 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bifunctional Pd sites on Pd4/Co3O4 nanowires for high selectivity of pyrazine electrocatalytic hydrogenation with water as the hydrogen source
Fangcheng Qiua,
Siyi Chenb,
Xin Zhenga,
Hanyu Lia,
Bowen Chenb,
Shaowen Tanb,
Xue Hanb and
Shengping Wang *b
*b
aElectric Power Research Institute, Yunnan Power Grid Co., Ltd, Kunming 650220, China
bFaculty of Material Science and Chemistry, China University of Geosciences, Wuhan 430074, China. E-mail: spwang@cug.edu.cn
First published on 19th February 2026
Abstract
Liquid organic hydrogen carriers (LOHCs) are considered highly promising materials for hydrogen storage. However, hydrogenation and dehydrogenation of LOHCs often require harsh reaction conditions. The hydrogenation of nitrogen-containing heterocyclic organic molecules in aqueous media is particularly challenging. This work presents an efficient electrochemical hydrogen storage system based on pyrazine as a nitrogen-containing organic hydrogen carrier. In this electrochemical system, water serves as both the solvent and the hydrogen source for pyrazine hydrogenation. Pd4/Co3O4/NF demonstrates high electrochemical activity under ambient temperature and pressure. At −0.25 V (vs. RHE), the hydrogenation conversion rate of pyrazine reaches 99.4% with the selectivity rate of 97.1%. Pd acts as the catalytic active center. It not only promotes water electrolysis to generate H* but also facilitates the adsorption of pyrazine, providing sites for its hydrogenation. Pd4/Co3O4/NF maintains high catalytic activity after 8 cycles, showing excellent stability. This electrochemical hydrogenation of pyrazine proceeds without using hydrogen gas. Therefore, the system offers superior safety performance.
1 Introduction
The rapid depletion of fossil fuels and severe environmental pollution have made the development of new energy sources a major global focus.1 Hydrogen, as a clean energy carrier, is regarded as an ideal alternative to address the world's energy and environmental challenges.2 However, its large-scale application is hindered by the challenge of developing efficient, safe, and economical storage and transportation methods.3 Currently, compressed hydrogen storage is widely used due to its low cost and simple operation.4 Yet, it suffers from low volumetric density and safety concerns. Liquid hydrogen storage offers higher density, suitable for large-scale, long-distance transport.5 However, it requires expensive cryogenic equipment and faces evaporation loss risks. Solid-state hydrogen storage materials, like carbon-based materials, metal hydrides, and MOFs, provide better safety.6 However, they often require harsh conditions for hydrogenation/dehydrogenation and show poor reversibility. Recently, liquid organic hydrogen carriers (LOHCs) have shown great promise for large-scale hydrogen logistics.7 They offer good reversibility, high hydrogen capacity, and compatibility with existing fuel infrastructure. Aromatic hydrocarbons of LOHCs have been widely studied with capacities of 6.1–7.3 wt%, but their catalytic hydrogenation and dehydrogenation typically need high temperatures (200–400 °C). The research indicates that nitrogen-containing heterocyclic compounds (N-LOHCs) can significantly lower the energy demands of these processes.8 Examples like N-propylcarbazole, 1-methylindole, N-ethylindole, and 7-ethylindole demonstrate excellent performance.9–13 The thermal catalytic hydrogenation/dehydrogenation for N-LOHCs still requires temperatures of 120–200 °C. It often relies on flammable hydrogen gas or other expensive, toxic hydrogen sources, posing safety and environmental risks.Electrocatalytic technology holds broad applications in the hydrogen energy sector. Compared to thermal catalysis, it offers advantages such as milder reaction conditions, easily tunable selectivity, and the direct use of water as a hydrogen source.14 Pan et al. employed water as the hydrogen source and achieved complete conversion of 10 mM quinoline with nearly 100% selectivity over a non-precious Cu-based catalyst after 1 h at a potential of −1.275 V (vs. Hg/HgO). Their work revealed that Cu+–Cu0 dual sites promote hydrogenation via a proton-coupled electron transfer (PCET) mechanism, and the coupled system (ECH||BAOR) operates at a lower voltage with higher efficiency.15 Guo et al. reported a fluorine-modified cobalt (Co–F) catalyst that enables high-yield hydrogenation of quinoline at room temperature. Under an optimal potential of −1.1 V (vs. Hg/HgO), 0.1 mmol quinoline was fully converted within 6 h with 99% selectivity.16 A MoNi4 catalyst achieved 99% yield of tetrahydroquinoxaline from 0.3 mM quinoxaline within 1 h through consecutive PCET steps at a very low overpotential of −0.05 V (vs. RHE).17 Such electrochemical hydrogen storage systems are safer, more environmentally friendly, and simpler to operate, demonstrating promising application prospects. Existing research has predominantly focused on quinoline and quinoxaline. In these compounds, hydrogenation and dehydrogenation occur only in the N-heterocyclic part of the bicyclic structure, resulting in a relatively low gravimetric hydrogen storage capacity. Moreover, both compounds exhibit poor solubility, making them less ideal as substrates for electrocatalytic LOHC-based hydrogen storage. Recent studies have used Rh/KB as a cathode catalyst in an anion-exchange membrane (AEM) electrolyzer to reduce nitrogen-containing aromatic compounds and for the first time accomplished the electrocatalytic hydrogenation of pyrazine with a yield of 96%.18 Pyrazine offers a considerable theoretical hydrogen storage capacity of 6.97% and is fully miscible with water, rendering it a more promising candidate for hydrogen storage carriers.
The efficiency of LOHC electrochemical hydrogenation (ECH) is primarily determined by the availability of H*. Therefore, developing electrocatalysts with excellent water dissociation capability and modulating the adsorption/desorption behavior of organic molecules on the electrode surface are key to achieving highly efficient LOHC conversion. Noble metals such as Pd, Pt, Ru, and Ir are widely studied due to their optimal H* adsorption/desorption properties.19,20 Pd exhibits strong H* adsorption capability, second only to Pt among them. Additionally, its cost is significantly lower. Under ambient conditions, H+ ions can occupy the octahedral sites in the Pd lattice, leading to the formation of PdHn.21 Pd shows high adsorption selectivity, fast kinetics, and good reversibility, making it highly promising for ECH. It demonstrates the highest catalytic activity for the hydrogenation of aromatic aldehydes.22 The weak adhesion between the active Pd phase and the substrate typically results in a low loading mass, which limits the number of available active sites.23 Furthermore, Pd nanoparticles are prone to detach from the substrate during prolonged operation or at high current densities. Thus, strong interfacial contact is crucial for electron conductivity and catalytic stability. Co3O4 can anchor and disperse catalyst nanoparticles as a substrate material. This enhances the loading of active components and provides abundant catalytic sites.24 The strategies involving defect engineering, doping, and constructing heterointerfaces have emerged as promising approaches to tune the electronic structure of Co3O4 and improve its overall catalytic performance.25 For instance, the strong electronic coupling at the interface between RuO2 and Co3O4 accelerates reaction kinetics and enhances catalyst stability. The heterointerface in RuO2@Co3O4 not only directly participates in the reaction but also modulates the d-band center of the catalyst. This optimization adjusts the adsorption strength of key intermediates, thereby improving the hydrogen evolution reaction (HER) activity.26 Liao et al. confirmed the presence of a strong metal–support interaction between Au nanoparticles and oxygen vacancy-rich Co3O4 nanorods (Co3O4 NRs–OVs). This interaction facilitates electron transfer from Co3O4 NRs–OVs to Au. The increased electron density on the Au surface selectively adsorbs the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group of vanillin and promotes catalytic hydrogen transfer for alcohol production.27 Similarly, an Ir–Co3O4 electrocatalyst with low Ir content and abundant oxygen vacancies (OV) has been developed. This catalyst features rich three-dimensional active sites, rapid electron transfer, and oxygen-vacancy-induced optimized kinetics for oxygenated intermediate formation. These properties collectively enhance the oxygen evolution reaction (OER) performance in acidic environments.28
O group of vanillin and promotes catalytic hydrogen transfer for alcohol production.27 Similarly, an Ir–Co3O4 electrocatalyst with low Ir content and abundant oxygen vacancies (OV) has been developed. This catalyst features rich three-dimensional active sites, rapid electron transfer, and oxygen-vacancy-induced optimized kinetics for oxygenated intermediate formation. These properties collectively enhance the oxygen evolution reaction (OER) performance in acidic environments.28
In this work, a series of Pdx/Co3O4/NF as ECH electrocatalysts of pyrazine with heterointerfaces and high OV content were synthesized via a room-temperature reduction method. The Co3O4 layer was directly grown on the nickel foam (NF) surface. This approach not only provided abundant anchoring sites for Pd loading but also created a tight interfacial structure. It avoided issues associated with polymer binders, thereby effectively improving the utilization of active components. Cobalt atoms modulated the electronic structure of the catalysts. Through strong metal–support interaction, they regulated the adsorption and activation of reaction substrates. This enhancement strengthened the adsorption–desorption behavior of Pd towards H* atoms and organic molecules. Pyrazine was selected as the hydrogen carrier; the pyrazine hydrogenation to produce piperazine had a theoretical hydrogen storage density of 6.97 wt%. The effects of Pd loading, catalyst morphology, and electrochemical performance on the ECH behavior of pyrazine were systematically investigated, and the hydrogenation mechanism was also studied. Pd4/Co3O4/NF exhibited high electrocatalytic activity for the hydrogenation of pyrazine and dehydrogenation of piperazine. It achieved optimal hydrogenation conversion and selectivity. The hydrogenation mechanism revealed that the water-mediated ECH process completely eliminates safety risks associated with the use of external hydrogen gas. In summary, this electrochemical hydrogen storage system is mild, rapid, efficient, and simple. It overcomes the practical limitations of traditional methods.
2 Experimental
2.1 Catalyst preparation
Preparation of Pdx/Co3O4/NF. NF (2 cm × 3 cm × 3 mm) was sequentially ultrasonically cleaned in absolute ethanol, 2 M hydrochloric acid, and absolute ethanol again, for 5 min each. Afterwards, it was rinsed thoroughly and air-dried. The homogeneous solution prepared by dissolving 1.746 g of Co(NO3)2·6H2O (0.03 M), 0.617 g of NH4F (0.083 M), and 1.401 g of urea (0.05 M) in 50 mL of deionized water, along with the pretreated NF, was transferred into a 100 mL Teflon-lined autoclave. The autoclave was subjected to a hydrothermal reaction at 120 °C for 6 h. The resulting precursor was collected and sequentially washed with absolute ethanol and deionized water. It was then dried in a vacuum oven at 60 °C for 8 h. Finally, the precursor was calcined in a muffle furnace at 400 °C for 3 h under an air atmosphere to obtain Co3O4/NF.Different amounts of PdCl2 (1.2 mM, 1.8 mM, 2.4 mM, 3.0 mM) and corresponding amounts of NaCl (2.4 mM, 3.6 mM, 4.8 mM, 6.0 mM) were added to separate 50 mL portions of deionized water. These mixtures were magnetically stirred in a constant-temperature water bath at 40 °C for 30 min until they turned clear and orange-yellow. The Co3O4/NF precursor was fixed and fully immersed in each clear solution. Stirring was continued at 40 °C until the solution color changed from orange-yellow to colorless and transparent. The catalyst was then retrieved, rinsed with deionized water, and dried overnight at room temperature. The resulting catalysts were designated as Pd2/Co3O4/NF, Pd3/Co3O4/NF, Pd4/Co3O4/NF, and Pd5/Co3O4/NF.
2.2 Material characterization
The morphology and elemental distribution of the samples were determined via a Hitachi SU8010 scanning electrode microscope (SEM) equipped with an X-ray energy dispersive spectrometer (EDS). The crystallinity of the electrocatalysts was characterized by X-ray diffraction (XRD) using a Bruker/D8 advance powder diffractometer with Cu Kα radiation (λ = 1.5418 Å) with a scanning range of 5° to 90° and a scan rate of 10° min−1. X-ray photoelectron spectroscopy (XPS) was carried out using a Thermo Fisher ESCALAB 250Xi X-ray photoelectron spectrometer with Al Kα X-ray radiation. The binding energy of C 1s = 284.80 eV was used as the energy standard for charge correction.The concentrations of pyrazine/piperazine in the electrolyte were quantified using gas chromatography (GC, Panno A60). The GC system was equipped with an AB-1 column (30 m × 0.32 mm × 0.25 μm). The injector temperature, detector temperature, and oven temperature were all set at 300 °C. The oven temperature program involved ramping from 70 °C to 180 °C at a rate of 10 °C min−1.
To ensure analytical reliability, a strict standardization protocol was followed. High-purity commercial standards of pyrazine and piperazine were used to identify their GC retention times. The standard solutions (0.1–1.0 mg mL−1) were analyzed in quintuplicate to establish a linear calibration curve relating peak area to concentration. All catalytic samples were quantified using this external standard method to calculate pyrazine conversion and piperazine selectivity (Fig. S1).
2.3 Electrochemical measurements
A catalyst electrode (10 mm × 10 mm) served as the working electrode. A platinum electrode (10 mm × 10 mm) and an Hg/HgO electrode were used as the counter electrode and reference electrode, respectively. The Hg/HgO electrode was filled with 1 M KOH as the internal electrolyte without a salt bridge. The electrolyte in the working electrode compartment was 40 mL of 1 M KOH containing 10 mM pyrazine (or just 40 mL of 1 M KOH for control experiments), with a magnetic stirring rate of 400 rpm. The counter electrode compartment contained 40 mL of 1 M KOH (pH = 13.8).
All measured potentials were converted to the reversible hydrogen electrode (RHE) scale using the Nernst equation (eqn (1)).
| ERHE (V) = E(Hg/HgO) + 0.059 × pH + E0(Hg/HgO) = E(Hg/HgO) + 0.059 × pH + 0.098 | (1) |
η = a + b × log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j j
| (2) |
| ECSA = Cdl/Cs | (3) |
 | (4) |
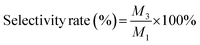 | (5) |
3 Results and discussion
3.1 Material characterization
 | ||
| Fig. 1 (a) XRD patterns and (b) Co 2p, (c) O 1s, (d) Pd 3d XPS spectra of Co3O4/NF and Pd4/Co3O4/NF. | ||
The Co 2p XPS spectrum of Co3O4/NF showed two main peaks at 779.79 and 795.04 eV, which were attributed to Co 2p3/2 and Co 2p1/2, respectively (Fig. 1b). Specifically, the peaks at 779.49 and 794.68 eV were assigned to Co3+, and those at 781.18 and 796.68 eV were assigned to Co2+.33 The peak intensity of Co3+ was significantly higher than that of Co2+, which was consistent with the XRD results. For the Pd 3d spectrum of Pd4/Co3O4/NF, the peak at 343.67 eV was identified as a satellite peak, and those at 335.67 and 340.94 eV were assigned to Pd0 3d5/2 and Pd Pd0 3d3/2, respectively (Fig. 1d). A peak corresponding to Pd2+ 3d5/2 was observed at 337.72 eV.34 In the Co 2p spectrum, the peaks at 780.54 and 796.45 eV belonged to Co 2p3/2 and Co 2p1/2, respectively (Fig. 1b). The separation of these peaks by a spin–orbit splitting confirmed the coexistence of Co3+ and Co2+.35 The peaks at 780.32 and 795.00 eV were attributed to Co3+, and those at 782.29 and 797.34 eV were attributed to Co2+. Additionally, the peaks at 786.29, 789.66 and 802.78 eV were identified as satellite peaks associated with Co 2p3/2 and Co 2p1/2, characteristic of the spinel structure.35
Compared with the Co 2p3/2 binding energy of Co3O4/NF, that of Pd4/Co3O4/NF increased. This suggested spontaneous electron transfer from cobalt (electronegativity of 1.88) to the more electronegative palladium (electronegativity of 2.20), rendering the Pd species more electron-rich.34 The increased electron filling caused a downshift in the d-band center of Pd. According to the d-band center theory, this downshift typically led to a moderate weakening of adsorption energy. This optimized adsorption strength contributed to higher activity and selectivity. The results further confirmed that Pd primarily existed in the metallic Pd0 state. The presence of Pd2+ indicated a strong interaction between the Pd nanoparticles and the substrate. This interaction was beneficial for enhancing the long-term stability of Pd4/Co3O4/NF during the ECH process (Fig. 1d).
Both Co3O4/NF and Pd4/Co3O4/NF exhibited three oxygen states. OL was attributed to typical metal–oxygen bonds, OV was associated with oxygen vacancies related to defects with low oxygen coordination and OC resulted from the physical and chemical adsorption of water on the material's surface.36 In the O 1s spectrum of Pd4/Co3O4/NF, the peaks at 529.55, 531.13, and 532.45 eV corresponded to OL, OV and OC, respectively (Fig. 1c). Compared to Co3O4/NF, the OV concentration in Pd4/Co3O4/NF was significantly higher. These positively charged defect centers indirectly influenced the adsorption capacity of Pd metal sites by altering the local electron density. The OV increase promoted catalysis and enhanced the intrinsic catalytic activity.28
3.2 Electrochemical tests
The commercial Pd/C catalyst exhibited a hydrogen evolution current of −31.0 mA cm−2 at −0.25 V (vs. RHE). The current remained essentially unchanged after the addition of pyrazine, and a net current only appeared at more negative potentials (Fig. S2a). This is primarily due to the binder used to immobilize Pd/C on the NF surface, which is nearly non-conductive and partially hinders the catalytic performance of Pd/C. Moreover, Pd/C shows relatively poor dispersion, resulting in an electrochemically active surface area much smaller than that of Pd4/Co3O4/NF.
3.3 Pyrazine ECH mechanism of Pd4/Co3O4/NF
4 Conclusion
A series of Pdx/Co3O4/NF (x = 2, 3, 4, 5) as electrocatalysts of pyrazine ECH were successfully synthesized via hydrothermal and room-temperature reduction methods. Their ECH mechanism and the performance of pyrazine were systematically investigated. Among them, Pd4/Co3O4/NF demonstrated the optimal electrochemical activity. It achieved a remarkable conversion rate of 99.4% with a selectivity rate of 97.1% under ambient temperature and pressure. Furthermore, Pd4/Co3O4/NF maintained high electrocatalytic activity and a largely unchanged microstructure after chronoamperometry of 8 cycles, highlighting its excellent stability. Mechanistic studies revealed that Pd acted as the catalytic center with dual functions, which promoted water electrolysis to generate H* and also facilitated the adsorption of pyrazine, providing the active sites for ECH. The direct growth of the Co3O4 layer on the NF substrate offered abundant anchoring sites for Pd, resulting in a tight interfacial structure. The Co atoms modulated the electronic structure and through strong metal–support interactions enhanced the adsorption–desorption behavior of H* and organic molecules on Pd. Compared to traditional hydrogen storage systems, pyrazine ECH with Pd4/Co3O4/NF was safer, more environmentally friendly, and simpler. It will eliminate the need for harsh reaction conditions and is compatible with green electricity generated from wind, water, and other renewable sources, demonstrating broad application prospects and providing an important reference for future research.Nonetheless, this study also acknowledges a limitation, that is, the obtained faradaic efficiency remains relatively low. This aspect leaves considerable room for improvement in future work. Further optimization of catalyst design and reaction conditions should focus on enhancing charge utilization efficiency while maintaining high conversion and selectivity, which is crucial for the practical deployment of electrocatalytic LOHC systems.
Conflicts of interest
There are no conflicts to declare.Data availability
All study data are included in the article and/or supplementary information (SI) Appendix.Supplementary information: electrochemical tests and corresponding parameters, and SEM images, EDS mapping of the catalysts. See DOI: https://doi.org/10.1039/d5lf00408j.
Acknowledgements
This work was supported by the R&D Project of China Southern Power Grid Co., Ltd (YNKJXM20240023).References
- M. Barani, K. Löffler, P. C. del Granado, N. Moskalenko, E. Panos, F. M. Hoffart, C. von Hirschhausen, M. Kannavou, H. Auer, K. Hainsch, T. G. Grandón, S. Mathisen and A. Tomasgard, European energy vision 2050 and beyond: Designing scenarios for Europe's energy transition, Renewable Sustainable Energy Rev., 2026, 225, 116074, DOI:10.1016/j.rser.2025.116074.
- Z. Wang, X. Liu, W. Gu, L. Cheng, H. Wang, J. Liu, L. Li, J. Wang, M. Zhang, Y. Wang and Y. Shen, Power-to-hydrogen-to-power as a pathway for wind and solar renewable energy utilization in China: Opportunities and challenges, Renewable Sustainable Energy Rev., 2026, 226, 116195, DOI:10.1016/j.rser.2025.116195.
- M. Habibi, M. G. Hosseini and K. Wang, Toward sustainable energy: A comprehensive review of hydrogen production, storage, and utilization, Renewable Sustainable Energy Rev., 2026, 226, 116193, DOI:10.1016/j.rser.2025.116193.
- Z. Hua, W. Gao, S. Chi, X. Wang and J. Zheng, Development status and challenges of high-pressure gaseous hydrogen storage vessels and cylinders in China, Renewable Sustainable Energy Rev., 2025, 214, 115567, DOI:10.1016/j.rser.2025.115567.
- X. Xu, H. Xu, J. Zheng, L. Chen and J. Wang, A high-efficiency liquid hydrogen storage system cooled by a fuel-cell-driven refrigerator for hydrogen combustion heat recovery, Energy Convers. Manage., 2020, 226, 113496, DOI:10.1016/j.enconman.2020.113496.
- J. K. Abifarin, J. F. Torres and Y. Lu, 2D materials for enabling hydrogen as an energy vector, Nano Energy, 2024, 129, 109997, DOI:10.1016/j.nanoen.2024.109997.
- Y. Li, X. Guo, S. Zhang and Y. He, A perspective review on N-heterocycles as liquid organic hydrogen carriers and their hydrogenation/dehydrogenation catalysts, Energy Fuels, 2024, 38, 12447–12471, DOI:10.1021/acs.energyfuels.4c01633.
- K. C. Tan, T. He, Y. S. Chua and P. Chen, Recent advances of catalysis in the hydrogenation and dehydrogenation of N-heterocycles for hydrogen storage, J. Phys. Chem. C, 2021, 125, 18553–18566, DOI:10.1021/acs.jpcc.1c04783.
- L. Liu, Q. Zhang, C. Deng, C. Li, Y. Zhao, R. Gao, T. Zhu, Y. Dong, H. Cheng and M. Yang, Pyridine N-induced intra-pore and interfacial dual-confined Pd0-Pdδ+ synergistic catalysis for ultra-stable dehydrogenation of dodecahydro-N-propylcarbazole, Appl. Catal., B, 2024, 351, 123987, DOI:10.1016/j.apcatb.2024.123987.
- M. Yang, G. Cheng, D. Xie, T. Zhu, Y. Dong, H. Ke and H. Cheng, Study of hydrogenation and dehydrogenation of 1-methylindole for reversible onboard hydrogen storage application, Int. J. Hydrogen Energy, 2018, 43, 8868–8876, DOI:10.1016/j.ijhydene.2018.03.134.
- Y. Dong, H. Zhao, Y. Zhao, M. Yang, H. Zhang and H. Cheng, Study of catalytic hydrogenation and dehydrogenation of 2,3-dimethylindole for hydrogen storage application, RSC Adv., 2021, 11, 15729–15737, 10.1039/d1ra01552d.
- Y. Dong, M. Yang, Z. Yang, H. Ke and H. Cheng, Catalytic hydrogenation and dehydrogenation of N-ethylindole as a new heteroaromatic liquid organic hydrogen carrier, Int. J. Hydrogen Energy, 2015, 40, 10918–10922, DOI:10.1016/j.ijhydene.2015.05.196.
- Z. Chen, M. Yang, T. Zhu, Z. Zhang, X. Chen, Z. Liu, Y. Dong, G. Cheng and H. Cheng, 7-ethylindole: A new efficient liquid organic hydrogen carrier with fast kinetics, Int. J. Hydrogen Energy, 2018, 43, 12688–12696, DOI:10.1016/j.ijhydene.2018.03.088.
- O. Lebedeva, D. Kultin, A.
 alenchuk and L.
alenchuk and L.  ustov, Advances and prospects in electrocatalytic hydrogenation of aromatic hydrocarbons for synthesis of “loaded” liquid organic hydrogen carriers, Curr. Opin. Electrochem., 2023, 38, 101207, DOI:10.1016/j.coelec.2022.101207.
ustov, Advances and prospects in electrocatalytic hydrogenation of aromatic hydrocarbons for synthesis of “loaded” liquid organic hydrogen carriers, Curr. Opin. Electrochem., 2023, 38, 101207, DOI:10.1016/j.coelec.2022.101207. - Y. Pan, Z. Bao, C. Wang, Z. Wang, P. Xu, X. Bai, X. Shi, H. Zheng, H.-E. Wang and L. Zheng, Electrochemical hydrogenation of quinoline enabled by Cu0-Cu+ dual sites coupled with efficient biomass valorization in aqueous solution, Adv. Funct. Mater., 2024, 35, 2414120, DOI:10.1002/adfm.202414120.
- S. Guo, Y. Wu, C. Wang, Y. Gao, M. Li, B. Zhang and C. Liu, Electrocatalytic hydrogenation of quinolines with water over a fluorine-modified cobalt catalyst, Nat. Commun., 2022, 13, 5297, DOI:10.1038/s41467-022-32933-6.
- M. Li, C. Liu, Y. Huang, S. Han and B. Zhang, Water-involving transfer hydrogenation and dehydrogenation of N-heterocycles over a bifunctional MoNi4 electrode, Chin. J. Catal., 2021, 42, 1983–1991, DOI:10.1016/S1872-2067(21)63834-2.
- N. Shida, Y. Shimizu, A. Yonezawa, J. Harada, Y. Furutani, Y. Muto, R. Kurihara, J. N. Kondo, E. Sato, K. Mitsudo, S. Suga, S. Iguchi, K. Kamiya and M. Atobe, Electrocatalytic hydrogenation of pyridines and other nitrogen-containing aromatic compounds, J. Am. Chem. Soc., 2024, 146, 30212–30221, DOI:10.1021/jacs.4c09107.
- Y. Inami, H. Ogihara, S. Nagamatsu, K. Asakura and I. Yamanaka, Synergy of Ru and Ir in the electrohydrogenation of toluene to methylcyclohexane on a Ketjenblack-supported Ru-Ir alloy cathode, ACS Catal., 2019, 9, 2448–2457, DOI:10.1021/acscatal.8b03610.
- G. Yang, V. Maliekkal, X. Chen, S. Eckstein, H. Shi, D. M. Camaioni, E. Baráth, G. L. Haller, Y. Liu, M. Neurock and J. A. Lercher, Rate enhancement of phenol hydrogenation on Pt by hydronium ions in the aqueous phase, J. Catal., 2021, 404, 579–593, DOI:10.1016/j.jcat.2021.11.003.
- S. Sarkar and S. C. Peter, An overview on Pd-based electrocatalysts for the hydrogen evolution reaction, Inorg. Chem. Front., 2018, 5, 2060–2080, 10.1039/c8qi00042e.
- L. C. Meyer, U. Sanyal, K. A. Stoerzinger, K. Koh, J. L. Fulton, D. M. Camaioni, O. Y. Gutiérrez and J. A. Lercher, Influence of the molecular structure on the electrocatalytic hydrogenation of carbonyl groups and H2 evolution on Pd, ACS Catal., 2022, 12, 11910–11917, DOI:10.1021/acscatal.2c03207.
- H. Sun, Z. Yan, F. Liu, W. Xu, F. Cheng and J. Chen, Self-supported transition-metal-based electrocatalysts for hydrogen and oxygen evolution, Adv. Mater., 2020, 32, 1806326, DOI:10.1002/adma.201806326.
- Z. Yan, H. Sun, X. Chen, H. Liu, Y. Zhao, H. Li, W. Xie, F. Cheng and J. Chen, Anion insertion enhanced electrodeposition of robust metal hydroxide/oxide electrodes for oxygen evolution, Nat. Commun., 2018, 9, 2373, DOI:10.1038/s41467-018-04788-3.
- H. Yuan, S. Wang, Z. Ma, M. Kundu, B. Tang, J. Li and X. Wang, Oxygen vacancies engineered self-supported B doped Co3O4 nanowires as an efficient multifunctional catalyst for electrochemical water splitting and hydrolysis of sodium borohydride, Chem. Eng. J., 2021, 404, 126474, DOI:10.1016/j.cej.2020.126474.
- Y. Jiang, H. Liu, Y. Jiang, Y. Mao, W. Shen, M. Li and R. He, Adjustable heterointerface-vacancy enhancement effect in RuO2@Co3O4 electrocatalysts for efficient overall water splitting, Appl. Catal., B, 2023, 324, 122294, DOI:10.1016/j.apcatb.2022.122294.
- Q. Liao, M. Shi, Q. Zhang, W. Cheng, P. Ji, X. Fu, H. Lai, R. Fan, J. Sheng and H. Li, Gold catalyst anchored to pre-reduced Co3O4 nanorods for the hydrodeoxygenation of vanillin using alcohols as hydrogen donors, ACS Appl. Mater. Interfaces, 2022, 14, 3939–3948, DOI:10.1021/acsami.1c18197.
- Z. Bai, L. Li, P. Yin and T. Lei, Ultralow iridium content and oxygen vacancy-rich Ir–Co3O4 electrocatalyst to boost acidic oxygen evolution, Int. J. Hydrogen Energy, 2025, 128, 76–84, DOI:10.1016/j.ijhydene.2025.04.230.
- C. C. L. McCrory, S. Jung, J. C. Peters and T. F. Jaramillo, Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction, J. Am. Chem. Soc., 2013, 135, 16977–16987, DOI:10.1021/ja407115p.
- C. Feng, L. Xuebin, C. Wei, W. Na and J. Yanqiao, Double core-shell hollow nanocage CuO@Co3O4 for the selective hydrogenolysis of C–O bonds of lignin without external hydrogen, Chem. Eng. J., 2024, 491, 152002, DOI:10.1016/j.cej.2024.152002.
- S. Wang, S. Zhang, Z. Zhang, Y. Tan, K. Liang, X. Guo and X. Kong, Reversible electrochemical hydrogen storage of quinoxaline utilizing Pd/NF dual-function electrocatalyst under mild conditions, Int. J. Hydrogen Energy, 2024, 49, 719–728, DOI:10.1016/j.ijhydene.2023.07.089.
- T. A. Revathy, K. Dhanapal, S. Dhanavel, V. Narayanan and A. Stephen, Pulsed electrodeposited dendritic Pd-Ni alloy as a magnetically recoverable nanocatalyst for the hydrogenation of 4-nitrophenol, J. Alloys Compd., 2018, 735, 1703–1711, DOI:10.1016/j.jallcom.2017.11.264.
- T. Wang, P. Wang, W. Zang, X. Li, D. Chen, Z. Kou, S. Mu and J. Wang, Nanoframes of Co3O4-Mo2N heterointerfaces enable high-performance bifunctionality toward both electrocatalytic HER and OER, Adv. Funct. Mater., 2022, 32, 2107382, DOI:10.1002/adfm.202107382.
- Z. Feng and X. Bai, Enhanced activity of bimetallic Pd-Ni nanoparticles on KIT-6 for production of hydrogen from dodecahydro-N-ethylcarbazole, Fuel, 2022, 329, 125473, DOI:10.1016/j.fuel.2022.125473.
- Z. Zhang, Z. Yang, C. Wei, Z. Liu and T. Mu, Facet-dependent electrocatalytic oxidation activity of Co3O4 nanocrystals for 5-hydroxymethylfurfural, Green Chem., 2023, 25, 8196–8206, 10.1039/d3gc02398b.
- M. Lan, B. Liu, R. Zhao, M. Dong, X. Wang, L. Fang and L. Wang, Dandelion-like CuCo2O4 arrays on Ni foam as advanced positive electrode material for high-performance hybrid supercapacitors, J. Colloid Interface Sci., 2020, 566, 79–89, DOI:10.1016/j.jcis.2020.01.077.
- M. Zeng and Y. Li, Recent advances in heterogeneous electrocatalysts for the hydrogen evolution reaction, J. Mater. Chem. A, 2015, 3, 14942–14962, 10.1039/c5ta02974k.
- W. Zhao, W. Fu, H. Yang, C. Tian, M. Li, Y. Li, L. Zhang, Y. Sui, X. Zhou, H. Chen and G. Zou, Electrodeposition of Cu2O films and their photoelectrochemical properties, CrystEngComm, 2011, 13, 2871–2877, 10.1039/c0ce00829j.
- Z.-C. Yao, J. Chai, T. Tang, L. Ding, Z. Jiang, J. Fu, X. Chang, B. Xu, L. Zhang, J.-S. Hu and L.-J. Wan, Manipulating hydrogenation pathways enables economically viable electrocatalytic aldehyde-to-alcohol valorization, Proc. Natl. Acad. Sci. U. S. A., 2025, 122, e2423542122, DOI:10.1073/pnas.2423542122.
- T. Cao, J. Cheng, Y. Xiang, L. Hu, X. Hu, L. Li, X. Huang and Z. Wei, Activating surface oxygen in Ce/Mo-Doped Ni oxyhydroxide for synergistically enhancing furfural oxidation and hydrogen evolution at ampere-level current densities, Angew. Chem., Int. Ed., 2025, 64, e202506017, DOI:10.1002/anie.202506017.
- C. Chauhan, T. Gupta and B. Mondal, Deciphering the role of second metal in M-Ni (M = Fe, Ni, and Mn) heterobimetallic electrocatalysts in controlling the HAT versus hydride transfer mechanism for the dehydrogenation of alcohols, Small, 2025, 21, 2410228, DOI:10.1002/smll.202410228.
- Z. Du, W. Ran, H. Zhao, J. Ma, J. Sun, B. Liu, S. Lin and R. Liu, Fully exposed Pd ensembles on ultrathin Co3O4 nanosheets: A reductive-oxidative dual-active catalyst for the detoxification of chlorophenol, ACS ES T Eng., 2024, 4, 1401–1411, DOI:10.1021/acsestengg.4c00003.
| This journal is © The Royal Society of Chemistry 2026 |







