Open Access Article
Open Access ArticleTransition-metal-free/boric acid catalysed mechanochemical synthesis of symmetrical and unsymmetrical azobenzenes: a sustainable approach
Tandra
Kundu
a,
Saptadwipa
Bhattacharjee
a,
Gyan
Chandra Pariyar
b,
Bijeta
Mitra
a and
Pranab
Ghosh
 *a
*a
aDepartment of Chemistry, University of North Bengal, Dist. Darjeeling, West Bengal, India. E-mail: pizy12@yahoo.com
bDepartment of Food Technology, University of North Bengal, Dist. Darjeeling, West Bengal, India. Fax: +91 (0353) 2699001; Tel: +91 (0353) 2776381
First published on 8th January 2026
Abstract
A mechanochemical protocol employing boric acid, an economical and environmentally benign inorganic acid catalyst, enables the synthesis of symmetrical and unsymmetrical azobenzenes from diverse amines. This grinding-based, solvent-free approach provides a sustainable alternative to conventional methods, avoiding harsh transition-metal catalysts. The use of oxone as a green oxidant, operation under ambient conditions, simplified work-up, and overall environmental compatibility highlight the advantages of this methodology.
Introduction
In recent years, sustainable development has become a central focus of modern organic synthesis. The urgent need to address global pollution and the ongoing environmental damage has stimulated the search for alternative strategies in organic synthesis.1 Conventional methods often rely on petroleum-based solvents, hazardous chemicals, and harsh reaction conditions, all of which contribute significantly to environmental pollution. To overcome these challenges, alternative strategies that minimize waste, reduce energy consumption, and eliminate toxic reagents are urgently needed. Mechanochemistry has emerged as a powerful green approach, offering solvent-free and energy-efficient pathways for the synthesis of organic molecules. By replacing traditional solvent-intensive protocols with grinding-based methodologies, mechanochemical synthesis provides an environmentally sustainable alternative that is particularly attractive for both organic and pharmaceutical chemistry.For chemists, the search for greener and eco-friendly catalysts and solvents remains both a challenge and an opportunity in the development of sustainable reaction strategies. Among various targets, aromatic azo-compounds have long attracted significant attention due to their wide range of applications in food additives, indicators, organic dyes, and therapeutic agents (Fig. 1).2,3 Owing to their remarkable photochemical properties, these compounds have also been widely employed in liquid crystals,4,5 smart polymers,6 photoswitches for biological systems,7,8 and photochromic ligands in optochemical genetics.9 More recently, through C–H activation or functionalization, azo compounds have been exploited for the synthesis of valuable derivatives such as o-alkoxyazobenzenes,10o-acylazobenzenes,11–14 and indole frameworks.15,16 In addition, they are increasingly utilized in the fabrication of optical filters and protective glass materials.
Literature surveys indicate that several methods have been reported for the synthesis of azo compounds, including the Mills17,18 and Wallach reactions, oxidation of amines, reduction of azobenzenes, thermolysis of azides, transformations of quinine acetals, dehydrogenation or metal-catalyzed coupling of arylhydrazines, triazene rearrangements, dimerization of diazonium salts, and opening of benzotriazoles.19–26 Among these, the simplest and most direct strategy is the one-step oxidation of amines. This transformation has been achieved using various catalysts such as gold nanoparticles on TiO2, silver nanoparticles, nickel peroxide, MnO2, NaBO3, BaMnO4, AgMnO4, Pb(OAc)4, Ag2CO3, Ce(OH)3O2H, RuCl3/H2O2, t-BuOI, t-BuOCl, galvinoxyl/K3Fe(CN)6, KO2, platinum and palladium nanowires, and Cu(I)-diaziridinone.27–46 A comparative overview of these reported processes is provided in Table 2.
Several methods have been reported for the synthesis of azobenzenes, each with certain advantages, such as high yields for specific amine derivatives. However, many of these protocols remain limited due to drawbacks including the use of expensive metal catalysts, toxic solvents, harsh conditions, long reaction times, moderate yields, and issues of functional group tolerance or metal contamination. The preparation and handling of metal catalysts, magnetic nanoparticles, or ionic liquids further increase cost and complexity. In contrast, mechanochemical synthesis offers a green, solvent-free, and energy-efficient alternative.47 It enables straightforward one-pot access to symmetrical and unsymmetrical azobenzenes under ambient conditions, without the need for transition-metal or lanthanide catalysts, thereby providing a more sustainable and eco-efficient pathway highly desirable in synthetic organic chemistry.
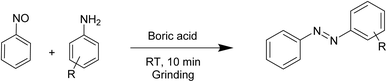
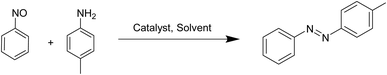
Among inorganic acids, boric acid is a weak acid (pKa = 9.2), moderately soluble in water, more soluble in warm water, and well known for its stability and environmental safety. Over the past decades, it has been widely applied in organic transformations48–52 due to its advantages, including commercial availability, chemical stability, and ease of handling, low cost, non-toxicity, and overall green nature. In this study, we explored the catalytic potential of boric acid in a mechanochemical framework for the sustainable synthesis of azobenzenes. We report a straightforward, solvent-free grinding protocol under mild conditions for the preparation of both symmetrical (Scheme 1) and unsymmetrical azobenzenes (Scheme 2), employing oxone53 as the oxidant in the synthesis of symmetric derivatives.
Results and discussion
Synthesis of symmetrical azobenzenes
To standardize the reaction protocol, aniline (1 mmol) was taken as the starting material and the progress of the reaction was monitored by TLC. Initially, the reaction was investigated in the absence of a catalyst under solvent conditions. Even after 30 min of grinding the desired product was not obtained (Table 1, Entry 1). In the absence of boric acid, only nitrobenzene was formed in the presence of oxone and it was confirmed by matching the TLC results with those of commercially purchased nitrobenzene (Table 1, Entry 1). Then the reaction was carried out by taking 1 mmol of aniline, 25 mol% of boric acid and 0.5 mmol of oxone under aqueous conditions for 30 min when only a small amount of the product was obtained (Table 1, Entry 2). Now under the same reaction conditions, different solvents were used to monitor the reaction. However, with ethanol, methanol, acetonitrile, chloroform, and toluene we obtained moderate yields of the desired products (Table 1, Entries 3–7). When we switched from different solvents to neat conditions in the presence of varying amounts of boric acid, we were excited to find that the desired product was obtained in good yields (82–88%) (Table 1, Entries 8–12). Now with the solvent being optimized, we increased the amount of oxone to 1.0 mmol but were surprised to find that the yield decreased considerably to 66% (Table 1, Entry 13). It might be due to the oxidation of aniline to nitrobenzene before undergoing the coupling reaction. However, reducing the amount of oxone to 0.25 mmol reduced the yield to 45% (Table 1, Entry 14). To explore the effect of the oxidant, we screened our reaction with various oxidants such as CH3COOOH, K2S2O8 and I2. However, either trace amounts of the product were obtained or no reaction was observed (Table 1, Entries 18–20). Thus, we chose oxone as the oxidant for this reaction. We further investigated the reaction by increasing the time to 15, 20, and 30 min which did not give any significant increase in the yield of the product (Table 1, Entries 15, 16, and 17). Hence finally we chose 1 mmol of aniline, 10 mol% of boric acid and 0.5 mmol of oxone54 as the optimal reaction conditions for the synthesis of symmetric azobenzenes under solvent-free conditions at room temperature for 10 min using the grinding method (Table 1, Entry 12). Therefore, boric acid shows great catalytic activity in the preparation of symmetric azobenzene by the mechanochemical process.| Entry | Oxone (mmol) | Solvent | Catalyst loading (mol%) | Time (min) | Yield (%)c |
|---|---|---|---|---|---|
| a Bold font represents the optimized conditions. b Reaction of aniline (1 mmol), varying amount of oxone and boric acid with various solvents at room temperature using the grinding method. c Isolated yield of product by column chromatography. d CH3COOOH as the oxidant. e K2S2O8 as the oxidant. f I2 as the oxidant. | |||||
| 1 | 0.5 | Neat | — | 30 | — |
| 2 | 0.5 | Water | 25 | 30 | 35 |
| 3 | 0.5 | Ethanol | 25 | 30 | 39 |
| 4 | 0.5 | Methanol | 25 | 30 | 41 |
| 5 | 0.5 | Acetonitrile | 25 | 30 | 50 |
| 6 | 0.5 | CHCl3 | 25 | 30 | 56 |
| 7 | 0.5 | Toluene | 25 | 30 | 60 |
| 8 | 0.5 | Neat | 25 | 30 | 88 |
| 9 | 0.5 | Neat | 30 | 30 | 87 |
| 10 | 0.5 | Neat | 20 | 20 | 85 |
| 11 | 0.5 | Neat | 15 | 10 | 82 |
| 12 | 0.5 | Neat | 10 | 10 | 82 |
| 13 | 1.0 | Neat | 10 | 10 | 66 |
| 14 | 0.25 | Neat | 10 | 10 | 45 |
| 15 | 0.5 | Neat | 10 | 15 | 82 |
| 16 | 0.5 | Neat | 10 | 20 | 84 |
| 17 | 0.5 | Neat | 10 | 30 | 84 |
| 18 | 0.5 | Neat | 10 | 30 | Traced |
| 19 | 0.5 | Neat | 10 | 30 | —e |
| 20 | 0.5 | Neat | 10 | 30 | —f |
After tuning various reaction conditions, we compared our results with the previously published literature (Table 2) to demonstrate the benefits of using boric acid as a catalyst in this procedure. The results show that previously published techniques had flaws including prolonged response times, low yields, severe reaction conditions, and so on. Compared to previously reported approaches, the suggested procedures are more efficient, cost-effective, and environmentally friendly.
| Sl no. | Catalyst | Conditions | Time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | Ag-nanoparticles (1 mmol) | KOH, DMSO, rt | 24 | 50–97 |
| 2 | Au/TiO2-nanoparticles (1.5 wt%) | Toluene, 100 °C, O2, 5 bar | 5–31 | 44–99 |
| 3 | t-BuOCl (1 mmol) | NaI (1 mmol), ether, rt | 1–36 | 44–97% |
| 4 | CuBr (5 mol%) | Diaziridinone (1.1 equiv.), CH3CN, rt | 2 | 70–98% |
| 5 | NaBO3 (0.01 mol) | Glacial acetic acid (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cc), 45–50 °C cc), 45–50 °C |
3 | 6.52–50.42% |
| Present work | Boric acid (10 mol%) | Oxone (0.5 mmol), neat, rt, grinding | 10 min | 62–82% |
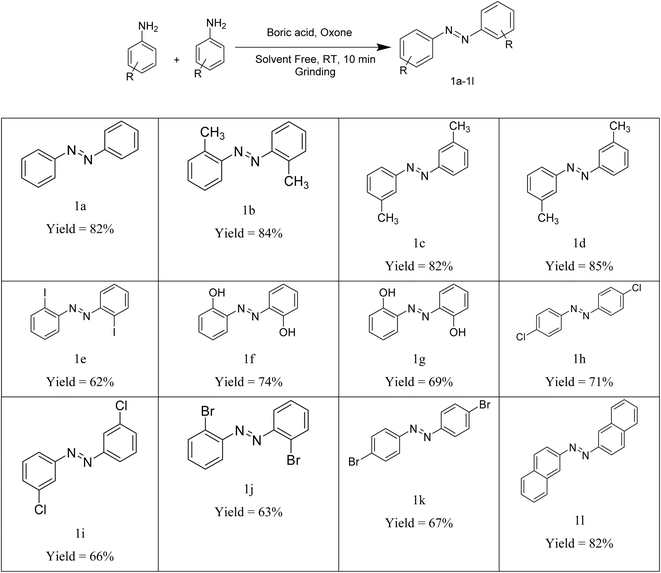
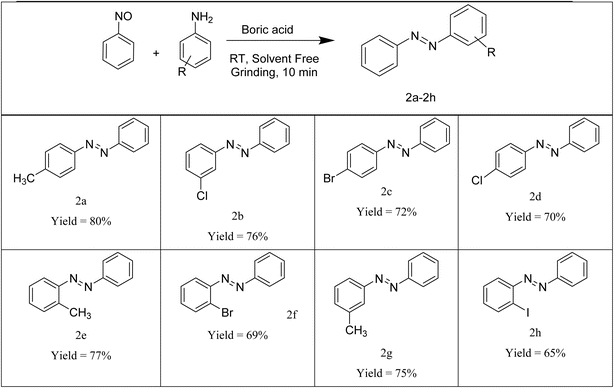
This protocol gave the desired products with a range of amines (Table 3). The progress of the reaction was monitored by TLC (eluent: ethyl acetate–petroleum ether). The product was collected through column chromatography. A number of amines were employed in the synthesis of symmetric azobenzenes in our methodology. As evidenced from Table 3, good yields of different products were obtained all at room temperature. The procedure was generalized for a range of amines having electron-donating and electron-withdrawing groups on the benzene ring as well as naphthyl rings. Better results were obtained for amines having the electron-donating effect compared to those having the electron-withdrawing effect. It may be due to the electron density on the nitrogen atom making them more nucleophilic. It was also observed that ortho-products gave fewer yields than the other products. This may be due to steric factors at the ortho positions.
| a Reaction conditions: aniline (1 mmol), oxone (0.5 mmol) and boric acid (10 mol%) under solvent-free conditions at room temperature using the grinding method. |
|---|
|
Synthesis of unsymmetrical azobenzenes
Now to broaden the catalytic activity, boric acid was also investigated for the synthesis of unsymmetrical azobenzenes. In our attempt to synthesize unsymmetric azobenzenes directly from amines, we chose aniline (1 mmol), p-toluidine (1 mmol), and oxone (0.5 mmol) as model substrates which gave only trace amounts of the desired product in the presence of 25 mol% of boric acid, with nitrobenzene as the main by-product. It was also confirmed by TLC using the commercially available one. Then to synthesise unsymmetrical azobenzenes we chose nitrosobenzene rather than aniline under the same conditions. At first, in the absence of a catalyst no desired product was obtained (Table 4, Entry 1). However, under the solvent-free mechanochemical conditions, the reaction of p-toluidine (1 mmol) with nitrosobenzene (1 mmol) in the presence of boric acid afforded the best yield of the targeted azobenzene (Table 4, Entries 8–12). Screening of solvents such as water, ethanol, methanol, chloroform, and acetonitrile gave only moderate results, confirming the superiority of the neat grinding method (Table 4, Entries 2–7). The yield of the product was not much affected even after increasing the time of the reaction (Table 4, Entries 13–15). Thus, the optimal protocol involved 1 mmol p-toluidine, 1 mmol nitrosobenzene, and 10 mol% of boric acid using the grinding process for 10 min, providing excellent yield of the unsymmetric azobenzene at room temperature (Table 4, Entry 12).| Entry | Solvent | Catalyst loading (mol%) | Time (min) | Yield (%)c |
|---|---|---|---|---|
| a Bold font represents the optimized conditions. b Reaction of amines (1 mmol), nitrosobenzene (1 mmol) and varying amount of boric acid at room temperature under different solvent conditions. c Isolated yield of the products using column chromatography. | ||||
| 1 | Neat | — | 30 | — |
| 2 | Water | 25 | 30 | 33 |
| 3 | Ethanol | 25 | 30 | 40 |
| 4 | Methanol | 25 | 30 | 47 |
| 5 | Acetonitrile | 25 | 30 | 56 |
| 6 | CHCl3 | 25 | 30 | 63 |
| 7 | Toluene | 25 | 30 | 69 |
| 8 | Neat | 25 | 20 | 86 |
| 9 | Neat | 30 | 20 | 86 |
| 10 | Neat | 20 | 15 | 84 |
| 11 | Neat | 15 | 10 | 82 |
| 12 | Neat | 10 | 10 | 80 |
| 13 | Neat | 10 | 15 | 80 |
| 14 | Neat | 10 | 20 | 81 |
| 15 | Neat | 10 | 30 | 82 |
Amines with diverse substituents were efficiently transformed into unsymmetric azobenzenes in good yields, as shown in Table 5. Both ortho- and para-substituted amines were compatible with the methodology, though para-substituted derivatives generally gave higher yields. The lower yields observed with ortho-substituted amines can be attributed to steric hindrance at the ortho position.
| a Reaction conditions: nitrosobenzene (1 mmol), amines (1 mmol) and boric acid (10 mol%) under neat conditions at room temperature using the grinding method. |
|---|
|
Table 6 shows the turnover frequency (TOF) and turnover number (TON). Boric acid can be used to attain higher turnover frequency (TOF) and turnover number (TON) values for both the symmetrical and unsymmetrical azobenzene synthesis under optimal circumstances.55
| Entry | Product | TON | TOF | Entry | Product | TON | TOF |
|---|---|---|---|---|---|---|---|
| 1 | 1a | 114 | 11.4 | 11 | 1k | 76 | 7.6 |
| 2 | 1b | 119 | 11.9 | 12 | 1l | 113 | 11.3 |
| 3 | 1c | 114 | 11.4 | 13 | 2a | 99 | 9.9 |
| 4 | 1d | 144 | 14.4 | 14 | 2b | 89 | 8.9 |
| 5 | 1e | 65 | 6.5 | 15 | 2c | 80 | 8.0 |
| 6 | 1f | 92 | 9.2 | 16 | 2d | 77 | 7.7 |
| 7 | 1g | 81 | 8.1 | 17 | 2e | 93 | 9.3 |
| 8 | 1h | 85 | 8.5 | 18 | 2f | 74 | 7.4 |
| 9 | 1i | 72 | 7.2 | 19 | 2g | 88 | 8.8 |
| 10 | 1j | 67 | 6.7 | 20 | 2h | 66 | 6.6 |
We studied the recyclability and reusability of the catalyst H3BO3 for the synthesis of symmetric and unsymmetric azobenzenes. It was found that after completion of the reaction, n-hexane was added to the residual mixture and then by simple filtration the catalyst was recovered and reused up to five times under the optimal conditions. After yielding an excellent amount of product in the 1st run, the catalyst was separated and collected to be used for the next run. In the 2nd run, the recovered catalyst provided the desired product in good yield (77%). The same process was just repeated for the 3rd, 4th and 5th run. We were glad to find that the catalytic activity of boric acid was retained up to the 5th run providing the desired product in good yields (58–82%) (Fig. 2).
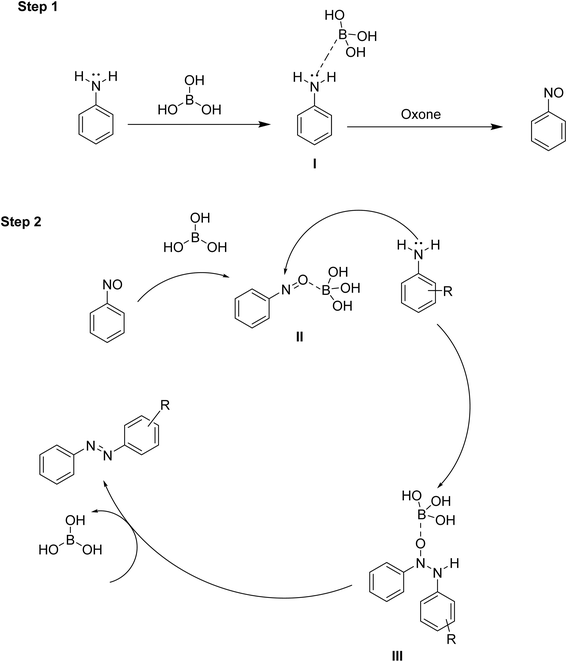
Mechanism
Based on the literature survey, a plausible mechanism has been proposed in Scheme 3. In the first step, boric acid coordinates with the lone pair of the amine nitrogen (I) through its electron-deficient boron centre. This coordination reduces the reactivity of nitrogen and directs the oxidation by oxone towards the formation of nitrosobenzene (detected by FT-IR spectra) rather than nitrobenzene. In the second step, boric acid may interact with the oxygen atom of nitrosobenzene (II), thereby activating it for nucleophilic attack by aniline at the nitrogen center. This leads to the formation of intermediate (III), which subsequently transforms into the targeted product, azobenzene. The first step of the mechanism is suitable only for the synthesis of symmetrical azobenzenes, whereas the second one is applicable for the synthesis of both symmetrical and unsymmetrical azobenzenes.Experimental details
The NMR data of (E)-azobenzene,56 the desired product of the model reaction, are given below: 1H NMR (300 M Hz, CDCl3) δ(ppm): 7.54–7.49 (m, 6H), 7.93 (d, J = 7.2 Hz, 4H); 13C NMR (75 M Hz, CDCl3) δ(ppm): 122.84, 129.10, 131.00, 152.62.
The NMR data of (E)-1-(4-methylphenyl)-2-phenyldiazene,57 the desired product of the model reaction, are given below: 1H NMR (400 M Hz, CDCl3) δ(ppm): 2.44 (s, 3H), 7.3 (d, J = 8.0 Hz, 2H), 7.5 (d, J = 8.0 Hz, 3H), 7.8 (d, J = 8.0, 2H), 7.9 (d, J = 7.6, 2H); 13C NMR (100 M Hz, CDCl3) δ(ppm): 22.69, 122.72, 122.85, 129.05, 129.74, 130.69, 141.56, 150.77, 152.74.
Conclusion
In conclusion, we have developed a simple, eco-friendly, and efficient mechanochemical strategy for the synthesis of both symmetrical and unsymmetrical azobenzenes. The use of inexpensive and non-toxic boric acid as a recoverable catalyst makes this method sustainable and environmentally benign. The process features easy reaction conditions and a straightforward setup, and eliminates the need for hazardous solvents. Overall, this work presents a facile and green approach for azobenzene synthesis, aligning with the principles of sustainable organic chemistry.Conflicts of interest
There are no conflicts to declare.Data availability
All the data put/used in writing the manuscript may be made available on request following the journal's policy.Supplementary information (SI): experimental details, 1H NMR and 13C NMR spectra of the synthesized compounds. See DOI: https://doi.org/10.1039/d5mr00133a.
Acknowledgements
The authors thank the Department of Chemistry, University of North Bengal for providing infrastructural support to carry out the laboratory work. One of the authors (S. B.) is thankful to DST/INSPIRE, New Delhi for financial support.References
- M. C. Bryan, B. Dillon, L. G. Hamann, G. J. Hughes, M. E. Kopach, E. A. Peterson, M. Pourashraf, I. Raheem, P. Richardson, D. Richter and H. F. Sneddon, J. Med. Chem., 2013, 56, 6007 Search PubMed.
- H. Böttcher, Zeitschrift für Chemie, 1989, 29, 387 Search PubMed.
- R. G. Anderson and G. Nickless, Anal., 1967, 92, 207 RSC.
- T. Ikeda, J. Mater. Chem., 2003, 13, 2037 RSC.
- C. J. Barrett, J. Mamiya, K. G. Yager and T. Ikeda, Soft Matter, 2007, 3, 1249 Search PubMed.
- G. S. Kumar and D. C. Neckers, Chem. Rev., 1989, 89, 1915 Search PubMed.
- A. A. Beharry and G. A. Woolley, Chem. Soc. Rev., 2011, 40, 4422 RSC.
- W. Jones, Solid State Ionics, 1995, 76, 173 CrossRef CAS.
- T. Fehrentz, M. Schönberger and D. Trauner, Angew. Chem., Int. Ed., 2011, 50, 12156 CrossRef CAS PubMed.
- Z. Yin, X. Jiang and P. Sun, J. Org. Chem., 2013, 78, 10002 CrossRef CAS PubMed.
- H. Li, P. Li, H. Tan and L. Wang, Chem. Eur. J., 2013, 19, 14432 CrossRef CAS PubMed.
- Z.-Y. Li, D.-D. Li and G.-W. Wang, J. Org. Chem., 2013, 78, 10414 CrossRef CAS PubMed.
- H. Song, D. Chen, C. Pi, X. Cui and Y. Wu, J. Org. Chem., 2014, 79, 2955 CrossRef CAS PubMed.
- F. Xiong, C. Qian, D. Lin, W. Zeng and X. Lu, Org. Lett., 2013, 15, 5444 Search PubMed.
- H. Li, P. Li and L. Wang, Org. Lett., 2013, 15, 620 CrossRef CAS PubMed.
- H. Wang, Y. Yu, X. Hong, Q. Tan and B. Xu, J. Org. Chem., 2014, 79, 3279 CrossRef CAS PubMed.
- C. Mills, J. Chem. Soc., Trans., 1895, 67, 925 RSC.
- A. Baeyer, Ber. Dtsch. Chem. Ges., 1874, 7, 1638 Search PubMed.
- E. Merino, Chem. Soc. Rev., 2011, 40, 3835 RSC.
- K. Haghbeen and E. W. Tan, J. Org. Chem., 1998, 63, 4503 Search PubMed.
- M. H. Davey, V. Y. Lee, R. D. Miller and T. J. Marks, J. Org. Chem., 1999, 64, 4976 Search PubMed.
- H. S. Fry and P. E. Bowman, J. Am. Chem. Soc., 1930, 52, 1531 Search PubMed.
- J. F. Vozza, J. Org. Chem., 1969, 34, 3219 Search PubMed.
- L. I. Smith and W. B. Irwin, J. Am. Chem. Soc., 1941, 63, 1036 CrossRef CAS.
- A. Thakuri, M. Banerjee and A. Chatterjee, iScience, 2022, 25, 104497 CrossRef CAS PubMed.
- J. Shen, J. Xu, Q. Zhu and P. Zhang, Org. Biomol. Chem., 2021, 19, 3119 RSC.
- S. Cai, H. Rong, X. Yu, X. Liu, D. Wang, W. He and Y. Li, ACS Catal., 2013, 3, 478 CrossRef CAS.
- A. Grirrane, A. Corma and H. García, Science, 2008, 322, 1661 CrossRef CAS PubMed.
- Y. Zhu and Y. Shi, Org. Lett., 2013, 15, 1942 CrossRef CAS PubMed.
- K. Nakagawa, S. Shiba, M. Horikawa, K. Sato, H. Nakamura, N. Harada and F. Harada, Synth. Commun., 1980, 10, 305 CrossRef CAS.
- H. K. Hombrecher and K. Lüdtke, Tetrahedron, 1993, 49, 9489 CrossRef CAS.
- S. M. Mehta and M. V. Vakilwala, J. Am. Chem. Soc., 1952, 74, 563 CrossRef CAS.
- E. Baer and A. L. Tosoni, J. Am. Chem. Soc., 1956, 78, 2857 CrossRef CAS.
- H. Firouzabadi and Z. Mostafavipoor, Bull. Chem. Soc. Jpn., 1983, 56, 914 CrossRef CAS.
- H. Firouzabadi, B. Vessal and M. Naderi, Tetrahedron Lett., 1982, 23, 1847–1850 CrossRef CAS.
- M. Hedayatullah, J. Pierre Dechatre and L. Denivelle, Tetrahedron Lett., 1975, 16, 2039 CrossRef.
- H. Firouzabadi and N. Iranpoor, Synth. Commun., 1984, 14, 875 Search PubMed.
- G. Crank and M. Makin, Aust. J. Chem., 1984, 37, 845 CrossRef CAS.
- X.-Y. Wang, Y.-L. Wang, J.-P. Li, Z.-F. Duanb and Z.-Y. Zhang, Synth. Commun., 1999, 29, 2271 Search PubMed.
- S. Okumura, C.-H. Lin, Y. Takeda and S. Minakata, J. Org. Chem., 2013, 78, 12090 Search PubMed.
- S. Minakata, Y. Takeda and S. Okumura, Synthesis, 2013, 45, 1029 Search PubMed.
- D. Zhang, J. Mcleod, L. Hu, S. Lu, Y. Ma, J. Zhong, Z. Jiang, H. Gu and X. Sun, J. Phys. Chem. C, 2016, 120, 14712 Search PubMed.
- B. Ortiz, P. Villanueva and F. Walls, J. Org. Chem., 1972, 37, 2748 Search PubMed.
- K. Seth, S. R. Roy, A. Kumar and A. K. Chakraborti, Catal. Sci. Technol., 2016, 6, 2892 Search PubMed.
- G. Barak and Y. Sasson, J. Org. Chem., 1989, 54, 3484 CrossRef CAS.
- K. Orito, T. Hatakeyama, M. Takeo, S. Uchiito, M. Tokuda and H. Suginome, Tetrahedron, 1998, 54, 8403 Search PubMed.
- G. W. Wang, Chem. Soc. Rev., 2013, 42, 7668 RSC.
- H. Kiyani and F. Ghorbani, Res. Chem. Intermed., 2013, 41, 2653 Search PubMed.
- T. A. Houston, B. L. Wilkinson and J. T. Blanchfield, Org. Lett., 2004, 6, 679 Search PubMed.
- R. K. Mylavarapu, K. Gcm, N. Kolla, R. Veeramalla, P. Koilkonda, A. Bhattacharya and R. Bandichhor, Org. Process Res. Dev., 2007, 11, 1065 Search PubMed.
- Z. Hosseinzadeh, A. Ramazani, N. Razzaghi-Asl, K. Slepokura and T. Lis, Turk. J. Chem., 2019, 43, 464 Search PubMed.
- T. B. Nguyen, J. Sorres, M. Q. Tran, L. Ermolenko and A. Al-Mourabit, Org. Lett., 2012, 14, 3202 Search PubMed.
- H. Hussain, I. R. Green and I. Ahmed, Chem. Rev., 2013, 113, 3329–3371 Search PubMed.
- G. C. Pariyar, T. Kundu, B. Mitra, S. Mukherjee and P. Ghosh, ChemistrySelect, 2020, 5, 9781–9786 Search PubMed.
- S. Bhattacharjee, P. Basak and P. Ghosh, Tetrahedron Lett., 2025, 159, 155518 Search PubMed.
- Á. Georgiádes, S. B. Ötvös and F. Fülöp, ACS Sustain. Chem. Eng., 2015, 3, 3388–3397 Search PubMed.
- M. Wang, J. Ma, M. Yu, Z. Zhang and F. Wang, Catal. Sci. Technol., 2016, 6, 1940–1945 RSC.
| This journal is © The Royal Society of Chemistry 2026 |