Maternal high-fat diet before pregnancy triggered off abnormal carnitine mapping and gut microbiota development in offspring
Chen
Zhang
,
Shiqi
Sun
,
Yufei
Song
,
Xinyi
Pan
,
Liyun
Shi
 and
Cuiyuan
Jin
and
Cuiyuan
Jin
 *
*
Key Laboratory of Artificial Organs and Computational Medicine of Zhejiang Province, Institute of Translational Medicine, Zhejiang Shuren University, Hangzhou, 310015, P. R. China. E-mail: jincuiyuan@zjsru.edu.cn
First published on 25th November 2025
Abstract
Maternal high-fat diet (HFD) can increase the risk of metabolic disturbances in offspring by modifying their gut microbiota, which may persist into adulthood. However, how HFD before pregnancy affects the carnitine mapping and alters the development of the gut microbiota in offspring remains to be explored. In this study, 4-week-old C57BL/6J mice were initially fed HFD for 10 weeks. The mice were subsequently mated and maintained on a normal diet during pregnancy. We investigated lipid metabolism, intestinal barrier function, and gut microbiota development in the offspring at various time points. We discovered that the carnitine development curve of the offspring in the HFD group was significantly different from that of the offspring in the control group. The expression of genes involved in lipid metabolism was altered significantly in the liver and colon. The development of the gut microbiota also significantly changed, and the degree of microbial invasion of the mucus increased. Specifically, the abundance of Akkermansia and Muribaculaceae increased significantly in the offspring, whereas the abundance of Bacteroides and Prevotellaceae UGG 001 decreased significantly in both the dams and offspring after maternal HFD before pregnancy. Maternal HFD before pregnancy, on the one hand, altered the developmental trajectory of the gut microbiota in offspring, impaired intestinal barrier function, and increased susceptibility to microbial infections. On the other hand, maternal HFD before pregnancy triggered an abnormal carnitine profile and disrupted lipid metabolism in the offspring.
Introduction
A growing body of evidence supports the theory of “developmental programming”, which posits that the maternal environment plays a critical role in shaping fetal and infant development, thereby modifying disease susceptibility later in life.1 Maternal obesity and unhealthy dietary patterns, particularly those rich in fat and sugar, have been increasingly recognized as critical determinants of offspring metabolic health.2,3 Such diets can induce maternal gut microbiota dysbiosis, systemic inflammation, and metabolic dysfunction, which may be vertically transmitted to the offspring, thereby predisposing them to obesity and related comorbidities.4,5 The impact of the maternal diet on offspring health has become a critical area of research, particularly given the increasing prevalence of high-fat diets (HFDs) and their potential long-term consequences. Epidemiological and animal studies have demonstrated that maternal HFD substantially elevates the risk of various diseases in offspring,6 including obesity,7 nonalcoholic fatty liver disease (NAFLD),8 neurodevelopmental disorders,9 colitis,10 cardiovascular disease,11 allergic asthma,12 and potentially even cancer.13One of the key mechanisms is the disruption of the gut microbiota, which plays a crucial role in host health throughout the entire lifespan of the host. In a nonhuman Macaca fuscata (Japanese macaque) primate model, the gut microbiota composition of offspring during the first year of life was altered when the mother consumed an HFD during pregnancy and lactation, irrespective of the postweaning diet.14 This association was further validated in a population-based birth cohort study involving mothers and infants.15 In mouse models, maternal low-fiber diet modified the composition of the microbiota in breast milk, thereby impairing its vertical transmission and disrupting the establishment of the gut microbiota in offspring.16 Consumption of maternal low-fiber high-fat diet during lactation not only continued to alter the gut microbial communities in the offspring but also predisposed them to develop diet-induced obesity (DIO), which is characterized by weight gain, hypercholesterolemia, dysregulated blood glucose levels, and insulin resistance.5 Research has shown that maternal HFD can alter the composition and function of the gut microbiota in offspring, potentially leading to metabolic disorders and chronic inflammation.17
In addition to the gut microbiota, metabolism is another critical pathway affected by maternal diet. Emerging evidence suggests that maternal HFD can disrupt carnitine metabolism in offspring, leading to abnormal carnitine mapping and potentially contributing to metabolic dysregulation.18 Carnitine is essential for fatty acid oxidation and energy metabolism, and its proper function is vital for maintaining metabolic homeostasis.19 Our focus on this metabolic pathway stemmed from the central role that carnitine plays in fatty acid β-oxidation and its well-established clinical significance.20 In fact, the measurement of free carnitine (C0) and acylcarnitines (C2–C18) serves as the most specific and direct clinical diagnostic tool for disorders of fatty acid oxidation and organic acidemia in newborns.21 However, research investigating the effects of maternal HFD on carnitine metabolism in offspring remains relatively scarce. These findings position the carnitine profile as a highly sensitive biomarker that is capable of revealing latent metabolic dysfunction programmed by maternal diet before the onset of more overt pathological conditions.
Increasing evidence indicates that maternal HFD may have lifelong effects on offspring health. However, research exploring the effects of HFD before pregnancy on offspring health remains scarce. Existing research has shown that the adverse physiological effects associated with a history of HFD-induced obesity can persist over the long term. Even after transitioning back to a standard diet and attaining a non-overweight condition, the body continues to exhibit a physiological profile comparable to that observed during the period of high-fat dietary intake.22 Furthermore, following successful weight loss, adipocytes in the human body retain an “obesity memory”, which can influence a range of gene expression processes within these cells. More importantly, these epigenomic alterations remain present for an extended period, even after body weight has been reduced to a healthy range.23 Consequently, we hypothesized that consumption of maternal high-fat diet prior to conception may lead to enduring alterations in metabolic profiles and the gut microenvironment, which may persist even following a subsequent transition to a standard healthy diet during gestation and lactation, thereby potentially exerting long-term developmental and health effects on offspring. These findings highlight the need for interventions targeting preconception dietary habits to mitigate adverse outcomes in offspring.24 In summary, the interplay between the maternal diet, gut microbiota, and carnitine metabolism is complex and multifaceted. Elucidating the mechanisms through which maternal HFD induces abnormalities in these pathways is crucial for developing interventions to enhance offspring health.
In this study, we aimed to elucidate the effects of maternal high-fat diet consumption prior to pregnancy on maternal metabolic health, as well as on the developmental trajectories of carnitine metabolism, gut microbiota colonization, and intestinal barrier function in offspring. We fed normal female mice with an HFD for 10 weeks and then allowed them to mate and give birth to their offspring. The offspring mice were fed with a normal diet to observe the effects of the maternal HFD before pregnancy on them.
Materials and methods
Animal experimental design
Four-week-old specific pathogen-free C57BL/6 mice were purchased from the China National Laboratory Animal Resource Center (Shanghai, China) and housed at Zhejiang Shuren University. After 3 days of adaptation, all the females were divided into 2 groups, ensuring that the average body weight was consistent. One group was fed a normal diet (ND group), and the other was fed a high-fat diet (60% kcal from fat diets, 12492M) (HFD group) for 10 weeks. The body weight, food intake, and serum indices of the mice were recorded during the modeling period. The males and females were mated at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, after which the males were removed when the females became pregnant. During this period, all the dams and the offspring mice were fed a normal diet until the end of the study. The dams were sacrificed during the late pregnancy period (marked as F0-LTP) or after production. The offspring were sacrificed at the ages of 10 days (marked as F1-10), 20 days (marked as F1-20), 4 weeks (marked as F1-28) and 7 weeks (marked as F1-49). The serum, liver, colon, colonic contents, amniotic fluid and placenta were collected for measurement or stored at −80 °C until processing. All animal procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals of Zhejiang Shuren University and approved by the Animal Ethics Committee of Zhejiang Shuren University.
1 ratio, after which the males were removed when the females became pregnant. During this period, all the dams and the offspring mice were fed a normal diet until the end of the study. The dams were sacrificed during the late pregnancy period (marked as F0-LTP) or after production. The offspring were sacrificed at the ages of 10 days (marked as F1-10), 20 days (marked as F1-20), 4 weeks (marked as F1-28) and 7 weeks (marked as F1-49). The serum, liver, colon, colonic contents, amniotic fluid and placenta were collected for measurement or stored at −80 °C until processing. All animal procedures were performed in accordance with the Guidelines for Care and Use of Laboratory Animals of Zhejiang Shuren University and approved by the Animal Ethics Committee of Zhejiang Shuren University.
16S rRNA gene sequencing and analysis
DNA was extracted from the colonic contents, and the V4 variable region of the 16S rRNA gene was sequenced on an Illumina NovaSeq 6000 platform. The sequenced libraries were processed using QIIME2 (ver. 2020.6) and denoised and clustered into amplicon sequence variants (ASVs) with Deblur. Taxonomic classification was performed using the SILVA 99% rRNA gene reference database (ver. 138). Taxa bar plot, alpha diversity, and beta diversity analyses were performed using the R package (ver. 3.2.0).Measurement of triglyceride, cholesterol and glucose levels
Serum samples were obtained by collecting blood before sacrifice. The blood samples were then centrifuged at 3000 rpm for 10 min at room temperature. The quantified liver tissue was weighed, after which two grinding beads and quantitative normal saline were added. Liver homogenate was obtained by grinding with a grinder. Triglyceride (TG), total cholesterol (TC) and glucose levels in the serum or liver were quantified with the kits purchased from the Nanjing Jiancheng Institute of Biotechnology (Jiancheng, China). The analyses strictly followed the manufacturer's instructions.Gene expression analysis
RNA was extracted from the tissue samples using the TRIzol method with RNAiso Plus (TaKARA, Japan) according to the manufacturer's instructions, followed by cDNA synthesis using a 5× HiScript II qRT SuperMix II Kit (Vazyme, China). Data acquisition was performed using a CFX Opus96 Real-time PCR system (Bio-Rad, USA) using the ChamQ Universal SYBR qPCR Master Mix (Vazyme, China). Cycle threshold (Ct) values were normalized to those of 18S. The primer pair data are included in Table S1.Analysis of colon bacterial 16S/colon 18S
A 5 mm-long colon sample was placed in sterile phosphate buffered saline (PBS), cut along the intestinal direction with ophthalmic scissors and then washed. DNA was extracted from the tissue samples using a One-tube General Sample DNA Kit (Sangon Biotech, China) consisting of the Qlysis-G reagent, proteinase K and buffer NST. Data acquisition was performed using a CFX Opus96 Real-time PCR system (Bio-Rad, USA) using the ChamQ Universal SYBR qPCR Master Mix (Vazyme, China). The primer pair data are included in Table S1.Carnitine and amino acid assays
Amniotic fluid and blood samples were dropped on cards, naturally dried, and stored in a 2–8 °C refrigerator for the subsequent analysis. Carnitine and amino acid levels were analyzed using a PerkinElmer NeoBase™ non-derived MSMS Kit (PerkinElmer, USA) and flow injection tandem mass spectrometry. Data were acquired using a Waters Acquity™ TQD system (Waters Corporation, USA) equipped with a triple quadrupole detector. The main carnitines examined in this analysis included C0, acetylcarnitine (C2), propionylcarnitine (C3), malonylcarnitine/3-hydroxy-butyrylcarnitine (C3DC/C4OH), butyrylcarnitine (C4), methylmalonyl/3-hydroxy-isovalerylcarnitine (C4DC/C5OH), isovalerylcarnitine (C5), tiglylcarnitine (C5:1), glutarylcarnitine/3-hydroxy-hexanoylcarnitine (C5DC/C6OH), hexanoylcarnitine (C6), adipylcarnitine (C6DC), octanoylcarnitine (C8), octenoylcarnitine (C8:1), decanoylcarnitine (C10), decenoylcarnitine (C10:1), decadienoylcarnitine (C10:2), dodecanoylcarnitine (C12), dodecenoylcarnitine (C12:1), tetradecanoylcarnitine (C14), tetradecenoylcarnitine (C14:1), tetradecadienoylcarnitine (C14:2), 3-hydroxy-tetradecanoylcarnitine (C14OH), hexadecanoylcarnitine (C16), hexadecenoylcarnitine (C16:1), 3-hydroxy-hexadecanoylcarnitine (C16OH), 4-hydroxy-hexadecenoylcarnitine (C16:1OH), octadecanoylcarnitine (C18), octadecenoylcarnitine (C18:1), octadecadienoylcarnitine (C18:2), 3-hydroxy-octadecanoylcarnitine (C18OH), and 3-hydroxy-octadecenoylcarnitine (C18:1OH). The main amino acids examined in this analysis included alanine (ALA), arginine (ARG), citrulline (CIT), glycine (GLY), leucine/isoleucine/hydroxyproline (LEU/ILE/PRO-OH), methionine (MET), ornithine (ORN), phenylalanine (PHE), proline (PRO), tyrosine (TYR), and valine (VAL).Statistical analysis
The data are expressed as the mean ± standard error, and analysis of variance (ANOVA) was performed using SPSS 24.0. The images were plotted using GraphPad 10 and Keynote.Results
Maternal HFD before pregnancy affects the carnitine and amino acid levels in the dams and offspring
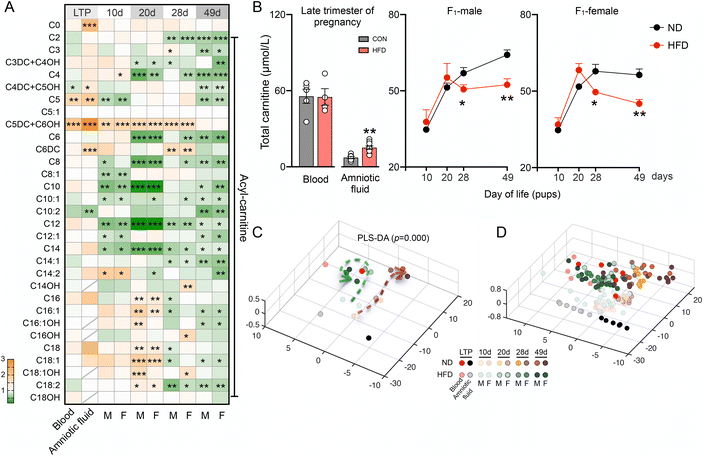
To investigate the effects of HFD before pregnancy on the dams and offspring, we fed 4-week-old female mice with an HFD for 10 weeks and the control group was fed a normal diet. During the modeling period, we discovered that the body weight and food intake of the mice did not significantly differ (Fig. S1A and B). Serum glucose levels at different time points tended to increase in the HFD group during the initial stage of modeling but tended to be consistent (Fig. S1C). After 10 weeks of HFD consumption, serum TG and TC levels were significantly higher in the HFD group than in the ND group (Fig. S1D and E). The female mice then became pregnant, during which time they were fed a normal diet.We measured carnitine and amino acid levels in the dams and their offspring, and these indices were applied to screen for neonatal inherited metabolic diseases in clinical practice. The results revealed that an HFD before pregnancy affected the carnitine levels in the amniotic fluid and blood of the dams in the late stages of pregnancy (Fig. 1A and B). The main manifestations were that C4DC + C5OH was significantly decreased and C5 and C5DC + C6OH were significantly increased in the blood of the dams, whereas C10:2 was significantly decreased and C0, C5DC + C6OH, C5, C5DC + C6OH, C6DC, and total carnitine were significantly increased in the amniotic fluid of the dams (Fig. 1A and B). The HFD before pregnancy also strongly affected the carnitine levels in the offspring, which generally tended to decrease until 7 weeks of age (Fig. 1A). Furthermore, we discovered that the total carnitine levels were not significantly different before weaning (10 and 20 days old) but were significantly lower after weaning in both male and female offspring (28 and 49 days old) (Fig. 1B). As the mice aged, the positions of the control and HFD samples changed unidirectionally according to the partial least squares discriminant analysis (PLS-DA) plots, with greater differences in the HFD-fed mice, suggesting deceleration of normal age-related carnitine development (Fig. 1C and D and S2A).
Although the amino acid developmental trajectory was not clear, the HFD before pregnancy also significantly altered the amino acid composition (Fig. S2B and C). Combined with the coefficient loading diagram (Fig. S2D and E), we found that C0, C2, ALA and GLY could be considered as potential markers of the effects of maternal HFD before pregnancy. In the HFD group, C0 and ALA increased significantly from 20 days of age to adulthood, whereas C2 decreased from 4 weeks of age and GLY decreased from 20 days of age (Fig. S2F). These significant fluctuations in amino acid and carnitine levels in the offspring indicated that HFD before pregnancy strongly affects the metabolism of fatty acids and amino acids in the offspring.
Maternal HFD before pregnancy persistently affects the liver lipid metabolism of the offspring
On the basis of the changes of carnitine in the amniotic fluid, genes involved in fatty acid β-oxidation were examined in the placentas of mice during late pregnancy. The HFD strongly affected placental gene expression (Fig. S3), indicating that maternal HFD before pregnancy may affect lipid metabolism in offspring. As shown in Table 1, HFD before pregnancy had little effect on the dam hepatic TG and TC levels in the late pregnancy. The hepatic TG level in the HFD group was significantly greater than that in the ND group during lactation (10 and 20 days of age), but lower than that in the ND group at the age of 49 days (Table 1). The hepatic TC level of male mice in the HFD group was significantly lower than that in the ND group at the age of 10 and 28 days, and that of female mice at the age of 49 days was also significantly lower than that in the ND group (Table 1).| TG (μmol g−1) | TC (μmol g−1) | ||||
|---|---|---|---|---|---|
| ND | HFD | ND | HFD | ||
| The presented values are the means ± SEMs. An asterisk indicates a significant difference (* at p < 0.05, ** at p < 0.01 and *** at p < 0.001) between the ND and HFD groups. | |||||
| F0-LTP | 2.33 ± 0.53 | 1.85 ± 0.76 | 0.45 ± 0.11 | 0.32 ± 0.04 | |
| F1-10d | M | 5.03 ± 0.50 | 8.70 ± 1.35* | 4.37 ± 0.93 | 1.45 ± 0.33* |
| F | 4.26 ± 0.65 | 6.88 ± 0.75* | 2.56 ± 0.74 | 2.35 ± 0.42 | |
| F1-20d | M | 5.90 ± 0.38 | 10.83 ± 0.63*** | 5.28 ± 0.22 | 5.88 ± 0.54 |
| F | 5.79 ± 0.32 | 10.20 ± 1.69** | 5.81 ± 0.43 | 5.60 ± 0.29 | |
| F1-28d | M | 11.43 ± 0.71 | 12.31 ± 2.08 | 7.72 ± 0.40 | 3.46 ± 0.91*** |
| F | 11.24 ± 0.76 | 10.84 ± 0.56 | 5.10 ± 0.53 | 5.47 ± 0.51 | |
| F1-49d | M | 24.82 ± 2.33 | 18.21 ± 1.14* | 6.31 ± 0.65 | 7.62 ± 0.56 |
| F | 31.36 ± 2.32 | 15.83 ± 1.67*** | 8.42 ± 0.82 | 6.14 ± 0.48* | |
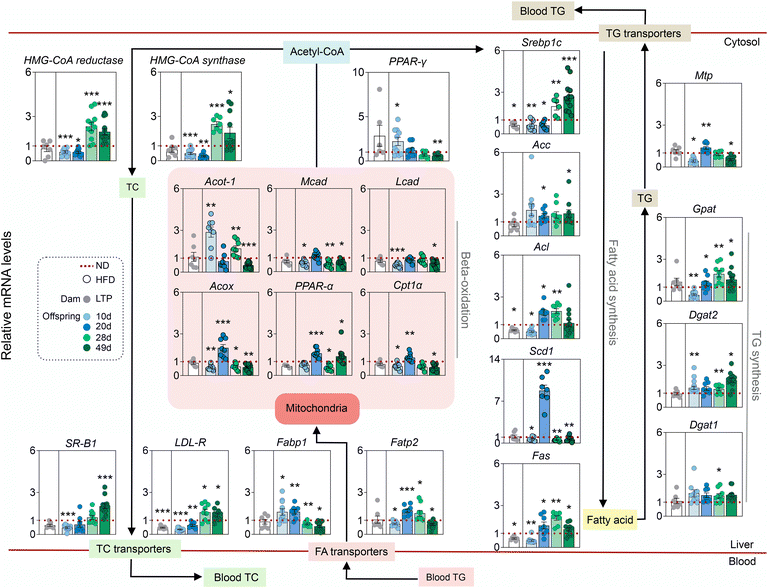
We also examined the expression of lipid metabolite-related genes in the livers of the mice. At the gene expression level, HFD before pregnancy had a stronger effect on the offspring than the dams (Fig. 2). In the dams, the HFD significantly downregulated the gene expression of the TC transporter (LDL-R) and fatty acid synthesis (Srebp1c, Acl and Fas). In the offspring, HFD before pregnancy changed the expression of genes related to TC metabolism, fatty acid metabolism and TG synthesis. A series of results revealed that HFD before pregnancy disturbed hepatic lipid metabolism in the offspring, and this trend could continue with age.
Maternal HFD before pregnancy affects the development of the gut microbiota in the offspring
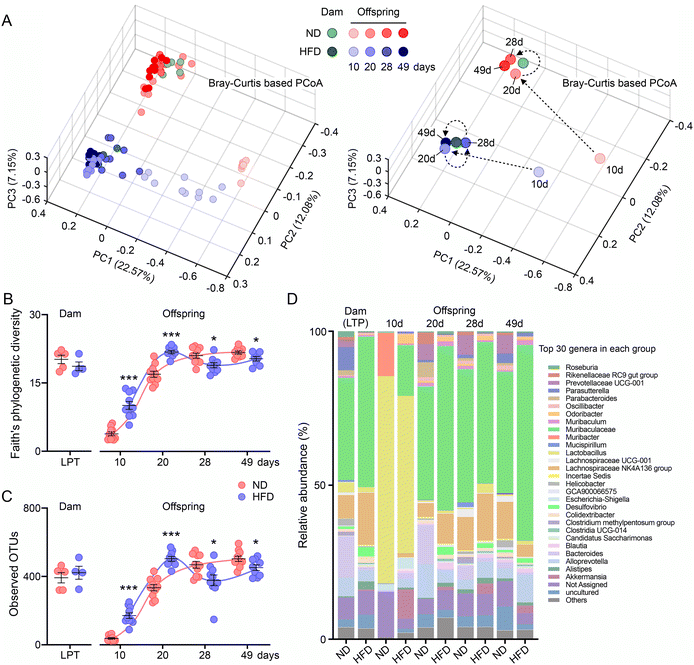
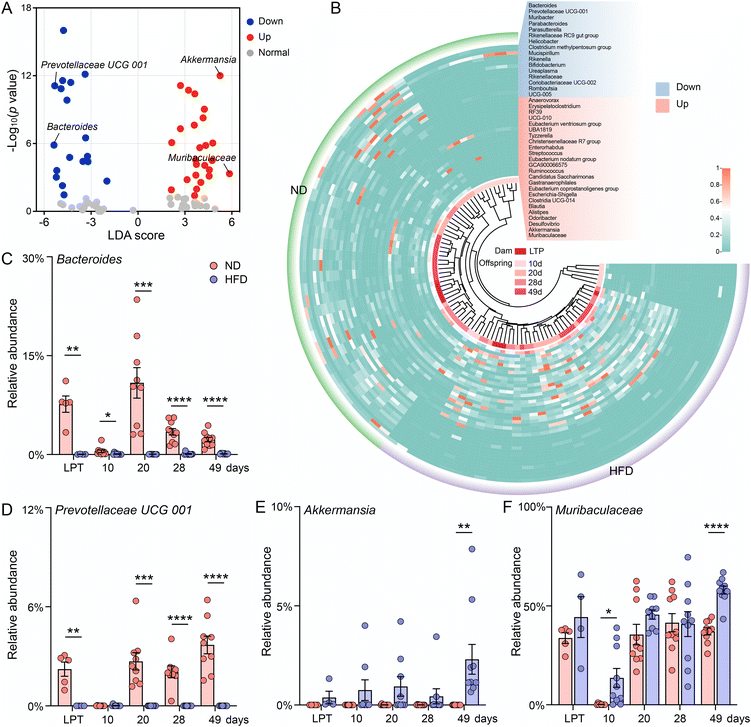
To examine the development of the gut microbiota in offspring in response to maternal HFD before pregnancy, we studied a series of colon content samples. In general, pre-pregnancy HFD had little effect on the dams (Table S2 and Fig. 3 and S4). In contrast, the Shannon, Simpson, Chao1 and ACE indices of the offspring in the HFD group significantly increased during lactation (10 and 20 days old), whereas the Shannon index significantly decreased after weaning (28 and 49 days old), and the Chao1 and ACE indices significantly decreased at the age of 28 days (Table S2). At the phylum level, Firmicutes and Proteobacteria were predominant in the ND group at 10 days of age, whereas Bacteroidetes, Firmicutes and Proteobacteria were predominant in the HFD group (Fig. S4A). Similarly, the gut microbiota was dominated by aerobes and facultatively anaerobes in pups at 10 days of age, but pre-pregnancy HFD led to the premature colonization of anaerobes (Fig. S4C). Principal co-ordinate analysis (PCoA) based on the Bray–Curtis distance revealed that pre-pregnancy HFD significantly altered the microbial community structure (Fig. S5B–E) and caused a deviation in the developmental trajectory of the gut microbiota in the offspring (Fig. 3A). HFD before pregnancy also significantly increased Faith's phylogenetic diversity and the number of taxa observed during lactation (10 and 20 days old), but significantly decreased these parameters after weaning (28 and 49 days old) (Fig. 3B and C).Significant alterations in the microbial composition were observed across multiple taxonomic levels, from phylum to genus, in both dams and offspring (Fig. 3D, S4A, and S4B). LEfSe analysis revealed that particular taxa changes were associated with maternal HFD prior to pregnancy. Notably, maternal HFD consistently decreased the abundance of Bacteroides and Prevotellaceae UGG 001 in both the dams and offspring (Fig. 4C and D), whereas the abundance of Akkermansia and Muribaculaceae increased exclusively in the offspring (Fig. 4E and F). Maternal HFD before pregnancy also significantly increased the abundance of potentially pathogenic bacteria in the intestines of adult offspring (Fig. S4D). Maternal HFD before pregnancy significantly altered the gut microbiota development in the offspring, increasing microbial diversity during lactation but decreasing it after weaning, and shifted the microbial community structure and composition across multiple taxonomic levels.
Maternal HFD before pregnancy altered the co-abundance network
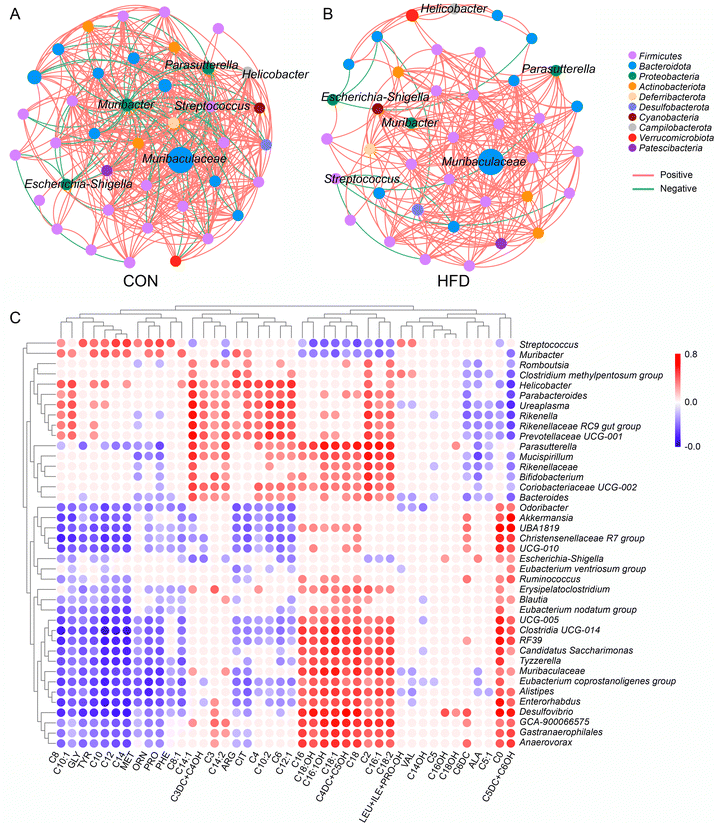
In addition, a comprehensive analysis was conducted to assess the influence of maternal pre-pregnancy HFD on the co-abundance network of the gut microbiota. The co-abundance network of the gut microbiota in the control group revealed a diverse microbial community (Fig. 5A). Phyla such as Firmicutes, Bacteroidota, Proteobacteria, and Actinobacteria were also well-represented, indicating a complex and stable microbial network. In contrast, the co-abundance network from the HFD group exhibited a less diverse structure (Fig. 5B). Notable genera included Helicobacter, Escherichia–Shigella, Mumbacter, Streptococcus, and Mumbaculaceae. The reduction in taxonomic diversity suggested that the HFD may simplify microbial interactions and reduce community complexity. Correlation analysis between the gut bacteria and the metabolites such as carnitine and amino acids revealed multiple significant associations (Fig. 5C). Several bacterial taxa showed potential relationships with these metabolites, including Parasutterella, Helicobacter, Bifidobacterium, Akkermansia, Escherichia–Shigella, Ruminococcus, and many others. These findings suggested that specific gut microbes may be involved in or influenced by the metabolism of carnitine and amino acids, which could have implications for host health under different dietary regimens.Maternal HFD before pregnancy affects the colon barrier function in the dams and offspring
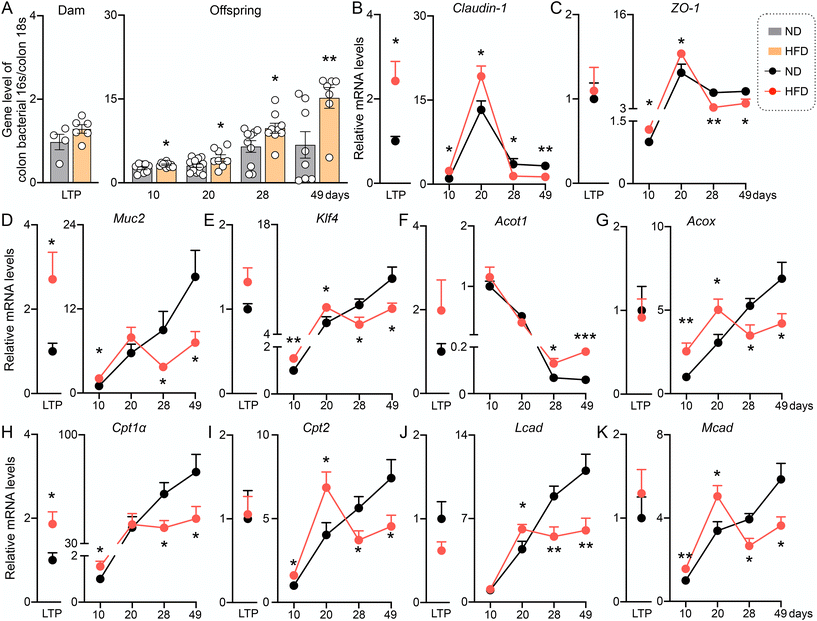
To investigate whether maternal HFD exposure before pregnancy affects the development of the intestinal barrier and induces functional defects in offspring mice, we performed real-time quantitative PCR analysis on colonic tissues from the offspring at various time points. After the dams were fed an HFD before pregnancy, the ratio of 16S to 18S in the colon of the offspring was significantly increased at 10, 20, 28 and 49 days post-natal (Fig. 6A), suggesting a persistent microbial infection in the offspring intestines. The expression of tight junction-related genes (Claudin-1 and Zo-1) and mucus secretion-related genes (Klf4 and Muc2) in the colons of the offspring also changed significantly (Fig. 6B–E). In addition, genes involved in fatty acid β-oxidation (Acot1, Acox, Cpt1α, Cpt2, Lcad and Mcad) were all affected by maternal HFD before pregnancy (Fig. 6F–K). We observed an up-regulation trend in the expression of most genes prior to weaning, followed by a down-regulation trend post-weaning. This shift may be associated with the weaning process and the introduction of solid foods. On the whole, the HFD before pregnancy affected the development of the intestinal barrier in both dams and offspring, which had a long-term effect on the offspring.Discussion
Diet is the main contributor to the species and functions of the gut microbiota.25 Previous studies have focused primarily on investigating the influence of HFD on relevant mechanisms and exploring how HFD-induced obesity or other diseases can be treated with specific therapies. Limited research has focused on the association between maternal HFD intake and the growth and development of the intestinal tract in offspring. In this study, we observed the metabolic function, gut microbiota and intestinal barrier of offspring mice of dams that were fed an HFD before pregnancy and returned to a normal diet during pregnancy. Through this experiment, we concluded that HFD prior to pregnancy leads to significant alterations in the maternal gut microbiota, atypical gut microbiota development and intestinal barrier function in the offspring, and dysregulation of amino acid and lipid metabolism in the offspring. This study not only highlights the long-term metabolic and microbial consequences of maternal HFD but also provides a foundation for future research aimed at developing nutritional or microbial interventions to prevent or reverse these effects.The changes in lipid and amino acid metabolism in late pregnant female mice were minimal (Fig. 1 and 2 and S1 and S2), suggesting that the influence of HFD before pregnancy on maternal physiology and various aspects of body function is limited. This is due to the normal development of maternal body tissues and organs, which possess a certain self-regulatory capacity. As a result, lipid and amino acid metabolism in maternal organisms is unlikely to be significantly affected by HFD, or HFD may only disturb maternal mice in only a few specific aspects, such as increasing the carnitine level in the amniotic fluid and up-regulating the expression of genes related to fatty acid β-oxidation in the placenta (Fig. 1 and S3).
Interestingly, PCoA based on the Bray–Curtis distance revealed that pre-pregnancy HFD significantly altered the microbial community structure in late pregnant female mice (Fig. S5A). Although the offspring were fed a normal chow diet, the gut microbiota of the offspring was disturbed (Fig. 3, 4, S4 and S5). Significant alterations in the α- and β-diversity of the gut microbiota of the offspring from the maternal HFD group were observed. During lactation, the phylogenetic diversity of the gut microbiota in the offspring of the HFD group was significantly greater than that in the ND group. However, after weaning, this diversity significantly decreased compared with that in the ND group. Our findings show that maternal HFD before pregnancy alters the developmental trajectory of the gut microbiota of the offspring, which aligns with the established principle that the mother is the primary source of the microbiome of an infant.26 Epidemiological evidence indicates that a significant proportion—approximately 58.5%—of the infant gut microbiota is derived from the mother.27 This vertical transmission is a multifaceted process, occurring through birth, breastfeeding (contributing up to 49.9% of the early microbiota), and potentially even in utero. Furthermore, this transfer is not random but is selective and can involve mechanisms such as horizontal gene transfer.28 Therefore, maternal HFD before conception likely alters the maternal microbial ‘seed’ population, which is then vertically transmitted, thereby disrupting the normal colonization and development of the gut microbiota in the offspring and contributing to the metabolic phenotypes we observed.
In addition, maternal HFD consistently decreased the abundance of Bacteroides in both the dams and offspring (Fig. 4). A recent study suggested a positive correlation between neonatal growth and Bacteroides abundance.29 Notably, members of the Prevotellaceae family play context-dependent roles in host metabolism and inflammation.30,31 Although some species are associated with improved metabolic outcomes, others may promote inflammation under certain dietary conditions. The consistent reduction in Prevotellaceae UCG-001 observed in our study may reflect a loss of beneficial functions, contributing to metabolic dysregulation in the offspring. That is, the changes in gut microbiota diversity and relative abundance in the offspring can be attributed to mother-to-infant transmission mechanisms, including vertical transmission of the gut microbiota, elevated carnitine levels in maternal amniotic fluid and breastfeeding.
The down-regulated expression levels of tight junction genes and mucus secretion-related genes reveal the hidden dangers associated with the integrity of the intestinal barrier (Fig. 4). Moreover, maternal HFD before pregnancy led to persistent microbial infection in the offspring intestines. It also damaged the integrity of the intestinal barrier of the offspring mice, leading to a defective physical barrier. A well-developed and mature gut epithelial barrier is essential for maintaining a healthy and stable gut environment, enabling offspring to effectively respond to infections, for nutrient synthesis and absorption, and to establish optimal communication with the brain and other organs.32 That is, a pre-pregnancy HFD can negatively impact the health of offspring by disrupting the intestinal homeostasis.
Carnitine plays a fundamental role in the process of fatty acid oxidation, and the total carnitine content represents the level of lipid metabolism in various parts of the human body.33 Although the total carnitine level did not significantly change during offspring lactation, the total carnitine level after weaning was significantly lower than that in the ND group (Fig. 1). This phenomenon may be attributed to the compensatory effect of breast milk on the carnitine concentration of the offspring during lactation, but the offspring could not maintain themselves after switching to a solid diet; thus, the decreasing trend became more obvious with age. We speculated that the effects of HFD before pregnancy on the offspring might have already occurred during pregnancy.
According to our results, TG and TC were elevated in the early life of the offspring (Table 1), mainly because of maternal breastfeeding, which also indicated that even if the chow diet was restored during pregnancy, the influence of the HFD before pregnancy on mothers still occurred. However, the TG and TC contents in the liver of the offspring tended to decrease during the adult phase. The trend was opposite during lactation and after weaning, which may be due to the change in the diet of the offspring from breast milk to solid food.
The down-regulated genes associated with TG synthesis, fatty acid synthesis and fatty acid metabolism indicated that HFD before pregnancy may inhibit TG and fatty acid metabolism in the offspring liver and that the increase in TG during lactation may come from breast milk. Second, TC synthesis and transport showed the same downward trend, which directly reflected the disorder of fatty acid metabolism and the levels of TG and TC. This disorder not only occurs early in life but also has a lasting effect on the long-term health of offspring. The newly born offspring were in the growth and development stage, and all body tissues and organs were in an imperfect state of function. The abnormal composition of intestinal microorganisms shaped the body development of the offspring, contributing to defects in fat metabolism, which was proven by the differences in the carnitine development curve and abnormal metabolism of amino acids. Beyond the hepatic lipid metabolism and carnitine profiles detailed in our results, maternal HFD is known to have a profound deleterious effect on systemic fatty acid metabolism. It promotes a hepatic prolipogenic state and significantly reduces the availability of long-chain polyunsaturated fatty acids (PUFAs), such as docosahexaenoic acid (DHA).34 Given that PUFAs are crucial for brain development, influencing neurogenesis, synaptic plasticity, and inflammatory responses, their deficiency may represent a key mechanistic link between maternal HFD and an increased risk of neurodevelopmental disorders in the offspring. Future investigations stemming from this work will aim to directly examine the impact of pre-pregnancy HFD on offspring brain lipid composition and neurodevelopment.
Clinical studies have demonstrated that maternal diets rich in fiber, omega-3 fatty acids, and polyphenols during pregnancy and lactation are associated with improved metabolic and immune outcomes in offspring. For instance, adherence to a Mediterranean diet—characterized by high consumption of fruits, vegetables, legumes, whole grains, and olive oil—during pregnancy has been linked to a reduced risk of obesity and allergic diseases in children.35,36 Additionally, ensuring adequate intake of dietary fiber supports a healthy maternal gut microbiota, which is crucial for vertical transmission and the establishment of a beneficial offspring microbiome.37 Similarly, omega-3 supplementation, particularly DHA, during pregnancy and lactation has been shown to support optimal neurodevelopment and may mitigate pro-inflammatory metabolic profiles in the offspring.38 These findings highlight the potential of targeted nutritional guidance for expectant and nursing mothers to promote long-term offspring health. Our findings align with growing evidence that maternal HFD compromises offspring metabolic health through disrupted carnitine metabolism and gut microbiota development. Notably, the reduction in beneficial taxa such as Bacteroides and Prevotellaceae UCG-001 may contribute to impaired barrier function and metabolic inflammation. Future studies should explore whether dietary fiber supplementation or targeted microbial interventions can mitigate these transgenerational effects.
Conclusions
As a crucial component of daily life, maintaining a balanced diet has long been associated with good health.39 Our study showed that HFD not only disrupts maternal gut microbiota diversity and carnitine levels, leading to an imbalanced state, but also has both short- and long-term effects on offspring. Importantly, this HFD pattern, which closely resembles the Western diet, can induce a range of adverse reactions, even in the absence of noticeable weight changes or severe intestinal pathological alterations. Such intestinal abnormalities may significantly compromise the health of offspring. Therefore, during pregnancy or while nurturing offspring, addressing this potential issue is essential to mitigate the adverse effects of unhealthy dietary patterns.Given the detrimental impact of HFD on the gut microbiota composition and metabolic health, dietary interventions such as increased dietary fiber and supplementation with prebiotics may serve as a promising strategy to restore microbial diversity and function. By promoting the growth of beneficial bacteria, increasing the production of short-chain fatty acids, and enhancing the integrity of the intestinal barrier, mitigating the transgenerational effects of maternal HFD may be possible.
Author contributions
Chen Zhang: conceptualization, methodology, formal analysis, data curation, and writing – original draft preparation. Shiqi Sun: data curation, validation and formal analysis. Yufei Song: data curation and validation. Xinyi Pan: formal analysis and validation. Liyun Shi: methodology and project administration. Cuiyuan Jin: project administration, funding acquisition, and writing – reviewing and editing.Conflicts of interest
The authors declare no competing interests.Data availability
The data supporting the findings of this study will be made available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo03845f.
Acknowledgements
This research was supported by the Zhejiang Provincial Natural Science Foundation of China under Grant No. LTGY24H260005 and the Scientific Research Foundation of Zhejiang Shuren University (2022R041).References
- S. Zeng, M. Zhou, D. Mu and S. Wang, Clinical implications of maternal multikingdom transmissions and early-life microbiota, Lancet Microbe, 2025, 6(7), 101042 CrossRef PubMed.
- C. Barrera, R. Valenzuela and R. Chamorro, et al., The impact of maternal diet during pregnancy and lactation on the fatty acid composition of erythrocytes and breast milk of Chilean women, Nutrients, 2018, 10(7), 839 CrossRef PubMed.
- M. L. Kearns and C. M. Reynolds, The impact of non-nutritive sweeteners on fertility, maternal and child health outcomes: a review of human and animal studies, Proc. Nutr. Soc., 2024, 83(4), 280–292 CrossRef CAS PubMed.
- Y. Guo, Z. Wang and L. Chen, et al., Diet induced maternal obesity affects offspring gut microbiota and persists into young adulthood, Food Funct., 2018, 9(8), 4317–4327 RSC.
- J. Zou, V. L. Ngo and Y. Wang, et al., Maternal fiber deprivation alters microbiota in offspring, resulting in low-grade inflammation and predisposition to obesity, Cell Host Microbe, 2023, 31(1), 45–57 CrossRef CAS PubMed.
- C. Zhang, W. Zhong and L. Miao, et al., Relationship of maternal high-fat diet to intestinal barrier integrity and disease in offspring, Nutr. Res., 2025, 140, 77–92 CrossRef CAS PubMed.
- Y. Xu, D. Yang and L. Wang, et al., Maternal high fat diet in lactation impacts hypothalamic neurogenesis and neurotrophic development, leading to later life susceptibility to obesity in male but not female mice, Adv. Sci., 2023, 10(35), e2305472 CrossRef PubMed.
- H. Peng, J. Li and H. Xu, et al., Offspring NAFLD liver phospholipid profiles are differentially programmed by maternal high-fat diet and maternal one carbon supplement, J. Nutr. Biochem., 2023, 111, 109187 CrossRef CAS PubMed.
- A. M. Ceasrine, B. A. Devlin and J. L. Bolton, et al., Maternal diet disrupts the placenta-brain axis in a sex-specific manner, Nat. Metab., 2022, 4(12), 1732–1745 CrossRef CAS PubMed.
- S. Bibi, Y. Kang, M. Du and M. J. Zhu, Maternal high-fat diet consumption enhances offspring susceptibility to DSS-induced colitis in mice, Obesity, 2017, 25(5), 901–908 CrossRef CAS PubMed.
- F. Chen, K. Cao and H. Zhang, et al., Maternal high-fat diet increases vascular contractility in adult offspring in a sex-dependent manner, Hypertens. Res., 2021, 44(1), 36–46 CrossRef PubMed.
- H. Lin, Y. Zhao and Y. Zhu, et al., Maternal high-fat diet aggravates allergic asthma in offspring via modulating CD4+ T-Cell differentiation, Nutrients, 2022, 14(12), 2508 CrossRef CAS PubMed.
- Y. Sun, Q. Wang and Y. Zhang, et al., Multigenerational maternal obesity increases the incidence of HCC in offspring via miR-27a-3p, J. Hepatol., 2020, 73(3), 603–615 CrossRef CAS PubMed.
- J. Ma, A. L. Prince and D. Bader, et al., High-fat maternal diet during pregnancy persistently alters the offspring microbiome in a primate model, Nat. Commun., 2014, 5, 3889 CrossRef CAS PubMed.
- D. M. Chu, K. M. Antony and J. Ma, et al., The early infant gut microbiome varies in association with a maternal high-fat diet, Genome Med., 2016, 8, 77 CrossRef PubMed.
- M. A. Sikder, R. B. Rashid and T. Ahmed, et al., Maternal diet modulates the infant microbiome and intestinal Flt3L necessary for dendritic cell development and immunity to respiratory infection, Immunity, 2023, 56, 1098–1114 CrossRef CAS PubMed.
- L. Ding, B. D. Weger and J. Liu, et al., Maternal high fat diet induces circadian clock-independent endocrine alterations impacting the metabolism of the offspring, iScience, 2024, 27(7), 110343 CrossRef CAS PubMed.
- H. Peng, H. Xu and J. Wu, et al., Maternal high-fat diet disrupted one-carbon metabolism in offspring, contributing to nonalcoholic fatty liver disease, Liver Int., 2021, 41(6), 1305–1319 CrossRef CAS PubMed.
- W. K. Wu, Y. L. Lo and J. Y. Chiu, et al., Gut microbes with the gbu genes determine TMAO production from L-carnitine intake and serve as a biomarker for precision nutrition, Gut Microbes, 2025, 17(1), 2446374 CrossRef PubMed.
- F. Xiang, Z. Zhang and J. Xie, et al., Comprehensive review of the expanding roles of the carnitine pool in metabolic physiology: beyond fatty acid oxidation, J. Transl. Med., 2025, 23(1), 324 CrossRef PubMed.
- I. Scala, G. Parenti and G. Andria, Universal screening for inherited metabolic diseases in the neonate (and the fetus), J. Matern.-Fetal Neonat. Med., 2012, 25(Suppl 5), 4–6 CrossRef PubMed.
- M. Hata, E. M. M. A. Andriessen and M. Hata, et al., Past history of obesity triggers persistent epigenetic changes in innate immunity and exacerbates neuroinflammation, Science, 2023, 379(6627), 45–62 CrossRef CAS PubMed.
- L. C. Hinte, D. Castellano-Castillo and A. Ghosh, et al., Adipose tissue retains an epigenetic memory of obesity after weight loss, Nature, 2024, 636(8042), 457–465 CrossRef CAS PubMed.
- J. Zhang, F. Zhang and X. Didelot, et al., Maternal high fat diet during pregnancy and lactation alters hepatic expression of insulin like growth factor-2 and key microRNAs in the adult offspring, BMC Genomics, 2009, 10, 478 CrossRef PubMed.
- H. C. Wastyk, G. K. Fragiadakis and D. Perelman, et al., Gut-microbiota-targeted diets modulate human immune status, Cell, 2021, 184(16), 4137–4153 CrossRef CAS PubMed.
- P. Ferretti, E. Pasolli and A. Tett, et al., Mother-to-infant microbial transmission from different body sites shapes the developing infant gut microbiome, Cell Host Microbe, 2018, 24, 133–145 CrossRef CAS PubMed.
- D. Bogaert, G. J. van Beveren and E. M. de Koff, et al., Mother-to-infant microbiota transmission and infant microbiota development across multiple body sites, Cell Host Microbe, 2023, 31(3), 447–460 CrossRef CAS PubMed.
- T. Vatanen, K. S. Jabbar and T. Ruohtula, et al., Mobile genetic elements from the maternal microbiome shape infant gut microbial assembly and metabolism, Cell, 2022, 185(26), 4921–4936 CrossRef CAS PubMed.
- L. L. Neves, A. B. Hair and G. A. Preidis, A systematic review of associations between gut microbiota composition and growth failure in preterm neonates, Gut Microbes, 2023, 15(1), 2190301 CrossRef PubMed.
- L. Wang, R. Hu and S. Ma, et al., Dihydroquercetin attenuated Prevotella copri-caused intestinal injury by modulating gut microbiota and bile acids in weaned piglets, Anim. Nutr., 2024, 20, 303–310 CrossRef PubMed.
- J. Gong, Q. Zhang and R. Hu, et al., Effects of Prevotella copri on insulin, gut microbiota and bile acids, Gut Microbes, 2024, 16(1), 2340487 CrossRef PubMed.
- J. K. Gustafsson and M. E. V. Johansson, The role of goblet cells and mucus in intestinal homeostasis, Nat. Rev. Gastroenterol. Hepatol., 2022, 19(12), 785–803 CrossRef PubMed.
- M. Almannai, M. Alfadhel and A. W. El-Hattab, Carnitine inborn errors of metabolism, Molecules, 2019, 24(18), 3251 CrossRef CAS PubMed.
- L. Mercado-López, Y. Muñoz and C. Farias, et al., High-fat diet in perinatal period promotes liver steatosis and low desaturation capacity of polyunsaturated fatty acids in dams: a link with anxiety-like behavior in rats, Nutrients, 2025, 17(7), 1180 CrossRef PubMed.
- A. Zaragoza-Martí, N. Ruiz-Ródenas and I. Herranz-Chofre, et al., Adherence to the Mediterranean Diet in Pregnancy and its benefits on maternal-fetal health: a systematic review of the literature, Front. Nutr., 2022, 9, 813942 CrossRef PubMed.
- M. Fan, Y. Chu and Y. Zheng, et al., Association of pregnancy diet with metabolic adverse outcomes in pregnant women and their children: a systematic review and meta-analysis, Ann. Nutr. Metab., 2025, 81(3), 123–140 CrossRef CAS PubMed.
- S. E. Maher, E. C. O'Brien and R. L. Moore, et al., The association between the maternal diet and the maternal and infant gut microbiome: a systematic review, Br. J. Nutr., 2023, 129(9), 1491–1499 CrossRef CAS PubMed.
- M. P. Herrero Jiménez, S. Del Pozo de la Calle, C. Cuadrado Vives and D. Escobar Sáez, Nutritional supplementation in pregnant, lactating women and young children following a plant-based diet: A narrative review of the evidence, Nutrition, 2025, 136, 112778 CrossRef PubMed.
- M. Liu, S. Li and S. Cao, et al., Let food be your medicine - dietary fiber, Food Funct., 2024, 15(15), 7733–7756 RSC.
| This journal is © The Royal Society of Chemistry 2026 |