Open Access Article
Open Access ArticleSelf-assembled α/β-dipeptides as dielectrics to improve the behavior of multilayered conducting polymer pseudocapacitors
Dulce A.
Quintana-Romero
ab,
Adrián
Fontana-Escartín
*ac,
Maria Luisa
Gelmi
 *c,
Merve
Gul
ac,
Maria M.
Pérez-Madrigal
*c,
Merve
Gul
ac,
Maria M.
Pérez-Madrigal
 ac,
Raffaella
Bucci
ac,
Raffaella
Bucci
 *b and
Carlos
Alemán
*b and
Carlos
Alemán
 *acd
*acd
aIMEM-BRT Group, Departament d’Enginyeria Química, EEBE, Universitat Politècnica de Catalunya. BarcelonaTech (UPC), Av. Eduard Maristany, 16, Barcelona 08019, Spain. E-mail: adrian.fontana@upc.edu; carlos.alema@upc.edu
bDepartment of Pharmaceutical Sciences, University of Milan, via Venezian 21, Milano 20133, Italy. E-mail: raffaella.bucci@unimi.it
cBarcelona Research Centre in Multiscale Science and Engineering (CCEM), Universitat Politècnica de Catalunya. BarcelonaTech (UPC), Av. Eduard Maristany, 16, Barcelona 08019, Spain. E-mail: marialuisa.gelmi@unimi.it
dInstitute for Bioengineering of Catalonia (IBEC), The Barcelona Institute of Science and Technology, Baldiri Reixac 10-12, 08028 Barcelona, Spain
First published on 2nd March 2026
Abstract
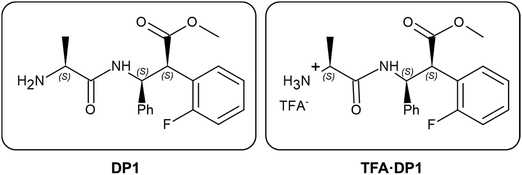
Multilayered films composed of two different materials, where the less conducting one acts as a dielectric separating the layers made of the other, are known to function as efficient electrodes for electrochemical supercapacitors. In this work, we extend this concept, which was previously applied only to soft supercapacitors made from two or more conducting polymers with markedly different electrochemical activities, to examine for the first time the utilization of α/β-dipeptides in conducting polymer electrodes for symmetric supercapacitors. Specifically, we investigate the α/β-dipeptides formed by L-alanine and syn-3-amino-2-(2-fluorophenyl)-3-phenylpropanoic acid in two forms: (1) an α/β-dipeptide with –NH2 as the free terminal amino group; and (2) an α/β-dipeptide with a protonated amino group stabilized through an ionic pair with trifluoroacetate (TFA·DP1). After examining the self-assembly of DP1 and TFA·DP1 on electropolymerized poly(3,4-ethylenedioxythiophene) (PEDOT) films, we fabricated three-layered electrodes consisting of two PEDOT layers separated by an intermediate layer of self-assembled DP1 or TFA·DP1, and subsequently characterized them. The best electrochemical response was achieved with the TFA·DP1-containing three-layered electrodes. These electrodes were then used to assemble symmetric supercapacitors, which were characterized by cyclic voltammetry, galvanostatic charge–discharge analysis and electrochemical impedance spectroscopy. The specific capacitance of devices incorporating TFA·DP1 between submicrometric PEDOT layers, which performed better than those in which the self-assembled peptide was omitted, was around 47 mF cm−2. In these optimized devices, the intermediate TFA-DP1 assemblies act as ultrathin, randomly distributed dielectrics connected in series with one another and in parallel with the PEDOT layers.
Introduction
Over the past fifteen years, significant efforts have been made to develop soft electrochemical energy storage devices, such as batteries and supercapacitors (SCs), in a simple and cost-effective manner.1–4 SCs are considered efficient electrochemical energy storage devices for power drifting applications due to their excellent power density, moderate energy density, ultrafast charge/discharge rates and long cycle life.5–7 Based on the type of charge storage exhibited by the SC electrodes, SCs are classified into three types:8,9 (1) electrochemical double layer capacitors (EDLCs), which store charge via a charge–discharge process (electro-sorption) in an electric double layer on porous electrodes (e.g. activated carbons and carbon nanotubes); (2) pseudocapacitors, which store charge by reversible faradaic redox reactions (e.g. transition metal oxides and electronically conducting polymers); and (3) battery-type hybrid capacitors, which combine the benefits of an EDLC and a pseudocapacitor to provide higher energy and power densities.Among the conjugated conducting polymers used to fabricate electrochemical pseudocapacitors, poly(3,4-ethylenedioxythiophene) (PEDOT) has been extensively employed. This is due to its excellent pseudocapacitance, which derives from the following features: its superior ionic charge mobility, outstanding electrical conductivity, fast and reversible redox reactions coupled with doping/de-doping processes, exceptional thermal and chemical stability, and resilience to expansion and contraction during electrochemical reactions.10–14 Indeed, PEDOT is frequently used in combination with other electrochemically active materials (e.g. clays,15,16 other conducting polymers,17,18 graphene,19,20 and MXenes21,22) to obtain effective hybrid and composite electrodes. The engineering of these complex electrodes involves the underlying use and sometimes even the explicit search for synergy between the materials being combined.23
On the other hand, peptide-based materials, which are exploited for their ability to self-assemble into well-shaped three-dimensional micro- and nanostructures, offer a variety of potential applications in environmentally friendly energy storage technologies. Thus, different α-peptide materials have been used to fabricate SCs taking advantage of the versatility of peptide sequences and various peptide self-assembly pathways.24–32 Due to the difficulties in the scalability of their synthesis, peptides are usually combined with other materials, forming hybrid or multilayered composite materials. For example, Hu et al.32 sandwiched Fmoc-Glu-Phe-NH2 dipeptide fibers coated with a TiO2 layer and a polyvinyl alcohol (PVA)/H3PO4 gel electrolyte between two polyethylene terephthalate (PET) films to obtain an outstanding areal capacitance of 8.6 mF cm−2.
In this work, we go one step forward investigating for the first time the utilization of α/β-dipeptides in electrodes for pseudocapacitors. More specifically, we have considered dipeptides involving L-alanine (L-Ala) and syn-3-amino-2-(2-fluorophenyl)-3-phenylpropanoic acid (β-Fpg), which are expected to exhibit well-shaped self-assemblies due to the aromatic aryl groups, the lipophilic fluorine atom, and the extended conformation typically driven by syn-β2,3-amino acids.33,34 Indeed, the protected α/β-dipeptide Boc-L-Ala-β-2S,3S-Fpg-OMe was reported to self-assemble into well-defined nanotubes stabilized by intermolecular hydrogen bonds, π–π stacking, as well as Cπ–H⋯O and Cπ–H⋯F interactions.35 Moreover, when the same dipeptide is further functionalized with a cationic construct the same interactions promote the formation of spherical structures.36 More recently, we also investigated the electrochemical behavior of peptidomimetics incorporating the β-2S,3S-Fpg residue. Our findings highlighted the importance of forming highly ordered nanomaterials, which were drop-cast onto PEDOT substrates to enhance electrochemical performance.37 In order to enhance the self-assembly capacity and the electrochemical response, both the formation of intermolecular electrostatic interactions and ionic transport have been promoted by selecting both the N-unprotected dipeptide (DP1 in Scheme 1) and the corresponding peptide protonated at the terminal amino group stabilized by forming an ionic pair with trifluoroacetate (TFA·DP1 in Scheme 1). Accordingly, the charge transport in the protonated peptide is expected to arise not only from the electron transfer flowing through stacked aromatic rings of β-2S,3S-Fpg, like in the neutral peptide, but also from the proton and/or counterion transport.
More specifically, herein, we present multilayer composite electrodes for pseudocapacitors that have been obtained by promoting the self-assembly of DP1 and TFA·DP1 between two anodically polymerized PEDOT films (Scheme 2). For this purpose, the self-assembly of the peptides, which have been drop cast on the surface of a PEDOT substrate, has been investigated considering not only the conditions used to dissolve the peptides (i.e. solvent mixtures and concentrations) but also the roughness of the PEDOT film. In this work, the roughness of PEDOT films has been tuned by varying the polymerization time (tp), which also affects the film thickness. The resulting peptide-coated PEDOT films (PEDOT(#) + DP1 and PEDOT(#) + TFA·DP1, where # refers to tp) have been covered by adding another layer of anodically polymerized PEDOT, which has been prepared using a tp identical to that of the first layer. Hereafter, the resulting 3-layered electrodes have been denoted 2PEDOT(#)-DP1 and 2PEDOT(#)-TFA·DP1. After evaluating the electrochemical behavior of the fabricated electrodes, those with better properties have been selected as electrodes for constructing SCs. The performance and stability of such devices have been analyzed by cyclic voltammetry (CV), galvanostatic charge–discharge (GCD) analysis and electrochemical impedance spectroscopy (EIS). Results indicate that electrochemical symmetric SCs prepared using 3-layered electrodes made of TFA·DP1 peptide assemblies located between two nano-structured PEDOT layers behave excellently and are very promising for use in wearable and implantable bioelectronic devices.
Methods
Peptide characterization
This work focusses on an α/β-dipeptide, which is based on the L-Ala-β-Fpg sequence with a specific stereochemistry of the β2,3-diaryl-amino acid. The protected dipeptide was prepared according to a known procedure35 from L-Ala and syn-3-amino-2-(2-fluorophenyl)-3-phenylpropanoic acid.The procedure used for the N-terminal deprotection of the dipeptide is available in the literature as well as the characterization data for the free dipeptide (DP1).36 The protonated form, TFA·DP1, was obtained directly by evaporating the reaction mixture without performing any extraction steps.
Since TFA·DP1 characterization is not present in the literature, the NMR and MS (ESI) data are reported below, as well as the spectra in the SI.
TFA·DP1: 1H NMR (300 MHz, CD3OD): δ 7.66–7.60 (m, 1H), 7.50–7.44 (m, 2H), 7.42–7.28 (m, 4H), 7.25–7.18 (m, 1H), 7.18–7.10 (m, 1H), 5.71 (d, J = 11.7, 1H), 4.67 (d, J = 11.7, 1H), 3.68–3.58 (m, 1H), 3.44 (s, 3H), 1.22 (d, J = 6.76, 3H).
13C NMR (75 MHz, CDCl3): δ = 15.8, 48.3 (d, J = 2.4), 48.4, 51.2, 54.5, 115.1 (d, J = 23.3), 122.5 (d, J = 14.2), 124.3 (d, J = 3.6), 127.2, 127.8, 128.3, 129.2 (d, J = 3.1), 129.5 (d, J = 8.5), 139.8, 161.1 (d, J = 245.3), 171.0, 172.1.
19F NMR (282 MHz, CDCl3): δ = 77.0 (s, 3F, CF3COO−), −119.3 to −119.4 (m, 1F, FPh).
MS (ESI): m/z calcd for [C19H21FN2O3]: 344.15; found: m/z 345.8 [M+H]+.
PEDOT polymerization
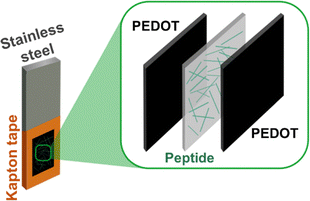
Steel sheets (1.0 × 2.0 cm2) were sanded and washed with Milli-Q® water, ethanol and acetone in an ultrasonic bath. Cleaned steel samples were covered with Kapton® tape leaving a central 0.5 × 0.5 cm2 window (Scheme 2). The steel sheet was immersed in a three-electrode electrochemical cell with 10 mL of acetonitrile solution containing 50 µM of 3,4-ethylenedioxythiophene (EDOT) monomer and 0.1 M of LiClO4 as a supporting electrolyte. The electrochemical setup was completed with the counter and the reference electrodes, which were a stainless-steel sheet of 10.0 × 1.0 cm2 and Ag|AgCl in 3 M KCl, respectively. Then, a constant potential of 1.2 V was applied for 60 s to stabilize the system and, subsequently, potential sweeps between –0.5 V and +1.4 V were conducted at a scan rate of 50 mV s−1 for 3 min or 15 s. The resulting films were denoted PEDOT(3m) and PEDOT(15s), respectively.Once the peptide was self-assembled on the PEDOT(3m) and PEDOT(15s) films generated using the procedure described above, the system was coated with a second PEDOT layer that was electrogenerated using as an electrolytic medium of 0.1 M LiClO4 aqueous solution in which 50 µM of EDOT monomer was dissolved. The electrochemical setup was identical to that described above but the operational parameters were adapted to the new polymerization medium. More specifically, a potential of 1.0 V was applied for 60 s to stabilize the system and, subsequently, potential sweeps between –0.5 V and +1.0 V were conducted at a scan rate of 50 mV s−1 for 3 min or 15 s (i.e. in all cases, the tp used for the layer prepared in aqueous solution was identical to the one used for the layer obtained in acetonitrile). All electrochemical polymerizations were performed at room temperature.
Peptide colloidal suspension
Peptide colloidal suspension (100 µL) was prepared from 100 mg mL−1 stocks using hexafluoroisopropanol (HFIP) as a solvent. The peptide concentration was adjusted by adding Milli-Q water (H2O), ethanol (EtOH) or more HFIP, as a co-solvent in the necessary volumes. This choice was based on the utilization of biocompatible solvents, which is an intrinsic property of both PEDOT and peptides, as has been extensively reported in the literature. More specifically, peptide concentrations of 8, 4, 2 and 0.5 mg mL−1 were studied using HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O and HFIP
H2O and HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH mixtures with 98
EtOH mixtures with 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 20
2, 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80, 40
80, 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60, 60
60, 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 and 80
40 and 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratios (in%). These ratios were chosen to prospect the whole range of mixtures, while the solubility of the peptides is preserved. Finally, 100 µL aliquots were placed on PEDOT substrates and stored at room temperature until dryness. The dielectric constant and the boiling point (ε and Tb, respectively) of the used solvents are ε = 16.7 and Tb = 59 °C for HFIP, ε = 24.3 and Tb = 78.2 °C for EtOH, and ε = 78.5 and Tb = 100 °C for H2O. Accordingly, the polarity of HFIP:H2O and HFIP:EtOH mixtures increased with the content of H2O and EtOH, respectively, while their volatility decreased.
20 ratios (in%). These ratios were chosen to prospect the whole range of mixtures, while the solubility of the peptides is preserved. Finally, 100 µL aliquots were placed on PEDOT substrates and stored at room temperature until dryness. The dielectric constant and the boiling point (ε and Tb, respectively) of the used solvents are ε = 16.7 and Tb = 59 °C for HFIP, ε = 24.3 and Tb = 78.2 °C for EtOH, and ε = 78.5 and Tb = 100 °C for H2O. Accordingly, the polarity of HFIP:H2O and HFIP:EtOH mixtures increased with the content of H2O and EtOH, respectively, while their volatility decreased.
Characterization
Electrochemical characterization of symmetrical supercapacitors
Symmetrical SCs, which have been prepared as described in the next section, were characterized by CV, galvanostatic charge–discharge (GCD) analysis and electrochemical impedance spectroscopy (EIS). At least three independent replicas were performed for all experiments (n ≥ 3).The loss of electrochemical activity (LEA, in %) against the increasing number of voltammetric cycles was expressed as:
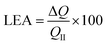 | (1) |
The specific capacitance (sc; in F cm−2) of the conducting polymer in the electrodes was calculated as:
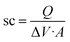 | (2) |
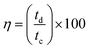 | (3) |
Results and discussion
Preparation and characterization of the PEDOT substrate
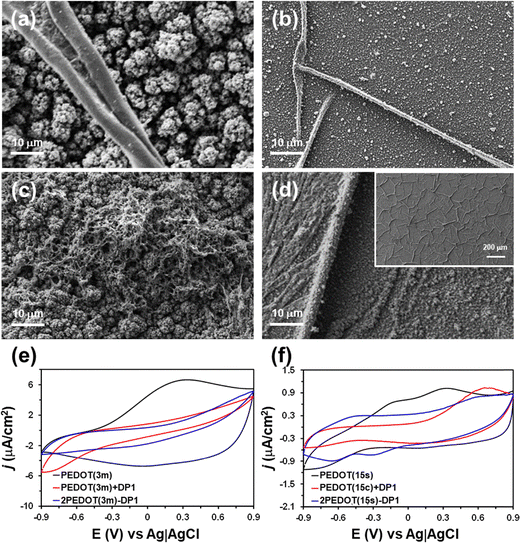
Fig. 1 shows representative SEM micrographs of the morphologies found at the surface of PEDOT films electrodeposited using acetonitrile as a solvent and tp = 3 min and 15 s, hereafter denoted PEDOT(3m) and PEDOT(15s). These values of tp were selected to obtain films with very different surface morphologies and thicknesses. PEDOT(3m) displayed a globular morphology formed by the aggregation of micrometric globules (Fig. 1a), thus evidencing a three-dimensional (3D) growth. Conversely, PEDOT(15s) films showed a significantly flatter morphology with a dense packing of nanometric particles, which was closer to two-dimensional (2D) growth (Fig. 1b). These results indicated the formation of multiple nucleation centers during the first steps of the electropolymerization process, giving rise to a relatively flat film. After that, there was preferential growth over some nucleation centers, probably due to steric effects, resulting in the formation of nanometric particles first (tp = 15 s) and micrometric globules later (tp = 3 min). The morphological differences between PEDOT(3m) and PEDOT(15s) were expected to play a key role in peptide self-assembly, since these substrates act as templates during this process. | ||
| Fig. 1 Representative SEM images of PEDOT films prepared in (a and b) acetonitrile and (c and d) water using a tp of (a and c) 3 min and (b and d) 15 s. | ||
Other crucial parameters for understanding both the peptide self-assembly and the electrode performance are the thicknesses (L) and the Rq of the films. These were determined using profilometry (for all films) and AFM scratching technology (for PEDOT(15s) films, since the roughness was too high for PEDOT(3m)), the latter having a higher level of accuracy due to its precise tip and lower loading force (Fig. S1). In addition to the good agreement between profilometry and AFM scratching, results (Table 1) confirmed that both L and Rq were much higher for PEDOT(3m) than for PEDOT(15s).
| Solvent | Technique | PEDOT(3m) | PEDOT(15s) | ||
|---|---|---|---|---|---|
| L | R q | L | R q | ||
| Acetonitrile | Profilometry | 8.4 ± 2.4 µm | 4.7 ± 1.6 µm | 1.4 ± 0.3 µm | 251 ± 41 nm |
| AFM | — | — | 1.1 ± 0.4 µm | 299 ± 72 nm | |
| Water | Profilometry | 3.4 ± 1.0 µm | 1.4 ± 0.4 µm | 0.6 ± 0.1 µm | 300 ± 51 nm |
| AFM | — | — | 0.4 ± 0.1 µm | 228 ± 59 nm | |
Although they were not used as templates in the self-assembly of the peptides, to better understand the functioning of the 3-layered electrodes, L and Rq were also determined for the PEDOT(3m) and PEDOT(15s) films electrogenerated in water. The values of L and Rq obtained for films produced using the same tp were significantly lower in water than in acetonitrile. These observations indicated that the polymerization rate relative to deposition charge density of PEDOT synthesized in acetonitrile was 2.5 (tp = 3 min) and 4.5 (tp = 15 s) times higher than that achieved in aqueous solution. These observations were fully consistent with data reported in the literature.38 On the other hand, SEM images showed that the cauliflower structure of PEDOT aggregates in PEDOT(3m) films was much less pronounced in water (Fig. 1c), while the morphology of PEDOT(15s) was less homogeneous in water (Fig. 1d) than in acetonitrile. These differences were attributed to the different solubility of EDOT oligomers produced during the initial stages of electropolymerization in these solvents.39
Optimizing the assembly of DP1 on PEDOT
The self-assembly of DP1 was examined using a bottom-up approach in which the concentrations and solvents listed in Table S1 were considered (labeled as conditions A–T). Initially, all experiments were conducted using PEDOT(3m) substrates. Assays with a dilute peptide concentration (0.5 mg mL−1) using HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O and HFIP
H2O and HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH mixtures (conditions A–D in Table S1) evidenced that DP1 aggregation was favored when the polarity and volatility of the medium decreased and increased, respectively (i.e. when the content of HFIP in the mixtures increased from 2% to 20%). Despite these changes, the amount of peptide was not high enough to promote the formation of fibrillary or regularly shaped structures, as shown in Fig. S2.
EtOH mixtures (conditions A–D in Table S1) evidenced that DP1 aggregation was favored when the polarity and volatility of the medium decreased and increased, respectively (i.e. when the content of HFIP in the mixtures increased from 2% to 20%). Despite these changes, the amount of peptide was not high enough to promote the formation of fibrillary or regularly shaped structures, as shown in Fig. S2.
Optical micrographs recorded for assemblies derived from 2 mg mL−1 peptide solutions (conditions E–N in Table S1), which are displayed in Fig. S3, confirmed the importance of the peptide concentration, as well as of the polarity and volatility of the medium. When the polarity and volatility of the medium were low and high, respectively, unshaped peptide aggregates were obtained (conditions E–H), while when there was a balance between polarity and volatility with 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 and 40
40 and 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 ratios (conditions I–K), well-shaped microfibers were formed. Finally, when the volatility was very high (conditions L–N), the peptide acted as a coating covering the whole PEDOT surface.
60 ratios (conditions I–K), well-shaped microfibers were formed. Finally, when the volatility was very high (conditions L–N), the peptide acted as a coating covering the whole PEDOT surface.
In order to favor the formation of more abundant and bigger fibers, the concentration of DP1 was increased to 4 mg mL−1, while the solvent![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) co-solvent ratios were maintained at 40
co-solvent ratios were maintained at 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 and 60
60 and 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 (conditions O–R in Table S1). Results displayed in Fig. S4 show that the peptide preserved well-defined and shaped fibers only for HFIP:H2O mixtures, while unstructured heterogeneous aggregates that partially coated the PEDOT surface were derived from HFIP:EtOH mixtures. Thus, the polarity and volatility of HFIP:EtOH, which were lower and higher, respectively, than those of HFIP:H2O for the same ratio, did not allow the formation of fibers when peptide molecules are in excess. Instead, increasing the peptide concentration had a positive effect on the self-assembly to form fibers when the medium is 60
40 (conditions O–R in Table S1). Results displayed in Fig. S4 show that the peptide preserved well-defined and shaped fibers only for HFIP:H2O mixtures, while unstructured heterogeneous aggregates that partially coated the PEDOT surface were derived from HFIP:EtOH mixtures. Thus, the polarity and volatility of HFIP:EtOH, which were lower and higher, respectively, than those of HFIP:H2O for the same ratio, did not allow the formation of fibers when peptide molecules are in excess. Instead, increasing the peptide concentration had a positive effect on the self-assembly to form fibers when the medium is 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 and 40
40 and 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 HFIP
60 HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O.
H2O.
Three-layered DP1-containing electrode manufacturing
At this stage, 8 mg mL−1DP1 solution in 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 HFIP
60 HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (condition S) was selected to construct three-layered electrodes, which were prepared using both PEDOT(3m) and PEDOT(15s) films. Fig. 2a and b display representative SEM micrographs of the fibers formed on two such substrates by dropping 100 µL of the prepared DP1 solution. The thickness of the formed fibers was much higher for the roughest surface (i.e. fibers up to 10 µm thick were observed on PEDOT(3m), while the average thickness of the fibers obtained on PEDOT(15s) was 1–2 µm). Interestingly, Shezad et al.40 reported that a rough surface decelerates the two-dimensional diffusion of peptides on the surface, which causes a retardation of the kinetic pathway for the formation of new nuclei and fibers. However, this only applied when extremely flat polymeric surfaces were compared (i.e. Rq of the polymeric substrates studied by Shezad et al.40 ranged between 0.26 and 1.81 nm). The Rq of the two PEDOT surfaces examined in this work (Table 1) was incommensurately large relative to those examined by Shezad et al.,40 suggesting that such observation is not valid at the micrometric scale. Indeed, results displayed in Fig. 2a and b suggest that the inter-peptide association is more favored for the PEDOT(3m) substrate than for the PEDOT(15s) one. Thus, in the former PEDOT substrate the enthalpic contribution dominated and accelerated the aggregation process, whereas in the latter one the entropic contribution competed with the enthalpic one.
H2O (condition S) was selected to construct three-layered electrodes, which were prepared using both PEDOT(3m) and PEDOT(15s) films. Fig. 2a and b display representative SEM micrographs of the fibers formed on two such substrates by dropping 100 µL of the prepared DP1 solution. The thickness of the formed fibers was much higher for the roughest surface (i.e. fibers up to 10 µm thick were observed on PEDOT(3m), while the average thickness of the fibers obtained on PEDOT(15s) was 1–2 µm). Interestingly, Shezad et al.40 reported that a rough surface decelerates the two-dimensional diffusion of peptides on the surface, which causes a retardation of the kinetic pathway for the formation of new nuclei and fibers. However, this only applied when extremely flat polymeric surfaces were compared (i.e. Rq of the polymeric substrates studied by Shezad et al.40 ranged between 0.26 and 1.81 nm). The Rq of the two PEDOT surfaces examined in this work (Table 1) was incommensurately large relative to those examined by Shezad et al.,40 suggesting that such observation is not valid at the micrometric scale. Indeed, results displayed in Fig. 2a and b suggest that the inter-peptide association is more favored for the PEDOT(3m) substrate than for the PEDOT(15s) one. Thus, in the former PEDOT substrate the enthalpic contribution dominated and accelerated the aggregation process, whereas in the latter one the entropic contribution competed with the enthalpic one.
It is well-known that multilayered electrodes made layer-by-layer by alternating compounds with markedly different electrochemical properties exhibit better performance than monolayered electrodes prepared using the same amount of a single electroactive material.18,41,42 This behavior, which was mainly studied using PEDOT layers separated by a less conductive intermediate layer (e.g. a less conductive polymer, like poly(N-methyl-pyrrole) and exfoliated clays), was attributed to the dielectric breakage induced by the latter. Inspired by such studies, another PEDOT layer was electrodeposited on the surface of the PEDOT film coated with DP1 fibers (Scheme 2). This second layer of PEDOT was electropolymerized using a LiCO4 aqueous solution as the electrolytic medium instead of a LiClO4 acetonitrile solution (see Methods section), which avoided dissolving the DP1 fibers in an organic solvent.
Fig. 2c and d displays SEM micrographs of the two resulting three-layered electrodes: (1) PEDOT(3m) electropolymerized using LiClO4 dissolved in acetonitrile + DP1 fibers + PEDOT(3m) electropolymerized using LiClO4 dissolved in water, hereafter named 2PEDOT(3m)-DP1; and (2) PEDOT(15s) electropolymerized using LiClO4 dissolved in acetonitrile + DP1 fibers + PEDOT(15s) electropolymerized using LiClO4 dissolved in water, hereafter named 2PEDOT(15s)-DP1. As can be seen, in both cases DP1 fibers were completely coated by the second PEDOT layer. However, a significant difference was found between the two electrodes. While the thickness of the second PEDOT layer was large enough to hide the intermediate DP1 fibers in 2PEDOT(3m)-DP1 (Fig. 2c), the shape of the DP1 fibers can be clearly recognized below the second layer of PEDOT in 2PEDOT(15s)-DP1 (Fig. 2d). Thus, the thin thickness of the second layer allowed the fibers to act as a template in the second electrode.
The thickness of 2PEDOT(3m)-DP1 and 2PEDOT(15s)-DP1 electrodes was 14.3 ± 3.1 µm and 4.9 ± 0.5 µm, as determined by profilometry. It is worth noting that these values were higher than those obtained as the sum of individual films polymerized in acetonitrile and in water (according to Table 1: 11.8 ± 3.4 and 2.0 ± 0.4 µm for tp = 3 min and 15 s, respectively). This effect was much more pronounced for 2PEDOT(15s)-DP1 in which the thickness of the film was more than twice the thickness of two individual films. It should be noted that, although this increase can initially be associated with the peptide fibers in the intermediate layer, the increase in thickness must be mainly attributed to the synergistic effect of the first PEDOT layer on the polymerization of the second layer. In order to demonstrate this, bilayer PEDOT systems were prepared, eliminating the intermediate peptide layer. The thickness of the resulting systems, called 2PEDOT(3m) and 2PEDOT(15s), was similar to those of 2PEDOT(3m) + DP1 and 2PEDOT(15s) + DP1 (i.e. the difference in the thickness of the bilayer and trilayer systems was smaller than 10%).
Finally, the electrochemical characterization was performed by CV using a potential range from –0.9 V to +0.9 V. Fig. 2e and f compares representative cyclic voltammograms recorded for: (1) PEDOT(3m) and PEDOT(15s) films without peptide; (2) the same films coated with peptide fibers, hereafter denoted PEDOT(3m) + DP1 and PEDOT(15s) + DP1, respectively; and (3) the three-layered electrodes, 2PEDOT(3m)-DP1 and 2PEDOT(15s)-DP1. As was expected, the electrochemical response increased with the amount of conducing polymer, being much higher for systems prepared using tp = 3 min than for systems obtained using tp = 15 s. Furthermore, the electrochemical activity, which is related to the voltammetric charge and, therefore, to the area of the voltammogram, is much higher for uncoated PEDOT films than for peptide-coated PEDOT + DP1 films, regardless of tp. This feature indicates that, as expected, the electrical response of the peptide is lower than that of PEDOT, suggesting that DP1 fibers can effectively induce a dielectric breakage between layers and improve the properties of PEDOT-based multilayered electrodes.
On the other hand, 2PEDOT(3m)-DP1 and 2PEDOT(15s)-DP1 electrodes displayed different behaviors. While the electroactivity of 2PEDOT(3m)-DP1 was even smaller than that of PEDOT(3m) + DP1, the electrochemical activity was greater for 2PEDOT(15s)-DP1 than for PEDOT(15s) + DP1. These results, together with the thickness of deposited fibers (i.e. up to 10 µm thick) and the Rq of PEDOT(3m) (Table 1), suggest that, probably, the second PEDOT(3m) layer is not in contact with the first one, which could explain the reduction in the electroactivity. Instead, the increase in electroactivity of 2PEDOT(15s)-DP1 with respect to PEDOT(15s) + DP1 suggests a good coupling between successive layers. However, the favorable inter-connection between the two thin conducting polymer layers is locally interrupted by DP1 peptide fibers distributed in the intermediate layer. Although aromatic-rich DP1 fibers exhibit electrochemical response, the electrical response of the peptide fibers is clearly lower than that of PEDOT layers and, therefore, the electrical behaviors of DP1/DP1 and PEDOT/DP1 interfaces in 2PEDOT(15s)-DP1 are expected to be very different. By similarity with previously reported multilayered electrodes, these interfaces have been inferred as series and parallel connections, respectively, while the peptide fibers forming the intermediate layer are considered as micro-dielectrics with respect to PEDOT layers. Accordingly, these micro-dielectric elements are interconnected in series while they form parallel interconnections with PEDOT(15s) layers. Nevertheless, from a practical point of view, the electrochemical activities of 2PEDOT(3m)-DP1 and 2PEDOT(15s)-DP1 electrodes were lower than those of PEDOT(3m) and PEDOT(15s). Therefore, the DP1 peptide was discarded for the preparation of electrodes for energy storage applications.
Optimizing the self-assembly of TFA·DP1 on PEDOT
The strategy used to explore the self-assembly of TFA·DP1 on PEDOT(3m) was similar to that described above for DP1. In this case, the peptide concentrations and solvent mixtures (conditions a–k in Table S2) were selected considering both the results obtained for DP1 and the solubility of TFA·DP1. When the TFA·DP1 concentration was as low as 0.5 mg mL−1 and the HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O ratio was varied from 2
H2O ratio was varied from 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 to 80
98 to 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 (conditions a–c in Table S2), poorly defined fibrillary structures could be observed when the volatility of the solvent:co-solvent mixture was the highest (Fig. S5). No significant improvement was observed when the peptide concentration was increased to 2 mg mL−1, or even when the volatility of the medium was enhanced by replacing water by EtOH (Fig. S6; conditions d–i in Table S2). After several tests and increasing the peptide concentration to 4 mg mL−1, we noted that the only condition that produced a slight nanostructuring of TFA·DP1 was when a medium with intermediate polarity and volatility, 98
20 (conditions a–c in Table S2), poorly defined fibrillary structures could be observed when the volatility of the solvent:co-solvent mixture was the highest (Fig. S5). No significant improvement was observed when the peptide concentration was increased to 2 mg mL−1, or even when the volatility of the medium was enhanced by replacing water by EtOH (Fig. S6; conditions d–i in Table S2). After several tests and increasing the peptide concentration to 4 mg mL−1, we noted that the only condition that produced a slight nanostructuring of TFA·DP1 was when a medium with intermediate polarity and volatility, 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 EtOH
2 EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HFIP (condition j in Table 2), was used. This is reflected in Fig. 3a, which shows representative SEM micrographs of the peptide coating. As is shown, TFA·DP1 organizes in ultrathin nanofibrillary structures that form a continuous network without altering the intrinsic porosity of the PEDOT(3m) substrate.
HFIP (condition j in Table 2), was used. This is reflected in Fig. 3a, which shows representative SEM micrographs of the peptide coating. As is shown, TFA·DP1 organizes in ultrathin nanofibrillary structures that form a continuous network without altering the intrinsic porosity of the PEDOT(3m) substrate.
| C 1s | O 1s | S 2p | N 1s | F 1s | |
|---|---|---|---|---|---|
| 2PEDOT(3m)-TFA·DP1 | 54.50 | 42.32 | 0.71 | 1.94 | 0.53 |
| 2PEDOT(15s)-TFA·DP1 | 58.00 | 38.73 | 0.68 | 1.47 | 1.11 |
| 2PEDOT(3m) | 60.24 | 35.82 | 3.83 | 0.11 | — |
Three-layered TFA·DP1-containing electrode manufacturing
At this stage, an 8 mg mL−1TFA·DP1 solution in 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 98 HFIP
98 HFIP![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH was selected to manufacture both PEDOT(3m)- and PEDOT(15s)-based three-layered electrodes. However, the increase of the peptide concentration from 4 to 8 mg mL−1 produced some changes in the assembly of the unprotected peptide with respect to that displayed in Fig. 3a, especially when the PEDOT substrate was the roughest. Fig. 3b and c shows the assemblies obtained for the peptide deposited on PEDOT(3m) and PEDOT(15s), respectively. While TFA·DP1 formed globular aggregates of variable size on PEDOT(3m) (Fig. 3b), long fibrillary structures of micrometric thickness were regularly observed on PEDOT(15m) (Fig. 3c). These results support the aforementioned hypothesis, indicating that, when the peptide concentration is very high, the roughest substrate, PEDOT(3m), prioritizes an enthalphic contribution that, in this case, is dominated by strong non-specific electrostatic interactions, giving rise to disordered aggregates. In contrast, both the entropic and enthalpic contributions compete when the peptide is deposited on PEDOT(15s), facilitating the formation of ordered self-assembled structures.
EtOH was selected to manufacture both PEDOT(3m)- and PEDOT(15s)-based three-layered electrodes. However, the increase of the peptide concentration from 4 to 8 mg mL−1 produced some changes in the assembly of the unprotected peptide with respect to that displayed in Fig. 3a, especially when the PEDOT substrate was the roughest. Fig. 3b and c shows the assemblies obtained for the peptide deposited on PEDOT(3m) and PEDOT(15s), respectively. While TFA·DP1 formed globular aggregates of variable size on PEDOT(3m) (Fig. 3b), long fibrillary structures of micrometric thickness were regularly observed on PEDOT(15m) (Fig. 3c). These results support the aforementioned hypothesis, indicating that, when the peptide concentration is very high, the roughest substrate, PEDOT(3m), prioritizes an enthalphic contribution that, in this case, is dominated by strong non-specific electrostatic interactions, giving rise to disordered aggregates. In contrast, both the entropic and enthalpic contributions compete when the peptide is deposited on PEDOT(15s), facilitating the formation of ordered self-assembled structures.
SEM images of the electrodes derived by adding the layer electrogenerated in water, hereafter denoted 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1, are shown in Fig. 3d and e. As occurred for DP1-based electrodes, the TFA·DP1 was completely coated and hidden by the second layer of conducting polymer in 2PEDOT(3m)-TFA·DP1 (Fig. 3d), whereas the shape of the coated peptide fibers is easily identified in 2PEDOT(15s)-TFA·DP1 (Fig. 3e). However, since TFA·DP1 is water-soluble, there is a possibility that the peptide acted as a template that dissolved in the electrolytic medium during the incorporation of the third layer. In order to confirm that TFA·DP1 remained in the intermediate layer of the electrode after electropolymerizing the last PEDOT layer using an aqueous electrolytic medium, XPS studies were performed for both 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 electrodes. The XPS survey spectra, which are displayed in Fig. 4a and b, indicated that the two electrodes contain fluorine, oxygen, nitrogen, carbon and sulfur intensities at approximately 687, 532, 401, 285 and 167 eV, respectively.
The atomic percent compositions of 2PEDOT(3m) (control), 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 are compared in Table 2. The composition of the control was consistent with that expected for perchlorate-doped PEDOT, with a small fraction of nitrogen contaminating the sample. However, the S 2p![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N 1s ratio, which was approximately 35 for 2PEDOT(3m), decreased to 0.36 and 0.46 for 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1, respectively, evidencing a source of nitrogen different from the impurities found in the 2PEDOT(3m) electrode (Fig. S7). The high resolution N 1s spectra of 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 electrodes showed two peaks centered at around 402 and 400 eV, which were associated with the N–H and N–C bonds of the peptide group, respectively (Fig. 4c and d). The presence of TFA·DP1 at the electrode was also confirmed by the content of fluorine (Table 2), which was not detected for the control. The high resolution spectra of F 1s reflected a single peak at around 687 eV, which was assigned to the F–C bonds from both the TFA stabilizing ions and the β-2S,3S-Fpg residue (Fig. S8).
N 1s ratio, which was approximately 35 for 2PEDOT(3m), decreased to 0.36 and 0.46 for 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1, respectively, evidencing a source of nitrogen different from the impurities found in the 2PEDOT(3m) electrode (Fig. S7). The high resolution N 1s spectra of 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 electrodes showed two peaks centered at around 402 and 400 eV, which were associated with the N–H and N–C bonds of the peptide group, respectively (Fig. 4c and d). The presence of TFA·DP1 at the electrode was also confirmed by the content of fluorine (Table 2), which was not detected for the control. The high resolution spectra of F 1s reflected a single peak at around 687 eV, which was assigned to the F–C bonds from both the TFA stabilizing ions and the β-2S,3S-Fpg residue (Fig. S8).
Fig. 4e and f compare the cyclic voltammograms recorded for the 3-layered 2PEDOT(3m)/TFA·DP1 and 2PEDOT(15s)/TFA·DP1 electrodes with those obtained for uncoated and peptide-coated conducting polymer films (i.e. PEDOT(3m), PEDOT(15s), PEDOT(3m) + TFA·DP1 and PEDOT(15s) + TFA·DP1). As can be seen, the electrochemical activities of 2PEDOT(3m)-TFA·DP1, PEDOT(3m) + TFA·DP1 and PEDOT(3m) were very similar, suggesting that any peptide-induced effect was practically negated by the excessive thickness and roughness of the PEDOT layers (Table 1). Conversely, the area of the voltammogram recorded for 2PEDOT(15s)-TFA·DP1 was 41% and 38% times greater than those of PEDOT(15s)-TFA·DP1 and PEDOT(15s), respectively, evidencing that the dielectric breakage induced by the intermediate peptide fibers is manifested when PEDOT layers are thin and flat enough (i.e. thickness ≤ 1.5 Å and Rq ≤ 300 nm). Thus, in PEDOT(15s)-TFA·DP1, self-assembled TFA·DP1 structures behave as micro-dielectrics with respect to PEDOT(15s) layers, forming parallel interconnections. Furthermore, the improvement with respect to PEDOT(15s)-DP1 has been attributed to the electrical response of TFA anions (i.e. ions flow), which is added to electronic response (i.e. electrons flow). Although the peptide-induced improvement was much more evident for 2PEDOT(15s)-TFA·DP1 than for 2PEDOT(3m)-TFA·DP1, both electrodes were considered for constructing symmetric SCs.
Electrochemical performance of 2PEDOT-TFA·DP1 electrodes in symmetrical capacitors
SCs can be constructed using three different configurations. Type 1 uses two identical electrodes made with a p-doped conducting polymer (symmetric SCs), while type 2 uses two different p-doped conducting polymers as electrodes (asymmetric SCs). Finally, electrodes in type 3 are made with the same conducting polymer but using the p-and n-doped forms as positive and negative electrodes, respectively. In this work, type 1 symmetric SCs were constructed using two identical electrodes of 3-layered 2PEDOT(3m)-TFA·DP1 or 2PEDOT(15s)-TFA·DP1, and an acetonitrile solution containing 0.1 M LiClO4, as an electrolytic medium, as indicated in Scheme 3.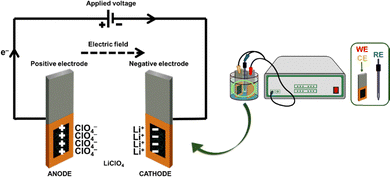 | ||
| Scheme 3 Sketch of the symmetric SCs constructed using 2PEDOT(3m)-TFA·DP1 or 2PEDOT(15s)-TFA·DP1 electrodes. | ||
The electrochemical behavior of SCs containing 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 electrodes was investigated by CV, GCD curves and EIS. Results were compared with those obtained for control electrochemical SC devices, which were prepared using two identical 2-layered PEDOT films as electrodes, 2PEDOT(3m) or 2PEDOT(15s); more specifically, electrodes prepared without incorporating the TFA·DP1 peptide at the intermediate layer.
For 2PEDOT(3m)-TFA·DP1, the reversal peak currents are linearly proportional to the square root of the scan rate (Fig. 5c, right), indicating that the substrate redox process is limited by diffusion. The relative similarity between 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1 indicated that the electrochemical processes were independent of peptide–conducting polymer interactions, as they were probably shielded by the external thick PEDOT(3m) layer. In the case of 2PEDOT(15s)-TFA·DP1, the reversal current is linearly proportional to the scan rate (Fig. 5d, right), which suggests that a surface confined process occurs. This is due to the physical adsorption of ions to the thin PEDOT(15s) layer via coulombic forces, which possibly occurs via the mediation of surface-confined, but still accessible, TFA·DP1 peptide fibers.
The stability of the two TFA·DP1-containing devices was evaluated by considering 200 cycles of CV. Selected voltammograms recorded for SCs made of 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1 are compared in Fig. 6a and b. The behavior of the two systems was completely different, as is also evidenced by the variation of the LEA (eqn (1)) with the number of CV cycles (Fig. 6c). For 2PEDOT(3m), the area of the voltammograms increased for the first 20 cycles (Fig. 6a), reflecting a self-stabilizing effect that increased the electroactivity by almost 20%. After that, the electroactivity decreased with increasing number of cycles, even though the LEA after 200 cycles remained positive (i.e. the voltammetric charge was higher after 200 cycles than after 2 cycles). This evidenced the high electrochemical stability of 2PEDOT(3m) electrodes. In contrast, the electroactivity of 2PEDOT(3m)-TFA·DP1 decreased rapidly with increasing number of cycles (Fig. 6b). Indeed, the LEA, which exhibited negative values during the 200 cycles, evidenced a reduction in the electroactivity close to 75% (Fig. 6c). This was attributed to the poor contact between the PEDOT layers at the interface with the peptide, which becomes worse when the peptide degrades by applying consecutive oxidation–reduction cycles.
 | ||
| Fig. 6 (a–f) Electrochemical stability of the studied symmetrical SCs. Cyclic voltammograms recorded after 2, 5, 10, 25, 50, 75, 100, 150 and 200 consecutive redox cycles for (a) 2PEDOT(3m), (b) 2PEDOT(3m)-TFA·DP1, (d) 2PEDOT(15s) and (e) 2PEDOT(15s)-TFA·DP1 electrodes. Variation of the loss of electroactivity (LEA; eqn (1)) with the number of redox cycles for (c) 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1 and (f) 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1. | ||
Fig. 6d and e display the cyclic voltammograms recorded after 2, 5, 10, 25, 50, 75, 100, 150 and 200 consecutive redox cycles recorded for the 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1 electrodes. As can be seen, the electrochemical stability of the two electrodes is extremely high. Specifically, the reduction in the electroactivity after 200 cycles with respect to the second cycle was as low as 2% and 5% for 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1, respectively (Fig. 6f), confirming that, in the latter electrode, the intermediate peptide layer works efficiently allowing the contact between the two outer PEDOT layers.
On the other hand, Fig. 7a compares the variation of the difference between electrochemical activities (ΔEA) for electrodes prepared applying the same tp with and without incorporating the intermediate peptide layer (i.e. 2PEDOT(3m)-TFA·DP1vs. 2PEDOT(3m) and 2PEDOT(15s)-TFA·DP1vs. 2PEDOT(15s)). As was previously observed (Fig. 5), the ΔEA of 2PEDOT(3m) is twice that of 2PEDOT(3m)-TFA·DP1 at the 2nd cycle (i.e. ΔEA = ∼50%). This difference became much more pronounced with increasing number of cycles, stabilizing at ∼11% after 75 cycles. Conversely, the electroactivities of 2PEDOT(15s)-TFA·DP1 and 2PEDOT(15s) were very similar, differing by less than 8%, regardless of the number of redox cycles. The specific capacitance (sc; eqn (2)) derived from the recorded voltammograms reflects this behavior (Fig. 7b). While the specific capacitance is much higher for 2PEDOT(3m) than for 2PEDOT(3m)-TFA·DP1 after 2 and 200 redox cycles (207.6 vs. 26.0 mF cm−2 and 228.3 vs. 6.8 mF cm−2, respectively), the specific capacitances obtained for 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1 are very similar and independent of the number of redox cycles (i.e. around 47 mF cm−2 in all cases).
Electrodes fabricated by combining conducting polymers with other intrinsically conducting materials (e.g. graphene, carbon nanotubes and metallic nanoparticles) often exhibit specific capacitances exceeding those obtained in this work for 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15)-TFA·DP1, in some cases by up one order of magnitude.43 However, the specific capacitances measured for the α/β-dipeptide-based supercapacitors studied here are comparable to those reported for devices employing electrodes with similar constituents (i.e. PEDOT and peptides). For example, supercapacitors assembled with electrodes composed of a polypeptide hydrogel loaded with PEDOT microparticles, further interconnected through an additional semi-interpenetrated PEDOT network formed by electropolymerization, displayed a specific capacitance of 45.4 ± 0.7 mF cm−2.44 In contrast, PEDOT electrodes coated with aromatic α-peptides exhibited areal specific capacitances several orders of magnitude lower than those achieved in the present study.30
 | ||
| Fig. 8 Representative GCD curves for (a) 2PEDOT(3m), (b) 2PEDOT(3m)-TFA·DP1, (c) 2PEDOT(15s) and (d) 2PEDOT(15s)-TFA·DP1. | ||
The GCD curves recorded for 2PEDOT(15s) (Fig. 8c) showed important differences with respect to those obtained for 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1. First, the reached voltage increased with the current intensity, while the charging time was significantly high (i.e. between 3 and 10 s depending on the current intensity). These features indicated that the flow of electrons from the first to the second layer is not continuous, affecting both the charging time and the reached voltage. Second, a significant voltage drop (Vdrop) appeared at the beginning of the discharging process. This effect, which increased with the current intensity from Vdrop = 0.11 V at 0.058 mA to Vdrop = 0.58 V at 0.66 mA, was attributed to the electrode interfacial resistance to charge transport, which affected the current flow.
Finally, the GCD curves obtained for 2PEDOT(15s)-TFA·DP1 (Fig. 8d) show improvements with respect to those of the rest of the systems. First, the cell voltage was 0.8 V for all tested current intensities, including the lowest one. Furthermore, the voltage drop was significantly lower than that observed for 2PEDOT(15s), improving the shape of the curves. Most importantly, the coulombic efficiency was significantly tuned by decreasing the current intensity, which was due to the charging time that was lower for 2PEDOT(15s)-TFA·DP1 than for the other systems. For example, at a current intensity of 0.22 mA, tc was 2.15 and 0.54 s for 2PEDOT(3m) and 2PEDOT(15s)-TFA·DP1, respectively (Fig. 8a and d). The behavior of tc allowed the latter to achieve a η of 25% when the applied current intensity was 0.05 mA only (with td and tc of 0.56 and 2.22 s, respectively). Instead, for 2PEDOT(3m), tc increased much more than td when the applied current was lower than 0.22 mA, leading to η lower than 13%.
 | ||
| Fig. 9 Nyquist plots recorded for SCs prepared using: (a) 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1; and (b) 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1. (c) RS and (d) RP values derived from EIS data. | ||
The diameter of the semicircle corresponded to the charge transfer resistance (RP) at the electrode/electrolyte interface, also known as the interface reaction resistance. Independently of the tp, the RP, which included the effects induced by the multilayered configuration of the electrodes, was significantly lower for the peptide-containing electrodes than for those without peptide (Fig. 9d). The RP values obtained for 2PEDOT(3m) and 2PEDOT(3m)-TFA·DP1 were 69.3 and 10.7 Ω cm−1, respectively, while those of 2PEDOT(15s) and 2PEDOT(15s)-TFA·DP1 were 45.8 and 1.0 Ω cm−1, respectively. This indicated that peptide-containing electrodes presented better interfacial contact with the electrolyte, which enhanced the ion transport and reduced the interfacial resistance. On the other hand, the lower resistance of the electrodes prepared using tp = 15 s in relation to those obtained using 3 min was totally consistent with our previous observations. Thus, our results showed that the smaller the thickness of the films, the more favorable the peptide-induced reorganization of the capacitive and conductive paths is.
Overall, the performance of SCs based on 2PEDOT(15s)-TFA·DP1 electrodes was intermediate between those reported for devices prepared using 3-layered electrodes with poly(N-methylpyrrole) (PNMPy)18 and clays16 at the intermediate layer (hereafter referred to as 2PEDOT-PNMPy and 2PEDOT-clay, respectively) instead of TFA·DP1 assemblies. The performance of 2PEDOT-PNMPy, which improved by decreasing the tp, as in this work, was attributed to a synergistic effect produced by the favorable interaction between the PEDOT and PNMPy layers at the corresponding interfaces, with the PNMPy intermediate sheet promoting the dielectric breakage effect (i.e. the electrical conductivity was ∼105 S cm−1 higher for PEDOT than for PNMPy).18,45 Instead, the very low performance of SCs based on 3-layered 2PEDOT-clay electrodes was attributed to the completely inert electrochemical nature of the clays.46 The electrochemical response of TFA·DP1 assemblies was primarily due to electron transport between stacked rings (i.e. intermolecular electronic contribution) and the movement of ions that stabilize the amine. This response was lower than that of PNMPy in which the charge moved along the polymer molecules both at the electronic level (intramolecular delocalization of polarons and bipolarons) and at the ionic level (counterions that neutralize the charge of the PNMPy chains). In the case of clays, the ions were strongly trapped between the charged silicate layers, and consequently, these materials did not exhibit electrochemical response.
On the other hand, it is worth noting that the use of electro-responsive peptide assemblies could be readily extended to the fabrication of various types of asymmetric supercapacitors. This could be achieved either by employing different conducting polymers at each electrode while using the same peptide in both, or by using different assembled peptides at each electrode while keeping the conducting polymer identical. Furthermore, TFA·DP1 peptide assemblies could also be integrated into flexible supercapacitors based on conducting polymer hydrogels. This could be accomplished either by incorporating pre-formed TFA·DP1 assemblies during the gelation process or by promoting the formation of peptide assemblies within a preformed 3D hydrogel network (i.e. infiltrating the peptide into the hydrogel).
Conclusions
In summary, we demonstrated that small α/β-dipeptides DP1 and TFA·DP1 can self-assemble into well-ordered structures on electropolymerized PEDOT substrates. In this novel self-assembly approach, in which the organization of the peptide molecules is conditioned by the very high roughness and particular morphology of such a conducting polymer, a synergy between the semi-conductive substrate and the self-assembled structures was sought in the electrochemical response. In order to prove such synergy, we systematically studied the electrochemical behavior of 3-layered electrodes consisting of two PEDOT layers separated by an intermediate layer of one of the self-assembled peptides constructed for the first time. Furthermore, the influence of the PEDOT layer thickness was taken into account, considering polymerization times of 3 min and 15 s. Electrochemical evaluation of the resulting electrodes revealed that the synergy between PEDOT and the self-assembled peptide only occurred with TFA·DP1. This was attributed to the fact that such a peptide, when forming ordered structures, not only transported electrons through the stacked aromatic rings, but also the counterions associated with the charged amino terminal group.Symmetric SCs prepared using 2PEDOT(3m)-TFA·DP1 and 2PEDOT(15s)-TFA·DP1 were characterized and compared with those obtained using electrodes without peptide at the intermediate layer. Peptide-containing systems not only displayed lower electrical resistance but also good voltammetric response and charge–discharge capacity. Our results suggest that self-assembled peptide nano- and micro-structured materials can perform excellently as biocompatible active components in electrochemical SCs, which are promising for use in wearable, implantable, and biodegradable devices. The proof of concept reported in the present study could be extended to de novo designed peptide sequences. We therefore envision that molecular engineering aimed, on the one hand, to having sequences that promote electronic and ionic transport efficiently, and on the other hand, to facilitate obtaining ordered self-assembled structures on soft semi-conducting substrates, will have broad and important applications in bioelectronics.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study have been included in the article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01170a.Other datasets generated during this study, such as raw and processed data, are available from the corresponding author upon reasonable request.
Acknowledgements
This publication received funding from the European Union's research and innovation program under the Marie Skłodowska-Curie grant agreement No. 101072645. The IMEM-BRT group thanks the Agència de Gestió d'Ajuts Universitaris i de Recerca (2021 SGR 00387) for financial support. This work is part of Maria de Maeztu Units of Excellence Programme CEX2023-001300-M funded by MCIN/AEI/10.13039/501100011033. Support for the research of C. A. was also received through the prize “ICREA Academia 2022” for excellence in research funded by the Generalitat de Catalunya.References
- X. Fan, C. Zhong, J. Liu, J. Ding, Y. Deng, X. Han, L. Zhang, W. Hu, D. P. Wilkinson and J. Zhang, Chem. Rev., 2022, 122, 17155–17239 Search PubMed
.
- Y. Jia, J. Zhang, D. Kong, C. Zhang, D. Han, J. Han, Y. Tao, W. Lv and Q.-H. Yang, Adv. Funct. Mater., 2022, 32, 2204272 CrossRef CAS
.
- L. Liu, X. Zhang, Y. Liu and X. Gong, ACS Appl. Electr. Mater., 2025, 7, 2233–2270 CrossRef CAS
.
- S. Zhang, Y. Liu, J. Hao, G. G. Wallace, S. Beirne and J. Chen, Adv. Funct. Mater., 2022, 32, 2103092 CrossRef CAS
.
- J. Chen and P. S. Lee, Adv. Energy Mater., 2021, 11, 2003311 CrossRef CAS
.
- L. Li, J. Meng, M. Zhang, T. Liu and C. Zhang, Chem. Commun., 2022, 58, 185–207 RSC
.
- H. A. Khan, M. Tawalbeh, B. Aljawrneh, W. Abuwatfa, A. Al-Othman, H. Sadeghifar and A. G. Olabi, Energy, 2024, 295, 131043 CrossRef CAS
.
- C. Padwal, H. D. Pham, L. T. My Hoang, S. Mundree, A. K. Nanjundan, S. G. Krishnan and D. Dubal, ChemSusChem, 2024, 17, e202301866 CrossRef CAS PubMed
.
- S. S. Shah, F. Niaz, M. A. Ehsan, H. T. Das, M. Younas, A. S. Khan, H. U. Rahman, S. M. A. Nayem, Md. M. Oyama and A. Aziz, J. Energy Storage, 2024, 79, 110152 CrossRef CAS
.
- Y. Yu, C. Yang, Y. Jiang, J. Zhu, Y. Zhao, S. Liang, K. Wang, Y. Zhou, Y. Liu and J. Zhang, Small, 2023, 19, 2303779 CrossRef CAS PubMed
.
- A. M. Bryan, L. M. Santino, Y. Lu, S. Acharya and J. M. D’Arcy, Chem. Mater., 2016, 28, 5989–5998 CrossRef CAS
.
- C. Bauer, M. Kirchner and A. Krueger, Energy Adv., 2024, 3, 1422–1430 RSC
.
- M. N. Gueye, A. Carella, J. Faure-Vincent, R. Demadrille and J.-P. Simonato, Prog. Mater. Sci., 2020, 108, 100616 CrossRef CAS
.
- M. G. Tadesse, N. Simon and J. F. Lübben, Adv. Energy Sustainability Res., 2024, 5, 2400006 CrossRef CAS
.
- X. Wang, P. Wang, W. Tian, H. Bu and K. Zhang, ACS Sustainable Chem. Eng., 2023, 11, 17510–17518 CrossRef CAS
.
- D. Aradilla, D. Azambuja, F. Estrany, M. T. Casas, C. A. Ferreira and C. Alemán, J. Mater. Chem., 2012, 22, 13110–13122 RSC
.
- F. Niu, X. Han, H. Sun, Q. Li, X. He, Z. Liu, J. Sun and Z. Lei, ACS Sustainable Chem. Eng., 2021, 9, 4146–4156 CrossRef CAS
.
- D. Aradilla, F. Estrany and C. Alemán, J. Phys. Chem. C, 2011, 115, 8430–8438 CrossRef CAS
.
- W. Yan, J. Li, G. Zhang, L. Wang and D. Ho, J. Mater. Chem. A, 2020, 8, 554–564 RSC
.
- M. F. Jimoh, G. S. Carson, M. B. Anderson, M. F. El-Kady and R. B. Kaner, Adv. Funct. Mater., 2025, 35, 2405569 CrossRef CAS
.
- T. Cheng, X. L. Yang, S. Yang, L. Li, Z. T. Liu, J. Qu, C. F. Meng, X. C. Li, Y. Z. Zhang and W. Y. Lai, Adv. Funct. Mater., 2022, 33, 2210997 CrossRef
.
- X. Liu, Y. Chen, H. Zhang, L. Zhuo, Q. Huang, W. Zhang, H. Chen and Q. Ling, J. Colloid Interface Sci., 2024, 660, 735–745 CrossRef CAS PubMed
.
- P. Gomez-Romero, Adv. Mater., 2001, 13, 163–174 Search PubMed
.
- S. Chakraborty, K. El Battioui and T. Beke-Somfai, Small Sci., 2024, 4, 2300217 Search PubMed
.
- K. Hu, C. Zheng, X. Miao and L. Wang, J. Mater. Chem., 2018, 6, 8047–8052 RSC
.
- Y. Tian, J. Li, A. Wang, Q. Li, H. Jian and S. Bai, Macromol. Biosci., 2023, 23, 2300171 Search PubMed
.
- O. S. Tiwari, Mol. Syst. Des. Eng., 2022, 7, 171–181 RSC
.
- J. Zhao, Q. Liu, X. Tong, Y. Wang, K. Cai and W. Ji, Adv. Funct. Mater., 2024, 34, 2401466 CrossRef CAS
.
- M. R. Biradar, H. A. Mirgane, S. V. Bhosale and S. V. Bhosale, J. Energy Chem., 2024, 99, 253–276 CrossRef CAS
.
- A. Fontana-Escartín, E. Rosa, C. Diaferia, S. Lanzalaco, A. Accardo and C. Alemán, J. Colloid Interface Sci., 2025, 679, 441–454 CrossRef PubMed
.
-
Y. Wang, K. Wan, C. Liu and W. Zhang, In Peptide Nanomaterials for Energy and Environmental Science Applications, Peptide Nano-Chemistry and Nanotechnology, ed G. Wei, Springer, Singapore, 2025 Search PubMed
.
- K. Hu, C. Zheng, M. An, X. Ma and L. Wang, J. Mater. Chem. A, 2018, 6, 8047–8052 RSC
.
- R. Bucci, A. Contini, F. Clerici, E. M. Beccalli, F. Formaggio, I. Maffucci, S. Pellegrino and M. L. Gelmi, Front. Chem., 2019, 7, 192 CrossRef CAS PubMed
.
- R. P. Cheng, S. H. Gellman and W. F. DeGrado, Chem. Rev., 2001, 101, 3219–3232 CrossRef CAS PubMed
.
- A. Bonetti, S. Pellegrino, P. Das, S. Yuran, R. Bucci, N. Ferri, F. Meneghetti, C. Castellano, M. Reches and M. L. Gelmi, Org. Lett., 2015, 17, 4468–4471 CrossRef CAS PubMed
.
- R. Bucci, P. Das, F. Iannuzzi, M. Feligioni, R. Gandolfi, M. L. Gelmi, M. Reches and S. Pellegrino, Org. Biomol. Chem., 2017, 15, 6773–6779 RSC
.
- M. Gul, D. A. Quintana-Romero, A. Fontana-Escartín, M. L. Gelmi, M. M. Pérez-Madrigal, E. Chiesa, R. Bucci and C. Alemán, Adv. Mater. Interfaces, 2025, e00254 CrossRef CAS
.
- Y. Zhang, L. Li and B. He, RSC Adv., 2024, 14, 30045–30054 RSC
.
- E. Poverenov, M. Li, A. Bitler and M. Bendikov, Chem. Mater., 2010, 22, 4019–4025 CrossRef CAS
.
- K. Shezad, K. J. Zhang, M. Hussain, H. Dong, C. X. He, X. J. Gong, X. L. Xie, J. T. Zhu and L. Shen, Langmuir, 2016, 32, 8238–8244 CrossRef CAS PubMed
.
- D. Aradilla, M. M. Pérez-Madrigal, F. Estrany, D. Azambuja, J. I. Iribarren and C. Alemán, Org. Electr., 2013, 14, 1483–1495 CrossRef CAS
.
- D. Amoura, M. Sánchez-Jiménez, F. Estrany, L. Makhloufi and C. Alemán, Eur. Polym. J., 2015, 69, 296–307 CrossRef CAS
.
- I. O. Oladele, S. O. Adelani, A. S. Taiwo, I. M. Akinbamiyorin, O. F. Alanrewaju and A. O. Orisawayi, RSC Adv., 2025, 15, 7509–7534 RSC
.
- M. C. G. Saborío, S. Lanzalaco, G. Fabregat, J. Puiggalí, F. Estrany and C. Alemán, J. Phys. Chem. C, 2018, 122, 1078–1090 CrossRef
.
- F. Estrany, D. Aradilla, R. Oliver, E. Armelin and C. Aleman, Eur. Polym. J., 2008, 44, 1323–1330 CrossRef CAS
.
- N. Borras, F. Estrany and C. Alemán, Org. Electr., 2017, 51, 322–331 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |