Open Access Article
Open Access ArticleResting-state brain connectivity following extra virgin olive oil intake in healthy adults: a randomised crossover pilot neuroimaging substudy
Rocío M.
Gutiérrez-Romero
 ab,
Carolina
Donat-Vargas
ab,
Carolina
Donat-Vargas
 abcd,
Emma
Muñoz-Moreno
abcd,
Emma
Muñoz-Moreno
 e,
Alejandro
Hinojosa-Moscoso
e,
Polina
Galkina
e,
Alejandro
Hinojosa-Moscoso
e,
Polina
Galkina
 abf,
Camila
Arancibia-Riveros
abf,
Camila
Arancibia-Riveros
 agh,
Inés
Domínguez-López
agh,
Inés
Domínguez-López
 bfi,
Sara
Hurtado-Barroso
bfi,
Sara
Hurtado-Barroso
 fj,
Maria
Pérez
fj,
Maria
Pérez
 abf,
Anna
Vallverdú-Queralt
abf,
Anna
Vallverdú-Queralt
 abf,
Rosa
Casas
abf,
Rosa
Casas
 bfk,
Ramón
Estruch
bfk,
Ramón
Estruch
 bfk and
Rosa M.
Lamuela-Raventós
bfk and
Rosa M.
Lamuela-Raventós
 *abf
*abf
aPolyphenol Research Group, Department of Nutrition, Food Sciences and Gastronomy, Faculty of Pharmacy and Food Sciences, University of Barcelona (UB), 08028 Barcelona, Spain. E-mail: rogutierrez@ub.edu; carolina.donat@ub.edu; polinagalkina@ub.edu; mariaperez@ub.edu; avallverdu@ub.edu; lamuela@ub.edu
bInstitut de Nutrició i Seguretat Alimentària (INSA), University of Barcelona (UB), 08921 Santa Coloma de Gramanet, Spain
cEpidemiology and Public Health Networking Biomedical Research Centre (CIBERESP), Instituto de Salud Carlos III, 28029 Madrid, Spain
dUnit of Cardiovascular and Nutritional Epidemiology, Institute of Environmental Medicine, Karolinska Institutet, 17177 Stockholm, Sweden
eMagnetic Resonance Imaging Core Facility, Hospital Clínic, Institut d'Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), 08036 Barcelona, Spain. E-mail: emunozm@recerca.clinic.cat; ahinojosa@recerca.clinic.cat
fCIBER de Fisiopatología de la Obesidad y Nutrición (CIBEROBN), Instituto de Salud Carlos III, 28029 Madrid, Spain
gInstitut de Recerca Sant Joan de Déu, 08950 Barcelona, Spain. E-mail: camila.arancibia@sjd.es
hEndocrinology Department, Hospital Sant Joan de Déu, 08950 Barcelona, Spain
iDepartment of Molecular Epidemiology, German Institute of Human Nutrition Potsdam-Rehbruecke, 14558 Nuthetal, Germany. E-mail: ines.dominguez-lopez@dife.de
jDepartment of Biomedical Science, Faculty of Medicine and Health Sciences, Universitat Internacional de Catalunya, 08195 Sant Cugat del Vallès, Spain. E-mail: shurtado@uic.es
kDepartment of Internal Medicine, Hospital Clínic, Institut d'Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS), University of Barcelona (UB), 08036 Barcelona, Spain. E-mail: rcasas1@recerca.clinic.cat; restruch@clinic.cat
First published on 31st March 2026
Abstract
This pilot randomised crossover neuroimaging substudy (n = 9) identified increased resting-state occipital functional connectivity following daily consumption of (poly)phenol-rich extra virgin olive oil compared with regular olive oil. Urinary hydroxytyrosol-glucuronide excretion increased significantly, and a significant interaction between intervention and metabolite levels was observed on occipital connectivity. These preliminary findings require replication in larger, adequately powered studies.
Introduction
Brain function and connectivity can be influenced by dietary factors, including foods rich in (poly)phenols. Previous evidence suggests that (poly)phenol-rich diets, such as the Mediterranean diet (MD), are associated with enhanced cognitive performance and brain connectivity, particularly in older adults.1,2Extra-Virgin Olive Oil (EVOO) is one of the main dietary sources of (poly)phenols within the MD. Unlike regular olive oil (OO), EVOO retains its antioxidant phenolic compounds due to natural minimal processing.3,4 Previous research has shown that greater adherence to MD is associated with stronger functional brain connectivity, especially in older adults.5
EVOO phenolic compounds as a whole exhibit strong antioxidants and anti-inflammatory properties. These bioactives, including oleuropein, hydroxytyrosol (HT), and related secoiridoids, neutralize reactive oxygen species, reduce oxidative damage to lipids, proteins, and DNA, and modulate signalling pathways involved in neuroinflammation and synaptic plasticity.6–8 Moreover, recent epidemiological evidence indicates that regular EVOO consumption is associated with reduced risk of cognitive decline and dementia, reinforcing its role in brain protection within the MD framework.9,10
Among these compounds, HT is particularly relevant due to its potent antioxidant and neuroprotective activity. Importantly, HT is rapidly metabolised into HT-glucuronide, which is widely recognised as a validated biomarker of EVOO intake.11
Despite growing evidence linking (poly)phenol-rich dietary patterns with cognitive and neurovascular outcomes, no previous study, to our knowledge, has evaluated the effects of EVOO consumption on human brain connectivity using resting-state functional magnetic resonance imaging (rs-fMRI). While prior research has primarily focused on older populations, examining EVOO intake in healthy young adults may help detect early alterations in functional connectivity before age-related decline occurs.
This pilot randomised crossover trial was designed to explore whether daily EVOO intake is associated with differences in resting-state functional connectivity in healthy young adults. We combined rs-fMRI with urinary biomarker analysis to assess changes in brain connectivity in parallel with (poly)phenol metabolites. We hypothesised that short-term consumption of high-(poly)phenol EVOO, compared with low-(poly)phenol olive oil, would induce measurable differences in resting-state connectivity patterns.
Materials and methods
Study design and participants
This study is based on data from the HEVOOC trial (NCT05898113), a randomised, crossover clinical study aimed to evaluate the effects of daily consumption of ‘Corbella’ EVOO compared to regular OO in healthy young adults (n = 28).Sample size was calculated based on the brain-derived neurotrophic factor (BDNF) variable using https://www.surveysystem.com/sscalc.htm for paired comparisons, with a significance level of 5% and 80% power, considering an expected dropout rate of 20%.
Participants were randomly allocated to one of two intervention sequences: (1) EVOO followed by regular olive oil (OO), or (2) the reverse sequence, with a 4-week washout period between phases. Each intervention phase lasted 4 weeks, during which participants consumed either EVOO or OO at a dose of 0.7 g per kg body weight per day, containing 227.7 mg kg−1 and 12.3 mg kg−1 total (poly)phenols, respectively. Participants were instructed to maintain their habitual diet and incorporate the assigned oil, either raw and/or cooked, at any time of the day.
Prior to the study, a 7-day run-in period was conducted, during which participants replaced EVOO with low-polyphenol olive oil and followed standardised dietary restrictions, avoiding olives, high-(poly)phenol foods (such as coffee, citrus fruits, apples, and berries), and limiting alcohol to a maximum of one glass of wine or beer per week. Participants were recruited in Barcelona, Spain, from June to September 2023, through online and institutional advertisements at Hospital Clínic and the University of Barcelona.
Inclusion criteria were age between 18 and 35 years, BMI < 30 kg m−2, absence of chronic diseases, and non-smokers. Exclusion criteria included food allergies or intolerances, adherence to extreme or restrictive diets, excessive alcohol intake (>30 g day−1 for men, >20 g day−1 for women), pregnancy, menopause, or breastfeeding For the present neuroimaging substudy (n = 9), participants from the HEVOOC cohort underwent rs-fMRI assessments. Participant flow throughout the study is presented in the SI (Fig. S1). All participants provided written informed consent. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Hospital Clínic (HCB/2023/0074) and the University of Barcelona (IRB00003099).
Neuroimaging acquisition and analysis
Image acquisition and analysis were performed at the IDIBAPS Magnetic Resonance Imaging Core Facility. MRI scans were conducted using a 3T Siemens PRISMA scanner and included both T1-weighted structural images (TR = 2.5 s, TE = 4.37 ms, voxel size: 1 × 1 × 1 mm3) and rs-fMRI acquisitions (TR = 800 ms, TE = 37 ms, total volumes = 450; voxel size: 2 × 2 × 2 mm3).The neuroimaging protocol included structural and rs-fMRI, analysed via independent component analysis (ICA) and permutation testing. Preprocessing steps comprised: (1) slice timing correction using FSL (v.6.0.7.1),12 (2) motion correction and detection of motion outliers: motion parameters were estimated using SPM12,13 together with frame displacement (FD) and DVARS computation.14 Motion parameters, FD, and DVARS were included as confound regressors in the BOLD signal estimation; (3) detection of motions outliers: a quality criteria of mean FD <0.5 mm was applied or if more than 10% of volumes with FD >0.5 mm; (4) registration of functional images to the T1-weighted structural image to mitigate EPI distortion using ANTs Registration (v.2.4.4);15 and (5) spatial smoothing with sigma = 3 mm and band-pass filtering between (0.01 and 0.15 Hz) using Nilearn (v.0.10.1).16 After preprocessing, images were registered into the standard MNI space using elastic registration by ANTs. Visual inspection of raw and pre-processed images was performed during the pipeline to ensure image quality.
Urinary (poly)phenol metabolites
Urinary metabolites were measured in 24 hour urine samples using liquid chromatography (Agilent 1290 Infinity II) coupled to high-resolution mass spectrometry (Agilent 6560 Ion Mobility QTOF LC/MS) operated in negative ionization mode. Analyses were performed at the Separation Techniques Unit of the Scientific and Technological Centers (CCiTUB), Universitat de Barcelona. Sample preparation and analysis followed a previously described protocol developed by our research group.17 Based on prior literature, the analysis specifically targeted (poly)phenolic compounds characteristic of EVOO: HT, HT-glucuronide, and HT-sulfate.18,19Covariate assessment
Covariates were selected based on prior knowledge of their potential influence on both (poly)phenol metabolites and resting state brain connectivity.20–24 These included total energy intake, assessed as a continuous variable (kcal day−1) using a validated 151-item food frequency questionnaire (FFQ);25 and adherence to the MD, evaluated using the 17-item MD Adherence Screener (MEDAS),26 which provides a score reflecting higher or lower MD adherence.Objective physical activity was measured with ActiGraph® wGT3X-BT accelerometers (ActiGraph LLC, Pensacola, FL). Each device was programmed with the participant's study ID and worn on the non-dominant wrist continuously for 7 consecutive days prior to each study visit. Data were analysed with ActiLife software (v6.13.4). Moderate-to-vigorous physical activity (MVPA) was defined as ≥1952 counts per minute (cpm), using Freedson adult cut-points,27 and expressed as the average daily minutes above this threshold. All covariates were measured at baseline and adjusted for in statistical models.
Statistical analysis
The neuroimaging analyses were conducted in a small subset of participants as part of a pilot, exploratory substudy. Accordingly, these analyses were intended to generate preliminary insights rather than to provide definitive or generalisable whole-brain inferences.ICA was performed using the MELODIC toolbox within the FSL to identify resting-state brain networks. Dual regression was applied to obtain subject-specific spatial maps and time series. Group-level comparisons were conducted with the FSL randomize tool and threshold-free cluster enhancement (TFCE) to evaluate differences in brain activation across conditions (baseline vs. washout, pre-OO vs. post-OO, and pre-EVOO vs. post-EVOO). Statistical significance for imaging data was set at p < 0.005. Cohen's d-effect was computed voxel-wise and average in the cluster showing significant differences identified using randomise. Bootstrapping was then applied to the mean cluster connectivity in each subject to estimate the corresponding confidence interval.
To evaluate the effects of the dietary interventions on brain connectivity and urinary biomarkers, linear mixed-effects models (LMMs) were used to account for the repeated-measures crossover design. Each model included a random intercept for participant ID to account for within-subject correlation. Model 1 included intervention type, period, and the baseline value of the outcome variable. Model 2 additionally adjusted for baseline total energy intake, Mediterranean diet adherence score, and moderate-to-vigorous physical activity. Associations between urinary HT-glucuronide levels and brain connectivity were examined using models that included an interaction term between metabolite levels and intervention. Model assumptions were assessed by visual inspection of residual Q–Q plots and residuals versus fitted value plots to evaluate normality and homoscedasticity. When normality was not satisfied, variables were log-transformed prior to analysis. Standardised effect sizes (Cohen's d) were calculated for the main intervention effects by dividing the estimated fixed-effect coefficient by the residual standard deviation of the model. All analyses were performed using Stata 17.0 (StataCorp, College Station, TX) and statistical significance was set at p < 0.05.
Results
Participant characteristics
Of the 28 participants enrolled in the HEVOOC randomised clinical trial, nine healthy adults consented to undergo additional neuroimaging assessments and were therefore included in the present exploratory analysis. Baseline characteristics are presented in Table 1. Participants were normotensive, had no cardiometabolic risk factors, and exhibited a moderate adherence to the MD (median score: 10/17).| Characteristics | Participants (n = 9) |
|---|---|
| Data are presented as median [IQR]. Abbreviations: SBP – systolic blood pressure; DBP – diastolic blood pressure; BMI – body mass index; TC – total cholesterol; HDL-C – high-density lipoprotein cholesterol; LDL-C – low-density lipoprotein cholesterol; MVPA – moderate-to-vigorous physical activity; MedDiet – Mediterranean diet adherence score. | |
Sex, male![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) female (n) female (n) |
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
| Age (years) | 31 [27;32] |
| Blood pressure measures | |
| SBP (mmHg) | 112.5 [104; 121] |
| DBP (mmHg) | 73.7 [69; 73] |
| Anthropometric and metabolic parameters | |
| Weight (kg) | 64.1 [58.1; 75.1] |
| BMI (kg m−2) | 23 [21.2; 25.1] |
| Body fat (%) | 24.7 [22.5; 29.4] |
| Muscle mass (%) | 34.1 [29.5; 37.7] |
| Visceral fat (%) | 6 [3; 7] |
| Waist circumference (cm) | 76.5 [73; 84] |
| TC (mg dL−1) | 165 [153; 216] |
| LDL-C (mg dL−1) | 95 [78; 122] |
| HDL-C (mg dL−1) | 52 [48; 56] |
| Questionary of healthy habits | |
| MVPA min day−1 | 303 [276.9; 321.4] |
| MedDiet score | 10 [9; 11] |
| Total energy intake (Kcal day−1) | 2210.6 [2128.2; 2725.1] |
Effects of EVOO on resting-state functional connectivity
Analysis of rs-fMRI data (TFCE-corrected, FSL Randomize; p < 0.005) revealed increased activation in a visual network localised to the left occipital cortex, including its superior and inferior lateral areas, following EVOO intake (Fig. 1a). The average voxel-wise Cohen's d within the significant cluster was 1.058 with a bootstrapped 95% CI [0.147, 0.464] for the mean cluster connectivity change. Fig. 1b displays the average visual network connectivity before and after each intervention, showing notably higher post-EVOO connectivity levels compared to regular OO. Extracted mean connectivity values were analysed using LMMs, functional connectivity was significantly greater following EVOO compared with regular OO consumption in the fully adjusted model (Model 2: β = 0.20; 95% CI: 0.03, 0.37; p = 0.016). Cohen's d = 1.46. Results were consistent in the minimally adjusted model (Table S1).Together, these findings support an association between EVOO intake and increased intrinsic functional connectivity in occipital regions involved in visual processing.
Effect of EVOO on urinary HT-glucuronide and its association with occipital connectivity
Urinary log-transformed HT-glucuronide excretion were significantly higher following EVOO compared with regular OO consumption in the fully adjusted model (β = 1.24; 95% CI: 0.13, 2.35; p = 0.028) Similar estimates were observed in the minimally adjusted model (Table S1). In exploratory analyses, changes in urinary log-transformed HT-glucuronide levels were positively associated with changes in occipital functional connectivity in the fully adjusted model (β = 3.34; 95% CI: −0.02, 6.72; p = 0.052). Estimates from the minimally adjusted model were comparable (Table S1).Interaction between EVOO Intake and HT-glucuronide on brain activity
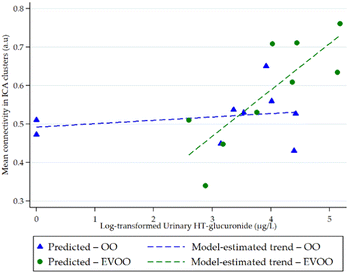
Notably, a significant interaction was observed between urinary HT-glucuronide levels and intervention group on occipital network activation in the fully adjusted model (β = 0.13; 95% CI: 0.05, 0.21; p = 0.002). Similar estimates were obtained in the minimally adjusted model (Model 1: β = 0.14; 95% CI: 0.06, 0.21; p < 0.001), indicating that the interaction effect was robust to additional adjustment. Fig. 2 plots occipital network activation against log-transformed urinary HT-glucuronide levels for each intervention, illustrating that the association between HT-glucuronide and brain activation differed by intervention.Discussion
This exploratory pilot study provides initial evidence that daily intake of 0.7 g per kg body weight of EVOO for one month was associated with increased resting-state functional connectivity within an occipital network, compared with regular, low-(poly)phenol OO. Activation increases were localised to the left occipital cortex, areas involved in visuospatial attention, motion perception (superior), and object recognition (inferior). EVOO consumption also significantly increased urinary HT-glucuronide excretion. Notably, a significant interaction between HT-glucuronide excretion and intervention was observed on occipital functional connectivity, indicating that the relationship between phenolic metabolite levels and visual network connectivity differed according to the intervention.These results complement prior studies associating MD adherence with improved brain connectivity28 but uniquely isolate EVOO's contribution. While previous work identified interesting effects of EVOO consumption on frontoparietal brain regions involved in working memory,20 our findings point to specific modulation of visual cortical networks even in the absence of external stimuli. Activation was specifically localised in the left occipital cortex, a region implicated in visuospatial attention and motion perception in its superior portion, and object recognition in its inferior segment.29,30
Urinary HT-glucuronide increased significantly following EVOO consumption. The observed interaction between HT-glucuronide levels and intervention further supports the hypothesis that EVOO-specific phenolics may play a relevant role in mediating its neurological benefits.
Pharmacokinetic studies show that over 60% of phenolic metabolites detected post-EVOO consumption circulate as conjugates, mainly glucuronides and sulfates.31 While urinary HT-glucuronide is a validated biomarker of EVOO consumption,32 and may contribute to observed effects, it is likely that other co-occurring bioactive compounds in EVOO also play a role in modulating brain connectivity.
In addition to human studies, preclinical evidence supports the neuromodulatory role of EVOO. Recent work in obese rats demonstrated that EVOO supplementation reduced hypothalamic inflammation (arcuate nucleus) and improved metabolic parameters compared to controls suggesting broader neuromodulatory effects across distinct brain regions.33 Although these findings concern different brain regions and mechanistic pathways, they provide biological plausibility for the cerebral effects of EVOO.
Strengths of this study include its nutritional randomised crossover design with a control, fMRI-based neural assessment, phenolic profiling, and within-subject control. The inclusion of healthy young participants helps control age-related variability in brain function. However, the study has limitations. Most notably, the small sample size (n = 9) may limit statistical power and the generalizability of the findings, particularly with respect to the neuroimaging outcomes. Additionally, the relatively short duration of the intervention (one month) may be insufficient to capture long-term neuroplastic changes. While the crossover design improves internal validity by allowing each participant to serve as their own control, replication of these results in larger, adequately powered samples, ideally at least double the current size, is necessary to confirm the robustness and reproducibility of the observed effects.
Conclusions
One month of consuming EVOO (0.7 g per kg body weight per day) was associated with preliminary differences in resting-state functional connectivity and higher urinary excretion of HT-glucuronide in healthy young adults, compared to one month of consuming regular, highly refined OO. The observed interaction between olive oil-derived phenolic metabolites and functional connectivity suggests a potential neuro-modulatory effect of EVOO. However, these findings should be interpreted with caution due to the exploratory nature and small sample size of the neuroimaging substudy. These novel findings warrant further investigation in larger, adequately powered, and long-term studies to confirm the observed associations and to explore the underlying mechanisms of the (poly) phenols present in EVOO.Author contributions
Conceptualization, M. P., A. V.-Q., R. C., R. E. and R. M. L.-R.; methodology, R. M. G.-R., E. M.-M., A. H., P. G., C. A.-R. and I. D.-L.; software, E. M.-M. and A. H.; validation, C. D.-V., I. D.-L. and R. M. L.-R.; formal analysis, R. M. G.-R., C. D.-V., E. M.-M., A. H., P. G., C. A.-R. and I. D.-L.; investigation, R. M. G.-R., E. M.-M., A. H., P. G., C. A.-R. and S. H.-B.; resources, R. C. and R. E.; data curation, R. M. G.-R. and C. D.-V.; writing – original draft, R. M. G.-R.; writing – review & editing, C. D.-V., E. M.-M., A. H., P. G., C. A.-R., S. H.-B., I. D.-L., M. P., A. V.-Q., R. C., R. E. and R. M. L.-R.; visualization, R. M. G.-R.; supervision, R. M. L.-R.; project administration, R. M. L.-R.; funding acquisition, R. E. and R. M. L.-R. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
R. M. L.-R. reported personal fees from Cerveceros de España, UNIDECO, Adventia, Wine in Moderation, and Ecoveritas S. A., all outside the submitted work. R. E. reported grants from the Fundación Dieta Mediterránea and Fundación Cerveza y Salud, as well as personal fees for lectures from Brewers of Europe, Fundación Cerveza y Salud, Instituto Cervantes (Albuquerque, Milan, Tokyo), Pernod Ricard, and the Wine and Culinary International Forum. He also received non-financial support for the organization of a national nutrition congress and for feeding trials with products from Grand Fountain and Uriach Laboratories (Spain). The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.Data availability
The data supporting this article have been included as part of the supplementary information (SI), including Supplementary Dataset 1. Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo05016b.Acknowledgements
This work was supported by CDTI [IDI-20210929], Fundació Bosch-Gimpera [311463], and the grant PID2023-147307OB-I00 funded by MICIU/AEI/10.13039/501100011033 and by ERDF/EU. We would like to thank Oli Migjorn for their collaboration and supplying the Corbella EVOO; Generalitat de Catalunya (GC) [2021-SGR-00334], CIBEROBN from the Instituto de Salud Carlos III (ISCIII) of the Ministerio de Ciencia, and the INSA-UB Unit of Excellence (María de Maeztu CEX2021-001234-M funded by MICIN/AEI/FEDER, UE). R. M. L.-R. would like to thank for the GC the ICREA academia recognition. R. M. G. R. acknowledges the support of the Carlos Antonio López Scholarship Program (BECAL), Paraguay, for doctoral funding. We thank the CCiTUB and staff members for their support and advice on LC-ESI-MS/MS technique. The authors sincerely thank all the volunteers who participated in the HEVOOC study for their time, commitment, and invaluable contribution to this research.References
- W. Yang, K. Cui, X. Li, J. Zhao, Z. Zeng, R. Song, X. Qi and W. Xu, Effect of Polyphenols on Cognitive Function: Evidence from Population-based Studies and Clinical Trials, J. Nutr., Health Aging, 2021, 25, 1190–1204 CrossRef CAS PubMed.
- C. L. Cheatham, D. C. Nieman, A. P. Neilson and M. A. Lila, Enhancing the Cognitive Effects of Flavonoids With Physical Activity: Is There a Case for the Gut Microbiome?, Front. Neurosci., 2022, 16, 833202 CrossRef PubMed.
- C. Donat-Vargas, E. Lopez-Garcia, J. R. Banegas, M. Martínez-González, F. Rodríguez-Artalejo and P. Guallar-Castillón, Only virgin type of olive oil consumption reduces the risk of mortality. Results from a Mediterranean population-based cohort, Eur. J. Clin. Nutr., 2023, 77, 226–234 CrossRef CAS PubMed.
- A. Olmo-Cunillera, M. Pérez, A. López-Yerena, M. M. Abuhabib, A. Ninot, A. Romero-Aroca, A. Vallverdú-Queralt and R. M. Lamuela-Raventós, Oleacein and Oleocanthal: Key Metabolites in the Stability of Extra Virgin Olive Oil, Antioxidants, 2023, 12, 1776 CrossRef CAS PubMed.
- A. M. Gaynor, E. Varangis, S. Song, Y. Gazes, C. Habeck, Y. Stern and Y. Gu, Longitudinal association between changes in resting-state network connectivity and cognition trajectories: The moderation role of a healthy diet, Front. Hum. Neurosci., 2023, 16, 1043423 CrossRef PubMed.
- M. Gonçalves, N. Vale and P. Silva, Neuroprotective Effects of Olive Oil: A Comprehensive Review of Antioxidant Properties, Antioxidants, 2024, 13, 762 CrossRef PubMed.
- A. Boronat, G. Serreli, J. Rodríguez-Morató, M. Deiana and R. de la Torre, Olive Oil Phenolic Compounds’ Activity against Age-Associated Cognitive Decline: Clinical and Experimental Evidence, Antioxidants, 2023, 12, 1472 CrossRef CAS PubMed.
- P. Papadopoulou, A. Polissidis, G. Kythreoti, M. Sagnou, A. Stefanatou and T. C. Theoharides, Anti-Inflammatory and Neuroprotective Polyphenols Derived from the European Olive Tree, Olea europaea L., in Long COVID and Other Conditions Involving Cognitive Impairment, Int. J. Mol. Sci., 2024, 25, 11040 CrossRef CAS PubMed.
- A.-J. Tessier, M. Cortese, C. Yuan, K. Bjornevik, A. Ascherio, D. D. Wang, J. E. Chavarro, M. J. Stampfer, F. B. Hu, W. C. Willett and M. Guasch-Ferré, Consumption of Olive Oil and Diet Quality and Risk of Dementia-Related Death, JAMA Netw. Open, 2024, 7, e2410021 CrossRef PubMed.
- M. Morvaridzadeh, M. Alami, N. Zoubdane, H. Sidibé, H. Berrougui, T. Fülöp, M. Nguyen and A. Khalil, High-Tyrosol/Hydroxytyrosol Extra Virgin Olive Oil Enhances Antioxidant Activity in Elderly Post-Myocardial Infarction Patients, Antioxidants, 2025, 14, 867 CrossRef CAS PubMed.
- I. Domínguez-López, A. López-Yerena, A. Vallverdú-Queralt, M. Pallàs, R. M. Lamuela-Raventós and M. Pérez, From the gut to the brain: the long journey of phenolic compounds with neurocognitive effects, Nutr. Rev., 2024, 83, 533–546 CrossRef PubMed.
- M. Jenkinson, C. F. Beckmann, T. E. J. Behrens, M. W. Woolrich and S. M. Smith, FSL, Neuroimage, 2012, 62, 782–790 CrossRef PubMed.
- Statistical Parametric Mapping: The Analysis of Functional Brain Images, ed. K. J. Friston, J. Ashburner, S. J. Kiebel, T. E. Nichols and W. D. Penny, Academic Press, London, 2006 Search PubMed.
- J. D. Power, A. Mitra, T. O. Laumann, A. Z. Snyder, B. L. Schlaggar and S. E. Petersen, Methods to detect, characterize, and remove motion artifact in resting state fMRI, Neuroimage, 2014, 84, 320–341 CrossRef PubMed.
- A. Abraham, F. Pedregosa, M. Eickenberg, P. Gervais, A. Mueller, J. Kossaifi, A. Gramfort, B. Thirion and G. Varoquaux, Machine learning for neuroimaging with scikit-learn, Front. Neuroinform., 2014, 8, 14 Search PubMed.
- B. B. Avants, N. J. Tustison, G. Song, P. A. Cook, A. Klein and J. C. Gee, A reproducible evaluation of ANTs similarity metric performance in brain image registration, Neuroimage, 2011, 54, 2033–2044 CrossRef PubMed.
- F. M. Campins-Machado, R. Casas, R. M. Lamuela-Raventós, P. Galkina, M.Á Martínez-González, M. Fitó, E. Ros, R. Estruch, I. Domínguez-López and M. Pérez, Microbiota-derived resveratrol metabolites: New biomarkers of red wine consumption are inversely associated with inflammation in a longitudinal study of a Mediterranean population, J. Nutr., Health Aging, 2025, 29, 100542 CrossRef PubMed.
- R. Fuccelli, R. Fabiani and P. Rosignoli, Hydroxytyrosol Exerts Anti-Inflammatory and Anti-Oxidant Activities in a Mouse Model of Systemic Inflammation, Molecules, 2018, 23, 3212 CrossRef PubMed.
- L. Rubió, M. Farràs, R. de La Torre, A. Macià, M.-P. Romero, R. M. Valls, R. Solà, M. Farré, M. Fitó and M.-J. Motilva, Metabolite profiling of olive oil and thyme phenols after a sustained intake of two phenol-enriched olive oils by humans: Identification of compliance markers, Food Res. Int., 2014, 65, 59–68 CrossRef.
- P. Faulkner, P. Allen, A. Costabile, M. H. Schoemaker, F. Imakulata and P. Hepsomali, Greater resting state functional connectivity of the medial prefrontal cortex with the thalamus, caudate, and putamen in individuals who adhere to the Mediterranean style diets, Eur. J. Nutr., 2025, 64, 34 CrossRef PubMed.
- D. E. A. Jensen, K. P. Ebmeier, T. Akbaraly, M. G. Jansen, A. Singh-Manoux, M. Kivimäki, E. Zsoldos, M. C. Klein-Flügge and S. Suri, Association of Diet and Waist-to-Hip Ratio With Brain Connectivity and Memory in Aging, JAMA Netw. Open, 2025, 8, e250171 CrossRef PubMed.
- C. Favari, J. F. Rinaldi de Alvarenga, L. Sánchez-Martínez, N. Tosi, C. Mignogna, E. Cremonini, C. Manach, L. Bresciani, D. Del Rio and P. Mena, Factors driving the inter-individual variability in the metabolism and bioavailability of (poly)phenolic metabolites: A systematic review of human studies, Redox Biol., 2024, 71, 103095 CrossRef CAS PubMed.
- J. J. Pruzin, H. Klein, J. S. Rabin, A. P. Schultz, D. R. Kirn, H. Yang, R. F. Buckley, M. R. Scott, M. Properzi, D. M. Rentz, K. A. Johnson, R. A. Sperling and J. P. Chhatwal, Physical activity is associated with increased resting–state functional connectivity in networks predictive of cognitive decline in clinically unimpaired older adults, Alzheimers Dement, 2022, 14, e12319 Search PubMed.
- E. T. Rolls, R. Feng and J. Feng, Lifestyle risks associated with brain functional connectivity and structure, Hum. Brain Mapp., 2023, 44, 2479–2492 CrossRef PubMed.
- C. Juton, S. Castro-Barquero, R. Casas, T. Freitas, A. M. Ruiz-León, F. Crovetto, M. Domenech, F. Crispi, E. Vieta, E. Gratacós, R. Estruch and H. Schroder, Reliability and Concurrent and Construct Validity of a Food Frequency Questionnaire for Pregnant Women at High Risk to Develop Fetal Growth Restriction, Nutrients, 2021, 13, 1629 CrossRef CAS PubMed.
- H. Schröder, M. Fitó, R. Estruch, M. A. Martínez-González, D. Corella, J. Salas-Salvadó, R. Lamuela-Raventós, E. Ros, I. Salaverría, M. Fiol, J. Lapetra, E. Vinyoles, E. Gómez-Gracia, C. Lahoz, L. Serra-Majem, X. Pintó, V. Ruiz-Gutierrez and M.-I. Covas, A Short Screener Is Valid for Assessing Mediterranean Diet Adherence among Older Spanish Men and Women, J. Nutr., 2011, 141, 1140–1145 CrossRef PubMed.
- P. S. Freedson, E. Melanson and J. Sirard, Calibration of the Computer Science and Applications, Inc. accelerometer, Med. Sci. Sports Exercise, 1998, 30, 777–781 CrossRef CAS PubMed.
- A. M. Gaynor, E. Varangis, S. Song, Y. Gazes, D. Noofoory, R. S. Babukutty, C. Habeck, Y. Stern and Y. Gu, Diet moderates the effect of resting state functional connectivity on cognitive function, Sci. Rep., 2022, 12, 16080 CrossRef CAS PubMed.
- K. Grill-Spector, Z. Kourtzi and N. Kanwisher, The lateral occipital complex and its role in object recognition, Vision Res., 2001, 41, 1409–1422 CrossRef CAS PubMed.
- K. Nagy, M. W. Greenlee and G. Kovács, The lateral occipital cortex in the face perception network: An effective connectivity study, Front. Psychol., 2012, 3, 141 Search PubMed.
- C. Alemán-Jiménez, R. Domínguez-Perles, S. Medina, I. Prgomet, I. López-González, A. Simonelli-Muñoz, M. Campillo-Cano, D. Auñón, F. Ferreres and Á. Gil-Izquierdo, Pharmacokinetics and bioavailability of hydroxytyrosol are dependent on the food matrix in humans, Eur. J. Nutr., 2021, 60, 905–915 CrossRef PubMed.
- C. Santangelo, C. Filesi, R. Varí, B. Scazzocchio, T. Filardi, V. Fogliano, M. D'Archivio, C. Giovannini, S. Morano and R. Masella, Consumption of extra-virgin olive oil rich in phenolic compounds: urinary excretion of glucuronides as biomarkers, J. Endocrinol. Invest., 2016, 39, 1295–1301 CrossRef CAS PubMed.
- A. Peres, R. Dantas, A. Ferreira, A. C. Silveira, G. Raphaelli, L. Felipe, L. Souza, M. Costa, A. Machado, D. Machado, R. Herrmann, J. de Oliveira, C. A. Netto, A. Wyse, C. Dalmaz and R. Bast, Extra virgin olive oil supplementation reduces inflammation in the arcuate nucleus of the hypothalamus and improves metabolic parameters in obese rats, Nutr. Neurosci., 2026, 29, 92–107 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |