Open Access Article
Open Access ArticleTailoring carbon nanomaterial architectures for CO2 capture: structure–property relationships, surface engineering, and future perspectives
Jagdeep
Singh†
 *a,
Ashish
Gupta†
*a,
Ashish
Gupta†
 b,
Zhiqi
Zhu
b,
Zhiqi
Zhu
 c,
Santosh K.
Tiwari
c,
Santosh K.
Tiwari
 d and
A. S.
Dhaliwal
e
d and
A. S.
Dhaliwal
e
aDepartment of Physics and Materials Science, Thapar Institute of Engineering and Technology, Patiala, 147004, Punjab, India. E-mail: jagdeep.singh1@thapar.edu; Tel: +91-84279-04455
bDepartment of Physics, National Institute of Technology, Kurukshetra, Haryana 136119, India
cSchool of Materials Science and Engineering, Central South University, Changsha 410083, China
dCentre for New Materials and Surface Engineering, Department of Chemistry, NMAM Institute of Technology, Nitte (Deemed to be University), Nitte, 574110, Karnataka, India
eDepartment of Physics, Sant Longowal Institute of Engineering and Technology, Longowal, 148106, Punjab, India
First published on 8th January 2026
Abstract
The rapid rise in atmospheric CO2 concentrations, now exceeding 420 ppm, necessitates the urgent deployment of scalable carbon capture, utilization, and storage (CCUS) technologies to mitigate global warming. This review offers a comprehensive analysis of carbon nanomaterials (CNMs) as a transformative class of adsorbents, providing a sustainable alternative to energy-intensive amine scrubbing. CNMs span all dimensional regimes, ranging from zero-dimensional (0D) fullerenes and carbon dots, through one-dimensional (1D) carbon nanotubes and two-dimensional (2D) graphene, to three-dimensional (3D) hierarchical foams, which exhibit exceptional physicochemical properties, notably high specific surface areas and highly tunable pore architectures. Various surface engineering approaches, including tuning surface chemistry and pore architecture and heteroatom functionalization, have been explored to enhance adsorption capacity and selectivity, as well as enable multiple regeneration cycles. Through structure–property–performance analysis, it has been concluded that ultra-micropores (<0.7 nm) are favorable, which further enhance adsorption capacity at low pressures, the isosteric heat of adsorption (35–50 kJ mol−1), and cycling stability. Furthermore, the surface modification of CNMs through nitrogen doping, amine functionalization, and hybrid composite engineering achieved CO2 adsorption capacities of up to ∼9 mmol g−1 at modest pressures, along with low-temperature regeneration (<100 °C), resulting in energy-efficient performance. This article also outlines ongoing challenges and research frontiers, emphasizing the need to enhance the CO2/N2 selectivity ratio, develop sustainable and scalable synthesis methods, incorporate techno-economic evaluations, and bridge laboratory-scale performance with industrial implementation. Later, a comparative analysis of the modified CNMs with standardized MOFs in terms of capacity is also discussed in detail. This analysis synthesizes current advancements and identifies knowledge gaps, offering a prospective outlook on the development and future trajectories of CNM-based adsorbents in greenhouse gas mitigation and achieving net-zero emission targets.
1. Introduction
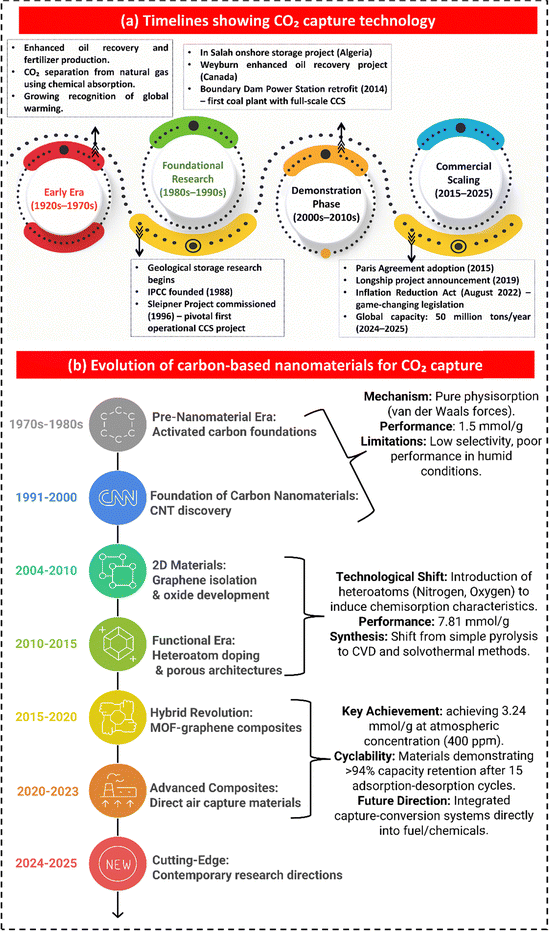
The global energy needs are still mainly met by the burning of fossil fuels, which make up 65–70% of the world's energy use.1–4 The substantial reliance on carbon-intensive energy sources, including power plants, biomass, and bioenergy, has led to an increase in CO2 emissions, resulting in atmospheric CO2 concentrations exceeding 420 ppm.5–7 Such elevated greenhouse gas concentrations have intensified global warming, as reflected in an average rise in the Earth's surface temperature of ∼0.06 °C, as shown in Fig. 1(a), underscoring an accelerating climate inequality driven by anthropogenic activities.8,9 According to the Intergovernmental Panel on Climate Change (IPCC), limiting global warming to below 2 °C, preferably to 1.5 °C above pre-industrial levels, requires not only significant reduction in emissions but also the active removal of CO2 from the atmosphere.10–13Among the portfolio of negative emission technologies, carbon capture, utilisation, and storage (CCUS) has emerged as a foundation strategy for deep decarbonization.14–17 CCUS integrates the capture of CO2 from point sources or the atmosphere with either its conversion into value-added products or its permanent isolation via long-term geological sequestration, thereby enabling substantial reduction in greenhouse gas emissions and supporting net-zero or even net-negative carbon trajectories.18–23 Reflecting its growing strategic importance, the global CCUS sector has expanded rapidly across all stages of development as shown in Fig. 1(d and e).24 As of July 2025, 77 facilities were operational worldwide, with a combined capture capacity of 64 Mtpa. The broader project pipeline included 734 facilities with a total capacity of 513 Mtpa. Notably, capacity at the FEED stage increased from 180 to 262 Mtpa, while 44 Mtpa was under active construction, signalling a clear shift toward large-scale deployment. Keeping the importance in mind, researchers marked a global surge in publications on CCUS, as depicted in Fig. 1(b), which highlights the country-wise distribution and temporal evolution of research in this domain.
Various materials, including liquid sorbents (chemical solvents such as amines, monoethanolamine, and diethanolamine), solid adsorbents (alumina, zeolites, and metal–organic frameworks (MOFs)), and membranes (e.g., polysulfone and polyimides), are being utilised in CCUS. Among these, CNMs have emerged as strong alternatives to the materials mentioned above for CO2 capture due to their exceptionally high surface area, superior structural engineering, lower energy demands for regeneration, enhanced operational flexibility, and cost-effectiveness.25–27 Unlike zeolites with rigid topologies and limited performance, they offer a useful platform where pore structures and functionalities can be precisely engineered for diverse capture needs.28–31 Their performance advantages are evident across key parameters, including surface areas of up to 2688 m2 g−1, a CO2 uptake of ∼8–9 mmol g−1, and superior adsorption capacity.32,33 They also achieve exceptional CO2/N2 selectivity and enable low-energy regeneration at temperatures below 100 °C, compared to 300–500 °C required for zeolites. Additionally, their tolerance to humidity levels below 5% contributes to increased reliability in practical applications.34,35 These materials exhibit superior cycling stability and effective thermal conductivity, facilitating efficient heat management and ensuring reliable long-term performance. These attributes lead to energy savings, lower operational costs, and improved process stability. CNMs demonstrate cost-effectiveness and scalability due to their ability to be synthesised from readily available precursors, including biomass and polymers.36 Their customizable pore structures and functional groups enable the tailoring of adsorption energetics and kinetics, while their chemical inertness and mechanical strength ensure that they can withstand harsh industrial environments.
Researchers have explored a broad spectrum of CNMs with different dimensionalities, ranging from 0D to 3D, for CCUS.37–43 The dimensionality of these materials fundamentally governs key performance parameters, such as specific surface area, pore size distribution, surface functionality, diffusion pathways, and adsorption energetics. While 0D carbons (carbon dots and fullerenes) lack intrinsic porosity and contribute mainly through surface functionalization and 1D carbons (CNTs) suffer from bundling-induced porosity loss and diffusion limitations, 2D materials (graphene, GO, and rGO) offer improved surface accessibility and CO2/N2 selectivity. Still, they are constrained by irreversible restacking and moisture sensitivity. In contrast, 3D hierarchical carbon architectures effectively combine ultra-micropores for strong CO2 confinement with meso- and macroporous networks for rapid mass transport, resulting in superior adsorption capacity, selectivity, and low regeneration energy. Consequently, although lower-dimensional carbons provide valuable chemical tunability, 3D hierarchical CNMs emerge as the most promising platforms for scalable and industrially relevant CO2 capture, particularly under flue gas and direct air capture (DAC) conditions.
Advanced functionalization techniques, including nitrogen doping, surface oxidation, and amine grafting, are extensively employed to improve CO2 capture in CNM-based adsorbents by modifying surface chemistry and adsorption energetics.44–46 Nitrogen doping introduces Lewis basic sites, which enhance CO2 interactions, thereby improving capacity, selectivity, and cycling stability.47 The controlled surface oxidation enhances CO2 selectivity, although excessive oxidation increases the sensitivity to moisture.32 Amine functionalization enables the highest CO2 uptake through reversible carbamate formation.48,49 However, it is constrained by pore blockage and stability challenges at elevated loadings.50 Consequently, synergistic combination of heteroatom doping with moderate oxidation or reduced amine loading on hierarchical carbon frameworks offers the most balanced and scalable CCUS performance.
In response to international climate agreements, numerous governments have made CCUS a key component of their plans to reduce carbon emissions. The US has endorsed its commitment to the Paris Agreement and plans to reach net-zero emissions by 2050.51 The federal government is investing a significant amount of money in the research and development of CCUS. China, which releases the most carbon dioxide into the atmosphere, aims to be carbon neutral by 2060 and has begun constructing large-scale collection facilities.51 India aims to reduce the emission intensity of its GDP by 45% by 2030 and achieve net-zero emissions by 2070. The emphasis will be on biomass-derived carbon sources, including biochar.51 The European Union, Japan, and South Korea also aim to accelerate the adoption of CCUS by implementing stringent rules and regulations.51
It is prominent that there has been an exponential increase in research papers related to CNM-based materials for CO2 collection and conversion in recent years, as illustrated in Fig. 1(b). This increase suggests that an increasing number of academics and businesses are interested in developing multifunctional absorbents that can effectively capture CO2 and convert it into valuable products, such as fuels, chemicals, and construction materials.52–54 However, turning new ideas from the lab into business remains a significant challenge. Some of the main problems are improving pore structures for selective CO2 uptake at low partial pressures, enhancing the durability of materials in real-world conditions (such as moisture and temperature fluctuations), reducing the costs of synthesis and regeneration, and improving cycling stability over time.55 A significant obstruction is the lack of a holistic structure–property–performance paradigm, which hinders the predictive design of materials and their scalability.52,56 Additionally, a systems-level approach is necessary for successful integration into industrial processes.38 This approach includes process engineering, techno-economic analysis, and life cycle evaluation. It is essential to determine how much these materials can genuinely benefit the environment and how much carbon they emit throughout their entire life cycle.
The number of review articles on CCUS in the literature has grown substantially, presenting a chronological overview of various reviews published over the past four years, along with their primary focus areas, as shown in Table 1.
| Year | Focus of the article | Source |
|---|---|---|
| 2021 | Critical technology review: an in-depth update on post-combustion technologies (absorption, adsorption, membrane, and cryogenic), emphasizing hybrid systems and future directions for decarbonization | Raganati et al.57 |
| 2021 | Adsorption-based post-combustion capture: a focused review on adsorption technologies, specifically evaluating adsorbent materials (activated carbons, zeolites, and MOFs), gas–solid contacting systems (fixed/fluidized beds), and regeneration strategies (PSA and TSA) | Raganati et al.58 |
| 2022 | Amine-functionalized solid materials: solid porous supports (silica, carbons, MOFs, and polymers), comparing their CO2 adsorption capacities and regeneration performance | Hack et al.59 |
| 2022 | Energy sector applications: a review of capture technologies (pre-combustion, oxy-fuel, and post-combustion) explicitly applied to energy sectors, with a particular focus on thermoelectric plants and bioenergy (ethanol plants). | Sílvio et al.60 |
| 2023 | 20-year reflective: an examination of CO2 capture methods developed over the last two decades, covering absorption, gas–solid reactions, adsorption, cryogenic processes, membrane processes, and natural inclusion | Kammerer et al.61 |
| 2023 | Process intensification: a review focusing on novel technologies to reduce costs in post-combustion capture, including rotating packed beds, loop reactors, mop fans, and advanced solvent developments | Joel et al.62 |
| 2023 | Power plant scenarios: an assessment identifying post-combustion capture as the best option for retrofitting power plants and proposing future scenarios such as ultrasound regeneration and synergistic solvent effects | Alalaiwat et al.63 |
| 2024 | Capture and mineral storage: a review linking CO2 capture (liquid, solid, microencapsulated) directly with mineral storage solutions (in situ and ex situ mineralization) | Liu et al.64 |
| 2024 | Hybrid configurations: a review of process developments in post-combustion capture from power plant flue gases, identifying combined two-stage hybrid configurations as the optimal route for efficiency and purity | Obi et al.65 |
| 2024 | Environmental assessment: a comprehensive review of direct and indirect capture methods (including reforestation) with a strong focus on environmental impact assessment (e.g., global warming potential) and economic sustainability | Goren et al.66 |
Previously published articles in the literature provide insight into process-level engineering, offering detailed technological summaries. However, these studies offer limited specific information about materials. Along with zeolites and MOFs, CNM-based adsorbents such as activated carbon, biochar, CNTs, MWCNTs, fullerenes, GO, rGO, and carbon dots are emerging nanomaterials with structural diversity and tunability. The existing literature reveals a significant gap in the absence of a dimensionality-driven (0D–3D) framework that systematically correlates diffusion behaviour, adsorption kinetics, and CO2 affinity with the underlying architecture of CNMs. Furthermore, the integration of capture-to-utilization remains underexplored, particularly in existing reviews, and is notably absent in the catalytic conversion of captured CO2 using metal-free carbon-based catalysts, which are predominantly focused on capture or storage alone. Despite their importance for low-pressure and DAC applications, key mechanistic aspects, including the role of ultra-microporosity (<0.7 nm), optimal adsorption enthalpy (35–50 kJ mol−1), and heteroatom-induced Lewis acid–base interactions, remain insufficiently explored. Moreover, many existing reviews continue to rely on pre-2020 paradigms, overlooking recent advances in functionalized, hybrid CNMs.
Hence, by addressing these gaps, the present review adopts a materials science-centric perspective that explores the fundamentals and current advancements in CNM-based materials for CO2 capture, with a focus on nanostructured carbon families (0D to 3D). We have extensively studied their structural, surface, morphological, and functional attributes and connected these behaviors with CO2 adsorption efficacy. We also discussed more advanced techniques for modifying materials, such as physical activation, heteroatom doping, and surface functionalization, and gained an understanding of the mechanisms that improve CO2 capture efficiency. Moreover, in real-world situations, the detailed comparison of different CNMs for CO2 capture performance has been explored. The complicated challenges and their portable solutions are also discussed in detail to align with global decarbonization objectives. This approach positions CNMs as versatile platforms for global carbon dioxide management and next-generation CCUS technologies.
2. CCUS: a historical perspective and a key climate mitigation strategy
The foundations of CO2 capture and climate science were laid as early as 1896, when Svante Arrhenius published the first quantitative study linking atmospheric CO2 levels to global temperature increases.67 Later, in the 1920s and 1930s, industrial interests arose around this issue and secured the first patents focused on removing CO2 from natural gas streams. A breakthrough 1930 patent on amine-based chemical absorption, which laid the groundwork for modern CO2 capture technologies, led to the first large-scale use of captured CO2 in the 1970s. In 1972, the world's first commercial enhanced oil recovery (EOR) project (SACROC) using injected CO2 was developed in Texas. In 1980, pilot-scale studies scientifically validated the feasibility of injecting CO2 into geological formations. In 1988, the Intergovernmental Panel on Climate Change (IPCC) was established to address the challenge of reducing CO2 emissions.68,69 The timelines for CO2 capture, utilization, and storage technologies are shown in Fig. 2(a). In 1996, the Sleipner Project in Norway became the world's first industrial-scale CCUS operation, successfully storing CO2 in deep saline aquifers and proving that large-scale climate mitigation was technically achievable. Between 2000 and 2008, global R&D efforts, such as the Salah storage site in Algeria, significantly advanced capture, transport, and storage technologies. Globally, CCUS projects store over 45 million tons of carbon dioxide, equivalent to the emissions of approximately 10 million passenger vehicles, every year. The International Energy Agency (IEA) predicts that CCUS may contribute to a 17% reduction in CO2 emissions by 2025.From a materials perspective, the field of CNM-based adsorbents for CO2 capture has undergone a significant transformation from 1985 to 2025, as shown in Fig. 2(b), transitioning from basic activated carbons, which primarily rely on weak physisorption, to sophisticated, multifunctional systems designed for low-energy and DAC. Initial activated carbons, while chemically stable, exhibited restricted uptake (<1.5 mmol g−1), thereby underscoring the significance of ultramicroporosity. The discovery of CNTs and fullerenes in the 1990s introduced nanoscale carbon architectures, thereby illuminating the impact of dimensionality on adsorption processes.70 Furthermore, the isolation of graphene and the subsequent development of graphene oxide between 2004 and 2010 facilitated the creation of atomically thin, functionalized surfaces with enhanced CO2 affinity, despite the performance limitations imposed by restacking.71 The period from 2010 to 2015 witnessed further progress, particularly in heteroatom doping and hierarchical porosity, marking a functional era and resulting in record capacities. Based on research spanning from 2015 to 2020, especially on carbon–MOF composites, it was found that these composites significantly improved both adsorption capacity and kinetics, achieving values as high as 9.02 mmol g−1 (GrO@HKUST-1 composites) and simultaneously enhancing stability and regenerability.72 Subsequently, from 2020 onwards, CNMs have been re-engineered for DAC. The comparative analysis of key performance metrics for CO2 capture materials is highlighted in Table 2.57,59,73–84
| Metric | Year | |||||
|---|---|---|---|---|---|---|
| 1990 | 2000 | 2010 | 2015 | 2020 | 2025 | |
| CO2 capacity (mmol g−1) | <1 | 0.8–1.2 | 2–3 | 4–5 | 6–8 | 9+ |
| Energy (kJ mol−1) | 150–185 | 120–160 | 60–100 | 40–50 | 25–40 | 12–20 |
| CO2/N2 selectivity | 2–5 | 3–8 | 10–25 | 30–50 | 60–85 | 100–150 |
| Cycle time | Hours | Hours | 30–60 min | 10–30 min | 5–15 min | <5 min |
| Thermal stability | Limited | Moderate | Good | Good-excellent | Excellent | Excellent |
| Humidity tolerance | Poor | Poor | Moderate | Good | Excellent | Excellent |
| Cost ($ per ton CO2) | >500 | 300–500 | 100–200 | 80–150 | 150–300 | 100–300 |
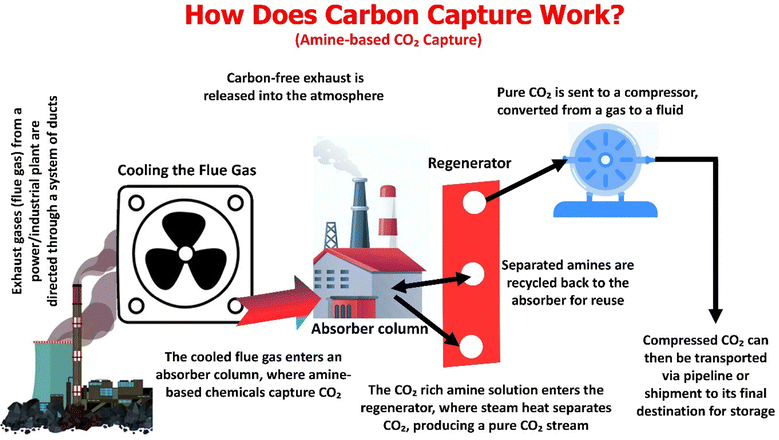
The CCUS process consists of three main steps and is illustrated in Fig. 3. The described process functions as a continuous chemical looping system, wherein an amine-based solvent selectively sequesters CO2 from flue gas, followed by its release for subsequent compression and storage.85–87 The primary stages, as depicted in the accompanying figure, along with their respective technical difficulties, are outlined below:
Flue gas intake and cooling: initially, the flue gas, which exits the power plant and primarily consists of N2, CO2, water vapor, and minor impurities (SOx, NOx, and particulates), is cooled to approximately 40–50 °C.88 This cooling step is crucial for optimizing the amine/CO2 reaction, as its efficiency diminishes significantly at higher temperatures. Significant challenges encompass solvent poisoning due to SOx/NOx, which leads to the formation of heat-stable salts, and particulate fouling, which intensifies pressure drop and increases maintenance requirements.
CO2 absorption (absorber column): following cooling, the gas ascends through the absorber column, while a lean amine solution is introduced from the top. CO2 is chemically absorbed through chemisorption, resulting in a CO2-saturated solvent and a processed flue gas. Significant challenges stem from the oxidative breakdown of amines when exposed to oxygen, corrosion caused by the byproducts of this degradation, and reaction kinetics that necessitate large column dimensions and substantial capital expenditures.89
Solvent regeneration (stripper/regenerator): the CO2-laden amine is subjected to heating (generally between 100 and 120 °C) to reverse the absorption process, thereby releasing high-purity CO2 and regenerating the lean solvent for subsequent use. The primary obstacle is the considerable parasitic energy requirement for steam production, which can account for 20–30% of the plant's total output. Furthermore, the thermal degradation of the solvent and losses of amine via vapor slip diminish both operational efficiency and the solvent's operational lifespan.87 Moreover, the high energy penalties associated with solvent regeneration (stripping), significant equipment corrosion requiring expensive metallurgy, and the oxidative degradation of amines, which leads to the emission of toxic byproducts, are also concerns.
CO2 compression: the liberated CO2 undergoes compression to supercritical pressures to facilitate efficient transport.90 This process requires rigorous dehydration protocols to prevent the formation of carbonic acid and subsequent corrosion, alongside multistage compression that incorporates intercooling to regulate both heat generation and energy expenditure.
Regarding transport and geological storage, supercritical CO2 is conveyed through pipelines or vessels and subsequently injected into deep geological formations, including saline aquifers and depleted oil or gas reservoirs located 2500 feet below ground.66 Such storage reduces the likelihood of atmospheric leakage and ensures safer long-term containment. CCUS will be a key part of this plan to help lower greenhouse gas emissions from cement, steel, and chemical manufacturing, which are hard to decarbonize and fix. Pipeline corrosion may be a possible risk if the purity of CO2 (absence of moisture, H2S, SO2, N2, O2, etc.) is compromised. In summation, despite the technological maturity of amine-based carbon capture and storage, its substantial energy requirements, solvent degradation, corrosion susceptibility, and long-term storage risks continue to drive research into advanced materials and alternative capture methodologies.
3. Methods of CO2 capture
The three primary methods of CO2 capture are depicted in Fig. 4: (i) pre-combustion, (ii) oxyfuel combustion, and (iii) post-combustion, which are further subdivided into different categories.91–93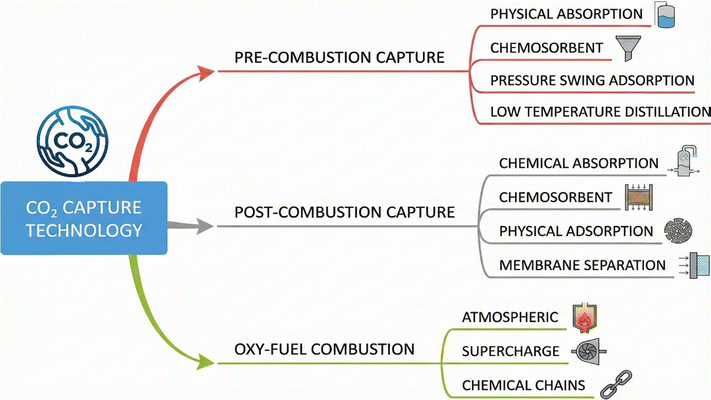 | ||
| Fig. 4 CO2 capture pathways and primary capture techniques in pre-combustion, post-combustion, and oxy-fuel processes. | ||
In the pre-combustion process, the fuel (coal or natural gas) is pre-treated before combustion.94 To prepare coal for use, it is gasified at low oxygen levels, producing syngas that is primarily composed of carbon monoxide and hydrogen gas and relatively free from other harmful gases.95 The steam-water gas shift process that occurs in the syngas converts the CO gas into CO2, producing more hydrogen gas. The high CO2 concentration (>20%) in the H2/CO2 fuel gas combination facilitates the extraction of CO2.96 The hydrogen is then burnt in an air atmosphere to produce mostly nitrogen gas and water vapour. Coal-fueled Integrated Gasification Combined Cycle (IGCC) power plants may employ this pre-combustion capture technique to increase efficiency by 7–8%. Two examples of physical solvents that work well for CO2 separation are Rectisol (based on methanol) and Selexol (polyethylene glycol ethers). While these are effective, they are expensive and often require complex thermal management. CNM-based materials offer a compelling alternative here due to their tunable surface chemistry and pore structures. Unlike Rectisol and Selexol, which rely on bulk liquid handling, solid-state carbon-based adsorbents (such as activated carbon, GO, and CNT-based filters) can potentially reduce the energy penalty associated with solvent circulation and regeneration. Furthermore, these adsorbents exhibit excellent material compatibility in these reducing syngas environments, resisting the chemical degradation that can plague organic solvents over time. This approach helps to generate syngas for chemical synthesis and carbon-free fuels. Although high costs limit its wider adoption, it is primarily employed in the fertilizer and hydrogen production sectors (Fig. 5).
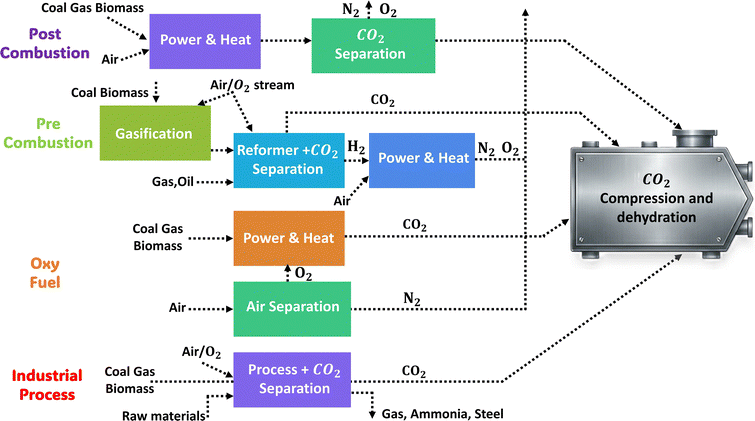 | ||
| Fig. 5 Graphical representation of major CO2 capture routes with integrated energy and industrial systems before transport, storage, and utilization. | ||
Whereas, burning fuel with extremely pure oxygen (≥95%) instead of air is known as oxy-fuel combustion capture.97,98 As a result, the exhaust gas contains negligible nitrogen, which significantly reduces thermal NOx.99 Using highly pure oxygen for combustion results in exhaust gases mainly composed of CO2, H2O, and SO2.100 Electrostatic precipitator and flue gas desulphurization techniques may remove particulates and sulfur dioxide. The residual gases contain a high percentage of CO2 (80–98%), which can be compressed, transported, and stored.101 Nevertheless, this approach consumes a significant amount of oxygen from the air separation unit, which increases expenses and requires substantial energy.102 Additionally, oxygen poses risks and impacts boiler performance.103 Moreover, flue gas recycling is necessary to control boiler temperature due to the higher combustion rate of oxygen-rich flue gas. Oxyfuel combustion remains a promising CO2 capture technique, despite these obstacles, with area for improvement.
However, post-combustion CO2 capture is a commonly used technology in the chemical industry because it captures CO2 after fuel combustion and can be retrofitted into existing power plants with minimal modifications.104,105 Monoethanolamine (MEA) and diethanolamine (DEA) are utilized in amine-based chemisorption, a widely used method that has drawbacks including solvent loss, corrosion, hazardous byproducts, and expensive regeneration.106 CNMs are gaining a significant role in post-combustion scenarios to address these specific drawbacks. In terms of energy efficiency, solid sorbents based on graphene or functionalized CNTs typically require significantly lower regeneration heat (1.2–2.5 GJ per ton CO2) compared to the latent heat of water vaporization inherent in aqueous amine systems (3.5–4.5 GJ per ton CO2).32 From a cost perspective, while pristine graphene is expensive, biomass-derived activated carbon and biochar offer a low-cost, sustainable route to high-surface-area materials that can compete economically with amine solvents on a large scale. Additionally, CNMs demonstrate superior chemical stability against oxidative degradation and corrosion, issues that severely limit the lifespan of amine solvents in oxygen-rich flue gas streams.
Chemical looping, which captures CO2, is another promising method for capturing CO2. However, it has high operational costs.107 Adsorption using novel solid sorbents is gaining interest as an alternative due to its efficiency. Similarly, oxy-fuel combustion, where fuel burns with pure oxygen and recycled flue gas, is another critical approach for CO2 capture. Key advantages include ease of integration, operational flexibility without plant disruption, and broad industrial adaptability. This method remains a promising, cost-effective solution for large-scale CO2 reduction. A detailed comparative summary highlighting the key differences between these technologies, along with their respective advantages and disadvantages, is presented in the accompanying figure (Fig. 6 and Table 3).
| Metric | Pre-combustion capture | Oxy-fuel combustion | Post-combustion capture |
|---|---|---|---|
| Thermodynamic driving force | Pressure-driven separation: elevated CO2 partial pressure enables low-energy physical separation processes | Phase-change–driven separation: near-pure CO2 streams allow condensation-based capture | Chemically driven separation: dilute CO2 streams require high-energy chemical bond formation and cleavage |
| Specific energy demand | ∼2.5 MJ kg−1 CO2 (lowest): energy is primarily associated with syngas generation and CO2 compression | ∼3.0–4.0 MJ kg−1 CO2: energy consumption is primarily driven by the electricity demand of the air separation unit (ASU) | ∼3.5–4.5 MJ kg−1 CO2 (highest): substantial steam input required for solvent regeneration (reboiler duty) |
| Efficiency | High: CO2 separation occurs upstream of expansion, minimizing irreversibility and exergy destruction | Moderate: significant combustion-related exergy losses, partially mitigated through latent heat recovery | Low: high entropy generation arising from CO2 separation from nitrogen-diluted flue gas |
| CNM integration potential | Transformative: membrane reactors enable simultaneous reaction and separation, shifting equilibrium, and eliminating intermediate cooling steps | High: membrane-based oxygen separation can replace cryogenic ASUs, substantially reducing parasitic energy losses | Incremental to moderate: advanced solid sorbents reduce sensible heat penalties, but the fundamental entropy constraint remains |
4. Key requirements of various CNMs for CO2 adsorption materials
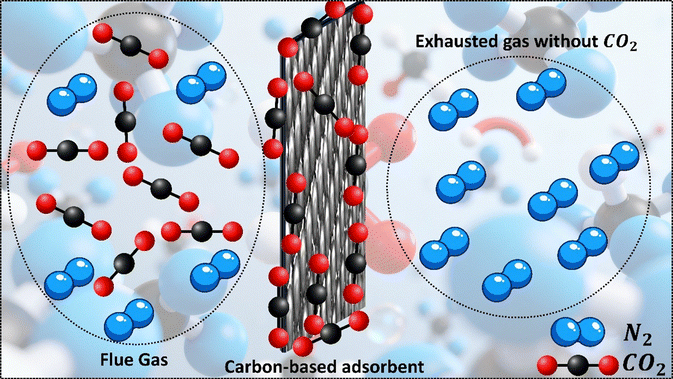
In recent years, solid adsorbents based on carbon (activated carbons, carbon nanotubes, graphene-based materials, and biochar) have gained significant attention over conventional solvent-based systems due to their several advantages, such as high efficiency, lower energy penalties, and operational flexibility.108–110 Due to their large surface areas, diverse pore architectures, chemical stability, and cost-effectiveness, these materials are beneficial for post-combustion and DAC applications.111 Recent breakthroughs in material synthesis, such as the creation of hierarchically porous carbons and nitrogen-doped frameworks, have significantly enhanced CO2 absorption and selectivity compared to other gases, including N2 and H2O.112Adsorption occurs when gas-phase molecules adhere to a nearby solid surface due to interactions between the gas-phase molecules and the solid surface.113 It can be categorized into two main types: physisorption (physical adsorption) and chemisorption (chemical adsorption). Physisorption occurs when molecules adhere to the surface of an adsorbent through weak van der Waals forces, with an enthalpy of approximately 10 kJ mol−1.114,115 The process could be over quickly because these interactions are weak. Chemisorption refers to the interaction between adsorbed molecules and the solid surface, where the interaction is stronger.116 These interactions can involve sharing electrons and atoms or the creation of new chemical species or radicals. The hydroxyl (–OH) and carboxyl (–COOH) groups enhance hydrogen bond interactions, leading to significant chemisorption, and exhibit an adsorption enthalpy of more than 100 kJ mol−1.117 It is also more difficult to reverse, which makes regeneration of the sorbent involved in chemisorption more challenging. For applications that utilize adsorption, the type of adsorbent material is crucial because it directly impacts the effectiveness of these processes. When selecting the right materials, key factors to consider are their performance under post-combustion conditions, where CO2 pressure is low, as well as their associated costs. The adsorbent's structure, including its pore structure, surface chemistry, and overall design, also significantly impacts its ability to capture CO2 effectively. The following is a detailed explanation of the main features (Fig. 7).
Surface area: the large surface area and high pore volume significantly affect the CO2 adsorption capacity of porous materials.118 An increased surface area results in a higher number of active sites available for CO2 molecule binding, thereby enhancing the overall uptake capacity.119 The balance between surface area and pore volume is essential for optimizing adsorption efficiency. The optimization of pore structure holds significant importance, and an optimal pore network ensures that active sites are easily accessible to CO2 molecules, thereby avoiding diffusion limitations that may impede adsorption.119 Efficient penetration of CO2 into the material occurs when pores are optimally distributed and possess suitable sizes, allowing for effective interaction with the available adsorption sites. The optimized pore architecture directly enhances CO2 capture efficiency. Porous materials, including activated carbon, graphene, and carbon nanotubes (CNTs), serve as prime examples of these principles. Activated carbons are characterized by their extensive pore networks and can exhibit surface areas that range from 400 to over 3000 m2 g−1. Graphene possesses a two-dimensional structure that provides an exceptionally high theoretical surface area. When engineered into porous configurations, it can achieve remarkable structure. Similarly, carbon nanotubes (CNTs), characterized by their hollow, tubular structures, offer high surface areas and adjustable interlayer spacing, allowing for precise control over pore size and accessibility. The ability of these materials to be engineered with extensive surface areas and customized pore volumes renders them highly effective and suitable candidates for the advanced CO2 capture process. Ongoing research on the development of these materials aims to optimize their characteristics further to achieve enhanced adsorption capacities and efficiencies.
Pore size, volume, and adsorption mechanism: a well-developed microporous structure is essential for efficient CO2 adsorption. The adsorption behavior depends on the pore size, affecting how different materials perform under various pressure conditions. Due to the strong molecular interactions in confined spaces, microporous materials are particularly effective for low-pressure adsorption. In contrast, mesoporous structures are more suitable for high-pressure adsorption, where physical adsorption dominates. Micropores (<2 nm) serve as active sites for CO2 capture through physisorption, where CO2 molecules interact with the surface via weak intermolecular forces, including hydrogen bonding and ionic interactions. Research indicates that CNM-based materials with a high ultra-micropore concentration exhibit significantly improved CO2 uptake even at low pressures (∼0.15 bar).32 The optimal pore size for CO2 physisorption is slightly larger than the kinetic diameter of CO2 molecules (0.33 nm), ensuring efficient molecular trapping and adsorption.120
Adsorption–desorption efficiency: the efficiency of adsorption and desorption depends on the pore structure of the material. Adsorption and desorption cycles must be accelerated for large-scale industrial applications, and a well-engineered adsorbent with an optimized pore structure guarantees effective mass transfer and quick kinetics.
In addition to the above-stated surface properties, the various other criteria for high-performance CO2 adsorbents for industrial applications are described below:
4.1. Heat of adsorption in CO2 capture
The heat of adsorption (Qst) is a critical factor in determining the interaction strength between CO2 molecules and adsorbent materials, directly influencing the feasibility of CO2 capture. In post-combustion scenarios, where CO2 concentrations are relatively low, adsorption based on physisorption is weaker.121 In physisorption, porous carbon materials exhibit Qst values ranging from 15 to 50 kJ mol−1, ensuring low energy consumption for regeneration. In contrast, chemisorption-based adsorbents (Qst > 80 kJ mol−1), such as amine-functionalized carbons or doped materials, are better suited for post-combustion CO2 capture due to their ability to form stronger covalent or ionic bonds with CO2. For industrial-scale CO2 capture, achieving an optimal balance between adsorption efficiency and regeneration energy consumption requires a Qst range of 35–50 kJ mol−1. Functionalizing porous materials with amine groups can also enhance CO2 adsorption by increasing Qst to approximately 50 kJ mol−1. Conversely, mesoporous and macroporous adsorbents are more suitable for pre-combustion CO2 capture due to their larger pore sizes, which enable efficient gas diffusion and removal. The optimal Qst range for industrial use is 25–50 kJ mol−1, striking a balance between high adsorption capacity and energy-efficient regeneration.4.2. Chemical, thermal, and mechanical stability in CO2 capture
To achieve effective CO2 capture in practical applications, adsorbents must demonstrate outstanding chemical, thermal, and mechanical stability. In post-combustion environments, characterized by flue gases with elevated temperatures ranging from 40 to 80 °C and a low CO2 pressure of approximately 15 bar, maintaining the structural integrity of the adsorbent is essential. Exposure to water vapor in flue streams can lead to swelling and subsequent degradation, compromising the efficiency of the adsorbent. CNMs exhibit significant advantages attributed to their elevated thermal and chemical stability, ensuring prolonged durability and reusability. Certain carbon frameworks have demonstrated the capability to adsorb and desorb CO2 for over 100 cycles, exhibiting a capacity loss of less than 10%, which highlights their durability. Contaminants present in flue gas, including N2, NO2, and SO2, can adversely affect CO2 selectivity, thereby complicating the performance of adsorbents.122 To resolve these challenges, it is essential to design adsorbent materials that can endure humid environments while preserving mechanical and thermal stability, ensuring that optimal performance is not compromised.4.3. Surface chemistry and functionalization
The surface chemistry of adsorbent materials is crucial for controlling the adsorption of CO2 and the selectivity of the process.122 Adding functional groups to different heteroatoms (such as nitrogen and boron) or chemical groups can significantly improve the ability to adsorb. Nitrogen doping (via amines, amides, or pyrrolic/pyridinic N) makes chemisorption stronger by making acid–base interactions stronger. Studies show that CO2 absorption is 20–50% higher in nitrogen-doped carbon materials as compared to undoped. Nitrogen-doped graphene aerogels are better at capturing CO2 because they have basic nitrogen sites that interact well with CO2 molecules. Adding carboxyl, phenolic, and carbonyl groups to the surface also makes it more polar, which facilitates the bonding of CO2 to hydrogen, thereby increasing the surface's effectiveness in adsorption.123 Adding sulfur or phosphorus atoms to the material, instead of nitrogen, creates more active sites and alters the distribution of electrons. Including metals like Mg, Ca, or transition metals in these materials can also boost the adsorption effectiveness by increasing CO2 binding affinity through Lewis acid–base interactions.4.4. CO2 adsorption capacity
The adsorption capacity is a crucial factor in determining how effectively CO2 can be collected, as it directly influences the rate at which CO2 is absorbed from gas mixtures. For CO2 capture to work well, rapid adsorption kinetics are necessary. This means that porous materials can swiftly trap CO2 molecules. However, in other cases, microporous materials may hinder the movement of CO2, potentially impairing overall efficiency. Physisorption allows CO2 molecules to enter small gaps in microporous materials (with pore diameters ≤0.33 nm), thereby accelerating diffusion at low pressures. At higher pressures, both micro- and macropores increase in volume, significantly altering the adsorption process. It typically takes only 2 to 4 minutes for materials to absorb a significant amount of CO2 to reach complete equilibrium. Amine-functionalized adsorbents may be engineered to enhance CO2 adsorption rates by reinforcing hydrogen bonding interactions with CO2. Chemical activation using KOH, ZnCl2, or H3PO4 has also been found to improve the ability of CNM-based adsorbents to capture CO2.4.5. Adsorbent costs and the synthesis process
Choosing the appropriate adsorbent requires carefully weighing several key criteria. For industrial-scale applications, the production and synthesis processes must be both cost-effective and environmentally friendly, while also being resource-efficient. CNM-based materials must be affordable and easily produced in large quantities to enable widespread use.124 Using biochar and other abundant, renewable materials offers a low-cost way to capture CO2. Additionally, new technologies have enabled the large-scale production of materials like graphene oxide, thereby facilitating their practical use.1254.6. Material regeneration and recyclability after CO2 capture
A key part of CCUS is the excellent regeneration of adsorbent materials, which makes them usable for an extended period while using less energy. The last stage of the CCUS cycle, CO2 desorption from the sorbent, is equally as critical as the first step, i.e., adsorption. Traditional CCUS techniques rely on the chemical absorption of CO2, followed by thermal desorption from aqueous alkanolamine solvents at 100–120 °C.126 This method requires a significant amount of energy and causes amine degradation, making it less affordable to reuse. Solid nanoporous materials may be used instead of amine-based solvents, especially those that include N-donor functional groups, such as pyridine, imidazole, and tetrazole.127 Dipole–quadrupole interactions between CO2 molecules and nitrogen sites enhance the adsorption capacity of these materials for CO2, requiring lower regeneration temperatures compared to water-based solutions. Additionally, the strong interactions between CO2 molecules and both the surface and interior pores enhance adsorption efficacy, thereby decreasing material degradation across multiple cycles. Therefore, porous materials rich in nitrogen offer a long-term, cost-effective method for absorbing a significant amount of CO2. They are also more recyclable and can be utilized in CCUS applications for an extended period.1284.7. Resistance to moisture and impurities in flue gas
In addition to CO2 and NO2, industrial flue gas contains water vapor and trace amounts of other pollutants, such as NOx and SOx.129 An ideal adsorbent must exhibit high tolerance to these impurities to maintain the efficiency and cost-effectiveness of the CO2 capture process. Common sorbents, such as activated carbon and zeolites, often face reduced adsorption capacity in humid environments, creating a significant challenge. To address this, adsorbents are designed to minimize competitive adsorption of H2O and other contaminants, thereby improving CO2 selectivity.130 Developing materials with inherent moisture resistance eliminates the need for pre-drying or additional purification steps, simplifying the process and making large-scale carbon capture more economically viable.1314.8. Selectivity
In this way, along with the above-discussed points, selectivity is a crucial concern that permits further exploration. The purity of captured CO2 is significantly influenced by the selectivity of the sorbent, which refers to its ability to preferentially adsorb CO2 over other gases present in the flue gas mixture. Selectivity is commonly quantified as the ratio of CO2 adsorption capacity to that of competing gases, such as N2. A high CO2/N2 selectivity ensures efficient separation, leading to high-purity CO2 for further utilization or storage.1324.9. Comparative performance of CNMs in CO2 capture
CNMs exhibit superior performance compared to other materials in CO2 capture. With surface areas of up to 2688 m2 g−1 and pore volumes of 0.6–1.5 cm3 g−1, they offer higher capacity and enhanced molecular sieving through ultra-micropores (<0.7 nm). Their tunable heats of adsorption (15–40 kJ mol−1) enable efficient regeneration, while capacities reach 5–8 mmol g−1 at 1 bar and up to 15 mmol g−1 at 5 bar, well above industrial benchmarks. The detailed performance matrix of these materials compared to the industry standard is highlighted in Table 4.| Performance metrics | Industry standard | Performance of CNMs | Advantage |
|---|---|---|---|
| Optimal surface area range | 800–1500 m2 g−1 | 1500–2688 m2 g−1 | Higher surface area |
| Critical pore size distribution | 70% micropores (<2 nm) | 60–85% ultra micropores (<0.7 nm) | Enhanced molecular sieving |
| Required pore volume | 0.4–0.8 cm3 g−1 | 0.6–1.5 cm3 g−1 | Higher volume |
| Target heat of adsorption | 25–35 kJ mol−1 | 15–40 kJ mol−1 | Tunable energy requirements |
| Minimum CO2 capacity (1 bar) | 3–5 mmol g−1 | 5–8 mmol g−1 | High capacity |
| Maximum CO2 capacity (5 bar) | 6–10 mmol g−1 | 8–12 mmol g−1 | Improvement |
| CO2/N2 selectivity target | 10–50 | 15–200 | High selectivity |
| Cycling stability requirement | >100 cycles | >300 cycles | Longer cycle life |
| Regeneration temperature | <120 °C | <100 °C | Lower regeneration temperature |
| Maximum material cost | <$100 per kg | $20–200 per kg | Cost-competitive at scale |
| Production scalability | Moderate | Good to excellent | Superior for most types |
| Environmental stability | Good under dry conditions | Excellent chemical stability | Enhanced durability |
5. Utilization of captured CO2
Currently, the predominant use of captured CO2 is enhanced oil recovery (EOR), where CO2 is injected into oil reservoirs to facilitate the extraction of additional crude oil.133 In addition to EOR, there is growing interest in using CO2 to make fuels, chemicals, and industrial materials through catalytic and electrochemical processes.134 In contrast to liquid amines, which necessitate energy-intensive desorption processes, functionalized carbons, such as nitrogen-doped porous carbons and CNTs, can serve a dual purpose. They can both seize CO2 and facilitate the direct conversion of CO2 into methane or syngas through the support of metal catalysts, including nickel and ruthenium, thus mitigating overall energy expenditures.Furthermore, developing globular pathways facilitates the conversion of captured CO2 into valuable products. These include polymers, construction materials, and advanced CNMs, such as CNTs and graphene, which can be produced via electrochemical or chemical vapor deposition methods.135 Biological approaches, employing algae or bacteria, present an additional utilization strategy. In these methods, CNMs, such as GO scaffolds, augment CO2 absorption and biomass production. Consequently, these sophisticated utilization strategies collectively underscore the potential of CNMs to enhance the economic feasibility and environmental impact of carbon capture and storage.136–138
Table 5 highlights recent progress in CNM-based catalysts enabling the electrochemical conversion of CO2 into value-added C2+ hydrocarbons, alcohols, and N-containing products through C–C and C–N coupling pathways. A key feature across these systems is the use of metal-free or heteroatom-doped carbon frameworks, which overcome limitations associated with metal catalysts, such as high cost, scarcity, and susceptibility to poisoning, while offering tunable electronic structures and abundant active sites.
| Catalyst type | Catalyst material | Coupling reaction | Final products | Performance metrics (faradaic efficiency/yield) | Ref. |
|---|---|---|---|---|---|
| Metal-free | N-doped graphene quantum dots (NGQDs) | C–C (CO2, H2O) | C2H4, C2H5OH | FE = 90% at −0.74 V, I = 46 mA cm−2 at −0.86 V | 139 |
| N-doped carbon (c-NC) with ordered cylindrical mesopores | C–C (CO2, H2O) | C2H5OH | FE = 77% at −0.56 V, selectivity 100% | 140 | |
| B and N codoped nanodiamonds (BND) | C–C (CO2, H2O) | C2H5OH | FE = 93.2% at −1.0 V, Y = 90 mg h−1 cm−2 | 141 | |
| F-doped CNTs | C–N (CO2, NO3−) | CO(NH2)2 (Urea) | FE = 18.0% at −0.65 V, I = 3.5 mA cm−2Y = 6.36 mmol g−1 h−1 | 142 | |
| N-doped carbon | C–N (CO2, NO3−) | CO(NH2)2 (Urea) | FE = 62% at −0.5 V, Y = 596.1 µg mg−1 h−1 | 143 |
6. CNM-based adsorbents for CO2 capture
Materials intended for CO2 capture must combine cost-effectiveness, wide availability, and high performance to satisfy global demands. Among potential candidates, carbon stands out as an exceptionally versatile element, offering a wide array of allotropes and the ability to form structures across all dimensional scales from zero-dimensional (0D) carbon dots and fullerenes, to one-dimensional (1D) carbon nanotubes, two-dimensional (2D) graphene sheets, and three-dimensional (3D) architectures such as carbon foams and activated carbon.144–147 0D materials (fullerenes and carbon dots) represent the lowest dimensional limit with cage-like sp2-carbon frameworks and high theoretical surface-to-volume ratios.148 However, pristine fullerenes interact weakly with CO2via physisorption, and their strong tendency to aggregate into fullerite crystals drastically reduces accessible surface area. Although ionization or functionalization can theoretically enhance CO2 binding, electronic instability, aggregation, and prohibitively high synthesis costs restrict 0D carbons to significant roles as functional additives rather than practical sorbents.149,150 1D materials, such as carbon nanotubes (CNTs), offer dual adsorption domains on their inner lumen and outer surfaces.110 While confinement within nanotube interiors can enhance CO2 binding, mass-transfer resistance and weak interactions with smooth graphitic walls limit practical uptake. Chemical functionalization (e.g., amine grafting) improves selectivity and capacity but introduces trade-offs by blocking pores and slowing kinetics. 2D materials (graphene and GO) provide extremely high theoretical surface areas and tunable surface chemistry.32,39 Their primary limitation is irreversible restacking driven by π–π interactions, which collapses porosity. Structural engineering strategies, such as pillaring with CNTs, metal oxides, or MOFs, successfully preserve interlayer spacing and enhance CO2 diffusion. 3D hierarchical porous carbons represent the most technologically mature and scalable class of materials. By integrating micro-, meso-, and macropores into a continuous framework, these materials overcome diffusion limitations inherent to lower-dimensional systems. Micropores offer high adsorption potential, while meso- and macropores facilitate rapid mass transport.151As discussed in the earlier section, this study focuses on specific CNM-based adsorbents for the collection and subsequent transformation of CO2 into value-added products. To provide a comprehensive picture, we discuss some materials in depth and then summarize their comparison with other essential CNMs. This method facilitates easier comparison of their performance, benefits, and drawbacks in the context of CO2 capture. Table 6 provides a summary of several CNMs discussed, focusing on their structural characteristics, physicochemical properties, and specialized functions in CO2 adsorption. Some of the main benefits, including a large surface area, adjustable porosity, and the capacity to transform with functional groups, are also included, along with some of the drawbacks, such as the difficulty of regeneration and expense. This comparison enables us to identify the most suitable candidates for CCUS applications that are both effective and long-lasting.
| Carbon material | Surface area (m2 g−1) | Pore structure | Functionalization/modification | CO2 adsorption characteristics | Notes | Ref. |
|---|---|---|---|---|---|---|
| Activated carbon | 500–2500 | Microporous with some mesopores | Physical (steam or CO2) or chemical (KOH, H3PO4) activation | High capacity at high pressure; low selectivity without functional groups | Inexpensive, scalable, sensitive to humidity | 152 |
| Carbon nanotubes | 100–1000 | Mostly mesoporous; can be microporous | Acid/base treatment; amine or metal doping | Moderate CO2 uptake; fast kinetics; tunable properties | Excellent thermal/mechanical stability | 153 |
| Graphene oxide | 300–700 | Layered, mesoporous/microporous | Oxygen groups (–OH, –COOH, and epoxy) naturally present | Moderate uptake; improved selectivity due to polar groups | Good dispersion in composites; lower conductivity | 154 |
| Reduced graphene oxide | 500–1200 | Less oxidized than GO; microporous/mesoporous | Partially removed O-groups by chemical or thermal reduction | Increased capacity due to the restoration of the π-conjugated network | Hydrophobic; can be used in composite adsorbents | 154 |
| Carbon aerogels | 400–1500 | Highly porous (micro–meso) | Controlled sol-gel synthesis; surface functionalization is possible | High capacity; excellent regeneration due to open structure | Fragile; high synthesis cost | 155 |
| Carbon xerogels | 400–1200 | Micro- and mesoporous | Like aerogels, ambient drying | Comparable to aerogels but with more practical processing | Useful for large-scale applications | 156 |
| Ordered mesoporous carbon | 1000–2000 | Highly ordered mesopores | Can be doped with N, amines, or metals | High CO2 uptake; tunable pore architecture | Made using mesoporous silica templates | 157 |
| Biochar | 200–900 | Microporous and mesoporous | Depends on the pyrolysis temperature and the feedstock | Low-to-moderate CO2 uptake; potential for amine grafting | Renewable; cost-effective; environmental benefit | 158 |
| MOF-derived porous carbon | 1000–3000 | Microporous, hierarchical, and possible | Direct carbonization; N-doping from ligands | Very high surface area; high selectivity with N-doping | Retains structural features of MOFs; thermal stability is enhanced | 159 |
| Activated carbon fibers | 1000–2500 | Predominantly microporous | Surface oxidation, amine impregnation | High CO2 capture rate and capacity due to fiber morphology | Excellent for rapid adsorption/desorption cycles | 152 |
| Hard carbon | 300–600 | Microporous with closed pores | N-doping or physical activation | Moderate uptake; cost-effective | Commonly used in energy and storage devices | 160 |
| Soft carbon | <100 | Mostly nonporous | Not typically used for CO2 capture | Poor adsorption performance | Used more for electrodes than adsorption | 161 |
6.1. Activated carbon-based adsorbents
Porous materials, particularly activated carbon, have attracted significant global research interest owing to their tunable pore architecture, exceptionally high surface area, and remarkable chemical and thermal stability.162,163 These attributes contribute to activated carbon's high CO2 adsorption capacity, complemented by excellent selectivity and ease of regeneration. A key advantage of activated carbon is its sustainable production, as it can be derived from waste biomass and agricultural residues, making it both cost-effective and environmentally friendly.164,165 This section of the review discusses the various precursor materials employed in the synthesis of activated carbon and subsequently applied for CO2 adsorption.166–171Sawdust, a common byproduct of woodworking, is traditionally used in rural areas as a fuel source for heat and light. However, it has shown significant promise as a precursor for producing activated carbon or biochar for CO2 adsorption applications172–176 Foorzinezhad et al.177 recently synthesized activated carbon from Iranian sawdust using a pure CO2 activation process. Sawdust was activated at six different temperatures, ranging from 700 °C to 1100 °C. SEM images (Fig. 8) show the morphological evolution of carbon with increasing temperature. The unactivated carbon (Fig. 8a and b) exhibits a smooth, amorphous surface with minimal porosity. At 700 °C (Fig. 8c), initial pore formation is observed, although it is partially filled with ash. Increasing temperature (800–1100 °C) significantly improves porosity due to byproduct removal. At 1000 °C, smaller pores merge within larger structures (inset, Fig. 8f), attributed to the formation of tunnel-like channels through the volatile gasification process. However, above 1000 °C, the structure becomes irregular and heterogeneous, potentially reducing surface area and CO2 adsorption capacity.
 | ||
| Fig. 8 FE-SEM micrographs of (a, b) untreated sawdust, (c) A-CWO-700, (d) A–CS–800, (e) A–CS–900, (f, g) A–CS–1000, (h) A–CS–1050, and (i) A–CS–1100; (j) Nitrogen adsorption–desorption isotherms and (k) corresponding pore size distribution of A–CS–1000 calculated using NLDFT (reproduced from ref. 177 under the Creative Commons Attribution (CC BY) license); (l) BET adsorption–desorption isotherm of activated carbon (HPAC and HPC) (reproduced from ref. 178 with permission from [Springer Nature], copyright [2023]). | ||
BET surface area increased with temperature, reaching a maximum of 1651 m2 g−1 at 1000 °C before declining to 1163 m2 g−1 at 1100 °C, as shown in Fig. 8(j). This trend directly influenced CO2 uptake, with the highest adsorption capacity observed in samples treated at 1000 °C, attributed to the optimized surface area. Fig. 8(k) shows the pore size distribution curve for the 1000 °C-treated sample, obtained using non-linear Density Functional Theory (DFT), which demonstrates the presence of ultra-micropores (0.4–1.76 nm). Specifically, as the activation temperature increased from 700 °C to 1000 °C, CO2 adsorption capacity increased from 3.3 to 9.2 mmol g−1 after 35 minutes but slightly declined to 8.6 mmol g−1 at 1100 °C, likely due to the loss of active sites, functional groups, and structural alterations that weakened the adsorbent–adsorbate interactions. Moreover, operational parameters influenced the adsorption performance, with the highest uptake recorded at a low flow rate of 50 mL min−1. Temperature also played a crucial role; the 1000 °C heat-treated sample captured 10.64 mmol g−1 of CO2 at 0 °C, compared to 9.2 mmol g−1 at 50 °C.
Dry fruit shells, a significant byproduct of agro-industries, have been effectively utilized as precursors for the production of activated carbon (AC), with applications in CO2 adsorption.179–182 Koli et al.178 employed Terminalia catapa (Indian almond) shells to synthesize AC through acid activation, followed by heat treatment under air (HPC) and nitrogen (HPAC) atmospheres. The resulting materials were utilized for CO2 capture. The surface area and pore structure play a significant role in determining their CO2 capture capacity. Fig. 8(l) shows the nitrogen adsorption–desorption curve for HPC and HPAC, demonstrating a small hysteresis at p/p0 = 0.4. This confirms the presence of meso- and micro-pore structures in both samples.178 Notably, HPAC exhibited a higher surface area (∼616 m2 g−1) and CO2 adsorption capacity (2.3 mmol g−1) compared to HPC, which showed low CO2 uptake (1.7 mmol g−1) owing to a lower surface area (244 m2 g−1). Similarly, Xu et al.180 developed K2CO3-activated porous carbon from peanut shells, achieving a remarkable CO2 adsorption capacity of 5.7 mmol g−1 at 273 K, which significantly outperformed unmodified peanut shells (1.54 mmol g−1) and sunflower seed shells (1.46 mmol g−1).179,180 In another study, activated carbon derived from walnut shells exhibited a CO2 adsorption capacity of 2.1 mmol g−1.170 Olive tree pruning residues have also been explored as a biomass source for AC synthesis aimed at CO2 capture.183,184 AC derived from this biomass has shown impressively high surface areas, ranging from 786 to 1985 m2 g−1.
Recently, Ramos et al.185 developed a high-performance CO2 adsorbent by using olive mill waste through chemical activation with potassium hydroxide (KOH). The optimized activated carbon (designated OMW-1) exhibited an impressive CO2 uptake performance of 2.4 mmol g−1 in pure CO2 and, critically, 0.84 mmol g−1 in 15% vol CO2, making it suitable for realistic industrial applications. SEM analysis reveals that activation parameters critically govern pore development and CO2 adsorption behavior of OMW-derived activated carbon (Fig. 9). Activation at 650 °C (OMW-1) yields well-distributed, uniform cavities with a stable micro–mesoporous framework, whereas 950 °C (OMW-2) causes excessive thermal decomposition of cellulose, hemicellulose, and lignin, leading to enlarged, non-uniform pores and partial structural collapse. Increasing activation time from 45 to 75 min promotes gradual formation of interconnected pores. However, prolonged treatment (120 min) induces surface cracking and degradation, especially at higher temperatures. The KOH/biochar ratio further modulates pore architecture. The 2:1 ratio (OMW-1) provides a balanced micro–mesopore network optimal for CO2 capture, while higher ratios (4:1 and 6:1) progressively increase microporosity (up to 96.3%) but reduce total pore volume and accessibility, diminishing practical adsorption efficiency. These morphological trends directly explain adsorption performance: OMW-1, with 94.6% microporosity and a narrow average pore size of ∼4.8 Å, exhibits the highest CO2 uptake (105.7 mg g−1), whereas OMW-3, despite its ultra-high surface area (2577 m2 g−1), shows inferior uptake due to excessive meso/macropore contribution.
 | ||
| Fig. 9 SEM micrographs recorded at 1000× magnification for the activated carbon samples: (a) OMW-1, (b) OMW-2, (c) OMW-3, (d) OMW-4, (e) OMW-5, and (f) OMW-6, illustrating the evolution of surface morphology under different activation conditions (reproduced from ref. 185 under the Creative Commons Attribution (CC BY 4.0) license). | ||
Thus, based on the above studies, it can be concluded that increasing activation temperatures generally reduces surface area and pore volume due to structural collapse. However, CO2 adsorption capacity improved when the activation temperature was raised from 500 °C to 600 °C, before declining at even higher temperatures. The highest CO2 uptake (265.8 mg g−1 at 0 °C) was reported for the AC sample activated at 600 °C with a carbon-to-KOH ratio of 1:3 (AC600 3/1).183
A clear performance pattern is observed across various biomass feedstocks. Precursors with inherently high lignocellulosic content, such as coconut and walnut shells, consistently exhibit superior microporosity compared to soft biomass, including leaves and stalks, when activated under identical conditions.186,187 Nevertheless, the synthesis method often has a greater influence on the final performance than the origin of the feedstock. Chemical activation with KOH or H3PO4 generally yields surface areas 1.5–2 times higher than those achieved through physical activation with CO2 or steam, thereby enabling higher CO2 gravimetric uptake, despite the trade-off of broader pore size distributions, which may compromise selectivity at low partial pressures.
In recent years, significant attention has been directed toward modifying and functionalizing AC materials to enhance the availability of active adsorption sites for CO2 capture. The incorporation of heteroatoms, such as nitrogen, oxygen, and sulfur, into the carbon framework has emerged as an effective strategy to enhance CO2 uptake. Since CO2 is inherently acidic, it can strongly interact with materials containing basic surface functionalities, such as amine groups.167 Amine-functionalized AC exhibits superior CO2 adsorption capacities compared to pristine or unmodified AC (SAC). Functionalization with various amine compounds, such as diethylenetriamine (DETA), triethylenetetramine (TETA), and tetraethylenepentamine (TEPA), has been explored in the literature, where TETA introduces both primary and secondary amine groups.188 Due to its shorter chain length, DETA facilitates a higher degree of amine functionality incorporation into the AC structure.
Additionally, the pore architecture plays a critical role in determining adsorption performance, as it governs the accessibility of adsorbate molecules to the adsorbent surface. While amine functionalization can generate mesoporous structures, as evidenced by type IV hysteresis loops, it often reduces surface area due to pore blockage by the relatively large molecular weight of organic amines. Fig. 10(a) presents FTIR analysis of SAC composites modified with varying concentrations of DETA and TEPA, while Fig. 10(c) illustrates the proposed reaction mechanism. This mechanism involves the formation of a zwitterionic intermediate upon interaction between the lone pair of electrons in the amine and the carbon atom present in CO2, followed by deprotonation by a free amine group to yield carbamate species, as confirmed by FTIR spectra (Fig. 10a and b) after CO2 adsorption.189 Remarkably, a 30% DETA–SAC composite achieved an adsorption capacity of 78.62 mg g−1 under 20% CO2 concentration conditions and at a gas flow rate of 800 mL min−1.
 | ||
| Fig. 10 (a) FT-IR spectra of various carbon materials; (b) magnified view of the region highlighted by the orange dashed circle; (c) schematic illustration of the potential CO2 adsorption mechanisms on carbon surfaces functionalized with primary and secondary amine groups (reproduced from ref. 189 with permission from [Elsevier], copyright [2024]). | ||
Further advancements include the work conducted by Lotfinezhad et al., who employed nitrogen doping on jujube and date seed-derived activated carbon using urea as the nitrogen source following KOH activation. Nitrogen incorporation occurs through the thermal decomposition of urea, which releases free amine radicals that interact with hydroxyl groups on the activated carbon surface, forming nitrogen-functional groups and creating a microporous architecture.190 Ospino et al. reported that nitrogen doping enhances CO2 capture capacity and selectivity but compromises AC's textural properties. A notable drawback of nitrogen-doped activated carbon is its diminished CO2 adsorption performance under humid conditions, attributed to the strong water affinity of the nitrogen-functionalized surfaces.191
It has been observed that the CO2 adsorption capacity increases significantly as the CO2 pressure increases. For example, at 90 vol% CO2, the adsorption reached 2.33 mmol g−1, while at 10 vol% CO2, it dropped to 1.16 mmol g−1. This improvement at higher pressures is due to CO2 molecules interacting more with the accessible adsorption sites on the material's surface. Even when the same synthesis circumstances and procedures are used, the choice of precursor is crucial in determining the effectiveness of the final adsorption. For instance, nitrogen-doped activated carbon made from date seeds exhibited better CO2 absorption (2.93 mmol g−1) compared to activated carbon made from jujube seeds. This was mainly because the microporous structure of the date seeds was more developed. Another important aspect that affects how things adsorb is temperature. A rise in CO2 capture temperature typically makes it more challenging to collect CO2 while all other factors remain constant. Specifically, increasing the temperature from 25 °C to 50 °C for date-seed-derived AC resulted in a significant 42.7% decrease in adsorption capacity.190
Composites, compared to activated carbon alone, have shown significant improvements in their ability to capture CO2. MXenes, a class of two-dimensional transition metal carbides and nitrides, are one of the most promising materials to be added to activated carbon matrices. They have lately gotten a lot of attention for their use in gas adsorption. DFT tests show that clean MXenes can hold around 0.36 mg g−1 of CO2, mainly because they are very good at transferring charge from the MXene surface to CO2 molecules. Adding activated carbon to the spaces between the layers of MXenes helps prevent MXenes from restacking, thereby increasing the surface area available for adsorption. Adding just 2.5% MXene to the activated carbon matrix made a significant difference in the amount of CO2 it could hold, increasing from 46.46 cm3 g−1 to 67.83 cm3 g−1.
Beyond adsorption capacity, gas selectivity is a critical parameter for separation processes. The MXene–AC composite exhibited superior CO2 selectivity, with a measured uptake of 67.83 cm3 g−1, compared to only 13.65 cm3 g−1 for CH4 and 5.29 cm3 g−1 for N2. Even pristine AC showed respectable selectivity, adsorbing 46.46 cm3 g−1 of CO2, versus 11.004 cm3 g−1 for CH4 and 3.992 cm3 g−1 for N2. Equilibrium adsorption, pure component analysis, and the ideal adsorbed solution theory (IAST) all confirmed the selectivity tests, which showed that CO2 > CH4 > N2 was the order of preference. The improved selectivity of the MXene–AC composite is attributed to the chemisorption process facilitated by the presence of titanium carbide functional groups, as indicated by the FTIR signal at 105 cm−1. In a separate investigation, Gorbounov et al.192 produced acid-treated carbon by using a nitration mixture (HNO3 and H2SO4) on physically activated biomass. This one-step treatment enhanced surface oxidation and nitrification, resulting in an adsorption capacity of 0.96 mmol g−1 at 50 °C. Combining acid treatment with plasma activation has also been demonstrated to be an effective method for increasing surface area, porosity, and CO2 collection capability. Zhiping Ye et al.193 used a two-step treatment method, first utilizing HNO3 acidification and then exposing the sample to cold plasma, as shown in Fig. 11. Adsorption measurements revealed a substantial increase in CO2 uptake: 81% at 298 K and 64% at 273 K, compared to untreated carbon. Mechanistically, the acid treatment introduced oxygen-rich functional groups (e.g., carboxyl, nitro, and phenolic) that provided active sites for subsequent nitrogen doping, enhancing CO2 and NO2 adsorption. The plasma treatment, using N2/Ar gases, generated high-energy species (ionized N2 and Ar+), where Ar+ ions played a dominant role in etching the AC surface, creating microporous structures. In contrast, reactive N2 species enabled effective nitrogen doping.
 | ||
| Fig. 11 Schematic illustration of surface modification of activated carbons (ACs) via acid treatment and cold plasma techniques, highlighting the introduction of functional groups and enhancement of surface activity (reproduced from ref. 193 with permission from [Elsevier], copyright [2025]). | ||
This section highlights that the efficacy of porous carbons in capturing CO2 is influenced by the interplay between pore structure and surface characteristics, both of which are significantly impacted by the activation and functionalization methods employed. Chemical activation, especially when employing KOH, proves highly efficient in creating ultrahigh surface areas and hierarchical pore structures, thereby facilitating rapid adsorption kinetics and substantial capacities. Nevertheless, these benefits are offset by diminished carbon yields, more demanding processing conditions, and the requirement for extensive post-treatment. Conversely, physical activation utilizing CO2 or steam presents a more yield-preserving and scalable approach, yielding narrowly distributed ultramicropores (<1 nm) that augment adsorption enthalpy and CO2/N2 selectivity through size-sieving effects, thus rendering this method particularly appealing for post-combustion capture under conditions of low CO2 partial pressures.
Beyond textural optimization, amine functionalization emerges as a powerful strategy to improve low-pressure CO2 uptake through strong chemisorptive interactions. The incorporation of amines such as PEI or TEPA significantly enhances affinity toward CO2, particularly under dilute conditions, but at the expense of surface area and pore accessibility due to pore filling and diffusion limitations. Notably, the superior tolerance and, in some cases, enhancement of CO2 capture performance under humid conditions distinguish amine-functionalized carbons from purely physisorptive systems, highlighting their potential for realistic flue-gas environments. Overall, the insights consolidated in this review emphasize that no single strategy is universally optimal; rather, the rational integration of activation methods and surface functionalization, tailored to specific capture conditions, is essential for the development of next-generation CNM-based adsorbents for efficient and practical CO2 capture. A thorough comparison of precursor materials, activation and surface modification techniques, textural characteristics (including specific surface area), and the corresponding CO2 adsorption performance metrics of activated carbon-based adsorbents reported in recent literature is given in Table 7.
| Precursor material | Activation/modification | Surface area (m2 g−1) | CO2 uptake (mmol g−1) | Q st (kJ mol−1) | Selectivity (CO2/N2) | Ref. |
|---|---|---|---|---|---|---|
| Note: the gas composition (e.g., purity and mixed-gas environments) and operating pressures used across different studies may differ. | ||||||
| Sunflower seed shell | Carbonization and activation by KOH at 773 K | 1790 | 1.46 | 38–23.5 | 7.2 | 179 |
| Indian almond shell | Heat treatment in air (HPC) and nitrogen (HPAC) | 244 | 1.7 | 43 | 41.80 | 182 |
| 616 | 2.3 | |||||
| Commercial AC | 1 M HNO3 + H2SO4 and cold plasma treatment | 354 (acid) | 2.7 (298 K) | 14–27 | CO2/N2 (40.5) | 193 |
| 468 (plasma) | 3.3 (273 K) | |||||
| 339 (acid+ plasma) | ||||||
| AC | Not mentioned | 1025 | 46.46 cm3 g−1 | 26.7–31.3 | CO2/N2 (11.79) | 167 |
| CO2/CH4 (4.97) | ||||||
| Date-palm waste | Activation by KOH | 1506.3 | 6.71 at 0 °C and 4.214 at 25 °C | 35 | — | 194 |
| Starch | N-doped biochars with rich-oxygen functional groups | 409.3 | 1.2 at 0.15 bar CO2 | — | — | 195 |
| Waste tyre | TEPA loading (5%) | 109.78 | 43.88 mg g−1 | — | — | 188 |
| CO2 flow rate of 100 mL min−1 | ||||||
| Peanut shell 1 | Carbonization and activation of K2CO3 | 1150 | 5.7 at 273 K | 29.7–42.6 | — | 180 |
| Peanut shell 2 | Carbonization and activation at 500 °C | 956 | 1.54 | 21.5–60 | 5.6 | 179 |
| Sawdust | Activation using CO2 at 1000 °C | 1651 | 10.64 (0 °C) | — | 207.52 (0 °C) | 177 |
| 1100 °C | 1163 | 9.2 (50 °C) | 40.2 (25 °C) | |||
| 8.6 (50 °C) | ||||||
| Date seed | KOH-carbon activation (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
786 | 1.21 | — | — | 190 |
| Jujube seeds | 698 | 1.16 | — | — | ||
| Date seed | KOH activation and N-doping | 864 | 1.31 at 25 C | — | — | 190 |
| Walnut shell | CO2 activation | 810 | 2.1 | — | — | 166 |
| Olive tree pruning | 600 °C, carbon KOH ratio 1:3 | 786–1985 | 265.8 at 0 °C | 23.8 | — | 183 |
| KOH activation at 788.5 °C, carbon KOH ratio (1:6.38) | 3526 | 5.1 at 0 °C and 1 bar | 19.4 (chemical) | — | 184 | |
| 24.2 (physical) | ||||||
| AC | Acid treated (HNO3 and H2SO4) | 221 | 0.96 at 50 °C | 30.5 | — | 192 |
| Chitosan-based AC | N-functionalized | 2262 | 5.3 at 25 °C and 1 bar | 20–29 | 1–10 | 196 |
| Chestnut shell | Carbonized (500 °C) material, boron-doped/activated with KBrO2 | 683 | 3.15 mmol g−1 (298 K) | 21–38 | — | 197 |
| 4.22 mmol g−1 (273 K) | ||||||
| Coconut shell | Carbonized (500 °C) treated with thiourea and KOH (700 °C) | 1315 | 4.38 mmol g−1 (298 K) | — | 17 | 198 |
| 6.46 (273 K) | ||||||
| Lotus petioles | Carbonised (500 °C) treated with sodium phytate (700 °C) | 525 | 2.51 (298 K) | 15–44 | 19 | 199 |
| 3.34 (273 K) | ||||||
6.2. Fullerene-based adsorbents
Fullerene, a zero-dimensional (0D) carbon allotrope, is characterized by its distinctive cage-like structure, typically denoted by its number of carbon atoms, such as C60 or C70. Due to their unique geometry and electronic properties, fullerenes have garnered global research interest, particularly for adsorption applications involving fuels and greenhouse gases, such as NO2 and CO2.200 However, the inherent adsorption mechanism of fullerenes is limited primarily to weak physical interactions, which allows only a monolayer of gas molecules to bind to the first layer of active sites. This limitation mainly stems from the absence of intrinsic porosity in pristine fullerenes, rendering surface modification or decoration essential to enhance their adsorption performance.Studies have shown that pure C60 exhibits negligible CO2 adsorption because of its nonporous nature. Interestingly, when a single CO2 molecule adsorbs onto a metal-supported fullerene (C60) surface, the process becomes exoergic, with a reported adsorption energy of –1.57 eV. Doping strategies, such as the incorporation of titanium (Ti) or calcium (Ca) atoms onto the fullerene surface, have been reported to significantly enhance CO2 adsorption capacity.201,202 This improvement stems from enhanced charge transfer interactions between the dopant and CO2 molecules, as well as modifications to the adsorption energy and enthalpy profiles compared to undoped (bare) fullerenes. Metal or metal oxide doping thus provides a promising route to transform fullerenes from passive surfaces into highly active materials for CO2 capture.
Recent advancements in DFT and other computational techniques have highlighted the potential of fullerenes in CO2 adsorption. Bottani et al.201 conducted a Monte Carlo simulation study, which suggested that CO2 can be adsorbed onto fullerene surfaces if voids are present within the structure. Perham Rezaee et al.203 examined the CO2 adsorption properties of boron (B), phosphorus (P), and nitrogen (N) doped C20 fullerenes (C20-nXn, where X represents B, P, or N). Their findings revealed that boron and nitrogen doping enhanced CO2 adsorption more effectively than phosphorus doping. These stronger interactions are attributed to the higher concentration of the highest occupied molecular orbitals (HOMO) and lowest unoccupied molecular orbitals (LUMO) associated with B and N, which are primarily localized on the dopant atoms and adjacent carbon atoms, as shown in Fig. 12. Nitrogen doping activates the π-electrons, making the N–C bond a preferred site for electrophilic or nucleophilic attachment.203 When an electric field was applied, the CO2 adsorption capacity of the doped fullerene increased (from −0.53 eV to −0.71 eV) due to chemisorption processes that followed initial physisorption. In a separate study, phosphorus-decorated fullerene (P-C24N24) demonstrated selective CO2 adsorption from a CO2–N2 mixture.204 Using an electric field, in conjunction with DFT calculations, revealed that the binding distance between carbon and oxygen decreased (from 0.012 to 0.013 au) with increasing electric field strength, signalling the onset of chemisorption alongside physisorption. This method allows for controlled desorption of CO2 by toggling the electric field, providing a potential pathway for CO2 regeneration. Boron-doped fullerenes (e.g., B38, B40, and B80) have garnered particular interest in CO2 capture applications. While B80 has yet to be experimentally realized, its theoretical potential remains promising. Mahsa Kabiri et al. demonstrated the CO2 capture potential of B40 fullerenes, citing their amphoteric nature and practicality in the literature. B40 exhibited an impressive CO2 adsorption capacity of 13.87 mmol g−1 and good selectivity.205
 | ||
| Fig. 12 (a) HOMO plots of pristine C20 and doped C20−nXn structures (n = 1, 2, 3; X = B, N, P), illustrating the spatial distribution of the highest occupied molecular orbitals; (b) LUMO plots of pristine C20 and doped C20−nXn structures (n = 1, 2, 3; X = B, N, P), showing the lowest unoccupied molecular orbitals. In both plots, red and green represent the positive and negative phases of the wave function, respectively. Atom color coding: pink—boron, blue—nitrogen, yellow—phosphorus, grey—carbon (reproduced from ref. 203 under the Creative Commons Attribution (CC BY) license). | ||
However, doping with a transition metal is necessary to improve performance due to the energy barrier (1.2 V) associated with CO2 adsorption and its low dipole moment. Kabiri et al. showed that doping B40 with yttrium (Y) resulted in a higher adsorption energy (−0.75 eV) for CO2 molecules compared to pristine B40.206 Furthermore, CO2 adsorption in Y-doped B40 is dominated by chemisorption, as evidenced by the overlap between Y's 4d orbitals and CO2's LUMO, confirming the strong interaction between yttrium and CO2.206 Esrafili et al. explored scandium (Sc)-decorated porphyrin-like fullerene (C24) for efficient CO2 capture via adsorption. Similarly, Sebastian Anila et al. prepared imidazolium cation polyanionic fulleride ((IM+)n C60 (CN)n)n− ionic liquids, noting an increase in binding energy with a greater number of CN anions. The molecular electrostatic potential (MESP) analysis revealed that the excess electrons in the complex were delocalized over the unsaturated carbon centers of the fullerene, while the CN groups remained neutral. This increase in CN groups enhanced the interaction with CO2, converting non-covalent interactions into stronger covalent bonding. Further studies on nitrogen-doped C60 fullerene (C60–2nN2n) revealed that nitrogen doping increases the electron density of the fullerene molecule, improving the CO2 interaction.206 By attaching a CN-group to the carbon center (forming N-cyanofulleride), CO2 adsorption was further enhanced, as confirmed by MESP analysis, which indicated an electron-rich anion facilitating stronger interactions with CO2.207 While much of the research on fullerene-based CO2 adsorption has been theoretical, there is a limited amount of experimental work on its real-world applications. However, the potential for fullerene-based materials in CO2 capture remains a promising avenue for further investigation.208 The curvature of the fullerene cage has a significant influence on its interaction with CO2 molecules. Smaller cages with higher curvature, such as C20 and B40, exhibit localized electron density and enhanced Lewis basicity compared to the larger, more diffuse C60 cage. This higher charge concentration on the cage surface strengthens the dipole–quadrupole interactions with CO2, theoretically resulting in higher binding energies and improved capture affinity for highly curved analogues.
In conclusion, CO2 capture via fullerene-based methods is predominantly theoretical. Although experimental realization is limited, advances in density functional theory (DFT) and Monte Carlo simulations have provided critical blueprints for designing next-generation adsorbents. Moreover, the theoretical studies summarized above are not merely abstract calculations; they serve as essential pre-screening and mechanistic guides for experimentalists.
6.3. CNT-based CO2 adsorbents
CNTs can be classified into two primary types: single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs). SWCNTs consist of a single graphene sheet wrapped into a tube form, while MWCNTs comprise multiple graphene sheets wrapped concentrically.209 The synthesis of CNTs typically involves arc discharge, laser ablation, or CVD. Although arc discharge and laser ablation are known for producing high-quality CNTs, CVD enables the production of large quantities with the potential for alignment.This section of the review focuses on the role of CNTs in CO2 capture and discusses the most recent advancements in this area. Over the last two decades, CNTs have garnered significant attention for CO2 absorption due to their lightweight nature, high surface area, conductive properties, hydrophobicity, and tunable surface chemistry. The hydrophobicity of CNTs minimizes the impact of water interaction, while their hollow tubular structure increases the surface area for adsorption inside and outside the tube. The adsorption mechanism differs fundamentally between pristine and functionalized CNTs. Pristine CNTs rely on weak van der Waals forces (physisorption) and often suffer from low selectivity. In contrast, acid treatment (introducing –COOH groups) enhances surface polarity, while amine functionalization shifts the mechanism toward chemisorption. Primary amines facilitate the formation of carbamates, significantly increasing the heat of adsorption (Qst). However, excessive functionalization can lead to agglomeration and blockage of the inner nanotube channels, reducing the accessibility of internal pore volume to gas molecules.
Cinke et al.210 conducted a temperature-dependent study on SWCNTs synthesized via the HiPco process for CO2 adsorption. They found that the CO2 adsorption capacity of purified SWCNTs was twice that of activated carbon and raw HiPco at 35 °C. As with other carbon materials, amine functionalization is an effective method to enhance CO2 adsorption in CNTs. This method also benefits from lower energy requirements for CO2 desorption compared to traditional amine solutions such as monoethanolamine (MEA).211,212
Recently, Khoshraftar et al.213 reported modified MWCNTs, synthesized using Fe–Ni/AC catalysts to achieve exceptional CO2 adsorption capacity, reaching a maximum of 424.08 mg g−1 at 25 °C and 10 bar pressure. The catalyst modification, which reduced the surface area from 240 m2 g−1 (pristine MWCNTs) to 11 m2 g−1 (modified M-MWCNTs), paradoxically resulted in an increased adsorption capacity. This was attributed to the introduction of new adsorption sites and favorable interactions between functional groups, which enhance CO2 binding at lower temperatures.
Lourenco et al.211 compared pristine CNTs with COOH-functionalized and amine-functionalized MWCNTs, using N1–(3-trimethoxysilylpropyl) diethylenetriamine (DETASi) as the functionalizing agent. They found that pristine MWCNTs and MWCNT-OH could not adsorb CO2 due to the absence of functional groups. In contrast, COOH-functionalized MWCNTs adsorbed 0.1% CO2 per gram of material, indicating weak interactions between the carboxylic group and CO2. However, MWCNTs functionalized with DETASi showed a significant improvement in CO2 adsorption, reaching 2.11%. Specifically, CO2 adsorption capacities were 0.48 mmol g−1 and 0.43 mmol g−1 for CNT-COOH-DETASi and CNT-SD-DETASi, respectively. Due to their high surface area, CNTs also help mitigate amine accumulation issues and aid in the structural arrangement of nanoparticles. Ronghuan Xu et al.214 impregnated CNTs with silica nanoparticles and tetraethylenepentamine (TEPA) to enhance CO2 adsorption. The composite was heat-treated at 90 °C to remove moisture before CO2 adsorption. Fig. 13(b–e) reveals that, at 50% amine loading, the TEPA-impregnated CNT–SiO2 composite exhibited an increased CO2 adsorption capacity of 2.70 mmol g−1, compared to 2.18 mmol g−1 for the TEPA–SiO2 composite alone. This result highlights the role of CNTs in promoting pore filling and rearranging SiO2 nanoparticles at higher TEPA viscosities. The study also found that increasing the amine functionalization weight fraction (up to 60%) has further enhanced CO2 adsorption.
 | ||
| Fig. 13 (a) Experimental setup for the fabrication of M-MWCNTs (reproduced from ref. 213 under the Creative Commons Attribution (CC BY) license); effect of amine loading and support type on CO2 capture performance; (b) and (c) illustrate the CO2 adsorption rate, while (d) and (e) present the total CO2 uptake and amine efficiency, respectively, with experimental conditions: 0.2 vol% CO2 in N2, a gas flow rate of 100 ccm, and a temperature of 25 °C (reproduced from ref. 214 with permission from [Elsevier], copyright [2025]). | ||
Ying Wang et al.215 incorporated sulfur and nitrogen doping to further improve the CO2 adsorption capacity of CNTs by introducing dual heteroatoms. The CNTs were in situ grown from coal activated by KOH, followed by doping with nitrogen and sulfur atoms (KS-NTC). To enhance nitrogen concentration, dual urea treatment was carried out (KN-NTC). Both KN-NTC and KS-NTC exhibited high surface areas of 1789 m2 g−1 and 1875 m2 g−1, respectively, due to low pore sizes (<0.7 nm), high pore volumes (∼0.8 cm3 g−1), and additional defect structures. These defects were attributed to surface etching mediated by the reduction and activation of CNTs.215 The formation of g-C3N4 at 500 °C enhanced the graphitic character, and at higher temperatures (900 °C), it converted to ammonia, creating a porous structure and introducing N-doping. Fig. 14(a and b) shows TEM images of KS-NTC samples demonstrating a tube-like structure with open and closed end configurations, which shows intact structure integrity of carbon nanotubes formed from coal. The CO2 adsorption capacities of KN-NTC and KS-NTC were evaluated at 25 °C and 0 °C under 0.15 bar pressure of CO2 as shown in Fig. 14(c and d). KN-NTC showed capacities of 0.815 mmol g−1 at 25 °C and 1.6 mmol g−1 at 0 °C, while KS-NTC demonstrated higher adsorption capacities of 0.973 mmol g−1 at 25 °C and 1.829 mmol g−1 at 0 °C. At 1 bar pressure, both samples exhibited significantly higher adsorption capacities, KN-NTC at 5.81 mmol g−1 and KS-NTC at 5.66 mmol g−1 at 0 °C. Additionally, CO2 adsorption was far superior to N2 adsorption (∼0.5 mmol g−1), confirming the selectivity of the composite for CO2 over N2.
 | ||
| Fig. 14 (a and b) TEM images of the KS-NTC sample; (c) CO2 and N2 adsorption capacities of the carbon nanotube composite measured at 25 °C; (d) CO2 and N2 adsorption capacities at 0 °C (reproduced from ref. 215 with permission from [Elsevier], copyright [2024]). | ||
Due to their high surface area and lightweight properties, CNTs are highly effective even in very low quantities, making them cost-effective materials for CO2 capture. Mohammad Heidari et al.216 explored the use of small amounts of CNTs (2.5, 5, and 10 wt%) as additives in the development of CaZrO3–CaO xerogels, thereby improving their structural and textural properties for CO2 capture. This study investigated the effects of varying Ca/Zr molar ratios (15/1 and 30/1) on the performance of the xerogels to minimize the use of expensive zirconium precursors while maximizing CO2 capture efficiency. The inclusion of CNTs led to the formation of highly porous structures, significantly enhancing the durability and efficiency of CaO-based adsorbents during cyclic CO2 capture processes. The xerogels containing 5 wt% CNTs achieved the highest CO2 capture capacities: 0.164 g CO2 per g adsorbent for the 15/1 Ca/Zr ratio and 0.149 g CO2 per g for the 30/1 ratio, demonstrating substantial improvements over traditional CaO sorbents. After 15 cycles under challenging CO2 capture conditions, the total amount of captured CO2 increased from 2.01 g CO2 per g for the 15/1 ratio to 2.92 g CO2 per g and from 1.96 g CO2 per g to 3.01 g CO2 per g for the 30/1 ratio, reflecting excellent cyclic performance. The incorporation of CNTs reduced the crystallite sizes of CaO by 15.84% and 33.1% for the 15/1 and 30/1 ratios, respectively, contributing to a higher surface area and enhanced CO2 adsorption. Furthermore, the xerogels exhibited substantial increases in pore volume (50.57% for the 15/1 ratio and 90.55% for the 30/1 ratio), promoting greater CO2 diffusion and capture efficiency. Yuhang Zhang et al.217 introduced MWCNTs/carbon foam (CF) nanocomposites (MCF), synthesized from liquefied larch sawdust, providing a novel approach to utilizing biomass for the creation of advanced materials. These MCF nanocomposites exhibit a unique hierarchical porous structure with ultra-micropores (0.50–0.80 nm) and mesopores (approximately 3.70–3.90 nm), thereby enhancing their potential for gas adsorption. The MCF-2 sample demonstrated impressive CO2 adsorption capacities of 4.58 mmol g−1 at 0 °C and 3.19 mmol g−1 at 25 °C, indicating its effectiveness in CO2 capture. At 25 °C, MCF-2 exhibited a CO2/N2 selectivity ratio of 23.71, highlighting its potential for CO2 separation from nitrogen in practical applications. The MCF nanocomposites showed high isosteric heats of adsorption, ranging from 23.32 to 36.48 kJ mol−1, suggesting strong interactions between CO2 molecules and the material, which is beneficial for efficient capture. Furthermore, MCF-2 demonstrated excellent recyclability, maintaining its CO2 capture capacity over multiple adsorption–desorption cycles, indicating low energy requirements for regeneration. Incorporating MWCNTs also significantly enhanced the mechanical strength of the carbon foam, resulting in a 113% increase in compressive strength compared to traditional biomass-based carbon foams. Kavitha Ramadass et al.26 highlighted the potential of naturally occurring materials, such as halloysite nanotubes, for creating advanced carbon nanostructures with superior properties for CO2 capture. Activated halloysite nanocarbon (AHNC) exhibited an impressive CO2 adsorption capacity of 25.7 mmol g−1 at 0 °C and 30 bar pressure, significantly outperforming other materials, such as mesoporous carbon, activated carbon, and MWCNTs. This high capacity is attributed to the material's large surface area and unique pore structure. The AHNC exhibited a specific surface area of 1646 m2 g−1, 74 times greater than the raw halloysite's surface area (22.5 m2 g−1). Additionally, the AHNC exhibited excellent cycling stability, retaining its CO2 capture efficiency over multiple adsorption–desorption cycles. It is highly suitable for practical applications, as it suggests that the material can be reused without significant performance loss.26 Immobilizing active materials on high-surface-area materials, particularly porous ones, can provide many active sites for CO2 capture. Yingjun Li et al.218 proposed a core–shell material composed of CNTs with ionic polymers and a porous structure, featuring activated epoxide rings for enhanced catalytic activity. They prepared bipyridine-functionalized Zn(II) ionic polymers, which showed enhanced performance in CO2 capture. Table 8 summarizes the functionalization strategies, specific surface areas, and corresponding CO2 adsorption performance parameters of CNT-based adsorbents reported in the literature. This comparison clarifies how surface functionalization affects adsorption capacity, selectivity, and kinetics, providing critical insights into structure–property relationships and guiding the rational design of high-efficiency CNT-based sorbents for advanced CO2 capture applications.
| CNT type | Functionalization/treatment | Surface area (m2 g−1) | CO2 capacity (mmol g−1) | Q st (kJ mol−1) | Selectivity (CO2/N2) | Ref. |
|---|---|---|---|---|---|---|
| Note: the gas composition (e.g., purity and mixed-gas environments) and operating pressures used across different studies may vary. | ||||||
| MWCNTs | Produced via CVD and modified by Fe–Ni/AC catalysts | 240, but after modification, it was reduced to 11 | 424.08 mg g−1 at 25 °C and 10 bar | — | — | 213 |
| f-MWCNTs | Functionalized MWCNTs by a simultaneous combination of two amines | 155 | 2.35 (modified MWCNTs) and 1.48 (MWCNT–COOH) | — | — | 219 |
| f-MWCNTs | Functionalized MWCNTs with 1,6-diaminohexane | 262.69 | 253.99 mg g−1 at 30 °C and 17 bar | 21–17 | 212 | |
| Silica particle -MWCNT | TEPA impregnation | — | 2.70 (Si-MWCNT-TEPA) | — | — | 214 |
| 2.18 (Si-TEPA) | ||||||
| Heteroatom doping | N(KN-NTC), doped carbon nanotubes | 1789 | 3.31 (25 °C) and 5.81 (0 °C) at 1 bar | 27–34 | 19 | 215 |
| Heteroatom doping | N-S(KS-NTC) doped carbon nanotubes | 1875 | 3.63 (25 °C) and 5.66 (0 °C) at 1 bar | 31–27 | 18 | |
| COOH-functionalized | Commercial | 121 | 0.1% CO2 g−1 | — | 0% | 211 |
| CNT-COOH-DETASi | Reflux for 16 h | 74 | 0.48 | — | 1.46% | |
| CNT-SD-DETASi | Reflux for 16 h | 88 | 0.25 | — | 1.89% | |
| MWCNT (5%)-CaZrO3-CaO xerogels | Heat treatment and dessication at 150 °C (3 h) and 850 °C (1.5 h) | 17.83 | 0.164 g (CO2 g−1) (Ca/Zr 15) | — | — | 216 |
| 20.63 | 0.149 g (CO2 per g) Ca/Zr 30) | |||||
6.4. Graphene-based adsorbents
Graphene, a 2D carbon material, is considered one of the most explored allotropes of carbon due to its unique structure, which is sp2-hybridized and consists of a single atomic-thick layer of carbon atoms.220–223 Single- or bilayer graphene can be synthesized using CVD, but the top-down wet chemical method is often preferred for large-scale production. This approach produces GO, which can be reduced and exfoliated to yield multilayer or few-layer graphene.224,225 The presence of defects in graphene or GO enhances their CO2 adsorption properties. Additionally, the 2D structure of graphene provides a high surface area, and its properties can be tuned to optimize CO2 capture.This section focuses on recent advancements in CO2 capture using graphene-based materials. Graphene and GO are widely studied for CO2 adsorption, infiltration, and capture.226–229 However, pure graphene with few layers tends to have low CO2 adsorption capacity due to its multi-layered structure, which acts as a diffusion barrier. Furthermore, its hydrophobic and nonpolar nature results in low selectivity towards CO2.230 To improve CO2 capture, several strategies have been employed, including tuning the surface defects and increasing the surface area of GO and reduced graphene oxide (rGO).231 Techniques including physical activation, hydrothermal treatment, chemical treatment, thermal treatment, and plasma treatment have been explored to enhance their adsorption properties. Graphene derivatives can adsorb CO2 through physisorption and chemisorption, and the adsorption route is primarily governed by the pore structure, surface area, and functional groups (oxygen, nitrogen, and sulfur) present on the graphene sheets. GO, being oxygen-rich, can support chemisorption, whereas rGO's defect structure favours physisorption.232 However, chemisorption is most effective under low CO2 pressures or when sufficient adsorption sites are available; otherwise, physisorption predominates.
Surface modification of GO through UV activation has been reported by Anish Mathai Varghese et al.233 to improve CO2 adsorption. In their study, a Cu-BTC MOF/GO composite was developed, and it was observed that the surface area of the pure Cu-BTC MOF decreased after growth on GO. However, the surface area was restored when the GO was subjected to UV irradiation for 10 minutes. The Cu-BTC MOF/UV-GO composite exhibited a 45% increase in CO2 adsorption capacity, reaching 5.14 mmol g−1 at 25 °C and 1 bar, compared to 3.55 mmol g−1 for the pure Cu-BTC MOF.233 At 0 °C, the adsorption capacity increased to 9.5 mmol g−1. Additionally, the CO2/N2 selectivity for UV-activated GO was 21, compared to 19.32 for the pure Cu-BTC MOF.
Similar to other carbon-based adsorbents, graphene-based materials have been functionalized with amine groups to further enhance CO2 adsorption.234–236 Amination of graphene is challenging due to the lack of oxygenated groups, but GO, with its abundant oxygen-containing functional groups, offers more opportunities for functionalization. rGO, depending on the production method, can retain these oxygenated groups while also maintaining conductivity, making it more suitable for amine linkage. Polyethyleneimine (PEI) is often used as an amine precursor to form composites with rGO. These PEI–rGO composites have demonstrated high CO2 uptake at low pressures, with one study reporting an uptake of 0.61 mmol g−1 at 4 pKa of CO2. At higher CO2 pressures (101 kPa), the adsorption capacity increased to 1.03 mmol g−1. Interestingly, at 50 °C, the adsorption capacity at low pressure decreased by 72%, but at high pressure, it increased by 50%, reaching 1.55 mmol g−1. This behavior is attributed to an increase in the number of available CO2 adsorption sites at higher temperatures and pressures.226
Due to their stability, primary amines such as 3-aminopropyltrimethoxysilane (APTES) are particularly effective for amine linkage.237 MP Jerome et al.238 explored the functionalization of cellulose-coated GO with APTES to create a composite with a 3D porous structure and abundant oxygen functionalities. Waste white paper was used to extract cellulose (84.9%) through bleaching and alkali treatment. The cellulose-coated GO was then functionalized with APTES and freeze-dried to form an aerogel. Fig. 15 presents SEM images of paper, extracted cellulose, cellulose aerogel, GO aerogel, CGO, CGO-0.25 APTES, and CGO-1.5 APTES.
 | ||
| Fig. 15 SEM images of (a) raw paper, (b) extracted cellulose, (c) the cellulose aerogel, (d) the graphene oxide (GO) aerogel, (e) the cellulose–GO (CGO) aerogel, (f) the CGO aerogel modified with 0.25% APTES, and (g) the CGO aerogel modified with 1.5% APTES (reproduced from ref. 238 with permission from [Elsevier], copyright [2025]). | ||
The rough surface of the paper is attributed to residual impurities from the papermaking process, such as lignin, calcite, and other additives. After alkali and bleaching treatments, the cellulose fibers become fibrillated and free from contaminants. Notably, extracted cellulose exhibits increased porosity along its fiber walls, likely due to the removal of lignin and hemicellulose, which enhances the flexibility of the fibers. Some pores may also be intrinsic, aiding plant fiber physiology. The native cellulose aerogel exhibits a 3D porous network, potentially offering a higher surface area than compact fibers, though this depends on cellulose content. In contrast, the GO aerogel shows a wrinkled, sheet-like morphology. The CGO aerogel combines these features with GO flakes anchored to cellulose fibrils in a porous matrix. Strong cellulose–GO interactions and high GO content (33.3 wt%) result in loosely layered pore walls rather than a rigid fibrous structure. APTES modification induces subtle morphological changes (Fig. 15f–g). With increasing APTES concentration, the GO sheets and cellulose fibrils appear more compact and integrated. The FTIR peak at 1565 cm−1 confirmed the successful formation of an amine linkage. Plain GO exhibited a CO2 capture capacity of 0.57 mmol g−1, higher than plain cellulose (0.2 mmol g−1). The cellulose–GO composite functionalized with APTES showed a remarkable increase in adsorption capacity, reaching 2.52 mmol g−1 at 0.25% APTES and 1.34 mmol g−1 at 1.5% APTES, significantly outperforming both cellulose and GO. This enhancement is attributed to the increased basicity of the amine groups, which facilitates chemisorption. The chemisorption mechanism is explained through specific equations detailing the interaction between CO2 and the amine groups on the surface.
| RNH2 + CO2 ↔ RNH2+COO− | (a) |
| RNH2 + RNH2+COO− ↔ RNHCOO− + NH3+ | (b) |
Further adsorption of CO2 in the pores of graphene derivatives occurs through physisorption. However, pore blocking and self-polymerization can occur at higher concentrations of amine groups, as seen in the case of 1.5% APTES, which reduces the surface area and decreases performance. The plasma technique (cold plasma) is another non-chemical method that has gained attention. This technique utilizes various gases, such as nitrogen (N2), argon (Ar), ammonia (NH3), and hydrogen (H2), to induce pore formation, functionalization, and morphological improvements in graphene without altering its primary structure. Navik et al.239 applied cold plasma surface modification to graphene-based aerogels using a N2/H2 mixture (0.5 mL min−1) at treatment times ranging from 2.5 to 10 minutes. The plasma treatment introduced C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vacancies and functionalized the graphene surface with amine groups, creating nanovoids and edges. This process occurs due to highly energized nitrogen ions, atoms, or molecules interacting with the graphene surface, replacing carbon atoms with N–H groups. The CO2 adsorption capacity of the treated graphene increased significantly, from 1.6 mmol g−1 to 3.3 mmol g−1 (when exposed to simulated flue gas) and 1.3 mmol g−1 (DAC). Additionally, the selectivity for CO2 improved dramatically from 42 to 87 after just 5 minutes of plasma treatment. The treatment also enhanced the load-bearing capacity of the graphene aerogel.
C vacancies and functionalized the graphene surface with amine groups, creating nanovoids and edges. This process occurs due to highly energized nitrogen ions, atoms, or molecules interacting with the graphene surface, replacing carbon atoms with N–H groups. The CO2 adsorption capacity of the treated graphene increased significantly, from 1.6 mmol g−1 to 3.3 mmol g−1 (when exposed to simulated flue gas) and 1.3 mmol g−1 (DAC). Additionally, the selectivity for CO2 improved dramatically from 42 to 87 after just 5 minutes of plasma treatment. The treatment also enhanced the load-bearing capacity of the graphene aerogel.
In recent years, there has been growing interest in using waste materials to synthesize graphene for CO2 capture.225 Bryan E. Arango Hoyos et al. synthesized graphene oxide (GO) foam from commercial bamboo using double thermal decomposition. The GO foam was carbonized at varying temperatures (873 K, 973 K, and 1073 K), resulting in different oxidation levels, as determined by XPS analysis. The degree of oxidation varied from 9% at 873 K (GO 873 K) to 3% at 1073 K (GO 1073 K). The results showed that a higher degree of oxidation led to increased surface area, with GO 873 K reaching 570.9 m2 g−1. GO foam exhibited superior CO2 retention performance compared to traditional adsorbents, such as zeolite and silica gel. After 2500 seconds of CO2 adsorption, no desorption was observed even after 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 seconds, demonstrating the material's ability to retain CO2 at room temperature. Complete desorption occurred at 673 K, making the GO foam highly reusable. The CO2 adsorption efficiency was the highest for GO 1073 K (92.20%), followed by GO 973 K (89.38%) and GO 873 K (86.28%).227 While the 2D structure of graphene offers a high surface area, it can also limit gas transport when used in pellet form. Researchers have explored the use of one-dimensional graphene nanoribbons (GNRs) to address this issue. These nanoribbons enable enhanced gas transport due to their complex structure. Fau-type zeolites (faujasites) combined with graphene oxide (GO) nanoribbons have shown an advantage in pelletization without structure-directing agents. CO2 adsorption tests, using a CO2:N2 mixture (15:85), showed that zeolite pellets with 12% GO nanoribbons exhibited a CO2 uptake of 4.61 mmol g−1 with high selectivity for CO2 (76.5) compared to N2.228 Furthermore, CO2 desorption occurred in the 125–150 °C temperature range, allowing for complete material regeneration. Nitrogen flow during the regeneration process also made the process energy-efficient.
000 seconds, demonstrating the material's ability to retain CO2 at room temperature. Complete desorption occurred at 673 K, making the GO foam highly reusable. The CO2 adsorption efficiency was the highest for GO 1073 K (92.20%), followed by GO 973 K (89.38%) and GO 873 K (86.28%).227 While the 2D structure of graphene offers a high surface area, it can also limit gas transport when used in pellet form. Researchers have explored the use of one-dimensional graphene nanoribbons (GNRs) to address this issue. These nanoribbons enable enhanced gas transport due to their complex structure. Fau-type zeolites (faujasites) combined with graphene oxide (GO) nanoribbons have shown an advantage in pelletization without structure-directing agents. CO2 adsorption tests, using a CO2:N2 mixture (15:85), showed that zeolite pellets with 12% GO nanoribbons exhibited a CO2 uptake of 4.61 mmol g−1 with high selectivity for CO2 (76.5) compared to N2.228 Furthermore, CO2 desorption occurred in the 125–150 °C temperature range, allowing for complete material regeneration. Nitrogen flow during the regeneration process also made the process energy-efficient.
A novel 2D allotrope of carbon, known as holey penta-hexagonal graphene (HPhG), was also investigated for CO2 adsorption due to its ample pores and superior stability. Although bare HPhG exhibits a CO2 adsorption capacity of 2.46 mmol g−1, researchers have been working on composites of HPhG to enhance its performance. Tiantian Quio et al. developed holey GO composites doped with alkali earth metals (Li, Na, K, Ca, and Mg) to improve the adsorption capacity of HPhG. Among the doped composites, Ca-doped HPhG exhibited the highest CO2 adsorption capacity of 11.51 mmol g−1, followed by Li-doped HPhG (10.84 mmol g−1), Na-doped HPhG (9.73 mmol g−1), K-doped HPhG (9.73 mmol g−1), and Mg-doped HPhG (9.67 mmol g−1). This enhancement is attributed to the strong van der Waals and coulombic interactions between CO2 and the metal-doped HPhG surface. The intense quadrupole moment and polarizability of the CO2 molecules contribute to the high selectivity of the doped composite towards CO2, especially in the presence of methane and nitrogen gases. A thorough summary of the structure–property–performance relationships of GO-based adsorbents for CO2 capture is presented in Table 9, which includes key factors such as specific surface area, adsorption capacity, selectivity, and isosteric heat of adsorption (Qst). This comparative study provides important insights for the logical design and optimization of high-performance GO-based sorbents by highlighting how customized structural engineering and surface functionalization control adsorption thermodynamics, affinity, and separation efficiency.147
| Type of material | Treatment | Surface area (m2 g−1) | CO2 adsorption (mmol g−1) | Q st (kJ mol−1) | Selectivity | Ref. |
|---|---|---|---|---|---|---|
| Note: the gas composition (e.g., purity and mixed-gas environments) and operating pressures used across different studies may vary. | ||||||
| Cu-BTC MOF/GO | UV activation | 1323.49 | 5.14 (25 °C) | 22.5 | 21 (0.1 bar) | 233 |
| 9.5 (0 °C) | 14.35 (1 bar) CO2/N2 | |||||
| PEI–rGO composite | Amine functionalization (71%) | — | 0.61 at 4 pKa | — | — | 226 |
| 1.03 at 101 pKa | ||||||
| 1.55 (50 °C) | ||||||
| Holey graphene (HPhG) | Li, Na, K, Mg, Ca doped | — | 11.51 (Ca-HPhG) | 43.37 | ∼2385 (CO2/CH4) | 147 |
| ∼1366 (CO2/N2) at 298 K 1 bar | ||||||
| Graphene nanoribbon (GNR) | Fau-zeolite + GNR (12%) pellet | 613 | 4.61 | — | 76.5 (CO2/N2) | 228 |
| Graphene oxide | Ionic liquid and C4mimBF4 | — | 247.99 GPU permeation | — | 13.58 (CO2/H2) | 229 |
| 44.82 (CO2/N2) | ||||||
| 75.45 (CO2/CH4) | ||||||
6.5. Carbon foam
Carbon foam is a porous 3D carbon material characterized by interconnected pores, lightweight properties, and high compressive strength.240 It can be synthesized using various methods, including the template method or by incorporating surfactants, foaming agents, and catalysts. Carbon foam is typically produced from resins and polymers such as phenolic resin, coal tar pitch, and other synthetic polymers.Diego Fernando et al.241 synthesized carbon foam using novolac-type phenolic resin. In their process, n-pentane and dichloromethane were used as foaming agents, while NaOH, NH3, and phosphoric acid served as catalysts. The presence of the –OH bond (900–3650 cm−1) indicated the phenolic group, and two peaks near 2980 cm−1 and 3000 cm−1 confirmed the formation of a methylene bridge during the polymerization of the resin to form a carbon foam. Carbon foam synthesized at 600 °C using phosphoric acid and DCM exhibited the highest surface area (984 cm2) and, consequently, the highest adsorption capacity (103.3 mg g−1). Recently, the thermal transformation of biowaste has also been explored as a method to produce carbon foam, offering both structural benefits and the added advantage of waste recycling. Beizhang et al.242 used palm kernel shells to prepare a multiporous carbon foam. Fig. 16(a–c) illustrates the schematic representation of the synthesis process for lignin-based carbon foam derived from palm kernel shells, along with the proposed reaction mechanism. The carbon foam obtained through this method was subjected to high-temperature heat treatment (600 °C), which increased CO2 adsorption. This improvement was attributed to the foam's multiporous structure (macro- and mesoporous), which enhanced CO2 accommodation by increasing diffusion capacity.
 | ||
| Fig. 16 (a) Schematic representation of the synthesis process for lignin-based carbon foam derived from palm kernel shells; (b and c) proposed reaction mechanism illustrating the key steps involved in the formation of the carbon foam structure (reproduced from ref. 242 with permission from [Elsevier], copyright [2022]); GO foams obtained employing the DTD and characterization methods, (d) 873.15 K (GO 9.00%), (e) 973.15 K (GO 5.25%) (reproduced from ref. 227 under the Creative Commons Attribution (CC BY) license). | ||
Hoyos et al.227 investigated the potential of eco-friendly GO foams, produced from bamboo waste, as effective adsorbents for carbon capture. The GO foams were synthesized as shown in Fig. 16(d–e)via a double thermal decomposition process, with varying temperatures employed to regulate oxidation levels. The foams were subsequently assessed through experimental CO2 adsorption tests and DFT simulations. The results indicated that the GO foams exhibited significantly enhanced CO2 capture efficiency, ranging from 86.28% to 92.20%, compared to traditional adsorbents such as zeolite and silica gel. The foam with 3% oxidation showed the best adsorption efficiency, while the foam oxidized at 9% had the highest material yield. DFT calculations indicate that CO2 capture primarily occurs through physisorption. This process is driven by weak van der Waals forces, with hydroxyl (–OH) groups playing a key role. In addition, the GO foams demonstrated excellent regenerability, completely regaining their adsorption capacity after thermal desorption at 673.15 K.
Zhou et al.243 developed hierarchically porous N-doped carbon foams (HPNCFs) for efficient CO2 capture by maximizing nitrogen content and microporous surface area through precise control of carbonization temperature and the PBC/His molar ratio. Among the samples, HPNCF-1.0–700 exhibits optimal performance, delivering CO2 adsorption capacities of 3.06 mmol g−1 (25 °C) and 4.13 mmol g−1 (0 °C) at 760 torr, along with high uptake under flue–gas conditions (0.81 mmol g−1 at 114 torr) and an exceptional CO2/N2 selectivity of 24. Raising the carbonization temperature from 700 °C to 900 °C increases the total surface area but sharply reduces nitrogen content, micropore surface area, CO2 capacity, and selectivity. This trend highlights the dominant role of micropores (0.77–1.9 nm) and nitrogen functionalities, particularly pyrrolic N, in CO2 adsorption, which outweighs the benefits of increased macroporosity. The relatively high isosteric heat of adsorption (26.5 kJ mol−1) indicates strong yet reversible physisorption driven by dipole–π interactions.
A study conducted by Vorokhta et al.244 reveals a clear trade-off between nitrogen doping and porosity in carbon foams, where increasing nitrogen content enhances CO2 affinity but progressively suppresses pore development, necessitating careful optimization. Among the samples, the triethanolamine-derived nitrogen-doped carbon foam (NCF-TEA, ∼7 at% N) delivered the highest CO2 uptake (5.14 mmol g−1 at 273 K, 100 kPa), outperforming the undoped foam despite possessing a substantially lower surface area. In contrast, further nitrogen enrichment (∼13 at% in NCF-MEA) severely reduced micropore volume and specific surface area, leading to diminished adsorption capacity due to pore blockage during pyrolysis. The enhanced performance at optimal doping arises from a synergistic interplay between pore architecture and surface chemistry. CO2 uptake is governed by ultra-micropores (<0.7 nm), where overlapping adsorption potentials maximize confinement, while nitrogen functionalities (pyridinic, pyrrolic, and amine groups) act as Lewis base sites that strengthen CO2 binding via acid–base and hydrogen-bonding interactions. This chemical enhancement compensates for surface area loss up to the optimal nitrogen level.
In advanced CNM-based adsorbents, the way pores are connected hierarchically, rather than just the total surface area, has become the most important factor in determining how quickly substances are adsorbed, how selective the process is, and how easily the adsorbent can be reused.245 By seamlessly integrating macropores and mesopores with microporous areas, diffusion limitations are reduced, mass transport is accelerated, and the active adsorption sites are used almost completely.246,247 In this structural context, macropores serve as pathways for transport, while the selectivity for CO2 is largely determined by the distribution of micropore sizes (less than 2 nm) and the surface chemistry, specifically the presence of nitrogen-containing functional groups that increase affinity via Lewis acid–base interactions.248 Because CO2 uptake occurs quickly and can be easily reversed through physisorption, these materials require less energy to regenerate and exhibit excellent stability over multiple cycles, which is superior to traditional chemisorbents.
In addition to how well they adsorb, the mechanical and thermal stability of materials are crucial for their practical use. Carbon foams provide a clear engineering advantage over fragile aerogels and loose powders, which are prone to wear, compaction, and inefficient heat transfer.248 The interconnected strut network, which is rigid, ensures structural integrity while also allowing for a quick thermal response. This is crucial for temperature swing adsorption (TSA) processes. Because carbon foams conduct heat so effectively, they can withstand repeated heating and cooling cycles without degrading. Experimental studies demonstrate that hierarchical porous carbons and foams can retain over 95% of their capacity after more than 50 cycles of adsorption and desorption, supporting these benefits.244,249 The combined results underscore the importance of hierarchical pore connectivity, along with structural and thermal stability, as key design principles for the next generation of multifunctional CO2 capture systems The structure–property–performance correlations of various carbon-form-based adsorbents for CO2 capture are summarized in Table 10, highlighting the effects of material type and post-treatment techniques on specific surface area, CO2 adsorption capacity, and CO2/N2 selectivity. To optimize carbon-based sorbents for high-efficiency, selective CO2 separation, this comparative analysis clarifies key structure-function relationships and provides useful design guidelines.
| Type of material | Treatment | Surface area (m2 g−1) | CO2 adsorption (mmol g−1) | Selectivity | Ref. |
|---|---|---|---|---|---|
| Note: the gas composition (e.g., purity and mixed-gas environments) and operating pressures used across different studies may vary. | |||||
| Novalac-type phenolic resin | 600 °C using phosphoric acid and DCM | 984 m2 g−1 | 103.3 mg g−1 | — | 241 |
| Palm kernel shells | Hydrothermal carbonization and acid-alkali treatment | 335.97 m2 g−1 | 1.25 mmol g−1 (35 °C 1 bar) | 34 | 242 |
| 0.86 mmol g−1 (50 °C 1 bar) | |||||
6.6. Carbon dots
Carbon dots (CDs) are zero-dimensional nanomaterials with ultra-low sizes (1–10 nm) and multifunctional properties, positioning them as promising candidates for CO2 capture applications. While their potential has been recognized, CDs have historically been underutilized compared to their carbon-based counterparts. Recent advances demonstrate substantial progress in optimizing their performance through composite architectures, heteroatom doping, and pilot-scale validation.The foundational work of Sahoo et al.250 utilized amine-doped carbon dots for CO2 capture from a 30 L automated pilot plant. They synthesized CDs from citric acid and ethylenediamine and then blended them with methyl diethanolamine, piperazine, and 2-amino-2-methyl-1-propanol as amine precursors. The CO2 source was flue gas (10% CO2). The solvent system consisting of amine-blended CDs exhibited enhanced CO2 adsorption, with the capacity increasing as the concentration of CDs increased to 100 mg L−1. This improvement is attributed to enhanced Brownian motion, which increases the mass transfer rate. However, agglomeration occurs at higher concentrations, which inhibits mass transfer and diminishes performance. Despite this, the CDs maintained stable CO2 capture efficiency for over 200 hours without significant degradation. Samandari et al.251 used lignin-derived carbon dots functionalized with oxygen and nitrogen groups for CO2 capture. Simulation studies revealed that in a single gas environment, the CO2/N2 and CO2/O2 selectivity ratios were 3.6 and 6.7, respectively, at 300 K. Additionally, Broud et al.252 also observed the effectiveness of amine-functionalized CDs, with selectivity values of 4.1 for CO2/N2 and 3.1 for CO2/O2. Notably, it was found that in the case of flue gas, the selectivity for CO2 was higher compared to pure gas mixtures. This is attributed to CO2 having a higher displacement for N2 and O2 compared to CO2 itself. Although CO2 preference is higher than that for N2 and O2, the selectivity toward N2 and O2 depends on the positioning of the functionalities, with interior hydroxyl functionalization increasing the preference for N2 over O2. The performance drop observed at higher CD concentrations is primarily attributed to particle agglomeration.253 As the concentration increases, strong van der Waals forces and hydrogen bonding between surface groups cause the 0D dots to cluster. This aggregation not only reduces the effective surface area available for gas interaction but can also mask active amine/functional sites buried within the clusters (Fig. 17).254
 | ||
| Fig. 17 (a) Representative snapshot from molecular simulation illustrating CO2–carbon interactions. Color scheme: carbon atoms of graphite and CO2 (gray), carbon atoms of carbon quantum dots (CQDs, green), oxygen (red), and hydrogen (white) (reproduced from ref. 251 with permission from [American Chemical Society], copyright [2024]); photographic images of amine-stable carbon dots under (b) visible light and (c) UV irradiation, demonstrating their optical response. TEM micrographs of (d and e) water-stable carbon quantum dots and (f, g) amine-stable carbon quantum dots; (h) Effect of CD loading on CO2 absorption capacity in a mixed amine solution (reproduced from ref. 250 with permission from [Elsevier], copyright [2025]). | ||
In summary, recent advances reveal the transformative potential of CDs as a scalable, highly selective, and previously underexploited platform for CO2 capture. Moving beyond laboratory-scale demonstrations, the pilot-scale validation reported by Sahoo et al.250 represents a pivotal milestone toward industrial implementation. The study shows that amine-blended CD systems significantly enhance gas–liquid mass transfer while maintaining structural and functional stability for over 200 hours under realistic flue gas conditions containing 10% CO2. Complementary investigations by Samandari et al.251 and Broud et al.252 further establish the chemical tunability of CDs, demonstrating that targeted oxygen and nitrogen-based functionalization, and, critically, the spatial distribution of these groups, can be precisely engineered to achieve high CO2 selectivity over N2 and O2. This level of control over surface chemistry positions CDs as a uniquely adaptable class of adsorbents and absorbents, capable of bridging molecular-scale interactions with process-level performance.
Although both carbon dots and fullerenes (e.g., C60) belong to the family of zero-dimensional carbon allotropes, CDs offer several decisive advantages for CO2 capture applications. Most notably, CDs are intrinsically hydrophilic, owing to abundant surface hydroxyl, carboxyl, and amine functionalities. This property enables their seamless dispersion in aqueous amine solvents, forming efficient CD-based nanofluids that enhance CO2 absorption kinetics without requiring the complex surface modifications typically necessary for hydrophobic fullerenes, which often rely on toxic organic solvents for processing.250 From a manufacturing perspective, CDs are inherently more scalable and cost-effective. They can be synthesized via simple, low-temperature bottom-up routes using inexpensive and renewable precursors such as citric acid or lignin.255 In contrast, fullerene production typically involves energy-intensive arc-discharge methods, followed by costly purification steps, which limit its economic feasibility at scale.256,257 Finally, the structurally disordered and defect-rich surfaces of CDs facilitate versatile heteroatom doping (N, S, and P), allowing fine-tuning of CO2 binding strength and selectivity.251 Fullerenes, while chemically robust, possess rigid and stoichiometric structures that are far less amenable to the flexible surface engineering required for optimized CO2 capture. Collectively, these advantages position carbon dots as a practically superior and industrially relevant alternative to other zero-dimensional carbon materials for next-generation CO2 capture technologies.
7. Techno-economic analysis of CNMs for CO2 capture technologies
Aqueous amine-based absorption technology for CO2 capture is currently at a Technology Readiness Level (TRL) of 9 and is a well-established method. Nevertheless, the primary challenges associated with this technology stem from its thermodynamic and economic limitations. Solvent regeneration presents a considerable challenge, driven by substantial energy costs (3.5–4.5 GJ per tonne CO2), thermal breakdown, and the production of hazardous byproducts. Consequently, the materials science field has focused on enhancing this technology through the application of CNMs as innovative adsorbents. This section will examine the various research and techno-economic data published over the last five years.CNM-based CO2 capture adsorbents, which are produced from agricultural and animal waste, present a promising avenue for value-added applications. The reported production costs for these materials range from USD 1.44 to 3.12 per kg, depending on the specific characteristics of the feedstock and the activation method employed.258–260 A key factor influencing both cost and efficacy is the selection of either physical or chemical activation, with the associated trade-offs being significantly dependent on the precursor material. Pricing analyses reveal that physically activated carbons typically demonstrate lower and more stable market prices across a diverse array of raw materials, approximately USD 0.96 to 1.92 per kilogram.261 This cost advantage is attributable to the streamlined process flows, the lack of corrosive chemical reagents, and the reduced need for extensive post-treatment procedures. Therefore, physical activation is particularly well-suited for large-scale, cost-sensitive applications, especially when utilizing waste-derived materials such as used tires and carbon black.
In contrast, chemical activation usually results in higher product costs, particularly for fossil fuel-derived precursors such as petroleum coke (USD 5.76 kg−1), lignite (USD 4.22 kg−1), and charcoal (USD 3.84 kg−1).261 These increased costs are mainly due to reagent use (e.g., H3PO4 and KOH), the need for corrosion-resistant reactors, extensive washing steps, and wastewater treatment. However, chemical activation often yields better textural properties, including a larger surface area, increased microporosity, and a higher carbon yield at lower activation temperatures. These performance enhancements can justify the increased expense in high-value applications, such as gas separation, energy storage, and catalysis. It is significant that biomass-derived precursors, such as wood, exhibit similar product costs for both activation methods (approximately USD 1.54 kg−1), implying that advantageous feedstock chemistry can, to some extent, mitigate the cost disadvantages linked to chemical activation. Consequently, these observations suggest that physical activation is most suitable for commodity-grade, high-throughput production, while chemical activation is strategically beneficial for performance-focused markets, thereby underscoring the need to select a precursor-specific and application-oriented process.261
Furthermore, enhancing the commercial feasibility of activated carbons derived from biomass and waste necessitates a combination of process intensification and targeted market positioning. Scaling up production to large-capacity facilities (>50 t day−1) can significantly reduce unit costs through improved thermal efficiency, bulk procurement, and streamlined logistics. In parallel, upgrading existing activation routes through energy recovery and chemical recycling has the potential to lower production costs by 15–25%. Additional savings can be achieved by reducing capital expenditure through shared utilities, co-locating carbon production with existing industrial infrastructure, and prioritizing locally available feedstocks to stabilize supply chains and transportation costs. Furthermore, hybrid activation strategies that combine steam and chemical activation warrant attention, as they enable a more balanced trade-off between pore development, reagent consumption, and overall cost–performance metrics. Finally, integrating life cycle assessment (LCA) into process design is essential for identifying emission hotspots, quantifying environmental benefits, and substantiating sustainability claims, including eligibility for carbon credits and compliance with emergent regulatory frameworks.
8. Comparative analysis of CNMs over commercial MOFs for CO2 capture
This section presents a comparative evaluation of MOFs and CNMs for CO2 capture technologies, highlighting their comparative features in Table 11. MOFs are already being used commercially because they possess a dual adsorption mechanism that combines open metal sites with framework-specific binding, offering a capacity of up to 12 mmol g−1.262,263 CNMs, on the other hand, primarily utilize physisorption, which is reinforced by interactions with heteroatoms. They also exhibit excellent chemical and thermal stability in harsh flue gas environments, maintaining structural integrity even in the presence of moisture, SOx, NOx, and elevated temperatures. CNMs are inexpensive ($0.01–50 per kg), readily available, and can be produced in bulk quantities, compared to MOFs, which are more expensive ($10–200 per kg).125 However, CNMs currently have a lower capacity, but their performance is significantly improving through four primary engineering approaches. Heteroatom doping introduces active sites, such as pyridinic, pyrrolic, and graphitic nitrogen, which increases CO2 affinity by nearly 35%.264,265 Dual N, S-doping in graphene results in a good adsorption capacity for CO2 capture under mild conditions (<100 °C, 1 atm) due to extra charge transfer from graphene to CO2.266 It is expected that the N–B co-doped systems will be available for sale in the next 5 years. Hierarchical pore engineering further enhances the adsorption kinetics of CNM-based adsorbents by combining micropores (<1 nm) for high-density uptake with mesopores (2–50 nm) for rapid diffusion, allowing for 95–98% retention after 100 cycles using optimized KOH activation strategies.267 Amine functionalization creates a chemisorption pathway, resulting in the highest CO2 capacities (5–8 mmol g−1).268 Grafted amine systems are more durable and drop less than 6% of their capacity over five cycles. But they remain sensitive to moisture. New strategies, such as tri-doping (N + B + O), the addition of single-atom metal sites, and operando electrochemical doping, are expected to further enhance performance and adsorption capacities up to 5–7 mmol g−1.269 These combined engineering methods provide an emerging pathway toward scalable, industrial-grade CNMs for next-generation CO2 capture systems. Hybrid systems that combine carbon supports with MOF coatings offer the best features of both materials, significantly enhancing performance. For instance, two hybrid materials composed of MOFs with MWCNTs exhibit an increase in adsorption capacity, from 64% to 76% (i.e., 5.8 to 9.52 mmol g−1 and from 4.53 to 8 mmol g−1), respectively, at 298 K and 18 bar. This increment has been attributed to the increase in micropore volume of MOFs by the incorporation of MWCNTs.270 Hence, it can be concluded that MOFs are the most effective performers and are employed commercially. However, engineered CNMs are rapidly improving and becoming strong candidates for scalable, next-generation CO2 capture solutions.| Feature | CNM-based | Commercial MOFs |
|---|---|---|
| Primary mechanism | Physisorption (physical) | Physisorption and chemisorption |
| CO2 capacity | Moderate (3 to 8 mmol g−1) | High (5 to 9 mmol g−1) |
| Selectivity | Moderate (improved with N-doping) | High (intrinsic to structure) |
| Moisture stability | High (hydrophobic) | Low (hydrophilic/degrades) |
| Heat of adsorption | Low/moderate (20–30 kJ mol−1) | Moderate/high (25–50 kJ mol−1) |
| Regeneration energy | Low (energy efficient) | High (energy intensive) |
| Cost | Low (biomass/waste precursors) | High (metals/ligands) |
9 Challenges and probable solutions in CNM-based CO2 capture technologies
Using CNM-based materials to absorb CO2 is very promising, as they can be tailored to have different levels of porosity, possess a large surface area, are thermally stable, and are relatively inexpensive. However, several important scientific and technical issues still need to be addressed before they can be used on a large scale. The main problems are discussed here, along with possible solutions. CNM-based materials often exhibit limited CO2/N2 selectivity due to their primarily non-polar surfaces and the absence of specialized binding sites, which complicates the efficient separation of CO2 from mixed gas streams, such as flue gases that contain N2 or O2. Their efficacy is further impaired by moisture sensitivity, as water vapor contends with CO2 for active sites and may destroy oxygen-containing functional groups, resulting in a loss of stability and adsorption capacity. Another significant issue is that CO2 does not absorb well at low partial pressures, which is often the case in post-combustion situations. CNMs that mainly depend on physisorption do not interact well with CO2 in these circumstances, which means they do not collect it well enough. Additionally, CNM-based adsorbents modified with amines or heteroatoms often lose their integrity when heated and cooled repeatedly, leading to structural breakdown and a decline in their adsorption capacity over time. The pore structure of carbon materials derived from biomass or waste is also a concern, as they often exhibit uneven distributions across micro-, meso-, and macropores, which renders their adsorption performance unreliable. Additionally, the high expense of synthesis and functionalization, particularly in techniques such as amine grafting or KOH activation, makes it more challenging to generate revenue and creates hazardous waste, posing a significant safety and environmental issue. Finally, scaling up and integrating into the industry are still significant challenges. Even though lab tests have shown encouraging results, it is challenging to scale up these materials into large-scale systems, as they are not very strong, may easily lose pressure, and are not always compatible with current CCUS technology. For CNM-based CO2 collection systems to function effectively in real-world applications, these problems must be addressed.The suggested portable solution to the above highlighted challenges is described below:
To address the constraints inherent in non-polar surfaces, heteroatom doping approaches will focus on optimizing pyridinic nitrogen functionalities. These functionalities feature a localized lone pair, which facilitates robust Lewis acid-base interactions with CO2, thereby substantially improving selectivity for CO2 over N2, surpassing the capabilities of simple physisorption.271 Moreover, the functionalization of CNMs with amines will be strategically designed to influence the moisture-enhanced chemisorption mechanism. In this mechanism, the presence of water vapor within flue gas alters the reaction stoichiometry from 2:1 (carbamate) to 1:1 (bicarbonate), effectively doubling the theoretical efficiency of the amine.272 To make them less sensitive to moisture, hydrophobic coatings such as fluorinated or alkylsilane layers will be used to keep water out while still allowing CO2 to pass through. Additionally, moisture-resistant features such as sterically hindered amines and ionic-liquid grafted surfaces will be employed to ensure that CO2 affinity remains constant under humid conditions. This chemical modification will be integrated with ultra-micropore engineering, specifically targeting pores smaller than 0.7 nm, to maximize adsorption potential through the overlapping of van der Waals fields. This chemical modification will be integrated with ultra-micropore engineering, specifically targeting pores smaller than 0.7 nm, to maximize adsorption potential through the overlapping of van der Waals fields. Consequently, this approach will ensure a high uptake capacity, even under conditions of low partial pressure. Chemisorption-active sites, including metal–nitrogen coordination centers, will also be added to increase binding energy while yet allowing for reversible desorption for cyclic usage. Using thermally stable linkers, such as pyridine and imidazole, can help address the issue of low regeneration stability. These linkers will hold functional groups in place and stop them from coming loose as the temperature rises over 100 °C. Long-term stability will be achieved through a comprehensive strategy that addresses moisture, oxidation, and contaminants. To prevent capillary condensation and water competition, hydrophobic functionalization will be applied to physisorbents using fluorinated alkylsilanes. For amine-based systems, oxidative degradation will be reduced by shifting from thermal swing to Vacuum Swing Adsorption (VSA) or Electrical Swing Adsorption (ESA). This change will help avoid the high temperatures that can deactivate amines by catalyzing their breakdown.58 Furthermore, the irreversible binding of SOx and NOx will be managed by using sterically hindered amines and tertiary amine functionalities. These materials have lower binding energies for CO2 molecules, allowing for some regeneration and extending the lifespan of the adsorbent in real-world flue gas environments.273
Template-assisted synthesis using silica templates or block copolymers will be used to ensure that the pores are evenly spaced and that the best pore architecture is achieved. To create hierarchical micro- and mesoporous networks, we will utilize controlled activation methods, such as steam or CO2 activation, combined with programmable heating. Using sustainable precursors, such as biochar, lignocellulosic biomass, or agricultural byproducts, can lower the cost and environmental impact of synthesis. Green activation technologies, such as H3PO4 treatment, CO2 activation, or one-pot activation–doping processes, will facilitate processing and reduce chemical waste. For use in factories, CNM-based adsorbents will be formed into strong structural shapes, such as pelletization or structured packings, including those utilizing 3D-printed supports, which are necessary. However, these methods often block the pores, thereby reducing the available surface area. As a means of process intensification, CNMs' adsorbents might be more efficiently utilized within rotating packed beds, where centrifugal forces alleviate pressure-drop constraints and substantially improve mass transfer compared to traditional static beds. This will allow the use of CNM-based CO2 capture technologies on a larger scale. Table 12 briefly summarizes the key challenges in commercial-scale CO2 capture and conversion, their primary causes, and potential solutions, providing critical insights into technological, material, and process bottlenecks to support scalable and economically viable industrial implementation.
| Key challenge | Primary cause | Possible solutions |
|---|---|---|
| Low CO2/N2 selectivity | Weak physisorption, non-polar surfaces | N-doping, amine functionalization |
| Sensitivity to humidity | Hydrophilic surface groups | Hydrophobic pore design, stable functionalities |
| Low CO2 uptake at low pressure | Lack of ultra-micropores, weak adsorption forces | Ultra-micropore tuning, chemical adsorption sites |
| Poor regeneration stability | Functional group degradation | Robust chemistry, low-energy regeneration methods |
| Inconsistent pore structure | Variability in synthesis | Template methods, advanced activation control |
| High synthesis/functionalization cost | Expensive precursors or chemicals | Biomass sources, green chemistry approaches |
| Scale-up and integration difficulties | Lack of pilot studies or structured forms | Engineering design, modular reactors, performance validation |
10. Future directions: from nanoscale design to industrial deployment
Despite remarkable advances in the nanoscale design of CNMs, their large-scale deployment in CCUS remains constrained by challenges related to process integration, manufacturability, and real-world operating conditions. Future research must therefore evolve beyond material-centric performance metrics and adopt a systems-oriented, application-driven paradigm. A central direction is the development of multifunctional and integrated materials that combine capture, conversion, and transport within a single platform. Rather than treating adsorption and utilization as isolated steps, next-generation CNMs should enable seamless process coupling to minimize energy penalties and improve overall process efficiency. Equally critical is addressing the form factor challenge. Translating nanoscale advantages into industrially viable architectures will require advances in shaping, compaction, and structuring of CNMs into mechanically robust, structured adsorbents that ensure low pressure drop, fast mass transfer, and compatibility with cyclic operation.Future CCUS strategies will also focus on hybrid process integration, in which CNMs operate within membranes, solvents, or other capture technologies as high-performance polishing or intensification units, enabling stringent purity targets while reducing the burden on primary separation stages. To ensure practical relevance, material design must increasingly account for realistic gas environments, including moisture and trace contaminants. Improving chemical and structural resilience under these conditions through targeted surface engineering will be crucial for sustaining long-term performance and stability. Given the large design space of CNMs, machine learning and AI-guided frameworks are expected to identify optimal process–structure relationships tailored to specific operating conditions, thereby reducing reliance on empirical trial-and-error methods. Finally, the transition from laboratory success to industrial credibility will depend on rigorous techno-economic and life-cycle assessments. Future studies should benchmark CNMs against incumbent materials using standardized metrics, such as energy penalty, capture cost, and environmental footprint, to ensure that performance gains translate into tangible system-level benefits. Hence, the future of CNM-based CCUS lies in shifting the guiding question from maximum adsorption capacity to maximum system efficiency under industrially relevant conditions. The convergence of multifunctional material design, scalable architectures, hybrid process integration, data-driven optimization, and transparent economic validation will ultimately determine the viability of CNMs in next-generation CCUS technologies.
11. Conclusion
In conclusion, this review presents a comprehensive and critical examination of the importance of CNM-based adsorbents as scalable and cost-effective solutions for mitigating rising CO2 emissions. Substantial advancements in CNMs have been achieved over the past few decades, underscoring sustained efforts to enhance capture efficiency with long-term sustainability while reducing environmental and energy penalties. The effective CO2 mitigation relies not only on maximizing the specific surface area, but also on the precise engineering of pore topology and surface energetics. Low-dimensional allotropes, including 0D and 1D, exhibit distinctive electronic properties and transport pathways. However, 3D hierarchical carbon structures stand out as the most promising materials for industrial use. These frameworks strike an ideal balance between capacity and kinetics by integrating ultra-micropores (<0.7 nm) for high-density CO2 molecular storage, along with mesoporous channels that reduce diffusion resistance during rapid pressure changes. Moreover, the various strategies for heteroatom functionalization, including nitrogen doping and amine grafting, carefully adjust adsorption enthalpies to make capture reversible and achieve long-term cycling stability. From a physics perspective, the core scientific insight derived from this review is that the good performance adsorbent based on CNMs must operate within this thermodynamic window defined by the isosteric heat of adsorption, QST, which must be tuned to a value of 35–50 kJ mol−1. In this window, CNMs adsorb strongly enough to capture CO2 from dilute flue gas streams (chemisorption-like affinity) yet weak enough to allow for complete desorption below 100 °C (physisorption-like regenerability). The amine functionalization yields the highest capacities through carbamate formation, resulting in nitrogen-doped carbons that offer superior long-term stability and moisture tolerance, thereby mitigating the degradation issues often observed in amine-grafted composites. However, optimisation of heteroatom doping is necessary to maintain the ultramicroporous structure. To effectively translate laboratory-scale advances into industrially viable retrofitting solutions, future adsorbent materials must meet stringent performance criteria under realistic post-combustion conditions, including a CO2 uptake of ≥3 mmol g−1 at low partial pressure (0.15 bar), a CO2/N2 selectivity exceeding 20 to ensure high-purity separation from flue gas, and a regeneration energy penalty of ≤50 kJ mol−1, thereby maintaining clear economic and energetic advantages over conventional aqueous amine-based capture systems.To summarize, this review provides valuable insights for advancing CO2 capture strategies using CNMs as state-of-the-art materials, offering excellent metrics for effective and large-scale deployment in mitigating greenhouse gas emissions.
Author contributions
JS: conceptualization, acquisition of data, investigation, analysis and interpretation, writing – original draft, writing – review and editing, visualization, validation, software, resources; AG: writing – original draft, writing – review and editing, validation; ZZ and SKT: writing – review & editing, validation; ASD: writing – review and editing, proof-reading; all authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Abbreviations
| CCUS | Carbon capture, utilization, and storage |
| CO2 | Carbon dioxide |
| CNMs | Carbon nanomaterials |
| CNTs | Carbon nanotubes |
| IPCC | Intergovernmental Panel on Climate Change |
| IEA | International Energy Agency |
| EOR | Enhanced oil recovery |
| DAC | Direct air capture |
| MOFs | Metal–organic frameworks |
| GO | Graphene oxide |
| rGO | Reduced graphene oxide |
| SWCNTs | Single-walled carbon nanotubes |
| MWCNTs | Multi-walled carbon nanotubes |
| Q st | Heat of adsorption (isosteric heat) |
| ACs | Activated carbons |
| TEPA | Tetraethylenepentamine |
| DETA | Diethylenetriamine |
| TETA | Triethylenetetramine |
| DFT | Density functional theory |
| BET | Brunauer–Emmett–Teller (surface area analysis) |
| NLDFT | Non-linear density functional theory |
| SEM | Scanning electron microscopy |
| FTIR | Fourier transform infrared spectroscopy |
| IAST | Ideal adsorbed solution theory |
| MESP | Molecular electrostatic potential |
| PEI | Polyethyleneimine |
| APTES | 3-Aminopropyltrimethoxysilane |
| AHNC | Activated halloysite nanocarbon |
Data availability
No primary research results, software, or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
J. Singh gratefully acknowledges the Department of Physics and Materials Science and the authorities of Thapar Institute of Engineering and Technology (TIET), Patiala, for providing state-of-the-art research infrastructure, advanced technical support, and a highly conducive academic environment that collectively enabled the successful execution and completion of this work. SKT also thanks the SPARC project (P3808) under the Indo-UK scheme for its help. Additionally, the authors express gratitude to all the researchers in this field who are mentioned in this paper.References
-
D. J. Soeder, Fracking and the environment: A scientific assessment of the environmental risks from hydraulic fracturing and fossil fuels, 2020 Search PubMed
.
- R. Heede and N. Oreskes, Glob. Environ. Chang., 2016, 36, 12–20 Search PubMed
.
- S. Paraschiv and L. S. Paraschiv, Energy Reports, 2020, 6, 237–242 CrossRef
.
-
J. Singh, A. S. Dhaliwal, K. Sharma, R. Sehgal and V. Kumar, Conjugated Polymers for Next-Generation Applications, Synthesis, Properties and Optoelectrochemical Devices, Vol. 1, 2022, pp. 505–538 Search PubMed
.
- Y. Liu, J. Liu, M. Chang and C. Zheng, Fuel, 2012, 95, 521–527 Search PubMed
.
- Y. Liu, J. Liu, Y. S. Lin and M. Chang, J. Phys. Chem. C, 2014, 118, 6744–6751 Search PubMed
.
- A. Majumdar and J. Deutch, Joule, 2018, 2, 805–809 CrossRef
.
- M. R. Raupach, G. Marland, P. Ciais, C. Le Quéré, J. G. Canadell, G. Klepper and C. B. Field, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 10288–10293 CrossRef CAS PubMed
.
- M. Wang, J. Liu, J. Hu and F. Liu, Ind. Eng. Chem. Res., 2015, 54, 9805–9812 Search PubMed
.
-
A. Kumar, S. Ogita and Y. Y. Yau, Biofuels: Greenhouse gas mitigation and global warming: Next generation biofuels and role of biotechnology, 2018 Search PubMed
.
- Intergovernmental Panel on Climate Change (IPCC), Climate Change 2021 – The Physical Science Basis, 2023.
- T. R. Anderson, E. Hawkins and P. D. Jones, Endeavour, 2016, 40, 178–187 Search PubMed
.
- V. Ramanathan and Y. Feng, Atmos. Environ., 2009, 43, 37–50 CrossRef CAS
.
-
M. Jarraud and A. Steiner, Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation, Special Report of the Intergovernmental Panel on Climate Change, 2012, vol. 9781107025, pp. 3–22 Search PubMed
.
- International Energy Agency, Glob. Energy Rev. 2021, 2021, 1–32.
- M. Yuan, G. Gao, X. Hu, X. Luo, Y. Huang, B. Jin and Z. Liang, Ind. Eng. Chem. Res., 2018, 57, 6189–6200 CrossRef CAS
.
- P. Wienchol, A. Szlęk and M. Ditaranto, Energy, 2020, 198, 117352 Search PubMed
.
- N. Mac Dowell, P. S. Fennell, N. Shah and G. C. Maitland, Nat. Clim. Chang., 2017, 7, 243–249 Search PubMed
.
- P. D. Sutrisna, S. Kawi, K. Khoiruddin, P. C. W. B. Mustika, N. Prasetya and I. G. Wenten, Carbon Capture Sci. Technol., 2025, 14, 100366 CAS
.
- Y. Song, Z. Hu, X. Yang, Y. An and Y. Lu, Environ. Res., 2025, 274, 121271 CrossRef CAS PubMed
.
- M. Bui, C. S. Adjiman, A. Bardow, E. J. Anthony, A. Boston, S. Brown, P. S. Fennell, S. Fuss, A. Galindo, L. A. Hackett, J. P. Hallett, H. J. Herzog, G. Jackson, J. Kemper, S. Krevor, G. C. Maitland, M. Matuszewski, I. S. Metcalfe, C. Petit, G. Puxty, J. Reimer, D. M. Reiner, E. S. Rubin, S. A. Scott, N. Shah, B. Smit, J. P. M. Trusler, P. Webley, J. Wilcox and N. Mac Dowell, Energy Environ. Sci., 2018, 11, 1062–1176 RSC
.
- R. Stuart Haszeldine, Science, 2009, 325, 1647–1652 CrossRef PubMed
.
- A. Thorbjörnsson, H. Wachtmeister, J. Wang and M. Höök, Energy Strateg. Rev., 2015, 7, 18–28 Search PubMed
.
- I. Global CCS, GLOBAL STATUS OF CCS 2025, 2025.
- M. Atif, H. Z. Haider, R. Bongiovanni, M. Fayyaz, T. Razzaq and S. Gul, Surf. Interfaces, 2022, 31, 102080 Search PubMed
.
- K. Ramadass, C. I. Sathish, S. Mariaruban, G. Kothandam, S. Joseph, G. Singh, S. Kim, W. Cha, A. Karakoti, T. Belperio, J. B. Yi and A. Vinu, ACS Appl. Mater. Interfaces, 2020, 12, 11922–11933 CrossRef CAS PubMed
.
- J. Singh and A. S. Dhaliwal, J. Phys. Chem. Solids, 2022, 160, 110358 CrossRef CAS
.
- H. Sharma and A. Dhir, Environ. Chem. Lett., 2021, 19, 851–873 Search PubMed
.
-
Materials for Carbon Capture, ed. D. E. Jiang, S. M. Mahurin, S. Dai, 2019, p. 376 Search PubMed
.
- J. Wang, L. Huang, R. Yang, Z. Zhang, J. Wu, Y. Gao, Q. Wang, D. O’Hare and Z. Zhong, Energy Environ. Sci., 2014, 7, 3478–3518 RSC
.
- K. Tian, Z. Wu, F. Xie, W. Hu and L. Li, Energy Fuels, 2017, 31, 12477–12486 CrossRef CAS
.
- P. Mehra and A. Paul, ACS Omega, 2022, 7, 34538–34546 CrossRef CAS PubMed
.
- R. Shi, B. Liu, Y. Jiang, X. Xu, H. Wang, Z. Zeng and L. Li, ACS Omega, 2021, 6, 30716–30725 CrossRef CAS PubMed
.
- A. Das, S. D. Peu, M. S. Hossain, M. M. A. Nahid, F. R. Bin Karim, H. Chowdhury, M. H. Porag, D. B. P. Argha, S. Saha, A. R. M. T. Islam, M. M. Salah and A. Shaker, Heliyon, 2023, 9, e22341 CrossRef CAS PubMed
.
- D. Bonenfant, M. Kharoune, P. Niquette, M. Mimeault and R. Hausler, Sci. Technol. Adv. Mater., 2008, 9, 013007 Search PubMed
.
- J. Singh, Z. Zhu, S. Han, N. Wang and S. K. Tiwari, J. Alloys Compd., 2025, 1042, 183755 Search PubMed
.
- Y. J. Park, H. Lee, H. L. Choi, M. C. Tapia, C. Y. Chuah and T. H. Bae, npj 2D Mater. Appl., 2023, 7, 61 CrossRef CAS
.
- B. Dziejarski, J. Serafin, K. Andersson and R. Krzyżyńska, Mater. Today Sustainability., 2023, 24, 100483 CrossRef
.
- J. Singh, S. K. Tiwari, S. Bhowmik, K. S. Hada and A. S. Dhaliwal, Curr. Appl. Phys., 2025, 75, 50–62 CrossRef
.
- G. Singh, J. Lee, A. Karakoti, R. Bahadur, J. Yi, D. Zhao, K. Albahily and A. Vinu, Chem. Soc. Rev., 2020, 49, 4360–4404 RSC
.
- M. S. Khosrowshahi, M. A. Abdol, H. Mashhadimoslem, E. Khakpour, H. B. M. Emrooz, S. Sadeghzadeh and A. Ghaemi, Sci. Rep., 2022, 12, 8917 Search PubMed
.
- A. H. Farmahini, S. Krishnamurthy, D. Friedrich, S. Brandani and L. Sarkisov, Chem. Rev., 2021, 121, 10666–10741 CrossRef CAS PubMed
.
- V. Georgakilas, J. A. Perman, J. Tucek and R. Zboril, Chem. Rev., 2015, 115, 4744–4822 CrossRef CAS PubMed
.
- W. Al-Hajri, Y. De Luna and N. Bensalah, Energy Technol., 2022, 10, 2200498 Search PubMed
.
- U. H. Bhatti, M. S. Alivand, F. Barzagli, M. G. Sanku, J. Gascon and K. A. Mumford, ACS Sustainable Chem. Eng., 2023, 11, 11955–11964 Search PubMed
.
- K. Gong, F. Du, Z. Xia, M. Durstock and L. Dai, Science, 2009, 323, 760–764 Search PubMed
.
- L. Spessato, V. A. Duarte, J. M. Fonseca, P. A. Arroyo and V. C. Almeida, J. CO2 Util., 2022, 61, 102013 CrossRef CAS
.
- N. Rao, M. Wang, Z. Shang, Y. Hou, G. Fan and J. Li, Energy Fuels, 2018, 32, 670–677 CrossRef CAS
.
- L. Wang, S. Fan, X. Li, M. O. Tadé and S. Liu, ACS Omega, 2022, 7, 40184–40194 Search PubMed
.
- L. B. Hamdy, C. Goel, J. A. Rudd, A. R. Barron and E. Andreoli, Mater. Adv., 2021, 2, 5843–5880 Search PubMed
.
- M. Crippa, D. Guizzardi, M. Banja, E. Solazzo, E. Schaaf, F. Pagani, F. Monforti-Ferrario, J. J. G. Olivier, R. Quadrelli, M. Risquez Martin, P. Moharamli, Taghavi, G. Grassi, S. Rossi, D. Oom, A. Branco, J. San Miguel, E. Vignati, M. Muntean, E. Schaaf, E. Solazzo, F. Monforti-Ferrario, J. J. G. Olivier and E. Vignati, Fossil CO2 emissions of all world countries - 2020 Report, 2022 Search PubMed.
- J. Chen, Y. Xu, P. Liao, H. Wang and H. Zhou, Carbon Capture Sci. Technol., 2022, 4, 100052 CAS
.
- J. Artz, T. E. Müller, K. Thenert, J. Kleinekorte, R. Meys, A. Sternberg, A. Bardow and W. Leitner, Chem. Rev., 2018, 118, 434–504 CrossRef CAS PubMed
.
- F. Almomani, A. Abdelbar and S. Ghanimeh, Sustainability, 2023, 15, 10438 Search PubMed
.
- T. M. McDonald, W. R. Lee, J. A. Mason, B. M. Wiers, C. S. Hong and J. R. Long, J. Am. Chem. Soc., 2012, 134, 7056–7065 CrossRef CAS PubMed
.
- B. Olfe-Kräutlein, T. Strunge and A. Chanin, Front. Energy Res., 2021, 9 DOI:10.3389/fenrg.2021.742709
.
- F. Raganati and P. Ammendola, Energy Fuels, 2024, 38, 13858–13905 CrossRef CAS
.
- F. Raganati, F. Miccio and P. Ammendola, Energy Fuels, 2021, 35, 12845–12868 CrossRef CAS
.
- J. Hack, N. Maeda and D. M. Meier, ACS Omega, 2022, 7, 39520–39530 CrossRef CAS PubMed
.
- S. Vaz, A. P. Rodrigues de Souza and B. E. Lobo Baeta, Clean. Eng. Technol., 2022, 8, 100456 CrossRef
.
- S. Kammerer, I. Borho, J. Jung and M. S. Schmidt, Int. J. Environ. Sci. Technol., 2023, 20, 8087–8104 CrossRef
.
- A. S. Joel and Y. M. Isa, J. Chem. Technol. Biotechnol., 2023, 98, 838–855 CrossRef CAS
.
- D. Alalaiwat and E. Khan, Rev. Environ. Sci. Biotechnol., 2023, 22, 799–822 CrossRef CAS
.
- H. Liu, H. Lu and H. Hu, Renew. Sustain. Energy Rev., 2024, 189, 113908 Search PubMed
.
- D. Obi, S. Onyekuru and A. Orga, Int. J. Sustain. Energy, 2024, 43, 2317137 Search PubMed
.
- A. Yagmur Goren, D. Erdemir and I. Dincer, Environ. Res., 2024, 240, 117503 CrossRef CAS PubMed
.
- S. Arrhenius and Futur Nat, Doc. Glob. Chang., 2013, 9, 303–312 Search PubMed
.
- J. D. Figueroa, T. Fout, S. Plasynski, H. McIlvried and R. D. Srivastava, Int. J. Greenh. Gas Control, 2008, 2, 9–20 CrossRef CAS
.
- M. Z. Jacobson, Energy Environ. Sci., 2009, 2, 148–173 Search PubMed
.
- S. Iijima, Nature, 1991, 354, 56–58 CrossRef CAS
.
- M. Isah, R. Lawal and S. A. Onaizi, Green Chem. Eng., 2025, 6, 305–334 CrossRef CAS
.
- F. Xu, Y. Yu, J. Yan, Q. Xia, H. Wang, J. Li and Z. Li, Chem. Eng. J., 2016, 303, 231–237 Search PubMed
.
- D. G. Boer, J. Langerak and P. P. Pescarmona, ACS Appl. Energy Mater., 2023, 6, 2634–2656 Search PubMed
.
- J. S. Zou, Z. P. Wang, Y. H. Andaloussi, J. Xue, W. Zhang, B. E. G. Lucier, Z. Zhang, Y. Jia, X. C. Wu, J. Li, Y. Huang, M. J. Zaworotko, G. Chen, S. Chen and Y. L. Peng, Nat. Commun., 2025, 16, 2598 CrossRef CAS PubMed
.
- M. S. Lee, M. Park, H. Y. Kim and S. J. Park, Sci. Rep., 2016, 6, 23224 Search PubMed
.
- B. Yuan, G. Zhan, Z. Chen, Y. Li, L. Wang, C. You and J. Li, Int. J. Greenh. Gas Control, 2022, 118, 103673 CrossRef CAS
.
- C. Chen, M. Kosari, M. Jing and C. He, Environ. Funct. Mater., 2022, 1, 253–266 Search PubMed
.
- S. Gaikwad, Y. Kim, R. Gaikwad and S. Han, J. Environ. Chem. Eng., 2021, 9, 105523 Search PubMed
.
- R. V. Siriwardane, M. S. Shen, E. P. Fisher and J. Losch, Energy Fuels, 2005, 19, 1153–1159 CrossRef CAS
.
- R. L. Siegelman, E. J. Kim and J. R. Long, Nat. Mater., 2021, 20, 1060–1072 Search PubMed
.
- Y. M. Abdellatif, R. Surkatti, R. Muhammad, A. Sodiq, N. Nassar, T. Al-Ansari and A. I. Amhamed, Energy Convers. Manag., 2026, 347, 120544 CrossRef CAS
.
- M. M. Devadiga, S. Sarangi, A. S. Bhat, M. Osial, K. Joseph, S. Olusegun, J. Singh, N. Wang, L. A. Ribeiro, Jr. and S. K. Tiwari, Energy Environ. Sci., 2026 10.1039/d6ee00134c
.
- L. Espinal, D. L. Poster, W. Wong-Ng, A. J. Allen and M. L. Green, Environ. Sci. Technol., 2013, 47, 11960–11975 Search PubMed
.
- J. W. Park, S. Heo, J. G. Yeo, S. Lee, J. K. Kim and J. H. Lee, Membranes, 2025, 15(7), 200 CrossRef CAS PubMed
.
- M. H. Jenab, M. Vahidi and M. Mehrabi, J. Chinese Chem. Soc., 2006, 53, 283–286 CrossRef CAS
.
- R. Idem, M. Wilson, P. Tontiwachwuthikul, A. Chakma, A. Veawab, A. Aroonwilas and D. Gelowitz, Ind. Eng. Chem. Res., 2006, 45, 2414–2420 CrossRef CAS
.
- G. T. Rochelle, Science, 2009, 325, 1652–1654 Search PubMed
.
- B. Aghel, S. Janati, S. Wongwises and M. S. Shadloo, Int. J. Greenh. Gas Control, 2022, 119, 103715 Search PubMed
.
- W. Emori, I. I. Udoh, O. O. Ekerenam, A. I. Ikeuba, I. I. N. Etim, C. N. Njoku, E. F. Daniel, D. I. Njoku, P. C. Uzoma, S. K. Kolawole and O. S. Olanrele, Greenh. Gases Sci. Technol., 2023, 13, 876–904 Search PubMed
.
- A. K. Sleiti, W. A. Al-Ammari, L. Vesely and J. S. Kapat, J. Energy Resour. Technol. Trans. ASME, 2022, 144, 092106 CrossRef CAS
.
- F. M. Orr, Energy Environ. Sci., 2009, 2, 449–458 Search PubMed
.
- M. Cao, L. Zhao, D. Xu, R. Ciora, P. K. T. Liu, V. I. Manousiouthakis and T. T. Tsotsis, J. Memb. Sci., 2020, 605, 118028 Search PubMed
.
- D. Jansen, M. Gazzani, G. Manzolini, E. Van Dijk and M. Carbo, Int. J. Greenh. Gas Control, 2015, 40, 167–187 Search PubMed
.
- A. Skorek-Osikowska, K. Janusz-Szymańska and J. Kotowicz, Energy, 2012, 45, 92–100 CrossRef CAS
.
- F. Dai, S. Zhang, Y. Luo, K. Wang, Y. Liu and X. Ji, Processes, 2023, 11, 1765 Search PubMed
.
- A. A. Olajire, Energy, 2010, 35, 2610–2628 CrossRef CAS
.
- A. Kather and G. Scheffknecht, Naturwissenschaften, 2009, 96, 993–1010 Search PubMed
.
- D. Y. C. Leung, G. Caramanna and M. M. Maroto-Valer, Renew. Sustain. Energy Rev., 2014, 39, 426–443 Search PubMed
.
- B. J. P. Buhre, L. K. Elliott, C. D. Sheng, R. P. Gupta and T. F. Wall, Prog. Energy Combust. Sci., 2005, 31, 283–307 Search PubMed
.
- S. Yadav and S. S. Mondal, Fuel, 2022, 308, 122057 CrossRef CAS
.
- I. Pfaff and A. Kather, Energy Procedia, 2009, 1, 495–502 Search PubMed
.
- S. M. Carpenter and H. A. Long, Integr. Gasif. Comb. Cycle Technol., 2017, 445–463 Search PubMed
.
- T. Burdyny and H. Struchtrup, Energy, 2010, 35, 1884–1897 Search PubMed
.
- A. Mukherjee, J. A. Okolie, A. Abdelrasoul, C. Niu and A. K. Dalai, J. Environ. Sci., 2019, 83, 46–63 CrossRef CAS PubMed
.
- M. Kanniche, R. Gros-Bonnivard, P. Jaud, J. Valle-Marcos, J. M. Amann and C. Bouallou, Appl. Therm. Eng., 2010, 30, 53–62 CrossRef CAS
.
- D. Loachamin, J. Casierra, V. Calva, A. Palma-Cando, E. E. Ávila and M. Ricaurte, ChemEngineering, 2024, 8, 129 CrossRef CAS
.
- H. A. Alalwan and A. H. Alminshid, Sci. Total Environ., 2021, 788, 147850 Search PubMed
.
- D. Obi, S. Onyekuru and A. Orga, Energy Sci. Eng., 2025, 13, 980–994 CrossRef CAS
.
- J. Singh and A. S. Dhaliwal, Int. J. Environ. Sci. Technol., 2023, 20, 11483–11500 CrossRef CAS
.
- K. J. Hughes, K. A. Iyer, R. E. Bird, J. Ivanov, S. Banerjee, G. Georges and Q. A. Zhou, ACS Appl. Nano Mater., 2024, 7, 18695–18713 CrossRef CAS
.
- M. Pardakhti, T. Jafari, Z. Tobin, B. Dutta, E. Moharreri, N. S. Shemshaki, S. Suib and R. Srivastava, ACS Appl. Mater. Interfaces, 2019, 11, 34533–34559 CrossRef CAS PubMed
.
- R. Morales-Ospino, L. Reinert, M. T. Izquierdo, A. Celzard, L. Duclaux and V. Fierro, Environ. Res., 2025, 273, 121211 CrossRef CAS PubMed
.
- A. N. Shafawi, A. R. Mohamed, P. Lahijani and M. Mohammadi, J. Environ. Chem. Eng., 2021, 9, 106869 Search PubMed
.
- H. E. Ashkanani, R. Wang, W. Shi, N. S. Siefert, R. L. Thompson, K. Smith, J. A. Steckel, I. K. Gamwo, D. Hopkinson, K. Resnik and B. I. Morsi, Int. J. Greenh. Gas Control, 2020, 101, 103135 CrossRef CAS
.
- P. Pourhakkak, A. Taghizadeh, M. Taghizadeh, M. Ghaedi and S. Haghdoust, Interface Sci. Technol., 2021, 33, 1–70 Search PubMed
.
- M. E. Majchrowicz, D. W. F. Wim Brilman and M. J. Groeneveld, Energy Proc., 2009, 1, 979–984 CrossRef CAS
.
- Y. Liu and J. Wilcox, Environ. Sci. Technol., 2012, 46, 1940–1947 Search PubMed
.
- M. Sai Bhargava Reddy, D. Ponnamma, K. K. Sadasivuni, B. Kumar and A. M. Abdullah, RSC Adv., 2021, 11, 12658–12681 RSC
.
- S. K. Tiwari, M. Bystrzejewski, A. De Adhikari, A. Huczko and N. Wang, Prog. Energy Combust. Sci., 2022, 92, 101023 Search PubMed
.
- E. Fleming, I. Kholmanov and L. Shi, Carbon, 2018, 136, 380–386 CrossRef CAS
.
- L. Zou, Y. Sun, S. Che, X. Yang, X. Wang, M. Bosch, Q. Wang, H. Li, M. Smith, S. Yuan, Z. Perry and H. C. Zhou, Adv. Mater., 2017, 29, 1700229 CrossRef PubMed
.
- Y. Hou, Y. Chen, X. He, F. Wang, Q. Cai and B. Shen, Chem. Eng. J., 2024, 490, 151424 CrossRef CAS
.
- M. Gorbounov, P. Halloran and S. Masoudi Soltani, J. CO2 Util., 2024, 86, 102908 Search PubMed
.
- A. P. Khedulkar, R. G. Bobade, R. An Doong, B. Pandit, N. M. Ky, R. C. Ambare, T. D. Hoang and K. J. Kumar, Mater. Today Chem., 2025, 46, 102688 CrossRef
.
- V. Ramar and A. Balraj, Energy Fuels, 2022, 36, 13479–13505 CrossRef CAS
.
- C. H. Yu, C. H. Huang and C. S. Tan, Aerosol Air Qual. Res., 2012, 12, 745–769 Search PubMed
.
- R. Singh, L. Wang, K. Ostrikov and J. Huang, Adv. Mater.
Interfaces, 2024, 11, 2202290 Search PubMed
.
- I. Khan, A. Altaf, S. Sadiq, S. Khan, A. Khan, S. Khan, M. Humayun, A. Khan, R. A. Abumousa and M. Bououdina, Chem. Eng. J. Adv., 2025, 21, 100691 Search PubMed
.
- Z. Zhu and B. Xu, Separations, 2022, 9, 307 Search PubMed
.
- H. Ma, H. Fu, Y. Tong, A. Umar, Y. M. Hung and X. Wang, Carbon Capture Sci. Technol., 2025, 15, 100441 CAS
.
- S. Kundu, T. Khandaker, M. A. A. M. Anik, M. K. Hasan, P. K. Dhar, S. K. Dutta, M. A. Latif and M. S. Hossain, RSC Adv., 2024, 14, 29693–29736 Search PubMed
.
- D. A. Kang, C. Murphy and H. K. Jeong, Microporous Mesoporous Mater., 2024, 369, 113038 Search PubMed
.
- L. Yang, W. Rui, Z. Qingmin, Z. Yuanlong, F. Xin and X. Zhaojie, Unconv. Resour., 2024, 4, 100096 Search PubMed
.
- O. Awogbemi and D. A. Desai, Discov. Nano, 2025, 20, 29 Search PubMed
.
- D. Gendron and M. Zakharova, Appl. Chem., 2024, 4, 236–269 Search PubMed
.
- Y. K. Leong, Y. J. Huang and J. S. Chang, Int. J. Hydrogen Energy, 2024, 998–1007 Search PubMed
.
- T. Iglina, P. Iglin and D. Pashchenko, Sustainability, 2022, 14, 3801 CrossRef CAS
.
- J. Zhang, Z. Xia and L. Dai, Sci. Adv., 2015, 1, 1500564 CrossRef PubMed
.
- J. Wu, S. Ma, J. Sun, J. I. Gold, C. Tiwary, B. Kim, L. Zhu, N. Chopra, I. N. Odeh, R. Vajtai, A. Z. Yu, R. Luo, J. Lou, G. Ding, P. J. A. Kenis and P. M. Ajayan, Nat. Commun., 2016, 7, 13869 CrossRef CAS PubMed
.
- Y. Song, W. Chen, C. Zhao, S. Li, W. Wei and Y. Sun, Angew. Chem., Int. Ed., 2017, 56, 10840–10844 CrossRef CAS PubMed
.
- Y. Liu, Y. Zhang, K. Cheng, X. Quan, X. Fan, Y. Su, S. Chen, H. Zhao, Y. Zhang, H. Yu and M. R. Hoffmann, Angew. Chem., Int. Ed., 2017, 56, 15607–15611 CrossRef CAS PubMed
.
- X. Liu, P. V. Kumar, Q. Chen, L. Zhao, F. Ye, X. Ma, D. Liu, X. Chen, L. Dai and C. Hu, Appl. Catal., B, 2022, 316, 121618 CrossRef CAS
.
- Y. Li, S. Zheng, H. Liu, Q. Xiong, H. Yi, H. Yang, Z. Mei, Q. Zhao, Z. W. Yin, M. Huang, Y. Lin, W. Lai, S. X. Dou, F. Pan and S. Li, Nat. Commun., 2024, 15, 176 CrossRef CAS PubMed
.
- Y. Zhu, L. Chen, J. Pan, S. Jiang, J. Wang, G. Zhang and K. Zhang, Prog. Mater. Sci., 2025, 148, 101373 CrossRef CAS
.
- M. S. Mohtasim and B. K. Das, Renew. Sustain. Energy Rev., 2025, 218, 115779 Search PubMed
.
- A. M. Afridi, M. Aktary, S. Shaheen Shah, S. I. Mitu Sheikh, G. Jahirul Islam, M. Nasiruzzaman Shaikh and M. Abdul Aziz, Chem. Rec., 2024, 24, e202400144 Search PubMed
.
- T. Qiao, H. Pan, Z. Han, Z. Zhang, S. Zhou, X. Lu and Y. Li, Fuel, 2025, 394, 135129 Search PubMed
.
- S. Assyl, S. Botakoz and Z. Saule, Discov. Nano, 2025, 20, 115 Search PubMed
.
- T. Zhao, Q. Wang, Y. Kawazoe and P. Jena, Carbon, 2018, 132, 249–256 Search PubMed
.
- A. J. Clancy, M. K. Bayazit, S. A. Hodge, N. T. Skipper, C. A. Howard and M. S. P. Shaffer, Chem. Rev., 2018, 118, 7363–7408 Search PubMed
.
- S. K. Gebremariam, A. M. Varghese, S. Ehrling, Y. Al Wahedi, A. AlHajaj, L. F. Dumée and G. N. Karanikolos, ACS Appl. Mater. Interfaces, 2024, 16, 50785–50799 Search PubMed
.
- J. Serafin and B. Dziejarski, Environ. Sci. Pollut. Res., 2024, 31, 40008–40062 Search PubMed
.
- S. K. Verma, P. Tripathi and A. Bhatnagar, Nanomater. Carbon Dioxide Capture Convers. Technol., 2022, 245–260 Search PubMed
.
- R. Balasubramanian and S. Chowdhury, J. Mater. Chem. A, 2015, 3, 21968–21989 Search PubMed
.
- B. Cui, X. Ju, H. Ma, S. Meng, Y. Liu, J. Wang, D. Wang and Z. Yang, Sep. Purif. Technol., 2025, 354, 128794 CrossRef CAS
.
- S. Wang, Y. Xu, M. Yan, L. Zhang and Z. Liu, J. Non. Cryst. Solids, 2018, 499, 101–106 CrossRef CAS
.
- D. Gang, Z. Uddin Ahmad, Q. Lian, L. Yao and M. E. Zappi, Chem. Eng. J., 2021, 403, 126286 CrossRef CAS
.
- S. Jung, Y. K. Park and E. E. Kwon, J. CO2 Util., 2019, 32, 128–139 CrossRef CAS
.
- H. Ma, X. Ju, B. Cui, S. Meng, Y. Liu, J. Li, D. Wang and Z. Yang, ACS Appl. Mater. Interfaces, 2025, 17, 13836–13850 CrossRef CAS PubMed
.
- M. T. Dunstan, F. Donat, A. H. Bork, C. P. Grey and C. R. Müller, Chem. Rev., 2021, 121, 12681–12745 CrossRef CAS PubMed
.
- Y. Shen, Fuel Process. Technol., 2022, 236, 107437 CrossRef CAS
.
- M. Umar, B. O. Yusuf, M. Aliyu, I. Hussain, A. M. Alhassan, M. M. Awad, O. A. Taialla, B. Ali, K. R. Alhooshani and S. A. Ganiyu, Coord. Chem. Rev., 2025, 526, 216380 Search PubMed
.
- Y. Mochizuki, J. Bud, E. Byambajav and N. Tsubouchi, Carbon Resour. Convers., 2024, 100237 Search PubMed
.
- M. S. Khosrowshahi, H. Mashhadimoslem, H. Shayesteh, G. Singh, E. Khakpour, X. Guan, M. Rahimi, F. Maleki, P. Kumar and A. Vinu, Adv. Sci., 2023, 10, 2304289 CrossRef CAS PubMed
.
- P. Devi, A. Gupta and A. Kumar, J. Energy Storage, 2024, 97, 102506 Search PubMed
.
- M. K. Al Mesfer, Environ. Sci. Pollut. Res., 2020, 27, 15020–15028 CrossRef CAS PubMed
.
- M. Aliyu, B. O. Yusuf, A. H. S. Abdullahi, A. I. Bakare, M. Umar, A. S. Hakeem and S. A. Ganiyu, Sep. Purif. Technol., 2024, 340, 126737 CrossRef CAS
.
- O. F. Cruz, I. Campello-Gómez, M. E. Casco, J. Serafin, J. Silvestre-Albero, M. Martínez-Escandell, D. Hotza and C. R. Rambo, Carbon Lett., 2023, 33, 727–735 CrossRef
.
- C. Jin, J. Sun, Y. Chen, Y. Guo, D. Han, R. Wang and C. Zhao, Sep. Purif. Technol., 2021, 276, 119270 Search PubMed
.
- S. Wang, Y. R. Lee, Y. Won, H. Kim, S. E. Jeong, B. Wook Hwang, A. Ra Cho, J. Y. Kim, Y. Cheol Park, H. Nam, D. H. Lee, H. Kim and S. H. Jo, Chem. Eng. J., 2022, 437, 135378 CrossRef CAS
.
- X. L. Zhu, P. Y. Wang, C. Peng, J. Yang and X. Bin Yan, Chin. Chem. Lett., 2014, 25, 929–932 Search PubMed
.
- A. D. Igalavithana, S. W. Choi, J. Shang, A. Hanif, P. D. Dissanayake, D. C. W. Tsang, J. H. Kwon, K. B. Lee and Y. S. Ok, Sci. Total Environ., 2020, 739, 139845 Search PubMed
.
- K. Kumaraguru, P. Saravanan, L. Nagarajan, V. Saravanan, R. Rajeshkannan and M. Rajasimman, Biomass Convers. Biorefinery, 2023, 30447–30466 Search PubMed
.
- K. Li, X. Niu, D. Zhang, H. Guo, X. Zhu, H. Yin, Z. Lin and M. Fu, Environ. Pollut., 2022, 306, 119399 CrossRef CAS PubMed
.
- K. Li, D. Zhang, X. Niu, H. Guo, Y. Yu, Z. Tang, Z. Lin and M. Fu, Sci. Total Environ., 2022, 826, 154133 Search PubMed
.
- H. Patel, H. Weldekidan, A. Mohanty and M. Misra, Carbon Capture Sci. Technol., 2023, 8, 100128 Search PubMed
.
- S. Foorginezhad, M. M. Zerafat, M. Asadnia and G. Rezvannasab, Mater. Chem. Phys., 2024, 317, 129177 Search PubMed
.
- A. Koli, R. Dhabbe, J. Shen, R. K. Motkuri and S. Sabale, Biomass Convers. Biorefinery, 2024, 14, 18789–18801 Search PubMed
.
- S. Deng, B. Hu, T. Chen, B. Wang, J. Huang, Y. Wang and G. Yu, Adsorption, 2015, 21, 125–133 Search PubMed
.
- Y. Xu, Y. Liu, W. Zhan, D. Zhang, Y. Liu, Y. Xu and Z. Wu, Biomass Bioenergy, 2024, 183, 100339 Search PubMed
.
- A. E. Ogungbenro, D. V. Quang, K. A. Al-Ali, L. F. Vega and M. R. M. Abu-Zahra, J. Environ. Chem. Eng., 2018, 6, 4245–4252 Search PubMed
.
- A. Koli, A. K. Battu, R. K. Motkuri and S. Sabale, Biomass Convers. Biorefinery, 2024, 14, 10177–10188 Search PubMed
.
- P. B. Ramos, F. Jerez, M. Erans, A. Mamaní, M. F. Ponce, M. F. Sardella, E. S. Sanz-Pérez, R. Sanz, A. Arencibia and M. A. Bavio, Biomass Bioenergy, 2025, 194, 107669 Search PubMed
.
- P. B. Ramos, A. Mamaní, M. Erans, F. Jerez, M. F. Ponce, M. F. Sardella, A. Arencibia, M. A. Bavio, E. S. Sanz-Pérez and R. Sanz, Energy Fuels, 2024, 38, 6102–6115 Search PubMed
.
- P. B. Ramos, A. Mamani, M. F. Sardella, A. Arencibia, R. Sanz, E. S. Sanz-Pérez, M. A. Bavio and M. Erans, Energy Fuels, 2025, 39, 5442–5452 Search PubMed
.
- A. Okhovat, A. Ahmadpour, F. Ahmadpour and Z. Khaki Yadegar, ISRN Chem. Eng., 2012, 2012, 1–10 Search PubMed
.
- M. Sevilla, N. Alam and R. Mokaya, J. Phys. Chem. C, 2010, 114, 11314–11319 Search PubMed
.
- P. Toh-ae, N. Timasart, D. Tumnantong, T. Bovornratanaraks and S. Poompradub, Sci. Rep., 2024, 14, 17100 Search PubMed
.
- Y. Yang, Y. Liu, S. Liu, Y. Zhao, Q. Zhang, L. Su, Z. Chen and M. Zhao, Chem. Eng. Sci., 2024, 292, 119968 Search PubMed
.
- M. Lotfinezhad, M. Tahmasebpoor and C. Pevida, Environ. Res., 2024, 263, 120017 Search PubMed
.
- R. Morales-Ospino, A. Taurbekov, J. Castro-Gutiérrez, M. T. Izquierdo, M. Atamanov, Z. Mansurov, A. Celzard and V. Fierro, Carbon, 2025, 234, 119978 Search PubMed
.
- M. Gorbounov, L. Hecquet-Perrot, S. Ignatova, P. Hewitson and S. M. Soltani, Next Mater., 2025, 6, 100321 Search PubMed
.
- Z. Ye, C. Han, M. Pan, L. Zhao, N. Liu, A. Nikiforov, K. Leus, N. De Geyter, R. Morent, J. Chen and J. Li, Chem. Eng. J., 2025, 507, 160530 Search PubMed
.
- E. H. Al-Ghurabi, M. M. Boumaza, W. Al-Masry and M. Asif, Sci. Rep., 2025, 15, 17132 Search PubMed
.
- Y. Long, H. Tian, C. H. Lee, H. Li, Z. Zeng, Z. Yang, G. Zhu, X. Chen and L. Liu, Sep. Purif. Technol., 2025, 359, 130476 Search PubMed
.
- J. A. O. Chagas, B. P. Pinto, A. L. de Lima and C. J. A. Mota, Energy Fuels, 2025, 39, 15039–15050 Search PubMed
.
- J. Wang, Y. Yin, X. Liu, Y. Liu, Q. Xiao, L. Zhao, M. Demir, M. Ö. Alaş Çolak, L. Wang and X. Hu, Sep. Purif. Technol., 2025, 376, 134079 Search PubMed
.
- J. Shao, Y. Wang, M. Che, Q. Xiao, M. Demir, M. K. Al Mesfer, L. Wang, X. Hu and Y. Liu, J. Energy Inst., 2025, 123, 102273 Search PubMed
.
- Y. Zhi, J. Shao, J. Wang, X. Liu, Q. Xiao, M. Demir, U. B. Simsek, L. Wang and X. Hu, Molecules, 2025, 30, 3990 CrossRef CAS PubMed
.
- Z. Zhang, Z. P. Cano, D. Luo, H. Dou, A. Yu and Z. Chen, J. Mater. Chem. A, 2019, 7, 20985–21003 Search PubMed
.
- A. Martínez-Alonso, J. M. D. Tascón and E. J. Bottani, J. Phys. Chem. B, 2001, 105, 135–139 Search PubMed
.
- N. Kuganathan, Fuels, 2022, 3, 176–183 Search PubMed
.
- P. Rezaee, S. A. Asl, M. H. Javadi, S. Rezaee, R. Morad, M. Akbari, S. S. Arab and M. Maaza, Sci. Rep., 2024, 14, 12388 Search PubMed
.
- A. A. Khan, R. Ahmad, I. Ahmad and X. Su, J. Mol. Graph. Model., 2021, 103, 107806 Search PubMed
.
- H. Dong, B. Lin, K. Gilmore, T. Hou, S. T. Lee and Y. Li, Curr. Appl. Phys., 2015, 15, 1084–1089 Search PubMed
.
- M. Kabiri and A. Bahrami, Comput. Theor. Chem., 2024, 1233, 114482 Search PubMed
.
- S. Anila and C. H. Suresh, New J. Chem., 2023, 47, 3047–3054 Search PubMed
.
- A. S. Palakkal and R. S. Pillai, Sep. Purif. Technol., 2023, 325, 124650 Search PubMed
.
- J. Singh and A. S. Dhaliwal, Polym. Bull., 2024, 81, 13155–13182 Search PubMed
.
- M. Cinke, J. Li, C. W. Bauschlicher, A. Ricca and M. Meyyappan, Chem. Phys. Lett., 2003, 376, 761–766 Search PubMed
.
- M. A. O. Lourenço, M. Fontana, P. Jagdale, C. F. Pirri and S. Bocchini, Chem. Eng. J., 2021, 414, 128763 Search PubMed
.
- N. Salehi Ardali, S. Riahi, M. Abbasi and N. Mohammadpour, Fuel, 2023, 338, 127213 Search PubMed
.
- Z. Khoshraftar, A. Ghaemi and A. Hemmati, Sci. Rep., 2024, 14, 5130 Search PubMed
.
- R. Xu, S. Kim, H. Ahn, H. S. Kim, M. Kim and Y. T. Kang, Sep. Purif. Technol., 2025, 356, 129922 Search PubMed
.
- Y. Wang, C. Zhang, M. Tang, J. Liu, J. Yuan, Y. Zhao and G. Zhang, J. Environ. Chem. Eng., 2024, 12, 114452 Search PubMed
.
- M. Heidari, S. Borhan Mousavi, F. Rahmani, P. T. Clough and S. Ozmen, Energy Convers. Manag., 2022, 274, 116461 Search PubMed
.
- Y. Zhang, J. Sun, J. Tan, C. H. Ma, S. Luo, W. Li and S. Liu, Fuel, 2021, 305, 121622 Search PubMed
.
- Y. Li, W. Zha, J. Zhang, W. Chen, Q. Yang, S. Zhong and Y. Z. Lei, Fuel, 2025, 382, 133809 Search PubMed
.
- T. S. Mirmiran, S. Riahi, M. Abbasi and M. Mohammadi-Khanaposhtani, J. Environ. Chem. Eng., 2025, 13, 119213 Search PubMed
.
- M. I. Katsnelson, Mater. Today, 2007, 10, 20–27 Search PubMed
.
- K. Bansal, J. Singh and A. S. Dhaliwal, Fullerenes, Nanotubes Carbon Nanostruct., 2024, 32, 651–663 Search PubMed
.
- K. Bansal, J. Singh and A. S. Dhaliwal, Inorg. Nano-Metal Chem., 2024, 54, 1145–1153 Search PubMed
.
- K. Bansal, J. Singh and A. S. Dhaliwal, Fullerenes, Nanotubes Carbon Nanostruct., 2023, 31, 277–287 Search PubMed
.
- A. K. Pathak, M. Borah, A. Gupta, T. Yokozeki and S. R. Dhakate, Compos. Sci. Technol., 2016, 135, 28–38 Search PubMed
.
- C. Kumar, A. Gupta, P. Saharan, M. Singh and S. R. Dhakate, Diam. Relat. Mater., 2023, 140, 110433 Search PubMed
.
- J. Ai, Z. Bacsik, K. Hallstensson, J. Yuan, A. Sugunan and N. Hedin, Chem. Eng. J., 2025, 506, 159963 Search PubMed
.
- B. E. Arango Hoyos, H. F. Osorio, E. K. Valencia Gómez, J. Guerrero Sánchez, A. P. Del Canto Palominos, F. A. Larrain and J. J. Prías Barragán, Sci. Rep., 2023, 13, 14476 Search PubMed
.
- W. Choi, Y. Ho Cho, T. Kim, C. H. Lee, J. Y. Kim, S. H. So, K. Eum, C. H. Lee and D. W. Kim, Sep. Purif. Technol., 2025, 354, 129270 Search PubMed
.
- H. Dong, Y. Wang, W. Ding, Y. Qiu and H. He, Sep. Purif. Technol., 2025, 354, 129158 Search PubMed
.
- Y. Wang, H. Wang, T. C. Zhang, S. Yuan and B. Liang, J. Power Sources, 2020, 472, 228610 Search PubMed
.
- M. He, H. Zhao, J. Jia, W. Zhou, Z. Wang, K. An, Y. Jiao, X. Yang, X. Zhang and T. Fan, Environ. Res., 2025, 267, 120701 Search PubMed
.
- S. Chakraborty, R. Saha and S. Saha, Environ. Sci. Pollut. Res., 2023, 31, 67633–67663 Search PubMed
.
- A. M. Varghese, K. S. K. Reddy, N. Bhoria, S. Singh, J. Pokhrel and G. N. Karanikolos, Chem. Eng. J., 2021, 420, 129677 Search PubMed
.
- Z. Zhou, E. Davoudi and B. Vaferi, J. Environ. Chem. Eng., 2021, 9, 106202 Search PubMed
.
- Y. Hosseini, M. Najafi, S. Khalili, M. Jahanshahi and M. Peyravi, Mater. Chem. Phys., 2021, 270, 124788 Search PubMed
.
- V. V. Chaban and N. A. Andreeva, Phys. Chem. Chem. Phys., 2022, 24, 25801–25815 Search PubMed
.
- Y. Nan, D. Gomez-Maldonado, K. Zhang, H. Du, D. C. Whitehead, M. Li, X. Zhang and M. S. Peresin, Carbohydr. Polym. Technol. Appl., 2024, 8, 100585 Search PubMed
.
- M. P. Jerome, A. Mathai Varghese, S. Kuppireddy, G. N. Karanikolos and N. Alamoodi, Sep. Purif. Technol., 2025, 360, 131089 Search PubMed
.
- R. Navik, E. Wang, X. Ding, H. Yunyi, Y. Liu and J. Li, J. Energy Chem., 2025, 100, 653–664 Search PubMed
.
- A. Sharma, R. Kumar, A. Gupta, P. R. Agrawal, N. Dwivedi, D. P. Mondal, A. K. Srivastava and S. R. Dhakate, Mater. Today Commun., 2022, 30, 103055 Search PubMed
.
- D. F. Maquíñez-Buitrago, J. M. Ramos-Rincón, L. Giraldo and J. C. Moreno-Piraján, Hybrid Adv., 2024, 6, 100219 Search PubMed
.
- B. Zhang, Y. Jiang and R. Balasubramanian, Resour. Conserv. Recycl., 2022, 185, 106453 CrossRef CAS
.
- M. Zhou, Y. Lin, H. Xia, X. Wei, Y. Yao, X. Wang and Z. Wu, Nano-Micro Lett., 2020, 12, 58 Search PubMed
.
- M. Vorokhta, M. I. M. Kusdhany, D. Vöröš, M. Nishihara, K. Sasaki and S. M. Lyth, Chem. Eng. J., 2023, 471, 144524 Search PubMed
.
- L. F. Cai, J. M. Zhan, J. Liang, L. Yang and J. Yin, Sci. Rep., 2022, 12, 3118 Search PubMed
.
-
X. Liu, C. Wang, C. Chen, Z. Pan, C. Gao, W. Lai, J. Zhao, T. Tian and W. Xiao, Coord. Chem. Rev., 2025, 544, 216927 Search PubMed
.
- L. Estevez, D. Barpaga, J. Zheng, S. Sabale, R. L. Patel, J. G. Zhang, B. P. McGrail and R. K. Motkuri, Ind. Eng. Chem. Res., 2018, 57, 1262–1268 Search PubMed
.
- L. Wang, Y. Ma, H. Liu, Y. Guo, B. Yang and B. Chang, Sep. Purif. Technol., 2025, 354, 128910 Search PubMed
.
- L. Zheng, W. B. Li and J. L. Chen, RSC Adv., 2018, 8, 29767–29774 Search PubMed
.
- P. C. Sahoo, R. Singh, P. Sivagurunathan, D. Singh, M. Kumar, R. P. Gupta and U. Srivastava, Int. J. Greenh. Gas Control, 2025, 142, 104340 Search PubMed
.
- M. Samandari, M. T. Broud, D. P. Harper and D. J. Keffer, J. Phys. Chem. B, 2024, 128, 8530–8545 Search PubMed
.
- M. T. Broud, M. Samandari, L. Yu, D. P. Harper and D. J. Keffer, J. Phys. Chem. C, 2023, 127, 13639–13650 Search PubMed
.
- Y. Ru, G. I. N. Waterhouse and S. Lu, Aggregate, 2022, 3, e296 Search PubMed
.
- Y. Liu, D. Zhong, L. Yu, Y. Shi and Y. Xu, Nanomaterials, 2023, 13, 437 Search PubMed
.
- A. Khayal, V. Dawane, M. A. Amin, V. Tirth, V. K. Yadav, A. Algahtani, S. H. Khan, S. Islam, K. K. Yadav and B. H. Jeon, Polymers, 2021, 13, 3190 Search PubMed
.
- C. Wang, A. Inazaki, T. Shirai, Y. Tanaka, T. Sakuta, H. Takikawa and H. Matsuo, Thin Solid Films, 2003, 425, 41–48 Search PubMed
.
- R. Dubrovsky, V. Bezmelnitsyn and A. Eletskii, Carbon, 2004, 42, 1063–1066 Search PubMed
.
- C. Ng, W. E. Marshall, R. M. Rao, R. R. Bansode and J. N. Losso, Ind. Crops Prod., 2003, 17, 209–217 Search PubMed
.
- I. M. Lima, A. McAloon and A. A. Boateng, Biomass Bioenergy, 2008, 32, 568–572 Search PubMed
.
- C. A. Toles, W. E. Marshall, M. M. Johns, L. H. Wartelle and A. McAloon, Bioresour. Technol., 2000, 71, 87–92 Search PubMed
.
- G. G. Stavropoulos and A. A. Zabaniotou, Fuel Process. Technol., 2009, 90, 952–957 Search PubMed
.
- D. A. Yang, H. Y. Cho, J. Kim, S. T. Yang and W. S. Ahn, Energy Environ. Sci., 2012, 5, 6465–6473 Search PubMed
.
- M. Ding, R. W. Flaig, H. L. Jiang and O. M. Yaghi, Chem. Soc. Rev., 2019, 48, 2783–2828 Search PubMed
.
- Y. Li, Y. Wang, N. Liu, B. Wang and R. Dong, Colloids Surf., A, 2023, 658, 130732 Search PubMed
.
- N. Kostkova, M. Vorokhta, M. Kormunda, R. Pilar, G. Sadovska, P. Honcova, E. Mikyskova, J. Moravkova and P. Sazama, Microporous Mesoporous Mater., 2024, 379, 113286 Search PubMed
.
- J. Li, M. Hou, Y. Chen, W. Cen, Y. Chu and S. Yin, Appl. Surf. Sci., 2017, 399, 420–425 Search PubMed
.
- H. Li, J. Bin Niu, K. J. Tay, M. C. Tan and H. Y. Low, ACS Sustainable Chem. Eng., 2024, 12, 2911–2920 Search PubMed
.
- H. J. Esfahani, A. Ghaemi and S. Shahhosseini, Sci. Rep., 2024, 14, 18871 Search PubMed
.
- Y. Gong and T. He, Small Methods, 2023, 7, 2300702 Search PubMed
.
- F. Eshraghi, M. Anbia and S. Salehi, J. Environ. Chem. Eng., 2017, 5, 4516–4523 Search PubMed
.
- H. Kiuchi, R. Shibuya, T. Kondo, J. Nakamura, H. Niwa, J. Miyawaki, M. Kawai, M. Oshima and Y. Harada, Nanoscale Res. Lett., 2016, 11, 127 Search PubMed
.
- R. Ben Said, J. M. Kolle, K. Essalah, B. Tangour and A. Sayari, ACS Omega, 2020, 5, 26125–26133 Search PubMed
.
- F. Rezaei and C. W. Jones, Ind. Eng. Chem. Res., 2013, 52, 12192–12201 Search PubMed
.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |