Open Access Article
Open Access ArticlePalladium-catalyzed asymmetric sequential hydroalkylation and hydroamination of 1,3-enynes with 3-hydroxyindoles
Qiuyu
Li†
a,
Ruixue
Wu†
a,
Tianbao
Wu†
b,
Renkang
Wei
a,
Zhijiao
Li
a,
Shang
Gao
 a,
Minyan
Wang
*b,
Hequan
Yao
a,
Minyan
Wang
*b,
Hequan
Yao
 *a and
Aijun
Lin
*a and
Aijun
Lin
 *a
*a
aState Key Laboratory of Natural Medicines (SKLNM) and Department of Medicinal Chemistry, School of Pharmacy, China Pharmaceutical University, Nanjing, 210009, P. R. China. E-mail: hyao@cpu.edu.cn; ajlin@cpu.edu.cn
bState Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering, Nanjing University, Nanjing 210093, China
First published on 12th January 2026
Abstract
Transition-metal catalyzed asymmetric hydrofunctionalization of unsaturated hydrocarbons has emerged as an efficient method to access diverse chiral value-added compounds. In contrast, asymmetric sequential hydrofunctionalization cyclization, which could incorporate two functional groups across carbon–carbon multiple bonds to construct chiral complex cyclic compounds, has only been sporadically explored. Herein, we report a palladium-catalyzed asymmetric sequential hydroalkylation and hydroamination of readily available 1,3-enynes with 3-hydroxyindoles. This redox-neutral process provides an efficient route for constructing a broad spectrum of enantioenriched pyrido[1,2-a]indoles and derivatives with high atom- and step-economy. Preliminary mechanistic investigations reveal that this transformation proceeds via an intermolecular enyne hydroalkylation pathway to produce an allene intermediate. Subsequent intramolecular hydroamination of the allene intermediate occurs via an axial-to-center chirality transfer process. Density functional theory (DFT) studies were conducted to probe the origin of enantioselectivities.
Green foundation1. This palladium-catalyzed asymmetric sequential hydroalkylation/hydroamination of 1,3-enynes achieves 100% atom economy under redox-neutral conditions, minimizing waste by incorporating all atoms of the starting materials into the final products.2. This asymmetric sequential hydrofunctionalization cyclization can install two distinct functional groups across the unsaturated hydrocarbons in one pot with excellent step economy. 3. The enantioenriched pyrido[1,2-a]indoles and derivatives are constructed with high efficiency with excellent chemo-, enantio-, and diastereo-selectivity (up to >20 |
Introduction
With increasing concerns about environmental sustainability, preparing value-added molecules such as bioactive compounds and pharmaceuticals, from readily available materials in a redox-neutral, atom- and step-economical manner has been a hot topic for the synthetic and medicinal communities.1 One of the versatile strategies to achieve this goal is to develop transition-metal catalyzed hydrofunctionalization of unsaturated hydrocarbons, which provides synthetically valuable products with 100% atom utilization.2 Over the past decades, great efforts have been made in the catalytic asymmetric hydrofunctionalization of alkenes,3 alkynes,4 allenes,5 and dienes,6 facilitating the construction of alkyl and allyl compounds. Recently, the asymmetric hydrofunctionalization of conjugated enynes has garnered attention and warrants further study. In this context, the groups of Buchwald, Hoveyda, Malcolmson, Engle, He and Luo have independently gained elegant achievements for the synthesis of optically pure allene and propargyl compounds (Scheme 1a).7 However, these advancements have predominantly concentrated on the introduction of a single functional motif onto unsaturated hydrocarbons.Sequential reactions, which could incorporate several transformations into a single sequence, represent a powerful strategy to rapidly install two or more functional groups to construct complex molecules in a step-economical fashion.8 In this regard, asymmetric sequential hydrofunctionalization of conjugated enynes provides a unique approach to assembling structurally diverse chiral cyclic compounds. Nevertheless, only a few asymmetric variants have been disclosed to date. In 2021, the Shao group developed a palladium-catalyzed sequential hydroalkylation of 1,3-enynes (double C–C bond formation) to produce a panel of chiral spiro compounds in good yields.9 In 2022, Meng and coworkers described a Co-catalyzed sequential hydrosilylation of 1,3-enynes (double C–Si bond formation) to generate enantioenriched cyclic alkenylsilane scaffolds with a high level of enantioinduction.10 In addition, our group reported a Pd-catalyzed sequential hydroamination of 1,3-enynes and ureas via the formation of double C–N bonds, yielding valuable chiral imidazolidinone architectures (Scheme 1b).11 Despite this progress, the installation of two distinct functional groups via the asymmetric sequential hydrofunctionalization of enynes has thus far proven elusive.12
As an extension of our interest in the catalytic hydrofunctionalization of unsaturated hydrocarbons,11,13 we seek to integrate hydroalkylation and hydroamination with 3-hydroxyindoles as nucleophiles, fulfilling the more challenging asymmetric sequential hydrofunctionalization of 1,3-enynes (Scheme 1c). However, the realization of this procedure in one pot presents several sticky issues. (1) The reaction might terminate prematurely at the first stage, leading to the formation of allene or diene products. (2) Both 3-hydroxyindoles and 1,3-enynes contain multiple reaction sites, complicating precise control over chemo- and regioselectivity. (3) Achieving control over the enantioselectivity and diastereoselectivity of the products poses an additional challenge.
Herein, we report an unprecedented palladium-catalyzed asymmetric sequential hydroalkylation and hydroamination reaction between 3-hydroxyindoles and 1,3-enynes to yield optically pure pyrido[1,2-a]indoles and derivatives in good efficiency. This process demonstrates excellent regio-, chemo-, and stereoselectivities, along with commendable atom- and step-economy. These scaffolds constitute a structurally fascinating part of indole alkaloids, which are prevalent in a variety of natural products and bioactive compounds.14 Preliminary mechanistic studies disclose that this consecutive reaction proceeds through an intermolecular enyne hydroalkylation pathway to yield an allene intermediate. The subsequent intramolecular hydroamination of the allene intermediate involves an axial-to-center chirality transfer process.
Results and discussion
Optimization studies of the racemic process
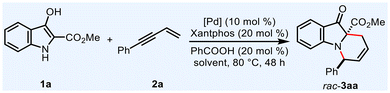
To validate the feasibility of this strategy, we commenced our investigation on the racemic process with commercially available 3-hydroxyindole-2-carboxylate 1a and enyne 2a as model substrates. Employing Pd(OAc)2 as the catalyst, Xantphos as the ligand, PhCOOH as the acid additive and DCM as the reaction medium, we successfully isolated product 3aa in 30% yield with 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (Table 1, entry 1). Subsequent optimization identified that Pd(acac)2 or [Pd(π-allyl)Cl]2 as the catalyst and toluene as the solvent significantly enhanced both productivity and diastereoselectivity (Table 1, entries 2–5).
1 dr (Table 1, entry 1). Subsequent optimization identified that Pd(acac)2 or [Pd(π-allyl)Cl]2 as the catalyst and toluene as the solvent significantly enhanced both productivity and diastereoselectivity (Table 1, entries 2–5).
| Entry | [Pd] source | Solvent | Yield (%) | dr |
|---|---|---|---|---|
| a Standard conditions: 1a (0.2 mmol), 2a (0.4 mmol), [Pd] (10 mol%), Xantphos (20 mol%), PhCOOH (20 mol%), solvent (1.0 mL), at 80 °C under an Ar atmosphere for 48 h. b Isolated yield. c The dr values were determined by crude 1H NMR. | ||||
| 1 | Pd(OAc)2 | DCM | 30 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 2 | Pd(OAc)2 | 1,4-Dioxane | 54 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | Pd(OAc)2 | Toluene | 59 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 4 | Pd(acac)2 | Toluene | 65 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 5 | [Pd(π-allyl)Cl]2 | Toluene | 75 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
Optimization studies of the asymmetric process
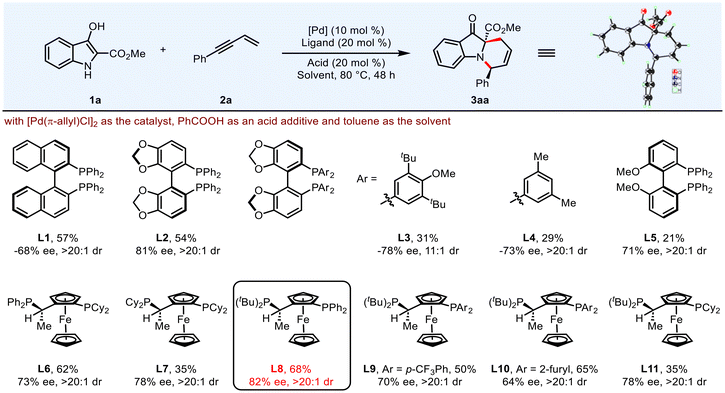
Encouraged by the success in racemic transformation, we endeavored to interpret this transformation into an enantioselective process. Combined with [Pd(π-allyl)Cl]2 as the catalyst, PhCOOH as the acid additive and toluene as the solvent, a variety of chiral BINAP-type bisphosphines were evaluated (Table 2, L1–L4). When (S)-SEGPHOS (L2) was employed as the chiral ligand, product 3aa could be achieved in 54% yield with 81% ee and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. Modifying the electronic and steric properties of the ligands resulted in inferior outcomes (L3 and L4). Upon assessing biphenyl-type and JosiPhos-type chiral ligands, L8 emerged as the optimal, furnishing 3aa in 68% yield with 82% ee. Solvent screening indicated that PhCF3 provided the best results (Table 2, entries 1–4). Exploration of the palladium catalysts revealed that [Pd(π-allyl)Cl]2 was the most promising one (Table 2, entries 5–7). Different types of acids were then examined, but no superior yield and enantioselectivity were obtained (Table 2, entries 8–11). Next, lowering the reaction temperature can efficiently boost the ee values (Table 2, entries 12–15), and the enantioselectivity could be increased to 95% when the reaction was carried out at 30 °C (Table 2, entry 15). Elongation of the reaction time to 72 h afforded the product 3aa in 84% isolated yield with 95% ee and >20
1 dr. Modifying the electronic and steric properties of the ligands resulted in inferior outcomes (L3 and L4). Upon assessing biphenyl-type and JosiPhos-type chiral ligands, L8 emerged as the optimal, furnishing 3aa in 68% yield with 82% ee. Solvent screening indicated that PhCF3 provided the best results (Table 2, entries 1–4). Exploration of the palladium catalysts revealed that [Pd(π-allyl)Cl]2 was the most promising one (Table 2, entries 5–7). Different types of acids were then examined, but no superior yield and enantioselectivity were obtained (Table 2, entries 8–11). Next, lowering the reaction temperature can efficiently boost the ee values (Table 2, entries 12–15), and the enantioselectivity could be increased to 95% when the reaction was carried out at 30 °C (Table 2, entry 15). Elongation of the reaction time to 72 h afforded the product 3aa in 84% isolated yield with 95% ee and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (Table 2, entry 16). Notably, in the absence of an acidic additive, the transformation still proceeded, albeit with reduced yield and enantioselectivity (Table 2, entry 17), underscoring the critical role of the acidic additive in this reaction. The absolute configuration of 3aa was confirmed by X-ray diffraction and those of others were assigned by analogy.
1 dr (Table 2, entry 16). Notably, in the absence of an acidic additive, the transformation still proceeded, albeit with reduced yield and enantioselectivity (Table 2, entry 17), underscoring the critical role of the acidic additive in this reaction. The absolute configuration of 3aa was confirmed by X-ray diffraction and those of others were assigned by analogy.
| Entry | [Pd] source | Solvent | Additive | T/°C | Yield (%) | ee (%) | dr |
|---|---|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 2a (0.4 mmol), [Pd] (10 mol%), ligand (20 mol%) and acid (20 mol%) in solvent (1.0 mL), under an argon atmosphere. b Yields of 3aa were determined by GC with n-dodecane as the internal standard. c The dr and ee values were determined by chiral HPLC analysis. d 72 h. e Isolated yield. | |||||||
| 1 | [Pd(π-allyl)Cl]2 | Toluene | PhCOOH | 80 | 68 | 82 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 2 | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 80 | 75 | 86 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | [Pd(π-allyl)Cl]2 | Mesitylene | PhCOOH | 80 | 76 | 82 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 4 | [Pd(π-allyl)Cl]2 | THF | PhCOOH | 80 | 65 | 67 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 5 | Pd(dba)2 | PhCF3 | PhCOOH | 80 | 71 | 86 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 6 | Pd(OAc)2 | PhCF3 | PhCOOH | 80 | 63 | 81 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 7 | Pd(acac)2 | PhCF3 | PhCOOH | 80 | 72 | 84 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 8 | [Pd(π-allyl)Cl]2 | PhCF3 | 4-F-C6H4COOH | 80 | 70 | 85 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 9 | [Pd(π-allyl)Cl]2 | PhCF3 | PhMe2CCOOH | 80 | 53 | 82 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 10 | [Pd(π-allyl)Cl]2 | PhCF3 | Ph3CCOOH | 80 | 67 | 84 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 11 | [Pd(π-allyl)Cl]2 | PhCF3 | 1-AdCOOH | 80 | 65 | 82 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 12 | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 60 | 71 | 90 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 13 | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 50 | 68 | 90 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 14 | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 40 | 63 | 92 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 15 | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 30 | 60 | 95 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 16d | [Pd(π-allyl)Cl]2 | PhCF3 | PhCOOH | 30 | 86 (84)e | 95 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 17d | [Pd(π-allyl)Cl]2 | PhCF3 | — | 30 | 23 | 80 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
Substrate scope in asymmetric transformation
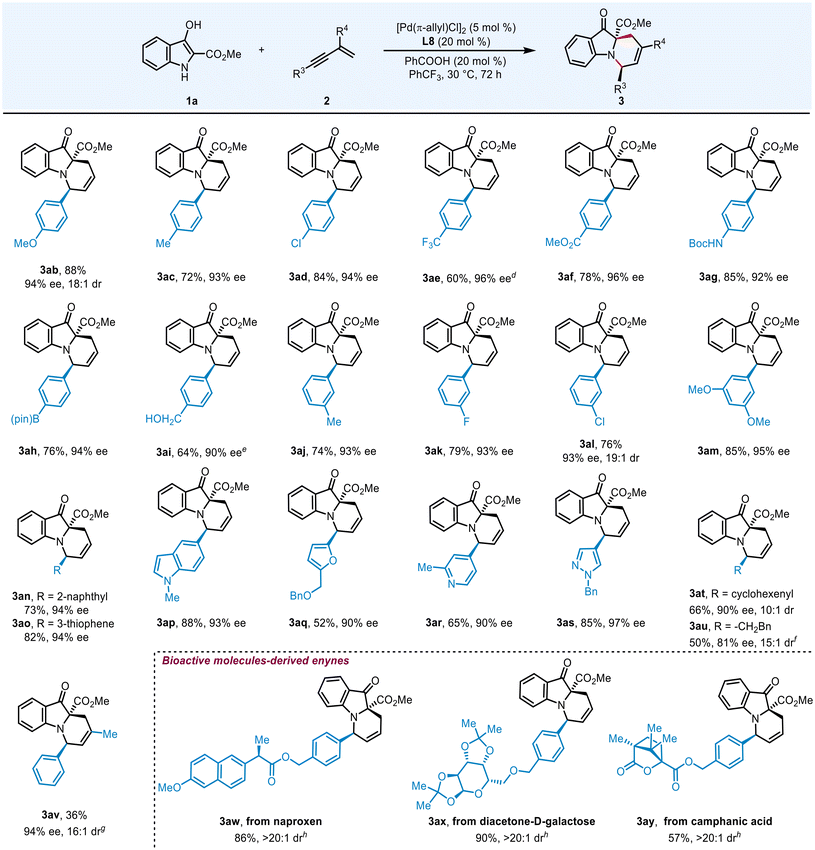
Having determined the optimal conditions, we then investigated the substrate scope of 3-hydroxyindoles. As illustrated in Table 3, a preliminary examination of ester groups at the C2-position was initially performed. Switching the methyl group to other alkyl groups, such as ethyl and benzyl, this reaction could proceed smoothly as well and delivered 3ba and 3ca in 74% and 73% yields, respectively. A bulkier tertiary butyl group was also compatible with this catalytic system, yielding product 3da in 63% yield with 91% ee and 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. However, amide-substituted product 3ea could only be isolated in 43% yield even under 60 °C for 5 days, with 50% starting material recovered. Both electron-donating (–Me and –OMe) and electron-withdrawing (–F, –Cl, and –CO2Me) substituents at various positions of the aromatic rings were well tolerated, generating products 3fa–na in 61–88% yields with 93–96% ee and >20
1 dr. However, amide-substituted product 3ea could only be isolated in 43% yield even under 60 °C for 5 days, with 50% starting material recovered. Both electron-donating (–Me and –OMe) and electron-withdrawing (–F, –Cl, and –CO2Me) substituents at various positions of the aromatic rings were well tolerated, generating products 3fa–na in 61–88% yields with 93–96% ee and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. In addition, polyaromatic fused rings, such as substrate 1oa, underwent this sequential hydroalkylation and hydroamination reaction smoothly, furnishing 3oa in 84% yield with 94% ee.
1 dr. In addition, polyaromatic fused rings, such as substrate 1oa, underwent this sequential hydroalkylation and hydroamination reaction smoothly, furnishing 3oa in 84% yield with 94% ee.
a Reaction conditions: 1 (0.2 mmol), 2a (0.4 mmol), [Pd(π-allyl)Cl]2 (5 mol%), L8 (20 mol%) and PhCOOH (20 mol%) in PhCF3 (1.0 mL), 30 °C, 72 h, under an argon atmosphere. All products were obtained with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, unless otherwise stated.
b Isolated yield.
c The dr and ee values were determined by chiral HPLC analysis.
d 60 °C, 5 d.
e 96 h. 1 dr, unless otherwise stated.
b Isolated yield.
c The dr and ee values were determined by chiral HPLC analysis.
d 60 °C, 5 d.
e 96 h.
|
|---|

|
Subsequently, we directed our focus towards exploring the substrate scope of conjugated enynes (Table 4). The reaction involving enynes with electron-donating and electron-withdrawing substituents at the para-position of the phenyl ring generated chiral polycyclic indolines 3ab–ad and 3af in 72–88% yields with 93–96% ee. The introduction of a trifluoromethyl group was also tolerated, delivering product 3ae in 60% yield, albeit with the concomitant formation of a diene byproduct. Functional groups such as NHBoc (3ag), Bpin (3ah) and unprotected hydroxyl (3ai) were all well accommodated, maintaining good yields (64–85%) and stereoselectivities (90–94% ee, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr). Excellent outcomes were also observed when meta-methyl, fluoride, chlorine and 3,5-dimethoxy were incorporated on the phenyl ring. In addition to aryl moieties, other fused arenes and heterocycles, such as naphthalene (2n), thiophene (2o), indole (2p), furan (2q), pyridine (2r) and parazole (2s), were all compatible with this transformation, delivering the desired products 3an–as in 52–88% yields with 90–97% ee. Switching the aromatic rings to a cyclohexene motif, this reaction performed smoothly as well and compound 3at was isolated in 66% yield with 90% ee. Efforts to broaden the diversity of enyne substrates, such as alkyl enynes and those with substituents at R4, proved unsuccessful under the standard conditions. Under slightly modified conditions (with L9 as the chiral ligand), 3au was obtained in 50% yield and 81% ee, while the diene byproduct was also formed. In addition, utilizing L2 as the chiral ligand enabled the reaction of 2v to generate 3av with good stereoselectivity, although in a lower yield at the expense of concurrent diene byproduct formation. The practicality of this protocol was assessed in the late stage sequential hydrofunctionalization of enynes derived from diverse biologically active molecules. Specifically, the reaction proceeded smoothly with enynes tethered to pharmaceuticals such as naproxen (2w), diacetone-D-galactose (2x), and camphanic acid (2y) to produce the target adducts in 57–90% yields with >20
1 dr). Excellent outcomes were also observed when meta-methyl, fluoride, chlorine and 3,5-dimethoxy were incorporated on the phenyl ring. In addition to aryl moieties, other fused arenes and heterocycles, such as naphthalene (2n), thiophene (2o), indole (2p), furan (2q), pyridine (2r) and parazole (2s), were all compatible with this transformation, delivering the desired products 3an–as in 52–88% yields with 90–97% ee. Switching the aromatic rings to a cyclohexene motif, this reaction performed smoothly as well and compound 3at was isolated in 66% yield with 90% ee. Efforts to broaden the diversity of enyne substrates, such as alkyl enynes and those with substituents at R4, proved unsuccessful under the standard conditions. Under slightly modified conditions (with L9 as the chiral ligand), 3au was obtained in 50% yield and 81% ee, while the diene byproduct was also formed. In addition, utilizing L2 as the chiral ligand enabled the reaction of 2v to generate 3av with good stereoselectivity, although in a lower yield at the expense of concurrent diene byproduct formation. The practicality of this protocol was assessed in the late stage sequential hydrofunctionalization of enynes derived from diverse biologically active molecules. Specifically, the reaction proceeded smoothly with enynes tethered to pharmaceuticals such as naproxen (2w), diacetone-D-galactose (2x), and camphanic acid (2y) to produce the target adducts in 57–90% yields with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr.
1 dr.
a Reaction conditions: 1a (0.2 mmol), 2 (0.4 mmol), [Pd(π-allyl)Cl]2 (5 mol%), L8 (20 mol%) and PhCOOH (20 mol%) in PhCF3 (1.0 mL), 30 °C, 72 h, under an argon atmosphere. All products were obtained with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, unless otherwise stated.
b Isolated yield.
c The dr and ee values were determined by chiral HPLC analysis.
d 40 °C.
e 50 °C, 96 h.
f
L9, 50 °C, 72 h.
g
L2 (20 mol%), 80 °C, 48 h.
h The dr values were determined by 1H NMR. 1 dr, unless otherwise stated.
b Isolated yield.
c The dr and ee values were determined by chiral HPLC analysis.
d 40 °C.
e 50 °C, 96 h.
f
L9, 50 °C, 72 h.
g
L2 (20 mol%), 80 °C, 48 h.
h The dr values were determined by 1H NMR.
|
|---|
|
Mechanistic studies
Based on previous reports,9,11 we hypothesized that the intermolecular hydroalkylation of 3-hydroxyindole 1 and enyne 2 should form an allene intermediate. Subsequent intramolecular hydroamination of the allene was expected to produce the corresponding polycyclic indoline 3. To verify this hypothesis, the reaction of 1a with enyne 2a was carried out under the standard conditions and quenched after 12 h. Then, the product 3aa was isolated in 23% yield, with considerable amounts of 1a remaining. However, the allene intermediate 4 was not detected (Scheme 2a). Furthermore, we attempted the synthesis of an allene intermediate in the presence of Pd(acac)2 and Xantphos. When the reaction was performed at 50 °C, rac-3aa was formed in 20% yield, along with diene rac-5 in 55% yield. Again, allene rac-4 was not detected (Scheme 2b). To gain insight into the reaction process, we prepared enantioenriched allene 4via a palladium-catalyzed allylic reaction of allenyl carbonate (see the SI for more details).15 Treatment of allene 4 with Xantphos as the ligand afforded product 3aa in 64% yield with 96% ee and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. Subsequently, allene 4 was subjected to asymmetric conditions with L8 as the ligand, resulting in the intramolecular hydroamination product 3aa in 37% yield, along with 39% yield of diene 5 in 96% ee. Compound 3aa was obtained in 71% yield with trace diene detected when the reaction was carried out at 50 °C (Scheme 2c). These data indicated that chiral allene 4 could efficiently transform into enantiopure 3aavia axial-to-center chirality transfer. Moreover, the diene product might be generated via Pd–H migratory insertion of allene followed by β-H elimination.6m,16 These observations prompted us to speculate that diene might also serve as an intermediate in this Pd-catalyzed asymmetric sequential hydrofunctionalization reaction. To corroborate this hypothesis, chiral diene 5 was subjected to the conditions with L8 as the ligand, but the diene was fully recovered (Scheme 2d, top panel). When the reaction was performed at 50 °C, product 3aa was isolated in 20% yield with 96% ee (Scheme 2d, bottom panel). Collectively, these observations suggest that allene acts as the crucial intermediate of this sequential hydrofunctionalization, whereas diene was the by-product, as it cannot be converted into the target product under standard conditions. Furthermore, deuterium-labeling experiments were conducted. As shown in Scheme 2e, the reaction of 3-hydroxyindole 1a–D with enyne 2a in PhCF3 in the presence of PhCOOD provided 3aa-D in 65% yield, exhibiting 69% and 72% deuterium incorporation at the α- and β-positions, respectively. This result indicated that this reaction might involve two successive Pd–H migratory insertions.
1 dr. Subsequently, allene 4 was subjected to asymmetric conditions with L8 as the ligand, resulting in the intramolecular hydroamination product 3aa in 37% yield, along with 39% yield of diene 5 in 96% ee. Compound 3aa was obtained in 71% yield with trace diene detected when the reaction was carried out at 50 °C (Scheme 2c). These data indicated that chiral allene 4 could efficiently transform into enantiopure 3aavia axial-to-center chirality transfer. Moreover, the diene product might be generated via Pd–H migratory insertion of allene followed by β-H elimination.6m,16 These observations prompted us to speculate that diene might also serve as an intermediate in this Pd-catalyzed asymmetric sequential hydrofunctionalization reaction. To corroborate this hypothesis, chiral diene 5 was subjected to the conditions with L8 as the ligand, but the diene was fully recovered (Scheme 2d, top panel). When the reaction was performed at 50 °C, product 3aa was isolated in 20% yield with 96% ee (Scheme 2d, bottom panel). Collectively, these observations suggest that allene acts as the crucial intermediate of this sequential hydrofunctionalization, whereas diene was the by-product, as it cannot be converted into the target product under standard conditions. Furthermore, deuterium-labeling experiments were conducted. As shown in Scheme 2e, the reaction of 3-hydroxyindole 1a–D with enyne 2a in PhCF3 in the presence of PhCOOD provided 3aa-D in 65% yield, exhibiting 69% and 72% deuterium incorporation at the α- and β-positions, respectively. This result indicated that this reaction might involve two successive Pd–H migratory insertions.
Based on the above mechanistic studies and detailed density functional theory (DFT) calculations (see the SI for details), a plausible catalytic cycle for this reaction of conjugated enynes and 3-hydroxyindoles is proposed in Scheme 2f.9,11 Oxidative addition of Pd(0) with the acid affords PdH species A. Regioselective alkyne insertion to the PdH species A leads to the formation of η1-butadienyl-Pd intermediate B-1, which could be reversibly converted to the allenylic-Pd intermediate B-2via isomerization. Ligand exchange between 3-hydroxyindole 1 and intermediate B-1 or B-2 affords intermediates C-1 or C-2, respectively. Due to the considerable energy barrier derived from computational studies, the pathway involving direct 1,1′-reductive elimination from intermediate C-2 is energetically disfavored and can be effectively precluded. The chiral carbon center of the indole skeleton is mainly established via enantioselective 3,3-reductive elimination from intermediate C-1 (Scheme 3). The reaction proceeds preferentially through transition state TS-D-1-Rvia a 3,3-elimination pathway, which features an energy barrier of 1.3 kcal mol−1 lower than that of the competing pathway viaTS-D-1-S, thereby kinetically favoring the formation of the chiral allene intermediate 4 with R-configuration. To elucidate the origin of enantioselectivity, non-covalent interactions between the substrate and the chiral ligand moiety in transition states TS-D-1-R and TS-D-1-S were analyzed using the Independent Gradient Model based on Hirshfeld partition (IGMH).17 Structural analyses indicate that the more favorable transition state TS-D-1-R is stabilized by enhanced hydrogen-bonding interactions between C–H bonds of the chiral ligand and the oxygen atom of the indole framework. These interactions contribute significantly to the reduced activation energy barrier of TS-D-1-R, making it 1.3 kcal mol−1 lower than that of TS-D-1-S. The direct electrophilic attack by the proton of benzoic acid on the palladium-activated allene intermediate D governs the enantioselectivity of the second stereocenter in the product. The pathway proceeding through the transition state TS-E-Z-R to form intermediate E exhibits an energy barrier of 12.8 kcal mol−1, representing the kinetically most favorable route. Compared to the analogous pathway through the transition state TS-E-Z-S, the formation of intermediate E proceeds with the lowest energy barrier (12.8 kcal mol−1 for TS-E-Z-Rvs. 15.2 kcal mol−1 for TS-E-Z-S). This distinct energy disparity substantiates the high enantioselectivity observed in the desired product 3aa. The activation energies for forming the olefin architecture with an E geometry via transition states TS-E-E-R and TS-E-E-S are 24.5 and 22.6 kcal mol−1, respectively. The disparity primarily stems from steric constraints imposed by the indole framework during the trajectory of electrophilic attack by benzoic acid. These values considerably exceed those required for the generation of the Z-olefin architecture. Given the computationally derived energy barriers, which are markedly elevated, this pathway can be confidently excluded.
Subsequently, the carboxylate anion in intermediate E can selectively abstract a hydrogen atom from either the indole nitrogen atom or the β-carbon atom of the ester group, leading to the formation of desired product 3aa or the conjugated diene byproduct 5, respectively. Overall, the formation of the conjugated diene byproduct proceeds reversibly via a lower energy barrier, enabling its generation under relatively mild reaction conditions and establishing it as the kinetically controlled product. In contrast, the pathway affording the desired product 3aa involves a moderately higher activation barrier, suggesting its dominance under thermodynamic control. These computational insights align consistently with the experimental outcomes (Scheme 2c).
Further study
To exemplify the synthetic utility of the current protocol, a gram-scale reaction of 1a and 2a was performed, and product 3aa was achieved in 85% yield with 94% ee and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (Scheme 4a). Ent-3aa was obtained in 84% yield and 94% ee by employing ent-L8 as the chiral ligand (Scheme 4b). Several transformations with respect to 3aa were then executed (Scheme 4c). Bromination and iodination of 3aa were efficiently achieved using N-bromosuccinimide (NBS) and N-iodosuccinimide (NIS), producing compounds 6 and 7 with yields of 89% and 93%, respectively. The allyl Grignard reagent could selectively attack the ketone moiety to form tertiary alcohol 8 in 86% yield. In addition, the treatment of 3aa with LiAlH4 resulted in the isolation of diol 9 in 87% yield with excellent diastereoselectivity. Selective reduction of the carbonyl group in 3aa to an alcohol 10 using NaBH4 allowed for a subsequent Mitsunobu reaction to form the corresponding amide derivative 11. Compounds 12 and 13 were easily constructed via the Beckmann rearrangement and DIBAL-H mediated ring-expansion rearrangement.18 In the presence of K2OsO2(OH)4 and NMO, bridged compound 14 was formed via dihydroxylation and intramolecular esterification. The absolute configurations of 8 and 9 were confirmed by X-ray diffraction. Moreover, a linear relationship between the ee values of L8 and the product 3aa was observed, which indicated that one molecule of the chiral ligand participated in controlling the stereoselectivity of the reaction (Scheme 4d).
1 dr (Scheme 4a). Ent-3aa was obtained in 84% yield and 94% ee by employing ent-L8 as the chiral ligand (Scheme 4b). Several transformations with respect to 3aa were then executed (Scheme 4c). Bromination and iodination of 3aa were efficiently achieved using N-bromosuccinimide (NBS) and N-iodosuccinimide (NIS), producing compounds 6 and 7 with yields of 89% and 93%, respectively. The allyl Grignard reagent could selectively attack the ketone moiety to form tertiary alcohol 8 in 86% yield. In addition, the treatment of 3aa with LiAlH4 resulted in the isolation of diol 9 in 87% yield with excellent diastereoselectivity. Selective reduction of the carbonyl group in 3aa to an alcohol 10 using NaBH4 allowed for a subsequent Mitsunobu reaction to form the corresponding amide derivative 11. Compounds 12 and 13 were easily constructed via the Beckmann rearrangement and DIBAL-H mediated ring-expansion rearrangement.18 In the presence of K2OsO2(OH)4 and NMO, bridged compound 14 was formed via dihydroxylation and intramolecular esterification. The absolute configurations of 8 and 9 were confirmed by X-ray diffraction. Moreover, a linear relationship between the ee values of L8 and the product 3aa was observed, which indicated that one molecule of the chiral ligand participated in controlling the stereoselectivity of the reaction (Scheme 4d).
Conclusions
In summary, we have developed an unprecedented Pd-catalyzed sequential hydroalkylation and hydroamination of 1,3-enynes with 3-hydroxyindoles. This redox-neutral protocol offers an efficient approach for the synthesis of optically pure pyrido[1,2-a]indoles and derivatives bearing one tertiary stereocenter and one quaternary stereocenter. This reaction proceeds in a high atom- and step-economic manner with good functional group compatibility. Preliminary mechanistic investigations suggest that the transformation proceeds via an intermolecular enyne hydroalkylation pathway to form an allene intermediate. Subsequent intramolecular hydroamination of the allene intermediate occurs via an axial-to-center chirality transfer process. We believe this research will inspire and stimulate efforts for designing more innovative sequential hydrofunctionalization cyclization reactions, thereby providing a toolkit for the synthesis of chiral complex molecules, and driving the advancement of organic green chemistry. Computational studies were conducted to probe the origin of the enantioselectivities. Further investigations on other catalytic asymmetric sequential hydrofunctionalization reactions are currently underway in our laboratory.Author contributions
Q. L., R. W. and T. W. contributed equally to this work.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed experimental procedures and characterization data for new compounds. See DOI: https://doi.org/10.1039/d5gc05558j.CCDC 2268989 (3aa), 2321444 (8) and 2321211 (9) contain the supplementary crystallographic data for this paper.19a–c
Acknowledgements
The authors acknowledge generous financial support from the National Natural Science Foundation of China (22371299), the Natural Science Foundation of Jiangsu Province (BK20242068 and BK20251564), the Open Project of State Key Laboratory of Natural Medicines (SKLNMKF202401), the China Postdoctoral Science Foundation (2024M763657), the Postdoctoral Fellowship Program of CPSF (GZB20250246), the Project Program of the State Key Laboratory of Natural Medicines (SKLNMZZ202211), the Fundamental Research Funds for the Central Universities (2632024ZD09 and 2632025PY03), the Jiangsu Funding Program for Excellent Postdoctoral Talent (2025ZB316), and the Innovation and Entrepreneurship (Shuangchuang) Program of Jiangsu Province (2024). We are also grateful to the High-Performance Computing Center of Nanjing University for performing the numerical calculations in this paper on its blade cluster system.References
- (a) J. H. Clark, Green Chem., 2006, 8, 17 RSC; (b) I. T. Horváth and P. T. Anastas, Chem. Rev., 2007, 107, 2169 CrossRef PubMed; (c) R. Noyori, Nat. Chem., 2009, 1, 5 Search PubMed; (d) P. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39, 301 RSC; (e) P. J. Dunn, Chem. Soc. Rev., 2012, 41, 1452 RSC.
- For selected reviews, see: (a) L. Huang, M. Arndt, K. Gooßen, H. Heydt and L. J. Gooßen, Chem. Rev., 2015, 115, 2596 CrossRef CAS PubMed; (b) P. Koschker and B. Breit, Acc. Chem. Res., 2016, 49, 1524 CrossRef CAS PubMed; (c) J. Chen, J. Guo and Z. Lu, Chin. J. Chem., 2018, 36, 1075 CrossRef CAS; (d) G. Li, X. Huo, X. Jiang and W. Zhang, Chem. Soc. Rev., 2020, 49, 2060 RSC; (e) J. Li and Y. Shi, Chem. Soc. Rev., 2022, 51, 6757 Search PubMed; (f) Y. Li, X. Lu and Y. Fu, CCS Chem., 2024, 6, 1130 CrossRef CAS.
- For selected reviews, see: (a) J. Chen and Z. Lu, Org. Chem. Front., 2018, 5, 260 RSC; (b) Y. He, J. Chen, X. Jiang and S. Zhu, Chin. J. Chem., 2022, 40, 651 CrossRef CAS; (c) X.-Y. Sun, B.-Y. Yao, B. Xuan, L.-J. Xiao and Q.-L. Zhou, Chem. Catal., 2022, 2, 3140 CAS; (d) W. Zhao, H.-X. Lu, W.-W. Zhang and B.-J. Li, Acc. Chem. Res., 2023, 56, 308 CrossRef CAS PubMed. For selected examples, see: (e) A. L. Rez-nichenko, H. N. Nguyen and K. C. Hultzsch, Angew. Chem., Int. Ed., 2010, 49, 8984 CrossRef CAS PubMed; (f) K. Manna, S. Xu and A. D. Sadow, Angew. Chem., Int. Ed., 2011, 50, 1865 CrossRef CAS PubMed; (g) H.-L. Teng, Y. Luo, B. Wang, L. Zhang, M. Nishiura and Z. Hou, Angew. Chem., Int. Ed., 2016, 55, 15406 CrossRef CAS PubMed; (h) Y. Xi and J. F. Hartwig, J. Am. Chem. Soc., 2016, 138, 6703 CrossRef CAS PubMed; (i) Y. Xi, S. Ma and J. F. Hartwig, Nature, 2020, 588, 254 CrossRef CAS PubMed; (j) F. Zhou, Y. Zhang, X. Xu and S. Zhu, Angew. Chem., Int. Ed., 2019, 58, 1754 CrossRef CAS PubMed; (k) Y. He, C. Liu, L. Yu and S. Zhu, Angew. Chem., Int. Ed., 2020, 59, 9186 CrossRef CAS PubMed; (l) X. Ren, Z. Wang, C. Shen, X. Tian, L. Tang, X. Ji and K. Dong, Angew. Chem., Int. Ed., 2021, 60, 17693 CrossRef CAS PubMed; (m) T. Qin, G. Lv, H. Miao, M. Guan, C. Xu, G. Zhang, T. Xiong and Q. Zhang, Angew. Chem., Int. Ed., 2022, 61, e202201967 CrossRef CAS PubMed; (n) H. S. Slocumb, S. Nie, V. M. Dong and X.-H. Yang, J. Am. Chem. Soc., 2022, 144, 18246 CrossRef CAS PubMed; (o) W. Zhao, K.-Z. Chen, A.-Z. Li and B.-J. Li, J. Am. Chem. Soc., 2022, 144, 13071 CrossRef CAS PubMed.
- For selected reviews, see: (a) A. M. Haydl, B. Breit, T. Liang and M. J. Krische, Angew. Chem., Int. Ed., 2017, 56, 11312 CrossRef CAS PubMed; (b) J. Chen, W.-T. Wei, Z. Li and Z. Lu, Chem. Soc. Rev., 2024, 53, 7566 RSC. For selected examples, see: (c) Q.-A. Chen, Z. Chen and V. M. Dong, J. Am. Chem. Soc., 2015, 137, 8392 Search PubMed; (d) P. Koschker, M. Kähny and B. Breit, J. Am. Chem. Soc., 2015, 137, 3131 CrossRef CAS PubMed; (e) F. A. Cruz and V. M. Dong, J. Am. Chem. Soc., 2017, 139, 1029 CrossRef CAS PubMed; (f) F.-T. Sheng, S.-C. Wang, J. Zhou, C. Chen, Y. Wang and S. Zhu, ACS Catal., 2023, 13, 3841 CrossRef CAS; (g) A. Huang, H.-R. Xu, Z.-J. Yang, X.-S. Xue and Z.-T. He, Nat. Synth., 2025, 4, 1565 CrossRef CAS.
- For selected reviews, see: (a) R. Blieck, M. Taillefer and F. Monnier, Chem. Rev., 2020, 120, 13545 CrossRef CAS PubMed; (b) S. V. Sieger, I. Lubins and B. Breit, ACS Catal., 2022, 12, 11301 CrossRef CAS. For selected examples, see: (c) K. L. Butler, M. Tragni and R. A. Widenhoefer, Angew. Chem., Int. Ed., 2012, 51, 5175 CrossRef CAS PubMed; (d) M. L. Cooke, K. Xu and B. Breit, Angew. Chem., Int. Ed., 2012, 51, 10876 CrossRef CAS PubMed; (e) K. Xu, N. Thieme and B. Breit, Angew. Chem., Int. Ed., 2014, 53, 2162 CrossRef CAS PubMed; (f) C. Li, M. Kähny and B. Breit, Angew. Chem., Int. Ed., 2014, 53, 13780 CrossRef CAS PubMed; (g) A. M. Haydl, K. Xu and B. Breit, Angew. Chem., Int. Ed., 2015, 54, 7149 CrossRef CAS PubMed; (h) Z. Yang and J. Wang, Angew. Chem., Int. Ed., 2021, 60, 27288 CrossRef CAS PubMed; (i) M.-Q. Tang, Z.-J. Yang, A.-J. Han and Z.-T. He, Angew. Chem., Int. Ed., 2025, 64, e202413428 CrossRef CAS PubMed.
- For selected reviews, see: (a) G. J. P. Perry, T. Jia and D. J. Procter, ACS Catal., 2020, 10, 1485 CrossRef CAS; (b) N. J. Adamson and S. J. Malcolmson, ACS Catal., 2020, 10, 1060 CrossRef CAS; (c) A. Flaget, C. Zhang and C. Mazet, ACS Catal., 2022, 12, 15638 CrossRef CAS PubMed; (d) Y. C. Wang, J.-B. Liu and Z.-T. He, Chin. J. Org. Chem., 2023, 43, 2614 CrossRef CAS. For selected examples, see: (e) S.-Z. Nie, R. T. Davison and V. M. Dong, J. Am. Chem. Soc., 2018, 140, 16450 CrossRef CAS PubMed; (f) N. J. Adamson, K. C. E. Wilbur and S. J. Malcolmson, J. Am. Chem. Soc., 2018, 140, 2761 CrossRef CAS PubMed; (g) Q. Zhang, H. Yu, L. Shen, T. Tang, D. Dong, W. Chai and W. Zi, J. Am. Chem. Soc., 2019, 141, 14554 CrossRef CAS PubMed; (h) Q. Zhang, D. Dong and W. Zi, J. Am. Chem. Soc., 2020, 142, 15860 CrossRef CAS PubMed; (i) A. Y. Jiu, H. S. Slocumb, C. S. Yeung, X.-H. Yang and V. M. Dong, Angew. Chem., Int. Ed., 2021, 60, 19660 CrossRef CAS PubMed; (j) Q. Li, Z. Wang, V. M. Dong and X.-H. Yang, J. Am. Chem. Soc., 2023, 145, 3909 CrossRef CAS PubMed; (k) X.-X. Chen, H. Luo, Y.-W. Chen, Y. Liu and Z.-T. He, Angew. Chem., Int. Ed., 2023, 62, e202307628 CrossRef CAS PubMed; (l) S.-Q. Yang, A.-J. Han, Y. Liu, X.-Y. Tang, G.-Q. Lin and Z.-T. He, J. Am. Chem. Soc., 2023, 145, 3915 CrossRef CAS PubMed; (m) M.-Q. Tang, Z.-J. Yang and Z.-T. He, Nat. Commun., 2023, 14, 6303 CrossRef CAS PubMed; (n) Y.-C. Wang, Z.-X. Xiao, M. Wang, S.-Q. Yang, J.-B. Liu and Z.-T. He, Angew. Chem., Int. Ed., 2023, 62, e202215568 CrossRef CAS PubMed.
- For review, see: (a) L. Li, S. Wang, A. Jakhar and Z. Shao, Green Synth. Catal., 2023, 4, 124 CAS. For examples, see: (b) Y. Yang, I. B. Perry, G. Lu, P. Liu and S. L. Buchwald, Science, 2016, 353, 144 CrossRef CAS PubMed; (c) Y. Huang, J. del Pozo, S. Torker and A. H. Hoveyda, J. Am. Chem. Soc., 2018, 140, 2643 CrossRef CAS PubMed; (d) L. Bayeh-Romero and S. L. Buchwald, J. Am. Chem. Soc., 2019, 141, 13788 CrossRef CAS PubMed; (e) D.-W. Gao, Y. Xiao, M. Liu, Z. Liu, M. K. Karunananda, J. S. Chen and K. M. Engle, ACS Catal., 2018, 8, 3650 CrossRef CAS PubMed; (f) H. Tsukamoto, T. Konno, K. Ito and T. Doi, Org. Lett., 2019, 21, 6811 CrossRef CAS PubMed; (g) N. J. Adamson, H. Jeddi and S. J. Malcolmson, J. Am. Chem. Soc., 2019, 141, 8574 CrossRef CAS PubMed; (h) S.-Q. Yang, Y.-F. Wang, W.-C. Zhao, G.-Q. Lin and Z.-T. He, J. Am. Chem. Soc., 2021, 143, 7285 CrossRef CAS PubMed; (i) C. You, M. Shi, X. Mi and S. Luo, Nat. Commun., 2023, 14, 2911 CrossRef CAS PubMed.
- For selected reviews, see: (a) X. Zeng, Chem. Rev., 2013, 113, 6864 CrossRef CAS PubMed; (b) Z. Cheng, J. Guo and Z. Lu, Chem. Commun., 2020, 56, 2229 RSC. For selected examples, see: (c) A. Heutling, F. Pohlki, I. Bytschkov and S. Doye, Angew. Chem., Int. Ed., 2005, 44, 2951 CrossRef CAS PubMed; (d) Y. Lee, H. Jang and A. H. Hoveyda, J. Am. Chem. Soc., 2009, 131, 18234 CrossRef CAS PubMed; (e) S.-L. Shi and S. L. Buchwald, Nat. Chem., 2015, 7, 38 Search PubMed; (f) Z. Zuo, J. Yang and Z. Huang, Angew. Chem., Int. Ed., 2016, 55, 10839 CrossRef CAS PubMed; (g) J. Guo, X. Shen and Z. Lu, Angew. Chem., Int. Ed., 2017, 56, 615 Search PubMed; (h) J. Guo, B. Cheng, X. Shen and Z. Lu, J. Am. Chem. Soc., 2017, 139, 15316 Search PubMed; (i) J. Guo, H. Wang, S. Xing, X. Hong and Z. Lu, Chem, 2019, 5, 881 CrossRef CAS; (j) M. Hu and S. Ge, Nat. Commun., 2020, 11, 765 CrossRef CAS PubMed; (k) D.-W. Gao, Y. Gao, H. Shao, T.-Z. Qiao, X. Wang, B. B. Sanchez, J. S. Chen, P. Liu and K. M. Engle, Nat. Catal., 2020, 3, 23 CrossRef CAS PubMed; (l) J. Chen, X. Shen and Z. Lu, J. Am. Chem. Soc., 2020, 142, 14455 CrossRef CAS PubMed; (m) S. Jin, K. Liu, S. Wang and Q. Song, J. Am. Chem. Soc., 2021, 143, 13124 CrossRef CAS PubMed; (n) S. Jin, J. Li, K. Liu, W.-Y. Ding, S. Wang, X. Huang, X. Li, P. Yu and Q. Song, Nat. Commun., 2022, 13, 3524 CrossRef CAS PubMed; (o) S. Wang, K. Chen, J. Niu, X. Guo, X. Yuan, J. Yin, B. Zhu, D. Shi, W. Guan, T. Xiong and Q. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202410833 CAS; (p) H. Wang, X. Jie, Q. Chong and F. Meng, Nat. Commun., 2024, 15, 3427 CrossRef CAS PubMed.
- L. Li, S. Wang, P. Luo, R. Wang, Z. Wang, X. Li, Y. Deng, F. Peng and Z. Shao, Nat. Commun., 2021, 12, 5667 CrossRef CAS PubMed.
- W. Lu, Y. Zhao and F. Meng, J. Am. Chem. Soc., 2022, 144, 5233 CrossRef CAS PubMed.
- Q. Li, X. Fang, R. Pan, H. Yao and A. Lin, J. Am. Chem. Soc., 2022, 144, 11364 CrossRef CAS PubMed.
- B.-Y. Xie and Z.-T. He, ACS Catal., 2024, 14, 9742 CrossRef CAS.
- (a) D. Zhang, M. Li, J. Li, A. Lin and H. Yao, Nat. Commun., 2021, 12, 6627 CrossRef CAS PubMed; (b) Q. Li, J. Li, J. Zhang, S. Wu, Y. Zhang, A. Lin and H. Yao, Angew. Chem., Int. Ed., 2023, 62, e202313404 CrossRef CAS PubMed; (c) Q. Wu, Q. Zhang, S. Yin, A. Lin, S. Gao and H. Yao, Angew. Chem., Int. Ed., 2023, 62, e202305518 CrossRef CAS PubMed; (d) M. Xu, Q. Lu, B. Gong, W. Ti, A. Lin, H. Yao and S. Gao, Angew. Chem., Int. Ed., 2023, 62, e202311540 CrossRef CAS PubMed.
- (a) G. Bartoli, G. Bencivenni and R. Dalpozzo, Chem. Soc. Rev., 2010, 39, 4449 RSC; (b) B. P. Pritchett and B. M. Stoltz, Nat. Prod. Rep., 2018, 35, 559 RSC; (c) A. E. Nugroho, W. Zhang, Y. Hirasawa, Y. Tang, C. P. Wong, K. Kaneda, A. H. A. Hadi and H. Morita, J. Nat. Prod., 2018, 81, 2600 CrossRef CAS PubMed; (d) Y. Hong, Y. Y. Zhu, Q. He and S.-X. Gu, Bioorg. Med. Chem., 2022, 55, 116597 CrossRef CAS PubMed.
- S. Song and S. Ma, Chin. J. Chem., 2020, 38, 1233 CrossRef CAS.
- (a) I. Crouch, T. Dreier and D. Frantz, Angew. Chem., Int. Ed., 2011, 50, 6128 CrossRef CAS PubMed; (b) G. Cera, M. Lanzi, F. Bigi, R. Maggi, M. Malacriab and G. Maestri, Chem. Commun., 2018, 54, 14021 RSC.
- T. Lu and Q. Chen, J. Comput. Chem., 2022, 43, 539 CrossRef CAS PubMed.
- (a) H. Zhao, C. P. Vandenbossche, S. G. Koenig, S. P. Singh and R. P. Bakale, Org. Lett., 2008, 10, 505 CrossRef CAS PubMed; (b) Y. Iwama, K. Okano, K. Sugimoto and H. Tokuyama, Org. Lett., 2012, 14, 2320 CrossRef CAS PubMed; (c) S.-L. Cai, Y. Li, C. Yang, J. Sheng and X.-S. Wang, ACS Catal., 2019, 9, 10299 CrossRef CAS.
- (a) CCDC 2268989: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2g526y; (b) CCDC 2321444: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hxn9d; (c) CCDC 2321211: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hxdsm.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |