 Open Access Article
Open Access ArticleBio-based antimicrobial nanocomposites derived from tannery waste for functional leather coating: circular valorization of chrome shaving dust
Sharmin Akter
Liza
a,
Md.
Abdulla-Al-Mamun
 *a,
Md.
Nazmul Islam
a,
Md. Rezaul Karim
Rana
b and
Md. Shariful
Islam
c
*a,
Md.
Nazmul Islam
a,
Md. Rezaul Karim
Rana
b and
Md. Shariful
Islam
c
aInstitute of Leather Engineering and Technology, University of Dhaka, 44-50 Hazaribagh, Dhaka-1209, Bangladesh. E-mail: mamun.ilet@du.ac.bd
bMolecular Biology Laboratory, Department of Biochemistry and Molecular Biology, Faculty of Biological Sciences, University of Dhaka, Dhaka 1000, Bangladesh
cDepartment of Veterinary and Animal Sciences, University of Rajshahi, Rajshahi-6205, Bangladesh
First published on 13th January 2026
Abstract
This study aimed to establish a waste valorization approach by developing bio-based antimicrobial nanocomposites and exploring their potential reuse within a circular economy framework. Protein was extracted from chrome-tanned shaving dust through eco-friendly microbial degradation using Bacillus thuringiensis SRL4A. Four types of metal–protein-based nanocomposites, namely protein–ZnO, Ag–protein, polyaniline (PANI)/protein–ZnO, and PANI/Ag–protein, were synthesized using the protein hydrolysate derived from tannery solid waste and incorporated into leather finishing formulations. Characterization techniques, including TKN, amino acid analysis, SDS-PAGE, UV-vis spectroscopy, FT-IR, XRD, zeta potential analysis, TGA, SEM, and EDX, were employed to determine the functional groups, crystallographic structure, thermal stability, morphology, and composition of the nanocomposites. TKN analysis confirmed that the protein content in the sample obtained from shaving dust was 73.08% and other analyses confirmed the successful incorporation of metal particles into the protein matrix. The particle sizes of the synthesized nanocomposites were 32 nm (protein–ZnO), 34 nm (Ag–protein), 44 nm (PANI/protein–ZnO), and 45 nm (PANI/Ag–protein). Cytotoxicity tests revealed that all nanocomposites were non-toxic to living cells. The minimum inhibitory concentration (MIC) values for protein–ZnO were 0.325 mg mL−1 against Gram-positive B. cereus and 1.25 mg mL−1 against Gram-negative E. coli. For Ag–protein, PANI/protein–ZnO, and PANI/Ag–protein, the MIC was 0.625 mg mL−1 against both B. cereus and E. coli. All nanocomposites exhibited antimicrobial activity, with protein–ZnO showing the highest zone of inhibition (ZOI = 20 mm) against B. cereus and Ag–protein showing the highest ZOI (22 mm) against E. coli. Leather samples coated with varying amounts of nanocomposites in different finishing formulations demonstrated effective antibacterial activity and met the standard requirements for finish film quality and mechanical performance. Notably, protein–ZnO-treated leather exhibited the highest ZOI (18 mm) against B. cereus, while Ag–protein-treated leather achieved the highest ZOI (14 mm) against E. coli. This work presents a sustainable approach to repurposing tannery waste into high-performance antimicrobial materials, with potential applications in the leather, textile, packaging, and biomedical industries, as well as the valorization of protein derived from tannery waste.
1. Introduction
In recent years, bio-nanocomposites have attracted considerable attention owing to their eco-friendly nature and diverse potential applications across various fields.1 These hybrid materials comprise both inorganic or organic nanoparticles (NPs) and bio-based polymers, with the nanoparticles typically exhibiting at least one dimension on the nanometre scale.2 Among these, metal–protein-based nanocomposites have emerged as promising candidates. Proteins possess several advantageous characteristics, such as biocompatibility, flexibility, abundant availability, biodegradability, and multiple reactive sites, often referred to as ‘cavities’, that make them ideal building blocks for high-performance nanocomposites.3During nanocomposite synthesis, proteins are commonly employed as capping agents that function as stabilizers, limiting nanoparticles' overgrowth and preventing their aggregation or coagulation. Capping ligands play a crucial role in maintaining the stability of the interface between nanoparticles and their synthesis medium.4 Due to their amphiphilic nature, capping agents impart functionality and enhance compatibility with other phases, facilitating improved integration within nanocomposite systems.5 Proteins utilize their polar head groups to coordinate with metal atoms, while their non-polar tails interact with the surrounding medium. This amphiphilic nature makes proteins highly effective as capping agents in the synthesis of nanocomposites. Metal–protein nanocomposites exhibit a versatile range of properties, including high mechanical strength,6 biodegradability, thermal stability, and light barrier capabilities.7,8 Additionally, they demonstrate vapor permeability, surface adhesion, and notable antifungal and antibacterial activities.9
Among the various promising applications of nanocomposites, antibacterial functionality stands out as one of the most compelling. Their ability to disrupt microbial activity through mechanisms such as physical interaction, oxidative stress induction, and controlled release of antimicrobial agents makes them powerful candidates for integration into medical devices, packaging materials, and environmental systems.10,11 The structural versatility and tunable surface properties of nanocomposites further enhance their efficacy in targeting a wide range of pathogenic microorganisms.12,13
Leather finishing represents a pivotal stage in the leather manufacturing process, wherein raw hides undergo surface treatments to acquire enhanced durability, aesthetic appeal, and functional performance. This phase is instrumental in imparting key properties such as hydrophobicity, colour fastness, rub resistance, gloss, and uniform texture to the final product.14 Conventional finishing formulations typically employ film-forming agents based on proteins, such as casein, or synthetic resins like polyurethanes and polyacrylates. Protein-based systems, particularly those utilizing casein, offer a natural appearance and thermostability, though they may exhibit limitations in water resistance and rub fastness.15 In contrast, resin-based formulations provide superior mechanical strength, abrasion resistance, and chemical stability, making them widely adopted in industrial leather finishing applications.16
The leather finishing process traditionally involves the use of environmentally detrimental substances, including volatile organic compounds (VOCs), non-biodegradable resins, and potentially hazardous cross-linking agents, all of which aim to enhance the physical and mechanical properties of leather.14 These chemical inputs contribute significantly to air pollution and pose serious risks to both environmental integrity and occupational health. In response to these challenges, various strategies have been proposed, most of which centre on the use of cross-linking agents to improve film-forming capabilities. However, to align with contemporary sustainability goals, it is imperative to further innovate leather finishing technologies by improving product performance while reducing reliance on harmful chemicals. This includes the advancement of bio-nanocomposite-based finishing systems that integrate highly compatible base and top coats. Despite the critical importance of this finishing phase, limited progress has been made in developing formulations that utilize proteins derived from industry-generated waste. Recent research has explored the synergistic application of such protein-based materials with nanotechnology, offering a promising pathway toward circular and eco-conscious leather manufacturing.14,17,18
Previous studies have reported the utilization of various biopolymers in nanocomposite formulations, including egg albumen,19,20 fish skin gelatin,21 fenugreek and mung bean proteins,22 corn silk extract,23 chitosan,24 soy protein,25 and whey protein.26 All these systems, metal atoms such as aluminum, lead, silver, gold, iron, cobalt, zinc, cadmium, titanium, and copper, have been incorporated to enhance functional properties.27 More recently, polyaniline (PANI)-functionalized metal nanocomposites have gained attention due to PANI's high electrical conductivity, environmental stability, facile synthesis, and notable antibacterial and antifungal activities.20 The polymer exhibits multiple oxidation states, and its physicochemical properties, including color, conductivity, and antimicrobial efficacy, are influenced by its oxidation and protonation levels.
In parallel, to mitigate microbial contamination on leather surfaces resulting from prolonged human contact, several nanocomposite-based treatments have been developed to impart antifungal and antibacterial characteristics. Notable examples include Ag–NiO2 composites,28 polyacrylate–ZnO nanocomposites,29 TiO2–polyacrylate emulsions,30 and casein–ZnO hybrid systems.31
In this study, a protein hydrolysate derived from tannery shaving dust – a significant solid waste stream containing approximately 15–30% proteinaceous material – was employed as a capping agent for the synthesis of metal–protein nanocomposites.32 Tannery shaving dust predominantly comprises chromium-cross-linked collagen, a by-product of leather processing that is typically discarded through environmentally detrimental practices such as open burning or landfilling. These disposal methods contribute to soil and air pollution, raising serious ecological concerns.
To the best of our knowledge, this is the first report detailing the utilization of protein extracted from such waste for the fabrication of nanocomposites, including protein–ZnO, Ag–protein, PANI/protein–ZnO, and PANI/Ag–protein systems. The protein hydrolysate was obtained via microbial degradation, a bio-based approach that facilitates the breakdown of complex collagen structures into functional peptides suitable for nanomaterial synthesis. Zinc and silver were selected as the metal components due to their well-documented antibacterial efficacy and compatibility with protein matrices.
The resulting nanocomposites were subsequently integrated into leather coating formulations to evaluate their potential as sustainable and antimicrobial alternatives in tannery finishing processes. This approach not only valorizes industrial waste but also aligns with circular economy principles by transforming hazardous residues into value-added functional materials.
2. Materials and methods
2.1. Chemicals and materials
All reagents used in this study were of analytical grade, except for those employed in leather finishing, which were of industrial grade. Zinc sulfate (ZnSO4·7H2O), silver nitrate (AgNO3), sodium borohydride (NaBH4), ammonium persulfate ((NH4)2S2O8), ferric chloride (FeCl3), and ethanol (C2H5OH) were procured from Sigma-Aldrich and Merck Chemical Groups. Purified sodium hydroxide (NaOH) pellets, magnesium oxide (MgO), and hydrochloric acid (HCl, 37%) were obtained from Active Fine Chemicals Ltd, Dhaka, Bangladesh. Nutrient broth and Mueller–Hinton agar media were sourced from HiMedia Laboratories Pvt. Ltd, India. Leather finishing chemicals, including pigment, isopropyl alcohol (IPA), acrylic resin, polyurethane (PU), protein binder/lustre, compact binder, nitrocellulose (NC) lacquer, and silicon, were supplied by Stahl, Germany. The Institute of Leather Engineering and Technology, University of Dhaka, provided goat crust leather.2.2. Extraction of the protein hydrolysate from tannery shaving dust
Shaving dust collected from a tannery was dried at 100 °C for 30 min. Distilled water (10 mL g−1 of chrome shavings) and tryptone (12% w/w) were added, followed by sterilization via autoclaving. After cooling to 50–60 °C, a bacterial cell suspension of Bacillus thuringiensis SRL4A (PP802975) at 35% v/w was inoculated. The mixture was incubated for 100 h at 150 rpm in a rotary shaker. Post-incubation, the hydrolysate was centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 20 min, filtered, and treated with MgO and NaOH at 55 °C for 2 h to precipitate chromium as insoluble Cr(OH)3.33 Throughout this study, the resulting dechromed protein hydrolysate is referred to as “protein”. The protein was subjected to various characterization analyses, including determination of the final chromium content in the protein sample. The process and result of the analyses are reported in our previous paper.33
000 rpm for 20 min, filtered, and treated with MgO and NaOH at 55 °C for 2 h to precipitate chromium as insoluble Cr(OH)3.33 Throughout this study, the resulting dechromed protein hydrolysate is referred to as “protein”. The protein was subjected to various characterization analyses, including determination of the final chromium content in the protein sample. The process and result of the analyses are reported in our previous paper.33
2.3. Synthesis of nanocomposites
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) NaBH4 = 1
NaBH4 = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) was dissolved in 6 mL of DI water. While the AgNO3–protein hydrolysate solution was stirred vigorously, the NaBH4 solution was added dropwise. Upon the addition of NaBH4, the solution's colour changed from pale yellow to deep brown, accompanied by the formation of microbubbles on the surface. To ensure complete homogenization, the mixture was sonicated for 10 min, resulting in a brownish-red solution with minimal residual bubbling. The final product was collected by centrifugation, and the Ag–protein nanocomposite was obtained via freeze-drying of the precipitate (Fig. 1b).
2) was dissolved in 6 mL of DI water. While the AgNO3–protein hydrolysate solution was stirred vigorously, the NaBH4 solution was added dropwise. Upon the addition of NaBH4, the solution's colour changed from pale yellow to deep brown, accompanied by the formation of microbubbles on the surface. To ensure complete homogenization, the mixture was sonicated for 10 min, resulting in a brownish-red solution with minimal residual bubbling. The final product was collected by centrifugation, and the Ag–protein nanocomposite was obtained via freeze-drying of the precipitate (Fig. 1b).
2.4. Finishing leather with the nanocomposites
The finishing formulations applied to upper leather are detailed in Tables 1 and 2. Four leather specimens were sprayed twice with the seasonal coat formulations, first incorporating nanocomposites and subsequently without nanocomposites, using a spray gun. The coated specimens were dried in a heater at 40 °C and then pressed at 70 kg cm−2 at 80 °C. Following this, a single layer of top coat formulation was applied to each specimen using one coat (two cross sprays), dried again at 40 °C, and pressed at 80 kg cm−2 at 80 °C. An additional leather specimen was finished using a conventional formulation, also prepared according to Tables 1 and 2. All chemical quantities are expressed in parts by weight.| Ingredients | Conventional (1 layer) | Experimental (2 layers) |
|---|---|---|
| Pigment | 30 | — |
| IPA | 30 | 24 |
| Acrylic resin | 100 | — |
| PU | 60 | — |
| Protein binder/lustre | 100 | 476 |
| Compact binder | 30 | — |
| Water | 650 | 495 |
| Nanocomposites | — | 5 |
| Ingredients | Conventional (1 layer) | Experimental (1 layer) |
|---|---|---|
| NC lacquer | 645 | 645 |
| Silicon | 30 | 30 |
| Water | 325 | 325 |
2.5. Characterization techniques
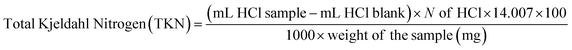 | (1) |
Amino acid profiling of the samples was conducted in accordance with AOAC Official Method 982.30, designated for the analysis of amino acids in foods and feeds.36 The analysis was performed using a Sykam Amino Acid Analyzer (Model S433; Sykam GmbH, Germany), which operates on ion-exchange chromatography coupled with post-column ninhydrin derivatization for quantification.
For sample preparation, 0.20–1.00 g of a finely powdered sample was hydrolyzed with 6 M hydrochloric acid (HCl) containing 0.1% (w/v) phenol to prevent oxidative degradation of tyrosine. The acid hydrolysis was carried out in sealed glass ampoules at 110 °C for 24 hours. Upon completion of hydrolysis, the reaction mixture was cooled to room temperature, and the pH was carefully adjusted to the range of 2.9–3.1 using a sodium hydroxide (NaOH) solution to ensure compatibility with the chromatographic system.
The resulting hydrolysate was filtered through a 0.25 µm syringe filter to remove particulates and transferred to the autosampler vials. Quantification of individual amino acids was achieved by comparing the retention times and peak areas of the sample chromatograms with those of authenticated amino acid standards analyzed under identical conditions.
Fourier-transform infrared (FTIR) spectroscopy was employed to identify the presence of specific functional groups and to monitor alterations in the secondary structure of the samples. The spectra were recorded using a Bruker ALPHA II FT-IR spectrometer (Bruker Optik GmbH, Germany) within the wavenumber range of 4000–500 cm−1, at a spectral resolution of 4 cm−1. This technique enabled the detection of characteristic vibrational modes associated with molecular bonds, thereby facilitating a detailed understanding of the chemical interactions and structural modifications occurring within the material matrix.
Surface charge properties were assessed through zeta potential (ζ-potential) measurements using a zeta potential instrument (Malvern Panalytical Ltd., UK). Prior to analysis, the samples were sonicated to ensure uniform dispersion and subsequently injected into a 100 µL measurement cell. The instrument determined the electrophoretic mobility of the particles, which was then used to calculate the ζ-potential, offering valuable information regarding colloidal stability and surface interactions.
The thermal stability and decomposition behavior of the samples were evaluated using thermogravimetric analysis (TGA), conducted on a TGA 8000 thermogravimetric analyzer (PerkinElmer Inc., USA). Approximately 5 mg of each sample was placed in a platinum crucible mounted on the microbalance pan. The samples were heated from 50 °C to 700 °C at a constant rate of 10 °C min−1 under a nitrogen atmosphere maintained at a flow rate of 20 mL min−1. The resulting thermograms provided quantitative data on weight loss events, enabling interpretation of thermal degradation stages and compositional stability.
In conjunction with FESEM analysis, elemental composition and distribution were assessed using an integrated Energy-Dispersive X-ray (EDX) spectroscopy system. This technique enabled qualitative and semi-quantitative identification of the constituent elements present in the sample matrix, thereby complementing the morphological observations with compositional insights.
2.6. Antibacterial evaluation of the nanocomposite and nanocomposite-coated leather
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and 0.2% gentamycin to prevent microbial contamination.
1) and 0.2% gentamycin to prevent microbial contamination.
A cell suspension containing 1.5 × 104 cells per 100 µL was seeded into sterile 96-well microplates and incubated at 37 °C in a humidified atmosphere containing 5% CO2 to allow for cell attachment and stabilization. After 24 hours of incubation, 25 µL of the autoclaved nanocomposite sample was introduced into each well. The treated cells were further incubated for 48 hours under the same conditions.
Cytotoxic effects were evaluated using an inverted light microscope by observing morphological changes, including cell shrinkage, detachment, and loss of membrane integrity. All treatments were performed in duplicate to ensure reproducibility and statistical reliability.
For the agar well diffusion assay, Mueller–Hinton agar plates were uniformly inoculated with bacterial suspensions standardized to 1 × 106 CFU mL−1 (0.5 McFarland standard) using sterile cotton swabs to ensure even distribution across the agar surface. Wells of 6 mm diameter were aseptically punched into the agar using a sterile cork borer. Each well was carefully loaded with 50 µL of the nanocomposite sample, which had been previously sterilized under ultraviolet (UV) light for 15 minutes to eliminate potential contaminants. Care was taken to dispense the sample slowly to prevent overflow and ensure consistent diffusion. The inoculated plates were incubated at 37 °C for 24 hours under aerobic conditions. Following incubation, the diameter of the zone of inhibition (ZOI) was measured in millimeters using a sterile ruler, and the results were recorded to quantify antimicrobial activity.
In parallel, the antimicrobial performance of nanocomposite-coated leather was evaluated using a modified disk diffusion protocol. A leather specimen (5 mm diameter) was sterilized under UV light for 15 minutes and placed directly onto the inoculated agar surface, ensuring that the finished film side was in contact with the medium. The plates were incubated under identical conditions (37 °C for 24 hours), and the ZOI surrounding the leather samples was measured to determine the extent of bacterial inhibition.
2.7. Mechanical tests of coated leather
Prior to testing, all leather specimens were conditioned under standardized laboratory conditions at a temperature of 23 ± 2 °C and a relative humidity of 65 ± 3%, in accordance with ISO 2419 guidelines, for 48 hours. Subsequent performance assessments were conducted using established SATRA methodologies to evaluate mechanical durability, thermal resistance, water vapor permeability, and adhesive bond strength. The SATRA TM-08 protocol was employed to assess surface damage and colour transfer under both dry and wet abrasion conditions. In the dry rub fastness test, a cotton felt pad was rotated over the leather surface under a 2.5 kg load, simulating mechanical wear. For wet rub fastness, the same procedure was performed without any applied load, with the felt pre-moistened to replicate exposure to moisture. Post-abrasion, the extent of colour change and staining on both the leather sample and the cotton felt was evaluated using the ISO 105-A02 grey scale for colour change and ISO 105-A03 for staining, providing a semi-quantitative measure of fastness performance. Thermal resistance was determined using the SATRA TM-49 method, which evaluates the visible effects of brief contact between the leather surface and a heated metal block. The test identifies changes such as discoloration, shrinkage, burning, melting, or smearing. Each specimen was graded using the ISO 105-A02 grey scale, allowing for standardized comparison of thermal degradation across samples. Water vapor transmission characteristics were measured according to SATRA TM-172, a method designed to assess breathability in leathers and textiles intended for footwear and apparel applications. Each leather specimen was affixed to a test pot containing desiccant-grade silica gel and exposed to controlled air conditions for 16 hours. The water vapor permeability (WVP) was calculated based on the mass change of the silica gel using the following equation: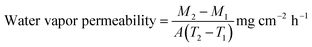 | (2) |
Bond strength was evaluated through a custom adhesion test. Epoxy resin was uniformly applied to a pre-conditioned leather strip, which was then bonded to a rigid plastic plate. The assembly was air-dried for 30 minutes, followed by compression under a 2 kg load for 24 hours to ensure uniform adhesion. The bonded specimen was subsequently subjected to incremental loading until detachment occurred. The maximum load at failure was recorded, providing a quantitative measure of adhesive integrity between the leather and polymer substrate.
3. Results and discussion
3.1. Characteristic analysis of proteins and composites
The amino acid composition of collagen derived from the shaving dust sample is presented in Table S1 of the supplementary information. Biochemically, collagen is characterized by a distinctive repeating tripeptide sequence (Gly–X–Y), in which glycine occupies every third residue. Consequently, glycine constitutes approximately one-third of the total amino acid content in collagen.39 In alignment with this structural motif, our analysis revealed glycine as the most abundant amino acid, with a concentration of 134.83 mg g−1. The next most prevalent residues were proline (PRO) and its hydroxylated derivative hydroxyproline (HYP), which frequently occupy the X and Y positions in the collagen sequence. These amino acids are critical biochemical markers for estimating total collagen content due to their structural role in stabilizing the triple helix. In our study, the combined concentration of proline and hydroxyproline was 97.13 mg g−1, reflecting the collagen-rich nature of the sample and corroborating previously reported values. In addition to these dominant residues, glutamic acid and alanine were also detected in appreciable quantities, with concentrations of approximately 60 mg g−1 and 57 mg g−1, respectively. These findings further support the characteristic amino acid profile of collagen and are consistent with the established literature. Moreover, the final chromium content in the dechromed protein hydrolysate was also found to be 1.40 ± 0.37 ppm.
Fig. S1 in the supporting information presents the UV-vis absorption spectra of all synthesized samples. The protein–ZnO nanocomposite exhibited a distinct SPR absorption peak at approximately 355 nm, corresponding to a band gap of 3.49 eV. This value exceeds the typical band gap of bulk ZnO (∼3.3 eV), indicating a quantum confinement effect and confirming the nanoscale dimensions of the ZnO particles.42 The protein solution alone showed a characteristic absorption peak at 280 nm, attributed to aromatic amino acid residues. This peak was also observed for the protein–ZnO nanocomposite, suggesting successful incorporation of protein within the composite matrix.
An asymmetric tail on the higher wavelength side of the SPR peak was noted, likely due to light scattering effects caused by the inhomogeneous distribution of nanocrystalline ZnO particles, which are insoluble in aqueous media.43 Such scattering phenomena are more pronounced in larger metallic nanoparticles, such as silver, which exhibit broadened and red-shifted SPR peaks due to enhanced scattering and aggregation tendencies.
In the silver–protein nanocomposite, a prominent SPR band was observed at 405 nm, characteristic of silver nanoparticles. The absence of peaks in the 335–560 nm range suggests that no significant aggregation occurred during synthesis.44 The sharp and intense nature of the SPR peak further implies a high yield of well-dispersed nanoparticles.
The PANI/Ag–protein nanocomposite displayed a strong absorption peak at 411 nm, along with a broad rising absorption near 800 nm. The peak at 411 nm is attributed to the SPR of silver and ZnO nanoparticles embedded within the polyaniline (PANI) matrix and is overlapped by the polaronic transition of PANI, which occurs at a similar wavelength.45 The broad absorption at higher wavelengths, accompanied by a free carrier tail, confirms the presence of the conducting emeraldine salt (ES) phase of PANI,46 indicating successful polymerization and integration of the nanocomposite components.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations of peptide linkages, contributing approximately 80% of the signal. In the FTIR spectrum of pure protein hydrolysate (Fig. 2a), a broad absorption band at 3290 cm−1 was observed, corresponding to the O–H stretching vibrations. Additionally, characteristic peaks at 1631 cm−1, 1548 cm−1, and 1240 cm−1 were assigned to amide I, amide II, and amide III bands, respectively. Peaks at 1448 cm−1 and 1082 cm−1 were attributed to C–H deformation and C–O stretching, consistent with the functional groups present in collagen.48
O stretching vibrations of peptide linkages, contributing approximately 80% of the signal. In the FTIR spectrum of pure protein hydrolysate (Fig. 2a), a broad absorption band at 3290 cm−1 was observed, corresponding to the O–H stretching vibrations. Additionally, characteristic peaks at 1631 cm−1, 1548 cm−1, and 1240 cm−1 were assigned to amide I, amide II, and amide III bands, respectively. Peaks at 1448 cm−1 and 1082 cm−1 were attributed to C–H deformation and C–O stretching, consistent with the functional groups present in collagen.48
 | ||
| Fig. 2 FT-IR spectra of (a) protein, (b) protein–ZnO, (c) Ag–protein, (d) PANI/protein–ZnO, and (e) PANI/Ag–protein. | ||
The FTIR spectra of protein–ZnO and Ag–protein nanocomposites (Fig. 2b and c) showed absorption bands at positions nearly identical to those of the pure protein hydrolysate, indicating the retention of protein structural motifs. Notably, the intensification and broadening of these bands suggest the formation of a composite, likely due to protein deprotonation during nanoparticle incorporation.49 Key absorption bands included 3290 cm−1 (N–H stretching), 1631 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching from –CO–NH– groups), and 576 cm−1 and 1082 cm−1, which correspond to metal-oxide stretching vibrations, consistent with literature reports.50
O stretching from –CO–NH– groups), and 576 cm−1 and 1082 cm−1, which correspond to metal-oxide stretching vibrations, consistent with literature reports.50
In the Ag–protein nanocomposite, a noticeable change in the intensity of the amide I band at 1631 cm−1 was observed, along with modifications in other vibrational bands compared to the pure protein hydrolysate. These spectral changes may be attributed to the formation of polypeptides and amino acids during hydrolysis, which act as stabilizing agents by coating the silver nanoparticles through interactions with their functional groups.51,52
The FTIR spectra of PANI/protein–ZnO and PANI/Ag–protein nanocomposites (Fig. 2d and e) revealed characteristic bands at 3292, 1560, 1431, 1300, 1124, 806, and 661 cm−1, resembling those of the pure protein hydrolysate, thereby confirming composite formation. The band at 3292 cm−1 corresponds to N-H stretching vibrations (both symmetric and asymmetric) of polyaniline (PANI). Peaks at 1560 cm−1 were assigned to quinoid and benzenoid ring stretching modes, while the band near 1240 cm−1 is associated with the quinic unit, indicative of PANI protonation. Distinctive peaks of pure PANI at 1286 cm−1 and 1478 cm−1 correspond to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of benzenoid and quinonoid rings, respectively. Additionally, an out-of-plane C–H bending vibration at 806 cm−1 was observed, characteristic of p-substituted benzene rings, and a C–C stretching peak at 1124 cm−1 was also detected. These findings are consistent with previous reports,53,54 further validating the successful incorporation of PANI and protein into the nanocomposite matrix.
C stretching vibrations of benzenoid and quinonoid rings, respectively. Additionally, an out-of-plane C–H bending vibration at 806 cm−1 was observed, characteristic of p-substituted benzene rings, and a C–C stretching peak at 1124 cm−1 was also detected. These findings are consistent with previous reports,53,54 further validating the successful incorporation of PANI and protein into the nanocomposite matrix.
 | ||
| Fig. 3 XRD patterns of prepared (a) protein–ZnO, (b) Ag–protein, (c) PANI/protein–ZnO and (d) PANI/Ag–protein nanocomposites. | ||
Fig. 3c and d present the XRD patterns of PANI/protein–ZnO and PANI/Ag–protein nanocomposites, respectively. Both spectra exhibit a semi-crystalline nature, characterized by two broad peaks at low diffraction angles, centered around 20° and 26°, corresponding to the (020) and (200) planes. These reflections are typically associated with periodicity parallel and perpendicular to the polymer backbone, and their sharpness and intensity reflect the degree of chain alignment and crystallite population within the polymer matrix.58,59 In the present study, the lack of sharpness in these peaks indicates poor orientation of PANI chains, likely due to the incorporation of inorganic nanoparticles and protein components.
In the PANI/Ag–protein nanocomposite, additional peaks at 37° and 45° confirm the presence of silver nanoparticles, and their retention within the polymer matrix suggests successful incorporation and stabilization of silver within the PANI framework. Conversely, the XRD pattern of PANI/protein–ZnO revealed a reduction in ZnO crystallinity compared to the pure ZnO phase, indicating that the amorphous nature of PANI may inhibit ZnO crystal growth.60 Nonetheless, characteristic ZnO diffraction peaks were still discernible at 30°, 32°, and 35°, confirming the presence of ZnO nanoparticles within the composite. The slight displacement of ZnO peaks in the ternary PANI/protein–ZnO nanocomposite suggests strong interfacial interactions between ZnO and the PANI matrix. Moreover, several weaker ZnO reflections were absent, likely due to the amorphous influence of polyaniline, which can suppress the crystallinity of embedded inorganic phases.61 These findings collectively affirm the formation of hybrid nanocomposites with distinct structural characteristics governed by the interplay between organic and inorganic components.
The initial weight loss observed across all samples occurred below 150 °C and is attributed to the evaporation of residual moisture and loosely bound water molecules within the matrix. The second decomposition stage, typically occurring between 200 and 350 °C, corresponds to the degradation of peptide linkages within the protein backbone. In the case of PANI/protein–ZnO and PANI/Ag–protein composites, this stage also encompasses the elimination of the acid dopant (HCl) and low-molecular-weight oligomers of aniline from the polyaniline (PANI) backbone, as previously reported.62,63 Additionally, the decomposition of protein-derived peptide bonds may contribute to this phase.
The third major weight loss, occurring at higher temperatures (above 400 °C), is primarily associated with the breakdown of the polymeric matrix and the thermal degradation of carbonaceous residues. This stage also involves the decomposition of inorganic constituents such as ZnO and Ag nanoparticles, as well as other thermally labile compounds embedded within the matrix.64,65 Furthermore, the release of structural water and the cleavage of hydroxyl groups initially bound to the PANI matrix may contribute to the final degradation phase.
Among all the tested samples, the protein–ZnO composite exhibited the highest thermal stability, followed sequentially by Ag–protein, pure protein, PANI/protein–ZnO, and Ag–protein–PANI. This trend suggests that the incorporation of ZnO nanoparticles enhances the thermal resistance of the protein matrix more effectively than Ag nanoparticles or PANI-based modifications.
 | ||
| Fig. 5 SEM analysis of (a) protein–ZnO (a1 and a2), (b) Ag–protein (b1 and b2), (c) PANI/protein–ZnO (c1 and c2) and (d) PANI/Ag–protein (d1 and d2). | ||
In Fig. 5b1, the Ag–protein nanocomposite displays small, spherical silver nanoparticles (AgNPs) uniformly dispersed within the protein matrix, indicating successful encapsulation and stabilization. Fig. 5c1 depicts the integration of ZnO nanoparticles within the rod-like structure of polyaniline (PANI), forming the PANI/protein–ZnO composite. The FESEM image of PANI/Ag–protein (Fig. 5d1) reveals that the AgNPs, exhibiting a somewhat spherical morphology, are homogeneously dispersed throughout the PANI polymer matrix. Additionally, agglomerated fibrous structures are evident on the surfaces of both PANI/protein–ZnO and PANI/Ag–protein composites, as shown in the supplementary figures (Fig. S2a and b), indicating partial clustering of polymer chains and embedded nanoparticles.
The particle size distribution for each nanocomposite was analyzed by fitting the histogram data to a log-normal distribution function, as illustrated in Fig. 5(a2–d2). The calculated average particle sizes were found to be 32 nm for protein–ZnO, 34 nm for Ag–protein, 44 nm for PANI/protein–ZnO, and 45 nm for PANI/Ag–protein, respectively. These values reflect the influence of polymeric encapsulation and nanoparticle loading on particle growth and dispersion.
To further confirm the elemental composition and assess the chemical purity of the nanocomposites, energy dispersive X-ray (EDX) spectroscopy analysis was performed (Fig. S3). The EDX spectra verified the presence of key elements expected in the formulations. Signals corresponding to carbon, nitrogen, and oxygen were consistent with the protein matrix, while the detection of silver and zinc confirmed the successful incorporation of AgNPs and ZnO nanoparticles. The elemental profiles obtained from EDX analysis are in agreement with the theoretical composition of the respective nanocomposites, thereby validating the synthesis approach and material integrity.
Fig. 6 illustrates the zeta potential profiles of the synthesized nanocomposites across varying pH conditions. The protein–ZnO nanocomposite exhibited zeta potential values ranging from +4.1 mV to 13.6 mV, with the highest value observed at pH 8. The Ag–protein nanocomposite demonstrated a broader range, from +6 mV to −31.7 mV at pH 10, indicating enhanced colloidal stability under alkaline conditions. Similarly, the PANI/protein–ZnO and PANI/Ag–protein composites showed zeta potential ranges of +5 mV to −27 mV and +5 mV to −27.6 mV, respectively. These variations suggest that the surface charge of the nanocomposites is highly pH-dependent, with near-neutral values under acidic and neutral conditions and increasingly negative values under basic conditions.
The predominance of negative zeta potential values across the nanocomposites implies improved colloidal stability, reduced aggregation tendency, and enhanced biocompatibility. Such characteristics are particularly advantageous for biomedical applications, including bone tissue engineering, where negatively charged surfaces have been shown to promote cell viability and integration.69 Furthermore, the tunable surface charge behavior of these nanocomposites underscores their versatility in diverse physiological environments, supporting their potential use in targeted delivery systems and implantable biomaterials.
3.2. Cytotoxicity evaluation of composites
The cytotoxicity profiles of the synthesized nanocomposites were evaluated using a standard cell viability assay, and the results are depicted in Fig. 7. Both protein–ZnO (Fig. 7b) and Ag–protein (Fig. 7c) composites demonstrated excellent biocompatibility, with cell viability consistently exceeding 95% after 24 and 48 hours of incubation. These findings suggest that the incorporation of ZnO and Ag nanoparticles within the protein matrix does not elicit any significant cytotoxic response under the tested conditions, thereby affirming their suitability for biomedical and packaging applications. In contrast, the PANI-functionalized composites, namely PANI/protein–ZnO (Fig. 7d) and PANI/Ag–protein (Fig. 7e), exhibited moderately reduced cell viability, with approximately 80% of cells remaining viable at both time points. Although this level of viability still falls within an acceptable range for certain applications, the slight decrease may be attributed to the intrinsic oxidative properties or surface charge characteristics of polyaniline, which could influence cellular interactions. Further investigation into the dose-dependent effects and long-term biocompatibility of PANI-containing systems is warranted to fully elucidate their safety profile. | ||
| Fig. 7 In vitro cell cytotoxicity evaluation of (a) control, (b) protein–ZnO, (c) Ag–protein, (d) PANI/protein–ZnO, and (e) PANI/Ag–protein. | ||
Fig. S8 illustrates the cytotoxicity assessment of the synthesized protein-based composites by determining their median lethal dose (LD50) values and corresponding cell survival viability. The LD50 of the protein–ZnO composite was found to exceed 1 mg mL−1, with a survival viability of 72.07%, suggesting relatively low toxicity at the tested concentration. In comparison, the Ag–protein composite exhibited an LD50 of 1 mg mL−1 with a viability of 72.87%, while the PANI/protein–ZnO and PANI/Ag–protein composites demonstrated lower LD50 values of 0.76 mg mL−1 and 0.507 mg mL−1, with survival viabilities of 40.05% and 34.79%, respectively. The native protein control showed an LD50 of 1 mg mL−1 and a viability of 69.83%. Notably, except for the PANI/Ag–protein composite, all tested materials maintained cell viability above 40% at their respective LD50 concentrations. This observation suggests that most composites do not exhibit pronounced cytotoxicity at the limit concentration, whereas the PANI/Ag–protein composite demonstrates comparatively higher toxicity, potentially due to synergistic effects between polyaniline and silver nanoparticles.
3.3. Minimum inhibitory concentration (MIC) assessment of nanocomposites
The minimum inhibitory concentration (MIC) is defined as the lowest concentration of an antimicrobial agent required to inhibit the visible growth of microorganisms under in vitro conditions. In this study, the MIC values of the synthesized nanocomposites were evaluated against two bacterial strains: Bacillus cereus (Gram-positive) and Escherichia coli (Gram-negative), as presented in Tables 3 and 4, respectively.| Dilution of nanocomposites | 5 mg mL−1 0.5% | 2.5 mg mL−1 0.25% | 1.25 mg mL−1 0.12% | 0.625 mg mL−1 0.06% | 0.325 mg mL−1 0.03% | 0.156 mg mL−1 0.01% |
|---|---|---|---|---|---|---|
| Positive (+): turbidity indicating growth; negative (−): no turbidity indicating the absence of growth. | ||||||
| Protein–ZnO | − | − | − | − | − | + |
| Ag–protein | − | − | − | − | + | + |
| PANI/protein–ZnO | − | − | − | − | + | + |
| PANI/Ag–protein | − | − | − | − | + | + |
| Dilution of nanocomposites | 5 mg mL−1 0.5% | 2.5 mg mL−1 0.25% | 1.25 mg mL−1 0.12% | 0.625 mg mL−1 0.06% | 0.325 mg mL−1 0.03% | 0.156 mg mL−1 0.01% |
|---|---|---|---|---|---|---|
| Positive (+): turbidity indicating growth; negative (−): no turbidity indicating the absence of growth. | ||||||
| Protein–ZnO | − | − | − | + | + | + |
| Ag–protein | − | − | − | − | + | + |
| PANI/protein–ZnO | − | − | − | − | + | + |
| PANI/Ag–protein | − | − | − | − | + | + |
Following incubation, the turbidity of each test tube was visually compared with two controls: one containing nutrient broth without bacterial inoculation, and another serving as the untreated bacterial control. Any turbidity attributable to the nanocomposite itself was disregarded to ensure accurate interpretation. The MIC was determined as the concentration at which no visible bacterial growth was observed, indicating complete inhibition of microbial proliferation.
The protein–ZnO nanocomposite exhibited MIC values of 0.325 mg mL−1 against B. cereus and 1.25 mg mL−1 against E. coli, demonstrating higher efficacy against the Gram-positive strain. In contrast, the Ag–protein, PANI/protein–ZnO, and PANI/Ag–protein composites each exhibited an MIC value of 0.625 mg mL−1 against both bacterial strains, indicating broad-spectrum antimicrobial activity. These findings are consistent with previous reports on the antimicrobial efficacy of silver nanoparticles, including their inhibitory effects against Staphylococcus aureus.37
The observed differences in MIC values may be attributed to variations in bacterial cell wall structure, nanoparticle composition, and the synergistic effects of the protein and polymer matrices. The enhanced activity of protein–ZnO against B. cereus highlights its potential as a selective antimicrobial agent, while the uniform MIC values of the other composites suggest their suitability for applications requiring broad-spectrum antibacterial coverage.
3.4. Finishing of leather with the nanocomposites
The synthesized nanocomposites were incorporated into the finishing formulation of leather, as illustrated in Fig. 8. Owing to their proteinaceous matrix, the composites functioned effectively as both fillers and binders, contributing to the structural integrity and adhesion of the finishing layer. Additionally, the presence of metal oxides such as ZnO and Ag endowed the formulation with antimicrobial properties, thereby enhancing the hygienic quality of the treated leather surface. Beyond their functional roles, the nanocomposites also imparted subtle coloration to the leather. Notably, the polyaniline (PANI)-based nanocomposites introduced two distinct shades of green, as evident in Fig. 8d and 8e. This chromatic contribution is attributed to the intrinsic color of PANI and its interaction with the protein–metal matrix, providing both aesthetic enhancement and functional benefits. | ||
| Fig. 8 Nanocomposite coated leather samples: (a) conventional (no nanocomposite was used), (b) protein–ZnO, (c) Ag–protein, (d) PANI/Ag–protein, and (e) PANI/protein–ZnO. | ||
Furthermore, the treated leather samples demonstrated improved surface glazing and enhanced mechanical properties, including increased flexibility and abrasion resistance. These improvements suggest that the nanocomposite-based finishing formulations not only provide antimicrobial protection and visual appeal but also contribute to the durability and performance of the final leather product.
3.5. Antimicrobial activities of nanocomposites and coated finished leather
The antimicrobial efficacy of the synthesized nanocomposites was assessed using the agar well diffusion method in Petri dishes, with a protein solution serving as the control. The tests were conducted against two bacterial strains: Bacillus cereus (a Gram-positive bacterium) and Escherichia coli (a Gram-negative bacterium), with incubation periods ranging from 18 to 24 hours. The results are visually represented in Fig. 9, and the corresponding zones of inhibition are quantitatively reported in Table 5. | ||
| Fig. 9 Antimicrobial activity of protein–ZnO (S-1), Ag–protein (S-2), PANI/protein–ZnO (S-3), PANI/Ag–protein (S-4), and protein (S-5) against (a) B. cereus and (b) E. coli. | ||
| Bacteria | Protein–ZnO (S-1) | Ag–protein (S-2) | PANI/protein–ZnO (S-3) | PANI/Ag-Protein (S-4) | Control (Protein) (S-5) |
|---|---|---|---|---|---|
| B. cereus zone of inhibition (mm) | 20 ± 0.70 | 18 ± 1 | 15 ± 1 | 13 ± 1 | 0 |
| E. coli zone of inhibition (mm) | 18 ± 1 | 22 ± 0.50 | 12 ± 0.50 | 12 ± 1 | 0 |
Zone of inhibition (ZOI) values for the four nanocomposite samples were compared with those of the positive (commercial antibiotic) and negative (protein, S-5) controls to evaluate antimicrobial activity, as shown in Fig. S4 and Table S7. For each, a two-tailed two-sample t-test was performed using Excel's T.TEST function. A significance threshold of p < 0.05 was used. All four samples showed statistically significantly larger ZOI compared to the negative control (p < 0.05), indicating effective antimicrobial activity for both B. cereus and E. coli bacteria. When compared to the positive control, all the samples showed a significant difference (p < 0.05), suggesting a slightly weaker efficacy compared to the commercial antibiotic for B. cereus and E. coli. However, the Ag–protein sample had a p-value greater than 0.05 for E. coli, which is statistically not significant, indicating that it has a similar effect to commercial antibiotics.
The antimicrobial mechanisms of the constituent nanoparticles are well-documented. Zinc oxide (ZnO) nanoparticles exert bactericidal effects by disrupting bacterial cell membranes, reducing cell surface hydrophobicity, and inhibiting the transcription of genes responsible for oxidative stress resistance. ZnO also enhances intracellular bacterial elimination by stimulating the production of reactive oxygen species (ROS), which interact with vital cellular components such as proteins, lipids, and DNA, ultimately inducing apoptosis.70
Silver nanoparticles (AgNPs), on the other hand, owe their antimicrobial properties primarily to the release of silver ions and their high surface area-to-volume ratio.71 These ions attach to essential bacterial structures, particularly thiol (–SH) groups, thereby compromising cellular integrity.72 Additionally, AgNPs generate ROS and free radicals that damage the bacterial cell wall and interfere with respiratory enzymes, leading to impaired DNA replication and cell death.73
Polyaniline (PANI), a conductive polymer, also contributes to antimicrobial activity, which is influenced by its molecular weight. PANI synthesized via chemical oxidative polymerization using ammonium persulfate typically has a molecular weight below 100 kDa,74 enabling it to penetrate bacterial cells and interact with intracellular biomolecules. The NH3+ and –C6H5 moieties of PANI bind to bacterial membranes through electrostatic and hydrophobic interactions, respectively. These interactions disrupt membrane integrity, induce cell lysis, and facilitate ROS-mediated damage to proteins and lipids.
As shown in Fig. 9, the protein control (S-5) exhibited negligible antibacterial activity, confirming its role as a stabilizing agent rather than an active antimicrobial component. However, all four nanocomposite formulations demonstrated measurable zones of inhibition against both bacterial strains. The protein matrix played a crucial role in stabilizing the nanoparticles, preventing aggregation and potentially enhancing their interaction with microbial cells by modifying surface characteristics.
Among the tested formulations, protein–ZnO and Ag–protein nanocomposites exhibited superior antimicrobial activity against both B. cereus and E. coli, indicating effective nanoparticle stabilization and bioavailability. In contrast, the PANI-containing composites PANI/protein–ZnO and PANI/Ag–protein showed comparatively reduced antimicrobial efficacy, possibly due to altered nanoparticle surface properties that may hinder ROS generation or ion release.
Interestingly, both PANI-based composites demonstrated stronger antibacterial effects against B. cereus, with zones of inhibition of 15 mm and 13 mm, respectively. Their efficacy against E. coli was slightly diminished, with inhibition zones of 12 mm. This differential activity may reflect the inherent structural differences between Gram-positive and Gram-negative bacteria, as well as the complex interactions between PANI and the embedded nanoparticles.
The antibacterial efficacy of leather samples coated with various nanocomposite formulations was evaluated against Bacillus cereus (a Gram-positive bacterium) and Escherichia coli (a Gram-negative bacterium), as illustrated in Fig. 10 and Fig. S9 and summarized in Table 6. The tested samples included: S-1 (protein–ZnO), S-2 (Ag–protein), S-3 (PANI/protein–ZnO), S-4 (PANI/Ag–protein), and S-5 (conventional aniline finish).
| Bacteria | Protein–ZnO (S-1) | Ag–protein (S-2) | PANI/protein–ZnO (S-3) | PANI/Ag–protein (S-4) | Control (S-5) | |
|---|---|---|---|---|---|---|
| *Here, (−) indicates that no zone of inhibition was obtained for the sample control (S-5). | ||||||
| B. cereus (zone of inhibition (mm)) | Abrasion/rubbed leather | 18 ± 2 | 20 ± 2 | 16 ± 2 | 13 ± 1 | − |
| 8 ± 0.70 | 10 ± 0.50 | 6 ± 0.40 | 4 ± 0.80 | − | ||
| E. coli (zone of inhibition (mm)) | Abrasion/rubbed leather | 12 ± 2 | 14 ± 1 | 12 ± 0.5 | 10 ± 1 | − |
| 10 ± 1 | 12 ± 1 | 8 ± 0.80 | 7 ± 0.60 | − | ||
The control sample (S-5), coated with a conventional aniline-based finish, exhibited no observable antibacterial activity against either bacterial strain, confirming the absence of inherent antimicrobial properties in traditional leather finishing agents. In contrast, all nanocomposite-treated samples demonstrated measurable zones of inhibition, indicating effective antibacterial performance.
Among the tested formulations, S-2 (Ag–protein) showed the highest zone of inhibition against B. cereus, followed by S-1 (protein–ZnO), suggesting that silver-based nanocomposites possess superior bactericidal properties against Gram-positive bacteria. The PANI-incorporated samples (S-3 and S-4) exhibited comparatively lower antibacterial efficacy, consistent with previous observations regarding the interaction of PANI with nanoparticles and its potential influence on antimicrobial mechanisms.
The Gram-negative strain E. coli exhibited greater resistance to the nanocomposite treatments than B. cereus. This can be attributed to the structural complexity of the E. coli cell envelope, which comprises a thin peptidoglycan layer surrounded by an outer membrane rich in lipoproteins, lipopolysaccharides, and phospholipids. This multilayered barrier reduces permeability and limits the penetration of reactive oxygen species (ROS) and metal ions, thereby diminishing the antimicrobial efficacy of surface treatments.75
Overall, the results indicate that nanocomposite-coated leather samples are more effective against Gram-positive bacteria, with Ag–protein and protein–ZnO formulations showing the most promising antibacterial performance. These findings support the potential applications of such nanocomposites in functional leather finishing, particularly for products requiring enhanced hygienic properties.
To validate the proposed mechanism underlying the release of Zn2+ and Ag+ ions, the antimicrobial activity of synthetically prepared solutions of Zn2+ and Ag+ at concentrations equivalent to 1 M, comparable to those present in the nanocomposite suspensions employed for zone of inhibition (ZOI) assays, was examined (Fig. S6). The Zn2+ solution produced nearly identical inhibitory effects against the Gram-positive strain Bacillus cereus, with ZOI values ranging from 15 to 20 mm. Similarly, for the Gram-negative strain Escherichia coli, the Ag+ solution demonstrated comparable antimicrobial performance, yielding ZOI values in the range of 20 to 25 mm. These findings confirm that the observed antibacterial activity in the nanocomposite suspensions can be attributed, at least in part, to the release of Zn2+ and Ag+ ions.
It is well known that zinc and silver ions, which produce reactive oxidative stress (ROS) that includes superoxide anions (O2−), hydroxyl radicals (–OH), and hydrogen peroxide (H2O2), are formed in aqueous solutions. To test this hypothesis, the effects of scavengers on bacterial death induced by reactive oxygen species were investigated by using the antioxidant ascorbic acid (AA). B. cereus and E. coli were grown in four separate agar plates, and a well was created (hole) on the plates by using the upper side of the micropipette tips. 10 µL of AA (5 mM) solution + 10 µL of nanocomposite (0.5 M) solution samples were applied into the holes created by micropipette tips. One hole is kept for control using only ascorbic acid. Similarly, the nanocomposite (without AA) at the same concentration and amount was applied to the other two plates. One hole is kept for control using only protein. The plates were kept in the incubator for 24 hours and the ZOI was measured and the diameter of the ZOI (with AA and without AA) was compared. The bacteria's survival is shown in Fig. S7. It is clearly observed that the presence of AA, a scavenger of ROS, was neutralized, and no bacterial death occurred. On the other hand, without AA bacteria, death was certain. Thus, it can be concluded that the hydroxyl radicals and hydrogen peroxide produced by metal–protein nanocomposites play a role in the process of killing bacteria.
3.6. Mechanical properties of nanocomposite coated finished leather
The observed fastness properties affirm the suitability of the developed finishes for practical applications where repeated handling and exposure to moisture are expected. Moreover, the consistent performance across both dry and wet conditions underscores the stability of the nanocomposite coatings and their potential for commercial leather finishing.
All tested samples met or exceeded the minimum bond strength requirements, indicating satisfactory adhesion across the formulations, Notably, samples S-1 through S-4, which incorporated nanocomposite finishes with reduced binder content compared to the conventional formulation (Sample-5), still demonstrated robust adhesion and mechanical strength. This suggests that, despite their lower binder concentrations, nanocomposite systems effectively anchor to the leather surface, likely due to enhanced interfacial interactions facilitated by the nanoscale components.
These findings affirm the structural integrity and practical viability of the experimental finishes, supporting their application in high-performance leather products where both aesthetic and mechanical properties are essential.
4. Conclusions
This study successfully developed and characterized four protein-based metal nanocomposites Protein–ZnO, Ag–protein, PANI/protein–ZnO, and PANI/Ag–protein for applications in functional leather finishing. These nanocomposites, synthesized by integrating proteins with metal nanoparticles, demonstrated promising antimicrobial efficacy and material performance, addressing the growing need for biofunctional coatings in leather processing. Comprehensive physicochemical characterization using UV-vis spectroscopy, FT-IR, XRD, zeta potential analysis, TGA, FESEM, and EDX confirmed the successful incorporation of ZnO and Ag nanoparticles within the protein and polyaniline matrices. The resulting nanocomposites exhibited particle sizes ranging from 32 to 45 nm and favorable surface charge profiles, contributing to their colloidal stability and bioactivity. Cytotoxicity assessments indicated that the formulations were non-toxic, supporting their potential for safe use in consumer products. Antimicrobial evaluations, including minimum inhibitory concentration (MIC) and zone of inhibition (ZOI) tests against Bacillus cereus and Escherichia coli, revealed significant antibacterial activity. Protein–ZnO showed the highest efficacy against B. cereus (ZOI = 20 mm), while Ag–protein was the most effective against E. coli (ZOI = 22 mm). When applied to leather substrates, the nanocomposite finishes enhance key performance attributes, including surface glaze, antimicrobial protection, and coloration. Notably, protein–ZnO-treated leather exhibited the highest ZOI (18 mm) against B. cereus, and Ag–protein-treated leather showed a ZOI of 14 mm against E. coli, outperforming conventional finishes. These findings demonstrate the potential of protein-based metal nanocomposites to improve the functional and aesthetic properties of low-grade crust leather while preserving its natural texture and appearance. The incorporation of bioactive nanoparticles offers a sustainable and effective approach to leather finishing, with implications for hygiene, durability, and consumer comfort. Future research may explore the electrical conductivity and electroactive behavior of polyaniline-based nanocomposites, paving the way for advanced applications in smart textiles, wearable electronics, and multifunctional leather products.Author contributions
Abdulla-Al-Mamun M: conceptualization, methodology, writing – reviewing, supervision, and project administration; Liza SA: formal analysis, investigation, data curation, writing – original draft preparation, equal contribution of the first authorship; Nazmul Islam M: protein and amino acid analysis; Rezaul Karim Rana M: SDS-PAGE analysis; Shariful Islam M: supervision. All authors have read and agreed to the publication of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
This manuscript contains all of the data generated or analyzed during this research. The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01510c.Acknowledgements
The authors would like to express their gratitude for the essential analytical and technical support provided by the Centre for Advanced Research in Sciences (CARS), Dhaka University, and the Bangladesh Council for Scientific and Industrial Research (BCSIR).References
- D. R. Bagal-Kestwal, R. M. Kestwal and B. H. Chiang, Bio-based Nanomaterials and Their Bionanocomposites, Nanomaterials and Nanocomposites: Zero-to Three-Dimensional Materials and Their Composites, Wiley-VCH Verlag GmbH & Co. KGaA, 2016, pp. 255–330 DOI:10.1002/9783527683772.ch8.
- H. Ahari, L. Golestan, S. A. A. Anvar, I. Cacciotti, F. Garavand, A. Rezaei, M. A. Sani and S. M. Jafari, Bio-nanocomposites as food packaging materials; the main production techniques and analytical parameters, Adv. Colloid Interface Sci., 2022, 310, 102806, DOI:10.1016/j.cis.2022.102806.
- A. Rodzik, P. Pomastowski, G. N. Sagandykova and B. Buszewski, Interactions of whey proteins with metal ions, Int. J. Mol. Sci., 2020, 21(6), 2156, DOI:10.3390/ijms21062156.
- R. Javed, M. Zia, S. Naz, S. O. Aisida, N. U. Ain and Q. Ao, Role of capping agents in the application of nanoparticles in biomedicine and environmental remediation: recent trends and future prospects, J. Nanobiotechnol., 2020, 18(1), 172, DOI:10.1186/s12951-020-00704-4.
- S. Gulati, M. Sachdeva and K. K. Bhasin, Capping agents in nanoparticle synthesis: Surfactant and solvent system, AIP Conf. Proc., 2018, 1953(1), 030214, DOI:10.1063/1.5032549.
- J. F. Floriano, M. C. da Silva, M. M. de Oliveira and A. A. de Souza, Metal-protein nanocomposites: A sustainable approach for antimicrobial applications in leather finishing, J. Clean. Prod., 2023, 408, 137245, DOI:10.1016/j.jclepro.2023.137245.
- W. Xiao, Z. Liu, W. Luo, Y. Gao, L. Chang, Y. Li and P. Xu, Specific and unbiased detection of polyubiquitination via a sensitive non-antibody approach, Anal. Chem., 2020, 92(1), 1074–1080, DOI:10.1021/acs.analchem.9b04594.
- R. Hu, Y. Li, Y. Yang and M. Liu, Mass spectrometry-based strategies for single-cell metabolomics, Mass Spectrom. Rev., 2023, 42(1), 67–94, DOI:10.1002/mas.21689.
- M. Darder, P. Aranda and E. Ruiz-Hitzky, Bionanocomposites: A New Concept of Ecological, Bioinspired, and Functional Hybrid Materials, Adv. Mater., 2007, 19, 1309–1319, DOI:10.1002/adma.200602328.
- S. C. Warangkar, M. R. Deshpande, N. D. Totewad and A. A. Singh, Antibacterial, Antifungal and Antiviral Nanocomposites: Recent Advances and Mechanisms of Action, Biocomposites – Recent Advances, IntechOpen, 2022 DOI:10.5772/intechopen.108994.
- A. Mobed, M. Hasanzadeh and F. Seidi, Anti-bacterial activity of gold nanocomposites as a new nanomaterial weapon to combat photogenic agents: recent advances and challenges, RSC Adv., 2021, 11, 34688–34698, 10.1039/D1RA06030A.
- M. Harun-Ur-Rashid, T. Foyez, S. B. N. Krishna, S. Poda and A. B. Imran, Recent advances of silver nanoparticle-based polymer nanocomposites for biomedical applications, RSC Adv., 2025, 15, 8480–8505, 10.1039/D4RA08220F.
- L. M. Ditu, R. Bucuresteanu, M. Ionita, A. Neacsu and I. Calinescu, Antimicrobial and Antibiofilm Properties of Nanocomposite Surfaces with Biomedical Applications, Nanocomposites – Properties, Preparations and Applications, IntechOpen, 2024 DOI:10.5772/intechopen.115120.
- M. Gargano, A. Bacardit, G. Sannia and V. Lettera, From Leather Wastes back to Leather Manufacturing: The Development of New Bio-Based Finishing Systems, Coatings, 2023, 13(4), 775, DOI:10.3390/coatings13040775.
- C. Winter, C. Borges Agustini and M. Gutterres, Behavior of Polymer Films and its Blends for Leather Finishing, Trends Text. Eng. Fashion Technol., 2018, 1(4), 93–102, DOI:10.31031/TTEFT.2018.01.000518.
- X. Wu, J. Wu, C. Mu, C. Wang and W. Lin, Advances in antimicrobial polymer coatings in the leather industry: a comprehensive review, Ind. Eng. Chem. Res., 2021, 60(42), 15004–15018, DOI:10.1021/acs.iecr.1c02600.
- M. R. Khan, M. S. R. Shakil, B. Vorselaars, M. Rahman, I. A. Jawad and W. Ahmed, Nanotechnology in Leather Manufacturing, ed. Jackson, M. J., Ahmed, W., in Micro and Nanomanufacturing, Springer Nature, Switzerland, 2024, vol. 2, pp. 23–52 DOI:10.1007/978-3-031-70499-4_2.
- B. Sahu, A. Ramesh and F. Zameer, Impact of nanomaterials on leather: a nano-Saga from processing to application, Clean Technol. Environ. Policy, 2024, 1–28, DOI:10.1007/s10098-024-02912-0.
- T. S. Vijayakumar, S. Mahboob, G. Bupesh, S. Vasanth, K. A. Al-Ghanim, F. Al-Misned and M. Govindarajan, Facile synthesis and biophysical characterization of egg albumen-wrapped zinc oxide nanoparticles: a potential drug delivery vehicle for anticancer therapy, J. Drug Delivery Sci. Technol., 2020, 60, 102015, DOI:10.1016/j.jddst.2020.102015.
- A. M. Alahe, S. A. Liza, R. K. Raha and M. Abdulla-Al-Mamun, Protein-Based Polymeric Metal–Nanocomposite Derived From Egg Albumin: A Highly Effective Antimicrobial Agent Against Bacteria, Appl. Organomet. Chem., 2025, 39(1), e7780, DOI:10.1002/aoc.7780.
- Y. A. Arfat, S. Benjakul, T. Prodpran, P. Sumpavapol and P. Songtipya, Properties and antimicrobial activity of fish protein isolate/fish skin gelatin film containing basil leaf essential oil and zinc oxide nanoparticles, Food Hydrocoll., 2014, 41, 265–273, DOI:10.1016/j.foodhyd.2014.04.023.
- M. Sitohy, A. R. Al-Mohammadi, A. Osman, S. Abdel-Shafi, N. El-Gazzar, S. Hamdi, S. H. Ismail and G. Enan, Silver-protein nanocomposites as antimicrobial agents, Nanomaterials, 2021, 11(11), 3006, DOI:10.3390/nano11113006.
- A. Amani, M. Montazer and M. Mahmoudirad, Synthesis of applicable hydrogel corn silk/ZnO nanocomposites on polyester fabric with antimicrobial properties and low cytotoxicity, Int. J. Biol. Macromol., 2019, 123, 1079–1090, DOI:10.1016/j.ijbiomac.2018.11.093.
- G. J. C. Canama, M. C. L. Delco, R. A. Talandron and N. P. Tan, Synthesis of chitosan-silver nanocomposite and its evaluation as an antibacterial coating for mobile phone glass protectors, ACS Omega, 2023, 8(20), 17699–17711, DOI:10.1021/acsomega.3c00191.
- Y. Xiao, Y. Liu, S. Kang, K. Wang and H. Xu, Development and evaluation of soy protein isolate-based antibacterial nanocomposite films containing cellulose nanocrystals and zinc oxide nanoparticles, Food Hydrocoll., 2020, 106, 105898, DOI:10.1016/j.foodhyd.2020.105898.
- E. Yuan, M. Zhou, S. Nie and J. Ren, Interaction mechanism between ZnO nanoparticles-whey protein and its effect on toxicity in GES-1 cells, J. Food Sci., 2022, 87(6), 2417–2426, DOI:10.1111/1750-3841.16193.
- N. Strambeanu, L. Demetrovici, D. Dragos and M. Lungu, Nanoparticles: Definition, classification and general physical properties. in Nanoparticles' Promises and Risks: Characterization, Manipulation, and Potential Hazards to Humanity and the Environment, Springer International Publishing, Cham, 2014, pp. 3–8 DOI:10.1007/978-3-319-11728-7_1.
- C. Gaidau, A. Petica, M. Ignat, O. Iordache, L. M. Ditu and M. Ionescu, Enhanced photocatalysts based on Ag-TiO2 and Ag-N-TiO2 nanoparticles for multifunctional leather surface coating, Open Chem., 2016, 14(1), 383–392, DOI:10.1515/chem-2016-0040.
- J. Liu, J. Ma, Y. Bao, J. Wang, H. Tang and L. Zhang, Polyacrylate/surface-modified ZnO nanocomposite as film-forming agent for leather finishing, Int. J. Polym. Mater. Polym., 2014, 63(16), 809–814, DOI:10.1080/00914037.2014.886217.
- W. Chen, L. Feng and B. Qu, In situ synthesis of poly (methyl methacrylate)/MgAl layered double hydroxide nanocomposite with high transparency and enhanced thermal properties, Solid State Commun., 2004, 130(3–4), 259–263, DOI:10.1016/j.ssc.2004.01.031.
- Y. Wang, J. Ma, Q. Xu and J. Zhang, Fabrication of antibacterial casein-based ZnO nanocomposite for flexible coatings, Mater. Des., 2017, 113, 240–245, DOI:10.1016/j.matdes.2016.09.082.
- S. Saravanabhavan, K. J. Sreeram, J. Raghava Rao and B. Unni Nair, The three pot solution for chromium, tannins and solid wastes: Recovery and reuse technique for spent semi-chrome liquor and chrome shavings, J. Soc. Leather Technol. Chem., 2004, 88(5), 202–207 CAS.
- S. A. Liza and M. Abdulla-Al-Mamun, Microbial degradation of tannery chrome-solid waste using Bacillus thuringiensis: optimization of collagen hydrolysate extraction via response surface methodology, RSC Sustainability, 2025, 3, 3198–3214, 10.1039/D4SU00666F.
- K. E. Ramohlola, G. R. Monana, M. J. Hato, K. D. Modibane, K. M. Molapo, M. Masikini, S. B. Mduli and E. I. Iwuoha, Polyaniline-metal organic framework nanocomposite as an efficient electrocatalyst for hydrogen evolution reaction, Composites, Part B, 2018, 137, 129–139, DOI:10.1016/j.compositesb.2017.11.016.
- H. K. Mahre, L. Dalheim, G. K. Edvinsen, E. O. Elvevoll and I. J. Jensen, Protein Determination_Method Matters, Foods, 2018, 7(1), 5, DOI:10.3390/foods7010005.
- P. Weber, Determination of amino acids in food and feed by microwave hydrolysis and UHPLC-MS/MS, J. Chromatogr. B:Anal. Technol. Biomed. Life Sci., 2022, 1209, 123429, DOI:10.1016/j.jchromb.2022.123429.
- P. Parvekar, J. Palaskar, S. Metgud, R. Maria and S. Dutta, The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of silver nanoparticles against Staphylococcus aureus, Biomater. Investig. Dent., 2020, 7(1), 105–109, DOI:10.1080/26415275.2020.1796674.
- F. W. Sosulski and G. I. Imafidon, Amino acid composition and nitrogen-to-protein conversion factors for animal and plant foods, J. Agric. Food Chem., 1990, 38(6), 1351–1356, DOI:10.1021/jf00096a011.
- M. Arnold and A. Barbul, Nutrition and wound healing, Plast. Reconstr. Surg., 2006, 117(7S), 42S–58S, DOI:10.1097/01.prs.0000225432.17501.6c.
- D. Acharya, B. Mohanta, P. Pandey, M. Singha and F. Nasiri, Optical and antibacterial properties of synthesised silver nanoparticles, Micro Nano Lett., 2017, 12(4), 223–226, DOI:10.1049/mnl.2016.0666.
- R. Das and S. Sarkar, Optical properties of silver nano-cubes, Opt. Mater., 2015, 48, 203–208, DOI:10.1016/j.optmat.2015.07.038.
- D. K. Singh, D. K. Pandey, R. R. Yadav and D. Singh, A study of nanosized zinc oxide and its nanofluid, Pramana, 2012, 78(5), 759–766, DOI:10.1007/s12043-012-0275-8.
- L. Shi, J. Zhou and S. Gunasekaran, Low temperature fabrication of ZnO–whey protein isolate nanocomposite, Mater. Lett., 2008, 62(28), 4383–4385, DOI:10.1016/j.matlet.2008.07.038.
- M. Venkatesham, D. Ayodhya, A. Madhusudhan, A. Santoshi Kumari, G. Veerabhadram and K. Girija Mangatayaru, A novel green synthesis of silver nanoparticles using gum karaya: characterization, antimicrobial and catalytic activity studies, J. Clust. Sci., 2014, 25(2), 409–422, DOI:10.1007/s10876-013-0620-1.
- M. V. Fuke, A. Vijayan, P. Kanitkar, M. Kulkarni, B. B. Kale and R. C. Aiyer, Ag-polyaniline nanocomposite cladded planar optical waveguide based humidity sensor, J. Mater. Sci.: Mater. Electron., 2009, 20(8), 695–703, DOI:10.1007/s10854-008-9787-x.
- M. S. Tamboli, M. V. Kulkarni, R. H. Patil, W. N. Gade, S. C. Navale and B. B. Kale, Nanowires of silver-polyaniline nanocomposite synthesized via in situ polymerization and its novel functionality as an antibacterial agent, Colloids Surf., B, 2012, 92, 35–41, DOI:10.1016/j.colsurfb.2011.11.006.
- S. Krimm and J. Bandekar, Vibrational spectroscopy and conformation of peptides, polypeptides, and proteins, Adv. Protein Chem., 1986, 38, 181–364, DOI:10.1016/S0065-3233(08)60528-8.
- R. Murali, A. Anumary, M. Ashokkumar, P. Thanikaivelan and B. Chandrasekaran, Hybrid biodegradable films from collagenous wastes and natural polymers for biomedical applications, Waste Biomass Valorization, 2011, 2(3), 323–335, DOI:10.1007/s12649-011-9072-8.
- D. Pradhan, L. B. Sukla, B. B. Mishra and N. Devi, Biosorption for removal of hexavalent chromium using microalgae Scenedesmus sp, J. Cleaner Prod., 2019, 209, 617–629, DOI:10.1016/j.jclepro.2018.10.288.
- N. Jayarambabu, B. S. Kumari, K. V. Rao and Y. T. Prabhu, Germination and growth characteristics of mungbean seeds (Vigna radiata L.) affected by synthesized zinc oxide nanoparticles, Int. J. Curr. Eng. Technol., 2014, 4(5), 3411–3416 Search PubMed.
- J. Zhao, J. Wang and G. Ma, Extremely concentrated silver nanoparticles stabilized in aqueous solution by Bovine Serum Albumin (BSA), Nano-Struct. Nano-Objects, 2019, 19, 100349, DOI:10.1016/j.nanoso.2019.100349.
- M. Ragothaman, A. K. Villalan, A. Dhanasekaran and T. Palanisamy, Bio-hybrid hydrogel comprising collagen-capped silver nanoparticles and melatonin for accelerated tissue regeneration in skin defects, Mater. Sci. Eng. C., 2021, 128, 112328, DOI:10.1016/j.msec.2021.112328.
- Y. B. Wankhede, S. B. Kondawar, S. R. Thakare and P. S. More, Synthesis and characterization of silver nanoparticles embedded in polyaniline nanocomposite, Adv. Mater. Lett., 2013, 4(1), 89–93, DOI:10.5185/amlett.2012.icnano.108.
- K. R. Reddy, B. C. Sin, K. S. Ryu, J. C. Kim, H. Chung and Y. Lee, Conducting polymer functionalized multi-walled carbon nanotubes with noble metal nanoparticles: synthesis, morphological characteristics and electrical properties, Synth. Met., 2009, 159(7–8), 595–603, DOI:10.1016/j.synthmet.2008.11.030.
- S. Muthusamy and J. Charles, In situ synthesis and characterization of polyaniline/prussian blue/zinc oxide nanocomposite, Polym. Bull., 2019, 76(1), 119–137, DOI:10.1007/s00289-018-2350-x.
- M. Vanaja and G. Annadurai, Coleus aromaticus leaf extract mediated synthesis of silver nanoparticles and its bactericidal activity, Appl. Nanosci., 2013, 3(3), 217–223, DOI:10.1007/s13204-012-0121-9.
- Y. Li, Z. Li and F. Zheng, Polyaniline/silver/cerium nitrate ternary composite: Synthesis, characterization and enhanced electrochemical properties, J. Appl. Polym. Sci., 2015, 132(46), 42785, DOI:10.1002/app.42785.
- L. J. Pan, L. Pu, Y. Shi, T. Sun, R. Zhang and Y. O. Zheng, Hydrothermal synthesis of polyaniline mesostructures, Adv. Funct. Mater., 2006, 16(10), 1279–1288, DOI:10.1002/adfm.200500543.
- H. S. Abdulla and A. I. Abbo, Optical and electrical properties of thin films of polyaniline and polypyrrole, Int. J. Electrochem. Sci., 2012, 7(11), 10666–10678, DOI:10.1016/S1452-3981(23)16893-3.
- R. Paulraj, P. Shankar, G. K. Mani, L. Nallathambi and J. B. B. Rayappan, Fabrication of PANI-ZnO nanocomposite thin film for room temperature methanol sensor, J. Mater. Sci., 2017, 28(15), 10799–10805, DOI:10.1007/s10854-017-6857-y.
- S. Muthusamy and J. Charles, In situ synthesis and characterization of polyaniline/prussian blue/zinc oxide nanocomposite, Polym. Bull., 2019, 76(1), 119–137, DOI:10.1007/s00289-018-2350-x.
- R. Kumar, R. D. Anandjiwala and A. Kumar, Thermal and mechanical properties of mandelic acid-incorporated soy protein films, J. Therm. Anal. Calorim., 2016, 123(2), 1273–1279, DOI:10.1007/s10973-015-5035-9.
- J. A. Lee, M. J. Yoon, E. S. Lee, D. Y. Lim and K. Y. Kim, Preparation and characterization of cellulose nanofibers (CNFs) from microcrystalline cellulose (MCC) and CNF/polyamide 6 composites, Macromol. Res., 2014, 22(7), 738–745, DOI:10.1007/s13233-014-2121-y.
- M. O. Ansari and F. Mohammad, Thermal stability, electrical conductivity and ammonia sensing studies on p-toluenesulfonic acid doped polyaniline: titanium dioxide (pTSA/Pani: TiO2) nanocomposites, Sens. Actuators, B, 2011, 157(1), 122–129, DOI:10.1016/j.snb.2011.03.036.
- J. G. Wang, Y. Yang, Z. H. Huang and F. Kang, Interfacial synthesis of mesoporous MnO2/polyaniline hollow spheres and their application in electrochemical capacitors, J. Power Sources, 2012, 204, 236–243, DOI:10.1016/j.jpowsour.2011.12.057.
- A. G. Kaningini, S. Azizi, N. Sintwa, K. Mokalane, K. C. Mohale, F. N. Mudau and M. Maaza, Effect of optimized precursor concentration, temperature, and doping on optical properties of ZnO nanoparticles synthesized via a green route using bush tea (Athrixia phylicoides DC.) leaf extracts, ACS Omega, 2022, 7(36), 31658–31666, DOI:10.1021/acsomega.2c00530.
- A. R. Maheo, B. S. M. Vithiya, T. A. Arul Prasad, V. L. Mangesh, T. Perumal, W. H. Al-Qahtani and M. Govindasamy, Cytotoxic, antidiabetic, and antioxidant study of biogenically improvised elsholtzia blanda and chitosan-assisted zinc oxide nanoparticles, ACS Omega, 2023, 8(12), 10954–10967, DOI:10.1021/acsomega.2c07530.
- J. D. Clogston and A. K. Patri, Zeta potential measurement, Characterization of nanoparticles intended for drug delivery, Humana press, Totowa, NJ, 2010, pp. 63–70 DOI:10.1007/978-1-60327-198-1_6.
- K. Cheng, W. Weng, H. Wang and S. Zhang, In vitro behavior of osteoblast-like cells on fluoridated hydroxyapatite coatings, Bio. Mater., 2005, 26(32), 6288–6295, DOI:10.1016/j.biomaterials.2005.03.041.
- Y. Xie, Y. He, P. L. Irwin, T. Jin and X. Shi, Antibacterial activity and mechanism of action of zinc oxide nanoparticles against Campylobacter jejuni, Appl. Environ. Microbiol., 2011, 77(7), 2325–2331, DOI:10.1128/AEM.02149-10.
- X. Chen and H. J. Schluesener, Nanosilver: a nanoproduct in medical application, Toxicol. Lett., 2008, 176(1), 1–12, DOI:10.1016/j.toxlet.2007.10.004.
- J. R. Morones, J. L. Elechiguerra, A. Camacho, K. Holt, J. B. Kouri, J. T. Ramírez and M. J. Yacaman, The bactericidal effect of silver nanoparticles, Nanotechnology, 2005, 16(10), 2346, DOI:10.1088/0957-4484/16/10/059.
- Y. Y. Loo, Y. Rukayadi, M. A. R. Nor-Khaizura, C. H. Kuan, B. W. Chieng, M. Nishibuchi and S. Radu, In vitro antimicrobial activity of green synthesized silver nanoparticles against selected Gram-negative foodborne pathogens, Front. Microbiol., 2018, 9, 1555, DOI:10.3389/fmicb.2018.01555.
- H. S. Kolla, S. P. Surwade, X. Zhang, A. G. MacDiarmid and S. K. Manohar, Absolute molecular weight of polyaniline, J. Am. Chem. Soc., 2005, 127(48), 16770–16771, DOI:10.1021/ja055327k.
- F. Fu, L. Li, L. Liu, J. Cai, Y. Zhang, J. Zhou and L. Zhang, Construction of cellulose based ZnO nanocomposite films with antibacterial properties through one-step coagulation, ACS Appl. Mater. Interfaces, 2015, 7(4), 2597–2606, DOI:10.1021/am507639b.
- D. Milašienė, V. Jankauskaitė and R. Arcišauskaitė, Prediction of stress relaxation in laminated leather layers, Mater. Sci., 2003, 9(1), 73–79 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




