Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Unveiling the oxygen adsorption potential of ZIFs: investigating the impact of organic ligands, metal centres, and synthesis methods
Hamidreza
Mahdavi
 *ab,
Leena
Melag
c,
Abdollah
Khosravanian
a,
Joydip
Mondal
*ab,
Leena
Melag
c,
Abdollah
Khosravanian
a,
Joydip
Mondal
 c,
Zongli
Xie
c,
Zongli
Xie
 *b,
Benny D.
Freeman
*ad and
Matthew R.
Hill
*b,
Benny D.
Freeman
*ad and
Matthew R.
Hill
 *e
*e
aDepartment of Chemical and Biological Engineering, Monash University, Clayton, VIC 3800, Australia. E-mail: benny.freeman@monash.edu; hamidreza.mahdavi1@monash.edu
bCSIRO, Manufacturing, Private Bag 10, Clayton South, VIC 3169, Australia. E-mail: zongli.xie@csiro.au
cCSIRO, Mineral Resources, Private Bag 10, Clayton South, VIC 3169, Australia
dJohn J. McKetta Jr. Department of Chemical Engineering, The University of Texas at Austin, 2501 Speedway, Austin, TX 78712, USA. E-mail: benny.freeman@utexas.edu
eDepartment of Materials Science and Engineering, Monash University, Clayton, VIC 3800, Australia. E-mail: matthew.hill@monash.edu
First published on 17th April 2026
Abstract
Zeolitic imidazolate frameworks (ZIFs) have emerged as promising candidates for high O2 adsorption capacity, O2/N2 selectivity and cyclability due to their tunable chemical and structural properties governed by the selection of organic ligands, metal centres and synthesis methods. Therefore, this research engineers ZIF-7, ZIF-8, ZIF-62 and their mixed-metal derivatives containing cobalt (Co) or iron (Fe) (ZIF-8-derived Fe-mIM where applicable) to improve O2 adsorption. Because performance metrics trade off, no single ZIF maximised all objectives, and application-specific leads were identified. The highest O2 capacity was delivered by ZIF-8(Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10)-MS, reaching 0.35 mmol g−1 with selectivity of 2.01, and 9.9% decrease over 3 cycles. The greatest selectivity was achieved by ZIF-8-derived Fe-mIM material (Zn
10)-MS, reaching 0.35 mmol g−1 with selectivity of 2.01, and 9.9% decrease over 3 cycles. The greatest selectivity was achieved by ZIF-8-derived Fe-mIM material (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 0
Fe = 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100)-MS, giving selectivity of 2.60 at a low adsorption of 0.09 mmol g−1 and 9.6% loss over 3 cycles. The most robust 3-cycle performance was observed for ZIF-8-US, which retained capacity with 0.5% loss while providing 0.26 mmol g−1 and selectivity of 1.06. For a balanced generalist over 3 cycles, ZIF-62-US offered the strongest overall combination, providing 0.34 mmol g−1, selectivity of 1.01, and 1.3% loss. These results underscore the delicate balance among O2 adsorption performance with structural integrity, coordination chemistry, adsorption characteristics, and cyclability in ZIFs shaped by organic ligand, metal centres, and synthesis method choices.
100)-MS, giving selectivity of 2.60 at a low adsorption of 0.09 mmol g−1 and 9.6% loss over 3 cycles. The most robust 3-cycle performance was observed for ZIF-8-US, which retained capacity with 0.5% loss while providing 0.26 mmol g−1 and selectivity of 1.06. For a balanced generalist over 3 cycles, ZIF-62-US offered the strongest overall combination, providing 0.34 mmol g−1, selectivity of 1.01, and 1.3% loss. These results underscore the delicate balance among O2 adsorption performance with structural integrity, coordination chemistry, adsorption characteristics, and cyclability in ZIFs shaped by organic ligand, metal centres, and synthesis method choices.
Introduction
Oxygen (O2) plays a crucial role in various industrial and environmental applications.1,2 Therefore, the efficient capture, storage, and separation of O2 from mixed gas streams has attracted significant interest from both academic and industrial sectors.3–5 The development of advanced adsorbent materials that are capable of selectively adsorbing O2 has the potential to considerably improve process efficiencies and decrease energy consumption.6–9 For example, zeolites are scaffolds for gas molecular sieving,10,11 due to their cost-effective production,12 exceptional thermal and chemical stability,13 and ordered microporous structures.14 However, their rigid frameworks and limited tunability limit their potential.15,16 Moreover, many zeolites are N2-selective because N2 typically interacts more strongly with charged sites than O2, making O2-selective adsorption more challenging.17 These restrictions drive exploration of materials that maintain zeolite-like stability while providing enhanced structural and chemical flexibility.Zeolitic imidazolate frameworks (ZIFs), a Zn-based subclass of metal–organic frameworks (MOFs), can efficiently integrate the thermal and chemical stability of zeolites with the tunability of MOFs, enabling precise control over pore size, shape, and surface chemistry.18,19 This tunability arises from flexible organic ligand design and the incorporation of bimetallic centres into the framework via various synthesis methods, resulting in versatile structures customised for targeted adsorption and separation applications.20–26 These characteristics position ZIFs as viable candidates for efficient O2-selective processes.27 For example, O2 adsorption on ZIF-8 shows a distinct gate-opening mechanism, indicating adsorption-induced framework flexibility linked to organic ligand motion.27 Neutron total scattering combined with Empirical Potential Structure Refinement (EPSR) modelling further indicates that following gate opening, the additional O2 is mainly accommodated by redistribution towards pore aperture regimes.28 In another study, multigas (N2/O2/Ar/CO) adsorption measurements reveal that deformation and multistep adsorption in ZIF-8 are gas-dependent, highlighting the importance of framework flexibility in adsorption behaviour.29 However, a fundamental question remains: to what extent do organic ligand identity, metal composition, and synthesis method each govern O2 separation performance?
The organic ligand in ZIFs plays a major role in defining the framework's structure, stability, and adsorption characteristics.30,31 Organic ligand selection adjusts the pore size, shape, and surface chemistry, thereby regulating O2–framework interactions and enabling O2-selective adsorption.32–36 On the other hand, bimetallic ZIFs introduce synergistic metal–metal interactions that improve adsorption capacity, selectivity, and stability compared to monometallic systems.37–40 Transition metals with unpaired d-electrons such as cobalt (Co) and iron (Fe), often demonstrate strong O2 chemisorption that leads to enhanced adsorption and selectivity.1,36,41 Adjusting the relative metal content provides an extra parameter to optimise performance for certain applications.42 Finally, the synthesis method controls crystallinity, porosity, and surface chemistry, and thus O2 adsorption, with each method offering distinct property/scalability trade-offs.43,44 A variety of synthesis methods are available, including magnetic stirring (MS), overhead stirring (OS),33,45 ultrasonication (US),46 a combination of OS and US (OS & US),45,46 ball milling (BM),47 and microwave synthesis (MW).48 Collectively, these three factors (organic ligand, metal, and synthesis) provide a rational design space for optimising O2 separation in ZIFs.
To date, no systematic comparison has been reported that identifies the individual effects of organic ligand chemistry, mono- versus bimetallic centre, and synthesis route while maintaining a constant framework topology within each framework series. Therefore, the field lacks clear guidance on the selection of the optimal composition and scalable strategy that yields the most effective O2-selective material. This study addresses this gap by elucidating how organic ligand chemistry and metal composition govern O2 adsorption, selectivity, and cyclability under a controlled synthesis, and evaluating synthesis methods on the best-performing compositions. Additionally, understanding the relationship between material properties and synthesis conditions provides essential insights for the industrial-scale production of ZIFs. Therefore, ZIF-7, ZIF-8, and ZIF-62 synthesised using the MS method were used to investigate the effects of organic ligands and metal centres on O2 adsorption capacity, selectivity, and structural stability. These three ZIFs exhibit diverse organic ligand chemistries and flexibility/aperture regimes, which serve as a controlled contrast set for assessing the effects of organic ligand identity and framework response on O2 adsorption, O2/N2 selectivity, and cycle stability. This selection was not only based on maximising BET surface area or pre-selecting materials that were known to be the most O2-selective a priori, but rather on enabling a controlled comparison of ligand identity/flexibility and metal-centre chemistry relevant to O2/N2 separation. Additionally, they were selected as widely used benchmark frameworks that are easily synthesised and modified via scalable methods. The primary metal ion in all three ZIFs is zinc (Zn), but their organic ligand selection varies. ZIF-7 utilises benzimidazole (bIM), ZIF-8 employs 2-methylimidazole (mIM), and ZIF-62 uses a combination of imidazole (IM) and bIM. Furthermore, mixed-metal derivatives of ZIF-8 incorporating Co and Fe (ZIF-8-derived Fe-mIM where applicable) were investigated, using different molar ratios of Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co and Zn
Co and Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe to assess the influence of bimetallic systems on O2 adsorption performance. The research extends to bimetallic ZIF-7 and ZIF-62 systems with Zn
Fe to assess the influence of bimetallic systems on O2 adsorption performance. The research extends to bimetallic ZIF-7 and ZIF-62 systems with Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co and Zn
Co and Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe mol% of 90
Fe mol% of 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, and similar assessments were carried out. Finally, to evaluate scalability and processability, a variety of synthesis methods including the MS, OS, US, OS & US, BM, and MW methods were examined on a selected group of candidates.
10, and similar assessments were carried out. Finally, to evaluate scalability and processability, a variety of synthesis methods including the MS, OS, US, OS & US, BM, and MW methods were examined on a selected group of candidates.
Experimental
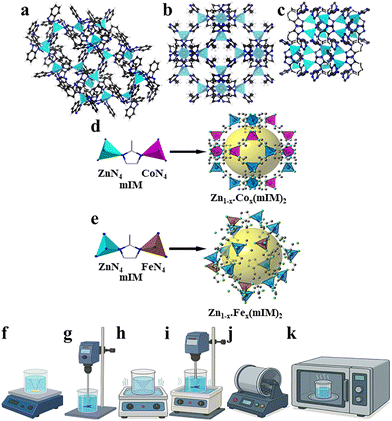
The synthesis of ZIF-7 (Zn(bIm)2), ZIF-8 (Zn(mIM)2), and ZIF-62 ([Zn(IM)2−X(bIM)X] where X ≈ 0.25) (Fig. 1a–c) and their bimetallic derivatives with Co and Fe (Fig. 1d and e) was conducted using various synthesis methods, including the MS, OS, US, OS & US, BM, and MW methods (Fig. 1f–k). Initially, the impact of different organic ligands with similar metal centres, then mixed metal centres of varying compositions on crystallinity, porosity, structural integrity, and ultimately, O2 adsorption performance has been explored. The influence of various synthesis methods on these properties of the chosen samples was studied systematically. The synthesis methods followed the established procedures, as outlined in the SI, Section S2. A comprehensive characterisation was conducted to assess the structural, chemical, thermal, and morphological aspects of the synthesised materials. The methodologies utilised encompassed powder X-ray diffraction (PXRD) for crystallinity and phase identification, Fourier-transform infrared spectroscopy (FTIR) for evaluating functional group preservation, low-pressure gas adsorption for determining Brunauer–Emmett–Teller (BET) surface areas and pore size distributions (PSDs), thermogravimetric analysis (TGA) for investigating thermal stability, and scanning electron microscopy (SEM) coupled with energy-dispersive X-ray spectroscopy (EDX) for analysing the morphology and elemental composition (see SI, Sections S3–S7 for detailed methodologies and results). Gas adsorption measurements, including O2 isotherms over multiple cycles, N2 isotherms, and O2/N2 selectivity assessments, were conducted using a Micromeritics 3Flex gas adsorption analyser (see SI, Sections S8 and S9). These investigations resulted in critical insights into the gas adsorption capabilities and selectivity of ZIFs and their bimetallic variations with respect to the organic ligand and metal centre types, mixed metal centres with varied compositions, and different synthesis methods, highlighting their potential for separation applications. The naming convention used in this manuscript is as follows: ZIF-No. (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co or Fe = x
Co or Fe = x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) xx) − y, where No. can be one of 7, 8, or 62; the variable x represents the molar percentage of Zn, xx denotes the molar percentage of Co or Fe, and y indicates the synthesis method. For example, ZIF-8 (Zn
xx) − y, where No. can be one of 7, 8, or 62; the variable x represents the molar percentage of Zn, xx denotes the molar percentage of Co or Fe, and y indicates the synthesis method. For example, ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) – MS represents ZIF-8 including 90 mol% Zn and 10 mol% Co synthesised via the MS method. In this work, the term ZIF-8 is reserved for samples that retain the characteristic ZIF-8 PXRD reflections; samples that lose long-range order (Fe-rich) are denoted ZIF-8-derived Zn/Fe-mIM materials rather than ZIF-8.
10) – MS represents ZIF-8 including 90 mol% Zn and 10 mol% Co synthesised via the MS method. In this work, the term ZIF-8 is reserved for samples that retain the characteristic ZIF-8 PXRD reflections; samples that lose long-range order (Fe-rich) are denoted ZIF-8-derived Zn/Fe-mIM materials rather than ZIF-8.
Results and discussion
Effect of organic ligands on O2 adsorption performance
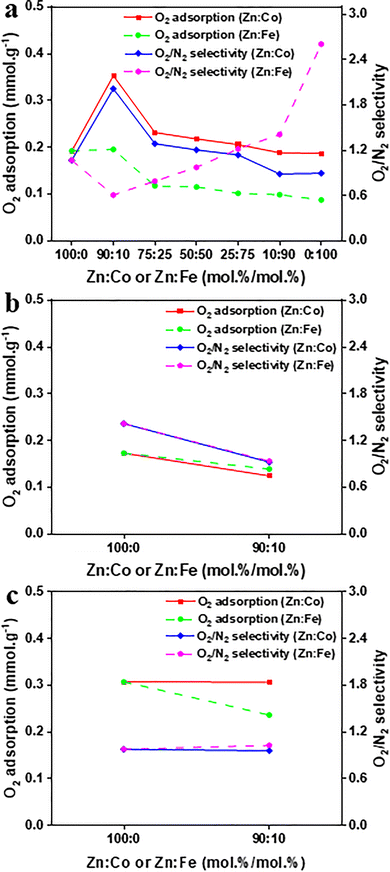
As illustrated in Fig. 2 (detailed in Fig. S11), the adsorption capacity follows the order of ZIF-62 > ZIF-8 > ZIF-7, whereas O2/N2 selectivity follows ZIF-7 > ZIF-8 > ZIF-62 (Table S3). This behaviour primarily highlights how organic ligand chemistry controls pore size, framework flexibility, and host–guest interactions, in addition to surface area.49 ZIF-62 features a mixed organic ligand composition of imidazole (IM) and bulkier benzimidazole (bIM).50 This mixed-organic ligand composition exhibits Type-I isotherms with an adsorption-induced gate-opening (narrow-to-expanded-pore) mechanism associated with IM and bIM organic ligands motion and framework distortion,50 with an average BET surface area and ultramicropore dominated PSD of 7–10 Å (Fig. S5 and Table S1). This is consistent with a balanced rigidity-flexibility that may temporarily expand the effective pore size, thereby increasing accessible pore volume and diffusion pathways.50 Meanwhile, it preserves confinement and improves O2–framework interactions without preventing diffusion, because stronger O2 interactions can promote access to/expansion of the framework and O2 equilibrates faster than N2.50 These factors are consistent with the high O2 adsorption capacity observed for ZIF-62. Interestingly, ZIF-62 also shows the highest N2 adsorption capacity likely owing to similar factors of optimal pore size and framework flexibility. Its selectivity of 0.98 (Table S3) which slightly favours N2, is consistent with the interplay of pore size, kinetic limitations, interaction strength, and molecular sieving.50 In ZIF-8, gas-induced motion of the mIM organic ligand increases flexibility and pore accessibility,51 with higher BET surface area and a micropore-dominated PSD of ∼10–16 Å (Fig. S5 and Table S1). Even so, its O2 adsorption capacity is lower than ZIF-62, probably because the larger pores may reduce confinement-driven O2–framework interaction strength (i.e., poorer ‘fit’), even if diffusion is facilitated.50,52 Its N2 adsorption capacity is also lower than ZIF-62, suggesting that the larger pore environment may provide weaker confinement-driven interactions and/or earlier saturation effects for N2.49,52 This trend also suggests that pore size and interaction strength matter more than surface area for gas adsorption.49 The selectivity of 1.07 (Table S3) still reflects a preference for O2, as slight differences in interaction strength and diffusion kinetics in the flexible framework favour O2 over N2.51 In contrast, ZIF-7 is more aperture-constrained owing to its bulkier bIM organic ligand with phenyl rings and its sorption behaviour is governed by guest-responsive gate-opening driven by organic ligand motion.53–55 It exhibits a lower BET surface area and a PSD peaked at ∼17–27 Å (Fig. S5 and Table S1) which likely reflects larger PSDs, while gas adsorption remains controlled by a comparatively restrictive pore aperture and a higher gate-opening barrier associated with the bIM organic ligand.53 As a result, it shows the lowest O2 adsorption capacity. Similarly, its N2 adsorption capacity is the lowest, indicating that restricted access (diffusional limitations and higher-pressure gate-opening) limits adsorption under the measurement conditions, despite the larger apparent PSD features.53 The observed selectivity of 1.42 (Table S3) suggests a stronger preference for O2, which may reflect differential diffusional/kinetic constraints and/or specific interactions within the narrow-pore form.53,54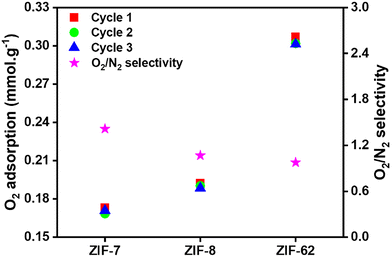 | ||
| Fig. 2 Effect of organic ligands on O2 adsorption, O2/N2 selectivity, and cyclability of ZIFs, synthesised using the MS method. | ||
Regarding cyclability and structural stability (Fig. 2, detailed in Fig. S15), ZIF-62 and ZIF-8 demonstrate significant stability and consistent adsorption capacity across multiple cycles, whereas ZIF-7 exhibits a gradual decline in performance. ZIF-62's mixed organic ligands enable an adsorption-induced gate-opening response,50 with minimal irreversible deformation.56 This flexibility can help maintain performance despite ZIF-62's comparatively lower surface area.23,49 Similarly, ZIF-8 adapts to repeated adsorption–desorption cycles due to its framework flexibility arising from mIM-driven organic ligand motions that help maintain pore accessibility.57,58 In contrast, although ZIF-7 also undergoes gate-opening,54,55 its transition occurs at higher pressures than ZIF-62,49 supporting cycling stability for gas adsorption applications.54 ZIF-7 experiences a decrease in adsorption capacity across cycles which may be associated with the steric constraints of the bIM organic ligand that limit flexibility and structural adaptability and with a less compliant response compared with ZIF-62 under these conditions.49 This may result in accumulated structural fatigue under cyclic stress. These findings underscore the importance of organic ligand-mediated flexibility of ZIFs in ensuring long-term cycling stability for gas adsorption applications.
Effect of mixed metal centres of varying compositions on O2 adsorption performance
Incorporating Co or Fe metal ions into the ZIF framework enables isovalent metal substitution that induces structural rearrangements.59 These rearrangements change the gate-opening energy barrier,60 pore sizes and interconnectivity,61 thereby affecting framework stability,59 and O2 adsorption capacity.62 To investigate these effects, ZIF-8 was selected for its moderate O2 adsorption and selectivity, whereas ZIF-7 demonstrates low adsorption but strong selectivity, and ZIF-62 displays the inverse trend.The introduction of Co into ZIF-8 at low content (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) significantly improves O2 adsorption (0.35 mmol g−1) and selectivity (2.01) (Fig. 3a; detailed in Fig. S11a and Table S3). This enhancement is likely associated with Co2+'s partially occupied d-orbitals, which may improve O2–framework interactions and affect the gate-opening which leads to preferential O2 adsorption.41 Moreover, its ionic radius (Co2+ = 0.58 Å) and preference for tetrahedral coordination align closely with Zn2+ (0.60 Å).63 Therefore, the ZIF-8 framework experiences minor distortion at low-level substitution as evidenced by retention of the framework with small lattice changes reflected in PXRD peak shifts without phase change (Fig. S1), along with minor FTIR band shifts (Fig. S3) and similar bimodal degradation profiles in TGA with modest shifts across the Co series (Fig. S7). As a result, the structure appears stable, with no clear evidence of major framework disruption at low-level substitution.59 Thus, the primary cause of the increase in O2/N2 selectivity in this regime is the local adsorption environment and changes in adsorption-site chemistry (Co-associated O2–framework interactions), rather than a change in pore structure. However, as the Co content increases beyond Zn
10) significantly improves O2 adsorption (0.35 mmol g−1) and selectivity (2.01) (Fig. 3a; detailed in Fig. S11a and Table S3). This enhancement is likely associated with Co2+'s partially occupied d-orbitals, which may improve O2–framework interactions and affect the gate-opening which leads to preferential O2 adsorption.41 Moreover, its ionic radius (Co2+ = 0.58 Å) and preference for tetrahedral coordination align closely with Zn2+ (0.60 Å).63 Therefore, the ZIF-8 framework experiences minor distortion at low-level substitution as evidenced by retention of the framework with small lattice changes reflected in PXRD peak shifts without phase change (Fig. S1), along with minor FTIR band shifts (Fig. S3) and similar bimodal degradation profiles in TGA with modest shifts across the Co series (Fig. S7). As a result, the structure appears stable, with no clear evidence of major framework disruption at low-level substitution.59 Thus, the primary cause of the increase in O2/N2 selectivity in this regime is the local adsorption environment and changes in adsorption-site chemistry (Co-associated O2–framework interactions), rather than a change in pore structure. However, as the Co content increases beyond Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, both O2 adsorption and selectivity gradually decrease. At Zn
10, both O2 adsorption and selectivity gradually decrease. At Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 0
Co = 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100, these values fall below those of monometallic ZIF-8. This trend suggests that progressive Co incorporation can change the flexibility of the aperture-opening process and may accumulate local distortions that diminish the effective accessibility of pore windows and thereby alter gas–framework interactions.61 Consequently, the contribution of the most O2-favourable sites may be diluted and/or less selective adsorption domains may increase, despite the BET surface area rising to 1667 m2 g−1 at Zn
100, these values fall below those of monometallic ZIF-8. This trend suggests that progressive Co incorporation can change the flexibility of the aperture-opening process and may accumulate local distortions that diminish the effective accessibility of pore windows and thereby alter gas–framework interactions.61 Consequently, the contribution of the most O2-favourable sites may be diluted and/or less selective adsorption domains may increase, despite the BET surface area rising to 1667 m2 g−1 at Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co mol% of 0
Co mol% of 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 (Table S1). Because ZIF-8 is flexible, BET surface area derived from N2 at 77 K does not necessarily represent dynamic pore accessibility at 298 K.57 Thus, the selectivity trend does not correspond to the BET surface area trend, which means that surface area alone is not the governing descriptor for O2/N2 selectivity in the Co series. Simultaneously, N2 adsorption at a Zn
100 (Table S1). Because ZIF-8 is flexible, BET surface area derived from N2 at 77 K does not necessarily represent dynamic pore accessibility at 298 K.57 Thus, the selectivity trend does not correspond to the BET surface area trend, which means that surface area alone is not the governing descriptor for O2/N2 selectivity in the Co series. Simultaneously, N2 adsorption at a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 is initially lower than ZIF-8, suggesting that minimal Co substitution preferentially enhances O2 adsorption over N2, perhaps owing to particular Co-associated O2–framework interactions (Fig. S13a). At higher Co contents, N2 adsorption increases and ultimately exceeds ZIF-8, suggesting that structural or chemical modifications enhance the affinity for the more polarizable gas such as N2,62 which contributes to the observed reduction in O2/N2 selectivity even when overall BET surface area remains high.
10 is initially lower than ZIF-8, suggesting that minimal Co substitution preferentially enhances O2 adsorption over N2, perhaps owing to particular Co-associated O2–framework interactions (Fig. S13a). At higher Co contents, N2 adsorption increases and ultimately exceeds ZIF-8, suggesting that structural or chemical modifications enhance the affinity for the more polarizable gas such as N2,62 which contributes to the observed reduction in O2/N2 selectivity even when overall BET surface area remains high.
Whereas, the introduction of Fe into ZIF-8 at a low content (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) slightly enhances O2 adsorption (0.20 mmol g−1) but results in the lowest selectivity (0.61) (Fig. 3a; detailed in Fig. S11b and Table S3). This behaviour may arise from Fe2+'s unpaired d-electrons, which may change O2–framework interactions and the gate-opening barrier,36,41 and thus less preferential O2 binding. Moreover, a slightly larger radius and coordination mismatch (Fe2+ = 0.63 Å versus Zn2+ = 0.60 Å; (Fe2+ is more coordination-flexible than Zn2+))63 that introduces local strain, promotes defect formation, and drives framework distortion even at low substitution levels.59,61 These defects can introduce additional less-specific adsorption domains that preferentially increase N2 adsorption relative to O2, in turn suppressing selectivity at low Fe loading.64 This is supported by PXRD peak broadening (Fig. S1), a less-faceted particle morphology (Fig. S9), and TGA evidence of earlier mass loss with reduced thermal stability (Fig. S7). As Fe content increases, O2 adsorption decreases to 0.09 mmol g−1 with 9.6% loss in the ZIF-8-derived Fe-mIM material (Zn
10) slightly enhances O2 adsorption (0.20 mmol g−1) but results in the lowest selectivity (0.61) (Fig. 3a; detailed in Fig. S11b and Table S3). This behaviour may arise from Fe2+'s unpaired d-electrons, which may change O2–framework interactions and the gate-opening barrier,36,41 and thus less preferential O2 binding. Moreover, a slightly larger radius and coordination mismatch (Fe2+ = 0.63 Å versus Zn2+ = 0.60 Å; (Fe2+ is more coordination-flexible than Zn2+))63 that introduces local strain, promotes defect formation, and drives framework distortion even at low substitution levels.59,61 These defects can introduce additional less-specific adsorption domains that preferentially increase N2 adsorption relative to O2, in turn suppressing selectivity at low Fe loading.64 This is supported by PXRD peak broadening (Fig. S1), a less-faceted particle morphology (Fig. S9), and TGA evidence of earlier mass loss with reduced thermal stability (Fig. S7). As Fe content increases, O2 adsorption decreases to 0.09 mmol g−1 with 9.6% loss in the ZIF-8-derived Fe-mIM material (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 0
Fe = 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100), but selectivity increases to 2.60, indicating its peak value among these samples. Fe substitution, with Fe2+ being more coordination-flexible than Zn2+, perturbs the intrinsic tetrahedral Zn-mIM node environment characteristic of ZIF-8, resulting in a significant reduction in BET surface area (Table S1) and compounding strain from the Fe2+/Zn2+ size mismatch.59,65 High Fe contents (ZIF-8-derived Zn/Fe-mIM (Zn
100), but selectivity increases to 2.60, indicating its peak value among these samples. Fe substitution, with Fe2+ being more coordination-flexible than Zn2+, perturbs the intrinsic tetrahedral Zn-mIM node environment characteristic of ZIF-8, resulting in a significant reduction in BET surface area (Table S1) and compounding strain from the Fe2+/Zn2+ size mismatch.59,65 High Fe contents (ZIF-8-derived Zn/Fe-mIM (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 75
Fe = 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25–0
25–0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100)) can lose ZIF-8 long-range order and promote significant structural disorder or partial amorphisation, thereby compromising structural stability,64,66 consistent with the extensive distortion needed to accommodate less compatible Fe nodes.59,61 While increased Fe distorts the structure and diminishes overall capacity, Fe-centred local chemistry and steric constraints can still favour O2 over N2, thereby gradually improving selectivity.36,67 In the high-Fe regime, the increase in selectivity correlates with the diminished accessible porosity/pore-window restriction (Table S1), which likely restricts N2 adsorption more than O2, resulting in a selectivity-capacity trade-off.36,61 At a Zn
100)) can lose ZIF-8 long-range order and promote significant structural disorder or partial amorphisation, thereby compromising structural stability,64,66 consistent with the extensive distortion needed to accommodate less compatible Fe nodes.59,61 While increased Fe distorts the structure and diminishes overall capacity, Fe-centred local chemistry and steric constraints can still favour O2 over N2, thereby gradually improving selectivity.36,67 In the high-Fe regime, the increase in selectivity correlates with the diminished accessible porosity/pore-window restriction (Table S1), which likely restricts N2 adsorption more than O2, resulting in a selectivity-capacity trade-off.36,61 At a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, N2 adsorption surpasses monometallic ZIF-8, indicating that early-stage Fe substitution improves overall adsorption capacity, preferentially for N2 owing to the defects (Fig. S13b). As the Fe content rises, the accumulation of size/coordination mismatch narrows pore size and reduces accessible porosity.61 This leads to a transition from non-specific defect-assisted adsorption at low Fe to more O2-selective environments at high Fe, resulting in decreased N2 adsorption.61 This trade-off presumably indicates both chemistry and steric changes resulting from framework deformation and partial amorphisation.66 Simultaneously, the higher selectivity at greater Fe contents supports the idea that partial Fe substitution can enhance specific gas-framework interactions, though overall adsorption capacity declines.
10, N2 adsorption surpasses monometallic ZIF-8, indicating that early-stage Fe substitution improves overall adsorption capacity, preferentially for N2 owing to the defects (Fig. S13b). As the Fe content rises, the accumulation of size/coordination mismatch narrows pore size and reduces accessible porosity.61 This leads to a transition from non-specific defect-assisted adsorption at low Fe to more O2-selective environments at high Fe, resulting in decreased N2 adsorption.61 This trade-off presumably indicates both chemistry and steric changes resulting from framework deformation and partial amorphisation.66 Simultaneously, the higher selectivity at greater Fe contents supports the idea that partial Fe substitution can enhance specific gas-framework interactions, though overall adsorption capacity declines.
Across the ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co) series, all compositions show a gradual reduction in O2 adsorption over consecutive cycles, likely due to partial pore obstruction or framework degradation. Among them, monometallic ZIF-8 and bimetallic ZIF-8 (Zn
Co) series, all compositions show a gradual reduction in O2 adsorption over consecutive cycles, likely due to partial pore obstruction or framework degradation. Among them, monometallic ZIF-8 and bimetallic ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) combine favourable adsorption capacity, selectivity, and cycling stability. This is due to the limited disruption of Zn–organic ligand bonds, which preserves structural integrity (Fig. S15a). The Zn
10) combine favourable adsorption capacity, selectivity, and cycling stability. This is due to the limited disruption of Zn–organic ligand bonds, which preserves structural integrity (Fig. S15a). The Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe series shows a similar trend, with each composition exhibiting a slight decline in O2 adsorption over cycles. However, formulations with low Fe content, such as a Zn
Fe series shows a similar trend, with each composition exhibiting a slight decline in O2 adsorption over cycles. However, formulations with low Fe content, such as a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, attain an effective equilibrium of adsorption and selectivity, coupled with comparatively steady cyclic performance, indicating their suitability for practical applications (Fig. S15b).
10, attain an effective equilibrium of adsorption and selectivity, coupled with comparatively steady cyclic performance, indicating their suitability for practical applications (Fig. S15b).
To expand the parametric space where functional bimetallic ZIF can be synthesised, the Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 and the Zn
10 and the Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 in ZIF-7 (Fig. 3b), and ZIF-62 (Fig. 3c) were also assessed (detailed in Fig. S11c and d, along with Table S3). The replacement of Zn with either Co or Fe in ZIF-7 lowers both O2 adsorption and selectivity. This is attributed to the relatively aperture-constrained structure of ZIF-7, which is less capable of accommodating metal substitution, resulting in a lower number of effective O2 binding sites.53 Concurrently, the enhanced N2 adsorption indicates that the modified pore shape generates new pore environments that are more suited to the larger N2 molecule (Fig. S13c). Both ZIF-7 variations exhibit reduced adsorption beyond the first cycle, suggesting partial pore obstruction or framework deformation induced by metal substitution (Fig. S15c). In ZIF-62, the mixed-organic ligand (IM/bIM) framework tolerates low-level substitution better.59 ZIF-62 (Zn
10 in ZIF-7 (Fig. 3b), and ZIF-62 (Fig. 3c) were also assessed (detailed in Fig. S11c and d, along with Table S3). The replacement of Zn with either Co or Fe in ZIF-7 lowers both O2 adsorption and selectivity. This is attributed to the relatively aperture-constrained structure of ZIF-7, which is less capable of accommodating metal substitution, resulting in a lower number of effective O2 binding sites.53 Concurrently, the enhanced N2 adsorption indicates that the modified pore shape generates new pore environments that are more suited to the larger N2 molecule (Fig. S13c). Both ZIF-7 variations exhibit reduced adsorption beyond the first cycle, suggesting partial pore obstruction or framework deformation induced by metal substitution (Fig. S15c). In ZIF-62, the mixed-organic ligand (IM/bIM) framework tolerates low-level substitution better.59 ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) demonstrates a minor decrease in O2 adsorption with a slight increase in N2 adsorption which results in reduced selectivity. These trends suggest that Co substitution in ZIF-62 slightly changes the pore environment and weakens O2 physisorption relative to N2.59,61 However, the overall impact remains small due to the flexible framework's ability to dilute local distortions.59 The Zn
10) demonstrates a minor decrease in O2 adsorption with a slight increase in N2 adsorption which results in reduced selectivity. These trends suggest that Co substitution in ZIF-62 slightly changes the pore environment and weakens O2 physisorption relative to N2.59,61 However, the overall impact remains small due to the flexible framework's ability to dilute local distortions.59 The Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe (90
Fe (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) derivative exhibits a larger drop in both O2 and N2 adsorptions, with N2 decreasing a bit more, leading to a modest increase in selectivity (Fig. S13d). This is ascribed to Fe-induced defects/size-mismatch that constrict windows and reduce accessible porosity, preferentially excluding N2 over O2.59,64 ZIF-62 (Zn
10) derivative exhibits a larger drop in both O2 and N2 adsorptions, with N2 decreasing a bit more, leading to a modest increase in selectivity (Fig. S13d). This is ascribed to Fe-induced defects/size-mismatch that constrict windows and reduce accessible porosity, preferentially excluding N2 over O2.59,64 ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) has good stability likely due to the flexible framework accommodating Fe-induced strain without catastrophic rearrangement, whereas Zn
10) has good stability likely due to the flexible framework accommodating Fe-induced strain without catastrophic rearrangement, whereas Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co reveals a gradual decrease in O2 adsorption, indicative of slow framework deformation or emerging pore obstruction around Co sites (Fig. S15d).
Co reveals a gradual decrease in O2 adsorption, indicative of slow framework deformation or emerging pore obstruction around Co sites (Fig. S15d).
Effect of synthesis methods on O2 adsorption performance
The impact of various synthesis methods on the structural stability, adsorption capacity, and degradation resistance of ZIFs was assessed by evaluating the changes in adsorption performance over multiple cycles. ZIF-8 and ZIF-62 were selected as monometallic frameworks covering organic ligand flexibility and baseline O2 capacity, along with ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) and ZIF-62 (Zn
10) and ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) to isolate metal centre effects. Together, these four materials include the extremes in capacity, selectivity, and stability, thereby enabling a sensitive evaluation of how synthesis methods translate O2 adsorption performance.
10) to isolate metal centre effects. Together, these four materials include the extremes in capacity, selectivity, and stability, thereby enabling a sensitive evaluation of how synthesis methods translate O2 adsorption performance.
The US method achieves the highest initial O2 adsorption capacity for ZIF-8 at 0.26 mmol g−1 (Fig. 4a; detailed in Fig. S12a and S14a, along with Table S4), demonstrating remarkable stability with just a 0.5% decrease over three cycles (Fig. S16a). This exceptional performance likely arises from improved nucleation and more homogeneous dispersion of nuclei under sonochemical conditions,43 along with cavitation-induced shock waves and microjets that intensify local mixing and mass transfer during coordination formation.68 US-assisted ZIF syntheses are also widely associated with smaller crystals and narrower PSDs,46 consistent with the observed PSD (∼10–16 Å) and increased surface area (1506 m2 g−1vs. 1402 m2 g−1), as confirmed by SEM images (Fig. S10) and gas adsorption measurements (Fig. S6 and Table S2). The MS method yields an initial capacity of 0.19 mmol g−1 with a slight decline of 2.0%, which suggests good stability, but a lesser capacity compared to the US method. This is consistent with the MS method achieving uniform bulk mixing through a rotating magnetic field, though with weaker shear fields. In contrast, the OS method yields a reduced starting capacity of 0.17 mmol g−1 and a more significant 4.7% decline, indicating worse stability relative to MS, likely attributable to less uniform crystallite formation despite intense mixing, good control over shear forces, and dead zones prevention. The OS & US method produces 0.18 mmol g−1 but shows a significant 16.1% decline, suggesting a surprising lack of stability that could reflect inadequate synthesis conditions rather than a synergistic effect between the methods. This lack of synergy is expected because intensive agitation can disrupt stable cavitation clouds and redistribute bubbles, resulting in a reduction in local ultrasonic intensity and the formation of generally nonuniform nucleation and growth.68 The resulting morphological heterogeneity is in accordance with the SEM observations and may result in a lower cyclability than the most effective single-field method. The BM method yields the lowest capacity of 0.13 mmol g−1, reflecting an 10.9% decrease; attributable to high-energy milling inducing defects that undermine the microporous structure.44,64 This milling-induced disorder, arising from direct mechanical treatment of the precursors, is evidenced by additional and broadened PXRD peaks (Fig. S2a), FTIR band broadening (Fig. S4a), severe BET surface area loss (401 m2 g−1 and Table S2), and a higher low-temperature mass loss (∼200 °C; Fig. S8a). These changes reflect the typical BM method trade-off: rapid processing and particle-size reduction at the cost of coordination defects and a partial loss of long-range order,64,66 which leads to the collapse of N2-accessible microporosity and an increase of non-selective adsorption domains (Table S2). This results in fewer well-defined micropore adsorption sites for O2 adsorption are available,66 leading to decreased O2/N2 selectivity (Table S4). The MW method achieves 0.19 mmol g−1 with an 7.8% decline, equivalent to the MS method in capacity but exhibiting much worse stability. This is likely owing to rapid heating and fast kinetics that can shift nucleation and growth balance,69 resulting in defective structures with altered adsorption,70 as supported by SEM images showing rod-like and less homogeneous crystals (Fig. S10).
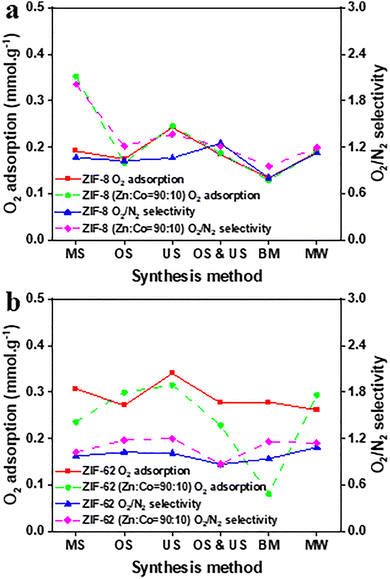 | ||
Fig. 4 Effect of synthesis methods on O2 adsorption, and O2/N2 selectivity of (a) ZIF-8, and ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90 Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10), along with (b) ZIF-62, and ZIF-62 (Zn 10), along with (b) ZIF-62, and ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90 Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10). 10). | ||
For ZIF-8 with a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co mol% of 90
Co mol% of 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, framework retention with minor lattice changes is confirmed by PXRD and FTIR (Fig. S2, and S4). The MS method delivers the maximum initial O2 adsorption capacity of 0.35 mmol g−1 (Fig. 4a; detailed in Fig. S12b and S14b, along with Table S4), which may reflect that partial Co substitution can modify adsorption energetics and O2–framework interactions in ZIF-8.61,62 The MS method exhibits the highest O2/N2 selectivity (Table S4), despite similar surface area and PSD (Table S2). This suggests that method-dependent variations in the effective adsorption landscape, such as Co incorporation uniformity46 and a reduced proportion of non-selective defect domains64 (Fig. S10), rather than surface area, are the primary factors. This maximises the availability of Co-associated O2-favourable sites while minimising N2 adsorption. A 9.9% decline across cycles indicates potential instability (Fig. S16b), perhaps due to distortions that reduce pore accessibility. The US method demonstrates a modest capacity of 0.26 mmol g−1, accompanied by a 10.8% decline, suggesting poorer stability than expected. The OS method produces a reduced capacity of 0.17 mmol g−1 with a 3.9% decline, indicating better stability than the MS or US methods. The OS & US method yields 0.19 mmol g−1 with a 7.5% decrease, demonstrating enhanced stability compared to the MS and US methods, but with lower capacity, as the combined fields can improve reactant dispersion via simultaneous acoustic and mechanical effects.68,69 The MW method reaches 0.19 mmol g−1 with only a 1.2% reduction, which implies that rapid crystallisation stabilises the Co-substituted framework. This is consistent with MW's potential for rapid reaction kinetics and potentially more uniform nucleation control.69 In contrast, the BM method exhibits poor performance at 0.13 mmol g−1, with a significant 34.8% decline, indicative of rapid degradation resulting from mechanical stress. High-energy milling is expected to introduce coordination defects and partially disrupt long-range order,44,66,71 as reflected by changes in PXRD and FTIR (Fig. S2 and S4) and porosity (Table S2). In the mixed-metal Zn
10, framework retention with minor lattice changes is confirmed by PXRD and FTIR (Fig. S2, and S4). The MS method delivers the maximum initial O2 adsorption capacity of 0.35 mmol g−1 (Fig. 4a; detailed in Fig. S12b and S14b, along with Table S4), which may reflect that partial Co substitution can modify adsorption energetics and O2–framework interactions in ZIF-8.61,62 The MS method exhibits the highest O2/N2 selectivity (Table S4), despite similar surface area and PSD (Table S2). This suggests that method-dependent variations in the effective adsorption landscape, such as Co incorporation uniformity46 and a reduced proportion of non-selective defect domains64 (Fig. S10), rather than surface area, are the primary factors. This maximises the availability of Co-associated O2-favourable sites while minimising N2 adsorption. A 9.9% decline across cycles indicates potential instability (Fig. S16b), perhaps due to distortions that reduce pore accessibility. The US method demonstrates a modest capacity of 0.26 mmol g−1, accompanied by a 10.8% decline, suggesting poorer stability than expected. The OS method produces a reduced capacity of 0.17 mmol g−1 with a 3.9% decline, indicating better stability than the MS or US methods. The OS & US method yields 0.19 mmol g−1 with a 7.5% decrease, demonstrating enhanced stability compared to the MS and US methods, but with lower capacity, as the combined fields can improve reactant dispersion via simultaneous acoustic and mechanical effects.68,69 The MW method reaches 0.19 mmol g−1 with only a 1.2% reduction, which implies that rapid crystallisation stabilises the Co-substituted framework. This is consistent with MW's potential for rapid reaction kinetics and potentially more uniform nucleation control.69 In contrast, the BM method exhibits poor performance at 0.13 mmol g−1, with a significant 34.8% decline, indicative of rapid degradation resulting from mechanical stress. High-energy milling is expected to introduce coordination defects and partially disrupt long-range order,44,66,71 as reflected by changes in PXRD and FTIR (Fig. S2 and S4) and porosity (Table S2). In the mixed-metal Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co framework, node heterogeneity can further increase local strain, making the material more susceptible to persistent defect formation and pore blockage under the BM method.59,64 This reduces accessible Co-associated O2-favourable sites and accelerates capacity degradation (Table S4).
Co framework, node heterogeneity can further increase local strain, making the material more susceptible to persistent defect formation and pore blockage under the BM method.59,64 This reduces accessible Co-associated O2-favourable sites and accelerates capacity degradation (Table S4).
For ZIF-62, method-dependent porosity variations are evident from BET surface area and PSD, while the BM method exhibits defect/secondary-phase signatures in PXRD and FTIR (Fig. S2c and S4c). Across syntheses, TGA indicates relatively similar thermal stability (Fig. S8c). The US method significantly outperforms other methods. It achieves an initial capacity of 0.34 mmol g−1 (Fig. 4b; detailed in Fig. S12c and S14c, along with Table S4), and shows a negligible 1.3% decline (Fig. S16c). These results underscore that the US method can intensify mixing, accelerate nucleation, and tune crystal size in ways that improve accessible porosity and reduce performance-limiting heterogeneity.72 The MS method shows a capacity of 0.31 mmol g−1 and a 1.9% decrease, which indicates robust performance in both capacity and stability. The OS method produces 0.27 mmol g−1 with a 6.1% reduction, while the OS & US method yields 0.28 mmol g−1 with a 3.8% decrease. The MW method yields 0.26 mmol g−1 with a 4.7% drop. This intermediate stability is consistent with the MW method generally providing rapid, relatively homogeneous heating and shortened crystallisation kinetics, which can alter defect densities and adsorption outcomes.69 Although BM introduces defect and secondary-phase signatures in PXRD and FTIR (Fig. S2c and S4c), ZIF-62 maintains a comparable initial O2 capacity (0.28 mmol g−1). This is likely because milling is followed by a thermal treatment phase that can partially restore the framework connectivity. Indeed, the extent and consequences of disorder are strongly chemistry- and process-dependent.71 Consequently, any residual defects appear stable over 3 cycles and do not produce measurable capacity loss, and BM does not translate into the same loss of micropore functionality or O2/N2 selectivity observed for directly milled frameworks (Tables S2 and S4).
For ZIF-62 with a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe mol% of 90
Fe mol% of 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, the US method reaches the highest initial capacity of 0.32 mmol g−1 (Fig. 4b; detailed in Fig. S12d and S14d, along with Table S4); however, a 12.6% decline suggests low stability (Fig. S16d). The OS method yields 0.30 mmol g−1 with a 16.7% reduction, suggesting that Fe incorporation may increase the framework's sensitivity to local distortions and accessibility losses during cycling, consistent with mixed-metal node effects reported for ZIFs.59 The MW method yields 0.29 mmol g−1 with a 3.6% reduction, achieving a favourable balance between capacity and stability despite a low surface area that underestimates O2-accessible ultramicroporosity in this flexible structure,51 presumably owing to the fast and more homogeneous heating that stabilises the Fe-substituted structure.69 This is consistent with PXRD and FTIR evidence of framework preservation (Fig. S2d and S4d). The MS method yields 0.24 mmol g−1 with a 0.9% decrease, which implies increased stability but reduced capacity. The OS & US method yields 0.23 mmol g−1, reflecting a 9.5% decrease, which is modest in every aspect. The BM method yields the lowest capacity (0.08 mmol g−1) and the largest decline (18.0%). For the mixed-metal ZIF-62 (Zn
10, the US method reaches the highest initial capacity of 0.32 mmol g−1 (Fig. 4b; detailed in Fig. S12d and S14d, along with Table S4); however, a 12.6% decline suggests low stability (Fig. S16d). The OS method yields 0.30 mmol g−1 with a 16.7% reduction, suggesting that Fe incorporation may increase the framework's sensitivity to local distortions and accessibility losses during cycling, consistent with mixed-metal node effects reported for ZIFs.59 The MW method yields 0.29 mmol g−1 with a 3.6% reduction, achieving a favourable balance between capacity and stability despite a low surface area that underestimates O2-accessible ultramicroporosity in this flexible structure,51 presumably owing to the fast and more homogeneous heating that stabilises the Fe-substituted structure.69 This is consistent with PXRD and FTIR evidence of framework preservation (Fig. S2d and S4d). The MS method yields 0.24 mmol g−1 with a 0.9% decrease, which implies increased stability but reduced capacity. The OS & US method yields 0.23 mmol g−1, reflecting a 9.5% decrease, which is modest in every aspect. The BM method yields the lowest capacity (0.08 mmol g−1) and the largest decline (18.0%). For the mixed-metal ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10), this stronger detrimental impact is consistent with milling-induced disorder reducing accessible porosity and adsorption sites,64 as well as in mixed-metal ZIF-62, additional heterogeneity at Zn/Fe nodes can amplify local strain,59 while the Zn–Fe ionic-radius and coordination mismatch can further bias the structure toward persistent disorder and partial pore blockage.63 that are not fully mitigated during the subsequent thermal treatment phase. As a result, the number of accessible adsorption sites is substantially reduced (Table S2), leading to poor capacity retention (Fig. S16d) and more pronounced defect and secondary-phase signatures in PXRD and FTIR (Fig. S2d and S4d).
10), this stronger detrimental impact is consistent with milling-induced disorder reducing accessible porosity and adsorption sites,64 as well as in mixed-metal ZIF-62, additional heterogeneity at Zn/Fe nodes can amplify local strain,59 while the Zn–Fe ionic-radius and coordination mismatch can further bias the structure toward persistent disorder and partial pore blockage.63 that are not fully mitigated during the subsequent thermal treatment phase. As a result, the number of accessible adsorption sites is substantially reduced (Table S2), leading to poor capacity retention (Fig. S16d) and more pronounced defect and secondary-phase signatures in PXRD and FTIR (Fig. S2d and S4d).
Mechanistic framework and design rules
O2 adsorption performance is governed by a coupled balance between (i) adsorption-site chemistry (metal–O2 interactions and the availability of O2-favourable sites), (ii) effective pore accessibility under operating conditions (gate-opening/aperture dynamics and restrictions caused by defects or strain), and (iii) structural integrity under cycling (resistance to irreversible disorder and pore blockage). The dominant contribution depends on the regime: in monometallic ZIFs, flexibility governed by organic ligands and aperture response mainly dictate capacity and cyclability; in bimetallic ZIFs with low substitution, selectivity enhancements are mostly influenced by changes in local site chemistry while maintaining the framework, whereas at higher substitution levels or during defect-prone processing, limitations in accessibility and non-selective domains can dominate, resulting in trade-offs between selectivity and capacity. Synthesis methods modulate these same factors by controlling crystallinity, defect density, and compositional uniformity, which explains why materials with similar BET surface area and PSD can still exhibit different O2/N2 selectivity and stability. To place these design rules in context, a representative literature benchmarking summary of O2 adsorption, O2/N2 selectivity, and recyclability for closely related adsorbents is provided in Table S5.Among the synthesis methods, the US method improves O2 adsorption capacity and stability in monometallic ZIFs through optimised nucleation and pore accessibility. Whereas in partial metal-substituted ZIFs, it increased initial capacities, particularly in ZIF-8 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10), but sometimes compromises long-term stability. The MW method demonstrates potential, attaining a robust equilibrium of capacity and stability in bimetallic ZIFs. The OS and OS & US methods provide dependable durability with modest performance, appropriate for long-term operations; however, the OS & US method falls short of expectations in ZIF-8. The BM method shows framework- and protocol-dependent behaviour: direct milling can induce persistent disorder and pore blockage, whereas BM followed by thermal treatment phase (as in ZIF-62) can partially recover framework connectivity, though mixed-metal systems may remain more susceptible to milling-induced heterogeneity. These results highlight the need to customise synthesis methods to particular ZIF compositions to optimise O2 adsorption and provide insight to develop scalable ZIFs for industrial gas separation and storage.
10), but sometimes compromises long-term stability. The MW method demonstrates potential, attaining a robust equilibrium of capacity and stability in bimetallic ZIFs. The OS and OS & US methods provide dependable durability with modest performance, appropriate for long-term operations; however, the OS & US method falls short of expectations in ZIF-8. The BM method shows framework- and protocol-dependent behaviour: direct milling can induce persistent disorder and pore blockage, whereas BM followed by thermal treatment phase (as in ZIF-62) can partially recover framework connectivity, though mixed-metal systems may remain more susceptible to milling-induced heterogeneity. These results highlight the need to customise synthesis methods to particular ZIF compositions to optimise O2 adsorption and provide insight to develop scalable ZIFs for industrial gas separation and storage.
Conclusions
This study evaluated ZIFs for efficient and sustainable O2 capture and separation. O2 adsorption performance in ZIFs is governed by the interplay of organic ligands, metal centres (including secondary metals in bimetallics), and the synthesis method, so a holistic design strategy is required to maximise adsorption capacity, selectivity, and cyclability. In monometallics, gate-opening was enabled by the mixed IM + bIM organic ligand set in ZIF-62 and the highest adsorption was recorded (e.g., 0.34 mmol g−1 for ZIF-62 – US), whereas in ZIF-8 with mIM a flexible but slightly larger pore was obtained (moderate adsorption, 0.19 mmol g−1 for ZIF-8 – MS), and in ZIF-7 with bIM the more aperture-constrained pores were produced with the highest selectivity of 1.42 at lower capacity. Furthermore, Co2+ incorporation into ZIF-8 increased O2 adsorption and selectivity at low loading (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 90
Co = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 – MS: 0.35 mmol g−1; selectivity of 2.01) but a higher 3-cycle loss was incurred (9.9%). Fe2+ substitution was shown to reduce capacity at high loading (ZIF-8-derived Fe-mIM material (Zn
10 – MS: 0.35 mmol g−1; selectivity of 2.01) but a higher 3-cycle loss was incurred (9.9%). Fe2+ substitution was shown to reduce capacity at high loading (ZIF-8-derived Fe-mIM material (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 0
Fe = 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100): selectivity 2.60 but 0.09 mmol g−1), yet in ZIF-62 a modest Fe level improved the balance (Zn
100): selectivity 2.60 but 0.09 mmol g−1), yet in ZIF-62 a modest Fe level improved the balance (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 – MW: 0.29 mmol g−1; selectivity 1.15; 3.6% loss over 3 cycles). Finally, the capacity and cycle stability were generally improved by the US method in monometallics (ZIF-62 – US: 0.34 mmol g−1; selectivity 1.01; 1.3% loss; ZIF-8 – US: 0.26 mmol g−1; selectivity 1.06; 0.5% loss). In bimetallics, stability was better preserved by the MS or MW methods (e.g., ZIF-62 (Zn
10 – MW: 0.29 mmol g−1; selectivity 1.15; 3.6% loss over 3 cycles). Finally, the capacity and cycle stability were generally improved by the US method in monometallics (ZIF-62 – US: 0.34 mmol g−1; selectivity 1.01; 1.3% loss; ZIF-8 – US: 0.26 mmol g−1; selectivity 1.06; 0.5% loss). In bimetallics, stability was better preserved by the MS or MW methods (e.g., ZIF-62 (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe = 90
Fe = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) – MW: 0.29 mmol g−1; selectivity 1.15; 3.6% loss). Therefore, considering balanced performance across three cycles, ZIF-62 – US emerged as the lead candidate. These results clarify the essential roles of organic ligand design, metal substitution, and synthesis optimisation in customising ZIFs for improved O2 adsorption and separation.
10) – MW: 0.29 mmol g−1; selectivity 1.15; 3.6% loss). Therefore, considering balanced performance across three cycles, ZIF-62 – US emerged as the lead candidate. These results clarify the essential roles of organic ligand design, metal substitution, and synthesis optimisation in customising ZIFs for improved O2 adsorption and separation.
Author contributions
H. M. and L. M. shared co-first authorship, having collaborated closely on the project where they made equal contributions. They jointly led the project administration, conceptual and experimental design, analysis, interpretation of results, and writing the draft. A. K. contributed to the analysis, interpretation of results, and writing the draft, and J. M. contributed to the experimental component of the study. B. D. F. and M. R. H. contributed to the project administration, conceptual and experimental design, analysis, and interpretation of results.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available: chemicals, synthesis methods, characterisation methodologies including PXRD, FTIR, low-pressure gas sorption, TGA, and SEM/EDX, along with gas adsorption measurement, and O2/N2 selectivity. See DOI: https://doi.org/10.1039/d6ma00271d.Acknowledgements
The authors acknowledge the use of facilities within the CSIRO Mineral Resources laboratories, Monash, Monash X-ray Platform, and Monash Centre for Electron Microscopy.References
- A. L. Sutton, L. Melag, M. M. Sadiq and M. R. Hill, Angew. Chem., Int. Ed., 2022, 61, e202208305 CrossRef CAS PubMed.
- J. Emsley, Nature's building blocks: an AZ guide to the elements, Oxford University Press, USA, 2011 Search PubMed.
- R. J. Allam, Energy Procedia, 2009, 1, 461–470 CrossRef CAS.
- M. Dobson, Int. J. Tuberc. Lung Dis., 2001, 5, 520–523 CAS.
- L. Melag, M. M. Sadiq, K. Konstas, F. Zadehahmadi, K. Suzuki and M. R. Hill, RSC Adv., 2020, 10, 40960–40968 RSC.
- O. Talu, J. Li, R. Kumar, P. M. Mathias, J. D. Moyer Jr and J. M. Schork, Gas Sep. Purif., 1996, 10, 149–159 CrossRef CAS.
- N. K. Jensen, T. E. Rufford, G. Watson, D. K. Zhang, K. I. Chan and E. F. May, J. Chem. Eng. Data, 2012, 57, 106–113 CrossRef CAS.
- J. C. White, P. K. Dutta, K. Shqau and H. Verweij, Langmuir, 2010, 26, 10287–10293 CrossRef CAS PubMed.
- M. Nazari, F. Zadehahmadi, M. M. Sadiq, A. L. Sutton, H. Mahdavi and M. R. Hill, Commun. Mater., 2024, 5, 170 CrossRef.
- K. Möller and T. Bein, Chem. Soc. Rev., 2013, 42, 3689–3707 RSC.
- Y. Li and J. Yu, Nat. Rev. Mater., 2021, 6, 1156–1174 CrossRef CAS.
- A. Khaleque, M. M. Alam, M. Hoque, S. Mondal, J. B. Haider, B. Xu, M. A. H. Johir, A. K. Karmakar, J. L. Zhou, M. B. Ahmed and M. A. Moni, Environ. Adv., 2020, 2, 100019 CrossRef.
- L. Zhang, K. Chen, B. Chen, J. L. White and D. E. Resasco, J. Am. Chem. Soc., 2015, 137, 11810–11819 CrossRef CAS PubMed.
- P. Lu, J. Xu, Y. Sun, R. Guillet-Nicolas, T. Willhammar, M. Fahda, E. Dib, B. Wang, Z. Qin, H. Xu, J. Cho, Z. Liu, H. Yu, X. Yang, Q. Lang, S. Mintova, X. Zou and V. Valtchev, Nature, 2024, 636, 368–373 CrossRef CAS PubMed.
- M. Eddaoudi, D. F. Sava, J. F. Eubank, K. Adil and V. Guillerm, Chem. Soc. Rev., 2015, 44, 228–249 RSC.
- E. Pérez-Botella, S. Valencia and F. Rey, Chem. Rev., 2022, 122, 17647–17695 CrossRef.
- H. Demir, S. J. Stoneburner, W. Jeong, D. Ray, X. Zhang, O. K. Farha, C. J. Cramer, J. I. Siepmann and L. Gagliardi, J. Phys. Chem. C, 2019, 123, 12935–12946 CrossRef CAS.
- D. Zou, D. Liu and J. Zhang, Energy Environ. Mater., 2018, 1, 209–220 CrossRef CAS.
- F. S. Butt, A. Lewis, F. Dingwall, N. A. Mazlan, N. Radacsi, X. Fan, X. Chen, Y. Yang, S. Yang and Y. Huang, Mater. Today Chem., 2023, 34, 101804 CrossRef CAS.
- K. Noh, J. Lee and J. Kim, Isr. J. Chem., 2018, 58, 1075–1088 CrossRef CAS.
- M. A. Nazir, S. Ullah, M. U. Shahid, I. Hossain, T. Najam, M. A. Ismail, A. u Rehman, M. R. Karim and S. S. A. Shah, Sep. Purif. Technol., 2025, 356, 129828 CrossRef CAS.
- S. Wang, L. Luo, A. Wu, D. Wang, L. Wang, Y. Jiao and C. Tian, Coord. Chem. Rev., 2024, 498, 215464 CrossRef CAS.
- Z. Zheng, Z. Rong, H. L. Nguyen and O. M. Yaghi, Inorg. Chem., 2023, 62, 20861–20873 CrossRef CAS.
- S. Sumbal, Z. Aslam, U. Irshad, S. Anwar, A. Abbas, W. Ahmad and A. Hamza, Appl. Organomet. Chem., 2024, 38, e7747 CrossRef CAS.
- S. K. Nandigama, V. R. Bheeram and S. B. Mukkamala, Environ. Chem. Lett., 2019, 17, 447–454 CrossRef CAS.
- P. Atchutha Rao, H. Padhy, A. Venkateswara Rao, R. Kumar Ganta, S. Bevara, S. Maddila and S. Babu Mukkamala, Inorg. Chem. Commun., 2024, 165, 112576 CrossRef CAS.
- B. Russell, J. Villaroel, K. Sapag and A. D. Migone, J. Phys. Chem. C, 2014, 118, 28603–28608 CrossRef CAS.
- R. Boada, S. Diaz-Moreno, S. E. Norman and D. T. Bowron, Mol. Phys., 2019, 117, 3456–3463 CrossRef CAS.
- C. O. Ania, E. García-Pérez, M. Haro, J. J. Gutiérrez-Sevillano, T. Valdés-Solís, J. B. Parra and S. Calero, J. Phys. Chem. Lett., 2012, 3, 1159–1164 CrossRef CAS PubMed.
- K. S. Park, Z. Ni, A. P. Côté, J. Y. Choi, R. Huang, F. J. Uribe-Romo, H. K. Chae, M. O’Keeffe and O. M. Yaghi, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 10186–10191 CrossRef CAS.
- R. Banerjee, A. Phan, B. Wang, C. Knobler, H. Furukawa, M. O'Keeffe and O. M. Yaghi, Science, 2008, 319, 939–943 CrossRef CAS PubMed.
- A. Phan, C. J. Doonan, F. J. Uribe-Romo, C. B. Knobler, M. O’Keeffe and O. M. Yaghi, Acc. Chem. Res., 2010, 43, 58–67 CrossRef CAS PubMed.
- H. Mahdavi, H. Zhang, L. K. Macreadie, C. M. Doherty, D. Acharya, S. J. D. Smith, X. Mulet and M. R. Hill, Nano Res., 2022, 15, 3533–3538 CrossRef CAS.
- H. Mahdavi, N. T. Eden, C. M. Doherty, D. Acharya, S. J. D. Smith, X. Mulet and M. R. Hill, ACS Appl. Mater. Interfaces, 2022, 14, 23392–23399 CrossRef CAS.
- R.-B. Lin, S. Xiang, W. Zhou and B. Chen, Chem, 2020, 6, 337–363 CAS.
- D. E. Jaramillo, A. Jaffe, B. E. R. Snyder, A. Smith, E. Taw, R. C. Rohde, M. N. Dods, W. DeSnoo, K. R. Meihaus, T. D. Harris, J. B. Neaton and J. R. Long, Chem. Sci., 2022, 13, 10216–10237 RSC.
- X. Li, S. Li, J. Liu, J. Zhang, Y. Ren and J. Zhao, RSC Adv., 2024, 14, 20780–20785 RSC.
- J. Wang, H. Wang, X. Qi, G. Zhi and J. Wang, Environ. Sci. Pollut. Res., 2024, 31, 32935–32949 CrossRef CAS.
- K. F. Kayani, RSC Adv., 2024, 14, 31777–31796 RSC.
- D. Li, A. Yadav, H. Zhou, K. Roy, P. Thanasekaran and C. Lee, Global Challenges, 2024, 8, 2300244 CrossRef PubMed.
- A. S. Rosen, M. R. Mian, T. Islamoglu, H. Chen, O. K. Farha, J. M. Notestein and R. Q. Snurr, J. Am. Chem. Soc., 2020, 142, 4317–4328 CrossRef CAS PubMed.
- L. Chen, H.-F. Wang, C. Li and Q. Xu, Chem. Sci., 2020, 11, 5369–5403 RSC.
- B. Chen, Z. Yang, Y. Zhu and Y. Xia, J. Mater. Chem. A, 2014, 2, 16811–16831 RSC.
- P. Kukkar, K.-H. Kim, D. Kukkar and P. Singh, Coord. Chem. Rev., 2021, 446, 214109 CrossRef CAS.
- H. Mahdavi, M. M. Sadiq, S. J. D. Smith, X. Mulet and M. R. Hill, J. Mater. Chem. A, 2023, 11, 16846–16853 RSC.
- B. Yao, S.-K. Lua, H.-S. Lim, Q. Zhang, X. Cui, T. J. White, V. P. Ting and Z. Dong, Microporous Mesoporous Mater., 2021, 314, 110777 CrossRef CAS.
- S. Al Abdulla, R. Sabouni, M. Ghommem and A. H. Alami, Heliyon, 2023, 9, e21349 CrossRef CAS PubMed.
- F. Hillman, J. M. Zimmerman, S.-M. Paek, M. R. A. Hamid, W. T. Lim and H.-K. Jeong, J. Mater. Chem. A, 2017, 5, 6090–6099 RSC.
- H. Mahdavi, A. Robin, N. T. Eden, A. Khosravanian, M. M. Sadiq, K. Konstas, S. J. D. Smith, X. Mulet and M. R. Hill, Langmuir, 2024, 40, 17387–17395 CrossRef CAS PubMed.
- J. Gandara-Loe, R. Bueno-Perez, A. Missyul, D. Fairen-Jimenez and J. Silvestre-Albero, ACS Appl. Nano Mater., 2021, 4, 3519–3528 CrossRef CAS.
- T. Tian, M. T. Wharmby, J. B. Parra, C. O. Ania and D. Fairen-Jimenez, Dalton Trans., 2016, 45, 6893–6900 RSC.
- C. Zheng, D. Liu, Q. Yang, C. Zhong and J. Mi, Ind. Eng. Chem. Res., 2009, 48, 10479–10484 CrossRef CAS.
- T. Xiao and D. Liu, Mater. Today Energy, 2019, 14, 100357 CrossRef.
- P. Zhao, G. I. Lampronti, G. O. Lloyd, M. T. Wharmby, S. Facq, A. K. Cheetham and S. A. T. Redfern, Chem. Mater., 2014, 26, 1767–1769 CrossRef CAS PubMed.
- P. Zhao, G. I. Lampronti, G. O. Lloyd, E. Suard and S. A. T. Redfern, J. Mater. Chem. A, 2014, 2, 620–623 RSC.
- J. Song, R. Pallach, L. Frentzel-Beyme, P. Kolodzeiski, G. Kieslich, P. Vervoorts, C. L. Hobday and S. Henke, Angew. Chem., Int. Ed., 2022, 61, e202117565 CrossRef CAS PubMed.
- D. Fairen-Jimenez, R. Galvelis, A. Torrisi, A. D. Gellan, M. T. Wharmby, P. A. Wright, C. Mellot-Draznieks and T. Düren, Dalton Trans., 2012, 41, 10752–10762 RSC.
- D. Fairen-Jimenez, S. A. Moggach, M. T. Wharmby, P. A. Wright, S. Parsons and T. Düren, J. Am. Chem. Soc., 2011, 133, 8900–8902 CrossRef CAS PubMed.
- R. S. Madsen, M. Stepniewska, Y. Yang, A. Qiao, W. M. Winters, C. Zhou, J. König, J. C. Mauro and Y. Yue, RSC Adv., 2022, 12, 10815–10824 RSC.
- J. G. Vitillo and L. Gagliardi, Chem. Mater., 2021, 33, 4465–4473 CrossRef CAS.
- P. Krokidas, S. Moncho, E. N. Brothers, M. Castier and I. G. Economou, Phys. Chem. Chem. Phys., 2018, 20, 4879–4892 RSC.
- A. Awadallah-F, F. Hillman, S. A. Al-Muhtaseb and H.-K. Jeong, J. Nanomater., 2019, 2019, 6130152 Search PubMed.
- R. D. Shannon, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- I. Bechis, A. F. Sapnik, A. Tarzia, E. H. Wolpert, M. A. Addicoat, D. A. Keen, T. D. Bennett and K. E. Jelfs, Chem. Mater., 2022, 34, 9042–9054 CrossRef CAS PubMed.
- B. Shen, B. Wang, L. Zhu and L. Jiang, Nanomaterials, 2020, 10, 1636 CrossRef CAS PubMed.
- Q. Ma, H. Jin and Y. Li, Chem. – Eur. J., 2020, 26, 13137–13141 CrossRef CAS PubMed.
- E. D. Bloch, L. J. Murray, W. L. Queen, S. Chavan, S. N. Maximoff, J. P. Bigi, R. Krishna, V. K. Peterson, F. Grandjean, G. J. Long, B. Smit, S. Bordiga, C. M. Brown and J. R. Long, J. Am. Chem. Soc., 2011, 133, 14814–14822 CrossRef CAS.
- D. Meroni, R. Djellabi, M. Ashokkumar, C. L. Bianchi and D. C. Boffito, Chem. Rev., 2022, 122, 3219–3258 CrossRef CAS PubMed.
- N. A. Khan and S. H. Jhung, Coord. Chem. Rev., 2015, 285, 11–23 CrossRef CAS.
- K. Behrens, S. S. Mondal, R. Nöske, I. A. Baburin, S. Leoni, C. Günter, J. Weber and H.-J. Holdt, Inorg. Chem., 2015, 54, 10073–10080 CrossRef CAS PubMed.
- S. Nikmehr, M. Kazemzad, M. M. Sabzehmeidani, L. Nikzad, T. Ebadzadeh and M. Mousaei, Results Chem., 2023, 5, 100973 CrossRef CAS.
- C. Vaitsis, G. Sourkouni and C. Argirusis, Ultrason. Sonochem., 2019, 52, 106–119 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |