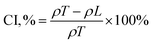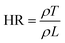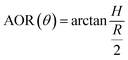Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Research progress on plant-based protein materials in food 3D printing: forming mechanisms, stabilization mechanisms, and applications
Shishuai
Cui
ab,
Tianlong
Xiao
ad,
Yichen
Zheng
a,
Shinuo
Cao
ac,
Xinyu
Zhang
a,
Chao
Wu
a,
Fei
Xiang
a,
Qiang
Wang
 a,
Bo
Jiao
a,
Xiaojie
Ma
*a and
Aimin
Shi
a,
Bo
Jiao
a,
Xiaojie
Ma
*a and
Aimin
Shi
 *ab
*ab
aInstitute of Food Science and Technology, Chinese Academy of Agricultural Sciences, Comprehensive Utilization Laboratory of Cereal and Oil Processing, Ministry of Agriculture and Rural, Beijing 100193, China. E-mail: maxiaojie@caas.cn; shiaimin@caas.cn
bCollege of Food Science and Engineering, Qingdao Agricultural University, Qingdao 266109, China
cCollege of Food Science and Technology, Nanchang University, Nanchang 330047, China
dCollege of Light Industry and Food Sciences, Zhongkai University of Agriculture and Engineering, Guangzhou 510225, China
First published on 6th March 2026
Abstract
The integration of plant-based foods with 3D printing technology offers a revolutionary solution for addressing future food security, sustainable manufacturing, and personalized nutrition. However, the inherent rheological limitations and processing instabilities of native plant proteins remain critical bottlenecks restricting their widespread adoption in additive manufacturing. This review aims to systematically summarize the research progress of plant protein-based materials across three primary food 3D printing technologies: Binder Jetting (BJT), extrusion-based printing, and inkjet printing. First, the forming and stabilization mechanisms under different printing modes are critically analyzed, focusing on the powder–binder interfacial wetting in BJT, the shear-thinning and thixotropic recovery of emulsion gels in extrusion, and the piezoelectric jettability and rapid gelation kinetics of hydrogels in inkjet printing. Second, strategies to optimize the printability of plant proteins via molecular modification (e.g., enzymatic hydrolysis, covalent/non-covalent cross-linking) and multi-scale structural design (e.g., polysaccharide complexation, bimodal particle size distribution) are elaborated. The review further showcases innovative applications in whole-cut meat analogues, personalized dysphagia diets, and functional bioactive carriers. Finally, the challenges regarding food safety (microbial and allergen risks), regulatory lag, and consumer acceptance are critically discussed. Future perspectives highlight the immense potential of multi-material co-extrusion, 4D printing, and Artificial Intelligence (AI)-driven inverse formulation design in accelerating the industrialization of plant-based food 3D printing.
Sustainability spotlightThis review systematically explores the forming and stabilization mechanisms of plant protein-based materials in food 3D printing. This technology can promote the high-value utilization of abundant plant protein resources, enable nutrition customization through digital manufacturing, and reduce raw material waste, providing a technological pathway for establishing a low-carbon, efficient, and personalized sustainable future food system, supporting the United Nations Sustainable Development Goals (SDG 2, 3, 12). |
1 Introduction
Additive Manufacturing (AM), commonly referred to as 3D printing, utilizes computer-aided techniques to fabricate objects with complex three-dimensional structures. Since its inception in the 1980s, AM technology has gained widespread recognition and has been applied across diverse fields.1 Governments worldwide have implemented strategic initiatives to promote its development. For instance, China has emphasized accelerating the advancement of its manufacturing sector to capitalize on the ongoing technological revolution and industrial transformation.2 Currently, as a cornerstone of intelligent manufacturing, 3D printing technology is highly valued and actively promoted on a global scale.3Researchers expect overpopulation to lead to a shortage of food resources by the middle of the twenty-first century. Moreover, environmental safety and public health issues, among others, pose great challenges to the sustainable supply of food and nutritional health.4 Currently, increased greenhouse gas emissions, loss of biodiversity, and geopolitical conflicts have exacerbated the weakening of global supply chains and increased the demand for food for the future. Foods prepared on the basis of plant proteins are important future foods.5 Excellent prospects include the use of plant proteins to produce food products with a fiber structure similar to that of meat products,6 giving the product an appearance and taste similar to animal meat.
Protein is a fundamental nutrient for human health. In recent years, the global demand for protein has surged rapidly, placing immense pressure on limited resources such as land and water. Traditionally, animal-derived proteins are regarded as superior due to their complete essential amino acid profiles and high digestibility. However, the heavy environmental burden of the livestock industry highlights the need for sustainable alternatives. In this context, plant proteins serve as a critical food source. Research indicates that plant proteins are ideal biomaterials characterized by their low cost, wide distribution, and high nutritional value.7 They not only help alleviate food resource shortages but also enable the provision of customized food services for groups with diverse dietary needs. Furthermore, their excellent functional properties, biocompatibility, and safety facilitate their successful application in food 3D printing.8
3D printing technology is uniquely positioned to address future food supply challenges and meet consumer demands for personalized nutrition, such as diets for dysphagia or specific nutritional deficiencies.9 Currently, the primary food 3D printing technologies include binder jetting, extrusion, and inkjet printing. The exploration of 3D food printing can be traced back to the early 21st century, but the use of plant protein as an ink material has only made significant progress in recent years.10 Extrusion-based printing remains the most widely researched method due to its cost-effectiveness, material versatility, and capability to process high-viscosity plant protein pastes for customized dietary needs.11 Binder jetting printing (inkjet printing combined with powder bed technology to construct three-dimensional structures) in the field of materials has already achieved the creation of highly porous and full-color structures, and its future applications in the food sector are promising.12,13 As illustrated in Fig. 1, there is an intrinsic relationship between plant protein-based materials and these printing methods. Recent advancements have focused on multi-nozzle extrusion systems to construct sophisticated analogues; for instance, Li et al. successfully printed the fatty and lean portions of plant-based fish products by co-extruding distinct protein emulsions, achieving a structural precision rate of 90%.14 Despite these advances, the application depth varies significantly across methods. Binder Jetting, while mature in biomedical and engineering fields, is still in its infancy within the food sector.15 Research on the molding and stabilization mechanisms of plant protein powders in BJT remains limited. Similarly, food inkjet printing, which shares material requirements with hydrogel processing, requires further exploration regarding ink stability and printability.
 | ||
| Fig. 1 The relationship between plant protein-based materials and food 3D printing is briefly described. | ||
Therefore, this review aims to provide a comprehensive overview of the progress in plant protein-based 3D printing. We systematically analyze the forming and stabilization mechanisms of plant protein materials across extrusion, binder jetting, and inkjet printing methods, focusing on the interplay between raw materials, printing parameters, and post-processing. This review offers theoretical insights to accelerate the industrial application of plant proteins in future food manufacturing.
2 Characterization of different food 3D printing methods and properties
Currently, the food industry has explored several mainstream printing methods. Before delving into the various food printing methods, this research has compiled and analyzed the current research progress to visually demonstrate the research intensity and latest achievements in different food printing methods, detailed information is provided in Table 1.| 3D printing methods | Raw materials | Goal | Achievements | Reference |
|---|---|---|---|---|
| Extrusion 3D printing | Soybean protein isolate/xanthan gum/rice starch composite gel; starch/carrageenan emulsion gel | Construct plant-based fish with complex muscle and fat morphology |

|
14 |
| Soybean protein isolate/wheat flour/rice protein blend | Match nutritional profile of real meat (approx. 25% protein) |

|
16 | |
| Pea fiber, pea starch, and pea protein composite | Quantitative study on macronutrients' effect on printability |

|
17 | |
| Flaxseed protein/sodium alginate complexes | Stabilize curcumin-loaded high internal phase emulsions (HIPEs) |
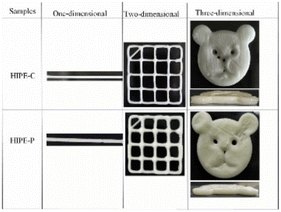
|
18 | |
| Pea protein with different pectin types | Enhance physicochemical properties via ultrasonic treatment |

|
19 | |
| Pea protein, corn starch, flaxseed oil and transglutaminase | Create salmon fillet simulants with fibrous texture |

|
20 | |
| Pea isolate protein–inulin coupling | Develop solid fat substitutes |

|
21 | |
| Pea protein/glycyrrhetinic acid/carrageenan | Improve rheology and functional properties |

|
22 | |
| White mushroom powder + soybean protein isolate | Develop dysphagia diet |

|
23 | |
| Binder jetting printing | Pea protein and powdered sugar; binder: copovidone and thiamine | Screen parameters for nutrient stability and accuracy | Established optimal binder/powder ratios for precise nutrient dosing. The printed structures maintained high stability and accurate vitamin distribution post-processing | 24 |
| Calcium caseinate/wheat starch/medium chain triglycerides; binder: tween 20 | Explore complex structures and binder saturation |
|
25 |
2.1 Binder jet printing
Technically, BJT combines powder bed fusion principles with inkjet technology.31 The printheads are typically categorized as continuous or Drop-on-Demand (DOD). For food applications, DOD systems (thermal or piezoelectric) are preferred due to their precision and have become a common choice.32 The BJT system generally consists of a powder recoating mechanism (roller or blade), a printhead, and a build platform. The printing process, as illustrated in Fig. 2, proceeds as follows: first, the recoating mechanism spreads a thin, uniform sacrificial layer of powder onto the platform. Next, the printhead selectively ejects binder droplets onto the powder bed according to digital slice data. The interaction between the liquid binder and powder particles forms liquid bridges, which initially bind the particles together. To enhance the structural integrity of each layer against shear forces during subsequent powder spreading, heating lamps (e.g., IR heaters) are often employed to facilitate solvent evaporation or rapid recrystallization. This process—powder spreading followed by binder jetting—is repeated layer by layer until the green part (printed blank) is completed. Finally, the printed object undergoes post-processing, such as high-temperature baking or drying, to remove excess moisture and strengthen the solid bridges between particles, thereby obtaining the finished product.33 While BJT has been a research hotspot in the pharmaceutical field since 2015 for developing orally disintegrating tablets,34 its application in the food sector focuses on creating low-moisture products with customized textures and flavors. Since the binder content is typically low, the final product retains the natural properties of the edible powder substrate, meeting consumer demands for clean-label and healthy foods.35
2.1.2.1 Powder flowability. Powder flowability is a determinant factor for the uniformity of the powder bed layer. Poor flowability leads to uneven spreading and defects in the printed green part.38 Since flowability is influenced by the physical properties of the powder and the stress environment, its liquidity evaluation method cannot be represented by a single measure. The materials field currently tends to use multi-indicator collaborative statistical methods to determine excellent fluidity.39 The core indicators of powder flowability primarily include static flowability, angle of repose, and dynamic flowability:
2.1.2.1.1 Static fluidity. Static fluidity refers to the tendency of a powder to resist flow in the absence of external driving forces or during low-speed movement, reflecting its ability to maintain a stable accumulation pattern.40 Static fluidity is commonly characterized using Carr's Index (CI) and Hausner's Ratio (HR). Carr's Index quantifies the compressibility and flow potential of the powder, while Hausner's ratio reflects the strength of internal friction and adhesion between powder particles, which can provide key guidance for material manufacturability, print quality and final product performance.41 The Carr's index to Hausner's ratio is mainly obtained by calculating the loose packing density (ρL) and knockdown packing density (ρT) with the following equations:42
Generally, particles exhibit good flowability when Carr's index < 16 and Hausner's ratio < 1.19.43 However, unmodified plant protein powders often exhibit HR values exceeding 1.25 due to strong inter-particle cohesive forces and irregular morphology, indicating a need for flow aids or granulation to meet binder jetting requirements.44
2.1.2.1.2 Angle of repose (AOR). Angle of repose (AOR) quantifies the resistance to flow by the angle between the inclined plane of the cone formed by the natural accumulation of powder and the horizontal plane,45 and is directly related to the uniformity of powder spreading, the quality of the interlayer bonding, and the performance of the final product during the printing process. The angle of repose depends on many factors, such as particle friction, cohesion, shape, drop height, and error, and is calculated using the following formula:
where H represents the critical height of the powder cone at equilibrium, and R represents the radius of the cone's base. Crucially, H and R are not independent geometric variables but are the integrated outcome of the powder's physical properties. Particle friction and irregular shape hinder flow, promoting a taller cone (increased H) relative to its base, thus raising θ. Cohesive forces further enhance this resistance to spreading. Conversely, a higher drop height may impart greater kinetic energy to particles, leading to a wider base (increased R) and a potentially lower θ. Therefore, this simple formula encapsulates complex granular interactions, with measurement variability in H and R accounting for experimental error. A thorough analysis of these parameters provides deep insight into powder behavior essential for process optimization.46
2.1.2.1.3 Dynamic fluidity. Dynamic fluidity refers to the ability of a powder to flow continuously under the drive of external forces (such as vibration, airflow, mechanical stirring), reflecting its efficiency in overcoming cohesion and friction resistance to achieve stable transportation. Dynamic fluidity directly affects the stability of the powder supply system layer thickness uniformity, which is a key parameter to ensure continuous production.47
For plant protein materials, dynamic fluidity is a critical predictor of incomplete layering. Due to the irregular morphology and high surface energy of milled protein particles, they tend to agglomerate under dynamic stress. A powder with poor dynamic fluidity may appear stable statically but will drag or tear during recoating, severely affecting the uniformity of layer thickness and the density of the printed green part.48 Indicators such as Basic Flow Energy (BFE) and Specific Energy (SE), measured via powder rheometry, are often used to quantify this dynamic behavior.
2.1.2.1.4 Comprehensive evaluation strategy. Given the complexity of plant protein powders—where factors like moisture content, particle shape, size distribution, and surface chemistry interact—relying on a single flowability indicator is often insufficient to predict printability.49 Therefore, current research advocates for a multi-indicator collaborative assessment. Statistical approaches, such as entropy-weighted TOPSIS (Technique for Order of Preference by Similarity to Ideal Solution) and Principal Component Analysis (PCA),50 are increasingly applied to weigh static (CI, HR, AoR) and dynamic metrics. This establishes a comprehensive printability score, allowing for the precise screening of plant protein formulations that balance flowability with binder wettability.
2.1.2.2 Binder sprayability. Binder sprayability refers to the ability of the liquid binder to form stable, spherical micro-droplets without generating satellite droplets or clogging the nozzle.51 This property is strictly governed by the fluid's physical properties—specifically density (ρ), dynamic viscosity (η), and surface tension (γ)—and their interaction with the nozzle diameter (α) and jetting velocity (v).52
In the context of food 3D printing, three key dimensionless numbers are employed to systematically predict the printability of the binder solution: the Reynolds number (Re), Weber number (We), and Ohnesorge number (Oh). These are defined as follows:
| Re = vρα/η = (inertial forces)/(viscous forces) |
| We=(v2ρα)/γ = (inertial forces)/(surface forces) |
| Oh = √We/Re = (viscous forces)/(surface and inertial forces) |
| Z = 1/Oh |
2.1.2.3 Characteristics of the printed product. The quality of plant protein-based foods produced via Binder Jetting is rigorously evaluated through three primary dimensions: mechanical properties, dimensional accuracy, and microstructural characteristics. These indicators directly reflect the efficacy of the powder–binder interaction and the optimization of printing parameters.
2.1.2.3.1 Mechanical properties (stability). The mechanical integrity of the printed product relies fundamentally on the bonding force between the plant protein particles and the cured binder.55 Since plant protein powders are non-melting, the structural strength is derived from the solid bridges formed by the binder (e.g., dried Gum Arabic) between particles. Key indicators include hardness, fracturability, and compressive strength. A common defect in plant-based BJT is the weak green part, where insufficient binder penetration or poor wettability (due to lack of surfactants like Tween) leads to delamination or collapse during depowdering.
2.1.2.3.2 Dimensional accuracy. Accuracy is defined by the deviation between the designed digital model and the actual printed object. For plant protein powders, accuracy is heavily influenced by particle size distribution and binder diffusion. Large, irregular protein particles often result in high surface roughness (Ra) and stair-stepping effects.56 Conversely, excessive binder saturation can cause unwanted spreading, reducing resolution. Accuracy is typically quantified by calculating the dimensional deviation rate (%) along the X, Y, and Z axes.
2.1.2.3.3 Microstructural characterization. As emphasized by recent studies, macro-scale properties are dictated by micro-scale interactions. Scanning Electron Microscopy (SEM) and X-ray Micro-Computed Tomography (µ-CT) are essential tools for characterizing these features. (1) Inter-particle bonding: SEM is used to visualize the morphology of solid bridges and the wetting behavior of the binder on the protein surface. It can reveal defects such as cracks or incomplete fusion caused by the hydrophobic nature of proteins. (2) Porosity and internal structure: unlike extrusion, BJT naturally produces porous structures.57 µ-CT is used to analyze pore size distribution and interconnectivity. For food applications, this internal porosity is crucial as it directly determines the product's crispness, texture.58
2.2 Extrusion 3D printing
Beyond the fundamental PAM-FDM dichotomy, specific subcategories are relevant in food printing. Ready-to-Eat (RTE) printing typically falls under the PAM paradigm, where the formulated ink itself is edible and requires no further extensive cooking post-printing; structural integrity is achieved through the ink's rheology or mild in situ stabilization (e.g., self-supporting gels). Conversely, hot extrusion shares a conceptual similarity with FDM in its use of temperature to induce flow but is distinct in application. It is employed for food materials that are thermoplastic within a safe food temperature range (e.g., chocolate, certain cheeses, or starch-based melts). Here, the material is heated to become extrudable and solidifies primarily through cooling. Unlike industrial FDM with high-melting polymers, food-grade hot extrusion operates at much lower temperatures and focuses on materials that remain edible after the thermal cycle. Therefore, while PAM is defined by its non-thermal, rheology-driven mechanism, hot extrusion is defined by thermally-induced viscosity reduction within food-safe limits.
2.2.2.1 Rheological properties of inks. The printability of extrusion inks relies on a delicate balance between flowability during extrusion and shape retention after deposition. Shear-thinning behavior is critical; it allows the ink's viscosity to decrease significantly under the high shear stress within the nozzle to facilitate flow.68 This behavior is typically described by the power law (Ostwald-de Waele) model:
η = K![[small gamma, Greek, dot above]](https://www.rsc.org/images/entities/i_char_e0a2.gif) n−1 n−1 |
2.2.2.2 Performance of the printed product. The macroscopic quality of printed food is assessed through two primary dimensions, dimensional accuracy and textural properties.72
2.2.2.2.1 Dimensional accuracy. This metric reflects the fidelity of the printing process. It is quantified by measuring the deviation rate between the digital model and the actual printed geometry (e.g., height, diameter). For plant protein-based inks, dimensional deviations often indicate specific rheological failures. For instance, die swell (expansion at the nozzle exit) suggests excessive elasticity, while structural collapse indicates insufficient yield stress to support gravity. Therefore, high dimensional accuracy serves as a direct validator of the ink's self-supporting capacity.73
2.2.2.2.2 Textural properties. Texture profile analysis provides a quantitative map of the food's mechanical behavior. Key parameters include hardness, springiness, cohesiveness, and chewiness. These mechanical attributes play a decisive role in consumer acceptance, particularly for plant-based meat analogues where mimicking the fibrous texture of animal muscle is critical.74 Furthermore, texture analysis serves as a diagnostic tool; for example, low cohesiveness values often reveal poor interlayer fusion or weak gel network formation within the printed object.
2.2.2.3 Microstructural characterization. Macro-scale texture and mechanical performance are intrinsically dictated by the micro-scale structural organization of the printed food. For plant protein-based extrusion, microstructural analysis is critical for verifying the formation of gel networks and the alignment of protein fibers.75
2.2.2.3.1 Microscopic techniques. Scanning electron microscopy is the primary tool for observing surface morphology and internal cross-linking. It reveals whether the plant protein matrix has formed a dense, continuous network or a loose, porous structure. Confocal laser scanning microscopy allows for the non-destructive observation of component distribution. By fluorescently labeling proteins and lipids separately, researchers can visualize the positioning of oil droplets within the protein gel matrix, which directly correlates with the juiciness and mouthfeel of the printed product.76 In the context of plant-based meat analogues, the shear-induced alignment of protein molecules during extrusion is a key quality indicator. Microscopic images can quantify the degree of anisotropy (fiber orientation). A highly aligned microstructure indicates successful imitation of animal muscle fibers. Conversely, observing the interface between deposited layers helps identify defects such as delamination or incomplete fusion, providing a fundamental explanation for poor mechanical strength in the finished product.
2.3 Inkjet printing
Inkjet technologies are primarily categorized into Continuous Inkjet (CIJ) and Drop-on-Demand (DOD).81 CIJ systems generate a continuous stream of droplets, deflecting selected ones onto the substrate while recycling the rest. Although efficient, CIJ is generally unsuitable for food 3D printing. It requires conductive inks and the recycling process increases the risk of contamination and material degradation.82 DOD systems eject droplets only when required, offering higher precision and minimizing waste. They are further divided by their actuation mechanism: thermal and piezoelectric. Fig. 4 is a basic conceptual diagram of the piezoelectric inkjet printing device.
With the advantages of tunable mechanical properties, good porosity, and easy fabrication, hydrogels have been prominently used in various cutting-edge scientific research. Hydrogels are extremely adaptable and can be fabricated into a variety of styles, so they are a potential raw material for inkjet printing.83 The most prominent feature of natural hydrogel materials is their excellent biocompatibility, but both natural and artificial hydrogels have certain drawbacks, so the development of stable and easy-to-regulate hydrogels suitable for the food field has become a hot issue. To ensure the effect of food inkjet printing, the ink raw materials should meet the characteristics of low viscosity, good fluidity and fast solidification, and combined with the concept of future food that has emerged in recent years, the use of plant protein-based materials to stabilize the hydrogel has become a good choice for future research and development.84
For plant protein-based hydrogels, piezoelectric DOD is the superior choice. Thermal inkjet printheads heat the ink (often >300 °C) to create a vapor bubble for ejection.85 This high temperature can cause denaturation of heat-sensitive plant proteins and destabilize the hydrogel network.86 In contrast, piezoelectric printheads utilize a crystal that deforms under voltage to mechanically eject droplets. This cold process protects the bioactivity and structural integrity of protein molecules. Therefore, current research on plant-based food printing predominantly utilizes piezoelectric DOD technology to ensure both print fidelity and nutritional retention.
Ink ejectability and latency: the primary criterion is whether the ink can form stable droplets. As discussed in Section 2.1.2.2, the Z value (1 < Z < 10) remains the gold standard for predicting jetting behavior. However, for plant protein inks, latency (open time) is a critical unique indicator. Protein solutions tend to form films or gels at the nozzle tip due to solvent evaporation, leading to immediate clogging or misdirected jetting (skewing). Therefore, characterizing the evaporation rate and crusting behavior of the ink is essential for continuous printing. Furthermore, since piezoelectric nozzles typically have diameters of 20–501 µm, the particle size of protein aggregates must be strictly controlled (usually <1 µm) to prevent physical blockage.87
Substrate interaction and spreading: upon impact, the droplet must spread appropriately without excessive bleeding. This behavior is governed by the contact angle between the ink and the substrate. Plant proteins, acting as surfactants, significantly lower the liquid's surface tension. While this aids ejection, it often causes over-spreading on hydrophilic surfaces, reducing print resolution. Therefore, characterizing the equilibrium contact angle and spreading dynamics is vital to ensure sharp feature definition.88
Since inkjet inks are low-viscosity fluids (typically <20 mPa s), they lack inherent shape retention. The printed structure relies entirely on rapid post-deposition solidification. Characterizing the gelation time (sol–gel transition speed) is crucial. If gelation is too slow, the droplets will coalesce into a puddle; if too fast, they may clog the nozzle.89 For plant proteins, monitoring the viscosity change over time under specific triggers (e.g., pH change or calcium ion contact) provides the necessary data to optimize this phase transition.
3 Exploration of stabilization mechanisms and defects in plant protein-based 3D printing
3.1 Binder jet printing based on plant proteins
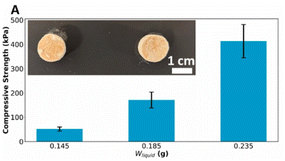
To address global nutritional challenges, plant proteins have emerged as a focal point for food innovation.90,91 Protein powders offer distinct advantages for binder jet printing, including low cost, high nutrient density, and tunable adhesion properties. Currently, over 30 types of plant proteins, spanning grains, legumes, and oilseeds—are commercially used and readily accessible.92 However, native plant protein powders often exhibit technical defects such as high cohesion, strong hygroscopicity, and low bulk density.93 These characteristics hinder the flowability required for uniform powder spreading (recoating). Therefore, modifying the protein structure or incorporating flow aids (e.g., anti-caking agents) is often necessary to facilitate the printing process.94Research on plant-based BJT is still in its nascent stages compared to other methods. A notable investigation by the University of Connecticut focused on pea protein substrates. Chadwick et al. demonstrated that blending pea protein with granulated sugar effectively reinforced the printed structure. Textural analysis revealed that sugar particles filled the inter-powder voids, reducing the porosity of the final product to 37.3% and significantly enhancing compressive strength. The mechanism relies on the sugar reducing the free volume within the powder bed and the binder inducing partial swelling of starch inclusions to bridge the particles.95,96 Although this study utilized pharmaceutical-grade binders (copovidone), it validated the feasibility of using BJT for precise, customized nutritional delivery.24
Beyond physical blending, molecular engineering strategies such as enzymatic processing play a decisive role in optimizing raw materials. By precisely shearing peptide bonds, enzymatic treatment can regulate the molecular weight and solubility of plant proteins.97 For BJT, increased solubility enhances the interaction between the powder and the water-based binder, promoting stronger liquid bridge formation. However, precise process control is critical; excessive hydrolysis may generate bitter peptides or weaken the structural integrity of the post-processed solid network.98
Thermodynamics governs the compatibility between the powder and binder, with wettability—quantified by the three-phase contact angle—being a key determinant.102 A major challenge in plant-based BJT is that many proteins (e.g., zein or high-purity soy isolate) possess hydrophobic surfaces, often resulting in a contact angle greater than 90°. This hydrophobicity prevents effective binder penetration, leading to weak delamination. To address this, optimizing the binder formulation by adding surfactants (e.g., Tween 80) is essential to lower surface tension and ensure a contact angle close to 90°, which balances the need for print accuracy with sufficient infiltration strength.103
Beyond chemical compatibility, the physical quality of the powder bed is a prerequisite for printing stability.101,104 Unmodified plant protein powders often exhibit low bulk density, a high tendency for moisture absorption, and strong electrostatic cohesion.105 These properties frequently cause clumping or dragging defects during the recoating process, creating large pores that compromise resolution.106,107 Consequently, ingredient processing methods play a decisive role in dictating printability by altering particle morphology and size distribution.108 For instance, spherical powders produced via spray drying exhibit superior flowability compared to irregular, rod-like particles resulting from conventional hammer milling.109
To further optimize the powder bed density, particle size distribution strategies are often employed. A trade-off exists where large particles flow well but leave voids, while fine particles yield high resolution but are prone to agglomeration.110 A bimodal particle size distribution strategy effectively resolves this by using fine-milled protein particles (<30 µm) to fill the voids between coarser particles (>80 µm), thereby significantly increasing the tapped density and mechanical strength of the final product.111 Additionally, exogenous regulation, such as coating protein particles with polyphenols or polysaccharides (e.g., maltodextrin) to reduce surface electrostatic charge, has proven effective in improving flowability and ensuring uniform powder spreading.112
Following printing, the fragile green part requires stabilization through post-processing, which differs fundamentally from industrial ceramic sintering. In the field of materials, binder jet printed green bodies need to undergo post-processing, which mainly includes curing, depowdering, and sintering. For plant-based foods, the curing phase is effectively replaced by thermal drying or baking, which removes moisture and induces physicochemical transformations—specifically starch gelatinization or protein denaturation (at temperatures >70 °C). These changes solidify the liquid bridges (e.g., Gum Arabic) into rigid solid bridges, significantly enhancing mechanical strength.114 Depowdering (removing excess powder) presents specific challenges for plant proteins due to their high hygroscopicity and adhesiveness; therefore, precise humidity control is often required during vacuuming or gentle brushing to prevent the powder from sticking to the product surface. To further refine texture, secondary processing techniques are increasingly employed: for example, freeze-drying is utilized to create porous, crispy plant-based snacks by preventing structural collapse, while microwave heating can be used to induce puffing and rapid densification.115
3.2 Extruded 3D printing based on plant proteins
Plant protein-based emulsions and gels are emerging as premier candidates for food extrusion due to their excellent biocompatibility, nutritional value, and tunable rheology.116 As discussed in Section 2.2, it is critical to distinguish this process from fused deposition modeling. Plant protein inks are typically processed via pressure-assisted microsyringe or direct ink writing at room or mild temperatures. This approach avoids thermal degradation of bioactive proteins, making it compatible with a wide range of vegetable protein-based emulsions and gels.117Beyond single-nozzle printing, recent research has pivoted toward multi-material extrusion. This technique utilizes multiple independent nozzles to co-extrude different inks—such as a rigid protein paste for the structural skeleton and a softer fat mimetic for juiciness—thereby constructing heterogeneous food structures that closely mimic real meat or complex confectionery.
To ensure successful printing, this section focuses on the stabilization mechanisms and defect control of plant protein inks.118 The molding mechanism is governed by three core factors: (1) rheological adaptability;119 (2) self-supporting capacity; and (3) interfacial stability.120
Covalent modification can precisely regulate viscoelasticity. For instance, Feng et al. engineered a ternary composite of epigallocatechin gallate (EGCG), high-methoxy pectin, and pea protein via covalent cross-linking. This strategy significantly enhanced the interfacial properties of the protein, preventing oil droplet coalescence. The resulting high-internal-phase emulsion exhibited a robust gel network (high storage modulus, G′), enabling the printing of structures with smooth surfaces and high shape fidelity.123 Similarly, Wu et al. demonstrated that the sol–gel strength is directly linked to particle concentration. By using ultrafine soybean protein isolate (SPI) particles to stabilize Pickering emulsions, they observed that increasing particle concentration from 1.0% to 1.6% enhanced both viscosity and G'. This rheological evolution minimized die swell and improved printing accuracy to over 99%. This underscores that building a robust internal network (reflected in high G′) is the core mechanism for ensuring shape accuracy.124
Non-covalent interactions, such as electrostatic complexation with polysaccharides, are equally effective. Li et al. modified walnut protein emulsions with xanthan gum. They found a direct correlation between xanthan dosage and the gel's structural recovery. Insufficient gum led to weak, loose structures, whereas optimal addition (1.5%) formed a dense network that balanced flowability with self-supporting capacity (G′ > G″). This viscoelastic equilibrium enabled the fabrication of complex geometries with sharp contours.125 Thus, whether through covalent or non-covalent means, the key is strictly regulating the G′/G″ ratio to match the specific shear conditions of the printer.
Different polysaccharides stabilize plant protein gels through distinct mechanisms.128 Weiwei et al. investigated the interaction between soybean protein nanoparticles and various polysaccharides in Pickering emulsions. They found that non-charged polysaccharides (e.g., Locust Bean Gum) stabilized the system primarily by increasing the aqueous phase viscosity. In contrast, charged polysaccharides like Xanthan Gum (XG) and Chitosan (CS) bound to protein nanoparticles via electrostatic interactions, creating a robust interfacial layer. Notably, the XG-reinforced gel at pH 7 exhibited the highest mechanical recovery (97.45%), providing sufficient strength for self-supporting structures, while the CS-stabilized gel demonstrated the lowest deformation rate (5.5%), making it ideal for high-fidelity printing.129
The choice of stabilizer also dictates the textural flexibility of the printed product.130 Zhao et al.131 doped quinoa protein emulsions with various hydrocolloids. They observed that Inulin improved the gel's flexibility, facilitating smoother extrusion flow. Conversely, polysaccharides like fucoidan enhanced rigidity, which is beneficial for shape retention but may require higher extrusion pressures.131 By balancing these additives, plant protein inks can be tailored for specific applications, ranging from customized dysphagia diets to fibrous plant-based meat analogues.132
3.3 Inkjet printing based on plant proteins
Unlike the powder–binder mechanism in BJT, inkjet printing constructs food by directly depositing liquid droplets that solidify upon impact. This section focuses on piezoelectric drop-on-demand technology, as it serves as the standard for processing plant protein-based inks.133 Piezoelectric printing involves three precise steps: droplet formation, ejection via mechanical deformation, and deposition.134 The core advantage of this method lies in its non-thermal actuation. Unlike thermal inkjet heads that vaporize fluid at high temperatures, piezoelectric actuators operate at ambient temperature. This cold process is critical for plant proteins, as it prevents thermal denaturation and preserves the bioactive functionality of the ink during the jetting process.135The primary material for food inkjet printing is the hydrogel, yet developing plant protein-based hydrogels faces a critical rheological paradox. The ink requires low viscosity (typically <20 mPa s) for successful ejection but high viscosity for post-deposition shape retention.136 Pure plant protein solutions often fail to strike this balance, leading to either nozzle clogging at high concentrations or structural collapse at low concentrations.137 To address this, current research focuses on protein–polysaccharide composite systems, where proteins function as nutrients and surfactants, while polysaccharides (e.g., alginate, pectin) serve as gelling agents. The stabilization of these systems relies on a rapid sol–gel transition immediately after deposition, facilitated by intermolecular forces such as hydrogen bonds, hydrophobic interactions, and disulfide bonds.138 Furthermore, to achieve true 3D build-up, dynamic triggers are introduced to lock the liquid droplets into a solid state. Common strategies include ion-induced gelation, where calcium ions (Ca2+) on the substrate instantly cross-link anionic polymer chains, and pH-driven aggregation, which induces rapid solidification by adjusting the local environment to the protein's isoelectric point.139
The interaction between the droplet and the substrate determines the print resolution. Plant protein inks, due to their low surface tension, tend to spread excessively on hydrophilic forming platforms, leading to pixel blurring.146 To counter this, surface modification of the substrate (e.g., applying a hydrophobic coating) or adjusting the substrate temperature to induce immediate gelation is often employed.147 This ensures that the ejected protein droplets maintain their spherical morphology upon impact rather than spreading into irregular puddles.148
3.4 Other advanced technologies
One promising approach to overcome the textural homogeneity of single-material printing is the implementation of co-axial and multi-material extrusion systems.149 Co-axial extrusion utilizes concentric nozzles to construct core–shell filaments; for instance, a lipid-rich emulsion can be encapsulated within a rigid protein paste shell to mimic intramuscular fat, thereby enhancing juiciness and preventing moisture loss during cooking. Similarly, multi-nozzle systems allow for the precise spatial distribution of distinct protein inks—such as pea protein for muscle and textured soy protein for connective tissue—enabling the construction of heterogeneous structures that closely resemble whole-cut meat products.150To achieve micro-scale fiber alignment that surpasses the resolution of mechanical extrusion, electrohydrodynamic printing, also known as electrospinning, is being explored as a complementary strategy.151 This technique employs a high-voltage electric field to pull charged plant protein solutions into nanofibers. These aligned nanofibers act as scaffolds to guide the orientation of subsequent protein layers, drastically improving the anisotropic mechanical properties and fibrous chewiness of meat analogues.152
Furthermore, the concept of 4D printing introduces time as the fourth dimension, allowing printed foods to undergo programmed morphological or color changes in response to external stimuli like pH, heat, or moisture.153 By incorporating pH-sensitive plant pigments (e.g., anthocyanins) into protein inks, researchers can create foods that exhibit interactive color shifts upon contact with acidic seasonings. Additionally, designing specific hydration pathways within the protein matrix allows flat printed films to self-fold into complex 3D shapes during boiling, offering novel solutions for packaging efficiency and creating interactive dining experiences.
4 Application of different printing methods in the food field
While consumer awareness of 3D printed food is still evolving, the technology has successfully transitioned from conceptual prototypes to functional food products. Due to their excellent processing adaptability, plant proteins are currently employed to construct three major product categories:154 plant-based meat analogues, personalized nutritional foods, and complex functional confectionery.155Among these applications, the fabrication of plant-based meat analogues is the most prominent sector, where extrusion-based printing (specifically PAM) dominates due to its ability to mimic the fibrous texture of animal muscle.156 For instance, Ben-Arye et al.157 successfully printed a soy protein-based scaffold exhibiting high anisotropy, simulating the muscle fiber structure of beef and achieving a chewiness comparable to cooked meat. Furthermore, through multi-material co-extrusion technology, researchers have constructed biphasic systems with complex internal structures. Jung et al. co-extruded red beet-colored pea protein (mimicking muscle) with a coconut oil emulsion (mimicking fat) to produce a marbled wagyu beef alternative. Notably, this product released juice upon cooking similar to real meat, significantly enhancing the sensory experience.158
In contrast to the dense textures produced by extrusion, binder jetting excels in fabricating porous, crispy personalized nutritional snacks. Due to its unique powder binding mechanism, BJT generates highly aerated internal structures, making it ideal for developing foods tailored to specific populations (e.g., elderly patients with dysphagia). Zhu et al. utilized a calcium caseinate and natural starch system to develop a high-protein casual snack with tunable hardness and rapid oral dissolution rates, effectively addressing feeding safety issues for specific groups.25 Simultaneously, BJT demonstrates immense potential in the precise delivery of micronutrients. Chadwick et al. demonstrated that this technology can precisely distribute vitamins within a pea protein matrix. By achieving dosage control through localized binder deposition, this method avoids the thermal degradation of heat-sensitive nutrients, providing a new approach for manufacturing personalized dietary supplements.24
Although inkjet printing is less common in bulk food production, it has found a unique niche in surface decoration and functional bioactive delivery. Plant protein hydrogels are frequently used as protective carriers for sensitive compounds like probiotics or polyphenols. Recent studies have utilized piezoelectric inkjet systems to deposit alginate/pea protein microbeads loaded with probiotics onto food surfaces, using the protein matrix to protect bioactives from gastric digestion.159 Additionally, by incorporating pH-sensitive plant pigments (e.g., anthocyanins from purple sweet potato), researchers have developed interactive 4D foods. Patterns printed on protein matrices change color in response to environmental pH shifts (e.g., spraying with lemon juice), offering highly attractive visual interactive experiences for high-end dining.160
While traditional extrusion and binder jetting have laid the foundation for food 3D printing, they often struggle to replicate the complex hierarchical architecture of natural tissues, such as the vascular networks or aligned myofibers found in meat. To address this limitation, researchers are increasingly adapting advanced strategies from tissue engineering and bio-printing to plant-based food fabrication (Fig. 5).155
5 Prospects and challenges
5.1 Prospects and dilemmas of 3D printing of plant protein-based foods
The diversity of plant proteins and printing methods leads to significant variations in print quality, hindering the standardization required for widespread home and personalized use.161,162 Unlike traditional processing, the additive manufacturing workflow involves unique pre-treatment and post-processing steps that lack comprehensive safety assessments. Therefore, establishing a database of optimal printing parameters for various plant substrates is crucial.The sustainability value of plant protein production is superior to that of animal proteins. Core advantages include revolutionary reductions in carbon footprints and the integration of a circular economy model. This is demonstrated by: (1) land intensification and efficient raw material utilization;163 (2) significant carbon emission reductions across the life cycle; (3) high-value utilization of agricultural waste within an industrial symbiosis system; and (4) global policy synergy. For instance, the EU's Carbon Border Adjustment Mechanism (CBAM) encourages the prioritization of low-carbon plant proteins, aligning with the 2019 concept of food of the future.164,165 Theoretically, Binder Jetting offers high productivity and nutritional customization. However, in practice, the mechanism of structural formation varies significantly between binders and plant proteins, necessitating high-throughput screening to determine optimal conditions. Furthermore, plant protein powders often yield parts with low mechanical strength and irregular porous structures due to inconsistent particle sizes. Similarly, inkjet printing faces rheological limitations. While plant protein hydrogels hold promise for delivering nutrients, their high viscosity often limits printing speed. Additionally, post-processing steps like freeze-drying increase complexity and cost. For example, while a mixture of sodium caseinate and concentrated milk protein has been shown to enhance the gel network and shape retention, such high-performance formulations can be costly.166
A critical, often overlooked dilemma is food safety. Plant protein pastes and hydrogels typically possess high water activity and nutrient density, creating an ideal breeding ground for pathogenic microorganisms (e.g., Salmonella or Listeria) if left in the printer nozzle at room temperature. Unlike thermal extrusion, the mild processing temperatures of PAM or Inkjet are insufficient to sterilize the food. Furthermore, the dead zones in complex nozzle geometries make thorough cleaning difficult, leading to biofilm formation. Additionally, the use of diverse plant sources (soy, nuts, wheat) introduces the risk of allergen cross-contamination in multi-material printers, necessitating strict cleaning protocols and dedicated equipment standards.
5.2 Laws and regulations
Global regulation of plant protein 3D printed foods exhibits a pattern of leniency and strictness. The EU adopts a strict approach, requiring safety assessments by the EFSA for new plant proteins (e.g., the 18-month approval for mung bean protein) and banning traditional names like milk on labels. Conversely, the U.S. follows a decentralized approach, with the FDA reviewing raw material safety and the USDA overseeing labeling. China focuses on material safety standards for food contact parts (e.g., banning leaded nozzles) but lacks specific regulations for printed food products [cite]. Emerging markets like Singapore favor innovation, allowing ingredients to be marketed if they meet GRAS (Generally Recognized As Safe) standards. The central contradiction lies in the blurring of technological classifications and the absence of transnational standards, highlighting the need for internationally recognized safety frameworks.5.3 Consumer acceptance
Consumer attitudes toward plant protein 3D printed food are characterized by rational observation and scenario differentiation. Positive drivers include sensory breakthroughs (e.g., high-fidelity plant-based steaks), personalized health benefits (e.g., high-protein pasta for fitness), and environmental values. However, barriers remain significant: price premiums (>30%), concerns over food safety (technophobia), and cultural resistance in traditional dietary regions.To bridge the gap between technological feasibility and market acceptance, the transformation of plant proteins into functional inks must integrate biotechnological tools. Enzyme engineering and synthetic biology act as molecular scissors, precisely splicing protein structures to overcome inherent limitations.167,168 In the long term, AI-assisted formulations could enable the precise assembly of synthetic biology-derived proteins into customized textures.169 However, as most of these technologies are still in the laboratory phase, enhancing product transparency and public communication is imperative.
5.4 Future functionality exploration
To transcend natural limitations, synthetic biology is driving a paradigm shift. Bacterial platforms (e.g., E. coli) enable the mass production of bioactive components like cyclic peptides. These rigid structures can enhance ink yield stress (>250 Pa) while encapsulating heat-sensitive nutrients (e.g., Vitamin D3) within their hydrophobic cavities, preventing degradation during extrusion.170,171Furthermore, microbial fermentation can optimize the nutritional profile. For instance, fermenting soy protein with Lactobacillus plantarum has been shown to increase essential amino acid bioavailability by over 100% and reduce anti-nutritional factors.172,173 Future breakthroughs should focus on: (1) multi-scale computational models (Molecular Dynamics and Machine Learning) to predict rheological responses; (2) in situ enzyme activation systems within the nozzle to trigger cross-linking only at the moment of extrusion; and (3) emulsion electrostatic deposition for spatially resolved nutrient delivery. These innovations will propel plant-based 3D printing from simple shape imitation to a new era of dual precision in texture and nutrition.
5.5 Artificial intelligence and machine learning: accelerating ink development
Traditionally, the development of plant protein-based inks relies heavily on trial-and-error experiments, which are time-consuming and resource-intensive due to the complex, non-linear relationships between rheological parameters and printing quality. With the advent of Industry 4.0, Artificial Intelligence and Machine Learning are emerging as powerful tools to decouple these complexities and predict printability with high accuracy.Recent studies have demonstrated the efficacy of ML in optimizing food ink formulations. For instance, ref. 155 established a machine learning-enabled framework that combined rheological data with image-based assessment. By training algorithms on visual datasets of printed structures, they successfully predicted the printability of polysaccharide-based inks, significantly reducing the experimental workload. Similarly, they utilized a Gaussian process regression (GPR) model to predict the printability of complex food formulations. This data-driven approach allows researchers to input raw material parameters (e.g., protein concentration, gum type) and output predicted printing outcomes (e.g., shape fidelity), thereby realizing inverse design of food inks.174
For plant protein-based 3D printing, future research should focus on building comprehensive databases linking Molecular Structure – Rheology – Print Quality. By integrating Multi-Layer Perceptrons (MLP) or Convolutional Neural Networks (CNN), researchers can accelerate the screening of plant protein sources and optimize processing parameters, shifting the paradigm from empirical observation to intelligent prediction.
6 Conclusion
In summary, advancing the large-scale application of plant proteins in food 3D printing requires addressing critical challenges spanning from fundamental science to industrialization. First, research must prioritize optimizing the modification processes of plant proteins. Precise regulation of their rheological properties and gelation behavior is essential to enhance structural precision and storage stability while controlling costs. Second, it is imperative to establish specific standards and regulations. A unified system for evaluating the safety of printed plant-based materials—covering microbial risks and allergen management—is urgently needed to support commercialization. Ultimately, by developing novel plant-based inks that combine nutritional functionality with printability, and integrating these with AI-driven process control, we can unlock the immense potential of 3D printing for personalized nutrition and sustainable food manufacturing.Author contributions
Shishuai Cui: writing – original draft preparation and revision, conceptualization, visualization. Tianlong Xiao and Yichen Zheng: visualization. Shinuo Cao: conceptualization. Xinyu Zhang, Chao Wu and Fei Xiang: validation. Qiang Wang and Bo Jiao: project administration and resources. Xiaojie Ma and Aimin Shi: conceptualization, supervision, validation, writing – review & editing, project administration and resources.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are included within the article. Additional datasets generated or analyzed during the current work are available from the corresponding author upon reasonable request.Acknowledgements
This research was funded by the National Key Research and Development Plan (2022YFF1101400), National Natural Science Foundation of China (32472271 and 32172149), and Agricultural Science and Technology Innovation Project (CAAS-ASTIP-2023-IFST).Notes and references
- A. Bernard, J. P. Kruth, J. Cao, G. Lanza, S. Bruschi, M. Merklein, T. Vaneker, M. Schmidt, J. W. Sutherland, A. Donmez and E. J. da Silva, CIRP J. Manuf. Sci. Technol., 2023, 47, 18–58, DOI:10.1016/j.cirpj.2023.08.005
.
- E. Forcael, M. Medina, A. Opazo-Vega, F. Moreno and G. Pincheira, Autom. ConStruct., 2025, 169, 105888, DOI:10.1016/j.autcon.2024.105888
.
- S. Kumar Panda, K. Charan Rath, S. Mishra and A. Khang, Mater. Today: Proc., 2023 DOI:10.1016/j.matpr.2023.10.138
.
- K. Pandian, M. R. A. F. Mustaffa, G. Mahalingam, A. Paramasivam, A. John Prince, M. Gajendiren, A. R. Rafiqi Mohammad and S. T. Varanasi, J. Hazard. Mater. Adv., 2024, 16, 100479, DOI:10.1016/j.hazadv.2024.100479
.
- Y. Li, N. Xiang, Y. Zhu, M. Yang, C. Shi, Y. Tang, W. Sun, K. Sheng, D. Liu and X. Zhang, Trends Food Sci. Technol., 2024, 147, 104439, DOI:10.1016/j.tifs.2024.104439
.
- T. Su, B. Le, W. Zhang, K. H. Bak, P. O. Soladoye, Z. Zhao, Y. Zhao, Y. Fu and W. Wu, Food Res. Int., 2024, 186, 114351, DOI:10.1016/j.foodres.2024.114351
.
- G. Karabulut, B. G. Subası, P. Ivanova, G. Goksen, V. Chalova and E. Capanoglu, Food Res. Int., 2025, 202, 115553, DOI:10.1016/j.foodres.2024.115553
.
- Y. Wang, D. J. McClements, C. Bai, X. Xu, Q. Sun, B. Jiao, S. Miao, Q. Wang and L. Dai, Trends Food Sci. Technol., 2024, 153, 104691, DOI:10.1016/j.tifs.2024.104691
.
- Y. Liu, Y. Zhang, L. Cai, Q. Zeng and P. Wang, Int. J. Biol. Macromol., 2024, 272, 132884, DOI:10.1016/j.ijbiomac.2024.132884
.
- H. A. Neamah and J. Tandio, Heliyon, 2024, 10(13), e33882, DOI:10.1016/j.heliyon.2024.e33882
.
- P. De Wever, B. Van Ostaeyen, R. Kargl, K. S. Kleinschek and P. Fardim, Int. J. Biol. Macromol., 2025, 294, 139399, DOI:10.1016/j.ijbiomac.2024.139399
.
- T.-Y. Cheng, Y.-C. Weng, C.-H. Chen and Y.-C. Liao, Addit. Manuf., 2023, 74, 103734, DOI:10.1016/j.addma.2023.103734
.
- D. Gamdha, K. Saurabh, B. Ganapathysubramanian and A. Krishnamurthy, Finite Elem. Anal. Des., 2025, 251, 104410, DOI:10.1016/j.finel.2025.104410
.
- J. Li, H. Hu, R. Niu, Q. Zhu, S. Yao, J. Zhou, D. Liu and E. Xu, Food Phys., 2025, 2, 100028, DOI:10.1016/j.foodp.2024.100028
.
- Y. Wang, N. Genina, A. Mullertz and J. Rantanen, Basic Clin. Pharmacol. Toxicol., 2024, 134(3), 325–332, DOI:10.1111/bcpt.13974
.
- Y. Qiu, D. J. McClements, J. Chen, C. Li, C. Liu and T. Dai, Food Res. Int., 2023, 167, 112635, DOI:10.1016/j.foodres.2023.112635
.
- A. Venkatachalam, A. Balasubramaniam, P. F. C. Wilms, L. Zhang and M. A. I. Schutyser, Food Hydrocoll., 2023, 142, 108760, DOI:10.1016/j.foodhyd.2023.108760
.
- Y. Chen, Y.-S. Wang, Y.-H. Li, Z.-Y. Li, Y.-Z. Ma, X.-Y. Qin and H.-H. Chen, Colloids Surf., A, 2023, 673, 131863, DOI:10.1016/j.colsurfa.2023.131863
.
- M. Gu, H. Gu, V. Raghavan and J. Wang, Future Foods, 2024, 9, 100377, DOI:10.1016/j.fufo.2024.100377
.
- Y. Wu, Z. Li, L. Yang, H. Zheng and C. Xue, Food Hydrocoll., 2024, 155, 110242, DOI:10.1016/j.foodhyd.2024.110242
.
- W. Jiang, W. Li, J. Li, D. J. McClements, C. Ma, S. Chen, X. Liu and F. Liu, Food Hydrocoll., 2023, 142, 108820, DOI:10.1016/j.foodhyd.2023.108820
.
- Q. Lin, M. Shang, X. Li, S. Sang, L. Chen, J. Long, A. Jiao, H. Ji, C. Qiu and Z. Jin, Food Hydrocoll., 2024, 147, 109347, DOI:10.1016/j.foodhyd.2023.109347
.
- K. Xiao, J. Zhang, L. Pan and K. Tu, Food Res. Int., 2024, 175, 113760, DOI:10.1016/j.foodres.2023.113760
.
- E. Chadwick, G. P. S. R. Bertola, X. Lin, A. H. Barrett, M. Okamoto, X. Lu and A. W. K. Ma, J. Food Eng., 2025, 391, 112465, DOI:10.1016/j.jfoodeng.2024.112465
.
- S. Zhu, P. Vazquez Ramos, O. R. Heckert, M. Stieger, A. J. van der Goot and M. Schutyser, J. Food Eng., 2022, 332, 111124, DOI:10.1016/j.jfoodeng.2022.111124
.
- S. Beg, W. H. Almalki, A. Malik, M. Farhan, M. Aatif, Z. Rahman, N. K. Alruwaili, M. Alrobaian, M. Tarique and M. Rahman, Drug Discovery Today, 2020, 25(9), 1668–1681, DOI:10.1016/j.drudis.2020.07.007
.
- P. Szymczyk-Ziółkowska, M. B. Łabowska, J. Detyna, I. Michalak and P. Gruber, Biocybern. Biomed. Eng., 2020, 40(2), 624–638, DOI:10.1016/j.bbe.2020.01.015
.
- K. Sen, T. Mehta, S. Sansare, L. Sharifi, A. W. K. Ma and B. Chaudhuri, Adv. Drug Delivery Rev., 2021, 177, 113943, DOI:10.1016/j.addr.2021.113943
.
- M. Ziaee and N. B. Crane, Addit. Manuf., 2019, 28, 781–801, DOI:10.1016/j.addma.2019.05.031
.
- Q. Chen, E. Juste, M. Lasgorceix, F. Petit and A. Leriche, Open Ceram., 2022, 9, 100218, DOI:10.1016/j.oceram.2022.100218
.
- S. Henry, A. Samaro, F. H. Marchesini, B. Shaqour, J. Macedo, V. Vanhoorne and C. Vervaet, Int. J. Pharm., 2021, 598, 120361, DOI:10.1016/j.ijpharm.2021.120361
.
- H. Yang, Z. Shan, D. Yan, J. Shi, H. Shi and J. Huang, Addit. Manuf. Front., 2024, 3(2), 200138, DOI:10.1016/j.amf.2024.200138
.
- Z. Zhao, R. Liu, J. Chen and X. Xiong, J. Mater. Res. Technol., 2024, 29, 4394–4405, DOI:10.1016/j.jmrt.2024.02.148
.
- I. Seoane-Viaño, T. Pérez-Ramos, J. Liu, P. Januskaite, E. Guerra-Baamonde, J. González-Ramírez, M. Vázquez-Caruncho, A. W. Basit and A. Goyanes, J. Controlled Release, 2024, 365, 348–357, DOI:10.1016/j.jconrel.2023.11.022
.
- A. Derossi, R. Caporizzi, D. Azzollini and C. Severini, J. Food Eng., 2018, 220, 65–75, DOI:10.1016/j.jfoodeng.2017.05.015
.
- M. S. Ramalingam, K. N. Chaithanya Kumar, S. Sharma, S. S. Joshi and N. B. Dahotre, Virtual Phys. Prototyp., 2025, 20(1), e2547091, DOI:10.1080/17452759.2025.2547091
.
- G. Karabulut, G. Goksen and A. Mousavi Khaneghah, J. Agric. Food Res., 2024, 15, 101017, DOI:10.1016/j.jafr.2024.101017
.
- Y. Zhao, Y. Cui, Y. Hasebe, H. Bian, K. Yamanaka, K. Aoyagi, T. Hagisawa and A. Chiba, Powder Technol., 2021, 393, 482–493, DOI:10.1016/j.powtec.2021.08.006
.
- D. Sivan, K. Satheesh Kumar, A. Abdullah, V. Raj, I. I. Misnon, S. Ramakrishna and R. Jose, J. Mater. Sci., 2024, 59(7), 2602–2643, DOI:10.1007/s10853-024-09379-w
.
- J. Zegzulka, D. Gelnar, L. Jezerska, R. Prokes and J. Rozbroj, Sci. Rep., 2020, 10(1), 21004, DOI:10.1038/s41598-020-77974-3
.
- W. Du, T. Hu, Q. MacKenzie and D. Singh, J. Manuf. Process., 2025, 147, 100–111, DOI:10.1016/j.jmapro.2025.04.095
.
- S. S. Gaikwad, A. M. Kothule, Y. Y. Morade, S. S. Patil, U. D. Laddha, S. J. Kshirsagar and K. S. Salunkhe, Int. J. Pharm., 2023, 635, 122699, DOI:10.1016/j.ijpharm.2023.122699
.
- W. Nan, M. Pasha, U. Zafar, S. Nadimi, W. P. Goh and M. Ghadiri, Powder Technol., 2024, 436, 119471, DOI:10.1016/j.powtec.2024.119471
.
- D. Kong, J. Chen, H. Wang and Y. He, Int. J. Agric. Biol. Eng., 2023, 16(6), 263–272, DOI:10.25165/j.ijabe.20231606.7089
.
- H. M. Beakawi Al-Hashemi and O. S. Baghabra Al-Amoudi, Powder Technol., 2018, 330, 397–417, DOI:10.1016/j.powtec.2018.02.003
.
- K. Fang, D. Zhang, D. Wang, Z. Liu, M. Zhang and S. Zhang, J. Build. Eng., 2023, 69, 106237, DOI:10.1016/j.jobe.2023.106237
.
- A. Mussatto, R. Groarke, A. O’Neill, M. A. Obeidi, Y. Delaure and D. Brabazon, Addit. Manuf., 2021, 38, 101807, DOI:10.1016/j.addma.2020.101807
.
- S. Schiano, L. Chen and C.-Y. Wu, Powder Technol., 2018, 337, 78–83, DOI:10.1016/j.powtec.2017.08.064
.
- C. E. Collins, M. Pasha and T. A. Bell, Powder Technol., 2025, 453, 120648, DOI:10.1016/j.powtec.2025.120648
.
- J. Márquez-Barja, C. T. Calafate, J.-C. Cano and P. Manzoni, Comput. Commun., 2011, 34(8), 985–997, DOI:10.1016/j.comcom.2010.11.010
.
- F. Dini, S. A. Ghaffari, J. Jafar, R. Hamidreza and S. Marjan, Met. Powder Rep., 2020, 75(2), 95–100, DOI:10.1016/j.mprp.2019.05.001
.
- M. A. Bae, K. H. Kim and J. H. Baek, Int. J. Metalcast., 2023, 17(4), 2500–2507, DOI:10.1007/s40962-023-01014-9
.
- A. Bąk and W. Podgórska, Colloids Surf., A, 2016, 504, 414–425, DOI:10.1016/j.colsurfa.2016.05.091
.
- L. Bai, F. Liu, X. Xu, S. Huan, J. Gu and D. J. McClements, J. Food Eng., 2017, 207, 35–45, DOI:10.1016/j.jfoodeng.2017.03.021
.
- A. Mostafaei, P. Rodriguez De Vecchis, M. J. Buckenmeyer, S. R. Wasule, B. N. Brown and M. Chmielus, Mater. Sci. Eng., C, 2019, 102, 276–288, DOI:10.1016/j.msec.2019.04.011
.
- W. Zhao, X. Qin, K. Feng, K. Zou, H. Nan, J. Wu, X. Qie and Q. Wei, J. Mater. Res. Technol., 2025, 3071–3084, DOI:10.1016/j.jmrt.2025.12.163
.
- Y. Liu, S. Cao and E. Yilmaz, Powder Technol., 2026, 471, 122102, DOI:10.1016/j.powtec.2025.122102
.
- S. S. Butt, I. K. Mohammed, V. Raghavan, J. Osborne, H. Powell and M. N. Charalambides, J. Food Eng., 2018, 238, 112–121, DOI:10.1016/j.jfoodeng.2018.05.039
.
- Y. Zhong, B. Wang, W. Lv, Y. Wu, Y. Lv and S. Sheng, Food Chem., 2024, 458, 140294, DOI:10.1016/j.foodchem.2024.140294
.
- S. C. Altıparmak and S. I. C. Daminabo, Addit. Manuf. Front., 2024, 3(1), 200106, DOI:10.1016/j.amf.2023.200106
.
- X. Wang, M. Zhang, A. S. Mujumdar and J. Li, Food Res. Int., 2023, 164, 112404, DOI:10.1016/j.foodres.2022.112404
.
- D. A. Rau, M. J. Bortner and C. B. Williams, Addit. Manuf., 2023, 75, 103745, DOI:10.1016/j.addma.2023.103745
.
- R. Karyappa, D. Zhang, Q. Zhu, R. Ji, A. Suwardi and H. Liu, Addit. Manuf., 2024, 79, 103903, DOI:10.1016/j.addma.2023.103903
.
- A. T. Tyowua, D. Harbottle and B. P. Binks, Adv. Colloid Interface Sci., 2024, 332, 103274, DOI:10.1016/j.cis.2024.103274
.
- Y. Ma and L. Zhang, Curr. Opin. Food Sci., 2022, 44, 100803, DOI:10.1016/j.cofs.2021.12.012
.
- T. Funami, S. Ishihara, K. Maeda and M. Nakauma, Food Hydrocoll., 2025, 162, 110901, DOI:10.1016/j.foodhyd.2024.110901
.
- M. G. Bedoya, D. R. Montoya, G. Tabilo-Munizaga, M. Pérez-Won and R. Lemus-Mondaca, Trends Food Sci. Technol., 2022, 128, 38–52, DOI:10.1016/j.tifs.2022.05.013
.
- M. A. Sourov, S. I. Emu, M. S. H. Thakur, M. M. Ikram, A. Morshed and M. M. Rahman, Sci. Rep., 2025, 15(1), 9842, DOI:10.1038/s41598-024-74058-4
.
- P. Thareja, S. Swarupa, S. Ahmad and M. E. Jinugu, Curr. Opin. Colloid Interface Sci., 2025, 77, 101918, DOI:10.1016/j.cocis.2025.101918
.
- J. Pan, X. Chen, J. Liu, Z. Wang, T. Aziz, M. Alhomrani, W. F. Alsanie, A. S. Alamri, H. Cui and L. Lin, Innov. Food Sci. Emerg. Technol., 2025, 106, 104257, DOI:10.1016/j.ifset.2025.104257
.
- Y. Ma, M. A. I. Schutyser, R. M. Boom and L. Zhang, Innov. Food Sci. Emerg. Technol., 2022, 81, 103135, DOI:10.1016/j.ifset.2022.103135
.
- J. Wang, Q. Jiang, Z. Huang, A. H. Muhammad, A. Gharsallaoui, M. Cai, K. Yang and P. Sun, Food Hydrocoll., 2024, 153, 109942, DOI:10.1016/j.foodhyd.2024.109942
.
- Y. Wang, C. Bai, D. J. McClements, X. Xu, Q. Sun, B. Jiao, Q. Wang and L. Dai, Food Hydrocoll., 2023, 145, 109097, DOI:10.1016/j.foodhyd.2023.109097
.
- D. De Angelis, A. J. van der Goot, A. Pasqualone and C. Summo, Curr. Opin. Food Sci., 2024, 58, 101192, DOI:10.1016/j.cofs.2024.101192
.
- F. Aghababaei, D. J. McClements, M. Pignitter and M. Hadidi, Food Chem.: X, 2025, 26, 102280, DOI:10.1016/j.fochx.2025.102280
.
- R. Wang, C. Y. Su, D. Li and L. J. Wang, Food Chem., 2025, 495(3), 146512, DOI:10.1016/j.foodchem.2025.146512
.
- H. Wijshoff, Curr. Opin. Colloid Interface Sci., 2018, 36, 20–27, DOI:10.1016/j.cocis.2017.11.004
.
- E. Enríquez, J. J. Reinosa, V. Fuertes and J. F. Fernández, Ceram. Int., 2022, 48(21), 31080–31101, DOI:10.1016/j.ceramint.2022.07.155
.
- B. Derby, Annu. Rev. Mater. Res., 2010, 40(1), 395–414, DOI:10.1146/annurev-matsci-070909-104502
.
- L. Hakim, R. K. Deshmukh, Y. S. Lee and K. K. Gaikwad, Sustainable Food Technol., 2024, 2(4), 876–892, 10.1039/d4fb00036f
.
- H. Gudapati, M. Dey and I. Ozbolat, Biomaterials, 2016, 102, 20–42, DOI:10.1016/j.biomaterials.2016.06.012
.
- J. Wang and G. T. C. Chiu, Mechatronics, 2023, 94, 103031, DOI:10.1016/j.mechatronics.2023.103031
.
- Y. Wu, Y. Zhang, M. Yan, G. Hu, Z. Li, W. He, X. Wang, A. Abulimit and R. Li, Appl. Mater. Today, 2024, 36, 102036, DOI:10.1016/j.apmt.2023.102036
.
- S. Chen and X. Luo, Appl. Food Res., 2024, 4(2), 100459, DOI:10.1016/j.afres.2024.100459
.
- D. Zhao, H. Zhou, Y. Wang, J. Yin and Y. Huang, Addit. Manuf., 2021, 48, 102451, DOI:10.1016/j.addma.2021.102451
.
- E. Parry, D.-J. Kim, A. A. Castrejón-Pita, S. J. Elston and S. M. Morris, Opt. Mater., 2018, 80, 71–76, DOI:10.1016/j.optmat.2018.04.038
.
- J. Zhang, H. Zhu, D. Liu, Y. Li and C. Huang, Mater. Today Commun., 2024, 41, 110866, DOI:10.1016/j.mtcomm.2024.110866
.
- T. Karbowiak, F. Debeaufort and A. Voilley, Crit. Rev. Food Sci. Nutr., 2006, 46(5), 391–407, DOI:10.1080/10408390591000884
.
- B. Han, G. Y. Yun, J. W. Boley, S. H. Kim, J. Y. Hwang, G. T. C. Chiu and K. Park, Int. J. Heat Mass Transfer, 2016, 97, 15–25, DOI:10.1016/j.ijheatmasstransfer.2016.01.030
.
- M. Upadhyay, T. Sivarupan and M. El Mansori, J. Manuf. Process., 2017, 29, 211–220, DOI:10.1016/j.jmapro.2017.07.017
.
- T. Ceyhan, G. S. Tomar and A. Can Karaca, Colloids Surf., B, 2025, 253, 114691, DOI:10.1016/j.colsurfb.2025.114691
.
- A. Badjona, B. Cherono, R. Bradshaw and B. Dubey, Food Hydrocoll., 2025, 162, 110997, DOI:10.1016/j.foodhyd.2024.110997
.
- O. K. Ozturk, A. M. Salgado, D. R. Holding, O. H. Campanella and B. R. Hamaker, Food Hydrocoll., 2023, 134, 108044, DOI:10.1016/j.foodhyd.2022.108044
.
- C. Hollestelle, L. Divoux, C. Michon and D. Huc-Mathis, Food Hydrocoll., 2024, 157, 110348, DOI:10.1016/j.foodhyd.2024.110348
.
- X. Lv, F. Ye, L. Cheng, S. Fan and Y. Liu, Ceram. Int., 2019, 45(10), 12609–12624, DOI:10.1016/j.ceramint.2019.04.012
.
- E. Chadwick, A. H. Barrett, M. Okamoto, Y. Suleiman, G. P. S. R. Bertola, S. Shahbazmohamadi, A. Shetty, Y. Li and A. W. K. Ma, J. Food Eng., 2024, 378, 112112, DOI:10.1016/j.jfoodeng.2024.112112
.
- Y. Pei, S. Yan, Y. Liao, B. Qi, Y. Huang and Y. Li, Food Res. Int., 2025, 207, 116056, DOI:10.1016/j.foodres.2025.116056
.
- S. J. Ye, H. J. Park and M. Y. Baik, Food Sci. Biotechnol., 2025, 34(2), 349–363, DOI:10.1007/s10068-024-01705-x
.
- S. Liao, J. Chen, C. Huang, C. Gao, L. Peng and Z. Huang, J. Eur. Ceram. Soc., 2025, 45(6), 117167, DOI:10.1016/j.jeurceramsoc.2024.117167
.
- Y. Wang, A. Müllertz and J. Rantanen, Eur. J. Pharm. Sci., 2022, 178, 106280, DOI:10.1016/j.ejps.2022.106280
.
- Z. Yang, Y. Zhang and W. Yan, Acta Mater., 2023, 260, 119298, DOI:10.1016/j.actamat.2023.119298
.
- M. Kozakiewicz-Latała, K. P. Nartowski, A. Dominik, K. Malec, A. M. Gołkowska, A. Złocińska, M. Rusińska, P. Szymczyk-Ziółkowska, G. Ziółkowski, A. Górniak and B. Karolewicz, Eur. J. Pharm. Biopharm., 2022, 170, 144–159, DOI:10.1016/j.ejpb.2021.11.001
.
- T. Cheny, C. Colin and B. Verquin, Addit. Manuf., 2024, 87, 104231, DOI:10.1016/j.addma.2024.104231
.
- S. R. Jaggannagari, W. H. Kan, L. N. S. Chiu, G. Proust, A. Huang, Y. Gan and R. K. Annabattula, Addit. Manuf., 2025, 97, 104571, DOI:10.1016/j.addma.2024.104571
.
- T. Toniazzo, A. M. Fioroto, E. da Silva Araújo and C. C. Tadini, Dry. Technol., 2023, 41(10), 1651–1662, DOI:10.1080/07373937.2023.2172028
.
- Z. Zhao, W. Liu, J. Ren, C. Wang, Y. He, X. Zhang and X. Chen, J. Manuf. Syst., 2024, 72, 59–73, DOI:10.1016/j.jmsy.2023.11.006
.
- A. Vasilenko, S. Koynov, B. J. Glasser and F. J. Muzzio, Powder Technol., 2013, 239, 366–373, DOI:10.1016/j.powtec.2013.02.011
.
- L. Yang, S. Wang, H. Zhang, C. Du, S. Li and J. Yang, LWT, 2022, 167, 113834, DOI:10.1016/j.lwt.2022.113834
.
- J. Li, Z. Wang, H. Xiu, X. Zhao, F. Ma, L. Liu, C. Yi, M. Zhang, E. Kozliak and Y. Ji, Powder Technol., 2022, 399, 117194, DOI:10.1016/j.powtec.2022.117194
.
- M. Liu, X. Yang, Z. Liu and B. Li, Powder Technol., 2025, 449, 120420, DOI:10.1016/j.powtec.2024.120420
.
- L. Chen, W. Chen, S. Zhang, S. Zou, T. Cheng and D. Zhu, J. Mater. Res. Technol., 2023, 27, 4043–4052, DOI:10.1016/j.jmrt.2023.10.203
.
- S. Cao, F. Xiang, S. Li, X. Ma, H. Hu, Q. Guo, B. Jiao, D. Agyei, Q. Wang and A. Shi, LWT, 2024, 206, 116596, DOI:10.1016/j.lwt.2024.116596
.
- K. Zhao, Z. Su, Z. Ye, W. Cao, J. Pang, X. Wang, Z. Wang, X. Xu and J. Zhu, J. Mater. Res. Technol., 2023, 27, 5449–5469, DOI:10.1016/j.jmrt.2023.11.045
.
- S. Bafaluy Ojea, J. Torrents-Barrena, M. T. Pérez-Prado, R. Muñoz Moreno and F. Sket, J. Mater. Res. Technol., 2023, 23, 3974–3986, DOI:10.1016/j.jmrt.2023.02.051
.
- J. E. Lawrence, M. P. Lawrence, K. Fezzaa, S. J. Clark and N. B. Crane, Addit. Manuf., 2024, 89, 104269, DOI:10.1016/j.addma.2024.104269
.
- D. Lin, A. L. Kelly and S. Miao, Trends Food Sci. Technol., 2020, 102, 123–137, DOI:10.1016/j.tifs.2020.05.024
.
- R. Wu, J. Jiang, F. An, X. Ma and J. Wu, Trends Food Sci. Technol., 2024, 149, 104545, DOI:10.1016/j.tifs.2024.104545
.
- Y. Sun, Z.-H. Yi, T. Shen, H.-W. Xiong, X. Kang, L. Zhang and K.-C. Zhou, Trans. Nonferrous Met. Soc. China, 2024, 34(7), 2108–2119, DOI:10.1016/s1003-6326(24)66528-3
.
- N. I. A. Pereira, M. D. S. Oliveira, B. C. C. Reis, B. L. Nascimento, C. R. Carneiro, T. R. Arruda, E. N. R. Vieira and B. R. D. C. Leite Junior, Food Res. Int., 2024, 192, 114849, DOI:10.1016/j.foodres.2024.114849
.
- H. Chen, D. Fang, X. Wang, Y. Gong, Y. Ji and H. Pan, Int. J. Pharm., 2024, 665, 124668, DOI:10.1016/j.ijpharm.2024.124668
.
- M. Zhu, H. Wang, T. Sun, J. Lv, K. Wang, S. Huan, Z. Li, Y. Liu, S. Liu, D. J. McClements and L. Bai, Food Hydrocoll., 2025, 162, 110943, DOI:10.1016/j.foodhyd.2024.110943
.
- J. Yu, X.-Y. Wang, D. Li, L.-J. Wang and Y. Wang, Food Hydrocoll., 2022, 131, 107824, DOI:10.1016/j.foodhyd.2022.107824
.
- T. Feng, X. Wang, X. Wang, X. Zhang, Y. Gu, S. Xia and Q. Huang, Food Chem., 2021, 350, 129251, DOI:10.1016/j.foodchem.2021.129251
.
- C. Wu, Z. Liu, L. Zhi, B. Jiao, H. Hu, X. Ma, J. Zhu, M. Pignitter, Q. Wang and A. Shi, Food Hydrocoll., 2023, 144, 109012, DOI:10.1016/j.foodhyd.2023.109012
.
- H. Li, W. Wang, N. Li, Z. Liu, D. Xu, H. Pu, W. Liang, H. Mo and L. Hu, J. Food Eng., 2023, 357, 111607, DOI:10.1016/j.jfoodeng.2023.111607
.
- P. Zhu, Y. Chu, J. Yang and L. Chen, Food Hydrocoll., 2024, 157, 110391, DOI:10.1016/j.foodhyd.2024.110391
.
- S. Gao and X. Qiu, Colloids Surf., A, 2025, 705, 135742, DOI:10.1016/j.colsurfa.2024.135742
.
- J. Zhao, B. Chang, J. Wen, Y. Fu, Y. Luo, J. Wang, Y. Zhang and X. Sui, Food Chem., 2025, 468, 142462, DOI:10.1016/j.foodchem.2024.142462
.
- W. Wei, L. Cui and Z. Meng, Int. J. Biol. Macromol., 2025, 287, 138637, DOI:10.1016/j.ijbiomac.2024.138637
.
- K. Zhao, Y. Hao, X. Guo, Y. Chang and X. Shen, Int. J. Biol. Macromol., 2024, 280, 135648, DOI:10.1016/j.ijbiomac.2024.135648
.
- K. Zhao, Y. Hao, X. Guo, Y. Chang and X. Shen, Food Hydrocoll., 2024, 280, 135648, DOI:10.1016/j.ijbiomac.2024.135648
.
- C. Chao, J. H. Lee, I. W. Kim, R. Y. Choi, H. W. Kim and H. J. Park, Food Biosci., 2023, 54, 102924, DOI:10.1016/j.fbio.2023.102924
.
- M. Murugan, S. K. Ramasamy, G. Venkatesan, J. Lee, S. Barathi, S. Kandasamy and P. K. Sarangi, Food Chem., 2024, 459, 140348, DOI:10.1016/j.foodchem.2024.140348
.
- T. Mehta, H. Aziz, K. Sen, S.-Y. Chang, V. Nagarajan, A. W. K. Ma and B. Chaudhuri, Int. J. Pharm., 2024, 656, 124037, DOI:10.1016/j.ijpharm.2024.124037
.
- M. Özkan, K. Dimic-Misic, A. Karakoc, S. G. Hashmi, P. Lund, T. Maloney and J. Paltakari, Org. Electron., 2016, 38, 307–315, DOI:10.1016/j.orgel.2016.09.001
.
- A. K. Tamo, L. D. W. Djouonkep and N. B. S. Selabi, Int. J. Biol. Macromol., 2024, 270, 132123, DOI:10.1016/j.ijbiomac.2024.132123
.
- G. Gao, T. Yonezawa, K. Hubbell, G. Dai and X. Cui, Biotechnol. J., 2015, 10(10), 1568–1577, DOI:10.1002/biot.201400635
.
- N. Bu, H. Lin, D. Zhang, X. Chen, P. Tan, Y. Zhong, L. Huang, J. Pang, L. Wang and R. Mu, J. Food Eng., 2025, 387, 112325, DOI:10.1016/j.jfoodeng.2024.112325
.
- Q. Wang, T. Chen, Y. Zhu, Y. Ning, Y. Li, S. Yan and B. Qi, Food Res. Int., 2025, 201, 115643, DOI:10.1016/j.foodres.2024.115643
.
- L. Li, Y. Liu, X. Tan, F. Teng and Y. Li, Carbohydr. Polym., 2025, 349, 122959, DOI:10.1016/j.carbpol.2024.122959
.
- D. Odelli, S. R. Rodrigues, L. Silva de Sousa, N. F. Nogueira Silva, A. L. López Martínez, L. S. Queiroz, F. Casanova and A. F. d. Carvalho, Food Hydrocoll., 2024, 156, 110310, DOI:10.1016/j.foodhyd.2024.110310
.
- R. Boni, E. A. Blackburn, D.-J. Kleinjan, M. Jonaitis, F. Hewitt-Harris, M. Murdoch, S. Rosser, D. C. Hay and L. Regan, J. Struct. Biol., 2023, 215(3), 107981, DOI:10.1016/j.jsb.2023.107981
.
- M. Zhao, P. Han, H. Mu, S. Sun, J. Dong, J. Sun, S. Lu, Q. Wang and H. Ji, Food Chem.: X, 2025, 102174, DOI:10.1016/j.fochx.2025.102174
.
- M. A. Saleem, X. Xiao, S. Mamoon, G. Li and T. Liu, Precis. Eng., 2025, 92, 63–76, DOI:10.1016/j.precisioneng.2024.11.012
.
- Y. Zhou, L. Qi, H. Lian, J. Luo and L. Su, Addit. Manuf., 2024, 84, 104088, DOI:10.1016/j.addma.2024.104088
.
- H.-Y. Jun, S.-J. Kim and C.-H. Choi, Nanomaterials, 2021, 11(12), 3441, DOI:10.3390/nano11123441
.
- X. Wang, H. Tian, H. Guo, C. Yan, H. Ran, R. Zhang, C. Wang, X. Li, X. Chen and J. Shao, Addit. Manuf., 2024, 79, 103940, DOI:10.1016/j.addma.2023.103940
.
- H. Zhang, J. Hui, L. Jingxiang, C.-H. Lee, Z. Yan, J. j. Wang, L. Guo and Z. Xu, J. Mater. Res. Technol., 2023, 24, 4405–4426, DOI:10.1016/j.jmrt.2023.04.059
.
- W. L. Ng, K. Sachdeva, P. L. Conway, S. Wuertz and P. Bartolo, J. Future Foods, 2025 DOI:10.1016/j.jfutfo.2025.07.031
.
- S. David, I. Ianovici, G. Guterman Ram, Y. Shaulov Dvir, N. Lavon and S. Levenberg, Adv. Sustain. Syst., 2024, 8(6), 2300499, DOI:10.1002/adsu.202300499
.
- B. A. S. Dagès, J. A. Fabian, D. Polakova, M. Rysova, P. D. Topham, J.-B. R. G. Souppez, M. P. Hanga and E. Theodosiou, Food Bioprod. Process., 2025, 149, 118–129, DOI:10.1016/j.fbp.2024.11.012
.
- R. Eslami, A. Azizi and M. Najafi, Polym. Polym. Compos., 2020, 29(5), 436–443, DOI:10.1177/0967391120923440
.
- H. Chu, W. Yang, L. Sun, S. Cai, R. Yang, W. Liang, H. Yu and L. Liu, Micromachines, 2020, 11(9), 796, DOI:10.3390/mi11090796
.
- O. Parizel, C. Sulmont-Rossé, G. Fromentin, J. Delarue, H. Labouré, R. Benamouzig and A. Marsset-Baglieri, Appetite, 2016, 104, 44–51, DOI:10.1016/j.appet.2015.11.018
.
- Y. Lu, R. Rai and N. Nitin, Food Res. Int., 2023, 173, 113384, DOI:10.1016/j.foodres.2023.113384
.
- K. Schüler, D. M. C. Marques, A. Gusmão, M. Jabouille, M. Leite, J. M. S. Cabral, P. Sanjuan-Alberte and F. C. Ferreira, Food Hydrocoll., 2024, 157, 110369, DOI:10.1016/j.foodhyd.2024.110369
.
- T. Ben-Arye, Y. Shandalov and S. Ben-Shaul,
et al.
, Nat Food, 2020, 1, 210–220, DOI:10.1038/s43016-020-0046-5
.
- M. Jung, Y. Lee, S. O. Han and J. E. Hyeon, J. Microbiol. Biotechnol., 2024, 34(5), 994–1002, DOI:10.1016/j.plana.2024.100127
.
- Z. Guo, B. Yang, N. Liang, X. Huang, J. Shi, Z. Li, P. Paximada and Z. Xiaobo, J. Food Eng., 2025, 386, 112258, DOI:10.1016/j.jfoodeng.2024.112258
.
- Y. Cheng, Y. Fu, L. Ma, P. L. Yap, D. Losic, H. Wang and Y. Zhang, Food Hydrocoll., 2022, 132, 107855, DOI:10.1016/j.foodhyd.2022.107855
.
- U. Shashikumar, A. Saraswat, K. Deshmukh, C. M. Hussain, P. Chandra, P.-C. Tsai, P.-C. Huang, Y.-H. Chen, L.-Y. Ke, Y.-C. Lin, S. Chawla and V. K. Ponnusamy, Adv. Colloid Interface Sci., 2024, 328, 103163, DOI:10.1016/j.cis.2024.103163
.
- M. G. Dancausa Millán and M. G. Millán Vázquez de la Torre, Int. J. Gastron. Food Sci., 2024, 37, 100963, DOI:10.1016/j.ijgfs.2024.100963
.
- S. P. Sarathy, H. Ravikumar, P. Nanjan, N. Alagesan and B. L. Chua, Int. J. Biol. Macromol., 2025, 321, 146465, DOI:10.1016/j.ijbiomac.2025.146465
.
- J. de Boer and H. Aiking, Ecol. Econ., 2011, 70(7), 1259–1265, DOI:10.1016/j.ecolecon.2011.03.001
.
- A. Hoehnel, E. Zannini and E. K. Arendt, Trends Food Sci. Technol., 2022, 128, 238–252, DOI:10.1016/j.tifs.2022.08.007
.
- Y. Liu, Y. Yu, C. Liu, J. M. Regenstein, X. Liu and P. Zhou, LWT, 2019, 102, 338–346, DOI:10.1016/j.lwt.2018.12.053
.
- A. R. T. Cirunay, C. Barry-Ryan and O. McAuliffe, Crit. Rev. Food Sci. Nutr., 2025, 1–25, DOI:10.1080/10408398.2025.2560680
.
- M. Broeders, P. Herrero-Hernandez, M. P. T. Ernst, A. T. van der Ploeg and W. W. M. P. Pijnappel, iScience, 2020, 23(1), 100789, DOI:10.1016/j.isci.2019.100789
.
- P. Zeng, H. Wang, P. Zhang and S. S. Y. Leung, Biotechnol. Adv., 2024, 73, 108371, DOI:10.1016/j.biotechadv.2024.108371
.
- J. Koukara and K. K. Papadopoulou, Biochem. Biophys. Res. Commun., 2023, 654, 55–61, DOI:10.1016/j.bbrc.2023.02.061
.
- S. R. Lokireddy, S. R. Kunchala and R. Vadde, World J. Microbiol. Biotechnol., 2025, 41(3), 90, DOI:10.1007/s11274-025-04302-0
.
- G. Galili and R. Amir, Plant Biotechnol. J., 2012, 11(2), 211–222, DOI:10.1111/pbi.12025
.
- M. D'Este, M. Alvarado-Morales and I. Angelidaki, Biotechnol. Adv., 2018, 36(1), 14–25, DOI:10.1016/j.biotechadv.2017.09.001
.
- R. Maldonado-Rosas, M. Alfaro-Ponce, E. Cuan-Urquizo and V. Tejada-Ortigoza, J. Food Eng., 2025, 395, 112534, DOI:10.1016/j.jfoodeng.2025.112534
.
| This journal is © The Royal Society of Chemistry 2026 |