Open Access Article
Open Access ArticlePhosphorous acid-mediated novel synthesis of MnPO4·H2O and Mn2P2O7 with a unique flower-like morphology: a new approach to manganese phosphate supercapacitors
Prakash Bobde ab,
Kajal Uphadec,
Sharath Kandambethc,
Shikha Wadhwad and
Ranjit Kumar
ab,
Kajal Uphadec,
Sharath Kandambethc,
Shikha Wadhwad and
Ranjit Kumar *e
*e
aTechknowgreen Research Laboratory-9, Building G A-4, 3rd Floor, Waters Square, New DP Road, Pimpri-Chinchwad, Pune, 411027, India
bDepartment of Research & Development, UPES University, Energy Acres Building, Bidholi, Dehradun, 248007, Uttarakhand, India
cDepartment of Chemistry, Shiv Nadar Institution of Eminence, Delhi-NCR, 201314, India
dApplied Science Cluster, School of Engineering, UPES University, Energy Acres Building, Bidholi, Dehradun, 248007, Uttarakhand, India
eCenter for Advanced Materials, Department of Chemical Engineering, Shiv Nadar Institution of Eminence, Delhi-NCR, 201314, India. E-mail: ranjit.kumar@snu.edu.in
First published on 22nd April 2026
Abstract
Manganese phosphates in various forms have garnered substantial interest because of their applications in energy storage, catalysis, and materials science. In this study, we present a novel phosphorous acid (H3PO3)-mediated approach for the facile synthesis of MnPO4·H2O and Mn2P2O7 with a unique flower-like morphology. As precursors, KMnO4 and H3PO3 were chosen to hydrothermally synthesize MnPO4·H2O. The crystalline Mn2P2O7 was produced by heating MnPO4·H2O for two hours at 700 °C. XRD, FTIR, FESEM, TEM, and other techniques were used to characterize the materials. The monoclinic structure of MnPO4·H2O and Mn2P2O7 was confirmed by XRD. A possible formation mechanism is proposed, highlighting the role of H3PO3 as both a phosphorus source and a structure-directing agent. The electrochemical study showed that Mn2P2O7 has a specific capacitance of 169 F g−1, whereas that of MnPO4·H2O is 63 F g−1 at a current density of 1 A g−1. This approach provides a straightforward, economical, and scalable pathway to manganese phosphates with a unique morphology, paving the way for a potential future material with enhanced performance in electrochemical and catalytic applications.
1. Introduction
Manganese phosphates, encompassing compounds such as MnPO4·H2O and Mn2P2O7, represent a versatile class of materials that have gained substantial attention recently for their promising roles in energy storage systems, catalytic processes, and advanced materials engineering.1 Transition metal phosphate materials exhibit intriguing characteristics due to their extensive use in electric, magnetic, dielectric, ceramic, laser host, and catalytic procedures.2,3 A large number of studies have documented the ion exchange properties of transition metal phosphates.3–5 Transition metal phosphates are extensively used as adsorbents, ionic conductors, catalysts, ion exchangers, catalyst carriers and catalysts due to their diverse structures.6 These materials have found applications in supercapacitors, magnetic materials, heat resistant materials, friction materials, and molecular recognition, because of their exceptional physical and chemical characteristics.7 As a result, transition metal phosphates have recently gained popularity as a materials science research area.4,8,9The phosphate block unit is used to separate the metal phosphate compounds into PO43−, HPO42−, H2PO4−, P2O74−, and P4O124− units.1 Metal(III) phosphates are generally insoluble in water and commonly occur in amorphous, crystalline, or partially crystalline forms. These metal phosphates have varying levels of crystallinity and pH-dependent surface charge characteristics.6 The use of MnPO4·H2O as a functional material, such as a cathode material for lithium-ion batteries, is made easier by manganese's various oxidation states.6,8,10 Notably, the morphology of these phosphates plays a critical role in enhancing their performance; hierarchical structures, such as flower-like architectures, offer increased surface area, improved ion diffusion pathways, and better electrochemical accessibility compared to bulk forms. It is crucial to look for a low-cost, straightforward synthesis approach for the bulk production of MnPO4·H2O to increase its use.6,8,11–13 Christensen et al.14 initially prepared MnPO4·H2O by combining manganese(III) nitrate with phosphoric acid and nitric acid, as well as by oxidizing manganese(II) nitrate with phosphoric acid. Other researchers used manganese(II) carbonate as a source and oxidized it by the action of nitric acid and phosphoric acid as part of an additional preparation technique.15–17 Zhang et al. used Mn(NO3)2 with H3PO4 to synthesize hydrothermally controlled MnPO4·H2O, at 130 °C in 16 h.18 Hu et al. synthesized microporous manganese phosphate using organophosphonic acid from bulk manganese oxide via a soft templating method.19 These previously documented techniques involved the evolution of harmful gases, including NO2 and CO2, and were time-consuming at high temperatures.20
The precipitation, sol–gel, and hydrothermal processes used to prepare manganese phosphate hydrate allowed for a close mixing of the solution's constituent elements, which facilitated quick homogenous nucleation, producing finer particles and higher-purity materials.21–24 Mixed orthophosphates, like LiMPO4 (where M can be Fe, Co, Ni, or Mn), provide good electrochemical performances in lithium-ion battery cathode materials and are another area of interest for solution-based approaches.17,20–23 Manganese phosphate, when prepared using the typically high-temperature process and using concentrated acid (conc. HNO3), causes particle aggregation and sintering, which improves its electrochemical performance.20
Many methods were used to prepare MnPO4·H2O, and each had its own pros and cons. The precipitation procedure was often used to produce MnPO4·H2O. This method involved mixing soluble manganese salts with phosphate sources in water. Although this method is easy to use and reasonably economical, it does not give precise control over crystal size, shape, and purity. The wide size range of the particles produced affects the material's properties and performance.21–24
Researchers have also used the sol–gel method, which involves breaking down and combining metal alkoxides or salts to create a sol. This sol is then converted to manganese phosphate monohydrate via gelation. While the sol–gel method allows for better control over the composition and consistency of the final product, it is often slow and requires high temperatures during the gelation process.16,24,25 Researchers have studied microwave irradiation to encourage the nucleation and growth of cobalt manganese phosphate crystals.26 This method reduces the time of synthesis and improves control over particle size. But it is important to carefully manage the synthesis conditions to prevent overheating and ensure consistent microwave energy distribution, as this could cause unwanted side effects.26,27 The hydrothermal method was chosen for manganese phosphate monohydrate because it produces high-purity, well-defined crystals with better properties. Its scalability, energy efficiency, and eco-friendliness made it a good option for large-scale industrial production of MnPO4·H2O.28,29 In the hydrothermal synthesis method, the reaction takes place in a closed vessel under high temperatures and high pressures.28,29
For the synthesis of MnPO4·H2O, similar challenges persisted despite using various synthesis techniques in the past. These challenges included complex or slow synthesis processes, difficulties in achieving high purity, trouble in controlling crystal size and shape, and the possible use of harmful chemicals or conditions in certain methods.20,30 To overcome these limitations and boost the production of MnPO4·H2O with better properties for various applications, such as environmental clean-up, battery technology, and catalysis, scientists have previously explored various synthesis methods.20,30 Furthermore, one-pot syntheses that integrate reduction and phosphorylation steps without additional oxidants or pH regulators are underrepresented, particularly for achieving hierarchical flower-like structures in a facile manner. Traditional reductive approaches often employ harsh agents, leading to byproduct formation and purification challenges, while morphology tuning typically demands multi-step processing or specialized precursors.
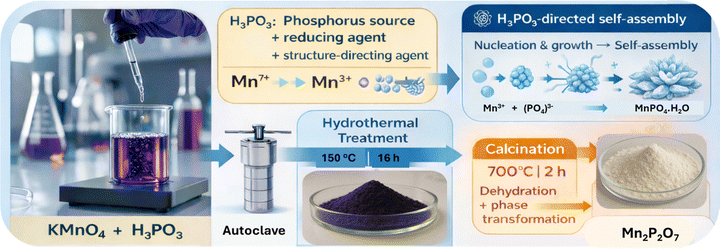
The present work introduces a phosphorous acid-mediated reductive pathway, which differs fundamentally in reaction chemistry from conventional phosphate-based methods that use Mn(II) or Mn(III) salts as precursors, and enables the formation of unique flower-like morphologies without external templating agents. Here we present hydrothermal synthesis of nanocrystalline MnPO4·H2O at 150 °C using only KMnO4 and H3PO3 as precursors. In this approach, H3PO3 acts both as a reducing agent and a phosphate source, enabling a simple, rapid, low-cost, and eco-friendly one-pot process that avoids the use of additional manganese salts and does not release any harmful gases. Subsequent calcination at 700 °C conveniently converts MnPO4·H2O into Mn2P2O7. To the best of our knowledge, this is the first reported synthesis of manganese phosphates via reduction with phosphorus acid (H3PO3). The simplicity of the process, combined with the high phase purity and unique morphology achieved, offers a significant advancement over conventional multi-reagent routes and provides an efficient pathway for producing these materials for potential applications in catalysis, supercapacitors, and battery materials.
2. Experimental
2.1. Materials and chemicals
KMnO4 (AR grade) and H3PO3 (AR grade) were purchased from Fisher Scientific, India. The Teflon-lined stainless steel autoclave (250 mL) was purchased from Shilpa Enterprises, Nagpur, Maharashtra.2.2. Synthesis
2.3. Characterization
MnPO4·H2O and Mn2P2O7 were characterized using Fourier transform infrared (FTIR) spectroscopy (PerkinElmer Frontier FTIR). FTIR was used to identify the surface functional groups present in MnPO4·H2O and Mn2P2O7. X-ray diffraction (XRD) (Bruker D8-Advance Eco) was utilized to analyze their crystalline structures. The surface morphology and structural features of MnPO4·H2O and Mn2P2O7 were examined using a JEOL JSM-7600PLUS field emission scanning electron microscope (FE-SEM). High-resolution transmission electron microscopy (HRTEM) images were acquired using a JOEL JEM2100 HRTEM, and the images were obtained using an accelerating voltage of 300 kV. The thermal stability of the materials was studied using thermogravimetric analysis (TGA2 System, Mettler Toledo) under N2 flow at a heating ramp of 10 °C per minute. The specific surface area was measured by the Brunauer–Emmett–Teller (BET) method using a Microtrac BELSORP MAX G instrument.2.4. Electrochemical study
Electrochemical charge storage experiments were conducted using a Bio-Logic SP-50e potentiostat in a three-electrode configuration. The working electrode was fabricated by depositing the active material onto carbon paper (1 cm × 3 cm). For electrode preparation, 8 mg of Mn2P2O7 or MnPO4·4H2O powder was dispersed with 1 mg of conductive carbon and 1 mg of Nafion binder in 1 mL of ethanol. Following 10 minutes of sonication, the homogeneous suspension was drop-cast onto the carbon paper substrate. The final deposited mass of the sample was 1 mg. Prior to electrochemical testing, the prepared electrodes were dried under an IR lamp. A graphite counter electrode and Hg–HgO reference electrode were utilized, with measurements conducted in 1.0 M KOH electrolyte at ambient temperature.3. Results and discussion
3.1. Characterization
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
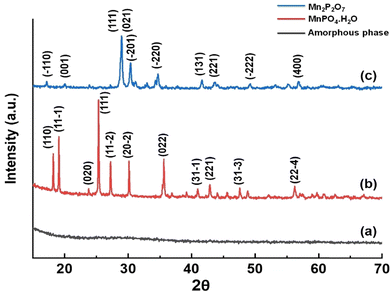
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ), where λ is the wavelength of Cu-Kα radiation, D is 0.89 (constant), θ is the diffraction angle, and β is the full width at half maximum (FWHM).20 The reflections at 2θ values 18.2(110), 19.1(11
θ), where λ is the wavelength of Cu-Kα radiation, D is 0.89 (constant), θ is the diffraction angle, and β is the full width at half maximum (FWHM).20 The reflections at 2θ values 18.2(110), 19.1(11![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), 23.8 (020), 25.3(111), 27.2(11
), 23.8 (020), 25.3(111), 27.2(11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) ), 30.2(20
), 30.2(20![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) ), 35.6(022), 41.0(31
), 35.6(022), 41.0(31![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), 42.9(221), 47.6(31
), 42.9(221), 47.6(31![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) and 56.2(22
) and 56.2(22![[4 with combining macron]](https://www.rsc.org/images/entities/char_0034_0304.gif) ) match perfectly with the peaks of MnPO4·H2O. The lattice parameters of MnPO4·H2O closely match those reported in the standard reference JCPDS# 440071. The peaks in Fig. 2(c) at 2θ values 17.1(
) match perfectly with the peaks of MnPO4·H2O. The lattice parameters of MnPO4·H2O closely match those reported in the standard reference JCPDS# 440071. The peaks in Fig. 2(c) at 2θ values 17.1(![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 10), 20.0(001), 28.7(111), 28.9(021), 30.4(
10), 20.0(001), 28.7(111), 28.9(021), 30.4(![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 01), 34.7(
01), 34.7(![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 20), 41.6(131), 43.5(221), 49.2(
20), 41.6(131), 43.5(221), 49.2(![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 22), and 56.9 (400) were matched with the Mn2P2O7 (JCPDS# 290891). These results indicate that Mn2P2O7 exhibits a monoclinic crystal structure with the space group C2/m. The average crystallite size of 30.82 nm for the Mn2P2O7 sample was calculated using the Scherrer equation.20 The lattice parameters of Mn2P2O7 match those of the standard data (JCPDS# 290891). The XRD data confirm the phase purity of the synthesized material (Table 1).
22), and 56.9 (400) were matched with the Mn2P2O7 (JCPDS# 290891). These results indicate that Mn2P2O7 exhibits a monoclinic crystal structure with the space group C2/m. The average crystallite size of 30.82 nm for the Mn2P2O7 sample was calculated using the Scherrer equation.20 The lattice parameters of Mn2P2O7 match those of the standard data (JCPDS# 290891). The XRD data confirm the phase purity of the synthesized material (Table 1).
| Lattice parameters | MnPO4·H2O | Mn2P2O7 | ||||
|---|---|---|---|---|---|---|
| This work | PDF#440071 | Difference (this work-PDF) | This work | PDF#290891 | Difference (this work-PDF) | |
| a (Å) | 6.947 | 6.928 | 0.019 | 6.657 | 6.636 | 0.021 |
| b (Å) | 7.483 | 7.478 | 0.005 | 8.592 | 8.584 | 0.008 |
| c (Å) | 7.386 | 7.372 | 0.014 | 4.540 | 4.545 | −0.005 |
| β (°) | 112.28 | 112.30 | −0.020 | 102.86 | 102.78 | 0.080 |
| Average crystallite size | 56.17 nm | 30.82 nm | ||||
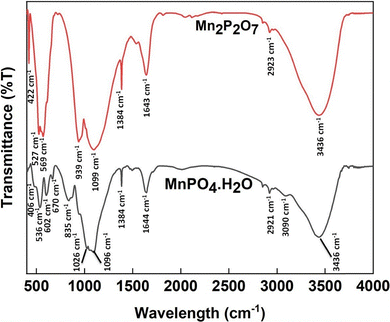
The FTIR spectra of Mn2P2O7 shown in Fig. 3 indicate the fundamental vibrations of the P2O74− anion. Among the spectrum's most notable characteristics are the strong bands located at 1099, 939, 569, 527, and 422 cm−1. υas (PO3), υas (P–O–P), δ (PO3), δ (PO3), and ρ (PO3) can all be attributed to these bands.1 The symmetric and asymmetric stretching vibrations of the P–O–P bridge (υas POP and υs POP) observed in the 700–970 cm−1 range confirm P2O74− anions with bent P–O–P angles.1 Comparing the two spectra, it can be clearly seen that the peaks at 835 and 602 cm−1 in MnPO4·H2O disappear completely in Mn2P2O7.17 These two peaks are assigned to the stretching vibrations of P–OH and the bending vibration of Mn–O–H, respectively, which disappear after calcination when the structure is converted to Mn2P2O7.1,17
![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 01), 2.58 Å (220), and 2.17 Å (131), which would produce rings at corresponding reciprocal distances. The observed pattern's ring distribution and spotty nature are consistent with nanocrystalline Mn2P2O7, often seen in derivatives from thermal decomposition of precursors like manganese phosphates.34
01), 2.58 Å (220), and 2.17 Å (131), which would produce rings at corresponding reciprocal distances. The observed pattern's ring distribution and spotty nature are consistent with nanocrystalline Mn2P2O7, often seen in derivatives from thermal decomposition of precursors like manganese phosphates.34
 | ||
| Fig. 5 TEM micrographs of MnPO4·H2O (a) and (b) and Mn2P2O7 (d) and (e), and SAED patterns of (c) MnPO4·H2O and (f) Mn2P2O7. | ||
In comparison, MnPO4·H2O has a distinct monoclinic structure (space group C2/c). Its prominent d-spacings are larger overall, such as 4.86 Å (110), 4.64 Å (11![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ), 3.51 Å (111), 3.32 Å (11
), 3.51 Å (111), 3.32 Å (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) ), 2.95 Å (20
), 2.95 Å (20![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) ), and 2.52 Å (022). An SAED pattern for MnPO4·H2O would thus feature inner rings at smaller reciprocal distances (larger d-values) compared to Mn2P2O7, reflecting its different unit cell and bonding (orthophosphate PO43− groups with coordinated water vs. pyrophosphate P2O74− in Mn2P2O7).
), and 2.52 Å (022). An SAED pattern for MnPO4·H2O would thus feature inner rings at smaller reciprocal distances (larger d-values) compared to Mn2P2O7, reflecting its different unit cell and bonding (orthophosphate PO43− groups with coordinated water vs. pyrophosphate P2O74− in Mn2P2O7).
3.2. Plausible mechanism of MnPO4·H2O and Mn2P2O7 formation
The synthesis of manganese phosphate monohydrate (MnPO4·H2O) using phosphorous acid (H3PO3) and potassium permanganate (KMnO4) as the sole manganese source involves a redox-driven precipitation reaction under controlled conditions. KMnO4 is a strong oxidizing agent in aqueous solution, where it dissociates to form permanganate ions (MnO4−). The manganese in MnO4− is in the +7 oxidation state (Mn(VII)). In the presence of a reducing agent, MnO4− can be reduced to lower oxidation states, such as Mn(III), which is required for the formation of MnPO4·H2O, where manganese exists as Mn3+.35 The progress of the reaction was monitored by UV-vis spectroscopy (Fig. S1) and the colour change of the solution. The colour of the solution changed from purple (Mn7+) to brown pink (Mn3+), which was also indicated by the decrease in absorbance of typical peaks of MnO4− species (490–570 nm) as H3PO3 was added to the KMnO4 solution. Possibly, some Mn2+ also forms transiently or as an intermediate during the initial reduction step. However, the hydrothermal treatment oxidises any remaining Mn2+ phase (or dissolved Mn2+) to the Mn3+ product, because of the presence of MnO4−.Phosphorous acid (H3PO3) serves a dual purpose. First, it acts as a reducing agent, capable of reducing Mn(VII) in MnO4− to Mn(III). Second, upon oxidation, H3PO3 is converted to phosphate ions (PO43−), which are essential for forming the MnPO4·H2O product. The oxidation of H3PO3 to H3PO4 (phosphoric acid) provides the phosphate groups necessary for the precipitation reaction. H3PO3 plays another significant role as a structure-directing agent. The presence of PO33− ions in the solution helps in the development of a unique flower-like morphology as was evidenced in the FESEM images.36,37
Redox reaction: The synthesis begins with the redox reaction between MnO4− and H3PO3 (eqn (1)). In an acidic aqueous medium, the permanganate ion is reduced, and H3PO3 is oxidized. The simplified redox reaction can be represented as follows:
Reduction half-reaction:
| MnO4− + 8H+ + 4e− → Mn3+ + 4H2O | (1) |
Here, Mn(VII) is reduced to Mn(III).
Oxidation half-reaction:
| H3PO3 + H2O → H3PO4 + 2H+ + 2e− | (2) |
H3PO3 is oxidized to H3PO4, releasing phosphate ions (PO43−) upon dissociation of H3PO4 in solution (eqn (2)).
The overall redox reaction can be written as
| MnO4− + H3PO3 + 4H+ → Mn3+ + H3PO4 + 2H2O | (3) |
Formation of MnPO4·H2O: once Mn3+ and PO43− ions are generated in the solution, they combine to form the insoluble MnPO4·H2O precipitate (eqn (4)). The reaction can be written as:
| Mn3+ + PO43− + H2O → MnPO4·H2O (s) | (4) |
The monohydrate form incorporates one water molecule per formula unit, which is typical for manganese phosphate synthesized under aqueous conditions. The reaction conditions, such as pH, temperature, and concentration, are critical to ensure the formation of the monohydrate phase with high crystallinity and purity.
The synthesis typically occurs in an aqueous medium under mild conditions resulting in the formation of an amorphous manganese phosphate. Treatment of this amorphous manganese phosphate by the hydrothermal method at 150 °C results in a crystalline MnPO4·H2O phase as confirmed by X-ray diffraction analysis. The pH of the solution influences the dissociation of H3PO4 and the stability of the Mn3+ ions. An acidic (pH ∼ 1.5) environment is maintained to facilitate the redox reaction and prevent the formation of undesirable manganese oxides or hydroxides.
Upon heating to 700 °C, the anhydrous MnPO4 undergoes a condensation reaction, where two orthophosphate units combine, releasing oxygen to form manganese pyrophosphate (Mn2P2O7) (eqn (5)).38 The reaction can be written as:
| 2MnPO4 → Mn2P2O7 + ½O2 | (5) |
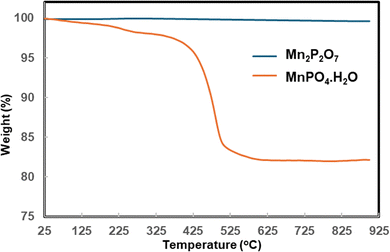
Mn2P2O7, which is a thermally stable pyrophosphate phase, consists of P2O74− anions (pyrophosphate groups) coordinated with Mn2+ cations, forming a crystalline lattice. The transformation of MnPO4·H2O to Mn2P2O7 upon calcining at 700 °C proceeds through dehydration to form MnPO4, followed by thermal decomposition and condensation of the phosphate units, resulting in the stable pyrophosphate phase. The TGA data support this, with the observed weight losses aligning with the loss of water and structural reorganisation, resulting in the formation of Mn2P2O7.
H3PO3's dual role as a reducing agent and phosphate source further streamlines the process, reducing the number of reagents and minimizing byproducts. The redox-driven mechanism enables precise control over the formation of MnPO4·H2O, ensuring high phase purity with unique morphology. This synthesis method for MnPO4·H2O under appropriate aqueous conditions and subsequent calcination to produce Mn2P2O7 offers an efficient route to this material for applications in catalysis, supercapacitors, battery materials, and other fields.
3.3. Electrochemical performance of Mn2P2O7
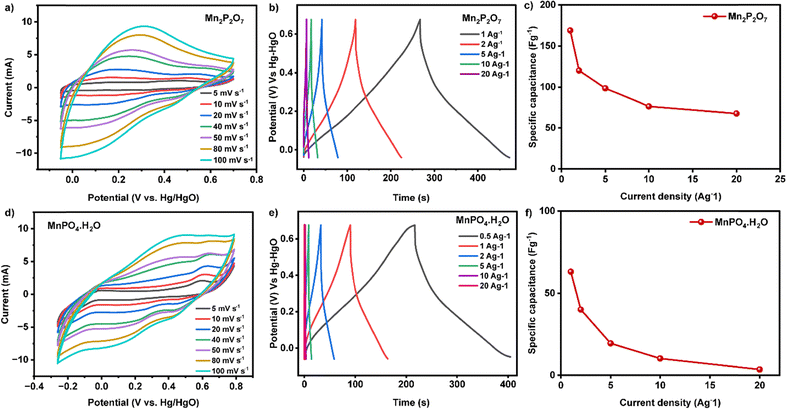
To study the electrochemical properties of Mn2P2O7 and MnPO4·H2O, a three-electrode configuration was employed with 1 M KOH electrolyte, a reference electrode of Hg–HgO, and a counter electrode made of graphite. Cyclic voltammetry (CV) measurements of Mn2P2O7 conducted at various scan rates revealed that the capacitance contribution predominantly occurs in the positive potential window (−0.04 V to 0.67 V vs. Hg–HgO), as shown in Fig. 8. The CV curve of Mn2P2O7 at 5 mV s−1 exhibited two distinct redox peaks at approximately 0.38 V and 0.47 V (vs. Hg–HgO).Similarly, MnPO4·4H2O displayed capacitive behaviour in the positive potential range (−0.26 V to 0.8 V) at 10 mV s−1 scan rate. The CV profile of MnPO4 showed characteristic peaks at 0 V and 0.6 V. To further assess the charge storage capabilities of these materials, galvanostatic charge/discharge (GCD) measurements were performed by varying the current density from 1 to 20 A g−1.
The following formula was used to calculate specific capacitance (Cs, F g−1) from the GCD curves:
| Cs = 2I∫Vdt/m(ΔV)2 |
GCD analysis revealed that Mn2P2O7 showed a specific capacitance value of 169 F g−1 at 1 A g−1 current density. Although the specific capacitance decreased upon increasing current density, Mn2P2O7 still retained 40% of its initial specific capacitance value even at a high current density. In contrast, MnPO4 exhibited a lower specific capacitance value of 63 F g−1 at 1 A g−1 and showed a drastic decrease in specific capacitance (5.5%) at higher current density (20 A g−1), yielding only 3.5 F g−1.39
When compared with MnPO4·H2O, Mn2P2O7 exhibits a markedly superior electrochemical performance. At a current density of 1 A g−1, Mn2P2O7 delivers a specific capacitance of 169 F g−1, which is nearly 2.7 times higher than that of MnPO4·H2O (63 F g−1). This significant enhancement can be attributed to the distinct phosphate framework and higher density of electrochemically active manganese sites in Mn2P2O7, which facilitate more effective faradaic redox reactions (Mn2+/Mn3+). Additionally, the pyrophosphate structure is likely to provide improved structural robustness and better ion diffusion pathways compared to the hydrated orthophosphate phase, thereby contributing to enhanced charge-storage capability. The relatively lower capacitance of MnPO4·H2O may arise from its more compact crystal structure and the presence of coordinated water molecules, which can impede electron transport and limit the utilization of the active material. The electrochemical performance of Mn2P2O7, with a specific capacitance of 169 F g−1 at a current density of 1 A g−1, highlights its potential as a promising pseudocapacitive electrode material for supercapacitor applications. This moderate yet significant capacitance, achieved in a pure and unmodified form, reflects the intrinsic redox activity of manganese species and the reversible faradaic charge-storage mechanism of the phosphate framework. Owing to its structural stability, earth abundance, and environmentally benign nature, Mn2P2O7 is particularly attractive for cost-effective and sustainable energy storage systems. While the relatively low electrical conductivity of phosphate-based materials may limit its performance, Mn2P2O7 can serve as an efficient base material for further performance enhancement through nanostructuring or hybridization with conductive matrices. Therefore, Mn2P2O7 holds considerable potential for use in next-generation supercapacitors, especially in composite or asymmetric device configurations where improved power and energy densities are desired.39
Mn2P2O7 exhibited excellent specific capacity retention (100%) over 250 cycles at a current density of 5 A g−1 as shown in Fig. S3. Cyclic voltammetry (CV) at varying scan rates (5 to 100 mV s−1), was conducted within the selected potential window (0 V to 0.4 V), to calculate the electrochemically active surface area. The ECSA values are found to be 0.41 cm2 for the Mn2P2O7 sample.
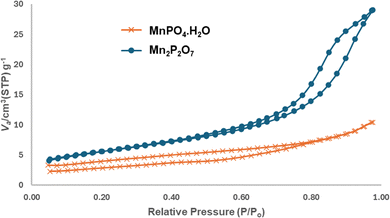
BET surface area calculations showed that Mn2P2O7 possessed a higher surface area (25.0 m2 g−1) than MnPO4·H2O (8.1 m2 g−1). Since capacitance directly correlates with the electrode surface area, MnPO4·H2O's limited surface area likely hindered its charge storage performance. This structural limitation explains why MnPO4·H2O exhibited substantially lower capacitance than Mn2P2O7, particularly at higher charging rates, where efficient charge distribution becomes crucial.
Although the specific capacitance of our Mn2P2O7 (169 F g−1 at 1 A g−1) is lower than certain nanostructured or composite Mn2P2O7 electrodes reported recently, it remains competitive among pure-phase materials synthesized via simple routes (Table 2). The performance gap relative to binder-free Ni-foam or polymer–composite electrodes is primarily attributed to differences in electrode configuration, conductivity, and mass loading. The flower-like morphology obtained through our H3PO3-mediated approach provides a good ion-accessible surface area while maintaining structural stability after calcination. Our future plans include making nanocomposites of Mn2P2O7 with other conductive materials to improve the supercapacitor performance.
| Material | Specific capacitance | Current density (A g−1) | Electrolyte (M KOH) | Electrode configuration | Ref. |
|---|---|---|---|---|---|
| Flower-like Mn2P2O7 | 169 F g−1 | 1 | 1 | Drop-cast on carbon paper | This work |
| Mn2P2O7 nanosheets (freeze-dried) | ∼140–180 F g−1 | 1 | 3 | Ni foam | 40 |
| Mn2P2O7 nanoclusters | ∼120–160 F g−1 | 1 | 1 | Three electrode | 41 |
| Mn2P2O7 (from Mn-organic phosphate, calcined at 550 °C) | 230.9 F g−1 | 0.5 | 3 | Three electrode | 39 |
| Mn2P2O7@polypyrrole (sunflower-like composite) | 658 F g−1 | 1 | 1 | Ni foam | 42 |
| Mn2P2O7 (in PANI hybrid study) | 750 C g−1 | 1 | 4 | Three electrode | 43 |
4. Conclusion
In conclusion, this study demonstrates a novel and efficient hydrothermal synthesis of MnPO4·H2O using KMnO4 and H3PO3 as precursors, followed by thermal treatment at 700 °C for 2 hours to yield crystalline Mn2P2O7. Unlike conventional methods that often require multiple manganese salts or complex phosphate sources, this approach streamlines the reaction by leveraging the strong oxidizing properties of KMnO4 and the dual functionality of H3PO3. This reduces the number of reagents, minimizes byproducts, and enhances phase purity, as confirmed by XRD analysis showing pure monoclinic phases for both compounds. These findings contribute to the advancement of the synthesis methodologies for manganese-based materials, with potential applications in catalysis, energy storage, anti-corrosion coatings, etc., paving the way for further exploration of tailored reaction conditions to optimize crystallite size and phase purity. Mn2P2O7 demonstrated a specific capacitance of 169 F g−1 at 1 A g−1, highlighting Mn2P2O7 as a promising manganese phosphate-based electrode material and a viable platform for further optimization toward high performance supercapacitor applications.Author contributions
Prakash Bobde: writing – review and editing, writing – original draft, validation, methodology, and investigation; Kajal Uphade: investigation; Sharath Kandambeth: investigation; Shikha Wadhwa: writing – review and editing, and resources; Ranjit Kumar: writing – review and editing, writing – original draft, resources, and conceptualisation.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors declare that all the data supporting the findings of this study are available within the paper. Should any raw data files be needed in another format, they are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ma01523e.
Acknowledgements
We acknowledge the characterization facility at the Indian Institute of Petroleum, Dehradun, for FESEM and TEM analysis. We would also like to thank the central characterization facility at UPES, Dehradun, for this study.References
- B. Boonchom, R. Baitahe, Z. Joungmunkong and N. Vittayakorn, Grass blade-like microparticle MnPO4·H2O prepared by a simple precipitation at room temperature, Powder Technol., 2010, 203(2), 310–314, DOI:10.1016/J.POWTEC.2010.05.022.
- H. Zhao and Z. Y. Yuan, Insights into Transition Metal Phosphate Materials for Efficient Electrocatalysis, ChemCatChem, 2020, 3797–3810, DOI:10.1002/cctc.202000360.
- S. Karafiludis, T. W. Ryll, A. G. Buzanich, F. Emmerling and T. M. Stawski, Phase stability studies on transition metal phosphates aided by an automated synthesis, CrystEngComm, 2023, 25(30), 4333–4344, 10.1039/d3ce00386h.
- G. Renaudin, S. Gomes and J. M. Nedelec, First-row transition metal doping in calcium phosphate bioceramics: A detailed crystallographic study, Materials, 2017, 10(1), 92, DOI:10.3390/ma10010092.
- X. Li, X. Xiao, Q. Li, J. Wei, H. Xue and H. Pang, Metal (M = Co, Ni) phosphate based materials for high-performance supercapacitors, Inorg. Chem. Front., 2018, 5(1), 11–28, 10.1039/C7QI00434F.
- G. Qiu, Z. Gao, H. Yin, X. Feng, W. Tan and F. Liu, Synthesis of MnPO4·H2O by refluxing process at atmospheric pressure, Solid State Sci., 2010, 12(5), 808–813, DOI:10.1016/j.solidstatesciences.2010.02.014.
- G. Tatrari, M. Karakoti, C. Tewari, S. Pandey, B. S. Bohra and A. Dandapat, et al., Solid waste-derived carbon nanomaterials for supercapacitor applications: a recent overview, Mater. Adv., 2021, 2(5), 1454–1484, 10.1039/D0MA00871K.
- N. Stegmann, C. Ochoa-Hernández, K. N. Truong, H. Petersen, C. Weidenthaler and W. Schmidt, The Mechanism and Pathway of Selective Partial Oxidation of n-Butane to Maleic Anhydride Studied on Titanium Phosphate Catalysts, ACS Catal., 2023, 13(24), 15833–15840, DOI:10.1021/acscatal.3c03172.
- C. Nithya, Transition Metal Phosphates/Phosphonates for Lithium-Ion Batteries, in Engineering Materials, 2023, pp. 283–299 DOI:10.1007/978-3-031-27062-8_16.
- T. Liu, C. Chen, S. Liu, Z. Chen, Z. Pu and Q. Huang, et al., Transition metal phosphides as noble-metal-alternative co-catalysts for solar hydrogen production, Coord. Chem. Rev., 2024, 521, 216145, DOI:10.1016/j.ccr.2024.216145.
- N. L. W. Septiani, Y. V. Kaneti, K. B. Fathoni, K. Kani, A. E. Allah and B. Yuliarto, et al., Self-assembly of two-dimensional bimetallic nickel-cobalt phosphate nanoplates into one-dimensional porous chainlike architecture for efficient oxygen evolution reaction, Chem. Mater., 2020, 32(16), 7005–7018, DOI:10.1021/acs.chemmater.0c02385.
- D. Zhang, L. L. Hu, Y. G. Sun, J. Y. Piao, X. S. Tao and Y. S. Xu, et al., Construction of uniform transition-metal phosphate nanoshells and their potential for improving Li-ion battery performance, J. Mater. Chem. A, 2018, 6(19), 8992–8999, 10.1039/c8ta01320a.
- Y. Wang, G. Hu, Z. Peng, K. Du, B. Zhang and Y. Cao, Self-assembly synthesis of dumbbell-like MnPO4·H2O hierarchical particle and its application in LiMnPO4/C cathode materials, Ceram. Int., 2021, 47(14), 19687–19693, DOI:10.1016/J.CERAMINT.2021.03.306.
- O. T. Christensen, Beiträge zur Kenntniss der Oxyde des Mangans, J. Prakt. Chem., 1883, 28(1), 1–37, DOI:10.1002/PRAC.18830280101.
- X. Bao, W. Bin Zhang, L. Zhang, Y. W. Guo, X. Zhou and X. L. Zhang, et al., Alkali cation intercalation manganese phosphate hydrate boosting electrochemical kinetics for pseudocapacitive energy storage, J. Materiomics, 2022, 8(4), 833–842, DOI:10.1016/j.jmat.2022.01.005.
- Y. Wang, G. Hu, Z. Peng, K. Du, B. Zhang and Y. Cao, Self-assembly synthesis of dumbbell-like MnPO4·H2O hierarchical particle and its application in LiMnPO4/C cathode materials, Ceram. Int., 2021, 47(14), 19687–19693, DOI:10.1016/j.ceramint.2021.03.306.
- B. Boonchom, S. Youngme, S. Maensiri and C. Danvirutai, Nanocrystalline serrabrancaite (MnPO4·H2O) prepared by a simple precipitation route at low temperature, J. Alloys Compd., 2008, 454(1–2), 78–82, DOI:10.1016/J.JALLCOM.2006.12.064.
- Y. Zhang, Y. Liu, S. Fu, F. Guo and Y. Qian, Hydrothermally Controlled Growth of MnPO4·H2O Single-Crystal Rods, Bull. Chem. Soc. Jpn., 2006, 79(2), 270–275, DOI:10.1246/bcsj.79.270.
- Y. Hu, Y. Zhang, C. Li, L. Wang, Y. Du and G. Mo, et al., Guided Assembly of Microporous/Mesoporous Manganese Phosphates by Bifunctional Organophosphonic Acid Etching and Templating, Adv. Mater., 2019, 31(25), 1–8, DOI:10.1002/adma.201901124 , PubMed PMID: 31062894.
- B. Boonchom, S. Youngme, S. Maensiri and C. Danvirutai, Nanocrystalline serrabrancaite (MnPO4·H2O) prepared by a simple precipitation route at low temperature, J. Alloys Compd., 2008, 454(1–2), 78–82, DOI:10.1016/j.jallcom.2006.12.064.
- S. Yang, X. Li, Y. Li, Y. Wang, X. Jin and L. Qin, et al., Effect of Proton Transfer on Electrocatalytic Water Oxidation by Manganese Phosphates, Angew. Chem., Int. Ed., 2023, 62(1), e202215594, DOI:10.1002/anie.202215594 , PubMed PMID: 36342503.
- S. Yang, K. Yue, X. Liu, S. Li, H. Zheng and Y. Yan, et al., Electrocatalytic water oxidation with manganese phosphates, Nat. Commun., 2024, 15(1), 1410, DOI:10.1038/s41467-024-45705-1 , PubMed PMID: 38360868.
- J. Yang, T. Yu, X. Jiang, X. Zhang, J. Guo and Y. Chen, et al., Hydrated manganese hydrogen phosphate coated membrane with excellent anticrude oil-fouling property for separating crude oil from diverse wastewater, Surf. Coat. Technol., 2023, 454, DOI:10.1016/j.surfcoat.2022.129215.
- S. Munkaila, R. Dahal, M. Kokayi, T. Jackson and B. P. Bastakoti, Hollow Structured Transition Metal Phosphates and Their Applications, Chem. Rec., 2022, 22(8), e202200084, DOI:10.1002/tcr.202200084 , PubMed PMID: 35815949.
- A. Bouddouch, E. Amaterz, R. Haounati, Y. Naciri, A. Taoufyq and B. Bakiz, et al., Synthesis, characterization and luminescence properties of manganese phosphate Mn3(PO4)2, Mater. Today: Proc., 2020, 16–21, DOI:10.1016/j.matpr.2019.08.058.
- J. Cherusseri, S. A. Thomas, A. K. Pandey, M. A. Zaed, N. K. Farhana and R. Saidur, Rapid synthesis of cobalt manganese phosphate by microwave-assisted hydrothermal method and application as positrode material in supercapatteries, Sci. Rep., 2024, 14(1), 1–18, DOI:10.1038/s41598-024-77278-w , PubMed PMID: 39489744.
- S. H. Jhung, J. S. Chang, J. W. Yoon, J. M. Grenèche, G. Férey and A. K. Cheetham, Synthesis of transition-metal-incorporated nickel phosphate molecular sieves TMI-VSB-1, Chem. Mater., 2004, 16(26), 5552–5555, DOI:10.1021/cm049081e.
- J. Zhao, Q. Jing, T. Zhou, X. Zhang, W. Li and H. Pang, Controllable Synthesis of Manganese Organic Phosphate with Different Morphologies and Their Derivatives for Supercapacitors, Molecules, 2024, 29(17), 4186, DOI:10.3390/molecules29174186 , PubMed PMID: 39275034.
- Y. Zhang, Y. Liu, S. Fu, F. Guo and Y. Qian, Hydrothermally controlled growth of MnPO4·H2O single-crystal rods, Bull. Chem. Soc. Jpn., 2006, 79(2), 270–275, DOI:10.1246/bcsj.79.270.
- H. Uchiyama, I. Okumura, S. Inoue, M. Kato and M. Wakahara, Aqueous Synthesis of Manganese Phosphate Hydrate Crystals for Creating Inorganic Pigment Materials, Inorg. Chem., 2021, 60(19), 14779–14785, DOI:10.1021/acs.inorgchem.1c02008 , PubMed PMID: 34553908.
- M. A. G. Aranda and S. Bruque, Characterization of manganese(III) orthophosphate hydrate, Inorg. Chem., 2002, 29(7), 1334–1337, DOI:10.1021/IC00332A009.
- G. Herzberg and L. C. Bryce, Infrared and Raman Spectra of Polyatomic Molecules, J. Phys. Chem., 2002, 50(3), 288, DOI:10.1021/J150447A021.
- M. Tsuboi, Vibrational Spectra of Phosphite and Hypophosphite Anions, and the Characteristic Frequencies of PO3- and PO2- Groups, J. Am. Chem. Soc., 2002, 79(6), 1351–1354, DOI:10.1021/JA01563A026.
- Y. Huang, N. A. Chernova, Q. Yin, Q. Wang, N. F. Quackenbush and M. Leskes, et al., What Happens to LiMnPO4 upon Chemical Delithiation?, Inorg. Chem., 2016, 55(9), 4335–4343, DOI:10.1021/acs.inorgchem.6b00089.
- Y. Song, J. Jiang, J. Ma, S. Y. Pang, C. Luo and W. Qin, Oxidation of inorganic compounds by aqueous permanganate: Kinetics and initial electron transfer steps, Sep. Purif. Technol., 2017, 183, 350–357, DOI:10.1016/j.seppur.2017.04.015.
- Y. Zhu, G. Hasegawa, K. Kanamori and K. Nakanishi, Comprehensive studies on phosphoric acid treatment of porous titania toward titanium phosphate and pyrophosphate monoliths with pore hierarchy and a nanostructured pore surface, Inorg. Chem. Front., 2018, 5(6), 1397–1404, 10.1039/C8QI00146D.
- J. C. Munyemana, H. He, S. Ding, J. Yin, P. Xi and J. Xiao, Synthesis of manganese phosphate hybrid nanoflowers by collagen-templated biomineralization, RSC Adv., 2018, 8(5), 2708–2713, 10.1039/c7ra12628j.
- B. Boonchom, S. Youngme, S. Maensiri and C. Danvirutai, Nanocrystalline serrabrancaite (MnPO4·H2O) prepared by a simple precipitation route at low temperature, J. Alloys Compd., 2008, 454(1–2), 78–82, DOI:10.1016/J.JALLCOM.2006.12.064.
- J. Zhao, Q. Jing, T. Zhou, X. Zhang, W. Li and H. Pang, Controllable Synthesis of Manganese Organic Phosphate with Different Morphologies and Their Derivatives for Supercapacitors, Molecules, 2024, 29(17), 4186, DOI:10.3390/molecules29174186 , PubMed PMID: 39275034.
- H. Chevulamaddi and V. R. Kalagadda, Impressive electrochemical characteristics of freeze-dried nanosheet structured Mn2P2O7 for affordable electrochemical capacitor, Surf. Interfaces, 2024, 55, 105340, DOI:10.1016/J.SURFIN.2024.105340.
- H. Chevulamaddi and V. R. Kalagadda, Redox Mediator–assisted Electrochemical Behaviour of Mn2P2O7 Nanoclusters for Symmetric Supercapacitors, Fuel, 2025, 398, 135543, DOI:10.1016/J.FUEL.2025.135543.
- R. BoopathiRaja, S. Vadivel, M. Parthibavarman, S. Prabhu and R. Ramesh, Effect of polypyrrole incorporated sun flower like Mn2P2O7 with lab waste tissue paper derived activated carbon for asymmetric supercapacitor applications, Surf. Interfaces, 2021, 26, 101409, DOI:10.1016/J.SURFIN.2021.101409.
- E. A. Alabdullkarem and J. Khan, Mn2P2O7–polyaniline hybrid composite as a promising electrode material for advanced symmetric supercapacitors, RSC Adv., 2025, 15(30), 24831–24843, 10.1039/D5RA04149J.
| This journal is © The Royal Society of Chemistry 2026 |