Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dual ionic conductivity in Ba3InGa2O7.5: correlating structure and electrochemical properties
Oliver J.
Wagstaff
 *,
Archie D.
Collins
,
John S. O.
Evans
and
Ivana Radosavljević
Evans
*,
Archie D.
Collins
,
John S. O.
Evans
and
Ivana Radosavljević
Evans

Department of Chemistry, Durham University, Lower Mountjoy, Durham DH1 3LE, UK. E-mail: ivana.radosavljevic@durham.ac.uk
First published on 16th March 2026
Abstract
Perovskite-related materials derived from the brownmillerite-type Ba2In2O5 and those with the formula A3OhTd2O7.5 (A = 2+ cation, Oh = octahedral 3+ cation, Td = tetrahedral 3+ cation) are two distinct classes of ionic conductors. We report the synthesis of Ba3InGa2O7.5, a material spanning these families that demonstrates promising electrochemical behaviour. X-ray powder diffraction analysis shows it adopts a monoclinic P2/c structure at room temperature (a = 7.9557(13) Å, b = 5.8762(9) Å, c = 18.2237(3) Å and β = 91.570(1)°) and undergoes a phase transition to space group Cmcm at 600 °C. Humidified variable temperature X-ray powder diffraction and thermogravimetric analysis demonstrate that Ba3InGa2O7.5 can uptake significant amounts of H2O without decomposition when exposed to moisture-rich atmospheres. Complex impedance studies reveal dual ionic conductivity, with either proton or oxide ion transport dominating depending on temperature and experimental conditions. Ba3InGa2O7.5 exhibits the highest total conductivity for an unsubstituted A3OhTd2O7.5-type material.
Introduction
Materials displaying ionic conductivity through proton or oxide ion mobility are important in several developing technologies such as separation membranes, fuel reformers, sensors, supercapacitors and as electrolyte materials for solid oxide fuel cells (SOFCs).1–6 Adoption of SOFCs at large-scale is hindered by the high temperatures (>700 °C) at which conventional electrolyte materials, such as yttria-stabilised zirconia and gadolinia-doped ceria, exhibit sufficient ionic conductivity for use in fabricated devices.7,8 The identification of materials that conduct at intermediate or low temperatures is important for the wider application of these technologies.Various perovskite and perovskite-related materials have been investigated for exhibiting oxide ion, proton, mixed ionic and even triple (O2−/e−/H+) conductivity at different temperatures.9–12 Examples include the proton conducting barium zirconates and cerates (∼300–600 °C) or W6+-doped BaScO2.5 (< 400 °C), oxide ion conducting La0.8Sr0.2Ga0.8Mg0.2O3−δ (∼500–900 °C) and Ba3MoNbO8.5 (500–600 °C), and dual conducting Ba7Nb4MoO20 (300–800 °C).13–20
Brownmillerite-type materials are a perovskite-related structural family that have been studied as both ionic and mixed ionic-electronic conductors depending on composition.21–24 The brownmillerite structure is derived from the cubic perovskite through the replacement of one-sixth of the oxide ions with vacancies. The ordered vacancies produce alternating layers of BO4 and BO6 polyhedra, with individual BO4 tetrahedra connecting to form chains along one direction of the unit cell. At higher temperatures it is possible for these vacancies to disorder to produce first tetragonal and then cubic structures.25 A marked increase in oxide ion conductivity often accompanies the disorder, illustrated by Ba2In2O5.26 The conductivity of Ba2In2O5 sharply increases from a modest σ ∼ 10−3 S cm−1 at 800 °C to 0.1 S cm−1 at ∼940 °C through this transition. Attempts to stabilise the disordered phases to room temperature via partial substitution of In3+ (e.g. Ln3+, Zr4+, Ti4+, Mo5+) have been successful, but are often associated with decreases in maximum conductivity.27–30
Separately, it has been found that Ba2In2O5 converts to a series of hydrated Ba2In2O5(H2O)y (0 < y < 1) orthorhombic phases when annealed in wet atmospheres. These phases exhibit dual protonic-oxide ion below 400 °C.31–36
Investigations of the solid solution Ba2(In1−xGax)2O5 have demonstrated the lowering of the order–disorder transition to <800 °C (x ≤ 0.2), and stabilisation of a disordered cubic phase to room temperature (0.25 ≤ x ≤ 0.45).37–39 The material with an equal amount of In and Ga (x = 0.5) has been found to adopt both the cubic disordered phase and an ordered brownmillerite phase (with octahedral In3+ and tetrahedral Ga3+) depending on the annealing temperature.40 The maximum total conductivity of these materials decreases with Ga3+ content, although all except Ba2InGaO5 exhibit higher conductivity than Ba2In2O5 in the intermediate temperature range (500–800 °C). The conductivity of Ba2InGaO5 is almost two orders of magnitude lower than Ba2In2O5 at 800 °C, a result of the reduced oxygen vacancy mobility induced by the cation ordering.40,41 Additionally, Ba2InGaO5 displays a small amount of proton conductivity (σ ∼ 10−7 S cm−1) below 500 °C.
Only limited work beyond the midpoint of the In1−xGax solid solution range exists, with the hypothetical end member Ba2Ga2O5 reported as thermodynamically unstable with respect to a mixture of BaO and BaGa2O4.42,43 Increasing the Ga-content to x = 2/3 would produce a compound isoformulaic with a structurally-distinct class of perovskite derived ionic conductors: the A3OhTd2O7.5 materials (A = 2+ cation, Oh = octahedral 3+ cation, Td = tetrahedral 3+ cation).
Similarly to the brownmillerites, A3OhTd2O7.5 compounds display varied transport properties dependent on composition, with the donor-doped Ba2.9La0.1YGa2O7.55 exhibiting pure oxide ion conductivity whilst the acceptor-doped Ba1.5Sr1.5YGa1.8Zn0.2O7.4 and Sr3YGa1.8Zn0.2O7.4 display majority proton conductivity in the intermediate temperature region (≤ 800 °C).44–47 Given the superior electrochemical performance of Ba2In2O5-based conductors, it is of interest to expand the Ba2(In1−xGax)2O5 series to x = 2/3 and investigate the properties of Ba3InGa2O7.5. One previous study on this material suggests that it may be a moderate dual ionic conductor (σ ∼ 10−4 S cm−1 at 800 °C).48 However, the measurements reported were carried out on a sample that contained unidentified impurities, warranting further investigation.
In the present work we explore the new A3OhTd2O7.5 ionic conductor Ba3InGa2O7.5 to investigate its electrochemical properties, and report a sample with maximum total conductivity amongst the largest for A3OhTd2O7.5 materials that also exhibits dual oxide ion and proton conductivity. The average structure between room temperature and 1000 °C is studied using X-ray powder diffraction. The conductivity is investigated using electrochemical impedance spectroscopy, and additional information on moisture uptake is gained through thermogravimetric analysis.
Experimental
Sample preparation
Polycrystalline samples of Ba3InGa2O7.5 (∼2 g) were initially prepared from stoichiometric amounts of BaCO3 (Sigma-Aldrich, ≥99.9%), In2O3 (Sigma-Aldrich, 99.99%) and Ga2O3 (Sigma-Aldrich, ≥99.99%) based on the method of Kochetova et al.48 Reagents were mixed and ground together under ethanol, then heated in alumina crucibles for up to 108 h at 1200 °C with a heating and cooling rate of 5 °C min−1. Intermediate regrinding and pelleting were carried out until powder X-ray diffraction patterns of each sample were constant. An off-white powder was obtained. Subsequent syntheses aimed at increasing product purity were modified by adjusting the relative amounts of starting reagents so that 95 wt% of stoichiometric Ga2O3 was used (giving a Ba![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In
In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ga molar ratio in the starting reagents of 3
Ga molar ratio in the starting reagents of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.91).
1.91).
Structural characterisation
Ambient X-ray diffraction data were collected on a Bruker D8 advance powder diffractometer in Bragg–Brentano geometry using Cu Kα radiation and a Lynx-eye detector. Measurements for final Rietveld analysis were collected with a 2θ range of 10° ≤ 2θ ≤ 120°.Variable temperature X-ray diffraction patterns were recorded with an Anton Parr HTK1200 furnace attachment. X-ray diffraction patterns were collected on heating and cooling at 20 °C intervals between 25 and 1000 °C over a 2θ range of 10 ≤ 2θ ≤ 90°. Collection times were 30 minutes at each temperature. A final pattern was collected for 12h at 1000 °C. A further series of measurements were collected under flowing wet nitrogen (pH2O ∼ 0.02 atm) between 25 and 1000 °C with two heating regimes: 20 °C intervals on heating and cooling for 25–500 °C, and 160 °C intervals for 500–1000 °C. Furnace temperatures were calibrated using an external alumina standard. Analysis was carried out via Rietveld refinement implemented in the TOPAS Academic software.49–51
Electrochemical characterisation
For impedance spectroscopy a sample of Ba3InGa2O7.5 was pressed into a 10 mm pellet and sintered to achieve a high density (a pressure of 5 tonnes and sintering at 1200 °C for 50 h achieved a relative density of 85%). Platinum ink (Metalor) was applied to the face of the pellet before mounting on a Probostat A-6 cell placed inside a vertically oriented tube furnace. Platinum wire electrodes were attached to each face and set by heating to 1000 °C for 30 min. Impedance measurements were collected at ∼20 °C intervals on heating and cooling between 20 and 1000 °C at 2 °C min−1 using a Solartron 1260 frequency response analyser in the range of 10−1–107 Hz. Further measurements were carried out under both dry and wet flowing nitrogen (pH2O ∼ 0.02 atm) with multiple cycles of heating and cooling to ensure equilibrium had been achieved. The results were analysed using the ZView/ZPlot Software (Scribner Associates).Thermogravimetric analysis
Mass changes were measured using a PerkinElmer Thermogravimetric Analysis (TGA) 8000 instrument. A powdered sample of Ba3InGa2O7.5 was heated and cooled between 30 and 1000 °C at a rate of 10 °C min−1 under flowing air.Results and discussion
Synthesis
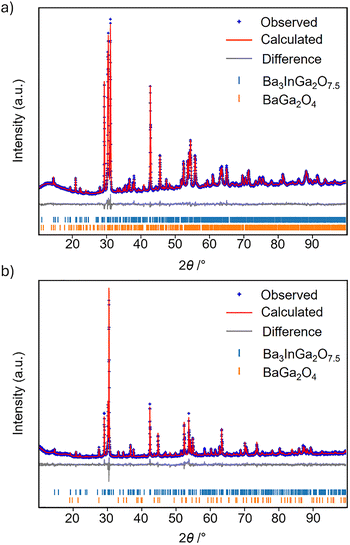
Powder diffraction patterns of samples of Ba3InGa2O7.5 produced using stoichiometric reagent amounts at 1200 °C were similar to those previously reported.48 Fitting the data with the P2/c structural model of Ba3ErGa2O7.552 (Fig. 1) accounted for the majority of the observed peaks. However, two observed weak peaks at 2θ ∼ 28.0 and 33.2° were not predicted by this model (Fig. S1). Instead, these peaks were readily fitted by including BaGa2O4 as a secondary phase that made up ∼10 wt% of the sample. Further heating of this sample for a total period in excess of 100 h did not decrease the BaGa2O4 weight percentage, whilst raising the synthesis temperature to 1300 °C resulted in melting. Our results suggest that the sample of Ba3InGa2O7.5 previously reported was ≤90% pure instead of the single phase claimed. By contrast, our modified synthesis procedure, using 95 wt% of stoichiometric Ga2O3 resulted in a sample with >98 wt% Ba3InGa2O7.5 after heating at 1200 °C for 108 h. This suggests that some Ba- and In-volatisation occurs at the high reaction temperature. | ||
| Fig. 1 View of the P2/c structure of A3OhTd2O7.5 materials (A2+ in grey, Oh3+ in purple, Td3+ in green/orange and O2− in red). | ||
Room temperature structure
The powder X-ray diffraction pattern of Ba3InGa2O7.5 was fitted with the P2/c structural model of Ba3ErGa2O7.5. This is a 12-fold superstructure of the cubic perovskite (2ap × √2ap × 3√2ap) with the octahedra (Oh) and tetrahedra (Td) ordered to give layers of corner-sharing Ga2O7 tetrahedra separated by slabs of [(GaO4)(InO6)]2 (Fig. 1). All peaks were fitted satisfactorily with this model except two low-intensity peaks at 28.0 and 33.2°, which were accounted for by the inclusion of 1.7(2) wt% BaGa2O4.Refinement of the fractional occupancies of the Oh and Td sites confirmed the expected overall In:Ga ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 but indicated that partial In/Ga disorder occurs between the Oh and Td2O7 sites with the Td site in the slab occupied entirely by Ga3+. This is consistent with the bonding preferences of these species, with In3+ and Ga3+ known to adopt both tetrahedral and octahedral geometries.53,54 The use of a model where the total occupancies of In3+ and Ga3+ across the sites was constrained to unity showed no difference compared to one with freely refined occupancies. Final refined occupancies gave an In
2 but indicated that partial In/Ga disorder occurs between the Oh and Td2O7 sites with the Td site in the slab occupied entirely by Ga3+. This is consistent with the bonding preferences of these species, with In3+ and Ga3+ known to adopt both tetrahedral and octahedral geometries.53,54 The use of a model where the total occupancies of In3+ and Ga3+ across the sites was constrained to unity showed no difference compared to one with freely refined occupancies. Final refined occupancies gave an In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ga ratio of 0.86(2)
Ga ratio of 0.86(2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.14(2) for the Oh site and the reverse for the Td site. Inclusion of the partial disorder resulted in a weighted R-factor (Rwp) of 2.130%, compared with 2.152% for the fully ordered structural model. A single isotropic atomic displacement parameter was used for each atom type, except for the partially occupied sites which used an equated value. Some small anisotropic strain broadening was accounted for using the peak shape model described by Stephens.55 Cell parameters of a = 7.9557(13) Å, b = 5.8762(9) Å, c = 18.2237(3) Å and β = 91.570(1)° were obtained with the final fit shown in Fig. 2a. Crystallographic parameters for Ba3InGa2O7.5 are given in Table S1.
0.14(2) for the Oh site and the reverse for the Td site. Inclusion of the partial disorder resulted in a weighted R-factor (Rwp) of 2.130%, compared with 2.152% for the fully ordered structural model. A single isotropic atomic displacement parameter was used for each atom type, except for the partially occupied sites which used an equated value. Some small anisotropic strain broadening was accounted for using the peak shape model described by Stephens.55 Cell parameters of a = 7.9557(13) Å, b = 5.8762(9) Å, c = 18.2237(3) Å and β = 91.570(1)° were obtained with the final fit shown in Fig. 2a. Crystallographic parameters for Ba3InGa2O7.5 are given in Table S1.
 | ||
| Fig. 2 Rietveld refinements against laboratory powder X-ray diffraction pattern of Ba3InGa2O7.5 (a) At room temperature in space group P2/c. (b) At 1000 °C in space group Cmcm. | ||
High temperature structure
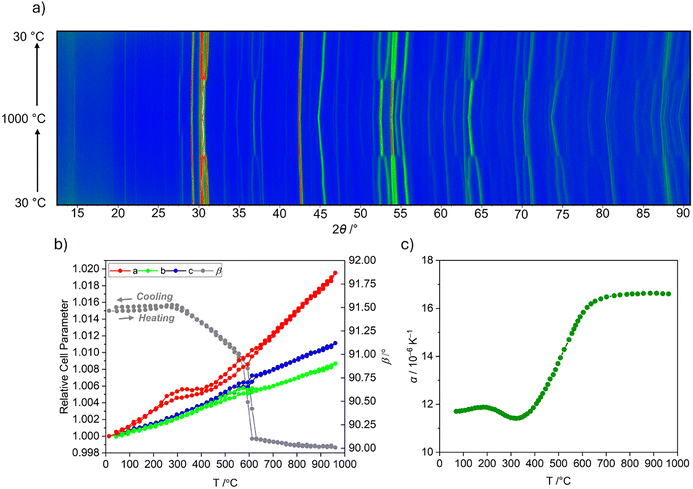
Materials in the A3OhTd2O7.5 family are known to undergo phase transitions as a function of temperature.45–47,52,56 Unit-cell parameters of Ba3InGa2O7.5 between room temperature and 1000 °C were therefore determined from variable temperature X-ray powder diffraction patterns analysed by Rietveld fitting (Fig. 3a). Three distinct regions of cell parameter behaviour can be identified. Between 25 and 400 °C the b- and c-cell axes increase linearly whilst the a-axis initially expands before contracting between 320 and 400 °C (Fig. 3b). Similarly to the a-axis, the monoclinic angle (β) remains approximately constant below 320 °C then begins to decrease. This is likely related to moisture uptake/loss by Ba3InGa2O7.5, as is the case for some other A3OhTd2O7.5 materials.45,46 This process must be rapid and on the timescale of the measurements (∼30 min), as the behaviour is reversible on both heating and cooling. Between 400 and ∼570 °C the three unit-cell axes and the monoclinic angle evolve linearly. From 570 °C the monoclinic angle rapidly decreases to 90° coinciding with subtle discontinuities in the a-, b-, and c-axes, suggesting a monoclinic-to-orthorhombic phase transition. The extracted volumetric thermal expansion coefficient (Fig. 3c) is consistent with this interpretation. There is a small feature around 200–400 °C, caused by moisture uptake and loss as in other compounds.45,46,57 The thermal expansion then increases approximately linearly until the phase transition at 600 °C.Inspection of the diffraction patterns also confirms the presence of the 1.7(2) wt% secondary BaGa2O4 phase. A discontinuity occurs at around 800 °C for the peaks at 2θ ∼28 and 33°, which is associated with the known phase transition from the P63α-phase to the P6322 β-phase (Fig. S2).43,58
Unlike for Ba3YGa2O7.5, no thermal hysteresis is associated with the monoclinic-to-orthorhombic transition temperature of Ba3InGa2O7.5.47 However, the phase transition is characterised by the merging of several reflections including the most intense (21![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) and (213) peaks around 2θ ∼31°. Therefore, one sensible starting model for the high-temperature structure of Ba3InGa2O7.5 is the P21/a model of Ba3YGa2O7.5 that appears orthorhombic due to a monoclinic angle (γ ∼ 107.8°) which transforms to almost exactly 90° in the P2/c setting (Fig. S3a).47,59 An alternative description is the orthorhombic Cmcm model adopted by Ba1.5Sr1.5YGa2O7.5, in which disorder exists along the Td2O7 chains (Fig. S3b).46 Rietveld refinements of both of these models against the highest temperature X-ray diffraction pattern show similar quality fits, with all major peaks accounted for by both models. The Rwp for the fit in P21/a is marginally lower (6.63 vs. 6.90%); however, several weak peaks that are present above the phase transition for Ba3YGa2O7.5 are not evident in the Ba3InGa2O7.5 patterns. (Fig. S4). These peaks are not predicted for the Cmcm model. Accordingly, we assign the Cmcm model to the high-temperature structure of Ba3InGa2O7.5, with cell parameters of a = 5.9251(3) Å, b = 18.4230(7) Å, c = 8.0957(3) Å. Refinement of partial In/Ga occupancies showed a ratio of 0.88(3)
) and (213) peaks around 2θ ∼31°. Therefore, one sensible starting model for the high-temperature structure of Ba3InGa2O7.5 is the P21/a model of Ba3YGa2O7.5 that appears orthorhombic due to a monoclinic angle (γ ∼ 107.8°) which transforms to almost exactly 90° in the P2/c setting (Fig. S3a).47,59 An alternative description is the orthorhombic Cmcm model adopted by Ba1.5Sr1.5YGa2O7.5, in which disorder exists along the Td2O7 chains (Fig. S3b).46 Rietveld refinements of both of these models against the highest temperature X-ray diffraction pattern show similar quality fits, with all major peaks accounted for by both models. The Rwp for the fit in P21/a is marginally lower (6.63 vs. 6.90%); however, several weak peaks that are present above the phase transition for Ba3YGa2O7.5 are not evident in the Ba3InGa2O7.5 patterns. (Fig. S4). These peaks are not predicted for the Cmcm model. Accordingly, we assign the Cmcm model to the high-temperature structure of Ba3InGa2O7.5, with cell parameters of a = 5.9251(3) Å, b = 18.4230(7) Å, c = 8.0957(3) Å. Refinement of partial In/Ga occupancies showed a ratio of 0.88(3)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.12(3) at the octahedral interslab site, consistent with the value obtained at room temperature. The final fit, shown in Fig. 2b with crystallographic parameters in Table S2, gave an Rwp of 6.90%.
0.12(3) at the octahedral interslab site, consistent with the value obtained at room temperature. The final fit, shown in Fig. 2b with crystallographic parameters in Table S2, gave an Rwp of 6.90%.
Moisture uptake
Proton conductivity in oxygen-deficient perovskite-type materials is often driven by moisture uptake, through deliberate exposure during fabrication or testing, or from the ambient environment whilst stored. Uptake during storage is especially important as it can lead to sample degradation limiting electrochemical performance.60–62 Thermogravimetric analysis (TGA) shows that a sample of Ba3InGa2O7.5 exposed to laboratory atmosphere for several days experiences a ∼0.4% mass loss (equivalent to ∼1.7 moles of H2O per Ba3InGa2O7.5 formula unit) by 375 °C on heating (Fig. S5), consistent with the behaviour observed in the variable temperature powder diffraction. The mass then remains approximately constant on heating to 1000 °C and cooling to room temperature. Despite the moisture uptake, Ba3InGa2O7.5 remains stable for extended periods when stored in closed containers.Both the TGA and conductivity exhibited under wet atmospheres (below) demonstrate that Ba3InGa2O7.5 can take up moisture under suitable conditions. To investigate whether Ba3InGa2O7.5 forms a structurally distinct hydrated phase similar to Ba2In2O5, variable temperature diffraction patterns were collected under wet flowing nitrogen. The patterns demonstrate that the P2/c structure persists until the Cmcm phase transition at 600 °C (Fig. S6). The extracted cell parameters are larger for each cell axis below 500 °C under humid conditions relative to those in air (Fig. S7), confirming the uptake of H2O. The a-axis (parallel to the Td2O7 chains) increases by the largest amount, which agrees with previous observations of moisture uptake in Sr3YGa1.8Zn0.2O7.4 and Ba3YGa1.8Zn0.2O7.4.46,47 This suggests that water molecules occupy vacant sites within the Td2O7 layers. These layers have been associated with long range ionic mobility in A3OhTd2O7.5-type materials.46
Conductivity
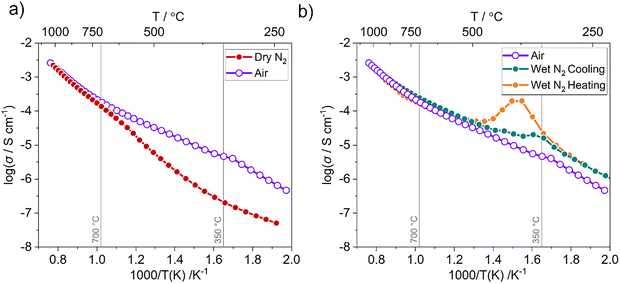
The conductivity of Ba3InGa2O7.5 was measured in air and under flowing dry and wet nitrogen from ∼250 to 1000 °C. The complex plane plots at all temperatures showed two responses, an electrode response at low frequencies and a semicircular one at high frequencies (Fig. S8). The forms of these responses changed with temperature. At lower temperatures (<400 °C), overlapping responses formed a depressed semicircle followed by a 45° spike. At higher temperatures a single semicircular signal was observed, whilst the electrode response tapered into an arc. The evolution of the electrode response is typical for finite Warburg type diffusion and majority ionic conductivity.63,64 The capacitance was extracted from the apex of the arc using the relationship ωRC = 1. This gave a value of ∼10−12 F cm−1, indicating a bulk response.63 The total conductivity at all temperatures was derived from the resistance given by the intercept with the real axis.65Arrhenius plots of the total conductivities are shown in Fig. 4. In air the maximum total conductivity is 2.6 × 10−3 S cm−1. This is an order of magnitude higher than other unsubstituted A3OhTd2O7.5 materials and comparable to the maximum conductivity of the best ionic conductor in this material family, Ba2.9La0.1YGa2O7.55.47 The behaviour is likely due to the presence of In3+ within the Td2O7 chains, which stabilise migrating species through temporary coordination to the metal centre. The conductivity is greater than that reported by Kochetova et al. at comparable temperatures (> 500 °C).48 This could be due to different sample relative density (not stated in that work) but is likely due to the ∼10% BaGa2O4 present in the previous work.
The Arrhenius curve can be separated into three regions with distinct gradients: T > 700 °C, 350 < T < 700 °C, and T < 350 °C. The boundaries of these regions approximately align with the P2/c ↔ Cmcm phase transition and the loss of moisture as determined from TGA. There is no abrupt increase in conductivity at the P2/c ↔ Cmcm phase transition temperature. This suggests that despite the O1 disorder in the Cmcm structural model, individual Td2O7 chains remain locally ordered at high temperature, preserving the –Td–O–Td–□– arrangement. This is consistent with other A3OhTd2O7.5-type conductors.44,45,47,56 Local disorder within these chains would require that some –□–Td–□– units are formed. Such 3-coordinate TdO3 units were not observed in ab initio molecular dynamics simulations.46
Activation energies were determined from plots of ln(σT) against 1/T. Above 700 °C the activation energy was 0.97(2) eV, a value consistent with oxide ions being the majority charge carrier.66–68 Between 400 and 700 °C a proton component due to moisture uptake is likely as the activation energy decreases to 0.58(1) eV. This is confirmed by measurements on a pre-dried sample in dry N2 that showed similar conductivity to that in air above 700 °C (majority oxide ion conductivity), whilst below 700 °C the conductivity was substantially lower with minimal change in gradient.
Fig. 4b compares conductivity data collected in air with data collected under wet N2. A bump was observed on heating under wet N2 between ∼330 and 470 °C, whilst on cooling there is a plateau over the same temperature range (Fig. 4b). The behaviour under wet conditions was reproducible through multiple heating and cooling cycles. The hysteresis of conductivity due to the gain and loss of protons is similar to that for other substituted barium indate systems.29,69,70 On heating the increasing mobility of the protons with temperature is counteracted by their loss after 380 °C, resulting in a decrease in the total number of charge carriers. Beyond ∼700 °C the conductivity under wet N2 converges with the measurements collected under air and dry N2. Approximate proton transport numbers were calculated from the data measured under wet and dry N2 across the measured temperature range (Fig. 5) via tH = (σwet−σdry)/σwet. These confirm that protons dominate conductivity at lower temperatures and remain the majority charge carrier until 700 °C. By 1000 °C the conductivity is almost entirely oxide-derived in nature (tH < 0.09). The low temperature moisture uptake is also presumably responsible for the slight bump in the conductivity measured in air at ∼350 °C, due to small amounts of atmospheric moisture.
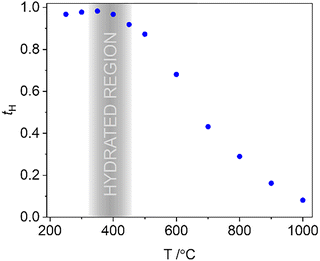 | ||
| Fig. 5 Proton transport numbers calculated from the total conductivity in wet and dry N2 of Ba3InGa2O7.5. | ||
The conductivity of Ba3InGa2O7.5 is lower than that for the Ba2(In1−xGax)2O5 materials with 0.1 ≤ x ≤ 0.45 between 500 and 1000 °C.37–39,41 This is likely the result of the persistence of oxide vacancy ordering at high temperatures. However, the conductivity is comparable to Ba2In2O5 in the brownmillerite temperature region (<800 °C) and significantly higher than that reported for Ba2InGaO5.26,40
Conclusions
Ba3InGa2O7.5 was prepared by conventional solid-state synthesis. X-ray diffraction analysis indicates that it adopts the monoclinic P2/c structure of other Ba-containing A3OhTd2O7.5 materials at room temperature instead of the orthorhombic brownmillerite or cubic perovskite structures of other members of the Ba2(In1−xGax)2O5 solid solution. Despite there being partial In/Ga disorder, refinements show that the In:Ga ratio is the targeted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. Variable temperature powder diffraction studies show a reversible discontinuous monoclinic-to-orthorhombic phase transition at 600 °C and measurable moisture uptake below 400 °C; this was confirmed by thermogravimetric analysis. Despite the moisture uptake, Ba3InGa2O7.5 remains stable when exposed to deliberately humidified atmospheres and can be stored without decomposition for periods of at least several weeks.
2. Variable temperature powder diffraction studies show a reversible discontinuous monoclinic-to-orthorhombic phase transition at 600 °C and measurable moisture uptake below 400 °C; this was confirmed by thermogravimetric analysis. Despite the moisture uptake, Ba3InGa2O7.5 remains stable when exposed to deliberately humidified atmospheres and can be stored without decomposition for periods of at least several weeks.
Impedance spectroscopy shows Ba3InGa2O7.5 to be a dual ionic conductor, exhibiting both oxide ion and proton conductivity. The maximum total conductivity is the highest reported for an unsubstituted A3OhTd2O7.5 conductor (2.6 × 10−3 S cm−1). The atmospheric moisture content used for the experiments has a significant impact on the conductivity. Below 400 °C proton conductivity dominates under humidified conditions, reaching a maximum of 2 × 10−4 S cm−1. Dual oxide ion and proton conductivity exists between 400 and ∼750 °C, whilst oxide ion conductivity dominates at the highest temperatures, as well as under dry conditions.
Overall, the conductivity behaviour reflects a crossover from proton to oxide ion transport. This leads to Ba3InGa2O7.5 having the highest total conductivity of any A3OhTd2O7.5-type conductor at intermediate temperatures, in addition to remaining competitive at higher temperatures.
Conflicts of interest
There are no conflicts to declare.Data availability
Additional data supporting this article are included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01495f.Acknowledgements
The authors thank Durham University for a Durham Doctoral Studentship (DDS) for OJW and for research leave (IRE and JSOE) during which this article was written. We also thank Gary Oswald and Doug Carswell for assistance with VT-PXRD and TGA.References
- N. N. M. Tahir, N. A. Baharuddin, A. A. Samat, N. Osman and M. R. Somalu, J. Alloys Compd., 2022, 894, 162458 CrossRef CAS.
- P. Colomban, Solid State Ion., 2019, 334, 125–144 CrossRef CAS.
- P. J. Megía, A. J. Vizcaíno, J. A. Calles and A. Carrero, Energy Fuels, 2021, 35, 16403–16415 CrossRef.
- L. Malavasi, C. A. J. Fisher and M. S. Islam, Chem. Soc. Rev., 2010, 39, 4370 RSC.
- J. B. Goodenough, Nature, 2000, 404, 821–823 CrossRef CAS PubMed.
- D. J. L. Brett, A. Atkinson, N. P. Brandon and S. J. Skinner, Chem. Soc. Rev., 2008, 37, 1568 RSC.
- D. Pérez-Coll, E. Sánchez-López and G. C. Mather, Solid State Ion., 2010, 181, 1033–1042 CrossRef.
- J. P. Goff, W. Hayes, S. Hull, M. T. Hutchings and K. N. Clausen, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 14202–14219 CrossRef CAS.
- M. Papac, V. Stevanović, A. Zakutayev and R. O’Hayre, Nat. Mater., 2021, 20, 301–313 CrossRef CAS PubMed.
- Y. Zhang, R. Knibbe, J. Sunarso, Y. Zhong, W. Zhou, Z. Shao and Z. Zhu, Adv. Mater., 2017, 29, 1700132 CrossRef PubMed.
- S. Fop, J. Mater. Chem. A, 2021, 9, 18836–18856 RSC.
- X. Yang, A. J. Fernández-Carrión and X. Kuang, Chem. Rev., 2023, 123, 9356–9396 CrossRef CAS PubMed.
- L. Yang, S. Wang, K. Blinn, M. Liu, Z. Liu, Z. Cheng and M. Liu, Science, 2009, 326, 126–129 CrossRef CAS PubMed.
- K. Saito, K. Umeda, K. Fujii, K. Mori and M. Yashima, J. Mater. Chem. A, 2024, 12(22), 13310–13319 RSC.
- M. Morales, J. J. Roa, J. Tartaj and M. Segarra, J. Eur. Ceram. Soc., 2016, 36, 1–16 CrossRef CAS.
- S. Fop, J. M. S. Skakle, A. C. McLaughlin, P. A. Connor, J. T. S. Irvine, R. I. Smith and E. J. Wildman, J. Am. Chem. Soc., 2016, 138, 16764–16769 CrossRef CAS PubMed.
- J. E. Auckett, K. L. Milton and I. R. Evans, Chem. Mater., 2019, 31, 1715–1719 CrossRef CAS.
- S. Fop, K. S. McCombie, E. J. Wildman, J. M. S. Skakle, J. T. S. Irvine, P. A. Connor, C. Savaniu, C. Ritter and A. C. Mclaughlin, Nat. Mater., 2020, 19, 752–757 CrossRef CAS PubMed.
- M. Yashima, T. Tsujiguchi, Y. Sakuda, Y. Yasui, Y. Zhou, K. Fujii, S. Torii, T. Kamiyama and S. J. Skinner, Nat. Commun., 2021, 12, 556 CrossRef CAS PubMed.
- T. Ishihara, H. Matsuda and Y. Takita, J. Am. Chem. Soc., 1994, 116, 3801–3803 CrossRef CAS.
- F. Ramezanipour, J. E. Greedan, A. P. Grosvenor, J. F. Britten, L. M. D. Cranswick and V. O. Garlea, Chem. Mater., 2010, 22, 6008–6020 CrossRef CAS.
- P. Jiang, J. Li, A. Ozarowski, A. W. Sleight and M. A. Subramanian, Inorg. Chem., 2013, 52, 1349–1357 CrossRef CAS PubMed.
- C. A. Fuller, Q. Berrod, B. Frick, M. R. Johnson, S. J. Clark, J. S. O. Evans and I. R. Evans, Chem. Mater., 2019, 31, 7395–7404 CrossRef CAS PubMed.
- C. A. Fuller, Q. Berrod, B. Frick, M. R. Johnson, M. Avdeev, J. S. O. Evans and I. R. Evans, Chem. Mater., 2020, 32, 4347–4357 CrossRef CAS.
- S. A. Speakman, J. W. Richardson, B. J. Mitchell and S. T. Misture, Solid State Ion., 2002, 149, 247–259 CrossRef CAS.
- J. B. Goodenough, J. E. Ruiz-Diaz and Y. S. Zhen, Solid State Ion., 1990, 44, 21–31 CrossRef CAS.
- T. Q. Ta, T. Tsuji and Y. Yamamura, J. Alloys Compd., 2006, 408–412, 253–256 CrossRef CAS.
- P. Berastegui, S. Hull, F. J. García-García and S.-G. Eriksson, J. Solid State Chem., 2002, 164, 119–130 CrossRef CAS.
- V. Jayaraman, A. Magrez, M. Caldes, O. Joubert, M. Ganne, Y. Piffard and L. Brohan, Solid State Ion., 2004, 170, 17–24 CrossRef CAS.
- A. Rolle, R. N. Vannier, N. V. Giridharan and F. Abraham, Solid State Ion., 2005, 176, 2095–2103 Search PubMed.
- G. B. Zhang and D. M. Smyth, Solid State Ion., 1995, 82, 153–160 CrossRef CAS.
- T. Schober, J. Friedrich and F. Krug, Solid State Ion., 1997, 99, 9–13 Search PubMed.
- W. Fischer, G. Reck and T. Schober, Solid State Ion., 1999, 116, 211–215 Search PubMed.
- M. Karlsson, A. Matic, C. S. Knee, I. Ahmed, S. G. Eriksson and L. Börjesson, Chem. Mater., 2008, 20, 3480–3486 CrossRef CAS.
- A. Perrichon, M. M. Koza, Z. Evenson, B. Frick, F. Demmel, P. Fouquet and M. Karlsson, Chem. Mater., 2023, 35, 6713–6725 CrossRef CAS.
- A. Perrichon, M. Jiménez-Ruiz, L. Mazzei, S. M. H. Rahman and M. Karlsson, J. Mater. Chem. A, 2019, 7, 17626–17636 RSC.
- H. Yamamura, Y. Yamada, T. Mori and T. Atake, Solid State Ion., 1998, 108, 377–381 CrossRef CAS.
- Y. Uchimoto, M. Kinuhata, H. Takagi, T. Yao, T. Inagaki and H. Yoshida, Proc. Vol., 1999, 1999–19, 317 CrossRef.
- T. Yao, Y. Uchimoto, M. Kinuhata, T. Inagaki and H. Yoshida, Solid State Ion., 2000, 132, 189–198 CrossRef CAS.
- C. Didier, J. Claridge and M. Rosseinsky, J. Solid State Chem., 2014, 218, 38–43 CrossRef CAS.
- H. Yamamura, H. Hamazaki, K. Kakinuma, T. Mori and H. Haneda, J. Korean Phys. Soc., 1999, 35, 200 Search PubMed.
- C. E. Mohn, N. L. Allan and S. Stølen, Solid State Ion., 2006, 177, 223–228 CrossRef CAS.
- V. Kahlenberg and C. Weidenthaler, Solid State Sci., 2002, 4, 963–968 CrossRef CAS.
- K. Saito, K. Fujii and M. Yashima, J. Solid State Chem., 2022, 306, 122733 CrossRef CAS.
- C. A. Fuller, J. I. Murrell, D. A. Blom, T. Vogt, W. Zhang, P. S. Halasyamani, I. R. Evans and J. S. O. Evans, Chem. Mater., 2022, 34, 3185–3196 CrossRef CAS PubMed.
- O. J. Wagstaff, M. Avdeev, S. J. Clark, J. S. O. Evans and I. R. Evans, Chem. Mater., 2025, 37, 3492–3503 CrossRef CAS.
- C. A. Fuller, D. A. Blom, T. Vogt, I. R. Evans and J. S. O. Evans, J. Am. Chem. Soc., 2022, 144, 615–624 CrossRef CAS PubMed.
- N. Kochetova, V. Cherepanova, A. Pikalova and A. Gilev, Chim. Techno Acta, 2024, 11, 202411102 CrossRef CAS.
- A. A. Coelho, J. Evans, I. Evans, A. Kern and S. Parsons, Powder Diffr., 2011, 26, S22–S25 CrossRef CAS.
- R. E. Dinnebier, A. Leineweber and J. S. O. Evans, Rietveld Refinement: Practical Powder Diffraction Pattern Analysis using TOPAS, De Gruyter STEM, 2018, vol. 1 Search PubMed.
- H. M. Rietveld, J. Appl. Crystallogr., 1969, 2, 65–71 CrossRef CAS.
- A. M. Abakumov, R. V. Shpanchenko, O. I. Lebedev, G. Van Tendeloo, S. Amelinckx and E. V. Antipov, Acta Crystallogr. A, 1999, 55, 828–839 CrossRef PubMed.
- D. Waroquiers, X. Gonze, G.-M. Rignanese, C. Welker-Nieuwoudt, F. Rosowski, M. Göbel, S. Schenk, P. Degelmann, R. André, R. Glaum and G. Hautier, Chem. Mater., 2017, 29, 8346–8360 CrossRef CAS.
- R. D. Shannon, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- P. W. Stephens, J. Appl. Crystallogr., 1999, 32, 281–289 CrossRef CAS.
- R. Simura, Y. Suzuki and H. Yamane, Acta Crystallogr., Sect. C: Struct, Chem., 2023, 79, 464–471 CrossRef CAS PubMed.
- I. R. Evans, J. A. K. Howard and J. S. O. Evans, Chem. Mater., 2005, 17, 4074–4077 CrossRef CAS.
- V. Kahlenberg, R. X. Fischer and J. B. Parise, J. Solid State Chem., 2000, 154, 612–618 CrossRef CAS.
- R. A. Tamazyan, Sov. Phys. - Crystallogr., 1987, 32, 519 Search PubMed.
- J. Nielsen and M. Mogensen, Solid State Ion., 2011, 189, 74–81 CrossRef CAS.
- Y. L. Huang, C. Pellegrinelli and E. D. Wachsman, J. Electrochem. Soc., 2015, 163, F171 CrossRef.
- J. Nielsen, A. Hagen and Y. L. Liu, Solid State Ion., 2010, 181, 517–524 CrossRef CAS.
- J. T. S. Irvine, D. C. Sinclair and A. R. West, Adv. Mater., 1990, 2, 132–138 CrossRef CAS.
- A. C. Lazanas and M. I. Prodromidis, ACS Meas. Sci. Au, 2023, 3, 162–193 CrossRef CAS PubMed.
- M. Diaz-Lopez, J. F. Shin, M. Li, M. S. Dyer, M. J. Pitcher, J. B. Claridge, F. Blanc and M. J. Rosseinsky, Chem. Mater., 2019, 31, 5742–5758 CrossRef CAS PubMed.
- W. Zhang, K. Fujii, T. Ishiyama, H. Kandabashi and M. Yashima, J. Mater. Chem. A, 2020, 8, 25085–25093 RSC.
- Z. Cheng, Y. An, R. Wang, J. C. Neuefeind, X. Yang, R. Cong, T. Yang and P. Jiang, Inorg. Chem., 2024, 63, 22022–22032 CrossRef CAS PubMed.
- J. T. S. Irvine, J. W. L. Dobson, T. Politova, S. García Martín and A. Shenouda, Faraday Discuss., 2007, 134, 41–49 RSC.
- A. Manthiram, J. F. Kuo and J. B. Goodenough, Solid State Ionics, 1993, 62, 225–234 CrossRef CAS.
- D. Tyagi, A. N. Shirsat, B. Saha and S. Varma, J. Alloys Compd., 2021, 877, 160298 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |