Rhodium catalyst immobilization in trialkylamine-functionalized ionic liquids as a new efficient way to promote biphasic reductive hydroformylation of methyl 10-undecenoate
Camille
Galand
 a,
Jérémy
Ternel
a,
Jérémy
Ternel
 a,
Nicolas
Kania
a,
Nicolas
Kania
 a,
Frédéric
Capet
b,
Hervé
Bricout
a,
Frédéric
Capet
b,
Hervé
Bricout
 a,
Sébastien
Tilloy
a,
Sébastien
Tilloy
 a,
Eric
Monflier
a,
Eric
Monflier
 a and
Michel
Ferreira
a and
Michel
Ferreira
 *a
*a
aUniv. Artois, CNRS, Centrale Lille, Univ. Lille, UMR 8181, Unité de Catalyse et Chimie du Solide (UCCS), Rue Jean Souvraz, SP 18, 62300, Lens, France
bUniv. Lille, CNRS, Centrale Lille, Univ. Artois, UMR 8181, Unité de Catalyse et Chimie du Solide (UCCS), 59000, Lille, France. E-mail: michel.ferreira@univ-artois.fr
First published on 2nd April 2026
Abstract
In this work, trialkylamine-functionalized ionic liquids were assessed for their dual role as solvents and ligands in the biphasic reductive hydroformylation of methyl 10-undecenoate (MU), a renewable substrate derived from castor oil. Systematic tuning of the aminated ionic liquids through cation and anion variation identified 1-(2-piperid-1-yl-ethyl)-3-ethanolimidazolium hexafluorophosphate, [PEEtOHim][PF6] combined with dodecane as the most effective biphasic couple, enabling efficient rhodium reductive hydroformylation with a turnover frequency in alcohols of 33 h−1 while limiting rhodium leaching in the apolar phase to as little as 1.5% of its initial loading. Under optimized conditions, the system was recycled for at least nine consecutive runs with constant selectivity in alcohols, highlighting the robustness of the catalytically active species stabilized within the ionic liquid phase. A cumulative total turnover number in alcohols of 1282 was achieved through this atom-economical tandem process, further demonstrating the potential of aminated ionic liquids in biphasic catalytic systems. This work underscores the relevance of ionic liquid-based strategies for sustainable catalysis and the valorization of renewable feedstocks, bridging high-performance chemistry with the principles of green chemistry.
Green foundation1. This work presents an innovative all-in-one catalytic system in which a ligand is covalently linked to an ionic liquid, combining catalyst stabilization and solvent function within a single greener medium. Applied for the first time to reductive hydroformylation, this approach enhances process efficiency while reducing environmental impact.2. We achieved 98% metal retention within the ionic liquid phase in a biphasic catalytic setup, reaching a total turnover number in alcohols (TTON) of 1282 over nine consecutive cycles with no loss in activity. The ionic liquids employed were thoroughly characterized, and their robustness within the system was established. 3. To further strengthen the sustainability of this methodology, future work should prioritize strategies aimed at improving catalyst stability to ensure consistent performance across multiple cycles. Exploration of alternative, such as amine-based deep eutectic solvents or biphasic IL/scCO2 catalytic systems could lead to improved environmental performance. |
Introduction
Primary alcohols represent a highly valuable class of products for the chemical industry. They find direct applications as solvents and disinfectants, as well as key intermediates in the production of detergents, plasticizers, and polymers.1 Among the different synthetic routes, reductive hydroformylation is a particularly attractive option from both economic and environmental perspectives, since it provides direct access to primary alcohols from olefins by combining hydroformylation and hydrogenation as successive steps with a 100% atomic efficiency from syngas.2,3Various metals have been reported to catalyse this transformation through single- or dual-catalyst strategies, including palladium4 and ruthenium,5,6 with cobalt and rhodium remaining the most extensively investigated.2 Historically, homogeneous cobalt-based catalytic systems have demonstrated the ability to promote direct alcohol formation, albeit under harsh reaction conditions. These conditions also induce significant isomerisation and hydrogenation activity, leading to the concurrent production of paraffins alongside alcohols.7 More recently, studies have highlighted a renewed interest in a cobalt-based catalytic system operating in milder conditions. However, this system relies on a highly basic phosphine ligand that is prone to oxidation and requires photoactivation, which poses significant limitations for large-scale industrial applications.8 Rhodium-based catalysts exhibit higher activity and selectivity in hydroformylation but the use of basic phosphine ligands is also essential to enable efficient one-step conversion of olefins into alcohols by enhancing the hydrogenation step.9 Consequently, despite extensive research into phosphane-modified rhodium catalysts, industrial processes for alkyl alcohol synthesis continue to be based on two-step processes, with aldehydes being isolated as intermediates.10,11
Catalytic systems operating without phosphorus ligands have also been investigated. Rh/tertiary amine complexes have shown high efficiency in promoting the one-step tandem reductive hydroformylation of olefins.12–25 These systems offer several advantages: beyond their ability to promote aldehyde hydrogenation, amine ligands display greater stability toward oxidation and lower toxicity compared to the widely used phosphorus ligands. However, a major challenge remains unsolved: the lack of a satisfactory solution for complete catalyst recycling. Biphasic catalysis addresses this issue by combining the high reactivity of homogeneous systems with the advantage of simplified catalyst separation and recovery. In this context, several rhodium-based catalytic biphasic systems have been investigated in recent studies.26–31
Following the significant progress achieved in biphasic hydroformylation using ionic liquids,32,33 we aimed to design a system suitable for tandem reductive hydroformylation. In the initial development of biphasic ionic liquid systems for hydroformylation reaction, ruthenium was evaluated as a catalyst for tandem reductive hydroformylation in [Bu4P][Br] under harsh conditions (180 °C, 80 bar CO/H2 at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio), using a relatively high catalyst loading of 3 mol%. Under optimized conditions, starting from RuO2 as a precursor, the system achieved moderate selectivity of around 65% toward alcohols.34 [HRu3(CO)11]− was highlighted by IR spectroscopy as being the catalytically active species. A key advantage of this system is the exceptional stability of the ruthenium carbonyl cluster species in the quaternary phosphonium salt matrix, even under low CO/H2 pressure and high temperature, unlike their homogeneous counterparts. This behaviour is likely attributed to the formation of ion pairs of the type [Bu4P][Ru cluster], which facilitate the efficient separation of alcohol and aldehyde products by distillation and support catalyst recycling without any loss of activity. In the rhodium/trialkylamines catalytic systems, anionic rhodium clusters are considered as active species during reductive hydroformylation,15 revealing the strong potential of ionic liquids as a development platform for this transformation. Building on this approach, our team has developed a biphasic catalytic system compatible with ionic liquids, enabling the tandem reductive hydroformylation of methyl 10-undecenoate, a biobased substrate of interest.35 This renewable feedstock, derived industrially from castor oil, was chosen for its strong potential in polymer chemistry, as it can be further transformed through reductive hydroformylation followed by reduction or oxidation into diols or dicarboxylic acids, both essential precursors for the production of polyester-12. In this catalytic system, an ionic tertiary amine, N,N-dimethyltaurinate salt, enabled the immobilization of the rhodium catalyst in the polar ionic liquid phase. Remarkably, the system operates efficiently with just two equivalents of potassium N,N-dimethyltaurinate per rhodium, in contrast to the typically high amine/Rh ratios required for such transformation, highlighting the beneficial stabilizing effect of the ionic liquid 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide, [BMIM][NTf2] on the catalytic species. Under optimized conditions, the substrate was nearly fully converted into alcohols. Although the system exhibited high selectivity toward alcohols, recycling experiments revealed a limitation: catalytic activity declined after a few cycles. This loss was attributed to amine leaching into the organic phase, as reintroducing a fresh amine charge restored activity. Another constraint was the poor solubility of the ligand in [BMIM][NTf2], which prevented the use of higher amine/Rh ratios. Replacing the potassium cation with butylmethylimidazolium (BMIM+) improved solubility, allowing an amine/Rh ratio of 10 and extending catalyst performance over successive runs compared to the potassium analogue. However, this modification also led to increased rhodium leaching, passing from 0.8% per cycle with the potassium-based ligand to 4.2% with the BMIM+ version, likely due to the enhanced solubility of the ligand in the non-polar heptane phase.
2 ratio), using a relatively high catalyst loading of 3 mol%. Under optimized conditions, starting from RuO2 as a precursor, the system achieved moderate selectivity of around 65% toward alcohols.34 [HRu3(CO)11]− was highlighted by IR spectroscopy as being the catalytically active species. A key advantage of this system is the exceptional stability of the ruthenium carbonyl cluster species in the quaternary phosphonium salt matrix, even under low CO/H2 pressure and high temperature, unlike their homogeneous counterparts. This behaviour is likely attributed to the formation of ion pairs of the type [Bu4P][Ru cluster], which facilitate the efficient separation of alcohol and aldehyde products by distillation and support catalyst recycling without any loss of activity. In the rhodium/trialkylamines catalytic systems, anionic rhodium clusters are considered as active species during reductive hydroformylation,15 revealing the strong potential of ionic liquids as a development platform for this transformation. Building on this approach, our team has developed a biphasic catalytic system compatible with ionic liquids, enabling the tandem reductive hydroformylation of methyl 10-undecenoate, a biobased substrate of interest.35 This renewable feedstock, derived industrially from castor oil, was chosen for its strong potential in polymer chemistry, as it can be further transformed through reductive hydroformylation followed by reduction or oxidation into diols or dicarboxylic acids, both essential precursors for the production of polyester-12. In this catalytic system, an ionic tertiary amine, N,N-dimethyltaurinate salt, enabled the immobilization of the rhodium catalyst in the polar ionic liquid phase. Remarkably, the system operates efficiently with just two equivalents of potassium N,N-dimethyltaurinate per rhodium, in contrast to the typically high amine/Rh ratios required for such transformation, highlighting the beneficial stabilizing effect of the ionic liquid 1-butyl-3-methylimidazolium bis(trifluoromethanesulfonyl)imide, [BMIM][NTf2] on the catalytic species. Under optimized conditions, the substrate was nearly fully converted into alcohols. Although the system exhibited high selectivity toward alcohols, recycling experiments revealed a limitation: catalytic activity declined after a few cycles. This loss was attributed to amine leaching into the organic phase, as reintroducing a fresh amine charge restored activity. Another constraint was the poor solubility of the ligand in [BMIM][NTf2], which prevented the use of higher amine/Rh ratios. Replacing the potassium cation with butylmethylimidazolium (BMIM+) improved solubility, allowing an amine/Rh ratio of 10 and extending catalyst performance over successive runs compared to the potassium analogue. However, this modification also led to increased rhodium leaching, passing from 0.8% per cycle with the potassium-based ligand to 4.2% with the BMIM+ version, likely due to the enhanced solubility of the ligand in the non-polar heptane phase.
A further extension of this work was subsequently published by our team.36 In this study, various commercially available trialkylamines, less soluble in the ionic liquid phase [BMIM][NTf2] than in the organic heptane phase, were employed in combination with rhodium precursor to catalyze the reductive hydroformylation of methyl 10-undecenoate. Among the tested amines, triethylamine proved to be the most effective, both in promoting alcohol formation and in retaining the catalyst within the ionic liquid phase. Notably, increasing the alkyl chain length reduced amine solubility in the ionic liquid layer, thereby limiting aldehyde hydrogenation. As in our earlier work using N,N-dimethyltaurinate salts, the reductive hydroformylation reaction proceeded efficiently despite a relatively low amine-to-rhodium ratio in the ionic liquid phase, with strong catalyst immobilization confirmed by successful scale-up to a miniplant continuous flow process. Indeed, after 25 hours of uninterrupted operation, a TON of 232 for alcohol production and 780 for hydroformylation was achieved, with rhodium loss into the organic phase limited to 7.4% of the initial charge.
Building on these results and aiming to investigate catalyst behaviour under high amine concentrations in the ionic liquid phase, we employed a series of ionic liquids featuring a covalently bound tertiary amine on an imidazolium backbone. This structural integration was designed to minimize ligand leaching and enhance catalytic performance. These functionalized ionic liquids were assessed both as reaction media and as ligands in combination with rhodium for the reductive hydroformylation of the bio-based methyl 10-undecenoate substrate. A range of reaction parameters was explored to optimize primary alcohol production, and both the recyclability of the catalytic system and its applicability to other substrates were evaluated.
Results and discussion
Effect of trialkylamine-functionalized ionic liquid loading on catalytic activity
Different N-functionalized ionic liquids were synthesized by an adapted existing procedure and fully characterized (Scheme 1).37 The synthesis begins with the quaternization of the nucleophilic nitrogen, affording a corresponding ammonium salt. Subsequent neutralization of this intermediate salt enables an anion metathesis with an organic counterion, ultimately inducing phase separation due to the pronounced hydrophobicity of the resulting ionic liquid. Complete removal of chloride from the ionic liquid phase is achieved through successive aqueous washes, with the absence of residual chloride confirmed by ion chromatography. Some of these compounds, and particularly [PEMim][PF6], were previously employed as solvents in biphasic hydroformylation of 1-octene using an ionic Rh(III) complex bearing a phosphine ligand. However, alcohol formation was not observed, most likely due to competitive coordination at the rhodium centre between the phosphine ligand and the tertiary amine moiety, which inhibited the second-step reduction of the intermediate aldehyde.37 To assess this hypothesis, [PEMim][PF6] was employed as the sole solvent/ligand in the reductive hydroformylation of methyl 10-undecenoate (MU) (Fig. 1a). The reaction pathway presents a broad product distribution. While the desired alcohol is obtained via a tandem hydroformylation and reduction sequence, side reactions may lead to the formation of isomeric variants of the starting MU as well as methyl 10-undecanoate. Isomers may further undergo transformation into branched aldehydes and alcohols. Experiments were conducted at 80 °C under a total pressure of 80 bar, using a CO/H2 gas mixture introduced at a stoichiometric ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, following optimization of the reaction conditions (see paragraph IX, Tables S4, S5 and S6, SI). Rhodium leaching into the organic phase was systematically quantified using inductively coupled plasma optical emission spectrometry (ICP-OES) (Fig. 1b).
1, following optimization of the reaction conditions (see paragraph IX, Tables S4, S5 and S6, SI). Rhodium leaching into the organic phase was systematically quantified using inductively coupled plasma optical emission spectrometry (ICP-OES) (Fig. 1b).
 | ||
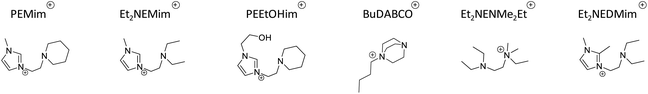
| Scheme 1 (a) Synthetic route to ionic liquids used in this study. (b) Different N-functionalized ionic liquids acronyms. (c) Ionic liquids components. | ||
As a control experiment, the reaction was carried out without any nitrogen donor ligand in heptane. Under these homogeneous conditions, the aldehyde intermediate was formed exclusively, reaching an 96% yield after 6 h.
Indeed, under CO/H2 pressure, rhodium species generated from the Rh(acac)(CO)2 precursor, mainly polynuclear complexes,38,39 are capable of catalyzing the hydroformylation of the starting substrate. Moreover, such a low l/b (linear to branched) aldehyde ratio of 1.0 is commonly reported for rhodium catalysts when no coordinating ligand is present, reflecting minimal regioselectivity. The absence of alcohol products confirms that, under such conditions, the subsequent reduction of the intermediate aldehyde cannot proceed. As a second control experiment, the reaction was carried out under biphasic conditions using the unmodified ionic liquid [BMIM][PF6] (75 molar equivalents relative to rhodium, corresponding to approximately 3% of the overall liquid volume). The results were comparable to the previous test. Given the similarity of the catalytic active species involved, i.e. neutral rhodium-hydrido-carbonyl complexes, a preferential migration into the organic phase was observed. This behaviour led to substantial rhodium leaching from the ionic liquid into the organic layer, as confirmed by rhodium content analysis of the isolated product phase. Indeed, 215 ppm of rhodium was detected following the catalytic run, representing nearly 72% of the initial rhodium charge introduced into the reactor. Once again, a l/b aldehyde ratio as low as 1.0 was consistent with unmodified rhodium catalyst being the active species.40 Moreover, no alcohol formation was detected. A series of experiments was then conducted with increasing amounts of the nitrogen donor ligand [PEMim][PF6]. While the addition of 2 molar equivalents of ligand relative to rhodium was sufficient to initiate alcohol formation (8% yield), confirming the essential role of a coordinative amine moiety in enabling the subsequent hydrogenation of the intermediate aldehyde, rhodium retention in the ionic liquid phase remained limited, with 70% of the initial rhodium content leaching into the organic phase. Increasing the amount of nitrogen-functionalized ionic liquid led to a concomitant rise in alcohol yield and improved rhodium immobilization in the heavy phase, with only 5% leaching observed when 75 equivalents were used. Under these conditions, a 71% alcohol yield was achieved after 6 h, corresponding to a turnover frequency (TOF) of 30 h−1. This value represents a fivefold enhancement in alcohol TOF compared to our previously described ionic liquid-based catalytic system employed under the same conditions (6 h−1),35,36 thereby highlighting the improved efficiency of our new catalytic system. The maximum alcohol yield of around 70% was obtained when using between 25 and 150 equivalents of ligand per rhodium. At higher ligand loadings, rhodium immobilization improved significantly, with leaching reduced to just 3%. However, this gain in immobilization was offset by a progressive reduction in alcohol yield, reaching only 44% yield at 300 equivalents. A similar trend was observed upon varying the loading of another N-functionalized ionic liquid, such as the [Et2NEMim][PF6] (Scheme 1) (see paragraph IX Table S7, entries 7–12, SI), confirming that the amount of ligand significantly affects both rhodium retention and catalytic efficiency. Furthermore, increasing the ligand loading positively influences the overall l/b ratio (aldehyde and alcohol), which progressively rises from approximately 1.0 in the absence of ligand to 1.4 at 300 equivalents. The catalytic system was consequently optimized using 75 equivalents of trialkylamine-functionalized ionic liquid relative to the rhodium catalyst, thereby providing an ionic liquid volume of approximately 0.4 mL (i.e. 3.4% of the overall liquid volume), which enabled efficient phase separation while minimizing both ligand consumption and rhodium leaching.
Counterion screening
The counterion of the ionic liquid was varied to evaluate its influence on the reaction outcome. Several combinations were tested, and the observed effects are summarized in Table 1. The highest alcohol yield (71%) was obtained in the presence of the hexafluorophosphate counterion (entry 2). Other weakly coordinating anions, such as BF4− and Tf2N−, also enabled alcohol formation, albeit with lower yields of 42% and 16%, respectively. Comparable trends have been reported in biphasic hydroformylation systems based on 1-butyl-3-methylimidazolium (BMIM+) ionic liquids, where substrate solubility is strongly governed by the identity of the anion.41 Although olefins generally display higher solubilities in Tf2N−-based ionic liquids, the greater coordinating ability of this anion has been cited as a factor contributing to reduced TOFs.42 In a similar manner, the use of more strongly coordinating anions such as (CN)2N− or Cl− led to a marked decrease in substrate conversion and alcohol production. Notably, in the presence of chloride, methyl 10-undecenoate conversion dropped to 40%, with no detectable alcohol formation. The competitive coordination of the ionic liquid constituents, i.e. anion and amine, to the rhodium center compromises substrate conversion efficiency. Indeed, the significant variations in alcohol yield observed highlight the strong influence of the counterion on catalytic activity. Rhodium leaching into the organic phase remained similar across all systems, ranging between 3% and 5%. The AcO−-based ionic liquid differs from the other systems in its noticeably lower thermal stability, as evidenced by its ATG thermogram (see Fig. S76, SI). This reduced stability likely contributes to the diminished catalytic activity observed, together with the higher level of rhodium leaching measured in this medium.| Entry | Counter ion (X) | Conv.b (%) |
Y
(Ald)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]d
(%) [l/b]d |
Y
(Alc)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]e
(%) [l/b]e |
Global [l/b]f |
Y
(Iso)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Y
(Sat)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Rh leaching (%) | TOF(alc) (h−1) |
|---|---|---|---|---|---|---|---|---|---|
a Experimental conditions: Rh(acac)(CO)2 (6.0 mg, 23.3 μmol, 1 equiv.), [PEMim][X] (75 equiv.), methyl 10-undecenoate (1.325 mL, 5.9 mmol, 250 equiv.), heptane (10 mL), 80 bar CO/H2 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio. 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio.
|
|||||||||
| 1 | BF4− | 83 | 28 [1.1] | 42 [3.3] | 2.1 | 8 | 5 | 3.1 | 18 |
| 2 | PF6− | 99 | 20 [0.4] | 71 [1.8] | 1.3 | 6 | 2 | 5.0 | 30 |
| 3 | Tf2N− | 95 | 53 [1.5] | 16 [2.9] | 1.7 | 11 | 15 | 3.8 | 7 |
| 4 | (CN)2N− | 72 | 44 [1.9] | 3 [—] | 1.9 | 14 | 11 | 1.9 | 1 |
| 5 | AcO− | 75 | 43 [1.8] | 5 [—] | 1.8 | 16 | 11 | 12.4 | 1 |
| 6 | Cl− | 40 | 22 [1.8] | 0 [—] | 1.8 | 7 | 11 | 4.9 | 0 |
As this study sought to establish fundamental structure–activity relationships by identifying the ionic liquid (cation/anion pair) that maximized alcohol yield in the reductive hydroformylation, PF6− was selected for further optimization because it delivered the highest catalytic activity while ensuring efficient catalyst immobilization in the ionic liquid phase. Yet PF6− is well known to be hydrolytically unstable,43 and although NMR analysis of the ionic-liquid phase revealed no detectable signs of decomposition, despite the absence of particular precautions during the catalytic experiments (see Fig. S62, S63 and S64, SI), its limited long-term stability and environmental persistence remain concerns when compared with more robust anions such as Tf2N−. Enhancing the sustainability of the process will therefore require replacing PF6− with a more stable and environmentally benign anion in future developments.
Kinetic monitoring
To gain deeper insight into the mechanistic aspects of the reductive hydroformylation of methyl 10-undecenoate, the reaction was kinetically monitored over time (Fig. 2a).Experimental conditions were kept constant compared to the previous studies but the total volume in the reactor was increased to get enough solution to sample the reaction over time. The formation of aldehydes was observed from the earliest stages, indicating that the reaction proceeds without any detectable induction period. The TOF(ald) measured in this system during the first 30 minutes of reaction reached approximately 142 h−1. For comparison, the sulfonated analogue of xantphos used in the biphasic hydroformylation of 1-decene in [BMIM][PF6] at 50 bar of CO/H2 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and 80 °C affords a catalytic system with a TOF(ald) of about 33 h−1, which can be increased to 75 h−1 when switching to 1-octene at 40 bar (CO/H2, 1
1) and 80 °C affords a catalytic system with a TOF(ald) of about 33 h−1, which can be increased to 75 h−1 when switching to 1-octene at 40 bar (CO/H2, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and employing TPPTS as the phosphorus ligand.44,45
1) and employing TPPTS as the phosphorus ligand.44,45
The evolution of the overall l/b ratio initially starts with the highest value of 2.4, reflecting the preferential reactivity of the terminal alkene isomer. As the reaction progresses, however, the l/b ratio decreases, indicating the involvement of isomerization processes. This suggests that terminal isomers display higher reactivity and are converted more rapidly than the internal ones, most likely because of reduced steric hindrance. Complete conversion of methyl 10-undecenoate is achieved after approximately 3 hours, marking a clear transition between the hydroformylation and hydrogenation stages. The delay in alcohol formation relative to aldehyde production highlights the slower kinetics of the second aldehyde reduction step. This kinetic profile is consistent with our previous studies dealing with reductive hydroformylation in ionic liquids.35 Following the detection of a small amount of rhodium leaching into the organic phase (approximately 5% of the initial catalyst loading, Table 1, entry 2), a hot phase separation test was performed after 3 hours to determine the location of the catalytically active species in the reaction (Fig. 2b). After decantation, the organic phase was transferred under CO/H2 to a separate reactor and subjected to an additional 3-hour reaction in the same reaction conditions (80 bar of CO/H2, 80 °C). The lack of alcohol formation at 6 h confirmed the hydrogenation activity interruption, thereby indicating that the second step of the tandem reaction takes place exclusively within the ionic liquid phase. Nonetheless, hydroformylation activity was detected in the organic phase, represented by the transformation of internal isomers into branched aldehydes, attributable to the presence of rhodium species resulting from the observed leaching. This catalytic activity was comparable to that obtained under classical biphasic conditions, with yields in isomers decreasing from 18% at 3 h to 9% at 6 h. This suggests that hydroformylation is mainly catalyzed by rhodium species located in the apolar phase which may account for the higher hydroformylation activity of this system compared with the more strongly immobilized phosphorus-based systems previously reported using TPPTS or sulfoxantphos as ligands. Rhodium leaching quantified during hot phase separation showed a maximum concentration in the apolar phase, in contrast to the lower values determined after cooling the autoclave to ambient temperature. Indeed, this experiment evidenced almost twice the leached fraction, reaching around 10% of the initial rhodium content. This behaviour confirms the dynamic partitioning of rhodium hydrido complexes between the two phases at elevated temperature under CO/H2 pressure, with the ionic liquid phase acting as a reservoir of hydroformylation catalytic species. Moreover, complementary HPLC measurements revealed that ionic liquid leaching (around 500 ppm) occurred alongside rhodium leaching, a correlation that may help explain the metal loss observed in the organic phase of this system.
Tuning catalysis through ligand design
We next examined the influence of the N-coordinating cation on the catalytic performance (Table 2). Modifying the N-functionalization from a piperidine to a diethylamino moiety resulted in no significant change in reactivity (Table 2, entries 1 and 2).| Entry | Ionic liquid | Conv.b (%) |
Y
(Ald)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]d
(%) [l/b]d |
Y
(Alc)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]e
(%) [l/b]e |
Global [l/b]f |
Y
(Iso)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Y
(Sat)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Rh leaching (%) | TOF(alc) (h−1) | H_ |
|---|---|---|---|---|---|---|---|---|---|---|
a Experimental conditions: Rh(acac)(CO)2 (6.0 mg, 23.3 μmol, 1 equiv.), ionic liquid (75 equiv.), methyl 10-undecenoate (1.325 mL, 5.9 mmol, 250 equiv.), heptane (10 mL), 80 bar CO/H2 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio. 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio.
|
||||||||||
| 1 | [PEMim][PF6] | 99 | 20 [0.4] | 71 [1.8] | 1.3 | 6 | 2 | 5.0 | 29 | 9.47 |
| 2 | [Et2NEMim][PF6] | 100 | 23 [0.4] | 63 [2.1] | 1.4 | 9 | 6 | 7.2 | 26 | 9.10 |
| 3 | [PEEtOHim][PF6] | 99 | 19 [0.3] | 78 [1.7] | 1.2 | 2 | 0 | 1.5 | 33 | 9.43 |
| 4 | [BuDABCO][Tf2N] | 100 | 77 [1.1] | 0 | 1.1 | 2 | 21 | 45.6 | 0 | n.d. |
| 5 | [Et2NENMe2Et][PF6] | 99 | 31 [0.7] | 66 [1.9] | 1.3 | 3 | 0 | 4.9 | 26 | 8.70 |
| 6 | [Et2NEMim][PF6] (100 °C) | 100 | 41 [0.4] | 49 [2.1] | 1.4 | 3 | 7 | 12.8 | 21 | 9.10 |
| 7 | [Et2NEDMim][PF6] (100 °C) | 100 | 26 [0.6] | 50 [2.1] | 1.4 | 14 | 10 | 2.1 | 21 | 9.30 |
In contrast, introducing an alcohol arm onto the imidazole scaffold markedly enhanced hydrogenation activity, with the alcohol yield increasing from 71% to 78% when using [PEEtOHim][PF6] (Table 2, entry 3). This ionic liquid provided the highest alcohol yield while simultaneously minimizing rhodium leaching to only 2%. A similar phenomenon had already been demonstrated earlier by Kaneda and co-workers, who showed that hydroxyl functionalities exert a strong positive influence on the performance of rhodium catalyst immobilized on polystyrene modified with trialkylamine and alcohol groups in the reductive hydroformylation of olefins.46 In their system, the absence of hydroxyl groups resulted in very low catalytic activity, whereas increasing the concentration of hydroxyl functionalities led to higher alcohol yields. The introduction of these groups was proposed to stabilize active rhodium carbonyl cluster anions. Later, another research group reported a comparable effect in PEG300 modified with amino groups used as a stationary phase for rhodium immobilization. Incorporating polar functional groups, particularly alcohol moieties, significantly enhanced the hydrogenation activity of the rhodium catalyst during the reductive hydroformylation of 1-pentene.30 Another study provided direct evidence that supramolecular substrate orientation can accelerate the key hydrogenation step in phosphorus-based catalysts. Indeed, Breit demonstrated that a guanidinium-functionalized phosphine ligand increases hydrogenation rates by H-bonding to the aldehyde intermediate and activating it toward reduction.47 By analogy, the superior performance of [PEEtOHim][PF6] may similarly arise from the ability of its hydroxyl group to weakly stabilize intermediate aldehydes in the same way, creating a structured microenvironment that facilitates the hydrogenation step as well. Although direct structural evidence is still lacking, this hypothesis is consistent with the observed high activity.
Remarkably, no alcohol formation was observed when employing the non-imidazolium-based trialkylamine ionic liquid derived from mono-N-alkylated 1,4-diazabicyclo[2.2.2]octane (DABCO) (Table 2, entry 4). Furthermore, the limited capacity of this ionic liquid to effectively immobilize the rhodium catalyst was demonstrated by substantial leaching, with 45.6% of the initial rhodium content detected in the organic phase. This behaviour raises important questions about the presumed inertness of the N-functionalized imidazolium ionic liquids employed in this study. In base-catalyzed organic reactions, the stability of such ionic liquids is frequently challenged due to the potential formation of N-heterocyclic carbenes (NHCs) via acid–base equilibria. Although the pKa values strongly favour retention of the imidazolium form (pKa ≈ 24 for the imidazolium salts and ≈11 for trialkylamines),48 the observation of side-products in various transformations conducted in ionic liquids suggests that NHCs may actively contribute to the reaction mechanism.49 Nevertheless, previous studies on [PEMim][PF6] stability have shown that counterion interactions can disrupt intramolecular C8–H⋯Npiperidine coordination (see Fig. 3 for the numbering) and proton transfer, thereby making carbene generation from these basic ILs unlikely in the absence of an external base and then concluding that this IL was suitable for application as dual solvent/base in organic/organometallic transformations.50 Additional evidence was provided by the molecular structures of [PEMim][PF6] and [PEEtOHim][PF6], associated with the more active catalytic systems, resolved by single-crystal X-ray diffraction (Fig. 3). Indeed, the resolved structure confirmed the lack of hydrogen bonding between the C8–H and the piperidine nitrogen, as the intermolecular distance is too long to allow proton transfer from C8 to Npiperidine.
To rigorously test this hypothesis, we employed the methylated analogue of [Et2NEMim][PF6], i.e.[Et2NEDMim][PF6], which lacks the acidic proton at the C8 position of the imidazolium ring, and compared results obtained with both ionic liquids. The diethylamino derivative was selected owing to the lower melting point of its methylated analogue (82 °C), in contrast to the 125 °C observed for the methylated piperidine-based compound [PEDMim][PF6] (see paragraph VII, SI). Accordingly, the reaction was carried out at 100 °C to ensure complete melting of the ionic liquid (Table 2, entries 6 and 7). The product distribution remained comparable between reactions performed in [Et2NEMim][PF6] and [Et2NEDMim][PF6] suggesting an absence of carbene-type intermediates under the tested conditions. Furthermore, systematic NMR analyses of both the ionic liquid and organic phases post-reaction revealed no formation of side products that could arise from carbene intermediates (Fig. S61, SI).
Similarly, the catalytic performance observed with the non-imidazolium based ionic liquid [Et2NENMe2Et][PF6] clearly demonstrated the absence of catalytically active carbenes in this reaction. Indeed, the product distribution was comparable to that obtained with imidazolium-based [PEMim][PF6] or [Et2NEMim][PF6] with a similar TOF(alc) value of 26 h−1.
In order to rationalize the distinct reactivity observed with [BuDABCO][Tf2N], we evaluated the basicity of the ligands used, as this property plays a decisive role in determining the activity of the reductive hydroformylation catalysts.15 For this purpose, the reverse Hammett function (H_) was determined using the Hammett indicator method in methanol solution, according to eqn (1):51,52
| H\_ = pKBH + log[B−]/[BH] | (1) |
Although [PEEtOHim][PF6] enables the formation of a more active catalytic system for the reductive hydroformylation of MU, its synthesis is affected by lower yields. In contrast, [PEMim][PF6] benefits from a simpler synthetic protocol, prompting further optimization efforts to be carried out using this ionic liquid.
Solvent screening and optimization
Several solvents were tested as organic phases in combination with [PEMim][PF6] and are listed in Table 3. Two distinct solvent categories can be identified. Solvents used in entries 1 to 5 lead to biphasic catalytic systems upon reaction completion, whereas those employed in entries 6 to 8 result in homogeneous systems. Similarly, the use of the neat substrate without any organic solvent (entry 10), combined with an increase in the substrate/catalyst ratio to 2500, aimed at maintaining a constant liquid volume in the reactor, resulted in a homogeneous reaction medium.| Entry | Solvent | Conv.b (%) |
Y
(Ald)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]d
(%) [l/b]d |
Y
(Alc)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%) [l/b]e
(%) [l/b]e |
Y
(Iso)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Y
(Sat)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (%)
(%) |
Global [l/b]f | Rh leaching (%) | TOF(alc) (h−1) |
|---|---|---|---|---|---|---|---|---|---|
a Experimental conditions: Rh(acac)(CO)2 (6.0 mg, 23.3 μmol, 1 equiv.), [PEMim][PF6], (592 mg, 75 equiv.), methyl 10-undecenoate (1.325 mL, 5.9 mmol), solvent (10 mL), 80 bar CO/H2 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio.
g
[PEMim][PF6] = 0.110 g mL−1 (150 equiv.). H = homogeneous.
h MU/Rh = 2500. 1), 80 °C, 6 h.
b Methyl 10-undecenoate conversion.
c
Y
(X) = yield in (X); (Ald) = aldehydes; (Alc) = alcohols; (Iso) = methyl 10-undecenoate isomers; and (Sat) = saturated compound = methyl undecanoate.
d Linear to branched ratio for aldehydes.
e Linear to branched ratio for alcohols.
f Global linear to branched ratio.
g
[PEMim][PF6] = 0.110 g mL−1 (150 equiv.). H = homogeneous.
h MU/Rh = 2500.
|
|||||||||
| 1 | Dodecane | 100 | 15 [0.3] | 75 [1.8] | 9 | 1 | 1.4 | 2.4 | 32 |
| 2 | Heptane | 99 | 22 [0.4] | 68 [1.8] | 8 | 1 | 1.3 | 5.0 | 29 |
| 3 | Cyclohexane | 100 | 40 [0.7] | 54 [2.2] | 6 | 0 | 1.3 | 7.5 | 23 |
| 4 | Pinane | 98 | 59 [0.5] | 32 [1.9] | 7 | 0 | 0.9 | 9.1 | 14 |
| 5 | Toluene | 100 | 71 [1] | 24 [2.0] | 5 | 0 | 1.2 | 19.5 | 10 |
| 6 | Acetonitrile (H) | 98 | 63 [2.0] | 0 | 21 | 14 | 2.0 | — | 0 |
| 7 | Isopropanol (H) | 86 | 42 [1.9] | 0 | 19 | 24 | 1.9 | — | 0 |
| 8 | Me-THF (H) | 26 | 13 [1.7] | 0 | 3 | 10 | 1.7 | — | 0 |
| 9 | Me-THFg (H) | 99 | 67 [1.7] | 0 | 20 | 13 | 1.7 | — | 0 |
| 10 | Noneh (H) | 67 | 42 [2.1] | 0 | 19 | 6 | 2.1 | — | 0 |
Under biphasic conditions, full conversion of the substrate was consistently observed, accompanied by alcohol formation in proportions that correlated with solvent apolarity. Indeed, the highest alcohol yield of 75% was recorded with the [PEMim][PF6]/dodecane combination, corresponding to an alcohol TOF of 32 h−1 (Table 3, entry 1). In contrast, replacing dodecane with toluene led to a substantial drop in alcohol yield (24%) and an accumulation of aldehydes (71%) (Table 3, entry 5). This effect can be attributed to the decreasing apolarity of the organic phase, which correlates with a pronounced increase in Rh leaching (up to 19.5% in toluene). As the organic phase becomes more polar, partial dissolution of the ionic liquid promotes rhodium transfer into the organic layer. Moreover, under reductive hydroformylation conditions in the presence of amines, anionic Rh clusters are known to form.15,55 Although these species are discussed in more detail only in the mechanistic section, it is reasonable to assume that they are more soluble in a more polar organic phase, thereby further contributing to the observed leaching. Given that hydrogenation to alcohols occurs exclusively in the ionic-liquid phase, as shown by the previously performed hot phase-separation test, any Rh that migrates into the organic phase is catalytically ineffective for this step, which directly explains the observed decrease in alcohol formation.
Consistent with this behaviour, earlier studies reported that adding toluene as a co-solvent in the hydroformylation of 1-octene in [BMIM][PF6] negatively affects performance, with a decrease in the l/b ratio attributed to catalyst leaching into the organic phase.44
Unfortunately, employing an apolar solvent derived from biorenewable sources, such as pinane (Table 3, entry 4), consistent with green chemistry principles and aimed at reducing the environmental footprint of the process, did not improve the catalytic performance, yielding only 32% alcohol.
Under homogeneous conditions, the lack of phase separation compromises the sustainability of the system, particularly with regard to catalyst recyclability. Moreover, the absence of alcohol formation is noteworthy. A plausible explanation involves the dilution of the ionic liquid, which reduces the availability of the amine ligand for rhodium coordination. The competition with the solvent may prevent the generation of hydrogenative species, thereby confining the catalytic activity to hydroformylation. Upon increasing the ionic liquid concentration in 2-MeTHF (entry 9), the system remained homogeneous. Under these conditions, hydroformylation activity significantly improved, with aldehyde yield rising from 13% to 67%. However, no alcohol formation was observed, indirectly suggesting that catalyst confinement within the ionic liquid phase is a key factor for generating hydrogenative species. Supporting this hypothesis, an experiment was conducted in biphasic conditions, using [PEMim][PF6] in comparable ligand concentration (i.e. 0.110 g mL−1) in [BMIM][PF6] as the ionic liquid solvent (paragraph IX, Table S9, entry 3, SI). Under these conditions, complete substrate conversion was achieved along with a 22% alcohol yield, confirming the stabilizing role of the ionic liquid in promoting the formation of hydrogenative catalytic species.
To explore the versatility of the catalytic [PEMim][PF6]/heptane system, its performance was tested on more complex substrates, including the Very High Oleic Sunflower Oil (VHOSO), a natural feedstock (paragraph IX, Table S10, SI). Among all substrates tested, methyl 10-undecenoate yielded the highest alcohol TOF of 30 h−1. This can be attributed not only to its terminal double bond, which is more reactive, but also to its superior solubility in the ionic liquid phase, facilitating efficient conversion. In contrast, substrates bearing internal double bonds and exhibiting lower solubility in the ionic liquid phase showed markedly reduced reactivity in hydroformylation and, consequently, in the tandem aldehyde reduction. This was evidenced by the low alcohol yield of 4% for methyl oleate and the complete absence of alcohol formation with VHOSO. Given the lower reactivity observed with other substrates, optimizing the ionic liquid formulation for each specific substrate (polarity and solubility) seems necessary to achieve efficient conversion.
Evaluation of system recyclability and mechanistic insights
The main objective of this study was to investigate the recyclability of the most promising rhodium catalytic system composed of [PEEtOHim][PF6] (highest TOF(alc)) and dodecane (lowest Rh leaching). To evaluate the robustness of the catalyst under the reaction conditions, nine consecutive batch runs were performed using the same ionic liquid phase. At the end of each run (6 h), the system was cooled to room temperature to allow phase separation. As shown in Fig. 4b for the final medium extracted from the autoclave, this occurred instantaneously, with a well-defined interface. This enabled easy recovery of the organic phase at the end of the reaction. Then, the autoclave was subsequently pressurized at 10 bar of CO/H2 mixture and maintained at room temperature overnight.After depressurization the following day, a fresh dodecane phase containing methyl 10-undecenoate was introduced. The autoclave was subsequently sealed and re-pressurized under CO/H2 to initiate the next run. The results are summarized in Fig. 4a.
Although the solvent differed slightly, the high alcohols yield obtained in the previous experiment (Table 2, entry 3) was reproduced in the first run along with complete conversion of MU.
Despite only 3.2% of the initial rhodium content leaching into the organic phase after this 1st run, a significant decrease in catalytic activity was observed, with MU conversion dropping to 78% in the second run. As noted earlier, it is now well established that Rh(acac)(CO)2 undergoes cluster formation under CO/H2 pressure in the presence of amines. Moreover, these rhodium hydrido carbonyl clusters could undergo deprotonation by the amine leading to anionic metallic species that are expected to be highly stabilized within an ionic environment such as [PEEtOHim][PF6]. These species are readily identifiable by their characteristic strong purple coloration26,56 (Fig. 4b), their IR-ATR spectra which revealed characteristic stretching and bending absorption bands corresponding to terminal and bridging carbonyl ligands of clusters and their 13C NMR spectrum exhibiting a clear doublet at 207.6 ppm, attributable to CO-bound rhodium anionic species (paragraph IV, Fig. S65, S66 and S68, SI).15 Therefore, a plausible explanation for the observed decrease in activity between run 1 and run 2 is the reduced rhodium atom utilization which arises from clusterization as molecular complexes transform into clusters during the initial run.
This transformation of the rhodium precursor occurs only in the initial run, after which the system appears to stabilize, as evidenced by alcohols yields around 60% and substrate conversion close to 80%, up to run 5. Between run 5 and 6, a slight decrease in alcohol production with yields around 50% was observed, after which the activity remained stable until the ninth run. It is important to note that the catalyst was maintained under CO/H2 pressure for an entire weekend between run 5 and 6, which may have led to the formation of slightly altered species. Spectroscopic data, however, did not provide evidence for such changes. To exclude the possibility that these rhodium species had evolved into larger nanoparticles responsible for the slight decrease in activity, transmission electron microscopy (TEM) analysis was conducted on a post-reaction medium. While Energy-dispersive X-ray (EDX) mapping revealed a homogeneous distribution of rhodium within the ionic liquid phase, no nanoparticles or large agglomerates were detected, supporting their confinement within the cluster domain (see Fig. S77–S84, SI). Although Rh leaching was detected throughout the recycling experiments (26% of the initial rhodium content was lost after the nine runs), likely contributing to the gradual decline in catalytic activity, the product distribution remained essentially constant, with alcohol selectivity stabilized around 70%. Such behaviour clearly emphasizes the robustness of the catalytically active species. By combining the alcohol production across all runs, a total turnover number (TTN) of 1282 was achieved, underscoring the productivity of the catalytic system. Due to equilibria among rhodium complexes in this system, a cocktail of catalytic species57,58 is likely active, with anionic rhodium clusters playing a pivotal role. To illustrate this, Scheme 2 presents credible equilibria between the different rhodium species present in the ionic liquid. The anionic species (III), (IV) and (V) are considered as “sleeping species” due to the lack of hydride required to hydroformylate the substrate or hydrogenate the intermediate aldehyde. In contrast, the hydrido carbonyl complexes (I) and the hydrido carbonyl species containing amine coordinated ligand (II) correspond to catalytically active rhodium species involved in hydroformylation and reduction, respectively.
 | ||
| Scheme 2 Equilibria between the different Rh species in the ionic liquid phase. The term “[Rh]” may refer either to mononuclear complexes or to cluster species. | ||
Indeed, under the conditions used, the subsequent reduction of the intermediate aldehyde cannot proceed without rhodium coordination to a tertiary amine moiety, which enhances the hydride character of the catalytic species and thereby facilitates aldehyde reduction. Species (I), which are active in the hydroformylation reaction, tend to leach into the organic phase due to their neutral character. They are likely responsible for most of the hydroformylation activity observed during recycling experiments and as evidenced by the previous hot separation test. Moreover, this test also confirmed that rhodium species (II) didn't leach as no hydrogenation activity in the organic phase was detected, suggesting that the active species remained confined to the ionic liquid phase. While this leaching remains low in the optimized system (around 2%), it may nonetheless compromise the long-term sustainability of the process over successive recycling runs. These findings suggest that, while the system exhibits promising potential for recycling, further improvements are required to enhance its long-term stability and reduce rhodium leaching. Future efforts should therefore focus on strategies to stabilize the catalyst and ensure sustained performance over more cycles.
Conclusion
In summary, the present study highlights the efficiency of a catalytic ionic liquid designed with an integrated ligand/solvent architecture, which delivers significantly improved TOF values compared to previously reported imidazolium-based systems. Systematic tuning of the IL through variation of anions and functionalization of the imidazolium ring identified [PEEtOHim][PF6] as the most effective candidate for promoting the reductive hydroformylation of MU. The polar rhodium catalyst, responsible for aldehyde hydrogenation, was immobilized within the ionic liquid phase, underscoring the strong potential of such biphasic systems for selective reductive hydroformylation. Our results suggest that the built-in coordinating functionality of the ionic liquid creates a locally enriched ligand environment around rhodium, thereby stabilizing active species and enhancing catalytic turnover. This interpretation is supported by parallels with other active amino-functionalized solvents used in catalytic biphasic systems, such as alkanolamines26,27 or polymer-supported amines,28,59 which have demonstrated comparable TOF(alc) values and similarly rely on the presence of coordinating amine groups to stabilize active species, modulate phase behavior, and enhance catalytic performance. These observations collectively underline the structural advantages of integrating ligand properties directly into the solvent phase.Nevertheless, rhodium leaching into the organic phase was detected, likely as the classical hydridocarbonyl complex. These rhodium species retained hydroformylation activity but were unable to catalyze aldehyde hydrogenation, likely contributing to the decline in tandem reaction efficiency during recycling.
Looking ahead, the integrated ligand/solvent design principle opens promising avenues for further optimization. One strategy would be to explore the use of biphasic ionic liquid/scCO2 systems. Operating under supercritical CO2 has been shown to significantly limit Rh leaching by enhancing phase separation and reducing metal solubility in the product phase. In such a configuration, careful structural tuning of the ionic liquid and particularly by introducing longer alkyl chains (e.g., octyl or decyl instead of butyl), would be advantageous to prevent excessive extraction of the substrate by scCO2 before it can react in a continuous-flow setup.60 A similar approach has already been reported for a biphasic PEG-amine catalytic phase extracted with scCO2, where the reductive hydroformylation of α-olefins achieved excellent catalyst retention, with only 0.1% rhodium loss over nine consecutive runs, making it one of the most efficient systems reported to date in terms of rhodium immobilization.30 Another complementary approach would be to structurally tune the ionic liquid to strengthen its coordination ability toward rhodium. Introducing bidentate ligands into the IL structure could increase the binding strength of the catalytic phase and thereby reduce metal migration into the organic phase. Finally, immobilizing ionic liquids that contain the catalyst onto solid supports is attractive because it lowers the amount of ionic liquid needed and helps overcome their inherently high viscosity, which is known to restrict H2 and CO diffusion in hydroformylation systems, slowing down the overall catalytic process.61 Such developments could significantly enhance the robustness and industrial relevance of reductive hydroformylation processes.
Experimental
All chemicals were purchased from Fisher Scientific or Merck and were used without prior purification. Pinane solvent was provided by SARL SICO-CHEM and POC SARL (France). Hydroformylation experiments were carried out in a 25 mL autoclave (Parr instrument company) equipped with a mechanical stirrer. All reactions involving metal-aminated IL catalysts were performed under air atmosphere and each run was performed at least twice to ensure reproducibility.The NMR spectra were recorded at 298 K on a Bruker Avance Neo 400 spectrometer operating at 9.4 T field strength (400 MHz for 1H nuclei and 100 MHz for 13C nuclei) equipped with a 5 mm BBFO SmartProbe (1H/19F/31P – 109Ag) and an automatic sample loading system.
Rhodium concentrations were determined through inductively coupled plasma optical emission spectroscopy experiments using a SpectroGreen ICP-OES. Samples composed of an aliquot of the organic phase were digested before ICP experiments in nitric acid via microwave mineralization using a MARS 6 microwave digestion system.
Synthetic procedures
Author contributions
C. G.: investigation, validation, visualization and writing – original draft. J. T.: investigation, formal analysis and visualization. N. K.: formal analysis and visualization. F. C.: formal analysis and visualization. H. B.: validation, formal analysis and visualization. S. T.: validation and supervision E. M.: conceptualization, funding acquisition, validation and supervision. M. F.: conceptualization, funding acquisition, supervision and project administration. All authors contributed to writing– reviewing and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6gc01402j.All other relevant data are available from the corresponding author upon reasonable request.
CCDC 2506183–2506185 ([PEDMim][PF6], [PEMim][PF6] and [PEEtOHim][PF6]) contain the supplementary crystallographic data for this paper.62a–c
Acknowledgements
University of Artois is gratefully acknowledged for its financial support. This research was funded, in whole or in part, by the Hauts-de-France Regional Council and the CPER BiHauts Eco de France project. For the TEM analyses, the Chevreul Institute is acknowledged for its help in the development of this work through the ARCHI-CM project supported by the French Ministère de l'Enseignement Supérieur et de la Recherche, the Hauts-de-France Region, the ERDF program of the European Union and the Métropole Européenne de Lille. We are also grateful to Bastien Léger and Ahmed Addad for the TEM analyses and Anne Marie Blanchenet for the ultramicrotomy experiments. We thank Dr Antoine Piccirilli (La Fabrique Végétale®) for providing pinane. We thank Dominique Prévost for technical assistance.References
- P. Munkajohnpong, C. Kesornpun, S. Buttranon, J. Jaroensuk, N. Weeranoppanant and P. Chaiyen, Biofuels, Bioprod. Biorefin., 2020, 14, 986–1009 CrossRef CAS.
- G. M. Torres, R. Frauenlob, R. Franke and A. Börner, Catal. Sci. Technol., 2015, 5, 34–54 RSC.
- P. Eilbracht, L. Bärfacker, C. Buss, C. Hollmann, B. E. Kitsos-Rzychon, C. L. Kranemann, T. Rische, R. Roggenbuck and A. Schmidt, Chem. Rev., 1999, 99, 3329–3365 CrossRef CAS PubMed.
- D. Konya, K. Q. Almeida Leñero and E. Drent, Organometallics, 2006, 25, 3166–3174 CrossRef CAS.
- L. Wu, I. Fleischer, R. Jackstell, I. Profir, R. Franke and M. Beller, J. Am. Chem. Soc., 2013, 135, 14306–14312 CrossRef CAS PubMed.
- I. Fleischer, K. M. Dyballa, R. Jennerjahn, R. Jackstell, R. Franke, A. Spannenberg and M. Beller, Angew. Chem., 2013, 125, 3021–3025 CrossRef.
- A. Börner and R. Franke, Hydroformylation Fundamentals, Processes, and Applications in Organic Synthesis, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2016 Search PubMed.
- C. S. MacNeil, L. N. Mendelsohn, T. P. Pabst, G. Hierlmeier and P. J. Chirik, J. Am. Chem. Soc., 2022, 144, 19219–19224 CrossRef CAS PubMed.
- J. K. MacDougall, M. C. Simpson, M. J. Green and D. J. Cole-Hamilton, J. Chem. Soc., Dalton Trans., 1996, 1161 RSC.
- R. Franke, D. Selent and A. Börner, Chem. Rev., 2012, 112, 5675–5732 CrossRef CAS PubMed.
- G. M. Torres, R. Frauenlob, R. Franke and A. Börner, Catal. Sci. Technol., 2015, 5, 34 RSC.
- B. Fell and A. Geurts, Chem. Ing. Tech., 1972, 44, 708–712 CrossRef CAS.
- A. T. Jurewicz, L. D. Rollmann and D. D. Whitehurst, in Homogeneous Catalysis—II, American Chemical Society, 1974, vol. 132, pp. 240–251 Search PubMed.
- J. Ternel, A. Lopes, M. Sauthier, C. Buffe, V. Wiatz, H. Bricout, S. Tilloy and E. Monflier, Molecules, 2021, 26, 7322 CrossRef CAS PubMed.
- T. Rösler, K. R. Ehmann, K. Köhnke, M. Leutzsch, N. Wessel, A. J. Vorholt and W. Leitner, J. Catal., 2021, 400, 234–243 CrossRef.
- D. N. Gorbunov, M. V. Nenasheva, M. V. Terenina, Y. Kardasheva, E. R. Naranov, A. L. Bugaev, A. V. Soldatov, A. L. Maximov, S. Tilloy, E. Monflier and E. A. Karakhanov, Appl. Catal., A, 2022, 647, 118891 CrossRef CAS.
- A. Zouhair, M. Ferreira, H. Bricout, N. Kania, B. Léger, J. Ternel, A. Ponchel, M. Lahcini, S. Tilloy and E. Monflier, Catal. Sci. Technol., 2026, 16, 2009–2020 RSC.
- T. Mizoroki, M. Kioka, M. Suzuki, S. Sakatani, A. Okumura and K. Maruya, Bull. Chem. Soc. Jpn., 1984, 57, 577–578 CrossRef CAS.
- D. L. Hunter, S. E. Moore, R. A. Dubois and P. E. Garrou, Appl. Catal., 1985, 19, 275–285 CrossRef CAS.
- D. L. Hunter, S. E. Moore, P. E. Garrou and R. A. Dubois, Appl. Catal., 1985, 19, 259–273 CrossRef CAS.
- L. Alvila, T. A. Pakkanen, T. T. Pakkanen and O. Krause, J. Mol. Catal., 1992, 71, 281–290 CrossRef CAS.
- L. L. W. Cheung, G. Vasapollo and H. Alper, Adv. Synth. Catal., 2012, 354, 2019–2022 CrossRef CAS.
- T. Vanbésien, E. Monflier and F. Hapiot, Green Chem., 2016, 18, 6687–6694 RSC.
- S. Fuchs, D. Lichte, M. Dittmar, G. Meier, H. Strutz, A. Behr and A. J. Vorholt, ChemCatChem, 2017, 9, 1436–1441 CrossRef CAS.
- C. Becquet, F. Berche, H. Bricout, E. Monflier and S. Tilloy, ACS Sustainable Chem. Eng., 2021, 9, 9444–9454 CrossRef CAS.
- M. Nenasheva, D. Gorbunov, M. Karasaeva, A. Maximov and E. Karakhanov, Mol. Catal., 2021, 516, 112010 CAS.
- S. Püschel, E. Hammami, T. Rösler, K. R. Ehmann, A. J. Vorholt and W. Leitner, Catal. Sci. Technol., 2022, 12, 728–736 RSC.
- D. N. Gorbunov, M. V. Nenasheva, E. A. Kuvandykova, S. V. Kardashev and E. A. Karakhanov, Pet. Chem., 2023, 63, 594–606 CrossRef CAS.
- S. Püschel, J. Sadowski, T. Rösler, K. R. Ehmann, A. J. Vorholt and W. Leitner, ACS Sustainable Chem. Eng., 2022, 10, 3749–3756 CrossRef PubMed.
- T. Rösler, J. Betting, S. Püschel, A. J. Vorholt and W. Leitner, Green Chem., 2022, 24, 6578–6588 RSC.
- W. Abdallah, J. Ternel, M. Ferreira, H. Bricout, S. Tilloy and E. Monflier, Coord. Chem. Rev., 2026, 557, 217749 CrossRef CAS.
- M. Haumann and A. Riisager, Chem. Rev., 2008, 108, 1474–1497 CrossRef CAS PubMed.
- J. Dupont, B. C. Leal, P. Lozano, A. L. Monteiro, P. Migowski and J. D. Scholten, Chem. Rev., 2024, 124, 5227–5420 CrossRef CAS PubMed.
- J. F. Knifton, J. Mol. Catal., 1987, 43, 65–77 CrossRef CAS.
- A. El Mouat, C. Becquet, J. Ternel, M. Ferreira, H. Bricout, E. Monflier, M. Lahcini and S. Tilloy, ACS Sustainable Chem. Eng., 2022, 10, 11310–11319 CrossRef CAS.
- A. El Mouat, W. Abdallah, J. Ternel, M. Ferreira, H. Bricout, A. J. Vorholt, H. Stieber, S. Stoertte, E. Monflier, M. Lahcini and S. Tilloy, ChemSusChem, 2025, 18, e202401384 CrossRef CAS PubMed.
- S. Chen, Y. Wang, W. Yao, X. Zhao, G. Vo-Thanh and Y. Liu, J. Mol. Catal. A: Chem., 2013, 378, 293–298 CrossRef CAS.
- B. N. Leidecker, D. P. Fuentes, C. Wei, M. Sawall, K. Neymeyr, R. Franke, A. Börner and C. Kubis, New J. Chem., 2024, 48, 18365–18375 RSC.
- Y. Jiao, M. S. Torne, J. Gracia, J. W. (Hans) Niemantsverdriet and P. W. N. M. van Leeuwen, Catal. Sci. Technol., 2017, 7, 1404–1414 RSC.
- C. P. Mehnert, R. A. Cook, N. C. Dispenziere and E. J. Mozeleski, Polyhedron, 2004, 23, 2679–2688 CrossRef CAS.
- Y. Chauvin, L. Mussmann and H. Olivier, Angew. Chem., Int. Ed. Engl., 1996, 34, 2698–2700 CrossRef.
- F. Favre, H. Olivier-Bourbigou, D. Commereuc and L. Saussine, Chem. Commun., 2001, 2, 1360–1361 RSC.
- B. Wang, L. Qin, T. Mu, Z. Xue and G. Gao, Chem. Rev., 2017, 117, 7113–7131 CrossRef CAS PubMed.
- J. Dupont, S. M. Silva and R. F. de Souza, Catal. Lett., 2001, 77, 131–133 CrossRef CAS.
- A. Sharma, C. J. Lebigue, R. M. Deshpande, A. A. Kelkar and H. Delmas, Ind. Eng. Chem. Res., 2010, 49, 10698–10706 CrossRef CAS.
- K. Kaneda, H. Kuwahara and T. Imanaka, J. Mol. Catal., 1992, 72, L27–L30 CrossRef CAS.
- L. Diab, T. Šmejkal, J. Geier and B. Breit, Angew. Chem., Int. Ed., 2009, 48, 8022–8026 CrossRef CAS PubMed.
- T. L. Amyes, S. T. Diver, J. P. Richard, F. M. Rivas and K. Toth, J. Am. Chem. Soc., 2004, 126, 4366–4374 CrossRef CAS PubMed.
- S. Raiguel, W. Dehaen and K. Binnemans, Green Chem., 2020, 22, 5225–5252 RSC.
- K. K. Laali, A. Jamalian and G. L. Borosky, J. Phys. Org. Chem., 2016, 29, 346–351 CrossRef CAS.
- W. Li, Z. Zhang, B. Han, S. Hu, J. Song, Y. Xie and X. Zhou, Green Chem., 2008, 10, 1142 RSC.
- P. McNeice, P. C. Marr and A. C. Marr, Catal. Sci. Technol., 2021, 11, 726–741 RSC.
- F. Rived, M. Rosés and E. Bosch, Anal. Chim. Acta, 1998, 374, 309–324 CrossRef CAS.
- W. Abdallah, M. Ferreira, S. Laclef, D. Mathiron, S. Pilard, R. Froidevaux, M. D. S. L. Moreira, H. Bricout, E. Monflier and S. Tilloy, Eur. J. Lipid Sci. Technol., 2025, 127, 1–12 CrossRef.
- P. Chini, G. Longoni and V. G. Albano, in Advances in Organometallic Chemistry, 1976, vol. 14, pp. 285–344 Search PubMed.
- D. Gorbunov, M. Nenasheva, A. Maximov and E. Karakhanov, Appl. Catal., A, 2024, 670, 119538 CrossRef CAS.
- A. L. Maximov and M. P. Egorov, Chin. J. Catal., 2025, 78, 7–24 CrossRef CAS.
- A. S. Kashin and V. P. Ananikov, J. Org. Chem., 2013, 78, 11117–11125 CrossRef CAS PubMed.
- M. V. Nenasheva, B. O. Protsenko, K. D. Kulaev, E. R. Naranov, A. L. Trigub, A. L. Maximov, A. A. Guda and D. N. Gorbunov, Mol. Catal., 2026, 590, 115654 CAS.
- P. B. Webb, M. F. Sellin, T. E. Kunene, S. Williamson, A. M. Z. Slawin and D. J. Cole-Hamilton, J. Am. Chem. Soc., 2003, 125, 15577–15588 CrossRef CAS PubMed.
- C. Van Doorslaer, J. Wahlen, P. Mertens, K. Binnemans and D. De Vos, Dalton Trans., 2010, 39, 8377 RSC.
- (a) CCDC 2506183: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3wmc; (b) CCDC 2506184: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3wnd; (c) CCDC 2506185: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3wpf.
| This journal is © The Royal Society of Chemistry 2026 |