 Open Access Article
Open Access ArticleEvaluation of the underutilized Malpighia glabra L. fruits as a future functional food: nutritional composition, phenolic profile, biological activities, and synergistic effects with pharmaceutical drugs
Varongsiri
Kemsawasd
 ,
Sirinapa
Thangsiri
,
Yuraporn
Sahasakul
,
Amornrat
Aursalung
,
Woorawee
Inthachat
,
Piya
Temviriyanukul
,
Sirinapa
Thangsiri
,
Yuraporn
Sahasakul
,
Amornrat
Aursalung
,
Woorawee
Inthachat
,
Piya
Temviriyanukul
 ,
Suwapat
Kittibunchakul
,
Suwapat
Kittibunchakul
 and
Uthaiwan
Suttisansanee
and
Uthaiwan
Suttisansanee
 *
*
Food and Nutrition Academic and Research Cluster, Institute of Nutrition, Mahidol University, Salaya, Phuttamonthon, Nakhon Pathom 73170, Thailand. E-mail: varongsiri.kem@mahidol.ac.th; sirinapa.tha@mahidol.ac.th; yuraporn.sah@mahidol.ac.th; amornrat.aur@mahidol.ac.th; woorawee.int@mahidol.ac.th; piya.tem@mahidol.ac.th; suwapat.kit@mahidol.ac.th; uthaiwan.sut@mahidol.ac.th; Tel: +66-(0)-2800-2380 (ext. 422)
First published on 7th April 2026
Abstract
Malpighia glabra L., commonly known as acerola or Barbados cherry, is a non-commercial tropical fruit species in Thailand that is primarily consumed fresh by local communities. Limited scientific data have restricted its potential for future food applications. In this study, we investigated the nutritional composition, phenolic profiles, and in vitro biological activity of M. glabra fruits. Results demonstrated that the ripened fruits (100 g fresh weight) provided low energy (36.9 kcal) with negligible fat content but exceptionally high vitamin C content (838 mg). The ethanolic extract exhibited strong antioxidant activities as examined by the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay (250.98 µmol Trolox equivalent (TE) per g dry weight (DW)), ferric ion reducing antioxidant power (FRAP) assay (642.35 µmol TE per g DW), and oxygen radical absorbance capacity (ORAC) assay (443.90 µmol TE per g DW). These activities were attributed to the phenolic composition, of which rutin was the predominant compound, as detected by liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS). The extract also demonstrated in vitro inhibition of α-glucosidase, with a half-maximal inhibitory concentration (IC50) of 4.81 mg mL−1, and β-secretase, with an IC50 value of 5.45 mg mL−1. Synergistic interactions were observed in vitro between the extract and acarbose (an anti-diabetic drug) and the extract and donepezil (an anti-Alzheimer's drug). These findings characterize the nutritional composition and in vitro bioactivities of ripened M. glabra fruits, supporting further investigation into their potential development as functional food ingredients.
1. Introduction
Malpighia glabra L. is a tropical shrub that belongs to the Malpighiaceae family that bears fruits commonly called acerola or Barbados cherry. This plant originates from Central America, Northern South America, and the Caribbean Islands and has been effectively cultivated in other tropical areas such as Thailand, where the local populations consume it mainly as a fresh fruit rather than as a commercial product. The fruits can be consumed at different maturity stages: green (immature), orange to orange-red (intermediate), and bright red (mature). The mature or ripened fruits are predominantly consumed due to their sweet-sour taste, while the fruits in other maturity stages have only a sour taste. Nutritional compositions and phytochemical profiles vary according to the fruit maturity stages. Among the Malpighia species, M. emarginata and M. punicifolia have received significant research attention due to their nutritional properties. The fully ripe fruits of Malpighia species contain high levels of vitamin C, with concentrations between 1347 and 1882 mg per 100 g fresh weight (FW).1–4M. emarginata fruits (100 g FW) also possess relatively low caloric values of 39 kcal, derived from macronutrient constituents, including 0.7 g of protein, 8.6 g of carbohydrates, and 0.3 g of fat,3 while only protein (1.1 g) and total sugar (5.5 g) are reported in M. punicifolia fruits (100 g FW).1,4 Although the levels of other vitamins are generally below the U.S. recommended daily allowance (USRDA), M. emarginata fruits (100 g FW) contain appreciable amounts of niacin (0.4 mg) and pantothenic acid (0.3 mg), along with moderate amounts of some essential minerals including potassium (163.6 mg), magnesium (20.2 mg), and calcium (13.4 mg).5 By contrast, limited nutritional information is available for ripened M. glabra fruits. Previous studies identified β-carotene, α-carotene, and β-cryptoxanthin as the three main carotenoids found at levels above 0.04 µg g−1, resulting in an overall carotenoid content of 30.5 µg g−1.6 Vitamin A content of 454 retinol equivalents (RE) per 100 g (ref. 6) and a vitamin C content of 72.1 mg per g dry weight7 were also detected. Only one report detailing the nutritional composition of M. glabra fruits was found in the FoodEXplorer Database (European Food Information Resource, EuroFIR), with Japan listed as the place of origin. However, this report did not specify fruit maturity (stating only ‘raw sour’), while information on the sugar content was missing and many vitamin levels remained undetected.2 Therefore, filling this knowledge gap will extend the current understanding of the nutritional composition of M. glabra fruits at a specific maturity stage (ripened) for further food applications and development.Similar to the nutritional compositions, only the phytochemical profile of M. emarginata has been widely investigated, with limited reports on M. glabra. Only three previous reports have documented phytochemical analyses of different fruit maturity stages of M. glabra fruits utilizing high-performance liquid chromatography with diode array detection (HPLC-DAD), revealing that this species possesses diverse bioactive phytochemicals, particularly phenolics.8–10 Based on methanolic extracts, ripened M. glabra fruits are rich in rutin,9 while another report suggests abundant quantities of epicatechin.10 Using similar techniques, rutin and quercetin were detected in M. glabra water extract.8 Aqueous ethanol has been previously reported as an optimal solvent for the extraction of phenolics from many fruits.11–13 Therefore, in this study, we investigated the phenolic profile using an 80% (v/v) aqueous ethanolic extraction of ripened M. glabra fruits. The selection of 80% (v/v) aqueous ethanol for phenolic extraction was supported by previous studies employing response surface methodology (RSM) to systematically optimize extraction conditions for various fruits, with results consistently identifying ethanol concentrations between 70% and 90% as optimal.11,14 The advantages of utilizing aqueous ethanolic extraction include its lower toxicity compared with methanol and higher extraction effectiveness than water alone.11 Additionally, while the identification of phenolics in M. glabra fruits has been limited to the HPLC-DAD technique, liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) with 26 phenolic standards (commonly found in most plants) was employed in this study. The advantages of LC-ESI-MS/MS over the HPLC-DAD technique include increased accuracy, sensitivity, and selectivity.15,16
The phytochemicals in Malpighia spp. fruits have been associated with various health-promoting properties, with previous studies suggesting their potential for preventing oxidative stress-related diseases, enhancing immune function, and providing anti-inflammatory benefits.17,18 Recent clinical investigations have reported their potential benefits for cardiovascular health and in enhancing metabolic syndrome parameters, along with their neuroprotective effects.19,20 While most biological properties have been extensively investigated in M. emarginata fruits, M. glabra remains underexplored in terms of its functional properties, despite its promising phytochemical profile. Regarding anti-diabetic and anti-hyperlipidemia properties, M. emarginata has been widely studied both in vivo and in vitro.21,22 Even though the methanolic extract of M. emarginata residual bagasse from pulp extraction was unable to inhibit the lipid-degrading enzyme lipase, it showed inhibitory activities against the carbohydrate-hydrolyzing enzymes α-amylase and α-glucosidase in vitro.22 However, no data are available for the insulin-maintaining enzyme dipeptidyl peptidase-IV (DPP-IV) for any Malpighia spp. Likewise, as medicinal targets for Alzheimer's disease treatment, three significant enzymes, including the neurotransmitter-degrading enzymes acetylcholinesterase (AChE) and butyrylcholinesterase (BChE), and β-amyloid-generating enzyme β-secretase (BACE-1), have been used as enzyme templates for structure-based drug design. Interestingly, norfriedelanes isolated from the branches and roots of M. emarginata showed inhibitory effects against AChE,23 and dark chocolate with added M. glabra fruit extract exhibited AChE inhibitory activity.24 However, no studies have been conducted on BChE and BACE-1 inhibition for any Malpighia spp.
Comprehensive information on the nutritional profile at a specific maturity stage, phenolic profiling using LC-ESI-MS/MS, phenolic extraction using aqueous ethanolic conditions, enzyme inhibitory activities, and synergistic interactions with synthetic drugs remains lacking for M. glabra ripened fruits. This knowledge gap hinders the recognition and utilization of these underutilized fruits as valuable sources of phytochemicals for health promotion and limits their potential valorization as functional food ingredients. To address this research lacuna, this study has analyzed the nutritional compositions of ripened M. glabra fruits, as well as the phenolic profile and in vitro bioactivity screening of M. glabra aqueous ethanolic extract. A particular emphasis was placed on the capacity of the fruits to inhibit target enzymes, including lipase, α-amylase, α-glucosidase, DPP-IV, AChE, BChE, and BACE-1. Furthermore, this study has explored the synergistic interactions between M. glabra extract and conventional synthetic medications. The research results characterize key phytochemical and bioactive properties that support the potential of M. glabra fruits as functional food ingredients and warrant further investigation of their health-promoting properties. These findings contribute to cultural resource management practices, promoting the sustainable preservation and potential utilization of M. glabra fruits for future food and nutraceutical applications.
2. Materials and methods
2.1 Sample collection, preparation, and extraction
Ripened fruits of M. glabra (3–4 weeks after flowering) were gathered from the Ban Phra Sub-district, Mueang District, Prachinburi Province, Thailand (14°07′6.53″ N, 101°24′28.08″ E), in October 2023 (Fig. 1). The color of the fresh samples was measured using a ColorFlex EZ spectrophotometer (Hunter Associates Laboratory, VA, USA), and the results were expressed in CIELAB units as lightness (L*) of 26.70 ± 0.26 (indicating a darker shade), a red-green axis (a*) of 34.23 ± 0.36 (showing a reddish tendency), and a blue-yellow axis (b*) of 19.48 ± 0.48 (indicating a yellowish tendency). Using a digital vernier caliper (Protronics Co., Ltd, Pathum Thani, Thailand) with a 0.01 mm/0.0005″ measuring range, ten fruit samples were measured, revealing an average diameter of 24.34 ± 1.04 mm and an average height of 18.69 ± 0.88 mm. The plant sample was preserved at the Sireeruckhachati Nature Learning Park of Mahidol University (Nakhon Pathom Province, Thailand) with the assigned voucher number PBM-006356. The plant specimen was verified and authenticated by Dr Sunisa Sangvirotjanapat from Mahidol University, Thailand. | ||
| Fig. 1 (A) Fresh fruits of Malpighia glabra L. and (B and C) sectioned fruit sliced lengthwise and crosswise, respectively. | ||
Fresh M. glabra fruits were first washed with deionized water and then freeze-dried using a PL9000 freeze dryer (Heto Lab Equipment, Allerod, Denmark). Following drying, the fruits were processed into a fine powder using a Philips 600 W grinder (Philips Electronics Co., Ltd, Jakarta, Indonesia). The powder characteristics were as follows: color analysis expressed in CIELAB units as L* of 64.81 ± 0.24, a* of 22.31 ± 0.09, and b* of 16.99 ± 0.02; a moisture content of 4.70% ± 0.34% was measured using a Halogen HE53 moisture analyzer (Mettler-Toledo AG, Greifensee, Switzerland); a particle size of 60 mesh, as analyzed by a Retsch AS200 sieving machine (Retsch GmbH, Haan, Germany). For preservation, the powdery sample was sealed in aluminum foil bags and stored at −20 °C until needed for further analysis.
Following a previously established method,25 the extraction process began by mixing 3 g of the powdery sample with 15 mL of 80% (v/v) aqueous ethanol. The mixture was shaken for 4.5 h in a WNE45 water bath shaker (Memmert GmBh, Eagle, WI, USA) maintained at 30 °C. The mixture was then subjected to centrifugation at 3800g for 10 min using a Hettich® ROTINA 38R centrifuge (Andreas Hettich GmbH, Tuttlingen, Germany). The supernatant was then filtered through a 0.22 µm polyethersulfone (PES) syringe filter and stored at −20 °C for subsequent analysis. The extract exhibited a pH of 5.5 measured using a pH meter (a-AB33 pH model, Ohaus Corporation, Parsippany, NJ, USA).
2.2 Determination of nutritive values
Standard AOAC (Association of Official Analytical Chemists) protocols were employed to determine the nutritional content of M. glabra fruits, using methods previously reported in the literature.25 The comprehensive analysis included measurements of energy, moisture, protein, carbohydrates, fats, and ash. Additionally, specific sugar components (fructose, glucose, sucrose, and maltose), dietary fiber fractions (soluble, insoluble, and total dietary fiber), B-complex vitamins (B1, B2, B3, B5, B6, B7, B9, and B12), vitamin C, macrominerals (calcium, sodium, potassium, and magnesium), and microminerals (iron and zinc) were evaluated. All analyses were performed at the Institute of Nutrition, Mahidol University's Accredited Laboratory, which operates under ISO/IEC 17025: 2017 standards for laboratory quality systems.The moisture content was determined using a Memmert UNE 500 hot air oven (Memmert GmBh, Eagle, WI, USA) following AOAC method 925.45 (2019). The sample was dried at 95 °C–105 °C with heat distribution until it reached a constant weight. The moisture content was then calculated as the difference between the initial and final weights using eqn (1) as follows:
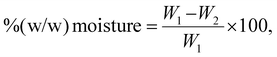 | (1) |
Using the Kjeldahl method as described in AOAC method 991.20 (2019), the protein content was analyzed through a multi-step process. First, the sample protein was hydrolyzed with concentrated sulfuric acid (H2SO4) to produce ammonium sulfate ((NH4)2SO4). A strong alkaline solution was then applied to convert ammonium sulfate to ammonia (NH3), which was subsequently distilled and titrated with standard acid to determine the total nitrogen content. The nitrogen percentage was multiplied by the standard factor of 6.25 to obtain the final protein content, using eqn (2) and (3) as follows:
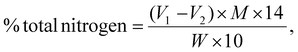 | (2) |
| % Protein = % total nitrogen × 6.25, | (3) |
The fat content was calculated according to AOAC method 922.06 (2019), which involved acid hydrolysis followed by a 6 h extraction with petroleum ether using a HT1043 Soxhlet system (Foss Tecator, Hoganas, Sweden). The total fat content was then calculated using eqn (4) as follows:
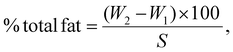 | (4) |
The ash content was measured following AOAC method 930.30 (2019) by incinerating the sample at 550 °C in a CWF 1100 muffle furnace (Carbolite Gero Ltd, Sheffield, UK), then weighing the remaining residue.
The total carbohydrate content was calculated using eqn (5), which considers the amounts of fat, protein, moisture, and ash present. The energy content was then determined using eqn (6) as follows:
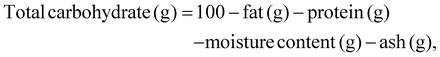 | (5) |
| Energy (kcal) = (total carbohydrate × 4) + (protein × 4) + (fat × 9). | (6) |
Fructose, glucose, sucrose, and maltose were analyzed using AOAC method 980.13 (2019). The analysis employed an ultra-fast liquid chromatography (UFLC) system (Shimadzu Corporation, Kyoto, Japan) coupled with an Alltech® model 800 evaporative light scattering detector (ELSD) (BUCHI Corporation, New Castle, DE, USA). The separation was performed on a Shodex Asahipak NH2P-50 4E column (250 × 4.6 mm, 5 µm particle size; Shodex Group, Kanagawa, Japan) using an isocratic mobile phase of 76% (v/v) aqueous acetonitrile at a constant flow rate of 1.0 mL min−1.
The total dietary fiber (TDF) was calculated as the sum of insoluble dietary fiber (IDF) and soluble dietary fiber (SDF), following enzymatic gravimetric methods from AOAC 993.19 and 991.42 (2019), respectively. The analysis began with sample digestion using three enzymes: α-amylase, amyloglucosidase, and protease. The IDF was then collected by filtration, and the residue was washed with warm deionized water. To determine the SDF content, the filtrate was combined with the washing water, and the soluble fiber was precipitated using ethanol.
The analysis of vitamin B1 (thiamin) and B2 (riboflavin) was conducted according to the in-house method INMU-TM-FCH-08 based on AOAC methods 942.23 (2019) and 970.65 (2019), respectively.26 The HPLC system used for analysis included a Luna® C18(2) 100 Å column (250 × 4.6 mm, 5 µm particle size; Phenomenex, Torrance, CA, USA), a FP-920 fluorescence detector (JASCO International Co., Ltd, Tokyo, Japan), and a LC-20AT pump (Shimadzu Scientific Instrument, Columbia, MD, USA). The vitamins were separated using an isocratic mobile phase of 50% (v/v) aqueous methanol at a constant flow rate of 1.0 mL min−1.
The analysis of vitamin B3 (niacin) was performed following AOAC method 961.14 (2019). The separation was conducted using an HPLC system equipped with a 1200 series G1310A isocratic pump (Agilent Technologies, Santa Clara, CA, USA), a Luna® C8(2) 100 Å column (250 × 4.6 mm, 5 µm particle size; Phenomenex, Torrance, CA, USA), and a VWD, 1100 series G1314B variable wavelength detector (Agilent Technologies, Santa Clara, CA, USA). The vitamin was eluted using an isocratic mobile phase of 15% (v/v) aqueous methanol at a constant flow rate of 1.0 mL min−1.
The analysis of vitamins B5 (pantothenic acid), B6 (pyridoxine), B7 (biotin), B9 (folic acid), and B12 (cobalamin) employed microbiological methods based on measuring the microbial growth in selective media lacking only the specific vitamin being tested. Each analysis followed specific protocols for sample preparation and extraction, including acid/alkaline hydrolysis at elevated temperatures. The procedure involved adding culture media to serial dilutions of vitamin standards or samples, followed by inoculation with specific microorganisms and incubation. Microbial growth was quantified utilizing a UV/visible single beam spectrophotometer (a Jenway™ model 7315 from Bibby Scientific Ltd, Staffordshire, UK) at 620 nm, except for vitamin B9, which was measured at 630 nm. Unless stated otherwise, in-house methods based on AOAC (2023) were employed. Vitamin B5 was analyzed using Lactobacillus plantarum ATCC 8014 (methods 960.46 and 945.74); B6 using Saccharomyces carlsbergensis (method 961.15); B7 using L. plantarum ATCC 8014 (AOAC 1980 Ch 43.150–43.158);27 B9 using L. casei ATCC 7469 (methods 960.46, 2004.05); and B12 using L. leichmannii ATCC 7830 (methods 960.46 and 952.20).
The vitamin C content was evaluated according to the previously published method.28 The analysis was performed using an HPLC system equipped with a Zorbax ODS column (250 × 4.6 mm, 5 µm particle size; Agilent Technologies, Santa Clara, CA, USA), a Waters 515 pump (Waters Corporation, Milford, MA, USA), and a UV/Vis detector (a UV-975 model from JASCO International Co., Ltd, Tokyo, Japan). The vitamin was separated using an isocratic mobile phase of 0.5% (w/v) KH2PO4 buffer (adjusted pH to 2.5 with H3PO4) at a constant flow rate of 0.8 mL min−1.
Minerals including potassium (K), sodium (Na), magnesium (Mg), calcium (Ca), zinc (Zn), and iron (Fe) were analyzed using an S series atomic absorption spectrometer (AAS) (Thermo Electron Corporation, UK) following AOAC method 985.35 (2019).
2.3 Determination of bioactive compounds
The types and quantities of phenolics present in M. glabra aqueous ethanolic extract were investigated using LC-ESI-MS/MS, following well-established methods and conditions from the previous literature.29 Briefly, the extract was subjected to a vacuum pump (a DTC-22 diaphragm from EYELA Co., Ltd, Shanghai, China) until dry. The dried extract was then reconstituted in 62.5% (v/v) aqueous ethanol to a concentration of 10 mg mL−1, and 10 µL of this solution was injected onto an Accucore RP-MS column (2.1 × 100 mm, 2.6 µm particle size; Thermo Fisher Scientific, Bremen, Germany) connected to an LC-ESI-MS/MS system, which consisted of a TSQ Quantis Triple Quadrupole mass spectrometer (MS), a Dionex ultra high-performance liquid chromatography (UHPLC) system, and a diode array detector (Thermo Fisher Scientific, Bremen, Germany). Analysis of the acquired data was performed using a Chromeleon 7 chromatography data system (Thermo Fisher Scientific, Bremen, Germany). Solvent A (acetonitrile) and solvent B (Milli-Q water with 18.2 MΩ cm resistivity at 25 °C containing 0.1% v/v formic acid) formed the gradient mobile phase system, delivered at a 0.5 mL min−1 flow rate as detailed below: 10% solvent A at time 0.0–0.1 min, 80% solvent A at time 0.1–8.0 min, 10% solvent A at time 8.0–8.1 min, and 10% solvent A at time 8.1–10.0 min.Twenty-six authentic standards of phenolics, including fisetin (≥90.0% HPLC), vanillic acid (≥97% HPLC), rosmarinic acid (≥98% HPLC), and mangiferin (≥98.0% TLC) were purchased from Sigma-Aldrich (St Louis, MO, USA), while galangin (≥98.0% HPLC), gallic acid (97.5%–102.5% T), and rutin (≥94% HPLC) were purchased from Wuhan ChemFaces Biochemical Co., Ltd (Hubei, China). Besides, isorhamnetin (≥99.0% HPLC) was purchased from Extrasynthese (Genay, France), while chlorogenic acid (>98.0% HPLC, T), (−)-epigallocatechin gallate (>98.0% HPLC), p-coumaric acid (>98.0% GC, T), caffeic acid (>98.0% HPLC, T), luteolin (>98.0% HPLC), quercetin (>98.0% HPLC, E), apigenin (>98.0% HPLC), genistein (>98.0% HPLC), cinnamic acid (>98.0% HPLC), 3,4-dihydroxybenzoic acid (≥97% T), syringic acid (>97.0% T), 4-hydroxybenzoic acid (>99.0% GC, T), sinapic acid (>99.0% GC, T), hesperidin (>90.0% HPLC, T), kaempferol (>97.0% HPLC), naringenin (>93.0% HPLC, T), ferulic acid (>98.0% GC, T), and myricetin (>97.0% HPLC) were purchased from Tokyo Chemical Industry (Tokyo, Japan). The LC-ESI-MS/MS parameters and validations of all phenolic standards are shown in SI Tables S1 and S2.
The total phenolic content (TPC) of M. glabra aqueous ethanolic extract was determined using a colorimetric assay, following a well-established, previously reported protocol30 without any modifications. Folin-Ciocalteu's phenol was used as a reagent, and a standard curve was generated using gallic acid at concentrations ranging from 0 to 200 µg mL−1. Using eqn (7) with a coefficient of determination (R2) of 0.9981, the TPC was calculated and reported as milligrams of gallic acid equivalent (GAE) per g dry weight (DW) as follows:
| y = 0.0054x + 0.0303, | (7) |
The total flavonoid content (TFC) of M. glabra aqueous ethanolic extract was also examined using a colorimetric assay, following an established method as previously reported31 without any modifications. Aluminum chloride was employed as a reagent, and a standard curve was generated using quercetin at concentrations ranging from 0 to 100 µg mL−1. The TFC was calculated using eqn (8) with R2 of 0.9920 and reported as milligrams of quercetin equivalent (QE) per g DW as follows:
| y = 0.0017x + 0.0119, | (8) |
Total anthocyanin content (TAC) was measured using a pH differential technique with sodium acetate buffers at pH 4.5 and 1.0, at absorbance values of 520 nm and 700 nm. This method followed the previously reported protocol25 with some modifications to the sample extraction as follows. The powdery sample (1 g) was mixed with 90% (v/v) aqueous ethanol (pH 1, 10 mL) and shaken for 3 h in the WNE45 water bath shaker (Memmert GmBh, Eagle, WI, USA) maintained at 80 °C. A calibration curve was created using cyanidin-3-O-glucoside standards ranging from 2 to 60 µg mL−1. The results were expressed as milligrams of cyanidin-3-O-glucoside equivalents (mg C3GE) per g DW.
The chemicals and reagents used for TPC, TFC, and TAC assays were purchased from Sigma-Aldrich (St Louis, MO, USA). Visualization and analysis of the assay reactions were performed using a SpectraMax Plus 384 microplate reader (Molecular Devices, LLC, Sunnyvale, CA, USA), with data processing conducted using the SoftMax Pro analysis software (version 6.5.1).
2.4 Determination of antioxidant potentials
The antioxidant potential of M. glabra aqueous ethanolic extract was evaluated using three different methods that cover two main antioxidant mechanisms: single-electron transfer (SET) and hydrogen-atom transfer (HAT). For SET mechanisms, two end-point spectroscopic tests were used as 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging and ferric ion reducing antioxidant power (FRAP) assays. The HAT mechanism was assessed using the kinetically measured oxygen radical absorbance capacity (ORAC) assay. All assays were performed using established methods as previously reported.32 Briefly, the DPPH radical-scavenging assay used the DPPH radical reagent with an endpoint measurement taken at 520 nm. The FRAP assay employed 2,4,6-tri(2-pyridyl)-S-triazine (TPTZ) reagent, ferric chloride solution, and acetate buffer. An endpoint measurement was performed at 600 nm. The ORAC assay consisted of sodium fluorescein and 2,2′-azobis(2-amidinopropane) dihydrochloride (AAPH) as the main reagents, while a kinetic measurement was taken at 485 nm (excitation) and 528 nm (emission). All chemicals and reagents for the assays were purchased from Sigma-Aldrich (St Louis, MO, USA). The measurements were conducted using a Synergy™ HT 96-well UV-visible microplate reader and the Gen 5 data analysis software (version 2.09, BioTek Instruments, Inc., Winooski, VT, USA). Results were standardized using Trolox for a calibration curve and expressed as µmol Trolox equivalent (TE) per g DW.2.5 Determination of enzyme inhibitory activities
The inhibitory activities of M. glabra aqueous ethanolic extract (pH 5.5) were evaluated against several key enzymes implicated in non-communicable diseases (NCDs), particularly diabetes (α-amylase, α-glucosidase, and DPP-IV), hyperlipidemia (lipase), and Alzheimer's disease (AChE, BChE, and BACE-1). The enzyme assays were conducted following previously reported protocols,25,31,33 as outlined in Table 1. All chemicals and reagents were purchased from Sigma-Aldrich (St Louis, MO, USA).| Assay | Assay components | ||||
|---|---|---|---|---|---|
| Enzyme | Substrate | Indicator | Extract | Detection wavelength | |
| a Candida rugosa lipase (type VII, ≥700 units per mg). b Electrophorus electricus AChE (1000 units per mg). c Equine serum BChE (≥10 units per mg). d Porcine pancreatic α-amylase (type VII, ≥10 units per mg). e Saccharomyces cerevisiae α-glucosidase (type I, ≥10 units per mg protein). f Recombinant human dipeptidyl peptidase-IV (≥10 units per mg). g DTNB was prepared as a 200 mM stock solution in absolute methanol and diluted to 16 mM with 50 mM KPB (pH 7.0) before use. ACh: acetylthiocholine; AChE: acetylcholinesterase; BACE-1: β-secretase; BCh: butyrylthiocholine; BChE: butyrylcholinesterase; BSA: bovine serum albumin; DMPTB: 2,3-dimercapto-1-propanol tributyrate; DPP-IV: dipeptidyl peptidase-IV; DTNB: 5,5′-dithiobis(2-nitrobenzoic acid); EDTA: ethylenediaminetetraacetic acid; FRET: fluorescence resonance energy transfer; Gly-Pro-pNA: Gly-Pro-p-nitroanilide hydrochloride; KPB: potassium phosphate buffer; pNPG: p-nitrophenyl-α-D-glucopyranoside; and pNPM: 2-chloro-4-nitrophenyl-α-D-maltotrioside. | |||||
| Lipase | 100 µL of 20 µg mL−1 lipase in 50 mM Tris (pH 8.0) containing 10 mM KCl, 1 mM EDTA, and 2 mg mL−1 BSAa | 50 µL of 0.2 mM DMPTB in 50 mM Tris (pH 7.2) containing 10 mM KCl, 1 mM EDTA, and 10% (v/v) Triton X-100 | 10 µL of 16 mM DTNB in 50 mM KPB (pH 7.0)g | 40 µL | 412 nm |
| AChE | 100 µL of 0.25 µg mL−1 AChE in 50 mM KPB (pH 7.0)b | 50 µL of 0.32 mM ACh in 50 mM KPB (pH 7.0) | |||
| BChE | 100 µL of 1.5 µg mL−1 BChE in 50 mM KPB (pH 7.0) containing 1 mM MgCl2c | 50 µL of 0.4 mM BCh in 50 mM KPB (pH 7.0) | |||
| α-Amylase | 100 µL of 0.06 mg mL−1 α-amylase in 50 mM KPB (pH 7.0) containing 200 mM KCld | 50 µL of 1 mM pNPM in 50 mM KPB (pH 7.0) containing 200 mM KCl | 50 µL | 405 nm | |
| α-Glucosidase | 100 µL of 0.1 U mL−1 α-glucosidase in 50 mM KPB (pH 7.0)e | 50 µL of 2 mM pNPG in 50 mM KPB (pH 7.0) | 50 µL | ||
| DPP-IV | 50 µL of 0.02 U mL−1 DPP-IV in 100 mM Tris-HCl (pH 8.0)f | 25 µL of 12 mM Gly-Pro-pNA in 100 mM Tris-HCl (pH 8.0) + 100 µL of 100 mM Tris-HCl (pH 8.0) | 25 µL | ||
| BACE-1 | BACE-1 FRET assay kit (Sigma-Aldrich, St Louis, MO, USA) following the manufacturer's recommendations | λ ex = 320 nm | |||
| λ em = 405 nm | |||||
The inhibitory assays for α-glucosidase, α-amylase, DPP-IV, lipase, AChE, and BChE were measured kinetically, while the BACE-1 inhibition assay used an end-point detection method. All assays were performed using the Synergy™ HT 96-well UV-visible microplate reader as previously mentioned. The results were expressed as a percentage of inhibition, calculated using eqn (9) as follows:
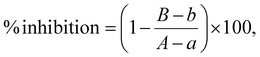 | (9) |
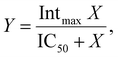 | (10) |
2.6 Preliminary synergistic screening
Potential synergistic interactions between commercial drugs and M. glabra aqueous ethanolic extract were examined through preliminary screening using well-established methods outlined in the previous literature.34 The synergistic evaluation of α-glucosidase inhibition was performed using M. glabra aqueous ethanolic extract, acarbose (anti-diabetic drug), and their combinations at different concentrations. Instead of acarbose, the synergistic evaluation of BACE-1 inhibition was performed using donepezil as an anti-Alzheimer's disease drug. The synergistic effects were analyzed by comparing experimental values (EV) against theoretical values (TV) as shown in eqn (11) as follows: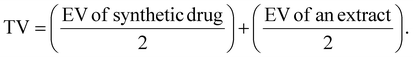 | (11) |
The synergistic effects were interpreted using the following criteria: a synergistic interaction was confirmed when the EV exceeded the TV by more than 5%, an antagonistic interaction was identified when the TV surpassed the EV by more than 5%, and an additive effect was determined when the difference between TV and EV was less than 5%.
2.7 Statistical analysis
The results are presented as averages with standard deviations (SD), based on three independent sets of samples, each performed in triplicate (n = 3). To determine statistical significance, the data underwent one-way analysis of variance (ANOVA) followed by Duncan's multiple comparison test, with differences considered significant at p < 0.05.3. Results
3.1 Nutritional compositions
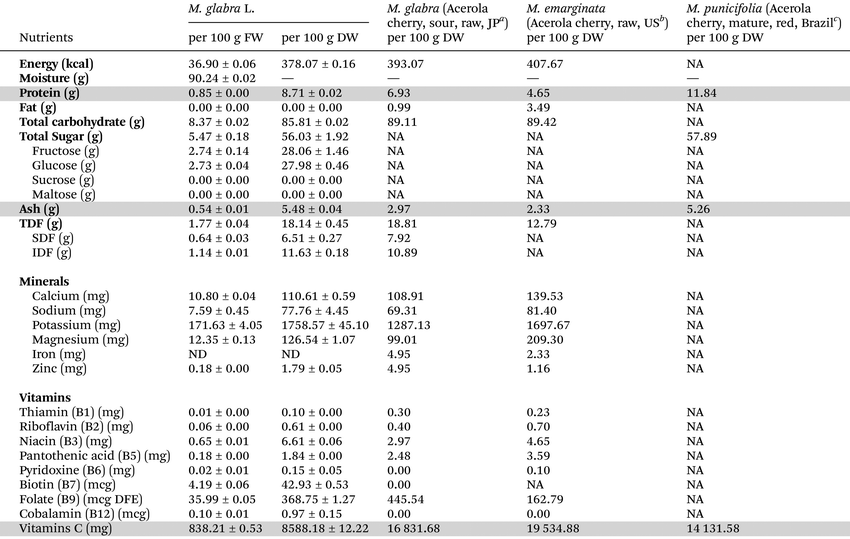
The results for the nutritional analysis of M. glabra fruits are shown as per 100 g FW and DW as indicated in Table 2. It was found that the fruits had a high moisture content (approximately 90%), while their energy mainly came from carbohydrates (approx. 86%). The fruits had a low protein content (approx. 9%) and no fat. Among the different types of carbohydrates, more than half (approx. 56%) were in the form of sugar (glucose and fructose contents in equivalent ratios), while sucrose and maltose were not detected. TDF content accounted for approx. 18%, and most dietary fiber was in the form of IDF, which was 1.8-fold higher than SDF. Among the investigated macrominerals, potassium was predominantly present (approx. 2%), followed by magnesium, calcium, and sodium, respectively (all were less than 0.2%). Microminerals, including iron and zinc, were also examined. While a trace amount of zinc was detected (less than 0.002%), no iron was detected (limit of detection (LOD) = 0.01 mg per 100 g). Interestingly, M. glabra fruits contained high vitamin C content (approx. 9%), while vitamin B3 was the highest among the B-complex vitamins (approx. 0.006%, with total B vitamins of approx. 0.01%).| All data are displayed as mean ± standard deviation (SD) of triplicated experiments from three independent sets of samples (n = 3). Grey shading indicates available data for comparing our results with all three previous reports. DW: dry weight; FW: fresh weight; TDF: total dietary fiber; SDF: soluble dietary fiber; IDF: insoluble dietary fiber; DFE: dietary folate equivalents; NA: not available; ND: not detected; and -: not applicable.a European food information resource (EuroFIR), FoodEXplorer database: acerola cherry fruit, sour type, raw (Food ID 0156123), country of origin: Japan, values converted to a dry-weight basis using a moisture content of 89.9%.b U.S. department of agriculture (USDA), food data central (FDC) database: acerola cherry fruit (West Indian Cherry), raw (FDC ID 171686), country of origin: USA, values converted to a dry weight basis using a moisture content of 91.4%.c Acerola cherry fruit, mature, red, country of origin: Brazil; values converted to a dry weight basis using a moisture content of 92.4%.4 |
|---|
|
3.2 Identification and quantitation of bioactive compounds
The targeted phenolic profile analyzed by LC-ESI-MS/MS only detected rutin (5.26 µg g−1 extract, Fig. 2 and Table 3) among 26 authentic phenolic standards (gallic acid, caffeic acid, sinapic acid, cinnamic acid, syringic acid, ferulic acid, rosmarinic acid, 3,4-dihydroxybenzoic acid, p-coumaric acid, 4-hydroxybenzoic acid, chlorogenic acid, vanillic acid, fisetin, rutin, genistein, apigenin, luteolin, hesperidin, (−)-epigallocatechin gallate, isorhamnetin, galangin, kaempferol, myricetin, naringenin, mangiferin, and quercetin). The LC-ESI-MS/MS chromatogram indicated that even though potential compounds eluted at the same time as the reference standards, the mass spectrometry fragmentation patterns, specifically the precursor and quantifier ions, showed significant differences (Fig. 2 and SI Tables S1–S3). Additionally, spectroscopic assays indicated the presence of TPC of 71.83 mg GAE per g DW, TFC of 6.80 mg QE per g DW, and TAC of 0.41 mg C3GE per g DW (Table 3). | ||
| Fig. 2 Liquid chromatography-electrospray ionization tandem mass spectrometry (LC-ESI-MS/MS) chromatogram of the aqueous ethanolic extract of Malpighia glabra L. | ||
| Phenolic content | Ion mass | Parent ions (m/z) | SRM transitions (m/z) and collision energy (V) | RF lens (V) | Amount |
|---|---|---|---|---|---|
| All data are displayed as mean ± standard deviation (SD) of triplicated experiments from three independent sets of samples (n = 3). DW: dry weight; GAE: gallic acid equivalent; QE: quercetin equivalent; and C3GE: cyanidin-3-O-glucoside equivalent. | |||||
| Targeted phenolics by LC-ESI-MS/MS | |||||
| Rutin (µg g−1 extract) | [M + H] | 611.2 | 303.13 (20.80 V) and 465.20 (12.71 V) | 198 | 5.26 ± 0.50 |
| TPC (mg GAE per g DW) | 71.83 ± 2.17 | ||||
| TFC (mg QE per g DW) | 6.80 ± 0.38 | ||||
| TAC (mg C3GE per g DW) | 0.41 ± 0.04 | ||||
3.3 In vitro biological activities
The antioxidant potential of M. glabra aqueous ethanolic extract was evaluated using DPPH radical scavenging, FRAP, and ORAC assays. The extract exhibited antioxidant activities of 250.98, 642.35, and 443.90 µmol TE per g DW, respectively (Table 4). Additionally, it exhibited DPPH radical scavenging activity with a half maximal effective concentration (EC50) of 154.73 µg mL−1.| Biological activities | Sample | Positive control |
|---|---|---|
| Malpighia glabra extract | Synthetic drug | |
| All data are displayed as mean ± standard deviation (SD) of triplicated experiments from three independent sets of samples (n = 3). AChE: acetylcholinesterase; BACE-1: β-secretase; BChE: butyrylcholinesterase; DPP-IV: dipeptidyl peptidase IV; DW: dry weight; IC50: half maximal inhibitory concentration; NA: not available; and ND: not detected.a The DPPH radical scavenging activity was also determined as a half maximal effective concentration (EC50) of 154.73 ± 16.13 µg mL−1.b Inhibitory activity was not detected up to the final extract concentration of 20 mg mL−1.c and the inhibitory activity was measured using the final extract concentration of 20 mg mL−1. | ||
| Antioxidant activities (µmol TE per g DW) | ||
| DPPH radical scavenging activitya | 250.98 ± 20.87 | NA |
| FRAP activity | 642.35 ± 20.93 | NA |
| ORAC activity | 443.90 ± 31.35 | NA |
| Enzyme inhibitions (IC 50 ) | ||
| Lipase | 10.94 ± 0.25 mg mL−1 | Orlistat (4.67 ± 0.38 µM) |
| α-Amylase | NDb | Acarbose (8.46 ± 0.45 µM) |
| α-Glucosidase | 4.81 ± 0.45 mg mL−1 | Acarbose (1.71 ± 0.11 mM) |
| DPP-IV | 23.73% ± 1.97% inhibitionc | Saxagliptin (0.27 ± 0.01 µM) |
| AChE | 15.90 ± 0.44 mg mL−1 | Donepezil (0.16 ± 0.01 µM) |
| BChE | 20.03 ± 0.40 mg mL−1 | Donepezil (35.56 ± 1.57 µM) |
| BACE-1 | 5.45 ± 0.20 mg mL−1 | Donepezil (0.14 ± 0.01 mM) |
In vitro assays were conducted to evaluate the inhibitory activity of the fruit extract against several enzymes, and the results are presented in Table 4. The fruit extract demonstrated a half-maximal inhibitory concentration (IC50) of 10.94 mg mL−1 against lipase. In comparison with orlistat, used as a positive control in the lipase inhibitory assay (IC50 = 4.67 µM or 2.32 µg mL−1), the fruit extract exhibited substantially lower inhibitory activity. Additionally, low inhibitory activities of the fruit extract were observed in α-amylase and DPP-IV inhibitory assays. The inhibitory ability against α-amylase was undetected up to the final extract concentration of 20 mg mL−1, while the fruit extract exhibited DPP-IV inhibitory activity of 23.73% at the extract concentration of 20 mg mL−1. Compared with saxagliptin (a positive control in DPP-IV inhibitory assay) with the IC50 of 0.27 µM or 0.09 µg mL−1, the DPP-IV inhibitory activity was considerably lower. However, the fruit extract effectively inhibited α-glucosidase with the IC50 of 4.81 mg mL−1 compared with acarbose, a commercially available antidiabetic agent, with the IC50 of 1.71 mM or 1.1 mg mL−1. According to these results, even though the fruit extract exhibited lower α-glucosidase inhibitory strength than acarbose (approximately 4.4-fold lower), the potential for synergistic interactions with the drug was further investigated. Interestingly, the fruit extract inhibited all key enzymes relevant to Alzheimer's disease in vitro. The IC50 values of 15.90 and 20.03 mg mL−1 against AChE and BChE, respectively, were detected in the fruit extract. Compared with donepezil, an anti-Alzheimer's disease therapeutic drug, with an IC50 of 0.16 µM (0.06 µg mL−1) against AChE and 35.56 µM (13.49 µg mL−1) against BChE, these inhibitory strengths of the fruit extract were considerably low. However, strong inhibitory strength of the fruit extract was observed in the BACE-1 inhibitory assay, in which the fruit extract exhibited the IC50 of 5.45 mg mL−1 (or approximately 100-fold lower IC50, indicating stronger inhibition) compared with donepezil with the IC50 of 0.14 mM (53.86 µg mL−1). Therefore, the in vitro synergistic effect between the fruit extract and donepezil against BACE-1 was also further investigated.
3.4 Synergistic effects between Malpighia glabra L. extract and synthetic drugs
To investigate potential synergistic interactions between M. glabra aqueous ethanolic extract and conventional pharmacological agents in vitro, enzyme inhibitory assays were conducted utilizing M. glabra aqueous ethanolic extract combined with either acarbose or donepezil, evaluated individually and in combination at inhibitory concentrations of 10%, 20%, and 30% (corresponding to IC10, IC20, and IC30, respectively). The combination of M. glabra aqueous ethanolic extract with acarbose resulted in dose-dependent inhibition, with synergistic effects demonstrated across all tested combinations in vitro (Table 5). The most efficacious combination, achieving approximately 50% inhibition of α-glucosidase, consisted of acarbose at IC30 combined with M. glabra extract at IC30, representing a 2.4-fold enhancement relative to the IC10 combination. Notably, this combination reduced the acarbose concentration required to achieve IC50 by 1.7-fold compared with acarbose alone. Similarly, synergistic interactions were observed across all tested combinations in vitro, with the most pronounced effect demonstrated when combining donepezil at IC20 with M. glabra extract at IC30 (Table 6). This combination achieved approximately 50% inhibition of BACE-1 and reduced the donepezil concentration required to achieve IC50 by approximately 2.5-fold compared with donepezil alone. However, no additional enhancement was observed at the IC30 combination, suggesting possible saturation of the enzyme active site.| Drug and extract | α-Glucosidase inhibition (%) | |||
|---|---|---|---|---|
| Acarbose | M. glabra extract | Experiment value (EV) | Theoretical value (TV) | Interpretation |
| The data on α-glucosidase inhibitory activities are demonstrated as mean ± standard deviation (SD) of three independent sets of samples analyzed in triplicate (n = 3). | ||||
| IC10 (0.31 mg mL−1) | 13.88 ± 1.09 | |||
| IC20 (0.75 mg mL−1) | 23.49 ± 1.10 | |||
| IC30 (1.25 mg mL−1) | 36.97 ± 1.35 | |||
| IC10 (0.38 mg mL−1) | 8.99 ± 0.05 | |||
| IC20 (0.63 mg mL−1) | 15.70 ± 1.12 | |||
| IC30 (1.25 mg mL−1) | 27.84 ± 0.96 | |||
| IC10 | IC10 | 20.83 ± 2.00 | 11.44 | Synergistic |
| IC10 | IC20 | 27.25 ± 2.62 | 14.79 | Synergistic |
| IC10 | IC30 | 34.94 ± 1.40 | 20.86 | Synergistic |
| IC20 | IC10 | 34.71 ± 2.00 | 16.24 | Synergistic |
| IC20 | IC20 | 34.95 ± 2.62 | 19.60 | Synergistic |
| IC20 | IC30 | 37.73 ± 1.40 | 25.67 | Synergistic |
| IC30 | IC10 | 35.05 ± 0.18 | 22.98 | Synergistic |
| IC30 | IC20 | 40.92 ± 1.20 | 26.34 | Synergistic |
| IC30 | IC30 | 49.28 ± 1.05 | 32.41 | Synergistic |
| Drug and extract | BACE-1 inhibition (%) | |||
|---|---|---|---|---|
| Donepezil | M. glabra extract | Experiment value (EV) | Theoretical value (TV) | Interpretation |
| The data on BACE-1 inhibitory activities are demonstrated as mean ± standard deviation (SD) of three independent sets of samples analyzed in triplicate (n = 3). | ||||
| IC10 (5.0 µg mL−1) | 9.32 ± 0.20 | |||
| IC20 (10.0 µg mL−1) | 19.96 ± 0.44 | |||
| IC30 (20.0 µg mL−1) | 27.06 ± 0.87 | |||
| IC10 (0.4 mg mL−1) | 14.20 ± 1.14 | |||
| IC20 (1.0 mg mL−1) | 25.12 ± 1.34 | |||
| IC30 (2.0 mg mL−1) | 35.04 ± 2.57 | |||
| IC10 | IC10 | 41.61 ± 0.01 | 11.76 | Synergistic |
| IC10 | IC20 | 44.21 ± 0.52 | 17.22 | Synergistic |
| IC10 | IC30 | 46.39 ± 2.07 | 22.18 | Synergistic |
| IC20 | IC10 | 33.82 ± 0.67 | 17.08 | Synergistic |
| IC20 | IC20 | 38.91 ± 4.23 | 22.54 | Synergistic |
| IC20 | IC30 | 48.88 ± 2.10 | 27.50 | Synergistic |
| IC30 | IC10 | 41.94 ± 3.49 | 20.63 | Synergistic |
| IC30 | IC20 | 38.44 ± 3.22 | 26.09 | Synergistic |
| IC30 | IC30 | 41.80 ± 2.85 | 31.05 | Synergistic |
4. Discussion
M. glabra is a tropical shrub, with the fruits consumed at various maturity stages from green (unripe) to bright red (ripe). While congeners M. emarginata and M. punicifolia are nutritionally well-documented,3,4M. glabra has received limited attention despite notable carotenoid and vitamin A contents.6 The nutritional composition of the fruit varies depending on the ripening stage,35 but only one maturity-unspecified report exists for M. glabra,2 representing a critical knowledge gap for food applications. The ripened M. glabra fruits showed comparable carbohydrate (85.81 g per 100 g DW) and protein (8.71 g per 100 g DW) levels to other Malpighia species (Table 2). However, the absence of detectable fat, versus 0.99–1.62 g per100 g DW in congeners, resulted in lower energy density (378.07 kcal versus typical Malpighia values), representing both an advantage for low-calorie diets and a limitation for energy-dense applications.2–4 Total sugars (56.03 g per 100 g DW) matched M. punicifolia (57.89 g per 100 g DW), though the fructose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glucose ratio (∼2
glucose ratio (∼2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) differed markedly from M. emarginata juice (166
1) differed markedly from M. emarginata juice (166![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 44
44![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 fructose
1 fructose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glucose
glucose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sucrose), suggesting distinct metabolic profiles.4,35 Vitamin C content (838.21 mg per 100 g FW) exceeded Thai RDIs by 838% (MOPH), establishing M. glabra as an exceptional source, but was 1.6–2.3-fold lower than previous Malpighia reports detailing M. glabra Japan (1700 mg per 100 g FW), M. emarginata U.S. (1680 mg per100 g FW), and M. punicifolia Brazil (1074 mg per 100 g FW).2–4 Key advantages include the unique presence of B7 (biotin) and B12 (cobalamin), plus elevated B3 (niacin) and B2 (riboflavin) compared with previous reports.2,3 Dietary fiber (18.14 g per 100 g DW) and minerals (calcium, sodium, potassium, and magnesium) aligned with typical Malpighia ranges.2,3 Therefore, ripened M. glabra fruits offer distinct nutritional advantages, including exceptional B-vitamin diversity, substantial vitamin C (despite lower levels than congeners), high fiber, and negligible fat, as suitable for nutrient-dense, low-fat dietary applications. These compositional differences reflect cultivar variation, environmental factors, and analytical methods, highlighting the unique nutritional niche of M. glabra within Malpighia.
sucrose), suggesting distinct metabolic profiles.4,35 Vitamin C content (838.21 mg per 100 g FW) exceeded Thai RDIs by 838% (MOPH), establishing M. glabra as an exceptional source, but was 1.6–2.3-fold lower than previous Malpighia reports detailing M. glabra Japan (1700 mg per 100 g FW), M. emarginata U.S. (1680 mg per100 g FW), and M. punicifolia Brazil (1074 mg per 100 g FW).2–4 Key advantages include the unique presence of B7 (biotin) and B12 (cobalamin), plus elevated B3 (niacin) and B2 (riboflavin) compared with previous reports.2,3 Dietary fiber (18.14 g per 100 g DW) and minerals (calcium, sodium, potassium, and magnesium) aligned with typical Malpighia ranges.2,3 Therefore, ripened M. glabra fruits offer distinct nutritional advantages, including exceptional B-vitamin diversity, substantial vitamin C (despite lower levels than congeners), high fiber, and negligible fat, as suitable for nutrient-dense, low-fat dietary applications. These compositional differences reflect cultivar variation, environmental factors, and analytical methods, highlighting the unique nutritional niche of M. glabra within Malpighia.
Our M. glabra aqueous ethanolic extract exhibited 7-fold higher TPC (71.83 mg GAE per g DW) than the methanolic extract of M. glabra10 and 5.5–6.9-fold higher than other Malpighia species.36,37 TFC was 2-fold higher than previously reported M. glabra extract.10 However, TAC (0.41 mg C3GE per g DW) was 2.5-fold lower than reported for ripened M. glabra fruits, consistent with observations that anthocyanin content increases while other phenolics decrease during ripening in Malpighia species.1,36,38 The LC-ESI-MS/MS analysis with 26 phenolic standards detected only rutin (5.26 µg g−1) in M. glabra aqueous ethanolic extract, consistent with rutin concentrations in M. emarginata (∼5 µg g−1).36 Previous HPLC-DAD studies of M. glabra methanolic extracts identified rutin alongside lower quantities of gallic acid, ellagic acid, epicatechin, caffeic acid, catechin, quercetin, kaempferol, and various phenolic acids.9,10 Although several of these compounds were included in our LC-ESI-MS/MS standards, they were not detected in our samples. These differences likely reflect variations in cultivars, maturity stages, collection locations, harvesting season, extraction protocols, and analytical methodologies.38,39
The discrepancy between the limited LC-ESI-MS/MS detection (only rutin) and the substantial TPC with relatively low TFC was attributed to several factors. Among the 26 phenolic standards (14 flavonoids and 12 phenolic acids) employed in the LC-ESI-MS/MS analysis, the targeted phenolic profile indicated only the presence of rutin. This suggests that the sample contained numerous phenolic compounds that were not included in the standard reference library. The LC-ESI-MS/MS method employed in this study utilized a targeted approach that detected only compounds with available authentic standards. Consequently, many phenolic compounds remained undetected despite their contribution to the overall phenolic content, as measured by the Folin–Ciocalteu method (TPC). Furthermore, the high TPC-to-TFC ratio observed in this study indicates that the predominant phenolic constituents were non-flavonoid phenolics. Given the classification of phenolics,40 the sample may be rich in phenolic acids, coumarins, or tannins, which contribute significantly to the total phenolic content but are not quantified by the flavonoid-specific TFC assay. Matrix effects and the complexity of the fruit extract influence compound ionization efficiency and detection sensitivity in LC-ESI-MS/MS analysis.41 The detection limits of this analytical method for certain phenolic compounds in a complex matrix proved insufficient for identification, particularly for compounds present at low concentrations or those experiencing ion suppression during electrospray ionization.
A strong correlation between TPC and antioxidant activity has been established in many previous studies.42–44 Our study demonstrated DPPH radical scavenging, FRAP, and ORAC activities of aqueous ethanolic extracts of M. glabra ripened fruits, demonstrating that antioxidative effects were exerted through both the SET and HAT mechanisms. Our DPPH radical scavenging activity (EC50 = 154.73 µg mL−1) was weaker than for previously reported methanolic extracts (EC50 = 8.69–46.20 µg mL−1)9,19 but stronger than ethanolic extracts (EC50 = 1.2 mg mL−1),10 indicating solvent-dependent extraction efficiency. Our extract exhibited 5.2-fold higher ORAC activity than M. emarginata juice,45 while FRAP activity substantially exceeded well-known antioxidant sources such as blueberry, blackcurrant, and cranberry (∼66–75 µmol TE per g DW).46 These results demonstrate considerable in vitro antioxidant capacity, supporting further investigation into potential applications as a natural antioxidant source.
In vitro enzyme inhibition assays were conducted to evaluate the M. glabra aqueous ethanolic extract against several enzymes, with activities compared with corresponding pharmaceutical agents. The extract inhibited lipase with an IC50 of 10.94 mg mL−1, approximately 2350-fold higher than orlistat (2.32 µg mL−1), precluding direct therapeutic application without substantial compound enrichment. This weak activity aligns with the predominance of rutin, which exhibits ∼4000-fold lower lipase inhibition than orlistat.47 No prior lipase inhibition data exist for M. glabra. Our screening results are consistent with findings from M. emarginata methanolic extract, which similarly showed no lipase inhibitory activity in vitro.22
M. glabra aqueous ethanolic extract exhibited selective α-glucosidase inhibition (IC50 = 4.81 mg mL−1) with no detectable α-amylase activity and weak DPP-IV inhibition (23.73% at 20 mg mL−1). These findings align with prior Malpighia research, in which M. emarginata methanolic extract inhibited both α-amylase and α-glucosidase in vitro.22 Although the extract required 4.4-fold higher mass concentration than acarbose (IC50 = 1.1 mg mL−1), all tested extract-acarbose combinations in the preliminary synergistic screening exhibited synergistic inhibition exceeding predicted additive effects. This selective inhibition and synergy likely reflected rutin bioactivity. Rutin demonstrates 1.4–1.6-fold lower IC50 values than acarbose (137.8–753.5 µM versus 217.6–1053.3 µM).48,49 Molecular docking revealed the lower binding energy of rutin (−10.5 kcal mol−1) compared with acarbose (−8.2 kcal mol−1; PDB ID: 3AXH), with 13 non-bonding interactions (9 hydrogen bonds, 2 hydrophobic, 2 pi-anion) versus only 7 hydrogen bonds for acarbose.50 Additional studies identified rutin interactions with Glu322, Lys324, and Asp325, residues proximal to the catalytic triad (Asp215, Glu277, and Asp352), indicating active site interference.49 Enzyme kinetics studies confirmed mixed-type inhibition by rutin, with stronger affinity for free enzyme than enzyme–substrate complexes, suggesting dual binding modes as (1) competitive-like inhibition near the active site and (2) the formation of ternary inhibitor-enzyme–substrate complexes.
This study presents the first report of AChE, BChE, and BACE-1 inhibition by any Malpighia species. M. glabra extract inhibited AChE, BChE, and BACE-1 in vitro, with the strongest potency against BACE-1 (IC50 = 5.45 mg mL−1), achieving activity comparable with donepezil (IC50 = 53.86 µg mL−1) on a mass basis, while cholinesterase inhibition was weaker (IC50 = 15.90 and 20.03 mg mL−1, respectively). All extract-donepezil combinations exhibited synergistic BACE-1 inhibition in the preliminary synergistic screening. This selective inhibition and synergy likely reflected the enzyme-specific potencies of rutin and complementary binding with donepezil. Rutin demonstrates substantially stronger BACE-1 inhibition (IC50 = 5.95 µM) versus cholinesterases (IC50 = 0.22–0.29 mM), representing ∼37–49-fold higher potency against BACE-1.51,52In silico studies revealed comparable BACE-1 binding energies for rutin (−7.04 kcal mol−1) and donepezil (−8.83 kcal mol−1).53 Mechanistically, donepezil primarily targets the catalytic dyad, while flavonoids like rutin typically inhibit BACE-1 through non-covalent interactions with catalytic pocket residues and the modulation of flap region dynamics.54 The BACE-1 flap transitions between open and closed conformations, altering active site geometry and accessibility.55 The hypothesis was posited that M. glabra extract may stabilize the closed-flap conformation, allosterically restricting substrate access and enhancing the binding occupancy of donepezil at the catalytic dyad, thereby producing synergistic inhibition through dual-site engagement. However, this mechanism requires structural validation through molecular dynamics simulations or X-ray crystallography.
While most bioactivities were explained in terms of the only detected phenolic compound, it must be acknowledged that the limited identification of specific phenolic compounds through LC-ESI-MS/MS analysis precludes definitive attribution of the observed bioactivities solely to phenolic composition. While the high TPC values suggest a significant presence of phenolic compounds, non-phenolic antioxidants, particularly vitamin C, which is abundant in many fruit extracts, may contribute substantially to the observed antioxidant and biological activities. The Folin–Ciocalteu assay, while widely used for phenolic quantification, is non-specific and can react with various reducing substances, including ascorbic acid and other non-phenolic compounds. Therefore, the bioactive effects reported in this study may result from a synergistic combination of both phenolic and non-phenolic antioxidants present in the extract, rather than exclusively from phenolic compounds. Future studies employing comprehensive metabolomic profiling and specific antioxidant quantification (e.g., HPLC analysis of vitamin C) would provide more definitive insights into the specific bioactive constituents responsible for the observed activities.
Our in vitro synergy assessment offers simplicity, transparency, and direct applicability as a preliminary screening tool. However, it has inherent limitations, including binary classification, evaluation at only three concentration levels, and the lack of comprehensive dose–response surface analysis. More sophisticated models, such as the Chou–Talalay combination index, Bliss independence, Loewe additivity, or ZIP methods, could provide more robust quantitative assessments with statistical confidence intervals and mechanistic insights.56,57 Importantly, the simplified empirical synergy models applied in vitro may not predict in vivo behavior, as these findings do not account for pharmacokinetic interactions, metabolism, or bioavailability differences that occur in complex biological systems. Therefore, animal studies and clinical trials are critical to confirm whether the observed synergistic interactions provide genuine therapeutic advantages in diabetes management. Future investigations should employ multiple synergy models simultaneously, utilize response surface methodology across broader concentration ranges, conduct mechanistic studies, and perform pharmacokinetic-pharmacodynamic modeling with in vivo validation to assess clinical translatability for type II diabetes and Alzheimer's disease management.
5. Conclusions
This study presents preliminary evidence of the nutritional richness and in vitro bioactivities of M. glabra fruits, particularly their high vitamin C and total phenolic contents. Although only rutin was identified among the targeted phenolic compounds, the aqueous ethanolic extract demonstrated strong antioxidant capacity and inhibitory effects against α-glucosidase and BACE-1, with apparent synergistic interactions when combined with standard drugs. These findings suggest that M. glabra warrants further investigation to determine whether these in vitro observations have any relevance to potential functional food applications. However, critical limitations constrain the interpretation of our findings. First, only rutin was identified among 26 phenolic standards, leaving substantial phytochemical composition uncharacterized. Untargeted metabolomics would provide more comprehensive profiling. Second, the molecular mechanisms underlying the apparent synergistic interactions remain speculative and require experimental validation. Third, safety profiles, optimal dosages, and long-term consumption effects remain unexplored. Future research should prioritize in vivo validation, bioavailability assessments, comprehensive phytochemical profiling, and mechanistic investigations to determine whether these in vitro observations have any biological relevance. Only after substantial preclinical evidence would clinical trials be warranted to evaluate potential health benefits and establish feasibility for functional food applications.Author contributions
Varongsiri Kemsawasd: writing – review and editing, writing – original draft, and conceptualization. Sirinapa Thangsiri: writing – original draft, visualization, validation, investigation, formal analysis, and data curation. Yuraporn Sahasakul: writing – original draft, visualization, validation, investigation, formal analysis, data curation, and conceptualization. Amornrat Aursalung: writing – original draft, investigation, and data curation. Woorawee Inthachat: writing – original draft, investigation, and data curation. Piya Temviriyanukul: writing – original draft and conceptualization. Suwapat Kittibunchakul: writing – review and editing, writing – original draft, formal analysis, visualization, data curation, and conceptualization. Uthaiwan Suttisansanee: writing – review and editing, writing – original draft, visualization, validation, supervision, resources, project administration, methodology, funding acquisition, formal analysis, and conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Tables S1 and S2, the parameters and validations of phenolic standards using liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) in the selective reaction monitoring (SRM) mode, Table S3, a liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) integration result of Malpighia glabra L. extract. See DOI: https://doi.org/10.1039/d5fo03782d.Acknowledgements
The article processing charge (APC) was paid by Mahidol University.References
- L. Delva and R. Goodrich-Schneider, Antioxidant activity and antimicrobial properties of phenolic extracts from acerola (Malpighia emarginata DC) fruit, Int. J. Food Sci. Technol., 2013, 48(5), 1048–1056 CrossRef CAS.
- EuroFIR, Foodexplorer, Version 2010–2025 [Available from: https://www.eurofir.org/foodexplorer/food_detail.php.
- USDA, Fooddata central: Foundation foods (version 12.0, october 2024) [Available from: https://fdc.nal.usda.gov.
- A. L. Vendramini and L. C. Trugo, Chemical composition of acerola fruit (Malpighia punicifolia L.) at three stages of maturity, Food Chem., 2000, 71(2), 195–198 CrossRef CAS.
- T. Mezadri, M. S. Fernández-Pachón, D. Villaño, M. C. García-Parrilla and A. M. Troncoso, The acerola fruit: Composition, productive characteristics and economic importance, Arch. Latinoam. Nutr., 2006, 56(2), 101–109 Search PubMed.
- M. L. Cavalcante and D. B. Rodriguez-Amaya, Carotenoid composition of the tropical fruits Eugenia uniflora and Malpighia glabra, in Developments in food science, ed. G. Charalambous, Elsevier, 1992, vol. 29, pp. 643–650 Search PubMed.
- L. G. Marques, M. C. Ferreira and J. T. Freire, Freeze-drying of acerola (Malpighia glabra L.), Chem. Eng. Process., 2007, 46(5), 451–457 Search PubMed.
- R. da Silva Nunes, V. F. S. Kahl, M. da Silva Sarmento, M. F. Richter, L. V. Costa-Lotufo, F. A. R. Rodrigues, J. A. Abin-Carriquiry, M. M. Martinez, S. Ferronatto, A. de Barros Falcão Ferraz and J. da Silva, Antigenotoxicity and antioxidant activity of acerola fruit (Malpighia glabra L.) at two stages of ripeness, Plant Foods Hum. Nutr., 2011, 66(2), 129–135 CrossRef CAS PubMed.
- E. M. M. Nascimento, F. F. G. Rodrigues, W. D. Costa, R. N. P. Teixeira, A. A. Boligon, E. O. Sousa, F. F. G. Rodrigues, H. D. M. Coutinho and J. G. M. da Costa, HPLC and in vitro evaluation of antioxidant properties of fruit from Malpighia glabra, (Malpighiaceae) at different stages of maturation, Food Chem. Toxicol., 2018, 119, 457–463 CrossRef CAS PubMed.
- S. V. Shreelakshmi, N. Chaitrashree, M. S. Nazareth, S. S. Kumar, N. P. Shetty and P. Giridhar, Bioactive compounds and antioxidant activity during ripening of malpighia glabra fruits, J. Food Sci. Technol., 2024, 61(4), 717–726 CrossRef CAS PubMed.
- M. Alothman, R. Bhat and A. A. Karim, Antioxidant capacity and phenolic content of selected tropical fruits from Malaysia, extracted with different solvents, Food Chem., 2009, 115(3), 785–788 CrossRef CAS.
- J. JakopiČ, R. VeberiČ and F. Štampar, Extraction of phenolic compounds from green walnut fruits in different solvents, Acta Agric. Slov., 2009, 93(1), 11–15 Search PubMed.
- I. El Mannoubi, Impact of different solvents on extraction yield, phenolic composition, in vitro antioxidant and antibacterial activities of deseeded Opuntia stricta fruit, J. Umm Al-Qura Univ. Appl. Sci., 2023, 9(2), 176–184 CrossRef.
- D. R. Pompeu, E. M. Silva and H. Rogez, Optimisation of the solvent extraction of phenolic antioxidants from fruits of Euterpe oleracea, using response surface methodology, Bioresour. Technol., 2009, 100(23), 6076–6082 CrossRef CAS PubMed.
- B. R. Kumar, Application of HPLC and ESI-MS techniques in the analysis of phenolic acids and flavonoids from green leafy vegetables (glvs), J. Pharm. Anal., 2017, 7(6), 349–364 CrossRef PubMed.
- L. Tribalat, O. Paisse, G. Dessalces and M. F. Grenier-Loustalot, Advantages of LC-MS-MS compared to LC-MS for the determination of nitrofuran residues in honey, Anal. Bioanal. Chem., 2006, 386(7–8), 2161–2168 CrossRef CAS PubMed.
- D. D. Leffa, G. T. Rezin, F. Daumann, L. M. Longaretti, A. L. F. Dajori, L. M. Gomes, M. C. Silva, E. L. Streck and V. M. de Andrade, Effects of acerola (Malpighia emarginata DC.) juice intake on brain energy metabolism of mice fed a cafeteria diet, Mol. Neurobiol., 2017, 54(2), 954–963 CrossRef CAS PubMed.
- A. Prakash and R. Baskaran, Acerola, an untapped functional superfruit: A review on latest frontiers, J. Food Sci. Technol., 2018, 55(9), 3373–3384 CrossRef CAS PubMed.
- B. M. M. de Oliveira, P. Z. Serpa, M. E. da Costa Zanatta, B. A. Aires, A. M. Steffler, L. B. Somensi, B. J. Cury, A. C. dos Santos, L. Venzon, T. Boeing, L. Mota da Silva and W. A. Roman Junior, Gastroprotective and gastric healing effects of the aqueous extract of casearia sylvestris in rodents: Ultrasound, histological and biochemical analyzes, J. Ethnopharmacol., 2022, 298, 115660 CrossRef CAS PubMed.
- V. Lima, E. Melo, L. Lima and P. Nascimento, Flavonoids in acerola (Malpighia sp L.) selections. 1- anthocyanins and flavonols content, Cienc. Rural, 2000, 30, 1063–1064 CrossRef.
- L. F. Laurindo, L. N. Takeda, N. Mendes Machado, A. M. M. B. Otoboni, R. Goulart, V. C. S. Catharin, L. R. Silva, S. M. Barbalho and R. Direito, Health benefits of acerola (Malpighia spp) and its by-products: A comprehensive review of nutrient-rich composition, pharmacological potential, and industrial applications, Food Biosci., 2024, 62, 105422 CrossRef.
- T. R. Marques, A. A. Caetano, A. A. Simão, F. C. d. O. Castro, V. de Oliveira Ramos and A. D. Corrêa, Metanolic extract of Malpighia emarginata bagasse: Phenolic compounds and inhibitory potential on digestive enzymes, Rev. Bras. Farmacogn., 2016, 26(2), 191–196 CrossRef CAS.
- J.-Q. Liu, X.-R. Peng, X.-Y. Li, T.-Z. Li, W.-M. Zhang, L. Shi, J. Han and M.-H. Qiu, Norfriedelins a–c with acetylcholinesterase inhibitory activity from acerola tree (Malpighia emarginata), Org. Lett., 2013, 15(7), 1580–1583 CrossRef CAS PubMed.
- D. Zarić, M. Rakin, M. Bulatović, T. Krunić, I. Lončarević, B. Pajin and Z. Blaževska, Influence of added extracts of herbs (Salvia lavandulifolia, Salvia officinalis) and fruits (Malpighia glabra) on rheological, textural, and functional (ACHE-inhibitory and antioxidant activity) characteristics of dark chocolate, J. Food Meas. Charact., 2024, 18(1), 772–782 CrossRef.
- L. K. Luu, S. Thangsiri, Y. Sahasakul, A. Aursalung, W. Inthachat, P. Temviriyanukul, N. On-Nom, C. Chupeerach and U. Suttisansanee, Nutrients, phytochemicals and in vitro disease prevention of Nephelium hypoleucum kurz fruit, Nutrients, 2023, 15(4), 950 CrossRef CAS PubMed.
- P. Wimalasiri and R. B. H. Wills, Simultaneous analysis of thiamin and riboflavin in foods by high-performance liquid chromatography, J. Chromatogr., A, 1985, 318, 412–416 CrossRef CAS PubMed.
- W. Horwitz, Official methods of analysis of aoac international, AOAC International, Washington, DC, USA, 2023 Search PubMed.
- I. Odriozola-Serrano, T. Hernández-Jover and O. Martín-Belloso, Comparative evaluation of uv-HPLC methods and reducing agents to determine vitamin C in fruits, Food Chem., 2007, 105(3), 1151–1158 CrossRef CAS.
- P. Sirichai, S. Kittibunchakul, S. Thangsiri, N. On-Nom, C. Chupeerach, P. Temviriyanukul, W. Inthachat, O. Nuchuchua, A. Aursalung, Y. Sahasakul, S. Charoenkiatkul and U. Suttisansanee, Impact of drying processes on phenolics and in vitro health-related activities of indigenous plants in Thailand, Plants, 2022, 11(3), 294 CrossRef CAS PubMed.
- C. Khemthong, S. Thangsiri, W. Chathiran, W. Inthachat, C. Chupeerach, P. Temviriyanukul, N. On-Nom, W. Srichamnong and U. Suttisansanee, The effect of pre- and post-harvesting techniques on phenolics, antioxidant activities and key enzyme inhibitions of commercially available ready-to-drink teas, J. Agric. Food Res., 2025, 21, 101853 CAS.
- P. Temviriyanukul, S. Kittibunchakul, P. Trisonthi, W. Inthachat, D. Siriwan and U. Suttisansanee, Analysis of phytonutrients, anti-mutagenic and chemopreventive effects of tropical fruit extracts, Foods, 2021, 10(11), 2600 CrossRef CAS PubMed.
- N. On-Nom, S. Thangsiri, W. Inthachat, P. Temviriyanukul, Y. Sahasakul, A. Aursalung, C. Chupeerach and U. Suttisansanee, Optimized conditions for the extraction of phenolic compounds from Aeginetia indica L. And its potential biological applications, Molecules, 2024, 29(5), 1050 CrossRef CAS PubMed.
- P. Natnicha, T. Piya and S. Uthaiwan, Investigation of anthocyanidins and anthocyanins for targeting α-glucosidase in diabetes mellitus, Prev. Nutr. Food Sci., 2020, 25(3), 263–271 CrossRef PubMed.
- W. Inthachat, N. Promyos, U. Suttisansanee, R. Chamchan, S. Thangsiri, B. Chantong, P. Pitchakarn and P. Temviriyanukul, Microwave-assisted optimization extraction for green chili paste and investigation of its alpha-glucosidase inhibitory activity with molecular docking and molecular dynamics simulation for anti-diabetic potential, J. Agric. Food Res., 2025, 22, 102130 CAS.
- A. M. Righetto, F. M. Netto and F. Carraro, Chemical composition and antioxidant activity of juices from mature and immature acerola (Malpighia emarginata DC), Food Sci. Technol. Int., 2005, 11(4), 315–321 CrossRef CAS.
- I. Carneiro Ferreira, V. Pereira da Silva, J. C. Vilvert, F. de França Souza, S. T. de Freitas and M. dos Santos Lima, Brazilian varieties of acerola (Malpighia emarginata DC.) produced under tropical semi-arid conditions: Bioactive phenolic compounds, sugars, organic acids, and antioxidant capacity, J. Food Biochem., 2021, 45(8), e13829 CrossRef CAS PubMed.
- A. Vasavilbazo-Saucedo, N. Almaraz-Abarca, H. González-Ocampo, J. Ávila-Reyes, L. Gonzalez, A. Luna, A. Delgado and R. Torres-Ricario, Phytochemical characterization and antioxidant properties of the wild edible acerola Malpighia umbellata rose, Cienc. Tecnol. Aliment., 2018, 16, 698–706 CAS.
- J. C. Vilvert, S. T. de Freitas, L. F. dos Santos, T. S. Ribeiro and C. M. Veloso, Phenolic compounds in acerola fruit and by-products: An overview on identification, quantification, influencing factors, and biological properties, J. Food Meas. Charact., 2024, 18(1), 216–239 CrossRef.
- T. Belwal, S. M. Ezzat, L. Rastrelli, I. D. Bhatt, M. Daglia, A. Baldi, H. P. Devkota, I. E. Orhan, J. K. Patra, G. Das, C. Anandharamakrishnan, L. Gomez-Gomez, S. F. Nabavi, S. M. Nabavi and A. G. Atanasov, A critical analysis of extraction techniques used for botanicals: Trends, priorities, industrial uses and optimization strategies, TrAC, Trends Anal. Chem., 2018, 100, 82–102 CrossRef CAS.
- R. J. Robbins and S. R. Bean, Development of a quantitative high-performance liquid chromatography–photodiode array detection measurement system for phenolic acids, J. Chromatogr., A, 2004, 1038(1), 97–105 CrossRef CAS PubMed.
- R. Dams, M. A. Huestis, W. E. Lambert and C. M. Murphy, Matrix effect in bio-analysis of illicit drugs with LC-MS/MS: Influence of ionization type, sample preparation, and biofluid, J. Am. Soc. Mass Spectrom., 2003, 14(11), 1290–1294 CrossRef CAS PubMed.
- G. Clarke, K. N. Ting, C. Wiart and J. Fry, High correlation of 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging, ferric reducing activity potential and total phenolics content indicates redundancy in use of all three assays to screen for antioxidant activity of extracts of plants from the Malaysian rainforest, Antioxidants, 2013, 2(1), 1–10 CrossRef CAS PubMed.
- Y. M. Muflihah, G. Gollavelli and Y.-C. Ling, Correlation study of antioxidant activity with phenolic and flavonoid compounds in 12 Indonesian indigenous herbs, Antioxidants, 2021, 10(10), 1530 CrossRef CAS PubMed.
- H. Zhao, W. Fan, J. Dong, J. Lu, J. Chen, L. Shan, Y. Lin and W. Kong, Evaluation of antioxidant activities and total phenolic contents of typical malting barley varieties, Food Chem., 2008, 107(1), 296–304 CrossRef CAS.
- T. Mezadri, D. Villaño, M. S. Fernández-Pachón, M. C. García-Parrilla and A. M. Troncoso, Antioxidant compounds and antioxidant activity in acerola (Malpighia emarginata, dc.) fruits and derivatives, J. Food Compos. Anal., 2008, 21(4), 282–290 CrossRef CAS.
- J. S. Kim, Antioxidant activities of selected berries and their free, esterified, and insoluble-bound phenolic acid contents, Prev. Nutr. Food Sci., 2018, 23(1), 35–45 CrossRef CAS PubMed.
- M.-M. Li, Y.-T. Chen, J.-C. Ruan, W.-J. Wang, J.-G. Chen and Q.-F. Zhang, Structure-activity relationship of dietary flavonoids on pancreatic lipase, Curr. Res. Food Sci., 2023, 6, 100424 CrossRef CAS PubMed.
- L. Han, H. Wang, J. Cao, Y. Li, X. Jin, C. He and M. Wang, Inhibition mechanism of α-glucosidase inhibitors screened from tartary buckwheat and synergistic effect with acarbose, Food Chem., 2023, 420, 136102 CrossRef CAS PubMed.
- T. K. Hyun, S. H. Eom and J.-S. Kim, Molecular docking studies for discovery of plant-derived α-glucosidase inhibitors, Plant Omics, 2014, 7(3), 166–170 Search PubMed.
- T. Maradesha, S. M. Patil, K. A. Al-Mutairi, R. Ramu, S. V. Madhunapantula and T. Alqadi, Inhibitory effect of polyphenols from the whole green jackfruit flour against α-glucosidase, α-amylase, aldose reductase and glycation at multiple stages and their interaction: Inhibition kinetics and molecular simulations, Molecules, 2022, 27(6), 1888 CrossRef CAS PubMed.
- A. O. Ademosun, G. Oboh, F. Bello and P. O. Ayeni, Antioxidative properties and effect of quercetin and its glycosylated form (rutin) on acetylcholinesterase and butyrylcholinesterase activities, J. Evidence-Based Complementary Altern. Med., 2015, 21(4), NP11–NP7 CrossRef PubMed.
- L.-H. Ye, R. Zhang and J. Cao, Screening of β-secretase inhibitors from Dendrobii caulis, by covalently enzyme-immobilized magnetic beads coupled with ultra-high-performance liquid chromatography, J. Pharm. Biomed. Anal., 2021, 195, 113845 CrossRef CAS PubMed.
- S. Viswanathan, T. Arumugam, K. Subramanian, R. Sivaraj, V. Ramesh and A. H. R. Vasanthi, Molecular docking analysis of flavonoids with ACHE and BACE-1, Bioinformation, 2024, 20(2), 103–109 CrossRef PubMed.
- N. I. S. Mohamed Yusof, Z. L. Abdullah, N. Othman and F. Mohd Fauzi, Structure–activity relationship analysis of flavonoids and its inhibitory activity against bace1 enzyme toward a better therapy for Alzheimer's disease, Front. Chem., 2022, 10, 2022 Search PubMed.
- R. Chakraborty, V. Vepuri, S. D. Mhatre, B. E. Paddock, S. Miller, S. J. Michelson, R. Delvadia, A. Desai, M. Vinokur, D. J. Melicharek, S. Utreja, P. Khandelwal, S. Ansaloni, L. E. Goldstein, R. D. Moir, J. C. Lee, L. P. Tabb, A. J. Saunders and D. R. Marenda, Characterization of a Drosophila Alzheimer's disease model: Pharmacological rescue of cognitive defects, PLoS One, 2011, 6(6), e20799 CrossRef CAS PubMed.
- T. C. Chou, Drug combination studies and their synergy quantification using the chou-talalay method, Cancer Res., 2010, 70(2), 440–446 Search PubMed.
- B. Yadav, K. Wennerberg, T. Aittokallio and J. Tang, Searching for drug synergy in complex dose–response landscapes using an interaction potency model, Comput. Struct. Biotechnol. J., 2015, 13, 504–513 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |
