Tandem electrocatalysis for continuous production of a zero-carbon cement clinker precursor and formate
Xiaoling
Xue
ab,
Haozhen
Li
ab,
Liuke
Duan
ab,
Zhefei
Pan
 *ab,
Xun
Zhu
*ab,
Xun
Zhu
 *ab,
Dingding
Ye
*ab,
Dingding
Ye
 ab,
Yang
Yang
ab,
Yang
Yang
 ab,
Hong
Wang
ab,
Hong
Wang
 ab,
Xiao
Yan
ab,
Xiaolong
Zu
c,
Xiao
Zhang
def,
Liang
An
ab,
Xiao
Yan
ab,
Xiaolong
Zu
c,
Xiao
Zhang
def,
Liang
An
 def,
Rong
Chen
def,
Rong
Chen
 *ab and
Qiang
Liao
*ab and
Qiang
Liao
 *ab
*ab
aKey Laboratory of Low-grade Energy Utilization Technologies and Systems (Chongqing University), Ministry of Education, Chongqing, 400030, China. E-mail: zhefei.pan@cqu.edu.cn; zhuxun@cqu.edu.cn; rchen@cqu.edu.cn; lqzx@cqu.edu.cn
bInstitute of Engineering Thermophysics, Chongqing University, Chongqing 400030, China
cInnovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), Xiamen 361005, China
dDepartment of Mechanical Engineering, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong SAR, China
eResearch Institute for Advanced Manufacturing, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong SAR, China
fHong Kong Polytechnic University, Shenzhen Research Institute, Shenzhen, China
First published on 26th December 2025
Abstract
The cement industry faces a critical challenge in decarbonization, as conventional production processes contribute significantly to global CO2 emissions through both fossil fuel combustion and limestone calcination. This study presents an innovative electrochemical tandem system designed to address these twin carbon challenges simultaneously. Our system achieves fossil-free clinker precursor synthesis, while simultaneously converting limestone-derived CO2 into valuable formate through direct electrochemical processes, creating a sustainable cement production pathway. The engineered three-chamber flow reactor architecture demonstrates exceptional operational stability exceeding 100 hours achieved through strategic compartmentalization and optimized operational parameters. Comprehensive techno-economic analysis confirms the system's viability as a transformative paradigm, offering both environmental and economic benefits compared to traditional cement production methods.
Green foundation1. This work establishes a closed-loop carbon cycle by coupling CO2 electrolysis with cement clinker production, converting process-emitted CO2 into formate and eliminating fossil fuel combustion.2. Continuous electrochemical production of Ca(OH)2 was achieved with a faradaic efficiency greater than 90% and stable operation exceeding 100 hours, coupled with CO2 to formate conversion at a faradaic efficiency of about 60%, providing quantitative evidence of technical viability. 3. Further greening involves replacing precious metals with Earth-abundant alternatives, utilizing renewable power, and sourcing calcium from industrial waste. Alongside reactor optimization to reduce energy consumption, these advancements require rigorous durability testing and life-cycle assessments to confirm their long-term scalability and environmental impact. |
Introduction
The accelerating urbanization and industrial expansion have significantly increased cement demand for modern infrastructure development.1–4 However, this surge exacerbates environmental burdens, as conventional cement production accounts for approximately 8% of anthropogenic CO2 emissions and 7% of industrial energy consumption, primarily due to limestone calcination and fossil fuel combustion.5–8 To address this dilemma, emerging electrochemical strategies powered by renewable energy show promise in decoupling cement production from fossil fuel dependence.9–12Electrification efforts in cement production have seen incremental progress, mainly through electric heating systems replacing fossil fuel combustion and the production of clean fuels via electricity for sintering kilns.10,13–15 However, these modifications remain fundamentally limited by multiple energy conversions, leading to significant electrical waste, and an unaltered limestone calcination chemistry resulting in persistently severe carbon emissions.16–18 Electrochemical clinker synthesis from limestone represents a breakthrough approach that directly integrates electrical energy into cement chemistry.19–21 While traditional H-type reactors have demonstrated conceptual viability at the lab scale,22–24 they suffer from critical operational barriers, including prohibitively low current densities (<10 mA cm−2) and the complexity of product separation coupled with discontinuous operation. Flow reactors theoretically enable continuous clinker production through sustained reactant feeding.25–27 However, they suffer from improper reaction coupling and insufficient product–reactant segregation, which induce severe membrane fouling and electrode clogging, ultimately collapsing operational stability.19 Additionally, the CO2 released from limestone decomposition constitutes a significant source of carbon emissions in cement production. Despite the severity of this emission source, direct conversion and utilization of such CO2 streams remain underexplored. To date, only Xie et al. have reported CO2 reduction through a pulsed potential approach, but with a disappointingly low faradaic efficiency (FE) of less than 20%.23 This low efficiency underscores significant challenges in practical implementation. Furthermore, the broader research community has largely overlooked systematic strategies for integrating CO2 capture and direct conversion technologies into cement manufacturing workflows, leaving a critical gap in decarbonizing this emission-intensive sector.21,28,29
Building upon this foundation, this study proposes a tandem electrochemical system that innovatively addresses the dual challenges. Through strategic reaction zone compartmentalization and operational parameter optimization, a three-chamber flow reactor achieves continuous production of high-purity calcium hydroxide (Ca(OH)2), specifically by generating it in a middle chamber physically decoupled from the electrochemical reaction zones. This design effectively prevents solid product deposition on electrode surfaces, thereby maintaining electrochemical continuity while enabling uninterrupted production and collection of the target product. By integrating in situ CO2 capture and conversion, the system achieves zero-carbon clinker precursor production while co-generating formate with high efficiency. This tandem architecture establishes a direct pathway for converting renewable electricity into zero-carbon cement. Fig. 1 shows the comparison of the traditional cement production process with a novel electrochemically mediated pathway designed to reduce the associated energy demand and CO2 emissions. The upper panel delineates the conventional route wherein raw materials are extracted through quarrying and subsequently crushed, ground, and calcined. The resultant material is sintered in a high-temperature kiln, cooled, milled, and eventually distributed via logistics, underpinning a process characterized by significant energy consumption and substantial CO2 emissions. In contrast, the tandem electrocatalytic approach presented here integrates two electrochemical processes into the cement production cycle, effectively transforming CO2 from a waste byproduct into a valuable product of formate. This integrated method leverages fundamental electrochemical and precipitation reactions to enable a lower-carbon, more energy-efficient production route. The heart of this strategy is the tandem electrochemical processes. In the first step, the calcium ions (Ca2+) and hydroxide ions (OH−) are extracted from calcium carbonate (CaCO3) and water, respectively. Driven by the electric field, Ca(OH)2 is generated by the precipitation reaction. In the second step, the CO2 emitted from the first step is captured by the alkali and electrolytically reduced to formate. Consequently, a zero-carbon cement clinker precursor and value-added formate are produced eventually. Compared to the conventional CCUS (Carbon Capture, Utilization and Storage) pathway, a well-established industrial carbon reduction process, this in situ electrochemical conversion approach eliminates intermediate steps, reduces capital investment in equipment, and mitigates transportation risks associated with CO2 handling.28 Following precipitation, the Ca(OH)2 is further processed via sintering powered by the byproduct hydrogen in the first step that forms high-quality cement. Collectively, this integrated scheme not only captures CO2 that would otherwise be emitted, but also utilizes it as a feedstock, thereby closing the carbon loop within the manufacturing process. By coupling CO2 utilization with the formation of key cement precursors, the electrochemically mediated pathway offers a promising route to reduce greenhouse gas emissions and overall energy consumption, aligning industrial cement production with modern sustainability and decarbonization goals.
 | ||
| Fig. 1 Comparative schematics of conventional thermal versus tandem electrochemical cement production pathways. | ||
Results and discussion
Electrode characterization
To achieve efficient electrochemical production of Ca(OH)2 and CO2 conversion, different electrode materials were employed and characterized using scanning electron microscopy (SEM) and elemental mapping. As shown in Fig. 2a, the anode responsible for the oxygen evolution reaction (OER) during the production of Ca(OH)2 is Ir-coated Ti felt. The SEM image reveals a porous and fibrous structure, which enhances the electrochemically active surface area and facilitates the OER. The corresponding elemental mapping confirms the uniform distribution of Ir on the Ti felt, ensuring effective catalytic activity and stability during operation. The high conductivity and corrosion resistance of Ir/Ti electrodes make them well-suited for a long-term OER in a neutral or acidic environment. The cathode used for Ca(OH)2 production is Pt-coated Ti felt. Fig. 2b presents the SEM image, demonstrating a similar porous structure to the anode, which aids in efficient ion and electron transport. The X-ray photoelectron spectroscopy (XPS) spectra of Pt and Ir elements are shown in SI Fig. 1. The elemental mapping verifies the homogeneous dispersion of Pt over the Ti felt substrate. This uniform distribution maximizes the population of accessible active sites and facilitates charge transfer, which are key factors contributing to the excellent catalytic activity for hydroxide ion generation.30,31 The Pt/Ti electrode facilitates the precipitation of Ca(OH)2 by increasing the local alkalinity in the middle chamber, playing a crucial role in the electrochemical reactor.The electrochemical conversion of CO2 from limestone decomposition is a critical pathway for cement industry carbon reduction. Among diverse CO2 reduction products, formate stands out as the most promising high-value-added product, owing to its exceptional economic viability and energy storage potential.32–34 However, efficient formate production faces dual challenges, including a high thermodynamic barrier for CO2 activation and a severe hydrogen evolution reaction (HER).35,36 These challenges drive innovations in electrocatalyst design.37 Notably, bismuth oxycarbonate (Bi2O2CO3) emerges as an optimal candidate due to its unique electronic structure and nanoconfinement effects.38,39 To leverage these advantages, we synthesized lattice distortion-enriched bismuth oxycarbonate nanosheets via a hydrothermal method and deposited on carbon paper to fabricate the cathode. The crystalline phase of the product was identified by X-ray diffraction (XRD). The XRD pattern (SI Fig. 2) confirms the successful synthesis of Bi2O2CO3, while the high-resolution transmission electron microscopy (HRTEM) image (SI Fig. 3) reveals clear lattice fringes, further corroborating its high crystallinity. As shown in Fig. 2c, the SEM image reveals the porous structure of the Bi2O2CO3 catalyst layer, which provides a high surface area favorable for reactant adsorption and electrochemical conversion. The elemental mapping confirms the uniform distribution of Bi across the carbon substrate. The magnified SEM image further highlights the sheet-like features of the Bi2O2CO3 catalyst, which contribute to enhanced catalytic activity and selectivity toward formate formation. The hierarchical structure constructed by the catalyst layer and carbon paper serves as an effective support, facilitating mass and electron transport. The relevant electrochemical characterization demonstrates its outstanding performance in charge transfer and CO2 conversion capability. A substantial electrochemically active surface area (ECSA), as shown in SI Fig. 4a, indicates a high density of active sites. Furthermore, linear sweep voltammetry (LSV) confirms its high CO2 reduction activity (SI Fig. 4b), and electrochemical impedance spectroscopy (EIS) reveals highly efficient charge transfer kinetics (SI Fig. 4c). The anode employed in this setup was identical to that used for Ca(OH)2 production (Ir-coated Ti felt).
Electrochemical production of calcium hydroxide
To investigate the feasibility of electrochemical Ca(OH)2 production for cement clinker production, a three-chamber flow reactor is constructed. The reactor is composed of three compartments (anode, middle chamber, and cathode) separated by a cation exchange membrane (CEM) and an anion exchange membrane (AEM) (SI Fig. 5). At the anode, a mixed suspension of CaCO3 particles and Ca(NO3)2 solution is fed into the reactor. Ir catalyzes the oxygen evolution reaction to produce protons, oxygen, and electrons, and protons react with CaCO3 to produce Ca2+ and CO2, as shown in eqn (1) and (2):| 2H2O → O2 + 4H+ + 4e− | (1) |
| 2H+ + CaCO3 → H2O + CO2 + Ca2+ | (2) |
Meanwhile, at the cathode, KOH solution is fed into the reactor. Pt catalyzes the hydrogen evolution reaction to produce OH− and hydrogen, as shown in eqn (3):
| 2H2O + 2e− →H2 + 2OH− | (3) |
Driven by the electric field, Ca2+ and OH− transport through the CEM and AEM, respectively, and form Ca(OH)2 when exceeding the solubility limit.
Fig. 3a illustrates dynamic changes in electrolyte solutions and gaseous products across chambers during the 100-minute operation. The gradual accumulation of gaseous products demonstrates sustained electrochemical evolution of hydrogen and oxygen. Concurrently, the clarification of the anode chamber solution (left) indicates the continuous conversion of limestone into CO2 and Ca2+ by H+ generated in situ, followed by migration to the middle chamber. Correspondingly, the middle chamber (right) exhibits progressive formation of precipitates. The crystalline structure of the precipitated product was analyzed using XRD, as presented in Fig. 3b. The diffraction peaks of the collected solid phase match well with the reference pattern of Ca(OH)2 (PDF#44-1481). This authenticates the formation of Ca(OH)2 as the product of the electrochemically driven synthesis. Further morphological characterization was conducted using SEM, as depicted in Fig. 3c. The precipitates exhibit a typical platelet-like structure, which is characteristic of Ca(OH)2, further supporting our XRD findings. To evaluate the operating stability of the electrochemical reactor, different AEMs were tested, and their voltage profiles over time are shown in Fig. 3d. Among the tested membranes, FAA-3-50 AEM demonstrated the most stable performance, with minimal voltage fluctuations over the experimental period, and its membrane structure remained intact after the reaction (SI Fig. 6a and b). In contrast, PiperION A40 and Sustainion AEMs exhibited noticeable increases in voltage. SEM images revealed structural damage in the post-reaction Sustainion AEM (SI Fig. 6c and d), indicating potential membrane degradation or fouling that could hinder long-term operation.
Compared to complex collection procedures such as drying and centrifugation required for solid products, gaseous products offer significant advantages in collection convenience and detectability via gas chromatography. Consequently, the performance of this electrocatalytic system was evaluated by monitoring the generation rates of O2 and CO2. The faradaic efficiency of O2 (FE O2) serves as a critical indicator of electrode electrochemical activity, while the CO2 evolution efficiency (FE CO2) directly reflects the conversion efficiency of calcium carbonate, as shown in Fig. 3e. The FE based on O2 remained near 100% across different anode flow rates, while it based on CO2 is lower than 90% at low flow rates with minor variations attributed to mass transport effects. When the flow rate is low with insufficient CaCO3 supply, the generated protons cannot be effectively consumed by CaCO3 to produce CO2. Unfavorably, the protons directly transport through the CEM to the middle chamber without contributing to the Ca2+ generation. When the flow rate is higher than 10 mL min−1, the mass transport effects are eliminated by sufficient CaCO3 supply. Concurrently, the voltage increased with higher flow rates, likely due to the increased ion transport resistance. When more solid CaCO3 particles enter the anode, the conduction of ions and electrons in the anode can be negatively affected. The influence of current density on the FE and reactor voltage was investigated and is presented in Fig. 3f. The FE remained relatively high across a broad range of current densities, although a slight decline was observed at higher current densities due to unmatched Ca2+ transport to the middle chamber. The voltage exhibited a strong dependence on current density, increasing linearly as expected from Ohmic losses. Temperature plays a crucial role in electrochemical reactions, affecting both reaction kinetics and FE. As illustrated in Fig. 3g, increasing the operating temperature led to a noticeable decline in reactor voltage, due to enhanced ionic conductivity and reaction kinetics at elevated temperatures. Meanwhile, the FE stayed high and stable. A high FE above 90% demonstrates that electrochemically driven synthesis of Ca(OH)2 is a viable approach, offering direct and controllable production under ambient conditions. The choice of the anion exchange membrane is critical for long-term operation, as evidenced by the stability tests. Additionally, optimizing the current density and operating temperature is essential to balance efficiency and energy consumption. The promising stability with minimal voltage fluctuations observed in the optimized system suggests that this method could provide a sustainable alternative to conventional lime production methods, which are energy-intensive and contribute significantly to CO2 emissions.
Operation stability of electrochemical Ca(OH)2 production
To evaluate the long-term stability of the electrochemical Ca(OH)2 production process, the reactor was operated at a current density of 100 mA cm−2, and its performance was monitored over time. As shown in Fig. 4a, the applied voltage remained stable at approximately 4 V throughout the 100-hour operation, while the FE consistently exceeded 90%. The middle chamber of the reactor was refreshed via in situ chemical cleaning with 0.5 M HNO3 for 10 min without reactor disassembly when the reactor voltage exceeded 5 V. Detailed voltage behaviors are shown in SI Fig. 7. These results indicate that the electrochemical system maintains high efficiency and stability under continuous operation, demonstrating its potential for scalable applications in sustainable cement production. It is noteworthy that the complex ion transport processes involving both ion-exchange membranes and the middle chamber contributed to a significant ohmic loss. Furthermore, potential membrane fouling could further increase the overall internal resistance, leading to a slight upward drift in the operating cell voltage over time. Consequently, the overall optimization of this electrochemical system, aiming to reduce the operating voltage and enhance the long-term durability, constitutes a primary focus of our future research.Electrochemical CO2 conversion performance
To achieve the production of a zero-carbon cement clinker precursor, the CO2 derived from the CaCO3 decomposition should be captured and converted, which accounts for nearly half of the total CO2 emissions. The produced CO2 was captured by reacting with a mixed solution of KOH and polyethylene glycol, resulting in the precipitation of KHCO3.40 The crystallographic structure of the resulting solid product was analyzed using XRD, as presented in Fig. 4b. The characteristic diffraction peaks align well with the reference pattern of KHCO3 (PDF#12-0292), confirming the successful carbonation reaction. After CO2 capture, the KHCO3 solution was fed into another electrochemical reactor for CO2 conversion. Consequently, a tandem system was established to produce a zero-carbon cement clinker precursor and formate (SI Fig. 8). The CO2-conversion reactor is a membrane-electrode-assembly reactor, which consists of a Bi2O2CO3 cathode for CO2 conversion to formate, an Ir/Ti felt anode for oxygen evolution, and a bipolar membrane for ion conduction. On the anode, KOH solution is fed and OH− is oxidized to oxygen, water, and electrons, as shown in eqn (4).| 4OH− → O2 + 2H2O + 4e− | (4) |
Within the bipolar membrane, water is dissociated into OH− and protons (eqn (5)).
| H2O → OH− + H+ | (5) |
The OH− and protons transport in an opposite direction to the anode and cathode, respectively, driven by the electric field. On the cathode, KHCO3 solution is supplied and it first reacts with protons from the bipolar membrane to produce local CO2. Then the locally produced CO2 is reduced to formate and OH− (eqn (6) and (7)).
| HCO3− + H+ → CO2 + 2H2O | (6) |
| 2CO2 + 2H2O + 4e− → 2HCOO− + 2OH− | (7) |
In parallel with Ca(OH)2 production, electrochemical CO2 conversion was investigated at various current densities. Fig. 4c illustrates the FE and voltage profiles for different current densities. Among the detected products, formate exhibited the highest FE, exceeding 60% from 50 mA cm−2 to 200 mA cm−2. The voltage increased moderately with increasing current density, consistent with the expected behavior of electrochemical processes. The high selectivity toward formate suggests that the Bi2O2CO3 catalyst effectively promotes the two-electron reduction pathway of CO2. To assess the long-term stability of the electrochemical CO2 conversion, the reactor was operated at a current density of 100 mA cm−2 over 100 h. Fig. 4d shows that the applied voltage remained stable at approximately 3.2 V, while the FE for formate production remained around 60%. This result indicates that the catalyst and electrode materials maintain their activity and selectivity over time, making them viable for continuous CO2 utilization. This suggests that the developed system not only mitigates CO2 emissions, but also generates valuable chemical products, contributing to a circular carbon economy.
Techno-economic analysis
To assess the economic feasibility of the proposed electrochemical process for simultaneous production of a zero-carbon cement clinker precursor and formate, a detailed cost analysis was conducted across different operating conditions. The results provide insights into cost distribution, sensitivity to key parameters, and potential cost optimization strategies for commercial implementation.The production costs of Ca(OH)2 and formate at varying current densities are presented in Fig. 5a. The cost of Ca(OH)2 production (left column) is primarily influenced by electricity consumption, followed by raw materials (clay and limestone), catalyst, and membrane. As current density increases, the cost per ton of cement shows a slight decrease due to improved process efficiency but increases at higher current densities due to rising energy requirements. The costs of capital expenditures (CAPEX) and fixed operational expenditures (OPEX) occupy a smaller ratio. For formate production (right column), the cost structure is similar, with electricity being the dominant contributor. The trend indicates that increasing current density initially reduces the production cost due to enhanced reaction rates but leads to diminishing returns at higher current densities due to increased voltage requirements and lower faradaic efficiencies. The integrated cost analysis of the coupled system is shown in Fig. 5b. The total production cost exhibits a U-shaped trend, where an optimal current density (100 mA cm−2) minimizes the cost by balancing energy consumption and production efficiency, reaching a total cost of 426 USD per t cement. At lower current densities, lower reaction rates result in higher fixed OPEX, while at excessive current densities, increased energy demand outweighs efficiency gains. This suggests that an intermediate current density is most economically viable for this coupled electrochemical process. When the profit of formate production is considered, the net cost of per ton cement can be obtained by deducting the profit from the total cost of per ton cement. As a result, the lowest net cost is 63 USD per t cement at 100 mA cm−2. Electricity price is a critical factor influencing production costs. By optimizing these parameters, the integrated process can achieve competitive costs while significantly reducing CO2 emissions. From an industrial perspective, adopting this technology could contribute to the decarbonization of cement production by replacing conventional lime production with an electrochemical alternative that simultaneously captures and utilizes CO2.
Fig. 5c illustrates the relationship between electricity cost, current density, and net production cost. The results indicate that at lower electricity prices, the electrochemical process becomes cost-competitive with conventional methods. However, at higher electricity prices, the cost advantage diminishes, highlighting the importance of sourcing low-cost renewable electricity to achieve economic viability. Our realized net cost of 63 USD per t cement is lower than the market price, indicating that our method is economically competitive. A sensitivity analysis of various operational and economic parameters on net cost is presented in Fig. 5d. EI and EII present the Ca(OH)2 and formate production, respectively. The sensitivity analysis for EI and EII is shown in SI Fig. 9. The cost of electricity has the most significant impact on total production cost, followed by the membrane electrode assembly (MEA) replacement frequency, FE, and electrolyzer lifetime. Voltage and electrolyzer capital costs also contribute substantially, suggesting that improving system efficiency and extending equipment lifetime are critical for cost reduction. The positive effect on CO2 emission reduction is evaluated. The conventional thermal method emits 1010 kg CO2 per t cement,28,41 where energy emission accounts for 500 kg CO2 per t cement and feedstock emission accounts for 510 kg CO2 per t cement. Energy emission is from the burning of fossil fuels to create high-temperature conditions, and feedstock emission is from the CaCO3 decomposition. The CO2 emission is reduced to 925 kg CO2 per t cement when the Ca(OH)2 is produced by an electrochemical method, and further to 904 kg CO2 per t cement when both the Ca(OH)2 and formate productions are based on an electrochemical method. If the emission effect of electricity can be reduced, the optimistic CO2 emission could be drastically reduced to 241 kg CO2 per t. While reducing carbon emissions, it also brings considerable carbon tax benefits compared to traditional thermal methods (SI Fig. 10).
Overall, this approach presents a viable pathway for zero-carbon cement clinker precursor production while simultaneously enabling CO2 valorization, offering a scalable and economically feasible solution to mitigate climate impacts in the cement and chemical industries. Future research could focus on optimizing electrode materials and architectures to enhance catalytic efficiency and durability, particularly under high current densities. Additionally, scaling up the tandem electrocatalysis system and evaluating its performance under industrially relevant conditions will be essential for its practical implementation in large-scale manufacturing. Key challenges to address include reducing material costs, refining reactor design, and extending operational stability to facilitate the transition from laboratory-scale research to commercial application. Furthermore, integrating this technology with renewable energy sources and conducting comprehensive environmental assessments will be crucial in establishing its potential for sustainable construction materials and carbon-neutral industrial processes.
Conclusions
This study developed a groundbreaking electrochemical tandem system that effectively addresses the dual challenges in cement production: fossil fuel dependence and process CO2 emissions. The system innovatively captures and directly converts limestone-derived CO2 into formate, while simultaneously utilizing electrochemically generated H2 to calcine Ca(OH)2 for zero-carbon cement production. Through optimized three-chamber flow reactor design and reaction conditions, the system achieves unprecedented 100-hour stable operation, overcoming the stability limitations of conventional electrochemical approaches. The techno-economic analysis demonstrated the system's dual benefits: eliminating traditional process carbon emissions while producing valuable formate at over 60% FE. This work not only provides a viable decarbonization solution for the cement industry, but also establishes a new paradigm for industrial electrification transforming emissions into useful products. The modular design exhibits excellent scalability potential, paving the way for large-scale implementation. Future research will focus on reactor scale-up and integration with renewable energy systems to accelerate commercial adoption.Materials and methods
Materials
Bismuth nitrate pentahydrate and ethylene glycol were purchased from Shanghai Macklin Co., Ltd. Calcium nitrate, potassium nitrate, potassium hydroxide, calcium carbonate, and diethanolamine were obtained from Shanghai Aladdin Co., Ltd. Ethanol, potassium bicarbonate and urea were purchased from Chengdu Chron Chemical Co., Ltd. Nafion dispersion (5 wt%) and carbon paper (HCP030N) were acquired from Shanghai Hesen Co., Ltd. The cation exchange membrane (Nafion N115), anion exchange membranes (Fumasep FAA-3-PK-75, Fumasep FAA-3-50, Sustainion X37-50 Grade T, and PiperION A40), and bipolar membrane (Fumasep FBM-PK) were purchased from SCI Materials Hub. The platinum-coated titanium felt (1 mg cm−2) and iridium-coated titanium felt (1 mg cm−2) were purchased from Kunshan Mingzhu Electronics Material Co., Ltd. Deionized water was supplied by a laboratory water purification system (Hitech, Smart-S15UVF).Electrode fabrication
In this work, we specifically exclusively fabricated the Bi2O2CO3 cathode for bicarbonate reduction to formate, while utilizing commercially available electrodes for all other components. Carbon paper was used as the cathode electrode substrate for electrolyzing bicarbonate. Bi2O2CO3 nanosheets, serving as the catalyst, were synthesized via a hydrothermal method.38 To prepare the catalyst ink, 20 mg of the synthesized catalyst was dispersed in 2 mL of ethanol solvent, followed by the addition of 21.05 mg of Nafion dispersion (5 wt%). The mixture was then ultrasonicated for 30 minutes to ensure a uniform dispersion. Thereafter, the ink was repeatedly sprayed onto the carbon paper and dried until the catalyst loading amount reached 1 mg cm−2.Characterization of electrodes
The morphological and microstructural features of the electrodes and synthesized products were examined using a scanning electron microscope (SEM, Hitachi SU8020). Crystalline phase identification was performed via powder X-ray diffraction (XRD, Bruker D8 ADVANCE) with Cu-Kα radiation. Surface elemental composition and chemical bonding states were assessed by X-ray photoelectron spectroscopy (XPS, Thermo Scientific ESCALAB 250Xi).Construction of the reaction system
For the electrochemical synthesis reactor of Ca(OH)2, the components are arranged from bottom to top in the order of an end plate, iridium-coated titanium felt, cation exchange membrane, middle chamber, anion exchange membrane, platinum-coated titanium felt, and another end plate, connected and secured by bolts. To ensure airtightness and prevent leakage, PTFE gaskets were installed between the end plates and the middle chamber. The anode chamber, middle chamber, and cathode chamber were supplied with 1 M Ca(NO3)2 (containing 30 g L−1 CaCO3), 1 M KNO3, and 1 M KOH, respectively, using peristaltic pumps. The flow rate in the middle chamber was maintained at 50 mL min−1. A constant current was applied using an electrochemical workstation (Gamry Interface 1010 E, USA), and the products were collected to quantify the FE. Gas products from the anode and cathode chambers were collected using gas bags, while solid products from the middle chamber reservoir were collected via vacuum filtration.For electrochemical reduction of bicarbonate, the assembly from top to bottom comprises an end plate, iridium-coated titanium felt, bipolar membrane, Bi2O2CO3 cathode, and another end plate. Similarly, gaskets were installed to prevent leakage. The anode and cathode chambers were supplied with 1 M KOH and 3 M KHCO3, respectively, using peristaltic pumps at a flow rate of 50 mL min−1. Before starting the experiment, the cathode chamber reservoir was purged with N2 to eliminate air interference. A constant current was applied using a DC regulated power supply (Tongmen eTM-L303SPD), and the gaseous products were collected using gas bags.
Production quantification
The produced gas was measured by gas chromatography (Shimadzu Nexis GC-2030) with a FID and TCD. FE was calculated using the following equation: | (8) |
The formate content in the catholyte was determined using an ion chromatograph (ICS-5000, ThermoFisher, USA) equipped with an anion self-regenerating suppressor (4 mm, ASRS 300) and an anion analytical column (4 × 250 mm, AS11-HC). The FE was calculated using eqn (9).
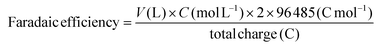 | (9) |
Techno-economic analysis
When conducting the techno-economic analysis, full consideration was given to capital expenditure (CAPEX), electricity cost, raw material cost, the replacement cost of catalysts and membranes, as well as fixed operational expenditures (OPEX) of the two electrochemical processes. The calculations were based on a production capacity of 3000 t day−1.CAPEX primarily comprises the cost of the main electrolyzer unit and the balance of plant (BOP) required to support the core electrochemical reactions.
The total electrolyzer cost can be calculated as follows:
 | (10) |
For EII, the unit area cost of the electrolyzer was 920 USD per m2,42 Considering that the EI requires an additional intermediate chamber made of POM, the single electrolyzer cost is set at 1200 USD per m2. And the capacity factor was 0.9. The actual operating area (S) of EI under the current production capacity can be calculated as:
 | (11) |
In this equation, n denotes the number of electrons transferred in the electrochemical process, and FE represents the faradaic efficiency for EI, calculated as 95%. Furthermore, based on eqn (2), the CO2 generated from limestone decomposition for a cement plant with a production capacity of 3000 tons is 1530 tons. Therefore, SII can be derived from eqn (12).
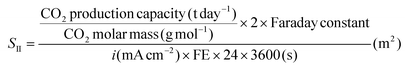 | (12) |
In this equation, the number 2 represents that the electrochemical synthesis of formate is a two-electron reaction. FE stands for faradaic efficiency, which is calculated based on the experimentally measured value.
The CAPEX was calculated using the following equation:
 | (13) |
The BOP cost, which includes the thermal management system, the fluid handling and control system, and the power conversion system, was estimated at 35% of the total CAPEX, and an electrolyzer lifetime of 20 years was assumed, which is a common value for other electrochemical processes.43
For both EI and EII, the electricity cost can be calculated as follows:
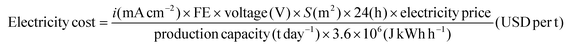 | (14) |
The respective operating voltage and faradaic efficiency, as obtained experimentally at the given current density, were employed for the calculations. The electricity price was based on the current market conditions, which was 0.05 USD per kWh.20
For the cement synthesis process, the raw materials are limestone (purity 0.95, 12 USD per t) and clay (SiO2 content 70%, 16 USD per t).41 For EII, the raw material cost is the carbon capture cost, which is USD 70 per tCO2.
The replacement costs of catalysts and membranes can be calculated using the following formulas:
 | (15) |
 | (16) |
The price of the catalysts is 28 USD per gram for platinum, 50 USD per gram for iridium,28 and the price for the self-made Bi2O2CO3 catalyst is calculated to be 10 USD per gram. The price of the membrane, referenced from PEM calculation processes, was set at 180 USD per m2.43
Operational expenditures (OPEX) primarily comprise installation costs and maintenance costs. The installation costs account for 35% of the CAPEX,44 while the annual maintenance costs amount to 3% of the CAPEX, which includes 1% for maintenance labor and 2% for spare parts replacement.41 The OPEX can be calculated using the following equation:
 | (17) |
For formate revenue calculation, the separation and purification cost was estimated as 60 USD per t,42 while the market price is 740 USD per t.45
Author contributions
X. X., Z. P., X. Z., R. C. and Q. L. conceived the project and designed the experiments. X. X., H. L., L. D., D. Y., Y. Y., H. W., X. Y. and X. Z. performed the experimental study. X. X., X. Z. and L. A. performed the TEA study. X. X., Z. P., X. Z., R. C. and Q. L. wrote the paper with support from all authors. Z. P., X. Z., R. C. and Q. L. supervised this project.Conflicts of interest
The authors declare no competing interests.Data availability
The data reported in this study are available from the corresponding author upon reasonable request. Supplementary information (SI) is available. Detailed electrode characterization, system photo images, and additional experimental and techno-economic analysis results are all included in the Supplementary Information (Fig. S1–S10). See DOI: https://doi.org/10.1039/d5gc05143f.Acknowledgements
The authors gratefully acknowledge the financial support of the National Natural Science Foundation of China (52161160333), the Joint Fund for Innovation and Development of Chongqing Natural Science Foundation (CSTB2022NSCQ-LZX0013), the Chongqing Talents (CSTB2024YCJH-KYXM0082), the Research Grants Council of the Hong Kong Special Administrative Region, China (N_PolyU559/21), and the Research Institute for Advanced Manufacturing at the Hong Kong Polytechnic University (CDJQ). Xiao Zhang acknowledges the support from the Research Grants Council of the Hong Kong Special Administrative Region, China (PolyU25213824), the National Natural Science Foundation of China (22205187), the Shenzhen Municipal Science and Technology Innovation Commission (JCYJ20230807140402006), and the Department of Science and Technology of Guangdong Province (2023A1515110123, 2024A1515012390).References
- G. Habert, S. A. Miller, V. M. John, J. L. Provis, A. Favier, A. Horvath and K. L. Scrivener, Nat. Rev. Earth Environ., 2020, 1, 559–573 CrossRef.
- J. Wei, K. Cen and Y. Geng, Environ. Sci. Pollut. Res., 2019, 26, 6409–6423 CrossRef CAS PubMed.
- O. E. Ige, D. V. Von Kallon and D. Desai, Clean Technol. Environ. Policy, 2024, 26, 579–597 CrossRef CAS.
- E. Benhelal, E. Shamsaei and M. I. Rashid, J. Environ. Sci., 2021, 104, 84–101 CrossRef CAS.
- R. M. Andrew, Earth Syst. Sci. Data, 2019, 11, 1675–1710 CrossRef.
- P. S. Fennell, S. J. Davis and A. Mohammed, Joule, 2021, 5, 1305–1311 CrossRef.
- C. Chen, R. Xu, D. Tong, X. Qin, J. Cheng, J. Liu, B. Zheng, L. Yan and Q. Zhang, Environ. Res. Lett., 2022, 17, 044007 CrossRef CAS.
- K. Guo, D. Liu, Y. Zhang, H. Feng and Q. Li, Chem. Eng. J., 2025, 519, 165155 CrossRef CAS.
- Y. Guo, L. Luo, T. Liu, L. Hao, Y. Li, P. Liu and T. Zhu, J. Environ. Sci., 2024, 136, 682–697 CrossRef PubMed.
- S. Q. Parra and M. C. Romano, J. Cleaner Prod., 2023, 425, 138913 CrossRef.
- Y. Yao, J. Lyu, X. Li, C. Chen, F. Verpoort, J. Wang, Z. Pan and Z. Kou, DeCarbon, 2024, 5, 100062 CrossRef.
- H. Feng, D. Liu, Y. Zhang, X. Shi, O. C. Esan, Q. Li, R. Chen and L. An, Adv. Energy Mater., 2022, 12, 2200469 CrossRef CAS.
- G. Habert, C. Billard, P. Rossi, C. Chen and N. Roussel, Cem. Concr. Res., 2010, 40, 820–826 CrossRef CAS.
- K. L. Scrivener, V. M. John and E. M. Gartner, Cem. Concr. Res., 2018, 114, 2–26 CrossRef CAS.
- K. Liu, X. Zhang, J. Li, Y. Liu, M. Wang and H. Cui, Green Chem., 2024, 26, 4677–4683 RSC.
- R. Maddalena, J. J. Roberts and A. Hamilton, J. Cleaner Prod., 2018, 186, 933–942 CrossRef CAS.
- K. Kukreja, M. K. Soni, B. Mohapatra and D. K. Panda, Sustainable Energy Technol. Assess., 2023, 57, 103300 CrossRef.
- X. Yu, W. Zhang, L. Ma, J. Tang, W. Lu, J. Li, J. Zhang and X. Xu, Green Chem., 2025, 27, 731–742 RSC.
- Z. Zhang, B. A. W. Mowbray, C. T. E. Parkyn, C. Waizenegger, A. S. R. Williams, E. W. Lees, S. Ren, Y. Kim, R. P. Jansonius and C. P. Berlinguette, Energy Environ. Sci., 2022, 15, 5129–5136 RSC.
- B. A. W. Mowbray, Z. B. Zhang, C. T. E. Parkyn and C. P. Berlinguette, ACS Energy Lett., 2023, 8, 1772–1778 CrossRef CAS.
- Z. B. Zhang, ACS Energy Lett., 2023, 8, 3927–3934 CrossRef CAS.
- L. D. Ellis, A. F. Badel, M. L. Chiang, R. J.-Y. Park and Y.-M. Chiang, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 12584–12591 CrossRef CAS PubMed.
- Q. Xie, L. Wan, Z. Zhang and J. Luo, iScience, 2023, 26, 106015 CrossRef CAS PubMed.
- N. P. Martínez, P. F. Troncoso, V. Gazzano, D. Ramírez-Amaya, M. González, I. Navarrete, R. Canales and P. Dreyse, J. Cleaner Prod., 2024, 468, 143112 CrossRef.
- Q. Chen, X. Wang, Y. Zhou, Y. Tan, H. Li, J. Fu and M. Liu, Adv. Mater., 2024, 36, 2303902 CrossRef CAS PubMed.
- D. Wu, F. Jiao and Q. Lu, ACS Catal., 2022, 12, 12993–13020 CrossRef CAS.
- Z.-Z. Niu, L.-P. Chi, R. Liu, Z. Chen and M.-R. Gao, Energy Environ. Sci., 2021, 14, 4169–4176 RSC.
- P. J. M. Monteiro, S. A. Miller and A. Horvath, Nat. Mater., 2017, 16, 698–699 CrossRef PubMed.
- S. Nie, J. Zhou, F. Yang, M. Lan, J. Li, Z. Zhang, Z. Chen, M. Xu, H. Li and J. G. Sanjayan, J. Cleaner Prod., 2022, 334, 130270 Search PubMed.
- A. Wang, G. Long, J. Chen, X. An, T. Yao and C. Li, Adv. Energy Mater., 2024, 14, 2400647 CrossRef CAS.
- W. Xia, M. Ma, Z. Li, L. Qiao, K. Chi, X. Guo, T. Liu, D. Wu, D. Cao and D. Cheng, Appl. Catal., B, 2024, 354, 124074 CrossRef CAS.
- L. Ma, Y. Wang, M. Zhang, J. Li, L. Zhang, X. Zhu, Q. Fu and Q. Liao, J. Power Sources, 2025, 641, 236874 CrossRef CAS.
- Y. Luo, J. Chen, N. Han and Y. Li, Acc. Mater. Res., 2025, 6, 462–472 CrossRef CAS.
- L. Tang, J. Ran, X. Huang, C. Ma, Y. Shao, J. Niu, H. Qiu and Y. Zhang, DeCarbon, 2025, 9, 100121 CrossRef.
- Z. Li, X. Wei, Z. Zhu, W. Jiang, Y. Hou, R. Yuan, Y. Wang, D. Xie, J. Wang, Y. Lin, R. Wu, Q. Kong and J. S. Chen, DeCarbon, 2025, 9, 100112 CrossRef.
- K. Liu, P. Lin, J. Li, Y. Liu, M. Wang, H. Cui and S. Yi, Adv. Funct. Mater., 2025, 35, 2424357 CrossRef CAS.
- Y. Sun, X. Liu, M. Zhu, Z. Zhang, Z. Chen, S. Wang, Z. Ji, H. Yang and X. Wang, DeCarbon, 2023, 2, 100018 CrossRef.
- Z. Wang, Xiaolong Zu, Xiaodong Li, L. Li, Y. Wu, S. Wang, P. Ling, Y. Zhao, Y. Sun and Y. Xie, Nano Res., 2022, 15, 6999–7007 CrossRef CAS.
- Y. Xiao, D. Liu, J. Yang, J. Feng, W. Gu, L. Qiao, W. F. Ip and H. Pan, Nano Lett., 2025, 25, 6548–6555 CrossRef CAS PubMed.
- M. Shen, L. Ji, D. Cheng, Z. Wang, Q. Xue, S. Feng, Y. Luo, S. Chen, J. Wang, H. Zheng, X. Wang, P. Sautet and J. Zhu, Joule, 2024, 8(7), 1999–2015 CrossRef CAS.
- R. K. Miao, N. Wang, S.-F. Hung, W.-Y. Huang, J. Zhang, Y. Zhao, P. Ou, S. Wang, J. P. Edwards, C. Tian, J. Han, Y. Xu, M. Fan, J. E. Huang, Y. C. Xiao, A. H. Ip, H. Liang, E. H. Sargent and D. Sinton, ACS Energy Lett., 2023, 8, 4694–4701 CrossRef CAS.
- P. Yue, Z. Kang, Q. Fu, J. Li, L. Zhang, X. Zhu and Q. Liao, Appl. Energy, 2021, 304, 117768 CrossRef CAS.
- H. Shin, K. U. Hansen and F. Jiao, Nat. Sustainability, 2021, 4, 911–919 CrossRef.
- F.-Y. Chen, A. Elgazzar, S. Pecaut, C. Qiu, Y. Feng, S. Ashokkumar, Z. Yu, C. Sellers, S. Hao, P. Zhu and H. Wang, Nat. Catal., 2024, 7, 1032–1043 CrossRef CAS.
- P. Zhu and H. Wang, Nat. Catal., 2021, 4, 943–951 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




