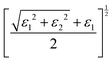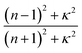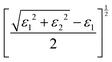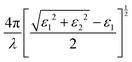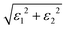Open Access Article
Open Access ArticleStrain-tunable mechanical, electronic and optical properties of La2AlGaO6 hybrid perovskites: a first-principles investigation
Chaithanya Purushottam
Bhat
 ,
Jyoti
Dagar
,
Ashwin K.
Godbole
and
Debashis
Bandyopadhyay
,
Jyoti
Dagar
,
Ashwin K.
Godbole
and
Debashis
Bandyopadhyay
 *
*
Department of Physics, Birla Institute of Technology and Science Pilani, Rajasthan – 333031, India. E-mail: bandy@pilani.bits-pilani.ac.in; Debashis.bandy@gmail.com
First published on 24th March 2026
Abstract
Perovskite materials, well-known for their structural flexibility and multifunctional characteristics, continue to attract attention for advanced electronic and optoelectronic applications. In this work, the elastic and strain-engineered electronic, mechanical, and optical properties of orthorhombic La2AlGaO6 (LAGO) – a hybrid perovskite formed by combining LaAlO3 and LaGaO3 – are systematically investigated using first-principles density functional theory (DFT) calculations. Structural optimizations were performed using the generalized gradient approximation (GGA). Mechanical stability was verified through the Born–Huang criteria, and the computed elastic constants (C11, C12, C33, C44, and C66) were employed to derive key mechanical parameters, including Young's modulus, bulk modulus, shear modulus, Poisson's ratio, Cauchy's pressure, and anisotropy factor, providing insights into the material's ductility, hardness, and elastic anisotropy. Furthermore, the influence of biaxial strain on the electronic band structure, total and partial density of states (DOS/PDOS), and Fermi energy was analyzed, revealing pronounced band gap modulation under both compressive and tensile strains, accompanied by significant changes in optical behavior. The strong coupling between elastic response and electronic structure underscores LAGO's potential for tunable device applications, where mechanical stimuli can effectively tailor its electronic and optical functionalities.
1. Introduction
The investigation of perovskite materials has gathered significant interest in recent years, mainly due to their exceptional structural versatility and promising functionalities across numerous applications.1–6 Due to their easily tunable electronic and optical properties, perovskites have found extensive utility in optoelectronics, catalysis, energy storage, and conversion, and in many other systems. Features, such as customizable band structures, high charge-carrier mobility, superior defect tolerance, and strong light-harvesting capabilities, have propelled their use in advanced technologies like solar cells, batteries, and energy storage.7–12 Furthermore, doping techniques applied to perovskite materials have appeared as transformative tools, enabling fine-tuning of the luminescence, enhancing the structural stability, and optimizing device performance metrics such as external quantum efficiency (EQE) in light-emitting diodes.13–15 These modifications highlight the critical role of dopants in improving the efficiency and durability of perovskite-based devices.16–20 ABO3-type perovskites and their hybrid counterparts, such as A2BB′O6 structures, exhibit unique crystal symmetries and electronic configurations that make them particularly appealing for both central research and practical applications.21–24 Recent studies have established the efficacy of anion-doped perovskite oxides as catalysts in various chemical reactions, where alterations to the lattice structure, metal–oxygen bond strength, and defect configurations significantly influence catalytic activity.25,26 Furthermore, investigations into the electronic properties of these materials have emphasized the importance of orbital hybridization between transition metal 3d and oxygen 2p states, which governs their electronic behavior and stability.27 Recent research on their different elastic coefficients, lattice vibration, and thermodynamic properties has deepened our understanding of their mechanical robustness and structural adaptability.28 The ability to tune the electronic properties of perovskite materials using external factors, such as strain, presents an exciting opportunity for the development of advanced electronic devices. To study such important materials, density functional theory (DFT) has emerged as one of the most effective computational methods for understanding the electronic, mechanical, and optical properties of these materials.29–35Study on the mechanical, electronic, and optical properties of LAGO perovskite, with an emphasis on their implications for diverse applications. Analyzing these properties is vital for appraising the material's toughness, damage resistance, and suitability for industrial purposes.36 Computational techniques, including finite element analysis (FEA), serve as powerful tools to simulate material behavior under various conditions, providing predictive insights into mechanical performance through parameters such as Young's modulus.37,38
Elastic constants are essential for deriving key material attributes like sound velocity, Debye temperature, and melting point, all of which are integral to both scientific research and technological innovations. Additionally, changes in elastic properties during phase transitions offer valuable information on complex phenomena in systems such as heavy fermion materials and superconductors.39,40 The full anisotropic elastic tensor enables precise predictions of a material's mechanical response, thereby supporting the development of tailored composite materials with desired stiffness and elasticity profiles.41,42 In geophysics, elastic constants are instrumental in interpreting seismic data by correlating them with acoustic velocity measurements.43,44
Hooke's law, which defines the proportionality between stress and strain, forms the theoretical foundation for understanding elastic behavior in materials. The elastic constants (Cij) relate stress (σ) and strain (εi) as (σiCij).45,46 The number of independent elastic constants depends on the material's symmetry. For tetragonal symmetry, as seen in LAGO perovskite, the constants include C11, C12, C33, C44, and C66, while cubic symmetry involves C11, C12, and C44. These constants provide critical insights into the mechanical stability, ductility, anisotropy, and elasticity of the material, thereby informing its practical applicability across various fields. In this study, we also employ DFT to investigate the effects of strain on the mechanical, electronic, and optical properties of La2AlGaO6 (LAGO) perovskite, providing valuable insights into its potential for strain-engineered electronic devices.
2. Computational
All electronic structure calculations were carried out using spin-polarized density functional theory (DFT) as implemented in the Vienna ab initio Simulation Package (VASP).47–50 The interactions between the ion cores and valence electrons were described using the projector augmented wave (PAW) method.51 All calculations employed the Perdew–Burke–Ernzerhof (PBE) exchange–correlation functional52 and included van der Waals interactions through Grimme's D2 dispersion correction.53 Convergence criteria were set at 10−6 eV for energy and 0.005 eV Å−1 for forces on each atom. The plane-wave basis set had an energy cut-off of 520 eV, which is 130% that of the ENMAX of the oxygen atom. We used the LAGO supercell La8Al4Ga4O24 with an 8 × 8 × 8 k-point mesh for relaxation. For Brillouin-zone sampling during structural optimization, we performed a systematic k-point convergence test starting from coarse grids (2 × 2 × 2) and increasing up to denser ones (8 × 8 × 8). At each step, total energy and forces were monitored to identify a mesh where the variations fall below a few 10−3 eV per atom – indicating convergence. This procedure ensured that the electronic integration is sufficiently accurate for geometry relaxation. Additionally, in a similar double-perovskite DFT study, an 8 × 8 × 8 Monkhorst–Pack mesh is typical for unit cells, with smaller meshes used for supercells after convergence checks. For density of states and band structure calculations, we employed the Γ-centered k-path. The calculated band structures of La2AlGaO6 perovskite by using the PBE functional are plotted with high symmetry points following the K-path as given, Γ → M → Z → Γ → Z → R → A → Z|X → R|M → A in the first Brillouin zone.3. Results and discussion
3.1 Elastic properties of LAGO perovskite
In this study, our primary objective is to investigate the mechanical properties of pure LAGO perovskite by calculating various elastic constants. These constants are essential for characterizing the material's mechanical behavior under different conditions. In the present framework for our work, it is significant to acknowledge the work of Naher et al.,54 who conducted an extensive study on the mechanical properties of Mo5PB2. Their research provides valuable insights into the methodologies used for analyzing elastic properties in complex materials, which serve as a foundation for our investigation into LAGO perovskite. Their detailed work on Mo5PB2 has helped inform our approach to understanding the mechanical characteristics of perovskites and the significance of elastic constants in determining material behavior.While examining the mechanical properties of materials with tetragonal structures, the mechanical stability can be measured using the Born stability criteria, referred to as the Born and Huang criteria.55 These principles provide a set of necessary conditions that must be met for a material to be mechanically stable under minor perturbations. The stability conditions are expressed through specific constraints, which are precisely represented in eqn (1). These constraints involve the material's elastic constants and ensure that the material will maintain structural integrity when subjected to external stresses, helping to predict whether the material will undergo any unstable deformations or transitions.
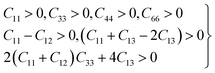 | (1) |
| C11 = 230.133, C12 = 94.528, C13 = 94.659, |
| C33 = 226.821, C44 = 78.649, C66 = 101.128 |
The elastic constants C11 and C33 characterize the resistance to linear compressions along the [100] and [001] directions, respectively, in LAGO. A higher value of C11 compared to C33 indicates a stronger bonding strength/compressibility along the [100] direction relative to the [001] direction in LAGO. Regarding shear deformation resistance, represented by the elastic constant C44, it is notably lower than both C11 and C33 in LAGO. This suggests that LAGO is more susceptible to deformation via shear stress compared to unidirectional stress along its principal crystallographic directions. Observing that C11 exceeds C33 indicates stronger bonding strength/compressibility along the [100] direction compared to the [001] direction in LAGO. The elastic constant C44, which signifies resistance to shear deformation, is notably smaller in LAGO compared to C11 and C33. This indicates that LAGO exhibits a greater tendency to deform under shear stress than under uniaxial stress along its principal crystallographic axes. Additionally, the elastic constant C66 represents the shear resistance of the (100) plane in the [110] direction. In LAGO, the value of (C11 + C12) exceeding C33 suggests that the bonding within the (001) plane is mechanically more rigid than along the c-axis. Consequently, the elastic tensile modulus is higher in the (001) plane than along the c-axis.
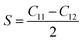 | (2) |
The shear constant S, also known as the tetragonal shear modulus, measures the stiffness of a material and its ability to resist shear deformation under stress in the (110) plane along the [110] direction. The sign of S is critical for assessing stability: a positive value indicates dynamical stability, whereas a negative value points to instability. For LAGO, the computed value of S is 67.8025 GPa, suggesting that the material is dynamically stable.
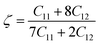 | (3) |
The Kleinman parameter (ξ) spans from zero to one (1 ≤ ξ ≤ 1) for a solid and ξ values of 0 and 1 denote the lower and upper boundaries, respectively. A ξ value of 0 signifies minimal bond bending contribution to endure external stress, whereas ξ equal to 1 indicates minimal bond stretching/contracting contribution to resist externally applied stress. The computed ξ for LAGO is 0.55, indicating that the mechanical strength of LAGO is influenced by both bond bending and bond stretching/contracting contributions.
The general expressions for Voigt bulk modulus (BV) and shear modulus (GV), and Reuss bulk modulus (BR) and shear modulus (GR) are as follows:
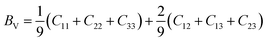 | (4) |
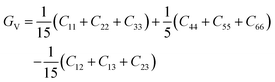 | (5) |
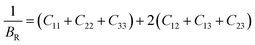 | (6) |
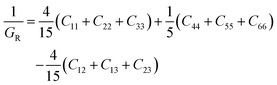 | (7) |
Hill illustrated that the Voigt and Reuss equations act as upper and lower limits for the actual polycrystalline constants. He proposed that a pragmatic estimation of the bulk and shear moduli could be attained by averaging these extremes. Hence, the elastic moduli can be approximated using Hill's average as follows:
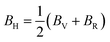 | (8) |
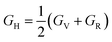 | (9) |
The calculated values of these parameters are shown in Table 1.
| Mechanical properties | Voigt | Reuss | Hill |
|---|---|---|---|
| Bulk modulus: B (GPa) | 139.42 | 139.414 | 139.417 |
| Young's modulus: E (GPa) | 198.43 | 194.718 | 196.577 |
| Shear modulus: G (GPa) | 78.57 | 76.829 | 77.698 |
By using these parameters, other elastic parameters can be defined, measured, and discussed in the following sections.
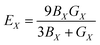 | (10) |
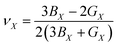 | (11) |
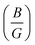 .
Once more, Pugh introduced a simple correlation to predict whether materials exhibit brittle or ductile characteristics, proposing that the ratio of bulk to shear modulus
.
Once more, Pugh introduced a simple correlation to predict whether materials exhibit brittle or ductile characteristics, proposing that the ratio of bulk to shear modulus 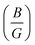 serves as an indicator. Pugh established a critical threshold for the transition from ductile to brittle behavior: if
serves as an indicator. Pugh established a critical threshold for the transition from ductile to brittle behavior: if 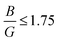 , the material behaves brittlely; otherwise, it behaves ductilely. For LAGO, with a
, the material behaves brittlely; otherwise, it behaves ductilely. For LAGO, with a 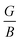 value of 1.77, slightly exceeding the specified condition, we anticipate the compound to demonstrate brittle behavior.
value of 1.77, slightly exceeding the specified condition, we anticipate the compound to demonstrate brittle behavior.
| C″ = C12 − C44 | (12) |
A positive Cauchy pressure usually indicates ductility, whereas a negative value suggests brittleness. In the instance of LAGO, the positive Cauchy pressure strongly indicates the compound's brittleness. Furthermore, Cauchy pressure is employed to describe the atomic bonding along the angular direction in a material. If this value is positive, it indicates ionic bonding, and for a negative value, it is covalent bonding in nature. In LAGO, with a Cauchy pressure value of 15.9 GPa, it exhibits bonding similar to ionic bonding.
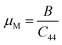 | (13) |
The machinability index also provides insights into a material's plasticity and dry lubrication behavior. The equation suggests that a combination of high overall strength and low shear resistance enhances machinability and dry lubrication performance. Materials with a high μM value exhibit superior dry lubrication, reduced feed forces, lower friction coefficients, and improved plastic deformation potential. With a μM value of 1.77, LAGO is predicted to exhibit excellent machinability, comparable to that of technologically significant MAX phase compounds, alloys, and other layered ternaries.
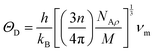 | (14) |
The average velocity of the sound wave νm, can be determined as:
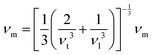 | (15) |
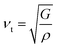 | (16) |
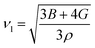 | (17) |
The calculated values of these parameters of LAGO are shown in Table 2.
| C″ (GPa) | μ M | Θ D (K) | ν m (m s−1) | ν t (m s−1) | ν l (m s−1) |
|---|---|---|---|---|---|
| 15.9 | 1.77 | 502.4 | 3907.037 | 3513.001 | 6212.822 |
 | (18) |
 | (19) |
To derive a singular anisotropy measure, Chung and Buessem58 introduced the following empirical approach:
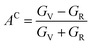 | (20) |
The universal anisotropy factor is limited to values of zero or greater. A value of zero for AU denotes isotropy within the crystal, whereas any non-zero value indicates the existence of anisotropy. Usually, in a low-range isotropic crystal, AU equals AC, and both are zero. In the case of LAGO, where AC is 0.0 and AU is 0.11, this suggests that the material exhibits minimal anisotropy.
3.2 Effect of strain on electronic properties
It is important to note that among these three different structures, we have reported only the strain dependent study on a cubic system as a choice. Details of the optimized structure parameters and corresponding energies are reported in the SI.
These changes are strongly dependent on both the magnitude and sign of the applied strain, highlighting the asymmetric response of the system to compressive and tensile deformation. The strain-dependent band structure of LAGO perovskite is shown in Fig. 2a and b.
For clarity and comprehensive understanding, the discussion of the strain-dependent electronic structure is organized into four subsections. Each subsection corresponds to specific strain regimes and includes a comparative analysis between equivalent compressive and tensile strains (e.g., ±4% and ±6%). Particular emphasis is placed on the evolution of orbital hybridization, shifts in the valence and conduction band edges, symmetry-driven transitions, and the structural origins of observed discontinuities in electronic parameters. This detailed breakdown allows for an in-depth interpretation of the strain-induced modulation of the electronic landscape in La2AlGaO6, paving the way for its potential application in strain-tunable electronic and optoelectronic devices.
3.2.2.1 Electronic evolution under mild strain (0 to −4% and 0 to +4%). In the strain interval between −4% and +4%, the electronic structure of La2AlGaO6 exhibits quasi-linear and reversible behavior. The calculated band structures remain semiconducting, with no apparent closure of the band gap or abrupt changes in orbital character. The band gap varies moderately, from approximately 3.10 eV at −4% compressive strain to 2.90 eV at +4% tensile strain. This slight asymmetry in band gap evolution reflects the intrinsic non-centrosymmetric bonding environment in the perovskite lattice. Under mild compressive strain (−4%), the decrease in lattice parameter along the z-axis results in shortened La–O, Al–O, and Ga–O bond lengths. This compression enhances the crystal field splitting and the orbital hybridization between O 2p states and the unoccupied cationic orbitals (primarily La 5d, Ga 4s, and Al 3s). The increased orbital overlap leads to a greater energy separation between the bonding and antibonding states, resulting in an upward shift of the conduction band minimum (CBM) and a corresponding increase in band gap.
Conversely, under an equivalent tensile strain (+4%), bond elongation leads to a decrease in p–d and p–s hybridization, weakening the electronic coupling between the anions and cations. This causes a reduction in crystal field strength and narrows the band gap due to the downward movement of the CBM. However, the VBM position remains relatively stable, as it is dominated by strongly localized O 2p states. Overall, this strain range represents a nearly elastic regime where the system maintains its structural symmetry, and the changes in electronic properties are driven primarily by bond length modulation.
3.2.2.2 Asymmetric behavior under moderate strain (−4% to −6% and 4% to +6%). The application of moderate strain magnitudes (|ε| = 6%) leads to a nonlinear and asymmetric evolution of the electronic structure, reflecting the onset of anisotropic lattice distortion and partial breakdown of orbital degeneracy. For 6% comprehensive strain, the band gap reaches its maximum value (∼3.35 eV), accompanied by the highest Fermi energy observed in the compressive regime (∼7.5 eV). The band structure becomes more dispersive, especially around the CBM, indicating an enhancement in carrier mobility. This behavior is consistent with a further strengthening of orbital hybridization under compression, particularly involving the antibonding interactions between O 2p and Ga/Al s–d states. The enhanced orbital repulsion pushes the CBM higher, increasing the band gap. In contrast, at +6% tensile strain, the band gap drops to ∼2.75 eV, and the Fermi level shifts downward to ∼6.1 eV. This reduction is associated with a reduction in orbital overlap due to increased interatomic distances, leading to a lower CBM energy and flatter conduction band, which indicates reduced carrier delocalization and higher effective masses.
A direct comparison of +6% and −6% strain reveals a clear electronic asymmetry, with compressive strain inducing a more significant band gap widening than the narrowing induced by the same magnitude of tensile strain. This asymmetric response can be attributed to the non-linear coupling between strain and internal octahedral distortions, which affects orbital orientations and interactions differently in tension and compression.
Under compressive strain, the contraction of the octahedra enhances the orbital overlap, increasing the crystal field strength and promoting stronger bonding–antibonding splitting. This leads to the lifting of the CBM and deepening of the VBM, enhancing the electronic band gap and stabilizing the semiconducting state. The bands near the Fermi level are more dispersive in this regime, suggesting increased orbital delocalization and higher carrier mobility. In contrast, tensile strain reduces the hybridization strength as the metal–oxygen bond lengths elongate, leading to a lower CBM and a mild upward shift in the VBM. The frontier orbitals become less hybridized, resulting in more localized electronic states. The flattening of the bands near the Fermi level – most prominent beyond +6% – is indicative of suppressed electronic coupling and may signal a transition toward semimetallic or low-mobility electronic behavior.
This evolution in orbital interaction strength is reflected in the trends of the conduction and valence band edge positions (Fig. 3, bottom panel), where the CBM drops significantly more under tensile strain than the VBM rises, accounting for the net band gap reduction.
As the strain transitions from compressive to tensile, especially from 0% to +8%, the hybridization behavior shifts significantly. Tensile strain elongates the lattice along the c-axis, reducing orbital overlap and thus weakening the hybridization between the O-2p and Al/Ga p-orbitals. Consequently, the conduction band edge moves to higher energy, and the valence band becomes more localized, resulting in a wider band gap. The PDOS under tensile strain shows reduced intensity and sharper peaks, indicating more localized electronic states due to diminished orbital mixing. In particular, the weakening of the Al–O and Ga–O bonds under tension suppresses the p–p and p–s interactions responsible for the original band structure. The gradual decoupling of these orbitals leads to a more ionic character and reduced carrier mobility. Most significantly, across the entire strain range, the La-f states (omitted in the right column of the PDOS for clarity) are not significantly involved in the states near the band edges and thus do not influence the electronic transitions directly. The electronic behavior is predominantly governed by the hybridization between the oxygen 2p orbitals and the p/s orbitals of Al and Ga. The ability to modulate this hybridization through strain engineering highlights the material's potential for electronic and optoelectronic device applications, such as strain-sensitive transistors or band gap-engineered photovoltaics.
Moreover, the transition from a highly hybridized to a weakly hybridized regime may cause certain electronic states to shift rapidly in energy, manifesting as band reordering or band crossing phenomena. These structural effects are more pronounced under tensile strain due to the softening of the lattice and reduction in octahedral rigidity. Hence, the discontinuous trends in electronic parameters serve as signatures of incipient phase transitions or electronic reconstructions driven by strain-induced lattice instabilities, underscoring the need for careful strain control in practical applications.
This comprehensive analysis reveals that La2AlGaO6 is an electronically versatile material, where strain acts as a tuning knob for band gap, band edge positions, Fermi level, and hybridization. It is important to mention here that the present method of electronic structure calculation underestimates the band gap. Therefore, we have tested the band structure calculation of zero strain LAGO perovskite using HSE06. We found that using the HSE06 hybrid functional, the band gap increased by an amount 0.74 eV. To avoid the computational cost, in the present report, we have used the computational method as described in the methodology. The asymmetric and nonlinear response to tensile and compressive deformation, coupled with hybridization-driven band structure evolution, offers a rich platform for strain-engineered optoelectronic applications, such as tunable insulators, photodetectors, or piezoelectric devices. These parameters are presented in Table 3.
| Strain | c (Å) | Gap (eV) | E f (eV) | E − E0 (eV) | BE (eV) |
|---|---|---|---|---|---|
| −8 | 8.155 | 2.688 | 7.277 | −0.434 | 1.690 |
| −6 | 8.235 | 2.842 | 7.026 | 0.395 | 0.860 |
| −4 | 8.393 | 2.994 | 6.766 | 0.689 | 0.566 |
| −3 | 8.551 | 3.065 | 6.636 | 0.542 | 0.713 |
| −2 | 8.710 | 3.135 | 6.516 | 0.527 | 0.728 |
| −1 | 8.868 | 3.256 | 6.396 | 0.315 | 0.940 |
| 0 | 8.353 | 3.267 | 6.276 | 0 | 1.255 |
| 1 | 7.839 | 3.341 | 6.166 | −0.240 | 1.496 |
| 2 | 7.760 | 3.052 | 6.156 | −0.816 | 2.072 |
| 3 | 7.681 | 3.162 | 5.944 | −0.966 | 2.222 |
| 4 | 7.601 | 2.803 | 6.086 | −1.876 | 3.132 |
| 6 | 7.443 | 2.561 | 6.016 | −3.125 | 4.380 |
| 8 | 7.284 | 2.242 | 5.956 | −4.516 | 5.772 |
These strain-dependent changes in electronic structure illustrate the versatility of La2AlGaO6 as a strain-engineerable perovskite. By tuning the biaxial strain, through epitaxial growth on mismatched substrates or external mechanical stress, researchers can systematically manipulate the positions of the HOMO and LUMO to tailor the band alignment and carrier dynamics. This level of control provides a powerful strategy for optimizing device performance across a wide range of applications, from high-efficiency solar cells to UV detectors and dielectric materials. The above variation can be understood with the help of the DOS/PDOS variation of LAGO with the applied strain, as shown in Fig. 4. The contribution of La-f is not shown in the figure because of its dominating contributions. With the application of strain, the HOMO–LUMO gaps have been changed because of the variation of the HOMO, which contains mainly Ga-d contributions, as shown in the DOS plot.
3.3 Strain-dependent optical properties of LAGO (uniaxial strain along z, −8% → +8%)
To investigate the influence of mechanical deformation on the optical response of the La2AlGaO6 (LAGO) perovskite, we analyzed its optical properties under uniaxial strain ranging from −8% (compressive) to +8% (tensile) along the crystallographic c-axis. The variation of different optical parameters is shown in Fig. 4. The optical parameters were computed using the frequency-dependent complex dielectric function, from which the real and imaginary parts (ε1(ω) and ε2(ω)), as well as derived properties, such as the refractive index, absorption coefficient, reflectivity, optical conductivity, and energy loss function, were evaluated. The relation between different optical parameters and the real and imaginary parts of the dielectric constants is shown in Table 4.Therefore, uniaxial strain along the c-axis from −8% to +8% tunes LAGO's optical response in a systematic way: compressive strain raises the transition energies, enhances UV absorption and reflectivity, and hardens plasmon resonances, while tensile strain lowers transition energies, weakens absorption and reflectivity, and softens the plasmonic response, making strain engineering a practical route to tailor LAGO for either active UV optoelectronic/photocatalytic roles or passive transparent/antireflective applications.
Conclusions
In the present report, we conducted an inclusive first-principles investigation into the mechanical, electronic, and optical properties of La2AlGaO6 (LAGO) perovskite under applied uniaxial external strain. The calculated elastic constants satisfy the Born stability criteria, confirming the mechanical stability of LAGO. Analysis of the elastic constants revealed a greater resistance to linear compression along the c-axis, while a lower shear resistance was observed, indicating a susceptibility to deformation under shear stress. The positive tetragonal shear modulus and Kleinman parameter value of 0.55 highlight the dynamic stability of LAGO and validate that both bond bending and stretching contribute significantly to its mechanical strength. Bulk and shear moduli, evaluated via Voigt, Reuss, and Hill approximations, further support the material's mechanical robustness. The calculated Young's modulus and Poisson's ratio (0.26) suggest that LAGO is a stiff, brittle material, with significant ionic character in its chemical bonding. Pugh's ratio and Cauchy pressure analyses align with these findings, indicating a predominantly brittle nature and ionic bonding. Moreover, the machinability index of 1.77 points to excellent plasticity and dry lubrication potential, comparable to that of technologically important layered materials. Finally, the near-zero universal anisotropy factor confirms that LAGO is almost elastically isotropic. In addition, the strain effect on the electronic structure of LAGO perovskite has been comprehensively analyzed, revealing significant changes in the band gap and its nature. Based on the detailed analysis of strain-dependent electronic and optical properties of La2AlGaO6 perovskite, it is evident that uniaxial strain along the c-axis significantly modulates its band structure and optical response. Compressive strain enhances orbital hybridization between O-2p and Al/Ga p-states, leading to band gap narrowing and increased electronic delocalization, while tensile strain weakens this hybridization, resulting in band gap widening and more localized states. These variations are directly reflected in the optical spectra, where shifts in the absorption edges, dielectric functions, and refractive indices correlate with the changes in electronic structure. Such tunability through strain engineering demonstrates the potential of La2AlGaO6 for advanced optoelectronic and strain-sensitive applications, where precise control over band gap and light–matter interactions is essential. The results highlight the sensitivity of the electronic structure to strain, which can be exploited to engineer materials with tailored properties for optoelectronic applications. These findings offer valuable insights into the potential of strain-engineering as a strategy for enhancing the performance of LAGO-based devices across various technological domains, including solar cells, light-emitting diodes, and piezoelectric devices. These results provide critical insights into the mechanical performance of La2AlGaO6, suggesting it as a promising candidate for applications requiring mechanical stability, thermal resilience, and good machinability in high-performance environments.Author contributions
Miss Chaithanya Purushottam Bhat (first author) was primarily responsible for data analysis, organizing the research paper, conducting the literature survey, setting references, and reviewing and revising the manuscript draft. Miss. Joyti Dagar (second author) was responsible for the data correction, graphical draft checking, and optical property calculations. Mr Ashwin K. Godbole (third author) was responsible for data analysis, writing the script, and checking the draft. Prof. Debashis Bandyopadhyay acted as the corresponding author and took on a broader set of duties, including project coordination, conceptualization, VASP simulations, manuscript writing, and review, as well as project administration. These CRediT statements offer a comprehensive and transparent overview of each author's contributions, ensuring proper acknowledgment of their roles in the project.Conflicts of interest
The authors confirm that they have no financial interests or personal relationships that could be perceived as conflicting with the research presented in this paper.Data availability
Supplementary information (SI) contains the coordinates of the structures presented in Fig. 1 and details of k-points, cut-off energies, etc. See DOI: https://doi.org/10.1039/d6ma00060f.To ensure transparency and research reproducibility and enable reuse of our findings, all data, software, and code that support the results of this study are presented in the form of table and graphs.
Acknowledgements
One of the authors, Debashis Bandyopadhyay, is thankful to the Science and Engineering Research Board (SERB), Government of India, for the research funding provided under sanction order CRG/2022/003249, which enabled this work. To improve the quality of the Graphical Abstract, language and ensure proper citation, the author(s) used Grammarly, Copilot, Turnitin and other free AI software while drafting this manuscript. The AI tools no way influenced the results/data of the work. After utilizing these tools, the author(s) reviewed and made any necessary revisions, and they assume complete responsibility for the publication's content.References
- B. Han, B. Cai, Q. Shan, J. Song, J. Li, F. Zhang, J. Chen, T. Fang, Q. Ji, X. Xu and H. Zeng, Stable, efficient red perovskite light-emitting diodes by (a, d)-CsPbI3 phase engineering, Adv. Funct. Mater., 2018, 28, 1804285, DOI:10.1002/adfm.201804285.
- J. Song, J. Li, X. Li, L. Xu, Y. Dong and H. Zeng, Quantum dot light-Emitting diodes based on inorganic perovskite cesium lead halides (CsPbX3), Adv. Mater., 2015, 27, 7162–7167, DOI:10.1002/adma.201502567.
- K. Lin, J. Xing, L. N. Quan, F. P. G. de Arquer, X. Gong, J. Lu, L. Xie, W. Zhao, D. Zhang, C. Yan, W. Li, X. Liu, Y. Lu, J. Kirman, E. H. Sargent, Q. Xiong and Z. Wei, Perovskite light-emitting diodes with external quantum efficiency exceeding 20 percent, Nature, 2018, 562, 245–248, DOI:10.1038/s41586-018-0575-3.
- Z. Shi, Y. Li, Y. Zhang, Y. Chen, X. Li, D. Wu, T. Xu, C. Shan and G. Du, High-efficiency and air-stable perovskite quantum dots light-emitting diodes with an all-inorganic heterostructure, Nano Lett., 2017, 17, 313–321, DOI:10.1021/acs.nanolett.6b04116.
- J. Song, J. Li, L. Xu, J. Li, F. Zhang, B. Han, Q. Shan and H. Zeng, Room-temperature triple-ligand surface engineering synergistically boosts ink stability, recombination dynamics, and charge injection toward EQE-11.6% perovskite QLEDs, Adv. Mater., 2018, 30, 1800764, DOI:10.1002/adma.201800764.
- Y. Dong, Y. Gu, Y. Zou, J. Song, L. Xu, J. Li, J. Xue, X. Li and H. Zeng, Improving All-inorganic perovskite photodetectors by preferred orientation and plasmonic effect, Small, 2016, 12, 5622–5632, DOI:10.1002/smll.201602366.
- J. Song, L. Xu, J. Li, J. Xue, Y. Dong, X. Li and H. Zeng, Monolayer and few-layer all-inorganic perovskites as a new family of two-dimensional semiconductors for printable optoelectronic devices, Adv. Mater., 2016, 28, 4861–4869, DOI:10.1002/adma.201600225.
- B. A. Koscher, J. K. Swabeck, N. D. Bronstein and A. P. Alivisatos, Essentially trap free CsPbBr3 colloidal nanocrystals by post synthetic thiocyanate surface treatment, J. Am. Chem. Soc., 2017, 139, 6566–6569, DOI:10.1021/jacs.7b02817.
- J. Xue, Z. Zhu, X. Xu, Y. Gu, S. Wang, L. Xu, Y. Zou, J. Song, H. Zeng and Q. Chen, Narrowband perovskite photodetector-based image array for potential application in artificial vision, Nano Lett., 2018, 18, 7628–7634, DOI:10.1021/acs.nanolett.8b03209.
- A. Abate, M. Saliba, D. J. Hollman, S. D. Stranks, K. Wojciechowski, R. Avolio, G. Grancini, A. Petrozza and H. J. Snaith, Supramolecular halogen bond passivation of organic-inorganic halide perovskite solar cells, Nano Lett., 2014, 14, 3247–3254, DOI:10.1021/nl500627x.
- Q. Liao, K. Hu, H. Zhang, X. Wang, J. Yao and H. Fu, Perovskite micro disk micro lasers self-assembled from solution, Adv. Mater., 2015, 27, 3405–3410, DOI:10.1002/adma.201500449.
- Y. Gao, C. Huang, C. Hao, S. Sun, L. Zhang, C. Zhang, Z. Duan, K. Wang, Z. Jin, N. Zhang, A. V. Kildishev, C. W. Qiu, Q. Song and S. Xiao, Lead halide perovskite nanostructures for dynamic color display, ACS Nano, 2018, 12, 8847–8854, DOI:10.1021/acsnano.8b02425.
- J. Song, T. Fang, J. Li, L. Xu, F. Zhang, B. Han, Q. Shan and H. Zeng, Organic-inorganic hybrid passivation enables perovskite QLEDs with an EQE of 16.48%, Adv. Mater., 2018, 30, 1805409, DOI:10.1002/adma.201805409.
- T. Chiba, Y. Hayashi, H. Ebe, K. Hoshi, J. Sato, S. Sato, Y. J. Pu, S. Ohisa and J. Kido, Anion-exchange red perovskite quantum dots with ammonium iodine salts for highly efficient light-emitting devices, Nat. Photonics, 2018, 12, 681, DOI:10.1038/s41566-018-0260-y.
- S. Hou, M. K. Gangi Shetty, Q. Quan and D. N. Congreve, Efficient blue and white perovskite light-emitting diodes via manganese doping, Joule, 2018, 2, 2421–2433, DOI:10.1016/j.joule.2018.08.005.
- J. Song, S. A. Kulinich, J. Li, Y. Liu and H. Zeng, A general one-pot strategy for the synthesis of high-performance transparent-conducting-oxide nanocrystal inks for all-solution-processed devices, Angew. Chem., Int. Ed., 2015, 54, 462–466, DOI:10.1002/anie.201408621.
- A. Scarangella, R. Reitano, F. Priolo and M. Miritello, Bismuth doping of silicon compatible thin films for telecommunications and visible light emitting devices, Mater. Sci. Semicond. Process., 2019, 92, 47–57, DOI:10.1016/j.mssp.2018.04.017.
- D. M. Jang, K. Park, D. H. Kim, J. Park, F. Shojaei, H. S. Kang, J. P. Ahn, J. W. Lee and J. K. Song, Reversible halide exchange reaction of organometal trihalide perovskite colloidal nanocrystals for full-range band gap tuning, Nano Lett., 2015, 15, 5191–5199, DOI:10.1021/acs.nanolett.5b01430.
- W. Rehman, D. P. McMeekin, J. B. Patel, R. L. Milot, M. B. Johnston, H. J. Snaith and L. M. Herz, Photovoltaic mixed-cation lead mixed-halide perovskites: links between crystallinity, photo-stability and electronic properties, Energy Environ. Sci., 2017, 10, 361–369, 10.1039/C6EE03014A.
- M. Lu, X. Zhang, X. Bai, H. Wu, X. Shen, Y. Zhang, W. Zhang, W. Zheng, H. Song, W. W. Yu and A. L. Rogach, Spontaneous silver doping and surface passivation of CsPbI3 perovskite active layer enable light-emitting devices with an external quantum efficiency of 11.2%, ACS Energy Lett., 2018, 3, 1571–1577, DOI:10.1021/acsenergylett.8b00835.
- L. Protesescu, S. Yakunin, S. Kumar, J. Bar, F. Bertolotti, N. Masciocchi, A. Guagliardi, M. Grotevent, I. Shorubalko, M. I. Bodnarchuk, C. J. Shih and M. V. Kovalenko, Dismantling the 'Red Wall' of colloidal perovskites: highly luminescent formamidinium and formamidinium-cesium lead iodide nanocrystals, ACS Nano, 2017, 11, 3119–3134, DOI:10.1021/acsnano.7b00116.
- H. Huang, M. I. Bodnarchuk, S. V. Kershaw, M. V. Kovalenko and A. L. Rogach, Lead halide perovskite nanocrystals in the research spotlight: stability and defect tolerance, ACS Energy Lett., 2017, 2, 2071–2083, DOI:10.1021/acsenergylett.7b00547.
- Q. A. Akkerman, G. Raino, M. V. Kovalenko and L. Manna Genesis, Genesis, challenges and opportunities for colloidal lead halide perovskite nanocrystals, Nat. Mater., 2018, 17, 394–405, DOI:10.1038/s41563-018-0018-4.
- Z. Li, M. Yang, J. Park, S. Wei, J. J. Berry and K. Zhu, Stabilizing perovskite structures by tuning tolerance factor: formation of formamidinium and cesium lead iodide solid-state alloys, Chem. Mater., 2015, 28, 284–292, DOI:10.1021/acs.chemmater.5b04107.
- W. J. Mir, Y. Mahor, A. Lohar, M. Jagadeeswararao, S. Das, S. Mahamuni and A. Nag, Postsynthesis doping of Mn and Yb into CsPbX3 (X = Cl, Br, or I) perovskite nanocrystals for down-conversion emission, Chem. Mater., 2018, 30, 8170–8178, DOI:10.1021/acs.chemmater.8b03066.
- Y. Liu, W. Wang, X. Xu, J. P. Marcel Veder and Z. Shao, Recent advances in anion-doped metal oxides for catalytic applications, J. Mater. Chem. A, 2019, 7(13), 7280–7300, 10.1039/C8TA09913H.
- T. Jia, Z. Zeng, H. Q. Lin, Y. Duan and P. Ohodnicki, First-principles study on the electronic, optical and thermodynamic properties of ABO3 (A = La, Sr, B, Fe, Co) perovskites, RSC Adv., 2017, 7, 38798–38804, 10.1039/c7ra06542f.
- E. G. Rini, M. N. Rao, S. L. Chaplot, N. K. Gaur and R. K. Singh, Phonon dynamics of lanthanum manganite LaMnO3 using an interatomic shell model potential, Phys. Rev. B:Condens. Matter Mater. Phys., 2000, 75, 214301, DOI:10.1103/PhysRevB.75.214301.
- A. Hassan, M. Ismail, A. H. Reshak, Z. Zada, A. A. Khan, F. U. Rehman, M. Arif, K. Siraj, S. Zada, G. Murtaza and M. M. Ramli, Effect of heteroatoms on structural, electronic and spectroscopic properties of polyfuran, polythiophene and polypyrrole: A hybrid DFT approach, J. Mol. Struct., 2023, 1274, 134484, DOI:10.1016/j.molstruc.2022.134484.
- H. Yu, H. Huang, A. H. Reshak, S. Auluck, L. Liu, T. Ma and Y. Zhang, Coupling ferroelectric polarization and anisotropic charge migration for enhanced CO2 photoreduction, Appl. Catal., B, 2021, 284, 119709, DOI:10.1016/j.apcatb.2020.119709.
- R. Singla, S. Kumar, T. A. Hackett, A. H. Reshak and M. K. Kashyap, Genesis of magnetism in graphene/MoS2 van der Waals heterostructures via interface engineering using Cr-adsorption, J. Alloys Compd., 2021, 859, 157776, DOI:10.1016/j.jallcom.2020.157776.
- D. M. Hoat, S. Amirian, H. Alborznia, A. Laref, A. H. Reshak and M. Naseri, Strain effect on the electronic and optical properties of 2D Tetra-hexcarbon: a DFT-based study, Indian J. Phys., 2021, 95(11), 2365–2373, DOI:10.1007/s12648-020-01913-1.
- J. Thakur, M. K. Kashyap, H. S. Saini and A. H. Reshak, Half metallicity and magnetism in graphene containing mono-vacancies decorated with Carbon/Nitrogen adatom, J. Alloys Compd., 2016, 663, 100–106, DOI:10.1016/j.jallcom.2015.12.117.
- M. Husain, N. Rahman, A. H. Reshak, Zulfiqar, A. Habib, S. Ali, A. Laref, A. M. M. A. Bakri and J. Bila, Insight into the physical properties of the inter-metallic titanium-based binary compounds, Eur. Phys. J. Plus, 2021, 136, 624, DOI:10.1140/epjp/s13360-021-01590-x.
- S. Tabassam, A. H. Reshak, G. Murtaza, S. Muhammad, A. Laref, M. Yousaf, A. M. M. A. Bakri and J. Bila, Co2YZ (Y = Cr, Nb, Ta, V and Z = Al, Ga) Heusler alloys under the effect of pressure and strain, J. Mol. Graphics Modell., 2021, 104, 107841, DOI:10.1016/j.jmgm.2021.107841.
- J. Pelleg, Mechanical properties of materials, Springer Science & Business Media, 2012, vol. 190 Search PubMed.
- K. Balasubramanian, N. Rajeswari and K. Vaidheeswaran, Analysis of mechanical properties of natural fibre composites by experimental with FEA, Mater. Today: Proc., 2020, 28, 1149–1153 CAS.
- S. Eshraghi and S. Das, Micromechanical finite-element modeling and experimental characterization of the compressive mechanical properties of polycaprolactone–hydroxyapatite composite scaffolds prepared by selective laser sintering for bone tissue engineering, Acta Biomater., 2012, 8(8), 3138–3143 CrossRef CAS PubMed.
- C. G. F. Blum, et al., Exploring the details of the martensite–austenite phase transition of the shape memory Heusler compound Mn2NiGa by hard x-ray photoelectron spectroscopy, magnetic and transport measurements, Appl. Phys. Lett., 2011, 98(25), 252501 CrossRef.
- J. Winterlik, B. Balke, G. H. Fecher, C. Felser, M. C. M. Alves, F. Bernardi and J. Morais, Structural, electronic, and magnetic properties of tetragonal Mn3−xGa: Experiments and first-principles calculations, Phys. Rev. B:Condens. Matter Mater. Phys., 2008, 77, 054406 CrossRef.
- Z. Hashin and S. Shtrikman, A variational approach to the theory of the elastic behaviour of multiphase materials, J. Mech. Phys. Solids, 1963, 11(2), 127–140 CrossRef.
- T. Zohdi and P. Wriggers, Aspects of the computational testing of the mechanical properties of microheterogeneous material samples, Int. J. Numer. Methods Eng., 2001, 50(11), 2573–2599 CrossRef.
- O. L. Anderson, E. Schreiber, R. C. Liebermann and N. Soga, Some elastic constant data on minerals relevant to geophysics, Rev. Geophys., 1968, 6(4), 491–524 CrossRef CAS.
- B. B. Karki, L. Stixrude and R. M. Wentzcovitch, Highpressure elastic properties of major materials of Earth's mantle from first principles, Rev. Geophys., 2001, 39(4), 507–534 CrossRef CAS.
- H. B. Huntington, The elastic constants of crystals, Solid State Phys., 1958, 7, 213–351 CAS.
- M. A. Alam, M. A. Hadi, M. T. Nasir, M. Roknuzzaman, F. Parvin, M. A. K. Zilani, A. K. M. A. Islam and S. H. Naqib, Structural, Elastic, and Electronic Properties of Newly Discovered Li2PtSi3 Superconductor: Effect of Transition Metals, J. Supercond. Novel Magn., 2016, 29(10), 2503–2508 CrossRef CAS.
- G. Kresse and J. Hafner, Ab initio molecular dynamics for liquid metals, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 47, 558–561, DOI:10.1103/PhysRevB.47.558.
- G. Kresse and J. Hafner, Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 49, 14251–14269, DOI:10.1103/PhysRevB.49.14251.
- G. Kresse and J. Furthmüller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set, Comput. Mater. Sci., 1996, 6, 15–50, DOI:10.1016/0927-0256(96)00008-0.
- G. Kresse and J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169–11186, DOI:10.1103/physrevb.54.11169.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953–17979, DOI:10.1103/physrevb.50.17953.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. Lett., 1996, 77, 3865–3868, DOI:10.1103/PhysRevLett.77.3865.
- S. Grimme, Semiempirical GGA-type density functional constructed with a long-range dispersion correction, J. Comput. Chem., 2006, 27, 1787–1799, DOI:10.1002/jcc.20495.
- M. I. Naher, M. A. Afzal and S. H. Naqib, A comprehensive DFT based insights into the physical properties of tetragonal superconducting Mo5PB2, Results Phys., 2021, 28, 104612, DOI:10.1016/j.rinp.2021.104612.
- M. Born and K. Huang, Dynamical theory of crystal lattices in The International Series of Monographs of Physics, Oxford University Press, Amen House, London, 1954 (Clarendon press) Search PubMed.
- L. Kleinman, Phys. Rev., 1962, 128, 2614, DOI:10.1103/PhysRev.128.2614.
- S. I. Ranganathan and M. Ostoja-Starzewski, Universal Elastic Anisotropy Index, Phys. Rev. Lett., 2008, 101, 055504, DOI:10.1103/PhysRevLett.101.055504.
- D. H. Chung and W. R. Buessem, The Elastic Anisotropy of Crystals, J. Appl. Phys., 1967, 38, 2010–2012, DOI:10.1063/1.1709819.
| This journal is © The Royal Society of Chemistry 2026 |