Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
CsPbBr3 nanocrystals supported on a partially oxidized Ti2N MXene for photothermal CO2 conversion
Kevin
Mego
 a,
Emanuela
Accardo
a,
Emanuela
Accardo
 b,
Pedro
Ruiz-Campos
b,
Pedro
Ruiz-Campos
 c,
Herme G.
Baldoví
c,
Herme G.
Baldoví
 *b and
Pedro
Atienzar
*b and
Pedro
Atienzar
 *a
*a
aInstituto de Tecnología Química, Consejo Superior de Investigaciones Científicas, Universitat Politècnica de València, Avenida de los Naranjos s/n, 46022, Valencia, Spain. E-mail: pedatcor@itq.upv.es
bDepartamento de Química, Universitat Politècnica de València, Camino de Vera s/n, 46022, Valencia, Spain. E-mail: hergarba@itq.upv.es
cUniversidad Científica del Sur, Panamericana Sur Km 19, 150142, Lima, Peru
First published on 29th December 2025
Abstract
The development of new photo-thermal catalysts for the transformation of CO2 into fuels is of great interest, offering a clean and sustainable approach to reducing the carbon footprint. Herein, we present a novel hybrid material composed of a nanocrystalline metal halide perovskite (CsPbBr3) supported on a two-dimensional titanium nitride (Ti2N) MXene. Additionally, we demonstrate the importance of forming an external TiO2 layer through partial oxidation of the MXene (POM–Ti2N), which introduces catalytic centers and enhances photogenerated charge separation. Remarkable activity in the formation of CH4 and CO was observed, with yields of 321 µmol g−1 and 480 µmol g−1, respectively. The selectivity of the reaction was found to be temperature dependent. The mechanism was thoroughly investigated using XPS and photoluminescence studies. XPS analysis revealed a significant chemical interaction between the CsPbBr3 nanocrystals and the POM–Ti2N MXene after the formation of the composite. Photoluminescence measurements revealed a considerably shorter emission lifetime for the hybrid catalyst (τave = 1.73 ns) compared to that of the CsPbBr3 nanoparticles (τave = 25.32 ns), indicating strong interaction with the MXene. Furthermore, this research highlights the potential of combining metal halide perovskites with MXenes and the importance of controlling their interface for photo-thermal reactions.
1. Introduction
Researchers are making significant efforts to address climate change by developing CO2 utilization technologies that convert this molecule into chemicals. Solar-driven photothermal catalysis is a compelling and sustainable approach that harnesses light and heat energy to synergistically convert CO2 into fuels and valuable chemicals.1,2 There are many photothermal catalytic pathways that offer sustainable routes for CO2 valorization,3,4 such as the Sabatier reaction, which has been one of the most studied reactions for over a century since its discovery. This reaction consists of the hydrogenation of CO2 to CH4, which is thermodynamically favorable but is hindered by slow reaction kinetics; therefore, the use of high temperatures is necessary to activate the CO2 molecule. The photothermal catalytic mechanism includes multiple proton-coupled electron transfer to CO2 to form CH4, which requires control of intermediate species on catalytic surfaces, often resulting in reduced selectivity and lower overall activity.5In the classical thermal methanation process, the most studied metallic active centers are Ni, Ru, and Rh, which require high temperatures (300 to 400 °C) and pressures (5–20 bar) to achieve high efficiencies. However, the photothermal pathway can promote the Sabatier reaction under milder conditions, employing the same active sites supported on a semiconductor, with metal oxides being the most commonly used due to their robustness under reaction conditions. Recent studies showed that nickel (Ni) has emerged as an optimal compromise, delivering Ru-like activity when properly nanostructured on TiO2 or CeO2 supports.6–8 Other researchers used single atoms and nanoparticles of ruthenium supported on CeAlOx, achieving formidable catalytic performances.9 Although a huge number of materials have been already explored to boost the photocatalytic side of the photothermal reaction (e.g. metal oxides, MOFs, COFs, metal complexes, C3N4…),10–13 there is still a need to boost this reaction at lower temperatures (<250 °C).
Metal halide perovskite nanocrystal (NC) semiconductors have been extensively studied because they offer a wide range of applications due to their excellent optoelectronic properties, such as broad absorption in the UV-vis spectrum, tunable light absorption range, long charge diffusion length, and high molar extinction coefficients.14 All these properties make them potential candidates for multiple photocatalytic applications. However, to improve the photocatalytic activity of pristine CsPbBr3, it has been combined with other materials, on the one hand, to improve charge separation properties through charge migration and, on the other hand, to add better active catalytic sites.15,16 In this regard, MXene materials as a new class of two-dimensional (2D) structures have recently attracted great attention due to their interesting properties such as rich surface chemistry, modifiable electronic structure and thermal stability. MXenes are transition metal carbides or nitrides with the general formula Mn+1XnTx (n = 1, 2 or 3), where M corresponds to a transition metal, X can be C or N, and Tx indicates the surface functional groups (usually –F, –O or –OH). These functional groups play an important role in the adsorption/desorption of molecules and electron transfer properties. Currently, in photothermal reduction of CO2, there are a few examples in the literature studying the catalytic properties of MXene carbides containing perovskite NCs. For instance, aerogels of a CsPbBr3/Ti3C2Tx MXene showed that the Ti3C2Tx MXene's conductive network promotes charge separation from photoexcited CsPbBr3, while its surface terminations (–O, –F, –and OH) improve CO2 adsorption, outperforming individual components.17
In addition, partially oxidized 2D transition metal carbide MXenes also offer the possibility to decorate the titanium nitride MXene with TiO2 moieties. According to the work of Pazniak et al.,18 the enhanced charge carrier exchange at the TiO2/MXene interface enhances the sensing properties due to the formation of a depletion region between the metal and the semiconductor interface.19
The novelty of this work lies in the use of the titanium nitride (Ti2N) MXene in the Sabatier reaction for the first time. Therefore, within this context, we have been the first investigating the formation of a novel hybrid material composed of nanocrystalline CsPbBr3 supported on a two-dimensional titanium nitride (Ti2N) MXene for photo-thermal CO2 conversion. To date, there has been no report describing this specific combination. Although bare metal nitride MXenes can exhibit certain limitations in this reaction, we addressed this by forming external TiO2 moieties through partial oxidation of the Ti2N MXene and combining it with the perovskite nanocrystals. We evaluated catalytic activity under different conditions and assessed its reusability, concluding that this hybrid is capable of performing CO2 revalorization. XPS analysis and photophysical studies were carried out. Finally, we propose a working mechanism based on the formation of a heterojunction architecture, featuring an interlayer that inhibits direct charge recombination.
2. Results and discussion
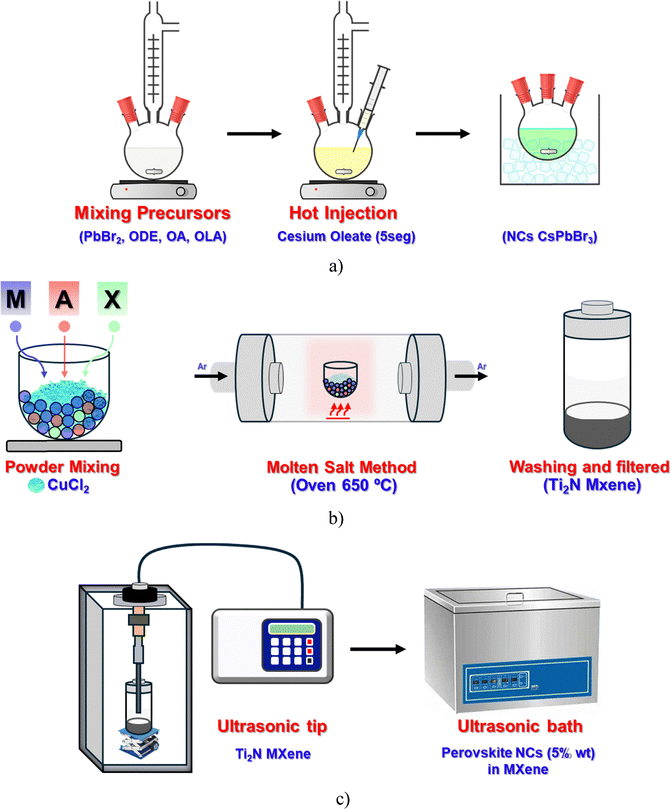
CsPbBr3 perovskite NCs and the Ti2N MXene were prepared separately according to the synthetic procedure described in detail in the Materials and Methods section (see SI) and illustrated in Scheme 1. The synthesis of CsPbBr3 NCs was carried out by the hot injection method.20Scheme 1a shows the conventional preparation of the NCs through three sequential steps: (i) mixing the precursors in a three-neck flask at elevated temperature, (ii) rapid injection of the cesium precursor, and (iii) immediate quenching in an ice-cold water bath.The preparation of the partially oxidized multilayer Ti2N MXene (POM–Ti2N) involves two main stages: (i) first the MAX phase was treated with CuCl2 using the molten salt method at high temperature (650 °C), followed by copper removal using persulfate solution, and (ii) afterwards the solid was filtered and thoroughly washed with deionized water. It is during the purification with persulfate salts at 70 °C that a thin layer of small nanoparticles of TiO2 was grown on top of the surface resulting in the partial oxidation of the Ti2N MXene. (see Scheme 1b). Actually, TiO2's presence is residual if the purification is carried out during the same time and at room temperature.
Finally, the perovskite NCs were supported on POM–Ti2N by dispersing POM–Ti2N by ultrasonication in hexane and adding dropwise a concentrated suspension of NCs. After some time, the solid was recovered by filtration. More details are described in the Materials and methods section in the SI (Scheme 1c).
The synthesis of M–Ti2N was carried out according to the publication by Xinbo Liu et al., following the molten salt method, in which molar ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 1
3 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 with respect to Ti2NAl
4 with respect to Ti2NAl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CuCl2 were used. In our case, a ratio of 1
CuCl2 were used. In our case, a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 was chosen to synthesise the multi-layered T2N MXene.21 In fact, analysis carried out with X-ray diffraction spectroscopy shown in Fig. S4 confirms that the titanium nitride MXene synthesized by the molten salt method leads to a nitride with the crystal structure of α-TiN with characteristic peaks at 37.9°, 43.3° and 62.7° corresponding, respectively, to the planes (004), (103), and (110).22,23
4 was chosen to synthesise the multi-layered T2N MXene.21 In fact, analysis carried out with X-ray diffraction spectroscopy shown in Fig. S4 confirms that the titanium nitride MXene synthesized by the molten salt method leads to a nitride with the crystal structure of α-TiN with characteristic peaks at 37.9°, 43.3° and 62.7° corresponding, respectively, to the planes (004), (103), and (110).22,23
A slight modification of the purification procedure during the bath with persulphate salts, such as raising the temperature of the process or increasing the concentration of these salts, leads to modification of the chemistry of the surface of the MXene layers by superficially oxidising the M–Ti2N layers as can also be seen in Fig. S4 with the appearance of new TiO2-related signals at 2θ = 25.3°, 47.9°, and 54.4° and 2θ = 27.4° and 43.3° assigned to (101), (202), and (211) anatase and (110) and (111) rutile crystalline planes.
Furthermore, XRD analyses were performed for all the materials. The XRD results were consistent with the ICSD data file #204325,24 which confirms the cubical crystalline structure of the CsPbBr3 NCs, evidenced by the presence of the peaks at the angles 15° and 30°, which represent the (001) and (002) crystalline planes of the cubic perovskite crystals (Fig. 1b). Once the NCs@POM–Ti2N composite has been formed, it is not possible to visualise the signals due to the low concentration of NCs in the sample (<5% by weight) and the small amount of material used during the analysis (Fig. 1a).
Moreover, high-resolution transmission electron microscopy (HR-TEM) was used to study the CsPbBr3 NCs and the Ti2N MXene independently. Fig. 2a shows that the perovskite NC particles have a similar size and crystallinity. The particle size distribution of NCs was estimated using HR-TEM, yielding an average size of 5 nm (Fig. 2b).
High resolution field emission scanning electron microscopy (HR-FESEM) is a useful tool to distinguish between the formation of a multi-layered Ti2N MXene (M–Ti2N) and POM–Ti2N. A soft purification treatment with 0.1 M sodium persulfate at room temperature leads easily to purification of M-Ti2N with a minor structural defect, as shown in Fig. 2c, whereas the same concentration of persulfate, but treating the MXene at 70 °C, resulted in slight oxidation of the MXene surface, forming POM–Ti2N (see Fig. 2d). Also, HR-FESEM images in Fig. S5 reveal the formation of rugosity on the surface and edges of the Ti2N MXene layers with the appearance of small crystals of TiO2 homogenously dispersed. Additionally, interplanar distances measured using HR-TEM on layers of the POM–T2N sample confirm the simultaneous presence of Ti2N and TiO2 crystals (Fig. 2e) After the formation of the composite NCs@POM–Ti2N no evident changes are seen by HR-FESEM in the partially oxidized MXene particle (Fig. 2f); however, the dispersion of NC particles on the POM–Ti2N MXene is confirmed from the DF STEM image in Fig. S6. This image showed that elements that form the perovskite particles are well dispersed and distributed homogenously over the support. A clearer image that shows the physical presence of perovskite NCs on the POM–Ti2N support is shown in Fig. S7, from which we can estimate a loading of 5% per weight of NCs on the POM–Ti2N MXene.
To elucidate the fine structure of the hybrid, HR-TEM images of NCs@POM–Ti2N are presented in Fig. 2g and h. As shown in Fig. 2g, TiO2 nanoparticles are uniformly anchored onto the Ti2N MXene sheets, forming a surface-decorated25 hybrid. This observation confirms that the partial surface transformation of Ti2N into TiO2 occurs during processing, yielding a coherent TiO2/Ti2N heterostructure.26
The distinct lattice fringes observed for both phases further verify the successful formation of the composite (Fig. 2h). Spacings of 3.50 Å and 2.70 Å, as well as 2.10 Å, correspond to the (101) planes of anatase TiO226 and the (01![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) planes of Ti2N,27,28 respectively. In addition, the lattice fringes of 3.00 Å are assigned to the (200) planes of CsPbBr3 nanocrystals,29 indicating their favourable deposition across the entire TiO2/Ti2N MXene surface.
0) planes of Ti2N,27,28 respectively. In addition, the lattice fringes of 3.00 Å are assigned to the (200) planes of CsPbBr3 nanocrystals,29 indicating their favourable deposition across the entire TiO2/Ti2N MXene surface.
Interestingly, the Ti2N nanosheets appear partially intercalated among the surface-grown TiO2 nanoparticles, generating a textured template onto which CsPbBr3 NCs are subsequently anchored. This morphological arrangement is entirely consistent with the structural model illustrated in Fig. 8a for NCs@POM–Ti2N. Such an architecture provides an enlarged interfacial contact area, thereby facilitating efficient charge-carrier migration throughout the heterostructure.
The thermal stability of CsPbBr3 and the POM–Ti2N MXene was investigated using thermogravimetric analysis (TGA) (Fig. S8). The results indicate that the POM–Ti2N MXene exhibits high thermal stability, showing only a small percentage of weight loss over the entire temperature range studied. In addition, CsPbBr3 (NCs) shows a sharp weight loss around 600 °C, indicating thermal stability up to this temperature. All of the above indicates the stability of the materials under the catalytic conditions studied.
The surface change induced by the partial oxidation of M–Ti2N produces profound changes in the optical absorption of the material. As shown in Fig. S9, when titanium nitride is present, the absorbance spans the UV region to the entire visible range. However, when oxidation occurs, its absorbance becomes significantly more prominent in the UV region up to 380 nm due to the appearance of TiO2 domains on the M–Ti2N structure. For the sake of comparison, diffuse reflectance UV-vis spectra of perovskite NCs, the POM–Ti2N MXene, and the composite NCs@POM–Ti2N are also plotted in Fig. 3. Herein, we can see the POM–Ti2N MXene UV-vis spectra exhibiting a strong band absorption with an edge at 380 nm, characteristic of TiO2 absorption. In the case of CsPbBr3 NCs, they display characteristic UV-vis absorption within the wavelength range of λ < 540 nm. The UV-vis spectrum also verifies the formation of the composite NCs and POM–Ti2N MXene as the supported material combines both absorptions, expanding the photophysical response of the initial POM–Ti2N in the visible region.
The optical band gaps (Eg) of the synthesized materials were determined using UV-vis diffuse reflectance spectroscopy (DRS), analyzed through Tauc plot extrapolations (Fig. S10, SI).30 The extracted band gap values were found to be 2.28 eV31 for CsPbBr3 NCs and 3.26 eV for POM–Ti2N, respectively.
To further elucidate the superior optoelectronic behavior of the perovskite material, the valence band edge positions (ENHEv) relative to the normal hydrogen electrode (NHE) were investigated using X-ray photoelectron spectroscopy (XPS).32–34 The ENHEv values were calculated based on eqn (1):
| ENHEv = Efv + φsp + ENHE0 | (1) |
Based on the calculated valence band (VB) positions and the corresponding optical band gaps, the conduction band minimum (CB) energies were inferred for each material. The estimated VB potentials referenced to the NHE were 1.14 eV for CsPbBr3 NCs and 2.30 eV for POM–Ti2N, as shown in Fig. S11(a and b). Moreover, the CB values of CsPbBr3 NCs and POM–Ti2N are −1.14 and −0.96 eV, respectively. Regarding the POM–Ti2N MXene material, we expect two distinct regions: an outer region corresponding to the partially oxidized Ti2N MXene with TiOx groups exhibiting electron-withdrawing behavior, and an inner region mainly of the Ti2N MXene with a metallic character, which facilitates efficient electron extraction. Consequently, energy band diagrams were constructed, providing comprehensive insight into the electronic structure of the materials investigated (Fig. 3b).
With the aim to understand the mechanism of the catalytic process that occurs during the photo-thermal reaction, steady-state and lifetime emission were studied. The PL spectrum of the CsPbBr3 NCs exhibits a strong emission peak at 530 nm; however, for the sample CsPbBr3@POM–Ti2N MXene, we observed a decrease in PL intensity along with a blue shift in the peak maximum to 513 nm. This shift to the blue can be attributed to the Burstein–Moss effect.35 This effect is related to the change in carrier concentration caused by the electronic coupling between the perovskite nanocrystals and the MXene material. In addition, the decrease in emission intensity can be ascribed to the transfer of photogenerated carriers between the CsPbBr3 NCs and the POM–Ti2N MXene.36
Time-resolved fluorescence spectroscopy (Fig. 4) was used to investigate the lifetime emission of pure CsPbBr3 NCs and the supported CsPbBr3@POM–Ti2N MXene catalyst. The best fitting corresponds to a biexponential decay model, where the short lifetime is attributed to nonradiative recombination of electron–hole pairs and surface trap states and the long lifetime to the radiative recombination.37,38
The emission lifetime of the CsPbBr3@POM Ti2N MXene (τave = 1.73 ns) was relatively shorter than that of the corresponding CsPbBr3 nanoparticles (τave = 25.32 ns). This difference in the emission lifetime between both materials indicates the emergence of a nonradiative pathway from the significant electronic interaction between CsPbBr3 nanoparticles and the partially oxidized MXene.18 This provides an alternative interfacial pathway for the excited electrons of CsPbBr3 to transfer to the oxidized MXene and relax non-radiatively, allowing them to participate in the catalytic reaction.35,39
X-ray photoelectron spectroscopy (XPS) measurements were conducted to determine the chemical states of lead (Pb), bromine (Br), cesium (Cs), titanium (Ti), nitrogen (N) and oxygen (O) in the CsPbBr3 NCs, POM–Ti2N, and NCs@POM–Ti2N samples (Fig. S12–S14 show the full survey scans). Fig. S12 displays the XPS core spectra of Cs 3d, Pb 4f, and Br 3d states, respectively, calibrated using the adventitious C 1s peak set to a binding energy (BE) of 284.8 eV.40 In the case of CsPbBr3 NCs, the BE peaks for Cs 3d at 724.0 and 738.0 eV correspond to the Cs 3d5/2 and Cs 3d3/2 signals (Fig. 5a), while the Pb 4f spectrum shows two peaks at 138.0 and 142.9 eV, attributed to the Pb 4f7/2 and Pb 4f5/2 levels respectively, of the Pb2+ state, consistent with previous literature reports41,42 (Fig. 5b). Additionally, two Br 3d peaks are observed at 67.9 and 69.0 eV, corresponding to Br 3d5/2 and Br 3d3/2 contributions43,44 (Fig. 5c). Further examination of the Ti 2p spectrum for the NCs@POM–Ti2N composite reveals that the Ti 2p3/2 and Ti 2p1/2 levels corresponding to Ti–N in POM–Ti2N appear at BE values of 458.5 and 464.2 eV, respectively. The Ti 2p3/2 and Ti 2p1/2 levels associated with Ti–O bonds characterize the POM–Ti2N MXene, with various oxidation states from Ti2+ to Ti4+ presenting at BE values around 455.7–459.8 and 461–466 eV, respectively45,46 (Fig. 5d).
 | ||
| Fig. 5 XPS spectra of CsPbBr3 NCs, POM–Ti2N (below), and NCs@POM–Ti2N composites (top): (a) Cs 3d spectra, (b) Pb 4f spectra, (c) Br 3d spectra, (d) Ti 2p spectra, and (e) N 1s spectra. | ||
Further evidence of the formation of the composite NCs@POM–Ti2N is found in the XPS spectrum shown in Fig. 5, which shows the elemental XPS signals of CsPbBr3 and the POM–Ti2N MXene before and after the formation of the composite. Due to the effective formation of an interface between perovskites and the POM–Ti2N MXene, subtle shifts in the Pb, Br, Ti, and N peak positions are observed, as shown below. Specifically, the binding energies for Pb 4f and Br 3d in the NCs@POM–Ti2N composite shift towards more positive values, indicating a reduction in electron density on CsPbBr3.47 Conversely, the binding energies assigned to Ti 2p and N 1s (from Ti–N bonding) exhibit a negative shift, suggesting an increase in electron density on the POM–Ti2N MXene particles (Fig. 5d and e).48 The Ti 2p spectrum of the NCs@POM–Ti2N composite shows a clear shift of the main peak positions to lower BE values compared to the POM-Ti2N MXene alone, indicating a chemical interaction between the perovskite nanocrystals and the POM–Ti2N MXene structure.
In addition, despite partial oxidation of the nitride MXene, Ti–N bonds characteristic of titanium nitride predominate, as evidenced by Ti 2p3/2 and Ti 2p1/2 signals at binding energies of approximately 458.5 eV and 464.2 eV, respectively. Additionally, the presence of an N 1s signal around 399.5 eV confirms the continued existence of N–Ti bonds,49,50 and the peak at 400.8 eV may be associated with nitrogen atoms in the Ti(N, O) oxynitride lattice (Fig. 5e).
These findings indicate that, although some surface oxidation occurs, forming TiO2, the internal structure of POM–Ti2N predominantly retains its titanium–nitride bonds, thus facilitating an effective electronic interaction with the CsPbBr3 nanocrystals. In summary, all these observations support the notion of electronic interaction between the orbitals that form CsPbBr3 NCs and the POM–Ti2N MXene.
To investigate the reducibility and metal–support interactions within the hybrid, H2 temperature-programmed reduction (H2-TPR) measurements were conducted (Fig. 6a). Pure TiO2 exhibits only a weak, broad reduction event centred at ∼535 °C,51 characteristic of kinetically hindered lattice-oxygen removal and indicative of strongly bound oxygen species with low surface mobility. A similar trend has been observed for TiO2 nanosheets by other authors.52 Conversely, the POM–Ti2N composite exhibits a markedly different H2 consumption behavior, revealing two pronounced low-temperature reduction peaks with maxima centered at 314 °C and 381 °C. The presence of these features at significantly lower temperatures reflects the formation of highly labile surface oxygen species generated at the Ti2N/TiO2 interface.53 According to the reducibility criterion, where lower peak temperatures correspond to more weakly bonded oxygen species such as O−/O2−,54 these events confirm the emergence of readily accessible active oxygen centres not present in pristine TiO2. This enhanced reducibility is attributed to a strong metal–support interaction (SMSI) between the almost conductive Ti2N core and the ultrathin surface-oxidised TiO2 shell.55 The Ti2N framework promotes electron donation, activating H2 molecules and facilitating the formation of hydrides, which collectively weakens the surface Ti–O bonds and triggers stepwise reduction of the MXene surface. Concurrently, the high-temperature feature centred at ∼530 °C originates from the reduction of oxidised TiO2 moieties anchored on the POM surface, consistent with the consumption of more strongly bound lattice oxygen. Taken together, these redox characteristics demonstrate that the POM–Ti2N hybrid possesses substantially improved oxygen lability and electronic coupling compared to pristine TiO2. Such behaviour is essential for catalytic activation, as the generation of easily reducible oxygen species and the formation of a robust Ti2N–TiO2 heterojunction provide the active environment required for efficient H2 dissociation, CO2 adsorption, and subsequent photothermal reduction to CO and CH4.
Furthermore, the CO2 adsorption behaviour of POM–Ti2N and TiO2 was further examined by CO2 temperature-programmed desorption (CO2-TPD), as illustrated in Fig. 6b. POM–Ti2N and TiO2 exhibit a desorption peak in the low-temperature region (∼105 °C),56 which can be attributed to physisorbed CO2 weakly interacting with surface hydroxyl groups. In the case of TiO2, this low-temperature feature dominates the profile, indicating a limited population of strongly adsorbed carbonate species.57 In contrast, POM–Ti2N displays a markedly enhanced desorption signal extending into the high-temperature region (350–700 °C), with a pronounced maximum centred at approximately 525 °C. This peak reflects the formation of strongly bound carbonate species, resulting from the interaction of CO2 with basic surface sites generated during the formation of the TiO2 moieties within the POM–Ti2N heterostructure. The significantly increased desorption intensity suggests an enlarged density of active sites capable of stabilising chemisorbed CO2 species. These results collectively indicate that the incorporation of Ti2N and its partial oxidation to TiO2 enhance both the strength and capacity of CO2 adsorption.
Periodic DFT calculations were performed with VASP (PAW-PBE including dispersion corrections;58,59 full computational details are provided in the SI) on three representative Ti-based supports designed to mimic the POM–Ti2N composite: a Ti2N MXene monolayer (conductive nitride core), a rock-salt TiN(100) slab (nitride surface), and rutile TiO2 (110) surfaces with and without a surface oxygen vacancy, VO (partially oxidized TiO2/TiOx domains) (Fig. S15). CO2 and H2 were individually adsorbed at low coverage on each model, exploring several initial geometries. Among these, Ti2N exhibited the largest thermodynamic affinity for CO2 (Eads = −3.32 eV),60,61 whereas stoichiometric TiO2(110) bound to CO2 only weakly (Eads = −0.10 eV), in line with previous DFT and surface-science studies.62,63 Introducing a surface oxygen vacancy on TiO2(110) markedly strengthened CO2 adsorption (Eads = −1.11 eV) and led to bent, vacancy-anchored configurations, consistent with the enhanced reactivity of defective rutile surfaces.64–66 In contrast, molecular H2 adsorption was found to be very weak on all Ti-based supports (|Eads(H2)| ≤ 0.05 eV), while an artificially initialized, dissociated H2 state on Ti2N was strongly stabilized (Eads = −7.57 eV per H2), indicating that nitride sites act as a deep thermodynamic sink for atomic hydrogen rather than as strong chemisorption sites for molecular H2.67 Taken together with the H2-TPR and CO2-TPD data, these DFT trends support a bifunctional picture in which oxygen-deficient TiO2/TiOx regions and TiO2/Ti2N interfacial sites provide the main CO2-binding and activation centres, whereas the TiN/Ti2N scaffold supplies electronic/photothermal conductivity and preferentially stabilizes dissociated hydrogen species, as described in detail in the DFT analysis (see SI).
Catalytic activity of the NCs@POM–Ti2N material
First, the catalytic activity of NCs@POM–Ti2N was studied at temperatures ranging between 160 and 250 °C, either in the dark (thermal catalysis) or under light irradiation (photo-thermal catalysis). Fig. 7 and Fig. S16 and S17 summarize the catalytic results obtained under light and dark conditions. In both cases, under light or dark conditions, at low temperatures selectivity towards CH4 is 100% but as temperature increases selectivity shifts to CO. Additionally, maximum conversion is achieved above 200 °C, whereas at 250 °C the activity drops. This fact may be due to changes happening on the support as the colour of POM–Ti2N shifted from light grey to dark grey at these temperatures. On the other hand, as shown in Fig. 7, under light irradiation production of CO2-derived products is always higher compared to dark conditions. These differences in the catalytic activity indicate that light effectively assists the CO2 conversion reaction in NCs@POM–Ti2N.A deeper analysis of catalytic data summarized in Table 1 brings further arguments for the photo-thermal catalytic pathway, especially, which is more evident at lower temperatures. A comparison of CO2 conversion or CH4 production at temperatures of 160 °C or 180 °C leads to conversions 5 to 4-fold higher when photo-thermal conditions are applied. Interestingly, both thermal and photothermal conditions share similar trends displaying higher conversions at 220 °C; in both cases, CO production starts at 200 °C and has a Gaussian plot in which 220 °C seems to be the optimum temperature conditions. It seems that at higher temperatures than 180 °C desorption of reaction intermediates is favored and the complete hydrogenation of CO2 to CH4 is difficult.
| T reaction (°C) | Conditions | Selectivity for CH4 (%) | Selectivity for CO (%) |
|---|---|---|---|
| 160 | Dark | 100 | 0 |
| Light | 100 | 0 | |
| 180 | Dark | 100 | 0 |
| Light | 100 | 0 | |
| 200 | Dark | 70 | 30 |
| Light | 60 | 40 | |
| 220 | Dark | 36 | 64 |
| Light | 28 | 72 | |
| 250 | Dark | 22 | 78 |
| Light | 15 | 85 |
For comparison, we studied the photo-thermal activity for CO2 reduction using CsPbBr3 nanocrystals and CsPbBr3@POM–Ti2N MXene at 200 °C. (Fig. 8). As can be seen, our CsPbBr3@POM–Ti2N catalyst presents the highest activity for CH4 production compared to pristine CsPbBr3 NCs or POM–Ti2N MXene alone. In fact, while the components of the composite CsPbBr3 and M–Ti2N MXene are not as active as the pristine POM–Ti2N we assume that the catalytically active sites are present in the oxidized side of POM–Ti2N. (Fig. 8a) However, selectivity changes occurred when POM–Ti2N was loaded with the perovskite due to the suppression of CO production and a change in selectivity towards CH4. This indicates a synergistic effect between both materials when they form a combined material.
In addition, the recyclability of CsPbBr3@POM–Ti2N MXene was evaluated over five consecutive cycles under identical reaction conditions, as illustrated in Fig. 8b. The results indicate a stable CH4 production yield for the first three cycles (only a 3% reduction in activity per cycle), suggesting desirable photostability of the catalyst, even with each cycle lasting 23 hours. Interestingly, while CH4 remained the primary product, CO was observed as a byproduct of the reaction. We attribute this emergent CO formation to surface restructuring and the gradual alteration of active sites, likely due to subtle changes in the surface composition or morphology of CsPbBr3@POM–Ti2N during the initial reaction cycle. This shift in selectivity, with CO generation emerging, may be explained by changes in the binding and activation of CO2 at newly exposed or modified active sites on the catalyst surface. This argument is reinforced by XPS data shown in Fig. 5 which reveal the electronic polarization in Cs and Ti atoms after the formation of CsPbBr3@POM–Ti2N, suggesting that perovskite NCs are not randomly deposited, if not interacting somehow. Photocatalytic CO2 reduction involves a series of complex, multi-step processes, including light absorption, charge separation, reactant adsorption, and catalytic reduction at active sites.68 Minor modifications in any of these steps, particularly in the structure of surface-active sites, can significantly impact product selectivity. In this case, the MXene support likely plays a dual role: enhancing electron–hole separation and simultaneously protecting CsPbBr3 nanocrystals from rapid degradation, as confirmed by the absence of notable changes in XRD patterns (Fig. 1, black line) and consistent XPS spectra. Nevertheless, with continued cycling, slight restructuring of the surface may expose new catalytic sites that favor CO production. By the fourth cycle, a reduction of approximately 30% in CH4 yield was observed, possibly due to gradual surface degradation or partial deactivation of active sites, while CO production remained stable. This indicates that, despite a decrease in CH4 yield, the modified surface continues to facilitate CO2 reduction to CO, suggesting persistent reactivity at specific catalytic sites that favor CO production over CH4. In fact, while CsPbBr3@POM–Ti2N has higher selectivity towards CH4, its individual components (the M–Ti2N MXene, the POM–Ti2N MXene, and CsPbBr3 nanocrystals) are also selective to CO. This change in catalytic activity agrees with superficial chemicophysical changes on the POM–Ti2N surface. This matches with the observation made using XPS in terms of electronic densities of atomic elements that compose CsPbBr3 NCs and POM–Ti2N. These findings underscore the importance of optimizing both the initial surface properties and long-term stability of active sites to maintain selective CO2 photoreduction over extended reaction cycles.
Proposed mechanism (photocatalytic CO2 reduction)
According to XPS and UV-vis measurements, the energy alignments of the CB and VB of CsPbBr3 NCs, POM–Ti2N, and CsPbBr3@POM–Ti2N were calculated. From a thermodynamic point of view, the reduction of CO2 to CO and CH4 occurs in all cases, see Fig. 9b. However, CO2 conversion increases considerably in the case of the CsPbBr3@POM-Ti2N composite, especially upon UV-vis light exposure. This fact, plus the intrinsic heterojunction morphology of the composite (see Fig. 9a), suggests that photogenerated electrons can flow from the conduction band (CB) of the perovskite to the CB of the TiO2 layer that eventually will pass to Ti2N, thus inducing effective charge separation enabled by a type II heterojunction formed between the CsPbBr3 NCs and the POM-Ti2N MXene structure. Additionally, the difference in potential between the two materials generates an internal electric field (IEF) that further enhances charge separation and transfer efficiency.69 The direction of the IEF is shown by XPS results (Fig. 5).The TiO2 layer also reduces direct recombination between the perovskite and the MXene, forming an additional small electric field that promotes electron flow into the MXene and avoids recombination. The introduction of such an interlayer to reduce direct recombination is a well-known strategy in the field of dye-sensitized solar cells (DSSCs) and hybrid solar cells.70,71
Thus, these electrons that are injected into the MXene can reduce CO2 into either CO or CH4 through multielectron transfer pathways:72
| CO2 + 2H+ + 2e− → CO + H2O (E0redox = −0.53 V) | (2) |
| CO2 + 8H+ + 8e− → CH4 + 2H2O (E0redox = −0.24 V) | (3) |
Moreover, these reactions proceed concurrently, with their predominance governed by the interplay between thermodynamic feasibility and kinetic accessibility. Thermodynamically, the formation of CH4 is favored; however, kinetically, CO production is more accessible as it requires fewer electrons (2e−vs. 8e− for CH4). Experimental data from Table 1 and Fig. 7 confirm this duality. At lower temperatures (160–180 °C), CH4 selectivity remains at 100%, indicating a thermodynamically driven process. However, as the temperature increases (200–250 °C), a significant shift in selectivity toward CO is observed, dropping CH4 selectivity to 15% under illumination at 250 °C. These results suggest that at higher reaction temperatures, kinetic factors increasingly govern product distribution.
Therefore, the temperature-dependent behavior observed in the CsPbBr3@POM-Ti2N composite under light irradiation illustrates a nuanced balance between thermodynamics and kinetics.73
In summary, the superior photocatalytic activity of CsPbBr3@POM-Ti2N can be attributed to several synergistic factors. First, the composite exhibits enhanced light absorption across the visible spectrum compared to the individual components (Fig. 8a), generating more charge carriers. Second, the heterojunction architecture suppresses electron–hole recombination, thereby ensuring greater availability of charge carriers for surface redox reactions. Lastly, the 2D/0D structural configuration—where zero-dimensional CsPbBr3 NCs are uniformly distributed over a two-dimensional layers of POM–Ti2N support—maximizes the active surface area and facilitates efficient interfacial charge migration (Fig. 9).
3. Conclusions
In conclusion, this study highlights the successful synthesis and performance of a novel hybrid heterojunction composed of CsPbBr3 nanocrystals supported on a partially oxidized Ti2N MXene (POM-Ti2N), specifically engineered for efficient photo-thermal CO2 conversion into CH4 and CO. Preparation of the hybrid material was achieved by simple ultrasonic impregnation, enabling the uniform deposition of CsPbBr3 NCs onto the surface of the 2D POM–Ti2N sheets. The partial oxidation of Ti2N was fundamental to introducing oxygen-containing functional groups, which improved surface hydrophilicity and provided anchoring sites for the nanocrystals. This facilitated strong interfacial interactions, critical for enhancing the overall photocatalytic activity.The proposed mechanism involves a synergistic interplay between light absorption, charge separation, thermodynamic, and kinetic effects. CsPbBr3 NCs serve as efficient light harvesters due to their strong absorption in the visible range and excellent charge transport properties. Upon irradiation, electron–hole pairs are generated in the perovskite, with electrons being efficiently transferred to the POM-Ti2N support. The partially oxidized MXene acts both as a photothermal catalyst and an electron sink, avoiding charge recombination to promote the catalytic reduction of CO2, while simultaneously accelerating charge separation and transport. The presence of oxygen vacancies and various oxidation states from Ti2+ to Ti4+ on the POM–Ti2N surface further facilitates CO2 adsorption and activation, which are essential for driving the CH4 and CO formation pathway with high selectivity.
In terms of recyclability, the hybrid catalyst demonstrated excellent structural integrity, maintaining its catalytic activity over multiple reaction cycles. Post-reaction analyses showed minimal degradation of the CsPbBr3 NCs, retaining the interfacial architecture and confirming the robustness of the catalyst under operational conditions. Overall, the CsPbBr3@POM–Ti2N hybrid catalyst demonstrates a promising strategy for the design of stable and highly active photocatalysts, combining the tunable optoelectronic properties of perovskite NCs with the multifunctionality of 2D MXenes. The enhancement in CH4 and CO production under light-driven conditions underscores the potential of this hybrid catalyst in advancing solar-to-fuel technologies.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the results of this study are available from the corresponding author upon reasonable request.Additional supporting information can be found online in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01122a.
Acknowledgements
This research was funded by the Spanish Ministry of Science and Innovation through MCIN/AEI/10.13039/501100011033/and “FEDER a way of making Europe” and the Severo Ochoa Centre of Excellence program (CEX2021-001230-S). K. M. acknowledges the PRE2022-104119 grant for the project “Catalytic processes: catalysis for energy and process intensification by applying instrumental tools to monitor chemical processes.” P. A. received funding from the PID2021-123163OB-I00 grant. H. G. B. was supported by the RYC2022-037287-I grant, PAID-06-23. Also, this research leading to these results has received funding from the European Union's Horizon Europe research and innovation programme (Project DAM4CO2, Double-Active Membranes for a sustainable CO2 cycle; HORIZON-EIC-2022-PATHFINDERCHALLENGES-01-Number: 101115488). We thank the technical team at the Instituto de Tecnología Química (ITQ) for their support in facilitating the characterization.References
- X. Jiao, K. Zheng, L. Liang, X. Li, Y. Sun and Y. Xie, Fundamentals and Challenges of Ultrathin 2D Photocatalysts in Boosting CO2 Photoreduction, Chem. Soc. Rev., 2020, 49(18), 6592–6604, 10.1039/D0CS00332H.
- G. Liao, G. Ding, B. Yang and C. Li, Challenges in Photocatalytic Carbon Dioxide Reduction, Precis. Chem., 2024, 2(2), 49–56, DOI:10.1021/PRECHEM.3C00112/ASSET/IMAGES/LARGE/PC3C00112_0004.JPEG.
- J. Yu, A. Muhetaer, Q. Li and D. Xu, Solar Energy-Driven Reverse Water Gas Shift Reaction: Photothermal Effect, Photoelectric Activation and Selectivity Regulation, Small, 2024, 20(42), 2402952, DOI:10.1002/SMLL.202402952.
- Y. Zhang, L. Ma, T. Wang and X. Li, MnO2 Coated Fe2O3 Spindles Designed for Production of C5+ Hydrocarbons in Fischer–Tropsch Synthesis, Fuel, 2016, 177, 197–205, DOI:10.1016/J.FUEL.2016.03.023.
- P. Liu, Z. Huang, X. Gao, X. Hong, J. Zhu, G. Wang, Y. Wu, J. Zeng and X. Zheng, Synergy between Palladium Single Atoms and Nanoparticles via Hydrogen Spillover for Enhancing CO2 Photoreduction to CH4, Adv. Mater., 2022, 34(16), 2200057, DOI:10.1002/ADMA.202200057.
- N. D. M. Ridzuan, M. S. Shaharun, M. A. Anawar and I. Ud-Din, Ni-Based Catalyst for Carbon Dioxide Methanation: A Review on Performance and Progress, Catal., 2022, 12(5), 469, DOI:10.3390/CATAL12050469.
- S. Carenco, A. Tuxen, M. Chintapalli, E. Pach, C. Escudero, T. D. Ewers, P. Jiang, F. Borondics, G. Thornton, A. P. Alivisatos, H. Bluhm, J. Guo and M. Salmeron, Dealloying of Cobalt from CuCo Nanoparticles under Syngas Exposure, J. Phys. Chem. C, 2013, 117(12), 6259–6266, DOI:10.1021/JP4000297/SUPPL_FILE/JP4000297_SI_001.PDF.
- S. Xiao, L. Wang, Y. Tang, Z. Yang, H. Wang, C. Guo, T. Zhao, Y. Jiang, X. Wen and F. Wang, Interfacial Structure Engineering Enhances Photo-Thermal CO2 Hydrogenation over Ni-CeO2 Nanocomposites, Chem Catal., 2025, 5(7), 101361, DOI:10.1016/J.CHECAT.2025.101361.
- Y. Tang, H. Wang, C. Guo, L. Wang, T. Zhao, S. Xiao, J. Liu, Y. Jiang, Y. Zhao, X. D. Wen and F. Wang, Synergies Between Atomically Dispersed Ru Single Atoms and Nanoparticles on CeAlOx for Enhanced Photo-Thermal Catalytic CO2 Hydrogenation, Adv. Mater., 2025, e12793, DOI:10.1002/ADMA.202512793;PAGE:STRING:ARTICLE/CHAPTER.
- X. Li, C. Wang and J. Tang, Methane Transformation by Photocatalysis, Nat. Rev. Mater., 2022, 7(8), 617–632, DOI:10.1038/s41578-022-00422-3.
- T. Luo, L. Gilmanova and S. Kaskel, Advances of MOFs and COFs for Photocatalytic CO2 Reduction, H2 Evolution and Organic Redox Transformations, Coord. Chem. Rev., 2023, 490, 215210, DOI:10.1016/J.CCR.2023.215210.
- P. Wang, F. Yang, J. Qu, Y. Cai, X. Yang, C. M. Li and J. Hu, Recent Advances and Challenges in Efficient Selective Photocatalytic CO2 Methanation, Small, 2024, 20(32), 2400700, DOI:10.1002/SMLL.202400700.
- Z. Shang, X. Feng, G. Chen, R. Qin and Y. Han, Recent Advances on Single-Atom Catalysts for Photocatalytic CO2 Reduction, Small, 2023, 19(48), 2304975, DOI:10.1002/SMLL.202304975.
- S. Ullah, J. Wang, P. Yang, L. Liu, S. E. Yang, T. Xia, H. Guo and Y. Chen, All-Inorganic CsPbBr 3 Perovskite: A Promising Choice for Photovoltaics, Mater. Adv., 2021, 2(2), 646–683, 10.1039/D0MA00866D.
- Y. H. Chen, J. K. Ye, Y. J. Chang, T. W. Liu, Y. H. Chuang, W. R. Liu, S. H. Liu and Y. C. Pu, Mechanisms behind Photocatalytic CO2 Reduction by CsPbBr3 Perovskite-Graphene-Based Nanoheterostructures, Appl. Catal., B, 2021, 284, 119751, DOI:10.1016/J.APCATB.2020.119751.
- X. Wang, J. He, X. Chen, B. Ma and M. Zhu, Metal Halide Perovskites for Photocatalytic CO2 Reduction: An Overview and Prospects, Coord. Chem. Rev., 2023, 482, 215076 CrossRef CAS.
- X. Li, J. Liu, G. Jiang, X. Lin, J. Wang and Z. Li, Self-Supported CsPbBr3/Ti3C2Tx MXene Aerogels towards Efficient Photocatalytic CO2 Reduction, J. Colloid Interface Sci., 2023, 643, 174–182, DOI:10.1016/J.JCIS.2023.04.015.
- H. Pazniak, I. A. Plugin, M. J. Loes, T. M. Inerbaev, I. N. Burmistrov, M. Gorshenkov, J. Polcak, A. S. Varezhnikov, M. Sommer, D. V. Kuznetsov, M. Bruns, F. S. Fedorov, N. S. Vorobeva, A. Sinitskii and V. V. Sysoev, Partially Oxidized Ti3C2Tx MXenes for Fast and Selective Detection of Organic Vapors at Part-per-Million Concentrations, ACS Appl. Nano Mater., 2020, 3(4), 3195–3204, DOI:10.1021/ACSANM.9B02223/ASSET/IMAGES/MEDIUM/AN9B02223_M001.GIF.
- H. Wang, R. Peng, Z. D. Hood, M. Naguib, S. P. Adhikari and Z. Wu, Titania Composites with 2
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D Transition Metal Carbides as Photocatalysts for Hydrogen Production under Visible-Light Irradiation, ChemSusChem, 2016, 9(12), 1490–1497, DOI:10.1002/CSSC.201600165.
D Transition Metal Carbides as Photocatalysts for Hydrogen Production under Visible-Light Irradiation, ChemSusChem, 2016, 9(12), 1490–1497, DOI:10.1002/CSSC.201600165. - K. Vighnesh, S. Wang, H. Liu and A. L. Rogach, Hot-Injection Synthesis Protocol for Green-Emitting Cesium Lead Bromide Perovskite Nanocrystals, ACS Nano, 2022, 16(12), 19618–19625, DOI:10.1021/ACSNANO.2C11689/ASSET/IMAGES/LARGE/NN2C11689_0004.JPEG.
- X. Liu, Y. Li, H. Ding, L. Chen, S. Du, Z. Chai and Q. Huang, Topotactic Transition of Ti4AlN3 MAX Phase in Lewis Acid Molten Salt, J. Mater., 2023, 9(6), 1032–1038, DOI:10.1016/J.JMAT.2023.03.012.
- W. B. Wang, X. Zhong, Z. Y. He, Z. X. Wang and P. Z. Zhang, Plasma Niobium Surface Alloying of Pure Titanium and Its Oxidation at 900 °C, Chin. J. Aeronaut., 2007, 20(2), 111–114, DOI:10.1016/S1000-9361(07)60015-6.
- H. E. Lai, R. M. S. Yoo, A. Djire and P. B. Balbuena, Investigation of the Vibrational Properties of 2D Titanium Nitride MXene Using DFT, J. Phys. Chem. C, 2024, 128(8), 3327–3342, DOI:10.1021/ACS.JPCC.3C06717/ASSET/IMAGES/LARGE/JP3C06717_0012.JPEG.
- A. Balvanz, K. S. Bayikadi, Z. Liu, T. S. Ie, J. A. Peters and M. G. Kanatzidis, Unveiling the Monoclinic Phase in CsPbBr3-XClx Perovskite Crystals, Phase Transition Suppression and High Energy Resolution γ-Ray Detection, J. Am. Chem. Soc., 2024, 146, 31836–31848, DOI:10.1021/JACS.4C10872/ASSET/IMAGES/LARGE/JA4C10872_0006.JPEG.
- J. Casanova-Chafer, R. Garcia-Aboal, K. Mego, S. B. Malik, P. Atienzar and E. Llobet, Lead-Free Perovskite Nanocrystals Decorating Graphene for Detecting Nerve Agents, ACS Appl. Electron. Mater., 2024, 6(9), 6974–6981, DOI:10.1021/ACSAELM.4C01220/SUPPL_FILE/EL4C01220_SI_001.PDF.
- J. Low, L. Zhang, T. Tong, B. Shen and J. Yu, TiO2/MXene Ti3C2 Composite with Excellent Photocatalytic CO2 Reduction Activity, J. Catal., 2018, 361, 255–266, DOI:10.1016/J.JCAT.2018.03.009.
- S. Akhtar, S. Roy, T. T. Tran, J. Singh, A. S. Sharbirin and J. Kim, Low Temperature Step Annealing Synthesis of the Ti2AlN MAX Phase to Fabricate MXene Quantum Dots, Appl. Sci., 2022, 12(9), 4154, DOI:10.3390/APP12094154.
- A. S. Sharbirin, S. Roy, T. T. Tran, S. Akhtar, J. Singh, D. L. Duong and J. Kim, Light-Emitting Ti2N (MXene) Quantum Dots: Synthesis, Characterization and Theoretical Calculations, J. Mater. Chem. C, 2022, 10(16), 6508–6514, 10.1039/D2TC00568A.
- P. Mandal, A. Roy, S. Mannar and R. Viswanatha, Growth Mechanistic Insights into Perovskite Nanocrystals: Dimensional Growth, Nanoscale Adv., 2020, 2(11), 5305–5311, 10.1039/D0NA00732C.
- K. Mabhouti, P. Norouzzadeh and M. Taleb-Abbasi, Effects of Fe, Co, or Ni Substitution for Mn on La0.7Sr0.3MnO3 Perovskite: Structural, Morphological, and Optical Analyses, J. Non-Cryst. Solids, 2023, 610, 122283, DOI:10.1016/J.JNONCRYSOL.2023.122283.
- M. I. Saleem, S. Yang, M. Sulaman, J. Hu, P. V. Chandrasekar, Y. Shi, R. Zhi, A. Batool and B. Zou, All-Solution-Processed UV-IR Broadband Trilayer Photodetectors with CsPbBr3 Colloidal Nanocrystals as Carriers-Extracting Layer, Nanotechnology, 2020, 31(16), 165502, DOI:10.1088/1361-6528/AB667B.
- A. J. Chacón-García, H. G. Baldovi, A. A. Babaryk, A. Rodríguez-Diéguez, S. Navalón, Y. Pérez, H. García and P. Horcajada, Robust Hybrid Bismuth Perovskites as Potential Photocatalysts for Overall Water Splitting, Nano Res., 2024, 17(5), 4593–4601, DOI:10.1007/S12274-023-6254-1/METRICS.
- P. Salcedo-Abraira, R. Serrano-Nieto, C. Biglione, M. Cabrero-Antonino, S. M. F. Vilela, A. A. Babaryk, D. Tilve-Martínez, A. Rodriguez-Diéguez, S. Navalón, H. García and P. Horcajada, Two Cu-Based Phosphonate Metal-Organic Frameworks as Efficient Water-Splitting Photocatalysts, Chem. Mater., 2023, 35(11), 4211–4219, DOI:10.1021/ACS.CHEMMATER.3C00054/SUPPL_FILE/CM3C00054_SI_003.CIF.
- K. Mego, P. Ruiz-Campos, H. G. Baldoví and P. Atienzar, OER Activity Promoted by Organic Ligand-Free Cs2Pt(Cl, Br)6 Perovskite Photocatalyst for Solar-Driven Water Splitting, Adv. Energy Sustainability Res., 2025, 2500105, DOI:10.1002/AESR.202500105.
- A. Pan, X. Ma, S. Huang, Y. Wu, M. Jia, Y. Shi, Y. Liu, P. Wangyang, L. He and Y. Liu, CsPbBr3 Perovskite Nanocrystal Grown on MXene Nanosheets for Enhanced Photoelectric Detection and Photocatalytic CO2 Reduction, J. Phys. Chem. Lett., 2019, 10(21), 6590–6597, DOI:10.1021/acs.jpclett.9b02605.
- X. Tang, Z. Zu, Z. Zang, Z. Hu, W. Hu, Z. Yao, W. Chen, S. Li, S. Han and M. Zhou, CsPbBr3/Reduced Graphene Oxide Nanocomposites and Their Enhanced Photoelectric Detection Application, Sens. Actuators, B, 2017, 245, 435–440, DOI:10.1016/J.SNB.2017.01.168.
- J. Casanova-Chafer, R. Garcia-Aboal, E. Llobet and P. Atienzar, Enhanced CO2 Sensing by Oxygen Plasma-Treated Perovskite-Graphene Nanocomposites, ACS Sens., 2024, 9(2), 830–839, DOI:10.1021/ACSSENSORS.3C02166/ASSET/IMAGES/LARGE/SE3C02166_0008.JPEG.
- J. A. Peters, Z. Liu, R. Yu, K. M. McCall, Y. He, M. G. Kanatzidis and B. W. Wessels, Carrier Recombination Mechanism in CsPbB R3 Revealed by Time-Resolved Photoluminescence Spectroscopy, Phys. Rev. B, 2019, 100(23), 235305, DOI:10.1103/PHYSREVB.100.235305/FIGURES/6/MEDIUM.
- C. Maduwanthi, C. A. Jong, W. S. Mohammed and S. H. Hsu, Stability and Photocurrent Enhancement of Photodetectors by Using Core/Shell Structured CsPbBr3/TiO2 Quantum Dots and 2D Materials, Nanoscale Adv., 2024, 6(9), 2328–2336, 10.1039/D3NA01129A.
- D. Fang, F. He, J. Xie and L. Xue, Calibration of Binding Energy Positions with C1s for XPS Results, J. Wuhan Univ. Technol., Mater. Sci. Ed., 2020, 35(4), 711–718, DOI:10.1007/S11595-020-2312-7/METRICS.
- M. Li, X. Zhang, K. Matras-Postolek, H. S. Chen and P. Yang, An Anion-Driven Sn2+ Exchange Reaction in CsPbBr3 Nanocrystals towards Tunable and High Photoluminescence, J. Mater. Chem. C, 2018, 6(20), 5506–5513, 10.1039/C8TC00990B.
- X. Liu, S. Ren, Z. Li, J. Guo, S. Yi, Z. Yang, W. Hao, R. Li and J. Zhao, Flexible Transparent High-Efficiency Photoelectric Perovskite Resistive Switching Memory, Adv. Funct. Mater., 2022, 32(38), 2202951, DOI:10.1002/ADFM.202202951.
- P. Pandey, A. Sengupta, S. Parmar, U. Bansode, S. Gosavi, A. Swarnkar, S. Muduli, A. D. Mohite and S. Ogale, CsPbBr3-Ti3C2T x MXene QD/QD Heterojunction: Photoluminescence Quenching, Charge Transfer, and Cd Ion Sensing Application, ACS Appl. Nano Mater., 2020, 3(4), 3305–3314, DOI:10.1021/ACSANM.0C00051/SUPPL_FILE/AN0C00051_SI_001.PDF.
- X. Di, L. Shen, J. Jiang, M. He, Y. Cheng, L. Zhou, X. Liang and W. Xiang, Efficient White LEDs with Bright Green-Emitting CsPbBr3 Perovskite Nanocrystal in Mesoporous Silica Nanoparticles, J. Alloys Compd., 2017, 729, 526–532, DOI:10.1016/J.JALLCOM.2017.09.213.
- J. Halim, M. R. Lukatskaya, K. M. Cook, J. Lu, C. R. Smith, L. Å. Näslund, S. J. May, L. Hultman, Y. Gogotsi, P. Eklund and M. W. Barsoum, Transparent Conductive Two-Dimensional Titanium Carbide Epitaxial Thin Films, Chem. Mater., 2014, 26(7), 2374–2381, DOI:10.1021/CM500641A/SUPPL_FILE/CM500641A_SI_001.PDF.
- J. Halim, K. M. Cook, M. Naguib, P. Eklund, Y. Gogotsi, J. Rosen and M. W. Barsoum, X-Ray Photoelectron Spectroscopy of Select Multi-Layered Transition Metal Carbides (MXenes, Appl. Surf. Sci., 2016, 362, 406–417, DOI:10.1016/J.APSUSC.2015.11.089.
- C. Cheng, B. He, J. Fan, B. Cheng, S. Cao and J. Yu, An Inorganic/Organic S-Scheme Heterojunction H2-Production Photocatalyst and Its Charge Transfer Mechanism, Adv. Mater., 2021, 33(22), 2100317, DOI:10.1002/ADMA.202100317.
- Q. Tang, Z. Sun, S. Deng, H. Wang and Z. Wu, Decorating G-C3N4 with Alkalinized Ti3C2 MXene for Promoted Photocatalytic CO2 Reduction Performance, J. Colloid Interface Sci., 2020, 564, 406–417, DOI:10.1016/J.JCIS.2019.12.091.
- L. Liuhe, X. Lifang and M. Xinxin, XPS of Ti + TiN + (N,C) Multilayer Films Deposited by Filtered Cathodic Arc Deposition with Controlled Feed Gas Flow Rate, Surf. Coat. Technol., 1999, 120–121, 618–621, DOI:10.1016/S0257-8972(99)00426-0.
- D. C. Coyac, E. Kabachkov, E. Kurkin, N. Vershinin, I. Balikhin, V. Berestenko, A. Michtchenko, Y. Shulga, D. C. Coyac, E. Kabachkov, E. Kurkin, N. Vershinin, I. Balikhin, V. Berestenko, A. Michtchenko and Y. Shulga, Pt Supported on Plasma-Chemical Titanium Nitride for Efficient Room-Temperature CO Oxidation, Mod. Res. Catal., 2022, 11(1), 1–11, DOI:10.4236/MRC.2022.111001.
- H. Zhu, Z. Qin, W. Shan, W. Shen and J. Wang, Pd/CeO2–TiO2 Catalyst for CO Oxidation at Low Temperature: A TPR Study with H2 and CO as Reducing Agents, J. Catal., 2004, 225(2), 267–277, DOI:10.1016/J.JCAT.2004.04.006.
- C. Guo, L. Wang, Y. Tang, Z. Yang, Y. Zhao, Y. Jiang, X. Wen and F. Wang, Enhanced Photo-Thermal CO2 Methanation with Tunable RuxNi1-x Catalytic Sites: Alloying Beyond Pure Ru, Adv. Funct. Mater., 2025, 35(6), 2414931, DOI:10.1002/adfm.202414931.
- J. Wang, K. Vikrant, S. A. Younis, K. H. Kim and P. M. Heynderickx, Low-Temperature Oxidative Removal of Benzene from the Air Using Titanium Carbide (MXene)-Supported Platinum Catalysts, Chemosphere, 2024, 350, 141114, DOI:10.1016/J.CHEMOSPHERE.2024.141114.
- G. I. Panov, K. A. Dubkov and E. V. Starokon, Active Oxygen in Selective Oxidation Catalysis, Catal. Today, 2006, 117(1–3), 148–155, DOI:10.1016/J.CATTOD.2006.05.019.
- N. G. Akalework, C. J. Pan, W. N. Su, J. Rick, M. C. Tsai, J. F. Lee, J. M. Lin, L. D. Tsai and B. J. Hwang, Ultrathin TiO2-Coated MWCNTs with Excellent Conductivity and SMSI Nature as Pt Catalyst Support for Oxygen Reduction Reaction in PEMFCs, J. Mater. Chem., 2012, 22(39), 20977–20985, 10.1039/C2JM34361D.
- Y. Tang, H. Wang, C. Guo, Z. Yang, T. Zhao, J. Liu, Y. Jiang, W. Wang, Q. Zhang, D. Wu, Y. Zhao, X. D. Wen and F. Wang, Ruthenium–Cobalt Solid-Solution Alloy Nanoparticles for Enhanced Photopromoted Thermocatalytic CO2 Hydrogenation to Methane, ACS Nano, 2024, 18(17), 11449–11461, DOI:10.1021/ACSNANO.4C02416.
- W. N. R. Wan Isahak, Z. A. Che Ramli, M. W. Mohamed Hisham and M. A. Yarmo, The Formation of a Series of Carbonates from Carbon Dioxide: Capturing and Utilisation, Renewable Sustainable Energy Rev., 2015, 47, 93–106, DOI:10.1016/J.RSER.2015.03.020.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A Consistent and Accurate Ab Initio Parametrization of Density Functional Dispersion Correction (DFT-D) for the 94 Elements H-Pu, J. Chem. Phys., 2010, 132(15), 154104, DOI:10.1063/1.3382344/926936.
- S. Grimme, S. Ehrlich and L. Goerigk, Effect of the Damping Function in Dispersion Corrected Density Functional Theory, J. Comput. Chem., 2011, 32(7), 1456–1465, DOI:10.1002/JCC.21759;JOURNAL:JOURNAL:1096987X;WGROUP:STRING:PUBLICATION.
- A. Jurado, K. Ibarra, Á. Morales-García, F. Viñes and F. Illas, Adsorption and Activation of CO2 on Nitride MXenes: Composition, Temperature, and Pressure Effects, Chem. Phys. Chem., 2021, 22(23), 2456–2463, DOI:10.1002/CPHC.202100600;REQUESTEDJOURNAL:JOURNAL:14397641;PAGEGROUP:STRING:PUBLICATION.
- R. Morales-Salvador, J. D. Gouveia, Á. Morales-García, F. Viñes, J. R. B. Gomes and F. Illas, Carbon Capture and Usage by MXenes, ACS Catal., 2021, 11(17), 11248–11255, DOI:10.1021/ACSCATAL.1C02663.
- D. C. Sorescu, J. Lee, W. A. Al-Saidi and K. D. Jordan, CO2 Adsorption on TiO2(110) Rutile: Insight from Dispersion-Corrected Density Functional Theory Calculations and Scanning Tunneling Microscopy Experiments, J. Chem. Phys., 2011, 134(10), 104707, DOI:10.1063/1.3561300/70606.
- W. J. Yin, M. Krack, B. Wen, S. Y. Ma and L. M. Liu, CO2 Capture and Conversion on Rutile TiO2(110) in the Water Environment: Insight by First-Principles Calculations, J. Phys. Chem. Lett., 2015, 6(13), 2538–2545, DOI:10.1021/ACS.JPCLETT.5B00798.
- D. C. Sorescu, J. Lee, W. A. Al-Saidi and K. D. Jordan, Coadsorption Properties of CO2 and H2O on TiO2 Rutile (110): A Dispersion-Corrected DFT Study, J. Chem. Phys., 2012, 137(7), 74704, DOI:10.1063/1.4739088/192030.
- G. B. Raupp and J. A. Dumesic, Adsorption of Carbon Monoxide, Carbon Dioxide, Hydrogen, and Water on Titania Surfaces with Different Oxidation States, J. Phys. Chem., 2002, 89(24), 5240–5246, DOI:10.1021/J100270A024.
- B. Wei and M. Calatayud, Hydrogen Activation on Anatase TiO2: Effect of Surface Termination, Catal. Today, 2022, 397–399, 113–120, DOI:10.1016/J.CATTOD.2021.11.020.
- M. López, Á. Morales-García, F. Viñes and F. Illas, Thermodynamics and Kinetics of Molecular Hydrogen Adsorption and Dissociation on MXenes: Relevance to Heterogeneously Catalyzed Hydrogenation Reactions, ACS Catal., 2021, 11(21), 12850–12857, DOI:10.1021/ACSCATAL.1C03150.
- J. Fu, K. Jiang, X. Qiu, J. Yu and M. Liu, Product Selectivity of Photocatalytic CO2 Reduction Reactions, Mater. Today, 2020, 32, 222–243, DOI:10.1016/J.MATTOD.2019.06.009.
- Y. Zhang, W. Chen, M. Zhou, G. Miao and Y. Liu, Efficient Photocatalytic CO2Reduction by the Construction of Ti3C2/CsPbBr3QD Composites, ACS Appl. Energy Mater., 2021, 4(9), 9154–9165, DOI:10.1021/ACSAEM.1C01406/ASSET/IMAGES/MEDIUM/AE1C01406_M008.GIF.
- E. Palomares, J. N. Clifford, S. A. Haque, T. Lutz and J. R. Durrant, Control of Charge Recombination Dynamics in Dye Sensitized Solar Cells by the Use of Conformally Deposited Metal Oxide Blocking Layers, J. Am. Chem. Soc., 2003, 125(2), 475–482, DOI:10.1021/JA027945W/ASSET/IMAGES/LARGE/JA027945WF00006.JPEG.
- P. Atienzar, T. Ishwara, B. N. Illy, M. P. Ryan, B. C. O’Regan, J. R. Durrant and J. Nelson, Control of Photocurrent Generation in Polymer/ZnO Nanorod Solar Cells by Using a Solution-Processed TiO2 Overlayer, J. Phys. Chem. Lett., 2010, 1(4), 708–713, DOI:10.1021/JZ900356U/ASSET/IMAGES/LARGE/JZ-2009-00356U_0003.JPEG.
- X. Chang, T. Wang and J. Gong, CO2 Photo-Reduction: Insights into CO2 Activation and Reaction on Surfaces of Photocatalysts, Energy Environ. Sci., 2016, 9(7), 2177–2196, 10.1039/C6EE00383D.
- L. Hu, X. Sai, X. Liu, Z. Chen, G. Wang and X. Yi, Influence of Environmental Conditions on Electrocatalytic CO2 Reduction, ChemCatChem, 2024, 16(6), e202301335, DOI:10.1002/CCTC.202301335.
| This journal is © The Royal Society of Chemistry 2026 |