 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Multifaceted advances in TiO2-based photocatalysts for PFAS degradation: a critical review of mechanisms, modifications, and challenges
Avtar
Singh
 * and
Thiagarajan
Soundappan
* and
Thiagarajan
Soundappan

Department of Chemistry, School of Science, Navajo Technical University, Crownpoint, NM-87313, USA. E-mail: avtar.singh@navajotech.edu; avtarldh007@gmail.com
First published on 19th January 2026
Abstract
The global persistence of per- and polyfluoroalkyl substances (PFAS), often referred to as “forever chemicals,” poses significant environmental and human health risks, driving regulatory action and intensified research into effective remediation strategies. Photocatalysis has emerged as a promising, sustainable approach for PFAS degradation via light-driven redox processes. Titanium dioxide (TiO2) remains the benchmark photocatalyst due to its chemical stability, low toxicity, and strong oxidative potential; however, its practical application is limited by rapid electron–hole recombination, restricted visible-light absorption, and pH-dependent surface charge behavior, necessitating acidic conditions for optimal performance. These constraints reduce its efficacy against the robust carbon–fluorine bonds characteristic of PFAS and complicate large-scale deployment. Recent advances focus on multifunctional TiO2-based systems, including metal and nonmetal doped systems, carbonaceous composites, heterojunctions, molecularly imprinted polymers, and adsorptive concentrate-and-destroy supports. Integration with advanced oxidation and reactor-level engineering approaches, such as photoelectrocatalysis (i.e., photocatalysis under applied bias) and hybrid oxidants, has further advanced photocatalytic degradation technology under broader operating conditions. This review provides a critical comparative assessment of these strategies, highlighting mechanistic insights, structure–activity relationships, and practical limitations related to pH, stability, and scalability. By consolidating recent innovations and operational considerations, this work offers guidance for the rational design of efficient, field-relevant, and sustainable photocatalytic technologies for global PFAS remediation.
1. Introduction
Scientific and technological progress has transformed modern society, improving healthcare, sanitation, transportation, and daily life. Central to these advances, materials science has enabled breakthroughs, from polymers to semiconductors and nanomaterials, elevating global living standards. However, history highlights the unintended environmental consequences of innovation, exemplified by chlorofluorocarbons (CFCs), asbestos, dichlorodiphenyltrichloroethane (DDT), and plastics.1–4 Today, per- and polyfluoroalkyl substances (PFAS) represent a pressing concern. Often termed “forever chemicals,” PFAS are characterized by strong C–F bonds, high chemical stability, bioaccumulation potential, and extreme environmental persistence.5–8 First synthesized in the late 1930s and commercialized by the 1940s,9 PFAS have been extensively applied in Teflon™ coatings, Scotchgard™ treatments, aqueous film-forming foams (AFFFs), textiles, semiconductor manufacturing, food packaging, and aerospace industries.9,10 United States (US) annual production of basic fluoropolymers reached 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons in 2018, forming the start of the PFAS value chain10,11 and contributing to widespread contamination of industrial zones, remote ecosystems, Arctic wildlife, and human blood samples worldwide.12 Their persistence, mobility, and toxicity now pose severe environmental and public health challenges.13
000 tons in 2018, forming the start of the PFAS value chain10,11 and contributing to widespread contamination of industrial zones, remote ecosystems, Arctic wildlife, and human blood samples worldwide.12 Their persistence, mobility, and toxicity now pose severe environmental and public health challenges.13
Diverse remediation strategies have been explored for PFAS contamination, including adsorption (activated carbon, ion-exchange resins, MOFs, and engineered clays);14–20 advanced oxidation processes (AOPs) such as Fenton chemistry, persulfate activation, and electrochemical oxidation;21–23 photocatalysis;24 thermal methods (incineration and supercritical water oxidation);24–26 biological approaches with PFAS-tolerant microbial consortia;27–29 plasma-based treatments;30,31 membrane separation (nanofiltration and reverse osmosis);32,33 and hybrid strategies integrating ultrasound, photolysis, or catalysis.34–36 Among these, photocatalysis has gained attention as a sustainable, environmentally benign approach. By harnessing sunlight or artificial UV light, photocatalysis generates reactive oxygen species (ROS) capable of cleaving robust C–F bonds.24,37–39 While numerous new strategies and materials are being explored in this field, a few recent examples of advancements include carbazole-cored super-photoreductants for low-temperature PFAS and PTFE defluorination,40 CdIn2S4 micro-pyramids,41 Fe/g-C hybrids,42 rGO/WO3 nanoflowers,43 and Pb-doped TiO2/rGO composites,44 many achieving near-complete PFAS degradation through synergistic redox and radical-mediated pathways.
Within this context, TiO2 remains at the core of photocatalysis, alone and in hybrid systems, owing to its high stability, low toxicity, cost-effectiveness, and strong oxidative potential under UV irradiation.45–49 The d0 electronic configuration of Ti4+ results in Ti 3d-O 2p hybridization, producing a ∼3.0–3.2 eV bandgap suitable for UV-driven photocatalysis. Upon excitation, TiO2 generates electron–hole (e−–h+) pairs that drive ROS formation, particularly ˙OH and O2˙−, which are central to pollutant degradation.50,51 Photocatalytic performance is strongly influenced by crystal polymorphs (anatase, rutile, and brookite), lattice structure, and facet-specific reactivity. For example, anatase {001} facets (surface energy ∼0.90 J m−2) exhibit a higher reactivity than {101} facets (∼0.44 J m−2).52–54 A representative strategy involves selective Pt single-atom deposition on facet-engineered TiO2, stabilization of {001} facets using fluoride ions, APTMS (aminopropyltrimethoxysilane) anchoring to guide Pt precursors to {101}, and mild UV irradiation to reduce Pt to single atoms. This spatially separates oxidative and reductive sites; holes migrate to {001} for ˙OH generation, while Pt atoms on {101} trap electrons for reductive defluorination, enabling simultaneous oxidative–reductive PFAS degradation. Surface chemistry further modulates TiO2 activity, with hydroxyl groups, coordinatively unsaturated Ti sites, oxygen vacancies, and Ti3+ centers serving as active sites for adsorption, charge separation, and visible-light absorption.55–60 Controlled defect introduction via hydrogenation, electrochemical reduction, ion-thermal processing, ultrasonication, NaBH4 treatment, microwave irradiation, plasma, or laser ablation enhances photocatalytic efficiency by suppressing e−–h+ recombination and extending optical response. Additional strategies include metal and nonmetal doping for bandgap tuning, heterojunction construction and fabrication of plasmonic–carbonaceous composites for charge separation, co-catalyst loading to enhance e−–h+ utilization, surface functionalization to promote pollutant adsorption, modulation of reactive species for optimized ROS balance, fabrication of hierarchical nanostructures, and formation of mixed anatase–rutile interfaces that facilitate charge migration.61–65 Hydrophilic or fluorophilic surface tailoring has been shown to enhance interactions with polar and fluorinated substrates.66,67 Synergistic integration of TiO2 photocatalysis with electrochemical,68 photo-Fenton,69 or piezocatalytic70 processes generates additional radical pathways, while immobilization in thin films and porous monoliths improves scalability. TiO2-based photocatalytic systems, owing to their intrinsic stability, structural tunability, and broad adaptability, continue to serve as a cornerstone for ROS-mediated pollutants degradation. Fig. 1 shows key strategies for enhancing photocatalysts' performance in environmental remediation.
This review presents a comprehensive evaluation of TiO2-based photocatalysts for PFAS degradation, covering pristine and modified materials, including defect-engineered, single-atom, carbonaceous, heterojunction, and adsorptive designs. It critically examines operational parameters, mechanistic pathways, and structural modifications that govern adsorption, charge separation, reactive species generation, and oxidative–reductive defluorination. Hybrid and integrated strategies, such as photoelectrocatalysis, advanced oxidation, and reactor-level innovations, are discussed to address inherent limitations like charge recombination, UV dependence, and mass-transfer constraints. By connecting mechanistic insights with practical considerations, including stability, recyclability, matrix effects, energy efficiency, and scalability, this review provides guiding principles for designing next-generation TiO2-based photocatalysts that are efficient, robust, and field-relevant for sustainable PFAS remediation.
2. Chemical, environmental, and health context of PFAS
Per- and polyfluoroalkyl substances (PFAS) are a global environmental concern due to their chemical stability, persistence, and widespread application. PFAS originated in 1938 when Dr Roy J. Plunkett at DuPont accidentally synthesized polytetrafluoroethylene (PTFE) while developing chlorofluorocarbon refrigerants.71,72 Commercialized as Teflon™ in 1948, PTFE's robust C–F bonds confer thermal and chemical stability, catalyzing thousands of PFAS compounds used in firefighting foams, surfactants, textiles, microelectronics, and other applications.73,74 These properties also make PFAS “forever chemicals,” leading to persistence and bioaccumulation in soil, water, air, and biota.75–78 Despite phase-outs of legacy PFAS such as perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS), new analogues continue to be produced with similar mobility and toxicity.79 PFAS comprise over 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 compounds, broadly classified as non-polymeric or polymeric (Fig. 2).80,81 Non-polymeric PFAS contain a hydrophobic perfluoroalkyl chain (–CnF2n–) linked to polar groups (carboxylate and sulfonate), enabling surfactant applications like aqueous film-forming foams (AFFFs), metal plating, and cleaning agents.82 Polymeric PFAS, including PTFE and FEP (fluorinated ethylene propylene), are high-molecular-weight fluoropolymers used in cookware, electronics, and waterproof fabrics. The chain length and functional groups influence mobility, bioaccumulation, and degradability: short-chain PFAS are highly mobile yet hard to remove, whereas long-chain PFAS readily bioaccumulate.83,84 Emerging variants with unknown fate challenge regulatory and analytical frameworks, prompting tools like PFAS-Atlas for chemical mapping and prioritization.85,86
000 compounds, broadly classified as non-polymeric or polymeric (Fig. 2).80,81 Non-polymeric PFAS contain a hydrophobic perfluoroalkyl chain (–CnF2n–) linked to polar groups (carboxylate and sulfonate), enabling surfactant applications like aqueous film-forming foams (AFFFs), metal plating, and cleaning agents.82 Polymeric PFAS, including PTFE and FEP (fluorinated ethylene propylene), are high-molecular-weight fluoropolymers used in cookware, electronics, and waterproof fabrics. The chain length and functional groups influence mobility, bioaccumulation, and degradability: short-chain PFAS are highly mobile yet hard to remove, whereas long-chain PFAS readily bioaccumulate.83,84 Emerging variants with unknown fate challenge regulatory and analytical frameworks, prompting tools like PFAS-Atlas for chemical mapping and prioritization.85,86
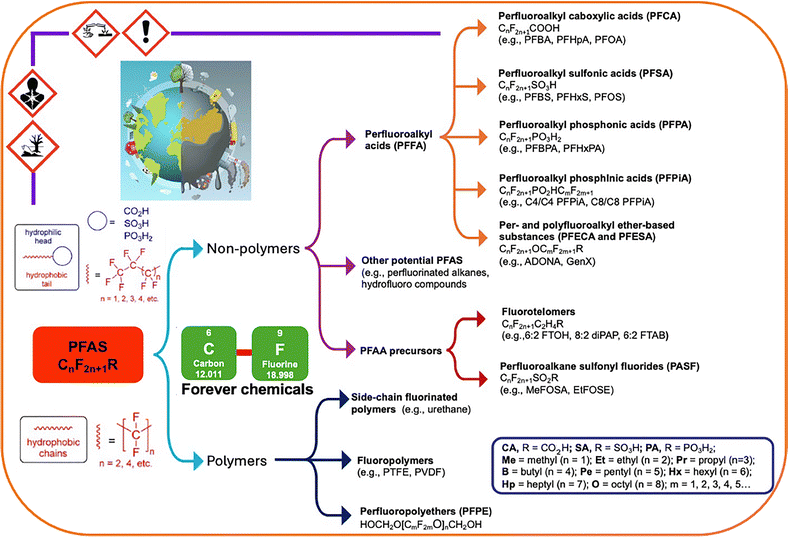 | ||
| Fig. 2 Classification of PFAS based on structural features: categorized into polymeric and non-polymeric groups, redraw from ref. 81. A few parts of this figure are adapted from open-source graphical elements available on Freepik (https://www.freepik.com), designed by macrovector/Freepik. | ||
PFAS are detected in surface water, groundwater, drinking water, soils, vegetation, and biota worldwide, including remote Arctic and Antarctic regions.87–91 HRMS and LC-MS/MS detect PFAS at parts-per-trillion levels, revealing widespread contamination and bioaccumulation.92–94 Human exposure occurs via ingestion, inhalation, and dermal contact.95–97 Epidemiological studies, the C8 Health Project (>69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 individuals), link chronic PFOA exposure to testicular and kidney cancers, thyroid disorders, ulcerative colitis, and pregnancy-induced hypertension.98,99 PFAS interact with nuclear receptors (PPARα, CAR, and PXR), perturb lipid metabolism, immunity, and hormonal regulation, and cross the placental barrier.100–102 Blood levels correlate with diet, smoking, and use of PFAS-containing products.103,104
000 individuals), link chronic PFOA exposure to testicular and kidney cancers, thyroid disorders, ulcerative colitis, and pregnancy-induced hypertension.98,99 PFAS interact with nuclear receptors (PPARα, CAR, and PXR), perturb lipid metabolism, immunity, and hormonal regulation, and cross the placental barrier.100–102 Blood levels correlate with diet, smoking, and use of PFAS-containing products.103,104
Ecologically, PFAS induce biochemical disturbances at environmental concentrations. Metabolomic studies reveal altered gene expression in zebrafish larvae, liver glycogen depletion, and shifts in earthworm metabolites;105–107 rodent studies report neurological and metabolic effects at low doses.108,109 Risk assessments indicate low-to-moderate global risk, though industrial discharges can elevate exposure.110–112 AFFFs remain a major source, with contamination documented at military, industrial, and civilian sites.112–120 Research now focuses on fluorine-free alternatives, supported by United States Department of Defence (DoD), Strategic Environmental Research and Development Program (SERDP) and Environmental Security Technology Certification Program (ESTCP) initiatives.120,121 In parallel, international regulations are also emerging: most Australian states restrict PFAS foams for non-aviation use,122,123 and in 2023, five European Union (EU) member states proposed banning all PFAS in firefighting foams.124 These exemptions highlight the need to integrate material innovation, regulation, and deployment to mitigate legacy and future PFAS exposure.
3. Prospective approaches to PFAS elimination
PFAS contamination in aqueous and soil matrices presents distinct remediation challenges. Water treatment strategies include adsorption, membrane filtration, and advanced oxidative or reductive methods, such as photocatalysis, electrochemical oxidation, electron beam irradiation, and hydrothermal alkaline treatment.125–127 While adsorption and membrane separation effectively concentrate PFAS, they often generate secondary waste requiring destructive post-treatment.125 Advanced oxidative and reductive processes target mineralization but typically demand high energy, specialized catalysts, or extreme conditions, limiting large-scale applicability. Hybrid treatment trains that combine selective concentration with destructive pathways improve degradation efficiency while mitigating cost and secondary pollution.125 However, complex aqueous matrices containing competing ions or natural organic matter can inhibit reactive species, underscoring the need for site-specific optimization.128 As summarized in Fig. 3, PFAS treatment technologies include both destructive methods, such as photocatalysis, electrochemical oxidation, and hydrothermal or mechanochemical degradation, and non-destructive approaches, including adsorption, membrane filtration, and phytoremediation.125 Hybrid systems that combine selective concentration with destructive pathways are also highlighted, illustrating strategies to enhance mineralization efficiency while minimizing secondary pollution.Soil remediation faces additional hurdles due to heterogeneity and strong C–F bonds promoting PFAS retention.129 Physical approaches, such as soil washing or amendment-induced immobilization, usually provide only temporary containment.130,131 Thermal strategies, including incineration and smoldering combustion (>500–900 °C), effectively destroy PFAS but require careful control of toxic fluorinated emissions.129,132–135 Energy-efficient smoldering of PFAS-laden granular activated carbon (GAC) and soils has achieved complete destruction, ∼44% of PFAS on GAC and ∼16% in soils, with emissions efficiently scrubbed by GAC.136 Hydrothermal and supercritical water oxidation under alkaline conditions can remove >95% of PFAS, though effluent management remains critical.137 Mechanochemical degradation using high-energy ball milling with additives (KOH and persulfate) provides non-thermal, onsite alternatives.138,139 Advanced oxidative and reductive methods, including electron beam irradiation and use of persulfate-based systems, generate radicals capable of cleaving C–F bonds but face high energy demands and matrix interference.125,138 Electrochemical remediation shows promise, particularly for short-chain PFAS, while phytoremediation offers a low-cost, sustainable option by sequestering PFAS in biomass for subsequent treatment.140–147
Artificial intelligence (AI) and machine learning (ML) offer transformative potential for catalyst design and process optimization. By analyzing large PFAS datasets and reaction pathways, ML can predict outcomes and guide selection of optimal materials and operational conditions.148–151 Hybrid systems combining photocatalysis with electrochemical or mechanochemical processes exploit synergistic effects, enhancing mineralization efficiency and reducing by-product formation. Circular economy strategies, such as mechanochemical defluorination recovering fluorine as reusable salts, further enable sustainable PFAS waste management.152
Among destructive methods, photocatalysis is particularly attractive, as it harnesses solar energy under ambient conditions to generate reactive oxygen species capable of mineralizing PFAS into environmentally benign products. Catalyst design approaches, including size-exclusion, charge heterogeneity, amphiphilic surface modification, and molecular imprinting, enhance PFAS adsorption and orientation, improving degradation even in complex matrices.153–155 TiO2-based photocatalysts are especially prominent due to their intrinsic tunability and versatility, allowing strategies such as facet engineering, doping, and heterojunction construction to create structurally and functionally diverse photocatalysts. This multifaceted platform underpins the focus of this review on strategies to enhance photocatalytic efficiency and selectivity for PFAS degradation.
4. Mechanistic foundations and limitations of TiO2
Titanium dioxide (TiO2) is widely recognized as a benchmark photocatalyst due to its strong oxidation potential, chemical stability, and cost-effectiveness.156 Upon absorption of photons with energy equal to or greater than its band gap (∼3.2 eV for anatase), electrons are excited from the valence band to the conduction band, generating e−–h+ pairs. Valence-band holes (h+) serve as potent oxidants, while conduction-band e− participate in interfacial reduction reactions, including oxygen reduction, leading to secondary reactive species under aerobic conditions3,157–159 (Fig. 4a). The overall photocatalytic efficiency is governed by the quantity of photo-generated charge carriers, their oxidation–reduction potentials, and charge-carrier recombination dynamics.Per- and polyfluoroalkyl substances (PFAS), particularly perfluorocarboxylates (PFCAs) and perfluorosulfonates (PFSAs), possess exceptionally strong carbon–fluorine (C–F) bonds,160 which underpin their environmental persistence and resistance to conventional radical-mediated oxidation.125 For representative compounds such as PFOA and PFOS, the fully fluorinated alkyl chains lack abstractable hydrogen atoms, rendering hydroxyl radical (˙OH) attack largely ineffective for initiating degradation.161 As a result, indirect oxidation pathways involving ˙OH contribute minimally to PFAS transformation, and reaction initiation is increasingly attributed to direct interfacial electron transfer between the pollutant and the photoexcited semiconductor via valence-band (VB) holes.161–163 For PFCAs, adsorption onto TiO2 surfaces facilitates direct VB hole oxidation, triggering Kolbe-type decarboxylation of the head group (Fig. 4b) and generating carbon-centered radicals that undergo stepwise chain shortening to progressively shorter PFCAs (e.g., PFHpA, PFHxA, PFPeA, PFBA, and PFPrA), accompanied by the formation of CO2 and fluoride ions.125,162,163 These transformations are surface-controlled, making adsorption affinity and interfacial charge-transfer efficiency the primary determinants of degradation kinetics. Although photoexcited electrons react readily with dissolved oxygen to form superoxide radicals (˙O2−), this species is widely regarded as ineffective for PFAS degradation, and its role remains controversial.162 In oxygenated systems, electron scavenging by O2 further suppresses reductive pathways by limiting the availability of excited electrons.164
Complementary photoreductive pathways (Fig. 4c) have also been reported, particularly under anoxic conditions and in the presence of hole scavengers.161,162 In these systems, conduction-band electrons or hydrated electrons (eaq−) are injected into the PFAS molecule, leading to direct reductive cleavage of C–F bonds (Fig. 4c) or, in some cases, reductively induced decarboxylation of PFCAs.165 However, such reductive mechanisms are strongly disfavored under environmentally relevant conditions due to efficient electron scavenging by oxygen and the generally low sorption affinity of semiconductors for PFAS.166 While some studies report that ˙O2− scavengers hinder PFOA degradation, suggesting a possible role in initiating decarboxylation via nucleophilic substitution, others propose that ˙O2− may indirectly generate ˙OH under acidic conditions, thereby contributing to oxidative pathways.162 Consequently, the role of superoxide remains unresolved. Ateia et al.163 proposed that PFAS degradation is primarily triggered by direct electron transfer to valence-band holes, with ˙OH and ˙O2− playing a minimal role in initiating the reaction. In contrast, earlier studies conducted by Hori et al.167 showed that the presence of moisture does not alter the primary degradation rate of fluorotelomer alcohols on TiO2 but does enhance the breakdown of fluorinated byproducts and the formation of CO2. This suggests that ˙OH radicals may be more important for the secondary oxidation and mineralization of intermediates rather than for the initial activation of PFAS molecules.
TiO2's intrinsic properties, including polymorphism, defect tolerance, electronic tunability, high dielectric constant, and chemical robustness, support diverse modification strategies. Its polymorphs (anatase, rutile, and brookite) can accommodate dopants, enabling band-gap modulation and the introduction of defect or mid-gap states that extend light absorption and influence charge-carrier dynamics. Facet engineering, particularly exposure of high-energy anatase {001} facets (∼0.90 J m−2), provides selective adsorption and reaction sites that enhance surface redox processes.168 Surface features such as coordinatively unsaturated Ti sites, oxygen vacancies, and Ti3+ species improve charge separation, broaden optical absorption, and facilitate PFAS adsorption and interfacial electron transfer.169–171 Morphological flexibility, from nanoparticles to ordered mesoporous films, allows tuning of surface area, porosity, and light-harvesting efficiency, directly influencing adsorption, charge migration, and catalytic performance.172,173 Combined with chemical robustness under diverse aqueous conditions, TiO2 remains durable for long-term photocatalytic applications.174
Compared with alternative photocatalysts such as ZnO, g-C3N4, or CdS, which can suffer from photocorrosion, limited charge mobility, or toxicity, TiO2 provides a balanced combination of stability, tunability, and cost-effectiveness.175,176 This multifaceted nature underpins rational design strategies, including doping, defect engineering, composite formation, and morphological control, that enhance PFAS degradation. The following sections focus on these intrinsic and engineered advances, highlighting their relevance for efficient PFAS mineralization under environmentally realistic conditions.
5. Pristine TiO2 photocatalysis performance and challenges
Among TiO2 polymorphs, anatase (Eg ≈ 3.2 eV) is the most widely employed for photocatalysis due to its more negative conduction band potential, slower e−–h+ recombination, and higher surface area compared to rutile (∼3.0 eV) and brookite (∼3.3 eV).177,178 Rutile offers superior thermal stability, and brookite remains underexplored due to synthetic challenges, while anatase generally exhibits higher photocatalytic activity for pollutant degradation under typical conditions, partly because its formation is kinetically favored at temperatures below ∼600 °C, leading to smaller crystallite sizes and a higher density of active surface sites.179,180 TiO2 synthesis via chemical (sol–gel, hydrothermal, and precipitation), physical (thermal oxidation and vapor deposition), or green/biological routes imparts control over size, crystallinity, morphology, and surface chemistry, which govern charge dynamics, reactive oxygen species (ROS) generation, and PFAS adsorption.181–184 Both commercial TiO2 (e.g., P25) and laboratory-tailored variants serve as references for mechanistic studies and catalyst development.Foundational studies show that while TiO2 efficiently mineralizes conventional organics, PFAS resist degradation due to strong C–F bonds and thermodynamically stable perfluoroalkyl backbones.185–187 Perfluorocarboxylic acid (PFCA) degradation typically follows a photo-Kolbe decarboxylation pathway, initiated at the terminal carboxyl group (Fig. 5a).186 Solution pH is a critical factor influencing TiO2 photocatalytic activity, as it affects surface charge, PFAS adsorption, aggregation, and reactive species generation.187 The point of zero charge (pHpzc) of TiO2 is approximately 6.25. PFOA exists as an anion (C7F15COO−) above its pKa (2.8).187 When pH ranges from 2.8 to 6.25, adsorption of C7F15COO− on the positively charged TiO2 surface (Ti–OH2+) is enhanced by electrostatic interactions, facilitating oxidative degradation. Above pH 6.25, the surface is negatively charged (Ti–O−), reducing adsorption and photocatalytic efficiency. Acidic conditions thus favor PFCA adsorption and degradation, whereas perfluorosulfonic acids (PFSAs) show limited reactivity, highlighting functional group-dependent degradability.185 Panchangama et al. reported >99% decomposition of long-chain PFCAs (PFOA, PFNA, and PFDA) under acidic conditions, though overall mineralization reached only 38–54%.186 Solution pH also influences the distribution and reactivity of key species. Acidic conditions favor heterogeneous ROS-driven oxidation, while alkaline conditions can enhance the formation of hydrated electrons (eaq−) in homogeneous systems, supporting reductive mineralization pathways. High H3O+ concentrations in acidic media can scavenge eaq−, reducing reductive contributions, whereas alkaline conditions stabilize eaq−, facilitating C–F bond cleavage and minimizing short-chain PFCA accumulation.161 Optimal TiO2 loadings (∼0.66 g L−1) enabled complete PFOA removal within 7 h; higher doses reduced efficiency due to light scattering and aggregation. Sonication-assisted TiO2 photocatalysis under near-neutral conditions improved mass transfer, ROS formation, and dispersion, achieving 64% PFOA degradation over 8 h, with sol–gel TiO2 outperforming P25.187 Surface charge effects were decisive: below the point of zero charge (∼pH 6–7), TiO2 is positively charged, enhancing adsorption; above it, repulsion limits efficiency (Fig. 5b).
 | ||
| Fig. 5 (a) Proposed photocatalytic decomposition cycle of PFCAs on TiO2 under UV light, involving ionization, electron transfer, decarboxylation, and oxidation. Successive CF2-unit losses yield shorter-chain PFCAs until complete mineralization to CO2 and F− ions. Reproduced from ref. 186 with permission from Elsevier BV, copyright 2009; (b) schematic of TiO2 surface charge effects on PFOA degradation across pH: Ti–OH2+ dominates at acidic pH, Ti–OH at neutral (pH 6–7, zero-point charge), and Ti–O− at basic pH. Despite expected higher degradation at low pH due to electrostatic attraction, experiments showed greater PFOA removal at basic pH (10), indicating that radical-driven processes outweigh surface charge effects. Reproduced from ref. 187 with permission from Elsevier BV, copyright 2009;187 (c) photocatalytic degradation of PFOA by TiO2 under 254 nm UV. Adsorption was <8%. After 180 min, removal reached 10.5% (O2), 12.4% (N2), 6.6% with oxalic acid (O2), and 86.7% with oxalic acid (N2). At pH 2.47 (HClO4), only 17.8% removal occurred. Rate constants were 1.16 × 10−2 min−1 (oxalic acid) vs. 1.0 × 10−3 min−1 (HClO4). Defluorination was 16.5% with TiO2/oxalic acid but <1% otherwise. Reproduced from ref. 188 with permission from Elsevier BV, copyright 2011; proposed reaction mechanism for PFOA degradation in the presence of a TiO2 photocatalyst, incorporating both photo-redox (d) and β-scission (e) pathways. Reproduced from ref. 189 with permission from Elsevier BV, copyright 2015.189 | ||
Reductive TiO2 photocatalysis complements oxidative pathways for highly resistant PFAS. Conduction-band electrons, stabilized by hole scavengers (e.g., oxalic acid), facilitate the formation of CO2˙− radicals that promote stepwise C–F bond cleavage.188,190–192 Wang et al. achieved 86.7% PFOA degradation at pH 2.47 with oxalic acid (as a hole scavenger) versus 17.8% with perchloric acid (used to maintain the same pH but without providing hole-scavenging ability), with the pseudo-first-order rate constant k = 1.16 × 10−2 min−1 (Fig. 5c), over 11 times higher than that under oxidative-only conditions.188 Kinetic and mechanistic studies highlight two key PFAS degradation pathways under UV/O2, a stepwise photo-redox route generating shorter-chain PFCAs and a direct β-scission pathway producing CO2 and HF189 (Fig. 5d and e). Fluoride accumulation on TiO2 surfaces can passivate active sites, limiting mineralization (∼32%) and fluoride release (∼29%). Oxygen availability modulates pathway selectivity: varying atmospheres (air, O2, and N2) shifts the balance between photo-redox and β-scission.193 Biosynthesized TiO2 nanoparticles offer sustainable alternatives. Plant extracts or microorganisms produce nanoscale TiO2 with tunable size and morphology, enhancing PFAS adsorption and ROS generation.194–196Albizia lebbeck-derived TiO2 achieved 95.6% PFOS degradation and 56.1% defluorination at pH 2 under UV-vis irradiation.196 Smaller crystallites (8.8 nm at pH 12 vs. 12.1 nm at pH 8) improved charge separation and surface activity, illustrating the dual advantage of green synthesis: environmental benignity and enhanced photocatalytic efficacy. All the above studies indicated that pristine TiO2 efficiently decomposes PFAS under optimized conditions, yet challenges including surface interactions, catalyst passivation, functional group specificity, and reaction condition control constrain complete mineralization, highlighting the need for advanced material design and hybrid strategies.
Murgolo et al.197 evaluated nanostructured TiO2 films on stainless steel mesh and P25 TiO2 (50–100 mg L−1) for groundwater PFOS (200–400 µg L−1) under UV (254 nm, 60 min, pH 7.85), observing negligible degradation and highlighting the importance of reaction optimization. Furtado et al.198 applied response surface methodology (RSM) to assess pH, catalyst dosage, and irradiation time. Acidic pH enhanced TiO2 protonation and PFOS adsorption, while moderate catalyst loadings optimized light penetration and active site availability. Pareto analysis identified pH as the dominant factor (Fig. 6a), and response surface plots (Fig. 6b) indicated optimal removal at low pH and moderate-to-high TiO2 loadings. Under optimized conditions (1.45 g L−1 TiO2, pH 4.0), PFOS (100 µg L−1) degradation reached 83% after 8 h, with a predicted maximum oxidation of 86% and an initial rate of 0.64 h−1 (Fig. 6c).
 | ||
| Fig. 6 Optimization of PFOS removal using TiO2/UV photocatalysis via response surface methodology (RSM): (a) Pareto chart showing the relative effects of suspension pH and TiO2 dosage on PFOS removal; bars crossing the significance line (p = 0.05) indicate statistically significant factors, highlighting pH as the dominant parameter, (b) response surface plot highlighting maximum removal at low pH with moderate catalyst loading, (c) time-dependent PFOS degradation measured by HPLC-MS/MS: (blue dots) TiO2/UV photocatalysis, (red dots) direct photolysis. Reproduced from ref. 198 with permission from Elsevier BV, copyright 2021; (d) FT-IR spectra of TiO2 during PFOA photodegradation: early spectra (2–4 h) show adsorption of PFOA and intermediates via C–F and carboxylate vibrations, while signals decrease after 9 h, reflecting progressive mineralization. Reproduced from ref. 199 with permission from Elsevier BV, copyright 2014; (e) stepwise decomposition of PFOA under UV-visible light, showing direct photolysis and TiO2-assisted photocatalysis. PFOA undergoes sequential C–C cleavage, F− elimination, and hydrolysis to form shorter-chain perfluorocarboxylic acids, ultimately mineralizing to CO2 and F−. Reproduced from ref. 200 with permission from American Chemical Society, copyright 2011; (f) mass balance of representative PFAA precursors after 4 h of TiO2/UV treatment, showing chain-length–dependent PFCA distributions. Reproduced from ref. 201 with permission from American Chemical Society, copyright 2022.201 | ||
Sansotera et al. studied PFOA photomineralization on commercial P25 TiO2, monitoring TOC and fluoride release and characterizing the photocatalyst by XPS and XRD.199 The reaction followed pseudo-first-order kinetics (kapp = 0.033–0.057 h−1) via Kolbe decarboxylation, producing C7 radicals, shorter-chain PFCAs, and HF. FT-IR spectra (Fig. 6d) indicated adsorption via carboxylate and C–F interactions within 2–4 h, with transient perfluorinated alcohols at 1210 cm−1, followed by signal reduction after 9 h as mineralization progressed. XPS indicated early-stage surface fluorine (85–90%) with mineralization plateauing at ∼32% and fluoride release at ∼29%. XRD revealed stable anatase (∼25 nm) and partially amorphized rutile (47 → 33 nm), highlighting that surface interactions, radical dynamics, and adsorption, rather than crystal structure, control efficiency.
The UV irradiation type strongly affects TiO2 performance. Low-pressure (LPUV, 254 nm) lamps provide narrow spectra, whereas medium-pressure (MPUV, 200–400 nm) lamps deliver higher photon flux and broader irradiation.202 Ochiai et al.200 achieved near-complete degradation of 5 mM PFOA using 1.5 wt% P25 under MPUV (250–375 nm, ∼600 mW cm−2 at 254 nm), with pseudo-first-order kinetics (k = 8.6 × 10−2 dm3 h−1), ∼6× faster than direct photolysis and 5–100× faster than other catalysts (e.g., tungstic heteropoly acid 2.1 × 10−3 and β-Ga2O3 1.7 × 10−2 dm3 h−1).203,204Fig. 6e describes the TiO2-assisted photocatalytic pathway where MPUV light initiates C–C bond cleavage in PFOA, leading to sequential shortening of perfluoroalkyl chains and eventual mineralization into CO2 and F−.
PhotoTOP is a TiO2-based platform for PFAS precursor characterization that operates under mild conditions (∼60 °C) without added chemicals, enabling direct LC-MS analysis with minimal matrix interference.201 It uses anatase TiO2 (∼390 nm, pHpzc 3.6) to generate steady-state hydroxyl radicals (˙OH, 1.1–1.5 × 10−12 M), allowing controlled oxidation that preserves the original perfluoroalkyl chain lengths. This contrasts with the higher-temperature TOP assay (85 °C) and persulfate-based methods, which often produce ultrashort PFCAs such as trifluoroacetic acid due to harsher conditions and stronger chain-shortening potential.201,205,206 PhotoTOP quantitatively converted eight PFAA precursors into PFCAs with mass balances of 82–115% after 4 h (Fig. 6f), performing reliably across complex matrices including soil extracts (15 mg L−1 dissolved organic carbon (DOC)), PFAS-coated papers, and technical mixtures. Unlike traditional methods, it also allows direct oxidation of solids, capturing non-extractable precursors, although this proceeds approximately 10× slower than extract oxidation. Compared to literature-reported TOP assays and chemical oxidation approaches, PhotoTOP offered milder, more selective oxidation, higher tolerance to matrix effects, and mechanistic insight into precursor-PFCA transformations (R2 = 0.88). Its main limitation is the need for a controlled UV setup, but this enables reproducible ˙OH generation and predictable oxidation kinetics. PhotoTOP provided a practical and mechanistically informative approach that bridges analytical characterization and photocatalytic treatment of PFAS precursors, complementing existing methods in the literature.
6. TiO2 modification and hybrid strategies
Pristine TiO2 generates reactive species capable of degrading PFAS; however, low visible-light absorption, rapid e−–h+ recombination, and the high stability of C–F bonds limit its performance. To address these challenges, TiO2 modifications and hybrid strategies aim to enhance light harvesting, charge separation, and surface reactivity. Approaches include metal and non-metal doping, single-atom incorporation, semiconductor coupling, immobilization on supports, and hybrid system design, all intended to maximize photocatalytic potential for persistent contaminants.6.1. Modified TiO2 systems
Metal and non-metal doping of TiO2 is a widely employed strategy to enhance photocatalytic activity under visible light, addressing the intrinsic wide bandgap of anatase TiO2 (∼3.2 eV) that limits UV-only activation207–210 (Fig. 7). Metal doping introduces mid-gap states, improves visible-light absorption, promotes charge separation, and generates oxygen vacancies that facilitate PFAS adsorption and reactive oxygen species formation.211 Transition metals (Fe, Co, and Cu) create Ti3+ centers and oxygen defects; noble metals (Ag, Au, and Pt) enhance interfacial charge transfer and suppress e−–h+ recombination; rare-earth metals (La3+, Ce3+, Gd3+, and Nd3+) serve as electron traps and increase surface area.211–215 Dopants are incorporated via sol–gel, hydrothermal, electrospinning, or surface impregnation methods.216,217 Non-metal doping extends visible-light activity without metal toxicity by introducing states above the valence band and narrowing the bandgap. Substitutional N (∼0.14 eV above the VB) and interstitial N (∼0.73 eV) reduce the bandgap to ∼2.6 eV, while S lowers it from 3.203 eV to 2.753–2.545 eV depending on concentration.218–221 Excessive doping can create recombination centers, necessitating careful optimization.222 Although, these modifications are extensively studied in general photocatalysis, their application for PFAS degradation on TiO2 remains emerging. Early reports suggest that such modifications enhance visible-light absorption, promote charge separation, and increase ROS generation, thereby potentially improving degradation kinetics and defluorination efficiency. | ||
| Fig. 7 Schematic illustrating the effects of metal and non-metal dopants on the properties and activity of TiO2-based photocatalysts. | ||
Estrillan et al.223 demonstrated visible-light photocatalytic degradation of PFOA using Fe and Nb co-doped TiO2 (Fe:Nb-TiO2) via sol–gel synthesis. XRD confirmed retention of the anatase phase with reduced crystallite size (11.4 nm), suppressing anatase-to-rutile transformation. Under UV-vis irradiation (200–600 nm) with 0.1 mM PFOA, pH 4.3, and a catalyst loading of 0.5 g L−1, Fe:Nb-TiO2 achieved nearly double the degradation efficiency of SG–TiO2, whereas AO-TiO2 P25 showed minimal activity. Enhanced performance was attributed to larger surface area (>120 m2 g−1vs. ∼114 m2 g−1 for SG–TiO2), Fe-induced bandgap narrowing, and Nb sites that trap photogenerated electrons, suppressing recombination and extending visible-light absorption. Maximum PFOA removal occurred at pH 4 due to electrostatic attraction between negatively charged perfluorooctanoate anions and positively charged TiO2, facilitating valence band hole-driven oxidation. At neutral or higher pH, surface speciation shifts to Ti–OH or Ti–O−, weakening adsorption, while the Fe–TiO2 point of zero charge (pzc 7.2) and PFOA's low pKa (2.8) limit adsorption and photocatalytic activity under alkaline conditions.186,187
UV-activated TiO2 modified with transition metals such as Fe and Cu was studied for PFOA degradation (Fig. 8a–c).224 XRD characterization revealed mixed anatase-rutile phases, with Fe–TiO2 containing zero-valent Fe and Cu–TiO2 showing Cu0 and Cu2O. Under UV irradiation (0.5 g L−1 catalyst, pH 5, 298 K, 12 h), Cu–TiO2 achieved 91% PFOA decomposition with 19% defluorination, outperforming UV/TiO2 (14% decomposition) and UV/Fe–TiO2 (69% decomposition, 9% defluorination). Enhanced activity of Cu–TiO2 arises from stronger electron trapping via Cu0/Cu+ species, suppressing e−/h+ recombination more effectively than Fe2+/Fe0 (standard reduction potentials: +0.34 V vs. −0.44 V), increasing valence band hole availability.225 Cu deposition also introduces localized energy levels in the TiO2 bandgap, enhancing UV absorption, while redox cycling of Cu facilitates electron transfer and perfluoroalkyl radical formation. Studied by Li et al.,226 noble-metal-doped TiO2 (Pt, Pd, and Ag) was investigated for PFOA degradation. Unlike transition metals, noble metals with high work functions efficiently capture electrons, suppress e−/h+ recombination, and enhance valence band hole-driven oxidation, with localized surface plasmon resonance (LSPR) extending absorption into the visible range.227,228 Li et al.226 reported Pt-, Pd-, and Ag-modified TiO2 (5 nm metal nanoparticles on 20–40 nm TiO2 aggregates) for PFOA degradation. At pH 3, 60 mg L−1 PFOA and 0.5 g L−1 catalyst, Pt–TiO2, achieved 100% decomposition and 34.8% defluorination after 7 h UV irradiation, with the pseudo-first-order rate constant 12.5× higher than that of pure TiO2. Pd–TiO2 and Ag–TiO2 achieved 94.2% and 57.7% decomposition with defluorination of 25.9% and 8.1%, respectively. Larger work functions of Pt (5.65 eV), Pd (5.55 eV), and Ag (4.26 eV) form Schottky barriers, efficiently trapping conduction-band electrons. Degradation proceeds via hVB+ -driven stepwise CF2 cleavage, producing shorter-chain PFCAs and fluoride ions (Fig. 8d and e). Rate constants increase with higher initial PFOA concentrations, demonstrating sustained photoactivity under elevated pollutant loads.
 | ||
| Fig. 8 Photocatalytic degradation of PFOA using TiO2-based materials: (a) comparison of degradation and defluorination efficiency: bare TiO2 (∼14% degradation in 12 h), Fe–TiO2 (∼69% degradation, 9% defluorination), (b) Cu–TiO2 (∼91% degradation, 19% defluorination), (c) schematic of PFOA decomposition under UV/TiO2: ˙OH-mediated C–C bond cleavage and stepwise defluorination to shorter-chain PFCAs and F−, with Fe or Cu doping enhancing ˙OH generation and electron–hole separation. Reproduced from ref. 224 with permission from Elsevier BV, copyright 2015; (d) rate constants for PFOA degradation using pure TiO2 and noble-metal-doped TiO2; Pt and Pd show superior electron-trapping effects, (e) mechanism of PFOA degradation on TiO2 and M–TiO2: UPLC-MS showed stepwise CF2 loss, mainly forming PFHpA (up to 19.1 mg L−1). Pt–TiO2 and Pd–TiO2 accelerated PFHpA generation (16–19 mg L−1 in 3–5 h) vs. Ag–TiO2 (9.7 mg L−1 in 7 h). About 80% F− remained in solution, with ∼20% lost to adsorption/gas. Decomposition followed Kolbe decarboxylation and HF elimination, yielding shorter PFCAs, CO2, and F−. Reproduced from ref. 226 with permission from Elsevier BV, copyright 2016; photocatalytic decomposition of PFOA by Pb–TiO2: (f) PFOA decomposition and defluorination at different pH values and under different atmospheres: highest at pH 3 (∼99%/30%) with air; low under N2 (∼26%/1%), (g) mechanistic pathway: UV-excited Pb–TiO2 generates perfluoroalkyl radicals that undergo stepwise HF elimination and hydrolysis, forming shorter-chain PFCAs and finally CO2 and F−. Pb improves efficiency by trapping electrons and reducing electron–hole recombination. Reproduced from ref. 229 with permission from Elsevier BV, copyright 2016. | ||
Pb-modified TiO2 has explored as an effective non-noble alternative. Pb introduced via photodeposition maintains the anatase-rutile framework, with XRD confirming Pb0, PbO, and PbO2 incorporation without lattice disruption.229 Pb species act as electron traps, enhancing charge separation. Under UV irradiation, Pb–TiO2 exhibited a pseudo-first-order rate constant of 0.5136 h−1, 32.5× higher than that of UV/TiO2 (0.0158 h−1), achieving nearly complete PFOA decomposition (99.9%) in 12 h, with a half-life of 1.3 h compared to 43.9 h for pristine TiO2. Acidic conditions (pH 3) and O2-rich environments further promoted decomposition via ˙OH formation, while N2 sparging drastically reduced activity (Fig. 8f). Mechanistic analysis (Fig. 8g) indicates sequential C–C cleavage, ˙OH attack, and chain shortening to CO2 and F−, with Pb acting as an efficient electron trap. The optimal Pb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 ratio (2
TiO2 ratio (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100) and catalyst loading (0.5–1.0 g L−1) maximized degradation efficiency. Compared to noble-metal doped-TiO2, Pb–TiO2 leverages acidic, oxygenated conditions to achieve faster kinetics and more complete degradation, highlighting the potential of strategic heavy-metal doping for PFAS remediation. Noble-metal doped TiO2 catalysts (Pt, Pd, and Ag) achieve faster and more complete PFOA degradation with higher defluorination than transition-metal doped TiO2, highlighting the effect of the dopant type on both reaction kinetics and partial mineralization. Pb–TiO2 also shows high activity, reaching 99.9% degradation and 22.4% defluorination in 7 h,229 comparable to Pt–TiO2 but slightly lower in defluorination. Among transition metals, Cu–TiO2 shows moderate performance, while Fe–TiO2 is less effective, and Fe:Nb–TiO2 mainly promotes stepwise chain-shortening.223,228
100) and catalyst loading (0.5–1.0 g L−1) maximized degradation efficiency. Compared to noble-metal doped-TiO2, Pb–TiO2 leverages acidic, oxygenated conditions to achieve faster kinetics and more complete degradation, highlighting the potential of strategic heavy-metal doping for PFAS remediation. Noble-metal doped TiO2 catalysts (Pt, Pd, and Ag) achieve faster and more complete PFOA degradation with higher defluorination than transition-metal doped TiO2, highlighting the effect of the dopant type on both reaction kinetics and partial mineralization. Pb–TiO2 also shows high activity, reaching 99.9% degradation and 22.4% defluorination in 7 h,229 comparable to Pt–TiO2 but slightly lower in defluorination. Among transition metals, Cu–TiO2 shows moderate performance, while Fe–TiO2 is less effective, and Fe:Nb–TiO2 mainly promotes stepwise chain-shortening.223,228
6.2. Single-atom catalysis (SAC) on TiO2
Single-atom catalysts (SACs) have gained considerable attention in photocatalysis due to maximal metal atom utilization, uniform active sites, and unique reaction pathways compared to conventional nanoparticles.230–232 TiO2-based SACs are particularly attractive for PFAS remediation, as isolated metal atoms can modulate charge separation, electronic structure, and interfacial interactions, enhancing pollutant degradation.233,234Weon et al. (2021) demonstrated the integration of facet-engineered TiO2 with single-atom Pt (Pt1) cocatalysts.52 Anatase TiO2 crystals with {001} and {101} facets were synthesized, with fluoride ions stabilizing {001} (∼80% surface area) and APTMS anchors selectively immobilizing PtCl62− on {101}, followed by photoreduction to atomically dispersed Pt1 (∼0.2 nm, confirmed by HAADF-STEM). This design exploited intrinsic band-driven charge separation; electrons migrated to {101} and holes to {001}, ensuring site-specific Pt1 placement and maximizing electron utilization. Pt1/facet-engineered TiO2 exhibited substantially more ˙OH generation than PtNP/TiO2 and showed ∼15× higher PFOA degradation (Fig. 9a). The single-atom design facilitated (i) efficient electron transfer from the TiO2 conduction band to Pt1, (ii) selective proton reduction forming Pt–Hδ− species, and (iii) hydrogen spillover onto TiO2 to generate reactive Ti–Hδ+ species that cleave C–F bonds, forming Ti–F (Fig. 9b). XPS confirmed Ti–F formation, and fluorine mass balance indicated ∼58% retention in Ti–F bonds, with minimal free F− or HF. This accumulation of Ti–F bonds represents a key catalyst deactivation pathway, as surface fluorination progressively blocks active Ti–OH sites and suppresses interfacial redox reactions. Importantly, Weon et al. demonstrated that this fluoride-induced deactivation is fully reversible. A mild NaOH washing step converted inactive Ti–F back to catalytically active Ti–OH (>Ti–F + OH− ↔ >Ti–OH + F−), restoring photocatalytic activity without Pt loss or aggregation. This regeneration strategy directly addresses long-term durability concerns by decoupling high defluorination efficiency from permanent catalyst poisoning. Moreover, the atomically dispersed Pt1 remained stable during reaction and regeneration cycles, indicating strong metal–support interactions and resistance to sintering or leaching, two common degradation routes in Pt nanoparticle-based systems.
 | ||
| Fig. 9 Photocatalytic hydrodefluorination of PFOA using Pt-loaded TiO2: (a) facet-engineered TiO2 shows negligible PFOA degradation, Pt nanoparticle/TiO2 improves performance moderately, and single-atom Pt1/facet-engineered TiO2 achieves the highest degradation, with the pseudo-first-order rate constant ∼15× higher than that of PtNP/TiO2, demonstrating efficient C–F bond hydrodefluorination. (b) Single-atom Pt1/TiO2 and Pt1/facet-engineered TiO2 show higher PFOA degradation than Pt nanoparticles due to efficient hydrogen spillover. Ti–F bonds form (58% of fluorine), with negligible free fluoride. Performance decline from Ti–F accumulation is reversible via NaOH. Facet engineering with Pt1 shifts the pathway from O2˙− to surface hydrogen species, enabling effective hydrodefluorination. Reproduced from ref. 52 with permission from American Chemical Society, copyright 2021;52 photocatalytic defluorination of PFOA by Bi single-atom-loaded TiO2. (c) Pure TiO2 shows low PFOA degradation (∼15%) and defluorination (∼2%). Fe or Pd doping slightly improves activity, while Bi single atoms (Bi SAs) significantly enhance defluorination (up to 70%), (d) under a xenon lamp, N-10Bi/TiO2 achieves complete PFOA removal within 2 h and high defluorination (∼85%) after 4 h, outperforming Cl-10Bi/TiO2, (e) short-chain PFCAs (PFHpA, PFHxA, and PFPeA) are detected as intermediates. N-10Bi/TiO2 shows rapid defluorination and maintains >65% activity over four cycles, demonstrating stability and practical potential. Reproduced from ref. 235 with permission from American Chemical Society, copyright 2023. | ||
As an alternative to noble-metal modification strategies, non-precious single-atom catalysts (SACs) have gained increasing attention. In this context, Bi single-atom catalysts (Bi-SACs) on TiO2, synthesized via green, UV-assisted methods, exhibit uniform metal dispersion and strong photocatalytic performance.235 N–Bi/TiO2 (from Bi(NO3)3) and Cl–Bi/TiO2 (from BiCl3 forming BiOCl nanoclusters) were investigated for PFOA degradation. N-10Bi/TiO2 achieved ∼85% defluorination under xenon lamp irradiation in 4 h, while Cl-10Bi/TiO2 achieved ∼76% (Fig. 9c–e). Pristine TiO2 and Fe- or Pd-modified TiO2 showed negligible activity. Enhanced performance of Bi–TiO2 arises from improved charge separation (photocurrent, EIS), side-on PFOA adsorption, and Bi⋯F interactions that polarize C–F bonds, as supported by DRIFTS, DFT, and FDTD simulations.236 Unlike Pt1/TiO2 systems that proceed via reductive hydrodefluorination, Bi–TiO2 operates through oxidative-defluorination, enabling rapid breakdown of PFOA and shorter-chain PFCAs. N-10Bi/TiO2 also demonstrated excellent recyclability, maintaining >90% PFOA removal over four cycles with only a slight decrease in defluorination (∼65%). These results establish Bi–TiO2 as an efficient, stable, and sustainable non-noble-metal SAC for PFAS remediation.
6.3. Hybrid photocatalysts
Hybrid photocatalysts have been widely investigated for PFAS remediation, wherein TiO2 is integrated with complementary materials to tailor its physicochemical and photochemical characteristics and broaden its functional scope. This section discusses key hybridization strategies studied for PFAS degradation, including coupling TiO2 with other semiconductors or oxides to form heterojunctions or Z-scheme architectures, use of carbon-modified TiO2 to facilitate electron transport and pollutant adsorption, and the integration of advanced material hybrids.Yao et al. developed mesoporous Sb2O3/TiO2 heterojunctions for PFOA degradation.239 SEM and TEM revealed bowl-like anatase TiO2 particles (∼10 nm) uniformly embedded with Sb2O3 nanocrystals, producing red-shifted light absorption and a narrowed bandgap (3.30 → 2.91 eV). At 10 ppm PFOA and 0.25 g L−1 catalyst, 3%-Sb2O3/TiO2 achieved 81.7% removal in 120 min, with the pseudo-first-order rate constant 4.2× faster than P25 (12.6 × 10−3 min−1vs. 6.3 × 10−3 min−1, Fig. 10a). ESR, PL, and TR-PL analyses identified O2˙− and h+ as dominant reactive species, while ˙OH was negligible, indicating a shift from conventional hydroxyl-mediated pathways (Fig. 10b).
 | ||
| Fig. 10 Photocatalytic degradation of PFOA: (a) comparison of calculated degradation rate constants for P25, TiO2, and TiO2 modified with 1%, 3%, 7%, and 10% Sb2O3, (b) schematic showing how the 3%-Sb2O3/TiO2 heterojunction enhances photocatalytic PFOA degradation. Reproduced from ref. 239 under the Creative Commons Attribution (CC BY), Frontiers, 2021; (c) PFOA degradation under simulated solar light, where P25/BiOBr achieved 99.7% removal in 100 min, significantly higher than that of BiOBr (13.4%), commercial In2O3 (16.8%), P25, or blank, attributed to its high adsorption capacity (qm = 21.94 mg g−1), (d) proposed photodegradation pathway over P25/BiOBr: holes generated on P25 migrate to BiOBr, oxidizing adsorbed PFOA, followed by stepwise decarboxylation and hydrolysis, producing short-chain PFCAs and ultimately CO2 and F−. Reproduced from ref. 240 with permission from Elsevier BV, copyright 2022; (e) fluoride ion release profiles during PFOA degradation under natural sunlight irradiation using BN/TiO2, compared with controls. Reproduced from ref. 37 with permission from Elsevier BV, copyright 2022; (f) degradation rate constants for different x% ReS2–TiO2 samples, (g) proposed electron transfer mechanism in the ReS2–TiO2 S-scheme heterostructure, illustrating band bending and internal electric field formation driven by Fermi level differences, (h) proposed photocatalytic mechanism of the ReS2–TiO2 system under UV irradiation, showing charge transfer, oxygen vacancy trapping, radical generation (˙O2− and ˙OH), and subsequent PFOA degradation. Reproduced from ref. 241 with permission from Elsevier BV, copyright 2025. | ||
BiOBr-modified P25 (P25/BiOBr) forms a p–n heterojunction, facilitating efficient charge separation.240 Under simulated solar irradiation, P25/BiOBr achieved 99.7% PFOA degradation within 100 min, outperforming P25 (<1%), BiOBr alone (13.4%), and commercial In2O3 (16.8%, Fig. 10c).242 ATR-FTIR and adsorption studies indicated unidentate binding of PFOA to Bi3+, enhancing surface adsorption (qm = 21.94 mg g−1) and direct hole-mediated oxidation. Mechanistically, photogenerated holes dominated, with negligible contribution from ˙OH, electrons, or superoxide anions, consistent with BiOBr's VB energy (1.99 V vs. NHE) being insufficient for water oxidation (Fig. 10d).243,244 In real water from Tangxun Lake (pH 3.5), degradation was efficient but slowed at natural pH 6.8 due to reduced adsorption and competing ions (Cl−, SO42−, and NO3−) and NOM, low NO3− or humic acid (∼10 mg L−1) slightly enhanced removal (as NO3− can act as scavengers of electrons), while higher concentrations (>100 mg L−1) inhibited it.245
The development of BN/TiO2 composites advanced PFAS photocatalysis degradation.37 Combining commercial TiO2 (P25, 80 wt% anatase/20 wt% rutile, 21 nm) with hexagonal BN (∼1 µm) via simple calcination forms a type-II n–n heterojunction that promotes efficient interfacial charge separation.246,247 Ultraviolet photoelectron spectroscopy (UPS) and diffuse reflectance UV-vis (DR-UV) spectroscopy revealed valence band positions of −7.0 eV (TiO2) and −6.9 eV (BN), with band gaps of 3.1 eV and 5.4 eV, respectively. This configuration allows TiO2 photoexcitation under UV-C (254 nm) and UV-A (365 nm), while BN primarily facilitates e−–h+ separation. Transient photocurrent measurements showed ∼47× and ∼70× higher steady-state current for BN/TiO2 relative to BN and TiO2, indicating enhanced charge separation and reduced recombination. Under UV-A, BN/TiO2 degraded 120 µM PFOA to <2.4 µM within 120 min (t½ = 55 min), compared to 77 µM remaining with TiO2 alone (t½ ≈ 350 min), achieving 37% versus 19% defluorination. Under UV-C, BN/TiO2 further accelerated mineralization (t½ = 13 min) and reached >50% defluorination after 60 min, outperforming BN (t½ = 10 min, 29% defluorination) and TiO2 (t½ = 80 min, 7% defluorination, Fig. 10e). BN/TiO2 also showed practical advantages, requiring fluence doses of ∼4–8 J cm−2 and EE/Ooutput values of 102 and 253 kWh m−3 order−1 under UV-C and UV-A, representing >10× improved energy efficiency compared to TiO2.
Chen et al. constructed an S-scheme ReS2–TiO2 heterostructure via electrostatic self-assembly, combining oppositely charged semiconductors to enhance interfacial electron transfer and suppress recombination.241 Transmission and electron microscopy (TEM) and elemental mapping confirmed uniform Re, S, Ti, and O distribution, while XPS and Raman analyses revealed increased oxygen vacancies (21 to 26%) and surface –OH groups (13 to 17%), boosting radical generation. Photocatalytic tests demonstrated that 2% ReS2–TiO2 achieved 98% PFOA removal with ∼75% defluorination in 120 min under UV, surpassing TiO2 (62%) and ReS2 alone. Radical trapping and ESR studies identified ˙O2− and ˙OH as dominant species, with ReS2 coupling significantly enhancing their formation. In situ XPS and DFT calculations elucidated the S-scheme mechanism: an internal electric field drives electron transfer from TiO2 to ReS2 in the dark, while upon illumination, TiO2 CB electrons migrate to the ReS2 VB, promoting charge separation, extending carrier lifetime, accelerating electron transfer, and increasing active site availability (Fig. 10f–h).248,249 For TiO2 heterojunctions, 3% Sb2O3/TiO2 achieved 81.7% PFOA degradation in 2 h,239 BN/TiO2 degraded 120 µM PFOA to <2.4 µM in 120 min with 37% defluorination under UV-A and >50% under UV-C in 60 min,37 and ReS2–TiO2 (2 wt%) removed 98% PFOA with ∼75% defluorination in 120 min.241
Fe/TNTs@AC demonstrated a “concentrate-and-destroy” mechanism, achieving 91.3% PFOA removal in 4 h under UV irradiation, outperforming TNTs@AC (23.8%), non-calcined Fe/TNTs@AC (68.7%), and calcined TNTs@AC (83.3%) (Fig. 11a).255 Fe modification and calcination enhanced defluorination (∼62%), 1.5–4× higher than that of other materials. Optimal calcination (550 °C) and 1 wt% Fe loading maximized performance. Fe/TNTs@AC adsorbed ∼99% of PFOA over pH 4–11 within 2 h, with α-Fe2O3/TNTs interacting electrostatically with the carboxylate head and AC providing hydrophobic and anion–π interactions with the tail.256 Adsorption orientation shifted from side-on (acidic/neutral) to tail-on (alkaline), slightly reducing defluorination at pH ≥ 9. Reusability tests confirmed stable adsorption (>99%) and ∼60% defluorination over six cycles, with minimal Fe (<1.7 mg L−1) and no Ti leaching. Mechanistic studies revealed h+-driven oxidation as dominant, with ˙OH mediating stepwise defluorination of perfluoroalkyl radicals, ˙O2− was negligible (Fig. 11b). Fukui index and ESP analyses identified the carboxylate head as the primary reactive site, supporting the proposed degradation pathway.257
 | ||
| Fig. 11 (a) Photocatalytic defluorination of PFOA, showing ∼62% conversion of organic fluorine to F− by Fe/TNTs@AC under UV irradiation, significantly higher than that of TNTs@AC and other controls, (b) proposed photocatalytic degradation mechanism of PFOA on iron (hydroxide-impregnated nanoparticles under UV irradiation, showing adsorption, hole/electron generation, radical formation, and stepwise defluorination via photo-Kolbe-like decarboxylation. Reproduced from ref. 255 with permission from Elsevier BV, copyright 2020; (c) kinetics of PFOS defluorination for neat AC, TNTs@AC, and Ga/TNTs@AC with varying Ga content under UV irradiation, (d) proposed mechanism for enhanced solid-phase photocatalytic degradation and defluorination of PFOS by Ga/TNTs@AC, highlighting multi-point adsorption, Ga-induced oxygen vacancies, charge separation, and generation of reactive species (h+ and O2˙−). Reproduced from ref. 258 with permission from Elsevier BV, copyright 2021;258 (e) Photodegradation and defluorination of PFOA pre-adsorbed on In/TNTs@AC under UV irradiation, showing rapid degradation (>50% in 30 min) and ∼60% defluorination after 4 h, (f) proposed stepwise photocatalytic degradation and defluorination pathways of PFOA on In/TNTs@AC, highlighting dual adsorption, In-facilitated redox cycling, and formation of perfluoroalkyl intermediates during UV irradiation. Reproduced from ref. 259 under the Creative Commons CC-BY licence, Elsevier BV, 2022. | ||
Titanate nanotubes (TNTs) have been further modified to enhance photocatalytic PFAS degradation through metal doping and carbon integration.258–261 Gallium-doped TNT-activated carbon composites (Ga/TNTs@AC) demonstrated synergistic adsorption-photocatalysis for PFOS removal. Both TNTs@AC and 2%Ga/TNTs@AC achieved near-complete PFOS adsorption within 10 min, far exceeding bare AC (equilibrium in 120 min) despite its higher surface area.258 Langmuir analysis indicated a maximum surface-area-normalized adsorption capacity of 0.225 mg m−2 for 2%Ga/TNTs@AC, ∼2.1× higher than that of AC, highlighting the combined effects of Ga doping and AC in creating electrostatic, hydrophobic, and π-anion binding sites.260,261 Under UV irradiation, 2%Ga/TNTs@AC achieved 75% PFOS degradation and 66.2% defluorination within 4 h, significantly outperforming TNTs@AC (13.1%/6.2%) and AC alone (Fig. 11c), with the underlying mechanism illustrated in Fig. 11d. The system exhibited broad pH tolerance (3.5–10.5), with alkaline conditions favoring O2˙− generation and defluorination. XPS and EPR confirmed Ga3+ induced oxygen vacancies that acted as electron traps, improving charge separation and ROS formation, supported by reduced PL emission.
Indium-doped TNT–AC composites (In/TNTs@AC) were developed for PFOA remediation.259 Characterization showed an interwoven TNT-AC framework, with In present mainly as In2O3 and minor In0 phases.262,263 XRD confirmed anatase retention with reduced interlayer spacing, while FTIR and N2 adsorption indicated partial surface-group removal and slightly reduced pore volume, favoring PFOA adsorption. Batch tests achieved >99% PFOA removal within 30 min via hydrophobic and electrostatic/Lewis acid–base interactions at In2O3 sites.264 Under UV irradiation, In/TNTs@AC achieved nearly complete PFOA degradation within 4 h with ∼60% defluorination, outperforming AC, TNTs@AC, and In2O3@AC due to synergistic adsorption and enhanced charge separation via heterojunction formation (Fig. 11e and f). TNT-based composites exhibit clear dopant-dependent behavior in PFAS degradation.255,258,259 Fe modification promotes redox-driven pathways and enhances defluorination, Ga doping improves oxygen-vacancy-mediated charge separation favoring sulfonated PFAS removal, and In doping enables combined hydrophobic and Lewis acid–base adsorption with In2O3 heterojunction-assisted photocatalysis, favoring carboxylated PFAS. Overall, dopant selection governs charge transport, vacancy chemistry, and PFAS-specific interactions, providing a mechanistic basis for rational design of TNT-based photocatalysts.
Multi-walled carbon nanotubes (MWCNTs) serve as supports to improve adsorption and charge separation.265 TiO2–MWCNT composites synthesized via sol–gel with varying TiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MWCNT ratios were tested under 365 nm UV (8 h) with 30 mg L−1 PFOA. Adsorption equilibrium on MWCNTs occurred within 30 min (∼18% PFOA). A 5
MWCNT ratios were tested under 365 nm UV (8 h) with 30 mg L−1 PFOA. Adsorption equilibrium on MWCNTs occurred within 30 min (∼18% PFOA). A 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 TiO2-MWCNT composite achieved 89% PFOA degradation, surpassing P25 (37%) and physical mixtures (60%) (Fig. 12a). Optimized 10
1 TiO2-MWCNT composite achieved 89% PFOA degradation, surpassing P25 (37%) and physical mixtures (60%) (Fig. 12a). Optimized 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 loading yielded 94% degradation, whereas 20
1 loading yielded 94% degradation, whereas 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 caused aggregation and reduced efficiency.
1 caused aggregation and reduced efficiency.
 | ||
Fig. 12 Photocatalytic degradation of PFOA and PFOS over various TiO2-based and modified photocatalysts: (a) comparison with a physical mixture of P25 and MWCNTs (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Reproduced from ref. 265 with permission from Elsevier BV, copyright 2012;265 (b) comparison of PFOA degradation after 12 h under UV irradiation using TiO2, TiO2–rGO composite, and GO control, showing enhanced removal with TiO2–rGO due to improved light penetration and reduced electron–hole recombination, (c) proposed photocatalytic degradation pathway of PFOA over TiO2–rGO, highlighting the role of rGO in enhancing charge separation, radical generation (˙OH and O2˙−), stepwise perfluoroalkyl cleavage, and eventual formation of CO2 and F−. reproduced from ref. 266 with permission from Elsevier BV, copyright 2018; (d) PFOA removal under UVC irradiation using TiO2, Pb, rGO, TiO2–Pb, TiO2/rGO, and TiO2–Pb/rGO, showing enhanced degradation with composite materials due to synergistic effects of rGO and Pb doping. Reproduced from ref. 267 with permission from Wiley, copyright 2023. 1). Reproduced from ref. 265 with permission from Elsevier BV, copyright 2012;265 (b) comparison of PFOA degradation after 12 h under UV irradiation using TiO2, TiO2–rGO composite, and GO control, showing enhanced removal with TiO2–rGO due to improved light penetration and reduced electron–hole recombination, (c) proposed photocatalytic degradation pathway of PFOA over TiO2–rGO, highlighting the role of rGO in enhancing charge separation, radical generation (˙OH and O2˙−), stepwise perfluoroalkyl cleavage, and eventual formation of CO2 and F−. reproduced from ref. 266 with permission from Elsevier BV, copyright 2018; (d) PFOA removal under UVC irradiation using TiO2, Pb, rGO, TiO2–Pb, TiO2/rGO, and TiO2–Pb/rGO, showing enhanced degradation with composite materials due to synergistic effects of rGO and Pb doping. Reproduced from ref. 267 with permission from Wiley, copyright 2023. | ||
TiO2 photocatalysis is significantly boosted through integration with reduced graphene oxide (rGO), which has been studied for the degradation of various pollutants.266–269 Ruiz et al. investigated TiO2–rGO composites for PFOA degradation under UV irradiation.266 While bare TiO2 removed only 24 ± 11% of PFOA after 12 h, TiO2–rGO achieved 93 ± 7% removal under identical conditions (Fig. 12b), representing an ∼4-fold improvement. The enhanced activity is attributed to: (i) one-atom-thick rGO sheets that reduce light screening, (ii) efficient electron transfer from TiO2 to rGO, and (iii) prolonged charge-carrier lifetimes, which enhance ˙OH and O2˙− generation (Fig. 12c). Total organic carbon decreased by 62%, and 20% of fluorine was removed through combined adsorption (6.8%) and partial volatilization of short-chain products. Kinetic analysis revealed chain-length-dependent degradation: kPFPeA = 2.14 h−1 > kPFHxA = 0.54 h−1 > kPFHpA = 0.27 h−1 > kPFOA = 0.163 h−1, indicating faster degradation of shorter-chain PFCAs. In subsequent studies, TiO2–rGO degraded both dichloroacetic acid (DCA) and PFOA, but complete mineralization occurred only for DCA, highlighting the strong recalcitrance of fluorinated compounds.268 Chowdhury et al. developed a synergistic Pb-doped TiO2/rGO system (TiO2–Pb/rGO) for PFOA remediation under UVC irradiation267 (Fig. 12d). The composite achieved 98% PFOA removal and 32% defluorination, surpassing TiO2–Pb, TiO2/rGO, and TiO2–Fe/rGO. Pb outperformed Fe for defluorination (34% vs. 15%) due to its more favorable standard reduction potential. The TiO2–Pb/rGO system was effective under UVA, UVB, and UVC, with UVC slightly more efficient, achieving a low electrical energy per order (EEO) of 4.05 kWh m−3 for 98% PFOA removal.
Ultra-small TiO2 quantum dots (QDs) have been developed and uniformly distributed on supports to enhance photocatalytic efficiency by maximizing the active surface area.269–271 Sodium dodecyl sulfate (SDS) was used to regulate Ti3+ ion distribution, minimizing TiO2 particle size. The resulting QDs (∼2.55 nm) were assembled into three-dimensional sulfonated graphene (SG) aerogels, forming 3D SG–TiO2 QD aerogels.272 Compared to conventional TiO2 nanoparticles (15–40 nm), these QDs exhibit enhanced photon absorption and reduced e–h+ recombination. Photocatalytic PFOA degradation followed first-order kinetics, with QDa (SG–TiO2 QDs synthesized at higher SDS concentration) achieving the highest rate constant (kapp = 1.898 × 10−4 s−1), outperforming QDb (SG–TiO2 QDs synthesized at lower SDS concentration) (1.530 × 10−4 s−1) and aggregated TiO2 NPs (9.283 × 10−5 s−1) (Fig. 13a). The synergistic combination of SG aerogels and TiO2 QDs enhanced adsorption, promoted uniform QD dispersion, reduced band gap, and improved photogenerated charge utilization, enabling persistent and high-efficiency PFAS degradation even in flow-through setups (Fig. 13b).
 | ||
| Fig. 13 (a) PFOA degradation profiles and simulated concentrations over time for 3D SG–TiO2 QDa, 3D SG–TiO2 QDb, 3D SG–TiO2 NP, and 3D SG, showing faster kinetics for smaller TiO2 QDs and supporting stepwise formation of shorter-chain PFCAs, (b) proposed mechanism for PFOA removal by 3D SG–TiO2 QDa, showing combined adsorption on the hydrophobic aerogel surface and photocatalytic degradation via h+-induced and ˙OH-mediated pathways, leading to stepwise formation of shorter-chain PFCAs and fluoride release. Reproduced from ref. 272 with permission from Elsevier BV, copyright 2020; (c) photodegradation of PFOA over TiO2, Exfol.CQD/TiO2, and MCQD/TiO2 under UVC (200–280 nm). Exfoliated CQDs enhance electron transfer, reduce electron–hole recombination, and improve photocatalytic efficiency compared to bare TiO2, (d) proposed mechanism for PFOA decomposition using TiO2 and CQD/TiO2 photocatalysts. Upon light irradiation, electron–hole pairs are generated; holes in the valence band directly oxidize PFOA, while electrons in the conduction band participate in reactive oxygen species formation, enabling stepwise defluorination and formation of shorter-chain PFCAs. Reproduced from ref. 273 with permission from Elsevier BV, copyright 2025; (e) defluorination efficiency of PFOA and short-chain PFCAs (C3–C6) under UVC and visible light using TiO2 and PCQD/TiO2 photocatalysts. The PCQD/TiO2 composite showed significantly higher defluorination percentages (46.6–56.2% for PFOA and 10.9–12.9% for short-chain PFCAs) compared to pristine TiO2, (f) PFCA degradation over PCQD/TiO2 proceeds via two main pathways: chain shortening, where reactive holes and radicals break C–C bonds to form shorter-chain PFCAs, and H/F exchange, where α–C–F bonds are sequentially replaced by hydrogen, releasing fluoride ions. Reactive oxygen species (˙OH and O2˙−) generated by the photocatalyst drive these processes. Reproduced from ref. 274 with permission from Elsevier BV, copyright 2025. | ||
Carbon quantum dots (CQDs) have emerged as effective modifiers of TiO2 photocatalysts, enhancing visible-light absorption and promoting efficient charge separation.275,276 High-purity CQDs synthesized via hydrothermal treatment of mussel shell biomass (MCQD) and electrochemical exfoliation of graphite rods (Exfol.CQD)273 were coupled with TiO2 to form composite photocatalysts. Photocatalytic experiments under UVC and visible light demonstrated substantially improved degradation of perfluorooctanoic acid (PFOA) and short-chain PFCAs (C3–C6). Stepwise decomposition of longer-chain PFCAs (e.g., PFHxA-C6 and PFPeA-C5) to shorter intermediates (PFBA-C4 and PFPrA-C3) was observed, with higher intermediate concentrations under UVC irradiation (PFHxA 11.5 µg L−1, PFPeA 11.9 µg L−1) than under visible light (PFHxA 9.8 µg L−1, PFPeA 8.2 µg L−1), reflecting more rapid generation of reactive species at higher photon energies. Corresponding defluorination efficiencies reached 32% (visible) and 34.5% (UVC) for Exfol. CQD/TiO2, compared to 11.6% and 21.8% for bare TiO2. Photocurrent and IMVS/IMPS analyses confirmed faster electron transfer (τt = 0.317 ms) and longer hole lifetimes (TR = 35.9 ms), highlighting the role of CQDs in charge separation. Fig. 13c shows improved PFOA photodegradation by CQD/TiO2 under UVC due to enhanced charge separation, while Fig. 13d illustrates a mechanism involving direct hole oxidation and ROS-driven stepwise defluorination.
Similarly, peanut-shell biomass-derived CQDs (PCQD/TiO2) extended TiO2's photoresponse from UV to visible light and reduced the band gap from 3.04 to 2.92 eV, acting as electron reservoirs and further enhancing e−/h+ separation.274 PFOA and other long-chain PFCAs degraded into shorter intermediates, with defluorination efficiencies significantly improved (PFOA: 56.2% UVC, 46.6% visible vs. 23.2% and 11.6% for bare TiO2) (Fig. 13e). Both Exfol.CQD and PCQD composites exhibited stepwise PFCA degradation driven by superoxide (˙O2−), hydroxyl radicals (˙OH), and photogenerated holes, maintaining stable performance over multiple cycles. Fig. 13f illustrates the mechanism: extended light absorption, improved e−–h+ separation, and reactive radical generation drive chain shortening, decarboxylation, and H/F exchange, achieving efficient mineralization. Among the three CQDs, PCQD/TiO2 demonstrates the highest overall efficiency, achieving 67.2% degradation of long-chain PFOA under UVC and 48.5% under visible light, while also effectively degrading short-chain PFCAs such as PFHxA (41.3%) and PFPeA (52.1%). Exfol.CQD/TiO2 performs comparably for long-chain PFOA (69% UVC, 50% visible) but is less effective for short-chain PFCAs (PFHxA 40%, PFPeA 35%, PFBA 31.6%, and PFPrA 21.6%). In contrast, mussel shell-derived CQDs (MCQD)/TiO2 consistently exhibits the lowest activity (PFOA 29% UVC, 20% visible) with limited short-chain PFCA data. This quantitative comparison underscores that both the CQD precursor and synthesis method critically influence TiO2 photocatalytic performance, with biomass-derived PCQDs offering a sustainable and broadly effective approach for PFAS remediation under both UVC and visible light.
In one approach, hydrothermally synthesized MXene/TiO2 composites demonstrated a layered structure with TiO2 nanoparticles anchored on MXene sheets, as confirmed by SEM, XRD, and XPS.282 These composites coupled rapid adsorption with photocatalysis: PFOA adsorption reached equilibrium within 30 min (qm ≈ 16.06 mg g−1), following pseudo-second-order kinetics and Langmuir isotherms, with efficiency strongly influenced by pH. Under UV irradiation, MXene/TiO2 achieved 94.6% degradation and 58.4% defluorination within 9 h, surpassing UV alone, UV/MXene, or UV/TiO2 systems (Fig. 14a). The Schottky barrier at the MXene–TiO2 interface facilitated hole transfer, improving e−–h+ separation and extending catalyst durability. Comparative analysis with advanced photocatalytic systems including MXene nanosheets, Ti4O7/MXene, COFs/MOFs and ZIF-67/Au-PCN reveals that the MXene/TiO2 system has competitive advantages in persistent pollutant remediation. In a separate study, an intercalation strategy using deep eutectic solvents (DESs) was developed to overcome the limited interlayer spacing of Ti3C2 MXene, which typically hinders in situ TiO2 crystal growth.283 The resulting Ti3C2-DES exhibited expanded c-lattice parameters compared to the hydrofluoric acid (HF)-etched MXene, allowing greater water intercalation and more effective Ti atom oxidation. This promoted in situ TiO2 crystal growth with exposed (001) and (101) facets, forming heterojunctions for enhanced charge separation. Combined with the high conductivity of the Ti3C2 substrate, this system enabled efficient carrier transport, achieving nearly 100% PFOA removal and 49% defluorination in 16 h. In contrast, Ti3C2-HF/TiO2 and commercial P25 TiO2 exhibited only modest efficiencies (22% removal, 12% defluorination, and 30% removal, 9% defluorination, respectively) (Fig. 14b and c), consistent with the morphological and mechanistic insights highlighted in Fig. 14d and e. Quantitatively, these results illustrate that faster C–F bond cleavage does not always correspond to higher overall PFOA removal. Altogether, these studies282,283 demonstrate that MXene/TiO2 hybrids can be engineered through hydrothermal or DES-driven strategies to maximize synergistic adsorption-photocatalysis, enhance charge separation, and significantly boost PFAS degradation, highlighting the versatility of MXenes as co-catalysts and structural supports for next-generation water treatment photocatalysts.
 | ||
| Fig. 14 (a) Photocatalytic degradation of PFOA under different systems: UV alone, MXene, hydrothermally treated MXene (180 °C), TiO2, and MXene/TiO2 composite. The MXene/TiO2 composite achieved the highest degradation (94.6%) due to enhanced light absorption and improved electron–hole separation, while UV alone showed minimal PFOA removal (13.96%). Reproduced from ref. 282 with permission from Elsevier BV, copyright 2025; photocatalytic degradation of PFOA using Ti3C2/TiO2-based catalysts: (b) PFOA removal under adsorption, UV photolysis, Ti3C2-HF/TiO2, P25, and Ti3C2/TiO2–80, (c) fluoride ion (F−) generation and defluorination efficiency for the same systems, (d) TiO2 distribution within Ti3C2-DES showing intercalation into interlayers, (e) TiO2 growth on the Ti3C2-HF surface for comparison. The intercalation strategy (Ti3C2/TiO2–80) significantly enhances both PFOA degradation (>99.9%) and defluorination (49%) compared to conventional TiO2 systems. Reproduced from ref. 283 with permission from Elsevier BV, copyright 2020; three-dimensional RS (response surface) diagrams illustrating the optimal operating conditions for maximum treatment efficiency: (f) effect of catalyst dosage (0.5–0.7 g) and zeolite mixing ratio (∼10%) on efficiency; (g) influence of the zeolite ratio (∼10%) and reaction temperature (up to 85 °C); (h) effect of the zeolite ratio (∼10%) and pH (up to 13). The diagrams indicate that treatment efficiency improves with decreasing zeolite ratio, increasing temperature, and higher pH under the specified conditions. Reproduced from ref. 284 with permission from Elsevier BV, copyright 2024; (i) PFOA degradation by MIL-125, F-MIL-125, TiO2@MIL-125, and F-TiO2@MIL-125 showed fastest kinetics for F-TiO2@MIL-125 with Kapp = 1.221 × 10−4 s−1, higher than that of other TiO2-based catalysts, while only TiO2@MIL-125 and F-TiO2@MIL-125 continued significant degradation after 2 h. Reproduced from ref. 66 with permission from Elsevier BV, copyright 2022.66 | ||
TiO2/zeolite nanocomposite materials were studied for photocatalytic pentafluoropropionic acid (PFPA) degradation, leveraging the complementary properties of both components.284 TiO2 provides photocatalytic activity, while zeolites offer thermally and chemically stable scaffolds with high surface area and porous, hydrophobic structures that pre-concentrate pollutants, prevent nanoparticle aggregation, and, when modified (e.g., alum-coated), enhance fluoride adsorption. Waste-derived Na–Y zeolite (68.7% purity) supported TiO2via sol–gel or hydrothermal methods, resulting in higher surface area (273 m2 g−1vs. 46.3 m2 g−1 for P25), reduced bandgap (2.75 eV vs. 3.28 eV), and preserved structural integrity (EDX Ti![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Si
Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al = 42.4
Al = 42.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 34.9
34.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22.7). Systematic evaluation using ANOVA and response surface (RS) methodology identified pH (alkaline, pH 13) as the most influential factor, followed by dosage and the TiO2/zeolite ratio, with reaction temperature contributing moderately. The RS 3D plots (Fig. 14f–h) visually depict the interplay between these factors, highlighting the optimal ranges of dosage, zeolite ratio, temperature, and pH for maximum defluorination. Under these optimal conditions (0.5 g L−1 catalyst, 10% TiO2/zeolite, 85 °C, pH 13), PFPA defluorination reached 58.7–60.2%, with elevated surface area, pore structure, and charge properties promoting ˙OH-mediated degradation. These results highlight the synergy of TiO2 and zeolite scaffolds and demonstrate a statistically validated approach for optimizing photocatalytic PFAS remediation.
22.7). Systematic evaluation using ANOVA and response surface (RS) methodology identified pH (alkaline, pH 13) as the most influential factor, followed by dosage and the TiO2/zeolite ratio, with reaction temperature contributing moderately. The RS 3D plots (Fig. 14f–h) visually depict the interplay between these factors, highlighting the optimal ranges of dosage, zeolite ratio, temperature, and pH for maximum defluorination. Under these optimal conditions (0.5 g L−1 catalyst, 10% TiO2/zeolite, 85 °C, pH 13), PFPA defluorination reached 58.7–60.2%, with elevated surface area, pore structure, and charge properties promoting ˙OH-mediated degradation. These results highlight the synergy of TiO2 and zeolite scaffolds and demonstrate a statistically validated approach for optimizing photocatalytic PFAS remediation.
Further advancement is represented by F-TiO2@MIL-125, integrating tailored adsorption and photocatalytic functionalities.66 While MOF (MIL-125(Ti)) displayed smooth plate-like morphology (1 nm), TiO2@MIL-125 retained disc-like structure with smoother surfaces, and F-TiO2@MIL-125 preserved structural integrity with superficial nano-debris despite fluorine functionalization. BET analysis showed a surface area decrease from 1201.71 m2 g−1 (MIL-125) to 85.25 m2 g−1 (F-TiO2@MIL-125), accompanied by an increase in pore diameter (2.13 → 12513.48 nm), favoring PFOA diffusion. F-TiO2@MIL-125 exhibited rapid adsorption (within 200 s) and superior monolayer capacities (185.151 µmol g−1), outperforming MIL-125, F-MIL-125, and TiO2@MIL-125. Photocatalytic tests (Fig. 14i) under simulated sunlight demonstrated that only TiO2@MIL-125 and F-TiO2@MIL-125 sustained PFOA degradation beyond 2 h, with F-TiO2@MIL-125 achieving the highest rate constant (Kapp = 1.221 × 10−4 s−1), surpassing TiO2–rGO (0.453 × 10−4 s−1) and fluorinated TiO2 (0.360 × 10−4 s−1). This enhanced performance was attributed to a narrowed bandgap (3.12 eV), redshifted absorption (460 nm), increased VB electron density, and improved electron–hole separation, as confirmed by UV-vis, Mott–Schottky, photocurrent, and EIS analyses. Mechanistically, PFOA removal involved F⋯F interactions for enrichment, followed by h+ and ˙OH-mediated oxidation through stepwise defluorination. These studies highlight that MXenes, zeolites, and MIL-125-derived supports enhance TiO2 photocatalysis via complementary mechanisms: adsorption, heterojunction-facilitated charge separation, structural stability, and tailored surface chemistry, achieving superior PFAS removal and defluorination compared to conventional TiO2 catalysts.
6.4. Advanced photocatalysis-centric approaches
Recent developments in TiO2-based photocatalysis have focused on integrated strategies that improve overall performance by combining structural and functional modifications. Among these, photoelectrocatalysis (PEC) combines photocatalysis with an applied bias to accelerate interfacial reactions, while advanced oxidation processes (AOPs) exploit synergistic reactive oxygen species (ROS) pathways to mineralize persistent contaminants efficiently.285–288 Molecularly imprinted photocatalysts (MIPs) offer selective adsorption and degradation by creating tailored recognition sites for specific pollutants.289–291 This section highlights the mechanistic advantages and potential of these advanced strategies for PFAS degradation. | ||
| Fig. 15 (a) Photoelectrocatalytic degradation of PFOA on GO/TNAs under UV light was tested with external bias from 0 to 2.0 V. Degradation increased with applied potential due to enhanced electron transfer from TNAs to graphene oxide sheets. Reproduced from ref. 295 with permission from Elsevier BV, copyright 2018;295 photocatalytic degradation of PFAS using the BEEP reactor: (b) concentration changes of PFCAs after 24 h, showing complete removal of long chains (C12–C17) and progressive breakdown of shorter chains (C8–C12) into smaller intermediates, (c) removal of PFSAs, with the longest PFDS (C10) achieving 90% degradation, while shorter chains (C7–C9) show lower removal, (d) mass spectra of PFTeDA (C14) during BEEP treatment, illustrating stepwise decarboxylation and formation of smaller PFCAs ([C8F17]− and [C6F13]−), (e) MS spectra of PFOS (C8) showing ∼30% removal and 15% defluorination after 24 h, highlighting BEEP's efficiency in degrading both long- and short-chain PFAS, including resistant compounds like PFOS and PFOA. Reproduced from ref. 296 with permission from Royal Society of Chemistry, under CC BY-NC 3.0;296 effect of UV power and PFAS chain length on removal efficiency: (f) influence of UV intensity on PFOA removal: in photolysis (PL), reducing power from P1 to P2 decreased removal from 68% to 50%, whereas in photoelectrocatalysis (PEC), removal remained high (65–72%) due to the catalytic activity of the TiO2-coated mesh. (g) Removal of PFAS with different chain lengths under PL and PEC: in PL at P2, removal increased with decreasing chain length (PFHpA 18.5% → PFBA 61%), while at P1, removal ranged 55–61% for PFHpA, PFHxA, and PFPeA and 89% for PFBA. In PEC, removal rates at P2 were equal or higher than at P1, demonstrating efficient degradation across all chain lengths. Reproduced from ref. 297 under the Creative Commons CC-BY 4.0. | ||
Field-relevant PEC studies further support these findings. In groundwater from the Veneto Region, Italy, the site of one of the largest PFAS environmental disasters in Europe, a total of 48 PFAS species, including long- and short-chain compounds, were monitored, with up to 9 species detected in the wells.297 The PEC treatment was carried out using immobilized TiO2 photoanodes (TiO2 film grown on titanium mesh by plasma electrolytic oxidation), achieved overall PFAS reduction of 63% in well 1 and 65% in well 2, with individual removal efficiencies ranging from 96% (PFOA) to 1% (PFBS).297 The average degradation order followed PFOA > PFHpA > PFHxA = PFPeA > PFBA for PFCAs and PFOS > PFHpS for PFSAs. Laboratory tests in ideal PFOA solutions confirmed that PEC degradation proceeds via decarboxylation followed by stepwise CF2-unit loss, with transient formation of shorter-chain intermediates. Laboratory comparisons between photolysis (PL) and PEC under two UV lamp power settings (P1 = higher, P2 = lower) illustrate the advantages of bias-assisted PEC: as shown in Fig. 15f, reducing UV intensity from P1 to P2 decreased PL removal from 68% to 50%, while PEC removal remained stable or slightly increased, ranging from 65% to 72%.297Fig. 15g shows that for individual PFCAs, PL removal strongly depended on the chain length and light intensity, whereas PEC maintained consistent or higher removal across all chain lengths regardless of UV power. These results indicate that PEC is less dependent on radiation density and that bias-assisted charge separation is a key driver of robust kinetics. In terms of operational efficiency, PEC was more energy-efficient than PL: EEO values were 36 kWh m−3 for PEC versus 46–59 kWh m−3 for PL, outperforming most other advanced oxidation processes and showing comparable performance to plasma and advanced reduction processes. PEC, including advanced configurations such as BEEP, has shown potential for both laboratory and field-relevant PFAS-contaminated waters, enabling high removal and defluorination rates while maintaining energy efficiency and stability under varying UV conditions. These studies also indicate the importance of non-target analyses to further elucidate reaction pathways and identify by-products, as well as the potential value of pilot-scale reactors for evaluating energy consumption and process feasibility.
Photocatalytic ozonation represents a highly effective TiO2-AOP strategy.302,303 Pairing TiO2 with ozone generates additional ˙OH, complementing photocatalytic oxidation. Using this approach, 99.1% PFOA degradation and 44.3% defluorination were achieved within 4 h (Fig. 16a).304 TiO2/PMS systems were evaluated for PFOA degradation under both visible and UV light.305 Under optimized conditions (0.25 g L−1 TiO2, 0.75 g L−1 PMS, pH 3, 300 W visible light), 50 mg L−1 PFOA was degraded nearly completely within 8 h (Fig. 16b), with a pseudo-first-order rate constant of 0.310 h−1, approximately 11× higher than that of TiO2 alone (0.028 h−1). Scavenger studies indicated that sulfate radicals (SO4˙−) from PMS and photogenerated holes (h+) from TiO2 were the primary reactive species. In real wastewater, PFOA removal decreased to 65% (influent) and 82% (effluent) in 8 h (k = 0.136 and 0.070 h−1) due to coexisting organic compounds, highlighting matrix effects (Fig. 16c). Under UV light (254 and 185 nm, 32 W), degradation reached nearly 100% within 1.5 h, reflecting higher catalyst absorbance and stronger photon energy. For comparison with other TiO2-based systems, TiO2/PMS exhibits the highest apparent kinetics (k = 1.09 h−1),305 followed by Pt-modified TiO2 (k = 0.726 h−1) and Pd-modified TiO2 (k = 0.438 h−1), while Ag-modified TiO2 shows the lowest reaction rate (k = 0.126 h−1).226 This comparison reflects differences in kinetic performance among TiO2-based systems when evaluated strictly by apparent rate constants.
 | ||
Fig. 16 (a) PFOA degradation and defluorination over 4 h under different treatments. Sole ozonation: 0.5% F defluorination; direct UV photolysis: 17.5% decomposition, 8.3% defluorination; TiO2 photocatalysis (UV/TiO2/O2): 46.4% decomposition; UV/O3: 27.1% decomposition, 10.6% defluorination; photocatalytic ozonation (UV/TiO2/O3) achieved 99.1% decomposition and 44.3% C–F cleavage, highlighting strong synergistic effects. Reproduced from ref. 304 with permission from Elsevier BV, copyright 2016; photocatalytic degradation of PFOA (50 mg L−1) using TiO2/PMS: (b) under weak visible light (30 W) or darkness, PMS, TiO2, and TiO2/PMS showed negligible removal. Under powerful visible light (300 W), TiO2 alone removed ∼20% of PFOA, PMS alone was ineffective, and TiO2/PMS achieved nearly 100% degradation within 8 h, demonstrating a strong synergistic effect, (c) in real wastewater samples, TiO2/PMS under 300 W visible light degraded 65–82% of PFOA within 8 h (k = 0.136 h−1 for influent, 0.070 h−1 for effluent), lower than that in pure water (k = 0.310 h−1) due to coexisting organics. UV light (254 and 185 nm) maintained degradation efficiency comparable to pure water, showing stable photocatalytic performance in complex matrices. Reproduced from ref. 305 with permission from Elsevier BV, copyright 2020; (d) fate of PFAS in pilot-scale AOP systems. Most targeted PFAS showed no significant removal, except PFHxA and PFHxS, which decreased by 7% and 11%, respectively, in the O3/H2O2 effluent at Site 3. All other PFAS increased after treatment, with PFBS rising by 405% and PFOA/PFOS increasing by 14% and 13%, respectively. Variations between AOP technologies and sites reflect differences in precursor composition and source water quality. Reproduced from ref. 306 with permission from Elsevier BV, copyright 2022; (e) PFAS degradation after 4 h under different treatments. Photocatalysis and photocatalytic ozonation showed significantly higher removal of total PFASs, PFOA, PFHxS, and 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS compared to photolysis (p < 0.05). Ozonation alone removed only ∼4% and was not statistically different from adsorption. Reproduced from ref. 307 with permission from Elsevier BV, copyright 2022. 2 FTS compared to photolysis (p < 0.05). Ozonation alone removed only ∼4% and was not statistically different from adsorption. Reproduced from ref. 307 with permission from Elsevier BV, copyright 2022. | ||
Venkatesan et al.306 evaluated seven pilot-scale AOPs for PFAS and 1,4-dioxane removal from New York groundwater, including UV/TiO2. While these AOPs transformed PFAS precursors, complete mineralization was not achieved, and concentrations of individual PFAS often increased due to precursor conversion. Specifically, average increases were observed for PFBS (≈405%, range 0–1220%), PFOA (≈14%, 0–48%), PFOS (≈13%, 3–25%), PFHxA (≈1%, −7 to 9%), PFHpA (≈3.8%, 0–9.5%), PFHxS (≈3.3%, −11 to 13%), and PFNA (≈2%, 0–5.2%) (Fig. 16d). These increases were dependent on UV and oxidant dose, indicating that hydroxyl radical-based AOPs primarily facilitated precursor transformation rather than full degradation. Importantly, at one site, PFOA concentrations exceeded the New York State drinking water standard (10 ng L−1) after treatment, highlighting the importance of considering AOP-induced PFAS formation when designing treatment systems. These findings highlight the challenges associated with AOPs, including limited removal efficiency and the potential for increasing certain PFAS concentrations, which complicates water treatment and regulatory compliance.
Photocatalytic ozonation, combining WO3/TiO2 catalysts with ozone under UVA-visible light, has been applied for the removal of five PFAS (PFOA, PFHxS, PFBS, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTS, and GenX) from water.307 Four catalysts with varying WO3 contents (0, 1, 3, and 5 wt%) were synthesized and the 5 wt% WO3/TiO2 catalyst (TW5) exhibited optimal properties and achieved 82% methylene blue removal in model tests. PFAS degradation after 4 h remained low (ΣPFAS removal ≈16%, range 4–26%), with photocatalytic ozonation only marginally improving removal compared to photocatalysis alone (Fig. 16e). Photolysis and ozone photolysis were less effective, and ozonation alone had a negligible effect. These results demonstrate both the potential and current limitations of AOPs for PFAS removal, highlighting the need to optimize catalyst design and treatment conditions across multiple PFAS types.
2 FTS, and GenX) from water.307 Four catalysts with varying WO3 contents (0, 1, 3, and 5 wt%) were synthesized and the 5 wt% WO3/TiO2 catalyst (TW5) exhibited optimal properties and achieved 82% methylene blue removal in model tests. PFAS degradation after 4 h remained low (ΣPFAS removal ≈16%, range 4–26%), with photocatalytic ozonation only marginally improving removal compared to photocatalysis alone (Fig. 16e). Photolysis and ozone photolysis were less effective, and ozonation alone had a negligible effect. These results demonstrate both the potential and current limitations of AOPs for PFAS removal, highlighting the need to optimize catalyst design and treatment conditions across multiple PFAS types.
Similarly, research has applied MIPs for selective PFAS removal. One strategy involves modifying TiO2 nanotube arrays with MIPs, using PFAS molecules (e.g., PFOA) as templates. The imprinted cavities selectively capture target PFAS near the TiO2 surface, facilitating localized photocatalytic degradation.316 Wu et al. demonstrated that MIP–TiO2 nanotubes (MIP-TiO2 NTs) selectively degrade PFOA in real secondary effluent. The material showed higher degradation efficiency for PFOA than that for PFHA, PFOS, or 2,4-dichlorophenoxyacetic acid (2,4-D), with apparent rate constants significantly greater for PFOA. In secondary effluent (TOC 10.2 mg L−1, pH 7.2), MIP–TiO2 NTs achieved 81.1% PFOA removal in 8 h, comparable to 84% in pure water, whereas non-imprinted TiO2 NTs reached only 50.2%. Furthermore, surface-molecularly imprinted TiO2 nanotubes (S-MIP–TiO2 NTs) was studied for enhanced photocatalytic degradation of PFAS in secondary municipal wastewater effluents.317 S-MIP–TiO2 NTs achieved significantly higher PFOA degradation under 8 h UV irradiation compared to TiO2 NTs and non-imprinted controls, with defluorination efficiency markedly improved. Adsorption studies indicate selective binding driven by functional group recognition and electrostatic interactions at pH 4–6. Photocatalytic reactions follow Langmuir–Hinshelwood kinetics, resulting in rate constants exceeding those of nonmoleculary imprinted polymer-TiO2 NTs (NIP–TiO2 NTs). This study demonstrated that S-MIP–TiO2 NTs can selectively and efficiently degrade multiple short- and long-chain PFCs from real wastewater effluents at environmentally relevant concentrations. Complementing nanostructured MIP–TiO2 systems, 3D-printed TiO2–polylactic acid (PLA) composites were developed for pilot-scale PFAS treatment in landfill leachate.318 Customized 3D-printed tiles containing 20 wt% TiO2 achieved over 80% PFAS removal, including PFOS, PFNA, PFDA, and PFOSAm, within 24 h, with even higher efficiency upon extended retention. This study highlighted the potential of additive manufacturing for creating scalable and efficient photocatalytic systems adaptable to real-world PFAS remediation.
7. Critical assessment of parameters, mechanisms, and modifications
Table 1 summarizes the comparative performance of TiO2-based photocatalysts, including pristine, doped, composite, and immobilized systems, for PFAS degradation, highlighting key design strategies, degradation efficiencies, and operational stability. A critical comparative evaluation of these systems reveals clear performance hierarchies. Pristine TiO2 (including P25) exhibits low degradation efficiency and defluorination (<30%), serving mainly as a benchmark.186,193,239 Noble-metal-modified TiO2 (Pt–TiO2) achieves complete PFOA degradation in 7 h with high kinetics (0.727 h−1) and substantial defluorination (∼35%).52,226 Heterojunctions such as BiOBr/TiO2 provide high defluorination (∼65%) and full degradation in 8 h.235 Carbon-supported hybrids (Fe/TNTs@AC) combine fast kinetics, high degradation (∼90%), and excellent stability.255,259 Emerging MXene/TiO2 systems show up to 94.6% degradation and ∼58% defluorination, providing scalable, noble-metal-free alternatives.282,283 Photoelectrocatalytic TiO2 systems achieve the highest overall performance (∼99.5% degradation, 1.63 h−1) though requiring applied bias and more complex reactors.296,297| Material | PFAS studied | Amount of PC; amount of PFAS; solution volume | Light used | Rate constant | Degradation/removal; defluorination, time | Cycle | Ref. |
|---|---|---|---|---|---|---|---|
| Pristine TiO 2 photocatalysis | |||||||
| TiO2 | HFBA, TFA, PFPA, NFPA, PFOA, NFBS, HFOS | 2 g L−1; 1.5–6 mM; 50 mL | UV light, 75 W, 1.4–7.2 mW cm−2 | 0.0024–0.0123 h−1 | CO2: 12–54%, F−: 15–64%, 60 h | — | 185 |
| TiO2 | PFOA, PFDA, PFNA | 0.66 g L−1; 48–120 µM, with 0.15 M HClO4; — | UVC light, 16 W, 0.45 mW cm−2 | 0.1–0.94 h−1 | Decomposition: 86–100%, 7 h, mineralization: 30–54%, 7–48 h | — | 186 |
| TiO2 (commercial RdH; sol–gel; sonication-assisted) | PFOA (120 µM) | 0.66 g L−1; 120 µM; 2 L (reactor) | UV light, 8–16 W | 0.0342 h−1 (RdH), 0.1309 h−1 (sol–gel) with ultrasound | Degradation: 22%, 7 h (RdH); ∼40%, 7 h sol–gel; ∼64%, 8 h (sol–gel + ultrasound) | — | 187 |
| TiO2 | PFOA + intermediates | 0.5 g L−1; 24 µM; 500 mL | UV light, 23 W | 1.16 × 10−2 min−1 | Degradation: 86.7%, 3 h; defluorination: 16.5%, 3 h | — | 188 |
| TiO2 (biosynthesized) | PFOS | —; 200 µg L−1; 50 mL | UV light | — | Degradation: 95.6%, 2.5 h; defluorination: 56.1%, 2.5 h | — | 196 |
| TiO2 | PFOS | 1.45 g L−1; 100 µg L−1; 100 mL | UV light, 250 W | 0.64 h−1 | Degradation: 86%, 8 h | — | 198 |
| TiO2 | PFOA | 0.66–1.0 g L−1; 0.0040–0.0120 M; 1 L | UV light, 500 W, 75–95 W m−2 | 0.0332–0.0572 h−1 | Mineralization: 32%, 4 h; F−: 29%, 6 h | — | 193 |
| TiO2 | PFOA | 1.5 wt%; 5 mM; 0.1 L | MPUV light, 600 mW cm−2 | — | 8.6 × 10−2 dm3 h−1, 4 h | — | 200 |
| TiO2 | PFBA, PFHxA, PFOA, PFHxS, PFOS; precursors: 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2/8 2/8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 diPAP, 6 2 diPAP, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2/8 2/8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 sFTSA, 6 2 sFTSA, 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2/8 2/8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 FTCA, PFOSA, N-EtFOSAA 2 FTCA, PFOSA, N-EtFOSAA |
430 mg L−1;100 µg L−1; solution: 1 L | UV/TiO2 (PhotoTOP), Pyrex glass, ∼60 °C, steady state | — | Degradation (of precursors):100%, 2–4 h | — | 201 |
| TiO2 | PFOA | 0.5 g L−1; 60 mg L−1; 120 mL | UV light, 125 W, 5.3 mW cm−2 | 0.058 h−1 | Degradation: 31.1%, 7 h; defluorination: 3.3%, 7 h | — | 226 |
| TiO2 (mesoporous) | PFOA | 0.25 g L−1; 10 ppm; 20 mL | UV light, 4 W | 6.3 × 10−3 min−1 | Degradation: 55.9%, 2 h | — | 239 |
| TiO2 | PFOA | 0.1–0.2 g L−1; 10 mg L−1 | UV light, 30 W; Xe lamp, 300 W | — | Degradation: 15%, 4 h | — | 235 |
| TiO2 | PFOA + intermediates PFHpA, PFHxA, PFPeA, PFBA | 0.05–0.5 g L−1; 0.24 mmol L−1; 0.8 L | Mercury lamp 200–600 nm | Degradation: 24 ± 11%, 12 h | — | 266 | |
| TiO2 | PFOA | 0.3 g L−1; 0.24 mmol L−1; 0.8 L | UV-A/B/C, visible, 150 W | 0.018 h−1 | Degradation: 10%; defluorinaion: 2% | — | 268 |
| TiO2 | PFPA | 0.5 g L−1; 10 mg L−1; solution: n/a | UV light, 8 h | — | TiO2: 48.4% defluorination | 284 | |
| TiO2 | PFOA | 0.33–0.66 g L−1; 10 mg L−1; 21 mL | UV light, 15 W | 0.0068 h−1 | — | — | 267 |
| UV/TiO2 pilot system | PFBS, PFHxA, PFHpA, PFHxS, PFOA, PFOS, PFNA | TiO2 slurry; PFAS 0.3–8.2 ng L−1; flow 26.5 L min−1 | 32 low-pressure UV lamps (7 kW) | Not reported | PFAS increase with UV power; ΣPFAS up to 103% at Site 2 | 1 Phase | 306 |
| Doped-TiO 2 systems | |||||||
| Fe:Nb–TiO2 | PFOA | 0.5 g L−1; 0.1 mM; 600 mL | MP mercury lamp 150 W, | — | Stepwise degradation to shorter PFCAs (C6, C5, and C4), fluoride ions released | — | 223 |
| Fe–TiO2 | PFOA | 0.5 g L−1; 50 mg L−1; 1 L | UV light, 400 W, 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 lux 000 lux |
0.0015 min−1 | Degradation: 69%, 12 h; defluorination: 9%, 12 h | — | 224 |
| Cu–TiO2 | PFOA | 0.5 g L−1; 50 mg L−1; 1 L | UV light, 400 W, 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 lux 000 lux |
0.0031 min−1 | Degradation: 91%, 12 h; defluorination:19%, 12 h | — | 224 |
| Pt–TiO2 | PFOA | 0.5 g L−1; 60 mg L−1; 120 mL | UV light, 125 W, 5.3 mW cm−2 | 0.7267 h−1 | Degradation: 100%. 7 h; defluorination: 34.8%, 7 h | — | 226 |
| Pd–TiO2 | PFOA | 0.5 g L−1; 60 mg L−1; 120 mL | UV light, 125 W, 5.3 mW cm−2 | 0.4369 h−1 | Degradation: 94.2%, 7 h; defluorination: 25.9%, 7 h | — | 226 |
| Ag–TiO2 | PFOA | 0.5 g L−1; 60 mg L−1; 120 mL | UV light, 125 W, 5.3 mW cm−2 | 0.1257 h−1 | Degradation: 57.7%, 7 h; defluorination: 8.1%, 7 h | — | 226 |
| Pb–TiO2 | PFOA | 0.5 g L−1; 50 mg L−1; 1.1 L (reactor) | UV light | 0.5136 h−1 | Degradation: 99.9%, 12 h; defluorination: 22.4%, t1/2 1.3 h | — | 229 |
| TiO2–Pb | PFOA | 0.33–0.66 g L−1; 10 mg L−1; 21 mL | UV light, 15 W | 0.0571 h−1 | Removal: 80%, 24 h; defluorination: 12%, 24 h | — | 267 |
| Single-atom catalysis (SACs) on TiO 2 | |||||||
| Facet-engineered TiO2, PtNP/facet-engineered TiO2, Pt1/facet-engineered TiO2 | PFOA | 0.25 g L−1; 100 µM; 80 mL | UV light, 5 W, 7.87 mW cm−2 | Negligible, NA, 15× PtNP | Significant | 10 (Pt1) | 52 |
| N/Cl-Bi/TiO2 variants | PFOA | 0.2 g L−1; 10 mg L−1; 100 mL | UV light, 30/300 W | — | Defluorination: 55–85%, 4 h | 4 | 235 |
| TiO 2 coupled with other semiconductors or oxides | |||||||
| 1–10%-Sb2O3/TiO2, | PFOA | 0.25 g L−1; 10 ppm; 20 mL | UV light, 4 W | 12.6 × 10−3 min−1 | Degradation: 10–81.8%, 2 h | 5 | 239 |
| P25/BiOBr | PFOA | 0.5 g L−1; 100 mg L−1; 1 L | 300 W, Xe lamp, 320–780 nm, 120 mW cm−2 | — | Degradation: 99.7%, 100 min; defluorination: 39.8%, 100 min | 4 | 240 |
| 1–5% ReS2–TiO2 | PFOA, PFHxA, PFPeA, PFOS, PFDA, PFHpA | 25 mg, 2 mg L−1 | UV light | 0.021–0.030 min−1 | PFOA, degradation: 98%, 2 h; defluorination ∼75%, 2 h | — | 241 |
| BN/TiO2 | PFOA | 0.5 g/L; 120 µM; 20 mL | 365 nm UV-A, 254 nm UV-C, natural sunlight | t½ = 55 min (365 nm UV-A), t½ = 13 min (254 nm UV-C), t½ = 1.7 h (sunlight DI water), t½ = 4.5 h (sunlight model water) | Defluorination: 37% (365 nm, 4 h), >50% (254 nm, 1 h), 60% (sunlight, DI water, 4 h), 68% (sunlight, DI water, 7 h), 31% (sunlight, model water, 7 h) | 4 | 36 |
| Carbon-based hybrid photocatalysts | |||||||
| TiO2–MWCNT composites | PFOA | 1.6 g L−1; 30 mg L−1; 250 mL | UV light, 300 W | — | Removal: 94–100%, 8 h | — | 265 |
| TNTs@AC | PFOA | 4.0 g L−1; 100 µg L−1; 10 mL | UV light, 21 mW cm−2 | Removal: 83.3%, 4 h; defluorination: 41%, 4 h, | — | 255 | |
| Fe/TNTs@AC | PFOA | 4.0 g L−1; 100 µg L−1; 10 mL | UV light, 21 mW cm−2 | 0.918 h−1 | Removal: 91.3%, 4 h; defluorination: ∼62%, 4 h | 6 | 255 |
| In/TNTs@AC | PFOA | 4 g L−1; 400 µg L−1; 10 mL | UV light, 2.28 mW cm−2 | — | Removal: 100%, 4 h; defluorination: 60%, 4 h | 4 | 259 |
| TiO2–rGO (5 wt% rGO) | PFOA | 0.1 g L−1; 0.24 mmol L−1; 0.8 L | UV-A/B/C, visible, 150 W | 0.211 h−1 | Removal: 86%, 8 h; defluorination: 30%, 8 h | — | 268 |
| TiO2/rGO | PFOA | 0.33–0.66 g L−1; 10 mg L−1; 21 mL | UV light, 15 W | 0.0410 h−1 | Removal: 70%, 24h; defluorination: 25% | — | 267 |
| TiO2–Pb/rGO | PFOA | 0.33–0.66 g L−1; 10 mg L−1; 21 mL | UV light, 15 W | 0.2193 h−1 | Removal: 98%, 24 h; defluorination: 32%, 24 h | — | 267 |
| 3D SG–TiO2 QDa | PFOA, | 4.8 mg/200 mL;0.30 mmol L−1; 200 mL | UV light, 150 W | 1.898 × 10−4/s | — | — | 272 |
| 3D SG–TiO2 QDb | PFOA | 4.8 mg/200 mL; 0.30 mmol L−1; 200 mL | UV light | 1.530 × 10−4/s | — | — | 272 |
| 3D SG–TiO2 NP | PFOA | 4.8 mg/200 mL; 0.30 mmol L−1; 200 mL | UV light | 9.283 × 10−5/s | — | — | 272 |
| MCQD/TiO2 | PFOA | 0.1 g L−1; 100 µg L−1; 100 mL | UV light, visible light | — | UV: degradation: 29%; visible: degradation: 20% | — | 273 |
| Exfol.CQD/TiO2 | PFOA | 0.1 g L−1; 100 µg L−1; 100 mL | UV light, visible light | — | UV: degradation: 69%, defluorination: 34.5%; visible: degradation: 50%; defluorination: 32.0% | 3 | 273 |
| Exfol.CQD/TiO2 | PFHxA, PFPeA, PFBA, PFPrA | 0.1 g L−1; 100 µg L−1; 100 mL | Visible light | — | PFHxA: degradation: 40%; PFPeA: degradation: 35%; PFBA: degradation: 31.6% PFPrA: degradation: 21.6% | — | 273 |
| PCQD/TiO2 | PFOA | 1 g L−1; 100 µg L−1; 100 mL | UV light, 4 × 16 W; visible light, 200 W | — | Defluorination: 56.2% (UV), 46.6% (visible) | 3 | 274 |
| PCQD/TiO2 | Short-chain PFCAs (C3–C6) | 1 g L−1; 100 µg L−1 each; 100 mL | UV light, 4 × 16 W; visible light, 200 W | — | Defluorination: 10.9% (visible), 12.9% (UV) | — | 274 |
| Emerging hybrid systems | |||||||
| MXene/TiO2 | PFOA | 20 mg; 20–100 mg L−1; 150 mL | UV light | 0.3296–0.1135 h−1 | Removal: 62.97–94.64%; defluorination: 58.4%, 9 h | — | 282 |
| Ti3C2/TiO2–80 | PFOA | 200 mg L−1; 20 µM | UV light, 20 mW cm−2 | — | Removal: >99.9%, F− 49%,16 h | 4 | 283 |
| Ti3C2-HF/TiO2 | PFOA | 200 mg L−1; 20 µM | UV light, 20 mW cm−2 | — | Removal: 22%, F− 12%,16 h | — | 283 |
| TiO2/zeolite | PFPA | 0.5-1.0 g L−1; 10 mg L−1; 1L | UV light, 16W | — | Removal: 54.5%, 8 h | — | 284 |
| F-TiO2@MIL-125 | PFOA, PFHpA, PFHxA, PFPeA | 4 mg; 300 μmol L−1 (PFOA); 200 mL | Visible light | 1.221 × 10−4/s | PFOA: rapid decrease in the first 2 h. | 7 | 66 |
| Photoelectrocatalysis (PEC) | |||||||
| GO/TNA | PFOA | 5 × 5 cm2; 0.121–2.42 mM; 1.4 L | UV light, 8 W; UV light + external bias (0–2 V) | 0.34 h−1 (25 °C, UV); 1.63 h−1 (75 °C, UV); | UV only: 83% in 4 h; PEC (2 V): 99.5% | 3 | 295 |
| TiO2 nanoporous (BEEP photoreactor) | PFTeDA (C14), PFNA (C9), PFOS, 42 PFAS mixture | TiO2 grown on inner Ti tubes; PFAS 100 ppm (single analyte)/42 PFAS mixture; 2 L solution | UVA 365 nm LED + 2 V bias (BEEP) | — | PFTeDA: below detection in 12 h; PFOS: ∼15% defluorination in 24 h; 42 PFAS: longer chains fully degraded in 24 h; smaller chains formed | — | 296 |
| TiO2 photoanode (plasma electrolytic oxidation on Ti mesh; anatase 58%, rutile 42%) | PFOA, PFHpA, PFHxA, PFPeA, PFBA, PFOS, PFHpS, PFHxS, PFBS | TiO2 mesh; PFOA 2 µg L−1 in ultrapure water; groundwater: 873–1604 ng L−1 total PFAS; reactor volume 1 L | UV (two lamp powers, P1 and P2); photoelectrocatalysis (PEC) | PFOA: 0.00461–0.00652 min−1 (PEC P1); PFHpA: 0.00242–0.00428 min−1; PFHxA: 0.00142–0.00309 min−1; PFPeA: 0.00179–0.00315 min−1; PFBA: 0.00265–0.00292 min−1; PFOS: 0.00261 min−1; PFHpS: 0.00134 min−1 | PFOA 96%, PFHpA 73%, PFHxA 75%, PFPeA 69%, PFBA 77%, PFOS 68%, PFHpS 40%, PFBS 1% @ 600 min | 2 | 297 |
| TiO 2 nanotube arrays (TNAs) with molecularly imprinted polymers (MIPs) | |||||||
| S-MIP–TiO2 NTs, NIP-TiO2 NTs, TiO2 NTs | PFBA, PFPeA, PFHxA, PFHpA, PFOA | Active area: 400 cm2 L−1; 10–109 ng L−1, Wastewater: 10 µg L−1; solution: 300 mL (adsorption) | UV light, 8 W | TiO2 NTs 0.0225, NIP-TiO2 NTs 0.0316, S-MIP–TiO2 NTs 0.0732 min−1 | S-MIP–TiO2 NTs > NIP-TiO2 NTs > TiO2 NTs | 5 | 317 |
| TiO2 NTs, NIP-TiO2 NTs, MIP–TiO2 NTs | PFOA, PFHA, PFOS, | —; 50 µmol L−1; competitive: PFOA 50 µmol L−1 + other PFASs 25 µmol L−1;— | UV light | TiO2 NTs 0.0011, NIP-TiO2 NTs 0.0022, MIP–TiO2 NTs 0.0036 min−1 | TiO2 NTs 41%, NIP-TiO2 NTs 67%, MIP–TiO2 NTs 84%; secondary effluent: UV 18%, NIP-TiO2 NTs 50.2%, MIP–TiO2 NTs 81.1%; F− release after 8 h: photolysis 5.4%, TiO2 NTs 9.7%, NIP-TiO2 NTs 15.7%, MIP–TiO2 NTs 30.2%; TOC removal by MIP–TiO2 NTs ∼46%;— | — | 316 |
| PLA/TiO2 composite tiles | PFOS, PFOA, PFHxA, PFBA, PFNA, PFDA, PFOSAm | 14.7 wt% TiO2; 850 cm2 chamber surface; PFOS 130 ng L−1, PFOA 1100–1500 ng L−1, PFHxA 2900 ng L−1, PFBA 2500 ng L−1, PFNA 29 ng L−1, PFDA 30 ng L−1, PFOSAm 5.7 ng L−1; PFUnDA/PFDOA near detection limit; — | UV light, ∼0.34 W m−2; cumulative 0.086–0.129 MJ m−2 | — | Mass removal rates: PFOS 2962–3072 ng m−2 day−1;degradation: PFOS 91–95%, PFNA 89–93%, PFDA 86–95%, PFOSAm 89–92%, PFOA 39–78%; short-chain PFASs increased due to longer-chain degradation; HRT 24 h vs. 36 h; better removal at 36 h | — | 318 |
| Hybrid advanced oxidation processes (AOPs) | |||||||
| TiO2/O3 | PFOA | 0.2 g L−1; 10 mg L−1; 1 L | UV light, 28 W | — | Using O3: 99.1% degradation, 44.3% C–F, 4 h | — | 304 |
| TiO2/PMS | PFOA | 0.25 g L−1; 50 mg L−1; 200 mL | 300 W, 400–770 nm | 0.310 h−1 | 100%, 8 h | — | 305 |
| TiO2/PMS | PFOA | 0.25 g L−1; 50 mg L−1; 200 mL | 32 W, 254 nm | 1.09 h−1 | 98%, 1.5 h | — | 305 |
A comprehensive understanding of TiO2-based PFAS photocatalysis necessitates the simultaneous evaluation of operational parameters, mechanistic pathways, and structural modifications (Fig. 17). These factors collectively determine degradation efficiency, selectivity, and stability. In this section, we critically assess the influence of key operational parameters, elucidate mechanistic insights into reactive species generation and C–F bond cleavage, and examine how structural and compositional modifications enhance charge separation and photocatalytic reactivity.
7.1. Influence of operational parameters
7.2. Mechanistic insights
A commonly proposed degradation pathway for PFOA begins with photoinduced oxidative decarboxylation, producing perfluoroalkyl radicals (˙CnF2n+1). These radicals then undergo reactions with O2 or H2O to form perfluoroalcohol intermediates, which subsequently yield shorter-chain PFCAs through β-scission and hydrolysis. The chain-shortening continues stepwise until CO2 and F− are released. This mechanism has been supported by LC-MS and ion chromatography analyses, revealing time-dependent decreases in PFOA and corresponding increases in fluoride ions.186,188,239 While oxidative routes dominate under oxygen-rich conditions, reductive pathways have also been recognized as critical for effective defluorination, especially under UV irradiation in the presence of organic hole scavengers (e.g., methanol or oxalic acid). The conduction band electrons in TiO2 can react with O2 to form O2˙− or directly attack the carbon–fluorine bonds via hydrated electrons (eaq−) or CO2˙− intermediates, enabling partial reduction of highly stable PFAS.188 This dual-pathway mechanism, where oxidative and reductive routes operate concurrently, explains why systems with balanced oxygen availability and sacrificial donors often exhibit superior degradation rates compared to purely oxidative setups.52Moreover, the crystal phase and exposed facets of TiO2 critically influence charge separation and ROS generation. Anatase TiO2 typically exhibits superior photocatalytic activity compared to rutile, owing to its higher surface area and lower e−–h+ recombination rates.37,52,223 Surface modifications, such as fluorination or oxygen vacancies (TiO2–x), further enhance electron trapping and extend carrier lifetimes, promoting ˙OH radical formation and effective C–F bond cleavage.235,274 Incorporation of carbon-based materials, including graphene oxide or MWCNTs, improves electron conductivity and facilitates π–π interactions with PFAS molecules, enhancing charge transfer and radical-mediated degradation.265
The reaction kinetics of PFAS degradation generally follow a pseudo-first-order model, highlighting the importance of electron transfer between TiO2 and adsorbed PFAS molecules. Degradation rates can be influenced by PFAS' chain length, surface adsorption, and operational conditions such as pH, dissolved oxygen, light intensity, temperature, and the presence of co-contaminants. Short-chain PFAS may degrade more slowly due to weaker adsorption, whereas long-chain PFAS, despite stronger initial adsorption, often undergo slower complete mineralization. These insights underscore the need to optimize both the interplay of surface adsorption and e−–h+ utilization and the operational parameters to achieve efficient defluorination and total organic carbon removal in TiO2-based PFAS remediation systems.
7.3. Structural and compositional modifications
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio combined adsorption with efficient electron storage to suppress recombination and drive stepwise CF2 cleavage.265,266,268 Exfoliated CQD/TiO2 composites demonstrated markedly superior PFOA degradation (69% under UVC, 50% under visible) compared to bare TiO2 (41%/24%), linking improved interparticle electron transfer and reduced aggregation to measurable gains in both conversion and defluorination stability.273 These results collectively show that conductive carbon frameworks are among the most practical routes to visible-light activity and suppressed recombination without relying solely on scarce noble metals.
1 ratio combined adsorption with efficient electron storage to suppress recombination and drive stepwise CF2 cleavage.265,266,268 Exfoliated CQD/TiO2 composites demonstrated markedly superior PFOA degradation (69% under UVC, 50% under visible) compared to bare TiO2 (41%/24%), linking improved interparticle electron transfer and reduced aggregation to measurable gains in both conversion and defluorination stability.273 These results collectively show that conductive carbon frameworks are among the most practical routes to visible-light activity and suppressed recombination without relying solely on scarce noble metals.
8. Challenges and knowledge gaps
Despite the remarkable progress summarized above, the practical application of TiO2-based photocatalytic systems for PFAS degradation remains constrained by a combination of scientific, engineering, and environmental challenges (Fig. 17). These challenges extend beyond catalytic activity alone and encompass incomplete mineralization, energy efficiency, reactor scalability, matrix effects, catalyst stability, and mechanistic uncertainty. Addressing these interconnected limitations is essential for translating laboratory-scale successes into viable treatment technologies.8.1. Incomplete mineralization and transformation limitations
The foremost limitation of TiO2-based PFAS photocatalysis is incomplete mineralization, even when high apparent degradation efficiencies are reported. In many systems, the disappearance of parent PFAS compounds does not correspond to full defluorination or conversion to CO2 and inorganic fluoride. Instead, short-chain perfluorinated intermediates, particularly C2–C5 perfluorocarboxylates, tend to accumulate, some of which are equally or more recalcitrant and mobile than their long-chain precursors.186,304 This behavior arises from the exceptionally high C–F bond dissociation energy and the reduced adsorption affinity of short-chain PFAS toward TiO2 surfaces. Consequently, extended irradiation times or secondary treatment stages are often required, underscoring that achieving selective and complete defluorination, rather than partial molecular transformation, remains a central mechanistic and catalyst-design challenge.8.2. Material stability and environmental safety
Material stability and environmental safety represent critical challenges for the practical deployment of modified TiO2 photocatalysts. Under prolonged irradiation, surface processes such as Ti–F bond accumulation can occur, leading to active-site passivation and suppression of further photocatalytic reactions. In parallel, leaching of dopant metals (e.g., Cu, Fe, Sb, and Pb) or the release of nanoscale catalyst fragments during operation poses risks of secondary contamination and long-term performance deterioration.239,283 Although several modified TiO2 systems exhibit acceptable recyclability over five to ten laboratory cycles with minimal activity loss, extended aging, fouling, mechanical integrity, and durability under realistic continuous-flow conditions remain largely uncharacterized. Beyond catalytic performance, comprehensive evaluation of toxicity and environmental compatibility is essential prior to large-scale implementation. Modified TiO2 systems must be assessed for the formation of potentially harmful transformation by-products, metal ion release, and long-term ecological impacts under realistic water treatment conditions. Life-cycle considerations, including material synthesis, operational stability, and end-of-life disposal, remain largely overlooked in current photocatalysis studies but are critical for ensuring sustainable deployment.Emerging composite materials further complicate stability and safety considerations. For example, MXene–TiO2 hybrids are susceptible to surface oxidation and structural degradation in aqueous and oxidative environments,319 particularly under prolonged illumination and in the presence of dissolved oxygen. Such degradation can reduce electrical conductivity, disrupt interfacial charge transfer, impair photocatalytic efficiency, and increase the likelihood of metal species release. Consequently, systematic long-term stability, leaching, toxicity, and environmental impact assessments under realistic operational conditions are required before modified TiO2 catalysts, especially advanced composite systems, can be considered viable for sustained PFAS remediation.
8.3. Water matrix effects and process complexity
Matrix effects represent a significant challenge for the real-world application of photocatalytic systems. Constituents commonly present in natural and wastewater matrices, such as bicarbonates, chlorides, natural organic matter (NOM), and competing inorganic anions, can substantially suppress photocatalytic activity by scavenging ROS or occupying active surface sites.320,321 Suspended solids, colored components, natural antioxidants, and mixed pollutants in real water matrices can further reduce photon penetration and pollutant-catalyst contact, exacerbating activity losses relative to idealized laboratory conditions. Beyond reducing degradation rates, these matrix components can alter reaction pathways and shift product distributions. Laboratory experiments conducted under idealized aqueous conditions often overestimate performance compared to complex environmental matrices. Dissolved oxygen (DO) is a critical chemical factor that interacts with water matrix effects. DO can enhance PFAS degradation in systems dominated by holes (h+) by suppressing electron–hole recombination and promoting hydroxyl radical formation. Conversely, in systems where electron-mediated or direct photolysis pathways are dominant, high DO can suppress degradation efficiency. Matrix components and DO together influence real-water photocatalytic performance, affecting both reaction rates and pathways. Therefore, standardized testing protocols that incorporate realistic water chemistry, including DO levels, are essential for meaningful cross-study comparisons and scalable reactor design.8.4. Energy efficiency and practical considerations
The energy required for PFAS photocatalytic degradation is a critical factor in evaluating the feasibility of treatment technologies.125 Photocatalytic systems relying on UV-C irradiation or high-intensity mercury lamps may exhibit high electrical energy per order (EE/O) values when photon utilization is inefficient, potentially limiting large-scale implementation unless reactor design and irradiance are carefully optimized.322 Despite its importance, EE/O is not reported consistently across studies, complicating direct techno-economic comparisons. Some studies have considered energy consumption metrics to evaluate and compare system performance. For example, BN/TiO2 composites have been reported to achieve lower EE/O values than bare TiO2, with lamp output-based EE/O values of ∼102 kWh m−3 order−1 under 254-nm irradiation and ∼253 kWh m−3 order−1 under 365-nm irradiation.37 A TiO2–Pb/rGO system achieved 98% PFOA removal with an EE/O of 4.05 kWh m−3.267 PEC generally requires lower energy than PL, with EEO ranges of 20–150 kWh m−3 (PEC) and 60–240 kWh m−3 (PL).297 In ultrapure water, PEC EEO values for PFOA (35.8–62.6 kWh m−3) are much lower than sonolysis and photochemical oxidation and comparable to plasma and advanced reduction processes. Higher energy demand in groundwater is due to matrix complexity and lower PFAS concentrations.8.5. Hybrid photocatalytic systems and operational complexity
Hybrid photocatalytic systems, including TiO2/PMS and TiO2/O3, have been widely investigated for the enhanced degradation of persistent pollutants by combining direct photocatalytic oxidation with radical-mediated pathways.323,324 Although these systems often outperform standalone photocatalysis under laboratory conditions, their practical implementation remains constrained by chemical complexity and operational limitations. A major challenge is the formation of undesirable transformation products. The generation of ROS (e.g., ˙OH and SO4˙−) can promote rapid pollutant degradation, but competing side reactions are common in real water matrices.325 In the presence of nitrate or nitrite, reactive nitrogen species may form and induce nitration reactions, leading to nitrogen-containing by-products that can be more persistent and toxic than the parent compounds. As a result, high removal efficiencies do not necessarily correspond to reduced environmental risk, highlighting the importance of transformation product analysis and toxicity evaluation alongside conventional performance metrics.Scalability is further limited by operational complexity. Hybrid systems depend on external oxidants such as persulfate, ozone, or hydrogen peroxide, which increase energy consumption, chemical usage, and process cost. Precise control of oxidant dosing and reaction conditions is required to avoid inefficiencies, secondary reactions, or residual oxidant release. Alternative hybrid configurations, such as photocatalysis combined with adsorption, membrane processes, or photoelectrocatalysis, can improve specific aspects of performance but introduce additional challenges related to reactor design, mass transfer, fouling, and material stability.326,327
8.6. Engineering challenges and scalability considerations
TiO2-based photocatalysis for PFAS degradation is still largely at the laboratory research stage, with most studies conducted under controlled ideal conditions. While these studies demonstrate the significant potential for photocatalysts, translating laboratory-scale findings into continuous-flow or field applications faces multiple engineering, operational, and scalability challenges.9. Conclusions and perspectives
TiO2-based photocatalysis has emerged as a versatile and promising strategy for PFAS remediation, offering sustainable pathways to break the highly resilient C–F bond. Advances in material modification, heterojunction engineering, and integration with conductive or carbonaceous supports have enhanced charge separation, light harvesting, and reactive species generation, leading to significant progress in PFAS degradation and establishing TiO2-based systems as a leading and well-studied photocatalytic platform for PFAS treatment. In summary, this review not only highlighted recent advances in TiO2-based photocatalysis for PFAS remediation but also critically assessed the influence of operational parameters, mechanistic pathways, structural and compositional modifications, hybrid and system-level strategies, and associated engineering and environmental challenges, providing a comprehensive overview of opportunities and remaining mechanistic gaps.Looking forward, future research should prioritize hybrid and system-level strategies to improve efficiency and scalability. Photoelectrocatalytic configurations under low external biases can enhance charge separation, improve defluorination, and reduce energy consumption; coupling TiO2 with conductive or tailored carbon materials such as graphene, carbon nitride, or MXenes can facilitate interfacial charge transport, lower overpotentials, and improve catalyst stability; and S-scheme heterojunctions help maintain strong redox potentials and suppress charge recombination. Integrated “concentrate-and-destroy” approaches, where TiO2-coated adsorbents capture PFAS from dilute streams followed by in situ photodegradation, may overcome mass-transfer limitations and minimize secondary waste. To bridge laboratory innovation with field-scale applications, future efforts must emphasize environmental compatibility, sustainability, and safety, with rigorous evaluation of catalyst toxicity, transformation byproducts, and life-cycle impacts, combined with durable, scalable reactor designs and energy-efficient operation. Coordinated advances across materials science, process engineering, and policy frameworks, guided by molecular design principles and data-driven discovery, can transform TiO2-based photocatalysis from a promising research platform into a field-ready solution for one of today's most persistent forever chemicals.
Author contributions
Avtar Singh: conceptualization, investigation, methodology, visualization, writing – original draft, writing – review and editing, supervision; Thiagarajan Soundappan: writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
The authors express their gratitude to Dr Yogesh Singla, Navajo Technical University (NTU), NM, USA, alongside their other collaborators and peers for their invaluable insights in shaping this manuscript. The authors also extend their appreciation to the publishers for granting permission to reproduce figures used in this publication.References
- I. T. Cousins, C. A. Ng, Z. Wang and M. Scheringer, Why Is High Persistence Alone a Major Cause of Concern?, Environ. Sci.: Processes Impacts, 2019, 21(5), 781–792, 10.1039/C8EM00515J.
- S. Jensen, A. Johnels, M. Olsson and G. Otterlind, DDT and PCB in Marine Animals from Swedish Waters, Nature, 1969, 224, 247–250, DOI:10.1038/224247a0.
- A. Kaushik, A. Singh, V. K. Gupta and Y. K. Mishra, Nano/Micro-Plastic, an Invisible Threat Getting into the Brain, Chemosphere, 2024, 361, 142380, DOI:10.1016/j.chemosphere.2024.142380.
- A. H. Butzlaff, J. Deighton, T. Le, K. Nguyen, C. Bonilla, Z. Chen, M. Kirshner, N. Shikuma, J. Peccia, K. McNeill and C. P. Higgins, PFAS, 6-PPD-Q, and Microplastics in Urban Sewer Overflows: Co-Occurrence and High-Rate Treatment Assessment, npj Emerg. Contam., 2025, 1, 5, DOI:10.1038/s44454-025-00010-4.
- E. L. Schymanski, J. Zhang, P. A. Thiessen, P. Chirsir, T. Kondic and E. E. Bolton, Per- and Polyfluoroalkyl Substances (PFAS) in PubChem: 7 Million and Growing, Environ. Sci. Technol., 2023, 57(44), 16918–16928, DOI:10.1021/acs.est.3c04855.
- A. K. Tokranov, Z. R. Hopkins, B. D. Lindsey and B. C. Jurgens, PFAS Are Widespread, Not Ubiquitous: Clarifying Misconceptions About the Prevalence of “Forever Chemicals”, Environ. Sci. Technol., 2025, 59(24), 11947–11949, DOI:10.1021/acs.est.5c03878.
- J. D. Boettger, N. M. DeLuca, M. A. Zurek-Ost, K. E. Miller, C. Fuller, K. D. Bradham, P. Ashley, W. Friedman, E. A. Pinzer, D. C. Cox, G. Dewalt, K. K. Isaacs, E. A. C. Hubal and J. P. McCord, Emerging Per- and Polyfluoroalkyl Substances in Tap Water from the American Healthy Homes Survey II, Environ. Sci. Technol., 2025, 59(5), 2686–2698, DOI:10.1021/acs.est.4c08037.
- K. M. Ríos-Bonilla, D. S. Aga, J. Lee, M. König, W. Qin, J. R. Cristobal, G. E. Atilla-Gokcumen and B. I. Escher, Neurotoxic Effects of Mixtures of Perfluoroalkyl Substances (PFAS) at Environmental and Human Blood Concentrations, Environ. Sci. Technol, 2024, 58(38), 16774–16784, DOI:10.1021/acs.est.4c06017.
- L. G. T. Gaines, Historical and Current Usage of Per- and Polyfluoroalkyl Substances (PFAS): A Literature Review, Am. J. Ind. Med., 2023, 66, 353–378, DOI:10.1002/ajim.23362.
- (a) D. O’Hagan, Understanding Organofluorine Chemistry: An Introduction to the C–F Bond, Chem. Soc. Rev., 2008,(37), 308–319, 10.1039/B711844A; (b) S. Jeon, E. Lee, T. Min, Y. Lee, G. Lee, A. Kim and H. Yoon, Efficient Removal of Short-Chain Perfluoroalkyl Substances (PFAS) Using Asymmetric Membrane Capacitive Deionization, Sep. Purif. Technol., 2024, 348, 127728, DOI:10.1016/j.seppur.2024.127728.
- The Report Socio-Economic Assessment of the US Fluoropolymer Industry, Executive Summary. Doc Ref. 41442-WOD-XX-XX-RP-OP-0002_A_C02, February 2020, 2020.
- G. Leter, C. Consales, P. Eleuteri, R. Uccelli, I. O. Specht, G. Toft, T. Moccia, A. Budillon, B. A. G. Jönsson, C. H. Lindh, A. Giwercman, H. S. Pedersen, J. K. Ludwicki, V. Zviezdai, D. Heederik, J. P. E. Bonde and M. Spanò, Exposure to Perfluoroalkyl Substances and Sperm DNA Global Methylation in Arctic and European Populations, Environ. Mol. Mutagen., 2014, 55, 591–600, DOI:10.1002/em.21874.
- S. E. Fenton, A. Ducatman, A. Boobis, J. C. DeWitt, C. Lau, C. Ng, J. S. Smith and S. M. Roberts, Per- and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research, Environ. Toxicol. Chem, 2021, 40(3), 606–630, DOI:10.1002/etc.4890.
- Y. He, X. Cheng, S. J. Gunjal and C. Zhang, Advancing PFAS Sorbent Design: Mechanisms, Challenges, and Perspectives, ACS Mater. Au, 2024, 4(2), 108–114, DOI:10.1021/acsmaterialsau.3c00066.
- P. S. Pauletto and T. J. Bandosz, Activated Carbon versus Metal–Organic Frameworks: A Review of Their PFAS Adsorption Performance, J. Hazard. Mater., 2022, 425, 127810, DOI:10.1016/j.jhazmat.2021.127810.
- E. Loukopoulos, S. Marugán-Benito, D. Raptis, E. Tylianakis, G. E. Froudakis, A. Mavrandonakis and A. E. Platero-Prats, Chemically Tailored Metal–Organic Frameworks for Enhanced Capture of Short- and Long-Chain Per- and Polyfluoroalkyl Substances from Water, Adv. Funct. Mater., 2024, 34, 2409932, DOI:10.1002/adfm.202409932.
- R.-R. Liang, S. Xu, Z. Han, Y. Yang, K.-Y. Wang, Z. Huang, J. Rushlow, P. Cai, P. Samorì and H.-C. Zhou, Exceptionally High Perfluorooctanoic Acid Uptake in Water by a Zirconium-Based Metal–Organic Framework through Synergistic Chemical and Physical Adsorption, J. Am. Chem. Soc., 2024, 146(14), 9811–9818, DOI:10.1021/jacs.3c14487.
- R. Mukhopadhyay, B. Sarkar, K. N. Palansooriya, J. Y. Dar, N. S. Bolan, S. J. Parikh, C. Sonne and Y. S. Ok, Natural and Engineered Clays and Clay Minerals for the Removal of Poly- and Perfluoroalkyl Substances from Water: State-of-the-Art and Future Perspectives, Adv. Colloid Interface Sci, 2021, 297, 102537, DOI:10.1016/j.cis.2021.102537.
- C. M. Luft, T. C. Schutt and M. K. Shukla, Properties and Mechanisms for PFAS Adsorption to Aqueous Clay and Humic Soil Components, Environ. Sci. Technol., 2022, 56(14), 10053–10061, DOI:10.1021/acs.est.2c00499.
- C. Gomri, B. T. Benkhaled, M. Cretin and M. Semsarilar, Adsorbent Material Used for the Treatment of Per- and Poly-Fluoroalkyl Substances (PFAS): A Short Review, Macromol. Chem. Phys., 2024, 225, 2400012, DOI:10.1002/macp.202400012.
- S. J. Smith, M. Lauria, L. Ahrens, P. McCleaf, P. Hollman, S. B. Seroka, T. Hamers, H. P. H. Arp and K. Wiberg, Electrochemical Oxidation for Treatment of PFAS in Contaminated Water and Fractionated Foam, A Pilot-Scale Study, ACS ES&T Water, 2023, 3(4), 1201–1211, DOI:10.1021/acsestwater.2c00660.
- Y. Bashir, R. Raj, M. M. Ghangrekar, A. K. Nema, S. Das, L. Zhang, C. Si, F. Zeng, X. Duan, D. Zhang, W. Xu and J. Shi, Critical Assessment of Advanced Oxidation Processes and Bio-Electrochemical Integrated Systems for Removing Emerging Contaminants from Wastewater, RSC Sustainability, 2023, 1, 1912–1931, 10.1039/D3SU00112A.
- L. Zhang, C. Si, F. Zeng, X. Duan, D. Zhang, W. Xu and J. Shi, Persulfate Activation for Efficient Remediation of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonic Acid (PFOS) in Water: Mechanisms, Removal Efficiency, and Future Prospects, J. Environ. Chem. Eng., 2024, 12(1), 111422, DOI:10.1016/j.jece.2023.111422.
- C.-S. Cao, J. Wang, L. Yang, J. Wang, Y. Zhang and L. Zhu, A Review on the Advancement in Photocatalytic Degradation of Poly/Perfluoroalkyl Substances in Water: Insights into the Mechanisms and Structure–Function Relationship, Sci. Total Environ., 2024, 946, 174137, DOI:10.1016/j.scitotenv.2024.174137.
- S. Rosansky, S. M. Al-Dirani, C. G. Scheitlin, K. Dasu, M. Dzurnak, X. Xia, C. Orth, M. McCauley and L. Mullins, Field Demonstration of PFAS Destruction in Various Alcohol-Resistant AFFFs Using Supercritical Water Oxidation (SCWO), ACS ES&T Water, 2024, 4(10), 4486–4496, DOI:10.1021/acsestwater.4c00499.
- J. T. McDonough, J. Kirby, C. Bellona, J. A. Quinnan, N. Welty, J. Follin and K. Liberty, Validation of Supercritical Water Oxidation to Destroy Perfluoroalkyl Acids, Remediation, 2022, 32, 75–90, DOI:10.1002/rem.21711.
- J. P. Skinner, A. Raderstorf, B. E. Rittmann and A. G. Delgado, Biotransforming the “Forever Chemicals”: Trends and Insights from Microbiological Studies on PFAS, Environ. Sci. Technol., 2025, 59(11), 5417–5430, DOI:10.1021/acs.est.4c04557.
- J. A. LaFond, P. B. Hatzinger, J. L. Guelfo, K. Millerick and W. A. Jackson, Bacterial Transformation of Per- and Poly-Fluoroalkyl Substances: A Review for the Field of Bioremediation, Environ. Sci.: Adv., 2023, 2, 1019–1041, 10.1039/D3VA00031A.
- L. P. Wackett, Nothing Lasts Forever: Understanding Microbial Biodegradation of Polyfluorinated Compounds and Perfluorinated Alkyl Substances, Microb. Biotechnol., 2022, 15(3), 773–792, DOI:10.1111/1751-7915.14018.
- C. Nau-Hix, N. Multari, R. K. Singh, S. Richardson, P. Kulkarni, R. H. Anderson, T. M. Holsen and S. M. Thagard, Field Demonstration of a Pilot-Scale Plasma Reactor for the Rapid Removal of Poly- and Perfluoroalkyl Substances in Groundwater, ACS ES&T Water, 2021, 1(3), 680–687, DOI:10.1021/acsestwater.0c00170.
- R. K. Singh, N. Multari, C. Nau-Hix, S. Woodard, M. Nickelsen, S. Mededovic Thagard and T. M. Holsen, Removal of Poly- and Per-Fluorinated Compounds from Ion Exchange Regenerant Still Bottom Samples in a Plasma Reactor, Environ. Sci. Technol., 2020, 54(21), 13973–13980, DOI:10.1021/acs.est.0c02158.
- N. Kim, J. Elbert and E. Shchukina, et al., Integrating Redox-Electrodialysis and Electrosorption for the Removal of Ultra-Short- to Long-Chain PFAS, Nat. Commun., 2024, 15, 8321, DOI:10.1038/s41467-024-52630-w.
- C. Liu, X. Zhao, A. F. Faria, K. Y. Deliz Quiñones, C. Zhang, Q. He, J. Ma, Y. Shen and Y. Zhi, Evaluating the Efficiency of Nanofiltration and Reverse Osmosis Membrane Processes for the Removal of Per- and Polyfluoroalkyl Substances from Water: A Critical Review, Sep. Purif. Technol., 2022, 302, 122161, DOI:10.1016/j.seppur.2022.122161.
- O. S. Awoyemi, R. Naidu and C. Fang, Advancements on Ultrasonic Degradation of Per- and Polyfluoroalkyl Substances (PFAS): Toward Hybrid Approaches, Environments, 2024, 11, 187, DOI:10.3390/environments11090187.
- X. Chen, T. Yuan, X. Yang, S. Ding and M. Ma, Insights into Photo/Electrocatalysts for the Degradation of Per- and Polyfluoroalkyl Substances (PFAS) by Advanced Oxidation Processes, Catalysts, 2023, 13(9), 1308, DOI:10.3390/catal13091308.
- T.-H. Kim, S.-H. Lee, H. Y. Kim, K. Doudrick, S. Yu and S. D. Kim, Decomposition of Perfluorooctane Sulfonate (PFOS) Using a Hybrid Process with Electron Beam and Chemical Oxidants, Chem. Eng. J., 2019, 361, 1363–1370, DOI:10.1016/j.cej.2018.10.195.
- L. Duan, B. Wang, K. N. Heck, C. A. Clark, J. Wei, M. Wang, J. Metz, G. Wu, A.-L. Tsai, S. Guo, J. Arredondo, A. D. Mohite, T. P. Senftle, P. Westerhoff, P. Alvarez, X. Wen, Y. Song and M. S. Wong, Titanium Oxide Improves Boron Nitride Photocatalytic Degradation of Perfluorooctanoic Acid, Chem. Eng. J., 2022, 448, 137735, DOI:10.1016/j.cej.2022.137735.
- J. P. Guin, J. A. Sullivan and K. R. Thampi, Challenges Facing Sustainable Visible Light Induced Degradation of Poly- and Perfluoroalkyls (PFA) in Water: A Critical Review, ACS Eng. Au, 2022, 2(3), 134–150, DOI:10.1021/acsengineeringau.1c00031.
- F. Liu, X. Guan and F. Xiao, Photodegradation of Per- and Polyfluoroalkyl Substances in Water: A Review of Fundamentals and Applications, J. Hazard. Mater., 2022, 439, 129580, DOI:10.1016/j.jhazmat.2022.129580.
- H. Zhang, J. X. Chen, J. P. Qu and Y. B. Kang, Photocatalytic Low-Temperature Defluorination of PFASs, Nature, 2024, 635(8039), 610–617, DOI:10.1038/s41586-024-08179-1.
- M. A. Hamza, A. J. Keltie, R. K. Matthews, M. L. Day and C. J. Shearer, CdIn2S4 Micro-Pyramids for Reductive Photocatalytic Degradation of Perfluorooctanesulfonic Acid, Small, 2025, e04601, DOI:10.1002/smll.202504601.
- R. Moreira, E. B. Esfahani and A. F. Zeidabadi, et al., Hybrid Graphenic and Iron Oxide Photocatalysts for the Decomposition of Synthetic Chemicals, Commun. Eng., 2024, 3, 114, DOI:10.1038/s44172-024-00267-4.
- K. Gates, S. Rai, A. Pramanik, O. P. Kolawole, S. Kundu, M. Ucak-Astarlioglu, M. K. Shukla, A. Al-Ostaz and P. C. Ray, Insight into the Photocatalytic Degradation Mechanism for “Forever Chemicals” PFNA by Reduced Graphene Oxide/WO3 Nanoflower Heterostructures, ACS Omega, 2025, 10(10), 10675–10684, DOI:10.1021/acsomega.5c00054.
- N. Chowdhury and H. Choi, Photocatalytic Degradation of Perfluorooctanoic Acid on Pb-Doped TiO2 Coated with Reduced Graphene Oxide, Water Environ. Res., 2023, 95(5), e10871, DOI:10.1002/wer.10871.
- K. E. Huang, C. W. Lai, J. C. Juan, Y. L. Pang, C. S. Khe, I. A. Badruddin, F. Gapsari and K. Anam, Recent Advances in Titanium Dioxide Bio-Derived Carbon Photocatalysts for Organic Pollutant Degradation in Wastewater, iScience, 2025, 28(5), 112368, DOI:10.1016/j.isci.2025.112368.
- C. B. Anucha, I. Altin, E. Bacaksiz and V. N. Stathopoulos, Titanium Dioxide (TiO2)-Based Photocatalyst Materials Activity Enhancement for Contaminants of Emerging Concern (CECs) Degradation: In the Light of Modification Strategies, Chem. Eng. J. Adv., 2022, 10, 100262, DOI:10.1016/j.ceja.2022.100262.
- R. Daghrir, P. Drogui and D. Robert, Modified TiO2 for Environmental Photocatalytic Applications: A Review, Ind. Eng. Chem. Res., 2013, 52(10), 3581–3599, DOI:10.1021/ie303468t.
- B. P. Narasaiah, P. Banoth, A. G. B. Dominguez, B. K. Mandal, C. K. Kumar, C. H. W. Barnes, L. D. L. S. Valladares and P. Kollu, Biogenic Photo-Catalyst TiO2 Nanoparticles for Remediation of Environment Pollutants, ACS Omega, 2022, 7(30), 26174–26189, DOI:10.1021/acsomega.2c01763.
- J. Arun, S. Nachiappan, G. Rangarajan, R. P. Alagappan, K. P. Gopinath and E. Lichtfouse, Synthesis and Application of Titanium Dioxide Photocatalysis for Energy, Decontamination and Viral Disinfection: A Review, Environ. Chem. Lett., 2023, 21(1), 339–362, DOI:10.1007/s10311-022-01503-z.
- A. Singh, J. Dhau, R. Kumar, R. Badru, P. Singh, Y. K. Mishra and A. Kaushik, Tailored Carbon Materials (TCM) for Enhancing Photocatalytic Degradation of Polyaromatic Hydrocarbons, Prog. Mater. Sci., 2024, 144, 101289, DOI:10.1016/j.pmatsci.2024.101289.
- L. Liu and X. Chen, Titanium Dioxide Nanomaterials: Self-Structural Modifications, Chem. Rev., 2014, 114(19), 9890–9918, DOI:10.1021/cr400624r.
- S. Weon, M.-J. Suh, C. Chu, D. Huang, E. Stavitski and J.-H. Kim, Site-Selective Loading of Single-Atom Pt on TiO2 for Photocatalytic Oxidation and Reductive Hydrodefluorination, ACS ES&T Eng., 2021, 1(3), 512–522, DOI:10.1021/acsestengg.0c00210.
- N. Roy, Y. Sohn and D. Pradhan, Synergy of Low-Energy {101} and High-Energy {001} TiO2 Crystal Facets for Enhanced Photocatalysis, ACS Nano, 2013, 7(3), 2532–2540, DOI:10.1021/nn400041p.
- M. Lazzeri, A. Vittadini and A. Selloni, Structure and Energetics of Stoichiometric TiO2 Anatase Surfaces, Phys. Rev. B, 2001, 63(15), 155409, DOI:10.1103/PhysRevB.63.155409.
- Z. Li, S. Wang, J. Wu and W. Zhou, Recent Progress in Defective TiO2 Photocatalysts for Energy and Environmental Applications, Renewable Sustainable Energy Rev., 2022, 156, 111980, DOI:10.1016/j.rser.2021.111980.
- T. Bak, W. Li, J. Nowotny, A. J. Atanacio and J. Davis, Photocatalytic Properties of TiO2: Evidence of the Key Role of Surface Active Sites in Water Oxidation, J. Phys. Chem. A, 2015, 119(36), 9465–9473, DOI:10.1021/acs.jpca.5b05031.
- X. Chen, L. Liu and F. Huang, Black Titanium Dioxide (TiO2) Nanomaterials, Chem. Soc. Rev., 2015, 44, 1861–1885, 10.1039/C4CS00330F.
- X. Qu, J. Lin, W. Qiang, C. Chen and D. Sun, Self-Doped Defect-Mediated TiO2 with Disordered Surface for High-Efficiency Photodegradation of Various Pollutants, Chemosphere, 2022, 308(Part 2), 136239, DOI:10.1016/j.chemosphere.2022.136239.
- D. Zhang, M. Yang and S. Dong, Hydroxylation of the Rutile TiO2(110) Surface Enhancing Its Reducing Power for Photocatalysis, J. Phys. Chem. C, 2015, 119(3), 1451–1456, DOI:10.1021/jp510427v.
- F. Teng, M. Li, C. Gao, G. Zhang, P. Zhang, Y. Wang, L. Chen and E. Xie, Preparation of Black TiO2 by Hydrogen Plasma Assisted Chemical Vapor Deposition and Its Photocatalytic Activity, Appl. Catal., B, 2014, 148–149, 339–343, DOI:10.1016/j.apcatb.2013.11.015.
- S. Adhikari, D. Sarkar and G. Madras, Hierarchical Design of CuS Architectures for Visible Light Photocatalysis of 4-Chlorophenol, ACS Omega, 2017, 2(7), 4009–4021, DOI:10.1021/acsomega.7b00669.
- W. Wang and S. M. Mukhopadhyay, Hierarchical Nanostructured Surface Design for Robust and Flexible Multifunctional Devices, Carbon Trends, 2021, 5, 100096, DOI:10.1016/j.cartre.2021.100096.
- J. Yi, G. Zhang, Y. Wang, W. Qian and X. Wang, Recent Advances in Phase-Engineered Photocatalysts: Classification and Diversified Applications, Materials, 2023, 16(11), 3980, DOI:10.3390/ma16113980.
- C. Qiu, J. Shen, J. Lin, D. Liu, D. Li, J. Zhang, Z. Zhang, H. Lin, X. Wang and X. Fu, Construction of the Rutile/Anatase Micro-Heterophase Junction Photocatalyst from Anatase by Liquid Nitrogen Quenching Method, ACS Appl. Energy Mater., 2021, 4(9), 10172–10186, DOI:10.1021/acsaem.1c02066.
- M. A. Ahmed, S. A. Mahmoud and A. A. Mohamed, Interfacially Engineered Metal Oxide Nanocomposites for Enhanced Photocatalytic Degradation of Pollutants and Energy Applications, RSC Adv, 2025, 15, 15561–15603, 10.1039/D4RA08780A.
- Z. Kong, L. Lu, C. Zhu, J. Xu, Q. Fang, R. Liu and Y. Shen, Enhanced Adsorption and Photocatalytic Removal of PFOA from Water by F-Functionalized MOF with In-Situ-Growth TiO2: Regulation of Electron Density and Bandgap, Sep. Purif. Technol., 2022, 297, 121449, DOI:10.1016/j.seppur.2022.121449.
- R. J. C. Fernandes, A. R. Silva, B. D. Cardoso, P. J. G. Coutinho and L. Pereira, Potential of Photocatalytic Nanomaterials for PFOA and PFOS Degradation: Challenges and Opportunities, J. Environ. Chem. Eng., 2025, 13(1), 115201, DOI:10.1016/j.jece.2024.115201.
- S. S. Thind, B. A. Ryane, J. B. Hayden, I. Chagunda, M. Paul and J. S. McIndoe, Bias Enhanced Electro-Photocatalysis on TiO2 Nanoporous Materials for Decomposition of Forever Chemicals in Saltwater, Environ. Sci.: Adv., 2025, 4, 1024–1034, 10.1039/D4VA00423J.
- S. Verma, B. Mezgebe, C. A. Hejase, E. Sahle-Demessie and M. N. Nadagouda, Photodegradation and Photocatalysis of Per- and Polyfluoroalkyl Substances (PFAS): A Review of Recent Progress, Next Mater., 2024, 2, 100077, DOI:10.1016/j.nxmate.2023.100077.
- A. Veciana, S. Steiner, Q. Tang, V. Pustovalov, J. Llacer-Wintle, J. Wu, X.-Z. Chen, T. Manyiwa, V. U. Ultra Jr, B. Garcia-Cirera, J. Puigmartí-Luis, C. Franco, D. J. Janssen, L. Nyström, S. Boulos and S. Pané, Breaking the Perfluorooctane Sulfonate Chain: Piezocatalytic Decomposition of PFOS Using BaTiO3 Nanoparticles, Small Sci., 2024, 4, 2400337, DOI:10.1002/smsc.202400337.
- S. Ebnesajjad, in Discovery and History of Fluoropolymers, Introduction to Fluoropolymers, ed S. Ebnesajjad, William Andrew Publishing, Boston, 2013, pp. 17–35 DOI:10.1016/B978-1-4557-7442-5.00003-6.
- The History of Fluoropolymers, APS News Archives, American Physical Society, April 2021, https://www.aps.org/archives/publications/apsnews/202104/history.cfm (accessed Aug 12, 2025) Search PubMed.
- Z. Habib, M. Song, S. Ikram and Z. Zahra, Overview of Per- and Polyfluoroalkyl Substances (PFAS), Their Applications, Sources, and Potential Impacts on Human Health, Pollutants, 2024, 4(1), 136–152, DOI:10.3390/pollutants4010009.
- J. Glüge, M. Scheringer, I. T. Cousins, J. C. DeWitt, G. Goldenman, D. Herzke, R. Lohmann, C. A. Ng, X. Trier and Z. Wang, An Overview of the Uses of Per- and Polyfluoroalkyl Substances (PFAS), Environ. Sci.: Processes Impacts, 2020, 22, 2345–2373, 10.1039/D0EM00291G.
- C. Gao, D. S. Drage, M. A.-E. Abdallah, F. Quan, K. Zhang, S. Hu, X. Zhao, Y. Zheng, S. Harrad and W. Qiu, Factors Influencing Concentrations of PFAS in Drinking Water: Implications for Human Exposure, ACS ES&T Water, 2024, 4(11), 4881–4892, DOI:10.1021/acsestwater.4c00533.
- A. F. Peritore, E. Gugliandolo, S. Cuzzocrea, R. Crupi and D. Britti, Current Review of Increasing Animal Health Threat of Per- and Polyfluoroalkyl Substances (PFAS): Harms, Limitations, and Alternatives to Manage Their Toxicity, Int. J. Mol. Sci, 2023, 24(14), 11707, DOI:10.3390/ijms241411707.
- S. Y. Wee and A. Z. Aris, Environmental Impacts, Exposure Pathways, and Health Effects of PFOA and PFOS, Ecotoxicol. Environ. Saf., 2023, 267, 115663, DOI:10.1016/j.ecoenv.2023.115663.
- N. M. DeLuca, J. M. Minucci, A. Mullikin, R. Slover and E. A. Cohen Hubal, Human Exposure Pathways to Poly- and Perfluoroalkyl Substances (PFAS) from Indoor Media: A Systematic Review, Environ. Int., 2022, 162, 107149, DOI:10.1016/j.envint.2022.107149.
- H. Mahoney, Y. Xie, M. Brinkmann and J. P. Giesy, Next Generation Per- and Polyfluoroalkyl Substances: Status and Trends, Aquatic Toxicity, and Risk Assessment, Eco Environ. Health, 2022, 1(2), 117–131, DOI:10.1016/j.eehl.2022.05.002.
- K. I. Kirkwood-Donelson, J. M. Minucci, A. Mullikin, R. Slover and E. A. Cohen Hubal, Uncovering Per- and Polyfluoroalkyl Substances (PFAS) with Nontargeted Ion Mobility Spectrometry–Mass Spectrometry Analyses, Sci. Adv., 2023, 9, eadj7048, DOI:10.1126/sciadv.adj7048.
- E. J. Itumoh, S. Data, J. L.-Y. Chen, M. Kah, L. P. Padhye and E. M. Leitao, Addressing the Persistence of Per- and Polyfluoroalkyl Substances (PFAS): Current Challenges and Potential Solutions, RSC Sustainability, 2024, 2, 3183–3201, 10.1039/D4SU00152D.
- K. A. Barzen-Hanson, S. C. Roberts, S. Choyke, K. Oetjen, A. McAlees, N. Riddell, R. McCrindle, P. L. Ferguson, C. P. Higgins and J. A. Field, Discovery of 40 Classes of Per- and Polyfluoroalkyl Substances in Historical Aqueous Film-Forming Foams (AFFFs) and AFFF-Impacted Groundwater, Environ. Sci. Technol., 2017, 51(4), 2047–2057, DOI:10.1021/acs.est.6b05843.
- W. S. Chambers, J. G. Hopkins and S. M. Richards, A Review of Per- and Polyfluorinated Alkyl Substance Impairment of Reproduction, Front. Toxicol., 2021, 3, 732436, DOI:10.3389/ftox.2021.732436.
- M. G. Evich, M. J. B. Davis, J. P. McCord, B. Acrey, J. A. Awkerman, D. R. U. Knappe, A. B. Lindstrom, T. F. Speth, C. Tebes-Stevens, M. J. Strynar, Z. Wang, E. J. Weber, W. M. Henderson and J. W. Washington, Per- and Polyfluoroalkyl Substances in the Environment, Science, 2022, 375(6580), eabg9065, DOI:10.1126/science.abg9065.
- A. Su, Y. Cheng, C. Zhang, Y.-F. Yang, Y.-B. She and K. Rajan, An Artificial Intelligence Platform for Automated PFAS Subgroup Classification: A Discovery Tool for PFAS Screening, Sci. Total Environ., 2024, 921, 171229, DOI:10.1016/j.scitotenv.2024.171229.
- S. Yaghoobian, M. A. Ramirez-Ubillus, L. Zhai and J. H. Hwang, A Perspective of Emerging Trends in Integrated PFAS Detection and Remediation Technologies with Data Driven Approaches, Chem. Sci., 2025, 16(30), 13564–13573, 10.1039/d5sc01624j.
- U.S. Environmental Protection Agency. PFAS Analytical Methods Development and Sampling Research. https://www.epa.gov/water-research/pfas-analytical-methods-development-and-sampling-research (accessed Aug 12, 2025).
- M. Jovanović, V. Müller, J. Feldmann and E. Leitner, Analysis of Per- and Polyfluoroalkyl Substances (PFAS) in Raw Materials Intended for the Production of Paper-Based Food Contact Materials,Evaluating LC-MS/MS versus Total Fluorine and Extractable Organic Fluorine, Food Addit. Contam., Part A, 2024, 41(5), 525–536, DOI:10.1080/19440049.2024.2332334.
- R. Kause, S. van Leeuwen, K. Krätschmer, B. van Dooren, R. Keppels, H. Makarem, L. R. A. P. Hoogenboom, L. de Pagter-de Witte and B. J. A. Berendsen, Development and Application of a Liquid Chromatography–Tandem Mass Spectrometry Method for the Analysis of 20 Perfluoroalkyl Substances in Fruit and Vegetables at Sub-Parts-per-Trillion Levels, J. Agric. Food Chem., 2024, 72(33), 18731–18741, DOI:10.1021/acs.jafc.4c01172.
- J. Garnett, C. Halsall, H. Winton, H. Joerss, R. Mulvaney, R. Ebinghaus, M. Frey, A. Jones, A. Leeson and P. Wynn, Increasing Accumulation of Perfluorocarboxylate Contaminants Revealed in an Antarctic Firn Core (1958–2017), Environ. Sci. Technol., 2022, 56(16), 11246–11255, DOI:10.1021/acs.est.2c02592.
- L. Zhu, R. Bossi, P. N. Carvalho, F. F. Rigét, J. H. Christensen, P. Weihe, E. C. Bonefeld-Jørgensen and K. Vorkamp, Suspect and Non-Target Screening of Chemicals of Emerging Arctic Concern in Biota, Air and Human Serum, Environ. Pollut., 2024, 360, 124605, DOI:10.1016/j.envpol.2024.124605.
- R. Kause, S. van Leeuwen, K. Krätschmer, B. van Dooren, R. Keppels, H. Makarem, L. R. A. P. Hoogenboom, L. de Pagter-de Witte and B. J. A. Berendsen, Development and Application of a Liquid Chromatography–Tandem Mass Spectrometry Method for the Analysis of 20 Perfluoroalkyl Substances in Fruit and Vegetables at Sub-Parts-per-Trillion Levels, J. Agric. Food Chem., 2024, 72(33), 18731–18741, DOI:10.1021/acs.jafc.4c01172.
- Y. Lai, J. P. Koelmel, D. I. Walker, E. J. Price, S. Papazian, K. E. Manz, D. Castilla-Fernández, J. A. Bowden, V. Nikiforov, A. David, V. Bessonneau, B. Amer, S. Seethapathy, X. Hu, E. Z. Lin, A. Jbebli, B. R. McNeil, D. Barupal, M. Cerasa, H. Xie, V. Kalia, R. Nandakumar, R. Singh, Z. Tian, P. Gao, Y. Zhao, J. Froment, P. Rostkowski, S. Dubey, K. Coufalíková, H. Seličová, H. Hecht, S. Liu, H. H. Udhani, S. Restituito, K.-M. Tchou-Wong, K. Lu, J. W. Martin, B. Warth, K. J. Godri Pollitt, J. Klánová, O. Fiehn, T. O. Metz, K. D. Pennell, D. P. Jones and G. W. Miller, High-Resolution Mass Spectrometry for Human Exposomics: Expanding Chemical Space Coverage, Environ. Sci. Technol., 2024, 58(29), 12784–12822, DOI:10.1021/acs.est.4c01156.
- Z. Zahra, M. Song, Z. Habib and S. Ikram, Advances in Per- and Polyfluoroalkyl Substances (PFAS) Detection and Removal Techniques from Drinking Water, Their Limitations, and Future Outlooks, Emerg. Contam., 2025, 11(1), 100434, DOI:10.1016/j.emcon.2024.100434.
- S. Poothong, E. Papadopoulou, J. A. Padilla-Sánchez, C. Thomsen and L. S. Haug, Multiple Pathways of Human Exposure to Poly- and Perfluoroalkyl Substances (PFASs): From External Exposure to Human Blood, Environ. Int., 2020, 134, 105244, DOI:10.1016/j.envint.2019.105244.
- N. M. DeLuca, J. M. Minucci, A. Mullikin, R. Slover and E. A. Cohen Hubal, Human Exposure Pathways to Poly- and Perfluoroalkyl Substances (PFAS) from Indoor Media: A Systematic Review, Environ. Int., 2022, 162, 107149, DOI:10.1016/j.envint.2022.107149.
- M. H. Dehghani, M. Aghaei and P. Bashardoust, et al., An Insight into the Environmental and Human Health Impacts of Per- and Polyfluoroalkyl Substances (PFAS): Exploring Exposure Pathways and Their Implications, Environ. Sci. Eur., 2025, 37, 81, DOI:10.1186/s12302-025-01122-9.
- K. Steenland, T. Fletcher, C. R. Stein, S. M. Bartell, L. Darrow, M.-J. Lopez-Espinosa, P. B. Ryan and D. A. Savitz, Review: Evolution of Evidence on PFOA and Health Following the Assessments of the C8 Science Panel, Environ. Int., 2020, 145, 106125, DOI:10.1016/j.envint.2020.106125.
- C. Boston, S. Keck, A. Naperala and J. Collins, The Evolution of PFAS Epidemiology: New Scientific Developments Call into Question Alleged “Probable Links” between PFOA and Kidney Cancer and Thyroid Disease, Front. Public Health, 2025, 13, 1532277, DOI:10.3389/fpubh.2025.1532277.
- J. T. Szilagyi, V. Avula and R. C. Fry, Perfluoroalkyl Substances (PFAS) and Their Effects on the Placenta, Pregnancy, and Child Development: A Potential Mechanistic Role for Placental Peroxisome Proliferator-Activated Receptors (PPARs), Curr. Environ. Health Rep, 2020, 7(3), 222–230, DOI:10.1007/s40572-020-00279-0.
- T. C. Ho, H. T. Wan, W. K. Lee, T. K. Y. Lam, X. Lin, T. F. Chan, K. P. Lai and C. K. C. Wong, Effects of In Utero PFOS Exposure on Epigenetics and Metabolism in Mouse Fetal Livers, Environ. Sci. Technol., 2023, 57(40), 14892–14903, DOI:10.1021/acs.est.3c05207.
- T. T. Lai, Y. Eken and A. K. Wilson, Binding of Per- and Polyfluoroalkyl Substances to the Human Pregnane X Receptor, Environ. Sci. Technol., 2020, 54(24), 15986–15995, DOI:10.1021/acs.est.0c04651.
- C. Holder, E. A. Cohen Hubal, J. Luh, M. G. Lee, L. J. Melnyk and K. Thomas, Systematic Evidence Mapping of Potential Correlates of Exposure for Per- and Poly-Fluoroalkyl Substances (PFAS) Based on Measured Occurrence in Biomatrices and Surveys of Dietary Consumption and Product Use, Int. J. Hyg. Environ. Health, 2024, 259, 114384, DOI:10.1016/j.ijheh.2024.114384.
- L. Dunder, S. Salihovic and S. Elmståhl, et al., Associations between Per- and Polyfluoroalkyl Substances (PFAS) and Diabetes in Two Population-Based Cohort Studies from Sweden, J. Expo. Sci. Environ. Epidemiol., 2023, 33, 748–756, DOI:10.1038/s41370-023-00529-x.
- F. Dunn, S. E. Paquette, K. D. Pennell, J. S. Plavicki and K. E. Manz, Metabolomic Changes Following GenX and PFBS Exposure in Developing Zebrafish, Aquat. Toxicol., 2024, 271, 106908, DOI:10.1016/j.aquatox.2024.106908.
- G. M. Sinclair, S. M. Long, N. Singh, T. L. Coggan, M. P. J. Askeland and O. A. H. Jones, Exposure to Environmentally Relevant Levels of PFAS Causes Metabolic Changes in the Freshwater Amphipod Austrochiltonia subtenuis, Metabolites, 2022, 12, 1135, DOI:10.3390/metabo12111135.
- E. Ortiz-Villanueva, J. Jaumot, R. Martínez, L. Navarro-Martín, B. Piña and R. Tauler, Assessment of Endocrine Disruptors Effects on Zebrafish (Danio rerio) Embryos by Untargeted LC-HRMS Metabolomic Analysis, Sci. Total Environ., 2018, 635, 156–166, DOI:10.1016/j.scitotenv.2018.03.369.
- J. Ning, C. Ding, H. Xu, Z. Liu, Q. Guan, Y. Xia and Q. Xu, Effect of Per- and Polyfluoroalkyl Substances on Neurodevelopment: Evidence-Based Risk Assessment in the TRAEC Strategy Context, Environ. Int., 2024, 191, 109003, DOI:10.1016/j.envint.2024.109003.
- J. M. Brown-Leung and J. R. Cannon, Neurotransmission Targets of Per- and Polyfluoroalkyl Substance Neurotoxicity: Mechanisms and Potential Implications for Adverse Neurological Outcomes, Chem. Res. Toxicol., 2022, 35(8), 1312–1333, DOI:10.1021/acs.chemrestox.2c00072.
- X. C. Hu, D. Q. Andrews, A. B. Lindstrom, T. A. Bruton, L. A. Schaider, P. Grandjean, R. Lohmann, C. C. Carignan, A. Blum, S. A. Balan, C. P. Higgins and E. M. Sunderland, Detection of Poly- and Perfluoroalkyl Substances (PFASs) in U.S. Drinking Water Linked to Industrial Sites, Military Fire Training Areas, and Wastewater Treatment Plants, Environ. Sci. Technol. Lett., 2016, 3(10), 344–350, DOI:10.1021/acs.estlett.6b00260.
- A. Gezahegn, L. B. Merga and S. Mammo, Industrial Effluents Caused Environmental Pollution and Its Potential Ecological and Human Health Impacts in Ethiopia: A Review, Waste Manage. Bull., 2025, 100240, DOI:10.1016/j.wmb.2025.100240.
- F. Wang, L. Xiang, K. S.-Y. Leung, M. Elsner, Y. Zhang, Y. Guo, B. Pan, H. Sun, T. An, G. Ying, B. W. Brooks, D. Hou, D. E. Helbling, J. Sun, H. Qiu, T. M. Vogel, W. Zhang, Y. Gao, M. J. Simpson, Y. Luo, S. X. Chang, G. Su, B. M. Wong, T.-M. Fu, D. Jobst, K. J. Ge, C. Coulon, F. Harindintwali, J. D. Zeng, X. Wang, H. Fu, Y. Wei, Z. Lohmann, R. Chen, C. Song, Y. Sanchez-Cid, C. Wang, Y. El-Naggar, A. Yao, Y. Huang, Y. Law, J. C.-F. Gu, C. Shen, H. Gao, Y. Qin, C. Li, H. Zhang, T. Corcoll, N. Liu, M. Alessi, D. S. Li, H. Brandt, K. K. Pico, Y. Gu, C. Guo, J. Su, J. Corvini, P. Ye, M. Rocha-Santos, T. He, H. Yang, Y. Tong, M. Zhang, W. Suanon, F. Brahushi, F. Wang, Z. Hashsham, S. A. Virta, M. Yuan, Q. Jiang, G. Tremblay, L. A. Bu, Q. Wu, J. Peijnenburg, W. Topp, E. Cao, X. Jiang, X. Zheng, M. Zhang, T. Luo, Y. Zhu, L. Li, X. Barceló, D. Chen, J. Xing, B. Amelung, W. Cai, Z. Naidu, R. Shen, Q. Zhu, Y.-G. Schaeffer, A. Rillig, M. C. Wu, F. Yu and G. Tiedje, Emerging Contaminants: A One Health Perspective, Innovation, 2024, 5(4), 100612, DOI:10.1016/j.xinn.2024.100612.
- J. E. Zenobio, B. N. Nzeribe, M. Hilyard and B. DiGuiseppi, Deciphering PFAS in Rainwater: Sources, Distribution, and Environmental Impact, Remediat. J., 2025, 35, e70029, DOI:10.1002/rem.70029.
- W.-L. Li, H. Hung, A. Kasperkiewicz, C. Shunthirasingham, J. Chisamore, M. Rabu, R. Park, C.-Y. Huo, P. Lee and H. Dryfhout-Clark, Assessing the Shifts in Atmospheric Per- and Polyfluoroalkyl Substances (PFAS) Levels in the Great Lakes and Implications for the Environmental Transport and Fate, ACS ES&T Air ASAP, 2025, 2, 1927–1938, DOI:10.1021/acsestair.5c00121.
- L. Zhang, Y. Zhang, Q. Gong, Z. Yang and D. Sun, Uncovering the Effects of the North Pacific Subtropical Gyre on Per- and Polyfluoroalkyl Substances Distribution in the Tropical Western Pacific, J. Hazard. Mater., 2025, 494, 138631, DOI:10.1016/j.jhazmat.2025.138631.
- R. Lohmann, K. Abass, E. C. Bonefeld-Jørgensen, R. Bossi, R. Dietz, S. Ferguson, K. J. Fernie, P. Grandjean, D. Herzke, M. Houde, M. Lemire, R. J. Letcher, D. Muir, A. O. De Silva, S. K. Ostertag, A. A. Rand, J. Søndergaard, C. Sonne, E. M. Sunderland, K. Vorkamp, S. Wilson and P. Weihe, Cross-Cutting Studies of Per- and Polyfluorinated Alkyl Substances (PFAS) in Arctic Wildlife and Humans, Sci. Total Environ., 2024, 954, 176274, DOI:10.1016/j.scitotenv.2024.176274.
- P.-F. Yan, S. Dong, K. D. Pennell and N. L. Cápiro, A Review of the Occurrence and Microbial Transformation of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Film-Forming Foam (AFFF)-Impacted Environments, Sci. Total Environ., 2024, 927, 171883, DOI:10.1016/j.scitotenv.2024.171883.
- B. J. Ruyle, C. P. Thackray, C. M. Butt, D. R. LeBlanc, A. K. Tokranov, C. D. Vecitis and E. M. Sunderland, Centurial Persistence of Forever Chemicals at Military Fire Training Sites, Environ. Sci. Technol., 2023, 57(21), 8096–8106, DOI:10.1021/acs.est.3c00675.
- D. Salvatore, K. Mok, K. K. Garrett, G. Poudrier, P. Brown, L. S. Birnbaum, G. Goldenman, M. F. Miller, S. Patton, M. Poehlein, J. Varshavsky and A. Cordner, Presumptive Contamination: A New Approach to PFAS Contamination Based on Likely Sources, Environ. Sci. Technol. Lett., 2022, 9(11), 983–990, DOI:10.1021/acs.estlett.2c00502.
- M. Filipovic, A. Woldegiorgis, K. Norström, M. Bibi, M. Lindberg and A.-H. Österås, Historical Usage of Aqueous Film Forming Foam: A Case Study of the Widespread Distribution of Perfluoroalkyl Acids from a Military Airport to Groundwater, Lakes, Soils and Fish, Chemosphere, 2015, 129, 39–45, DOI:10.1016/j.chemosphere.2014.09.005.
- M. Ateia, J. V. Buren, W. Barrett, T. Martin and G. G. Back, Sunrise of PFAS Replacements: A Perspective on Fluorine-Free Foams, ACS Sustainable Chem. Eng., 2023, 11, 7986–7996, DOI:10.1021/acssuschemeng.3c01124.
- T. Paige, T. De Silva, S. Buddhadasa, S. Prasad, D. Nugegoda and V. Pettigrove, Background Concentrations and Spatial Distribution of PFAS in Surface Waters and Sediments of the Greater Melbourne Area, Australia, Chemosphere, 2024, 349, 140791, DOI:10.1016/j.chemosphere.2023.140791.
- EPA NSW. Regulation of PFAS Firefighting Foams. https://www.epa.nsw.gov.au/Your-environment/Chemicals/PFAS-in-NSW/Regulation-of-PFAS-firefighting-foams Search PubMed.
- ECHA. ECHA Publishes Updated PFAS Restriction Proposal. https://echa.europa.eu/-/echa-publishes-updated-pfas-restriction-proposal Search PubMed.
- C. S. Tshangana, S. T. Nhlengethwa and S. Glass, et al., Technology Status to Treat PFAS-Contaminated Water and Limiting Factors for Their Effective Full-Scale Application, npj Clean Water, 2025, 8, 41, DOI:10.1038/s41545-025-00457-3.
- S. J. Smith, M. Lauria, L. Ahrens, P. McCleaf, P. Hollman, S. Bjälkefur Seroka, T. Hamers, H. P. Arp and K. Wiberg, Electrochemical Oxidation for Treatment of PFAS in Contaminated Water and Fractionated Foam-A Pilot-Scale Study, ACS ES&T Water, 2023, 3(4), 1201–1211, DOI:10.1021/acsestwater.2c00660.
- H. N. Hussain, M. I. Jilani, F. Imtiaz, T. Ahmed, M. B. Arshad, M. Mudassar and M. N. Sharif, Advances in the Removal of Polyfluoroalkyl Substances (PFAS) from Water Using Destructive and Non-Destructive Methods, Green Anal. Chem., 2025, 12, 100225, DOI:10.1016/j.greeac.2025.100225.
- Z. Ren, U. Bergmann and T. Leiviskä, Reductive Degradation of Perfluorooctanoic Acid in Complex Water Matrices Using the UV/Sulfite Process, Water Res, 2021, 205, 117676 Search PubMed.
- Y.-G. Kang, Q. T. Birch, M. N. Nadagouda and D. D. Dionysiou, Advanced destruction technologies for PFAS in soils: Progress and challenges. Curr. Opin. Environ, Sci. Health, 2023, 33, 100459, DOI:10.1016/j.coesh.2023.100459.
- C. Grimison, E. R. Knight, T. M. H. Nguyen, N. Nagle, S. Kabiri, J. Bräunig, D. A. Navarro, R. S. Kookana, C. P. Higgins, M. J. McLaughlin and J. F. Mueller, The efficacy of soil washing for the remediation of per- and poly-fluoroalkyl substances (PFASs) in the field, J. Hazard. Mater., 2023, 445, 130441, DOI:10.1016/j.jhazmat.2022.130441.
- R. Napoli, F. Fazzino, F. G. A. Vagliasindi and P. P. Falciglia, Sustainable remediation strategies and technologies of per- and polyfluoroalkyl substances (PFAS)-contaminated soils: A critical review, Sustainability, 2025, 17(14), 6635, DOI:10.3390/su17146635.
- A. Alinezhad, P. C. Sasi, P. Zhang, B. Yao, A. Kubátová, S. A. Golovko, M. Y. Golovko and F. Xiao, An investigation of thermal air degradation and pyrolysis of per- and polyfluoroalkyl substances and aqueous film-forming foams in soil, ACS ES&T Eng., 2022, 2(2), 198–209, DOI:10.1021/acsestengg.1c00335.
- J. Wang, Z. Lin, X. He, M. Song, P. Westerhoff, K. Doudrick and D. Hanigan, Critical review of thermal decomposition of per- and polyfluoroalkyl substances: Mechanisms and implications for thermal treatment processes, Environ. Sci. Technol., 2022, 56(9), 5355–5370, DOI:10.1021/acs.est.2c02251.
- R. Mahinroosta and L. Senevirathna, A review of the emerging treatment technologies for PFAS contaminated soils, J. Environ. Manage., 2020, 255, 109896, DOI:10.1016/j.jenvman.2019.109896.
- F. Wang, X. Lu, X.-Y. Li and K. Shih, Effectiveness and mechanisms of defluorination of perfluorinated alkyl substances by calcium compounds during waste thermal treatment, Environ. Sci. Technol., 2015, 49(9), 5672–5680, DOI:10.1021/es506234b.
- A. L. Duchesne, J. K. Brown, D. J. Patch, D. Major, K. P. Weber and J. I. Gerhard, Remediation of PFAS-contaminated soil and granular activated carbon by smoldering combustion, Environ. Sci. Technol., 2020, 54(19), 12631–12640, DOI:10.1021/acs.est.0c03058.
- S. Hao, Y. J. Choi, R. A. Deeb, T. J. Strathmann and C. P. Higgins, Application of hydrothermal alkaline treatment for destruction of per- and polyfluoroalkyl substances in contaminated groundwater and soil, Environ. Sci. Technol., 2022, 56(10), 6647–6657, DOI:10.1021/acs.est.2c00654.
- L. P. Turner, B. H. Kueper, K. M. Jaansalu, D. J. Patch, N. Battye, O. El-Sharnouby, K. G. Mumford and K. P. Weber, Mechanochemical remediation of perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) amended sand and aqueous film-forming foam (AFFF) impacted soil by planetary ball milling, Sci. Total Environ., 2021, 765, 142722, DOI:10.1016/j.scitotenv.2020.142722.
- N. J. Battye, D. J. Patch, D. M. D. Roberts, N. M. O’Connor, L. P. Turner, B. H. Kueper, M. E. Hulley and K. P. Weber, Use of a horizontal ball mill to remediate per- and polyfluoroalkyl substances in soil, Sci. Total Environ., 2022, 835, 155506, DOI:10.1016/j.scitotenv.2022.155506.
- Y. Yan, Z. Wei, X. Duan, M. Long, R. Spinney, D. D. Dionysiou, R. Xiao and P. J. J. Alvarez, Merits and limitations of radical vs. nonradical pathways in persulfate-based advanced oxidation processes, Environ. Sci. Technol., 2023, 57(33), 12153–12179, DOI:10.1021/acs.est.3c05153.
- W. H. DiGuiseppi, C. J. Newell, G. Carey, P. R. Kulkarni, Z. Xia, J. Stults, T. L. Maher, E. F. Houtz, R. Mora, R. Wice, P. W. Tomiczek, S. D. Richardson, J. Xiong, J. Hale, J. P. Hnatko, R. McGregor, J. T. McDonough, A. Oka, R. Thomas and J. Hatton, Available and emerging liquid treatment technologies for PFASs, Remediation, 2024, 34(2), e21782, DOI:10.1002/rem.21782.
- S. J. Smith, M. Lauria, L. Ahrens, P. McCleaf, P. Hollman, S. B. Seroka, T. Hamers, H. P. H. Arp and K. Wiberg, Electrochemical oxidation for treatment of PFAS in contaminated water and fractionated foam-A pilot-scale study, ACS ES&T Water, 2023, 3(4), 1201–1211, DOI:10.1021/acsestwater.2c00660.
- M. Sörengård, G. Niarchos, P. E. Jensen and L. Ahrens, Electrodialytic per- and polyfluoroalkyl substances (PFASs) removal mechanism for contaminated soil, Chemosphere, 2019, 232, 224–231, DOI:10.1016/j.chemosphere.2019.05.088.
- E. Kavusi, B. S. Khalaf Ansar, S. Ebrahimi, R. Sharma, S. S. Ghoreishi, K. Nobaharan, S. Abdoli, Z. Dehghanian, B. A. Lajayer, V. Senapathi, G. W. Price and T. Astatkie, Critical review on phytoremediation of polyfluoroalkyl substances from environmental matrices: Need for global concern, Environ. Res., 2023, 217, 114844, DOI:10.1016/j.envres.2022.114844.
- A. Bhattacharya, J. Fathima, S. Varghese, P. Chatterjee and V. Gadhamshetty, Advances in bioremediation strategies for PFAS-contaminated water and soil, Soil Environ. Health, 2025, 3(1), 100126, DOI:10.1016/j.seh.2024.100126.
- S. L. Nason, S. Thomas, C. Stanley, R. Silliboy, M. Blumenthal, W. Zhang, Y. Liang, J. P. Jones, N. Zuverza-Mena, J. C. White, C. L. Haynes, V. Vasiliou, M. P. Timko and B. W. Berger, A comprehensive trial on PFAS remediation: Hemp phytoextraction and PFAS degradation in harvested plants, Environ. Sci.: Adv., 2024, 3, 304–313, 10.1039/D3VA00340J.
- W. Zhang, H. Cao, S. M. Subramanya, P. Savage and Y. Liang, Destruction of perfluoroalkyl acids accumulated in Typha latifolia through hydrothermal liquefaction, ACS Sustainable Chem. Eng., 2020, 8(25), 9257–9262, DOI:10.1021/acssuschemeng.0c03249.
- M. H.-U. Rahman, R. Sikder, T. A. Tonmoy, M. M. Hossain, T. Ye, N. Aich and V. Gadhamshetty, Transforming PFAS Management: A Critical Review of Machine Learning Applications for Enhanced Monitoring and Treatment, J. Water Process Eng., 2025, 70, 106941, DOI:10.1016/j.jwpe.2025.106941.
- A. Hosseinzadeh, A. Altaee, X. Li and J. L. Zhou, Machine Learning-Based Modeling and Analysis of Perfluoroalkyl and Polyfluoroalkyl Substances Controlling Systems in Protecting Water Resources, Curr. Opin. Chem. Eng., 2023, 42, 100983, DOI:10.1016/j.coche.2023.100983.
- M. Caldas Ramos, C. J. Collison and A. D. White, A Review of Large Language Models and Autonomous Agents in Chemistry, Chem. Sci., 2025, 16, 2514–2572, 10.1039/D4SC03921A.
- D. B. Olawade, J. O. Ijiwade, O. Fapohunda, A. O. Ige, D. O. Olajoyetan and O. Z. Wada, Predictive Modeling of PFAS Behavior and Degradation in Novel Treatment Scenarios: A Review, Process Saf. Environ. Prot., 2025, 196, 106869, DOI:10.1016/j.psep.2025.106869.
- L. Yang, Z. Chen, C. A. Goult, T. Schlatzer, R. S. Paton and V. Gouverneur, Phosphate-Enabled Mechanochemical PFAS Destruction for Fluoride Reuse, Nature, 2025, 640(8057), 100–106, DOI:10.1038/s41586-025-08698-5.
- S. Glass, H. A. Santiago-Cruz and W. Chen, et al., Merits, Limitations and Innovation Priorities for Heterogeneous Catalytic Platforms to Destroy PFAS, Nat. Water, 2025, 3, 644–654, DOI:10.1038/s44221-025-00433-8.
- D. B. Miklos, et al., Evaluation of Advanced Oxidation Processes for Water and Wastewater Treatment, A Critical Review, Water Res, 2018, 139, 118–131, DOI:10.1016/j.watres.2018.03.042.
- Y. Wang, M. Warner, K. Li, G. L. Hawkins and Q. Huang, Assessing Explicit Models of Per- and Polyfluoroalkyl Substances Adsorption on Anion Exchange Resins by Rapid Small-Scale Column Tests, Chemosphere, 2022, 300, 134547, DOI:10.1016/j.chemosphere.2022.134547.
- L. Liu and X. Chen, Titanium Dioxide Nanomaterials: Self-Structural Modifications, Chem. Rev., 2014, 114(19), 9890–9918, DOI:10.1021/cr400624r.
- X. Yang and D. Wang, Photocatalysis: From Fundamental Principles to Materials and Applications, ACS Appl. Energy Mater., 2018, 1(12), 6657–6693, DOI:10.1021/acsaem.8b01345.
- C. Mu, C. Lv, X. Meng, J. Sun, Z. Tong and K. Huang, In Situ Characterization Techniques Applied in Photocatalysis: A Review, Adv. Mater. Interfaces, 2023, 10, 2201842, DOI:10.1002/admi.202201842.
- Y. Nosaka and A. Y. Nosaka, Generation and Detection of Reactive Oxygen Species in Photocatalysis, Chem. Rev., 2017, 117(17), 11302–11336, DOI:10.1021/acs.chemrev.7b0016.
- M. K. Wilsey, T. Taseska, Z. Meng, W. Yu and A. M. Müller, Advanced Electrocatalytic Redox Processes for Environmental Remediation of Halogenated Organic Water Pollutants, Chem. Commun., 2023, 59, 11895–11922, 10.1039/D3CC03176D.
- S. Wang, Q. Yang, F. Chen, J. Sun, K. Luo, F. Yao, X. Wang, D. Wang, X. Li and G. Zeng, Photocatalytic Degradation of Perfluorooctanoic Acid and Perfluorooctane Sulfonate in Water: A Critical Review, Chem. Eng. J., 2017, 328, 927–942, DOI:10.1016/j.cej.2017.07.076.
- S. Bertucci and P. Lova, Exploring Solar Energy Solutions for Per- and Polyfluoroalkyl Substances Degradation: Advancements and Future Directions in Photocatalytic Processes, Sol. RRL, 2024, 8, 2400116, DOI:10.1002/solr.202400116.
- J.-M. Arana Juve, J. A. Donoso Reece, M. S. Wong, Z. Wei and M. Ateia, Photocatalysts for Chemical-Free PFOA Degradation – What We Know and Where We Go from Here?, J. Hazard. Mater., 2024, 462, 132651, DOI:10.1016/j.jhazmat.2023.132651.
- Y. Qi, H. Cao, W. Pan, C. Wang and Y. Liang, The Role of Dissolved Organic Matter during Per- and Polyfluorinated Substance (PFAS) Adsorption, Degradation, and Plant Uptake: A Review, J. Hazard. Mater., 2022, 436, 129139, DOI:10.1016/j.jhazmat.2022.129139.
- S. Verma, R. S. Varma and M. N. Nadagouda, Remediation and Mineralization Processes for Per- and Polyfluoroalkyl Substances (PFAS) in Water: A Review, Sci. Total Environ., 2021, 794, 148987, DOI:10.1016/j.scitotenv.2021.148987.
- H. Park, C. D. Vecitis, J. Cheng, W. Choi, B. T. Mader and M. R. Hoffmann, Reductive Defluorination of Aqueous Perfluorinated Alkyl Surfactants: Effects of Ionic Headgroup and Chain Length, J. Phys. Chem. A, 2009, 113(4), 690–696, DOI:10.1021/jp807116q.
- S. Kutsuna, Y. Nagaoka, K. Takeuchi and H. Hori, TiO2-Induced Heterogeneous Photodegradation of a Fluorotelomer Alcohol in Air, Environ. Sci. Technol., 2006, 40(21), 6824–6829, DOI:10.1021/es060852.
- X.-Q. Gong and A. Selloni, Reactivity of Anatase TiO2 Nanoparticles: The Role of the Minority (001) Surface, J. Phys. Chem. B, 2005, 109(42), 19560–19562, DOI:10.1021/jp055311g.
- C. Liu, Y. Qin, W. Guo, Y. Shi, Z. Wang, Y. Yu and L. Wu, Visible-Light-Driven Photocatalysis over Nano-TiO2 with Different Morphologies: From Morphology through Active Site to Photocatalytic Performance, Appl. Surf. Sci., 2022, 580, 152262, DOI:10.1016/j.apsusc.2021.152262.
- I. A. Janković, Z. V. Saponjić, E. S. Džunuzović and J. M. Nedeljković, New Hybrid Properties of TiO2 Nanoparticles Surface Modified with Catecholate Type Ligands, Nanoscale Res. Lett., 2009, 5(1), 81–88, DOI:10.1007/s11671-009-9447-y.
- A. Wypych, I. Bobowska, M. Tracz, A. Opasinska, S. Kadlubowski, A. Krzywania-Kaliszewska, J. Grobelny and P. Wojciechowski, Dielectric Properties and Characterisation of Titanium Dioxide Obtained by Different Chemistry Methods, J. Nanomater., 2014, 124814, DOI:10.1155/2014/124814.
- S. Reghunath, D. Pinheiro and S. K. R. Devi, A Review of Hierarchical Nanostructures of TiO2: Advances and Applications, Appl. Surf. Sci. Adv., 2021, 3, 100063, DOI:10.1016/j.apsadv.2021.100063.
- A. Singh, V. Goyal, J. Singh and M. Rawat, Structural, Morphological, Optical and Photocatalytic Properties of Green Synthesized TiO2 NPs, Curr. Res. Green Sustainable Chem., 2020, 3, 100033, DOI:10.1016/j.crgsc.2020.100033.
- K. Nakata and A. Fujishima, TiO2 Photocatalysis: Design and Applications, J. Photochem. Photobiol., C, 2012, 13(3), 169–189, DOI:10.1016/j.jphotochemrev.2012.06.001.
- M. Nemiwal, T. C. Zhang and D. Kumar, Recent Progress in g-C3N4, TiO2 and ZnO Based Photocatalysts for Dye Degradation: Strategies to Improve Photocatalytic Activity, Sci. Total Environ., 2021, 767, 144896, DOI:10.1016/j.scitotenv.2020.144896.
- N. O. Etafo, M. O. Bamidele, A. Bamisaye and Y. A. Alli, Revolutionizing Photocatalysis: Unveiling Efficient Alternatives to Titanium (IV) Oxide and Zinc Oxide for Comprehensive Environmental Remediation, J. Water Process Eng., 2024, 62, 105369, DOI:10.1016/j.jwpe.2024.105369.
- L. Liu, H. Zhao, J. M. Andino and Y. Li, Photocatalytic CO2 Reduction with H2O on TiO2 Nanocrystals: Comparison of Anatase, Rutile, and Brookite Polymorphs and Exploration of Surface Chemistry, ACS Catal., 2012, 2(8), 1817–1828, DOI:10.1021/cs300273q.
- D. R. Eddy, G. A. N. Sheha, M. D. Permana, N. Saito, T. Takei, N. Kumada, Irkham, I. Rahayu, I. Abe, Y. Sekine, T. Oyumi and Y. Izumi, Study on Triphase of Polymorphs TiO2 (Anatase/Rutile/Brookite) for Boosting Photocatalytic Activity of Metformin Degradation, Chemosphere, 2024, 351, 141206, DOI:10.1016/j.chemosphere.2024.141206.
- A. Di Paola, M. Bellardita and L. Palmisano, Brookite, the Least Known TiO2 Photocatalyst, Catalysts, 2013, 3, 36–73, DOI:10.3390/catal3010036.
- M. El Mchaouri, S. Mallah, D. Abouhajjoub, W. Boumya, R. Elmoubarki, A. Essadki, N. Barka and A. Elhalil, Engineering TiO2 Photocatalysts for Enhanced Visible-Light Activity in Wastewater Treatment Applications, Tetrahedron Green Chem, 2025, 6, 100084, DOI:10.1016/j.tgchem.2025.100084.
- N. M. Alabdallah, S. M. Alluqmani, H. M. Almarri and A. A. Al-Zahrani, Physical, Chemical, and Biological Routes of Synthetic Titanium Dioxide Nanoparticles and Their Crucial Role in Temperature Stress Tolerance in Plants, Heliyon, 2024, 10(4), e26537, DOI:10.1016/j.heliyon.2024.e26537.
- J. Wu, G. R. Bai and J. A. Eastman, et al., Synthesis of TiO2 Nanoparticles Using Chemical Vapor Condensation, MRS Online Proc. Libr., 2005, 879, Z7.12, DOI:10.1557/PROC-879-Z7.12.
- M. Cargnello, T. R. Gordon and C. B. Murray, Solution-Phase Synthesis of Titanium Dioxide Nanoparticles and Nanocrystals, Chem. Rev., 2014, 114(19), 9319–9345, DOI:10.1021/cr500170p.
- U. Öztürk and A. S. Çitak, Synthesis of Titanium Dioxide Nanoparticles with Renewable Resources and Their Applications: Review, Turk. J. Chem., 2022, 46(5), 1345–1357, DOI:10.55730/1300-0527.3443.
- R. Dillert, D. Bahnemann and H. Hidaka, Light-Induced Degradation of Perfluorocarboxylic Acids in the Presence of Titanium Dioxide, Chemosphere, 2007, 67(4), 785–792, DOI:10.1016/j.chemosphere.2006.10.023.
- S. C. Panchangam, A. Y.-C. Lin, K. L. Shaik and C.-F. Lin, Decomposition of Perfluorocarboxylic Acids (PFCAs) by Heterogeneous Photocatalysis in Acidic Aqueous Medium, Chemosphere, 2009, 77(2), 242–248, DOI:10.1016/j.chemosphere.2009.07.003.
- S. C. Panchangam, A. Y.-C. Lin, J.-H. Tsai and C.-F. Lin, Sonication-Assisted Photocatalytic Decomposition of Perfluorooctanoic Acid, Chemosphere, 2009, 75(5), 654–660, DOI:10.1016/j.chemosphere.2008.12.065.
- Y. Wang and P. Zhang, Photocatalytic Decomposition of Perfluorooctanoic Acid (PFOA) by TiO2 in the Presence of Oxalic Acid, J. Hazard. Mater., 2011, 192(3), 1869–1875, DOI:10.1016/j.jhazmat.2011.07.026.
- S. Gatto, M. Sansotera, F. Persico, M. Gola, C. Pirola, W. Panzeri, W. Navarrini and C. L. Bianchi, Surface Fluorination on TiO2 Catalyst Induced by Photodegradation of Perfluorooctanoic Acid, Catal. Today, 2015, 241(Part A), 8–14, DOI:10.1016/j.cattod.2014.04.031.
- P. S. Surdhar, S. P. Mezyk and D. A. Armstrong, Reduction Potential of the CO2− Radical Anion in Aqueous Solutions, J. Phys. Chem., 1989, 93, 3360–3363 CrossRef CAS.
- Y. Li and F. Wasgestian, Photocatalytic Reduction of Nitrate Ions on TiO2 by Oxalic Acid, J. Photochem. Photobiol., A, 1998, 112, 255–259 CrossRef CAS.
- L. L. Perissinotti, M. A. Brusa and M. A. Grela, Yield of Carboxyl Anion Radicals in the Photocatalytic Degradation of Formate over TiO2 Particles, Langmuir, 2001, 17, 8422–8427 CrossRef CAS.
- M. Sansotera, F. Persico, V. Rizzi, W. Panzeri, C. Pirola, C. L. Bianchi, A. Mele and W. Navarrini, The Effect of Oxygen in the Photocatalytic Oxidation Pathways of Perfluorooctanoic Acid, J. Fluor. Chem., 2015, 179, 159–168, DOI:10.1016/j.jfluchem.2015.06.019.
- M. Peydayesh, M. Bagnani, W. L. Soon and R. Mezzenga, Turning Food Protein Waste into Sustainable Technologies, Chem. Rev., 2023, 123(5), 2112–2154, DOI:10.1021/acs.chemrev.2c00236.
- A. Ghareeb, A. Fouda and R. M. Kishk, et al., Unlocking the Potential of Titanium Dioxide Nanoparticles: An Insight into Green Synthesis, Optimizations, Characterizations, and Multifunctional Applications, Microb. Cell Fact., 2024, 23, 341, DOI:10.1186/s12934-024-02609-5.
- M. Saheed, T. Jimoh Oladejo, E. Rabi, E. Muhammed Binin, A. Azeezah Taiwo, S. Damola Taye, S. Abdulmumuni, O. Adekunle Jelili, A. Hassana Ladio and A. Saka Abdulkareem, et al., Photocatalytic Degradation and Defluorination of Per- and Poly-Fluoroalkyl Substances (PFASs) Using Biosynthesized TiO2 Nanoparticles under UV–Visible Light, Eng. Proc., 2023, 37, 114, DOI:10.3390/ECP2023-14630.
- S. Murgolo, V. Yargeau, R. Gerbasi, F. Visentin, N. El Habra, G. Ricco, I. Lacchetti, M. Carere, M. L. Curri and G. Mascolo, A New Supported TiO2 Film Deposited on Stainless Steel for the Photocatalytic Degradation of Contaminants of Emerging Concern, Chem. Eng. J., 2017, 318, 103–111, DOI:10.1016/j.cej.2016.05.125.
- R. X. Furtado, S. de, C. A. Sabatini, M. Zaiat and E. B. Azevedo, Perfluorooctane Sulfonic Acid (PFOS) Degradation by Optimized Heterogeneous Photocatalysis (TiO2/UV) Using the Response Surface Methodology (RSM), J. Water Process Eng., 2021, 41, 101986, DOI:10.1016/j.jwpe.2021.101986.
- M. Sansotera, F. Persico, C. Pirola, W. Navarrini, A. Di Michele and C. L. Bianchi, Decomposition of Perfluorooctanoic Acid Photocatalyzed by Titanium Dioxide: Chemical Modification of the Catalyst Surface Induced by Fluoride Ions, Appl. Catal., B, 2014, 148–149, 29–35, DOI:10.1016/j.apcatb.2013.10.038.
- T. Ochiai, Y. Iizuka, K. Nakata, T. Murakami, D. A. Tryk, Y. Koide, Y. Morito and A. Fujishima, Efficient Decomposition of Perfluorocarboxylic Acids in Aqueous Suspensions of a TiO2 Photocatalyst with Medium-Pressure Ultraviolet Lamp Irradiation under Atmospheric Pressure, Ind. Eng. Chem. Res., 2011, 50(19), 10943–10947, DOI:10.1021/ie1017496.
- J. Zweigle, B. Bugsel, C. Capitain and C. Zwiener, PhotoTOP: PFAS Precursor Characterization by UV/TiO2 Photocatalysis, Environ. Sci. Technol., 2022, 56(22), 15728–15736, DOI:10.1021/acs.est.2c05652.
- Y.-q Gao, X.-m Zhong, X.-t Ren, F.-x Tian, N.-y Gao and C. Li, Synergistic Mechanism and Toxicity Reduction Potential in Medium-Pressure UV/Chlorine/TiO2 System for Metronidazole Removal, J. Water Process Eng., 2024, 65, 105903, DOI:10.1016/j.jwpe.2024.105903.
- H. Hori, E. Hayakawa, H. Einaga, S. Kutsuna, K. Koike, T. Ibusuki, H. Kiatagawa and R. Arakawa, Decomposition of Environmentally Persistent Perfluorooctanoic Acid in Water by Photochemical Approaches, Environ. Sci. Technol., 2004, 38, 6118 CrossRef CAS PubMed.
- B. Zhao and P. Zhang, Photocatalytic Decomposition of Perfluorooctanoic Acid with β-Ga2O3 Wide Bandgap Photocatalyst, Catal. Commun., 2009, 10, 1184 CrossRef CAS.
- J. Janda, K. Nodler, M. Scheurer, O. Happel, G. Nurenberg, C. Zwiener and F. T. Lange, Closing the gap,Inclusion of ultrashort-chain perfluoroalkyl carboxylic acids in the total oxidizable precursor (TOP) assay protocol., Environ. Sci. Process Impacts, 2019, 21(11), 1926–1935 RSC.
- E. F. Houtz and D. L. Sedlak, Oxidative conversion as a means of detecting precursors to perfluoroalkyl acids in urban runoff, Environ. Sci. Technol., 2012, 46(17), 9342–9349 CrossRef CAS PubMed.
- M. Jiang, K. Nozaki, T. Mokudai, Y. Nakano, M. Uo, K. Yamashita, S. Ohara and N. Wakabayashi, Enhancing the Photocatalytic Activity and Antibacterial Efficiency of TiO2 Nanosheets via Doping with Ag, Cu, or Ce, ACS Appl. Nano Mater., 2025, 8(22), 11568–11581, DOI:10.1021/acsanm.5c01768.
- V. Etacheri, C. Di Valentin, J. Schneider, D. Bahnemann and S. C. Pillai, Visible-Light Activation of TiO2 Photocatalysts: Advances in Theory and Experiments, J. Photochem. Photobiol., C, 2015, 25, 1–29, DOI:10.1016/j.jphotochemrev.2015.08.003.
- C. Thambiliyagodage and L. Usgodaarachchi, Photocatalytic Activity of N, Fe and Cu Co-Doped TiO2 Nanoparticles under Sunlight, Curr. Res. Green Sustainable Chem., 2021, 4, 100186, DOI:10.1016/j.crgsc.2021.100186.
- A. Khlyustova, N. Sirotkin, T. Kusova, A. Kraev, V. Titov and A. Agafonov, Doped TiO2: The Effect of Doping Elements on Photocatalytic Activity, Mater. Adv., 2020, 1, 1193–1201, 10.1039/D0MA00171F.
- S. Mathew, B. K. John, T. Abraham and B. Mathew, Metal-Doped Titanium Dioxide for Environmental Remediation, Hydrogen Evolution and Sensing: A Review, ChemistrySelect, 2021, 6, 12742 CrossRef CAS.
- S. G. Kumar and L. Gomathi Devi, Review on Modified TiO2 Photocatalysis under UV/Visible Light: Selected Results and Related Mechanisms on Interfacial Charge Carrier Transfer Dynamics, J. Phys. Chem. A, 2011, 115(46), 13211–13241, DOI:10.1021/jp204364a.
- C. He, Y. Yu, X. Hu and A. Larbot, Influence of Silver Doping on the Photocatalytic Activity of Titania Films, Appl. Surf. Sci., 2002, 200, 239–247, DOI:10.1016/S0169-4332(02)00927-3.
- A.-W. Xu, Y. Gao and H.-Q. Liu, The Preparation, Characterization, and Their Photocatalytic Activities of Rare-Earth-Doped TiO2 Nanoparticles, J. Catal, 2002, 207, 151–157, DOI:10.1006/jcat.2002.3539.
- N. U. Saqib, R. Adnan and I. A. Shah, A Mini-Review on Rare Earth Metal-Doped TiO2 for Photocatalytic Remediation of Wastewater, Environ. Sci. Pollut. Res., 2016, 23, 15941–15951, DOI:10.1007/s11356-016-6984-7.
- L. Yang, J. Zhang, H. Tong, X. Li, D. Zhao and K. Lan, Synthesis of Mesoporous Titania Nanomaterials for Evolving Photocatalytic Applications, Adv. Energy Mater., 2025, e02405, DOI:10.1002/aenm.202502405.
- J. Watthanaarun, V. Pavarajarn and P. Supaphol, Titanium (IV) Oxide Nanofibers by Combined Sol–Gel and Electrospinning Techniques: Preliminary Report on Effects of Preparation Conditions and Secondary Metal Dopant, Sci. Technol. Adv. Mater., 2005, 6, 240–245, DOI:10.1016/j.stam.2005.02.002.
- R. Shah, D. Khan, A. Al-Anazi, W. Ahmad, I. Ullah, N. S. Shah and J. A. Khan, Non-Metal Doped ZnO and TiO2 Photocatalysts for Visible Light Active Degradation of Pharmaceuticals and Hydrogen Production: A Review, Appl. Catal., Open, 2025, 204, 207043, DOI:10.1016/j.apcato.2025.207043.
- P. S. Basavarajappa, S. B. Patil, N. Ganganagappa, K. Raghava Reddy, A. V. Raghu and C. Venkata Reddy, Recent Progress in Metal-Doped TiO2, Non-Metal Doped/Co-Doped TiO2 and TiO2 Nanostructured Hybrids for Enhanced Photocatalysis, Int. J. Hydrogen Energy, 2020, 45(13), 7764–7778, DOI:10.1016/j.ijhydene.2019.07.241.
- C. Di Valentin, G. Pacchioni, A. Selloni, S. Livraghi and E. Giamello, Characterization of Paramagnetic Species in N-Doped TiO2 Powders by EPR Spectroscopy and DFT Calculations, J. Phys. Chem. B, 2005, 109(23), 11414–11419, DOI:10.1021/jp051756t.
- F. FengHui and C. ChengBu, DFT Description on Electronic Structure and Optical Absorption Properties of Anionic S-Doped Anatase TiO2, J. Phys. Chem. B, 2006, 110(36), 17866–17871, DOI:10.1021/jp0635462.
- R. Daghrir, P. Drogui and D. Robert, Modified TiO2 for Environmental Photocatalytic Applications: A Review, Ind. Eng. Chem. Res., 2013, 52(10), 3581–3599, DOI:10.1021/ie303468t.
- C. R. Estrellan, C. Salim and H. Hinode, Photocatalytic Decomposition of Perfluorooctanoic Acid by Iron and Niobium Co-Doped Titanium Dioxide, J. Hazard. Mater., 2010, 179(1–3), 79–83, DOI:10.1016/j.jhazmat.2010.02.060.
- M.-J. Chen, S.-L. Lo, Y.-C. Lee and C.-C. Huang, Photocatalytic Decomposition of Perfluorooctanoic Acid by Transition-Metal Modified Titanium Dioxide, J. Hazard. Mater., 2015, 288, 168–175, DOI:10.1016/j.jhazmat.2015.02.004.
- M. S. Vohra, S. Kim and W. Choi, Effects of Surface Fluorination of TiO2 on the Photocatalytic Degradation of Tetramethylammonium, J. Photochem. Photobiol. A Chem., 2003, 160(1–2), 55–60, DOI:10.1016/S1010-6030(03)00221-1.
- M. Li, Z. Yu, Q. Liu, L. Sun and W. Huang, Photocatalytic Decomposition of Perfluorooctanoic Acid by Noble Metallic Nanoparticles Modified TiO2, Chem. Eng. J., 2016, 286, 232–238, DOI:10.1016/j.cej.2015.10.037.
- C. Vanlalhmingmawia, S. M. Lee and D. Tiwari, Plasmonic Noble Metal Doped Titanium Dioxide Nanocomposites: Newer and Exciting Materials in the Remediation of Water Contaminated with Micropollutants, J. Water Process Eng., 2023, 51, 103360, DOI:10.1016/j.jwpe.2022.103360.
- F. Zhan, J. Qi, R. Li, H. Zhao, Y. Liu and P. La, Synthesis, Photocatalytic Applications and Future Prospects of Noble Metal-Modified Semiconductors Fabricated via Photodeposition, Phys. Chem. Chem. Phys., 2025, 27, 13770–13792, 10.1039/D5CP01191D.
- M.-J. Chen, S.-L. Lo, Y.-C. Lee, J. Kuo and C.-H. Wu, Decomposition of Perfluorooctanoic Acid by Ultraviolet Light Irradiation with Pb-Modified Titanium Dioxide, J. Hazard. Mater., 2016, 303, 111–118, DOI:10.1016/j.jhazmat.2015.10.011.
- B. Huang, Z. Wu, H. Zhou, J. Li, C. Zhou, Z. Xiong, Z. Pan, G. Yao and B. Lai, Recent Advances in Single-Atom Catalysts for Advanced Oxidation Processes in Water Purification, J. Hazard. Mater., 2021, 412, 125253, DOI:10.1016/j.jhazmat.2021.125253.
- T. Wei, J. Zhou and X. An, Recent Advances in Single-Atom Catalysts (SACs) for Photocatalytic Applications, Mater. Rep. Energy, 2024, 4(3), 100285, DOI:10.1016/j.matre.2024.100285.
- S.-M. Wu and P. Schmuki, Single Atom Cocatalysts in Photocatalysis, Adv. Mater., 2025, 37, 2414889, DOI:10.1002/adma.202414889.
- U. Kerketta, A. B. Tesler and P. Schmuki, Single-Atom Co-Catalysts Employed in Titanium Dioxide Photocatalysis, Catalysts, 2022, 12, 1223, DOI:10.3390/catal12101223.
- H. Sun, R. Tang and J. Huang, Considering Single-Atom Catalysts as Photocatalysts from Synthesis to Application, iScience, 2022, 25(5), 104232, DOI:10.1016/j.isci.2022.104232.
- X. Liu, K. Gong, X. Duan, W. Wei, T. Wang, Z. Chen, L. Zhang and B.-J. Ni, Photo-Induced Bismuth Single Atoms on TiO2 for Highly Efficient Photocatalytic Defluorination of Perfluorooctanoic Acid: Ionization of the C–F Bond, ACS ES&T Eng., 2023, 3(10), 1626–1636, DOI:10.1021/acsestengg.3c00177.
- Z. Cui, A. Jafarzadeh, Y. Hao, L. Liu, L. Li and Y. Zheng, Prediction of the Decomposition Tendency of C5F10O on Discharged Metal Surfaces, IEEE Trans. Dielectr. Electr. Insul., 2023, 30, 1365–1367, DOI:10.1109/TDEI.2023.3260035.
- C. B. Anucha, I. Altin, E. Bacaksiz and V. N. Stathopoulos, Titanium Dioxide (TiO2)-Based Photocatalyst Materials Activity Enhancement for Contaminants of Emerging Concern (CECs) Degradation: In the Light of Modification Strategies, Chem. Eng. J. Adv., 2022, 10, 100262, DOI:10.1016/j.ceja.2022.100262.
- M. Dahl, Y. Liu and Y. Yin, Composite Titanium Dioxide Nanomaterials, Chem. Rev., 2014, 114(19), 9853–9889, DOI:10.1021/cr400634p.
- X. Yao, J. Zuo, Y.-J. Wang, N.-N. Song, H.-H. Li and K. Qiu, Enhanced photocatalytic degradation of perfluorooctanoic acid by mesoporous Sb2O3/TiO2 heterojunctions, Front. Chem., 2021, 9, 690520, DOI:10.3389/fchem.2021.690520.
- F. Chen, A. He, Y. Wang, W. Yu, H. Chen, F. Geng, Z. Li, Z. Zhou, Y. Liang, J. Fu, L. Zhao and Y. Wang, Efficient Photodegradation of PFOA Using Spherical BiOBr Modified TiO2 via Hole-Remained Oxidation Mechanism, Chemosphere, 2022, 298, 134176, DOI:10.1016/j.chemosphere.2022.134176.
- A. Chen, H. Ji, Z. Xu, Z. Wang, H. Xie and L. Zhang, Unique S-Scheme Structures in Electrostatic Self-Assembled ReS2–TiO2 for High-Efficiency Perfluorooctanoic Acid Removal, J. Colloid Interface Sci., 2025, 681, 63–70, DOI:10.1016/j.jcis.2024.11.164.
- X. Li, P. Zhang, L. Jin, T. Shao, Z. Li and J. Cao, Efficient Photocatalytic Decomposition of Perfluorooctanoic Acid by Indium Oxide and Its Mechanism, Environ. Sci. Technol., 2012, 46(10), 5528–5534, DOI:10.1021/es204279u.
- X. Zhang, R. Li, M. Jia, S. Wang, Y. Huang and C. Chen, Degradation of Ciprofloxacin in Aqueous Bismuth Oxybromide (BiOBr) Suspensions under Visible Light Irradiation: A Direct Hole Oxidation Pathway, Chem. Eng. J., 2015, 274, 290–297, DOI:10.1016/j.cej.2015.03.077.
- Y.-F. Fang, W.-H. Ma, Y.-P. Huang and G.-W. Cheng, Exploring the Reactivity of Multicomponent Photocatalysts: Insight into the Complex Valence Band of BiOBr, Chem. – Eur. J., 2013, 19(9), 3224–3229, DOI:10.1002/chem.201202602.
- L. Liang, S. Gao, J. Zhu, L. Wang, Y. Xiong, X. Xia and L. Yang, The Enhanced Photocatalytic Performance toward Carbamazepine by Nitrogen-Doped Carbon Dots Decorated on BiOBr/CeO2: Mechanism Insight and Degradation Pathways, Chem. Eng. J., 2020, 391, 123599, DOI:10.1016/j.cej.2019.123599.
- B. Singh, G. Kaur, P. Singh, K. Singh, J. Sharma, M. Kumar, R. Bala, R. Meena, S. K. Sharma and A. Kumar, Nanostructured BN–TiO2 Composite with Ultra-High Photocatalytic Activity, New J. Chem., 2017, 41(20), 11640–11646 RSC.
- L. Duan, B. Wang, K. Heck, S. Guo, C. A. Clark, J. Arredondo, M. Wang, T. P. Senftle, P. Westerhoff, X. Wen, Y. Song and M. S. Wong, Efficient Photocatalytic PFOA Degradation over Boron Nitride, Environ. Sci. Technol. Lett., 2020, 7(8), 613–619, DOI:10.1021/acs.estlett.0c00434.
- C. Yang, G. Zhang, Y. Meng, G. Pan, Z. Ni and S. Xia, Direct Z-Scheme CeO2@LDH Core–Shell Heterostructure for Photodegradation of Rhodamine B by Synergistic Persulfate Activation, J. Hazard. Mater., 2021, 408, 124908, DOI:10.1016/j.jhazmat.2020.124908.
- Y. Du, Y. Meng, G. X. Pan, H. Shen, Y. Y. Yao, B. Xie, Z. M. Ni and S. J. Xia, Comparison of Performance and Mechanism for Single and Double Vacancies MoS2 Catalyzing Water Gas Shift Reaction, Int. J. Hydrogen Energy, 2023, 48, 24628–24639, DOI:10.1016/j.ijhydene.2022.06.198.
- E. D. H. Kong, C. W. Lai, J. C. Juan, Y. L. Pang, C. S. Khe, I. A. Badruddin, F. Gapsari and K. Anam, Recent Advances in Titanium Dioxide Bio-Derived Carbon Photocatalysts for Organic Pollutant Degradation in Wastewater, iScience, 2025, 28(5), 112368, DOI:10.1016/j.isci.2025.112368.
- B. Xia, P. Cao, X. Tang, Q. Shi, Y. Lv and X. Zhang, Promoting Interfacial Charge Separation for Efficient Photocatalysis of Organic Dyes Using ESM-Solidified TiO2-Based Coupling Materials, Mater. Today Commun., 2025, 45, 112436, DOI:10.1016/j.mtcomm.2025.112436.
- M. R. Meirelles, A. S. Giroto, K. Furukawa and M. Gonçalves, Valorization of Lignocellulosic Biomass for Photocatalytic Applications: Development of Activated Carbon–TiO2 Composites, ACS Sustainable Resour. Manag, 2025, 2(3), 524–535, DOI:10.1021/acssusresmgt.4c00512.
- X. Dong, H. Li, Y. Guo, C. Ge and J. Zha, Construction of Hierarchical Black TiO2/Carbon Fiber: Enhanced Photocatalytic Activity Based on Schottky Heterojunctions, ACS Appl. Bio Mater., 2024, 7(8), 5397–5410, DOI:10.1021/acsabm.4c00572.
- N. T. Padmanabhan, N. Thomas, J. Louis, D. T. Mathew, P. Ganguly, H. John and S. C. Pillai, Graphene Coupled TiO2 Photocatalysts for Environmental Applications: A Review, Chemosphere, 2021, 271, 129506, DOI:10.1016/j.chemosphere.2020.129506.
- F. Li, Z. Wei, K. He, L. Blaney, X. Cheng, T. Xu, W. Liu and D. Zhao, A Concentrate-and-Destroy Technique for Degradation of Perfluorooctanoic Acid in Water Using a New Adsorptive Photocatalyst, Water Res, 2020, 185, 116219, DOI:10.1016/j.watres.2020.116219.
- T. Xu, H. Ji, Y. Gu, T. Tong, Y. Xia, L. Zhang and D. Zhao, Enhanced Adsorption and Photocatalytic Degradation of Perfluorooctanoic Acid in Water Using Iron (Hydr)oxides/Carbon Sphere Composite, Chem. Eng. J., 2020, 388, 124230, DOI:10.1016/j.cej.2020.124230.
- Z. Wei, S. Luo, R. Xiao, R. Khalfin and R. Semiat, Characterization and Quantification of Chromate Adsorption by Layered Porous Iron Oxyhydroxide: An Experimental and Theoretical Study, J. Hazard. Mater., 2017, 338, 472–481, DOI:10.1016/j.jhazmat.2017.05.049.
- Y. Zhu, T. Xu, D. Zhao, F. Li, W. Liu, B. Wang and B. An, Adsorption and Solid-Phase Photocatalytic Degradation of Perfluorooctane Sulfonate in Water Using Gallium-Doped Carbon-Modified Titanate Nanotubes, Chem. Eng. J., 2021, 421(Part 1), 129676, DOI:10.1016/j.cej.2021.129676.
- J.-M. Arana Juve, F. Li, Y. Zhu, W. Liu, L. D. M. Ottosen, D. Zhao and Z. Wei, Concentrate and Degrade PFOA with a Photo-Regenerable Composite of In-Doped TNTs@AC, Chemosphere, 2022, 300, 134495, DOI:10.1016/j.chemosphere.2022.134495.
- Q. Yu, R. Zhang, S. Deng, J. Huang and G. Yu, Sorption of Perfluorooctane Sulfonate and Perfluorooctanoate on Activated Carbons and Resin: Kinetic and Isotherm Study, Water Res., 2009, 43(4), 1150–1158, DOI:10.1016/j.watres.2008.12.001.
- N. Wibowo, L. Setyadhi, D. Wibowo, J. Setiawan and S. Ismadji, Adsorption of Benzene and Toluene from Aqueous Solutions onto Activated Carbon and Its Acid and Heat Treated Forms: Influence of Surface Chemistry on Adsorption, J. Hazard. Mater., 2007, 146, 237–242, DOI:10.1016/j.jhazmat.2006.12.011.
- R. N. Gayen, S. Hussain, R. Bhar and A. K. Pal, Synthesis and Characterization of Indium Phosphide Films Prepared by Co-Evaporation Technique, Vacuum, 2012, 86, 1240–1247, DOI:10.1016/j.vacuum.2011.11.005.
- Z. M. Detweiler, S. M. Wulfsberg, M. G. Frith, A. B. Bocarsly and S. L. Bernasek, The Oxidation and Surface Speciation of Indium and Indium Oxides Exposed to Atmospheric Oxidants, Surf. Sci., 2016, 648, 188–195, DOI:10.1016/j.susc.2015.10.026.
- M. Kosmulski, Pristine Points of Zero Charge of Gallium and Indium Oxides, J. Colloid Interface Sci., 2001, 238, 225–227, DOI:10.1006/jcis.2001.7484.
- C. Song, P. Chen, C. Wang and L. Zhu, Photodegradation of Perfluorooctanoic Acid by Synthesized TiO2–MWCNT Composites under 365 nm UV Irradiation, Chemosphere, 2012, 86(8), 853–859, DOI:10.1016/j.chemosphere.2011.11.034.
- B. Gomez-Ruiz, P. Ribao, N. Diban, M. J. Rivero, I. Ortiz and A. Urtiaga, Photocatalytic Degradation and Mineralization of Perfluorooctanoic Acid (PFOA) Using a Composite TiO2–rGO Catalyst, J. Hazard. Mater., 2018, 344, 950–957, DOI:10.1016/j.jhazmat.2017.11.048.
- N. Chowdhury and H. Choi, Photocatalytic Degradation of Perfluorooctanoic Acid on Pb-Doped TiO2 Coated with Reduced Graphene Oxide, Water Environ. Res., 2023, 95(5), e10871, DOI:10.1002/wer.10871.
- M. J. Rivero, P. Ribao, B. Gomez-Ruiz, A. Urtiaga and I. Ortiz, Comparative Performance of TiO2–rGO Photocatalyst in the Degradation of Dichloroacetic and Perfluorooctanoic Acids, Sep. Purif. Technol., 2020, 240, 116637, DOI:10.1016/j.seppur.2020.116637.
- W. Li, C. Zhuang, Y. Li, C. Gao, W. Jiang, Z. Sun and K. Qi, Anchoring Ultra-Small TiO2 Quantum Dots onto Ultra-Thin and Large-Sized MXene Nanosheets for Highly Efficient Photocatalytic Water Splitting, Ceram. Int., 2021, 47(15), 21769–21776, DOI:10.1016/j.ceramint.2021.04.192.
- C. Dimitriou, L. Belles, N. Boukos and Y. Deligiannakis, {TiO2/TiO2(B)} Quantum Dot Hybrids: A Comprehensible Route toward High-Performance [>0.1 mol g−1 h−1] Photocatalytic H2 Production from H2O, ACS Catal., 2024, 14(23), 17919–17934, DOI:10.1021/acscatal.4c05001.
- M. Ikram, M. A. Ul Haq, A. Haider, J. Haider, A. Ul-Hamid, I. Shahzadi, M. A. Bari, S. Ali, S. Goumri-Said and M. B. Kanoun, The Enhanced Photocatalytic Performance and First-Principles Computational Insights of Ba Doping-Dependent TiO2 Quantum Dots, Nanoscale Adv, 2022, 4(18), 3996–4008, 10.1039/d2na00361a.
- C. Zhu, J. Xu, S. Song, J. Wang, Y. Li, R. Liu and Y. Shen, TiO2 Quantum Dots Loaded Sulfonated Graphene Aerogel for Effective Adsorption-Photocatalysis of PFOA, Sci. Total Environ., 2020, 698, 134275, DOI:10.1016/j.scitotenv.2019.134275.
- M. Nejatpour, A. M. Ünsür, B. Yılmaz, M. Gül, B. Ozden, S. Barisci and M. Dükkancı, Enhanced Photodegradation of Perfluorocarboxylic Acids (PFCAs) Using Carbon Quantum Dots (CQDs) Doped TiO2 Photocatalysts: A Comparative Study between Exfoliated Graphite and Mussel Shell-Derived CQDs, J. Environ. Chem. Eng., 2025, 13(1), 115382, DOI:10.1016/j.jece.2025.115382.
- A. M. Ünsür, M. Nejatpour, M. Dükkancı, B. Ozden, S. Samai, C. Parlak and S. Barisci, Green Approach for Perfluorocarboxylic Acids (PFCAs) Removal with Density Functional Theory (DFT) Insights: Peanut-Shell Biomass-Based Carbon Quantum Dots (PCQDs) Coupled with TiO2 Photocatalyst, J. Hazard. Mater., 2025, 495, 139060, DOI:10.1016/j.jhazmat.2025.139060.
- N. A. Pechnikova, K. Domvri, K. Porpodis, M. S. Istomina, A. V. Iaremenko and A. V. Yaremenko, Carbon Quantum Dots in Biomedical Applications: Advances, Challenges, and Future Prospects, Aggregate, 2025, 6, e707, DOI:10.1002/agt2.707.
- M. A. Akhtar, J. Anum, S. S. Ahmed, M. A. Ahmed, A. M. Khan, C. M. Sayes and A. Rahim, Eco-Friendly Synthesis of N-Doped Carbon Quantum Dots from Bombax ceiba Stem: A Photophysical Study with Exploratory Sensing of Fe3+/Cd2+ and Dye Degradation, J. Water Process Eng., 2025, 78, 108704, DOI:10.1016/j.jwpe.2025.108704.
- X. Li, Y. Bai, X. Shi, N. Su, G. Nie, R. Zhang, H. Nie and L. Ye, Applications of MXene (Ti3C2Tx) in Photocatalysis: A Review, Mater. Adv., 2021, 2(5), 1570–1594, 10.1039/D0MA00938E.
- M. Urso, L. Bruno, S. Dattilo, S. C. Carroccio and S. Mirabella, Band Engineering versus Catalysis: Enhancing the Self-Propulsion of Light-Powered MXene-Derived Metal–TiO2 Micromotors To Degrade Polymer Chains, ACS Appl. Mater. Interfaces, 2024, 16(1), 1293–1307, DOI:10.1021/acsami.3c13470.
- M. Rostami, A. Badiei, M. Alijani, A. Das and G. M. Ziarani, Nanoarchitectonic MOF-Derived Materials for Enhanced Photocatalytic Activity in Organic Contaminant Removal: A Review, Alex. Eng. J., 2025, 126, 448–479, DOI:10.1016/j.aej.2025.04.092.
- I. Abánades Lázaro, H. Szalad, P. Valiente, J. Albero, H. García and C. Martí-Gastaldo, Tuning the Photocatalytic Activity of Ti-Based Metal–Organic Frameworks through Modulator Defect-Engineered Functionalization, ACS Appl. Mater. Interfaces, 2022, 14(18), 21007–21017, DOI:10.1021/acsami.2c02668.
- S. J. Armaković and S. Armaković, Zeolite-Supported TiO2 for Enhanced Photocatalytic Performance in Environmental Applications: A Review, Catalysts, 2025, 15(2), 174, DOI:10.3390/catal15020174.
- K. Zhang, C. Li, L. Xu, Z. He, Z. Zhang, J. Sumita, J. A. Ghumro and K. Dong, MXene and Its Composites Combined with Photocatalytic Degradation of Perfluorooctanoic Acid: Efficiency and Active Species Study, Environ. Res., 2025, 278, 121690, DOI:10.1016/j.envres.2025.121690.
- H. Song, Y. Wang, Z. Ling, D. Zu, Z. Li, Y. Shen and C. Li, Enhanced Photocatalytic Degradation of Perfluorooctanoic Acid by Ti3C2 MXene-Derived Heterojunction Photocatalyst: Application of Intercalation Strategy in DESs, Sci. Total Environ., 2020, 746, 141009, DOI:10.1016/j.scitotenv.2020.141009.
- S. Halder, F.-C. Chou, T.-K. Wan, Y.-T. Tsai, P.-C. Cheng and Y.-C. Lin, Enhanced Photodegradation of Pentafluoropropionic Acid (PFPA) via TiO2–Synthetic Zeolite Composites: A Sustainable Approach for Effective Defluorination, J. Water Process Eng., 2024, 62, 105296, DOI:10.1016/j.jwpe.2024.105296.
- N. M. Chauke and M. Raphulu, A Review: Simultaneous “One-Pot” Pollution Mitigation and Hydrogen Production from Industrial Wastewater Using Photoelectrocatalysis Process, Mater. Today Catal., 2024, 5, 100052, DOI:10.1016/j.mtcata.2024.100052.
- C. Dong, W. Fang, Q. Yi and J. Zhang, A Comprehensive Review on Reactive Oxygen Species (ROS) in Advanced Oxidation Processes (AOPs), Chemosphere, 2022, 308, 136205, DOI:10.1016/j.chemosphere.2022.136205.
- A. Tufail, W. E. Price and F. I. Hai, A Critical Review on Advanced Oxidation Processes for the Removal of Trace Organic Contaminants: A Voyage from Individual to Integrated Processes, Chemosphere, 2020, 260, 127460, DOI:10.1016/j.chemosphere.2020.127460.
- N. Taoufik, W. Boumya, M. Achak, M. Sillanpää and N. Barka, Comparative Overview of Advanced Oxidation Processes and Biological Approaches for the Removal of Pharmaceuticals, J. Environ. Manage., 2021, 288, 112404, DOI:10.1016/j.jenvman.2021.112404.
- Y. Luo, X. Feng, Z. Chen and X. Shen, Molecularly Imprinted Photocatalysts: Fabrication, Application and Challenges, Mater. Adv., 2022, 3, 8830–8847, 10.1039/D2MA00848C.
- A. Kubiak, M. Jaruga, A. Lusina, T. Nazim, K. Sobańska, P. Pietrzyk and M. Cegłowski, Real-World Application of Molecularly Imprinted TiO2-Graphite Photocatalysts: Efficient Pharmaceutical Removal under Energy-Optimized LED System, J. Water Process Eng., 2025, 69, 106894, DOI:10.1016/j.jwpe.2024.106894.
- M. Guo, Y. Hu, R. Wang, H. Yu and L. Sun, Molecularly Imprinted Polymer-Based Photocatalyst for Highly Selective Degradation of Methylene Blue, Environ. Res., 2021, 194, 110684, DOI:10.1016/j.envres.2020.110684.
- J. Yu, J. González-Cobos, F. Dappozze, P. Vernoux, A. Caravaca and C. Guillard, Basic Comprehension and Recent Trends in Photoelectrocatalytic Systems, Green Chem, 2024, 26, 1682–1708, 10.1039/D3GC03371F.
- D. Liu, X. Wan, T. Kong, W. Han and Y. Xiong, Single-Atom-Based Catalysts for Photoelectrocatalysis: Challenges and Opportunities, J. Mater. Chem. A, 2022, 10, 5878–5888, 10.1039/D1TA08252C.
- P. Fernandez-Ibanez, S. McMichael, A. Rioja Cabanillas, S. Alkharabsheh, A. Tolosana Moranchel and J. A. Byrne, New Trends on Photoelectrocatalysis (PEC): Nanomaterials, Wastewater Treatment and Hydrogen Generation, Curr. Opin. Chem. Eng., 2021, 34, 100725, DOI:10.1016/j.coche.2021.100725.
- K. Park, I. Ali and J.-O. Kim, Photodegradation of Perfluorooctanoic Acid by Graphene Oxide-Deposited TiO2 Nanotube Arrays in Aqueous Phase, J. Environ. Manage., 2018, 218, 333–339, DOI:10.1016/j.jenvman.2018.04.016.
- S. S. Thind, B. A. Ryane, J. B. Hayden, I. Chagunda, M. Paul and J. S. McIndoe, Bias Enhanced Electro-Photocatalysis on TiO2 Nanoporous Materials for Decomposition of Forever Chemicals in Saltwater, Environ. Sci.: Adv., 2025, 4, 1024–1034, 10.1039/D4VA00423J.
- A. P. Tucci, S. Murgolo, C. De Ceglie, G. Mascolo, M. Carmagnani, P. Ronco, M. Bestetti and S. Franz, Photoelectrocatalytic Advanced Oxidation of Perfluoroalkyl Substances in Groundwaters of the Veneto Region, Italy, Catal. Today, 2025, 450, 115205, DOI:10.1016/j.cattod.2025.115205.
- A. Fernandes, P. Makoś, Z. Wang and G. Boczkaj, Synergistic Effect of TiO2 Photocatalytic Advanced Oxidation Processes in the Treatment of Refinery Effluents, Chem. Eng. J., 2020, 391, 123488, DOI:10.1016/j.cej.2019.123488.
- F. Tanos, A. Razzouk, G. Lesage, M. Cretin and M. Bechelany, A Comprehensive Review on Modification of Titanium Dioxide-Based Catalysts in Advanced Oxidation Processes for Water Treatment, ChemSusChem, 2024, 17, e202301139, DOI:10.1002/cssc.202301139.
- A. H. Navidpour, S. Abbasi, D. Li, A. Mojiri and J. L. Zhou, Investigation of Advanced Oxidation Process in the Presence of TiO2 Semiconductor as Photocatalyst: Property, Principle, Kinetic Analysis, and Photocatalytic Activity, Catalysts, 2023, 13, 232, DOI:10.3390/catal13020232.
- A. Fazli, A. Athanassiou and D. Fragouli, Current Approaches and Challenges in Advanced Oxidation Processes for Nanoplastic Degradation, Adv. Sci., 2025, 12, e04352, DOI:10.1002/advs.202504352.
- A. Šuligoj, M. Kete, U. Černigoj, F. Fresno and U. Lavrenčič Štangar, Synergism in TiO2 Photocatalytic Ozonation for the Removal of Dichloroacetic Acid and Thiacloprid, Environ. Res., 2021, 197, 110982, DOI:10.1016/j.envres.2021.110982.
- M. A. Zazouli, M. Yousefi, F. Ghanbari and E. Babanezhad, Performance of Photocatalytic Ozonation Process for Pentachlorophenol (PCP) Removal in Aqueous Solution Using Graphene-TiO2 Nanocomposite (UV/G-TiO2/O3), J. Environ. Health Sci. Eng., 2020, 18(2), 1083–1097, DOI:10.1007/s40201-020-00529-1.
- J. Huang, X. Wang, Z. Pan, X. Li, Y. Ling and L. Li, Efficient Degradation of Perfluorooctanoic Acid (PFOA) by Photocatalytic Ozonation, Chem. Eng. J., 2016, 296, 329–334, DOI:10.1016/j.cej.2016.03.116.
- B. Xu, M. B. Ahmed, J. L. Zhou and A. Altaee, Visible and UV Photocatalysis of Aqueous Perfluorooctanoic Acid by TiO2 and Peroxymonosulfate: Process Kinetics and Mechanistic Insights, Chemosphere, 2020, 243, 125366, DOI:10.1016/j.chemosphere.2019.125366.
- A. K. Venkatesan, C.-S. Lee and C. J. Gobler, Hydroxyl-Radical Based Advanced Oxidation Processes Can Increase Perfluoroalkyl Substances beyond Drinking Water Standards: Results from a Pilot Study, Sci. Total Environ., 2022, 847, 157577, DOI:10.1016/j.scitotenv.2022.157577.
- B. Lashuk, M. Pineda, S. AbuBakr, D. Boffito and V. Yargeau, Application of Photocatalytic Ozonation with a WO3/TiO2 Catalyst for PFAS Removal under UVA/Visible Light, Sci. Total Environ., 2022, 843, 157006, DOI:10.1016/j.scitotenv.2022.157006.
- S. Satyam and S. Patra, Innovations and Challenges in Adsorption-Based Wastewater Remediation: A Comprehensive Review, Heliyon, 2024, 10(9), e29573, DOI:10.1016/j.heliyon.2024.e29573.
- B. G. Fouda-Mbanga, O. P. Onotu and Z. Tywabi-Ngeva, Advantages of the Reuse of Spent Adsorbents and Potential Applications in Environmental Remediation: A Review, Green Anal. Chem., 2024, 11, 100156, DOI:10.1016/j.greeac.2024.100156.
- S. Sanzana, A. Fenti, P. Iovino and A. Panico, A Review of PFAS Remediation: Separation and Degradation Technologies for Water and Wastewater Treatment, J. Water Process Eng., 2025, 74, 107793, DOI:10.1016/j.jwpe.2025.107793.
- J. Wang, M. Zhu, A. Sun, R. Yuan, H. Chen and B. Zhou, The Influence Mechanism of Dissolved Organic Matter on the Photocatalytic Oxidation of Pharmaceuticals and Personal Care Products, Molecules, 2025, 30, 2266, DOI:10.3390/molecules30112266.
- A. Fattahi, I. Jaciw-Zurakowsky, N. Srikanthan, L. Bragg, R. Liang, N. Zhou, M. Servos and M. Arlos, Effect of Background Water Matrices on Pharmaceutical and Personal Care Product Removal by UV-LED/TiO2, Catalysts, 2021, 11, 576, DOI:10.3390/catal11050576.
- E. O. Anjorin, M. O. Alfred, B. Sotunde, E. A. Nnamani, A. A. Bayode, E. I. Unuabonah, B. Helmreich and M. O. Omorogie, Overview of the Mechanism of Degradation of Pharmaceuticals by Persulfate/Peroxysulfate Catalysts, ChemBioEng Rev, 2024, 11, e202300079, DOI:10.1002/cben.202300079.
- N. A. Samah, M.-J. Sánchez-Martín, R. M. Sebastián, M. Valiente and M. López-Mesas, Molecularly Imprinted Polymer for the Removal of Diclofenac from Water: Synthesis and Characterization, Sci. Total Environ., 2018, 631–632, 1534–1543, DOI:10.1016/j.scitotenv.2018.03.087.
- M. Cantarella, S. C. Carroccio, S. Dattilo, R. Avolio, R. Castaldo, C. Puglisi and V. Privitera, Molecularly Imprinted Polymer for Selective Adsorption of Diclofenac from Contaminated Water, Chem. Eng. J., 2019, 367, 180–188, DOI:10.1016/j.cej.2019.02.146.
- Y. Wu, Y. Li, A. Tian, K. Mao and J. Liu, Selective Removal of Perfluorooctanoic Acid Using Molecularly Imprinted Polymer-Modified TiO2 Nanotube Arrays, Int. J. Photoenergy, 2016, 7368795, DOI:10.1155/2016/7368795.
- L. Hu, Y. Li and W. Zhang, Characterization and Application of Surface-Molecular-Imprinted-Polymer Modified TiO2 Nanotubes for Removal of Perfluorinated Chemicals, Water Sci. Technol, 2016, 74(6), 1417–1425, DOI:10.2166/wst.2016.321.
- A. D. McQueen, O. Tedrow and M. L. Ballentine, et al., Demonstration of Photocatalytic Degradation of Per- and Polyfluoroalkyl Substances (PFAS) in Landfill Leachate Using 3D Printed TiO2 Composite Tiles, Water Air Soil Pollut., 2022, 233, 444, DOI:10.1007/s11270-022-05911-3.
- Z. Khalid, F. Hadi, J. Xie, V. Chandrabose and J.-M. Oh, The Future of MXenes: Exploring Oxidative Degradation Pathways and Coping with Surface/Edge Passivation Approach, Small, 2025, 21, 2407856, DOI:10.1002/smll.202407856.
- J. Brame, M. Long, Q. Li and P. Alvarez, Inhibitory Effect of Natural Organic Matter or Other Background Constituents on Photocatalytic Advanced Oxidation Processes: Mechanistic Model Development and Validation, Water Res, 2015, 84, 362–371, DOI:10.1016/j.watres.2015.07.044.
- I. Y. Qudsieh, M. A. Ali and I. M. Maafa, Effect of Water Matrix on Photocatalytic Degradation of Organic Pollutants in Water: A Literature Review, Rev. Chem. Eng., 2025, 41(6), 539–573, DOI:10.1515/revce-2024-0093.
- S. A. Younis and K.-H. Kim, Heterogeneous Photocatalysis Scalability for Environmental Remediation: Opportunities and Challenges, Catalysts, 2020, 10(10), 1109, DOI:10.3390/catal10101109.
- E. H. Khader, S. A. Muslim, N. M. C. Saady, N. S. Ali, I. K. Salih, T. J. Mohammed, T. M. Albayati and S. Zendehboudi, Recent Advances in Photocatalytic Advanced Oxidation Processes for Organic Compound Degradation: A Review, Desalin. Water Treat., 2024, 318, 100384, DOI:10.1016/j.dwt.2024.100384.
- N. Gaur, D. Dutta, A. Singh, R. Dubey and D. V. Kamboj, Recent Advances in the Elimination of Persistent Organic Pollutants by Photocatalysis, Front. Environ. Sci., 2022, 10, 872514, DOI:10.3389/fenvs.2022.872514.
- P. Kumari and A. Kumar, Advanced Oxidation Process: A Remediation Technique for Organic and Non-Biodegradable Pollutants, Results Surf. Interfaces, 2023, 11, 100122, DOI:10.1016/j.rsurfi.2023.100122.
- W.-L. Wang, Q.-Y. Wu, N. Huang, Z.-B. Xu, M.-Y. Lee and H.-Y. Hu, Potential Risks from UV/H2O2 Oxidation and UV Photocatalysis: A Review of Toxic, Assimilable, and Sensory-Unpleasant Transformation Products, Water Res, 2018, 141, 109–125, DOI:10.1016/j.watres.2018.05.005.
- T. Dong, H. Y. M. Cheung and J. H. K. Man, et al., Recent Advances in Oxidant-Involved Photoelectrochemical Systems for Sustainable Wastewater Treatment: Mechanisms, Applications, and Perspectives, npj Mater. Sustainability, 2025, 3, 41, DOI:10.1038/s44296-025-00084-6.
- K. Wetchakun, N. Wetchakun and S. Sakulsermsuk, An Overview of Solar/Visible Light-Driven Heterogeneous Photocatalysis for Water Purification: TiO2- and ZnO-Based Photocatalysts Used in Suspension Photoreactors, J. Ind. Eng. Chem., 2019, 71, 19–49 Search PubMed.
- R. Binjhade, R. Mondal and S. Mondal, Continuous Photocatalytic Reactor: Critical Review on the Design and Performance, J. Environ. Chem. Eng., 2022, 10(3), 107746, DOI:10.1016/j.jece.2022.107746.
- S. Chang, X. Yang, Y. Sang and H. Liu, Highly Efficient Photocatalysts and Continuous-Flow Photocatalytic Reactors for Degradation of Organic Pollutants in Wastewater, Chem. Asian J., 2016, 11, 2352–2363 CrossRef CAS PubMed.
- S. Basha, A. Vijeev, K. S. Pranavi, A. Chattopadhyay, A. R. Pai and K. K. Mahato, Ultraviolet Light-Emitting Diode Technologies in Water Disinfection, Water Res. X, 2025, 29, 100454, DOI:10.1016/j.wroa.2025.100454.
- Z. U. Zango, K. H. Ibnaouf, A. Haruna, M. A. Lawal, I. A. Wadi, M. A. Ibrahem, F. B. Shittu and H. Adamu, Toward Sustainable PFAS Elimination: Engineering TiO2 Photocatalysts for Complete Mineralization and Defluorination, J. Sci.: Adv. Mater. Devices, 2025, 10(4), 101041, DOI:10.1016/j.jsamd.2025.101041.
| This journal is © The Royal Society of Chemistry 2026 |




