Open Access Article
Open Access ArticleHigh-throughput C–H activation in a bead-beater homogenizer: fast and regioselective access to 2-arylindoles
Mainak
Banerjee
 *a,
Shravya
B.
a,
Aboy
Chatterjee
a,
Shamima
Hussain
b and
Amtita
Chatterjee
*a,
Shravya
B.
a,
Aboy
Chatterjee
a,
Shamima
Hussain
b and
Amtita
Chatterjee
 *a
*a
aDepartment of Chemistry, Birla Institute of Technology and Science Pilani, KK Birla Goa Campus, Zuarinagar, Goa 403726, India. E-mail: mainak@goa.bits-pilani.ac.in; amrita@goa.bits-pilani.ac.in
bUGC-DAE CSR, Kalpakkam Node, Kokilamedu, TN 603104, India
First published on 7th January 2026
Abstract
High-throughput mechanochemical synthesis is rarely attempted. A bead-beater homogenizer with 24–64 slots offers a unique opportunity for high-throughput mechanosynthesis (HTMS). In this study, C–H bond activation is achieved by homogenizing multiple samples in one go in simple polypropylene (PP) vials with stainless steel balls via liquid-assisted grinding (LAG), with enough frictional force being employed to achieve this challenging transformation. The method demonstrates the preparation of synthetically useful 2-arylindoles through a regioselective reaction between N-alkylated indoles and aryl iodides, catalyzed by Pd(II) and facilitated by C2–H activation in the absence of phosphine ligands, using a bead-beater homogenizer. 5 mol% of Pd(OAc)2 was sufficient to catalyze C2-activation of indoles in the presence of a small volume of EtOAc as the LAG agent, affording 2-arylindoles in high to excellent yields within 9 min. The method demonstrated excellent tolerance to structural variations, including electron-rich and electron-deficient substituents in both indoles and iodoarenes. However, unprotected indoles or indoles with deactivated five-membered rings could not participate well in the reaction. This simple, high-yielding, ambient-temperature high-throughput mechanochemical protocol is devoid of a formal workup step, and with a low E-factor (4.5) and a high Eco-scale score (73.5), it augments well with sustainability matrices.
Introduction
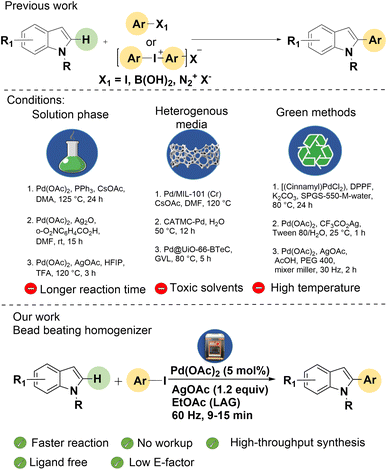
Mechanochemistry has emerged as a transformative strategy in modern organic synthesis, providing a solvent-free and energy-efficient alternative to conventional solution-phase methods.1 Recognized by IUPAC as one of the ten chemical innovations poised to change the conventional synthetic strategies,2 mechanochemistry uses mechanical force to drive chemical reactions, thereby reducing solvent waste and energy requirements.3 Over the past two decades, mechanochemistry has advanced from simple mortar-and-pestle grinding to sophisticated techniques employing mixer mills, planetary ball mills for batch reaction, and even twin-screw extruders for continuous-flow mechanochemistry.4 Other specialized approaches include cryo-mechanochemistry,5 photo-mechanochemistry with LED-equipped jars,6 electro-mechanochemistry using piezoelectric materials,7 and thermo-mechanochemistry assisted by external heating.8 Despite these advances, most of these instruments allow only a single or a couple of reactions to be performed at a time, which restricts their application in rapid optimization, parallel screening, and library synthesis.9 High-throughput experimentation (HTE) is designed to execute multiple experiments in parallel, often using multi-well plates, to accelerate reaction discovery, optimization, and combinatorial synthesis while reducing time and resource consumption.10 Only a few efforts have attempted to combine high-throughput methods with mechanochemical techniques.9,11 One of the examples is the demonstration of a mini-cell homogenizer in the synthesis of quinoxalines via cyclocondensation by our group.9C–H activation has emerged as a powerful strategy in synthetic chemistry, allowing the direct functionalization of otherwise inert C–H bonds.12 Its atom- and step-economical nature has enabled diverse bond constructions, avoiding pre-functionalization of the substrates.13 Recently, mechanochemical C–H activation has proven to be highly effective for constructing C–C and C–heteroatom bonds, in particular, for heteroaromatic sp2 C–H activation;14,15 however, sheer mechanochemical force, employed by high-end milling equipment like a mixer-mill or planetary ball-mill, is commonly required for C–H activation.
2-Arylindoles constitute an important class of heterocycles that occur in numerous natural products and pharmaceuticals, highlighting their value as privileged scaffolds in drug discovery (Scheme 1).16 For example, bazedoxifene is used for the treatment of osteoporosis,17 while other derivatives have been reported as anti-coagulant,17 NorA efflux pump inhibitor,18 anticancer,19 anti-inflammatory,20 and anti-MRSA agents.21 Indole is one of the widely explored scaffolds in C–H activation.22,23 Consequently, numerous strategies have been developed for the synthesis of 2-arylindoles.22–26 A pioneering study on the direct C-2 and C-3 arylation of indoles with aryl halides was reported by Sames and co-workers27 using phosphine ligands. Later, Sanford28 and Larrosa29 independently achieved phosphine ligand-free, regioselective C-2 arylation of indoles under acidic conditions. Building on these studies, various improved protocols have been reported using a broad spectrum of transition-metal catalysts, predominantly palladium, with increasing use of 3d transition metals.25 Typical coupling partners include aryl halides,30–33 boronic acids,34,35 arene diazonium salts,36,37 and aryl iodonium salts.38,39 Despite these advances, most methods still demand high temperatures (>100 °C), toxic solvents such as DMF, and extended reaction times. The reliance on phosphine ligands40 and protecting groups further reduces the efficiency and sustainability. Alternative approaches employing heterogeneous supports, such as Pd catalysts on porous materials39,41,42 or nanoreactors,43,44 have been explored, alongside greener methods in aqueous media,45,46 micellar media,47,48 and mechanochemical synthesis.49,50 Nevertheless, elevated temperatures and long times are often required. In continuation of our sustained interest in the development of mechanochemical methods for C–H activation,50,51 and devising new tools and strategies for mechanochemistry,9 we envisioned that a bead-beater homogenizer with multiple reaction slots could be explored to serve the dual purpose of high-throughput mechanosynthesis (HTMS) and C–H bond activation. Using Pd(OAc)2 as the catalyst with EtOAc as a liquid-assisted grinding (LAG) agent, C2-arylation of indoles via HTMS was achieved in a bead-beater homogenizer under ambient conditions within 9–15 minutes (Scheme 2).
 | ||
| Scheme 1 Some examples of biologically active molecules and drug candidates with a 2-arylindole substructure. | ||
Results and discussion
Our initial studies focused on optimizing the C-2 arylation under homogenization conditions using a model reaction between N-methylindole (1a) and iodobenzene (2a). The reaction was performed in a 2 mL polypropylene vial using an iGene Labserve homogenizer with variable rotor heads, capable of accommodating up to 64 vials simultaneously for parallel reactions. In order to achieve optimized conditions in a time- and energy-efficient manner, 12 reactions were carried out at a time. The reactions were repeated to get consistent results. The optimized conditions were established as 1a (1 equiv.), 2a (1.1 equiv.), Pd(OAc)2 (5 mol%), AgOAc (1.2 equiv.), EtOAc as the LAG agent (200 µL), and 1 g of 2 mm stainless steel (SS) balls at 60 Hz frequency, under which 3a was obtained in 81% yield within 9 min (entry 1, Table 1). The reaction conducted under neat conditions furnished 3a in 72% yield (entry 2, Table 1). Further replacement of EtOAc with other milling auxiliaries, such as PEG-400, EtOH, H2O, or SiO2, resulted in a decreased yield of 3a (entries 3–6, Table 1). Thus, EtOAc proved to be the most effective LAG agent, likely due to better mixing. Reducing the stoichiometric ratio of iodobenzene (2a) to 1.0 equiv. led to a decrease in the yield of 3a (entry 7, Table 1). However, further increasing the amount to 1.2 equiv. resulted in no significant improvement (entry 8, Table 1). Optimization was next directed towards adjusting the number of stainless steel balls and the homogenization frequency to evaluate their influence on the reaction kinetics. Reducing the ball amount to 0.5 g (16 balls) led to a slower reaction (15 min) and a slightly lower yield (78%), while increasing it to 2 g (64 balls) produced a similar yield (80%) without a significant improvement in the reaction efficiency (entries 9 and 10, Table 1). A change in the ball material from SS to zirconia resulted in a similar outcome in terms of time for completion and the yield of 3a (entry 11, Table 1). Meanwhile, reducing the homogenization frequency to 50 Hz or 40 Hz resulted in a slower reaction rate, extending the time required for complete conversion to 18 min (entries 12 and 13, Table 1). Reactions were also attempted with PhBr and PhOTf instead of PhI; however, they were sluggish and afforded only 12% and 14% yields of 3a, respectively, even after 30 min of homogenization, indicating that the cross-coupling is much more facile with iodoarenes than other coupling partners (entries 14 and 15, Table 1). Next, the optimal mol% of the catalyst, Pd(OAc)2, was investigated by conducting reactions with 2 mol% and 10 mol% of the catalyst (entries 16 and 17, Table 1). The reaction with 2 mol% of Pd(OAc)2 was sluggish, whereas 10 mol% of the catalyst offered a similar outcome to that with 5 mol%. Unlike earlier reports,28,50 the presence of acetic acid had little impact on the outcome, as the reaction proceeded efficiently even in its absence with a comparable yield (entry 18, Table 1). Then, cheaper oxidants were evaluated as a replacement for AgOAc while keeping other parameters the same (entries 19–23, Table 1). However, AgOAc was found to be the best oxidant; replacing it with CuOAc or Ag2CO3 reduced the yields to 35% and 62%, respectively, whereas DDQ, 1,4-benzoquinone (BQ), and K2S2O8 gave only trace or low conversions. Therefore, the optimized conditions were arrived at as the use of 5 mol% Pd(OAc)2 as the catalyst, 1.2 equiv. of AgOAc as the oxidant in the presence of 200 µL of EtOAc as the LAG per 0.5 mmol of 1a and 0.55 mmol of 2a (1.1 equiv.) taken in a 2 mL polypropylene vial, and milling with 1 g of SS balls of 2 mm diameter (32 no.) at 60 Hz frequency in a bead-beater homogenizer to get the highest yields of 2-arylindoles.| Entry | Variation from standard conditions | Time | Isolated yield |
|---|---|---|---|
| a Experimental conditions: the reactions were performed using an iGene Labserve bead-beater homogenizer with 1 g of 2 mm stainless steel (SS) balls in a 2 mL polypropylene tube using 1a (0.5 mmol), 2a (0.55 mmol), Pd(OAc)2 (5 mol%), AgOAc (0.6 mmol) and EtOAc (200 µL), which were milled together at 60 Hz frequency. b Auxiliary taken was 2–3 times the weight of 1b. c Both starting materials were partially recovered. | |||
| 1 | None | 9 min | 81 |
| 2 | Neat homogenization | 9 min | 72 |
| 3 | PEG-400 as LAG | 9 min | 77 |
| 4 | H2O as LAG | 9 min | 70 |
| 5 | EtOH as LAG | 9 min | 76 |
| 6 | SiO2 as milling auxiliary | 15 min | 75b |
| 7 | 1.0 equiv. of 2a | 15 min | 75 |
| 8 | 1.2 equiv. of 2a | 9 min | 81 |
| 9 | 0.5 g of SS balls instead of 1 g | 15 min | 78 |
| 10 | 2 g of SS balls instead of 1 g | 9 min | 80 |
| 11 | 1 g of zirconia balls | 9 min | 76 |
| 12 | 40 Hz instead of 60 Hz | 18 min | 78 |
| 13 | 50 Hz instead of 60 Hz | 12 min | 79 |
| 14 | PhBr instead of 2a | 30 min | 12c |
| 15 | PhOTf instead of 2a | 30 min | 14c |
| 16 | 2% of Pd(OAc)2 | 15 min | 45b |
| 17 | 10% of Pd(OAc)2 | 9 min | 79 |
| 18 | AcOH as the additive | 9 min | 80 |
| 19 | CuOAc (1.2 equiv.) instead of AgOAc | 15 min | 35 |
| 20 | Ag2CO3 (1.2 equiv.) instead of AgOAc | 15 min | 62 |
| 21 | DDQ (1.2 equiv.) instead of AgOAc | 15 min | Trace |
| 22 | BQ (1.2 equiv.) instead of AgOAc | 15 min | Trace |
| 23 | K2S2O8 instead of AgOAc | 15 min | Trace |
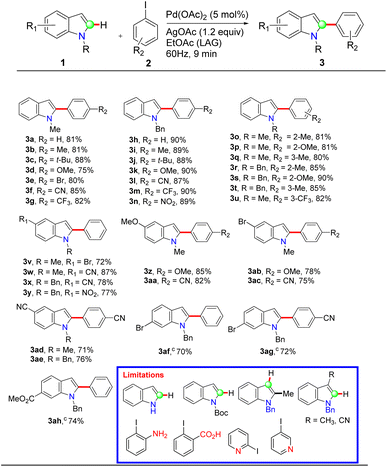
Subsequently, the method was validated by reacting various indole derivatives (1) with several electronically varied iodoarenes (2). To demonstrate high-throughput mechanosynthesis, 12 reactions were set up in parallel to achieve time- and energy-efficiency, and facilitate faster substrate screening. Towards this, 0.5 mmol of indoles (1) and iodoarenes (2, 1.1 equiv.) were taken in 2 mL polypropylene vials containing 1 g of 2 mm SS balls; then, Pd(OAc)2 (5 mol%) AgOAc (1.2 equiv.), and EtOAc (200 µL) were added and the mixtures were homogenized using an iGene Labserve homogenizer set to 60 Hz (Table 2). A pulse of 180 s was set with 1 minute of pause time. The progress of the reactions was monitored via TLC at 3 min intervals, and complete conversion was mostly observed within 9 min unless otherwise mentioned in Table 2. In a few cases, the reactions took 15 min to achieve complete conversion. Upon completion, the crude reaction mixture was transferred from the vial using a small amount of EtOAc, and the stainless steel balls were easily separated using a magnetic retriever. As the entire volume was approximately 1 mL, silica gel (230–400 mesh) was added to it, and the resulting slurry was subjected to flash chromatography. The desired 2-arylindoles (3) were isolated using a work-up-free approach. Notably, no trace of C3-arylated indole was observed or isolated, indicating that the method is highly C2-selective. The 1H and 13C spectroscopic data of the isolated 2-arylindoles (3) were in full agreement with previously reported literature values.50 To evaluate the substrate scope, various aryl iodides (2) were explored with N-methylindole (1a) as the constant coupling partner. Initially, iodoarenes bearing electron-donating (Me, OMe) and electron-withdrawing substituents (CN, CF3, Br) were reacted with N-methylindole, affording the corresponding 2-arylindoles in good to excellent yields ranging from 75% to 88% (3a–3g, Table 2). Therefore, the variation of the electronic behavior in the iodoarenes (2) hardly has any effect on the yield or the reaction kinetics. Notably, N-methylindole also efficiently coupled with sterically hindered iodoarenes, such as those bearing a tert-butyl group (3c, Table 2), indicating that steric bulk in the substrate has minimal effect on the reaction outcome. Substrates containing an additional halogen substituent, such as bromine on the iodoarene, selectively afforded the corresponding monoarylated products due to the difference in reactivity (3e, Table 2). Subsequently, N-benzylindoles (1b) were examined using a similar variety of iodoarenes. Again, the reactions proceeded smoothly, displaying excellent functional group tolerance and delivering the desired products in slightly higher yields (87–90%) (3h–3n, Table 2). The influence of electronic effects at the ortho- and meta-positions of the iodoarenes (2) was also investigated. In all cases, the reaction proceeded efficiently, highlighting the protocol's compatibility with electronically diverse and sterically demanding substrates. Next, the influence of N-substitution on the indole core was also evaluated. However, unprotected indoles or those with an electron-withdrawing group at the N-atom (e.g., N-Boc) hardly afforded any product even after prolonged homogenization, indicating that the optimum conditions are mostly suitable for N-alkylindoles and not for indoles with a deactivated ring. Furthermore, a series of C-5 substituted indole derivatives were reacted with iodobenzene to afford the corresponding products in high to excellent yields, with hardly any electronic effect on the yield or reaction time (3v–3ae, Table 2). However, substitution with electron-withdrawing groups at the C-6 position resulted in slightly lower yields (70–74%) and required longer reaction times (15 min) (3af–3ah, Table 2), likely due to the proximity of the EWG to the reaction site to influence the reactivity. When either the C-2 or C-3 position of the indole ring is blocked, the reaction fails to proceed. This lack of reactivity is attributed to the absence of a feasible C-3 → C-2 migration pathway (Scheme 3), resulting in the intermediate reverting to the starting material. This behavior can be attributed to the higher stability of organometallic intermediates when the metal is bound to the electron-deficient C-2 center adjacent to the nitrogen atom.27 However, aryl iodides bearing a strongly coordinating or polar functional group, such as –NH2 or –CO2H, or heteroaryl iodides, did not furnish any desired product, likely due to the deactivation of the low concentration of palladium catalyst;52 in particular, the influence is much more pronounced as the total volume is nominal. Overall, this protocol offers a rapid and sustainable strategy for the C-2 arylation of indoles, featuring broad substrate scope, high functional group tolerance, and compatibility with both electron-rich and electron-poor aryl iodides. The combination of high-throughput mechanosynthesis, short reaction time (9 min), and workup-free isolation underscores its efficiency, scalability, and operational simplicity.
| a In each case, 0.5 mmol of indole derivative (1) and 0.55 mmol of iodoarene (2), Pd(OAc)2 (5 mol%), AgOAc (1.2 equiv.), and EtOAc (200 µL) as the LAG medium were homogenized for 9 min (unless otherwise stated) in a homogenizer with 1 g of 2 mm SS balls in a 2 mL polypropylene tube at 60 Hz frequency. b All yields reported here are isolated yields of the products (3) after column purification. c Homogenized for 15 min. |
|---|
|
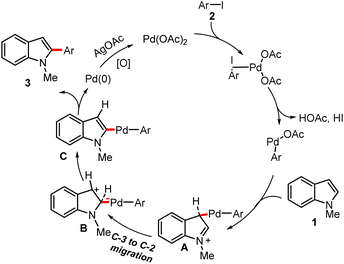
In line with previous reports on ligand-free C-2 arylation of indoles,29,50 a plausible Pd(II)/Pd(0) catalytic cycle is proposed (Scheme 3). Presumably, Pd(OAc)2 undergoes oxidative addition with aryl iodide (2) to generate the Ar–Pd(II)–OAc complex, which attaches to the preferred C-3 position of indole, affording intermediate A. A subsequent 1,2-palladium migration furnishes the C-2 bound intermediate B, which then undergoes reductive elimination to deliver the C-2 arylated indole (3) along with the elimination of Pd(0). The catalytic cycle is completed by AgOAc-mediated re-oxidation of Pd(0) to Pd(II). As mentioned earlier, C-2 or C-3 substituted indoles showed no product formation, which is in accordance with the proposed pathway involving C-3 activation, Pd migration to C-2, and deprotonation.
Sustainability matrices and comparatives
The sustainability of the homogenizer-based high-throughput C-2 arylation protocol was assessed using green metrics such as E-factor and Eco-scale scores. The E-factor for the model reaction for the formation of 3a was calculated to be 4.5, indicating low waste generation. Meanwhile, the Eco-scale score was determined to be 73.5 for 3a, positioning the method near the “excellent” category (Tables S1 and S2, SI). In comparison with conventional homogeneous, heterogeneous, and other green methods, the homogenizer approach uniquely combines ambient conditions with fast reaction kinetics (9–15 min) while maintaining high yields (70–90%). Previously reported methods generally rely on reactions in toxic organic solvents at elevated temperatures, or prolonged reaction times (hours to days), often affording moderate yields, leading to higher E-factors (up to 60) and lower Eco-scale scores (Table S3, SI).27–29,53–59 However, in all cases, setting a single reaction at a time makes them time- and energy-intensive. Notably, for previous mechanochemical methods,49,50 even though they afforded similar yields and E-factor, the reactions took hours to complete and required high-end milling equipment (mixer-mill). Overall, the present protocol represents a sustainable, high-throughput alternative for C-2 arylation of indoles, offering time- and energy-efficiency, achieving higher yields, and better green metrics than existing methodologies.Conclusions
In summary, we have developed a highly efficient, phosphine-free mechanochemical protocol for the regioselective C-2 arylation of N-alkyl indoles by coupling with iodoarenes using a bead-beater homogenizer. Utilizing 5 mol% Pd(OAc)2 as the catalyst and silver acetate as the oxidant, the heteroaryl C–H activation proceeded smoothly in simple polypropylene tubes with stainless steel balls, employing minimal EtOAc as an LAG agent. This method represents a rare example of high-throughput mechanosynthesis (HTMS), enabling simultaneous processing of 12 reactions under ambient conditions. Under optimized homogenization conditions, 34 examples of 2-arylindoles were synthesized in high to excellent yields within just 9–15 minutes. The method displays a broad substrate scope, tolerating both electron-donating and electron-withdrawing substituents on the iodoarenes and indoles. The simplicity of the reaction set up, time- and energy-efficiency, and excellent green metrics (low E-factor, high Eco-scale score) make it a sustainable and more viable alternative for C2-arylation of indoles. This work demonstrates the utility of a bead-beater homogenizer for high-throughput experimentation in the thriving area of C–H bond activation for the first time.Author contributions
The manuscript was written through the contributions of all authors. SB carried out optimization studies, most of the substrate scope validation, and spectral data interpretation; Aboy carried out some C2–H activation with C-5/C-6 substituted indoles; AC contributed with supervision and revision of the manuscript; SH was engaged in reviewing the manuscript and provided input from time-to-time; MB was engaged in the design of the project, investigation, overviewing the entire project, supervision, manuscript writing, and reviewing. All authors have given approval for the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, E-factor, eco-Scale calculation spectral details and spectra. See DOI: https://doi.org/10.1039/d5mr00124b.Acknowledgements
MB is grateful to the UGC-DAE, CRS/2021-22/04/631 project for the financial support. SB is indebted to BITS Pilani, KK Birla Goa campus, for the JRFship. We acknowledge the central sophisticated instrumentation facilities (CSIFs) of BITS Pilani, KK Birla Goa campus, for NMR analysis, and BITS Pilani, Pilani and Hyderabad campus, for HRMS facilities. We also acknowledge that, while drafting certain parts of the manuscript, a free AI tool, ChatGPT, was utilized.References
- F. Gomollón-Bel, Chem. Int., 2019, 41, 12–17, DOI:10.1515/ci-2019-0203.
- N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714, 10.1039/D2CS00997H.
- I. R. Speight, K. J. Ardila-Fierro, J. G. Hernández, F. Emmerling, A. A. L. Michalchuk, F. García, E. Colacino and J. Mack, Nat. Rev. Methods Primers, 2025, 5, 29, DOI:10.1038/s43586-025-00401-2.
- M. Rashid, S. Kasana, V. Nigam, Md. M. Islam, R. Sanan, B. D. Kurmi, V. Asati, G. D. Gupta and P. Patel, Mol. Divers., 2025 DOI:10.1007/s11030-025-11176-9.
- D. M. Baier, C. Spula, S. Fanenstich, S. Grätz and L. Borchardt, Angew. Chem., Int. Ed., 2023, 62, e202218719, DOI:10.1002/anie.202218719.
- C. Schumacher, J. G. Hernández and C. Bolm, Angew. Chem., Int. Ed., 2020, 59, 16357–16360, DOI:10.1002/anie.202003565.
- K. Kubota, T. Endo and H. Ito, Chem. Sci., 2024, 15, 3365–3371, 10.1039/D3SC05796H.
- Y. Pang, J. W. Lee, K. Kubota and H. Ito, Angew. Chem., Int. Ed., 2020, 59, 22570–22576, DOI:10.1002/anie.202009844.
- A. Chatterjee, D. Mahanta, S. Hussain, A. Chatterjee and M. Banerjee, RSC Mech. Chem., 2025, 2, 72–78, 10.1039/D4MR00100A.
- S. Felten, M. Shevlin and M. H. Emmert, in Enabling Tools and Techniques for Organic Synthesis, ed. S. G. Newman, Wiley, 2023, pp. 197–257, DOI:10.1002/9781119855668.
- K. Martina, L. Rotolo, A. Porcheddu, F. Delogu, S. R. Bysouth, G. Cravotto and E. Colacino, Chem. Commun., 2018, 54, 551–554, 10.1039/C7CC07758K.
- C. J. Ann Mary, D. Vasudevan, P. Nagarajan, S. Suhas, A. Vijayan, K. V. Radhakrishnan and Y. N. Sudhakar, Polyhedron, 2024, 264, 117260, DOI:10.1016/j.poly.2024.117260.
- J. Grover, G. Prakash, N. Goswami and D. Maiti, Nat. Commun., 2022, 13, 1085, DOI:10.1038/s41467-022-28707-9.
- M. Banerjee, A. Chatterjee, S. Aneja and A. Chatterjee, J. Org. Chem., 2025, 90, 5323–5335, DOI:10.1021/acs.joc.4c03135.
- N. K. Narayanan and M. Schnürch, ChemCatChem, 2025, 17, e00457, DOI:10.1002/cctc.202500457.
- A. Dorababu, RSC Med. Chem., 2020, 11, 1335–1353, 10.1039/D0MD00288G.
- S. Yaragorla and D. Arun, J. Org. Chem., 2022, 87, 14250–14263, DOI:10.1021/acs.joc.2c01753.
- Y.-S. Yang, J. Yoo, J. Jeon, J. H. Bak, J.-W. Shin, H.-J. Won, H. S. Hwang, J. H. Kim, J. Sim and N.-J. Kim, RSC Adv., 2025, 15, 28131–28135, 10.1039/d5ra04628a.
- X. Wang, X. Xun, H. Song, Y. Liu and Q. Wang, Org. Lett., 2022, 24, 4580–4585, DOI:10.1021/acs.orglett.2c01674.
- S. Siddique, K. R. Ahmad, S. K. Nawaz, A. R. Raza, S. N. Ahmad, R. Ali, I. Inayat, S. Suleman, M. A. Kanwal and M. Usman, Sci. Rep., 2024, 14, 11028, DOI:10.1038/s41598-024-61715-x.
- S. Daly, K. Hayden, I. Malik, N. Porch, H. Tang, S. Rogelj, L. V. Frolova, K. Lepthien, A. Kornienko and I. V. Magedov, Bioorg. Med. Chem. Lett., 2011, 21, 4720–4723, DOI:10.1016/j.bmcl.2011.06.081.
- P. Kumar, P. J. Nagtilak and M. Kapur, New J. Chem., 2021, 45, 13692–13746, 10.1039/D1NJ01696B.
- J. A. Leitch, Y. Bhonoah and C. G. Frost, ACS Catal., 2017, 7, 5618–5627, DOI:10.1021/acscatal.7b01785.
- P. Sihag, M. S. Keerthana and M. Jeganmohan, in Transition-Metal-Catalyzed C–H Functionalization of Heterocycles, ed T. Punniyamurthy and A. Kumar, Wiley, 2023, pp. 193–250, DOI:10.1002/9781119774167.ch5.
- R. A. Jagtap and B. Punji, Asian J. Org. Chem., 2020, 9, 326–342, DOI:10.1002/ajoc.201900554.
- H. Ma, T. Yu, L. Chi, C. Huang, X. Li, R. Zhang and C. Deng, J. Mol. Model., 2022, 28, 267, DOI:10.1007/s00894-022-05265-x.
- B. S. Lane and D. Sames, Org. Lett., 2004, 6, 2897–2900, DOI:10.1021/ol0490072.
- N. R. Deprez, D. Kalyani, A. Krause and M. S. Sanford, J. Am. Chem. Soc., 2006, 128, 4972–4973, DOI:10.1021/ja060809x.
- N. Lebrasseur and I. Larrosa, J. Am. Chem. Soc., 2008, 130, 2926–2927, DOI:10.1021/ja710731a.
- Y. Gao, W. Zhu, L. Yin, B. Dong, J. Fu, Z. Ye, F. Xue and C. Jiang, Tetrahedron Lett., 2017, 58, 2213–2216, DOI:10.1016/j.tetlet.2017.04.066.
- Y. Taskesenligil, M. Aslan, T. Cogurcu and N. Saracoglu, J. Org. Chem., 2023, 88, 1299–1318, DOI:10.1021/acs.joc.2c00716.
- T. Murugesan, C. Sivarajan, C. M. Jayakumari, R. K. Singh, S. R. Vennapusa and A. Kaliyamoorthy, J. Org. Chem., 2021, 86, 10838–10851, DOI:10.1021/acs.joc.1c01123.
- Y. Taskesenligil, F. Lafzi, H. Kilic and N. Saracoglu, J. Heterocycl. Chem., 2019, 56, 3289–3296, DOI:10.1002/jhet.3724.
- Y.-S. Yang, J. Yoo, J. Jeon, J. H. Bak, J.-W. Shin, H.-J. Won, H. S. Hwang, J. H. Kim, J. Sim and N.-J. Kim, RSC Adv., 2025, 15, 28131–28135, 10.1039/d5ra04628a.
- X. Zhu, J.-H. Su, C. Du, Z.-L. Wang, C.-J. Ren, J.-L. Niu and M.-P. Song, Org. Lett., 2017, 19, 596–599, DOI:10.1021/acs.orglett.6b03746.
- H. P. L. Gemoets, I. Kalvet, A. V. Nyuchev, A. V. Erdmann, V. Hessel, F. Schoenebeck and T. Noël, Chem. Sci., 2017, 8, 1046–1055, 10.1039/c6sc02595a.
- C. Liu, T. Miao, L. Zhang, P. Li, Y. Zhang and L. Wang, Chem.–Asian J., 2014, 9, 2584–2589, DOI:10.1002/asia.201402274.
- T. Mandal, T. K. Dutta, S. Mohanty and J. Choudhury, Chem. Commun., 2021, 57, 10182–10185, 10.1039/d1cc04429j.
- I. Anastasiou, V. N. Velthoven, E. Tomarelli, A. Lombi, D. Lanari, P. Liu, S. Bals, D. E. De Vos and L. Vaccaro, ChemSusChem, 2020, 13, 2786–2791, DOI:10.1002/cssc.202000378.
- D. Takeda, M. Yamashita, K. Hirano, T. Satoh and M. Miura, Chem. Lett., 2011, 40, 1015–1017, DOI:10.1246/cl.2011.1015.
- Y. Huang, T. Ma, P. Huang, D. Wu, Z. Lin and R. Cao, ChemCatChem, 2013, 5, 1877–1883, DOI:10.1002/cctc.201200957.
- L. Zhang, P. Li, C. Liu, J. Yang, M. Wang and L. Wang, Catal. Sci. Technol., 2014, 4, 1979–1988, 10.1039/C4CY00040D.
- S. Mandal, P. Sarkar and P. Ghosh, Org. Biomol. Chem., 2024, 22, 7438–7447, 10.1039/D4OB00886C.
- J. Malmgren, A. Nagendiran, C. Tai, J.-E. Bäckvall and B. Olofsson, Chem.–Eur. J., 2014, 20, 13531–13535, DOI:10.1002/chem.201404017.
- O. Moncea, D. Poinsot, A. A. Fokin, P. R. Schreiner and J. Hierso, ChemCatChem, 2018, 10, 2915–2922, DOI:10.1002/cctc.201800187.
- P. Xu and X. H. Duan, New J. Chem., 2021, 45, 19425–19431, 10.1039/d1nj03400f.
- G. N. Vaidya, S. Fiske, H. Verma, S. K. Lokhande and D. Kumar, Green Chem., 2019, 21, 1448–1454, 10.1039/C8GC03413C.
- Z. Xu, Y. Xu, H. Lu, T. Yang, X. Lin, L. Shao and F. Ren, Tetrahedron, 2015, 71, 2616–2621, DOI:10.1016/j.tet.2015.03.051.
- C. Wu, J. Lv, H. Fan, W. Su and X. Cai, Chem.–Eur. J., 2024, 30, e202304231, DOI:10.1002/chem.202304231.
- D. Das, Z. T. Bhutia, A. Chatterjee and M. Banerjee, J. Org. Chem., 2019, 84, 10764–10774, DOI:10.1021/acs.joc.9b01280.
- D. Das, A. A. Bhosle, P. C. Panjikar, A. Chatterjee and M. Banerjee, ACS Sustainable Chem. Eng., 2020, 8, 19105–19116, DOI:10.1021/acssuschemeng.0c07465.
- C. Liu, L. Wang and H. Ge, Chem. Sci., 2025, 16, 12449–12454, 10.1039/D5SC02745D.
- X. Wang, D. V. Gribkov and D. Sames, J. Org. Chem., 2007, 72, 1476–1479, DOI:10.1021/jo061979v.
- C. Liu, L. Ding, G. Guo, W. Liu and F.-L. Yang, Org. Biomol. Chem., 2016, 14, 2824–2827, 10.1039/c5ob02569a.
- Y.-S. Yang, S. Lee, S. H. Son, H.-S. Yoo, Y. H. Jang, J.-W. Shin, H.-J. Won, J. Sim and N.-J. Kim, Org. Chem. Front., 2022, 9, 5906–5911, 10.1039/D2QO01326F.
- L. Duan, R. Fu, B. Zhang, W. Shi, S. Chen and Y. Wan, ACS Catal., 2016, 6, 1062–1074, DOI:10.1021/acscatal.5b02147.
- C. Cai and G.-P. Lu, Synlett, 2012, 23, 2992–2996, DOI:10.1055/s-0032-1317702.
- T. Miao, P. Li, G.-W. Wang and L. Wang, Chem.–Asian J., 2013, 8, 3185–3190, DOI:10.1002/asia.201300913.
- S. Islam and I. Larrosa, Chem.–Eur. J., 2013, 19, 15093–15096, DOI:10.1002/chem.201302838.
| This journal is © The Royal Society of Chemistry 2026 |