Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Click-enabled tetrazine ionic liquid as an advanced material for chemo-selective gas detection via inverse electron-demand Diels–Alder reaction
Chien-Yuan
Chen
,
Yu-Hsin
Chung
,
Yu-Qian
Qiu
,
Wen-Chun
Su
,
Wen-Ting
Hsiao
and
Yen-Ho
Chu
 *
*
Department of Chemistry and Biochemistry, National Chung Cheng University, Chiayi 62102, Taiwan, Republic of China. E-mail: cheyhc@ccu.edu.tw; Fax: +886 52721040; Tel: +886 52729139
First published on 15th January 2026
Abstract
A room-temperature 1,2,4,5-tetrazine ionic liquid (TIL) was synthesized in eight steps with an overall yield of 8.5% as a new functional material for chemoselective target gas detection. The TIL, bearing two electron-withdrawing substituents on the tetrazine ring, served as a reactive probe for alkene and alkyne vapors through the rapid inverse electron-demand Diels–Alder (IEDDA) [4+2] cycloaddition. When thin-coated on a quartz crystal microbalance (QCM), the TIL enabled real-time monitoring of gaseous styrene, strained cycloalkenes, and cyclooctyne with distinct frequency responses and high sensitivity. These results demonstrate that tetrazine-based ionic liquids can act as both reaction media and chemo-sensing elements. This study establishes a concise synthetic route and introduces an IEDDA reaction-based platform for the selective and sensitive detection of unsaturated hydrocarbons.
1. Introduction
Click chemistry provides a versatile platform for constructing functional molecular architectures with high efficiency and selectivity. Based on this principle, we report the concise synthesis and development of a room-temperature 1,2,4,5-tetrazine ionic liquid (TIL) (Fig. 1) as a new functional material for the rapid and chemo-selective detection of target alkene and alkyne gases, monitored in real time by quartz crystal microbalance (QCM) analysis.1,2The 1,2,4,5-tetrazine framework is a benzene-like heterocycle in which four CH units are replaced by electronegative nitrogen atoms.3 It serves as a key structural motif in energy-rich4 and optoelectronic5 materials and, unlike the aromatic and inert benzene ring, readily undergoes inverse electron-demand Diels–Alder (IEDDA) reactions with dienophiles. This unique reactivity has enabled widespread applications across chemical biology, materials chemistry, and polymer science.6–10
Ionic liquids (ILs) are organic salts that remain liquid below 100 °C. Unlike neutral molecular solvents, ILs are entirely composed of cations and anions with asymmetric geometries and delocalized charges, leading to weak interionic coordination and low melting points.11–13 This ionic nature imparts ILs with highly tunable physicochemical properties, including adjustable polarity, excellent solvation ability toward both small and macromolecules, outstanding thermal and chemical stability, and negligible volatility.13 Moreover, when used as reaction media, ILs are known to stabilize polar transition states and reactive intermediates, thereby facilitating diverse organic transformations.14
Motivated by these insights, we designed and synthesized the TIL through a click-assembly strategy, aiming to integrate the reactivity of tetrazines with the functional versatility of ILs. The resulting material demonstrates notable potential for efficient alkene and alkyne capture under mild conditions.
2. Experimental
Detailed 1H and 13C NMR and HRMS spectra of TIL, the HRMS spectrum of the TIL–styrene adduct (Fig. S1), the QCM measurement protocol, and procedures for in-solution click reactions monitored spectrophotometrically are summarized in the SI.2.1. Synthesis of TIL
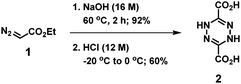
The synthesis of TIL was carried out according to the synthetic pathway illustrated in Fig. 2.To a round-bottom flask containing sodium hydroxide (2.27 g, 56.82 mmol) was added deionized water (4 mL). The 16 M NaOH solution was stirred at 60 °C. Ethyl diazoacetate 1 (1.4339 g, 10.9 mmol, 13% in dichloromethane) was added dropwise to the NaOH solution being stirred. After 2 h, the reaction slurry was poured into methanol (100 mL). The precipitate was collected by suction filtration and washed with methanol several times. The solvent was removed in vacuo to afford sodium 1,4-dihydro-1,2,4,5-tetrazine-3,6-dicarboxylate (1.085 g, 92% yield) as a yellow-brown solid. This product was only soluble in water and no 1H NMR signals were observed.
The sodium dicarboxylate (1.0828 g, 5.011 mmol) was dissolved in deionized water (20 mL) in a round-bottom flask, and the solution was stirred in an ice–salt bath. HCl (1 mL, 12 M) was added dropwise to the solution of sodium dicarboxylate while stirring. After 1 h, the dicarboxylic acid (product 2) precipitated from the aqueous medium. The product was collected by suction filtration and washed with diethyl ether several times. The solvent was removed in vacuo to afford 1,4-dihydro-1,2,4,5-tetrazine-3,6-dicarboxylic acid 2 (0.631 g, 60% yield) as a yellow solid. 1H NMR (400 MHz, DMSO-d6) δ 8.85 (s, 2 × NH, 2H); 13C NMR (100 MHz, DMSO-d6) δ 140.18, 160.45; ESI-HRMS m/z [M − H]− calculated for C4H4N4O4 171.0154, found 171.0158 ([M − H]−).
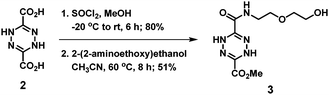
Thionyl chloride (0.5 mL) was added dropwise to a stirred cooled solution of methanol (3 mL) at −20 °C. A suspension of dicarboxylic acid 2 (0.2634 g, 15.3 mmol, 1.0 equiv.) in methanol was added in small portions to the cooled solution. The reaction mixture was then warmed to room temperature and stirred for 6 h to effect esterification. After 6 h, the solvent and thionyl chloride were removed under reduced pressure to afford a yellow solid. This crude product was purified by silica chromatography (ethyl acetate/hexane = 2/1, v/v) to give dimethyl 1,4-dihydro-1,2,4,5-tetrazine-3,6-dicarboxylate (0.246 g, 80% yield) as an orange solid. 1H NMR (400 MHz, CDCl3) δ 3.93 (s, 2 × CH3, 6H), 7.47 (s, 2 × NH, 2H).
To a stirred solution of dimethyl ester (0.4624 g, 2.309 mmol, 1.5 equiv.) in acetonitrile (4 mL) at 60 °C, 2-(2-aminoethoxy)ethanol (0.162 g, 1.539 mmol, 1.0 equiv.) was added. After 3 h, the solvent was removed under reduced pressure to afford the crude product. The crude product was purified by silica chromatography (methanol/dichloromethane = 1/20, v/v) to give methyl 6-((2-(2-hydroxyethoxy)ethyl)carbamoyl)-1,4-dihydro-1,2,4,5-tetrazine-3-carboxylate 3 (0.2158 g, 51% yield) as an orange solid. 1H NMR (400 MHz, CDCl3) δ 3.52–3.58 (m, OCH2CH2N, 2H), 3.58–3.66 (m, OCH2CH2N + HOCH2CH2O, 4H), 3.76 (t, J = 4 Hz, HOCH2CH2O, 2H), 3.92 (s, CH3, 3H), 7.19 (s, NHCO, 1H), 7.31 (s, NH, 1H), 7.77 (s, NH, 1H); 13C NMR (100 MHz, CDCl3) δ 39.50, 53.70, 61.80, 69.35, 72.32, 138.51, 140.10, 157.57, 159.44; ESI-HRMS m/z [M + Na]+ calculated for C9H15N5O5 296.0965, found 296.0958 ([M + Na]+).
