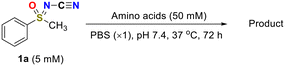Open Access Article
Open Access ArticleN-Cyano sulfoximine-mediated thiazole ligation with N-terminal cysteine under mild aqueous conditions
Eunsil
Kim
ab,
Ji Young
Hyun
ab,
Ji Ho
Song
ab,
Hwan Jung
Lim
 *ab and
Seong Jun
Park
*ab and
Seong Jun
Park
 *ab
*ab
aDepartment of Drug Discovery, Korea Research Institute of Chemical Technology (KRICT), 141 Gajeong-ro, Yuseong-gu, Daejeon 34114, Republic of Korea. E-mail: sjunpark@krict.re.kr; Fax: +82 42 860 7160; Tel: +82 42 860 7175
bPharmaceutical Chemistry, University of Science & Technology, Daejeon 34113, Republic of Korea
First published on 16th December 2025
Abstract
An N-terminal cysteine-selective click reaction employing N-cyano sulfoximines enables rapid thiazole formation under mild conditions. These three-dimensional, hydrophilic scaffolds offer high selectivity, tunable reactivity, and improved drug-like properties. The platform holds promise for bioorthogonal conjugation and ligand design in drug discovery applications.
Efficient and selective chemical ligation methods have greatly advanced chemical biology and materials science.1–3 Among these, click reactions such as the copper-catalyzed azide–alkyne cycloaddition (CuAAC)4,5 and sulfur(VI) fluoride exchange (SuFEx) are powerful tools for modular synthesis. In particular, SuFEx using sulfuryl fluoride (SO2F2) affords stable linkages, though the resulting connectivities are typically limited to planar geometries.6,7
The shift from two-dimensional (2D) to three-dimensional (3D) molecular design is emerging as a promising strategy in medicinal chemistry to enhance aqueous solubility without compromising permeability or efficacy. Adoption of non-planar molecular architectures provides an effective strategy to address solubility challenges in drug development (Fig. 1).8–12
Thionyl tetrafluoride (SOF4) enables the formation of tetrahedral iminosulfur oxydifluorides with two reactive S–F handles, offering precise spatial control and polyvalency for constructing 3D architectures in biomolecular engineering and materials science.13–15 However, SuFEx chemistry remains limited by reagent availability, substrate scope, and scalability.11,16
To establish a practical biomimetic approach, we selected readily accessible sulfoximines that enable the construction of three-dimensional molecular architectures.17 Bioisosteric replacement of sulfone or sulfonamide groups with sulfoximine moieties significantly enhances aqueous solubility and has facilitated the clinical progression of a lead compound.18–20
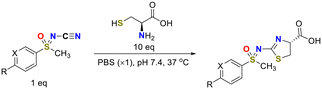
N-Cyano sulfoximines21–32 are particularly attractive due to their ease of synthesis (Fig. 2a), high aqueous solubility, three-dimensional molecular features, and the presence of a reactive cyano group amenable to click-type conjugation. Their potential as bioorthogonal platforms is further supported by the well-established reactivity of cyano groups—particularly cyanopyridines—with aminothiols.33–42
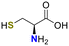
Importantly, N-terminal cysteine, a naturally encoded amino acid, was deliberately selected as the reaction partner43,44 to ensure that the product more closely resembles a naturally occurring structure rather than a purely synthetic one (Fig. 2b). This biomimetic strategy provides a distinct advantage by aligning chemical reactivity with biological relevance and compatibility.
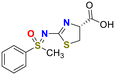
To evaluate the reactivity, we first examined the model reaction of phenylmethyl sulfoximine 1a with cysteine. The desired click reaction proceeded smoothly to afford the corresponding thiazole 2a when the reaction was carried out in PBS (pH 7.4) at 37 °C in the presence of 10 equivalents of cysteine (entry 1, Table 1).45
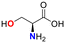
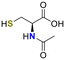
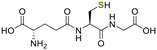
No reaction occurred when N-cyano sulfoximine 1a was treated with serine, N-acetylcysteine, or glutathione (GSH), indicating high selectivity towards cysteine (entry 2 to 4, Table 1).
To modulate the reactivity of the nitrile group in the electrophilic N-cyano sulfoximine, various electron-withdrawing or electron-donating groups were introduced onto the phenyl ring. Additionally, heteroatoms were incorporated into the aromatic system to further tune the electron density.
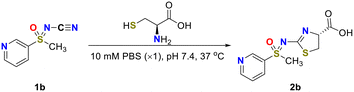
The reactions of 1a and 1b were completed after 72 h and 25 h, respectively (entry 1 and 2, Table 2). For N-cyano sulfoximine 1b, which contains a heteroatom within the aromatic ring, a markedly shorter reaction time was observed (entry 2, Table 2).
For para-substituted N-cyano sulfoximines 1c and 1d, poor aqueous solubility prevented dissolution under standard buffer conditions; thus, 2.5% DMSO was added to achieve complete dissolution prior to the reaction. N-Cyano sulfoximine 1d, bearing an electron-withdrawing group, afforded the desired thiazole product 2d with a higher conversion ratio compared to 1c, which possesses an electron-donating group (entry 3 and 4, Table 2).
For comparison, the corresponding N-cyano sulfonamide 1ba was also subjected to the reaction. As shown in the X-ray crystal structures in Fig. 3, the sulfonamide 1ba adopts the 2D conformation, whereas the sulfoximine 1b exhibits the 3D structure. The sulfoximine 1b also demonstrated approximately 1.7-fold higher solubility. Furthermore, in the case of the sulfonamide 1ba, the desired thiazole product was not obtained upon reaction with cysteine.
 | ||
| Fig. 3 Comparison between N-cyano sulfonamide 1ba and sulfoximine 1b.46 | ||
Based on previous findings indicating high reactivity and good solubility under buffer conditions, N-cyano sulfoximine 1b was selected to evaluate the effect of buffer pH on the reaction completion time for thiazole 2b formation.
Under acidic conditions (pH 4), no reaction was observed, whereas under mildly acidic conditions (pH 6), the reaction reached completion after 72 hours. Under neutral conditions (pH 7.4), the reaction of N-cyano sulfoximine 1b was complete after 24 h. Under basic conditions (pH 8.0–9.0), the reaction was found to reach completion within 7 hours. Interestingly, under strongly basic conditions (pH 10), the reaction was found to reach completion within just 1 hour (Fig. 4).
To shorten the reaction completion time at neutral pH, the effect of varying the stoichiometric ratio between N-cyano sulfoximine 1b and cysteine was investigated. As illustrated in Table 3, an excess of the electrophile N-cyano sulfoximine 1b resulted in a faster reaction completion.47
Collectively, the results indicate that the use of 10 equivalents of N-cyano sulfoximine 1b and 1 equivalent of cysteine at neutral pH leads to complete conversion within 3 hours, representing the optimised condition.
A comparison of the predicted physicochemical properties between the reported azidophile48 and the N-cysteinophile 1b developed in this study revealed that compound 1b exhibits significantly more hydrophilic characteristics.49 The compound 1b was predicted to exhibit greater hydrophilicity than TMTHSI bearing a sulfoximine moiety, as reported by Liskamp and co-workers (Fig. 5).50
 | ||
| Fig. 5 Prediction of the physicochemical properties of azidophiles and N-cysteinophile 1b.49 | ||
To evaluate the practical applicability of the developed reaction, the transformation between N-cyano sulfoximine-derived methionine 1e and N-terminal cysteine was carried out. Gratifyingly, the reaction reached completion within 1.5 h under mild aqueous conditions (Fig. 6).51 This result is significant as it demonstrates the ability to link the two naturally occurring sulfur-containing amino acids, methionine and cysteine.
To assess site selectivity under the optimized conditions, peptides bearing either N-terminal or internal cysteine residues were examined. Peptide 1 (CGKSRF) bearing an N-terminal cysteine readily underwent ligation with N-cyano sulfoximine 1b, affording the expected product after 28 h, as confirmed by LC-MS. In contrast, Peptide 2 (KSCGRF), containing an internal cysteine, showed no reaction even after 72 h. Notably, efficient ligation was also observed with the longer Peptide 3 (CGCGESGKSTIVKQMK), which features an N-terminal cysteine, completing the reaction within 3 h (Fig. 7).53
In summary, we have developed a novel N-cyano sulfoximine-based click reaction that proceeds selectively with N-terminal cysteine to form thiazole linkages under mild, aqueous conditions. Systematic evaluation of structure–reactivity relationships revealed that electronic and solubility properties of the N-cyano sulfoximines significantly influence reaction kinetics. Among the tested analogs, compound 1b, featuring a heteroaryl moiety, exhibited enhanced reactivity and superior aqueous solubility. These findings highlight the potential of N-cyano sulfoximines as versatile and biocompatible electrophilic warheads for bioorthogonal conjugation. The practical utility of this method was further demonstrated by its successful application to a methionine-derived N-cyano sulfoximine substrate 1e. Notably, ligation occurred selectively at N-terminal cysteines, as shown by efficient conjugation of peptides 1 and 3, while no reaction was observed with the internal cysteine of peptide 2, highlighting the potential of this approach for bioconjugation and chemical biology applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5md00948k.CCDC 2481304 and 2481305 contain the supplementary crystallographic data for this paper.54a,b
Acknowledgements
This research was supported by the Korea Drug Development Fund funded by Ministry of Science and ICT, Ministry of Trade, Industry and Energy, and Ministry of Health and Welfare (RS-2025-02223190, Republic of Korea) and by KRICT (KK2431-20 and KK2531-20).Notes and references
- R. E. Bird, S. A. Lemmel, X. Yu and Q. A. Zhou, Bioconjugate Chem., 2021, 32, 2457–2479 CrossRef PubMed.
- K. M. Hartung and E. M. Sletten, Chem, 2023, 9, 2095–2109 Search PubMed.
- F. M. Zielke and F. P. J. T. Rutjes, Top. Curr. Chem., 2023, 381, 1–30 CrossRef.
- J. E. Hein and V. V. Fokin, Chem. Soc. Rev., 2010, 39, 1302–1315 RSC.
- E. Haldón, M. C. Nicasio and P. J. Pérez, Org. Biomol. Chem., 2015, 13, 9528–9550 RSC.
- J. Dong, L. Krasnova, M. G. Finn and K. B. Sharpless, Angew. Chem., Int. Ed., 2014, 53, 9430–9448 CrossRef CAS.
- T. A. Fattah, A. Saeed and F. Albericio, J. Fluorine Chem., 2018, 213, 87–112 CrossRef.
- F. Lovering, J. Bikker and C. Humblet, J. Med. Chem., 2009, 52, 6752–6756 CrossRef CAS.
- M. Ishikawa and Y. Hashimoto, J. Med. Chem., 2011, 54, 1539–1554 CrossRef CAS.
- M. A. Walker, Expert Opin. Drug Discovery, 2014, 9, 1–13 Search PubMed.
- Z. He, W. Yang, F. Yang, J. Zhang and L. Ma, Eur. J. Med. Chem., 2024, 279, 116842 CrossRef CAS.
- B. Das, A. T. K. Baidya, A. T. Mathew, A. K. Yadav and R. Kumar, Bioorg. Med. Chem., 2022, 56, 116614 CrossRef CAS PubMed.
- S. Li, P. Wu, J. E. Moses and K. B. Sharpless, Angew. Chem., Int. Ed., 2017, 56, 2903–2908 CrossRef CAS.
- B. Gao, S. Li, P. Wu, J. E. Moses and K. B. Sharpless, Angew. Chem., Int. Ed., 2018, 57, 1939–1943 CrossRef CAS PubMed.
- D. Zeng, W.-P. Deng and X. Jiang, Natl. Sci. Rev., 2023, 10, 1–19 Search PubMed.
- A. S. Barrow, C. J. Smedley, Q. Zheng, S. Li, J. Dong and J. E. Moses, Chem. Soc. Rev., 2019, 48, 4731–4758 RSC.
- M. Andresini, A. Tota, L. Degennaro, J. A. Bull and R. Luisi, Chem. – Eur. J., 2021, 27, 17293–17321 CrossRef CAS.
- U. Lücking, Org. Chem. Front., 2019, 6, 1319–1324 RSC.
- P. Mäder and L. Kattner, J. Med. Chem., 2020, 63, 14243–14275 CrossRef PubMed.
- Y. Han, K. Xing, J. Zhang, T. Tong, Y. Shi, H. Cao, H. Yu, Y. Zhang, D. Liu and L. Zhao, Eur. J. Med. Chem., 2021, 209, 112885 CrossRef CAS PubMed.
- D. Swern, I. Ikeda and G. F. Whitfield, Tetrahedron Lett., 1972, 13, 2635–2638 CrossRef.
- T. E. Varkey, G. F. Whitfield and D. Swern, J. Org. Chem., 1974, 39, 3365–3372 CrossRef CAS.
- J. E. G. Kemp, D. Ellis and M. D. Closier, Tetrahedron Lett., 1979, 20, 3781–3784 CrossRef.
- O. G. Mancheño and C. Bolm, Org. Lett., 2007, 9, 2951–2954 CrossRef PubMed.
- O. G. Mancheño, O. Bistri and C. Bolm, Org. Lett., 2007, 9, 3809–3811 CrossRef PubMed.
- A. Pandey and C. Bolm, Synthesis, 2010, 17, 2922–2925 Search PubMed.
- C. M. M. Hendriks, P. Lamers, J. Engel and C. Bolm, Adv. Synth. Catal., 2013, 355, 3363–3368 CrossRef CAS.
- C. A. Dannenberg, L. Fritze, F. Krauskopf and C. Bolm, Org. Biomol. Chem., 2017, 15, 1086–1090 RSC.
- S. M. Kim, O.-Y. Kang, H. J. Lim and S. J. Park, ACS Omega, 2020, 5, 10191–10199 CrossRef CAS PubMed.
- M. Klein and S. R. Waldvogel, Angew. Chem., 2021, 60, 23197–23201 CrossRef CAS PubMed.
- N. Amri and T. Wirth, J. Org. Chem., 2021, 86, 15961–15972 CrossRef CAS.
- Y. J. Seo, E. Kim, I. S. Oh, J. Y. Hyun, J. H. Song, H. J. Lim and S. J. Park, RSC Adv., 2023, 13, 24445–24449 RSC.
- R. M. Oballa, J.-F. Truchon, C. I. Bayly, N. Chauret, S. Day, S. Crane and C. Berthelette, Bioorg. Med. Chem. Lett., 2007, 17, 998–1002 CrossRef CAS.
- V. Ehmke, J. E. Q. Quinsaat, P. Rivera-Fuentes, C. Heindl, C. Freymond, M. Rottmann, R. Brun, T. Schirmeister and F. Diederich, Org. Biomol. Chem., 2012, 10, 5764–5768 RSC.
- O. V. Maltsev, V. Walter, M. J. Brandl and L. Hintermann, Synthesis, 2013, 45, 2763–2767 CrossRef.
- C. Nitsche, H. Onagi, J.-P. Quek, G. Otting, D. Luo and T. Huber, Org. Lett., 2019, 21, 4709–4712 CrossRef.
- Z. Chen, M. Chen, Y. Cheng, T. Kowada, J. Xie, X. Zheng and J. Rao, Angew. Chem., Int. Ed., 2020, 59, 3272–3279 Search PubMed.
- F.-J. Chen and J. Gao, Chem. – Eur. J., 2022, 28, e202201843 Search PubMed.
- M. Proj, N. Strašek, S. Pajk, D. Knez and I. Sosic, Bioconjugate Chem., 2023, 34, 1271–1281 CrossRef PubMed.
- C. Nitsche, Synlett, 2024, 35, 1067–1071 Search PubMed.
- V. J. Thombare, Y. Wu, K. Pamulapati, M. Han, J. Tailhades, M. J. Cryle, K. D. Roberts, T. Velkov, J. Li and N. A. Patil, Chem. – Eur. J., 2024, 30, e202401674 CrossRef PubMed.
- T. Yano, T. Yamada, H. Isida, N. Ohashi and T. Itoh, RSC Adv., 2024, 14, 6542–6547 RSC.
- A. Istrate, M. B. Geeson, C. D. Navo, B. B. Sousa, M. C. Marques, R. J. Taylor, T. Journeaux, S. R. Oehler, M. R. Mortensen, M. J. Deery, A. D. Bond, F. Corzana, G. Jiménez-Osés and G. J. L. Bernardes, J. Am. Chem. Soc., 2022, 144, 10396–10406 CrossRef PubMed.
- R. Padanha, R. A. N. Cavadas, P. Merino, J. P. M. António and P. M. P. Gois, Org. Lett., 2023, 25, 5476–5480 CrossRef PubMed.
- The reaction was considered complete after 72 hours, as N-cyano sulfoximine 1a was no longer detected by LC/MS analysis.
- CCDC 2481304 (1ba) and CCDC 2481305 (1b) contain the supplementary crystallographic data for this paper. These data are provided free of by The Cambridge Crystallographic Centre.
- The completion time was determined by 1H NMR spectroscopy (please see the SI).
- M. F. Debets, J. C. M. van Hest and F. P. J. T. Rutjes, Org. Biomol. Chem., 2013, 11, 6439–6455 RSC.
- A. Daina, O. Michielin and V. Zoete, Sci. Rep., 2017, 7, 42717 CrossRef PubMed.
- J. Weterings, C. J. F. Rijcken, H. Veldhuis, T. Meulemans, D. Hadavi, M. Timmers, M. Honing, H. Ippel and R. M. J. Liskamp, Chem. Sci., 2020, 11, 9011–9016 RSC.
- The synthetic procedure52 for 1e, together with full reaction details, is provided in the SI.
- L. Buglioni, V. Bizet and C. Bolm, Adv. Synth. Catal., 2014, 356, 2209–2213 CrossRef.
- We would also like to point out that longer peptides may adopt an α-helical conformation, which could potentially reduce the reactivity of internal cysteines.
- (a) CCDC 2481304: Experimental Crystal Structure Determination, 2025, DOI:10.5517/fiz.icsd.cc2p9023; (b) CCDC 2481305: Experimental Crystal Structure Determination, 2025, DOI:10.5517/fiz.icsd.cc2p9034.
| This journal is © The Royal Society of Chemistry 2026 |