Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Combining cellulose substrates and perovskites in sustainable solar cells is possible: a systematic literature review offering realistic solutions
Joaquín
Valdez García
 *a,
Mahboubeh
Hadadian
*a,
Mahboubeh
Hadadian
 a,
Vidushi
Aggarwal
a,
Vidushi
Aggarwal
 a,
Sirius
Yli-Paavola
a,
Sirius
Yli-Paavola
 a,
Joice
Kaschuk
a,
Joice
Kaschuk
 b,
Riikka
Suhonen
b,
Riikka
Suhonen
 c,
Marja
Välimäki
c,
Marja
Välimäki
 c and
Kati
Miettunen
c and
Kati
Miettunen
 *a
*a
aDepartment of Mechanical and Materials Engineering, Faculty of Technology, University of Turku, Vesilinnantie 5, Turku FI-20500, Finland. E-mail: jvagar@utu.fi; kati.miettunen@utu.fi
bPhysical Chemistry and Soft Matter Group, Wageningen University & Research, Wageningen, the Netherlands
cVTT Technical Research Centre of Finland Ltd, Digital Technologies, Kaitoväylä 1, Oulu FI-90590, Finland
First published on 11th February 2026
Abstract
The aim of this article is to provide direction for the advancement of cellulose films as sustainable substrates for perovskite solar cells (PSCs). Cellulose, the most abundant biopolymer on Earth, represents a viable, renewable alternative to glass and synthetic polymers when subjected to appropriate modifications. It can be customized via crosslinking, plasticization, and functionalization to increase flexibility and solvent resistance while decreasing gas permeation, surface roughness, and thermal expansion. The adoption of cellulose can drive transformative changes in PSC processing, facilitating the integration of sustainable electrode materials and greener alternatives to toxic solvents, as well as the replacement of high-temperature treatments. Although the literature contains numerous solutions to specific challenges, these findings are scattered across different fields and must be critically assessed for PSC suitability. In this article, we critically review alternative fabrication methods and form a step-by-step multidisciplinary strategy to alter both cellulose and PSC fabrication protocols for the development of sustainable next-generation solar cells.
Green foundation1. Here we discussed the integration of cellulose substrates in perovskite solar cells, the improvement of cellulose films with some green additives, the replacement of perovskite solvents with greener options, and alternative fabrication methods to reduce solvent usage.2. We believe the integration of cellulose substrates in perovskite solar cells can improve the recyclability of solar energy devices, as well as accelerate the production of cheaper solar cells via printing and other wet deposition methods. 3. The perovskite solar cell field needs to anticipate and adapt to future needs through improved designs that focus on recyclability and the inclusion of renewable materials. Similarly, the cellulose field will have to adapt to the needs of the optoelectronics industry. In our review, we delineate how these two fields could come together through a green chemistry approach to retain their sustainability. |
Introduction
Solar energy harvesting technologies are crucial for procuring clean energy. To beat climate change and quench our need for electricity, vast quantities of solar panels need to be manufactured and deployed. Perovskite solar cells (PSCs) have emerged as alternatives to silicon thanks to their high-volume and cost-efficient production, their high efficiency, and low-levelized cost of energy.1 Their environmental, social, and economic sustainability is taking center-stage as they enter the market, necessitating rigorous attention to both material selection and assembly processes.The fabrication of PSCs involves depositing very thin layers (<1 µm) of materials with distinct optoelectronic functions onto a substrate that ranges from 50 µm to 2 mm in thickness. The substrate constitutes the bulk of the device, thereby playing a major role in the device's life-cycle environmental impact2 as well as in the recovery of environmentally hazardous and economically valuable components.3 Historically, a variety of materials have been employed as substrates in flexible solar cells, each with their own advantages and disadvantages (Table 1).
| Property | Glass | Metal foil | PET or PEN | Cellulose |
|---|---|---|---|---|
| a Requires modifications. | ||||
| Transparency | ✓ | ✗ | ✓ | ✓a |
| Flexibility | Flexible up to 100 µm | ✓ | ✓ | ✓a |
| Resistance to solvents used in PSC fabrication | ✓ | ✓ | ✓ | ✓a |
| Low surface roughness | ✓ | ✗ | ✓ | ✓a |
| Oxygen blocking | ✓ | ✓ | ✗ | ✗a |
| Water vapor blocking | ✓ | ✓ | ✗ | ✓a |
| Low coefficient of thermal expansion (CTE) | ✓ | ✓ | ✗ | ✓a |
| Recyclability in photovoltaics | ✓ | ✓ | ✗ | ✓ |
| Price | $$$ | $$ | $ | $ |
Glass is desirable because of its excellent optical transmittance, which allows the majority of incident light to enter the active layer; its chemical inertness regarding the solvents used in PSC fabrication; its mechanical robustness; its ability to withstand high temperatures; and it has low oxygen and water vapor transmission rates (OTR and WVTR, respectively).4 However, its main drawback is rigidity, which renders it unsuitable for high-volume roll-to-roll (R2R) printing techniques. While flexible glass (FG) has been utilized to fabricate solar cells, its adoption is hindered by its inherent brittleness,5 high cost,6 and the limited market for solar glass waste following recycling.7,8
Metal foils are inherently highly conductive, excellent at blocking oxygen and moisture, highly flexible, and have shown some potential in emerging solar cells.9,10 Nonetheless, their opacity and high surface roughness represent major impediments.5 Their opacity requires that the top contact be transparent and flexible, which limits the choice of top electrode materials.11 Furthermore, their surface roughness is in the order of microns, which requires planarization (i.e., polishing) or the use of passivation layers.5 Synthetic polymers, such as polyethylene terephthalate (PET) and polyethylene naphthalate (PEN), are the preferred materials for R2R fabrication because of their high transparency and flexibility, low cost, and low weight. Despite their positive properties, synthetic polymer substrates are limited by thermal and solvent instability and require low oxygen and water vapor barrier layers to block gas and moisture.5
Major advancements have been made in using cellulose and its derivatives to produce films with traits desirable for solar cell substrates.12,13 Cellulose materials can already reach transparency and surface roughness levels comparable to glass, and similar mechanical properties to those of synthetic polymers with an increased thermal stability.14 Cellulose is a nontoxic, low-cost material and the most abundant biopolymer on the planet; it can be obtained from various renewable sources and side streams. The modification of cellulose for optoelectronics begins with achieving high transparency. When unmodified, in their macroscopic structure, cellulose fibers form long networks that form light-scattering pores, resulting in opaque, white materials like paper and cotton.15 These materials can be transformed into transparent films through several strategies, including modification and defibrillation. The most consolidated and reliable approach is to produce cellulose derivatives, which involves replacing one or more of the three reactive hydroxyl groups (–OH) on the cellulose chain with a functional group (esters, ethers, etc.; see Fig. 1).16,17 The degree of substitution (DS), which denotes the number of –OH groups substituted, can range from 0 to 3 and largely dictates the properties of the resulting material.18
 | ||
| Fig. 1 Chemical structure of various cellulose derivatives. R can be a hydrogen or one of the groups around the cellulose molecule, reproduced from ref. 19 with permission from the American Chemical Society,19 Copyright 2022. | ||
Another critical aspect of cellulose is its solubility and interaction with solvents. The hierarchical and recalcitrant structure of unmodified cellulose originates from the semicrystalline structure and extensive intra- and intermolecular hydrogen bond network between chains, which prevents the dissolution of cellulose in most commonly used solvents (ethanol, acetone, etc.).17 These molecular interactions are altered when cellulose is modified, resulting in cellulose derivatives that are soluble in common solvents, depending on the functional group and DS. Furthermore, the cellulose hydrophilicity and its amorphous regions facilitate strong water bonding and trapping, allowing unmodified cellulose materials to absorb larger quantities without being dissolved.20 Notably, cellulose derivatives may exhibit divergent swelling behaviors because certain functional groups have high levels of hydrophobicity compared to those in unmodified cellulose.
In the cellulose field there is a concept known as the “cellulose gap”. Cotton demand in textile production is very high; cotton production is not expected to meet this demand, as its production is very water intensive, and the textile industry is working on developing cellulose fibers that can complement cotton.21 Conversely, cellulose sourced from bacteria is quite costly and is used in specialized applications like biomedicine.22 Currently, wood is the biggest alternative to cotton due to low costs and large availability.21 As such, most of the commercially available cellulose to make films, membranes, and hydrogels is sourced from wood. Unlike in fibers, nanofibers,23 and nanocrystals,24 after derivatizing and functionalizing polymeric cellulose and cellulose nanocrystals for transparent films in optoelectronic applications, its source has little impact other than the degree of polymerization (DP). At this point, its functional groups, its DS, and whether or not it has been crosslinked or plasticized have a much greater impact on its properties.
From a processability standpoint, unmodified cellulose cannot be processed in a manner similar to synthetic polymers due to its high glass transition temperatures: the amount of thermal energy required to break the intermolecular hydrogen bonds of unmodified cellulose is so high that cellulose degrades before melting.18 One of the strategies used to produce cellulose substrates involves the dissolution and regeneration (solvent removal) of films.25 The properties of these films depend on a variety of factors, ranging from their new functional groups, their DS and size of the chain, their drying conditions, the surface on which they were cast, and their porosity, among others. Controlling these parameters is key to making viable cellulose substrates for solar cells.
Two properties stand out when comparing cellulose films to currently used synthetic polymers: their solvent resistance and low thermal expansion. During fabrication, solar cell layers expand and contract along with the substrate, which leads to cracks that can reduce device performance and stability. Cellulose has a very low coefficient of thermal expansion (CTE) (0.1–158 ppm K−1 (ref. 26 and 27)) compared with synthetic polymers (12–122 ppm K−1 (ref. 28–30)), resulting in a more consistent PSC fabrication. Furthermore, some cellulose derivatives exhibit strong resistance to many solvents commonly used in PSC fabrication (see section Solvent resistance), while others can be crosslinked to obtain similar durability.31
A seldom-discussed advantage of cellulose is that it facilitates the recycling of solar cells. Glass and encapsulant resins are among the biggest hindrances associated with solar panel recycling, because they complicate separating valuable materials such as silver and high-quality silicon from shredded solar panel waste.32 Glass, PET, and PEN coated with a thin layer of indium tin oxide (ITO) are the most commonly used substrates in PSCs. ITO accounts for most of a PSC's carbon emissions due to the energy-intensive vacuum deposition process required.33 It can be recovered from glass using a chemical bath,8 but panel encapsulation makes this difficult.34 In contrast to glass substrates, PET and PEN can be easily burned in pyrolysis processes to extract valuable metals.3 However, aside from bioPET,35 they are derived from fossil sources. Thin cellulose substrates further concentrate the value of solar cell materials, making recycling economically viable at the end of the product's lifespan.3 Since solar cells are meant to be encapsulated to maximize stability and efficiency over time, cellulose is not expected to degrade since it is not in an environment with bacteria, soil, and moisture.19 Therefore, the recycling route for solar cells made on renewable cellulose is to degrade it only at the end of the device lifetime to allow for material recovery, either by burning it or by degrading it through photochemical means.19
As polymers are the most popular substrate materials when making flexible devices, most research is focused on the same. Notably, cells with a power conversion efficiency (PCE) lower than 20% are made of MAPbI3 perovskite, one of the first perovskite compositions used, and higher efficiencies are reached by using perovskites with more complicated stoichiometries and compositions. While there is still room for improvement, Li et al.'s work demonstrated that cellulose films are viable substrates for PSCs and can achieve 13.1% efficiency.36 Nevertheless, physical vapor deposition techniques are costly to implement and require high initial investments; cellulose, being the quintessential printing material, calls for solution-based R2R-compatible deposition methods. Table 2 provides a summary of high-performing flexible PSCs.
| Substrate | Structure | PCE [%] | J SC [mA cm−2] | V OC [V] | FF [%] | Active area [cm2] | Ref. |
|---|---|---|---|---|---|---|---|
| FG | FG/ITO/SnO2/m-TiO2/MAPbI3/Spiro-OMeTAD/Au | 15.8 | 22.83 | 1.09 | 79.1 | 0.08 | 37 |
| Titanium foil | Ti/TiO2/MAPbI3/PTAA/Graphene/PDMS | 15.0 | 18.70 | 1.08 | 74.4 | 1 | 38 |
| Steel foil | Stainless steel/ITO/NiOx/MeO-2PACz/(CsFAMA) ((CsPbI3)0.13(FAPbI3)0.85(MAPbBr3)0.02)/DMI/C60/BCP/Ag | 20.70 | 23.30 | 1.11 | 80.15 | 1.012 | 39 |
| PET | PET/ITO/CBD-SnO2/(RbFAMA)Pb(ICl)3/Spiro-OMeTAD/Au | 25.65 | 25.90 | 1.19 | 83.39 | 1 | 40 |
| PEN | PEN/ITO/SnO2-DNS//(CsFAMA)Pb(BrICl)3/Spiro-OMeTAD/Au | 23.16 | 24.85 | 1.13 | 82.2 | 1 | 41 |
| PI | PI/ITO/PF-doped SnO2/(CsFAMA)Pb(BrICl)3/Spiro-OMeTAD/Au | 23.31 | 24.87 | 1.16 | 80.80 | 1 | 42 |
| Cellulose | Cellophane/TiO2/Ag/TiO2/C60/CPTA/MAPbI3/Spiro-OMeTAD/Au | 13.19 | 17.81 | 1.04 | 71.30 | Unspecified, <1 | 36 |
The PSC fabrication process imposes many requirements on cellulose (see Table 1). Many strategies are available to achieve these properties, but these are typically found in fields very different from photovoltaics, such as food packaging, drug delivery, and battery membrane technologies, which have varying requirements for cellulose films. Furthermore, their applicability requires a critical assessment. This article fills the existing gap in the literature by reviewing relevant and potential solutions. The aim of our analysis is to shed light on the modifications required for cellulose to work as a good substrate in PSCs and on ways to adapt PSC fabrication methods to cellulose films, addressing both cellulose and solar cell audiences. The article begins with an analysis of PSC substrate materials and fabrication processes currently used, followed by a strategy on how to modify both the cellulose films and the typical PSC processing routes. As the need for clean energy sources increases, the more our devices need to adapt to sustainable designs. Understanding how to adapt biobased alternatives becomes critical for the development of next-generation solar cells.
The process of making a solar cell
A PSC is a stack of functional layers (Fig. 2); what layers are included and how they are arranged depends on many factors. The first component to consider is the active layer, and its bandgap dictates which charge transport layers (CTLs) can be used and how they need to be modified to match their band levels. Accordingly, the deposition sequence depends on the CTL deposition methods. Since the perovskite layer is vulnerable to solvents and high temperatures, it is advisable to first deposit the CTL that requires solvents and temperatures incompatible with the perovskite. The PSC structure is defined by whether the electron transport layer (ETL) or the hole transport layer (HTL) is deposited first, namely NIP (or regular) or PIN (or inverted), respectively.43PSC fabrication is generally done on glass or polymeric substrates covered with a transparent conductive oxide (TCO). The methods used greatly depend on the fabrication scale; spin coating on glass/fluorine-doped tin oxide (FTO) or glass/ITO is favored at a laboratory scale, while coating or printing on PET/ITO or PEN/ITO is more common in high-volume industrial fabrication. Nevertheless, the deposition steps for both are quite similar (Fig. 3). The first step is the TCO patterning, normally done through chemical etching or laser patterning, although this process may be skipped in certain situations.44 Next, the substrate is washed in different solvents to remove any residues that could interfere with the first CTL deposition. The substrate is treated with UV-ozone (UV-O3) or plasma to increase its wettability, and the first CTL is immediately deposited, typically followed by thermal annealing. The active layer is then deposited, followed by another annealing step to crystallize the perovskite. Once the substrates have cooled down enough, the next CTL is deposited, followed by the thermal evaporation of the top electrode.45 Depending on the materials used and the associated process, some steps are carried out inside a nitrogen-filled glovebox to prevent oxygen and moisture from interfering with the layers. Current research is focused on adapting PSC production to less restrictive environments and moving away from expensive vacuum-based, energy-intensive fabrication methods and special atmospheres, and toward wet deposition methods that are easier to implement.
 | ||
| Fig. 3 Schematic of typical fabrication processes for PSCs on glass (upper panel) and the proposed or modified processes designed for compatibility with cellulose substrates (lower panel). | ||
The regular wet-method processing route presents many challenges when using unmodified cellulose films. First, the acid to etch ITO could damage cellulose. Many researchers have proposed the use of metallic nanowires instead, considering ITO's poor sustainability, but this approach requires pre-patterning them, as they cannot be removed if embedded. Second, solvent washing can be problematic; some solvents may cause the cellulose fibers to redisperse or may infiltrate the film and cause it to swell. While UV-O3 treatments break the carbon bonds in organic matter to volatilize contaminants on inorganic surfaces, this can damage cellulose films and make them more reactive, increasing solvent absorption. Third, additional plasticizers may be required to ensure that cellulose has sufficient flexibility during the PSC fabrication process. These plasticizers degrade at around 120 °C,46 which limits the thermal steps in the process. Lastly, thermal annealing can decrease the mechanical properties of cellulose, making it more difficult to handle.47
Considering the above, we devised a PSC processing route on cellulose substrates based on strategies scattered across different fields to further advance the integration of biobased materials into next-generation solar cells (Fig. 3).
Step 1: preparing the cellulose suspensions and solutions
Before casting cellulose films, the cellulose solutions must be homogeneously produced and altered to provide cellulose with additional properties to endure PSC fabrication. The first thing to consider is the solvents used. Two options in this regard are (i) to employ a cellulose type compatible with the selected solvent or (ii) to crosslink the cellulose chains to avoid redispersion. Crosslinking can also decrease the film's thermal expansion. Next, cellulose can be mixed with plasticizers to increase the resulting film's mechanical strength and flexibility. Finally, different additives can be mixed in the cellulose matrix to give the resulting films gas-blocking properties for PSC protection.Table 3 shows how commonly used solvents in solar cell fabrication affect cellulose films. The effect 15 different solvents have on cellulose films is judged by their visual appearance. After rubbing a solvent-soaked cotton swab on each film, cellulose redisperses and resettles when dry. Solvent affects the films as they can break, buckle, and roughen; these effects can prevent the deposition of different layers during PSC fabrication, especially the first ones. The test was adapted from the ASTM D5402 to judge the quality of organic coatings. The film preparation description along with pictures for every entry, in line with Fig. 4, can be found in the SI, Table S1.
| Solvent | Carboxymethylated cellulose (CMC) | Cellulose nanocrystals (CNC) | Cellulose acetate (CA) | Ethyl cellulose (EC) | Hydroxyethyl cellulose (HEC) | Cellophane |
|---|---|---|---|---|---|---|
| Water | ✗ | ✗ | ✓ | ✓ | ✓ | ✓ |
| Ethanol | ✓ | ✓ | ✓ | ✗ | ✗ | ✓ |
| Isopropanol | ✓ | ✓ | ✓ | ✗ | ✗ | ✓ |
| Acetone | ✓ | ✓ | ✗ | ✗ | ✓ | ✓ |
| Methanol | ✓ | ✓ | ✓ | ✓ | ✗ | ✓ |
| γ-Valerolactone (GVL) | ✓ | ✓ | ✗ | ✓ | ✓ | ✓ |
| Dimethyl sulfoxide (DMSO) | ✓ | ✓ | ✗ | ✗ | ✓ | ✓ |
| γ-Butyrolactone (GBL) | ✓ | ✓ | ✗ | ✗ | ✓ | ✓ |
| Dimethylformamide (DMF) | ✓ | ✓ | ✗ | ✗ | ✓ | ✓ |
| Chloroform | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| Chlorobenzene | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| 1,2-Dichlorobenzene | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| Toluene | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| Diethyl ether | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
| Acetonitrile | ✓ | ✓ | ✓ | ✗ | ✓ | ✓ |
The first solar cell layers are prone to damaging the cellulose directly. Particular attention needs to be paid to the solvent compatibility of the first layers that could come in contact with cellulose. For example, the first layer may be too thin or too porous and thus allow solvents to reach the substrate and damage it. A type of cellulose that can withstand all the solvents used should ideally be chosen, or the cellulose should be crosslinked to increase its solvent resistance.
In the case of cellulose, a molecule or a short polymer chain bonds two parallel cellulose chains and prevents them from redispersing or redissolving, thus granting cellulose films solvent resistance and/or modifying their properties. Crosslinking occurs along the active sites on the cellulose chains, which can be hydroxyl groups or other functional groups, and depending on their nature, a catalyst might be required. For instance, 1,2,3,4-butanetetracarboxylic acid (BTCA) has been used to crosslink cotton fibers and thereby obtain cotton fabrics with wrinkle and fire resistance. It can also be used to increase the water resistance of cellulose films (Fig. 5). Cellulose nanofiber (CNF) films crosslinked with BTCA remained stable in water even when submerged for 5 months. Sodium hypophosphite (SHP) is used as a catalyst for hydroxyl radical reactions in both cellulose and BTCA.49
 | ||
| Fig. 5 Crosslinked cellulose with BTCA, adapted from ref. 49 with permission from Elsevier.49 Copyright 2021. | ||
A similar approach can be taken for hybrid films. For example, crosslinking CMC with cornstarch using epichlorohydrin (ECH) results in water-resistant films. ECH reacted and simultaneously bonded with CMC and starch active sites to form 3-propyleneglycol, which acts as a plasticizer that increases the resulting films’ flexibility.50 However, ECH is quite toxic, and a safer alternative is required. Citric acid is another small molecule that has been used to crosslink cellulose. Transparent films made of regenerated bacterial cellulose can be crosslinked with citric acid without a catalyzer, although this process is carried out in toxic N,N-dimethylacetamide.51 This solvent can potentially be replaced by safer ionic liquids or deep eutectic solvents.52
Crosslinking can also be catalyzed using UV light. EC can be crosslinked with ethoxylated trimethylolpropane triacrylate through UV light to obtain films with increased solvent resistance and tensile strength. These films have been used as substrates in organic solar cells (OSCs) with almost 19% efficiency.31
| Material | CTE [ppm K−1 or 10−6 °C−1] | Testing conditions | Ref. |
|---|---|---|---|
| FG | 0.31 | Unspecified | 54 |
| Steel foil | 16 | Taken from the literature | 56 |
| Titanium foil | 8.9 | Taken from the literature | 56 |
| PET | 15–117 | A thermal expansion analyzer (TMA 402 F3, NETZSCH, Germany) was used to evaluate the CTE of the TSV sample at a heating rate of 10 °C min−1 in the range of 10–100 °C under nitrogen flow | 28–30 |
| Commercial sample | |||
| The CTEs of samples measuring 20 mm long and 20 mm wide were evaluated using a thermal expansion analyzer (NETZSCH DIL 402 C, Germany). The measurements were conducted three times from 25 °C to 100 °C by elevating the temperature at a rate of 10 °C min−1 under an inert gas atmosphere and taking the average value | |||
| PEN | 13–20 | Commercial sample | 29 and 53 |
| Polyimide (PI) | 12–60 | A thermal expansion analyzer (TMA 402 F3, NETZSCH, Germany) was used to evaluate the CTE of the TSV sample at a heating rate of 10 °C min−1 in the range of 10–100 °C under nitrogen flow | 28, 29 and 53 |
| Commercial sample | |||
| Polycarbonate (PC) | 60–122 | Commercial sample | 29 and 30 |
| The CTEs of samples measuring 20 mm long and 20 mm wide were evaluated using a thermal expansion analyzer (NETZSCH DIL 402 C, Germany). The measurements were conducted three times from 25 °C to 100 °C by elevating the temperature at a rate of 10 °C min−1 under an inert gas atmosphere and taking the average value | |||
| Cellulose microfiber composite | 0.17 | Measured with a TMA-SS120S (Seiko Instruments). Heating rate of 10 °C min−1. The specimen with an original length of 15 mm was subjected to a uniaxial stress of 0.12 MPa | 26 |
| Microcrystalline cellulose crosslinked with epichlorohydrin | 6.9 | Measured with a thermomechanical analyzer (TMA-SS6100, Seiko Instruments) | 55 |
| Self-organized CNC (self-organized/shear-organized) | ∼25/9–158 | Digital image correlation (DIC) methodology used. Images were captured at 25, 40, 60, 80, and 100 °C. A 10 min stabilization time was allowed at each temperature before capturing images | 27 |
| CNC | 2.7 | Atmospheric condition of nitrogen and heating rate of 5 °C min−1, calculated over the specified regions from 50 to 150 °C and each 5 °C range | 57 |
| CNC/CNF | 11.86–17.65 | The temperature used in CTE tests was increased from 20 to 120 °C at a rate of 3 °C min−1 | 58 |
| CNF | 8.5 | Measured using a thermomechanical analyzer (TMA/SS6100, SII nanotechnology Inc.). Specimens were 25 mm long and 3 mm wide with a 20 mm span. The measurements were carried out three times with a heating rate of 5 °C min−1 under a nitrogen atmosphere in tensile mode under a load of 3 g. The CTE values were determined as the mean values at 20–150 °C in the second run | 59 |
| CMC | 38 | Measured using a TMA Q400 | 60 |
| CA | 55.3 | Commercial sample | 53 |
| Cellophane (flow direction/transverse direction) | 18.2/33.6 | Commercial sample | 53 |
Although CTE is an important property that highlights the suitability of cellulose as a substrate material for optoelectronics, it is seldom reported in the literature and requires further research. It is unclear whether the cellulose moisture content has an effect on CTE and, if so, to what extent. Table 4 also includes the testing conditions used to obtain CTE values. There seems to be no apparent standard way to measure CTE between different materials. It would be useful to report the percentage of thermal expansion similar to how Nishino et al. do in their work.26 This approach would be useful to determine if there is a difference in CTE due to water loss and plasticizer degradation.
| Material | Plasticizer | Tensile strength (MPa) | WVP (g µm (m2day)−1) (23 °C, 50% RH) | Oxygen permeability (OP) cm3 µm m−2 day−1 (23 °C, 50% RH) | Ref. | |||
|---|---|---|---|---|---|---|---|---|
| Without plasticizer | With plasticizer | Without plasticizer | With plasticizer | Without plasticizer | With plasticizer | |||
| CNC | Glycerol | 62.3 | 32.9 | 2601 | 2669 | 134 | 494 | 65 |
| CNC | Sorbitol | 62.3 | 39.2 | 2601 | 399 | 134 | 153 | 65 |
| CNC | Maltitol | 62.3 | 47.5 | 2601 | 97 | 134 | 29 | 65 |
| CNC | Maltitol | 64.2 ± 10.9 | 32.4 ± 2.3 | 6.06 × 10−11 g Pa−1 s−1 m−1 | 3.67 × 10−12 (g Pa−1 s−1 m−1) | 21.68 cm3 µm m−2 day−1 atm−1 | 4.75 cm3 µm m−2 day−1 atm−1 | 66 |
| CA | Glycerol and triethyl citrate | 34 | 8 | 1.0 × 10−4 g Pa−1 s−1 m−1 | 7.0 × 10−5 g Pa−1 s−1 m−1 | — | — | 67 |
| Regenerated cellulose crosslinked with Ca2+ | Glycerol | 4.5 ± 4 | 0.8 ± 4 | 1.48 ± 3 × 10−8 g Pa−1 s−1 m−1 | 10.12 ± 9.65 × 10−8 g Pa−1 s−1 m−1 | — | — | 63 |
| Regenerated cellulose crosslinked with Ca2+ | Sorbitol | 4.5 ± 4 | 0.3 ± 1 | 1.48 × 10−8 g Pa−1 s−1 m−1 | 1.08 ± 10 × 10−10 g Pa−1 s−1 m−1 | — | — | 63 |
Glycerol is a well-known plasticizer in the cellulose field due to its chemical similarity. It interacts with hydroxyl radicals and reduces hydrogen bonding between cellulose chains, which reduces the interactions between chains when the films are bent, thereby increasing their flexibility.61 The higher the plasticizer concentration in a film, the more space it occupies between the polymer chains and the greater the flexibility.62 Glycerol is compatible with many cellulose derivatives, such as CMC,61 CA,62 microcrystalline cellulose,46,63 and CNC.64 The effects of glycerol's interactions with hydroxyl groups vary depending on the cellulose radical groups and DS.
While glycerol is a good plasticizer, it is also known for increasing OTR and WVTR. Research showed that mixing glycerol with other polyols, such as maltitol and sorbitol, can greatly decrease CNC films’ gas permeability (from 2601 to 97 g µm m−2 day−1 for water and from 134 to 20 cc µm m−2 day−1 for oxygen) while maintaining good flexibility.65 Maltitol, in particular, was proved to increase CNC films’ elongation at break to levels similar to PET and other plastics, and it has the added benefit of increasing optical transmittance while decreasing surface roughness.66
Different cellulose types have been combined to study the resulting synergistic effects on their barrier and mechanical properties. Fernández-Santos et al. tested CNC films with varying proportions of CNF and CMC.69 At 90% RH, CNC films with more than 40% CMC showed increased resistance to water vapor, hence lower WVTR values which rapidly reached those of pure CMC of 3.38 g m−2 day−1. CMC contributed to better water resistance by forming dense, compact networks, whereas the free –OH groups and crystals of CNF led to higher water vapor permeation and reduced barrier water properties. Similar findings on the effects of CMC and CNF were also obtained by another research group.70 Pure cellulose films could be fine-tuned to obtain oxygen and water barrier properties that exponentially increase PCE and device stability. These properties are especially desirable for PSC application in humid conditions.
CMC films combined with sodium montmorillonite (Na-MMT) nanoclay exhibited a 39% reduction in water vapor permeability compared to pristine CMC films.71 This enhanced water barrier property could be attributed to the formation of strong hydrogen bonds between the –OH groups of CMC and Na-MMT, which hinder the availability of –OH groups for moisture absorption. The incorporation of TiO2, which has inherent UV-shielding properties, led to a further decrease in water vapor permeability by 50%. CMC films comprising 5 wt% Na-MMT and 1 wt% TiO2 had good tensile strength (6.18 ± 2.01 MPa) and Young's modulus (28.93 ± 5.74 MPa) but became non-transparent. Thus, nanocomposite CMC films would require further optimization to be employed as substrates in PSCs. In our previous work, we explored the use of montmorillonite clay (MMT) to produce transparent films (60–80% at 550 nm) with improved gas barrier properties. We found that MMT induces some improvements in the barrier properties,72 but they are far from what is needed for stable PSCs; the required levels for PSCs range from 10−3 to 10−6 g m−2 day−1 for WVTR, and from 10−2 to 10−5 cm3 m−2 day−1 for OTR.73 Thus, a separate protective coating is needed to achieve acceptable gas barrier levels (see Coatings to increase barrier properties section). WVTR and OTR values for some PSC substrate materials are given in Table 6.
| Material | Thickness [µm] | WVTR [g (m2 day)−1] | OTR [cm3 (m2 day)=] | Ref. | |
|---|---|---|---|---|---|
| Without modification | With modification | ||||
| FG (NEP G-Leaf™) | 30–700 | — | Below 1 × 10−5 detection limit | Below 1 × 10−2 detection limit | 74 |
| PET | 100 | — | 3.9–17 | 1.8–7.7 | 75 |
| PEN | 100 | — | 7.3 | 3 | 75 |
| PI | 100 | — | 0.4–21 | 0.04–17 | 75 |
| CMC crosslinked with CA | 50–100 | 33.8 | Less than 0.02 g mm m2 day−1 | — | 76 |
| CNF with montmorillonites (MMT) clay and polyethylenimine (PEI) | 35 ± 3 | 30 wt% ML-MMTs: 41.5 | 20.9 g mm m2 day−1 | — | 77 |
| O-MMTs-CNFs: 78.7 | |||||
Step 2: casting cellulose films
Simple casting is the most common approach for cellulose film production. This involves dissolving cellulose in a suitable solvent at specific concentrations, followed by intensive stirring until a homogeneous solution is obtained, which is then cast on a container and left to dry. Many variables can affect the resulting film's characteristics, such as the cellulose concentration in solution, solvent type, shape and surface of the container, and drying temperature and environment.In our previous work on CNF films, we demonstrated that films made with CNF suspensions with lower concentration led to higher transparency levels and lower surface roughness.78 This was attributed to the increase in cellulose agglomerates in high cellulose concentrations. Similarly, the solvent choice can also impact film formation. Changing the solvent from acetone to N-methyl-2-pyrrolidone (NMP) was shown to have an overall positive impact on composite CA and CNF film formation, specifically by increasing the films’ transparency and mechanical strength and lowering their surface roughness and water absorption.79 These improvements can be explained by CNF's increased dispersion in NMP compared to acetone.
The container used for casting has a big influence on film transparency. Cellulose films cast on hydrophobic surfaces are denser, more transparent, and have lower WVTR and higher tensile strengths than films cast on hydrophilic substrates.80 As the suspension on hydrophilic surfaces dries, the cellulose fibers sediment and form random fiber networks. On hydrophobic surfaces, the solvent experiences tension due to its interaction with the container, and capillary forces drive the fibers to align, ultimately resulting in high transparency.
The drying conditions have a notable impact on film formation too. In the case of CNF films, hot press drying has been found to increase mechanical strength, lower oxygen and water vapor transmission rates, and lower surface roughness, primarily due to the improved consolidation of the CNF layers.81 In the case of EC, drying at 6 °C produces better films than drying at room temperature, even with added plasticizers.82 In the case of CA, humidity greatly affects film transparency and uniformity.83 The slow and gradual evaporation of the solvent is believed to be one of the main parameters contributing to high-quality films with both EC and CA.
| Material | Surface roughness (Rq) [nm] | Ref. |
|---|---|---|
| Glass/ITO | 2.15 | 86 |
| Glass/FTO | 45.43 | 86 |
| Glass/polished FTO | 14.96 | 86 |
| FG/ITO | 4.3–30.3 | 87 |
| Titanium foil | 25.34 | 88 |
| Stainless steel foil (untreated and polished) | 57.4, 25.2 | 89 |
| PET/ITO | 1.8–4.4 | 90 |
| PEN/ITO | 4.66 | 91 |
| Hydroxypropyl methyl cellulose/AgNWs | 7.07 | 92 |
| Nanocellulose paper/PEDOT:PSS | 6.54 | 14 |
The surface roughness of cellulose films greatly depends on the casting surface.84 Cellulose chains assemble while the film dries and thus roughly adopt the container's surface roughness, so it is prudent to use the smoothest container possible. Some types of cellulose, such as CMC, EC, and CA, peel off easily from glass containers, whereas CNC and CNF films remain firmly attached to hydrophilic surfaces. For the latter, it is ideal to use plastic containers or to treat hydrophilic surfaces (like glass) with a hydrophobic coating. Many of these coatings are highly toxic siliconizing agents, so caution is advised.
A film's roughness may be very high even after using a smooth substrate. In such cases, films can either be coated with a planarization layer or hot pressed. Depositing SiNx on CNF films has been shown to reduce the surface roughness to 1 nm and provide the added benefit of greatly reducing the WVTR to less than 0.02 g m−2 day−1.85 Alternatively, CNF films can be hot pressed at 120 °C for 10 min at 1.1 MPa.81 Hot pressing is mainly useful for films with very high roughness, as it helps to reduce roughness on a macro level.
| Material | Thickness [µm] | WVTR [g (m2 day)−1] (23 °C and 50% RH) | OTR [cm3 (m2 day)−1] | Ref. | ||
|---|---|---|---|---|---|---|
| Without coating | With coating | Without coating | With coating | |||
| CA crosslinked with PMDA and coated with OCTS | 153–215 | 77.0 ± 2.5 | 29.5 ± 1.7 | 280 (23 °C and 80% RH) | 24.6 ± 0.8 | 93 |
| CNF coated with SiNx | 41.4 | 13.60 ± 0.42 (38 °C and 50% RH) | Below 0.02 detection limit | 10.83 ± 0.99 cm3 m−2 day−1 bar−1 (70% RH) | — | 85 |
| Paper coated with Al2O3 through atomic layer deposition | 60 | 589.9 ± 15.1 | 2.2 ± 0.5 | — | — | 94 |
| Cellophane coated with SiOx | 35 | 65.85 (23 °C and 85% RH) | 1.28 | 10.44 (23 °C and 0% RH) | 0.82 | 95 |
Castro-Hermosa et al. have reported that low gas barrier levels are expected for PSCs,73 outside the current range for modified cellulose films, but their discussion about gas barrier properties is focused on encapsulated devices and not solely on the substrate. For instance, thermoplastic starch (TPS) films reinforced with CNF have OTR levels below 2 × 10−4 cm3 m−2 day−1 and WVTR levels below 2.24 × 10−2 g (m2 day)−1 and still maintain an optical transmittance of over 80% at 550 nm.96 Films like these have relatively high surface roughness levels, and their resistance to the solvents used in PSCs is yet unclear. It could be possible to combine PSCs made on cellulose films with a TPS/CNF encapsulant, using transparent biobased materials for both the substrate and the encapsulant.
Step 3: depositing the bottom electrode
FTO and ITO, the most typical electrode materials for PSCs, face issues when combined with cellulose. The use of FTO is limited to glass substrates due to its high-temperature processing requirements, and while ITO can be sputtered at low temperatures, indium is a scarce element. More sustainable electrodes are being investigated. Among them, conductive polymers and metallic nanowires have been shown to be compatible with cellulose. A comparison of various electrode materials and substrates is presented in Table 9.| Conductive substrate | Thickness [µm] | Transmittance [%] | Sheet resistance [Ω sq.−1] | Ref. |
|---|---|---|---|---|
| FG (Corning Willow Glass)/ITO | 100 | ∼80 (at 550 nm) | 12.6 ± 0.1 | 111 |
| Titanium foil | — | 0 | ∼3.6 | 112 |
| Stainless steel foil | — | 0 | <0.5 | 113 |
| PET/ITO | 188 | 80–90 (at 550 nm) | 14.5–17.3 | 90 |
| PEN/ITO | 125 | ∼82 (at 550 nm) | 15 | |
| EC/AgNWs | 8 | 88 (400–1000 nm) | 21.04 | 31 |
| Cellophane/TiO2/Ag/TiO2 | — | 82.1 (400–800 nm) | 9.5 | 114 |
Poly(3,4-ethylenedioxythiophene) (PEDOT) is a well-investigated conductive polymer whose sheet resistance can reach around 20 Ω sq.−1,97 close to the usual values of TCO. However, a notable drawback is that thick layers of PEDOT are required to achieve low resistivity, which significantly reduces its transparency. PEDOT's conductivity can be greatly increased by doping and crosslinking the polymer. Polystyrene sulfonate (PSS) is a common dopant used to increase the conductivity of PEDOT and simultaneously decrease electrode thickness and increase transparency. With the addition of a conductivity and flexibility enhancer (CFE), PEDOT:PSS:CFE can reach conductivities of over 4100 S cm−1 and a transmittance of 85%, comparable to commercial ITO on PET. Perovskite solar modules consisting of this electrode were shown to reach 10.9% PCE on a 25 cm2 active area.98 PEDOT and PEDOT:PSS are compatible with cellulose, as PEDOT evaporates at low temperatures, and PEDOT:PSS can be spin coated or printed. Their inks are usually water-based, which means that water-resistant cellulose is required (see Table 3).
N-doped poly(benzodifurandione) (n-PBDF) is a novel polymer electrode made as a TCO alternative. Similar to PEDOT:PSS, it is solution-based and can be deposited at low temperatures via spin coating, inkjet printing, blade coating, and spraying. Sheet resistances 90.9 and 78.8 Ω sq.−1 can be obtained by spraying layers of 65–75 nm thickness. FAPbI3 PSCs were built on PET/n-PBDF substrates with 11.24% PCE.99
Metallic nanowires have gained interest as promising ITO substitutes due to their excellent features, which include low sheet resistance, high transparency, and easy deposition via spraying or spin coating. Their high transparency stems from the random deposition of nanowires and the resulting mesh-like structures, leaving holes through which light can travel. Silver nanowires (AgNWs) are the most commonly used nanowires thanks to their high conductivity of around 12 Ω sq.−1 and 94.4% transmittance,100 although more sustainable options such as copper101,102 and nickel103 are also being investigated. To attain high conductivities, electrical losses need to be reduced by welding nanowire junctions via thermal annealing,104 laser sintering,105 or chemical processes.106,107
Poor substrate bonding strength and high surface roughness are the major disadvantages of metallic nanowires. Addressing these typically requires nanowires to be embedded in a different material, either by pressing the material into the substrate107 or by mixing the two in a conductive material matrix to improve conductivity.108 One way to solve both of the aforementioned issues when working with cellulose is to spray the nanowires on a rigid surface and then cast the cellulose. This greatly reduces the surface roughness and keeps the nanowires from moving inside the cellulose film (Fig. 6).109 AgNWs were used as electrodes on cellulose to make OSCs, with an impressive 18.71% efficiency level,31 proving that cellulose is suitable for the development of high-efficiency devices.
 | ||
| Fig. 6 Pre-patterned AgNWs embedded in a hydroxypropyl methyl cellulose film. (a) Fabrication procedure, (b) photograph of the cellulose film embedded with nanowires, (c) SEM micrograph of film with embedded nanowires, (d) SEM micrograph of film with coated nanowires, reproduced from ref. 109 with permission from the American Chemical Society.109 Copyright 2020. | ||
The highest-performing PSC made on cellulose was made using an oxide/metal/oxide electrode (OMO), specifically TiO2/Ag/TiO2, and reached 13.19% PCE.36 Devices made with OMO electrodes were made using MAPbI3, which partly explains their low performance compared to lower bandgap perovskites. The OMO electrode was sequentially sputtered and reached a sheet resistance of 3.6 Ω sq.−1, well below the 30 Ω sq.−1 of commercial PET/ITO but with similar optical transmittance.110 OMO electrodes have the advantage of being easy to deposit, but due to the need for vacuum deposition, their scalability would depend on a high initial investment compared to wet methods.
When attaching a film to a carrier, there are many things to consider. First, the carrier and the adhesive surface should be as smooth as possible to prevent protrusions that could compromise the cellulose surface, which would, in turn, affect the deposition quality of the subsequent layers. Second, an adhesive with an adequate thermal resistance must be chosen based on the highest temperature expected during PSC processing. Third, if the device is to be characterized while it is on the carrier, the optics of the adhesive have to be considered, as these will impact how much light reaches the active area. Fourth, if bending tests are to be done, the device needs to be peeled off the carrier without being damaged.
Step 4: solvent washing
Conventional glass and plastic substrates are normally washed with ultrasonication baths in different solvents.45 In laboratory settings, the quality of the deposited layers is ensured by washing the substrate to eliminate all the loose particles on its surface and leftovers from the etching process, such as zinc remnants and the Kapton tape adhesive. Substrates are washed in a soap water bath, followed by an acetone bath and an isopropanol bath, and then dried with nitrogen gas. For applications that require high hydrophilicity and no traces of organic components, the substrates are subjected to a chemical bath in a piranha solution. This mixture of sulfuric acid and hydrogen peroxide decomposes most organic matter and is, therefore, unsuitable for synthetic polymers and cellulose films.For small-scale spin-coating processes, a compatible solvent is dynamically spin coated at high speed over a cellulose film to clean the surface. In large-scale R2R fabrication processes, the substrates are washed with an organic solvent, such as isopropanol or ethanol, followed by an air plasma corona treatment. Web cleaning may also be performed, which involves using rolls made of a sticky elastomer to remove dust particles from the substrate surface.115,116 Substrates can also undergo UV-O3 or plasma treatments, as discussed below (see section Step 5: wettability).
Cellulose is compatible only with some of these cleaning methods; ultrasonication in a solvent bath risks cellulose redispersal in the solvent. The recommended approach is to dynamically spin coat a compatible solvent (i.e., something that would not redissolve the film) quickly, spray the substrate surface with N2, and treat the film inside a plasma cleaner. Web cleaning and corona treatments are suitable methods for removing dust and cleaning the cellulose surface in large-scale processes.
Step 5: wettability
In many cases, the surface that needs to be coated has poor wettability, which prevents the ink from completely covering it. To address this issue, surfaces are typically subjected to UV-O3 or plasma treatment. These treatments have a dual purpose: to remove organic matter left on the surface after cleaning and to increase the surface wettability. Surfactants can also be used instead of surface treatments to increase the ink's spreadability over the substrate.UV exposure has been shown to increase cellulose swelling and reduce the material's mechanical properties.118 These effects occur because bond scissions between glucose units in the cellulose chains create more sites to form hydrogen bonds with water, as evidenced by the reduced cellulose chain length.119 UV light can penetrate the cellulose bulk and greatly increase the material's hydrophilicity, in addition to affecting the surface. This could prove disadvantageous when using cellulose substrates, as inks would get absorbed into the substrate and prevent layers from forming on top.
Another common technique for surface cleaning involves the use of plasma. An electric current is passed through a gas to ionize it, and these ions bombard the substrate surface, resulting in etching or an increase in bonding attributes. When the plasma exposure energy is low, this effect is confined to the substrate surface and affects only the top molecular layers, not the bulk, reaching a depth of around 4–6 Å.120 Gas ionization is controlled by applying an energy field at radio frequencies around 13.56 MHz, as these have been found to produce homogeneous results.
Since cellulose is often a porous material, an increase in its hydrophilicity can render cellulose films absorbent. This is undesirable, as the material would then absorb the solutions used to make layers on top of it. Similar to UV-O3 treatment, plasma can break the molecular oxygen bonds to form free oxygen radicals. However, plasma only affects the substrate surface and is thus more suitable than UV-O3.
A useful alternative to surface treatments is to include a surfactant to reduce surface tension and facilitate the deposition of inks in a controlled manner. When PEDOT:PSS is used as an electrode, Tween-80 can be used to improve its spread over glass, without any surface treatment. This approach has the benefit of decreasing PEDOT:PSS sheet resistance to 202 Ω sq.−1 (ref. 121). When used as an HTL, PEDOT:PSS has a tendency to corrode the perovskite layer, which greatly decreases the perovskite stability. Triton-X 100 (TX) can be used to increase the spreadability of PEDOT:PSS and insulate the perovskite, which would, in turn, lead to reduced charge interface recombination. This method has been shown to increase PCE from 11.12% to 16.23%.122
TX can also be used to greatly reduce the surface tension of SnO2 nanoparticle (NP) inks intended for R2R processes. Excessive TX acts as an impurity and decreases device performance, so isopropanol (IPA) is mixed with water to further decrease the surface tension.123 Alternatively, SnO2 can be mixed with polyethylene oxide–polypropylene oxide–polyethylene oxide (P123) to increase the ETL spread, compactness, and uniformness. This dopant also increases device stability, thanks to the increased binding interaction between SnO2 and the perovskite layer, and 85% of the initial performance is retained after 1000 h of storage in air.124
Step 6: depositing the first CTL
Depending on the chosen device architecture, namely NIP or PIN, an n-type or a p-type semiconductor material is deposited on top of the bottom electrode. TiO2 is the most commonly used ETL—a legacy from dye-sensitized solar cells when perovskite was discovered as a photoactive material.125 Mesoporous titania (m-TiO2) acts as a scaffold for the perovskite; both materials remain in close contact, enabling charge separation and electron transport to the bottom electrode. A thin layer of compact titania (c-TiO2) is deposited before m-TiO2 to better match the band levels between the perovskite, the ETL, and the bottom electrode. Both layers of TiO2 need to be sintered at temperatures over 400 °C, outside the temperature range of synthetic polymers and cellulose.45 Tin oxide (SnO2) is an alternative to TiO2 that can be annealed at very low temperatures; it is commonly sold as nanoparticles suspended in water126 or can be made from tin chloride in ethanol.127,128 ZnO may also be used as an ETL in PSCs, but it has been shown to degrade the perovskite quickly. Therefore, SnO2 is the only viable low-temperature ceramic ETL so far. SnO2 is typically annealed at temperatures between 150 °C and 180 °C,129 although there are methods to anneal it at room temperature.128,130 This is the highest temperature used for an annealing process during PSC fabrication, as the perovskite layer is normally annealed at 100 °C.45 It is also possible to top c-TiO2 with SnO2 for better band alignment and greater efficiency.131Inverted structure devices have a wide range of options for their first CTL. Nickel oxide (NiOx) is the most popular p-type HTL that is deposited on top of the bottom electrode. It can be deposited using various techniques, including spraying and sputtering, atomic layer deposition, and chemical baths. NiOx has different stoichiometries depending on the deposition method, which affects its electrical properties and band alignment with the perovskite. Spin-coated NiOx nanoparticles without annealing were used to make PSCs with 21.6% efficiency.132
Other than metal oxides, polymers have successfully been integrated into PSCs. PEDOT:PSS was the first polymer to be used as an HTL in inverted structure PSCs, and it can reach efficiencies of 19.19%.133 Nonetheless, PEDOT:PSS significantly affects perovskite performance and stability due to its high hygroscopicity and acidity.132 PSS corrodes the perovskite and provokes ion migrations to the ITO electrode. Substituting this dopant with hydrophobic poly(ethylene-co-vinyl acetate) (EVA) improves perovskite adhesion to the electrode and prevents ion migration, resulting in devices with efficiencies close to 20%.
Poly[bis(4-phenyl)(2,4,6-trimethylphenyl)amine] (PTAA) is another polymer used to make HTLs in inverted structure PSCs. It has the advantage of forming very smooth hydrophobic layers after annealing and is more stable than PEDOT:PSS.132 However, these properties also lead to the perovskite solution dewetting too rapidly, resulting in nonuniform layers. This problem can be solved by using UV-O3 or plasma treatment, but as discussed earlier, these treatments can damage organic layers. Researchers have opted to add surfactants to perovskite solutions to increase their spreadability. Dodecylammonium bromide (DDABr) proved to be a useful additive, resulting in PSCs with an impressive 24.3% efficiency, and up to 19.8% efficiency in 13.18 cm2 aperture area devices.134 PEDOT and PTAA can be deposited in a variety of ways; it is most apt to introduce these polymers in inks meant for printing or spraying.
The use of self-assembled monolayers (SAMs) has surged recently and has led to a variety of record-breaking PSCs, reaching efficiencies over 26% when combined with NiOx.135 Unlike PEDOT and PTAA, SAMs are small-sized molecules and are usually spin coated or sprayed onto the electrode or other CTLs. These molecules consist of three parts: an anchoring group that bonds the SAM to the substrate surface, a spacer group that determines the SAM tilt angle and connects the other two groups, and a terminal group that modifies the perovskite work function and inhibits interfacial charge recombination.136 Each group can be adjusted based on the substrate surface and the influence on perovskite crystal growth, making them highly versatile. Their anchoring is driven by heat and is thus greatly affected by the substrate morphology; thus, the deposition technique needs to be optimized for different surfaces.136
Special care is required during this stage, as the charge transport material solvent can have a significant impact on the substrate. Thermal evaporation and sputtering have little effect on the substrate, but these processes are more expensive to implement in R2R processes. Printing, spraying, and slot-die coating all involve the use of solutions and inks, making the choice of material and solvent a critical issue at this stage (see Table 3).
Substituting water with ethanol. Commercial colloidal SnO2, the most common ETL requiring low-temperature processing, is usually sold in water suspensions. This has a negative impact on water-soluble cellulose films (namely CMC and CNC; Table 3). One way to avoid water on this layer is to use SnO2 on ethanol instead. SnO2 can be synthesized from SnCl2·2H2O in ethanol. Specifically, SnCl2·2H2O reacts with ethanol to form Sn(OH)2, which then decays into SnO and finally oxidizes to SnO2.128
PEDOT:PSS is another popular PSC material that is sold in water. Water can be removed from the solution and replaced with ethanol through ultrafiltration and dialysis.137 This approach has been used to make top electrodes for OSCs, reaching 14% efficiency.138
Step 7: first CTL thermal annealing
Many layers require thermal annealing in order to grow crystalline structures. TiO2 is a very common ETL material, but its use is restricted to glass substrates because it requires annealing at 500 °C. New TiO2 processing techniques have been developed to overcome this obstacle. Sputtering is one deposition technique that can be used at room temperatures and is scalable to high-volume fabrication. However, sputtered films normally require a thermal annealing process afterwards. To improve crystallinity and avoid high-temperature defects, sputtered TiO2 films can be annealed using UV light. This method has produced PSCs with a remarkable PCE of 24.75% as well as perovskite modules on glass/FTO and PEN/ITO with PCEs of 18.82% and 14.61%, respectively.139 It is also possible to deposit TiO2 layers at room temperature using wet chemistry methods. For instance, a sol–gel titanium diisopropoxide bis(acetylacetonate) solution can be annealed with a laser at room temperature; this method has resulted in devices with 17.1% PCE on PEN/ITO substrates.140SnO2 is used instead of TiO2 for the low-temperature processing of PSCs, yet it still requires temperatures over 150 °C to be annealed. Multiple approaches have been developed to overcome this. SnO2 layers can be made using intense pulse light,141 ultrasonic vibrations,130 negative pressure,142 UV light,143 or amine fumes (Fig. 7),128 and none of these methods require thermal annealing.
 | ||
| Fig. 7 Room temperature annealing of SnO2 with amine fumes, adapted from ref. 128 with permission from Elsevier.128 Copyright 2022. | ||
Nickel oxide is the most-used metal oxide HTL in PIN-structure PSCs. To overcome the need for high-temperature annealing, NiOx NPs made with a tetramethylammonium hydroxide surfactant can be used. These require annealing at 150 °C, which is a more tolerable temperature than the normal annealing temperature of 400 °C. This fabrication method has resulted in PSCs with 18.85% PCE on glass/FTO.144 Recently, a new HTL material known as DFBT-PMTP has been synthesized as an annealing-free and additive-free option for inverted PSCs; it has been shown to achieve 21.23% PCE.145 SAMs such as MeO-2PACz can also be deposited as HTLs and annealed at temperatures below 100 °C to obtain high efficiencies without any thermal treatment.146
Step 8: perovskite deposition
Perovskite crystal formation is susceptible to temperature and solvent vapor changes in the environment. While there are ways to deposit the perovskite layer by evaporation, it is typically deposited with solution-based methods, as these are cheaper and easier to implement.147,148 Perovskites have an ABX3 structure, and different A cations and X anions are mixed to shift the perovskite band level and adjust the perovskite Goldschmidt tolerance factor. The A cations are usually small molecules such as methylammonium (MA) or formamidinium (FA) or big atoms such as cesium or rubidium. Iodine is the most common anion, followed by bromine and chloride.149 The wet-method processing route consists of two different approaches for fabricating the perovskite layer—the one-step solution process (OSSP) and the two-step solution process (TSSP); both are illustrated in Fig. 8. | ||
| Fig. 8 Comparison of OSSP and TSSP, reproduced from ref. 150 with permission from Elsevier.150 Copyright 2020. | ||
The OSSP involves mixing the perovskite precursors together in a single solution. When the precursors are deposited, the solvent needs to be removed to start the nucleation and crystallization of the perovskite layer. In spin coating, this is normally done by using an antisolvent, which removes the solvent from the substrate without redissolving the perovskite layer. In large-scale methods, the perovskite solvent choice is very important, as the solvent is removed using an N2 air knife via the gas quenching method, and this is affected by the solvent evaporation temperature and the atmosphere solvent saturation.151 Methods such as flash infrared annealing and photonic curing are also R2R compatible.152,153
In the TSSP, both precursors are deposited separately. The BX3 precursors are typically deposited first. The AX precursor is dissolved in a different solvent that would remove the BX3 solvent, thus acting as an antisolvent. In both cases, the substrates are thermally annealed to remove any solvent traces and to promote grain growth.
The OSSP and TSSP each have their advantages and disadvantages. The OSSP is more straightforward since mixing all the precursors allows for an easier deposition, and it has a defined stoichiometry. However, the choice of antisolvent and how it is deposited greatly affects perovskite formation,154 and the OSSP tends to result in big crystals with many pinholes, without homogeneous surface coverage.150,155 Lead halide deposition in the TSSP is easier to control and more homogeneous, but there is little control over the resulting perovskite stoichiometry.155 There are arguments in favor of both methods for high-volume production: the OSSP allows for the precise control of precursor compositions, enables the use of complicated perovskite formulations in high-efficiency devices, and has a more economic implementation.156 TSSP allows for more consistent film morphology, leading to reproducible devices.157 Regardless of which process is used, more than one solvent is required, often with distinct properties, and their interactions with the cellulose substrate need to be carefully considered.
Substituting DMF
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) DMSO with ethanol and acetonitrile.
Ethanol and acetonitrile are incapable of dissolving lead iodide (PbI2) on their own and are commonly used as cosolvents. Ethanol can be mixed with N,N-dimethylacetamide (DMA) and alkylammonium chloride (RNH3Cl) to form FAPbI3;159 it is worth noting that DMA is a toxic solvent, but it is used in small amounts. In a similar fashion, acetonitrile can be mixed with 1,2-dimethoxyethane (DME) to make FAPbI3160 or with isopropyl acetate (IA) to make MAPbI3.161
DMSO with ethanol and acetonitrile.
Ethanol and acetonitrile are incapable of dissolving lead iodide (PbI2) on their own and are commonly used as cosolvents. Ethanol can be mixed with N,N-dimethylacetamide (DMA) and alkylammonium chloride (RNH3Cl) to form FAPbI3;159 it is worth noting that DMA is a toxic solvent, but it is used in small amounts. In a similar fashion, acetonitrile can be mixed with 1,2-dimethoxyethane (DME) to make FAPbI3160 or with isopropyl acetate (IA) to make MAPbI3.161
The use of a mixture of ethanol and acetonitrile was a significant advancement in PSC fabrication in recent years. In their work, Rezaee et al. fabricated MAPbI3 PSCs on glass/ITO and PET/ITO via bar coating and slot-die coating, respectively, at room temperature and attained efficiencies of 19.5%.162 A similar solvent mixture, DMSO with acetonitrile and ethanol, was used in the production of a wide-bandgap perovskite on tandem modules. The PSCs reached efficiencies up to 21.5%, and 23.8% in 20 cm2 tandem devices.163
Substituting DMF
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) DMSO with water and IPA.
Water is one of the main factors contributing to PSC degradation due to the critical role played by moisture in perovskite decomposition. This has led to researchers regarding water as unsuitable for PSC manufacturing. However, water and other green options can replace DMF and DMSO, the most commonly used solvents in lead-based PSC production. In the TSSP, BX2 precursors are deposited first, followed by AX. PbI2 has poor solubility in water, so it is replaced by Pb(NO3)2. Once dry, the substrate is submerged in a methylammonium iodide bath in IPA and thermally annealed to form the perovskite layer (Fig. 9). The crystallization pathway of perovskites formed from aqueous precursors differs from that of conventional organic-solvent systems, often producing films with larger grains.164–166 Interestingly, studies showed water-processed PSCs exhibit enhanced stability compared with DMF-based devices due to the absence of excess PbI2, the presence of larger crystalline domains, and Pb(NO3)2-derived residues that can act as water scavengers. Despite these advantages, some challenges still remain for aqueous precursor processing, including incomplete surface coverage, pinhole formation driven by Ostwald ripening, and the need for multi-cycle deposition to achieve uniform films. Surfactants such as potassium oleate have been introduced to lower the surface tension of water and promote better wetting. Using this strategy, PSCs have achieved 24% PCE, with unencapsulated devices retaining 92.5% of their initial performance after 90 days at 25 °C and 30% RH in ambient air. Under combined thermal and light stress (1 sun, 60 °C), these devices maintain 90.8% of their original PCE after 800 h, illustrating that properly controlled aqueous processing can deliver both high efficiency and long-term operational stability.
DMSO with water and IPA.
Water is one of the main factors contributing to PSC degradation due to the critical role played by moisture in perovskite decomposition. This has led to researchers regarding water as unsuitable for PSC manufacturing. However, water and other green options can replace DMF and DMSO, the most commonly used solvents in lead-based PSC production. In the TSSP, BX2 precursors are deposited first, followed by AX. PbI2 has poor solubility in water, so it is replaced by Pb(NO3)2. Once dry, the substrate is submerged in a methylammonium iodide bath in IPA and thermally annealed to form the perovskite layer (Fig. 9). The crystallization pathway of perovskites formed from aqueous precursors differs from that of conventional organic-solvent systems, often producing films with larger grains.164–166 Interestingly, studies showed water-processed PSCs exhibit enhanced stability compared with DMF-based devices due to the absence of excess PbI2, the presence of larger crystalline domains, and Pb(NO3)2-derived residues that can act as water scavengers. Despite these advantages, some challenges still remain for aqueous precursor processing, including incomplete surface coverage, pinhole formation driven by Ostwald ripening, and the need for multi-cycle deposition to achieve uniform films. Surfactants such as potassium oleate have been introduced to lower the surface tension of water and promote better wetting. Using this strategy, PSCs have achieved 24% PCE, with unencapsulated devices retaining 92.5% of their initial performance after 90 days at 25 °C and 30% RH in ambient air. Under combined thermal and light stress (1 sun, 60 °C), these devices maintain 90.8% of their original PCE after 800 h, illustrating that properly controlled aqueous processing can deliver both high efficiency and long-term operational stability.
 | ||
| Fig. 9 PSC fabrication using water as a solvent, reproduced from ref. 167 with permission from the American Chemical Society.167 Copyright 2024. | ||
Moreover, while improved morphology, fewer defects, and controlled crystallization pathways explain much of the enhanced stability, broader degradation pathways, such as ion migration remain considerations.
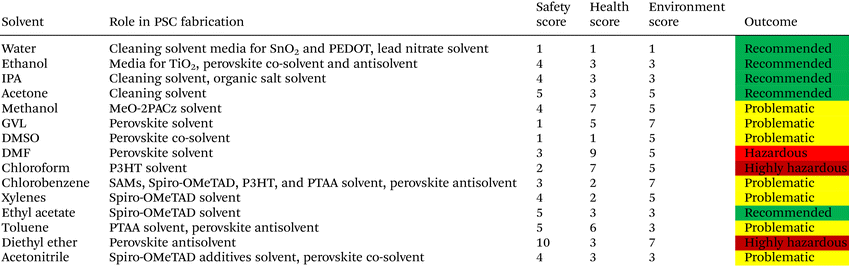
Using less toxic and more environmentally responsible solvent systems is essential for enabling safe and sustainable manufacturing. Green solvents, defined as low-toxicity, biodegradable, non-flammable with minimal environmental persistence, offer safer alternatives. Simple alcohols such as ethanol and IPA, water, and bio-derived solvents (e.g., GVL) exhibit lower toxicity, biodegradability, and minimal bioaccumulation potential.170 In addition, bio-derived solvents produced from renewable feedstocks offer further reductions in life-cycle impacts and align well with circular-economy principles. Taken together, both toxicity data and life-cycle assessment findings underscore that replacing hazardous polar aprotic solvents with greener alternatives can significantly reduce the environmental footprint associated with perovskite film fabrication. Table 10 presents the list of solvents used in the fabrication of PSCs classified based on Safety (S), Health (H), and Environment (E).
Step 9: thermal annealing
After the solvent is removed, the perovskite is annealed to promote crystal growth and remove any leftover solvent. This process is usually done at 100 °C for 40–60 minutes. As discussed earlier, high temperatures can cause cellulose to lose moisture and buckle, and additives like plasticizers to degrade, so it is prudent to minimize thermal treatments. The following methods can be used to anneal the perovskite layer at room temperature.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 ratio of 2-methoxy ethanol and tetrahydrofuran (THF) and used it to make MAPbI3 perovskite devices with up to 18% efficiency, without using an antisolvent or thermal annealing. The key finding was that THF helped accelerate the solvent removal by lowering the vapor pressure of the solvent mixture, facilitating the crystallization of the perovskite with just a nitrogen gas flow.146 Caution is advised as both of these solvents are toxic. A similar approach could be found using non-toxic and greener solvents instead.
9 ratio of 2-methoxy ethanol and tetrahydrofuran (THF) and used it to make MAPbI3 perovskite devices with up to 18% efficiency, without using an antisolvent or thermal annealing. The key finding was that THF helped accelerate the solvent removal by lowering the vapor pressure of the solvent mixture, facilitating the crystallization of the perovskite with just a nitrogen gas flow.146 Caution is advised as both of these solvents are toxic. A similar approach could be found using non-toxic and greener solvents instead.
Another way to avoid the thermal annealing of perovskite is by doping the antisolvent with 3,4-ethoxylenedioxythiophene (EDOT). The dopant fills any halide vacancies and thus helps passivate any defects in the perovskite layer, and it increases cell stability by reducing halide ion migration. This technique also slightly increases the open-circuit voltage and device stability in ambient air. Cells made with this approach have attained 20.12% PCE on glass/FTO and 15.58% on unspecified flexible substrates.177
Step 10: depositing the second CTL
With solution-based processes, special care needs to be taken to use a compatible solvent during the second CTL deposition to avoid affecting the perovskite active layer. Additionally, since the perovskite has already been deposited by this stage, the rest of the process needs to proceed at a low temperature.In NIP devices, 2,2′,7,7′-tetrakis(N,N-di-p-methoxyphenyl-amine)9,9′-spirobifluorene (Spiro-OMeTAD) is the most popular material for HTLs. Initially used as an HTL in light-emitting diodes and dye-sensitized solar cells, Spiro-OMeTAD was adapted to PSCs due to its easy processability, good morphology, and ability to match the bands of perovskite types.178 Its conductivity is much lower than that of most perovskites, so Spiro-OMeTAD needs to be doped; the most common dopants are 4-tert-butyl pyridine (tBP), LiTFSI, and FK209.179 The biggest drawback of Spiro-OMeTAD is its low stability, arising from the dopants’ interactions and degradations when in contact with perovskite, water, and oxygen.178 Promising new HTL materials similar to Spiro-OMeTAD have been recently developed and have proved to be easier to synthesize and more stable.180,181
PTAA has also been used as an HTL in devices with the NIP structure. It requires doping with tBP and LiTFSI to become a p-type material, but the dopant concentration needed is four times less than that of Spiro-OMeTAD.182 PTAA has many advantages: it is an amorphous, thermally stable material that is soluble in a wide range of organic solvents and forms very smooth films.182,183 There is no clear winner between PTAA and Spiro-OMeTAD, as they both have advantages and disadvantages. PTAA is more mechanically and thermally stable, but Spiro-OMeTAD has been the preferred HTL material in record-breaking PSCs.182
Poly(3-hexylthiophene) (P3HT) is the third most commonly used organic material for the HTL in NIP PSCs. It exhibits good hole mobility without dopants and is inexpensive and simple to process. It is speculated to be a good replacement for Spiro-OMeTAD, as its low cost makes it a very attractive option for large-scale production.184 P3HT devices have been reported to attain an impressive efficiency level of 24.6%.185 The molecular structures of the different organic HTLs are illustrated in Fig. 13.
 | ||
| Fig. 13 Molecular structures of organic HTLs, adapted from ref. 186 with permission from Wiley.186 Copyright 2021. | ||
The deposition of metal oxide transport layers on the perovskite layer is uncommon because of the high temperatures necessary for deposition and annealing. One way to get around this issue is by synthesizing metal oxide NPs. NiOx NPs printed in NIP devices and SnO2 NPs in PIN devices have been reported to achieve 25.17% and 16.5% efficiency, respectively.187,188
For PIN structure devices, fullerene and its derivatives (C60 and PCBM) are the most popular ETL choices, thanks to their low-temperature processing, high electron conductivity, and appropriate band alignment with perovskites. However, good quality C60 layers require thermal evaporation, whereas PCBM is processable in solution but dimerizes under light and absorbs the perovskite degradation products, thus losing stability.189
Substituting chlorobenzene with ethyl acetate. There is an urgent need to replace chlorobenzene (CB), a common antisolvent for lead-based PSCs and a common solvent for Spiro-OMeTAD, with an alternative, as it is highly toxic to animals and the environment, and large volumes are used during fabrication.190 CB can be replaced with ethyl acetate (EA), a safer, greener alternative. Notably, devices made with Spiro-MeOTAD and EA have outperformed devices made with CB, at PCEs of 23.3% and 21.0%, respectively.191 In a study that compared many solvents for Spiro-OMeTAD, it was proved that CB can be substituted with p-xylene, a less toxic and more industrially compatible solvent, and achieve an identical PCE.192
Step 11: top electrode
The last step in PSC fabrication is the deposition of the top electrode. Thermally evaporated metals such as gold, silver, aluminum, and copper are the most typical materials for this layer. Their main advantages are their high conductivity, ease of processability, and proper work function. Their biggest drawback is metal ion diffusion into the other layers, which lowers device stability.193AgNWs can be used as top electrodes, and when combined with a TCO as a bottom electrode, they enable the easy fabrication of bifacial PSCs.194 When an opaque substrate is used, such as titanium foil, the top electrode needs to be transparent to let light into the active layer.
Carbon is among the newest electrode materials proposed for PSCs. Carbon electrodes are normally used in mesoporous recyclable devices but have been adapted to work as top electrodes in planar devices. A composite carbon electrode can be hot pressed on top of Spiro-OMeTAD or P3HT (Fig. 14). Devices made with this novel technique have achieved efficiency levels of over 19%.195 A similar approach has been taken in an R2R process involving a silver-carbon composite, which proves the scalability of this material.196
 | ||
| Fig. 14 Cross-section of hot-pressed composite carbon electrode over a PSC: (a) optical microscope image, (b) SEM image, reproduced from ref. 195 with permission from Springer Nature.195 Copyright 2025. | ||
Outlook
Actionable points
Summary
Over the last decade, PSCs have been considered promising options for circumventing the costly and energy-intensive fabrication of silicon. PSCs have undergone huge leaps in efficiency and stability, and now, before they enter the market, the question of their sustainability is taking center stage. Science and industry are increasingly aiming to integrate PSCs into a circular economy model, and cellulose might be one of the key factors. However, combining cellulose and PSCs comes with many intricacies that have rarely been explored; it is not as easy as merely swapping one material for another. A successful outcome requires both the cellulose and the PSC fields to come together and understand the other's needs and limitations, including acceptable gas barrier levels, appropriate transparency and conductivity values, and the solvent resistance of cellulose films, among others.Cellulose has the potential to be a sustainable substrate but only after careful modifications and optimization. What would PSC fabrication on cellulose look like? First, cellulose needs to be crosslinked to increase its solvent resistance and reduce its CTE and water uptake. Next, plasticizers and other compounds have to be included to increase the flexibility of cellulose films, and coatings are needed to reduce gas permeation. Cellulose needs to be dried properly during casting for maximum transparency and minimum surface roughness. The most critical point arises during the deposition of the bottom electrode and the first CTL, as these have the biggest impacts on the deposition of the next layers and the overall quality of the device. Solvent compatibility and an appropriate surface morphology are crucial parameters at this stage. Finally, cleaning treatments, ink deposition, and annealing treatments need to be adapted to PSC processing to avoid damaging the substrate.
The traditional PSC processing route has to evolve to accommodate more sustainable approaches. Here, we offer some realistic solutions for most of the steps involved, which can be implemented with varying levels of engineering and optimization. The most challenging aspect is ensuring that the gas barrier properties of cellulose, particularly the oxygen barrier, surpass those of PET and PEN; OTR and WVTR levels need to improve to ensure that solar cells are both sustainable and long lasting. Otherwise, this technology would only be suitable for applications with short lifespans, such as some sensors and indoor photovoltaics. Gas barriers can be improved by pairing cellulose-based PSCs with biobased encapsulants, which would ensure the recyclability of the devices. Environmental, social, and economic sustainability and the high-throughput production of PSCs is within reach, and the methods have been laid out in this article. We hope that the outlined recommendations help perovskite and cellulose experts come together in bringing forth the next generation of solar technology.
Author contributions
J. V. G.: conceptualization, investigation, visualization, methodology, writing – original draft, writing – review and editing, project administration; M. H.: writing – original draft, writing – review and editing; V. A.: investigation, writing – original draft; S. Y.: investigation; J. K.: writing – review and editing; R. S.: writing – review and editing; M. V.: writing – review and editing; K. M.: conceptualization, supervision, funding acquisition, writing – review and editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article has been included as part of the supplementary information (SI). Supplementary information: Solvent resistance test film preparation and Table S1. Supplementary information is available. See DOI: https://doi.org/10.1039/d5gc05281e.Acknowledgements
J. V. G. thanks the Research Council of Finland, project ECOSOL (347275). M. H. thanks PROFI7/SUSMAT funding provided by the Research Council of Finland. V. A., S. Y. and K. M. thank Circular Materials Bioeconomy Network funded by the Ministry of Education and Culture, Finland (CIMANET, Decision No. VN/3137/2024-OKM-6). J. K. acknowledges that she is part of the Sector Plan Engineering II, which is funded by the Dutch Ministry of Education, Culture, and Science (OCW). M. V. and R. S. acknowledge the support provided by the Research Council of Finland (RCF), Printed intelligence infrastructure funding, decision 358621 and the RCF Flagship Programme, Photonics Research and Innovation (PREIN), decision number 346545. The authors thank Väinö Anttalainen/University of Turku for photographing our cellulose solvent resistance experiment samples.References
- C. Yang, W. Hu, J. Liu, C. Han, Q. Gao, A. Mei, Y. Zhou, F. Guo and H. Han, Light: Sci. Appl., 2024, 13, 227 CrossRef PubMed.
- R. G. Charles, A. Doolin, R. García-Rodríguez, K. V. Villalobos and M. L. Davies, Energy Environ. Sci., 2023, 16, 3711–3733 Search PubMed.
- E. S. Akulenko, M. Hadadian, A. Santasalo-Aarnio and K. Miettunen, Heliyon, 2023, 9, e13584 Search PubMed.
- S. Kim, H. Van Quy and C. W. Bark, Mater. Today Energy, 2021, 19, 100583 Search PubMed.
- P. Subudhi and D. Punetha, Prog. Photovoltaics, 2023, 31, 753–789 Search PubMed.
- Y. Ma, Z. Lu, X. Su, G. Zou and Q. Zhao, Adv. Energy Sustainability Res., 2022, 4 DOI:10.1002/aesr.202200133.
- M. Tao, V. Fthenakis, B. Ebin, B. Steenari, E. Butler, P. Sinha, R. Corkish, K. Wambach and E. S. Simon, Prog. Photovoltaics, 2020, 28, 1077–1088 Search PubMed.
- D. Le Khac, S. Chowdhury, A. Soheil Najm, M. Luengchavanon, A. Mebdir Holi, M. Shah Jamal, C. Hua Chia, K. Techato and V. Selvanathan, Sol. Energy, 2024, 267, 112214 Search PubMed.
- G. S. Han, S. Lee, M. L. Duff, F. Qin and J.-K. Lee, ACS Appl. Mater. Interfaces, 2018, 10, 4697–4704 Search PubMed.
- B. T. Feleki, R. K. M. Bouwer, M. M. Wienk and R. A. J. Janssen, Sol. RRL, 2022, 6, 2100898 CrossRef.
- J. Liu, T. Ye, D. Yu, S. (Frank) Liu and D. Yang, Angew. Chem., Int. Ed., 2023, 62 DOI:10.1002/anie.202307225.
- L. Huang, Y. Li, Z. Zheng, Y. Bai, T. P. Russell and C. He, Mater. Horiz., 2024, 11, 1560–1566 RSC.
- B. Liu, J. Zeng, P. Li, M. Kui, J. Li and K. Chen, Chem. Eng. J., 2024, 498, 155273 CrossRef.
- L. Gao, L. Chao, M. Hou, J. Liang, Y. Chen, H.-D. Yu and W. Huang, npj Flexible Electron., 2019, 3, 4 CrossRef.
- G. Dinesh and B. Kandasubramanian, Mater. Chem. Phys., 2022, 281, 125707 CrossRef.
- H. Qi, Novel Functional Materials Based on Cellulose, Springer International Publishing, 2017 Search PubMed.
- B. Lindman, G. Karlström and L. Stigsson, J. Mol. Liq., 2010, 156, 76–81 CrossRef.
- H. Qi, Novel Functional Materials Based on Cellulose, Springer International Publishing, Cham, 2017 Search PubMed.
- N. B. Erdal and M. Hakkarainen, Biomacromolecules, 2022, 23, 2713–2729 CrossRef PubMed.
- S. Jing, L. Wu, A. P. Siciliano, C. Chen, T. Li and L. Hu, ACS Nano, 2023, 17, 22196–22226 CrossRef PubMed.
- O. A. El Seoud, M. Kostag, K. Jedvert and N. I. Malek, Macromol. Mater. Eng., 2020, 305 DOI:10.1002/mame.201900832.
- A. Forte, F. Dourado, A. Mota, B. Neto, M. Gama and E. C. Ferreira, Int. J. Life Cycle Assess., 2021, 26, 864–878 CrossRef.
- A. Mazega, A. F. Lehrhofer, R. J. Aguado, A. Potthast, R. Marquez, T. Rosenau and M. Delgado-Aguilar, Cellulose, 2025, 32, 5227–5246 CrossRef.
- A. Babaei-Ghazvini, B. Vafakish, R. Patel, K. J. Falua, M. J. Dunlop and B. Acharya, Int. J. Biol. Macromol., 2024, 258, 128834 CrossRef PubMed.
- T. Heinze, O. A. El Seoud and A. Koschella, in Cellulose Derivatives, Springer Cham, 1st edn, 2018, pp. 259–292 Search PubMed.
- T. Nishino, I. Matsuda and K. Hirao, Macromolecules, 2004, 37, 7683–7687 CrossRef.
- J. A. Diaz, X. Wu, A. Martini, J. P. Youngblood and R. J. Moon, Biomacromolecules, 2013, 14, 2900–2908 CrossRef PubMed.
- T. Zhang, P. Yang, M. Chen, K. Yang, Y. Cao, X. Li, M. Tang, W. Chen and X. Zhou, ACS Appl. Mater. Interfaces, 2019, 11, 36010–36019 CrossRef PubMed.
- W. A. MacDonald, J. Mater. Chem., 2004, 14, 4 RSC.
- B. Xiong, W. Zhong, Q. Zhu, K. Liu, M. Li, G. Sun and D. Wang, Nanoscale, 2017, 9, 19216–19226 RSC.
- Z. Xiao, J. Liu, X. Chen, Z. Suo, X. Cao, N. Xu, Z. Yao, C. Li, X. Wan and Y. Chen, J. Mater. Chem. A, 2025, 13, 2301–2308 RSC.
- Š. Jech, N. Bartie, G. Tas, K. Miettunen, R. Serna-Guerrero and A. Santasalo-Aarnio, Sol. Energy Mater. Sol. Cells, 2025, 286, 113561 CrossRef.
- V. Larini, C. Ding, F. Faini, G. Pica, G. Bruni, L. Pancini, S. Cavalli, M. Manzi, M. Degani, R. Pallotta, M. De Bastiani, C. Ma and G. Grancini, Adv. Funct. Mater., 2023, 34 DOI:10.1002/adfm.202306040.
- G. Imbuluzqueta, F. J. Cano, U. Iglesias, J. Aizpurua, J. M. Hernández, N. Yurrita, W. Cambarau and O. Zubillaga, Sol. Energy, 2025, 300, 113799 CrossRef.
- G. Fredi and A. Dorigato, Adv. Ind. Eng. Polym. Res., 2021, 4, 159–177 Search PubMed.
- H. Li, X. Li, W. Wang, J. Huang, J. Li, Y. Lu, J. Chang, J. Fang and W. Song, Sol. RRL, 2019, 3 DOI:10.1002/solr.201800317.
- X. Dai, Y. Deng, C. H. Van Brackle, S. Chen, P. N. Rudd, X. Xiao, Y. Lin, B. Chen and J. Huang, Adv. Energy Mater., 2019, 10 DOI:10.1002/aenm.201903108.
- J. H. Heo, D. H. Shin, M. L. Lee, M. G. Kang and S. H. Im, ACS Appl. Mater. Interfaces, 2018, 10, 31413–31421 CrossRef PubMed.
- C. Zhou, Y. Xu, Y. Li, K. Du, X. Li, X. Dong, L. Li, N. Yuan and J. Ding, Sol. RRL, 2023, 8 DOI:10.1002/solr.202300901.
- N. Ren, L. Tan, M. Li, J. Zhou, Y. Ye, B. Jiao, L. Ding and C. Yi, iEnergy, 2024, 3, 39–45 Search PubMed.
- X. Chen, W. Cai, T. Niu, H. Wang, C. Liu, Z. Zhang, Y. Du, S. Wang, Y. Cao, P. Liu, W. Huang, C. Ma, B. Yang, S. (Frank) Liu and K. Zhao, Energy Environ. Sci., 2024, 17, 6256–6267 RSC.
- R. Xu, F. Pan, J. Chen, J. Li, Y. Yang, Y. Sun, X. Zhu, P. Li, X. Cao, J. Xi, J. Xu, F. Yuan, J. Dai, C. Zuo, L. Ding, H. Dong, A. K.-Y. Jen and Z. Wu, Adv. Mater., 2023, 36 DOI:10.1002/adma.202308039.
- A. Miglani, S. B. Ogale and O. S. Game, Small, 2025, 21 DOI:10.1002/smll.202411355.
- J. Valdez García, M. Hadadian, R. Nizamov, P. Mäkinen, N. Lamminen, P. Vivo and K. Miettunen, R. Soc. Open Sci., 2024, 11 DOI:10.1098/rsos.241012.
- M. Saliba, J.-P. Correa-Baena, C. M. Wolff, M. Stolterfoht, N. Phung, S. Albrecht, D. Neher and A. Abate, Chem. Mater., 2018, 30, 4193–4201 CrossRef.
- J. J. Benitez, P. Florido-Moreno, J. M. Porras-Vázquez, G. Tedeschi, A. Athanassiou, J. A. Heredia-Guerrero and S. Guzman-Puyol, Int. J. Biol. Macromol., 2024, 273, 132956 CrossRef PubMed.
- Q. Wu, Y. Meng, K. Concha, S. Wang, Y. Li, L. Ma and S. Fu, Ind. Crops Prod., 2013, 48, 28–35 CrossRef.
- Z. Wang, M. Zhu, J. Li, C. Hu, J. Li, R. Xiong and C. Huang, Nano Energy, 2024, 129, 109974 CrossRef.
- H. Yang, J. Edberg, V. Gueskine, M. Vagin, M. G. Say, J. Erlandsson, L. Wågberg, I. Engquist and M. Berggren, Carbohydr. Polym., 2022, 278, 118938 CrossRef PubMed.
- G. Hu, X. Lan, B. Peng, J. Liao and Y. Xiong, Int. J. Biol. Macromol., 2024, 260, 129476 CrossRef PubMed.
- Q. Yu, L. Yang, S. Wang, L. Zhang and D. Sun, Cellulose, 2023, 30, 10273–10284 CrossRef.
- W. Ge, J. Shuai, Y. Wang, Y. Zhou and X. Wang, Polym. Chem., 2022, 13, 359–372 RSC.
- M. Nogi and H. Yano, Appl. Phys. Lett., 2009, 94 DOI:10.1063/1.3154547.
- A. A. Goje, N. A. Ludin, P. N. A. Fahsyar, U. Syafiq, P. Chelvanathan, A. D. A.-G. Syakirin, M. A. Teridi, M. A. Ibrahim, M. S. Su'ait, S. Sepeai and A. S. H. M. Yasir, Mater. Renew. Sustain. Energy, 2024, 13, 155–179 CrossRef.
- B. Guo, W. Chen and L. Yan, ACS Sustainable Chem. Eng., 2013, 1, 1474–1479 CrossRef.
- T. H. Yim, S. H. Koo and H. Lee, Curr. Appl. Phys., 2010, 10, S241–S243 CrossRef.
- L. Chen, H. Yu, M. Dirican, D. Fang, Y. Tian, C. Yan, J. Xie, D. Jia, H. Liu, J. Wang, F. Tang, X. Zhang and J. Tao, Adv. Mater. Interfaces, 2020, 7 DOI:10.1002/admi.202000928.
- X. Sun, Q. Wu, X. Zhang, S. Ren, T. Lei, W. Li, G. Xu and Q. Zhang, Cellulose, 2018, 25, 1103–1115 CrossRef.
- M. Nogi, S. Iwamoto, A. N. Nakagaito and H. Yano, Adv. Mater., 2009, 21, 1595–1598 CrossRef.
- Y. Wang, K. Li, X. Li, H. Cui, G. Liu, H. Xu, X. Wu, W. Yao, B. Zhong, X. Huang, H. Wang and T. Wu, Carbon, 2019, 152, 873–881 CrossRef.
- A. B. Catel, F. F. Camilo and C. Molina, Opt. Mater., 2025, 165, 117092 CrossRef.
- A. Bouftou, K. Aghmih, F. Lakhdar, N. Abidi, S. Gmouh and S. Majid, Measurement: Food, 2024, 15, 100180 Search PubMed.
- S. Paudel, S. Regmi and S. Janaswamy, Food Packag. Shelf Life, 2023, 37, 101090 CrossRef.
- E. Csiszár and S. Nagy, Carbohydr. Polym., 2017, 174, 740–749 CrossRef PubMed.
- P. Ahokas, V. Kunnari, J. Majoinen, A. Harlin and M. Mäkelä, ACS Sustainable Chem. Eng., 2025, 13, 10771–10779 CrossRef PubMed.
- N. Nasiri, H. E. Cainglet, G. Garnier and W. Batchelor, Cellulose, 2024, 31, 7421–7436 Search PubMed.
- S. C. Teixeira, R. R. A. Silva, T. V. de Oliveira, P. C. Stringheta, M. R. M. R. Pinto and N. de F. F. Soares, Food Biosci., 2021, 42, 101202 Search PubMed.
- S. Guzman-Puyol, J. Hierrezuelo, J. J. Benítez, G. Tedeschi, J. M. Porras-Vázquez, A. Heredia, A. Athanassiou, D. Romero and J. A. Heredia-Guerrero, Int. J. Biol. Macromol., 2022, 209, 1985–1994 Search PubMed.
- J. Fernández-Santos, C. Valls, O. Cusola and M. B. Roncero, Int. J. Biol. Macromol., 2022, 211, 218–229 Search PubMed.
- K. Chi and J. M. Catchmark, Food Hydrocolloids, 2018, 80, 195–205 CrossRef.
- B. Fathi Achachlouei and Y. Zahedi, Carbohydr. Polym., 2018, 199, 415–425 Search PubMed.
- J. Valdez García, A. Boding, X. Yang, R. Nizamov, M. S. Reid, K. Junel, K. Miettunen, T. Abitbol and J. Kaschuk, Int. J. Biol. Macromol., 2025, 292, 139203 Search PubMed.
- S. Castro-Hermosa, M. Top, J. Dagar, J. Fahlteich and T. M. Brown, Adv. Electron. Mater., 2019, 5 DOI:10.1002/aelm.201800978.
- Nippon Electric Glass, Ultra-thin glass: G-Leaf, https://www.neg.co.jp/en/products/g-leaf/index.html, (accessed 17 September 2025).
- Q.-H. Lu and F. Zheng, in Advanced Polyimide Materials, Elsevier, 2018, pp. 195–255 Search PubMed.
- T. J. Hickman, L. Tao, N. Stingelin and J. C. Meredith, RSC Sustainability, 2024, 2, 3451–3455 Search PubMed.
- C. Sun, G. Li, J. Wang, Z. Fang, F. Qin, K. Chen, J. Zhou and X. Qiu, Cellulose, 2022, 29, 7111–7124 Search PubMed.
- J. J. Kaschuk, Y. Al Haj, J. Valdez García, A. Kamppinen, O. J. Rojas, T. Abitbol, K. Miettunen and J. Vapaavuori, Carbohydr. Polym., 2024, 332, 121877 CrossRef PubMed.
- A. W. Cindradewi, R. Bandi, C.-W. Park, J.-S. Park, E.-A. Lee, J.-K. Kim, G.-J. Kwon, S.-Y. Han and S.-H. Lee, Polymers, 2021, 13, 2990 CrossRef PubMed.
- M. N. Mirvakili, S. G. Hatzikiriakos and P. Englezos, Phys. Fluids, 2021, 33 DOI:10.1063/5.0043881.
- I. Hasan, J. Wang and M. Tajvidi, Cellulose, 2021, 28, 11345–11366 CrossRef.
- G. Horvat, K. Žvab, Ž. Knez and Z. Novak, Polymers, 2022, 14, 2399 CrossRef PubMed.
- A. Kramar, I. Rodríguez Ortega, G. González-Gaitano and J. González-Benito, Cellulose, 2023, 30, 2037–2052 CrossRef.
- K. Shanmugam, H. Nadeem, C. Browne, G. Garnier and W. Batchelor, Colloids Surf., A, 2020, 589, 124396 CrossRef.
- F. Karasu, L. Müller, H. Ridaoui, M. Ibn ElHaj, G. Flodberg, C. Aulin, L. Axrup and Y. Leterrier, Front. Chem., 2018, 6 DOI:10.3389/fchem.2018.00571.
- P. Holzhey, M. Prettl, S. Collavini, C. Mortan and M. Saliba, Sci. Rep., 2023, 13, 6375 CrossRef PubMed.
- M. Fahland, O. Zywitzki, T. Modes, K. Vondkar, T. Werner, C. Ottermann, M. Berendt and G. Pollack, Thin Solid Films, 2019, 669, 56–59 CrossRef.
- H. E. Lee, Y. Ji, Y. D. Kim, K. Kim and H. O. Seo, Mater. Res. Bull., 2025, 185, 113321 CrossRef.
- S. Lee, S.-W. Lee, S. Bae, J.-K. Hwang, W. Lee, D. Pyun, S.-H. Jeong, K. Kim, J.-S. Hwang, S. Cho, D. Kim, Y. Kang and H.-S. Lee, Energies, 2023, 16, 8061 CrossRef.
- M. Socol, N. Preda, A. Costas, G. Petre, A. Stanculescu, I. Stavarache, G. Popescu-Pelin, S. Iftimie, A. Stochioiu, A. M. Catargiu and G. Socol, Appl. Phys. A, 2025, 131, 17 Search PubMed.
- G. Zhang, H. Wu, C. Chen, T. Wang, J. Yue and C. Liu, Nanoscale Res. Lett., 2015, 10, 76 Search PubMed.
- Z. Guo, X. Li, N. Li, X. Liu, L. Hao, Y. Wang, W. Deng, H. Bai, J. Liang and Z. Chen, Phys. Chem. Chem. Phys., 2024, 26, 4524–4532 RSC.
- J. J. Kaschuk, M. Borghei, K. Solin, A. Tripathi, A. Khakalo, F. A. S. Leite, A. Branco, M. C. Amores de Sousa, E. Frollini and O. J. Rojas, ACS Appl. Polym. Mater., 2021, 3, 2393–2401 CrossRef PubMed.
- M. N. Mirvakili, H. Van Bui, J. R. van Ommen, S. G. Hatzikiriakos and P. Englezos, ACS Appl. Mater. Interfaces, 2016, 8, 13590–13600 Search PubMed.
- S. Yue, S. Wang, D. Han, S. Huang, M. Xiao and Y. Meng, Cellulose, 2022, 29, 8293–8303 CrossRef.
- M. Fazeli, M. Keley and E. Biazar, Int. J. Biol. Macromol., 2018, 116, 272–280 CrossRef PubMed.
- R. Yewale, P. Damlin, M. Salomäki and C. Kvarnström, Mater. Today Commun., 2020, 25, 101398 Search PubMed.
- X. Hu, X. Meng, L. Zhang, Y. Zhang, Z. Cai, Z. Huang, M. Su, Y. Wang, M. Li, F. Li, X. Yao, F. Wang, W. Ma, Y. Chen and Y. Song, Joule, 2019, 3, 2205–2218 CrossRef.
- P. Kumar, W.-J. Lee, Y. Tang, H. Yang, W. Xu, P. Rout, L. You, J. A. Romo, B. Pradhan, J. Mei and L. Dou, EES Sol., 2025, 1, 529 RSC.
- Y. Ding, S. Xiong, L. Sun, Y. Wang, Y. Zhou, Y. Li, J. Peng, K. Fukuda, T. Someya, R. Liu and X. Zhang, Chem. Soc. Rev., 2024, 53, 7784–7827 RSC.
- J. Sun, X. Li, Z. Chen, S. Yan, L. Qin, J. Zeng, S. Wang, J. Xu, L. Zhao, W. Zhou, Q. Wang, H. Gong, A. Lu and J. Yu, Org. Electron., 2018, 63, 392–397 CrossRef.
- R. J. B. Pinto, M. A. Martins, J. M. F. Lucas, C. Vilela, A. J. M. Sales, L. C. Costa, P. A. A. P. Marques and C. S. R. Freire, ACS Appl. Mater. Interfaces, 2020, 12, 34208–34216 CrossRef PubMed.
- J. Kim, W. J. da Silva, A. R. bin Mohd Yusoff and J. Jang, Sci. Rep., 2016, 6, 19813 CrossRef PubMed.
- M. Lagrange, D. P. Langley, G. Giusti, C. Jiménez, Y. Bréchet and D. Bellet, Nanoscale, 2015, 7, 17410–17423 RSC.
- S. Han, S. Hong, J. Ham, J. Yeo, J. Lee, B. Kang, P. Lee, J. Kwon, S. S. Lee, M. Yang and S. H. Ko, Adv. Mater., 2014, 26, 5808–5814 CrossRef PubMed.
- J. Gong, X. Fan, Z. Zong, M. Yang, Y. Sun and G. Zhao, RSC Adv., 2023, 13, 15531–15539 RSC.
- S. J. Lee, Y.-H. Kim, J. K. Kim, H. Baik, J. H. Park, J. Lee, J. Nam, J. H. Park, T.-W. Lee, G.-R. Yi and J. H. Cho, Nanoscale, 2014, 6, 11828–11834 Search PubMed.
- M. Betker, C. Harder, E. Erbes, J. E. Heger, A. E. Alexakis, B. Sochor, Q. Chen, M. Schwartzkopf, V. Körstgens, P. Müller-Buschbaum, K. Schneider, S. A. Techert, L. D. Söderberg and S. V. Roth, ACS Appl. Nano Mater., 2023, 6, 13677–13688 CrossRef.
- D. Li, L. Wang, W. Ji, H. Wang, X. Yue, Q. Sun, L. Li, C. Zhang, J. Liu, G. Lu, H.-D. Yu and W. Huang, ACS Appl. Mater. Interfaces, 2021, 13, 1735–1742 CrossRef CAS PubMed.
- B. Kang and F. Yan, Energy Environ. Sci., 2025, 18, 3917–3954 Search PubMed.
- X. Wang, H. Jin, R. C. R. Nagiri, B. Z. L. Poliquit, J. Subbiah, D. J. Jones, N. Kopidakis, P. L. Burn and J. Yu, Sol. RRL, 2019, 3 DOI:10.1002/solr.201800286.
- J. Linnemann, J. Giorgio, K. Wagner, G. Mathieson, G. G. Wallace and D. L. Officer, J. Mater. Chem. A, 2015, 3, 3266–3270 RSC.
- Y. Galagan, D. J. D. Moet, D. C. Hermes, P. W. M. Blom and R. Andriessen, Org. Electron., 2012, 13, 3310–3314 CrossRef CAS.
- H. Li, X. Li, W. Wang, J. Huang, J. Li, S. Huang, B. Fan, J. Fang and W. Song, Sol. Energy, 2019, 188, 158–163 CrossRef CAS.
- F. C. Krebs, T. Tromholt and M. Jørgensen, Nanoscale, 2010, 2, 873 RSC.
- N. Espinosa, R. García-Valverde, A. Urbina and F. C. Krebs, Sol. Energy Mater. Sol. Cells, 2011, 95, 1293–1302 CrossRef CAS.
- M. Österberg, M. S. Peresin, L.-S. Johansson and T. Tammelin, Cellulose, 2013, 20, 983–990 CrossRef.
- M. Zuber, K. M. Zia, I. A. Bhatti, Z. Ali, M. U. Arshad and M. J. Saif, Int. J. Biol. Macromol., 2012, 51, 743–748 CrossRef CAS PubMed.
- C. Wang, Z. Yuan, A. Wang, J. Qu, Z. Fang and Y. Wen, Cellulose, 2020, 27, 2041–2051 CrossRef CAS.
- R. A. Wolf, Atmospheric pressure plasma for surface modification, John Wiley & Sons, Hoboken, N.J, 1st edn, 2013 Search PubMed.
- J. L. Carter, C. A. Kelly and M. J. Jenkins, Polym. J., 2023, 55, 253–260 CrossRef CAS.
- D. Shin, D. Kang, J.-B. Lee, J.-H. Ahn, I.-W. Cho, M.-Y. Ryu, S. W. Cho, N. E. Jung, H. Lee and Y. Yi, ACS Appl. Mater. Interfaces, 2019, 11, 17028–17034 Search PubMed.
- S. Y. Lee, G. Y. Park, N. Kim, M. Kang, K. Kim, S. Cho, W. Choi, J. S. Myung and D. S. Ham, Energy Technol., 2023, 12 DOI:10.1002/ente.202301058.
- Z. Xu, X. Zhou, X. Li and P. Zhang, Sol. RRL, 2022, 6 DOI:10.1002/solr.202200092.
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- F. Gayot, E. Bruhat, M. Bouttemy, M. Frégnaux, E. De Vito, J.-P. Kleider, S. Cros and M. Manceau, ACS Appl. Energy Mater., 2023, 6, 11849–11860 Search PubMed.
- A. A. Eliwi, M. Malekshahi Byranvand, P. Fassl, M. R. Khan, I. M. Hossain, M. Frericks, S. Ternes, T. Abzieher, J. A. Schwenzer, T. Mayer, J. P. Hofmann, B. S. Richards, U. Lemmer, M. Saliba and U. W. Paetzold, Mater. Adv., 2022, 3, 456–466 Search PubMed.
- Z. Li, Z. Wang, C. Jia, Z. Wan, C. Zhi, C. Li, M. Zhang, C. Zhang and Z. Li, Nano Energy, 2022, 94, 106919 Search PubMed.
- S. Y. Park and K. Zhu, Adv. Mater., 2022, 34 DOI:10.1002/adma.202110438.
- X. Zhang, F. Zabihi, H. Xiong, M. Eslamian, C. Hou, M. Zhu, H. Wang and Q. Zhang, Chem. Eng. J., 2020, 394, 124887 Search PubMed.
- J. Wang, Z. Dong, J. Wang, J. Zhang, Z. Zhai, F. Qiu, J. Wu, Y. Lin and J. Zhang, J. Mater. Chem. A, 2023, 11, 22206–22215 RSC.
- S. Li, X. Wang, N. Huang, S. He, L. Qiu and Y. Qi, EnergyChem, 2024, 6, 100135 CrossRef CAS.
- A. Кabdiyeva, A. Zeinidenov, D. Abeuov, X. Rozhkova and A. Aimukhanov, Opt. Mater., 2026, 169, 117581 Search PubMed.
- L. Zhou, Y. Du, J. Zhang, X. Feng, D. Chen, W. Zhu, H. Xi, J. Zhang, C. Zhang and Y. Hao, Chem. Eng. J., 2025, 511, 161813 Search PubMed.
- S. Liu, J. Li, W. Xiao, R. Chen, Z. Sun, Y. Zhang, X. Lei, S. Hu, M. Kober-Czerny, J. Wang, F. Ren, Q. Zhou, H. Raza, Y. Gao, Y. Ji, S. Li, H. Li, L. Qiu, W. Huang, Y. Zhao, B. Xu, Z. Liu, H. J. Snaith, N.-G. Park and W. Chen, Nature, 2024, 632, 536–542 Search PubMed.
- Y. Zheng, T. Niu, L. Chao, Y. Xia and Y. Chen, J. Energy Chem., 2025, 107, 74–86 CrossRef CAS.
- J. J. Lee, S. Gandla, B. Lim, S. Kang, S. Kim, S. Lee and S. Kim, NPG Asia Mater., 2020, 12, 65 Search PubMed.
- X. Dong, X. Zhou, Y. Liu, S. Xiong, J. Cheng, Y. Jiang and Y. Zhou, Energy Environ. Sci., 2023, 16, 1511–1519 RSC.
- Y. Yoo, G. Seo, H. J. Park, J. Kim, J. Jang, W. Cho, J. H. Kim, J. Shin, J. S. Choi, D. Lee, S.-W. Baek, S. Lee, S. M. Kang, M. Kim, Y.-E. Sung and S. Bae, J. Mater. Chem. A, 2024, 12, 1562–1572 RSC.
- G. C. Wilkes, X. Deng, J. J. Choi and M. C. Gupta, ACS Appl. Mater. Interfaces, 2018, 10, 41312–41317 Search PubMed.
- A. H. Ghahremani, B. Martin, A. Gupta, J. Bahadur, K. Ankireddy and T. Druffel, Mater. Des., 2020, 185, 108237 CrossRef CAS.
- P. Wang, X. Zhang, Z. Zhang, H. Ling, L. Tao, K. Sohail, X. Li, X. Fu, X. Zhang, R. Wang, Y. Wang, J. Luo, M. J. Ko, J. Chen and Y. Li, ACS Appl. Energy Mater., 2023, 6, 6554–6562 Search PubMed.
- Z. Xu, L. Huang, Y. Jiang, Z. Li, C. Chen, Z. He, J. Liu, Y. Fang, K. Wang, G. Zhou, J.-M. Liu and J. Gao, ACS Appl. Mater. Interfaces, 2022, 14, 41037–41044 Search PubMed.
- P.-H. Lee, T.-T. Wu, C.-F. Li, D. Głowienka, Y.-H. Sun, Y.-T. Lin, H.-W. Yen, C.-G. Huang, Y. Galagan, Y.-C. Huang and W.-F. Su, Chem. Eng. J., 2021, 412, 128746 CrossRef CAS.
- Y. Wang, Q. Chen, J. Fu, Z. Liu, Z. Sun, S. Zhang, Y. Zhu, X. Jia, J. Zhang, N. Yuan, Y. Zhou, B. Song and Y. Li, Chem. Eng. J., 2022, 433, 133265 CrossRef CAS.
- E. J. Cassella, E. L. K. Spooner, J. A. Smith, T. Thornber, M. E. O’Kane, R. D. J. Oliver, T. E. Catley, S. Choudhary, C. J. Wood, D. B. Hammond, H. J. Snaith and D. G. Lidzey, Adv. Energy Mater., 2023, 13 DOI:10.1002/aenm.202203468.
- F. U. Kosasih, E. Erdenebileg, N. Mathews, S. G. Mhaisalkar and A. Bruno, Joule, 2022, 6, 2692–2734 CrossRef CAS.
- S.-R. Bae, D. Y. Heo and S. Y. Kim, Mater. Today Adv., 2022, 14, 100232 Search PubMed.
- S. Tao, I. Schmidt, G. Brocks, J. Jiang, I. Tranca, K. Meerholz and S. Olthof, Nat. Commun., 2019, 10, 2560 Search PubMed.
- S. Chaudhary, S. K. Gupta and C. M. Singh Negi, Mater. Sci. Semicond. Process., 2020, 109, 104916 CrossRef CAS.
- M. Fievez, P. J. Singh Rana, T. M. Koh, M. Manceau, J. H. Lew, N. F. Jamaludin, B. Ghosh, A. Bruno, S. Cros, S. Berson, S. G. Mhaisalkar and W. L. Leong, Sol. Energy Mater. Sol. Cells, 2021, 230, 111189 CrossRef CAS.
- S. Sánchez and A. Hagfeldt, in Perovskite Solar Cells, Wiley, 2021, pp. 33–89 Search PubMed.
- M. A. Slimani, S. G. Cloutier and R. Izquierdo, Nanomaterials, 2024, 14, 886 CrossRef CAS PubMed.
- A. D. Taylor, Q. Sun, K. P. Goetz, Q. An, T. Schramm, Y. Hofstetter, M. Litterst, F. Paulus and Y. Vaynzof, Nat. Commun., 2021, 12, 1878 CrossRef CAS PubMed.
- M. Li, Y.-M. Xie, X. Xu, Y. Huo, S.-W. Tsang, Q.-D. Yang and Y. Cheng, Org. Electron., 2018, 63, 159–165 CrossRef CAS.
- D. Angmo, G. DeLuca, A. D. Scully, A. S. R. Chesman, A. Seeber, C. Zuo, D. Vak, U. Bach and M. Gao, Cell Rep. Phys. Sci., 2021, 2, 100293 CrossRef CAS.
- P. Kumar, Appl. Phys. A, 2024, 130, 566 CrossRef CAS.
- J. Han, R. H. Kim, S. Huang, J. Kim and J. S. Yun, Sol. RRL, 2024, 8 DOI:10.1002/solr.202400262.
- H.-S. Yun, H. W. Kwon, M. J. Paik, S. Hong, J. Kim, E. Noh, J. Park, Y. Lee and S. Il Seok, Nat. Energy, 2022, 7, 828–834 Search PubMed.
- X. Huang, F. Cao, S. Zhan, Q. Feng, M. Zhu, Z. Su, X. Gao, J. Yin, J. Li, N. Zheng and B. Wu, Joule, 2023, 7, 1556–1573 CrossRef CAS.
- Y. Yang, Y. Wang, Z. Qu, K. Zhang, T. Liang, S. Chen, W. Lv, F. Min, Y. Chen, Y. Qiao and Y. Song, Angew. Chem., Int. Ed., 2023, 62 DOI:10.1002/anie.202300971.
- E. Rezaee, D. I. Kutsarov, J. Zhang, G. Koutsourakis, B. Li, F. A. Castro and S. R. P. Silva, Small Methods DOI:10.1002/smtd.202300564.
- C. Duan, H. Gao, K. Xiao, V. Yeddu, B. Wang, R. Lin, H. Sun, P. Wu, Y. Ahmed, A. D. Bui, X. Zheng, Y. Wang, J. Wen, Y. Wang, W. Ou, C. Liu, Y. Zhang, H. Nguyen, H. Luo, L. Li, Y. Liu, X. Luo, M. I. Saidaminov and H. Tan, Nat. Energy, 2025, 10, 318–328 CrossRef CAS.
- Y. Zhang, L. Ren, P. Zhai, J. Xin, J. Wu, Q. Zhang, X. Chen, K. Zhao, L. Zhang and S. (Frank) Liu, Energy Environ. Sci., 2024, 17, 296–306 Search PubMed.
- D. V. Shinde, L. Pyeon, M. Pei, G.-W. Kim, H. Yang and T. Park, ACS Appl. Mater. Interfaces, 2017, 9, 14023–14030 Search PubMed.
- T. Hsieh, M. Pylnev, E. Palomares and T. Wei, Adv. Funct. Mater., 2020, 30 DOI:10.1002/adfm.201909644.
- M. T. Hoang, Y. Yang, N. D. Pham and H. Wang, J. Phys. Chem. Lett., 2024, 15, 6880–6889 CrossRef CAS PubMed.
- Y. Yang, Z. Huang, H. Gao, Z. Xu, W. Fang, Y. Chen, Y. Hu, Z. Yi, J. Huang and H. Zhu, RSC Adv., 2024, 14, 32370–32388 Search PubMed.
- R. Vidal, J.-A. Alberola-Borràs, S. N. Habisreutinger, J.-L. Gimeno-Molina, D. T. Moore, T. H. Schloemer, I. Mora-Seró, J. J. Berry and J. M. Luther, Nat. Sustain., 2020, 4, 277–285 CrossRef.
- N.-G. Park, Nat. Sustain., 2020, 4, 192–193 Search PubMed.
- E. Greul, P. Docampo and T. Bein, Z. Anorg. Allg. Chem., 2017, 643, 1704–1711 CrossRef CAS.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, Green Chem., 2016, 18, 288–296 RSC.
- B. Wang, Z.-G. Zhang, S. Ye, H. Rao, Z. Bian, C. Huang and Y. Li, J. Mater. Chem. A, 2016, 4, 17267–17273 RSC.
- H. Xiong, F. Zabihi, H. Wang, Q. Zhang and M. Eslamian, Nanoscale, 2018, 10, 8526–8535 Search PubMed.
- M.-R. Ahmadian-Yazdi and M. Eslamian, Mater. Today Commun., 2018, 14, 151–159 CrossRef CAS.
- H. Dong, S. Pang, F. He, H. Yang, W. Zhu, D. Chen, H. Xi, J. Zhang, Y. Hao and C. Zhang, Sol. RRL, 2021, 5 DOI:10.1002/solr.202100097.
- C. Dong, J. Chen, C.-H. Chen, Y.-R. Shi, W.-F. Yang, K.-L. Wang, Z.-K. Wang and L.-S. Liao, Nano Energy, 2022, 94, 106866 CrossRef CAS.
- Y. Dong, F. M. Rombach, G. Min, H. J. Snaith, C.-T. Lin, S. A. Haque and T. J. Macdonald, Mater. Sci. Eng., R, 2025, 162, 100875 CrossRef.
- T. H. Schloemer, J. A. Christians, J. M. Luther and A. Sellinger, Chem. Sci., 2019, 10, 1904–1935 RSC.
- S. Daskeviciute-Geguziene, A. Magomedov, M. Daskeviciene, K. Genevičius, N. Nekrašas, V. Jankauskas, K. Kantminiene, M. D. McGehee and V. Getautis, Chem. Commun., 2022, 58, 7495–7498 RSC.
- S. Daskeviciute, Y. Zhang, M. Daskeviciene, K. Rakstys, J. Petrulevicius, V. Jankauskas, V. Getautis and M. K. Nazeeruddin, Sol. RRL, 2025, 9 DOI:10.1002/solr.202500034.
- F. M. Rombach, S. A. Haque and T. J. Macdonald, Energy Environ. Sci., 2021, 14, 5161–5190 RSC.
- Y. Ko, Y. Kim, C. Lee, Y. Kim and Y. Jun, ACS Appl. Mater. Interfaces, 2018, 10, 11633–11641 CrossRef CAS PubMed.
- H. Kassem, A. Salehi and M. Kahrizi, Energy Technol., 2024, 12 DOI:10.1002/ente.202301032.
- M. J. Jeong, K. M. Yeom, S. J. Kim, E. H. Jung and J. H. Noh, Energy Environ. Sci., 2021, 14, 2419–2428 RSC.
- Y. Zhang, Y. Ren, X. Xie, Y. Wei, L. He, L. Fang, J. Zhang, Y. Yuan and P. Wang, Adv. Funct. Mater., 2021, 32 DOI:10.1002/adfm.202108855.
- Y. Xu, Z. Chu, H. Wang, Z. Xing, B. Fan, X. Hu and Y. Chen, Sci. China: Chem., 2024, 67, 2335–2340 CrossRef CAS.
- S. Chapagain, B. Martin, P. Armstrong, C. L. Perkins, M. O. Reese, T. Druffel and C. A. Grapperhaus, ACS Appl. Energy Mater., 2023, 6, 4496–4502 CrossRef CAS.
- W. Zhang, X. Guo, Z. Cui, H. Yuan, Y. Li, W. Li, X. Li and J. Fang, Adv. Mater., 2024, 36 DOI:10.1002/adma.202311025.
- S. Hyun Park, I. Su Jin and J. Woong Jung, Chem. Eng. J., 2021, 425, 131475 CrossRef CAS.
- B. Guo, X. Chen, H. Luo, G. O. Odunmbaku, T. Jiang, N. A. N. Ouedraogo, Z. Huang, Q. Gao, B. Zhang, Y. Ouyang, Y. Pan, T. Xia, C. Wang, K. Zahid, C. Li, S. Chen, Y. Zheng, Z. Ma and K. Sun, Sol. RRL, 2023, 8 DOI:10.1002/solr.202300934.
- F. Isabelli, F. Di Giacomo, H. Gorter, F. Brunetti, P. Groen, R. Andriessen and Y. Galagan, ACS Appl. Energy Mater., 2018, 1, 6056–6063 CrossRef CAS.
- A. Hassan, M. I. Syauqi, Y. Liu, Z. Ke, W. Lin, Z. Wang, Y. Jin and R. Azmi, Mater. Sci. Eng., R, 2025, 166, 101023 Search PubMed.
- Q. Fu, J. He, J. Zheng, Y. Xing, Q. Wang, J. Xiong, W. Zhu, R. Xuan, A. Fu, X. Gan, L. Huang, X. Liu, Y. Zhu and J. Zhang, J. Alloys Compd., 2024, 1001, 175058 CrossRef CAS.
- B. Yu, J. Shi, Y. Li, S. Tan, Y. Cui, F. Meng, H. Wu, Y. Luo, D. Li and Q. Meng, Nat. Commun., 2025, 16, 3328 CrossRef CAS PubMed.
- L. J. Sutherland, D. Vak, M. Gao, T. A. N. Peiris, J. Jasieniak, G. P. Simon and H. Weerasinghe, Adv. Energy Mater., 2022, 12 DOI:10.1002/aenm.202202142.
- J. Batta-Mpouma, G. Kandhola, J. Sakon and J.-W. Kim, Biomacromolecules, 2022, 23, 4085–4096 CrossRef CAS PubMed.
- C. Liu, X. Li, T. Liu, Z. Liu, N. Li, Y. Zhang, C. Xiao and X. Feng, J. Membr. Sci., 2016, 512, 1–12 CrossRef CAS.
- P. Wu, S. Wang, X. Li and F. Zhang, Matter, 2022, 5, 1137–1161 CrossRef CAS.
- G. Zhang, Y. Zheng, H. Wang, G. Ding, F. Yang, Y. Xu, J. Yu and Y. Shao, Joule, 2024, 8, 496–508 CrossRef CAS.
- R. N. Hussein and T. B. Carmichael, ACS Appl. Electron. Mater., 2025, 7, 9249–9266 CrossRef CAS.
- Y. Yang, J. Zhao, H. Yang, X. Yang, Y. Lu, Z. Huang, S. Duo, Z. Xiong and X. Hu, Nat. Commun., 2025, 16, 8993 CrossRef CAS PubMed.
- J. Li, X. Qiao, B. He, Y. Zhang, S. Pal, L. Sun, M. Bilal, Z. Su, X. Gao, J. Briscoe, I. Abrahams, M. Li, Z. Li and Y. Lu, Energy Environ. Sci., 2025, 18, 5632–5642 RSC.
- Y. Xie, H. Sui, X. Chen, X. Wang, C. Li, H. Zhao, H. Zhong, C. Yao, S. Zhang and Q. Zhang, Electrochim. Acta, 2026, 545, 147731 CrossRef CAS.
- S. Castro-Hermosa, J. Dagar, A. Marsella and T. M. Brown, IEEE Electron Device Lett., 2017, 38, 1278–1281 CAS.
- L. Gao, L. Chao, M. Hou, J. Liang, Y. Chen, H.-D. Yu and W. Huang, npj Flexible Electron., 2019, 3, 4 CrossRef.
- P. Holzhey, M. Prettl, S. Collavini, N. L. Chang and M. Saliba, Joule, 2023, 7, 257–271 CrossRef CAS.
- L. McGovern, E. C. Garnett, S. Veenstra and B. van der Zwaan, Sustainable Energy Fuels, 2023, 7, 5259–5270 RSC.
- T. Okoroafor, A. Maalouf, S. Oez, V. Babu, B. Wilk and S. Resalati, J. Cleaner Prod., 2022, 373, 133665 CrossRef CAS.
- X. Tian, S. D. Stranks and F. You, Nat. Sustain., 2021, 4, 821–829 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |