Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
An experimental investigation of the Mpemba effect
Anna
Janni
 *,
Santiago Botero
Ampudia
and
E. Dan
Dahlberg
*,
Santiago Botero
Ampudia
and
E. Dan
Dahlberg
University of Minnesota, Minneapolis, Minnesota, USA. E-mail: janni018@stanford.edu
First published on 16th January 2026
Abstract
The Mpemba effect is not well defined, as it does not distinguish between an initially hotter water sample reaching 0 °C before an initially colder sample or an initially colder sample supercooling and entering its liquid-ice phase transition after an initially hotter sample. These two definitions are explored by measuring the cooling curves of deionized water samples in lidded tins on an expanded polystyrene tray; deionized water in lidded tins on a baking sheet; deionized water in unlidded plastic beakers in an expanded polystyrene tray; tap water in lidded tins on an expanded polystyrene tray; and deionized water in lidded tins in an expanded polystyrene tray. From 176 measurements, 46 and 58 instances of the Mpemba effect as defined in the two aforementioned ways are observed. The temperature measurements align well with Newton's law of cooling, allowing for temperature exponential decay rates to be extracted. It can be concluded that Mpemba effect observations arise from two paths: fluctuations in the convective air currents that dominate the cooling process and variations in supercooling among water samples. The largest temperature differences that exhibited the Mpemba effect were those with tap water, which indicates ions may also be relevant for both interfacial energy transfer and supercooling.
1 Introduction
The Mpemba effect is the claim that hot water begins to freeze before cold water.1–3 While studies assert observations of the Mpemba effect, the counterintuitive phenomenon remains unexplained.2 There was a careful experiment conducted by James Brownridge in a well-controlled environment that showed no Mpemba effect, as expected.4 However, this experiment does not explain the numerous reports of the Mpemba effect.4 Previous research attributes the effect to a number of causes, including faster evaporation rates,2 increased tendencies to supercool,5 and higher levels of dissolved gas2 in hot water compared to cold water, but none of these potential causes have been extensively tested. The importance of developing an understanding of this counterintuitive phenomenon is evident by the number of current areas of research, like quantum simulations and trapped-ion systems, that claim Mpemba-like effects. The question remains: under what conditions does the Mpemba effect occur? In an effort to find the conditions under which the Mpemba effect occurs and explain the frequent observations of it, we systematically measured cooling curves of deionized and tap water samples with different initial temperatures and under different experimental conditions. From our quantitative measurements of the time-dependent temperature, we are able to determine the thermal coupling to the freezer environment using Newton's law of cooling.6–10 Our measurements indicate that previous explanations are not supported and instead point to two reasonable explanations, one for each definition of the Mpemba effect described below. The reasons for the Mpemba effect are the fluctuations in air convection coupling the samples to the freezer environment and the fluctuations in supercooling. Our systematic investigation differs from other studies' that merely compare the time it takes for freezing to occur. Understanding the Mpemba effect is important, as there are many technologies that require air-cooled heat exchanges in their operation.The Mpemba effect has two possible definitions. One is based on the time it takes a water sample to reach 0 °C, and the other is based on the time it takes a water sample to enter the start of its liquid-ice phase transition after supercooling. In our work, we consider both definitions. As water cools, its temperature T decays exponentially in time t according to Newton's law of cooling,
| T(t) = Tf + (T0 − Tf)e−κt | (1) |
In order for the Mpemba effect—as defined by an initially hotter sample reaching 0 °C before an initially colder sample—to occur, the exponential decay rate for a hotter sample must be greater than the exponential decay rate for a colder sample. The rate at which a sample cools depends on how much of its water evaporates,12 whether frost accumulates on a sample container,13 and what thermal contact a container has with its environment.11 As such, we measured water samples with and without lids placed on expanded polystyrene, on a baking sheet, and in wells in an expanded polystyrene tray. In order for the Mpemba effect, as defined as an initially hotter sample beginning its liquid-ice phase transition before an initially colder sample, to occur, a colder sample needs to supercool for a longer time than a hotter sample. Whether and how a sample supercools depends on the presence of impurities, or nucleation sites, in the water sample.14,15 To test this correlation between impurities and supercooling, we measured tap water samples and deionized water samples.
2 Experimental procedure
For most of our measurements, we heated 35 mL water samples to different temperatures (according to the heating methods detailed below) before placing them in our laboratory freezer—an 8.7-cubic-foot Kenmore chest freezer. The laboratory freezer temperature ranged between −14 and −17 °C in its refrigeration cycle. To test whether evaporation causes the Mpemba effect, we measured water samples in 6.35 cm diameter tin cans with and without lids and 1.81 cm diameter plastic beakers without lids (see Fig. 1). To vary the thermal contact with the freezer, we placed samples on an expanded polystyrene tray, in expanded polystyrene wells, or on a metal baking sheet (see Fig. 1). To test whether the presence of impurities in water causes the Mpemba effect, we prepared samples of tap water and deionized water. As samples cooled in the freezer, we measured and recorded their temperatures with thermocouples situated in the center (radially and vertically) of sample containers. Thermocouples measured sample temperatures with an error of ±0.7 °C every 2 seconds.To heat water samples, we filled a beaker with deionized or tap water and set it on a hot plate. Once the water reached a boil, we poured out a sample of 35 mL and then put the beaker back on the hot plate. After we poured out a sample, we waited around 10 minutes to let the previously poured sample cool and then poured another. We continued the process until we had prepared 6 to 8 samples, each at a different temperature. Then we put the samples in the freezer.
We placed samples randomly on the sample holder (baking sheet or expanded polystyrene) and found no correlation between our results and a sample's location. The sample holder was positioned in the center of the freezer elevated to be on the order of 15 cm from the closed lid. The thermocouple wires connected to the meters did not hamper the closing of the freezer lid.
We prepared a portion of tap water samples following a different procedure. We took water directly from the tap and poured eight 35 mL samples. Then, we heated 4 samples to around 80 to 90 °C on a hot plate before placing all samples—4 at room temperature and 4 heated—into the freezer. While deionized water measurements compared samples at a range of initial temperatures, certain tap water measurements compared samples at only two different initial temperatures—room temperature and between 80 to 90 °C.
We measured 44 samples of deionized water in lidded tins on an expanded polystyrene tray in 7 runs of 6 to 8 samples; 34 samples of deionized water in lidded tins on a baking sheet in 5 runs; 18 samples of deionized water in unlidded plastic beakers in an expanded polystyrene tray in 3 runs; 40 samples of tap water in lidded tins on an expanded polystyrene tray in 5 runs; and 40 samples of tap water in lidded cans in an expanded polystyrene holder in 5 runs. In the last case, the expanded polystyrene surrounded the entire tin container, except for the top surface of the tin's lid. From each sample cooling curve, we extracted its cooling rate, the time it took to reach 0 °C, and the time it took to reach 0 °C after possible supercooling.
The extracted cooling rates from the aforementioned measurements showed a range of thermal conductivities and led us to surmise that Mpemba effect observations originated due to variations in convective currents that cooled samples.
3 Discussion of experimental results
When water cools, its temperature follows eqn (1) until around 4 °C, the temperature of water's maximum density.16eqn (1) can be linearized by taking the logarithm of both sides, yielding| ln(T(t) − Tf) = ln(T0 − Tf) − κt. | (2) |
We fit our data between 20 and 4 °C to eqn (2) to determine κ. Fig. 2 shows a typical cooling curve (with some supercooling), and the inset shows its corresponding linearized data between 20 and 4 °C. The slope of the linear fit in Fig. 2 is −κ. Note that all data were similar to those shown in Fig. 2.
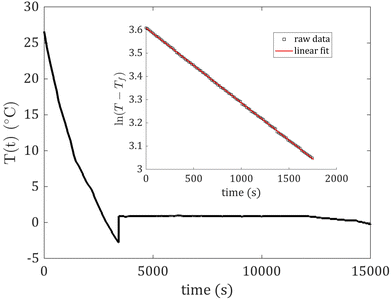 | ||
| Fig. 2 A typical cooling curve (with some supercooling) for deionized water in a lidded tin on expanded polystyrene. The inset shows the corresponding linearized data versus time and the linear fit. | ||
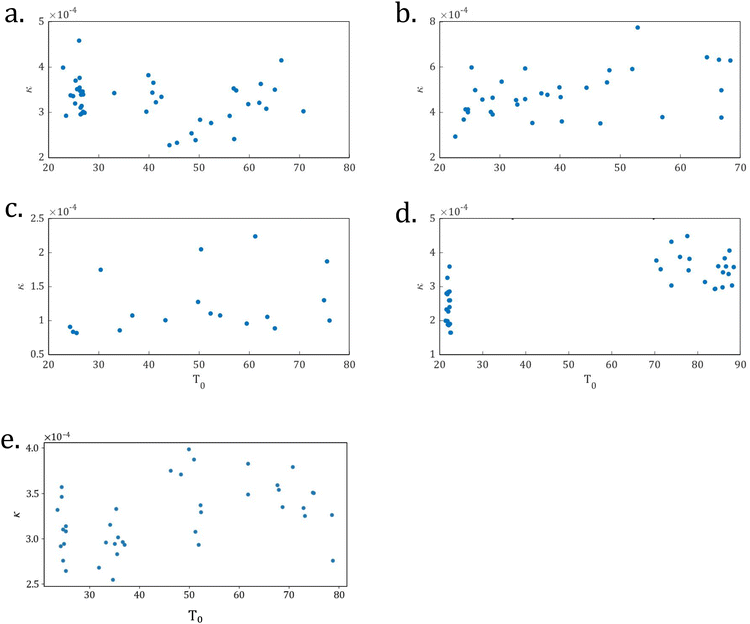
We determined a value of κ for all samples and, as shown in Fig. 3, plotted κ versus initial temperature T0. Note that the errors in the data, found using the covariance matrix from our fits to extract κ, are approximately the same size as the plotted symbols. For the Mpemba effect (defined as an initially hotter sample reaching 0 °C before an initially colder sample) to occur consistently, we would expect a positive correlation between κ and T0. As seen in Fig. 3(a)–(d), there was no such trend. However, as seen in these same figures, there was a significant spread in the measured values of κ. The Mpemba effect (defined as an initially hotter sample reaching 0 °C before an initially colder sample), which we observed, is due to this spread in κ.
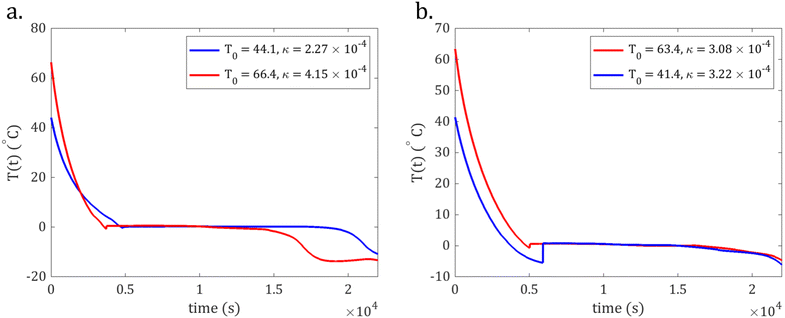
To count the occurrences of the Mpemba effect, we compared the time it took samples to reach 0 °C. If an initially hotter sample of at least 3 °C reached 0 °C in less time than an initially colder or multiple initially colder samples, we counted this as one occurrence. When samples entered their liquid-ice phase transitions, their cooling curves became constant at thermocouple readings between −0.7 and 0.7 °C. Thus, we assigned a ±0.7 °C error to each temperature measurement and only counted Mpemba effects in which a hot sample's initial temperature exceeded a cold sample's initial temperature by 3 °C to account for this error. The initial temperatures generally ranged from 22 to 72 °C in a given trial. We only compared temperature curves from the same trial. Of the 25 trials consisting of 176 samples, we counted 46 instances of the Mpemba effect as defined by an initially hotter sample reaching 0 °C before an initially colder sample or initially colder samples in a given trial. In Table 1, we summarize these results, and Fig. 4(a) shows an example of such an instance.
| Sample type | Mpemba effect count | Number of trials | Number of samples | Mpemba effect frequency |
|---|---|---|---|---|
| Deionized water in lidded tins on expanded polystyrene | 9 | 7 | 44 | 0.205 |
| Deionized water in lidded tins on a baking sheet | 21 | 5 | 34 | 0.618 |
| Deionized water in unlidded plastic in expanded polystyrene | 5 | 3 | 18 | 0.278 |
| Tap water in lidded tins on expanded polystyrene | 7 | 5 | 40 | 0.175 |
| Deionized water in lidded tins in an expanded polystyrene holder | 4 | 5 | 40 | 0.125 |
| Total | 46 | 25 | 176 | 0.261 |
As stated earlier, one definition of the Mpemba effect refers to instances in which an initially hotter sample enters its liquid-ice phase transition before an initially colder sample. Instances of supercooling as well as scatter in κ cause this Mpemba effect to occur. To count this Mpemba effect, we again only compared temperature curves from the same trial. And, if an initially hotter (by at least 3 °C) sample reached its liquid-ice phase transition in less time than an initially colder or multiple initially colder samples, we counted this as one effect. We counted 58 instances of this Mpemba effect. In Table 2, we summarize these results, and Fig. 4(a) and (b) show examples of such instances.
| Sample type | Mpemba effect count | Number of trials | Number of samples | Mpemba effect frequency |
|---|---|---|---|---|
| Deionized water in lidded tins on expanded polystyrene | 17 | 7 | 44 | 0.386 |
| Deionized water in lidded tins on a baking sheet | 19 | 5 | 34 | 0.559 |
| Deionized water in unlidded plastic in expanded polystyrene | 5 | 3 | 18 | 0.278 |
| Tap water in lidded tins on expanded polystyrene | 13 | 5 | 40 | 0.325 |
| Deionized water in lidded tins in an expanded polystyrene holder | 4 | 5 | 40 | 0.125 |
| Total | 58 | 25 | 176 | 0.330 |
To determine whether the Mpemba effect occurred only when samples had close initial temperatures or when samples had significant temperature differences, we plotted the number of Mpemba effect counts in bins of initial 5 °C temperature differences. We did this for both types of the Mpemba effect (see Fig. 5 and 6). As previously mentioned, some of the tap water data compared samples initially at two different temperatures (around room temperature and between 80 and 90 °C) not initially at a range of different temperatures. This explains the separation of tap water counts (in purple) in Fig. 5 and 6.
As previously mentioned, in Newton's law of cooling, κ equals hA/mc, where c is a sample's specific heat capacity, m is its mass, h is its coefficient of heat transfer, and A is its surface area.11 From water sample type to water sample type, we expect κ to have slight variations. For example, a water sample placed in or on an expanded polystyrene tray is expected to have a different heat transfer coefficient h than a sample placed on a metal baking sheet. We saw this trend in our data. Samples in the expanded polystyrene wells had an average value of κ = 1.2 × 10−4, which is less than the average value of κ = 4.8 × 10–4 for samples in tins on a baking sheet.
While we expect κ to vary from sample type to sample type, we do not expect it to vary for samples of the same type—especially for lidded samples on expanded polystyrene where evaporation and frost effects are eliminated. So, why do we see a spread in κ for this data set and others? In our experiment, the overall heat transfer coefficient h from the water to the freezer air can be modeled by
 | (3) |
The standard deviation of κ is a measure of its variation. When we compared the standard deviations of the κ's for a given type of sample, we found that the highest Mpemba effect frequency, 0.618 for deionized water in lidded tins on a baking sheet (see Table 1), corresponds to the highest standard deviation in κ, σ = 1.0 × 10−4. And, the lowest Mpemba effect frequency—0.125 for tap water in lidded cans in the expanded polystyrene wells (see Table 1)—corresponds to the smallest standard deviation of κ, σ = 3.6 × 10−5. We attribute this small variation in σ to be due to the expanded polystyrene wells limiting convective air currents. Accordingly, these samples saw fewer occurrences of the Mpemba effect. This leads to the conclusion that the Mpemba effect corresponds to a spread in κ caused by convective currents.
To further connect convection to the Mpemba effect, we calculate the Rayleigh number for sample water and freezer air to show that convection, rather than conduction, dominates the cooling process of water samples. For air in the freezer at a pressure of 1 atm and average temperature between −14 and −17 °C, we calculate Ra ∼ 107. The critical Rayleigh value above which convection occurs is on the order of 103, indicating that convection dominates the heat transfer process from sample to freezer.18 This convection process contributes to the overall cooling rate of samples quantified by κ.
There has been a body of research attempting to understand convection currents that includes experiments in air,19 liquids,20 atmosphere,21 and, additionally, theory associated with plasmas19 and weather.22 However, given the complexity of convection, there is not a simple method to generally quantify fluctuations in air currents.
4 Supercooling
Of the 176 samples we measured, we saw 84 samples supercool. Initially hot and initially cold water samples supercooled across three of the four sample types we measured. Table 3 lists supercooling counts and frequencies for each sample type we measured. On occasion, an initially hotter water sample supercooled for a longer time than an initially colder sample, and this contributed to instances of the Mpemba effect counted in Table 2. However, across all sample types, hot water did not consistently supercool for a longer time or to a cooler temperature than initially colder water, and thus, supercooling did not enable the Mpemba effect to consistently occur.| Sample type | Supercooling count | Number of samples | Supercooling frequency |
|---|---|---|---|
| Deionized water in lidded tins on expanded polystyrene | 18 | 44 | 0.409 |
| Deionized water in lidded tins on a baking sheet | 32 | 34 | 0.941 |
| Deionized water in unlidded plastic in expanded polystyrene | 0 | 18 | 0.000 |
| Tap water in lidded tins on expanded polystyrene | 34 | 40 | 0.85 |
| Deionized water in lidded tins in an expanded polystyrene holder | 0 | 40 | 0.000 |
| Total | 84 | 176 | 0.500 |
While we observed multiple instances of supercooling, we did not identify the cause. However, trends in our data aligned with existing research. Water samples placed in expanded polystyrene wells did not supercool while 94 % of water samples in tins on a baking sheet did supercool. The former samples cooled at a slower rate than other samples, and the latter cooled at a faster rate than other samples. These results align with prior research that posits a connection between the rate at which water cools to 0 °C and whether and for how long that water supercools.23,24
While this describes the overwhelming trend we observed in supercooling, we also saw that deionized water in lidded tins on a baking sheet had the highest rate of supercooling; 95% of these samples supercooled. An increased presence of nucleation sites decreases the likelihood that a sample supercools.25 In water, ions act as nucleation sites, making deionized water more likely to supercool than tap water, which has more impurities. That said, the overall rate of supercooling in tap water was 85% as opposed to 37% for deionized water. This counterintuitive result likely indicates that other variables, like cooling rate to 0 °C, more heavily influenced supercooling tendencies, or that the thermocouple in a sample acted as a nucleation site.
5 Conclusion
We attribute common experimental observations of the Mpemba effect to two causes: a variation in supercooling between samples and a spread in the thermal contact a sample makes with its cooling agent due to a variation in thermal convection currents. We posit that the latter contributes to the cause of Mpemba effect observations seen by Vynnycky, Ball, Mpemba, Ibekwe, Osborne, Tang, and Burridge,1–3,13,26–28 and the former contributes to the cause of Mpemba effect observations seen by Brownridge, Tan, and Burridge.4,5,28Recent research attributes the Mpemba effect to faster evaporation rates,2 increased tendencies to supercool,5 and higher levels of dissolved gas2 in hot water compared to cold water. If faster evaporation rates in hot water compared to cold water cause the Mpemba effect, we would expect a higher frequency of Mpemba effect occurrences in unlidded measurements than in lidded measurements; we saw the opposite (see Tables 1 and 2). If increased tendencies to supercool in hot water compared to cold water cause the Mpemba effect, we could expect to count significantly more instances of the Mpemba effect that accounts for supercooling than of the Mpemba effect that ignores it. Out of 176 measurements, we only counted 12 more instances of the Mpemba effect that accounts for supercooling than of the effect that ignores it (see Tables 1 and 2). In our experiment, supercooling contributes to the Mpemba effect but alone does not explain it. If higher levels of dissolved gas in hot water compared to cold water cause the Mpemba effect, we would expect to see a higher frequency of Mpemba effect occurrences in tap water than in deionized water; we saw the opposite (see Tables 1 and 2).
We saw the highest frequency of the Mpemba effect for data with the largest spread in κ values, and the lowest frequency of the Mpemba effect for data with the smallest spread in κ values. We attribute the spread in the κ values to uncontrolled convective currents in the freezer. This conclusion aligns with past research on the Mpemba effect conducted by Brownridge and with current research on turbulent convection conducted by Lohse and Shishkina.4,29 When Brownridge measured samples in a vacuum chamber heated and cooled through radiation (that is, without convective currents), of 138 measurements, no Mpemba effect occurred.4 But what happens when convective currents are controlled in a freezer, not in vacuum? We attempted to address this question by isolating sample tin walls from convective currents by placing the tins in an expanded polystyrene holder. When we did, we saw the smallest spread in κ and the lowest frequency of Mpemba effect counts. We attribute the remaining spread in κ and corresponding counts of the Mpemba effect to the exposure of each sample's lid top to convective currents in the freezer. In their studies of thermal convection, Lohse and Shishkina realize the difficulty of controlling convective currents due to convection's sensitivity to the geometry, boundary conditions, and turbulent heat flow in a system.29 We predict that had each sample's lid been covered by expanded polystyrene and isolated from a freezer's convective currents, there would have been smaller spread in κ and no counts of the Mpemba effect. We suggest groups with the relevant expertise perform experiments to investigate controlled convection and its connection to the Mpemba effect.
Lastly, we note the samples with the largest spread between the initial and final temperatures are those with tap water. This suggests ions in the water play a significant role. In the case of the Mpemba effect due to fluctuations in κ, it is most likely the energy transfer at the interface that is responsible. The mechanism for this is unclear and could be related to the greater mass of the ions or the fact that they are charged. As to the supercooled events, it is known that ions suppress the nucleation process of ice formation.30 For both Mpemba effects, since the concentration of ions is low, the fluctuations would be expected to be large.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available within the article and from the corresponding author upon reasonable request.Acknowledgements
This work was supported by the National Science Foundation (NSF) (Grant No. DMR 2103704) and U.S. Department of Energy, Office of Basic Energy Sciences, Division of Materials Science and Engineering, under Award No. DE-SC0013599.Notes and references
- M. Vynnycky and S. Kimura, Int. J. Heat Mass Transfer, 2014, 80, 243–255 CrossRef.
- P. Ball, Phys. World, 2006, 19, 19–21 CrossRef CAS.
- E. Mpemba and D. G. Osborne, Phys. Educ., 1979, 14, 410 CrossRef.
- J. D. Brownridge, Am. J. Phys., 2011, 79, 78–84 CrossRef CAS.
- K. C. Tan, W. Ho, J. I. Katz and S. J. Feng, Am. J. Phys., 2016, 84, 293–300 CrossRef CAS.
- A. Zhang, G. Xia and C. Wu, Nat. Commun., 2025, 16, 301 CrossRef PubMed.
- L. K. Joshi, J. Franke, A. Rath, F. Ares, S. Murciano, F. Kranzl, R. Blatt, P. Zoller and B. Vermersch, Phys. Rev. Lett., 2024, 133, 010402 Search PubMed.
- A. Nava and R. Egger, Phys. Rev. Lett., 2024, 133, 136302 Search PubMed.
- K. Zatsarynna, A. Nava, R. Egger and A. Zazunov, Phys. Rev. B, 2025, 111, 104506 Search PubMed.
- T. Vu and H. Hayakawa, Phys. Rev. Lett., 2025, 134, 107101 Search PubMed.
- P. Nithiarasu, R. W. Lewis and K. N. Seetharamu, in Fundamentals of the Finite Element Method for Heat and Fluid Flow, John Wiley & Sons, Ltd, Hoboken, New Jersey, 2004, ch. 4, p. 151 Search PubMed.
- G. S. Kell, Am. J. Phys., 1969, 37, 564–565 CrossRef CAS.
- A. Osborne, New Sci., 1969, 43, 662 Search PubMed.
- K. Zachariassen and E. Kristiansen, Cryobiology, 2000, 41, 257–259 CrossRef CAS PubMed.
- P. G. Debenedetti and H. E. Stanley, Phys. Today, 2003, 56, 40–46 Search PubMed.
- R. M. Lynden-Bell, S. C. Morris, J. D. Barrow, J. L. Finney and C. Harper, in Water and Life—The Unique Properties of H2O, CRC Press, Boca Raton, Florida, 1st edn, 2010, p. 78 Search PubMed.
- J. H. Lienhard, in A Heat Transfer Textbook, Dover Publications, Mineola, New York, 5th edn, 2019, pp. 78–85 Search PubMed.
- J. Kunecs, in Dimensionless Physical Quantities in Science and Engineering, Thermomechanics, Elsevier, Waltham, Massachusetts, 2012, ch. 5, pp. 173–283 Search PubMed.
- K. Talluru, H. Pan, J. Patternson and K. Chauhan, Int. J. Heat Fluid Flow, 2020, 84, 108590 Search PubMed.
- Y.-H. He and K.-Q. Xia, Phys. Rev. Lett., 2019, 122, 014503 Search PubMed.
- M. Karasewicz, M. Waclawczyk, P. Ortiz-Amezcua, L. Janicka, P. Poczta, C. K. Borges and I. S. Stachlewska, Atmos. Chem. Phys., 2024, 24, 13231–13251 CrossRef CAS.
- P. Bechtold, N. Semane, P. Lopez, J. Chaboureau, A. Beljaars and N. Bormann, J. Atmos. Sci., 2014, 71, 734–753 CrossRef.
- N. R. Gokhale, J. Atmos. Sci., 1965, 22, 212–216 Search PubMed.
- A. Gholaminejad and R. Hosseini, J. Electron. Cool. Therm. Control, 2013, 3, 1–6 Search PubMed.
- S.-L. Chen and C.-L. Chen, Inorg. Chem., 1999, 38, 339–351 Search PubMed.
- R. T. Ibekwe and J. P. Cullerne, Phys. Educ., 2016, 51, 78–84 Search PubMed.
- Z. Tang, W. Huang, Y. Zhang, Y. Liu and L. Zhao, InfoMat, 2023, 5, e12352 Search PubMed.
- H. C. Burridge and O. Hallstadius, Proc. R. Soc. A, 2020, 1–14 Search PubMed.
- D. Lohse and O. Shishkina, Phys. Today, 2023, 76, 26–32 CrossRef.
- H. R. Pruppacher and M. Neiburger, J. Atmos. Sci., 1963, 20, 376–385 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |