Open Access Article
Open Access ArticleMicrofluidic determination of minimum miscibility pressure (MMP) in dynamic CO2/n-decane flow
Junyi
Yang
and
Peichun Amy
Tsai
 *
*
Department of Mechanical Engineering, University of Alberta, Edmonton, Alberta, Canada T6G 2R3. E-mail: peichun.amy.tsai@ualberta.ca
First published on 18th March 2026
Abstract
Carbon dioxide enhanced oil recovery (CO2-EOR) has been recognized as a viable pathway for carbon capture, utilization, and storage (CCUS). Among its variants, miscible CO2-EOR offers a considerable additional oil recovery of approximately 5–20%, making the determination of minimum miscibility pressure (MMP) a critical design consideration. In this study, we employ a high-pressure microfluidic platform to investigate the miscibility transition between CO2 and n-decane at temperatures (T) of 40, 50, 70, and 90 °C. At T = 40 °C, with increasing pressure (P), microfluidic visualization reveals a series of distinct flow regimes: dripping, quasi-steady jetting, unsteady jetting, transitional, and ultimately diffusive regimes. In the diffusive regime, miscibility is achieved through intensive mixing, leading to the disappearance of the fluid–fluid interface. Based on these microfluidic observations, we propose a new criterion for MMP determination: the minimum pressure required to reach the diffusive regime for the dynamic CO2–oil flow. The experimentally determined MMP values show good agreement with previous microfluidic studies and predictions from the Peng–Robinson equation of state (PR-EOS). Furthermore, the MMP increases linearly with temperature from 40 to 90 °C, consistent with the reduced solubility of CO2 in n-decane at higher temperatures. This microfluidic method provides a rapid and visual approach to assess miscibility transitions in CO2-EOR applications.
1. Introduction
Carbon dioxide enhanced oil recovery (CO2-EOR) has been widely implemented as a large-scale and cost-effective method under carbon capture, utilization, and storage (CCUS) strategies to mitigate rising greenhouse gas emissions. In CO2-EOR, captured CO2 is injected into existing petroleum reservoirs to enhance oil recovery—a process that not only enables low-cost production but also reduces the net carbon footprint of the extracted oil.1–4 Since the 1990s, the number of CO2-EOR projects has steadily increased and is projected to continue growing through 2040.5 Various CO2-EOR approaches exist, including immiscible injection, miscible injection, CO2 foam-assisted EOR, and the huff-n-puff method.4,6–8 Compared with immiscible CO2-EOR, miscible and near-miscible operations have shown particular promise,9–12 as they are more effective in improving sweep efficiency and the overall recovery factor.11 Miscibility refers to the ability of two fluids to mix uniformly and form a single-phase mixture.13 In CO2-EOR, achieving miscibility between injected CO2 and in situ oil can significantly improve oil recovery through mechanisms such as oil swelling and viscosity reduction,9,14,15 vaporizing and condensing gas drive processes,16,17 and the mitigation of unfavorable flow phenomena including gravity override18–21 and viscous fingering,22–28 which commonly reduce recovery efficiency in conventional gas injection methods.29In miscible CO2-EOR, the minimum miscibility pressure (MMP) is one of the most critical parameters, defining the pressure above which CO2 and oil become fully miscible.13 MMP is influenced by several factors, such as temperature, oil composition, and injected gas composition.30 Two commonly used definitions for miscibility are first-contact and multi-contact miscibility.13,30,31 First-contact miscibility refers to a thermodynamic condition in which two fluids form a single-phase mixture upon contact, regardless of their mixing ratio.13 In contrast, multi-contact miscibility is more commonly encountered in practical CO2-EOR operations, as reservoir oils typically contain multiple components, including light, intermediate, and heavy fractions.32,33 Multi-contact miscibility is achieved through repeated contact and component exchange between the injected gas and reservoir oil, involving both vaporization and condensation mechanisms.13,31
Several long-established, macroscopic experimental techniques have been developed since the 1980s to determine MMP, including the vanishing interfacial tension (VIT) method,34,35 rising bubble apparatus (RBA),36 and slim tube test (STT).32,37 The VIT method involves measuring interfacial tension, typically using a pendant drop technique, to construct an IFT–pressure curve and extrapolate the pressure at which IFT approaches zero, which is taken as the MMP.34,35 However, this method is sensitive to fluid composition and may result in deviations in MMP estimation.31,38 The RBA involves injecting a gas bubble into a column of oil and observing its shape and rising behavior, which offers qualitative insight into miscibility conditions.36 While relatively simple and fast to perform, RBA may not reliably capture the vaporizing and condensing mechanisms involved in multi-contact miscibility, due to the short interaction time and lack of continuous phase exchange between gas and oil.31,39 The STT uses a coiled tube packed with porous media, such as sand or glass beads, to simulate reservoir flow conditions.32,37 CO2 is injected at varying pressures, and oil recovery is measured; MMP is operationally identified as the pressure at which the recovery factor transitions from a rapid increase to a slower rate of change, which typically occurs at a recovery of ≈90–95%.13,32,37 The STT is widely considered the most accurate laboratory method for representing multi-contact miscibility in CO2-EOR.30,31,33 However, it is time-consuming (often requiring 2–6 weeks to complete), lacks standardized design in tube dimensions and packing material, and can be costly to perform.30,31,33 In addition to the above-mentioned experimental techniques, MMP can also be estimated using empirical correlations40–42 and equation-of-state- (EOS-) based methods.43,44
In contrast, more recent microfluidic approaches offer complementary capabilities, including reduced sample volumes, faster measurements, and direct visualization at the pore scale. Microfluidic platforms, hence, have recently emerged as versatile tools for studying fluid displacement,12,25,45–54 mass transport,55–66 and phase behavior67–72 at the pore scale. When operated under high-pressure conditions, these systems can mimic the temperature and pressure environments of subsurface reservoirs, making them particularly useful for investigating CO2–oil MMP.73–82
Microfluidic systems, when combined with fluorescence microscopy, allow direct visualization of phase behavior at the CO2–oil interface, with the oil phase typically labeled using a fluorescent dye. For example, Nguyen et al.73 were among the first to examine MMP using a microfluidic T-junction, where CO2 and oil were co-injected, and miscibility was evaluated by monitoring the fluorescence intensity at a fixed detection region.73 Below MMP, the presence of segmented CO2 bubbles led to periodic drops in fluorescence intensity; above MMP, a more uniform signal indicated complete mixing.73 Microfluidic analogs of the vanishing interface technique (VIT) have also been developed, where oil is trapped in a dead-end microchannel and the average fluorescence intensity is monitored to determine whether a homogeneous mixture is formed.74–78 Similarly, the “slim-tube-on-a-chip” design mimics the concept of conventional slim-tube tests by injecting CO2 into oil-saturated microfluidic porous media, where multi-contact miscibility is evaluated based on the recovery factor.79–82 Unlike conventional slim tubes, microfluidic platforms enable faster testing (within hours instead of weeks) and allow precise control over pore size, geometry, and permeability, offering more consistent and tunable experimental conditions.
Compared to conventional laboratory methods, microfluidic approaches offer several remarkable advantages for studying miscibility. These include low fluid consumption, real-time optical access, rapid testing, and precise control over the porous medium geometry and flow conditions.83–87 Microfluidic platforms are particularly well suited for studying miscibility because their characteristic length scale, typically ranging from 100 nm to 100 μm,88 is comparable to the pore scale of subsurface porous media.84 At this scale, the reduced channel dimensions result in a high surface-to-volume ratio, which enhances interfacial contact and promotes mass transfer,89 thereby making microfluidic systems more representative of multi-contact miscibility development in porous reservoirs. Moreover, recent microfluidic studies have extended into the nanofluidic regime, where channel depths on the order of 10 to 100 nm introduce strong molecular–wall interactions and confinement effects.75,78 The measurement of MMP under such nanoconfinement is particularly relevant for CO2-EOR in tight formations such as shale. For example, parachor-EOS modeling based on interfacial tension predictions suggests that reducing the channel depth from 1 μm to 10 nm can lower the MMP of CO2–octane from 15.10 to 10.10 MPa at 160 °C.78
Building on the advantages of microfluidics, this work introduces a new, dynamic, and visually intuitive method for determining MMP based on flow regime transitions in continuous flow. While previous microfluidic approaches using static geometries such as dead-end channels have provided valuable insights into equilibrium phase behavior,74–76,78 our work introduces an original approach and a unique perspective that captures the transient evolution of miscibility under dynamic, flowing conditions. We develop a high-pressure microfluidic platform capable of operating under reservoir-like conditions (15 MPa, 90 °C) and use it to visualize interfacial dynamics in CO2–n-decane systems across a range of temperatures and pressures. By identifying the transition from dripping, jetting, and transitional flows to a diffusive regime, we define MMP through direct visualization of miscibility with high precision. This approach enables real-time assessment of phase behavior and provides new insight into the dynamics of interfacial deformation and mixing near the miscibility threshold. The experimental MMP values are compared with predictions from the Peng–Robinson equation of state (PR-EOS) and previous microfluidic experimental data, confirming the method's accuracy, reproducibility, and potential for broader applications in energy and chemical systems.
2. Experiments
This study investigates the transition to miscibility between CO2 and n-decane from 40 to 90 °C, where miscibility typically occurs at pressures on the order of 1 to 101 MPa. A robust microfluidic setup was developed to operate with current gas supply up to 15 MPa and 90 °C, enabling operation under reservoir-relevant conditions.2.1. Materials
The materials used included n-decane (≥99%, CAS No. 124-18-5, Thermo Fisher Scientific), Nile red dye (CAS No. 7385-67-3, Sigma-Aldrich), and CO2 (99.99% purity, 4.0 anaerobic grade, Linde). For microfabrication, prime-grade silicon (Si) wafers and round borofloat glass substrates, each with a diameter of 100 mm, were used.2.2. Microfluidic chip fabrication
The microfluidic chip was fabricated using silicon and glass, materials commonly employed in high-pressure microfluidics.90,91 On the front side, the silicon wafer was first cleaned with a piranha solution and primed with hexamethyldisilazane (HMDS) vapor to enhance photoresist adhesion (YES Oven). A layer of photoresist (AZ 1512) was spin-coated and patterned using direct-write photolithography with a maskless aligner (Heidelberg MLA150). Deep reactive ion etching (DRIE) was then performed using a high-rate single-stage Bosch recipe (Oxford Estrelas) to etch the microchannels to a depth of 50 μm.Following the two-sided lithography method adapted from Qi et al.,91 the fluidic ports were etched through the remaining 450 μm-thick wafer from the backside using a bilayer of AZ4330 photoresist. This double-sided approach was adopted to eliminate the need for mechanical drilling, which previously caused silicon cracking under elevated pressure.
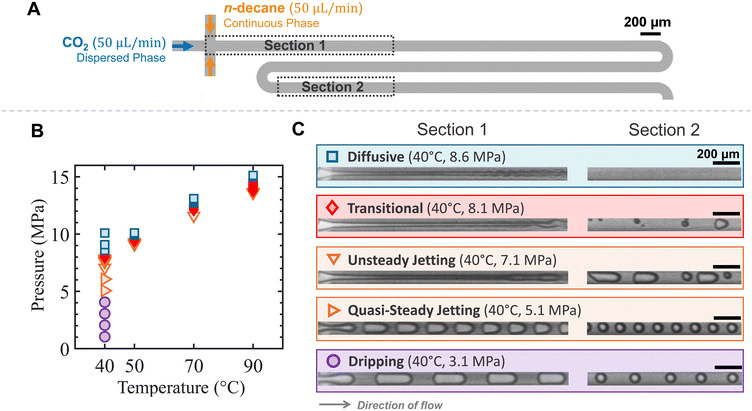
Residual photoresist was removed with a photoresist asher (PVA TePla Ion 10Q) followed by piranha cleaning. The patterned silicon wafer was then anodically bonded to a borofloat glass wafer at 370 °C with 600 V using an AML wafer bonder, forming a sealed chip. Finally, the bonded wafer stack was diced into individual chips using a dicing saw (Disco 3240). An image of the final silicon–glass microfluidic chip used in the experiments is shown in Fig. 1A, with overall chip dimensions of 25 × 15 mm. A microscopy view of the microfluidic design is shown in Fig. 1B, illustrating the CO2–decane flow within the serpentine microchannel.
2.3. Experimental setup
A stainless steel microfluidic chip holder was designed to securely house the microfluidic chip, as shown in Fig. 1C. The chip holder was fabricated using a CNC mill (Tormach PCNC 1100) and a milling–drilling machine (Optimum BF 30 Vario). Internal fluidic channels were precision-drilled to the interface with external 1/16″ stainless steel tubing. The chip was compressed and secured with four bolts, which also clamped a sapphire viewing window between the chip and the holder for real-time microscopy observations. The modular holder design enables replacement of the microfluidic chip with alternative layouts for other high-pressure experiments.As illustrated in Fig. 1C, a high-pressure syringe pump (Teledyne ISCO 100DX) was used for CO2 injection and pressure control. A pressure transducer (OMEGA PX309) was installed upstream of the CO2 microfluidic inlet to provide accurate monitoring of the CO2 inlet pressure.
For n-decane injection, a high-pressure syringe pump (Chemyx Fusion 6000X) was used with a 6 mL stainless steel syringe and an inline pressure gauge to verify that the target pressure was reached. Downstream pressure was regulated using a back pressure regulator (Equilibar ZF Series) with an electronic pilot controller (Equilibar EPR-3000), which maintained a stable back-pressure setpoint.
Temperature control was achieved using a temperature controller (OMEGA CN740). An aluminum heating plate was mounted on the stainless steel chip holder and secured with Kapton tape. A K-type thermocouple was inserted into a hole drilled 1 mm away from the microfluidic channel to provide accurate temperature readings. Thermal paste was applied beneath the heating plate and inside the thermocouple hole to ensure efficient thermal conductivity.
2.4. Experimental procedures
We investigated the miscibility transition for CO2 and n-decane at temperatures of T = 40, 50, 70, and 90 °C. For experiments at 40 °C, an initial coarse pressure ramp was performed from 1 to 10 MPa in 1 MPa increments to observe flow behavior across a broad pressure range. Subsequently, a finer pressure ramp was performed from 7.5 to 8.5 MPa in 0.1 MPa increments to determine MMP more precisely. For temperatures of 50, 70, and 90 °C, only the fine pressure ramp (0.1 MPa increments) was performed to precisely determine the MMP values. For each pressure and temperature condition, the experiment was repeated at least three times to ensure reproducibility.To enhance contrast between the oil and CO2 phases, n-decane was mixed with Nile red at a concentration of 10 g m−3. The solution was stirred with a magnetic stirrer until fully homogenized, and then degassed in a vacuum desiccator for 45 minutes until no visible gas bubbles remained.
Prior to each experiment, the target temperature and back pressure were set using the temperature controller and back pressure regulator, respectively. CO2 was gradually pressurized to the desired testing pressure using a high-pressure syringe pump and introduced into the microfluidic system. Simultaneously, the oil phase (n-decane) was also pressurized to the same target pressure with its valve kept closed. Once both pressure and temperature reached their targets, the decane valve was opened, and both CO2 and decane were co-injected into the microfluidic chip. Both injection pumps were set to maintain a constant flow rate of 50 μL min−1 throughout the experiment. The system was allowed to stabilize for at least 5 minutes before recording began.
After the experiments were completed at a given pressure, both CO2 and n-decane lines were closed. The back pressure was then adjusted for the next setpoint, and each fluid was re-pressurized separately. Once stabilized at the new pressure, the fluids were reintroduced at the same flow rate of ![[Q with combining dot above]](https://www.rsc.org/images/entities/i_char_0051_0307.gif) = 50 μL min−1.
= 50 μL min−1.
2.5. Imaging acquisition and data analysis
The microfluidic chip holder was placed on an inverted microscope with 5× or 10× objectives. A mercury lamp (HXP 200C) was used for illumination. A high-speed camera was attached to the microscope side port to capture image sequences at 7500 frames per second (fps).Initial image processing was conducted in ImageJ,92 including cropping the region of interest (ROI), applying a bandpass filter, and subtracting the background to improve illumination uniformity and reduce noise. Data processing, including equation of state calculations and figure generation, was carried out using MATLAB.
3. Results and discussion
3.1. Microfluidic flow regimes and visualization
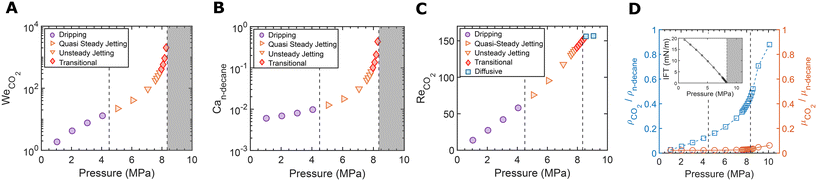
We performed experiments by simultaneously injecting CO2 and n-decane at a constant flow rate of 50 μL min−1 into a microfluidic flow-focusing device, where CO2 was the dispersed phase and n-decane was the continuous phase. A schematic of the microchannel layout is shown in Fig. 2A.At T = 40 °C, increasing the pressure from 1 to 10 MPa resulted in a series of distinct flow regime transitions, as illustrated by the data points in Fig. 2B and the corresponding snapshots in Fig. 2C. The observed flow regimes include dripping, quasi-steady jetting, unsteady jetting, transitional, and diffusive regimes. A detailed discussion of each regime is provided in sections 3.1.1 to 3.1.4.
For T = 50, 70, and 90 °C, finer pressure steps of 0.1 MPa were applied over a narrower pressure range to capture the miscibility transition. As shown in Fig. 2B, similar flow regime transitions were observed, from unsteady jetting to transitional, and ultimately to diffusive regimes.
The transition from dripping to jetting can be triggered when either inertial forces from the dispersed phase or viscous shear from the continuous phase become large enough to overcome interfacial tension.95,97 Each mechanism is represented by a corresponding dimensionless number:
(1) Inertia-surface tension balance: when the Weber number of the dispersed phase (WeCO2) exceeds a critical value, the inertial force of the moving CO2 thread overcomes the restoring force of surface tension, leading to the transition from dripping to jetting.
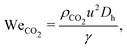 | (1) |
![[Q with combining dot above]](https://www.rsc.org/images/entities/i_char_0051_0307.gif) /A is the superficial velocity (i.e., the volumetric flow rate
/A is the superficial velocity (i.e., the volumetric flow rate ![[Q with combining dot above]](https://www.rsc.org/images/entities/i_char_0051_0307.gif) divided by the microchannel cross-sectional area A), Dh = 2HW/(H + W) is the hydraulic diameter, and γ is the interfacial tension (IFT) between CO2 and n-decane, estimated using the empirical model developed by Pan and Trusler.94
divided by the microchannel cross-sectional area A), Dh = 2HW/(H + W) is the hydraulic diameter, and γ is the interfacial tension (IFT) between CO2 and n-decane, estimated using the empirical model developed by Pan and Trusler.94
(2) Viscous-surface tension balance: when the capillary number of the continuous phase (Can-decane) exceeds a critical value, the viscous shear exerted by the flowing n-decane balances and overcomes surface tension, also triggering the transition to jetting.
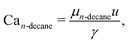 | (2) |
Both WeCO2 and Can-decane increase with pressure (Fig. 3A and B). The increase in We is particularly pronounced during the transition from dripping to jetting, reflecting the growing inertia of the inner CO2 stream as its density increases with pressure due to compression.
 | ||
| Fig. 3 Variation of dimensionless numbers and fluid properties with pressure at T = 40 °C. (A) Weber number of the CO2 phase, calculated using eqn (1), showing its pressure dependence. (B) Capillary number of the n-decane phase, defined by eqn (2), as a function of pressure. (C) Reynolds number of the CO2 phase, defined by eqn (3), as a function of pressure. (D) Pressure-dependent variation of fluid properties: density ratio ρCO2/ρn-decane (blue □, left axis),93 viscosity ratio μCO2/μn-decane (red ○, right axis),93 and IFT (inset, calculated using the empirical correlation from ref. 94 based on pendant drop measurements). The shaded region marks the pressure range where IFT approaches zero; We and Ca are not computed in this regime. | ||
The change in flow behavior can be further explained by the pressure-dependent variation in fluid properties, illustrated in Fig. 3D. Between 1 and 5 MPa, the CO2 density increases steadily, while the interfacial tension with decane decreases, as seen in the inset of Fig. 3D. This combined effect of higher density and lower interfacial tension causes the Weber number to increase, due to increasing inertial force and weaker surface tension. The steady rise in the Weber number promotes the transition from dripping to quasi-steady jetting.
In this pressure range, the Weber number continues to rise due to increasing CO2 density and decreasing interfacial tension. The capillary number also increases more significantly as interfacial tension drops further, as seen in Fig. 3B. The increasing trend in We and Ca indicates an increasing influence of inertial and viscous forces relative to surface tension, leading to delayed droplet pinch-off. The Reynolds number of the CO2 stream, defined as
| ReCO2 = ρCO2uDh/μCO2, | (3) |
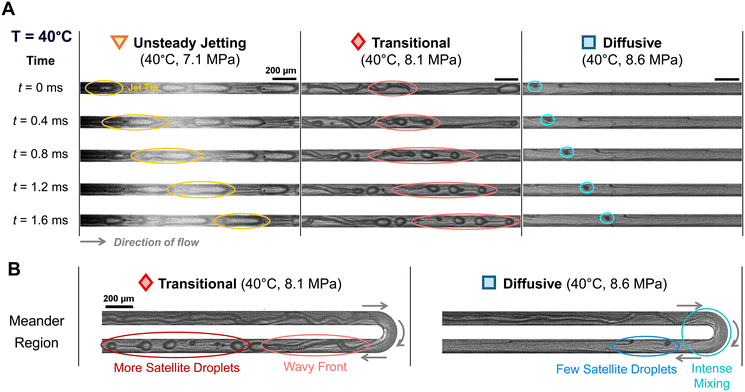
To better visualize the jet-tip dynamics at elevated pressures, time-lapse snapshots were captured at 40 °C and 7.1 MPa to highlight the instability at the jet tip. As shown in Fig. 4A (left), frames acquired every 0.4 ms reveal a weak and unstable jet tip, with inefficient pinch-off and variation in droplet size.
Similar unsteady jetting behavior was consistently observed at 50, 70, and 90 °C (Fig. 5–7 (left)). At elevated temperatures, the onset of unsteady jetting occurred at correspondingly higher pressures, consistent with reduced interfacial tension and changes in fluid properties.
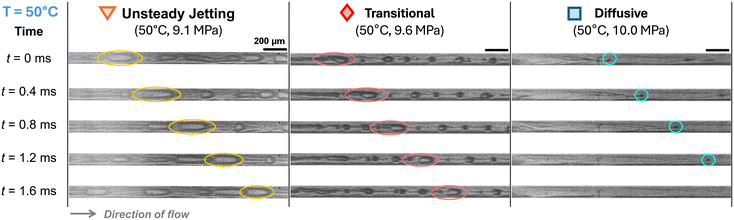 | ||
| Fig. 5 Time-lapse snapshots at T = 50 °C showing the evolution of the CO2 jet tip at 9.1 MPa (unsteady jetting), 9.6 MPa (transitional), and 10.0 MPa (diffusive). Scale bars: 200 μm. | ||
 | ||
| Fig. 6 Time-lapse snapshots at T = 70 °C showing the evolution of the CO2 jet tip at 11.6 MPa (unsteady jetting), 12.5 MPa (transitional), and 12.8 MPa (diffusive). Scale bars: 200 μm. | ||
In the pressure range corresponding to transitional flow, the inset in Fig. 3D shows IFT values of approximately 1 to 2 mN m−1, a range commonly regarded as near-miscible in EOR applications.98 Based on both the IFT values and observed flow characteristics, this regime is interpreted as near-miscible.
Transitional flow was also observed at elevated temperatures of 50, 70, and 90 °C. Compared to the transitional regime at 40 °C, droplets at higher temperatures (50–90 °C) generally appear smaller, although no consistent size trend is evident due to the unstable nature of the flow. As T increased, the CO2 jet also appeared thinner and more laminar, with breakup occurring further downstream – sometimes even near the channel exit. Although the flow pattern appears different from that at 40 °C, it is still considered transitional, since a visible interface was present and discrete droplets eventually formed, consistent with low-IFT conditions.
These changes with increasing temperature can be explained by variations in fluid properties. Under the pressure and temperature conditions corresponding to the snapshots in Fig. 4A (middle) and 7 (middle), the viscosity of decane decreased by 37.5%, while the density of CO2 increased by 24.1%. The reduction in viscous drag from the outer decane stream, combined with the increased inertial force from the inner CO2 stream, led to a more extended CO2 jet and a delayed breakup point further downstream.
The time-lapse images at 40 °C and 8.5 MPa captured the mixing evolution, as shown in Fig. 4B (right). As the CO2 jet passed through the meandering region, streak-like patterns emerged and the jet appeared stretched and filamented. This pattern suggested intensive mixing with only a few satellite droplets. Further downstream, the interface disappeared entirely, suggesting that the two fluids transitioned into a homogeneous phase.
The miscibility was not achieved immediately upon contact near the junction. This observation is consistent with the findings of Cubaud and Notaro,99 where miscible threads exhibited different flow behaviors depending on the Péclet number (Pe), defined as:
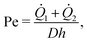 | (4) |
![[Q with combining dot above]](https://www.rsc.org/images/entities/i_char_0051_0307.gif) 1 and
1 and ![[Q with combining dot above]](https://www.rsc.org/images/entities/i_char_0051_0307.gif) 2 are the flow rates of the two fluids, D is the diffusivity, and h is the channel height.99 The Péclet number represents the ratio of advective to diffusive transport. In our study, Pe ≈ 6 × 103 at 40 °C, which exceeds the ultra-diffusive threshold of PeD = 1.5 × 103 reported by Cubaud and Notaro.99 The higher Pe suggests that the diffusive rate was insufficient to produce an ultra-diffusive interface immediately at the junction. Instead, we observed a gradual development of miscibility, characterized by a transient mixing zone that evolved as the fluids flowed downstream.
2 are the flow rates of the two fluids, D is the diffusivity, and h is the channel height.99 The Péclet number represents the ratio of advective to diffusive transport. In our study, Pe ≈ 6 × 103 at 40 °C, which exceeds the ultra-diffusive threshold of PeD = 1.5 × 103 reported by Cubaud and Notaro.99 The higher Pe suggests that the diffusive rate was insufficient to produce an ultra-diffusive interface immediately at the junction. Instead, we observed a gradual development of miscibility, characterized by a transient mixing zone that evolved as the fluids flowed downstream.
At 50 and 70 °C, a similar diffusive flow was observed. At 90 °C, the mixing dynamics became even more pronounced. As shown in Fig. 7 (right), chaotic advection led to tangled streak patterns and rapid interface breakdown.
In this study, the onset of the diffusive regime is used to define the minimum miscibility pressure (MMP). Microfluidic visualization shows that mixing does not begin immediately after the flow-focusing junction due to limited diffusion. A diffusive thread gradually forms downstream, with a blurred interface and intensified mixing enhanced by recirculation in the meandering channel. The appearance of this flow structure marks the transition to a single phase and provides a clear visual indicator of miscibility.
In addition, we conducted a supplementary grayscale intensity analysis at section 2 to compare the unsteady, transitional, and diffusive regimes (Fig. 8). Fig. 8B presents the grayscale profiles extracted from the corresponding experimental snapshots in Fig. 8A. Grayscale values were normalized to the range of 0–1, averaged along the vertical direction, and plotted against distance along the flow direction. As shown in Fig. 8B, sharp drops in intensity indicate the locations of the dark CO2–oil interfaces—pronounced in the unsteady jetting regime, less frequent in the transitional regime, and absent in the diffusive regime, where the grayscale profile becomes flat, resulting in a more uniform gray color. Fig. 8C shows the histogram analysis of the same ROI. Compared with the unsteady jetting and transitional regimes, the probability of darker intensities (approximately 0.2–0.4) drops by orders of magnitude in the diffusive regime. This change reflects the disappearance of distinct interfaces as the pressure exceeds the MMP. Both methods—direct visualization of the diffusive regimes and grayscale intensity analysis—yield consistent MMP results. While MMP values in this work can be conveniently identified through direct visualization, the quantitative grayscale intensity analysis (e.g., Fig. 8B and C) also serves as an alternative approach.
To evaluate whether the visual criterion for miscibility is influenced by flow conditions, we conducted additional experiments at 40 °C using different combinations of CO2 and n-decane flow rates, ranging from 25 to 100 μL min−1, at pressures between 8 and 9 MPa. While varying the flow rate altered specific aspects of the two-phase flow morphology—including the droplet size, breakup location, and thread width—the pressure at which the flow transitioned from transitional to fully diffusive regimes remained unchanged. For example, in the diffusive regime, reducing the CO2 flow rate from 100 μL min−1 to 25 μL min−1 decreased the initial thread width from 60.5 μm to 36.0 μm and changed the interface curvature near the inlet. These results are consistent with previous observations of Re and Pe dependence of the viscous thread morphology of miscible flows in a flow-focusing microchannel.99
The agreement of the regime transition pressure for different flow rates confirms that full miscibility is a thermodynamic effect, driven by a vanishing interfacial tension at the MMP, rather than by hydrodynamic effects. Therefore, the fixed flow rate of 50 μL min−1 used in the main experiments provides a valid basis for determining MMP, while further studies could explore flow-rate effects, particularly changes in the flow morphology, under other regimes such as dripping or jetting.
3.2. Minimum miscibility pressure (MMP)
We compared the minimum miscibility pressure (MMP) determined from our microfluidic experiments with predictions from a thermodynamic model based on the Peng–Robinson equation of state (PR-EOS).100 The PR-EOS is widely used in the petroleum industry for modeling the phase behavior and thermophysical properties of CO2–hydrocarbon mixtures, offering a practical balance between simplicity and accuracy.33,101,102 The following section provides a brief overview and computational workflow of the PR-EOS approach, while detailed equations and implementation steps are presented in Appendix A.PR-EOS is a thermodynamic model using a cubic equation of state that describes the pressure–volume–temperature (P–V–T) relationships for real fluids, accounting for intermolecular forces and molecular volume.100,102,105 It defines pressure (P) as a function of molar volume (v) and temperature (T):100
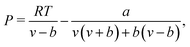 | (5) |
In our study, PR-EOS was used to compute the pressure–composition (P–x) phase envelope for the CO2–decane system. The envelope is bounded by two curves: the bubble point curve (open ○ in Fig. 9A), constructed by varying the CO2 mole fraction in the liquid phase (x1), which marks the pressure at which the first bubble of vapor forms from a liquid mixture, and the dew point curve (filled ◆ in Fig. 9A), constructed by varying the CO2 mole fraction in the vapor phase (y1), which marks the pressure at which the first drop of liquid condenses from a vapor mixture.13 These two curves are computed by satisfying vapor–liquid equilibrium,13 which ensures that there is no net mass transfer between the phases, i.e., thermodynamic equilibrium is achieved.106
 | ||
| Fig. 9 Prediction of the minimum miscibility pressure (MMP) using the Peng–Robinson equation of state (PR-EOS). (A) Phase envelopes of CO2/n-decane mixtures at T = 40, 50, 70, and 90 °C, computed using PR-EOS. The phase envelope consists of the bubble point curve (open ○), which plots the bubble point pressure Pbubble against the CO2 mole fraction in the liquid phase (x1), and the dew point curve (filled ◆), which plots the dew point pressure Pdew against the CO2 mole fraction in the vapor phase (y1). MMP is determined at the highest point of P on the phase envelope, above which the two components form a fully miscible, single-phase mixture at all proportions. (B) Flow chart illustrating the bubble point calculation procedure. CO2 is treated as component 1 and n-decane as component 2. For each specific CO2 liquid mole fraction x1, the corresponding bubble point pressure Pbubble and vapor mole fraction y1 are obtained using a Newton–Raphson method based on PR-EOS with the van der Waals mixing rule. The iteration converges when the fugacity residual between liquid and vapor phases, ‖F = fL − fV‖, falls below a tolerance of 10−6, satisfying vapor–liquid equilibrium (VLE). By the same token, dew point pressures (Pdew) are calculated analogously by fixing y1 and solving for x1 and Pdew. Each converged solution contributes to the construction of the phase envelopes in Fig. 9A. | ||
Together, the bubble point and dew point curves form the phase envelope. In Fig. 9A, they appear as a single continuous boundary (liquid or vapor) enclosing the two-phase region, where vapor and liquid coexist. Outside this region, only a single phase exists. The peak of the envelope corresponds to the pressure above which the mixture becomes single-phase at all compositions, also known as the first-contact minimum miscibility pressure (FC-MMP).31
The workflow in Fig. 9B outlines the bubble point calculation procedure. Following common convention, we define x1 and x2 as the mole fractions of CO2 (component 1) and n-decane (component 2) in the liquid phase, respectively, such that x1 + x2 = 1. Similarly, y1 and y2 denote the mole fractions of CO2 and n-decane in the vapor phase. The subscript L refers to the liquid phase and V to the vapor phase. For a binary mixture of CO2 and n-decane under isothermal conditions, we fix the temperature T and x1, and solve for two unknowns: the bubble point pressure Pbubble and y1. These parameters must satisfy vapor–liquid equilibrium (VLE), where the fugacity of each component is equal in both phases: f1,L = f1,V and f2,L = f2,V.
Under the PR-EOS formulation, fugacity depends nonlinearly on pressure and composition through the compressibility factor Z and other mixture-specific EOS parameters.100 As a result, the VLE conditions form a coupled system of two nonlinear equations in two unknowns: Pbubble and y1. To enforce these conditions numerically, we define a residual vector F = fL − fV, and solve the resulting nonlinear system using the Newton–Raphson method. Convergence is reached when the residual norm ‖F‖ falls below 10−6, indicating that thermodynamic equilibrium has been achieved within an acceptable tolerance. Initial guesses for Pbubble and y1 are estimated using the Wilson correlation107 for better convergence. Additional computational details are provided in Appendix A. Each converged solution yields a point on the phase envelope shown in Fig. 9A.
By repeating this calculation over a range of compositions, we traced the full phase envelopes at each temperature. The resulting envelopes for T = 40, 50, 70, and 90 °C are shown in Fig. 9A. As the temperature increases, the envelopes shift toward higher pressures, with the corresponding MMP increasing from 8.26 MPa at 40 °C to 9.73 MPa at 50 °C, 12.88 MPa at 70 °C, and 15.43 MPa at 90 °C. The CO2–decane phase envelopes determined by PR-EOS in this study are benchmarked against the experimental data from Jiménez-Gallegos et al.103 and EOS models reported by Nascimento et al.104 and Pan and Trusler94 at 40 and 70 °C, showing good agreement as presented in Fig. 11 in Appendix B.
 | ||
Fig. 10 Comparison of minimum miscibility pressure (MMP) determined from our microfluidic experiments using dynamic flow ( ) and Peng–Robinson EOS predictions ( ) and Peng–Robinson EOS predictions ( ) at T = 40, 50, 70, and 90 °C. Literature data using dead-end microfluidic channels to determine CO2–decane MMP from Bao et al.75 ( ) at T = 40, 50, 70, and 90 °C. Literature data using dead-end microfluidic channels to determine CO2–decane MMP from Bao et al.75 ( ), Shi et al.76 ( ), Shi et al.76 (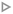 ), Pan et al.77 ( ), Pan et al.77 ( ) and Tao et al.78 ( ) and Tao et al.78 ( ). Error bars represent the scatter across three independent experimental datasets, calculated as 0.5 × (MAX − MIN). A linear regression of the experimental data is shown as a dashed line, given by MMP (MPa) = 0.136 × T (°C) + 2.976, with a coefficient of determination R2 = 0.997. ). Error bars represent the scatter across three independent experimental datasets, calculated as 0.5 × (MAX − MIN). A linear regression of the experimental data is shown as a dashed line, given by MMP (MPa) = 0.136 × T (°C) + 2.976, with a coefficient of determination R2 = 0.997. | ||
 | ||
| Fig. 11 Benchmarking of PR-EOS-predicted phase envelopes in this work for the CO2–n-decane system against experimental data from Jiménez-Gallegos et al.103 at 71.6 °C, and PR-EOS models from Pan and Trusler94 and Nascimento et al.104 at 40 and 70.9 °C. The present model shows good agreement across the full two-phase region. | ||
Within the tested temperature range (T = 40–90 °C), MMP increases nearly linearly with temperature. This strong correlation from microfluidic measurement aligns with previous studies identifying temperature as a dominant factor affecting MMP, based on sensitivity analysis.42
The observed increase in MMP with temperature reflects the reduced solubility of CO2 in n-decane at elevated temperatures.108 Since CO2 dissolution is an exothermic process, adding thermal energy shifts the equilibrium against solubility, thereby requiring higher pressure to achieve single-phase conditions.6,108 Our observed slope of MMP increasing with temperature (ΔMMP/ΔT = 0.136 MPa °C−1) is comparable to that reported for light crude oils in VIT-based MMP studies, where ΔMMP/ΔT = 0.1448 MPa °C−1.109 As noted by Lashkarbolooki et al.,109 this slope may increase for a heavier crude oil.
A recent micro/nanofluidic study by Tao et al.78 explored a wider temperature range (50–180 °C) and reported a non-monotonic trend in MMP. Based on thermodynamic modeling, the authors found that for a 1 μm deep channel, the MMP increases with temperature up to 140 °C, and then begins to decrease beyond this transitional point.78 This behavior was attributed to changes in the CO2–oil phase equilibrium: at lower T, MMP increases due to decreasing CO2 solubility, while at higher T, the oil phase exhibits gas-like behavior that enhances mass transfer, reduces interfacial tension, and lowers the pressure required for miscibility.78 Additionally, the transitional temperature decreased from 140 °C to 110 °C as the channel depth was reduced from 1 μm to 10 nm, highlighting the pronounced effect of nanoconfinement.78
The close agreement between microfluidic MMP measurements and PR-EOS predictions may be attributed to enhanced mixing and mass transfer at the fluid interface. The small length scale of the microfluidic platform, along with the meandering channel geometry, promotes passive mixing and increases interfacial contact between CO2 and decane. The resulting rapid and continuous component exchange likely allows the local composition to approach near-equilibrium, leading to flow regime transitions that closely match PR-EOS predictions, despite the system being dynamically operated.
Nguyen et al.73 pioneeringly introduced a microfluidic flow-based method for quantifying MMP in microfluidics, using fluorescence intensity profiles in a T-junction geometry to monitor composition changes.73 In contrast, our approach determines MMP by identifying hydrodynamic flow regime transitions—from transitional to diffusive flow—in a flow-focusing geometry. This strategy eliminates the need for fluorescent tracers, enables real-time assessment with standard optical imaging, and directly captures miscibility-driven morphological changes. Furthermore, our method spans a broader temperature range (40–90 °C) and is validated against Peng–Robinson EOS predictions, demonstrating applicability under diverse thermodynamic conditions.
While the applicability of this approach to more complex fluid systems, such as CO2 with multi-component crude oil, remains to be investigated, the microfluidic method provides valuable and unique insight into the transient evolution of fluid interfaces, including interface formation, droplet breakup, and mixing behavior, which cannot be captured by thermodynamic EOS models. In contrast to EOS-based predictions, which require careful model selection and parameter tuning,31,102 microfluidic visualization provides a direct visual indicator for miscibility transitions under reservoir-relevant conditions.
4. Conclusions
In this study, we developed and utilized a high-pressure microfluidic platform with a flow-focusing junction to investigate the miscibility transition between CO2 and a model light oil, n-decane, under dynamic co-flow conditions at temperatures of 40, 50, 70, and 90 °C. At 40 °C, five distinct flow regimes were observed as the pressure increased from 1 to 10 MPa: dripping, quasi-steady jetting, unsteady jetting, transitional, and diffusive regimes. At higher temperatures (50, 70, and 90 °C), we focused on the latter three regimes, where similar transitions were observed with increasing pressure. The flow regime transitions were driven by the increasing dominance of inertial and viscous forces over surface tension, arising from pressure-dependent changes in CO2 properties. Specifically, increasing pressure led to higher density and viscosity of CO2, along with a reduction in interfacial tension between CO2 and decane.The minimum pressure at which the diffusive regime first appeared was used as the criterion for determining the MMP experimentally. The diffusive regime was characterized by intense mixing and the disappearance of a sharp interface downstream, indicating fully miscible conditions. Miscibility was not achieved immediately at the flow-focusing junction but developed progressively along the microchannel, enhanced by mixing induced in the meander region.
The experimentally determined MMP values showed good agreement with Peng–Robinson EOS predictions with deviations less than 2.25%. Over the tested range of 40 to 90 °C, MMP increased linearly with temperature, consistent with the decreasing solubility of CO2 in decane at higher temperatures.
These findings highlight the potential of microfluidic flow visualization as a reliable and rapid method for identifying miscibility transitions. In addition to MMP determination, the transitional regime observed just below the diffusive regime provides further insight into near-miscible conditions. Within this pressure range, the low IFT values are likely sufficient to suppress capillary trapping and enhance sweep efficiency. In CO2-EOR applications where full miscibility cannot be achieved due to operational or reservoir constraints, operating near the transitional regime could still enable effective oil displacement.
A Peng–Robinson EOS-based prediction of minimum miscibility pressure (MMP)
This appendix provides the thermodynamic formulation and solution algorithm used to calculate the phase envelope of the CO2–n-decane system using the Peng–Robinson equation of state (PR-EOS).100The overall solution algorithm follows the steps illustrated in Fig. 9B. For each temperature T, we construct the bubble point curve (open ○ in Fig. 9A) by incrementally varying the CO2 mole fraction in the liquid phase (x1). At each x1, the goal is to determine the corresponding bubble point pressure (Pbubble) and CO2 mole fraction in the vapor phase (y1) that satisfy vapor–liquid equilibrium (VLE) conditions for both components. This requires solving the Peng–Robinson EOS to compute fugacities in both phases and enforcing fi,L = fi,V for i = 1 and 2, resulting in a nonlinear system with two equations and two unknowns. The system of nonlinear equations is solved using the Newton–Raphson method.
A1 Input properties and initial guess
We begin by specifying fluid properties for CO2 (component 1) and n-decane (component 2). For each component, the critical temperature Tc,i, critical pressure Pc,i, and acentric factor ωi are sourced from the DIPPR 801 database.110 The Wilson correlation107 provides an initial estimate of the vapor mole fraction (y1) and bubble point pressure (Pbubble) for improved convergence in the Newton–Raphson iteration.A2 Peng–Robinson EOS parameters
For implementation, the cubic form of the PR-EOS is used, where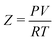 is the compressibility factor, reflecting how much a real fluid deviates from ideal gas behavior:100
is the compressibility factor, reflecting how much a real fluid deviates from ideal gas behavior:100| Z3 − (1 − B)Z2 + (A − 3B2 − 2B)Z − (AB − B2 − B3) = 0. | (6) |
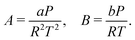 | (7) |
A3 Pure component properties
To compute A and B, the mixture parameters a and b must first be calculated from pure-component properties. For each component i in the mixture, the pure-component parameters ai and bi are defined by:100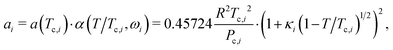 | (8) |
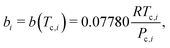 | (9) |
| κi = 0.37464 + 1.54226ωi − 0.26992ωi2. | (10) |
A4 van der Waals mixing rule
The mixture parameters a and b are calculated based on the pure component parameters (ai, bi), the component mole fractions (xi, xj), and the intermolecular interactions (aij). These are evaluated using the classical van der Waals mixing rules:100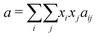 | (11) |
| aij = (1 − kij)ai1/2aj1/2, | (12) |
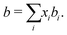 | (13) |
With the mixture parameters a and b determined, the constants A and B are computed using eqn (7). These values are then used to solve the cubic equation in eqn (6) for the compressibility factor Z. In the two-phase region, eqn (6) yields three real roots: the smallest root corresponds to the liquid phase compressibility ZL, and the largest to the vapor phase compressibility ZV.100
A5 Fugacity and vapor–liquid equilibrium
The liquid and vapor compressibility factors (ZL and ZV) are used to determine the fugacity of each component (k = 1, 2) in the liquid and vapor phases, respectively.100 Fugacity is the effective partial pressure of a component in a real mixture, given by:100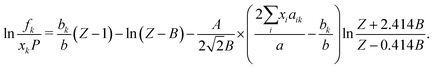 | (14) |
At vapor–liquid equilibrium, the fugacity of each component must be equal in both phases.100 To maintain consistency with the rest of the appendix, we use i to represent component indices in the following discussion:100
| fi,L = fi,V for component i = 1, 2. | (15) |
We define the fugacity residual vector:
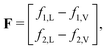 | (16) |
A6 Newton–Raphson method
The Newton–Raphson method is used to solve the nonlinear system of equations F = 0, where F is defined in eqn (16). The variable vector is X = [y1, Pbubble]T, and an initial guess is provided using the Wilson correlation107 to improve convergence. The residual vector F is evaluated at each iteration, and the solution is updated iteratively using the Newton–Raphson scheme:111| Xn+1 = Xn − J−1F(Xn), | (17) |
Finally, the MMP is identified as the highest pressure on the phase envelope, above which the mixture becomes single-phase at all compositions.
B Benchmark of PR-EOS phase envelopes
This appendix presents a benchmark of the calculated phase envelopes using the PR-EOS formulation described in Appendix A. Model predictions for the CO2–n-decane system are compared against experimental data103 and previously published results94,104 at 40 °C and approximately 70 °C. Data at 70.9 °C and 71.6 °C were used as the closest available references.94,103 This comparison confirms that the present implementation (shown in solid lines) captures the two-phase behavior with high accuracy across the full range of conditions. Following successful benchmarking, we extended the PR-EOS calculations to the temperature ranges of interest that have not been previously reported in the literature.C Temperature-dependent variation of dimensionless parameters at MMP
Fig. 12 illustrates the variation of dimensionless parameters at the minimum miscibility pressure (MMP) across different temperatures, indicated by different colors. The vertical dashed lines mark the MMP for each temperature, and the corresponding horizontal lines denote the value of each dimensionless parameter at that transition point.In Fig. 12A, the density ratio ρCO2/ρn-decane at MMP shows no clear trend with temperature and remains approximately 0.5. In Fig. 12B, the viscosity ratio μCO2/μn-decane at MMP increases with temperature. In Fig. 12C, despite changes in pressure and temperature, the Reynolds number of the CO2 stream at MMP remains close to 150 for different temperatures. Based on the definition of the Reynolds number and the fixed flow velocity u and characteristic length L in our setup, this nearly constant value of Re suggests that the kinematic viscosity of CO2 (μ/ρ) at the miscibility transition changes very little with temperatures.
D Grayscale intensity analysis of flow regimes
We performed a grayscale intensity analysis for one set of experiments at 40 °C, covering three characteristic flow regimes: unsteady jetting (7.1 MPa), transitional (8.4 MPa), and diffusive (8.5 MPa) regimes. Grayscale profiles were extracted from both section 1 and section 2 of the microfluidic channel using ImageJ. The selected regions of interest (ROI) correspond directly to the snapshots shown in Fig. 13. At each x-location along the flow direction, grayscale values were averaged across the channel width (vertical direction) and then normalized between 0 and 1 for comparison. In these profiles, smaller grayscale values correspond to darker regions in the snapshot, such as CO2–oil interfaces, which appear as valleys in the curve.As shown in Fig. 13, the section 1 profiles close to the initial flow junction were similar for all three regimes. In section 2, however, the unsteady jetting case exhibited large oscillations and multiple local minima due to distinct CO2 bubble boundaries appearing dark, the transitional regime showed fewer fluctuations from smaller, less frequent bubbles, and the diffusive regime displayed an almost flat profile, indicating the absence of visible interfaces.
Author contributions
Junyi Yang: investigation, methodology, software, formal analysis, writing – original draft, visualization. Peichun Amy Tsai: conceptualization, methodology, validation, software, formal analysis, writing – revision, visualization, supervision, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information: Video S1. Experimental video clips showing the complete set of flow regimes at T = 40 °C. See DOI: https://doi.org/10.1039/D5LC00616C.The data supporting this article have been included as part of the SI.
Acknowledgements
We gratefully acknowledge the support from the Canada First Research Excellence Fund (CFREF), the Future Energy System (FES T02-P05 CCUS projects) at the University of Alberta, and the Canada Foundation for Innovation (CFI 34546). J. Y. acknowledges the support of the Graduate Student Scholarship from Alberta Innovates (AI-GSS) and the Future Energy System Legacy Builders Program (FES T02-P05-LBP1). P. A. T. holds a Canada Research Chair (CRC) in Fluids and Interfaces and gratefully acknowledges funding from the Natural Sciences and Engineering Research Council of Canada (NSERC) and Alberta Innovates (AI) (ADVANCE 202102818), in particular the NSERC Canada Research Chairs Program (CRC 233147) and Discovery Grant (RGPIN-2020-05511). We thank the nanoFAB facility at the University of Alberta for support with microfabrication, particularly Aaron Hryciw for process consultation, and Breanna Cherkawski, Gustavo de Oliveira Luiz, and Scott Munro for training and technical assistance. We also acknowledge the Elko Engineering Garage at the University of Alberta for support with microfluidic chip holder fabrication, and thank Nicolas Marchand for training and assistance.Notes and references
- International Energy Agency (IEA), Can CO2-EOR really provide carbon-negative oil?, 2019, https://www.iea.org/commentaries/can-co2-eor-really-provide-carbon-negative-oil, accessed: 2025-01-01 Search PubMed.
- J. H. Ching, P. Chen and P. A. Tsai, Phys. Rev. Fluids, 2017, 2, 1–9 CrossRef.
- N. Moradpour, J. Yang and P. A. Tsai, Curr. Opin. Colloid Interface Sci., 2024, 101845 CrossRef CAS.
- N. Moradpour, R. Azadi and P. A. Tsai, Colloids Surf., A, 2025, 705, 135533 CrossRef CAS.
- International Energy Agency (IEA), Whatever happened to enhanced oil recovery?, 2018, https://www.iea.org/commentaries/whatever-happened-to-enhanced-oil-recovery, accessed: 2025-01-01 Search PubMed.
- S. K. Prasad, J. S. Sangwai and H. S. Byun, J. CO2 Util., 2023, 72, 102479 CrossRef CAS.
- A. Alinejad and H. Dehghanpour, Fuel, 2024, 359, 130387 CrossRef CAS.
- A. Alinejad, J. Behseresht and H. Dehghanpour, SPE J., 2025, 30, 251–271 CrossRef CAS.
- N. Mungan, SPE West. Reg. Meetin, Long Beach, California, 1991, pp. 113–122 Search PubMed.
- F. B. Thomas, N. Holowach, X. Zhou, D. B. Bennion and D. W. Bennion, Proc. - SPE Symp. Improv. Oil Recover, 1994, pp. 307–320 Search PubMed.
- N. Zhang, M. Yin, M. Wei and B. Bai, Fuel, 2019, 241, 459–467 CrossRef CAS.
- J. Sun, L. Sun and B. Bao, Fuel, 2025, 392, 134835 CrossRef CAS.
- C. Whitson and M. Brule, Phase Behavior, Society of Petroleum Engineers Inc., 2000, vol. 20 Search PubMed.
- A. Abedini and F. Torabi, Energy Fuels, 2014, 28, 774–784 CrossRef CAS.
- X. Zhang, L. Li, Y. Su, Q. Da, J. Fu, R. Wang and F. Chen, Appl. Energy, 2023, 348, 121518 CrossRef CAS.
- A. Zick, 61st Annu. Tech. Conf. Exhib. Soc. Pet. Eng., 1986, p. 15493 Search PubMed.
- F. I. Stalkup, 62nd Annu. Tech. Conf. Exhib. Soc. Pet. Eng., 1987, p. 16715 Search PubMed.
- J. Han, M. Lee, W. Lee, Y. Lee and W. Sung, Appl. Energy, 2016, 161, 85–91 CrossRef CAS.
- X. Chen, Y. Li, X. Tang, H. Qi, X. Sun and J. Luo, Energy, 2021, 226, 120294 CrossRef CAS.
- Y. Hao, Z. Li, Y. Su, C. Kong, H. Chen and Y. Meng, Energy, 2022, 254, 124349 CrossRef CAS.
- S. Yu, L. Yiqiang, Q. Huan, C. Jinxin and T. Xuechen, 16th Int. Conf. Appl. Energy, 2025 Search PubMed.
- P. Saffman and G. Taylor, Proc. R. Soc. London, Ser. A, 1958, 245, 312–329 CAS.
- R. L. Chuoke, P. Von Meurs, J. Member and A. C. Von Der Poel, Transactions of the AIME, 1959, 216, 188–194 CrossRef.
- T. T. Al-Housseiny, P. A. Tsai and H. A. Stone, Nat. Phys., 2012, 8, 747–750 Search PubMed.
- Y. Guo, F. Liu, J. Qiu, Z. Xu and B. Bao, Energy, 2022, 256, 124524 CrossRef CAS.
- J. Ren, W. Xiao, W. Pu, Y. Tang, Y. Bernabé, Q. Cheng and L. Zheng, Energy, 2024, 297, 131211 CrossRef CAS.
- G. Bongrand and P. A. Tsai, Phys. Rev. E, 2018, 97, 2–5 CrossRef PubMed.
- A. Pouplard and P. A. Tsai, Sci. Rep., 2024, 14, 10–17 CrossRef PubMed.
- N. Kumar, M. Augusto Sampaio, K. Ojha, H. Hoteit and A. Mandal, Fuel, 2022, 330, 125633 CrossRef CAS.
- M. Almobarak, Z. Wu, D. Zhou, K. Fan, Y. Liu and Q. Xie, Petroleum, 2021, 7, 245–253 CrossRef.
- B. Dindoruk, R. Johns and F. M. Orr, SPE Reservoir Eval. Eng., 2021, 24, 367–389 CrossRef CAS.
- W. F. Yellig and R. S. Metcalfe, JPT, J. Pet. Technol., 1980, 32, 160–168 CrossRef CAS.
- G. Song, Y. Meng, C. Zhang, Z. Zhao and Q. Yang, ACS Omega, 2024, 9, 14747–14765 CrossRef CAS PubMed.
- D. N. Rao, Fluid Phase Equilib., 1997, 139, 311–324 CrossRef CAS.
- S. C. Ayirala and D. N. Rao, SPE Soc. Pet. Eng., 2006, 1, 349–363 Search PubMed.
- R. L. Christiansen and H. K. Haines, SPE Reservoir Eng., 1987, 2, 523–527 CrossRef CAS.
- K. Zhang, N. Jia, F. Zeng, S. Li and L. Liu, J. Pet. Sci. Eng., 2019, 183, 106366 CrossRef CAS.
- K. Jessen and F. M. Orr, SPE Reservoir Eval. Eng., 2008, 933–939 CrossRef CAS.
- D. Zhou and F. M. Orr, SPE J., 1998, 3, 19–25 CrossRef.
- R. B. Alston, G. P. Kokolis and C. F. James, SPEJ, Soc. Pet. Eng. J., 1985, 25, 268–274 CrossRef CAS.
- O. S. Glaso, SPEJ, Soc. Pet. Eng. J., 1985, 25, 927–934 CrossRef.
- H. Chen, B. Li, I. Duncan, M. Elkhider and X. Liu, Fuel, 2020, 278, 118272 CrossRef CAS.
- E. H. Benmekki and G. A. Mansoori, SPE Reservoir Eng., 1988, 3, 559–564 CrossRef CAS.
- S. N. Ashrafizadeh and A. Ameri Ghasrodashti, Chem. Eng. Res. Des., 2011, 89, 690–696 CrossRef CAS.
- K. Ma, R. Liontas, C. A. Conn, G. J. Hirasaki and S. L. Biswal, Soft Matter, 2012, 8, 10669–10675 RSC.
- C. A. Conn, K. Ma, G. J. Hirasaki and S. L. Biswal, Lab Chip, 2014, 14, 3968–3977 RSC.
- X. Zheng, N. Mahabadi, T. S. Yun and J. Jang, J. Geophys. Res.:Solid Earth, 2017, 122, 1634–1647 CrossRef CAS.
- Y. Li, F. Kazemifar, G. Blois and K. T. Christensen, Water Resour. Res., 2017, 5, 6178–6196 CrossRef.
- M. Saadat, J. Yang, M. Dudek, G. Øye and P. A. Tsai, J. Pet. Sci. Eng., 2021, 203, 108647 CrossRef CAS.
- J. Yang and P. A. Tsai, Chem. Eng. Sci., 2024, 300, 120543 CrossRef CAS.
- S. Yang, H. Li, S. Suo and Z. Wu, Adv. Water Resour., 2024, 191, 104757 CrossRef.
- L. Li, J. Zheng, Y. Shi, Y. Su, Y. Hao and Z. Chen, Energy Fuels, 2024, 38, 7997–8008 CrossRef CAS.
- J. Wang, J. Sun, J. Shi and B. Bao, Lab Chip, 2025, 25, 1981–1992 RSC.
- N. Moradpour and P. A. Tsai, Microfluidic Investigation of CO2 Foam Flow in a Heterogeneous Porous Medium, Lab Chip, 2025 10.1039/D5LC00544B , in press.
- N. Liu, C. Aymonier, C. Lecoutre, Y. Garrabos and S. Marre, Chem. Phys. Lett., 2012, 551, 139–143 CrossRef CAS.
- M. Abolhasani, M. Singh, E. Kumacheva and A. Günther, Lab Chip, 2012, 12, 1611–1618 RSC.
- A. Sell, H. Fadaei, M. Kim and D. Sinton, Environ. Sci. Technol., 2013, 47, 71–78 CrossRef CAS PubMed.
- N. Qin, J. Z. Wen and C. L. Ren, Phys. Rev. E, 2017, 95, 1–15 Search PubMed.
- N. Qin, J. Z. Wen, B. Chen and C. L. Ren, Appl. Phys. Lett., 2018, 113, 033703 CrossRef.
- S. G. Lefortier, P. J. Hamersma, A. Bardow and M. T. Kreutzer, Lab Chip, 2012, 12, 3387–3391 RSC.
- T. H. M. Ho, J. Yang and P. A. Tsai, Lab Chip, 2021, 21, 3942–3951 RSC.
- T. H. M. Ho, D. Sameoto and P. A. Tsai, Chem. Eng. Res. Des., 2021, 174, 116–126 CrossRef CAS.
- J. Yang and P. A. Tsai, Biomicrofluidics, 2024, 18, 051301 CrossRef CAS PubMed.
- S. Yang, G. Kong, Z. Cao and Z. Wu, Chem. Eng. J. Adv., 2023, 16, 100518 CrossRef CAS.
- S. Virappane, R. Azadi, N. Mukherjee and P. A. Tsai, Phys. Fluids, 2024, 36, 102019 CrossRef CAS.
- Y. Guo, F. Li, S. Saber, M. Zargartalebi, S. S. Sun, Y. C. Xiao, B. Bao, Z. Xu and D. Sinton, Lab Chip, 2025, 25(12), 2918–2925, 10.1039/D4LC00772G.
- F. Mostowfi, S. Molla and P. Tabeling, Lab Chip, 2012, 12, 4381–4387 RSC.
- W. Song, H. Fadaei and D. Sinton, Environ. Sci. Technol., 2014, 48, 3567–3574 CrossRef CAS PubMed.
- B. Bao, J. Riordon, Y. Xu, H. Li and D. Sinton, Anal. Chem., 2016, 88, 6986–6989 CrossRef CAS PubMed.
- Y. Xu, J. Riordon, X. Cheng, B. Bao and D. Sinton, Angew. Chem., 2017, 129, 14150–14155 CrossRef.
- B. Bao, J. Riordon, F. Mostowfi and D. Sinton, Lab Chip, 2017, 17, 2740–2759 RSC.
- M. J. Fornerod, E. Amstad and S. Guldin, Mol. Syst. Des. Eng., 2020, 5, 358–365 RSC.
- P. Nguyen, D. Mohaddes, J. Riordon, H. Fadaei, P. Lele and D. Sinton, Anal. Chem., 2015, 87, 3160–3164 CrossRef CAS PubMed.
- A. Sharbatian, A. Abedini, Z. Qi and D. Sinton, Anal. Chem., 2018, 90, 2461–2467 CrossRef CAS PubMed.
- B. Bao, J. Feng, J. Qiu and S. Zhao, ACS Omega, 2021, 6, 943–953 CrossRef CAS PubMed.
- J. Shi, L. Tao, Y. Guo, X. He, Y. Li and B. Bao, Fuel, 2024, 362, 130876 CrossRef CAS.
- X. Pan, L. Sun, F. Chen, X. Huo, Y. Wang, C. Feng, X. Zheng and Z. Zhang, Energy Fuels, 2024, 38, 10904–10913 CrossRef CAS.
- L. Tao, W. Liu, J. Shi, Y. Guo, W. Qin and B. Bao, Chem. Eng. Sci., 2025, 302, 120828 CrossRef CAS.
- H. Zou, A. C. Slim and A. Neild, Anal. Chem., 2019, 91, 13681–13687 CrossRef CAS PubMed.
- H. Zou, H. Kang, A. C. Slim and A. Neild, Lab Chip, 2020, 20, 3582–3590 RSC.
- F. Ungar, S. Ahitan, S. Worthing, A. Abedini, K. Uleberg and T. Yang, J. Pet. Sci. Eng., 2022, 208, 109415 CrossRef CAS.
- D. Pereponov, M. Tarkhov, D. B. Dorhjie, A. Rykov, I. Filippov, E. Zenova, V. Krutko, A. Cheremisin and E. Shilov, Energies, 2023, 16, 4994 CrossRef CAS.
- D. Qin, Y. Xia, J. A. Rogers, R. J. Jackman and G. M. Whitesides, Microsyst. Technol. Chem. Life Sci., Springer, Berlin, Heidelberg, 1998, vol. 194, pp. 2–20 Search PubMed.
- V. A. Lifton, Lab Chip, 2016, 16, 1777–1796 RSC.
- D. Sinton, Lab Chip, 2014, 14, 3127–3134 RSC.
- A. Abedini, S. Ahitan, Z. Barikbin, V. Soni, J. Ratulowski and D. Sinton, Energy Fuels, 2022, 36, 8578–8590 CrossRef CAS.
- S. S. Datta, I. Battiato, M. A. Fernø, R. Juanes, S. Parsa, V. Prigiobbe, E. Santanach-Carreras, W. Song, S. L. Biswal and D. Sinton, Lab Chip, 2023, 23, 1358–1375 RSC.
- N. Convery and N. Gadegaard, Micro Nano Eng., 2019, 2, 76–91 CrossRef.
- S. Marre and K. F. Jensen, Chem. Soc. Rev., 2010, 39, 1183–1202 RSC.
- S. Marre, A. Adamo, S. Basak, C. Aymonier and K. F. Jensen, Ind. Eng. Chem. Res., 2010, 49, 11310–11320 CrossRef CAS.
- Z. B. Qi, L. Xu, Y. Xu, J. Zhong, A. Abedini, X. Cheng and D. Sinton, Lab Chip, 2018, 18, 3872–3880 RSC.
- C. A. Schneider, W. S. Rasband and K. W. Eliceiri, Nat. Methods, 2012, 9, 671–675 CrossRef CAS PubMed.
- National Institute of Standards and Technology (NIST), NIST Chemistry WebBook, SRD 69, 2025, https://webbook.nist.gov/chemistry/, accessed: 2025-01-01 Search PubMed.
- Z. Pan and J. P. M. Trusler, Fluid Phase Equilib., 2023, 566, 113700 CrossRef CAS.
- J. K. Nunes, S. S. Tsai, J. Wan and H. A. Stone, J. Phys. D: Appl. Phys., 2013, 46, 114002 CrossRef PubMed.
- T. Cubaud and T. G. Mason, Phys. Fluids, 2008, 20, 053302 CrossRef.
- A. S. Utada, A. Fernandez-Nieves, H. A. Stone and D. A. Weitz, Phys. Rev. Lett., 2007, 99, 1–4 CrossRef PubMed.
- A. Skauge and K. Serbie, Soc. Pet. Eng. - SPE EOR Conf. Oil Gas West Asia 2014 Driv. Integr. Innov. EOR, 2014, pp. 891–905 Search PubMed.
- T. Cubaud and S. Notaro, Phys. Fluids, 2014, 26, 122005 CrossRef.
- D.-Y. Peng and D. B. Robinson, Ind. Eng. Chem. Fundam., 1976, 15, 59–64 CrossRef CAS.
- X. Li, D. Yang, X. Zhang, G. Zhang and J. Gao, Fluid Phase Equilib., 2016, 417, 77–86 CrossRef CAS.
- J. S. Lopez-Echeverry, S. Reif-Acherman and E. Araujo-Lopez, Fluid Phase Equilib., 2017, 447, 39–71 CrossRef CAS.
- R. Jiménez-Gallegos, L. A. Galicia-Luna and O. Elizalde-Solis, J. Chem. Eng. Data, 2006, 51, 1624–1628 CrossRef.
- F. P. Nascimento, M. L. Paredes, A. P. D. Bernardes and F. L. Pessoa, J. Supercrit. Fluids, 2019, 154, 1–9 CrossRef.
- Y. S. Wei and R. J. Sadus, AIChE J., 2000, 46, 169–196 CrossRef CAS.
- İ. Tosun, The Thermodynamics of Phase and Reaction Equilibria, Elsevier, 2nd edn, 2021, pp. 365–478 Search PubMed.
- G. Wilson, AIChE Annual Meeting, Cleveland, Ohio, 1968 Search PubMed.
- M. Abdurrahman, A. K. Permadi, A. Arsad, A. F. Abdul Rahman, W. Bae, U. Z. Husna, A. L. Pang and R. Fauzi, ACS Omega, 2023, 8, 8703–8711 CrossRef CAS PubMed.
- M. Lashkarbolooki, M. J. Eftekhari, S. Najimi and S. Ayatollahi, J. Supercrit. Fluids, 2017, 127, 121–128 CrossRef CAS.
- Design Institute for Physical Property Research/AIChE, DIPPR's Project 801 Database, 2025, https://www.aiche.org/dippr, accessed: 2025-01-01 Search PubMed.
- J. M. Ortega, Iterative solution of nonlinear equations in several variables, Academic Press, 1970 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |