Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
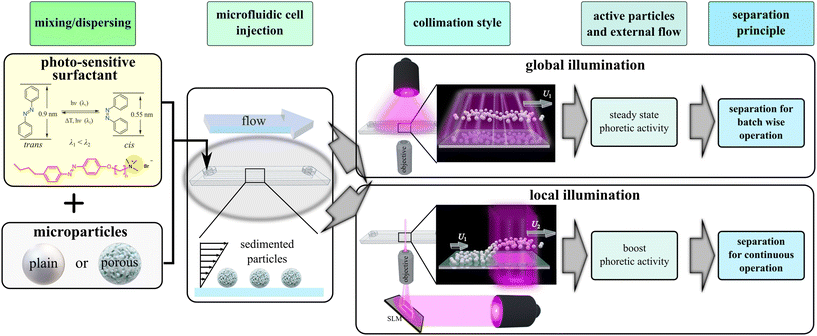
Phototunable hopping of microparticles enables surface-selective continuous separation via microfluidics
Fabian
Rohne
 a,
Daniela
Vasquez Muñoz
a,
Yulia
Gordievskaya
a,
Cevin
Braksch
a,
Isabel
Meier
a,
Anjali
Sharma
a,
Sarah
Loebner
a,
Anne
Nitschke
b,
Nino
Lomadze
a,
Andreas
Taubert
b,
Svetlana
Santer
a,
Daniela
Vasquez Muñoz
a,
Yulia
Gordievskaya
a,
Cevin
Braksch
a,
Isabel
Meier
a,
Anjali
Sharma
a,
Sarah
Loebner
a,
Anne
Nitschke
b,
Nino
Lomadze
a,
Andreas
Taubert
b,
Svetlana
Santer
 a and
Marek
Bekir
a and
Marek
Bekir
 *a
*a
aInstitute of Physics and Astronomy, University of Potsdam, 14476 Potsdam, Germany. E-mail: marek.bekir@uni-potsdam.de
bInstitute of Chemistry, University of Potsdam, 14476 Potsdam, Germany
First published on 2nd April 2026
Abstract
High surface area materials are central to next-generation technologies, yet their purification remains limited by lab-scale methods. We present a continuous-flow technique that separates microparticles by surface area and porosity. The method relies on light-responsive surfactants that generate surface-localized photochemical activity under illumination, scaling with particle surface area. Particles with sufficient activity undergo light-induced “hopping” into an overlying channel, enabling deflection across streamlines and obstacles. Non-porous particles remain confined, while porous particles are diverted to secondary outlets. We demonstrate control of hopping efficiency through illumination parameters, establishing a scalable platform for high-throughput, surface-sensitive purification.
Introduction
Next-generation applications increasingly demand advanced functional materials with highly specialized properties. Among the wide array of critical material characteristics, one universally advantageous and frequently required attribute, particularly for dynamic and responsive systems, is a high specific surface area. Materials exhibiting high surface areas offer enhanced interfacial contact with their environment, thereby significantly improving mass and energy exchange processes and enabling a more efficient response to external stimuli.This makes porous microparticles of growing considerable interest, as their high surface-to-volume ratios underpin performance enhancements across a diverse range of technological applications.1 These include heterogeneous catalysis,2–4 next-generation battery systems,5–7 fabrication of porous 2d macroscopic interfaces with colloidal lithography,8 heat-energy storage media,9 and adsorption-based technologies such as activated carbon for wastewater treatment,10–12 among others.13–16 In each of these fields, the porous architecture of the particles directly influences functional efficiency, making morphological control, especially porosity, a central design criterion.
However, the scalable fabrication of such porous microparticles remains a significant challenge. Conventional synthesis methods often yield particles with broad distributions in size and structural heterogeneity, particularly in terms of porosity and internal surface area. Such variability can severely compromise performance consistency in end-use applications. For instance, in catalysis or electrochemical energy storage, minor deviations in surface area can lead to disproportionately large drops in activity or capacity.
Therefore, beyond size-based classification, it is important to develop strategies for the selective separation and fractionation of particles based on their porosity or, effectively, their accessible surface area. Despite the widespread use of particle sizing techniques at both laboratory17–21 and industrial scales,22–24 methods for surface based (includes porosity based) separation have only recently gained traction, driven largely by advances in surface-chemistry-mediated fractionation approaches.25 Recent advances have demonstrated the integration of phoretic or osmotic activity with microfluidic platforms, wherein colloidal particles are suspended in aqueous media containing dissolved salts.25 By employing controlled injection protocols to establish localized salt concentration gradients, active colloids can be guided toward specific regions within the microchannel. Under laminar flow conditions, this method facilitates selective migration, segregation, and subsequent collection of particles through spatially separated outlets under continuous operation. Rather than relying on microfluidic-based fractionation techniques, Zheng et al. demonstrated that particles can be locally separated and remotely guided to predefined location down to micron scale through spatially controlled light illumination. This approach is enabled by functionalizing the particles with light-responsive dyes, allowing selective manipulation via externally applied illumination patterns.26 An alternative approach for particle separation leverages differences in interfacial properties by exploiting surface morphology characteristics such as roughness, porosity, and surface charge. This strategy offers enhanced flexibility, enabling the discrimination of particles with identical sizes but distinct surface chemistries or interfacial compositions.27,28
Surface-sensitive fractionation can be realized through the integration of microfluidic technology with light-induced phoretic activity of microparticles, as illustrated in Scheme 1. Initially, microparticles are suspended in a solution containing a photo-responsive surfactant, {(6-[4-(4-hexylphenylazo)-phenoxyl-butyl-trimethylammoniumbromide], abbreviated AzoC6, (Scheme 1))},27 and subsequently introduced into the microfluidic channel. Sedimented microparticles move along with the carrier fluid flow inside the separation cell. Upon illumination, the photo-responsive surfactant renders the particles to be phoretic/osmotic active due to the release of cis isomers from particles interface.27,28 The bottom wall acts as a reflective boundary for the cis-isomers, which cause a hovering of the sedimented particles during light illumination. It has been previously demonstrated that hovering height and translational velocity of the particles depends on the strength of the light induced activity and thus on the interfacial properties of the particles themselves.27,28
Two principal illumination strategies may be employed: global27,28 and locally29 collimated light profiles. In the case of global collimation, as illustrated in Scheme 1, the entire separation region is uniformly illuminated. This results in a sustained velocity difference between distinct particle fractions, enabling separation based on retention principles.30,31 The method benefits from low-cost instrumentation and straightforward sample preparation; however, its primary limitation lies in throughput.28,32 Since the separation process operates in a batchwise manner, it is less suited for high-throughput applications involving large quantities of dispersed particles.
To improve the throughput, one needs to transition from a batchwise to a continuous separation mode. This forms the central focus of the present study, in which we propose a framework for continuous, surface-sensitive separation based on locally collimated light illumination, as depicted in Scheme 1. In this approach, particles entering the illuminated region experience a transient enhancement in phoretic activity, which can be sufficiently strong to enable them to traverse physical obstacles. Specifically, this activity allows certain particles to be levitated into a secondary, elevated microchannel, from which they are extracted via a separate outlet. Such selective fractionation based on interfacial properties is feasible only for particles exhibiting sufficiently strong light-induced phoretic responses.27 For example, only particles with high surface porosity may generate enough propulsion to surmount step heights of approximately 100 μm along otherwise planar interfaces. Such high levitation requires strong phoretic activity, which can be achieved by using the principles of l-LDDO only when the applied wavelength is in the UV range.33,35,36
We previously demonstrated particle fractionation in batchwise operation using a modified field-flow fractionation (FFF) principle.36 This approach was shown to be effective for several distinct particle pair types, including particles with porous versus non-porous interfacial structures,27 particles composed of different polymeric materials by tuning the wavelength of the applied illumination,28 and particles exhibiting only minor differences in surface functionalization through the use of a chemically active interface.31
The approach presented in this work departs from these earlier studies27,28,31,36 by translating the separation concept from batchwise to continuous operation. While the same aqueous photosensitive surfactant solution is employed, the separation mechanism is fundamentally changed by replacing global illumination with spatially localized light collimation. This modification enables particle levitation above obstacles and enables continuous selective fractionation of one species. In the following, we first analyze the levitation strength as a function of illumination wavelength, followed by a demonstration of how this effect can be employed as a continuous separation mechanism.
Theory: from velocity as a proxy to predict levitation height as a function of the wavelength
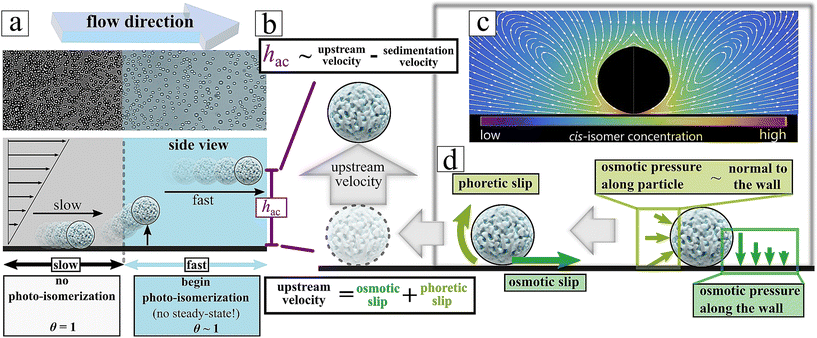
The levitation dynamics of particles suspended in fluid within a laminar microchannel are strongly governed by the local hydrodynamic environment. Owing to their comparatively large mass and micron-scale dimensions, yet still being much smaller than the characteristic geometric length scale of the channel (height = 0.54 mm), the particles undergo gravitational sedimentation. Consequently, they remain confined to a narrow near-wall region, either in direct contact with the bottom surface or levitated only a few micrometers above it. In this regime, the local flow field can be approximated by a linear shear profile, consistent with low-Reynolds-number hydrodynamics under Stokes flow conditions. When a particle is levitated at a height hac above the lower wall (see for illustration Fig. S1), it is situated in a region of laminar flow with a spatially varying velocity gradient. In this regime, conservation of mass and momentum leads to the development of a net drift velocity U, which scales with the local shear rate S. This shear rate is evaluated at a vertical position equal to the sum of the particle radius and levitation height, hac = a + hlev, where the flow profile remains approximately linear. Consequently, the particle's translational motion is governed by its hydrodynamic interaction with the spatially varying flow field at that elevation. Under the assumption that particles are positioned at the mid-width of a rectangular microchannel (Scheme 1) Brenner et al. derived an asymptotic expression,37,38 where the hovering tendency is connected with translational velocity as:27,28,36,37,39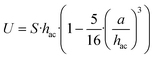 | (1) |
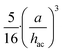 is very small and can be omitted in first approximation. This assumption holds true for strongly hovered particles discussed later in the results and discussion section under local light collimation. Then eqn (1) simplify into:
is very small and can be omitted in first approximation. This assumption holds true for strongly hovered particles discussed later in the results and discussion section under local light collimation. Then eqn (1) simplify into:| U ∼ S·hac = S·(a + hlev), | (2) |
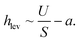 | (3) |
Results and discussion
Systematic investigation of velocity boost as a function of the wavelength
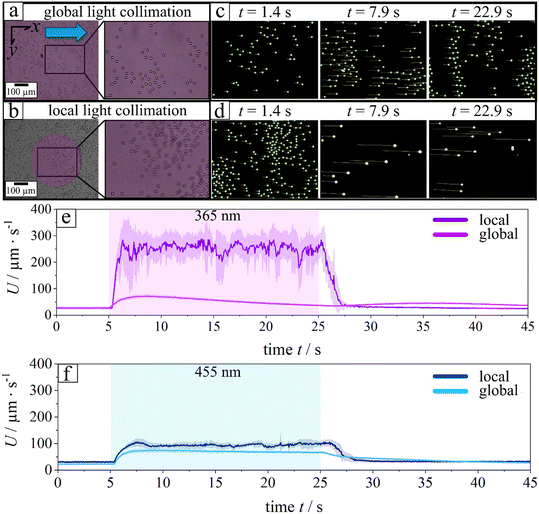
Before introducing the concept of continuous separation, we first address the rationale for employing UV light illumination. This choice is motivated by a systematic investigation of particle hovering behavior as a function of illumination wavelength. In all experiments, two illumination strategies were implemented: local and global collimation. The corresponding illuminated regions are shown for UV light in Video S1 and as snapshot series in Fig. 1a–d. Particle motion was quantified by tracking individual trajectories across the illumination region. For each frame, the mean particle velocity was calculated from all crossing trajectories, and the temporal evolution of the average velocity (bold line) together with the standard deviation (shaded region) is reported for local (Fig. 1b and d) and global collimation (Fig. 1a and c). All experiments were conducted in a rectangular microfluidic channel (height h = 0.54 mm, width w = 3.8 mm, length L = 17 mm) at a constant volumetric flow rate of 150 μL min−1, corresponding to a shear rate of 14.98 s−1 as determined from eqn (13) (Materials and methods section).Under local light collimation and particularly for UV light data exhibit a constant strong boost of entering microparticles and thus the average velocity maintains for the whole-time frame of illumination, while at global light particles experience a transient boost with subsequent decay to the initial velocity at no illumination. It is important to note that collimation from the bottom substrate minimizes the optical path length through the microfluidic channel. This improves the boosting tendency of chemical activity of the microparticles and strongly impact the levitation height (Fig. 2).
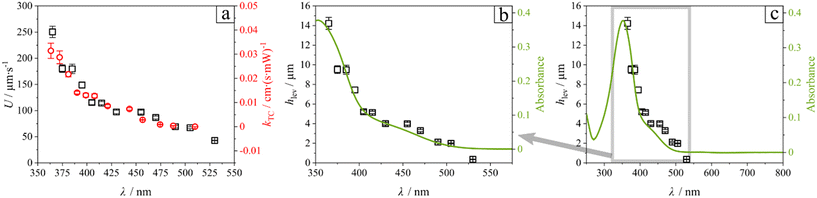
The average steady-state velocity, plotted in Fig. 3c and d for the final 5 s of illumination (t = 20–25 s), reveals that light-induced motion varies not only in velocity profile as a function of wavelength but also in absolute magnitude. Velocities under local illumination (see Fig. 3c) are consistently higher compared to global collimation (see Fig. 3d). Although the applied power was kept constant in all experiments (P = 11.5 mW), local collimation through the objective confines the light to a rectangular spot of only a few micrometers (200 × 300 μm), additionally loses power output leaving the objective to 0.8 mW, thereby substantially increasing the local intensity. This enhanced intensity amplifies the phoretic/osmotic activity, which scales with light intensity. As a result, particles exhibit stronger activity, attain larger hovering heights (hac), and reach higher velocities in agreement with eqn (1). In the following, we discuss in more detail the differences in motion patterns between the two collimation strategies.
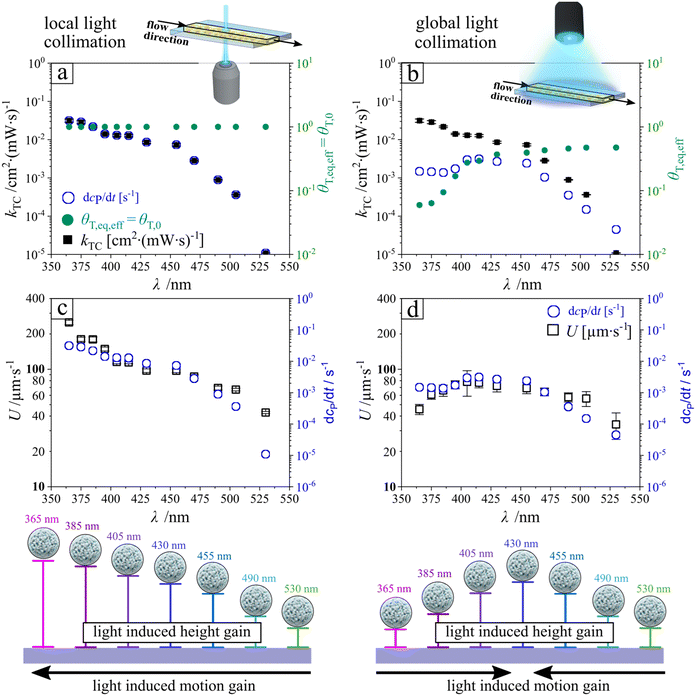 | ||
| Fig. 3 Key photochemical parameters under local (left, a and c) and global (right, b and d) illumination. (a and b) Steady-state isomer flux, dcP/dt, trans-isomer coverage θT, and trans-to-cis rate constant kTC as a function of wavelength, indicating local photoactivity and effective l-LDDO strength. (c and d) Corresponding particle velocities under the same spectral conditions, differing only in illumination geometry. Data in (b and d) are reproduced from ref. 28 to highlight the impact of illumination collimation on particle dynamics. Note, that local light collimation from the bottom of the substrate minimizes the optical pathway and drastically improves the boosting in levitation. | ||
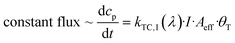 | (4) |
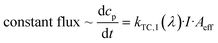 | (5) |
Data exhibit that the velocity follows the pattern of the trans isomer absorption spectrum. We explain this relation by the fact that the value of the photoisomerization rate constant is the product of the quantum yield φ, the extinction coefficient ε and the applied intensity of illumination I:40
| kphotoisomerization = φ·ε·I. | (6) |
Apparently, we observe that the levitation of the particle can be lower than the height of the obstacle as data in Fig. 5e and Video S4 clearly demonstrate a wall lift migration, while calculated levitation height hlev is much lower. We estimated the needed minimum elevation height of particles by calculating the value of hlev using eqn (3) in absence of any obstacle, where the applied illumination intensity has been precisely varied displayed in Fig. S5a. Then we compared the value of hlev with the hopping potential over the obstacle by performing same measurements with same variation in intensity displayed in Fig. S5b. Data exhibits that already 5 μm of levitation, approximately one diameter, is enough to successfully hop particles over the obstacle. Thus, besides the light induced levitation, hydrodynamic effects appear to play a critical role in the system, particularly in regions proximal to obstacles, where particle lift forces are significantly amplified. In general, when particles are in close proximity to channel walls or the bottom substrate, they experience a transverse pressure gradient. This gradient induces a hydrodynamic lift force directed away from the lower boundary (substrate) toward the center of the channel. This near-wall lift force FNw given by:41–43
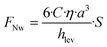 | (7) |
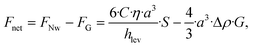 | (8) |
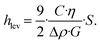 | (9) |
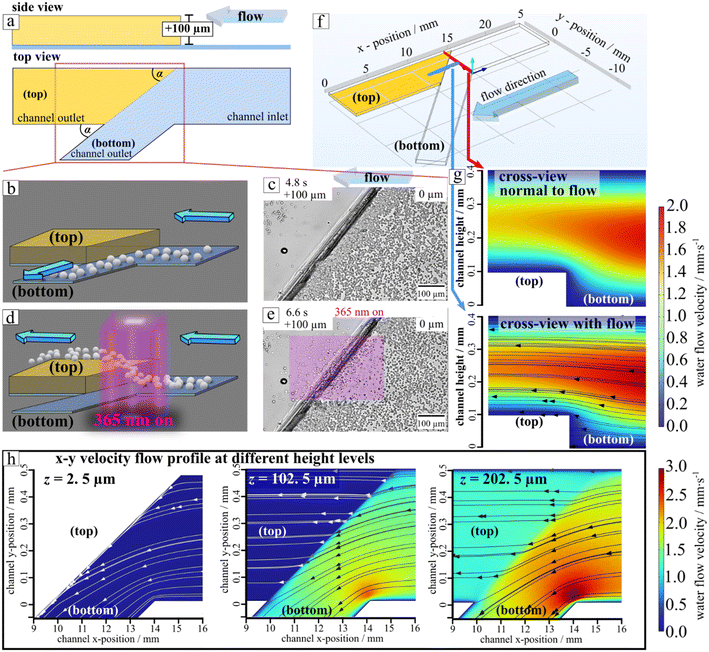
Thus, to further support the demonstration of the separation potential of hopping particles traversing diagonal obstacles, we perform numerical simulations of the fluid flow field using the COMSOL Multiphysics software. The simulation domain is illustrated in Fig. 5f, and representative cross-sectional velocity field profiles are presented in Fig. 5g both in the plane normal to the flow (top) and along the flow direction (bottom). As shown in Fig. 5g, the computed flow streamlines indicate that fluid can only traverse around the obstacle at specific vertical (z) positions within the microchannel. And if the light induced hovering reaches the critical vertical position the remaining distance can be overcome from the carrier fluid.
Notably, at an elevation corresponding to the particle radius (z = 2.5 μm), streamlines are directed from the vicinity of the wall toward outlet channel (bottom) (Fig. 5h, left). This result aligns with experimental observations from Video S4, where microparticles are passively advected along the wall when the illumination is off. At higher vertical positions specifically at the top of the obstacle (z = 102.5 μm, Fig. 6h, middle) and significantly above it (z = 202.5 μm, Fig. 5h, right) the flow streamlines are directed toward the upper outlet channel (top), with some extend in lateral deflection toward outlet (bottom). These flow characteristics correspond well with experimental trajectories of particles that are transiently elevated under UV illumination, demonstrating the influence of vertical position on particle routing via the local flow field.
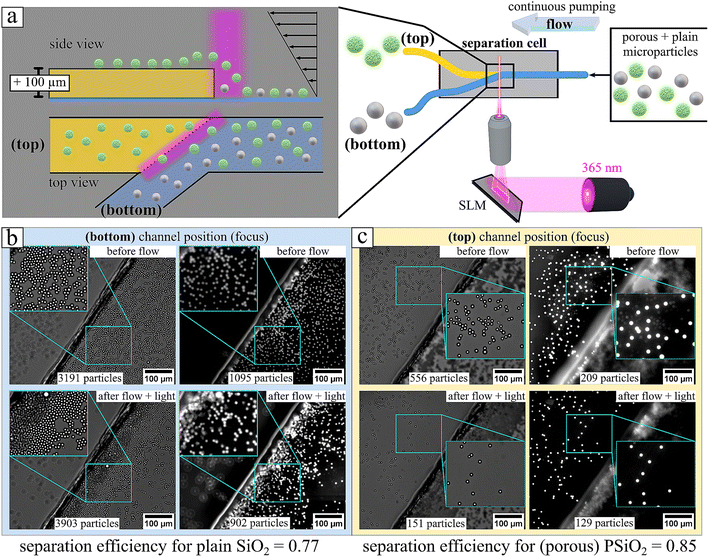
The hopping principle can be used to selectively fractionate particles based on interfacial properties. Levitation over high obstacles requires strong phoretic activity, which scales with the effective surface area Aeff and determines the trans-to-cis isomer flux. Under UV illumination (365 nm), porous particles with large Aeff levitate higher, whereas plain particles with small Aeff show negligible lift. A binary mixture of porous and plain particles thus separates, with porous particles elevated into the higher channel while plain particles remain below (Fig. 6, Video S5). Fluorescein dye in the solution enhances emission from porous particles, providing a visual confirmation of selective levitation during simultaneous UV illumination and emission microscopy.45,46 The pronounced elevation observed for the porous particles can be attributed to their substantially increased specific surface area compared to that of the plain particle interface. An influence arising solely from differences in intrinsic surface charge between the two particle types can be excluded. As shown in Fig. S4, both silica microparticles exhibit comparable negative zeta potentials in aqueous medium, consistent with the presence of deprotonated surface hydroxyl groups. Upon addition of a cationic surfactant (1 mM), both particle types display a positive zeta potential, which can be ascribed to the adsorption of the surfactant and the formation of a multilayer at the particle interface at the given concentration.47 Under UV irradiation, the zeta potential remains positive but decreases in magnitude. This reduction is attributed to photoisomerization-induced desorption processes: the predominance of cis isomers leads to partial desorption, resulting in a reduced surface coverage by the remaining trans isomers in accordance with the trans/cis population ratio.46 These observations confirm that the distinct elevation behavior of porous particles is not governed by differences in net surface charge, but rather by their enhanced interfacial area and the associated amplification of interfacial interactions.
Furthermore, we note that particle inertia is negligible under the investigated flow conditions. Consequently, the light-induced hopping behavior is interpreted to be governed primarily by advection along the streamlines rather than inertial effects. Based on the theoretical considerations presented in the literature,36 we calculated a Stokes number (St ≈ 3.32 × 10−4) much smaller than 1 for the system as detailed in SI section S8. The obtained result confirms that inertial contributions are minimal and do not significantly influence the particle dynamics.
Furthermore, to quantify the separation efficiency over an operation time of 30 s separation we calculated the number of particles in bright field and emission recording before and after separation. Bright field recording reveals the total number of all particles, porous and plain particles together, so NPSiO2 + NSiO2, while emission recording reveals only the porous particles, NPSiO2. Thus, one has all important information to calculate the separation efficiency, SE, with respect to porous microparticles equals the fraction of porous particles:
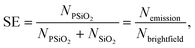 | (10) |
Analyzing now the bottom channel (bottom) initially we have a binary mixture of plain and porous particles with a ratio of 0.34 = 1095/3191, in good agreement with value for the initial top channel to be 0.37. After separation we observe a strong accumulation of plain particles, yet porous particles are still present, and it yields according to eqn (10) a SEPSiO2 for porous to be 0.23 = 902/3903. Given the fact that we expect at the bottom channel (bottom) an enriched population of plain particles accordingly the value of SESiO2 = 1 − SEPSiO2 and yields a value of 0.77 demonstrating an accumulation of the plain particle in the same order as for the porous in the upper channel. Quantification in the bottom channel should be approached with caution, as cumulative stacking of multiple particles leads to a significant increase in concentration, making precise particle counting impossible. Although the mixture is clearly not purified, the visible multilayer formation of plain particles indicates that these particles comprise the dominant fraction.
From Video S5 (Fig. 6), we demonstrated selective fractionation of porous particles. Continuous flow allows new particles to be hovered into the upper channel (top) while others are deflected into the lower channel (bottom) by the 40° wall angle. This enables continuous, surface-sensitive separation of particles with strong osmotic/phoretic activity, i.e., those with large surface areas. In this example, porous and plain silica particles of equal size were separated. We emphasize that a direct quantitative relationship between surface area and chemical activity cannot be established due to several additional factors, including surfactant diffusion within pores, surface functionalization, and electrostatic interactions. These factors may either limit or enhance the dynamic exchange of trans–cis isomers under illumination, thereby affecting the effective activity (e.g., the measured U(λ)). This conclusion is drawn from a comparison between surface areas measured via nitrogen sorption and surface areas inferred from chemical activity measurements.35 Nitrogen sorption measurements indicate an approximately fifth-fold larger pore volume for porous particles compared to non-porous particles displayed in Fig. S21 (data taken from reference),48 whereas an approximately 33-fold stronger chemical activity is reported for the same system in the absence of external flow.35 This might be attributed that the effective average pore diameter of the porous particles is measured 12 nm, in comparison to non-porous particles to be 2.5 nm (see Fig. S21b), which is in the dimension of the surfactant molecule size to be approximately 2 nm.49 Thus the strength in chemical activity and the separation potential shown in this work critically depends on the pore size.47,48 A detailed discussion is provided in the SI (section S6).
Unlike traditional batch-based methods, such as chromatographic or flow-field fractionation,27,29,30,38 this approach allows continuous operation, enhancing throughput and efficiency. To substantiate this claim, we estimate the mass throughput of fractionated porous particles by quantifying the number of particles crossing the obstacle within the detection area shown in Video S5. Specifically, number of crossing particles were counted within a region of 360 × 410 μm2 over an observation period of 30 s under continuous illumination.
The calculation in section S9 (SI) and data presented in Fig. S22 indicate that, assuming a homogeneous particle concentration across the channel width w, approximately 1585 particles per second are deflected into the upper channel via hydrodynamic levitation. Considering a particle diameter of 5 μm and a material density of 1.8 g cm−3, this corresponds to a processed sample mass of approximately 0.672 mg h−1 during one hour of continuous operation. Although the resulting throughput remains within the milligram range, thus being primarily suited for laboratory-scale applications, it exceeds the throughput reported for comparable batch-wise fractionation strategies in the literature27,29,30,38 by roughly two orders of magnitude.
For comparison, batch-based fractionation typically requires at least 20 min per cycle to achieve complete binary separation under quiescent conditions. In addition to the separation time itself, several preparatory and post-processing steps are required: particle collection after separation, reinjection of a fresh suspension into the separation chamber, careful reconnection of pumping tubes to avoid air bubble formation within tubing and the separation cell, and the re-establishment of fully sedimented initial conditions. These auxiliary procedures generally require an additional preparation time of approximately 10 min. Consequently, a single operational cycle, including separation and preparation, takes about 30 min, allowing for only two cycles per hour of operation. Under these conditions, the achievable sample throughput amounts to approximately 0.004 mg h−1. This comparison clearly demonstrates that the continuous fractionation scheme substantially enhances processing efficiency, increasing the achievable sample throughput by a factor of approximately 160 relatives to conventional batch-based methodologies.27,29,30,38
The principle of separation relies on temporarily hovering particles from a lower to a higher wall position via microfluidics and illumination of a photoswitchable surfactant at an appropriate wavelength, easily optimized using UV-vis spectroscopy. For continuous separation, it is essential that the injected particles fully sediment onto the bottom substrate before encountering the obstacle. From the injection point to the separation wall, particles are transported along streamlines according to the local flow velocity at their respective vertical positions within the channel.
To provide a simplified estimate of the required sedimentation time and the corresponding horizontal travel distance (i.e., the minimum required injection channel length), we assume that the microparticles are introduced at the top of the rectangular microfluidic channel (i.e., at h). Furthermore, we assume that the sedimentation velocity is not significantly influenced by the perpendicular laminar flow, since it introduces no turbulence in the system. Accordingly, the description of the sedimentation velocity by a low Reynolds number is rectified. Under this assumption, the buoyancy-corrected gravitational force is balanced by the viscous drag force according to Stokes' law, allowing estimation of the terminal sedimentation velocity. Additionally, the particle will follow the streamlines of the laminar flow inside the microfluidic channel during sedimentation. The motion along the streamlines of the laminar flow is perpendicular to the sedimentation motion. Hence, we can calculate the sedimentation time (tsed = 107.5 s, see section S7 in the SI) independently of motion along the laminar flow, and then calculate the distance traveled along the flow, which is 44 mm. Accordingly, the distance between the injection point and the separation wall should be at least 44 mm or longer (see detailed calculations in the section S7, SI).
Furthermore, precise adjustment of the local illumination position relative to the wall is essential. For accurate positioning of the illumination spot, it is crucial that the particles reach their maximum levitation height at the moment they cross the wall or obstacle. This requirement arises because the levitation process is not instantaneous: particles typically require approximately 3–4 seconds after illumination to reach their fully developed hovering state. Accordingly, the time required for levitation must match the longitudinal travel time of the particles toward the obstacle, such that maximum levitation aligns with the crossing of the obstacle. In our previous study, we mapped the particle velocity field (x–y position) under localized UV illumination on a planar substrate and observed that the maximum levitation height was attained after the particles had traveled a defined distance following UV excitation.36 Consequently, the illumination spot should be positioned at a corresponding upstream distance on the order of 160 μm before the particles encounter the wall. An increase in flow rate extends the longitudinal distance traveled within the same levitation time under identical illumination conditions. Therefore, with increasing flow rate, the distance between the local light spot and the obstacle must be increased proportionally to ensure that maximum levitation is achieved at the point of crossing.
Additionally, we recommend employing the highest possible light intensity in order to generate the strongest transient levitation enhancement. For strong light-induced chemical activity, as required for the temporal hopping of particles over obstacles, the appropriate choice of surfactant concentration is crucial. The total surfactant concentration should be well above the CMC of the trans isomer (CMCtrans = 0.5 mM). This ensures that even under illumination, a sufficient fraction of trans isomers remains in solution, allowing a rapid dynamic exchange between adsorbing trans molecules (with adsorption rate proportional to concentration) and desorbing cis molecules.35 This dynamic exchange sustains a pronounced cis-isomer gradient in the vicinity of the particles and thereby maintains strong light-induced chemical activity. Only above a critical trans-isomer concentration does the interface remain fully covered with trans molecules. At total surfactant concentrations of 2 mM or higher, the interface remains saturated even under UV illumination,47,50 whereas at lower concentrations significant net desorption at water–glass interfaces has been reported.50 At the same time, concentrations exceeding 4 mM should be avoided, as this corresponds to the CMC of the cis isomer (CMCcis = 4.0 mM). In this regime, the cis isomer itself becomes surface-active, which reduces the cis concentration gradient and consequently weakens the chemical activity. Therefore, in the experiments presented in this work, the surfactant concentration was set to 1 mM, representing a regime well above CMCtrans yet below CMCcis, thereby ensuring robust and reproducible light-induced levitation.
Future improvements include tuning wall height to increase selectivity for less active particles and adjusting wall angles to minimize accumulation of non-hovered particles. Overall, the results demonstrate the feasibility of continuous, surface-sensitive particle separation in microfluidic devices and highlight the potential for further optimization for high-purity, high-throughput applications.
To upscale and generalize the continuous separation principle based on selective light-induced hopping of one particle fraction across others, two boundary conditions are critical: (i) a sufficiently high activity contrast between particle fractions, and (ii) at least one fraction exhibiting intrinsically strong chemical activity. In the following, we qualitatively discuss key engineering parameters governing transferability and scale-up of the separation concept, with particular emphasis on surface/material properties, particle concentration, and the choice of photosensitive surfactant.
Surface and material properties
Light-induced levitation of microparticles has been demonstrated for a broad range of interfacial systems, including porous interfaces,27,28 chemically functionalized surfaces (hydrophobic and hydrophilic moieties),27,31 and polymer-modified microparticles.30 For many of these systems, the l-LDDO strength is moderate, resulting in equilibrium levitation heights on the order of 1–2 μm above the bottom substrate. For overcoming macroscopic obstacles (e.g., >100 μm), however, a substantially higher levitation force is required. Since the applied photosurfactant carries a cationic headgroup,51 preferential adsorption is expected at anionic interfaces,46 particularly those with high specific surface area. Consequently, negatively charged and porous materials (e.g., silica-based particles27,28 and activated carbon) are anticipated to exhibit enhanced interfacial surfactant accumulation and thus stronger l-LDDO activity. Similarly, microgels and highly hydrated polymer networks, especially those incorporating anionic functional groups can generate pronounced chemical gradients under illumination.34,52 From a separation engineering perspective, these material classes provide a larger activity contrast window and are therefore particularly suitable for selective fractionation based on light-triggered hopping over obstacles.Particle concentration effects
The hopping mechanism is comparatively robust over several orders of magnitude in particle concentration.36 However, experimental observations indicate that levitation height decreases with increasing particle number density.36 This behavior can be attributed to collective effects such as local depletion of product gradients. As a result, the effective activity contrast between fractions reduces with increasing concentration, rendering selective separation progressively more challenging. The extent of this limitation depends on the natural light induced activity strength of the respective particle fractions. For scale-up considerations, concentration-dependent transport phenomena therefore represent a key parameter.Photosensitive surfactant selection
The choice of photosensitive surfactant constitutes a central control parameter in the separation process. In the present study, an azobenzene-based cationic surfactant was employed, for which photoisomerization kinetics in bulk33 and at interfaces,35,50 as well as adsorption/desorption dynamics at solid–liquid interfaces (with50 and without illumination47), have been extensively characterized in the literature. Reported kinetic data indicate that the relative rates of adsorption/desorption and photoisomerization enable a sustained dynamic exchange between trans- and cis-isomers under continuous illumination. This dynamic steady state generates sufficiently strong and spatially confined chemical gradients at solid–liquid-interfaces to induce active levitation of microparticles in aqueous media. Importantly, the propulsion strength and equilibrium position can be precisely modulated via illumination wavelength28 and intensity,35 providing a high degree of external process control.Variations in hydrophobic chain length and headgroup architecture of the surfactant molecule significantly affect interfacial packing density,53 adsorption equilibria, and desorption kinetics of both isomers, and therefore directly modulate the magnitude of the light-induced chemical activity. While LDDO has been reported for surfactants with different alkyl chain lengths, the strongest effects to date have been observed for the compound presented in Scheme 1. Current literature suggests that cationic headgroups with minimal steric demand promote higher interfacial accumulation compared to bulkier analogues,52,53 thereby enhancing gradient formation.
Nevertheless, the selection of an optimal photosensitive surfactant is inherently application-specific and multidimensional, involving trade-offs between molecular volume change upon isomerization, photostability, quantum yield, adsorption strength, and dynamic exchange potential.53 Among available photoresponsive systems,54–58 azobenzene derivatives remain particularly attractive due to their pronounced conformational change during isomerization and low photodegradation, albeit at moderate quantum yield.59 The latter can be compensated by applying high photon flux (i.e., increased illumination intensity), enabling high conversion rates and sustained gradient formation.27,28 For a comprehensive discussion on material-specific classification of light-induced chemical activity and surfactant selection strategies, the reader is referred to the corresponding perspective article published elsewhere.53
Conclusion
We demonstrated that with the combination of a photoactive surfactant and microfluidics one can separate equally sized microparticles by their differences in their interface, where the principle of separation could run continuously.The principle works by hovering sedimented particles over obstacles/walls much bigger than the size of the particles. To that end particles are induced osmotically/phoretically active, which produces, leads to selective levitation of only strongly active particles. Then, simultaneously, an external fluid flow carries the particles in an upper channel position. Now particles with difference in surface properties likewise porous versus plain particle are hovering differently high and by so, only the porous (∼strong active) can be guided into the higher channel. The plain particles remain effectively non-hovered and are deflected from the wall, where the flow guides them into the lower channel. Such strong levitation requires locally collimated light through the microscope objective, which not only focuses on the applied light intensity but also needs a special wavelength of illumination. Both parameters must increase the initial burst (∼initial photoisomerization kinetics) of adsorbed trans-isomers by that porous particles are temporary so strong active to pass by a wall lift height of 100 um. For azobenzene containing surfactants such strong activities could be achieved with UV light (365 nm).
To predict the wavelength needed for high levitation (∼strong activity) of the particles, this can be estimated from the extinction coefficient of the photo switch, a strait forward value quickly to be obtained from UV-vis spectroscopy. The physical principles result from the photoisomerization rate constant, which is proportional to the extinction coefficient.
Materials and methods
Microfluidic chambers and microparticles
Microfluidic experiments were conducted using commercially available flow chambers (μ-SlideVI, Ibidi GmbH), featuring a glass-bottom coverslip suitable for high-resolution optical microscopy. Each chamber has a void volume of 40 μL.Two types of silica-based colloidal particles were employed in the study: commercial porous silica microparticles with mean diameters of (3 ± 1) μm and (5 ± 1) μm, otherwise unmodified, were obtained from micromod GmbH (sicastar® 43-00-503). These particles possess internal porosity. Non-porous (plain) silica colloids with narrowly distributed diameters of (4 ± 0.1) μm and (5 ± 0.1) μm otherwise unmodified, were sourced from microparticles GmbH (SiO2-F-SC260-2).
Azobenzene containing surfactant
The azobenzene-containing surfactant, C4-Azo-OC6TMAB (azobenzene-functionalized trimethylammonium bromide), was synthesized in accordance with previously reported methodologies. Structurally (Scheme 1), the molecule consists of a cationic trimethylammonium bromide headgroup connected to an azobenzene core via a hexamethylene spacer. The azobenzene unit is further functionalized with a terminal butyl chain, enhancing its amphiphilic character.A stock solution of the surfactant was prepared at a concentration of 10 mM in ultrapure water (Milli-Q, resistivity > 18 MΩ cm) and subsequently diluted to 1 mM for experimental applications involving microparticle dispersions.
The photophysical behavior of the surfactant was characterized via UV-visible absorption spectroscopy. In its thermodynamically stable trans-configuration, the molecule exhibits a π–π* transition centered at 351 nm. Upon photoisomerization to the cis-isomer, the absorption profile changes markedly, showing a blue-shifted π–π* transition at 313 nm and the emergence of an n–π* transition at 437 nm. The cis-isomer is metastable and undergoes thermal relaxation back to the trans-form with a half-life of approximately 48 hours under ambient dark conditions or under continuous red-light irradiation (λ = 625 nm) at 23 °C.33
Sample preparation
Aqueous colloidal dispersions are mixed with a stock solution of photosensitive surfactants to reach a final surfactant concentration of 1 mM. The mixtures are equilibrated for a minimum of 24 hours prior to any measurements to ensure homogeneous distribution and equilibration of the surfactant within the dispersion. For measurements, the surfactant–colloid mixture is introduced into the microfluidic chamber, where gravitational sedimentation leads to the accumulation of particles on the bottom glass surface. The chamber is connected to a syringe pump (PhD Ultra, Harvard Apparatus), enabling controlled fluid manipulation. The entire fluidic system including the chamber, syringe, and connecting tubing is prefilled with the same aqueous solution containing 1 mM of the photosensitive surfactant to maintain chemical consistency throughout the setup. All samples are handled and stored under red light or in the dark to prevent unintended photo-isomerization of the azobenzene-containing surfactant. Measurements are carried out at ambient conditions, with the temperature maintained at T = 23 ± 1 °C.Sample preparation with fluorescein dye
Fluoresceine was used as the fluorescent probe for all emission imaging experiments. Prior to mixing with particle mixture of porous (PSiO2) and plain (SiO2) particles (D = 5 μm, cparticles,total = 0.3 mg mL−1, ratio SiO2/PSiO2 = 2/1), the surfactant solution (c = 1 mM) is mixed with fluorescein (c = 15 μM). Similar as above, the entire fluidic system including the chamber, syringe, and connecting tubing contain the surfactant-dye solution. Details of measurement conditions are shown in SI (section S5).Preparation of modified glass substrate for microfluidic integration
A standard borosilicate microscope glass slide (commercially available) was modified to create a patterned surface suitable for microfluidic chamber. Two parallel layers of transparent pressure-sensitive adhesive tape (Tesa GmbH) were applied to the substrate surface. These tape layers were aligned and trimmed such that the resulting boundary edges formed an angle of ∼40° relative to the anticipated fluid flow direction within the final microfluidic chamber. Selective regions of the adhesive tape, corresponding to the future microchannel area, were carefully scraped using a precision utility blade under light pressure to avoid damaging the underlying glass. This defined the channel bottom substrate of the microfluidic channel. Following the tape removal, residual adhesive in the exposed regions was immediately cleaned using a minimal volume of high-purity methanol. The cleaning was performed with care to minimize solvent interaction with the remaining adhered tape sections, preserving their integrity. The substrate was then left to dry under ambient conditions until complete methanol evaporation was confirmed.Subsequently, the cleaned and patterned glass substrate was aligned and bonded to a commercial microfluidic channel (sticky-Slide VI0.4, Ibidi GmbH), which incorporates a pre-defined chamber geometry. The microfluidic channel dimensions are approximately: length L = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm, width w = 3.8
mm, width w = 3.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm, and height h = 0.54
mm, and height h = 0.54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) mm corresponding to a void volume of approximately 40 μL.
mm corresponding to a void volume of approximately 40 μL.
Zeta potential measurements
Zeta potential is calculated from the electrophoretic mobility measured using commercial zetasizer (Nano-ZS, Malvern Instruments, Ltd., Malvern, UK). Microparticles (cmass = 0.1 mg ml−1) are dispersed in MilliQ or aqueous solution containing the azobenzene surfactant solution (c = 1 mM). Prior to measurements dispersion are sonicated and immediately injected into the measurement cell, an directly measured. Data containing UV illumination is exposed for 12 min under irradiation with UV light (365 nm, I = 10 mW cm−2), followed by sonication for 10 s.Nitrogen sorption analysis
The porous and non-porous commercial stock colloidal suspension was purified by two successive washes with Millipore-grade water to eliminate residual contaminants. The cleaned suspensions were subsequently freeze-dried overnight under high vacuum (<1 mbar) using a Schlenk line. The resulting dry powders and approximately 100 mg were used for N2 sorption.Nitrogen sorption measurements
Prior to analysis, the dried samples were accurately weighed, transferred into analysis cells, and degassed under vacuum at 300 °C for 3 h using a BELPREP VAC III preparation system (Microtrac). Nitrogen adsorption–desorption isotherms were recorded with a BELSORP MAX surface area and porosity analyzer (Microtrac). Depending on the porosity of the samples, measurement times ranged from 18 h (porous particles) to 43 h (non-porous particles). Adsorption data are presented as the adsorbed nitrogen volume plotted against the corresponding relative pressure.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 974
974![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 542 domain elements, 286
542 domain elements, 286![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 916 boundary elements, and 6669 edge elements. (Fig. S6). Flow within the system was modeled under steady-state laminar conditions with a fixed volumetric flow rate of 150 μL min−1, since Reynolds number under study is low, Re ∼100. A no-slip boundary condition was applied to all walls.
916 boundary elements, and 6669 edge elements. (Fig. S6). Flow within the system was modeled under steady-state laminar conditions with a fixed volumetric flow rate of 150 μL min−1, since Reynolds number under study is low, Re ∼100. A no-slip boundary condition was applied to all walls.
The 625 nm wavelength served as image acquisition tool, as it does not induce photo-isomerization of the photosensitive surfactant. In contrast, the secondary wavelength (X nm) was selected based on its ability to induce photo-isomerization. The LED illumination system was configured to enable both global and localized collimation. For global illumination, the light was collimated through the ocular pathway, resulting in uniform exposure across the entire sample area. For localized illumination, the light beam was first shaped using a spatial light modulator (SLM), enabling precise spatial control of the illumination pattern. The modulated beam was then directed through the objective lens to selectively illuminate defined regions of the sample at the focal plane.
This dual-mode collimation approach allowed flexible photoactivation strategies, facilitating both homogeneous and spatially resolved control of light-responsive surfactants. A detailed schematic of the optical setup and beam collimation pathways is provided in Fig. S23 (SI, microscope setup).
After transmission through the sample, all wavelengths below 625 nm were effectively blocked using a 580 nm long-pass optical filter (Thorlabs GmbH), thereby ensuring that only the red light (625 nm) reached the detection system. This optical filtering enabled selective imaging under non-photoactivating conditions, minimizing interference from photoresponsive wavelengths during video acquisition. All LED light sources used in the study were obtained from Thorlabs GmbH, with the following center wavelengths and model numbers: 365 nm (M365LP1), 375 nm (M375L4), 385 nm (M385L3), 395 nm (M395L5), 405 nm (M405L4-C1), 415 nm (M415L4), 430 nm (M430L5), 455 nm (M455L4), 490 nm (M490L4), 505 nm (M505L4-C1), 530 nm (M530L4), and 625 nm (M625L4). Video recordings were performed using a Hamamatsu ORCA-Flash camera at a frame rate of 30 frames per second, capturing dynamics exclusively under red-light illumination.
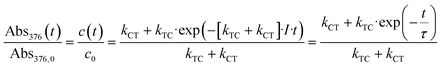 | (11) |
This allows calculation of kTC given by:
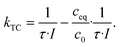 | (12) |
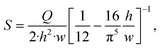 | (13) |
Video tracking
Motion videos were acquired using an inverted Olympus microscope (IX73) equipped with a Hamamatsu ORCA-Flash 4.0 scientific CMOS camera or Olympus MFD 2 – IX53, 73.Depending on the configuration, frame rates were set to 30 fps (ORCA-Flash 4.0) or 15 fps (MFD2-controlled IX53/IX73).
Image acquisition was conducted under red illumination (I = 0.5 mW cm−2) to minimize photochemical disturbances. For illumination, a collimated LED at wavelength λ was directed either from above over the condenser (global illumination) or from below over the objective (illumination) depending on experimental requirements. Prior to each recording session, the aqueous surfactant solution (c = 1 mM) with pre-dispersed microparticles was irradiated at λ for 1 minute at a power of 11.5 mW to ensure attainment of photo-stationary state and equilibrium isomer population. Motion recording was performed by previously sedimented all microparticles onto the bottom glass interface of the rectangular microfluidic channel with 17 mm channel length, 3.8 mm channel depth and 0.54 mm channel height (μ-slideVI with a glass bottom cover slip (Ibidi GmbH)).
A typical 45-second motion-recording sequence captures microparticle trajectories along flow streamlines with the following precisely defined illumination protocol:
1. Initial phase (0–5 s)
A 5-second period without irradiation, allowing observation of baseline particle motion under passive flow conditions.
2. Irradiation phase (5–25 s)
A 20-second interval of illumination at wavelength X (I = 11.5 mW cm−2), activating photoresponsivity behavior while continuously recording particle dynamics.
3. Post-illumination phase (25–45 s)
A final 25-second interval without irradiation, capturing relaxation or persistent effects in the absence of light.
This structured timing ensures a controlled analysis of both the immediate and lasting photo driven responses of the microparticles within the flow system. Each measurement was performed under applied constant pressure driven flow using a syringe pump (Ph.D. ultra, Harvard apparatus) with a flow rate of 150 μL min−1.
Statistical analysis. Statistical analysis is done similar to our previous publication27,28,31 with same set of software Python software package. We calculated the time resolved average velocity U from the sample size n (number of particles) of individual velocity Ui for each frame-to-frame and we averaged using the arithmetic mean to obtain:
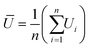 | (14) |
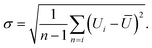 | (15) |
Author contributions
M. B. designed and supervised the project and wrote the manuscript. F. R., D. V. M. performed experiments, analysis, and wrote manuscript together with S. S. C. B., I. M., S. L. and A. S. performed experiments and analysis. N. L. synthesized the photosensitive surfactant. Y. G. performed COMSOL simulations. All the authors were involved in the preparation of the manuscript. All the authors have read and approved the final manuscript.Conflicts of interest
The authors declare no competing financial interests.Data availability
The data supporting this article have been included as part of the SI.Primary data (uploaded Video files) supporting this article have been included as part of the supplementary information (SI) uploaded on publisher website.
Supplementary information is available. See DOI: https://doi.org/10.1039/d5lf00341e.
Acknowledgements
M. B. acknowledges financial support by the National German Science Foundation (DFG) through the grant BE 7745/1-1. A. T. and A. N. acknowledge funding from the University of Potsdam (grant 5317000).References
- X. Yang, F. Yu, H. Shang, Z. Li, S. Wang, Y. Xing and X. Gui, Powder Technol., 2025, 453, 120594 CrossRef CAS.
- Y. Deng, Y. Cai, Z. Sun, J. Lia, C. Liu, J. Weng, W. Li, C. Liu, Y. Wang and D. Zhao, J. Am. Chem. Soc., 2010, 132(24), 8466 CrossRef CAS PubMed.
- M. A. M. Gijs, F. Lacharme and U. Lehmann, Chem. Rev., 2010, 110, 1518 CrossRef CAS PubMed.
- Y. Ding, Y. Yan, H. Wang, X. Wang, T. Hu, S. Tao and G. Li, ACS Appl. Mater. Interfaces, 2018, 10, 48 Search PubMed.
- H. Kim, B. Han, J. Choo and J. Cho, Angew. Chem., Int. Ed., 2008, 47, 10151–10154 CrossRef CAS PubMed.
- H. Liu, X. Liu, W. Li, X. Guo, Y. Wang, G. Wang and D. Zhao, Adv. Energy Mater., 2017, 7, 1700283 CrossRef.
- A. Bielefeld, D. A. Weber and J. Janek, J. Phys. Chem. C, 2018, 123, 1626–1634 CrossRef.
- Y. Wang, M. Zhang, Y. Lai and L. Chi, Nano Today, 2018, 22, 36–61 CrossRef CAS.
- Y. Kim, X. Dong, S. Chae, G. Asghar, S. Choi, B. Jun Kim, J.-Y. Choi and H. K. Yu, Adv. Mater., 2022, 35, 2204775 CrossRef PubMed.
- R. Ahmed, I. Block, F. Otte, C. Günter, A. Duarte-Rodrigues, P. Hesemann, A. Banerji and A. Taubert, Chemistry, 2023, 5, 1124–1137 CrossRef CAS.
- S. Wong, N. Ngadi, I. M. Inuwa and O. Hassan, J. Cleaner Prod., 2018, 175, 361–375 CrossRef CAS.
- I. Block, H. M. Rawel, T. Klamroth, C. Günter, J. Kim, F. Loepthien, S. K. Gahlaut, I. Bald and A. Taubert, ACS Omega, 2025, 10, 4614–4623 CrossRef CAS PubMed.
- D. G. Ghosh Dastidar, S. Saha and M. Chowdhury, Int. J. Pharm., 2018, 548, 34–48 CrossRef CAS PubMed.
- S. Pöttgen, M. Mazurek-Budzyńska and C. Wischke, Int. J. Pharm., 2025, 672, 125340 CrossRef PubMed.
- L. Wu, S. Bai and Y. Sun, Biotechnol. Prog., 2008, 19, 1300–1306 CrossRef PubMed.
- K. J. Stine, Adv. Carbohydr. Chem. Biochem., 2017, 74, 61–136 CrossRef PubMed.
- P. Bayat and P. Rezai, Soft Matter, 2018, 14, 5356 RSC.
- J. Oakey, J. Allely and D. W. M. Marr, Biotechnol. Prog., 2002, 18(6), 1439 CrossRef CAS PubMed.
- M. Yamada, M. Nakashima and M. Seki, Anal. Chem., 2004, 76(18), 5465 CrossRef CAS PubMed.
- J. F. Ashley, C. N. Bowman and R. H. Davis, AIChE J., 2013, 59(9), 3444 CrossRef CAS.
- G. Brans, A. Van Dinther, B. Odum, C. G. P. H. Schroën and R. M. Boom, J. Membr. Sci., 2007, 290(1–2), 230 CrossRef CAS.
- M. Iranmanesh and J. Hulliger, Chem. Soc. Rev., 2017, 46, 5925 RSC.
- A. Dalili, E. Samiei and M. Hoorfar, Analyst, 2019, 144, 87 RSC.
- C. Sophonsiri and E. Morgenroth, Chemosphere, 2004, 55(5), 691 CrossRef CAS PubMed.
- A. Chakra, C. Puijk, G. T. Vladisavljević, C. Cottin-Bizonne, C. Pirat and G. Bolognesi, J. Colloid Interface Sci., 2025, 693, 137577 CrossRef CAS PubMed.
- J. Zheng, J. Chen, Y. Jin, Y. Wen, Y. Mu, C. Wu, Y. Wang, P. Tong, Z. Li, X. Hou and J. Tang, Nature, 2023, 617, 499–506 CrossRef CAS PubMed.
- M. Bekir, M. Sperling, D. Vasquez-Muñoz, C. Braksch, A. Böker, N. Lomadze, M. N. Popescu and S. Santer, Adv. Mater., 2023, 35, 2300358 CrossRef CAS PubMed.
- D. Vasquez Muñoz, F. Rohne, I. Meier, A. Sharma, N. Lomadze, S. Santer and M. Bekir, Small, 2024, 20, 202403546 Search PubMed.
- V. Muraveva, M. Bekir, N. Lomadze, R. Großmann, C. Beta and S. Santer, Interplay of diffusio- and thermo-osmotic flows generated by single light stimulus, Appl. Phys. Lett., 2022, 120, 231905 CrossRef CAS.
- A. Sharma, F. Rohne, D. Vasquez-Muñoz, S.-H. Jung, N. Lomadze, A. Pich, S. Santer and M. Bekir, Small Methods, 2024, 8, 202400226 CrossRef PubMed.
- D. Vasquez Munoz, F. Rohne, I. Meier, C. Braksch, N. Lomadze, A. Heraji Esfahani, A. Nitschke, A. Taubert, S. Santer, M. Hartlieb and M. Bekir, Small Sci., 2024, 4, 2400146 CrossRef CAS PubMed.
- D. Vasquez-Muñoz, M. Nicola Popescu, A. Sharma, F. Rohne, I. Meier, P. Ortner, S. Loebner, J. R. Benson, N. Lomadze, S. Eickelmann, S. Santer and M. Bekir, Small, 2025, 21, 2500012 CrossRef PubMed.
- P. Arya, J. Jelken, N. Lomadze, S. Santer and M. Bekir, J. Chem. Phys., 2020, 152, 024904 CrossRef CAS PubMed.
- A. Sharma, M. Bekir, N. Lomadze and S. Santer, Molecules, 2021, 26, 19 CrossRef CAS PubMed.
- M. Bekir, A. Sharma, M. Umlandt, N. Lomadze and S. Santer, Adv. Mater. Interfaces, 2022, 9, 2102395 CrossRef CAS.
- F. Rohne, D. Vasquez-Muñoz, S. Santer and M. Bekir, J. Chromatogr. A, 2025, 1762, 466368 CrossRef CAS PubMed.
- J. Happel and H. Brenner, Low Reynolds Number Hydrodynamics, Prentice-Hall, Englewood Cliffs, NJ, 1965 Search PubMed.
- H. Brenner, The slow motion of a sphere through a viscous fluid towards a plane surface, Chem. Eng. Sci., 1961, 16, 242–251 CrossRef CAS.
- F. Rohne, D. Vasquez Muñoz, I. Meier, N. Lomadze, S. Santer and M. Bekir, Lab Chip, 2025, 25, 4106–4118 RSC.
- E. Titov, A. Sharma, N. Lomadze, P. Saalfrank, S. Santer and M. Bekir, ChemPhotoChem, 2021, 5, 926 CrossRef CAS.
- P. S. Williams, M. H. Moon, Y. Xu and J. C. Giddings, Chem. Eng. Sci., 1996, 51, 4477–4488 CrossRef CAS.
- P. S. Williams, T. Koch and J. C. Giddings, Chem. Eng. Commun., 1991, 111, 121–147 CrossRef.
- P. S. Williams, L. Seungho and J. C. Giddings, Chem. Eng. Commun., 1994, 130, 143–166 CrossRef CAS.
- I. S. Woo, E. C. Jung and S. Lee, Talanta, 2015, 132, 945–953 CrossRef CAS PubMed.
- A. K. Mathur, C. Agarwal, B. S. Pangtey, A. Singh and B. N. Guptam, Int. J. Cosmet. Sci., 1988, 10, 213–218 CrossRef CAS PubMed.
- J. K. Salem, I. M. El-Nahhal and S. F. Salama, Chem. Phys. Lett., 2019, 730, 445–450 CrossRef CAS.
- M. Umlandt, D. Feldmann, E. Schneck, S. Santer and M. Bekir, Adsorption of Photoresponsive Surfactants at Solid−Liquid Interface, Langmuir, 2020, 36, 14009 CrossRef CAS PubMed.
- F. Rohne, D. Vasquez Muñoz, I. Meier, A. Nitschke, F. Schmitt, N. Lomadze, M. Reifarth, A. Taubert, S. Santer and M. Bekir, Particle-Based Detection of Surface Chemistry via Optical Microscopy—Integrating Microfluidics, Light-Induced Activity of Colloids and Data Science, Small Methods, 2026, 10, e02329 CrossRef CAS PubMed.
- M. Montagna and O. Guskova, Photosensitive Cationic Azobenzene Surfactants: Thermodynamics of Hydration and the Complex Formation With Poly(methacrylic acid), Langmuir, 2018, 34, 311–321 CrossRef CAS PubMed.
- M. Umlandt, P. Ortner, N. Lomadze, M. Bekir, S. Santer and Y. D. Gordievskaya, Langmuir, 2025, 41, 29567–29577 CrossRef CAS PubMed.
- S. Santer, J. Phys. D: Appl. Phys., 2017, 51, 013002 CrossRef.
- M. Bekir, S. Loebner, A. Kobyshev, N. Lomadze and S. Santer, Photosensitive Spherical Polymer Brushes: Light Triggered Process of Particle Repulsion, Processes, 2023, 11, 773 CrossRef CAS.
- M. Bekir, J. Gurke and M. Reifarth, ChemSystemsChem, 2024, 202400026 CrossRef.
- M. Schnurbus, R. A. Campbell, J. Droste, C. Honnigfort, D. Glikman, P. Gutfreund, M. R. Hansen and B. Braunschweig, J. Phys. Chem. B, 2020, 124, 6913 CrossRef CAS PubMed.
- C. Honnigfort, L. Topp, N. García Rey, A. Heuer and B. Braunschweig, J. Am. Chem. Soc., 2022, 144, 4026 CrossRef CAS PubMed.
- M. Reifarth, M. Bekir, A. M. Bapolisi, E. Titov, F. Nußhardt, J. Nowaczyk, D. Grigoriev, A. Sharma, P. Saalfrank, S. Santer, M. Hartlieb and A. Böker, Angew. Chem., Int. Ed., 2022, 61, e202114687 CrossRef CAS PubMed.
- R. Klajn, Chem. Soc. Rev., 2014, 43, 148 RSC.
- D. H. Waldeck, Photoisomerization Dynamics of Stilbenes, Chem. Rev., 1991, 91, 415–436 CrossRef CAS.
- L. W. Giles, C. F. J. Faul and R. F. Tabor, Mater. Adv., 2021, 2, 4152 RSC.
- M. J. Boussinesq, J. Math. Pures Appl., 1868, 13, 377–424 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |