Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Metabolomic profiling of urinary phenolic compounds in postmenopausal women after consumption of dark chocolate, green tea, and fruit juice
Lorena
Sánchez-Martínez
ab,
Nicole
Tosi
 c,
Nicola Luigi
Bragazzi
c,
Nicola Luigi
Bragazzi
 c,
Letizia
Bresciani
c,
Letizia
Bresciani
 c,
Daniele
Del Rio
c,
Daniele
Del Rio
 cd,
María Jesús
Periago
*ab,
Pedro
Mena†
cd,
María Jesús
Periago
*ab,
Pedro
Mena†
 cd and
Rocío
González-Barrio
cd and
Rocío
González-Barrio
 *ab
*ab
aDepartment of Food Technology, Food Science and Nutrition, University of Murcia, Campus Mare Nostrum, Campus de Espinardo, 30100 Murcia, Spain. E-mail: rgbarrio@um.es; mjperi@um.es; Tel: +34 868889641
bBiomedical Research Institute of Murcia Pascual Parrilla – IMIB, 30120 Murcia, Spain
cDepartment of Food and Drugs, University of Parma, Parma, Italy
dMicrobiome Research Hub, University of Parma, Parma, Italy
First published on 2nd February 2026
Abstract
Postmenopausal women are approximately twice as likely to develop cardiometabolic diseases (CMDs) as premenopausal women. Plant-based diets rich in fruits and vegetables, due to their high content of bioactive compounds such as (poly)phenols, represent a promising strategy to reduce the risk of CMDs in this population. However, the cardioprotective effects of (poly)phenols depend largely on inter-individual variability, which is strongly influenced by the gut microbiota composition. Menopause is often associated with gut dysbiosis, characterized by a reduced microbial diversity and a lower abundance of beneficial bacteria. This imbalance in the gut microbiota profile of postmenopausal women could influence (poly)phenol metabolism and, consequently, the health benefits attributed to (poly)phenol-rich food (PP-rich food) consumption. Therefore, this study aimed to assess the impact of a daily consumption of PP-rich foods (dark chocolate, green tea and fruit juice) for 2 months on the urinary phenolic profile in postmenopausal women at high cardiometabolic risk. To this end, 116 urinary phenolic metabolites were determined using UHPLC-ESI-QqQ-MS/MS. Dietary intervention with PP-rich foods led to a significant increase in the urinary excretion of phenolic metabolites derived from gut microbiota activity and phase II metabolism. Notably, there was a significant increase in the excretion of glucuronidated and/or sulfated conjugates of phenyl-γ-valerolactones, phenylvaleric acids, phenylacetic acids, benzoic acids, and urolithins. It is expected that these phenolic metabolites could be the bioactive compounds responsible for the potential beneficial effects derived from the daily intake of dark chocolate, green tea, and fruit juice on the reduction of the risk of CMDs in postmenopausal women.
Introduction
The hormonal transition towards menopause is a critical stage in women's lives, involving multiple physiological, psychological, and social changes that can significantly impact their health and quality of life. These changes are driven mainly by a sudden decline in circulating oestrogen levels. Although primarily involved in reproductive processes, oestrogens also play essential roles in several physiological functions, including blood pressure regulation, body fat distribution, and lipid metabolism.1,2 Consequently, postmenopausal women often experience several metabolic alterations, such as elevated blood pressure, increased insulin resistance, redistribution of body fat to the abdominal area, elevated low-density lipoprotein cholesterol (LDL-C), and decreased high-density lipoprotein cholesterol (HDL-C) levels. These changes contribute to a significantly higher risk of developing cardiometabolic diseases (CMDs).1,3 Postmenopausal women are approximately twice as likely to develop CMDs as premenopausal women.4Plant-based diets rich in fruit and vegetables have emerged as a promising strategy to improve health outcomes and quality of life in postmenopausal women at high risk of CMDs. This is primarily due to their high content of bioactive compounds such as (poly)phenols. Consumption of phenolic compounds has been associated with a decreased risk of non-communicable diseases, including neurodegenerative diseases, cancer, and cardiovascular diseases (CVDs), among others.5,6 Moreover, several epidemiological studies have demonstrated an inverse association between adherence to a Mediterranean diet based on (poly)phenol-rich plant foods and mortality in individuals at high risk of CVDs.7,8 In particular, in the PREDIMED study, researchers observed a significant 37% reduction in all-cause mortality when comparing individuals with the highest (1235 mg per day) versus the lowest (483 mg per day) quintiles of (poly)phenol intake.9 Similarly, Mendonça et al.10 showed that individuals with a high intake of flavonoids (highest quintile) had a 47% lower incidence of developing cardiovascular events than those with a low intake of flavonoids (lowest quintile). Researchers suggested that oranges, dark chocolate, nuts, and tea could be the plant foods responsible for the cardioprotective effects, since these (poly)phenol-rich foods (PP-rich foods) were the most relevant contributors to the dietary intake of phenolic compounds.
Clinical trials testing the cardioprotective effect of several PP-rich foods have observed a high inter-individual variability, concluding that the magnitude of the beneficial effect of (poly)phenol intake on human health may be influenced by different factors such as the structure and the concentration of the ingested compound, the bioavailability in the body, and the gut microbiome.11–13 Microbial catabolism occurring in the large intestine and human phase II conjugation occurring in enterocytes and hepatocytes give origin to phenolic metabolites that are potentially more bioactive than their precursors.12,14 Since these compounds are more easily absorbed and have a longer half-life in the body, phenolic metabolites may exert a beneficial effect on target organs and tissues greater than that of native compounds present in foods.12
Gut microbiota composition and functionality are the most important factors influencing (poly)phenol bioavailability and so determining inter-individual differences in the physiological response to (poly)phenol intake.11 In this context, it has been suggested that postmenopausal women may have an imbalance in the composition and activity of the gut microbiota due to the abrupt decline in circulating oestrogen levels. In particular, postmenopausal women experience a decrease in the Firmicute/Bacteroidetes ratio, as well as in the relative abundance of short-chain fatty acid (SCFA) producers such as Faecalibacterium, Bifidobacterium and Ruminococcus. Other studies have reported that postmenopausal gut microbiota is characterized by a lower abundance of the Lachnospiraceae family and a higher abundance of the genera Prevotella, Parabacteroides, Bacteroides, and Bilophila.15–17 In addition, menopause-related obesity further exacerbates gut dysbiosis in postmenopausal women by reducing the microbial diversity and depleting key beneficial taxa, including Akkermansia muciniphila.18 This imbalance in the gut microbiota profile of postmenopausal women could influence (poly)phenol metabolism and, consequently, the impact of consuming PP-rich foods on the cardiometabolic risk associated with menopause.
Therefore, this study aimed to assess the impact of a chronic daily consumption of PP-rich foods on the urinary phenolic profile in postmenopausal women at high cardiometabolic risk.
Materials and methods
Participants and dietary intervention
A total of 25 postmenopausal women aged 45–65 years at high risk of CMDs were enrolled in a dietary intervention trial, and the study design has been previously published.19 The study protocol was approved by the Research Ethics Committee of the University of Murcia (CEI:ID:3636/2022) and was registered as a clinical research study on Clinical-Trials.gov (NCT 05255367). Briefly, a 3-month trial was conducted, consisting of a 1-month control period during which participants followed their habitual diet, followed by a 2-month experimental period in which their habitual diet was daily supplemented with 16.6 g of 85% dark chocolate, one cup of green tea (1 sachet infused in ∼200 mL of boiled water), and 100 mL of fruit juice prepared from commercially refrigerated pomegranate juice (40%), orange juice (30%), and berry juice containing red grapes and blackcurrants (30%), as shown in Fig. S1. The dietary intervention provided a total amount of 1226 µmol of (poly)phenols per day (647.17 µmol from 85% dark chocolate, 191.60 µmol from green tea, and 387.00 µmol from the mix of fruit juice containing pomegranate, orange and berries). Throughout the dietary trial, participants were instructed to maintain their habitual lifestyle and dietary patterns. Adherence to the study protocol was monitored through personal interviews, and dietary intake was assessed using a 24-hour dietary recall. The collected data were analyzed using Nutrium® software (v.2019) in combination with Phenol-Explorer 3.6 database as previously reported.19 The characterization of the phenolic profile of the PP-rich foods included in the dietary intervention has recently been published.19At the baseline (Time 0), beginning (Time 1), and end of the dietary intervention (Time 2), 24 h urine samples were collected, divided into aliquots, and stored at −80 °C until analysis.
Urine sample analysis
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, v/v) and with phosphoric acid 40% (1
5, v/v) and with phosphoric acid 40% (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, v/v) to limit phenolic–protein interactions, and finally the diluted samples were vortexed. Once the plate was activated with 250 µL of methanol and 250 µL of water 0.2% acetic acid, 600 µL of diluted urine were added and the plate was washed with 200 µL of water and 200 µL of water 0.2% acetic acid. Water (40 µL) was added into each well of the final collection plate, and samples were eluted with 90 µL of methanol 0.1% formic acid in three steps. Finally, the final volume (130 µL) was transferred to a vial until analysis.
2, v/v) to limit phenolic–protein interactions, and finally the diluted samples were vortexed. Once the plate was activated with 250 µL of methanol and 250 µL of water 0.2% acetic acid, 600 µL of diluted urine were added and the plate was washed with 200 µL of water and 200 µL of water 0.2% acetic acid. Water (40 µL) was added into each well of the final collection plate, and samples were eluted with 90 µL of methanol 0.1% formic acid in three steps. Finally, the final volume (130 µL) was transferred to a vial until analysis.
The identification of the compounds under study was carried out by comparing the retention time with authentic standards and/or MS/MS fragmentation patterns, using Xcalibur software 2.1 (Thermo Fisher Scientific Inc.). The nomenclature used for phenolic metabolites follows the current recommendations described by Kay et al.,22 as well as Curti et al.23 Quantification was performed with calibration curves of standards, while some metabolites were quantified with the most structurally similar compound when standards were not available. Metabolite data are expressed as amount (μmol) excreted in 24 h urine.
Statistical analysis
Results were expressed as mean values ± standard error of the mean (SEM). Normality and homoscedasticity tests were performed using the Kolmogorov–Smirnov and Levene's tests, respectively. Comparisons within time points (Time 0, Time 1, and Time 2) were made by Friedman's test for dependent samples, and a Wilcoxon's post hoc signed-rank test was used to assess differences between pairs. Differences were considered significant at p-value < 0.05. All statistical analyses were performed using the SPSS statistical package (version 25, SPSS, Inc., Chicago, IL, USA).Principal component analysis (PCA) was carried out to elucidate the effect of the dietary intervention with PP-rich foods on the urinary excretion of phenolic metabolites. PCA was performed using SIMCA 16.0.1 software (Sartorius Stedim Data Analytics, Umea, Sweden). Datasets were not subjected to any data transformation, but unit variance (UV) scaled.24 The quality of the model and data interpretability were evaluated using the R2X and Q2 parameters. Partial least squares discriminant analysis (PLS-DA) was performed to determine which metabolites had the greatest impact on the urinary phenolic profile after 2-month dietary intervention with PP-rich foods in postmenopausal women, using VIP (variable importance in projection) score values and VIP score > 1.0, p < 0.05 in Student's t test, and log2FC < −1 (significantly decreased) or log2FC > 1 (significantly increased) in a Volcano plot. Plots were performed with MetaboAnalyst25,26 and Rstudio with the ggplot2 package.
Results
Urinary excretion of phenolic metabolites after dietary intervention
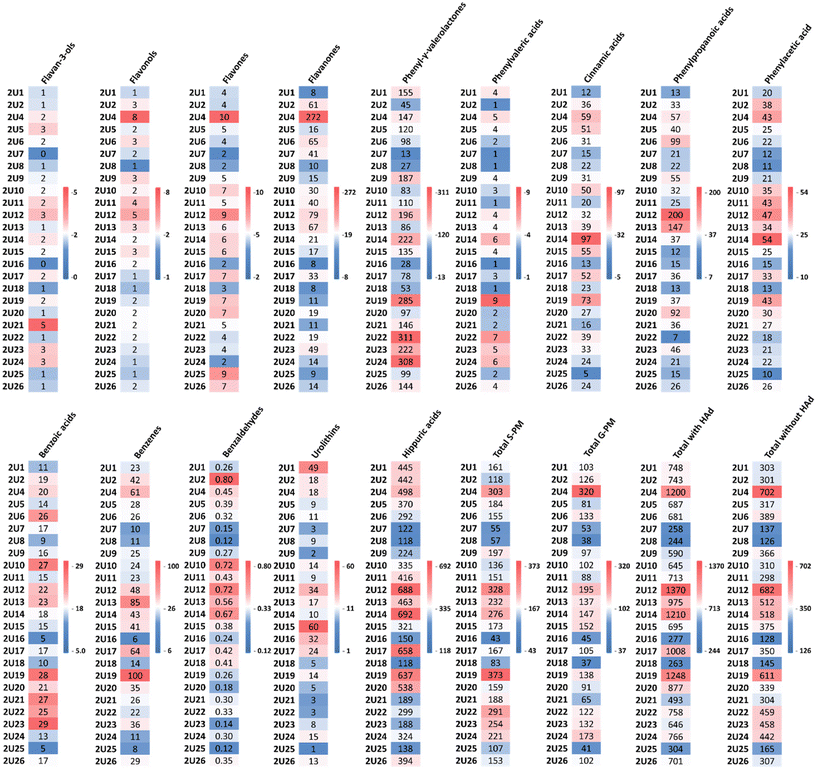
After a 2-month dietary intervention with PP-rich foods, a total of 116 gut microbiota and phase II-derived metabolites were identified in 24 h urine samples (Table 1). Phenolic metabolites derived from different (poly)phenol classes, including flavan-3-ols, flavonols, flavones, flavanones, anthocyanins, and ellagitannins, which characterize the foods supplemented within the dietary intervention. To better characterize the phenolic metabolite profile before and after the dietary intervention, phenolic metabolites were grouped into different classes based on their aglycon moieties: flavan-3-ol monomers (n = 5), flavonols (n = 6), flavones (n = 1), flavanones (n = 4), phenyl-γ-valerolactones (PVLs, n = 24), phenylvaleric acids (PVAs, n = 3), cinnamic acids (n = 18), phenylpropanoic acids (n = 14), phenylacetic acids (n = 6), benzoic acids (n = 18), benzenes (n = 6), benzaldehydes (n = 1), and ellagic acid derivatives and urolithins (n = 8), and hippuric acid (n = 2). Furthermore, Table 1 summarizes the total urinary excretion of sulphated and glucuronidated phenolic metabolites, providing insights into the (poly)phenol bioavailability ingested through the PP-rich foods over a 2-month period by postmenopausal women.| Phenolic metabolites | Time 0 | Time 1 | Time 2 | p value | log2 fold-change (T2 vs. T1) |
|---|---|---|---|---|---|
| Mean ± SEM (µmol) | |||||
| The amounts excreted (µmol) and the log2 fold-change (calculated by dividing the amount excreted at Time 2 by the amount excreted at Time 1) are indicated. T1: Time 1; T2: Time 2; LOQ: limit of quantification; NC: non-log2-fold-change. Data are indicated as mean ± SEM and are expressed as µmol excreted in 24 h urine. Compounds presenting different letters (a–c) indicate significant differences among time points (p value <0.05). Nomenclature of phenolic metabolites is reported as proposed by Kay et al.,22 as well as Curti et al.,23 and the abbreviations are reported under brackets. | |||||
| FLAVAN-3-OLS | |||||
| Methoxy-(epi)gallocatechin-glucuronide (MeO-EGC-Glc) | <LOQb | <LOQb | 0.10 ± 0.02a | <0.001 | NC |
| (−)-Epicatechin (EC) | 0.02 ± 0.01 | 0.02 ± 0.01 | 0.01 ± 0.00 | 0.267 | −1.00 |
| (Epi)catechin-glucuronide isomer 1 (EC-Glc-iso1) | 0.16 ± 0.04b | 0.17 ± 0.04b | 0.60 ± 0.10a | <0.001 | 1.82 |
| (Epi)catechin-glucuronide isomer 2 (EC-Glc-iso2) | 0.31 ± 0.03b | 0.37 ± 0.04b | 0.64 ± 0.05a | <0.001 | 0.79 |
| (Epi)catechin-sulfate (EC-S) | 0.09 ± 0.02b | 0.12 ± 0.03b | 0.41 ± 0.06a | <0.001 | 1.77 |
| FLAVONOLS | |||||
| Myricetin-glucuronide (Myricetin-Glc) | 0.29 ± 0.06b | 0.36 ± 0.06b | 0.62 ± 0.11a | 0.017 | 0.78 |
| Quercetin-3-glucuronide (Quercetin-3-Glc) | 0.29 ± 0.04b | 0.29 ± 0.05b | 0.41 ± 0.05a | 0.036 | 0.50 |
| Kaempferol-glucuronide (Kaempferol-Glc) | 0.24 ± 0.04 | 0.24 ± 0.04 | 0.36 ± 0.05 | 0.056 | 0.58 |
| Myricetin-sulfate (Myricetin-S) | 0.75 ± 0.16 | 1.08 ± 0.27 | 0.74 ± 0.14 | 0.544 | −0.55 |
| Quercetin-3-sulfate (Quercetin-3-S) | 0.28 ± 0.13 | 0.29 ± 0.16 | 0.15 ± 0.07 | 0.797 | −0.95 |
| Kaempferol-sulfate (Kaempferol-S) | 0.09 ± 0.02 | 0.11 ± 0.02 | 0.10 ± 0.01 | 0.289 | −0.14 |
| FLAVONES | |||||
| Apigenin-glucuronide (Apigenin-Glc) | 2.54 ± 0.22b | 2.48 ± 0.20b | 5.28 ± 0.44a | <0.001 | 1.09 |
| FLAVANONES | |||||
| Hesperetin-glucuronide (Hesperetin-Glc) | 23.98 ± 7.34 | 25.47 ± 9.82 | 19.07 ± 5.92 | 0.978 | −0.42 |
| Naringenin-glucuronide (Naringenin-Glc) | 16.49 ± 6.61 | 26.15 ± 11.12 | 12.04 ± 3.14 | 0.600 | −1.12 |
| Hesperetin-sulfate (Hesperetin-S) | 8.79 ± 4.15 | 9.09 ± 4.93 | 4.75 ± 2.03 | 0.722 | −0.94 |
| Naringenin-sulfate (Naringenin-S) | 1.39 ± 0.30 | 1.69 ± 0.35 | 1.58 ± 0.20 | 0.331 | −0.10 |
| PHENYL-γ-VALEROLACTONES (PVLs) | |||||
| 5-(3′,4′,5′-Trihydroxyphenyl)-γ-valerolactone (3′,4′,5′-TriHPVL) | 0.03 ± 0.01b | 0.02 ± 0.01b | 0.09 ± 0.02a | <0.001 | 2.17 |
| 5-(Dihydroxyphenyl)-γ-valerolactone-glucuronide (3′,4′,5′) isomer 1 (DiHPVL-Glc-iso1) | <LOQ | 0.02 ± 0.00 | 0.03 ± 0.01 | 0.059 | 0.58 |
| 5-(Dihydroxyphenyl)-γ-valerolactone-glucuronide (3′,4′,5′) isomer 2 (DiHPVL-Glc-iso2) | <LOQc | 0.13 ± 0.06b | 1.43 ± 0.32a | <0.001 | 3.46 |
| 5-(Dihydroxyphenyl)-γ-valerolactone-sulfate (3′,4′,5′ isomers) (DiHPVL-S) | 0.19 ± 0.03ab | 0.12 ± 0.03b | 0.32 ± 0.07a | 0.020 | 1.42 |
| 5-(Hydroxyphenyl)-γ-valerolactone-methoxy-glucuronide (3′,4′,5′) isomer 1 (HPVL-MeO-Glc-iso1) | 0.02 ± 0.01b | 0.03 ± 0.01b | 0.17 ± 0.04a | <0.001 | 2.50 |
| 5-(Hydroxyphenyl)-γ-valerolactone-methoxy-glucuronide (3′,4′,5′) isomer 2 (HPVL-MeO-Glc-iso2) | <LOQb | <LOQb | 0.13 ± 0.07a | <0.001 | NC |
| 5-(Hydroxyphenyl)-γ-valerolactone-methoxy-sulfate (3′,4′,5′) isomer 1 (HPVL-MeO-S-iso1) | 0.02 ± 0.01b | 0.04 ± 0.02b | 0.61 ± 0.12a | <0.001 | 3.93 |
| 5-(3′,4′-Dihydroxyphenyl)-γ-valerolactone (3′,4′-DiHPVL) | 0.14 ± 0.06b | 0.11 ± 0.04b | 1.33 ± 0.32a | <0.001 | 3.60 |
| 5-(3′,5′-Dihydroxyphenyl)-γ-valerolactone (3′,5′-DiHPVL) | 0.06 ± 0.01b | 0.05 ± 0.02b | 0.25 ± 0.04a | <0.001 | 2.32 |
| 5-(5′-Hydroxyphenyl)-γ-valerolactone-3′-glucuronide (5′-HPVL-3′-Glc) | 0.05 ± 0.02b | 0.12 ± 0.06b | 2.26 ± 0.46a | <0.001 | 4.24 |
| 5-(5′-Hydroxyphenyl)-γ-valerolactone-3′-sulfate (5′-HPVL-3′-S) | 0.12 ± 0.05b | 0.21 ± 0.06b | 7.28 ± 1.61a | <0.001 | 5.12 |
| 5-(3′-Hydroxyphenyl)-γ-valerolactone-4′-glucuronide (3′-HPVL-4′-Glc) | 0.71 ± 0.13b | 0.87 ± 0.17b | 4.13 ± 0.95a | <0.001 | 2.25 |
| 5-(4′-Hydroxyphenyl)-γ-valerolactone-3′-glucuronide (4′-HPVL-3′-Glc) | 2.35 ± 0.43b | 2.56 ± 0.51b | 11.88 ± 2.55a | <0.001 | 2.21 |
| 5-(Hydroxyphenyl)-γ-valerolactone-sulfate (3′,4′ isomers) (HPVL-S) | 23.50 ± 4.12b | 23.64 ± 4.11b | 84.45 ± 10.39a | <0.001 | 1.84 |
| 5-Phenyl-γ-valerolactone-methoxy-glucuronide (3′,4′) isomer 1 (PVL-MeO-Glc-iso1) | 0.68 ± 0.10b | 0.64 ± 0.10b | 2.12 ± 0.26a | <0.001 | 1.73 |
| 5-Phenyl-γ-valerolactone-methoxy-glucuronide (3′,4′) isomer 2 (PVL-MeO-Glc-iso2) | 0.63 ± 0.06ab | 0.42 ± 0.06b | 0.76 ± 0.10a | 0.008 | 0.86 |
| 5-Phenyl-γ-valerolactone-methoxy-sulfate (3′,4′) isomer 1 (PVL-MeO-S-iso1) | 0.12 ± 0.02b | 0.12 ± 0.02b | 0.42 ± 0.07a | <0.001 | 1.81 |
| 5-Phenyl-γ-valerolactone-methoxy-sulfate (3′,4′) isomer 2 (PVL-MeO-S-iso2) | 0.17 ± 0.04b | 0.21 ± 0.04b | 0.70 ± 0.09a | <0.001 | 1.74 |
| 5-(3′-Hydroxyphenyl)-γ-valerolactone (3′-HPVL) | 0.01 ± 0.01ab | <LOQb | 0.04 ± 0.02a | 0.025 | NC |
| 5-Phenyl-γ-valerolactone-4′-glucuronide (PVL-4′-Glc) | 0.37 ± 0.27b | 0.46 ± 0.19b | 1.13 ± 0.38a | <0.001 | 1.30 |
| 5-Phenyl-γ-valerolactone-3′-glucuronide (PVL-3′-Glc) | 0.20 ± 0.09b | 0.16 ± 0.05b | 1.48 ± 0.33a | <0.001 | 3.21 |
| 5-Phenyl-γ-valerolactone-4′-sulfate (PVL-4′-S) | 0.27 ± 0.04ab | 0.20 ± 0.02b | 0.39 ± 0.06a | 0.049 | 0.96 |
| 5-Phenyl-γ-valerolactone-3′-sulfate (PVL-3′-S) | 0.74 ± 0.22b | 0.53 ± 0.11b | 4.42 ± 0.97a | <0.001 | 3.06 |
| 5-Phenyl-γ-valerolactone-sulfate-glucuronide (3′,4′ isomers) (PVL-S-Glc) | 2.54 ± 0.40b | 2.77 ± 0.58b | 9.93 ± 1.28a | <0.001 | 1.84 |
| PHENYLVALERIC ACIDS (PVAs) | |||||
| 4-Hydroxy-5-(3′,4′-dihydroxyphenyl)valeric acid (4-OH-3′,4′-DiHPVA) | <LOQb | <LOQb | 0.03 ± 0.01a | <0.001 | NC |
| 4-Hydroxy-5-(hydroxyphenyl)valeric acid-sulfate (3′,4′ isomers) (4-OH-HPVA-S) | 0.61 ± 0.09b | 0.80 ± 0.16b | 2.75 ± 0.36a | <0.001 | 1.78 |
| 4-Hydroxy-5-(phenyl)valeric acid-glucuronide (3′,4′ isomers) (4-OH-PVA-Glc) | 0.08 ± 0.02b | 0.07 ± 0.02b | 0.51 ± 0.09a | <0.001 | 2.87 |
| CINNAMIC ACIDS (CAs) | |||||
| 3′,4′-Dihydroxycinnamic acid (3′,4′-DiHCA) | 0.04 ± 0.01b | 0.06 ± 0.01b | 0.13 ± 0.02a | <0.001 | 1.12 |
| 3′-Hydroxycinnamic acid-4′-glucuronide (3′-HCA-4′-Glc) | 0.16 ± 0.03ab | 0.10 ± 0.01b | 0.17 ± 0.02a | 0.016 | 0.77 |
| 4′-Hydroxycinnamic acid-3′-glucuronide (4′-HCA-3′-Glc) | 0.32 ± 0.05 | 0.32 ± 0.06 | 0.42 ± 0.08 | 0.496 | 0.39 |
| 3′-Hydroxycinnamic acid-4′-sulfate (3′-HCA-4′-S) | 0.55 ± 0.09 | 0.57 ± 0.10 | 0.66 ± 0.09 | 0.651 | 0.21 |
| 4′-Hydroxycinnamic acid-3′-sulfate (4′-HCA-3′-S) | 4.99 ± 0.60ab | 3.76 ± 0.42b | 6.50 ± 0.93a | 0.024 | 0.79 |
| 4′-Hydroxy-3′-methoxycinnamic acid (3′-MeO-4′-HCA) | 0.16 ± 0.02 | 0.15 ± 0.02 | 0.15 ± 0.02 | 0.872 | 0.00 |
| 3′-Methoxycinnamic acid-4′-glucuronide (3′-MeO-CA-4′-Glc) | 7.47 ± 0.78 | 8.05 ± 0.79 | 9.99 ± 1.15 | 0.222 | 0.31 |
| 4′-Methoxycinnamic acid-3′-glucuronide (4′-MeO-CA-3′-Glc) | 2.14 ± 0.22 | 1.62 ± 0.18 | 2.05 ± 0.32 | 0.249 | 0.34 |
| 3′-Methoxycinnamic acid-4′-sulfate (3′-MeO-CA-4′-S) | 3.22 ± 0.27 | 3.69 ± 0.53 | 4.24 ± 0.52 | 0.646 | 0.20 |
| 4′-Methoxycinnamic acid-3′-sulfate (4′-MeO-CA-3′-S) | 0.21 ± 0.02 | 0.20 ± 0.03 | 0.21 ± 0.03 | 0.625 | 0.07 |
| N-(4′-Hydroxy-3′-methoxy-cinnamoyl)glycine (3′-MeO-4′-HCA-Gly) | 5.99 ± 0.63 | 6.70 ± 0.95 | 6.12 ± 0.87 | 0.884 | −0.13 |
| 2′-Hydroxycinnamic acid (2′-HCA) | 0.14 ± 0.03 | 0.15 ± 0.03 | 0.15 ± 0.03 | 0.370 | 0.00 |
| 3′-Hydroxycinnamic acid (3′-HCA) | 0.09 ± 0.02 | 0.14 ± 0.04 | 0.13 ± 0.03 | 0.388 | −0.11 |
| 4′-Hydroxycinnamic acid (4′-HCA) | 0.36 ± 0.06 | 0.32 ± 0.04 | 0.55 ± 0.10 | 0.286 | 0.78 |
| Cinnamic acid-4′-glucuronide (CA-4′-Glc) | 0.64 ± 0.34 | 0.23 ± 0.04 | 0.45 ± 0.15 | 0.448 | 0.97 |
| Cinnamic acid-3′-glucuronide (CA-3′-Glc) | 0.12 ± 0.03 | 0.17 ± 0.06 | 0.21 ± 0.05 | 0.185 | 0.30 |
| Cinnamic acid-4′-sulfate (CA-4′-S) | 1.22 ± 0.17 | 1.10 ± 0.21 | 1.14 ± 0.19 | 0.599 | 0.05 |
| Cinnamic acid-3′-sulfate (CA-3′-S) | 1.37 ± 0.45 | 0.90 ± 0.18 | 1.91 ± 0.52 | 0.167 | 1.09 |
| PHENYLPROPANOIC ACIDS (PPAs) | |||||
| 3-(3′,4′-Dihydroxyphenyl)propanoic acid (3′,4′-DiHPPA) | 0.94 ± 0.15 | 1.55 ± 0.33 | 1.47 ± 0.31 | 0.388 | −0.08 |
| 3-(3′-Hydroxyphenyl)propanoic acid-4′-glucuronide (3′-HPPA-4′-Glc) | 0.09 ± 0.04b | 0.28 ± 0.11ab | 0.70 ± 0.29a | 0.003 | 1.32 |
| 3-(4′-Hydroxyphenyl)propanoic acid-3′-glucuronide (4′-HPPA-3′-Glc) | 0.01 ± 0.01b | 0.03 ± 0.03ab | 0.09 ± 0.03a | 0.006 | 1.58 |
| 3-(4′-Hydroxyphenyl)propanoic acid-3′-sulfate (4′-HPPA-3′-S) | 9.43 ± 1.82 | 8.30 ± 2.42 | 12.36 ± 4.06 | 0.304 | 0.57 |
| 3-(4′-Hydroxy-3′-methoxyphenyl)propanoic acid (3′-MeO-4′-HPPA) | 13.55 ± 2.42 | 18.12 ± 3.78 | 14.29 ± 3.34 | 0.387 | −0.34 |
| 3-(3′-Hydroxy-4′-methoxyphenyl)propanoic acid (4′-MeO-3′-HPPA) | 1.61 ± 0.63 | 1.37 ± 0.48 | 2.11 ± 1.34 | 0.531 | 0.62 |
| 3-(3′-Methoxyphenyl)propanoic acid-4′-glucuronide (3′-MeO-PPA-4′-Glc) | 1.09 ± 0.15 | 0.91 ± 0.16 | 1.03 ± 0.18 | 0.511 | 0.18 |
| 3-(4′-Methoxyphenyl)propanoic acid-3′-glucuronide (4′-MeO-PPA-3′-Glc) | 3.85 ± 0.79 | 2.65 ± 0.53 | 2.71 ± 0.44 | 0.266 | 0.03 |
| 3-(3′-Methoxyphenyl)propanoic acid-4′-sulfate (3′-MeO-PPA-4′-S) | 0.91 ± 0.20 | 0.81 ± 0.19 | 1.19 ± 0.30 | 0.767 | 0.55 |
| 3-(4′-Methoxyphenyl)propanoic acid-3′-sulfate (4′-MeO-PPA-3′-S) | 0.63 ± 0.23 | 0.45 ± 0.16 | 0.37 ± 0.06 | 0.530 | −0.28 |
| 3-(3′-Hydroxyphenyl)propanoic acid (3′-HPPA) | 1.58 ± 0.42 | 1.24 ± 0.23 | 1.46 ± 0.40 | 0.780 | 0.24 |
| 3-(Phenyl)propanoic acid-4′-glucuronide (PPA-4′-Glc) | 1.08 ± 0.28 | 0.57 ± 0.08 | 0.92 ± 0.20 | 0.674 | 0.69 |
| 3-(Phenyl)propanoic acid-3′-glucuronide (PPA-3′-Glc) | 0.51 ± 0.13 | 0.41 ± 0.06 | 0.49 ± 0.08 | 0.622 | 0.26 |
| 3-(Phenyl)propanoic acid-sulfate (3′,4′ isomers) (PPA-S) | 6.21 ± 1.29ab | 4.00 ± 0.62b | 6.27 ± 1.09a | 0.043 | 0.65 |
| PHENYLACETIC ACID (PAAs) | |||||
| 4′-Hydroxy-3′-methoxyphenylacetic acid (3′-MeO-4′-HPAA) | 3.47 ± 0.53b | 3.69 ± 0.54b | 5.51 ± 0.74a | 0.021 | 0.58 |
| 3′-Hydroxy-4′-methoxyphenylacetic acid (4′-MeO-3′-HPAA) | 3.00 ± 0.49 | 2.94 ± 0.47 | 4.02 ± 0.88 | 0.960 | 0.45 |
| 3′-Methoxy-phenylacetic acid-4′-sulfate (3′-MeO-PAA-4′-S) | 1.25 ± 0.28 | 1.41 ± 0.33 | 1.55 ± 0.31 | 0.781 | 0.14 |
| 4′-Methoxy-phenylacetic acid-3′-sulfate (4′-MeO-PAA-3′-S) | 0.72 ± 0.09 | 0.80 ± 0.11 | 0.99 ± 0.15 | 0.579 | 0.31 |
| 3′-Hydroxyphenylacetic acid (3′-HPAA) | 6.93 ± 1.16 | 7.69 ± 1.16 | 10.59 ± 1.49 | 0.081 | 0.46 |
| 4′-Hydroxyphenylacetic acid (4′-HPAA) | 5.05 ± 0.58 | 5.39 ± 0.48 | 4.87 ± 0.54 | 0.651 | −0.15 |
| BENZOIC ACIDS (BAs) | |||||
| 3,4,5-Trihydroxybenzoic acid (3,4,5-TriHBA) | 0.02 ± 0.01b | 0.02 ± 0.01b | 0.09 ± 0.01a | <0.001 | 2.17 |
| 4-Methoxy-3,5-dihydroxybenzoic acid (4-MeO-3,5-DiHBA) | 0.07 ± 0.02b | 0.07 ± 0.04b | 0.13 ± 0.02a | <0.001 | 0.89 |
| 4-Hydroxy-3,5-dimethoxybenzoic acid (3,5-DiMeO-4-HBA) | 0.08 ± 0.01 | 0.11 ± 0.03 | 0.10 ± 0.01 | 0.274 | −0.14 |
| Hydroxy-methoxybenzoic acid-sulfate (MeO-HBA-S) | 0.17 ± 0.04b | 0.14 ± 0.04b | 0.26 ± 0.03a | 0.001 | 0.89 |
| 3,4-Dihydroxybenzoic acid (3,4-DiHBA) | 0.32 ± 0.03 | 0.38 ± 0.07 | 0.46 ± 0.05 | 0.106 | 0.28 |
| 3-Hydroxybenzoic acid-4-glucuronide (3-HBA-4-Glc) | 0.06 ± 0.02b | 0.09 ± 0.02b | 0.11 ± 0.02a | 0.045 | 0.29 |
| 4-Hydroxybenzoic acid-3-glucuronide (4-HBA-3-Glc) | 0.03 ± 0.01 | 0.05 ± 0.03 | 0.09 ± 0.03 | 0.162 | 0.85 |
| Hydroxybenzoic acid-sulfate (3,4 isomers) (HBA-S) | 0.20 ± 0.07 | 0.21 ± 0.11 | 0.62 ± 0.18 | 0.108 | 1.56 |
| 4-Hydroxy-3-methoxybenzoic acid (3-MeO-4-HBA) | 0.05 ± 0.02b | 0.14 ± 0.05b | 0.47 ± 0.07a | <0.001 | 1.75 |
| 3-Methoxybenzoic acid-4-glucuronide (3-MeO-BA-4-Glc) | 1.72 ± 0.30 | 1.38 ± 0.23 | 2.15 ± 0.35 | 0.175 | 0.64 |
| 4-Methoxybenzoic acid-3-glucuronide (4-MeO-BA-3-Glc) | 2.18 ± 0.33 | 1.46 ± 0.22 | 2.30 ± 0.36 | 0.118 | 0.66 |
| 3′-Methoxybenzoic acid-4-sulfate (3′-MeO-BA-4-S) | 2.23 ± 0.60 | 1.98 ± 0.37 | 2.30 ± 0.30 | 0.252 | 0.22 |
| 4′-Methoxybenzoic acid-3-sulfate (4′-MeO-BA-3-S) | 0.36 ± 0.05 | 0.28 ± 0.05 | 0.39 ± 0.05 | 0.167 | 0.48 |
| 4-Hydroxybenzoic acid (4-HBA) | 4.43 ± 0.56 | 4.48 ± 0.45 | 4.80 ± 0.59 | 0.671 | 0.10 |
| Benzoic acid-4-glucuronide (BA-4-Glc) | 0.04 ± 0.01 | 0.04 ± 0.01 | 0.04 ± 0.01 | 0.973 | 0.00 |
| Benzoic acid-3-glucuronide (BA-3-Glc) | 0.20 ± 0.05 | 0.23 ± 0.08 | 0.21 ± 0.04 | 0.752 | −0.13 |
| Benzoic acid-4-sulfate (BA-4-S) | 1.00 ± 0.19 | 1.15 ± 0.24 | 1.27 ± 0.21 | 0.635 | 0.14 |
| Benzoic acid-3-sulfate (BA-3-S) | 1.17 ± 0.17b | 0.99 ± 0.16b | 2.21 ± 0.38a | 0.003 | 1.16 |
| BENZENES (BZs) | |||||
| 1,2,3-Trihydroxybenzene (1,2,3-TriOH-Benz) | 0.04 ± 0.04 | 0.14 ± 0.10 | 0.34 ± 0.15 | 0.157 | 1.28 |
| 2-Hydroxy-3/6-methoxybenzene-1-sulfate or 3-hydroxy-2-methoxybenzene-1-sulfate (MeO-Pyr-S) | 0.04 ± 0.02b | 0.05 ± 0.02b | 0.26 ± 0.09a | 0.033 | 2.38 |
| 1,2-Dihydroxybenzene (1,2-DiOH-Benz) | 3.59 ± 0.86 | 3.44 ± 1.02 | 3.33 ± 0.80 | 0.822 | −0.05 |
| 2-Hydroxy-benzene-1-glucuronide (Cat-Glc) | 0.82 ± 0.11 | 0.61 ± 0.09 | 0.77 ± 0.10 | 0.350 | 0.34 |
| 2-Hydroxy-benzene-1-sulfate (Cat-S) | 22.89 ± 3.06 | 21.14 ± 2.49 | 27.57 ± 3.73 | 0.444 | 0.38 |
| 2-Hydroxy-4/5-methylbenzene-1-sulfate (Met-Cat-S) | 1.15 ± 0.14 | 1.11 ± 0.18 | 1.37 ± 0.22 | 0.600 | 0.30 |
| BENZALDEHYDES (BALs) | |||||
| 4-Hydroxybenzaldehyde (4-HBAL) | 0.30 ± 0.03 | 0.35 ± 0.04 | 0.37 ± 0.04 | 0.547 | 0.08 |
| ELLAGIC ACID DERIVATIVES AND UROLITHINS (UROs) | |||||
| Dimethylellagic acid-glucuronide (DMEA-Glc) | 0.16 ± 0.03b | 0.13 ± 0.02b | 0.38 ± 0.05a | <0.001 | 1.55 |
| 3,8,9-Trihydroxy-urolithin (Uro-C) | 0.06 ± 0.06 | 0.07 ± 0.05 | 0.07 ± 0.02 | 0.053 | 0.00 |
| 3,8-Dihydroxy-urolithin and/or 3,9-dihydroxy-urolithin ((ISO)Uro-A) | 0.54 ± 0.42ab | 0.35 ± 0.12b | 1.89 ± 0.56a | <0.001 | 2.43 |
| 3-Hydroxy-urolithin-8-glucuronide and/or 3-Hydroxy-urolithin-9-glucuronide ((ISO)Uro-A-Glc) | 4.19 ± 2.88b | 3.19 ± 0.89b | 8.82 ± 1.38a | <0.001 | 1.47 |
| 3-Hydroxy-urolithin-8-sulfate and/or 3-hydroxy-urolithin-9-sulfate ((ISO)Uro-A-S) | 0.42 ± 0.41a | 0.05 ± 0.02c | 0.12 ± 0.03b | <0.001 | 1.26 |
| 3-Hydroxy-urolithin (Uro-B) | 0.25 ± 0.21ab | 0.01 ± 0.01b | 0.21 ± 0.11a | 0.050 | 4.39 |
| Urolithin-3-glucuronide (Uro-B-Glc) | 4.72 ± 3.68 | 1.18 ± 0.33 | 4.08 ± 1.84 | 0.380 | 1.79 |
| Urolithin-3-sulfate (Uro-B-S) | 0.08 ± 0.07 | 0.01 ± 0.01 | <LOQ | 0.490 | NC |
| HIPPURIC ACIDS | |||||
| 4-Hydroxyhippuric acid (4-HHA) | 11.85 ± 1.24 | 14.11 ± 1.71 | 14.07 ± 1.28 | 0.310 | 0.00 |
| Hippuric acid (HA) | 309.12 ± 31.99 | 323.16 ± 34.96 | 348.31 ± 36.57 | 0.736 | 0.11 |
| Total sulfated phenolic metabolites | 97.52 ± 0.27b | 91.29 ± 0.31b | 182.57 ± 0.37a | <0.001 | 1.00 |
| Total glucuronidated phenolic metabolites | 83.38 ± 0.35b | 87.37 ± 0.41b | 112.89 ± 0.31a | 0.011 | 0.37 |
| Total (with hippuric acid derivatives) | 555.02 ± 52.63b | 575.36 ± 58.90b | 724.09 ± 63.90a | 0.032 | 0.33 |
| Total (without hippuric acid derivatives) | 234.02 ± 25.29b | 238.09 ± 31.83b | 361.71 ± 32.22a | 0.023 | 0.60 |
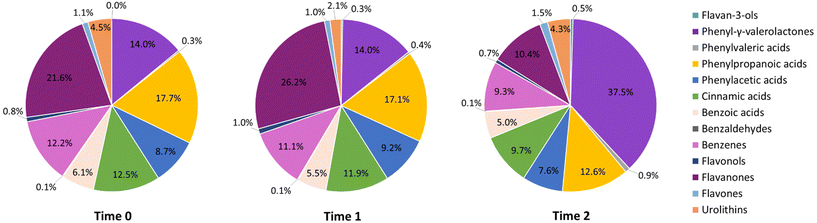
Fig. 1 shows the phenolic metabolite profile of 24 h urine samples of postmenopausal women at different time points. Metabolite levels are expressed as a percentage of total phenolic excretion, excluding hippuric acid and 4-hydroxyhippuric acid, as they have been reported to originate not only from sources related to PP-rich foods ingested.27 During the control period, from Time 0 to Time 1, the phenolic metabolite profile remained stable and was represented mainly by phenolic acids (44.1%) [phenylpropanoic acids (17.1%), cinnamic acids (11.9%), phenylacetic acids (9.2%), benzoic acids (5.5%), and PVAs (0.4%)], followed by flavanones (26.2%), PVLs (14.0%), and benzenes (11.1%), and to a lesser extent by urolithins (2.1%), flavones (1.0%), flavonols (1.0%), and others (0.5%) including flavan-3-ols and benzaldehydes. Whereas, after the 2-month dietary intervention with PP-rich foods (Time 2), the phenolic metabolite profile shifted and was characterized by PVLs, which doubled their concentration (37.5%), followed by phenolic acids (35.8%) [phenylpropanoic acids (12.6%), cinnamic acids (9.7%), phenylacetic acids (7.6%), benzoic acids (5.0%), and PVAs (0.9%)], flavanones (10.4%), benzenes (9.3%), urolithins (4.3%), flavones (1.5%), and flavan-3-ols (0.5%).
In general, the daily intake of dark chocolate, green tea, and fruit juice led to a shift in the urinary profile of phenolic classes (Table 2), with a significant increase in total phenolic metabolite excretion (p = 0.023), rising from a mean value of 238.09 µmol at the beginning of the dietary intervention (Time 1) to a 361.71 µmol at the end of it (Time 2). This increase was mainly linked to a significant increase in the excretion of phase II derivatives of flavan-3-ols, PVLs, and PVAs, which achieved a mean value of 1.76, 135.75, and 3.29 μmol, respectively (p < 0.001), at the end of the dietary intervention, showing log2 fold-changes of 1.35, 2.02, and 1.92, respectively. All these metabolites derive from flavan-3-ols, present in high quantities in 85% dark chocolate and green tea.
| Phenolic classes | Time 0 | Time 1 | Time 2 | p value | log2 fold-change (T2 vs. T1) |
|---|---|---|---|---|---|
| Mean ± SEM (µmol) | |||||
| The amounts excreted (µmol) and the log2 fold-change (calculated dividing the amount excreted at Time 2 by the amount excreted at Time 1) are indicated. T1: Time 1; T2: Time 2; EA: ellagic acid; HAs: hippuric acid derivatives. Data are indicated as mean ± SEM and are expressed as µmol excreted in 24 h urine. Compounds presenting different letters (a,b) indicate significant differences (p value < 0.05) among time points. | |||||
| Flavan-3-ols | 0.59 ± 0.08b | 0.69 ± 0.10b | 1.76 ± 0.19a | <0.001 | 1.35 |
| Flavonols | 1.94 ± 0.31 | 2.37 ± 0.46 | 2.39 ± 0.30 | 0.280 | 0.01 |
| Flavones | 2.54 ± 0.22b | 2.48 ± 0.20b | 5.28 ± 0.44a | <0.001 | 1.09 |
| Flavanones | 50.65 ± 15.13 | 62.41 ± 21.36 | 37.44 ± 10.64 | 0.513 | −0.74 |
| Phenyl-γ-valerolactones | 32.92 ± 5.30b | 33.42 ± 5.71b | 135.75 ± 16.92a | <0.001 | 2.02 |
| Phenylvaleric acids | 0.69 ± 0.09b | 0.87 ± 0.16b | 3.29 ± 0.42a | <0.001 | 1.92 |
| Cinnamic acids | 29.19 ± 2.40 | 28.23 ± 2.81 | 35.19 ± 4.22 | 0.687 | 0.32 |
| Phenylpropanoic acids | 41.50 ± 6.41 | 40.69 ± 7.32 | 45.46 ± 9.08 | 0.846 | 0.16 |
| Phenylacetic acid | 20.43 ± 2.04b | 21.91 ± 2.02ab | 27.53 ± 2.45a | 0.037 | 0.33 |
| Benzoic acids | 14.33 ± 1.28ab | 13.19 ± 1.24b | 18.00 ± 1.38a | 0.018 | 0.45 |
| Benzenes | 28.53 ± 3.56 | 26.48 ± 3.29 | 33.65 ± 4.68 | 0.513 | 0.35 |
| Benzaldehydes | 0.31 ± 0.03 | 0.35 ± 0.04 | 0.37 ± 0.04 | 0.417 | 0.08 |
| EA derivatives and urolithins | 10.43 ± 7.74b | 5.00 ± 1.31b | 15.59 ± 2.92a | <0.001 | 1.64 |
| Hippuric acids | 320.97 ± 32.62 | 337.27 ± 35.82 | 362.38 ± 36.96 | 0.607 | 0.10 |
| Total (with HAs) | 555.02 ± 52.633b | 575.36 ± 58.90b | 724.09 ± 63.90a | 0.032 | 0.33 |
| Total (without HAs) | 234.02 ± 25.29b | 238.09 ± 31.83b | 361.71 ± 32.22a | 0.023 | 0.60 |
A significant increase in the excretion of benzoic acids was also shown, from a mean value of 13.19 µmol at Time 1 to 18.00 µmol at Time 2 (p = 0.018), with a log2 fold-change of 0.45. Benzoic acids are present in berry-containing fruit juice and can also derive from the microbial metabolism of anthocyanins and other phenolic compounds. Finally, a significant increase in the excretion of urolithins was reported, with these metabolites reaching a mean value of 15.59 μmol at Time 2 (p < 0.001) and a log2 fold-change of 1.64. Urolithins may arise from the microbial metabolism of ellagitannins and ellagic acid present in pomegranate and berry-containing fruit juice.
Multivariate analysis reveals the dietary intervention effect on the phenolic metabolic profile in postmenopausal women
Multivariate analysis was carried out to explore globally the effect of the 2-month dietary intervention on the phenolic metabolic profile of postmenopausal women. A PCA model was built considering all metabolites as variables (Fig. 2A) except for hippuric acid derivatives (hippuric acid and 4-hydroxyhippuric acid), which were excluded because they have a clear heterogeneous origin.27 Two principal components (PCs) explained 30.4% of the total variability, with PC1 and PC2 explaining 20.6% and 9.8% of the observed variation, respectively. In the score plot (Fig. 2A), two different clusters can be distinguished: one including observations at the baseline and the beginning of the dietary intervention (Time 0 and Time 1) and another cluster representing the end of the dietary intervention (Time 2). Moreover, the loading plot revealed an increase in the excretion of phenolic metabolites after the 2-month dietary intervention with PP-rich foods (quantitative effect). These findings highlight the consistent urinary excretion of phenolic metabolites in postmenopausal women during their habitual diet and reveal the significant impact of a 2-month daily intake of PP-rich foods on the urinary excretion profile of phenolic metabolites.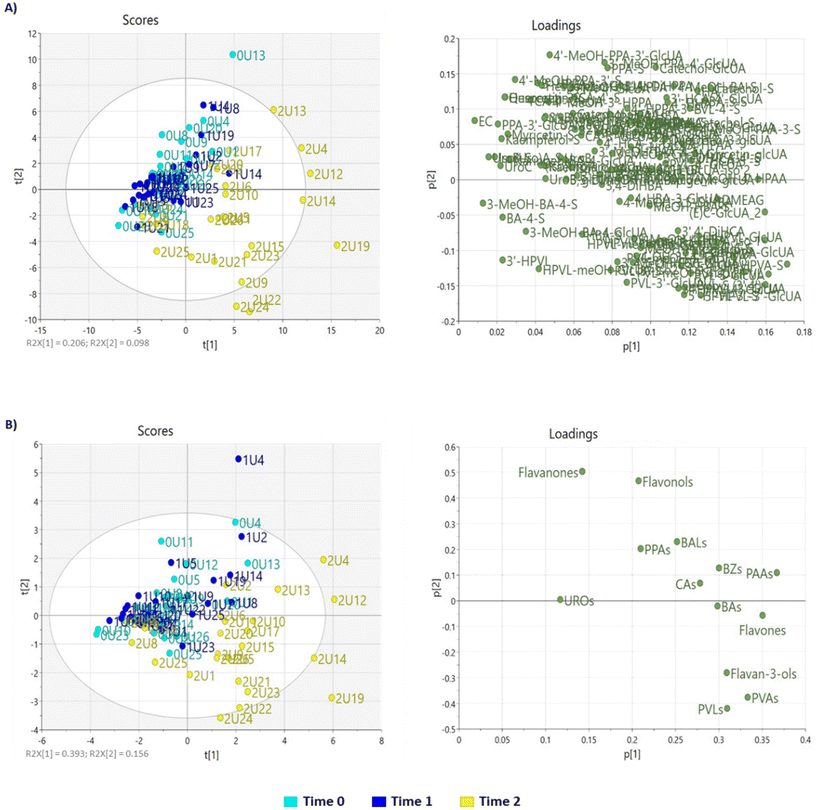 | ||
| Fig. 2 Score and loading plots resulting after PCA on autoscaled data for individual phenolic metabolites (A) and classes of phenolic metabolites (B) obtained at the baseline (Time 0), beginning (Time 1) and end of the 2-month dietary intervention (Time 2). Labels in the score plots indicate the sample number. Metabolite abbreviations in the loading plots are indicated in Table 1. The first number in the codes refer to the time point [Time 0 (0), Time 1 (1) or Time 2 (2)] and the second one refers to the subject number (from U1 to U26). | ||
To further explore the impact of the dietary intervention on the urinary phenolic profile, metabolite classes, rather than individual phenolic metabolites, were considered as variables. In this case (Fig. 2B), two PCs explained 54.9% of the total variability, with PC1 and PC2 explaining 39.3% and 15.6% of the observed variation, respectively. The resulting score and loading plots further supported the impact of the daily dietary intake of PP-rich foods for 2 months on the urinary profile of phenolic metabolites. Furthermore, the loading plot discriminated the metabolite classes into two main groups, one with a positive score for PC2 that included flavanones, flavonols, small phenolic acids (except for benzoic acids), benzenes and benzaldehydes and the other with a negative score for PC2 that included the main metabolites derived from flavan-3-ol metabolism (i.e., phase II derivatives of flavan-3-ols, PVLs, and PVAs). This was expected, since flavan-3-ols were the most abundant class in the PP-rich foods supplemented with the dietary intervention (63.7% of the total (poly)phenols consumed).19
Then, a PLS-DA model was performed to determine which phenolic metabolites had the greatest impact on the urinary excretion profile. The Volcano plot (Fig. 3A) showed that a total of 41 phenolic metabolites were significantly modified after the intake of PP-rich foods (p < 0.001), with 31 metabolites increasing their excretion level and 10 decreasing. The VIP score plot (Fig. 3B) showed that 15 metabolites contributed in a relevant way to the modulation of the urinary excretion after the intervention (VIP score > 1.25), of which 13 increased their excretion level and 2 decreased [4′-HPAA and 4-HBA]. The metabolites characterizing the excretion profile after the intervention are derived specifically from flavan-3-ol sources and include 9 PVLs, 1 PVA, 2 phase II conjugates of flavan-3-ols, and 3,4,5-TriHBA. On the other hand, the metabolites that decrease after the dietary intervention are phenylacetic and benzoic acids that can be derived from sources different from (poly)phenols, including aromatic amino acids and catecholamines.13,28
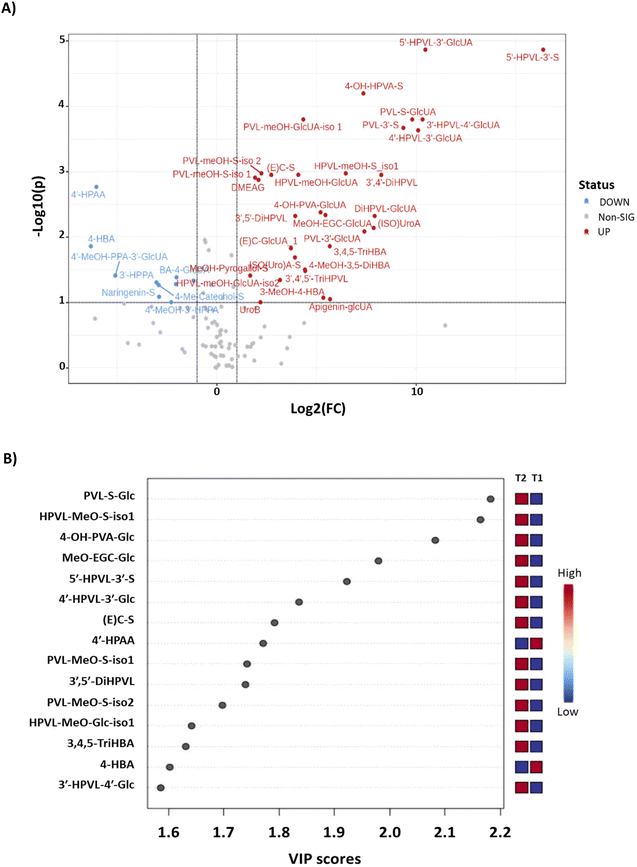 | ||
| Fig. 3 Volcano plot (A) and VIP scores plot (B) of phenolic metabolites excreted in 24 h urine of postmenopausal women after dietary intervention with PP-rich foods for 2 months. Significantly increased metabolites appear in red (log2FC > 1), significantly decreased metabolites appear in blue (log2FC < −1), and non-significantly different metabolites appear in grey. The size of the dots represents the VIP score value (a larger dot size represents a higher VIP value). The relative abundance of metabolites is indicated by a coloured scale from blue (low) to red (high). The codes T1 and T2 correspond to the time points Time 1 and Time 2, respectively. Phenolic metabolite abbreviations in the plots are indicated in Table 1. | ||
Finally, to explore inter-individual differences in phenolic metabolite profiles, a heatmap based on the phenolic metabolite classes identified after the dietary intervention was obtained (Fig. 4). This analysis revealed substantial inter-individual variability in phenolic metabolite excretion following the consumption of PP-rich foods, allowing the identification of three phenolic-metabolite-producer phenotypes (low, medium, and high), according to thirtile calculated as previously described.29 Excluding hippuric acids (since their origin is not exclusively to the (poly)phenol metabolism), 32% of the participants were classified as low producers, 32% as medium producers, and 36% as high producers, with mean total phenolic metabolite excretion values of 200 µmol, 334 µmol, and 530 µmol, respectively. A similar distribution was observed across individual metabolite classes. For instance, in the case of phenyl-γ-valerolactones, low producers (32%) exhibited a mean value of 52 µmol, medium producers (32%) showed a mean value of 119 µmol, while high producers (36%) reached a mean value of 226 µmol. A comparable trend was observed for urolithins, with mean excretion values of 4 µmol, 11 µmol, and 30 µmol in low, medium and high producers, respectively. However, we were unable to identify the urolithin metabotype (UM-A, UM-B and UM-0), as we were unable to stratify the participants according to their urolithin production because the analytical method used did not allow us to differentiate between urolithin A and isourolitin A.
Moreover, although most participants excreted higher amounts of sulfate-conjugated phenolic metabolites than glucuronide-conjugated forms, notable inter-individual variability was also evident within sulfate conjugates. Based on the same tertile approach, participants were categorized into low (32%), medium (32%), and high (36%) sulfate-conjugated phenolic metabolite producers, with the corresponding mean excretion values of 94 µmol, 167 µmol, and 275 µmol, respectively.
Discussion
(Poly)phenol metabolism has been widely studied in several intervention studies, which have evaluated the pharmacokinetics and urinary excretion of phenolic metabolites generated after the consumption of (poly)phenol-rich extracts or foods.30,31 However, there is a lack of information on the excretion profile of specific target populations like postmenopausal women. This work contributed to a better understanding of the metabolism of (poly)phenols in this population. Indeed, metabolomic analysis of 24 h urine samples allowed us to identify and quantify 116 phenolic metabolites derived from microbiota-mediated breakdown and/or human phase II conjugation of dietary phenolic compounds. The intake of PP-rich foods for 2 months showed the capability of modulating the excretion profile of phenolic metabolites in postmenopausal women, increasing PVLs, urolithins, and benzoic acids. All these phenolic metabolites are derived from the microbial metabolism of the native compounds found in the PP-rich foods provided during the intervention, which included 85% dark chocolate, green tea, and a fruit juice made of pomegranate, berries, and orange.These findings demonstrate the potential ability of a (poly)phenol-rich experimental diet to increase the concentration of circulating phenolic metabolites that may exert beneficial effects in reducing the risk of CMDs in postmenopausal women. Moreover, the results confirm the pivotal role of the gut microbiota in the biotransformation that (poly)phenols undergo in humans. Furthermore, a notable inter-individual variability in the urinary excretion of phenolic metabolites was observed, showing three phenolic-metabolite-producer phenotypes (low, medium, and high). Previous studies have also reported substantial inter-individual differences in phenolic metabolite production following the consumption of PP-rich foods. For instance, Vallejo et al.29 stratified participants into low, medium, and high excretors of phase II flavanone metabolites and phenolic acids following orange juice consumption. These variations were found to correlate with differences in the participants’ gut microbiota profiles.11,12
Although our current study focuses on postmenopausal women, it is worth noting that hormonal status, microbiota composition and metabolic capacity may differ considerably in premenopausal women, and thus urinary phenolic metabolites’ excretion profiles may also differ. Previous research on (poly)phenol absorption and hormonal status is limited and inconsistent. In this context, García-Villalba et al.32 reported the differences in plasma pharmacokinetic and urinary excretion of hydroxytyrosol metabolites after the acute intake of an oleuropein-rich olive leaf extract by pre- and postmenopausal women. Postmenopausal women showed higher plasma absorption of olive phenolic metabolites, while premenopausal women excreted more sulfated compounds in urine, suggesting that hormone-associated changes may influence (poly)phenol metabolism and elimination, likely due to age-related declines in phase II enzyme expression. However, in another study, Faughnan et al.33 found similar urine excretion of daidzein independent of the age or hormonal status.
After the 2-month dietary intervention, phase II derivatives of flavan-3-ols, PVLs, and PVAs were the metabolites that increased the most in urine. These phenolic metabolites are derived explicitly from the metabolic pathway of flavan-3-ols34,35 and, in fact, two PVLs [i.e., 5-(4′-hydroxyphenyl)-γ-valerolactone-3′-glucuronide and 5-(hydroxyphenyl)-γ-valerolactone-3′-sulfate] have been proposed as biomarkers of the intake of flavan-3-ol-rich foods such as green tea, cocoa and cranberry.30,36–38 Since more than 50% of the (poly)phenols ingested were flavan-3-ols via dark chocolate and green tea,19 a significant increase in specific metabolites of this (poly)phenol class was expected after the dietary intervention. Our findings are in line with those reported by Hidalgo-Liberona et al.,31 who showed a significant increase in the urinary excretion of gut microbiota and phase II-derived metabolites of flavan-3-ols, PVLs, and PVAs following a 4-week dietary intervention similar to that of the present study, but in older subjects.
A significant increase in the excretion of phase II derivatives of urolithins was also observed after the 2-month dietary intervention. Urolithins derived from the microbial catabolism of ellagitannins and ellagic acids present in the fruit juice supplemented, which contained pomegranate and berries, including blackcurrants.19 These findings are supported by several studies showing that urolithins are the main biomarkers of the consumption of foods containing ellagitannins and ellagic acids, such as pomegranate and some berries.39–41 However, a great inter-individual variability in urolithin excretion was also reported in the literature, which has led to the identification of urolithin metabotypes.42,43 In this context, three metabotypes have been identified based on urolithin production: urolithin metabotype A (UM-A), including individuals producing only 3,8-dihydroxy-urolithin (urolithin A); urolithin metabotype B (UM-B), including individuals characterized by the excretion of 3,8-dihydroxy-urolithin (urolithin A), 3,9-dihydroxy-urolithin (isourolithin A), and 3-hydroxy-urolithin (urolithin B); and urolithin metabotype 0 (UM-0), with no production of urolithins.42 In the present study, although 24 out of 25 participants were able to produce urolithins, we were unable to stratify them according to their metabotype due to the method limitations, as mentioned previously.
The intake of 85% dark chocolate, green tea, and fruit juice also increased the urinary excretion of non-specific metabolites, including benzoic acids. Since benzoic acids are the primary phenolic metabolites generated after the consumption of anthocyanin-rich foods,44–46 this result may suggest that native benzoic acids and anthocyanins present in fruit juice contributed to the significant increase in benzoic acids observed. However, other (poly)phenol classes, including flavan-3-ols [i.e., (−)-epicatechin],47 flavanones [i.e., naringenin-7-rutinoside],48 and flavonols [i.e., quercetin-3-glucoside]49 could also have influenced the significant increase in the urinary excretion of benzoic acids. Similarly, other phase II derivatives of non-specific metabolites, including phenylpropanoic acids, phenylacetic acids, and benzenes, may result from cocoa-based products,38 green tea,37,50 and orange juice,51 products being part of the PP-rich food portfolio of this study.
Our findings suggest that phase II derivatives of flavan-3-ols, PVLs, PVAs, benzoic acids, and urolithins are the main phenolic metabolites in circulation after daily consumption of dark chocolate, green tea, and mix-fruit juice, and could be responsible for the potential beneficial effects of PP-rich foods on cardiometabolic health in postmenopausal women. According to the scientific literature, PVLs and PVAs are known to decrease the expression of inflammatory and cell adhesion biomarkers (i.e., TNF-α, sVCAM-1 and sICAM-1).52,53 Urolithins have been recognized for their ability to control blood pressure, reduce the atherosclerosis, and improve the insulin sensitivity,54–58 while benzoic acids exhibit anti-inflammatory and antithrombotic properties.59–61 Therefore, the phenolic metabolites excreted in the urine of the participants of the present study are expected to be responsible for the potential beneficial effects of PP-rich food consumption on the reduction of the risk of CMDs in postmenopausal women. Further studies are needed to corroborate the findings found in the present study for elucidating the mechanisms of action of these phenolic metabolites in promoting health and determine the optimal (poly)phenol dose required to produce phenolic metabolites capable of exerting protective effects. Furthermore, future research should explore the potential synergistic effects derived from the consumption of various PP-rich foods capable of generating a wide range of phenolic metabolites. Investigating these synergies among different (poly)phenol classes could be helpful in the development of dietary patterns aimed at improving the health of the general population and of specific at-risk subgroups such as postmenopausal women. This is particularly relevant given that (poly)phenols are widely distributed in many plant-based foods, which are often consumed together throughout the day.
Conclusions
The phenolic metabolite urinary profile of postmenopausal women was modulated after the 2-month dietary intervention with PP-rich foods, due to the significant increase in the excretion of phenolic metabolites, including phase II derivatives of flavan-3-ols, PVLs, PVAs, benzoic acids, and urolithins. These metabolites could be the bioactive compounds responsible for the potential beneficial effects derived from the daily intake of dark chocolate, green tea, and fruit juice in reducing the risk of CMDs in postmenopausal women.Nevertheless, due to the inter-individual variability observed in response to PP-rich food intake and the need for microbiota-related information, future research should focus on identifying specific phenolic metabotypes and enterotypes that may influence the beneficial effects of dietary (poly)phenols on cardiometabolic parameters in this category of subjects. This future research will contribute to the improvement of the scientific understanding of the protective role of (poly)phenols against CMDs for specific population groups.
Author contributions
Conceptualization, M. J. P., and R. G. B.; methodology, L. S. M., N. T., L. B., P. M., M. J. P., and R. G. B.; formal analysis, L. S. M., N. T., N. L. B., and R. G. B.; investigation, L. S. M., N. T., N. L. B., L. B., P. M., and R. G. B.; resources, D. D. R., M. J. P., P. M., and R. G. B.; data curation, L. S. M., P. M., and R. G. B.; writing – original draft preparation, L. S. M. and R. G. B.; writing – review and editing, L. S. M., N. T., D. D. R., P. M., and R. G. B.; supervision, P. M., and R. G. B.; project administration, M. J. P.; funding acquisition, D. D. R., M. J. P., P. M, and R. G. B. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated for this article are available in the Digitum repository at https://hdl.handle.net/10201/159069.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04088d.
Acknowledgements
We thank all the volunteers for their participation in the study. The authors also want to thank Fundación Séneca (Agencia de Ciencia y Tecnología de la Región de Murcia, Spain) for funding the project (CARDIOGENE project, grant agreement 20904/PI/2018), Fundación Robles Chillida (Murcia, Spain) for the financial help provided to L. S. M., and the Spanish Ministry of Universities for the grant provided to R. G. B. within the program ‘Requalification of the Spanish University System 2021–2023’ (Next Generation EU funds). This work was also supported by the European Research Council (ERC) under the European Union's Horizon 2020 Research and Innovation Programme (PREDICT-CARE project, grant agreement no. 950050).References
- K. L. Marlatt, D. R. Pitynski-Miller, K. M. Gavin, K. L. Moreau, E. L. Melanson and N. Santoro, et al., Body composition and cardiometabolic health across the menopause transition, Obesity, 2022, 30(1), 14–27 Search PubMed . Available from: https://onlinelibrary.wiley.com/doi/10.1002/oby.23289.
- R. E. Nappi, P. Chedraui, I. Lambrinoudaki and T. Simoncini, Menopause: a cardiometabolic transition, Lancet Diabetes Endocrinol., 2022, 10(6), 442–456, DOI:10.1016/S2213-8587(22)00076-6.
- A. H. E. M. Maas, G. Rosano, R. Cifkova, A. Chieffo, D. van Dijken and H. Hamoda, et al., Cardiovascular health after menopause transition, pregnancy disorders, and other gynaecologic conditions: a consensus document from European cardiologists, gynaecologists, and endocrinologists, Eur. Heart J., 2021, 42, 967–984 Search PubMed . Available from: https://academic.oup.com/eurheartj/article/42/10/967/6120040.
- R. De Marchi, C. M. Dell'Agnolo, T. C. R. Lopes, A. A. F. Gravena, M. D. O. Demitto and S. C. R. Brischiliari, et al., Prevalence of metabolic syndrome in pre- and postmenopausal women, Arch. Endocrinol. Metab., 2017, 61(2), 160–166 Search PubMed.
- A. Rodriguez-Mateos, M. Le Sayec and A. Cheok, Dietary (poly)phenols and cardiometabolic health: from antioxidants to modulators of the gut microbiota, Proc. Nutr. Soc., 2024, 1–11 Search PubMed . Available from: https://www.cambridge.org/core/product/identifier/S0029665124000156/type/journal_article.
- R. D. Mendonça, N. C. Carvalho, J. M. Martin-Moreno, A. M. Pimenta, A. C. S. Lopes and A. Gea, et al., Total polyphenol intake, polyphenol subtypes and incidence of cardiovascular disease: The SUN cohort study, Nutr., Metab. Cardiovasc. Dis., 2019, 29(1), 69–78, DOI:10.1016/j.numecd.2018.09.012.
- C. Tang, X. Wang, L. Q. Qin and J. Y. Dong, Mediterranean diet and mortality in people with cardiovascular disease: A meta-analysis of prospective cohort studies, Nutrients, 2021, 13(8), 2623, DOI:10.3390/nu13082623.
- N. Becerra-Tomás, S. Blanco Mejía, E. Viguiliouk, T. Khan, C. W. C. Kendall and H. Kahleova, et al., Mediterranean diet, cardiovascular disease and mortality in diabetes: A systematic review and meta-analysis of prospective cohort studies and randomized clinical trials, Crit. Rev. Food Sci. Nutr., 2020, 60(7), 1207–1227, DOI:10.1080/10408398.2019.1565281.
- A. Tresserra-Rimbau, A. Medina-Remón, J. Pérez-Jiménez, M. A. Martínez-González, M. I. Covas and D. Corella, et al., Dietary intake and major food sources of polyphenols in a Spanish population at high cardiovascular risk: The PREDIMED study, Nutr., Metab. Cardiovasc. Dis., 2013, 23(10), 953–959 CrossRef CAS PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0939475312002451.
- R. D. Mendonça, N. C. Carvalho, J. M. Martin-Moreno, A. M. Pimenta, A. C. S. Lopes and A. Gea, et al., Total polyphenol intake, polyphenol subtypes and incidence of cardiovascular disease: The SUN cohort study, Nutr., Metab. Cardiovasc. Dis., 2019, 29(1), 69–78 Search PubMed . Available from: https://linkinghub.elsevier.com/retrieve/pii/S0939475318302953.
- C. Favari, J. F. Rinaldi de Alvarenga, L. Sánchez-Martínez, N. Tosi, C. Mignogna and E. Cremonini, et al., Factors driving the inter-individual variability in the metabolism and bioavailability of (poly)phenolic metabolites: A systematic review of human studies, Redox Biol., 2024, 71, 103095, DOI:10.1016/j.redox.2024.103095.
- L. Narduzzi, V. Agulló, C. Favari, N. Tosi, C. Mignogna and A. Crozier, et al., (Poly)phenolic compounds and gut microbiome: new opportunities for personalized nutrition, Microbiome Res. Rep., 2022, 1(2), 16 Search PubMed.
- N. Tosi, L. Bresciani, C. Favari, A. Rosi, R. Dodi and A. Bertelé, et al., How does the flavan-3-ol structure condition the quali-quantitative production of phenolic metabolites? Insights from a 3-arm intervention, Food Chem., 2025, 491, 145207, DOI:10.1016/j.foodchem.2025.145207.
- P. Mena, C. Favari, A. Acharjee, S. Chernbumroong, L. Bresciani and C. Curti, et al., Metabotypes of flavan-3-ol colonic metabolites after cranberry intake: elucidation and statistical approaches, Eur. J. Nutr., 2022, 61(3), 1299–1317, DOI:10.1007/s00394-021-02692-z.
- J. A. Santos-Marcos, O. A. Rangel-Zuñiga, R. Jimenez-Lucena, G. M. Quintana-Navarro, S. Garcia-Carpintero and M. M. Malagon, et al., Influence of gender and menopausal status on gut microbiota, Maturitas, 2018, 116, 43–53 CrossRef PubMed.
- B. A. Peters, J. Lin, Q. Qi, M. Usyk, C. R. Isasi and Y. Mossavar-Rahmani, et al., Menopause Is Associated with an Altered Gut Microbiome and Estrobolome, with Implications for Adverse Cardiometabolic Risk in the Hispanic Community Health Study/Study of Latinos, mSystems, 2022, 7(3), 00273–22, DOI:10.1128/msystems.00273-22.
- T. C. A. da Silva, J. A. G. Oliveira, L. Â. G. de Moraes, I. Rosse, A. Góes-Neto and V. A. de Carvalho Azevedo, et al., Microbiota and Postmenopause: The resilience of intestinal bacteria in the face of female hormonal aging, PLoS One, 2025, 20, 1–19, DOI:10.1371/journal.pone.0324712.
- V. Singh, Y. J. Park, G. D. Lee, T. Unno and J. H. Shin, Dietary regulations for microbiota dysbiosis among post-menopausal women with type 2 diabetes, Crit. Rev. Food Sci. Nutr., 2023, 63(29), 9961–9976, DOI:10.1080/10408398.2022.2076651.
- L. Sánchez-Martínez, R. González-Barrio, J. García-Alonso, P. Mena and M. J. Periago, Assessing the Impact of (Poly)phenol-Rich Foods on Cardiometabolic Risk in Postmenopausal Women: A Dietary Trial, Antioxidants, 2024, 13(8), 973 CrossRef PubMed . Available from: https://www.mdpi.com/2076-3921/13/8/973.
- M. Domínguez-Fernández, Y. Xu, P. Young Tie Yang, W. Alotaibi, R. Gibson and W. L. Hall, et al., Quantitative Assessment of Dietary (Poly)phenol Intake: A High-Throughput Targeted Metabolomics Method for Blood and Urine Samples, J. Agric. Food Chem., 2021, 69(1), 537–554, DOI:10.1021/acs.jafc.0c07055.
- F. Castello, G. Costabile, L. Bresciani, M. Tassotti, D. Naviglio and D. Luongo, et al., Bioavailability and pharmacokinetic profile of grape pomace phenolic compounds in humans, Arch. Biochem. Biophys., 2018, 646, 1–9, DOI:10.1016/j.abb.2018.03.021.
- C. D. Kay, M. N. Clifford, P. Mena, G. J. McDougall, C. Andres-Lacueva and A. Cassidy, et al., Recommendations for standardizing nomenclature for dietary (poly)phenol catabolites, Am. J. Clin. Nutr., 2020, 112(4), 1051–1068 Search PubMed.
- C. Curti, M. N. Clifford, C. D. Kay, P. Mena, A. Rodriguez-Mateos and D. Del Rio, et al., Extended recommendations on the nomenclature for microbial catabolites of dietary (poly)phenols, with a focus on isomers, Food Funct., 2025, 16(10), 3963–4000 RSC . Available from: https://xlink.rsc.org/?DOI=D4FO06152G.
- P. Mena, C. Favari, A. Acharjee, S. Chernbumroong, L. Bresciani and C. Curti, et al., Metabotypes of flavan-3-ol colonic metabolites after cranberry intake: elucidation and statistical approaches, Eur. J. Nutr., 2022, 61(3), 1299–1317, DOI:10.1007/s00394-021-02692-z.
- Z. Pang, J. Chong, G. Zhou, D. A. De Lima Morais, L. Chang and M. Barrette, et al., MetaboAnalyst 5.0: Narrowing the gap between raw spectra and functional insights, Nucleic Acids Res., 2021, 49(W1), W388–W396 CrossRef CAS PubMed.
- Z. Pang, G. Zhou, J. Ewald, L. Chang, O. Hacariz and N. Basu, et al., Using MetaboAnalyst 5.0 for LC–HRMS spectra processing, multi-omics integration and covariate adjustment of global metabolomics data, Nat. Protoc., 2022, 17(8), 1735–1761 CrossRef CAS PubMed.
- M. N. Clifford, I. A. Ludwig, G. Pereira-Caro, L. Zeraik, G. Borges and T. M. Almutairi, et al., Exploring and disentangling the production of potentially bioactive phenolic catabolites from dietary (poly)phenols, phenylalanine, tyrosine and catecholamines, Redox Biol., 2024, 71, 103068, DOI:10.1016/j.redox.2024.103068.
- M. N. Clifford and N. Kuhnert, LC–MS Characterization and Quantification of Known and Unknown (Poly)phenol Metabolites—Possible Pitfalls and Their Avoidance, Mol. Nutr. Food Res., 2022, 66(21), 1–10 CrossRef PubMed.
- F. Vallejo, M. Larrosa, E. Escudero, M. P. Zafrilla, B. Cerdá and J. Boza, et al., Concentration and solubility of flavanones in orange beverages affect their bioavailability in humans, J. Agric. Food Chem., 2010, 58(10), 6516–6524 Search PubMed.
- C. Favari, P. Mena, C. Curti, G. Istas, C. Heiss and D. Del Rio, et al., Kinetic profile and urinary excretion of phenyl-γ-valerolactones upon consumption of cranberry: a dose–response relationship, Food Funct., 2020, 11(5), 3975–3985 Search PubMed . Available from: http://xlink.rsc.org/?DOI=D0FO00806K.
- N. Hidalgo-Liberona, R. González-Domínguez, E. Vegas, P. Riso, C. Del Bo’ and S. Bernardi, et al., Increased Intestinal Permeability in Older Subjects Impacts the Beneficial Effects of Dietary Polyphenols by Modulating Their Bioavailability, J. Agric. Food Chem., 2020, 68(44), 12476–12484 CrossRef CAS PubMed . Available from: https://pubs.acs.org/doi/10.1021/acs.jafc.0c04976.
- R. García-Villalba, M. Larrosa, S. Possemiers, F. A. Tomás-Barberán and J. C. Espín, Bioavailability of phenolics from an oleuropein-rich olive (Olea europaea) leaf extract and its acute effect on plasma antioxidant status: comparison between pre- and postmenopausal women, Eur. J. Nutr., 2014, 53(4), 1015–1027 CrossRef . Available from: https://link.springer.com/10.1007/s00394-013-0604-9.
- M. S. Faughnan, A. Hawdon, E. Ah-Singh, J. Brown, D. J. Millward and A. Cassidy, Urinary isoflavone kinetics: the effect of age, gender, food matrix and chemical composition, Br. J. Nutr., 2004, 91(4), 567–574 CrossRef CAS PubMed.
- G. Di Pede, L. Bresciani, F. Brighenti, M. N. Clifford, A. Crozier and D. Del Rio, et al., In Vitro Faecal Fermentation of Monomeric and Oligomeric Flavan-3-ols: Catabolic Pathways and Stoichiometry, Mol. Nutr. Food Res., 2022, 2101090 Search PubMed.
- N. Tosi, L. Mancabelli, G. Alessandri, F. Turroni, M. Ventura and D. Del Rio, et al., Optimization of an In Vitro Colonic Fermentation: Insights into Flavan-3-ol Catabolism and Microbiota Modulation, J. Agric. Food Chem., 2025, 16429–16443, DOI:10.1021/acs.jafc.5c06932.
- P. Mena, L. Bresciani, N. Brindani, I. A. Ludwig, G. Pereira-Caro and D. Angelino, et al., Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity, Nat. Prod. Rep., 2019, 36(5), 714–752 Search PubMed.
- P. Mena, I. A. Ludwig, V. B. Tomatis, A. Acharjee, L. Calani and A. Rosi, et al., Inter-individual variability in the production of flavan-3-ol colonic metabolites: preliminary elucidation of urinary metabotypes, Eur. J. Nutr., 2019, 58(4), 1529–1543, DOI:10.1007/s00394-018-1683-4.
- M. Gómez-Juaristi, B. Sarria, S. Martínez-López, L. B. Clemente and R. Mateos, Flavanol bioavailability in two cocoa products with different phenolic content. A comparative study in humans, Nutrients, 2019, 11(7), 1–19 CrossRef PubMed.
- R. González-Barrio, G. Borges, W. Mullen and A. Crozier, Bioavailability of anthocyanins and ellagitannins following consumption of raspberries by healthy humans and subjects with an ileostomy, J. Agric. Food Chem., 2010, 58(7), 3933–3939 CrossRef PubMed.
- B. Cerdá, F. A. Tomás-Barberán and J. C. Espín, Metabolism of Antioxidant and Chemopreventive Ellagitannins from Strawberries, Raspberries, Walnuts, and Oak-Aged Wine in Humans: Identification of Biomarkers and Individual Variability, J. Agric. Food Chem., 2005, 53(2), 227–235 Search PubMed . Available from: https://pubs.acs.org/doi/10.1021/jf049144d.
- R. García-Villalba, M. V. Selma, J. C. Espín and F. A. Tomás-Barberán, Identification of Novel Urolithin Metabolites in Human Feces and Urine after the Intake of a Pomegranate Extract, J. Agric. Food Chem., 2019, 67(40), 11099–11107 Search PubMed.
- F. A. Tomás-Barberán, A. González-Sarrías, R. García-Villalba, M. A. Núñez-Sánchez, M. V. Selma and M. T. García-Conesa, et al., Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status, Mol. Nutr. Food Res., 2017, 61(1), 1–35 Search PubMed . Available from: https://onlinelibrary.wiley.com/doi/10.1002/mnfr.201500901.
- A. González-Sarrías, R. García-Villalba, M. Romo-Vaquero, C. Alasalvar, A. Örem and P. Zafrilla, et al., Clustering according to urolithin metabotype explains the interindividual variability in the improvement of cardiovascular risk biomarkers in overweight-obese individuals consuming pomegranate: A randomized clinical trial, Mol. Nutr. Food Res., 2017, 61(5), 1–14 Search PubMed.
- L. Tian, Y. Tan, G. Chen, G. Wang, J. Sun and S. Ou, et al., Metabolism of anthocyanins and consequent effects on the gut microbiota, Crit. Rev. Food Sci. Nutr., 2019, 59(6), 982–991, DOI:10.1080/10408398.2018.1533517.
- I. Fernandes, A. Faria, V. de Freitas, C. Calhau and N. Mateus, Multiple-approach studies to assess anthocyanin bioavailability, Phytochem. Rev., 2015, 14(6), 899–919 Search PubMed . Available from: https://link.springer.com/10.1007/s11101-015-9415-3.
- R. González-Barrio, C. A. Edwards and A. Crozier, Colonic catabolism of ellagitannins, ellagic acid, and raspberry anthocyanins: In vivo and in vitro studies, Drug Metab. Dispos., 2011, 39(9), 1680–1688 Search PubMed.
- G. Di Pede, P. Mena, L. Bresciani, T. M. Almutairi, D. Del Rio and M. N. Clifford, et al., Human colonic catabolism of dietary flavan-3-ol bioactives, Mol. Aspects Med., 2022, 101107, DOI:10.1016/j.mam.2022.101107.
- G. Pereira-Caro, B. Fernández-Quirós, I. A. Ludwig, I. Pradas, A. Crozier and J. M. Moreno-Rojas, Catabolism of citrus flavanones by the probiotics Bifidobacterium longum and Lactobacillus rhamnosus, Eur. J. Nutr., 2018, 57(1), 231–242 Search PubMed.
- X. Zhu, G. Ding, S. Ren, J. Xi and K. Liu, The bioavailability, absorption, metabolism, and regulation of glucolipid metabolism disorders by quercetin and its important glycosides: A review, Food Chem., 2024, 458, 140262, DOI:10.1016/j.foodchem.2024.140262.
- S. Roowi, A. Stalmach, W. Mullen, M. E. J. Lean, C. A. Edwards and A. Crozier, Green tea flavan-3-ols: Colonic degradation and urinary excretion of catabolites by humans, J. Agric. Food Chem., 2010, 58(2), 1296–1304 Search PubMed.
- G. Pereira-Caro, T. Polyviou, I. A. Ludwig, A. M. Nastase, J. M. Moreno-Rojas and A. L. Garcia, et al., Bioavailability of orange juice (poly)phenols: The impact of short-term cessation of training by male endurance athletes, Am. J. Clin. Nutr., 2017, 106(3), 791–800 Search PubMed.
- D. Álvarez-Cilleros, M. E. López-Oliva, S. Ramos and M. Á. Martín, Preventive effect of cocoa flavanols against glucotoxicity-induced vascular inflammation in the arteria of diabetic rats and on the inflammatory process in TNF-α-stimulated endothelial cells, Food Chem. Toxicol., 2020, 146, 111824, DOI:10.1016/j.fct.2020.111824.
- E. M. Campos, P. Stehle and M. C. Simon, Microbial metabolites of flavan-3-ols and their biological activity, Nutrients, 2019, 11(10), 1–27 Search PubMed.
- J. Li, R. Liao, S. Zhang, H. Weng, Y. Liu and T. Tao, et al., Promising remedies for cardiovascular disease: Natural polyphenol ellagic acid and its metabolite urolithins, Phytomedicine, 2023, 116, 154867, DOI:10.1016/j.phymed.2023.154867.
- A. M. Toney, D. Fox, V. Chaidez, A. E. Ramer-Tait and S. Chung, Immunomodulatory role of urolithin a on metabolic diseases, Biomedicines, 2021, 9(2), 1–18 CrossRef PubMed.
- R. García-Villalba, J. A. Giménez-Bastida, A. Cortés-Martín, M. Á. Ávila-Gálvez, F. A. Tomás-Barberán and M. V. Selma, et al., Urolithins: a Comprehensive Update on their Metabolism, Bioactivity, and Associated Gut Microbiota, Mol. Nutr. Food Res., 2022, 66(21), 2101019 Search PubMed , Available from: https://onlinelibrary.wiley.com/doi/10.1002/mnfr.202101019.
- J. I. Jiménez-Loygorri, B. Villarejo-Zori, Á. Viedma-Poyatos, J. Zapata-Muñoz, R. Benítez-Fernández and M. D. Frutos-Lisón, et al., Mitophagy curtails cytosolic mtDNA-dependent activation of cGAS/STING inflammation during aging, Nat. Commun., 2024, 15(1), 830, DOI:10.1038/s41467-024-45044-1.
- A. M. Toney, R. Fan, Y. Xian, V. Chaidez, A. E. Ramer-Tait and S. Chung, Urolithin A, a Gut Metabolite, Improves Insulin Sensitivity Through Augmentation of Mitochondrial Function and Biogenesis, Obesity, 2019, 27(4), 612–620 Search PubMed.
- J. H. Choi and S. Kim, In Vitro Antithrombotic, Hematological Toxicity, and Inhibitor Studies of Protocatechuic, Isovanillic, and p-Hydroxybenzoic Acids from Maclura Tricuspidata (Carr.) Bur, Molecules, 2022, 27(11), 3496, DOI:10.3390/molecules27113496.
- G. Akbari, Molecular mechanisms underlying gallic acid effects against cardiovascular diseases: An update review, Avicenna J. Phytomed., 2020, 10(1), 11–23 CAS.
- S. J. Uddin, M. Afroz, S. M. N. K. Zihad, M. S. Rahman, S. Akter and I. N. Khan, et al., A Systematic Review on Anti-diabetic and Cardioprotective Potential of Gallic Acid: A Widespread Dietary Phytoconstituent, Food Rev. Int., 2022, 38(4), 420–439, DOI:10.1080/87559129.2020.1734609.
Footnote |
| † Co-last author. |
| This journal is © The Royal Society of Chemistry 2026 |