Next-generation borophene nanostructures for dual-domain sensing: from structural optimisation to biomedical and environmental applications
Chayanika
Hazarika
ab,
Pulakesh
Borah
 ab,
Rituparna
Duarah
ab,
Rituparna
Duarah
 *ab and
Manash R.
Das
*ab and
Manash R.
Das
 *ab
*ab
aMaterials Sciences Group, Coal Energy and Materials Sciences Division, CSIR-North East Institute of Science and Technology, Jorhat 785006, Assam, India. E-mail: rituparnaduarah@gmail.com; mnshrdas@yahoo.com
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad 201002, India
First published on 22nd October 2025
Abstract
Borophene, a novel two-dimensional (2D) material composed of atomically thin boron sheets, has emerged as a formidable contender to graphene due to its unique structural and electronic attributes. Borophene-derived nanosheets, quantum dots and nanocomposites are revolutionising advanced sensing technologies by integrating high-performance electronic properties with biologically and environmentally relevant detection capabilities. Their exceptional charge carrier mobility, tunable bandgap, and intrinsic catalytic activity endow them with remarkable plasmonic, optical, and enzyme-mimetic properties, enabling ultra-sensitive and selective detection of biomolecules and pollutants. This review critically examines the synthesis, structural evolution, and functional tuning of borophene-derived nanostructures (nanosheets, quantum dots and nanocomposites), with a focus on surface engineering strategies, such as doping, functionalisation, and hybridisation, that enhance sensing performance. The mechanisms underlying fluorescence, colorimetric, and electrochemical sensing modalities are explored, along with the emerging role of borophene-based nanozymes in catalytic biosensing. A comparative evaluation of borophene sheets, quantum dots and nanocomposites is presented, highlighting their advantages over conventional 2D materials. Finally, current challenges and future perspectives are discussed, marking the transformative potential of borophene-based nanostructures in next-generation multifunctional sensing technologies.
Wider impactThe precise design of atomic-scale architectures with tunable electronic and interfacial properties is pivotal for advancing miniaturised, high-precision analytical systems in biomedical and environmental sensing. Borophene, a structurally polymorphic and electronically versatile 2D boron allotrope, offers a highly responsive platform owing to its anisotropic charge transport, multi-centre bonding, and surface adaptability. This review highlights borophene-based quantum dots, nanosheets, and nanocomposites as multifunctional transducers in fluorescence, colorimetric, and electrochemical sensing platforms, capable of detecting a broad spectrum of analytes including hydrogen peroxide (H2O2), glucose, dopamine, cholesterol, etc. in biomedical diagnostics, as well as heavy metals, nitroaromatics, and volatile organic compounds (VOCs) in environmental monitoring. By systematically correlating synthetic methodologies and surface functionalisation strategies with catalytic, optical, and electronic transduction mechanisms, this work lays the groundwork for the development of ultra-sensitive, multifunctional sensors, poised to revolutionise precision diagnostics, real-time environmental monitoring, and wearable biointerfaces, thereby contributing meaningfully to global health security and sustainable technology solutions. |
1. Introduction
Xenes, a class of two-dimensional (2D) monoelemental materials, encompass a broad range of elements, including boron and carbon-based structures such as graphitic carbon nitride1 as well as germanene,2 phosphorene,3 hexagonal boron nitride,4 stanene,5 silicene,6 and plumbene.7 These materials have gained significant attention due to their exceptional mechanical strengths, high surface-to-volume ratios, and outstanding electrical conductivity. Moreover, they have propelled notable progress in nanotechnology, opening new avenues for advanced applications in sensor development, energy storage, and conversion technologies. Since then, the field has witnessed rapid expansion, with each newly discovered Xene offering distinct properties that enable a wide spectrum of applications, from optoelectronics to biomedicine.Among these emerging Xenes, borophene, a 2D allotrope of boron, has emerged as a promising material.8 It is an electrically versatile and structurally dynamic member of the Xene family which is redefining the frontier of 2D materials beyond graphene. Often regarded as a ‘cousin of graphene’, borophene has advantages over graphene, due to its strongly anisotropic structure, and the fact that its electronic and magnetic properties can be orientation-controlled for flexible applications.9 It distinguishes itself through its unique structural features, arising from the complex and versatile bonding behavior of boron atoms10 which provide borophene with remarkable mechanical and electrical qualities. High electrical conductivity gives it its inherent metallic nature, and the lattice's strong B–B bonds provide it with exceptional mechanical strength and critical strain properties.11 Apart from conventional covalent bonding, boron uniquely exhibits three-center, two-electron (3c–2e) bonding,12,13 which facilitates the formation of a wide variety of atomic structures with diverse sizes and bonding configurations.14 Experimentally, several borophene structures such as β12, χ3, and graphene-like phases have been synthesized. To date, four distinct phases of borophene 2-Pmmn, β12, χ3, and graphene-type, under ultrahigh vacuum (UHV) conditions on atomically clean Ag(111) or Al(111) substrates have been reported, each exhibiting unique structural and electronic characteristics.15 Notably, the 2-Pmmn phase represents a buckled configuration in which boron atoms in adjacent rows are corrugated in a zigzag pattern, while remaining uncorrugated along the perpendicular in-plane direction. Remarkably, this structure exhibits negative Poisson's ratios in both in-plane directions.16 Interestingly, borophene synthesized from bulk boron exhibits enhanced symmetry and structural stability, serving as a structural basis for the evolution of various borophene phases, including the 2-Pmmn phase. Bulk boron predominantly consists of B7 clusters arranged in a hexagonal lattice, with an additional boron atom occupying the center of each hollow hexagon, contributing to borophene's unique geometry, multi-center bonding characteristics, and the emergence of structurally diverse polymorphs.17,18 This is attributed to the fact that boron allotropes such as B7, B13, and B36 naturally adopt planar or pseudo-planar configurations in bulk, driven by their high symmetrical stability. These structural patterns provide a favorable basis for the formation and stabilization of two-dimensional boron-based nanomaterials.19 For instance, Piazza et al. reported the planar B36-structure enclosing hexagonal holes in 2014.20 The study provided indirect experimental validation for the existence of 2D boron structures with defined hexagonal hole densities, thereby introducing the term “borophene” in analogy to graphene. Concurrently, another investigation demonstrated that the B35 cluster, featuring double hexagonal holes and high structural flexibility, could serve as a stable and robust foundation for the realization of borophene.18,20 To date, only a limited number, approximately 16, of such boron-based 2D structures have been theoretically proposed, experimentally synthesised, and thermodynamically validated through structural modulation and computational simulations.21
Since the successful synthesis of 2D boron sheets on Ag(111) substrates, interest in 2D boron materials has significantly increased.22 Owing to its structural similarities with graphene and MXenes, borophene can be fabricated using comparable techniques, broadly classified into top-down and bottom-up approaches. Among these, chemical vapor deposition (CVD) is a widely adopted top-down strategy which relies on the pyrolysis of a boron precursor under high temperature and vacuum, where the volatile compound decomposes to generate metallic vapour that subsequently deposits onto the substrate. Using diborane as the boron source, researchers successfully achieved the growth of monoatomic borophene sheets through CVD.23 A wide range of strategies have been explored for the fabrication of borophene, including CVD, liquid-phase exfoliation (LPE), sonochemical exfoliation, mechanical exfoliation, molecular beam epitaxy (MBE), thermal decomposition, and epitaxial growth.24 Recent innovations such as sonochemical exfoliation and a modified Hummers’ method have enabled the production of free-standing borophene, addressing scalability limitations and paving the way for its integration into renewable energy devices and ultrafast sensors.25,26 However, fabricating ultrathin borophene from bulk remains challenging due to the limitations inherent in top-down approaches.27 Borophene has garnered significant attention across various disciplines including materials science, nanotechnology, physics, chemistry, and condensed matter, due to its exceptional properties and versatile potential.27–29 In particular, boron-based nanostructures exhibit outstanding physical and chemical characteristics, making them highly suitable for a wide spectrum of scientific and technological applications, such as field emission (FE) devices, high-energy fuels, high-temperature components, protective coatings, and nuclear engineering.30,31 Furthermore, borophene exhibits high bending flexibility and remarkable mechanical strength along multiple directions, arising from its unique boron–boron multicenter bonding, as revealed by theoretical studies.19,20 These characteristics suggest that borophene is a promising candidate for advanced wearable devices and electronic skin (e-skin) applications.32,33 These advancements align with borophene's growing prominence in analytical sensing, where its derivatives, particularly borophene, exhibit intrinsic enzyme-mimicking (nanozyme) activities, such as peroxidase (POD)-like behavior, facilitating the catalytic oxidation of chromogenic substrates for rapid and visual colorimetric detection.34 Simultaneously, the tunable fluorescence, high photostability, and strong affinity for biomolecules make borophene quantum dots (BQDs) ideal for fluorometric sensing,10 while borophene's exceptional electron mobility and surface reactivity enhance its performance in electrochemical platforms. Together, these multifunctional capabilities underline the versatility of borophene-based materials for integrated health diagnostics, environmental monitoring, and smart wearable technologies, which is reflected by the steadily increasing number of publications on “borophene” over the years (Fig. 1), highlighting its rising prominence in materials research.
 | ||
| Fig. 1 Number of scientific publications on ‘borophene’ over the last ten years (data procured from Web of Science on 27/09/2025). | ||
Accordingly, the total citations generated by borophene-based studies have increased exponentially. Although a few review articles on borophene have primarily focused on its synthesis strategies and broad technological applications, highlighting its superior electronic, optical, thermal, and elastic properties compared to graphene and other related materials there remains a lack of comprehensive reviews dedicated to its application in the sensitive and selective detection of biomolecules and environmental pollutants.
Addressing this gap, the present review aims to provide an in-depth overview of the latest advancements in borophene-based nanostructures for fluorometric, colorimetric, and electrochemical sensing strategies in health and environmental monitoring (Scheme 1). Several existing review articles35–37 lack a comprehensive discussion on specific sensing mechanisms such as fluorometric, electrochemical, and nanozyme-based approaches. Moreover, they do not provide detailed examples of borophene-based sensors in key application areas like health diagnostics and environmental monitoring. Additionally, there is an insufficient focus on the diverse nanostructures of borophene and their role in enhancing sensing performance. These gaps highlight the need for a more focused and application-oriented review of borophene nanomaterials in sensor development.
So, in this review article, we aim to address the gap by critically examining the synthesis, structural evolution, and functional tuning of borophene-derived materials, including quantum dots, nanosheets, and nanocomposites with a particular emphasis on surface engineering strategies that enhance their sensing performance and highlight the unique physicochemical properties of borophene that enable high sensitivity and selectivity. This review explores the emerging role of borophene-based nanozymes in catalytic biosensing, drawing attention to their POD-like activities and potential in real-time monitoring applications. A comparative evaluation of different borophene nanostructures is presented, highlighting their advantages over conventional 2D materials. Specifically, this work not only consolidates existing knowledge but also identifies key challenges and opportunities for future research, outlining promising directions for achieving enhanced sensitivity, selectivity, stability, and real-world applicability in areas such as environmental monitoring and biomedicine, diagnostics, etc. Finally, the review concludes with critical insights into the current challenges and future prospects of borophene research, emphasizing its growing potential as a next-generation sensing material. This analysis is crucial for accelerating the translation of borophene-based sensors from theoretical concepts to practical devices, paving the way for significant advancements in material science and sensor technology.
2. Synthesis and functional evolution of borophene-based nanomaterials
Borophene, a monoatomic layer of boron featuring trigonal and hexagonal ring arrangements, exhibitsexceptional material properties that have garnered significant research interest in recent years.8 Beyond its 2D form, researchers have explored borophene in various dimensions 0D, 1D, and even 3D due to its versatile structural adaptability.38–43 The strategic design and dimensional engineering of borophene-based nanomaterials are crucial for tailoring their physicochemical properties, enabling applications in catalysis, sensing, energy storage, and biomedicine. Synthesis methods significantly affect borophene's morphology, stability, and functionalization potential. Due to their structural similarities, graphene and borophene can be synthesized using similar top-down and bottom-up approaches.11 The majority of standard techniques for exfoliating graphene from bulk graphite are not appropriate for synthesizing boron sheets because bulk boron does not have a layered structure like graphite possesses. As a result, improved techniques are needed for the synthesis of borophene, mainly LPE,44 MBE,45 and CVD.15,23 Borophene's unique atomic structure and exceptional surface anchoring capabilities make it a highly promising material for sensing applications, including light, gas, molecular, and mechanical detection. However, controlled synthesis remains essential to overcome its structural limitations and enhance stability and dispersibility for practical use.2.1 Precision engineering of borophene sheets, borophene quantum dots, and borophene nanocomposites
However, it restricts large-scale applications and inhibits research advancement due to its reliance on substrates and extremely high vacuum conditions for deposition. To address these limitations, Ranjan et al. have developed a novel, facile, and scalable liquid-phase synthesis (Fig. 2a) of freestanding borophene nanosheets via sonochemical exfoliation and reduction strategy using borophene oxide, enabling the production of freestanding borophene atomic nanosheets under more accessible conditions.47 Acetone as a solvent proved highly effective, yielding borophene monolayers as shown via transmission electron microscope (TEM) (Fig. 2b) and high resolution transmission electron microscope (HRTEM) (Fig. 2c) analysis, with crystallinity evident in the inset. Atomic force microscopy (AFM) confirmed the presence of few-layer borophene nanosheets with variable thickness and large lateral dimensions. The Raman analysis indicated an anisotropic nature and structural defects. X-ray photoelectron spectroscopy (XPS) revealed characteristic B 1s peaks for borophene and signals corresponding to surface oxidation (Fig. 2d–h). In a similar way, Yadav et al. synthesized 2D borophene via surfactant-assisted LPE using sonication, centrifugation, and washing, offering a scalable and eco-friendly route for producing high-quality nanosheets. The nanosheets showed dual-phase crystallinity (β12, χ3), good dispersion stability, high surface areas, and a 3.53 eV band gap, indicating potential for electrochemical sensing.48
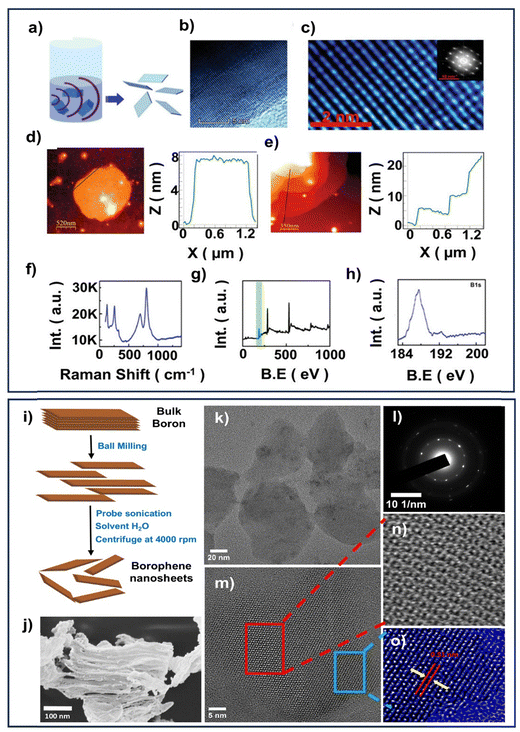 | ||
| Fig. 2 (a) Schematic diagram illustrating the sonochemical exfoliation synthesis of borophene, (b) TEM image, (c) HRTEM image of borophene synthesized in acetone solvent (inset shows the electron diffraction pattern), (d) and (e) AFM images, (f) Raman spectrum, (g) XPS survey spectrum of borophene, and (h) high-resolution XPS B 1s scan. Reprinted with permission from ref. 47. Copyright 2019, Wiley. (i) Schematic representation of the synthesis procedure for borophene nanosheets, (j) SEM image, (k) and (m)–(o) HRTEM images of borophene nanosheet at various magnifications, and (l) SAED pattern of borophene nanosheets. Reprinted with permission from ref. 34. Copyright 2024, Royal Society of Chemistry. | ||
In another work, Kiraly et al. reported the synthesis of borophene under ultrahigh vacuum by thermally evaporating boron onto a clean Au(111) substrate. Unlike on Ag(111), where boron stays surface-bound, Au(111) allows boron to diffuse into the bulk during deposition and later segregate to the surface upon cooling, forming borophene islands. This process modifies the native surface structure of Au(111), creating a trigonal network that enables templated growth. With increased boron concentration, this order is lost, leading to larger sheet formation. Scanning tunneling microscopy (STM) showed embedded borophene islands with ordered patterns revealing the characteristic herringbone surface reconstruction of the substrate, which is defined by Shockley partial dislocations aligned along the (112) directions. These dislocations delineate alternating face-centered cubic (fcc) and hexagonal close-packed (hcp) striped regions, while XPS and secondary ion mass spectrometry (SIMS) revealed boron diffusion into subsurface layers and induced strain. Spectroscopic studies and theoretical calculations indicated the metallic nature of borophene, with smaller islands exhibiting confinement-related electronic features.49
Guoan Tai et al. synthesized borophene nanosheets on carbon cloth via CVD using sodium borohydride and hydrogen. Uniform growth at 700 °C for 30 min produced strongly adherent, structurally intact nanosheets. TEM, HRTEM, SAED, and FFT confirmed crystalline α′-2H-borophene, while XPS, Raman, and FTIR indicated characteristic B–B and B–H bonds.40 Chowdhury et al. synthesized few-layer borophene via temperature-assisted electrochemical exfoliation, using thermally modified boron as the cathode and platinum as the anode in ionic liquids or aqueous electrolytes. Raman spectroscopy confirmed characteristic vibrational modes, while zeta potential and particle size analysis indicated high colloidal stability and suitability for further applications.50 Mazaheri et al. reported the synthesis of 2D boron sheets using CVD. Diborane (B2H6) was thermally decomposed at 830 K in a low-pressure CVD chamber. This bottom-up method produced atomically thin borophene nanosheets with a dominant χ3 phase. The nanosheets were stabilized by electron transfer from the Al substrate. The process is compatible with conventional deposition systems. It enables scalable production of large-area crystalline borophene for nanoelectronics and sensing.23 Guo et al. also reported that borophene, with its graphene-like structure, high conductivity, and stability, is ideal for scalable synthesis via low-pressure CVD to obtain tetragonal single-crystalline sheets with excellent ambient stability.51
In addition, eco-friendly approaches have been developed to synthesise metal-free, biocompatible borophene nanosheets, further broadening their applicability in catalysis and biosensing. Borah et al. recently synthesized freestanding borophene nanosheets via an eco-friendly LPE method. Ball-milled boron powder was dispersed in deionized water and exfoliated using low-power ultrasonication, avoiding hazardous solvents and reducing energy use. The process yielded stable, biocompatible nanosheets with good dispersibility due to their high surface charge (Fig. 2i). SEM images of the synthesized borophene nanosheet material, presented in Fig. 2j, further revealed its sheet-like morphology. Characterization by TEM and HRTEM confirmed the formation of thin, transparent sheets exhibiting dual-phase crystalline features, with an interplanar spacing of approximately 0.51 nm, as shown in the Fig. 2(k)–(o). SAED patterns indicated a polycrystalline nature, with lattice fringes corresponding to known boron phases, including β-rhombohedral and mixed α-sheet structures, reflecting the structural complexity and potential functionality of the borophene nanosheet.34
Moreover, numerous innovative methods have been investigated for the synthesis of borophene nanosheets, each specifically designed to improve its structural, electrical, and functional characteristics for a variety of uses. By optimizing borophene's shape, stability, and performance for a given application, each method opens the door for its incorporation into catalytic processes, energy storage, electromagnetic shielding, and biological sensing.
Gogoi et al. synthesized BQDs via a hydrothermal-assisted LPE method using deionized water as the sole solvent, offering an environmentally friendly approach. The process involved hydrothermal treatment followed by ultrasonication and centrifugation to yield colloidally stable, fluorescent BQDs with excellent aqueous stability (Fig. 3a). TEM analysis showed uniformly distributed, spherical nanoparticles with slight aggregation. HRTEM revealed distinct lattice fringes corresponding to β-rhombohedral boron, while SAED patterns confirmed their polycrystalline nature. These BQDs exhibited size-dependent optical and catalytic properties, making them suitable for biosensing and nanozyme applications.10 In the same manner, Wang et al. reported crystalline BQDs, synthesized via a low-temperature, probe-assisted LPE method using a ternary solution of boric acid, hydrogen peroxide, and isopropyl alcohol. In this process, boric acid promoted nucleation, while hydrogen peroxide enhanced exfoliation through a sonochemical mechanism driven by cavitation-induced shockwaves (Fig. 3b). This approach yielded a stable dispersion of BQDs with good monodispersity, dispersibility, and two-photon fluorescence properties. TEM analysis showed well-dispersed, spherical nanoparticles with an average lateral size of ∼3.5 nm. HRTEM revealed lattice fringes with ∼0.22 nm spacing, corresponding to the (404) plane of β-rhombohedral boron, and FFT analysis indicated the presence of β12 and χ3 borophene-like phases. AFM confirmed their ultrathin, few-layered nature with discrete height profiles, validating successful exfoliation and the quantum dot morphology.53 Powder XRD patterns of the as-collected BQDs disclose the crystal structure, which can be indexed to the β-rhombohedral boron (PDF #80-0323),54 indicating that the BQDs still have a crystalline boron structure and are derived from the bulk boron.
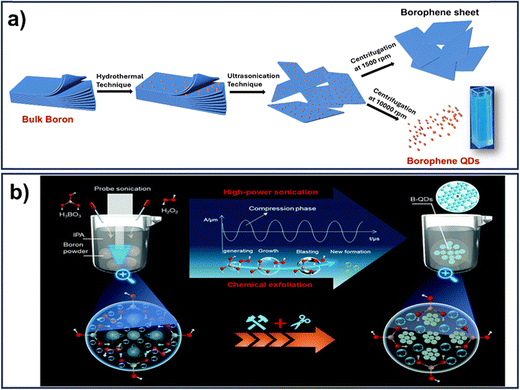 | ||
| Fig. 3 (a) Schematic representation of the synthesis procedure of borophene QDs via a hydrothermal-assisted sonication technique; reprinted with permission from ref. 10 Copyright 2024 American Chemical Society. (b) Schematic representation of the synthesis procedure of a liquid-phase exfoliation method in a low-temperature environment. Reprinted with permission from ref. 53 Copyright 2021, Royal Society of Chemistry. | ||
Joshi et al. developed a green, probe-assisted LPE method to synthesize water-dispersible BQDs using isopropyl alcohol, boric acid, and hydrogen peroxide. In situ oxidation and edge functionalization, followed by ultrasonication, produced stable BQDs with strong blue fluorescence. TEM showed uniformly dispersed, quasi-spherical nanoparticles, while HRTEM confirmed crystalline lattice spacing corresponding to the (021) plane of β-rhombohedral boron. Exposure to dehydroepiandrosterone (DHEA) induced aggregation and enhanced fluorescence via aggregation-induced emission, highlighting their potential for sensitive biosensing.55 Further advancing the synthesis of BQDs, Aswal et al. synthesized ultrasmall BQDs via a two-step top-down LPE approach using isopropyl alcohol. The process involved mechanical pulverization of boron chunks followed by prolonged tip sonication in an ice bath to minimize thermal degradation. After solvent removal and redispersion in water, stable and highly fluorescent BQDs were obtained. TEM analysis revealed uniformly spherical particles with size reduction upon extended sonication, reaching an average lateral size of ∼4.1 nm for the 10 h sample. HRTEM showed clear lattice fringes with ∼0.36 nm spacing, corresponding to the (204) plane of β-rhombohedral boron, indicating high crystallinity. AFM confirmed few-layer morphology with discrete height profiles, validating successful exfoliation and good dispersibility.56 Barman et al. synthesized Arg(BQDs) via a two-step approach involving mixed-solvent-assisted exfoliation of boron powder followed by arginine-mediated surface functionalization under hydrothermal conditions. The exfoliation yielded few-layer borophene nanosheets, which were then fragmented into QDs in the presence of Arg. The resulting B-Arg QDs exhibited strong blue fluorescence and good colloidal stability. The author mentioned that TEM analysis showed nearly spherical particles with an average lateral size of ∼3.3 nm, and HRTEM confirmed crystalline features with lattice spacing corresponding to β-rhombohedral boron. AFM of the precursor sheets indicated a few-layer morphology, supporting effective exfoliation. XPS analysis revealed B–B and B–O bonds, and the emergence of B–N and N–C peaks confirmed successful Arg functionalization, indicating covalent integration on the QDs surface.57 Yashwanth et al. reported that BQDs were synthesized via a sonochemical LPE method using isopropyl alcohol, with boric acid and hydrogen peroxide facilitating mild oxidation and surface functionalization. The process yielded fluorescent and water-stable BQDs. TEM analysis revealed spherical particles with an average size of ∼6 nm, and HRTEM showed lattice fringes corresponding to β-rhombohedral boron. In the BQDs/TiO2 nanocomposite, TEM confirmed homogeneous decoration of BQDs on TiO2 nanoparticles with clear interfacial contact, while HRTEM showed additional lattice planes of anatase TiO2. SAED patterns indicated polycrystallinity. XPS confirmed B–B, B–O, and B2O3 bonds, along with interfacial oxygen bridging and electronic interaction with TiO2, suggesting successful integration and potential for improved photocatalytic performance.58
Building on these advancements, Turkmen et al. synthesized a novel PEDOT:PSS/β12-borophene nanocomposite via a simple solution-based method at room temperature. The β12-borophene nanosheets were prepared through green, surfactant-free LPE in DMF under ambient conditions, with ultrasonication facilitating exfoliation via shear forces and cavitation. TEM and HRTEM analyses confirmed the formation of freestanding nanosheets with structural anisotropy and clear lattice fringes corresponding to β12-phase borophene. FFT patterns indicated high crystallinity, while XRD analysis validated phase purity and structural integrity, showing diffraction peaks consistent with β-rhombohedral boronphenedot.65
Again, smart fabrics capable of harvesting ambient energy and providing diverse sensing functionalities through triboelectric effects have garnered significant interest for next-generation healthcare electronics. As a potential triboelectric material, a unique borophene/ecoflex nanocomposite was developed by Chen et al., that provides flexibility, mechanical stability, durability, and tailorability as shown in Fig. 4a. Borophene nanosheets were synthesized using a modified LPE method, where bulk boron powder was dispersed in N-methyl-2-pyrrolidone and subjected to sequential ultrasonication and centrifugation. The exfoliated nanosheets were isolated, washed, and vacuum-dried, yielding highly crystalline and uniform borophene suitable for nanocomposite fabrication. HRTEM micrographs revealed ultrathin, quasi-transparent sheets with minimal folding and distinct lattice fringes showing an interplanar spacing of 0.35 nm, corresponding to the (1![[0 with combining macron]](https://www.rsc.org/images/entities/char_0030_0304.gif) 2) planes of β-rhombohedral boron. SAED patterns confirmed the polycrystalline nature of the sheets. AFM analysis showed uniform sheet morphology and layer thicknesses consistent with few-layer borophene. XPS confirmed dominant boron peaks with minimal oxidation, indicating the high purity and structural integrity of the nanosheet.66
2) planes of β-rhombohedral boron. SAED patterns confirmed the polycrystalline nature of the sheets. AFM analysis showed uniform sheet morphology and layer thicknesses consistent with few-layer borophene. XPS confirmed dominant boron peaks with minimal oxidation, indicating the high purity and structural integrity of the nanosheet.66
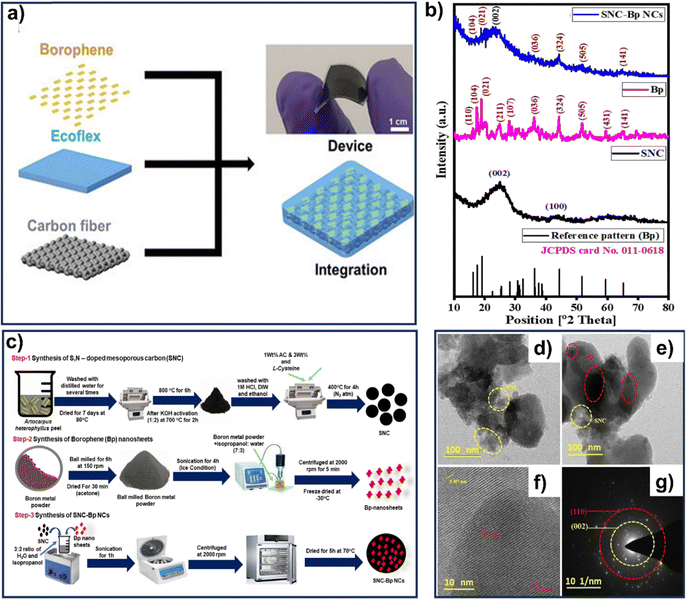 | ||
| Fig. 4 (a) A schematic integration process of producing a fabric-based triboelectric nanogenerator; reprinted with permission from ref. 66 Copyright 2022 Wiley. (b) XRD spectrum of borophene nanosheet with stimulated reference pattern, SNC, borophene nanosheet, and SNC-BNS NCs; (c) schematic representation of the steps involved in synthesizing SNC-BNS NCs; (d)–(f) HR-TEM images of SNC-BNS NCs; (g) SAED pattern of SNC-BNS. Reprinted with permission from ref. 67 Copyright 2023 Elsevier. | ||
Chinnalagu et al. reported the synthesis of 2D borophene nanosheets anchored onto S, N-doped mesoporous carbon (SNC-BNSs) to form a nanocomposite. Borophene nanosheets were synthesized via a LPE approach, involving pre-milling of boron powder followed by probe sonication in an isopropanol-water mixture. This surfactant-free, green method ensured minimal defects and good dispersibility. Post-sonication, the exfoliated nanosheets were isolated and dried to obtain stable borophene powder as depicted in Fig. 4c. HRTEM revealed thin, stacked nanosheets with sheet-like morphology and high crystallinity, as evidenced by distinct lattice fringes corresponding to β-rhombohedral planes (Fig. 4d–f). The SAED pattern illustrates the presence of both amorphous and polycrystalline SNC and Bp-nanosheets in the SNC-BNSs with circular rings of various diameters distinctly attributable to the planes of (002) and (110) of SNC and borophene nanosheet (Fig. 4g). XRD patterns matched the rhombohedral boron phase, confirming phase purity and long-range order. The SNC-BNS carry both the characteristic peaks of SNC and borophene nanosheet and the availability of (002) confirms the successful incorporation of SNC into the borophene nanosheet (Fig. 4b). The combination of HRTEM and XRD validated the effective exfoliation and structural integrity of the borophene sheets.67 Cihat Taşaltın reported the synthesis of PAN: β-rhombohedral borophene composite by sonicating borophene and PAN in N,N-dimethylformamide (DMF) via physical exfoliation. For composite preparation, borophene PAN were co-dispersed in DMF, ultrasonicated, and drop-cast onto a gold electrode to form a uniform film for electrochemical sensing applications. HRTEM analysis revealed well-defined nanosheets with hexagonal morphology and distinct lattice fringes, confirming the β-rhombohedral phase, further supported by FFT and XRD patterns. Zeta potential measurements indicated stable dispersion and surface charge modulation across pH values. The integration of borophene into the PAN matrix enhanced the electrochemical performance, attributed to improved electron transport and interfacial charge distribution.68 Mabhulusa et al. developed free-standing borophene sheets synthesized through prolonged sonication of boron powder in acetone, followed by centrifugation and drying. For nanocomposite fabrication, borophene was dispersed in water and combined with palladium and nickel oxide nanoflowers (NiONF) under ultrasonic agitation to yield a PdNiONF/B hybrid. HRTEM analysis showed borophene nanosheets with crystalline lamellar morphology and a lattice spacing of 0.89 nm, characteristic of the β12-borophene phase. The PdNiONF/B composite displayed multilayered flakes decorated with well-dispersed Pd and NiO nanoparticles. Distinct lattice fringes matched those of hexagonal borophene, face-centered cubic Pd, and cubic NiO. Edge views confirmed stacked architectures, validating successful integration of Pd and NiO within the borophene matrix.69 However, the potential of porous borophene structures remains largely unexplored. To bridge this gap, Yang et al. reported a multifunctional borophene hybrid foam (named CMB foam), integrating the exceptional properties of MXene with borophene in a three-dimensional porous framework.70 Borophene nanosheets were synthesized via aqueous-phase liquid exfoliation of boron powder under controlled ultrasonication conditions, followed by sedimentation to obtain a stable dispersion. The porous configuration increases surface area, reducing the transport pathway for electrolyte ions and significantly improving energy storage performance. CMB foam also exhibits excellent thermal management and insulation properties. HRTEM analysis revealed ultrathin, layered sheets with lateral dimensions in the sub-micron range and distinct lattice fringes (0.504 nm) corresponding to the (104) planes of β-rhombohedral boron. SAED patterns confirmed preserved crystallinity. XPS spectra showed dominant B–B bonding with minimal oxidation, indicating structural integrity and electronic stability. AFM analysis corroborated the formation of few-layer borophene with smooth topography and uniform thickness, validating successful aqueous exfoliation.70 Wu et al. reported an epitaxial growth of α′-borophene on multilayer graphene which enabled the fabrication of a borophene–graphene/n-Si Schottky photodetector exhibiting a broadband photoresponse (255–940 nm), high responsivity, superior detectivity, and rapid response/recovery times. These results clearly establish borophene as a promising material for next-generation optoelectronic devices, demonstrating enhanced performance compared to devices based on pristine borophene or graphene. Importantly, this work addresses key challenges in borophene synthesis by achieving well-defined vertical heterostructures with graphene.71
3. Properties of the borophene nanostructure relevant to sensing applications
3.1 Electronic properties
Borophene, a metallic Dirac material composed of elemental boron, is believed to exhibit exceptional mechanical and electrical properties. It displays a remarkable range of electronic characteristics, such as intrinsic metallicity, highly directional conductivity, and exceptional carrier mobility, which positions borophene as one of the most promising materials for advanced electronic sensing platforms. Unlike graphene, which often necessitates chemical doping or bandgap engineering to exhibit semiconducting behavior, borophene inherently functions as a metal with anisotropic conductivity.15 This unique property facilitates swift charge transport along specific crystallographic directions, enabling enhanced performance in electronic applications. Xu et al. reported that the predicted highest electron mobility reaches 2.01 × 104 cm2 V−1 s−1, implying the possibility of a semiconductor device with low-power consumption.72 This significant mobility surpasses that of many traditional two-dimensional materials, highlighting borophene's potential in high-performance electronic devices. In sensor applications, these exceptional properties translate directly into outstanding electrical sensitivity. Even subtle alterations in the local electronic environment, prompted by molecular adsorption or binding, can lead to substantial changes in the material's conductivity. Theoretical models for gas sensing have illustrated borophene's remarkable responsiveness to various gases such as ammonia (NH3), nitrogen monoxide (NO), and hydrogen sulfide (H2S), showcasing high selectivity and efficient charge transfer.73 Furthermore, the metallic characteristics of borophene enable zero-barrier electron injection in field-effect transistor (FET)-based sensors, significantly boosting device performance for rapid response and low-voltage detection. This combination of features makes borophene an exciting material for future technological advancements in sensing and electronic applications.3.2 Optical properties
The optical properties of borophene present a fascinating opportunity for advancements in optoelectronic and photonic sensors. This 2D material, borophene, characterized by its delocalized electronic states and unique anisotropic structure, demonstrates robust absorption across a wide spectrum, spanning the ultraviolet, visible, and near-infrared regions. Such characteristics make borophene exceptionally well-suited for the development of broadband photodetectors.74 Furthermore, borophene's inherent in-plane anisotropy gives rise to direction-dependent optical transitions, allowing for innovative designs in polarization-sensitive photodetectors and cutting-edge optical biosensors. The ability of borophene to support plasmonic resonances within the visible and near-infrared ranges is particularly noteworthy due to its high carrier density (approximately 1019 m−2), as this feature is rare among 2D materials.75 This property enables borophene to act as a substrate for localized surface plasmon resonance (LSPR)-based sensing, facilitating the detection of analytes through minute changes in the refractive index near its surface.76Recent theoretical studies have revealed that the optical conductivity of borophene can be dynamically modulated through external electric fields, providing exciting possibilities for real-time tunability in sensor readouts. In addition, the interactions between borophene and fluorescent molecules or quantum dots can be harnessed for innovative fluorescence quenching-based detection methods, significantly broadening their application potential in biosensing and molecular diagnostics. This remarkable versatility positions borophene as a promising material in the field of advanced sensor technology.
3.3 Mechanical and thermal properties
Mechanically, borophene stands out as one of the most robust two-dimensional materials known, showcasing impressive attributes such as high stiffness, tensile strength, and remarkable stretchability.77–80 These characteristics make it particularly well-suited for advanced wearable and deformable sensor systems. The Young's modulus of borophene varies based on its crystalline phase and orientation, with values frequently surpassing 200 GPa nm. This positions borophene as a formidable competitor to graphene in terms of strength, while its unique buckled structure grants it superior flexibility in-plane. Cutting-edge first-principles simulations reveal that borophene can endure strains of up to 20% without succumbing to structural failure, a trait that is exceptionally beneficial for flexible sensor substrates. This extraordinary mechanical resilience ensures that borophene maintains consistent and reliable sensing performance even under conditions of bending, twisting, or stretching, making it invaluable for applications such as strain sensors, e-skins, and soft robotics.11 In these innovative applications, borophene-based films or composites can function dually as both the sensing layer and the electrical conducting channel. This dual functionality simplifies device design while enhancing overall reliability.Borophene demonstrates outstanding thermal properties, including stability and conductivity, which are closely tied to its structure. Different phases, such as β12, δ6, and χ3, have been identified experimentally17,34,81 with the δ6 phase being particularly thermally unstable when detached from its substrate. To manage this instability, Zhou et al.79 developed empirical potentials for molecular dynamics simulations to analyze interactions between low-energy structures and borophene. Another approach to enhance stability is through full hydrogenation of borophene.82 Recent studies highlight that borophene exhibits superior lattice thermal conductivity in the ballistic regime, surpassing graphene, due to the lightweight boron atoms, short B–B bonds, and strong structural anisotropy.83 Molecular dynamics (MD) simulations show that thermal conductivity increases with specimen length until it approaches its phonon mean free path, but becomes inhibited in infinitely long structures due to buckling, which enhances out-of-plane phonon scattering.84 The unique bulk structure of borophene results in anisotropic thermal transport. Low-frequency phonons behave similarly to graphene with nearly isotropic transmission, while high-frequency phonons show one-dimensional transmission, leading to ultrahigh thermal conductivity.83 The differences in phonon scattering rates result in varying thermal conductivity and properties. For instance, the β12 phase of borophene is characterized by significant anisotropy, in contrast to the isotropic nature of the α-phase. Furthermore, the α phase is inherently stable, while the δ6 phase lacks thermal stability.83,85
4. Mechanistic insights and sensing modalities of borophene-based nanomaterials
In the following section of this review, we are going to discuss in detail the various sensing techniques along with their mechanism of action for the detection of various analytes. We mainly focus on colorimetric, fluorometric, and electrochemical-based sensing of borophene-based nanomaterials in this review.4.1 Colorimetric detection mechanism of borophene-based nanomaterials
Colorimetric detection techniques offer several advantages over traditional analytical methods such as UV-vis spectroscopy, HPLC, mass spectrometry, and atomic absorption spectroscopy. These advantages make them particularly appealing for environmental monitoring and on-site diagnostics. Unlike conventional methods, which often require complex instruments, trained personnel, and lengthy sample preparation, colorimetric sensors provide rapid, simple, and cost-effective detection with minimal equipment. The ability of colorimetric sensors to produce visible color changes allows for immediate interpretation without the need for specialized tools, supporting real-time monitoring and point-of-care testing. Additionally, the integration of functional nanomaterials and recognition elements such as aptamers or nanozymes enhances their sensitivity and selectivity, often achieving detection limits comparable to those of laboratory-based techniques. Furthermore, the portability, low operational costs, and ability to conduct large-scale analysis give colorimetric sensors a significant advantage in field applications and resource-limited settings where traditional techniques are impractical or inaccessible. In this section, the focus is primarily on the mechanism of action of colorimetric sensors based on borophene and its nanocomposites.Enzymatic and nanozyme-based catalysis constitutes one of the most effective methods for colorimetric sensing, particularly for the detection of various biological and environmental analytes such as glucose, H2O2, heavy metals, antioxidants, etc. The core principle relies on catalytic oxidation reactions that produce a visible color change, usually involving a chromogenic substrate such as 3,3′,5,5′-tetramethylbenzidine (TMB), 2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), or o-phenylenediamine (OPD), etc. Natural enzymes, such as horseradish peroxidase (HRP), have historically been used in biosensing because of their high specificity and catalytic efficiency. However, these natural enzymes often encounter issues such as high costs, low stability under different temperature and pH conditions, and complicated immobilization processes. These challenges have prompted the development of nanozymes, nanomaterials that replicate enzyme-like functions while being more stable, cost-effective, and easier to handle.
The mechanism for nanozyme-mediated colorimetric sensing generally involves a colorless substrate (like TMB) being oxidized in the presence of H2O2, facilitated by either a natural enzyme or a nanozyme. This oxidation transforms TMB into a blue-colored oxidized product, which can be observed visually or measured using a UV-vis spectrophotometer. For instance, Gogoi et al. reported that BQDs are capable of mimicking the activity of natural peroxidase enzymes by catalyzing the decomposition of H2O2 into ROS, primarily ˙OH radicals.10 In the presence of H2O2, BQDs facilitate the oxidation of chromogenic substrates such as TMB, which produces a blue-colored oxidized product measured at 652 nm, and ABTS, which yields a green-colored oxidized product measured at 414 nm. Initially, TMB or ABTS adsorbs onto the surface of BQDs through electrostatic interactions, leading to electron transfer from the substrate to the BQDs and the subsequent generation of ˙OH radicals from H2O2. The generated radicals oxidize the substrates, causing a visible color change (Fig. 5a). When antibiotics like oxytetracycline (OTC) or tetracycline (TC) are introduced, they interact with the BQDs or scavenge the ˙OH radicals, inhibiting the oxidation of TMB or ABTS and resulting in a reduction in color intensity. This decrease in absorbance is proportional to the antibiotic concentration, enabling quantification. Mechanistic validation was achieved through radical trapping experiments with terephthalic acid (TA) and various scavengers (IPA, methanol, EDTA, and benzoquinone), which confirmed ˙OH radicals as the primary reactive species in the system.10
 | ||
| Fig. 5 Plausible mechanism for (a) oxidation of TMB and ABTS by the POD-like activity of BQDs; reprinted with permission from ref. 10 Copyright 2024 American Chemical Society. (b) Generation of color via oxidation of TMB using dual enzymatic activities of Au@BNSs. Reprinted with permission from ref. 87 Copyright 2025 American Chemical Society. | ||
Borah et al. reported a colorimetric sensor based on metal-free, biocompatible borophene nanosheet photonanozyme for the selective and colorimetric detection of DA.34 Borophene nanosheet exhibited intrinsic POD-like activity that is significantly enhanced under visible light irradiation. The mechanism involves the photoexcitation of borophene nanosheet, which have a direct bandgap of approximately 2.36 eV, making them suitable for absorbing light at 440 nm. When exposed to light, electrons in the borophene nanosheet are excited from the valence band to the conduction band, generating electron–hole pairs. These photoexcited charge carriers catalyze the decomposition of H2O2 into ROS, including ˙OH radicals, O2˙− anions, and photogenerated holes (h+). These ROS are primarily responsible for oxidizing the chromogenic substrate TMB into its blue-colored oxidized form, which exhibits strong absorbance at 652 nm. DA, which contains electron-rich catechol groups, readily reacts with or scavenges ROS due to its high redox activity. When DA is added to the borophene nanosheet + H2O2 + TMB system, it competes with TMB for the available ROS, effectively suppressing TMB oxidation. This leads to a noticeable decrease in blue coloration, which can be measured spectrophotometrically. The extent of decrease in the absorbance at 652 nm is directly proportional to the concentration of DA present. This strategy provides a sensitive and selective platform for DA detection, with minimal interference from structurally similar biomolecules.
On the other hand, the nanozyme-assisted colorimetric detection of some analytes involves a two-step cascade reaction. One such analyte is glucose, and the colorimetric detection of glucose using borophene-based sensor was studied by Sun et al.86 In the study, oxidized boron nanosheets (ox-BNSs) were used as nanozyme catalysts for the colorimetric detection of glucose through a two-step cascade mechanism. In the first step, glucose being enzymatically oxidized by glucose oxidase (GOx), a natural enzyme, resulted in the production of gluconic acid and H2O2. The H2O2 thus generated then produced ROS through a POD-mimicking reaction catalyzed by ox-BNSs, where the colorless substrate TMB was oxidized to form a blue-colored product (ox-TMB), leading to a measurable absorbance at 652 nm. The ox-BNSs exhibited intrinsic POD-like activity due to their oxygen-rich surface and multilayered structure, which promote efficient electron transfer during the catalytic cycle. The intensity of the resulting blue color is directly proportional to the glucose concentration, facilitating sensitive quantification.
In a recent study, Borah et al. reported gold nanoparticle decorated borophene nanosheets (Au@BNSs) for the colorimetric detection of glucose.87 The colorimetric sensing mechanism that employs Au@BNSs is based on their intrinsic dual enzyme-mimetic activities, GOx- and POD-like activities, allowing for effective glucose detection. In the presence of glucose, Au@BNSs facilitate its oxidation to gluconic acid and H2O2, effectively mimicking the action of natural GOx. This conversion takes place through the interaction of glucose with the electron-rich gold nanoparticles (AuNPs) on the borophene surface, activating molecular oxygen (O2) and creating a dioxogold intermediate (such as Au+–O22− or Au2+–O22−). This process enables the transfer of electrons from glucose to O2, resulting in the production of H2O2. The in situ generated H2O2 is then decomposed by the POD-like activity of Au@BNSs, producing highly reactive ˙OH radicals, which subsequently oxidize the chromogenic substrate TMB into blue colored OxTMB, leading to a visible color change (as shown in Fig. 5b). The combination of conductive, hydroxyl-functionalized borophene nanosheets and catalytic AuNPs enhances substrate affinity, promotes electron transfer, and ensures high catalytic efficiency and stability.
4.2 Mechanistic insights into the fluorometric sensing of borophene-based nanomaterials
BQDs and their derivatives have garnered significant attention in fluorometric sensing due to their remarkable photoluminescence (PL) characteristics. The strong and tunable PL exhibited by these nanomaterials arises primarily from surface states, edge effects, and quantum confinement inherent to their two-dimensional nanostructure. These unique features enable efficient electron excitation and relaxation processes, producing intense fluorescence emission as electrons return to their ground state from higher energy levels. The presence of particular analytes can modify this fluorescence through a variety of interactions, leading to sensitive and selective detection. Such modulation typically occurs via mechanisms like photoinduced electron transfer (PET), fluorescence resonance energy transfer (FRET),88 or the inner filter effect (IFE), all of which influence fluorescence intensity, emission wavelength, and excited-state lifetimes. Turn-on or turn-off fluorescent chemosensors are commonly developed by modulating fluorescence intensity through mechanisms such as PET,89 intramolecular charge transfer (ICT),90 chelation-enhanced fluorescence (CHEF),91 aggregation-induced emission (AIE),92 or aggregation-caused quenching (ACQ).93 In contrast, ratiometric fluorescent chemosensors rely on emission wavelength shifts, which are typically governed by ICT, FRET, through-bond energy transfer (TBET), or excited-state intramolecular proton transfer (ESIPT).94 Together, these diverse photophysical mechanisms enable the design of highly sensitive and selective fluorescent sensing platforms. In all cases, the sensing strategy relies on strong analyte and QD interactions, optical overlap, and changes in fluorescence behavior to enable highly sensitive, selective, and often visually distinguishable detection.Gogoi et al. reported that BQDs exhibit strong blue fluorescence, with an emission maximum around 400 nm when excited at 310 nm. This fluorescence arises from quantum confinement effects and surface state emissions.10 Upon the stepwise addition of oxytetracycline (OTC) and tetracycline (TC), a systematic quenching of fluorescence intensity was observed, as shown in Fig. 6a, which was attributed to IFE rather than dynamic or static quenching mechanisms. In this IFE-based sensing mechanism, the absorption spectra of OTC and TC substantially overlap with both the excitation and emission spectra of BQDs. This spectral overlap leads to the reabsorption of excitation and/or emitted photons by the analytes, resulting in diminished fluorescence intensity. This turn-off fluorescence behavior provides a simple, sensitive, and effective approach for detecting tetracycline-class antibiotics in aqueous media, further highlighting the potential of BQDs as dual-mode nanozyme platforms.10
 | ||
| Fig. 6 (a) Schematic representation of fluorescence “turn on–off” of borophene QDs with OTC and TC interaction; reprinted with permission from ref. 10 Copyright 2021 Elsevier. (b) Schematic representation of DHEA sensing using BQDs as a nanoprobe via a fluorescence enhancement mechanism; reprinted with permission from ref. 55 Copyright 2021 American Chemical Society. (c) Schematic representation of a borophene-based composite/hybrid modified electrochemical biosensor system and its sensing mechanisms. Reprinted with permission from ref. 104 Copyright 2024 Wiley. | ||
Joshi et al. reported that the turn-on fluorometric sensing of dehydroepiandrosterone (DHEA) using BQDs is primarily governed by an aggregation-induced emission (AIE) mechanism.55 This response is driven by strong intermolecular interactions between DHEA molecules and surface-functionalized BQDs, which trigger nanoparticle aggregation as shown in Fig. 6b. Upon DHEA addition, high-resolution TEM and dynamic light scattering (DLS) confirmed the transformation of BQDs from discrete nanodots into large aggregates, indicating DHEA-induced clustering. This aggregation restricted intramolecular motions and reduced non-radiative decay pathways, resulting in enhanced fluorescence, a characteristic feature of AIE. Supporting spectroscopic evidence includes a red shift in UV-vis absorption (from 304 to 309 nm), FTIR spectral changes indicating functional group interactions, and a notable decrease in zeta potential (from +31.2 mV to +8.8 mV), suggesting electrostatic screening and surface charge neutralization. Fluorescence lifetime measurements further confirmed the AIE mechanism, showing an increase from 1.09 ns (BQDs alone) to 1.72 ns (BQDs + DHEA), indicative of enhanced radiative decay. Mechanistically, DHEA interacts with B–OH and B–O groups on the BQD surface, likely through hydrogen bonding, π–π stacking, and hydrophobic interactions, promoting aggregation and fluorescence enhancement. This AIE-driven “turn-on” response enables sensitive and selective fluorometric detection of DHEA at nanomolar levels.
Aswal et al. investigated the fluorescence sensing mechanism of BQDs, synthesized via a contamination-free LPE method.56 These BQDs exhibit strong visible PL with a high quantum yield of ∼40% and excitation-independent emission, indicative of uniform size distribution and intrinsic defect states. The sensing mechanism for Fe3+ involves a pronounced quenching of PL intensity (over 90%) upon interaction, attributed primarily to photoinduced charge transfer (PCT) and partial energy transfer (PET) processes. The quenching is facilitated by the spectral overlap between BQD emission and Fe3+ absorption, along with the strong coordination between Fe3+ and hydroxyl functional groups on the BQD surface. Notably, the PL quenching displays a nonlinear response to Fe3+ concentration, deviating from the classical Stern–Volmer behavior, suggesting a complex quenching mechanism involving static and dynamic components. These findings highlight the mechanistic basis for using BQDs as selective and sensitive fluorescent nanosensors for Fe3+ ions.60 While fluorescence-based detection offers rapid, non-invasive analysis with high sensitivity, exploring alternative transduction mechanisms is equally important to broaden application scopes. In this context, electrochemical sensing has emerged as a powerful approach due to its simplicity, low cost, and ability to provide real-time quantitative data. The following section delves into the electrochemical sensing mechanisms of borophene-based materials, highlighting their behavior, electron transfer capabilities, and application in sensing platforms.
4.3 Electrochemical sensing mechanism of borophene-based nanomaterials
Borophene has emerged as a highly promising material for electrochemical sensing due to its superior chemical reactivity, high electron mobility95–97 and tunable anisotropic conductivity. Its textured and perforated surface provides abundant active sites for analyte adsorption, enhancing sensitivity and facilitating efficient charge transfer. These features, combined with its excellent intrinsic catalytic activity, make borophene particularly suitable for enzyme-free and catalytic sensing platforms. Additionally, its fast electron transport capabilities allow for rapid signal transduction, while hybridization with other materials improves stability and current response.98 These advantages collectively establish borophene as a highly promising material for next-generation electrochemical sensors, offering exceptional sensitivity, selectivity, and responsiveness.63,97,99,100In addition to these merits, the strong interfacial interactions of borophene with various chemical species further contribute to its effectiveness in analyte recognition.101,102 Under certain electrochemical circumstances, borophene can remain stable, which is essential for guaranteeing the long-term functionality and dependability of sensors. However, environmental conditions may affect its stability.103 The borophene-based composite/hybrid electrochemical biosensor system and its analytical sensing mechanisms are illustrated in Fig. 6c. This suggests that borophene's electrical performance is greatly improved by developing hybrids or composites with other materials. When compared to pristine borophene, these borophene-based materials show significantly larger current outputs, suggesting that the composite or hybrid structure has improved electrical conductivity. The identification of the target analyte, the transduction of the recognition event into a measurable signal, the amplification of this signal to improve detectability, and the identification and quantification of the analyte based on the amplified response are the four main steps that usually comprise the electrochemical sensing process.104
Gülsen Baytemir reported that the electrochemical sensing of DA using a borophene–PANI nanocomposite as a non-enzymatic sensor is attributed to the enhanced redox activity at the modified electrode interface.105 DA, a redox-active catecholamine, undergoes a reversible two-electron, two-proton oxidation to form DA-o-quinone. This redox process is significantly facilitated by the synergistic interaction between PANI and borophene. PANI provides redox-active sites and proton conductivity, while borophene contributes a high electroactive surface area, excellent electrical conductivity, and π–π interactions with DA, which promote selective adsorption. Additionally, borophene acts as an efficient electron mediator, accelerating charge transfer at the electrode surface. The hybrid nanocomposite structure not only lowers the overpotential but also amplifies the electrochemical signal, resulting in improved sensitivity and selectivity. As a result, the sensor demonstrated a high sensitivity of 385.05 µA µM−1 cm−2 and a low detection limit of 0.017 µM, outperforming PANI-only systems and highlighting the pivotal role of borophene in enhancing electrochemical DA sensing.
Similarly, Güngör et al. reported that the electrochemical sensing of urea using a CuPc–borophene nanocomposite sensor operates via a non-enzymatic redox mechanism enhanced by the synergistic interplay between copper(II) phthalocyanine (CuPc) and borophene nanosheets.106 Upon hydrolysis, urea produces ions like ammonium and carbonate, which enhance the conductivity of the sensing layer. Simultaneously, CuPc undergoes redox cycling, where the Cu2+/Cu+ couple facilitates electron transfer. Cu2+ is chemically reduced by urea to Cu+ and then electrochemically re-oxidized, producing a measurable current response. CuPc serves as an efficient electrocatalytic center due to its redox-active metal core and π-conjugated structure, while borophene significantly improves the electrical conductivity, surface area for analyte adsorption, and dispersion of CuPc. This synergistic effect lowers the redox overpotential, boosts signal intensity, and accelerates electron transfer kinetics, resulting in a high sensitivity of 10.43 µA µM−1 cm−2, a low detection limit of 0.05 µM, and a rapid response time of approximately one minute.
Again, Kaimal et al. reported freestanding borophene nanosheet, synthesized via solvothermal-assisted sonochemical exfoliation of bulk boron in acetone, which were utilized to modify a glassy carbon electrode (GCE) with a Nafion binder, forming the borophene nanosheet modified GCE (BNSs/GCE) sensor for electrochemical detection of As(III). The sensing mechanism is primarily driven by borophene's exceptional electrical conductivity and metallic nature, which significantly enhance electron transfer kinetics, as evidenced by increased peak currents and reduced peak-to-peak separation in cyclic voltammetry compared to bare GCE. Upon interaction with As(III), the BNSs/GCE exhibits distinct anodic peaks at +0.74 V and +0.58 V, corresponding to the redox transitions of arsenic, facilitated by the abundant surface-active sites and high electron mobility of borophene. Scan rate-dependent studies reveal a linear relationship between peak current and the square root of scan rate, indicating a diffusion-controlled, surface-confined electrochemical oxidation process. Furthermore, electrochemical impedance spectroscopy (EIS) confirms a substantially lower charge transfer resistance for BNSs/GCE, validating the improved interfacial conductivity and rapid electron transfer characteristics that underpin the efficient and selective sensing of As(III).100
5. Emerging sensing applications of borophene-based nanomaterials
Borophene, a two-dimensional boron-based material, exhibits outstanding mechanical strength, electrical conductivity, thermal stability, and a high surface-to-volume ratio, making it a compelling candidate for next-generation sensor technologies. Moreover, their biocompatibility and surface functionalization capabilities make them promising candidates in the field of drug delivery systems, bio-imaging, sensing and diagnostic applications. Its inherent chemical reactivity and ability to form hybrid structures with metals, metal oxides, and polymers significantly enhance the sensitivity, selectivity, and response time of sensing platforms. Borophene-based sensors have shown remarkable promise across a range of applications, including medical diagnostics, environmental monitoring, and wearable electronics. Conventional detection techniques, although accurate and reliable, often suffer from limitations such as high operational costs, complex instrumentation, and the need for trained personnel. These drawbacks have fuelled growing interest in portable, cost-effective, and rapid sensing alternatives. In this context, borophene-based nanozyme-assisted colorimetric, fluorescence, and electrochemical sensors are emerging as efficient and user-friendly platforms for on-site and point-of-care diagnostics in both environmental and biomedical domains. In Table 1, the detection of various environmental pollutants and biomarkers using different 2D nanomaterial-based systems is summarized.| Target analyte | 2D nanomaterial | Sensing technique | Limit of detection (LOD) | Ref. | |
|---|---|---|---|---|---|
| Environmental pollutant | As(III) ions | PbO/rGO | Electrochemical sensing | 0.01 µM | 115 |
| Gr-nPt | Electrochemical sensing | 1.1 nM | 116 | ||
| Fe3O4/rGO | Electrochemical sensing | 0.12 ppb | 117 | ||
| Au/RGO | Electrochemical sensing | 0.1 ppb | 118 | ||
| Oxytetracycline | Graphene–gold nanoparticles | Colorimetric sensing | 0.091 µM | 119 | |
| Biographene | Colorimetric sensing | 2.66 µM | 120 | ||
| Tetracycline | Amino-functionalized Al-MOF | Fluorometric sensing | 26.16 nM | 121 | |
| Fe3+ ions | Graphene quantum dots | Fluorometric sensing | 0.45 µM | 122 | |
| S-GQDs | Fluorometric sensing | 4.2 nM | 123 | ||
| H2S gas | Cu/GO | Gas sensing | 4.155 ppm | 124 | |
| SnO2/rGO | Gas sensing | 43 ppb | 125 | ||
| PANI/GO | Gas sensing | 1 ppm | 126 | ||
| Nitrogen dioxide | MoS2 | Gas sensing | 150 ppb | 127 | |
| Biomarker | Glucose | Cu–Ag/rGO | Colorimetric (nanozyme based) | 3.8 µM | 128 |
| Ni-MoS2 | Electrochemical sensing | 0.31 μM | 129 | ||
| Au/MOF | Colorimetric (POD, Gox) | 8.5 µM | 130 | ||
| Dopamine (DA) | Pt/h-BN nanosheets | Colorimetric (nanozyme based) | 0.76 µM | 131 | |
| CG nanoparticles | Colorimetric (nanozyme based) | 1.15 µM | 132 | ||
| Graphdiyne quantum dots (GDY QDs) | Colorimetric (nanozyme based) | 8.65 µM | 133 | ||
| Glutathione (GSH) | MnPc@GNP | Colorimetric (nanozyme based) | 1.23 µM | 134 | |
| AuAg–rGO | Colorimetric (nanozyme based) | 38 nM | 135 | ||
| Hydrogen peroxide (H2O2) | Ni-MOF nanosheets | Colorimetric (nanozyme based) | 8 nM | 136 | |
| PtS2 nanosheets | Colorimetric (nanozyme based) | 29.95 µM | 137 | ||
| Folic acid | rGo–AuNPs | Electrochemical sensing | 3.1 nM | 138 | |
| MoS2 | Electrochemical sensing | 7.5 nM | |||
| Graphene–ZnO | Electrochemical sensing | 6 nM | |||
5.1 Borophene-based nanomaterials for environmental sensing
Borophene-based sensors have emerged as promising tools for environmental monitoring due to their exceptional surface reactivity, high electrical conductivity, and adaptability in forming hybrids and composites. These properties enable the sensitive and selective detection of diverse environmental contaminants even at trace levels, including antibiotics, heavy metals, industrial toxins, organic pollutants, toxic gases, and humidity. Among these, antibiotic contamination poses a significant threat to human and environmental health due to widespread use and improper disposal. Broad-spectrum antibiotics such as OTC and tetracycline (TC), though extensively employed in healthcare, agriculture, and animal husbandry, can lead to toxicity and antibiotic resistance if misused. Similarly, heavy metals like Fe3+ while essential in biological systems, can cause serious health disorders when present in excess. In addition, the growing impact of air pollutants like NOx, SOx, CO, and H2S on public health and the environment has shown the importance of developing low-cost, high-performance borophene-based gas sensors for real-time environmental surveillance. To address all these, borophene-based nanostructures have been developed as efficient sensing platforms for their detection. Here in this section, we will be discussing the application of borophene-based sensors in the field of environmental sensing.For instance, Gogoi et al. reported that the intrinsic POD-mimicking activity of BQDs has been effectively harnessed for the colorimetric detection of broad-spectrum antibiotics such as OTC and TC.10 In this approach, commonly used chromogenic substrates TMB and ABTS were employed in the presence of hydrogen peroxide (H2O2). BQDs facilitated the decomposition of H2O2 to generate ˙OH radicals, which oxidized TMB and ABTS into colored products with characteristic absorbance peaks at 652 nm and 414 nm, respectively. However, the presence of OTC or TC led to a concentration-dependent decrease in absorbance intensity due to their ˙OH scavenging ability, allowing quantification based on this inhibition. The assay demonstrated good linearity in the 0–200 µM range with detection limits of 1.10 µM (OTC) and 1.02 µM (TC) for TMB, and 1.03 µM (OTC) and 1.02 µM (TC) for ABTS as shown in Fig. 7a–h. Notably, the catalytic performance remained stable over 120 days of storage, indicating high robustness and shelf-life. In parallel, BQDs also exhibited strong blue fluorescence with an emission maximum around 400 nm upon 310 nm excitation. The fluorescence intensity was progressively quenched with increasing concentrations of OTC and TC (0–300 µM), attributed to the inner filter effect (IFE) arising from spectral overlap between the antibiotic absorption and BQD excitation/emission bands. This turn-off fluorescence response enabled sensitive and rapid quantification, yielding LODs of 1.14 µM (OTC) and 1.08 µM (TC). Importantly, the dual-mode (colorimetric and fluorometric (Fig. 7i) sensing platform was validated in real sample matrices including river water, milk, and animal tissue lysates, achieving recovery rates between 96% and 103%. These results underline the practical applicability, stability, and versatility of BQDs for antibiotic detection in complex environmental and biological samples. Moreover, the selectivity of BQDs for colorimetric detection of OTC and TC was assessed using TMB and ABTS substrates in the presence of common interfering molecules (e.g., erythromycin, ciprofloxacin, norfloxacin, diclofenac, ofloxacin, enrofloxacin). A significant absorbance change was observed only with OTC and TC, while interference from other compounds was negligible. These results confirm the high selectivity and sensitivity of the BQD-based assay for OTC and TC detection.10
 | ||
| Fig. 7 Colorimetric sensing of OTC and TC using (a)–(d) TMB substrate; (e)–(h) ABTS substrate; (i) schematic representation of both colorimetric and fluorometric sensing of BQD; reprinted with permission from ref. 10 Copyright 2024 American Chemical Society. (j) Schematic representation of As3+ sensing via an electrochemical method; reprinted with permission from ref. 100 Copyright 2023 American Chemical Society. (k) PL quenching ratios of BQDs in the presence of different metal ions, including mixed ions; (l) PL spectra (with 360 nm excitation) of BQDs in the presence of Fe3+ aqueous medium at different concentrations; (m) PL quenching ratio as a function of Fe3+ ion concentration showing a nonlinear Stern–Volmer plot; (n) TRPL decay profile of BQDs without and with various concentrations of Fe3+, reprinted with permission from ref. 56 Copyright 2024 American Chemical Society. (o) Schematic showing the sensing mechanism in the ECR sensor. Reprinted with permission from ref. 110 Copyright 2024 American Chemical Society. | ||
Kaimal et al. reported the development of borophene nanosheet-modified glassy carbon electrode (BS/GCE) for the electrochemical detection of As(III) ions in groundwater.100 They reported the synthesis of few-layered, large flake-sized freestanding borophene (BS) using a solvothermal-assisted sonochemical exfoliation method. By adding the synthesized borophene to a glassy carbon electrode (GCE) and seeing how it behaved using cyclic voltammetry (CV) and differential pulse voltammetry (DPV), the electrochemical performance was assessed. With a remarkable low limit of detection (LOD) of 0.1 µM, the BS-modified GCE demonstrated a linear response toward As(III) concentrations ranging from 0.1 to 20 µM. Importantly, the sensor's practical value was confirmed through testing with actual groundwater samples from areas affected with arsenic. Excellent sensitivity, robust selectivity, outstanding repeatability, and reusability were all displayed by the BS-modified electrode. Additionally, it demonstrated strong anti-interference capabilities, guaranteeing accurate detection in intricate environmental matrices. Its potential for public health monitoring is highlighted by the fact that the LOD obtained is much lower than the allowable limits for As(III) in drinking water, with similar precision in real sample analysis, the borophene-based electrochemical sensor provides a more affordable, quick, and field-deployable alternative to traditional high-end analytical techniques like inductively coupled plasma mass spectrometry (ICP-MS). With significant implications for environmental monitoring and public health protection, this work highlights the potential of borophene nanosheets as an effective electrode material for creating portable, affordable sensors for As(III) detection100 (Fig. 7j).
Aswal et al. reported the selective and ultrasensitive fluorometric detection of Fe3+ ions as an environmental pollutant using BQDs. They investigated the fluorescence sensing mechanism of BQDs, which exhibit strong and stable photoluminescence (PL) due to their unique quantum confinement effects and electronic structure. Additionally, unlike other 2D quantum dots, the BQDs exhibit excitation-independent emission, meaning that their fluorescence wavelength remains constant regardless of the excitation energy. With a special affinity for Fe2+ ions, these BQDs exhibit exceptional sensitivity and selectivity for metal ion detection. The fluorescence is considerably suppressed (>90% quenching ratio) upon contact with Fe3+, with an ultralow detection limit of roughly 5 nM. The fluorescence quenching observed in BQDs upon interaction with Fe3+ ions is governed by multiple synergistic mechanisms. One key process is photoinduced electron transfer (PET), wherein excited-state electrons are transferred from the BQDs to Fe3+ ions, thereby diminishing the fluorescence signal. Additionally, the inner filter effect (IFE) contributes to the quenching, as Fe3+ ions absorb light at both the excitation and emission wavelengths, leading to an apparent reduction in fluorescence intensity. Furthermore, charge trapping at surface states plays a significant role; the adsorption of Fe3+ ions onto the BQD surface introduces trap states or defects that disrupt electron–hole recombination, further suppressing photoluminescence. Collectively, these mechanisms account for the high quenching efficiency and sensitivity of BQDs toward Fe3+ detection. Density functional theory (DFT) calculations supported the experimental findings by confirming the feasibility of charge transfer between the BQDs and Fe3+ ions. A comprehensive study was also conducted on the coordination behavior of various metal ions (Fe3+, Cu2+, Ag+, Zn2+, K+, Pb2+, Mg2+, Na+, Ni2+, Hg2+, Mn2+, Ce4+, and Fe2+) with BQDs using both PL measurements and DFT analyses. Among these, Fe3+ showed the most pronounced quenching effect, highlighting the sensor's selectivity. A nonlinear Stern–Volmer response was observed in the fluorescence quenching behavior. Fig. 7k shows a histogram that amply illustrates the enhanced sensitivity and selectivity of BQDs toward Fe3+. As Fe3+ concentrations increased, Fig. 7l shows there was a gradual decrease in PL intensity. Although a linear trend was visible in the lower concentration range (50–500 nM), which is comparable to observations with BiO2Se2107 and graphene QDs,108 the nonlinear quenching profile (Fig. 7m) significantly deviates from the standard linear Stern–Volmer model. The charge transfer process was further validated by time-resolved PL measurements. When 0.5 µM Fe3+ was added, the average carrier lifetime decreased from 1.8 ns to 0.9 ns (Fig. 7n), indicating quicker recombination brought on by electron transfer. All things considered, this work emphasizes how surface contacts, quantum confinement, and photoinduced charge transfer control the fluorescence sensing behavior of BQDs. With promising uses in biosensing, environmental monitoring, and optoelectronic devices, the results demonstrate the potential of BQDs as efficient fluorescence-based sensors for trace-level detection of Fe3+.56 Tahani et al. reported that the borophene nanosheet-modified glassy carbon electrode (BPS/GCE) was developed for the electrochemical detection of lead (Pb2+) and mercury (Hg2+) ions. The electrochemical behavior of borophene-modified glassy carbon electrodes (GCEs) was evaluated using cyclic voltammetry (CV) and differential pulse voltammetry (DPV). The sensor exhibited a broad linear detection range of 0.1–100 µM, with remarkably low detection limits of 0.005 µM for Pb2+ and 0.01 µM for Hg2+. These values underscore the sensor's high sensitivity and strong potential for trace-level heavy metal ion detection. The electrochemical sensing mechanism is attributed to borophene's large surface area, metallic conductivity, and strong affinity for metal ions, which facilitate efficient adsorption and electron transfer. CV measurements revealed distinct oxidation peaks for Pb2+ (−0.09 V) and Hg2+ (+0.38 V), confirming their redox activity at the modified electrode surface. EIS demonstrated significantly reduced charge transfer resistance for the borophene-modified electrode compared to the bare GCE, indicating enhanced conductivity and improved electron mobility. DPV analyses provided precise quantification and confirmed the linearity of the calibration plots for both Pb2+ and Hg2+ detection. Selectivity studies showed minimal interference from other common metal ions such as Cu2+, Cd2+, Co2+, and Ni2+. However, a slight overlap in the oxidation peaks of Hg2+ and Pb2+ was observed, which may affect the accuracy of Pb2+ detection in the presence of high Hg2+ concentrations. Long-term stability tests indicated that the sensor retained approximately 94% of its original current response for Pb2+ after 15 days, demonstrating excellent durability and repeatability. Overall, the study highlights the promise of borophene nanosheets as an effective electrochemical sensing platform for the detection of toxic heavy metals, offering significant advantages in terms of sensitivity, selectivity, cost-efficiency, and applicability to real-world environmental monitoring.109
Hosseini et al. developed a novel electro-chemoresistance (ECR) sensor leveraging borophene's high surface activity, strong gas adsorption, and excellent conductivity for efficient H2S detection.110 Borophene, synthesized via Al-assisted CVD, enables dual-mode sensing by combining changes in electrical resistance (chemoresistance) with electrochemical responses from redox reactions on its surface. This dual-signal mechanism enhances both sensitivity and reliability. Compared to conventional sensors, the ECR device demonstrated 10-fold higher sensitivity than standard electrochemical sensors and 4-fold higher than standalone chemoresistive sensors, proving highly effective for detecting toxic and corrosive H2S111,112 in petroleum, natural gas, and biogas applications. Upon exposure to H2S, the sensor displayed a rapid response time of 6–8 seconds, along with high accuracy and repeatability. Significant variations in borophene's conductivity following H2S adsorption were detected by EIS, demonstrating its crucial role in improving sensor performance. In order to enable gas permeation and preserve sensor stability, the sensor's structural components include a graphite working electrode covered with borophene, a reference electrode, a counter electrode, and an electrolyte layer. All of these components are encased in a Nafion gas diffusion membrane. As illustrated in Fig. 7(o), the sensor mechanism involves H2S adsorption on the borophene surface, which alters the sheet conductance and thus the chemoresistive component (R cr) of the equivalent circuit. Simultaneously, electrochemical reactions at the electrode generate electrons and protons (H+ ions), contributing to a measurable electrochemical response. This synergistic interaction between borophene's electrical and electrochemical properties offers a robust platform for real-time and selective gas monitoring. Overall, this work positions borophene-based ECR sensors as promising candidates for next-generation gas sensing technologies, with wide-ranging applications in industrial safety, environmental monitoring, and healthcare diagnostics. Nitrogen dioxide (NO2) is a toxic gas and a significant air pollutant, known to cause serious health effects such as respiratory illnesses, and contribute to environmental problems, including acid rain. Similarly, Casanova et al. explore a sonochemical exfoliation method for synthesizing borophene nanolayers and evaluate their gas sensing capabilities, particularly for NO2 detection in humid conditions. The presence of humidity boosts sensitivity by ∼50%, with LOD and limit of quantification (LOQ) of 23 ppb and 76 ppb, respectively. Borophene demonstrates rapid recovery, repeatability, and selectivity against common interferents (CO2, benzene). NO2 adsorption increases charge carrier concentration, lowering resistance, with humidity acting synergistically to improve responses. This study highlights borophene's potential for practical air quality monitoring, offering a cost-effective, scalable, and high-performance alternative to traditional gas sensors.113 Zhao et al. reported an ultrasensitive NO2 gas sensor developed from a mixed-dimensional heterostructure combining 2D borophene sheets with 0D BC2N quantum dots. Both theoretical and experimental studies showed that NO2 interacts most strongly with this material, causing high charge transfer and clear changes in its electronic structure. The sensor demonstrated remarkable performance at room temperature, with a 108% response even at very low concentrations (0.2 ppm) and an ultrahigh 1170% response at 30 ppm NO2, greatly outperforming pristine borophene and many other low-dimensional sensors. This improvement is attributed to the cooperative effects at the 0D–2D interface, which enhance charge transfer, orbital interactions, and fast adsorption–desorption processes, making the heterostructure a highly promising platform for selective NO2 detection.114
5.2 Borophene-based nanomaterials for biomedical sensing
Biomedical sensing refers to the application of sensors for the detection and measurement of biological and chemical substances associated with human health. These sensors can detect various biomarkers, including glucose, DA, glutathione (GSH), H2O2, folic acid, etc. The primary objective of the sensing is to facilitate early disease diagnosis, health monitoring, and real-time detection of physiological changes. In recent times, biomedical sensors commonly employ nanomaterials, enzymes, or bio-recognition elements such as antibodies or aptamers to achieve high levels of sensitivity and selectivity. In this context, colorimetric sensing techniques are particularly advantageous due to their simplicity, affordability, and capacity to deliver visible results without the need for complex instruments, making them well-suited for point-of-care testing. In this section, we will be discussing the application of borophene-based sensors in the field of biomedical sensing. For instance, Cihat Taşaltın developed an innovative glucose sensor utilizing a nanocomposite made of polyacrylonitrile (PAN) and β-rhombohedral borophene and reported the detection of glucose by a non-enzymatic electrochemical oxidation mechanism. PAN provides nitrogen sites that interact weakly with hydroxyl groups (–OH) of glucose, assisting in its adsorption. Borophene significantly enhances this process by improving charge transfer efficiency, attributed to its negative zeta potential and high electrical conductivity. During voltammetric measurements, glucose is directly oxidized at the electrode surface, generating a measurable redox current. Importantly, the addition of the PAN:borophene nanocomposite shifted the redox peaks and enhanced the performance of the sensor, yielding a sensitivity of 8.43 µA mM−1 cm−2. The group reported a linear range of 1.5–12 mM with a LOD of 0.099 mM for the electrochemical sensing of glucose using the PAN:borophene-based sensor. The improvements in the electrochemical sensing are linked to borophene's unique electron-deficient structure, which promotes effective electron injection into the sensing layer, resulting in more robust and stable electrochemical signals compared to PAN alone.68Along the same lines, Taşaltın et al. developed a novel glucose sensor combining β12-borophene nanosheets with PANI to form a conductive nanofiber network.139 The borophene was prepared via environmentally friendly, sonication-assisted exfoliation of boron microparticles in DMF, and subsequently mixed with PANI in NMP, resulting in a randomly interconnected network. In this composite, glucose detection occurs through a non-enzymatic redox reaction at the electrode surface, where PANI enhances glucose adsorption and redox activity, while borophene facilitates electron transfer due to its high conductivity and negatively charged surface. This synergistic effect increases the number of electroactive sites, enabling more efficient glucose oxidation and producing a current signal proportional to glucose concentration. As a result, the PANI–borophene sensor demonstrated superior performance over PANI alone, achieving a sensitivity of 96.93 µA mM−1 cm−2 and a LoD of 0.5 mM, effectively covering the physiological glucose range of 1–12 mM. Similarly, Baytemir et al. developed a novel nanocomposite by combining nickel phthalocyanine (NiPc) and borophene nanosheets, resulting in a highly sensitive glucose sensor that does not require enzymes.140 Here, the mechanism of glucose detection relies on the redox cycling between Ni(II) and Ni(III) in the NiPc matrix. In the presence of glucose, Ni(II) is oxidized to Ni(III), which then catalyzes the oxidation of glucose to gluconolactone while being reduced back to Ni(II), thus forming a continuous catalytic cycle. Borophene plays a crucial role as an electron acceptor and conductive enhancer, significantly increasing the overall current response through rapid charge transfer. This improved electron mobility is attributed to borophene's metallic nature, high surface area, and nanostructured morphology, as confirmed by HRTEM, XRD, and SEM imaging. The resulting sensor demonstrated outstanding performance, with a sensitivity of 10.31 µA mM−1 cm−2 and a LOD as low as 0.15 µM. This is far superior to the NiPc-only sensor, which exhibited a sensitivity of 0.08 µA mM−1 cm−2 and a LoD of 3 µM. The study highlights the synergy between NiPc and borophene, illustrating the role of borophene in significant enhancement of the electrochemical properties and sensing capability, making the composite highly effective for glucose detection under physiological conditions. Recently, Baytemir developed an innovative sensor using a PANI/borophene nanocomposite for the sensitive and selective detection of DA.105 The DA detection mechanism is based on a non-enzymatic electrochemical redox reaction, where applied voltage oxidizes DA to DA-quinone, releasing electrons. PANI facilitates this process through its conjugated backbone and interactions with –NH2/–OH groups, while borophene enhances conductivity, electron transfer, and DA adsorption by increasing surface roughness and active sites. This synergy leads to a significant rise in redox peak currents, with the sensor achieving high sensitivity (385.05 µA µM−1 cm−2) and a low detection limit (0.017 µM), surpassing the performance of PANI alone.
In a recent study, Güngör et al. introduced a novel biosensor utilizing a copper phthalocyanine (CuPc)–borophene nanocomposite for the sensitive and selective detection of urea.106 The group reported a non-enzymatic redox process as the mechanism for urea sensing. When urea interacts with the CuPc in the nanocomposite, it experiences electrocatalytic oxidation, resulting in the reduction of Cu(II) to Cu(I). This redox cycle is coupled with the re-oxidation of Cu(I) back to Cu(II) at the electrode surface, producing a measurable anodic current. The inclusion of borophene significantly enhances this electrochemical reaction by improving the electrical conductivity, charge transfer rate, and electroactive surface area of the sensing layer. These enhancements result in a greater current response and increased sensitivity, with the CuPc–borophene sensor achieving a sensitivity of 10.43 µA mM−1 cm−2 and a low detection limit of 0.05 µM, outperforming CuPc alone. The study draws attention to the potential of borophene as a valuable additive for improving the performance of phthalocyanine-based sensors, providing fast response times, high stability, and strong selectivity for urea, even within complex matrices, making it an excellent candidate for clinical and environmental urea monitoring applications.
Recently, Ahmed et al. introduced an innovative BQD-based nanozyme platform designed for the sensitive detection of H2O2 and cardiac biomarkers such as cholesterol and choline.141 These hydrothermally synthesized BQDs demonstrated strong blue photoluminescence, high colloidal stability, and significantly enhanced POD-like activity when compared to pristine borophene. Structural analyses confirmed that they have a β-rhombohedral crystalline phase with a lattice spacing of 0.41 nm, which contributed to their excellent electroactive surface area. The primary detection mechanism is based on the nanozymatic oxidation of TMB in the presence of H2O2, facilitated by the BQDs. At neutral pH and under ambient conditions, the BQDs showed a strong affinity for both TMB and H2O2, as indicated by the Michaelis–Menten kinetics (Km for TMB is 0.12 mM; for H2O2 is 0.65 mM). Notably, the nanozymatic activity was significantly amplified by as much as 50-fold when an electrochemical field was applied, which accelerated both the redox cycle and color development. The BQDs promoted electron transfer and worked synergistically with the reactive oxygen species (ROS) such as ˙OH, ˙OOH, generated during the process, thereby enhancing TMB oxidation and reducing the detection time to just 30 s, in contrast to the traditional 30 min incubation required by conventional nanozyme assays. This advancement resulted in an exceptionally low LOD of 3.5 nM for H2O2. In the detection of cardiac biomarkers, cholesterol and choline were enzymatically oxidized using cholesterol oxidase and choline oxidase, respectively, producing stoichiometric quantities of H2O2, which was then rapidly quantified using the BQD/TMB system. The sensor achieved LoD values of 0.13 µM for cholesterol and 0.38 µM for choline, with a linear detection range of 10–100 µM for both analytes. Important selectivity assays demonstrated minimal interference from other common biomolecules and ions, such as uric acid, cysteine, ascorbic acid, and metal cations (Ca2+, Mg2+), ensuring reliable detection even in simulated blood serum. In addition to its sensitivity, the BQDs also exhibited benefits in stability, scalability, and biocompatibility, establishing them as a potent resource for next-generation label-free biosensing platforms in cardiovascular diagnostics.
Recently, Borah et al. developed a metal-free, biocompatible borophene nanosheet as a photonanozyme that enables colorimetric detection of DA by utilizing its unique photo-enhanced POD-like activity.34 Here, the POD-like activity of borophene nanosheet was significantly enhanced by exposure to light (440 nm), and it gradually decreased upon addition of DA. This inhibition-based sensing strategy enables selective and sensitive detection of DA, with a linear response in the range of 5–40 µM and a LOD of 6.6 µM. The photonanozyme sensor showed minimal interference from other biomolecules, including glucose, uric acid, cysteine, and ascorbic acid, and achieved high recovery rates (93–105%) in real samples such as fetal bovine serum (FBS) and brain tissue lysate, confirming its practical applicability. The study emphasizes the potential of light-driven, metal-free borophene nanozymes as effective and environmentally friendly platforms for the selective detection of neurotransmitters and other redox-active biomarkers (Fig. 8a).
 | ||
| Fig. 8 Schematic representation of the detection of (a) DA biomolecules using the photo-enhanced POD-like activity of borophene nanosheet. Reprinted with permission from ref. 34 Copyright 2024 Royal Society of Chemistry. (b) Glucose by combining the activities of GOx natural enzyme and POD-like activity of ox-BNSs. Reprinted with permission from ref. 86 Copyright 2023 American Chemical Society. | ||
Sun et al. reported a multilayered oxidized boron nanosheet (ox-BNSs), abundant in oxygen-containing functional groups, such as B–O and –OH, for the colorimetric detection of glucose.86 These nanosheets displayed strong blue fluorescence and impressive POD-like catalytic activity, effectively mimicking natural enzymes. The mechanism for glucose sensing involves a two-step enzymatic and nanozyme cascade. Initially, glucose is oxidized by glucose oxidase (GOx), which produces H2O2. Subsequently, the ox-BNSs catalyze the oxidation of TMB in the presence of the generated H2O2, leading to the formation of a distinct blue-colored product (OxTMB) with strong absorbance at 652 nm. The sensor showed a low detection limit of 2.5 µM, high selectivity for glucose compared to other sugars, and excellent performance in human serum, validating its potential for practical applications. Notably, the synthesis of the ox-BNSs did not involve organic solvents, surfactants, or templates, rendering this method green, cost-effective, and scalable. Their layered structure and high surface reactivity facilitate electron transfer and catalysis, making them an ideal nanozyme platform for effective glucose biosensing applications (Fig. 8b).
In 2024, Bu et al. developed a reusable, non-enzymatic colorimetric H2O2 sensor based on functionalized few-layer borophene (FFB) decorated with cerium-doped iron oxide nanoparticles (Ce-FeONPs).142 The Ce-FeONPs were grown in situ on FFB via a precipitation method, forming a hybrid nanozyme (FFB-Ce-FeONPs) that catalyzes the oxidation of TMB in the presence of H2O2, producing blue-colored OxTMB. This mimics natural peroxidase activity and enables sensitive colorimetric detection of H2O2, with a low detection limit of 0.03 mM and a linear range of 0.08–1 mM. Notably, the FFB component provides a reductive function, allowing it to convert OxTMB back to TMB, which permits multiple reuse cycles of the sensor. This reversible redox behavior, which is rare among nanozymes, enables the detection of H2O2 in 10 consecutive tests with minimal signal loss. Kinetic analysis showed a low Km value of 0.462 mM and a high Vmax, indicating strong substrate affinity and enhanced catalytic efficiency compared to undoped Ce-FeONPs. The hybrid's performance results from the synergistic interactions between the redox-active Fe2+/Fe3+ and Ce3+/Ce4+ pairs, along with the electron-deficient, high-surface-area borophene matrix, which enhances substrate adsorption and electron transfer. Overall, this system represents a robust, recyclable, and cost-effective nanozyme platform for H2O2 detection, with potential applications in biosensing, food safety, and environmental analysis.
Ahmed et al. developed a borophene-based nanozyme for electrochemically enhanced colorimetric detection of H2O2 by hydrothermally treating borophene with sodium thiocyanate (NaSCN) at 80 °C for one hour.143 This modification added radical-scavenging SCN− groups to the borophene surface and significantly improved its POD-like catalytic activity, allowing for effective oxidation of chromogenic substrates. The detection mechanism relies on the oxidation of TMB in the presence of H2O2, catalyzed by the SCN-modified borophene nanozyme. The presence of SCN groups helps concentrate ˙OH radicals produced from the decomposition of H2O2 near the borophene surface, enhancing electron transfer and speeding up TMB oxidation. By applying an electrochemical potential (ranging from −1 to +1 V) to the sensing surface through cyclic voltammetry, the oxidation rate of TMB is further increased, resulting in a deep-blue product that has a linear correlation with the concentration of H2O2. This electrochemical enhancement reduced the detection time to under 30 s and improved the LOD to 38 nM. The key attributes of borophene, including its high anisotropic conductivity, large surface area, and p-type semiconducting nature, were further enhanced by SCN modification, creating a p–n junction-like system that reduced charge transfer resistance and improved electrocatalytic efficiency.
Joshi et al. reported a novel fluorescent nanosensor based on BQDs for the sensitive detection of dehydroepiandrosterone (DHEA), a steroid hormone biomarker linked to pediatric adrenocortical carcinoma.55 The resulting BQDs displayed a strong blue fluorescence (excitation/emission at 370/428 nm), a high quantum yield of 24.12%, and an average particle size of 1.6 nm, which provided excellent water dispersibility and stability. The detection mechanism for DHEA is based on a fluorescence enhancement effect as illustrated in Fig. 9a. When DHEA is introduced, it binds electrostatically to the surface of the BQDs, resulting in their aggregation. This aggregation causes aggregation-induced emission (AIE), leading to a significant increase in fluorescence intensity. Evidence from dynamic light scattering (DLS), zeta potential measurements, Fourier-transform infrared (FT-IR) spectroscopy, and fluorescence lifetime analyses supported this interaction and aggregation mechanism. The system showed high specificity for DHEA compared to other biologically relevant molecules and achieved a low LOD of 18.72 nM, with a wide dynamic range of 0.1–125 µM. In addition, BQD-coated paper strips enabled visual detection under UV light, and the sensor exhibited outstanding performance in real samples such as urine and plasma, with recovery rates nearing 99%. The study highlights the potential of BQDs as a biocompatible, label-free, and highly sensitive platform for biomarker detection, paving the way for affordable, fluorescence-based biosensors suitable for clinical diagnostics and point-of-care applications.
 | ||
| Fig. 9 Schematic illustration of (a) DHEA sensing via fluorescence enhancement of BQDs. Reprinted with permission from ref. 55 Copyright 2023 Elsevier. (b) Detection of folic acid using fluorescence quenching of B-Arg QDs. Reprinted with permission from ref. 57 Copyright 2024 American Chemical Society. | ||
A recent study conducted by Barman et al. developed a highly sensitive and selective fluorescent nanosensor that utilizes arginine-functionalized BQDs (B-Arg QDs) to detect folic acid (FA), an important biomarker for DNA synthesis and neurological function as depicted in Fig. 9b.57 B-Arg QDs were synthesized through a two-step process, in which initially, borophene nanosheets were synthesized from bulk boron powder using an LPE technique in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ethanol–water mixture, carried out under prolonged ultrasonication. Following this, the nanosheets underwent functionalization with Arg via a hydrothermal reaction at 180 °C, resulting in small (1–5.5 nm), water-stable B-Arg QDs that exhibit pronounced blue fluorescence. The mechanism of detection of FA operates on a “turn-off” principle characterized by fluorescence quenching. Interaction with folic acid causes a significant decrease in the fluorescence intensity of the B-Arg QDs due to a combination of static quenching and IFE. This quenching occurs as FA binds electrostatically to the surface of the B-Arg QDs, forming ground-state complexes that hinder the excitation of the QDs. Time-resolved photoluminescence (TRPL) studies validated the static quenching mechanism, as the fluorescence lifetimes remained nearly constant upon the addition of FA. Additionally, the spectral overlap between FA absorption and B-Arg QDs excitation bands suggested that IFE also plays a role. This sensor demonstrated an impressive LOD of 14.91 nM in aqueous solutions, with comparable sensitivity in real samples, including human blood serum (LOD 14.22 nM), artificial urine (LOD 14.32 nM), and FA tablets (LOD 14.94 nM), all within a linear range of 0–6.66 µM. The B-Arg QDs exhibited exceptional selectivity, showing minimal response to common biomolecules and metal ions, while also achieving high recovery rates (92–105%) in complex matrices.
50 ethanol–water mixture, carried out under prolonged ultrasonication. Following this, the nanosheets underwent functionalization with Arg via a hydrothermal reaction at 180 °C, resulting in small (1–5.5 nm), water-stable B-Arg QDs that exhibit pronounced blue fluorescence. The mechanism of detection of FA operates on a “turn-off” principle characterized by fluorescence quenching. Interaction with folic acid causes a significant decrease in the fluorescence intensity of the B-Arg QDs due to a combination of static quenching and IFE. This quenching occurs as FA binds electrostatically to the surface of the B-Arg QDs, forming ground-state complexes that hinder the excitation of the QDs. Time-resolved photoluminescence (TRPL) studies validated the static quenching mechanism, as the fluorescence lifetimes remained nearly constant upon the addition of FA. Additionally, the spectral overlap between FA absorption and B-Arg QDs excitation bands suggested that IFE also plays a role. This sensor demonstrated an impressive LOD of 14.91 nM in aqueous solutions, with comparable sensitivity in real samples, including human blood serum (LOD 14.22 nM), artificial urine (LOD 14.32 nM), and FA tablets (LOD 14.94 nM), all within a linear range of 0–6.66 µM. The B-Arg QDs exhibited exceptional selectivity, showing minimal response to common biomolecules and metal ions, while also achieving high recovery rates (92–105%) in complex matrices.
A study by Kumar et al. introduced a new paper-based electrochemical biosensor that is highly sensitive for detecting serum amyloid A (SAA), an important protein biomarker related to infectious diseases such as swine flu.144 This biosensor incorporates hydrothermally synthesized borophene nanosheets embedded in a PEDOT:PSS matrix. These nanosheets were evenly integrated into Whatman paper through a straightforward dip-coating technique and underwent DMSO treatment to improve conductivity. The final electrode, called borophene/CP, exhibited excellent flexibility and could be bent (up to 40 cycles) while being environmentally friendly, allowing for smoke-free disposal with no toxic residue. The detection of SAA utilized a sandwich-type electrochemical immunoassay, where anti-SAA antibodies were immobilized onto the conducting paper modified by borophene. When the biosensor was exposed to the SAA antigen, specific interactions between antibody and antigen enabled electron transfer across the interface, leading to a measurable increase in the current signal. The resulting immunoelectrode demonstrated a detection limit of 0.49 ng mL−1, a broad linear detection range from 1 ng mL−1 to 60 µg mL−1, and was reported to exhibit excellent selectivity, stability up to 42 days, and reproducibility. The inclusion of borophene nanosheets significantly enhanced the electroactive surface area, charge transport, and biocompatibility of the sensing platform. In a recent study by Wekalao et al., the group introduced a hybrid terahertz biosensor designed to improve malaria detection by utilizing borophene-based nanocomposites.145 This innovative biosensor leverages the unique optoelectronic properties of borophene within a multilayer resonator structure. The design includes two rectangular resonators coated with borophene, symmetrically embedded in a square ring resonator coated with black phosphorus, alongside a circular resonator coated with graphene, all constructed on a silicon dioxide (SiO2) substrate. Borophene is produced using CVD technique and is transferred through wet transfer techniques, significantly boosting the sensor's performance due to its high electron mobility, strong plasmonic response, and excellent surface adsorption capabilities. The detection of malaria is based on shifts in resonance frequencies, namely 40 GHz and 30 GHz, that reflect changes in the refractive index of red blood cells infected with plasmodium. These frequency shifts are enhanced by borophene's capacity to localize and magnify terahertz field interactions within the resonator components. The sensor achieves a peak sensitivity of 700 GHz per refractive index unit (RIU) and enhances a quality factor greater than 10.5, making it highly effective for early-stage, label-free malaria diagnosis.
Again, humidity levels critically influence human health, as elevated moisture can exacerbate respiratory conditions and encourage fungal growth in damp environments, underscoring the urgent need for effective sensing technologies. The developed device enables real-time monitoring of environmental humidity, which is essential for maintaining optimal indoor air quality in hospitals, preventing skin and respiratory illnesses, and advancing smart building automation systems. So, in this context, Hou et al. experimentally demonstrated a flexible and ultrasensitive borophene–MoS2 heterostructured humidity sensor (Fig. 10a), addressing the growing need for wearable sensing devices in health monitoring and human–machine interface (HMI) applications.146 Although the p–n junction of borophene–MoS2 had previously been predicted to exhibit superior humidity sensitivity, this study was the first to experimentally validate these claims. The fabricated sensor exhibited an exceptional sensitivity of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500% at 97% relative humidity (RH), a value over 90 and 70 times higher than pristine borophene and MoS2, respectively. This makes it the most sensitive chemiresistive sensor among all 2D material-based counterparts reported to date. Furthermore, the device demonstrated rapid response and recovery times of 2.5 s and 3.1 s, respectively (Fig. 10e), excellent mechanical flexibility (Fig. 10h and i), long-term operational stability, and strong selectivity for water vapor over other organic vapors. The performance enhancement is attributed to efficient charge transfer and the Grotthuss proton-hopping mechanism within the heterostructure, facilitating fast adsorption–desorption of water molecules. When exposed to increasing RH levels (0–97%), the current and sensitivity increased proportionally, as shown in the real-time measurements (Fig. 10b and c). The device showed excellent repeatability with three consecutive 97% RH exposure/recovery cycles (Fig. 10d), and reversible performance even under repeated bending, indicating suitability for wearable electronics (Fig. 10f and g). To demonstrate practical utility in health diagnostics, the sensor was used for real-time breath monitoring. It could distinguish between resting and post-exercise breathing rates by tracking rapid fluctuations in current during inhalation and exhalation cycles (Fig. 10j and k), highlighting its potential for low-cost, non-invasive preliminary diagnosis of respiratory conditions. Beyond contact-based applications, the sensor was integrated into a non-contact sensing platform. By detecting humidity emitted from human fingertips, the sensor's current output was inversely correlated with fingertip distance, from 71.9% sensitivity at 2 mm to 9.5% at 10 mm (Fig. 10l). Additionally, the sensor was incorporated into a smart LED circuit. As a fingertip approached, the induced current triggered the LED to switch on (ON state), while retraction turned it off (OFF state), visually demonstrating its non-contact sensing capability (Fig. 10m and n). These findings firmly establish borophene–MoS2 heterostructures as powerful candidates for next-generation wearable electronics, respiratory diagnostics, and HMI systems.145 In another work, Hou et al. developed structurally stable borophene-based heterostructures and demonstrated their effectiveness in high-performance humidity sensing. Remarkably, at 85% relative humidity (RH), the borophene–graphene sensor exhibited a sensitivity nearly 700 times higher than that of pristine graphene, ranking it among the most sensitive 2D material-based chemiresistive humidity sensors reported to date. The enhanced performance is attributed to a well-established charge transfer mechanism between adsorbed water molecules and the borophene–granule interface, which modulates the sensor's electrical resistance. The response characteristics were evaluated under various RH environments (0%, 11%, 33%, 43%, 67%, 75%, and 85%), with a strong linear correlation observed between sensor sensitivity and humidity concentration. Repeatable exposure/recovery cycles under RH pulses (0% to 85%) confirmed the device's excellent reversibility and repeatability. Furthermore, the time-dependent response and recovery behavior were assessed at 85% RH, revealing rapid switching dynamics. Stability tests over extended periods demonstrated consistent performance under varying humidity levels (43%, 67%, 75%, and 85% RH), indicating strong long-term reliability. Importantly, the borophene–graphene heterostructure sensor maintained its sensitivity and structural integrity even after repeated mechanical bending, confirming its suitability for flexible and wearable electronics. The sensor, fabricated on a PET substrate displayed negligible performance degradation under applied bending strain highlighting its potential for integration into next-generation portable and stretchable sensing devices. This work not only advances the understanding of interfacial charge transfer interactions in boron-based 2D heterostructures but also underscores the viability of borophene–graphene composites for real-time, high-performance environmental sensing applications.32 Another one developed by Wu et al. was an ultrasensitive and durable borophene-based humidity sensor tailored for advanced human-centric applications. Their approach addresses key limitations of conventional fabrication techniques, such as drop-coating, which often suffer from poor control over sensing layer thickness, limited water resistance, and significant device-to-device variability.147 Hou et al. (2023) presented a comprehensive review on borophene, covering its evolution, synthesis strategies, and diverse applications in fields such as energy storage, energy conversion, energy harvesting, sensors, and information storage.37 They particularly emphasized borophene's unique physicochemical properties, including its high surface area, anisotropic conductivity, and strong adsorption capacity, which enable high sensitivity and rapid response in gas, humidity, and pressure sensors. In addition, their work reported the development of a high-performance wearable pressure sensor based on hydrogenated borophene nanosheets integrated into tissue paper substrates.33 The sensor exhibited excellent electromechanical properties, including high sensitivity (2.16 kPa−1), a wide detection range (0–120 kPa), low detection limit (10 Pa), rapid response, and durability over 1000 cycles. Due to borophene's semiconducting nature and mechanical flexibility, the device enabled real-time monitoring of physiological signals such as pulse, respiration, and human motion, and was further applied in e-skin arrays and human–machine interfaces, highlighting its potential for wearable healthcare monitoring and soft robotics. Together, these findings highlight borophene's strong potential for advancing next-generation sensing technologies and wearable electronics.33
500% at 97% relative humidity (RH), a value over 90 and 70 times higher than pristine borophene and MoS2, respectively. This makes it the most sensitive chemiresistive sensor among all 2D material-based counterparts reported to date. Furthermore, the device demonstrated rapid response and recovery times of 2.5 s and 3.1 s, respectively (Fig. 10e), excellent mechanical flexibility (Fig. 10h and i), long-term operational stability, and strong selectivity for water vapor over other organic vapors. The performance enhancement is attributed to efficient charge transfer and the Grotthuss proton-hopping mechanism within the heterostructure, facilitating fast adsorption–desorption of water molecules. When exposed to increasing RH levels (0–97%), the current and sensitivity increased proportionally, as shown in the real-time measurements (Fig. 10b and c). The device showed excellent repeatability with three consecutive 97% RH exposure/recovery cycles (Fig. 10d), and reversible performance even under repeated bending, indicating suitability for wearable electronics (Fig. 10f and g). To demonstrate practical utility in health diagnostics, the sensor was used for real-time breath monitoring. It could distinguish between resting and post-exercise breathing rates by tracking rapid fluctuations in current during inhalation and exhalation cycles (Fig. 10j and k), highlighting its potential for low-cost, non-invasive preliminary diagnosis of respiratory conditions. Beyond contact-based applications, the sensor was integrated into a non-contact sensing platform. By detecting humidity emitted from human fingertips, the sensor's current output was inversely correlated with fingertip distance, from 71.9% sensitivity at 2 mm to 9.5% at 10 mm (Fig. 10l). Additionally, the sensor was incorporated into a smart LED circuit. As a fingertip approached, the induced current triggered the LED to switch on (ON state), while retraction turned it off (OFF state), visually demonstrating its non-contact sensing capability (Fig. 10m and n). These findings firmly establish borophene–MoS2 heterostructures as powerful candidates for next-generation wearable electronics, respiratory diagnostics, and HMI systems.145 In another work, Hou et al. developed structurally stable borophene-based heterostructures and demonstrated their effectiveness in high-performance humidity sensing. Remarkably, at 85% relative humidity (RH), the borophene–graphene sensor exhibited a sensitivity nearly 700 times higher than that of pristine graphene, ranking it among the most sensitive 2D material-based chemiresistive humidity sensors reported to date. The enhanced performance is attributed to a well-established charge transfer mechanism between adsorbed water molecules and the borophene–granule interface, which modulates the sensor's electrical resistance. The response characteristics were evaluated under various RH environments (0%, 11%, 33%, 43%, 67%, 75%, and 85%), with a strong linear correlation observed between sensor sensitivity and humidity concentration. Repeatable exposure/recovery cycles under RH pulses (0% to 85%) confirmed the device's excellent reversibility and repeatability. Furthermore, the time-dependent response and recovery behavior were assessed at 85% RH, revealing rapid switching dynamics. Stability tests over extended periods demonstrated consistent performance under varying humidity levels (43%, 67%, 75%, and 85% RH), indicating strong long-term reliability. Importantly, the borophene–graphene heterostructure sensor maintained its sensitivity and structural integrity even after repeated mechanical bending, confirming its suitability for flexible and wearable electronics. The sensor, fabricated on a PET substrate displayed negligible performance degradation under applied bending strain highlighting its potential for integration into next-generation portable and stretchable sensing devices. This work not only advances the understanding of interfacial charge transfer interactions in boron-based 2D heterostructures but also underscores the viability of borophene–graphene composites for real-time, high-performance environmental sensing applications.32 Another one developed by Wu et al. was an ultrasensitive and durable borophene-based humidity sensor tailored for advanced human-centric applications. Their approach addresses key limitations of conventional fabrication techniques, such as drop-coating, which often suffer from poor control over sensing layer thickness, limited water resistance, and significant device-to-device variability.147 Hou et al. (2023) presented a comprehensive review on borophene, covering its evolution, synthesis strategies, and diverse applications in fields such as energy storage, energy conversion, energy harvesting, sensors, and information storage.37 They particularly emphasized borophene's unique physicochemical properties, including its high surface area, anisotropic conductivity, and strong adsorption capacity, which enable high sensitivity and rapid response in gas, humidity, and pressure sensors. In addition, their work reported the development of a high-performance wearable pressure sensor based on hydrogenated borophene nanosheets integrated into tissue paper substrates.33 The sensor exhibited excellent electromechanical properties, including high sensitivity (2.16 kPa−1), a wide detection range (0–120 kPa), low detection limit (10 Pa), rapid response, and durability over 1000 cycles. Due to borophene's semiconducting nature and mechanical flexibility, the device enabled real-time monitoring of physiological signals such as pulse, respiration, and human motion, and was further applied in e-skin arrays and human–machine interfaces, highlighting its potential for wearable healthcare monitoring and soft robotics. Together, these findings highlight borophene's strong potential for advancing next-generation sensing technologies and wearable electronics.33
 | ||
| Fig. 10 Fabrication and characterization of the borophene–MoS2 heterostructured humidity sensor: (a) schematic of the heterostructured sensor; (b) real-time response under RH switching; (c) sensitivity (ΔI/I0) at different RH levels; (d) cycling stability at 97% RH; (e) response/recovery at 97% RH; (f) long-term stability at 43–97% RH; (g) hysteresis curve at various RH values; (h) schematic of bent sensor on PET; (i) response under bent and flat states at 67% RH. Multifunctional applications of the borophene–MoS2 humidity sensor: (j) schematic of health diagnostics via humidity sensing; (k) response to slow/fast respiration; (l) response at varying fingertip distances; (m) current response with/without fingertip at 15 V; (n) photographs of fingertip non-contact switch with LED ON/OFF. Reprinted with permission from ref. 146 Copyright 2021 Royal Society of Chemistry. | ||
6. Challenges and future perspectives
Borophene has rapidly established itself as one of the most intriguing members of the 2D material family, due to its exceptional carrier mobility, mechanical compliance, metallic conductivity, plasmonic tunability, and superior catalytic activity. However, despite these advantages, its practical deployment remains significantly restricted by several critical challenges. Most theoretical studies are conducted under idealised conditions, where borophene is considered perfectly stable and structurally pure, thereby predicting extraordinary mechanical, thermal, electronic and optical properties.148,149 In contrast, experimental realisations often encounter difficulties related to stability, large-scale synthesis, reproducibility, and environmental sensitivity, which collectively hinder its immediate transition to real-world applications. This gap between theoretical predictions and experimental feasibility continues to be the major bottleneck in translating the exceptional potential of borophene into practical technologies such as energy storage, catalysis, plasmonics, and sensing. However, experimental studies frequently encounter the aforementioned challenges, which significantly hinder the realisation of these envisioned applications. The synthesis of borophene since its discovery in 2015 by Mannix et al.15 has underscored its fundamental limitations. Early growth relied on epitaxial deposition under ultra-high vacuum (UHV) on metallic substrates such as Ag(111) and Au(111), which validated its existence but simultaneously restricted scalability and hindered substrate transferability. Moreover, borophene displayed high sensitivity to ambient conditions; Zhang et al.150 reported its rapid oxidative degradation and structural collapse upon air exposure, as well as significant out-of-plane buckling on weakly interacting substrates, which compromised mechanical integrity. Unlike more structurally robust 2D analogues such as graphene or hexagonal boron nitride, borophene does not exhibit a fixed intrinsic crystal lattice but instead demonstrates polymorphism dictated by the growth substrate. This polymorphic nature, while scientifically intriguing, further complicates its reliable synthesis and device integration.To circumvent these limitations, liquid-phase strategies have been developed. Sonication-assisted exfoliation in organic solvents such as DMF and isopropanol produced borophene nanosheets,151 while subsequent approaches enabled freestanding sheets in aqueous media through the reduction of borophene oxide.47 More recently, aqueous-phase synthesis routes have been reported, offering a step towards environmentally friendly and potentially scalable production.34 Despite these advances, key issues remain unresolved. Borophene continues to exhibit high susceptibility to oxidation, difficulty in achieving controlled sheet thickness and morphology, and an absence of reproducible, cost-effective, and industrially viable synthesis methods. Furthermore, the lack of defect-free, large-area films significantly restricts its applicability in electronic and optoelectronic devices.
Addressing these challenges requires a multipronged approach. First, stability enhancement strategies are crucial. Surface functionalisation, heterostructure design, and encapsulation using chemically inert 2D materials (such as graphene, MoS2, or h-BN) could offer effective protection against oxidative degradation.26,99,152 Similarly, integration into polymer or hybrid nanocomposites provides opportunities for structural reinforcement, passivation, and multifunctionality. Second, advances in synthesis and processing will be indispensable. Developing substrate-independent, large-area growth techniques, refining LPE for uniform nanosheet production, and devising scalable transfer methodologies are essential steps towards industrial relevance. Third, defect engineering and controlled doping strategies may allow for tunability of electronic and catalytic properties, while simultaneously mitigating instability issues. Emerging approaches that combine in situ characterisation with machine learning or AI-guided simulations also have potential to optimise growth conditions and predict stable polymorphs. From an application standpoint, real progress will depend on integrating borophene into device-level platforms. In energy storage, designing borophene-based heterostructures with graphene or transition-metal dichalcogenides could improve electrode stability and charge transport. In catalysis and sensing, functionalised borophene may offer high sensitivity and selectivity, provided long-term reliability is achieved. Moreover, the biomedical domain, where borophene's catalytic and electronic properties could be leveraged for biosensing or therapeutic applications, remains largely unexplored but highly promising, contingent on the development of biocompatible and stable formulations. The pathway from fundamental discovery to practical application of borophene is at a critical juncture. Overcoming challenges in synthesis, stability, reproducibility, and scalability will be pivotal. At the same time, future perspectives lie in hybridisation, defect and interface engineering, encapsulation, and AI-assisted materials design. By bridging these scientific and technological gaps, borophene may ultimately transition from an exciting laboratory material into a cornerstone for next-generation devices across electronics, optoelectronics, catalysis, energy systems, and biomedical technologies.
This review provides an overview of borophene synthesis in various forms, sheets, quantum dots, and nanocomposites, via techniques like MBE, CVD, and LPE. It highlights borophene's key properties, such as metallic conductivity, anisotropic mechanical strength, high surface reactivity, and unique plasmonic and enzyme-mimetic behaviors, making it a promising material for next-gen sensing technologies. This review covers detection methods like fluorescence, colorimetric, and electrochemical techniques, demonstrating borophene's potential for ultra-sensitive monitoring of biomolecules, antibiotics, heavy metals, and pollutants. Borophene's versatility positions it as a key material for advancements in materials science, nanotechnology, and bioanalytics.
Conflicts of interest
There are no conflicts to declare.Data availability
This review article does not involve the generation of new datasets. All data and materials discussed are derived from previously published studies, which are cited in the manuscript.Acknowledgements
The authors gratefully acknowledge the Director, CSIR-NEIST, Jorhat, India, for providing permission and necessary support to carry out this work (MS No. CSIR-NEIST/PUB/2025/161). M. R. D. sincerely acknowledges the Council of Scientific and Industrial Research (CSIR), India, for the financial support through Research Grant (Project No. OLP-2504A).References
- G. Wu, Y. Hu, Y. Liu, J. Zhao, X. Chen, V. Whoehling, C. Plesse, G. T. M. Nguyen, F. Vidal and W. Chen, Nat. Commun., 2015, 6, 7258 CrossRef CAS PubMed.
- C. Li, J. Kang, J. Xie, Y. Wang, L. Zhou, H. Hu, X. Li, J. He, B. Wang and H. Zhang, J. Mater. Chem. C, 2020, 8, 16318–16325 RSC.
- R. Irshad, K. Tahir, B. Li, Z. Sher, J. Ali and S. Nazir, J. Ind. Eng. Chem., 2018, 64, 60–69 Search PubMed.
- X. Li, S. Chen, Q. Liu, Y. Luo and X. Sun, Chem. Commun., 2021, 57, 8039–8042 Search PubMed.
- S. K. Sahoo and K. Wei, Adv. Mater. Interfaces, 2019, 6, 1900752 Search PubMed.
- S. Ghosal, A. Bandyopadhyay, S. Chowdhury and D. Jana, Rep. Prog. Phys., 2023, 86, 096502 Search PubMed.
- Y. Han, M. Kolmer, J. W. Evans and L.-L. Wang, J. Phys.:Condens. Matter, 2025, 37, 373001 CrossRef CAS PubMed.
- P. Ranjan, J. M. Lee, P. Kumar and A. Vinu, Adv. Mater., 2020, 32, 2000531 CrossRef CAS.
- B. Peng, H. Zhang, H. Shao, Z. Ning, Y. Xu, G. Ni, H. Lu, D. W. Zhang and H. Zhu, Mater. Res. Lett., 2017, 5, 399–407 Search PubMed.
- D. Gogoi, C. Hazarika, G. Neog, P. Mridha, H. K. Bora, M. R. Das, S. Szunerits and R. Boukherroub, ACS Appl. Mater. Interfaces, 2024, 16, 14645–14660 CrossRef CAS PubMed.
- Y. Duo, Z. Xie, L. Wang, N. Mahmood Abbasi, T. Yang, Z. Li, G. Hu and H. Zhang, Coord. Chem. Rev., 2021, 427, 213549 Search PubMed.
- M. Fujimori, T. Nakata, T. Nakayama, E. Nishibori, K. Kimura, M. Takata and M. Sakata, Phys. Rev. Lett., 1999, 82, 4452 CrossRef CAS.
- X. Sun, X. Liu, J. Yin, J. Yu, Y. Li, Y. Hang, X. Zhou, M. Yu, J. Li, G. Tai and W. Guo, Adv. Funct. Mater., 2017, 27, 1603300 CrossRef.
- T. Jian, X. Chen, S. D. Li, A. I. Boldyrev, J. Li and L. S. Wang, Chem. Soc. Rev., 2019, 48, 3550–3591 RSC.
- A. J. Mannix, X. F. Zhou, B. Kiraly, J. D. Wood, D. Alducin, B. D. Myers, X. Liu, B. L. Fisher, U. Santiago, J. R. Guest, M. J. Yacaman, A. Ponce, A. R. Oganov, M. C. Hersam and N. P. Guisinger, Science, 2015, 350, 1513–1516 CrossRef CAS PubMed.
- M. Gao, Q. Z. Li, X. W. Yan and J. Wang, Phys. Rev. B, 2017, 95, 024505 CrossRef.
- B. Feng, J. Zhang, Q. Zhong, W. Li, S. Li, H. Li, P. Cheng, S. Meng, L. Chen and K. Wu, Nat. Chem., 2016, 8, 563–568 Search PubMed.
- A. P. Sergeeva, I. A. Popov, Z. A. Piazza, W. L. Li, C. Romanescu, L. S. Wang and A. I. Boldyrev, Acc. Chem. Res., 2014, 47, 1349–1358 Search PubMed.
- A. Banks, J. Chem. Educ., 1990, 67, 14 Search PubMed.
- Z. A. Piazza, H. S. Hu, W. L. Li, Y. F. Zhao, J. Li and L. S. Wang, Nat. Commun., 2014, 5, 1–6 Search PubMed.
- Z. Zhang, E. S. Penev and B. I. Yakobson, Chem. Soc. Rev., 2017, 46, 6746–6763 RSC.
- H. Shu, F. Li, P. Liang and X. Chen, Nanoscale, 2016, 8, 16284–16291 Search PubMed.
- A. Mazaheri, M. Javadi and Y. Abdi, ACS Appl. Mater. Interfaces, 2021, 13, 8844–8850 CrossRef CAS PubMed.
- G. H. Gupta, S. Kadakia, D. Agiwal, T. Keshari and S. Kumar, Mater. Adv., 2024, 5, 1803–1816 RSC.
- N. Sarma, H. Das and P. Saikia, ACS Sens., 2025, 10, 622–641 CrossRef CAS PubMed.
- P. Pal and M. Nandi, Dalton Trans., 2024, 54, 38–58 Search PubMed.
- M. Ou, X. Wang, L. Yu, C. Liu, W. Tao, X. Ji and L. Mei, Adv. Sci., 2021, 8, 2001801 Search PubMed.
- P. G. C. A. Macilon, T. F. Santos, E. V. Santos, B. R. Carvalho and J. H. O. Nascimento, Emergent Mater., 2025, 1–42 Search PubMed.
- R. K. Mishra, J. Sarkar, K. Verma, I. Chianella, S. Goel and H. Y. Nezhad, Nano Mater. Sci., 2024, 2589–9651 Search PubMed.
- E. Halvacı, D. İkballı, A. Özengül, B. Sevimli, A. Tumeh, Z. Güzel, M. S. Erdoğan, G. Kaya, Ö. Uysal and F. Şen, Int. J. Boron Sci. Nanotechnol., 2024, 86–102 Search PubMed.
- S. Tatiya, M. Pandey and S. Bhattacharya, J. Micromanuf., 2020, 3, 159–173 CrossRef.
- C. Hou, G. Tai, B. Liu, Z. Wu and Y. Yin, Nano Res., 2020, 14, 2337–2344 CrossRef.
- C. Hou, G. Tai, Y. Liu, R. Liu, X. Liang, Z. Wu and Z. Wu, Nano Energy, 2022, 97, 107189 CrossRef CAS.
- P. Borah, D. J. Baruah, P. Mridha, R. Baishya, H. K. Bora and M. R. Das, Chem. Commun., 2024, 20, 2417–2420 RSC.
- A. Rahman, M. T. Rahman, M. A. Chowdhury, S. Bin Ekram, M. M. K. Uddin, M. R. Islam and L. Dong, Sens. Actuators, A, 2023, 359, 114468 CrossRef CAS.
- P. Kumar, G. Singh, R. Bahadur, Z. Li, X. Zhang, C. I. Sathish, M. R. Benzigar, T. Kim Anh Tran, N. T. Padmanabhan, S. Radhakrishnan, J. C. Janardhanan, C. Ann Biji, A. Jini Mathews, H. John, E. Tavakkoli, R. Murugavel, S. Roy, P. M. Ajayan and A. Vinu, Prog. Mater. Sci., 2024, 146, 101331 Search PubMed.
- C. Hou, G. Tai, Y. Liu, Z. Wu, X. Liang and X. Liu, Nano Res. Energy, 2023, 2, 9120051 CrossRef.
- U. Anwar, J. Ren, L. Malfatti and P. Innocenzi, J. Photochem. Photobiol., A, 2025, 116399 CrossRef CAS.
- I. Muhammad, U. Younis, H. Xie, A. A. Khan, A. Khaliq, A. Samad, U. Schwingenschlögl and Q. Sun, Chem. Mater., 2021, 33, 2976–2983 CrossRef CAS.
- G. Tai, M. Xu, C. Hou, R. Liu, X. Liang and Z. Wu, ACS Appl. Mater. Interfaces, 2021, 13, 60987–60994 CrossRef CAS.
- N. Taşaltın, S. Güllülü and S. Karakuş, Inorg. Chem. Commun., 2022, 136, 109150 CrossRef.
- C. Sharma, M. K. Gupta, S. Badatya, A. K. Srivastava and N. Sathish, Commun. Mater., 2023, 4, 54 Search PubMed.
- Q. Zhong, L. Kong, J. Gou, W. Li, S. Sheng, S. Yang, P. Cheng, H. Li, K. Wu and L. Chen, Phys. Rev. Mater., 2017, 1, 021001 CrossRef.
- U. Rajput, M. F. Akhtar, V. Walve, I. Mulani, G. Vyas, S. Bhowmick, P. Kumar and A. Deshpande, J. Phys. Chem. C, 2024, 128, 4104–4110 CrossRef CAS.
- H. Ekici, Uluslar. Dogu Anadolu Fen Muhendislik Ve Tasar. Derg., 2023, 126–142 CrossRef.
- W. Li, K. Wu and L. Chen, Prog. Surf. Sci., 2023, 100704 CrossRef CAS.
- P. Ranjan, T. K. Sahu, R. Bhushan, S. S. Yamijala, D. J. Late, P. Kumar and A. Vinu, Adv. Mater., 2019, 31, 1900353 Search PubMed.
- S. Yadav, M. A. Sadique, N. Sathish and R. Khan, New J. Chem., 2025, 49, 7910–7917 RSC.
- B. Kiraly, X. Liu, L. Wang, Z. Zhang, A. J. Mannix, B. L. Fisher, B. I. Yakobson, M. C. Hersam and N. P. Guisinger, ACS Nano, 2019, 13, 3816–3822 CrossRef CAS PubMed.
- M. A. Chowdhury, M. M. K. Uddin, M. B. A. Shuvho, M. Rana and N. Hossain, Appl. Surf. Sci. Adv., 2022, 11, 100308 CrossRef.
- Z. Guo, H. Lin, H. Zhang, Y. Tian, Y. Chen, J. Chen, S. Deng and F. Liu, Cryst. Growth Des., 2023, 23, 4506–4513 CrossRef CAS.
- A. Zhao, Y. Han, Y. Che, Q. Liu, X. Wang, Q. Li, J. Sun, Z. Lei, X. He and Z.-H. Liu, J. Mater. Chem. A, 2021, 9, 24036–24043 Search PubMed.
- H. Wang, D. An, M. Wang, L. Sun, Y. Li, H. Li, N. Li, S. Hu and Y. B. He, Mater. Adv., 2021, 2, 3269–3273 RSC.
- L. Wang, S. M. Xu, S. Guan, X. Qu, G. I. N. Waterhouse, S. He and S. Zhou, J. Mater. Chem. B, 2020, 8, 9881–9887 RSC.
- D. J. Joshi, N. I. Malek, T. Jung Park and S. K. Kailasa, J. Mol. Liq., 2023, 385, 122294 CrossRef CAS.
- S. Aswal, K. Ghosh and P. K. Giri, ACS Appl. Nano Mater., 2024, 7, 11634–11644 CrossRef CAS.
- N. Barman, K. J. Goswami, N. Sultana and N. S. Sarma, ACS Appl. Nano Mater., 2024, 7, 17351–17363 CrossRef CAS.
- H. J. Yashwanth, K. Hareesh, S. R. Rondiya, R. J. Choudhary and S. D. Dhole, Appl. Surf. Sci., 2024, 646, 158910 CrossRef CAS.
- T. E. Somesh, D. T. Tran, S. Jena, Y. Bai, S. Prabhakaran, D. H. Kim, N. H. Kim and J. H. Lee, Chem. Eng. J., 2024, 481, 148266 Search PubMed.
- Y. Lin, M. Yu, X. Li, W. Gao, L. Wang, X. Zhao, M. Zhou, X. Yao, M. He and X. Zhang, J. Mater. Chem. C, 2021, 9, 15877–15885 RSC.
- J. Yu, Z. Fan, F. Yu, H. Zhao, P. Yao, S. Chen, H. Yang, C. Zhu and J. Xu, Chem. Eng. J., 2023, 145592 CrossRef CAS.
- M. Alikhani, S. Hajibaba, M. Moayedi and Y. Abdi, J. Storage, 2024, 99, 113301 CrossRef CAS.
- A. Dube, S. J. Malode, M. Alruwaili, A. N. Alodhayb, T. Maiyalagan and N. P. Shetti, ChemElectroChem, 2025, 12, 202400670 CrossRef.
- A. G. Rojek, G. Leniec and E. Mijowska, Appl. Surf. Sci., 2025, 684, 161851 CrossRef CAS.
- T. A. Türkmen, N. Taşaltın, C. Taşaltın, G. Baytemir and S. Karakuş, Inorg. Chem. Commun., 2022, 139, 109329 CrossRef.
- S. Chen, S. Huang, H. Wu, W. Pan, S. Wei, C. Peng, I. Ni, B. T. Murti, M. Tsai, C. Wu and P. Yang, Adv. Sci., 2022, 9, 2201507 CrossRef CAS.
- D. K. Chinnalagu, B. Murugesan, M. Arumugam, K. Chinniah, S. Ganesan, Y. Cai and S. Mahalingam, J. Energy Storage, 2023, 74, 109328 CrossRef.
- C. Taşaltın, Inorg. Chem. Commun., 2021, 133, 108973 Search PubMed.
- W. Mabhulusa, K. E. Sekhosana and X. Fuku, ChemElectroChem, 2024, 11, e202400138 CrossRef CAS.
- H. Yang, F. Bao, S. Chen, S. Liu, H. Huang, L. Wang, H. Liu, J. Yu, C. Zhu and J. Xu, ACS Appl. Mater. Interfaces, 2024, 16, 56072 Search PubMed.
- Z. Wu, C. Shifan, Z. Wu, Y. Liu, W. Shao, X. Liang, C. Hou and G. Tai, Nano Res., 2023, 17, 3053–3060 CrossRef.
- X. Yan, S. Wang, Y. Sun, Y. Liu, Y. Wang and G. Yang, J. Phys. Chem. Lett., 2023, 9698–9704 CrossRef CAS PubMed.
- S. Mohanty, D. Panda, A. Dash, S. Sovan Kumar, R. R. Padhi, S. Guhathakurata and S. Mallik, J. Electron. Mater., 2023, 52, 4434–4454 CrossRef CAS.
- Y. Abdi, A. Eskandari, Z. Alavi, A. Khamsavi, M. Etminan, M. Zahedi, M. Taleb and N. Talebi, Adv. Mater. Interfaces, 2024, 12, 2400894 Search PubMed.
- J. Zhang, Z. Zhang, X. Song, H. Zhang and J. Yang, Nanomaterials, 2021, 11, 1165 Search PubMed.
- J. Nong, F. Feng, C. Min, X. Yuan and M. Somekh, Opt. Lett., 2021, 46, 725–728 CrossRef PubMed.
- R. Ahluwalia, N. Ng, D. J. Srolovitz, M. Q. Le, B. Mortazavi and T. Rabczuk, Nanotechnology, 2016, 27, 445709 CrossRef PubMed.
- B. Mortazavi, O. Rahaman, A. Dianat and T. Rabczuk, Phys. Chem. Chem. Phys., 2016, 18, 27405–27413 Search PubMed.
- Y. P. Zhou and J. W. Jiang, Sci. Rep., 2017, 7, 45516 Search PubMed.
- Z. Zhang, Y. Yang, E. S. Penev and B. I. Yakobson, Adv. Funct. Mater., 2017, 27, 1605059 CrossRef.
- X. Wu, J. Dai, Y. Zhao, Z. Zhuo, J. Yang and X. C. Zeng, ACS Nano, 2012, 6, 7443–7453 Search PubMed.
- G. Liu, H. Wang, Y. Gao, J. Zhou and H. Wang, Phys. Chem. Chem. Phys., 2017, 19, 2843–2849 RSC.
- H. Zhou, Y. Cai, G. Zhang and Y.-W. Zhang, npj 2D Mater. Appl., 2017, 1, 14 CrossRef.
- H. Sun, Q. Li and X. G. Wan, Phys. Chem. Chem. Phys., 2016, 18, 14927–14932 RSC.
- T. Tsafack and B. I. Yakobson, Phys. Rev. B, 2016, 93, 165434 Search PubMed.
- R. Sun, J. Chen, W. Zhang, Y. Huang, J. Zheng and Y. Chi, Anal. Chem., 2023, 95, 11475–11482 CrossRef CAS PubMed.
- P. Borah, D. J. Baruah, P. Mridha, R. Duarah, R. Baishya and M. R. Das, ACS Omega, 2025, 29, 32269–32281 Search PubMed.
- L. He, B. Dong, Y. Liu and W. Lin, Chem. Soc. Rev., 2016, 45, 6449–6461 Search PubMed.
- D. Escudero, Acc. Chem. Res., 2016, 49, 1816–1824 CrossRef CAS PubMed.
- D. Udhayakumari, J. Fluoresc., 2024, 35, 3799–3828 Search PubMed.
- P. Naksen, S. Boonruang, N. Yuenyong, H. L. Lee, P. Ramachandran, W. Anutrasakda, M. Amatatongchai, S. Pencharee and P. Jarujamrus, Talanta, 2022, 242, 123305 Search PubMed.
- Y. Hong, J. W. Y. Lam and B. Z. Tang, Chem. Commun., 2009, 4332 RSC.
- X. Ma, R. Sun, J. Cheng, J. Liu, F. Gou, H. Xiang and X. Zhou, J. Chem. Educ., 2015, 93, 345–350 Search PubMed.
- A. C. Sedgwick, L. Wu, H. H. Han, S. D. Bull, X. P. He, T. D. James, J. L. Sessler, B. Z. Tang, H. Tian and J. Yoon, Chem. Soc. Rev., 2018, 47, 8842–8880 RSC.
- V. Chaudhary, S. Sonu, B. A. Taha, P. Raizada, S. Rustagi, S. Chahal, P. Singh, A. Khosla and V. H. Nguyen, J. Mater. Sci. Technol., 2025, 218, 236–262 CrossRef CAS.
- S. Yadav, M. A. Sadique, A. Kaushik, P. Ranjan, R. Khan and A. K. Srivastava, J. Mater. Chem. B, 2022, 10, 1146–1175 RSC.
- M. A. Sadique, S. Yadav, P. Ranjan and R. Khan, Engineering Materials, Springer Nature Singapore, Singapore, 2024, pp. 175–199 Search PubMed.
- Z. Wu, X. Liang, Y. Liu, M. Xu, R. Zhu and G. Tai, Angew. Chem., Int. Ed., 2024, 64, e202416041 CrossRef PubMed.
- S. S. Emadian, S. Varagnolo, A. Kumar, P. Kumar, P. Ranjan, V. Pyeshkova, N. Vangapally, N. P. Power, S. Pitchaimuthu, A. Chroneos, S. Gopalan, P. Sonar and S. Krishnamurthy, Energy Environ. Mater., 2025, 8, e12881 CrossRef CAS.
- R. Kaimal, M. C. Maridevaru, A. Dube, J. J. Wu, A. Sambandam and M. Ashokkumar, Ind. Eng. Chem. Res., 2023, 62, 15418–15427 CrossRef CAS.
- S. N. Nangare, Z. G. Khan, A. G. Patil and P. O. Patil, J. Mol. Struct., 2022, 133387 CrossRef CAS.
- S. Mondal, S. Priya, S. Lata, S. Kumar, H. Ranjan, S. Rustagi, M. A. Sadique, S. Yadav, J. Bora, S. Malik and R. Khan, Engineering Materials, Springer Nature Singapore, Singapore, 2024, pp. 45–65 Search PubMed.
- Y. V. Kaneti, D. P. Benu, X. Xu, B. Yuliarto, Y. Yamauchi and D. Golberg, Chem. Rev., 2022, 122, 1000–1051 CrossRef CAS PubMed.
- S. Ahmed, A. Ansari, S. Kashif Ali, B. R. Patil, F. Riyaz, A. Khan and P. Ranjan, Anal. Sens., 2024, 4, e202400034 CAS.
- G. Baytemir, Appl. Phys. A: Mater. Sci. Process., 2023, 129, 85 CrossRef CAS.
- S. Güngör, C. Taşaltın, İ. Gürol, G. Baytemir, S. Karakuş and N. Taşaltın, Appl. Phys. A: Mater. Sci. Process., 2022, 128, 89 CrossRef.
- S. Paul, S. Nandi, M. Das, A. Bora, M. T. Hossain, S. Ghosh and P. K. Giri, Nanoscale, 2023, 15, 12612–12625 Search PubMed.
- R. Das, H. Sugimoto, M. Fujii and P. K. Giri, ACS Appl. Mater. Interfaces, 2020, 12, 4755–4768 Search PubMed.
- T. I. Al Muhimeed, Chin. J. Phys., 2025, 95, 752–764 Search PubMed.
- S. M. Hosseini, A. Imanpour, M. Rezavand, A. M. Arjmandi Tash, S. Antonini, S. J. Rezvani and Y. Abdi, ACS Mater. Lett., 2024, 6, 933–942 CrossRef CAS.
- T. L. Guidotti, Int. J. Toxicol., 2010, 29, 569–581 CrossRef CAS PubMed.
- D. Girardin, F. Berger, A. Chambaudet and R. Planade, Sens. Actuators, B, 1997, 43, 147–153 CrossRef CAS.
- J. Casanova-Chafer and C. Bittencourt, ACS Appl. Electron. Mater., 2025, 7, 2305–2312 CrossRef CAS.
- Z. Zhao, X. Liu, Y. Liu, Z. Wu, K. Xiong and G. Tai, Angew. Chem., Int. Ed., 2025, 64(18), e202501550 CrossRef CAS PubMed.
- G. K. Ramesha and S. Sampath, Sens. Actuators, B, 2011, 160, 306–311 CrossRef CAS.
- G. K. Raghu, D. Antony and P. Malingappa, Int. J. Smart Nano Mater., 2014, 5, 17–32 CrossRef.
- P. Devi, C. Sharma, P. Kumar, M. Kumar, B. K. S. Bansod, M. K. Nayak and M. L. Singla, J. Hazard. Mater., 2017, 322, 85–94 Search PubMed.
- W. W. Li, F. Y. Kong, J. Y. Wang, Z. D. Chen, H. L. Fang and W. Wang, Electrochim. Acta, 2015, 157, 183–190 CrossRef CAS.
- F. Yuan, H. Zhao, X. Wang and X. Quan, Anal. Lett., 2016, 50, 544–553 CrossRef.
- C. Hazarika, D. Dutta, E. Roy, R. Duarah, R. Konwar and M. R. Das, Chem. – Asian J., 2025, 20(21), e00820 CrossRef CAS PubMed.
- C. Li, L. Zhu, W. Yang, X. He, S. Zhao, X. Zhang, W. Tang, J. Wang, T. Yue and Z. Li, J. Agric. Food Chem., 2019, 67, 1277–1283 CrossRef CAS PubMed.
- X. Zhu, Z. Zhang, Z. Xue, C. Huang, Y. Shan, C. Liu, X. Qin, W. Yang, X. Chen and T. Wang, Anal. Chem., 2017, 89, 12054–12058 CrossRef CAS PubMed.
- S. Li, Y. Li, J. Cao, J. Zhu, L. Fan and X. Li, Anal. Chem., 2014, 86, 10201–10207 CrossRef CAS PubMed.
- S. Liu, X. Yang and W. Feng, Appl. Opt., 2019, 58, 2152 CrossRef CAS PubMed.
- Z. Song, Z. Wei, B. Wang, Z. Luo, S. Xu, W. Zhang, H. Yu, M. Li, Z. Huang, J. Zang, F. Yi and H. Liu, Chem. Mater., 2016, 28, 1205–1212 CrossRef CAS.
- A. Bibi, Y. R. M. Rubio, L. Xian-Lun, N. Sathishkumar, C. Y. Chen, K. S. Santiago, H. T. Chen, Y. F. Lin and J.-M. Yeh, Synth. Met., 2021, 282, 116934 Search PubMed.
- K. Urs, T. Carey, L. Tsetseris, S. Liu, K. Synnatschke, Z. Sofer, J. N. Coleman, J. C. Wenger, S. Biswas and J. D. Holmes, ACS Appl. Nano Mater., 2025, 8, 2141–2152 Search PubMed.
- G. Darabdhara, B. Sharma, M. R. Das, R. Boukherroub and S. Szunerits, Sens. Actuators, B, 2017, 238, 842–851 CrossRef CAS.
- J. Huang, Y. He, J. Jin, Y. Li, Z. Dong and R. Li, Electrochim. Acta, 2014, 136, 41–46 CrossRef CAS.
- Y. Huang, M. Zhao, S. Han, Z. Lai, J. Yang, C. Tan, Q. Ma, Q. Lu, J. Chen, X. Zhang, Z. Zhang, B. Li, B. Chen, Y. Zong and H. Zhang, Adv. Mater., 2017, 29, 1700102 CrossRef PubMed.
- M. N. Ivanova, E. D. Grayfer, E. E. Plotnikova, L. S. Kibis, G. Darabdhara, P. K. Boruah, M. R. Das and V. E. Fedorov, ACS Appl. Mater. Interfaces, 2019, 11, 22102–22112 CrossRef CAS PubMed.
- P. K. Boruah, P. Borthakur, G. Neog, B. Le Ouay, N. U. Afzal, P. Manna and M. R. Das, ACS Appl. Nano Mater., 2023, 6, 1667–1677 CrossRef CAS.
- S. R. Ahmed, A. G. Cardoso, H. V. Cobas, P. Das, A. Chen, S. Srinivasan and A. R. Rajabzadeh, ACS Appl. Nano Mater., 2023, 6, 8434–8443 CrossRef CAS.
- D. J. Baruah, A. Thakur, E. Roy, K. Roy, S. Basak, D. Neog, H. K. Bora, R. Konwar, V. Chaturvedi, M. V. Shelke and M. R. Das, ACS Appl. Mater. Interfaces, 2023, 15, 47902–47920 CrossRef CAS PubMed.
- P. Das, P. K. Boruah, P. Sarmah, R. Dutta, R. Boukherroub and M. R. Das, ChemistrySelect, 2022, 7, e202203415 CrossRef CAS.
- J. Chen, Y. Shu, H. Li, Q. Xu and X. Hu, Talanta, 2018, 189, 254–261 CrossRef CAS.
- W. Zhang, X. Li, T. Cui, S. Li, Y. Qian, Y. Yue, W. Zhong, B. Xu and W. Yue, Microchim. Acta, 2021, 188, 174 Search PubMed.
- B. Ghrib, Results Chem., 2025, 102629 CrossRef CAS.
- C. Taşaltın, T. A. Türkmen, N. Taşaltın and S. Karakuş, J. Mater. Sci., 2021, 32, 10750–10760 Search PubMed.
- G. Baytemir, İ. Gürol, S. Karakuş, C. Taşaltın and N. Taşaltın, J. Mater. Sci., 2022, 33, 16586–16596 CAS.
- S. R. Ahmed, M. Sherazee, P. Das, J. Dondapati, S. Srinivasan and A. R. Rajabzadeh, ACS Appl. Nano Mater., 2023, 6, 19939–19946 CrossRef CAS.
- Y. Bu and B. S. Kim, Biosens. Bioelectron., 2024, 260, 116448 CrossRef CAS PubMed.
- S. R. Ahmed, M. Sherazee, P. Das, M. Shalauddin, S. Akhter, W. J. Basirun, S. Srinivasan and A. R. Rajabzadeh, Biosens. Bioelectron., 2024, 246, 115857 CrossRef CAS PubMed.
- Y. Kumar, S. Shankar, R. Chandra and S. Kumar, ACS Appl. Nano Mater., 2024, 7, 19925–19935 CrossRef CAS.
- J. Wekalao, S. M. Njoroge and O. Elamri, Braz. J. Phys., 2025, 55, 126 CrossRef CAS.
- C. Hou, G. Tai, Y. Liu, Z. Wu, Z. Wu and X. Liang, J. Mater. Chem. A, 2021, 9, 13100–13108 RSC.
- Z. Wu, X. Liang, Z. Zhao, Q. Wu, X. Liu, Y. Liu, M. Xu, C. Hou and G. Tai, Chem. Eng. J., 2024, 500, 156881 CrossRef CAS.
- Y. Yao and G. Liu, Mol. Phys., 2021, 1966114 CrossRef.
- M. Ishaq, R. A. Shehzad, M. Yaseen, S. Iqbal, K. Ayub and J. Iqbal, J. Mol. Model., 2021, 27, 188 CrossRef CAS PubMed.
- Z. Zhang, Y. Yang, G. Gao and B. I. Yakobson, Angew. Chem., 2015, 127, 13214–13218 Search PubMed.
- H. Li, L. Jing, W. Liu, J. Lin, R. Y. Tay, S. H. Tsang and E. H. T. Teo, ACS Nano, 2018, 12, 1262–1272 CrossRef CAS PubMed.
- S. Faramarzi and T. Movlarooy, ACS Appl. Mater. Interfaces, 2024, 16, 25966–25976 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |