Photocatalytic modular synthesis of functionalized cyclotetrasiloxane (D4) monomers toward tailored polysiloxanes
Shaowei
Chen†
a,
Zhiqiang
Fang†
a,
Xiaoqian
He
a,
Qi
Jia
b,
Yanchuan
Zhao
 b and
Xiao
Shen
b and
Xiao
Shen
 *a
*a
aThe Institute for Advanced Studies, Engineering Research Center of Organosilicon Compounds & Materials, Ministry of Education, State Key Laboratory of Metabolism and Regulation in Complex Organisms, Wuhan University, Wuhan 430072, China. E-mail: xiaoshen@whu.edu.cn
bState Key Laboratory of Fluorine and Nitrogen Chemistry and Advanced Materials and Shanghai Hongkong Joint Laboratory in Chemical Synthesis, Shanghai Institute of Organic Chemistry, University of Chinese Academy of Sciences, Chinese Academy of Sciences, Shanghai 200032, China
First published on 5th December 2025
Abstract
Polysiloxanes possess immense utility across consumer and industrial sectors, yet conventional approaches to their functionalization remain constrained by reliance on platinum catalysts and limited group compatibility. We present a photocatalytic radical C–H functionalization strategy that enables direct, modular modification of octamethylcyclotetrasiloxane (D4) without noble-metal catalysts. This approach accommodates diverse functional groups, including cyano, pyridyl, sulfone, and phosphorus-based substituents, and facilitates efficient access to functionalized polysiloxanes via ring-opening polymerization. A streamlined one-pot protocol further enhances synthetic efficiency. The resulting modified polysiloxanes exhibit substantially improved thermal stability, underscoring the potential of this methodology to expand the landscape of high-performance silicone materials.
Green foundation1. This work advances green chemistry by introducing a noble-metal-free photocatalytic platform for direct C–H functionalization of D4, moving beyond traditional Pt-catalyzed methods and enhancing the sustainability of polysiloxane synthesis.2. Our method achieves a high-yielding, modular synthesis of diverse functionalized D4 monomers under mild conditions, eliminating reliance on precious metals and enabling a streamlined one-pot, two-step process from D4 to tailored polymers. 3. The green profile could be further elevated by developing continuous-flow protocols to scale the reaction. |
Polysiloxanes are integral to numerous consumer and industrial technologies, owing to their exceptional thermal stability, weather resistance, electrical insulation, and physiological inertness.1–3 These materials feature a siloxane backbone with organic side chains that can be tailored to fine-tune performance. A key precursor in silicone manufacturing is octamethylcyclotetrasiloxane (D4), whose controlled ring-opening polymerization affords products ranging from silicone oils to elastomeric rubbers (Scheme 1a).4,5 Introducing functional groups into the side chains further enhances material properties and broadens application scope.6 For example, nitrile-modified polysiloxanes have been widely employed in Li-ion batteries, soft actuators, and capacitive light-emitting devices.7–9 Conventional strategies for preparing functionalized polysiloxanes from D4 rely heavily on platinum-catalyzed hydrosilylation (Scheme 1b).10–13 These routes typically involve either prefunctionalized siloxane monomers or hydrosilylation-derived chlorosilanes. However, such methods remain constrained by their dependence on noble-metal catalysts and limited functional group compatibility, which restricts structural diversity and practical utility.
Recently, photocatalytic radical hydrogen atom transfer (HAT) mediated C–H functionalization has emerged as a powerful tool for direct molecular diversification from simple substrates.14–27 This strategy enables selective modification of inert C–H bonds without prefunctionalization, offering a more straightforward and versatile route to complex molecular architectures. Inspired by recent advances in radical C–H functionalization of organosilicon compounds,28–32 we envisioned applying this approach to synthesis of functionalized D4. This method enables a noble-metal-free route to valuable silicone monomers, circumventing the industry's longstanding platinum dependence. Moreover, photocatalytic reactions typically proceed under mild and environmentally benign conditions, exhibit excellent functional group tolerance. Herein, we report a photocatalytic radical C–H functionalization of commercially available D4 that enables efficient and modular incorporation of diverse functional groups. The resulting monomers undergo direct ring-opening polymerization to yield modified polysiloxanes (Scheme 1c). This method operates without noble-metal catalysts, significantly expands the scope of accessible silicones, and delivers polymers with improved thermal stability, as confirmed by thermogravimetric analysis, highlighting its potential for advancing the design of high-performance silicone materials.
We initiated our investigation by optimizing the reaction conditions using D4 and acrylonitrile (1a) as model substrates (for details, see SI Tables S1–S5). After extensive screening, the optimal conditions were identified as follows: 3.0 equivalents of D4 and 1.0 equivalent of 1a were reacted in the presence of FeCl3 (10 mol%) and LiCl (50 mol%) in CH3CN under 400 nm irradiation. Under these conditions, the desired C–H functionalization product 2a was obtained in 89% isolated yield (SI Table S5, entry 2). It is worth noting that this reaction proceeds most efficiently in acetonitrile, which offers high solubility for the reactants, a low boiling point to facilitate solvent recovery, and a safer profile compared to more hazardous alternatives. With the optimized conditions in hand, we proceeded to evaluate the substrate scope for synthesizing functionalized D4 monomers (Scheme 2). A variety of monosubstituted alkenes bearing electron-withdrawing groups, including ester, carbonyl, sulfonyl, and 2-pyridyl substituents, proved to be suitable reaction partners, affording the corresponding products 2b–2e in 53–83% yields. Given that the incorporation of fluorine-containing groups can enhance the hydrophobic and oleophobic properties of polysiloxanes, we also examined alkenes featuring trifluoromethyl groups.33 These substrates participated smoothly in the transformation, delivering products 2f–2g in 71–78% yield. We next turned to the synthesis of difunctionalized D4 monomers bearing two cyano groups. A range of such derivatives (2h–2v) were successfully prepared in 43–83% yield. The reaction demonstrated broad functional group tolerance, accommodating methyl, tert-butyl, fluoride, bromide, iodide, trifluoromethyl, and ester substituents. Substituents on the aromatic ring of the dicyanoalkene, whether electron-donating or electron-withdrawing, are well tolerated. However, replacing the aryl group with an alkyl group leads to a significant decrease in yield (such as 2v). Furthermore, D4 monomers functionalized with maleimide groups (2w–2aa) were synthesized in 67–91% yield. Interestingly, when allylic sulfone was used as the acceptor, the product 2ab was obtained without desulfonylation. To further demonstrate the versatility of this methodology, we explored the introduction of heteroatom-containing functional groups using diverse radical acceptors. Substrates S-(p-tolyl) 4-methylbenzenesulfonothioate and ClPPh2 furnished the corresponding products 2ac and 2ad, bearing arylthio and diarylphosphino groups, in 71% and 67% yield, respectively. It is worthy to note that compound 2a could be obtained on a 50 mmol scale reaction (10.12 g, 58% yield). However, the reactions of styrene and cyclohexene failed, probably because of the polarity mismatch with the alkyl radical derived from D4.
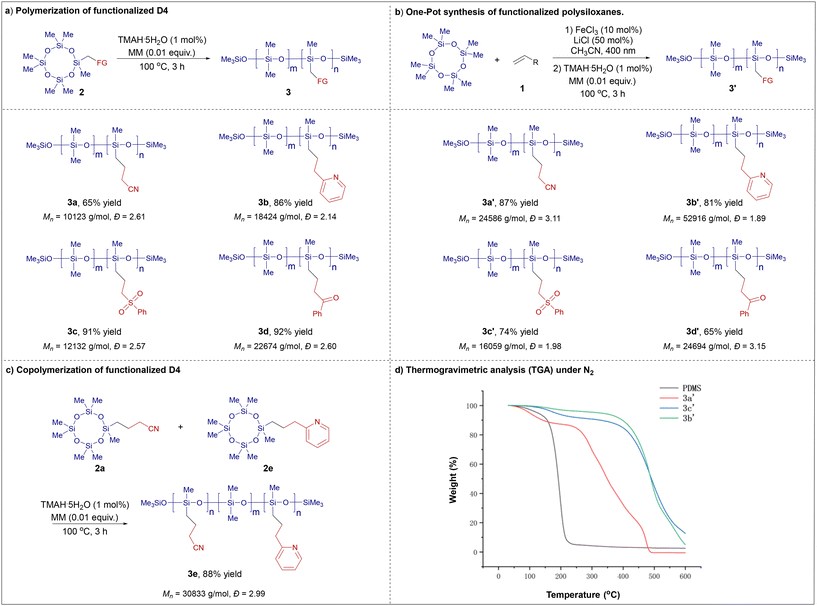
With the modular synthesis of functionalized D4 monomers accomplished, we next turned to their application in polymer synthesis (Scheme 3). Through systematic screening, we identified suitable ring-opening polymerization conditions: stirring the monomer at 100 °C for 3 hours in the presence of 1 mol% TMAH·5H2O as a basic catalyst. Under these conditions, polymerization proceeded smoothly, yielding functionalized polysiloxanes. For example, cyano-substituted polysiloxane 3a was obtained in 65% yield after workup (Scheme 3a). NMR analysis confirmed a cyano group content of 25%, consistent with the feed ratio of the functionalized monomer. This polymerization strategy was successfully extended to other functional monomers. Pyridine-containing polysiloxane 3b was synthesized in 86% yield under similar conditions. Additionally, polysiloxanes bearing sulfone and ketone functional groups (3c and 3d) were prepared in 91% and 92% yield, respectively. Gel permeation chromatography (GPC) was used to determine the molecular weight characteristics (Mw, Mn, and Đ) of the resulting polymers and high Mw values were obtained.
While the copolymerization of functionalized monomers with D4 provided a viable route to modified polysiloxanes, we envisioned a more streamlined one-pot approach that directly converts D4 into functionalized polysiloxanes without isolating the intermediate monomer (Scheme 3b). This process begins with the radical C–H functionalization of D4, followed by direct polymerization of the crude mixture after simple workup (filtration and concentration) upon addition of TMAH·5H2O. The functional group content in the final polymer could be readily controlled by adjusting the initial feed ratio of D4 to the radical acceptor. Using this method, modified polysiloxanes 3a′–3d′ were successfully synthesized. Encouraged by these results, we further explored the copolymerization of two different functional monomers, 2a and 2e, which afforded copolymer 3e in 88% yield, demonstrating the versatility of this approach for accessing diverse polysiloxane architectures (Scheme 3c). Again, high Mw values were obtained for these polymers.
To evaluate the thermal stability of the synthesized functionalized polysiloxanes, thermogravimetric analysis (TGA) was performed under an inert atmosphere (Scheme 3d). Unmodified polydimethylsiloxane (PDMS) exhibited a decomposition onset temperature (Td) of 156 °C. In contrast, the cyano-substituted polysiloxane showed a markedly enhanced stability, with Td increasing by 100 °C to 256 °C. More notably, polysiloxanes incorporating pyridine and sulfone functional groups demonstrated a further substantial increase in thermal resistance, exhibiting decomposition temperatures as high as 430 °C. These results clearly indicate that side-chain functionalization via our developed method significantly enhances the thermal stability of polysiloxanes, underscoring the potential of this strategy to tailor the properties of silicone-based polymeric materials.
Conclusions
In summary, we have developed a modular and efficient strategy for the direct C–H functionalization of octamethylcyclotetrasiloxane (D4) via a radical hydrogen atom transfer pathway. This method enables the introduction of diverse functional groups, including cyano, pyridyl, sulfonyl, ketone, and other heteroatom-containing substituents, into the D4 framework under mild photocatalytic conditions, without relying on precious metal catalysts. The resulting functionalized D4 monomers undergo facile ring-opening polymerization to afford well-defined polysiloxanes with tailored side-chain functionalities. Furthermore, a streamlined one-pot, two-step protocol has been established to directly access modified polysiloxanes from unmodified D4, bypassing the need for intermediate isolation. Thermogravimetric analysis confirms that the introduced functional groups significantly enhance the thermal stability of the resulting silicone materials. This work establishes a general and versatile platform for the synthesis of functional silicones, opening up new avenues for the design of high-performance silicone-based polymers.Conflicts of interest
The authors declare no competing interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details and characterization of all compounds. See DOI: https://doi.org/10.1039/d5gc05780a.Acknowledgements
We are grateful to National Key R&D Program of China (2022YFA1506100), National Natural Science Foundation of China (22471201, 22531010, 22501209, 22503064), the China Postdoctoral Science Foundation (2023TQ0252, 2023M742687), the Postdoctoral Foundation of Hubei Province (211000032) and the Postdoctoral Fellowship Program of CPSF (GZC20231960) for financial support. We thank Dr Liya Ma from the Core Facility of Wuhan University for her assistance with TGA analysis.References
- J. E. Mark, Acc. Chem. Res., 2004, 37, 946–953 CrossRef CAS PubMed.
- N. Đ. Vũ, A. Boulegue-Mondière, N. Durand, J. Munsch, M. Boste, R. Lhermet, D. Gajan, A. Baudouin, S. Roldán-Gómez, M. Perrin, V. Monteil and J. Raynaud, Science, 2025, 388, 392–400 CrossRef.
- Grand View Research, Silicone market size, share & trends analysis report by product (fluids, gels, resins, elastomers), by end-use (electronics, transportation, construction, healthcare, industrial processes, energy), by region, and segment forecasts, 2024–2030, Report GVR-1-68038-063-7, Grand View Research, 2023, https://www.grandviewresearch.com/industry-analysis/silicone-market.
- L. Shi, A. Boulègue-Mondière, D. Blanc, A. Baceiredo, V. Branchadell and T. Kato, Science, 2023, 381, 1011–1014 CrossRef CAS PubMed.
- N. S. Sarai, T. J. Fulton, R. L. O'Meara, K. E. Johnston, S. Brinkmann-Chen, R. R. Maar, R. E. Tecklenburg, J. M. Roberts, J. C. T. Reddel, D. E. Katsoulis and F. H. Arnold, Science, 2024, 383, 438–443 CrossRef CAS.
- B. Boutevin, F. Guida-Pietrasanta and A. Ratsimihety, Side Group Modified Polysiloxanes, ed. R. G. Jones and W. Ando and J. Chojnowski, in Silicon-Containing Polymers, Springer, Dordrecht, 2000 Search PubMed.
- S. Xie, A. Nikolaev, O. A. Nordness, L. C. Llanes, S. D. Jones, P. M. Richardson, H. Wang, R. J. Clément, J. Read de Alaniz and R. A. Segalman, Macromolecules, 2022, 55, 5723–5732 CrossRef CAS.
- J. Von Szczepanski, P. M. Danner and D. M. Opris, Adv. Sci., 2022, 9, 1–9 Search PubMed.
- P. M. Danner, T. Pleij, G. Siqueira, A. V. Bayles, T. R. Venkatesan, J. Vermant and D. M. Opris, Adv. Funct. Mater., 2024, 34, 2313167 CrossRef CAS.
- N. Dong, J. Duan, C. Zhang, X. Gu, L. Feng and L. Liu, Polymer, 2025, 323, 128163 CrossRef CAS.
- T. Wang, J. Yang and Q. Cao, Nat. Commun., 2023, 14, 1342 CrossRef CAS PubMed.
- S. Kinoshita, S. Watase, K. Matsukawa and Y. Kaneko, J. Am. Chem. Soc., 2015, 137, 5061–5065 CrossRef CAS PubMed.
- J. McGee, J. Kunzler, R. Ozark and J. Salamone, US Pat, US20100118261A1, 2010 Search PubMed.
- H. Cao, X. Tang, H. Tang, Y. Yuan and J. Wu, Chem Catal., 2021, 1, 523–598 CAS.
- L. Capaldo, D. Ravelli and M. Fagnoni, Chem. Rev., 2022, 122, 1875–1924 CrossRef CAS PubMed.
- P. Bellotti, H.-M. Huang, T. Faber and F. Glorius, Chem. Rev., 2023, 123, 4237–4352 CrossRef CAS PubMed.
- D. L. Golden, S.-E. Suh and S. S. Stahl, Nat. Rev. Chem., 2022, 6, 405–427 CrossRef CAS PubMed.
- L. Capaldo and D. Ravelli, Eur. J. Org. Chem., 2017, 2056–2071 CrossRef CAS.
- S. Sarkar, K. P. S. Cheung and V. Gevorgyan, Chem. Sci., 2020, 11, 12974–12993 RSC.
- H. Yi, G. Zhang, H. Wang, Z. Huang, J. Wang, A. K. Singh and A. Lei, Chem. Rev., 2017, 117, 9016–9085 CrossRef CAS PubMed.
- Z. Ye, Y. Lin and L. Gong, Eur. J. Org. Chem., 2021, 5545–5556 CrossRef CAS.
- L. Stateman, K. Nakafuku and D. Nagib, Synthesis, 2018, 1569–1586 CAS.
- J. L. Jeffrey, J. A. Terrett and D. W. C. MacMillan, Science, 2015, 349, 1532–1536 CrossRef CAS PubMed.
- P.-F. Zhong, J.-L. Tu, Y. Zhao, N. Zhong, C. Yang, L. Guo and W. Xia, Nat. Commun., 2023, 14, 6530–6540 CrossRef CAS PubMed.
- H. Cao, Y. Kuang, X. Shi, K. L. Wong, B. B. Tan, J. M. C. Kwan, X. Liu and J. Wu, Nat. Commun., 2020, 11, 1956 CrossRef CAS PubMed.
- A. Hu, J.-J. Guo, H. Pan and Z. Zuo, Science, 2018, 361, 668–672 CrossRef CAS PubMed.
- H.-S. Wang, H.-C. Li, X.-Y. Yuan, K. Sun, X.-L. Chen, L. Qu and B. Yu, Green Chem., 2025, 27, 4655–4663 RSC.
- X. He, Y. Zhang, S. Liu, W. Zhang, Z. Liu, Y. Zhao and X. Shen, Nat. Synth., 2024, 4, 188–195 CrossRef.
- C. Shu, A. Noble and V. K. Aggarwal, Nature, 2020, 586, 714–719 CrossRef CAS PubMed.
- M. Wang, Y. Huang and P. Hu, Science, 2024, 383, 537–544 CrossRef CAS PubMed.
- R. Sang, W. Han, H. Zhang, C. M. Saunders, A. Noble and V. K. Aggarwal, J. Am. Chem. Soc., 2023, 145, 15207–15217 CrossRef CAS PubMed.
- J.-L. Tu, A.-M. Hu, L. Guo and W. Xia, J. Am. Chem. Soc., 2023, 145, 7600–7611 CrossRef CAS PubMed.
- R. Shi, X. Han, L. Zhu, J. Hao, H. Wang and C. Zhou, Polymer, 2024, 308, 127428 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |


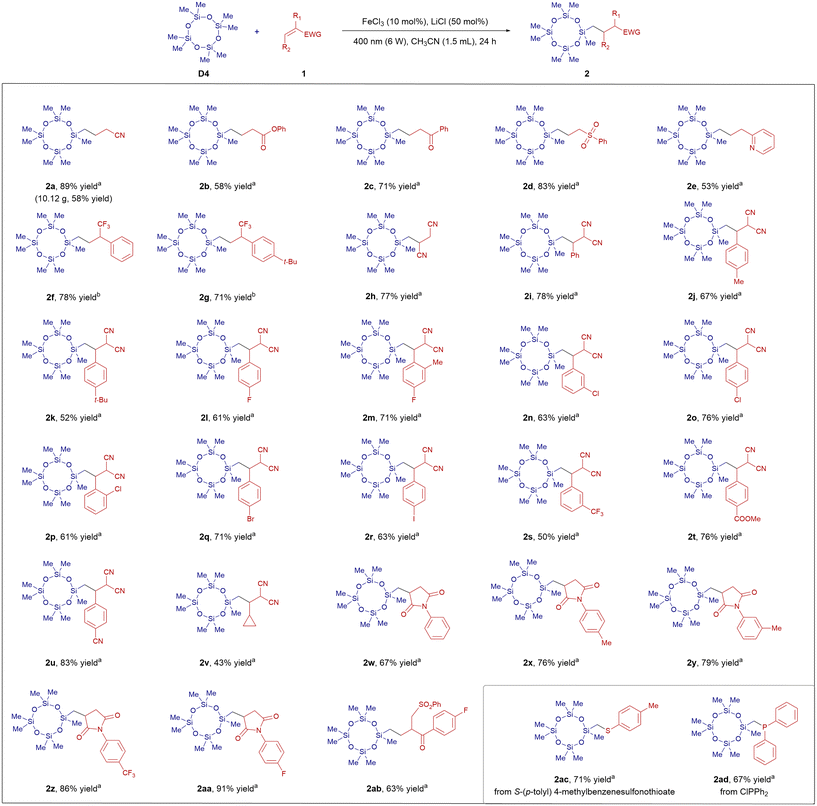
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D4 (0.6 mmol), 1 (0.2 mmol), FeCl3 (10 mol%), LiCl (50 mol%), CH3CN (1.5 mL), 400 nm (6 W), 24 h.
D4 (0.6 mmol), 1 (0.2 mmol), FeCl3 (10 mol%), LiCl (50 mol%), CH3CN (1.5 mL), 400 nm (6 W), 24 h.