Open Access Article
Open Access ArticleDirecting chiral induction in hollow helical organic nanotubes
Indradip
Mandal
 ,
Justin
Vouillamoz
,
Justin
Vouillamoz
 and
Andreas F. M.
Kilbinger
and
Andreas F. M.
Kilbinger
 *
*
Department of Chemistry, University of Fribourg, Chemin du Musée 9, 1700 Fribourg, Switzerland. E-mail: andreas.kilbinger@unifr.ch
First published on 26th January 2026
Abstract
Helical aromatic oligoamides were synthesized via a polymerization strategy from amino acid-derived monomers to probe side-chain steric effects on helical chirality. Circular dichroism revealed tunable, reversible helicity dictated by substituent bulk. This work establishes tubular oligoamides as structurally precise yet responsive foldamers, enabling controlled chiroptical activity through targeted side-chain engineering.
A helical architecture is a hallmark of structural organization in both natural and synthetic systems,1 enabling precise spatial arrangement of functional groups2 and often conferring unique chiroptical properties.3 Inspired by motifs in proteins, nucleic acids, and polysaccharides, chemists have developed foldamers, which are synthetic oligomers that adopt stable, predictable conformations and act as versatile platforms for chiral recognition,4 catalysis,5 molecular transport,6 and responsive materials.7 Among these, aromatic oligoamides have attracted sustained interest for their predictable folding, high stability, and modular synthetic design.
Tubular aromatic oligoamides developed by Gong and co-workers8,9 form a distinct class within this family. Built from meta-alkoxy-linked aromatic amide units reinforced by intramolecular three-centre hydrogen bonds, these rigid backbones fold into persistent cylindrical helices with internal cavities and tunable outer surfaces. Such tubular foldamers assemble into columnar stacks via π–π interactions,10 creating continuous channels for molecular encapsulation11 or transport.12 While structurally precise and robust, these helices have not been extensively explored for their chiroptical properties; despite their well-defined helicity, parent systems have rarely been examined for circular dichroism (CD) activity or systematic control of helical chirality.13
Other aromatic foldamer systems demonstrate that peripheral substituents can strongly influence helicity,14 self-assembly,15 and optical properties.16 Among them, quinoline-derived oligoamides, pioneered by Huc and co-workers, have been studied extensively for helical folding,17 chiral induction,18 and functional group organization along a rigid aromatic helix. In such systems, remote stereocenters,19 steric bias,20 and hydrogen-bond-mediated conformational control have been exploited to achieve absolute helical handedness and switchable chirality in solution and the solid state.21 Additionally, Zeng and co-workers22 have reported macrocyclic aromatic oligomers with specific functional end groups that stack into columnar assemblies, highlighting how noncovalent forces can control supramolecular helicity. Li23 and others24 have designed aromatic macrocycles25 and helices26 bearing hydrogen-bonding and π-stacking motifs that drive hierarchical organization into nanorods, fibrils, and other anisotropic structures. These studies illustrate a general principle: the periphery of a foldamer and its side chains or appended functionalities can be as critical as the backbone in dictating folding propensity, supramolecular organization, and emergent chiral properties.27
In helical aromatic foldamers, side chains can influence conformation through steric effects,28 electronic modulation of aromatic stacking, and secondary noncovalent interactions.29 Such influences can bias helix handedness,30 alter the helical pitch,31 and modulate aggregation, with potential to enable reversible chiral switching.32 However, a systematic investigation into how side-chain design affects the helicity and chiroptical signatures of “tubular” aromatic oligoamides, such as those developed by the Gong group, has not yet been reported.
Herein, we present a detailed study of side-chain-functionalized tubular aromatic foldamers that display measurable and tunable CD activity. In the absence of chiral induction, the helices typically exist as a racemic mixture of left- and right-handed forms. Incorporation of chiral side chains or chiral initiators can bias this equilibrium to favor one handedness, which is reflected in their chiroptical signals. By introducing chiral substituents of varied steric demand, we examine how peripheral modifications influence the magnitude of chiral induction. Helices bearing achiral side chains were biased toward a preferred helical sense by introducing a chiral initiating group into the backbone, exploiting the well-known ‘sergeant-and-soldiers’ effect. Variable-temperature CD spectroscopy reveals distinct side-chain-dependent trends, including reversible modulation of CD intensity. Structural analysis suggests these effects arise from the interplay between side-chain packing. This work demonstrates, for the first time, that Gong-type tubular foldamers can be rendered chiroptically active through targeted side-chain engineering. The resulting CD signals provide direct access to their helicity in solution, bridging the gap between structurally precise yet previously uncharacterized chiral-responsive foldamer systems.
A recently developed polymerization method33–35 was employed by our group to synthesize oligomers and polymers directly from aromatic amino acid monomers (Fig. 1), as it can produce presumed helical foldamers in a single step. The latest phosphine-based reagent, R (Fig. 1), was used to activate the carboxylic acid to the corresponding acid chloride in the presence of an amine under slow monomer addition conditions (Scheme 1). Initially, monomer A, bearing minimally sterically demanding methoxy groups, was polymerized using the chiral initiator I1 to yield PA1. Although the polymerization was targeted to form only a 16-mer (A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I1 = 16
I1 = 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
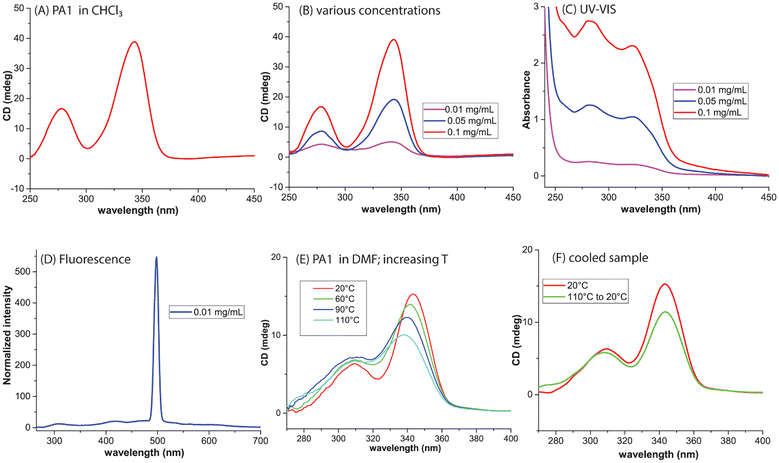
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), precipitation of the resulting polymer was observed. This limited the solubility and hindered subsequent characterization by size-exclusion chromatography (SEC) in DMF and via NMR spectroscopy. Fortunately, CD spectroscopy could be performed in both CHCl3 and DMF due to the low concentration needed for the measurements. In CHCl3, a positive Cotton effect was observed (Fig. 2A). Concentrations from 0.01 to 0.1 mg mL−1 showed a proportional increase in both CD (Fig. 2B) and absorbance intensities (Fig. 2C). Fluorescence measurements were also conducted in CHCl3 (0.01 mg mL−1) (Fig. 2D) using the excitation wavelength λ = 274 nm where the absorbance value was 0.1 AU (Fig. S). In DMF, a lower-intensity CD signal was detected, and heating the solution up to 110 °C caused a slight decrease in CD intensity (Fig. 2E). Upon cooling to 20 °C and equilibrating for one hour, the CD intensity recovered almost to that of a freshly prepared sample at 20 °C (Fig. 2F), indicating that heat can act as a stimulus for these chiral initiator-induced helices. To increase solubility and thereby to obtain control over the helix synthesis during polymerization, monomer B was prepared (see the SI) and initiated with I1 at a B
1), precipitation of the resulting polymer was observed. This limited the solubility and hindered subsequent characterization by size-exclusion chromatography (SEC) in DMF and via NMR spectroscopy. Fortunately, CD spectroscopy could be performed in both CHCl3 and DMF due to the low concentration needed for the measurements. In CHCl3, a positive Cotton effect was observed (Fig. 2A). Concentrations from 0.01 to 0.1 mg mL−1 showed a proportional increase in both CD (Fig. 2B) and absorbance intensities (Fig. 2C). Fluorescence measurements were also conducted in CHCl3 (0.01 mg mL−1) (Fig. 2D) using the excitation wavelength λ = 274 nm where the absorbance value was 0.1 AU (Fig. S). In DMF, a lower-intensity CD signal was detected, and heating the solution up to 110 °C caused a slight decrease in CD intensity (Fig. 2E). Upon cooling to 20 °C and equilibrating for one hour, the CD intensity recovered almost to that of a freshly prepared sample at 20 °C (Fig. 2F), indicating that heat can act as a stimulus for these chiral initiator-induced helices. To increase solubility and thereby to obtain control over the helix synthesis during polymerization, monomer B was prepared (see the SI) and initiated with I1 at a B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I1 ratio of 8
I1 ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. SEC analysis of the precipitated polymer PB1 (from cold methanol) in DMF showed a number-average molar mass (Mn) of 4.8 kDa (theoretical Mn = 5.3 kDa) with a dispersity of 1.09. CD spectra were recorded in a range of solvents at a fixed concentration (0.1 mg mL−1), including CHCl3, DMF, a CHCl3/methanol (1
1. SEC analysis of the precipitated polymer PB1 (from cold methanol) in DMF showed a number-average molar mass (Mn) of 4.8 kDa (theoretical Mn = 5.3 kDa) with a dispersity of 1.09. CD spectra were recorded in a range of solvents at a fixed concentration (0.1 mg mL−1), including CHCl3, DMF, a CHCl3/methanol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
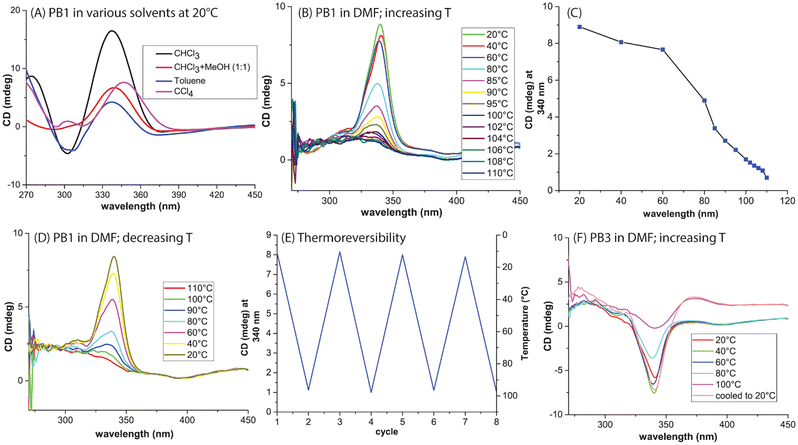
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture, and CCl4. In all cases, a positive Cotton effect was observed (Fig. 3A), though with varying intensities. CHCl3 showed the highest intensity signal among the solvents tested here. Temperature-dependent CD measurements were further performed in CHCl3 and DMF. Heating a chloroform solution to 40 °C did not alter the CD signal (Fig. S3) whereas increasing temperature showed a continuous decrease of the CD signal in DMF (Fig. 3B). Plotting of the CD absorption maximum (at 340 nm) vs. temperature yielded a cross-over point at around T = 80–85 °C which resembled a melting transition of PB1 in DMF (Fig. 3C). When the same solution was gradually cooled to 20 °C, the CD signal returned to almost its original value indicating a reversible process (Fig. 3D). This phenomenon indicates that PB1 is thermally stable yet dynamic. The polymer underwent reversible unfolding and refolding of its helices in response to temperature changes. This phenomenon should be similar in magnitude and transition temperature for similar helices. However, this scenario was less pronounced in the case of PA1 compared to PB1. We assume that the three center hydrogen bonding strength is of similar magnitude for PA1 and PB1, and in fact, all helices synthesized here. The enthalpic benefit from π-interactions between phenyl rings of adjacent helical turns, however, will be highly dependent on the helical pitch, which will be a function of steric bulk of the attached side chains.
1) mixture, and CCl4. In all cases, a positive Cotton effect was observed (Fig. 3A), though with varying intensities. CHCl3 showed the highest intensity signal among the solvents tested here. Temperature-dependent CD measurements were further performed in CHCl3 and DMF. Heating a chloroform solution to 40 °C did not alter the CD signal (Fig. S3) whereas increasing temperature showed a continuous decrease of the CD signal in DMF (Fig. 3B). Plotting of the CD absorption maximum (at 340 nm) vs. temperature yielded a cross-over point at around T = 80–85 °C which resembled a melting transition of PB1 in DMF (Fig. 3C). When the same solution was gradually cooled to 20 °C, the CD signal returned to almost its original value indicating a reversible process (Fig. 3D). This phenomenon indicates that PB1 is thermally stable yet dynamic. The polymer underwent reversible unfolding and refolding of its helices in response to temperature changes. This phenomenon should be similar in magnitude and transition temperature for similar helices. However, this scenario was less pronounced in the case of PA1 compared to PB1. We assume that the three center hydrogen bonding strength is of similar magnitude for PA1 and PB1, and in fact, all helices synthesized here. The enthalpic benefit from π-interactions between phenyl rings of adjacent helical turns, however, will be highly dependent on the helical pitch, which will be a function of steric bulk of the attached side chains.
 | ||
| Fig. 1 Chemical structures of the monomer (A–J), initiators (I1–I3) and the phosphine reagent (R) used in this study. | ||
It is, therefore, reasonable to assume that helices carrying bulkier side chains give rise to larger helical pitches. This in turn should decrease the strength of aromatic interactions and facilitate helical racemization. For chirally initiated helices in particular, a larger pitch will reduce the steric contact between the initiator group and the first “overlapping”, i.e. a helical monomer unit and hence weaken the chiral induction.
We, therefore, believe that the loss of the CD signal is due to a loss of chiral bias rather than proof of complete unfolding of the helix.
To understand the effect of side chain steric bulk on parameters, such as the helical pitch, we carried out simple molecular dynamics simulations using an implicit solvent model (Hawkins–Cramer–Truhlar, see the SI for details). Helices composed of 60 phenyl rings were simulated for all monomers A–I for 100 ns, with the last 50 ns used for averaging geometric parameters.
We calculated the helical pitches for poly-A (3.45 Å) and poly(B) (5.35 Å). This difference in pitch of almost 2 Å shows how polymers of these densely side-chain decorated monomer units are very susceptible to small steric effects. Therefore, assuming a larger pitch for PB1 compared to PA1, we can assume that weakened aromatic interactions in PB1 could be responsible for a more pronounced thermal response in CD-spectroscopy. To examine the effect of the helix length on helicity, PB2 was synthesized under the same conditions as PB1, except using a B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I1 ratio of 16
I1 ratio of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. A chloroform-soluble polymer was obtained; however, PB2 precipitated at the SEC characterization concentration (2 mg mL−1) in DMF, and SEC in chloroform yielded no elution, the reason for which remains unclear. Despite these challenges, CD analysis of PB2 revealed a Cotton effect similar to that of PB1, confirming the presence of helical folding in longer oligomers in solution (SI, Fig. S5). However, at the same mass concentration (0.1 mg mL−1) the CD intensity of PB2 was lower than that of PB1, indicating that the chiral bias does not propagate along the entire length of the helix for this type of monomer (Fig. S8). Moreover, PB1 exhibited a thermoreversible behaviour, with CD signals decreasing upon heating and largely recovering upon cooling for several cycles, indicating reversible helical folding (Fig. 3E). Initiator I2 was used to synthesize PB3 (I2
1. A chloroform-soluble polymer was obtained; however, PB2 precipitated at the SEC characterization concentration (2 mg mL−1) in DMF, and SEC in chloroform yielded no elution, the reason for which remains unclear. Despite these challenges, CD analysis of PB2 revealed a Cotton effect similar to that of PB1, confirming the presence of helical folding in longer oligomers in solution (SI, Fig. S5). However, at the same mass concentration (0.1 mg mL−1) the CD intensity of PB2 was lower than that of PB1, indicating that the chiral bias does not propagate along the entire length of the helix for this type of monomer (Fig. S8). Moreover, PB1 exhibited a thermoreversible behaviour, with CD signals decreasing upon heating and largely recovering upon cooling for several cycles, indicating reversible helical folding (Fig. 3E). Initiator I2 was used to synthesize PB3 (I2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B = 1
B = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8), which, as expected, showed a negative Cotton effect (Fig. 3F) and a similar thermal response during CD analysis.
8), which, as expected, showed a negative Cotton effect (Fig. 3F) and a similar thermal response during CD analysis.
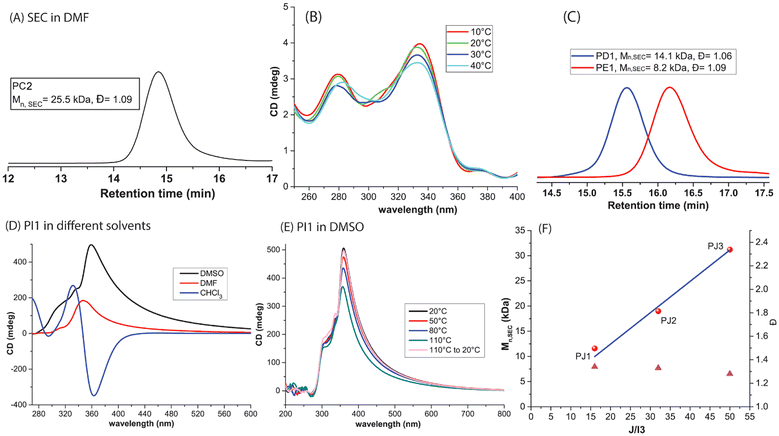
To improve the solubility of the helical polymers in both nonpolar (CHCl3 during synthesis) and polar (DMF for SEC analysis) solvents, a pentyl tert-butoxycarbonyl (BOC) side group was introduced into monomer C. Monomer C was polymerized using R and I1 under slow monomer addition (0.07 mL h−1) conditions. The resulting 20-mer, PC1, exhibited controlled Mn (11 kDa) and dispersity (1.08) in SEC (DMF). Subsequently, a 50-mer, PC2, was synthesized with similarly controlled Mn and dispersity (Fig. 4A). CD analysis of PC2 revealed a positive Cotton effect in CHCl3, although the signal intensity was comparatively weak (Fig. 4B). Heating a CHCl3 solution up to 40 °C did not result in any loss of CD signals. Despite the longer chain length, the foldamer retained its helical conformation, consistent with the classical sergeant-and-soldiers effect. MD simulations of a 60mer of poly(C) allowed the calculation of a pitch of 4.14 Å, which was much shorter than that of polymers of A and B. MD simulations show that attractive H-bonds amongst the side chain BOC urethane groups are responsible for a more compact and less sterically interfering periphery of the helix, which manifests in a smaller pitch and hence reduced thermoresponsive behavior in CD-measurements.
To investigate the effect of steric crowding on the solution-state helicity of these foldamers, ethylhexyl side chains were introduced to afford monomers D and E. The only difference between the two monomers is that D is racemic, whereas E is the enantiomerically pure (S)-isomer (see the SI). Mosher's ester derivatives were prepared from the synthesized pure (S) alcohol and the commercial racemic alcohol to prove the enantiomeric purity of the synthesized side groups. 19F and 13C NMR comparison of both esters unequivocally proved that the chiral (S)-2-ethylhexan-1-ol was indeed enantiomerically pure (see Fig. S12–S15). Monomer D was polymerized using I1 to target a 25-mer (PD1), which was well-characterized by SEC (Fig. 4C). As expected, PD1 did not exhibit any CD signals in CHCl3, CCl4, or DMF. A similar observation was made for PE1 (synthesized from E with I3, see the SI), which was striking given that each repeat unit contains two chiral side chains. MD simulations of a 60mer of poly(E) gave a pitch of 6.43 Å.
These results strengthen our hypothesis that steric side chain bulk affects the helical pitch, which in turn results in a loss of the ability to be chirally biased by the initiator or side chains.
Additionally, PD1 and PE1 showed good solubility in DMF and can be readily characterized by SEC, despite their inherent hydrophobicity. This observation aligns with our hypothesis that a larger helical pitch allows solvent molecules to interact with the exposed amide and phenyl groups, enhancing polymer solubility in DMF. In fact, the molecular dynamics simulated pitches (see the SI) can only be considered as minimum values, useful for comparing the effect of different substituents. We believe that the excellent solubility of PD1 and PE1 can only be explained by solvation of the helices, which would significantly expand their helical pitches if the three center H-bonds remain unbroken.
Next, a systematic study of the effect of steric groups on CD behavior was performed by designing monomers F–I. Monomer F has a slightly lower steric bulk than D and E; G contains alternating chiral branched and achiral straight alkyl groups; H bears a methoxy group on one side and a bulky (S)-configured ethylhexyl side chain on the other, whereas I has one methoxy group on one side and a chiral (S)-2-methylbutyl on the other. F, G, and H could all be polymerized in a controlled manner (see the SI). PF1 (pitch: 5.80 Å, see the SI) and PH1 (pitch: 5.10 Å, see the SI) were CD silent, whereas PG1 (pitch: 5.27 Å, see the SI) showed a very weak CD signal in CCl4 (Fig. S7 and S8).
Gratifyingly, PI1 (pitch: 4.90 Å, see the SI) showed a strong CD signal across multiple solvents and temperatures, including DMSO (Fig. 4D). In CHCl3, a bisignate signal was initially observed but disappeared when the solution was kept at room temperature overnight, likely due to aggregation-induced precipitation. In DMF and DMSO, PI1 consistently showed a positive Cotton effect, with the highest intensity in DMSO. Notably, the CD signal in polar, potentially H-bond-disrupting solvents persisted for extended periods without racemization. Heating the DMSO solution to 100 °C did not alter the CD signal much (Fig. 4E), which also remained stable at room temperature for over 45 days. Interestingly, unlike PB1, here, the CD intensity did not decrease to almost zero upon heating in either DMSO or DMF (Fig. S9). The dissymmetry factor (g-factor) of PI1 in DMSO at room temperature was calculated as 9.8 × 10−3 (see the SI). This high g-factor suggests significant chiral asymmetry in the electronic transitions of PI1 in a polar solvent such as DMSO.
The monomer structures of H and I only differ slightly in that the stereogenic center in H carries an ethyl and a butyl group, whereas the stereogenic center of I carries a methyl and an ethyl group. This slight difference in the steric bulk leads to a 0.2 Å (see MD simulations) larger helical pitch for H (5.10) compared to I (4.90) and complete loss of any helical bias in H. It is important to note that branched side chains, which are often used for increased solubility, can easily affect the helical folding if the steric bulk due to the branching group is too close to the helical core.
Due to strong aggregation, PI1 was insoluble for NMR or SEC analysis. To overcome this, a protection–deprotection strategy was employed. Monomer J was prepared in one step from I and polymerized to three different lengths (PJ1: 16-mer, PJ2:32-mer, and PJ3: 50-mer). A controlled polymerization could be achieved as shown by the linear relationship between observed Mn and monomer (J) to initiator (I3) ratio employed (Fig. 4F). As expected, PJ1 did not show any CD signal. Following trifluoroacetic acid treatment and trituration from methanol, the resulting polymer exhibited a sharp CD signal in CHCl3, confirming the pH-triggered helical folding of the polymer (Fig. S12). Fluorescence spectra of both protected and deprotected PJ1 revealed an additional emission peak at around 420 nm, which is likely attributable to folding of the helical backbone (Fig. S13).
Conclusions
In summary, we demonstrate that the helicity and chiroptical activity of tubular aromatic oligoamide foldamers can be precisely modulated through side-chain design and chain length. Minimally sterically demanding side chains yield strong, thermoresponsive CD signals, while bulky substituents suppress the signal, highlighting the critical role of steric effects and likely the helical pitch. Notably, the introduction of optimized side chains further enables stable CD responses, even in polar solvents and at elevated temperatures. These findings were further supported by molecular dynamics simulations of polymers constructed from all monomers. These results establish Gong-type meta-alkoxy-linked meta-aminobenzamide-based tubular foldamers as a versatile platform for controlling solution-state helicity and designing chiral-responsive materials through side-chain engineering. These results also suggest that in all applications of these polymers as tube-like structures or channels, the side-chains must be designed carefully in order not to widen the helical pitch beyond the point where aromatic interactions are lost.Author contributions
IM conceived the project. IM and JV synthesized the monomers. IM performed the polymerizations and analyses. AFMK supervised the research. All authors reviewed the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: figures, data, detailed experimental protocols and characterisation of new compounds. See DOI: https://doi.org/10.1039/d5ob01868d.Acknowledgements
IM, JV, and AFMK thank the Department of Chemistry, University of Fribourg, Switzerland, for support.References
- T. Leigh and P. Fernandez-Trillo, Nat. Rev. Chem., 2020, 4, 291 Search PubMed.
- M. Zwillinger, P. S. Reddy, B. Wicher, P. K. Mandal, M. Csékei, L. Fischer, A. Kotschy and I. Huc, Chem. – Eur. J., 2020, 26, 17366 Search PubMed.
- E. Yashima, K. Maeda, H. Iida, Y. Furusho and K. Nagai, Chem. Rev., 2009, 109, 6102 CrossRef CAS PubMed.
- T. Ikai, T. Matsumoto, S. Takeda, K. Oki and E. Yashima, Macromolecules, 2025, 58, 6943 Search PubMed.
- L. Zhou, K. He, N. Liu and Z.-Q. Wu, Polym. Chem., 2022, 13, 3967 Search PubMed.
- S. Farooq, J. A. Malla, M. Nedyalkova, R. V. M. Freire, I. Mandal, A. Crochet, S. Salentinig, M. Lattuada, C. T. McTernan and A. F. M. Kilbinger, Angew. Chem., Int. Ed., 2025, 64, e202504170 Search PubMed.
- M. Lago-Silva, M. Fernández-Míguez, R. Rodríguez, E. Quiñoá and F. Freire, Chem. Soc. Rev., 2024, 53, 793 Search PubMed.
- B. Gong, Acc. Chem. Res., 2008, 41, 1376 CrossRef CAS PubMed.
- T. A. Sobiech, Y. Zhong and B. Gong, Org. Biomol. Chem., 2022, 20, 6962 RSC.
- X. Wei, G. Zhang, Y. Shen, Y. Zhong, R. Liu, N. Yang, F. Y. Al-mkhaizim, M. A. Kline, L. He, M. Li, Z.-L. Lu, Z. Shao and B. Gong, J. Am. Chem. Soc., 2016, 138, 2749 Search PubMed.
- Y. Zhong, T. A. Sobiech, B. Kauffmann, B. Song, X. Li, Y. Ferrand, I. Huc and B. Gong, Chem. Sci., 2023, 14, 4759 RSC.
- A. J. Helsel, A. L. Brown, K. Yamato, W. Feng, L. Yuan, A. J. Clements, S. V. Harding, G. Szabo, Z. Shao and B. Gong, J. Am. Chem. Soc., 2008, 130, 15784 CrossRef CAS PubMed.
- J. Cao, M. Kline, Z. Chen, B. Luan, M. Lv, W. Zhang, C. Lian, Q. Wang, Q. Huang, X. Wei, J. Deng, J. Zhu and B. Gong, Chem. Commun., 2012, 48, 11112 Search PubMed.
- K. Kuyama, F. Takeda, A. Ikeda, K. Tanaka, K. Katagiri, M. Kawahata, Y. Okada, N. Kobayashi, H. Kagechika and A. Tanatani, Org. Biomol. Chem., 2025, 23, 4927 Search PubMed.
- Z. Pang, T. Qi and B. Li, Org. Biomol. Chem., 2021, 19, 5555 RSC.
- V. Laffilé, K. Moreno, E. Merlet, N. McClenaghan, Y. Ferrand and C. Olivier, Org. Biomol. Chem., 2023, 21, 3644 Search PubMed.
- S. Wang, J. Sigl, L. Allmendinger, V. Maurizot and I. Huc, Chem. Sci., 2025, 16, 1136 Search PubMed.
- H. Jiang, C. Dolain, J.-M. Léger, H. Gornitzka and I. Huc, J. Am. Chem. Soc., 2004, 126, 1034 Search PubMed.
- V. Maurizot, C. Dolain and I. Huc, Eur. J. Org. Chem., 2005, 2005, 1293 CrossRef.
- A. M. Kendhale, L. Poniman, Z. Dong, K. Laxmi-Reddy, B. Kauffmann, Y. Ferrand and I. Huc, J. Org. Chem., 2011, 76, 195 Search PubMed.
- T. Qi, V. Maurizot, H. Noguchi, T. Charoenraks, B. Kauffmann, M. Takafuji, H. Ihara and I. Huc, Chem. Commun., 2012, 48, 6337 RSC.
- H. Zhao, W. Q. Ong, F. Zhou, X. Fang, X. Chen, S. F. Y. Li, H. Su, N.-J. Cho and H. Zeng, Chem. Sci., 2012, 3, 2042 Search PubMed.
- D. Zhang, W. Wang and Z. Li, Chem. Rec., 2015, 15, 233 Search PubMed.
- K. Kreger, F. Behrendt and J. C. Brendel, J. Polym. Sci., 2025, 63, 4450–4465 CrossRef CAS.
- C. Ren, V. Maurizot, H. Zhao, J. Shen, F. Zhou, W. Q. Ong, Z. Du, K. Zhang, H. Su and H. Zeng, J. Am. Chem. Soc., 2011, 133, 13930 Search PubMed.
- S. Koppireddi, C.-Z. Liu, H. Wang, D.-W. Zhang and Z.-T. Li, CrystEngComm, 2019, 21, 2626 RSC.
- T. Ikai, K. Nakamura, K. Mizumoto, K. Oki and E. Yashima, Angew. Chem., Int. Ed., 2023, 62, e202301127 Search PubMed.
- A. Tanatani, A. Yokoyama, I. Azumaya, Y. Takakura, C. Mitsui, M. Shiro, M. Uchiyama, A. Muranaka, N. Kobayashi and T. Yokozawa, J. Am. Chem. Soc., 2005, 127, 8553 CrossRef CAS PubMed.
- S. E. Miller, A. M. Watkins, N. R. Kallenbach and P. S. Arora, Proc. Natl. Acad. Sci. U. S. A., 2014, 111, 6636 Search PubMed.
- T. Ikai, Y. Morita, T. Majima, S. Takeda, R. Ishidate, K. Oki, N. Suzuki, H. Ohtani, H. Aoi, K. Maeda, K. Okoshi and E. Yashima, J. Am. Chem. Soc., 2023, 145, 24862 Search PubMed.
- A. Yokoyama, T. Saiki, H. Masu, I. Azumaya and T. Yokozawa, Polymer, 2018, 134, 175 Search PubMed.
- S. Kobayashi, K. Morino and E. Yashima, Chem. Commun., 2007, 2351 RSC.
- S. Pal, D. P. T. Nguyen, A. Molliet, M. Alizadeh, A. Crochet, R. D. Ortuso, A. Petri-Fink and A. F. M. Kilbinger, Nat. Chem., 2021, 13, 705 Search PubMed.
- S. Pal, L. Hong, R. V. M. Freire, S. Farooq, S. Salentinig and A. F. M. Kilbinger, Macromolecules, 2023, 56, 7984 CrossRef CAS.
- D. P. T. Nguyen, S. Farooq, N. Meyer and A. F. M. Kilbinger, Polym. Chem., 2025, 16, 2639 RSC.
| This journal is © The Royal Society of Chemistry 2026 |