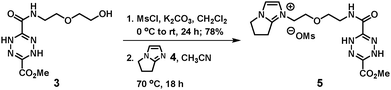
A suspension of monoamide 3 (0.1347 g, 0.493 mmol, 1.0 equiv.) and K2CO3 (0.204 g, 1.48 mmol, 3.0 equiv.) in dichloromethane (9 mL) was stirred in an ice bath. A solution of methanesulfonyl chloride (0.085 g, 0.74 mmol, 1.5 equiv.) in dichloromethane (1 mL) was added dropwise to the ice-chilled suspension. After completion of the addition, the reaction mixture was allowed to warm to room temperature and stirred for 12 h. At that time, the unreacted starting material was about 50% (monitored by TLC). An additional portion of the methanesulfonyl chloride solution (1 mL, 1.5 equiv.) and K2CO3 (3.0 equiv.) was then added to the reaction mixture. After a further 12 h, the reaction was diluted with deionized water and the aqueous phase was extracted with dichloromethane. The combined organic extracts were dried over Na2SO4 and concentrated under reduced pressure to give an orange crude product. The crude product was purified by silica chromatography (methanol/dichloromethane = 1/20, v/v) to give methyl 6-((2-(2-((methylsulfonyl)oxy)ethoxy)ethyl)carbamoyl)-1,4-dihydro-1,2,4,5-tetrazine-3-carboxylate (0.135 g, 78% yield) as an orange solid. 1H NMR (400 MHz, CDCl3) δ 3.06 (s, SCH3, 3H), 3.51–3.58 (m, OCH2CH2N, 2H), 3.62 (t, J = 4 Hz, OCH2CH2N, 2H), 3.73–3.77 (m, MsOCH2CH2O, 2H), 3.92 (s, CH3, 3H), 4.35–4.40 (t, J = 4 Hz, MsOCH2CH2O, 2H), 7.12 (s, NHCO, 1H), 7.28 (s, NH, 1H), 7.72 (s, NH, 1H).
To a solution containing the methanesulfonate (0.12 g, 0.342 mmol, 1.2 equiv.) and 6,7-dihydro-5H-pyrrolo[1,2-a]imidazole 4 (0.031 g, 0.285 mmol, 1.0 equiv.) previously developed in our laboratory,15 acetonitrile (0.2 mL) was added and the solution was stirred at 70 °C for the alkylation reaction. After 18 h, the reaction was completed (monitored by TLC). The resulting ionic salt was dissolved in deionized water and extracted with dichloromethane to remove excess methanesulfonate. The combined organic phase was concentrated under reduced pressure to give [DHTz-3C-im][OMs] 5 (0.073 g, 56% yield) as an orange solid. 1H NMR (400 MHz, DMSO-d6) δ 2.30 (s, SCH3, 3H), 2.65–2.68 (m, NCH2CH2CH2, 2H), 3.11–3.15 (m, NCH2CH2CH2, 2H), 3.24–3.31 (m, OCH2CH2N, 2H), 3.47 (t, J = 4 Hz, OCH2CH2N, 2H), 3.70 (t, J = 6 Hz, OCH2CH2N, 2H), 3.80 (s, CH3, 3H), 4.19–4.25 (m, N+CH2CH2O + N CH2CH2CH2, 4H), 7.60 (d, J = 4 Hz, im-H, 2H), 8.30 (t, J = 4 Hz, NHCO, 1H), 8.97 (d, J = 4 Hz, NH, 1H); 13C NMR (400 MHz, DMSO-d6) δ 23.19, 25.98, 29.48, 38.99, 48.36, 48.70, 53.48, 68.52, 68.86, 118.24, 126.46, 139.79, 141.47, 153.33, 158.40, 159.69; ESI-HRMS m/z [M]+ calculated for C15H22N7O4+ 364.1728, found 364.1731 ([M]+).
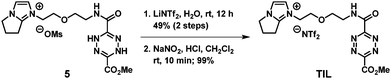
To a solution containing [DHTz-3C-im][OMs] 5 (0.390 g, 0.850 mmol, 1.0 equiv.) in deionized water (5 mL), LiNTf2 (0.268, 0.935 mmol, 1.1 equiv.) was added and the solution was stirred at room temperature for 12 h to effect ion exchange. Upon completion, the reaction mixture was extracted with dichloromethane. The combined organic extracts were dried over Na2SO4 and concentrated under reduced pressure to give the orange crude product. The crude material was purified by silica chromatography (methanol/dichloromethane = 1/20, v/v) to afford [DHTz-3C-im][NTf2] (0.2679 g, 49% yield) as an orange solid.
A sample vial containing [DHTz-3C-im][NTf2] (22.26 mg, 0.0345 mmol, 1.0 equiv.) in dichloromethane (10 mL) was sealed with a septum and PTFE tubing, and stirred at room temperature. In a separate three-neck round–bottom flask with septums, NaNO2 (111 mg, 1.6 mmol, 46 equiv.) in deionized water (25 mL) was treated with hydrochloric acid (2 mL, 12 M) added via syringe to generate NOx gas. Nitrogen from a balloon was used as a carrier gas to transfer the NOx to the reaction vial via PTFE tubing. After 10 min, the solution color changed from yellow to red. The solvent was then removed under reduced pressure, affording the desired TIL (22.1 mg, quantitative yield) as a red liquid. 1H NMR (400 MHz, CD3CN) δ 2.66–2.78 (m, NCH2CH2CH2, 2H), 3.12 (t, J = 6 Hz, NCH2CH2CH2, 2H), 3.67 (s, CH3, 3H), 3.77 (m, OCH2CH2N, 2H), 4.11–4.26 (m, N+CH2CH2O + N CH2CH2CH2 + OCH2CH2N, 8H), 7.14 (d, J = 2 Hz, im-H, 1H), 7.25 (t, J = 2 Hz, im-H, 1H), 8.33 (s, NH, 1H); 13C NMR (100 MHz, CD3CN) δ 23.38, 25.93, 39.68, 48.57, 49.21, 54.36, 68.64, 69.38, 120.31 (q, JCF = 319 Hz), 126.24, 153.80, 159.02, 159.16, 159.61, 161.23; ESI-HRMS m/z [M]+ calculated for C15H20N7O4+ 362.1571, found 362.1564 ([M]+).
3. Results and discussion
3.1. Synthesis of TIL
Fig. 2 outlines our synthesis of TIL in which the key, highly reactive tetrazine element was obtained by oxidizing the dihydrotetrazine-conjugated IL in the final step. We commenced the total synthesis of TIL from commercially available ethyl diazoacetate (1) using the procedure reported by Boger and co-workers15 under basic conditions, followed by acid neutralization to afford the dihydrotetrazine dicarboxylic acid (2) with a combined yield of 55% over two steps. Subsequent acid activation and amide bond formation of 2 with 2-(2-aminoethoxy)ethanol afforded compound 3, bearing a flexible diethylene glycol linker. This intermediate then underwent a sequence of three reactions: alcohol mesylation, nucleophilic substitution with the bicyclic 6,7-dihydro-5H-pyrrolo[1,2-a]imidazole (4) previously developed in our laboratory,16 to yield the dihydrotetrazine-conjugated IL (5) as a shelf-stable solid, and lastly, anion metathesis. The final oxidation of the dihydrotetrazine moiety using sodium nitrite under acidic conditions produced the desired TIL as a viscous red liquid at room temperature. The synthesis of TIL was concise in the number of steps, and in our hands, the overall isolated yield of 8.5% over eight steps was considered satisfactory.3.2. IEDDA-based, chemo-selective detection of alkene and alkyne gases by TIL-coated QCM
The absence of vapor pressure ensures that room-temperature ILs remain stable and do not evaporate from QCM quartz chips, rendering the TIL highly suitable for reactions with target gaseous molecules. In other words, the chemo-selective gas detection using a thin TIL coating on QCM chips takes place when analyte gases rapidly diffuse into the TIL layer, where the specific IEDDA reaction with the target gases occurs and can be readily monitored by resonance frequency decreases (ΔF) using QCM. Fig. 3 illustrates our custom-built TIL-on-QCM gas analysis setup integrated with a commercial QCM922A detector (Princeton Applied Research, Oak Ridge, TN, USA) (see Fig. S1 for photos of a lab-built QCM-based gas sensing system and its reaction chamber, SI).In this study, we focused on the IEDDA reactions enabling the detection of alkynes and alkenes by TIL for two main reasons. First, unsubstituted tetrazine is known to react selectively with both alkene and alkyne molecules,9 and second, unlike organic azides used in Cu(I)-catalyzed azide–alkyne cycloaddition (CuACC) reactions,17,18 the reactivity of 1,2,4,5-tetrazines can be finely tuned by modulating their electron deficiency through functionalization at the 3- and 6-positions. In our case, the reaction rate needed to be sufficiently fast to allow gaseous alkenes and alkynes to react efficiently with TIL, enabling their detection by QCM. Furthermore, due to their intrinsic absorbance in the 510–550 nm range,6–9 these rapid click reactions could also be conveniently monitored spectrophotometrically in solutions.
3.3. IEDDA reactivity of TIL toward alkene and alkyne gases on QCM
We first examined the reactivities of acyclic, unconstrained alkene and alkyne gases using this system. As shown in Fig. 4, TIL exhibited no apparent reactivity toward, and thus failed to detect, terminal (1-pentene, vinylcyclohexane, 1-pentyne, and phenylacetylene) or internal (2-pentene, 2-methyl-2-butene, and 2-pentyne) alkenes and alkynes. Likewise, dichloromethane used as the solvent for sample preparation was completely inert toward TIL.Pleasingly, TIL was found to readily react with styrene (ΔF = −116 Hz) but not to phenylacetylene (ΔF = 0 Hz). This positive QCM result confirmed with a previous study that a phenyl-substituent with π conjugation raised the HOMO energy of a terminal alkene and therefore facilitating its IEDDA reaction with the TIL where its electron-withdrawing groups significantly lower the LUMO energy of tetrazine diene, ultimately resulting in strong interactions and, finally, a fast response on QCM.19
Furthermore, the pronounced difference in reactivity between styrene and phenylacetylene toward 1,2,4,5-tetrazines has been computationally elucidated by Houk and co-workers,20 who demonstrated that alkenes exhibit higher HOMO energies and smaller distortion energies than alkynes in IEDDA reactions, accounting for their greater reactivity. Lastly, compared with styrene, TIL reacted much more slowly with sterically hindered β-methylstyrene (ΔF = −2.4 Hz) (Fig. 4).
3.4. Quantitative analysis of click reactions of styrene gas with TIL
As TIL proved effective for chemo-selective target gas analysis, we next carried out a detailed quantitative study on its reaction with styrene gas. The results are shown in Fig. 5. TIL exhibited a linear QCM frequency response across the tested concentrations range (0–3.00 ppm) (experimental sensorgrams in Fig. 5A and a fitting line in Fig. 5B). This TIL was highly sensitive to styrene gas: a resonance frequency decrease of 10 Hz (ΔF = −10 Hz) corresponded to a detectable concentration of 0.23 ppm (10/43.748, Fig. 5B). In addition, ESI-HRMS analysis confirmed the formation of the expected TIL-styrene adduct isolated from quartz chip, giving a measured molecular ion at m/z = 438.2146 ([M]+, calcd = 438.2136) corresponding to [C23H28N5O4]+ (Fig. S2, SI).3.5. TIL reactions with cycloalkenes and cyclooctyne
In addition to unstrained, acyclic alkenes and alkynes (Fig. 4), we also examined cycloalkenes (2.50 ppm each) as dienophiles for their IEDDA reactions with TIL to evaluate its performance as a new functional material for label-free target gas analysis. As shown in Fig. 6A, TIL displayed clear reactivity toward cyclopentene (ΔF = −24 Hz), cycloheptene (ΔF = −39 Hz) and cyclooctene (ΔF = −13 Hz), but remained unreactive toward the essentially unstrained cyclohexene (ΔF = 0 Hz). Similar reactivity trends of cycloalkenes in IEDDA reactions with 3,6-bis(trifluoromethyl)-1,2,4,5-tetrazine in dioxane solvent were previously reported by Sauer and co-workers.21 These observations have been further supported by computational studies, which attribute the reactivity of tetrazines toward cycloalkenes to their strong interaction energies (i.e., smaller HOMO–LUMO gaps leading to stronger orbital interactions) and ring distortion acceleration.20,22As strain-promoted click cycloaddition reactions have proven to be powerful tools with wide applications in chemistry, materials science, and polymer research,6–9 we further investigated the reactivity of TIL toward a highly strained cyclooctyne gas.23,24 As depicted in Fig. 6B, the IEDDA reaction of cyclooctyne with TIL was found to be ultrafast even at ppb levels (ΔF = −72 Hz at 250 ppb and ΔF = −135 Hz at 500 ppb), leading to efficient capturing of cyclooctyne. Due to its significant pre-distorted geometry that favors cycloaddition,20TIL responded sensitively to cyclooctyne (Fig. 6B) and was also capable of detecting cyclooctene at 500 ppb by QCM (ΔF = −135 and −5.6 Hz, respectively) (Fig. 6A).
3.6. In-solution click reaction of styrene with TIL
In addition to the TIL-on-QCM study of gaseous IEDDA reactions involving various alkenes and alkynes on quartz surfaces, we further examined the reactivity of TIL with styrene in acetonitrile solution at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 molar ratio. Remarkably, under these experimental conditions, the highly electron-deficient TIL reacted completely with styrene within 5 min, exhibiting a half-life (t1/2) of less than 30 s (Fig. 7). This reaction could be conveniently monitored spectrophotometrically by tracking the disappearance of the characteristic visible absorption band of 1,2,4,5-tetrazines typically observed between 510 and 550 nm, although the corresponding molar extinction coefficient is relatively low (∼500–1000 M−1 cm−1).7
10 molar ratio. Remarkably, under these experimental conditions, the highly electron-deficient TIL reacted completely with styrene within 5 min, exhibiting a half-life (t1/2) of less than 30 s (Fig. 7). This reaction could be conveniently monitored spectrophotometrically by tracking the disappearance of the characteristic visible absorption band of 1,2,4,5-tetrazines typically observed between 510 and 550 nm, although the corresponding molar extinction coefficient is relatively low (∼500–1000 M−1 cm−1).7
4. Conclusion
In this work, we demonstrated the concise chemical synthesis of a room-temperature 1,2,4,5-tetrazine ionic liquid (TIL) as a new functional material specifically designed for click-based, chemo-selective detection of alkenes and alkynes by QCM. The TIL was synthesized in eight steps with an overall acceptable isolated yield of 8.5%, obtained as a red ionic liquid. Due to the presence of two electron-withdrawing groups at the 3- and 6-positions of the 1,2,4,5-tetrazine core, the TIL exhibited high diene reactivity and proved well suited for IEDDA reactions with alkene and alkyne dienophiles. In QCM measurements, successful detections of gaseous alkenes and alkynes were achieved using the TIL thin-coated on quartz chips through rapid IEDDA reactions. To the best of our knowledge, this represents the first report employing IEDDA reactions for the sensitive detection of alkene and alkyne gases using ionic liquid materials on QCM platforms.Author contributions
Yen-Ho Chu: conceptualized the study, secured funding, designed the experiments, interpreted the data, and drafted the manuscript. Chien-Yuan Chen: performed the experiments. Yu-Hsin Chung: contributed to the development of the lab-built QCM device. Yu-Qian Qiu, Wen-Chun Su and Wen-Ting Hsiao: reproduced the synthesis of TIL and optimized the final step of the TIL synthesis. All authors discussed the results and approved the final version of the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
All data supporting the findings of this study are available in the supplementary information (SI). Supplementary information: Fig. S1, 1H and 13C NMR, and high-resolution mass spectrometry (HRMS) spectra and data of 2, 3, 5 and TIL, procedure for QCM measurements, reactions of TIL toward styrene in CH3CN measured by NanoDrop UV-Vis spectrophotometer. See DOI: https://doi.org/10.1039/d5ma01455g.Acknowledgements
This work was supported by grants MOST 109-2113-M-194-005-MY2 (the Ministry of Science and Technology) and NSTC 111-2113-M-194-010-MY2 (National Science and Technology Council), Taiwan, Republic of China.References
- C. I. Cheng, Y.-P. Chang and Y.-H. Chu, Biomolecular interactions and tools for their recognition: Focus on the quartz crystal microbalance and its diverse surface chemistries and applications, Chem. Soc. Rev., 2012, 41, 1947–1971 Search PubMed.
- A. Chang, H.-Y. Li, I.-N. Chang and Y.-H. Chu, Affinity ionic liquids for chemoselective gas sensing, Molecules, 2018, 23, 2380 Search PubMed.
- G. Clavier and P. Audebert, s-Tetrazines as building blocks for new functional molecules and molecular materials, Chem. Rev., 2010, 110, 3299–3314 CrossRef CAS PubMed.
- L. L. Fershtat, Recent advances in the synthesis and performance of 1,2,4,5-tetrazine-based energetic materials, FirePhysChem, 2023, 3, 78–87 CrossRef CAS.
- G. N. Lipunova, E. V. Nosova, G. V. Zyryanov, V. N. Charushina and O. N. Chupakhin, 1,2,4,5-Tetrazine derivatives as components and precursors of photo- and electroactive materials, Org. Chem. Front., 2021, 8, 5182–5205 Search PubMed.
- H. Sun, Q. Xue, C. Zhang, H. Wu and P. Feng, Derivatization based on tetrazine scaffolds: Synthesis on tetrazine derivatives and their biomedical applications, Org. Chem. Front., 2022, 9, 481–498 RSC.
- H. Wu and N. K. Devaraj, Advances in tetrazine bioorthogonal chemistry driven by the synthesis of novel tetrazines and dienophiles, Acc. Chem. Res., 2018, 51, 1249–1259 Search PubMed.
- B. L. Oliveira, Z. Guo and G. J. L. Bernardes, Inverse electron demand Diels-Alder reactions in chemical biology, Chem. Soc. Rev., 2017, 46, 4895–4950 RSC.
- J. Sauer, D. K. Heldmann, J. Hetzenegger, J. Krauthan, H. Sichert and J. Schuster, 1,2,4,5-Tetrazine: Synthesis and reactivity in [4 + 2] cycloadditions, Eur. J. Org. Chem., 1998, 2885–2896 Search PubMed.
- A. Yu, X. He, T. Shen, X. Yu, W. Mao, W. Chi, X. Liu and H. Wu, Design strategies for tetrazine fluorogenic probes for bioorthogonal imaging, Chem. Soc. Rev., 2025, 54, 2984–3016 RSC.
- Y. Pei, Y. Zhang, J. Ma, M. Fan, S. Zhang and J. Wang, Ionic liquids for advanced materials, Mater. Today NANO, 2022, 17, 100159 Search PubMed.
- G. Kaur, H. Kumar and M. Singla, Diverse applications of ionic liquids: A comprehensive review, J. Mol. Liq., 2022, 351, 118556 CrossRef CAS.
- S. Sowmiah, V. Srinivasadesikan, M.-C. Tseng and Y.-H. Chu, On the chemical stabilities of ionic liquids, Molecules, 2009, 14, 3780–3813 CrossRef CAS PubMed.
- C.-W. Chen, M.-C. Tseng, S.-K. Hsiao, W.-H. Chen and Y.-H. Chu, Transimination reactions in [b-3C-im][NTf2] ionic liquid, Org. Biomol. Chem., 2011, 9, 4188–4193 Search PubMed.
- D. L. Boger, J. S. Panek and M. Patel, Preparation and Diels-Alder reaction of a reactive, electron-deficient heterocyclic azadiene: dimethyl 1,2,4,5-tetrazine-3,6-dicarboxylate, Org. Synth., 1992, 70, 79 Search PubMed.
- H.-C. Kan, M.-C. Tseng and Y.-H. Chu, Bicyclic imidazolium-based ionic liquids: synthesis and characterization, Tetrahedron, 2007, 63, 1644–1653 CrossRef CAS.
- P. Thirumurugan, D. Matosiuk and K. Jozwiak, Click chemistry for drug development and diverse chemical–biology applications, Chem. Rev., 2013, 113, 4905–4979 CrossRef CAS PubMed.
- M.-C. Tseng and Y.-H. Chu, Reaction-based azide gas sensing with tailored ionic liquids measured by quartz crystal microbalance, Anal. Chem., 2014, 86, 1949–1952 CrossRef CAS PubMed.
- X. Shang, X. Song, C. Faller, R. Lai, H. Li, R. Cerny, W. Niu and J. Guo, Fluorogenic protein labeling using a genetically encoded unstrained alkene, Chem. Sci., 2017, 8, 1141–1145 Search PubMed.
- F. Liu, Y. Liang and K. N. Houk, Theoretical elucidation of the origins of substituent and strain effects on the rates of Diels–Alder reactions of 1,2,4,5-tetrazines, J. Am. Chem. Soc., 2014, 136, 11483–11493 Search PubMed.
- F. Thalhammer, U. Wallfahrer and J. Sauer, Reaktivität einfacher offenkettiger und cyclischer dienophile bei Diels-Alder-reaktionen mit inversem elektronenbedarf, Tetrahedron Lett., 1990, 31, 6851–6854 Search PubMed.
- F. Liu, R. S. Paton, S. Kim, Y. Liang and K. N. Houk, Diels–Alder reactivities of strained and unstrained cycloalkenes with normal and inverse-electron-demand dienes: activation barriers and distortion/interaction analysis, J. Am. Chem. Soc., 2013, 135, 15642–15649 CrossRef CAS PubMed.
- L. Brandsma and H. D. Verkruijsse, An improved synthesis of cyclooctyne, Synthesis, 1978, 290 CrossRef CAS.
- W. Chen, D. Wang, C. Dai, D. Hamelberg and B. Wang, Clicking 1,2,4,5-tetrazine and cyclooctynes with tunable reaction rates, Chem. Commun., 2012, 48, 1736–1738 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |