Limosilactobacillus reuteri NCU-37 alleviates leuprorelin-induced perimenopausal syndrome in infertile women by modulating the gut microbiota: a randomized controlled trial
Xiaoyun
Wu†
a,
Weijun
Chen†
b,
Qianyu
Yong
c,
Qingwei
Zeng
c,
Jie
You
c,
Kangtai
Zeng
d,
Ting
Li
c,
Jing
Wei
e,
Yupei
Xie
*a,
Tangchang
Xu
*c and
Tingtao
Chen
 *ace
*ace
aReproductive Medicine Center of The First Affiliated Hospital, Jiangxi Medical College, Nanchang University, Nanchang 330031, China. E-mail: chentingtao1984@163.com; chentingtao@ncu.edu.cn; xieyupei1128@163.com
bThe Reproductive Hospital, Jiangxi University of Traditional Chinese Medicine, Nanchang 330004, China
cSchool of Pharmacy, Jiangxi Medical College, Nanchang University, Nanchang 330031, China. E-mail: xu2950129@163.com
dSchool of Public Health, Jiangxi Medical College, Nanchang University, Nanchang 330031, China
eJiangxi Province Key Laboratory of Bioengineering Drugs, Institute of Translational Medicine, Jiangxi Medical College, Nanchang University, Nanchang 330031, China
First published on 1st December 2025
Abstract
Leuprorelin-induced perimenopausal syndrome (LIPS) represents a common challenge faced by infertile women undergoing assisted reproductive procedures, characterized primarily by hot flashes, night sweats, and mood swings. Currently, there is a shortage of safe approaches that can effectively alleviate these symptoms without compromising the therapeutic outcomes of pregnancy. In this study, 100 infertile women with LIPS (placebo group, MP group; probiotic group, ML group) and 50 healthy women were enrolled to evaluate the therapeutic effects of Limosilactobacillus reuteri NCU-37 on LIPS. The results showed that, compared to the MP group, NCU-37 significantly reduced the Modified Kupperman Index (MKI) scores (MP vs. ML = 15.00 vs. 8.00, p < 0.01), Hamilton Anxiety Scale (HAMA) scores (11.50 vs. 7.00, p < 0.01), Hamilton Depression Scale (HAMD) scores (9.00 vs. 7.00, p < 0.01), and Athens Insomnia Scale (AIS) scores (7.50 vs. 5.00, p < 0.05). Additionally, NCU-37 significantly increased serum hormone levels, including anti-Müllerian hormone (AMH), estradiol (E2), follicle-stimulating hormone (FSH), and luteinizing hormone (LH), compared to the placebo group (p < 0.05). These results indicate that NCU-37 effectively improves perimenopausal symptoms, mood disorders, and hormone levels. Meanwhile, NCU-37 altered the composition and diversity of the gut microbiota in LIPS patients, resulting in a significant increase in beneficial bacteria (Blautia) and a reduction in pathogenic bacteria (Bacteroides). Interestingly, NCU-37 increased the pregnancy rate in LIPS patients. In conclusion, this study demonstrates for the first time that the probiotic NCU-37 alleviates LIPS by improving perimenopausal symptoms, mood disorders, hormone levels, and the gut microbiota, thereby providing data to support its clinical application.
1. Introduction
Leuprolide-induced perimenopausal syndrome (LIPS) is a clinical condition caused by a hypoestrogenic state triggered by leuprolide during assisted reproductive therapy.1,2 It is primarily characterized by vasomotor symptoms (e.g., hot flashes and night sweats), mood disorders (e.g., anxiety and depression), and sleep disturbances (e.g., difficulty falling asleep and nighttime awakenings), which may adversely affect pregnancy outcomes and the quality of life of infertile patients.3,4 Currently, studies show that approximately 40% of patients treated with leuprorelin experience moderate to severe menopausal-like symptoms (e.g., hot flashes), significantly reducing treatment compliance and quality of life, and even affecting reproductive outcomes.5,6 Although hormone therapy (HT) can effectively alleviate symptoms, its use is strictly limited due to potential interference with reproductive efficacy.7 Therefore, developing safe and effective intervention strategies is of great clinical significance for improving LIPS in these patients.Numerous pieces of evidence suggest a close association between the composition of the gut microbiota and perimenopausal symptoms.8,9 For instance, Liu et al. conducted a clinical study revealing that, compared to healthy women, patients experiencing perimenopausal symptoms exhibited gut microbiota dysbiosis, which was characterized by reduced abundances of hormone-related bacteria such as Aggregatibacter segnis, Bifidobacterium animalis, and Acinetobacter guillouiae.10 Zheng et al. further reported that perimenopausal women have decreased abundances of Collinsella, Rothia, Haemophilus, Sphingomonas, and Lactococcus compared to younger women, while the abundances of Phascolarctobacterium, Paraprevotella, and Acinetobacter are significantly increased.11 Additionally, Zong et al. established a bilateral ovariectomized (OVX) rat model and utilized 16S rRNA sequencing technology to demonstrate that fecal microbiota transplantation (FMT) can trigger hot flash symptoms in rats by modulating the composition of the gut microbiota.12 In summary, while there is a close association between the gut microbiota and perimenopausal symptoms in women, further research is needed to determine whether modulating the gut microbiota can clinically alleviate LIPS.
Probiotics are live microorganisms that confer health benefits to the host and play a significant role in improving human mental health disorders, including perimenopausal symptoms in women.13,14Limosilactobacillus reuteri is a Gram-positive probiotic bacterium commonly found in the intestines of humans and animals, which has been shown to alleviate symptoms related to mental disorders by modulating the gut microbiota, enhancing immune function, and inhibiting the growth of harmful bacteria.15,16 Xie et al. established a depression-susceptible mouse model induced by chronic social defeat stress and demonstrated that treatment with L. reuteri 3 ameliorated depressive-like behaviors, reduced the relative abundances of Clostridiales and Adlercreutzia, prevented the decline in the abundances of Lactobacillus, Allobaculum, and Sutterella, and significantly increased the abundance of Bifidobacterium.17 Jiang et al. evaluated the therapeutic effects of L. reuteri E9 on a pentylenetetrazol (PTZ)-induced sleep disorder model in zebrafish. The results revealed that this bacterial strain significantly upregulates inhibitory neurotransmitters (including GABA, taurine, glycine, and serine), exerting a sedative effect by reducing epileptic seizures and hyperactive behaviors, which indicates that L. reuteri E9 can modulate key inhibitory neurotransmitters and sleep-related receptors through the microbiota–gut–brain axis, thereby improving sleep disorders.18 Meanwhile, Ma et al. discovered that L. reuteri L6 can improve anxiety- and depression-like behaviors in mice by suppressing IL-6 expression and modulating the gut microbiota.19 However, in the context of perimenopause, Britton et al. found that L. reuteri ATCC PTA 6475 inhibits bone loss in an ovariectomized mouse model of menopause by regulating microbiota composition.20 Currently, there is a severe lack of effective treatments for LIPS. The efficacy and mechanism of action of probiotic intervention as a potential novel therapy have not been reported.
This study recruited 50 healthy women and 100 infertile women with LIPS to evaluate the ameliorative effects of L. reuteri NCU-37 on their symptoms. Assessments using the Modified Kupperman Index (MKI), Hamilton Anxiety Scale (HAMA), Hamilton Depression Scale (HAMD), and Athens Insomnia Scale (AIS) revealed that NCU-37 significantly alleviated menopausal symptoms and reduced anxiety, depression, and insomnia in LIPS patients. Additionally, the probiotic notably regulated hormone levels (e.g., increased estradiol levels) and restored the diversity and composition of the gut microbiota. Our results provide clinical data to support the development of microbial agents for treating LIPS in infertile women.
2. Materials and methods
2.1 Clinical design and subjects
This study employed a randomized controlled trial design and was conducted at Nanchang Reproductive Hospital from October 2024 to August 2025. The study participants included healthy subjects and infertile women with LIPS. All subjects were fully informed about the study details and voluntarily provided written informed consent before participation. The inclusion criteria comprised two groups. Normal healthy women: (1) healthy women with regular menstrual cycles (cycle length 21–35 days, duration 2–7 days); (2) aged 20–40 years; (3) voluntary participation with signed informed consent. Infertile women with LIPS: (1) infertile women aged 20–40 years (due to female factors or male factors contributing to female infertility) with baseline hormone levels (prior to leuprorelin intervention) as follows: FSH: 3–10 mIU mL−1, LH: 3–10 mIU mL−1, E2: 10–70 pg mL−1, AMH ≥ 1.2 ng ml−1, and a total antral follicle count > 7; (2) meeting the indications for in vitro fertilization (IVF); (3) no unexplained abnormal uterine or vaginal bleeding; (4) presence of perimenopausal syndrome symptoms after leuprorelin use, such as hot flashes, sweating, mood swings, insomnia, fatigue, and joint pain. One or more symptoms must be present and meet certain severity and duration criteria to fulfill the diagnostic standards for perimenopausal syndrome; (5) treatment history: no recent use (e.g., within the past 6 months) of other hormonal medications or therapies that may affect study outcomes; (6) informed consent: patients must fully understand the study content, purpose, risks, and benefits, and provide signed informed consent indicating voluntary participation.Additionally, the exclusion criteria also applied to both groups. Normal healthy women: (1) irregular menstruation in the past two months; (2) planning pregnancy, or currently pregnant, lactating, or in the lactation period; (3) history of abortion (medical or surgical) within the past six months; (4) use of antibiotics, contraceptive pills, or similar medications within the past month; (5) infertility due to female factors or male factors contributing to female infertility; (6) absence of major organ dysfunction or other severe diseases; (7) current participation in other clinical trials. Patients with LIPS: (1) pregnant or lactating women should not be included; (2) unexplained abnormal uterine or vaginal bleeding; (3) nonorganic or surgically induced premature ovarian failure, or diminished ovarian function due to chromosomal abnormalities; (4) severe dysfunction of major organs (e.g., heart, liver, and kidneys); (5) presence of other endocrine disorders; (6) use of antibiotics or oral probiotics within the past month; (7) allergy to gonadotropin-releasing hormone agonists (e.g., leuprorelin) or their excipients; (8) other severe diseases: including serious genetic, somatic, or psychiatric disorders contraindicated for pregnancy, as well as acute genitourinary infections or sexually transmitted diseases; (9) recent exposure to harmful substances: such as teratogenic levels of radiation, toxic substances, or teratogenic drugs/narcotics during the exposure period; (10) other ineligible conditions: including specific exclusion criteria defined in the study design, such as certain types of endometriosis or specific severity levels of menopausal syndrome symptoms.
All procedures and protocols involved in this study were thoroughly reviewed and formally approved by the Medical Ethics Committee of Nanchang Reproductive Hospital (Ethics Approval No.: 2024024). All operations were strictly conducted in accordance with the approved norms and standards. This study fully adhered to the ethical principles established in the Declaration of Helsinki. The trial was also registered with the Chinese Clinical Trial Registry (Registration No.: ChiCTR2400089614).
2.2 Randomization and blinding
The random allocation in this trial was generated using computer-based random codes assigned by staff not involved in the study. A total of 100 infertility patients with LIPS were assigned in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio to either the placebo group (MP group, n = 50) or the probiotic group (ML group, n = 50). Subsequently, designated personnel labeled the packaging of the placebo and probiotic supplements with the corresponding unique sequential numeric identifiers based on this random sequence, with the blinding maintained throughout the process. All participants and personnel administering the interventions remained blinded. Both the placebo and the L. reuteri NCU-37 probiotic preparation were identical in visual appearance, texture, odor, and unit weight, making them physically indistinguishable. L. reuteri NCU-37 was isolated from a fresh fecal sample of a 26-year-old healthy person from Nanchang, Jiangxi Province, China. The strain has been deposited in the China General Microbiological Culture Collection Center (CGMCC No. 33627).
1 ratio to either the placebo group (MP group, n = 50) or the probiotic group (ML group, n = 50). Subsequently, designated personnel labeled the packaging of the placebo and probiotic supplements with the corresponding unique sequential numeric identifiers based on this random sequence, with the blinding maintained throughout the process. All participants and personnel administering the interventions remained blinded. Both the placebo and the L. reuteri NCU-37 probiotic preparation were identical in visual appearance, texture, odor, and unit weight, making them physically indistinguishable. L. reuteri NCU-37 was isolated from a fresh fecal sample of a 26-year-old healthy person from Nanchang, Jiangxi Province, China. The strain has been deposited in the China General Microbiological Culture Collection Center (CGMCC No. 33627).
2.3 Interventions
Eligible infertile female patients first received a single subcutaneous injection of 3.75 mg leuprorelin on menstrual cycle days 2–3. Those developing perimenopausal symptoms were then screened between days 7–15 for enrollment in the randomized study. Intervention with NCU-37 or placebo was initiated only after all enrolled patients had completed randomization. Then, both the L. reuteri NCU-37 preparation and the placebo were individually packaged in composite aluminum foil bags and stored protected from light at 4 °C. Each sachet (2 g per sachet) used in the probiotic group contained L. reuteri NCU-37 with maltodextrin as the carrier, with a viable count of 2.0 × 109 CFU. The sachets for the placebo group contained only maltodextrin without any active components. Participants in both groups were instructed to orally take one sachet twice daily in the same manner for a total intervention period of 4 weeks.2.4 Data collection and outcomes
Baseline data, including age and body mass index (BMI), were systematically recorded by trained clinicians for both healthy participants and infertility patients with LIPS. Fasting venous blood (5 mL) was collected from all subjects in the morning. After centrifugation for serum separation, the samples were stored at −80 °C until analysis. Serum hormone levels were then measured using a fully automated chemiluminescence immunoassay system (UniCel DxI 800 Access, Beckman Coulter, USA). Specifically, multiple hormones related to perimenopausal status were assessed before and after the intervention, including follicle-stimulating hormone (FSH, mIU mL−1), luteinizing hormone (LH, mIU mL−1), estradiol (E2, pg mL−1), progesterone (P, ng mL−1), and anti-Müllerian hormone (AMH, ng mL−1). The severity of perimenopausal symptoms, anxiety, depression, and sleep disturbances were quantitatively assessed using multiple emotion-related questionnaires administered before and after the intervention. These included the MKI,21 HAMA,22 HAMD,23 and AIS.24 Furthermore, fresh fecal samples were collected from participants in both groups after the intervention and immediately stored at −80 °C for subsequent analysis of the gut microbiota.2.5 RT-qPCR
Fecal samples (180–220 mg) collected from participants in each group after the intervention were placed in 2 mL centrifuge tubes. Total DNA was extracted from human feces using a Fecal Genomic DNA Extraction Kit (DP328, Tiangen, China). Real-time quantitative reverse transcription polymerase chain reaction (RT-qPCR) was then performed on the ViiA 7 Real-Time PCR System (Applied Biosystems, USA) using the TB Green Premix Ex Taq™ (with Tli RNaseH Plus) kit (RR420A, Takara, Japan) to quantitatively detect the relative abundance of Lactobacillus (gene: Lac) in the fecal bacterial samples. The results were normalized using the bacterial reference gene 16S rDNA (16S). The relative expression levels were calculated using the 2−ΔΔCt method, with 35 samples tested per group. The primer sequences were as follows: Lac: forward primer 5′-CACCGCTACACATGGAG-3′, reverse primer 5′-AGCAGTAGGGAATCTTCCA-3′. 16S: forward primer 5′-ACTCCTACGGGAGGCAGCAGT-3′, reverse primer 5′-TATTACCGCGGCTGCTGGC-3′.2.6 High-throughput sequencing
Total genomic DNA was extracted from fecal samples collected from participants in each group after the intervention using the Fecal Bacterial Genomic DNA Extraction Kit (DP328, Tiangen, China) according to the manufacturer's instructions. The concentration and purity of the extracted genomic DNA were measured using a spectrophotometer (NanoDrop, Thermo Fisher Scientific, USA). Qualified DNA samples were subsequently sent to Personalbio Biotechnology Co., Ltd (Shanghai, China) for 16S rDNA amplicon sequencing. During sequencing, the V4 region of the 16S rDNA gene in each sample was amplified via PCR using specific primer pairs (forward 520F: 5′-AYTGGGYDTAAAGNG-3′; reverse 802R: 5′-TACNVGGGTATCTAATCC-3′). The amplification products were subjected to high-throughput sequencing on the Illumina HiSeq 2000 platform (Illumina, California, USA). The resulting sequencing data were analyzed using QIIME2 (version 2019.4) to assess microbial community α- and β-diversity. Taxonomic composition and intergroup structural differences at the phylum and genus levels were also examined. All raw sequencing data generated in this study have been deposited in the National Center for Biotechnology Information (NCBI) public database under the accession number PRJNA1335389 and are publicly accessible for further analysis.2.7 Sample size
Based on preliminary pre-experiment data, the MKI scores were 3.36 ± 1.69 in the healthy control group, 13.6 ± 3.56 in the placebo group, and 8.45 ± 2.54 in the probiotic group. The minimum sample size was calculated using the One-Way Analysis of Variance F-Tests. Under the assumption of a two-sided significance level of α = 0.05 and a statistical power of 0.90, the minimum required sample sizes for the three groups were 9, 9, and 15, respectively. To further enhance the robustness of the study and mitigate the impact of dropouts, loss to follow-up, and other uncontrollable factors, the final sample size per group was increased to 50 participants.2.8 Statistical analysis
All statistical analyses were performed using GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA). Data are presented as mean ± standard deviation (SD) or median (interquartile range), as appropriate. Normality of data distribution was assessed using the Shapiro–Wilk test. Continuous variables were analyzed using one-way repeated measures analysis of variance (ANOVA). For normally distributed data, multiple comparisons were conducted using Tukey's test; for non-normally distributed data, Dunn's test was applied as a post hoc method. Correlations between variables were examined using Pearson correlation analysis. A p-value of less than 0.05 was considered statistically significant.3. Results
3.1 Baseline characteristics of participants
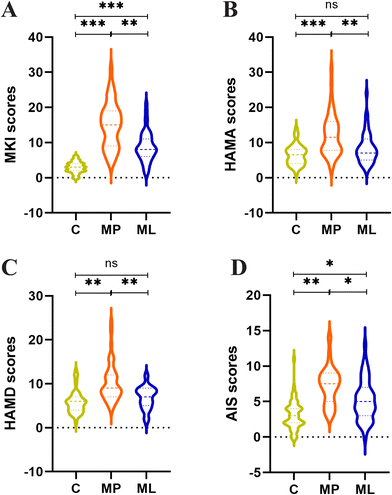
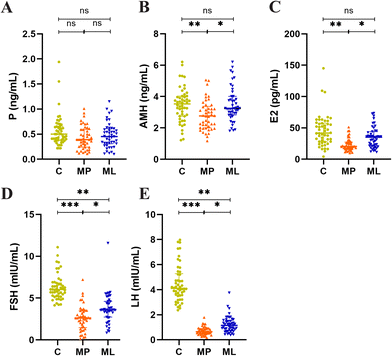
A total of 243 women were initially recruited for this study. After rigorous screening based on inclusion and exclusion criteria, 45 individuals did not meet the inclusion criteria, 11 declined to participate, and 37 withdrew for other reasons. Ultimately, 150 subjects were enrolled and randomly assigned to the healthy control group (C group, n = 50), the placebo group (MP group, n = 50), or the probiotic group (ML group, n = 50). During follow-up, a total of 143 participants completed the clinical trial, with 50 in Group C, 46 in Group MP, and 47 in Group ML, due to loss to follow-up and intervention discontinuation (Fig. 1).Baseline characteristics analysis showed that the mean ages of the three groups were 33.50, 34.00, and 35.00 years, respectively, with no significant difference among groups (p = 0.7926) (Table 1 and Table S1). The mean BMI values were 22.55 ± 2.62, 22.46 ± 2.88, and 22.48 ± 3.14, respectively, with no statistically significant difference (p = 0.9880). The MKI scores were 3.00, 13.50, and 13.00 in the three groups, showing a significant difference (p < 0.0001). Compared with the C group, both the MP group and the ML group had significantly higher HAMA scores (anxiety), HAMD scores (depression), and AIS scores (insomnia). These results indicate that infertile women with LIPS exhibited severe perimenopausal symptoms along with mild anxiety, depression, and insomnia tendencies compared to the healthy population, while no significant differences were observed between the MP group and the ML group in these indicators. Furthermore, hormone testing results revealed that both the MP group and the ML group had significantly lower levels of E2, P, and AMH compared to the C group, but no significant differences were found between the MP group and the ML group. In summary, no significant differences were observed in any baseline indicators between the placebo and probiotic groups, indicating high homogeneity at the start of the study.
| Variables | C (n = 50) | MP (n = 46) | ML (n = 47) | p-Value |
|---|---|---|---|---|
| BMI: body mass index; MKI: Modified Kupperman Index; HAMA: Hamilton Anxiety Scale; HAMD: Hamilton Depression Scale; AIS: Athens Insomnia Scale. FSH: follicle-stimulating hormone; LH: luteinizing hormone; E2: estradiol; P: progesterone; AMH: anti-Müllerian hormone. C: healthy group (n = 50); MP: placebo group (n = 46); ML: probiotic group (n = 47). Data are shown as mean ± standard deviation (SD) and median (interquartile range). The normality of the datasets was analyzed using the Shapiro–Wilk test. Continuous variables were compared using one-way repeated measures ANOVA, followed by Tukey's test (BMI and FSH) or Dunn's test (Age, MKI, HAMA, HAMD, AIS, LH, E2, P and AMH) for multiple comparisons. Significance levels are indicated as *p < 0.05. The data comparison between the MP group and the ML group showed no significant differences. | ||||
| Age (years) | 33.50 (30.75–36.00) | 34.00 (30.75–37.00) | 35.00 (30.00–37.00) | 0.7926 |
| BMI (kg m−2) | 22.55 ± 2.62 | 22.46 ± 2.88 | 22.48 ± 3.14 | 0.9880 |
| MKI scores | 3.00 (2.00–4.00) | 13.50 (9.75–19.50) | 13.00 (9.00–18.00) | <0.0001 |
| HAMA scores | 6.00 (5.00–7.25) | 9.50 (8.00–14.25) | 12.00 (8.00–15.00) | <0.0001 |
| HAMD scores | 5.50 (4.00–7.00) | 9.00 (7.00–13.25) | 9.00 (8.00–13.00) | <0.0001 |
| AIS scores | 3.00 (2.00–4.00) | 6.00 (4.00–9.00) | 7.00 (5.00–9.00) | <0.0001 |
| FSH (mIU mL−1) | 6.29 ± 2.42 | 2.57 ± 1.26 | 2.51 ± 1.57 | <0.0001 |
| LH (mIU mL−1) | 4.07 (2.96–5.83) | 0.58 (0.50–0.86) | 0.69 (0.42–0.90) | <0.0001 |
| E2 (pg mL−1) | 46.70 (32.88–53.92) | 20.36 (16.59–30.28) | 21.16 (15.28–28.22) | <0.0001 |
| P (ng mL−1) | 0.53 (0.33–0.69) | 0.39 (0.23–0.63) | 0.40 (0.21–0.60) | 0.0318 |
| AMH (ng mL−1) | 3.68 (2.45–4.13) | 2.33 (1.95–3.66) | 2.44 (1.72–3.72) | 0.0072 |
3.2 L. reuteri NCU-37 significantly improved perimenopausal syndrome and mood disorders in patients with LIPS
Numerous studies have shown that women in the perimenopausal period often experience significant menopausal symptoms, which are frequently accompanied by mood disorders such as anxiety and depression, as well as sleep problems.25 To evaluate these symptoms, we conducted a 4-week intervention using L. reuteri NCU-37 in women with LIPS. As shown in Table S2 and Fig. 2, compared with the MP group, the ML group exhibited significant reductions in all assessed scores: the MKI score decreased from 15.00 to 8.00 (p < 0.01, Fig. 2A); the HAMA score decreased from 11.50 to 7.00 (p < 0.01, Fig. 2B); the HAMD score decreased from 9.00 to 7.00 (p < 0.01, Fig. 2C); and the AIS score decreased from 7.50 to 5.00 (p < 0.05, Fig. 2D). These results indicate that L. reuteri NCU-37 significantly alleviates perimenopausal syndrome and related mood disorders (including anxiety, depression, and insomnia).3.3 L. reuteri NCU-37 significantly increased hormone levels in patients with LIPS
Research indicates that leuprolide causes a marked reduction in E2 levels by reversibly inhibiting ovarian function, thereby inducing an artificial perimenopausal syndrome.26 Therefore, we measured key hormone levels associated with perimenopause, including E2, P, and AMH. The results showed that, compared to the MP group, the ML group exhibited an increase in P levels (MP vs. ML = 0.39 vs. 0.45), although the difference was not statistically significant (Fig. 3A). Meanwhile, the ML group demonstrated a significant increase in AMH levels compared to the MP group (MP vs. ML = 2.76 vs. 3.24, p < 0.05; Fig. 3B). The E2 levels were also significantly elevated (MP vs. ML = 20.46 vs. 35.01, p < 0.05; Fig. 3C). Additionally, the FSH levels increased significantly (MP vs. ML = 2.60 vs. 3.60, p < 0.05; Fig. 3D), as did LH levels (MP vs. ML = 0.64 vs. 1.15, p < 0.05; Fig. 3E). Interestingly, in terms of pregnancy outcomes, infertile women with LIPS who received L. reuteri NCU-37 intervention showed an increased pregnancy rate (MP vs. ML = 65.2% vs. 72.3%), suggesting that probiotic intervention may help improve pregnancy outcomes in infertile women (Table S2). These findings indicate that L. reuteri NCU-37 significantly improves hormone levels in infertile women with LIPS.3.4 L. reuteri NCU-37 significantly reduced gut microbiota diversity in patients with LIPS
To further investigate the role of the gut microbiota in LIPS, we conducted high-throughput sequencing on fecal samples from the C (n = 35), MP (n = 35), and ML (n = 35) groups after the intervention. The α-diversity analysis revealed that compared with the C group, the MP group showed significant increases in the indices of Chao1, Faith_pd, Shannon, and Observed_species (p < 0.01). Following probiotic intervention, all these indices were significantly decreased in the ML group compared to the MP group (p < 0.05), indicating that L. reuteri NCU-37 effectively reduced the α-diversity of the gut microbiota (Fig. 4A). Principal coordinates analysis (PCoA) results showed partial overlap among samples from different groups, though certain intergroup differences remained observable (Fig. 4B). A Venn diagram revealed that a total of 5713 operational taxonomic units (OTUs) were generated across all samples, with 582 OTUs shared among the three groups. The C group, MP group, and ML group had unique OTUs of 953, 2183, and 1459, respectively (Fig. 4C). Intergroup distance comparison demonstrated significant differences between each pair of groups (p < 0.001) (Fig. 4D). Furthermore, rarefaction curves indicated that at the same sequencing depth, the MP group exhibited higher curve plateaus than both the C group and ML group, further confirming greater species richness in the MP group (Fig. 4E). These results suggest that L. reuteri NCU-37 may alleviate LIPS in infertile women by modulating gut microbiota diversity.3.5 L. reuteri NCU-37 modulated gut microbiota composition in patients with LIPS
Subsequently, we analyzed the taxonomic composition of the gut microbiota in each group following the intervention. At the phylum level, the fecal microbiota of all participants was primarily composed of Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria (Fig. 5A). Compared with the C group, the MP group showed a significant decrease in the relative abundance of Firmicutes (p < 0.001) and a significant increase in Bacteroidetes (p < 0.001) (Fig. 5B and C). These trends were reversed after probiotic intervention. Further analysis at the genus level focused on the top 30 genera. The fecal microbiota was dominated by Blautia, Collinsella, Bacteroides, Faecalibacterium, and Coprococcus (Fig. 5D). Compared with the C group, the MP group exhibited a significant increase in the relative abundance of Bacteroides and Parabacteroides (p < 0.001), which decreased in the ML group (no significant difference) after probiotic intervention. Additionally, Prevotella showed a significant decrease following probiotic intervention (p < 0.05). Notably, no significant differences were observed in Blautia or Coprococcus among the groups (Fig. 5E–I). Furthermore, we quantified the relative abundance of Lactobacillus in fecal samples using RT-qPCR. The results showed that compared with the C group, the abundance of this bacterium was significantly lower in the MP group (p < 0.05), while probiotic intervention significantly increased its abundance in the ML group compared to the MP group (p < 0.001) (Fig. 5J). These findings suggest that L. reuteri NCU-37 may alleviate LIPS in infertile women by modulating the gut microbiota, particularly by reducing the proliferation of potentially pathogenic bacteria such as Bacteroides.In addition, we performed differential species analysis. LEfSe results indicated that beneficial bacteria, such as Bifidobacterium, were significantly enriched in the C group, while the MP group was characterized by the enrichment of potentially harmful bacteria, including Bacteroides (Fig. 6A). Notably, after probiotic intervention, the ML group showed enrichment of the beneficial bacterium Akkermansia. Random forest analysis further supported these findings, indicating that Bacteroides played an important role in intergroup differences, with its abundance decreasing after intervention (Fig. 6B). These results imply that L. reuteri NCU-37 may improve LIPS in infertile women by reducing the abundance of Bacteroides in the gut microbiota.
3.6 Correlation analysis among the gut microbiota, clinical symptoms, and hormone levels
To investigate the associations between the gut microbiota, clinical symptoms, and hormone levels in patients with LIPS, we performed Pearson correlation analysis. The results revealed that the MKI score was positively correlated with Bacteroidetes (p < 0.001), Bacteroides (p < 0.05), Parabacteroides (p < 0.01), and Prevotella (p < 0.01), and significantly negatively correlated with Firmicutes (p < 0.001) and Blautia (p < 0.05). The HAMA score showed a significant positive correlation with Bacteroidetes (p < 0.01) and Prevotella (p < 0.001), and a significant negative correlation with Firmicutes (p < 0.05). The AIS score was significantly positively correlated with Bacteroidetes (p < 0.01) and Bacteroides (p < 0.01), and significantly negatively correlated with Firmicutes (p < 0.01). Furthermore, both FSH and LH were significantly negatively correlated with Bacteroidetes and Bacteroides, and significantly positively correlated with Firmicutes and Blautia. Notably, E2 was significantly negatively correlated with Bacteroidetes and Bacteroides (Fig. 7). These results indicate a close relationship between the severity of perimenopausal symptoms, hormonal changes, and structural imbalances in the gut microbiota among infertile women with LIPS.4. Discussion
With the rapid development of assisted reproductive technology (ART), infertile women receiving leuprorelin therapy have frequently experienced perimenopausal symptoms (such as hot flashes, mood swings, and sleep disturbances) due to hypoestrogenism.27,28 This issue extends beyond the scope of general adverse drug reactions and has become a significant clinical obstacle that may severely impact the outcomes of ART.29 Although HT can effectively relieve related symptoms, its use in this population remains limited due to potential adverse effects on the reproductive process.30,31 Now, probiotics, as active microorganisms that can improve immune and metabolic health, have shown potential value in regulating mood and sleep.32 However, the efficacy and underlying mechanisms of probiotic interventions for LIPS remain unexplored. Therefore, this study aims to evaluate the potential role of L. reuteri NCU-37 in improving the quality of life and promoting pregnancy outcomes in patients with LIPS, thereby offering new insights for addressing this clinical challenge.This study enrolled 50 healthy women and 100 infertile women with LIPS to evaluate the effects of probiotic intervention on their symptoms. Before intervention, compared with the C group, infertile women with LIPS showed significantly aggravated perimenopausal symptoms (including anxiety, depression, insomnia, and other mood disorders), along with significantly reduced hormone levels (Table 1). These findings are similar to the research by Snigdha et al.,33 further confirming the negative impact of perimenopausal symptoms on women's quality of life and aligning with multiple reports on age-related hormonal decline.34,35 The MKI score served as the primary endpoint to comprehensively and rapidly assess the severity and frequency of perimenopausal symptoms.36 Our findings demonstrated that NCU-37 supplementation for one month led to a significant reduction in the MKI score compared to the MP group, suggesting that NCU-37 intervention is effective in alleviating the core symptomatology associated with LIPS. Furthermore, Zhao et al. also reported a high prevalence of anxiety, depressive symptoms, and sleep problems among perimenopausal women, adversely affecting their physical and mental health.25 Our results confirmed this finding, demonstrating that NCU-37 significantly improved the anxiety, depression, and insomnia of patients compared to the MP group (Fig. 2). In conclusion, NCU-37 can significantly ameliorate perimenopausal symptoms and mood disorders in infertile women with LIPS.
Perimenopause, a critical endocrine transition phase in a woman's life, is widely recognized as a key physiological basis for the development of mood disorders due to its associated fluctuations in estrogen levels.37 E2 is a key hormone secreted by the ovaries, whose decline is considered a core mechanism underlying vasomotor symptoms, mood swings, and sleep disturbances in perimenopausal women.38,39 Similarly, our results indicated that NCU-37 intervention significantly increased the serum E2 levels in patients (p < 0.05), suggesting that the elevation of E2 may be one of the underlying mechanisms for its symptomatic improvement (Fig. 3). Additionally, we observed a significant increase in the levels of FSH and LH (p < 0.05), which was consistent with the studies by Lee et al.40 and Yun et al.41 FSH and LH are two key gonadotropins secreted by the pituitary gland.28 Chronic administration of leuprorelin suppresses pituitary function, leading to a sharp decline in FSH and LH levels, which in turn results in decreased estrogen production by the ovaries. In contrast, women undergoing natural perimenopause typically exhibit elevated levels of FSH and LH. This distinct hormonal profile suggests that NCU-37 may alleviate LIPS by modulating pituitary function. Additionally, AMH, secreted by granulosa cells of preantral and small antral follicles, is widely regarded as the most direct and stable biomarker for assessing ovarian reserve.42 The results showed a significant increase in serum AMH levels after probiotic intervention, indicating that probiotics may positively influence ovarian function. It is noteworthy that NCU-37 intervention increased the clinical pregnancy rate in infertile women with LIPS, suggesting that probiotic supplementation may positively influence pregnancy outcomes by improving overall physiological status. Additionally, the C group provides a critical baseline for evaluating the efficacy of NCU-37. By comparing with the C group, it has been confirmed that NCU-37 can guide physiological indicators back to normal ranges, which not only signifies symptom alleviation but also marks the restoration of physiological functions. In summary, supplementation with L. reuteri NCU-37 may alleviate perimenopausal symptoms in infertile women during ART by regulating hormone levels, offering significant clinical value for improving both pregnancy outcomes and quality of life.
With the rapid development of microbiome research technologies and ongoing methodological innovations, substantial evidence has demonstrated that the gut microbiota plays a key role in maintaining human health and regulating disease progression.43 Particularly in perimenopausal women, the gut microbiota can modulate core symptoms such as mood swings, sleep disturbances, and hot flashes by regulating estrogen levels, thereby alleviating clinical manifestations to some extent.8 Based on this, the present study used high-throughput sequencing technology to systematically compare the compositional structure of the fecal microbiota among each group. The results showed that after intervention with L. reuteri NCU-37, the previously disrupted gut microbiota diversity indices (including the Chao1 index, Faith_pd index, Shannon index, and Observed_species index) were significantly improved (Fig. 4). At the phylum level, compared with the placebo group, the relative abundance of Firmicutes significantly increased in the probiotic group, while the relative abundance of Bacteroidota decreased markedly. At the genus level, the abundance of Prevotella also significantly decreased due to probiotic intervention (Fig. 5). These results align with the study by Zheng et al.,9 indicating that targeted modulation of gut microbiota diversity can effectively correct microecological imbalances associated with menopausal syndrome, thereby exerting protective effects. L. reuteri, an important probiotic belonging to the phylum Firmicutes and genus Lactobacillus, naturally exists in the gut and breast milk of humans and various animals.44,45 Numerous studies have shown that this bacterium can influence brain function and behavior through multiple pathways (such as immune, neuroendocrine, and neural mechanisms), constituting the core of the so-called “gut–brain axis” communication.46 For example, Mo et al.16 demonstrated that L. reuteri DSM 17938 significantly improved LPS-induced depression and anxiety-like behaviors by regulating the gut microbiota and brain metabolic function. Liu et al.47 found that supplementation with L. reuteri or exogenous butyrate reversed depression-like behaviors, a mechanism involving reduced activation of hippocampal astrocytes and upregulation of Nrf2 expression. Soviji et al.48 also reported that ovarian progesterone could inhibit depression and anxiety-like behaviors by increasing the abundance of L. reuteri in the gut of ovariectomized mice. In summary, our results suggest that L. reuteri NCU-37 may improve anxiety and depression symptoms in patients with LIPS via the gut–brain axis mechanism. Based on the current results, it can be inferred that supplementation with L. reuteri NCU-37 may reshape the gut microbiota structure and diversity in infertile women with LIPS, thereby alleviating their related clinical symptoms. Furthermore, compared with traditional hormone therapy, NCU-37 demonstrates unique advantages across multiple dimensions. In terms of safety, this strain shows beneficial effects without causing any adverse symptoms. Regarding efficacy, it potentially acts through systemic mechanisms such as the “gut–brain axis”, not only gradually alleviating core perimenopausal symptoms but also producing positive improvements in mood and sleep quality. This microecological modulation-based approach may offer more physiological and sustainable benefits. In patient acceptance, NCU-37 significantly enhances treatment adherence owing to its “natural” properties, convenient oral administration, and elimination of requent medical monitoring. Consequently, NCU-37 represents a comprehensive interventional strategy for infertile female patients with mild to moderate perimenopausal symptoms.
Here, this study has several limitations: (1) the number of infertile women with LIPS included and the sample size tested were relatively small. Future studies should expand the sample size to obtain more reliable results. Furthermore, it is worth delving deeper into whether probiotic interventions yield differential improvement effects on infertile women across different age groups by including larger cohorts in future studies. (2) The subjects enrolled were infertile women who developed perimenopausal syndrome due to leuprorelin treatment. This population has specific pathophysiological conditions and cannot simulate naturally perimenopausal women. Further evaluation of the efficacy of L. reuteri NCU-37 is needed. (3) Although correlation analysis was conducted, our results did not integrate multiomics data (such as metagenomics and metabolomics) to elucidate the functional genes, pathways, or microbial metabolites (e.g., short-chain fatty acids and indoles) through which L. reuteri NCU-37 improves LIPS, nor did they clarify the underlying mechanism. (4) The study did not comprehensively record or control confounding factors during the intervention period, such as diet, lifestyle habits, and medication use. These factors may influence the gut microbiota and clinical symptoms. It is particularly important that future studies implement more rigorous dietary assessment methods, such as validated food frequency questionnaires or 24-hour dietary recalls, to better control for confounding factors and ensure an accurate evaluation of the NCU-37 intervention effects. (5) The underlying mechanism by which NCU-37 improves pregnancy rates remains unclear, though it is likely the result of combined effects involving optimization of the systemic endocrine environment for embryo implantation and enhancement of uterine receptivity. Future studies should establish animal models to investigate this further. (6) Due to the lack of pre-intervention microbiota data, we cannot determine whether the observed correlations between the microbiota and clinical indicators accurately reflect dynamic associations. Future studies should adopt an intensive longitudinal sampling design, dynamically monitoring both microbiota and host indicators at multiple time points before, during, and after the intervention, so as to provide more direct evidence for elucidating the causal relationship between microbiota and clinical phenotypes. (7) This study did not perform within-group comparative analyses before and after the intervention, which may to some extent limit the interpretation of the intervention effects.
5. Conclusions
In summary, our results provide the first evidence that L. reuteri NCU-37 can significantly alleviate LIPS in infertile women and promote the recovery of various perimenopause-related hormone levels, including FSH, LH, E2, and AMH. More importantly, probiotic intervention significantly increased the clinical pregnancy rate, suggesting its potential clinical value in improving fertility outcomes. Further investigation revealed that NCU-37 may exert its beneficial effects on both symptoms and reproductive outcomes by remodeling the diversity and composition of the gut microbiota (Fig. 8), thereby indirectly regulating endocrine function. Future studies should include larger sample sizes, long-term follow-up, and validation in naturally perimenopausal populations to fully elucidate the intervention effects of NCU-37 and promote its clinical translation. In future studies, we will utilize animal models to verify that NCU-37 alleviates LIPS symptoms through gut–brain axis-mediated neuroendocrine regulation and to further decipher its precise mechanisms. This research provides foundational data supporting the development of NCU-37 as a clinical microecological agent for adjuvant treatment of LIPS in infertile women. | ||
| Fig. 8 Schematic diagram of the effect of L. reuteri NCU-37 on perimenopausal symptoms, hormone levels, and the gut microbiota in infertile women induced by leuprolide. | ||
Author contributions
Xiaoyun Wu and Weijun Chen contributed to methodology, investigation, formal analysis, and writing – original draft. Qianyu Yong contributed to methodology. Qingwei Zeng contributed to resources and software. Jie You contributed to data curation. Kangtai Zeng contributed to supervision. Ting Li and Jing Wei contributed to visualization. Yupei Xie, Tangchang Xu and Tingtao Chen contributed to conceptualization and writing – review and editing. Tingtao Chen contributed to funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting this study are available in the article and its supplementary information (SI) (Tables S1 and S2). Raw sequences have been deposited in the NCBI database under the accession number PRJNA1335389.Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo04330a.
Acknowledgements
This work was supported by the Jiangxi Province Key Laboratory of Bioengineering Drugs (2024SSY07061, for Tingtao Chen).References
- H. Joffe, S. Crawford, N. Economou, S. Kim, S. Regan, J. E. Hall and D. White, A gonadotropin-releasing hormone agonist model demonstrates that nocturnal hot flashes interrupt objective sleep, Sleep, 2013, 36, 1977–1985 Search PubMed.
- S. Zhang, L. Wang, J. Zhang, W. An and L. Jia, The efficacy and safety of Kuntai capsule combined with leuprorelin acetate in the treatment of endometriosis: A protocol for systematic review and meta-analysis, Medicine, 2021, 100, e25080 CrossRef CAS.
- E. R. Duralde, T. H. Sobel and J. E. Manson, Management of perimenopausal and menopausal symptoms, Br. Med. J., 2023, 382, e072612 Search PubMed.
- M. Hickey, P. Basu, J. Sassarini, M. E. Stegmann, E. Weiderpass, K. Nakawala Chilowa, C. H. Yip, A. H. Partridge and D. J. Brennan, Managing menopause after cancer, Lancet, 2024, 403, 984–996 Search PubMed.
- S. Daya, J. Gunby, E. G. Hughes, J. A. Collins and M. A. Sagle, Randomized controlled trial of follicle stimulating hormone versus human menopausal gonadotrophin in in vitro fertilization, Hum. Reprod., 1995, 10, 1392–1396 Search PubMed.
- J. Donnez, J. Tomaszewski, F. Vazquez, P. Bouchard, B. Lemieszczuk, F. Baro, K. Nouri, L. Selvaggi, K. Sodowski, E. Bestel, P. Terrill, I. Osterloh and E. Loumaye, Ulipristal acetate versus leuprolide acetate for uterine fibroids, N. Engl. J. Med., 2012, 366, 421–432 CrossRef PubMed.
- A. J. Voedisch, Updates in nonhormonal therapy for perimenopausal and menopausal symptoms, Curr. Opin. Obstet. Gynecol., 2025, 37, 67–74 CrossRef PubMed.
- H. Wang, F. Shi, L. Zheng, W. Zhou, B. Mi, S. Wu and X. Feng, Gut microbiota has the potential to improve health of menopausal women by regulating estrogen, Front. Endocrinol., 2025, 16, 1562332 CrossRef PubMed.
- X. Zheng, J. Jia, X. He, C. H. Zhou, Y. Shao, B. Li, G. Lv and S. Chen, Protective effect of the functional food containing Dendrobium officinale on menopausal syndromes based on gut microbiota and metabolomics analysis, J. Funct. Foods, 2024, 118, 106275 CrossRef.
- Y. Liu, Y. Zhou, T. Mao, Y. Huang, J. Liang, M. Zhu, P. Yao, Y. Zong, J. Lang and Y. Zhang, The relationship between menopausal syndrome and gut microbes, BMC Womens Health, 2022, 22, 437 CrossRef PubMed.
- Y. Zheng, J. Ke, J. Song, X. Li, R. Kuang, H. Wang, S. Li and Y. Li, Correlation between daily physical activity and intestinal microbiota in perimenopausal women, Sports Med. Health Sci., 2025, 7, 230–236 CrossRef PubMed.
- Y. Zong, T. Mao, P. Yao, J. Liang, Y. Lai, Z. Chen, S. Chen, L. Huang, Y. Guo, M. Zhu, J. Zhao, Y. Liu, Y. Li, K. Guo, H. Tang, X. Ke and Y. Zhou, Effects of Guizhi and Erxian Decoction on menopausal hot flashes: Insights from the gut microbiome and metabolic profiles, J. Appl. Microbiol., 2024, 135, lxae016 CrossRef PubMed.
- R. Branch, M. C. Kruger, B. Kuhn-Sherlock and B. Ilesanmi-Oyelere, The effect of pre- and probiotic supplementation on inflammatory markers in postmenopausal women, Proc. Nutr. Soc., 2024, 83, E80 CrossRef.
- M. N. T. Lambert, A. C. Thorup, E. S. S. Hansen and P. B. Jeppesen, Combined red clover isoflavones and probiotics potently reduce menopausal vasomotor symptoms, PLoS One, 2017, 12, e0176590 Search PubMed.
- Z. Liu, Q. Cao, W. Wang, B. Wang, Y. Yang, C. J. Xian, T. Li and Y. Zhai, The impact of Lactobacillus reuteri on oral and systemic health: A comprehensive review of recent research, Microorganisms, 2024, 13, 45 Search PubMed.
- X. Mo, S. Guo, D. He, Q. Cheng, Y. Yang, H. Wang, Y. Ren, L. Liu and P. Xie, Lactobacillus reuteri, DSM 17,938 ameliorates LPS-induced depression-like and anxiety-like behaviors by modulating gut microbiota and brain metabolic function, Gut Pathog., 2025, 17, 65 Search PubMed.
- R. Xie, P. Jiang, L. Lin, J. Jiang, B. Yu, J. Rao, H. Liu, W. Wei and Y. Qiao, Oral treatment with Lactobacillus reuteri attenuates depressive-like behaviors and serotonin metabolism alterations induced by chronic social defeat stress, J. Psychiatr. Res., 2020, 122, 70–78 Search PubMed.
- Y. Jiang, L. Guo, H. He, H. Chen, T. Chen, Y. Liu and W. Zhao, Lactobacillus reuteri, E9 regulates sleep disorders through its metabolite GABA, Front. Biosci., 2025, 30, 39587 Search PubMed.
- X. Ma, J. W. Shin, J. H. Cho, S. W. Han and D. H. Kim, IL-6 expression-suppressing Lactobacillus reuteri strains alleviate gut microbiota-induced anxiety and depression in mice, Lett. Appl. Microbiol., 2024, 77, ovad144 Search PubMed.
- R. A. Britton, R. Irwin, D. Quach, L. Schaefer, J. Zhang, T. Lee, N. Parameswaran and L. R. McCabe, Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model, J. Cell Physiol., 2014, 229, 1822–1830 CrossRef PubMed.
- G. Zhou, D. Chen, L. Qian, D. Song, Q. Zhang, Y. Yan, J. Lin, L. Li, G. Wan, S. Li, L. Hou, Y. He, C. Qi and W. Yuan, Efficacy and safety of Kunxinning granules in patients with menopausal syndrome: a multicenter, randomized, double-blinded, and placebo-controlled trial, Front. Pharmacol., 2025, 16, 1512110 CrossRef PubMed.
- R. Li, J. Li, Y. Liu, D. Li, L. Chu, J. Zhang, X. Luo and Y. Zhang, Serum BDNF levels are involved in the diagnosis and treatment response in patients with PD, J. Affective Disord., 2023, 327, 31–37 CrossRef PubMed.
- C. N. Epperson, D. R. Rubinow, S. Meltzer-Brody, K. M. Deligiannidis, R. Riesenberg, A. D. Krystal, K. Bankole, M. Y. Huang, H. Li, C. Brown, S. J. Kanes and R. Lasser, Effect of brexanolone on depressive symptoms, anxiety, and insomnia in women with postpartum depression: Pooled analyses from 3 double-blind, randomized, placebo-controlled clinical trials in the HUMMINGBIRD clinical program, J. Affective Disord., 2023, 320, 353–359 CrossRef PubMed.
- Y. Mi and J. Wang, Effectiveness of personalized continuous care in wound care of patients with diabetic foot ulcers, Front. Endocrinol., 2025, 16, 1612047 CrossRef PubMed.
- Y. Zhao, H. Niu and S. Liu, Effects of aerobics training on anxiety, depression and sleep quality in perimenopausal women, Front. Psychiatry, 2022, 13, 1025682 CrossRef PubMed.
- J. Ling and Y. H. Wang, Association between depressive mood and body image and menopausal symptoms and sexual function in perimenopausal women, World J. Clin. Cases, 2023, 11, 7761–7769 CrossRef PubMed.
- Y. Osuga, P. L. Torng and C. J. Jeng, Clinical studies investigating the use of leuprorelin in Asian women with endometriosis: A review, J. Obstet. Gynaecol., 2019, 39, 291–296 CrossRef PubMed.
- M. Valenzise, C. Nasso, A. Scarfone, M. Rottura, G. Cafarella, G. Pallio, G. Visalli, E. Di Prima, E. Nasso, V. Squadrito, M. Wasniewska, P. Irrera, V. Arcoraci and F. Squadrito, Leuprolide and triptorelin treatment in children with idiopathic central precocious puberty: An efficacy/tolerability comparison study, Front. Pediatr., 2023, 11, 1170025 CrossRef.
- J. H. Check, C. Wilson, K. Levine, R. Cohen and D. Corley, Improved implantation and live delivered pregnancy rates following transfer of embryos derived from donor oocytes by single injection of leuprolide in mid-luteal phase, Clin. Exp. Obstet. Gynecol., 2015, 42, 429–430 CrossRef.
- R. Baber, Treating menopausal women: Have we lost our way?, Aust. N. Z. J. Obstet. Gynaecol., 2021, 61, 493–495 CrossRef.
- V. A. Flores, L. Pal and J. E. Manson, Hormone therapy in menopause: Concepts, controversies, and approach to treatment, Endocr. Rev., 2021, 42, 720–752 CrossRef.
- S. Tiwari and V. Paramanik, Role of probiotics in depression: Connecting dots of gut-brain-axis through hypothalamic-pituitary adrenal axis and tryptophan/kynurenic pathway involving indoleamine-2,3-dioxygenase, Mol. Neurobiol., 2025, 62, 7230–7241 CrossRef CAS.
- S. Snigdha, E. Lau, J. Ademola, R. Frost and P. Patel, Effect of a novel nutraceutical combination of EstroG-100 and γ-aminobutyric acid (GABA) in attenuating symptoms of menopause in healthy adult women: A randomized double-blinded placebo-controlled study, Menopause, 2025, 32, 1036–1045 CrossRef PubMed.
- A. Garavellas, P. Mallis, E. Michalopoulos and E. Nikitos, P-798 Comprehensive evaluation of autologous Platelet-Rich Plasma infusion in ovarian rejuvenation, Hum. Reprod., 2023, 38, dead093.1104 CrossRef.
- T. M. Gouvea, E. S. L. A. Cota and A. A. Lima, Correlation of serum anti-Mullerian hormone with hormonal and environmental parameters in Brazilian climacteric women, Sci. Rep., 2022, 12, 12065 CrossRef CAS.
- T. Zhao, X. Y. Zhang, R. S. Deng, Z. Tan, G. Y. Chen and X. H. Nong, Three new unsaturated fatty acids from marine-derived fungus Aspergillus, sp. SCAU150, Nat. Prod. Res., 2022, 36, 3965–3971 CrossRef CAS PubMed.
- Y. Liu, X. Fu, B. Guan, R. Cui and W. Yang, The role and mechanism of estrogen in perimenopausal depression, Curr. Neuropharmacol., 2025, 24, 36–57 CAS.
- C. S. Fonseca, N. S. S. Aquino, G. K. N. Goncalves, L. R. Drummond, L. M. Hipolito, J. F. Silva, K. S. C. Silva, P. C. Henriques, T. E. Domingues, A. C. R. Lacerda, S. Guatimosim, C. C. Coimbra, R. E. Szawka and A. M. Reis, Norepinephrine modulation of heat dissipation in female rats lacking estrogen, J. Neuroendocrinol., 2022, 34, e13188 CrossRef CAS.
- X. Wang, N. Ding, S. D. Harlow, J. F. Randolph, Jr., E. B. Gold, C. Derby, H. M. Kravitz, G. Greendale, X. Wu, K. Ebisu, J. Schwartz and S. K. Park, Associations between exposure to air pollution and sex hormones during the menopausal transition, Sci. Total Environ., 2024, 908, 168317 CrossRef CAS.
- S. H. Lee, T. J. Lim, E. J. Yun, K. H. Kim and S. Lim, Anti-menopausal effect of soybean germ extract and Lactobacillus gasseri in the ovariectomized rat model, Nutrients, 2023, 15, 4485 CrossRef CAS PubMed.
- T. J. Yun, Y. Kim, J. J. Lee, J. Y. Park and J. H. Kim, Protective effects of probiotics against menopausal symptoms in ovariectomized mice, Food Biosci., 2024, 61, 104611 CrossRef CAS.
- M. I. Cedars, Evaluation of female fertility-AMH and ovarian reserve testing, J. Clin. Endocrinol. Metab., 2022, 107, 1510–1519 CrossRef PubMed.
- H. C. Lai, T. L. Lin, T. W. Chen, Y. L. Kuo, C. J. Chang, T. R. Wu, C. C. Shu, Y. H. Tsai, S. Swift and C. C. Lu, Gut microbiota modulates COPD pathogenesis: Role of anti-inflammatory Parabacteroides goldsteinii, lipopolysaccharide, Gut, 2022, 71, 309–321 CrossRef CAS PubMed.
- B. S. Lee, O. H. Ban, W. Y. Bang, S. A. Chae, S. Oh, C. Park, M. Lee, S. J. Kim, J. Yang and Y. H. Jung, Safety assessment of Lactobacillus reuteri IDCC 3701 based on phenotypic and genomic analysis, Ann. Microbiol., 2021, 71, 10 CrossRef CAS.
- Z. Yu, J. Chen, Y. Liu, Q. Meng, H. Liu, Q. Yao, W. Song, X. Ren and X. Chen, The role of potential probiotic strains Lactobacillus reuteri in various intestinal diseases: New roles for an old player, Front. Microbiol., 2023, 14, 1095555 CrossRef.
- S. I. Zolfaghari, M. Rabbani Khorasgani and M. Noorbakhshnia, The effects of lactobacilli (L. rhamnosus, L. reuteri, L. Plantarum) on LPS-induced memory impairment and changes in CaMKII-α and TNF-α genes expression in the hippocampus of rat, Physiol. Behav., 2021, 229, 113224 CrossRef CAS.
- X. Liu, J. He, L. Cui, Y. Ye, M. Luo, H. Xu, Y. Zhai, Z. Zhao, T. Huang, Y. Li, J. L. Wu, J. Wen, Y. Wang and T. Zhou, Limosilactobacillus reuteri-butyrate axis in depression therapy: A key pathway discovered through a novel preclinical human flora-associated animal model, Pharmacol. Res., 2025, 220, 107941 CrossRef CAS PubMed.
- W. N. Sovijit, W. E. Sovijit, S. Pu, K. Usuda, R. Inoue, G. Watanabe, H. Yamaguchi and K. Nagaoka, Ovarian progesterone suppresses depression and anxiety-like behaviors by increasing the Lactobacillus population of gut microbiota in ovariectomized mice, Neurosci. Res., 2021, 168, 76–82 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |