Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Materials- and process-driven microstructural engineering for scalable dry-processed electrode manufacturing
Gwonsik Nam†
a,
Jaejin Lim†b,
Seungyeop Choi†b,
Sang Cheol Namc,
Kijoo Hongd,
Jisung Leed and
Yong Min Lee *ab
*ab
aDepartment of Battery Engineering, Yonsei University, 50 Yonsei-ro, Seodaemun-gu, Seoul 03722, Republic of Korea. E-mail: yongmin@yonsei.ac.kr
bDepartment of Chemical and Biomolecular Engineering, Yonsei University, 50 Yonsei-ro, Seodaemun-gu, Seoul 03722, Republic of Korea
cCathode Materials Research Center, POSCO Future M, 31 Sojeongsandan 4-ro, Sojeong-myeon, Sejong-si, 30002, Republic of Korea
dLIB Materials Research Center, POSCO N.EX.T Hub, POSCO Holdings, 100 Songdogwahak-ro, Yeonsu-gu, Incheon, 21985, Republic of Korea
First published on 24th February 2026
Abstract
Solvent-free dry-processed electrode (DPE) technology has become a promising platform for the manufacturing of lithium-ion batteries, offering a low carbon footprint, enhanced energy efficiency, and cost-effectiveness compared to conventional slurry-based processes. In addition to its process sustainability, the dry process enables the formation of a distinctive electrode microstructure, characterized by a well-interconnected pore network, low tortuosity, a broadened active surface area, and improved electron conduction pathways, which supports high energy density and power performance. Although considerable laboratory-scale progress has been made, several critical challenges, such as the non-uniform binder distribution, insufficient mechanical integrity during large-scale manufacturing, and limitations in current collector compatibility, still hinder the commercialization of DPEs while preserving their microstructural advantages. Overcoming these barriers requires synergistic innovations in terms of materials design and process engineering. This review highlights recent advancements in key material components, including active materials, conductive additives, binders, and current collectors, along with process technologies such as powder mixing, kneading, laminating, and calendering, all from the perspective of microstructural optimization. We discuss how materials innovation can address process limitations and, conversely, how novel process strategies can accommodate material constraints. Finally, this review provides a microstructure-centric perspective on the materials and process innovations required for ensuring the scalable production of high-performance and sustainable DPEs with unique microstructural features.
Wider impactOwing to the growing interest in gigafactory-scale and sustainable battery manufacturing, dry-processed electrodes (DPEs) have become a promising platform, eliminating toxic solvents and energy-intensive drying processes. This review highlights recent advances in both materials design and process engineering that enable the scalable fabrication of DPEs with structurally robust architectures. Unlike conventional slurry-based electrodes, DPEs have distinctive microstructural features that are not simply determined by the materials or processes alone but are co-shaped through their coupled, bidirectional interplay. These microstructures critically impact electrode performance and reliability, especially in thick electrodes. By characterizing the DPE manufacturing process into four key stages—mixing, kneading, laminating, and calendering—the origin of major defects, including non-uniformity, delamination, and heterogeneous densification, is identified and strategies for mitigating these issues are proposed. This review provides a practical framework linking materials chemistry and process engineering to microstructure and performance, offering valuable insights for researchers and engineers developing the next generation of high-performance, scalable, and sustainable electrode manufacturing technologies. |
1. Introduction
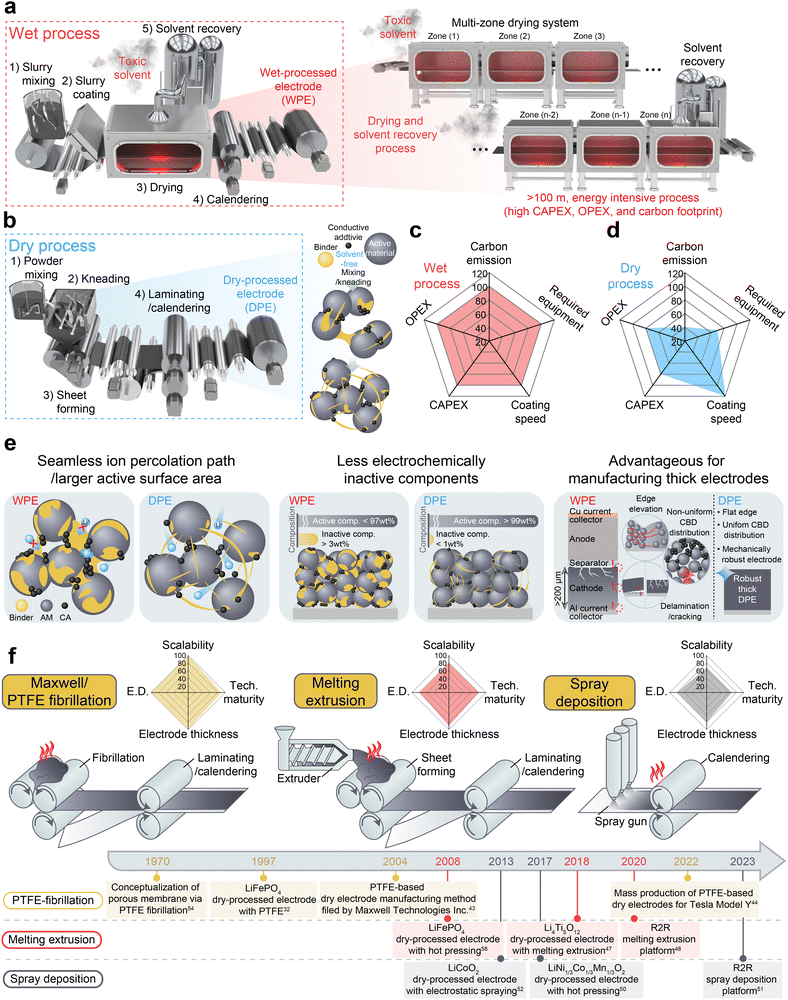
The growing urgency to mitigate the impacts of climate change has accelerated the global transition toward electrification, primarily enabled by lithium-ion battery (LIB) technologies. In recent years, the global sale of electric vehicles (EVs) has increased by >24%, highlighting the rapid expansion of the EV market.1–3 Despite this growth, the practical implementation of EVs remains hindered by inherent trade-offs among core battery attributes, particularly energy density, affordability, and safety, each with stringent and often conflicting requirements. To fully replace internal-combustion engine vehicles, LIBs must simultaneously meet performance targets, including high energy density (>350 Wh kgcell−1), low manufacturing costs (<100 USD kWhcell−1), long-term durability (>15-year service life), and high safety under actual operating conditions.4–7 These performance demands have driven extensive research on all major battery components, including cathodes, anodes, electrolytes, and separators. Traditionally, LIB development has focused on chemistry-driven materials-level innovation, leading to remarkable advances in terms of intrinsic electrochemical properties such as specific capacity, electrochemical stability, and cycle life.8–12 While such material-level improvements have resulted in notable performance gains, it is becoming increasingly evident that cell-level performance is not solely determined by chemistry alone. Instead, the architecture of electrodes, including design parameters such as composition, mass loading, and density, and microstructural features like component distribution, active surface area, and ion/electron conduction pathways play decisive roles in translating material properties into real-world device performance. Consequently, studies have recently moved beyond materials design to incorporate electrode- and cell-level engineering, highlighting the need for integrated approaches that optimize materials, structures, and processing in tandem.13–17Among the various electrode-level strategies, the design of thick electrodes is gaining increasing attention as a promising approach for simultaneously reducing the manufacturing cost per kilowatt-hour (kWh) and enhancing the energy density by minimizing the relative fractions of inactive components such as current collectors and separators. For instance, by increasing the cathode mass loading from 20 mg cm−2 (roughly equivalent to a thickness of ∼57 µm at an electrode density of 3.5 g cm−3) to 70 mg cm−2 (∼200 µm), the proportion of inactive components could be reduced from approximately 21% to 6%, while the gravimetric energy density could be increased from 475 Wh kg−1 to 541 Wh kg−1 (this estimation considered only the cathode and current collector).18–20 Despite these advantages, conventional wet-processed electrodes (WPEs) face intrinsic limitations when implemented in thick formats. Solvent evaporation during drying often induces problems such as carbon–binder domain (CBD) migration and electrode delamination, leading to structural heterogeneity and increased internal resistance.21–23 Additionally, wet processing depends on energy-intensive drying and toxic solvent recovery systems (e.g., N-methyl-2-pyrrolidone, NMP), thereby increasing both manufacturing costs and environmental burdens.
To overcome these limitations, dry-processed thick electrodes, which eliminate the need for solvents and the associated drying and recovery processes, have been adopted. By avoiding solvent-related emissions and substantially lowering energy consumption, dry-processed electrodes (DPEs) offer a significantly reduced carbon footprint.24
While dry process techniques provide distinct advantages, the comprehensive understanding of how materials selection and processing strategies co-shape the electrode microstructure in DPEs remains limited. This review critically examines recent advances in DPE technology by integrating materials innovation and process engineering within a microstructure-centric framework. This approach underscores the bi-directional interplay between materials and processes, highlighting how targeted materials design can mitigate the intrinsic limitations of processes, and, conversely, how advanced processing strategies can accommodate material constraints.
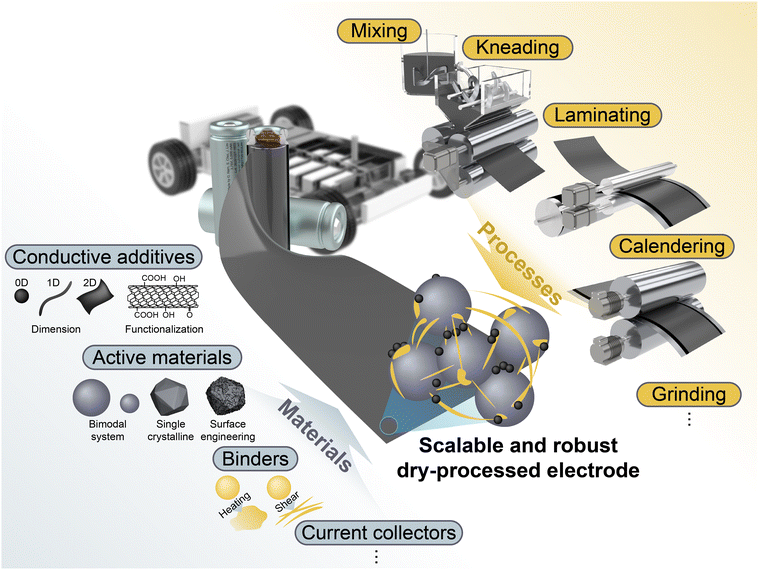
Key electrode components, including active materials, conductive additives, binders, and current collectors, are systematically discussed, alongside critical processing steps such as powder mixing, kneading, laminating, and calendering, with particular emphasis on how materials innovation and processing strategies result in microstructure evolution in DPEs. Furthermore, polytetrafluoroethylene (PTFE)-based binder systems, which continue to play a central role in DPE manufacturing, are examined, alongside emerging alternatives that present new opportunities for microstructural control and functional enhancement in DPEs. Ultimately, this review provides a unified, microstructure-driven perspective that bridges materials chemistry with process design, offering critical insights into the scalable manufacturing of high-performance, cost-effective, and environmentally sustainable DPEs.
2. Advantages and manufacturing methods of dry-processed electrodes
2.1. Advantages of the dry process, compared to the conventional slurry-based wet process
2.2. Typical manufacturing methods for dry-processed electrodes
Various manufacturing methods have been developed for DPEs, including PTFE-fibrillation-based methods (Maxwell technology),43,44 powder hot pressing,45,46 melting extrusion,47,48 vapor deposition,49 dry-spray deposition,50–52 and 3D printing.53 Among these, Maxwell/PTFE fibrillation, melting extrusion, and spray deposition demonstrate the highest technological maturity and scalability, especially in terms of roll-to-roll (R2R) compatibility and industrial applicability. Fig. 1f provides a comparative schematic overview of these representative dry fabrication methods.2.3. Materials- and process-driven dry-processed electrode engineering
As summarized in Fig. 2, several types of microstructural defects can be observed in DPEs, including (1) non-uniformity, (2) delamination, and (3) heterogeneous densification accompanied by crack formation. These issues arise from the complex interplay between processing conditions and material properties. For instance, a non-uniform distribution of components may result from improper shear conditions during mixing and kneading, while also being strongly affected by the particle size, morphology, surface chemistry, and thermodynamic properties of the active materials, conductive additives, and binders. This review dissects the DPE manufacturing process step-wise, including the mixing, kneading, laminating, and calendering processes, and critically examines the microstructural challenges that arise at each stage. We then discuss both process-level modifications and material-level innovations proposed in recent studies to mitigate these issues.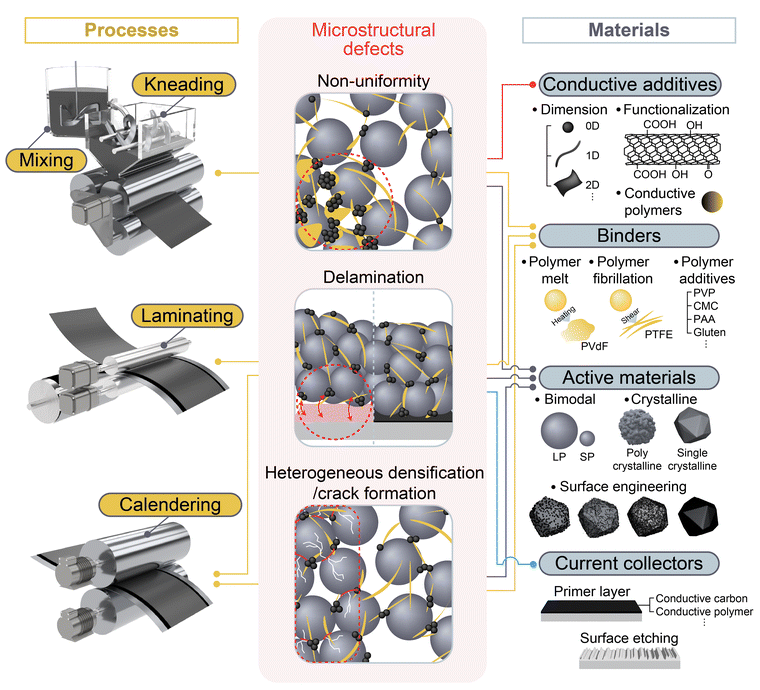 | ||
| Fig. 2 Microstructural challenges in DPE manufacturing and the interplay between manufacturing processes and materials design. | ||
3. Challenges and strategies for scalable dry-processed electrode manufacturing
PTFE-based DPEs are typically dry-mixed and fibrillated before lamination and calendering.30,55,65 Dry powder mixing strongly affects the microstructure of the resulting powder mixture, thus greatly influencing the electrochemical performance and mechanical integrity of the electrode. Owing to the absence of a dispersing solvent, achieving a homogeneous distribution of electrode components at the microscale remains a critical challenge in dry processing. Moreover, the shear-induced fibrillation of PTFE under mechanical mixing conditions contributes to the formation of a fibrous binder network that enhances mechanical robustness but requires a careful optimization of processing parameters. Finally, because of the hydrophobic nature of PTFE, an optimized lamination strategy is required to address interfacial adhesion issues, while careful control of calendering conditions is essential for mitigating particle microcracking in thick electrodes. The following sections systematically discuss recent strategies involving materials modification and processing control, to overcome the inherent challenges of each step.3.1. Powder mixing process
The powder mixing process is an important step for ensuring uniform dispersion of active materials, conductive additives, and binders. This process must be achieved through mechanical mixing, as the dispersant-assisted wet dispersion of conductive additives is not employed.66–69 In terms of material properties, carbon additives are inherently highly likely to agglomerate owing to the presence of van der Waals and π–π interactions as well as because of their high surface energy, making effective dispersion particularly challenging.57,70,71Oh et al.56 reported the effects of various carbon additives on DPEs. Carbon nanotubes (CNTs) exhibited intrinsically high electrical conductivity (∼200 S cm−1), approximately 100 times higher than that of conventional carbon black (Super P: ∼29.6 S cm−1, Ketjen black: ∼23.2 S cm−1). However, despite this superior intrinsic conductivity, their electrode-level advantage was substantially diminished due to poor dispersion of CNTs under physical mixing conditions. In contrast, conventional carbon black, despite its inherent tendency toward aggregation, showed comparatively more homogeneous dispersion than CNTs owing to its 0D morphological properties. To exploit their intrinsically high electrical conductivity and fibrous morphology, recent efforts focused on achieving homogeneous CNT dispersion, which enables the formation of a percolated electronic network.154–156 Accordingly, the surface modification strategies have been introduced, offering two key advantages: (1) reducing interparticle agglomeration; the surface functionalization introduces polar functional groups that weaken van der Waals attraction157,158 and (2) improving interfacial interactions with active materials due to hydrogen bonding and dipole–dipole interactions between active materials and carbon additive surfaces.57 However, most functionalization approaches rely on acid-based treatments, which often induce buckypaper-like structures, as CNTs inevitably undergo re-bundling during drying due to capillary forces, thereby limiting their dispersibility.159,160 This limitation becomes more pronounced during dry mixing, as even high shear stress is insufficient to disrupt re-bundled CNTs, ultimately hindering uniform CNT distribution. To mitigate the intrinsic limitations of carbon materials, Kim et al.72 introduced ozone-treated CNTs as shown in Fig. 3a. Conventional CNT functionalization is typically achieved using strong liquid-phase oxidants to generate surface functional groups. However, such an approach often causes severe damage to the sp2 carbon framework, leading to deterioration of electronic conductivity. In addition, acid-based mixing generally results in the formation of rigid buckypaper structures, making it unsuitable for dry processes. Contrastingly, ozone treatment not only preserves the initial fluffy powdery texture of CNTs but also introduces oxygen-containing functional groups, such as hydroxyl, carbonyl, and carboxylic-acid groups, onto the surface. X-ray microscopy (XRM) analysis revealed that, after pressing, an ozone-treated single-walled CNT (O-SWCNT) electrode with NCM-based cathode active materials maintained a porosity of 13.89%, whereas a Super P-NCM (SP) electrode retained a higher porosity of 20.90%. This difference was attributed to the partial aggregation of Super P particles, even under the application of high shear forces during calendering. As an alternative strategy, the pre-dispersion of carbon materials has been achieved by coating the active materials, rather than by directly modifying the carbon materials themselves.57,65,73 Koo et al.57 reported a uniform wrapping of a multi-walled CNT (MWCNT) on single-crystalline NCA (SC-NCA) cathode active materials, in which an anti-solvent-induced salt-out effect was employed to trigger MWCNT precipitation (Fig. 3b). In comparison, a conventional WPE containing 2 wt% carbon black (CB-wet) exhibited a highly non-uniform distribution of conductive additives relative to a DPE incorporating CNTs (CNT-Dry). This inhomogeneous conductive network resulted in the degradation of the electronic percolation pathway and, consequently, higher local resistance, as was clearly visualized using 2D scanning spreading resistance microscopy (2D-SSRM). Furthermore, 3D digital twin structural analysis revealed that the effective electronic conductivity (σs,eff) of a CNT-DPE was approximately 3.1 times higher than that of a CB-WPE, while the tortuosity of the electronic transport pathways was reduced by 36%. By pre-dispersing an MWCNT directly onto the cathode active material, CNT agglomeration was effectively mitigated, leading to a highly uniform carbon distribution. Consequently, both lithium-ion and electronic transport processes were significantly enhanced, allowing an exceptionally high active material fraction of up to 99.6 wt% within the electrode and achieving a high electrode density of 4.0 g cm−3. Moreover, Linh et al.74 proposed a facile pre-dispersion strategy using polyvinylpyrrolidone (PVP), a cost-effective and widely used polymeric dispersant, to facilitate homogeneous MWCNT dispersion (Fig. 3c). The PVP macromolecular chains absorbed onto the MWCNT surfaces, preventing aggregation and forming a stable dispersion in ethanol before electrode fabrication. The authors achieved a well-distributed conductive network that promoted effective electronic percolation in the electrode. Accordingly, PVP–MWCNT DPEs (PCDPEs) with areal capacities of 8 and 10 mAh cm−2 exhibited markedly higher discharge capacity and improved capacity retention than those of MWCNT-only DPEs (CDPEs), highlighting the advantages of enhanced conductive network formation in thick electrodes.
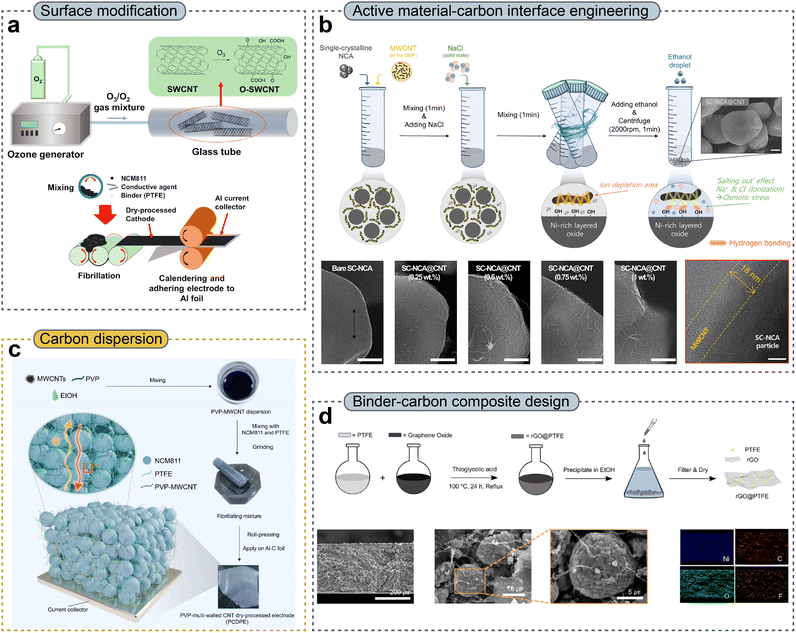 | ||
| Fig. 3 (a) Schematic illustration of the ozone treatment process for surface functionalization. Reproduced with permission from ref. 72. Copyright 2023, The Royal Society of Chemistry. (b) Processing schematic of MWCNT wrapping on SC-NCA (top) and corresponding SEM and TEM images (bottom). Reproduced with permission from ref. 57. Copyright 2025, Elsevier B.V. (c) The PVP–MWCNT coating process for improving MWCNT dispersibility. Reproduced with permission from ref. 74. Copyright 2025, Elsevier B.V. (d) Schematic procedure of the PTFE coating strategy on rGO (top) and SEM/EDS analyses of electrodes fabricated using rGO@PTFE (bottom). Reproduced with permission from ref. 79. Copyright 2025, Wiley-VCH GmbH. | ||
However, the above-mentioned strategies have primarily focused on carbon additives only, whereas achieving a uniform distribution of both carbon additives and binders remains a critical design consideration. Because PTFE is electrically insulating, its inhomogeneous dispersion can induce not only a heterogeneous current density but also deterioration of the mechanical integrity of the electrode structure.75–77 Accordingly, the spatial distribution of the CBD plays a decisive role in determining both the electronic percolation network and the mechanical robustness of the electrode, thus significantly influencing the overall electrochemical performance.21–23,78 Yoon et al.79 reported a wet-chemical process using thioglycolic acid to synthesize a PTFE-nanoparticle-anchored reduced graphene oxide (rGO@PTFE) composite (Fig. 3d). Owing to the presence of anchored PTFE nanoparticles, the rGO@PTFE composite effectively suppressed restacking of rGO sheets, mitigated aggregation, and promoted enhanced nanofibrillation behavior. As a result, the rGO@PTFE-based ultra-thick cathode (G@P_TC) exhibited a high lithium-ion transference number of 0.73, whereas the Super P-PTFE-based thick cathode (S-P_TC) showed a substantially lower value of 0.43. The improved lithium-ion transport in G@P_TC effectively alleviated concentration polarization in thick electrodes, enabling a more uniform ionic flux throughout the electrode. In addition, G@P_TC delivered superior electrochemical performance, achieving a maximum areal capacity of 15.2 mAh cm−2 and a volumetric capacity of 563 mAh cm−3, respectively, along with excellent rate capability and cycling stability.
In conclusion, achieving a homogeneous dispersion of inactive materials is critically important, as it determines the electrode microstructure and cell performance. To facilitate a systematic comparison, Table 1 summarizes various strategies for achieving a uniform dispersion of inactive components, including conductive carbon additives and binders.57,65,72–74,79–81 This comparison provides insights into how the targeted modification of inactive materials can promote uniform mixing behavior, thereby facilitating the scalable fabrication of thick DPEs.
| Materials | Surface modifier | Function | Approach | Electrode composition (wt%) | Electrochemical performance | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Active material | Conductive additive | Binder | Cell configuration | Loading level (mg cm−2) | Areal capacity (mAh cm−2) | Electrolyte | Voltage range | C-rate@temper-ature | Capacity retention@cycles | ||||||
| Active materials | Single crystalline LiNi0.8Co0.15Al0.05O2 | MWCNT | (1) Homogeneous carbon distribution on cathode materials | Salt-induced selective precipitation method | 99.6[NCA/MWCNT = 99.25/0.75(w/w)] | — | 0.4(PTFE) | Graphite||NCA | 23(NCA) | 4.6 | 1.15 M LiPF6 | 2.5–4.25 V | 0.5C@25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
95%@250 cycles | 57 |
| EC/EMC/DEC | |||||||||||||||
| 2/4/4 (v/v/v) | |||||||||||||||
| (2) Spatially uniform conductive pathways | |||||||||||||||
| (3) Enhanced electrode density (4.0 g cm−3) with a low binder composition (0.4 wt%) | 1 wt% VC | ||||||||||||||
| 1 wt% LiPO2F2 | |||||||||||||||
| Single crystalline LiNi0.8Co0.1Mn0.1O2 | MWCNT | (1) Ultralow binder content (∼0.3 wt%) | Pickering emulsion-assisted assembly method | 99.5[NCM811/MWCNT =99.7/0.3(w/w)] | — | 0.5(PTFE) | Graphite+Si||NCM811 | 51.9(NCM811) | 10 | 1.1 M LiPF6 | 3.0–4.3 V | 0.33C | 78%@500 cycles | 65 | |
| EC/DMC/EMC | |||||||||||||||
| 2/6/2 (v/v/v) | |||||||||||||||
| (2) Strong CNT–PTFE interfacial anchoring | |||||||||||||||
| 1 wt% VC | |||||||||||||||
| (3) 3D conductive network with improved tensile strength | 10 wt% FEC | ||||||||||||||
| Single crystalline LiNi0.8Co0.1Mn0.1O2 | SWCNT | (1) Homogeneous carbon distribution on cathode materials | Spray-drying-assisted composite formation method | 98 [NCM811/ SWCNT = 99.88/0.12(w/w)] | — | 2(PTFE) | Graphite+Si||NCM811 | 36(NCM811) | 7 | 1 M LiPF6 | 2.5–4.25 V | 1.0C | 89.40%@100 cycles | 73 | |
| EC/DMC | |||||||||||||||
| (2) Ultralow carbon additive content (0.12 wt%) | 3/7 (v/v) | ||||||||||||||
| 5 wt% FEC | |||||||||||||||
| Binder materials | PTFE | Graphene oxide | (1) Lowered percolation threshold | Chemical reduction–precipitation method | 90(NCM) | — | 10[PTFE/rGo = 3/7(w/w)] | Li metal||NCM | 85(NCM) | 15.2 | 1 M LiPF6 | 2.7–4.3 V | 0.1C | 82%@100 cycles | 79 |
| (2) Reduced interfacial contact between carbon additives and active materials | EC/DMC | ||||||||||||||
| 1/1 (v/v) | |||||||||||||||
| PTFE | MWCNT | (1) Good percolating network | Freeze-drying -induced composite formation method | 98(LCO) | — | 2[PTFE/MWCNT = 1/2(w/w)] | Li metal||LCO | 101(LCO) | 14.8 | 1 M LiPF6 | 3.0–4.3 V | 0.1C | 92.60%@20 cycles | 80 | |
| (2) Homogeneous electron transport | EC/DMC | ||||||||||||||
| (3) Improved mechanical properties | 1/1 (v/v) | ||||||||||||||
| PTFE | Carbon black | (1) Preventing agglomeration of the conducting agent and PTFE binder in the electrode | Charge-induced electrostatic assembly method | 98(NCMA) | 1(Super P) | 1[PTFE/Super P = 1/1(w/w)] | Graphite||NCMA | 24(NCMA) | 5 | 1.15 M LiPF6 | 2.8–4.3 V | 0.5C | 82%@200 cycles | 81 | |
| EC/EMC/DEC | |||||||||||||||
| 2/4/4 (v/v/v) | |||||||||||||||
| 1 wt% VC | |||||||||||||||
| 1 wt% LiPO2F2 | |||||||||||||||
| Conductive additives | SWCNT | — | (2) Increased surface hydrophilicity of carbon additives with decreased electrical conductivity | Ozone-induced surface modification method | 96(NCM811) | 2(O-SWCNT) | 2(PTFE) | Graphite||NCM811 | 30(NCM811) | 5.76 | 1 M LiPF6 | 2.5–4.25 V | 0.33C | 85.60%@100 cycles | 72 |
| EC/DEC | |||||||||||||||
| (3) Homogeneous distribution of carbon additive | 1/1 (v/v) | ||||||||||||||
| 10 wt% FEC | |||||||||||||||
| MWCNT | PVP | (1) Homogeneous distribution of carbon additive | Solvent-assisted pre-dispersion method | 97(NCM811) | 1[MWCNT/PVP = 10/1 (w/w)] | 2(PTFE) | Li metal||NCM811 | 40(NCM811) | 8 | 1 M LiPF6 | 2.8–4.4 V | 0.33C@25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
60.23%@50 cycles | 74 | |
| EC/DMC/EMC | |||||||||||||||
| (2) Improved charge transfer | 1/1/1 (v/v/v) | ||||||||||||||
| 1 wt% VC | |||||||||||||||
3.2. Kneading process
Kneading is an essential step for PTFE-based DPE processing, as it induces PTFE fibrillation. Apart from ensuring the homogeneous distribution of PTFE, the degree of fibrillation strongly determines the electrode microstructure and mechanical properties, thus considerably influencing electrochemical performance.82–84 Therefore, from both the perspectives of the intrinsic PTFE properties and processing conditions, systematic studies are required to elucidate how PTFE fibrillation evolves and how the resulting changes influence electrochemical behavior.Lee et al.76 systematically investigated the mechanical and electrochemical properties of electrodes with respect to the particle size of PTFE (Fig. 4a). Pristine PTFE (PTFE(L)) exhibited an average particle size of 492.31 µm, which decreased to 87.73 µm after 8 cycles of cryogenic freezer milling (CFM, PTFE(M)), and further to 6.44 µm after 16 CFM cycles (PTFE(S)). DPEs fabricated using PTFE with different particle sizes were evaluated using a surface and interfacial cutting analysis system (SAICAS) to assess interfacial adhesion, revealing distinct differences in adhesion behavior between PTFE(L) and PTFE(S) under both vertical and horizontal stresses at a 75% depth. Electrochemical impedance spectroscopy (EIS) analysis further demonstrated that decreasing the PTFE particle size led to reduced charge transport resistance, resulting in enhanced ionic and electronic conductivity. This improvement was attributed to the minimized contact area of the inherently insulating PTFE when employing smaller particles, which facilitated more efficient charge transport within the electrode. As shown in Fig. 4b, Lee et al.85 investigated how molecular weight (MW) influences the PTFE binder's fibrillation behavior. Compared with low-MW PTFE (PTFE(L)), high-MW PTFE (PTFE(H)) exhibited a lower degree of agglomeration and a more uniform fibrous morphology. This behavior stemmed from the sufficiently long polymer chains of PTFE(H), which could sustain and propagate fibril formation, thereby promoting effective fibrillation. Consequently, PTFE(H) showed superior fibrillation behavior and a more homogeneous distribution within the electrode. Composite cathode electrodes incorporating PTFE(H) not only exhibited lower electronic resistance but also demonstrated enhanced adhesion strength. Accordingly, an all-solid-state Li–In/LPSCl/NCM cell employing PTFE(H) delivered a high discharge capacity of 209.7 mAh g−1 and exhibited excellent cycling retention of 97.4% after 300 cycles, even at a high mass loading of 22.5 mg cm−2. Furthermore, Lee et al.86 tuned the degree of crystallinity of PTFE by heating it to 200 °C and then varied the cooling rate between 2 and 15 °C min−1. Thereafter, they applied crystallinity- controlled PTFE to compose cathode electrodes for all-solid-state batteries. The prepared PTFE samples with different crystallinities were characterized using X-ray diffraction (XRD), which confirmed the presence of amorphous (A-PTFE), semi-crystalline (SC-PTEF, conventional PTFE), and highly crystalline states (C-PTFE). Stress–strain measurements revealed that A-PTFE resulted in an increased mechanical strength; this enhanced strength effectively mitigated interparticle contact loss within the electrode. In electrochemical evaluations, a C-PTFE adopted NCM cathode electrode exhibited a high initial discharge capacity of 171 mAh g−1 and maintained a high capacity retention of 84.1% after 200 cycles. In summary, the intrinsic properties of PTFE, including its MW, particle size, and crystallinity, are critical in governing the degree of fibrillation and, consequently, the electrochemical performance.
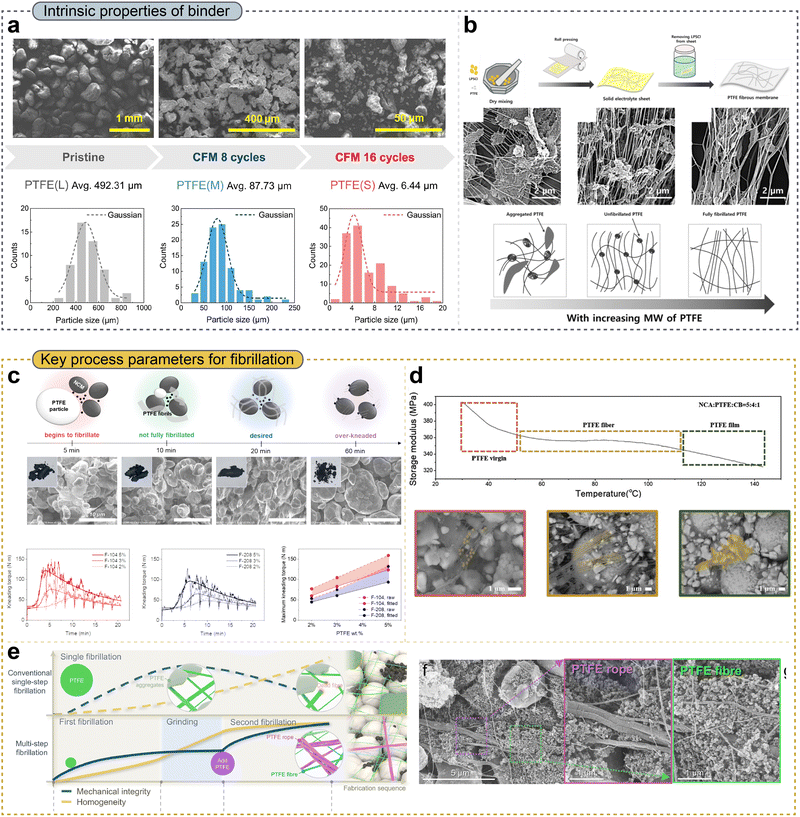 | ||
| Fig. 4 (a) SEM images (top) and particle size distribution (bottom) of cryo-pulverized PTFE powders. Reproduced with permission from ref. 76. Copyright 2024, Elsevier B.V. (b) Processing schematic of the solid electrolyte sheet formation (top). SEM image (middle) and a predictive fibrillation model for different molecular weights (bottom). Reproduced with permission from ref. 85. Copyright 2024, Elsevier B.V. (c) Schematic illustration of the morphological changes of the PTFE binder with kneading time (top), corresponding SEM images (middle), and kneading torque profiles (bottom). Reproduced with permission from ref. 55. Copyright 2024, Elsevier B.V. (d) Storage modulus of PTFE at different temperatures (top) and corresponding SEM images (bottom). Reproduced with permission from ref. 77. Copyright 2024, Wiley-VCH GmbH. (e) Schematic illustration of the multi-step fibrillation process (left) and SEM images of rope- and fiber-like PTFE morphologies at different feeding times (right). Reproduced with permission from ref. 88. Copyright 2025, The Royal Society of Chemistry. | ||
From a processing perspective, the degree of fibrillation varies with kneading parameters such as kneading time, temperature, and feeding steps, thus influencing both the mechanical integrity of electrodes and the ion percolation pathways. Oh et al.55 reported that the fibrillation morphology evolved markedly with kneading time (Fig. 4c). During the initial 5 min of kneading, fibrillation was initiated, while nearly complete fibrillation was achieved at 20 min. In contrast, prolonged kneading for 60 min resulted in an over-kneaded state, in which the fibrils were fractured, leading to a loss of mechanical integrity within the mixer. For F-104 and F-208 binders, although their intrinsic material properties, such as particle size, bulk density, and specific gravity, were comparable, their extrusion ratios differed significantly, ranging from 36 to 1500. During kneading, F-104, which had a lower extrusion ratio, exhibited a higher kneading torque than F-208. These observations highlight the fact that the optimal kneading process must be carefully tailored to the intrinsic properties of the PTFE binder. Moreover, Kim et al. reported that temperature is a critical processing factor during PTFE kneading (Fig. 4d).77 When fibrillation was conducted at 30 °C, insufficient fibrillation occurred, resulting in PTFE remaining largely in a virgin-like morphology and exhibiting a low tensile strength of 0.43 MPa. In contrast, kneading at 80 °C led to a well-developed, fully fibrous PTFE morphology, accompanied by a significant increase in tensile strength to approximately 0.85 MPa. When the kneading temperature was increased to 135 °C, which exceeds the glass transition temperature (Tg) of PTFE, the fibrils tended to coalesce into film-like structures, indicating excessive softening. Notably, fibrillation at an optimized temperature of 80 °C resulted in a more homogeneous distribution of PTFE and conductive additives, thereby facilitating continuous ion and electron transport pathways, which, in turn, reduced the ionic resistance (Rion) and charge transfer resistance (Rct). Lee et al.87 comprehensively investigated the effects of PTFE processing on tensile stress and ionic conductivity during the fabrication of solid electrolyte sheets. As the number of calendering loops increased, the tensile stress increased, whereas the ionic conductivity remained at comparable levels. Furthermore, similar tensile stresses were observed at uniaxial (perpendicular to the fibrillation direction) and biaxial fibrillation directions. However, when the uniaxial fibrillation was aligned parallel to the fibrillation direction, a lower tensile strength was attained. This degradation was attributed to the aggregation of 1D PTFE fibers. In contrast, biaxial fibrillation resulted in a random distribution of fibers, leading to an improved tensile strength. As an alternative strategy for achieving uniform fibrillation, Lee et al.88 introduced a dual-fibrous DPE fabrication method in which multistep kneading and grinding were performed as shown in Fig. 4e. This multistep grinding–kneading process enabled the uniform distribution of two types of PTFE fibers—yarn-like thin fibers and rope-like thick fibers—resulting in homogeneous microstructures at both the macroscopic and microscopic scales and leading to an enhanced electrochemical performance. The rope-like fibers improved the mechanical strength by reducing the edge roughness and enhancing the cohesion and adhesion within the electrode.
In summary, the selection of intrinsic material properties of the PTFE binder, together with the fibrillation processing conditions, governs the mechanical integrity and the ionic and electronic pathways of electrodes. Therefore, beyond carefully selecting PTFE binder properties, the optimization of fibrillation processes is essential for scalable DPE fabrication.
3.3. Laminating process
The laminating process provides sufficient adhesion between an electrode layer and a metallic current collector, thereby ensuring the mechanical integrity of the electrode. However, in PTFE-based DPEs, the bonding strength with metallic current collectors is limited; instead, interfacial adhesion is dominated by limited mechanical interlocking.89–91 In addition, PTFE exhibits a low surface energy of approximately 18 mN m−1, whereas aluminum (Al) current collectors have significantly higher surface energies of approximately 40 mN m−1.91,92 This noticeable difference in surface energy leads to a reduced work of adhesion at the electrode–current collector interface, further worsening the interfacial adhesion strength.93–95 Moreover, the interfacial adhesion between the electrode and the current collector is characterized by localized point-to-point contact, as PTFE provides binding through fibrillation. The effective contact area is severely limited, leading to inherently low interfacial adhesion strength. Addressing the weak interfacial adhesion, therefore, necessitates the development of advanced current collector designs and surface modification strategies.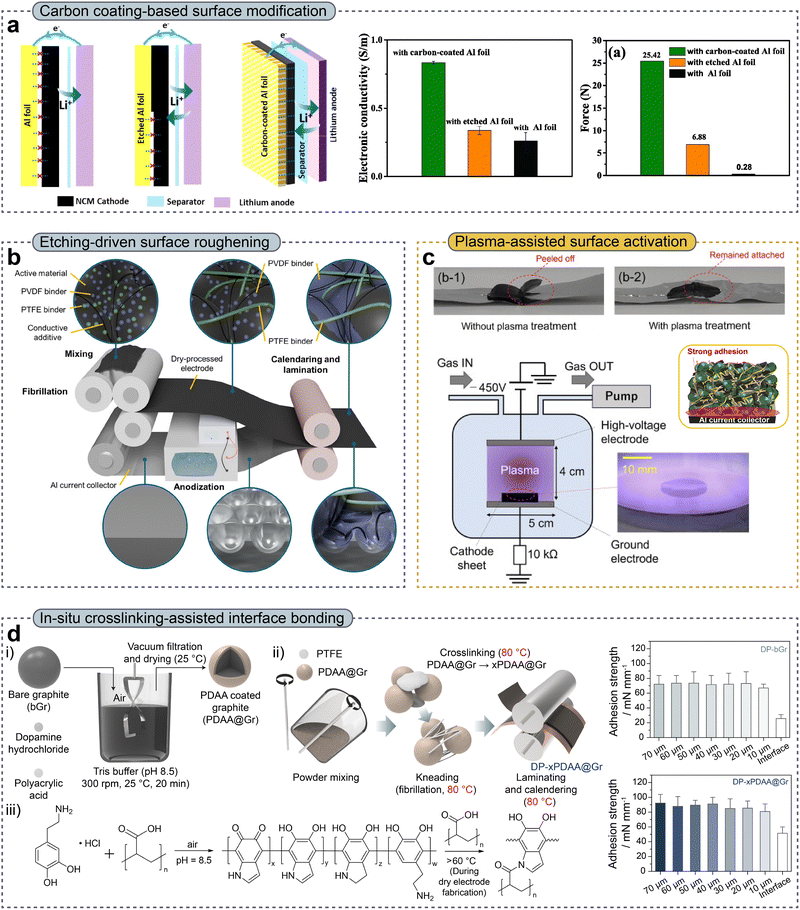 | ||
| Fig. 5 (a) Schematic illustration of electrodes fabricated on various current collectors (left), electronic conductivity (middle), and adhesion force (right). Reproduced with permission from ref. 100. Copyright 2024, Elsevier B.V. (b) Anodization process applied to aluminium current collectors to induce surface roughness. Reproduced with permission from ref. 101. Copyright 2025, Wiley-VCH GmbH. (c) Photographic images of the current collectors with and without plasma treatment (top). Schematic illustration of plasma treatment for surface activation (bottom). Reproduced with permission from ref. 108. Copyright 2025, Elsevier B.V. (d) Schematic illustration of crosslinking-assisted polymer interfacial bonding to reinforce the mechanical robustness of the electrode (left). Adhesion strength of DP-bGr and DP-xPDAA@Gr (right). Reproduced with permission from ref. 15. Copyright 2025, Wiley-VCH GmbH. | ||
The lamination process is a critical step for enhancing the interfacial adhesion between an electrode and a current collector, as the adhesion at the electrode–current collector interface directly influences electronic conductivity and contact resistance, ultimately dictating electrochemical performance. Although carbon coating, etching, plasma treatment, and crosslinking-assisted polymer bonding strategies have improved interfacial adhesion, from a processing perspective, carbon-coated Al current collectors still require systematic investigation to identify suitable conductive additives and binders, optimize the coating thickness, and determine the optimal fraction of carbon-coated Al within the electrode architecture. Moreover, the Al etching process requires optimization to fully exploit its potential in enhancing interfacial adhesion and electronic transport pathways.
3.4. Calendering process
Calendering has long been recognized as a critical post-fabrication step that strongly influences the mechanical integrity and electrochemical performance of battery electrodes. This process is believed to play a multifunctional role by simultaneously determining electrode density, particle–particle contact, adhesion to the current collector, and electronic and ionic transport properties.39,109,110 Beyond porosity reduction, calendering in DPEs performs an additional and essential function by continuing the fibrillation of polymeric binders such as PTFE. Rolling and pressing introduce substantial shear stresses that further extend, align, and redistribute the PTFE fibrils throughout the electrode thickness. This shear-induced fibrillation reinforces the mechanically interlocking binder network that connects active material particles and conductive additives, enhancing electrode cohesion and stabilizing particle–particle contact.32,111–113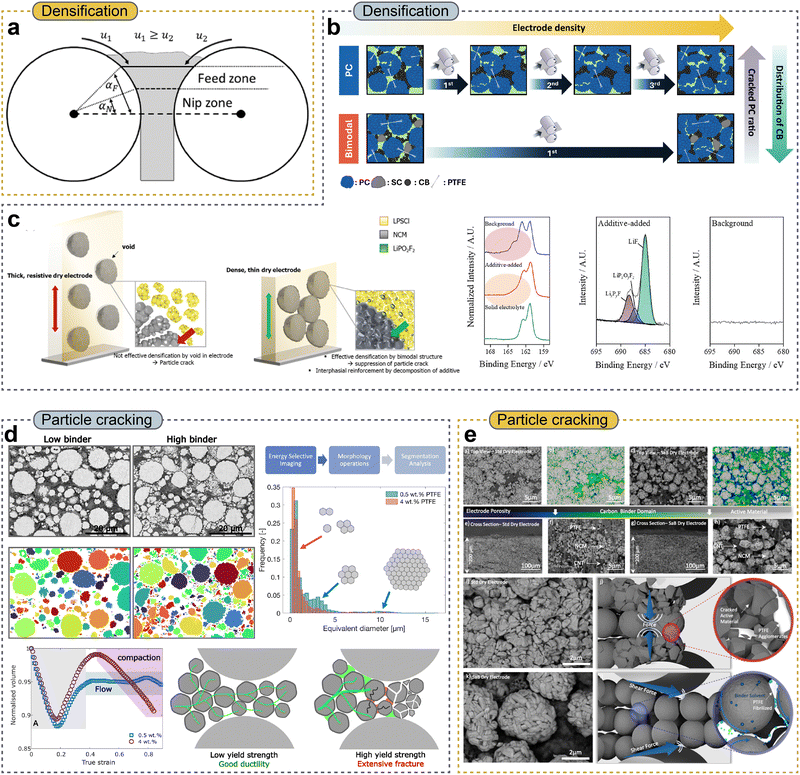 | ||
| Fig. 6 (a) Scheme of a vertical powder calendering process with the distinct gap zones and characteristic parameters. Reproduced with permission from ref. 112. Copyright 2023, American Chemical Society. (b) Schematic illustration of comparison of overall characteristics between PC and bimodal electrodes. Reproduced with permission from ref. 116. Copyright 2025, Elsevier B.V. (c) Scheme of the distinguished ability of LiPO2F2 as an electrode additive, and S 2p and F 1s XPS spectra of background and LiPO2F2-added electrodes after the formation in all-solid-state batteries. Reproduced with permission from ref. 119. Copyright 2023, Wiley-VCH GmbH. (d) Energy-selective backscattered electron micrographs and segmented images of NCM particles for electrodes with low (0.5 wt%) and high (4 wt%) PTFE binder contents (left), corresponding NCM particle size distributions (right), and normalized volume evolution under uniaxial compression with schematic illustrations of deformation behavior during calendering (bottom). Reproduced with permission from ref. 122. Copyright 2025, The Royal Society of Chemistry. (e) SEM images of standard DPEs and solvent-assisted binder (SaB) DPEs, including surface and cross-sectional morphologies (top) and high-magnification cross-sectional views with schematics illustrating microstructural evolution during calendering (bottom). Reproduced with permission from ref. 123. Copyright 2024, Elsevier B.V. | ||
Along with binder properties, the architecture of active materials also greatly determines the densification behavior during calendering.37,115–118 Hong et al.116 systematically demonstrated that bimodal particle-size distributions combining large polycrystalline (PC) and small single-crystalline (SC) particles enhanced packing density by efficiently filling interstitial voids, thereby reducing the number of calendering passes required to reach the target electrode density (Fig. 6b). Importantly, this densification proceeded through geometric rearrangement rather than particle fracture, resulting in a lower fraction of mechanically damaged particles compared to that of unimodal PC electrodes at comparable densities. Furthermore, the higher intrinsic packing density of bimodal electrodes suppressed elastic spring-back during electrochemical cycling, preserving particle–particle contact and electronic percolation networks. These effects collectively lead to improved electronic stability and cycling performance, underscoring the fact that effective densification in calendering arises from the coupled interactions between shear-induced binder fibrillation and particle-scale packing, rather than from compressive stress alone. Similarly, Kim et al.119 have provided direct evidence that conventional DPE preparation intrinsically exhibits incomplete and spatially non-uniform densification during calendering, contributing to persistent microstructural defects such as interparticle voids and particle cracking after rolling (Fig. 6c). In their study, voids were widely spread throughout the mixed powder before calendering, leading to a sparse bulk structure in the as-pressed electrode because the externally applied pressure was not evenly transmitted through the heterogeneous powder bed. Consequently, regions with residual voids exhibited poor mechanical load transfer during calendering. This resulted in persistent porosity as well as the development of microcracks within active materials and at interfacial points of contact. To mitigate these chemo-mechanical shortcomings, a bi-functionalized electrode additive approach was introduced, in which a finely dispersed electrolyte component (Li6PS5Cl) and a lithium difluorophosphate (LiPO2F2) additive were incorporated into the electrode mixture. The bimodal particle size distribution and strategic filling of interparticle voids by the additive facilitated a more uniform pressure distribution under calendering, enhancing powder packing and reducing residual voids and cracking. Additionally, the LiPO2F2 additive readily reacted with the surfaces of the active material during initial electrochemical formation, producing a conformal LiF/LixPFy surface layer, which further improved the chemo-mechanical stability and suppressed solid electrolyte degradation. These findings collectively underscore that achieving homogeneous densification during calendering is a prerequisite for suppressing void formation, ensuring uniform stress transfer, and maintaining the mechanical and electrochemical integrity of DPEs.
In summary, calendering is a critical step in DPE manufacturing and governs electrode densification, binder fibrillation, and mechanical integrity. Effective calendering in DPEs relies on shear-dominated deformation to promote uniform PTFE fibrillation and particle packing. Densification is strongly influenced by binder mechanics and particle architecture, while inadequate stress accommodation leads to non-uniform consolidation and calendering-induced particle cracking. Importantly, particle fracture is not intrinsic to dry processing but can be mitigated by controlling materials design and calendering pathways, allowing for mechanically robust and high-performance DPEs.
4. Advanced binders for next-generation dry-processed electrodes
4.1. Challenges in conventional PTFE binders
PTFE has become one of the most widely adopted binders for DPE owing to its exceptional and unique fibrillation behavior under shear.32,117,124 However, conventional PTFE binders present several fundamental challenges that limit their broader applicability (Fig. 7a). A major challenge is the inherently non-uniform dispersion of PTFE within composite electrodes. In DPE fabrication, binder dispersion is governed primarily by mechanical mixing and shear-driven fibrillation, rather than by solvent-mediated molecular diffusion. Under such conditions, PTFE particles do not always undergo uniform fibrillation throughout the electrode. Instead, localized PTFE agglomerates or uneven fibrillar clusters frequently form, resulting in spatial heterogeneity in binder distribution.57,88,125,126 This non-uniform binder network can induce localized ionic and electronic transport bottlenecks, particularly in regions where excessive PTFE aggregation disrupts continuous conductive pathways or blocks pore channels. Consequently, such structural heterogeneity compromises electrochemical performance by increasing local resistance and limiting the effectiveness of utilizing active materials.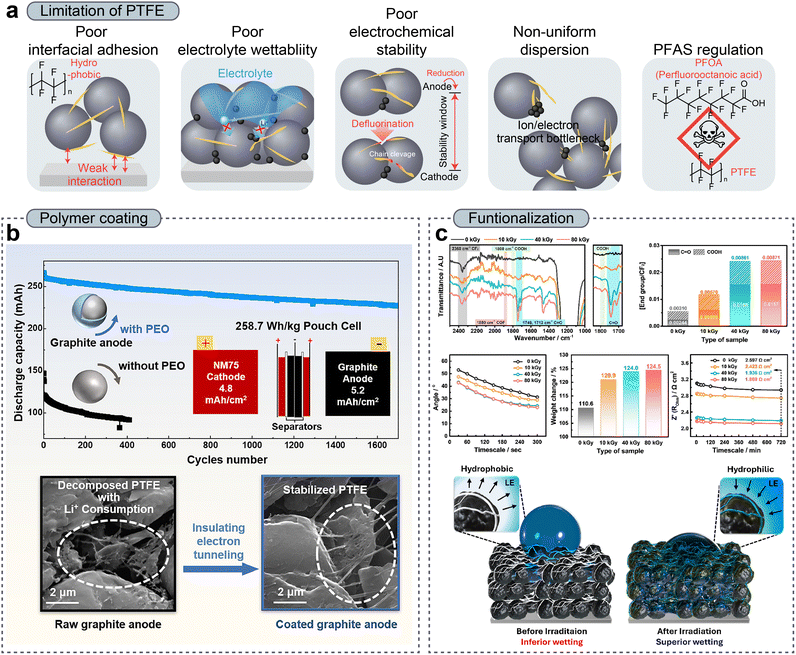 | ||
| Fig. 7 (a) Schematic illustration of challenges in the PTFE binder in DPEs. (b) Prevention of PTFE decomposition through PEO coating on a graphite anode. Reproduced with permission from ref. 129. Copyright 2024, Cell Press. (c) FT-IR spectra, molar ratio of the oxygen group, contact angle, weight change of the electrodes after being soaked for 1 h, and Rohm resistance of the electrodes over time as a function of irradiation dose (top) and schematic of electron beam-induced functionalization of fibrillated PTFE in post-fabricated dry electrodes (bottom). Reproduced with permission from ref. 134. Copyright 2025, Wiley-VCH GmbH. | ||
Furthermore, under low potential, PTFE may be susceptible to reductive decomposition owing to the low lower unoccupied molecular orbital (LUMO) level. When using PTFE as an anode binder, the defluorination of PTFE results in a lower initial coulombic efficiency (ICE) and chain cleavage, which degrade mechanical robustness. To address this, electrolyte additives such as FEC (fluoroethylene carbonate) can be used to form a proactive and stable FEC-derived solid electrolyte interphase (SEI)127,128 and polymer coating on active materials, providing an option for suppressing irreversible PTFE reactions.129,130 As shown in Fig. 7b, Z. Wei et al.129 systematically elucidated the electrochemical reduction and defluorination of PTFE binders in dry-processed graphite anodes and found that a thin PEO coating on graphite particles effectively suppressed PTFE decomposition by electronically decoupling the binder from the anode while maintaining lithium-ion transport, thereby enabling the development of high-loading DPEs with significantly improved ICE and practical pouch-cell performance.
Another critical limitation of PTFE binders is found in their poor interfacial adhesion to active materials and current collectors. As mentioned in the Laminating process section, the chemically inert and nonpolar nature of PTFE composed of C–F bonds results in weak interfacial interactions with metallic current collectors, where adhesion relies mainly on mechanical interlocking rather than on chemical bonding.89,90,131 In DPEs, where the binder content is typically minimized, insufficient interfacial adhesion can lead to delamination at the electrode–current collector interface and reduced mechanical robustness under calendering or cycling-induced stress. This issue becomes particularly problematic in thick, high-loading electrodes, where insufficient interfacial contact not only undermines structural integrity but also increases interfacial contact resistance, thereby limiting effective electronic transport across the electrode thickness. As electrode thickness increases, such interfacial electronic bottlenecks can promote non-uniform current distribution and depth-dependent electrochemical utilization, underscoring the need to address adhesion and interfacial design as critical factors in fabricating thick DPEs.
Beyond adhesion-related concerns, facile electrolyte penetration becomes increasingly critical in thick DPE configurations, where lithium-ion transport must be sustained across through-thickness pathways. While fibrillated PTFE networks can form open-channel microstructures that are more favorable for ion transport compared to conventional WPEs,132–135 the intrinsically low electrolyte affinity of PTFE remains a key limitation. In DPEs, where the porosity and tortuosity are carefully controlled through calendering, poor electrolyte wettability can cancel the benefits of optimized densification by limiting the effectiveness of ionic transport. Moreover, delayed or incomplete wetting can result in increased interfacial resistance, a non-uniform current distribution, and sluggish electrochemical activation. These effects are exacerbated in thick electrodes, where electrolyte penetration is already kinetically constrained. To address this, Won et al.151 proposed a post-fabrication functionalization strategy where fibrillated PTFE binders within DPEs are selectively modified via electron-beam (EB) irradiation to introduce oxygen-containing functional groups such as –COOH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O. This can effectively convert intrinsically hydrophobic PTFE surfaces into electrolyte-affinitive interfaces without compromising fibrillation behavior or mechanical integrity (Fig. 7c). As a result, the EB-treated DPEs exhibited markedly improved electrolyte wettability, enhanced electrolyte uptake, and significantly reduced ionic transport resistance throughout the entire electrode thickness. Importantly, this improvement has enabled uniform lithium-ion migration even in hyper-thick electrodes with mass loadings up to 100 mg cm−2, thereby mitigating through-thickness reaction inhomogeneity and suppressing localized overpotential accumulation. Similarly, Kanno et al.108 used a plasma-assisted dry process to introduce oxygen-containing functional groups (–OH) onto PTFE surfaces. By simply applying plasma treatment to a cathode sheet composed of PTFE, an active material, and a conductive additive, the electrolyte wettability and adhesion were improved, resulting in an enhanced electrochemical performance compared with that of a non-treated cathode. These findings clearly demonstrate that the surface modification of PTFE binders represents a critical design strategy for overcoming the electrolyte wetting limitations of DPEs, particularly under high mass loading conditions.
O. This can effectively convert intrinsically hydrophobic PTFE surfaces into electrolyte-affinitive interfaces without compromising fibrillation behavior or mechanical integrity (Fig. 7c). As a result, the EB-treated DPEs exhibited markedly improved electrolyte wettability, enhanced electrolyte uptake, and significantly reduced ionic transport resistance throughout the entire electrode thickness. Importantly, this improvement has enabled uniform lithium-ion migration even in hyper-thick electrodes with mass loadings up to 100 mg cm−2, thereby mitigating through-thickness reaction inhomogeneity and suppressing localized overpotential accumulation. Similarly, Kanno et al.108 used a plasma-assisted dry process to introduce oxygen-containing functional groups (–OH) onto PTFE surfaces. By simply applying plasma treatment to a cathode sheet composed of PTFE, an active material, and a conductive additive, the electrolyte wettability and adhesion were improved, resulting in an enhanced electrochemical performance compared with that of a non-treated cathode. These findings clearly demonstrate that the surface modification of PTFE binders represents a critical design strategy for overcoming the electrolyte wetting limitations of DPEs, particularly under high mass loading conditions.
Apart from performance-related concerns, environmental recycling and health-related challenges associated with PTFE are becoming increasingly imporant.136–140 PTFE is classified within the broader family of per- and polyfluoroalkyl substances (PFASs), a class of fluorinated compounds which are under increasing regulatory scrutiny due to their extreme environmental persistence, potential for bioaccumulation, and emerging links to human health hazards. Owing to its strong chemical and thermal stability, PTFE is often associated with the “forever chemical” concern, as degradation in the environment is slow and end-of-life handling is difficult.161,162 While this stability is advantageous for electrode integrity, it also creates challenges from a lifecycle perspective, particularly for waste treatment and recycling of PTFE-containing electrodes. As a result, persistent fluorinated components may increase cumulative environmental burden and limit efficient material recovery through recycling routes.163,164 In particular, increasing attention has been directed toward perfluorooctanoic acid (PFOA) and related long-chain PFAS, which were historically used as processing aids or surfactants during PTFE polymerization. Although PFOA is not intentionally present in the final PTFE product, residual traces and lifecycle-related considerations, including emissions during manufacturing and impacts during disposal, have led regulators to evaluate PTFE-containing materials within the broader PFAS framework. In summary, the challenges associated with non-uniform dispersion, interfacial instability, limited electrolyte wettability, electrochemical instability at low electrode potentials, and growing environmental, recycling, and health considerations under PFAS-related regulation support the need to move beyond conventional PTFE binders toward more sustainable material systems for DPEs.
4.2. Dual-binder and beyond-PTFE binder (PFAS-free) systems
While substantial efforts have been made to address the limitations of PTFE-based DPEs through process optimization and interfacial engineering, growing evidence suggests that many of these challenges are intrinsically linked to the fundamental physicochemical nature of PTFE. Consequently, research interest has been directed toward dual-binder strategies and beyond-PTFE, PFAS-free binder systems that provide both mechanical robustness and enhanced electrochemical functionality. Dual-binder systems are effective systems in which PTFE is combined with a secondary polymer binder, i.e., a co-binder. In this approach, PTFE primarily serves as a mechanically robust, fibrillating scaffold that enables solvent-free electrode fabrication, while the co-binder introduces other functionalities. Many of the co-binders employed in dual-binder DPE systems, including PVdF and hydrophilic polymers such as PAA, carboxymethyl cellulose (CMC), and PVP, have been extensively investigated in WPEs and are generally regarded as chemically and electrochemically stable within the practical operating voltage window of LIBs.165–168 Sung et al.90 demonstrated a representative dual-binder strategy that integrates PAA as a hydrogen-bonding co-binder into PTFE-based DPEs. By partially replacing PTFE with PAA while maintaining a low total binder content, a substantial enhancement in adhesion between the freestanding electrode film and the Al current collector was achieved, attributed to hydrogen bonding between the carboxyl groups (–COOH) of PAA and hydroxyl groups (–OH) present on the native Al2O3 layer on the Al current collector (Fig. 8a). This chemical adhesion mechanism stood in sharp contrast to the purely mechanical anchoring provided by PTFE fibrils alone and effectively mitigated interfacial delamination under both calendering-induced stress and long-term electrochemical cycling. Beyond adhesion, the introduction of PAA significantly improved the electrolyte wettability by increasing the surface energy of the electrode and promoting favorable interactions with polar carbonate-based electrolytes. Contact angle measurements and impedance analyses clearly confirm accelerated electrolyte infiltration, reduced interfacial resistance, and enhanced lithium-ion transport throughout the electrode thickness, particularly under high mass loading conditions. Importantly, these interfacial improvements were achieved without compromising the PTFE fibrillation behavior or mechanical integrity, enabling stable electrochemical performance in thick, high-density DPEs. Similarly, He et al.141 introduced a biomass-derived flour additive at low loading (1 wt%) into PTFE-based DPEs to form flour-infused DPEs with enhanced structural and transport properties. The crosslinking between protein and starch components in the flour, in combination with PTFE fibrillation, resulted in the formation of a highly flexible and mechanically resilient powder network with robust load transfer during calendering. Importantly, the modified microstructure exhibited a larger, more interconnected pore architecture that reduced the pore tortuosity in the electrode, thereby improving the ion transport kinetics compared with those of conventional PTFE-only DPEs. DPEs incorporating 1 wt% flour demonstrated superior mechanical integrity and enhanced rate capability and cycling stability, along with the suppressed irreversible phase transitions and intragranular/intergranular crack propagation under aggressive cycling conditions. These findings show that hybrid additive approaches, in which low-cost and sustainable powders are integrated into PTFE fibrillated matrices, can effectively address major bottlenecks in DPE performance by simultaneously reinforcing mechanical cohesion and ion transport behavior. Along with strategies that target interfacial adhesion and wettability, recent efforts have focused on enhancing the electrochemical stability of PTFE-based DPEs by integrating functional co-binders that stabilize the active material–binder interface under reductive conditions.142,143 For example, Lee et al.142 reported a dual-binder system consisting of PTFE and PVP for dry-processed graphite anodes, in which PVP effectively reduced direct contact between PTFE and the anode surface, suppressed PTFE decomposition, and facilitated the formation of a robust, inorganic-rich SEI that enhanced the lithium-ion kinetics and interfacial stability (Fig. 8b). Consequently, DPEs using the PVP/PTFE binary binder achieved an ultrahigh areal capacity (10 mAh cm−2) with significantly improved ICE and prolonged cycling stability compared with those of PTFE-only counterparts.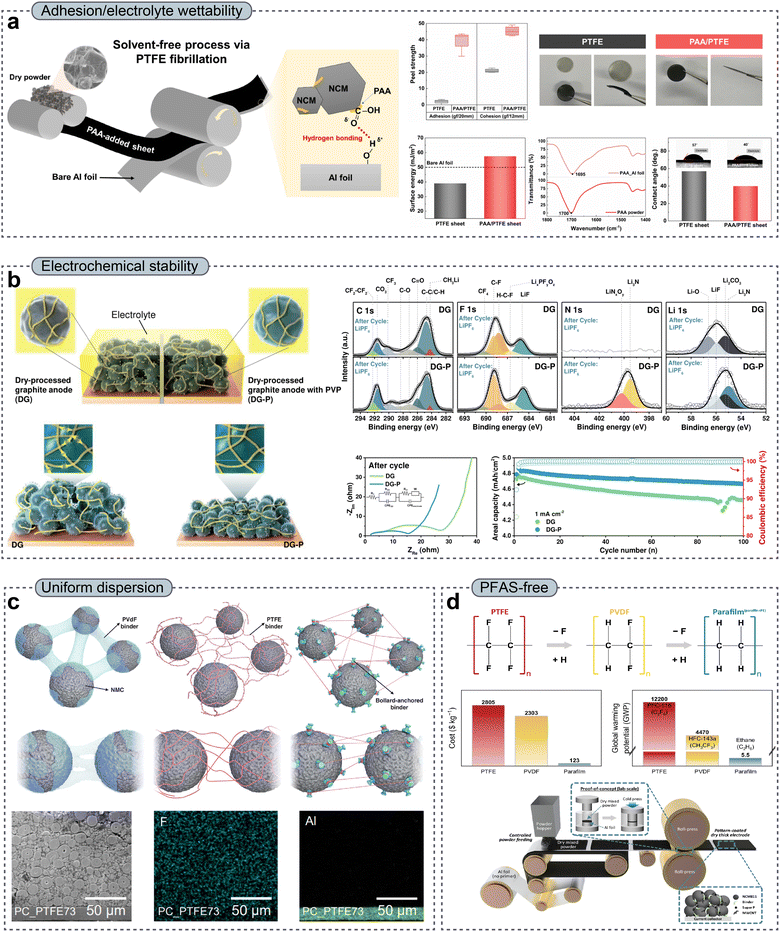 | ||
| Fig. 8 (a) Schematic illustration of the dry process using a PAA/PTFE dual binder and comparative analysis of adhesion properties of PTFE- and PAA/PTFE-based DPEs, correlated with post-cycling electrode morphologies after 100 cycles, surface energies of Al foil and binder sheets, FT-IR evidence of hydrogen bonding interactions, and contact angle measurements. Reproduced with permission from ref. 90. Copyright 2025, Elsevier B.V. (b) PTFE/PVP dual-binder for mitigating direct contact between PTFE and the anode surface, suppressing PTFE decomposition, and facilitating the formation of a robust, inorganic-rich SEI. Reproduced with permission from ref. 142. Copyright 2025, Wiley-VCH GmbH. (c) Schematic images of a cathode with PVdF (wet process), PTFE (dry process), and bollard-anchored dual-binder (dry process) and cross-sectional SEM/EDS images of a bollard-anchored dual-binder-based cathode. Reproduced with permission from ref. 145. Copyright 2024, Elsevier B.V. (d) Comparison of PTFE, PVDF, and Parafilm binders based on the structure, cost, environmental impact, and manufacturing process. Reproduced with permission from ref. 146. Copyright 2025, Springer Nature. | ||
Furthermore, recent studies are increasingly emphasizing the influence of binder dispersion and network uniformity on the mechanical integrity and electrochemical performance of DPEs.144,145 For instance, Kang et al.145 introduced a bollard-anchored dual-binder architecture in which a PAA-grafted-sodium carboxymethyl cellulose (PAA-g-CMC) functioned as a dispersion-controlling “bollard,” effectively anchoring PTFE fibrils and suppressing binder agglomeration (Fig. 8c). This architecture effectively suppressed binder agglomeration by immobilizing PTFE fibrils through multivalent hydrogen bonding and polymer–polymer anchoring, thereby preventing uncontrolled fibril bundling and the development of localized binder-rich domains during dry mixing and calendering. As a result, the dual-binder system promoted a more homogeneous binder network throughout the electrode thickness, significantly improved powder dispersion, and enabled uniform stress and charge transport even at high mass loadings (90 mg cm−2 and 15.6 mAh cm−2), while simultaneously reducing the overall PTFE content.
Building upon dual-binder strategies that partially alleviate the intrinsic limitations of PTFE, emerging studies are focusing on the development of beyond-PTFE, PFAS-free binder systems that can fundamentally replace fluorinated polymers in DPE manufacturing.47,60,146–148 In this context, two distinct material design strategies have been explored, namely PFAS-free binders that retain fibrillation behavior under shear and alternative binders that enable dry processing without relying on fibrillation mechanisms. Regarding the former, Schmidt et al.148 reported a renewable, fluorine-free protein-based binder (sericin) that could form fibrillar networks during shear-induced dry processing, similar to PTFE fibrillation. Despite its lower MW relative to that of PTFE, sericin underwent shear-induced fibril formation, generating a spiderweb-like network that effectively interconnected active materials and conductive additives in solvent-free lithium–sulfur cathodes. Importantly, the intrinsic polarity of the polypeptide binder enhanced the electrolyte wettability and interfacial interactions with polysulfide species, resulting in an electrochemical performance comparable to that of PTFE-based DPEs, even under electrolyte-lean conditions. In addition, this study demonstrated that the fibrillation-enabled mechanical integrity in DPEs is not exclusive to fluoropolymers and can be achieved using biodegradable, PFAS-free macromolecules with appropriate molecular architectures. In contrast, a fundamentally different approach was introduced by Kim et al.,146 who demonstrated fluorine-free DPEs based on a thermoplastic binder (Parafilm® M) that does not rely on fibrillation for mechanical cohesion. Instead, Parafilm, composed of paraffin and polyethylene, exploits its low Tg and thermoplastic flow behavior to establish interparticle bonding through mild pressure activation during dry pressing and calendering (Fig. 8d). This binder enabled primer-free adhesion to current collectors, a uniform binder distribution, and robust mechanical integrity without the formation of fibrillar networks. Notably, the absence of fluorinated moieties resulted in a wider electrochemical stability window, particularly at low potentials, while simultaneously addressing PFAS-related environmental and regulatory concerns. The successful fabrication of thick electrodes exceeding 5–9 mAh cm−2 along with long-term cycling stability underscores the fact that non-fibrillating, thermoplastic binders represent a viable and scalable alternative to PTFE-dominated DPE technologies. Similarly, Kim et al.132 designed PFAS-free DPEs based on styrene–butadiene rubber (SBR), which acts as a sticky adhesive during high-pressure calendering. The resulting DPEs exhibited an open-channel interfacial structure that promoted the formation of a favorable SEI rich in LiF, thereby enhancing cycling stability even at an ultrahigh areal loading of 10.6 mAh cm−2.
Collectively, the various dual-binder and beyond-PTFE (PFAS-free) binder systems reviewed in this section demonstrate that the challenges of DPE manufacturing can be addressed through multiple, function-oriented material design strategies rather than by relying on PTFE alone. To facilitate a systematic comparison, Table 260,90,132,141–143,145,146,148–150 summarizes representative binder systems with respect to their primary functional roles, the corresponding DPE fabrication approaches, and the resulting electrochemical performance, particularly in terms of capacity retention during cycling, thus providing a perspective on the effectiveness and scalability of emerging binder technologies for DPEs.
| (a) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Classification | System | Function | Approach | Electrode composition (wt%) | Ref. | |||
| Active material | Conductive additive | Binder | ||||||
| Dual binder | PTFE/PAA | NCM811 cathode | Improve adhesion and electrolyte wettability via polar functional groups of PAA | Twin-screw kneading of the mixed powder | 96.8(NCM811) | 1.2(carbon black) | 2[PTFE/PAA = 1/1(w/w)] | 90 |
| PTFE/High-gluten flour | NCM811, LCO cathode | Improve mechanical robustness via flour crosslinking | Hot-pressing of powder mixture | 90(LCO/NCM811) | 5[Super P/carbon fiber = 4/1(w/w)] | 5[PTFE/flour = 4/1(w/w)] | 141 | |
| PTFE/PAA-g-CMC | NCM622 cathode | (1) Anchoring PTFE to the electrode surface | (1) Pre-anchoring of PAA-g CMC | 93(NCM622) | 5(Super P) | 2[PTFE/PAA-g-CMC = 3/7(w/w)] | 145 | |
| (2) Improve Li ion transport and adhesion | (2) binder onto NCM particles | |||||||
| (3) PTFE-assisted fibrillation | ||||||||
| (4) Kneading and calendering | ||||||||
| PTFE/PVdF | Graphite anode | Functional binder when PTFE decomposes in the first lithiation | (1) PVdF-assisted powder premixing | 90(graphite) | 5(carbonblack) | 5[PTFE/PVdF = 3/2(w/w)] | 149 | |
| (2) PTFE-induced fibrillation | ||||||||
| (3) Hot calendering | ||||||||
| (4) Lamination | ||||||||
| PTFE/PVP | Graphite anode | (1) Reducing PTFE decomposition | (1) Powder premixing | 95.5(graphite) | 1(carbon black) | 3.5[PTFE/PVP = 6/1(w/w)] | 142 | |
| (2) Enhancing mechanical robustness | (2) Kneading and calendering | |||||||
| (3) Formation of a robust Li3N-rich SEI layer | (3) Lamination | |||||||
| PTFE/CMC | Graphite anode | (1) Reducing PTFE decomposition | Quasi-dry processing using ethanol to uniformly integrate CMC | 97(graphite) | 1(Super P) | 2[PTFE/CMC = 1/1(w/w)] | 143 | |
| (2) Improve Li ion transport and adhesion | ||||||||
| PFAS-free | Phenoxy resin | NCM811 cathode | Formation of a continuous and ductile binder network | Hot-pressing a powder composite using a press machine | 100(NCM811) | 1.5(CNT) | 3(phenoxy resin) | 150 |
| Sacrificial binder (PP, paraffin wax, stearic acid) | LFP cathode | Rheological control and structural support during forming, followed by debinding to create a porous, high–active-material electrode | Powder extrusion molding (PEM) technology | — | — | — | 60 | |
| Parafilm | NCM811 cathode | (1) Cost effectiveness | Dry mixed powder can be directly applied onto current collector and pressed into electrodes | 97(NCM811) | 2(Super P) | 1(Parafilm® M) | 146 | |
| (2) High electrochemical stability at low potentials | ||||||||
| (3) Providing natural adhesion without an additional primer coating | ||||||||
| Styrene–butadiene rubber(SBR) | Graphite anode | (1) Sticky adhesive in high-pressure-based calendering | (1) Synthesizing graphite composite powder via freeze-drying | 95(natural graphite) | 1[PTFE/Super P= 1/9(w/w)] | 4(SBR) | 132 | |
| (2) Elastic scaffold within the electrode | (2) Calendering between the two rollers in the nip zone | |||||||
| Sericin | Sulfur/carbon cathode | (1) High electrochemical stability at low potentials | Fibrillation through DRYtraec® process | 60(sulfur) | 37(ketjen black) | 3(sericin) | 148 | |
| (2) Shear induced fibrillation enabling interconnection of active materials | ||||||||
| (3) Improve electrolyte wettability via polar functional groups | ||||||||
| (b) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Classification | System | Function | Approach | Electrochemical performance | Ref. | ||||||
| Loading level (mg cm−2) | Areal capacity(mAh cm−2) | Cell configuration | Voltage range | C-rate@temperature | Capacity retention@cycle | ||||||
| Dual binder | PTFE/PAA | NCM811 cathode | Improve adhesion and electrolyte wettability via polar functional groups of PAA | Twin-screw kneading of the mixed powder | 30(NCM811) | ∼6 | Graphite+Si/C||NCM811 | 2.5–4.25 V | 1C@RT | 82.2%@300 cycles (PTFE/PAA)/63.5%@300 cycles (PTFE) | 90 |
| PTFE/High-gluten flour | NCM811, LCO cathode | Improve mechanical robustness via flour crosslinking | Hot-pressing of powder mixture | 10(LCO) / 9.0 (NCM811) | ∼1.4 (LCO) / ∼1.6 (NCM811) | Li metal||LCO /Li metal||NCM811 | 3.0–4.3 V /3.0–4.5 V | 0.5C / 2C@RT | 79.2%@560 cycle/80.3%@260 cycles | 141 | |
| PTFE/PAA-g-CMC | NCM622 cathode | (1) Anchoring PTFE to the electrode surface | (1) Pre-anchoring of PAA-g CMC | 90(NCM622) | 15.6 | Graphite|| NCM622 | 2.7–4.25 V | 0.5C@25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
84%@50 cycles | 145 | |
| (2) Improve Li ion transport and adhesion | (2) binder onto NCM particles | ||||||||||
| (3) PTFE-assisted fibrillation | |||||||||||
| (4) Kneading and calendering | |||||||||||
| PTFE/PVdF | Graphite anode | Functional binder when PTFE decomposes in the first lithiation | (1) PVdF-assisted powder premixing | 15(graphite) | ∼4.6 | Li metal||graphite | 0.01–1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V V |
0.23mA cm−2@RT | 95%@50 cycles | 149 | |
| (2) PTFE-induced fibrillation | |||||||||||
| (3) Hot calendering | |||||||||||
| (4) Lamination | |||||||||||
| PTFE/PVP | Graphite anode | (1) Reducing PTFE decomposition | (1) Powder premixing | - | 4.8 / 5.7 | Li metal|graphite / graphite||NCM811 | 0.05–1.5 V /2.8–4.25 V | 0.2C@25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
97.4%@100 cycles (PTFE/PVP), 94.3%@100 cycles (PTFE) / 93.0%@100 cycles (PTFE/PVP), 87.0%@100 cycles (PTFE) | 142 | |
| (2) Enhancing mechanical robustness | (2) Kneading and calendering | ||||||||||
| (3) Formation of a robust Li3N-rich SEI layer | (3) Lamination | ||||||||||
| PTFE/CMC | Graphite anode | (1) Reducing PTFE decomposition | Quasi-dry processing using ethanol to uniformly integrate CMC | 30(graphite) | ∼10 | Graphite||NCM811 | 2.7–4.3 V | 0.5C@RT | 48.96%@100 cycles (PTFE/CMC) | 143 | |
| (2) Improve Li ion transport and adhesion | 19.49%@100 cycles (PTFE) | ||||||||||
| PFAS-free | Phenoxy resin | NCM811 cathode | Formation of a continuous and ductile binder network | Hot-pressing a powder composite using a press machine | ∼40.0(NCM811) | ∼7.7 | Li metal||NCM811 | 3.0–4.35 V | 0.1C@25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
73.5%@50 cycles (dry phenoxy resin) | 150 |
| 63.0%@50 cycles (wet phenoxy resin) | |||||||||||
| Sacrificial binder (PP, paraffin wax, stearic acid) | LFP cathode | Rheological control and structural support during forming, followed by debinding to create a porous, high–active-material electrode | Powder extrusion molding (PEM) technology | — | 13.7 | Li metal||LFP | 2.0–4.0 V | 0.1C@20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) °C °C |
Good cyclability over 20 cycles | 60 | |
| Parafilm | NCM811 cathode | (1) Cost effectiveness | Dry mixed powder can be directly applied onto current collector and pressed into electrodes | 36.8(NCM811) | 6.6 (1st cycle) | Graphite||NCM811 | 3.0–4.3 V | 0.3C@25 °C | 63.1%@1000 cycles | 146 | |
| (2) High electrochemical stability at low potentials | |||||||||||
| (3) Providing natural adhesion without an additional primer coating | |||||||||||
| Styrene–butadiene rubber(SBR) | Graphite anode | (1) Sticky adhesive in high-pressure-based calendering | (1) Synthesizing graphite composite powder via freeze-drying | 45(LFP) | 5.6 (1st cycle) | Graphite||LFP | 2.45–3.9 V | 0.5C | 74.6%@200 cycles | 132 | |
| (2) Elastic scaffold within the electrode | (2) Calendering between the two rollers in the nip zone | ||||||||||
| Sericin | Sulfur/carbon cathode | (1) High electrochemical stability at low potentials | Fibrillation through DRYtraec® process | 2.5 ± 0.2(sulfur) | — | Li metal||Sulfur | 1.5–2.5 V | 0.1C@25 ± 1 °C | 836 mAh g−1@50 cycles (sericin) | 148 | |
| (2) Shear induced fibrillation enabling interconnection of active materials | 695 mAh g−1@50 cycles (PTFE) | ||||||||||
| (3) Improve electrolyte wettability via polar functional groups | |||||||||||
5. Summary and outlook: advanced materials and processes for scalable dry-processed electrode manufacturing
DPE manufacturing provides an alternative electrode fabrication route for LIBs by removing conventional solvent-dependent processing steps. As systematically reviewed in this paper, the primary advantages of dry processing (i.e., environmental sustainability, reduced manufacturing cost, and high coating speed) originate from the elimination of solvent handling and drying steps. However, as schematically illustrated in Fig. 9, the practical application of DPEs critically depends on optimization at both the process and material levels.Regarding processing perspectives, each step of DPE manufacturing, including powder mixing, kneading, sheet forming, laminating, and calendering, plays a critical role in determining the electrode microstructure. Non-uniform powder mixing leads to local heterogeneity in the distribution of binders and conductive additives, which subsequently manifests as spatial variations in electronic conductivity and mechanical strength. Similarly, insufficient or non-uniform PTFE fibrillation during kneading compromises the formation of a continuous fibrous binder network, resulting in poor cohesion within the bulk electrode, as well as weak adhesion at the electrode/current collector interface. Lamination and calendering, while essential for densification and interfacial contact, can introduce further constraints, such as crack formation and density gradients, when the mechanical compliance of the electrode film is not properly matched to the applied stress. These results show that process control plays a critical role in both microstructural stability and electrochemical performance.
Meanwhile, materials-level design also plays an important role in DPE performance. The fibrillation behavior of PTFE binders, for instance, is intrinsically related to the MW, particle size, and crystallinity, which, in turn, determines the robustness of the fibrous network formed during shear-driven processing. Conductive additives with different dimensions and surface energies respond differently to dry mixing and kneading, influencing percolation pathways and contact resistance. Likewise, the choice of the active material or polymeric binder affects electrochemical stability and mechanical stress accommodation during lamination and calendering. Importantly, these material properties collectively define the processing window for obtaining uniform microstructures.
The intrinsic limitations of PTFE-based DPEs highlight the need for next-generation binder concepts. From a scale-up perspective, thicker and higher-loading DPE architectures may require higher absolute binder input to maintain mechanical integrity and processability, even when the binder fraction is controlled and optimized. This trend can amplify the environmental, recycling, and regulatory burden of fluorinated binder systems at the cell-manufacturing level. Therefore, future binder design should target not only fibrillation capability and electrode robustness but also reduced environmental impact and safer material pathways. In this context, dual-binder strategies—in which PTFE serves as a mechanically robust fibrillating scaffold while secondary polymers introduce complementary functions—demonstrate that key performance bottlenecks in DPEs can be effectively addressed through synergistic materials design. Emerging PTFE-lean or PTFE-free systems, including co-binder systems and non-fluorinated polymer networks, provide an important transition route. In particular, bio-derived fibrillar candidates such as cellulose nanofibers (CNFs)89,91,169 and protein-based fibroin170,171 are attracting attention due to their potential to form mechanically supportive networks while offering improved environmental compatibility. More broadly, beyond-PTFE (PFAS-free) systems—including fibrillating fluorine-free macromolecules and non-fibrillating thermoplastic binders—further indicate that mechanical cohesion and scalable dry processing can be achieved through fundamentally different mechanisms.
In summary, scalable and mechanically robust DPE manufacturing should be treated as an integrated material–process–microstructure engineering problem. Only by simultaneously optimizing processing routes and materials design, while accounting for electrochemical performance, manufacturability, and sustainability, can DPEs realize their full potential as a cornerstone technology for next-generation battery manufacturing.
Author contributions
G. Nam: conceptualization, investigation, methodology, data curation, writing – original draft; J. Lim: conceptualization, investigation, visualization, writing – original draft; S. Choi: conceptualization, investigation, visualization, data curation, writing – original draft; S. C. Nam: funding acquisition, writing – review and editing; K. Hong: funding acquisition, writing – review and editing; J. Lee: funding acquisition, writing – review and editing; Y. M. Lee: conceptualization, supervision, project administration, funding acquisition, writing – original draft, writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This work was supported by the Korea Institute for Advancement of Technology (KIAT) grant funded by the Technology Innovation Program funded by the Ministry of Trade, Industry & Energy (MOTIE, Korea) (No. 2410009726 (RS-2025-00437259) and No. 2410010799 (RS-2025-00420590), HRD program for industrial innovation). This study was also supported by POSCO HOLDINGS, Korea. We also acknowledge the use of ChatGPT (version 5.2) solely for language editing purposes to improve linguistic quality and readability. The authors take full responsibility for the scientific content of the article and have carefully read and checked the manuscript to ensure technical accuracy.Notes and references
- T. L. Sheldon and R. Dua, Energy Res. Soc. Sci., 2025, 127, 104327 CrossRef.
- A. Kumar, Energy Storage, 2024, 6, e70000 CrossRef.
- R. Schmuch, R. Wagner, G. Hörpel, T. Placke and M. Winter, Nat. Energy, 2018, 3, 267–278 CrossRef CAS.
- A. Masias, J. Marcicki and W. A. Paxton, ACS Energy Lett., 2021, 6, 621–630 CrossRef CAS.
- J. Deng, C. Bae, A. Denlinger and T. Miller, Joule, 2020, 4, 511–515 CrossRef.
- Y. Liu, Y. Zhu and Y. Cui, Nat. Energy, 2019, 4, 540–550 CrossRef.
- A. Kwade, W. Haselrieder, R. Leithoff, A. Modlinger, F. Dietrich and K. Droeder, Nat. Energy, 2018, 3, 290–300 CrossRef.
- W. Li, Z. He, Y. Jie, F. Huang, Y. Chen, Y. Wang, W. Zhang, X. Zhu, R. Cao and S. Jiao, Adv. Funct. Mater., 2024, 34, 2406770 CrossRef CAS.
- H.-H. Ryu, H. H. Sun, S.-T. Myung, C. S. Yoon and Y.-K. Sun, Energy Environ. Sci., 2021, 14, 844–852 RSC.
- Z. Liu, Y. Zhang, S. Pan, Y. Chen, K. Yang, S. Wu, M. Liu, L. Hu, S. Jiang, X. Wang, G. Wang and M. Yao, Energy Environ. Sci., 2026, 15, 883 Search PubMed.
- J. Zheng, M. S. Kim, Z. Tu, S. Choudhury, T. Tang and L. A. Archer, Chem. Soc. Rev., 2020, 49, 2701–2750 RSC.
- N. Kim, Y. Kim, J. Sung and J. Cho, Nat. Energy, 2023, 8, 921–933 CrossRef CAS.
- J. Li, J. Fleetwood, W. B. Hawley and W. Kays, Chem. Rev., 2022, 122, 903–956 CrossRef CAS PubMed.
- J. Lim, S. Park, H. Lee, S. Choi, G. Nam, K.-G. Kim, J. Choi, Y.-G. Lee and Y. M. Lee, J. Energy Chem., 2025, 105, 87–95 CrossRef CAS.
- J. Lim, K. Kang, S. Choi, M. Song, W. Yang, G. Nam, M. Kwon, R. Hong, D. Kang, H. Kim and Y. M. Lee, Carbon Neutralization, 2025, 4, e70050 CrossRef CAS.
- J. Lim, D. Kang, C. Bak, S. Choi, M. Lee, H. Lee and Y. M. Lee, Nano-Micro Lett., 2025, 18, 75 CrossRef PubMed.
- J. Kang, J. Lim, H. Lee, S. Park, C. Bak, Y. Shin, H. An, M. Lee, M. Lee, S. Lee, B. Choi, D. Kang, S. Chae, Y. M. Lee and H. Lee, Adv. Sci., 2024, 11, 2403071 CrossRef CAS PubMed.
- J. Wu, X. Zhang, Z. Ju, L. Wang, Z. Hui, K. Mayilvahanan, K. J. Takeuchi, A. C. Marschilok, A. C. West, E. S. Takeuchi and G. Yu, Adv. Mater., 2021, 33, 2101275 CrossRef CAS PubMed.
- Y. Kuang, C. Chen, D. Kirsch and L. Hu, Adv. Energy Mater., 2019, 9, 1901457 CrossRef.
- H. Kang, J.-H. Kim, J. Lim, C. Heo, K.-S. Oh, H. Moon, S. Choi, J. Nam, Y. M. Lee and S.-Y. Lee, ACS Energy Lett., 2025, 10, 6223–6235 CrossRef CAS.
- Y. S. Zhang, N. E. Courtier, Z. Zhang, K. Liu, J. J. Bailey, A. M. Boyce, G. Richardson, P. R. Shearing, E. Kendrick and D. J. L. Brett, Adv. Energy Mater., 2022, 12, 2102233 CrossRef CAS.
- T. Lombardo, A. C. Ngandjong, A. Belhcen and A. A. Franco, Energy Storage Mater., 2021, 43, 337–347 CrossRef.
- J. Entwistle, R. H. Ge, K. Pardikar, R. Smith and D. Cumming, Renew. Sustain. Energy Rev., 2022, 166, 112624 CrossRef CAS.
- Y. Liu, R. Zhang, J. Wang and Y. Wang, iScience, 2021, 24, 102332 CrossRef CAS PubMed.
- T. Krüger, H. Gruhn, M. Mund, K. Dilger and M. W. Kandula, Energy Technol., 2025, 13, 2402221 CrossRef.
- H. Liu, X. Cheng, Y. Chong, H. Yuan, J.-Q. Huang and Q. Zhang, Particuology, 2021, 57, 56–71 CrossRef CAS.
- R. Sliz, J. Valikangas, H. Silva Santos, P. Vilmi, L. Rieppo, T. Hu, U. Lassi and T. Fabritius, ACS Appl. Energ. Mater., 2022, 5, 4047–4058 CrossRef CAS PubMed.
- S. Ahmed, P. A. Nelson, K. G. Gallagher and D. W. Dees, J. Power Sources, 2016, 322, 169–178 CrossRef CAS.
- Y. H. Liang, H. Long, Z. M. Huang, T. He, X. M. Chen, M. Li, H. Chen and S. Q. Zhang, Adv. Funct. Mater., 2025, e18619 Search PubMed.
- Y. Lu, C.-Z. Zhao, H. Yuan, J.-K. Hu, J.-Q. Huang and Q. Zhang, Matter, 2022, 5, 876–898 CrossRef CAS.
- R. Tao, Y. Gu, Z. Du, X. Lyu and J. Li, Nat. Rev. Clean Technol., 2025, 1, 116–131 CrossRef.
- W. Jin, G. Song, J.-K. Yoo, S.-K. Jung, T.-H. Kim and J. Kim, ChemElectroChem, 2024, 11, e202400288 CrossRef CAS.
- Y. Bi, Q. Li, R. Yi and J. Xiao, J. Electrochem. Soc., 2022, 169, 020521 CrossRef CAS.
- F. Degen, M. Winter, D. Bendig and J. Tübke, Nat. Energy, 2023, 8, 1284–1295 CrossRef CAS.
- H. Zhang, B. Xue, S. Li, Y. Yu, X. Li, Z. Chang, H. Wu, Y. Hu, K. Huang, L. Liu, L. Chen and Y. Su, Sci. Rep., 2023, 13, 7952 CrossRef CAS PubMed.
- D. L. Wood, J. D. Quass, J. Li, S. Ahmed, D. Ventola and C. Daniel, Drying Technol., 2018, 36, 234–244 CrossRef CAS.
- M. Abdollahifar, H. Cavers, S. Scheffler, A. Diener, M. Lippke and A. Kwade, Adv. Energy Mater., 2023, 13, 2300973 CrossRef CAS.
- S. Choi, J. P. Seo, J. Lim, C. B. Dzakpasu, Y. Roh, C. Bak, S. Kim, H. Lee and Y. M. Lee, Batter. Supercaps, 2025, 8, e202400638 CrossRef CAS.
- J. Lim, J. Song, K.-G. Kim, J. K. Koo, H. Lee, D. Kang, Y.-J. Kim, J. Park and Y. M. Lee, Small, 2025, 21, 2410485 CrossRef CAS PubMed.
- M. Ebner, D.-W. Chung, R. E. García and V. Wood, Adv. Energy Mater., 2014, 4, 1301278 CrossRef.
- S. Hein, T. Danner, D. Westhoff, B. Prifling, R. Scurtu, L. Kremer, A. Hoffmann, A. Hilger, M. Osenberg, I. Manke, M. Wohlfahrt-Mehrens, V. Schmidt and A. Latz, J. Electrochem. Soc., 2020, 167, 013546 CrossRef.
- T.-T. Nguyen, A. Demortière, B. Fleutot, B. Delobel, C. Delacourt and S. J. Cooper, npj Comput. Mater., 2020, 6, 123 CrossRef CAS.
- P. Mitchell, L. Zhong and X. Xi, Recyclable dry particle based adhesive electrode and methods of making same, US Pat., US7342770B2, 2008 Search PubMed.
- M. Ank, A. Sommer, K. Abo Gamra, J. Schöberl, M. Leeb, J. Schachtl, N. Streidel, S. Stock, M. Schreiber, P. Bilfinger, C. Allgäuer, P. Rosner, J. Hagemeister, M. Rößle, R. Daub and M. Lienkamp, J. Electrochem. Soc., 2023, 170, 120536 CrossRef CAS.
- X. Han, M. R. Funk, F. Shen, Y.-C. Chen, Y. Li, C. J. Campbell, J. Dai, X. Yang, J.-W. Kim, Y. Liao, J. W. Connell, V. Barone, Z. Chen, Y. Lin and L. Hu, ACS Nano, 2014, 8, 8255–8265 CrossRef CAS.
- M. Ryu, Y.-K. Hong, S.-Y. Lee and J. H. Park, Nat. Commun., 2023, 14, 1316 CrossRef CAS.
- M. E. Sotomayor, C. D. L. Torre-Gamarra, W. Bucheli, J. M. Amarilla, A. Varez, B. Levenfeld and J. Y. Sanchez, J. Mater. Chem. A, 2018, 6, 5952–5961 RSC.
- S. El Khakani, N. Verdier, D. Lepage, A. Prébé, D. Aymé-Perrot, D. Rochefort and M. Dollé, J. Power Sources, 2020, 454, 227884 CrossRef CAS.
- S. Shiraki, H. Oki, Y. Takagi, T. Suzuki, A. Kumatani, R. Shimizu, M. Haruta, T. Ohsawa, Y. Sato, Y. Ikuhara and T. Hitosugi, J. Power Sources, 2014, 267, 881–887 CrossRef CAS.
- J. Liu, B. Ludwig, Y. Liu, Z. Zheng, F. Wang, M. Tang, J. Wang, J. Wang, H. Pan and Y. Wang, Adv. Mater. Technol., 2017, 2, 1700106 CrossRef.
- Y. Liu, X. Gong, C. Podder, F. Wang, Z. Li, J. Liu, J. Fu, X. Ma, P. Vanaphuti, R. Wang, A. Hitt, Y. Savsatli, Z. Yang, M. Ge, W.-K. Lee, B. Yonemoto, M. Tang, H. Pan and Y. Wang, Joule, 2023, 7, 952–970 CrossRef CAS.
- H. Abe, A. Kondo, M. Naito, T. Wakimoto and M. Yamaguchi, Electrostatic dry coating of cathode materials for Li ion battery, in: 9th IMAPS/ACerS International Conference and Exhibition on Ceramic Interconnect and Ceramic Microsystems Technologies, CICMT 2013, Orlando, FL, 2013.
- B. Trembacki, E. Duoss, G. Oxberry, M. Stadermann and J. Murthy, J. Electrochem. Soc., 2019, 166, A923 CrossRef CAS.
- R. W. Gore, Process for producing porous products, US Pat., US3953566A, 1976 Search PubMed.
- H. Oh, G.-S. Kim, B. U. Hwang, J. Bang, J. Kim and K.-M. Jeong, Chem. Eng. J., 2024, 491, 151957 CrossRef CAS.
- H. Oh, G.-S. Kim, J. Bang, S. Kim and K.-M. Jeong, Energy Environ. Sci., 2025, 18, 645–658 RSC.
- J. K. Koo, J. Lim, J. Shin, J. K. Seo, C. Ha, W. T. A. Ran, J.-H. Lee, Y. Kwon, Y. M. Lee and Y.-J. Kim, Energy Storage Mater., 2025, 78, 104270 CrossRef.
- G.-T. Kim, G. B. Appetecchi, F. Alessandrini and S. Passerini, J. Power Sources, 2007, 171, 861–869 CrossRef CAS.
- L. Helmers, L. Froböse, K. Friedrich, M. Steffens, D. Kern, P. Michalowski and A. Kwade, Energy Technol., 2021, 9, 2000923 CrossRef CAS.
- C. de la Torre-Gamarra, M. E. Sotomayor, J.-Y. Sanchez, B. Levenfeld, A. Várez, B. Laïk and J.-P. Pereira-Ramos, J. Power Sources, 2020, 458, 228033 CrossRef CAS.
- L. Yuan, H. Liu and X. Jiang, J. Energy Storage, 2024, 90, 111912 CrossRef CAS.
- D.-W. Park, N. A. Cañas, N. Wagner and K. A. Friedrich, J. Power Sources, 2016, 306, 758–763 CrossRef CAS.
- M. Al-Shroofy, Q. Zhang, J. Xu, T. Chen, A. P. Kaur and Y.-T. Cheng, J. Power Sources, 2017, 352, 187–193 CrossRef CAS.
- Z. Du, C. J. Janke, J. Li, D. L. Wood III and C. Daniel, Method of solvent-free manufacturing of composite electrodes incorporating radiation curable binders, US Pat., US11289689B2, 2022 Search PubMed.
- J. Park, H. Oh, J.-H. Lim, S. Jung, N. Yoo, J.-K. Yoo, K.-M. Jeong and K.-Y. Park, Adv. Energy Mater., 2026, 16, e04005 CrossRef CAS.
- C.-C. Chang, L.-J. Her, H.-K. Su, S.-H. Hsu and Y. T. Yen, J. Electrochem. Soc., 2011, 158, A481 CrossRef CAS.
- C.-C. Chang, H.-K. Su, L.-J. Her and J.-H. Lin, J. Chin. Chem. Soc., 2012, 59, 1233–1237 CrossRef CAS.
- Y. Kim, J. S. Hong, S. Y. Moon, J.-Y. Hong and J. U. Lee, Carbon Lett., 2021, 31, 1327–1337 CrossRef.
- Y. J. Choi, E. J. C. Nacpil, J. Han, C. Zhu, I. S. Kim and I. Jeon, Adv. Energy Sustain. Res., 2024, 5, 2300219 CrossRef CAS.
- Q. Zhang, J.-Q. Huang, W.-Z. Qian, Y.-Y. Zhang and F. Wei, Small, 2013, 9, 1237–1265 CrossRef CAS PubMed.
- B. Ludwig, Z. Zheng, W. Shou, Y. Wang and H. Pan, Sci. Rep., 2016, 6, 23150 CrossRef CAS PubMed.
- H. Kim, J. H. Lim, T. Lee, J. An, H. Kim, H. Song, H. Lee, J. W. Choi and J. H. Kang, ACS Energy Lett., 2023, 8, 3460–3466 CrossRef CAS.
- I. Hwang, K.-E. Sung, J. Hong, G.-S. Kang and J. Yoon, Chem. Eng. J., 2025, 506, 160159 CrossRef CAS.
- C. N. Thi Linh, V. D. Thuc, D. D. Mai, M. C. Nguyen, D. T. Pham, W. J. Yu and D. Kim, Chem. Eng. J., 2025, 509, 161183 CrossRef CAS.
- S. Z. Damnali, B. Villard, M. Leeb, B. Tekin, T. Woehrle, R. Daub, A. Hoffmann and M. Hoelzle, J. Power Sources, 2026, 663, 238924 CrossRef CAS.
- K. Lee, Y. Jo, J. Seok Nam, H. Yu and Y.-J. Kim, Chem. Eng. J., 2024, 487, 150221 CrossRef CAS.
- J. Kim, K. Park, M. Kim, H. Lee, J. Choi, H. B. Park, H. Kim, J. Jang, Y.-H. Kim, T. Song and U. Paik, Adv. Energy Mater., 2024, 14, 2303455 CrossRef CAS.
- S. Jang, Y. Kang, H. Kim, J. Park and K. T. Lee, Small Struct., 2025, 6, 2400350 CrossRef CAS.
- J. Yoon, D. H. Kang, S. Shin, S. Yoo, H. Kim, J.-K. Lee, H.-J. Jin and Y. S. Yun, Adv. Funct. Mater., 2025, e22855 CrossRef.
- X. Shen, H. Yu, L. Ben, W. Zhao, Q. Wang, G. Cen, R. Qiao, Y. Wu and X. Huang, J. Energy Chem., 2024, 90, 133–143 CrossRef CAS.
- H. Choi, D. Moon, J. Sheem, J. K. Koo, S. Hong, S.-M. Oh and Y.-J. Kim, J. Electrochem. Soc., 2023, 170, 090511 CrossRef CAS.
- Z. Zhou, K. Lu, F. Chen, Y. Sun, X. Liu, Y. Niu, Y. Fan, X. Han and Y. Zheng, J. Power Sources, 2026, 666, 239147 CrossRef CAS.
- J. Hong, J. Yoon, J.-W. Park, Y.-C. Ha, J. Lee and I. Hwang, J. Power Sources, 2025, 655, 237925 CrossRef CAS.
- B. G. Meyer, G. Matthews, R. Scales, N. C. Mitchell, E. Darnbrough, R. A. House, D. E. J. Armstrong and P. S. Grant, ACS Mater. Lett., 2025, 7, 3444–3451 CrossRef.
- Y.-J. Lee, S.-Y. Kim, W.-J. Song, J. Cha and D.-W. Kim, Mater. Today Energy, 2025, 51, 101914 CrossRef CAS.
- D. Lee and A. Manthiram, Small Methods, 2023, 7, 2201680 CrossRef CAS.
- D. J. Lee, J. Jang, J.-P. Lee, J. Wu, Y.-T. Chen, J. Holoubek, K. Yu, S.-Y. Ham, Y. Jeon, T.-H. Kim, J. B. Lee, M.-S. Song, Y. S. Meng and Z. Chen, Adv. Funct. Mater., 2023, 33, 2301341 CrossRef CAS.
- K.-H. Lee, H. Shim, S. H. Lee, H.-J. Kim, C. Park, J. Choi, S.-J. Lee, Y.-K. Hong, J. Lyu, J. C. Kim, S. Park, H. Cha, W. Jin, J. Kim, S. Choi, S.-Y. Lee, S.-K. Jung, M. De Volder, T.-H. Kim and G. Song, Energy Environ. Sci., 2025, 18, 8446–8461 RSC.
- S. Jang, M.-H. Ryou, J.-Y. Bae, S.-H. Yu, S.-Y. Lee and J. Bang, Prog. Polym. Sci., 2025, 166, 101987 CrossRef CAS.
- K.-E. Sung, I. Hwang, J. Choi, S.-K. Jung and J. Yoon, Chem. Eng. J., 2025, 511, 161789 CrossRef CAS.
- N.-Y. Kim, J.-H. Kim, H. Koo, J. Oh, J.-H. Pang, K.-D. Kang, S.-S. Chae, J. Lim, K. W. Nam and S.-Y. Lee, ACS Energy Lett., 2024, 9, 5688–5703 CrossRef CAS.
- A. Komlev, D. Kudryavtseva, I. Neustroev, Y. Sudalenko, A. Altynnikov, A. Tsymbalyuk, A. Gagarin and R. Platonov, Coatings, 2025, 15, 644 CrossRef CAS.
- Y. Li, Y. Zhou, Y. Gu, B. Chen, B. Wang, J. Yan, J. Liu, F. Chen, D. Zhao and X. Liu, Vacuum, 2022, 196, 110763 CrossRef CAS.
- M. Nishino, Y. Okazaki, Y. Seto, T. Uehara, K. Endo, K. Yamamura and Y. Ohkubo, Polymers, 2022, 14, 394 CrossRef CAS PubMed.
- M. W. Finnis, J. Phys.: Condens. Matter, 1996, 8, 5811 CrossRef CAS.
- Y. Ye, R. Xu, W. Huang, H. Ai, W. Zhang, J. O. Affeld, A. Cui, F. Liu, X. Gao, Z. Chen, T. Li, X. Xiao, Z. Zhang, Y. Peng, R. A. Vila, Y. Wu, S. T. Oyakhire, H. Kuwajima, Y. Suzuki, R. Matsumoto, Y. Masuda, T. Yuuki, Y. Nakayama and Y. Cui, Nat. Energy, 2024, 9, 643–653 CrossRef CAS.
- H. Li, L. Wang, Y. Song, Z. Zhang, H. Zhang, A. Du and X. He, Adv. Funct. Mater., 2023, 33, 2305515 CrossRef CAS.
- Y. Ye, L.-Y. Chou, Y. Liu, H. Wang, H. K. Lee, W. Huang, J. Wan, K. Liu, G. Zhou, Y. Yang, A. Yang, X. Xiao, X. Gao, D. T. Boyle, H. Chen, W. Zhang, S. C. Kim and Y. Cui, Nat. Energy, 2020, 5, 786–793 CrossRef CAS.
- N. Li, J. Zhao, Y. Zhang, R. Song, N. Zhang, Y. Cui, J. Lin, H. Xu and Y. Huang, Adv. Funct. Mater., 2025, 35, 2419102 CrossRef CAS.
- F. Wang, S. Tang, Q. Han, S. Ji, J. Wang, B. Du, L. Xu, M. Guan, P. Lou, W. Zhang, Y.-C. Cao and S. Cheng, J. Colloid Interface Sci., 2025, 678, 57–65 CrossRef CAS PubMed.
- S. Y. Kim, T. K. Liu, H. S. Kim, D. H. Wang and J. H. Park, Adv. Funct. Mater., 2025, e18597 Search PubMed.
- Z. Hu, R. Zhao, J. Yang, C. Wu and Y. Bai, Energy Storage Mater., 2023, 59, 102776 CrossRef.
- T. Qin, H. Yang, Q. Li, X. Yu and H. Li, Ind. Chem. Mater., 2024, 2, 191–225 RSC.
- J. Yoon, J. Lee, H. Kim, J. Kim and H.-J. Jin, Polymers, 2024, 16, 254 CrossRef CAS PubMed.
- Y. Ohkubo, K. Ishihara, H. Sato, M. Shibahara, A. Nagatani, K. Honda, K. Endo and Y. Yamamura, RSC Adv., 2017, 7, 6432–6438 RSC.
- M. Zhianmanesh, A. Gilmour, M. M. M. Bilek and B. Akhavan, Appl. Phys. Rev., 2023, 10, 021301 CAS.
- J.-T. Kim, S. Kennedy, I. Phiri and S.-Y. Ryou, ACS Appl. Mater. Interfaces, 2024, 16, 11400–11407 CrossRef CAS PubMed.
- M. Kanno and I. Honma, J. Energy Storage, 2025, 137, 118658 CrossRef CAS.
- Y. Song, K. Gao, C. He, Y. Wu, S. Yang, N. Li, L. Yang, Y. Mao, W.-L. Song and H. Chen, Appl. Energy, 2023, 351, 121904 CrossRef CAS.
- J. Zhang, H. Huang and J. Sun, Powder Technol., 2022, 409, 117828 CrossRef CAS.
- B. Schumm, A. Dupuy, M. Lux, C. Girsule, S. Dörfler, F. Schmidt, M. Fiedler, M. Rosner, F. Hippauf and S. Kaskel, Adv. Energy Mater., 2025, 15, 2406011 CrossRef CAS.
- A. Gyulai, W. Bauer and H. Ehrenberg, ACS Appl. Energy Mater., 2023, 6, 5122–5134 CrossRef CAS.
- G. A. B. Matthews, S. Wheeler, J. Ramírez-González and P. S. Grant, Front. Energy Res., 2024, 11, 1336344 CrossRef.
- C. Wang, M. M. Thackeray and E. O. Fuller, Integrated battery electrode fabrication using dry powder coating method, US Pat., US20150303481A1, 2015 Search PubMed.
- K. Raju, L. Wheatcroft, M. C. Lai, A. Mahadevegowda, L. F. J. Piper, C. Ducati, B. J. Inkson and M. De Volder, J. Electrochem. Soc., 2024, 171, 080519 CrossRef CAS.
- S. Hong, J. K. Seo, C. Ha, S.-M. Oh and Y.-J. Kim, J. Power Sources, 2025, 638, 236621 CrossRef CAS.
- J. H. Kang, W. Jeong, M. S. Kang, H. W. Kim, D. W. Jeon, H. U. Lee, J. H. Hong, S. Han, M. Kim, S. Yang, D. Lee, P. J. Kim, T. Song, M. Yoon, L. Su, J. Choi and S. B. Cho, Commun. Mater., 2025, 7, 34 CrossRef.
- W. Chung, H. Lee, J. Shim and J. H. Bang, J. Electrochem. Sci. Technol., 2025, 16, 512–520 CrossRef CAS.
- H.-s Kim, J. Y. Jung, K. Kim, C. Hwang, J. Yu, M.-S. Park and W. Cho, Adv. Energy Mater., 2024, 14, 2303965 CrossRef CAS.
- Y. Lyu, S. Dong, T. Zhang and Z.-S. Guo, Eng. Fract. Mech., 2025, 321, 111120 CrossRef.
- J. Xu, B. Paredes-Goyes, Z. Su, M. Scheel, T. Weitkamp, A. Demortière and A. A. Franco, Batter. Supercaps, 2023, 6, e202300371 CrossRef.
- G. Matthews, B. Meyer, C. Doerrer, J. Ramírez-González, E. Darnbrough, N. Hallemans, D. Armstrong and P. S. Grant, J. Mater. Chem. A, 2025, 13, 18283–18291 RSC.
- T. J. Embleton, J. H. Choi, S.-J. Won, J. Ali, K. S. Saqib, K. Ko, M. Jo, J. Hwang, J. Park, J. H. Lee, J. Kim, M. K. Kim, J.-W. Jung, M. Park and P. Oh, Energy Storage Mater., 2024, 71, 103542 CrossRef.
- R. Tao, B. Steinhoff, X.-G. Sun, K. Sardo, B. Skelly, H. M. Meyer, C. Sawicki, G. Polizos, X. Lyu, Z. Du, J. Yang, K. Hong and J. Li, Chem. Eng. J., 2023, 471, 144300 CrossRef CAS.
- K. Zhang, D. Li, X. Wang, J. Gao, H. Shen, H. Zhang, C. Rong and Z. Chen, Materials, 2024, 17, 2349 CrossRef CAS PubMed.
- D. Kong, Q. Yang, Y. He and H. Hu, ACS Appl. Energy Mater., 2025, 8, 15438–15447 CrossRef CAS.
- S. Han, E.-H. Noh, S. Chae, K. Kwon, J. Lee, J.-S. Woo, S. Park, J. W. Lee, P. J. Kim, T. Song, W.-J. Kwak and J. Choi, J. Energy Storage, 2024, 96, 112693 CrossRef CAS.
- S. Han, W. Jeong, R. Kang, M. Kim, M. Jeong, H.-W. Lee, Y.-J. An, H.-J. Ji, M. Yoon, P. J. Kim, S. Sun, T. Song, D. Lee, W.-J. Kwak and J. Choi, EES Batteries, 2025, 1, 1267–1278 RSC.
- Z. Wei, D. Kong, L. Quan, J. He, J. Liu, Z. Tang, S. Chen, Q. Cai, R. Zhang, H. Liu, K. Xu, L. Xing and W. Li, Joule, 2024, 8, 1350–1363 CrossRef CAS.
- T. Lee, J. An, W. J. Chung, H. Kim, Y. Cho, H. Song, H. Lee, J. H. Kang and J. W. Choi, ACS Appl. Mater. Interfaces, 2024, 16, 8930–8938 CrossRef CAS PubMed.
- Y. Zhao, Z. Liang, Y. Kang, Y. Zhou, Y. Li, X. He, L. Wang, W. Mai, X. Wang, G. Zhou, J. Wang, J. Li, N. Tavajohi and B. Li, Energy Storage Mater., 2021, 35, 353–377 CrossRef.
- J. Kim, J. S. Park, J. J. Woo, S. Choi, J. Ma, J. Kim and S. Choi, ACS Energy Lett., 2025, 10, 2670–2678 CrossRef CAS.
- B. Chang, D. H. Yun, I. Hwang, J. K. Seo, J. Kang, G. Noh, S. Choi and J. W. Choi, Adv. Mater., 2023, 35, 2303787 CrossRef CAS PubMed.
- J. H. Kang, W. Jeong, M. S. Kang, H. W. Kim, D. W. Jeon, H. U. Lee, J. H. Hong, S. Han, M. Kim, S. Yang, D. Lee, P. J. Kim, T. Song, M. Yoon, L. Su, J. Choi and S. B. Cho, Commun. Mater., 2025, 7, 34 CrossRef.
- J.-K. Hu, H. Yuan, S.-J. Yang, Y. Lu, S. Sun, J. Liu, Y.-L. Liao, S. Li, C.-Z. Zhao and J.-Q. Huang, J. Energy Chem., 2022, 71, 612–618 CrossRef CAS.
- W. Shin, D. S. Kwon, M. Kim, J. Woo, H. G. Jung, J. Bang, J. Shim and J. K. Yoo, ChemSusChem, 2025, e202501943 Search PubMed.
- L. Meng, B. Song, Y. Lu, K. Lv, W. Gao, Y. Wang and G. Jiang, J. Environ. Sci., 2021, 107, 77–86 CrossRef CAS PubMed.
- E. K. Savvidou, A. Rensmo, J. P. Benskin, S. Schellenberger, X. Hu, M. Weil and I. T. Cousins, Environ. Sci. Technol., 2024, 58, 21908–21917 CrossRef CAS PubMed.
- A. A. Dolatabad, X. Zhang, J. Mai, A. Kubátová, J. Cao and F. Xiao, J. Hazard. Mater., 2025, 496, 139322 CrossRef CAS PubMed.
- J. Kang, H. Eom, D. Yoo, M.-L. Seol, J. W. Han, H. Song, J. Li and I. Nam, Energy Storage Mater., 2025, 82, 104662 CrossRef.
- R. He, W. Zhong, C. Cai, S. Li, S. Cheng and J. Xie, Adv. Energy Mater., 2024, 14, 2402109 CrossRef CAS.
- J. Lee, C. Y. Son, S. Han, S. Yang, P. J. Kim, D. Lee, J. W. Lee, W.-H. Ryu and J. Choi, Chem. Eng. J., 2025, 503, 158271 CrossRef CAS.
- C. Nguyen Thi Linh, V. Dong Thuc, T. A. Nguyen, T. D. Cam Ha and D. Kim, Mater. Today Energy, 2025, 54, 102071 CrossRef CAS.
- M.-J. Kim, J.-H. Cha and E.-S. Oh, J. Korean Battery Soc., 2024, 4, 67–73 CrossRef.
- J. Kang, H. Eom, S. Jang, D. Yoo, H. Lee, M. Kim, M.-L. Seol, J. W. Han, I. Nam and H. Song, Adv. Mater., 2025, 37, 2416872 CrossRef CAS PubMed.
- M. K. Kim, T. Yu, S. Jang, J. Lee, H. Oh, M. Jang, H. Cha, H. Lee, J. Kang, S. M. Lee, H. Shim, K.-H. Lee, G. Song, W. Jin, T.-H. Kim, S. Choi, K.-M. Jeong, J. T. Han, J.-K. Yoo, H.-G. Jung, S. Song, M. Park, J. Seong, D. Kim, H. Choi, M. Seong, M. J. Lim, W. R. Hwang, J. Nam, S. Jo and J. Kim, Nat. Commun., 2025, 16, 11174 CrossRef CAS PubMed.
- H.-M. Kim, B.-I. Yoo, J.-W. Yi, M.-J. Choi and J.-K. Yoo, Nanomaterials, 2022, 12, 3320 CrossRef CAS PubMed.
- F. Schmidt, S. Kirchhoff, K. Jägle, A. De, S. Ehrling, P. Härtel, S. Dörfler, T. Abendroth, B. Schumm, H. Althues and S. Kaskel, ChemSusChem, 2022, 15, e202201320 CrossRef CAS PubMed.
- Y. Zhang, S. Lu, F. Lou and Z. Yu, Energy Technol., 2022, 10, 2200732 CrossRef CAS.
- H. M. Kim, B. I. Yoo, J. W. Yi, M. J. Choi and J. K. Yoo, Nanomaterials, 2022, 12, 3320 CrossRef CAS PubMed.
- J. U. Won, B. K. Park, J. Choi and K. J. Kim, Adv. Energy Mater., 2025, 15, e04841 CrossRef CAS.
- Y. H. Liang, H. Long, Z. M. Huang, T. He, X. M. Chen, M. Li, H. Chen and S. Q. Zhang, Adv. Funct. Mater., 2025, e18619 Search PubMed.
- Y. Li, Y. Wu, Z. Wang, J. Xu, T. Ma, L. Chen, H. Li and F. Wu, Mater. Today, 2022, 55, 92–109 CrossRef CAS.
- C. Y. Lim, G. Park and K. J. Lee, Carbon Lett., 2025, 35, 675–685 CrossRef CAS.
- Z. Ye, Z. Zou and C. Jiang, Diamond Relat. Mater., 2023, 136, 109911 CrossRef CAS.
- B. Kim, D. K. Kim, J. Yu and Y. Yoo, Energy Environ. Mater., 2025, 8, e70019 CrossRef CAS.
- R. Shvartzman-Cohen, Y. Levi-Kalisman, E. Nativ-Roth and R. Yerushalmi-Rozen, Langmuir, 2004, 20, 6085–6088 CrossRef CAS PubMed.
- S. Mallakpour and S. Soltanian, RSC Adv., 2016, 6, 109916–109935 RSC.
- M. Maugey, W. Neri, C. Zakri, A. Derre, A. Penicaud, L. Noe, M. Chorro, P. Launois, M. Monthioux and P. Poulin, J. Nanosci. Nanotechnol., 2007, 7, 2633–2639 CrossRef CAS PubMed.
- H. Lee, J. Park, H. Cho, J. Lee and K.-H. Lee, RSC Adv., 2021, 11, 38152–38160 RSC.
- M. Hattori, T. Kiyono, Z. Zhao, M. Higashi, M. Fujishiro, Y. Kishikawa, J. Escorihuela and N. Shibata, Nat. Commun., 2025, 17, 669 CrossRef PubMed.
- W. Yu, L. Chen, X. Zhang, R. Lu, X. Zhu and F. Lu, Resour. Conserv. Recy., 2025, 215, 108143 CrossRef CAS.
- S. Liu, R. J. Letcher, M. L. Diamond, A. Blum and D. Chen, Nat. Commun., 2025, 17, 108 CrossRef PubMed.
- F. Xiao, B. Deng, D. Dionysiou, T. Karanfil, K. O’Shea, P. Roccaro, Z. J. Xiong and D. Zhao, Nat. Water., 2023, 1, 1004–1015 CrossRef.
- L. Qiu, Z. Shao, D. Wang, F. Wang, W. Wang and J. Wang, Carbohydr. Polym., 2014, 112, 532–538 CrossRef CAS PubMed.
- P. Parikh, M. Sina, A. Banerjee, X. Wang, M. S. D’Souza, J.-M. Doux, E. A. Wu, O. Y. Trieu, Y. Gong, Q. Zhou, K. Snyder and Y. S. Meng, Chem. Mater., 2019, 31, 2535–2544 CrossRef CAS.
- I. İ. Avcı Yayla, N. Yuca, E. Sezer and B. Ustamehmetoğlu, Energy Storage, 2022, 4, e338 CrossRef.
- S. Choi, T.-w Kwon, A. Coskun and J. W. Choi, Science, 2017, 357, 279–283 CrossRef CAS PubMed.
- Y.-K. Hong, J.-H. Kim, N.-Y. Kim, K.-S. Oh, H.-I. Kim, S. Ryu, Y. Ko, J.-Y. Kim, K.-H. Lee and S.-Y. Lee, Nano-Micro Lett., 2025, 17, 112 CrossRef CAS PubMed.
- Y. Guo, R. Soni, K. Coke, J. B. Robinson, F. Iacoviello, R. S. Young, R. Jervis, P. R. Shearing and T. S. Miller, ACS Sustain. Chem. Eng., 2025, 13, 13726–13739 CrossRef CAS PubMed.
- P. Dong, X. Zhang, J. Zamora, J. McCloy and M.-K. Song, J. Energy Chem., 2023, 80, 442–451 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |