 Open Access Article
Open Access ArticleFinancial analysis of shipping container-based mushroom cultivation
Mahsa Aliana,
Sunil P. Dhoubhadelb,
Sandesh Risald,
Pratikshya Tiwarid,
Weihang Zhuc and
Venkatesh Balan*ac
aDepartment of Civil and Environmental Engineering, Cullen College of Engineering, University of Houston, Houston, TX 77002, USA. E-mail: vbalan@uh.edu
bDepartment of Agriculture, Nutrition and Human Ecology, College of Agriculture, Food, and Natural Resources, Prairie View A&M University, Prairie View, TX 77446, USA
cDepartment of Engineering Technology, Cullen College of Engineering, University of Houston, Sugarland, TX 77479, USA
dDepartment of Mechanical and Aerospace Engineering, University of Houston, Houston, TX 77204, USA
First published on 29th April 2026
Abstract
Shipping-container mushroom farms offer a sustainable and scalable approach to year-round food production, enabling efficient resource use with minimal land and water requirements. This study presents a comprehensive financial assessment of container-based mushroom cultivation across four production scales (50, 100, 200, and 500 lb per day) using four commercially relevant species: Oyster, Enoki, Maitake, and Lion's Mane. Economic feasibility was evaluated over a 10-year project horizon using net present value (NPV), discounted cash flow, and payback period. Results show that small-scale systems (50 and 100 lb per day) are not financially viable for lower-value species such as Oyster, whereas medium and large-scale operations (200 and 500 lb per day) achieve positive NPVs and substantially shorter payback periods. At 200 lb per day, payback periods range from 4.4–4.6 years for Enoki and Maitake and 3.4 years for Lion's Mane, while Oyster requires 9.8 years to recover the initial investment. Financial performance improves further at 500 lb per day, with payback periods of 2.1–2.5 years for Lion's Mane, Maitake, and Enoki, and 3.4 years for Oyster. All reported payback periods include an additional one year for container installation, infrastructure setup, system commissioning, and ramp-up to steady production. Overall, Lion's Mane and Maitake deliver the highest returns due to substantial market value and efficient cost recovery. These results demonstrate that production scale and species selection are the dominant drivers of profitability in container-based mushroom cultivation, and the analytical framework provides a practical decision-support tool that can be extended to medicinal or specialty fungi.
Sustainability spotlightContainer-based mushroom cultivation represents a forward-thinking solution to sustainable food production, combining efficient resource use with minimal environmental impact. By enabling year-round growth in controlled environments, this approach significantly reduces land and water consumption while supporting local economies and enhancing food security. The scalable nature of container farming enables optimized production that meets market demand without the typical challenges associated with traditional agriculture. This study's financial analysis underscores how such innovative systems not only promote ecological sustainability but also present viable business opportunities, paving the way for broader adoption of sustainable, high-value crop production methods. |
1 Introduction
The global mushroom farming industry has expanded significantly over the past few years due to rising demand for mushrooms as a sustainable protein alternative, driven by their versatility in fast food, rich flavor profiles, and nutritional benefits, creating profitable opportunities for farms of all sizes.1,2 Major contributors to the mushroom industry include the United States, China, the Netherlands, Poland, and India.3 In the U.S., Pennsylvania and California are the top two mushroom-producing states, which produce over 80% of the national mushroom production.3 During the 2021–2022 season, U.S. mushroom crop sales totaled 702 million pounds, valued at $1.02 billion.4 While 70–80% of domestic production consists of Agaricus bisporus (Button), demand is steadily increasing for other species such as Pleurotus ostreatus (Oyster), Grifola frondosa (Maitake), Flammulina velutipes (Enoki), Hericium erinaceus (Lion's mane), and Lentinula edodes (Shiitake).5 This trend is fueled by consumer interest in culinary diversity, plant-based diets, and functional foods with medicinal and immune-boosting properties.Compared to most plant crops, domestic mushroom cultivation requires less space, has shorter growth cycles, and can be maintained year-round.6 These advantages make mushroom production a high-margin opportunity for farmers and entrepreneurs. Mushroom cultivation is a multi-stage biological process that integrates mechanical operations with strict hygiene protocols to produce optimal yield and high-quality mushrooms. Densified biomass pellets undergo a series of steps, including storage, blending, jarred, sterilization, inoculation, incubation, fruiting, harvesting, packaging, and distribution. Brown and blue droplets denote the dirty and clean steps, respectively, to ensure proper sanitation flow (Fig. 1).
Large-scale mushroom farms benefit from advanced technologies, including automated climate-control systems, AI-based environmental monitoring, vertical farming structures, robotic harvesting, improved substrate processing, and blockchain-enabled supply-chain tracking. Small and medium-scale growers may lack access to high-tech innovations. Still, they can remain competitive by focusing on local markets, adopting eco-friendly practices to produce high-quality mushrooms, and selling directly to customers.7 Small-scale mushroom farms can benefit from the growing consumer interest in organic, locally sourced products, which can also help them establish a strong brand identity and stand out in the market.8,9 However, a significant challenge for small producers is maintaining the specific temperature and humidity ranges required by different mushroom species. Without a heating, ventilation, and air conditioning (HVAC) system, cultivation is constrained to seasons when ambient conditions are favorable. For example, L. edodes (shiitake) and P. ostreatus (Oyster) thrive in cool, moist environments (55–65 °F), limiting production to spring and fall in many areas. Calocybe indica (milky white) prefers warm, humid conditions and is best cultivated in summer.10 This seasonal dependency restricts year-round productivity and introduces variability in output, posing a challenge to the reliability and scalability of small-scale mushroom farming.11 Traditional small-scale mushroom farming typically relies on regular rooms, basements, or barns with basic insulation, manual ventilation, and basic humidity control. They are low-cost and accessible, but they offer limited precision in maintaining environmental conditions, increasing the risk of contamination and yield loss.12 In contrast, shipping containers are a promising option with superior insulation, a modular layout, and the easy integration of automated climate control systems, which enable consistent, year-round production despite the higher upfront cost. This setup is widely used for hydroponic microgreens.13 Shipping containers are widely available, space-efficient, and stackable, and can be customized to provide specific environmental conditions for growing various mushroom species year-round. Their mobility and scalability make them suitable for rural or remote locations with limited infrastructure. Additionally, equipping container roofs with solar panels can reduce energy expenses, enhance sustainability, and make this approach more viable for small-scale farmers and agri-entrepreneurs.
To operate a profitable small-scale mushroom farm, producers must carefully evaluate both technological and economic variables, ranging from selecting the appropriate mushroom species and preparing the substrate to controlling the environment, implementing automation, managing capital expenditures (CapEx), operating costs (OpEx), understanding market demand, and devising a pricing strategy.14 These factors are crucial for designing cost-effective and efficient operations that support financial sustainability. Financial feasibility is recognized as an essential tool for optimizing feasibility and profitability. However, outcomes may vary by region, mushroom type, and production scale.15 Numerous studies emphasize the importance of financial analysis and TEA in ensuring sustainability and profitability. However, outcomes vary by region, species, and farm size. For example, larger farms often achieve better margins due to economies of scale and cost efficiency.16–18 Keneni found that sourcing raw materials significantly reduced transport costs and boosted profitability.19
Despite growing interest in controlled-environment agriculture, a comprehensive financial analysis of shipping-container-based mushroom farming has been absent in the U.S. context. This study fills that gap by evaluating shipping container production systems for four scales: 50, 100, 200, and 500 pounds per day, with potential applications in other global settings.
CapEx, OpEx, NPV, and payback period are evaluated. In addition to financial modeling, this study offers practical insights through visual layouts, container configurations, and land-use estimates, supporting system design and planning. Results show the importance of production scale, a suitable mushroom strain, and a financing strategy for profitability and provide a decision-making framework for small-scale growers, investors, and policymakers. This approach is adaptable for cultivating a wide range of high-value edible and medicinal mushrooms, further enhancing its utility and potential impact on sustainable food systems.
2 Methods and assumptions
2.1. Substrates and conditioning assumptions for mushroom production
In commercial mushroom production, several substrate formulations vary by species and are a critical determinant of yield and quality. Oyster, Maitake, Lion's mane, and Enoki mushrooms can all be cultivated on a range of lignocellulosic or nutrient-supplemented substrates. Typical formulations include hardwood sawdust amended with bran, Master's mix (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 hardwood sawdust and soy hulls), and pasteurized or pelleted straw. Oyster mushrooms are exceptionally adaptable and can utilize a wide range of agricultural residues, including corncobs, wheat straw, rice straw, cottonseed hulls, sugarcane bagasse, and soybean hulls, making them among the most flexible in terms of substrate selection.20 Maitake mushrooms, while more selective, perform well on hardwood sawdust supplemented with bran or gypsum, on oak-based sawdust blends, and in soy hull-enriched formulations that support dense, uniform mycelial colonization.21
50 hardwood sawdust and soy hulls), and pasteurized or pelleted straw. Oyster mushrooms are exceptionally adaptable and can utilize a wide range of agricultural residues, including corncobs, wheat straw, rice straw, cottonseed hulls, sugarcane bagasse, and soybean hulls, making them among the most flexible in terms of substrate selection.20 Maitake mushrooms, while more selective, perform well on hardwood sawdust supplemented with bran or gypsum, on oak-based sawdust blends, and in soy hull-enriched formulations that support dense, uniform mycelial colonization.21
Lion's mane cultivation typically relies on hardwood sawdust supplemented with bran, with oak-based sawdust fortified with wheat bran and calcium sources being especially effective. The species can also grow on beech or maple sawdust, soy-hull-rich mixes, and, in some cases, pelleted straw blends, although yields are generally lower on non-wood substrates.22 Enoki mushrooms are traditionally produced on hardwood sawdust supplemented with wheat bran and calcium carbonate. Still, they are also compatible with oak- or beech-based sawdust, Master's mix, and finely processed straw or corncob substrates when properly sterilized.23
These substrate formulations are widely adopted in commercial mushroom production due to their reliability, balanced nutrient profiles, broad availability, and consistently high biological efficiency. In this study, all substrates are assumed to be purchased in pre-processed, pelletized form to standardize input quality and facilitate transparent cost estimation.24 The pellets are hydrated in a paddle mixer to achieve a target moisture content of approximately 65–70%, which is optimal for fungal growth.25 Following hydration, the substrate is filled into bags or bottles and autoclaved to eliminate contaminants and establish a sterile environment for mycelial colonization.26
After sterilization and cooling, grain spawn is added at an inoculation rate of 5–20% (w/w), depending on the mushroom species, growth characteristics, substrate composition, and desired colonization speed. Higher spawn rates are commonly applied to species with slower mycelial expansion or when using denser substrates. In this analysis, a 10% spawn rate is assumed for Oyster and Enoki, reflecting their rapid and efficient colonization under standard production conditions. In contrast, Lion's mane and Maitake are modeled using a 15% spawn rate, acknowledging that these species typically benefit from higher inoculation levels to ensure uniform and reliable colonization, particularly in substrates that are more resistant to mycelial penetration. The equipment used for substrate conditioning and container filling is illustrated in Fig. S1.
After inoculation, the sealed substrate bags or bottles enter the incubation phase, during which fungal mycelium colonizes the substrate. Incubation is typically carried out in a dark or low-light condition under controlled temperature, humidity, and fresh-air exchange to promote rapid mycelial expansion. While optimal parameters vary by species, incubation generally requires elevated CO2 concentrations, high relative humidity (60–75%), and minimal ventilation. Upon complete substrate colonization, the system transitions to the fruiting phase, characterized by reduced temperatures, higher humidity, and increased air exchange to promote primordia formation and fruit-body development. Species-specific environmental requirements for both phases are summarized in Table 1.
| Mushroom species | Incubation conditions | Fruiting conditions | Reference |
|---|---|---|---|
| Oyster (Pleurotus ostreatus) | Temperature: 25–30 °C | Temperature: 20–25 °C | 27 |
| Humidity: 65–75% | Humidity: 85–95% | ||
| Light: dark/low light | Light: moderate diffuse light | ||
| Air exchange: minimal | CO2: <1000 ppm | ||
| Air exchange: high fresh-air flow | |||
| Maitake (Grifola frondosa) | Temperature: 15–20 °C | Temperature: 12–18 °C | 28 |
| Humidity: 60–70% | Humidity: 85–95% | ||
| Light: dark | Light: low-moderate light | ||
| CO2: <800 ppm | |||
| Air exchange: moderate | |||
| Lion's Mane (Hericium erinaceus) | Temperature: 20–25 °C | Temperature: 18–24 °C | 29 |
| Humidity: 65–70% | Humidity: 90–95% | ||
| Light: dark | Light: low diffuse light | ||
| CO2: <1000 ppm | |||
| Air exchange: gentle ventilation to avoid elongated spines | |||
| Enoki (Flammulina velutipes) | Temperature: 18–25 °C | Temperature: 15–18 °C | 30 |
| Humidity: 60–70% | Humidity: 85–95% | ||
| Light: dark | Light: very low light | ||
| CO2: <800 ppm | |||
| Air exchange: controlled, low airflow |
In this study, both incubation and fruiting are assumed to occur within a climate-controlled shipping container, where species-specific environmental conditions are maintained using integrated HVAC, humidification, and ventilation systems. Mature mushrooms are harvested manually, packaged, and prepared for distribution to buyers and end users. Details regarding substrate volume capacity within the shipping container and corresponding mushroom yields are provided in Table S1.
2.2. Mushroom variety, growth conditions, and market value
The mushroom varieties evaluated in this study are Oyster, Enoki, Maitake, and Lion's mane, and were selected based on their commercial relevance, cultivation feasibility, and market demand. Each species exhibits specific biological and environmental requirements that influence its suitability for small and medium-scale farming. Mushroom yield is demonstrated with biological efficiency percentage (BE), which is a measure of how effectively a mushroom crop converts substrate into mushrooms. It is calculated as the fresh weight of the harvested mushroom divided by the substrate's dry weight. For Oyster mushrooms, BE ranges from 80–150% on straw-based substrates, while Enoki mushrooms achieve 70–100% BE on sawdust. Maitake mushrooms can be grown on wood chips, have a lower BE of 35–45%, but command high market prices. Lion's mane is priced for their flavor, reaching a BE of 40–60%.19,31–33 Optimal temperature and humidity requirements differ by species and can substantially impact both yield and production costs (Table 1).Oyster, Enoki, Lion's mane, and Maitake mushrooms are tolerant of standard indoor conditions. Market prices reflect consumer demand and production cost, with Lion's mane priced at ∼$20–30 per lb, Maitake at $18–22 per lb, Enoki at $8–12 per lb, and Oyster mushrooms at ∼$6–8 per lb (Table 2).
| Type of mushroom | BE (%) | Assumed substrate | Average price ($ per lb) | Reference | |
|---|---|---|---|---|---|
| Oyster |  |
80–150 | Corncobs, wheat straw, rice straw, cottonseed hulls, sugarcane bagasse, and soybean hulls | 6–8 | 20 |
| Enoki |  |
70–100 | Hardwood sawdust, oak-based sawdust blends | 8–12 | 23 |
| Lion's Mane |  |
60–150 | Hardwood sawdust, wheat bran | 20–30 | 22 |
| Maitake |  |
35–45 | Hardwood sawdust, wheat bran | 18–22 | 21 |
By analyzing mushrooms with diverse growth parameters and economic values, this study aims to identify the most profitable and sustainable container-based cultivation method.
2.3. Organization of a shipping container mushroom farm
The shipping container mushroom farm will use standard 40 ft × 8 ft × 8.6 ft containers (320 ft2 each), with layouts precisely modeled in AutoCAD to optimize spatial organization. Each container is equipped with HVAC systems, circulation fans, humidifiers, and full-spectrum LED lighting, enabling precise control of temperature, humidity, airflow, and lighting conditions. The system is designed to accommodate four production scales, with the corresponding number of containers indicated in parentheses: 50 lb per day (7 containers), 100 lb per day (11 containers), 200 lb per day (17.5 containers), and 500 lb per day (33 containers). A minimum clearance of 15 feet is maintained around the installation for vehicle access and maneuverability. Each container is subdivided into growing chambers. These chambers are designed to operate in staggered cycles, allowing simultaneous cultivation of multiple species or growth stages. Additional containers are designed for specific functions such as substrate preparation, sterilization, inoculation, and harvesting, as well as walkways to provide smooth operational flow.The illustrated facility layout offers a scalable and efficient framework for container-based mushroom cultivation, designed to accommodate four production capacities: 50 and 100 pounds per day (Fig. 2A and B) and 200 and 500 lb per day (Fig. 3A and B).
The cultivation facility is organized into distinct functional zones to streamline workflow and optimize productivity. Each production scale corresponds to a defined number of shipping containers and land footprint, as detailed in Table S2: 50 lb per day (7 containers, 6000 ft2), 100 lb per day, (11 containers, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2), 200 lb per day (17.5 containers, 15
000 ft2), 200 lb per day (17.5 containers, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2) and 500 lb per day (33 container, 25
000 ft2) and 500 lb per day (33 container, 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2). All containers are aligned along a central walkway, with side doors to provide smooth access for staff, equipment, and harvest trays. This ergonomic layout facilitates efficient loading and unloading, routine cleaning, and continuous environmental monitoring.
000 ft2). All containers are aligned along a central walkway, with side doors to provide smooth access for staff, equipment, and harvest trays. This ergonomic layout facilitates efficient loading and unloading, routine cleaning, and continuous environmental monitoring.
Each container is outfitted with standardized infrastructure to maintain optimal growing conditions including: (i) ventilation system with intake, exhaust, and auxiliary air handling fans; (ii) 1-ton mini-split HVAC unit with dual-zone temperature control; (iii) ultrasonic or fogging humidifiers for precise humidity regulation; (iv) full-spectrum LED grow lights for consistent illumination; (v) water pump (0.3–0.5 HP) for automated misting or irrigation; and (vi) Wi-Fi-enabled smart controllers integrated with temperature, humidity, and CO2 sensors. Shared zones, such as the central walkway and substrate preparation area, are also climate-controlled to ensure consistent air quality and environmental stability across the entire facility.
An efficient contamination-control system is critical for reliable container-based mushroom production. To minimize the introduction of airborne spores, microorganisms, and dust, the container design incorporates a HEPA-filtered air intake and maintains a controlled positive-pressure environment. HEPA filtration removes fine particulate contaminants before air enters the cultivation chamber. At the same time, positive pressure ensures that airflow is directly outward through any small openings, thereby preventing infiltration of unfiltered ambient air. Together, these measures substantially reduce contamination risks during both substrate colonization and fruiting, supporting consistent yields and hygienic production conditions.34
2.4. Capital expenditure computation assumptions
CapEx refers to the long-term investments required to establish infrastructure and equipment before production and revenue generation.15,16,35,36 Major CapEx components include specialized growing systems, indoor production machinery, land acquisition or leasing, and the conversion of shipping containers into climate-controlled fruiting chambers. Essential equipment, such as autoclaves, industrial balances, filling machines, ribbon mixers, and pallet lifters, is scaled according to the production capacity, with the types and numbers of containers tailored to support daily outputs of 50, 100, 200, or 500 pounds. The specific equipment requirements and assumed cost at each scale are detailed in Tables S2 and S3, respectively. Estimated land requirements, including space for containers and vehicle access, are 6000 ft2, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2, 15
000 ft2, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2, and 25
000 ft2, and 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ft2 for respective production capacities. To account for unforeseen needs, a 20% markup is applied to the initial investment for additional supplies, and a 15% markup is applied to ongoing maintenance costs. Installation costs, including container retrofitting and system integration, are estimated at 30% of the initial investment. All cost estimations are based on 2025 market data, using average prices for equipment, containers, materials, and variable inputs.
000 ft2 for respective production capacities. To account for unforeseen needs, a 20% markup is applied to the initial investment for additional supplies, and a 15% markup is applied to ongoing maintenance costs. Installation costs, including container retrofitting and system integration, are estimated at 30% of the initial investment. All cost estimations are based on 2025 market data, using average prices for equipment, containers, materials, and variable inputs.
2.5. Operating expenditure computation assumptions
OpEx for daily mushroom cultivation constitutes a significant portion of total production costs and scales with output volume.7,37 These expenses include labor, substrates and spawn (assumed purchased ready-made), utilities, packaging, and distribution. In this study, OpEx is categorized into five main components: substrate and spawn, electricity, packaging, water, and labor.| BE (%) = (weight of fresh mushrooms)/(dry weight of substrate) | (1) |
Substrate costs, including shipping fees, are calculated. Total wet substrate and its associated water content are then determined using eqn (2):
| Wet weight of substrate = (dry weight of substrate)/(1 − moisture content %) | (2) |
To account for spawn, 10–15% of the total wet substrate is allocated for spawn preparation, and the associated cost is calculated accordingly.
| Water required = wet weight − wet mass of ingredients | (3) |
In this paper, the average of 2–4 gallons of water per pound of mushrooms is assumed for all calculations.39
Total electrical energy consumption (ETotal) is calculated as the sum of all non-HVAC electrical loads and the electricity consumed by the cooling system using eqn (4):
 | (4) |
This formulation distinguishes electrical power that is converted to heat (from lighting, fans, pumps, and environmental monitoring systems) from electrical power required to move that heat via cooling. The HVAC electricity demand is governed by the system's coefficient of performance (COP), which represents cooling efficiency (e.g., a COP of 3 indicates that 1 kWh of electricity removes 3 kWh of heat). This two-term approach allows accurate assessment of how biological heat generation, ventilation rates, and insulation levels influence total facility energy demand.
2.5.3.1. Fixed electrical loads. All devices operating within the container, excluding the active refrigeration cycle, were treated as fixed electrical loads, and their daily energy consumption was calculated as the sum of each component's rated power and operating time (eqn (5)).
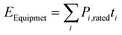 | (5) |
These loads include LED grow lights, whose energy use varies with shelf configuration, species-specific lighting requirements, and photoperiod, and which convert nearly all the electricity they consume into heat within the chamber. Additional contributors include humidifiers, circulation fans, and HEPA or laminar-flow units that operate for extended periods to maintain target humidity and air quality, as well as environmental sensors, automated valves, and control systems that draw continuous low-power loads. Pumps used for irrigation and misting add intermittent heat gains, while substrate-processing equipment (e.g., ribbon mixers) contributes additional thermal load on preparation days. Autoclaves and steamers represent the largest single electrical load (3–18 kW during sterilization); when operated inside the container, they introduce substantial internal heat that can exceed biological and environmental heat sources. Consequently, autoclave operation is preferably isolated in a separate, externally vented module to avoid oversizing of the HVAC system and excessive cooling demand.
2.5.3.2. HVAC thermal load. The total thermal load that the HVAC system must remove was modeled as the sum of four distinct heat sources, each representing a different physical mechanism contributing to temperature rise within the container (eqn (6)):
| Qthermal = Qtrans + Qvent + Qinternal + Qproduct | (6) |
2.5.3.2.1. Transmission heat load. Transmission heat is conductive heat transfer through the container walls, ceiling, and floor, driven by temperature differences between the external environment and the controlled interior. It is calculated as (eqn (7)):
| Qtrans = U × A × (Tamb − Tset) | (7) |
2.5.3.3. Internal electrical heat gain. Nearly all electrical devices operating within the container dissipate most of the energy they consume as heat, resulting in a substantial internal heat load. The internal electrical heat generation was estimated as (eqn (8)):
| Qinternal = ηheat × Eequipment | (8) |
2.5.3.4. Biological product heat. Actively growing mushrooms and colonized substrate continuously release metabolic heat through cellular respiration, representing a significant component of the total thermal load within the growing chamber. This heat generation was quantified as (eqn (9)):
| Qproduct = Wrasp × Mbio | (9) |
2.5.3.5. Ventilation heat load. Ventilation is essential in mushroom cultivation because fungal respiration generates substantial CO2, requiring a continuous supply of fresh air to maintain proper gas exchange and avoid growth inhibition. However, this necessary airflow imposes a thermal penalty, as outside air, typically warmer than the growing chamber, is introduced into the container and must be cooled to the species-specific setpoint temperature. The resulting ventilation heat load is computed as (eqn (10):
| Qvent = V × ρ × Cp × ΔT | (10) |
To convert the total thermal load into electrical energy consumption, the sum of the heat sources was divided by the HVAC system's coefficient of performance (COP) to determine the required cooling power. A COP of 3 was assumed, indicating that the HVAC system removes 3 kW of heat per 1 kW of electricity consumed. Daily cooling energy use (kWh) was then calculated by multiplying the cooling power by the system's daily operating hours, thereby directly converting the thermal load to refrigeration energy demand.43
 | ||
| Fig. 4 Average monthly high and low temperatures for Houston, Texas, expressed in degrees Celsius. Values were converted from historical climate data.44 | ||
For each month, the ambient temperature was represented by the average of the monthly high and low temperatures. Species-specific temperature differentials were then calculated using these monthly ambient temperatures and the required cultivation setpoints. These temperature differences were used to update monthly ventilation and transmission heat loads. To isolate the effect of climate alone, all other system parameters, including container insulation, surface area, airflow rate, HVAC coefficient of performance, and operating hours, were held constant, and the analysis was performed for a single growing container.
| OpEx = Σ(CSub + CL + CP + CW + CE) × (working days) | (11) |
2.6. Financial analysis and discounted returns
In this model, revenue is generated from the sale of fresh mushrooms and spent mushroom substrate (SMS). Fresh mushrooms are assumed to be sold at the average market price, while SMS is sold at $0.01 per pound (wet weight). In the first year, the number of harvesting days is reduced due to the initial cultivation period. The financial analysis of the mushroom farm uses a 10-year discounted cash flow analysis that incorporates both CapEx and OpEx.45 The farm design includes four production scales: 50, 100, 200, and 500 lb per day, each requiring equipment appropriate to its scale, with associated costs. Equipment depreciation, representing the reduction in asset value due to wear and tear over time, is calculated using the straight-line method over a 10-year lifespan with no salvage value and is included in annual OpEx (eqn (12)).| Depreciation = (purchase price − salvage value)/useful life | (12) |
The financing structure assumes an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 debt-to-equity ratio. Interest rates of 8% on debt and 10% on equity were considered based on market conditions and the relative risk of debt versus equity financing. A 30% corporate tax rate was applied, consistent with standard government tax regulations. Taxable income is determined by subtracting allowable deductions, such as annual depreciation and loan interest, from gross income.46 The income tax is calculated using eqn (13):
20 debt-to-equity ratio. Interest rates of 8% on debt and 10% on equity were considered based on market conditions and the relative risk of debt versus equity financing. A 30% corporate tax rate was applied, consistent with standard government tax regulations. Taxable income is determined by subtracting allowable deductions, such as annual depreciation and loan interest, from gross income.46 The income tax is calculated using eqn (13):
| Income tax = (gross income − depreciation − loan interest) × 0.30 | (13) |
To evaluate investment feasibility, future cash flows are discounted to present value using the Weighted Average Cost of Capital (WACC), which reflects the minimum return investors expect. WACC is a composite rate based on the cost of equity and the cost of debt, adjusted for their respective capital-structure weights and tax savings on interest (eqn (14)).47
| WACC = (equity wt. × cost of equity) + (debt weight × cost of debt) × (1 − tax rate) | (14) |
The key indicator used includes NPV, which measures profitability by subtracting the initial investment from the present value of projected returns. A positive NPV indicates financial viability.48 Additionally, the payback period was calculated to estimate the time required to recover the initial investment, providing insight into investment risk and capital recovery.46,49
3 Results and discussion
3.1. Processing substrate and producing mushrooms in a shipping container
Mushroom cultivation in shipping containers follows a modular, controlled process designed to optimize space, hygiene, and yield. The process begins in the “dirty zone”, where densified lignocellulosic biomass pellets are loaded into a hopper, conveyed to a ribbon blender, and mixed with supplements and water to moisten the substrate. The blended substrate is dispensed into bottles or bags using automated loaders, then sterilized in a double-door steam sterilizer. After sterilization, the bottles or bags are moved into the “clean zone”, where they are inoculated with mushroom spawn (solid/liquid) under aseptic conditions (Fig. 5).These inoculated substrates are then incubated at regulated temperatures, with low humidity (70%) and high CO2 levels, in dark conditions, to promote mycelial colonization. Once colonization is complete, environmental conditions are carefully adjusted to control lighting, airflow, and CO2 levels, promoting fruit body formation. Bottles (used for Enoki and Lion's mane mushrooms) are placed on racks, while polythene bags (used for Oyster and Maitake mushrooms) are suspended from rods. Mature mushrooms are then harvested, packed, and prepared for distribution within the container's compact, climate-controlled environment, enabling scalable, year-round production.
Fig. 6 illustrates the internal configuration of a standard 40 ft. long high-cube shipping container, divided into two independent growing chambers. Each chamber can accommodate either substrate bags or jars and is accessible via insulated doors. Fig. 6A depicts a jar-based system in which mycelium-inoculated jars are placed in trays and stacked on three-tier shelving. Fig. 6B illustrates a bag-based system in which inoculated substrate bags are vertically hung from rods, making it ideal for high-fruiting species.
The modular design supports flexible batch management and species-specific environmental control, minimizing cross-contamination and enabling continuous, high-efficiency mushroom cultivation. The container interior features a washable surface, allowing each chamber to be washed and sanitized between cultivation cycles to reduce the risk of contamination.
3.2. CapEx analysis
CapEx for establishing container-based mushroom farms was estimated across four production scales—50, 100, 200, and 500 pounds per day. CapEx includes costs for equipment, container procurement, land acquisition, and installation/construction, all of which scale with production capacity (Table 3).| Items | Mushroom farm's production capacity | |||
|---|---|---|---|---|
| 50 lb per day ($) | 100 lb per day ($) | 200 lb per day ($) | 500 lb per day ($) | |
| Autoclave | 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
180![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
180![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
| Industrial balance | 1700 | 1700 | 3400 | 5100 |
| Filling machine | 3000 | 3000 | 6000 | 6000 |
| Ribbon Mixer | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
| Industrial stacker | 7500 | 7500 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
| Other cost | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 440 440 |
31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 940 940 |
48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 380 380 |
48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 720 720 |
| Refrigerated container | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
125![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
262![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
| Regular container | 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
60,000 | 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
| Land | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
| Installation and construction | 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 842 842 |
99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 042 042 |
158![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 334 334 |
218![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 496 496 |
| Total | 241![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 982 982 |
429![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 182 182 |
686![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 114 114 |
946![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 816 816 |
Equipment costs encompass autoclaves, industrial balances, filling machines, ribbon mixers, and stackers, scaled to match operational throughput. Equipment costs begin at $106![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 140 for the 50 lb per day system and increase to $199
140 for the 50 lb per day system and increase to $199![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 140 for 100 lb per day, $305
140 for 100 lb per day, $305![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 280 for 200 lb per day, and $307
280 for 200 lb per day, and $307![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320 for 500 lb per day.
320 for 500 lb per day.
Container costs rise with production capacity and are divided into two categories: insulated, climate-controlled growing containers, and standard containers for substrate preparation, incubation, and logistics. For 50 lb per day, two growing containers and five regular containers are required. This increases to 4 and 7 for 100 lb per day, 10 and 7.5 for 200 lb per day, and 21 and 12 for 500 lb per day, respectively. Total container costs range from $65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (50 lb per day) to $358
000 (50 lb per day) to $358![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 (500 lb per day).
500 (500 lb per day).
Assuming an average land price of $2.50 per ft2, land acquisition costs range from $15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (50 lb per day) to $62
000 (50 lb per day) to $62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 (500 lb per day), with intermediate scale requiring $25
500 (500 lb per day), with intermediate scale requiring $25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (100 lb per day) and $37
000 (100 lb per day) and $37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 (200 lb per day). Installation and construction expenses, including retrofitting the container, HVAC setup, plumbing, and electrical work, range from $55
500 (200 lb per day). Installation and construction expenses, including retrofitting the container, HVAC setup, plumbing, and electrical work, range from $55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 842 to $218
842 to $218![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 496. The total estimated CapEx by scale is: 50 lb per day ($241
496. The total estimated CapEx by scale is: 50 lb per day ($241![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 982); 100 lb per day ($429
982); 100 lb per day ($429![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 182); 200 lb per day ($686
182); 200 lb per day ($686![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 114); 500 lb per day ($946
114); 500 lb per day ($946![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 816). These estimates highlight the direct relationship between production scale and capital investment, consistent with the existing literature emphasizing the need for proportionally scaled infrastructure to ensure operational efficiency and financial viability.45
816). These estimates highlight the direct relationship between production scale and capital investment, consistent with the existing literature emphasizing the need for proportionally scaled infrastructure to ensure operational efficiency and financial viability.45
3.3. OpEX analysis
The total OpEx for container-based mushroom cultivation was estimated across four production scales 50, 100, 200, and 500 lb per day to account for variability in substrate and spawn inputs, electricity usage, packaging, water, and labor costs (Fig. 7). A breakdown in electricity consumption and associated fees is provided in SI Table S4. These OpEx estimates align with earlier studies highlighting the importance of scalability assessments in mushroom production, particularly in resource-sensitive regions such as India, where output expansion significantly increases input demand.36The operating expenditure (OpEx) analysis across production scales of 50, 100, 200, and 500 lb per day reveals clear scale-dependent trends and species-specific differences in resource requirements. At the most minor scale (50 lb per day), daily OpEx remains relatively modest, totaling $253 for Oyster, $307 for Lion's mane, $301 for Maitake, and $257 for Enoki. These lower costs reflect reduced substrate throughput, limited environmental loads, and minimal labor and material-handling demands. As production increases to 100 lb per day, OpEx rises proportionally with greater substrate volumes, higher spawn consumption, and increased energy demand, reaching $477 for Oyster, $583 for Lion's mane, $568 for Maitake, and $480 for Enoki. At 200 lb per day, daily operating costs increase further to $691 for Oyster, $902 for Lion's mane, $870 for Maitake, and $695 for Enoki, driven by more frequent substrate preparation cycles, expanded labor requirements, and intensified HVAC operation.
At the largest production scale (500 lb per day), OpEx peaks at $1062 for Oyster, $1590 for Lion's mane, $1506 for Maitake, and $1070 for Enoki, reflecting substantial increases in energy consumption, packaging needs, and overall process throughput associated with high-volume production. Across all scales, Lion's mane and Maitake consistently exhibit higher operating costs due to stricter environmental control requirements and more labor-intensive handling. In contrast, Oyster and Enoki remain comparatively less costly to produce.
Examination of individual operating cost components reveals clear scale-dependent trends and, in some cases, species-independent behaviors. Water costs increase proportionally with production volume, as higher substrate throughput, greater humidification demand, and more frequent sanitation activities require increased water use. Daily water expenses rise from $2.8 at 50 lb per day to $27.8 at 500 lb per day, reflecting the combined contributions of misting, substrate hydration, soaking, and routine cleaning.
Labor costs also scale upward with increasing production capacity, as additional time is required for substrate preparation, inoculation, environmental monitoring, harvesting support, and general chamber maintenance. Total labor expenses increase from $180.0 per day at 50 lb per day to $600.0 per day at 500 lb per day. However, labor cost per unit of production declines with scale, demonstrating clear economies of scale. Harvesting and packaging costs increase nearly linearly with output because each unit of production requires manual harvesting, trimming, and packaging. These costs rise from $18.5 per day at 50 lb per day to $185.0 per day at 500 lb per day, reflecting the direct relationship between production volume and packaging material consumption and handling effort.
Substrate costs vary across species due to differences in substrate composition, bulk density, and moisture requirements. At the smallest production scale (50 lb per day), daily substrate expenses range from $5.2 for Oyster to $11.3 for Maitake, with Enoki remaining comparatively low at $5.3. As production increases to 500 lb per day, substrate costs scale accordingly, reaching $51.8 for Oyster, $90.0 for Lion's mane, $112.5 for Maitake, and $52.9 for Enoki. Spawn costs exhibit the most significant species-specific variation because they are directly influenced by biological efficiency. Species with higher biological efficiency require less substrate to achieve the same output; because spawn is applied at a fixed inoculation rate (10% w/w), lower substrate requirements translate directly into reduced spawn demand. Consequently, Oyster and Enoki exhibit relatively low spawn costs across all scales ($16.8–$171.6), whereas Lion's mane and Maitake, characterized by lower biological efficiency require greater substrate input and therefore incur substantially higher spawn costs ($65.6–$656.3).
Electricity costs vary across species because of differences in temperature setpoints, humidity control, and ventilation requirements. At the smallest production scale (50 lb per day), daily electricity expenses range from $30.2 for Oyster to $33.6 for Maitake. As production increases to 500 lb per day, electricity costs rise substantially to $150.3–$166.0 per day, reflecting the greater HVAC demand associated with larger cultivation volumes and intensified environmental control. Overall, Lion's mane and Maitake incur higher substrate, spawn, and electricity costs due to their lower biological efficiency and more stringent climate requirements. In contrast, Oyster and Enoki consistently remain the most cost-effective species across all production scales.39,50 The estimated energy consumption of 4 kWh per lb at the 500 lb per day scale is reasonable and consistent with literature values, as container-based mushroom cultivation requires additional HVAC, humidification, and environmental control energy compared with conventional systems. Robinson et al. reported total primary energy use of approximately 2.8 kWh per lb, with electricity as the dominant contributor, supporting the plausibility of higher energy intensity in more tightly controlled production systems.39
3.4. Sensitivity analysis of ventilation and transmission loads
To assess the impact of seasonal climate variability on container energy performance, a month-by-month temperature sensitivity analysis was conducted for both ventilation energy (Event), and transmission energy (Etrans) across four cultivated mushroom species. For each month, the effective ambient temperature was defined as the average of the monthly high and low temperatures, representing typical outdoor operating conditions for the container. These monthly ambient temperatures range from approximately 12 °C in the winter months (January and December) to 28–29 °C in the summer months (July and August), producing pronounced seasonal variability in thermal loading (Fig S2).Using these ambient conditions, a monthly temperature difference ΔT was calculated for each species based on its species-specific growing temperature (setpoint). The four mushrooms evaluated (Oyster, Maitake, Lion's mane, and Enoki), exhibit distinct thermal preferences, with Oyster requiring the warmest conditions and Enoki the coldest. Consequently, ΔT varies substantially across species and seasons. Oyster shows small or near-zero ΔT values during warmer months (May–September) and, in some cases, slightly positive values, reflecting minimal cooling requirements. In contrast, Maitake and Enoki exhibit consistently large positive ΔT values during summer because their cultivation temperatures must be maintained well below outdoor conditions. During winter months, Oyster and Lion's mane experience negative ΔT values, indicating net heat loss to the environment and a corresponding need for space heating rather than cooling.
These monthly ΔT values directly govern the ventilation energy (Event) and transmission energy (Etrans) requirements associated with air exchange and conductive heat transfer through the container envelope. When ΔT < 0, heat is lost from the container to the surroundings, resulting in negative values of Event and Etrans and indicating a heating demand. Conversely, when ΔT > 0, the HVAC system must remove excess heat, resulting in positive energy values associated with cooling demand. Oyster mushrooms, which grow at temperatures close to ambient conditions, require little to no cooling during spring and fall but exhibit substantial heating demand in winter (e.g., Event = −8.3 in January, −7.3 in February, and −7.9 in December). Lion's mane follows a similar trend, with pronounced winter heating requirements (Event = −7.1 in January and −6.8 in December) and only moderate cooling demand during summer, peaking at approximately 5.8 in August.
In contrast, Maitake and Enoki, which require significantly cooler growing conditions, experience sustained and elevated cooling loads during warm months. Maitake's cooling demand increases sharply from late spring into summer, reaching approximately 6.8 in May, 9.4 in June, 10.2 in July, and peaking at 10.5 in August. Enoki displays a comparable seasonal pattern, with cooling demand rising from 5.7 in May to 8.2 in June, 9.1 in July, and 9.3 in August. Elevated cooling requirements persist into early autumn for both species (e.g., 7.4 for Enoki and 8.5 for Maitake in September). Collectively, these results highlight that warm-climate conditions impose minimal cooling burdens for warm-growing species such as Oyster and Lion's mane, while generating substantial HVAC energy demand for colder-growing species, particularly Maitake and Enoki.
Transmission energy (Etrans) exhibits a seasonal pattern similar to ventilation energy but remains smaller in magnitude due to the container's high insulation (U = 0.07 W m−2 K−1). As outdoor temperatures exceed species-specific setpoints, E(trans) becomes increasingly positive, reflecting additional cooling demand. Warm-growing species such as Oyster experience modest summer transmission loads, rising from 0.1–0.5 kWh between May and August, peaking at 0.5 kWh in August. Colder-growing species, including Maitake and Enoki, show larger midsummer values due to greater temperature differentials: Maitake increases from 0.7 kWh in May to 1.1 kWh in August, while Enoki ranges from 0.6 kWh in May to 0.9 kWh from June through August. Lion's mane follows a similar trend, increasing from 0.2 kWh in May to 0.6 kWh in July and August (Fig S3).
During winter, all species exhibit negative transmission energy, indicating heating demand rather than cooling demand. For example, Oyster reaches 0.8 kWh in January and December, Lion's mane −0.7 kWh in January and 0.3 kWh in December, and Enoki −0.4 kWh in January and 0.3 kWh in December. Although transmission contributes less to total thermal load than ventilation, it responds strongly to monthly temperature fluctuations, reinforcing the pattern of high cooling demand in summer and heating demand in winter. Overall, the combined climate data, species-specific ΔT, and resulting E(vent) and E(trans) values demonstrate that ambient temperature strongly governs heating and cooling requirements. Species cultivated at lower setpoints (Enoki, Maitake) are considerably more sensitive to warm climates than those grown near ambient conditions (Oyster, Lion's mane).
3.5. Production cost analysis
The cost analysis across four production scales (50, 100, 200, and 500 lb per day), reveals clear economies of scale for all mushroom species evaluated. As production volume increases, the unit cost per pound declines substantially, highlighting the economic advantage of scaling controlled-environment mushroom operations. Oyster and Enoki exhibit the highest cost efficiency, with per-pound costs dropping from $5.1 at 50 lb per day to $2.1 at 500 lb per day, a nearly 59% reduction. Lion's mane, the most resource-intensive species, also shows significant cost savings, decreasing from $6.1 to $3.2 per pound (48% reduction), while Maitake follows a similar trend, falling from $6.0 to $3.0 per pound (50% reduction). These reductions result from more efficient utilization of substrate, spawn, electricity, and labor, as well as better amortization of fixed infrastructure, including climate-control systems, environmental sensors, and automated handling equipment.The steepest unit-cost reduction occurs between 200 lb per day and 500 lb per day, marking the production scale at which the benefits of scaling outweigh the added operational complexity. As output increases, fixed costs are distributed over a larger production volume, lowering per-pound costs without a proportional rise in inputs. Across all scales, Lion's mane remains the costliest to produce due to its strict environmental requirements and slower growth cycle. In contrast, Oyster and Enoki remain the most economical, suitable for both small- and large-scale cultivation. These results underscore that scaling production capacity is a key strategy for improving economic performance and achieving competitive unit costs in container-based mushroom farming. Consistent with Dushyant Kumar et al., larger-scale mushroom farms achieve lower per-unit production costs due to economies of scale, as fixed costs are spread over higher output and bulk purchasing reduces the unit costs of compost, spawn, labor, and energy.51
3.6. NPV analysis
Net Present Value (NPV) is a key metric for evaluating the financial feasibility of an investment, comparing the present value of projected cash inflows with the discounted value of capital and operating expenditures over the project lifetime. In this study, NPV was used to assess the profitability of container-based mushroom cultivation for four species Oyster, Enoki, Maitake, and Lion's mane under an 80% loan and 20% equity financing structure with a 30% corporate tax rate. All capital equipment and container infrastructure were depreciated using the straight-line method over a 10-year useful life (Fig. 8).At the smallest scale of 50 lb per day, only Maitake, Enoki, and Lion's mane achieve positive NPVs ($1686, $14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 746, and $235
746, and $235![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 297, respectively). At the same time, Oyster remains unprofitable (–$322
297, respectively). At the same time, Oyster remains unprofitable (–$322![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 758), reflecting the difficulty of covering fixed infrastructure and operational costs at low volumes. At 100 lb per day, profitability improves for all species except Oyster, with Lion's mane showing the strongest performance ($619
758), reflecting the difficulty of covering fixed infrastructure and operational costs at low volumes. At 100 lb per day, profitability improves for all species except Oyster, with Lion's mane showing the strongest performance ($619![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 195), followed by Enoki ($181
195), followed by Enoki ($181![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 765) and Maitake ($164
765) and Maitake ($164![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 066). Oyster's NPV remains negative (–$441
066). Oyster's NPV remains negative (–$441![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 563), though the deficit is smaller, signaling early benefits of scale.
563), though the deficit is smaller, signaling early benefits of scale.
At 200 lb per day, all species achieve positive NPVs for the first time, marking this as the minimum scale for universal financial viability. Lion's mane leads with $2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 011
011![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 248, followed by Enoki ($1
248, followed by Enoki ($1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 140
140![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 062), Maitake ($1
062), Maitake ($1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 106
106![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 910), and Oyster ($81
910), and Oyster ($81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 229). This scale demonstrates effective amortization of fixed costs and improved operational efficiency. At the largest scale (500 lb per day), profitability is maximized. Lion's mane remains the most lucrative ($7
229). This scale demonstrates effective amortization of fixed costs and improved operational efficiency. At the largest scale (500 lb per day), profitability is maximized. Lion's mane remains the most lucrative ($7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 538
538![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 331), followed by Enoki ($5
331), followed by Enoki ($5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 365
365![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 878), Maitake ($5
878), Maitake ($5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 284
284![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 834), and Oyster ($2
834), and Oyster ($2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 688
688![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 702). High output volumes, optimized energy use, reduced per-unit labor, and efficient substrate handling drive strong positive cash flows. Detailed revenue and discounted net cash flow data for the four species are presented in Tables S5–S8.
702). High output volumes, optimized energy use, reduced per-unit labor, and efficient substrate handling drive strong positive cash flows. Detailed revenue and discounted net cash flow data for the four species are presented in Tables S5–S8.
Production scale is the dominant driver of profitability in container-based mushroom cultivation. High-value species such as Lion's mane and Maitake consistently yield superior financial returns, whereas lower-margin species like Oyster become financially attractive only at larger scales. Annual revenues and discounted net cash flows across scales (50–500 lb per day) demonstrate clear economies of scale, with the most substantial gains observed at 200 and 500 lb per day. These results highlight the importance of both production scale and species selection in achieving sustainable, long-term financial performance.
3.7. Payback period evaluation
Payback period analysis demonstrates a strong dependence of financial feasibility on production scale for container-based mushroom farming (Fig. 9). At the smallest scale (50 lb per day), Oyster, Maitake, and Enoki mushrooms do not achieve payback within the 10-year project horizon, indicating high financial risk, whereas Lion's Mane reaches a payback period of 5.8 years. At 100 lb per day, Oyster remains unprofitable within the project timeframe, while payback periods improve to 8.0 years for Maitake, 7.7 years for Enoki, and 4.9 years for Lion's Mane, reflecting gradual cost recovery but continued capital constraints at small scales. At 200 lb per day, financial performance improves substantially across all species, with payback periods of 9.8 years for Oyster, 4.6 years for Maitake, 4.4 years for Enoki, and 3.4 years for Lion's Mane, demonstrating more efficient utilization of fixed infrastructure and operational scaling. The most favorable outcomes occur at 500 lb per day, where all species achieve rapid capital recovery, with payback periods of 3.4 years for Oyster, 2.5 years for Maitake, 2.4 years for Enoki, and 2.1 years for Lion's Mane.Importantly, all reported payback periods already include an additional one year to account for container installation, infrastructure setup, system commissioning, and ramp-up to steady mushroom production, providing a realistic estimate of time to profitability. The results highlight the economic advantage of scaling production, as larger systems distribute capital and operating costs over higher output, while small-scale farms remain financially vulnerable unless supported by premium pricing, external funding, or targeted efficiency improvements.
4 Conclusion and future directions
This study demonstrates that container-based mushroom farming offers a scalable, hygienic, and efficient platform for year-round production. The modular design enables flexible species selection, batch scheduling, and environmental zoning, while standardized infrastructure ensures consistent climate control and reduces the risk of contamination. Production scale was identified as the primary driver of financial performance. Small-scale systems (50–100 lb per day) incur high per-unit costs and long payback periods, particularly for low-margin species such as Oyster, making them financially challenging without subsidies, premium markets, or technological support. In contrast, larger systems (200–500 lb per day) benefit from economies of scale, achieving lower operating costs, positive NPVs, and faster capital recovery. High-value species such as Lion's mane consistently deliver quicker returns, highlighting their commercial viability.Beyond economics, containerized farms provide a controlled, climate-resilient solution that can operate independently of location or season. Future work should explore renewable energy integration (e.g., solar-assisted HVAC), location-specific energy optimization, automation of harvesting and substrate handling, and the application of artificial intelligence for real-time climate control. Incorporating life-cycle assessment and carbon footprint analysis would further elucidate environmental sustainability. Additional opportunities include cultivating medicinal fungi and vertical stacking to increase yield density. With appropriate technological and financial support, container-based mushroom cultivation has strong potential as a robust, scalable, and climate-resilient strategy for sustainable global food production.
Author contributions
Mahsa Alian: methodology, visualization, resources, writing – original draft, review & editing. Sunil P. Dhoubhadel: techno-economic analysis, editing; Sandesh Risal and Pratikshya Tiwari: setting up the model, calculations, and techno-economic analysis; Weihang Zhu: editing, and funding acquisition; Venkatesh Balan: conceptualization, methodology, resources, writing original draft, review & editing, and funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.Abbreviations
| NPV | Net Present Value |
| CapEx | Capital Expenditure |
| OpEx | Operational Expenditure |
| BE | Biological Efficiency |
| WACC | Weighted Average Cost of Capital |
| HVAC | Heating, Ventilation, and Air Conditioning |
Data availability
All data supporting the findings of this article are presented in the manuscript's tables and figures. No restrictions apply to the availability of these data.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6fb00006a.
Acknowledgements
This project was supported by the National Institute of Food and Agriculture, U.S. Department of Agriculture, under award numbers 2021-67022-34889 and 2023-51300-40853. We also acknowledge the University of Houston Energy Transition Institute, supporting two undergraduate students (Mr Ayush Patel and Mr Viet-Joseph Dinh) who contributed to specific components of the project. Thanks to Mr Shiresh Bhatnagar from Netventix, Sugarland, Texas, and his team for drafting some of the figures for this manuscript.References
- D. Sharma, S. Ramniwas, R. Mugabi, J. Uddin and G. A. Nayik, Revolutionizing mushroom processing: innovative techniques and technologies, Food Chem.: X, 2024, 23, 101774, DOI:10.1016/j.fochx.2024.101774.
- V. Balan, W. Zhu, H. Krishnamoorthy, D. Benhaddou, J. Mowrer, H. Husain and A. Eskandari, Challenges and opportunities in producing high-quality edible mushrooms from lignocellulosic biomass at small scale, Appl. Microbiol. Biotechnol., 2022, 106(4), 1355–1374, DOI:10.1007/s00253-021-11749-2.
- R. Skorbiansky, Commercial Organic Mushroom Production by State, USDA Econ Res Serv, 2023 Search PubMed.
- USDA, Agaricus and Specialty Mushroom Sales, Price, and value—United States: 2019–2020, 2020–2021, and 2021–2022, USDA, 2022 Search PubMed.
- L. Bhagarathi, G. Subramanian and P. N. B. DaSilva, A review of mushroom cultivation and production, benefits and therapeutic potentials, Int. J. Biol. Pharm. Health Sci., 2023, 15, 01–56, DOI:10.30574/wjbphs.2023.15.2.0333.
- N. Gupta, M. Mehta and K. Singh, Benefits, challenges and opportunities in mushroom production: a review, Pharma Innov., 2022, 11(3), 360–364 Search PubMed.
- O. Quaicoe, F. Asiseh and A. S. Aloka, Enhancing year-round profitability for small-scale ranchers: an economic analysis of integrated cattle and mushroom production system, Sustainability, 2024, 16(13), 532, DOI:10.3390/su16135320.
- J. Vunduk, D. Tura and A. Y. Biketova, Medicinal mushroom nutraceutical commercialization: two sides of a coin, Wild Mushrooms, 2022, 10, 89–131, DOI:10.1201/9781003152583-5.
- Farms that sell directly to consumers may stay in business longer, https://www.usda.gov/about-usda/news/blog/farms-sell-directly-consumers-may-stay-business-longerUSDA, 2016.
- J. Nongthombam, A. Kumar, B. G. Ladli, M. Madhushekhar and S. Patidar, A review on study of growth and cultivation of oyster mushroom, Plant Cell Biotechnol. Mol. Biol., 2021, 22(5), 55–65 Search PubMed.
- A. Sujatanagarjuna, S. Kia, D. F. Briechle and B. Leiding, MushR: a smart, automated, and scalable indoor harvesting system for gourmet mushrooms, Agriculture, 2023, 13(8), 1533, DOI:10.3390/agriculture13081533.
- K. A. Subbiah and V. Balan, A comprehensive review of tropical milky white mushroom (Calocybe indica), Mycobiology, 2015, 43(3), 184–194, DOI:10.5941/MYCO.2015.43.3.184.
- L. He, L. Fu, W. Fang, X. Sun, R. Suo, G. Li, G. Zhao, R. Yang and R. Li, IoT-based urban agriculture container farm design and implementation for localized produce supply, Comput. Electron. Agric., 2022, 203, 107445, DOI:10.1016/j.compag.2022.107445.
- N. Wagner, D. Morrish and M. Juarez, Identifying the influential factors, benefits and challenges of hydroponic shipping container farm businesses: a snapshot of farmers' perceptions, Renew. Agric. Food Syst., 2021, 36(6), 519–526, DOI:10.1017/S1742170521000211.
- N. Sergeyeva and T. Vasilchenko, Economic justification for mushroom cultivation, E3S Web Conf., 2024, 494, 04031, DOI:10.1051/e3sconf/202449404031.
- I. Kurniawan, M. Muthoifin, Z. A. Arif, H. A. Lubis, H. La Ramba, M. S. Apriantoro, A. I. Hambali and J. H. Srifyan, Mushroom cultivation for farmer existence: a normative and economic review, Solo Int. Collab. Publ. Soc. Sci. Humanit, 2023, 1(02), 119–131, DOI:10.61455/sicopus.v1i02.50.
- S. Bijla, Status of mushroom production: global and national scenario, Mushroom Res., 2023, 2024(6), 84–92, DOI:10.36036/MR.32.2.2023.146647.
- S. Bijla and V. Sharma, Economic analysis of shiitake mushroom cultivation as an agribusiness enterprise in India, Econ. Aff., 2023, 1101–1106, DOI:10.46852/0424-2513.2.2023.15.
- A. Keneni, Utilizing locally accessible substrate to maximize oyster mushroom (Pleurotus ostreatus) growth and bioconversion efficiency in Ambo, Central Ethiopia, Turk. J. Food Agric. Sci., 2023, 5(2), 68–77, DOI:10.53663/turjfas.1256506.
- K. Deepalakshmi and M. Sankaran, Pleurotus ostreatus: an oyster mushroom with nutritional and medicinal properties, J. Biochem. Technol., 2014, 28(2), 718–726 Search PubMed.
- B. Song, J. Ye, F. L. Sossah, C. Li, D. Li, L. Meng, S. Xu, Y. Fu and Y. Li, Assessing the effects of different agro-residue as substrates on growth cycle and yield of Grifola frondosa and statistical optimization of substrate components using simplex-lattice design, AMB Express, 2018, 8(1), 46, DOI:10.1186/s13568-018-0565-8.
- D. Gonkhom, T. Luangharn, M. Stadler and N. Thongklang, Cultivation and nutrient compositions of medicinal mushroom Hericium erinaceus in Thailand, Chiang Mai J. Sci., 2024, 51, 1, DOI:10.12982/CMJS.2024.028.
- S. Rezaeian and H. R. Pourianfar, A comparative study on bioconversion of different agro-wastes by wild and cultivated strains of Flammulina velutipes, Waste Biomass Valor., 2017, 8, 2631–2642, DOI:10.1007/s12649-016-9698-7.
- J. Y. Choi, J. H. Kim, Y. J. Kim, C. Y. Lee, I. S. Baek, T. M. Ha, C. J. Lee and G. J. Lim, Growth characteristics of oyster mushroom following addition of wheat straw pellet as a substitute for beet pulp, Mushroom J., 2022, 20(04), 270–273, DOI:10.14480/JM.2022.20.4.270.
- L. Wiesnerová, T. Hřebečková, I. Jablonský and M. Koudela, Effect of different water contents in the substrate on cultivation of Pleurotus ostreatus Jacq. P. Kumm., Folia Hortic., 2023, 35(01), 25–31, DOI:10.2478/fhort-2023-0002.
- D. Grimm, E. Sonntag and G. Rahmann, Evaluation of different pasteurization and sterilization methods for oyster mushroom substrates, J. Microbiol. Biotechnol. Food Sci., 2024, 13(05), e10428, DOI:10.55251/jmbfs.10428.
- J. Jarial RS, K. Jarial and J. N. Bhatia, Comprehensive review on oyster mushroom species (Agaricomycetes): Morphology, nutrition, cultivation and future aspects, Heliyon, 2024, 10(5), e26539, DOI:10.1016/j.heliyon.2024.e26539.
- S. M. Barreto, M. V. López and L. Levin, Effect of culture parameters on the production of the edible mushroom Grifola frondosa (maitake) in tropical weathers, World J. Microbiol. Biotechnol., 2007, 24(08), 1361–1366, DOI:10.1007/s11274-007-9616-z.
- S. A. Bacha, S. Ali, Y. Li, H. U. Rehman, S. Farooq, A. Mushtaq, S. A. Wahocho and S. M. Aslam, Lion's mane mushroom: new addition to food and natural bounty for human wellness, Int. J. Biosci., 2018, 13, 396–402, DOI:10.12692/ijb/13.4.396-8.
- S. Attaran Dowom, S. Rezaeian and H. R. Pourianfar, Agronomic and environmental factors affecting cultivation of the winter mushroom (Enokitake): achievements and prospects, Appl. Microbiol. Biotechnol., 2019, 103, 2469–2481, DOI:10.1007/s00253-019-09652-y.
- C. Muswati, K. Simango, L. Tapfumaneyi, M. Mutetwa and W. Ngezimana, Effects of different substrate combinations on growth and yield of oyster mushroom (Pleurotus ostreatus), Int. J. Agron., 2021, 1(2021), 9962285, DOI:10.1155/2021/9962285.
- S. Montoya, C. E. Orrego and L. Levin, Growth, fruiting and lignocellulolytic enzyme production by the edible mushroom Grifola frondosa (maitake), World J. Microbiol. Biotechnol., 2012, 28(4), 1533–1541, DOI:10.1007/s11274-011-0957-2.
- S. Tuğa, N. Çömlekçioğlu and M. Soylu, Effects of substrate formulation on yield and yield components in enoki (Flammulina velutipes) cultivation, Int. J. Agric. Life Sci., 2019, 9(1), 26–33 Search PubMed.
- F. J. Gea and M. J. Navarro, Mushroom diseases and control, in Edible and Medicinal Mushrooms: Technology and Applications, 2017, vol. 5, pp. 239–259, DOI:10.1002/9781119149446.ch12.
- S. Dhiman, R. Gangwar and D. Pant, Cost and return of oyster cultivation in Rajnandgaon district of Chhattisgarh, Pharma Innov., 2022, 11(9), 1043–1048 Search PubMed.
- S. Kumar, S. Susila and S. Singh, Cost and return of oyster cultivation in Rajnandgaon district of Chhattisgarh, Pharma Innov., 2022, 11(12), 4266–4269 Search PubMed.
- S. Sachan and R. Kumar, Cost benefit analysis and marketing of mushroom in Uttar Pradesh, Plant Arch., 2020, 20, 2532–2536 Search PubMed.
- R. R. Ahmed, M. A. Niloy, M. S. Islam, M. S. Reza, S. Yesmin, S. B. Rasul and J. Khandakar, Optimizing tea waste as a sustainable substrate for oyster mushroom (Pleurotus ostreatus) cultivation: biological efficiency and nutritional aspects, Front. Sustain. Food Syst., 2023, 7, 1308053, DOI:10.3389/fsufs.2023.1308053.
- B. Robinson, K. Winans, A. Kendall, J. Dlott and F. Dlott, A life cycle assessment of Agaricus bisporus mushroom production in the USA, Int. J. Life Cycle Assess., 2019, 24, 456–467, DOI:10.1007/s11367-018-1456-6.
- S. Kuruneru and Y. Too, A new numerical method for determining heat transfer and packing distribution in particle heat exchangers for concentrated solar power, Int. J. Heat Mass Tran., 2021, 1(90), 108805, DOI:10.1016/j.ijheatfluidflow.2021.108805.
- M.-A. Meilleur, D. Bastien and D. Monfet, Modeling mushrooms' carbon dioxide emission and heat exchange rates for synergistic cultivation with leafy greens, Sustainability, 2023, 15(24), 16740, DOI:10.3390/su152416740.
- M. Rabani, H. B. Madessa, N. Nord, P. Schild and M. Mysen, Performance assessment of all-air heating in an office cubicle equipped with an active supply diffuser in a cold climate, Build. Environ., 2019, 156, 123–136, DOI:10.1016/j.buildenv.2019.04.017.
- U.S. Energy Information Administration (EIA), EIA, 2024, https://www.eia.gov/electricity/annual/ElectricPowerAnnual.
- USA Climate Data, USA Climate Data database, 2025, https://www.usclimatedata.com/climate/houston/texas/united-states/ustx0617.
- B. B. Dey, M. A. Ador, M. M. Haque, J. Ferdous, M. A. Halim, M. B. Uddin and R. Ahmed, Strategic insights for sustainable growth of mushroom farming industry in Bangladesh using SWOT-AHP and TOPSIS, Heliyon, 2024, 10, e36956, DOI:10.1016/j.heliyon.2024.e36956.
- A. J. Auerbach, in Corporate Taxation in the US, Econ. Pol., 1987, pp. 375–431 Search PubMed.
- R. C. Macalalad, B. W. Yun and H. S. Hwang, Cost-benefit analysis of a smart greenhouse facility in Western Visayas and Northern Mindanao, Philippines, J. Int. Dev., 2024, 19, 19–40, DOI:10.34225/jidc.2024.19.2.19.
- R. Rismawati, D. I. Wardhana and A. N. Nalawati, Analysis of added value and financial feasibility of the oyster mushroom agroindustry in Jember Regency, Int. J. Appl. Sci., 2024, 3, 137–149, DOI:10.32528/ias.v3i2.637.
- A. Damodaran, Book value multiples, in Investment Valuation: Tools and Techniques for Determining the Value of Any Asset, 2012, pp. 511–541 Search PubMed.
- D. Ram Singh and K. B. Sharma, Cost benefit analysis and marketing of mushroom in Haryana, Agric. Econ. Res. Rev., 2010, 23, 165–171, DOI:10.22004/ag.econ.92164.
- U. Pradesh, D. Kumar, G. C. Patel and M. K. Mishra, A comprehensive economic study of production and marketing of mushrooms in Varanasi district, Int. J. Agric. Ext. Soc. Dev., 2024, 7, 360–365, DOI:10.33545/26180723.2024.v7.i11e.1342.
| This journal is © The Royal Society of Chemistry 2026 |








