Open Access Article
Open Access ArticleRed quinoa hydrolysate as a plant-based therapeutic alternative for damage induced by high cadmium concentrations to the vascular system in rats
Samia Hassan Husein
Kanaan
 a,
Paola Zambelli
Moraes
a,
Paola Zambelli
Moraes
 a,
Katye Yasmin de Souza
de Oliveira
a,
Fernando
Barbosa
a,
Katye Yasmin de Souza
de Oliveira
a,
Fernando
Barbosa
 b,
José Eudes
Gomes Pinheiro
b,
José Eudes
Gomes Pinheiro
 c,
Franck Maciel
Peçanha
c,
Franck Maciel
Peçanha
 a,
Dalton Valentim
Vassallo
a,
Dalton Valentim
Vassallo
 d,
Marta
Miguel-Castro
d,
Marta
Miguel-Castro
 *e and
Giulia Alessandra
Wiggers
*e and
Giulia Alessandra
Wiggers
 *a
*a
aCardiovascular Physiology Laboratory, Universidade Federal do Pampa, BR 472, Km 592, Uruguaiana, Rio Grande do Sul, Brazil. E-mail: giuliapecanha@unipampa.edu.br
bDepartment of Clinical Analyses, Toxicology and Food Sciences, School of Pharmaceutical Sciences of Ribeirão Preto, Universidade de São Paulo, Av. do Café s/n, 14049-903, Ribeirão Preto, São Paulo, Brazil
cPhysiotherapist, Empresa Brasileira de Serviços Hospitalares (EBSERH), Hospital das Clínicas, Universidade Federal de Uberlândia, Av. Pará, 1720 – Umuarama, Uberlândia – MG, 38405-320, Uberlândia, Brazil
dCardiac Electromechanical and Vascular Reactivity Laboratory, Universidade Federal do Espírito Santo, Marechal Campos, 1468, Vitória, Espírito Santo, Brazil
eInstituto de Ciencia y Tecnología de Alimentos y Nutrición (CSIC), C/Jose Antonio Novais, 6, 28040, Madrid, Spain. E-mail: marta.miguel@csic.es
First published on 20th March 2026
Abstract
Cadmium (Cd) is a highly toxic, non-essential element associated with cardiovascular dysfunction through oxidative stress, endothelial impairment, and inflammatory activation. Identifying plant-based dietary strategies, including quinoa, a low-cost pseudocereal with recognized bioactive properties, may offer a sustainable approach to mitigate these effects. This study aimed to characterize a bioactive red quinoa hydrolysate (RQH) and to investigate whether RQH prevents vascular damage induced by Cd exposure. Male Wistar rats were exposed to Cd with or without RQH treatment for 14 days. Co-treatment with RQH prevented the increase in systolic blood pressure (SBP) induced by Cd exposure. In the aorta and mesenteric resistance artery (MRA), RQH protected from increased contractile response while preserving nitric oxide (NO) bioavailability for vascular function. Additionally, RQH effectively mitigated oxidative stress and prevented the inflammatory cascade (COX-2 and TNF-alpha) resulting from Cd exposure. RQH could be used as a potential therapeutic dietary alternative for Cd-induced vascular damage. RQH exhibited antihypertensive, antioxidant, and anti-inflammatory properties, protecting against Cd-induced cardiovascular dysfunction.
1. Introduction
Living beings are continuously exposed to environmental contaminants from multiple sources, including cadmium (Cd), a toxic and non-essential element with a long biological half-life (25–30 years).1 Human exposure to Cd occurs through contaminated water, soil, and food, in addition to occupational activities, cigarette smoke, etc.2,3 In Brazil, environmental disasters have resulted not only in human casualties but also in severe ecological damage, with Cd concentrations in rivers and sediments reported to reach up to 70 times above regulatory limits.4The deleterious effects of Cd impact multiple organ systems, including the respiratory, renal, metabolic, nervous, reproductive, and cardiovascular systems.1 In the cardiovascular system, Cd has been associated with arterial hypertension, atherosclerosis, myocardial infarction, stroke, and heart failure, ultimately contributing to increased cardiovascular disease mortality.5,6 These effects are primarily attributed to Cd-induced vascular damage, which is highly dependent on dose and exposure duration. Cd exposure reduces endothelial nitric oxide availability, enhancing arterial vasoconstriction and sustaining a cycle of oxidative stress and inflammation in vascular tissue, as previously demonstrated in exposure to high doses of Cd.7–12
Given the human population's susceptibility to this metal contamination, the search for new therapeutic alternatives to mitigate cardiovascular damage induced by Cd is crucial. The side effects and limited accessibility of conventional treatments highlight the importance of exploring accessible, functional, and sustainable food alternatives that minimize environmental impact and preserve biodiversity.13,14
Pseudocereals, a group of underexploited foods, are nutrition-rich and have the potential to confer functional benefits due to their bioactive components, yet they remain underutilized in global markets.15 Quinoa (Chenopodium quinoa) is one of the principal representatives of pseudocereals, especially in Latin America, characterized by notable resistance to various abiotic stresses, including salinity, extreme temperatures, drought and nutrient-poor soils.16 Hydrolysis of quinoa proteins enhances their beneficial properties by generating bioactive peptides, and several studies have reported antioxidant, antihypertensive, and other bioactive properties for quinoa hydrolysates.17,18 However, in vivo evidence linking hydrolyzed quinoa to anti-inflammatory properties remains limited.
Recently, López-Moreno et al. (2023) demonstrated the antihypertensive and antioxidant properties of a red quinoa hydrolysate with alcalase (RQH) in spontaneously hypertensive rats (SHR).19 Considering the toxicity of Cd, and the need for natural therapeutic alternatives, RQH represents a promising natural bioactive agent for protecting against Cd-induced vascular damage. Therefore, this study aimed to investigate whether RQH treatment could protect the vascular system from damage caused by exposure to high doses of Cd, targeting key oxidative and inflammatory pathways activated by the metal. Additionally, peptide characterization of RQH was also performed, and potentially bioactive sequences were identified.
2. Methods
2.1. Animal care and protocols
Three-month-old male Wistar rats (∼350 g) were used. The animals were obtained from the Animal Facility of the Federal University of Pampa (UNIPAMPA) and maintained in an environment with constant temperature, humidity, and a 12 hour light–dark cycle, with free access to food and water in a dedicated enclosure. The experiments were conducted in accordance with the guidelines for biomedical research defined by the Brazilian Societies of Experimental Biology and approved by the Ethics Committee on the Use of Animals (CEUA) of the Federal University of Pampa, with protocol number 010/2023.The animals were randomly divided into 4 experimental groups (n = 10 per group) and treated for 14 days. During treatment, the animal's weight was monitored weekly, and daily feed and water intake were measured: (a) control: tap water via gavage + distilled water via intraperitoneal (i.p.); (b) cadmium (Cd): tap water via gavage + CdCl2 – 1 mg kg−1 i.p.20 (c) red quinoa hydrolysate (RQH): 1 g kg−1 day−1via gavage + distilled water i.p. (López-Moreno et al., 2023);19 and (d) cadmium + red quinoa hydrolysate (CdRQH): CdCl2 – 1 mg kg−1 i.p. + RQH 1 g kg−1 day−1via gavage. For daily administration via gavage in the experimental animals, the hydrolysate was resuspended in tap water.
2.2. Preparation of red quinoa hydrolysate (RQH)
Red quinoa seeds were purchased from Legumbres Astorga (Burgos, Spain). To obtain RQH, the quinoa was ground in a stone grinder (Conasi, Jaén, Spain), and the resulting flour was diluted in distilled water in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12.5 (w/v). Subsequently, hydrolysis was performed with Alcalase 2.4 LFG (Subtilisin (E.C. 3.4.21.62) 2.4 AU-A g−1), a protease enzyme produced by the bacterium Bacillus licheniformis (Novozymes – Copenhagen, Denmark). This process lasted 2 h in a thermostatic bath, regulated at a temperature of 55 °C, with constant stirring. For enzymatic inactivation, the sample was brought to a temperature of 90 °C for 15 minutes. Afterwards, the hydrolysate was centrifuged at 4500 rpm for 15 minutes, and the supernatant was collected and frozen at −20 °C for later lyophilization.19 The lyophilized hydrolysate was stored under vacuum and frozen at −20 °C.
12.5 (w/v). Subsequently, hydrolysis was performed with Alcalase 2.4 LFG (Subtilisin (E.C. 3.4.21.62) 2.4 AU-A g−1), a protease enzyme produced by the bacterium Bacillus licheniformis (Novozymes – Copenhagen, Denmark). This process lasted 2 h in a thermostatic bath, regulated at a temperature of 55 °C, with constant stirring. For enzymatic inactivation, the sample was brought to a temperature of 90 °C for 15 minutes. Afterwards, the hydrolysate was centrifuged at 4500 rpm for 15 minutes, and the supernatant was collected and frozen at −20 °C for later lyophilization.19 The lyophilized hydrolysate was stored under vacuum and frozen at −20 °C.
2.3. Systolic blood pressure (SBP) and vascular experiments
Systolic blood pressure (SBP) analysis was performed weekly (days 0, 7 and 14) indirectly using the caudal plethysmography method (LE 5002/V03/200L, Panlab S.L., Barcelona, Spain), according to the protocol described by Buñag (1973).21 The animals were acclimated to the procedure for one week prior to the first measurement. For each final SBP value, 10 consecutive recordings were obtained per animal, and their mean was used for analysis.At the end of the treatment (15th day), the animals were anesthetized (ketamine – 87 mg kg−1, i.p., and xylazine 13 mg kg−1, i.p.) and then euthanized by exsanguination. Whole blood was collected by direct puncture of the abdominal aorta for analysis of Cd content and after centrifugation at 3500 rpm for 10 minutes at a temperature of 4 °C, plasma was obtained and stored at −20 °C for biochemical analyses. In addition, the aorta and third order branches of the mesenteric resistance arteries (MRA) were extracted for the vascular reactivity experiments, and biochemical and immunofluorescence analyses.
Both arteries were carefully dissected, and rings of 2–4 mm were cut and mounted on their respective systems. Vascular reactivity in the aorta was assessed using an organ bath system (TSD125BX8 – Biopac Systems, Inc.; CA) connected to a data acquisition system (MP150WSW-SYS – Biopac Systems, Inc.; CA), and a basal tension of 1.5 g was applied to the aortic rings. MRA were mounted on a small-vessel dual chamber wire myograph (Powerlab 8/35 ADInstruments Pty Ltd, Castle Hill, Australia). Isometric tension recording was performed according to the method described by Nielsen & Owman (1971)22 and Mulvany & Halpern (1977),23 respectively. After a 30-min stabilization period, the preparations were maintained in Krebs–Henseleit solution (KHS in mM: 115 NaCl, 25 NaHCO3, 11.1 glucose, 4.7 KCl, 2.5 CaCl2, 1.2 MgSO4 7H2O, 1.2 KH2PO4, and 0.01 Na2EDTA, pH 7.4), and continuously bubbled with a carbogenic mixture (95% O2 and 5% CO2). The segments underwent an assessment of functional integrity and were contracted with potassium chloride (KCl, 75 mM – aorta and 120 mM – MRA), and the maximum contraction was recorded. Then, the arteries were pre-contracted (at 50% of the maximum contraction performed with KCl) with phenylephrine (Phe – (R)-(−)-phenylephrine hydrochloride – Sigma-Aldrich Cod. P6126) in the aorta and noradrenaline (NE – NE-norepinephrine bitartrate salt – Cod. A0937) in MRA.
A relaxation curve to acetylcholine (ACh – acetylcholine chloride – Sigma-Aldrich Cod. A6625 – 0.1–1 mmol L−1) was plotted, and when the segments were able to relax at least 80% of the pre-contraction, the integrity of the endothelium was verified. Afterwards, concentration–response curves to Phe or NE were plotted and, to investigate the possible pathways involved, drugs such as a non-selective inhibitor of nitric oxide synthase (L-NAME – N-Nitro-L-arginine methyl ester – Sigma-Aldrich Cod. RV04 – 100 μM), a non-selective inhibitor of reactive oxygen species – ROS (Tiron – 4,5-dihydroxy-1,3-benzenedisulfonic acid disodium salt monohydrate – Sigma-Aldrich Cod. 172553 – 1 mmol L−1), an enzyme that converts the superoxide anion into hydrogen peroxide (SOD – superoxide dismutase – Sigma-Aldrich Cod. S5395 – 150 U mL−1) and the selective inhibitor of cyclooxygenase-2, COX-2 (NS 398 – Sigma-Aldrich Cod. N194 – 1 μM) were incubated. The independent endothelium relaxation was evaluated through a concentration–response curve to sodium nitroprusside (SNP – acetylcholine chloride – Sigma-Aldrich Cod. 71778 – 1–30 μmol L−1).
2.4. Cd content in the blood
Cd levels in blood were measured using inductively coupled plasma mass spectrometry (ICP-MS), according to the methodology described by Batista et al. (2009).24 Briefly, samples were diluted in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio into a 15 mL polypropylene Falcon® tube (Becton Dickinson) with a solution containing 0.01% (v/v) Triton® X-100, 0.5% (v/v) nitric acid and 10 μg L−1 of each one of the internal standards, Rh and Ir. In order to verify the accuracy and precision of the proposed method, the reference materials (RM) of human blood from the L'Institut National de Santé Publique du Quebec (Canada) were analyzed.
50 ratio into a 15 mL polypropylene Falcon® tube (Becton Dickinson) with a solution containing 0.01% (v/v) Triton® X-100, 0.5% (v/v) nitric acid and 10 μg L−1 of each one of the internal standards, Rh and Ir. In order to verify the accuracy and precision of the proposed method, the reference materials (RM) of human blood from the L'Institut National de Santé Publique du Quebec (Canada) were analyzed.
2.5. Immunofluorescence for detection of NOX1, p47-phox, eNOS, COX-2 and TNFα
Briefly, segments of the aorta and MRA were fixed in 4% paraformaldehyde, washed in PBS and 30% sucrose, embedded in Tissue Tek O.C.T., frozen in liquid nitrogen, and stored at −80 °C. Then, 14 µm sections were made using a cryostat (Leica – CM1860), mounted on gelatinized slides, and subjected to washings with PBS and permeabilization with heated sodium citrate buffer for protein reactivation. After blocking, the slides were incubated with primary antibodies (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400) (NOX-1 – Sigma-Aldrich Cod. SAB 2108601, eNOS – Sigma-Aldrich Cod. SAB4502016, p47-phox – Sigma-Aldrich Cod. SAB4502809, COX-2 – Sigma-Aldrich SAB 5700721, and TNF-α – Sigma-Aldrich SAB5700627) for 24 h in a cold chamber. Next, they were incubated with Alexa Fluor (aorta 1
400) (NOX-1 – Sigma-Aldrich Cod. SAB 2108601, eNOS – Sigma-Aldrich Cod. SAB4502016, p47-phox – Sigma-Aldrich Cod. SAB4502809, COX-2 – Sigma-Aldrich SAB 5700721, and TNF-α – Sigma-Aldrich SAB5700627) for 24 h in a cold chamber. Next, they were incubated with Alexa Fluor (aorta 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 and MRA 1
400 and MRA 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 – Sigma-Aldrich Cod. A11001) 488 nm secondary antibody and stained with DAPI (4′,6-diamidino-2-phenylindole – 1
500 – Sigma-Aldrich Cod. A11001) 488 nm secondary antibody and stained with DAPI (4′,6-diamidino-2-phenylindole – 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 – Sigma-Aldrich MBD0015) for nuclear staining. Negative controls omitted the primary antibody. Slides were mounted with Fluoromount-G and images were captured on the EVOS® Floid® Cell Imaging Station (40×). Fluorescence quantification was performed using ImageJ, with at least N = 06 captures per group. Merged images (MERGE) represent nuclei (blue/DAPI) and primary–secondary antibody binding (green).25
1000 – Sigma-Aldrich MBD0015) for nuclear staining. Negative controls omitted the primary antibody. Slides were mounted with Fluoromount-G and images were captured on the EVOS® Floid® Cell Imaging Station (40×). Fluorescence quantification was performed using ImageJ, with at least N = 06 captures per group. Merged images (MERGE) represent nuclei (blue/DAPI) and primary–secondary antibody binding (green).25
2.6. Biochemical analysis of oxidative stress parameters
Plasma samples were diluted with 50 mM Tris-HCl (pH 7.4) at a concentration of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. Aortic rings and MRA were homogenized with 50 mM Tris-HCl (pH 7.4) at a ratio of 1
10. Aortic rings and MRA were homogenized with 50 mM Tris-HCl (pH 7.4) at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (weight – tissue
10 (weight – tissue![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) volume – Tris) and centrifuged, and the supernatant was diluted with 50 mM Tris-HCl (1
volume – Tris) and centrifuged, and the supernatant was diluted with 50 mM Tris-HCl (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) and frozen at −20 °C for use in the following biochemical analyses of oxidative stress parameters.
10) and frozen at −20 °C for use in the following biochemical analyses of oxidative stress parameters.
Reactive oxygen species (ROS) levels in homogenates from aorta and mesenteric resistance artery (MRA) samples were measured using the fluorimetric method described by Loetchutinat et al. (2005).26 We also measured lipid peroxidation levels in plasma and arteries by quantifying thiobarbituric acid reactive substances (TBARS) using the colorimetric method described by Ohkawa et al. (1979).27 Finally, the total antioxidant capacity (FRAP – ferric reducing antioxidant potential) in plasma and arteries was assessed by the ability of the samples to reduce ferric ions (Fe3+) to ferrous ions (Fe2+), following the method described by Benzie & Strain (1996).28
2.7. Characterization of RQH
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, with survey scans (400–1600 amu) followed by twenty data-dependent MS/MS scans (Top 20) using a 2 u isolation width, 35% normalized collision energy, and a 60 s dynamic exclusion.29
000, with survey scans (400–1600 amu) followed by twenty data-dependent MS/MS scans (Top 20) using a 2 u isolation width, 35% normalized collision energy, and a 60 s dynamic exclusion.29
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 949 entries; UniProt 07/2024, decoy-fusion). Searches were conducted in the nonspecific digestion mode, allowing up to two missed cleavages, with mass tolerances of 20 ppm for precursor ions and 0.6 Da for MS/MS fragments. False discovery rates (FDRs) were set at 0.01 for peptide spectrum matches (PSMs) and proteins. Only proteins with at least two unique peptides identified via LC-MS/MS were considered.30
949 entries; UniProt 07/2024, decoy-fusion). Searches were conducted in the nonspecific digestion mode, allowing up to two missed cleavages, with mass tolerances of 20 ppm for precursor ions and 0.6 Da for MS/MS fragments. False discovery rates (FDRs) were set at 0.01 for peptide spectrum matches (PSMs) and proteins. Only proteins with at least two unique peptides identified via LC-MS/MS were considered.30
Peptide identification began with the selection of molecular ions in the hydrolysate. Precursor ions, either singly or doubly charged, were fragmented, generating a series of fragments that allowed amino acid sequence determination. The obtained masses and partial sequences from ESI-MS/MS were matched against the Chenopodium quinoa protein database, considering enzyme specificity. Peptides were classified based on the agreement between theoretical and experimental data. Each identified peptide was assigned to its source protein with a confidence score. This study identified several RQH peptide sequences, selecting those with potential antioxidant activity based on structural analysis.
2.8. Statistical analysis
The results are expressed as mean ± standard error of the mean (SEM). Aorta vasoconstrictor responses are expressed as a percentage of the contraction induced by KCl, and MRA vasoconstrictor responses were expressed as mN mm−1. Vasodilator responses to ACh or SNP were expressed as a percentage of relaxation of the pre-contraction induced by Phe or NE. Two-way analysis of variance (ANOVA) for repeated measures was performed to analyze the results. When the ANOVA showed statistical significance, the Bonferroni post-hoc test was used. The results were considered statistically significant for p-values <0.05. Statistical analyses were performed using Prism Graph 8.0 software (GraphPad Software, San Diego, CA, United States).3. Results
At the beginning of the treatment, the animals in all groups studied exhibited similar body weights. By the end of the treatment, rats exposed to high doses of Cd had significantly reduced body weights compared to the beginning of the treatment and compared to the control group. Furthermore, cotreatment with RQH did not prevent the weight loss induced by the metal (initial body weight – control: 432.8 ± 18.2; RQH: 440.8 ± 11.9; Cd: 469.1 ± 20.3; CdRQH: 483.7 ± 19.3; final body weight – control: 467.0 ± 15.3; RQH: 455.6 ± 9.8; Cd: 379.4 ± 20.8*; CdRQH: 398.6 ± 15.9*, *vs. control – in g).Exposure to Cd for 14 days significantly increased blood Cd levels in the exposed animals. Cotreatment with RQH significantly reduced Cd levels when compared to the group that only received the metal; however, the levels remained elevated relative to the controls (control: 0.36 ± 0.01; RQH: 0.34 ± 0.02; Cd: 245.03 ± 11.02*; CdRQH: 199.21 ± 12.47*# μg L−1, *p < 0.05 vs. control; #p < 0.05 vs. Cd).
Water and feed consumption were reduced in rats exposed to Cd, and cotreatment with RQH (CdRQH group) significantly improved the water and feed intake (final mean daily feed consumption over the 14 day period – control: 27.1 ± 0.8; RQH: 26.0 ± 1.1; Cd: 8.4* ± 1.2; CdRQH: 16.2 ± 1.0#, in g day−1, *vs. control, #vs. Cd; final mean daily water consumption over the 14 day period – control: 56.0 ± 1.7; RQH: 50.5 ± 1.5; Cd: 18.9 ± 2.0*; CdRQH: 31.2 ± 2.0#, in mL day−1, *vs. control, #vs. Cd).
The initial SBP levels were similar between the groups and remained similar during the first 7 days of the experimental period (SBP day 0 – control: 124.5 ± 2.0; RQH: 123 ± 1.6; Cd: 123.3 ± 1.6; CdRQH: 124.8 ± 1.3 – in mmHg) (SBP day 7 – control: 123.6 ± 1.2; RQH: 122.7 ± 0.9; Cd: 123.7 ± 2.3; CdRQH: 120.3 ± 2.0 – in mmHg). However, at the end of the treatment, the animals of the Cd group exhibited a significant increase in SBP. Nevertheless, cotreatment with RQH prevented the increase of SBP induced by the metal, maintaining a level similar to the control group (SBP – control: 124.1 ± 1.0; RQH: 123.8 ± 1.5; Cd: 140.8 ± 2.6*; CdRQH: 122.9 ± 2.2#, *vs. control, #vs. Cd – in mmHg).
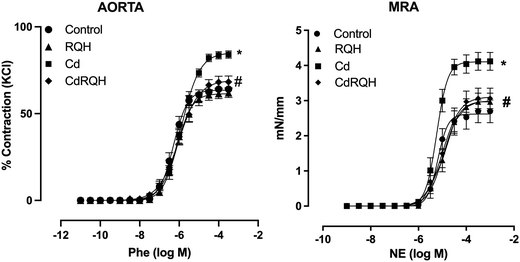
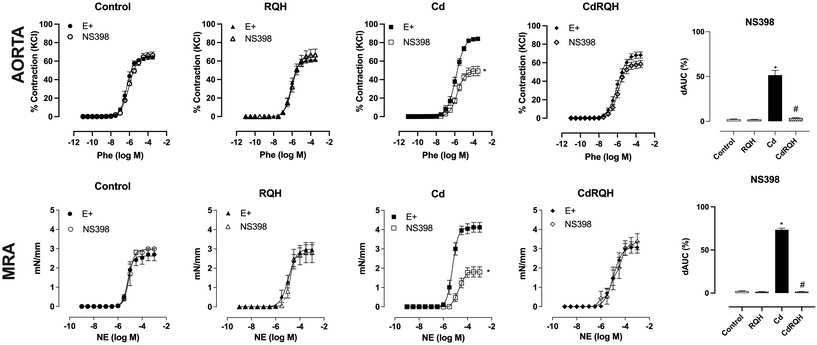
The contractile response to KCl in the aorta and MRA was similar in all groups (aorta – ΔKCl 75 mM – Ct: 2.0 ± 0.0; RQH: 2.0 ± 0.0; Cd: 1.9 ± 0.0; CdRQH: 2.1 ± 0.0 g and MRA – KCl 120 mM – Ct: 4.4 ± 0.3; RQH: 4.0 ± 0.2; Cd: 3.5 ± 0.3; CdRQH: 4.0 ± 0.2 mN mm−1). No differences were observed in the internal diameters of MRA among the groups (MRA – Ct: 258.9 ± 6.9; RQH: 282.1 ± 8.1; Cd: 255.1 ± 6.7; CdRQH: 259.2 ± 7.4 μm). There were no changes in the vasodilatory response, both endothelium dependent (ACh) and independent (NPS), in either artery, aorta, and MRA, of all experimental groups, as demonstrated by the absence of differences in maximal responses or sensitivity in both curves (SI Table). In contrast, the contractile response of both vessels of animals exposed only to Cd presented a significant increase in the maximal contractile response to Phe or NE, demonstrating vascular dysfunction promoted by Cd exposure. However, co-treatment with RQH prevented the increase in reactivity promoted by the metal, normalizing the contraction curve to control group levels (Fig. 1 and SI Table).
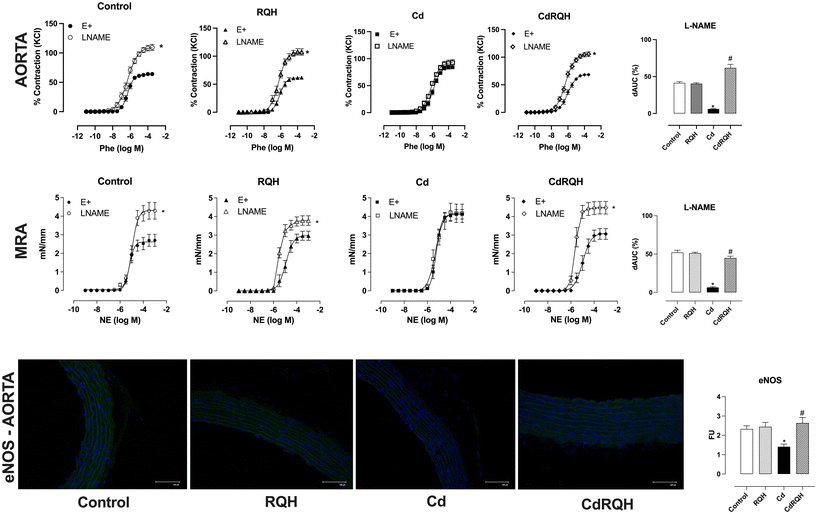
The possible pathways involved in the RQH vascular protection were investigated, focusing on the endothelium, which plays a key role in releasing substances that regulate vascular smooth muscle, primarily nitric oxide (NO). Vessels were incubated with L-NAME, a non-selective NO synthesis inhibitor. In both vascular beds, there was a significant reduction in the participation of NO in the contractile response to Phe/NE of animals exposed only to Cd (Cd group), indicating impairment of the nitric oxide (NO) pathway. Notably, co-treatment with RQH (CdRQH group) preserved the maintenance of NO bioavailability in both arteries, preventing dysfunction of this important vasorelaxant product derived from the endothelium (Fig. 2 and SI Table). To corroborate these functional findings and knowing the important participation of NO in the responses of the aorta, we investigated the expression of eNOS (endothelial nitric oxide synthase). In the aorta artery of Cd-exposed animals, a significant reduction in fluorescence was observed, indicating lower bioavailability of NO. In the arteries of the animals treated concomitantly with RQH, the fluorescence for eNOS was maintained, suggesting any impairment in the bioavailability of NO, reinforcing the functional finding (Fig. 2 – inferior panel).
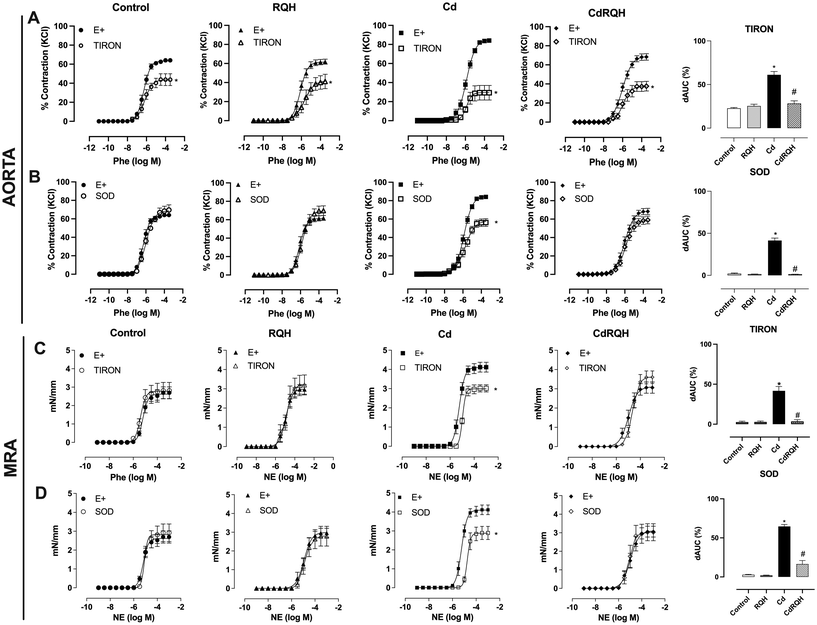
One of the main factors that reduces NO bioavailability in the vascular bed is the increased production of reactive oxygen species (ROS). Considering that the vascular damage caused by Cd exposure is directly associated with increased oxidative stress, and RQH has demonstrated antioxidant activity, we investigated the participation of ROS in the contractile response to Phe/NE. Incubation of aortic rings with Tiron, an ROS scavenger, reduced the contractile response to Phe in all groups studied. This reduction was significantly greater in the rings of animals exposed only to Cd, as clearly demonstrated in the graph of the area under the curve – dAUC (Fig. 3A and SI Table). In MRA, Tiron significantly decreased the maximum response to NE only in the Cd group, while no effect was observed in the other groups. Co-treatment with RQH provided vascular protection by preventing the increased participation of these reactive species in the contractile response of the vessels, reinforcing the potential antioxidant power for RQH (Fig. 3C and SI Table).
The superoxide anion (O2˙−) is a primary ROS that plays a central role in vascular oxidative stress in cadmium exposure models.9 To investigate its participation in the contractile response of the aorta and MRA, we incubated the arteries with SOD. SOD significantly reduced the contractile response in both vascular beds of animals exposed only to Cd and partially in the co-treated group (CdRQH) in the aorta, indicating the increased participation of O2˙−. Co-treatment with RQH completely prevented the participation of O2˙− in the contractile response in MRA and partially in the aorta, evidencing its antioxidant effect through O2˙− neutralization (Fig. 3B, D and SI Table).
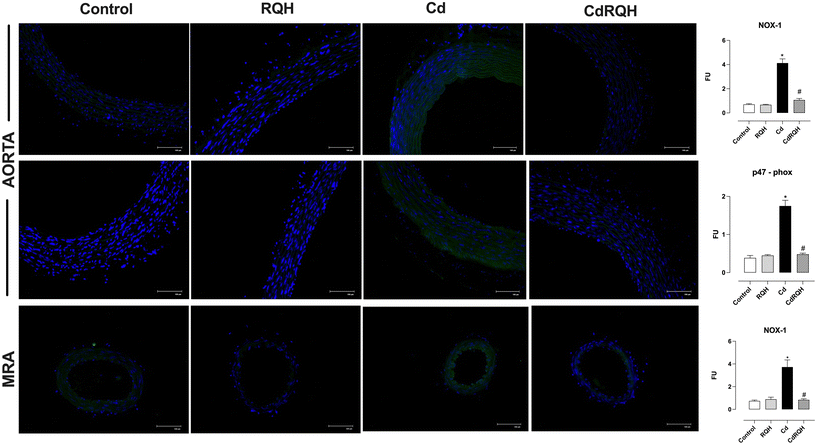
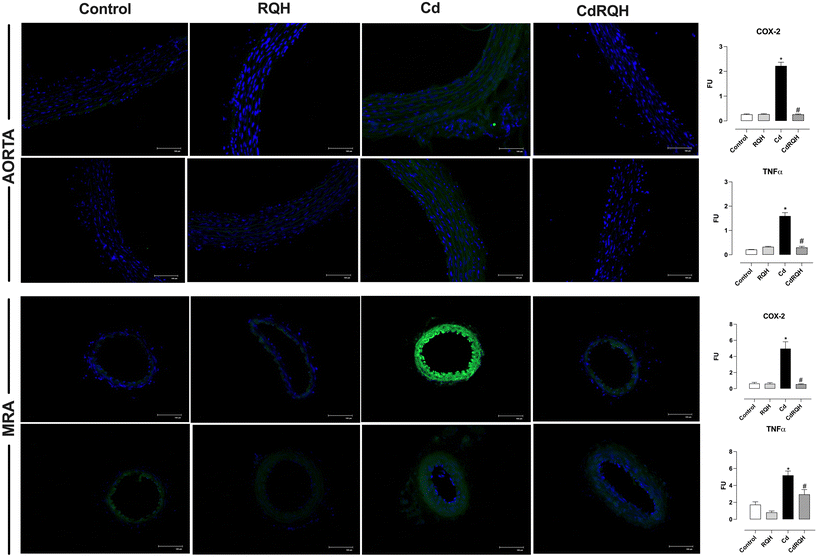
The NADPH oxidase enzyme complex is a major source of ROS in the vascular endothelium. Under exposure to Cd, this complex is overactivated, as previously demonstrated in this high-exposure model.9 Accordingly, we used immunofluorescence to evaluate the presence of NOX-1 and observed that co-treatment with RQH prevented the Cd-induced increase in NOX-1 fluorescence in both arteries (Fig. 4). Furthermore, p47-phox, a key regulatory subunit of the NADPH oxidase complex, expressed in aortic endothelial cells, was upregulated in Cd-exposed animals. Co-treatment with RQH significantly reduced the fluorescence emission of this subunit, supporting its antioxidant role (Fig. 4).
The biochemical parameters of oxidative stress corroborated the vascular functional findings. ROS levels were significantly elevated in the Cd group, and co-treatment with RQH prevented the increase of these harmful reactive species in the aorta and MRA. Lipid peroxidation, assessed by malondialdehyde (MDA) levels, was significantly increased in both arteries and in the plasma of animals exposed only to Cd. However, those co-treated with RQH efficiently maintained MDA levels similar to those of the control group. Evaluation of total antioxidant capacity revealed a significant increase in animals exposed only to Cd, while animals co-treated with RQH showed no increase in total antioxidant capacity in the aorta and MRA. No significant differences were observed in plasma between groups (Table 1).
| Control | RQH | Cd | CdRQH | |
|---|---|---|---|---|
| Data are presented as mean ± SEM (N = 10). Reactive oxygen species (ROS) are expressed as fluorescence units (FU), thiobarbituric acid reactive substances (TBARS) as nM per MDA, and ferric reducing antioxidant power (FRAP) as nM Trolox equivalents.a vs. Controlb vs. Cd (ANOVA-2 way, followed by the Bonferroni post-hoc test). | ||||
| Aorta | ||||
| ROS | 66.5 ± 3.5 | 65.3 ± 5.5 | 97.2 ± 6.8a | 64.1 ± 7.7b |
| TBARS | 0.249 ± 0.0 | 0.229 ± 0.0 | 0.372 ± 0.0a | 0.221 ± 0.0b |
| FRAP | 71.2 ± 17.3 | 132.5 ± 16.2 | 221.1 ± 20.4a | 97.1 ± 20.4b |
| MRA | ||||
| ROS | 85.3 ± 3.5 | 81.4 ± 3.8 | 149.0 ± 15.0a | 87.9 ± 6.8b |
| TBARS | 0.073 ± 0.0 | 0.081 ± 0.0 | 0.168 ± 0.0a | 0.102 ± 0.0b |
| FRAP | 74.4 ± 8.0 | 45.0 ± 5.6 | 162.0 ± 10.3a | 60.2 ± 6.7b |
| Plasma | ||||
| TBARS | 0.090 ± 0.0 | 0.093 ± 0.0 | 0.161 ± 0.0a | 0.076 ± 0.0b |
| FRAP | 10.4 ± 1.3 | 12.4 ± 1.1 | 9.5 ± 1.6 | 9.5 ± 2.0 |
Given RQH's vascular protection against Cd-induced damage, attributed to its antioxidant properties and the well-established link between oxidative stress and inflammatory signaling (COX-2 and cytokines), we assessed whether this pathway was also modulated by RQH treatment. The incubation of aorta and MRA rings with the selective COX-2 inhibitor, NS398, significantly reduced the contractile response of both arteries, indicating an increased participation of COX-2 and its derivatives in aorta and MRA vasoconstriction. Notably, co-treatment with RQH (CdRQH group) prevented the increased participation of COX-2 in the contractile response in both vessels, demonstrating, for the first time, vascular anti-inflammatory properties of this hydrolysate (Fig. 5).
Immunofluorescence analysis of COX-2 directly in the vascular bed revealed a significant increase in fluorescence in the Cd group, and co-treatment with RQH prevented the increase and activation of this inflammatory cascade in the aorta and MRA, corroborating the functional data observed. Once we identified that oxidative stress and the inflammatory pathway are the main mechanisms of Cd-induced damage, and that COX-2 induction may be due to the activation of inflammatory cytokines such as TNF-alpha, we also investigated the expression of this factor in the aorta and MRA. Fluorescence for TNF-α was increased in the Cd group, and the co-treatment with RQH prevented the exacerbation of this factor in the vascular bed of the aorta and MRA, further supporting the potential RQH anti-inflammatory effects (Fig. 6).
As a final step of this study, we aimed to demonstrate that RQH contains peptide sequences with biological properties capable of explaining the effects observed in our study. Several peptide sequences from RQH treated with alcalase for 2 hours were identified in this study. Based on studies of structure–activity reported in the literature, twelve peptide sequences with potential antioxidant, anti-inflammatory, and/or chelating activity were selected (Table 2). The predicted effects of each peptide are inferred from their amino acid composition, structure, and positioning of its amino acids, particularly the terminal residues.31 The antioxidant potential extensively reported for other hydrolysates is primarily associated with the presence of histidine (H), tyrosine (Y), and phenylalanine (F).32 Additionally, anti-inflammatory effects have been linked with aspartate,33 while the metal-chelating potential is associated with glutamate.34 Interestingly, none of the selected peptides had been previously described in the literature.
| Sequence | Molecular mass (Da) | Potential effect |
|---|---|---|
| Amino acids found in the presented sequences: A: alanine; D: aspartate; E: glutamate; F: phenylalanine; G: glycine; H: histidine; I: isoleucine; K: lysine; L: leucine; P: proline; Q: glutamine; R: arginine; T: threonine; V: valine; Y: tyrosine. | ||
| FDVGVK | 66![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 336 336 |
Antioxidant, anti-inflammatory |
| KLYGPE | 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 537 537 |
Antioxidant, anti-inflammatory, chelating |
| PIYEGY | 74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 034 034 |
Antioxidant, anti-inflammatory, chelating |
| TETVRFF | 89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 845 845 |
Antioxidant, anti-inflammatory |
| VDHPVFL | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 544 544 |
Antioxidant, anti-inflammatory, chelating |
| VPIYEGY | 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 941 941 |
Antioxidant, anti-inflammatory, chelating |
| YIRRGEAKL | 110![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 464 464 |
Antioxidant, anti-inflammatory, chelating |
| YPIIADPK | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 551 551 |
Antioxidant, anti-inflammatory |
| YTEQPL | 74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 936 936 |
Antioxidant, anti-inflammatory, chelating |
4. Discussion
Our findings suggested that co-treatment with RQH prevented Cd-induced cardiovascular damage by reducing the increase in systolic blood pressure. In the aorta and MRA exposed to Cd, RQH protected against heightened vascular reactivity while preserving NO bioavailability for vascular function. Moreover, RQH effectively mitigated oxidative stress and inhibited the inflammatory cascade (COX-2 and TNF-alpha) resulting from Cd exposure. RQH offering antihypertensive, antioxidant, and anti-inflammatory properties could be used as a potential therapeutic dietary alternative for Cd-induced vascular damage.Although RQH demonstrated cardiovascular protection, it did not prevent the body weight loss induced by Cd, corroborating previous findings using this Cd model.9–11 Weight loss due to Cd exposure may be linked to oxidative stress and inflammation in the gastrointestinal tract, with additional contributions from kidney and liver damage.10,35 Despite not preventing weight loss, RQH treatment significantly improved water and food intake in animals exposed to Cd, an effect potentially related to its enhanced organoleptic properties, which had been previously observed in SHR.19 RQH, derived from quinoa, is rich in proteins, carbohydrates, fibers, and lipids, potentially providing additional nutritional support for physiological and metabolic functions.16,19,36 Furthermore, quinoa's regulatory effects on the gut microbiota, even in its hydrolyzed form, may restore intestinal balance by promoting pre- and probiotic populations and reducing pathogenic bacteria, which could contribute to mitigating Cd-induced intestinal dysbiosis induced by inflammatory insult.37,38
Different doses and duration of Cd exposure in humans and in animal models have been associated with cardiovascular damage and the development of arterial hypertension,5,8,9,39 as demonstrated in this study. We show that RQH cotreatment prevented hypertension induced by Cd and protected the vasculature from metal toxicity. This effect was previously observed in SHR rats, where administration of 1 g kg−1 day−1 in drinking water for eight weeks reduced a blood pressure of ∼9.8 ± 4.5 mmHg.19
Despite the recognized cardioprotective and antihypertensive properties of quinoa, the specific mechanisms responsible for the health benefits of its bioactive compounds are still not fully understood.40 Some studies have demonstrated in vitro that specific peptide sequences exhibit ACE inhibitory potential.41In vivo, quinoa protein from the Mengli-I variety (200 and 400 mg kg−1) significantly reduced SBP in SHR animals.42 ACE-inhibitory peptides identified in these studies (YRNAIMAPHYN, TRGDIIAIPPGAVH, FHPFPR, and NIFRPF) include di- and tripeptides that are efficiently absorbed in the intestine, enhancing their antihypertensive effects.41,42 The peptide sequences identified in the present RQH may be promising candidates for the antihypertensive effect observed in co-treated animals, due to the presence of histidine (H), leucine (L), valine (V), and proline (P). These amino acids, found in the sequences TETVRFF, VDHPVFL, VPIYEGY, YIRRGEAKL, and KLYGPE, may exert a vasodilatory effect and influence nitric oxide (NO) production,43 contributing to the observed vascular protection.
Co-treatment with RQH significantly reduced blood Cd concentrations by approximately 20%, highlighting the hydrolysate's potential chelating effect. Although this circulating concentration remains high compared to the safe levels (∼0.3 µg L−1 in blood),44 the observed reduction provided beneficial effects and additional protection, possibly by decreasing Cd binding to essential biomolecules. This chelating potential may be related to the presence of cysteine residues, quercetin, and hydroxyl (–OH) groups in its structure, which confer potential chelating ability, which can form soluble complexes and/or exert antioxidant and anti-inflammatory activities.45–47
The identified peptide sequences contain hydroxyl groups (–OH) from tyrosine (Y), threonine (T), aspartate (D), or glutamate (E), which could contribute to the hydrolysate's chelating potential. Notably, YIRRGEAKL also exhibits chelating properties due to histidine (H), which interacts with metal cations via its imidazole ring.32 Additionally, glutamate (E) residues in PIYEGY, TETVRFF, VPIYEGY, YIRRGEAKL, and YTEQPL may enhance chelation through its carboxyl group.34 To our knowledge, no previous studies have demonstrated a relationship between red quinoa derived peptides consumption and Cd chelation in the body. However, compounds such as cysteines, polyphenols, and flavonoids have been shown to reduce Cd deposition in organs.48–50
RQH demonstrated antioxidant protection against vascular dysfunction by reducing ROS levels in the studied vessels and maintaining NO bioavailability effects in vascular responses, which was impaired by Cd exposure. In addition, RQH prevented the activation of the NADPH oxidase complex by reducing the expression of its isoform NOX-1 and subunit p47-phox, both of which were upregulated after Cd exposure. Notably, we demonstrated for the first time the anti-inflammatory effects of RQH; the treatment lowered the expression and involvement of COX-2 and its derivatives in the studied vessels.
Consequently, RQH treatment completely prevented the increased contractile response to Phe and NE in the aorta and MRA, contributing to its antihypertensive effect. The increase in the vasoconstrictor response observed after exposure to Cd has already been attributed to the reduction in endothelial modulation.8–11 Moreover, considering that NO is also linked to inflammatory signaling pathways,51 our findings suggest that the maintenance of NO bioavailability by RQH may expand its protective effects beyond vascular modulation, potentially interfering with key inflammatory cascades triggered by Cd exposure.
We show that co-treatment with RQH prevented Cd-induced NO reduction in the contractile response to Phe/NE, maintaining NO bioavailability and increasing eNOS expression in the aorta. This effect is likely due to RQH's high antioxidant activity, reducing ROS expression in the MRA and aorta.19 Following RQH's improvement of NO-mediated vascular responses in Cd-exposed arteries, we investigated the underlying pathways based on previous evidence that Cd promotes vascular dysfunction through the generation of the superoxide anion (O2˙−).9 As expected, Cd induced an increase in O2˙−, contributing to the enhanced contractile response in both vascular beds, demonstrated by the effects of SOD and Tiron incubation. In addition, elevated expression of NOX-1 and p47-phox, and increased oxidative stress parameters (ROS, TBARS, and FRAP) were also observed. However, co-treatment with RQH attenuated the oxidative stress induced by Cd, preventing not only the establishment of vascular damage, but also the upregulation of proteins that generate vascular oxidative stress.
Therefore, increased oxidative stress and exacerbation of ROS expression by the overactivation of NADPH oxidase, particularly the NOX-1 and NOX-2 isoforms, appear to be a central mechanism promoting Cd-induced endothelial dysfunction by reducing NO bioavailability.19,39,52 Notably, in this toxicological context, NADPH oxidase activation induced by Cd appears to occur, at least in part, through direct redox and inflammatory disturbances rather than exclusively through classical neurohumoral pathways typically described in essential hypertension models.39 The antioxidant effects of RQH, by preserving NO bioavailability and reducing ROS expression in both vascular beds, seem to be mediated by the inhibition of NADPH oxidase (NOX-1) activation in Cd-exposed animals.
Consistent with findings from other plant-derived compounds such as curcumin and soy casein, which showed antioxidant and cardioprotective effects in Cd-exposed models,53,54 the antioxidant effect of RQH was also confirmed in SHR rats, reducing lipid peroxidation in the kidneys, heart, and plasma, while increasing total antioxidant capacity in the kidneys and glutathione levels in the liver.19 Consistently, other studies have demonstrated quinoa's antioxidant properties using a low molecular weight polysaccharide, hydrolyzed quinoa, and quinoa extract in prostate tumor cells, human macrophages, and zebrafish larvae, respectively.55–57 Peptide sequences (ASPKPSSA and QFLLAGR) derived from hydrolyzed quinoa bran albumin using alcalase and trypsin were identified in the literature, demonstrating their ability to act as “scavengers” of the toxic reactive species.58 In the present study, nine peptide sequences identified also exhibited antioxidant properties, largely attributed to histidine (H), tyrosine (Y), leucine (L), and proline (P),59,60 which have been previously described in the literature.
In addition to its high oxidative effects on the cardiovascular system, Cd also exacerbates inflammatory cascades by increasing COX-2 and proinflammatory cytokines like TNF-α, as observed in this study and previously reported.8,9,11,61 In this context, our findings provide the first evidence of the anti-inflammatory effects of RQH, which until now had only been associated with antihypertensive and antioxidant properties. Specifically, RQH reduced COX-2 involvement in aortic and MRA vasoconstriction and also decreased the expression of COX-2 and TNF-α in both vascular beds, which were elevated by Cd exposure.
Among the peptide sequences identified in RQH, some exhibit anti-inflammatory potential, either independently or in synergy with their antioxidant properties. These include sequences containing histidine (H), proline (P), tyrosine (Y), aspartate (D), and glutamate (E), such as PIYEGY, YPIIADPK, YIRRGEAKL, and TETVRFF.33,62 It is important to note that the effects of this hydrolysate are possibly synergistic, and its therapeutic impact cannot be attributed to a single component alone. In fact, quinoa's anti-inflammatory properties may also be attributed to its content of polyphenols and flavonoids like quercetin, saponins, phenolic acids, and amino acids.63 Supporting this, in vitro and in vivo studies with quinoa-derived saponins and the peptide lunasin or quinoa itself showed reduced expression of inflammatory cytokines, such as TNF-α, IL-1β, and IL-6, suggesting an anti-inflammatory effect.64–68
Cd exposure induces interconnected cellular damage, including oxidative stress, inflammation, and apoptosis. Previous studies have linked increased TNF-α expression with apoptotic marker activation.11,69 In the present study, RQH demonstrated antioxidant properties and reduced TNF-α expression, potentially preventing the activation of the intrinsic apoptotic pathway and protecting the vasculature.
In conclusion, this study presents a novel therapeutic approach for Cd-induced cardiovascular damage. RQH proved to be a natural alternative to mitigate Cd cardiovascular damage, demonstrating antihypertensive, antioxidant, and newly identified anti-inflammatory properties. Given its plant-based origin and bioactive peptide composition, RQH represents a sustainable and potentially translatable intervention, offering a natural approach to prevent or reduce cardiovascular risks associated with metal exposure in humans.
Author contributions
Samia H. H. Kanaan: investigation, conceptualization, methodology, formal analysis, visualization, writing – original draft, review & editing. Paola Z. Moraes: investigation, methodology, formal analysis, writing – original draft, review & editing. Katye Y. de Souza de Oliveira: investigation, formal analysis, writing – review & editing. Fernando Barbora Jr.: investigation, writing – review & editing. José Eudes G. Pinheiro Jr.: conceptualization, formal analysis, writing – review & editing. Franck M. Peçanha: conceptualization, methodology, validation, writing – original draft, review & editing. Dalton V. Vassallo: resources, funding acquisition, writing – original draft, review & editing. Marta Miguel-Castro: conceptualization, methodology, validation, resources, project administration, funding acquisition, writing – original draft, review & editing. Giulia Alessandra Wiggers: conceptualization, methodology, formal analysis, validation, visualization, data curation, supervision, project administration, resources, funding acquisition, writing – original draft, review & editing. All authors have read and approved the final manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data for this article are available at Digital CSIC at https://digital.csic.es/.Supplementary information (SI) is available. Supplementary Table: Effect of RQH on sensitivity (pD2) and maximal effect (Emax) to Phe or NE in Aorta or MRA, respectively, of rats of all experimental groups and experimental conditions. See DOI: https://doi.org/10.1039/d5fo04828a.
Acknowledgements
This work was supported by the National Council for Scientific and Technological Development [PQ/CNPq 305360/2023-0]; Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES); Fundação de Amparo à Pesquisa do Rio Grande do Sul – FAPERGS/Brazil [PQG: 24/2551-0001363-6]; FAPES/CNPq/PRONEX [No. 80598773/17] and Spanish Government through the Agencia Estatal de Investigación (AEI) and Fondo Europeo de Desarrollo Regional (FEDER) [PID2022-138610OB-I00]; I-COOP + 2024 (COOPA24047). S. H. H. K. was supported by CNPq; P. Z. M was supported by CAPES/Brazil; and K. Y. S. O. was supported by FAPERGS/Brazil.References
- M. Peana, A. Pelucelli, C. T. Chasapis, S. P. Perlepes, V. Bekiari, S. Medici and M. A. Zoroddu, Biological Effects of Human Exposure to Environmental Cadmium, Biomolecules, 2022, 13(1), 36, DOI:10.3390/biom13010036 , PMID: 36671421; PMCID: PMC9855641.
- S. Satarug, S. H. Garrett, M. A. Sens and D. A. Sens, Cadmium, environmental exposure, and health outcomes, Environ. Health Perspect., 2010, 118(2), 182–190, DOI:10.1289/ehp.0901234 , PMID: 20123617; PMCID: PMC2831915.
- Á. Mérida-Ortega, L. López-Carrillo, K. Rangel-Moreno, N. Ramirez and S. J. Rothenberg, Tobacco Smoke Exposure and Urinary Cadmium in Women from Northern Mexico, Int. J. Environ. Res. Public Health, 2021, 18(23), 12581, DOI:10.3390/ijerph182312581 , PMID: 34886306; PMCID: PMC8656637.
- C. D. S. Vergilio, D. Lacerda, B. C. V. Oliveira, E. Sartori, G. M. Campos, A. L. S. Pereira, D. B. Aguiar, T. D. S. Souza, M. G. Almeida, F. Thompson and C. E. Rezende, Metal concentrations and biological effects from one of the largest mining disasters in the world (Brumadinho, Minas Gerais, Brazil), Sci. Rep., 2020, 10(1), 5936, DOI:10.1038/s41598-020-62700-w , PMID: 32246081; PMCID: PMC7125165.
- M. Tellez-Plaza, E. Guallar, B. V. Howard, J. G. Umans, K. A. Francesconi, W. Goessler, E. K. Silbergeld, R. B. Devereux and A. Navas-Acien, Cadmium exposure and incident cardiovascular disease, Epidemiology, 2013, 24(3), 421–429, DOI:10.1097/EDE.0b013e31828b0631 , PMID: 23514838; PMCID: PMC4142588.
- P. Verzelloni, T. Urbano, L. A. Wise, M. Vinceti and T. Filippini, Cadmium exposure and cardiovascular disease risk: A systematic review and dose-response meta-analysis, Environ. Pollut., 2024, 345, 123462, DOI:10.1016/j.envpol.2024.123462 . Epub 2024 Jan 29. PMID: 38295933.
- C. R. Nwokocha, A. Baker, D. Douglas, G. McCalla, M. Nwokocha and P. D. Brown, Apocynin ameliorates cadmium-induced hypertension through elevation of endothelium nitric oxide synthase, Cardiovasc. Toxicol., 2013, 13(4), 357–363, DOI:10.1007/s12012-013-9216-0 , PMID: 23703608.
- J. K. Angeli, C. A. Cruz Pereira, T. de Oliveira Faria, I. Stefanon, A. S. Padilha and D. V. Vassallo, Cadmium exposure induces vascular injury due to endothelial oxidative stress: the role of local angiotensin II and COX-2, Free Radical Biol. Med., 2013, 65, 838–848, DOI:10.1016/j.freeradbiomed.2013.08.167 , Epub 2013 Aug 22. PMID: 23973752.
- J. E. G. Pinheiro Júnior, P. Z. Moraes, M. D. Rodriguez, M. R. Simões, F. Cibin, S. Pinton, F. Barbosa Junior, F. M. Peçanha, D. V. Vassallo, M. Miguel and G. A. Wiggers, Cadmium exposure activates NADPH oxidase, renin-angiotensin system and cyclooxygenase 2 pathways in arteries, inducing hypertension and vascular damage, Toxicol. Lett., 2020, 333, 80–89, DOI:10.1016/j.toxlet.2020.07.027 , Epub 2020 Jul 30. PMID: 32738273.
- P. Z. Moraes, J. E. G. Pinheiro Júnior, C. S. Martinez, C. R. Moro, G. C. da Silva, M. D. Rodriguez, M. R. Simões, F. Barbosa Junior, F. M. Peçanha, D. V. Vassallo, M. Miguel and G. A. Wiggers, Multi-functional egg white hydrolysate prevent hypertension and vascular dysfunction induced by cadmium in rats, J. Funct. Foods, 2022, 94, 105131, DOI:10.1016/j.jff.2022.105131 , Epub 2022 Apr 26. PMID: 35469109.
- J. T. Piagette, J. E. G. Pinheiro Júnior, S. H. H. Kanaan, C. T. Herrera, L. O. Bastilhos, F. M. Peçanha, D. V. Vassallo, M. Miguel-Castro and G. A. Wiggers, Pretreatment with egg white hydrolysate protects resistance arteries from damage induced after treatment with accidental cadmium exposure values, J. Funct. Foods, 2023, 104, 105529, DOI:10.1016/j.jff.2023.105529 , Epub 2023 Mar 21. PMID: 36976563.
- Y. Ozarde, D. Purandare, S. Deshmukh and R. Gadhave, Heavy metals and cardiovascular health: Uncovering links and health challenges, J. Trace Elem. Med. Biol., 2025, 89, 127648, DOI:10.1016/j.jtemb.2025.127648 , Epub 2025 Apr 9. PMID: 40228399.
- H. Lynch, C. Johnston and C. Wharton, Plant-Based Diets: Considerations for Environmental Impact, Protein Quality, and Exercise Performance, Nutrients, 2018, 10(12), 1841, DOI:10.3390/nu10121841 , PMID: 30513704; PMCID: PMC6316289.
- M. Balali-Mood, N. Eizadi-Mood, H. Hassanian-Moghaddam, L. Etemad, M. Moshiri, M. Vahabzadeh and M. Sadeghi, Recent advances in the clinical management of intoxication by five heavy metals: Mercury, lead, chromium, cadmium and arsenic, Heliyon, 2025, 11(4), e42696, DOI:10.1016/j.heliyon.2025.e42696.
- A. Nandan, P. Koirala, A. Dutt Tripathi, U. Vikranta, K. Shah, A. J. Gupta, A. Agarwal and N. Nirmal, Nutritional and functional perspectives of pseudocereals, Food Chem., 2024, 448, 139072, DOI:10.1016/j.foodchem.2024.139072 , Epub 2024 Mar 20. PMID: 38547702.
- V. Angeli, P. Miguel Silva, D. Crispim Massuela, M. W. Khan, A. Hamar, F. Khajehei, S. Graeff-Hönninger and C. Piatti, Quinoa (Chenopodium quinoa Willd.): An Overview of the Potentials of the “Golden Grain” and Socio-Economic and Environmental Aspects of Its Cultivation and Marketization, Foods, 2020, 9(2), 216, DOI:10.3390/foods9020216 , PMID: 32092899; PMCID: PMC7074363.
- Z. Shi, Y. Hao, C. Teng, Y. Yao and G. Ren, Functional properties and adipogenesis inhibitory activity of protein hydrolysates from quinoa (Chenopodium quinoa Willd), Food Sci. Nutr., 2019, 7(6), 2103–2112, DOI:10.1002/fsn3.1052 , PMID: 31289658; PMCID: PMC6593480.
- D. Morales, M. Miguel and M. Garcés-Rimón, Pseudocereals: a novel source of biologically active peptides, Crit. Rev. Food Sci. Nutr., 2021, 61(9), 1537–1544, DOI:10.1080/10408398.2020.1761774 , Epub 2020 May 14. PMID: 32406747.
- M. López-Moreno, E. Jiménez-Moreno, A. Márquez Gallego, G. Vera Pasamontes, J. A. Uranga Ocio, M. Garcés-Rimón and M. Miguel-Castro, Red Quinoa Hydrolysates with Antioxidant Properties Improve Cardiovascular Health in Spontaneously Hypertensive Rats, Antioxidants, 2023, 12(6), 1291, DOI:10.3390/antiox12061291 , PMID: 37372021; PMCID: PMC10295081.
- R. Balaraman, O. D. Gulati, J. D. Bhatt, S. P. Rathod and K. G. Hemavathi, Cadmium-induced hypertension in rats, Pharmacology., 1989, 38(4), 226–234, DOI:10.1159/000138541 , PMID: 2755962.
- R. D. Buñag, Validation in awake rats of a tail-cuff method for measuring systolic pressure, J. Appl. Physiol., 1973, 34(2), 279–282, DOI:10.1152/jappl.1973.34.2.279 , PMID: 4686367.
- K. C. Nielsen and C. Owman, Contractile response and amine receptor mechanisms in isolated middle cerebral artery of the cat, Brain Res., 1971, 27(1), 33–42, DOI:10.1016/0006-8993(71)90370-2 . PMID: 4396591.
- M. J. Mulvany and W. Halpern, Contractile properties of small arterial resistance vessels in spontaneously hypertensive and normotensive rats, Circ. Res., 1977, 41(1), 19–26, DOI:10.1161/01.res.41.1.19 , PMID: 862138.
- B. L. Batista, D. Grotto, J. L. Rodrigues, V. C. Souza and F. Barbosa Jr., Determination of trace elements in biological samples by inductively coupled plasma mass spectrometry with tetramethylammonium hydroxide solubilization at room temperature, Anal. Chim. Acta, 2009, 646(1–2), 23–29, DOI:10.1016/j.aca.2009.05.022 , Epub 2009 May 21. PMID: 19523552.
- F. Jiménez-Altayó, A. M. Briones, J. Giraldo, A. M. Planas, M. Salaices and E. Vila, Increased superoxide anion production by interleukin-1beta impairs nitric oxide-mediated relaxation in resistance arteries, J. Pharmacol. Exp. Ther., 2006, 316(1), 42–52, DOI:10.1124/jpet.105.088435 , Epub 2005 Sep 23. PMID: 16183707.
- C. Loetchutinat, S. Kothan, S. Dechsupa, J. Meesungnoen, J. P. Jay-Gerin and S. Mankhetkorn, Spectrofluorometric determination of intracellular levels of reactive oxygen species in drug-sensitive and drug-resistant cancer cells using the 2′,7′-dichlorofluorescein diacetate assay, Radiat. Phys. Chem., 2005, 72(2–3), 323–331, DOI:10.1016/j.radphyschem.2004.06.011.
- H. Ohkawa, N. Ohishi and K. Yagi, Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction, Anal. Biochem., 1979, 95(2), 351–358, DOI:10.1016/0003-2697(79)90738-3 , PMID: 36810.
- I. F. Benzie and J. J. Strain, The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay, Anal. Biochem., 1996, 239(1), 70–76, DOI:10.1006/abio.1996.0292 , PMID: 8660627.
- R. Alonso, D. Pisa, A. I. Marina, E. Morato, A. Rábano, I. Rodal and L. Carrasco, Evidence for fungal infection in cerebrospinal fluid and brain tissue from patients with amyotrophic lateral sclerosis, Int. J. Biol. Sci., 2015, 11(5), 546–558, DOI:10.7150/ijbs.11084 , PMID: 25892962; PMCID: PMC4400386.
- Á. Sanchiz, E. Morato, A. Rastrojo, E. Camacho, S. G. González-de la Fuente, A. Marina, B. Aguado and J. M. Requena, The Experimental Proteome of Leishmania infantum Promastigote and Its Usefulness for Improving Gene Annotations, Genes, 2020, 11(9), 1036, DOI:10.3390/genes11091036 . PMID: 32887454; PMCID: PMC7563732.
- A. Tsugita, K. Takamoto, M. Kamo and H. Iwadate, C-terminal sequencing of protein. A novel partial acid hydrolysis and analysis by mass spectrometry, Eur. J. Biochem., 1992, 206(3), 691–696, DOI:10.1111/j.1432-1033.1992.tb16975.x , PMID: 1606956.
- R. J. Elias, S. S. Kellerby and E. A. Decker, Antioxidant activity of proteins and peptides, Crit. Rev. Food Sci. Nutr., 2008, 48(5), 430–441, DOI:10.1080/10408390701425615 , PMID: 18464032.
- A. F. G. Cicero, F. Fogacci and A. Colletti, Potential role of bioactive peptides in prevention and treatment of chronic diseases: a narrative review, Br. J. Pharmacol., 2017, 174(11), 1378–1394, DOI:10.1111/bph.13608 , Epub 2016 Sep 29. PMID: 27572703; PMCID: PMC5429326.
- M. C. Udechukwu, S. A. Collins and C. C. Udenigwe, Prospects of enhancing dietary zinc bioavailability with food-derived zinc-chelating peptides, Food Funct., 2016, 7(10), 4137–4144, 10.1039/c6fo00706f , PMID: 27713952.
- J. Nguyen, A. Patel, A. Gensburg, R. Bokhari, P. Lamar and J. Edwards, Diabetogenic and Obesogenic Effects of Cadmium in Db/Db Mice and Rats at a Clinically Relevant Level of Exposure, Toxics, 2022, 10(3), 107, DOI:10.3390/toxics10030107 , PMID: 35324731; PMCID: PMC8949435.
- S. Dakhili, L. Abdolalizadeh, S. M. Hosseini, S. Shojaee-Aliabadi and L. Mirmoghtadaie, Quinoa protein: Composition, structure and functional properties, Food Chem., 2019, 299, 125161, DOI:10.1016/j.foodchem.2019.125161 , Epub 2019 Jul 9. Erratum in: Food Chem., 2020, 310, 125318. DOI: 10.1016/j.foodchem.2019.125318. PMID: 31323439.
- T. Y. Wang, S. Y. Tao, Y. X. Wu, T. An, B. H. Lv, J. X. Liu, Y. T. Liu and G. J. Jiang, Quinoa Reduces High-Fat Diet-Induced Obesity in Mice via Potential Microbiota-Gut-Brain-Liver Interaction Mechanisms, Microbiol. Spectrum, 2022, 10(3), e0032922, DOI:10.1128/spectrum.00329-22 , Epub 2022 May 18. PMID: 35583337; PMCID: PMC9241864.
- J. Kim, J. Kim, Y. Jung, G. Kim, S. Kim and H. Shin, Prebiotic potential of proso millet and quinoa: Effects on gut microbiota composition and functional metabolic pathways, J. Microbiol., 2025, 63(7), e2503002, DOI:10.71150/jm.2503002 , Epub 2025 Jul 31. PMID: 40741631.
- C. C. Almenara, G. B. Broseghini-Filho, M. V. Vescovi, J. K. Angeli, O. Faria Tde, I. Stefanon, D. V. Vassallo and A. S. Padilha, Chronic cadmium treatment promotes oxidative stress and endothelial damage in isolated rat aorta, PLoS One, 2013, 8(7), e68418, DOI:10.1371/journal.pone.0068418 , PMID: 23874620; PMCID: PMC3709967.
- A. Jangra, V. Kumar, S. Kumar, R. Mehra and A. Kumar, Unraveling the Role of Quinoa in Managing Metabolic Disorders: A Comprehensive Review, Curr. Nutr. Rep., 2025, 14(1), 4, DOI:10.1007/s13668-024-00600-5 , PMID: 39753698.
- H. Guo, Y. Hao, A. Richel, N. Everaert, Y. Chen, M. Liu, X. Yang and G. Ren, Antihypertensive effect of quinoa protein under simulated gastrointestinal digestion and peptide characterization, J. Sci. Food Agric., 2020, 100(15), 5569–5576, DOI:10.1002/jsfa.10609 , Epub 2020 Jul 29. PMID: 32608025.
- H. Guo, Y. Hao, X. Fan, A. Richel, N. Everaert, X. Yang and G. Ren, Administration with Quinoa Protein Reduces the Blood Pressure in Spontaneously Hypertensive Rats and Modifies the Fecal Microbiota, Nutrients, 2021, 13(7), 2446, DOI:10.3390/nu13072446 , PMID: 34371955; PMCID: PMC8308759.
- N. Shobako, Hypotensive peptides derived from plant proteins, Peptides, 2021, 142, 170573, DOI:10.1016/j.peptides.2021.170573 , Epub 2021 May 21. PMID: 34023396.
- Agency for Toxic Substances and Disease Registry, Toxicological profile for cadmium, U.S. Department of Health and Human Services, Public Health Service, Atlanta, GA, 2012. Available from: https://www.atsdr.cdc.gov/toxprofiles/tp5.pdf Search PubMed.
- M. J. Kozioł, Chemical composition and nutritional evaluation of quinoa (Chenopodium quinoa Willd.), J. Food Compos. Anal., 1992, 5(1), 35–68, DOI:10.1016/0889-1575(92)90006-6.
- M. Leopoldini, N. Russo, S. Chiodo and M. Toscano, Iron chelation by the powerful antioxidant flavonoid quercetin, J. Agric. Food Chem., 2006, 54(17), 6343–6351, DOI:10.1021/jf060986h , PMID: 16910729.
- H. P. Burrieza, A. J. Rizzo, E. Moura Vale, V. Silveira and S. Maldonado, Shotgun proteomic analysis of quinoa seeds reveals novel lysine-rich seed storage globulins, Food Chem., 2019, 293, 299–306, DOI:10.1016/j.foodchem.2019.04.098 , Epub 2019 Apr 27. PMID: 31151615.
- A. Winiarska-Mieczan, The potential protective effect of green, black, red and white tea infusions against adverse effect of cadmium and lead during chronic exposure - A rat model study, Regul. Toxicol. Pharmacol., 2015, 73(2), 521–529, DOI:10.1016/j.yrtph.2015.10.007 , Epub 2015 Oct 22. PMID: 26472100.
- A. Winiarska-Mieczan, Protective effect of tea against lead and cadmium-induced oxidative stress-a review, Biometals, 2018, 31(6), 909–926, DOI:10.1007/s10534-018-0153-z , Epub 2018 Oct 13. PMID: 30317404; PMCID: PMC6245044.
- A. Wilczyńska and N. Żak, Polyphenols as the Main Compounds Influencing the Antioxidant Effect of Honey-A Review, Int. J. Mol. Sci., 2024, 25(19), 10606, DOI:10.3390/ijms251910606 , PMID: 39408935; PMCID: PMC11477350.
- P. Theofilis, M. Sagris, E. Oikonomou, A. S. Antonopoulos, G. Siasos, C. Tsioufis and D. Tousoulis, Inflammatory Mechanisms Contributing to Endothelial Dysfunction, Biomedicines, 2021, 9(7), 781, DOI:10.3390/biomedicines9070781 , PMID: 34356845; PMCID: PMC8301477.
- B. Lassègue and K. K. Griendling, NADPH oxidases: functions and pathologies in the vasculature, Arterioscler., Thromb., Vasc. Biol., 2010, 30(4), 653–661, DOI:10.1161/ATVBAHA.108.181610 , Epub 2009 Nov 12. PMID: 19910640; PMCID: PMC2841695.
- M. F. Pérez Díaz, M. Acosta, F. H. Mohamed, M. L. Ferramola, L. B. Oliveros and M. S. Gimenez, Protective effect of soybeans as protein source in the diet against cadmium-aorta redox and morphological alteration, Toxicol. Appl. Pharmacol., 2013, 272(3), 806–815, DOI:10.1016/j.taap.2013.07.016 , Epub 2013 Aug 3. PMID: 23916567.
- A. Tubsakul, W. Sangartit, P. Pakdeechote, V. Kukongviriyapan, K. Apaijit and U. Kukongviriyapan, Curcumin Mitigates Hypertension, Endothelial Dysfunction and Oxidative Stress in Rats with Chronic Exposure to Lead and Cadmium, Tohoku J. Exp. Med., 2021, 253(1), 69–76, DOI:10.1620/tjem.253.69 , PMID: 33473064.
- U. Gawlik-Dziki, M. Świeca, M. Sułkowski, D. Dziki, B. Baraniak and J. Czyż, Antioxidant and anticancer activities of Chenopodium quinoa leaves extracts - in vitro study, Food Chem. Toxicol., 2013, 57, 154–160, DOI:10.1016/j.fct.2013.03.023 , Epub 2013 Mar 26. PMID: 23537598.
- Y. Hu, J. Zhang, L. Zou, C. Fu, P. Li and G. Zhao, Chemical characterization, antioxidant, immune-regulating and anticancer activities of a novel bioactive polysaccharide from Chenopodium quinoa seeds, Int. J. Biol. Macromol., 2017, 99, 622–629, DOI:10.1016/j.ijbiomac.2017.03.019 , Epub 2017 Mar 6. PMID: 28274868.
- R. Vilcacundo, D. Barrio, C. Carpio, A. García-Ruiz, J. Rúales, B. Hernández-Ledesma and W. Carrillo, Digestibility of Quinoa (Chenopodium quinoa Willd.) Protein Concentrate and Its Potential to Inhibit Lipid Peroxidation in the Zebrafish Larvae Model, Plant Foods Hum. Nutr., 2017, 72(3), 294–300, DOI:10.1007/s11130-017-0626-1 , PMID: 28831659.
- Y. Zheng, X. Wang, Y. Zhuang, Y. Li, H. Tian, P. Shi and G. Li, Isolation of Novel ACE-Inhibitory and Antioxidant Peptides from Quinoa Bran Albumin Assisted with an In Silico Approach: Characterization, In Vivo Antihypertension, and Molecular Docking, Molecules, 2019, 24(24), 4562, DOI:10.3390/molecules24244562 , PMID: 31842519; PMCID: PMC6943578.
- R. Matsui, R. Honda, M. Kanome, A. Hagiwara, Y. Matsuda, T. Togitani, N. Ikemoto and M. Terashima, Designing antioxidant peptides based on the antioxidant properties of the amino acid side-chains, Food Chem., 2018, 245, 750–755, DOI:10.1016/j.foodchem.2017.11.119 , Epub 2017 Dec 5. PMID: 29287436.
- M. Laguerre, C. Bayrasy, J. Lecomte, B. Chabi, E. A. Decker, C. Wrutniak-Cabello, G. Cabello and P. Villeneuve, How to boost antioxidants by lipophilization?, Biochimie, 2013, 95(1), 20–26, DOI:10.1016/j.biochi.2012.07.018 , Epub 2012 Aug 1. PMID: 22884864.
- S. Arbi, M. J. Bester, L. Pretorius and H. M. Oberholzer, Adverse cardiovascular effects of exposure to cadmium and mercury alone and in combination on the cardiac tissue and aorta of Sprague-Dawley rats, J. Environ. Sci. Health, Part A: Toxic/Hazard. Subst. Environ. Eng., 2021, 56(6), 609–624, DOI:10.1080/10934529.2021.1899534 , Epub 2021 Mar 15. PMID: 33720805.
- M. Dadar, Y. Shahali, S. Chakraborty, M. Prasad, F. Tahoori, R. Tiwari and K. Dhama, Antiinflammatory peptides: current knowledge and promising prospects, Inflammation Res., 2019, 68(2), 125–145, DOI:10.1007/s00011-018-1208-x , Epub 2018 Dec 17. PMID: 30560372.
- A. Malek Mahdavi, Z. Javadivala, Y. Khalili and M. Khalili, Effects of Quinoa (Chenopodium quinoa) on inflammatory mediators: a systematic review of preclinical studies, Inflammopharmacology, 2023, 31(1), 241–251, DOI:10.1007/s10787-022-01091-y , Epub 2022 Nov 4. PMID: 36331709.
- Y. Yao, X. Yang, Z. Shi and G. Ren, Anti-inflammatory activity of saponins from quinoa (Chenopodium quinoa Willd.) seeds in lipopolysaccharide-stimulated RAW 264.7 macrophages cells, J. Food Sci., 2014, 79(5), H1018–H1023, DOI:10.1111/1750-3841.12425 , Epub 2014 Apr 8. PMID: 24712559.
- G. Ren, Y. Zhu, Z. Shi and J. Li, Detection of lunasin in quinoa (Chenopodium quinoa Willd.) and the in vitro evaluation of its antioxidant and anti-inflammatory activities, J. Sci. Food Agric., 2017, 97(12), 4110–4116, DOI:10.1002/jsfa.8278 , Epub 2017 Apr 6. PMID: 28218804.
- S. Erfidan, S. Dede, A. Usta, V. Yüksek and S. Çetin, The effect of quinoa (Chenopodium quinoa) on apoptotic, autophagic, antioxidant and inflammation markers in glucocorticoid-induced insulin resistance in rats, Mol. Biol. Rep., 2022, 49(7), 6509–6516, DOI:10.1007/s11033-022-07479-x , Epub 2022 May 26. PMID: 35618936.
- J. Liu, Z. Wang, P. Mai, Y. Hao, Z. Wang and J. Wang, Quinoa bran soluble dietary fiber ameliorates dextran sodium sulfate induced ulcerative colitis in BALB/c mice by maintaining intestinal barrier function and modulating gut microbiota, Int. J. Biol. Macromol., 2022, 216, 75–85, DOI:10.1016/j.ijbiomac.2022.06.194 , Epub 2022 Jul 2. PMID: 35792311.
- J. U. Obaroakpo, W. Nan, L. Hao, L. Liu, S. Zhang, J. Lu, X. Pang and J. Lv, The hyperglycemic regulatory effect of sprouted quinoa yoghurt in high-fat-diet and streptozotocin-induced type 2 diabetic mice via glucose and lipid homeostasis, Food Funct., 2020, 11(9), 8354–8368, 10.1039/d0fo01575j.
- D. J. Van Antwerp, S. J. Martin, I. M. Verma and D. R. Green, Inhibition of TNF-induced apoptosis by NF-kappa B, Trends Cell Biol., 1998, 8(3), 107–111, DOI:10.1016/s0962-8924(97)01215-4 , PMID: 9695819.
| This journal is © The Royal Society of Chemistry 2026 |