Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Carbon-supported NiS catalysts for sustainable transfer-hydrogenation reactions of nitroarenes
Ankit
Kumar
a,
Pascal A.
Roll
a,
Anna
Stephan
a,
Claus
Feldmann
 a,
Arjun
Neyyathala
a,
Paolo
Dolcet
b and
Schirin
Hanf
a,
Arjun
Neyyathala
a,
Paolo
Dolcet
b and
Schirin
Hanf
 *a
*a
aInstitute of Inorganic Chemistry, Karlsruhe Institute of Technology, Engesserstr. 15, 76131 Karlsruhe, Germany. E-mail: Schirin.hanf@kit.edu
bDepartment of Chemical Sciences, University of Padova, Via Francesco Marzolo 1, Padova 35131, Italy
First published on 2nd April 2026
Abstract
We report Ni3S2/C as an effective and environmentally friendly catalyst for the transfer hydrogenation of nitroarenes, using hydrazine monohydrate as a hydrogen donor under mild reaction conditions. The catalyst achieves complete conversion and excellent selectivity across a wide range of nitrobenzene derivatives, bearing both electron-withdrawing and electron-donating groups. Mechanistic studies indicate mainly a direct reduction pathway from nitrobenzene to aniline via nitrosobenzene, unlike Ni/C, which favours a condensation route. The incorporation of sulphur into Ni is shown to tune the selectivity and enhance sulfur tolerance, thereby enabling efficient hydrogenation of nitroarenes containing thiol groups. Moreover, Ni3S2/C is recyclable and effective on a gram-scale reaction, maintaining full conversion and high aniline yields. Compared to conventional noble- and non-noble-metal catalysts that require high-pressure molecular hydrogen, this system offers a cost-effective, robust, and sustainable alternative with strong potential for industrial-scale applications.
1. Introduction
Aniline and its derivatives are crucial intermediates in the production of fine chemicals, such as pharmaceuticals, agrochemicals, dyes, and polymers.1,2 Due to their high industrial demand, the development of cost-effective and sustainable methods for the synthesis of aniline and related compounds from readily available raw materials remains a key focus. The catalytic hydrogenation of nitroarenes has been a key method for producing aniline and its derivatives for over a century, valued for its operational simplicity, environmental compatibility, and adaptability to both laboratory- and industrial-scale applications.3–5 Traditionally, the hydrogenation of organic compounds has been carried out either through direct hydrogenation using molecular hydrogen6–11 or via catalytic transfer hydrogenation reactions,12–17 in which hydrogen is provided by alternative donor molecules instead of gaseous hydrogen. However, conventional hydrogenation methods face several challenges, including the high cost of hydrogen compression, transportation difficulties, safety risks, and the need for complex reactor systems with strict operating conditions.18–20 As a result, transfer hydrogenation reactions have gained significant attention, due to their operational simplicity, reduced equipment requirements, and the gradual and controlled release of hydrogen during the catalytic reactions.18–20 Transfer hydrogenation reactions commonly employ metal hydride reagents, such as sodium borohydride (NaBH4) and lithium aluminium hydride (LiAlH4). However, these approaches suffer from several drawbacks, including a high reagent consumption, poor atom economy, substantial metal salt waste generation, and complex product separation procedures.21,22 These drawbacks, together with the high cost of metal hydride reagents at industrial scales, significantly limit their practical application in selective hydrogenation reactions. Consequently, recent research has shifted towards the development of alternative hydrogen sources, like formic acid, alcohols, amine–boranes and hydrazine monohydrate, as more stable derivative to anhydrous hydrazine.12–17,23 Hydrazine monohydrate is particularly attractive for transfer hydrogenation reactions because it allows transformations to be carried out under mild, pressure-free conditions. By eliminating the need for high-pressure hydrogen gas and specialized equipment, it simplifies laboratory-scale studies, facilitates mechanistic investigations, and enables rapid catalyst screening. Although its large-scale application is limited by toxicity and handling concerns, hydrazine monohydrate remains a valuable and well-established model hydrogen donor. In contrast, molecular hydrogen is generally preferred for industrial processes because of its clean by-product profile and the availability of established infrastructure.To selectively activate hydrazine monohydrate and control its decomposition into nitrogen and hydrogen under mild reaction conditions, various metal-based catalytic systems have been developed.24,25 Currently, both homogeneous and heterogeneous catalysts are employed in transfer hydrogenation reactions. However, heterogeneous catalysts offer distinct advantages over their homogeneous counterparts, particularly in terms of catalyst separation and recovery.26,27 In this context, noble metal-based heterogeneous catalysts, such as Ru-OMC-800 (OMC: ordered mesoporous carbon),28 Ru/PNC-700 (PNC: phosphorous- and nitrogen-doped carbon),29 Ru/MoS2,30 Ir-MoOx/SiO2,31 Pd/CeO2,32 Pd/C,33 Pt/CeO2,34 ZrO2@Pt,35 Pt/TiO2/SBA-15,36 and Rh/COF (COF: covalent organic framework),37 have been explored. An overview is given in Table S1. While these catalysts exhibit excellent catalytic activity, their poor selectivity, high cost, and limited availability restrict their application in large-scale industrial applications.
To address these limitations, non-noble-metal-based heterogeneous catalysts have been developed for the chemoselective transfer hydrogenation of nitroarenes (Table S1). For example, iron-based materials, such as Fe@C,38 FeCeO2,39 Fe2O3/LRC-700 (LRC: lignin-derived carbon),40 and Fe–Fe3C–Fe3O4@CN (CN: nitrogen-doped carbon),41 as well as immobilized Fe in ionic liquids,42 and ultrasmall FeS2 nanoparticles,43 have demonstrated quantitative conversion and excellent selectivity toward anilines, typically using hydrazine monohydrate as the hydrogen source. Also, nickel-based catalysts, such as Ni/TiO2,44 Ni/C60,45 Ni–NC-700,46 Ni2P/AC (AC: activated carbon),47 and Ni/SiO2,48 have been reported in this context. For example, Sharma et al. developed Ni2P/AC nanocomposites and demonstrated their catalytic activity in the reduction of aromatic nitro compounds.47 Using hydrazine monohydrate as the hydrogen source at 70 °C, complete conversion of nitrobenzene (100%) and 93% yield of aniline were achieved in 2 h. Jiang et al. prepared highly dispersed Ni nanoparticles on SiO2 (Ni/SiO2) via atomic layer deposition. Under hydrazine-assisted hydrogenation conditions at 100 °C, an aniline yield of 45% was obtained after 2 h, which increased to 94% after 8 h reaction time.48 In parallel, copper-based systems (Cu/Celite),49 and a variety of cobalt-based architectures, ranging from supported nanoparticles to single-atom catalysts embedded in N-doped carbon matrices, such as Co@NC,50 Co@NC/AC,51 Co@NCT (NCT: nitrogen-doped carbon nanotube),52 Co@C-800-AL,53 Co-L@C-800,54 Co/NPC,55 Co/MA-800 (MA: macroalgae-derived carbon),56 Co@C/CNT,57 and Co@NC-600,58 have enabled efficient nitroarene reduction, employing alternative hydrogen donors such as methanol or formic acid. Despite these advances, many reported systems rely on elevated temperatures, long reaction times, or highly specific reaction environments. For instance, Dey et al. investigated a single-atom cobalt catalyst (Co@C-800-AL) for the transfer hydrogenation of nitroarenes using methanol as the hydrogen source. For nitrobenzene, complete conversion and 95% yield of aniline were achieved at 140 °C in 24 h with Cs2CO3 as base.53
The catalytic performance of these non-noble metal catalysts can be structurally and electronically tuned by the incorporation of p-block elements to form alloys or intermetallic compounds. Here, the formation of isolated active metal sites, in analogy to the well-known concept of frustrated single sites in homogenous catalysis, has been proposed.59 In this context, metal phosphides and sulphides have demonstrated very promising catalytic activity. For example, metal phosphides (MxPy; M = Ni, Co, Ru, Pd, and Pt) have been applied in hydrogenation reactions,60 water splitting,61 coupling62 and oxidation reactions,63 as well as the deoxygenation of sulfoxides.64 Similarly, metal sulphides, such as MoS2, WS2, RuS2, PdSx, and RhSx, have shown efficiency in hydrodesulfurization reactions,65 CO2 hydrogenation,66 and hydroformylation reactions.67 Although sulphur is typically considered to be a catalyst poison in heterogeneous catalysis,68 sulphide-based catalysts (e.g., MoS2, Co–Mo–S, and Ru–W–S) have successfully been employed in the hydrogenation of sulphur-containing nitroaromatic compounds. For instance, Corma et al. developed a Co–Mo–S catalyst capable of hydrogenating 4-nitrothioanisole to 4-aminothioanisole with complete conversion at 150 °C under 11 bar H2.69 More recently, Glorius et al. reported a Ru–W–S catalyst for the hydrogenation of sulphur-containing quinolines to tetrahydroquinolines.70 Zhang et al. synthesized a PdxSy/C catalyst in-situ for the hydrogenation of sulphur-containing nitroarenes to the corresponding amines.71 Similarly, Yu et al. explored Pd4S nanoparticles supported on sulphur-doped carbon (Pd4S/SC) for the selective hydrogenation of 4-chloronitrobenzene at 80 °C under 6 bar H2.72 Despite the high activity and selectivity of the before-mentioned catalysts, their reliance on high-pressure molecular hydrogen poses sustainability and safety challenges.
Building on the successful p-block/d-metal element combination strategy, we report the synthesis of a nickel sulphide-based catalyst (Ni3S2/C) for the transfer hydrogenation of nitroarenes. Ni3S2/C was employed under base-free conditions using hydrazine hydrate as the hydrogen source, thereby avoiding the need for molecular hydrogen. At this point, it is important to note that hydrazine monohydrate is toxic, corrosive, and potentially carcinogenic. Therefore, its handling requires strict safety measures, including the use of appropriate personal protective equipment (PPE), working in a well-ventilated fume hood, and adherence to institutional safety protocols. The reaction conditions were systematically optimised to maximise the product yield by varying key parameters. This work highlights the potential of nickel sulphide-based catalysts in sustainable organic transformations and opens avenues for large-scale applications in the chemical industry.
2. Experimental section
2.1. Reagents
Nickel chloride hexahydrate (Sigma-Aldrich, >98%), thiourea (Sigma-Aldrich, 99%), carbon (BASF SE), ZrO2 (BASF SE), Al2O3 (BASF SE), SiO2 (Cariact), hydrazine monohydrate (TCI, >98%), n-heptane (TCI, >99%), ethyl acetate (Fischer Scientific, 98%), n-decane (abcr, >99%), methanol (VWR, 99.8%), ethanol (Carl Roth, >99.5%), formic acid (Merk, 91%), nitrobenzene (Merck, >99%), (TCI, 98%), 2-chloronitrobenzene (TCI, 99%), 4-chloronitrobenzene (TCI, >98%), 2-nitrophenol (TCI, 98%), 4-nitrophenol (TCI, >99%), 4-nitrotoluene (TCI, >99%), 2-nitroanisole (TCI, >98%), 4-nitroanisole (>98%), 4-nitrobenzonitrile (TCI, 98%), 4-fluoronitrobenzene (TCI, >98%), 1,4-dinitrobenzene (TCI, 99%), 4-nitrothioanisole (abcr, 98%), 4-nitrophenyl phenyl sulphide (Thermo Scientific, 98%), 4-nitrothiophenol, 4-nitrobenzenethiol (TCI, >95%), nitroso benzene (TCI, 98%), and azobenzene (TCI, >98%) were purchased and used without any further purification. For the thermal treatment during catalyst synthesis, 5%H2/N2 (99.999%) was used and purchased from Air Liquide.2.2. Characterisation
Powder X-ray diffraction (PXRD) measurements were carried out using a Stoe STADI-MP diffractometer operating with a Ge-monochromatized Cu Kα radiation (λ = 1.541 Å) in transmission mode. For the measurements, about 5–10 mg of sample was placed between thin films of scotch tape and acetate paper, which were then placed into the sample holder. The measurements were carried out in the range of 2 to 92° with a step size of 3°. The shape, size, and chemical composition of the Ni3S2/C sample were studied by high-angle annular dark-field (HAADF) scanning transmission electron microscopy (STEM) combined with energy-dispersive X-ray spectroscopy (EDXS). The experiments were conducted with a FEI Tecnai Osiris microscope at 200 keV electron energy, which is equipped with a Super-X EDXS system comprising four silicon drift detectors. EDXS elemental maps were recorded and used to investigate the element distribution within the respective sample. The maps were analysed using the ESPRIT software (version 2.3) from Bruker. For the sample preparation, the solid sample was dispersed in ethanol under ultrasonication and then deposited over the amorphous carbon-coated 400 mesh Cu grid and dried under vacuum at 80 °C overnight. Thermogravimetric analysis (TGA) of the carbon support and the Ni3S2/C catalyst was performed over a temperature range of 30–1200 °C under air at a heating rate of 5 K min−1 using a NETZSCH STA 449 F3 Jupiter instrument. Raman spectroscopy was performed with an inVia Raman spectrometer (Renishaw) equipped with a frequency doubled Nd:YAG laser (532 nm, 100 mW) and a 50× Leica DM2500 optical microscope. A laser power of 10% was employed together with a 600 lines per mm grating. Spectra were recorded from 6 different points with an acquisition time of 30 s. All obtained spectra for each sample were converged. The software WiRE 4.4 (Renishaw) was used for the data treatment. X-ray photoelectron spectroscopy (XPS) was performed with a Thermo Scientific Escalab QXi. The spectra were collected employing a monochromatic Al Kα source (λ = 1486.6 eV), with a spot size of 650 µm and using a flood gun for charge compensation. For the XPS measurements, the catalyst was stored under an inert atmosphere in a glove box and immediately transferred to the XPS instrument with minimal exposure to air. The measurements were performed without any additional pre-treatment. The survey spectra were collected (0–1350 eV) with a pass energy of 100 eV and a step size of 1 eV, whereas detailed regions were collected with a pass energy of 20 eV and a step size of 0.05 eV. The specific surface area was measured by N2 physisorption using the Brunauer–Emmett–Teller (BET) method. The measurements were carried out with an AUTOSORB IQ-XR VITON analyzer (Anton Paar). The samples were treated at 150 °C for 3 h under reduced pressure before the measurement. The metal loading of the catalyst was determined via inductive coupled plasma optical emission spectroscopy (ICP-OES) measurements using an iCAP7600 DUO from Thermo-Fisher-Scientific. Before analysis, the samples were digested using acids and high pressure.2.3. Catalyst syntheses
Nickel sulphide nanoparticles supported on carbon (Ni3S2/C) were synthesized via a wet impregnation method followed by thermal treatment. For the preparation of a 10 wt% Ni catalyst, 225 mg of carbon (finely ground) was dispersed in 15 mL of distilled water in a 50 mL round-bottom flask and sonicated for 20 minutes. Subsequently, NiCl2·6H2O (0.425 mmol) and thiourea (2.55 mmol) were added to the suspension, and the mixture was stirred at room temperature for 2 h. The mixture was then heated at 70 °C overnight to remove the solvent completely. The resulting precursor was subjected to thermal treatment at 400 °C under a continuous flow of 5% H2/N2 gas at a heating rate of 5 °C min−1 for 4 h to obtain the Ni3S2/C catalyst. Similarly, other supported catalysts (Ni3S2/ZrO2, Ni3S2/Al2O3 and Ni3S2/SiO2) were also synthesized. For comparison, a reference Ni/C catalyst (10 wt% Ni) was prepared using the same procedure, excluding the addition of thiourea, and applying a slightly modified thermal treatment (500 °C, 5 °C min−1, 5 h).2.4. Catalytic testing
The catalytic performance was evaluated using nitrobenzene as model substrate. In a typical catalytic test, nitrobenzene (0.97 mmol, 100 µL), n-heptane (2 mL), and the catalyst (30 mg) were placed in a 25 mL Schlenk round-bottom flask. It is important to note that the size of the flask has an impact on the performance of the catalysts, since it affects the gas atmosphere of the head space. The mixture was sonicated for several minutes to ensure the homogeneous dispersion of the catalyst. Subsequently, hydrazine monohydrate (10 equiv.) was added, the flask was sealed with a stopper, and the Schlenk valve was closed. The reaction mixture was then heated to 70 °C in a preheated oil bath and stirred at 600 rpm for the desired reaction time. After completion of the reaction, the Schlenk valve was carefully opened to release any gas generated during the reaction, and subsequently the mixture was allowed to cool to room temperature. Ethyl acetate (2 mL) and n-decane (50 µL, used as an external standard) were added, and the resulting biphasic mixture was thoroughly mixed. The organic phase was then filtered through a 0.2 µm syringe filter, and 100 µL of the filtrate was diluted with 900 µL of methanol or ethyl acetate to obtain a homogeneous solution for further analysis. Product identification and quantification was performed using an Agilent GC-MS system comprising a 8860 GC system coupled with a 5977B mass selective detector (MSD). From the GC vial, 1 µL was automatically injected into the GC-MS system at a plunger speed of 1000 µL min−1, using a split ratio of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and an injection temperature of 300 °C. Chromatographic separation was achieved using a fused silica capillary column (inner diameter: 0.25 mm; film thickness: 0.25 µm; length: 30 m). The oven temperature program was as follows: initial hold at 50 °C for 2.25 min, ramping at 25 °C min−1 to 250 °C for 2 min, followed by a final hold at 300 °C for 1.0 min.
1 and an injection temperature of 300 °C. Chromatographic separation was achieved using a fused silica capillary column (inner diameter: 0.25 mm; film thickness: 0.25 µm; length: 30 m). The oven temperature program was as follows: initial hold at 50 °C for 2.25 min, ramping at 25 °C min−1 to 250 °C for 2 min, followed by a final hold at 300 °C for 1.0 min.
2.5. Recyclability studies
The catalytic reaction was performed as specified before. After the completion of the reaction, the catalyst was recovered from the reaction mixture by centrifugation, washed three times with ethyl acetate and acetone, dried overnight at 60 °C, and then reused for the next catalytic run.2.6. Gram scale reaction
To a 100 mL Schlenk round-bottom flask, nitrobenzene (19.4 mmol), Ni3S2/C (600 mg), n-heptane (40 mL), and hydrazine monohydrate (10 equiv.) were added and stirred at 70 °C at 600 rpm for the specified time. After completion, the reaction mixture was cooled to room temperature. Ethyl acetate (40 mL) and n-decane (1 mL, used as an external standard) were added to the reaction mixture. The organic phase of the mixture was then filtered through a 0.2 µm syringe filter, and 100 µL of the filtrate was diluted with 900 µL of ethyl acetate to prepare a homogeneous solution for further product analysis via GC-MS.3. Results and discussion
3.1. Catalyst characterisation
Nickel sulphide nanoparticles supported on carbon (Ni3S2/C) were synthesized via a wet impregnation method followed by a thermal treatment. To prepare a 10 wt% Ni-based catalyst, nickel chloride and thiourea (in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 molar ratio) were added to a carbon suspension in demineralised water. The mixture was stirred at room temperature for 2 h. The as-prepared material was then dried overnight and thermally treated under a continuous flow of 5% H2 in N2 to form the Ni3S2/C catalyst. Similarly, Ni/C (10 wt% Ni) was prepared as the reference catalyst using the same procedure, excluding the addition of thiourea, and applying a slightly modified thermal treatment (500 °C, 5 °C min−1, and 5 h). The thermal treatment conditions were intentionally selected based on the distinct chemical requirements of each catalyst system. For the Ni3S2/C catalyst, the thermal treatment at 400 °C (5 °C min−1, 4 h) was applied to promote the controlled sulfidation and stabilization of the Ni3S2 phase. Higher temperatures or stronger reducing environments can induce sulphur loss73 or partial reduction to metallic Ni. In contrast, the Ni/C catalyst requires the complete reduction of the Ni2+ precursor to metallic Ni, for which higher temperatures (500 °C) are required.
6 molar ratio) were added to a carbon suspension in demineralised water. The mixture was stirred at room temperature for 2 h. The as-prepared material was then dried overnight and thermally treated under a continuous flow of 5% H2 in N2 to form the Ni3S2/C catalyst. Similarly, Ni/C (10 wt% Ni) was prepared as the reference catalyst using the same procedure, excluding the addition of thiourea, and applying a slightly modified thermal treatment (500 °C, 5 °C min−1, and 5 h). The thermal treatment conditions were intentionally selected based on the distinct chemical requirements of each catalyst system. For the Ni3S2/C catalyst, the thermal treatment at 400 °C (5 °C min−1, 4 h) was applied to promote the controlled sulfidation and stabilization of the Ni3S2 phase. Higher temperatures or stronger reducing environments can induce sulphur loss73 or partial reduction to metallic Ni. In contrast, the Ni/C catalyst requires the complete reduction of the Ni2+ precursor to metallic Ni, for which higher temperatures (500 °C) are required.
The phase of the synthesized Ni3S2/C was examined by powder X-ray diffraction (PXRD), as shown in Fig. 1. The PXRD pattern exhibits characteristic peaks at 2θ values of 21.7°, 31.0°, 37.7°, 44.3°, 49.7°, and 55.1°, corresponding to the (010), (110), (111), (020), (120), and (121) planes, respectively. These reflections of Ni3S2/C are consistent with the heazlewoodite phase as a reference (JCPDS No. 01-071-1682).74
Raman spectra of the carbon support and Ni3S2/C composite exhibit the characteristic D band (∼1350 cm−1), associated with structural defects, and the G band (∼1600 cm−1), corresponding to graphitic sp2 carbon domains (Fig. S2a). The carbon support shows an ID/IG ratio of 0.96, indicative for partially disordered activated carbon. After deposition of Ni3S2 nanoparticles, the ID/IG ratio slightly increases to 0.99, indicating a minor increase in structural disorder, while preserving the overall carbon framework.75 Thermogravimetric analysis (TGA) demonstrates that both the support and the nickel sulfide catalyst are thermally stable below 400 °C. A noticeable mass loss is observed between 450 and 750 °C, attributable to carbon decomposition.76
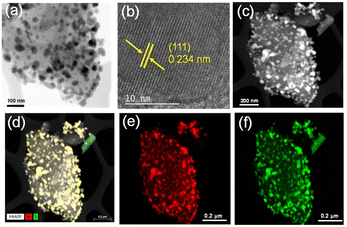
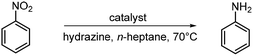
To analyse the size, structure and composition of Ni3S2/C, transmission electron microscopy (TEM) was carried out (Fig. 2) and particles with an average size of 39 ± 5.2 nm (Fig. S1) were observed. High-resolution (HR)-TEM images show well-ordered lattice fringes over the whole nanoparticle. A d-spacing of 0.234 nm (Fig. 2b) corresponds to the (111) plane of bulk Ni3S2 as a reference.74 Additionally, high-angle annular dark-field scanning (HAADF-S)TEM with energy-dispersive X-ray spectroscopy (EDXS) and element mapping (Fig. 2c–f) confirm a uniform distribution of Ni and S, and thus, of the active phase over the whole carbon support.
To verify the elemental composition, inductively coupled plasma atomic emission spectroscopy (ICP-AES) was performed. The analysis shows 9 wt% Ni and 3 wt% S, and a total of 12 wt% of the active phase on carbon. These values are consistent with the theoretical stoichiometry of Ni3S2 (Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S = 3
S = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). An analysis of the specific surface area, following the Brunauer–Emmett–Teller (BET) approach, further confirms the successful incorporation of Ni3S2 onto the porous carbon matrix. The pristine carbon support exhibits a high surface area of 1319 m2 g−1 and a pore volume of 0.60 mL g−1, while the Ni3S2 loaded sample shows reduced values of 418 m2 g−1 and 0.18 mL g−1, respectively. This decrease indicates that Ni3S2 nanoparticles partially occupy/block the pores of the carbon support.
2). An analysis of the specific surface area, following the Brunauer–Emmett–Teller (BET) approach, further confirms the successful incorporation of Ni3S2 onto the porous carbon matrix. The pristine carbon support exhibits a high surface area of 1319 m2 g−1 and a pore volume of 0.60 mL g−1, while the Ni3S2 loaded sample shows reduced values of 418 m2 g−1 and 0.18 mL g−1, respectively. This decrease indicates that Ni3S2 nanoparticles partially occupy/block the pores of the carbon support.
X-ray photoelectron spectroscopy (XPS) was employed to investigate the surface electronic structure of the Ni3S2/C catalyst (Fig. 3). The high-resolution Ni 2p spectrum (Fig. 3a) displays two primary spin-orbit doublets, corresponding to the Ni 2p3/2 and Ni 2p1/2 components. The peaks located at 855.5 and 872.9 eV are attributed to Ni2+ species, while the other peaks at 856.8 and 875.2 eV are assigned to Ni3+ species.77 The signals at 852.9 and 870.5 eV correspond to metallic Ni0.78 Furthermore, the satellite signals observed at 861.3 and 864.7 eV in the Ni 2p3/2 region and at 879.8 and 881.7 eV in the Ni 2p1/2 region are characteristic shake-up satellite peaks associated with Ni2+ species. The high-resolution S 2p spectrum (Fig. 3b) exhibits a dominant doublet centred at 161.8 eV (S 2p3/2) and 162.9 eV (S 2p1/2), which is characteristic of S2− species in Ni3S2/C. Additional peaks at 163.7 and 165.0 eV are attributed to surface polysulfide (Sn2−) species. A weak peak at 167.9 eV is assigned to sulphate species, likely formed due to surface oxidation of sulphur upon exposure to air. This is a well-known observation for sulfide-based materials.79 These results collectively confirm the presence of metal–sulphur bonding and are consistent with the characteristic electronic structure of Ni3S2 supported on carbon.
3.2. Catalytic transfer hydrogenation of nitrobenzene
The catalytic activity of the supported Ni3S2 catalysts (10 wt% Ni) was evaluated in the transfer hydrogenation of nitrobenzene to aniline using hydrazine monohydrate as the hydrogen source at 70 °C (Table 1). In the initial blank experiments, the reaction was conducted without a base and with carbon as the support (without any Ni) and no conversion was observed (Table 1, entries 1 and 2). This clearly indicates that the presence of a metal catalyst is essential for the transfer hydrogenation reaction.| Entry | Catalyst | t (h) | Conversion (%) | Aniline yield (%) |
|---|---|---|---|---|
| a Reaction conditions: nitrobenzene (0.97 mmol, 0.1 mL), catalyst (30 mg, 10 wt% Ni), n-heptane (2 mL), 70 °C, and hydrazine monohydrate (10 equiv.). Conversions and yields were determined by GC-MS with n-decane as an external standard. | ||||
| 1 | Without catalyst | 2 | — | — |
| 2 | Carbon support | 2 | — | — |
| 3 | Ni3S2/SiO2 | 2 | 74 | 15 |
| 4 | Ni3S2/Al2O3 | 2 | 93 | 20 |
| 5 | Ni3S2/ZrO2 | 2 | >99 | 26 |
| 6 | Ni3S2/C | 2 | >99 | 64 |
| 7 | Ni3S2/C | 5 | >99 | 85 |
| 8 | Ni3S2/C | 6 | >99 | 91 |
| 9 | Ni/C | 2 | >99 | 31 |
| 10 | Ni/C | 6 | >99 | 54 |
Subsequently, various supported Ni3S2 catalysts were screened using different support materials, such as ZrO2, Al2O3, and SiO2. These catalysts enabled nitrobenzene conversions ranging from 74% to >99%, with aniline yields between 15 and 26% (Table 1, entries 3–5). Notably, Ni3S2 supported on carbon (Ni3S2/C) showed superior performance with respect to Ni3S2 on other support materials, achieving complete nitrobenzene conversion (>99%) and a 64% aniline yield within 2 h at 70 °C (Table 1, entry 6). Extending the reaction times to 5 and 6 h, respectively, further improved the aniline yield to 85% and 91% (Table 1, entries 7 and 8). For comparison, the reaction was also performed using a sulphur-free Ni/C catalyst (10 wt% Ni) as reference system, which resulted in complete nitrobenzene conversion, but only 31% aniline yield in 2 h (Table 1, entry 9). In this reaction, azobenzene and azoxybenzene were identified as by-products (Fig. S8). Although the aniline yield could be improved to 54% after 6 h with Ni/C, the sulphide-containing catalyst (Ni3S2/C) achieved a significantly higher yield of 91%, clearly outperforming the sulphur-free Ni/C reference catalyst (Table 1, entries 8 and 10). The productivity of aniline over the Ni3S2/C catalyst was calculated (at 6 h) to be 4.9 mmol g−1 h−1, whereas the Ni/C catalyst exhibited a lower productivity of 2.9 mmol g−1 h−1 under identical reaction conditions. This ∼1.7-fold enhancement in productivity quantitatively highlights the crucial role of sulphur incorporation in improving catalytic performance for the transfer hydrogenation of nitrobenzene to aniline.
Given the exceptional catalytic performance of Ni3S2 on carbon in the transfer hydrogenation of nitrobenzene to aniline, it was selected as the catalyst for further reaction optimization studies. These investigations focused on the effects of reaction temperature, reaction time, amount of hydrazine monohydrate, and type of hydrogen donor (Fig. 4). Each reaction parameter was varied individually, while keeping other reaction conditions constant to understand its sole influence on the catalytic performance.
Temperature optimization was carried out in the range of 30–70 °C using nitrobenzene as the model substrate and hydrazine monohydrate as the hydrogen source (Fig. 4a and Table S2). At 30 °C, no conversion was observed. Increasing the temperature to 40 °C resulted in 21% conversion of nitrobenzene. Further temperature increments from 40 to 70 °C led to progressively higher conversion rates and improved aniline yields.
To investigate the effect of catalyst amount, reactions were performed using varying amounts of Ni3S2/C (0.87–5.2 mol%) under standard reaction conditions (70 °C, 2 h, n-heptane as the solvent, Fig. 4b and Table S3). Using 0.87 mol% of catalyst resulted in a 65% nitrobenzene conversion and 35% yield of aniline. Increasing the catalyst amount to 1.73–5.2 mol% enhanced both conversion and aniline yield (33–64%), likely due to the greater availability of catalytically active Ni sites.
The influence of hydrazine monohydrate quantity was examined in the range of 2 to 10 equiv. (Fig. 4c and Table S4). Here, a direct correlation was observed between the amount of hydrazine and aniline yield, which increased from 23 to 64%. This improvement can be attributed to the greater availability of hydrogen gas, which accelerates the reduction of nitrobenzene and its intermediates. A higher hydrazine concentration also enhances reaction kinetics, suppresses side reactions leading to condensation by-products, and maintains a strongly reducing environment throughout the reaction.
Additionally, various other hydrogen donors were explored, including isopropanol, methanol, ethanol, formic acid, and hydrazine monohydrate (Fig. 4d and Table S5). Among these, only hydrazine monohydrate effectively facilitated the hydrogenation of nitrobenzene to aniline.
3.3. Mechanistic investigations
After optimizing the reaction parameters, time-dependent studies were conducted to identify possible intermediates during the transfer hydrogenation of nitrobenzene over Ni/C and Ni3S2/C as catalysts (Fig. 5).For Ni/C, extending the reaction time from 0.5 h to 2 h resulted in an increase in aniline yield and azobenzene selectivity, accompanied by a decrease in azoxybenzene selectivity. Notably, nitrosobenzene was not detected at any stage using Ni/C as the catalyst. In contrast, when the reaction was performed with Ni3S2/C, the yield of aniline increased steadily with time, while the selectivity of other intermediates remained largely unchanged. Interestingly, nitrosobenzene was consistently observed as an intermediate throughout the reaction over Ni3S2/C.
To further probe the mechanistic pathway, control experiments were carried out using nitrosobenzene and azobenzene as substrates under the before optimized reactions conditions (Scheme S1). When nitrosobenzene was used with Ni3S2/C as the catalyst, complete conversion was achieved in 2 h, yielding aniline (91%) and azobenzene (9%). Similarly, over Ni/C, nitrosobenzene was fully converted, producing aniline (82%) and azobenzene (18%). However, when azobenzene was used as the substrate, both catalysts exhibited sluggish activity, with only trace amounts of aniline being detected. These findings, combined with literature reports,45 suggest that the reaction can proceed via two pathways: a direct pathway, involving nitrosobenzene and N-phenylhydroxylamine as intermediates, and a condensation pathway, which involves azoxybenzene and azobenzene (Fig. 6). Our results indicate that the reaction over Ni3S2/C proceeds mainly via a competitive pathway involving nitrosobenzene as an intermediate, whereas condensation pathways are more prominent over Ni/C. Surprisingly, the incorporation of sulphur into the nickel lattice appears to play a crucial role in modulating the reaction pathway, thereby enhancing the aniline selectivity in the transfer hydrogenation of nitrobenzene.
3.4. Recyclability study and gram-scale reaction
The recyclability of the Ni3S2/C catalyst was evaluated under optimized reaction conditions during four consecutive reaction runs (Fig. 7). After each catalytic cycle, the catalyst was recovered by centrifugation, thoroughly washed with ethyl acetate and acetone, and dried before reuse. The results demonstrate that the catalyst can be recycled for up to four consecutive runs with only a slight decrease in activity.The spent Ni3S2/C catalyst, recovered after the first catalytic cycle, was characterized by TEM, PXRD, and XPS to evaluate its bulk and surface stability. TEM analysis revealed only a marginal increase in average particle size from 39 to 40 nm, indicating negligible sintering during the reaction (Fig. S1 and S4). The PXRD pattern of the recovered catalyst showed no detectable phase transformation or formation of new crystalline phases, confirming the preservation of the bulk crystal structure under the reaction conditions (Fig. S5). Furthermore, XPS analysis of the Ni 2p and S 2p regions demonstrated that the surface chemical states of Ni and S species were retained after the reaction (Fig. S6). These findings correlate well with the stable catalytic performance observed during recycling experiments.
To assess the practical applicability of Ni3S2/C as the transfer hydrogenation catalyst, a gram-scale hydrogenation of nitrobenzene was performed by proportionally increasing all reaction components, including the catalyst amount (Scheme S2b). Here, the complete conversion of nitrobenzene (>99%) with 64% aniline yield was achieved in 2 h, which completely aligns with the small-scale reaction reported before. In the gram-scale reaction, the aniline yield could even be enhanced to 92% in 6 h reaction time (Scheme S2c). Under these conditions, the reaction proceeded efficiently, confirming the potential of Ni3S2/C as the catalyst for industrial-scale transfer hydrogenation processes.
3.5. Substrate scope
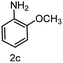
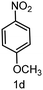
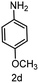
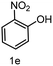
To evaluate the versatility of the Ni3S2/C catalyst, the transfer hydrogenation of various nitroarenes, bearing different functional groups, was investigated under optimized reaction conditions (Table 2). The catalyst showed excellent catalytic activity in the transfer hydrogenation of nitroarenes, containing both electron-donating and electron-withdrawing substituents, regardless of their steric hindrance (Table 2, entries 2–8). Also, halogen-substituted nitroarenes, which are typically prone to dehalogenation,80 were selectively converted to the corresponding aromatic amines with high selectivity (>99%, Table 2, entries 6–8). The transfer hydrogenation of 1,4-dinitrobenzene also proceeded efficiently, affording complete conversion with 60% selectivity towards the desired product (Table 2, entry 9). Furthermore, nitroarenes with additional reducible groups, such as 4-nitrobenzonitrile, underwent the selective reduction of the nitro group to the corresponding amine with 94% selectivity (Table 2, entry 10). Particularly noteworthy is the hydrogenation of nitroarenes with sulphur-containing functional groups, which has been rarely explored in the field of heterogeneous catalysis, due to the strong chemisorption of sulphur atoms on metal surfaces and subsequent catalyst poisoning.81 Despite this challenge, Ni3S2/C efficiently converted sulphur-containing nitroarenes into the corresponding amines (Table 2, entries 11–13). Interestingly, the Ni/C reference catalyst showed significantly lower activity for the hydrogenation of sulphur-containing nitroarenes, such as 4-nitrothioanisole, whereas Ni3S2/C achieved complete conversion with identical reaction conditions (Ni3S2/C: >99% conversion in 6 h versus Ni/C: 65% conversion in 6 h). These results highlight the robustness and high selectivity of Ni3S2/C as an efficient catalyst for the transfer hydrogenation of a broad range of nitroaromatic compounds.| Entry | Substrate | t (h) | Conversion (%) | Product | Selectivity (%) |
|---|---|---|---|---|---|
| a Reaction conditions: substrate (1 mmol), catalyst (30 mg, 10 wt%), n-heptane (2 mL), 70 °C, and hydrazine monohydrate (10 equiv.). Conversions and selectivities were determined by GC-MS with n-decane as an external standard*. | |||||
| 1 |
|
6 | >99 |
|
91 |
| 2 |
|
6 | >99 |
|
>99 |
| 3 |
|
6 | >99 |
|
>99 |
| 4 |
|
9 | 95 |
|
>99 |
| 5 |
|
6 | >99 |
|
>99 |
| 6 |
|
6 | >99 |
|
>99 |
| 7 |
|
7 | >99 |
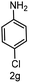
|
>99 |
| 8 |
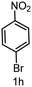
|
6 | >99 |
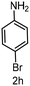
|
>99 |
| 9 |
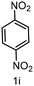
|
6 | >99 |
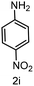
|
60 |
| 10 |
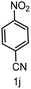
|
6 | >99 |
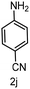
|
94 |
| 11 |
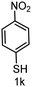
|
6 | >99 |
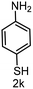
|
>99 |
| 12 |
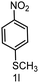
|
6 | >99 |
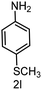
|
>99 |
| 13 |
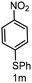
|
6 | >99 |
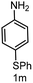
|
>99 |
A detailed comparison of the Ni3S2/C catalyst with previously reported systems is presented in Table S1. The analysis highlights several key advantages of Ni3S2/C over conventional catalysts. Ni3S2/C achieves complete conversion of nitrobenzene under mild reaction conditions (70 °C and atmospheric pressure), whereas many reported approaches require elevated temperatures, high hydrogen pressures, or noble metal catalysts, based on Pd, Pt, Ru, or Rh.28–37 While Fe- and Co-based catalysts (e.g., ImmFe-IL, FeS2 NPs, Fe–Fe3C–Fe3O4@CN, Co@C/CNT, Co-L@C-800, and Co@NCT) often achieve complete conversion and high selectivity with hydrazine or other hydrogen donors, Ni-based systems typically require higher reaction temperatures.44–46,48 When compared to the reported Ni2P/AC system, Ni2P/AC affords an aniline yield of 93% in 2 h in n-heptane, while Ni3S2/C initially reaches a lower aniline yield of 64% under comparable reaction conditions (Table S6).46 However, extending the reaction time to 6 h increases the yield of Ni3S2/C to 91%, demonstrating that high aniline yield/selectivity can be achieved without compromising the mild operating conditions. Importantly, Ni3S2/C uniquely exhibits high catalytic activity toward sulphur-containing nitroarenes, a substrate class that has not been previously addressed by any Ni-based transfer hydrogenation catalysts, including Ni2P/AC. This capability overcomes a well-known limitation of non-noble Ni catalysis and underscores the broad applicability and robustness of the herein reported Ni3S2/C.
4. Conclusions
This work establishes Ni3S2/C as an efficient and sustainable catalyst for the transfer hydrogenation of nitroarenes, using hydrazine monohydrate as the hydrogen source. The catalyst selectively reduces nitro groups in various aromatic substrates under mild reaction conditions, offering a practical alternative to conventional hydrogenation processes that rely on noble metals and molecular hydrogen. Mechanistic studies reveal that Ni3S2/C mainly promotes a direct reaction pathway from nitrobenzene to aniline, involving nitrosobenzene as a key intermediate, whereas Ni/C favours a condensation pathway via azoxybenzene and azobenzene. Control experiments confirmed the underlying reaction pathways and highlighted the critical role of sulphur incorporation into nickel when tuning the selectivity of transfer hydrogenation reactions towards aniline. Investigations of the substrate scope demonstrated the applicability of the system to a wide variety of nitroarene derivatives. Interestingly, Ni3S2/C significantly enhances sulphur resistance, as it also functions as a catalyst for the transfer hydrogenation of thiol-containing nitrobenzene. Beyond these insights, the catalyst demonstrated excellent recyclability, maintaining good activity over multiple catalytic cycles with minimal performance loss. Furthermore, a gram-scale hydrogenation of nitrobenzene to aniline was successfully performed, achieving complete conversion. This result underscores the industrial relevance and scalability of the Ni3S2/C based catalytic system. Compared to traditional catalyst systems, the presented Ni3S2/C catalyst is cost-effective and robust paving the way for broader adoption in fine chemical synthesis and pharmaceutical manufacturing. Future research will focus on expanding the substrate scope, exploring other sulphur-modified transition metal systems, and integrating this methodology into continuous-flow processes for large-scale applications.Conflicts of interest
There are no conflicts to declare.Data availability
Further information is available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01349f. Also, all additional data in open file formats has been uploaded in the NFDI4Cat Central Data Repository Repo4Cat (https://hdl.handle.net/21.11165/4cat/3edu-kaap).Acknowledgements
We thank the Federal Ministry of Research, Technology and Space (BMFTR) for funding through the, CARBO-DIOL2.0 - Von Kohlenhydraten aus sekundären Rohstoffquellen zu Chemierohstoffen“ project (project number: 03XP0612B). We thank Dr. Merve Kurt, Institute for chemical technology and polymer chemistry, KIT, for the Raman measurements. We would also like to thank Silvia Gross, University of Padova, for discussions regarding the XPS measurements.Notes and references
- Y. Chi, S. Zheng, X. Zhang and G. Li, Int. J. Hydrogen Energy, 2021, 46, 36124–36136 CrossRef CAS.
- K. Murugesan, M. Beller and R. V. Jagadeesh, Angew. Chem., Int. Ed., 2020, 59, 17408–17412 CrossRef CAS PubMed.
- H. Miao, H. Shiwei, K. Ma, L. Sun, W. Fufang, H. Wang and H. Li, Catal. Commun., 2018, 109, 33–37 CrossRef CAS.
- Y. M. Yuan, J. Y. Long, H. Xiwei, X. Dan, Y. Zhu and Z. Dong, ChemSusChem, 2018, 11, 4156–4165 CrossRef PubMed.
- H. Lei, F. Tang, F. Hao, H. Zhao, W. Liu, Y. Lv, P. Liu, W. Xiong and H. Luo, ACS Sustainable Chem. Eng., 2022, 10, 2947–2959 CrossRef.
- H. Qi, J. Yang, F. Liu, L. Zhang, J. Yang, X. Liu, L. Li, Y. Su, Y. Liu, R. Hao, A. Wang and T. Zhang, Nat. Commun., 2021, 12, 3295 CrossRef CAS PubMed.
- K. Murugesan, T. Senthamarai, V. G. Chandrashekhar, K. Natte, P. C. J. Kamer, M. Beller and R. V. Jagadeesh, Chem. Soc. Rev., 2020, 49, 6273–6328 RSC.
- H. Wei, X. Liu, A. Wang, L. Zhang, B. Qiao, X. Yang, Y. Huang, S. Miao, J. Liu and T. Zhang, Nat. Commun., 2014, 5, 5634 CrossRef CAS PubMed.
- T. Irrgang and R. Kempe, Chem. Rev., 2020, 120, 9583–9674 CrossRef CAS PubMed.
- Q. Sun, N. Wang, T. Zhang, R. Bai, A. Mayoral, P. Zhang, Q. Zhang, O. Terasaki and J. Yu, Angew. Chem., Int. Ed., 2019, 58, 18570–18576 CrossRef CAS.
- X. Sun, A. I. Olivos-Suarez, D. Osadchii, M. J. V. Romero, F. Kapteijn and J. Gascon, J. Catal., 2018, 357, 20–28 CrossRef.
- Z. An, P. Yang, D. Duan, J. Li, T. Wan, Y. Kong, S. Caratzoulas, S. Xiang, J. Liu, L. Huang, A. I. Frenkel, Y. Y. Jiang, R. Long, Z. Li and D. G. Vlachos, Nat. Commun., 2023, 14, 6666 CrossRef CAS PubMed.
- M. Pang, J. Y. Chen, S. Zhang, R. Z. Liao, C. H. Tung and W. Wang, Nat. Commun., 2020, 11, 1249 CrossRef CAS PubMed.
- D. Zhang, X. Gao, T. Cheng and G. Liu, Sci. Rep., 2014, 4, 5091 CrossRef.
- F. Wang, Z. Zhang, Y. Chen, V. Ratovelomanana-Vidal, P. Yu, G. Q. Chen and X. Zhang, Nat. Commun., 2022, 13, 7794 CrossRef CAS PubMed.
- F. Wang, O. Planas and J. Cornella, J. Am. Chem. Soc., 2019, 141, 4235–4240 CrossRef CAS.
- P. Hauwert, R. Boerleider, S. Warsink, J. J. Weigand and C. J. Elsevier, J. Am. Chem. Soc., 2010, 132, 16900–16910 CrossRef CAS PubMed.
- M. Subaramanian, G. Sivakumar, V. G. Landge, R. Kumar, K. Natte, R. V. Jagadeesh and E. Balaraman, J. Catal., 2023, 425, 386–405 CrossRef CAS.
- Y. Wei, Y. Liang, R. Luo and L. Ouyang, Org. Biomol. Chem., 2023, 21, 7484–7497 RSC.
- V. Goyal, T. Bhatt, C. Dewangan, A. Narani, G. Naik, E. Balaraman, K. Natte and R. V. Jagadeesh, J. Org. Chem., 2023, 88, 2245–2259 CrossRef CAS PubMed.
- J. Wang, A. D. Ebner and J. A. Ritter, J. Am. Chem. Soc., 2006, 128, 5949–5954 CrossRef CAS PubMed.
- J. Graetz, J. Wegrzyn and J. J. Reilly, J. Am. Chem. Soc., 2008, 130, 17790–17794 CrossRef CAS.
- W. Zhao, H. Li, H. Zhang, S. Yang and A. Riisager, Green Energy Environ., 2023, 8, 948–971 CrossRef CAS.
- D. Cantillo, M. M. Moghaddam and C. O. Kappe, J. Org. Chem., 2013, 78, 4530–4542 CrossRef CAS PubMed.
- M. G. Kallitsakis, K. D. Nikopoulos and I. N. Lykakis, ChemCatChem, 2025, 17, e202401927 CrossRef CAS.
- V. Dragutan, I. Dragutan, L. Delaude and A. Demonceau, Coord. Chem. Rev., 2007, 251, 765–794 CrossRef CAS.
- S. Enthaler, R. Jackstell, B. Hagemann, K. Junge, G. Erre and M. Beller, J. Organomet. Chem., 2006, 691, 4652–4659 CrossRef CAS.
- H. Liao, P. Weng, H. Huang, R. Tan, R. Zhu, Y. Liu and Z. Wang, RSC Adv., 2023, 13, 20876 RSC.
- V. Goyal, N. Sarki, M. K. Poddar, A. Narani, D. Tripathi, A. Ray and K. Natte, New J. Chem., 2021, 45, 14687–14694 RSC.
- Q. Liu, H. Fu, W. Wang, L. Zhu, Y. Fang, Z. Yang, A. Kroner, H. Ye and B. H. Chen, ACS Appl. Nano Mater., 2024, 7, 22517–22524 CrossRef CAS.
- J. Li, S. Wang, Z. Yu, H. Zhang, L. Zhu, C. Wu and H. Yu, Ind. Eng. Chem. Res., 2025, 64, 10844–10851 CrossRef CAS.
- X. Shi, X. Wang, X. Shang, X. Zou, W. Ding and X. Lu, ChemCatChem, 2017, 9, 3743–3751 CrossRef CAS.
- V. Goyal, N. Sarki, K. Natte and A. Ray, J. Ind. Chem. Soc., 2021, 98, 100014 CrossRef CAS.
- Q. Zhang, J. Bu, J. Wang, C. Sun, D. Zhao, G. Sheng, X. Xie, M. Sun and L. Yu, ACS Catal., 2020, 10, 10350–10363 CrossRef CAS.
- Y. Qu, H. An, X. Zhao and Y. Wang, Chem. Eng. J., 2024, 496, 153949 CrossRef CAS.
- Y. Zhang, L. J. Zhou, F. Wang and X. Zhao, Langmuir, 2022, 38, 7699–7708 CrossRef CAS.
- H. Tian, J. Zhou, Y. Li, Y. Wang, L. Liu, Y. Ai, Z. N. Hu, J. Li, R. Guo, Z. Liu, H. Sun and Q. Liang, ChemCatChem, 2019, 11, 5543–5552 CrossRef CAS.
- J. Yu, X. Zhang, R. Jiang, W. He, M. Xu, X. Xu, Q. Xiang, C. Yin, Z. Xiang, C. Ma, Y. Liu, X. Li and C. Lu, ACS Appl. Mater. Interfaces, 2024, 16, 8603–8615 CrossRef CAS PubMed.
- X. Zheng, B. Li, R. Huang, W. jiang, L. Shen, G. Lei, S. Wang, Y. Zhan and L. Jiang, ACS Catal., 2024, 14, 10245–10259 CrossRef CAS.
- N. Sarki, R. Kumar, B. Singh, A. Ray, G. Naik, K. Natte and A. Narani, ACS Omega, 2022, 7, 19804–19815 CrossRef CAS PubMed.
- X. Li, R. Sun, L. Zhao, J. Wang, Z. Wang, W. Li and X. Wang, Appl. Organomet. Chem., 2025, 39, e7974 CrossRef CAS.
- N. M. Patil, T. Sasaki and B. M. Bhanage, ACS Sustainable Chem. Eng., 2016, 4, 429–436 CrossRef CAS.
- J. P. Southouse, L. Lazzarin, A. O. Ibhadon and M. Grazia Francesconi, New J. Chem., 2021, 45, 17808–17815 RSC.
- K. J. A. Raj, M. G. Prakash, R. Mahalakshmy, T. Elangovan and B. Vishwanathan, Chin. J. Catal., 2012, 33, 1299–1305 CrossRef CAS.
- Y. Qu, H. Yang, S. Wang, T. Chen and G. Wang, Catal. Commun., 2017, 97, 83–87 CrossRef CAS.
- F. Yang, M. Wang, W. Liu, B. Yang, Y. Wang, J. Luo, Y. Tang, L. Hou, Y. Li, Z. Li, B. Zhang, W. Yang and Y. Li, Green Chem., 2019, 21, 704–711 RSC.
- D. Sharma, P. Choudhary, S. Kumar and V. Krishnan, J. Colloid Interface Sci., 2024, 657, 449–462 CrossRef CAS PubMed.
- C. Jiang, Z. Shang and X. Liang, ACS Catal., 2015, 5, 4814–4818 CrossRef CAS.
- K. Martina, M. J. Moran, M. Manzoli, M. V. Trukhan, S. Kuhn, T. V. Gerven and G. Cravotto, Org. Process Res. Dev., 2024, 28, 1515–1528 CrossRef CAS PubMed.
- Y. Zhou, A. Guo, H. Shen, Y. Wang, F. Zhang, X. Rong, Y. Li, X. Yan, L. Chen and B. Wang, ChemCatChem, 2025, 17, e202401902 CrossRef CAS.
- G. Zhang, L. Liu, Q. Zhu and X. Kong, Catal. Lett., 2023, 153, 1536–1542 CrossRef CAS.
- Z. Zhang, Y. Liu, J. Du, Y. Jiang, Z. Wang, R. Yun and B. Zheng, Inorg. Chem. Front., 2023, 10, 7028–7037 RSC.
- S. Dey, S. D. Thakur, A. Sau, D. Panja, T. Roy, J. Zhang, H. V. Annadata and S. Kundu, J. Catal., 2024, 439, 115759 CrossRef CAS.
- S. Dey, D. Panja, A. Sau, S. D. Thakur and S. Kundu, J. Org. Chem., 2023, 88, 10048–10057 CrossRef CAS PubMed.
- H. Jin, P. Li, P. Cui, J. Shi, W. Zhou, X. Yu, W. Song and C. Cao, Nat. Commun., 2022, 13, 1–9 CAS.
- V. Goyal, N. Sarki, B. Saint, A. Ray, M. Poddar, A. Bordoloi, A. Narani and K. Natte, ACS Appl. Nano Mater., 2020, 3, 11070–11079 CrossRef CAS.
- Z. Ma, J. Chen, M. Chen, L. Dong, W. Mao, Y. Long and J. Ma, ChemCatChem, 2024, 16, e202400336 CrossRef CAS.
- M. Yuan, Y. Long, J. Yang, X. Hu, D. Xu, Y. Zhu and Z. Dong, ChemSusChem, 2018, 11, 4156–4165 CrossRef CAS PubMed.
- S. Hanf, L. A. Rupflin, S. Iqbal, R. Gläser and S. A. Schunk, catalysts, 2020, 10, 510 CrossRef CAS.
- L. Karam, C. Fares, C. Weidenthaler and N. Neumann, Angew. Chem., Int. Ed., 2024, 63, e202404292 CrossRef CAS PubMed.
- Z. Zhao and Z. Y. Yuan, Smart Mater. Devices, 2025, 1, 202521 Search PubMed.
- A. Neyyathala, F. Flecken, F. Rang, C. Papke and S. Hanf, Chem. – Eur. J., 2024, 30, e202302825 CrossRef CAS PubMed.
- A. Neyyathala, F. Flecken and S. Hanf, ChemPlusChem, 2023, 88, e202200431 CrossRef CAS PubMed.
- H. Ishikawa, S. Yamaguchi, A. Nakata, K. Nakajima, S. Yamazoe, J. Yamasaki, T. Mizugaki and T. Mitsudome, JACS Au, 2022, 2, 419–427 CrossRef CAS PubMed.
- J. N. D. Leon, C. R. Kumar, J. A. Garcia and S. F. Moyado, Catalysts, 2019, 9, 87 CrossRef.
- L. Gong, W. Zhang, Y. Zhuang, K. Zhang, Q. Zhao, D. Xiao, S. Liu, Z. Liu and Y. Zhang, ACS Appl. Mater. Interfaces, 2024, 16, 66211–66218 CrossRef CAS PubMed.
- A. Neyyathala, E. Faco, S. De, D. Gashnikova, F. Maurer, J. D. Grunwaldt, S. A. Schunk and S. Hanf, Small Struct., 2025, 6, 2400260 CrossRef CAS.
- X. Lin, M. Zhang., X. Liu, J. Liu, H. Dai, W. Luo, J. Liu, R. Cao and Q. Yang, ACS Catal, 2024, 14, 16214–16223 CrossRef.
- I. Sorribes, L. Liu and A. Corma, ACS Catal., 2017, 7, 2698–2708 CrossRef CAS.
- L. Lückemeier, T. D. Vos, L. Schlichter, C. Gutheil, C. G. Daniliuc and F. Glorius, J. Am. Chem. Soc., 2024, 146(9), 5864–5871 CrossRef.
- Q. Zhang, W. Xu, X. Li, D. Jiang, Y. Xiang, J. Wang, J. Cen, S. Romano and J. Ni, Appl. Catal. A, 2015, 497, 17–21 CrossRef CAS.
- Z. Y. Wu, H. Nan, S. C. Shen, M. X. Chen, H. W. Liang, C. Q. Huang, T. Yao, S. Q. Chu, W. X. Li and S. H. Yu, CCS Chem., 2022, 4, 3051–3063 CrossRef CAS.
- A. Neyyathala, E. Fako, S. De, D. Gashnikova, F. Maurer, J. D. Grunwaldt, S. A. Schunk and S. Hanf, Small Struct., 2025, 6, 2400260 CrossRef CAS.
- M. Kumar, D. I. Jeong, N. Sarwar and D. H. Yoon, Ceram. Int., 2021, 47, 16852–16860 CrossRef CAS.
- Z. Liu, Y. Sun, X. Xu, J. Qu and B. Qu, ACS Omega, 2020, 5, 29231–29242 CrossRef CAS PubMed.
- S. Sharma, M. Kaur, C. Sharma, A. Choudhary and S. Paul, ACS Omega, 2021, 6, 19529–19545 CrossRef CAS PubMed.
- J. Tang, S. Ni, D. Chao, J. Liu, X. Yang and J. Zhao, Electrochim. Acta, 2018, 265, 709–716 CrossRef CAS.
- J. Zhang, T. Wang, P. Liu, Z. Liao, S. Liu, X. Zhuang, M. Chen, E. Zschech and X. Feng, Nat. Commun., 2017, 8, 15437 CrossRef CAS PubMed.
- X. Liu, Y. Li, N. Chen, D. Deng, X. Xing and Y. Wang, Electrochim. Acta, 2016, 213, 730–739 CrossRef CAS.
- J. Li, S. Ding, F. Wang, H. Zhao, J. Kou, M. Akram, M. Xu, W. Gao, C. Liu, H. Yang and Z. Dong, J. Colloid Interface Sci., 2022, 625, 640–650 CrossRef CAS.
- X. Liu, Y. Ren, M. Wang, X. Ren, J. Liu and Q. Yang, ACS Catal., 2022, 12, 11369–11379 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |