 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Combined effects of a low-dose multi-target supplement (CaHMB, CBP, and HA) on delaying musculoskeletal aging
Haoqi
Chen
a,
Junhong
Peng
a,
Shanshan
Guo
c,
Ting
Chen
a,
Si
Chen
a,
Xinyuan
Jin
a,
Wenge
Huang
d,
Chao
Xu
e,
Mengchu
Li
a,
Mengxing
Xie
a,
Mengtao
Yang
a,
Jinzhu
Pang
*bc and
Huilian
Zhu
 *ab
*ab
aDepartment of Nutrition, School of Public Health, Sun Yat-sen University, Guangzhou 510080, China. E-mail: zhuhl@mail.sysu.edu.cn
bSun Yat-sen University-Mengniu Joint Research Center of Nutrition and Health for Middle-Aged and Elderly, Sun Yat-sen University, Guangzhou 510080, China
cMengniu Institute of Nutrition Science, Global R&D Innovation Center, Inner Mongolia Mengniu Dairy (Group) Co., Ltd, Huhhot 01150, China. E-mail: pangjinzhu@mengniu.cn
dCenter of Experimental Animals, Sun Yat-sen University, Guangzhou 510080, China
eDepartment of Clinical Nutrition, Haikou Affiliated Hospital of Central South University Xiangya School of Medicine, No. 43 Renmin Avenue, Haikou 570208, China
First published on 31st March 2026
Abstract
Age-related musculoskeletal diseases underscore the importance of comprehensive dietary interventions. The individual benefits of calcium β-hydroxy-β-methylbutyrate (CaHMB), colostrum basic protein (CBP), and hyaluronic acid (HA) on the musculoskeletal system have been well documented; however, their combined effects remain unclear. This study aims to investigate the effects of combined supplementation with CaHMB, CBP, and HA on age-related musculoskeletal degeneration and to explore the underlying mechanisms, with a particular focus on the muscle–bone axis. Twelve-month-old male C57BL/6J mice received a 6-month dietary intervention. The combination (COM) group was supplemented with CaHMB, CBP, and HA at substantially reduced doses (37.5%, 20%, and 14%, respectively). The COM group showed higher lean mass (18%) and a greater muscle cross-sectional area (35%) compared to the OLD group, with significantly enhanced grip strength by 41% and exercise performances. The COM group presented less decline in whole-body bone mineral density (5%) than the OLD group. The bone microstructure was improved by increasing the bone volume fraction and trabecular thickness/number, while a decrease in trabecular separation was observed in the COM group. Musculoskeletal improvements either matched or surpassed the benefits of individual components. Mechanistically, interventions modulated the key mediators of the muscle–bone axis, significantly upregulating beneficial myokines (irisin and IGF-1) and osteokine osteocalcin, while downregulating negative regulators (myostatin and sclerostin). These changes were correlated with the observed phenotypic enhancements. The low-dose combination of CaHMB, CBP, and HA provides comprehensive benefits against age-related muscle and bone loss, likely by modulating the muscle–bone axis, and outperforms individual components. Our findings support its potential as a multi-targeted nutritional strategy for the aging population.
1. Introduction
The global population is undergoing irreversible aging, with the process exhibiting an accelerating trend. According to the World Health Organization (WHO), in 2020, there were about 1 billion people aged 60 or older worldwide, accounting for 13.5% of the global population (7.8 billion). This figure is expected to rise to nearly 2.1 billion by 2050.1 The health of the aging population has become a significant public health concern in the world. Aging involves progressive cellular degeneration driven by accumulated inflammation, oxidative stress, and apoptosis,2,3 which leads to a series of physiological declines, including impaired nutrient metabolism that increases malnutrition risk, causes deleterious changes in body composition, and results in the functional deterioration of key systems such as bone, muscle, joint, cognition, and immunity.4–6 Therefore, the elderly face a high burden of chronic diseases and prolonged disability, frequently leading to multimorbidity.7 Among these comorbid conditions, age-related musculoskeletal diseases, such as sarcopenia, osteoporosis and osteoarthritis, are frequently observed together.6Sarcopenia, characterized by the progressive loss of muscle mass and strength with aging,8 has a reported global prevalence ranging between 10% and 27%.9 Osteoporosis is defined by low bone mineral density (BMD) and deterioration of bone microstructure,10 with an estimated worldwide prevalence of 19.7%.11 Osteoarthritis is a progressive and degenerative joint disease characterized by articular cartilage degeneration, bone remodelling and osteophyte formation.12 The global prevalence of osteoarthritis is approximately 7.6% and knee osteoarthritis, as the most common form, exhibits a prevalence of about 43% in individuals aged 40 years and older.13 The musculoskeletal system reaches its peak function around age 30. After that, bone and muscle gradually decline, with this loss accelerating rapidly after age 45–50.14–16 Sarcopenia, osteoporosis and osteoarthritis are independently and synergistically associated with an increased risk of adverse health outcomes, including frailty, fractures, mobility disability, and mortality.6,13 Beyond the well-known biomechanical, neuroendocrine, and nutritional interactions,17 the musculoskeletal system also engages in molecular crosstalk through the secretion and reception of various cytokines, a network of interactions collectively termed the “muscle–bone axis”.18 Myokines, secreted by skeletal muscles, such as irisin,19 insulin like growth factor-1 (IGF-1)20 and myostatin (MSTN)21 can positively or negatively regulate bone health through autocrine, paracrine, and endocrine modes.22 Conversely, bone-derived osteokines like osteocalcin (OCN)23 and sclerostin (SOST)24 signal back to influence muscle mass and function.22 Accordingly, given the inseparable link between muscle, bone and joint, integrated management of sarcopenia, osteoporosis and osteoarthritis is imperative for the elderly.
β-Hydroxy β-methylbutyrate (HMB) is a natural metabolite of the essential amino acid leucine.25 Its commonly supplemented form is calcium HMB (CaHMB), the calcium salt of HMB. Extensive research, spanning from animal models to human clinical trials, has established the potential of HMB to positively modulate muscle mass and function.25–27 The primary mechanism of HMB involves stimulating muscle protein synthesis and inhibiting degradation.28,29 Colostrum basic protein (CBP) is a small bioactive protein extracted from colostrum, with a molecular weight ranging from 1 to 30 kDa.30 Although clinical evidence of CBP in humans remains limited, several animal experiments have shown that CBP could promote bone growth and increase bone strength through the direct regulation of osteoblast and osteoclast activity.30,31 Hyaluronic acid (HA), also known as hyaluronan, is a high-molecular-weight linear glycosaminoglycan. It is naturally and widely distributed throughout the human body, including in joints, skin, and the vitreous body of the eye.32 Oral HA supplementation has been widely studied for its potential benefits in multiple areas, including joint, skin and gastrointestinal health.33–36 Specifically, its role in osteoarthritis management is notable for enhancing muscle strength, reducing pain and stiffness, and modulating inflammation.37–39 Based on the broad beneficial effects of CaHMB, CBP, and HA on the musculoskeletal system, they are currently approved for use in a variety of food categories in many countries, primarily including sports nutrition foods, dairy products and foods for special medical purposes.40–43
Currently, most of the interventions primarily target sarcopenia, osteoporosis and osteoarthritis in isolation, often overlooking the critical interconnection within the musculoskeletal system. Moreover, despite the well-established effects of the individual administration of CaHMB, CBP, and HA on muscle, bone and joint, their combined effects remain largely unexplored. An important and unexplored aspect of this gap is a low-dose combination, formulated from human food regulation limits, that would represent a critical translational step to bridge nutritional science and practical application.
Therefore, this study aimed to evaluate the efficacy of a combined CaHMB, CBP, and HA supplement on delaying degenerative changes in muscle, bone, and joints as mice naturally progress to early old age and to explore the underlying molecular mechanisms involving the muscle–bone axis.
2. Materials and methods
2.1. Animals and experimental design
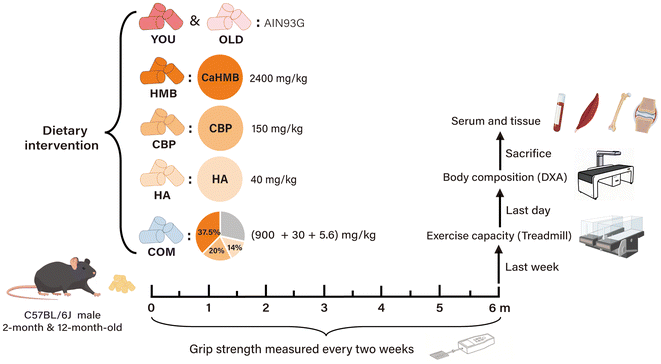
Male C57BL/6J mice aged 2 or 12-months were provided by Vital River Laboratory Animal Technology Co., Ltd (Beijing, China) and were housed in a pathogen-free facility (12 h day/night cycle, 20–26 °C, relative air humidity 40–60%) with ad libitum access to food and water. A 6-month dietary intervention was administered. 2-month-old mice (the young control group, YOU) received no intervention and were fed the standard AIN93G diet (Jiangsu Medicine, China) throughout the study, reaching 8-months of age at the end of the experiment. 12-month-old mice were randomly assigned to five groups and received the following dietary interventions for 6-months until euthanasia at 18-months of age: (1) the old control group (OLD), fed with the standard AIN93G diet; (2) the CaHMB group (HMB), fed with AIN93G containing 2400 mg kg−1 CaHMB;44 (3) the CBP group (CBP), fed with AIN93G containing 150 mg kg−1 CBP;45 (4) the HA group (HA), fed with AIN93G containing 40 mg kg−1 HA;35 and (5) the combination group (COM), fed with AIN93G supplemented with 900 mg kg−1 CaHMB, 30 mg kg−1 CBP, and 5.6 mg kg−1 HA. CaHMB was provided by Jiangyin Tsi Pharmaceutical Co., Ltd (Jiangsu, China), CBP was obtained from Seperex Nutritionals® Ltd (Otago, New Zealand), and HA was obtained from Shandong Bloomage Hyinc Biopharm Co. Ltd (Shandong, China).The dose in the COM group was set at the animal-feed equivalent of the highest level permitted in human food by relevant Chinese national regulations.41–43 Specifically, the regulations stipulate the following maximum addition limits in dairy products: CaHMB ≤ 3 g per 100 g, CBP ≤ 100 mg per 100 g, and HA ≤ 20 mg per 100 g. Therefore, taking a recommended daily human intake of 50 g of milk powder as an example, this would provide 1.5 g of CaHMB. The daily intake of humans is thus 1.5 g per 60 kg body weight (BW). According to the Meeh–Rubner formula, k = 9.1 for dose translation from a human to a mouse (based on the body surface area), and the equivalent mouse dose was calculated to be 225 mg per kg BW. This dose was subsequently converted to the dietary concentration based on the average mouse BW and food consumption. The doses for the three single-intervention groups were determined by referencing efficacious doses reported in the previous literature.35,44,45
The BW and food intake were recorded weekly. After 6-months of intervention, the mice were fasted for 10–12 h, anesthetized with pentobarbital sodium, and blood samples were collected. Serum was obtained by centrifugation at 1000g for 15 min and stored at −80 °C until analysis. The tibias, femurs, knee joints and hind limb muscles of all mice were dissected. The entire hind limb bones and 5 mm3 of quadriceps muscle (QA) samples were selected and fixed in 4% paraformaldehyde (PFA, Servicebio, China) for 24 h. All animal protocols were approved by the ethics committee of the School of Public Health, Sun Yat-sen University (permit no. 2024-066). Experimental design and group assignment are presented in Fig. 1.
2.2. Body composition measurement
At the end of the dietary intervention, body composition was examined. The mice were anesthetized by pentobarbital sodium and placed in a prone position on a dual-energy X-ray absorptiometer (DXA, Discovery W; Hologic Inc., USA). The mode of “small animal” was applied to record the body composition, including lean mass, fat mass and whole-body bone mineral density (BMD).2.3. Behaviour experiment
The grip strength was measured using a grasp meter (DS2-50N, China) every two weeks during the intervention. In brief, after acclimation, the mice were placed on a metal grid with all four limbs, and then their tails were pulled horizontally with a steadily increasing force until the mice released the grip. The peak force was recorded. The measurement was repeated four times per mouse, and the result was calculated as the average of the two highest values.The exercise capacity of mice was evaluated using a treadmill (XR-PT-10B, Shanghai Xinrun Information Technology Co., Ltd, China) after 6-month intervention. Before the formal test, the mice underwent a 3-day acclimation. The formal test consisted of a 15-minute run, starting at 15 m min−1 and increasing to 25 m min−1 over 2 min. The current intensity for electrical stimulation was set as 0.5 mA. The distance and the time of the running test were recorded.
2.4. Micro-CT
The left femur and tibia samples (N = 5 per group) collected from the mice were scanned by micro-CT (VNC-102, PINGSENG Healthcare (Kunshan) Inc., China), and the resulting images were reconstructed using Recon software. A standardized region of interest (ROI) 2 mm beneath the femoral and tibial growth plates was defined and analysed uniformly across all samples using Avatar software. The analysis covered the bone volume (BV/TV) fraction, BMD of femur and tibia, trabecular bone thickness (Tb·Th), trabecular bone separation (Tb·Sp), and trabecular bone number (Tb·N).2.5. Histological analysis
The right QA samples (N = 8 per group), right femur and tibia samples (N = 5 per group) and right knee joint (N = 5 per group) were harvested. Briefly, 5 mm3 QAs were fixed with 4% PFA for 24 h, paraffin-embedded, and sectioned at 4 μm for hematoxylin and eosin (H&E) staining (Servicebio, China). Four random fields from each skeletal muscle cross-section were photographed under an optical microscope equipped with a digital camera (ECLIPSE Ci-L, Nikon, Tokyo, Japan). The cross-sectional area (CSA) was evaluated using ImageJ 2.16.0 software.For the femur and tibia samples, following 24 h fixation in 4% PFA, the bone tissues were decalcified in 0.5 M EDTA (Servicebio, China) and paraffin-embedded. Tissues were sectioned at 8 μm and stained with H&E and tartrate-resistant acid phosphatase (TRAP) (Servicebio, China). To quantify osteoclasts, three random fields were analysed per sample using ImageJ to determine the osteoclast number per unit bone surface (OC·N/BS) and the osteoclast surface per unit bone surface (OC·S/BS).
Knee joints were processed identically to bone samples (fixation, decalcification, and embedding). The samples were sliced into 8-μm-thick sections and subjected to H&E and Safranin O and Fast Green staining (Servicebio, China). The medial tibial cartilage of the knee was examined microscopically, photographed, and scored using the Osteoarthritis Research Society International (OARSI) system for cartilage degeneration.46,47
2.6. ELISA
The levels of OCN (SEKM-0301, Solarbio, Beijing, China), bone-specific alkaline phosphatase (BALP) (ELK2689, ELK Biotechnology, Wuhan, China) and C-telopeptide of type I collagen (CTX-I) (ELK2223, ELK Biotechnology, Wuhan, China) in the serum, irisin (PI470, Beyotime, China), MSTN (SEKM-0158, Solarbio, Beijing, China) and IGF-1 (EK0378, BOSTER, Wuhan, China) in the QA tissues and SOST (EK1179, BOSTER, Wuhan, China) in the femur tissues were measured using respective ELISA kits according to the manufacturers’ instructions. All standard curves had an R2 > 0.99.2.7. Statistical analysis
The data were expressed as the mean ± standard deviation (SD). Statistical analysis was performed using SPSS 27.0 statistical software (IBM Corp., Armonk, New York, USA). Student's t test and one-way analysis of variance (ANOVA), followed by least-significant difference (LSD) and Spearman correlation analysis were used in this study. Adobe Illustrator 29.6 and GraphPad Prism 10.0 were used to draw the graphs. The p-value < 0.05 was considered statistically significant.3. Results
3.1. Muscle and bone loss in aged mice
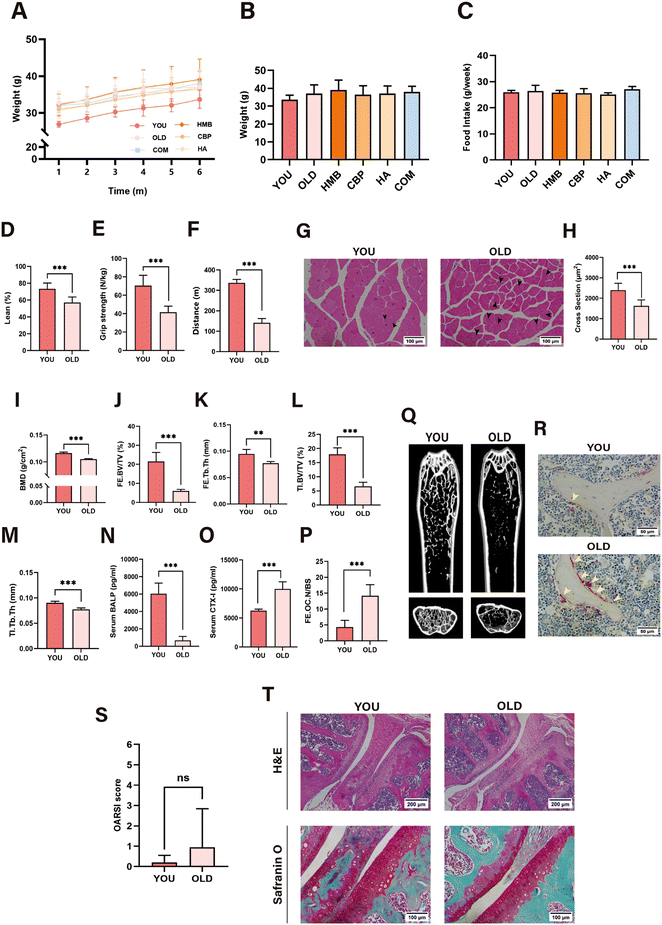
The BW increased progressively in all groups during the intervention period (Fig. 2A), with no significant differences observed among groups at the endpoint (Fig. 2B). Average weekly food intake did not differ among groups, which was 25.9 g per mouse per week throughout the intervention (Fig. 2C).Marked musculoskeletal deterioration was observed in the OLD group. Compared to the YOU group, the OLD group exhibited a significant decrease in the lean mass percentage (LM%) and muscle fibre CSA (Fig. 2D and H). Muscle function was also significantly impaired in the OLD group including the decrease of grip strength and the decrease of running distance and the time on the treadmill (Fig. 2E, F and Fig. S1A). Moreover, the whole-body BMD, BV/TV%, Tb·Th, and Tb·N of lower limb bones significantly reduced and the Tb·Sp significantly increased in the OLD group (Fig. 2I–M and Fig. S1B–D and F–H). Three-dimensional (3D) reconstruction and H&E staining further confirmed substantial trabecular bone loss in the aged mice (Fig. 2Q and Fig. S1E, I and J). Enhanced bone resorption in the OLD group was supported by the elevated CTX-I level and increased osteoclast numbers (Fig. 2O, P, and R and Fig. S1K and J–M), along with a decrease in the BALP level (Fig. 2N).
Notably, no overt knee osteoarthritis-related pathological changes were observed in the aged mice. In the OLD group, both the medial tibial plateau (MTP) and the medial femoral condyle (MFC) exhibited smooth articular surfaces without observable cartilage defects or significant synovitis-related changes, and the cartilage showed continuous red in Safranin O staining, similar to the YOU group (Fig. 2T). Furthermore, no significant differences were found between the OLD and YOU groups in the OARSI scores or MTP thickness (Fig. 2S and Fig. S1N).
Collectively, the naturally aged mice (18-months old after intervention) developed pronounced age-related muscle and bone loss accompanied by a functional decline, whereas spontaneous degenerative changes in the knee joint were not observed.
3.2. Intervention preserves muscle mass and function in aged mice
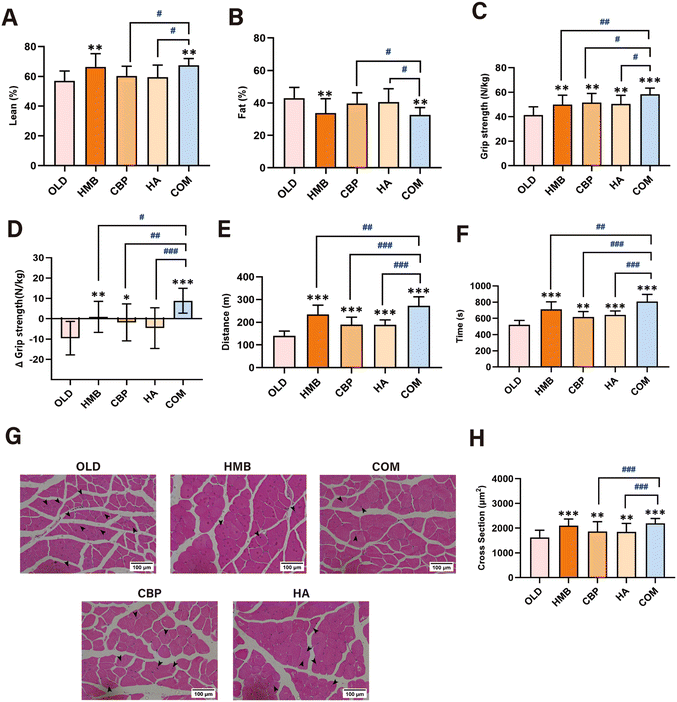
Following the 6-month intervention, the body composition was assessed by DXA. The HMB and COM groups effectively maintained LM% (COM vs. OLD: 67.4 ± 4.6% vs. 57.0 ± 6.6%, p = 0.002) and decreased FM% (COM vs. OLD: 32.6 ± 4.6% vs. 43.0 ± 6.6%, p = 0.002), and the efficacy of the COM group was significantly superior to both the CBP and HA groups (p < 0.05) (Fig. 3A and B). To further assess the muscle morphology, right QAs were subjected to H&E staining. All intervention groups prevented the decline of muscle fibre CSA in the aged mice. Specifically, the COM group showed the highest increase (1.35-fold), followed by the HMB group (1.29-fold), the CBP group (1.15-fold), and the HA group (1.14-fold). The COM group matched the effect of the HMB group (p = 0.213) and significantly exceeded the CBP and HA groups (p < 0.001) in preserving the muscle fibre CSA (Fig. 3G and H). A higher prevalence of nucleus centralization (NC) was observed in the OLD group, and both the COM and HMB groups could reduce the prevalence of NC, despite no significant difference between the two groups (Fig. 3G).Beyond improving the muscle mass, the intervention also exerted beneficial effects on muscle function, including grip strength and exercise performance. The aged mice in all intervention groups not only showed improved grip strength (Fig. 3C and D) but also exhibited extended running distance and time (Fig. 3E and F). Furthermore, these improvements in the COM group were superior to the three individual component groups.
In summary, our findings demonstrated that both CaHMB alone and the combination effectively preserved muscle mass and all interventions enhanced muscle function in aged mice. Moreover, the combination significantly outperformed both CBP and HA alone.
3.3. Intervention mitigates bone loss by improving bone metabolism in aged mice
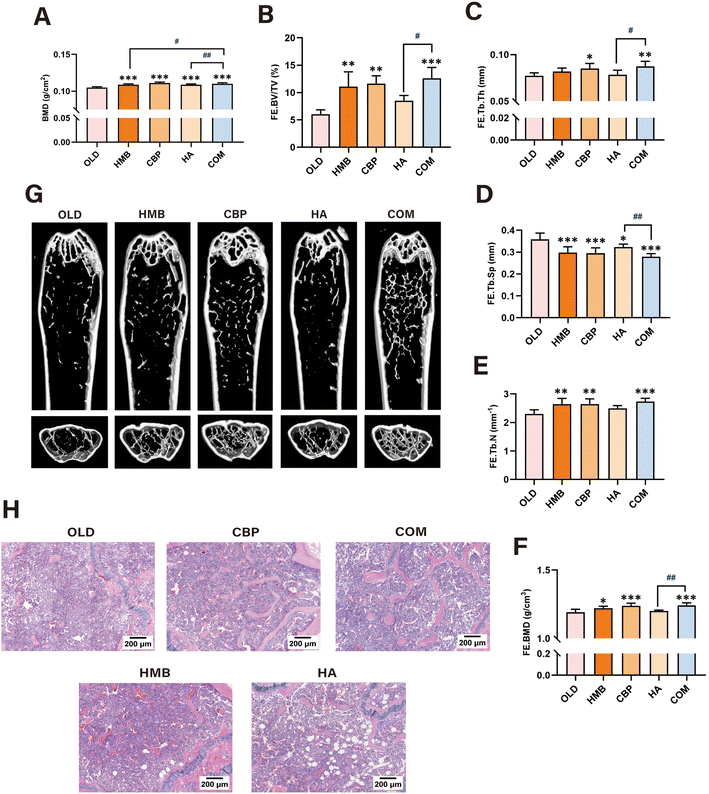
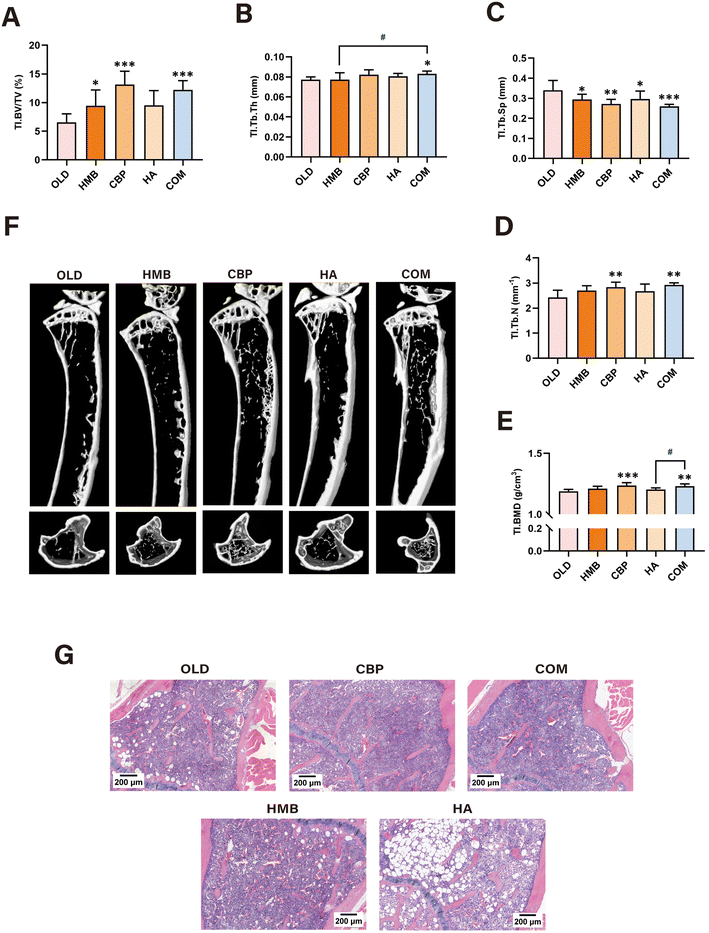
DXA analysis revealed all interventions preserved the whole-body BMD among groups, with the COM group showing the most significant improvement (COM vs. OLD: 0.1101 ± 0.0013 vs. 0.1049 ± 0.0011 g cm−2, p < 0.001). Notably, while the efficacy of the COM group was comparable to the CBP group (p = 0.096), it was significantly superior to both the HMB and HA groups (p < 0.05) (Fig. 4A).Micro-CT analysis of the femoral microstructure indicated that both CBP and COM groups significantly improved the femur (FE) microstructure in the aged animals, increasing the BV/TV% (COM: +109% and CBP: +93%), FE·BMD (COM: +4% and CBP: +4%), Tb·Th (COM: +13% and CBP: +10%), and Tb·N (COM: +19% and CBP: +15%) and decreasing Tb·Sp (COM: −22% and CBP: −18%), even though no statistically significant differences were found between the two groups across all parameters. What's more, apart from the Tb·N, COM group demonstrated significantly greater efficacy than the HA group in all parameters (Fig. 4B–G). 3D reconstruction images and H&E staining of FE showed age-related bone loss in the trabecular bone area, which was mitigated in both the CBP and COM groups (Fig. 4G and H).
Similarly, analysis of the tibial microstructure by micro-CT indicated that the CBP and COM groups markedly ameliorated tibia (TI) degeneration, with an increase in BV/TV% (COM: +87% and CBP: +100%), TI·BMD (COM: +3% and CBP: +4%), Tb·Th (COM: +7% and CBP: +6%), and Tb·N (COM: +21% and CBP: +17%) and a decrease in Tb·Sp (COM: −24% and CBP: −20%); however, the effectiveness of the two groups was statistically comparable. Additionally, the COM group outperformed the HA group in improving the TI·BMD and also showed a better effect than the HMB group in increasing Tb·Th (Fig. 5A–F). Similarly, tibial 3D reconstruction images and H&E staining demonstrated that both CBP and COM interventions attenuated the age-related loss of trabecular bone (Fig. 5F and G).
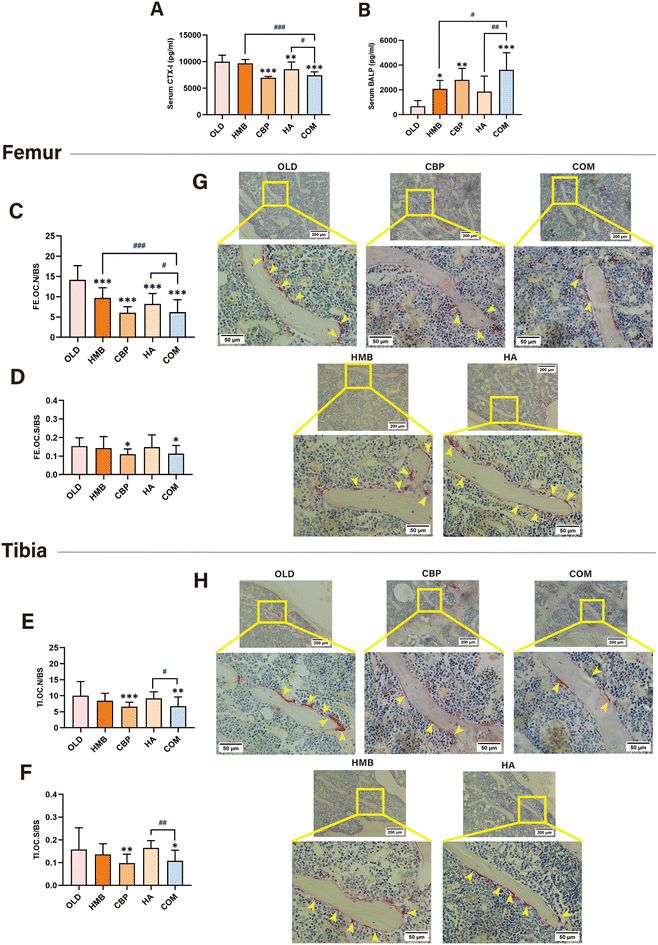
For the assessment of bone metabolism, we measured the serum levels of the bone turnover markers CTX-I and BALP and performed TRAP staining. Both COM and CBP groups reduced the CTX-I levels (COM: 7435 ± 627.9 and CBP: 6948 ± 266.5 pg ml−1) and increased the BALP levels (COM: 3607 ± 1389 and CBP: 2811 ± 927.1 pg ml−1) in aged mice, with no statistically significant difference observed between the two groups. However, the effect of the COM group was significantly superior to both the HMB and HA groups (Fig. 6A and B). TRAP staining and the following quantitative analysis demonstrated that COM and CBP were similarly effective in suppressing the overall osteoclast activity, reducing both OC·N/BS and OC·S/BS in the FE and TI (Fig. 6C–H). Moreover, COM was significantly more potent than the HMB and HA groups in inhibiting the OC·N/BS of FE (Fig. 6C) and surpassed the HA group in both the OC·N/BS and OC·S/BS of TI (Fig. 6E and F).
In conclusion, our results indicate that CBP alone and the combination attenuated bone loss by reducing bone resorption and improving bone metabolic homeostasis, thereby preserving bone mass and microstructure. Furthermore, the combination resulted in significantly greater improvements than either HMB or HA alone.
3.4. Molecular regulation of the muscle–bone axis
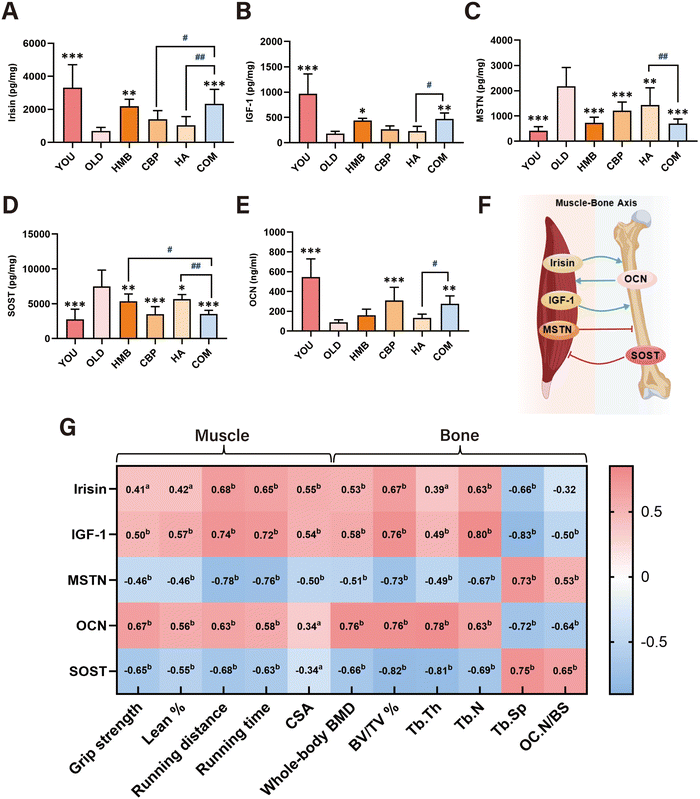
Given the superior musculoskeletal benefits observed with the combination treatment at relatively lower doses, we next investigated potential molecular mechanisms underlying its effects by examining key myokines and osteokines involved in muscle–bone crosstalk.Among myokines, positive factors such as irisin and IGF-1 promote bone formation and growth.20,48 In contrast, MSTN acts as a negative regulator, not only inhibiting muscle mass but also potentially accelerating bone loss21 (Fig. 7F). Our results showed that the levels of irisin and IGF-1 were significantly decreased, whereas the level of MSTN was significantly increased in the OLD group (Fig. 7A–C). Treatment with COM or HMB significantly elevated the levels of irisin (COM: 2338 ± 874.4 and HMB: 2190 ± 421.4 pg ml−1) and IGF-1 (COM: 469.8 ± 118.3 and HMB: 439.4 ± 42.4 pg ml−1) and suppressed the level of MSTN (COM: 696.2 ± 186.0, HMB: 722.9 ± 229.2 pg ml−1), with no significant difference between them. However, the COM group had significantly higher irisin levels than both CBP and HA groups and showed greater efficacy than the HA group in modulating IGF-1 and down-regulating MSTN (Fig. 7A–C).
Regarding osteokines, OCN enhances muscle regeneration and performance,23 while SOST not only inhibits osteoblast activity in bone but also impairs muscle function24 (Fig. 7F). The results revealed a significantly lower OCN level and a significantly higher SOST level in the OLD group (Fig. 7D and E). The COM and CBP groups comparably increased the OCN level (COM: 275.9 ± 79.4 and CBP: 307.8 ± 134.2 ng ml−1) and decreased the SOST level (COM: 3515 ± 549.7 and CBP: 3479 ± 1100 pg ml−1). COM outperformed the HMB and HA groups in lowering the SOST level and showed a stronger effect than the HA group in increasing the OCN level (Fig. 7D and E).
Spearman correlation analysis revealed that irisin, IGF-1 and OCN levels were positively correlated with muscle strength, lean mass, exercise performance, and bone microarchitectural parameters, while MSTN and SOST exhibited inverse correlations (Fig. 7G). These findings support the involvement of key myokines and osteokines in regulating musculoskeletal benefits through the muscle–bone axis.
Collectively, these results suggest that the combination of CaHMB, CBP and HA exerted the enhanced effects through an indirect mechanism: by concurrently boosting muscle and bone health and performance, the combination modulated the bidirectional signalling of the muscle–bone axis, leading to benefits beyond individual components.
4. Discussion
In this study, 12-month-old mice received a 6-month dietary intervention and were evaluated at 18-months of age (approximately 55–65 human years). This model allowed us to explore whether a long-term intervention across the middle and early aged status could exert effects on delaying the age-related musculoskeletal decline. We found that while individual components (CaHMB, CBP, and HA) conferred selective benefits on either muscle or bone, their low-dose combination produced broader musculoskeletal improvements, preserving both muscle mass and bone microstructure. Notably, these enhanced effects were potentially mediated by the modulation of key molecules of the muscle–bone axis.The early aged mice in this model showed clear degenerative losses in the muscle and bone but did not develop significant histological alterations in knee osteoarthritis under our experimental conditions. This outcome contrasts with some reports of high spontaneous osteoarthritis incidence in aged C57BL/6J mice (up to 80% at the age of 18-months).49,50 This might be attributed to factors including housing conditions, physical activity levels, genetic/epigenetic backgrounds, or inherent individual resistance to osteoarthritis. Furthermore, this natural aging model has not been widely standardized or adopted, and most studies in the field have relied on surgical or chemically induced osteoarthritis models or spontaneous models with specific genotypes to ensure more predictable and severe joint pathology.51–53 Additionally, aging is highly heterogeneous, which differentially impacts the progression of degenerative changes across tissues, and the pace of aging varies across muscle, bone, and joint.13,54,55 Therefore, it is possible that in our model, significant aging-related deterioration had already been observed in muscle and bone, while the knee joint remained in a relatively preserved state, highlighting a potential heterogeneity in tissue aging.
Consistent with the existing literature, the individual components in our study demonstrated tissue-specific benefits. The HMB groups showed improvements in muscle mass and function in aged animals (Fig. 3), aligning with prior evidence that CaHMB enhances muscle health in aged or sarcopenia-model animals and is supported by human randomized controlled trials (RCT) in the elderly.56–58 These effects were largely attributed to CaHMB's ability to stimulate muscle protein synthesis (e.g., via the mammalian target of the rapamycin/ribosomal protein S6 kinase (mTOR/p70S6K1) pathway),59,60 inhibit protein degradation (e.g., the suppression of the ubiquitin–proteasome pathway),61–64 and promote myocyte proliferation.65 On the other hand, the CBP group effectively maintained bone mass and improved bone metabolism in aged mice (Fig. 4–6), consistent with previous reports that CBP promotes bone health, likely through stimulating osteoblast activity30,31 and modulating calcium metabolism.45 However, research on CBP remains relatively limited, especially human intervention studies. These findings reinforce the targeted, yet limited, efficacy of single-nutrient approaches.
Notably, existing evidence points to the modest efficacy of single interventions in comprehensively addressing musculoskeletal aging, as each component primarily benefits a specific tissue, and findings across human studies were often inconsistent.66–68 In contrast, multi-target nutritional strategies have shown more promising results.69,70 A growing number of RCTs indicate that a range of different supplement strategies, employing various combinations of protein (e.g., whey), leucine or CaHMB, vitamin D, calcium, chondroitin sulfate, glucosamine, polyunsaturated fatty acids and other micronutrients, synergistically promote musculoskeletal health in older adults.71–77 Our study aligns with and extends this concept: while CaHMB and CBP exerted protective effects on muscle and bone, respectively, the low-dose combination achieved comparable or superior benefits across both tissues, despite containing each component at a substantially reduced dose (37.5%, 20%, and 14% of their respective monotherapy levels for CaHMB, CBP, and HA) (Fig. 3–6). This can be explained by a multi-target strategy, in which the combination simultaneously addresses the musculoskeletal system: CaHMB supports muscle health,26 CBP enhances bone metabolism,45 and HA contributes to joint and inflammatory modulation.78 This approach more closely reflects the physiological interconnectivity of the musculoskeletal system as a functional unit. This combined effect is probably mediated through the modulation of key myokines and osteokines of the muscle–bone axis.
As established in previous research, aging reduces the irisin, IGF-1, and OCN levels,79,80 while exercise can stimulate their expression and suppress the levels of MSTN and SOST.66,81 Consistent with this pattern, our ELISA assay results confirmed that aged mice exhibited significantly decreased levels of irisin, IGF-1, and OCN, alongside increased levels of MSTN and SOST, which was reversed by COM intervention (Fig. 7A–E). Notably, our correlation analysis further linked these COM-induced changes in myokines and osteokines to the specific improvements in muscle mass, function, and bone microstructure (Fig. 7G), consistent with findings from observational human studies.82–86 This association further supports the role of muscle–bone crosstalk in mediating the efficacy of the interventions.
On the one hand, the COM group significantly elevated the levels of beneficial myokines irisin and IGF-1 while suppressing the level of the negative regulator MSTN (Fig. 7A–C). As reported in the literature, irisin promotes bone formation by stimulating osteoblast activity through pathways such as Wnt/β-catenin,87 mitogen-activated protein kinase/extracellular signal-regulated protein kinase (MAPK/ERK),88 and adenosine monophosphate protein kinase (AMPK),89 while also inhibiting osteoclast formation through the receptor activator of the nuclear factor-κB ligand (RANKL)/the nuclear factor of the activated T cell (NFAT) c1 pathway.48,90 IGF-1 enhances osteoblast survival, proliferation, and differentiation,20,91,92 primarily via the Ras/MAPK and phosphoinositide 3′-kinase (PI3K)/Akt pathways.20 Conversely, MSTN inhibits muscle growth and promotes bone resorption93,94 by suppressing Wnt/β-catenin signaling95 and enhancing RANKL-mediated osteoclast activity.21,96 Beyond the myokines we discussed in this study, other muscle-derived factors like interleukin-6 (IL-6), fibroblast growth factor21 (FGF21), β-aminoisobutyric acid (BAIBA) and so on also played important roles in bone homeostasis.97–99 On the other hand, COM increased the level of osteokine OCN and decreased the level of SOST (Fig. 7D and E). OCN contributes to bone mineralization,100 regulates muscle function101 and promotes muscle cell proliferation and differentiation via p38MAPK and PI3K/Akt signaling,23 whereas SOST negatively regulates bone formation102 and might impair muscle mass82,103,104 and regeneration by inhibiting Wnt/β-catenin signaling.105 Osteokines such as transforming growth factor-β (TGF-β), FGF23, and RANKL are also important regulators of muscle metabolism and function.106–108
In addition to classical signalling molecules, extracellular vesicles (EVs) have emerged as a crucial communication medium in the muscle–bone axis,109,110 carrying bioactive cargos—including microRNAs (miRNAs)—that mediate efficient bidirectional crosstalk between muscle and bone.111 For instance, muscle-derived miR-27a-3p enhances osteoblast differentiation by activating β-catenin signaling,112 while miR-34a, which increases with age, accelerates bone marrow stem cell (BMSC) senescence by suppressing sirtuin 1 (SIRT1).113 Similarly, bone tissue also expresses a variety of miRNAs. Aged BMSCs downregulate miR-24,111 which is a promoter of myogenic differentiation,114 and upregulate miR-15b and miR-99b,111 which inhibit muscle cell differentiation115 and regulate muscle mass via the mTOR/Akt pathway.116Thus, future studies could further explore whether the combination supplement influences EV-mediated communication, particularly through the regulation of specific miRNAs involved in muscle–bone crosstalk.
While the existing literature predominantly focuses on single components or isolated musculoskeletal systems, our study provides the first systematic evaluation of the combined intervention of CaHMB, CBP, and HA. Our findings demonstrate that this combination has more comprehensive and multiple musculoskeletal system benefits at lower doses, potentially through regulating key signalling molecules (e.g., irisin, IGF-1, MSTN, OCN, and SOST) of the muscle–bone axis. Therefore, the low-dose, multi-target supplement presents enhanced potential for real-world generalization, providing direct evidence to support this nutritional strategy. However, several limitations of this study should also be acknowledged: (1) the study used only male mice. Given the crucial role of estrogen in maintaining bone density and muscle mass, and the higher prevalence of osteoporosis and sarcopenia in postmenopausal women, our results may not be directly generalizable to females. (2) The naturally aged model did not develop significant spontaneous osteoarthritis pathology in the knee joint, limiting the assessment of the intervention's effects on joint degeneration. (3) The experimental design included only the three single-component groups and the full combination group, without pairwise combination groups (e.g., CaHMB + CBP, CaHMB + HA, and CBP + HA). This limits our ability to identify which specific binary interactions might be driving the observed combined effects; therefore, a more precise mechanistic interpretation and optimal formulation refinement might be overlooked. (4) Our mechanistic investigation is correlative and lacks depth. The exploration of the muscle–bone axis was limited to a few proteins via ELISA and we did not assess other key mediators like miRNAs. Furthermore, without western blotting and quantitative real-time PCR data, changes in the expression and regulation of key functional molecules in muscle and bone tissue remain unexamined, narrowing the pathway-specific conclusions. Future studies should utilize animals of both sexes and other established age-related musculoskeletal disease models. Additionally, targeted molecular techniques and in vitro co-culture systems should be employed to clarify the precise signalling mechanisms. Finally, long-term, large-scale randomized controlled trials in elderly populations with musculoskeletal decline should be conducted to determine the clinical effects of the combination of CaHMB, CBP and HA.
5. Conclusions
This study suggested that CaHMB improved muscle mass and function in naturally aged animals, while CBP alleviated bone loss by improving bone metabolism. Furthermore, the combination of CaHMB, CBP, and HA had multiple musculoskeletal benefits at relatively lower doses compared to individual components. Specifically, the combination could maintain muscle mass, enhance grip strength and exercise performance, and reduce bone resorption, thereby mitigating bone loss. The possible mechanism involved the regulation of critical signalling molecules of the muscle–bone axis, including irisin, IGF-1, MSTN, OCN, and SOST. Our findings highlight the potential effects of CaHMB, CBP, and HA supplementation on delaying age-related progression of muscle and bone loss, and their combined application appears to provide comprehensive and multiple benefits. This provides new research perspectives and potential intervention targets for preventing and improving age-related musculoskeletal diseases, thereby contributing to the extension of health span.Author contributions
Haoqi Chen: writing – original draft, methodology, investigation, and conceptualization. Junhong Peng: investigation. Shanshan Guo: methodology and investigation. Ting Chen: investigation. Si Chen: methodology, writing – review & editing, and conceptualization. Xinyuan Jin: investigation. Wenge Huang: methodology and investigation. Chao Xu: methodology and investigation. Mengchu Li: methodology and investigation. Mengxing Xie: investigation. Mengtao Yang: methodology and investigation. Jinzhu Pang: investigation and conceptualization. Huilian Zhu: writing – review & editing, funding acquisition, and conceptualization. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data used to support the findings of this study are included within the article and are available from the corresponding author upon request.Supplementary information is available. See DOI: https://doi.org/10.1039/d6fo00352d.
Acknowledgements
This study was supported by the Hainan Provincial Natural Science Foundation of China (821RC1139).References
- World Health Organization, Decade of healthy ageing: Baseline report, 2020.
- J. Luo, K. Mills, S. le Cessie, R. Noordam and D. van Heemst, Ageing, age-related diseases and oxidative stress: What to do next?, Ageing Res. Rev., 2020, 57, 100982 CrossRef CAS PubMed.
- A. Calcinotto, J. Kohli, E. Zagato, L. Pellegrini, M. Demaria and A. Alimonti, Cellular senescence: Aging, cancer, and injury, Physiol. Rev., 2019, 99, 1047–1078 CrossRef CAS PubMed.
- C. López-Otín and G. Kroemer, Hallmarks of health, Cell, 2021, 184, 33–63 CrossRef.
- L. Culig, X. Chu and V. A. Bohr, Neurogenesis in aging and age-related neurodegenerative diseases, Ageing Res. Rev., 2022, 78, 101636 CrossRef CAS.
- E. Gielen, J. Dupont, M. Dejaeger and M. R. Laurent, Sarcopenia, osteoporosis and frailty, Metab., Clin. Exp., 2023, 145, 155638 CrossRef CAS PubMed.
- S. J. Wang, Y. L. Chen, L. J. Xiong, N. N. Jin, P. F. Zhao, Z. Liang, L. X. Cheng and L. Kang, Multimorbidity measures associated with cognitive function among community-dwelling older Chinese adults, Alzheimers Dement., 2024, 20, 6221–6231 CrossRef PubMed.
- A. J. Cruz-Jentoft, G. Bahat, J. Bauer, Y. Boirie, O. Bruyère, T. Cederholm, C. Cooper, F. Landi, Y. Rolland, A. A. Sayer, S. M. Schneider, C. C. Sieber, E. Topinkova, M. Vandewoude, M. Visser and M. Zamboni, EWWGSOP2 and E. G. EWGSOP2, Sarcopenia: Revised European consensus on definition and diagnosis, Age Ageing, 2019, 48, 16–31 CrossRef.
- F. Petermann-Rocha, V. Balntzi, S. R. Gray, J. Lara, F. K. Ho, J. P. Pell and C. Celis-Morales, Global prevalence of sarcopenia and severe sarcopenia: A systematic review and meta-analysis, J. Cachexia Sarcopenia Muscle, 2022, 13, 86–99 CrossRef.
- Y. Y. Shen, X. Huang, J. Y. Wu, X. L. Lin, X. Zhou, Z. Zhu, X. W. Pan, J. Y. Xu, J. Qiao, T. Y. Zhang, L. X. Ye, H. W. Jiang, Y. Z. Ren and P. F. Shan, The global burden of osteoporosis, low bone mass, and its related fracture in 204 countries and territories, 1990–2019, Front. Endocrinol., 2022, 13, 882241 CrossRef.
- P. L. Xiao, A. Y. Cui, C. J. Hsu, R. Peng, N. Jiang, X. H. Xu, Y. G. Ma, D. Liu and H. D. Lu, Global, regional prevalence, and risk factors of osteoporosis according to the World Health Organization diagnostic criteria: A systematic review and meta-analysis, Osteoporosis Int., 2022, 33, 2137–2153 CrossRef.
- C. W. Hutton, Osteoarthritis: The cause not result of joint failure?, Ann. Rheum. Dis., 1989, 48, 958–961 CrossRef CAS.
- S. a. Tang, C. Zhang, W. M. Oo, K. Fu, M. A. Risberg, S. M. Bierma-Zeinstra, T. Neogi, I. Atukorala, A.-M. Malfait, C. Ding and D. J. Hunter, Osteoarthritis, Nat. Rev. Dis. Primers, 2025, 11, 10 CrossRef PubMed.
- A. J. Cruz-Jentoft and A. A. Sayer, Sarcopenia, Lancet, 2019, 393, 2636–2646 CrossRef PubMed.
- S. N. Morin, W. D. Leslie and J. T. Schousboe, Osteoporosis: A review, J. Am. Med. Assoc., 2025, 334, 894–907 CrossRef CAS PubMed.
- B. L. Riggs, L. J. Melton, R. A. Robb, J. J. Camp, E. J. Atkinson, L. McDaniel, S. Amin, P. A. Rouleau and S. Khosla, A population-based assessment of rates of bone loss at multiple skeletal sites: Evidence for substantial trabecular bone loss in young adult women and men, J. Bone Miner. Res., 2008, 23, 205–214 CrossRef.
- M. R. Laurent, V. Dubois, F. Claessens, S. M. P. Verschueren, D. Vanderschueren, E. Gielen and F. Jardí, Muscle-bone interactions: From experimental models to the clinic? A critical update, Mol. Cell. Endocrinol., 2016, 432, 14–36 CrossRef CAS PubMed.
- A. F. Deng, F. X. Wang, S. C. Wang, Y. Z. Zhang, L. Bai and J. C. Su, Bone-organ axes: Bidirectional crosstalk, Mil. Med. Res., 2024, 11, 37 Search PubMed.
- G. Colaianni, T. Mongelli, S. Colucci, S. Cinti and M. Grano, Crosstalk between muscle and bone via the muscle-myokine irisin, Curr. Osteoporos. Rep., 2016, 14, 132–137 CrossRef CAS.
- D. D. Bikle, C. Tahimic, W. H. Chang, Y. M. Wang, A. Philippou and E. R. Barton, Role of IGF-I signaling in muscle bone interactions, Bone, 2015, 80, 79–88 CrossRef CAS.
- Y. Qin, Y. Peng, W. Zhao, J. Pan, H. Ksiezak-Reding, C. Cardozo, Y. Wu, P. Divieti Pajevic, L. F. Bonewald, W. A. Bauman and W. Qin, Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: A novel mechanism in muscle-bone communication, J. Biol. Chem., 2017, 292, 11021–11033 CrossRef CAS.
- F. Bosco, V. Musolino, M. Gliozzi, S. Nucera, C. Carresi, M. C. Zito, F. Scarano, M. Scicchitano, F. Reale, S. Ruga, J. Maiuolo, R. Macrì, L. Guarnieri, A. R. Coppoletta, R. Mollace, C. Muscoli, E. Palma and V. Mollace, The muscle to bone axis (and viceversa): An encrypted language affecting tissues and organs and yet to be codified?, Pharmacol. Res., 2021, 165, 105427 CrossRef CAS.
- S. F. Liu, F. Gao, L. Wen, M. Ouyang, Y. Wang, Q. Wang, L. P. Luo and Z. J. Jian, Osteocalcin induces proliferation via positive activation of the PI3K/Akt, P38 MAPK pathways and promotes differentiation through activation of the GPRC6A-ERK1/2 pathway in C2C12 myoblast cells, Cell. Physiol. Biochem., 2017, 43, 1100–1112 CrossRef CAS PubMed.
- I. G. P. S. Aryana, S. S. Rini and C. H. Soejono, Importance of sclerostin as bone-muscle mediator crosstalk, Ann. Geriatr. Med. Res., 2022, 26, 72–82 CrossRef PubMed.
- S. L. Nissen and N. N. Abumrad, Nutritional role of the leucine metabolite β-hydroxy β-methylbutyrate (HMB), J. Nutr. Biochem., 1997, 8, 300–311 CrossRef CAS.
- J. A. Rathmacher, L. M. Pitchford, J. R. Stout, J. R. Townsend, R. Jäger, R. B. Kreider, B. I. Campbell, C. M. Kerksick, P. S. Harty, D. G. Candow, B. M. Roberts, S. M. Arent, D. S. Kalman and J. Antonio, International society of sports nutrition position stand: β-Hydroxy-β-methylbutyrate (HMB), J. Int. Soc. Sports Nutr., 2025, 22, 2434734 CrossRef PubMed.
- H. M. Wu, Y. Xia, J. Jiang, H. M. Du, X. Y. Guo, X. Liu, C. L. Li, G. W. Huang and K. J. Niu, Effect of beta-hydroxy-beta-methylbutyrate supplementation on muscle loss in older adults: A systematic review and meta-analysis, Arch. Gerontol. Geriatr., 2015, 61, 168–175 CrossRef CAS PubMed.
- H. L. Eley, S. T. Russell, J. H. Baxter, P. Mukerji and M. J. Tisdale, Signaling pathways initiated by β-hydroxy-β-methylbutyrate to attenuate the depression of protein synthesis in skeletal muscle in response to cachectic stimuli, Am. J. Physiol.: Endocrinol. Metab., 2007, 293, E923–E931 CrossRef CAS.
- H. J. Smith, S. M. Wyke and M. J. Tisdale, Mechanism of the attenuation of proteolysis-inducing factor stimulated protein degradation in muscle by β-hydroxy-β-methylbutyrate, Cancer Res., 2004, 64, 8731–8735 CrossRef CAS PubMed.
- J. Lee, S.-H. Kwon, H.-M. Kim, S. N. Fahey, D. R. Knighton and A. Sansom, Effect of a growth protein-colostrum fraction on bone development in juvenile rats, Biosci. Biotechnol. Biochem., 2008, 72, 1–6 CrossRef CAS PubMed.
- K. Tsuji-Naito and R. W. Jack, Concentrated bovine milk whey active proteins facilitate osteogenesis through activation of the JNK-ATF4 pathway, Biosci. Biotechnol. Biochem., 2012, 76, 1150–1154 CrossRef CAS PubMed.
- I. S. Bayer, Hyaluronic acid and controlled release: A review, Molecules, 2020, 25, 2649 CrossRef CAS PubMed.
- M. Oe, S. Sakai, H. Yoshida, N. Okado, H. Kaneda, Y. Masuda and O. Urushibata, Oral hyaluronan relieves wrinkles: A double-blinded, placebo-controlled study over a 12-week period, Clin., Cosmet. Invest. Dermatol., 2017, 10, 267–273 CrossRef CAS PubMed.
- N. G. Kotla, S. R. Bonam, S. Rasala, J. Wankar, R. A. Bohara, J. Bayry, Y. Rochev and A. Pandit, Recent advances and prospects of hyaluronan as a multifunctional therapeutic system, J. Controlled Release, 2021, 336, 598–620 CrossRef CAS PubMed.
- M. H. Park, J. C. Jung, S. Hill, E. Cartwright, M. H. Dohnalek, M. Yu, H. J. Jun, S. B. Han, J. T. Hong and D. J. Son, FlexPro MD®, a combination of krill oil, astaxanthin and hyaluronic acid, reduces pain behavior and inhibits inflammatory response in monosodium iodoacetate-induced osteoarthritis in rats, Nutrients, 2020, 12, 956 CrossRef CAS.
- A. Marinho, C. Nunes and S. Reis, Hyaluronic acid: A key ingredient in the therapy of inflammation, Biomolecules, 2021, 11, 1518 CrossRef CAS PubMed.
- R. Solà, R. M. Valls, I. Martorell, M. Giralt, A. Pedret, N. Taltavull, M. Romeu, A. Rodríguez, D. Moriña, V. L. de Frutos, M. Montero, M. C. Casajuana, L. Pérez, J. Faba, G. Bernal, A. Astilleros, R. González, F. Puiggrós, L. Arola, C. Chetrit and D. Martinez-Puig, A low-fat yoghurt supplemented with a rooster comb extract on muscle joint function in adults with mild knee pain: a randomized, double blind, parallel, placebo-controlled, clinical trial of efficacy, Food Funct., 2015, 6, 3531–3539 RSC.
- D. Moriña, S. Fernández-Castillejo, R. M. Valls, A. Pedret, N. Taltavull, M. Romeu, M. Giralt, M. Montero, G. Bernal, J. Faba, L. Pérez-Merino, R. Gonzalez, M. C. Casajuana, A. Rodríguez, L. Arola, F. Puiggrós, I. Möller, C. Chetrit, D. Martinez-Puig and R. Solà, Effectiveness of a low-fat yoghurt supplemented with rooster comb extract on muscle strength in adults with mild knee pain and mechanisms of action on muscle regeneration, Food Funct., 2018, 9, 3244–3253 RSC.
- M. Oe, T. Tashiro, H. Yoshida, H. Nishiyama, Y. Masuda, K. Maruyama, T. Koikeda, R. Maruya and N. Fukui, Oral hyaluronan relieves knee pain: A review, Nutr. J., 2016, 15, 11 CrossRef PubMed.
- Chinese Institute of Food Science and Technology, Scientific consensus on application of calcium β-hydroxy-β-methylbutyrate (CaHMB), J. Chin. Inst. Food Sci. Technol., 2024, 24, 486–497 Search PubMed.
- National Health Commission of the People's Republic of China, Announcement on the Approval of Gamma-Aminobutyric Acid, Colostrum Basic Protein, Conjugated Linoleic Acid, Conjugated Linoleic Acid Glyceride, Lactobacillus plantarum (Strain ST-III), and Eucommia ulmoides Seed Oil as New Resource Foods (No. 12, 2009), https://www.nhc.gov.cn/wjw/c100175/200910/4b835c2b642245f497e3c35a545758d1.shtml, (accessed 22 July, 2024).
- National Health Commission of the People's Republic of China, Announcement on Ten New Food Ingredients Including Shea Butter (No. 7, 2017), https://www.nhc.gov.cn/wjw/c100175/201706/3855e4cb0e384021840409f3b6b88a3c.shtml, (accessed 24 July, 2024).
- National Health Commission of the People's Republic of China, Announcement on 15 “Three New Food” Items Including Isaria cicadae Miq (Artificial Cultivation) (No. 9, 2020), https://www.nhc.gov.cn/sps/c100088/202101/a677d9fc167241d4a49d4a22329d774e.shtml, (accessed 24 July, 2024).
- M. Munroe, Z. S. Mahmassani, S. Dvoretskiy, J. J. Reid, B. F. Miller, K. Hamilton, J. S. Rhodes and M. D. Boppart, Cognitive function is preserved in aged mice following long-term β-hydroxy β-methylbutyrate supplementation, Nutr. Neurosci., 2020, 23, 170–182 CrossRef CAS PubMed.
- Y. R. Zhang, Z. Y. Qiao, J. L. Yu, C. H. Shi, R. Quan, W. Zhang, R. Bi, H. L. Li, W. T. Qian, M. H. Wang and Y. X. Li, Effects of dietary colostrum basic protein on bone growth and calcium absorption in mice, Nutrients, 2024, 16, 664 CrossRef CAS PubMed.
- K. P. Pritzker, S. Gay, S. A. Jimenez, K. Ostergaard, J. P. Pelletier, P. A. Revell, D. Salter and W. B. van den Berg, Osteoarthritis cartilage histopathology: Grading and staging, Osteoarthritis Cartilage, 2006, 14, 13–29 CrossRef CAS PubMed.
- S. S. Glasson, M. G. Chambers, W. B. Van Den Berg and C. B. Little, The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse, Osteoarthritis Cartilage, 2010, 18(Suppl 3), S17–S23 CrossRef.
- L. Liu, J. Guo, X. Chen, X. Tong, J. Xu and J. Zou, The role of irisin in exercise-mediated bone health, Front. Cell Dev. Biol., 2021, 9, 668759 CrossRef PubMed.
- T. Lapveteläinen, T. Nevalainen, J. J. Parkkinen, J. Arokoski, K. Kiraly, M. Hyttinen, P. Halonen and H. J. Helminen, Lifelong moderate running training increases the incidence and severity of osteoarthritis in the knee joint of C57BL mice, Anat. Rec., 1995, 242, 159–165 CrossRef PubMed.
- G. Wilhelmi and R. Faust, Suitability of the C 57 Black Mouse as an Experimental Animal for the Study of Skeletal Changes Due to Ageing, with Special Reference to Osteo-Arthrosis and Its Response to Tribenoside, Pharmacology, 1976, 14, 289–296 CrossRef CAS PubMed.
- M. Alves-Simões, Rodent models of knee osteoarthritis for pain research, Osteoarthritis Cartilage, 2022, 30, 802–814 CrossRef PubMed.
- Y. Sanada, Y. Ikuta, C. Ding, M. Shinohara, D. Yimiti, H. Ishitobi, K. Nagira, M. Lee, T. Akimoto, S. Shibata, M. Ishikawa, T. Nakasa, K. Matsubara, M. K. Lotz, N. Adachi and S. Miyaki, Senescence-accelerated mice prone 8 (SAMP8) in male as a spontaneous osteoarthritis model, Arthritis Res. Ther., 2022, 24, 235 CrossRef CAS PubMed.
- M. Maumus, D. Noël, H. K. Ea, D. Moulin, M. Ruiz, E. Hay, X. Houard, D. Cleret, M. Cohen-Solal, C. Jacques, J. Y. Jouzeau, M. H. Lafage-Proust, P. Reboul, J. Sellam, C. Vinatier, F. Rannou, C. Jorgensen, J. Guicheux and F. Berenbaum, Identification of TGFβ signatures in six murine models mimicking different osteoarthritis clinical phenotypes, Osteoarthritis Cartilage, 2020, 28, 1373–1384 CrossRef CAS PubMed.
- J. Xiong, X. Zhu, Y. Guo, H. Tang, C. Dong, B. Wang, M. Liu, Z. Li and Y. Tu, Multi-omic underpinnings of heterogeneous aging across multiple organ systems, Cell Genomics, 2025, 5, 101032 CrossRef CAS PubMed.
- Y. Ding, Y. Zuo, B. Zhang, Y. Fan, G. Xu, Z. Cheng, S. Ma, S. Fang, A. Tian, D. Gao, X. Xu, Q. Wang, Y. Jing, M. Jiang, M. Xiong, J. Li, Z. Han, S. Sun, S. Wang, F. He, J. Yang, J. Qu, W. Zhang and G. H. Liu, Comprehensive human proteome profiles across a 50-year lifespan reveal aging trajectories and signatures, Cell, 2025, 188, 5763–5784 CrossRef CAS PubMed.
- J. R. Stout, A. E. Smith-Ryan, D. H. Fukuda, K. L. Kendall, J. R. Moon, J. R. Hoffman, J. M. Wilson, J. S. Oliver and V. A. Mustad, Effect of calcium β-hydroxy-β-methylbutyrate (CaHMB) with and without resistance training in men and women 65+ yrs: A randomized, double-blind pilot trial, Exp. Gerontol., 2013, 48, 1303–1310 CrossRef CAS PubMed.
- N. E. P. Deutz, S. L. Pereira, N. P. Hays, J. S. Oliver, N. K. Edens, C. M. Evans and R. R. Wolfe, Effect of β-hydroxy-β-methylbutyrate (HMB) on lean body mass during 10 days of bed rest in older adults, Clin. Nutr., 2013, 32, 704–712 CrossRef CAS PubMed.
- L. Berton, G. Bano, S. Carraro, N. Veronese, S. Pizzato, F. Bolzetta, M. De Rui, E. Valmorbida, I. De Ronch, E. Perissinotto, A. Coin, E. Manzato and G. Sergi, Effect of oral beta-hydroxy-beta-methylbutyrate (HMB) supplementation on physical performance in healthy old women over 65 years: An open label randomized controlled trial, PLoS One, 2015, 10, e0141757 CrossRef PubMed.
- F. Gerlinger-Romero, L. Guimarães-Ferreira, G. Giannocco and M. T. Nunes, Chronic supplementation of beta-hydroxy-beta methylbutyrate (HMβ) increases the activity of the GH/IGF-I axis and induces hyperinsulinemia in rats, Growth Horm. IGF Res., 2011, 21, 57–62 CrossRef CAS PubMed.
- D. J. Wilkinson, T. Hossain, D. S. Hill, B. E. Phillips, H. Crossland, J. Williams, P. Loughna, T. A. Churchward-Venne, L. Breen, S. M. Phillips, T. Etheridge, J. A. Rathmacher, K. Smith, N. J. Szewczyk and P. J. Atherton, Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism, J. Physiol.-London, 2013, 591, 2911–2923 CrossRef CAS.
- Y. Hao, J. R. Jackson, Y. Wang, N. Edens, S. L. Pereira and S. E. Alway, β-Hydroxy-β-methylbutyrate reduces myonuclear apoptosis during recovery from hind limb suspension-induced muscle fiber atrophy in aged rats, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2011, 301, R701–R715 CrossRef CAS PubMed.
- M. Holeček, Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions, J. Cachexia Sarcopenia Muscle, 2017, 8, 529–541 CrossRef.
- K. K. Noh, K. W. Chung, Y. J. Choi, M. H. Park, E. J. Jang, C. H. Park, C. Yoon, N. D. Kim, M. K. Kim and H. Y. Chung, β-Hydroxy β-methylbutyrate improves dexamethasone-induced muscle atrophy by modulating the muscle degradation pathway in SD rat, PLoS One, 2014, 9, e102947 CrossRef PubMed.
- M. D. Girón, J. D. Vílchez, S. Shreeram, R. Salto, M. Manzano, E. Cabrera, N. Campos, N. K. Edens, R. Rueda and J. M. López-Pedrosa, β-Hydroxy-β-Methylbutyrate (HMB) normalizes dexamethasone-induced autophagy-lysosomal pathway in skeletal muscle, PLoS One, 2015, 10, e0117520 CrossRef PubMed.
- R. Kornasio, I. Riederer, G. Butler-Browne, V. Mouly, Z. Uni and O. Halevy, Beta-hydroxy-beta-methylbutyrate (HMB) stimulates myogenic cell proliferation, differentiation and survival via the MAPK/ERK and PI3K/Akt pathways, Biochim. Biophys. Acta, 2009, 1793, 755–763 CrossRef CAS.
- S. H. Ahn, H. W. Jung, E. Lee, J. Y. Baek, I. Y. Jang, S. J. Park, J. Y. Lee, E. Choi, Y. S. Lee, S. Hong and B. J. Kim, Decreased serum level of sclerostin in older adults with sarcopenia, Endocrinol. Metab., 2022, 37, 487–496 CrossRef CAS PubMed.
- C. M. Prado, C. E. Orsso, S. L. Pereira, P. J. Atherton and N. E. P. Deutz, Effects of β-hydroxy β-methylbutyrate (HMB) supplementation on muscle mass, function, and other outcomes in patients with cancer: A systematic review, J. Cachexia Sarcopenia Muscle, 2022, 13, 1623–1641 CrossRef PubMed.
- X.-R. Gao, Y.-S. Chen and W. Deng, The effect of vitamin D supplementation on knee osteoarthritis: A meta-analysis of randomized controlled trials, Int. J. Surg., 2017, 46, 14–20 CrossRef PubMed.
- J. Kositsawat, G. Duque and B. Kirk, Nutrients with anabolic/anticatabolic, antioxidant, and anti-inflammatory properties: Targeting the biological mechanisms of aging to support musculoskeletal health, Exp. Gerontol., 2021, 154, 111521 CrossRef CAS PubMed.
- J. E. Moreira-Velasco, M. F. Contreras-Alvarado, H. Rammal, D. Rivas and G. Duque, Beyond calcium and vitamin D: Exploring creatine, β-Hydroxy-β-methylbutyrate, prebiotics and probiotics in osteosarcopenia, Nutrients, 2025, 17, 2332 CrossRef CAS PubMed.
- J. A. Rathmacher, L. M. Pitchford, P. Khoo, H. Angus, J. Lang, K. Lowry, C. Ruby, A. C. Krajek, J. C. Fuller Jr and R. L. Sharp, Long-term effects of calcium β-hydroxy-β-methylbutyrate and vitamin D3 supplementation on muscular function in older adults with and without resistance training: A randomized, double-blind, controlled study, J. Gerontol., Ser. A, 2020, 75, 2089–2097 CrossRef CAS.
- T. R. Hill, S. Verlaan, E. Biesheuvel, R. Eastell, J. M. Bauer, I. Bautmans, K. Brandt, L. M. Donini, M. Maggio, T. Mets, C. J. Seal, S. L. Wijers, C. Sieber, T. Cederholm and T. J. Aspray, A Vitamin D, calcium and leucine-enriched whey protein nutritional supplement improves measures of bone health in sarcopenic non-malnourished older adults: The PROVIDE study, Calcif. Tissue Int., 2019, 105, 383–391 CrossRef CAS PubMed.
- S. Seino, K. Sumi, M. Narita, Y. Yokoyama, K. Ashida, A. Kitamura and S. Shinkai, Effects of low-dose dairy protein plus micronutrient supplementation during resistance exercise on muscle mass and physical performance in older adults: A randomized, controlled trial, J. Nutr., Health Aging, 2018, 22, 59–67 CrossRef CAS PubMed.
- K. Liberman, R. Njemini, Y. Luiking, L. N. Forti, S. Verlaan, J. M. Bauer, R. Memelink, K. Brandt, L. M. Donini, M. Maggio, T. Mets, S. L. J. Wijers, C. Sieber, T. Cederholm and I. Bautmans, Thirteen weeks of supplementation of vitamin D and leucine-enriched whey protein nutritional supplement attenuates chronic low-grade inflammation in sarcopenic older adults: The PROVIDE study, Aging: Clin. Exp. Res., 2019, 31, 845–854 Search PubMed.
- M. Rondanelli, E. Cereda, C. Klersy, M. A. Faliva, G. Peroni, M. Nichetti, C. Gasparri, G. Iannello, D. Spadaccini, V. Infantino, R. Caccialanza and S. Perna, Improving rehabilitation in sarcopenia: A randomized-controlled trial utilizing a muscle-targeted food for special medical purposes, J. Cachexia Sarcopenia Muscle, 2020, 11, 1535–1547 CrossRef PubMed.
- Q. Wu, Z. Xu, W. Huang, X. Qi, J. Wu, J. Du and J. Kan, Effect of high plant protein/peptide nutrition supplementation on knee osteoarthritis in older adults with sarcopenia: A randomized, double-blind, placebo-controlled trial, Clin. Nutr., 2024, 43, 2177–2185 CrossRef CAS PubMed.
- W. Liu, Y. Yu, Z. Zhang, Z. Li, Y. Wu, L. Xie, Z. Xu and C. Chang, Nutrition supplementation combined with exercise versus exercise alone in treating knee osteoarthritis: A double-blinded, randomised, placebo-controlled trial, Age Ageing, 2025, 54, afaf010 CrossRef PubMed.
- R. Altman, A. Bedi, A. Manjoo, F. Niazi, P. Shaw and P. Mease, Anti-inflammatory effects of intra-articular hyaluronic acid: A systematic review, Cartilage, 2019, 10, 43–52 CrossRef CAS PubMed.
- K. J. Gries, V. S. Zysik, T. K. Jobe, N. Griffin, B. P. Leeds and J. W. Lowery, Muscle-derived factors influencing bone metabolism, Semin. Cell Dev. Biol., 2022, 123, 57–63 CrossRef CAS PubMed.
- P. Mera, K. Laue, M. Ferron, C. Confavreux, J. Wei, M. Galán-Díez, A. Lacampagne, S. J. Mitchell, J. A. Mattison, Y. Chen, J. Bacchetta, P. Szulc, R. N. Kitsis, R. de Cabo, R. A. Friedman, C. Torsitano, T. E. McGraw, M. Puchowicz, I. Kurland and G. Karsenty, Osteocalcin signaling in myofibers is necessary and sufficient for optimum adaptation to exercise, Cell Metab., 2016, 23, 1078–1092 CrossRef CAS PubMed.
- S. Welle, K. Bhatt and C. A. Pinkert, Myofibrillar protein synthesis in myostatin-deficient mice, Am. J. Physiol.: Endocrinol. Metab., 2006, 290, E409–E415 CrossRef CAS PubMed.
- J. A. Kim, E. Roh, S. H. Hong, Y. B. Lee, N. H. Kim, H. J. Yoo, J. A. Seo, N. H. Kim, S. G. Kim, S. H. Baik and K. M. Choi, Association of serum sclerostin levels with low skeletal muscle mass: The Korean Sarcopenic Obesity Study (KSOS), Bone, 2019, 128, 115053 CrossRef CAS PubMed.
- G. Colaianni, M. Errede, L. Sanesi, A. Notarnicola, M. Celi, R. Zerlotin, G. Storlino, P. Pignataro, A. Oranger, V. Pesce, U. Tarantino, B. Moretti and M. Grano, Irisin correlates positively with BMD in a cohort of older adult patients and downregulates the senescent marker p21 in osteoblasts, J. Bone Miner. Res., 2021, 36, 305–314 CrossRef CAS PubMed.
- L. F. Wu, D. C. Zhu, B. H. Wang, Y. H. Lu, P. He, Y. H. Zhang, H. Q. Gao, X. W. Zhu, W. Xia, H. Zhu, X. B. Mo, X. Lu, L. Zhang, Y. H. Zhang, F. Y. Deng and S. F. Lei, Relative abundance of mature myostatin rather than total myostatin is negatively associated with bone mineral density in Chinese, J. Cell. Mol. Med., 2018, 22, 1329–1336 CrossRef CAS PubMed.
- P. Y. Chen, Y. H. Lee, C. H. Chiang, H. H. Chang, C. W. Lu and K. C. Huang, Sex differences and positive dose-response relationships between serum osteocalcin levels and low muscle strength, Gerontology, 2023, 69, 1056–1064 CrossRef CAS.
- C. Ohlsson, D. Mellström, D. Carlzon, E. Orwoll, O. Ljunggren, M. K. Karlsson and L. Vandenput, Older men with low serum IGF-1 have an increased risk of incident fractures: the MrOS Sweden study, J. Bone Miner. Res., 2011, 26, 865–872 CrossRef CAS PubMed.
- J. A. Robinson, M. Chatterjee-Kishore, P. J. Yaworsky, D. M. Cullen, W. Zhao, C. Li, Y. Kharode, L. Sauter, P. Babij, E. L. Brown, A. A. Hill, M. P. Akhter, M. L. Johnson, R. R. Recker, B. S. Komm and F. J. Bex, Wnt/beta-catenin signaling is a normal physiological response to mechanical loading in bone, J. Biol. Chem., 2006, 281, 31720–31728 CrossRef CAS.
- X. Qiao, Y. Nie, Y. Ma, Y. Chen, R. Cheng, W. Yin, Y. Hu, W. Xu and L. Xu, Irisin promotes osteoblast proliferation and differentiation via activating the MAP kinase signaling pathways, Sci. Rep., 2016, 6, 18732 CrossRef CAS PubMed.
- W. Ye, J. Wang, D. Lin and Z. Ding, The immunomodulatory role of irisin on osteogenesis via AMPK-mediated macrophage polarization, Int. J. Biol. Macromol., 2020, 146, 25–35 CrossRef CAS PubMed.
- J. Zhang, P. Valverde, X. Zhu, D. Murray, Y. Wu, L. Yu, H. Jiang, M. M. Dard, J. Huang, Z. Xu, Q. Tu and J. Chen, Exercise-induced irisin in bone and systemic irisin administration reveal new regulatory mechanisms of bone metabolism, Bone Res., 2017, 5, 16056 CrossRef CAS PubMed.
- Y. Wang, Z. Cheng, H. Z. Elalieh, E. Nakamura, M. T. Nguyen, S. Mackem, T. L. Clemens, D. D. Bikle and W. Chang, IGF-1R signaling in chondrocytes modulates growth plate development by interacting with the PTHrP/Ihh pathway, J. Bone Miner. Res., 2011, 26, 1437–1446 Search PubMed.
- M. Zhang, S. Xuan, M. L. Bouxsein, D. von Stechow, N. Akeno, M. C. Faugere, H. Malluche, G. Zhao, C. J. Rosen, A. Efstratiadis and T. L. Clemens, Osteoblast-specific knockout of the insulin-like growth factor (IGF) receptor gene reveals an essential role of IGF signaling in bone matrix mineralization, J. Biol. Chem., 2002, 277, 44005–44012 Search PubMed.
- M. Bowser, S. Herberg, P. Arounleut, X. Shi, S. Fulzele, W. D. Hill, C. M. Isales and M. W. Hamrick, Effects of the activin A-myostatin-follistatin system on aging bone and muscle progenitor cells, Exp. Gerontol., 2013, 48, 290–297 Search PubMed.
- M. N. Elkasrawy and M. W. Hamrick, Myostatin (GDF-8) as a key factor linking muscle mass and bone structure, J. Musculoskeletal Neuronal Interact., 2010, 10, 56–63 CAS.
- J. Yang, L. Sun, X. Fan, B. Yin, Y. Kang, L. Tang and S. An, Effect of exercise on bone in poorly controlled type 1 diabetes mediated by the ActRIIB/Smad signaling pathway, Exp. Ther. Med., 2018, 16, 3686–3693 Search PubMed.
- B. Dankbar, M. Fennen, D. Brunert, S. Hayer, S. Frank, C. Wehmeyer, D. Beckmann, P. Paruzel, J. Bertrand, K. Redlich, C. Koers-Wunrau, A. Stratis, A. Korb-Pap and T. Pap, Myostatin is a direct regulator of osteoclast differentiation and its inhibition reduces inflammatory joint destruction in mice, Nat. Med., 2015, 21, 1085–1090 Search PubMed.
- W. Wei, P. A. Dutchak, X. Wang, X. Ding, X. Wang, A. L. Bookout, R. Goetz, M. Mohammadi, R. D. Gerard, P. C. Dechow, D. J. Mangelsdorf, S. A. Kliewer and Y. Wan, Fibroblast growth factor 21 promotes bone loss by potentiating the effects of peroxisome proliferator-activated receptor γ, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 3143–3148 Search PubMed.
- H. Li, H. Sun, B. Qian, W. Feng, D. Carney, J. Miller, M. V. Hogan and L. Wang, Increased expression of FGF-21 negatively affects bone homeostasis in dystrophin/utrophin double knockout mice, J. Bone Miner. Res., 2020, 35, 738–752 CrossRef CAS PubMed.
- X. W. Zhu, K. Ding, X. Y. Dai and W. Q. Ling, β-Aminoisobutyric acid accelerates the proliferation and differentiation of MC3T3-E1 cells via moderate activation of ROS signaling, J. Chin. Med. Assoc., 2018, 81, 611–618 CrossRef PubMed.
- T. Komori, Functions of osteocalcin in bone, pancreas, testis, and muscle, Int. J. Mol. Sci., 2020, 21, 7513 CrossRef CAS PubMed.
- B. Y. Wang, Y. F. Chen, A. W. Hsiao, W. J. Chen, C. W. Lee and O. K. Lee, Ginkgolide B facilitates muscle regeneration via rejuvenating osteocalcin-mediated bone-to-muscle modulation in aged mice, J. Cachexia Sarcopenia Muscle, 2023, 14, 1349–1364 Search PubMed.
- J. Huang, S. Romero-Suarez, N. Lara, C. Mo, S. Kaja, L. Brotto, S. L. Dallas, M. L. Johnson, K. Jähn, L. F. Bonewald and M. Brotto, Crosstalk between MLO-Y4 osteocytes and C2C12 muscle cells is mediated by the Wnt/β-catenin pathway, JBMR Plus, 2017, 1, 86–100 CrossRef CAS PubMed.
- M. C. Medeiros, N. Rocha, E. Bandeira, I. Dantas, C. Chaves, M. Oliveira and F. Bandeira, Serum sclerostin, body composition, and sarcopenia in hemodialysis patients with diabetes, Internet J. Nephrol., 2020, 2020, 4596920 Search PubMed.
- K. Amrein, S. Amrein, C. Drexler, H. P. Dimai, H. Dobnig, K. Pfeifer, A. Tomaschitz, T. R. Pieber and A. Fahrleitner-Pammer, Sclerostin and its association with physical activity, age, gender, body composition, and bone mineral content in healthy adults, J. Clin. Endocrinol. Metab., 2012, 97, 148–154 Search PubMed.
- I. Aryana, S. S. Rini and C. H. Soejono, Importance of Sclerostin as Bone-Muscle Mediator Crosstalk, Ann. Geriatr. Med. Res., 2022, 26, 72–82 CrossRef PubMed.
- R. Elsurer Afsar, B. Afsar and T. A. Ikizler, Fibroblast growth factor 23 and muscle wasting: A metabolic point of view, Kidney Int. Rep., 2023, 8, 1301–1314 CrossRef PubMed.
- R. C. Langen, A. M. Schols, M. C. Kelders, E. F. Wouters and Y. M. Janssen-Heininger, Inflammatory cytokines inhibit myogenic differentiation through activation of nuclear factor-kappaB, FASEB J., 2001, 15, 1169–1180 CrossRef CAS PubMed.
- C. L. Mendias, J. P. Gumucio, M. E. Davis, C. W. Bromley, C. S. Davis and S. V. Brooks, Transforming growth factor-beta induces skeletal muscle atrophy and fibrosis through the induction of atrogin-1 and scleraxis, Muscle Nerve, 2012, 45, 55–59 CrossRef CAS.
- S. Pluchino and J. A. Smith, Explicating exosomes: Reclassifying the rising stars of intercellular communication, Cell, 2019, 177, 225–227 CrossRef CAS PubMed.
- G. van Niel, G. D'Angelo and G. Raposo, Shedding light on the cell biology of extracellular vesicles, Nat. Rev. Mol. Cell Biol., 2018, 19, 213–228 CrossRef CAS PubMed.
- B. Kirk, G. Lombardi and G. Duque, Bone and muscle crosstalk in ageing and disease, Nat. Rev. Endocrinol., 2025, 21, 375–390 CrossRef CAS PubMed.
- Q. Xu, Y. Cui, J. Luan, X. Zhou, H. Li and J. Han, Exosomes from C2C12 myoblasts enhance osteogenic differentiation of MC3T3-E1 pre-osteoblasts by delivering miR-27a-3p, Biochem. Biophys. Res. Commun., 2018, 498, 32–37 CrossRef CAS PubMed.
- S. Fulzele, B. Mendhe, A. Khayrullin, M. Johnson, H. Kaiser, Y. Liu, C. M. Isales and M. W. Hamrick, Muscle-derived miR-34a increases with age in circulating extracellular vesicles and induces senescence of bone marrow stem cells, Aging, 2019, 11, 1791–1803 CrossRef CAS.
- Q. Sun, Y. Zhang, G. Yang, X. Chen, Y. Zhang, G. Cao, J. Wang, Y. Sun, P. Zhang, M. Fan, N. Shao and X. Yang, Transforming growth factor-beta-regulated miR-24 promotes skeletal muscle differentiation, Nucleic Acids Res., 2008, 36, 2690–2699 CrossRef CAS PubMed.
- M. J. Zhao, J. Xie, W. J. Shu, H. Y. Wang, J. Bi, W. Jiang and H. N. Du, MiR-15b and miR-322 inhibit SETD3 expression to repress muscle cell differentiation, Cell Death Dis., 2019, 10, 183 CrossRef PubMed.
- E. Zacharewicz, P. Della Gatta, J. Reynolds, A. Garnham, T. Crowley, A. P. Russell and S. Lamon, Identification of microRNAs linked to regulators of muscle protein synthesis and regeneration in young and old skeletal muscle, PLoS One, 2014, 9, e114009 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)