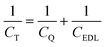Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Large improvement in the charge storage performance of a CoSb2O6–reduced graphene oxide (rGO) composite – probing the role of rGO through experiments and theoretical analyses
Parul
 a,
Surjit
Sahoo
a,
Surjit
Sahoo
 b,
M.
Amrutha
f,
Satyajit
Ratha
b,
M.
Amrutha
f,
Satyajit
Ratha
 c,
Brahmananda
Chakraborty
c,
Brahmananda
Chakraborty
 *de and
Saroj Kumar
Nayak
*a
*de and
Saroj Kumar
Nayak
*a
aIndian Institute of Technology Bhubaneswar, Argul, Jatani, Khordha, Odisha-752050, India. E-mail: nayaks@iitbbs.ac.in
bCentre for Interdisciplinary Research, SRM University-AP, Amaravati, Andhra Pradesh 522240, India
cInstitute of Technical Education and Research, S‘O’A Deemed to be University, Bhubaneswar, Odisha 751030, India
dHigh Pressure & Synchrotron Radiation Physics Division, Bhabha Atomic Research Centre, Trombay, Mumbai-400085, India. E-mail: brahma@barc.gov.in
eHomi J Bhabha National Institute, Bhabha Atomic Research Centre, Trombay, Mumbai-400085, India
fDepartment of Physics, Karpagam Academy of Higher Education (Deemed to be University), Coimbatore-641021, India
First published on 9th January 2026
Abstract
We present a detailed electrochemical comparison between cobalt antimony oxide (CoSb2O6) and its reduced graphene oxide (rGO) composite, prepared through a combination of hydrothermal and ultrasonic synthesis. The structural, morphological, and compositional properties of the as-synthesized material have been investigated through comprehensive characterization techniques. In a three-electrode configuration, the CoSb2O6/rGO composite exhibited a specific capacitance value of 1000 F g−1 at a mass-normalized current of 2 A g−1, which is at least 5-fold higher than that of pristine CoSb2O6 (i.e., 195.5 F g−1 at a mass-normalised current of 2 A g−1). It shows a high capacity retention of 93.18% over 3000 cycles. We have also tested a two-electrode symmetric device, fabricated from the CoSb2O6/rGO composite, which shows a specific capacitance of 227.79 F g−1 at a mass-normalised current of 1 A g−1. The CoSb2O6/rGO composite yields energy and power densities of 38.28 Wh kg−1 and 10.08 kW kg−1, respectively, and exhibits a cyclic stability of 98.54% even after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. Furthermore, we have employed density functional theory simulations to elucidate the structure and electronic characteristics of pristine CoSb2O6 and CoSb2O6/rGO composite systems. The enhancement in the electronic states near the Fermi level, resulting from the charge transfer from rGO to CoSb2O6, indicates improved conductivity of the CoSb2O6/rGO composite system. The considerably lower diffusion energy barrier and enhanced quantum capacitance of CoSb2O6/rGO compared to CoSb2O6 contribute to the superior supercapacitance performance, which supports our experimental findings.
000 cycles. Furthermore, we have employed density functional theory simulations to elucidate the structure and electronic characteristics of pristine CoSb2O6 and CoSb2O6/rGO composite systems. The enhancement in the electronic states near the Fermi level, resulting from the charge transfer from rGO to CoSb2O6, indicates improved conductivity of the CoSb2O6/rGO composite system. The considerably lower diffusion energy barrier and enhanced quantum capacitance of CoSb2O6/rGO compared to CoSb2O6 contribute to the superior supercapacitance performance, which supports our experimental findings.
1. Introduction
The demand for efficient, high-performance energy storage devices has become more important for powering a wide range of applications, including portable devices, electric cars, and smart city infrastructure. Supercapacitors have drawn significant interest because of their high power density, extended life cycle, and rapid charging time.1 Nevertheless, conventional supercapacitors frequently experience bottlenecks in terms of energy density, prompting the addition of innovative materials to improve their performance.2 Recently, there has been growing interest in utilizing hybrid materials to enhance charge storage performance. Despite the potential of these hybrid materials, issues persist in optimizing the energy–power spectrum for the device.3 Different types of electrode materials have been investigated for supercapacitors, e.g., carbonaceous materials, transition metal oxides (TMOs), and transition metal chalcogenides (TMCs). TMOs such as V2O5,4 NiO,5 MoO2,6 and MnO27 exhibit rich redox mechanisms, making them suitable for the fabrication of a pseudocapacitive electrode.8 Many binary metal oxides or some battery-type materials like NiCo2O4,9 CuFeS2,10 CuFe2O4,11 CuCo2O4,12 and CuWO413 have been reported to show good electrochemical performance for supercapacitors. With the rapid development of wearable, portable, and flexible electronic devices, there is a need for high-performance supercapacitors that combine long cycle stability, high power density, operational safety, and mechanical flexibility for practical applications. Recent works have shown that multifunctional electrodes, such as Cu3Mo2O9 nanostructures, are used in self-powered integration systems.14 So, to improve the electrochemical performance, we need to focus on key factors like high surface area, pore size distribution, and cyclic stability of the electrode materials. EDLCs (electric double-layer capacitors showing surface adsorption at the electrode and electrolyte interface) typically exhibit high specific power and long cycle life, while pseudocapacitors (which rely on reversible fast redox reactions) have an improved specific energy profile. Combining these two materials will integrate the electrochemical functionalities in a single system and contribute to the development of more sustainable energy storage solutions for the future, thus achieving a very good power–energy (storage property) balance along with good cycle performance (lifetime of the material/device).15 Moreover, hydrogel-based electrolytes such as PVA and S-PANa hydrogels have enabled flexible and safe devices that further expand practical applications.16 Such reports highlight the growing importance of a versatile system for integration and next-generation energy storage technologies.Within this expanding materials landscape, metal antimonates have emerged as a new class of promising electrode materials due to their stable crystal structure and strong faradaic redox activity. Here, we have synthesized cobalt antimony oxide, a pseudocapacitive material, and prepared its composite with reduced graphene oxide, aiming to improve the charge storage performance of the supercapacitor. We systematically optimize the weight percentage of reduced graphene oxide (i.e., variation within 5% to 20% by weight of cobalt antimony oxide) and observe the change in electrochemical performance depending on the variation of the rGO content.17 According to the reported literature, CoOx,18 Co3O4,19,20 Sb-doped Co3O4,21 and Sb2O322 show potential applications in various fields like energy storage, catalysis, and gas sensing.23 Co3O4, with multiple morphologies, has been widely used in supercapacitor applications. Using the successive ionic layer adsorption and reaction (SILAR) method, a thin cobalt oxide film on a copper substrate exhibited a specific capacitance of 165 F g−1.18 In another work, porous Co3O4 nanocubes were synthesized by a hydrothermal method, yielding a specific capacitance of 430.6 F g−1.19 Furthermore, electrodeposited cobalt oxide on a copper substrate exhibited a specific capacitance of 235 F g−1.20 Co2+ induced deposition in an Sb2O5/rGO composite showed enhanced interfacial interactions, showing potential for high-performance Li-ion/Na-ion storage applications.24 The incorporation of antimony enhances the electrochemical performance of the electrode. Sb can form binary and ternary alloys with metals like cobalt, as in CoSb2O6, which enhances both structural integrity and conductivity, leading to improved electrochemical performance. It has been observed that Sb can act as an essential dopant species, which can tune the electrochemical properties of the electrode material.21 Cobalt antimony oxide (CoSb2O6), which has a rutile-type structure,23 presents intriguing applications in both sodium- and lithium-ion batteries as an anode active material,25 catalysis, and the detection of various types of gases in sensors.26 However, the inclusion of antimony enhances the surface area, generates an increased number of redox-active sites, and improves the stability of the electrode, significantly strengthening the electrochemical performance. One of the strategic steps to further enhance the cycle life of CoSb2O6 (a pseudocapacitive material) is by combining it with a carbonaceous material like reduced graphene oxide (because of its high electrical conductivity, large surface area, and chemical stability)27 to form a composite/hybrid system. Several reports have observed that pristine metal oxides perform poorly compared with their rGO composites, indicating the significance of rGO in a composite/hybrid system. Despite these promising features, reports on the systematic optimization of CoSb2O6–rGO composite electrodes for supercapacitor applications are rare. Combining cobalt antimony oxide nanoparticles with a reduced graphene oxide substrate creates a three-dimensional conductive structure with improved mechanical and electrochemical stability, which has not been thoroughly explored. Addressing these gaps is critical for identifying the optimal composite structure and maximizing the redox activity of CoSb2O6 while benefiting from the excellent conductivity of rGO. Furthermore, the synergistic effect of rGO and cobalt antimony oxide, in the as-obtained composite, resulted in high electrochemical performance.
In this work, we compare the electrochemical performances of pristine cobalt antimony oxide and its rGO composite using state-of-the-art analytical methods. First, we discuss the detailed synthesis method for preparing the composite, which combines a hydrothermal reaction and ultrasonic treatment. In the next step, we perform detailed structural and morphological analyses on the samples to assess their dimensionality, crystal structure, and purity. Afterwards, we carry out detailed electrochemical analyses to determine the charge storage properties of the material in both three- and two-electrode configurations in an aqueous electrolyte medium.28 To validate our experimental findings and gain insight into the electronic properties and the interaction between CoSb2O6 and rGO, we implemented first-principles calculations based on density functional theory (DFT).
2. Experimental section
2.1 Chemicals
Cobalt(ous) chloride hexahydrate [CoCl2·6H2O, 99%] and antimony trichloride [SbCl3, 99%] were procured from the Central Drug House Pvt Ltd (India). Ammonia solution [NH3, 25%] and N-methyl-2-pyrrolidone [C5H9NO, 99.7%] were procured from Molychem, India. Graphene oxide [15–20 sheets] was purchased from Sigma-Aldrich. Poly(vinylidene fluoride) (PVDF) powder [(–CH2CF2–)]n and conductive carbon black [99+%, C] were procured from Alfa Aesar, India. All chemicals were of analytical grade and used without any further purification/modification.2.2 Synthesis of cobalt antimony oxide (CoSb2O6)
A hydrothermal approach was used to synthesize cobalt antimony oxide.29 First, solutions of 0.1 M cobalt chloride (dissolved in 20 ml of distilled water) and 0.1 M antimony trichloride (dissolved in 40 ml of distilled water) were combined and mixed in a beaker containing a total amount of 100 ml of distilled water, followed by ultrasonication for 10 minutes. Subsequently, the beaker was placed on a magnetic stirrer, and ammonia solution was gradually added until the pH reached approximately 9. The obtained solution was transferred to a Teflon-lined stainless-steel autoclave and placed in a digital hot-air oven for 16 hours at a temperature of 160 °C. Then, the product was centrifuged with ethanol and water repeatedly until the pH became neutral. Once all these procedures were completed, the sample was dried in an oven for 8 hours at a temperature of 80 °C. Lastly, the product was calcined in a furnace for 5 hours at 500 °C to remove volatile substances and obtain a stable product.2.3 Synthesis of the cobalt antimony oxide/reduced graphene oxide (CoSb2O6/rGO) composite
The cobalt antimony oxide/reduced graphene oxide composite was prepared using the ultra-sonication-assisted physical mixture method.30 Here, we synthesized different cobalt antimony oxide/reduced graphene oxide composites by varying the weight percentage of rGO (CoSb2O6–5% rGO, CoSb2O6–10% rGO, CoSb2O6–15% rGO, CoSb2O6–20% rGO). Firstly, different weight percentages (i.e., 5%, 10%, 15%, and 20%) of graphene oxide powder and the prepared cobalt antimony oxide nanoparticles were dispersed in ethanol by ultrasonication for 30 minutes (at 10-min intervals), which promoted exfoliation of rGO sheets and facilitated uniform anchoring of the CoSb2O6 particles onto the surface of rGO. Then the mixture was ground for 2 hours at room temperature to ensure uniform dispersion of CoSb2O6 particles on rGO sheets, and the reduction of graphene oxide was achieved by calcining the composite mixture for 1 h at 200 °C. Thus, the cobalt antimony oxide/reduced graphene oxide composite was obtained as the final product. The schematic diagram of the synthesis of cobalt antimony oxide and its rGO composite is shown in Fig. 1. | ||
| Fig. 1 Schematic representation of the preparation process of nanostructured CoSb2O6 and CoSb2O6-reduced graphene oxide composite materials, along with their future applications. | ||
2.4 Physical characterization
The XRD analyses were conducted using a Bruker D8 Advance X-ray diffractometer (operating at 40 kV and 40 mA, with Ni-filtered Cu-Kα radiation). Surface morphology was studied by field emission scanning electron microscopy (Merlin Compact with Gemini 1 electron column). An Oxford Instruments EDX spectrometer was used for energy dispersive X-ray spectroscopy (EDX). The chemical composition of pristine CoSb2O6 and the CoSb2O6–15% rGO composite was analyzed by X-ray photoelectron spectroscopy (XPS) using a Thermo Fisher Scientific ESCALAB instrument. The surface area and pore volume of cobalt antimony oxide and its reduced graphene oxide composites (CoSb2O6–5% rGO, CoSb2O6–10% rGO, CoSb2O6–15% rGO, and CoSb2O6–20% rGO) were determined using Brunauer–Emmett–Teller (BET) analysis explicitly using a Quantachrome Nova 2200 instrument. Additionally, high-resolution morphological, elemental, and compositional information was obtained using high-resolution transmission electron microscopy (HR-TEM, JEOL JEM 2100 Plus). Room temperature Raman spectra were recorded using a Renishaw InVia Raman microscope with 532 nm laser excitation.2.5 Electrochemical characterization
All electrochemical characterizations (cyclic voltammetry, galvanostatic charge–discharge cycling, and electrochemical impedance spectroscopy) were conducted using a multi-channel BioLogic electrochemical workstation (VSP-300). Here, nickel foam was used as the substrate for all as-synthesized materials. Initially, nickel foam was cleaned with diluted hydrochloric acid in a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and then gently washed with deionized water. After that, it was dried for 6 hours at 80 °C. For the electrode coating, a slurry was prepared in a weight ratio of 80
3 and then gently washed with deionized water. After that, it was dried for 6 hours at 80 °C. For the electrode coating, a slurry was prepared in a weight ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5. This included an 80% weight ratio of the active material, a 15% weight ratio of carbon black as the conductive material, in order to reduce the effect of the binder, and a 5% weight ratio of PVDF as the binding material, and the mixture was homogenously dispersed by using N-methyl-2-pyrrolidone (NMP). The slurry was mixed until a homogeneous mixture was obtained. Then, a slurry coating method (using the brush-paint technique) was employed to coat the nickel foam, followed by drying overnight at 80 °C. The mass loading for cobalt antimony oxide and its reduced graphene oxide composites was ∼1 mg. These steps were consistently applied to all rGO composites.
5. This included an 80% weight ratio of the active material, a 15% weight ratio of carbon black as the conductive material, in order to reduce the effect of the binder, and a 5% weight ratio of PVDF as the binding material, and the mixture was homogenously dispersed by using N-methyl-2-pyrrolidone (NMP). The slurry was mixed until a homogeneous mixture was obtained. Then, a slurry coating method (using the brush-paint technique) was employed to coat the nickel foam, followed by drying overnight at 80 °C. The mass loading for cobalt antimony oxide and its reduced graphene oxide composites was ∼1 mg. These steps were consistently applied to all rGO composites.
Three-electrode measurements were performed on cobalt antimony oxide and its reduced graphene oxide composites. The working electrode was coated on the nickel foam, with Ag/AgCl as the reference electrode and a platinum mesh electrode as the counter electrode. 1 M aqueous sodium sulphate was used as the electrolyte for all electrochemical measurements. A two-electrode (symmetric) configuration was employed to evaluate the charge storage performance of the CoSb2O6–15% rGO composite, where a glass fiber membrane/pre-filter (purchased from Merck Millipore Ltd) was used as the separator to isolate the electrodes. The specific capacitance (Csp) values of cobalt antimony oxide and its reduced graphene oxide composite electrodes were calculated from the cyclic voltammetry profiles and galvanostatic charge–discharge cycles using eqn (1) and (2), respectively:31
 | (1) |
| Csp = (I × Δt)/(ΔV × m) | (2) |
| Ed = (1/2 × Csp × (ΔV)2)/3.6 | (3) |
| Pd = Ed/Δt × 3600 | (4) |
2.6 Computational details
All simulations were conducted using density functional theory (DFT) within the context of the projector-augmented wave (PAW) method33 employing the Vienna ab initio simulation package (VASP).34 The generalized gradient approximation (GGA) method35 was employed to describe the exchange–correlation function, which accounts for ion–electron interactions. The structures were optimized until the force and energy convergence thresholds reached 0.01 eV Å−1 and 10−5 eV, respectively. The cut-off energy for the plane wave basis set expansion was set to 500 eV. A gamma-centred k-point mesh with grids of 4 × 5 × 1 was used for sampling the reciprocal space. To eliminate periodic interactions, a vacuum spacing of 30 Å was introduced above the surface of the monolayer in the supercell. Moreover, Grimme's DFT-D2 dispersion correction method36 was applied to account for van der Waals (vdW) interactions effectively.3. Results and discussion
3.1 Structural, morphological, and elemental analyses
Hereafter, CoSb2O6/rGO composites containing 5%, 10%, 15%, and 20% of rGO are denoted as CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20 for convenience. X-ray diffraction was used to analyze the material's crystal structure, phase, space group, and crystallite size. Fig. 2(a) compares CoSb2O6 and CoSb2O6/rGO composites, which perfectly match the JCPDS (reference) file no. 01-084-2062.37 All the peaks present in XRD correspond to the Bragg's angle at 19.12°, 21.36°, 27.101°, 33.380°, 34.817°, 38.701°, 39.336°, 43.51°, 53.030°, 55.886°, 59.767°, 63.189°, 66.813°, 67.554°, 73.505°, 80.664°, 83.260°, 86.310°, and 89.566° can be assigned to the (002), (101), (110), (112), (103), (004), (113), (210), (211), (213), (220), (006), (310), (116), (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 223), (206), (323), (008), (226), and (118) planes showing the crystal system of tetragonal structured CoSb2O6 and CoSb2O6/rGO composites. It shows a highly crystalline structure with growth oriented along the (110) lattice plane and the P42/mnm space group. To validate the lattice parameters a, b, and c, we have calculated d-spacing using Bragg's law as shown in eqn (5):
223), (206), (323), (008), (226), and (118) planes showing the crystal system of tetragonal structured CoSb2O6 and CoSb2O6/rGO composites. It shows a highly crystalline structure with growth oriented along the (110) lattice plane and the P42/mnm space group. To validate the lattice parameters a, b, and c, we have calculated d-spacing using Bragg's law as shown in eqn (5):nλ = 2d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ θ |
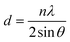 | (5) |
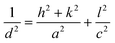 | (6) |
D = (kλ)/(β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ) θ) | (7) |
 | ||
| Fig. 2 (a) Comparison of the X-ray diffraction (XRD) patterns of CoSb2O6 and CoSb2O6/rGO composites; (b) Raman spectra of rGO, CoSb2O6, and its rGO composites. | ||
To explore more structural information, Raman spectroscopy was further employed. It is an essential tool for confirming the CoSb2O6/rGO composite phase, providing information on lattice defects. The Raman spectra of rGO, CoSb2O6, and its rGO composites are shown in Fig. 2(b). The Raman spectra of GO and rGO are shown in Fig. S3. The graphitic materials exhibit first-order bands at 1347 cm−1 and 1343 cm−1 (D), and 1570 cm−1 and 1571 cm−1 (G) for GO and rGO, respectively. rGO shows a moderate defect intensity (ID/IG), i.e., 0.47, and the corresponding plane crystallite size (La) is ∼40.9 nm (calculated using the Tuinstra–Koenig relation given in eqn (8)), considering partial restoration of the sp2 domain after reduction.39 In Fig. 2(b), CoSb2O6 displays three distinct phonon peaks at 514, 678, and 716 cm−1, which correspond to the vibrational modes of the cobalt antimony oxide lattice and confirm the formation of the oxide phase. All these oxide modes are present in all composites, indicating that the oxide phase is preserved upon compositing with rGO. In CoSb2O6/rGO composites, the Raman features of both CoSb2O6 and rGO are present and show systematic changes with varying rGO loadings (10–15%) (shown in Table S3). The D and G band positions for all composites are nearly constant, but the ID/IG ratio and calculated La values change with different compositions. From this, we measured the ID/IG ratio (Table S3) and calculated the crystallite sizes (La) using the Tuinstra–Koenig relation (i.e., eqn (8))40 (at 532 nm excitation) to be 36.26 (GO), 40.89 (rGO), 33.72 (5%), 30.51 (10%), and 26.94 (15%). This trend indicates the introduction of additional defects and distortion in the sp2 lattice, arising from strong interfacial interactions and nucleation of CoSb2O6 and rGO. The CoSb2O6/rGO_15 composite exhibits the highest defect density, providing abundant anchoring sites for nanoparticles, increasing the redox activity due to its accessible surface area, and enabling faster electron transfer.
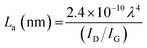 | (8) |
XPS was performed to examine the chemical composition and electronic states of CoSb2O6 and CoSb2O6/rGO_15. The high-resolution XPS spectra of Co 2p, Sb 3d, O 1s, and C 1s are shown in Fig. 3(a)–(d). The complete XPS survey spectrum of CoSb2O6 and CoSb2O6/rGO_15 is shown in Fig. 2(a). The XPS spectra were deconvoluted using Fityk software, resulting in perfect curve fitting. Fig. 2(b) shows the high-resolution Co 2p spectra of both CoSb2O6 and CoSb2O6–15% rGO, which were fitted using a Voigt profile (G![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) L = 70
L = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) and a Shirley-type background with two doublet peaks at 796.4–796.5 eV and 780.7 eV, corresponding to 2p1/2 and 2p3/2, respectively. In addition to these prominent peaks, two satellite peaks are observed at 802.6 eV and 785.95 eV. These satellite peaks are about 5–6 eV higher in binding energy than the prominent peaks, indicating that the electronic state of Co is approaching the +2 oxidation state, which is in good agreement with previously reported literature.41 The interaction with rGO modifies the electronic structure but does not alter the dominant oxidation state. Comparing the two samples, the Co 2p peaks in the CoSb2O6/rGO_15 composite show a slight binding energy shift, i.e., ∼0.1 to 0.7 eV. This shift indicates an increase in partial electron density around Co2+. In addition, the Co2+ peaks in the composite become noticeably sharper and more intense, reflecting a reduction in surface defects due to rGO addition, which enhances charge-transfer capability through the conductive rGO framework. In Fig. 2(c), the Sb 3d and O 1s core-level spectra of both pristine CoSb2O6 and the CoSb2O6–15% rGO composite were re-deconvoluted to address the overlap between Sb 3d5/2 and O 1s signals. Two high-resolution Sb 3d peaks are observed at 530.5–530.6 eV and 539.8–540.0 eV, corresponding to the 3d5/2 and 3d3/2 states, respectively. Due to the spin–orbit coupling, the energy separation between these two peaks is about 9–9.5 eV, corresponding to the higher number of oxidation states (+5) of antimony (Sb). Additionally, the peak shift in CoSb2O6/rGO_15 compared to pristine CoSb2O6 at 540 eV indicates increasing Sb5+ character.42 In CoSb2O6, minor peaks at 530.35 and 539.82 eV suggested the presence of reduced Sb species (Sb3+). The O–C peak at 532.09 eV was assigned to surface-adsorbed species or oxygen vacancies. A broad peak of O 1s in CoSb2O6/rGO_15 at 530.99 eV suggests overlapping with lattice oxygen, hydroxyl groups, and potentially functional groups from rGO. The XPS data of O 1s are shown in Fig. S4(a) and (b) for both pristine CoSb2O6 and the CoSb2O6–15% rGO composite. One peak is observed at 530.59 eV for O 1s, which is closely related to the binding energy of Sb 3d. The XPS peaks for C 1s are shown in Fig. 2(d); the main peak of carbon is observed at 284.09 eV, corresponding to C–C/C
30) and a Shirley-type background with two doublet peaks at 796.4–796.5 eV and 780.7 eV, corresponding to 2p1/2 and 2p3/2, respectively. In addition to these prominent peaks, two satellite peaks are observed at 802.6 eV and 785.95 eV. These satellite peaks are about 5–6 eV higher in binding energy than the prominent peaks, indicating that the electronic state of Co is approaching the +2 oxidation state, which is in good agreement with previously reported literature.41 The interaction with rGO modifies the electronic structure but does not alter the dominant oxidation state. Comparing the two samples, the Co 2p peaks in the CoSb2O6/rGO_15 composite show a slight binding energy shift, i.e., ∼0.1 to 0.7 eV. This shift indicates an increase in partial electron density around Co2+. In addition, the Co2+ peaks in the composite become noticeably sharper and more intense, reflecting a reduction in surface defects due to rGO addition, which enhances charge-transfer capability through the conductive rGO framework. In Fig. 2(c), the Sb 3d and O 1s core-level spectra of both pristine CoSb2O6 and the CoSb2O6–15% rGO composite were re-deconvoluted to address the overlap between Sb 3d5/2 and O 1s signals. Two high-resolution Sb 3d peaks are observed at 530.5–530.6 eV and 539.8–540.0 eV, corresponding to the 3d5/2 and 3d3/2 states, respectively. Due to the spin–orbit coupling, the energy separation between these two peaks is about 9–9.5 eV, corresponding to the higher number of oxidation states (+5) of antimony (Sb). Additionally, the peak shift in CoSb2O6/rGO_15 compared to pristine CoSb2O6 at 540 eV indicates increasing Sb5+ character.42 In CoSb2O6, minor peaks at 530.35 and 539.82 eV suggested the presence of reduced Sb species (Sb3+). The O–C peak at 532.09 eV was assigned to surface-adsorbed species or oxygen vacancies. A broad peak of O 1s in CoSb2O6/rGO_15 at 530.99 eV suggests overlapping with lattice oxygen, hydroxyl groups, and potentially functional groups from rGO. The XPS data of O 1s are shown in Fig. S4(a) and (b) for both pristine CoSb2O6 and the CoSb2O6–15% rGO composite. One peak is observed at 530.59 eV for O 1s, which is closely related to the binding energy of Sb 3d. The XPS peaks for C 1s are shown in Fig. 2(d); the main peak of carbon is observed at 284.09 eV, corresponding to C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C. Some other peaks are observed at 284.93 eV and 288.15 eV due to the respective functional groups C–OH (carboxylic) and C
C. Some other peaks are observed at 284.93 eV and 288.15 eV due to the respective functional groups C–OH (carboxylic) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (carbonyl).43 The dominant sp2 carbon peak reflects the presence of conductive graphene sheets. Meanwhile, the presence of oxygen-functionalized carbon peaks enhances electrolyte diffusion. The SI provides detailed data on XPS in a tabular form (Table S5 for pristine CoSb2O6 and Table S6 for CoSb2O6/rGO_15). When comparing the XPS spectra between CoSb2O6 and CoSb2O6/rGO_15, a minor chemical peak shift is present in CoSb2O6–15% rGO due to the addition of rGO to CoSb2O6.
O (carbonyl).43 The dominant sp2 carbon peak reflects the presence of conductive graphene sheets. Meanwhile, the presence of oxygen-functionalized carbon peaks enhances electrolyte diffusion. The SI provides detailed data on XPS in a tabular form (Table S5 for pristine CoSb2O6 and Table S6 for CoSb2O6/rGO_15). When comparing the XPS spectra between CoSb2O6 and CoSb2O6/rGO_15, a minor chemical peak shift is present in CoSb2O6–15% rGO due to the addition of rGO to CoSb2O6.
To analyse the porosity (pore diameter and pore volume), the surface area of the nanoparticles was measured using N2 adsorption and desorption isotherms (BET isotherms). Fig. 3(e) shows that all samples exhibit type IV isotherms with noticeable hysteresis loops (H3). The surface area of CoSb2O6/rGO_15 was determined to be 50 m2 g−1. Fig. 3(e) compares the BET isotherms of cobalt antimony oxide and its rGO composites. A comparison of the specific surface area, pore diameter, and pore volume is presented in Table 1. As shown in Table 1, when the weight percentage of rGO increases, the specific surface area also increases from 16.671 m2 g−1 for pristine CoSb2O6 to 61.804 m2 g−1 for 20% (by wt%) rGO in CoSb2O6. The maximum pore diameter ranges between 3 and 4 nm, indicating a mesoporous structure of cobalt antimony oxide and its rGO composites. This structure offers a large number of electrochemically active sites, thereby facilitating rapid ion absorption and faster charge transfer at the electrode–electrolyte interface.44 The highest pore diameter of CoSb2O6/rGO_15 is 3.82 nm, which significantly improves the electrochemical charge storage performance. After the addition of rGO, the surface area and porosity increase, which is favorable for electrochemical performance. Subsequently, the pore-size distribution graph of the synthesized materials is presented in Fig. 3(f) for further clarification. These graphs show the presence of mesoporous particles in their structure. As the percentage of reduced graphene oxide increases, the pore size also increases. Here, the pore size distribution of CoSb2O6/rGO_15 and CoSb2O6/rGO_20 is almost the same, even with a 5% increase in rGO, suggesting that the distribution remains the same. Therefore, the improved specific capacitance value can be attributed to the increased percentage of rGO, offering a large surface area.45
| Sample name | Surface area (m2 g−1) | Pore diameter (nm) | Pore volume (CC g−1) |
|---|---|---|---|
| CoSb2O6 | 16.67 | 3.05 | 0.0714 |
| CoSb2O6/rGO_5 | 24.20 | 3.40 | 0.1475 |
| CoSb2O6/rGO_10 | 35.24 | 3.39 | 0.1062 |
| CoSb2O6/rGO_15 | 50.00 | 3.82 | 0.1030 |
| CoSb2O6/rGO_20 | 61.80 | 3.38 | 0.1291 |
Field emission scanning electron microscopy was used to examine the surface morphology of CoSb2O6 and its rGO composites. Fig. 4 shows the micrographs of pristine CoSb2O6 and CoSb2O6/rGO_15. Fig. 4(a)–(c) shows the micrographs of CoSb2O6 at different magnifications and scales (i.e., 1 µm, 200 nm, and 200 nm). In these micrographs, we can observe the uniform formation of nanoparticles. Some of the nanoparticles show a larger size, which is due to the agglomeration effect between the nearby interconnected nanoparticles. Fig. 4(d)–(f) shows the micrographs of the CoSb2O6/rGO_15 composite at different magnifications and scales (i.e., 1 µm, 200 nm, and 200 nm), where the yellow circle indicates the rGO sheet in the composite and the red circle indicates the cobalt antimony decorated with the reduced graphene oxide composite. From Fig. 4(d)–(f), we can observe that all nanoparticles are uniformly distributed on the surface of reduced graphene oxide. Reduced graphene oxide behaves as a conductive substrate for the cobalt antimony oxide particles, which enhances the electrochemical performance.46 Fig. S5(a)–(c) of the SI show the SEM images of all other composites. The micrographs of CoSb2O6/rGO_5, CoSb2O6/rGO_10, and CoSb2O6/rGO_20 taken at a 50 KX magnification, shown in Fig. S5(a)–(c), demonstrate a uniform distribution of the reduced graphene oxide sheets. Energy dispersive spectroscopy (EDS) was used to determine the elemental composition. Fig. S6(a)–(e) shows cobalt antimony oxide with the presence of all elements: Co, Sb, and O. The measured Co![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Sb ratio is ∼1
Sb ratio is ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, which agrees with the composition of trirutile type CoSb2O6. A minor deviation from the ideal 1
2, which agrees with the composition of trirutile type CoSb2O6. A minor deviation from the ideal 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 stoichiometry in the oxygen content is expected because the EDS instrument shows limited sensitivity to the lighter elements, which tends to underestimate O relative to Co or Sb. Fig. S7(A)–(D) shows the elemental composition of CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively. Fig. S7[A(a)–(f)], [B(a)–(f)], [C(a)–(f)], and [D(a)–(f)] shows the elemental composition of CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively.
6 stoichiometry in the oxygen content is expected because the EDS instrument shows limited sensitivity to the lighter elements, which tends to underestimate O relative to Co or Sb. Fig. S7(A)–(D) shows the elemental composition of CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively. Fig. S7[A(a)–(f)], [B(a)–(f)], [C(a)–(f)], and [D(a)–(f)] shows the elemental composition of CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively.
HR-TEM images are presented in Fig. 5 to further understand the structure and morphology. Fig. 5(a)–(e) shows the morphology of CoSb2O6/rGO_15 at different scales from 500 to 5 nm. In Fig. 5(e), the d-spacing is approximately 0.32 nm, which corresponds to the (110) lattice plane of CoSb2O6, and the inset shows the selected area electron diffraction pattern, which reveals the polycrystalline nature of CoSb2O6/rGO_15. Fig. 5(f) shows the EDS image of CoSb2O6/rGO_15 at a scale of 500 nm, which reveals the presence of all elements in the layers.47 Fig. S8 shows the HR-TEM images, SAED pattern, and the bare cobalt antimony oxide's particle size. Fig. S8(a)–(f) shows the morphology of CoSb2O6 at different scales, from 200 nm to 5 nm. The inset of Fig. S8(a) shows the histogram of the particle size distribution corresponding to the 200 nm image, with the average particle size, measured from the histogram, being 36.61 nm. The HR-TEM image in Fig. S8(e) shows d-spacings of 0.3 nm and 0.46 nm, corresponding to the (111) and (101) planes of CoSb2O6, respectively. This interplanar spacing showed good agreement with the XRD data. Fig. S8(f) presents the SAED pattern for cobalt antimony oxide. The bright points in the SAED pattern indicate the highly crystalline structure of CoSb2O6. The concentric rings of nanoparticles correspond to the (101), (110), (112), (004), (213), and (310) planes of CoSb2O6, which are in good agreement with the XRD pattern. Additionally, the EDS analysis conducted during HR-TEM for CoSb2O6 and CoSb2O6/rGO_15, shown in Fig. S9(a)–(f) and Fig. 5(g)–(k), confirms the presence of all the desired elements and compositions. The above analysis confirms the successful formation of pure CoSb2O6 and CoSb2O6/rGO_15 composite nanostructures. The structure is nearly uniform in shape and size, making it highly suitable for supercapacitor applications.
3.2 Electrochemical analysis
To understand the electrochemical kinetics of cobalt antimony oxide and its rGO composite (at different weight percentages), we performed cyclic voltammetry (CV), galvanostatic charge–discharge cycling (GCD), and electrochemical impedance spectroscopy (EIS). First, we conducted three-electrode measurements for CoSb2O6 and all its rGO composites to enable systematic comparisons. The potential window for all CV curves was 0–1.1 V vs. Ag/AgCl in 1 M aqueous Na2SO4 electrolyte. Fig. 6(a) presents the CV comparison plot for rGO, pristine CoSb2O6, and CoSb2O6/rGO_15 (which shows the best performance among all composite materials) at 100 mV s−1. This plot shows that rGO displays a quasi-rectangular shape, revealing the material's capacitive or EDLC nature.48 For pristine CoSb2O6, the capacitance originated from the pseudocapacitance mechanism. The addition of 15% rGO to pristine CoSb2O6 leads to an increase in both area and peak current, resulting from the synergistic effect of rGO, which provides a more conductive surface for pristine CoSb2O6 nanoparticles.49 The cyclic voltammetry curve of CoSb2O6/rGO_15 is illustrated in Fig. 6(b) at various scan rates ranging from 5 mV s−1 to 100 mV s−1. The increase in the scan rate influences the peak current and potential response. The shape of the CV curves shows oxidation and reduction peaks, which clarify the redox behavior of CoSb2O6/rGO_15, indicating the pseudocapacitive behavior of the electrode material.50 At high scan rates, we observe that the peak shape is less sharp compared to low scan rates, indicating the presence of high diffusive resistance. Conversely, the diffusive resistance decreases at lower scan rates, resulting in the occurrence of anodic and cathodic peaks at nearly identical potentials. This is because the longer time to complete the cycle allows for greater interaction between the electrode and electrolytic ions, reflecting the electrochemically reversible nature of the electrode. From Fig. 6(c), which displays the plot of scan rate vs. specific capacitance, we calculate a maximum capacitance of 483.52 F g−1 at a scan rate of 5 mV s−1 for CoSb2O6/rGO_15. This value is four times greater than the specific capacitance value of 115.2 F g−1 for pristine CoSb2O6, indicating that rGO addition enhances the material's electrochemical performance.51 The comparison of cyclic voltammetry curves for pristine CoSb2O6, CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20 is shown in Fig. S11. Fig. S11(a)–(e) shows the CV plots of pristine CoSb2O6, CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively. To calculate the contribution of nickel (Ni) foam to the capacitance, the electrochemical response of bare Ni foam is presented in Fig. S10 of the SI. The bare Ni foam shows a negligible contribution to the tested potential window, confirming that its influence on the measured capacitance value is less.We can observe that the peak current response also increases as the weight percentage of rGO increases. This increase is due to the addition of rGO, which enhances the charge transfer at the electrode and electrolyte interface and provides a shorter diffusion length for the interaction of electrolytic ions. However, from Fig. S11(d) and (e), we see that, after adding an additional 5% rGO, the current response does not significantly increase and remains similar to that observed with the addition of 15% CoSb2O6. Furthermore, there is no significant increase in capacitance.52 The area under the curve and shape of all CV tests exhibit symmetric curves, and as the weight percentage increases, the area under the curve also expands. The reason behind this improvement with 20% rGO arises from the EDLC contribution, not from the faradaic contribution. rGO mainly contributes to the electric double-layer capacitance, while CoSb2O6 contributes to the pseudocapacitance. Pseudocapacitance sites saturate at 15%; an additional 5% rGO improves only the EDLC contribution, resulting in a smaller capacitance gain. Another reason is that increasing the rGO content reduces the relative mass fraction of CoSb2O6, which carries the majority of the pseudocapacitive charge. We observe a smaller increase in specific capacitance when adding 5% additional rGO to the EDLC (15% behavior). This combined effect explains why rGO shows very little increase in the capacitance relative to 15% rGO. From this cyclic voltammetry test, we can conclude that CoSb2O6/rGO_15 shows better electrochemical performance.
The galvanostatic charge–discharge cycling for pristine CoSb2O6 and its rGO composites was performed in a potential window of 0–1.1 V vs. Ag/AgCl using the same electrolyte. Fig. 6(d) shows the comparative GCD cycle for rGO, pristine CoSb2O6, and CoSb2O6/rGO_15 at a mass-normalised current value of 2 A g−1. Fig. 5(d) shows that rGO shows a triangular-shaped charge–discharge curve. In contrast, pristine CoSb2O6 deviates from the generally triangular shape of EDLC behavior, which is attributed to the surface diffusion control mechanism.53 However, the CoSb2O6/rGO_15 curve shows clear plateaus representing the electrode material's more diffusion-controlled or pseudocapacitive behavior. A detailed GCD study of CoSb2O6/rGO_15 is presented in Fig. 6(e) at various constant current values ranging from 2 A g−1 to 5 A g−1. This plot reveals a symmetrical and reversible curve for all current values.54 As the current value increases, the time taken to complete the cycle decreases, indicating that a large amount of charge is being transferred at low current values as the interaction time increases. At high current values, ions from the electrolyte do not get enough time to interact, and the inactive diffusion reaction activity is attributed to the incomplete coverage of the electrode surface, resulting in a lower capacitance value. From this analysis, we calculate the specific capacitance at different values, as shown in Fig. 6(f). The high capacitance value at 2 A g−1 is 1000 F g−1, varying as follows: 1000 F g−1, 821.7 F g−1, 707.07 F g−1, 620.15 F g−1, 565.33 F g−1, and 544.07 F g−1 at mass-normalised current values of 2 A g−1, 2.5 A g−1, 3 A g−1, 3.5 A g−1, 4 A g−1, 4.5 A g−1, and 5 A g−1, respectively. Given that the mass loading is 1 mg, a maximum specific capacitance value of 1000 F g−1 occurs at a mass-normalised current value of 2 A g−1, which is approximately 5-fold higher than that of pristine CoSb2O6 (i.e., 195.5 F g−1 at a mass-normalised current value of 2 A g−1). The detailed study of all electrode materials is shown in Fig. S12(a)–(f). Fig. S12(a)–(e) presents the GCD curves of CoSb2O6, CoSb2O6/rGO_5, CoSb2O6/rGO_10, CoSb2O6/rGO_15, and CoSb2O6/rGO_20, respectively. Pristine CoSb2O6 does not exhibit a proper EDLC triangular shape and shows some deviation. However, with increasing the percentage of rGO in pristine CoSb2O6, the time taken to complete the cycle increases and the shape of the curve changes to reflect a more diffusive contribution.55 It is observed that the presence of 20% rGO leads to an increased cycle time compared to the 15% rGO electrode. However, after increasing the rGO content to 20% (i.e., 15% + 5% more), the additional impact of rGO beyond 15% is negligible. As illustrated in Fig. S12(f), the comparison of specific capacitance at a current value of 2 mA reveals that the increase of specific capacitance for 5%, 10%, and 15% rGO is almost symmetrical. Nevertheless, the composite with 20% rGO shows increased specific capacitance, which is, however, not significant. Therefore, the increase beyond 15% may decrease the performance due to increased resistance across the reduced graphene oxide layers. From this analysis, we conclude that the presence of 15% rGO in pristine CoSb2O6 leads to a higher capacitance value of 1000 F g−1 at a mass-normalised current value of 2 A g−1. This enhancement is attributed to the synergistic effect of the EDLC charge storage of rGO and the pseudocapacitive charge storage mechanism of CoSb2O6, demonstrating that rGO addition enhances the material properties and electrochemical reaction pathways. We have calculated the specific capacity from the GCD curves of CoSb2O6 and its rGO composite, as shown in Fig. S13 (and the calculated values are presented in Table S6). The maximum specific capacity of CoSb2O6/rGO_15 is 1100 C g−1 at an applied mass-normalised current of 2 A g−1, whereas the pristine CoSb2O6 electrode exhibits a specific capacity of 215.05 C g−1 under the same conditions.
Electrochemical impedance spectroscopy (EIS) is a crucial technique for studying the electrochemical kinetics of supercapacitive electrode materials. Fig. 6(g) presents the Nyquist plot, with a zoomed-in view covering the frequency range of 100 kHz–0.1 Hz for rGO, CoSb2O6, and CoSb2O6/rGO_15. From this plot, we analyze the electrode material's electrolytic or solution resistance (Rs), which appears in the high-frequency region. The diameter of the semicircle in the high-frequency region represents the charge transfer resistance (Rct) at the electrode–electrolyte interface. In contrast, the line in the low-frequency region corresponds to the Warburg region.56 For pristine CoSb2O6, the solution resistance is 2.12 Ω and the charge transfer resistance is 3.11 Ω. To improve performance and reduce resistance, we developed a composite with rGO that exhibits the lowest solution resistance of 1.42 Ω, significantly lower than that of pristine CoSb2O6. Adding rGO enhances the performance of the CoSb2O6/rGO_15 composite, which has a solution resistance of 1.09 Ω. The semicircle's diameter near the real axis shows that CoSb2O6/rGO_15 has a lower Rct value of ∼2.58 Ω, indicating faster charge transfer at the electrode–electrolyte interface. The lower resistance in the composite material corresponds to improved electrochemical performance, contributing to higher specific capacitance. The EIS fitted parameters are presented in Table S9 of the SI. A Bode plot was generated to examine the deviation from the ideal capacitor angle (−90°) and the charge storage mechanism.57 The characteristic frequency, f0, for the phase angle −45° marks the point at which resistive and capacitive impedance are equal. f0 represents a time constant, 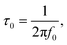 which defines how fast the device can charge and discharge. From Fig. S14, the phase angle for the CoSb2O6/rGO_15 composite electrode is −45.97°, indicating a predominant pseudocapacitive or diffusive charge storage behavior. For this Bode plot, the corresponding time constant is 0.836 ms. This lower time constant confirms excellent rate capability and fast ion transfer. Additionally, cyclic stability and capacity retention were tested over 3000 cycles. Fig. 6(h) shows that the CoSb2O6/rGO_15 composite retains 93.18% of its capacity after 3000 cycles.
which defines how fast the device can charge and discharge. From Fig. S14, the phase angle for the CoSb2O6/rGO_15 composite electrode is −45.97°, indicating a predominant pseudocapacitive or diffusive charge storage behavior. For this Bode plot, the corresponding time constant is 0.836 ms. This lower time constant confirms excellent rate capability and fast ion transfer. Additionally, cyclic stability and capacity retention were tested over 3000 cycles. Fig. 6(h) shows that the CoSb2O6/rGO_15 composite retains 93.18% of its capacity after 3000 cycles.
To quantitatively understand the charge storage process, the Trasatti plot was analyzed for both pristine CoSb2O6 and CoSb2O6/rGO_15, which allows us to assess both capacitive and pseudocapacitive contributions.58 Fig. S15(a) and (c) shows the y-intercept of the linear fit of 1/Cpvs. v1/2, while Fig. S15(b) and (d) presents the y-intercept of the linear fit of Cspvs. v−1/2 for pristine CoSb2O6 and the CoSb2O6/rGO_15 composite electrode. Then, we analyzed the overall percentage of capacitive and pseudocapacitive contributions. Fig. S15(e) shows that pristine CoSb2O6 exhibits an EDLC contribution of 6.98%, while the pseudocapacitive contribution is 93.02%. After adding rGO into CoSb2O6, the EDLC contribution increases to 15.08%, while the pseudocapacitive contribution decreases to 84.92%, facilitating faster charge transfer. The large surface area of rGO allows more ions to interact with the electrode material, enhancing storage performance.
We have employed the Dunn method to distinguish between the capacitive and pseudocapacitive effects.59 This helps clarify which reaction dominates in the respective electrode materials. For a quantitative description of both capacitive and diffusive control effects, we calculated the b-value using the following equation:
| ip = avb | (9) |
log(ip) = b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) log(v) + log log(v) + log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a | (10) |
Here, ip is the peak current, v is the scan rate, and a and b are constants. Plots of log(v) vs. log(i) for pristine CoSb2O6 and CoSb2O6/rGO_15 are shown in Fig. S16(a) and (b). The cathodic and anodic peak current values from the CV plot yielded b-values of 0.57 for the cathodic peak and 0.73 for the anodic peak for pristine CoSb2O6, suggesting a mixed mechanism of capacitive and diffusive contributions. A b-value near 1 suggests a capacitive contribution, while a b-value close to 0.5 indicates a diffusive contribution, with values between 0.5 and 1 representing pseudocapacitive contributions.60
Similarly, for CoSb2O6/rGO_15, the b-values were approximately 0.66 for the cathodic peak and 0.65 for the anodic peak, indicating that adding rGO reduces the resistance and enhances the charge transfer characteristics. The following equation was used to determine the respective contributions from both processes:
| i(v) = k1v = k2v1/2 | (11) |
We conducted two-electrode measurements on the CoSb2O6/rGO_15 composite symmetric supercapacitive device. The photographic image of the symmetric supercapacitor device is shown in Fig. S18. To assess the electrochemical behavior of the device, we performed CV, CD, and EIS measurements. Fig. 7(a) shows the CV measurement of symmetric devices at different scan rates. Particularly at higher scan rates, the CV curves show sharp redox peaks, signifying that redox behavior plays a crucial role in determining the capacitance of the symmetric device.61,62 Even at high potential sweep rates, consistent findings are observed at low to high scan rates, indicating excellent reversibility. Fig. 7(b) shows the GCD curve for 10 continuous cycles at a mass-normalised current value of 2.5 A g−1. Fig. 7(c) shows the GCD plot for the symmetric device at different current densities, ranging from 1 A g−1 to 5 A g−1. We calculated the capacitance from these data, revealing remarkable output values for the device. The specific capacitance at different mass-normalised current values is presented in Fig. 7(d), with a maximum capacitance of 227.79 F g−1 recorded at 1 A g−1. EIS was performed in the frequency range of 100 kHz to 0.1 Hz to further understand the device's chemical kinetics, as shown in Fig. 7(e). The calculated solution resistance (Rs) is 1.02 Ω, and the charge transfer resistance (Rct) is 2.25 Ω. These low values of Rs and Rct confirm the excellent performance of the symmetric device. Additionally, the Bode plot, displayed in Fig. 7(f), shows a phase angle of approximately −64.415°, indicating both EDLC and pseudocapacitive contributions, and for the symmetric device, the calculated response time τ0 is 0.439 ms. A lower time response for the device indicates faster ion transport and faster charge transportation through the device. To quantify the specific energy and power, the Ragone plot, in Fig. 7(g), has been generated, showing a maximum energy density of 38.28 Wh kg−1 with a corresponding power density of 1837.44 W kg−1 and a maximum power density of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 082.19 W kg−1. The Ragone plot indicates the high specific capacitance of the CoSb2O6/rGO_15 composite symmetric supercapacitive device. It shows a comparison with the performance of other SSD configurations such as the Mn0.2Co0.8 LDH (9.5 Wh kg−1),63 CoSe2 (21.1 Wh kg−1),64 Co(OH)2/GF (18 Wh kg−1),65 mesoporous carbon/MnO2 (2.32 Wh kg−1),66 ZnO (3.1 Wh kg−1),67 ZnO/GNR (6.8 Wh kg−1),67 and NPCs (16.9 Wh kg−1).68 A 10
082.19 W kg−1. The Ragone plot indicates the high specific capacitance of the CoSb2O6/rGO_15 composite symmetric supercapacitive device. It shows a comparison with the performance of other SSD configurations such as the Mn0.2Co0.8 LDH (9.5 Wh kg−1),63 CoSe2 (21.1 Wh kg−1),64 Co(OH)2/GF (18 Wh kg−1),65 mesoporous carbon/MnO2 (2.32 Wh kg−1),66 ZnO (3.1 Wh kg−1),67 ZnO/GNR (6.8 Wh kg−1),67 and NPCs (16.9 Wh kg−1).68 A 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000-cycle test was conducted at a mass-normalised current of 5 A g−1 to measure the electrode's stability, as shown in Fig. 7(h). The results indicate a capacity retention of 98.54% after 10
000-cycle test was conducted at a mass-normalised current of 5 A g−1 to measure the electrode's stability, as shown in Fig. 7(h). The results indicate a capacity retention of 98.54% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. For this cyclic stability, the coulombic efficiency remained close to 100% throughout the 10
000 cycles. For this cyclic stability, the coulombic efficiency remained close to 100% throughout the 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, as shown in Fig. S19, confirming the stability and absence of parasitic side reactions. For further clarification, Fig. S20 compares the first ten cycles with the last ten cycles, which reveals no substantial difference, affirming the device's exceptional stability. For the reproducibility analysis of the symmetric CoSb2O6/rGO_15 device based on five different fabricated cells, we present this analysis graph in Fig. S21 of the SI with the corresponding error bars (±SD). The standard deviation across the five devices was found to be 1.258, indicating very small device-to-device variations.
000 cycles, as shown in Fig. S19, confirming the stability and absence of parasitic side reactions. For further clarification, Fig. S20 compares the first ten cycles with the last ten cycles, which reveals no substantial difference, affirming the device's exceptional stability. For the reproducibility analysis of the symmetric CoSb2O6/rGO_15 device based on five different fabricated cells, we present this analysis graph in Fig. S21 of the SI with the corresponding error bars (±SD). The standard deviation across the five devices was found to be 1.258, indicating very small device-to-device variations.
3.3 Theoretical analysis
where D(E) and ϕG imply the density of states and electrode potential of the system under study, respectively, while the function FT(E) represents the thermal broadening owing to the thermal effect.
The function FT(E) can be expressed as
| FT(E) = (4kBT)−1sech2(E/2kBT) |
where CEDL stands for electric double-layer capacitance, which depends on the interfacial interaction between the electrode and electrolyte.
Fig. 9(b) shows the variation of quantum capacitance concerning the electrode potential for CoSb2O6 and CoSb2O6/rGO systems. Noticeably, the quantum capacitance is higher for most of the CoSb2O6/rGO composite electrode potential than for pristine CoSb2O6. At the Fermi level, the value of quantum capacitance for CoSb2O6 and CoSb2O6/rGO is 173.85 and 310.32 mF cm−2, respectively. The higher quantum capacitance of the CoSb2O6/rGO composite leads to a higher total capacitance, thereby achieving better charge storage performance than pristine CoSb2O6, further supporting our experimental results. Fig. S23 presents the variation of quantum capacitance for rGO. Here, we can observe that the quantum capacitance plot of rGO is graphene-like except for the band gap opening due to the introduction of a functional group, which results in a dip at the Fermi level.75
4. Conclusion
In summary, we report CoSb2O6 and CoSb2O6/rGO composites with different weight ratios of rGO (5%, 10%, 15%, and 20%) synthesized by a hydrothermal reaction and ultrasonic treatment, which show uniform morphology. The CoSb2O6/rGO_15 composite enables a high specific capacitance of ∼1000 F g−1 at a mass-normalised current of 2 A g−1. The combination of CoSb2O6 and rGO improves the conductivity and stability of pristine CoSb2O6, resulting in enhanced electrochemical performance of the material. Moreover, when the composite material was assembled into a symmetric supercapacitor device, it exhibited energy and power densities of 38.28 Wh kg−1 and 10.08 kW kg−1, respectively, and excellent cyclic stability of 98.54% even after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles. The structure and electronic properties, diffusion energy barrier, and the quantum capacitance calculated using density functional theory simulations corroborate our experimental results. The interaction of CoSb2O6 and rGO due to the charge transfer from rGO to CoSb2O6 resulted in an enhancement of electronic states near the Fermi level for the CoSb2O6/rGO composite system. The elevated quantum capacitance and lowered diffusion energy barrier contribute to the enriched charge storage and supercapacitance performance of the CoSb2O6/rGO composite system, supporting our experimental findings.
000 cycles. The structure and electronic properties, diffusion energy barrier, and the quantum capacitance calculated using density functional theory simulations corroborate our experimental results. The interaction of CoSb2O6 and rGO due to the charge transfer from rGO to CoSb2O6 resulted in an enhancement of electronic states near the Fermi level for the CoSb2O6/rGO composite system. The elevated quantum capacitance and lowered diffusion energy barrier contribute to the enriched charge storage and supercapacitance performance of the CoSb2O6/rGO composite system, supporting our experimental findings.
Author contributions
Parul: experimental analysis and writing of the original manuscript draft. Surjit Sahoo: technical discussion and editing. Amrutha M: DFT calculations and theoretical draft write-up. Satyajit Ratha: conceptualization and manuscript editing. Brahmananda Chakraborty: theoretical data supervision and DFT analysis. Saroj Kumar Nayak: manuscript supervision, technical discussion, and funding acquisition. All authors have approved the submission of the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma00956a.Acknowledgements
The authors acknowledge the financial assistance from NALCO (RP-274), DST, India, and the Ministry of Mines (RP-333) extended to carry out part of this research. Surjit Sahoo gratefully acknowledges the financial support from the Department of Science and Technology (DST), India, under the DST-INSPIRE Faculty Award [DST/INSPIRE/04/2023/000200]. The authors thank Dr Sk Musharaf Ali and Dr Saju Joseph for their constant support and encouragement. They also thank the BARC supercomputing facility and its staff for their valuable assistance and support.References
- S. Sahoo, S. Ratha, C. S. Rout and S. K. Nayak, Self-charging supercapacitors for smart electronic devices: a concise review on the recent trends and future sustainability, J. Mater. Sci., 2022, 57(7), 4399–4440, DOI:10.1007/s10853-022-06875-9.
- S. Sarkis and X. Huang, Cobalt Nanorods as Transition Metal Electrode Materials for Asymmetric Supercapacitor Applications, J. Phys. Chem. C, 2020, 124(38), 20746–20756, DOI:10.1021/acs.jpcc.0c05562.
- N. R. Chodankar, et al., True Meaning of Pseudocapacitors and Their Performance Metrics: Asymmetric versus Hybrid Supercapacitors, Small, 2020, 16(37), 2002806, DOI:10.1002/smll.202002806.
- A. Jain, et al., Vanadium oxide nanorods as an electrode material for solid state supercapacitor, Sci. Rep., 2022, 12(1), 21024, DOI:10.1038/s41598-022-25707-z.
- S. D. Dhas, et al., Synthesis of NiO nanoparticles for supercapacitor application as an efficient electrode material, Vacuum, 2020, 181, 109646, DOI:10.1016/j.vacuum.2020.109646.
- J. Rajeswari, P. S. Kishore, B. Viswanathan and T. K. Varadarajan, One-dimensional MoO2 nanorods for supercapacitor applications, Electrochem. Commun., 2009, 11(3), 572–575, DOI:10.1016/j.elecom.2008.12.050.
- J. Liu, et al., MnO2-based materials for supercapacitor electrodes: challenges, strategies and prospects, RSC Adv., 2022, 12(55), 35556–35578, 10.1039/D2RA06664E.
- C. Yu, M. Moslehpour, T. K. Tran, L. M. Trung, J. P. Ou and N. H. Tien, Impact of non-renewable energy and natural resources on economic recovery: Empirical evidence from selected developing economies, Resources Policy, 2023, 80, 103221, DOI:10.1016/j.resourpol.2022.103221.
- M. Pathak, S. M. Jeong and C. S. Rout, Spinel NiCo2O4 based hybrid materials for supercapacitors: Recent developments and future perspectives, J. Energy Storage, 2023, 73, 108881, DOI:10.1016/j.est.2023.108881.
- A. Ansari, R. A. Badhe, D. G. Babar and S. S. Garje, One pot solvothermal synthesis of bimetallic copper iron sulfide (CuFeS2) and its use as electrode material in supercapacitor applications, Appl. Surf. Sci. Adv., 2022, 9, 100231, DOI:10.1016/j.apsadv.2022.100231.
- Y. Guo, Y. Chen, X. Hu, Y. Yao and Z. Li, Tween modified CuFe2O4 nanoparticles with enhanced supercapacitor performance, Colloids Surf., A, 2021, 631, 127676, DOI:10.1016/j.colsurfa.2021.127676.
- J. Sun, C. Xu and H. Chen, A review on the synthesis of CuCo2O4-based electrode materials and their applications in supercapacitors, J. Materiomics, 2021, 7(1), 98–126, DOI:10.1016/j.jmat.2020.07.013.
- V. Gajraj and C. R. Mariappan, CuWO4: a promising multifunctional electrode material for energy storage as in redox active solid-state asymmetric supercapacitor and an electrocatalyst for energy conversion in methanol electro-oxidation, J. Electroanal. Chem., 2021, 895, 115504, DOI:10.1016/j.jelechem.2021.115504.
- N. U. H. Liyakath Ali, et al., Thermo-Electric Powered High Energy-Density Hybrid Supercapattery for Driving Overall Water Splitting: A Novel Trifunctional Builder for Self-Powered Hydrogen Production, Small, 2025, 21(25), 2504667, DOI:10.1002/smll.202504667.
- P. R. Bueno, Nanoscale origins of super-capacitance phenomena, J. Power Sources, 2019, 414, 420–434, DOI:10.1016/j.jpowsour.2019.01.010.
- N. U. H. L. Ali, P. Pazhamalai, A. Sathyaseelan, R. Swaminathan and S.-J. Kim, Sodium polyacrylate hydrogel electrolyte: Flexible rechargeable supercapacitor using thermally reduced graphene oxide nanosheets, J. Power Sources, 2025, 625, 235648, DOI:10.1016/j.jpowsour.2024.235648.
- M. S. Chang, T. Kim, J. H. Kang, J. Park and C. R. Park, The effect of surface characteristics of reduced graphene oxide on the performance of a pseudocapacitor, 2D Mater., 2015, 2(1), 014007, DOI:10.1088/2053-1583/2/1/014007.
- S. G. Kandalkar, J. L. Gunjakar and C. D. Lokhande, Preparation of cobalt oxide thin films and its use in supercapacitor application, Appl. Surf. Sci., 2008, 254(17), 5540–5544, DOI:10.1016/j.apsusc.2008.02.163.
- G.-S. Jang, S. Ameen, M. S. Akhtar and H.-S. Shin, Cobalt oxide nanocubes as electrode material for the performance evaluation of electrochemical supercapacitor, Ceram. Int., 2018, 44(1), 588–595, DOI:10.1016/j.ceramint.2017.09.217.
- S. G. Kandalkar, H.-M. Lee, H. Chae and C.-K. Kim, Structural, morphological, and electrical characteristics of the electrodeposited cobalt oxide electrode for supercapacitor applications, Mater. Res. Bull., 2011, 46(1), 48–51, DOI:10.1016/j.materresbull.2010.09.041.
- F. Ali, et al., Capacitive properties of novel Sb-doped Co3O4 electrode material synthesized by hydrothermal method, Ceram. Int., 2021, 47(22), 32210–32217, DOI:10.1016/j.ceramint.2021.08.114.
- N. Pradeep, V. Chaitra, V. Uma and A. N. Grace, Antimony oxide nanobelts: synthesis by chemical vapour deposition and its characterisation, Int. J. Nanotechnol., 2017, 14(9–11), 752–761, DOI:10.1504/IJNT.2017.086761.
- A. Guillén-Bonilla, et al., Dynamic Response of CoSb2O6 Trirutile-Type Oxides in a CO2 Atmosphere at Low-Temperatures, Sensors, 2014, 14(9), 15802–15814, DOI:10.3390/s140915802.
- X. Zhou, A. Wang, X. Zheng, D. Sun and Y. Yang, Uniformly anchoring Sb2O5 nanoparticles on graphene sheets via Co2+-induced deposition for enhanced lithium/sodium-ion storage, Inorg. Chem. Front., 2024, 11(14), 4167–4178, 10.1039/D4QI00827H.
- U. Ray, D. Das, A. Mitra, K. Das, S. B. Majumder and S. Das, Cobalt Antimony Oxide-Carbon Black Composite as Negative Electrode for Alkali-Ion Batteries: Synthesis and Electrochemical Characterization, Social Science Research Network, Rochester, NY, 2023, p. 4608624 DOI:10.2139/ssrn.4608624.
- H. Guillén-Bonilla, et al., Sensitivity of Mesoporous CoSb2O6 Nanoparticles to Gaseous CO and C3H8 at Low Temperatures, J. Nanomater., 2015, e308465, DOI:10.1155/2015/308465.
- J. Sun, B. Luo and H. Li, A Review on the Conventional Capacitors, Supercapacitors, and Emerging Hybrid Ion Capacitors: Past, Present, and Future, Adv. Energy Sustainability Res., 2022, 3(6), 2100191, DOI:10.1002/aesr.202100191.
- O. Ibukun and H. Jeong, Effects of Aqueous Electrolytes in Supercapacitors, New Phys.: Sae Mulli, 2019, 69, 154–158, DOI:10.3938/NPSM.69.154.
- A. Guillén-Bonilla, et al., Synthesis and characterization of cobalt antimonate nanostructures and their study as potential CO and CO2 sensor at low temperatures, J. Mater. Sci.: Mater. Electron., 2018, 29(18), 15632–15642, DOI:10.1007/s10854-018-9157-2.
- K. M. Anilkumar, M. Manoj, B. Jinsiha, V. S. Pradeep and S. Jayalekshmi, Mn3O4/reduced graphene oxide nanocomposite electrodes with tailored morphology for high power supercapacitor applications, Electrochim. Acta, 2017, 236, 424–433, DOI:10.1016/j.electacta.2017.03.167.
- M. A. Garakani, et al., Scalable spray-coated graphene-based electrodes for high-power electrochemical double-layer capacitors operating over a wide range of temperature, Energy Storage Mater., 2021, 34, 1–11, DOI:10.1016/j.ensm.2020.08.036.
- C. Lämmel, M. Schneider, M. Weiser and A. Michaelis, Investigations of electrochemical double layer capacitor (EDLC) materials – a comparison of test methods, Materialwiss. Werkstofftech., 2013, 44(7), 641–649, DOI:10.1002/mawe.201300122.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50(24), 17953–17979, DOI:10.1103/PhysRevB.50.17953.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59(3), 1758–1775, DOI:10.1103/PhysRevB.59.1758.
- Z. Wu and R. E. Cohen, More accurate generalized gradient approximation for solids, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73(23), 235116, DOI:10.1103/PhysRevB.73.235116.
- S. Grimme, Semiempirical GGA-type density functional constructed with a long-range dispersion correction, J. Comput. Chem., 2006, 27(15), 1787–1799, DOI:10.1002/jcc.20495.
- M. J. Kruszewski, K. Cymerman, E. Choińska, D. Moszczyńska and Ł. Ciupiński, A comparative study of oxidation behavior of Co4Sb12 and Co4Sb10.8Se0.6Te0.6 skutterudite thermoelectric materials fabricated via fast SHS-PPS route, J. Eur. Ceram. Soc., 2024, 44(6), 3760–3766, DOI:10.1016/j.jeurceramsoc.2024.01.044.
- H. Jensen, et al., Determination of size distributions in nanosized powders by TEM, XRD, and SAXS, J. Exp. Nanosci., 2006, 1(3), 355–375, DOI:10.1080/17458080600752482.
- S. Kumuda, U. Gandhi, U. Mangalanathan and K. Rajanna, Synthesis and characterization of graphene oxide and reduced graphene oxide chemically reduced at different time duration, J. Mater. Sci.: Mater. Electron., 2024, 35(9), 637, DOI:10.1007/s10854-024-12393-y.
- L. G. Cançado, et al., General equation for the determination of the crystallite size La of nanographite by Raman spectroscopy, Appl. Phys. Lett., 2006, 88(16), 163106, DOI:10.1063/1.2196057.
- T. J. Chuang, C. R. Brundle and D. W. Rice, Interpretation of the X-ray photoemission spectra of cobalt oxides and cobalt oxide surfaces, Surf. Sci., 1976, 59(2), 413–429, DOI:10.1016/0039-6028(76)90026-1.
- T. A. Evans and K.-S. Choi, Electrochemical Synthesis and Investigation of Stoichiometric, Phase-Pure CoSb2O6 and MnSb2O6 Electrodes for the Oxygen Evolution Reaction in Acidic Media, ACS Appl. Energy Mater., 2020, 3(6), 5563–5571, DOI:10.1021/acsaem.0c00526.
- Z. Li, X. Song, S. Cui, Y. Jiao and C. Zhou, Fabrication of macroporous reduced graphene oxide composite aerogels reinforced with chitosan for high bilirubin adsorption, RSC Adv., 2018, 8(15), 8338–8348, 10.1039/C8RA00358K.
- Z. Chen, et al., Nanoarchitectured Co3O4/reduced graphene oxide as anode material for lithium-ion batteries with enhanced cycling stability, Ionics, 2019, 25(12), 5779–5786, DOI:10.1007/s11581-019-03134-x.
- P. Lin, et al., The Nickel Oxide/CNT Composites with High Capacitance for Supercapacitor, J. Electrochem. Soc., 2010, 157(7), A818, DOI:10.1149/1.3425624.
- Parul, S. Sahoo, S. Ratha, G. Sanyal, B. Chakraborty and S. K. Nayak, Experimental and theoretical investigation on the charge storage performance of NiSb2O6 and its reduced graphene oxide composite – a comparative analysis, Phys. Chem. Chem. Phys., 2024, 26(48), 29962–29975, 10.1039/D4CP03249G.
- G. He, et al., Hydrothermal preparation of Co3O4@graphene nanocomposite for supercapacitor with enhanced capacitive performance, Mater. Lett., 2012, 82, 61–63, DOI:10.1016/j.matlet.2012.05.048.
- J. Jayachandiran, et al., Synthesis and Electrochemical Studies of rGO/ZnO Nanocomposite for Supercapacitor Application, J. Inorg. Organomet. Polym., 2018, 28(5), 2046–2055, DOI:10.1007/s10904-018-0873-0.
- M. Chandel, D. Moitra, P. Makkar, H. Sinha, H. S. Hora and N. N. Ghosh, Synthesis of multifunctional CuFe2O4-reduced graphene oxide nanocomposite: an efficient magnetically separable catalyst as well as high performance supercapacitor and first-principles calculations of its electronic structures, RSC Adv., 2018, 8(49), 27725–27739, 10.1039/C8RA05393F.
- D. Govindarajan, V. Uma Shankar and R. Gopalakrishnan, Supercapacitor behavior and characterization of RGO anchored V2O5 nanorods, J. Mater. Sci.: Mater. Electron., 2019, 30(17), 16142–16155, DOI:10.1007/s10854-019-01984-9.
- N. A. Devi, et al., Investigation of chemical bonding and supercapacitivity properties of Fe3O4-rGO nanocomposites for supercapacitor applications, Diamond Relat. Mater., 2020, 104, 107756, DOI:10.1016/j.diamond.2020.107756.
- I. Kotutha, T. Duangchuen, E. Swatsitang, W. Meewasana, J. Khajonrit and S. Maensiri, Electrochemical properties of rGO/CoFe2O4 nanocomposites for energy storage application, Ionics, 2019, 25(11), 5401–5409, DOI:10.1007/s11581-019-03114-1.
- S. Ghosh, P. Samanta, P. Samanta, N. C. Murmu and T. Kuila, Investigation of Electrochemical Charge Storage Efficiency of NiCo2Se4/RGO Composites Derived at Varied Duration and Its Asymmetric Supercapacitor Device, Energy Fuels, 2020, 34(10), 13056–13066, DOI:10.1021/acs.energyfuels.0c02152.
- B. Rani and N. K. Sahu, Electrochemical properties of CoFe2O4 nanoparticles and its rGO composite for supercapacitor, Diamond Relat. Mater., 2020, 108, 107978, DOI:10.1016/j.diamond.2020.107978.
- C. Xiang, M. Li, M. Zhi, A. Manivannan and N. Wu, A reduced graphene oxide/Co3O4 composite for supercapacitor electrode, J. Power Sources, 2013, 226, 65–70, DOI:10.1016/j.jpowsour.2012.10.064.
- A. C. Lazanas and M. I. Prodromidis, Electrochemical Impedance Spectroscopy–A Tutorial, ACS Meas. Sci. Au, 2023, 3(3), 162–193, DOI:10.1021/acsmeasuresciau.2c00070.
- S. A. El-Khodary, et al., Sonochemical assisted fabrication of 3D hierarchical porous carbon for high-performance symmetric supercapacitor, Ultrason. Sonochem., 2019, 58, 104617, DOI:10.1016/j.ultsonch.2019.104617.
- Z.-H. Huang, T.-Y. Liu, Y. Song, Y. Li and X.-X. Liu, Balancing the electrical double layer capacitance and pseudocapacitance of hetero-atom doped carbon, Nanoscale, 2017, 9(35), 13119–13127, 10.1039/C7NR04234E.
- J. Liu, et al., Advanced Energy Storage Devices: Basic Principles, Analytical Methods, and Rational Materials Design, Adv. Sci., 2018, 5(1), 1700322, DOI:10.1002/advs.201700322.
- Y. Lan, et al., Phosphorization Boosts the Capacitance of Mixed Metal Nanosheet Array for High Performance Supercapacitor Electrode, Nanoscale, 2018, 10(25), 11775–11781, 10.1039/C8NR01229F.
- Y. Wang, et al., Intercalating Ultrathin MoO3 Nanobelts into MXene Film with Ultrahigh Volumetric Capacitance and Excellent Deformation for High-Energy-Density Devices, Nano-Micro Lett., 2020, 12(1), 115, DOI:10.1007/s40820-020-00450-0.
- S. Dutt, et al., Flexible and Highly Stable Textile-Based Symmetric Supercapacitor Comprising Binder-Free MnO2/rGO-CF Nanocomposite Electrodes, J. Electron. Mater., 2023, 52(11), 7447–7458, DOI:10.1007/s11664-023-10683-5.
- M. Kour, et al., Highly stable electrodeposited 2D Mn0.2Co0.8 LDH nanoplatelets based symmetric supercapacitor electrode, Mater. Sci. Eng., B, 2023, 293, 116488, DOI:10.1016/j.mseb.2023.116488.
- I. Rabani, et al., 1D-CoSe 2 nanoarray: a designed structure for efficient hydrogen evolution and symmetric supercapacitor characteristics, Dalton Trans., 2020, 49(40), 14191–14200, 10.1039/D0DT02548H.
- U. M. Patil, et al., Enhanced Symmetric Supercapacitive Performance of Co(OH)2 Nanorods Decorated Conducting Porous Graphene Foam Electrodes, Electrochim. Acta, 2014, 129, 334–342, DOI:10.1016/j.electacta.2014.02.063.
- M. Murugan, et al., Development of Mesoporous Carbon Composites with Waste Plastics Derived Graphene and MnO2 for Supercapacitor Applications, J. Electrochem. Soc., 2023, 170(4), 040518, DOI:10.1149/1945-7111/acbf7b.
- V. Sahu, S. Goel, R. Kishore Sharma and G. Singh, Zinc oxide nanoring embedded lacey graphene nanoribbons in symmetric/asymmetric electrochemical capacitive energy storage, Nanoscale, 2015, 7(48), 20642–20651, 10.1039/C5NR06083D.
- N. Kumar, T. A. Wani, P. K. Pathak, A. Bera and R. R. Salunkhe, Multifunctional nanoarchitectured porous carbon for solar steam generation and supercapacitor applications, Sustainable Energy Fuels, 2022, 6(7), 1762–1769, 10.1039/D2SE00092J.
- A. Guillén-Bonilla, et al., Dynamic Response of CoSb2O6 Trirutile-Type Oxides in a CO2 Atmosphere at Low-Temperatures, Sensors, 2014, 14(9), 15802–15814, DOI:10.3390/s140915802.
- E. Sanville, S. D. Kenny, R. Smith and G. Henkelman, Improved grid-based algorithm for Bader charge allocation, J. Comput. Chem., 2007, 28(5), 899–908, DOI:10.1002/jcc.20575.
- S. Abbas, et al., Fe doped NiCo2O4@rGO nanosheets network boost electrochemical kinetics for high energy density and stable asymmetric coin cell supercapacitor, J. Energy Storage, 2024, 102, 114181, DOI:10.1016/j.est.2024.114181.
- M. Pathak, J. R. Jose, B. Chakraborty and C. S. Rout, High performance supercapacitor electrodes based on spinel NiCo2O4@MWCNT composite with insights from density functional theory simulations, J. Chem. Phys., 2020, 152(6), 064706, DOI:10.1063/1.5138727.
- G. M. Yang, H. Z. Zhang, X. F. Fan and W. T. Zheng, Density Functional Theory Calculations for the Quantum Capacitance Performance of Graphene-Based Electrode Material, J. Phys. Chem. C, 2015, 119(12), 6464–6470, DOI:10.1021/jp512176r.
- C. Zhan, J. Neal, J. Wu and D. Jiang, Quantum Effects on the Capacitance of Graphene-Based Electrodes, J. Phys. Chem. C, 2015, 119(39), 22297–22303, DOI:10.1021/acs.jpcc.5b05930.
- C. Zhan, J. Neal, J. Wu and D. Jiang, Quantum Effects on the Capacitance of Graphene-Based Electrodes, J. Phys. Chem. C, 2015, 119(39), 22297–22303, DOI:10.1021/acs.jpcc.5b05930.
| This journal is © The Royal Society of Chemistry 2026 |