DOI:
10.1039/D6MA00278A
(Paper)
Mater. Adv., 2026, Advance Article
Influence of Sm2O3 substitution on the mechanical properties, crystallization kinetics, radiation shielding and Judd–Ofelt analysis of borosilicate glasses
Received
27th February 2026
, Accepted 6th April 2026
First published on 7th April 2026
Abstract
Magnesium-containing borosilicate glass samples reinforced with varying concentrations of Sm2O3 (0–1.5 mol%) have been produced via the conventional melt quenching method in order to investigate the mechanical properties, activation energy of crystallization and transition, thermal stability, fragility index, radiation shielding (MAC, HVL, Zeff, EBAF) and Judd–Ofelt parameters. The X-ray diffraction pattern confirmed the glassy nature of the prepared samples owing to the lack of distinctive X-ray diffraction peaks. The elastic moduli Em and Rm increased from 86.26 to 107.95 GPa and 62.11 to 95.55 GPa, respectively, with an increase in Sm2O3 concentration. Crystallization kinetics, especially activation energies (Eg, Ex and Ec), were analyzed via the Kissinger and Augis–Bennett methods and were found to increase with increasing Sm2O3 concentration. The mass attenuation coefficient (MAC) and the effective atomic number (Zeff) increased, whereas the half-value layer (HVL) decreased as Sm2O3 content increased in the glass networks. The MS-1 sample has higher relative photon attenuation efficiency over a broad energy range than the other samples. The Judd–Ofelt parameters follow the same trend (Ω4 > Ω2 > Ω6) for all the prepared glass samples. To examine the suitability of Sm3+-substituted borosilicate glass for photonic applications, transition probability (Ar), branching ratio (βr), radiative lifetime (τR), and peak emission cross-section (σp) are obtained for each transition band. The examined glasses are promising candidates for laser applications as compared to the other Sm2O3 doped glass systems reported in the literature.
1 Introduction
Borosilicate glasses (BSGs) have shown great potential for use in laboratories due to their superior mechanical strength, chemical durability, and exceptional thermal shock resistance as compared to the other glasses, such as borate and soda-lime glasses.1,2 In the modern era, borosilicate glasses are also explored for solid-state lasers, radiation dosimetry, and radiation shielding applications.3–5 BSGs have been recognized as good shielding materials owing to their high density and better ionizing radiation absorbability.6
Presently, radiation shielding is a primary requirement in radiotherapy, industrial imaging, and aerospace engineering.7 It is environmentally important, uses radiological facilities and serving as a primary means of safeguarding personnel from exposure risks.8–10 To optimize gamma radiation shielding, various glasses have been synthesized using high atomic number (Z) compounds to increase their effective atomic number (Zeff) as well as density.11,12 The light transmission, absorbance, refractive index, and the optical band gap must be rigorously assessed to ensure the suitability of glasses for shielding applications.9,11,13 For instance, B2O3 enhances optical performance, while MgO improves the mechanical and thermal stability. Furthermore, the addition of PbO2, BaO, TiO2, GeO2, TeO2, etc. significantly increases the radiation attenuation abilities of the glasses.14,15
Rare earth elements play a vital role in advancing both the gamma radiation shielding and the optical performance of glass materials. Furthermore, the network of glass structure changes with the incorporation of rare earth elements, leading to an increase in density and refractive index, which is essential for superior attenuation efficiency and optical properties.16 Rare-earth doping significantly enhances luminescence efficiency, enabling advancements in high-performance lighting, display technologies, and phosphor-based devices. Samarium-doped borosilicate glasses have gained significant attention from the scientific community due to their unique physicochemical properties.17 Additionally, they are highly suitable for full-color displays and high-density optical memory devices owing to the inherent valence instability of the samarium ion. The interaction between radiation and matter is primarily characterized by the photoelectric effect (0.01–0.5 MeV), photon–electron scattering (>0.5 MeV), and electron–positron pair production (1.02 MeV).18,19
Glasses have emerged as an important class of materials for radiation shielding applications.20 Extensive research has been done on tellurite, borate, phosphate, germanate, and silicate oxides as the primary source of the glass-forming systems.21 Borate glasses offer a low melting point, high resistance to devitrification, superior density, and effective γ-ray attenuation, particularly in lead-borate compositions, which possess a diverse array of structural units.22 Concrete is a composite material composed of cement and aggregates such as sand, gravel/crushed stone, and optional additives including supplementary cementitious materials, fibers, and chemical admixtures. In the presence of water, these constituents form a durable material widely used in foundations, bridges, pavements, and buildings. Due to its cost-effectiveness, durability, high mechanical strength, and superior heat resistance, concrete has been widely investigated as a radiation shielding material.23,24 However, structural degradation, cracking, corrosion, high weight, and environmental concerns related to CO2 emissions during cement production restrict the use of borate glasses. Ceramics are composed of silica, quartz, and feldspar and exhibit excellent thermal stability, corrosion resistance, hardness, and electrical insulation. These properties make ceramics promising candidates for radiation shielding applications.23,25 Nevertheless, their inherent brittleness and susceptibility to cracking remain an important limitation.
Furthermore, crystallization kinetics are essential to understand the thermal stability of borosilicate glasses.26 A differential scanning calorimeter (DSC) is employed to determine the characteristic temperatures of the prepared glasses.27 The non-isothermal crystallization performance can be effectively characterized using the Augis–Bennett and Kissinger models to estimate the activation energies for crystallization and the glass transition of the prepared glasses.28,29 Additionally, DSC data are used to assess the Avrami constant (Ce), fragility index (If), and Lasocka parameters (X and Y), which provide critical insights into the local structure of the glasses. These factors govern the crystallization kinetics, and optical and mechanical properties of the glasses. Notably, there is always a scarcity of research that systematically correlates crystallization behavior with the mechanical performance of Sm2O3-containing borosilicate glasses. UV-Vis-NIR absorption spectra characterize the optical band gap and localized energy states as a result of Sm3+ incorporation. The spectral features correspond to electronic transitions within the Sm3+ 4f levels, enabling the investigation of the local bonding, structural modifications, and potential radiation-induced defect levels.30
The present study determine the effect of Sm2O3 substitution on the optical and mechanical properties, crystallization kinetics, and radiation shielding behavior of 50SiO2 + 40B2O3 + (10 − x)MgO + (x)Sm2O3 glass. The mechanical properties are determined using the Makishima–Mackenzie, Rocherulle and bond compression models and correlated to the crystallization kinetics. The unique electronegativity and field strength of MgO and Sm2O3 in the glass matrix are responsible for altering the network structure. Therefore, the aim of the present work is to examine the effect of Sm2O3 on the non-isothermal crystallization kinetics, mechanical properties, and radiation shielding capability of borosilicate glasses.
Problem statement: borosilicate glasses are widely used in photonic, laser, and radiation shielding applications due to their excellent thermal and chemical stability. However, the performance of these glasses strongly depends on the incorporation of the rare-earth ions, which can modify the glass network structure and influence their optical and mechanical properties. Among the rare-earth ions, Sm3+ has attracted considerable attention because of its characteristic orange-red emissions and potential applications in solid-state lasers and photonic devices. Despite several studies on Sm2O3-doped glass systems, a comprehensive study on the effect of Sm2O3 substitution on the structural rigidity, crystallization behavior, radiation shielding capability, and spectroscopic properties of borosilicate glasses remains limited. Particularly, the influence of Sm2O3 on the elastic properties, crystallization kinetics, and Judd–Ofelt parameters of the SiO2 + B2O3 + MgO glass system has not been systematically investigated yet.
Therefore, it is essential to explore the role of Sm2O3 substitution in modifying the glass network and the consequent impact on mechanical stability, crystallization resistance, and optical performance. For this purpose, Sm2O3-containing borosilicate glass compositions with improved structural and spectroscopic properties for advanced photonic and laser applications are fabricated using the melt and quench method.
Research gap: rare-earth-doped borosilicate glasses have gained significant interest due to their excellent chemical durability, thermal stability, and ability to host optically active ions for applications in photonics and lasers. Among different rare-earth ions, Sm3+ is a vital candidate due to its characteristic emission transitions and suitability for solid-state lasers and optical devices. Although several researchers have reported Sm2O3-doped phosphate, borate, and silicate glass systems for many applications, a systematic investigation of Sm2O3 substitution in MgO-modified borosilicate glass networks remains scarce. In particular, the combined effect of Sm2O3 on mechanical properties, crystallization kinetics, radiation shielding behavior, and Judd–Ofelt spectroscopic parameters has not been comprehensively explored in the glass systems.
2 Materials and methods
2.1 Glass synthesis
The glass compositions are prepared using the melt and quenching method according to the general formula| | |
50SiO2 + 40B2O3 + (10 − x)MgO + (x)Sm2O3
| (1) |
where x = 0, 0.5, 1, and 1.5 mol%. The selected compositions are given in Table 1. For convenience, the prepared glass samples are nominated as MS-0, MS-0.5, MS-1, and MS-1.5. Sm2O3, MgO, B2O3 and SiO2 were weighed using an electronic balance and ground using an agate mortar and pestle. Subsequently, the mixture was placed in a recrystallized alumina crucible and melted at 1550 °C. For homogeneity, the melt was held at 300, 600, 900, 1200, and 1500 °C for 1 hour (h). The melt was then quickly quenched between two copper plates. In the end, the glass samples were annealed at 400 °C for 4 hours. The physical picture of the as-synthesized samples is shown in Fig. 1. It is observed that the bulk density (Dg) increases from 2.32 to 2.48 g cm−3 with an increase in samarium ions (Sm3+) at the cost of lighter magnesium ions (Mg2+). It is due to the replacement of lighter magnesium ions (Mg2+) by the heavier samarium ions (Sm3+). In addition, such replacement alters the overall atomic packing density of the glass network.
Table 1 Composition in wt% and mol% of Sm2O3-containing magnesium borosilicate glasses along with their experimental density
| Glass ID ↓ |
Elemental composition (wt%) |
Oxide composition (mol%) |
Dg (g cm−3) |
| Si |
B |
Mg |
Sm |
O |
SiO2 |
B2O3 |
MgO |
Sm2O3 |
| MS-0 |
0.233 |
0.124 |
0.060 |
— |
0.581 |
50 |
40 |
10 |
— |
2.32 |
| MS-0.5 |
0.233 |
0.124 |
0.057 |
0.004 |
0.580 |
50 |
40 |
9.5 |
0.5 |
2.38 |
| MS-1 |
0.233 |
0.124 |
0.054 |
0.008 |
0.579 |
50 |
40 |
9 |
1 |
2.43 |
| MS-1.5 |
0.233 |
0.124 |
0.051 |
0.012 |
0.578 |
50 |
40 |
8.5 |
1.5 |
2.48 |
 |
| | Fig. 1 Photograph of as-synthesized glass samples melted and annealed at 1550 °C and 400 °C, respectively. | |
2.2 Structural and crystallization kinetics study
To analyze the structure of the as-prepared samples, XRD patterns were acquired using a PANalytical X'Pert Pro diffractometer with Cu Kα radiation (λ = 1.54 Å). The instrument was operated at an accelerating voltage of 40 kV and a current of 30 mA. Diffractograms were recorded in the 2θ range of 10°–90°, with subsequent data processing performed via the SmartLab Studio II software package. Differential scanning calorimetry (DSC) was performed using a Linseis PT-1600 high-temperature system. For each measurement, approximately 20 mg of as-prepared powder was placed in a platinum crucible and heated from room temperature to 1000 °C in a nitrogen atmosphere. To analyze the non-isothermal crystallization kinetics of the prepared samples, DSC measurements were done at four different heating rates (θ = 10, 20, 30, and 40 °C min−1) with aluminium oxide (purity = 99.9%) as a reference material. The activation energies are determined using the Kissinger and Augis–Bennett models. Subsequently, the characteristic temperatures and activation energies are used to estimate the Lasocka parameters (A and B), thermal stability (ΔS), fluctuation-free volume (Fv), Avrami's constant (Ac) and the fragility index (Fi).
2.3 Study of mechanical properties using three different models
2.3.1 Makishima–Mackenzie (M–M) approach. The mechanical properties are assessed using the Makishima–Mackenzie, Rocherulle and bond compression models. The Makishima–Mackenzie framework is used to calculate elastic moduli, precisely the Young's modulus (Em) and other essential mechanical parameters of heavy metal oxide-modified glass materials. This theoretical approach depends on the packing density (Pt) and the dissociation energy of the constituent oxides (Di). For a multi-component glass system, Pt is determined using the following equation:| |
 | (2) |
where xi represents the mole fraction of the ith oxide. Pf denotes the packing factor of the respective oxide constituent (ApOq) and is defined according to Makishima as follows:31| | |
Pf (cm3 mol−1) = Na × 4.18 × (pRA3 + qRO3)
| (3) |
The symbol De represents the total dissociation energy and Di represents the dissociation energy of constituent oxides per unit volume, expressed in kcal cm−3. The values of De for each oxide are taken from the literature which are defined as follows:31,32| |
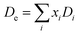 | (4) |
However, elastic moduli, like the Young's modulus (Em), shear modulus (Km) and bulk modulus (Rm), are calculated using the following equations:| | |
Em (GPa) = 83.6 × Pt × De
| (5) |
| |
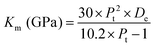 | (6) |
| | |
Rm (GPa) = 10 × Pt2 × De
| (7) |
| |
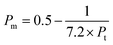 | (8) |
where Na, RA and RO, p, q and Pm are Avogadro's number, ionic radius of metal and oxygen, number of metal atoms, number of oxygen atoms, and Poisson's ratio, respectively.32,33
2.3.2 Rocherulle model. The Rocherulle model extends the theoretical framework established by Makishima and Mackenzie to evaluate the elastic behavior of the materials. The Rocherulle model introduces an alternative to calculate the packing factor (Pr), accounting for experimental evidence that suggests elastic moduli are independent of glass density. The total packing density (Rt) is defined as follows:| |
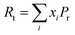 | (9) |
The value of Pr is determined using the following equation:| |
 | (10) |
The packing factor (Pr) is determined using the density of the individual constituent oxides rather than the bulk density of the glass sample. Further, similar mechanical properties as defined in the Makishima–Mackenzie approach can be calculated by substituting Rt in place of Pt.
2.3.3 Bond compression model. The bond compression model provides an idea about the quantitative interpretation of the experimental elastic moduli. According to this model, the bulk modulus (Bbc) can be written as follows:34| |
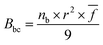 | (11) |
where r represents the bond length between the cation and anion, ![[f with combining macron]](https://www.rsc.org/images/entities/i_char_0066_0304.gif) denotes the average stretching force constant, and nb signifies the number of network bonds per unit volume of the glass. It is defined as follows:
denotes the average stretching force constant, and nb signifies the number of network bonds per unit volume of the glass. It is defined as follows:| |
 | (12) |
Here, NA represents Avogadro's number, nf denotes the number of network bonds per glass formula unit, and V signifies the molar volume of the glass. For three-dimensional multicomponent oxide glasses, the parameters Bbc and nb are defined as follows:| |
 | (13) |
| |
 | (14) |
Here, x denotes the molar fraction of the constituent oxides. The theoretical Poisson ratio of the multicomponent oxide glass systems can be determined as follows:34| |
 | (15) |
Here, nc represents the average cross-link density of the glass network, which is defined as follows:| |
 | (16) |
Here, (nc)i represents the number of cross-links per cation for the i-th oxide (defined as the number of bridging bonds per cation minus two), (Nc)i denotes the number of cations per formula unit, and  is the total number of cations per glass formula unit. Furthermore, the average atomic ring size (L) for a three-dimensional network structure can be expressed as:35
is the total number of cations per glass formula unit. Furthermore, the average atomic ring size (L) for a three-dimensional network structure can be expressed as:35| |
 | (17) |
Here,  represents the bond-bending force constant and is assumed to be proportional to the stretching force constant
represents the bond-bending force constant and is assumed to be proportional to the stretching force constant ![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) for the first approximation, and Be denotes the experimental bulk modulus. The stretching force constant
for the first approximation, and Be denotes the experimental bulk modulus. The stretching force constant ![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) of the glass network is expressed as
of the glass network is expressed as| |
 | (18) |
2.4 Radiation shielding analysis
2.4.1 MCNP code. Monte Carlo simulations may be used to solve highly complex physical problems. The Monte Carlo simulation code efficiently reproduces the experimental environment by accounting for the geometrical and physical characteristics of the employed equipment, cross-section values, and other databases used in experimental investigations. The literature review demonstrates that the Monte Carlo simulation method is frequently employed to evaluate the radiation shielding characteristics of various glasses.36,37 Using the Monte Carlo approach, μ/Dg is determined for the prepared glasses. The MCNP5 gamma-ray attenuation setup, which includes several simulation components like the glass sample, a Pb collimator for radiation, a point isotropic source, F4 tally mesh detection field, and Pb blocks to prevent the dispersed radiation, is shown in Fig. 2 as a 3-D image (obtained using Visual Editor VE). The F4 tally at the detection field was positioned at a distance of 70 cm along the same line as the point radiation source, and was used to calculate the average photon flux per MeV cm2 s−1 in the detecting field that passed through the detector. In this simulation, the glass sample was placed 50 cm from the source, between the F4 tally mesh (detection field) and the source. The average photon flux from the glass in the F4 detection field may be obtained by recording each average photon flux from the MCNP5 output file. The Beer–Lambert equation may then be used to determine and show the linear attenuation coefficient. The mass attenuation coefficients in various photon energies were then calculated by dividing each observed linear attenuation coefficient by the glass density. The characteristics of the material determine the structure of the MCNP5 code cell design (the cell structure of the modeled glasses was taken into consideration as a distinct cell in the input file). As a result, for each computation, the cell structure has been characterized as a unique glass sample with varying element mass percentages and densities, as shown in Table 1. When establishing material characteristics using the MCNP5 material card, the mass fractions of each ingredient are considered in accordance with the form criteria of the MCNP5 code. To depict a cell with certain material qualities, the material should then be set to Mn on the material card. The NPS value has been set at 108 particles.38,39 The linear attenuation coefficient (μ, cm−3), which represents the effectiveness of shielding materials, can be evaluated using the Beer–Lambert law as shown in eqn (19). Other factors such as the mass attenuation coefficient (MAC) and half value layer (HVL) can be extracted using eqn (20) and (21), where x represent the thickness of the sample in the following equation:18,40| |
 | (20) |
| |
 | (21) |
 |
| | Fig. 2 Gamma photon simulation setup via MCNPX visual editor. | |
2.4.2 Phy-X: PSD software. Complementary shielding parameters, including the effective atomic number (Zeff), exposure buildup factor (EBF), and energy absorption buildup factor (EABF), are computed using Phy-X: PSD software, which is developed by Sakar et al.41 This is a user-friendly computational tool designed for the rapid evaluation of attenuation factors. The input parameters required for the analysis include elemental compositions, material density, and the gamma photon energy range. The compositions of the candidate composites can be input into the Phy-X: PSD software in either mol percent (mol%) or weight percent (wt%). This software is available for any researcher at https://phy-x.net/.42
3 Results and discussion
3.1 X-ray diffraction (XRD) analysis
The XRD patterns of the as-quenched glass samples are illustrated in Fig. 3. The absence of Bragg reflections confirms the amorphous nature of the synthesized samples. Furthermore, the XRD patterns exhibit two diffuse humps at approximately 24° and 45°, which suggests that the incorporation of Sm2O3 promotes phase separation in the glass matrix.43 This diffuse scattering indicates the absence of long-range periodicity in the atomic arrangement, consistent with the presence of short-range order. This structural heterogeneity is attributed to the coexistence of discrete borate and silicate networks.44 Moreover, the broadening of the hump increases with an increase in Sm2O3 concentration, which leads to an increase in glass-forming ability.
 |
| | Fig. 3 XRD patterns of the as-prepared 50SiO2 + 40B2O3+ (10 − x)MgO + (x)Sm2O3 series. | |
3.2 Study of mechanical properties using various models
The study of fundamental structural units of oxide glasses is essential to understand their mechanical properties.45 However, the relative orientation of adjacent units remains consistent throughout a crystalline lattice, and a random distribution of orientations characterizes a vitreous state.
Table 2 summarizes the crystallographic parameters utilized to calculate the bond compression bulk modulus (Bbc), Poisson's ratio (σcal), and the average atomic ring size (L) of the glass samples. The elastic moduli are calculated through the bond compression model (Tables 3 and 4). The experimental bulk modulus (Be) exhibits an increase from 148 to 150 GPa with increasing Sm2O3 content. The increase in the bulk modulus is directly proportional to the number of bonds per unit volume (nb). nb increases from 7.68 to 7.77 × 1028 m−3 with the increasing content of Sm2O3 from 0 to 1.5 mol%, which is associated with an increase in the mean cross-link density (nc), as given in Table 3. It signifies improved glass rigidity, as evidenced by the formation of rigid BO4 units.46
Table 2 Parameters utilised in the calculations are sourced from the corresponding crystal structures of the individual oxides
| Oxide ↓ |
r (Å) |
nf |
nc |
![[f with combining macron]](https://www.rsc.org/images/entities/i_char_0066_0304.gif) (N m−1) (N m−1) |
| SiO2 |
1.62 |
4 |
2 |
432 |
| B2O3 |
1.37 |
3 |
2 |
410 |
| MgO |
1.82 |
6 |
4 |
282 |
| Sm2O3 |
2.30 |
6 |
4 |
210 |
Table 3 Calculated values of the average force constant (![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) ), number of network bonds per unit volume (nb), average cross-link density
), number of network bonds per unit volume (nb), average cross-link density  and Poisson's ratio (σcal) for the present glass system
and Poisson's ratio (σcal) for the present glass system
| Glass ID ↓ |
ρ (g cm−3) |
![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) (N m−1) (N m−1) |
nb × 1028 (m−3) |

|
σcal |
| MS-0 |
2.32 |
458.5 |
7.68 |
3.39 |
0.206 |
| MS-0.5 |
2.38 |
456.3 |
7.73 |
3.41 |
0.206 |
| MS-1 |
2.43 |
453.3 |
7.75 |
3.43 |
0.205 |
| MS-1.5 |
2.48 |
451.0 |
7.77 |
3.45 |
0.205 |
Table 4 Calculated values of the experimental bulk modulus (Be), bond compression bulk modulus (Bbc), ratio (Bbc/Be), and average atomic ring size (L) for the present glass system
| Glass ID ↓ |
Be (GPa) |
Bbc (GPa) |
Bbc/Be |
L (nm) |
| MS-0 |
148.01 |
152.47 |
1.03 |
0.410 |
| MS-0.5 |
149.10 |
151.74 |
1.01 |
0.409 |
| MS-1 |
149.94 |
150.74 |
1.00 |
0.408 |
| MS-1.5 |
150.88 |
149.97 |
0.99 |
0.407 |
Additionally, the Bbc/Be ratio (bond-bending constraints within the network) is directly proportional to the ring diameter (L). The reduction in average ring size (L) from 0.410 to 0.407 nm and Bbc/Be from 1.03 to 0.99 confirms the increased network connectivity.
The mechanical properties of the developed glass samples, particularly stiffness and structural integrity, can be characterized using the packing density, dissociation energy per unit volume, and elastic moduli. To determine these moduli, the Makishima–Mackenzie (M–M) model and the Rocherulle model are employed.29,47 Both the models help to correlate microscopic characteristics (effects of network formers and modifiers) by providing insights into the local structure configurations of the glasses. The M–M model adopts a macroscopic approach, utilizing experimentally determined bulk density to derive packing density. On the other hand, the Rocherulle model provides a microscopic perspective by calculating the packing density using the molecular weights and densities of the constituent oxides. The calculated elastic moduli (Em, Km and Rm) and Poisson's ratio (Pm) are summarized in Table 5. It is observed that the elastic moduli increase (Em and Rm increase from 86.26 to 107.95 GPa and from 62.11 to 95.55 GPa, respectively) with an increase in Sm2O3 concentration, consistent with an increase in the average number of bonds per unit volume and cross-link density. These variations suggest that the stiffness of the glass framework is primarily governed by the nature of the network bonding, especially alterations in cross-linking and coordination as observed in the Em and Rm trend.48,49 The transition from BO3 to BO4 with the formation of bridging oxygens (BOs) is responsible for enhancing the rigidity and elastic moduli of the present glass samples.46,50
Table 5 Calculated elastic moduli (Em, Km and Rm), Poisson's ratio (Pm), hardness (Vh), and packing density (Pt) using the Makishima–Mackenzie model of the present samples
| Glass ID ↓ |
Makishima–Mackenzie (M–M) model |
| Em (GPa) |
Km (GPa) |
Rm (GPa) |
Pm |
Vh (GPa) |
Pt (cm3 mol−1) |
| MS-0 |
86.26 |
36.24 |
62.11 |
0.269 |
8.42 |
0.602 |
| MS-0.5 |
93.68 |
38.80 |
72.83 |
0.286 |
8.59 |
0.650 |
| MS-1 |
100.80 |
41.28 |
83.80 |
0.300 |
8.73 |
0.695 |
| MS-1.5 |
107.95 |
43.77 |
95.55 |
0.312 |
8.87 |
0.740 |
The incorporation of Sm2O3 into the glass network results in a simultaneous increase in total dissociation energy (De) and packing density (Pt), as detailed in Table 7. The rise in De and density of network bonds is primarily associated with the high dissociation energy of Sm2O3 compared to other constituents.48,51 As De increases, the glass structure becomes progressively more compact, leading to the enhanced elastic moduli as given in Table 5. The present findings indicate that the prepared glass samples possess higher resistance to mechanical deformation. Further, the increase in total packing density and network stiffness is governed by the higher atomic mass, strong bonding between Sm and O and the nature of Sm3+ ions compared to other cations in the matrix.52 Specifically, Sm2O3 facilitates higher packing density by enabling ions to occupy interstitial voids within the glass network.
Poisson's ratio (Pm), defined as the ratio of lateral strain to longitudinal strain under an applied tensile force, exhibits a minimal increasing trend from 0.269 to 0.312. According to Souri,53 loosely packed glass networks permit higher atomic mobility, which lowers the lateral strain. Consequently, Pm tends to increase in more tightly packed structures or vice versa. In the present study, the increasing trend of Pm suggests that Sm2O3 promotes a more compact network configuration, which is also satisfied by the increasing trend of elastic moduli and structural rigidity.
The theoretical elastic moduli are calculated by using the Rocherulle model as presented in Table 6. The elastic moduli and Poisson's ratio increase with the increase of Sm2O3 content. Fig. 4 illustrates the variations in moduli obtained using the Makishima–Mackenzie and Rocherulle models as a function of Sm2O3 content. It is noticed that all the elastic moduli show an increasing trend with increasing Sm2O3 concentration. To assess the consistency of theoretical frameworks, the relative percentage deviation (Rd) of the Young's modulus is summarized in Table 7. The maximum relative deviation between models was 12.11%, indicating reasonable agreement.
Table 6 Calculated elastic moduli (Em, Km and Rm), Poisson's ratio (Pm), hardness (Vh), and packing density (Rt) using the Rocherulle model of the present samples
| Glass ID ↓ |
Rocherulle model |
| Em (GPa) |
Km (GPa) |
Rm (GPa) |
Pm |
Vh (GPa) |
Rt (cm3 mol−1) |
| MS-0 |
98.15 |
40.29 |
80.42 |
0.297 |
8.61 |
0.685 |
| MS-0.5 |
102.47 |
41.81 |
87.15 |
0.304 |
8.72 |
0.711 |
| MS-1 |
107.04 |
43.42 |
94.49 |
0.311 |
8.84 |
0.738 |
| MS-1.5 |
111.45 |
44.98 |
101.85 |
0.318 |
8.91 |
0.764 |
 |
| | Fig. 4 Variation in elastic moduli (Em, Km and Rm) with Sm2O3 content (mol%) using (a) the Makishima–Mackenzie model and (b) Rocherulle model. | |
Table 7 Total dissociation energy (De) and relative deviation (Rd) in the Young's modulus (Em) measured by using the Makishima–Mackenzie and Rocherulle models
| Glass ID ↓ |
M–M model |
Rocherulle model |
Relative deviation (Rd) |
De (GPa) |
| Pt |
Em (GPa) |
Rt |
Em (GPa) |
Em (%) |
| MS-0 |
0.60 |
86.26 |
0.68 |
98.15 |
12.11 |
17.14 |
| MS-0.5 |
0.65 |
93.68 |
0.71 |
102.47 |
8.57 |
17.24 |
| MS-1 |
0.69 |
100.80 |
0.73 |
107.04 |
5.82 |
17.35 |
| MS-1.5 |
0.74 |
107.95 |
0.76 |
111.45 |
3.14 |
17.45 |
Microhardness (Vh) is defined as the pressure required for eliminating free volume within the glass matrix and it characterizes network deformation. Under high hydrostatic pressure, the glass structure undergoes compaction, reducing free volume. The microhardness was determined using the following expression:44
| |
 | (22) |
Both the models show comparable hardness values that increase with increasing Sm
2O
3 content, attributed to the greater structural contribution of BO
4 units than BO
3 units in the glass network. Thus, it can be concluded that Sm
3+ ions with relatively high ionic field strength and strong Sm–O bonds are incorporated into the glass network. This leads to an increase in packing density and cross-linking within the borosilicate network, resulting in a more compact and rigid structure. Consequently, the elastic moduli such as the Young's modulus (
Em), bulk modulus (
Rm), and shear modulus (
Km) increase with increasing Sm
2O
3 concentration in place of MgO.
3.3 Study of non-isothermal crystallization kinetics
Differential scanning calorimetry (DSC) is employed to study the thermal performance of the glass samples. It provides both qualitative insights into structural changes and quantitative data regarding the characteristic temperatures. The glass transition (Tg), onset crystallization (Tc), and peak crystallization (Tp) temperatures were identified through DSC thermograms. In the DSC thermographs, Tg appears as an endothermic baseline shift, while crystallization and melting are demonstrated as exothermic and endothermic peaks, respectively. Fig. 5 illustrates DSC thermographs at four heating rates (10–40 °C min−1) of the prepared glass samples. Tg was determined from the peak derivative in the DSC peak (DDSC) curve (dH/dT), as shown in Fig. 6, while Tc and Tp were identified through the combined analysis of the slope change and the corresponding DDSC peak.
 |
| | Fig. 5 DSC curves for (a) MS-0 and (b) MS-0.5 obtained at different heating rates. | |
 |
| | Fig. 6 Derivative curve of differential scanning calorimetry (DDSC) for (a) MS-0 at 10 °C and (b) MS-0.5 at 30 °C. | |
The characteristic temperatures of the present glasses exhibit an increase with an increase in heating rates (Table 8), which is associated with the sluggish thermal events in kinetic effects.54,55 The glass transition is related to the structural relaxation time (tr) of the material that becomes comparable to the observation time (δT). At a constant heating rate, Tg is governed by the packing density and cross-linkages in the glass structure. Tg depends on the heating rate (α), which arises because Tg is inversely proportional to tr; a higher heating rate (α = δT/tr) reduces the time available for molecular rearrangement, thereby requiring a higher temperature to achieve the transition.55
Table 8 Characteristic temperatures and sintering window (ΔT) of the synthesized glasses at different heating rates
| Glass ID ↓ |
Heating rate (α) (°C min−1) |
Tg1 (°C) |
Tg2 (°C) |
〈Tg〉 (°C) |
Tx (°C) |
Tc (°C) |
ΔT = Tc − Tg (°C) |
| MS-0 |
10 |
425 ± 1 |
551 ± 1 |
488 ± 1 |
692 ± 1 |
790 ± 1 |
302 ± 2 |
| 20 |
491 ± 1 |
639 ± 1 |
565 ± 1 |
743 ± 1 |
851 ± 1 |
386 ± 2 |
| 30 |
525 ± 1 |
673 ± 1 |
599 ± 1 |
770 ± 1 |
878 ± 1 |
279 ± 2 |
| 40 |
590 ± 1 |
705 ± 1 |
647 ± 1 |
834 ± 1 |
942 ± 1 |
295 ± 2 |
| MS-0.5 |
10 |
442 ± 1 |
561 ± 1 |
501 ± 1 |
697 ± 1 |
832 ± 1 |
331 ± 2 |
| 20 |
501 ± 1 |
605 ± 1 |
553 ± 1 |
712 ± 1 |
871 ± 1 |
318 ± 2 |
| 30 |
517 ± 1 |
671 ± 1 |
594 ± 1 |
850 ± 1 |
904 ± 1 |
310 ± 2 |
| 40 |
524 ± 1 |
701 ± 1 |
612 ± 1 |
898 ± 1 |
939 ± 1 |
327 ± 2 |
| MS-1 |
10 |
481 ± 1 |
595 ± 1 |
538 ± 1 |
705 ± 1 |
869 ± 1 |
331 ± 2 |
| 20 |
502 ± 1 |
670 ± 1 |
586 ± 1 |
793 ± 1 |
873 ± 1 |
287 ± 2 |
| 30 |
552 ± 1 |
727 ± 1 |
639 ± 1 |
804 ± 1 |
879 ± 1 |
240 ± 2 |
| 40 |
561 ± 1 |
740 ± 1 |
650 ± 1 |
863 ± 1 |
919 ± 1 |
269 ± 2 |
| MS-1.5 |
10 |
510 ± 1 |
632 ± 1 |
571 ± 1 |
762 ± 1 |
875 ± 1 |
304 ± 2 |
| 20 |
539 ± 1 |
649 ± 1 |
594 ± 1 |
773 ± 1 |
896 ± 1 |
302 ± 2 |
| 30 |
572 ± 1 |
662 ± 1 |
617 ± 1 |
843 ± 1 |
908 ± 1 |
291 ± 2 |
| 40 |
601 ± 1 |
673 ± 1 |
637 ± 1 |
895 ± 1 |
951 ± 1 |
3145 ± 2 |
The substitution of Sm2O3 in place of MgO facilitates a structural modification in the borosilicate network, promoting the conversion of Q4 + Q2 into Q1 + Q3 units.46 This transition enhances network connectivity and polymerization, resulting in a more connected glass framework. Therefore, a general trend in the characteristic temperatures, i.e., increasing with increasing Sm2O3 content, is observed. It supports an increase in thermal stability with increasing Sm2O3 content, which is attributed to the shorter bond length, higher cross-link density, and a corresponding increase in overall network rigidity.
3.3.1 Lasocka parameters. The glass transition temperature (〈Tg〉) highly depends on the heating rate (α) and can be expressed as follows:| | |
〈Tg〉 = X + Y × ln(α)
| (23) |
The constants X and Y from the Lasocka equation were determined through linear regression of the 〈Tg〉 versus ln(α) plots, as illustrated in Fig. 7. The constant X represents the glass transition temperature extrapolated to a heating rate of 1 °C min−1, while Y is a configurational constant sensitive to the glass composition and quenching rate. Y is governed by the cooling rate and decreases with slower cooling rates. The specific values derived from the intercept (X) and slope (Y) are listed in Table 9. The incorporation of Sm2O3 into the SiO2–B2O3–MgO glass system is responsible for the systematic decrease in X (459.88 to 231.84 K). Conversely, Y increases with an increase in Sm2O3 content, suggesting that the MS-1.5 sample undergoes greater configurational modifications. The inverse relationship in X and Y for MS-1.5 glass indicates better resistance to devitrification.46 Additionally, enhanced stability is supported by the higher activation energy, as discussed in the subsequent sections.
 |
| | Fig. 7 The Lasocka plot shows the variation of Tg versus ln(β) for the as-prepared glasses. A straight line represents the linear fit passing through the data points, with error bars indicating the uncertainty from the linear fitting. | |
Table 9 Lasocka parameters and the frequency factor (ν) for as-prepared samples
| Glass ID ↓ |
Lasocka parameter |
ν (s−1) |
| X |
Y |
| MS-0 |
459.88 |
46.77 |
5.24 × 1031 |
| MS-0.5 |
311.62 |
81.80 |
8.91 × 1017 |
| MS-1 |
340.08 |
84.97 |
5.49 × 1015 |
| MS-1.5 |
231.84 |
110.71 |
1.20 × 1013 |
3.3.2 Crystallization activation energy. Glass initially undergoes a glass transition on heating, followed by crystallization to form a glass-ceramic material.56 The nucleation and growth phenomena occur during the crystallization process. It is governed by the network connectivity, chemical composition, viscosity, and the free energy difference between the phases. Glass transition and crystallization processes are kinetically controlled phenomena that proceed only when the available thermal energy is sufficient to overcome their respective activation energy barriers. To understand the barriers in the present glass samples, two distinct mathematical models were employed to determine the activation energies associated with glass transition and crystallization events. According to the Kissinger model,57 the activation energy for the glass transition is derived from the Tg and heating rate (α) as follows:| |
 | (24) |
In the above equation, C, Eg, and R are the constant, activation energy of the glass transition, and the universal gas constant, respectively. As illustrated in Fig. 8(a), the plot of 1000/〈Tg〉 and ln(〈Tg2〉/α) exhibits a linear trend, where the slope is utilized to calculate the activation energy (Eg) for all samples. The data are well fitted by linear regression, and the obtained Eg values are summarized in Table 10. Error bars (2%) are included to specify the uncertainty associated with the linear fitting process.
 |
| | Fig. 8 Activation energy of crystallization (Eg) of as-prepared glasses using the (a) Kissinger model and (b) Augis–Bennett model. | |
Table 10 obtained values of Eg, Ec and Er for the as-prepared samples
| Glass ID ↓ |
Kissinger model |
Augis–Bennett model |
| Eg (kJ mol−1) |
Ex (kJ mol−1) |
Ec (kJ mol−1) |
Eg (kJ mol−1) |
Ex (kJ mol−1) |
Ec (kJ mol−1) |
| MS-0 |
38 |
65 |
107 |
45 |
73 |
110 |
| MS-0.5 |
55 |
71 |
133 |
62 |
85 |
143 |
| MS-1 |
87 |
89 |
161 |
95 |
98 |
179 |
| MS-1.5 |
118 |
119 |
333 |
127 |
136 |
348 |
In the Augis–Bennett model, the relationship between the heating rate (α) and the glass transition temperature (〈Tg〉) is defined as follows:
| |
 | (25) |
Fig. 8(b) illustrates the plots of ln(〈
Tg〉/
α)
versus 1000/〈
Tg〉 for all glass samples. The Kissinger model is adapted to determine the crystallization activation energy by substituting the glass transition parameters (
Tg and
Eg) with the crystallization temperatures (
Tx or
Tc) and activation energies (
Ex or
Ec), respectively. The crystallization activation energy (
Ec) and the frequency factor (
ν) are evaluated using the Augis–Bennett model as follows:
| |
 | (26) |
The crystallization activation energies (
Ex and
Ec) are determined through graphical analysis using the Kissinger and Augis–Bennett models, as illustrated in
Fig. 9 and 10, respectively. The calculated
Ex and
Ec values are summarized in
Table 10.
Ec represents the energy barrier that must be overcome for the glass-to-crystal transition. Thus, higher
Ec values signify greater resistance to devitrification with enhanced thermal stability. The activation energies derived from the Kissinger and Augis–Bennett models have strong agreement, with minor discrepancies attributable to the distinct underlying assumptions of each framework. The reliability of the results is supported by an excellent correlation coefficient (
R2) of 0.99. The Augis–Bennett model can determine the frequency factor (
ν). The obtained values of the frequency factor are given in
Table 9. It represents the atomic attempt frequency required to surmount the crystallization barrier and provides critical insights into the availability of nucleation sites in the glass network.
47,58 Consequently,
ν serves as an essential parameter for assessing the overall stability of the glass samples. Both models consistently reveal a linear trend in
Ex and
Ec,
i.e., increasing with an increase in Sm
2O
3 content. Notably, the MS-0 sample exhibits minimum
Ec and maximum
ν values, signifying a higher tendency for crystallization facilitated by an elevated atomic attempt frequency. On the other hand, the MS-1.5 sample exhibits the highest
Ec and the lowest
ν, indicating greater thermal stability against devitrification compared to the other glass samples. This stability can be associated with the more uniform network structure that requires higher energy for atomic rearrangement. So, it can be concluded that the incorporation of Sm
2O
3 increases the activation energy by modifying the network connectivity with enhanced structural integrity of the borosilicate framework. Thus, it can be concluded that the Kissinger and Augis–Bennett models show an increase in crystallization activation energy (
Ec) with increasing Sm
2O
3 concentration. This behavior suggests that Sm
3+ ions suppress crystallization by hindering atomic diffusion and structural rearrangement in the glass matrix.
 |
| | Fig. 9 Activation energy of crystallization (Eg) of as-prepared glasses using the (a) Kissinger model and (b) Augis–Bennett model. | |
 |
| | Fig. 10 Activation energy of crystallization (Eg) of as-prepared glasses using the (a) Kissinger model and (b) Augis–Bennett model. | |
3.4 Avrami constant
The shape of the DSC crystallization peak provides essential information regarding the crystallization mechanism in glass samples. The sharp exothermic peaks indicate bulk crystallization, whereas broader peaks indicate surface crystallization.59 The full width at half maxima (FWHM; ΔTw) quantifies peak broadening. Consequently, the Avrami constant (CA), which characterizes the dimensionality and mechanism of crystallization, can be determined using ΔTw and activation energy (Ec) according to the following expression:60| |
 | (27) |
The Avrami constant (CA), typically ranging from 1 to 4, is a key parameter for interpreting the crystallization mechanism. When CA = 3, it signifies bulk crystallization consistent with the sharp exothermic peaks observed in the DSC thermographs. When CA = 1, it indicates surface-mediated crystallization. The heating rate also affects the Avrami constant because the heating rate (α) significantly influences the peak shape as shown in Table 11. It is observed that CA increases from 10 to 40 °C min−1 as α increases. It is also noticed that CA increases with increasing Sm2O3 content, which suggests that the substitution of Sm2O3 promotes bulk crystallization in the present glass samples. Generally, a higher CA reflects a stronger tendency toward volume-nucleated, multidimensional growth.61
Table 11 Various kinetic parameters of the as-prepared glasses at different heating rates
| Glass ID ↓ |
θ (°C min−1) |
CA |
Vf × 10−3 |
If |
ΔS (°C) |
| MS-0 |
10 |
1.20 |
90.16 |
4.81 |
60.64 |
| 20 |
1.59 |
104.38 |
4.16 |
73.78 |
| 30 |
2.70 |
110.66 |
3.92 |
50.30 |
| 40 |
3.86 |
119.53 |
3.63 |
49.24 |
| MS-0.5 |
10 |
1.76 |
67.18 |
6.46 |
89.19 |
| 20 |
2.69 |
74.15 |
5.85 |
91.43 |
| 30 |
2.46 |
79.65 |
5.45 |
28.18 |
| 40 |
3.62 |
82.06 |
5.29 |
21.90 |
| MS-1 |
10 |
1.81 |
47.08 |
9.22 |
100.89 |
| 20 |
2.37 |
51.28 |
8.47 |
39.18 |
| 30 |
3.34 |
55.92 |
7.76 |
281.6 |
| 40 |
4.67 |
56.88 |
7.63 |
23.17 |
| MS-1.5 |
10 |
2.76 |
37.38 |
11.62 |
60.16 |
| 20 |
3.13 |
38.88 |
11.17 |
62.53 |
| 30 |
3.79 |
40.39 |
10.75 |
30.65 |
| 40 |
4.15 |
41.70 |
10.41 |
27.60 |
3.4.1 Fluctuation free volume. The fluctuation-free volume (Vf) is an important parameter for studying the structural changes that govern glass kinetics during the glass transition and crystallization. Higher values of Vf indicate a more open network that facilitates atomic movement and structural rearrangements. The assessment of Vf is crucial for correlating network connectivity and plays a fundamental role in determining the overall mechanical and thermal stability of the glass samples. The fluctuation-free volume is calculated using the following expression:| |
 | (28) |
The obtained fluctuation-free volume (Vf) reveals the significant structural modifications in the glass network, with a clear heating rate and Sm2O3 concentration dependency. Vf decreases as the heating rate increases from 10 to 40 °C min−1 as given in Table 11. This phenomenon suggests that a higher heating rate provides sufficient time for the glass network to reach thermodynamic equilibrium, thereby trapping less free volume. This excess free volume helps structural modifications and higher atomic mobility.47 Further, the substitution of MgO with Sm2O3 in the borosilicate glass leads to a decrease in Vf. This trend validates the role of Sm2O3 as a network former, which enhances the borosilicate glass network by forming Si–O–B linkages and generating BOs. The resultant structure is more compact, less expandable, and less flexible. The minimal Vf of the MS-1.5 glass indicates a highly compact and rigid network, whereas the higher of Vf in the MS-0 glass reflects a more disordered configuration. This structural modification is consistent with the observed trend in the crystallization activation energy (Ec), where low Vf corresponds to high Ec, thereby confirming that Sm2O3 enhances the overall network rigidity. From a functional perspective, the dense network associated with the low Vf typically enhances mechanical robustness and chemical durability. Conversely, the more open network characterized by high Vf facilitates ionic transport and may control the optical characteristics. Therefore, the incorporation of Sm2O3 supports tuning the physical and chemical properties of these borosilicate glasses.
3.4.2 Fragility index. The fragility index of glass-forming liquids classifies them as either kinetically “strong” or “fragile”. Kinetically strong liquids typically exhibit substantial covalent character with discrete coordination, resulting in a well-defined network structure. Conversely, kinetically fragile liquids are often derived from simple molecular fluids with isotropic coordination and are characterized by ionic or van der Waals forces.62The glass transition temperature (〈Tg〉) is conventionally defined as the temperature at which the liquid's viscosity (η) abruptly increases to 1012 Pa s. However, this definition is not universally applicable, as certain molecular liquids exhibit viscosities below 1010 Pa s at the glass transition. To address such a discrepancy, Angell introduced the concept of fragility, in which the logarithm of viscosity is plotted against the inverse normalized temperature (〈Tg〉/T). In this case, strong glass formers (SiO2) display Arrhenius temperature dependency, whereas fragile glass formers (ortho-terphenyl) exhibit distinct non-Arrhenius behavior. Furthermore, fragile structures are known to undergo rapid structural degradation near the glass transition.63 Consequently, the kinetics of the glass transition are quantified by the fragility index (If), which is most commonly defined by the slope of the viscosity curve as it approaches Tg on an Angell plot.
| |
 | (29) |
Upon incorporating
Vf,
eqn (29) simplifies to
| |
 | (30) |
The fragility index (
If) serves as a key indicator of glass-forming kinetics. Kinetically strong (KS) melts typically exhibit an
If value near 16. Traditional glass formers such as SiO
2, GeO
2, P
2O
5, and As
2O
3 generally fall within the range of 17 to 20, but B
2O
3 is an exception at approximately 32.
62,64 Conversely, kinetically fragile (KF) melts display significantly higher values, approaching 200.
48 Table 11 represents the fragility indices of the prepared glass samples. The values range from 4.81 to 11.62, indicating that these compositions possess a favorable glass-forming ability consistent with kinetically strong characteristics. It is observed that glasses with higher Sm
2O
3 content exhibit higher fragility indices, which can be attributed to the interactions between borate and silicate structural units. In addition, the fragility index decreases with increasing heating rates. Finally, glass fragility is intrinsically linked to composition and bond topology, as modifications in local structural environments allow different regions to relax at different heating rates, resulting in a broad distribution of relaxation times.
3.4.3 Thermal stability. To evaluate the feasible processing window and resistance to devitrification of Sm2O3 modified SiO2−B2O3−MgO glasses, thermal stability is assessed using the working range (ΔS) and the Saad stability factor (ΔS). The working range (ΔT = Tc − 〈Tg〉) represents the temperature interval available for processing before the onset of crystallization. A larger ΔT value indicates enhanced thermal stability. Further, the Saad stability parameter (ΔS) provides an additional quantitative measure of this stability and is calculated as follows:| |
 | (31) |
The Saad stability factor (ΔS) analysis identifies an optimal Sm2O3 concentration for maximizing the thermal resistance of the glass system. The 1 mol% Sm2O3-doped sample (MS-1) demonstrates higher thermal stability, achieving a peak ΔS value of 100.89 as shown in Table 11. This enhancement is attributed to the formation of robust Si–O–B bonds, which increase network cross-linking and reduce the density of NBOs. On the other hand, the 1.5 mol% Sm2O3-doped sample (MS-1.5) exhibits the lowest stability (ΔS = 60.16), suggesting that the excessive Sm2O3 amount disrupts the glass network and promotes NBO formation. The undoped (MS-0) and 1 mol% Sm2O3 samples (MS-1) display intermediate stability, with values ranging from 60.64 to 49.24 and from 100.89 to 23.17, respectively. These findings highlight the need for proper optimization of Sm2O3 to tune thermal performance. Thus, MS-1 is identified as the most suitable glass for high-temperature processing and long-term applications in optoelectronics, solid-state devices, and protective coatings.
3.5 Gamma photon radiation shielding study
MAC serves as a vital parameter to study the radiation shielding capability of the materials based on the per-unit-mass. The incident gamma-photon energy and density of the glass matrix predominantly influence the behavior of materials. The MAC values show a pronounced decrease with an increase in gamma-photon energy, which is associated with the dominant gamma-photon interaction mechanisms (photoelectric (PE), Compton scattering (CS), and pair production (PP)) as shown in Fig. 11.65 Generally, density and MAC show an inverse relationship, which can be attributed to the substitution of MgO with Sm2O3 in glass samples. The incorporation of Sm2O3 improves the interaction cross-section, which leads to a change in the attenuation properties. Additionally, a distinct K-edge absorption feature centered around 48.6 keV is produced by increasing Sm2O3 content. This feature becomes more prominent with higher Sm2O3 content. Further, the increment in MAC values can be attributed to the higher effective atomic number and density introduced by Sm3+ ions, which increases the probability of photon interaction, particularly through the photoelectric effect at low photon energies. Fig. 12 and 13 depict the partial contributions of individual gamma-photon interaction mechanisms and their respective average percentages to the total MAC for the MS-0 sample across a broad gamma-photon energy range. Different patterns of interaction are observed with different energy domains. Precisely, the photoelectric effect (PE) dominates in the lower energy range, exhibiting a pronounced decline as gamma-photon energy increases.65 In contrast, the Compton scattering (CS) curve demonstrates a more gradual attenuation across the energy range. Rayleigh (coherent) scattering exhibits a nearly linear decreasing trend. The photoelectric effect is predominant in the lower energy range (0.015 to 0.3 MeV). The CS interaction dominates between 0.4 and 8 MeV and approaches 98% significance, while the PE is found at nearly 80% in the effective range (Fig. 13). At higher energies (between 10 and 15 MeV), a comparable trend is observed, with a prominent increase in the pair production (PP) interaction. It is attributed to its inverse-square dependence on gamma-photon energy, which governs the elastic scattering behavior of gamma-photons. The MAC values fall in the range of 1.95 × 10−2 to 3.93 (for MS-0) and 0.0199 to 4.98 (for MS-1.5) at 0.015 and 15 MeV, respectively. Overall, the relative contribution of each interaction is strongly influenced by both the gamma-photon energy and the compositions. The HVL denotes the material thickness necessary to reduce the incident gamma-photon intensity by 50%. It has a strong dependency on gamma-photon energy, which is associated with the gamma-photon–matter interaction mechanisms that vary across different energy regimes. The PE is dominant in the range of 10–100 keV and the HVL reveals a steep relationship with E (directly proportional to E3). In the case of CS between 100 and 1000 keV, the HVL exhibits a linear relationship with E, as shown in Fig. 14. On the other hand, in a high-energy zone where PP dominates (E > 1.022 MeV), the HVL follows an approximate inverse logarithmic trend with E (ln(E−1)). This behaviour in the PP region occurs due to the high atomic number (Z) of materials.37,66 Due to the overlapping energy dependence of PE and CS interactions, HVL values in the low and intermediate energy regions tend to converge closely. However, a more distinguishable gradient in HVL values is observed with an increase in the PP domain. Beyond the energy dependence, the HVL is also influenced by the composition of the shielding materials. At a constant gamma-photon energy, variations in material constituents yield different HVL values. For instance, at 10 MeV, the HVL spans from 13.3 cm in MS-0 to 12.1 cm in MS-1.5, indicating superior attenuation performance in the latter. These findings suggest that MS-1.5 exhibits enhanced gamma-photon radiation shielding efficiency, characterized by the lowest HVL across the evaluated energy spectrum (0.001 eV to 10 MeV).66
 |
| | Fig. 11 MAC values as a function of gamma photon energy for the MS series. | |
 |
| | Fig. 12 Partial interaction contributions to the total MAC vs. gamma photon energy for the MS-0 sample. | |
 |
| | Fig. 13 The average percentage contribution of PE absorption, CS and PP to the total MAC related to the MS-0 sample for 3 different gamma photon energy ranges. | |
 |
| | Fig. 14 Δ0.5 fluctuations for the provided samples as a function of gamma photon energy. | |
Fig. 15 illustrates the energy dependence of the effective atomic number (Zeff) of the prepared glass samples. As energy increases, it is observed that Zeff first decreases, then rises sharply, and finally gradually attenuates. The undoped glass sample (MS-0) exhibits a smooth, monotonic decrease, while Sm2O3-containing samples exhibit distinct, sharper maxima. This is attributed to the K-edge absorption of Sm3+, occurring near 46.8 keV, which induces a localized surge in photon interaction probability.66 Increased inner-shell electron interactions, particularly in the PE absorption regime, are responsible for the observed peak. The CS-dominant interaction mechanism causes Zeff to resume a downward trajectory beyond this energy threshold, and the impact of PE absorption diminishes. Zeff is more sensitive to the composition due to the presence of high atomic number dopants such as Sm2O3. Even minor additions of Sm3+ significantly enhance Zeff, especially within the photon energy range corresponding to the K-edge. Furthermore, the dependence of Zeff on compositional variation is inherently nonlinear. The incorporation of rare-earth elements introduces a non-proportional enhancement in Zeff, underscoring the complex interplay between elemental concentration and photon interaction cross-sections. As demonstrated in Fig. 15, low-weight fractions of Sm2O3 induce a substantial increase in Zeff in the vicinity of the absorption edge. At higher photon energy levels, the Zeff curves exhibit minimal deviation. Nevertheless, the undoped glass sample exhibits the lowest gamma-ray attenuation efficiency, while the Sm3+ containing glass sample shows superior shielding capability over a broad energy range.66
 |
| | Fig. 15 Variation of Zeff with gamma photon energy for the MS series. | |
In addition, the figure shows that Zeff values range from 8.43 to 11.2 (for MS-0) and from 8.5 to 15.4 (for MS-1.5). RPE% is the capacity of materials to attenuate ionizing radiation. Within the low-energy photon domain, a notable gradient in RPE% is observed, whereas in the intermediate- and high-energy regions, the curves are converged and approach zero, indicating minimal shielding differentiation at elevated energies. It is observed that RPE% is intrinsically governed by the density and composition. The incorporation of high atomic number (Z) additives into the glass matrix significantly improved the attenuation performance. As Sm2O3 content increases, the shielding capacity of the prepared glass samples increases.37 Furthermore, a strong positive correlation is established between gamma-photon attenuation and the corresponding RPE values, supporting that materials exhibiting higher photon interaction probabilities provide superior radiation mitigation. The MS-1.5 sample shows the highest radiation shielding efficiency across a broad energy spectrum compared to the other glass samples, as shown in Fig. 16. In addition, the MS-0 sample achieves 100% protection till 0.02 MeV while the MS-1.5 sample achieves 100% protection till 0.0263 MeV. In addition to this, the increasing trend of RPE with Sm2O3 concentration is due to the absorption of a greater fraction of incident gamma photons. The improvement in RPE is mainly associated with the increased photon interaction cross-section and the enhanced stopping power resulting from the higher atomic number of Sm.
 |
| | Fig. 16 Variations of evaluated RPE for the selected samples with photon energy. | |
The EBF quantifies the accumulation of scattered radiation within a shielding material. Fig. 19 and 20 illustrate the energy-dependent performance of EBF of the prepared glass samples. The dependency of EBF on material density reflects a multifaceted interaction influenced by incident photon energy, depth of penetration, and composition of the glass, particularly Zeff. It is observed that the relationship between EBF and photon energy is inherently nonlinear, exhibiting a pronounced maximum at intermediate energy levels, followed by a decline at both lower and higher energies. This intermediate-energy behavior is attributed to the compositional makeup of the synthesized glass matrices. Precisely, an increase in Sm3+ concentration is accompanied by a reduction in Mg2+ content, resulting in pronounced peaks. This phenomenon arises due to the substitution of a high-Z element (Sm3+) for a low-Z element (Mg2+).37 Moreover, the composition alteration enhances the probability of photoelectric interactions. It is evident, particularly by the sharp peaks near 46.8 keV in the PE region, as the Sm3+ concentration increases. Conversely, the occurrence of broader peaks around 1305 keV is associated with reduced Mg2+ content. These variations could also result from elemental substitution-induced changes in material density. Additionally, at a penetration depth of 15 mfp, the prepared glass samples demonstrate comparable behavior, with variations in EBF magnitude correlating to differences in the atomic structure and bulk density of the glass matrix. The same trend is also reported for EABF for the prepared glass samples – a parameter used to quantify how gamma-photons are absorbed and scattered within the materials in the wide energy ranges – as shown in Fig. 17 and 18. As expected, at fixed mfp, the EABF follows the trend EABF1.5 < EABF1 < EABF0.5 < EABF0. The MS-1.5 sample has the lowest EBF and EABF values, while the MS-0 sample possesses the highest values. The EABF values slightly decrease with increasing Sm2O3 concentration at most photon energies. This reduction suggests that photon penetration and multiple scattering inside the material become less significant as the glass becomes more effective at attenuating photons. The presence of heavier Sm3+ ions increases the probability of photon absorption before multiple scattering events occur.
 |
| | Fig. 17 EBF versus gamma photon energy at different penetration depths for all investigated samples. | |
 |
| | Fig. 18 Variations of the EBF for the MS series as a function of gamma photon energy at a penetration depth of 15 mfp. | |
 |
| | Fig. 19 EABF values versus gamma photon energy at different penetration depths for the MS series. | |
 |
| | Fig. 20 Changes of EABF values for the MS series as a function of gamma photon energy at a penetration depth of 15 mfp. | |
The energy dependency of the kerma relative to air (Kα) of the prepared glass samples is shown in Fig. 21. Kerma is a measure of energy transferred from radiation to matter and is maximum for the MS-1.5 glass sample in a wide energy range. This indicates that the MS-1.5 sample has the highest value of kinetic energy released per unit mass compared to the other samples.67 On the other hand, the gamma-photon loss in the MS-0 glass sample is small due to its lowest Kα. Furthermore, the MS-1.5 sample shows a sharp peak due to the higher Sm3+ content. Moreover, a descending trend in Kα is seen due to the increasing Sm3+ content as follows: Kα(MS-0) < Kα(MS-0.5) < Kα(MS-1) < Kα(MS-1.5).
 |
| | Fig. 21 The change of Kα of the present samples as a function of gamma photon energy. | |
The gamma-photon dose rate (Dr) has been quantitatively assessed for the prepared glass samples and illustrated in Fig. 22 and 23. The data indicate that the increase in sample thickness (denoted as x) leads to a corresponding decrease in Dr. This trend is consistently observed across all glass samples, typically exhibiting a distinct valley in the low-energy region. As thickness increases further, the respective Dr curves converge, suggesting the potential for graphical overlap. This convergence indicates reduced differentiation in shielding response at higher thickness levels. Furthermore, replacing high-Z Sm2O3 with MgO increases density and modifies the structural network, which improves Dr performance. For fixed thicknesses, the attenuation valleys become smooth with increasing Sm2O3 content, highlighting the critical influence of composition and density variation on radiation interaction performance. At elevated thicknesses, these valleys tend to vanish completely, resulting in a linearized response curve. This observation suggests the appearance of a near-linear relationship of thickness with Dr, reinforcing the dominant role of physical and chemical parameters in modulating shielding efficiency. The present results of radiation shielding are compared with the existing literature on Sm2O3-containing samples in Table 12.
 |
| | Fig. 22 Dr variation for the prepared samples as a function of gamma photon energy. | |
 |
| | Fig. 23 Dr variation for the prepared samples as a function of gamma photon energy at different thicknesses. | |
Table 12 Comparison of radiation shielding parameters of Sm2O3-doped or rare-earth doped glass systems at 0.662 MeV
| Glass ID ↓ |
MAC (cm2 g−1) |
HVL (cm) |
Zeff |
Ref. |
| MS-0 |
0.63 |
0.46 |
10.40 |
Present work |
| MS-0.5 |
0.69 |
0.42 |
11.18 |
Present work |
| MS-1 |
0.74 |
0.38 |
11.96 |
Present work |
| MS-1.5 |
0.80 |
0.34 |
12.71 |
Present work |
| Sm2O3 containing borate |
7.4–8.1 |
0.040–0.046 |
32–35 |
68 |
| PbO−B2O3−SiO2−Sm2O3 |
8.35 |
0.041 |
33.8 |
65 |
| La2O3/Nd2O3 containing borate |
6.72 |
0.052 |
30–32 |
69 |
| Sm2O3 contain borate |
0.19–1.44 |
1.02–2.72 |
— |
70 |
3.6 Study of oscillator strengths and Judd–Ofelt intensity parameters
Fig. 24 represents the absorbance spectra of the prepared glass samples in the wavelength range of 250–1250 nm. The absorbance bands are observed at 402, 475, 941, 1075, and 1219 nm wavelengths, which are assigned to 6H5/2 → 6P3/2, 6H5/2 → 4I11/2, 6H5/2 → 6F11/2, 6H5/2 → 6F9/2 and 6H5/2 → 6F7/2, respectively. The nephelauxetic ratio (Nr) is a vital parameter to identify the nature of the bond between metal and ligands. It is obtained by dividing the examined wavenumber of the as-quenched glass (νc) by the wavenumber of the concerned transition of an aqua ion (νa). The energies of concerned transitions (νc), average nephelauxetic ratios  , and bonding parameters (Bp) corresponding to the different transitions are determined and presented in Table 13. The relationship between
, and bonding parameters (Bp) corresponding to the different transitions are determined and presented in Table 13. The relationship between  and the bonding parameter (Bp) can be written as follows:
and the bonding parameter (Bp) can be written as follows:| |
 | (32) |
 |
| | Fig. 24 UV-Visible NIR absorption spectra of Sm2O3-containing magnesium borosilicate glasses. | |
Table 13 The energy of concerned transition (νc), average nephelauxetic ratios  , and bonding parameters (Bp) of as-prepared glasses
, and bonding parameters (Bp) of as-prepared glasses
| Transition ↓ |
νc (cm−1) |
| MS-0.5 |
MS-1 |
MS-1.5 |
| 6H5/2 → 6P3/2 |
24875.62 |
24937.65 |
24937.65 |
| 6H5/2 → 4I11/2 |
21052.63 |
21097.04 |
21052.63 |
| 6H5/2 → 6F11/2 |
10626.99 |
10626.99 |
10638.29 |
| 6H5/2 → 6F9/2 |
9302.32 |
9310.98 |
9319.66 |
| 6H5/2 → 6F7/2 |
8203.44 |
8196.72 |
8196.72 |
| |
 |
1.010 |
1.011 |
1.011 |
| Bp |
−0.991 |
−1.083 |
−1.082 |
The average nephelauxetic ratios are greater than 1  , and bonding parameters are less than zero (Bp < 0), which signifies more inter-electronic repulsions in metal–ligand bonds. Thus, it confirms the presence of ionic bonding between the Sm3+ ions and ligands in the prepared glasses.
, and bonding parameters are less than zero (Bp < 0), which signifies more inter-electronic repulsions in metal–ligand bonds. Thus, it confirms the presence of ionic bonding between the Sm3+ ions and ligands in the prepared glasses.
The absorption ability is measured in terms of oscillator strength. The experimental oscillator strength (fexp) and calculated oscillator strength (fcal) are determined using the 6H5/2 → 6P3/2, 6H5/2 → 4I11/2, 6H5/2 → 6F11/2, 6H5/2 → 6F9/2 and 6H5/2 → 6F7/2 transitions. The experimental oscillator strength can be calculated as follows:
| |
 | (33) |
where
A = log(
I/
I0) is the absorbance at wavenumber
![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif)
,
S is the rare-earth ion concentration in mol l
−1 and
R is the thickness of the material. The calculated oscillator strength of transition between the lowest level
ψJ and the excited level

of the samarium ion is determined using the following equation:
78| |
 | (34) |
where
ν denotes the wavenumber of a transition,
h is the Planck's constant,
p is the refractive index, and
Ωα (
α = 2, 4 and 6) are Judd–Ofelt (J–O) parameters. These parameters can be obtained by standard least-squares fitting of
fexp and
fcal.
J and
J′ are the total angular momentum of the ground state and upper state, and

signifies the doubly reduced matrix element of a unit tensor operator. The matrix elements are taken from the literature.
The obtained values of fexp and fcal are depicted in Table 14. It is revealed that the experimental oscillator strength (fexp) and the calculated oscillator strength (fcal) show approximately close agreement with each other, which is further confirmed by the root-mean-square value. The root-mean-square value (δrms) is determined by the following relation:79
| |
 | (35) |
The smaller value of
δrms represents the good fitting between the experimental oscillator strength (
fexp) and the calculated oscillator strength (
fcal). Therefore, it is assigned to the validity of Judd–Ofelt parameters of the prepared glass materials.
Table 14 experimental oscillator strength (fexp × 10−6), calculated oscillator strength (fcal × 10−6) and root-mean-square value (δrms) of as-prepared glasses
| Transition ↓ |
MS-0.5 |
MS-1 |
MS-1.5 |
| fexp |
fcal |
fexp |
fcal |
fexp |
fcal |
| 6H5/2 → 6P3/2 |
0.467 |
0.789 |
2.804 |
2.214 |
0.137 |
0.931 |
| 6H5/2 → 4I11/2 |
0.048 |
0.012 |
0.663 |
0.113 |
0.858 |
0.012 |
| 6H5/2 → 6F11/2 |
1.792 |
0.943 |
3.421 |
3.385 |
0.392 |
0.985 |
| 6H5/2 → 6F9/2 |
3.971 |
5.497 |
7.471 |
10.354 |
3.921 |
5.756 |
| 6H5/2 → 6F7/2 |
7.268 |
6.286 |
10.894 |
9.336 |
7.916 |
6.587 |
| |
| δrms (×10−6) |
0.91 |
1.51 |
1.17 |
Brian R. Judd80 and George S. Ofelt81 introduced a theory to understand the glass structure and radiative behavior of rare-earth-ion-activated materials. They examined Ω2, Ω4, and Ω6 parameters, which are known as Judd–Ofelt parameters. The Ω2 parameter confirms the symmetry as well as the covalent nature of the Sm–O bond. The Ω4 and Ω6 parameters are associated with the rigidity and viscosity of a material, respectively. The obtained values of Judd–Ofelt parameters are listed in Table 14. It is noticed that Ω4 attains a higher value than other parameters in all the prepared glass samples, which signifies the existence of better rigidity in all the prepared glasses. In the present report, the synthesized glasses follow the Ω4 > Ω2 > Ω6 trend, which signifies the excellent rigidity and lack of covalent nature.17
Further, the spectroscopic quality factor (χ) is calculated by dividing Ω4 by the Ω6 parameter. The obtained quality factors of MS-0.5, MS-1, and MS-1.5 glass samples are 1.10, 1.64, and 1.68, respectively. The present results are compared with the existing literature on Sm3+-doped samples (Table 15). It is observed that the MS-1.5 glass sample has a higher spectroscopic quality factor than others, which leads to a better ability in stimulated emission.
Table 15 The values of Ω2, Ω4 and Ω6 (10−20 cm2), trend of Judd–Ofelt (J–O) parameters, and spectroscopic quality factor (χ) of as-prepared glasses
| Glass ID ↓ |
Ω2 |
Ω4 |
Ω6 |
Trend |
χ |
Ref. |
| MS-0.5 |
3.72 |
3.93 |
3.58 |
Ω4 > Ω2 > Ω6 |
1.10 |
Present work |
| MS-1 |
3.98 |
4.02 |
2.45 |
Ω4 > Ω2 > Ω6 |
1.64 |
Present work |
| MS-1.5 |
4.10 |
4.69 |
2.80 |
Ω4 > Ω2 > Ω6 |
1.68 |
Present work |
| FTe1 |
1.33 |
1.40 |
1.14 |
Ω4 > Ω2 > Ω6 |
1.22 |
71 |
| Te1 |
1.16 |
1.28 |
1.03 |
Ω4 > Ω2 > Ω6 |
1.24 |
71 |
| Sm3+ doped borate |
2.39 |
4.71 |
6.53 |
Ω4 > Ω6 > Ω2 |
0.72 |
70 |
| Sm3+ doped fluorophosphate |
0.98 |
1.97 |
1.45 |
Ω4 > Ω6 > Ω2 |
1.35 |
72 |
| LBTAF |
0.27 |
2.52 |
2.47 |
Ω4 > Ω6 > Ω2 |
1.02 |
73 |
| CNGS |
1.26 |
3.52 |
3.44 |
Ω4 > Ω6 > Ω2 |
1.02 |
74 |
| Sm3+ doped Li2O–B2O3 |
0.38 |
3.01 |
2.01 |
Ω4 > Ω6 > Ω2 |
1.49 |
75 |
| Sm3+:BPWLTe |
2.62 |
4.64 |
2.91 |
Ω4 > Ω6 > Ω2 |
1.59 |
76 |
| F30 |
1.66 |
2.55 |
1.56 |
Ω4 > Ω2 > Ω6 |
1.63 |
77 |
3.7 Radiative properties
The probability of a radiative transition from an excited state to a subordinate energy level is computed as follows:| |
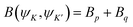 | (36) |
where Bp and Bq denote the transition probabilities associated with electric and magnetic dipole interactions, respectively. These parameters are determined using the following expressions:82| |
 | (37) |
| |
 | (38) |
The matrix elements  and
and 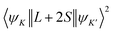 are evaluated within the intermediate coupling scheme, where ‖Uα‖ signifies the reduced matrix element of the unit tensor operator. These values are taken from the existing literature. Therefore, the total radiative transition probability (BS) for the excited state is obtained by aggregating the probabilities across all available decay channels, as follows:
are evaluated within the intermediate coupling scheme, where ‖Uα‖ signifies the reduced matrix element of the unit tensor operator. These values are taken from the existing literature. Therefore, the total radiative transition probability (BS) for the excited state is obtained by aggregating the probabilities across all available decay channels, as follows:
| |
 | (39) |
The branching ratio provides a measure of the relative intensity distribution among the diverse emission pathways originating from a given excited state. It is defined as follows:
| |
 | (40) |
Among the observed transitions, the
4G
5/2 →
6H
5/2 transition exhibits the highest branching ratio than others, suggesting its potential for laser oscillation. The experimental branching ratios (
βR) confirm the predominance of blue emission, following the hierarchical order (
6H
5/2 >
6H
7/2 >
6H
9/2) as given in
Table 16, which suggests that MS-1.5 glass is a suitable candidate for visible orange-red laser applications. Furthermore, the radiative lifetime (
τR) of the excited state is defined as being inversely proportional to the total radiative transition probability (1/
BT).
Table 16 Radiative transition probability (B), total radiative transition probability (BT), decay time (τR), branching ratio (βR) and stimulated emission cross section (σp (×10−22 cm2)) parameters of as-prepared glasses
| Glass ID ↓ |
Transitions (4G5/2→) |
λp (nm) |
Ar (s−1) |
AT (s−1) |
τR (ms) |
βR |
σp |
| MS-0.5 |
6H5/2 |
548 |
607.97 |
1633.08 |
0.61 |
0.65 |
4.28 |
| 6H7/2 |
600 |
535.01 |
0.24 |
1.06 |
| 6H9/2 |
645 |
490.09 |
0.11 |
1.03 |
| MS-1 |
6H5/2 |
548 |
619.96 |
1659.80 |
0.60 |
0.66 |
4.42 |
| 6H7/2 |
600 |
552.14 |
0.24 |
1.21 |
| 6H9/2 |
646 |
487.70 |
0.10 |
1.18 |
| MS-1.5 |
6H5/2 |
548 |
647.43 |
1723.13 |
0.58 |
0.69 |
4.89 |
| 6H7/2 |
600 |
555.14 |
0.25 |
1.52 |
| 6H9/2 |
646 |
520.54 |
0.06 |
1.44 |
The Judd–Ofelt parameters are used to evaluate radiative properties such as the branching ratio (βR), decay time (τR), and radiative and total radiative transition probabilities (Ar and AT) for the exciting level of Sm3+ ions in the prepared glass samples. The obtained values of radiative parameters are presented in Table 16. The emission at 548, 600, and 648 nm corresponds to the 4G5/2 → 6H5/2, 4G5/2 → 6H7/2, and 4G5/2 → 6H9/2 transitions, respectively. The 4G5/2 → 6H5/2 transition attains a higher radiative transition probability than other possible transitions. As Sm2O3 increases, the total radiative transition probability increases from 1633.080 to 1723.135 s−1. The decay times of MS-0.5, MS-1, and MS-1.5 glass samples are 0.61, 0.60, and 0.58 ms, respectively. The present findings signify that samarium oxide in the glass matrix plays a vital role in improving radiative emission. The present results of decay time and stimulated emission cross section are compared with the existing literature on Sm2O3-doped samples in Table 17.
Table 17 Comparison of stimulated emission cross section (σp (×10−22 cm2)) and decay time (τR) of Sm2O3-doped glass systems with existing literature
| Glass ID ↓ |
σp (4G5/2 → 6H7/2) |
τR (ms) |
Ref. |
| MS-0.5 |
1.06 |
0.61 |
Present work |
| MS-1 |
1.21 |
0.60 |
Present work |
| MS-1.5 |
1.52 |
1.68 |
Present work |
| Sm3+ doped Li2O–B2O3 |
2.95 |
0.65–1.62 |
75 |
| Sm3+:BPWLTe |
1.69 |
0.64 |
76 |
| SmWZnBTe |
5.36 |
1.17 |
74 |
The branching ratio is the most significant parameter of stimulated emission. Notably, the βR parameter has a higher value than 0.50 (50%) in all the prepared glass samples. The MS-1.5 glass has the highest value for all transitions as compared to the other glass samples. Moreover, the stimulated emission cross-section (σp) is calculated as shown in Table 16. The 4G5/2 → 6H5/2 transition has a higher value of the σp parameter compared to the other transitions. The stimulated emission cross-section increases with increasing Sm2O3 concentration. Therefore, the high Ω2, Ω4/Ω6 ratio, σp and τR values (Tables 15 and 17) of the Sm3+ doped glasses as compared to the existing literature make them suitable for laser and photonic applications.
4 Conclusion
A novel glass system with the formula 50SiO2 + 40B2O3 + (10 − x)MgO + (x)Sm2O3 (x = 0 to 1.5 mol%) was successfully synthesized via the conventional melt-quenching method. Increasing Sm2O3 doping introduced multifunctional enhancement across mechanical, thermal, optical, and radiation shielding properties of the borosilicate glasses.
• Density increased from 2.32 g cm−3 (MS-0) to 2.48 g cm−3 (MS-1.5) due to the substitution of heavier Sm3+ ions, which confirms a dense and more compact glass network.
• XRD confirmed the amorphous nature of all compositions.
• Elastic moduli such as the Young's, shear, and bulk modulus exhibited an increasing trend with Sm2O3 concentration, reflecting enhanced rigidity and mechanical integrity of the glass structure. These enhancements are associated with stronger Sm–O bonds that reinforce the borosilicate network.
• The glass transition temperature (〈Tg〉) increased from 488 °C (MS-0) to 571 °C (MS-1.5). Thermal stability (ΔT = Tc − Tg) increased from 302 °C to 331 °C, indicating better resistance to devitrification and higher structural integrity.
• The increasing trend of the Avrami constant from 1.20 to 2.76 with Sm2O3 substitution indicates that bulk crystallization dominates in the as-prepared glass samples.
• The Judd–Ofelt intensity parameters increased with increasing Sm2O3 content, indicating enhanced asymmetry and rigidity around the Sm3+ ions within the glass matrix.
• The mass attenuation coefficient (MAC) decreased significantly with increasing gamma photon energy, consistent with the prevailing photon interaction mechanisms. The MS-1 sample exhibited the highest relative photon attenuation efficiency (RPE) over a broad energy range as compared to the other samples. The exposure buildup factor (EABF) followed the trend EABF1.5 < EABF1 < EABF0.5 < EABF0. The kerma relative to air followed the order Kα(MS-0) < Kα(MS-0.5) < Kα(MS-1) < Kα(MS-1.5).
Overall, the incorporation of Sm2O3 significantly enhances the structural compactness, mechanical rigidity, radiation attenuation capability, and luminescence efficiency of SiO2–B2O3–MgO glasses. Among all compositions, the MS-1.5 sample consistently exhibited superior performance across most evaluated parameters, indicating its potential for multifunctional applications in photonics and radiation shielding.
5 Novelty of the study
The present research establishes a systematic investigation into the multifunctional development of rare-earth-modified borosilicate glasses. Precisely, it explores the Sm2O3 incorporation (0 to 1.5 mol%) effect on a tailored 50SiO2 + 40B2O3 + (10 − x)MgO + (x)Sm2O3 host matrix. The novelty of the present study is as follows:
• Comprehensive structure–property correlation: the present study provides a detailed understanding of the effect of Sm3+ ions on structural and active radiative/non-radiative centers.
• Advanced compositional optimization: the systematic distribution of Sm2O3 modifies the structure, thermal stability, and optical properties. This approach permits the fine-tuning of material properties while strictly preserving the amorphous integrity and phase homogeneity of the glass matrix.
• Synergistic integration of radiation shielding and photonic properties: the present research reports a correlation between photoluminescence efficiency and gamma-radiation attenuation with an increase in Sm2O3 content at the cost of MgO. The findings establish that MS-1.5 glass is a high-performance, dual-functional candidate for advanced solid-state lighting and radiation protection in nuclear and aerospace environments.
• Sm2O3-mediated structural reconstruction: Minimal Sm2O3 incorporation affects substantial network polymerization and structural rigidity. This transition is driven by the higher field strength of Sm3+ ions, which leads to an increase in the hardness as well as the elastic modulus. This structural evolution is validated through different theoretical models, i.e., Makishima–Mackenzie, Rocherulle and the bond compression model.
Conflicts of interest
No conflicts of interest exaist to disclose.
Data availability
Data will be made available on request.
Acknowledgements
This research did not receive dedicated funding from public, commercial, or non-profit organisations. Financial assistance for this project was provided by the Thapar Institute of Engineering and Technology (TIET), Patiala. The authors would like to express their gratitude to DST-TIET for granting access to characterisation facilities through the FIST-II scheme.
References
- A. Madheshiya, C. Gautam and S. Upadhyay, Preparation, optical and electrical properties of bismuth substituted lead titanate borosilicate glass and glass ceramics, J. Non-Cryst. Solids, 2018, 502, 118–127 CrossRef CAS.
- S. Kumar, K. Singh and D. Kumar, SiO2/B2O3 glass formers effect on transparency and mechanical properties of soda-lime borosilicate glasses for automobile applications, J. Non-Cryst. Solids, 2023, 618, 122530 CrossRef CAS.
- M. H. A. Mhareb, M. Alqahtani, Y. Alajerami, F. Alshahri, M. Sayyed, K. Mahmoud, N. Saleh, N. Alonizan, M. Al-Buriahi and K. M. Kaky, Ionizing radiation shielding features for titanium borosilicate glass modified with different concentrations of barium oxide, Mater. Chem. Phys., 2021, 272, 125047 CrossRef CAS.
- M. Mhareb, M. A. Morsy, H. Almarri, M. Sayyed, I. Alrammah, N. Alonizan, Y. Alajerami, Q. Drmosh, M. K. Hamad and G. N. Makhadmeh, et al., Gamma-ray induced effect on the structural and optical properties and durability of neodymium-doped zinc-bismuth-borotellurite glasses and glass ceramics, Opt. Mater., 2023, 137, 113572 CrossRef CAS.
- K. Wu, F. Wang, Q. Liao, H. Zhu, D. Liu and Y. Zhu, Synthesis of pyrochlore-borosilicate glass-ceramics for immobilization of high-level nuclear waste, Ceram. Int., 2020, 46(5), 6085–6094 CrossRef CAS.
- M. Y. Hanfi, A. K. Sakr, A. Ismail, B. M. Atia, M. S. Alqahtani and K. Mahmoud, Physical characterization and radiation shielding features of B2O3As2O3 glass ceramic, Nucl. Eng. Technol., 2023, 55(1), 278–284 CrossRef CAS.
- H. A. Fakhouri, B. Abughazaleh and M. Sayyed, Comparative evaluation of cubic spline, akima, and PCHIP interpolation methods for predicting radiation shielding parameters of heavy metal oxide glasses: numerical approximation for lead borate-based glasses using cubic splines, piecewise cubic hermite interpolating polynomial and akima interpolation, Nucl. Eng. Technol., 2026, 104232 CrossRef CAS.
- H. Al-Ghamdi, N. Alfryyan, N. A. Alsaif, A. Abdelghany, K. Higazy, A. Abouhaswa, Y. S. Rammah, K. M. Kaky, A. J. Kadhim and F. El-Agawany, Impact of TiO2-doped bismuth-boro-tellurite glasses: fabrication, physical and optical properties, and γ-ray protection competence for optical and radiation shielding applications, J. Electron. Mater., 2025, 54(2), 1432–1443 Search PubMed.
- M. R. Ahmed, N. A. Alsaif, N. Siddiqui, A. S. Prasad, M. Srinivas, S. K. Ahmmad, K. M. Kaky, A. J. Kadhim and Y. Rammah, Physical, thermal properties, FTIR and Raman spectroscopies as well as γ-ray attenuation capacity of borate glasses doped with Mn2+ ions: role of CaO/Al2O3 substitution, Opt. Mater., 2025, 158, 116461 CrossRef CAS.
- N. A. Alsaif, N. Alfryyan, H. Al-Ghamdi, A. Abdelghany, A. Abouhaswa, A. J. Kadhim, K. M. Kaky and Y. Rammah, Fabrication, structural, physical, optical characteristics and γ-ray attenuation capacity of borosilicate glasses doped with Gd2O3, Sci. Rep., 2024, 14(1), 30587 CrossRef CAS PubMed.
- M. Sayyed, M. Mhareb, K. M. Kaky, K. Mahmoud, A. J. Kadhim and Y. Maghrbi, Developing GTB glass system doped holmium oxide for radiation absorption and optical applications, J. Sci.: Adv. Mater. Devices, 2025, 10(2), 100872 CAS.
- A. Acikgoz, I. Izguden, Y. Tasgin, D. Yilmaz, G. Demircan, S. Kalecik and B. Aktas, Influence of praseodymium oxide on the structural, mechanical and photon, charged particles, and neutron shielding properties of alumina borate glass, Ceram. Int., 2024, 50(19), 34573–34584 CrossRef CAS.
- M. Sayyed, M. Mhareb, M. K. Hamad, A. J. Kadhim, H. Alsafi, K. Mahmoud and K. M. Kaky, Effects of MoO3 on the structural, physical, mechanical, optical, and ionizing shielding of borate-germanate-telluride
glass system, Ceram. Int., 2024, 50(22), 46008–46017 CrossRef CAS.
- M. Sayyed, M. Mhareb, K. M. Kaky, K. Mahmoud and A. J. Kadhim, A new approach toward enhancing the gamma-ray shielding efficiency: Zn ions effects on synthesized B2O3–PbO–BaO toward gamma-ray protection applications, Ann. Nucl. Energy, 2025, 210, 110860 CrossRef CAS.
- R. I. Mahdi, M. Y. Hanfi, M. Mhareb, M. Sayyed, A. J. Kadhim and K. M. Kaky, Transparent heavy-metal glass-ceramics reinforced with nano-PbO for next-generation X-and γ-ray shielding applications, Mater. Res. Bull., 2025, 113808 Search PubMed.
- M. Mhareb, M. K. Hamad, R. I. Mahdi, M. Sayyed, H. Mansour, A. J. Kadhim and K. M. Kaky, Tailoring the optical, mechanical, and gamma-ray-attenuation performance of GTB glasses by doping nano rare-earth (Gd, Yb, Tm), Radiat. Phys. Chem., 2025, 237, 113019 CrossRef CAS.
- V. Gupta, S. Kumar, S. Arya, R. Mishra and K. Singh, et al., Blue-to-white light generation using rare-earth dopants and magnesium phosphate glass host interaction, Opt. Mater., 2026, 174, 117898 CrossRef CAS.
- M. Sayyed, M. Mhareb and K. M. Kaky, Characterization of mechanical and radiation shielding features of borosilicate glasses doped with MoO3, Silicon, 2024, 16(5), 1955–1965 CrossRef CAS.
- J. Thumma and R. K. Guntu, Design and evaluation of TiO2-enhanced phosphate glasses for dual-functionality: photonic applications and radiation safety, Phys. B, 2025, 417735 CrossRef CAS.
- M. A. Imheidat, M. KhHamad, K. Naseer, M. Sayyed, N. Dwaikat, K. Cornish, Y. Alajerami, M. Alqahtani and M. Mhareb, Radiation shielding, mechanical, optical, and structural properties for tellurite glass samples, Optik, 2022, 268, 169774 CrossRef CAS.
- P. Kaur, D. Singh and T. Singh, Heavy metal oxide glasses as gamma rays shielding material, Nucl. Eng. Des., 2016, 307, 364–376 CrossRef CAS.
- A. Mostafa, H. M. Zakaly, S. Al-Ghamdi, S. A. Issa, M. Al-Zaibani, R. Ramadan and E. El Agammy, PbO–Sb2O3–B2O3–CuO glassy system: evaluation of optical, gamma and neutron shielding properties, Mater. Chem. Phys., 2021, 258, 123937 CrossRef CAS.
- M. Sayyed, M. Mhareb, M. K. Hamad, A. J. Kadhim, K. M. Kaky and Y. Maghrbi, Structural, physical, optical, mechanical, and radiation shielding investigation of tantalum pentoxide doped germanate-tellurite–borate based glass system, Results Phys., 2025, 75, 108356 CrossRef.
- M. Sayyed, K. M. Kaky, M. Mhareb, A. Al-keisy and S. Baki, Impact of cuo on TeO2–GeO2–ZnO–Al2O3–MgO glass system for ionizing shielding applications, J. Mater. Sci.: Mater. Electron., 2023, 34(36), 2277 CrossRef CAS.
- M. Hamad, N. Dwaikat, M. Mhareb, M. Sayyed, R. Hamad, Y. Alajerami, M. Almessiere, G. Saleh, A. H. Alomari and K. Ziq, Influence of erbium on structural, and charged particles, photons, and neutrons shielding properties of Ba1−xErxSnO3 perovskite ceramics, J. Rare Earths, 2024, 42(4), 724–732 CrossRef CAS.
- A. T. Patel and A. Pratap, Study of kinetics of glass transition of metallic glasses, J. Therm. Anal. Calorim., 2012, 110(2), 567–571 CrossRef CAS.
- A. M. Al-Baradi, E. A. Wahab and K. S. Shaaban, Preparation and characteristics of B2O3–SiO2–Bi2O3–TiO2–Y2O3 glasses and glass-ceramics, Silicon, 2022, 14(10), 5277–5287 CrossRef CAS.
- M. Kaur, G. Kaur, O. Pandey, K. Singh and V. Kumar, Influence of CaO/MgO ratio on the crystallization kinetics and interfacial compatibility with crofer 22APU and YSZ of strontium based alumino-borosilicate glasses for SOFC applications, Int. J. Hydrogen Energy, 2017, 42(25), 16244–16257 CrossRef CAS.
- S. Khan, S. Kumar, N. Jast, K. Abida, K. Singh and S. Singh, Luminescence, mechanical and non-isothermal crystallization kinetics properties of MnO-doped borosilicate glasses for photonics applications, J. Solid State Chem., 2025, 125608 Search PubMed.
- R. K. Guntu, P. Ashok, K. Sivaram, P. B. Shetty, S. Babu and M. Israr, Investigations on thermoluminescence, photoluminescence and radiation shielding properties of Dy2O3 doped Li2O–Al2O3–SiO2–Y2O3 glasses for thermoluminescent dosimeter applications, Appl. Phys. A, 2025, 131(9), 701 CrossRef CAS.
- A. Makishima and J. D. Mackenzie, Direct calculation of Young's modulus of glass, J. Non-Cryst. Solids, 1973, 12(1), 35–45 CrossRef CAS.
- A. A. El-Rehim, H. Zahran, I. Yahia, S. A. Makhlouf and K. S. Shaaban, Radiation, crystallization, and physical properties of cadmium borate glasses, Silicon, 2021, 13(7), 2289–2307 CrossRef CAS.
- H. Doweidar, Consideration of the boron oxide anomaly, J. Mater. Sci., 1990, 25(1), 253–258 CrossRef CAS.
- B. Bridge and A. Higazy, A model of the compositional dependence of the elastic moduli of polycomponent oxide glasses, Phys. Chem. Glasses, 1986, 27(1), 1–14 CAS.
- B. Bridge, N. Patel and D. Waters, On the elastic constants and structure of the pure inorganic oxide glasses, Phys. Status Solidi A, 1983, 77(2), 655–668 CrossRef CAS.
- O. H. Abd-Elkader, M. Nasrallah, M. Nasrallah, S. Aleya, M. O. Abdelkader and A. Saleh, Rapid fabrication, magnetic, and radiation shielding characteristics of NiFe2O4 nanoparticles, Opt. Mater. Express, 2024, 14(5), 1170–1185 CrossRef CAS.
- A. Saleh, Comparative shielding features for X/gamma-rays, fast and thermal neutrons of some gadolinium silicoborate glasses, Prog. Nucl. Energy, 2022, 154, 104482 CrossRef CAS.
- S. A. Saleh, S. Tajudin, M. Algethami, K. T. Osman, K. Said, A. S. Elshoukrofy and T. Y. Wais, Fatma elzahraa mansour, optimizing the integration between radiation protection, optical, and mechanical strength for advanced nuclear safety applications using tungsten-doped bismuth borate glasses, Radiat. Phys. Chem., 2026, 238, 113216 CrossRef.
- N. A. Abdelhakim, R. M. Shalaby, A. Abdelghany, M. Mitwalli, A. El-Farrash and A. Saleh, Integration between radiation shielding performance, structural evolution, and mechanical features of co-doped sodium phosphate glasses, Sci. Rep., 2026, 16, 1958 CrossRef CAS PubMed.
- A. Saleh, A. Kobyliukh, N. Basfer, S. Tajudin and U. Szeluga, Comprehensive examination of synthesis, microstructure, and radiation shielding effectiveness of multi-layered polymeric GNP-nanocomposites, Radiat. Phys. Chem., 2025, 237, 113078 CrossRef CAS.
- E. Sakar, Ö. F. Özpolat, B. Alm, M. Sayyed and M. Kurudirek, Phy-X/PSD: development of a user friendly online software for calculation of parameters relevant to radiation shielding and dosimetry, Radiat. Phys. Chem., 2020, 166, 108496 CrossRef CAS.
- N. Alharbiy, Z. Khattari, Y. Rammah and A. Saleh, Role of Al2O3, WO3, Nb2O5, and PbO on the physical, elasto-mechanical and radiation attenuation performance of borotellurite glasses, J. Mater. Sci.: Mater. Electron., 2023, 34(3), 191 CrossRef CAS.
- J. Kaur, S. Kumar, I. Mudahar and K. Singh, Tunable green-blue luminescence of Dy2O3 doped borosilicate glasses for optoelectronic devices, J. Mol. Struct., 2025, 1322, 140574 CrossRef CAS.
- L. U. Grema, S. Kumar, S. Khan, K. Singh and S. Umar, Thermo-mechanical, structural and electrical properties of Ba–La–Zn–Al-borosilicate glasses as sealants for application in SOFCs, J. Non-Cryst. Solids, 2026, 671, 123826 CrossRef CAS.
- A. Abd El-Moneim, Bond compression bulk modulus and Poisson's ratio of the polycomponent silicate glasses, Mater. Chem. Phys., 2001, 70(3), 340–343 CrossRef CAS.
- S. Kumar, S. Thakur, P. K. Jha, D. Kumar and K. Singh, Conversion of cool-to-warm orange-reddish light emitting diode by Sm2O3 addition in magnesium borosilicate glasses, J. Mol. Struct., 2024, 1312, 138432 CrossRef CAS.
- S. Khan, S. Kumar, G. Singla, K. Singh and P. K. Jha, Tuning crystallization, strength, and luminescence property of CuO modified sodium-phosphate glasses, ECS J. Solid State Sci. Technol., 2025, 14(8), 086004 CrossRef CAS.
- S. Kumar, K. Singh and D. Kumar, B2O3 anomaly effect on crystallization and mechanical properties of soda-lime silicate glasses, Mater. Today Commun., 2024, 39, 109247 CrossRef CAS.
- C. Bernard, S. Chaussedent, A. Monteil, M. Montagna, L. Zampedri and M. Ferrari, Simulation by molecular dynamics of erbium-activated silica-titania glasses, J. Sol-Gel Sci. Technol., 2003, 26(1), 925–929 CrossRef CAS.
- M. Halimah, W. Daud and H. Sidek, Elastic properties of TeO2–B2O3–Ag2O glasses, Ionics, 2010, 16(9), 807–813 CrossRef CAS.
- S. Inaba, S. Oda and K. Morinaga, Heat capacity of oxide glasses at high temperature region, J. Non-Cryst. Solids, 2003, 325(1–3), 258–266 CrossRef CAS.
- R. El-Mallawany, Tellurite glasses part 1. elastic properties, Mater. Chem. Phys., 1998, 53(2), 93–120 CrossRef CAS.
- D. Souri, Fragility, DSC and elastic moduli studies on tellurite-vanadate glasses containing molybdenum, Measurement, 2011, 44(10), 1904–1908 CrossRef.
- S. Khan, S. Kumar, S. Khan, K. Abida and K. Singh, Influence of ZrO2 on non-isothermal crystallization kinetics, structure and optical properties of calcium borosilicate glasses, Mater. Today Commun., 2024, 40, 109526 CrossRef CAS.
- S. Khan, S. Kumar, K. Abida and K. Singh, Non-isothermal crystallization kinetics, optical and structural properties of MgO-doped vanadate glasses, J. Therm. Anal. Calorim., 2024, 149(11), 5299–5311 CrossRef CAS.
- I.-C. Chen, P.-Y. Lin, T. T. Li and J.-Y. Chang, Kinetic study of the thermal crystallization behavior of hydrogenated amorphous silicon prepared by ECRCVD, ECS J. Solid State Sci. Technol., 2014, 3(5), N75 CrossRef CAS.
- H. E. Kissinger, et al., Variation of peak temperature with heating rate in differential thermal analysis, J. Res. Natl. Bur. Stand, 1956, 57(4), 217–221 CrossRef CAS.
- J. Augis and J. Bennett, Calculation of the avrami parameters for heterogeneous solid state reactions using a modification of the kissinger method, J. Therm. Anal., 1978, 13(2), 283–292 CrossRef CAS.
- A.-M. Hu, K.-M. Liang, M. Li and D.-L. Mao, Effect of nucleation temperatures and time on crystallization behavior and properties of Li2O–Al2O3–SiO2 glasses, Mater. Chem. Phys., 2006, 98(2–3), 430–433 CrossRef CAS.
- S. Danewalia, K. Singh and S. Arya, Influence of vanadium oxide on non-isothermal crystallization kinetics of zinc lithium borate glasses, J. Non-Cryst. Solids, 2021, 553, 120471 CrossRef CAS.
- R. Iordanova, E. Lefterova, I. Uzunov, Y. Dimitriev and D. Klissurski, Non-isothermal crystallization kinetics of V2O5–MoO3–Bi2O3 glasses, J. Therm. Anal. Calorim., 2002, 70(2), 393–404 CrossRef CAS.
- D. L. Sidebottom, Connecting glass-forming fragility to network topology, Front. Mater., 2019, 6, 144 CrossRef.
- O. A. Lafi and M. M. Imran, Compositional dependence of thermal stability, glass-forming ability and fragility index in some Se–Te–Sn glasses, J. Alloys Compd., 2011, 509(16), 5090–5094 CrossRef CAS.
- T. Mel'Nichenko, V. Rizak, T. Mel'Nichenko and V. Fedelesh, Parameters of the fluctuation free volume theory for glasses in the Ge–As–Se system, Glass Phys. Chem., 2004, 30(5), 406–414 CrossRef.
- M. Sayyed, M. K. Hamad and M. Mhareb, Radiation
shielding properties for a borosilicate glass: role of varying PbO, Opt. Mater., 2025, 159, 116602 CrossRef CAS.
- A. Ratep, A. Abdelaziem, M. Hanfi, K. Mahmoud and I. Kashif, Enhancing gamma-ray shielding with bismuth oxide-infused boron oxides, Opt. Quantum Electron., 2024, 56(2), 143 CrossRef CAS.
- H. Yilmaz Alan, Efficacy of barium and calcium additives in lithium silicate glasses for nuclear shielding applications, Phys. Scr., 2024, 99(7), 075314 CrossRef CAS.
- M. Sayyed, A. Bennal, M. Y. Hanfi, V. K. Bhovi and S. A. Issa, et al., Impact of Y2O3 and Sm2O3 doping on the radiation shielding properties of lead-borate glasses, Appl. Radiat. Isot., 2025, 112407 Search PubMed.
- A. H. Almuqrin, D. A. Aloraini, M. Sayyed and M. Alwehaibi, Structural and attenuation properties of rare-earth and barium oxide modified borate glasses for gamma ray protection, Open Chem., 2026, 24(1), 20250223 CrossRef CAS.
- J. Budida, C. S. Rao, N. R. Chand and R. K. Guntu, Spectral and structural tailoring of Sm2O3-doped LiF–La2O3–PbO–B2O3 glasses for advanced luminescent devices, J. Electron. Mater., 2025, 54(11), 10611–10647 CrossRef CAS.
- K. Boonin, P. Yasaka, P. Yamchumporn, N. Triamnak, S. Kothan and J. Kaewkhao, Scintillation respond and orange emission from Sm3+ ion doped tellurite and fluorotellurite glasses: a comparative study, Radiat. Phys. Chem., 2021, 189, 109754 CrossRef CAS.
- B. Burtan-Gwizdala, J. Cisowski, R. Lisiecki, K. J. Kowalska, B. Jarzabek, N. Nosidlak, M. Reben, A. M. Alshehri, K. I. Hussein and E. S. Yousef, Thermal, optical, and emission traits of Sm3+-ion-doped fluoride/chloride/oxide glass for red/orange laser fiber applications, Fibers, 2024, 12(11), 100 CrossRef CAS.
- B. Swetha, A. P. Chowdhury and M. Kokila, Sodium lanthanum boro-tellurite glasses with Sm3+ ion doping and gold nanoparticle embedding: a new glass matrix for solid state lighting devices, J. Alloys Compd., 2025, 1010, 177628 CrossRef CAS.
- S. Ghosh, J. Biswas and S. Jana, Deciphering the role of CIElab on emission mechanism of samarium (Sm3+) ions embedded boro-tellurite glasses for applications in optoelectronics devices, Phys. B, 2025, 699, 416819 CrossRef CAS.
- K. Sudhakar Reddy, Influence of samarium concentration on the luminescence characteristics of lithium borate glasses for reddish-orange emission applications, Int. J. Curr. Sci., 2025, 15(5), 355–363 Search PubMed.
- P. Aruna, S. Hajira, M. V. Ramanaiah, V. S. Reddy, K. N. Reddy and B. S. Reddy, Optical analysis of Eu3+ and Tb3+: B2O3–P2O5–WO3–Li2O–(ZnO/CdO/TeO2) glasses, Next Mater., 2025, 8, 100703 CrossRef CAS.
- K. Bansal, N. K. Mishra, I. Abdullahi, P. J. Singh, M. Tyagi and S. Singh, Studies of luminescence traits and judd-ofelt analysis of Sm3+ activated oxyfluoride glasses, Opt. Mater., 2024, 147, 114579 CrossRef CAS.
- S. Mahamuda, F. Syed, C. B. A. Devi, K. Swapna, M. Prasad, M. Venkateswarlu and A. Rao, Spectral characterization of Dy3+ ions doped phosphate glasses for yellow laser applications, J. Non-Cryst. Solids, 2021, 555, 120538 CrossRef CAS.
- A. Florez, V. Jerez and M. Florez, Optical transitions probabilities of Dy3+ ions in fluoroindate glass, J. Alloys Compd., 2000, 303, 355–359 CrossRef.
- B. R. Judd, Optical absorption intensities of rare-earth ions, Phys. Rev., 1962, 127(3), 750 CrossRef CAS.
- G. Ofelt, Intensities of crystal spectra of rare-earth ions, J. Chem. Phys., 1962, 37(3), 511–520 CrossRef CAS.
- J. S. Kumar, K. Pavani, A. M. Babu, N. K. Giri, S. Rai and L. R. Moorthy, Fluorescence characteristics of Dy3+ ions in calcium fluoroborate glasses, J. Lumin., 2010, 130(10), 1916–1923 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article d and
A. Salehe
d and
A. Salehe


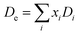
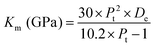
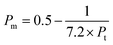
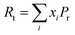

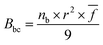
![[f with combining macron]](https://www.rsc.org/images/entities/i_char_0066_0304.gif) denotes the average stretching force constant, and nb signifies the number of network bonds per unit volume of the glass. It is defined as follows:
denotes the average stretching force constant, and nb signifies the number of network bonds per unit volume of the glass. It is defined as follows:




 is the total number of cations per glass formula unit. Furthermore, the average atomic ring size (L) for a three-dimensional network structure can be expressed as:35
is the total number of cations per glass formula unit. Furthermore, the average atomic ring size (L) for a three-dimensional network structure can be expressed as:35
 represents the bond-bending force constant and is assumed to be proportional to the stretching force constant
represents the bond-bending force constant and is assumed to be proportional to the stretching force constant ![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) for the first approximation, and Be denotes the experimental bulk modulus. The stretching force constant
for the first approximation, and Be denotes the experimental bulk modulus. The stretching force constant ![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) of the glass network is expressed as
of the glass network is expressed as


![[f with combining macron]](https://www.rsc.org/images/entities/i_char_0066_0304.gif) (N m−1)
(N m−1)![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) ), number of network bonds per unit volume (nb), average cross-link density
), number of network bonds per unit volume (nb), average cross-link density  and Poisson's ratio (σcal) for the present glass system
and Poisson's ratio (σcal) for the present glass system



















 , and bonding parameters (Bp) corresponding to the different transitions are determined and presented in Table 13. The relationship between
, and bonding parameters (Bp) corresponding to the different transitions are determined and presented in Table 13. The relationship between  and the bonding parameter (Bp) can be written as follows:
and the bonding parameter (Bp) can be written as follows:
 , and bonding parameters (Bp) of as-prepared glasses
, and bonding parameters (Bp) of as-prepared glasses
 , and bonding parameters are less than zero (Bp < 0), which signifies more inter-electronic repulsions in metal–ligand bonds. Thus, it confirms the presence of ionic bonding between the Sm3+ ions and ligands in the prepared glasses.
, and bonding parameters are less than zero (Bp < 0), which signifies more inter-electronic repulsions in metal–ligand bonds. Thus, it confirms the presence of ionic bonding between the Sm3+ ions and ligands in the prepared glasses.
![[small nu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0ce.gif) , S is the rare-earth ion concentration in mol l−1 and R is the thickness of the material. The calculated oscillator strength of transition between the lowest level ψJ and the excited level
, S is the rare-earth ion concentration in mol l−1 and R is the thickness of the material. The calculated oscillator strength of transition between the lowest level ψJ and the excited level  of the samarium ion is determined using the following equation:78
of the samarium ion is determined using the following equation:78
 signifies the doubly reduced matrix element of a unit tensor operator. The matrix elements are taken from the literature.
signifies the doubly reduced matrix element of a unit tensor operator. The matrix elements are taken from the literature.

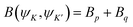


 and
and 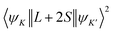 are evaluated within the intermediate coupling scheme, where ‖Uα‖ signifies the reduced matrix element of the unit tensor operator. These values are taken from the existing literature. Therefore, the total radiative transition probability (BS) for the excited state is obtained by aggregating the probabilities across all available decay channels, as follows:
are evaluated within the intermediate coupling scheme, where ‖Uα‖ signifies the reduced matrix element of the unit tensor operator. These values are taken from the existing literature. Therefore, the total radiative transition probability (BS) for the excited state is obtained by aggregating the probabilities across all available decay channels, as follows:
















