 Open Access Article
Open Access ArticleImprovement of structural, physicochemical, functional, and pasting properties and mass balance analysis of porous starch obtained from Indonesian local rice through microwave-assisted enzymatic hydrolysis
Elok Pawening
Maharani
 ab,
Priyanto
Triwitono
ab,
Priyanto
Triwitono
 a,
Yudi
Pranoto
a,
Yudi
Pranoto
 a and
Djagal Wiseso
Marseno
a and
Djagal Wiseso
Marseno
 *a
*a
aDepartment of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Universitas Gadjah Mada, Jalan Flora 1, Bulaksumur, Yogyakarta, 55281, Indonesia. E-mail: djagal@ugm.ac.id
bDepartment of Agricultural Product Technology, Faculty of Halal Industry, Universitas Nahdlatul Ulama Yogyakarta, Jalan Ringroad Barat, Banyuraden, Yogyakarta, 55293, Indonesia
First published on 30th March 2026
Abstract
This study aims to examine the impact of microwave treatment, enzymatic hydrolysis, and microwave-assisted enzymatic hydrolysis on the characteristics of porous rice starch. Microwave irradiation was applied to rice starch at 171 W g−1 for 15 s. Enzymatic hydrolysis was carried out using 100 U per g α-amylase and 100 U per g amyloglucosidase at 60 °C for 8 h in rice starch and 0.1 M CH3COONa buffer at pH 5.2. The results showed that among the samples, porous rice starch acquired by microwave-assisted enzymatic hydrolysis (MES) showed an increase in pore structure, crystallinity, solubility, pasting temperature, and adsorption ability, with oil and methylene blue uptake increased by 124.5 ± 2.80–158.45 ± 5.40% and 35.72 ± 3.66–53.67 ± 1.77% (P < 0.05), respectively. Pasting properties analysis indicates a significant decline in pasting, breakdown, final, and setback viscosity. Based on the findings of crystallinity tests, all samples showed crystal type A. In accordance with mass balance calculations, the mass of the incoming or outgoing materials of MES is 31.19 g, and the yield of MES obtained is 60.39 ± 0.72%. This study established that compared with microwave or enzymatic treatment, the microwave-initiated enzymatic method has great potential and novelty in porous rice starch production.
Sustainable spotlightThis study encourages sustainable food innovation by exploring novel approaches in porous rice starch preparation. Microwave assisted enzymatic hydrolysis was evaluated as an efficient and rapid method with great potential for the preparation of porous starch. This approach reduces processing time and solvent, and enhances the structural, physicochemical, functional, and pasting properties of porous rice starch, making it a potential flavoring agent, absorbent, carrier for volatile compounds, and encapsulant for delicate components such as probiotics, vitamins, and oils in food products and packaging. This promotes the integration of natural bioactive compounds into sustainable food systems. |
1 Introduction
Porous materials are economical and safe adsorbent matrices that have been extensively used in the food, medicinal, and environmental sectors.1 This is due to their advantages, namely high adsorption capacity and slow release of active substances. Porous matrices applied in the food industry not only focus on high adsorption capacity and protection of bioactive components, but also require safe, biodegradable, and low-cost characteristics.2 Xiao et al.3 analogized the physical properties of porous starch (PS) to an irregular honeycomb, while Belingheri et al.4 described PS as an adsorbent sponge matrix, with numerous pores distributed on the surface and extending into the core.5PS is produced from various commodities such as rice,3 corn,6 arrowroot,7 and canna.8 Different starch sources will produce different characteristics and morphology of PS.9 One of the main producers of rice worldwide is Indonesia. The International Rice Research Institute estimates that Indonesia produced 70 million metric tons of milled rice.10 Rice starch has been widely used in noodles,11 encapsulants,12 and fat replacers.13 However, rice starch also has various drawbacks, such as the amylose fraction, which causes heterogeneity and increases retrogradation. Therefore, it requires being transformed into PS to improve its structural, physicochemical, absorption capacity, and pasting properties. Porous particles have provided significant benefits as carriers of tomato flavour,4 for the encapsulation of red palm oil,14 and curcumin,15 active packaging capable of slowly releasing tea polyphenols,16 and even effluent purification.17 These bioactive components are encapsulated in porous starch through absorption or binding mechanisms.18
PS can be derived physically, chemically, enzymatically, and through a combination of treatments. Chemical treatments are often less recommended for use in the food industry. Organic solvents in solvent exchange methods can reduce the stability and increase the toxicity of porous starch, as well as increase production costs.5 The most commonly used method is enzymatic treatment because it is safe, has a mild reaction, produces specific products, and produces minimal by-products. However, enzymatic treatment sometimes results in pore collapse and is time-consuming, so it needs to be combined with other methods. The enzyme alpha-amylase cleaves α1,4-glycosidic bonds and creates oligosaccharides, which will prevent additional hydrolysis.8 Amyloglucosidase is an exo-enzyme that cleaves α1,4 and α1,6 bonds from the non-reducing end of the starch chain and produces glucose.6
The physical approach in PS production can cause changes in its structure and morphology.19 Chen et al.1 stated that extrusion, ultrasonication, and microwave irradiation are physical techniques commonly used in PS production. To achieve optimal physicochemical and functional properties, PS development is carried out with a combination of physical and biological techniques. The H2O and oil absorbency of porous corn starch obtained through the freeze-thaw method and enzymatic hydrolysis increased by 99.89% and 44.83%, respectively.20 There is an enhancement of the structure and absorptive ability of PS obtained through the combination of enzymatic methods with extrusion,21 and the combination of high-speed shear with enzymatic methods.3
Various methods can be used to optimize the absorption performance of porous starch, but they usually require long time, high cost, and complex processes. Therefore, a simple and rapid method that can improve the performance is needed, and currently not much attention has been paid. Microwave-assisted double enzymatic hydrolysis is considered an efficient technique for developing PS with good physicochemical and structural properties.
Molecular manipulation and modification techniques can be enhanced by microwaves.22 Furthermore, its advantages include a fast, simple, and easy operation. Jiang et al.23 reported that double enzymatic hydrolysis, preceded by microwave heating, resulted in significant increases in water and oil absorption, solubility, crystallinity, and pore surface area. Microwaves provide attachment points for enzyme molecules, making it easier for enzymes to break down starch bonds during the hydrolysis process. Jiang et al.23 and Su et al.18 have reported the structural and physicochemical properties of PS using the microwave-assisted enzymatic hydrolysis, but previous studies have not investigated the yield, mass balance, color, amylose content, methylene blue adsorption capacity, and pasting properties of PS. Subsequently, this research not only provides information on the physicochemical, morphological, and functional characteristics, but also analyzes its production efficiency through mass balance and yield, and also indicates its stability against retro-gradation by examining the pasting properties of starch. This highlights exciting prospects for porous starch as a high-performance food additive, particularly as an encapsulant for food active substances or active food packaging. This study also used local Indonesian rice with an amylose content of 28.58%, which has not been previously utilized. Therefore, these seven aspects represent the novelty of this research.
This work aims to assess the impact of the microwave process, double enzymatic hydrolysis, and combination methods on the structural, physicochemical, absorbency performance, mass balance, yield, and pasting properties of PS as an academic reference for the application of PS in the food, pharmaceutical, or environmental industries.
2 Experimental section
2.1 Materials
The local rice was procured from Toko Tani Berkah Nandur, Surakarta, Central Java, Indonesia. The amylose amount of the rice starch extracted is 28.58%. α-Amylase (AA) from Bacillus 3700 U g−1 and amyloglucosidase (AMG) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 U g−1 were supplied by Beijing Solarbio, China. All chemicals (acetic acid, sodium acetate, NaOH, etc.) were of analytical standard.
000 U g−1 were supplied by Beijing Solarbio, China. All chemicals (acetic acid, sodium acetate, NaOH, etc.) were of analytical standard.
2.2 Starch extraction
Rice starch (RS) was prepared via distilled H2O extraction of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (w/v) method with slight modification.24 Rice was soaked with H2O of 1
2 (w/v) method with slight modification.24 Rice was soaked with H2O of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (x/v) for 24 h. The rice was cleansed, crushed in a blender for 1 min, placed in a 100-mesh sieve, and divided at 1410×g for 15 min. The pellet was evaporated in an oven (Memmert) overnight at 50 °C, pulverized, and placed using a 100-mesh sieve to acquire RS.
2 (x/v) for 24 h. The rice was cleansed, crushed in a blender for 1 min, placed in a 100-mesh sieve, and divided at 1410×g for 15 min. The pellet was evaporated in an oven (Memmert) overnight at 50 °C, pulverized, and placed using a 100-mesh sieve to acquire RS.
2.3 Microwave treatment
The starch was heated with microwaves derived from the approach of Maharani et al.25 with little adjustments. 5 g of the sample was blended with 20 mL of 0.1 M CH3COONa buffer at pH 5.2. The slurry was put in a microwave oven (Electrolux, Sweden, 220 V, 800 W, 2450 MHz) at 171 W g−1 for 15 s. The blend was evaporated in an oven (Memmert) at 50 °C for 20 h, then pulverized in size and sieved using a 100-mesh screen. The microwave treatment and native starch were named as MS and NS, respectively.2.4 Enzymatic hydrolysis technique
The enzymatic process was conducted using the formula of Witasari et al.8 with slight adjustments. RS (5 g) was incorporated in 20 mL of 0.1 M CH3COONa buffer at pH 5.2. The dispersion was shaken on 80 rpm at 60 °C for 10 min. AA 100 U g−1 and AMG (100 U g−1) were incorporated into the solution, and processed at 80 rpm for 8 h at 60 °C. To deactivate the enzymes, 6 mL of 96% ethanol was mixed immediately. The suspension was separated at 1410×g for 10 min. The pellet was evaporated at 50 °C for 20 h, pulverized, and filtered using a 100-mesh screen. The enzymatically hydrolyzed PS was named ES.2.5 Combined treatment of microwave and enzymatic hydrolysis
The microwave-assisted enzymatic procedure was adopted from Jiang et al. with adjustments.23 RS (5 g) was dispersed in 20 mL of 0.1 M CH3COONa buffer at pH 5.2, then processed in a microwave for 15 s at 171 W g−1. Then, to the sample was added AA 100 U g−1 and AMG 100 U g−1, and conditioned at 80 rpm for 8 h at 60 °C in a shaker. The enzymatic hydrolysis was ended immediately by incorporating 6 mL of 96% ethanol. The slurry was spun off at 1410×g for 10 min. The pellet was evaporated at 50 °C for 20 h, crushed, and screened via a 100-mesh sieve. The PS obtained was named MES.2.6 Morphological features
The structure of starches was measured via scanning electron microscopy (JEOL JSM 6510, Japan) at 5 kV following the procedure of Han et al.26 The sample was coated with Au and obtained at 5000× and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnifications.
000× magnifications.
2.7 Crystallinity assay
Using X-ray diffraction (Bruker AXS, USA), the crystalline feature of rice PS was identified following the Witasari et al.8 procedure. The diffraction area was processed at 5–80° at 25 mA and 40 kV.2.8 FTIR spectroscopy
FTIR (Thermo Scientific Nicolet iS10, USA) was utilized to evaluate the functional groups of starches. The instrument was equipped with a Deuterated Tri Glycine Sulfate (DTGS) detector and Smart Omni Transmission accessory, operated in the 4000–400 cm−1 range, with 50 scans for every sample, and a 4 cm−1 resolution.2.9 Amylose content (AC)
0.1 g of native and rice PS was incorporated into 1 mL of 96% ethanol and 9 mL of NaOH in a 50 mL erlenmeyer flask. The mixture was placed at 100 °C for 10 min, kept for cooling, and added to 100 mL of distilled water. 5 mL of the liquid was blended with 1 mL of 1 N CH3COOH and 2 mL of 0.2% iodine, then made up to a total volume of 100 mL with H2O. Absorbance was determined after 20 min with a UV-vis spectrophotometer (Genesys, Thermo Fisher Scientific, UK) at 590 nm. The curve standard used is y = 22.65x + 0.0008, R2 = 0.9999, where x is the concentration (mg mL−1) and y is the absorbance. The AC of the PS was measured by using formula (1):| AC = {[sample concentration × sample volume × dilution factor]/sample weight} × 100% | (1) |
2.10 Color analysis
Color testing of native and PS was carried out using a chromameter (Konica Minolta CR410, Japan). The chromameter was configured with the L* (lightness), a* (red-green), and b* (yellow-blue) systems, illuminant D65, observer angle of 2°, aperture size of 5.0 mm, and a closed cone. The chromameter was calibrated using a standard white tile before the analysis. Native and porous starch samples were placed in a plate, and then the L*, a*, and b* values were measured in triplicate.2.11 Swelling power and solubility
Swelling power (SP) or solubility (S) tests was performed via the approach by Han et al.26 with few alterations. 10 mL of H2O was blended with 5 g of sample. The solution was treated at 85 °C for 30 min. The dispersion was separated at 1410×g for 15 min. The supernatant was placed in an oven overnight to obtain S (formula (2)). The pellet was used to measure SP, using formula (2):| SP (%) = the pellet weight/(the sample weight × [1 − S]) | (2) |
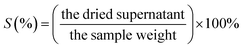 | (3) |
2.12 Mass balance and yield
Mass balance measurement refers to Vergara-Mendoza et al.27 with slight modifications. Mass balance and yield were measured for the PS produced by microwave treatment (MS), enzymatic hydrolysis (ES), and a combination of microwave treatment-assisted enzymatic hydrolysis (MES). For the MS, the mass balance process was carried out by weighing the input and output materials at each stage of microwave heating, drying, and sieving. For the ES, the mass balance test was measured by weighing the input and output materials at each stage of enzymatic hydrolysis, centrifugation, drying, and sieving. Meanwhile, for the MES, the test was conducted by weighing the input and output materials at each stage of microwave heating, enzymatic hydrolysis, centrifugation, drying, and sieving. As for yield evaluation, refer to Witasari et al.8 Yield is calculated by comparing the final product with the starting materials used, using formula (4).| Yield (%) = W1/W0 × 100% | (4) |
2.13 Absorptive capacity of water and palm oil
The absorbency ability was tested referring to the technique by Han et al.26 5 g of PS was incorporated into 8 mL of water or oil. Next the sample was left to stand for 30 min. The dispersion was separated at 1410×g for 15 min to obtain the sediment. The absorbency of water or oil can be quantified using formula (5): | (5) |
2.14 Methylene blue absorbency
The ability to absorb methylene blue (MB) was assessed using an approach defined by Witasari et al.8 0.025 g of rice starch was mixed with 11 mL of 0.01 mg per mL MB. The solution was subjected to stirring for 5 h, after which it was subjected to separation at 1410×g for 5 min. The measurement of the absorbance of the supernatant was conducted at 665 nm. The standard curve used is y = 190.8x − 0.0177, with R2 = 0.998, and y and x indicate absorbance and concentration (mg mL−1), respectively. The absorbency of MB was measured using formula (6):| MBAC (%) = ([C0 − C1]/C0) × 100% | (6) |
2.15 Pasting characteristics
Sample pasting characteristics were tested using a Rapid Visco Analyzer (RVA 4500, Perten Instruments). The starch was mixed with H2O. The moisture value of the samples was identified. The shaking speed was set at 960 and 160 rpm for 20 and 50 s, respectively. The sample was placed at 50–95 °C at a heating rate of 5.2°C min−1. The sample was conditioned at 95 °C for 5 min, then allowed to cool to 50 °C for 2 min.2.16 Experimental design and statistical analysis
At least three runs of the sample were conducted. The mean ± standard deviation was used to display the data. Analysis of variance (ANOVA) with Duncan or Kruskal–Wallis assessment support was implemented for analyzing the data using the SPSS program (version 25, IBM).3 Results and discussion
3.1 Structural properties
The morphology of starch is shown in Fig. 1. NS (Fig. 1a and b) exhibits a polygonal form with a soft appearance. Modified starch (Fig. 1c and d) exhibits micrographs of granules with few cracks and slight indentations. Microwaves produce few cracks and few uneven pores in the MS granules. The phenomenon of starch granules vibrating at high frequency in the presence of a microwave is attributable to the constant alteration of (+) and (−) polarities within the alternating electromagnetic field. Consequently, microwave energy is transformed into thermal energy within the starch. Thermal energy may accumulate inside granules as a result of a mechanism of loss of heat at their surface. At the same time, the water content in the starch granules can evaporate rapidly, creating high internal pressure within the granules. It is known that, under high pressure conditions, starch granules undergo a swelling and rupture process, which leads to the formation of pores.1ES exhibits polyhedral micrographs with more pores than MS (Fig. 1e and f). Variations in pore shapes, produced by enzymatic hydrolysis, include pinholes, sponge-like erosion, medium-sized pores, and internal and external corrosion.28 Initially, the enzyme hydrolyzes the starch in amorphous and irregular regions. Next, the enzyme hydrolyzes the non-reducing ends of the starch unit chains, which in turn causes the crystalline regions to hydrolyze. The starch becomes porous, forming a hollow internal structure.1
The enzyme α-amylase (AA) cleaves the α1,4-glucosidic bonds in the interior of the polysaccharide chain. AA will more easily reach the interior of the granule during starch swelling. The internal cleavage of the α-amylase enzyme will form new non-reducing ends that can be processed by saccharifying enzymes. The enzyme amyloglucosidase (AMG) cleaves the α1,4- and α1,6-glucosidic bonds of the non-reducing ends of the starch chain, creating sharp and deep pores.29 Therefore, it is proposed that the main explanation for the different surface pore patterns is the difference in the response patterns in the granules between these enzymes. In addition, the impacts of these two enzymes on PS can broaden its applications and offer an analytical basis for its preparation. Table 1 shows that the pore diameter of ES is larger than that of NS and MS. When compared with native arrowroot starch, arrowroot PS hydrolyzed by AA and AMG showed much larger pore sizes.7
| Sample | Pore diameter (µm) | Relative crystallinity (%) | 1047/1022 cm−1 | 1022/995 cm−1 |
|---|---|---|---|---|
| a Native starch (NS), microwave treatment starch (MS), enzymatically hydrolysed porous starch (ES), combined microwave and enzymatic hydrolysis treatment of porous starch (MES). | ||||
| NS | N/A | 10.2 | 1.41 | 0.38 |
| MS | N/A | 11.8 | 1.60 | 0.30 |
| ES | 0.606 | 12.8 | 1.56 | 0.29 |
| MES | 0.664 | 11.9 | 1.58 | 0.30 |
MES showed more pores, deeper pores, and denser pores than the other samples (Table 1). These pores were deeper and smaller than those in MS, and larger than those in ES (Fig. 1g and h). This indicates that MES has larger pores, a greater ability to hold the coated material more stably in the pores, and a high absorption capacity. Microwave irradiation can increase the rate of molecular alteration processes.22 Microwaves provide binding sites for enzyme molecules, thus facilitating enzyme access during enzymatic starch breakdown. As a result, enzymatic hydrolysis occurs more extensively, resulting in MES having a larger number of pores and denser pores.
3.2 Crystallinity determination
The amorphous and crystalline characteristics of starch were determined through XRD analysis. Fig. 2 shows clear diffraction peaks of NS at 2θ = 15.16°, 17.08°, 17.97°, and 23.12°, indicating an A-type diffraction pattern. The weakest peak at 19.98° indicates the interaction between amylose and lipids after the hydrothermal procedure.30 The modified starch presented a typical A-type XRD trend, implying that this treatment did not disrupt the crystallization of starch. Table 1 presents a slight increase in relative crystallinity (RC) for MS, ES, and MES compared to NS. MS showed a slight increase in crystallinity, which is comparable to the evaluation of Zhang et al.31 | ||
| Fig. 2 The XRD of native starch (NS), microwave-treated starch (MS), enzymatically hydrolysed porous starch (ES), combined microwave and enzymatic hydrolysis treatment of porous starch (MES). | ||
However, the results differ from those of Yang et al.32 who reported that waxy corn starch processed with microwave irradiation at 160 W g−1 for 5, 10, and 15 min experienced a decrease in crystallinity from 19.58% to 10.47%, 6.42%, and 2.91%, respectively. This phenomenon is likely due to the fact that, in this study, the application of microwave treatment at 171 W g−1 for 15 s probably did not produce sufficient alteration of the branched chain structure of starch. This difference in XRD data may be caused by variations in heating duration and starch moisture content.33
ES exhibits an increased proportion of crystalline area. This is because the amorphous area undergoes enzymatic hydrolysis, which can increase the percentage of crystalline area.34 The relatively higher crystallinity of MES compared to NS is likely due to the involvement of microwaves in facilitating conditions conducive to enzyme particles penetrating the granules.23 This is likely due to the thermal effect of microwaves causing particle expansion33 and generating forces that can damage the amorphous area.
The R1047/1022 value for PS presented in Table 1 indicates a more regular percentage of crystallinity compared to the amorphous area in NS.35 The higher the ratio value of 1047/1022, the higher the crystallinity and the higher the proportion of short-range ordered structures. The R1047/1022 values for ES and MES are greater than for NS, meaning the enzymatic process produces starch with high short-range order. This is related to the study by Zhang et al.,19 which found that the enzymatic process occurs in the amorphous zones of starch, leading to a reduction in the amorphous area and an increase in the short-range order of starch. The relative crystallinity pattern obtained is in accordance with the FTIR result.
3.3 FTIR spectroscopy
The functional groups of the starches were determined using FTIR. Fig. 3 presents the absorbance peaks in the range 3200–3400 cm−1 for starch, indicating the stretching of polymeric –OH groups.8 The absorbance at 2928 cm−1 detects the existence of methyl (–CH3) and methylene (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2) groups in the polysaccharide, which enhances the starch's lipophilicity.20 The absorbance at 1640 cm−1 is attributed to the bending vibration of the O–H bond in H2O trapped within the amorphous portions of starch.36 The peak intensity of NS at 3405.33 cm−1 wave number was 36.873, while the peak intensities of MS, ES, and MES were 30.13, 30.176, and 30.755, respectively. Meanwhile, the peak intensities of NS, MS, ES, and MES at 2928.46 cm−1 were 70.206, 67.701, 69, and 72.562, respectively. The decrease in intensity at the wave number of 3385 cm−1 indicates a reduction in hydrophilic groups, and the increase in the absorption peak at the wave number 2930 cm−1 indicates that the starch modified by microwave treatment followed by enzymatic hydrolysis is lipophilic, thus increasing the oil absorption capacity.
CH2) groups in the polysaccharide, which enhances the starch's lipophilicity.20 The absorbance at 1640 cm−1 is attributed to the bending vibration of the O–H bond in H2O trapped within the amorphous portions of starch.36 The peak intensity of NS at 3405.33 cm−1 wave number was 36.873, while the peak intensities of MS, ES, and MES were 30.13, 30.176, and 30.755, respectively. Meanwhile, the peak intensities of NS, MS, ES, and MES at 2928.46 cm−1 were 70.206, 67.701, 69, and 72.562, respectively. The decrease in intensity at the wave number of 3385 cm−1 indicates a reduction in hydrophilic groups, and the increase in the absorption peak at the wave number 2930 cm−1 indicates that the starch modified by microwave treatment followed by enzymatic hydrolysis is lipophilic, thus increasing the oil absorption capacity.
 | ||
| Fig. 3 The FTIR of native starch (NS), microwave-treated starch (MS), enzymatically hydrolysed porous starch (ES), and combined microwave and enzymatic hydrolysis treatment of porous starch (MES). | ||
The peak in the IR spectra's 800–1200 cm−1 area can be utilized to examine the crystalline composition and short-range organization of starch. Short-range order serves as an indicator of the atomic-scale chemical distributions inside a specific crystal system. This indicates a repeating pattern with minimal interatomic distance, frequently detected in amorphous areas.37 The ratio of 1047/1022 cm−1 (R1047/1022) signifies the crystallinity, while the diminished R1022/995 cm−1 denotes the enhanced order of the double-helical form of starches, as illustrated in Table 1.38
The absorbance R1047/1022 indicates the presence of a more ordered crystalline percentage compared to the amorphous areas in the starch.35 The 1047/1022 ratio values obtained in this study for PS increased in contrast to the NS, with values of 1.40, 1.60, 1.56, and 1.58 for NS, MS, ES, and MES, respectively. The better the R1047/1022 value, the higher the proportion of short-range ordered structures or the higher the crystallinity. The R1047/1022 values are presented in Table 1. The R1047/1022 value for ES is larger than NS, meaning the enzymatic process produces starch with high short-range order. This is related to the investigation of Zhang et al.19 which found that the enzymatic process occurs in the amorphous zones of starch, leading to a reduction in amorphous area and an improvement in the short-range order of the starch. Amorphous regions are easily hydrolyzed by the enzymes AA and AMG.26,28 In MES, the R1047/1022 value was 1.58 times higher than in ES. This is likely because microwaves can increase AA and AMG exposure to the amorphous areas of starch, thereby enhancing the degree of enzymatic hydrolysis. This is consistent with enzymatically hydrolyzed porous corn starch preceded by microwave treatment, which has a higher R1047/1022 value than NS.23 The obtained XRD values are in concordance with these results.
The 1022/995 cm−1 ratios for NS, MS, ES, and MES are 0.38, 0.30, 0.29, and 0.30, respectively. The implication of the microwave-aided enzymatic process on the double helix form within the starch is examined using the absorbance R1022/995 cm−1. A greater level of order among the particles can be determined by a smaller ratio. When compared to NS, the R1022/995 cm−1 for MS, ES, and MES declined, confirming that the double helix form of starch is not disrupted by either microwaves or enzymatic hydrolysis.
3.4 Physicochemical properties and mass balance
Microwave treatment, enzymatic hydrolysis, and their combination will result in different porous starch colors (Table 2). MES significantly reduced the lightness (L) and increased the redness (a) and yellowness (b) of PS. This is similar to what happened to porous arrowroot starch, which experienced a 4.8% decrease in brightness.7 The decrease in L value occurred in ES and MES because it was influenced by the color of the materials used, particularly the brown AA enzyme and the black AMG enzyme. The a and b values tended to improve after modification resulting in a redness and yellowish color of MS, ES, and MES. This is as stated by Wang et al.39 that the color of the packaging film made from polyvinyl alcohol and cinnamon essential oil-loaded microporous starch will be influenced by the color of the materials used.| Sample | L* | a* | b* | Amylose content (%) | Swelling power (g g−1) | Solubility (%) | Yield (%) |
|---|---|---|---|---|---|---|---|
| a Native starch (NS), microwave-treated starch (MS), enzymatically hydrolysed porous starch (ES), combined microwave and enzymatic hydrolysis treatment of porous starch (MES). Lightness (L*), red-green (a*), and yellow-blue (b*). Diverse letters in the column denote significant differences (P < 0.05). | |||||||
| NS | 97.06 ± 0.41b | 0.22 ± 0.07 a | 2.82 ± 0.26a | 28.58 ± 0.43b | 5.87 ± 0.15d | 1.01 ± 0.43a | N/A |
| MS | 96.91 ± 0.25b | 0.28 ± 0.05b | 3.94 ± 0.16b | 28.87 ± 1.32 ab | 4.99 ± 0.05a | 3.81 ± 0.32b | 84.87 ± 1.76c |
| ES | 94.02 ± 0.19a | 0.87 ± 0.04d | 4.48 ± 0.10c | 27.53 ± 1.04 ab | 5.81 ± 0.08c | 9.86 ± 0.97c | 68.83 ± 0.19b |
| MES | 94.04 ± 0.23a | 0.83 ± 0.05c | 4.44 ± 0.22c | 27.42 ± 0.50a | 5.70 ± 0.16b | 12.62 ± 2.78c | 60.39 ± 0.72a |
Microwave irradiation induces the transformation of starch from an ordered to a disordered structure; meanwhile, the thermal and non-thermal effects of microwaves have various impacts on starch morphology. The quick heating influence compacts the double helix configuration of amylopectin within the crystalline region, thereby distorting the amorphous region. Conversely, the non-thermal impact protects the amorphous region from disruption caused by rapid heating by inducing a change in the irregular lamellar shape.40 The amount of amylose did not differ significantly between MS and ES, compared to NS, as shown in Table 2. This is likely because the short time (15 s) did not cause damage to the amylopectin. This finding contradicts studies on waxy starch heated in microwaves for 3, 5, and 7 min, which showed an increase in amylose by 6.01% to 128.19%. Microwave-treated granules interact with H2 bonding forces in busil (Xanthosoma sagittifolium) starch granules, facilitating the degradation of amylopectin chains and producing linear amylose chains.41
Microwave-assisted enzymatic hydrolysis using AA and AMG reduced the amylose content in MES (Table 1). Amylose in arrowroot and canna starches also decreased due to enzymatic hydrolysis using AA and AMG.7,8 This decrease occurred because AA and AMG tend to hydrolyze the amorphous part of starch, which lowers amylose levels.42 Jiang et al.23 reported that microwaves play a role in providing attachment points that enzymes later use to hydrolyze starch. This creates a synergy between microwaves and enzymatic hydrolysis, resulting in a decrease in amylose.
MS, ES, and MES showed a significant decrease in swelling power (SP). Microwave heating damages amylopectin, leading to a decrease in its SP.23 The type of enzyme significantly influences the SP of starch. AMG and AA produced the highest and lowest SP, respectively. AMG induces the rearrangement of starch chains, thereby increasing structural complexity; as a result, the granular structure of starch becomes stronger, and its water-binding capacity increases.38 Meanwhile, SEM studies generally confirm that the development of hollow structures after enzymatic modification is associated with a decrease in SP. These structures weaken the starch granule structure. Alternatively, a surface with increased hydrophobicity was created on the inner walls of the needle holes, which minimized water binding43 caused by AA breaking α-(1,4) bonds, resulting in dextrins, which have a lower SP during gelatinization.44
Solubility (S) is a parameter that reflects how effectively starch dissolves in water or other solvents, and is closely related to the structure of PS. High solubility indicates increased pore formation.20 The power of starch suspensions to absorb H2O during gelatinization and retain H2O during centrifugation is measured by swelling power (SP). The binding process between starch and H2O can be determined through S and SP.18 Compared with NS, solubility showed a significant improvement in MS, ES, and MES (Table 2). This finding is similar to that of adlay starch treated with microwave heating for 3 and 6 min, which showed increases of 28.94% and 45.59%, respectively.45
The heating process can increase the solubility of MS. The H2 bonds in the branched chains of amylose and amylopectin rupture, making it simpler for the H2 bonds of H2O to combine with the –OH groups of amylose, thereby increasing the solubility of MS.46,47 High temperatures cause de-polymerization of starch molecules, converting amylose into simpler, more water-soluble linear chains.45
The high solubility in ES and MES is due to enzymatic hydrolysis, which induces the appearance of larger and deeper pores. The porous morphology provides sufficient access for H2O molecules to release solutes from the starch granules, thus increasing the solubility of ES and MES. MES has the highest solubility, confirming that microwave treatment followed by enzymatic hydrolysis results in more PS structure. This is similar to porous corn starch treated with microwaves and enzymatic hydrolysis, resulting in an 80.84% increase in solubility.23 In ES, there is a significant increase in solubility because AM carries out endo- and exo-corrosion processes that create tiny starch components from the soluble starch.34 AM damages amylopectin and induces glucose outside the molecule, causing starch chain rearrangement.
The amount of PS produced from microwave treatment, enzymatic hydrolysis, and their combination is shown through percentage yield (Table 2). The yields obtained at MS, ES, and MES are 84.87 ± 1.76%, 68.83 ± 0.19%, and 60.39 ± 0.72%, respectively (Table 2). ES and MES showed lower yields compared to MS. This is because AA and GA enzymes attack the amorphous areas,42 thus reducing amylose and yield. At high concentrations, AA can partially attack both amorphous and crystalline areas, leading to the formation of additional pores and making the granules more easily damaged.8 MES has the lowest yield due to the preliminary treatment with microwaves, which provides attachment points that facilitate enzyme molecules in hydrolyzing starch,22,23 thus further reducing the yield. MS has the highest yield compared to ES and MES because there is no enzyme to hydrolyze the starch.
Mass balance is analyzed by determining the incoming and outgoing materials flow in the system.27 Therefore, the mass entering the system must equal the mass exiting the system. Fig. 4 shows the mass balance of PS production according to (a) microwave treatment (MS), (b) enzymatic hydrolysis (ES), and (c) microwave followed by enzymatic hydrolysis (MES). In MS, the process stages are microwave heating, drying, grinding, and sieving. In the microwave process, the input used is 25 g, consisting of 5 g of rice starch and 20 mL of acetate buffer, while the mass of product obtained is 21.49 g. The output lost is gelatinized starch. Next, drying was carried out, resulting in 4.45 g of PS. The subsequent processes were grinding and sieving. After sieving, the final product of MS was 4.24 g. The amount of input or output materials was the same at 25 g.
 | ||
| Fig. 4 Mass balance of (a) microwave-treated starch (MS), (b) enzymatically hydrolysed porous starch (ES), and (c) combined microwave and enzymatic hydrolysis treatment of porous starch (MES). | ||
The preparation of ES begins with enzymatic hydrolysis, centrifugation, drying, grinding, and sieving of starch. Enzymatic hydrolysis used an input of 31.29 g, consisting of 5 g rice starch, 20 mL acetate buffer, 0.1351 g AA, 0.05 g AMG, and 6 mL ethanol used to stop the enzymatic hydrolysis. The AA and AMG break down starch molecules into glucose, with the glucose being included in the supernatant. The stages of this process are almost the same as the research on bioethanol production from petai peel (Parkia speciosa) using α-amylase and glucoamylase.48 The 7.30 g of precipitate was then dried, yielding 3.65 g with a moisture loss during drying of 3.65 g. Grinding and sieving were performed, resulting in a ES of 3.44 g. The total input and output of materials were both 31.29 g.
The production of MES was carried out similar to the preparation of ES, but it begins with microwave heating first. 5 g of rice starch and 20 mL of acetate buffer were subjected to microwave treatment, resulting in a yield of 22.4 g and gelatinized starch content of 2.56 g. The gelatinized starch produced at MES was less than that produced during the MS stage due to the difference in containers used. In MS, Petri dishes are used during the microwave process, so the heat received can directly reach the material, and the process of collecting gelatinized starch is also easier and more optimal. In MES, during the microwave process, the sample is placed in an Erlenmeyer flask to facilitate the next step, enzymatic hydrolysis. However, using an Erlenmeyer flask may result in less microwave heat being received and/or make it more difficult to collect gelatinized starch compared to using Petri dishes. Next, enzymatic hydrolysis and centrifugation were performed, resulting in a precipitate of 6.74 g and a supernatant of 21.89 g. The precipitate produced at this stage was less than that obtained in the production of ES because the weight of the starch had decreased due to the microwave heating. Then drying was carried out, resulting in a product of 3.19 g and evaporated water content of 3.55 g, which is almost the same as the drying water content at the ES stage. In the final stage, grinding and sieving were performed, yielding 3.02 g of MES and 0.17 g of sieving residue. The total amount of incoming or outgoing materials is 31.19 g. From these methods of making PS, the final products MS, ES, and MES were obtained in amounts of 4.24 g, 3.44 g, and 3.02 g, respectively, and their percentage yields are shown in Table 2.
3.5 Adsorption capacity
Microwave, enzymatic, and combination techniques showed an improvement in oil absorption capacity (OAC) and methylene blue absorption capacity (MBAC) and decline in water absorption capacity (WAC) in MS, ES, and MES (Fig. 5). The OAC values for NS, MS, ES, and MES are 124.50 ± 2.80%, 147.41 ± 5.30%, 155.15 ± 3.35%, and 158.45 ± 5.40%, respectively. Meanwhile, the MBAC values for NS, MS, ES, and MES are 35.72 ± 3.66%, 36.82 ± 1.61%, 53.24 ± 1.56%, and 53.67 ± 1.77%, respectively. These findings are identical to the study of Witasari et al.8 on porous edible canna starch resulting from enzymatic hydrolysis of AA and AMG with a ratio of 150![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 75, which showed higher levels in OAC and MBAC, and a decline in WAC. OAC increased due to an increase in methylene groups (–CH2) in FTIR, indicating that porous starch is lipophilic.20,49 AA and AMG enzymes facilitate oil penetration into the deep and wide pores of the starch. Hydrolysis using AA and AMG on wheat and corn starch resulted in an increase in OAC of 90.67% and 90.15%, respectively.50 Porous corn starch produced by microwave-initiated enzymatic hydrolysis also experienced an increase.23 The WAC values for NS, MS, ES, and MES are 96.92 ± 2.56%, 91.49 ± 2.53%, 89.94 ± 3.34%, and 85.85 ± 1.77%, respectively. The WAC decreased due to the reduction of hydroxyl groups in PS, as shown in the FTIR results. The hydrophilic properties of PS are affected by the type and concentration of the enzyme, while its hydrophobic properties are influenced by the type of enzyme.51
75, which showed higher levels in OAC and MBAC, and a decline in WAC. OAC increased due to an increase in methylene groups (–CH2) in FTIR, indicating that porous starch is lipophilic.20,49 AA and AMG enzymes facilitate oil penetration into the deep and wide pores of the starch. Hydrolysis using AA and AMG on wheat and corn starch resulted in an increase in OAC of 90.67% and 90.15%, respectively.50 Porous corn starch produced by microwave-initiated enzymatic hydrolysis also experienced an increase.23 The WAC values for NS, MS, ES, and MES are 96.92 ± 2.56%, 91.49 ± 2.53%, 89.94 ± 3.34%, and 85.85 ± 1.77%, respectively. The WAC decreased due to the reduction of hydroxyl groups in PS, as shown in the FTIR results. The hydrophilic properties of PS are affected by the type and concentration of the enzyme, while its hydrophobic properties are influenced by the type of enzyme.51
The methylene blue adsorption capacity of MS, ES, and MES is higher than that of NS. The pores formed by double enzymatic hydrolysis with the aid of microwave heating support the adsorption of methylene blue molecules. This is due to the increased surface area of the pores formed. Several studies have shown increased methylene blue adsorption by porous arrowroot starch7 and glutinous rice28 which are produced by enzymatic hydrolysis using AA and AMG. These results are consistent with SEM images showing larger and deeper pores in PS, thus supporting oil adsorption and MB capacity.
3.6 Pasting properties
These pasting properties reflect the gel viscosity, stability, and retro-gradation of starch, as shown in Table 3. The PSs with pretreatment(s) had substantially higher Pasting Temperature (PT) than the NS, following the order of NS < MS < MES < ES (Table 3). The higher PT implies that higher heat is necessary for structural disintegration and paste formation because treatment increases the strength of intramolecular bonds, thus shifting the initiation of pasting in PS to a higher temperature.32| Sample | PT (°C) | PV (cP) | BV (cP) | FV (cP) | SV (cP) |
|---|---|---|---|---|---|
| a Native starch (NS), microwave-treated starch (MS), enzymatically hydrolysed porous starch (ES), combined microwave and enzymatic hydrolysis treatment of porous starch (MES). Pasting temperature (PT), pasting viscosity (PV), breakdown viscosity (BV), final viscosity (FV), and setback viscosity (SV). Diverse letters in the column denote significant differences (P < 0.05). | |||||
| NS | 79.27 ± 0.21a | 4557.33 ± 66.61c | 3228.33 ± 105.45d | 2779.33 ± 9.87c | 1450.33 ± 34.03b |
| MS | 81.92 ± 0.35b | 3822.33 ± 165.18b | 2175.67 ± 134.81c | 3303.67 ± 83.94d | 1657 ± 54.03c |
| ES | 82.58 ± 0.20d | 2274.33 ± 63.96a | 1954.67 ± 45.18b | 522.33 ± 36.30b | 202.67 ± 10.69a |
| MES | 82.42 ± 0.33c | 2077.67 ± 90.36a | 1852.67 ± 45.72a | 379.67 ± 71.68a | 154.67 ± 27.79a |
MS, ES, and MES showed an improvement in PT and a decline in Pasting Viscosity (PV) and Breakdown Viscosity (BV). These results are similar to those observed in waxy corn starch, which denoted increased resistance to swelling and rupture.22 The PS had substantially lower PV than the NS, following the order of NS > MS > ES and MES (Table 3). The decrease in PV indicates that microwaves increase intermolecular and intramolecular hydrogen bonding, a consequence of starch chain association.32 Starch treated with microwaves, enzymatic hydrolysis, and a combination showed a decrease in PV compared to NS. The lower the PV, the greater the number of voids in the starch.52 This is consistent with the SEM results of PS. Enzymatic hydrolysis breaks bonds, reduces the molecular weight of the starch fraction, and reduces swelling during gelatinization. This is similar to the investigation carried out by Davoudi et al.6 The low PV value facilitates the application of PS in liquid food products.53 The reduction in PV is linked with hydrolysis in non-crystalline areas and the synthesis of low-molecular-weight dextrins,54 which minimizes the expansion force.55
The PS had substantially lower BV than the NS, following the order of NS > MS > ES > MES (Table 3). The decrease in BV in MS indicates that MS is less resistant to shear effects during heating.32 The starch treated with microwaves for 3 min also showed a decrease in BV.45 The enzymatic process lowered the BV of PS, which showed higher stability during shearing and heating. Porous corn starch processed with cyclodextrin glycosyltransferase also showed a decrease in BV.43 MES showed the lowest BV, indicating that MES has excellent prospects as a food ingredient when heated and stirred mechanically. Low BV is also associated with low PV and low swelling capacity.56 This is consistent with the results of the swelling power test on PS being lower than those of NS.
A decrease in Final Viscosity (FV) in ES and MES has been shown to correlate with amylose loss during the pasting phase.57 This aligns with the result of amylose value and XRD identified in ES and MES. The Setback Viscosity (SV) describes the starch molecules, particularly amylose, undergoing re-association during cooling and forming a gel structure. The propensity of amylose chains to retrograde and recrystallize is reflected in the SV.6 ES and MES have the lowest SV compared to NS and MS, making them suitable for preventing staling in bread products.
4 Conclusions
Microwave treatment assisted enzymatic hydrolysis, compared to other treatments, can improve pore structure and crystallinity, significantly increase solubility, oil and methylene blue absorption capacity, and pasting temperature, while reducing the pasting, breakdown, final, and setback viscosity. These findings confirm that porous rice starch could be potentially applied as an encapsulant or carrier for active substances in food products or packaging. Based on mass balance calculations, the mass of the incoming or outgoing materials of MES is 31.19 g, and the yield in the preparation of MES is 60.39 ± 0.72%. Next, research is necessary to measure the thermal characteristic, apply PS to food products or packaging, and evaluate the influence of adding PS on the overall stability and quality of the products.Author contributions
Elok Pawening Maharani: investigation, writing–original draft, conceptualization, data visualization, formal analysis; Priyanto Triwitono: supervision, methodology, validation; Yudi Pranoto: writing–review & editing, supervision, conceptualization; Djagal Wiseso Marseno: writing–review & editing, methodology, supervision, conceptualization, data curation, validation.Conflicts of interest
There are no conflicts of interest to declare.Data availability
All data produced or examined in this work are contained within this published publication and comply with research standards.Acknowledgements
The research was funded by the Indonesian Education Scholarship (BPI), the Center for Higher Education Funding and Assessment from the Ministry of Higher Education, Science, and Technology of Republic Indonesia, and the Indonesian Endowment Fund for Education (LPDP).References
- J. Chen, Y. Wang, J. Liu and X. Xu, Preparation, characterization, Physicochemical property and potential application of porous starch: a review, Int. J. Biol. Macromol., 2020, 148, 1169–1181, DOI:10.1016/j.ijbiomac.2020.02.055.
- D. J. McClements, Nanotechnology approaches for improving the healthiness and sustainability of the modern food supply, ACS Omega, 2020, 5, 29623–29630 CrossRef CAS PubMed.
- W. Xiao, H. He, Q. Dong, Q. Huang, F. An and H. Song, Effects of high-speed shear and double-enzymatic hydrolysis on the structural and physicochemical properties of rice porous starch, Int. J. Biol. Macromol., 2023, 234, 123692, DOI:10.1016/j.ijbiomac.2023.123692.
- C. Belingheri, A. Ferrillo and E. Vittadini, Porous starch for flavor delivery in a tomato-based food application, LWT–Food Sci. Technol., 2015, 60, 593–597, DOI:10.1016/j.lwt.2014.09.047.
- S. Jha, S. Sarkhel, S. Saha, B. Sahoo, A. Kumari, K. Chatterjee, P. M. Mazumder, G. Sarkhel, A. Mohan and A. Roy, Expanded porous-starch matrix as an alternative to porous starch granule: Present status, challenges, and future prospects, Food Res. Int., 2024, 175, 113771, DOI:10.1016/j.foodres.2023.113771.
- Z. Davoudi, M. H. Azizi and M. Barzegar, Porous corn starch obtained from combined cold plasma and enzymatic hydrolysis: Microstructure and physicochemical properties, Int. J. Biol. Macromol., 2022, 223, 790–797, DOI:10.1016/j.ijbiomac.2022.11.058.
- L. D. Witasari, S. Nisrina, A. I. T. Yani, A. A. Heryadi and Y. Pranoto, Characterization of porous starch produced from arrowroot (Maranta arundinacea L.) by enzymatic hydrolysis with α-amylase and glucoamylase, Carbohydr. Polym. Technol. Appl., 2024, 7, 100445, DOI:10.1016/j.carpta.2024.100445.
- L. D. Witasari, A. A. Heryadi, A. I. T. Yani, S. Nisrina, L. Purwitasari and Y. Pranoto, Characterization of porous starch produced from edible canna (Canna edulis Kerr.) via enzymatic hydrolysis using thermostable α-amylase and glucoamylase, Biocatal. Agric. Biotechnol., 2024, 55, 102990, DOI:10.1016/j.bcab.2023.102990.
- D. N. H. Latip, H. Samsudin, U. Utra and A. Karim, Modification methods toward the production of porous starch: a review, Crit. Rev. Food Sci. Nutr., 2021, 61, 2841–2862, DOI:10.1080/10408398.2020.1789064.
- J. G. Mogoginta, T. Murai and G. A. Annor, Profiles of Indonesian rice (Oryza sativa), Foods, 2024, 13, 1–14 CrossRef PubMed.
- C. Yi, H. Zhu, Y. Zhang, S. Wu and J. Bao, The role of indica starch in the mechanism of formation of fresh rice noodles, J. Cereal Sci., 2021, 99, 103212, DOI:10.1016/j.jcs.2021.103212.
- B. A. Ashwar, A. Gani, A. Gani A, A. Shah and F. A. Masoodi, Production of RS4 from rice starch and its utilization as an encapsulating agent for targeted delivery of probiotics, Food Chem., 2018, 239, 287–294, DOI:10.1016/j.foodchem.2017.06.110.
- F. Bagheri, M. Radi and S. Amiri, Evaluating the function of cross-linked rice starch as a fat replacer in low fat cream, Int. J. Dairy Technol., 2018, 71, 981–991 CrossRef CAS.
- B. S. Putra, S. Raharjo and L. D. Witasari, Encapsulation of red palm oil using porous starch from Canna edulis Kerr, Food Chem. Adv., 2025, 7, 100989 CrossRef.
- X. Han, P. Ma, M. Shen, H. Wen and J. Xie, Modified porous starches loading curcumin and improving the free radical scavenging ability and release properties of curcumin, Food Res. Int., 2023, 168, 112770, DOI:10.1016/j.foodres.2023.112770.
- Z. Miao, R. Lv, S. Teng, C. Cao and P. Lu, Development of antioxidant active packaging films with slow release properties incorporated with tea polyphenols-loaded porous starch microcapsules, Int. J. Biol. Macromol., 2022, 222, 403–412, DOI:10.1016/j.ijbiomac.2022.09.143.
- L. Bao, X. Zhu, H. Dai, Y. Tao, X. Zhou, W. Liu and Y. Kong, Synthesis of porous starch xerogels modified with mercaptosuccinic acid to remove hazardous gardenia yellow, Int. J. Biol. Macromol., 2016, 89, 389–395, DOI:10.1016/j.ijbiomac.2016.05.003.
- (a) X. Su, Y. Ji, S. Bai, Q. Xu, S. Xu, Z. Xu and N. Zhang, Structural and physicochemical properties of porous starch effected by different microwave involved stages under enzymatic hydrolysis, Int. J. Biol. Macromol., 2025, 294, 139317, DOI:10.1016/j.ijbiomac.2024.139317; (b) X. Han, P. Ma, M. Shen, H. Wen and J. Xie, Modified porous starches loading curcumin and improving the free radical scavenging ability and release properties of curcumin, Food Res. Int., 2023, 168, 112770, DOI:10.1016/j.foodres.2023.112770.
- B. Zhang, D. Cui, M. Liu, H. Gong, Y. Huang and F. Han, Corn porous starch: Preparation, characterization and adsorption property, Int. J. Biol. Macromol., 2012, 50, 250–256, DOI:10.1016/j.ijbiomac.2011.11.002.
- A. Q. Zhao, L. Yu, M. Yang, C. J. Wang, M. M. Wang and X. Bai, Effects of the combination of freeze-thawing and enzymatic hydrolysis on the microstructure and physicochemical properties of porous corn starch, Food Hydrocolloids, 2018, 83, 465–472, DOI:10.1016/j.foodhyd.2018.04.041.
- W. Wu, A. Jiao, E. Xu, Y. Chen and Z. Jin, Effects of Extrusion Technology Combined with Enzymatic Hydrolysis on the Structural and Physicochemical Properties of Porous Corn Starch, Food Bioprocess Technol., 2020, 13, 442–451 CrossRef CAS.
- M. Brasoveanu and M. R. Nemtanu, Behaviour of starch exposed to microwave radiation treatment, Starch/Staerke, 2013, 1–12 Search PubMed.
- K. Jiang, W. Wang, Q. Ma, J. Wang and J. Sun, Microwave-assisted enzymatic hydrolysis as a novel efficient way to prepare porous starch, Carbohydr. Polym., 2023, 301, 120306, DOI:10.1016/j.carbpol.2022.120306.
- D. K. Verma and P. P. Srivastav, Isolation, modification, and characterization of rice starch with emphasis on functional properties and industrial application: a review, Crit. Rev. Food Sci. Nutr., 2022, 62, 6577–6604, DOI:10.1080/10408398.2021.1903383.
- E. P. Maharani, P. Triwitono, Y. Pranoto and D. W. Marseno, Optimization of Microwave Treatment to Improve Adsorption Properties of Porous Rice Starch, Jurnal Teknik Pertanian Lampung, 2025, 14(5), 1553–1562, DOI:10.23960/jtepl.v14i5.1553-1562.
- X. Han, H. Wen, Y. Luo, J. Yang, W. Xiao, X. Ji and J. Xie, Effects of α-amylase and glucoamylase on the characterization and function of maize porous starches, Food Hydrocolloids, 2021, 116, 106661, DOI:10.1016/j.foodhyd.2021.106661.
- M. Vergara-Mendoza, G. R. Martinez, C. Blanco-Tirado and M. Y. Combariza, Mass Balance and Compositional Analysis of Biomass Outputs from Cacao Fruits, Molecules, Molecules, 2022, 27, 3717, DOI:10.3390/molecules27123717.
- L. D. Lacerda, D. C. Leite, R. M. D. Soares and N. P. da Silveira, Effects of α-Amylase, Amyloglucosidase, and Their Mixture on Hierarchical Porosity of Rice Starch, Starch, 2018, 70, 1–23, DOI:10.1002/star.201800008.
- M. Sujka and J. Jamroz, Starch granule porosity and its changes by means of amylolysis, Int. Agrophys., 2007, 21, 107–113 Search PubMed.
- S. O'Brien and Y. J. Wang, Susceptibility of annealed starches to hydrolysis by α-amylase and glucoamylase, Carbohydr. Polym., 2008, 72, 597–607 CrossRef.
- J. Zhang, Z. W. Wang and X. M. Shi, Effect of microwave heat/moisture treatment on physicochemical properties of Canna edulis Ker starch, J. Sci. Food Agric., 2009, 89, 653–664 Search PubMed.
- Q. Yang, L. Qi, Z. Luo, X. Kong, Z. Xiao, P. Wang and X. Peng, Effect of microwave irradiation on internal molecular structure and physical properties of waxy maize starch, Food Hydrocolloids, 2017, 69, 473–482, DOI:10.1016/j.foodhyd.2017.03.011.
- S. A. Oyeyinka, O. A. Akintayo, O. A. Adebo, E. Kayitesi and P. B. Njobeh, A review on the physicochemical properties of starches modified by microwave alone and in combination with other methods, Int. J. Biol. Macromol., 2021, 176, 87–95, DOI:10.1016/j.ijbiomac.2021.02.066.
- T. Keeratiburana, A. R. Hansen, S. Soontaranon, A. Blennow and S. Tongta, Porous high amylose rice starch modified by amyloglucosidase and maltogenic α-amylase, Carbohydr. Polym., 2020, 230, 115611, DOI:10.1016/j.carbpol.2019.115611.
- S. Garcia-Diaz, C. Hernandez-Jaimes, H. B. Escalona-Buendia, L. A. Bello-Perez, E. J. Vernon-Carter and J. Alvarez-Ramirez, Effects of CaCO3 treatment on the morphology, crystallinity, rheology and hydrolysis of gelatinized maize starch dispersions, Food Chem., 2016, 207, 139–147, DOI:10.1016/j.foodchem.2016.03.095.
- S. Wang, J. Wang, W. Zhang, C. Li, J. Yu and S. Wang, Molecular order and functional properties of starches from three waxy wheat varieties grown in China, Food Chem., 2015, 181, 43–50 CrossRef CAS PubMed.
- A. V. Ceguerra, M. P. Moody, R. C. Powles, T. C. Petersen, R. K. W. Marceau and S. P. Ringer, Short-range order in multicomponent materials, Acta Crystallogr., Sect. A:Found. Crystallogr., 2012, 68, 547–560 CrossRef CAS PubMed.
- Y. Xie, M. N. Li, H. Q. Chen and B. Zhang, Effects of the combination of repeated heat-moisture treatment and compound enzymes hydrolysis on the structural and physicochemical properties of porous wheat starch, Food Chem., 2019, 274, 351–359, DOI:10.1016/j.foodchem.2018.09.034.
- J. Wang, C. Chen and J. Xie, Loading oregano essential oil into microporous starch to develop starch/polyvinyl alcohol slow-release film towards sustainable active packaging for sea bass (Lateolabrax japonicus), Ind. Crops Prod., 2022, 188, 115679, DOI:10.1016/j.indcrop.2022.115679.
- Z. Han, Y. Li, D. Luo, Q. Zhao, J. Cheng and J. Wang, Structural variations of rice starch affected by constant power microwave treatment, Food Chem., 2021, 359, 129887, DOI:10.1016/j.foodchem.2021.129887.
- L. Hakim, P. Triwitono, Supriyanto and D. W. Marseno, Microwave treatment to optimize physicochemical properties of modified Busil (Xanthosoma sagittifolium) starch, Food Res., 2022, 6, 304–311 Search PubMed.
- L. Guo, J. Li, Y. Gui, Y. Zhu, B. Yu, C. Tan, Y. Fang and B. Cui, Porous starches modified with double enzymes: Structure and adsorption properties, Int. J. Biol. Macromol., 2020, 164, 1758–1765, DOI:10.1016/j.ijbiomac.2020.07.323.
- A. Dura and C. M. Rosell, Physico-chemical properties of corn starch modified with cyclodextrin glycosyltransferase, Int. J. Biol. Macromol., 2016, 87, 466–472, DOI:10.1016/j.ijbiomac.2016.03.012.
- T. d. S. Rocha, A. P. d. A. Carneiro and C. M. L. Franco, Effect of enzymatic hydrolysis on some physicochemical properties of root and tuber granular starches, Cienc. Tecnol. Aliment., 2010, 30, 544–551 CrossRef.
- E. Subroto, F. Filianty, R. Indiarto and A. A. Shafira, Physicochemical and functional properties of modified adlay starch (Coix lacryma-jobi) by microwave and ozonation, Int. J. Food Prop., 2022, 25, 1622–1634, DOI:10.1080/10942912.2022.2096061.
- S. Zeng, X. Wu, S. Lin, H. Zeng, X. Lu, Y. Zhang and B. Zheng, Structural characteristics and physicochemical properties of lotus seed resistant starch prepared by different methods, Food Chem., 2015, 186, 213–222 CrossRef CAS PubMed.
- A. Narwojsz, E. J. Borowska, M. Polak-Śliwińska and M. Danowska-Oziewicz, Effect of Different Methods of Thermal Treatment on Starch and Bioactive Compounds of Potato, Plant Foods Hum. Nutr., 2020, 75, 298–304 CrossRef CAS PubMed.
- E. A. Saputro, R. R. Yogaswara, N. Arviana, L. Aini, A. R. Y. Sunarti and R. E. Nugraha, Mass Balance Analysis of Bioethanol Production from Petai Peel (Parkia speciosa) through Enzymatic Process, International Journal of Eco-Innovation in Science and Engineering , 2021, 2(2), 30–33, DOI:10.4186/ijeise.upnjatim.ac.id.
- L. Purwitasari, M. P. Wulanjati, Y. Pranoto and L. D. Witasari, Characterization of porous starch from edible canna (Canna edulis Kerr.) produced by enzymatic hydrolysis using thermostable α-amylase, Food Chem. Adv., 2023, 2, 100152, DOI:10.1016/j.focha.2022.100152.
- L. Guo, Y. Yuan, J. Li, C. Tan, S. Janaswamy, L. Lu, Y. Fang and B. Cui, Comparison of functional properties of porous starches produced with different enzyme combinations, Int. J. Biol. Macromol., 2021, 174, 110–119 Search PubMed.
- L. Liu, W. Shen, W. Zhang, F. Li and Z. Zhu, Porous Starch and Its Applications, Functional Starch and Applications in Food, 2018, pp. 91–113 Search PubMed.
- Y. Benavent-Gil and C. M. Rosell, Comparison of porous starches obtained from different enzyme types and levels, Carbohydr. Polym., 2017, 157, 533–540, DOI:10.1016/j.carbpol.2016.10.047.
- B. Z. Ying, H. Kamilah, A. A. Karim and U. Utra, Effects of heat-moisture and alkali treatment on the enzymatic hydrolysis of porous sago (Metroxylon sagu) starch, J. Food Process. Preserv., 2020, 1–12 CAS.
- H. Singh, N. S. Sodhi and N. Singh, Structure and functional properties of acid thinned sorghum starch, Int. J. Food Prop., 2009, 12, 713–725 CrossRef CAS.
- S. Mehboob, T. M. Ali, F. Alam and A. Hasnain, Dual modification of native white sorghum (Sorghum bicolor) starch via acid hydrolysis and succinylation, Lwt, 2015, 64, 459–467, DOI:10.1016/j.lwt.2015.05.012.
- S. Ragaee and E. M. Abdel-Aal, Pasting properties of starch and protein in selected cereals and quality of their food products, Food Chem., 2006, 95, 9–18 CrossRef CAS.
- K. O. Falade and O. E. Ayetigbo, Effects of tempering (annealing), acid hydrolysis, low-citric acid substitution on chemical and physicochemical properties of starches of four yam (Dioscorea spp.) cultivars, J. Food Sci. Technol., 2017, 54, 1455–1466 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |


