Oyster hydrolysate via dynamic enzymolysis ameliorates male sexual dysfunction in mice
Chunli
Xu†
,
Yuhong
Huang†
,
Junran
Yin
,
Chengfei
Liu
,
Dongyu
An
,
Shiming
Deng
* and
Guijin
Liu
 *
*
Key Laboratory of Tropical Biological Resources of Ministry of Education, School of Pharmaceutical Sciences, Hainan University, Haikou, China. E-mail: dsm701@hainanu.edu.cn; liugj@hainanu.edu.cn
First published on 11th December 2025
Abstract
Male sexual dysfunction (MSD) poses a substantial global health burden, with current pharmacotherapies often limited by adverse effects, necessitating safer, natural alternatives. This study investigates the therapeutic potential of oyster enzymatic hydrolysate (OEH) produced via an optimized dynamic enzymolysis membrane reactor (DEMR) for ameliorating MSD. The DEMR process enhanced OEH yield by 15.9% and antioxidant capacity by 42.8% compared to conventional methods, producing a hydrolysate rich in small peptides (<1000 Da), essential amino acids, taurine, and zinc with potent in vitro antioxidant and immunomodulatory activities. In an adenine/estradiol benzoate-induced MSD mouse model, DEMR-OEH intervention (1500 mg kg−1) significantly restored sexual performance parameters to levels comparable to sildenafil. This efficacy was underpinned by a multi-mechanistic action: alleviating testicular oxidative stress via increased SOD/GSH activity and reduced MDA content, improving renal function indicated by decreased BUN/sCr levels, and normalizing endocrine homeostasis through the upregulation of serum testosterone and luteinizing hormone. Bioinformatic analysis further suggested involvement of oxytocin and cGMP-PKG signaling pathways. Our findings establish DEMR-derived OEH as a promising natural therapeutic agent for MSD, functioning through integrated antioxidant, renoprotective, and endocrine-modulating pathways.
1. Introduction
Male sexual dysfunction (MSD), encompassing disruptions in arousal, erection, penetration, or ejaculation, imposes a substantial global health burden.1 Its etiology involves multiple factors, including chronic diseases,2 exposure to endocrine-disrupting chemicals,3 environmental pollution,4 modern lifestyles,5 and medication side effects,6 among others. MSD is highly prevalent, affecting approximately 52% of men aged 40–70 and an estimated 10 million individuals in the United States alone.7,8 Current therapeutic interventions include pharmacotherapy, surgery, assisted reproduction, and emerging stem cell approaches.9–11 Conventional pharmacotherapies offer a certain efficacy, such as PDE5 inhibitors (e.g., sildenafil) enhancing the NO-cGMP pathway for vasodilation,12,13 clomiphene modulating hypothalamic–pituitary–gonadal axis (HPGA) function,14 and antioxidants (e.g., vitamin C, CoQ10) mitigating oxidative stress.15,16 However, their clinical utility is often hampered by significant adverse effects, including gynecomastia, psychiatric exacerbation, thrombotic events, arrhythmias, and gastrointestinal distress.17 This underscores an urgent need for safer, naturally-derived alternatives.Oyster (Crassostrea gigas), a marine bivalve designated among Chinese first medicinal foods, represents a promising natural resource.18 Rich in proteins, taurine, polyunsaturated fatty acids, polysaccharides, and notably zinc, oyster extracts exhibit diverse physiological activities, including significant antioxidant, antibacterial, anti-fatigue, and anticancer effects, with particular relevance to reproductive health.19 Crucially, oyster-derived peptides have shown efficacy in alleviating cyclophosphamide- and retinoic acid-induced reproductive dysfunction,20,21 enhancing the synthesis of serum sex hormone and reducing oxidative stress in Leydig cells,22 and improving sexual performance in exhausted male mice.23 These findings position oyster enzymatic hydrolysates (OEH) as promising candidates for novel MSD interventions. However, conventional enzymatic production methods face key limitations, including enzyme non-reusability, high costs, inefficiency, and poor control over peptide molecular weight (MW) distribution that critically impacts bioactivity.
The dynamic enzymolysis membrane reactor (DEMR) technology offers an advanced solution, integrating enzymatic hydrolysis, continuous product separation, and enzyme recycling within a single system.24 By utilizing selective membrane permeability under a transmembrane driving force, DEMR achieves continuous separation of low-MW target peptides while retaining high-MW substrates and enzymes for reuse. This approach overcomes product inhibition, drastically enhances enzyme utilization and cost-effectiveness, accelerates reaction kinetics, increases product yield, and most importantly, allows precise control over the MW profile of the resulting hydrolysate.25,26 Consequently, DEMR holds significant potential to yield OEH with superior consistency and optimized bioactivity compared to conventional methods.
Despite the compelling bioactivity of OEH and the technological advantages of DEMR, critical research gaps impede their combined application for MSD amelioration. To address these gaps, this study employs an integrated approach: (1) optimizing DEMR parameters for OEH production from fresh oysters using single-factor and orthogonal experimental designs; (2) characterizing the resulting OEH, focusing on MW distribution, amino acid composition, peptide profile, and contents of key micronutrients (taurine and zinc); (3) assessing key in vitro bioactivities, including antioxidant and immunomodulatory potential; (4) systematically evaluating the in vivo efficacy of DEMR-derived OEH against adenine/estrogen-induced MSD in mice through analysis of sexual behavior, organ-somatic indices, testicular oxidative stress markers (superoxide dismutase, SOD; malondialdehyde, MDA; glutathione, GSH), renal functional markers (serum creatinine, sCr; blood urea nitrogen, BUN), serum sex hormone (luteinizing hormone, LH; testosterone, T) levels, and histopathological examination of testicular and renal tissues. This study provides a scientific foundation for developing OEH as a novel, natural functional ingredient for ameliorating MSD through multiple pathways.
2. Materials and methods
2.1. Materials
Fresh oysters were purchased from the local seafood market in Haikou, China. Hollow fiber membrane column was purchased from Jingbiying Environmental Technology Co., Ltd (Henan, China). Neutral protease (2 × 105 U g−1) was obtained from Xinyangshao Bio-enzyme Co., Ltd (Henan, China). 2,2-Diphenyl-1-picrylhydrazyl radical (DPPH) and 2,2′-azinobis-(3-ethylbenzothiazolin-6-sulfonic acid) (ABTS) were purchased from Yuanye Bio-technology Co., Ltd (Shanghai, China). Lipopolysaccharide (LPS) was purchased from Solarbio Science & Technology Co., Ltd (Beijing, China). Adenine was purchased from Macklin Biochemical Co., Ltd (Shanghai, China). Sildenafil citrate tablets were purchased from Qilu Pharmaceutical Co., Ltd (Shandong, China). Hydrocortisone and estradiol benzoate injections were purchased from Shanghai Quanyu Biology Technology Co., Ltd (Shanghai, China). All other chemicals and reagents used were of analytical grade and commercially available.2.2. Preparation of OEH
A modified DEMR system (Fig. 1) was employed for continuous OEH production. Operation involved the following sequential steps. (1) Substrate preparation: fresh oysters were homogenized with distilled water at a fixed ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v). The resulting homogenate was filtered through a 300-mesh nylon bag to obtain the crude protein extract filtrate. (2) Initiation of DEMR: the filtrate was transferred to the reaction vessel, and neutral protease was added at a designated enzyme-to-substrate ratio (ESR). The mixture was adjusted to target pH, equilibrated to specified temperature, and pre-incubated with stirring for 10 min. (3) Continuous hydrolysis and separation: the mixture was pumped into a hollow fiber membrane column (MW cut-off, 10 kDa) using a peristaltic pump operating at 50 rpm. Throughout this process, a second peristaltic pump (15 rpm) was employed for continuous replenishment of the reaction solution to maintain a consistent liquid level. Low-MW hydrolysate permeates continuously passed through the membrane into the product collection tank, while high-MW components and active enzymes were retained and recirculated to the reaction vessel for further hydrolysis. (4) Product collection and processing: the hydrolysate collected in the product tank was concentrated by reverse osmosis, and subsequently freeze-dried to obtain the final OEH powder.
10 (w/v). The resulting homogenate was filtered through a 300-mesh nylon bag to obtain the crude protein extract filtrate. (2) Initiation of DEMR: the filtrate was transferred to the reaction vessel, and neutral protease was added at a designated enzyme-to-substrate ratio (ESR). The mixture was adjusted to target pH, equilibrated to specified temperature, and pre-incubated with stirring for 10 min. (3) Continuous hydrolysis and separation: the mixture was pumped into a hollow fiber membrane column (MW cut-off, 10 kDa) using a peristaltic pump operating at 50 rpm. Throughout this process, a second peristaltic pump (15 rpm) was employed for continuous replenishment of the reaction solution to maintain a consistent liquid level. Low-MW hydrolysate permeates continuously passed through the membrane into the product collection tank, while high-MW components and active enzymes were retained and recirculated to the reaction vessel for further hydrolysis. (4) Product collection and processing: the hydrolysate collected in the product tank was concentrated by reverse osmosis, and subsequently freeze-dried to obtain the final OEH powder.
In this study, enzyme retention rate in the reaction vessel was monitored spectrometrically (absorbance at 238 nm wavelength, A238). Enzyme loss rate was calculated to assess DEMR integrity, and compare dynamic with static enzymolysis. For DEMR process optimization, four critical parameters were systematically evaluated using an orthogonal array design. They are hydrolysis time (2, 3, 4, 5 h), temperature (30, 40, 50, 60 °C), pH (6, 6.5, 7, 7.5), and ESR (1000, 1200, 1400, 1600 U g−1). Optimization indexes were OEH yield (Y, defined as the ratio of freeze-dried powder weight to the total weight of fresh oyster) and DPPH radical scavenging activity (RSA). A comprehensive evaluation score (CES) integrating both indexes was calculated as follows:
 | (1) |
CES maximization was the optimization criterion. Following optimization, DEMR-OEH was desalted via Sephadex G-15 gel filtration. The desalted product was then lyophilized and stored at −20 °C for further use.
2.3. Analysis of main components and MW distribution
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 55
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, v/v/v), delivered at a flow rate of 0.5 mL min−1. Samples were prepared by dissolving a precise weight of OEH in the mobile phase, and filtered through a 0.22 µm syringe filter. An aliquot (10 µL) of the filtered solution was injected. Detection was performed at a wavelength of 220 nm. MW calibration was performed using a series of purified peptide/protein standards covering the expected MW range.
0.1, v/v/v), delivered at a flow rate of 0.5 mL min−1. Samples were prepared by dissolving a precise weight of OEH in the mobile phase, and filtered through a 0.22 µm syringe filter. An aliquot (10 µL) of the filtered solution was injected. Detection was performed at a wavelength of 220 nm. MW calibration was performed using a series of purified peptide/protein standards covering the expected MW range.
Chromatographic separation was performed on a homemade analytical column (15 cm × 100 μm, 1.7 μm). The mobile phase comprised (A) water containing 2% acetonitrile and 0.1% formic acid, and (B) 80% acetonitrile containing 0.1% formic acid. The gradient elution program was set as follows: 0–1 min, 8–17% B (flow rate 1.0 μL min−1); 1–5.5 min, 17–55% B (flow rate 0.4 μL min−1); 5.5–7 min, 55–99% B (flow rate 0.7 μL min−1); 7–8 min, 99% B (flow rate 1.0 μL min−1). The total run time was 8 min.
MS analysis was conducted in positive ion mode with data-dependent acquisition (PASEF). The ion mobility range (1/K0) was set from 0.75 to 1.3 V s cm−2, with a cycle time of 0.39 s. Full MS scans were acquired over the m/z range of 180–1800. Raw data were processed using Compass HyStar software (Bruker Daltonics) and searched against the target protein database for peptide identification.
2.4. Evaluation of in vitro antioxidant activities
In vitro antioxidant capacities of OEH were assessed by measuring its DPPH RSA,30 ABTS RSA,31 and ferric reducing antioxidant power (FRAP),32 according to established methods with minor modifications.For the DPPH assay, 100 μL of OEH solution was mixed with 100 μL of 0.1 mM ethanolic DPPH solution. After 30 min incubation (room temperature, dark), absorbance was measured at 517 nm using a microplate reader. For the ABTS assay, ABTS working solution (A734 nm = 0.70) was generated by reacting ABTS stock solution with potassium persulfate firstly. Then, OEH solution was mixed with diluted ABTS working solution. After 5 min incubation (room temperature, dark), absorbance was measured at 734 nm. The scavenging activity (%) was calculated as follows.
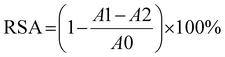 | (2) |
Where, A0, A1 and A2 represent the absorbance of blank control, sample and solution background respectively.
For FRAP assay, the FRAP working solution was freshly prepared by mixing 300 mM acetate buffer, 10 mM TPTZ solution, and 20 mM FeCl3 solution in a ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v/v). Then, OEH solution was mixed with FRAP working solution. After 10 min incubation (37 °C, dark), absorbance was measured at 593 nm. The antioxidant capacity was quantified using the standard curve generated from FeSO4 solutions and expressed as mmol Fe2+ equivalents per mL of OEH.
1 (v/v/v). Then, OEH solution was mixed with FRAP working solution. After 10 min incubation (37 °C, dark), absorbance was measured at 593 nm. The antioxidant capacity was quantified using the standard curve generated from FeSO4 solutions and expressed as mmol Fe2+ equivalents per mL of OEH.
2.5. Cell culture and cytotoxicity assay
Mouse monocyte/macrophage RAW 264.7 cells were cultured in Dulbecco's Modified Eagle's medium (DMEM) supplemented with 10% (v/v) fetal bovine serum and 1% (v/v) penicillin–streptomycin at 37 °C in a humidified 5% CO2 incubator. To assess cytotoxicity, logarithmic-phase cells were seeded into 96-well plates at a density of 1 × 105 cells per well and allowed to adhere for 24 h. Cells were then incubated for 24 h with various concentrations of OEH diluted in complete culture medium. Following treatment, the supernatant was aspirated and the wells were rinsed gently with phosphate-buffered saline to remove residual compounds. Each well subsequently received 100 μL of serum-free DMEM mixed with 10 μL of CCK-8 reagent. After incubating the plate at 37 °C for 1 h, absorbance was measured at 450 nm using a microplate reader. Cell viability was calculated according to the formula: | (3) |
2.6. Measurement of NO, TNF-α and IL-6 secretion
To assess the immunomodulatory activity of OEH, RAW 264.7 cells were seeded in 96-well plates at 1 × 105 cells per well and cultured for 24 h. Cells were subsequently co-treated with varying concentrations of OEH and 500 ng mL−1 LPS for 24 h to induce inflammatory activation. Following stimulation, cell-free supernatants were collected for parallel analysis: nitric oxide (NO) production was quantified using Griess reagent33 with absorbance measurement at 540 nm, calibrated against sodium nitrite standards; concurrently, the levels of interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) in supernatant were measured using commercial enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer's instructions, with cytokine concentrations interpolated from respective standard curves. LPS-only stimulated controls were included in all experiments, with treatments performed in triplicate wells.2.7. Animal experiments
Kunming mice (20 ± 2 g) were housed under controlled conditions (temperature 25 ± 2 °C, humidity 55 ± 5%, 12 h light/dark cycle) with ad libitum access to food and water. All experimental protocols were approved by the Animal Ethics Committee of Hainan University (approval no. HNUAUCC-2025-00390) in compliance with institutional and national guidelines. | (4) |
Serum biomarkers were further quantified through standardized assays. Reproductive hormones (LH and T) were analyzed via ELISA kits, while renal function markers (BUN and sCr) were determined using an automated biochemistry analyzer. For testicular oxidative stress evaluation, the right testis was homogenized in ice-cold saline (10%, w/v) and centrifuged at 3000 rpm (4 °C, 10 min). The supernatant was assayed for SOD activity, MDA content, and total protein concentration using manufacturer-specified commercial kits. Histopathological analysis was performed on the left testis and kidneys. Tissues were fixed in 4% paraformaldehyde, and processed through standard procedures of dehydration, clarification, paraffin embedding, and sectioning. Sections were mounted on slides, stained with hematoxylin and eosin (H&E), and examined under light microscopy for morphological alterations.
2.8. Statistical analysis
All experiments were repeated at least three times. Results were presented as mean ± standard deviation. Statistical analyses were performed using GraphPad Prism 8.0 and SPSS software. Intergroup comparisons were assessed by one-way analysis of variance (ANOVA) followed by the least significant difference test for multiple comparisons. Statistical significance was defined as p < 0.05.3. Results and discussion
3.1. DEMR process optimization
Enzyme retention within the reaction vessel is a fundamental prerequisite for the DEMR process. As shown in Fig. 2A, the DEMR system exhibited excellent enzyme retention efficiency. Throughout the 6 h hydrolysis period, the reaction vessel maintained 90.76% of its initial enzyme activity (ΔA238 < 0.2). Crucially, enzyme presence in the production tank remained negligible (A238 < 0.1), confirming effective membrane retention. Building on this validated system performance, parameters for optimal OEH production were further investigated.| Experiment number | Factors | Yield (%) | RSA (%) | CES | |||
|---|---|---|---|---|---|---|---|
| Temperature (°C) | pH | Time (h) | ESR (U g−1) | ||||
| 1 | 40 | 6 | 2 | 1200 | 5.48 | 55.95 | 0.33 |
| 2 | 40 | 6.5 | 3 | 1400 | 6.87 | 62.84 | 1.00 |
| 3 | 40 | 7 | 4 | 1600 | 6.03 | 57.82 | 0.52 |
| 4 | 50 | 6 | 3 | 1600 | 7.63 | 60.79 | 0.87 |
| 5 | 50 | 6.5 | 4 | 1200 | 7.82 | 54.56 | 0.35 |
| 6 | 50 | 7 | 2 | 1400 | 7.07 | 55.89 | 0.42 |
| 7 | 60 | 6 | 4 | 1400 | 6.25 | 51.54 | 0.00 |
| 8 | 60 | 6.5 | 2 | 1600 | 4.46 | 62.29 | 0.82 |
| 9 | 60 | 7 | 3 | 1200 | 6.62 | 60.12 | 0.75 |
| k1 | 0.62 | 0.40 | 0.52 | 0.48 | |||
| k2 | 0.54 | 0.72 | 0.87 | 0.47 | |||
| k3 | 0.52 | 0.57 | 0.29 | 0.74 | |||
| R | 0.52 | 2.25 | 4.10 | 1.85 | |||
| p-Value | 0.274 | 0.010 | 0.003 | 0.049 | |||
| Order of influence | Time > pH > ESR > temperature | ||||||
Based on the analysis of factor levels (k-values in Table 1), the optimal conditions for maximizing CES were determined as follows: temperature 40 °C, pH 6.5, hydrolysis time 3 h, and ESR 1400 U g−1. To validate the effectiveness of these conditions, three independent replicate experiments were conducted. The validation results demonstrated that DMER under optimal conditions produced OEH with a yield of 6.92 ± 0.10% and DPPH-RSA of 62.24 ± 1.42%. In contrast, static enzymolysis under the same conditions yielded only 5.97 ± 0.08% OEH and 43.58 ± 1.48% DPPH-RSA. Furthermore, analysis of the degree of hydrolysis (DH, calculated from the ratio of free amino nitrogen to total nitrogen) indicated that static enzymolysis required 5 h to achieve final hydrolysis, whereas DMER completed within 3 h (Fig. 2F), indicating a higher reaction rate under dynamic conditions. These results collectively demonstrate the effectiveness and superiority of DMER for enhancing OEH production and its antioxidant activity.
3.2. Comprehensive characterization of OEH main components
To elucidate the material basis for in vivo efficacy against MSD, we systematically characterized the principal physicochemical and bioactive components of OEH. The analysis focused on its molecular weight distribution, amino acid composition, peptide profile, and key micronutrients (taurine and zinc), all of which are critically implicated in reproductive health pathways.Further analysis of the OEH identified a comprehensive profile of 20 free amino acids, as detailed in Table 2. Alanine and tyrosine were the most abundant free amino acids, with contents of 16.86 ± 2.01 μg mg−1 and 16.85 ± 1.28 μg mg−1, respectively. The hydrolysate contained eight essential amino acids, which constituted 44.38% of the total free amino acids. Branched-chain amino acids represented 25.95%, and hydrophobic amino acids accounted for a substantial 55.73% of the total free amino acids.
| Amino acid | Content (μg mg−1) | Amino acid | Content (μg mg−1) |
|---|---|---|---|
| Glycine | 14.15 ± 0.800 | Glutamine | 2.53 ± 0.270 |
| Alanine | 16.86 ± 2.010 | Lysine | 4.73 ± 0.139 |
| γ-Aminobutyric acid | 0.11 ± 0.017 | Glutamic acid | 11.88 ± 1.003 |
| Serine | 0.10 ± 0.001 | Methionine | 5.05 ± 0.857 |
| Proline | 7.09 ± 0.665 | Histidine | 3.73 ± 0.434 |
| Valine | 11.55 ± 1.011 | Phenylalanine | 10.88 ± 1.030 |
| Threonine | 2.40 ± 0.103 | Arginine | 9.34 ± 0.330 |
| Isoleucine | 8.19 ± 0.326 | Tyrosine | 16.85 ± 1.277 |
| Leucine | 12.76 ± 0.457 | Tryptophan | 1.44 ± 0.044 |
| Ornithine | 5.32 ± 0.158 | Aspartic acid | 0.42 ± 0.125 |
| Rank | Peptide sequence | Length | Molecular mass (Da) | Observed mass (m/z) | Peak area | Bit scores |
|---|---|---|---|---|---|---|
| 1 | FNDAQRQATKDAGTIAG | 17 | 1762.86 | 882.4442 | 1520 | 49.8 |
| 2 | ATVGDVNTDRPGMLD | 15 | 1559.725 | 780.8753 | 3170 | 49.4 |
| 3 | ATVGDVNTDRPGM | 13 | 1331.614 | 666.8229 | 7730 | 43.9 |
| 4 | IIANDQGNRITPSY | 14 | 1560.79 | 781.4123 | 557 | 43.9 |
| 5 | LSMANAGKDTNGSQF | 15 | 1539.699 | 770.8686 | 613 | 43.9 |
| 6 | VTIAQGGVLPNIQ | 13 | 1308.74 | 655.3818 | 6100 | 42.6 |
| 7 | QVHPDTGVSSKAM | 13 | 1355.65 | 678.8417 | 788 | 40.5 |
| 8 | VEPSDTIENVKAK | 13 | 1428.746 | 715.3857 | 1000 | 40.1 |
| 9 | VGDVNTDRPGM | 11 | 1159.529 | 580.7772 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
38 |
| 10 | LLPGELAKHA | 10 | 1047.608 | 524.8154 | 1390 | 33.3 |
| 11 | IQDKEGIPPDQQR | 13 | 1522.774 | 762.4022 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
31.6 |
| 12 | DSGDGVSHTVPIYEG | 15 | 1531.679 | 766.8572 | 1390 | 31.2 |
| 13 | LDSGDGVSHTVPIYEG | 16 | 1644.763 | 823.3994 | 512 | 31.2 |
| 14 | DSGDGVTHTVPIY | 13 | 1359.631 | 680.83 | 913 | 28.6 |
| 15 | LTEAPLNPKANRE | 13 | 1451.773 | 726.9016 | 2360 | 27.8 |
| 16 | FAGDDAPRAVFPS | 13 | 1348.641 | 675.3348 | 2390 | 27.4 |
| 17 | LVIDNGSGMCK | 11 | 1135.537 | 568.7795 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
24.4 |
| 18 | IVTNWDDMEK | 10 | 1249.565 | 625.797 | 86![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
23.5 |
| 19 | LRVAPEEHPVL | 11 | 1258.703 | 630.3652 | 1240 | 22.7 |
| 20 | FSVVPSPK | 8 | 859.4803 | 430.7474 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
21.8 |
| 21 | LAQDEQDSVRL | 11 | 1272.631 | 637.3268 | 392 | 19.3 |
Structural characterization further revealed that the identified peptides are predominantly short-chain, with lengths concentrated between 9 and 13 amino acid residues (Fig. 3B). Their MW largely fell below 3 kDa, and isoelectric points (pI) clustered in the 4–7 range (Fig. 3C), features consistent with high bioavailability and commonly observed in antioxidant peptides.41 Grand average of hydropathicity (GRAVY) values exhibited a unimodal distribution centered at approximately −0.7 (Fig. 3D), indicating pronounced hydrophilicity, which predicts excellent solubility and low aggregation propensity. N- and C-terminal residue analysis uncovered distinct compositional biases (Fig. 3E). Hydrophobic amino acids, notably isoleucine, leucine, and valine, were highly enriched at the N-terminus, while leucine and methionine were prominent at the C-terminus.
Critically, several sequences correspond to peptides with documented biological activities relevant to MSD. For instance, FSVVPSPK is reported to attenuate ROS accumulation by promoting Nrf2-mediated HO-1 upregulation,42 and IVTNWDDMEK protects vascular endothelium from oxidative apoptosis via the HO-1/Nrf2 pathway.43 The peptide IQDKEGIPPDQQR has demonstrated anti-inflammatory activity by suppressing LPS-induced responses.44 Furthermore, the frequent occurrence of histidine, arginine, and lysine can facilitate radical quenching, NO synthesis, and vascular relaxation, which strengthens the premise for their role in mitigating MSD.
ICP-OES determined the zinc content to be 181.06 ± 2.15 mg kg−1, a high level characteristic of oyster-derived materials.39 Zinc is integral to male reproductive health, serving as a cofactor for testosterone biosynthesis, providing antioxidant defense to reproductive tissues, and being crucial for normative spermatogenesis and sperm function.48
Therefore, OEH is a complex mixture system comprising three pivotal classes of bioactive constituents: (1) a spectrum of low-MW peptides and free amino acids with structural predispositions for high bioavailability, and antioxidant capacity; (2) a diverse repertoire of specific bioactive peptides with documented roles in oxidative stress mitigation, immunomodulation, and vascular function; and (3) nutritionally significant levels of taurine and zinc, which are fundamental to endocrine function and reproductive tissue integrity. This multi-component profile provides a compelling, synergistic material basis for the efficacy of OEH in ameliorating MSD, which stems from concerted actions through interconnected pathways, such as oxidative stress reduction, immunomodulation, endocrine regulation, and vascular protection.
3.3. In vitro antioxidant activities of OEH
The radical scavenging capacity and reducing power of OEH were systematically evaluated to characterize its antioxidant potential, and the results were shown in Fig. 3G. OEH displayed a significant dose-dependent capacity to scavenge DPPH radicals. Scavenging efficiency rose from 22.77 ± 2.68% at 0.25 mg mL−1 to 75.39 ± 1.40% at 4 mg mL−1, yielding a half-maximal inhibitory concentration (IC50) of 1.67 mg mL−1. Similarly, OEH exhibited strong ABTS-RSA, where the scavenging rate rose from 29.83 ± 2.56% to 87.46 ± 2.19% over the same concentration range, and an IC50 of 0.78 mg mL−1 was obtained. Moreover, OEH demonstrated notable ferric ion reducing power, with reduction capacity increasing from 0.34 ± 0.03 to 0.79 ± 0.02 mmol Fe2+ equivalents per mL between 0.25 mg mL−1 and 4 mg mL−1. This dose-dependent behavior highlights electron-donating ability of OEH, a key characteristic of antioxidant agents. These results clearly indicate potent concentration-dependent antioxidant activity in vitro, consistent with its composition rich in low-MW antioxidant peptides and free amino acids.3.4. Cell cytotoxicity and immunomodulation of OEH
The cytotoxicity of OEH on RAW 264.7 macrophages was evaluated using the CCK-8 assay. As shown in Fig. 4A, OEH exhibited no significant cytotoxicity (p > 0.05) at concentrations ranging from 250 to 2000 μg mL−1, with cell viability consistently maintained above 95% compared to the untreated control. These findings confirm the high cytocompatibility of OEH and support the use of these concentrations in subsequent immunomodulatory assays. | ||
| Fig. 4 Effect of OEH treatment on (A) RAW 264.7 viability, (B) NO production, (C) IL-6 secretion, (D) TNF-α secretion. Dissimilar letters denote significant differences (p < 0.05). | ||
NO is a critical effector molecule released by activated macrophages, which disrupts pathogens through membrane damage and metabolic inhibition.49 OEH treatment induced a dose-dependent NO production (Fig. 4B), increasing from 14.38 ± 2.63 µM at 250 μg mL−1 to 32.74 ± 2.26 µM at 2000 μg mL−1. Notably, OEH treatment at concentrations exceeding 1000 µg mL−1 triggered significantly stronger NO production (p < 0.05) than that induced by LPS alone (21.72 ± 3.71 µM), demonstrating its potent macrophage-activating effect. To further investigate the immunomodulatory function of OEH, the secretion of key pro-inflammatory cytokines was evaluated. OEH treatment resulted in a significant and concentration-dependent upregulated of both IL-6 (Fig. 4C) and TNF-α (Fig. 4D) compared to the control group (p < 0.05), reaching levels comparable to those stimulated by LPS. This concerted enhancement of NO production and pro-inflammatory cytokine secretion (IL-6, TNF-α) demonstrates the significant immunostimulatory potential of OEH in macrophage activation.
3.5. MSD model selection
Three distinct murine models were evaluated for their suitability in studying OEH interventions against MSD, as outlined in Fig. 5A. Model 1 induced severe systemic toxicity, featuring persistent writhing, acute weight loss, and progression to hindlimb paralysis. Model 2 showed progressive deterioration, including early tremors, hypokinesis, bilateral ocular lesions, weight loss, and terminal intestinal infarction. In contrast, model 3 exhibited progressive but stable pathology (polyuria, polydipsia, hunched posture, and piloerection) without mortality. Although all models displayed elevated renal impairment markers (BUN and sCr, Fig. 5B and C), model 3 resulted in comparatively milder histopathological damage. Thus, model 3 was selected for subsequent therapeutic intervention studies.3.6. Effects of OEH on food intake and body weight
Based on the longitudinal monitoring over 30 days, distinct profiles in both food intake and body weight were observed across experimental groups (Fig. 6). The control group maintained stable daily food consumption and exhibited a consistent increase in body weight, reflecting normal growth. In contrast, the model group displayed a pronounced reduction in food intake, coupled with a severe decline in body weight, consistent with MSD-induced metabolic stress. Interventions with OEH-L, OEH-H, and SDF effectively restored both food intake and body weight, indicating systemic amelioration of dysfunction. These results suggest that OEH, particularly at higher doses, helps maintain metabolic homeostasis and overall health, thereby supporting the improvement of MSD.3.7. Restorative effects of OEH on sexual behavior parameters
OEH efficacy in restoring MSD was quantified through four validated behavioral parameters (Fig. 7). Compared with the control group, the model group exhibited prolonged ML (368.75 ± 52.80 s vs. 139.25 ± 36.60 s) and IL (394.63 ± 58.79 s vs. 182.38 ± 40.74 s), along with decreased MF (2.50 ± 1.41 s vs. 13.25 ± 3.11 s) and IF (1.13 ± 0.99 s vs. 10.00 ± 2.51 s) significantly. OEH intervention dose-dependently reversed these deficits. OEH-H reduced ML to 172.38 ± 38.24 s, elevated MF to 8.25 ± 2.96 s, shortened IL to 183.75 ± 38.80 s, and increased IF to 6.88 ± 2.90 s. Notably, OEH-H achieved statistical equivalence (p > 0.05) to the positive control (SDF) in all parameters. These results suggest OEH as a potent mitigator of MSD, functionally comparable to first-line therapeutics. | ||
| Fig. 7 Effects of OEH on sexual behavior parameters: (A) ML, (B) MF, (C) IL, and (D) IF. Dissimilar letters denote significant differences (p < 0.05). | ||
3.8. Mitigation effects of OEH on organ pathophysiological alterations
Renal and testicular structural integrity is essential for endocrine homeostasis and male reproductive function. Organ coefficient analysis (Table 4) revealed severe pathological alterations in the model group versus controls: kidney coefficient increased 43.27% (2.45 ± 0.46% vs. 1.71 ± 0.31%; p < 0.05); testis coefficient decreased 65.59% (0.32 ± 0.13% vs. 0.93 ± 0.09%; p < 0.05). Macroscopic examination (Fig. 8A) demonstrated concordant pathology, with model kidneys exhibiting diffuse gray-white lesions and testicular volume reduction. H&E histopathology (Fig. 8B and C) further confirmed severe tubular dilation with cast formation (red arrows) in renal tissue, and disrupted spermatogenic cell layers (green arrows) in testicular tissue, indicating multifunctional organ damage. OEH intervention dose-dependently reversed these changes. OEH-H significantly reduced kidney hypertrophy by 21.63% (1.92 ± 0.42%; p < 0.05 vs. model); increased testis mass 28.13% (0.41 ± 0.17%; p < 0.05 vs. model), with histological improvements comparable to SDF treatment.| Organ | Control | Model | OEH-L | OEH-H | SDF |
|---|---|---|---|---|---|
| **Values with different superscript letters differ significantly (p < 0.05). | |||||
| Heart | 0.50 ± 0.04a | 0.43 ± 0.05ab | 0.40 ± 0.05b | 0.40 ± 0.05b | 0.43 ± 0.06ab |
| Liver | 5.79 ± 1.15a | 4.39 ± 0.17b | 4.99 ± 0.65ab | 4.30 ± 0.58b | 4.22 ± 1.51b |
| Spleen | 0.33 ± 0.10a | 0.22 ± 0.08a | 0.23 ± 0.09a | 0.25 ± 0.07a | 0.28 ± 0.04a |
| Kidney | 1.71 ± 0.31a | 2.45 ± 0.46b | 2.13 ± 0.39a | 1.91 ± 0.42a | 1.92 ± 0.28a |
| Testis | 0.93 ± 0.09a | 0.32 ± 0.13b | 0.32 ± 0.10b | 0.41 ± 0.17b | 0.41 ± 0.08b |
3.9. Amelioration effects of OEH on testicular oxidative stress
Pathological oxidative stress in testicular tissue, characterized by disrupted redox balance, critically contributes to MSD. On the basis of its in vitro antioxidant capacity, OEH efficacy against adenine-estrogen-induced oxidative damage in murine testicular tissue was further assessed. As shown in Fig. 9A–C, the model group exhibited significant redox dysregulation compared to controls. Specifically, MDA content increased by 167.74% (3.32 ± 0.37 nmol mgprot−1vs. 1.24 ± 0.14 nmol mgprot−1; p < 0.05), while the activities of key antioxidant enzymes were markedly suppressed: SOD decreased by 42.81% (85.12 ± 7.19 U mgprot−1vs. 148.85 ± 17.88 U mgprot−1; p < 0.05) and GSH level decreased by 49.16% (0.91 ± 0.19 μmol mL−1vs. 1.79 ± 0.23 μmol mL−1; p < 0.05).Intervention with OEH dose-dependently reversed these abnormalities. Although OEH-L failed to significantly modulate either parameter (p > 0.05), OEH-H profoundly reduced MDA to 2.03 ± 0.20 nmol mgprot−1, and restored SOD and GSH levels to 104.07 ± 15.63 U mgprot−1 and 1.46 ± 0.29 μmol mL−1, respectively. Notably, OEH-H achieved therapeutic equivalence to SDF (p > 0.05 for all parameters). The SOD restoration demonstrates reactivation of endogenous antioxidant systems, while MDA reduction confirms attenuation of membrane lipid peroxidation. The concurrent elevation of GSH suggests potential activation of the Nrf2-Keap1-HO-1 pathway, a central regulatory axis for cellular antioxidant responses.50 Collectively, these results demonstrate that OEH functions as a redox-targeted therapeutic agent capable of ameliorating testicular oxidative stress through a multi-faceted mechanism.
3.10. Attenuation effects of OEH on renal dysfunction biomarkers
As important biomarkers for evaluating renal function, sCr and BUN reflect the glomerular filtration rate and excretory capacity. Impairment of renal function leads to the accumulation of sCr and BUN, which may directly or indirectly affect sexual function.23 As shown in Fig. 9D and E, the model group exhibited a significant increase in BUN levels, reaching 43.07 ± 12.13 mg dL−1 compared to the control group (19.90 ± 4.04 mg dL−1; p < 0.05). Similarly, sCr levels were significantly elevated to 82.07 ± 19.20 μM versus the control (28.56 ± 7.46 μM; p < 0.05). After intervention with OEH and SDF, there were some reductions in the levels of BUN and sCr. These results demonstrate the renoprotective effects of OEH, underscoring its therapeutic potential in MSD.3.11. Modulation effects of OEH on sex hormone levels
T and LH are critical regulators of testicular spermatogenesis, sexual function, and reproductive endocrine homeostasis. Sex hormone analysis (Fig. 9F and G) demonstrated severe endocrine disruption in the model group: LH decreased 48.36% versus control (2781.90 ± 116.30 pg mL−1vs. 5387.08 ± 401.08 pg mL−1; p < 0.05); T declined 54.57% (5.97 ± 0.47 nmol mL−1vs. 13.14 ± 0.70 nmol mL−1; p < 0.05). Intervention with OEH and SDF resulted in varying degrees of increase in LH and T levels in male mice. The dose-dependent LH elevation correlates with prior observations of spermatogenic recovery, suggesting pituitary axis reactivation. Contrastingly, T normalization implies direct testicular steroidogenesis modulation, potentially through oxidative stress mitigation.3.12. Mechanisms of OEH in ameliorating MSD
As summarized in Fig. 10, OEH significantly ameliorates MSD in mice through a multi-targeted mechanism, synergistically improving biochemical, physiological, and behavioral outcomes. The multifaceted bioactivities of OEH are attributed to its specific composition of bioactive peptides, free amino acids, taurine, and zinc. Key bioactive amino acids (e.g., lysine, tyrosine, and histidine) support critical physiological processes such as testosterone secretion, sperm motility, neuro-regulation, and vascular engorgement.51,52 Moreover, arginine serves as a precursor for NO, which plays a critical role in penile erection through GC-cGMP-PKG pathway.53 Additionally, the high bioavailability of low-MW peptides in OEH promotes tissue repair and helps alleviate fatigue, thereby supporting overall physiological recovery.18 The presence of taurine further contributes to neuroregulation and endocrine stability, while zinc acts as an essential cofactor for testosterone synthesis and antioxidant defense.Oxidative stress plays a critical role in impaired sperm function and testicular damage.54,55 OEH demonstrated potent concentration-dependent antioxidant activity, and OEH treatment markedly reduced MDA levels and enhanced SOD activity, along with restoring GSH levels indicating improved redox homeostasis. As reported, the transcription factor Nrf2 plays a pivotal role in regulating oxidative homeostasis, primarily through its interaction with Keap1.56 Modulation of testicular redox balance via the Keap1/Nrf2 pathway has been shown to ameliorate MSD.57 Furthermore, oxidative stress can activate apoptosis-related pathways such as JNK and PERK/CHOP,58 while concurrently inhibiting the PI3K/Akt pathway.59 We propose that OEH may activate the Nrf2-Keap1 pathway, suppress pro-apoptotic signals (JNK and PERK/CHOP), and promote PI3K/Akt-mediated cell survival, thereby protecting testicular tissue from oxidative injury. The coordinated action of antioxidant peptides, taurine, and zinc in OEH underpins this comprehensive protective effect against oxidative and apoptotic damage.
OEH treatment also improved renal function, as evidenced by decreased BUN and sCr levels. Elevated BUN and sCr are well-established biomarkers associated with acute kidney injury. Renal dysfunction is known to exacerbate MSD through multiple mechanisms, including hormonal imbalance, vascular injury, and systemic toxin accumulation.60 Previous studies have demonstrated that inhibition of NF-κB can alleviate apoptosis and inflammatory responses, thereby providing renal protection.61 Thus, the beneficial effects of OEH on MSD may be partially attributed to NF-κB inhibition and the consequent improvement of renal function. The anti-inflammatory and renoprotective properties of taurine and certain bioactive peptides in OEH probably contribute to this aspect of its efficacy.
Furthermore, OEH treatment was found to modulate the HPGA, a central regulatory system in male reproductive endocrinology. This effect was demonstrated by elevated T and LH levels. Within HPGA, LH is secreted by the anterior pituitary and acts upon testicular Leydig cells to promote the synthesis and secretion of T. T plays a critical role in spermatogenesis, the development of sexual organs, and the maintenance of male secondary sexual characteristics.62–64 The availability of key amino acids and zinc in OEH provides essential nutritional and catalytic support for this endocrine normalization.
3.13. Bioinformatic exploration of OEH action pathways
To complement the experimentally elucidated mechanisms and further delineate the putative signaling networks modulated by the peptide fraction of OEH, we conducted a bioinformatic analysis. It is noteworthy that this predictive approach primarily identifies direct, peptide-associated functions and may not fully encompass the integrated in vivo effects contributed by non-peptide constituents (e.g., taurine and zinc) or complex secondary physiological adaptations.Gene Ontology (GO) enrichment analysis revealed significant overrepresentation of key functional categories among OEH-associated peptides (Fig. 11A). Molecular functions (MF) such as antioxidant activity, biological processes (BP) including response to stimulus and detoxification were prominently enriched, and cellular component (CC) including cellular anatomical entity and protein-containing complex. This functional profile provides corroborative evidence at the molecular level demonstrated efficacy in OEH attenuating systemic and testicular oxidative stress. Concurrently, significant enrichment in transporter activity, cytoskeletal motor activity, and reproductive process implies a direct role for these peptides in fundamental physiological mechanisms underpinning erectile function, spanning molecular transport to tissue contractility.
 | ||
| Fig. 11 Bioinformatic analysis of OEH peptide functions and associated pathways. (A) GO functional classification across BP, MF, and CC categories. (B) KEGG pathway enrichment analysis. | ||
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis further identified significant associations with two critical signaling cascades pertinent to MSD pathophysiology (Fig. 11B). The oxytocin signaling pathway was prominently enriched (implicating 19 proteins). This finding suggests a potential role for OEH peptides in central neuromodulation of sexual arousal and pro-social sexual behavior, constituting a complementary mechanism to the observed peripheral endocrine normalization.65 The cGMP-PKG signaling pathway was also significantly modulated (involving 22 proteins). This provides a putative mechanistic link between the precursor role of arginine, NO production, and the downstream relaxation of corpus cavernosum smooth muscle essential for penile erection.53
Collectively, this bioinformatic exploration proposes a “center-periphery” axis for the bioactivity of OEH peptides. The model suggests that OEH ameliorates MSD via concerted actions on central arousal pathways (e.g., oxytocin) and peripheral erectile executors (e.g., cGMP-PKG). While this peptide-centric perspective does not encompass the full scope of mechanisms validated in our study, it offers a sophisticated, predicted molecular framework that is coherent with and extends the functional efficacy observed in vivo.
3.14. Practical feasibility and translational potential of OEH
The translational potential of OEH was assessed through dosage feasibility, formulation compatibility, stability, and bioavailability. The human equivalent dose (HED) calculated from the efficacious high-dose regimen in mice (1500 mg kg−1) corresponds to a daily intake of 8.5 g for a 70 kg adult, establishing a foundation for clinical translation.Oyster-derived peptides exhibit excellent formulation versatility, having been successfully incorporated into compressed candies meeting pharmacopeial standards,39 and combined with potato flour for specialized nutritional products,66 confirming feasible delivery via conventional supplements. Our preliminary stability assessment under refrigerated conditions (4 °C, 45 days) confirmed that OEH maintains critical quality attributes, showing negligible moisture absorption and preserved antioxidant capacity (DPPH-RSA: 75.39 ± 1.40% vs. 73.49 ± 2.77% after storage), indicating sufficient stability for practical storage and distribution.
The bioavailability of active components in OEH underlies its efficacy. The low-MW profile and free amino acids generated through dynamic enzymolysis ensure efficient systemic delivery of functional constituents. Specifically, bioavailable arginine supports NO-dependent vascular function, while antioxidant peptides mitigate testicular oxidative stress. Notably, zinc-binding peptides form stable complexes that enhance zinc absorption via a dual-pathway mechanism,67 providing superior bioavailability of this essential element for testosterone synthesis and spermatogenesis.68 Therefore, the achievable dosage, formulation adaptability, demonstrated stability, and enhanced bioavailability collectively establish OEH as a viable functional foods or dietary supplements for MSD management, effectively bridging experimental findings with practical applications.
4. Conclusion
This study successfully establishes an advanced DEMR process for the efficient production of OEH with optimized bioactivity, and systematically validates its profound efficacy in alleviating MSD in mice. Compared to conventional static enzymolysis, the optimized DEMR process enhanced the OEH yield by 15.9% and its DPPH-RSA by 42.8%. Component analysis indicates that OEH constitutes a complex mixture rich in low-MW peptides (94.66% <1000 Da), essential amino acids (44.38%), along with substantial taurine (16.48%) and zinc (181.06 ± 2.15 mg kg−1), exhibiting pronounced antioxidant and immunomodulatory activity in vitro.In an adenine/estradiol benzoate-induced MSD mouse model, intervention with high-dose DEMR-OEH (1500 mg kg−1) significantly restored sexual performance parameters, reducing ML from 368.75 ± 52.80 s to 172.38 ± 38.24 s and increasing IF from 1.13 ± 0.99 s to 6.88 ± 2.90 s, achieving efficacy comparable to sildenafil. This efficacy was underpinned by a multi-mechanistic action: alleviating testicular oxidative stress via enhancing SOD and GSH activity, along with reducing MDA content; improving renal function indicated by decreasing BUN and sCr levels; and normalizing endocrine homeostasis through the upregulation of T and LH. Bioinformatic analysis further suggested potential involvement of oxytocin and cGMP-PKG signaling pathways, supporting a central-peripheral synergistic mode of action.
As a natural hydrolysate derived from widely consumed oysters, OEH functions as a multi-target nutraceutical rather than a single-molecule pharmaceutical. The human equivalent dose of 8.5 g daily for a 70 kg adult is achievable through dietary supplements or functional foods, supported by its demonstrated formulation versatility, storage stability, and enhanced bioavailability of active components. This study highlights DEMR technology as a superior platform for generating consistent bioactive hydrolysates and positions OEH as a promising natural therapeutic candidate for MSD. These findings provide a robust scientific foundation for the further development of OEH as a functional nutraceutical or therapeutic agent in the management of male reproductive health, with future research directed towards elucidating the precise signaling pathway and identifying the specific bioactive peptide sequences responsible for the observed effects.
Author contributions
Chunli Xu: conceptualization, methodology, writing (original draft). Yuhong Huang: formal analysis, methodology, investigation, data curation. Junran Yin: validation, data curation. Chengfei Liu: formal analysis, data curation. Dongyu An: formal analysis, investigation. Shiming Deng: supervision, resources. Guijin Liu: conceptualization, writing (review & editing), funding acquisition.Conflicts of interest
The authors declare no conflict of interest.Data availability
The authors declare that the data supporting the findings of this study are available within the paper. Should any raw data files be needed in another format, they are available from the corresponding author upon reasonable request.Acknowledgements
Financial support from the National Natural Science Foundation of China (no. 82460699) is greatly appreciated.References
- L. Chen, G. Shi, D. Huang, Y. Li, C. Ma, M. Shi, B. Su and G. Shi, Male Sexual Dysfunction: A Review of Literature on Its Pathological Mechanisms, Potential Risk Factors, and Herbal Drug Intervention, Biomed. Pharmacother., 2019, 112, 108585 CrossRef.
- M. L. Eisenberg, S. Li, M. R. Cullen and L. C. Baker, Increased Risk of Incident Chronic Medical Conditions in Infertile Men: Analysis of United States Claims Data, Fertil. Steril., 2016, 105, 629–636 CrossRef.
- A. Giwercmanz, L. Rylander and Y. L. Giwercman, Influence of Endocrine Disruptors on Human Male Fertility, Reprod. BioMed. Online, 2007, 15, 633–642 CrossRef PubMed.
- H. Li, X. R. Wang, Y. F. Hu, Y. W. Xiong, H. L. Zhu, Y. C. Huang and H. Wang, Advances in Immunology of Male Reproductive Toxicity Induced by Common Environmental Pollutants, Environ. Int., 2024, 190, 108898 CrossRef.
- A. Agarwal, S. Baskaran, N. Parekh, C. L. Cho, R. Henkel, S. Vij, M. Arafa, M. K. Panner Selvam and R. Shah, Male Infertility, Lancet, 2021, 397, 319–333 CrossRef PubMed.
- D. Velez and S. Ohlander, Medical Therapies Causing Iatrogenic Male Infertility, Fertil. Steril., 2021, 116, 618–624 CrossRef PubMed.
- H. M. Tan, S. F. Tong and C. C. K. Ho, Men's Health: Sexual Dysfunction, Physical and Psychological Health—Is There a Link?, J. Sex. Med., 2012, 9, 663–671 CrossRef.
- P. F. Schnatz, S. K. Whitehurst and D. M. O'Sullivan, Sexual Dysfunction, Depression, and Anxiety Among Patients of an Inner-City Menopause Clinic, J. Women's Health, 2010, 19, 1843–1849 CrossRef.
- R. F. Adriansyah, R. Margiana, S. Supardi and P. Narulita, Current Progress in Stem Cell Therapy for Male Infertility, Stem Cell Rev. Rep., 2023, 19, 2073–2093 CrossRef.
- M. Assidi, Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics, Cells, 2022, 11, 1711 CrossRef CAS PubMed.
- A. Kaltsas, F. Dimitriadis, D. Zachariou, A. Zikopoulos, E. N. Symeonidis, E. Markou, D. M. B. Tien, A. Takenaka, N. Sofikitis and A. Zachariou, From Diagnosis to Treatment: Comprehensive Care by Reproductive Urologists in Assisted Reproductive Technology, Medicina, 2023, 59, 1835 CrossRef PubMed.
- G. Salvio, M. Cutini, A. Ciarloni, L. Giovannini, M. Perrone and G. Balercia, Coenzyme Q10 and Male Infertility: A Systematic Review, Antioxidants, 2021, 10, 874 CrossRef CAS.
- X. Zhou, H. Shi, S. Zhu, H. Wang and S. Sun, Effects of Vitamin E and Vitamin C on Male Infertility: A Meta-Analysis, Int. Urol. Nephrol., 2022, 54, 1793–1805 CrossRef PubMed.
- M. Huijben, R. L. N. Huijsmans, M. T. W. T. Lock, V. F. De Kemp, L. M. O. De Kort and J. H. M. K. Van Breda, Clomiphene Citrate for Male Infertility: A Systematic Review and Meta–analysis, Andrology, 2023, 11, 987–996 CrossRef.
- H. Glossmann, G. Petrischor and G. Bartsch, Molecular Mechanisms of the Effects of Sildenafil (VIAGRA®), Exp. Gerontol., 1999, 34, 305–318 CrossRef PubMed.
- N. D. Scherzer, T. V. Le and W. J. G. Hellstrom, Sildenafil's Impact on Male Infertility: What Has Changed in 20 Years?, Int. J. Impotence Res., 2019, 31, 71–73 CrossRef.
- K. M. Wheeler, D. Sharma, P. K. Kavoussi, R. P. Smith and R. Costabile, Clomiphene Citrate for the Treatment of Hypogonadism, Sex. Med. Rev., 2019, 7, 272–276 CrossRef.
- M. Xiao, L. Lin, H. Chen, X. Ge, Y. Huang, Z. Zheng, S. Li, Y. Pan, B. Liu and F. Zeng, Anti-Fatigue Property of the Oyster Polypeptide Fraction and Its Effect on Gut Microbiota in Mice, Food Funct., 2020, 11, 8659–8669 RSC.
- J. Miao, W. Liao, M. Kang, Y. Jia, Q. Wang, S. Duan, S. Xiao, Y. Cao and H. Ji, Anti-Fatigue and Anti-Oxidant Activities of Oyster (Ostrea Rivularis) Hydrolysate Prepared by Compound Protease, Food Funct., 2018, 9, 6577–6585 RSC.
- X. Zhang, Z. Peng, H. Zheng, C. Zhang, H. Lin and X. Qin, The Potential Protective Effect and Possible Mechanism of Peptides from Oyster (Crassostrea Hongkongensis) Hydrolysate on Triptolide-Induced Testis Injury in Male Mice, Mar. Drugs, 2021, 19, 566 CrossRef PubMed.
- M. Li, M. Zhou, Y. Wei, F. Jia, Y. Yan, R. Zhang, M. Cai and R. Gu, The Beneficial Effect of Oyster Peptides and Oyster Powder on Cyclophosphamide–induced Reproductive Impairment in Male Rats: A Comparative Study, J. Food Biochem., 2020, 44, 13468 Search PubMed.
- Y. Yan, M. Li, Y. Wei, F. Jia, Y. Zheng, G. Tao and F. Xiong, Oyster–derived Dipeptides RI, IR, and VR Promote Testosterone Synthesis by Reducing Oxidative Stress in TM3 Cells, Food Sci. Nutr., 2023, 11, 6470–6482 CrossRef.
- X. Luo, W. Liu, H. Zhong, Y. Yan, F. Feng and M. Zhao, Synergistic Effect of Combined Oyster Peptide and Ginseng Extracts on Anti-Exercise-Fatigue and Promotion of Sexual Interest Activity in Male ICR Mice, J. Funct. Foods, 2021, 86, 104700 CrossRef.
- S. Baldassarre, N. Babbar, S. Van Roy, W. Dejonghe, M. Maesen, S. Sforza and K. Elst, Continuous Production of Pectic Oligosaccharides from Onion Skins with an Enzyme Membrane Reactor, Food Chem., 2018, 267, 101–110 CrossRef PubMed.
- A. B. Sitanggang, A. Drews and M. Kraume, Enzymatic Membrane Reactors: Designs, Applications, Limitations and Outlook, Chem. Eng. Process., 2022, 180, 108729 CrossRef.
- Z. Su, J. Luo, X. Li and M. Pinelo, Enzyme Membrane Reactors for Production of Oligosaccharides: A Review on the Interdependence between Enzyme Reaction and Membrane Separation, Sep. Purif. Technol., 2020, 243, 116840 CrossRef.
- R. Fuertig, A. Ceci, S. M. Camus, E. Bezard, A. H. Luippold and B. Hengerer, LC–MS/MS-Based Quantification of Kynurenine Metabolites, Tryptophan, Monoamines and Neopterin in Plasma, Cerebrospinal Fluid and Brain, Bioanalysis, 2016, 8, 1903–1917 CrossRef PubMed.
- D. Virág, M. Király, L. Drahos, A. E. Édes, K. Gecse, G. Bagdy, G. Juhász, I. Antal, I. Klebovich, B. Dalmadi Kiss and K. Ludányi, Development, Validation and Application of LC–MS/MS Method for Quantification of Amino Acids, Kynurenine and Serotonin in Human Plasma, J. Pharm. Biomed. Anal., 2020, 180, 113018 CrossRef PubMed.
- G. An, M. Wei, Z. Wang, X. Zhao, X. Li and Z. He, Effects of Enzymolysis Method on the Preparation of Peptides from Wheat Flour, Food Biosci., 2022, 49, 101956 CrossRef.
- T. Fang, X. Zhang, S. Hu, Y. Yu, X. Sun and N. Xu, Enzymatic Degradation of Gracilariopsis Lemaneiformis Polysaccharide and the Antioxidant Activity of Its Degradation Products, Mar. Drugs, 2021, 19, 270 CrossRef PubMed.
- Z. Xia, J. Miao, B. Chen, J. Guo, Y. Ou, X. Liang, Y. Yin, X. Tong and Y. Cao, Purification, Identification, and Antioxidative Mechanism of Three Novel Selenium-Enriched Oyster Antioxidant Peptides, Food Res. Int., 2022, 157, 111359 CrossRef PubMed.
- M. Chauhan and P. S. Negi, Protective Effect of Phytochemicals-Rich Syzygium Malaccense Seed Extract against H2O2 Stimulated Inflammatory Response in RAW 264.7 Macrophages, Food Biosci., 2025, 66, 106230 CrossRef.
- S. Kim, C. H. Lee, J. Y. Yeo, K. W. Hwang and S. Y. Park, Immunostimulatory Activity of Stem Bark of Kalopanax Pictus in RAW 264.7 Macrophage, J. Herb. Med., 2022, 32, 100504 CrossRef.
- W. Jin, L. Yang, Y. Zhang, Y. Wang, Y. Li, Y. Zhao, L. Sun and F. Liu, Comprehensive Characterization and Metabolomics to Explore the Role of Hydrocortisone-Induced Yin Deficiency Syndrome in Mice, Biochem. Biophys. Rep., 2024, 40, 101868 Search PubMed.
- A. J. Van Der Slot, A. M. Zuurmond, A. J. Van Den Bogaerdt, M. M. W. Ulrich, E. Middelkoop, W. Boers, H. Karel Ronday, J. DeGroot, T. W. J. Huizinga and R. A. Bank, Increased Formation of Pyridinoline Cross-Links Due to Higher Telopeptide Lysyl Hydroxylase Levels Is a General Fibrotic Phenomenon, Matrix Biol., 2004, 23, 251–257 CrossRef.
- C. C. Sung, Y. C. Hsu, C. C. Chen, Y. F. Lin and C. C. Wu, Oxidative Stress and Nucleic Acid Oxidation in Patients with Chronic Kidney Disease, Oxid. Med. Cell. Longevity, 2013, 2013, 1–15 CrossRef.
- Z. Zhang, N. I. Dmitrieva, J. H. Park, R. L. Levine and M. B. Burg, High Urea and NaCl Carbonylate Proteins in Renal Cells in Culture and in Vivo, and High Urea Causes 8-Oxoguanine Lesions in Their DNA, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 9491–9496 CrossRef.
- D. Li, H. Ping, K. Li, J. Lin, X. Jiang and X. Zhang, Environmental Oestrogens Disrupt Testicular Descent and Damage Male Reproductive Health: Mechanistic Insight, J. Cell. Mol. Med., 2023, 27, 2095–2102 CrossRef PubMed.
- Q. Huang, H. Wu, X. Xiao, X. Qin and S. Liu, Preparation of Oyster Peptide and Pfaffia Glomerata Pressed Candy and Its Ameliorative Effect on Sexual Dysfunction in Male Mice, Food Sci. Nutr., 2024, 12, 5572–5586 CrossRef.
- M. Khalid, M. H. Alqarni, S. Wahab, S. Annadurai, M. A. Alamri, A. I. Foudah, T. M. Aljarba and J. Akhtar, Badruddeen and S. Ahmad, Ameliorative Sexual Behavior and Phosphodiesterase-5 Inhibitory Effects of Spondias Mangifera Fruit Extract in Rodents: In Silico, In Vitro, and In Vivo Study, J. Clin. Med., 2022, 11, 3732 CrossRef.
- J. Fang, J. Lu, Y. Zhang, J. Wang, S. Wang, H. Fan, J. Zhang, W. Dai, J. Gao and H. Yu, Structural Properties, Antioxidant and Immune Activities of Low Molecular Weight Peptides from Soybean Dregs (Okara), Food Chem.: X, 2021, 12, 100175 Search PubMed.
- Y. Oh, W. K. Jung and J. Y. Je, Protective Effect of Multifunctional Peptides PIISVYWK and FSVVPSPK on Oxidative Stress-Mediated HUVEC Injury through Antioxidant and Anti-Apoptotic Action, Process Biochem., 2023, 125, 121–129 CrossRef.
- C. X. Wang, C. C. Song, X. T. Liu, B. W. Qiao, S. Song and Y. H. Fu, ACE Inhibitory Activities of Two Peptides Derived from Volutharpa Ampullacea Perryi Hydrolysate and Their Protective Effects on H2O2 Induced HUVECs Injury, Food Res. Int., 2022, 157, 111402 CrossRef CAS PubMed.
- Y. Gao, X. Zhang, G. Ren, C. Wu, P. Qin and Y. Yao, Peptides from Extruded Lupin (Lupinus Albus L.) Regulate Inflammatory Activity via the P38 MAPK Signal Transduction Pathway in RAW 264.7 Cells, J. Agric. Food Chem., 2020, 68, 11702–11709 CrossRef CAS.
- M. O. Oyovwi, E. K. Nwangwa, B. Ben-Azu, R. A. Rotue, T. P. Edesiri, V. Emojevwe, J. C. Igweh and C. I. Uruaka, Prevention and Reversal of Chlorpromazine Induced Testicular Dysfunction in Rats by Synergistic Testicle-Active Flavonoids, Taurine and Coenzyme-10, Reprod. Toxicol., 2021, 101, 50–62 CrossRef CAS PubMed.
- I. A. Adedara, S. E. Alake, M. O. Adeyemo, L. O. Olajide, T. O. Ajibade and E. O. Farombi, Taurine Enhances Spermatogenic Function and Antioxidant Defense Mechanisms in Testes and Epididymis of L-NAME-Induced Hypertensive Rats, Biomed. Pharmacother., 2018, 97, 181–189 CrossRef CAS.
- J. Feng, W. Cao, B. Xu and Z. Wang, Structure-Based Method for the Discovery of Selective Inhibitors of PED 5 in Erectile Dysfunction Therapy from the Pacific Oyster Peptides (Crassostrea Gigas): Peptidomic Analysis, Molecular Docking, and Activity Validation, Int. J. Biol. Macromol., 2025, 295, 139494 CrossRef CAS PubMed.
- S. Marín De Jesús, R. M. Vigueras-Villaseñor, E. Cortés-Barberena, J. Hernández-Rodriguez, S. Montes, I. Arrieta-Cruz, S. G. Pérez-Aguirre, H. Bonilla-Jaime, O. Limón-Morales and M. Arteaga-Silva, Zinc and Its Impact on the Function of the Testicle and Epididymis, Int. J. Mol. Sci., 2024, 25, 8991 Search PubMed.
- M. Chalamaiah, W. Yu and J. Wu, Immunomodulatory and Anticancer Protein Hydrolysates (Peptides) from Food Proteins: A Review, Food Chem., 2018, 245, 205–222 CrossRef CAS PubMed.
- L. He, P. Li, L. H. Yu, L. Li, Y. Zhang, Y. Guo, M. Long, J. B. He and S. H. Yang, Protective Effects of Proanthocyanidins against Cadmium-Induced Testicular Injury through the Modification of Nrf2-Keap1 Signal Path in Rats, Environ. Toxicol. Pharmacol., 2018, 57, 1–8 CrossRef CAS PubMed.
- B. Kelly and E. L. Pearce, Amino Assets: How Amino Acids Support Immunity, Cell Metab., 2020, 32, 154–175 Search PubMed.
- H. J. Dong, D. Wu, S. Y. Xu, Q. Li, Z. F. Fang, L. Q. Che, C. M. Wu, X. Y. Xu and Y. Lin, Effect of Dietary Supplementation with Amino Acids on Boar Sperm Quality and Fertility, Anim. Reprod. Sci., 2016, 172, 182–189 Search PubMed.
- Y. F. Zhang, J. Y. Yang, X. P. Meng and X. L. Qiao, L-Arginine Protects against T-2 Toxin-Induced Male Reproductive Impairments in Mice, Theriogenology, 2019, 126, 249–253 CrossRef CAS.
- F. M. El-Demerdash, M. I. Yousef, F. S. Kedwany and H. H. Baghdadi, Cadmium-Induced Changes in Lipid Peroxidation, Blood Hematology, Biochemical Parameters and Semen Quality of Male Rats: Protective Role of Vitamin E and β-Carotene, Food Chem. Toxicol., 2004, 42, 1563–1571 Search PubMed.
- R. J. Aitken and J. R. Drevet, The Importance of Oxidative Stress in Determining the Functionality of Mammalian Spermatozoa: A Two-Edged Sword, Antioxidants, 2020, 9, 111 Search PubMed.
- A. T. Dinkova-Kostova and I. M. Copple, Advances and Challenges in Therapeutic Targeting of NRF2, Trends Pharmacol. Sci., 2023, 44, 137–149 CrossRef CAS PubMed.
- A. Alnajem and M. Al-Maghrebi, The Regulatory Effects of JAK2/STAT3 on Spermatogenesis and the Redox Keap1/Nrf2 Axis in an Animal Model of Testicular Ischemia Reperfusion Injury, Cells, 2023, 12, 2292 CrossRef CAS.
- B. Ma, J. Zhang, Z. Zhu, X. Bao, M. Zhang, C. Ren and Q. Zhang, Aucubin, a Natural Iridoid Glucoside, Attenuates Oxidative Stress-Induced Testis Injury by Inhibiting JNK and CHOP Activation via Nrf2 up-Regulation, Phytomedicine, 2019, 64, 153057 CrossRef CAS.
- J. Sun, H. Wang, B. Liu, W. Shi, J. Shi, Z. Zhang and J. Xing, Rutin Attenuates H2O2-Induced Oxidation Damage and Apoptosis in Leydig Cells by Activating PI3K/Akt Signal Pathways, Biomed. Pharmacother., 2017, 88, 500–506 CrossRef CAS PubMed.
- A. M. Abdelrahman, R. Al Maskari, H. Ali, P. Manoj and Y. Al Suleimani, Effect of Riociguat on Adenine-Induced Chronic Kidney Disease in Rats, Biology, 2025, 14, 161 CrossRef CAS.
- M. Wang and F. Gao, Suppression of nF-κB by ro 106-9920 Alleviates Ischemia/Reperfusion-Induced Renal Dysfunction and Inflammation via Modulation of Neutrophil Extracellular Trap Formation in Acute Kidney Injury Mice, Renal Failure, 2025, 47, 2545983 CrossRef.
- J. Chen, X. Yang, Z. He, W. Chen, Y. Zhao, J. Li, Y. Zong and R. Du, Cynomorium Songaricum Rupr. Flavonoids Improve Cyclophosphamide-Induced Reproductive Function Damage by Regulating the Testosterone Synthesis Pathway, Front. Pharmacol., 2024, 15, 1457780 CrossRef CAS.
- H. Schultz, X. Zhou, C. A. I. Alonso, L. Ongaro, Y. F. Lin, M. Loka, T. Brabletz, S. Brabletz, M. P. Stemmler, U. Boehm and D. J. Bernard, ZEB1 Inhibits LHβ Subunit Transcription When Overexpressed, but Is Dispensable for LH Synthesis in Mice, Endocrinology, 2024, 165, 116 CrossRef.
- S. C. Krzastek and R. P. Smith, Non-Testosterone Management of Male Hypogonadism: An Examination of the Existing Literature, Transl. Androl. Urol., 2020, 9, 160–170 CrossRef PubMed.
- M. R. Melis and A. Argiolas, Oxytocin, Erectile Function and Sexual Behavior: Last Discoveries and Possible Advances, Int. J. Mol. Sci., 2021, 22, 10376 CrossRef CAS PubMed.
- N. R. Han, J. E. Bae, H. W. An, H. J. Yun, B. Retnoaji, S. Lee and S. G. Lee, Influence of Potato Starch on the 3D Printing of Senior-Friendly Foods Enriched with Oyster Powder, LWT, 2025, 224, 117886 CrossRef CAS.
- S. Wang, X. Yang, S. Cheng and M. Du, The Structural Characteristics of Crassostrea Gigas Peptide-Zinc Complexes and the Bioactive Mechanism of Their Enhanced Zinc Absorption, Food Biosci., 2024, 58, 103722 Search PubMed.
- X. Yang, S. Wang, H. Liu, T. Zhang, S. Cheng and M. Du, A Dual Absorption Pathway of Novel Oyster-Derived Peptide-Zinc Complex Enhances Zinc Bioavailability and Restores Mitochondrial Function, J. Adv. Res., 2025, 78, 47–62 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |








