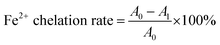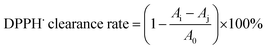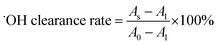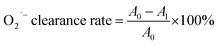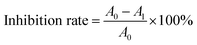Open Access Article
Open Access ArticleUnlocking the potential of cysteine–xylose Maillard reaction intermediates as natural flavor precursors: a comprehensive study on flavor regulation, storage stability, and antioxidant activity
Di
Kang
b,
Lin
Jiang
a,
Songjin
Zheng
b,
Yuan
Hu
a,
Haifeng
Wang
a,
Teng
Li
a,
Yuying
Fu
 *a and
Yun
Zhai
*a and
Yun
Zhai
 *a
*a
aSchool of Food Science and Biotechnology, Zhejiang Gongshang University, Hangzhou, 310018, People's Republic of China. E-mail: 18352538819@163.com; webfu@126.com; wangmumu542@163.com; 15125949093@163.com; whf4341514@163.com; tli0718@mail.zjgsu.edu.cn
bChina Tobacco Hebei Industrial Co., Ltd, Shijiazhuang, 050051, People's Republic of China. E-mail: 1283201616@qq.com; zsj211@126.com
First published on 11th March 2026
Abstract
2-Threityl-thiazolidine-4-carboxylic acid (TTCA) and Amadori rearrangement products (ARPs), intermediates of a cysteine–xylose (Cys–Xyl) Maillard reaction system, have been proven to form desirable flavors during thermal processing. The results showed that both TTCA and ARPs revealed excellent environmental stability at low temperatures (≤25 °C), neutral pH (7), and low water activity (0.113). Under storage conditions of 40 °C, pH 9, and 0.843 water activity, the loss rates of TTCA and ARPs reached 7.06% and 12.17%, 11.19% and 21.25% and 35.77% and 60.61%, respectively. Further research found that TTCA and ARP have increased Fe2+ chelating ability, ferric ion reducing capacity, and free radical scavenging ability within a concentration range of 0.5 to 3.0 mg mL−1. Additionally, different heat treatment conditions as well as the addition of different exogenous amino acids could regulate the flavor formation profile and characteristics of TTCA, making it suitable for different real food systems and broadening its processing adaptability. In particular, when the pH was increased from 5.5 to 8, the meaty flavor of the system gradually lightened, while flavor characteristics such as popcorn flavor, burnt flavor, and nutty flavor gradually strengthened. This research explored the potential of MRIs, natural flavor precursors, to serve as supplements and partial alternatives to synthetic additives. These findings provide a theoretical basis for developing more sustainable food systems and may help reduce the environmental footprint of food processing.
Sustainability spotlightIn the context of a growing global pursuit of sustainable food systems, the development of natural, efficient flavor precursors and antioxidants has become a critical direction for the food industry. The use of TTCA/ARPs as natural flavor precursors and antioxidants can contribute to: (1) reducing reliance on synthetic additives, supporting cleaner labels and potentially lowering the environmental burden associated with chemical synthesis; (2) minimizing food waste through enhanced product stability and extended shelf-life; and (3) optimizing energy efficiency in food processing, as flavor formation can be regulated under varied, potentially milder thermal conditions. Utilizing natural and renewable precursors (cysteine and xylose) in a value-added reaction process. |
1 Introduction
In the context of a growing global pursuit of sustainable food systems, the development of natural and efficient additives has become a critical direction for the food industry. Maillard reaction intermediates (MRIs) and Amadori/Heyns rearrangement compounds (ARPs/HRPs), which are naturally derived, hold significant potential as clean-label alternatives to synthetic flavor precursors and antioxidants.1,2 Compared to the complex and variable nature of complete Maillard reaction products (MRPs), MRIs provide a stable and controllable source for generating ideal flavor and color upon thermal processing.3 In particular, the Cys–Xyl system yields both the conventional ARP and a unique thiazolidine derivative, TTCA.4,5 Current studies have investigated the fundamental aspects of TTCA and ARPs,6,7 including their aqueous-phase tracing preparation mechanism, characteristic patterns of flavor formation and yield enhancement.8,9 These investigations preliminarily demonstrate their advantages in controlled flavor formation, underscoring their potential as flavor precursors.Existing studies confirm that the flavor intensity in thermally processed MRIs exhibits significant advantages; however, the profile of characteristic volatile compounds generated from MRIs remains less diverse than that of the volatile compounds derived from MRPs.8,9 Although adjusting the reaction parameters is a common regulatory approach, its ability to fully compensate for the lack of flavor complexity may be limited.10–13 The addition of exogenous amino acids presents a promising alternative for modulating the flavor, which could promote Strecker degradation between regenerated amino acids derived from MRI degradation and dicarbonyl compounds to form a characteristic flavor.14,15 Nevertheless, the systematic effect of different exogenous amino acids on the flavor profiles derived from specific intermediates, such as TTCA/ARP, remains unclear. Moreover, comprehensive approaches for regulating the flavor profile of TTCA/ARP are currently lacking. Besides, to enable flavor formation exclusively during subsequent thermal processing, MRIs must maintain stability during storage.16 However, MRIs can further degrade through rearrangement and condensation pathways.17,18 Thus, understanding their behavior during storage and heating is essential for commercial applications. Concurrently, developing natural antioxidants is crucial for food preservation.19–21 MRPs exhibit confirmed antioxidant, antiallergic, and antibacterial activities,22–24 primarily due to the presence of the chromophoric rings of melanoidins.22,25 Certain low-molecular-weight compounds in MRPs, especially reductones and heterocycles, exhibit a greater antioxidant activity than pure melanoidins.19 However, the specific contribution and efficacy of defined intermediate compounds like TTCA and ARPs have not been comparatively evaluated against MRPs.
Therefore, to address the current research gap regarding TTCA and ARPs, this study adopts a unique perspective to investigate the regulatory mechanisms of their flavor characteristics, storage stability, and antioxidant properties and aims to comprehensively evaluate the dual-function potential of TTCA and ARPs as flavor precursors and antioxidants through the following steps: (1) systematic assessment of the storage stability of TTCA and ARPs under different environmental stresses to provide essential data for their commercial storage; (2) elucidation of the directional regulation of the thermal degradation flavor profiles of TTCA by temperature, pH, and various exogenous amino acids to extend their processing adaptability; and (3) evaluation and comparison of the in vitro antioxidant activities of TTCA/ARPs with those of MRPs to clarify their roles and potential in different antioxidant pathways. Hence, this work provides essential data for the commercial storage and application of TTCA/ARPs, extends their processing adaptability, and clarifies their dual-function potential as natural flavor precursors and potential antioxidants, thereby supporting their development as sustainable food additives.
2 Materials and methods
2.1 Chemicals
Analytical grade L-cysteine, D-xylose, sodium hydroxide, 1,2,4-benzenetriol, phenanthroline, potassium ferricyanide, trichloroacetic acid, FeCl3, DPPH, ethanol, 1,10-phenanthroline, FeSO4, ascorbic acid, lecithin, thiobarbituric acid, H2O2, NaOH and HCl were purchased from Sigma-Aldrich Chemical Co. (Shanghai, China). LC-MS-grade water, acetonitrile, deuteroxide ammonium formate and 1,2-dichlorobenzene were purchased from Sinopharm Chemical Reagent Co. Ltd (Shanghai, China).2.2 Synthesis, purification and characterization of isomeric TTCA and ARPs
The preparation, purification and characterization of TTCA and ARPs were carried out by following the methods reported in our previous studies.4,26 An equimolar solution of Cys and Xyl (0.0827 mol L−1) was prepared using deionized water. The prepared solution was maintained at pH 7.4 ± 0.01 using NaOH (6 mol L−1), followed by the heat treatment for 40 min at 90 °C. The reaction vessel was quickly transferred to an ice-bath to terminate the reaction immediately. The obtained reactant was employed to examine the purity of TTCA and ARPs, which were preliminarily separated using an ion exchange resin (H+ Dowex 50WX4, 200–400 mesh, Acros Organics, SERVA, J&K, Beijing, China) with ammonium hydroxide (0.2 mol L−1) as the eluent, and further purification was carried out using a semi-preparative RP-HPLC equipped with an Xbridge Amide column (4.6 mm × 150 mm, 3.5 µm; Waters Co., Milford, MA, USA). The identification and characterization of TTCA and ARP purities were performed using UPLC-ESI-MS (Waters Synapt MALDI Q-TOF MS, USA) and NMR (Bruker DRX 400 MHz spectrometer). The specific procedures of the above-mentioned methods were accomplished by referring to our published study. The mass spectral and NMR spectral information is listed in the SI (Fig. S1 and Table S1).2.3 Assessment of the browning index of the Maillard reaction solution
To assess the browning degree of the melamine reaction solution at different temperatures, the absorbance at 420 nm (A420) was typically measured using a UV-visible spectrophotometer, which served as the browning index. The absorbance was determined by diluting the sample appropriately so that the A420 value was within a reasonable range (0.05 to 1.0).2.4 Construction of the Maillard thermal reaction model system using TTCA as the base material
Maillard thermal reaction model systems were established with TTCA as the base material. The pH of the TTCA solution (10 mmol L−1) was adjusted to 5.5, 7, and 8, respectively, using NaOH (6 mol L−1). The prepared samples were then treated at different temperatures (100 °C, 120 °C, and 140 °C) for 120 minutes.Additionally, an equimolar amount of different exogenous amino acids (Gly, Ala, Met, Leu, Ser, Thr, Pro, Val, Phe, His, Lys, Glu, and Asp) were added to the TTCA solution (10 mmol L−1). The pH of the system was adjusted to 7.0 using NaOH (6 mol L−1), and the thermal reaction was conducted at 120 °C for 120 minutes.
Using high-temperature/high-pressure-resistant glass reaction vessels set in an oil bath, the aforementioned thermal reactions were carried out. Following this, all the mixtures were immediately transferred to an ice bath to halt the reactions. Afterwards, the samples were collected and kept under 4 °C for further analysis.
2.5 Determination of the storage stability of TTCA and ARPs
To investigate the effect of different storage temperatures on the stability of TTCA and ARP intermediates in solid and solution forms, a solid pure lyophilized powder of TTCA and ARPs was placed in self-sealing bags or dissolved in deionized water and configured as a solution with a concentration of 100 mmol L−1, and placed in the ambient temperatures of 4 °C, 25 °C and 40 °C. The changes in concentration, A420, A294, and color were recorded every 10 days during storage, and storage stability was investigated over a period of 60 days.In addition, the storage stability of the TTCA and ARP system was observed by varying the pH of the system while maintaining the same ambient temperature, and the same metrics were also examined. For TTCA and ARP solid samples, the effect of ambient humidity on their storage stability was investigated. A fixed mass of the dried powder of the intermediate was taken in an open glass Petri dish and transferred to a sealed desiccator, the bottom of which was filled with different saturated salt solutions configured to maintain the ambient moisture activity of the system, and a comparison of the moisture activities of different saturated salt solutions at 25 °C is shown in Table S2.
2.6 Determination of the antioxidant properties of intermediates
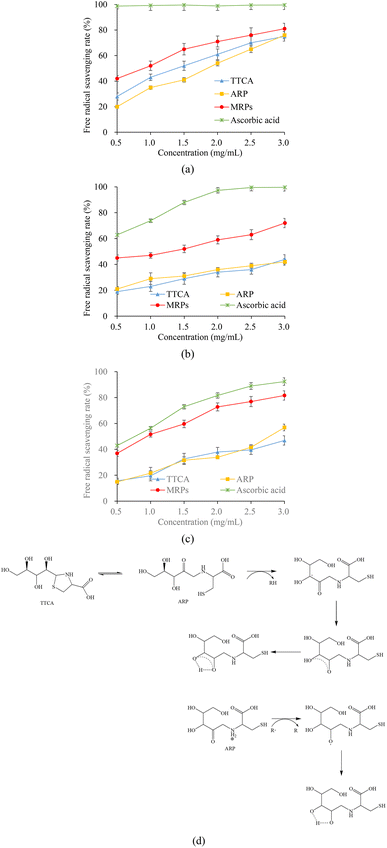
where A0 and A1 are the absorbance of the blank and sample, respectively.
where AS represents the absorbance of the sample; A1 denotes the absorbance of the control reaction solution, which contains 1,10-phenanthroline, FeSO4 and H2O2; and A0 corresponds to the absorbance of the blank solution, composed solely of FeSO4 and 1,10-phenanthroline.7
where A0 represents A327 of the blank and A1 represents A327 of the sample.7
where A0 and A1 denote the absorbance of the blank (deionized water as the substitute) and the sample, respectively.7
2.7 Identification and quantification of the characteristic volatile compounds
The analysis of volatile flavor compounds was performed using headspace solid-phase microextraction coupled with gas chromatography-mass spectrometry (HS-SPME-GC-MS). The sample solution (3 g) was added with 3 µL internal standard (1,2-dichlorobenzene, 0.018 µg µL−1 in methanol), which was then placed in a 20 mL headspace vial. The extraction and desorption procedures were carried out by following a previously published method,15 employing a divinylbenzene/carboxen/polydimethylsiloxane (CAR/PDMS/DVB, 75 µm) SPME fiber. Compound separation and identification were achieved using an Agilent 7890B gas chromatograph interfaced with a 5977B mass selective detector. Following 10 minutes of thermal desorption at 250 °C in the injection port, the volatiles were transferred onto an analytical column. Chromatographic separation was performed using a 30 m DB-Wax capillary column (0.25 mm i.d. and 0.25 µm film thickness). Specific instrumental conditions were adopted from our previous research.15Compound identification was based on matching the mass spectra against the NIST 17 and WILEY 07 databases, comparing the calculated Kovats retention indices (RI) with the values reported in the literature, and referencing literature data. To facilitate accurate RI matching, the retention indices of target compounds were calculated by analyzing a C7–C30n-alkane series under identical chromatographic conditions. For compounds with available reference standards, their contents were determined by the standard curve method with the internal standard calibration (see Table S3); for those without reference standards, quantification was performed just using the internal standard method.
2.8 Data analysis
We conducted all experiments in triplicate, and the data are presented as mean ± standard deviation. Data processing involved the following steps: (1) analysis using SPSS Statistics 22.0 and Microsoft Excel 2010 and (2) multiple comparisons via Duncan's multiple range test, where a p-value of less than 0.05 indicated statistical significance.3 Results and discussion
3.1 Regulating the characteristic flavor formation of the TTCA intermediate through parameter regulation
Given the established similarity in flavor formation pathways between TTCA and ARP (differing primarily in rate), TTCA was selected to investigate the effects of temperature and pH. Temperature is a key kinetic parameter in controlling the reaction progress. The appropriate increase in temperature is beneficial for accelerating effective collisions between substrates and the thermal degradation of cysteine.29 Comparative studying of heated TTCA models at 100 °C, 120 °C, and 140 °C revealed that the total volatile flavor substances increased markedly, with sulfur-containing compounds showing a leapfrog increase (Table S4 and Fig. 1a). The content of furfural (the dominant furan) increased significantly, reaching 11.039 µg L−1 at 140 °C, which was 3.3 times that at 100 °C. The concentrations of 2-methyl-3-furanthiol and 2-furfurylthiol also increased accordingly. This is because high temperatures promote the enolization of ARPs, leading to the rapid generation and accumulation of the direct precursors like 3-DX and 1-DX for furfural and 4-hydroxy-5-methyl-3(2H)-furanone (Fig. 1f).30 They also increase the generation of furan and 2-methylfuran, which are precursors to thiophene compounds (Fig. 1f). Furthermore, elevated temperatures intensified the thermal degradation of TTCA and free Cys (Fig. S2 and S3), resulting in an increased release of H2S. The boosted supply of these key precursors (α-dicarbonyls, furans, and H2S) strengthened their interactions, thereby promoting the formation of corresponding thiols and thiophenes (Fig. 1f).Key steps such as enolization, retro-aldolization, and Strecker degradation are pH-dependent, and the activity of key precursors like H2S and NH3 also varies with the pH values.3 Therefore, the influence of different reaction pH values (5.5, 7, and 8) on the TTCA thermal reaction system was studied. The characteristic volatile compounds were predominantly sulfur-containing (Table S5). Under the acidic condition (pH 5.5), thiols were most abundant, followed by thiophenes; no nitrogen-containing heterocycles were detected. As the pH increased from 5.5 to 8, thiol and furan contents decreased, while thiazoles increased sharply to 10.952 µg L−1. Notably, 2-acetylthiazole (nutty/popcorn odor) increased significantly with pH, comprising 27.01% of total thiazoles at pH 8. Pyrazines were detected only at pH 8 and 0.838 µg L−1, a concentration much lower than that of sulfur-containing compounds. Post-reaction, the system pH dropped (to 5.1, 4.9, and 4.7, respectively), due to organic acid formation from accelerated TTCA degradation at a higher initial pH. Under acidic conditions, TTCA undergoes 1,2-enolization, generating thiol and furan precursors (Fig. 1f).15 The low thiazole and pyrazine contents under acidic conditions are due to the superior competitiveness of H2S over NH3/amines in capturing reactive α-dicarbonyl intermediates including glyoxal, methylglyoxal, and 2,3-butandione.17 In contrast, alkaline conditions enhanced the nucleophilicity of deprotonated NH3/amines, enabling them to successfully compete for intermediates and drive the formation of thiazoles and pyrazines, imparting roasted notes.17 Alkaline conditions also favor the regeneration of Cys during TTCA degradation, promoting Strecker degradation with α-dicarbonyls and further amplifying thiazole/pyrazine formation (Fig. 1f).3
In summary, the increased temperature universally intensified flavor formation by elevating precursor supply and reaction rates. In contrast, the increased pH altered the fundamental reaction pathway, shifting the flavor profile from sulfur-rich/meaty notes toward nitrogen-containing heterocycles associated with the characteristics of roasted flavor. These findings confirmed that targeted parameter control (temperature and pH) can effectively steer the flavor formation pathways of TTCA, demonstrating its adaptable potential for generating diverse flavor profiles under different processing conditions.
3.2 Addition of exogenous amino acids regulating the formation of characteristic flavors of the TTCA reaction system
While TTCA intermediates produce intense flavors surpassing those of typical MRPs, their volatile compound diversity is limited.31 The formation of key flavor compounds such as pyrazines, pyridines, pyrroles, thiazoles, and Strecker aldehydes may require sufficient exogenous amino acids to participate in subsequent thermal reactions of the intermediates.15,31 To further elucidate the effects of different amino acids, those types with varying isoelectric points and side chain groups were selected to combine with TTCA for thermal reactions. The browning intensity was significantly higher (p < 0.05) in the systems with added amino acids than in the TTCA control (Fig. 1c), likely because amino acids react with carbonyl compounds in later stages to form colored polymers.32Volatile analysis revealed that all systems predominantly generated sulfur-containing compounds (Table 1 and Fig. 1d, e), which was driven by the high reactivity of H2S and NH3 released from TTCA degradation (Fig. 1f).10 Results showed that the side chains of the added amino acids had a negligible influence on sulfur-containing flavor formation (Table 1). High levels of 2-methyl-3-furanthiol and 2-furfuryl thiol were detected across all systems. This consistent presence indicated that added amino acids did not inhibit the key pathway leading to these furans—namely, the dehydration and cyclization of deoxyosones during TTCA degradation (Fig. 1f).33 Furthermore, sulfur heterocycles requiring the participation of α-dicarbonyls (e.g., 2-methyl-thiophene, thieno[3,2-b]thiophene, and thiazole) were detected, and were common to all systems supplemented with Gly, Lys, or Glu (Fig. 1f).34 This reinforces that the core pathway for sulfur-containing flavor generation is governed by α-dicarbonyl formation rather than by the specific side chains of the added amino acids. The addition of the three exogenous amino acids can promote the retro-aldol cleavage of deoxyosones, yielding more short-chain α-dicarbonyl compounds and reductive ketones, which expanded flavor diversity (including sulfur compounds and pyrazines) and contributed to enhanced browning.13 Gly supplementation produced the greatest variety of flavor compounds, possibly due to its propensity to form short-chain reactive aldehydes.35 Lys significantly increased flavor variety (p < 0.05), likely because its additional amino group boosted α-dicarbonyl formation via reactions with sugar chains.36
| Compounds | RIb | RIc | Control | Neutral | Acidic | Basic | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cys | Gly | Ala | Met | Glu | Asp | His | Lys | ||||
| a Notes: Results are presented as mean ± standard deviation; data within a row with different letters are significantly different (p < 0.05), as indicated by Duncan's multiple comparison tests (n = 3). b Linear retention indices calculated using a series of n-alkanes on a DB-W AX column (30 m × 0.25 mm × 0.25 µm). c Linear retention indices searched from https://www.flavornet.org and https://webbook.nist.gov/chemistry/.“ND”: not detected. Compounds marked with an asterisk (*) were quantified using the standard curve method with internal standard calibration. | |||||||||||
| Sufur-containing compounds | |||||||||||
| Thiols and sulfides | |||||||||||
| Dimethyl disulfide | 1085 | 1037 | ND | ND | ND | ND | 0.956 ± 0.013a | ND | ND | ND | ND |
| 3-Mercapto-2-butanone* | 1273 | 1283 | 1.798 ± 0.025a | ND | ND | ND | ND | ND | ND | ND | ND |
| 2-Methyl-3-furanthiol* | 1302 | 1305 | 1.388 ± 0.207f | 4.083 ± 0.092d | 11.569 ± 0.165b | 0.329 ± 0.008g | — | 7.327 ± 0.222c | 0.344 ± 0.007g | 2.536 ± 0.149e | 18.752 ± 1.061a |
| 3-Mercapto-2-pentanone* | 1352 | 1343 | 1.298 ± 0.021a | 0.438 ± 0.01b | ND | ND | ND | ND | ND | ND | ND |
| Dimethyl trisulfide | 1392 | 1370 | ND | ND | ND | ND | 2.031 ± 0.028a | ND | ND | ND | ND |
| 2-Furfurylthiol* | 1426 | 1402 | 4.107 ± 0.152d | 11.591 ± 0.26c | 15.254 ± 0.217b | 2.133 ± 0.003e | ND | 12.107 ±0.366c | 3.176 ± 0.061d | 0.38 ± 0.022f | 29.295 ± 1.658a |
| 3-Thiophenethiol* | 1564 | 1530 | 0.178 ± 0.011e | 12.892 ± 0.289a | 0.74 ± 0.011c | ND | ND | 0.405 ± 0.012e | ND | ND | 2.31 ± 0.131b |
| 2-Thiophenemethanethiol* | 1702 | 1713 | 0.196 ± 0.012d | 0.483 ± 0.011a | 0.267 ± 0.004c | ND | ND | 0.207 ± 0.006d | ND | ND | 0.41 ± 0.023b |
| 2-Methyl-3-[(2-methyl-3-thienyl)dithio]furan | 1732 | — | 0.098 ± 0.006d | ND | 2.804 ± 0.04b | ND | ND | 0.574 ± 0.017c | ND | ND | 4.428 ± 0.251a |
| 1,2,3-Trithiolane | 1794 | — | 0.378 ± 0.026a | ND | ND | ND | ND | ND | ND | ND | ND |
| 3,3′-Dithiobis[2-methyl-furan] | 2139 | — | ND | ND | ND | ND | ND | 3.946 ± 0.119b | ND | ND | 6.862 ± 0.388a |
| Furfuryl sulfide | 2274 | 2223 | ND | ND | 0.159 ± 0.002a | ND | ND | ND | 0.025 ± 0b | ND | ND |
| Bis(2-furfuryl)sulfide | 2419 | — | 0.042 ± 0.008b | 0.972 ± 0.022a | — | ND | ND | ND | ND | ND | ND |
| Subtotal | 9.483 ± 0.199d | 30.46 ± 0.684b | 30.794 ± 0.438b | 2.461 ± 0.011f | 2.987 ± 0.042ef | 24.566 ± 0.743c | 3.545 ± 0.068e | 2.916 ± 0.171ef | 62.057 ±3.512a | ||
| Kinds | 9 | 6 | 6 | 2 | 2 | 6 | 4 | 2 | 5 | ||
| Thiophenes | |||||||||||
| Thiophene* | 1016 | 1022 | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| 3-Methyl-thiophene | 1119 | 1106 | ND | ND | 10.192 ± 0.145a | ND | ND | ND | ND | ND | 0.083 ± 0.005b |
| 2,3-Dihydro-5-methyl-thiophene | 1129 | 1156 | 0 ± 0e | 0.058 ± 0.001d | 0.167 ± 0.002b | ND | ND | 0.126 ± 0.004c | ND | ND | 0.204 ± 0.012a |
| 2-Methyl-thiophene* | 1135 | 1095 | 1.128 ± 0.159c | 0.237 ± 0.005d | 0.147 ± 0.002d | 0.098 ± 0.002d | ND | 5.389 ± 0.163b | 0.141 ± 0.003d | 0.48 ± 0.028d | 12.21 ± 0.691a |
| 2-Ethyl-thiophene | 1179 | 1167 | ND | ND | ND | ND | ND | 0.062 ± 0.002c | ND | ND | 0.083 ± 0.005b |
| 2,5-Dimethyl-thiophene | 1216 | 1202 | ND | ND | 0.464 ± 0.007b | 0.76 ± 0.019a | ND | ND | ND | ND | 0.461 ± 0.026b |
| 2,3-Dimethyl-thiophene | 1232 | 1212 | ND | ND | 0.038 ± 0.001b | ND | ND | 0.207 ± 0.006a | ND | ND | ND |
| Dihydro-2-methyl-3(2H)-thiophenone | 1511 | 1506 | ND | ND | 1.167 ± 0.017b | ND | ND | ND | ND | ND | 1.77 ± 0.1a |
| 2-Thiophenecarboxaldehyde* | 1674 | 1679 | 2.196 ± 0.076b | 1.06 ± 0.024c | 0.541 ± 0.008e | ND | 3.109 ± 0.043a | ND | 0.125 ± 0.002f | 0.078 ± 0.005f | 0.695 ± 0.039d |
| 5-Methyl-2-thiophenecarboxaldehyde* | 1701 | 1785 | 3.649 ± 0.075a | 2.535 ± 0.057c | ND | ND | ND | ND | ND | ND | 2.808 ± 0.159b |
| 2-Acetyl-3-methylthiophene | 1754 | 1760 | ND | ND | 0.488 ± 0.007a | ND | ND | ND | ND | ND | 0.388 ± 0.022b |
| Thieno[3,2-b]thiophene | 1868 | 1843 | 1.302 ± 0.099b | 7.967 ± 0.179a | 0.659 ± 0.009d | 0.038 ± 0.001f | 0.063 ± 0.001f | 0.39 ± 0.012e | 0.192 ± 0.004f | 0.105 ± 0.006f | 0.964 ± 0.055c |
| 1-(2-Thienyl)-1-propanone | 1872 | — | ND | ND | 0.46 ± 0.007b | ND | ND | ND | ND | ND | 0.584 ± 0.033a |
| 2,5-Thiophenedicarboxaldehyde* | 1911 | — | 0.724 ± 0.055d | 1.261 ± 0.028a | 1.015 ± 0.014c | ND | 1.079 ± 0.015b | 0.292 ± 0.009e | ND | ND | ND |
| 2-Methylthieno[2,3-b]thiophene | 1947 | — | 0.19 ± 0.023c | 1.886 ± 0.042a | ND | ND | 0.012 ± 0.000d | ND | ND | ND | 0.24 ± 0.014b |
| Subtotal | 9.189 ± 0.392c | 15.003 ± 0.337b | 15.338 ± 0.218b | 0.895 ± 0.022f | 4.263 ± 0.059e | 6.466 ± 0.196d | 0.457 ± 0.009f | 0.663 ± 0.039f | 20.49 ± 1.16a | ||
| Kinds | 6 | 7 | 13 | 3 | 4 | 7 | 3 | 3 | 14 | ||
| Thiazoles | |||||||||||
| Thiazole* | 1240 | 1262 | 1.796 ± 0.128a | 0.461 ± 0.01d | 0.556 ± 0.008 cd | 0.131 ± 0.003f | — | 0.58 ± 0.018c | 0.353 ± 0.007e | 0.772 ± 0.045b | 0.506 ± 0.029 cd |
| 2-Methyl-thiazole* | 1272 | 1278 | 0.194 ± 0.017d | 0.579 ± 0.013b | ND | ND | ND | 0 ± 0e | ND | 0.612 ± 0.036a | 0.277 ± 0.016c |
| 2,5-Dimethyl-thiazole* | 1301 | 1326 | 2.203 ± 0.143a | 0.127 ± 0.003b | ND | ND | ND | 0.188 ± 0.006b | ND | ND | ND |
| 2-Ethyl-thiazole* | 1319 | 1304 | 0.273 ± 0.008a | 0.152 ± 0.003b | ND | ND | ND | 0 ± 0c | ND | ND | ND |
| 2,4,5-Trimethyl-thiazole* | 1373 | 1390 | 3.199 ± 0.127a | 0.928 ± 0.021b | 0.38 ± 0.005c | ND | ND | 0.442 ± 0.013c | ND | ND | 0.209 ± 0.012d |
| 4,5-Dimethyl-thiazole* | 1378 | 1843 | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| 5-Ethyl-2,4-dimethyl-thiazole* | 1437 | — | 0.587 ± 0.014a | 0.174 ± 0.004d | 0.339 ± 0.005c | ND | ND | ND | ND | ND | 0.38 ± 0.022b |
| 2-Ethyl-4-methylthiazole* | 1449 | 1410 | 1.278 ± 0.117a | 0.284 ± 0.006b | 0.219 ± 0.003b | 0.014 ± 0.000c | ND | ND | 0.018 ± 0c | ND | ND |
| 2-Acetylthiazole | 1646 | 1643 | 8.795 ± 0.213a | 0.538 ± 0.012c | ND | ND | 2.837 ± 0.04b | ND | ND | ND | ND |
| Subtotal | 18.325 ± 0.744a | 3.255 ± 0.073b | 1.494 ± 0.021c | 0.145 ± 0.004d | 2.837 ± 0.04b | 1.211 ± 0.037c | 0.371 ± 0.007d | 1.385 ± 0.081c | 1.371 ± 0.078c | ||
| Kinds | 8 | 9 | 4 | 2 | 1 | 3 | 2 | 2 | 4 | ||
| Total contents of sufur-containing compounds | 36.997 ± 1.1c | 48.718 ± 1.094b | 47.626 ± 0.678b | 3.501 ± 0.037g | 10.087 ± 0.14e | 32.243 ± 0.975d | 4.374 ± 0.084f | 4.963 ± 0.291f | 83.918 ± 4.749a | ||
| Total kinds of sufur-containing compounds | 23 | 22 | 23 | 6 | 6 | 16 | 9 | 7 | 23 | ||
| Nitrogen-containing heterocycles | |||||||||||
| Pyridine | 1189 | 1202 | ND | ND | ND | ND | ND | ND | ND | ND | 0.033 ± 0.002a |
| Methylpyrazine* | 1214 | 1263 | ND | 0.343 ± 0.008d | 1.623 ± 0.023b | ND | 0.423 ± 0.006d | 1.512 ± 0.046b | ND | 0.692 ± 0.041c | 4.055 ± 0.229a |
| 2-Pyridinecarboxaldehyde | 1186 | — | ND | ND | ND | ND | ND | ND | ND | ND | 1.239 ± 0.07a |
| Pyrazine* | 1216 | 1209 | ND | 0.063 ± 0.001 cd | 0.208 ± 0.003 cd | 0.039 ± 0.001d | 0.12 ± 0.002 cd | 0.119 ± 0.004 cd | 0.294 ± 0.006bc | 0.498 ± 0.029b | 5.699 ± 0.323a |
| 3-Methyl-pyridine* | 1307 | 1346 | ND | 0.076 ± 0.002a | ND | ND | ND | ND | ND | ND | ND |
| 2,5-Dimethyl-pyrazine* | 1320 | 1328 | ND | 0.679 ± 0.015b | 0.423 ± 0.006c | ND | 0.069 ± 0.001e | 0.402 ± 0.012c | ND | 0.129 ± 0.008d | 0.927 ± 0.052a |
| 2-(n-Propyl)-pyrazine | 1476 | 1428 | ND | ND | ND | ND | ND | ND | ND | ND | 0.378 ± 0.021a |
| Pyrrole | 1499 | 1518 | ND | ND | 0.228 ± 0.003a | ND | ND | 0.12 ± 0.004b | ND | ND | ND |
| 2-Methyl-1H-pyrrole | 1534 | 1551 | ND | ND | 0.371 ± 0.005b | 0.036 ± 0.001d | ND | 0.139 ± 0.004c | ND | ND | 0.411 ± 0.023a |
| 1H-Pyrrole-2-carboxaldehyde | 2002 | 2028 | ND | ND | ND | ND | 0.836 ± 0.012a | ND | ND | ND | ND |
| Subtotal | ND | 1.161 ± 0.026d | 2.853 ± 0.041b | 0.076 ± 0.002e | 1.448 ± 0.02d | 2.292 ± 0.069c | 0.294 ± 0.006e | 1.319 ± 0.077d | 12.741 ± 0.721a | ||
| Kinds | 0 | 4 | 5 | 2 | 2 | 5 | 1 | 3 | 7 | ||
| Oxygen-containing heterocycles | |||||||||||
| Furan* | 797 | 798 | 0.138 ± 0.01a | ND | ND | ND | ND | 0 ± 0b | 0 ± 0b | ND | ND |
| 2-Methyl-furan* | 851 | 829 | 0.019 ± 0.005f | ND | 6.884 ± 0.098b | 0.505 ± 0.012e | ND | 4.741 ± 0.143c | 0.276 ± 0.005ef | 1.589 ± 0.093d | 8.225 ± 0.465a |
| 2-Ethyl-furan | 913 | 949 | ND | ND | 0.041 ± 0.001b | ND | ND | 0.042 ± 0.001b | 0 ± 0c | ND | 0.089 ± 0.005a |
| Furfural* | 1457 | 1460 | 5.793 ± 0.155a | ND | 1.05 ± 0.015e | 0.242 ± 0.006g | 1.968 ± 0.027c | 1.019 ± 0.031e | 3.589 ± 0.069b | 1.369 ± 0.08d | 0.664 ± 0.038f |
| 2-Acetylfuran | 1491 | 1500 | ND | ND | ND | ND | ND | 1.189 ± 0.036a | 0.378 ± 0.007b | ND | ND |
| 1-(2-Furanyl)-ethanone* | 1497 | 1501 | 0.136 ± 0.077a | ND | ND | ND | ND | ND | ND | ND | ND |
| 5-Methylfurfural | 1542 | 1567 | ND | ND | 1.34 ± 0.019a | 0.703 ± 0.017b | ND | ND | ND | ND | ND |
| 2(5H)-Furanone* | 1748 | 1767 | 0.072 ± 0.005a | ND | ND | ND | ND | ND | ND | ND | ND |
| 4-Hydroxy-5-methyl-3(2H)-furanone* | 2108 | 2124 | ND | ND | ND | ND | ND | 0.19 ± 0.006a | ND | ND | ND |
| Subtotal | 6.158 ± 0.231c | ND | 9.314 ± 0.133a | 1.45 ± 0.036g | 1.968 ± 0.027f | 7.182 ± 0.217b | 4.243 ± 0.082d | 2.958 ± 0.174e | 8.978 ± 0.508a | ||
| Kinds | 5 | 0 | 4 | 3 | 1.000 | 5 | 3 | 2 | 3 | ||
| Total | 43.155 ± 1.223d | 49.878 ± 1.12c | 59.793 ± 0.851b | 5.027 ± 0.075f | 13.503 ± 0.188e | 41.717 ± 1.262d | 8.911 ± 0.172e | 9.24 ± 0.542e | 105.637 ± 5.978a | ||
| Total kinds | 28 | 26 | 32 | 11 | 9 | 26 | 13 | 12 | 33 | ||
Compared to the TTCA control, systems with Gly, Lys, or Glu showed a reduced thiazole content/variety but increased nitrogenous heterocycles, confirming greater NH3 participation in pyrazine formation (Fig. 1f).37 The TTCA-Lys thermal reaction system could generate pyrazines. At the system pH (7.0), some Lys residues remained undissociated and acted as nucleophiles, reacting with sugar residues to produce short-chain α-dicarbonyls that subsequently formed pyrazines38 (Fig. 1f). In contrast, TTCA-His produced less meat or roasting aroma (Table 1), as its imino group is less reactive than amino group to form carbonyl compounds as a nucleophile.29 Moreover, the imidazole group with strong basic properties has a lower reactivity, and is less likely to participate in the formation of flavor compounds.35 In addition to Lys, the TTCA thermal reaction system supplemented with Glu detected a considerable amount of pyrazine compounds, which is consistent with previous research.39 Beyond pyrazines, pyrroles were detected in all three systems, linked to reactions between α-dicarbonyls (MGO/GO) and NH3 (Fig. 1f).40 Among them, 2-methylpyrrole and pyrrole were synthesized via an aldol condensation reaction between MGO/GO and acetaldehyde, followed by nucleophilic addition with NH3 and dehydration. 2-Pyrrolecarboxaldehyde was produced by nucleophilic addition of NH3 to a 3-DX isomer, followed by dehydration and cyclization (Fig. 1f).41 The large amount of pyridine compounds in the TTCA-Lys reaction system was generated by the Strecker degradation reaction between Lys and α-dicarbonyl compounds, followed by subsequent dehydration reactions (Fig. 1f).42
Furans (mainly 2-methylfuran and furfural) were formed in TTCA reaction systems with Gly, Lys, or Glu but not in a TTCA-Cys system, indirectly confirming the critical role of H2S from Cys degradation in diverting precursors toward sulfur-containing flavors. In summary, supplementing TTCA with Gly or Lys enriched the overall flavor profile (sulfur compounds and pyrazines), while Glu specifically promoted pyrazine formation (p < 0.05). Exogenous amino acids help shift the reaction equilibrium by ensuring sufficient α-dicarbonyl precursors for key flavor pathways.43 Simultaneously, incorporating various exogenous amino acids can markedly enhance the browning development in the TTCA thermal reaction system. These results suggest that combining Gly, Lys, or Glu with TTCA could enhance TTCA's versatility for generating desired colors and flavors in diverse food applications.
3.3 Extremely high environmental stability of TTCA and ARP intermediates to act as natural flavoring additives
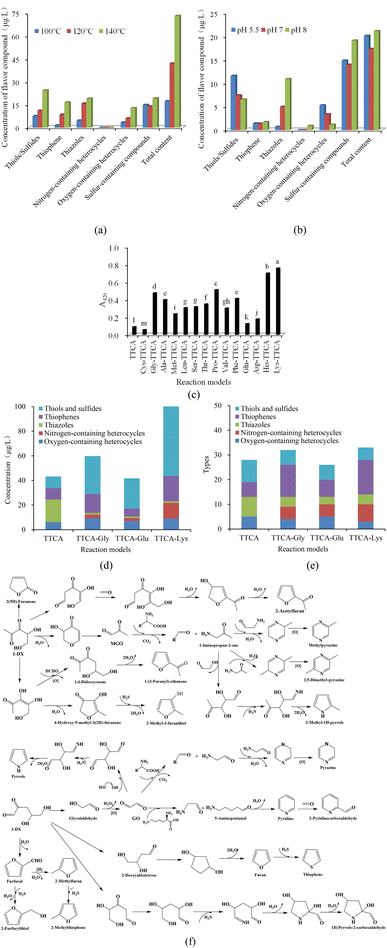
The storage environment is a key factor affecting the structure, content and physicochemical properties of all kinds of chemical products in the storage process.44 In this study, the storage stability of TTCA and ARP was investigated. The key finding is that TTCA exhibits superior storage stability compared to ARPs across all conditions tested, attributed to its stable cyclic molecular structure.3 Both intermediates were highly stable at 4 °C and 25 °C. Degradation became noticeable only under stress conditions: at 40 °C, and the TTCA concentration decreased by 7.06% after 60 days, less than ARP's 12.17% loss. The increased degradation at elevated temperatures is linked to water autoionization,45 which catalyzes the enolization and ring-opening of ARPs.44 Corresponding increases in the A294 and A420 values at 40 °C (Fig. 2c and d) indicate the formation of carbonyl intermediates and browning products.21The stability of TTCA and ARP solutions stored at pH 5.5, 7 and 9 was monitored for 60 days at room temperature (Fig. 2e–h). The intermediates maintained stable concentrations under acidic and neutral conditions (pH 5.5 and 7). In contrast, at pH 9, the concentrations of TTCA and ARPs decreased by 11.19% and 21.25% after 60 days, respectively. Meanwhile, the rise of A294 and A420 also indicate the alkaline-promoted degradation into downstream products. The slight decrease in TTCA concentration at pH 5.5—accompanied by opposing trends in A420 (increase) and A294 (decrease)—suggested a transformation distinct from degradation, potentially due to acid-catalyzed hydrolysis or reversion to N-xylosylamine.45 This phenomenon is attributed to the formation of rearrangement reaction by the deprotonation of imine positive ions, so the presence of H+ under acidic conditions could promote the reverse reaction.21
The moisture content and water activity values of food products are key indicators of their shelf life and eating quality.46 The stability of solid TTCA and ARP was assessed for 60 days under varying humidity levels. When stored under dry conditions (aw 0.113) for 60 days, the TTCA content decreased by only 13.5% (Fig. 2i). This exceptional stability implies lower storage losses, reduced packaging demands, and potentially extended shelf-life in food applications. Elevated moisture activity significantly accelerated the degradation and browning of both TTCA and ARP. Colorimetric analysis (L*, a*, b*, ΔE) showed that increased aw led to darker samples (decreased L*) and more pronounced overall color change (ΔE) (Table 2). Notably, the ΔE values for ARP were consistently higher than those for TTCA under all humidity conditions, further corroborating TTCA's superior stability. A semi-quantitative estimate indicates that TTCA's degradation rate in high humidity (aw 0.843) is approximately 60% slower than that of ARP (Fig. 2j).
| MRI | Indicator | a w | |||
|---|---|---|---|---|---|
| Control | 0.113 | 0.432 | 0.843 | ||
| TTCA | L* | 88.39 ± 0.03a | 81.36 ± 0.02b | 77.39 ± 0.04c | 72.34 ± 0.02d |
| a* | 5.19 ± 0.01d | 7.71 ± 0.03c | 9.19 ± 0.02b | 11.63 ± 0.04a | |
| b* | 11.13 ± 0.02d | 16.78 ± 0.04c | 22.36 ± 0.03b | 27.63 ± 0.01a | |
| ΔE | — | 9.36c | 16.22b | 23.90a | |
| ARP | L* | 87.68 ± 0.03a | 79.36 ± 0.03b | 62.18 ± 0.03c | 51.03 ± 0.03d |
| a* | 6.21 ± 0.03d | 11.39 ± 0.03c | 17.68 ± 0.03b | 21.39 ± 0.03a | |
| b* | 13.19 ± 0.03d | 20.38 ± 0.03c | 29.63 ± 0.03b | 37.68 ± 0.03a | |
| ΔE | — | 12.16c | 32.44b | 46.62a | |
In summary, TTCA possesses significantly greater environmental stability than ARP. Optimal storage conditions are low temperature, acidic-to-neutral pH, and low humidity, while high temperature, alkaline pH, and high moisture accelerate degradation. These results confirm TTCA's extended effective shelf life and underscore the necessity for cool, dry, and sealed storage for practical applications as a stable flavor precursor.
3.4 Antioxidant properties of MRIs demonstrate promising potential as alternatives to synthetic antioxidants
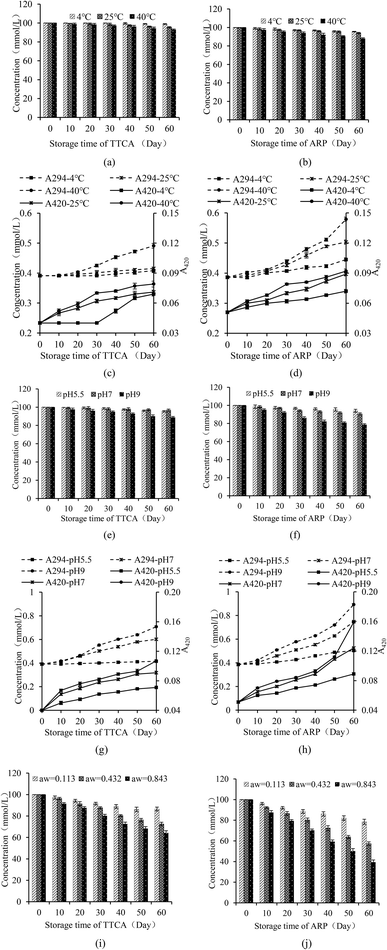
The antioxidant capacities of TTCA and ARP intermediates and complete Maillard reaction products (MRPs) were evaluated in this study. The chelating ability of Fe2+ helps to reduce the occurrence of lipid oxidation, and is an important indicator for evaluating antioxidants.7 A concentration-dependent Fe2+ chelating ability was observed for TTCA and ARPs (Fig. 3a). Fe2+ can coordinate with the carbonyl oxygen atom on the xylose segment of Amadori compounds, forming a five-membered chelating ring.7 Compared to the ARP molecule, the TTCA compound can provide stronger overall molecular polarity and more polar sites (including hydroxyl and carbonyl groups) in its possible tautomeric forms (Table S1).36 This enhanced polar characteristic allowed the TTCA molecule to interact more effectively with solvent water molecules in aqueous solutions through electrostatic interactions and dynamic hydrogen-bonding networks, thereby forming more stable solvated complexes.47 Therefore, TTCA compound molecules have more sites that can form complexes with Fe2+, and the complexes formed may be more stable than those formed by ARP, which likely directly leads to a higher Fe2+ chelating ability of TTCA (71.35%) than that of the ARP (51.03%) (Fig. 3a). Besides, MRPs always revealed a higher Fe2+ chelating ability than that of TTCA and ARP compounds. Some melanoidins in MRPs can be natural free radical scavengers in the food system.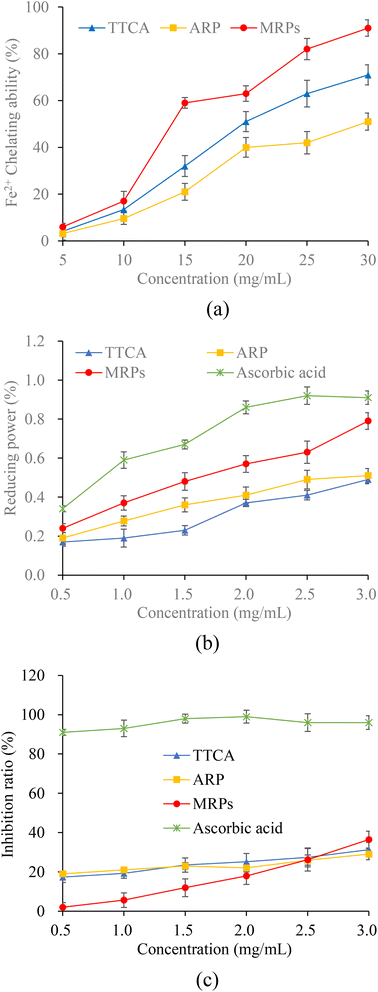 | ||
| Fig. 3 Fe2+ chelating activity (a) and reducing power (b) of TTCA, ARPs and MRPs and inhibition of TTCA, ARPs, MRPs and ascorbic acid on lipid peroxidation induced by iron (c). | ||
The reducing power, reflecting a substance's electron transfer ability, was measured to further evaluate the antioxidant activity.7 A positive correlation was observed between the concentration of MRIs (0.5–3 mg mL−1) and their reducing power (Fig. 3b). This can be attributed to reductive ketones and hydroxyl-containing compounds in the intermediates, which can donate electrons or hydrogen atoms to reduce the ferricyanide complex.23 Notably, ARPs exhibited stronger reducing properties than TTCA. This difference likely stems from the role of free thiol groups in ARPs, which possess strong electron-donating ability due to their S–H bonds. In contrast, the thiol groups in TTCA are involved in forming a stable five-membered ring, leaving no exposed free thiols available for reduction.48 Furthermore, MRPs exhibited a stronger reducing power than that of their intermediates at all tested concentrations, with a rapid increase as the concentration increased, which is consistent with the existing research results.47
The chelating action on transition metals and the reducing action can inhibit lipid oxidation. Within the concentrations of 0.5 to 2.5 mg mL−1, ARP/TTCA inhibited lipid peroxidation more than the MRPs, indicating ARP's better efficacy (Fig. 3c). As the concentration increased to 3.0 mg mL−1, the inhibition rate for lipid peroxidation of MRPs increased from 2.18% to 36.39%, exceeding that of ARP (29.18%) and TTCA (31.30%) at 3.0 mg mL−1 (Fig. 3c). The increased concentration of TTCA or ARP beyond a certain point did not lead to a corresponding significant improvement in the inhibition of lipid peroxidation. This observed plateau in efficacy could result from several factors, such as potential solubility limitations of TTCA and ARPs compared to MRPs (rich in aldehydes and ketones) in the emulsion system,27,30 or the attainment of a saturation point for their antioxidant action under these specific conditions. The results confirmed that TTCA or ARPs could be one reason for the antioxidant activity of the Maillard reaction during food processing and storage. However, at higher concentrations, the MRPs demonstrated superior performance compared to their precursors, suggesting that they possess better emulsion system compatibility and contain a greater number of compounds with strong antioxidant activity.
The DPPH free radical scavenging ability typically reflects the hydrogen-donating ability of antioxidants.49 The scavenging rates of TTCA and ARP increased with their concentration during the range of 0.5 to 3.0 mg mL−1 (Fig. 4a). Their effect was weaker than that of MRPs and considerably lower than that of ascorbic acid, which achieved nearly 100% scavenging across the same range. Similar concentration-dependent trends were observed for superoxide anion and hydroxyl radical scavenging (Fig. 4a and b). At a concentration of 3.0 mg mL−1, the superoxide anion radical scavenging activities of TTCA and ARP are functionally comparable to that of 0.5 mg mL−1 ascorbic acid, suggesting that approximately 6 g of TTCA could theoretically replace 1 g of ascorbic acid in antioxidant applications. MRPs consistently demonstrated a stronger radical scavenging ability than that of the intermediates, with activity rising sharply at higher concentrations (Fig. 4a–c), consistent with prior reports.47 This enhanced activity is attributed to the complex mixture of cross-linked polymers, aldehydes, ketones, and heterocyclic compounds formed in the later Maillard stages, which exert antioxidant effects through multiple pathways.27 In summary, while MRIs such as TTCA and ARP contribute to controlled flavor formation, the final MRPs exhibit superior antioxidant properties. Therefore, a combination of MRIs and MRPs holds potential as dual-function ingredients, serving as both natural flavor enhancers and antioxidants in food systems.
TTCA and ARP compounds, derived from xylose and cysteine, exhibit antioxidant properties distinct from a simple cysteine–xylose mixture, indicating that structural differences at the molecular level are responsible,50 while free sugars like pentoses, hexoses, and ketoses predominantly exist in a closed-ring form in solutions, with minimal reactive open-chain carbonyls.51 For MRIs, the conjugated electron cloud structure between the enol and aminium ions leads to an equilibrium of isomeric interconversion between their open-ring and closed-ring forms, with the open-chain conformation becoming a significant configuration in aqueous media.52 This provides abundant free carbonyl groups capable of donating electrons to neutralize free radicals.7 Based on this, a possible pathway for the intermediates to scavenge free radicals is proposed (Fig. 4d). TTCA and ARPs can donate hydrogen atoms to free radicals, stabilizing them and terminating chain reactions. The radical electron can localize on the carbon adjacent to the carbonyl group on the xylose residue, where resonance and intramolecular hydrogen bonding lead to a thermodynamically stable intermediate. Alternatively, the carbonyl group itself may be attacked, with the radical electron transferring to the oxygen atom, followed by stabilization via intramolecular hydrogen bonding.7,53,54 Research also found that the free radical scavenging ability of ARPs is consistent with its electron-donating or hydrogen-donating properties and specifically proposed that its metal chelating ability may also be related to this principle.39 Although this study indicates that TTCA and ARPs exhibited certain antioxidant activity, its long-term stability in complex food systems and the production costs at scale still require further evaluation. Therefore, MRIs can serve as natural flavor precursors to achieve controlled formation of processing flavors and provide partial antioxidant functions, suggesting a potential practical value.
4 Conclusion
This study demonstrates that the Xyl-Cys MRIs including TTCA and ARPs could act as dual-functional, sustainable platforms, integrating efficient flavor precursor properties with certain natural antioxidant activities. The flavor profile released from TTCA can be regulated from “meaty” to “roasted nutty” notes through simply modulating thermal processing parameters (pH and temperature). The addition of exogenous amino acids can further enrich the diversity of roasted flavor compounds such as pyrazines. This significantly enhances the processing adaptability of the flavor precursors of MRIs. Compared to MRPs, TTCA or the ARP exhibits superior stability for commercial applications, which can retain over 90% of its content after 60 days of storage under ambient, neutral, and dry conditions. This stability ensures consistent performance and reliability during the storage, transportation, and use of TTCA or ARP as an ingredient, significantly reducing the risks of raw material loss and flavor variability due to degradation. TTCA and ARP compounds revealed increased Fe2+ chelating ability, reducing power, and free radical scavenging ability with the increase in concentration, demonstrating certain antioxidant capabilities. By combining flavor enhancement with moderate oxidative stability contributed by MRIs, this approach provides a new direction for “clean-label” food development that may enable a partial reduction in reliance on synthetic antioxidants and flavor additives. In summary, this work supports the potential of TTCA or the ARP to act as a sustainable platform for natural flavoring and supporting antioxidant properties. It offers a feasible strategy for improving the food quality and stability and aligns with the pursuit of greener food processing practices.Author contributions
Di Kang and Songjin Zheng conceived the study and designed and supervised the research. Yun Zhai and Yuying Fu outlined the study, performed the literature search, and prepared the original draft and figures. Lin Jiang, Yuan Hu, Haifeng Wang and Teng Li assisted in preparing the figures and the draft. All the authors have read and approved the final version to be published.Conflicts of interest
The authors declare no competing financial interest. This article does not contain any study with human participants or animals performed by any of the authors.Data availability
All the data generated or analyzed during this study are included in the manuscript.Supplementary information (SI): data on the structural confirmation of TTCA, the degradation and flavor formation patterns under various thermal processing conditions (temperature, pH), as well as the dynamic changes of the key precursor (Cys). These data serve as robust supporting material for the conclusions drawn in the main text regarding the antioxidant activity, stability, and flavor contribution of TTCA and ARP. See DOI: https://doi.org/10.1039/d5fb00932d.
Acknowledgements
The research was supported by the Zhejiang Provincial Natural Science Foundation of China under Grant No. LQN25C200010, the Scientific Research Fund of Zhejiang Provincial Education Department under Grant No. Y202353299, and the Fundamental Research Funds for the Provincial Universities of Zhejiang under Grant No. QRK23003.References
- R. Bel-Rhlid, R. G. Berger and I. Blank, Bio-mediated generation of food flavors – Towards sustainable flavor production inspired by nature, Trends Food Sci. Technol., 2018, 78, 134–143 CrossRef CAS.
- G. P. Rizzi, Chemically Reducing Properties of Maillard Reaction Intermediates, 2019, pp. 35–40 Search PubMed.
- D. S. Mottram, Flavour formation in meat and meat products: a review, Food Chem., 1998, 62, 415–424 CrossRef CAS.
- Y. Zhai, H. Cui, K. Hayat, S. Hussain, M. U. Tahir, J. Yu, C. Jia, X. Zhang and C.-T. Ho, Interaction of Added l-Cysteine with 2-Threityl-thiazolidine-4-carboxylic Acid Derived from the Xylose–Cysteine System Affecting Its Maillard Browning, J. Agric. Food Chem., 2019, 67, 8632–8640 CrossRef CAS PubMed.
- F. Shahidi, L. J. Rubin and L. A. D'Souza, Meat flavor volatiles: a review of the composition, techniques of analysis, and sensory evaluation, Crit. Rev. Food Sci. Nutr., 1986, 24, 141–243 CrossRef CAS PubMed.
- Amadori Compounds of Cysteine and Their Role in the Development of Meat Flavor, https://www.researchgate.net/publication/289728874_Amadori_Compounds_of_Cysteine_and_Their_Role_in_the_Development_of_Meat_Flavor, accessed 4 December 2025.
- S. Benjakul, W. Visessanguan, V. Phongkanpai and M. Tanaka, Antioxidative activity of caramelisation products and their preventive effect on lipid oxidation in fish mince, Food Chem., 2005, 90, 231–239 CrossRef CAS.
- H. Cui, J. Yu, Y. Zhai, L. Feng, P. Chen, K. Hayat, Y. Xu, X. Zhang and C.-T. Ho, Formation and fate of Amadori rearrangement products in Maillard reaction, Trends Food Sci. Technol., 2021, 115, 391–408 CrossRef CAS.
- Y. Zhai, H. Cui, K. Hayat, S. Hussain, M. U. Tahir, S. Deng, Q. Zhang, X. Zhang and C.-T. Ho, Transformation between 2-Threityl-thiazolidine-4-carboxylic Acid and Xylose–Cysteine Amadori Rearrangement Product Regulated by pH Adjustment during High-Temperature Instantaneous Dehydration, J. Agric. Food Chem., 2020, 68, 10884–10892 CrossRef CAS PubMed.
- V. A. Yaylayan, Recent Advances in the Chemistry of Strecker Degradation and Amadori Rearrangement: Implications to Aroma and Color Formation, Food Sci. Technol. Res., 2003, 9, 1–6 CrossRef CAS.
- H. Zhang, H. Cui, X. Xia, S. Hussain, K. Hayat, X. Zhang and C.-T. Ho, Control Formation of Furans and Pyrazines Resulting from Dual Glycation Sites in Nα,Nε-Di(1-deoxy-d-xylulos-1-yl)lysine via Elevating Thermal Degradation Temperatures, J. Agric. Food Chem., 2024, 72, 25261–25274 CrossRef CAS PubMed.
- L. Feng, H. Cui, P. Chen, K. Hayat, X. Zhang and C.-T. Ho, Promoted Formation of Pyrazines and Sulfur-Containing Volatile Compounds through Interaction of Extra-Added Glutathione or Its Constituent Amino Acids and Secondary Products of Thermally Degraded N-(1-Deoxy-d-ribulos-1-yl)-Glutathione, J. Agric. Food Chem., 2022, 70, 9095–9105 CrossRef CAS PubMed.
- X. Xia, T. Zhou, H. Zhang, H. Cui, F. Zhang, K. Hayat, X. Zhang and C.-T. Ho, Simultaneously Enhanced Formation of Pyrazines and Furans during Thermal Degradation of the Glycyl-l-glutamine Amadori Compound by Selected Exogenous Amino Acids and Appropriate Elevated Temperatures, J. Agric. Food Chem., 2023, 71, 4346–4357 CrossRef CAS PubMed.
- Y. Zhai, H. Cui, Q. Zhang, K. Hayat, X. Wu, S. Deng, X. Zhang and C.-T. Ho, Degradation of 2-Threityl-Thiazolidine-4-Carboxylic Acid and Corresponding Browning Accelerated by Trapping Reaction between Extra-Added Xylose and Released Cysteine during Maillard Reaction, J. Agric. Food Chem., 2021, 69, 10648–10656 CrossRef CAS PubMed.
- Y. Zhai, H. Cui, K. Hayat, T. Li, X. Wu, Y. Fu, X. Zhang and C.-T. Ho, Regulated Formation of Inhibited Color and Enhanced Flavor Derived from Heated 2-Threityl-Thiazolidine-4-Carboxylic Acid with Additional Cysteine Targeting at Different Degradation Stages, J. Agric. Food Chem., 2023, 71, 14300–14311 CrossRef CAS.
- Y. Luo, S. Li and C.-T. Ho, Key Aspects of Amadori Rearrangement Products as Future Food Additives, Molecules, 2021, 26, 4314 CrossRef CAS.
- B. Kebede, T. Grauwet, T. Andargie, G. Sempiri, S. Palmers, M. Hendrickx and A. Van Loey, Kinetics of Strecker aldehyde formation during thermal and high pressure high temperature processing of carrot puree, Innovative Food Sci. Emerging Technol., 2017, 39, 88–93 CrossRef CAS.
- C. Kanzler, H. Schestkowa, P. T. Haase and L. W. Kroh, Formation of Reactive Intermediates, Color, and Antioxidant Activity in the Maillard Reaction of Maltose in Comparison to d-Glucose, J. Agric. Food Chem., 2017, 65, 8957–8965 CrossRef CAS PubMed.
- W. Wang, Y. Bao and Y. Chen, Characteristics and antioxidant activity of water-soluble Maillard reaction products from interactions in a whey protein isolate and sugars system, Food Chem., 2013, 139, 355–361 CrossRef CAS PubMed.
- L. N. Vhangani and J. Van Wyk, Antioxidant activity of Maillard reaction products (MRPs) derived from fructose–lysine and ribose–lysine model systems, Food Chem., 2013, 137, 92–98 CrossRef CAS PubMed.
- J. Limsuwanmanee, M. Chaijan, S. Manurakchinakorn, W. Panpipat, S. Klomklao and S. Benjakul, Antioxidant activity of Maillard reaction products derived from stingray (Himantura signifier) non-protein nitrogenous fraction and sugar model systems, LWT–Food Sci. Technol., 2014, 57, 718–724 CrossRef CAS.
- M.-N. Maillard, C. Billaud, Y.-N. Chow, C. Ordonaud and J. Nicolas, Free radical scavenging, inhibition of polyphenoloxidase activity and copper chelating properties of model Maillard systems, LWT–Food Sci. Technol., 2007, 40, 1434–1444 CrossRef CAS.
- I. G. Hwang, H. Y. Kim, K. S. Woo, J. Lee and H. S. Jeong, Biological activities of Maillard reaction products (MRPs) in a sugar–amino acid model system, Food Chem., 2011, 126, 221–227 CrossRef CAS.
- G. M. Williams, M. J. Iatropoulos and J. Whysner, Safety Assessment of Butylated Hydroxyanisole and Butylated Hydroxytoluene as Antioxidant Food Additives, Food Chem. Toxicol., 1999, 37, 1027–1038 CrossRef CAS PubMed.
- W.-K. Jung, P.-J. Park, C.-B. Ahn and J.-Y. Je, Preparation and antioxidant potential of maillard reaction products from (MRPs) chitooligomer, Food Chem., 2014, 145, 173–178 CrossRef CAS PubMed.
- H. Cui, K. Hayat, C. Jia, E. Duhoranimana, Q. Huang, X. Zhang and C.-T. Ho, Synergistic Effect of a Thermal Reaction and Vacuum Dehydration on Improving Xylose–Phenylalanine Conversion to N-(1-Deoxy-d-xylulos-1-yl)-phenylalanine during an Aqueous Maillard Reaction, J. Agric. Food Chem., 2018, 66, 10077–10085 CrossRef CAS PubMed.
- F. Gu, J. M. Kim, K. Hayat, S. Xia, B. Feng and X. Zhang, Characteristics and antioxidant activity of ultrafiltrated Maillard reaction products from a casein–glucose model system, Food Chem., 2009, 117, 48–54 CrossRef CAS.
- S. Wu, X. Dai, F. Shilong, M. Zhu, X. Shen, K. Zhang and S. Li, Antimicrobial and antioxidant capacity of glucosamine-zinc(II) complex via non-enzymatic browning reaction, Food Sci. Biotechnol., 2018, 27, 1–7 CrossRef CAS PubMed.
- T. Kocadağlı and V. Gökmen, Investigation of α-Dicarbonyl Compounds in Baby Foods by High-Performance Liquid Chromatography Coupled with Electrospray Ionization Mass Spectrometry, J. Agric. Food Chem., 2014, 62, 7714–7720 CrossRef.
- D. S. Mottram and J. S. Elmore, in Controlling Maillard Pathways To Generate Flavors, American Chemical Society, 2010, vol. 1042, pp. 143–155 Search PubMed.
- S. Deng, H. Cui, K. Hayat, Y. Zhai, Q. Zhang, X. Zhang and C.-T. Ho, Comparison of pyrazines formation in methionine/glucose and corresponding Amadori rearrangement product model, Food Chem., 2022, 382, 132500 CrossRef CAS PubMed.
- M. Friedman, Food Browning and Its Prevention: An Overview, J. Agric. Food Chem., 1996, 44, 631–653 CrossRef CAS.
- T. Zhou, M. Huang, H. Cui, P. Chen, K. Hayat, X. Zhang and C.-T. Ho, Exogenous Alanine Promoting the Reaction between Amadori Compound and Deoxyxylosone and Inhibiting the Formation of 2-Furfural during Thermal Treatment, J. Agric. Food Chem., 2024, 72, 5878–5886 CrossRef CAS.
- M. Liu, J. Yu, T. Zhou, H. Xu, K. Hayat, X. Zhang and C.-T. Ho, Formation Priority of Pyrazines and 2-Acetylthiazole Dependent on the Added Cysteine and Fragments of Deoxyosones during the Thermal Process of the Glycine–Ribose Amadori Compound, J. Agric. Food Chem., 2022, 70, 11643–11651 CrossRef CAS.
- H.-I. Hwang, T. G. Hartman and C.-T. Ho, Relative Reactivities of Amino Acids in Pyrazine Formation, J. Agric. Food Chem., 1995, 43, 179–184 CrossRef CAS.
- A.-N. Yu and A.-D. Zhang, The effect of pH on the formation of aroma compounds produced by heating a model system containing l-ascorbic acid with l-threonine/l-serine, Food Chem., 2010, 119, 214–219 CrossRef CAS.
- S. Deng, Y. Zhai, H. Cui, K. Hayat, X. Zhang and C.-T. Ho, Mechanism of Pyrazine Formation Intervened by Oxidized Methionines during Thermal Degradation of the Methionine–Glucose Amadori Compound, J. Agric. Food Chem., 2022, 70, 14457–14467 CrossRef CAS PubMed.
- T. Zhou, M. Huang, H. Cui, S. Hussain, K. Hayat, X. Zhang and C.-T. Ho, Disclosing the Nitrogen Sources via Isotope Labeling Technique and the Formation Mechanism of Pyrazine and Alkylpyrazines during the Heat Treatment of N-(1-Deoxy-d-xylulos-1-yl)-alanine and Exogenous Alanine, J. Agric. Food Chem., 2024, 72, 18630–18637 CrossRef CAS PubMed.
- H. Zhan, H. Cui, J. Yu, K. Hayat, X. Wu, X. Zhang and C.-T. Ho, Characteristic flavor formation of thermally processed N-(1-deoxy-α-d-ribulos-1-yl)-glycine: Decisive role of additional amino acids and promotional effect of glyoxal, Food Chem., 2022, 371, 131137 CrossRef CAS PubMed.
- A.-N. Yu and A.-D. Zhang, The effect of pH on the formation of aroma compounds produced by heating a model system containing l-ascorbic acid with l-threonine/l-serine, Food Chem., 2010, 119, 214–219 CrossRef CAS.
- X. Gao, Y. Duan, H. Song and P. Yang, Unraveling the mechanism of aroma formation in the maillard reaction of roasted Wuyi rock tea: Focus on furfural, tea pyrrole, and 1-ethyl-1H-Pyrrole, Food Biosci., 2025, 71, 107137 CrossRef CAS.
- R. Bel-Rhlid, R. G. Berger and I. Blank, Bio-mediated generation of food flavors – Towards sustainable flavor production inspired by nature, Trends Food Sci. Technol., 2018, 78, 134–143 CrossRef CAS.
- T. Zhou, M. Huang, H. Cui, P. Chen, K. Hayat, X. Zhang and C.-T. Ho, Exogenous Alanine Promoting the Reaction between Amadori Compound and Deoxyxylosone and Inhibiting the Formation of 2-Furfural during Thermal Treatment, J. Agric. Food Chem., 2024, 72, 5878–5886 CrossRef CAS PubMed.
- N. V. Harohally, S. M. Srinivas and S. Umesh, ZnCl2-mediated practical protocol for the synthesis of Amadori ketoses, Food Chem., 2014, 158, 340–344 CrossRef CAS.
- Amino-carbonyl reactions in food and biological systems : proceedings of the 3rd International Sympo, https://www.las.ac.cn/front/book/detail?id=7153bffee43b00b5bfe66b85b3efbe3a, accessed 4 December 2025 Search PubMed.
- X. Wang, C. Wei, D. Wei and C. Yang, Impact of Amadori rearrangement products of cysteine-xylose-glutamic acid on meat flavor development, Food Res. Int., 2026, 230, 118535 CrossRef CAS PubMed.
- D. A. Cortés Yáñez, M. Gagneten, G. E. Leiva and L. S. Malec, Antioxidant activity developed at the different stages of Maillard reaction with milk proteins, LWT–Food Sci. Technol., 2018, 89, 344–349 CrossRef.
- C. Kanzler, P. T. Haase, H. Schestkowa and L. W. Kroh, Antioxidant Properties of Heterocyclic Intermediates of the Maillard Reaction and Structurally Related Compounds, J. Agric. Food Chem., 2016, 64, 7829–7837 CrossRef CAS PubMed.
- J. A. Rufián-Henares and F. J. Morales, Functional properties of melanoidins: In vitro antioxidant, antimicrobial and antihypertensive activities, Food Res. Int., 2007, 40, 995–1002 CrossRef.
- B. Cämmerer, W. Jalyschko and L. W. Kroh, Intact carbohydrate structures as part of the melanoidin skeleton, J. Agric. Food Chem., 2002, 50, 2083–2087 CrossRef PubMed.
- D. M. T. Nguyen, J. P. Bartley and W. O. S. Doherty, Combined Fenton Oxidation and Chemical Coagulation for the Treatment of Melanoidin/Phenolic Acid Mixtures and Sugar Juice, Ind. Eng. Chem. Res., 2017, 56, 1385–1393 CrossRef CAS.
- H. Cui, C. Jia, K. Hayat, J. Yu, S. Deng, E. Karangwa, E. Duhoranimana, S. Xia and X. Zhang, Controlled formation of flavor compounds by preparation and application of Maillard reaction intermediate (MRI) derived from xylose and phenylalanine, RSC Adv., 2017, 7, 45442–45451 RSC.
- Y. Li, B. Jiang, T. Zhang, W. Mu and J. Liu, Antioxidant and free radical-scavenging activities of chickpea protein hydrolysate (CPH), Food Chem., 2008, 106, 444–450 CrossRef CAS.
- W. Wang, Y. Bao and Y. Chen, Characteristics and antioxidant activity of water-soluble Maillard reaction products from interactions in a whey protein isolate and sugars system, Food Chem., 2013, 139, 355–361 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |