 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Simulated gastrointestinal digestion of cocoa powder (INFOGEST): methylxanthine bioaccessibility and antioxidant properties
Leopoldo
Rivera-Manzorro
a,
María José
Aliaño-González
b,
Gerardo Fernández
Barbero
 b,
Nuria
Chinchilla
b,
Nuria
Chinchilla
 *a,
Francisco Antonio
Macías
*a,
Francisco Antonio
Macías
 a,
Miguel
Palma-Lovillo
b and
Ceferino
Carrera
b
a,
Miguel
Palma-Lovillo
b and
Ceferino
Carrera
b
aDepartment of Organic Chemistry, Faculty of Sciences, Institute of Biomolecules (INBIO), University of Cadiz, 11510 Puerto Real, Spain. E-mail: nuria.chinchilla@uca.es
bDepartment of Analytical Chemistry, Faculty of Sciences, Institute for Wine and Agri-Food Research (IVAGRO), University of Cadiz, Agri-Food Campus of International Excellence (ceiA3), 11510 Puerto Real, Spain
First published on 17th March 2026
Abstract
Cocoa (Theobroma cacao L.) is a rich source of bioactive compounds, including methylxanthines (theobromine and caffeine) and polyphenols, which are known for their stimulant, antioxidant, and cardioprotective properties. In this study, an ultrasound-assisted extraction (UAE) method was developed and optimized for methylxanthine recovery from commercial cocoa powders and cocoa-based drinking chocolate mixes (powdered products) using a Box–Behnken design and response surface methodology (BBD–RSM). The optimal conditions for this experiment were as follows: 67.81% EtOH, 50.10 °C, 36.02% amplitude, 0.3 s−1 cycle, and 10 minutes of extraction. In parallel, three samples (C-1, C-2, and C-4) were selected for in vitro digestion (INFOGEST 2.0) to assess the bioaccessibility and stability of bioactive compounds during gastrointestinal transit. Across the oral, gastric and intestinal phases, accumulative methylxanthine bioaccessibility reached more than 85%, indicating matrix-dependent release and possible co-extracted modulators. Antioxidant capacity, measured using DPPH assay, was high prior to digestion and showed phase-dependent changes during INFOGEST digestion, with lower values in the oral phase followed by higher measured antioxidant capacity in the gastric and/or intestinal phases, consistent with digestion-driven release of antioxidant compounds. The present study highlights the differential bioaccessibility of methylxanthines and the pivotal role of formulation factors in determining their gastrointestinal fate. Overall, the study establishes a sustainable, food-grade UAE workflow as a scalable starting point for obtaining methylxanthine-enriched cocoa extracts, while providing mechanistic insight into methylxanthine bioaccessibility in cocoa powder-based products during INFOGEST digestion; further work is required to translate these conditions into nutraceutical/functional ingredients (e.g., stability during concentration/drying, sensory and regulatory aspects).
Introduction
The growing consumer interest in food composition, the presence of additives, and their potential health effects has led to a substantial increase in the consumption of cocoa and its derivatives. Cocoa (Theobroma cacao L.), which is indigenous to the Amazonian region, is classified within the class Magnoliopsida, order Malvales, family Malvaceae, and genus Theobroma. It is a diploid fruit species (2n = 2x = 20) whose seeds, measuring 2–3 cm, are utilized in the production of chocolate, cocoa butter, and cocoa powder. The quantity of fruit required to produce 1 kg of dry cocoa varies between 15 and 31 fruits, depending upon the size of the fruit and its seed content.1,2Advancements in processing and transportation technologies have facilitated the dissemination of cocoa on a global scale, contributing to its widespread acceptance due to its favorable organoleptic properties, which encompass attributes such as flavor and texture. In the contemporary era, cocoa is consumed in a variety of forms and is recognized for its nutritional and gastronomic values. According to the Food and Agriculture Organization (FAO) and the International Cocoa Organization (ICCO), global cocoa production for the 2023–2024 season is estimated at 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 382
382![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tonnes. This figure underscores the economic significance of cocoa production and its prominent position in the global food market.3,4
000 tonnes. This figure underscores the economic significance of cocoa production and its prominent position in the global food market.3,4
Cocoa beans (seeds) and the cocoa solids derived from them are characterized by a complex chemical composition, encompassing water, carbohydrates, proteins, fiber, minerals, and bioactive compounds such as polyphenols, alkaloids, terpenoids, and lipids. These elements contribute to the sensory characteristics and biological benefits of cocoa.1,2 Among the various alkaloids present in cocoa, methylxanthines – specifically theobromine, caffeine, and theophylline – have been identified as the principal bioactive constituents.5,6 Caffeine, a widely consumed stimulant, functions as an adenosine receptor antagonist, thereby enhancing alertness and promoting dopamine release.7–10 Theobromine has been shown to exert anti-inflammatory, vasodilatory and bronchodilatory effects, with comparatively lower stimulation of the central nervous system.11–13 In contrast, theophylline, which is utilized clinically as a bronchodilator, must be administered with caution due to potential toxicity.14,15 The concentration and ratio of these methylxanthines are influenced by genetic, environmental, and technological factors. These factors directly affect the quality and potency of the physiological effects of cocoa.
Methylxanthines have been demonstrated to contribute to wakefulness, cognitive performance, vascular elasticity, blood pressure regulation, and reductions in body weight and body mass index among overweight individuals.16–19 From a clinical perspective, methylxanthines have been employed in the treatment of neonatal apnea, while caffeine has demonstrated neuroprotective properties in Alzheimer's disease models through the reduction of beta-amyloid deposition and the enhancement of memory and cognitive function.20,21 Collectively, flavonoids and methylxanthines modulate cardiovascular function, energy metabolism, inflammation, and neuronal activity, positioning cocoa as a functional food with both nutritional and therapeutic applications.
In this context, cocoa powders and related matrices may serve as affordable raw materials for obtaining methylxanthine-enriched extracts that could be incorporated into functional foods, nutraceutical formulations, or encapsulated supplements, provided that extraction relies on food-grade solvents and mild processing conditions. Therefore, there is interest in efficient and sustainable extraction approaches capable of concentrating methylxanthines while preserving their chemical integrity and enabling downstream standardization.
Cocoa-derived products differ markedly in composition and technological history (e.g., roasting, alkalization, fat content, milling, and the addition of sugar, starch, emulsifiers, and flavorings), which in turn may modulate methylxanthine release and interactions during digestion. Importantly, methylxanthine levels and profiles are strongly influenced by product category and cocoa solid content: cocoa powders and cocoa liquor/unsweetened cocoa mass generally contain high methylxanthine concentrations, whereas dark chocolate typically contains lower but still substantial levels depending on cocoa solids, milk chocolate shows intermediate levels, and white chocolate contains negligible methylxanthines due to the absence of cocoa solids. The present study focuses on usual commercial cocoa-bean-derived products in powder form, including unsweetened 100% cocoa products and cocoa-based drinking chocolate mixes containing sugar and/or starch and other additives.
These differences yield matrices with distinct release profiles and potential interactions during digestion. Consequently, the actual fraction of methylxanthines that becomes intestinally absorbable remains uncertain, and claims based solely on content cannot establish functional efficacy.
In vitro digestion is a laboratory approach that emulates human gastrointestinal conditions. It is used to evaluate bioaccessibility and stability of food constituents without involving absorption. The INFOGEST 2.0 consensus method is a standardized, static model that reproduces oral, gastric and intestinal environments using simulated fluids and physiologically relevant pH, enzymes and incubation schedules. This enables phase-resolved assessment in complex food matrices. The study identified three key strengths, namely, simplicity, high reproducibility and cross-laboratory comparability.22,23
In view of the marked effects of the matrix on commercial cocoa, it is imperative to ascertain which constituent of the methylxanthine complex becomes bioaccessible under gastrointestinal conditions, to establish a correlation between composition and the likely physiological impact. Accordingly, the aims of this study were (i) to develop and optimize an ultrasound-assisted extraction (UAE) process based on ethanol–water as a food-grade solvent system to maximize methylxanthine recovery from commercial cocoa products, with potential applicability for producing methylxanthine-enriched extracts as standardized cocoa-derived ingredients; and (ii) to apply the INFOGEST 2.0 static in vitro digestion model to selected cocoa products as consumed in order to determine phase-resolved methylxanthine bioaccessibility and relate release patterns to formulation- and matrix-related characteristics. Overall, the study elucidates the phase-dependent gastrointestinal behavior of cocoa methylxanthines and links these outcomes to matrix characteristics, providing a basis for future development of cocoa-derived methylxanthine-enriched ingredients.
Materials and methods
Samples
Six commercially available cocoa-bean-derived products in powder form were purchased from supermarkets in Cádiz (Andalusia, Spain) in October–November 2024 (Table 1). The set comprised four unsweetened products labelled 100% cocoa (natural or alkalized/with acidity regulators; defatted and/or ‘soluble’; and with or without cocoa butter) and two cocoa-based drinking chocolate mixes containing sugar and/or starch and additional ingredients (e.g., emulsifiers and flavorings). These products were selected to represent formulation-driven variability within cocoa powders and cocoa-based mixes, which is relevant for interpreting methylxanthine content and digestion behavior. Brand information is provided solely for traceability; however, throughout the manuscript the samples are primarily discussed according to product category and formulation/matrix characteristics. To optimize the extraction method, a homogeneous matrix was prepared by mixing three unsweetened 100% cocoa products (C-1, C-2, and C-3). After the optimization, each sample was analyzed individually under optimal conditions.| Code | Brand | Description/product type |
|---|---|---|
| C-1 | La chocolatera | 100% pure defatted cocoa powder, no added sugar, with acidity regulators (potassium carbonate and hydroxide) |
| C-2 | Chocolates valor | 100% pure natural soluble cocoa with cocoa butter (11%) and acidity regulators, no added sugar |
| C-3 | ColaCao | 100% pure natural cocoa powder with no added sugar or additives |
| C-4 | Chocolates valor | Drinking chocolate. Blend of defatted cocoa powder (20%), sugar, corn starch, emulsifier and flavorings |
| C-5 | Dulcinea | Fat-free cocoa powder with cocoa butter (16%) and acidity regulators, no added sugar |
| C-6 | Carrefour | Soluble mixture of fat-free cocoa powder (25%), sugar, acidity regulators, thickener, cinnamon and flavorings |
Chemicals and solvents
Ethanol (EtOH, 100% purity, VWR, Radnor, PA, USA) and Milli-Q water obtained using a Wasserlab Ecomatic system (Barbatáin, Spain) were utilized for the extraction of bioactive compounds. For the HPLC analysis, the mobile phase was composed of Milli-Q water acidified with 2% acetic acid (AcOH, ≥99%, Panreac, Castellar del Vallès, Spain) and methanol (MeOH, ≥99%, VWR, Radnor, PA, USA). Caffeine and theobromine standards (Sigma-Aldrich, St. Louis, MO, USA) were utilized for quantification purposes.The digestive phases were prepared using KCl, KH2PO4, NaHCO3, MgCl2·6H2O, NaCl, NaOH (≥99%) and HCl (37–38%), all from Panreac (Castellar del Vallès, Spain), and ammonium carbonate ((NH4)2CO3, ≥99%, Sigma-Aldrich, San Luis, MO, USA). Solutions were prepared using Milli-Q water. In vitro digestion was simulated using α-amylase from Aspergillus oryzae (75 U mL−1), pepsin from porcine gastric mucosa (2000 U mL−1), and lipase from porcine pancreas (100 U mL−1), all from Sigma-Aldrich (San Luis, MO, USA). The antioxidant capacity of the samples was assessed using DPPH and Trolox, and both had a purity of ≥98% (Sigma-Aldrich, San Luis, MO, USA).
Ultrasound-assisted extraction
UAE optimization was pursued with a dual purpose: (i) to ensure robust analytical quantification of total methylxanthines (initial content and post-digestion residues) to support phase-resolved bioaccessibility calculations, and (ii) to establish food-grade, mild processing conditions with potential applicability to the production of methylxanthine-enriched cocoa extracts.
The equipment employed in this study was a BANDELIN SONOPLUS HD 2200.2 (BANDELIN electronic GmbH & Co. KG, Berlin, Germany), coupled to an MS 72 ultrasonic probe with a 2 cm diameter tip. This apparatus facilitates meticulous regulation of pivotal variables including time, temperature, cycle and amplitude, which are indispensable for optimizing extraction performance. To maintain a constant temperature during the process, a water bath with a thermocouple thermometer was utilized for control purposes.
The design utilizes three levels for each variable: low (−1), medium (0), and high (1). A significant distinction between common factorial designs and other experimental designs is evident in the strategic configuration of experimental points. This strategic arrangement is pivotal in preventing combinations where all variables are simultaneously at their extreme values, whether maximum or minimum. This feature enables the acquisition of equivalent information while maintaining robust statistical precision, and consequently, a reduced number of experiments is required. Furthermore, BBD–RSM is distinguished by the selection of test points situated in intermediate areas within the range of each variable, thereby circumventing extreme combinations that could potentially compromise the stability of the process. This design offers significant benefits, including reduced energy and solvent consumption, thereby minimizing the potential environmental and economic consequences of less sustainable processes.24–26
Four factors were selected for extraction optimization based on the literature and the research group's previous experience: %EtOH (60–80–100%), temperature (30–50–70 °C), cycle (0.3–0.6–0.9 s−1) and amplitude (20–35–50%). The temperature range (30–70 °C) was selected to keep UAE under mild conditions and to minimize the risk of thermal alteration of cocoa constituents. Methylxanthines are regarded as thermally stable compounds, with reported thermal degradation occurring at substantially higher temperatures (e.g., caffeine degradation around 146 °C),27 and theobromine has been described as highly thermostable in extraction studies.
The design utilizes a rotational or partially rotational approach to generate second-order polynomial models, as described by eqn (1):
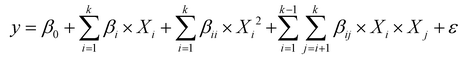 | (1) |
After the definition of these variables, the experimental design was subjected to adjustment through the implementation of the BBD–RSM, encompassing a total of 27 trials (Table 2) that were derived from the systematic combination of the four selected factors.
| Exp. | %EtOH | Temperature (°C) | Cycle (s−1) | Amplitude (%) | mg methylxanthines per g observed | mg methylxanthines per g adjusted | Error (%) |
|---|---|---|---|---|---|---|---|
| 1 | 80 | 50 | 0.6 | 35 | 13.23 | 13.46 | 1.74 |
| 2 | 60 | 50 | 0.6 | 50 | 13.53 | 13.71 | 1.33 |
| 3 | 80 | 50 | 0.6 | 35 | 13.59 | 13.46 | 0.93 |
| 4 | 80 | 70 | 0.6 | 50 | 13.40 | 13.29 | 0.77 |
| 5 | 80 | 70 | 0.6 | 20 | 13.28 | 13.29 | 0.08 |
| 6 | 100 | 30 | 0.6 | 35 | 7.01 | 7.29 | 3.89 |
| 7 | 80 | 30 | 0.6 | 50 | 13.36 | 13.09 | 2.03 |
| 8 | 80 | 50 | 0.9 | 20 | 13.22 | 13.36 | 1.05 |
| 9 | 60 | 50 | 0.9 | 35 | 13.79 | 13.54 | 1.82 |
| 10 | 60 | 70 | 0.6 | 35 | 13.62 | 13.45 | 1.23 |
| 11 | 80 | 30 | 0.9 | 35 | 13.19 | 13.28 | 0.69 |
| 12 | 60 | 50 | 0.6 | 20 | 13.51 | 13.65 | 1.00 |
| 13 | 80 | 30 | 0.3 | 35 | 13.15 | 13.17 | 0.17 |
| 14 | 60 | 30 | 0.6 | 35 | 13.76 | 13.80 | 0.31 |
| 15 | 80 | 70 | 0.3 | 35 | 13.44 | 13.50 | 0.48 |
| 16 | 100 | 50 | 0.9 | 35 | 8.39 | 8.07 | 3.83 |
| 17 | 80 | 70 | 0.9 | 35 | 13.20 | 13.34 | 1.01 |
| 18 | 100 | 70 | 0.6 | 35 | 7.96 | 8.02 | 0.78 |
| 19 | 60 | 50 | 0.3 | 35 | 14.01 | 14.07 | 0.43 |
| 20 | 80 | 50 | 0.3 | 20 | 13.49 | 13.39 | 0.77 |
| 21 | 100 | 50 | 0.3 | 35 | 7.60 | 7.59 | 0.12 |
| 22 | 100 | 50 | 0.6 | 50 | 7.65 | 7.67 | 0.26 |
| 23 | 100 | 50 | 0.6 | 20 | 7.76 | 7.74 | 0.32 |
| 24 | 80 | 30 | 0.6 | 20 | 13.26 | 13.10 | 1.19 |
| 25 | 80 | 50 | 0.9 | 50 | 13.16 | 13.36 | 1.58 |
| 26 | 80 | 50 | 0.6 | 35 | 13.56 | 13.46 | 0.76 |
| 27 | 80 | 50 | 0.3 | 50 | 13.41 | 13.38 | 0.25 |
Following the implementation of the BBD–RSM analysis and the determination of the optimal extraction conditions, the influence of extraction time was evaluated using intervals between 5 and 30 minutes. The objective of this study was to analyze how time affects the extraction process of these compounds and to determine the minimum time necessary to carry out the extraction while avoiding the degradation of the compounds of interest present in the solid sample.
Methylxanthine analysis
The mass spectrum was acquired in positive ionization mode using an electrospray source, with a desolvation temperature of 600 °C, a capillary voltage of 0.8 kV and a cone voltage of 15 V. The data were recorded in full scan mode in the m/z range of 50–700. The following bioactive compounds were identified based on their mass spectra [H+]: theobromine (m/z 181.17) and caffeine (m/z 195.18). In the individual analysis of sample C-4, vanillin (m/z 153.12) was also detected.
The experiment utilized a C18 column (250 × 4.6 mm, 5 µm, VWR) and the mobile phases consisted of water acidified with 2% acetic acid (phase A) and methanol (phase B). These phases were employed in isocratic mode, with a ratio of 80% A and 20% B, at a flow rate of 1 mL min−1. The total analysis time was 16 minutes.
In this study, theobromine and caffeine (Fig. 1) were identified and subsequently quantified using calibration curves that had been prepared in advance: theobromine; Y = 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 128X + 19
128X + 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 787; R2: 0.9992, and caffeine; Y = 12
787; R2: 0.9992, and caffeine; Y = 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 457X + 3640; R2: 0.9997. The total concentration of methylxanthines was calculated as the sum of both compounds, with no theophylline detected.
457X + 3640; R2: 0.9997. The total concentration of methylxanthines was calculated as the sum of both compounds, with no theophylline detected.
In vitro digestion
Following quantification of methylxanthine content in all cocoa products, three representative cocoa products (C-1, C-2 and C-4) were selected for in vitro digestion. Digestion was performed directly on the cocoa products as purchased (i.e., without prior solvent extraction), according to the INFOGEST 2.0 protocol.The in vitro digestion process was executed in three sequential phases (oral, gastric and intestinal) in accordance with the standardized protocol. The samples were incubated in a Climo-Shaker ISF4-X (Kuhner Shaker, Birsfelden, Switzerland). In the oral phase, the samples were subjected to incubation with simulated salivary fluid (SSF, pH 7.0) and α-amylase for a period of two minutes at a temperature of 37 °C. The gastric phase was reproduced using gastric fluid (SGF, pH 3.0) with pepsin, while the intestinal phase was carried out with intestinal fluid (SIF, pH 7.0) and pancreatin. Both phases were then subjected to an incubation period of 2 h at a temperature of 37 °C. Subsequent to the conclusion of each stage, aliquots were collected, subjected to centrifugation and filtration, and stored at −20 °C for subsequent analysis. The undigested cocoa residue was separated by means of centrifugation, freeze-dried, and stored for subsequent use in the extraction of bioactive compounds.
Antioxidant capacity
The antioxidant activity of the cocoa samples, both before and after in vitro digestion, was evaluated using the DPPH˙ (2,2-diphenyl-1-picrylhydrazil) assay. This allowed the stability of the bioactive compounds under gastrointestinal conditions to be assessed.28,29In the DPPH˙ assay (absorbance 515 nm), the reduction of free radicals was achieved by the addition of 15 μL of extract to 165 μL of DPPH solution (0.06 mM in methanol) and subsequent incubation for 30 min in the dark.28,29
The decrease in the rate of light absorption is indicative of the antioxidant capacity of the samples; DPPH˙ changes from purple to yellowish when interacting with the antioxidants present in cocoa. The percentage of inhibition was calculated according to eqn (2):
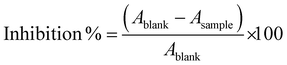 | (2) |
Statistical analysis
A one-factor analysis of variance (ANOVA) with 95% confidence was employed to evaluate the comparative outcomes of the initial phase of this study, encompassing the effects of solvent percentage and extraction temperature. The Duncan post-hoc methodology was used to facilitate the interpretation of the ANOVA results. In addition, the BBD–RSM design of experiments was developed and subsequently analysed, with 95% confidence and using Statgraphics Centurion software, version XVIII (Warrenton, VA, USA).Results and discussion
Univariate assessment of %EtOH and temperature
It is imperative to optimize the extraction conditions to ensure the efficient extraction of methylxanthines from cocoa samples. The composition of the solvent and the temperature employed during the extraction process are pivotal factors in determining the outcome.To evaluate the composition of the solvent as a variable in the extraction process, 0.5 g of sample was extracted with 20 mL of solvent while varying the proportion of EtOH between 0 and 100% (v/v). Ethanol–water was selected as the extraction medium due to its compatibility with food-grade processing and its relevance for potential translation of UAE conditions to the production of cocoa-derived methylxanthine-enriched ingredients.
The extractions were carried out for a period of 10 minutes while maintaining constant experimental conditions, namely 30% amplitude, 0.5 s−1 duty cycle and room temperature. The average concentration of methylxanthines per gram of sample was calculated based on the different percentages of EtOH and is shown in Fig. 2. The analyses were performed in duplicate.
 | ||
| Fig. 2 Evaluation of the percentage of ethanol in the extraction of methylxanthines (mg g−1 sample) (n = 2). Different letters indicate significant differences at the 95% confidence level. | ||
The extraction efficiency exhibited a direct correlation with the EtOH concentration, reaching a maximum close to 100%. ANOVA revealed significant differences (p = 0.016), indicating that the percentage of EtOH influences the extraction of methylxanthines. No significant differences were observed across the 0% to 60% EtOH range; the maximum value was recorded at approximately 80% EtOH. Based on these pivotal results, the low, medium, and high levels of EtOH (60%, 80%, and 100%) were established for the BBD–RSM design.
In addition, an evaluation was conducted of the influence of temperature (Fig. 3). Samples weighing 0.5 g were weighed and subjected to an extraction procedure using 20 mL of 80% ethanol solvent. The amplitude and cycle were maintained at a constant level of 30%, while the cycle was set at 0.5 s−1. The temperature evaluated was found to be in the range of 30–70 °C, as reported in the relevant literature, to avoid degradation of the compounds of interest.
 | ||
| Fig. 3 Temperature evaluation in the extraction of methylxanthines (mg g−1 sample) (n = 2). Different letters indicate significant differences at the 95% confidence level. | ||
The graph demonstrates a clear correlation between the extraction yield of methylxanthine and the temperature of the environment, with the yield increasing in proportion to the increase in temperature until it stabilizes at approximately 50 °C. ANOVA revealed significant differences (p = 0.017), thereby demonstrating that temperature has a considerable impact on methylxanthine extraction. These findings were instrumental in establishing the temperature range in the BBD–RSM design, wherein 30, 50, and 70 °C were designated as low, medium, and high levels, respectively.
BBD–RSM
A BBD–RSM experimental design was developed, taking into consideration four independent variables: ethanol percentage (%EtOH), temperature, cycle, and amplitude. The experimental design comprised a total of 27 experiments (Table 2), enabling a systematic evaluation of the linear, quadratic, and interactive effects of the selected variables on the efficiency of the methylxanthine extraction process. All experiments were performed randomly and the concentration of methylxanthines based on grams of sample was calculated for each experiment.The high quality of the fit is reflected in a coefficient of determination R2 = 0.9959, indicating that 99.59% of the observed variability in methylxanthine concentration is explained by the model. Furthermore, analysis of the Durbin–Watson statistic (p = 0.8862) confirmed the absence of autocorrelation in the model residuals, meaning that the results are reliable, with no repeated errors or hidden patterns. Finally, the average error between the observed and adjusted values was only 1.07%, with minimum errors close to 0.08% and maximum errors not exceeding 3.89%.
Therefore, to systematically evaluate the effects of the independent variables on the concentration of methylxanthines, a second-order polynomial model was developed to describe the relationship between the variables and the methylxanthine content, expressed in milligrams per gram of sample. The resulting mathematical model is shown in eqn (3):
 | (3) |
The equation incorporates both the main effects of each factor and the interactions between them and their quadratic components, thus facilitating accurate modelling of the non-linear and synergistic behaviors that may occur during extraction.
The results obtained from the ANOVA were found to be of paramount importance in identifying which variables exerted a statistically significant influence on the concentration of extracted methylxanthines. This analysis included the sum of squares, F-statistic, and the corresponding p-value for each term of the model applied to the methylxanthine extraction process from cocoa. A significance level of α < 0.05 (95% confidence) was established for the interpretation of results, with factors having p-values below this threshold considered to have a relevant effect on the response.
The analysis revealed that ethanol percentage (A) was the linear factor with the highest statistical significance (p < 0.001), highlighting its predominant effect on the extraction process. Furthermore, the quadratic term of ethanol percentage (AA) exhibited a highly significant effect (p < 0.001), indicating a nonlinear influence of the solvent on extraction efficiency. About the observed interactions, a significant effect was observed between the percentage of ethanol and the temperature (AB, p = 0.03), as well as between the percentage of ethanol and the cycle (AC, p = 0.05). These findings emphasize the importance of the combined effect of these factors. This finding suggests that the effect of ethanol on extraction does not act independently but is modulated by temperature and ultrasound cycle. In contrast, the remaining terms, including the linear effects of temperature (B), cycle (C), and amplitude (D), their respective interactions, and the quadratic terms of temperature (BB), cycle (CC), and amplitude (DD), did not exhibit statistically significant effects on methylxanthine concentration (p > 0.05). This does not imply that these factors have no influence on the process; rather, it suggests that, within the experimental range that was tested, their effects were not sufficiently strong or consistent to reach statistical significance.
Finally, the results of the ANOVA (Table 3) test were represented using a standardized Pareto chart (Fig. 4), which provides a clear, hierarchical visualization of the magnitude of the effect of each variable, as well as their interactions and quadratic terms, on the response variable: the concentration of extracted methylxanthines (mg g−1 of sample). The graph is organized such that the horizontal axis displays the standardized effects corresponding to each term in the model, thus enabling a direct comparison of their relative impact. The vertical axis identifies the individual factors, interaction terms and quadratic terms.
| Variable | Sum of squares | F-Value | p-Value |
|---|---|---|---|
| A: %EtOH | 107.12 | 2060.63 | 0.00 |
| B: temperature (°C) | 0.11 | 2.17 | 0.17 |
| C: cycle | 0.00 | 0.04 | 0.85 |
| D: amplitude (%) | 0.00 | 0.00 | 0.99 |
| AA | 37.71 | 725.34 | 0.00 |
| AB | 0.30 | 5.69 | 0.03 |
| AC | 0.25 | 4.90 | 0.05 |
| AD | 0.00 | 0.08 | 0.78 |
| BB | 0.13 | 2.54 | 0.14 |
| BC | 0.02 | 0.38 | 0.55 |
| BD | 0.00 | 0.00 | 0.97 |
| CC | 0.00 | 0.05 | 0.83 |
| CD | 0.00 | 0.00 | 0.97 |
| DD | 0.06 | 1.19 | 0.30 |
| Total error | 0.62 |
The red vertical line denotes the threshold of statistical significance with a 95% confidence level. The effects whose standardized value exceeds this limit are considered statistically significant, suggesting that they exert a real, rather than random, influence on the response variable.
The analysis of the diagram indicates a negative correlation between the percentage of ethanol (%EtOH) and the extraction of methylxanthines. This suggests that, within the experimental range evaluated, the concentration of extracted methylxanthines is higher when lower percentages of ethanol are used. The quadratic interaction of %EtOH also exerts a negative effect, thereby confirming that the relationship is not strictly linear. Consequently, it can be deduced that the maximum extraction value is not attained at the lower end of the %EtOH range under consideration.
The interaction between %EtOH and temperature was statistically significant (p = 0.034), surpassing the established threshold and indicating a positive synergistic effect beyond what would be expected additively.
A three-dimensional response surface (Fig. 5) was constructed based on the BBD–RSM design to further evaluate the interactions between variables. This representation facilitates the clear visualization of the combined effect of ethanol percentage (%EtOH) and extraction temperature on the concentration of extracted methylxanthines (mg g−1).
 | ||
| Fig. 5 Estimated response surface of the influence of %EtOH and temperature on methylxanthine concentration. | ||
The surface reveals an optimal plateau-shaped area, indicating a positive interaction between both variables at intermediate values, consistent with the negative effect of the quadratic term of %EtOH. The maximum concentrations were achieved with %EtOH ranging from 65 to 70% and a temperature close to 50 °C. The optimal conditions that have been predicted are presented in Table 4.
| Factor | Low | High | Optimal |
|---|---|---|---|
| %EtOH | 60 | 100 | 67.81 |
| Temperature (°C) | 30 | 70 | 50.10 |
| Cycle (s−1) | 0.3 | 0.9 | 0.3 |
| Amplitude (%) | 20 | 50 | 36.02 |
It is important to note that the optimal cycle corresponds to the lower limit of the evaluated range, coinciding with the minimum capacity of the equipment, and was not a significantly influential variable in the extraction of methylxanthines (p > 0.05). Conversely, the optimal values for temperature and %EtOH are within the evaluated ranges, thus rendering further expansion unnecessary in future optimizations.
Time optimization
After the establishment of the optimal conditions for UAE, a study was conducted to evaluate the effect of extraction time on methylxanthine yield. To achieve this objective, the work was conducted under the previously optimized conditions, utilizing time intervals ranging from 5 to 30 minutes (Fig. 6). | ||
| Fig. 6 Evaluation of extraction time for methylxanthines (mg g−1 sample). Different letters indicate significant differences at the 95% confidence level. | ||
The concentration of methylxanthines exhibited mean values ranging from 13.02 to 13.55 mg g−1, indicative of a relatively consistent overall trend over the observed period. However, statistical analysis using ANOVA (p = 0.019) confirmed the existence of significant differences between certain intervals.
Following a period of 10 minutes, the concentration stabilized, indicating that the extraction process had reached equilibrium. The present study demonstrates that an increase in the duration of the extraction process results in elevated levels of energy consumption and thermal stress, without concomitant improvement in yield. It is therefore concluded that a duration of 10 minutes is optimal in achieving balanced efficiency, sustainability, and performance.
Repeatability and intermediate precision
Subsequent to the determination of the optimal conditions for the extraction of methylxanthines from cocoa, a validation study was conducted to evaluate the repeatability (intra-day variability) and intermediate precision (inter-day variability) of the optimized method (Table 5). To ensure the reliability of the findings, nine extractions (n = 9) were carried out on the same day under the established conditions. In the case of intermediate precision, a total of 27 extractions were performed over a period of three days (n = 9 + 9 + 9). The coefficient of variation (C.V.) was utilized as a statistical reference parameter to ascertain the precision of the method.30| Repeatability (n = 9) | Intermediate precision (n = 27) | |
|---|---|---|
| mg methylxanthines per g sample | 13.79 | 13.92 |
| Standard deviation | 0.14 | 0.18 |
| C.V. (%) | 1.01 | 1.27 |
The C.V. values (1.01% and 1.27%, respectively) were well below the 5% threshold considered acceptable in chemical analysis, thus confirming the high precision of the method developed. The findings indicate that the procedure exhibits minimal intraday and interday variability, thereby substantiating its reliability and robustness for the quantification of methylxanthines in cocoa samples under controlled and reproducible conditions.
This optimization study yielded an average concentration of 13.92 ± 0.18 mg methylxanthines per gram of sample. Several studies have been conducted that have explored the potential for the recovery of methylxanthines from cocoa matrices by means of alternative methods. Maldonado et al. employed microwave-assisted extraction (MAE) utilizing 73% methanol at 67 °C for 56 minutes, a process that necessitated more severe conditions to ensure the optimal recovery of theobromine.31 In contrast, the UAE method utilized in this study offers a more efficient alternative by reducing temperature, time, and solvent consumption, while promoting the use of ethanol as a more sustainable option.
Pagliari et al. applied pressurized hot water extraction (PHWE) (69 bar, 7.5% ethanol, 3 cycles of 4 min) to successfully extract theobromine and caffeine at elevated temperatures (>90 °C).32 It is evident that alternative approaches, such as the solid–liquid extraction methods evaluated by Nguyen et al. (70% ethanol, 90 min) or the Soxhlet extraction performed by José González et al. (90% ethanol, 20 h), require a longer duration and larger solvent volumes, thus rendering them less efficient than UAE.33,34 It is important to note that the matrices used in these studies differ from the one employed in the present research, which limits the possibility of direct comparisons. In addition, there is a paucity of literature specifically focusing on the direct extraction of methylxanthines from cocoa. This highlights the need for further research into more sustainable and efficient extraction strategies to optimize the use of this valuable resource.
Applying the method to real samples
To optimize the extraction method, a homogeneous matrix was prepared by mixing three unsweetened 100% cocoa powders (C-1, C-2 and C-3), selected to represent the compositional space of the cocoa powder products analyzed. Following the determination of the optimal extraction conditions, the method was applied to six commercial samples to evaluate their methylxanthine content, with each extraction being performed in triplicate (Fig. 7).Samples C-1, C-2 and C-3 exhibited the highest methylxanthine contents, while an additional compound, identified as vanillin, was detected in C-4. In view of the elevated methylxanthine content and the compositional heterogeneity, it was decided that samples C-1, C-2 and C-4 should be subjected to further in vitro digestion studies.
From a translational perspective, the optimized UAE conditions rely on an ethanol–water system and moderate temperature with a short extraction time (10 min), which are consistent with mild, scalable processing. These features support the potential use of UAE to obtain methylxanthine-enriched cocoa extracts suitable for subsequent solvent removal, concentration, and standardization steps required in functional ingredient or nutraceutical manufacturing. While formulation and encapsulation were not addressed here, the process parameters established provide a practical starting point for developing cocoa-derived methylxanthine ingredients.
In vitro digestion
A static in vitro digestion model was employed to assess the bioaccessibility of methylxanthines, which is defined as the fraction of the ingested compound that is potentially absorbable. The digestion process was simulated in accordance with the INFOGEST 2.0 protocol, with aliquots collected from the oral, gastric, and intestinal phases subsequently analyzed using HPLC. The residual solid was then extracted via ultrasonication to determine the remaining methylxanthine content.Overall recovery (%) was determined by the summation of the maximum release observed in any phase, with this value then being added to the percentage of residual solid (remainder (%)). Values approaching 100% indicate complete recovery of the initial compound, whereas lower values indicate partial degradation. Values exceeding 100% should be regarded as quantitative artefacts (e.g. matrix effects, analytical variability and/or co-elution) and must not be interpreted as net formation of additional methylxanthines. Instead, a qualitative interpretation is proposed, whereby the results are understood to signify an enhancement in apparent extractability/release in subsequent phases under gastrointestinal-like conditions. Under INFOGEST conditions, changes in pH, ionic strength and enzyme activity can promote the dissociation of methylxanthines from the cocoa matrix and modify their partitioning between the digestive fluids and the solid residue, contributing to the observed phase-dependent release patterns and, in some formulations, to elevated apparent totals.
Among the unsweetened 100% cocoa powders (C-1 and C-2), methylxanthine bioaccessibility was clearly phase-dependent and reflected formulation-/matrix-related differences. In C-1 (defatted/alkalized cocoa powder), methylxanthine release increased gradually across digestion, reaching a maximum in the intestinal phase (46.19%), with a cumulative bioaccessibility of 86.13%, suggesting incomplete release under the simulated gastrointestinal conditions. In contrast, C-2 (soluble cocoa powder containing cocoa butter and alkalization-related additives) showed the highest intestinal-phase release (56.67%) and an apparent cumulative bioaccessibility above 100% (113.04%), which is indicative of matrix-driven effects and/or analytical overestimation rather than true physiological release. In the cocoa-based drinking chocolate mix (C-4), which contains sugar/starch and flavorings, high apparent release was observed in all phases (oral: 52.29%, gastric: 78.48%, intestinal: 70.83%), resulting in a cumulative bioaccessibility of 110.45%.
Furthermore, interactions with compounds such as vanillin have been shown to have a substantial impact on the release and quantification of the substance.35–37 Overall, these results (Table 6) support that both digestion phase and matrix composition substantially modulate methylxanthine bioaccessibility, and that specific formulation components may affect not only release behaviour but also analytical quantification.
| Code | Oral phase (%) | Gastric phase (%) | Intestinal phase (%) | % Remainder | Overall recovery (%) |
|---|---|---|---|---|---|
| C-1 | 13.68 | 25.93 | 46.19 | 39.94 | 86.13 |
| C-2 | 16.25 | 29.51 | 56.67 | 56.37 | 113.04 |
| C-4 | 52.29 | 78.48 | 70.83 | 31.97 | 110.45 |
Evaluation of antioxidant activity in vitro
In the present study, the bioaccessibility of methylxanthines and the antioxidant capacity of extracts were evaluated under both pre- and post-digestion conditions. The antioxidant activity exhibited by cocoa is predominantly attributable to the presence of polyphenols and other constituents. Consequently, the trends observed in the DPPH assay should be interpreted as reflecting the combined release and solubilization of antioxidant compounds during the digestive process, rather than the effect of methylxanthines alone.This approach facilitates the evaluation of the impact of the digestive process on the release, stability, and potential availability of bioactive compounds, as well as their antioxidant potential, thereby providing a comprehensive understanding of the availability of these components and their potential physiological effects following consumption.
Prior to in vitro digestion, the assays demonstrated high percentages of free radical inhibition, with DPPH values between 63 and 85% (corresponding to C-2 and C-4), indicating a significant antioxidant potential in the cocoa samples before digestion. The antioxidant capacity of the selected samples (C-1, C-2 and C-4) was evaluated after the simulated digestive process was completed, to assess the impact of extreme digestive conditions (such as pH variations and enzymatic activity) on the stability and effectiveness of the present antioxidant compounds. Due to the limited remaining sample amount, only one antioxidant activity assay could be applied. The DPPH method was chosen because of its simplicity and reproducibility in experiments.
Post-digestion antioxidant capacity assessed using DPPH showed a clear phase-dependent pattern (Table 7). For C-1, DPPH values increased from 11.42 ± 0.41 mg Trolox eq. per g in the oral phase to 22.73 ± 0.27 mg Trolox eq. per g in the gastric phase and 41.65 ± 3.33 mg Trolox eq. per g in the intestinal phase. A similar trend was observed for C-2, which increased from 38.52 ± 3.47 to 71.35 ± 1.68 and 137.04 ± 0.55 mg Trolox eq. per g in oral, gastric and intestinal phases, respectively, indicating a progressive increase in measured antioxidant capacity as digestion advanced. Although the release of methylxanthines has been observed to coincide with the process of matrix disintegration, the observed changes in DPPH are not attributable to methylxanthines alone. Instead, these changes are likely to reflect the digestion-driven behavior of multiple antioxidant constituents, particularly polyphenols.
| Code | Pre-digestion (mg Trolox eq. per g sample) | Post-digestion (mg Trolox eq. per g sample) | ||
|---|---|---|---|---|
| Oral | Gastric | Intestinal | ||
| C-1 | 81.91 ± 1.19 | 11.42 ± 0.41 | 22.73 ± 0.27 | 41.65 ± 3.33 |
| C-2 | 85.41 ± 0.38 | 38.52 ± 3.47 | 71.35 ± 1.68 | 137.04 ± 0.55 |
| C-3 | 85.03 ± 0.94 | — | — | — |
| C-4 | 63.61 ± 3.89 | 41.64 ± 4.02 | 65.00 ± 2.35 | 60.07 ± 1.28 |
| C-5 | 81.15 ± 4.74 | — | — | — |
| C-6 | 81.52 ± 1.68 | — | — | — |
For C-4, values remained comparatively high across phases, peaking in the gastric phase. The lower oral-phase values likely reflect the short residence time and dilution effects upon addition of simulated fluids, whereas the subsequent increases in the gastric and intestinal phases are consistent with digestion-driven release/solubilization of antioxidant constituents and matrix disintegration under changing pH and enzymatic conditions. Overall, these results support the idea that gastrointestinal digestion can enhance the bioaccessibility of antioxidant compounds, leading to higher measured antioxidant capacity in later phases.
Conclusions
In this research, an optimized ultrasound-assisted extraction method for extracting and quantifying total methylxanthines (caffeine and theobromine) from cocoa samples has been successfully developed. This method proved highly efficient, providing excellent extraction yields while remaining simple and environmentally friendly. In addition to improving methylxanthine recovery under food-grade solvent conditions, the optimized UAE parameters provide a methodological basis for obtaining methylxanthine-enriched cocoa extracts; however, additional studies are required before translation into nutraceutical/functional ingredients (e.g., stability during concentration/drying, sensory aspects, and regulatory considerations).Following the standardized simulated digestion protocol (INFOGEST 2.0), the bioaccessibility of the methylxanthines was estimated by comparing their concentrations with the initial values. The results showed recovery values ranging from 46% to 70%, depending on the sample analyzed. High antioxidant capacity was observed in the pre-digestion phase.
Regarding antioxidant properties, the INFOGEST results revealed a phase-dependent modulation rather than a uniform decline: although DPPH values were lower in the oral phase, measured antioxidant capacity increased in the gastric and/or intestinal phases, consistent with digestion-driven release and enhanced bioaccessibility of antioxidant compounds as digestion progressed. These findings highlight the importance of considering the sequential gastrointestinal environment when evaluating the functional potential of cocoa-derived bioactives.
The advantages of UAE, including short extraction times, operational simplicity and high efficiency, make this approach highly promising for the extraction and quantification of methylxanthines in cocoa. Overall, these results provide methodological and mechanistic support for future research on cocoa-derived formulations and for more detailed evaluation of bioactive behavior during gastrointestinal digestion.
Importantly, the present findings are restricted to cocoa powder-based products and cocoa-based drinking chocolate mixes; extrapolation to other cocoa-derived categories (e.g., cocoa liquor/cocoa mass, dark, milk, or white chocolate) should not be made without further targeted investigation.
Author contributions
Conceptualization: A.-G. M. J., C. C. and C. N.; methodology: A.-G. M. J. and C. C.; software: A.-G. M. J.; validation: A.-G. M. J., C. C. and C. N.; formal analysis: F. B. G., P.-L. M. and M. F. A.; investigation: R.-M. L.; data curation, A.-G. M. J. and C. N.; writing – original draft preparation: R.-M. L.; writing – review and editing: A.-G. M. J. and C. N.; supervision: C. C.; project administration: A.-G. M. J. and C. N.; funding acquisition: C. N. All authors read and approved the submission of the final manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo05193b.Acknowledgements
The authors are thankful for the financial support from Programa Operativo FEDER Andalucía 2021-2027 and Consejería de Universidad, Investigación e Innovación, Junta de Andalucía (Proyecto FEDER-UCA-2024-A2-49).References
- P. A. de Souza, L. F. Moreira, D. H. A. Sarmento and F. B. da Costa, Cacao—Theobroma cacao, in Exotic Fruits, ed. S. Rodrigues, E. de Oliveira Silva and E. S. de Brito, Academic Press, London, 2018, pp. 69–76 Search PubMed.
- X. Argout, J. Salse, J.-M. Aury, M. J. Guiltinan, G. Droc, J. Gouzy, M. Allegre, C. Chaparro, T. Legavre, S. N. Maximova, M. Abrouk, F. Murat, O. Fouet, J. Poulain, M. Ruiz, Y. Roguet, M. Rodier-Goud, J. F. Barbosa-Neto, F. Sabot, D. Kudrna, J. S. S. Ammiraju, S. C. Schuster, J. E. Carlson, E. Sallet, T. Schiex, A. Dievart, M. Kramer, L. Gelley, Z. Shi, A. Bérard, C. Viot, M. Boccara, A. M. Risterucci, V. Guignon, X. Sabau, M. J. Axtell, Z. Ma, Y. Zhang, S. Brown, M. Bourge, W. Golser, X. Song, D. Clement, R. Rivallan, M. Tahi, J. M. Akaza, B. Pitollat, K. Gramacho, A. D'Hont, D. Brunel, D. Infante, I. Kebe, P. Costet, R. Wing, W. R. McCombie, E. Guiderdoni, F. Quetier, O. Panaud, P. Wincker, S. Bocs and C. Lanaud, The genome of Theobroma cacao, Nat. Genet., 2011, 43, 101–108 CrossRef CAS PubMed.
- W. Food and Agriculture, World Food and Agriculture – Statistical Yearbook 2024, FAO, 2024.
- International Cocoa Organization, Cocoa Statistics – November 2024 Quarterly Bulletin of Cocoa Statistics, https://www.icco.org/november-2024-quarterly-bulletin-of-cocoa-statistics/, (accessed 17 February 2025).
- T. Aniszewski, in Alkaloids, Chemistry, Biology, Ecology, and Applications, ed. T. Aniszewski, Elsevier, Amsterdam, Elsevier, 2nd edn, 2015, ch. 1, pp. 1–97 Search PubMed.
- L. Bartella, L. Di Donna, A. Napoli, C. Siciliano, G. Sindona and F. Mazzotti, A rapid method for the assay of methylxanthines alkaloids: Theobromine, theophylline and caffeine, in cocoa products and drugs by paper spray tandem mass spectrometry, Food Chem., 2019, 278, 261–266 CrossRef CAS PubMed.
- Y. He, X. Shen, J. Zhu, L. Zhang, X. Wang, T. Zhou, J. Zhang, W. Li and X. Fan, A comprehensive review of caffeine population pharmacokinetics in preterm infants: Factors affecting clearance, Eur. J. Pharm. Biopharm., 2025, 208, 114659 CrossRef PubMed.
- R. Moratalla, Neurobiología de las metilxantinas, Trastor. Adict., 2008, 10, 201–207 CrossRef.
- A. Smith, Effects of caffeine on human behavior, Food Chem. Toxicol., 2002, 40, 1243–1255 CrossRef CAS PubMed.
- P. P. Perissinotti, M. C. S. Rivero-Echeto, L. Kargieman, F. Correa, N. Rodríguez-Leone, S. Junge, S. Villalba, M. A. Bernardi, V. Bisagno and F. J. Urbano, in Natural Molecules in Neuroprotection and Neurotoxicity, Elsevier, 2024, pp. 801–829 Search PubMed.
- M. Zhang, H. Zhang, L. Jia, Y. Zhang, R. Qin, S. Xu and Y. Mei, Health benefits and mechanisms of theobromine, J. Funct. Foods, 2024, 115, 106126 CrossRef CAS.
- I. Cova, V. Leta, C. Mariani, L. Pantoni and S. Pomati, Exploring cocoa properties: is theobromine a cognitive modulator?, Psychopharmacology, 2019, 236, 561–572 CrossRef CAS PubMed.
- H.-S. Afzal, A.-M. Uttra, S. Qasim, A. Malik and A. Mobashar, Theobromine as a multi-target therapeutic agent: Analgesic, anti-inflammatory, and anti-arthritic potential with network pharmacology insights, Ann. Pharm. Fr., 2025, 83, 1114–1129 CrossRef CAS PubMed.
- V. Dinesh, D. R. Varshini, A. A. Mohanan and V. T. Amrithanand, Navigating the storm: A case of severe theophylline toxicity and its management, Med. Rep., 2025, 12, 100191 Search PubMed.
- Y. W. Hartati, M. I. H. L. Zein, A. U. Ibrahim, C. Y. Kharismasari, R. S. Syafira and J. Irkham, Gunlazuardi and P. K. Jiwanti, Advanced aptamer-based sensors for monitoring theophylline, Clin. Chim. Acta, 2025, 571, 120200 CrossRef CAS PubMed.
- C. Heiss, P. Kleinbongard, A. Dejam, S. Perré, H. Schroeter, H. Sies and M. Kelm, Acute Consumption of Flavanol-Rich Cocoa and the Reversal of Endothelial Dysfunction in Smokers, J. Am. Coll. Cardiol., 2005, 46, 1276–1283 CrossRef CAS PubMed.
- H. D. Sesso, J. E. Manson, A. K. Aragaki, P. M. Rist, L. G. Johnson, G. Friedenberg, T. Copeland, A. Clar, S. Mora, M. V. Moorthy, A. Sarkissian, W. R. Carrick and G. L. Anderson, Effect of cocoa flavanol supplementation for the prevention of cardiovascular disease events: the COcoa Supplement and Multivitamin Outcomes Study (COSMOS) randomized clinical trial, Am. J. Clin. Nutr., 2022, 115, 1490–1500 CrossRef CAS PubMed.
- V. Socci, D. Tempesta, G. Desideri, L. De Gennaro and M. Ferrara, Enhancing Human Cognition with Cocoa Flavonoids, Front. Nutr., 2017, 4, 264265 Search PubMed.
- I. Y. Dimeji, K. S. Abass, H. L. Jabba, N. Murtala, S. W. Adeoye and Z. M. Baba, Cocoa flavonoids: A natural approach to regulating metabolic health and combating metabolic syndrome, Food Biosci., 2025, 71, 107210 CrossRef CAS.
- A. Oñatibia-Astibia, R. Franco and E. Martínez-Pinilla, Health benefits of methylxanthines in neurodegenerative diseases, Mol. Nutr. Food Res., 2017, 61(6), 1600670 CrossRef.
- J. W. Kim, M. S. Byun, D. Yi, J. H. Lee, S. Y. Jeon, G. Jung, H. N. Lee, B. K. Sohn, J.-Y. Lee, Y. K. Kim, S. A. Shin, C.-H. Sohn and D. Y. Lee, Coffee intake and decreased amyloid pathology in human brain, Transl. Psychiatry, 2019, 9, 270 CrossRef PubMed.
- D. Morales, A. Iriondo-DeHond and S. Fernández-Tomé, Application of the INFOGEST 2.0 standardized method to study the behavior of phenolic compounds throughout gastrointestinal digestion, Food Chem., 2025, 492, 145531 CrossRef CAS PubMed.
- A. Mackie and N. Rigby, InfoGest Consensus Method, in The Impact of Food Bioactives on Health: In Vitro and Ex Vivo Models, ed. K. Verhoeckx, P. Cotter, I. López-Expósito, C. Kleiveland, T. Lea, A. Mackie, T. Requena, D. Swiatecka and H. Wichers, Springer International Publishing, Cham, 2015, pp. 13–22 Search PubMed.
- S. L. C. Ferreira, R. E. Bruns, H. S. Ferreira, G. D. Matos, J. M. David, G. C. Brandão, E. G. P. da Silva, L. A. Portugal, P. S. dos Reis, A. S. Souza and W. N. L. dos Santos, Box-Behnken design: An alternative for the optimization of analytical methods, Anal. Chim. Acta, 2007, 597, 179–186 CrossRef CAS.
- N. Aslan and Y. Cebeci, Application of Box–Behnken design and response surface methodology for modeling of some Turkish coals, Fuel, 2007, 86, 90–97 CrossRef CAS.
- T.-F. Zhang, J.-F. Yang and D. K. J. Lin, Small Box–Behnken design, Stat. Probab. Lett., 2011, 81, 1027–1033 CrossRef.
- J. Grzelczyk, P. Fiurasek, A. Kakkar and G. Budryn, Evaluation of the thermal stability of bioactive compounds in coffee beans and their fractions modified in the roasting process, Food Chem., 2022, 387, 132888 CrossRef CAS PubMed.
- N. N. Batista, D. P. de Andrade, C. L. Ramos, D. R. Dias and R. F. Schwan, Antioxidant capacity of cocoa beans and chocolate assessed by FTIR, Food Res. Int., 2016, 90, 313–319 CrossRef CAS PubMed.
- R. Martínez, P. Torres, M. A. Meneses, J. G. Figueroa, J. A. Pérez-Álvarez and M. Viuda-Martos, Chemical, technological and in vitro antioxidant properties of cocoa (Theobroma cacao L.) co-products, Food Res. Int., 2012, 49, 39–45 CrossRef.
- J. W. Bartlett and C. Frost, Reliability, repeatability and reproducibility: analysis of measurement errors in continuous variables, Ultrasound Obstet. Gynecol., 2008, 31, 466–475 CrossRef CAS PubMed.
- Y. E. Maldonado and J. G. Figueroa, Microwave-Assisted Extraction Optimization and Effect of Drying Temperature on Catechins, Procyanidins and Theobromine in Cocoa Beans, Molecules, 2023, 28, 3755 CrossRef CAS PubMed.
- S. Pagliari, R. Celano, L. Rastrelli, E. Sacco, F. Arlati, M. Labra and L. Campone, Extraction of methylxanthines by pressurized hot water extraction from cocoa shell by-product as natural source of functional ingredient, LWT, 2022, 170, 114115 CrossRef CAS.
- V. T. Nguyen and N. H. Nguyen, Proximate Composition, Extraction, and Purification of Theobromine from Cacao Pod Husk (Theobroma Cacao L.), Technologies, 2017, 5, 14 CrossRef.
- J. González, M. Monan, J. Perez, E. Gómez, D. D. L. C. Salgado and D. Pérez, Determination of Theobromine and Caffeine in Theobroma cacao Husk from Ethanolic Extract by GC-MS after CC Separation, OALib J., 2019, 06, 1–9 Search PubMed.
- X. Chen, M. Wei, H. Zhang, C. Luo, Y. Chen and Y. Chen, Effect of vanillin and ethyl vanillin on cytochrome P450 activity in vitro and in vivo, Food Chem. Toxicol., 2012, 50, 1897–1901 CrossRef CAS PubMed.
- C. H. Risner and M. J. Kiser, High-performance liquid chromatography procedure for the determination of flavor enhancers in consumer chocolate products and artificial flavors, J. Sci. Food Agric., 2008, 88, 1423–4130 CrossRef CAS.
- N. J. Walton, M. J. Mayer and A. Narbad, Vanillin, Phytochemistry, 2003, 63, 505–515 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



