Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Beyond A1 milk concerns: dietary-relevant concentrations of β-casomorphin-7 show limited absorption but retain opioid-like activity in an intestinal cell model
Giorgia
Antonelli
 a,
Giovanni
Piccinini
a,
Giovanni
Piccinini
 b,
Mattia
Santoni
b,
Mattia
Santoni
 a,
Terenzio
Bertuzzi
a,
Terenzio
Bertuzzi
 c,
Davide
Risso
c,
Davide
Risso
 d,
Federico
Canzoneri
d,
Federico
Canzoneri
 d,
Roberto
Menta
d,
Nicole
Tosi
d,
Roberto
Menta
d,
Nicole
Tosi
 e,
Liliana
Milani
e,
Liliana
Milani
 b,
Cristian
Del Bo’
b,
Cristian
Del Bo’
 f,
Alessandra
Bordoni
f,
Alessandra
Bordoni
 ag,
Margherita
Dall'Asta
ag,
Margherita
Dall'Asta
 *c and
Francesca
Danesi
*c and
Francesca
Danesi
 ag
ag
aDepartment of Agricultural and Food Sciences (DISTAL), University of Bologna, Piazza Goidanich 60, 47521 Cesena, Italy
bDepartment of Biological, Geological, and Environmental Sciences (BiGeA), University of Bologna, Via Selmi 3, 40126 Bologna, Italy
cDepartment of Animal Science, Food and Nutrition, Università Cattolica del Sacro Cuore, Via Emilia Parmense 84, 29122 Piacenza, Italy. E-mail: margherita.dallasta@unicatt.it
dSoremartec Italia Srl, Ferrero Group, Piazzale Ferrero 1, 12051 Alba, Italy
eDepartment of Food and Drugs, University of Parma, Via Volturno 39, 43125 Parma, Italy
fDepartment of Food, Environmental and Nutritional Sciences (DeFENS), University of Milan, Via Celoria 2, 20133 Milan, Italy
gInterdepartmental Centre for Industrial Agri-Food Research (CIRI-AGRO), University of Bologna, Via Quinto Bucci 336, 47521 Cesena, Italy
First published on 9th March 2026
Abstract
Milk and dairy products are a significant source of bioactive peptides, including β-casomorphin-7 (BCM-7), a heptapeptide released from the enzymatic cleavage of the A1 variant of β-casein. While some studies have suggested that BCM-7 may have detrimental effects on gastrointestinal physiology, the European Food Safety Authority found no established cause-and-effect relationship between the oral intake of BCM-7 and non-communicable diseases. This study aimed to investigate the biological effects of BCM-7 at physiologically relevant concentrations, corresponding to the estimated exposure levels in the European population, on the mucus-producing HT29-MTX-E12 intestinal cell line. Cells were treated with BCM-7 concentrations ranging from 4 to 120 µM for 4 hours, and various endpoints were assessed, including cytotoxicity, mucus production and secretion, opioid activity, inflammation, and peptide translocation across the intestinal barrier. BCM-7 treatment did not significantly affect cell viability, metabolic activity, or membrane integrity, except for a slight increase in LDH release at the highest concentration tested. No changes in mucin gene transcription levels (MUC5AC, MUC2) or mucus secretion were observed. However, BCM-7 treatment increased the expression of the µ-opioid receptor compared to the untreated control, confirming its opioid-like properties. While the IL-8 gene mRNA level increased at 120 µM, protein secretion remained unchanged, suggesting limited inflammatory effect. BCM-7 translocation across the intestinal barrier was minimal and inversely concentration-dependent, with most peptides likely degraded by dipeptidyl peptidase-4. These findings demonstrate that physiologically relevant BCM-7 concentrations exhibit opioid-like properties but have limited impact on mucus production, inflammation, and translocation of the intact peptide across the epithelial barrier in this in vitro intestinal model. The results provide mechanistic insights into BCM-7 effects while highlighting the need for further research considering individual dietary patterns and complex milk component interactions.
1 Introduction
Milk is a well-established source of significant nutritional value, providing energy, high-quality proteins (approximately 32 g L−1), calcium, and essential vitamins. In 1880, the pioneering work of the Swedish scientist Olav Hammarsten revealed that milk proteins can be categorized into two distinct groups: caseins and whey proteins.1 Caseins account for 80% of the total protein content and can be further classified into α-, β-, and κ-casein fractions. Ancient genetic mutations in the bovine β-casein protein have led to the emergence of distinct variants. Approximately 5000 years ago, a point mutation occurred, resulting in the substitution of the proline amino acid at position 67 with histidine (His67). Cattle expressing this mutated form of β-casein are referred to as A1 cows, while those with the original proline variant are known as A2 cows. Notably, the A1 protein variant is commonly found in milk from crossbred and European cattle breeds, while the A2 variant is predominantly found in milk from indigenous cattle and buffalo breeds native to the Indian subcontinent.2Extensive research has demonstrated that the digestion of ‘A1 milk’, containing the His67 variant of the β-casein protein, by human digestive and microbial enzymes during the digestive process and cheese ripening can lead to the release of a specific heptapeptide, β-casomorphin-7 (BCM-7; Tyr-Pro-Phe-Pro-Gly-Pro-Ile). In contrast, the presence of proline at position 67 in the ‘A2 milk’ β-casein variant generates resistance to enzymatic cleavage at this position, reducing the amount of BCM-7 formed but not fully preventing its release.3 BCM-7 belongs to the class of opioid-like peptides termed casomorphins.4 This particular casomorphin has been extensively studied due to its potential health implications. Some studies in animals and humans have suggested that A1 β-casein consumption, with BCM-7 proposed as a key mediator, may have a detrimental impact on gastrointestinal (GI) physiology, including delayed intestinal transit, increased inflammatory markers, and altered mucus production.5–7 The proposed mechanism underlying these effects is attributed to BCM-7's ability to interact with opioid receptors, primarily the μ-opioid receptors (MORs), acting as an agonist and exerting opioid-like actions within the GI tract.8
Over the years, this hypothesis regarding the detrimental effects of BCM-7 has been investigated using in vitro and animal models, often employing supraphysiological or pharmacological concentrations. The European Food Safety Authority (EFSA) has evaluated studies on the potential association between β-casomorphins and related peptides and an increased risk of non-communicable diseases. However, based on the available evidence, no cause-and-effect relationship was established.9 Despite the lack of conclusive scientific evidence, many dairy industries have proactively started producing milk and dairy products (such as cheese, yogurt, and cream) claimed to be free from the β-casein A1 variant over the years.10,11 However, further research is warranted to evaluate the potential impact of BCM-7 on human physiology, considering the actual exposure levels of this peptide in the general population.
Our recent study was the first to estimate BCM-7 exposure levels for the European population across different consumption patterns.12 By combining BCM-7 peptide release data from in vitro simulated GI digestion of A1-containing milk and dairy products (fermented milk, yogurt, cheese, and milk powder) with European consumption data, we determined consistent exposure ranges across population groups: children (200–2357 μg day−1), adolescents (163–2594 μg day−1), and adults (132–2541 μg day−1).12 Considering BCM-7's molecular weight (790.9 g mol−1) and estimated duodenal volumes (30–54 mL), these daily exposure levels correspond to approximate duodenal luminal concentrations of ∼3–99 µM; we therefore selected 4, 40, 80 and 120 µM for in vitro testing, with 120 µM included as an upper bound to ensure coverage of the high-exposure scenario (Table S1). Building upon these exposure estimates, the present study investigates the biological effects of BCM-7 at physiologically relevant concentrations corresponding to the actual European dietary intake levels through milk and dairy consumption. Specifically, we focus on the impact of BCM-7 on intestinal mucus-producing goblet cells to elucidate the physiological relevance of this peptide at the GI level. The study evaluates cytotoxicity, bioactivity-related outcomes (mucus production and secretion, opioid activity, and inflammation), as well as the translocation of BCM-7 across the intestinal barrier. The findings from this research will contribute to a better understanding of the potential health implications associated with BCM-7 exposure at realistic dietary levels.
2 Materials and methods
2.1 Materials
The HT29-MTX-E12 cell line was kindly provided by Prof. Giromini (University of Milan, Italy). Dulbecco's Modified Eagle's medium (DMEM) with high glucose and no glutamine, MEM non-essential amino acid solution, and GlutaMAX supplement were purchased from Gibco (Waltham, MA, USA). Fetal Bovine Serum (FBS), penicillin–streptomycin, trypsin/EDTA, bovine serum albumin (BSA), and mucin from porcine stomach (type II) were obtained from Sigma-Aldrich (St Louis, MO, USA) or Merck (Darmstadt, Germany). β-Casomorphin-7 (BCM-7) was acquired from Bachem (Bubendorf, Switzerland). Transwells with 0.4 µm pore polyester membrane inserts were obtained from Corning (NY, USA).2.2 Cell culture growth and treatment conditions
HT29-MTX-E12 cells were cultured in DMEM containing 4.5 mg L−1 glucose, supplemented with 10% FBS (v/v), 1% penicillin–streptomycin (v/v), and 1% MEM non-essential amino acid solution (v/v). Cells were maintained under standard conditions (37 °C, 5% CO2), and the growth medium was changed every other day. Cells between passages 53 and 65 were used for experiments. Before conducting experiments, the absence of mycoplasma was routinely tested using the MycoBlue Mycoplasma Detector (Vazyme; Nanjing, China) and cellular senescence was determined using the Cellular Senescence Assay Kit (Cell Biolabs; San Diego, CA, USA).Undifferentiated cells were used for the PrestoBlue assay (1 × 104 cells per well in a 96-well plate), Trypan Blue exclusion assay, LDH assay (5 × 105 cells per well in a 96-well plate), and MOR quantification (3 × 105 cells per well in a 96-well plate). For cytokine quantification, gene transcription analysis, and mucus quantification, cells were seeded on polycarbonate inserts (12-well) at a density of 5 × 105 cells per insert and grown on an orbital shaker (Infors HT; Bottmingen, Switzerland) set to 55 rpm, for 21 days post-confluence. The development and integrity of the cellular monolayer over time were monitored by measuring the trans-epithelial electrical resistance (TEER) as previously described.13 TEER values were recorded every other day for 21 days post-confluence, following the replacement of the culture medium. The TEER of an empty insert was subtracted from each value.
In experiments assessing cell viability, cytotoxicity, and membrane integrity, cells were treated with 4, 40, 80, and 120 μM BCM-7 in FBS- and phenol red-free DMEM for 4 hours. The choice of BCM-7 concentration (Table S1) was guided by estimated duodenal BCM-7 levels in three European population groups (children, adolescents, and adults) corresponding to low, medium, and high milk and dairy consumption, respectively,12 assuming average duodenal volumes of 30 mL for children and 54 mL for adults.14,15 In subsequent experiments evaluating the bioactivity of the peptide, only the three highest concentrations were used.
2.3 Assessment of cytotoxicity
Cytotoxicity assessment was performed using three complementary assays: cell viability (Trypan Blue exclusion assay), metabolic activity (PrestoBlue assay), and membrane integrity (LDH release).16For the Trypan Blue exclusion assay, cells were washed twice with DPBS and then detached using ReagentPack Subculture Reagents (Lonza; Basel, Switzerland) according to the manufacturer's protocol. A 10 µL aliquot of the cell suspension was mixed with an equal volume of Trypan Blue (Bio-Rad Laboratories; Hercules, CA, USA), and 10 µL of the cell–Trypan Blue mixture was loaded into a dual-chamber slide. Cell counts were performed using a TC20 Automated Cell Counter (Bio-Rad), and the results represent the percentage of viable cells in the treated samples normalized to control values.
For the PrestoBlue assay, cells were washed twice with DPBS and incubated with PrestoBlue reagent (Invitrogen; Waltham, MA, USA) diluted in phenol red-free DMEM (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) for 30 minutes. The reaction was terminated by adding 3% sodium dodecyl sulfate (SDS), and the fluorescence of samples and blank (PrestoBlue reagent plus 3% SDS) was measured with an Infinite F200 microplate reader (Tecan; Männedorf, Switzerland) at an excitation wavelength (λ) of 560 nm and an emission λ of 590 nm. Results represent the percentage of metabolic activity in BCM-7 treated samples normalized to control values after background correction.
10) for 30 minutes. The reaction was terminated by adding 3% sodium dodecyl sulfate (SDS), and the fluorescence of samples and blank (PrestoBlue reagent plus 3% SDS) was measured with an Infinite F200 microplate reader (Tecan; Männedorf, Switzerland) at an excitation wavelength (λ) of 560 nm and an emission λ of 590 nm. Results represent the percentage of metabolic activity in BCM-7 treated samples normalized to control values after background correction.
Cell membrane integrity was assessed by measuring lactate dehydrogenase (LDH) release from the cytoplasm into the culture medium, as LDH release is a reliable indicator of membrane damage and compromised cellular integrity.17 The assay was conducted using the CyQUANT LDH Cytotoxicity Assay Kit (Invitrogen) following the manufacturer's protocol. Absorbance was measured at 490 nm and 680 nm (background) using an Infinite M200 microplate reader (Tecan). The LDH release was calculated as a percentage of the maximum LDH activity, which was determined by lysing control cells. The percentage of LDH release in BCM-7 treated cells was then normalized to the untreated control group and calculated as: LDH release (%) = [(BCM-7 LDH activity/maximum LDH activity)/(control LDH activity/maximum LDH activity)] × 100.
2.4 Mucus production assessment
Mucus production by HT29-MTX-E12 cells was assessed using two complementary methods: periodic acid-Schiff assay for quantitative analysis of secreted mucins in the cell culture media and alcian blue staining for qualitative visualization of acidic mucopolysaccharides in the cell-associated mucus layer.2.5 Quantitative PCR (qPCR) analysis of mRNA levels
Following the treatment described in Section 2.2, the cell monolayer was scraped, washed with DPBS, and stored at −80 °C until further processing. For cell lysis, 300 µL of TriReagent (Zymo Research; Irvine, CA, USA) was added to the frozen cell pellet, which was then subjected to three sonication cycles (30 seconds each) in a cold bath using a Bioruptor sonicator (Diagenode; Denville, NJ, USA). After sonication, the lysate was centrifuged at 3000g for 5 minutes at room temperature. Total RNA was extracted using the Direct-zol RNA MiniPrep kit (Zymo Research; Irvine, CA, USA) according to the manufacturer's instructions. RNA quantity and quality were assessed using a NanoDrop ND-2000 spectrophotometer (Thermo Fisher Scientific).For cDNA synthesis, 2 µg of total RNA was reverse-transcribed using the High-Capacity RNA-to-cDNA Kit (Applied Biosystems; Waltham, MA, USA). Quantitative PCR was performed using the CFX Connect Real-Time PCR Detection System (Bio-Rad Laboratories) following the TaqMan Fast Advanced Master Mix protocol (Applied Biosystems), as previously described.13 The target genes were selected to assess specific intestinal functions, including those encoding mucin glycoproteins [MUC2 (mucin-2) and MUC5AC (mucin-5AC)], intestinal brush border enzyme [DPP4 (dipeptidyl peptidase-4)], and proinflammatory cytokine [IL-8 (interleukin-8)]. All TaqMan probe sets used in this study were purchased from Applied Biosystems (Table S2). Relative gene transcription was calculated using the ΔCt method,22 with β-actin (ACTB), glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and peptidylprolyl isomerase A (PPIA) as the reference genes. Data processing was performed using Bio-Rad CFX Maestro 2.3 version 5.3.
2.6 Quantification of cytokines
Cytokine levels in the cell culture medium were quantified using the Bio-Plex Pro Human Cytokine 8-Plex Panel kit (Bio-Rad) following the manufacturer's protocol. In brief, cell culture supernatants were supplemented with 0.5% (w/v) BSA. Magnetic beads pre-coupled with cytokine-specific capture antibodies (50 µL) were added to each well and washed twice with the wash buffer provided in the kit using a Bio-Plex Handheld Magnetic Washer (Bio-Rad). Fifty µL of samples, standards, and blank were added to each well and the covered and sealed plate was incubated on a shaker (850 ± 50 rpm) for 30 minutes. After incubation, the plate was washed three times, and 25 µL of detection antibody was added to each well. The plate was incubated for an additional 30 minutes, followed by another washing step as previously done. Subsequently, 50 µL of streptavidin–phycoerythrin conjugate was added to each well and incubated for 10 minutes. The plate was then washed three times as previously done, and 125 µL of assay buffer was added to each well to resuspend beads. Finally, the plate was read using a Bio-Plex MAGPIX Multiplex Reader (Bio-Rad) with Bio-Plex Manager software (v. 6.2) for data acquisition and analysis. Cytokine concentrations were calculated based on the standard curves generated using the provided cytokine standards. The limit of detection (LOD) of each analyte in the assay is presented in Table S3. The cytokines measured in the immunoassay included interleukin-2 (IL-2), IL-4, IL-6, IL-8, IL-10, tumor necrosis factor α (TNFα), interferon γ (IFN-γ), and granulocyte-macrophage colony-stimulating factor (GM-CSF).2.7 Assessment of µ-opioid receptor (MOR) expression by cell-based ELISA
The expression of MOR was evaluated using a cell-based ELISA kit (Boster; Pleasanton, CA, USA), targeting the OPRM1-encoded protein (UniProt ID: P35372), according to the manufacturer's protocol. Following the treatment, cells were fixed with 100 µL of 4% formaldehyde for 20 minutes. The fixing solution was then removed, and cells were incubated with 100 µL of quenching buffer for 20 minutes. The plate was washed three times, and the wells were blocked with 200 µL of blocking buffer. After washing, the wells were incubated overnight with primary antibodies specific for MOR (rabbit polyclonal) and housekeeping protein glyceraldehyde 3-phosphate dehydrogenase (GAPDH, mouse monoclonal) included in the kit. The following day, the plate was washed and HRP-conjugated secondary antibodies (anti-rabbit IgG for MOR and anti-mouse IgG for GAPDH, included in the kit) were added and incubated for 1.5 hours. The colorimetric reaction was developed by adding 50 µL of substrate solution and stopped with 50 µL of stop solution. The absorbance was measured at a wavelength of 450 nm. Subsequently, the wells were washed three times, and cells were stained with crystal violet solution for 30 minutes. After rinsing the wells with tap water, 100 µL of SDS solution was added, and the absorbance was measured at λ = 595 nm. The absorbance values for the opioid receptor and GAPDH conditions were normalized to the absorbance of the cell nuclei (crystal violet). The minimum detectable expression of MOR was 5000 cells per well according to the manufacturer's protocol. Data are expressed as the ratio between the normalized absorbance of the opioid receptor condition and the GAPDH condition.2.8 Quantification of BCM-7 translocation and recovery
The concentrations of BCM-7 in the apical (AP; donor) and basolateral (BA; receiver) chambers were quantified using liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS). Samples were diluted (for AP samples: 0.100 mL sample + 0.900 mL mobile phase; for BA samples: 0.200 mL sample + 0.800 mL mobile phase) and filtered through a 0.45 µm filter. The LC-MS/MS system consisted of a LC 1.4 Surveyor pump (Thermo Fisher Scientific), a PAL 1.3.1 sampling system (CTC Analytics AG; Zwingen, Switzerland), and a Quantum Discovery Max triple quadrupole mass spectrometer. The system was controlled by Excalibur 1.4 software (Thermo Fisher Scientific).Chromatographic separation was achieved using an X Select HSS T3 column (2.5 μm particle size, 100 × 2.1 mm i.d.; Waters Corporation, MA, USA) with a gradient elution of H2O–CH3CN (both acidified with 0.2% formic acid). The linear gradient was programmed from 25% to 65% CH3CN within 4 min, followed by an isocratic period of 4 min and column conditioning for 6 min. The flow rate was set at 0.2 mL min−1. Ionization was performed in positive mode using an electrospray ionization (ESI) interface. The fragment ions monitored were 229, 286, 383, and 530 m/z (precursor ion M+ 791 m/z). The limit of detection (LOD) and the limit of quantification (LOQ) were determined to be 0.001 and 0.003 µmol L−1, respectively.
The percentage of translocation was calculated according to the following equation:23
| Translocation (%) = (CBL/C0) × 100 |
To compare the amount of the test compound detected at the end of the experiment with the initial nominal concentration, the peptide recovery percentage was also calculated, as described by Almeida et al.:24
| Recovery (%) = [(CAP + CBL)/C0] × 100 |
2.9 Statistical analysis
Statistical analysis was conducted using GraphPad Prism version 10.1.1 (GraphPad Software Inc.; La Jolla, CA, USA). Data are presented as means ± standard deviation (SD) of three independent experiments. Statistical significance among treatments was determined by one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test. A p-value of less than 0.05 was considered statistically significant.3 Results
3.1 Evaluation of monolayer integrity by trans-epithelial electrical resistance (TEER) measurement
The integrity of the HT29-MTX-E12 cell monolayers used in the experiments was assessed by measuring the trans-epithelial electrical resistance (TEER) (Fig. S1). Following Schimpel et al.25 and Dolan et al.,26 only monolayers exhibiting initial TEER values post-21 days of culture exceeding 500 Ω cm2 were selected for the experiments, ensuring the presence of a well-established and intact epithelial barrier.3.2 Cell viability, metabolic activity, and membrane integrity assessment
The potential detrimental effects of BCM-7 on HT29-MTX-E12 cells were evaluated by assessing cell viability (Trypan Blue exclusion assay), metabolic activity (PrestoBlue assay), and plasma membrane integrity (lactate dehydrogenase [LDH] release assay). As shown in Table 1, exposure to BCM-7 at the tested concentrations did not significantly affect cell viability or metabolic activity. However, treatment with 120 µM BCM-7 for 4 hours resulted in a significant increase in LDH release into the growth media compared to the lowest concentrations tested (4 and 40 µM).| BCM-7 | ||||
|---|---|---|---|---|
| 4 μM | 40 μM | 80 μM | 120 μM | |
| Data are presented as mean ± standard deviation (SD) of three independent experiments. Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test. Different letters within a row indicate significant differences among the treatment groups (p < 0.05). | ||||
| Cell viability (%) | 99.23 ± 3.12a | 102.40 ± 5.27a | 100.85 ± 4.32a | 99.85 ± 7.63a |
| Metabolic activity (%) | 104.99 ± 0.96a | 100.05 ± 2.64a | 100.73 ± 1.25a | 98.07 ± 3.23a |
| Plasma membrane integrity (%) | 91.79 ± 0.48b | 92.51 ± 0.75b | 95.26 ± 2.85a,b | 98.79 ± 1.69a |
3.3 Evaluation of mucin gene transcription level and mucus secretion
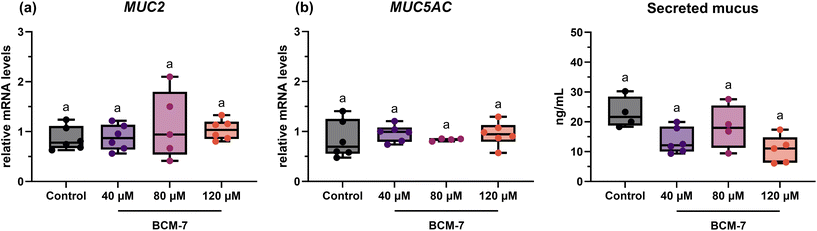
Several in vitro and in vivo studies have reported the ability of BCM-7 to increase mucin production.27–29 In this study, we investigated the effect of BCM-7 on the mRNA levels of MUC5AC and MUC2 genes, which encode for the major secreted mucins in the GI tract. Fig. 1 (panels a and b) presents the relative transcription levels of these genes in HT29-MTX-E12 cells treated with 40, 80, and 120 µM BCM-7 compared to the untreated control. Our results demonstrated no significant differences in the mRNA abundance of either MUC5AC or MUC2 transcripts between cells treated with the various BCM-7 concentrations and the untreated control.The effect of BCM-7 on mucus production and secretion by HT29-MTX-E12 cells was evaluated using both quantitative and qualitative methods. Quantitative analysis of secreted mucins using the periodic acid-Schiff assay revealed no significant differences in mucus concentrations across treatment conditions (Fig. 1, panel c). Treatment with BCM-7 at various concentrations (40, 80, and 120 μM) for 4 hours did not significantly alter the amount of secreted mucus compared to the untreated control, with values ranging from approximately 10 to 20 ng mL−1 across all groups.
Complementing these findings, HT29-MTX-E12 cells stained with alcian blue showed no discernible differences between the control and BCM-7-treated groups at various concentrations (Fig. 2). The staining intensity and distribution of mucus appeared similar across all experimental conditions. This lack of observable changes is consistent with the quantitative data obtained from the periodic acid-Schiff assay.
3.4 Quantification of µ-opioid receptor protein
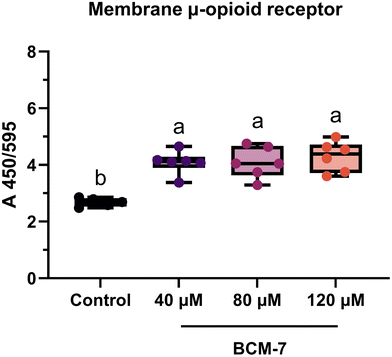
BCM-7 has been reported to exhibit opioid activity through the activation of MOR.3,30 In our study, we investigated the effect of BCM-7 treatment on the expression of MOR in HT29-MTX-E12 cells. As shown in Fig. 3, BCM-7 treatment (40, 80, and 120 µM) significantly increased the expression of MOR compared to the untreated control. To assess the lower limit of activity, we also tested the lowest dose (4 µM; data not shown), which resulted in activation of the same magnitude as the highest doses.3.5 Evaluation of cytokine gene transcription levels and protein secretion
To assess the potential inflammatory effect of BCM-7, we evaluated the mRNA levels of interleukin-8 (IL-8) and the secretion of various cytokines in the growth media of HT29-MTX-E12 cells. As shown in Table 2, treatment with 120 µM BCM-7 for 4 hours significantly increased the IL-8 gene transcription level compared to the untreated control. However, this trend was not reflected in the secretion of IL-8 in the growth media, as no significant differences were observed between the BCM-7 treatments and the untreated control (Table 2).| Control | BCM-7 | |||
|---|---|---|---|---|
| 40 μM | 80 μM | 120 μM | ||
| Data are presented as mean ± standard deviation (SD) of three independent experiments. Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test. Different letters within a row indicate significant differences among the treatment groups (p < 0.05). | ||||
| IL-8 gene transcription level | 1.015 ± 0.160b | 1.004 ± 0.327b | 0.806 ± 0.163b | 1.476 ± 0.098a |
| IL-8 protein secretion (pg mL−1) | 0.027 ± 0.006a | 0.027 ± 0.012a | 0.023 ± 0.006a | 0.023 ± 0.006a |
IL-8 was quantified in both compartments, apical and basolateral, but was primarily detected in the apical compartment, consistent with polarized epithelial secretion toward the luminal surface. Additionally, we evaluated the secretion of other cytokines, including IL-2, IL-4, IL-6, IL-10, TNFα, IFN-γ, and GM-CSF. However, their concentrations were below the lower limit of detection of the assay and thus could not be quantified.
3.6 Evaluation of the dipeptidylpeptidase-4 (DPP4) gene transcription level
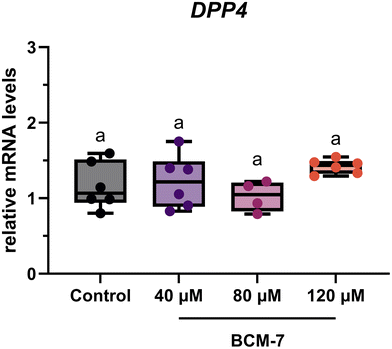
Dipeptidyl peptidase-4 (DPP4), a brush border enzyme encoded by the DPP4 gene, has been identified as the primary enzyme responsible for the hydrolysis of BCM-7 in the small intestine. To investigate whether BCM-7 exposure influences the transcription level of DPP4 gene, we assessed the mRNA level in HT29-MTX-E12 cells treated with various concentrations of BCM-7 (40, 80, and 120 µM). As illustrated in Fig. 4, the DPP4 gene transcription level remained unaffected by BCM-7 treatment at any of the tested concentrations after a 4-hour exposure period.3.7 Evaluation of BCM-7 translocation and recovery
The concentrations of BCM-7 in the apical (donor) and basolateral (receiver) chambers after 4 hours of treatment are presented in Table 3. The translocation of BCM-7 to the basolateral chamber represents a measure of potential intestinal absorption following oral exposure, as polarized intestinal cells grown on inserts mimic epithelial barriers where basolateral transport indicates systemic uptake.24 The results demonstrate that the majority of BCM-7 was removed from the apical chamber after 4 hours of treatment, with a decrease of approximately 98% for all concentrations tested (40 µM: 98.03%; 80 µM: 98.29%, and 120 µM: 98.05%). The lack of significant differences in the percentage decrease among the treatment groups (p = 0.7106) suggests that the translocation of BCM-7 from the apical chamber was not concentration-dependent within the range tested.| Control | BCM-7 | |||
|---|---|---|---|---|
| 40 μM | 80 μM | 120 μM | ||
| Data are presented as mean ± standard deviation (SD) of three independent experiments. Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test. Different letters within a row indicate significant differences among the treatment groups (p < 0.05). | ||||
| Apical (donor) chamber (μM) | <0.001 | 0.776 ± 0.085b | 1.366 ± 0.373b | 2.040 ± 0.487a |
| Basolateral (receiver) chamber (μM) | <0.001 | 0.050 ± 0.007a | 0.041 ± 0.009a,b | 0.035 ± 0.003b |
| Translocation (%) | — | 0.125 ± 00.018a | 0.051 ± 00.011b | 0.029 ± 00.003c |
| Recovery (%) | — | 1.939 ± 00.213a | 1.708 ± 00.466a | 1.955 ± 00.723a |
Interestingly, the concentration remaining in the AP chamber after treatment with 120 µM BCM-7 was significantly higher than that of the other concentrations. In contrast, the concentration of BCM-7 in the BL chamber showed an opposite trend, with lower concentrations observed at the higher treatment level. Similar recovery values across treatment conditions support the comparability of translocation kinetics between doses (Table 3); however, the low absolute recovery suggests that the majority of apically applied BCM-7 is likely subject to degradation, cellular uptake, or adsorption during the 4-hour incubation period, rather than remaining intact or crossing the epithelial barrier.
4 Discussion
BCM-7 is released during digestion of milk and dairy products after cleavage by proteases in the small intestine, where it exerts its biological activity. One of the most studied biological effects of BCM-7 is its mucus-secreting promoting effect. The intestinal mucus layer plays a pivotal role in maintaining intestinal homeostasis, as it acts as a physical and immunological barrier against food particles and bacteria and lubricates the luminal content.21,31 For this reason, we investigated the effect of BCM-7 using the human colon adenocarcinoma cell line HT29-MTX-E12, an established model for studying the effect of bioactive molecules on mucus production. When grown under appropriate conditions, this cell line exhibits mucus-secreting characteristics, making it suitable for this purpose. The HT29-MTX-E12 cell line mainly expresses the mucin-like glycoprotein MUC5AC, while MUC2 is produced in lower amounts.32 Furthermore, in order to promote the mucus phenotype, we have grown cells under mechanical stimulation.33 Unpublished data from our laboratories have demonstrated that this growth condition upregulates the transcription of mucin-encoding genes, compared to a static condition, enhancing the phenotype of the goblet cells.The first part of the study was designed to evaluate the effect of different doses of BCM-7 (4, 40, 80, and 120 µM) on cell viability, cytotoxicity, and membrane integrity on 21-day differentiated HT29-MTX-E12 cells treated for 4 hours. These concentrations were chosen based on the estimated daily consumption of β-casomorphin-7 in European children, teenagers, and adults.12 Data on dairy consumption were extrapolated from the European Food and Safety Authority database – FoodEx 2 system – and integrated with a previously established dataset of BCM-7 release from milk and dairy products. The concentration of 4 µM represents the 5th percentile of exposure (low exposure) in children, adolescents, and adults; 40 µM represents the 50th percentile of exposure (average exposure) in all population categories; 80 µM was used to represent the high-exposure scenario for adolescents/adults, whereas 120 µM was included to cover the upper-end estimate for children (Table S1).12 The 4 hours exposure time was chosen based on the physiological small intestine transit, which spans from 2 to 6 hours.34,35 While cell viability and metabolic activity remained unaffected across all concentrations tested, treatment with 120 µM BCM-7 resulted in a slightly increased LDH release compared to lower concentrations (4 and 40 µM), suggesting some membrane stress at the highest exposure levels. Our results showed that treatment with BCM-7 did not upregulate MUC5AC and MUC2 gene transcription levels. This was also confirmed by alcian blue histochemical staining. In agreement with our results, Zoghbi et al.27 showed no upregulation of MUC5AC in HT29-MTX cells after treatment with 100 µM BCM-7 for 4 and 8 hours. As demonstrated in their study, the upregulation of MUC5AC started between 8 and 24 hours; however, this is not physiologically relevant as the small intestine transit is shorter than 8 hours. Our results about mucus dynamics collectively suggest that short-term exposure to physiologically relevant concentrations of BCM-7 does not substantially impact mucus production or secretion in this intestinal epithelial cell model. The consistency between the quantitative assays and qualitative staining further supports this conclusion.
The second part of the study aimed to investigate the biological effects of BCM-7 on differentiated HT29-MTX-E12 cells and on its translocation across the intestinal barrier. The cells were differentiated for 21 days and then treated for 4 hours with the three highest doses of BCM-7 used in the first part of the study: 40 µM, the average exposure level for all European population groups considered; 80 µM, the high exposure level for adolescents and adults; 120 µM, the high exposure level for children.
Several studies have suggested that the mucus-secreting effect is promoted by the activation of the μ-opioid receptor by BCM-7. The opioid system consists of three transmembrane G-protein coupled receptors – μ- (MOR), κ- (KOR), and δ-opioid (DOR) receptors – which mediate motility and secretion in the gut. At the GI level, opioid agonists promote the absorption of fluids and electrolytes and decrease gastric emptying, motility, and intestinal propulsion.36 The presence of μ-opioid receptors and their localization on the plasma membrane in the HT29 cell line has been demonstrated by Nylund et al.37 and the activation of the MOR by BCM-7 as an exogenous agonist has been shown by several studies using in vitro and animal models. BCM-7 contains a tyrosine in its N-terminal portion and a phenylalanine as the third amino acid. These two amino acids confer a motif structure that fits well into the binding pocket of the opioid receptor.38 Trompette et al.28 demonstrated that administration of 120 μM BCM-7 for 30 minutes to isolated perfused rat jejunum increased mucus discharge by 500% compared to the control, which was inhibited by the opioid antagonist naloxone. Although we were not able to replicate mucus-inducing effects in our model at both transcriptional and secreted protein levels, our results confirmed the interaction of BCM-7 with the μ-opioid receptor, showing a dose-independent activation of MOR.
Studies investigating the correlation between BCM-7 and inflammation have yielded contradictory results. Fiedorowicz et al.39 reported that infant formulas containing 0.02–6 μM BCM-7 induced a significant increase in IL-8 release in a Caco-2 cell model. In contrast, our study found that the increase in gene transcript level induced by 120 μM BCM-7 did not translate to higher protein secretion in the culture medium. This discrepancy is unlikely to be due to the different cell lines used, as previous studies have shown that Caco-2 cells have lower basal IL-8 expression than HT29 cells.40 However, it is important to note that our HT29-MTX-E12 cell model differs from the original HT29 clone and may have a distinct and improved cytokine expression profile. Furthermore, proliferating Caco-2 cells, such as those used in Fiedorowicz's study,39 are more susceptible to inflammatory triggers than differentiated cells,41 which could contribute to the observed differences in IL-8 secretion. At a mechanistic level, IL-8 output is strongly regulated post-transcriptionally (e.g., via p38 MAPK-dependent mRNA stabilization),42 which may explain why IL-8 mRNA changes did not translate into increased secreted protein in our conditions. The potential inflammatory effect of BCM-7 was investigated by the same authors in an ex vivo model.43 Peripheral blood mononuclear cells extracted from healthy patients were treated with micro-, nano-, and picomolar concentrations of BCM-7 for 7 days. Nanomolar concentrations induced a significant increase in IL-4, and, for IL-8, picomolar concentrations also had an effect. Although this study sheds light on the biological effect of BCM-7 on human blood cells, it must be noted that the chosen incubation duration is not physiological, as peptidases that degrade peptides are present in blood serum.44,45 Barnett et al.6 compared the GI effects of A1 versus A2 β-casein consumption in Wistar rats fed experimental diets for 36–84 hours. Colonic myeloperoxidase activity was significantly higher in the A1 group compared to the A2 group, and this increase was attenuated by co-administration of naloxone, supporting the involvement of opioid-mediated pathways and providing physiological evidence consistent with a role for BCM-7 as a mediator of A1 β-casein effects. Human double-blind controlled trials on the effect of BCM-7 from A1A1 or A1A2 milk are very limited and do not allow general conclusions on the actual biological effect.46
In the small intestine, BCM-7 is hydrolyzed by DPP4, a proline-specific peptidase that cleaves X-P dipeptides from the N-terminus of polypeptides.47 To confirm the reliability of our model, we evaluated the gene transcription level of DPP4. Our results show a dramatic decrease in BCM-7 concentration in the apical chamber at the end of the 4-hour treatment with increasing concentrations of BCM-7, which was not reflected by a corresponding increase in the basolateral chamber. Although we did not evaluate the intracellular content of BCM-7, it is possible to speculate that the remaining portion of the heptapeptide was hydrolyzed by DPP4 into di- and tripeptides, which were then taken up by peptide transporter 1 (PepT1).48,49 In a study by Iwan et al.,50 a 99.4% decrease in the apical concentration of BCM-7 was found after 60 minutes of incubation in a Caco-2 model. The concentration decreased by only 33% after treatment with diprotin A (Ile-Pro-Ile), a DPP4 inhibitor, demonstrating the role of DPP4 in BCM-7 degradation. Osborne et al.51 identified metabolites resulting from the degradation of BCM-7, which were found to be consistent with the action of DPP4. The inverse relationship between BCM-7 concentration and translocation percentage observed in our study suggests that the transport of BCM-7 across the HT29-MTX-E12 cell monolayer may be saturable or subject to concentration-dependent limitations. In the Osborne study,51 the authors suggested that the passage of intact BCM-7 through the monolayer occurs via the sodium-coupled oligopeptide transporter 2 (SOPT2), which has been shown to be the transport pathway for the synthetic opioid pentapeptide DADLE.52 Another study correlated a decrease in TEER values following treatment of Caco-2 cells with BCM-7 with an increase in the basolateral BCM-7 concentration, suggesting a paracellular transport pathway.53 Further studies are needed to elucidate the main transport pathway of the BCM-7 peptide. It is important to note that the cell line used in the present study, HT29-MTX-E12, is a mucus-secreting cell line, unlike Caco-2 cells. The presence of mucus might have influenced the translocation of BCM-7 through the monolayer compared to the data present in the literature obtained using Caco-2 models. Additionally, we must acknowledge as a minor limitation that the monoculture models lack the complexity of the intact intestinal environment, including immune cell interactions,21 that may modulate BCM-7 effects. However, the consistent recovery percentages of the peptide across all tested concentrations obtained in this study indicate that the observed differences in the transport kinetics of BCM-7 are more likely due to biological factors, such as saturable transport mechanisms, rather than the presence of mucus affecting the cell monolayer's permeability. It should be noted that our study focused on acute 4-hour BCM-7 exposure, reflecting the physiological small intestine transit time.34,35 While this approach is appropriate for assessing immediate cellular responses, future studies examining repeated or prolonged exposure protocols could provide complementary insights into potential cumulative effects of regular dairy consumption.
5 Conclusions
While some studies in the literature suggest a possible link between A1 milk consumption and GI disorders,54 the exact mechanisms remain unclear. Our findings offer mechanistic insights into the role of BCM-7 in the GI tract in vitro. While in vitro cell models have inherent limitations, our differentiated HT29-MTX-E12 system provides a controlled environment to assess direct BCM-7 effects on intestinal epithelial cells. Confirmation in in vivo studies would strengthen these findings and address factors such as immune cell interactions and microbiome influences. It is important to note that BCM-7 can also be formed during milk processing, regardless of the β-casein variant,55,56 and that milk contains other peptides with opioid antagonist bioactivities.57 Therefore, the health implications of BCM-7 exposure should be considered within the broader context of milk's compositional complexity, individual dietary patterns, and peptide bioavailability. Until more conclusive evidence emerges, claims regarding the healthiness of A2 over A1 milk should be approached with caution.Author contributions
Giorgia Antonelli: data curation, formal analysis, investigation, writing – original draft, and writing – review & editing; Terenzio Bertuzzi: investigation; Alessandra Bordoni: writing – review & editing; Federico Canzoneri: project administration; writing – review & editing; Margherita Dall'Asta: conceptualization, funding acquisition, investigation, resources, and writing – review & editing; Francesca Danesi: conceptualization, data curation, formal analysis, funding acquisition, investigation, resources, writing – original draft, and writing – review & editing; Cristian Del Bo’: investigation, resources, and writing – review & editing; Roberto Menta: writing – review & editing; Liliana Milani: investigation and resources; Giovanni Piccinini: investigation; Davide Risso: conceptualization; Mattia Santoni: investigation and writing – review & editing; Nicole Tosi: investigation and writing – review & editing. All authors have read and approved the manuscript.Conflicts of interest
Federico Canzoneri is employed by Soremartec Italia Srl (Alba, Italy). At the time of conceptualization, both Roberto Menta and Davide Risso were employed by Soremartec Italia Srl. Roberto Menta is now retired, while Davide Risso is currently employed by Tate & Lyle Italy SpA. The other authors declare no competing interests.Data availability
The raw data supporting the findings of this article are available from the corresponding author on reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo04465k.
Acknowledgements
The authors acknowledge the funding from Soremartec Italia Srl (Alba, Italy) during the period September 2021–April 2023. This research was conducted independently, and the conclusions do not necessarily reflect the positions of the funding organization.References
- P. F. Fox, T. Uniacke-Lowe, P. L. H. McSweeney and J. A. O'Mahony, in Dairy chemistry and biochemistry, ed. P. F. Fox, T. Uniacke-Lowe, P. L. H. McSweeney and J. A. O'Mahony, Springer International Publishing, Cham, Switzerland, 2015, pp. 145–239 Search PubMed.
- C. Paramasivam, Bovine milk: A1 and A2 beta casein milk proteins and their impact on human health: a review, Agric. Rev., 2021, 43, 374–378 Search PubMed.
- Y. Jinsmaa and M. Yoshikawa, Enzymatic release of neocasomorphin and β-casomorphin from bovine β-casein, Peptides, 1999, 20, 957–962 CrossRef CAS PubMed.
- I. De Noni and S. Cattaneo, Occurrence of β-casomorphins 5 and 7 in commercial dairy products and in their digests following in vitro simulated gastro-intestinal digestion, Food Chem., 2010, 119, 560–566 Search PubMed.
- S. Jianqin, X. Leiming, X. Lu, G. W. Yelland, J. Ni and A. J. Clarke, Effects of milk containing only A2 beta casein versus milk containing both A1 and A2 beta casein proteins on gastrointestinal physiology, symptoms of discomfort, and cognitive behavior of people with self-reported intolerance to traditional cows’ milk, Nutr. J., 2016, 15, 35 CrossRef PubMed.
- M. P. Barnett, W. C. McNabb, N. C. Roy, K. B. Woodford and A. J. Clarke, Dietary A1 β-casein affects gastrointestinal transit time, dipeptidyl peptidase-4 activity, and inflammatory status relative to A2 β-casein in Wistar rats, Int. J. Food Sci. Nutr., 2014, 65, 720–727 CrossRef CAS PubMed.
- S. Brooke-Taylor, K. Dwyer, K. Woodford and N. Kost, Systematic review of the gastrointestinal effects of A1 compared with A2 β-casein, Adv. Nutr., 2017, 8, 739–748 CrossRef CAS PubMed.
- H. Teschemacher, Opioid receptor ligands derived from food proteins, Curr. Pharm. Des., 2003, 9, 1331–1344 CrossRef CAS PubMed.
- European Food Safety Authority, Review of the potential health impact of β-casomorphins and related peptides, EFSA J., 2009, 231, 1–107 Search PubMed.
- A. S. Truswell, The A2 milk case: a critical review, Eur. J. Clin. Nutr., 2005, 59, 623–631 CrossRef CAS PubMed.
- B. C. Esty and D. Fisher, The a2 Milk Company, https://www.hbs.edu/faculty/Pages/item.aspx?num=55342, Accessed 21/03/2024, 2024.
- F. Danesi, F. Rossi, G. Leni, G. Antonelli, A. Bordoni, T. Bertuzzi, M. Santoni, D. Risso, F. Canzoneri, R. Menta and M. Dall'Asta, Estimating β-casomorphin-7 exposure from milk and dairy product consumption: a comprehensive assessment for the European population, Int. J. Food Sci. Nutr., 2025, 76, 571–580 Search PubMed.
- M. Santoni, G. Piccinini, G. Liguori, M. R. Randi, M. Baroncini, L. Milani and F. Danesi, Enhanced intestinal epithelial co-culture model with orbital mechanical stimulation: a proof-of-concept application in food nanotoxicology, Front. Mol. Biosci., 2024, 11, 1529027 CrossRef CAS PubMed.
- E. Papadatou-Soulou, J. Mason, C. Parsons, A. Oates, M. Thyagarajan and H. K. Batchelor, Magnetic resonance imaging quantification of gastrointestinal liquid volumes and distribution in the gastrointestinal tract of children, Mol. Pharm., 2019, 16, 3896–3903 CrossRef CAS PubMed.
- C. Schiller, C. P. Fröhlich, T. Giessmann, W. Siegmund, H. Monnikes, N. Hosten and W. Weitschies, Intestinal fluid volumes and transit of dosage forms as assessed by magnetic resonance imaging, Aliment. Pharmacol. Ther., 2005, 22, 971–979 CrossRef CAS PubMed.
- P. O'Brien and J. R. Haskins, In vitro cytotoxicity assessment, Methods Mol. Biol., 2007, 356, 415–425 Search PubMed.
- F. K. Chan, K. Moriwaki and M. J. De Rosa, Detection of necrosis by release of lactate dehydrogenase activity, Methods Mol. Biol., 2013, 979, 65–70 Search PubMed.
- M. Mantle and A. Allen, A colorimetric assay for glycoproteins based on the periodic acid/Schiff stain, Biochem. Soc. Trans., 1978, 6, 607–609 Search PubMed.
- S. Yamabayashi, Periodic acid-Schiff-alcian blue: a method for the differential staining of glycoproteins, Histochem. J., 1987, 19, 565–571 Search PubMed.
- V. Manna and S. Caradonna, Isolation, expansion, differentiation, and histological processing of human nasal epithelial cells, STAR Protoc., 2021, 2, 100782 Search PubMed.
- T. Pelaseyed, J. H. Bergstrom, J. K. Gustafsson, A. Ermund, G. M. Birchenough, A. Schütte, S. van der Post, F. Svensson, A. M. Rodríguez-Piñeiro, E. E. Nystrom, C. Wising, M. E. Johansson and G. C. Hansson, The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system, Immunol. Rev., 2014, 260, 8–20 Search PubMed.
- J. Vandesompele, K. De Preter, F. Pattyn, B. Poppe, N. Van Roy, A. De Paepe and F. Speleman, Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes, Genome Biol., 2002, 3, research0034.1 Search PubMed.
- S. Neves-Coelho, R. P. Eleutério, F. J. Enguita, V. Neves and M. Castanho, A new noncanonical anionic peptide that translocates a cellular blood-brain barrier model, Molecules, 2017, 22, 1753 CrossRef PubMed.
- H. Almeida, A. C. F. Vieira, J. Teixeira, M. J. Gomes, P. Barrocas, T. Vasconcelos and B. Sarmento, in Drug Discovery and evaluation: Safety and pharmacokinetic assays, ed. F. J. Hock, M. R. Gralinski and M. K. Pugsley, Springer International Publishing, Cham, Switzerland, 2022, pp. 1–22 Search PubMed.
- C. Schimpel, B. Teubl, M. Absenger, C. Meindl, E. Fröhlich, G. Leitinger, A. Zimmer and E. Roblegg, Development of an advanced intestinal in vitro triple culture permeability model to study transport of nanoparticles, Mol. Pharm., 2014, 11, 808–818 Search PubMed.
- B. Dolan, J. Naughton, N. Tegtmeyer, F. E. May and M. Clyne, The interaction of Helicobacter pylori, with the adherent mucus gel layer secreted by polarized HT29-MTX-E12 cells, PLoS One, 2012, 7, e47300 CrossRef CAS PubMed.
- S. Zoghbi, A. Trompette, J. Claustre, M. El Homsi, J. Garzón, G. Jourdan, J. Y. Scoazec and P. Plaisancié, β-Casomorphin-7 regulates the secretion and expression of gastrointestinal mucins through a μ-opioid pathway, Am. J. Physiol.: Gastrointest. Liver Physiol., 2006, 290, G1105–G1113 CrossRef CAS PubMed.
- A. Trompette, J. Claustre, F. Caillon, G. Jourdan, J. A. Chayvialle and P. Plaisancié, Milk bioactive peptides and β-casomorphins induce mucus release in rat jejunum, J. Nutr., 2003, 133, 3499–3503 Search PubMed.
- P. Plaisancié, J. Claustre, M. Estienne, G. Henry, R. Boutrou, A. Paquet and J. Léonil, A novel bioactive peptide from yoghurts modulates expression of the gel-forming MUC2 mucin as well as population of goblet cells and Paneth cells along the small intestine, J. Nutr. Biochem., 2013, 24, 213–221 Search PubMed.
- H. Teschemacher, G. Koch and V. Brantl, Milk protein-derived opioid receptor ligands, Pept. Sci., 1997, 43, 99–117 CrossRef CAS.
- M. Herath, S. Hosie, J. C. Bornstein, A. E. Franks and E. L. Hill-Yardin, The role of the gastrointestinal mucus system in intestinal homeostasis: implications for neurological disorders, Front. Cell. Infect. Microbiol., 2020, 10, 248 CrossRef CAS PubMed.
- T. Lesuffleur, N. Porchet, J. P. Aubert, D. Swallow, J. R. Gum, Y. S. Kim, F. X. Real and A. Zweibaum, Differential expression of the human mucin genes MUC1 to MUC5 in relation to growth and differentiation of different mucus-secreting HT-29 cell subpopulations, J. Cell Sci., 1993, 106, 771–783 CrossRef CAS PubMed.
- J. Elzinga, B. van der Lugt, C. Belzer and W. T. Steegenga, Characterization of increased mucus production of HT29-MTX-E12 cells grown under semi-wet interface with mechanical stimulation, PLoS One, 2021, 16, e0261191 Search PubMed.
- Y. Y. Lee, A. Erdogan and S. S. Rao, How to assess regional and whole gut transit time with wireless motility capsule, J. Neurogastroenterol. Motil., 2014, 20, 265–270 CrossRef PubMed.
- A. M. Barnett, N. C. Roy, A. L. Cookson and W. C. McNabb, Metabolism of caprine milk carbohydrates by probiotic bacteria and Caco-2:HT29-MTX epithelial co-cultures and their impact on intestinal barrier integrity, Nutrients, 2018, 10, 949 CrossRef PubMed.
- M. Sobczak, M. Sałaga, M. A. Storr and J. Fichna, Physiology, signaling, and pharmacology of opioid receptors and their ligands in the gastrointestinal tract: current concepts and future perspectives, J. Gastroenterol., 2014, 49, 24–45 CrossRef CAS PubMed.
- G. Nylund, A. Pettersson, C. Bengtsson, A. Khorram-Manesh, S. Nordgren and D. S. Delbro, Functional expression of μ-opioid receptors in the human colon cancer cell line, HT-29, and their localization in human colon, Dig. Dis. Sci., 2008, 53, 461–466 Search PubMed.
- D. A. Clare and H. E. Swaisgood, Bioactive milk peptides: a prospectus, J. Dairy Sci., 2000, 83, 1187–1195 CrossRef CAS PubMed.
- E. Fiedorowicz, L. H. Markiewicz, K. Sidor, D. Świątecka, A. Cieślińska, M. Matysiewicz, K. Piskorz-Ogórek, E. Sienkiewicz-Szłapka, M. Teodorowicz, A. Świątecki and E. Kostyra, The influence of breast milk and infant formulae hydrolysates on bacterial adhesion and Caco-2 cells functioning, Food Res. Int., 2016, 89, 679–688 CrossRef CAS PubMed.
- A. Rieder, S. Grimmer, S. O. Kolset, T. E. Michaelsen and S. H. Knutsen, Cereal β-glucan preparations of different weight average molecular weights induce variable cytokine secretion in human intestinal epithelial cell lines, Food Chem., 2011, 128, 1037–1043 CrossRef CAS.
- J. Van De Walle, A. Hendrickx, B. Romier, Y. Larondelle and Y. J. Schneider, Inflammatory parameters in Caco-2 cells: effect of stimuli nature, concentration, combination and cell differentiation, Toxicol. In Vitro, 2010, 24, 1441–1449 CrossRef CAS PubMed.
- E. Hoffmann, O. Dittrich-Breiholz, H. Holtmann and M. Kracht, Multiple control of interleukin-8 gene expression, J. Leukocyte Biol., 2002, 72, 847–855 CrossRef CAS PubMed.
- E. Fiedorowicz, B. Jarmołowska, M. Iwan, E. Kostyra, R. Obuchowicz and M. Obuchowicz, The influence of μ-opioid receptor agonist and antagonist peptides on peripheral blood mononuclear cells (PBMCs), Peptides, 2011, 32, 707–712 CrossRef CAS PubMed.
- M. Grujic, I. Z. Matic, M. D. Crnogorac, A. D. Velickovic, B. Kolundzija, O. J. Cordero, Z. Juranic, S. Prodanovic, M. Zlatanovic, D. Babic and N. Damjanov, Activity and expression of dipeptidyl peptidase IV on peripheral blood mononuclear cells in patients with early steroid and disease modifying antirheumatic drugs naïve rheumatoid arthritis, Clin. Chem. Lab. Med., 2017, 55, 73–81 CrossRef CAS PubMed.
- S. De Pascale, G. Picariello, A. D. Troise, S. Caira, G. Pinto, F. Marino, A. Scaloni and F. Addeo, Ex vivo degradation of β-casomorphin-7 by human plasma peptidases: Potential implications for peptide systemic effects, J. Funct. Foods, 2024, 113, 106004 CrossRef CAS.
- M. Giribaldi, C. Lamberti, S. Cirrincione, M. G. Giuffrida and L. Cavallarin, A2 milk and BCM-7 peptide as emerging parameters of milk quality, Front. Nutr., 2022, 9, 842375 CrossRef PubMed.
- S. Arısoy and Ö. Üstün-Aytekin, Hydrolysis of food-derived opioids by dipeptidyl peptidase IV from Lactococcus lactis spp. lactis, Food Res. Int., 2018, 111, 574–581 CrossRef PubMed.
- S. A. Adibi, The oligopeptide transporter (Pept-1) in human intestine: Biology and function, Gastroenterology, 1997, 113, 332–340 CrossRef CAS PubMed.
- M. Brandsch, Drug transport via the intestinal peptide transporter PepT1, Curr. Opin. Pharmacol., 2013, 13, 881–887 CrossRef CAS PubMed.
- M. Iwan, B. Jarmołowska, K. Bielikowicz, E. Kostyra, H. Kostyra and M. Kaczmarski, Transport of μ-opioid receptor agonists and antagonist peptides across Caco-2 monolayer, Peptides, 2008, 29, 1042–1047 CrossRef CAS PubMed.
- S. Osborne, W. Chen, R. Addepalli, M. Colgrave, T. Singh, C. Tran and L. Day, In vitro transport and satiety of a beta-lactoglobulin dipeptide and beta-casomorphin-7 and its metabolites, Food Funct., 2014, 5, 2706–2718 RSC.
- S. Ananth, S. V. Thakkar, J. P. Gnana-Prakasam, P. M. Martin, P. S. Ganapathy, S. B. Smith and V. Ganapathy, Transport of the synthetic opioid peptide DADLE ([D-Ala2,D-Leu5]-enkephalin) in neuronal cells, J. Pharm. Sci., 2012, 101, 154–163 CrossRef CAS PubMed.
- E. Fiedorowicz, M. Kaczmarski, A. Cieślińska, E. Sienkiewicz-Szłapka, B. Jarmołowska, B. Chwała and E. Kostyra, β-casomorphin-7 alters μ-opioid receptor and dipeptidyl peptidase IV genes expression in children with atopic dermatitis, Peptides, 2014, 62, 144–149 CrossRef CAS PubMed.
- A. Summer, F. Di Frangia, P. Ajmone Marsan, I. De Noni and M. Malacarne, Occurrence, biological properties and potential effects on human health of β-casomorphin 7: Current knowledge and concerns, Crit. Rev. Food Sci. Nutr., 2020, 60, 3705–3723 CrossRef CAS PubMed.
- T. T. Lambers, S. Broeren, J. Heck, M. Bragt and T. Huppertz, Processing affects beta-casomorphin peptide formation during simulated gastrointestinal digestion in both A1 and A2 milk, Int. Dairy J., 2021, 121, 105099 CrossRef CAS.
- T. Asledottir, T. T. Le, B. Petrat-Melin, T. G. Devold, L. B. Larsen and G. E. Vegarud, Identification of bioactive peptides and quantification of β-casomorphin-7 from bovine β-casein A1, A2 and I after ex vivo gastrointestinal digestion, Int. Dairy J., 2017, 71, 98–106 CrossRef CAS.
- Z. Liu and C. C. Udenigwe, Role of food-derived opioid peptides in the central nervous and gastrointestinal systems, J. Food Biochem., 2019, 43, e12629 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |