Ionic atmosphere and temperature field–promoted dissociation and phase reconstruction of bastnaesite
Qionghua
Xie
abcde,
Yifan
Hao
acde,
Guocheng
Yao
acde,
Cong
Wang
b and
Liangshi
Wang
 *acde
*acde
aNational Engineering Research Center for Environment-friendly Metallurgy in Producing Premium Non-ferrous Metals, China GRINM Group Co., Ltd, Beijing 101407, China
bSchool of Metallurgy, Northeastern University, Shenyang, Liaoning 110819, China
cBeijing Engineering Research Center of Strategic Nonferrous Metals Green Manufacturing Technology, GRINM Resources and Environment Tech. Co., Ltd, Beijing 101407, China. E-mail: wangliangshi@grinm.com
dGeneral Research Institute for Nonferrous Metals, Beijing 100088, China
eGRIMAT Engineering Institute Co., Ltd, Beijing 101407, China
First published on 12th April 2026
Abstract
Bastnaesite serves as the primary global source mineral for light rare earth elements (LREEs). However, achieving green metallurgy in the rare earth industry still faces severe challenges, including resource wastage, environmental pollution induced by associated fluorine, and low comprehensive recovery rates of REEs. To address the critical issues of green and efficient REE extraction from bastnaesite and the comprehensive utilization of associated fluorine resources, this study proposes a novel coordinated leaching–selective precipitation strategy by introducing an SO42− ionic atmosphere and a temperature field. The results demonstrate that during the leaching process, SO42− effectively stabilizes [CeFx]4−x complexes by forming an ionic atmosphere, thereby retarding the reduction of Ce4+ and the formation of REF3. This significantly enhances the leaching efficiency of both non-cerium rare earth elements and total rare earth oxides (TREO leaching rate = 92%). Simultaneously, leveraging phase reconstruction under the temperature field enables regulation of the precipitation transition from [CeFx]4−x → CeF3, achieving a fluorine recovery rate >98% and yielding high-purity CeF3 (D50 = 1.3–1.9 μm). This process achieves highly efficient separation and extraction of rare earth elements, cerium, and associated fluorine. The proposed ionic atmosphere model provides a reference for developing short-process, green, and clean bastnaesite metallurgy, advancing the sustainable utilization of both rare earth and fluorine resources.
Green foundation1. This work achieves co-recovery of REEs and fluorine from bastnaesite via a synergistic leaching–selective precipitation strategy under SO42− ionic atmospheres, resolving persistent fluorine pollution challenges. The short-flow process eliminates alkali consumption, enabling zero fluorine-containing wastewater discharge while advancing green chemistry in rare earth metallurgy.2. SO42− ionic atmospheres stabilize [CeFx]4−x complexes during HCl leaching, inhibiting Ce4+ reduction and REF3 precipitation while boosting total REE recovery by 12%. Temperature-controlled regulation converts >98% of fluorine into size-controllable CeF3 products, transforming pollutants into functional materials. 3. The established controllable ionic atmosphere model provides a universal framework for sustainable mineral processing. Future enhancements through deeper ionic mechanism studies, high-value product engineering, and circular industrial chains will further reduce energy/carbon footprints, advancing toward a zero-waste, zero-pollution paradigm in rare earth metallurgy. |
Introduction
Rare earth elements (REEs) are regarded as essential for the advancement of clean energy technologies and the attainment of net-zero emissions, owing to their distinctive magneto-optical and electro-optical characteristics.1–4 Global supply risks of REEs have intensified due to intensive extraction, sustained consumption growth, and geopolitical tensions.5–8 REEs have therefore become strategically contested mineral resources globally. Bastnaesite dominates global REE reserves and production, accounting for ∼70% of worldwide REE output. The dominant extraction process—roast–acid leach treatment—is extensively adopted in major producing nations (e.g., China and USA) due to its operational simplicity, moderate capital investment, and cost efficiency.9 However, the high fluorine content in bastnaesite renders its metallurgical processing energy- and pollution-intensive.10–12 During metallurgical processing, fluorine is converted into HF gas, REF3 precipitates, and fluoride anions, severely impeding REE separation and causing substantial environmental degradation with compromised economic efficiency. These fluorine-induced low rare earth recovery and eco-hazard issues are recognized as critical constraints for bastnaesite metallurgy.13,14 Consequently, the efficient extraction of REEs from bastnaesite, coupled with the recovery of associated fluorine resources, has become essential. This integration is crucial for developing a sustainable rare earth industry and advancing the circular economy in this sector.15–17The recycling of fluorine, as a fundamental raw material for fluorine-based chemical products, plays a crucial role in mitigating the depletion of strategic resources and facilitating the high-value utilization of these products. To effectively recover REEs and fluorine from bastnaesite, researchers have focused their efforts on the research and development of environmentally sustainable smelting processes for bastnaesite. As illustrated in Fig. 1, three predominant methodologies have been developed based on alterations in fluorine-bearing phase morphology during bastnaesite processing: (i) fluorine gasification removal via roasting with water vapor/concentrated H2SO4;18–20 (ii) roasting–fluorine fixation using Ca/Mg/Al additives;21–23 and (iii) complexation leaching with Al-salts/H3BO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24–27 and co-extraction of Ce4+ and F− from the leachates.28–30 Although these processes recover fluorine as fluorite or cryolite, they fail to achieve high-value fluorine utilization. Furthermore, they exhibit increased complexity, excessive chemical consumption, and constrained economic–environmental viability, thereby limiting industrial scalability. Consequently, developing a novel short-flow process for efficient co-recovery of REEs and associated fluorine from bastnaesite is imperative.
24–27 and co-extraction of Ce4+ and F− from the leachates.28–30 Although these processes recover fluorine as fluorite or cryolite, they fail to achieve high-value fluorine utilization. Furthermore, they exhibit increased complexity, excessive chemical consumption, and constrained economic–environmental viability, thereby limiting industrial scalability. Consequently, developing a novel short-flow process for efficient co-recovery of REEs and associated fluorine from bastnaesite is imperative.
 | ||
| Fig. 1 Metallurgical process of bastnaesite and the current status of the comprehensive utilisation process of fluorine resources. | ||
Leveraging the strong coordination between rare earth and fluorine enables efficient co-recovery of REEs and fluorine from bastnaesite, offering a streamlined, additive-free, and environmentally benign process. Building on the coordination chemistry of [CeFx]4−x in HCl leaching systems, our research team has developed a novel technology that integrates low-temperature roasting, coordinated leaching, and reductive precipitation for the recovery of REEs and fluorine. This approach significantly enhances the yields of both non-cerium and total REEs, while simultaneously recovering fluorine as high-value CeF3.31 However, [CeFx]4−x is prone to reduction by Cl− ions during HCl leaching, compromising its stability (eqn (1)–(7)). Once destabilized, free F− ions readily combine with RE3+ to form REF3 solids, which partition into the slag (eqn (8)) and consequently reduce the leaching efficiency of non-cerium REEs.
| CeO2 + 4H+ → Ce4+ + 2H2O | (1) |
| xREOF + 2xH+ + Ce4+ → xRE3+ + [CeFx](4−x) + H2O | (2) |
| CeO2 + 4H+ + xF− → [CeFx](4−x) + 2H2O | (3) |
| Ce4+ + xH+ + xF− → [CeFx](4−x) + xH+ | (4) |
| [CeFx](4−x) → Ce4+ + xF− | (5) |
| Ce4+ + Cl− → Ce3+ + Cl2↑ | (6) |
| 3[CeFx](4−x) + 3Cl− → xCeF3↓ + (3 − x)Ce3+ + 1.5Cl2↑ | (7) |
| RE3+ + 3F− → REF3↓ | (8) |
The key to solving this problem lies in achieving “targeted protection” of the [CeFx]4−x complex at the molecular scale. This requires simultaneously blocking the approach of Cl− and preserving the Ce4+–F− coordination structure. To this end, this study proposes an ionic atmosphere regulation mechanism. High-charge-density counterions are introduced to form an ordered and dynamic electrostatic shielding layer around [CeFx]4−x through electrostatic attraction. This ionic atmosphere does not rely on strong chemical bonding. Instead, it effectively prevents Cl− from attacking and reducing Ce4+ through both steric hindrance and electrostatic repulsion. It also inhibits the formation of REF3 precipitates. As a result, efficient rare earth leaching and in situ fluorine fixation are achieved. Based on electronegativity principles and Hard–Soft Acid–Base (HSAB) theory, SO42− satisfies the coordination requirements for ionic atmosphere construction, demonstrating superior stability, applicability, and environmental compatibility among hard-base ions.32 Our previous work has demonstrated that [CeFx]4−x can exist stably within a sulfuric acid leaching system, indicating a beneficial role of SO42− in maintaining their stability.33 However, mechanisms for maintaining [CeFx]4−x–Cl− stability and achieving controlled conversion of [CeFx]4−x to CeF3 in HCl systems remain unexplored.
This study enhances bastnaesite dissociation via coordination chemistry by introducing SO42− during HCl leaching to establish an ionic atmosphere. This environment is crucial for maintaining the macroscopic kinetic stability of the [CeFx]4−x–Cl− system, intensifying leaching of valuable REEs, cerium, and fluorine. Subsequent phase reconstruction through selective precipitation utilizes temperature field effects within the [CeFx]4−x–SO42−–Cl− system. These effects destabilize [CeFx]4−x, thereby accelerating the transformation process from [CeFx]4−x to CeF3. The integrated short-flow process achieves efficient separation of high-value REEs, cerium, and associated fluorine. This study mitigates critical constraints in bastnaesite metallurgy such as ineffective fluorine resource recovery, refractory wastewater treatment, and substantial cerium resource losses.
Experimental
Reagents and materials
The bastnaesite concentrate used in this study originated from Mianning, Sichuan. The mineralogical analysis identified bastnaesite and fluorite as the predominant phases, accompanied by minor amounts of calcite, parisite, and quartz (Fig. 2(a)). REEs were predominantly enriched in bastnaesite, while fluorine distribution between bastnaesite and fluorite approximated a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (Fig. 2(b)). Differential thermal–thermogravimetric analysis under an air atmosphere demonstrated bastnaesite near-complete decomposition at 458.6 °C (Fig. 2(c)). Roasting the bastnaesite at 500 °C for 2 hours in a muffle furnace produced roasted bastnaesite, with the roasting process generating almost no gaseous fluoride emission.34 Phase compositions and chemical transformations pre-/post-roasting are shown in Fig. 2(d–f). Other reagents were purchased from Shanghai Macklin Biochemical Co., Ltd. All the reagents were of analytical purity and did not require further purification.
1 ratio (Fig. 2(b)). Differential thermal–thermogravimetric analysis under an air atmosphere demonstrated bastnaesite near-complete decomposition at 458.6 °C (Fig. 2(c)). Roasting the bastnaesite at 500 °C for 2 hours in a muffle furnace produced roasted bastnaesite, with the roasting process generating almost no gaseous fluoride emission.34 Phase compositions and chemical transformations pre-/post-roasting are shown in Fig. 2(d–f). Other reagents were purchased from Shanghai Macklin Biochemical Co., Ltd. All the reagents were of analytical purity and did not require further purification.
Experimental procedure
Leaching and deposition experiments for roasted bastnaesite were conducted in a thermostatic water bath. Precisely 4 mol L−1 HCl was pipetted into a 250 mL three-necked flask, followed by addition of a coordination agent (5 wt% relative to mineral mass). The flask was immersed in a 25 °C water bath with agitation at 250 rpm. Subsequently, 30 g of roasted bastnaesite was gradually added via a powder funnel with timing initiation. After 2 hours of leaching, the mixture was subjected to solid–liquid separation using a Büchner funnel, and the leachate volume was recorded. A 100 mL leachate aliquot was transferred to a 250 mL beaker, sealed with parafilm, and covered with a Petri dish in a 90 °C water bath for static deposit. Following solid–liquid separation, the deposited solids were oven-dried at 100 °C to constant weight for storage. The leaching rate and deposition rate were calculated as follows: | (9) |
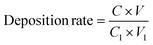 | (10) |
Analytical methods
An inductively coupled plasma emission spectrometer (Agilent 5110, USA) was used to determine the concentration of rare earth ions and impurity ions in solution. The Ce(IV) and total cerium (TCe) contents in the solution were titrated with ammonium ferrous sulfate standard solution using sodium diphenylamine sulfonate as an indicator. Fluoride ion concentration in the solution was measured using the fluoride ion selective electrode method. The component content and phase composition of the minerals and sediments were examined using an X-ray fluorescence spectrometer (SHIMADZU XRF-1800, Japan) and an X-ray diffractometer (Rigaku Ultima IV, Japan), and the particle size distribution of the sediments was determined using a laser particle sizer (Malvern Mastersizer 2000, UK). The morphology of the sediments was analysed using a scanning electron microscope (ZEISS Gemini SEM 300, Germany) at a magnification of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times.
000 times.
Results and discussion
Reaction thermodynamics and theoretical foundation
The chemical behavior of roasted bastnaesite during hydrochloric acid leaching displays significant complexity. To clarify the thermodynamic properties governing the reactions occurring in bastnaesite hydrochloric acid leaching, the variation of the Gibbs free energy change (ΔG) with temperature was plotted, as presented in Fig. 3(a). While the acid leaching of bastnaesite is exothermic and the ΔG values for trivalent rare earth oxides increase only slightly with temperature, CeO2 remains refractory under conventional conditions (ΔG > 0). In contrast, two key reactions are thermodynamically favored at higher temperatures: the precipitation of REF3 from RE3+ and F−, and the reduction of Ce4+ by Cl−. This indicates that the conversion of Ce4+ to Ce3+ and the formation of REF3 are both enhanced at higher temperatures. As an endothermic reaction, “RE3+ + 3F− → REF3” shifts the equilibrium toward precipitation with rising temperature. This is further supported by the strong temperature dependence of the solubility product (Ksp) of rare earth fluorides. For example, the Ksp of CeF3 decreases markedly from 8.0 × 10−16 at 25 °C to 9.3 × 10−18 at 100 °C, and further to 1.6 × 10−20 at 200 °C. This trend indicates that higher temperatures promote the formation of stable CeF3 precipitates. Notably, at equivalent temperatures, La3+ and Ce3+ exhibit stronger F− affinity than other higher-value light REEs (e.g., Pr and Nd). This suggests that by carefully controlling the leaching conditions, it is possible to suppress the fluorination of more valuable RREs in solutions dominated by Ce3+ and La3+.As the predominant rare earth element in bastnaesite, efficient CeO2 leaching is crucial for enhancing total rare earth extraction, particularly high-value Pr and Nd recovery. The strong bonding affinity between cerium and fluorine facilitates the coordination of Ce4+ and F− in the leaching solution, resulting in the formation of the cerium–fluorine complexes ([CeFx]4−x). Although both protonation of fluoride ions and their coordination with Ce4+ occur during hydrochloric acid leaching, the HF generated from protonation also undergoes coordination with the abundantly leached Ce4+ (eqn (3) and (4)). The Eh–pH diagrams of the Ce–Cl–F–H2O system (Fig. 3(b), SI S1) define the stability regions of [CeFx]4−x under highly acidic and oxidizing conditions, with [CeF3]+ exhibiting superior thermodynamic stability. However, due to the high concentration of Cl− in the system, [CeF3]+ can only persist under extremely acidic conditions (pH < −2), where the redox potential of E(Cl2/Cl−) exceeds that of E([CeF3]+/CeF3). The temperature-dependent potential profiles (Fig. 3(c)) show that E(Cl2/Cl−) is more sensitive to temperature than E(Ce4+/Ce3+). This difference increases the ΔE at higher temperatures, thereby accelerating the reduction of Ce4+ by Cl− and, in turn, diminishing the stabilization region of [CeFx]4−x. The ΔG (ΔG = G([CeF3]+)−G(Ce4+) − 3G(F−)) for the reaction “Ce4+ + 3F− ⇌ [CeF3]+” across temperatures (Fig. 3(d)) further evidence thermal field effects: [CeFx]4−x stabilizes at low temperatures but undergoes accelerated dissociation when heated. Collectively, acidity, potential, and temperature govern [CeFx]4−x generation and dissociation dynamics in leaching systems.
However, the stable form of [CeFx]4−x in the hydrochloric acid system has yet to be documented. The molecular structure of the [CeFx]4−x complexes was optimized using Density Functional Theory (DFT), as detailed in SI S2. Electrostatic interactions and molecular surface properties of the [CeFx]4−x molecules were predicted through quantitative analyses employing an efficient algorithm implemented in Multiwfn.35–37 The van der Waals surface electrostatic potential (ESP), average localised ionisation energy (ALIE), orbital-weighted Fukui functions, and orbital-weighted dual descriptors (Fig. 3(e)) alongside binding energies (Fig. 3(f)) of various [CeFx]4−x molecules demonstrate [CeF3]+ as the primary stable form during the coordinated leaching process. Moreover, the differences in electronic structure and coordination behavior between Ce and high-value rare earth elements (e.g., Pr and Nd) govern their distribution during leaching–selective precipitation. DFT calculations reveal that [PrFx]4−x exhibits lower spatial stability than [CeFx]4−x, while the binding energy difference between PrF3 and CeF3 is minimal, allowing Pr co-precipitation to be suppressed via aging control. Moreover, tetravalent rare earth fluorides show higher stability, with F− preferentially coordinating to tetravalent species, thereby enhancing leaching and enabling in situ fluorine fixation.
Coordinated leaching of bastnaesite under an SO42− ionic atmosphere and mechanism
The kinetic stability of the [CeFx]4−x–Cl− system is manifested macroscopically as an increase in the concentrations of the stable Ce4+, F− and RE3+ in leachates. Fig. 4(a) demonstrates that SO42− introduction significantly elevates Ce4+ and F− contents in the leachate, with F− enhancement being more pronounced. This confirms SO42−'s efficacy in stabilizing [CeFx]4−x, intensifying Ce4+ and F− leaching, sequestering fluorine as [CeFx]4−x ligands, and minimizing F−–RE3+ contact. Simultaneously, the F− concentration in the leaching solution differs depending on whether SO42− is introduced via H2SO4 or sulfates. H2SO4 marginally increases H+ concentration, promoting bastnaesite dissociation and enhancing [CeFx]4−x stability and F− concentration. The comparison in Table S2 reveals that SO42−—not H+—dominates system behavior during sulfuric acid addition. In addition, the introduction of SO42− boosts REE ion leaching (Fig. 4(b)), attributed to enhanced leaching of high-value REEs (e.g., Pr and Nd) and valuable components (Ce/F) upon [CeFx]4−x–Cl− stabilization. SO42− coordinated leaching systems effectively suppress the formation of colloidal REF3, yielding low-viscosity residues with rapid solid–liquid separation. Except for Ba (precipitated as BaSO4), other impurity ions show negligible changes in leaching (Fig. 4(c)). Total rare earth oxide (TREO) leaching rates calculated from residue (Fig. 4(g)) and leachate compositions exhibit consistency, with high-value REEs (Pr/Nd) comprising <1% of residues—merely 3–5% of total residue REEs. The dominant residue elements F and Ca indicate undissolved CaF2 phases from bastnaesite, amenable to subsequent beneficiation recovery.
The leaching solutions obtained from different leaching systems were subjected to aging treatment (Fig. 4(d and e)). The results demonstrate that Ce4+ and F− in the leachate can be efficiently deposited to form CeF3 under an SO42− ionic atmosphere. After the aging treatment, there was almost no Ce4+ in the leaching solution, and the system redox potential decreased (Fig. 4(f)). The SO42− coordinated system achieved >98.5% fluorine deposition efficiency. Subsequently, it is expected to achieve complete recovery of fluorine in the leaching solution by adjusting factors such as acidity and potential.
Quantitative trajectory analysis of intra-systemic ions yielded the Ce4+-centered radial distribution function (RDF), visualizing radial distances from Ce4+ to surrounding ions and their density enrichment (relative density) at specific distances. The RDF profiles of the Ce4+–Cl−–F−–SO42− system (Fig. 5(c)) reveal a distinct concentric ordering around Ce4+. The ions are arranged with F− in the innermost shell, SO42− in the intermediate shell, and Cl− in the outermost shell. Correspondingly, F− shows the highest density enrichment, followed by SO42−, while Cl− exhibits minimal enrichment. Despite greater Cl− population density (Fig. S2), SO42− can closely surround [CeFx]4−x to hinder the contact between Cl− and Ce4+.
Furthermore, Raman spectroscopy analysis was performed on leachates from both the HCl-only and SO42−-coordinated leaching systems. As shown in Fig. 5(d), the pre-leaching solutions of the sulfate-coordinated systems all exhibit a broad HSO4− peak at approximately 1060 cm−1, indicating that most SO42− species are protonated into dynamically disordered HSO4− prior to leaching. Slight differences in acidity arising from the sulfate source (H2SO4 or Na2SO4) lead to variations in the degree of protonation, as reflected by the presence of a weak SO42− shoulder peak at ∼980 cm−1 in the HCl + Na2SO4 pre-leaching solution. After leaching bastnaesite with the HCl + H2SO4/Na2SO4 solutions, the post-leaching spectra show a weakening of the HSO4− peak at ∼1060 cm−1, while the SO42− characteristic peaks in the 980–997 cm−1 region become stronger and sharper. Meanwhile, a distinct and well-defined SO42− peak appears at ∼506 cm−1.38,39 Additionally, clear peaks are observed at 291 cm−1 and 330 cm−1, attributable to Ce–F/Ce–F–S vibrations.39–41 These spectral changes indicate that acid consumption during rare earth dissolution shifts the sulfate protonation equilibrium, driving the conversion of HSO4− back to SO42−. The SO42− and HSO4− peaks in the post-leaching solution become sharper and more pronounced. This suggests that they are no longer disordered but are dynamically and relatively ordered “anchored” around the positively charged [CeFx]4−x complexes via electrostatic interactions. This observation aligns well with the outer-sphere coordination model of the ionic atmosphere. Meanwhile, the high charge of Ce4+ influences the electron cloud distribution of SO42− through its spatial electric field. This results in a slight shift of the characteristic Raman peak of SO42− from ∼980 cm−1 to ∼997 cm−1, suggesting that the involvement of sulfate also involves partial chemical interactions, such as polarization effects and hydrogen bonding. In contrast, the Ce–F peaks at 291 cm−1 and 330 cm−1 are very weak or absent in the HCl-only post-leaching solution. This confirms the instability of [CeFx]4−x complexes without the protective ionic atmosphere.
The Raman spectroscopy analysis further validates the rationality of the ionic atmosphere mechanism. During the coordinated leaching process, HSO4− is the dominant species of sulfate. As H+ is consumed by mineral dissolution, HSO4− deprotonates to form SO42−. In this dynamic equilibrium, HSO4− acts as a reservoir that continuously supplies SO42−. Deprotonated SO42 constructs a dynamic and ordered ionic atmosphere protective layer around the [CeFx]4−x complex, primarily through electrostatic interactions (Fig. 5(e)). This protective layer effectively prevents Cl− ions from approaching and reductively attacking the Ce4+ center through steric hindrance and electrostatic repulsion, thereby kinetically stabilizing the high-valence state of cerium. At the same time, this protective ionic atmosphere confines F− ions around Ce4+ and within the [CeFx]4−x complex, hindering the release of F− and its subsequent contact with RE3+ ions in the solution, thus preventing the formation of REF3 during the leaching process. Furthermore, weak chemical interactions assist in enhancing the density and stability of the ionic atmosphere, allowing SO42− to subtly maintain the stability of [CeFx]4−x without engaging in strong coordination competition with Ce4+. Ultimately, the macroscopic kinetic stability of the [CeFx]4−x–Cl− system promotes the leaching of high-valence REEs (e.g., Pr and Nd), cerium, and fluorine.
Precipitation–dissolution equilibrium of the CeF3 precipitation process
Aging treatment of the bastnaesite acid leachate aims to promote the [CeFx]4−x → CeF3 transformation, enabling efficient Ce4+ and F− recovery as CeF3 while retaining high-value REEs (e.g., Pr/Nd) in solution. To investigate precipitation–dissolution equilibria within the Ce4+–F−–SO42−–Cl− system under an SO42− ionic atmosphere and varying temperature fields, the acid leaching solution from SO42− coordinated leaching was subjected to static aging at 25 °C and 90 °C for varying durations, resulting in temperature and time-dependent supernatant–precipitate formation. Solution evolution during precipitation (Fig. S3) macroscopically demonstrates thermal field-enhanced [CeFx]4−x destabilization, accelerated [CeFx]4−x → CeF3 kinetics, and rare earth fluoride nuclei growth–agglomeration. Post-aging ion concentrations and deposition rates across temperatures (Fig. 6) further elucidate the thermal field-driven phase reconstruction process. Throughout the aging process, the ionic atmosphere environment of the Ce4+–F−–SO42−–Cl− system was progressively disrupted, leading to the dissociation of [CeFx]4−x or its direct reduction to Ce3+ by Cl−. Concurrently, free F− ions combined with RE3+ to form precipitates, resulting in a marked reduction in the concentrations of F− and Ce4+ within the system, as well as a decrease in the overall system potential.Deposition kinetics of cerium, non-cerium REEs, and impurity ions exhibit temperature-dependent disparities. At 25 °C (Fig. 6(a and c)), Ce4+ reduction and F− deposition proceed slowly and incompletely. Conversely, at 90 °C (Fig. 6(b and e)), near-complete Ce4+ reduction and F− deposition (>99%) occur within 20 minutes, confirming thermal field-accelerated [CeFx]4−x → CeF3 transformation. Although cerium dominates precipitation across temperatures, the 90 °C thermal field widens the deposition separation window between low-value (La/Ce) and high-value REEs (Pr/Nd). It is anticipated that controllable separation of La/Ce from high-value Pr/Nd can be achieved through further regulation of the aging process. Impurity ions, except for calcium, show no obvious change over time under different temperature fields (Fig. 6(d and f)), whereas elevated Ca2+ deposition at high temperatures risks compromising rare earth fluoride purity.
Compositional, phase, granulometric, and morphological analyses were performed on precipitates obtained under varying thermal fields and aging durations. Sediments predominantly contained Ce, La, and F elements (Tables S3 and S4). At 25 °C, the precipitates exhibited minimal Nd and negligible Pr content (Table S3), whereas processing at 90 °C resulted in increased Nd deposition and minor Pr co-precipitation (∼1% of total REEs, Table S4). The precipitation–dissolution equilibrium inherent to the deposition process results in fluctuations and increases in the content of rare-earth fluorides. Furthermore, Tables S3 and S4 indicate that metallic elements other than Ce, such as La, Nd, Ca, and Sr, persist in the CeF3 product. These impurities originate from the leachate of bastnaesite and undergo co-precipitation during the aging process. Due to the highly similar physicochemical properties of REEs, they readily form REF3 precipitates with low solubility products with fluoride ions during aging. While elements like Ca and Sr have a weaker affinity for fluoride compared to REEs, they may compete for F− in the initial precipitation stage, forming fluoride impurities. However, as the precipitation–dissolution dynamic progresses throughout aging, the content of these non-rare earth impurities fluctuates and decreases. The minimization of these impurities can be achieved by controlling the aging process parameters, including pH, system potential, aging time, and temperature.
The primary phases of the sediments are identified as rare earth fluorides, specifically CeF3 and LaF3 (Fig. 7(a and b)). However, the migration of impurities such as calcium (Ca) and aluminum (Al) into the fluorinated rare earth phases was evident following prolonged aging at 25 °C. The particle size distribution (Fig. 7(c and d)) and morphological alterations (Fig. S4 and S5) of the sediments demonstrate the modulation effects of temperature fields and aging time. The particle size of the sediments across different temperature fields exhibited a trend of initial increase followed by a decrease with aging time. Under an SO42− ionic atmosphere, the sediments predominantly showed a morphology characterized by spherical particle aggregates. The variations in particle size distribution (Fig. 7(c)) and morphology (Fig. S4) of the sediments at 25 °C reflect the processes of nucleation, agglomeration, and structural reorganization of rare-earth fluorides during the low-temperature aging process, while 90 °C sediments displayed uniform, significantly finer particles with consistent morphology (Fig. 7(d) and Fig. S5). Optimizing leaching–aging parameters (potential, temperature, duration, ionic atmosphere) via a generalized ionic atmosphere model enables controlled synthesis of CeF3 particles with tailorable morphology and size distribution (Fig. 7(e)).
Although the high-temperature aging process involves energy consumption, this energy input can be offset by the elimination of the alkali conversion step, the avoidance of fluorine-containing wastewater, and the economic value generated from high-purity CeF3 production. Notably, the high purity of CeF3 does not necessitate high energy input. While elevated temperatures favor the precipitation of rare earth fluorides, they also reduce the difference in solubility products between CeF3 and PrF3/NdF3. Aging at lower temperatures (60–90 °C) holds potential for achieving comparable selectivity through further optimization.
Overall, the coordinated leaching–selective precipitation process enables highly efficient recovery of REEs while directionally converting associated fluorine pollutants into high-value resources. This method offers distinct advantages over current mainstream bastnaesite metallurgical processes and other reported fluorine recovery technologies (Table 1). These include a shorter process flow, lower chemical consumption, higher utilization efficiency of REEs and fluorine-containing waste, as well as increased value of the resulting fluoride products. Building on previous research, this process demonstrates potential for exceptional rare earth and fluorine recovery, greener operation, and lower costs. With further adjustment and optimization of process parameters (e.g., solid-to-liquid ratio, acidity, sulfate concentration, and temperature), it can offer a superior green metallurgical solution for bastnaesite.
| Processes | Ecological–economic impact | Rare earth leaching efficiency | Fluoride product/fluorine recovery rate | Ref. |
|---|---|---|---|---|
| Oxidation roasting – HCl leaching – NaOH alkali transformation – HCl preferential dissolution | E-factor: 100 kg fluorine-containing wastewater per kg REO | ≥70% | — | 9 |
| High alkali consumption and carbon footprint | ||||
| Ca(OH)2 roasting – gravity separation – HCl leaching | High Ca(OH)2 consumption | 96.56% | CaF2/64.76% | 42 |
| High energy consumption in mineral processing | ||||
| Oxidation roasting – H2SO4 and H3BO3 high temperature leaching – Cyanex923 extraction (grade 5) – H2SO4/H2O2/NH4OH-step stripping (grade 4) – potassium salt precipitation | Complex process | 83.70% | KBF4/96.83% | 43 |
| High chemical reagent and energy consumption | ||||
| Oxidation roasting – sulfuric acid leaching – P113 Ce4+–F− co-extraction (grade 1) – H2SO4 + H2O2 stripping Ce4+–F− | Challenging synthesis of extractants | — | CeF3/∼30% | 30 |
| High costs | ||||
| Potential risk of organic fluorine-containing wastewater | ||||
| Oxidation roasting – bio-acid leaching – filtration – ascorbic acid reduction of Fe3+ – DEHPA extraction – HCI stripping | Untreated fluorine-containing raffinate | ∼90% | — | 44 |
| Oxidation roasting – H2SO4 leaching – HEH(EHP) extraction – Al3+ coordination scrubbing F− – HCl + H2O2 stripping Ce4+ – H2SO4 stripping Th4+ – precipitation | Complex process | >95 | Na3AlF6/97.30% | 45 and 46 |
| High chemical reagent consumption, cost, and energy consumption | ||||
| Oxidation roasting – HCl leaching – reductive precipitation (our previous work) | Loss of high-value non-cerium REEs during the leaching process | 90.3 | (La,Ce)F3/99% | 47 |
| Oxidation roasting – HCl leaching under an ionic atmosphere – selective precipitation of CeF3 | E-factor: <1 kg fluorine-containing wastewater per kg REO | ≥92% | CeF3/98.68% | This work |
| Zero fluoride-containing wastewater discharge | ||||
| Low reagent and energy consumption, low cost |
Aging mechanism of CeF3 under the action of a temperature field
To elucidate thermal field effects on cerium fluoride precipitation in Ce4+–F−–SO42−–Cl− systems, molecular dynamics simulations established kinetic models of Ce4+–Pr3+–Cl−–F−–SO42− systems across temperatures. Radial distribution functions centered on Ce4+ (Fig. 8(a)) and Pr3+ (Fig. 8(b)) reveal a stable Ce4+–F−–SO42− ionic atmosphere at 25 °C, where F− predominantly localizes around Ce4+ as [CeFx]4−x, with minimal F− and SO42− distribution near Pr3+. This indicates sluggish kinetics for the reduction of Ce4+ by Cl− and the binding of F− with RE3+ at low temperatures, resulting in slowed CeF3 precipitation. Concurrently, the predominant distribution of F− around Ce4+ leads to a reduction in the precipitation of high-value praseodymium and neodymium within the precipitation solution, consistent with experimental evidence from Fig. S3 and Table S3.Conversely, high temperatures (Fig. 8(d and e)) disrupt the ionic atmosphere of SO42− and destabilize [CeFx]4−x, weakening Ce4+–F− coordination while strengthening Ce4+–SO42− and Ce4+–Cl− coordination. The liberated Ce4+ is readily reduced by Cl− and subsequently combines with F− to precipitate. Thus, high temperature accelerates the precipitation rate of CeF3. Simultaneously, high temperature significantly enhances the coordination between Pr3+ and F− ions in the system, promoting the precipitation of non-cerium rare earth fluorides. Furthermore, temperature significantly influences ion diffusion and the selectivity of CeF3 precipitation (Fig. 8(c and f) and SI S8). In summary, as illustrated in Fig. 8(g), the temperature field regulates the reaction kinetics, ion transport pathways, and distribution within the Ce4+–F−–SO42−–Pr3+–Cl− system during cerium fluoride precipitation by influencing the coordination strength between rare earth cations and anions, as well as their diffusion intensity. This, in turn, governs the purity, particle size, and other characteristics of CeF3. Optimizing the aging temperature within the SO42− ionic atmosphere can enhance the selectivity of the cerium fluoride precipitation process, reducing the co-deposition of high-value Pr and Nd elements.
Conclusions
This study investigated the dissociation of bastnaesite and the reconstruction of the cerium fluoride phase under the influence of the SO42− ionic atmosphere and the temperature field. The research places particular emphasis on the coordinated leaching mechanism of bastnaesite within the ionic atmosphere, as well as the separation and recovery mechanisms of cerium fluoride from the leachate under the influence of the temperature field. The specific conclusions are as follows:(1) The dominant controlling factors for the coordinated leaching of bastnaesite and the precipitation process of cerium fluoride are temperature, potential, and the ionic atmosphere environment. At low temperatures and high acidity, and under oxidizing conditions, [CeFx]4−x complexes can exist stably. Increasing temperature and reducing conditions promote the transformation of [CeFx]4−x into CeF3. [CeF3]+ is identified as the primary stable form of [CeFx]4−x complexes.
(2) The SO42− ionic atmosphere environment enables macroscopic kinetic stability in the [CeFx]4−x–Cl− system during bastnaesite leaching. SO42− forms a stable ionic atmosphere around [CeFx]4−x during coordinated leaching, preventing the reduction of Ce4+ by Cl− and the formation of REF3. Simultaneously, it enhances the leaching of REEs, particularly high-value REEs and fluorine.
(3) The temperature field regulates the reaction rate, ion transport pathways, and distribution during cerium fluoride precipitation by affecting the coordination and diffusion intensity between rare earth ions and anions in the Ce4+–F−–SO42−–Pr3+–Cl− system, thereby influencing the purity, particle size, and other characteristics of CeF3.
In summary, this work preliminarily achieved the dissociation of the bastnaesite mineral phase and the reconstruction of the cerium fluoride phase under the synergistic effects of the SO42− ionic atmosphere and the temperature field. This approach advances the comprehensive utilization of both rare earth and fluorine resources in bastnaesite metallurgy. The ionic atmosphere model and the coordinated leaching–selective precipitation process established in this study hold potential for developing short-process, highly efficient separation and extraction processes for bastnaesite under various ionic atmospheres or temperature fields. This approach aims to achieve zero fluoride discharge, efficient leaching of high-value rare earths, and controllable purity, morphology, and particle size of the cerium fluoride product.
Author contributions
Qionghua Xie: conceptualization, methodology, data curation, writing – original draft and visualization. Yifan Hao: validation, investigation and formal analysis. Guocheng Yao: resources and project administration. Cong Wang: conceptualization and supervision. Liangshi Wang: conceptualization, writing – review & editing, supervision and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): thermodynamic data of the Ce–Cl–F–H2O system, DFT calculation, binding energy of CeF3 and PrF3, fluoride ion concentration in leachates from different leaching systems, molecular dynamics simulation, changes in the deposition process of the immersion liquid under different temperature fields, composition and morphology of sediments obtained using different temperature fields and aging times, and ion diffusion coefficients at different temperatures. See DOI: https://doi.org/10.1039/d5gc05704c.Additional data will be made available upon request.
Acknowledgements
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China 52174337.References
- Y. Chen, B. Zheng and F. Qu, Resour. Policy, 2020, 65, 101545, DOI:10.1016/j.resourpol.2019.101545.
- R. K. Jyothi, T. Thenepalli, J. W. Ahn, P. K. Parhi, K. W. Chung and J.-Y. Lee, J. Cleaner Prod., 2020, 267, 122048, DOI:10.1016/j.jclepro.2020.122048.
- W. Wen, C. Gao, Y. Xu and M. Sun, J. Cleaner Prod., 2024, 441, 140870, DOI:10.1016/j.jclepro.2024.140870.
- T. Cheisson and E. J. Schelter, Science, 2019, 363, 489–493, DOI:10.1126/science.aau7628.
- I. E. A. (IEA), The Role of Critical Minerals in Clean Energy Transitions. World Energy Outlook Special Report, 2021. https://www.iea.org/reports/the-role-of-critical-minerals-in-clean-energy-transitions.
- L. A. Attílio, Appl. Energy, 2025, 377, 124688, DOI:10.1016/j.apenergy.2024.124688.
- U.S. Geological Survey, Mineral commodity summaries 2025, Reston, VA, 2025. DOI:10.3133/mcs2024.
- A. Karati, H. Parmar, T. Riedemann, M. Besser, D. Prodius and I. C. Nlebedim, Nat. Commun., 2025, 16, 1–6, DOI:10.1038/s41467-025-59468-w.
- L. Wang, X. Huang, Y. Yu, L. Zhao, C. Wang, Z. Feng, D. Cui and Z. Long, J. Cleaner Prod., 2017, 165, 231–242, DOI:10.1016/j.jclepro.2017.07.107.
- J. C. K. Lee and Z. Wen, Nat. Sustain., 2018, 1, 598–605, DOI:10.1038/s41893-018-0154-5.
- Z. Weng and G. M. Mudd, in Wind Energy Engineering, ed. T. M. Letcher, Academic Press, 2nd edn, 2023, pp. 475–487. DOI:10.1016/B978-0-323-99353-1.00015-3.
- B. Deng, X. Wang, D. X. Luong, R. A. Carter, Z. Wang, M. B. Tomson and J. M. Tour, Sci. Adv., 2022, 8, eabm3132, DOI:10.1126/sciadv.abm3132.
- G. Bailey, P. J. Joyce, D. Schrijvers, R. Schulze, A. M. Sylvestre, B. Sprecher, E. Vahidi, W. Dewulf and K. Van Acker, Resour., Conserv. Recycl., 2020, 155, 104675, DOI:10.1016/j.resconrec.2019.104675.
- D. Talan and Q. Huang, Miner. Eng., 2022, 179, 107430, DOI:10.1016/j.mineng.2022.107430.
- Y. Geng, J. Sarkis and R. Bleischwitz, Nature, 2023, 619, 248–251, DOI:10.1038/d41586-023-02153-z.
- J. A. Mattocks, J. J. Jung, C.-Y. Lin, Z. Dong, N. H. Yennawar, E. R. Featherston, C. S. Kang-Yun, T. A. Hamilton, D. M. Park and A. K. Boal, Nature, 2023, 618, 87–93, DOI:10.1038/s41586-023-05945-5.
- B. Bernicot, G. Arrachart, S. Dourdain, N. Schaeffer, A. Dias and S. Pellet-Rostaing, Green Chem., 2025, 27, 10851–10864, 10.1039/D5GC01707F.
- S. Sun, Y. Yang, G. Tu and F. Xiao, J. Rare Earths, 2022, 40, 1963–1968, DOI:10.1016/j.jre.2021.12.008.
- Y. Yang, S. Sun and G. Tu, Nonferrous Met. Sci. Eng., 2021, 12, 126–132, DOI:10.13264/j.cnki.ysjskx.2021.04.016.
- J. Zhao, B. Li and X. Wei, J. Cleaner Prod., 2021, 327, 129489, DOI:10.1016/j.jclepro.2021.129489.
- P. Cen, W. Wu and X. Bian, Green Process. Synth., 2016, 5, 427–434, DOI:10.1515/gps-2016-0031.
- A. Shahbaz and M. Zahid, Can. Metall. Q., 2022, 62, 549–559, DOI:10.1080/00084433.2022.2102305.
- R. Chi, X. Zhang, G. Zhu, Z. A. Zhou, Y. Wu, C. Wang and F. Yu, Miner. Eng., 2004, 17, 1037–1043, DOI:10.1016/j.mineng.2004.04.010.
- M. Li, X.-W. Zhang, Z.-G. Liu, M.-T. Wang, J. Liu and J.-P. Yang, Rare Met., 2013, 32, 312–317, DOI:10.1007/s12598-013-0034-0.
- Y. Huang, Z. Dou, T.-a. Zhang and J. Liu, Hydrometallurgy, 2017, 173, 15–21, DOI:10.1016/j.hydromet.2017.07.004.
- M. Li, J. Hu, X. Zhang, Z. Liu and Y. Hu, Chin. J. Rare Met., 2016, 40, 695–700, DOI:10.13373/j.cnki.cjrm.2016.07.010.
- H. Zhang, D. Xie, H. Zhang, M. Jiang, H. Huang, Y. Wan, Y. Liao and S. Zhao, J. Rare Earths, 2021, 39, 186–193, DOI:10.1016/j.jre.2020.03.005.
- Y. Yan, J. Chen, K. Li, D. Zou, Y. Deng and D. Li, Sep. Purif. Technol., 2021, 258, 118026, DOI:10.1016/j.seppur.2020.118026.
- D. Zhang, W. Wang, Y. Deng, J. Zhang, H. Zhao and J. Chen, Chem. Eng. J., 2012, 179, 19–25, DOI:10.1016/j.cej.2011.06.021.
- M. Liu, D. Zou, D. Yu, S. Xiang, Y. Deng, J. Chen and J. Xiao, Sep. Purif. Technol., 2023, 317, 123909, DOI:10.1016/j.seppur.2023.123909.
- L. Wang, Z. Long, D. Cui, X. Huang, Y. Yu, Y. Xu and X. Feng, US Pat., 9752213B2, 2017 Search PubMed.
- D. Zhang, Geochemistry of Ore-Forming Processes, Geological Press, Beijing, 2020, pp. 70–71 Search PubMed.
- Y. Liu, Doctor's degree Fun damental science research, Beijing General Research Institute of Nonferrous Metals, 2002 Search PubMed.
- L. Zhao, L. Wang, G. Shuai, Z. Long, D. Cui and X. Huang, J. Rare Earths, 2018, 36, 758–764, DOI:10.1016/j.jre.2018.01.008.
- T. Lu and F. W. Chen, J. Mol. Graphics Modell., 2012, 38, 314–323, DOI:10.1016/j.jmgm.2012.07.004.
- J. Zhang and T. Lu, Phys. Chem. Chem. Phys., 2021, 23, 20323–20328, 10.1039/d1cp02805g.
- T. Lu and Q. X. Chen, in Conceptual Density Functional Theory, 2022, pp. 631–647. DOI:10.1002/9783527829941.ch31.
- K. Ben Mabrouk, T. H. Kauffmann, H. Aroui and M. D. Fontana, J. Raman Spectrosc., 2013, 44, 1603–1608, DOI:10.1002/jrs.4374.
- W. W. Rudolph and G. Irmer, Dalton Trans., 2017, 46, 4235–4244, 10.1039/C7DT00008A.
- W. W. Rudolph and G. Irmer, RSC Adv., 2015, 5, 84999–85008, 10.1039/C5RA16900C.
- P. Lai, Q. Lan, L. Huang, S. Liu, Y. Yang, H. Nie and X. Zhang, JOM, 2025, 77, 3187–3196, DOI:10.1007/s11837-024-07060-w.
- P. Cen, X. Bian, W. Wu and B. Li, Sep. Purif. Technol., 2021, 274, 118380, DOI:10.1016/j.seppur.2021.118380.
- Y. Li, D. Zou, J. Chen, Y. Deng and D. Li, Sep. Purif. Technol., 2023, 310, 123152, DOI:10.1016/j.seppur.2023.123152.
- W. Liu, J. Liu, E. Y. Owusu-Fordjour and X. Yang, Resour., Conserv. Recycl., 2025, 215, 108115, DOI:10.1016/j.resconrec.2024.108115.
- L. Wang, Y. Yu, X. Huang, Z. Long and D. Cui, Chem. Eng. J., 2013, 215–216, 162–167, DOI:10.1016/j.cej.2012.09.126.
- L. Wang, C. Wang, Y. Yu, X. Huang, Z. Long, Y. Hou and D. Cui, J. Hazard. Mater., 2012, 209–210, 77–83, DOI:10.1016/j.jhazmat.2011.12.069.
- L. Zhao, L. Wang, G. Shuai and Z. Long, presented in part at the Environmental Governance, Ecological Remediation and Sustainable Development, Cham, 2024, 597–606, DOI:10.1007/978-3-031-52901-6-59597.
| This journal is © The Royal Society of Chemistry 2026 |







