Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Copolymers with tailored microstructure as versatile platforms for ZIF-8 polyMOF growth
M. Celeste
Legarto
ab,
Agustín
Iborra
*a,
Juan J.
Romero†
a,
Juan M.
Padró
a,
Cristian
Villa Pérez
a,
Matías
Rafti
 b,
Isabel N.
Vega
a and
Juan M.
Giussi
a
b,
Isabel N.
Vega
a and
Juan M.
Giussi
a
aYPF Tecnología (Y-TEC), Berisso, Buenos Aires, Argentina. E-mail: agustin.iborra@ypftecnologia.com
bInstituto de Investigaciones Fisicoquímicas Teóricas y Aplicadas (INIFTA) – Departamento de Química – Facultad de Ciencias Exactas – Universidad Nacional de La Plata – CONICET – (1900), La Plata, Argentina
First published on 11th May 2026
Abstract
In this work, we report copolymers based on zinc dimethacrylate (ZnDMA) and poly(ethylene glycol)methyl ether methacrylate (PEGMA) as versatile platforms for the in situ growth of ZIF-8 polyMOFs. ZnDMA is a suitable monomer which can potentially optimize polyMOF growth, enabling improved control over the nucleation process and both final structure and morphology. The copolymers were synthesized both as bulk materials from precursor solutions and via surface-initiated Atom Transfer Radical Polymerization (ATRP). The composition and microstructure were investigated using the Fineman-Ross method, complemented by characterization through X-ray photoelectron spectroscopy (XPS), proton nuclear magnetic resonance (1H NMR), thermogravimetric analysis (TGA), aqueous size exclusion chromatography (SEC) and scanning electron microscopy (SEM). We found that variations in structural stability, composition and microstructure of the resulting poly(ZnDMA-co-PEGMA)@ZIF-8 materials can be traced back to conditions affecting polyMOF growth, mainly regarding efficient nucleation and controlled crystalline growth. Overall, these results demonstrate that the hereby proposed synthetic approach to polyMOFs can be employed for the rational design of functional nanostructured porous materials.
Introduction
The class of porous materials known as Metal Organic Frameworks (MOFs) has attracted a great deal of attention over the past three decades due to their exceptional physicochemical properties and structural tunability. Among their most remarkable features are high specific surface areas (reaching up to 7000 m2 g−1 under appropriate activation conditions),1–3 uniform and well-defined pore sizes stemming from their crystalline architectures,4–7 and the presence of coordinatively unsaturated metal sites that enable selective adsorption and heterogeneous catalysis.8 These unique attributes have stimulated an exponential growth in MOF-related research, leading to a vast number of reports addressing their potential in energy storage and conversion,9,10 selective adsorption,11,12 carbon capture,13 sensing/biosensing,14 water harvesting15,16 and remediation applications,17 among many other cutting-edge applications. One effective strategy for overcoming challenges that have long hindered large-scale synthesis of MOFs for industrial applications (limited processability and reduced chemical robustness under common real-life operating conditions) has been their integration into polymeric scaffolds. Although the concept of combining MOFs with polymers was proposed more than two decades ago,18 the formal introduction of the term “polyMOFs” and the recognition of these composite materials as a distinct class can be traced back to Cohen et al.19 and, more broadly, to the seminal contribution of Matzger et al.20 Since then, a wide range of synthetic strategies have been developed, yielding polyMOFs with diverse porous architectures, increased processability, and enhanced functionalities.21–31A widely adopted strategy for the creation of polyMOFs is the partial or complete substitution of the organic linker in a given MOF structure with a polymer bearing equivalent coordination moieties in appropriate orientations, e.g., replacing benzene dicarboxylate (bdc) in Zr-UiO-MOFs or Zn-IRMOFs with polyethers incorporating bdc units at predefined spacings.19,22,30,31 The trade-off for the reduced thermal stability and increased synthetic complexity is the emergence of remarkable features in polyMOFs, such as designed hierarchical porosity and stimuli-responsiveness.32 Furthermore, polyMOFs offer a unique pathway for the fabrication of free-standing structures, which are highly desirable in membrane technologies, including crosslinked polymeric brushes obtained via surface-initiated radical polymerization (SI-RAFT) on surface-functionalized nano- or microscale MOF units. This concept has been successfully applied for Zn-imidazolate ZIF-8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33 and Zr-based UiO-66-NH2 PolyMOFs.19,22
33 and Zr-based UiO-66-NH2 PolyMOFs.19,22
On a related note, polymeric brushes exposing suitable functional moieties can be used as three-dimensional primers for MOF thin-film growth on targeted substrates.34 Moreover, when monomers structurally analogous to the MOF organic linker are incorporated into the polymeric brush, the accessible porosity of the resulting polyMOF film can be precisely tuned by controlling the brush surface density, as demonstrated for ZIF-8 MOFs and imidazolate-functionalized surface-anchored polymeric brushes.35 The above mentioned ZIF-8 MOF (Zn(mim)2, mim2− = 2-methylimidazolate)36 is one of the most widely studied and belongs to the zeolitic imidazolate framework (ZIF) subclass, featuring a zeolite-like crystalline structure with SOD topology, highly ordered hydrophobic micropores (6 Å and 11.4 Å pore window and diameter respectively), and relatively high BET surface areas (up to 1600 m2 g−1). Owing to the versatility and high reproducibility of synthesis protocols,37 the use of ZIF-8 for multiple applications has been explored, including gas storage,38,39 separations,40,41 heterogeneous catalysis, or chemical sensing.42,43
Ultimately, the rational design of these PolyMOF with hierarchical nanoarchitectures is driven by their potential for diverse applications. In particular, their tunable nanoscale porosity and robust polymer–MOF interfaces make them promising candidates for carbon capture, utilization, and storage (CCUS),44,45 as well as for the selective separation of low molecular weight alkane/alkene/alkyne.46 Furthermore, by optimizing the synthesis under conditions aligned with green chemistry principles, this platform offers a sustainable pathway toward the development of scalable sorbents for environmental remediation.47
Inspired by the ideas discussed above, we evaluated the feasibility of employing tailor-made polymeric architectures based on the combination of zinc dimethacrylate (ZnDMA) and poly(ethylene glycol)methyl ether methacrylate (PEGMA) as platforms for the controlled growth of ZIF-8 polyMOFs. Our main hypothesis is that ad hoc poly(ZnDMA-co-PEGMA) copolymers, featuring homogeneously distributed Zn2+ coordination centers, can act as effective nucleation points which can guide polyMOF formation. The selection of ZnDMA represents a rational and strategic approach to circumvent common limitations in polyMOF synthesis, as it offers advantages over other metallic ion precursor sources. Namely, ZnDMA-based copolymers enable uniform and high-density metal ion loading along the polymeric backbone. In addition, the dimethacrylate structure provides a rigid and sterically constrained coordination environment, preventing premature Zn2+ leaching into the solution before 2-methylimidazole (HmIm) ligand present can trigger in situ nucleation. Finally, the intrinsic ionic nature of ZnDMA modulates the local dielectric environment, potentially lowering the activation energy required for ZIF-8 nucleation compared to other non-ionic zinc-sources.
As ZnDMA homopolymers exhibited poor water solubility due to their high degree of crosslinking, PEGMA was selected as a comonomer owing to its greater water solubility. The incorporation of PEGMA aimed to increase hydrophilicity, reduce the degree of crosslinking, and enable homogeneous polymer growth both in solutions and on surfaces. To this end, we systematically evaluated the influence of polymer architecture and composition on polyMOF formation by employing ZnDMA-co-PEGMA copolymers synthesized under both bulk and surface-anchored conditions. The resulting materials are comparatively analyzed in terms of their structural and morphological features, providing insight into the role of metal-bearing polymer design in directing ZIF-8 polyMOF growth (Scheme 1).
Experimental section
Materials
(3-Aminopropyl)triethoxysilane (APTES, >99%), α-bromoisobutyryl bromide (BIBB, 98%), triethylamine (TEA, >99%), zinc methacrylate (ZnDMA, >99.9%), poly(ethylene glycol)methyl ether methacrylate average Mn 500 g mol−1 (PEGMA, >99%), N,N,N′,N″,N″-pentamethyldiethylenetriamine (PMDTA, >99%), copper(I) chloride (CuCl, >99%), 2,2′-bipyridyl (Bpy, >99%), ethyl 2-bromoisobutyrate (EBIB), zinc nitrate hexahydrate (Zn(NO3)2·6H2O, 98%) and 2-methylimidazole (HmIm, 99%), were purchased from Sigma-Aldrich, St Louis USA, and used without further purification. Ammonia (30% aqueous solution) and sodium chloride (NaCl) was obtained from Anedra (Bs. As., Argentina). Solvents dichloromethane (DCM) and methanol (MeOH) grade RPE, also from Anedra, were distilled under Na and kept with 4 A molecular sieves until used. The dialysis tubing cellulose membrane with a typical molecular weight cut-off of 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 was provided by Aldrich. Cover glasses of 18 mm diameter were purchased from VWR international. Prior to use, coverslips were cleaned using piranha solution (H2SO4
000 was provided by Aldrich. Cover glasses of 18 mm diameter were purchased from VWR international. Prior to use, coverslips were cleaned using piranha solution (H2SO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O2 in a 1
H2O2 in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio) and thoroughly rinsed with ethanol. Bovine serum albumin (BSA monomer), 2 mg mL−1 in a 0.9% NaCl solution containing sodic azide, Mw = 66.4 kDa; Rh = 3.5 nm acquire from Thermo Scientific® (Waltham, Massachusetts, USA). Ultra-pure water was produced using a Milli-Q® purification apparatus (Millipore, Molsheim, France).
4 ratio) and thoroughly rinsed with ethanol. Bovine serum albumin (BSA monomer), 2 mg mL−1 in a 0.9% NaCl solution containing sodic azide, Mw = 66.4 kDa; Rh = 3.5 nm acquire from Thermo Scientific® (Waltham, Massachusetts, USA). Ultra-pure water was produced using a Milli-Q® purification apparatus (Millipore, Molsheim, France).
Methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PMDETA catalyst system (1
PMDETA catalyst system (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, molar ratio) was employed at a 2.5-fold molar excess relative to EBIB (7.5-fold molar ratio to monomers). In each experiment, ZnDMA and/or PEGMA were dissolved in methanol to obtain a total monomer concentration of 10 wt% and transferred into a Schlenk tube. After catalyst addition, the mixtures were purged with nitrogen at room temperature for 60 min. The solutions were then heated to 45 °C under stirring, and EBIB was added to initiate polymerization. Reactions proceeded for 24 h. Purification of the crude homo- and copolymers was performed in two steps. First, the reaction mixtures were precipitated into hexane, and the resulting waxy solids were dried. These solids were then redissolved in water and subjected to three dialysis cycles (12 h each) against fresh distilled water using a membrane with a molecular weight cutoff of 1500 g mol−1 and subsequently freeze-dried. Finally, the purified polymers were lyophilized and stored until further use. The absence of olefinic proton signals in the 1H NMR spectra confirmed complete monomer conversion and the high purity of the final products. The (ZnDMA
1, molar ratio) was employed at a 2.5-fold molar excess relative to EBIB (7.5-fold molar ratio to monomers). In each experiment, ZnDMA and/or PEGMA were dissolved in methanol to obtain a total monomer concentration of 10 wt% and transferred into a Schlenk tube. After catalyst addition, the mixtures were purged with nitrogen at room temperature for 60 min. The solutions were then heated to 45 °C under stirring, and EBIB was added to initiate polymerization. Reactions proceeded for 24 h. Purification of the crude homo- and copolymers was performed in two steps. First, the reaction mixtures were precipitated into hexane, and the resulting waxy solids were dried. These solids were then redissolved in water and subjected to three dialysis cycles (12 h each) against fresh distilled water using a membrane with a molecular weight cutoff of 1500 g mol−1 and subsequently freeze-dried. Finally, the purified polymers were lyophilized and stored until further use. The absence of olefinic proton signals in the 1H NMR spectra confirmed complete monomer conversion and the high purity of the final products. The (ZnDMA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PEGMA) molar feed ratios were varied as follows: (0.1
PEGMA) molar feed ratios were varied as follows: (0.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), (0.5
1), (0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), (1
1), (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and (1
1) and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5), corresponding to ZnDMA feed compositions (fZnDMA) of 0.1, 0.3, 0.5 and 0.7.
0.5), corresponding to ZnDMA feed compositions (fZnDMA) of 0.1, 0.3, 0.5 and 0.7.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PMDETA (1
PMDETA (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as the catalyst (7.5-fold molar ratio to monomers). The polymerization was conducted at 45 °C for 24 h under a nitrogen atmosphere. The reaction was terminated by exposure to air. The polymer-coated glass slides were then rinsed with MeOH and stored under vacuum until further use. As in bulk synthesis, the fraction of ZnDMA (fZnDMA) was varied as follows: 0.1, 0.3, 0.5 and 0.7.
1) as the catalyst (7.5-fold molar ratio to monomers). The polymerization was conducted at 45 °C for 24 h under a nitrogen atmosphere. The reaction was terminated by exposure to air. The polymer-coated glass slides were then rinsed with MeOH and stored under vacuum until further use. As in bulk synthesis, the fraction of ZnDMA (fZnDMA) was varied as follows: 0.1, 0.3, 0.5 and 0.7.
Results and discussion
Polymer synthesis and characterization
In a previous work, we employed 1-vinylimidazole as a ligand-bearing monomer in polymeric brushes to promote ZIF-8 surface growth as a strategy to obtain ZIF-8 MOF films with tunable porosity.35 In our synthetic approach, zinc dimethacrylate (ZnDMA) was employed as a metal-bearing monomer to promote ZIF-8 growth within the polymer scaffold, both from surface-anchored and bulk (colloidal dispersions) copolymers. Having in mind results obtained for polymerizations using different monomer compositions, we focused on determining the influence of both bulk and surface grafted poly(ZnDMAx-co-PEGMAy) scaffolds on polyMOF growth. In contrast, we employ ZnDMA which features Zn2+ as pre-defined nucleation sites along the polymer backbone, yielding faster growth kinetics, as no preliminary coordination step is required. This differs from ligand-bearing systems (e.g., poly-vinyl-imidazole or PVI), where nucleation depends on the diffusion and coordination of Zn2+ ions present. Moreover, the direct involvement of polymer-bound carboxylates in metal coordination supports enhanced polymer–MOF integration, in contrast to ligand-bearing systems that typically act as scaffolds and favor surface growth.The atom transfer radical copolymerization of zinc dimethacrylate (ZnDMA) with poly(ethylene glycol)methyl ether methacrylate (PEGMA, average Mn = 500 g mol−1) yielded a series of novel copolymers whose structures, 1H NMR spectra, and resonance signal assignments are presented in Fig. 1A. Four copolymers were synthesized with ZnDMA feed compositions (fZnDMA) of 0.1, 0.3, 0.5, and 0.7. To determine the compositions of the bulk copolymers, 1H NMR signals corresponding to the polymer backbone of both monomers and to the PEGMA side chains were analyzed, since ZnDMA lacks side-chain groups capable of producing detectable 1H NMR signals. The resonance at δ = 1.20–1.95 ppm (signal a) corresponds to the –CH2– backbone protons of both monomers, while the signal at δ = 0.50–1.15 ppm (b) is attributed to the –CH3 backbone protons. The peak at δ = 4.08 ppm (c) corresponds to the PEGMA –OCH2– group bonded to the carboxylate moiety, δ = 3.65 ppm (d) is assigned to the PEGMA –CH2O– units, δ = 3.55 ppm (e) to the terminal –OCH2– group, and δ = 3.38 ppm (f) to the terminal –OCH3 group of PEGMA. The copolymer compositions (FPEGMA) were estimated using from the integral ratio between the PEGMA terminal –OCH3 signal (δ = 3.4 ppm – corresponding to signal If) and the –CH2– backbone signal (δ = 1.95 ppm – corresponding to signal Ib). The composition values were obtained according to eqn (1):
 | (1) |
For each copolymer synthesis, Table 1 shows FZnDMA, polymer conversion, the mass-average molecular weight (Mw), and the dispersity (Đ = Mw/Mn).
| f ZnDMA | Conversion (%) | F ZnDMA | M w (g mol−1) | Đ |
|---|---|---|---|---|
| 0.1 | 9.0 | 0.15 | 1.40 × 104 | 1.16 |
| 0.3 | 9.5 | 0.51 | 1.09 × 104 | 1.53 |
| 0.5 | 18.5 | 0.75 | 1.45 × 104 | 1.65 |
| 0.7 | 20.5 | 0.87 | 9.94 × 103 | 1.14 |
It is observed that, for the same polymerization times, increasing the proportion of ZnDMA results in higher polymerization conversion, suggesting higher reactivity of ZnDMA. The Mw values remain around 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 in all cases, with lower dispersity values at the compositional extremes (fZnDMA 0.1 and 0.7), close to 1.1, supporting the hypothesis of a controlled polymerization. Dispersity values closer to 1.5 for fZnDMA 0.3 and 0.5 may indicate termination mechanisms more likely associated with disproportionation or chain transfer, in agreement with theoretical predictions.50 The thermogravimetric curves presented in Fig. 1B reveal significant differences in the initial degradation temperatures (IDT) of polyZnDMA and polyPEGMA, which are 410 °C and 260 °C, respectively. The polyZnDMA profile exhibits continuous weight loss from 100 °C up to the IDT, suggesting the presence of coordinated water molecules associated with Zn2+ ions, as reported by other authors.51 As observed, the polyPEGMA homopolymer shows no residual weight at 500 °C, whereas polyZnDMA shows a residual weight of approximately 34%. This remaining weight is consistent with the formation of ZnO, as previously suggested by McNeill et al. for polyZnDMA degradation.52 Furthermore, the residual weight increases as fZnDMA increases, in line with the increasing Zn2+ content in the copolymers. The TGA profiles of copolymers show two distinct thermal events, suggesting a block-like or sequential monomer microstructure along the copolymers chain.
000 g mol−1 in all cases, with lower dispersity values at the compositional extremes (fZnDMA 0.1 and 0.7), close to 1.1, supporting the hypothesis of a controlled polymerization. Dispersity values closer to 1.5 for fZnDMA 0.3 and 0.5 may indicate termination mechanisms more likely associated with disproportionation or chain transfer, in agreement with theoretical predictions.50 The thermogravimetric curves presented in Fig. 1B reveal significant differences in the initial degradation temperatures (IDT) of polyZnDMA and polyPEGMA, which are 410 °C and 260 °C, respectively. The polyZnDMA profile exhibits continuous weight loss from 100 °C up to the IDT, suggesting the presence of coordinated water molecules associated with Zn2+ ions, as reported by other authors.51 As observed, the polyPEGMA homopolymer shows no residual weight at 500 °C, whereas polyZnDMA shows a residual weight of approximately 34%. This remaining weight is consistent with the formation of ZnO, as previously suggested by McNeill et al. for polyZnDMA degradation.52 Furthermore, the residual weight increases as fZnDMA increases, in line with the increasing Zn2+ content in the copolymers. The TGA profiles of copolymers show two distinct thermal events, suggesting a block-like or sequential monomer microstructure along the copolymers chain.
DSC curves of poly(ZnDMAx-co-PEGMAy) copolymers and the polyPEGMA homopolymer exhibit complex thermal events. As previously reported, the glass transition temperature (Tg) of the polyPEGMA homopolymer is around −60 °C, and its melting temperature (Tm) is approximately −5 °C. In contrast, the polyZnDMA homopolymer does not show any detectable thermal transitions, likely due to its high degree of crosslinking.51–53 As shown in Fig. 1C, the Tg values of the copolymers with fZnDMA = 0.1 and 0.3 remain close to −60 °C. For fZnDMA = 0.5, the Tg appears to shift slightly towards lower temperatures, while for fZnDMA = 0.7, no clear thermal transition is observed. The melting peak corresponding to PEGMA is visible for fZnDMA values of 0.1, 0.3 and 0.5. However, the high ZnDMA content in copolymer with fZnDMA = 0.7 likely suppresses the melting peak. In agreement with the TGA profile, this thermal behavior suggests a block or sequential monomer microstructure along the copolymer chains.
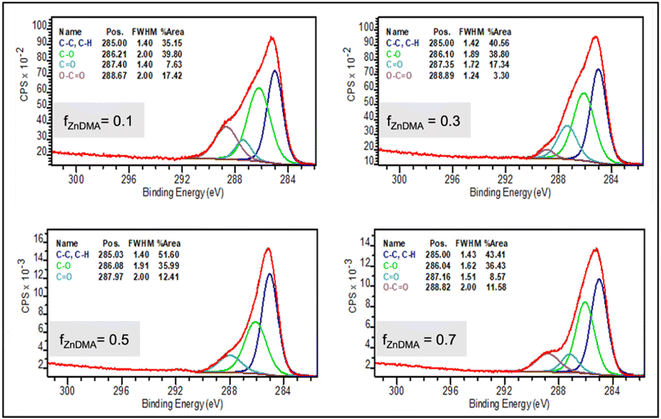
In comparison with bulk copolymerization, surface-initiated atom transfer radical copolymerizations (SI-ATRP) of ZnDMA with PEGMA 500 produce new brush copolymers, XPS spectra are shown in Fig. 2. Compositional analysis from the XPS data was performed by considering the C 1s and Zn 2p peak areas from the XPS survey spectra (at −284.5 and 1022.0 eV binding energies respectively). These values were corrected for adventitious carbon54 and normalized using the corresponding atomic sensitivity factors.55 The corrected data were then used to quantify the Zn and C contents in the sample, considering that for ZnDMA the Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C relation is 1
C relation is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. Also, from C 1s signal deconvolution, the relative contribution C species did not show significative changes, suggesting that the main contribution of this signal is related to copolymer backbone.
4. Also, from C 1s signal deconvolution, the relative contribution C species did not show significative changes, suggesting that the main contribution of this signal is related to copolymer backbone.
The FZnDMA values obtained under surface-initiated polymerization conditions were significantly different from those observed under bulk conditions, with values of 0.17, 0.34, 0.36, and 0.37 for fZnDMA = 0.1; 0.3; 0.5 and 0.7, respectively. As quantitative C determination is always challenging, XPS obtained results were compared with those derived from Raman spectroscopy, showing good agreement (as detailed in the SI).
Depth profiling studies were carried out to evaluate the homogeneity of the polymer layer and any possible lamellar segregation (see Fig. 3). A depth profiling experiment was performed for a selected poly(ZnDMAx-co-PEGMAy) surface brush corresponding to fZnDMA = 0.5 using Ar+ ions as reactive sputtering source, and quantifying C and Si amounts form C 1s and Si 2p peaks. Considering the sharp variation in C intensity for copolymer relative to Si (glass substrate), the thickness of the polymer brush can be estimated to be below 5 nm. Furthermore, the Zn composition remained constant along the polymer brush, indicating a homogeneous distribution with no evidence of lamellar segregation.
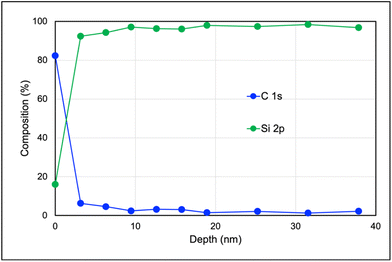 | ||
| Fig. 3 Depth profile result for poly(ZnDMAx-co-PEGMAy) surface bushes corresponding to fZnDMA = 0.5. | ||
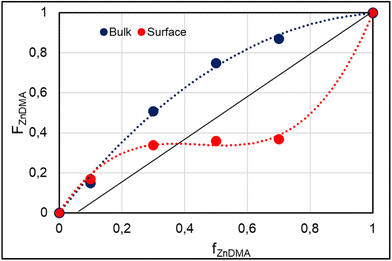
The comonomer–copolymer composition curves (see Fig. 4) reveal distinct trends for bulk and surface polymerizations. Notable differences are observed in the copolymerization behavior of this monomer pair when comparing bulk and surface-initiated copolymerization. In bulk polymerization, there is a clear trend toward higher FZnDMA values for each corresponding fZnDMA value. In surface-initiated copolymerization, however, for low fZnDMA values, the FZnDMA values are higher than fZnDMA (0.17 > 0.1 and 0.34 > 0.3) whereas at higher fZnDMA values, the FZnDMA values become lower than fZnDMA (0.36 < 0.5 and 0.37 < 0.7).
The determination of monomer reactivity ratios provides insight into the copolymerization behavior of monomer pairs. The application of the Fineman-Ross method under the evaluated conditions allowed us to obtain the following reactivity ratios: (a) bulk condition: r1(ZnDMA) = 3.26 and r2(PEGMA) = 0.60; (b) surface condition: r1(ZnDMA) = 0.13 and r2(PEGMA) = 0.43. The obtained values, with r1 > r2 for the bulk condition, suggest a higher reactivity of the ZnDMA monomer and the preferential reaction of the poly(ZnDMA) radical toward the ZnDMA monomer. Furthermore, the product r1 × r2 = 1.956 is consistent with an unbalanced copolymerization behavior, with a higher incorporation of ZnDMA and the possible formation of ZnDMA-enriched sequences. On the other hand, for the surface condition, the values of r1 and r2, together with the product r1 × r2 = 0.055, suggest a copolymerization with a strong tendency toward cross-reactivity, meaning that each monomer would preferentially react with the other monomer rather than with itself. This is consistent with an alternating or random statistical distribution that would minimize the formation of long sequences or blocks of ZnDMA or PEGMA. It should be noted that these values are derived from a limited experimental dataset (four composition points). Nevertheless, the observed qualitative trends are considered representative of the system behavior under the studied conditions.
The observed change in reactivity at the film interface can be rationalized by two hypotheses: (i) an intrinsic modification of radical reactivity upon anchoring, or (ii) a differential monomer consumption, where ZnDMA, being more reactive than PEGMA, is depleted faster at the interface. The latter explanation appears more consistent, as it does not require a change in the intrinsic cross-reactivity rate constants of the monomers. Instead, the enrichment of PEGMA at the interface relative to the bulk arises from the faster depletion of ZnDMA due to its higher reactivity. Despite the operating mechanism, this change results in alternating or random statistical distribution and minimizes the formation of long sequences or blocks of ZnDMA or PEGMA as observed for bulk copolymers.
Overall, these results demonstrate that the copolymer composition can be precisely tuned via ATRP, although reactivity asymmetries, particularly for PEGMA in confined geometries, must be considered for predictive control. The obtained copolymers provide chemically diverse surfaces for subsequent MOF growth studies.
The poly(ZDMAx-co-PEGMAy) bulk copolymer with fZnDMA = 0.3 was selected for preparation of poly(ZDMAx-co-PEGMAy)@ZIF-8-bulk polyMOF. DLS analysis (see Fig. 5A) reveals a single size population for both the polyMOF and the bare poly(ZDMAx-co-PEGMAy) copolymer, revealing a single size population for both materials. PolyMOF particle size remains relatively constant for pH 6, 4, and 8, indicating added stability towards moderate acidic and alkaline conditions. This result highlights the structural robustness of the hybrid material within the well-established stability window of ZIF-8-based frameworks. In this context, pH stability was prioritized as a key operational parameter for aqueous applications. In contrast, bare polymer scaffold exhibits a size variability consistent with a crosslinking structure featuring pH-sensitive units, namely methacrylate and zinc moieties.56,57 While pH = 6 size is approximately 450 nm, at pH = 4, partial protonation of the methacrylate –COO− groups reduce the polymer size to 170 nm, whereas for alkaline pH, partial hydroxylation of Zn2+ causes the polymer size to decrease down to 140 nm. Both processes modify the degree of crosslinking, leading to a significant size contraction, which is not reversible upon restitution of neutral pH conditions (see Fig. 5A).
TGA analysis (see Fig. 5B) shows the thermal profile of the polyMOF in comparison with the bare copolymer and bulk ZIF-8. Although the samples were previously dried at 60 °C, thermograms show an initial weight loss 100–150 °C corresponding to water present in ZIF-8 and polyMOF, but not in the bare polymer. According to its expected stability, ZIF-8 shows no additional thermal events until nearly 400 °C, followed by an abrupt weight loss around 450 °C, an event also present in the polyMOF. Comparison between polymer and polyMOF confirms the presence of similar features: the polymer exhibits a major weight loss near 250 °C and a smaller event around 400 °C. Both events are also present in the polyMOF, with the first one shifted slightly to lower temperatures, likely due to a thermal destabilization effect of ZIF-8 on the polymeric backbone. Aside from both DLS and TGA suggesting the successful integration of the MOF into a polyMOF composite, XRD patterns show evidence of expected diffraction peaks corresponding to ZIF-8 (as detailed in the SI). To obtain clear information regarding crystallinity, we acquired high-resolution TEM images while minimizing radiation damage for both the neat copolymer and the polyMOF samples. Wide field images were acquired under identical imaging conditions, allowing direct comparison. In the case of the polyMOF, regions with varying contrast can be observed within the same particle, which are not present in the bare copolymer. This suggests the possible inclusion of crystalline domains associated with the MOF component, due to its higher electron density. This observation is consistent with the difficulty of resolving distinct crystalline peaks in the XRD patterns. The relatively weak XRD signals observed for the polyMOF samples are to be expected due to intrinsic features of these hybrid materials, where polymer chains are interwoven within the MOF framework. This results in an effective dilution of the crystalline diffraction signal, while covalent tethering of polymer chains to metallic centers induces lattice distortion and microstrain, leading to peak broadening. In addition, polymer-induced nano-confinement may limit the growth of ZIF-8 crystallites to small domains (<10–20 nm), further reducing peak definition due to Scherrer broadening. Together, these effects are consistent with the attenuated diffraction patterns typically observed in polyMOF systems. While low-angle XRD would provide a more accurate probe of the expanded periodicities in these materials, additional HR-TEM analysis (as detailed in the SI) revealed lattice fringes of 1.6 ± 0.2 nm, confirming the presence of crystalline ordering distinct from amorphous phases or ZnO formation.
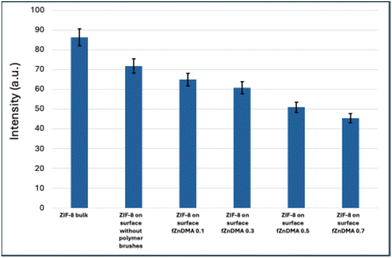
SEM analysis (see Fig. 6), which includes images corresponding to the extreme compositions (fZnDMA = 0.1 and 0.7), while images for the intermediate compositions (fZnDMA = 0.3 and 0.5) as well as bulk ZIF-8 and ZIF-8 on glass are presented in the SI, reveals a strong inverse correlation between FZnDMA and PolyMOF particle size for the surface-grafted polymeric platform (see Fig. 7); the following key aspects are proposed to rationalize such observations. The distribution of ZnDMA units is hypothesized to act as a spatial template dictating the density of nucleation sites. At high FZnDMA, the close spacing of Zn2+ ions is expected to generate a high local concentration, leading to rapid, simultaneous nucleation. Because the diffusing 2-methylimidazole (HmIm) ligand would be rapidly consumed by numerous adjacent nuclei, crystal growth may become sterically and chemically constrained, yielding smaller crystal sizes. Conversely, at low FZnDMA, the sparse distribution of Zn centers would result in fewer, isolated nuclei that can “harvest” HmIm from a larger volume, promoting larger, discrete ZIF-8 crystals.
 | ||
| Fig. 6 (A and B) SEM image of surface grafted polyMOF on poly(ZnDMA-co-PEGMA)@ZIF8-surface with 0.1 and 0.7fZnDMA. | ||
Another aspect that may contribute is the formation of ionic clusters. Being an ionic monomer in a relatively low-dielectric polymer environment, ZnDMA is likely prone to forming ionic clusters or aggregates driven by electrostatic interactions. These clusters are proposed not to behave strictly as single atomic nucleation sites but rather as “super-nucleation sites” that could trigger the growth of polycrystalline aggregates (e.g., raspberry-like structures) from a single dense ionic pocket. If these clusters are too large, Zn2+ ions may become partially trapped within them and thus be sterically shielded from the HmIm ligand, creating unreacted zones that could potentially compromise the mechanical integrity of the polymer–MOF interface. We further hypothesize that the interface may be governed by a nanoscale “dissolution–crystallization” mechanism; in this scenario, the interface would not correspond to a sharp boundary but rather to a gradient interpenetration region where methacrylate chains are threaded through the outer layers of the growing ZIF-8 crystals. The chain mobility (relative to the Tg during synthesis) is expected to influence how effectively ZnDMA units can rearrange to feed the growing nuclei without introducing excessive lattice strain into the MOF framework.
It is important to note that these mechanistic interpretations are inferential and consistent with the observed SEM trends, but they do not constitute direct experimental evidence. SEM primarily reflects the final morphology and does not independently resolve the nanoscale distribution of ZnDMA units or nucleation kinetics. We aim to perform experiments of scattering at wide and small angle (SAXS, WAXS) which can contribute to gain further insight on initial nucleation kinetics. Another possible way for this end would be to generate in situ quantitative 13C-NMR or MALDI-TOF MS to resolve the precise nanoscale distribution of ZnDMA units and monomer sequence fractions.
Conclusions
This study demonstrates the successful synthesis of poly(ZnDMA-co-PEGMA) copolymers with tailored microstructure across the full compositional range, both in bulk and as surface-grafted polymer brushes, establishing versatile platforms for ZIF-8 polyMOF growth. Under bulk conditions, the copolymerization behavior (r1(ZnDMA) > r2(PEGMA)) as estimated from the Fineman-Ross analysis revealed a higher reactivity of ZnDMA and a preferential reaction of poly(ZnDMA) radicals toward the ZnDMA monomer. Consequently, the copolymerization is suggested to proceed in an unbalanced manner, giving rise to blocky or sequential microstructures with enhanced ZnDMA incorporation and the possible formation of ZnDMA-enriched sequences, in agreement with thermal analyses. It should be noted that these reactivity ratios are derived from a limited experimental dataset (four composition points, some at moderate conversion), and therefore their absolute values may be subject to uncertainty; however, the observed qualitative trends are considered representative of the system behavior under the studied conditions. In contrast, under surface-initiated polymerization conditions, the copolymerization behavior (r1(ZnDMA) < r2(PEGMA) and r1 × r2 ≪ 1) suggests that the process is dominated by cross-reactivity, leading to alternating or random statistical microstructures and tending to minimize the formation of long ZnDMA or PEGMA sequences, as confirmed by X-ray photoelectron spectroscopy. This microstructural shift is proposed to enable the generation of chemically diverse polymer platforms suitable for the controlled growth of ZIF-8 polyMOFs. These mechanistic interpretations, including the relationship between copolymer microstructure and nucleation behavior, are consistent with the observed trends but remain inferential, as SEM primarily reflects the final morphology and does not directly resolve nanoscale ZnDMA distribution or nucleation kinetics. The bulk copolymer is proposed to act as an effective nucleation point for ZIF-8, yielding poly(ZnDMAx-co-PEGMAy)@ZIF-8-bulk polyMOFs that remained stable toward pH variations. In contrast, polymer brushes enabled the direct growth of ZIF-8 polyMOFs on surfaces, demonstrating that controlling FZnDMA can be used to modulate the size and surface coverage of the crystals, thereby enabling the design of polyMOF platforms with tunable nanoscale architectures. Overall, these results establish that copolymer composition and microstructure can be tuned via atom transfer radical polymerization and highlight the potential of ZnDMA–PEGMA copolymers as primers for the controlled growth of polyMOFs, while the proposed structure–property relationships should be interpreted in light of the aforementioned limitations and will be further explored in future studies.Author contributions
The manuscript was written through contributions of all authors. Experimental design: M. C. L., J. M. G., J. J. R., M. R.; monomer and polymer synthesis: M. C. L., A. I., J. M. G.; performance of experiments: M. C. L., A. I., J. M. P., C. V. P.; data analysis: J. M. G., J. J. R.; writing of the first manuscript draft: M. C. L., J. J. R.; writing of manuscript: M. R., A. I., I. N. V., J. M. G.; all authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting the findings of this study are available within the article and its supplementary information (SI). Additional data are available from the corresponding author upon reasonable request.Acknowledgements
The work was performed with funds of YPF Tecnología S.A. The authors would like to thank to YPF-Tecnología S. A (Y-TEC) for giving permission for this publication and resources. MCL gratefully recognize Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina, for postdoctoral fellowship. JMG, JJR, JMP, CVP and MR are researcher members of CONICET. AI, JMG, JMP, CVP, INV and JJR working at Y-TEC.References
- R. Robson, The Historical Development of the Concepts Underlying the Design and Construction of Targeted Coordination Polymers/MOFs: A Personal Account, Chem. Rec., 2024, 24, 5 CrossRef PubMed.
- H. C. Zhou, J. R. Long and O. M. Yaghi, Introduction to metal-organic frameworks, Chem. Rev., 2012, 112, 673–674 CrossRef CAS PubMed.
- Z. Wang, L. Liu, Z. Li, N. Goyal, T. Du and J. He, et al., Shaping of Metal-Organic Frameworks: A Review, Energy Fuels, 2022, 36, 2927–2944 CrossRef CAS.
- Z. Chen, K. O. Kirlikovali, P. Li and O. K. Farha, Reticular Chemistry for Highly Porous Metal-Organic Frameworks: The Chemistry and Applications, Acc. Chem. Res., 2022, 55, 579–591 CrossRef CAS PubMed.
- D. Li, A. Yadav, H. Zhou, K. Roy, P. Thanasekaran and C. Lee, Advances and Applications of Metal-Organic Frameworks (MOFs) in Emerging Technologies: A Comprehensive Review, Global Challenges, 2024, 8, 2300244 CrossRef PubMed.
- O. K. Farha, I. Eryazici, N. C. Jeong, B. G. Hauser, C. E. Wilmer and A. A. Sarjeant, et al., Metal-organic framework materials with ultrahigh surface areas: Is the sky the limit?, J. Am. Chem. Soc., 2012, 134, 15016–15021 CrossRef CAS PubMed.
- H. Furukawa, N. Ko, Y. B. Go, N. Aratani, S. B. Choi and E. Choi, et al., Ultrahigh porosity in metal-organic frameworks, Science, 2010, 329, 4242–4428 CrossRef PubMed.
- Z. Wang, Y. Cheng, C. Wang, R. Guo, J. You and H. Zhang, Optimizing the performance of Fe-based metal-organic frameworks in photo-Fenton processes: Mechanisms, strategies and prospects, Chemosphere, 2023, 339, 139673 CrossRef CAS PubMed.
- L. G. Gordeeva, Y. D. Tu, Q. Pan, M. L. Palash, B. B. Saha and Y. I. Aristov, et al., Metal-organic frameworks for energy conversion and water harvesting: A bridge between thermal engineering and material science, Nano Energy, 2021, 84, 105946 CrossRef CAS.
- X. Zhang, M. Yan, P. Chen, J. Li, Y. Li and H. Li, et al., Emerging MOFs, COFs, and their derivatives for energy and environmental applications, Innovation, 2025, 6, 100778 CAS.
- M. Xu, P. Cai, S. S. Meng, Y. Yang, D. S. Zheng and Q. H. Zhang, et al., Linker Scissoring Strategy Enables Precise Shaping of Metal–Organic Frameworks for Chromatographic Separation, Angew. Chem., Int. Ed., 2022, 61, 37 Search PubMed.
- S. K. Firooz and D. W. Armstrong, Metal-organic frameworks in separations: A review, Anal. Chim. Acta, 2022, 1234, 340208 CrossRef CAS PubMed.
- Y. R. Tao and H. J. Xu, A critical review on potential applications of Metal-Organic frameworks (MOFs) in adsorptive carbon capture technologies, Appl. Therm. Eng., 2024, 236, 121504 CrossRef.
- B. Mohanty, S. Kumari, P. Yadav, P. Kanoo and A. Chakraborty, Metal-organic frameworks (MOFs) and MOF composites based biosensors, Coord. Chem. Rev., 2024, 519, 2216102 CrossRef.
- H. Lin, Y. Yang, Y. C. Hsu, J. Zhang, C. Welton and I. Afolabi, et al., Metal–Organic Frameworks for Water Harvesting and Concurrent Carbon Capture: A Review for Hygroscopic Materials, Adv. Mater., 2024, 36, 12 Search PubMed.
- H. A. Almassad, R. I. Abaza, L. Siwwan, B. Al-Maythalony and K. E. Cordova, Environmentally adaptive MOF-based device enables continuous self-optimizing atmospheric water harvesting, Nat. Commun., 2022, 13, 4873 CrossRef CAS PubMed.
- J. H. Wang, F. Kong, B. F. Liu, N. Q. Ren and H. Y. Ren, Preparation strategies of waste-derived MOF and their applications in water remediation: A systematic review, Coord. Chem. Rev., 2025, 533, 216534 CrossRef CAS.
- T. Uemura, K. Kitagawa, S. Horike, T. Kawamura, S. Kitagawa and M. Mizuno, et al., Radical polymerisation of styrene in porous coordination polymers, Chem. Commun., 2005, 48, 5968 RSC.
- Z. Zhang, H. T. H. Nguyen, S. A. Miller and S. M. Cohen, PolyMOFs: A Class of Interconvertible Polymer-Metal-Organic-Framework Hybrid Materials, Angew. Chem., Int. Ed., 2015, 54, 6152–6157 CrossRef CAS PubMed.
- K. A. McDonald, J. I. Feldblyum, K. Koh, A. G. Wong-Foy and A. J. Matzger, Polymer@MOF@MOF: ‘grafting from’ atom transfer radical polymerization for the synthesis of hybrid porous solids, Chem. Commun., 2015, 51, 11994–11996 RSC.
- Y. Gu, M. Huang, W. Zhang, M. A. Pearson and J. A. Johnson, PolyMOF Nanoparticles: Dual Roles of a Multivalent polyMOF Ligand in Size Control and Surface Functionalization, Angew. Chem., Int. Ed., 2019, 58, 16676–16681 CrossRef CAS PubMed.
- N. Mukherjee, A. Das, S. Mukhopadhyay, S. K. Das and T. Jana, Grafting of Polymer Brushes on MOF Surface to Achieve Proton-Conducting Membranes, ACS Appl. Polym. Mater., 2024, 6, 846–858 CrossRef CAS.
- M. H. Zahir, A. Helal and A. S. Hakeem, Hybrid polyMOF Materials Prepared by Combining an Organic Polymer with a MOF and Their Application for Solar Thermal Energy Storage, Energy Fuels, 2021, 35, 10199–10209 CrossRef CAS.
- S. Fan, C. Liang, F. Feng, K. Wong, K. Wang and S. Jia, et al., Polymer-MOF Network Enabling Ultrathin Coating for Post-Combustion Carbon Capture, Angew. Chem., Int. Ed., 2025, 64, 10 Search PubMed.
- A. K. Bindra, D. Wang and Y. Zhao, Metal–Organic Frameworks Meet Polymers: From Synthesis Strategies to Healthcare Applications, Adv. Mater., 2023, 35, e2300700 CrossRef PubMed.
- X. Cui, G. Kong, S. Wei, Z. Zhang, Z. Kang and H. Guo, PolyMOF interlayers modulated interfacial polymerization of ultra-thin nanofiltration membranes with efficient and stable desalination performance, J. Membr. Sci., 2024, 702, 122780 CrossRef CAS.
- H. Kim and S. M. Cohen, Metal-Organic Frameworks Constructed from Branched Oligomers, Inorg. Chem., 2024, 63, 1853–1857 CrossRef CAS PubMed.
- F. Jin, J. Liu, Y. Chen and Z. Zhang, Tethering Flexible Polymers to Crystalline Porous Materials: A Win–Win Hybridization Approach, Angew. Chem., Int. Ed., 2021, 60, 1853–1857 CrossRef PubMed.
- M. Kalaj, K. C. Bentz, S. Ayala, J. M. Palomba, K. S. Barcus and Y. Katayama, et al., MOF-Polymer Hybrid Materials: From Simple Composites to Tailored Architectures, Chem. Rev., 2020, 120, 8267–8302 CrossRef CAS PubMed.
- J. M. Palomba, S. Ayala and S. M. Cohen, polyMOF Formation from Kinked Polymer Ligands via ortho-Substitution, Isr. J. Chem., 2018, 58, 1123–1126 CrossRef CAS.
- S. Ayala, Z. Zhang and S. M. Cohen, Hierarchical structure and porosity in UiO-66 polyMOFs, Chem. Commun., 2017, 53, 3058–3061 RSC.
- S. W. Li, M. F. Hsieh, T. Hong, P. Chen, K. Osada and X. Liu, et al., Block Copolymer-Stabilized Metal–Organic Framework Hybrids Loading Pd Nanoparticles Enable Tumor Remission Through Near-Infrared Photothermal Therapy, Adv. NanoBiomed Res., 2024, 4, 1 Search PubMed.
- Y. Katayama, M. Kalaj, K. S. Barcus and S. M. Cohen, Self-Assembly of Metal-Organic Framework (MOF) Nanoparticle Monolayers and Free-Standing Multilayers, J. Am. Chem. Soc., 2019, 141, 20000–20003 CrossRef CAS PubMed.
- M. Rafti, J. A. Allegretto, G. M. Segovia, J. S. Tuninetti, J. M. Giussi and E. Bindini, et al., Metal-organic frameworks meet polymer brushes: Enhanced crystalline film growth induced by macromolecular primers, Mater. Chem. Front., 2017, 1, 2256–2260 RSC.
- J. A. Allegretto, A. Iborra, J. M. Giussi, C. von Bilderling, M. Ceolín and S. Moya, et al., Growth of ZIF-8 MOF Films with Tunable Porosity by using Poly (1-vinylimidazole) Brushes as 3D Primers, Chem. – Eur. J., 2020, 26, 12388–12396 CrossRef CAS PubMed.
- K. S. Park, Z. Ni, A. P. Côté, J. Y. Choi, R. Huang and F. J. Uribe-Romo, et al., Exceptional chemical and thermal stability of zeolitic imidazolate frameworks, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 10186–10191 CrossRef CAS PubMed.
- J. A. Allegretto, D. Onna, S. A. Bilmes, O. Azzaroni and M. Rafti, Unified Roadmap for ZIF-8 Nucleation and Growth: Machine Learning Analysis of Synthetic Variables and Their Impact on Particle Size and Morphology, Chem. Mater., 2024, 36, 5814–5825 CrossRef CAS PubMed.
- D. Fairen-Jimenez, S. A. Moggach, M. T. Wharmby, P. A. Wright, S. Parsons and T. Düren, Opening the gate: Framework flexibility in ZIF-8 explored by experiments and simulations, J. Am. Chem. Soc., 2011, 133, 8900–8902 CrossRef CAS PubMed.
- C. O. Ania, E. García-Pérez, M. Haro, J. J. Gutiérrez-Sevillano, T. Valdés-Solís and J. B. Parra, et al., Understanding gas-induced structural deformation of ZIF-8, J. Phys. Chem. Lett., 2012, 3, 1159–1164 CrossRef CAS PubMed.
- J. Cousinsaintremi, T. Rémy, V. Vanhunskerken, S. Vandeperre, T. Duerinck and M. Maes, et al., Biobutanol separation with the metal-organic framework ZIF-8, ChemSusChem, 2011, 4, 1074–1077 CrossRef CAS PubMed.
- M. Bergaoui, M. Khalfaoui, A. Awadallah-F and S. Al-Muhtaseb, A review of the features and applications of ZIF-8 and its derivatives for separating CO2 and isomers of C3- and C4- hydrocarbons, J. Nat. Gas Sci. Eng., 2021, 96, 104389 Search PubMed.
- Y. Sun, T. Shu, J. Ma, Q. Dai, P. Peng and Z. Zhou, et al., Rational Design of ZIF-8 for Constructing Luminescent Biosensors with Glucose Oxidase and AIE-Type Gold Nanoclusters, Anal. Chem., 2022, 94, 3408–3417 CrossRef CAS PubMed.
- A. Paul, I. K. Banga, S. Muthukumar and S. Prasad, Engineering the ZIF-8 Pore for Electrochemical Sensor Applications-A Mini Review, ACS Omega, 2022, 7, 26993–27003 CrossRef CAS PubMed.
- D. M. D'Alessandro, B. Smit and J. R. Long, Carbon dioxide capture: Prospects for new materials, Angew. Chem., Int. Ed., 2010, 49, 6058–6082 CrossRef PubMed.
- S. R. Venna and M. A. Carreon, Highly permeable zeolite imidazolate framework-8 membranes for CO 2/CH4 separation, J. Am. Chem. Soc., 2010, 132, 76–78 CrossRef CAS PubMed.
- H. Bux, F. Liang, Y. Li, J. Cravillon, M. Wiebcke and J. Caro, Zeolitic imidazolate framework membrane with molecular sieving properties by microwave-assisted solvothermal synthesis, J. Am. Chem. Soc., 2009, 131, 16000–16001 CrossRef CAS PubMed.
- N. S. Bobbitt, M. L. Mendonca, A. J. Howarth, T. Islamoglu, J. T. Hupp and O. K. Farha, et al., Metal-organic frameworks for the removal of toxic industrial chemicals and chemical warfare agents, Chem. Soc. Rev., 2017, 46, 3357–3385 RSC.
- J. Schindelin, I. Arganda-Carreras, E. Frise, V. Kaynig, M. Longair and T. Pietzsch, et al., Fiji: An open-source platform for biological-image analysis, Nat. Methods, 2012, 9, 676–682 CrossRef CAS PubMed.
- J. J. Romero, M. C. De Rossi, C. Oses, C. V. Echegaray, P. Verneri and M. Francia, et al., Nucleus-cytoskeleton communication impacts on OCT4-chromatin interactions in embryonic stem cells, BMC Biol., 2022, 20, 6 CrossRef CAS PubMed.
- E. Mastan and S. Zhu, A Molecular Weight Distribution Polydispersity Equation for the ATRP System: Quantifying the Effect of Radical Termination, Macromolecules, 2015, 48, 18 CrossRef.
- A. B. Samui, V. G. Dalvi, L. Chandrasekhar, M. Patri and B. C. Chakraborty, Interpenetrating polymer networks based on nitrile rubber and metal methacrylates, J. Appl. Polym. Sci., 2006, 99, 5 CrossRef.
- I. C. McNeill, M. Zulfiqar and C. Urie, Thermal degradation of zinc polymethacrylate and a zinc methacrylate/methyl methacrylate copolymer, Polym. Degrad. Stab., 1984, 9, 239–250 CrossRef CAS.
- T. Kos, A. Anžlovar, D. Pahovnik, E. Žagar, Z. C. Orel and M. Žigon, Zinc-containing block copolymer as a precursor for the in situ formation of nano ZnO and PMMA/ZnO nanocomposites, Macromolecules, 2013, 46, 17 CrossRef.
- B. P. Payne, M. C. Biesinger and N. S. McIntyre, X-ray photoelectron spectroscopy studies of reactions on chromium metal and chromium oxide surfaces, J. Electron Spectrosc. Relat. Phenom., 2011, 184, 1–2 CrossRef.
- J. H. Scofield, TID-4500, UC-34 Physics THEORETICAL PHOTOIONIZATION CROSS SECTIONS FROM I TO 1500 keV. UCRL-51326. 1973.
- M. Eren and H. K. Can, Preparation of zinc methacrylate-methylmethacrylate-butyl acrylate emulsions and their application in exterior paints, Prog. Org. Coat., 2019, 135, 424–437 CrossRef CAS.
- Y. Zhang and M. Muhammed, Critical evaluation of thermodynamics of complex formation of metal ions in aqueous solutions - VI. Hydrolysis and hydroxo-complexes of Zn2+ at 298.15 K, Hydrometallurgy, 2001, 60, 3 CrossRef.
Footnote |
| † Present address: Institute for Integrative Biology of the Cell (I2BC), CEA, CNRS, Université Paris-Saclay, 91198 Gif-sur-Yvette, France. |
| This journal is © The Royal Society of Chemistry 2026 |