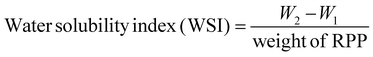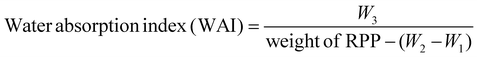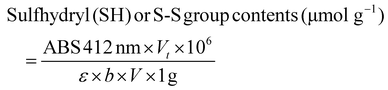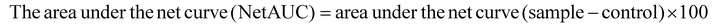Ameliorative potential of rice protein hydrolysates prepared through high-pressure processing against diabetic cardiomyopathy
Yu-Tsung
Cheng†
a,
Ping-Hsiu
Huang†
 b,
Yung-Jia
Chan
c,
Wen-Chien
Lu
d and
Po-Hsien
Li
b,
Yung-Jia
Chan
c,
Wen-Chien
Lu
d and
Po-Hsien
Li
 *e
*e
aCardiovascular Center, Taichung Veterans General Hospital, Taichung 407219, Taiwan, Republic of China
bDepartment of Food Science, National Chiayi University, Chia-Yi City 600355, Taiwan, Republic of China
cCollege of Biotechnology and Bioresources, Da-Yeh University, Changhua 515006, Taiwan, Republic of China
dDepartment of Food and Beverage Management, Chung-Jen Junior College of Nursing, Health Sciences and Management, Chia-Yi City, 60077, Taiwan, Republic of China
eDepartment of Food and Nutrition, Providence University, Taichung 433303, Taiwan, Republic of China. E-mail: pohsien0105@pu.edu.tw; Tel: +886-4-2632-8001 (ext. 15326)
First published on 9th December 2025
Abstract
Various functional foods, including meal replacements and nutritional supplements, are available to consistently regulate blood glucose levels in patients with type 2 diabetes mellitus (T2DM). This study proposes the development of high-pressure processing (HPP) treatment for rice protein (RP) flour to produce rice protein peptides (RPP). This dietary intervention aims to stabilize glycemic levels and mitigate myocardial damage caused by oxidative stress (OS) in patients with diabetic cardiomyopathy (DCM). This study demonstrated that the protein, branched-chain amino acid (BCAA), and peptide contents in RPP increased significantly following HPP treatments ranging from 200 to 600 MPa. Additionally, the study found that RPP effectively regulated blood glucose levels (621.85–181.73 mg dL−1) in rats with DCM, attenuated myocardial injury caused by OS, and prevented the development of myocardial infarction (MI) indices and cardiac oxidative-inflammatory parameters in an animal model. Notably, RPP may effectively inhibit nuclear factor (NF)-κB expression and activity, thereby reducing myocardial cellular pyroptosis. Consequently, the findings of this study contribute to the advancement and commercialization of RPP as a potential health food product, with the dual benefits of regulating blood glucose levels and providing antioxidant properties. Furthermore, this study offers novel perspectives for preventing and alleviating heart failure in DCM, thus guiding future research.
1. Introduction
The metabolic disorders commonly observed in individuals with T2DM exert a significant impact on multiple organ systems, thereby contributing to potentially severe complications that have been acknowledged as a major global public health threat.1,2 It is notable that patients with T2DM, in the absence of other heart diseases, develop peculiar cardiovascular complications, mainly resulting in myocardial dysfunction, known as diabetic cardiomyopathy (DCM), an extensively recognized clinical entity.3–5 Nevertheless, patients with T2DM who experienced suboptimal glycemic control and concurrent cardiovascular risk factors were at an elevated risk of developing DCM, which was observed in up to 30% of individuals with T2DM.5 Additionally, a higher susceptibility to DCM has been documented among elderly patients with T2DM.6 Advancement of these lesions is associated with mitochondrial dysfunction due to excessive accumulation of lipids in the myocardium, increased production of reactive oxygen species (ROS), and elevated glucose levels. Namely, it contributes to developing advanced glycation end-products that promote OS and sustained inflammation, leading to myocardial injury.3,5,7–10 Moreover, it has been recently demonstrated that cardiomyocyte pyroptosis is linked to the aberrant activation of the NF-κB signaling pathway in cardiovascular diseases, indicating that intervention in the expression of this pathway provides an apparent therapeutic target.11 This condition frequently lacks symptoms during the initial stages of DCM and frequently coincide with other diabetic complications, complicating the process of establishing a definitive diagnosis complex and prolonging the subclinical period.9,12 However, the most appropriate approach for identifying DCM involves discerning the left ventricle's structure and detecting functional changes through echocardiography, excluding other conditions that may contribute to heart failure.9,12,13 Unfortunately, the specific mechanisms have not been elucidated, and practical agents or inhibitors to inhibit this pathway are lacking.11 According to those mentioned above, these multifactorial pathological mechanisms play a pivotal role in the development of DCM, all intricately interconnected with controlling blood glucose levels.2,4,8,10,14 Nevertheless, it has been suggested that in addition to improving blood glucose levels, it is imperative to augment the antioxidant system within the body sufficiently to mitigate cardiac OS in rats with DCM.15The HPP technology is a commercially successful non-thermal sterilization method with diverse applications, including inactivating pathogenic microorganisms and preserving natural attributes such as shape, color, aroma, flavor, and nutrients in food products.16–18 This technology has demonstrated the ability to induce degradation of the cellular walls in plants or animals, resulting in enhanced permeability and improved intracellular mass transfer mechanisms.19–21 Specifically, it enhances dietary bioactive substances (such as polyphenols, peptides, free amino acids (AA), γ-aminobutyric acid, etc.) and starch (molecular/crystalline arrangement and functionalities).17,18,22,23 In addition, the bioactive peptides, comprising a range of several to fewer than 20 AA, can be directly absorbed by the human body to provide physiological functions such as antioxidant, anti-inflammatory, anti-cancer, antibacterial, intestine-modulatory, immunomodulatory, metal-chelating, and blood pressure-lowering.24,25 Therefore, the health benefits of functional foods conferred by these specific protein fragments’ bioactive peptides (antioxidant peptides) can maintain human health, food safety, and quality by reducing lipid peroxidation and OS.16 This was caused by free radicals produced during oxidative reactions in the body and foods.16 However, the presence of bitterness peptides resulting from enzymatic hydrolysis and deamidation poses concerns regarding food safety.26,27 This ultimately restricts the nutritional and application benefits of RP. Nevertheless, exploring alternative methods to enhance RP solubility could potentially overcome these challenges that are difficult to address simultaneously.27
In previous times, RP's relatively limited protein content and solubility have resulted in its lack of appeal, namely being a by-product of starch extraction, which has traditionally been regarded as having limited value.26 However, the suitability of RP as a protein for preventing obesity and DM has been demonstrated, with its positive impact on body performance and health being attributed to a specific protein fragment.28–31 The global market for RP witnessed a stable growth, expanding from USD 132.99 million in 2024 to USD 249.83 million in 2032, reflecting a robust Compound Annual Growth Rate (CAGR) of 8.2%.32 The key advantages of RP products lie in their hypoallergenic and nutritional properties, endowing them with a competitive edge over other plant-based protein alternatives.33–37 Notably, most RP is obtained through hydrolysis using alkaline or proteolytic enzymes. According to reports, the structure of RP was altered (such as indigestible prolamin) by alkaline extraction to enhance digestibility (in vivo) compared to rice flour and starch-degraded RP, thereby potentially improving the nutritional value of high-starch base foods, particularly for elderly individuals.38–40 Consequently, incorporating RP could be beneficial with higher bioavailability attributed to alkaline-extracted RP.38,41 In addition, it has been indicated that proteins with limited digestibility could be characterized as prolamins, which are lipid-rich core proteins deficient in lysine but abundant in cysteine.36 Furthermore, ingesting essential AA derived from proteins has been shown to markedly augment the relative abundance of the gut microbiota (Alistipes spp.), thereby facilitating the performance of muscle function in elderly individuals.42,43 In addition, a clinical study (n = 50) showed that dietary supplementation with highly bioavailable purified rice endothelial protein in maintenance hemodialysis patients led to a statistically significant 0.07 g kg−1 day−1 increase in urea kinetic-based normalized protein catabolic rate.40 The authors highlighted that this effect was achieved while maintaining stable serum phosphorus and potassium concentrations, mitigating the risk factors associated with elevated protein intake and mortality. In particular, proteins and protein hydrolysates exhibit potential as functional food ingredients through biological activities.37,44–47 These bioactive peptides derived from diverse sources exhibit the potential to reduce OS, modulate immune responses, mitigate inflammation, and enhance intestinal barrier integrity to fortify, uphold, or restore various physiological homeostatic processes.46,47 However, these biological activities are intricately associated with the AA composition, sequence, molecular weight, and hydrophobicity of peptides; specifically, peptides characterized by low molecular weight and pronounced hydrophobicity may exhibit enhanced anti-inflammatory properties.47–52
Based on the above, there have been many explorations of HPP achieving increased bioactivity content in various food materials and realizing the potential of having the desired functional food components. However, more information is needed on advanced practical applications for products and efficacy validation. Concurrently, developing a robust and pragmatic food matrix was deemed essential to enhance the assessment of our nutritious foods’ functionality, nutrient composition, bioaccessibility, bioavailability, etc., while exploring the interrelationships among individual food components in an in vivo model.53
Therefore, this study assessed the practical applicability of RPP by utilizing a previously successful functional food carrier in an established animal model of DCM.2 Specifically, this study evaluated the impact of HPP-treated RPP on the physiological parameters of DCM rats, encompassing growth performance, serum biochemistry, MI indices, and cardiac oxide-inflammatory markers. This study aimed to investigate the health-promoting efficacy of RPP as a dietary supplement for five consecutive months in rats with DCM to gain a more comprehensive understanding of RPP.
2. Materials and methods
2.1. Materials
The 80% RP flour [made from Vietnamese rice (cóm tấm; Oryza sativa L. subsp. indica Kato), >80% crude protein content and lower trace mineral (phosphorus and potassium) contents] was offered as a gift from Vedan Vietnam Enterprise Co., Ltd (Long Thanh, Vietnam). The corn flour (CF) and food-grade ingredients, such as cellulose, lard, and casein, were procured from a local market (Taichung, Taiwan). The chemicals used in this study were procured from Sigma-Aldrich® (Merck KGaA, Darmstadt, Germany) and used as is unless explicitly stated otherwise.2.2. Preparation of the extruded products
| Group | ||||
|---|---|---|---|---|
| Composition (%) | Control and diabetes (DM) | RPP | ||
| 200 | 400 | 600 | ||
| Casein | 16 | |||
| Lard | 8 | |||
| DL-Methionine | 0.2 | |||
| AIN-76 mineral mix | 1 | |||
| AIN-76 vitamin mix | 4 | |||
| Cholesterol | 0.5 | |||
| Cholic acid | 0.1 | |||
| Choline chloride | 0.2 | |||
| Cellulose | 5 | |||
| Corn starch | 25 | |||
| RPP flour | 30 | |||
| Rice bran | 10 | |||
2.3. Physicochemical properties of the HPP-treated RPP flour
where W1 represents the supernatant weight; W2 represents the constant weight; and W3 represents the precipitate weight.
To 2 mL of the above suspension was added 8 mL of 0.1 M PBS (pH 8.0), and the pH of the solution was adjusted to 8.0 with 1 M NaOH. Next, 1 mL of 2 mM 5,5-dithio-bis-(2-nitrobenzoic acid) (DTNB; Ellman's reagent) was added and allowed to stand for 40 min. Subsequently, 0.1 M PBS (pH 7.0) was added until the final volume was 11 mL and centrifuged (3000g) for 10 min. The supernatant was collected and filtered through a 0.45 μm membrane before the absorbance at 412 nm was measured, and then the SH content in RP was calculated using the following formula:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 cm−1); b indicates the optical range of the cuvette (1 cm); and V indicates the volume of the sample (L).
600 M−1 cm−1); b indicates the optical range of the cuvette (1 cm); and V indicates the volume of the sample (L).
Moreover, the S–S bond contents were determined using cysteine. Briefly, 1 mL of the above suspension was added to 50 μL of 0.1 M ethylenediaminetetraacetic acid (EDTA) solution and 3 mL of 0.1 M PBS (pH 8.0), followed by 2 μL of octanol and 0.48 g of urea, and then kept for dissolution. Next, 1 mL of NaBH4 solution (2.5%) was added and reacted in a water bath at 40 °C for 30 min. Afterward, 1 mL of an acidifier (pH 2.7; 10.4 g of NaH2PO4·H2O dissolved in 100 mL of 0.1 M HCl) and 2 mL of acetone were added, followed by blowing with N2 gas for 6–8 min at 25 °C (to remove excess NaBH4). Then, 1 mL of DTNB solution (2 mM) was added, allowed to stand for 40 min, and centrifuged for 10 min (3000g);then the supernatant was collected for filtration using a 0.45 μm membrane. The absorbance of the filtrate was measured at 412 nm, and the S–S bond contents in RP were calculated using the above formula.
2.3.6.1. 1,1-Diphenyl-2-picrylhydrazyl (DPPH). The methodology described by Wang et al.60 was employed to determine the DPPH radical scavenging capacity of the sample. The sample (1.5 mg mL−1) was mixed with a 150 μM DPPH reagent dissolved in an 80% (v/v) methanol solution. The mixture was subsequently shielded from any light exposure for 90 min, followed by conducting absorbance measurements at 517 nm using a microplate reader (Emax, Molecular Devices, LLC, San Jose, CA, USA). The DPPH radical scavenging capacity of the sample was calculated by employing the following equation:
where ABS 517 nm indicates the absorbance of RPP at 517 nm.
2.3.6.2. Oxygen radical absorbance capacity (ORAC). The ORAC of the RPP for this study was determined following the description of Asma et al.,61 with some modifications. ORAC has been extensively employed in the evaluation of the antioxidative potential of food items, utilizing the AAPH [2,2′-azobis(2-amidinopropane) dihydrochloride] radical to induce the formation of peroxyl radicals through heating in the presence of ample oxygen and expedite fluorescence degradation.62 The fluorescence change can serve as an indicator for assessing the antioxidant capacity of the samples. The sample in a 96-well plate was incubated in a water bath (37 °C) for 5 min, followed by the sequential addition of 50 μL of fluorescein sodium salt (78 nM) to each well, along with 50 μL of the sample and 25 μL of AAPH (221 mM), which were shaken for 10 s. The fluorescence values were recorded every 5 min for 90 min, with excitation at 485 nm and emission at 520 nm. The fluorescence value and time were used to generate a plot, from which the area under the sample and blank curves was derived. The equation provided below calculates the Net AUC by determining the area under the net curves. The standard substance 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) was employed, and linear regression was performed to establish the standard curves for determining the ORAC of the sample.
2.4. Experimental animals
The design and operations of the animal experiments in this study were performed with minor modifications based on our team's descriptions of the published study by Cheng et al.2 This study utilized male Wistar rats aged six weeks (body weight (BW) 200 ± 20 g) purchased from LASCO Co., Ltd (Taipei, Taiwan). Upon arrival at the laboratory, the animals were allowed 1 week of acclimatization before being randomly divided into groups (n = 6) (Fig. 1). These groups included the control group (standard diet, without induction or treatment), the DCM group (streptozotocin (STZ) induction for T2DCM on a standard diet), and the HPP treatment 200, 400, and 600 groups. Throughout the experiment, a controlled environment (pathogen-free; temperature and humidity were maintained at 25 ± 2 °C and 65 ± 5%; the dark and light periods were from 07:00 to 19:00 and 19:00 to 07:00) was provided for all rats, which were housed in cages made of plastic measuring 30 [L] × 20 [W] × 10 [H] cm3, with two rats per cage. The dietary formulas for each group were prepared according to the protocol described in section 2.2.2 and the diet was provided ad libitum, with daily renewal. All groups (except for the control group) were induced with T2DM, which underwent a 12 h fast before the injection of STZ (Millipore®, Merck KGaA). The STZ solution (40 mg kg−1) was prepared in a light-protected container (maintained in an ice bath) using ice-cold 0.01 mM citric acid buffer (pH 4.5), which was administered intravenously. The induction sessions were conducted consecutively for seven sessions with three-day intervals. On the last day of the experiment, all animals were subjected to a 24 h fasting period, during which their BWs and blood glucose levels were measured. Subsequently, euthanasia was performed by intraperitoneal injection of sodium pentobarbital (150 mg per kg BW). Afterward, a cardiac blood sample was obtained through puncture and subjected to centrifugation (at 4 °C, 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g) for 10 min, while the resulting serum was utilized for the subsequent biochemical analysis. The excised cardiac tissue was rinsed with a 0.9% NaCl solution and weighed. Additionally, one-third of the cardiac tissue was preserved in 10% formalin-buffered solution. The other cardiac tissue was homogenized in phosphate-buffered saline, followed by centrifugation (3000g) for 30 min. These steps were performed to facilitate the subsequent correlative analyses (section 2.5). All animal experiments complied with the regulations outlined by the Committee for Control and Supervision of Experiments on Animals and the National Institutes of Health, ensuring proper care and use of experimental animals. The protocol under code 20230807 A001 has been approved by the Committee on Animal Research at Providence University.
000g) for 10 min, while the resulting serum was utilized for the subsequent biochemical analysis. The excised cardiac tissue was rinsed with a 0.9% NaCl solution and weighed. Additionally, one-third of the cardiac tissue was preserved in 10% formalin-buffered solution. The other cardiac tissue was homogenized in phosphate-buffered saline, followed by centrifugation (3000g) for 30 min. These steps were performed to facilitate the subsequent correlative analyses (section 2.5). All animal experiments complied with the regulations outlined by the Committee for Control and Supervision of Experiments on Animals and the National Institutes of Health, ensuring proper care and use of experimental animals. The protocol under code 20230807 A001 has been approved by the Committee on Animal Research at Providence University.
2.5. Analysis of serum biochemistry and determination of factors related to cardiac oxidative inflammation
The other parameters employed in this study were determined following the procedures outlined by Cheng et al.2 and employing specialized ELISA kits (Abcam Plc.). According to the manufacturer's instructions, these factors in cardiac homogenates were quantified. Specifically, the determined immune-related factors were interleukin (IL)-6 (ab234570, sensitivity: 43 pg mL−1, range: 125–8000 pg mL−1), IL-1β (ab255730, sensitivity: 26.58 pg mL−1, range: 54.69–3500 pg mL−1), tumor necrosis factor (TNF-α; ab236712, sensitivity: 1.04 pg mL−1, range: 18.75–1200 pg mL−1), and NF-κB (ab100785 with a sensitivity of 25 pg mL−1 and a range of 82.3–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 pg mL−1).
000 pg mL−1).
2.6. Statistical analysis
The data were presented as mean ± standard deviation (SD). The RP extruded products underwent triplicate measurements (n = 3), while the animal experiments were conducted with 6 replicates per group (n = 6). In addition, the data were graphed using GraphPad Prism software (version 9, GraphPad Software, LLC, Boston, MA, USA) and subjected to one-way analysis of variance, followed by Newman–Keuls post-hoc analyses. A significance level of p < 0.05 was deemed statistically significant.3. Results and discussion
3.1. Effects of different HPP treatments on the compositions of rice protein peptide (RPP) flour
The present study showed that the protein, BCAA, and peptide contents of all RPP groups increased in response to increasing pressure (Fig. 2), with significant variations observed among the different groups (p < 0.05). Specifically, 600 MPa provides satisfactory performance for these substances. This also signified that treatment with HPP can provide satisfactory reinforcement at the level where RP already contains high levels of arginine and BCAA levels.36 This observed phenomenon can be ascribed to excessively high pressures (>400 MPa), which enhance the diffusion and collision rates of proteins or peptides, expedite protein–protein or peptide–peptide aggregation, and reduce the hydrolysis rate, thereby facilitating the production of bioactive peptides.18,25,44,66,67 However, the hydrolysates obtained consisted of intricate peptide mixtures, some of which may exhibit limited efficacy in vivo.44,66 Conversely, treatment with HPP facilitated the protein conformation and molecular chain extension change. Then, the exposure to specific or new restriction sites enabled the subsequent digestive enzymes to selectively recognize and hydrolyze AA sequences to generate peptides possessing distinct active motifs.16,25,37,44,66,68 Therefore, this study showed that the application of HPP effectively increased the protein, BCAA, and peptide contents in RPP flour.3.2. Effects of different HPP treatments on the physicochemical properties of rice protein peptide (RPP) flour
It is well known that the WSI of proteins forms the base for essential functional properties in food systems, including WHC, emulsification, foaming, oil-holding capacity, and gelation.27,69 It serves as a reliable indicator of the potential applications of proteins while effectively enhancing the solubility of RP, facilitating its utilization in a broader range of fields.27 However, the abundance of glutelin in RP leads to its poor solubility in aqueous solution, particularly with high concentrations, whereas the aggregation and cross-linking of disulfide bonds provide the primary constraints on its insolubility.69 The findings of this study showed that the WSI and WHC increased as the pressure increased in all RPP groups (Fig. 3A and B), which were significantly different from each other (p < 0.05). The above phenomenon may be attributed to the HPP treatment that makes the RPP flour particles and forms a dense 3-dimensional protein network structure, while a few starch gelatinizations occurred that could effectively cover the water, increasing the WHC.21 Conversely, fluctuations in temperature can lead to denaturation or aggregation of proteins, thereby diminishing part of the protein's hydration capacity,17 while the alteration may subsequently result in an elevated WSI. Another plausible explanation for the increase in solubility is that external forces (HPP, extrusion, heat treatments, ultrasonics, micro-fluidics, etc.) induce a higher exposure of the hydrophilic groups of the RP, thereby facilitating enhanced interaction between proteins and water molecules.27,69 It is worth mentioning that studies have reported that the non-covalent reactions (primarily hydrogen bonding) of protein–protein (RP-cod protein) with an alkaline solution trigger the sheet helix transition and enhance functional properties like solubility (more than 90% (w/v)), gelling, and emulsification.70,71 These observed phenomena have also been attributed to the exposure of hydrophilic sequences and the burial of hydrophobic regions.71 Interestingly, Abd Rahim et al.72 have reported that the salting out of ions at pH 7 disrupts the protein–water interaction, accumulating protein molecules while leading to enhanced protein–protein interactions and reduced solubility. Therefore, this study indicated that the 600 MPa-treated HPP provided improved RPP flour with satisfactory WSI and WHC.This study showed that the free SH content in RP flour from all HPP-treated groups increased with pressure (Fig. 3C), and there were significant differences (p < 0.05). Conversely, the S–S content showed the opposite trend (Fig. 3D), with significant differences among the groups (p < 0.05). Notably, it has been suggested that the poor solubility of RP may be attributed to the presence of intermolecular S–S bonds (glutelins and globulins) and the relatively high molecular weight.36 In comparison, the issue can be addressed by reducing these factors to enhance the extractability and bioavailability of RP.36 Therefore, this study's results indicated that HPP treatment of RPP can effectively dissociate and refold the protein structure and contribute to applications for deeper processing.
Regarding the free radical scavenging capacities, both DPPH and ORAC of the HPP-treated groups in this study exhibited a positive correlation with the pressure (Fig. 3E and F), and there were significant differences between each group (p < 0.05). These phenomena were attributed to releasing antioxidant peptides (or AA) from RPP, thereby providing high activity against oxidants and exhibiting antioxidant capacity.73–75 The enzymatic hydrolysis of RP was also found to yield antioxidant peptides or AA, thereby enhancing its antioxidant capacity, including DPPH and ABTS scavenging activity, reducing capacity, and ORAC.74–76 It is worth mentioning that Liu et al.77 also reported that the antioxidant ability of RP is related to the process conditions of alkaline hydrolysis, which involves the AA composition. Therefore, this study's results indicated that HPP treatment could remarkably improve the antioxidant ability of RP flour.
The molecular structure of the HPP-treated RPP in this study, determined by FTIR, showed absorption peaks ranging from 1000–1300 cm−1 in the infrared spectrum (Fig. 3G). These were attributed to functional groups such as C–C, C–O, and C–H stretching vibrations and C–OH bending vibrations.78,79 The peak marked absorption between 1010 and 1095 cm−1 was detected in all groups, indicating the presence of pyranose rings, whereas the peaks of carboxyl (COOCH3) and free carboxyl (COO–) group asymmetric and symmetric stretching were measured at 1600–1630 cm−1.79–81 The structural components, α-helix, β-sheet, β-turn, and aggregated strands of RPP, were exhibited at 1648–1660, 1626–1640, 1662–1684, and 1610–1628 cm−1, respectively.72,82,83 In addition, the peak ranges at 1600–1700 and 1500–1600 cm−1 were attributed to amides I and II, respectively, which included C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations and N–H bending vibrations, namely, protein secondary structure conformation.69 The presence of bound water was indicated by the peak observed at 1650 cm−1.79 Several weaker wave peaks at 1965 and 2150–2200 cm−1 belonged to the C–O bound folded state of the protein and the triple bond of alkyne stretching, respectively.72 The vibration at 2850 cm−1 corresponded to the helical structures formed by AA (lysine and arginine) and the turn structure of proline.72,84 Moreover, the peak profiles of 2800–3000 (the symmetric stretching mode of C–H bonds in the aliphatic side chain and aldehyde functional groups, including methyl and methylene) and 3100–3500 cm−1 (the vibration of amide A; hydroxyl groups) corresponded to the hydrophobicity and hydrophilicity of RPP, respectively.69,72,78,81 Yet, there was no evident change in the peak profiles of all groups, whereas the light spectrum increased with the increased stress of HPP, which agreed with the above results of the WSI and WHC.
O stretching vibrations and N–H bending vibrations, namely, protein secondary structure conformation.69 The presence of bound water was indicated by the peak observed at 1650 cm−1.79 Several weaker wave peaks at 1965 and 2150–2200 cm−1 belonged to the C–O bound folded state of the protein and the triple bond of alkyne stretching, respectively.72 The vibration at 2850 cm−1 corresponded to the helical structures formed by AA (lysine and arginine) and the turn structure of proline.72,84 Moreover, the peak profiles of 2800–3000 (the symmetric stretching mode of C–H bonds in the aliphatic side chain and aldehyde functional groups, including methyl and methylene) and 3100–3500 cm−1 (the vibration of amide A; hydroxyl groups) corresponded to the hydrophobicity and hydrophilicity of RPP, respectively.69,72,78,81 Yet, there was no evident change in the peak profiles of all groups, whereas the light spectrum increased with the increased stress of HPP, which agreed with the above results of the WSI and WHC.
This study showed that the diffraction peaks at 15°, 17°, 18° and 23° (2θ) for all groups were typically characteristic of the A-type crystalline structure of rice starch (Fig. 3H), which was consistent with the results reported by Chi et al.85 and Lu et al.31 However, increased HPP stress led to observing minor peaks at 20°–21° in the groups exposed to 400 and 600 MPa, indicating the formation of V-type crystallites. The observed phenomenon could be attributed to HPP and heat exposure, and the partial disruption of non-covalent bonds, including hydrogen and ionic bonds, within the intermolecular chains of few starch molecules in RPP.86 These disruptions alter the orientation and arrangement of starch molecules during crystallization, thereby perturbing the crystalline structure.85–87 Furthermore, this phenomenon also facilitates the endogenous assembly of lipid–amylose in the RPP, forming complex V-type crystalline structures.85,87 It is worth mentioning that Zhang et al.88 reported higher crystallinity of RP-free rice starch through gelatinization, which decreased with the incorporated RP contents. Lu et al.31 also reported that the complexation of rice starch with RP also caused a pronounced decrease in the relative crystalline value, namely, altering the rice starch's crystalline structure.
The present study showed that the RPP particles in all samples exhibited comparable characteristics, featuring smooth and continuous lamellar surfaces with irregular porosity and grooves (Fig. 3I).73,89 This observation suggests that HPP does not remarkably impact the shape of the protein particles; instead, higher stresses appear to result in a transition from loose grooves to tighter ones. This observed phenomenon can be attributed to the spray drying process of RP during manufacturing, wherein the elevated temperature of the hot air accelerates water evaporation by increasing the thermal energy transferred to protein-bound water (or droplets).73,90 In addition, water vapor moves from the center of the protein particle to its surface, preventing the internal pressure from distorting or expanding to form inward collapsing blowholes, namely, hollow craters,73,91 as observed by SEM. Moreover, it has been reported that HPP is unaffected by the presently achievable stress range for the primary structure of proteins, whereas the secondary structure may undergo minimal changes.37,44 Conversely, the compression affects tertiary and quaternary structures by disrupting non-covalent bonds such as hydrogen bonds or the exposure of hydrophobic groups.37 However, it does not impact covalent bonds, thus preserving the primary chemical integrity of flavoring compounds and colors in foodstuffs without altering their original flavor and nutritional properties, while the HPP finds extensive applications.37,92
3.3. Effects of HPP-treated RPP on the growth performance of the experimental animals
This study showed that the BW in all rats at 1–2 weeks was elevated in all four groups of induced DCM compared to the control group (Fig. 4A). This phenomenon was hypothesized to be related to the initial overeating of DM, which increased the BW. Unsurprisingly, the BW performance of the control group increased to the highest level as time increased to the end of the experiment. While the BW of the RPP-supplemented rats in the three groups was substantially higher than that of the DCM group, albeit lower than that of the control group (Fig. 4B), all groups exhibited a significantly different performance (p < 0.05). These phenomena mentioned above revealed that RPP was effective in controlling the BW with a decreased risk of obesity in rats, and the trends agreed with the results reported by Liu et al.93 Concurrently, the authors also noted that the influence of age on experimental animals did not limit the effects of RP. This study also suggested that the growth and metabolism of the three experimental animal groups fed RPP-prepared ES remained unaffected.94,95In addition, the heart weights of rats in each group were also consistently associated with the above trend of BW changes (Fig. 4C), where the DM group was the lowest, and there were significant differences among all groups (p < 0.05). The heart weight to BW ratio was the highest in the DCM group (Fig. 4D), whereas there were significant differences between the groups (p < 0.05), but the three RPP groups’ ratios were closer to the control group. The results showed that continuous supplementation of RPP effectively reduced the heart-to-body weight ratio in DCM rats, indicating a potential reversal of cardiac inflammation or attenuation of symptoms.2,96 Consequently, this intervention facilitated the restoration of impaired cardiac cells caused by DCM, thereby enhancing cardiac pump efficiency.2 Li et al.97 also revealed that obese T2DM mice exhibited pronounced myocardial lipotoxicity, characterized by enhanced uptake of free fatty acids and accumulation of ROS, glycogen, and lipid droplets. It is worth mentioning that excessive accumulation of free fatty acids in non-adipocytes can lead to glucose and lipid metabolic disorders, namely lipotoxicity and insulin resistance.97,98 However, clinical evidence has demonstrated myocardial lipid accumulation in patients’ hearts with insulin resistance and hyperglycemia.13
In terms of blood glucose levels, the results of this study showed that the DCM group exhibited the maximum (Fig. 4E). This also suggested that insulin utilization was impaired in the rats in the DCM group.99 Namely, the pattern of induction of DCM in this study was successfully established. In contrast, the blood glucose levels of the RPP groups decreased with increased HPP pressure, similar to the control group, whereas there were significant differences between all groups (p < 0.05). These phenomena also implied that continuous supplementation of high-pressure (600 MPa) treatment RPP of ES for five months facilitated the restoration of blood glucose levels in T2DM rats. Specifically, it alleviates impaired insulin utilization in rats.99 Notably, Higuchi et al.28 reported that rice endosperm protein administration was associated with markedly decreased fasting glucose, plasma insulin, and homeostatic model assessment of insulin resistance in adult mice fed a high-fat diet for 12 weeks. The authors also showed that the protein source provided to juvenile mice for six weeks substantially influenced the susceptibility to obesity and associated diseases induced by a high-fat diet during adulthood. It has also been reported that sustained hyperglycemia stimulates the production of ROS, consequently promoting the secretion of pro-inflammatory cytokines in DCM.1 Therefore, this study's results revealed that the elevated pressure of high-pressure (600 MPa) HPP-treated RPP of ES positively restored growth performance and blood glucose levels in DCM rats.
3.4. Effects of HPP-treated RPP on the serum biochemical parameters and myocardial infarction (MI) indices of the experimental animals
This study showed that the TG, TC, AST, and LDH levels in the three RPP groups were lower than those in the DCM group (Fig. 5A–D), while the values were closer to the control group. In particular, the RPP600 group exhibited more favorable results, and there were significant differences between the groups (p < 0.05). This study showed abnormalities in serum biochemical parameters in the DCM group, the basic module of metabolic disorders in rats.100 According to the report, apart from TC and TG, there is also a concomitant elevation in the low-density lipoprotein-cholesterol level.100Yang et al.94 provided evidence that the administration of RP reduced TG and cholesterol levels. This outcome was correlated with decreased protein digestibility and increased fecal excretion of neutral sterols. Notably, this mechanism was facilitated by converting cholesterol into bile acids within the body through enterohepatic circulation.94 Interestingly, Liu et al.93 also reported that continuous supplementation of RP for two weeks exhibited the potential to reduce TG levels in both plasma and liver among juvenile and adult rats. However, the results of a four-week clinical trial using rice endosperm protein, as Nakayama et al.101 reported, did not demonstrate any significant positive effects on subjective mood states and the insomnia severity index. The lack of efficacy may be attributed to the relatively short intervention period.101
This study also showed that AST was the highest in the DCM group, meaning that liver function and myocardial cells were impaired, but RPP supplementation effectively inhibited liver and myocardial cell injury in DCM rats. In contrast, AST exhibited a decrease in the three RPP groups, which agreed with the results reported by Gilani et al.100 and Wang et al.99 AST is distributed in cardiac, liver, and skeletal muscle cells.99 In addition, hepatic enzymes in the bloodstream, originating from the liver cytosol, indicate hepatocyte damage.100 Kalpanadevi et al.102 also reported that enzymes such as AST, alanine aminotransferase (ALT), and alkaline phosphatase (ALP) primarily aid in the breakdown of proteins by the liver, which were relatively high in rats supplemented with rice bran protein. In contrast, AST and ALT were typically elevated in response to acute hepatotoxicity, yet tended to decline with prolonged intoxication.102 Notably, LDH, TG, cholesterol, and CK play critical roles in understanding cardiac function, where LDH catalyzes the conversion of lactate to pyruvate, an important step in cellular energy production.102,103 Creatine kinase serves as an essential enzyme modulator in the production and utilization of high-energy phosphates in the contractile tissues.102,103 Kalpanadevi et al.102 reported that rats supplemented with rice bran protein exhibited markedly increased levels of LDH and enzymes such as creatine kinase and decreased cholesterol levels. Conversely, elevated cholesterol, CK, and LDH levels indicated that the control rats were more susceptible to cardiac dysfunction in the presence of protein malnutrition,102 which was comparable to the results observed in the present study. Therefore, the ameliorative effect of bioactive substances on cardiotoxicity was evaluated as a decrease in the serum levels of creatine phosphokinase, CK-MB, and LDH, which also included a reduction in the level of MDA in the OS index.104
Moreover, the same trends described above were observed for MI indices (TnT and CK-MB) in DCM rats of three RPP groups following continuous HPP-treated RPP flour-produced ES supplementation for five months (Fig. 5E and F). Specifically, the RPP 600 MPa group exhibited satisfactory performance in recovering the TnT content, with significant differences among the groups (p < 0.05). In contrast, the CK-MB activities of the three RPP groups were not significantly different from each other, and they were lower than that of the DCM group (p < 0.05). These phenomena were attributed to the structural and property changes in RP flour post-HPP treatment and the recognition of specific AA sequences during hydrolysis by enzymes in the rat's digestive tract, producing peptides with potent bioactivities (such as antioxidant capacities).25 However, the findings of this study indicated that the bioactivity of RPP played a crucial role in eliminating MI indices and alleviating irreversible myocardial damage induced by DCM. Thereby, the OS risk of heart failure was reduced, which was associated with elevated levels of TnT and CK-MB.2,105 Hence, this study suggested that ES produced from HPP-treated RPP flour supplemented for five consecutive months contributed to the recovery of serum biochemical parameters and MI indices in patients with DCM, contributing to health promotion.
3.5. Effects of HPP-treated RPP on the cardiac oxide-inflammatory parameters of the experimental animals
This study showed a positive correlation between the reduction of MDA content in rat serum in the three RPP groups and the pressure of HPP used (Fig. 6A), with significant differences among all groups (p < 0.05). Namely, the RPP600 group exhibited the best effect in reducing the MDA content, which was closer to the value of the control group. This finding also correlated with the antioxidant ability of HPP-treated RPP flour, as described above, whereas the highest MDA content, as expected, was found in the DCM group. It has been reported that these advances and related issues regarding DCM were attributable to an increase in MDA caused by OS, while a decrease in MDA levels could be attributed to the contribution of antioxidant enzymes (SOD and CAT).100,106 Specifically, it also confirmed that HPP-treated RPP-prepared ES exhibited antioxidant ability similar to the results reported by Gilani et al.100 and this was also consistent with the results of this study mentioned in section 3.2. This also implied that the RPP (peptide or AA) of the ES in this study still had antioxidant capacity after being metabolized by rat digestion, and the LMW of the hydrolysate (<10 kDa) following digestion also proved to exert more excellent antioxidant activity.107 The limitations of the present study lie in the absence of further confirmation regarding the distribution of undigested RPP and intestinal flora in the rat colon. This hinders our ability to elucidate both beneficial and adverse health effects resulting from the products derived from fermentative hydrolysis of these proteins (peptide or AA).108In terms of several cardiac oxide-inflammatory parameters (including CAT, GPx, GSH, and SOD), this study showed that all three RPP groups were significantly improved compared to the DCM group while they were closer to the control group (p < 0.05) (Fig. 6B–E). In particular, the 600 MPa group showed a satisfactory improvement. These findings indicated that in the DCM group, the continuous elevation of OS and the subsequent inflammatory response exhausted the endogenous antioxidants (such as SOD and GSH-Px) in the heart, thereby leading to cardiomyocyte damage.1
However, replenishing exogenous antioxidants through five consecutive months of supplementation with RPP-prepared ES effectively mitigated OS damage to cardiomyocytes, enhancing their antioxidant capabilities. This was similar to our previous report on the improvement and benefit of supplementation with extruded djulis snacks on DCM.2 Furthermore, dietary supplementation with RP has been reported to enhance endogenous antioxidant capacity (up-regulation of expression levels of SOD, GPx, etc.) in both juvenile and adult rats, while reducing the hepatic accumulation of MDA, carbonylated proteins, and ROS, leading to a decrease in the damage caused by OS.109 It has also been reported that bioactive peptides obtained from the enzymatic hydrolysis of whey proteins can also exert antioxidant effects in Caco-2 cell lines by enhancing the levels of both GSH and SOD, thereby safeguarding intestinal cells against OS.110,111 Therefore, the findings revealed the beneficial levels of all related antioxidant status. Thus, modifying the ES formula composition by incorporating the RPP reduced OS risk due to DCM, which aligns with the extruded djulis snack results previously reported by this team.2
Well known as a critical executive molecule in apoptosis, caspase-3 is activated in the early stages and cleaves the corresponding cytoplasmic and cytosolic substrates, leading to apoptosis. Notably, the activity of caspase-3 is dramatically reduced in the late stages of apoptosis and dead cells.112 This study showed that all three supplemental RPP groups exhibited a substantial reduction in caspase-3 levels compared to the DCM group (Fig. 6F), and there were significant differences among the groups (p < 0.05). Specifically, the decrease in the RPP600 group was closest to the control group. These phenomena can be attributed to the antioxidant ability linked to HPP-treated RPP flour, which mitigates OS in the myocardial tissue cells from T2DM-induced inflammation.
Conversely, for other cardiac oxide-inflammatory parameters such as IL-6, IL-1β, TNF-α, and NF-κB, this study showed that the three groups exhibited statistically significantly lower levels of these inflammatory parameters in the RPP group than in the DCM group (p < 0.05) (Fig. 6G–J). Specifically, the RPP600 group exhibited the greatest reduction, close to that of the control group. These results showed that by reducing the production of cardiac oxide-inflammatory parameters were alleviated, namely decreased levels of inflammatory cytokines (IL-6, IL-1β, and TNF-α) in the cardiac tissues of the DCM group.1,2
Subsequently, the aberrant accumulation of varying growth factors activated the NF-κB signaling pathway in cardiovascular disease, eventually contributing to cardiomyocyte pyroptosis.7,11 However, continuous supplementation of ES for 5 months in the three RPP groups exhibited a restraining effect on the aforementioned inflammatory cytokines and facilitated a healthy recovery of the myocardium.1,2 It is noteworthy that excessive free radical production has been reported to promote NF-κB triggering, which in turn contributes to gene transcription of encrypting inflammatory proteins.100 Wang et al.113 also reported that feeding RP for two weeks effectively down-regulated the hepatic protein expression and mRNA levels of NF-B1 in juvenile and adult rats, thereby inhibiting the activation of the NF-B metabolic pathway. The same authors also stated that RP supplementation for two weeks showed a decrease in the mRNA levels of pro-inflammatory cytokines [IL-1, IL-6, TNF-α, inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), and monocyte chemoattractant protein-1 (MCP-1)]. In addition, Wen et al.114 showed that trypsin-derived RP hydrolysates inhibited LPS-induced inflammatory responses and suppressed the NF-κB signaling pathway in RAW264.7 macrophages by inhibiting nitric oxide (NO) release and decreasing IL-1β, IL-6, TNF-α, and iNOS expression in a dose-dependent manner. However, increased NF-κB levels were also observed in the DCM group of this study, but effective mitigation of this phenomenon was also found in the three RPP groups by supplementation with RPP-prepared ES.
Moreover, it has been reported that rice endosperm protein administration also decreased the serum levels of IL-1β, IL-6, leptin, and lipopolysaccharide-binding protein in high-fat diet-induced obese C57BL/6J male mice (12 week old adults).28 The same authors also stated that in the serum, kidneys, and liver of the fed mice (6 week old juvenile), it was observed that RP intake contributed to lowering the levels of TNF-α and monocyte chemoattractant protein-1 apart from the biomarkers mentioned above. In addition, Ge et al.45 reported that egg white peptide supplementation ameliorated colonic inflammatory symptoms and modulated inflammatory factors, such as IL-6, in mice with ulcerative colitis attributed to altered gut flora. Notably, all of these changes in inflammatory parameters were also involved in the composition of the intestinal microbiota and variations in the levels of SCFAs.47 Galland et al.111 also reported that LMW (<5 kDa) peptides derived from whey protein isolate through digestive hydrolysis contributed to the attenuation of inflammation in astrocytes and microglia while enhancing IL-6 and TNF-α mRNA expression, thereby reducing the levels of these cytokines. Interestingly, a similar phenomenon has been observed in the Parkinson's disease model, wherein microglia exhibit heightened activation and release inflammatory molecules (such as IL-1β, IL-6, TNF-α, IL-18, and chemokines), thereby inducing neuronal damage.112
Protein digestion in the gastrointestinal tract involves complex mechanisms, while proteins are broken down and AA is released into the stomach. Then, partially digested proteins enter the small intestine along with other gastric contents for absorption, where they undergo hydrolysis by additional proteolytic enzymes to yield oligopeptides and free AA.115 Further investigation is necessary to comprehensively analyze the peptide and AA composition and the distribution of hydrolyzed substances during digestion to address the current limitations and ensure a more comprehensive understanding of these factors. However, the above results suggested that continuous supplementation of ES prepared from HPP-treated RPP flour may be a promising approach for mitigating DCM-induced cardiac oxidative-inflammatory parameters. Therefore, it also implies that patients can supplement or incorporate foods prepared with HPP-treated RPP flour in their daily diets to mitigate cardiac oxide inflammation caused by DCM, apart from receiving conventional therapeutic medications. Moreover, integrating HPP-RPP ES into their dietary regimen during hypoglycemia episodes can enhance general well-being.
4. Conclusions
This study revealed that HPP treatment for RPP enhanced the physicochemical properties. Incorporating RPP of ES formulation also proved that RPP ES's glucose modulation and antioxidant potential alleviated the OS in DCM and performed satisfactorily to improve cardiac oxide-inflammatory parameters. Specifically, five consecutive months of dietary supplementation effectively ameliorated OS-related myocardial damage in DCM rats. However, the detailed mechanism underlying the RPP-mediated NF-κB metabolic pathway and the extent of involvement of the gut microbiota remain unclear, necessitating further investigation. Another limitation of the present study was the validation phase of the animal model. It was necessary to examine whether consistent effects were maintained through human clinical trials before sufficient information could be obtained about the actual contribution of RPP-prepared ES. Therefore, this study offers valuable insights and information regarding the utilization of HPP for processing, enhancement of bioactive functionality, and integration with existing technologies.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this study.Data availability
The data that support the findings of this study are available from the corresponding author, Po-Hsien Li, upon reasonable request.Supplementary information is available. See DOI: https://doi.org/10.1039/d5fo01477h
Acknowledgements
This research was financially supported by the Taichung Veterans General Hospital/Providence University Joint Research Program (TCVGH-PU-1128102), Taiwan.References
- J. Zou, D. Sui, W. Fu, Y. Li, P. Yu, X. Yu and H. Xu, Total flavonoids extracted from the leaves of Murraya paniculata (L.) Jack alleviate oxidative stress, inflammation and apoptosis in a rat model of diabetic cardiomyopathy, J. Funct. Foods, 2021, 76, 104319 CrossRef CAS.
- Y.-T. Cheng, W.-C. Lu, Y.-J. Chan, P.-H. Huang, P.-Y. Chiang, R.-S. Chen and P.-H. Li, Effect of extruded djulis (Chenopodium formosanum) snacks on the ameliorative potential against diabetic cardiomyopathy, J. Funct. Foods, 2024, 116, 106154 CrossRef CAS.
- G. Murtaza, H. U. H. Virk, M. Khalid, C. J. Lavie, H. Ventura, D. Mukherjee, V. Ramu, S. Bhogal, G. Kumar, M. Shanmugasundaram and T. K. Paul, Diabetic cardiomyopathy - A comprehensive updated review, Prog. Cardiovasc. Dis., 2019, 62, 315–326 CrossRef PubMed.
- N. J. Byrne, N. S. Rajasekaran, E. D. Abel and H. Bugger, Therapeutic potential of targeting oxidative stress in diabetic cardiomyopathy, Free Radicals Biol. Med., 2021, 169, 317–342 CrossRef CAS PubMed.
- V. Rizza, L. Tondi, A. M. Patti, D. Cecchi, M. Lombardi, F. Perone, M. Ambrosetti, M. Rizzo, D. Cianflone and F. Maranta, Diabetic cardiomyopathy: pathophysiology, imaging assessment and therapeutical strategies, Int. J. Cardiol. Cardiovasc. Risk Prev., 2024, 23, 200338 Search PubMed.
- E. A. E. Farrag, M. O. Hammad, S. M. Safwat, S. Hamed and D. Hellal, Artemisinin attenuates type 2 diabetic cardiomyopathy in rats through modulation of AGE-RAGE/HMGB-1 signaling pathway, Sci. Rep., 2023, 13, 11043 CrossRef CAS PubMed.
- T. Nishikawa, D. Edelstein, X. L. Du, S.-i. Yamagishi, T. Matsumura, Y. Kaneda, M. A. Yorek, D. Beebe, P. J. Oates, H.-P. Hammes, I. Giardino and M. Brownlee, Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage, Nature, 2000, 404, 787–790 CrossRef CAS PubMed.
- R. A. Rasheed, A. S. Embaby, F. A. A. Elkhamisy, M. O. Mohamed, A. M. Abbasi, W. B. A. Ghaly, M. M. A. Elshaer, K. S. Almaary and M. A. M. Almoatasem, Ascorbic acid alleviates oxidative stress and improves major salivary glands’ structure and function in diabetic rats: A histological and immunohistochemical study, J. King Saud Univ., Sci., 2022, 34, 102273 CrossRef.
- E. Radzioch, B. Dąbek, M. Balcerczyk-Lis, W. Frąk, P. Fularski, E. Młynarska, J. Rysz and B. Franczyk, Diabetic cardiomyopathy—From basics through diagnosis to treatment, Biomedicines, 2024, 12, 765 CrossRef CAS PubMed.
- X. Song, C. Fan, C. Wei, W. Yu, J. Tang, F. Ma, Y. Chen and B. Wu, Mitochondria fission accentuates oxidative stress in hyperglycemia-induced H9c2 cardiomyoblasts in vitro by regulating fatty acid oxidation, Cell Biol. Int., 2024, 48, 1378–1391 CrossRef CAS PubMed.
- Z. Zhang, Z. Yang, S. Wang, X. Wang and J. Mao, Overview of pyroptosis mechanism and in-depth analysis of cardiomyocyte pyroptosis mediated by NF-κB pathway in heart failure, Biomed. Pharmacother., 2024, 179, 117367 CrossRef CAS PubMed.
- G. Borghetti, D. von Lewinski, D. M. Eaton, H. Sourij, S. R. Houser and M. Wallner, Diabetic cardiomyopathy: Current and future therapies. Beyond glycemic control, Front. Physiol., 2018, 9, 1–15 Search PubMed.
- R. Marfella, C. Sardu, G. Mansueto, C. Napoli and G. Paolisso, Evidence for human diabetic cardiomyopathy, Acta Diabetol., 2021, 58, 983–988 CrossRef PubMed.
- H. Bhagani, S. A. Nasser, A. Dakroub, A. F. El-Yazbi, A. A. Eid, F. Kobeissy, G. Pintus and A. H. Eid, The mitochondria: A target of polyphenols in the treatment of diabetic cardiomyopathy, Int. J. Mol. Sci., 2020, 21, 4962 CrossRef CAS PubMed.
- G. Yang, Z. Liu, S. Dong, X. Zhao, Z. Ge, Z. Cheng, X. Zhang and K. Wang, Duodenal-jejunal bypass surgery activates eNOS and enhances antioxidant system by activating AMPK pathway to improve heart oxidative stress in diabetic cardiomyopathy rats, J. Diabetes, 2024, 16, e13516 CrossRef CAS PubMed.
- S. A. Tadesse and S. A. Emire, Production and processing of antioxidant bioactive peptides: A driving force for the functional food market, Heliyon, 2020, 6, e04765 CrossRef CAS PubMed.
- I. Shorstkii, E. H. A. Mounassar, U. Bindrich, V. Heinz and K. Aganovic, Influence of High Hydrostatic Pressure and Pulsed Electric Field Treatment on moisture absorption of wheat grains, Chem. Ing. Tech., 2022, 96, 1098–1106 CrossRef.
- Y. Wang, C.-m. Ma, Y. Yang, B. Wang, X.-f. Liu, Y. Wang, X. Bian, G. Zhang and N. Zhang, Effect of high hydrostatic pressure treatment on food composition and applications in food industry: A review, Food Res. Int., 2024, 195, 114991 CrossRef CAS.
- Q. Xia and Y. Li, Mild high hydrostatic pressure pretreatments applied before soaking process to modulate wholegrain brown rice germination: An examination on embryo growth and physicochemical properties, Food Res. Int., 2018, 106, 817–824 CrossRef CAS PubMed.
- N. C. Santos, R. L. J. Almeida, G. M. da Silva, S. S. Monteiro, V. H. de Alcântara Ribeiro, A. P. de França Silva, V. M. de Alcântara Silva, L. M. de Sousa Rodrigues, A. M. M. C. N. André and M. M. de Almeida Mota, Influence of high hydrostatic pressure (HHP) pretreatment on plum (Prunus salicina) drying: Drying approach, physical, and morpho-structural properties of the powder and total phenolic compounds, J. Food Process. Preserv., 2022, 46, e16968 CAS.
- P.-H. Huang, Y.-T. Cheng, H.-C. Hsieh, W.-C. Ko, W.-C. Lu and P.-H. Li, Effects of transglutaminase on the physicochemical properties of surimi and kamaboko prepared by thermal and low level-pressure treatments, LWT–Food Sci. Technol., 2023, 183, 114863 CrossRef CAS.
- Q. Xia, L. Wang, C. Xu, J. Mei and Y. Li, Effects of germination and high hydrostatic pressure processing on mineral elements, amino acids and antioxidants in vitro bioaccessibility, as well as starch digestibility in brown rice (Oryza sativa L.), Food Chem., 2017, 214, 533–542 CrossRef CAS PubMed.
- J. E. Dominguez-Ayala, A. Soler, G. Mendez-Montealvo and G. Velazquez, Supramolecular structure and technofunctional properties of starch modified by high hydrostatic pressure (HHP): A review, Carbohydr. Polym., 2022, 291, 119609 CrossRef CAS.
- M. Chalamaiah, S. K. Ulug, H. Hong and J. Wu, Regulatory requirements of bioactive peptides (protein hydrolysates) from food proteins, J. Funct. Foods, 2019, 58, 123–129 CrossRef CAS.
- Y. Zhang, X. Jing, Z. Chen and X. Wang, Purification and identification of antioxidant peptides from millet gliadin treated with high hydrostatic pressure, LWT–Food Sci. Technol., 2022, 164, 113654 CrossRef CAS.
- L. Amagliani, J. O'Regan, A. L. Kelly and J. A. O'Mahony, The composition, extraction, functionality and applications of rice proteins: A review, Trends Food Sci. Technol., 2017, 64, 1–12 CrossRef CAS.
- L. Zheng, J. M. Regenstein, Z. Wang, H. Zhang and L. Zhou, Reconstituted rice protein: The raw materials, techniques and challenges, Trends Food Sci. Technol., 2023, 133, 267–276 CrossRef CAS.
- Y. Higuchi, M. Hosojima, H. Kabasawa, S. Kuwahara, S. Goto, K. Toba, R. Kaseda, T. Tanaka, N. Kitamura, H. Takihara, S. Okuda, M. Taniguchi, H. Arao, I. Narita and A. Saito, Rice endosperm protein administration to juvenile mice regulates gut microbiota and suppresses the development of high-fat diet-induced obesity and related disorders in adulthood, Nutrients, 2019, 11, 2919 CrossRef PubMed.
- A. A. Zaky, A. M. Abd El-Aty, A. Ma and Y. Jia, An overview on antioxidant peptides from rice bran proteins: extraction, identification, and applications, Crit. Rev. Food Sci. Nutr., 2020, 62, 1350–1362 CrossRef PubMed.
- S. Helmyati and M. Wigati, in Functional Foods and Nutraceuticals in Metabolic and Non-Communicable Diseases, ed. R. B. Singh, S. Watanabe and A. A. Isaza, Academic Press, 2022, pp. 151–162, DOI:10.1016/B978-0-12-819815-5.00005-7.
- X. Lu, R. Ma, J. Zhan, Z. Jin and Y. Tian, Mechanism of peptides from rice hydrolyzed proteins hindering starch digestion subjected to hydrothermal treatment, NPJ Sci. Food., 2022, 6, 37 CrossRef PubMed.
- MarketDataForecast, Global rice protein market size (2024 to 2032), https://www.marketdataforecast.com/market-reports/rice-protein-market (accessed on 11 Nov 2024).
- D. J. McClements and L. Grossmann, A brief review of the science behind the design of healthy and sustainable plant-based foods, NPJ Sci. Food., 2021, 5, 17 CrossRef PubMed.
- J. Jang and D.-W. Lee, Advancements in plant based meat analogs enhancing sensory and nutritional attributes, NPJ Sci. Food., 2024, 8, 50 CrossRef PubMed.
- S. L. R. Meza, L. Cañizares, B. Dannenberg, B. B. Peres, L. A. Rodrigues, C. Mardade, M. A. de Leon, C. A. Gaioso, I. Egea and M. de Oliveira, Sustainable rice bran protein: Composition, extraction, quality properties and applications, Trends Food Sci. Technol., 2024, 145, 104355 CrossRef.
- J. P. D. Wanasundara, H. Kumagai, A. P. Kasch, L. Van Gels and S. Peirce, in Sustainable Protein Sources, ed. S. Nadathur, J. P. D. Wanasundara and L. Scanlin, Academic Press, 2nd edn, 2024, pp. 55–75, DOI:10.1016/B978-0-323-91652-3.00008-3.
- L. Zheng, Y. San, Y. Xing and J. M. Regenstein, Rice proteins: A review of their extraction, modification techniques and applications, Int. J. Biol. Macromol., 2024, 268, 131705 CrossRef CAS PubMed.
- T. Kumagai, H. Kawamura, T. Fuse, T. Watanabe, Y. Saito, T. Masumura, R. Watanabe and M. Kadowaki, Production of rice protein by alkaline extraction improves its digestibility, J. Nutr. Sci. Vitaminol., 2006, 52, 467–472 CrossRef CAS PubMed.
- M. Kubota, Y. Saito, T. Masumura, T. Kumagai, R. Watanabe, S. Fujimura and M. Kadowaki, Improvement in the in vivo digestibility of rice protein by alkali extraction is due to structural changes in prolamin/protein Body-I particle, Biosci., Biotechnol., Biochem., 2010, 74, 614–619 CrossRef CAS PubMed.
- M. Hosojima, H. Shimada, Y. Obi, S. Kuwahara, R. Kaseda, H. Kabasawa, H. Kondo, M. Fujii, R. Watanabe, Y. Suzuki, M. Kadowaki, S. Miyazaki and A. Saito, A randomized, double-blind, crossover pilot trial of rice endosperm protein supplementation in maintenance hemodialysis patients, Sci. Rep., 2017, 7, 18003 CrossRef PubMed.
- T. Kumagai, R. Watanabe, M. Saito, T. Watanabe, M. Kubota and M. Kadowaki, Superiority of alkali-extracted rice protein in bioavailability to starch degraded rice protein comes from digestion of prolamin in growing rats, J. Nutr. Sci. Vitaminol., 2009, 55, 170–177 CrossRef CAS PubMed.
- C. Gong, C. Jiao, H. Liang, Y. Ma, Q. Wu and Y. Xie, Exome-based amino acid optimization: A dietary strategy to satisfy human nutritional demands and enhance muscle strength in breast tumor mice undergoing chemotherapy, J. Agric. Food Chem., 2024, 72, 7089–7099 CrossRef CAS PubMed.
- R. J. Klement, The optimal amino acid pattern for humans and its implications for nutrition of cancer patients, Transl Breast Cancer Res., 2024, 5, 25–30 CrossRef PubMed.
- F. Rivero-Pino, F. J. Espejo-Carpio and E. M. Guadix, Antidiabetic food-derived peptides for functional feeding: Production, functionality and in vivo evidences, Foods, 2020, 9, 983 CrossRef CAS PubMed.
- H. Ge, T. Li, Q. Yang, Y. Tang, J. Liu, Y. Yu and T. Zhang, Egg white peptides administration in enhancing pathological immune response and regulating intestinal bacteria abundance: A new strategy for relieving young mice colitis, Food Front., 2023, 4, 782–794 CrossRef CAS.
- C.-Y. Hou, S. H. Hazeena, S.-L. Hsieh, J.-Y. Ciou, C.-W. Hsieh, M.-K. Shih, M.-H. Chen, C.-W. Tu and P.-H. Huang, Investigation of the optimal production conditions for egg white hydrolysates and physicochemical characteristics, J. Food Sci. Technol., 2023, 60, 1600–1611 CrossRef CAS PubMed.
- Y. Zhou, D. Wang, J. Guo, Y. Zheng, H. Duan and W. Yan, Two protein-derived peptides from Bombyx mori attenuate colitis by enhancing intestinal barrier function and modulating gut microbiota ecology, Food Front., 2024, 5, 2652–2671 CrossRef CAS.
- E. B.-M. Daliri, D. H. Oh and B. H. Lee, Bioactive peptides, Foods, 2017, 6, 32 CrossRef PubMed.
- S. Guha and K. Majumder, Structural-features of food-derived bioactive peptides with anti-inflammatory activity: A brief review, J. Food Biochem., 2019, 43, e12531 CrossRef PubMed.
- H. Li, J. Wu, J. Wan, Y. Zhou and Q. Zhu, Extraction and identification of bioactive peptides from Panxian dry-cured ham with multifunctional activities, LWT–Food Sci. Technol., 2022, 160, 113326 CrossRef CAS.
- D. Wang, F. Cheng, Y. Wang, J. Han, F. Gao, J. Tian, K. Zhang and Y. Jin, The changes occurring in proteins during processing and storage of fermented meat products and their regulation by lactic acid bacteria, Foods, 2022, 11, 2427 CrossRef CAS PubMed.
- Q. Guo, P. Chen and X. Chen, Bioactive peptides derived from fermented foods: Preparation and biological activities, J. Funct. Foods, 2023, 101, 105422 CrossRef CAS.
- D. Mondal, M. Awana, S. Mandal, K. Pandit, A. Singh, C. O. Syeunda, S. J. Thandapilly and V. Krishnan, Functional foods with a tailored glycemic response based on food matrix and its interactions: Can it be a reality?, Food Chem.: X, 2024, 22, 101358 CAS.
- AOAC, in Official Methods of Analysis of AOAC INTERNATIONAL, ed. G. W. Latimer Jr. and G. W. Latimer Jr., Oxford University Press, 2023, DOI:10.1093/9780197610145.002.001.
- C. Dinnella, M. T. Gargaro, R. Rossano and E. Monteleone, Spectrophotometric assay using o-phtaldialdehyde for the determination of transglutaminase activity on casein, Food Chem., 2002, 78, 363–368 CrossRef CAS.
- P.-H. Huang, C.-S. Chiu, W.-C. Lu and P.-H. Li, Effect of compositions on physicochemical properties and rheological behavior of gelatinized adzuki-bean cake (Yokan), LWT–Food Sci. Technol., 2022, 168, 113870 CrossRef CAS.
- T.-H. Guo, P.-H. Huang, C.-W. Hsieh, C.-K. Chang, C.-Y. Huang, S.-L. Hsieh, M.-K. Shih and C.-Y. Hou, Effects on the quality in using freezing equipment (weak oscillating magnetic field) on hybrid giant tiger grouper (Epinephelus fuscoguttatus, × Epinephelus lanceolatus) fillets, J. Agric. Food Res., 2024, 15, 101002 CAS.
- Y. Guo, H. Hu, Q. Wang and H. Liu, A novel process for peanut tofu gel: Its texture, microstructure and protein behavioral changes affected by processing conditions, LWT–Food Sci. Technol., 2018, 96, 140–146 CrossRef CAS.
- S. Ou, K. C. Kwok, Y. Wang and H. Bao, An improved method to determine SH and –S–S– group content in soymilk protein, Food Chem., 2004, 88, 317–320 CrossRef CAS.
- C.-Y. Wang, Y.-W. Chen and C.-Y. Hou, Antioxidant and antibacterial activity of seven predominant terpenoids, Int. J. Food Prop., 2019, 22, 230–238 CrossRef CAS.
- U. Asma, M. L. Bertotti, S. Zamai, M. Arnold, R. Amorati and M. Scampicchio, A kinetic approach to oxygen radical absorbance capacity (ORAC): restoring order to the antioxidant activity of hydroxycinnamic acids and fruit juices, Antioxidants, 2024, 13, 222 CrossRef CAS PubMed.
- R. L. Prior, Oxygen radical absorbance capacity (ORAC): New horizons in relating dietary antioxidants/bioactives and health benefits, J. Funct. Foods, 2015, 18, 797–810 CrossRef CAS.
- C.-H. Lin, Y.-T. Huang, J.-Y. Ciou, C.-M. Cheng, G.-T. Wang, C.-M. You, P.-H. Huang and C.-Y. Hou, Circular economy and sustainable recovery of Taiwanese tilapia (Oreochromis mossambicus) byproduct—The large-scale production of umami-rich seasoning material application, Foods, 2023, 12, 1921 CrossRef CAS PubMed.
- Y.-T. Cheng, P.-H. Huang, Y.-J. Chan, P.-Y. Chiang, W.-C. Lu, C.-W. Hsieh, Z.-C. Liang, B.-W. Yan, C.-C. R. Wang and P.-H. Li, Investigate the composition and physicochemical properties attributes of banana starch and flour during ripening, Carbohydr. Polym. Technol. Appl., 2024, 100446, DOI:10.1016/j.carpta.2024.100446.
- M.-Y. Chou, Y.-C. Wong, S.-Y. Wang, C.-H. Chi, T.-H. Wang, M.-J. Huang, P.-H. Huang, P.-H. Li and M.-F. Wang, Potential antidepressant effects of a dietary supplement from Huáng qí and its complex in aged senescence-accelerated mouse prone-8 mice, Front. Nutr., 2023, 10, 1–12 Search PubMed.
- A. Quirós, R. Chichón, I. Recio and R. López-Fandiño, The use of high hydrostatic pressure to promote the proteolysis and release of bioactive peptides from ovalbumin, Food Chem., 2007, 104, 1734–1739 CrossRef.
- P. Garcia-Mora, E. Peñas, J. Frias, H. Zieliński, W. Wiczkowski, D. Zielińska and C. Martínez-Villaluenga, High-Pressure-Assisted enzymatic release of peptides and phenolics increases angiotensin converting enzyme I inhibitory and antioxidant activities of pinto bean hydrolysates, J. Agric. Food Chem., 2016, 64, 1730–1740 CrossRef CAS PubMed.
- W. Wang, P. Yang, L. Rao, L. Zhao, X. Wu, Y. Wang and X. Liao, Effect of high hydrostatic pressure processing on the structure, functionality, and nutritional properties of food proteins: A review, Compr. Rev. Food Sci. Food Saf., 2022, 21, 4640–4682 CrossRef CAS PubMed.
- X. Ji, Y. L. Xiong and J. Jiang, Tunable rice protein–starch composite soft gels: Structural role of ultrasound-modified protein, Food Hydrocolloids, 2024, 148, 109462 CrossRef CAS.
- R. Wang, T. Wang, W. Feng, Q. Wang and T. Wang, Rice proteins and cod proteins forming shared microstructures with enhanced functional and nutritional properties, Food Chem., 2021, 354, 129520 CrossRef CAS PubMed.
- Y. Xie, F. Yang, W. Shu, K. Zhao, Y. Huang, Q. Liu and Y. Yuan, Improved qualities of cod-rice dual-protein gel as affected by rice protein: Insight into molecular flexibility, protein interaction and gel properties, Food Res. Int., 2024, 197, 115176 CrossRef CAS PubMed.
- F. N. A. Rahim, W. Z. W. Ibadullah, N. Saari, N. A. Mustapha, F. H. Brishti, I.-F. M. Rashedi and R. Shukri, Effect of protein concentration, pH, and ionic strength on the adsorption properties of rice bran protein concentrates at the oil-water interface, Food Bioprod. Process., 2024, 147, 162–174 CrossRef.
- F. N. A. Rahim, W. Z. W. Ibadullah, N. Saari, F. H. Brishti, N. A. Mustapha, N. Ahmad and B. Arulrajah, The effect of alkaline extraction and drying techniques on the physicochemical, structural properties and functionality of rice bran protein concentrates, Int. J. Biol. Macromol., 2023, 242, 124908 CrossRef PubMed.
- W. Onsaard, S. Kate-Ngam and E. Onsaard, Physicochemical and antioxidant properties of rice bran protein hydrolysates obtained from different proteases, J. Food Meas. Charact., 2023, 17, 2374–2385 CrossRef.
- N. Wang, X. Cui, Y. Duan, S. Yang, P. Wang, A. S. M. Saleh and Z. Xiao, Potential health benefits and food applications of rice bran protein: research advances and challenges, Food Rev. Int., 2023, 39, 3578–3601 CrossRef CAS.
- H.-J. Chen, F.-J. Dai, C.-Y. Chen, S.-L. Fan, J.-H. Zheng, Y.-C. Huang, C.-F. Chau, Y.-S. Lin and C.-S. Chen, Evaluating the antioxidants, whitening and antiaging properties of rice protein hydrolysates, Molecules, 2021, 26, 3605 CrossRef CAS PubMed.
- Y. Liu, Z. Wang, H. Li, M. Liang and L. Yang, In vitro antioxidant activity of rice protein affected by alkaline degree and gastrointestinal protease digestion, J. Sci. Food Agric., 2016, 96, 4940–4950 CrossRef CAS PubMed.
- P.-H. Huang, T.-S. Wang and W.-C. Chang, Insights into the physicochemical and structural properties of pumpkin flour via synergistic treatments with debranching enzymes and freeze-thaw cycle, Food Hydrocolloids, 2024, 110421, DOI:10.1016/j.foodhyd.2024.110421.
- J. Luo, J. Wu, Y. Xu, H. Shao and P.-H. Huang, Isolation, purification, and structural elucidation of a water-soluble polysaccharide derived from Phellinus baumii Pilát mycelia, Heliyon, 2024, 10, e38239 CrossRef CAS PubMed.
- X. Xie, Y. Song, X. Bi, X. Liu, Y. Xing and Z. Che, Characterization of sea buckthorn polysaccharides and the analysis of its regulatory effect on the gut microbiota imbalance induced by cefixime in mice, J. Funct. Foods, 2023, 104, 105511 CrossRef CAS.
- P.-H. Huang, C.-S. Chiu, W.-C. Lu, R.-H. Huang, C.-C. R. Wang and P.-H. Li, Change in chemical composition and enhancement of intestinal microflora of acid hydrolyzed polysaccharides from Zizyphus jujube and Sterculia lychnophora, Arabian J. Chem., 2024, 17, 105598 CrossRef CAS.
- J. l. Zhai, L. Day, M.-I. Aguilar and T. J. Wooster, Protein folding at emulsion oil/water interfaces, Curr. Opin. Colloid Interface Sci., 2013, 18, 257–271 CrossRef CAS.
- L. Cui, J. Zhao, Q. Jia, J. Tang, F. Zhong, J. Lu and S. Zhou, A comparative study on stability and flavor of additive-free oat milk obtained by different heat pretreatments (blanching and microwave) of oat kernels, J. Cereal Sci., 2024, 120, 104057 CrossRef CAS.
- N. F. Rahmati, A. Koocheki, M. Varidi and R. Kadkhodaee, Adsorption of Speckled Sugar bean protein isolate at oil-water interface: Effect of ionic strength and pH, Int. J. Biol. Macromol., 2017, 95, 1179–1189 CrossRef CAS PubMed.
- C. Chi, W. Ren, Y. Yang, X. Guo, Y. Zhang, B. Chen, Y. He, H. Chen, X. Zheng and H. Wang, Starch ordered structures control starch reassembly behaviors during heat–moisture treatment for modulating its digestibility, Food Chem., 2024, 430, 136966 CrossRef CAS PubMed.
- N. Wang, Y. Dong, Y. Dai, H. Zhang, H. Hou, W. Wang, X. Ding, H. Zhang and C. Li, Influences of high hydrostatic pressure on structures and properties of mung bean starch and quality of cationic starch, Food Res. Int., 2023, 165, 112532 CrossRef CAS PubMed.
- P.-H. Huang, C.-S. Chiu, Y.-J. Chan, W.-C. Su, C.-C. R. Wang, W.-C. Lu and P.-H. Li, Effect of osmotic pressure and simultaneous heat-moisture phosphorylation treatments on the physicochemical properties of mung bean, water caltrop, and corn starches, Int. J. Biol. Macromol., 2024, 272, 132358 CrossRef CAS PubMed.
- Y. Zhang, C. Chen, Y. Chen and Y. Chen, Effect of rice protein on the water mobility, water migration and microstructure of rice starch during retrogradation, Food Hydrocolloids, 2019, 91, 136–142 CrossRef CAS.
- T. Roy, A. Singh, T. P. Sari and S. Homroy, Rice protein: Emerging insights of extraction, structural characteristics, functionality, and application in the food industry, J. Food Compos. Anal., 2023, 123, 105581 CrossRef CAS.
- M. R. I. Shishir and W. Chen, Trends of spray drying: A critical review on drying of fruit and vegetable juices, Trends Food Sci. Technol., 2017, 65, 49–67 CrossRef CAS.
- D. E. Walton and C. J. Mumford, Spray dried products—Characterization of particle morphology, Chem. Eng. Res. Des., 1999, 77, 21–38 CrossRef CAS.
- Y. Hu, H. Sun and T. Mu, Effects of different Zn2+ concentrations and high hydrostatic pressures (HHP) on Chlorophyll stability, Foods, 2022, 11, 2129 CrossRef CAS PubMed.
- B. Liu, Z. Wang, M. Liang and L. Yang, Rice protein reduces triglyceride levels through modulating cd36, mtp, fatp, and fabp expression in growing and adult rats, Foods, 2024, 13, 2704 CrossRef CAS PubMed.
- L. Yang, J. Chen, T. Xu, W. Qiu, Y. Zhang, L. Zhang, F. Xu and H. Liu, Rice protein extracted by different methods affects cholesterol metabolism in rats due to its lower digestibility, Int. J. Mol. Sci., 2011, 12, 7594–7608 CrossRef CAS PubMed.
- P.-H. Huang, Y.-T. Cheng, Y.-J. Chan, S.-J. Chen, J.-Y. Ciou, W.-C. Lu, W.-J. Hsu, C.-C. R. Wang and P.-H. Li, Anti-hyperlipidemic and antioxidant ability of HeShouWu (roots of Polygonum multiflorum Thunb.) and its complex formula, Arabian J. Chem., 2023, 16, 105280 CrossRef CAS.
- C. Wen, C. Liu, Y. Li, T. Xia, X. Zhang, S. Xue and O. J. Olatunji, Ameliorative potentials of the ethanolic extract from Lycium chinense leaf extract against diabetic cardiomyopathy. Insight into oxido-inflammatory and apoptosis modulation, Biomed. Pharmacother., 2022, 154, 113583 CrossRef CAS PubMed.
- X. Li, Y. Wu, J. Zhao, H. Wang, J. Tan, M. Yang, Y. Li, S. Deng, S. Gao, H. Li, Z. Yang, F. Yang, J. Ma, J. Cheng and W. Cai, Distinct cardiac energy metabolism and oxidative stress adaptations between obese and non-obese type 2 diabetes mellitus, Theranostics, 2020, 10, 2675–2695 CrossRef CAS PubMed.
- G. Jia, V. G. DeMarco and J. R. Sowers, Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy, Nat. Rev. Endocrinol., 2016, 12, 144–153 CrossRef CAS PubMed.
- H. Wang, Y. Fu, Q. Zhao, Z. Liu, C. Wang, Y. Xue and Q. Shen, Effects of heat-treated starch and protein from foxtail millet (Setaria italica) on type 2 diabetic mice, Food Chem., 2023, 404, 134735 CrossRef CAS PubMed.
- S. J. Gilani, M. N. Bin-Jumah, F. A. Al-Abbasi, M. S. Nadeem, S. S. Imam, S. Alshehri, M. M. Ghoneim, M. Afzal, S. I. Alzarea, N. Sayyed and I. Kazmi, Rosinidin flavonoid ameliorates hyperglycemia, lipid pathways and proinflammatory cytokines in streptozotocin-induced diabetic rats, Pharmaceutics, 2022, 14, 547 CrossRef CAS PubMed.
- R. Nakayama, D. Nishi, M. Sato, A. Ito, K. Uchiyama, Y. Higuchi, H. Takahashi and K. Ohinata, The effect of the rice endosperm protein hydrolysate on the subjective negative mood status in healthy humans: A randomized, double-blind, and placebo-controlled clinical trial, Nutrients, 2023, 15, 3491 CrossRef CAS PubMed.
- C. Kalpanadevi, S. P. Muthukumar, K. Govindaraju and R. Subramanian, Rice bran protein: An alternative plant-based protein to ameliorate protein malnourishment, J. Cereal Sci., 2021, 97, 103154 CrossRef CAS.
- M. Goicoechea, F. Cía, C. San José, A. Asensio, J. I. Emparanza, A. G. Gil, A. López de Cerain, P. Aldazabal, M. Azpitarte, D. Otaegui and A. López de Munain, Minimizing creatine kinase variability in rats for neuromuscular research purposes, Lab. Anim., 2008, 42, 19–25 CrossRef CAS PubMed.
- N. Z. H. Eleiwa, H. A. M. I. Khalifa and H. A. Nazim, Cardioprotective role of royal jelly in the prevention of celecoxib-mediated cardiotoxicity in adult male albino rats, J. Cardiothorac. Surg., 2024, 19, 135 CrossRef PubMed.
- A. Mahalakshmi and G. A. Kurian, Evaluating the impact of diabetes and diabetic cardiomyopathy rat heart on the outcome of ischemia-reperfusion associated oxidative stress, Free Radicals Biol. Med., 2018, 118, 35–43 CrossRef CAS PubMed.
- A. M. Alghamdi, F. A. Al-Abbasi, S. A. AlGhamdi, F. Fatima, S. I. Alzarea and I. Kazmi, Rosinidin inhibits NF-κB/Nrf2/caspase-3 expression and restores neurotransmitter levels in rotenone-activated Parkinson's disease, Saudi J. Biol. Sci., 2023, 30, 103656 CrossRef CAS PubMed.
- Z. Wang, Y. Liu, H. Li and L. Yang, Rice proteins, extracted by alkali and α-amylase, differently affect in vitro antioxidant activity, Food Chem., 2016, 206, 137–145 CrossRef CAS PubMed.
- S. Kashyap, B. H. R. Reddy, S. Devi and A. V. Kurpad, Potential impact of climate change on dietary grain protein content and its bioavailability-a mini review, Front. Nutr., 2024, 11, 1–9 Search PubMed.
- H. Li, M. Liang, Z. Wang, Y. Zhang, Q. Wu and L. Yang, Rice protein exerts endogenous antioxidant capacity via methionine sulfoxide reductase and the Nrf2 antioxidant system independent of age, J. Med. Food, 2020, 23, 565–574 CrossRef CAS PubMed.
- J. S. de Espindola, M. F. Taccóla, V. S. N. da Silva, L. D. D. Santos, B. C. Rossini, B. C. Mendonça, M. T. B. Pacheco and F. Galland, Digestion-resistant whey peptides promote antioxidant effect on Caco-2 cells, Food Res. Int., 2023, 173, 113291 CrossRef CAS PubMed.
- F. Galland, J. S. de Espindola, E. S. Sacilotto, L. G. V. C. Almeida, J. Morari, L. A. Velloso, L. D. dos Santos, B. C. Rossini and M. T. Bertoldo Pacheco, Digestion of whey peptide induces antioxidant and anti-inflammatory bioactivity on glial cells: Sequences identification and structural activity analysis, Food Res. Int., 2024, 188, 114433 CrossRef CAS PubMed.
- A. Sun, Y.-f. Li, Y. Miao, H.-x. Wang and L.-l. Zhang, Research on the mechanism of Ursolic acid for treating Parkinson's disease by network pharmacology and experimental verification, Heliyon, 2024, 10, e34113 CrossRef CAS PubMed.
- Z. Wang, M. Liang, H. Li, L. Cai and L. Yang, Rice protein exerts anti-inflammatory effect in growing and adult rats via suppressing NF-κB pathway, Int. J. Mol. Sci., 2019, 20, 6164 CrossRef CAS PubMed.
- L. Wen, Y. Chen, L. Zhang, H. Yu, Z. Xu, H. You and Y. Cheng, Rice protein hydrolysates (RPHs) inhibit the LPS-stimulated inflammatory response and phagocytosis in RAW264.7 macrophages by regulating the NF-κB signaling pathway, RSC Adv., 2016, 6, 71295–71304 RSC.
- Z. Yan, Y. Gui, C. Liu, X. Zhang, C. Wen, O. J. Olatunji, I. Suttikhana and T. J. Ashaolu, Gastrointestinal digestion of food proteins: Anticancer, antihypertensive, anti-obesity, and immunomodulatory mechanisms of the derived peptides, Food Res. Int., 2024, 189, 114573 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |