Open Access Article
Open Access ArticleAntioxidant pine nut protein hydrolysate as a therapeutic oral immunotherapy agent in a murine model of pine nut allergy
Nourelimane
Benzitoune
abcd,
David
Menchén-Martínez
a,
Daniel
Lozano-Ojalvo
a,
Nabil
Kadri
c,
Rosina
López-Fandiño
 a,
Elena
Molina
a and
Sara
Benedé
a,
Elena
Molina
a and
Sara
Benedé
 *a
*a
aInstituto de Investigación en Ciencias de la Alimentación (CIAL), CSIC-UAM, Nicolás Cabrera, 9, 28049, Madrid, Spain. E-mail: s.benede@csic.es
bLaboratoire de Gestion et Valorisation des Ressources Naturelles et Assurance Qualité (LGVRNAQ), Faculté des Sciences de la Nature et de la Vie et des Sciences de la Terre, Université de Bouira, 10000 Bouira, Algérie
cDépartement des Sciences Biologiques, Faculté des Sciences de la Nature et de la Vie et des Sciences de la Terre, Université de Bouira, 10000 Bouira, Algérie
dDépartement de Biologie, Faculté des Sciences de la Nature et de la Vie et des Sciences de la Terre, Université de Khemis Miliana, 44000 Ain defla, Algérie
First published on 2nd February 2026
Abstract
Oral immunotherapy (OIT) is one of the most promising treatments for food allergy, although no protocols have yet been described for pine nut allergy. We aim to produce a pine nut protein hydrolysate to be used as a treatment agent in OIT. Hydrolysates were produced using three proteases (pepsin, trypsin, and Alcalase) alone or in combination and their antioxidant activity, antigenicity, and immunomodulatory properties were determined in vitro. Pine nut-sensitized C57BL/6 mice were subjected to OIT with intact and hydrolysed pine nut protein. The trypsin hydrolysate showed higher antioxidant activity and lower IgE binding capacity than the intact protein, and it was able to desensitize mice in a similar way to the unhydrolyzed protein. Therefore, it could be regarded as a good candidate as a therapeutic agent in OIT on the grounds of its effectiveness in desensitizing allergic mice and its improved antioxidant and immunoreactive properties with respect to intact pine nut protein. Oral immunotherapy is one of the most promising treatments for food allergy, although no protocols have yet been described for pine nut allergy. To identify pine nut protein hydrolysates with potential for OIT, we applied enzymatic hydrolysis using food-grade enzymes and a two-step selection strategy. Hydrolysates were first screened for in vitro antioxidant activity and then assessed for their ability to modulate allergen-specific T-cell responses ex vivo. The trypsin-derived hydrolysate showed the most promising profile, leading to further evaluation of its IgE-binding properties and therapeutic potential in pine nut-sensitized C57BL/6 mice subjected to OIT with intact and hydrolysed pine nut protein. Mice treated with the trypsin hydrolysate showed reduced specific IgE and IgG1 levels and were protected against oral pine nut-induced anaphylaxis, similar to those treated with intact pine nut protein. Due to its ability to desensitize allergic mice, combined with lower IgE binding and higher antioxidant activity, the trypsin-hydrolysed pine nut protein appears to be a promising candidate for OIT.
Introduction
Food allergy has emerged as a pressing global public health concern, displaying a steadily increasing prevalence over the past few decades. It encompasses a range of immune-mediated adverse responses triggered by specific food proteins, affecting up to 10% of certain populations, with a predilection for industrialized regions and emerging economies.1Nine primary allergenic foods have been identified, including tree nuts, which are known for causing potentially fatal anaphylactic reactions, often triggered by IgE-mediated responses. Tree nut allergies are closely linked to specific protein families, such as 2S albumins, vicilins, legumins, and nonspecific lipid transfer proteins.2,3 Allergic reactions to pine nuts are relatively less common than to other nuts, such as walnuts, cashews, hazelnuts or almonds, although they are particularly alarming due to the significant frequency of severe anaphylactic responses (approximately 80%) triggered by even small quantities of pine nuts.4,5 A high proportion (70%) of pine nut allergic patients are monosensitized5–7 and there is limited cross-reactivity between pine nuts and more commonly consumed nuts, primarily due to the evolutionary differences between gymnosperms (pine nuts) and angiosperms (other nuts), which contribute to the distinct characteristics of pine nut allergy.4
Pine nut allergy management is still limited to strict dietary avoidance, basically because allergen-specific immunotherapy using the whole allergenic food is often accompanied by severe side effects.4 Oral immunotherapy (OIT) has been implemented mainly against peanut, egg, and milk allergies to induce immunomodulation and protect against accidental allergen exposure. However, concerns about its safety and ability to establish permanent tolerance persist.8,9 Alternative food allergy treatments have emerged, such as hydrolysed protein-based OIT, which offers the advantage of a reduced risk of adverse reactions compared to intact allergen-based OIT. In addition, the peptides resulting from the hydrolysis of allergens may have immunomodulatory properties maintaining and reinforcing barrier function and promoting immune homeostasis through their action on the cells of the innate and adaptive immune system.10–12 Peptides derived from food proteins may also exhibit antioxidant properties,13 which could have an additional beneficial effect during immunotherapy treatments as there is ample evidence that high levels of oxidative stress may play an important role in the pathogenesis of allergic disorders.14,15 An imbalance between reactive oxygen species (ROS) production and antioxidant defence during allergen encounter may favour a T helper 2 cell immune response.16 In fact, certain clinical trials suggest that specific antioxidants from diet or vitamin supplements might improve asthma control or lung function in asthmatic children and adults.17,18
To the best of our knowledge, there are no previous reports on the use of pine nut protein hydrolysates in OIT. To identify hydrolysates with optimal properties, after performing enzymatic hydrolysis of pine nut proteins using food-grade enzymes, either individually or sequentially, a two-step selection strategy was applied: first, hydrolysates were evaluated for antioxidant activity using multiple in vitro methods as the primary criterion; second, their ability to down-regulate ex vivo T-cell responses induced by the pine nut allergen was considered as a secondary criterion. Among the tested hydrolysates, the trypsin-derived hydrolysate exhibited the most favourable profile. After assessing its IgE-binding properties with sera from pine nut-allergic patients, we further investigated its therapeutic potential in vivo using the C57BL/6 mouse model.
Materials and methods
Preparation of protein isolate and protein hydrolysates
Pine nuts (Pinus pinea L), skinned and shelled, were bought from a local market (Blida, Algeria). To prepare pine nut protein isolate (PI), clean and dried pine nuts were ground and defatted by Soxhlet extraction using n-hexane. Then, a solution of defatted flour in MilliQ water at 125 mg mL−1 was prepared and centrifuged at 3000g for 10 min and the supernatant was freeze-dried.Pine nut protein hydrolysates were obtained using pepsin (P; Sigma-Aldrich, St. Louis, MO, USA) at an enzyme-to-substrate (E/S) ratio of 128.034 U mg−1 protein in 200 mM KCl/HCl buffer, pH 2.0, at 37 °C for 4 h; trypsin (T; Sigma-Aldrich) at an E/S ratio of 0.3377 U mg−1 protein in 50 mM phosphate buffer, pH 8.0, at 37 °C for 4 h; and Alcalase (A; Novozyme, Bagsvaered, Denmark) at an E/S ratio of 62.205 U mg−1 protein in 50 mM phosphate buffer, pH 8.0, at 55 °C for 3 h. Hydrolysis was also carried out using combinations of pepsin/trypsin (PT), pepsin/Alcalase (PA), trypsin/Alcalase (TA), and pepsin/trypsin/Alcalase (PTA). Sequential hydrolysis was initiated by adding the first enzyme and allowing it to hydrolyse the protein under the conditions mentioned above. Then, the pH was adjusted with 6 M NaOH, and the next enzyme was added and incubated for an additional 2 h at the indicated temperature. Upon completion of the hydrolysis, the reaction mixtures were heated at 85 °C for 15 min to inactivate enzymes and centrifuged at 8000g for 15 min. Supernatants were collected and freeze-dried for further analysis.
The protein content of PI and its hydrolysates was quantified by determining their nitrogen percentage by elemental analysis. The lipopolysaccharide present in the samples was removed by using Pierce™ high capacity endotoxin removal spin columns (Thermo Fisher Scientific, Waltham, MA, USA) and its absence was confirmed by using the transfected cell line THP1-XBlue™, stably expressing an NF-κB/AP-1-inducible secreted alkaline phosphatase reporter assessed by the QUANTI-Blue™ assay (InvitroGen, Carlsbad, CA, USA), following the manufacturer's instructions.
SDS-PAGE analyses
The SDS-PAGE analysis of the samples was performed as previously described.19 Concisely, the samples were dissolved in sample buffer containing 2-mercaptoethanol, heated at 95 °C for 5 min and loaded (30 µg protein per well) onto 4–12% Bis-Tris Criterion XT precast gel (Bio-Rad, Hercules, CA, USA). The gel was stained with Bio-safe Coomassie blue R-250 (Bio-Rad). As a molecular weight marker, Precision Plus Protein TM Dual Xtra Prestained Protein Standard (Bio-Rad) was used.Determination of antioxidant activity
The ABTS, DPPH, and OH radical scavenging activities of the samples were assessed, respectively, according to the methods described by Zhang et al.,20 Farvin et al.,21 and Mintah et al.22 The oxygen radical absorbance capacity (ORAC) of the samples was measured following the method described by Vasquez-Rojas et al.23 The ability of samples to chelate ferrous iron was evaluated based on the procedure described by Xia et al.,24 and the method described by He et al.25 was followed for the determination of the ferric reducing power (FRAP) of the samples.In vitro gastrointestinal digestion
The in vitro gastrointestinal digestion of PI and T was performed according to the INFOGEST 2.0 protocol.26 For the gastric phase, PI and T solutions (15 mg mL−1 in the final mixture), mixed with simulated gastric fluid, were adjusted to a pH of 3.0 with 1 M HCl. Pepsin from porcine gastric mucosa was added to achieve a concentration of 2000 U mL−1 in the final mixture, followed by a 2-hour incubation at 37 °C. Pepsin activity was halted by raising the pH to 7.0 using 1 M NaHCO3. Subsequently, gastric samples were mixed with simulated intestinal fluid. The pH was adjusted to 7.0 with 1 M NaOH, and CaCl2 (0.075 mM in the final digestion mixture) was introduced. To simulate the intestinal phase, pancreatin from porcine pancreas (100 U mL−1 in the final digestion mixture based on its trypsin activity) and bovine bile extract (10 mM in the final digestion mixture) were added. The mixtures were subjected to an additional 2-hour incubation and the process was halted with a Bowman–Birk protease inhibitor. Samples were immediately frozen and stored at −80 °C for subsequent analyses.Immunodetection assays
The human IgE binding capacity of samples was assessed by inhibition ELISA and western blotting as previously reported,27 using a pool of 5 sera from pine nut-allergic patients diagnosed through a compatible clinical history and positive skin prick test. Serum samples were obtained from the Institute of Food Science Research (CIAL) serum library in compliance with relevant institutional and national guidelines and regulations and in accordance with the Declaration of Helsinki. The study protocol was approved by the CIAL Bioethics Committee. Written informed consent was obtained from all participants prior to inclusion. Human samples were processed in a coded manner to protect participant confidentiality and stored at −80 °C until analysis.Ex vivo experiments in spleen cells from sensitized mice
The immunomodulatory properties of the hydrolysates were assessed on splenocytes from mice sensitized to pine nut protein. Six-week-old female C57BL/6 mice were purchased from Charles River Laboratories (Saint Germain sur l'Arbresle, France). Mice (n = 5) were sensitized to PI by the oral administration of 5 mg of PI protein per mouse plus 10 μg of cholera toxin (CT, List Biologicals, Campbell, CA) in PBS, during 3 consecutive days on the first week and once per week during the following 5 weeks (SI Fig. 1A). Mice were euthanized by CO2 inhalation and spleens were collected and processed under sterile conditions. Splenocytes from individual animals were cultured in 96-well plates (4 × 106 cells per mL) and stimulated in triplicate with PI alone or in combination with its hydrolysates (200 μg mL−1). Stimulation with concanavalin A (2.5 μg mL−1) was used as a positive control, and PBS as a negative control. Inactivated enzymes were also tested at a concentration equivalent to that present in 200 μg mL−1 hydrolysates. Supernatants were collected after 72 h at 37 °C in 5% CO2 and detection of IL-4 and IFN-γ was performed by ELISA, using commercial kits (Invitrogen).In vivo experiments in sensitized mice
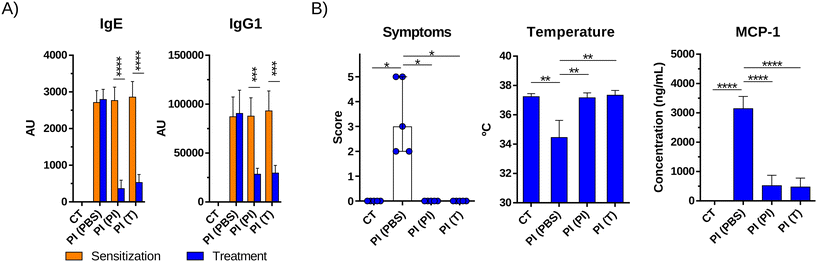
For the in vivo evaluation of the therapeutic capacity of pine nut hydrolysate with trypsin (T), three groups of six-week-old female C57BL/6 mice (n = 5) were sensitized to PI as described above, while mice from a fourth group (n = 5) just received 10 μg of CT (sham-sensitized mice). One week apart, mice from two of the PI-sensitized groups were orally treated with 5 mg of PI or T protein per mouse, three times per week for 3 weeks. Mice from the other PI-sensitized group and the sham-sensitized group received PBS during the therapy. Three days after the last treatment dose, all mice were orally challenged with 50 mg of PI (SI Fig. 1B). Anaphylactic responses were evaluated by measuring the body temperature with a rectal thermometer and scoring clinical signs 30 min after challenge with the scale described in Pablos-Tanarro et al.28 Blood samples were collected for pine nut specific IgE and IgG1 analyses by indirect ELISA as previously described.28 Mouse mast-cell protease 1 (MCP-1) was analysed using an ELISA Kit (Invitrogen).All protocols involving animals followed the European Legislation (Directive 2010/63/EU) and were approved by the Comunidad de Madrid (Ref PROEX 286.8/20).
Statistical analyses
Statistical analyses were performed using GraphPad Prism version 7.0e (GraphPad Software Inc., San Diego, CA). For Fig. 1, 2, and 3C, differences among groups were assessed by one-way ANOVA followed by Tukey's multiple comparisons test. For Fig. 3, differences were assessed using a two-tailed Student's t test. For Fig. 4 and 5A, data were analyzed by two-way ANOVA followed by Šidák's multiple comparisons test. Clinical sign scores (Fig. 5b) were analyzed using the Mann–Whitney U test. Statistical significance was set at P < 0.05.Results
Antioxidant and immunomodulant activity of pine nut hydrolysates
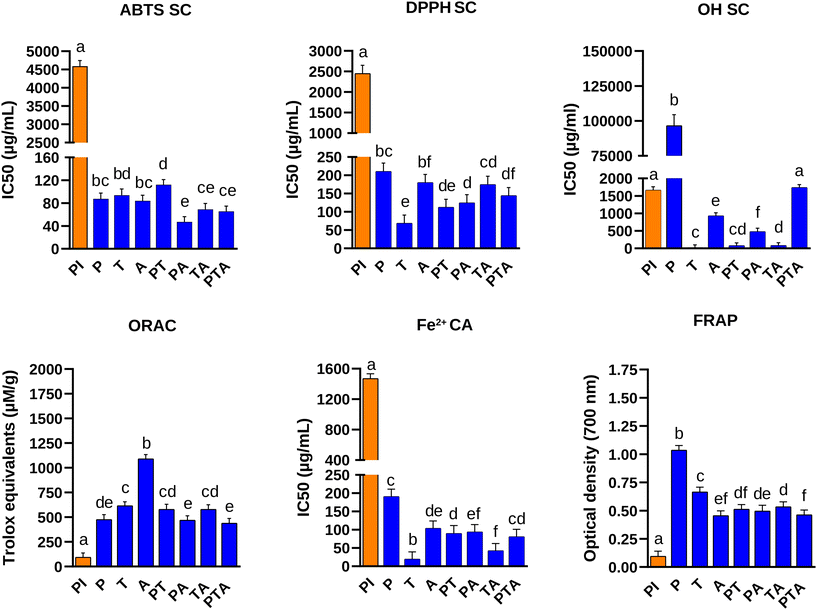
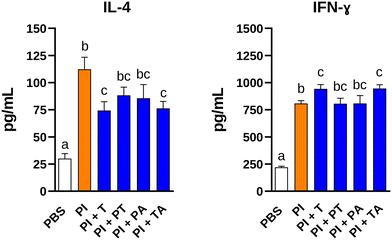
Under reducing conditions, PI showed protein bands from ∼100 to ∼10 kDa. Enzymatic hydrolysis removed most high-molecular-mass proteins, leaving only <10 kDa bands after Alcalase or combined enzyme treatments, while P and T retained bands up to ∼15 and 20 kDa, respectively (SI Fig. 2). The antioxidant capacity of the hydrolysates was measured by different in vitro methods (Fig. 1). Overall, all hydrolysates showed better antioxidant capacity than PI. Radical scavenging assays revealed that the lowest IC50 values were observed for PA in the ABTS assay, for T, PT, and PA in the DPPH assay, and for T, PT, PA, and TA in the OH radical assay. All hydrolysates showed ORAC antioxidant capacity values ranging from 439 to 615 µM trolox equivalents per g except for A which showed a higher capacity reaching up to 1090 µM trolox equivalents per g. T and TA were the hydrolysates that showed the highest ferrous iron chelating capacity with IC50 values of 19.5 and 42.4 µg mL−1, respectively. The hydrolysate that showed the best ferric reducing ability was P, but it was excluded from further consideration due to its comparatively low antioxidant capacity in the other assays performed. On the basis of the highest overall antioxidant capacity obtained with the 6 methods tested, we selected T, PT, PA, and TA for further evaluation of their immunomodulatory capacity.In order to test whether the allergen-induced responses on immune cells from pine nut sensitized mice (SI Fig. 1A) could be modulated by the hydrolysates, splenocytes were stimulated with the PI used for sensitization in the presence of its hydrolysates at a concentration of 200 μg mL−1 of each substance, and the impact on the secretion of Th2 (IL-4) and Th1 (IFN-γ) cytokines was determined (Fig. 2). PI-induced production of IL-4 was inhibited by co-incubation of spleen cells from PI-sensitised mice with the hydrolysates of PI with T and TA. On the other hand, these hydrolysates tended to promote the release of the Th1 cytokine IFN-γ, when used together with PI for the stimulation of the spleen cells from sensitized mice. However, PT and PA failed to reduce IL-4 secretion and increase the levels of IFN-γ induced by PI.
Characterisation of pine nut hydrolysate with trypsin
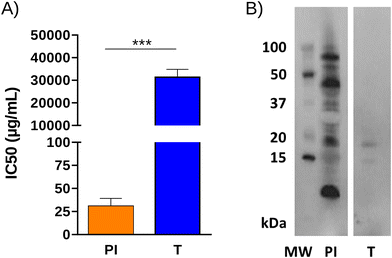
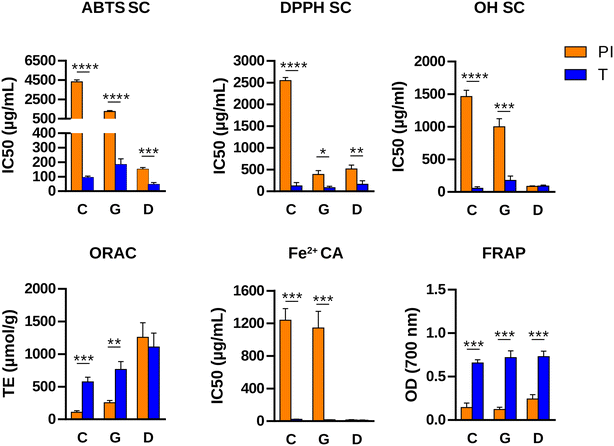
According to the results explained above, T and TA were the hydrolysates with the highest overall antioxidant capacity and were also able to modulate specific T-cell responses induced by the sensitizing allergen. Ultimately, we opted for T as a candidate for peptide immunotherapy because a single-step hydrolysis simplifies production and ensures high reproducibility. In order to evaluate its potential allergenicity, IgE binding was determined by inhibition ELISA using a pool of sera from 5 pine nut allergic patients. Fig. 3 shows that the IgE binding capacity of PI was significantly reduced after hydrolysis with trypsin, with EC50 values (concentration for 50% of maximum IgE binding) a thousand times higher. Western blot (Fig. 3B) showed that most protein bands in PI were recognized by patient IgE. T still retained residual IgE binding capacity, mainly due to two bands of approximately 15 and 20 kDa that remained unhydrolyzed.We next evaluated the influence of in vitro gastrointestinal digestion on the immunoreactive and antioxidant properties of the hydrolysate. The IgE binding capacity of both PI and T decreased significantly throughout the digestion process, with the percentage of IgE binding being the same for both samples after gastric digestion and remaining similar until the end of duodenal digestion (data not shown). Fig. 4 shows the results of the antioxidant activity determined by different methods after 2 h of simulated gastric digestion, followed by 2 h of duodenal digestion of PI and T. After undergoing pepsin action during gastric digestion, the antioxidant activity of T was significantly higher than that of PI. The same trend was observed after subsequent duodenal digestion when the antioxidant activity was measured by the ABTS and DPPH radical scavenging assays and the FRAP assay. However, when the antioxidant activity was measured by the OH radical scavenging, ferrous iron chelating, and ORAC methods, the values obtained for the gastrointestinal digests of PI and T were similar. Although in vitro digestion models do not fully replicate physiologically relevant conditions, these findings suggest that the hydrolysate may possess superior properties in vivo compared to the parent extract.
In vivo evaluation of the therapeutic capacity of pine nut hydrolysate with trypsin
Pine nut allergy was induced in mice by using the oral sensitization and boosting protocol shown in SI Fig. 1B. Starting at week 6 of the protocol, mice were given 4 weekly oral treatments with PI or T for 5 weeks and the concentrations of specific antibodies were measured in the serum. The levels of specific IgE and IgG1 were significantly reduced in both groups of treated mice, while the levels in the non-treated group remained unchanged (Fig. 5A). Three days after the last treatment dose, mice were orally challenged with PI and no visual signs of anaphylaxis or decrease in body temperature were detected in the treated groups (Fig. 5B). In contrast, the untreated group experienced obvious clinical signs of anaphylaxis and a significant drop in body temperature. Moreover, serum levels of MCP-1, a protease released by activated intestinal mast cells, which is indicative of mast cell degranulation, were significantly lower in the treated mice than in the non-treated ones (Fig. 5B).Discussion
We used enzymatic hydrolysis of pine nut protein isolate to produce protein hydrolysates with antioxidant and immunomodulatory activities. SDS-PAGE analysis showed protein bands (6–11 kDa) that resisted enzymatic cleavage and were present in all hydrolysates. Cabanillas et al. identified a band with a molecular mass of around 6 kDa under reducing conditions as a 2S albumin known as Pin p 1, the first characterized allergen in Pinus pinea L. nuts.5 This protein has been reported to be resistant to heat treatment and cleavage by digestive enzymes. However, a band with this molecular mass was reactive to human IgE only in the non-hydrolysed extract (PI).Our investigation delved into the antioxidant activity of pine nut protein hydrolysates using a diverse array of assays to cover the variety of mechanisms by which antioxidants inhibit oxidative routes, including inactivation of reactive oxygen species, scavenging of free radicals, chelation or sequestration of metal catalysts, or reduction of ferric ions, among others.29 The results obtained with each of the methodologies used depend on the presence of amino acids in peptides that serve as electron or hydrogen donors,30 with sequence position and hydrophobicity also playing crucial roles.31 The ABTS assay may be more useful than the DPPH assay for detecting the scavenging capacity of hydrophilic antioxidants in foods,32 while the presence of hydrophobic and aromatic amino acids could contribute to the ability of hydrolysates to neutralize DPPH radicals.33 The hydroxyl radical (OH) is one of the most toxic reactive oxygen radicals known, which causes damage to all important biomolecules involving DNA, proteins, and membranes.34 Notably, OH radical scavenging activity was observed in T, PT, and TA hydrolysates, likely attributed to the presence of amino acids with hydrogen atom donation capabilities that could mitigate hydrogen peroxide-induced oxidative stress,35 such as Arg and Lys, which can become exposed after trypsin cleaves at their carboxyl side. In the ORAC assay, A exhibited a particularly high antioxidant activity, probably attributed to the ability of this enzyme to expose hydrophobic amino acids during hydrolysis,36 potentially enhancing hydrophobic interactions between peptides and the oxidizing agent.31 Pine nut hydrolysates also displayed high Fe2+ chelating activity and substantial ferric reducing antioxidant power, which could be attributed to peptides containing sulfhydryl amino acids such as Cys and Met. These residues can bind heavy metals, reducing their pro-oxidant activity,37 and donate electrons to radicals, thereby stabilizing them and preventing oxidative damage.38
Assessment of the immunomodulatory capacity of pine nut protein hydrolysates on splenocytes from sensitized C57BL/6 mice revealed that, among the hydrolysates with the highest antioxidant activity, T and TA could be regarded as good candidates for peptide-based immunotherapy on the grounds of their potential to reduce ex vivo Th2 responses induced by pine nut protein, while also enhancing Th1 responses. Previous results from our group have shown that ovalbumin and egg white hydrolysates, selected for their low IgE binding and their ability to reduce Th2 responses in splenocytes from sensitized mice while enhancing Th1 responses, effectively promoted oral tolerance to the corresponding intact allergens in mouse models.39,40 This effect was associated with the induction of a tolerogenic environment in the intestine that promotes the expansion of regulatory T cells.41 Overall, these findings indicate that allergen-derived peptides capable of dampening inflammation and redirecting immune responses of specific cell populations can enhance the effectiveness of immunotherapy. A simple hydrolysis step with trypsin appears to be a cheaper and simpler method for the reproducible production of pine nut-derived antioxidant and immunomodulant peptides than combined hydrolysis with trypsin and Alcalase. Furthermore, there is evidence that bacterial enzymes, such as Alcalase, may induce sensitization in susceptible individuals,42 while the European Food Safety Authority considers that the risk of allergic sensitisation to trypsin from porcine pancreas is low.43 Therefore, the hydrolysate obtained with trypsin was selected for further examination, including the assessment of its potential hypoallergenic effect using sera from pine nut allergic subjects. Numerous studies have highlighted the efficacy of enzymatic hydrolysis for reducing the allergenicity of various food proteins.39,44 In our study, the IgE binding capacity of PI was significantly reduced after hydrolysis with trypsin, similarly to what has been previously reported for other hydrolysates of different food substrates obtained with this enzyme.45 During the digestion process, antioxidant peptides may be further hydrolysed by gastrointestinal enzymes, thereby altering their activities. In this study, the antioxidant activity of T was higher than that of PI after the action of digestive enzymes, as described in the case of other food peptides,46,47 although this effect was more pronounced at the end of the gastric phase.
To evaluate the use of the pine nut protein hydrolysate with trypsin as a therapeutic agent, once pine nut allergy was established in mice, T was administered for 3 weeks. Mice treated with the hydrolysate were protected against anaphylaxis induced by an oral pine nut challenge to the same extent as those treated with intact pine nut protein. Protection was accompanied by a reduction in serum pine nut-specific IgE and IgG1 levels, consistent with previous reports of successful desensitization in mouse models.39
The clinical efficacy of OIT is largely attributed to the activation of T cells by specific epitopes, which initiates counter-regulatory immune mechanisms that reduce Th2 cell activity and expansion.48 Fragmented allergens offer a distinct advantage in OIT because they preserve T cell-stimulating epitopes while lacking the ability to cross-link IgE on effector cells, thereby minimizing the risk of adverse reactions. However, the selection of peptides that balance immunostimulatory potential with reduced allergenicity remains challenging, as longer peptides provide broader T cell epitope coverage but increase the likelihood of IgE cross-linking.49 Since enzymatic hydrolysis typically generates short fragments with limited sequence coverage, potentially reducing the proportion of full-length T epitopes, the comparable desensitization achieved with PI and T suggests that the latter may exert superior immunomodulatory effects, possibly by promoting a shift from Th2 to Th1 responses and/or inducing regulatory T cells. This dual benefit of reduced allergenicity combined with preserved or enhanced immunomodulatory capacity positions the pine nut hydrolysate with trypsin as a promising candidate for safer and more effective OIT strategies. Such an approach could mitigate adverse reactions during therapy, which are a major barrier to patient adherence, although this aspect cannot be properly evaluated in murine models.
Conclusion
In conclusion, the hydrolysate of pine nuts with trypsin could be regarded as a good candidate for a therapeutic agent in OIT on the grounds of its effectiveness in desensitizing pine nut allergic mice with the added properties of having lower IgE binding capacity and higher antioxidant activity than intact pine nut protein.Author contributions
N. B.: investigation, methodology, and writing – original draft; D. M-M.: investigation; D. L-O. and N. K.: review; R. L-F. and E. M.: funding acquisition, conceptualization, validation, and writing – review & editing; and S. B.: investigation, methodology, writing – original draft, conceptualization, validation, and writing – review & editing. We acknowledge the Bioanalytical Techniques Unit (BAT) at CIAL for their technical support and services. D. M-M., D. L-O., R. L-F., E. M., and S. B. are members of the CSIC Interdisciplinary Thematic Platform (PTI) “Food Allergy”.Conflicts of interest
The authors declare that they have no competing interests.Data availability
The data generated during the current study are available from the corresponding author on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04650e.
Acknowledgements
This work was supported by the project PID2021-123236OB-I00. D. Menchén-Martínez was the recipient of an FPI grant (all from the Ministerio de Ciencia, Innovación y Universidades).References
- J. P. Lopes and S. Sicherer, Food allergy: epidemiology, pathogenesis, diagnosis, prevention, and treatment, Curr. Opin. Immunol., 2020, 66, 57–64 CrossRef CAS PubMed.
- M. P. Borres, S. Sato and M. Ebisawa, Recent advances in diagnosing and managing nut allergies with focus on hazelnuts, walnuts, and cashew nuts, World Allergy Organ. J., 2022, 15, 100641 CrossRef CAS PubMed.
- B. Elrahi, Z. Mehanna, S. Haidar, M. Serhan and H. F. Hassan, Food allergies and allergens in Lebanon: characterization and perceptions toward labeling, World Allergy Organ. J., 2023, 16, 100743 CrossRef PubMed.
- B. Cabanillas and N. Novak, Allergic reactions to pine nut: a review, J. Invest. Allergol. Clin. Immunol., 2015, 25, 329–333 CAS.
- B. Cabanillas, J. F. Crespo, S. J. Maleki, J. Rodriguez and N. Novak, Pin p 1 is a major allergen in pine nut and the first food allergen described in the plant group of gymnosperms, Food Chem., 2016, 210, 70–77 CrossRef CAS PubMed.
- B. Cabanillas, H. Cheng, C. C. Grimm, B. K. Hurlburt, J. Rodríguez, J. F. Crespo and S. J. Maleki, Pine nut allergy: clinical features and major allergens characterization, Mol. Nutr. Food Res., 2012, 56, 1884–1893 CrossRef CAS PubMed.
- J. F. Crespo, C. Bueno, M. Villalba, L. Monaci, C. Cuadrado, N. Novak and B. Cabanillas, Epitope mapping of the major allergen 2S albumin from pine nut, Food Chem., 2021, 339, 127895 CrossRef CAS PubMed.
- M. Vazquez-Ortiz and P. J. Turner, Improving the safety of oral immunotherapy for food allergy, Pediatr. Allergy Immunol., 2016, 27, 117–125 CrossRef PubMed.
- G. Pouessel and G. Lezmi, Oral immunotherapy for food allergy: translation from studies to clinical practice?, World Allergy Organ. J., 2023, 16, 100747 CrossRef CAS PubMed.
- D. Lozano-Ojalvo, E. Molina and R. López-Fandiño, Hydrolysates of egg white proteins modulate T- and B-cell responses in mitogen-stimulated murine cells, Food Funct., 2016, 7, 1048–1056 RSC.
- D. Lozano-Ojalvo and R. López-Fandiño, Immunomodulating peptides for food allergy prevention and treatment, Crit. Rev. Food Sci. Nutr., 2018, 58, 1629–1649 CrossRef CAS PubMed.
- D. Lozano-Ojalvo, M. Martínez-Blanco, L. Pérez-Rodríguez, E. Molina and R. López-Fandiño, Oral immunotherapy with egg peptides induces innate and adaptive tolerogenic responses, Mol. Nutr. Food Res., 2019, 63, e1900144 CrossRef PubMed.
- S. Benedé and E. Molina, Chicken egg proteins and derived peptides with antioxidant properties, Foods, 2020, 9, 735 CrossRef PubMed.
- R. P. Bowler and J. D. Crapo, Oxidative stress in allergic respiratory diseases, J. Allergy Clin. Immunol., 2002, 110, 349–356 CrossRef CAS PubMed.
- M. Han, D. Lee, S. H. Lee and T. H. Kim, Oxidative stress and antioxidant pathway in allergic rhinitis, Antioxidants, 2021, 10, 1266 CrossRef CAS PubMed.
- L. Utsch, C. Folisi, J. H. Akkerdaas, A. Logiantara, M. A. van de Pol, J. S. van der Zee, E. J. Krop, R. Lutter, R. van Ree and L. S. van Rijt, Allergic sensitization is associated with inadequate antioxidant responses in mice and men, Allergy, 2015, 70, 1246–1258 CrossRef CAS PubMed.
- C. H. Guo, P. J. Liu, K. P. Lin and P. C. Chen, Nutritional supplement therapy improves oxidative stress, immune response, pulmonary function, and quality of life in allergic asthma patients: an open-label pilot study, Altern. Med. Rev., 2012, 17, 42–56 Search PubMed.
- E. Pendezza, C. Nava, A. Leone, F. Riccaboni, A. Bosetti, G. V. Zuccotti, S. Bertoli and E. D'Auria, Antioxidant potential of the diet in Italian children with food allergies, Front. Nutr., 2023, 10, 1096288 CrossRef PubMed.
- S. Benedé, I. López-Expósito, R. López-Fandiño and E. Molina, Identification of IgE-binding peptides in hen egg ovalbumin digested in vitro with human and simulated gastroduodenal fluids, J. Agric. Food Chem., 2014, 62, 152–158 CrossRef PubMed.
- W. Zhang, N. Jia, Z. Zhu, Y. Wang, J. Wang and C. Xue, Changes of antioxidative activities and peptidomic patterns of Auxenochlorella pyrenoidosa protein hydrolysates: effects of enzymatic hydrolysis and decoloration processes, LWT, 2021, 152, 112306 CrossRef CAS.
- K. H. Farvin, L. L. Andersen, H. H. Nielsen, C. Jacobsen, G. Jakobsen, I. Johansson and F. Jessen, Antioxidant activity of cod (Gadus morhua) protein hydrolysates: in vitro assays and evaluation in 5% fish oil-in-water emulsion, Food Chem., 2014, 149, 326–334 CrossRef PubMed.
- B. K. Mintah, R. He, M. Dabbour, M. K. Golly, A. A. Agyekum and H. Ma, Effect of sonication pretreatment parameters and their optimization on the antioxidant activity of Hermetia illucens larvae meal protein hydrolysates, J. Food Process. Preserv., 2019, 43, e14093 Search PubMed.
- W. V. Vasquez-Rojas, D. Martín, B. Miralles, I. Recio, T. Fornari and M. P. Cano, Composition of Brazil nut (Bertholletia excelsa HBK), its beverage and by-products: a healthy food and potential source of ingredients, Foods, 2021, 10, 3007 CrossRef CAS PubMed.
- Y. Xia, F. Bamdad, M. Gänzle and L. Chen, Fractionation and characterization of antioxidant peptides derived from barley glutelin by enzymatic hydrolysis, Food Chem., 2012, 134, 1509–1518 CrossRef CAS PubMed.
- R. He, A. T. Girgih, S. A. Malomo, X. Ju and R. E. Aluko, Antioxidant activities of enzymatic rapeseed protein hydrolysates and the membrane ultrafiltration fractions, J. Funct. Foods, 2013, 5, 219–227 CrossRef CAS.
- A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assunção, S. Balance, T. Bohn, C. Bourlieu-Lacanal, R. Boutrou, F. Carrière, A. Clemente, M. Corredig, D. Dupont, C. Dufour, C. Edwards, M. Golding, S. Karakaya, B. Kirkhus, S. Le Feunteun, U. Lesmes, A. Macierzanka, A. R. Mackie, C. Martins, S. Marze, D. J. McClements, O. Ménard, M. Minekus, R. Portmann, C. N. Santos, I. Souchon, R. P. Singh, G. E. Vegarud, M. S. J. Wickham, W. Weitschies and I. Recio, INFOGEST static in vitro simulation of gastrointestinal food digestion, Nat. Protoc., 2019, 14, 991–1014 CrossRef CAS PubMed.
- L. Martín-Pedraza, C. Mayorga, F. Gomez, C. Bueno-Díaz, N. Blanca-Lopez, M. González, M. Martínez-Blanco, J. Cuesta-Herranz, E. Molina, M. Villalba and S. Benedé, IgE-reactivity pattern of tomato seed and peel nonspecific lipid-transfer proteins after in vitro gastrointestinal digestion, J. Agric. Food Chem., 2021, 69, 3511–3518 CrossRef PubMed.
- A. Pablos-Tanarro, D. Lozano-Ojalvo, E. Molina and R. López-Fandiño, Assessment of the allergenic potential of the main egg white proteins in BALB/c mice, J. Agric. Food Chem., 2018, 66, 2970–2976 CrossRef CAS PubMed.
- K. Jomova, S. Y. Alomar, S. H. Alwasel, E. Nepovimova, K. Kuca and M. Valko, Several lines of antioxidant defense against oxidative stress: antioxidant enzymes, nanomaterials with multiple enzyme-mimicking activities, and low-molecular-weight antioxidants, Arch. Toxicol., 2024, 98, 1323–1367 CrossRef CAS PubMed.
- A. Sila and A. Bougatef, Antioxidant peptides from marine by-products: isolation, identification and application in food systems. A review, J. Funct. Foods, 2016, 21, 10–26 CrossRef CAS.
- A. G. Samaranayaka and E. C. Li-Chan, Food-derived peptidic antioxidants: a review of their production, assessment, and potential applications, J. Funct. Foods, 2011, 3, 229–254 CrossRef CAS.
- A. Floegel, D.-O. Kim, S.-J. Chung, S. I. Koo and O. K. Chun, Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods, J. Food Compos. Anal., 2011, 24, 1043–1048 CrossRef CAS.
- S. Zhang, M. Zhang, J. Xing and S. Lin, A possible mechanism for enhancing the antioxidant activity by pulsed electric field on pine nut peptide glutamine–tryptophan–phenylalanine–histidine, J. Food Biochem., 2019, 43, e12714 Search PubMed.
- S. B. Nimse and D. Pal, Free radicals, natural antioxidants, and their reaction mechanisms, RSC Adv., 2015, 5, 27986–28006 RSC.
- A. Phaniendra, D. B. Jestadi and L. Periyasamy, Free radicals: properties, sources, targets, and their implication in various diseases, Indian J. Clin. Biochem., 2015, 30, 11–26 CrossRef CAS PubMed.
- V. G. Tacias-Pascacio, R. Morellon-Sterling, E. H. Siar, O. Tavano, Á. Berenguer-Murcia and R. Fernandez-Lafuente, Use of Alcalase in the production of bioactive peptides: a review, Int. J. Biol. Macromol., 2020, 165, 2143–2196 CrossRef CAS PubMed.
- U. Urbizo-Reyes, M. F. San Martin-González, J. Garcia-Bravo, A. L. M. Vigil and A. M. Liceaga, Physicochemical characteristics of chia seed (Salvia hispanica) protein hydrolysates produced using ultrasonication followed by microwave-assisted hydrolysis, Food Hydrocolloids, 2019, 97, 105187 CrossRef CAS.
- M. M. Aondona, J. K. Ikya, M. T. Ukeyima, T. W. J. Gborigo, R. E. Aluko and A. T. Girgih, In vitro antioxidant and antihypertensive properties of sesame seed enzymatic protein hydrolysate and ultrafiltration peptide fractions, J. Food Biochem., 2021, 45, e13587 CrossRef CAS PubMed.
- D. Lozano-Ojalvo, E. Molina and R. López-Fandiño, Hypoallergenic hydrolysates of egg white proteins modulate allergen responses induced ex vivo on spleen cells from sensitized mice, Food Res. Int., 2016, 89, 661–669 CrossRef CAS PubMed.
- D. Lozano-Ojalvo, L. Pérez-Rodríguez, A. Pablos-Tanarro, E. Molina and R. López-Fandiño, Hydrolysed ovalbumin offers more effective preventive and therapeutic protection against egg allergy than the intact protein, Clin. Exp. Allergy, 2017, 47, 1342–1354 CrossRef CAS PubMed.
- D. Lozano-Ojalvo, M. Martínez-Blanco, L. Pérez-Rodríguez, E. Molina, C. Peláez, T. Requena and R. López-Fandiño, Egg white peptide-based immunotherapy enhances vitamin A metabolism and induces RORγt+ regulatory T cells, J. Funct. Foods, 2019, 52, 204–211 CrossRef CAS.
- D. I. Bernstein, I. L. Bernstein, W. G. Gaines Jr., T. Stauder and E. R. Wilson, Characterization of skin prick testing responses for detecting sensitization to detergent enzymes at extreme dilutions: inability of the RAST to detect lightly sensitized individuals, J. Allergy Clin. Immunol., 1994, 94, 498–507 CrossRef CAS PubMed.
- EFSA Panel on Food Contact Materials, Enzymes and Processing Aids (CEP), C. Lambré, J. M. Barat Baviera, C. Bolognesi, P. S. Cocconcelli, R. Crebelli, D. M. Gott, K. Grob, E. Lampi, M. Mengelers, A. Mortensen, G. Rivière, I. L. Steffensen, C. Tlustos, H. Van Loveren, L. Vernis, H. Zorn, M. Andryszkiewicz, Y. Liu, S. Rainieri and A. Chesson, Safety evaluation of the food enzyme trypsin from porcine pancreas, EFSA J., 2022, 20, e07008 Search PubMed.
- C. Cuadrado, C. Arribas, A. Sanchiz, M. M. Pedrosa, P. Gamboa, D. Betancor, C. Blanco, B. Cabanillas and R. Linacero, Effects of enzymatic hydrolysis combined with pressured heating on tree nut allergenicity, Food Chem., 2024, 451, 139433 CrossRef CAS PubMed.
- S. J. Laly, A. Kumar, T. V. Sankar and S. K. Panda, In vitro characterisation of tropomyosin from flower tail shrimp, Metapenaeus dobsoni, and effect of hydrolysis on allergenicity of tropomyosin, Int. J. Food Sci. Technol., 2022, 57, 7434–7444 CrossRef CAS.
- S. Ketnawa, O. Martínez-Alvarez, S. Benjakul and S. Rawdkuen, Gelatin hydrolysates from farmed giant catfish skin using alkaline proteases and its antioxidative function of simulated gastro-intestinal digestion, Food Chem., 2016, 192, 34–42 CrossRef CAS PubMed.
- H. Li, H. Fan, Z. Wang, Q. Zhua and J. Wu, Effect of simulated gastrointestinal digestion on antioxidant and anti-inflammatory activities of bioactive peptides generated in sausages fermented with Staphylococcus simulans QB7, Food Sci. Hum. Wellness, 2024, 13, 1662–1671 CrossRef CAS.
- M. D. Kulis, S. U. Patil, E. Wambre and B. P. Vickery, Immune mechanisms of oral immunotherapy, J. Allergy Clin. Immunol., 2018, 141, 491–498 CrossRef CAS PubMed.
- S. R. Prickett, A. L. Voskamp, T. Phan, A. Dacumos-Hill, S. I. Mannering, J. M. Rolland and R. E. O'Hehir, Ara h 1 CD4+ T cell epitope-based peptides: candidates for a peanut allergy therapeutic, Clin. Exp. Allergy, 2013, 43, 684–697 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |