Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Alternative proteins from waste streams: a circular economy roadmap for sustainable food systems
Sunny
Dhiman
a,
Gunjan
Mukherjee
 *a,
Anu
Kumar
a and
Bhanu
Krishan
b
*a,
Anu
Kumar
a and
Bhanu
Krishan
b
aDepartment of Biotechnology, University Institute of Biotechnology, Chandigarh University, Mohali-140413, Punjab, India. E-mail: gunjanmukherjee@gmail.com
bDepartment of Biotechnology, Himachal Pradesh University, Summerhill, Shimla, Himachal Pradesh 171005, India
First published on 24th February 2026
Abstract
The escalating global demand for sustainable proteins calls for a paradigm shift from conventional animal- and plant-based production toward innovative, resource-efficient alternatives. This review delineates a circular economy roadmap for deriving alternative proteins from agri-food residues, industrial by-products, and organic waste streams. It systematically examines microbial, algal, fungal, and insect-based conversion systems, highlighting their bioprocess efficiencies, nutritional potential, technology readiness, and environmental performance. Emerging technologies such as enzymatic hydrolysis, fermentation biorefineries, and integrated waste-to-protein platforms are discussed as scalable approaches for valorizing waste into high-quality protein for food and feed applications. The study emphasizes that these waste-derived protein systems not only mitigate greenhouse gas emissions, land use, and water consumption but also enhance food security and resource circularity. Key challenges including safety assurance, contaminant control, regulatory barriers, and consumer acceptance, are critically analyzed alongside enabling policy, innovation, and scale-up strategies. By redefining waste as a bioresource, this review advances the vision of a regenerative, low-carbon, and nutritionally secure global food system.
Sustainability spotlightThis review delineates a circular economy roadmap for deriving alternative proteins from agri-food residues, industrial by-products, and organic waste streams. The development and adoption of alternative proteins from waste streams can contribute to the achievement of several Sustainable Development Goals (SDGs), providing a pathway towards a more sustainable and equitable food system. These encompass SDG 2 (Zero Hunger), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), highlighting the multifaceted benefits of alternative proteins in addressing global challenges. By providing sustainable protein sources, reducing environmental impacts, and promoting resource efficiency, alternative proteins can help achieve a more sustainable and equitable food system that supports both human nutrition and ecological health. |
1. Introduction
The global food system faces an unprecedented challenge as the world population is projected to reach 9.7 billion by 2050, necessitating a 70% increase in food production to meet growing nutritional demands.1,2 Protein, as an essential macronutrient crucial for human health and development, represents a particularly critical component of this challenge. With mounting pressure on natural resources, the supply of high-value protein has become increasingly limited.3 The growing awareness of healthy food choices has further propelled the demand for protein needed to adequately nourish the world's population. This demographic surge places enormous pressure on existing food production systems, specifically those responsible for supplying adequate and high-quality protein to meet nutritional requirements. Meeting this protein demand is a fundamental challenge, given the essential role that proteins play in human health as building blocks for tissue repair, growth, and immune function.Conventional animal-based protein production faces increasing scrutiny owing to its significant environmental footprint, various health concerns, diseases, and ethical considerations.4 Animal protein production is anticipated to increase by approximately 50–73% by 2050 to meet the demands of a rapidly growing and economically advancing global population.5 The World Health Organization (WHO) has emphasized the urgent need to substitute animal-based proteins with sustainable alternative protein sources.6
Animal agriculture is resource-intensive, linked to considerable greenhouse gas emissions, land degradation, and water use. For example, the production of 1 tonne of beef protein can emit between 45 and 640 tonnes of CO2-equivalents, use 1.5 million litres of water, and require over 200 hectares of land annually.7 The livestock sector accounts for roughly 14–18% of anthropogenic greenhouse gas emissions globally.8
Ruminant-based production, particularly beef, is a major emitter due to methane output and inefficient feed conversion, whereas poultry and pork systems, while less intensive, still require significant farmland for feed crops such as soy and maize.9 Plant-based proteins (e.g., cereals and legumes) are generally more resource-efficient and produce lower emissions than animal-sourced proteins. For instance, shifting towards plant-based diets in the United States could reduce arable land use by 51 million hectares globally.8 However, the production of protein-rich crops remains susceptible to climatic stresses, with projections indicating that legume yields could drop 10–49% in sensitive areas in certain climate scenarios if no adaptive measures are taken.10
There are also challenges linked to the nutrient density and protein quality of plant sources. Many staple plant proteins are often deficient in key amino acids such as lysine, and their digestibility/Protein Digestibility-Corrected Amino Acid Score (PDCAAS) (0.54–0.57) for unfermented barley/rice is lower than those of animal proteins.11 These factors underscore the necessity to diversify protein sources beyond conventional agriculture.
The convergence of environmental pressures, technological advances, and changing consumer preferences has catalysed the emergence of alternative protein sources as a transformative solution for future food systems. Alternative proteins, encompassing plant-based proteins, cultivated meat, fermentation-derived proteins, and novel protein sources such as insects and algae, offer the potential to decouple protein production from many of the negative externalities associated with conventional animal agriculture.12
According to the latest report by Global Market Insights Inc., the global alternative protein market was valued at USD 35.6 billion in 2024 and is projected to increase from USD 40.4 billion in 2025 to USD 130.1 billion by 2034, registering a compound annual growth rate (CAGR) of 13.9% (Fig. 1).13
This growth is driven by technological breakthroughs that have significantly improved the taste, texture, and nutritional profiles of alternative protein products, making them increasingly competitive with conventional animal proteins.3 Major food companies and technology firms have invested heavily in alternative protein research and development, accelerating innovation and market adoption.
The integration of alternative proteins into existing food systems represents more than a simple substitution; it embodies a fundamental shift toward more sustainable, resilient, and ethical protein production paradigms. As demand rises and environmental constraints become critical, alternative proteins from agri-food residues, by-products, and industrial effluents have seen increasing interest. Waste streams from agri-food residues, industrial by-products, and effluents offer a rich, underutilized resource for protein recovery. Technologies to convert these residues into high-value protein products include processing of lignocellulosic biomass, single-cell proteins derived from microorganisms, insect proteins, and other microbial proteins cultivated on low-cost substrates. These approaches align with circular economy (CE) principles by utilizing waste as a resource, minimizing environmental impact, and fostering resource efficiency.14–17
This paper aims to provide a comprehensive roadmap on how CE strategies can transform waste streams into sustainable protein-rich resources, contributing significantly to resilient and ecologically sound food systems. Previous studies broadly discuss alternative proteins across diverse sources. However, the production of waste-derived alternative proteins remains less explored. This review uniquely focuses on waste-to-protein valorization within the framework of circular economy principles. It provides a critical evaluation of how waste-derived alternative protein production pathways can be integrated into food system sustainability, considering environmental, economic, and technological dimensions to outline a clearer path toward a sustainable protein supply.
By unveiling the intersections of waste valorization, protein innovation, and circular economy, this review contributes novel insights vital for policymakers, researchers, and industry stakeholders seeking sustainable solutions for future protein demands and food security challenges.
2. Circular economy framework for alternative proteins
The circular economy represents a paradigm shift from the traditional linear “take-make-dispose” model to a regenerative system where resource inputs, waste, emissions, and energy leakage are minimized by closing material and energy loops. In food systems, this translates to a holistic approach that aims to minimize waste, maximize resource utilization, and ensure that materials are kept in use for as long as possible through various strategies, enhancing overall sustainability.18At its core, the circular economy in food systems emphasizes minimizing waste, maximizing resource utilization, and closing material loops to ensure that resources are used efficiently and sustainably.19
By implementing strategies such as reducing food loss and waste, valorizing by-products, and designing food systems for resource efficiency, the circular economy aims to decouple economic activity from the consumption of finite resources and minimize environmental impacts.20 This approach not only reduces the strain on natural resources but also creates economic opportunities through the development of innovative technologies and business models focused on resource recovery and reuse. The emphasis on resource efficiency also promotes the development of more sustainable agricultural practices, such as precision farming and integrated pest management, which reduce the environmental footprint of food production.
A key aspect of the CE in food systems is the valorization of by-products and waste streams, transforming them from liabilities into valuable resources that can be reintegrated into the food system or used in other industries.21 This includes converting agricultural residues, food processing waste, and other organic waste into valuable products like alternative proteins, biofuels, fertilizers, and packaging materials.22 For instance, agricultural residues such as crop stalks and leaves can be used as feedstock for producing biofuels or as a substrate for growing edible mushrooms, while food processing waste like fruit and vegetable peels can be processed into valuable ingredients for food and cosmetic products. Valorization not only reduces waste but also creates new revenue streams for businesses and enhances the overall sustainability of food production by reducing the reliance on virgin resources. This approach supports the development of a more resilient and resource-efficient food system that minimizes environmental impacts and promotes economic growth.23
Alternative proteins, especially those sourced from waste streams, are ideally positioned as bio-based solutions within a circular bioeconomy, providing a sustainable and innovative means of addressing the increasing global demand for protein while concurrently minimizing environmental consequences.14 They present a sustainable substitute to conventional protein sources through the utilization of renewable resources and the reduction of environmental impacts.24,25
The production of alternative proteins from waste streams can significantly reduce the environmental footprint compared to traditional animal-based proteins, offering a more sustainable and resource-efficient approach for meeting global protein demands.15 It necessitates reduced land, water, and energy inputs, while also diminishing greenhouse gas emissions and pollutant release.25 By utilizing waste as a feedstock, alternative protein production can also contribute to waste reduction and resource conservation, creating a closed-loop system that minimizes environmental impacts.20 For instance, insect farming, which uses organic waste as feed for the insects, requires significantly less land and water compared to traditional livestock farming and produces fewer greenhouse gas emissions. Similarly, the production of single-cell proteins from agricultural residues can reduce the environmental burden associated with waste disposal while simultaneously creating a valuable protein source. This approach supports the development of a more sustainable and resilient food system that minimizes environmental impacts and promotes resource efficiency.
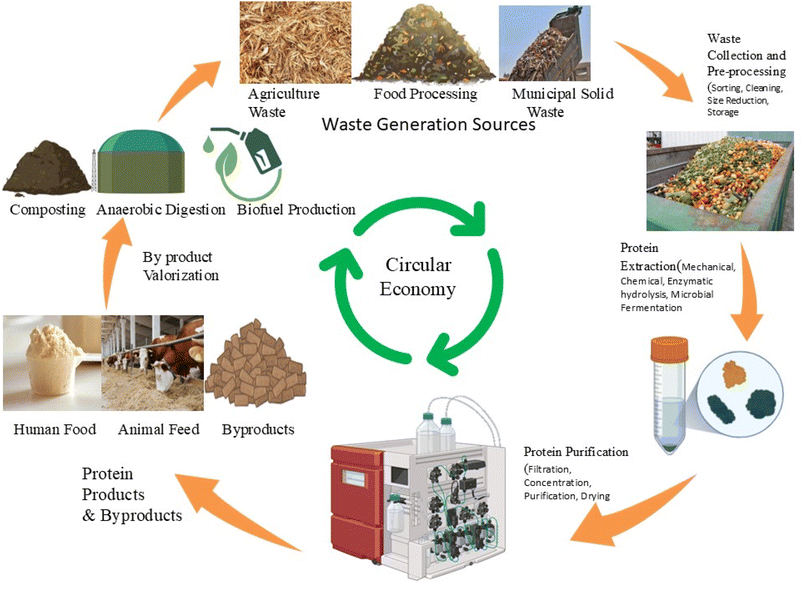
Several waste streams possess significant potential for protein recovery, making them valuable resources for alternative protein production within a circular economy framework.25 These include agricultural residues like crop residues and livestock manure, food processing waste such as fruit and vegetable peels and whey, and industrial effluents, each offering unique opportunities for protein extraction and valorization.25 Lignocellulosic biomass from agricultural and forestry residues stands out as an abundant and renewable feedstock for single-cell protein production, offering a sustainable alternative to conventional protein sources.25 Additionally, agro-industrial by-products, such as citric and yeast waste, can be fermented to produce protein-rich feed ingredients, further highlighting the potential of waste streams to contribute to a more sustainable food system.16 By identifying and utilizing these key waste streams, it is possible to create a more resource-efficient and environmentally friendly approach to protein production. Fig. 2 presents a waste to protein conversion model of the circular economy framework.
The circular bioeconomy emphasizes the efficient and sustainable use of biological resources, including waste materials, to create value-added products and services. By integrating alternative protein production into this framework, it is possible to create a more resilient and environmentally friendly food system that supports both human nutrition and ecological health. This approach also fosters innovation in agricultural practices, waste management, and food processing, leading to the development of new technologies and business models that contribute to a more sustainable economy.
3. Waste-to-protein conversion systems
The escalating global demand for sustainable protein sources necessitates innovative approaches to decouple protein production from traditional, resource-intensive agricultural practices. Waste streams, especially organic wastes arising from agricultural, industrial, and municipal sources, represent abundant and renewable raw materials that can be valorized into alternative proteins through biotechnological and biological processes. By integrating these substrates into production systems, it is possible to close nutrient loops, reduce reliance on finite resources, and lower greenhouse gas emissions.26The transformation of waste streams into alternative proteins requires a comprehensive understanding of substrate characteristics, conversion pathways, and recovery potentials. Different waste categories exhibit varying nutritional profiles, processing requirements, and scalability potential, necessitating targeted approaches for optimal protein recovery. Table 1 highlights the composition of different waste streams.
| Waste stream/substrate | Protein (%) | Carbohydrate (%) | Lipids (%) | Ash (%) | Fibre (%) | Reference |
|---|---|---|---|---|---|---|
| Aquaculture waste | 54–58 | <10 (low) | 12–16 | ∼8 | Low (<5) | 27 |
| Mixed biowaste | ∼40 | 10–20 | ∼30 | 8–17 | 7–16 | 28 |
| Coconut endosperm/soybean curd residue | 34–41 | 15–23 | 15–38 | 7–20 | 13–16 | 29 |
| Household/retail food waste | 9–30 | 40–70 | 7–22 | 4–10 | 5–20 | 30 and 31 |
| Municipal solid waste (MSW) compost | 2.4–4.0 | 30–44 | 1.5–3.2 | 25–35 | 13–19 | 32 |
| Anaerobic digestate | 4–7 | 20–40 | 1–3 | 12–20 | 23–31 | 33 |
| Agro-food lignocellulosic residues | 5–8 | 50–67 | 1–3 | 7–12 | 21–38 | 34 |
| Legume cell-powder | 23–29 | 36–44 | 0.6–2.6 | 2.8–4.9 | 24–26 | 35 |
| Pumpkin by-products | 17–21 | 50–57 | 3–7 | 6–11 | 12–21 | 36 |
| Macroalgae (seaweed) | 7–45 | 10–60 | 1–7 | 16–39 | 18–53 | 37 |
| Agro-industrial fruit residues (citrus/banana/wine pomace) | 3–7 | 40–74 | 1–4 | 5–9 | 18–32 | 38 |
| Green leafy crop residues | 6–24 | 43–55 | 1.2–2.5 | 15–21 | 13–27 | 39 |
| Yam peel | 3.5% | 81.73 | 4.17% | 5.4% | — | 40 |
All values are % of dry matter. Ranges represent variability due to source heterogeneity or the substrate used for bioconversion.
The integration of microbial biotechnology, insect farming, advanced fermentation processes, and biorefinery concepts offers multiple pathways for waste-to-protein conversion, each with distinct advantages and technological requirements.41
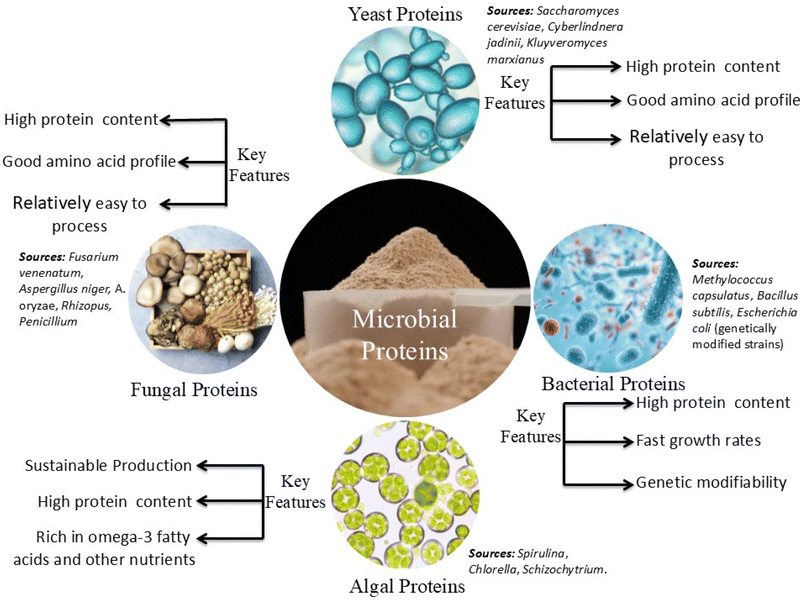
3.1 Microbial protein (Single-Cell Protein, SCP) production systems
Microbial protein, often referred to as Single-Cell Protein (SCP), represents a sustainable and innovative protein source derived from the cultivation of microorganisms including yeasts, bacteria, algae, and fungi on various waste substrates (Fig. 3). SCP production leverages organic wastes such as agricultural residues, food waste, and lignocellulosic biomass as low-cost, renewable feedstocks, enabling nutrient recycling and circular bioeconomy integration. These microbes convert carbon and energy from waste into biomass rich in protein, essential amino acids, vitamins, and minerals, which can be utilized as food supplements or animal feed.42Waste streams commonly used for yeast-based SCP production include agricultural residues (fruit peels and spent grains), food processing waste (potato, dates, Nyonya kuih, and confectionery side-streams), spent yeast from fermentations, and industrial effluents such as those from ethanol and beverage production.43,44,46–51 These substrates often require pre-treatment (e.g., enzymatic hydrolysis, acid hydrolysis, or lipid removal) to convert polysaccharides into fermentable sugars and reduce inhibitory compounds. For instance, the combined application of lipase and polyvinylpyrrolidone treatments on food residue hydrolysates increased the fermentable sugar yield and, subsequently, yeast biomass by up to 39%.52 Multiple yeast species have been explored, with Saccharomyces cerevisiae, Candida utilis, Cyberlindnera jadinii, Kluyveromyces marxianus, and Rhodotorula toruloides among the most prominent due to their ability to rapidly grow and efficiently convert various carbon sources to biomass.43,53,54 Optimal growth conditions typically involve aerobic fermentation at controlled pH (often 5.0–6.0) and temperature (25–30 °C). Addition of an appropriate nitrogen source (e.g., ammonium sulfate) is crucial. For A. oryzae grown on date waste, supplementation with 0.8% ammonium sulfate at pH 5.5 maximized the protein content to 13.8% in the SCP.55 Yield and protein content depend on the yeast strain, substrate composition, and fermentation strategy (batch, fed-batch, or chemostat).
Vieira-Lara et al. established a comprehensive quantitative physiological baseline for Cyberlindnera jadinii during growth on ethanol as a carbon substrate. The yeast exhibited a high maximum biomass yield on ethanol (0.83 g biomass per g ethanol) and strong energetic efficiency, with an ATP maintenance requirement of only 2.7 mmol g−1 h−1. Most C. jadinii strains (11 out of 13) demonstrated rapid growth (µmax > 0.4 h−1) even in vitamin-free mineral media, highlighting simple nutritional requirements, though strain CBS 621 was identified as thiamine auxotrophic. The prototrophic strain CBS 5947 achieved up to 100 g biomass per kg broth in fed-batch cultures with a biomass yield of 0.65 g per g ethanol, closely matching model predictions based on chemostat data. Notably, the yeast maintained a consistently high protein content (∼54% in chemostat and ∼42% in fed-batch cultures), with key metabolic enzymes—alcohol dehydrogenase, aldehyde dehydrogenase, and acetyl-CoA synthetase—enriched at low growth rates.43 Collectively, these findings demonstrate the high growth rate, energetic efficiency, and protein productivity of C. jadinii, underscoring its potential as a robust platform organism for sustainable single-cell protein and other ethanol-based bioproducts.
Gao et al. investigated the valorization of yellow wine lees and rice wastewater—organic-rich seasonal discharges—through microbial conversion into single-cell protein (SCP) using Candida utilis and Geotrichum candidum. Co-cultivation of the two yeasts achieved the highest biomass increase (4.2-fold higher than initial biomass) and crude protein yield (68.4%), outperforming monocultures. Process optimization via response surface analysis identified optimal fermentation conditions as 4 days of incubation, 6% (w/v) solid-to-liquid ratio, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 inoculation proportion, 18% inoculum dose, and 1.4 vvm aeration rate, yielding 66.3% crude protein content. The system achieved 78% soluble chemical oxygen demand (SCOD) and 55% total nitrogen removal, with acetic acid being the major residual carbon source. Importantly, the amino acid profile of the SCP closely matched commercial fishmeal and soybean meal standards, indicating its suitability for animal feed.53 These results demonstrate an efficient biotransformation strategy that couples wastewater remediation with the sustainable production of high-protein biomass.
1 inoculation proportion, 18% inoculum dose, and 1.4 vvm aeration rate, yielding 66.3% crude protein content. The system achieved 78% soluble chemical oxygen demand (SCOD) and 55% total nitrogen removal, with acetic acid being the major residual carbon source. Importantly, the amino acid profile of the SCP closely matched commercial fishmeal and soybean meal standards, indicating its suitability for animal feed.53 These results demonstrate an efficient biotransformation strategy that couples wastewater remediation with the sustainable production of high-protein biomass.
In another study, Thiviya et al. explored the potential of utilizing locally available fruit peel wastes—papaya (Carica papaya), watermelon (Citrullus lanatus), and banana (Musa spp.)—as low-cost substrates for SCP production using natural palmyrah toddy yeast under liquid-state fermentation. Physiochemical analyses of the peel extracts revealed that papaya peel supported the highest protein yield (52.4 ± 0.4%), followed by watermelon (45.2 ± 0.7%) and banana (30.4 ± 0.6%). Optimization of fermentation parameters with papaya peel extract showed maximum protein content (59.1 ± 0.8%) at 10% substrate concentration, pH 5.0, 25 °C, and 24 h of fermentation.46 The findings demonstrate that papaya peel waste serves as an excellent and sustainable substrate for producing protein-rich microbial biomass, thereby offering an effective approach to valorize fruit waste and mitigate associated environmental issues.
Tropea et al. investigated the production of SCP using mixed animal and agricultural residues—specifically fish waste along with pineapple, banana, apple, and citrus peels—as a complex fermentation substrate for Saccharomyces cerevisiae. Enzyme supplementation and nutrient enrichment optimized the fermentation conditions, resulting in a protein-enriched final substrate with a protein content of up to 40.19%, suitable for use in animal feed formulations. In addition to protein enrichment, the fermented substrate contained 14.46% crude lipid, 1.08% ash, and 6.29% lignin, while the soluble and insoluble sugar contents significantly decreased from 20.5% to 6.10% and 19.15% to 2.14%, respectively, indicating effective substrate utilization by the yeast.44 These findings demonstrate that integrating multiple food wastes in a single fermentation process offers an efficient biovalorization route for generating nutrient-rich SCP while simultaneously reducing environmental burdens from diverse organic wastes.
Díaz-Vázquez et al. explored the revalorization of tequila vinasses (TV)—a high-strength wastewater generated from tequila production in Jalisco, Mexico—through SCP production using mono- and mixed-culture yeast fermentations. Mixed cultures of Candida utilis, Rhodotorula mucilaginosa, and Kluyveromyces marxianus achieved superior pollutant removal and protein accumulation compared to monocultures, with C. utilis and K. marxianus contributing most effectively to COD, nitrogen, phosphorus, and sugar reduction. Although R. mucilaginosa populations declined due to the acidic and phenolic nature of TV, its presence enhanced amino acid enrichment and overall pollutant removal in the mixed culture. High-throughput sequencing and redundancy analysis confirmed dynamic community interactions linked to improved bioconversion efficiency. The mixed-culture fermentation yielded protein-rich biomass with significantly higher protein and amino acid content, highlighting its potential to transform tequila industry waste into valuable animal feed.50 This circular bioeconomy approach could enable the production of approximately 45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 664 tons of protein feed annually, offering a sustainable strategy for waste mitigation and resource recovery in the tequila and livestock sectors.
664 tons of protein feed annually, offering a sustainable strategy for waste mitigation and resource recovery in the tequila and livestock sectors.
Deuchande et al. focused on the valorization potential of spent yeast (SY), a byproduct generated from industrial fermentation processes used for producing beer and high-value biomolecules via genetically engineered microorganisms. SY samples derived from the production of beer and six biomolecules—β-farnesene (Fene), rebaudioside M (Reb M), manool (MN), patchouli (PAT), sandalwood (SDW), and sclareol (SCL)—were comprehensively characterized to assess their compositional diversity. SY from Fene, MN, and PAT fermentations exhibited comparable protein levels (35.9–44% dw), and balanced mineral profiles, and amino acid compositions, suggesting suitability for use as nitrogen-rich bulking materials. In contrast, SY from Reb M production contained notably higher lipid content (21 ± 2.6% dw), while SCL-derived SY displayed exceptionally high inorganic matter (45%), distinguishing it from other SY types (1.2–11.3% dw).48 These findings highlight significant compositional variability among spent yeast streams from precision fermentations and underscore the importance of such assessments in designing targeted and sustainable biorefinery or valorization strategies for industrial fermentation residues.
Yeast-based SCP platforms are among the most mature waste-to-protein technologies, benefiting from compatibility with established brewing, ethanol, and biotechnology infrastructure. Compared with bacterial systems, yeasts generally exhibit slightly lower maximum growth rates but greater tolerance to variable substrates and inhibitory compounds. Relative to algae and fungi, yeasts offer higher volumetric productivity and simpler reactor control, supporting superior scalability in industrial fermenters.
Most yeast SCP processes using industrial by-products fall within TRL 6–8, particularly for feed and selected food applications. They have higher scalability when substrates are sugar-rich and require minimal pre-treatment; however, extensive hydrolysis, detoxification, and nitrogen supplementation significantly increase costs and reduce competitiveness. Major implementation barriers include high energy demand for aeration, dependence on relatively clean waste streams, and limited consumer acceptance for food-grade applications. Thus, yeast SCP is a promising candidate as a near-term solution for feed and niche food markets rather than as a primary replacement for conventional dietary protein.
Substrate preparation often includes pretreatment (e.g., hydrolysis, sterilization, or pH adjustment) to make nutrients bioavailable and to reduce inhibitory substances.56–58 Typical bacterial genera used include purple non-sulfur bacteria (PNSB), notably Rhodopseudomonas palustris, valued for their efficiency on organic-rich wastewaters and co-products, Escherichia coli and recombinant variants for recombinant protein production from spent media, methanotrophs and hydrogen-oxidizing bacteria (e.g., Methyloparacoccus murrelli and Cupriavidus necator) capable of utilizing C1 gases derived from anaerobic digestion or waste gas streams, and Rhodococcus, Streptomyces, and Pseudomonas for high-value protein, lipid, or secondary metabolite co-production.49,56,58–60
Fermentation is usually carried out using batch, fed-batch, or continuous systems, with strict control of temperature, pH, aeration, and sometimes redox potential for optimal growth and yield.57 In many SCP processes, nitrogen limitation is engineered to enhance protein synthesis or lipid accumulation.
Bacterial SCP typically ranges from 35% to as high as 70% protein (dry basis) depending on the strain and substrate. Amino acid profiles can be comparable to plant-based or fishmeal proteins, but critical evaluation is necessary for their use in feed due to differences in digestibility and nucleic acid content.56,59,61 Aside from protein, bacterial SCP may also contribute valuable micronutrients, vitamins, lipids, and sometimes even speciality functional molecules (e.g., bacteriochlorophylls and coenzyme Q10).56 Waste-yeast used as a substrate can further enhance the profile through transferred nutrients.60
Wada et al. investigated the recovery of SCP from fuel synthesis process water (FSPW)—a nutrient-rich but underutilized waste stream abundant in arid regions such as the Middle East—using anaerobic fermentation with purple non-sulfur bacteria (PNSB). In 1-L batch fermentations with nutrient-amended feedstock (COD = 10.3 g L−1), approximately two-thirds of the chemical oxygen demand was degraded within 72 h at a rate of 2100 mg L−1 d−1, alongside complete nitrogen depletion, indicating nitrogen limitation. The process yielded a peak biomass concentration of 1.11 ± 0.037 g Volatile Suspended Solids per Litre (VSS per L), containing 35% protein, 32% lipid, 16% carbohydrate, and minor fractions of ash, carotenoids, bacteriochlorophylls, and coenzyme Q10.56 The amino acid composition of the microbial protein was comparable to soybean meal and met nutritional requirements for aquafeed, while metal analysis confirmed the absence of toxic elements. Overall, the results demonstrate that PNSB can effectively transform FSPW into protein- and lipid-rich biomass, offering a sustainable circular bioeconomy solution for aquafeed production and waste minimization in resource-limited regions.
Lynch and Connell explored the valorization of chemically defined spent media (CDSM) from cell culture bioprocesses as a nutrient source for secondary microbial fermentations aimed at recombinant protein production—advancing circular bioeconomy goals. Escherichia coli cultured in CDSM supplemented with 2% glycerol exhibited growth rates and recombinant protein yields comparable to those obtained in standard rich media Lysogeny Broth (LB) during both 4-hour expression screening and 16-hour preparative fermentation. Quantitative proteomic analysis via mass spectrometry revealed upregulation of protein synthesis machinery and downregulation of carbohydrate metabolism enzymes in CDSM-fed cultures, indicating adaptive resource reallocation to support protein expression.49 These findings demonstrate that CDSM, a major waste stream generated in large volumes by the bioprocessing industry, can be effectively repurposed as a fermentation feedstock, enabling sustainable recombinant protein production while reducing industrial waste generation.
Kerckhof et al. investigated the co-cultivation of methane-oxidizing bacteria (MOB) and hydrogen-oxidizing bacteria (HOB) as a sustainable approach for microbial protein (MP) production using biogas-derived substrates. Novel autotrophic HOB strains were isolated and paired with defined MOB cultures grown on a mixture of biogas and H2/O2 to explore potential synergistic interactions. Co-cultivation not only enabled simultaneous valorization of CH4 and CO2 but also enhanced biomass and protein yield, with several combinations showing 1.1–3.8-fold higher protein concentrations and 2.4–6.1-fold higher essential amino acid content compared to monocultures. The most promising consortium—Methyloparacoccus murrelli LMG 27482 and Cupriavidus necator LMG 1201—produced MP with an amino acid composition comparable to soybean and requiring 27–67% less biomass than conventional animal proteins to meet human nutritional needs.59 Although the MP was slightly lower in histidine, lysine, and threonine relative to tofu and fishmeal, the results demonstrate that MOB–HOB co-cultivation offers an innovative and safe strategy to generate high-quality microbial protein from waste-derived gaseous substrates, advancing circular and low-carbon food production systems.
Poladyan et al. examined the growth behaviour and bioenergetic responses of the chemolithoautotrophic bacterium Ralstonia eutropha H16 (Cupriavidus necator) during heterotrophic cultivation on various carbon sources, including fructose, glycerol, and lignocellulose-rich brewery spent grain hydrolysate (BSGH). The bacterium effectively utilized BSGH as both a carbon and energy source, achieving a biomass yield of approximately 0.7 g cell dry weight per L and displaying high activities of soluble (3.50 ± 0.02 U per mg protein) and membrane-bound hydrogenases (2.3 ± 0.03 U per mg protein). While growth on fructose and glycerol led to slight acidification and redox potential reduction, cultivation on BSGH maintained stable pH and Oxidation-Reduction Potential (ORP) conditions. ATPase activity was significantly lower under fructose–glycerol growth, suggesting an energy-limited state that triggered enhanced hydrogenase biosynthesis. The addition of ferricyanide modestly improved growth, whereas dithiothreitol temporarily suppressed it.57 Overall, the results demonstrate that organic wastes such as glycerol and BSGH are promising substrates for producing R. eutropha biomass enriched in O2-tolerant [NiFe]-hydrogenases, offering valuable prospects for biotechnological applications such as enzymatic fuel cells and bioenergy systems.
Liu et al. explored the substrate utilization preferences of Streptomyces sp. F-3 through functional proteomic and structural omics analyses to identify cost-effective nutrient sources for industrial cultivation. The strain displayed distinct nitrogen-source degradation abilities, showing poor utilization of intact globular proteins but efficient degradation of peptone and protein hydrolysates, particularly those derived from waste yeast. When grown on waste-yeast substrates, Streptomyces sp. F-3 exhibited rapid growth and high extracellular protein production, driven by three secreted S8 proteases that determined its nitrogen degradation specificity. Additionally, the strain exhibited substantial β-glucanase and chitinase activities, enabling the assimilation of yeast cell wall polysaccharides.60 These findings identify waste yeast—rich in peptone, β-glucan, and chitin—as an optimal, low-cost substrate for Streptomyces cultivation, expanding its valorization potential while offering a sustainable strategy for reducing industrial production costs.
Gómez Martínez et al. aimed to enhance the nutritional value of sugarcane bagasse through solid-state fermentation (SSF) with probiotic microorganisms, transforming this agroindustrial residue into a functional feed ingredient for sheep. Sugarcane bagasse was mixed with alfalfa flour, molasses, urea, and mineral supplements at different inclusion levels and fermented under controlled conditions. The compositional analysis revealed that the inclusion percentage significantly influenced ash, crude protein, organic matter, and digestibility, with the highest crude protein (16.3%) and dry matter digestibility (74.6%) achieved at lower bagasse inclusion levels. Microbiological evaluation showed increased populations of lactic acid bacteria and aerobic mesophiles during fermentation, while undesirable microbes such as molds, yeasts, and Salmonella were absent, confirming the microbiological safety of the process.61 Overall, the study demonstrates that SSF is an effective and sustainable approach for upgrading sugarcane bagasse into protein-enriched, digestible, and microbiologically safe feed, offering a practical strategy for valorizing agro-industrial waste in livestock nutrition.
Bacteria-based protein production from waste streams is a robust and scalable technology. It enables high-value protein yields (often 35–70% of cell mass) while simultaneously achieving waste remediation and supporting circular bioeconomy models.49,56–64 The diversity of bacterial strains, waste substrates, and fermentation strategies enables broad tailoring to local resource streams and nutritional targets.
Bacterial SCP systems demonstrate the highest theoretical conversion efficiencies among microbial platforms, with rapid growth rates and the capacity to utilize diverse substrates, including wastewaters and gaseous by-products. Compared with yeast systems, bacteria can exploit a broader substrate spectrum but are generally more sensitive to contamination and process fluctuations. Relative to algal and fungal platforms, bacterial systems achieve higher volumetric productivity but require stricter environmental control.
Heterotrophic bacterial SCP using agro-industrial effluents typically operates at TRL 6–8, whereas gas-fermentation and co-culture platforms remain at TRL 4–6. Practical feasibility is constrained by high aeration and sterilization costs, management of nucleic acid content, and regulatory barriers for food use. While bacterial systems offer strong potential for circular valorization of industrial wastes, their near-term deployment is most realistic in feed and industrial protein markets rather than direct human consumption.
Typical microalgae utilized include Chlorella vulgaris, Chlorella sorokiniana, Neochloris sp., Tetradesmus sp., and Synechocystis sp. for municipal/agricultural wastewaters.66,67Spirulina platensis is often used in feed trials and is easily cultivated on nutrient-rich effluents.73 Algae cultivation often occurs in open raceway ponds, high-rate algal ponds (HRAPs), or photobioreactors depending on scale and quality requirements.72,74 Dilution of effluents is common to avoid ammonia and salt inhibition. For instance, Chlorella species showed the highest productivity in 10–20% anaerobic digestate effluent, reaching up to 3.26 g per L biomass.66 High light intensity and continuous aeration optimize nutrient uptake and protein generation.70
Protein content in microalgae is commonly reported at 30–70% dry weight, with variances based on the species, substrate, and process.70,75Table 2 highlights various algal systems utilized for protein production.
| Algal species/consortia | Waste stream | Biomass yield | Protein content | Process features | References |
|---|---|---|---|---|---|
| a ADE-anaerobic digester effluent; NFP-nano-filtered pig slurry; PBR-photobioreactors; WRRFs-water resource recovery facilities. | |||||
| Chlorella vulgaris | Anaerobic digestate | 3.26 g L−1 (max) | High (not specified) | 10–20% ADE; robust growth | 66 |
| Chlorella & consortia | Pig slurry NFP | 0.55–0.58 d−1 (µ) | Low (N-limited) | Some consortia had optimal omega-6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratios 3 ratios |
67 |
| Chlorella sorokiniana | Wastewater (lab PBR) | 1.52 g L−1 d−1 | 388.2 mg g−1 (38.8%) | Nitrogen and P removal rates tracked; productivity peaks at moderate dilution | 70 |
| Macroalgae (Ulva, Cladophora) | Aquatic overgrowth/waste | — | “Modest” | Source of bioactives; less protein than microalgae | 76 |
| Mixed species (RAB system) | WRRF effluent | Site-dependent | Calculated fraction after conversion | WRRF integration; potential for >1000 MT protein-rich product per year | 72 |
| Spirulina platensis | Fish feed trial | Feed uptake study | Used at 5–15% of feed | Improved fish growth and meat quality | 73 |
Algal systems such as HRAPs and wetland-based oxidation ponds perform dual duties: wastewater treatment (nutrient and contaminant reduction) and resource recovery as protein-rich biomass.71,74Chlorella vulgaris and related species are particularly noted for high tolerance and remediation abilities, with HRAPs showing a nitrogen removal of 50–98% and phosphorus removal of 32–99% depending on the process.74 For instance, in East Kolkata Wetland, India, sewage-derived nutrient input is converted, using natural algal-bacterial processes, into protein through fish production, effectively treating over 900 MLD of sewage with BOD reductions of over 59%.71 In municipal WRRFs, rotating algal biofilm (RAB) systems can produce over 1000 metric tons of protein-rich co-products annually at a large scale, while reducing greenhouse gas emissions and supporting circular economy objectives.72
Harvested algal protein is processed into animal feed (esp. aquaculture and poultry), and food additives, or refined into biopolymers and speciality chemicals.25,72,73 Microalgae can provide all 20 essential amino acids, with good digestibility and functional properties. In feed studies, partial replacement with Spirulina platensis improved weight gain, protein efficiency, and body composition in catfish fed with 5–15% algae in the diet.73,75 Algae-based SCP production from waste streams efficiently recycles nutrients, reduces environmental pollution (N, P, and organic matter), and closes nutrient loops, supporting the circular bioeconomy paradigm.25,26,77 The process requires less land and water versus crop-based protein, with lower overall greenhouse gas emissions.45,75 Economic analyses suggest that energy costs, particularly artificial lighting, are the main operational barrier; leveraging sunlight or waste heat is critical for a competitive cost structure.70 Cofeeding waste-derived algae into integrated biorefineries further improves revenue and sustainability (biogas, bioethanol, bioplastics, and fertilizers).72,78
Algae-based SCP from waste streams is a technically viable, environmentally sound, and increasingly scalable method for sustainable protein generation, often yielding protein contents of 30–70% dry weight, and capable of treating large volumes of organic and nutrient-rich waste.25,26,45,65–68,70,72–76,78,79 Its full realization requires continued innovation in process optimization, valorization strategies, and policy support.
Algal systems provide unique environmental advantages by integrating wastewater treatment with protein production. Compared with microbial fermentation systems, algae require less chemical input but depend heavily on light availability and large surface areas, resulting in lower volumetric productivity. Relative to insect systems, algae offer superior nutrient removal but face higher harvesting and dewatering costs.
Most waste-based algal protein systems currently operate at TRL 4–6, with only a limited number of integrated wastewater-algae platforms approaching TRL 7. Scalability is restricted by seasonal variability, land demand, and energy-intensive harvesting. Consequently, algal protein is presently more viable for feed, additives, and co-product streams than for large-scale human food applications, despite strong environmental performance.
Common waste substrates include fruit peels (banana, orange, and pineapple), fibrous agri-waste (yam peel and rice polishings), food processing effluents (cheese whey and baker's yeast wastewater), crop residues (wheat straw and sugarcane bagasse), and specialty byproducts like date syrup press cake or potato peel. Key fungal genera used are Aspergillus (e.g., A. niger and A. oryzae), Penicillium, Trichoderma, Rhizopus, Fusarium, and Mucor. Strain selection depends on their ability to utilize the complex carbohydrates and lignocellulosic materials in these wastes, as well as their protein yield and nutritional profile.55,80 Fungi can be cultivated using two main fermentation approaches: submerged fermentation, which involves growth in liquid media (commonly in stirred-tank or bubble column fermenters) and allows precise control over parameters such as pH, oxygen, and nutrient supply; and solid-state fermentation, which takes place on moist solid substrates, closely mimicking natural fungal habitats and is typically used for processing low-moisture wastes.81,82
Fungal SCP is characterized by high protein (often 30–55% dry weight), moderate fat (2–11%), carbohydrates (14–21%), ash (4–16%), and fibers. The amino acid profile typically meets or exceeds that of conventional plant-based proteins, including all essential amino acids.80,83 Proximate analysis in various studies further confirms the presence of vitamins and minerals, enhancing the nutritional value.
Key process parameters affecting yield include fermentation time (ranging 5–10 days), temperature (typically 20–35 °C), pH (commonly acidic to mild, pH 4–5.5), and nitrogen supplementation source (e.g., ammonium sulfate often optimal).55
Ahmed et al. explored the potential of agricultural waste as a substrate for SCP production using filamentous fungi under submerged fermentation. Pineapple, orange, banana, sugarcane, and garlic wastes were evaluated, and fermentation parameters—temperature, pH, and nitrogen source—were optimized for the most promising fungal isolates. Among the tested combinations, Aspergillus niger cultivated on pineapple waste achieved the highest protein yield (9.79 ± 0.11 g L−1) after 10 days, followed by Penicillium citrinum on orange waste (9.41 ± 0.15 g L−1, 8 days) and Penicillium crustosum on banana waste (7.75 ± 0.11 g L−1, 6 days). Optimal SCP production occurred at 30 °C, pH 4.3, and with ammonium sulfate for A. niger; 30 °C, pH 5.0, and with ammonium sulfate for P. citrinum; and 20 °C, pH 5.0, and with ammonium sulfate for P. crustosum. Proximate composition revealed notable nutritional quality, with fat (2.74–10.73%), fiber (4.8–9.45%), carbohydrate (14.1–20.81%), ash (4.05–16.1%), total phenols (0.14–0.64%), and flavonoids (0.09–0.55%) varying across isolates. The amino acid analysis confirmed the presence of all essential amino acids in appreciable amounts.80 Overall, this work highlights the effective utilization of fruit and agro-wastes for SCP generation, introducing certain fungi as novel protein producers and demonstrating their potential as sustainable, nutrient-rich ingredients for food and feed applications.
In another study, Abdurrazaq et al. demonstrated the potential of agro-industrial waste, specifically yam peels, as an economical substrate for SCP production using Aspergillus niger. The fungal strain, isolated from garden soil and identified based on morphological characteristics, exhibited dark brown colonies with black conidial heads and pale-yellow reverses on Sabouraud dextrose agar, with microscopic observation confirming black globular conidia on extended conidiophores. Proximate analysis of yam peels revealed high carbohydrate content (81.73%), along with lipids (4.17%), proteins (3.5%), moisture (5.19%), ash (5.4%), and fiber (1.85%), indicating their suitability as a fermentation substrate. Submerged fermentation for seven days on a rotary shaker showed that A. niger achieved significantly higher biomass yield on a yam peel substrate (0.4 OD) compared to the commercial yeast extract peptone dextrose (YEPD) medium (0.23 OD).40 These findings underscore yam peels as a cost-effective and nutrient-rich alternative for SCP production while promoting sustainable waste valorization and reducing environmental pollution from agro-industrial residues.
Nadeem highlighted the potential of agro-industrial waste valorization for economic and environmental benefits by producing SCP from fungi using potato peels as a fermentation substrate. Four broth media—control, glucose, potato peel, and mixed—were formulated and supplemented with essential nutrients including potassium dihydrogen phosphate, magnesium sulphate, sodium chloride, and yeast extract. Among these, the mixed broth supported the highest dry cell biomass yield (0.523 g per 100 ml) and was selected for further optimization. Comparative fermentation trials conducted in stirred-tank and bubble column fermenters revealed superior fungal biomass production in the bubble column system, achieving an optimal yield of 5.45 g per 100 ml.81 These findings demonstrate that potato peels, rich in fermentable sugars and essential nutrients, serve as an effective and low-cost substrate for SCP production. The process not only offers a sustainable approach for producing protein-rich food supplements but also contributes to environmental pollution mitigation through the productive utilization of agro-industrial waste.
Ahmed et al. evaluated the potential of Trichoderma harzianum to produce FBP from rice polishings, an abundant agro-industrial byproduct. Fungal fermentation was optimized to achieve maximum biomass yield at 5% (w/v) rice polishings after 72 hours of incubation at 28 °C and pH 4, with a carbon-to-nitrogen ratio of 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 supporting the highest protein production. In a 75 L fermenter, the resulting FBP contained 49.5% crude protein, 32% true protein, 19.45% crude fiber, 9.62% ash, 11.5% cellulose, and 0.325% RNA. Amino acid profiling revealed that all essential amino acids were present in significant quantities, underscoring the nutritional quality of the fungal biomass.84 The study demonstrates that T. harzianum can efficiently convert rice polishings into protein-rich biomass suitable for use as a poultry feed supplement, offering a sustainable approach to valorize agricultural residues into high-value nutritional products.
1 supporting the highest protein production. In a 75 L fermenter, the resulting FBP contained 49.5% crude protein, 32% true protein, 19.45% crude fiber, 9.62% ash, 11.5% cellulose, and 0.325% RNA. Amino acid profiling revealed that all essential amino acids were present in significant quantities, underscoring the nutritional quality of the fungal biomass.84 The study demonstrates that T. harzianum can efficiently convert rice polishings into protein-rich biomass suitable for use as a poultry feed supplement, offering a sustainable approach to valorize agricultural residues into high-value nutritional products.
Asadollahzadeh et al. aimed to valorize spent liquors generated from acidic sulfite and neutral sulfite semi-chemical (NSSC) pulping processes by converting them into protein-rich fungal biomass. Three filamentous fungi—Aspergillus oryzae, Mucor indicus, and Rhizopus oryzae—were cultivated on diluted spent liquors in an airlift bioreactor operated at 35 °C, pH 5.5, and an aeration rate of 0.85 vvm. Among the tested conditions, A. oryzae achieved the highest biomass yield of 10.17 g L−1 when cultivated on 60% diluted spent sulfite liquor (SSL), followed by M. indicus (6.14 g L−1) and R. oryzae (5.47 g L−1). Cultivation of A. oryzae on 50% diluted spent NSSC liquor (SNL) yielded 3.27 g per L biomass. The resulting fungal biomasses were rich in nutrients, containing 407–477 g per kg protein, 31–114 g per kg fat, 56–89 g per kg ash, and 297–384 g per kg alkali-insoluble material (AIM). Furthermore, the amino acid, fatty acid, and mineral profiles of the fungal biomasses were comparable to those of conventional protein sources such as soybean meal.83 Overall, A. oryzae demonstrated superior performance in utilizing pulp and paper industry spent liquors, underscoring its potential for sustainable production of protein-rich fungal biomass and contributing to circular bioeconomy strategies.
Al-Farsi et al. focused on valorizing waste generated from the date industry by converting it into SCP through fungal fermentation. Five fungal strains were screened for their growth and protein production potential, with Aspergillus oryzae identified as the most efficient strain due to its robust biomass formation and high protein yield. Optimization experiments revealed that 0.8% ammonium sulfate served as the most effective nitrogen source, while a pH of 5.5 and a substrate-to-medium ratio of 75 g per 250 ml flask provided the best growth conditions. The SCP produced by A. oryzae demonstrated high nutritional quality, with essential amino acids constituting 46% of the total amino acid content.55 These findings highlight the feasibility of using date-processing waste as a low-cost substrate for SCP production, offering both an environmentally sustainable waste management strategy and an economic opportunity for the date industry.
Sar et al. explored the valorization of organic-waste-derived volatile fatty acids (VFAs) as substrates for fungal biomass production, focusing on the influence of cultivation modes on growth performance and biomass composition of Aspergillus oryzae. Agro-industrial VFA effluent was utilized in repeated-batch and fed-batch (stepwise and continuous-feeding) cultivation strategies to assess biomass productivity and quality. The continuous-feeding fed-batch mode achieved the highest dry biomass yield of 0.41 g dry biomass per g VFA fed, with the fungal culture exhibiting pellet morphology that simplified downstream harvesting. Biomass protein content varied with the operational mode, reaching 45–53% under continuous feeding and 34–42% under stepwise feeding. Moreover, the resulting fungal biomass was enriched with essential macronutrients and trace minerals such as magnesium, calcium, potassium, manganese, and iron—elements critical for animal nutrition.85 Overall, the findings demonstrate that the cultivation mode plays a pivotal role in determining the biomass yield and composition, and underscores the potential of VFA-derived fungal biomass as a sustainable, nutrient-dense ingredient for animal feed applications within a circular bioeconomy framework.
Many studies implement integrated bioprocesses, coupling protein production with enzyme, pigment, and biogas generation for improved waste valorization.86,87 Incorporation of bio-waste streams not only decreases environmental pollution but also creates a sustainable protein source, important for addressing global food insecurity.65,88 Scale-up experiments (e.g., use of bioreactors and bubble columns) demonstrate that such processes are technically feasible, and critical factors for industrial application are substrate preparation, process optimization, and cost-effective downstream processing.85 Solid-state processes offer low-input alternatives, especially where water use must be minimized.82
Fungal platforms combine the ability to degrade complex lignocellulosic wastes with relatively high protein yields, positioning them between bacterial/yeast systems and algae in terms of flexibility and productivity. Compared with bacteria and yeasts, fungi grow more slowly but tolerate more complex substrates. Relative to algae, fungi offer greater process stability and independence from climatic conditions.
Most fungal SCP systems fall within TRL 5–7, with mycoprotein representing the most advanced example. Solid-state fermentation offers low-input scalability in resource-limited settings, but industrial automation and consistent quality control remain challenging. Implementation barriers include long fermentation times, substrate pre-treatment, and downstream processing costs, making fungal protein most suitable for regional biorefineries and selected food and feed applications.
3.2 Insect-based systems
Insect-based protein production from waste streams has gained significant traction as a means of sustainably producing high-quality proteins while addressing the challenges of organic waste management. This approach leverages the remarkable ability of certain insects most notably the black soldier fly (Hermetia illucens), housefly (Musca domestica), mealworms (Tenebrio molitor), and superworms (Zophobas morio) to convert a variety of organic waste materials into biomass rich in protein and fat suitable for animal feed and, in some cases, human food.Insects are reared on diverse waste substrates, including food waste (fruit and vegetable residues), agricultural byproducts (bran, silage, and olive pulp), manure (poultry and pig), and even digestate from biogas plants. For example, black soldier fly larvae can achieve up to 98.9% biomass conversion on kitchen waste, 84.6% on vegetable waste, and 87.5% on fruit waste.89
High-nitrogen substrates like swill and pig manure lead to superior larval growth rates and process yields. One study found swill-fed black soldier fly larvae (BSFL) had a maximum growth rate (13.4 mg d−1) almost double that of a standard reference diet, while waste reduction efficiency reached 11.3 (waste reduction index), outperforming many conventional feedstocks.90 Agricultural wastes such as sugarcane bagasse require pre-treatment (thermal, enzymatic, and microbial) to liberate fermentable sugars, which can then support high larval bioconversion rates (13.4%) and yield larvae with crude protein content as high as 33% and fat at 28%.91 The bioconversion of organic waste into BSFL, which is a valuable source of lipids, and protein, is part of an ongoing creative effort in valorising waste. In this context, Aminuddin et al. explored the bioconversion of organic waste into valuable biomass using BSFL, a process that efficiently transforms waste into high-value products such as proteins, lipids, and chitin. The effects of waste type and moisture content on biomass conversion were evaluated, revealing that kitchen waste (KW) achieved the highest conversion efficiency (98.9 ± 0.71%) compared to vegetable waste (84.6 ± 1.41%) and fruit waste (87.5 ± 0.71%). Optimal conditions were achieved at 80% moisture content in KW, resulting in nearly complete biomass conversion (99.8 ± 0.58%). The resulting BSFL contained approximately 38–42% crude lipid, 34–41% crude protein, and 8–9% ash. Chitin, a valuable biopolymer of N-acetylglucosamine, was successfully isolated from the larvae through sequential deproteinization, demineralization, and decolorization, yielding 4–6% (g g−1) of a yellowish-grey powder. Fourier transform infrared (FTIR) spectroscopy confirmed the presence of characteristic chitin functional groups (–OH, –CH3CONH, and –CO), as well as triglyceride-associated X–H stretching in the lipid fraction.89 Overall, the study demonstrates that BSFL effectively upcycle organic waste into multiple bioproducts, contributing to sustainable waste management and resource recovery.
In another study, Kim et al. evaluated the potential of BSFL biorefineries as a sustainable alternative for producing protein-rich livestock feed and biodiesel from organic waste, reducing reliance on valuable feed crops and fossil fuel-based resources. The bioconversion efficiency of organic waste into BSFL biomass ranged from 32.0% to 35.8% after 24 days of cultivation. Both protein and lipid compositions of the larvae varied over time, with substrate type significantly influencing lipid accumulation—lower lipid content in the substrate resulted in reduced lipid deposition in the BSFL. Despite this variation, the estimated annual productivity of proteins (42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 471–48
471–48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 345 kg per ha per year) and lipids (41
345 kg per ha per year) and lipids (41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 642–64
642–64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 708 kg per ha per year) from BSFL cultivation exceeded that of conventional feed and biodiesel crops such as maize and soybean.92 Overall, the findings underscore the promise of BSFL-based insect biorefineries as an efficient and sustainable strategy to produce animal feed and biofuel precursors while valorizing organic waste and advancing circular bioeconomy principles.
708 kg per ha per year) from BSFL cultivation exceeded that of conventional feed and biodiesel crops such as maize and soybean.92 Overall, the findings underscore the promise of BSFL-based insect biorefineries as an efficient and sustainable strategy to produce animal feed and biofuel precursors while valorizing organic waste and advancing circular bioeconomy principles.
House fly larvae (HFL) are another promising candidate for sustainable protein production. In this context, Uushona et al. explored the potential of using organic waste—specifically pig manure and chicken layer droppings—as substrates for the production of HFL and evaluated the nutritional composition of the resulting biomass. A completely randomized design with three treatments (pig manure, chicken layer droppings, and a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of both) was used, each replicated six times. The results showed that pig manure yielded the highest HFL biomass, with significant increases in both wet (3.61 g) and dry (0.63 g) larval weights (P < 0.05). The introduction of HFL led to a 39–42% reduction in organic waste, demonstrating their efficiency in waste bioconversion. Nutritional analysis revealed that HFL biomass contained high protein levels (50–53%), along with considerable calcium (1.32–1.465%) and phosphorus (1.72–2.09%) contents.93 These findings indicate that both pig manure and chicken layer droppings are effective substrates for HFL cultivation, producing nutrient-rich larvae suitable as a sustainable protein source in animal feed. The study further recommends scaling up HFL production for integration into animal nutrition systems, aligning with circular bioeconomy principles.
1 mixture of both) was used, each replicated six times. The results showed that pig manure yielded the highest HFL biomass, with significant increases in both wet (3.61 g) and dry (0.63 g) larval weights (P < 0.05). The introduction of HFL led to a 39–42% reduction in organic waste, demonstrating their efficiency in waste bioconversion. Nutritional analysis revealed that HFL biomass contained high protein levels (50–53%), along with considerable calcium (1.32–1.465%) and phosphorus (1.72–2.09%) contents.93 These findings indicate that both pig manure and chicken layer droppings are effective substrates for HFL cultivation, producing nutrient-rich larvae suitable as a sustainable protein source in animal feed. The study further recommends scaling up HFL production for integration into animal nutrition systems, aligning with circular bioeconomy principles.
With increasing emphasis on sustainable protein production and agricultural waste management, insect farming presents a viable strategy for waste valorization. Liu et al. examined the influence of different agricultural waste-based diets on the growth performance, gut microbiota composition, and flavour quality of Zophobas morio Fabricius (Coleoptera: Tenebrionidae), offering valuable insights into its potential for waste recycling and edible insect production. Over a 32-day period, Z. morio larvae were fed wheat bran (WB), edible mushroom sticks (EMS), or apple pomace (AP), and their biological responses were analyzed using 16S rRNA sequencing and gas chromatography-ion mobility spectrometry (GC-IMS). Larvae fed with WB exhibited the highest weight gain, associated with an increased abundance of Lactobacillus and Lactococcus, while EMS feeding enhanced Spiroplasma populations without improving growth performance. In contrast, AP supported survival but limited weight gain, correlating with higher levels of Chryseobacterium and Kosakonia. The gut microbiota composition was found to modulate the volatile organic compound (VOC) profile, with Lactobacillus and Lactococcus promoting the formation of esters and ketones, Chryseobacterium contributing to sweet and fruity volatiles, and Spiroplasma potentially reducing alcohols and sulfur compounds.94 Overall, this study highlights the potential of Z. morio to serve as a sustainable bioconversion agent for agricultural waste while providing a nutritionally and sensorially favorable edible protein source.
Studies consistently highlight that insect meals have protein contents ranging from 34–57% (dry matter), with a complete amino acid profile suitable for animal industry needs.95,96 For instance, BSFL protein meal can replace 50–100% of soybean meal in poultry diets without negative impacts on performance (egg production and feed intake).97 In addition, amino acid and fatty acid profiles are influenced by substrate choice and can be optimized by feed formulation.98 In quails, up to 15% inclusion of defatted BSFL meal did not cause adverse effects on meat quality, with benefits for aspartic acid, glutamic acid, alanine, serine, tyrosine, and threonine content.99,100
Protein extraction from insect biomass can be achieved through chemical or enzymatic hydrolysis. For BSFL, protein and bioplastic precursor yields vary depending on the extraction method, with film-forming bioplastics displaying higher degradability at higher protein incorporations.101,102 Insect production also generates significant side streams (frass and exuviae). Frass exhibits biomethane potentials comparable to cow manure, making it a viable feedstock for biogas production (biomethane potential: 149–275 L per kg VS).103,104 Chitin and chitosan extraction from insect exuviae offers opportunities for additional high-value bioproducts, with process yields of purified chitin reported at approximately 14–17% of the starting biomass.105,106
Life cycle assessments show that insect-based protein can significantly decrease the carbon footprint of animal feed compared to soybean or fish meal, and can directly address food system greenhouse gas (GHG) emissions.107,108 Insect rearing closes nutrient loops in circular economy models, as demonstrated by university food systems using institutional food waste to feed local insect farms.109 Chemical and biological safety of insect meal depend on the waste origin and strict control of rearing conditions and substrate quality to avoid contaminant accumulation (e.g., heavy metals and mycotoxins). Reports show generally safe profiles, with specific vigilance needed for certain metals in some circumstances.110,111 Regulations in the European Union (EU) and elsewhere often restrict the use of animal byproducts or post-consumer waste in insect rearing; only plant-derived and certain pre-consumer organic substrates are currently allowed in many jurisdictions.112,113 Economic scaling and sector development require further upscaling, integration with other food and waste streams, and regulatory adaptation.114,115
Insect-based systems display exceptional tolerance to heterogeneous organic waste and high bioconversion efficiencies. Compared with microbial platforms, insects require simpler infrastructure and lower energy input, but offer less precise process control. Relative to algae and fungi, insects provide faster biomass accumulation and superior waste reduction efficiency.
Insect protein for animal feed has reached TRL 7–9 in several regions, surpassing most microbial and algal systems in market maturity. However, regulatory restrictions on substrates, biosecurity requirements, and cultural resistance limit their expansion into human food markets. Thus, insect farming represents one of the most practical near-term waste-to-protein strategies for feed, while its broader role in human diets depends primarily on regulatory evolution and social acceptance rather than technological capability.
Waste-to-protein platforms represent a convergent solution to two critical challenges: protein scarcity and organic waste management. Strategic platform selection should consider substrate availability, target application (feed versus food), required protein quality, advantages and constraints. Table 3 compares major waste-to-protein conversion systems in terms of protein content, TRL, suitability for food and/or feed applications, and their key advantages and limitations.
| System type | Typical waste substrates | Protein content range | TRL range | Feed vs. food suitability | Key advantages | Key limitations | Reference |
|---|---|---|---|---|---|---|---|
| a DM: Dry Matter; COD: Chemical Oxygen Demand; DW: Dry Weight: GRAS: generally recognized as safe; PHA: polyhydroxyalkanoates; PHB: polyhydroxybutyrate (a common type of PHA bioplastic); SCP: Single-Cell Protein; TN: Total Nitrogen; TP: Total Phosphorus; COD: Chemical Oxygen Demand; VFAs: Volatile Fatty Acids; NIR: near-infrared; RNA: ribonucleic acid. | |||||||
| Yeast (Saccharomyces cerevisiae) | Fruit peel hydrolysate | ∼50% DW protein; 21.40 ± 0.60 g per L protein | TRL 4–6 (lab–pilot scale in this study) | Both feed & food potential (species widely used, GRAS) | Utilizes fruit waste hydrolysate; high protein yield; simultaneous ethanol and protein production; robust, fast-growing industrial yeast | Nucleic acid content not assessed; RNA reduction likely needed for food use; scale-up and regulatory validation for waste substrates required | 116 |
| Yeast (Cyberlindnera jadinii) | Short-chain fatty acids (VFAs from waste) | 41.3% crude protein; 17.3% lipids | TRL 4–6 | Feed & food potential (edible, GRAS history) | Utilizes VFAs from anaerobic digestion; no strong substrate inhibition; produces ∼37% odd-chain fatty acids | Requires a controlled pH-stat process; scale-up and regulatory validation needed | 117 |
| Yeast (Yarrowia lipolytica) | Brewery spent grain + bakery leftovers | Balanced fatty acid profile (lipid-focused system) | TRL 3–5 (lab scale in this study) | Mainly feed/industrial; food potential not assessed | Non-conventional oleaginous yeast; grows on complex wastes; versatile metabolism; strong lipid synthesis | Protein production not quantified; system primarily lipid-oriented; scale-up and regulatory validation needed | 118 |
| Purple Non-Sulfur Bacteria (PNSB) | Agri-food waste (VFAs, carbohydrates, and alcohols), dairy and brewery effluents | Protein-rich biomass with high essential amino acid quality; rich in vitamins, pigments and antioxidants | TRL ∼4–6 | Both feed & food (emerging) | Near-unity carbon-to-biomass conversion under phototrophic growth; metabolic versatility; co-production of vitamins and pigments | Requires light; complex scale-up; limited industrial deployment | 119 |
| PNSB (Rhodopseudomonas palustris-dominated culture) | Synthetic VFA mixture (70% acetate, 10% propionate, and 20% butyrate) representative of fermented organic waste (OFMSW) | 62% crude protein (DW); PHA 12–18% DW (peak 0.18 g PHA per g DW) – true dual protein + bioplastic platform | TRL 5–6 (long-term lab–pilot continuous system, 160 days operation) | Primarily feed (aquaculture SCP); not yet food-grade | High protein SCP (up to 62%); essential amino acid profile meets fish dietary needs; simultaneous production of PHA and pigments; selective NIR phototrophic growth; contamination reduction via membrane-fed VFAs | Requires light and a membrane system; moderate biomass density; PHA fraction lower than in PHB-specialized systems; regulatory barriers for food use | 120 |
| Microalgae (Chlorella vulgaris) | Membrane-treated distillery wastewater (industrial alcohol wastewater after anaerobic + membrane treatment) | 49.6 ± 1.4% DW protein; balanced amino acid profile with elevated essential amino acids compared to the control | TRL 5–6 (lab-scale to pilot-scale demonstrated) | Primarily feed | High protein accumulation in wastewater; strong nutrient removal (80% TN, 94% TP, snd 72.24% COD); simultaneous biomass production and wastewater treatment; removes heavy metals (Ca, Mg, Mo, As, Pb, and Cu) | Moderate biomass productivity (0.04 g L−1 d−1); requires light and controlled conditions; digestibility limited by the rigid cell wall; large-scale cost and harvesting challenges | 121 |
| Microalgae (Scenedesmus obliquus) | Anaerobic digestate from food waste + waste sugar | ∼43.8% DW protein under mixotrophic growth | TRL ∼5–6 (pilot-scale demonstration) | Primarily feed; emerging food potential | Mixotrophy boosts biomass (up to 10.5 g L−1) and protein; strong N, P and sugar removal | Requires light, organic carbon, and culture pre-concentration; scale-up and regulatory validation needed | 122 |
| Fungi (Aspergillus oryzae) | Soy whey (tofu wastewater; soy-processing by-product) | 41.08–66.96% w/w protein (best condition: 55.93% w/w with N + mineral supplementation); protein yield up to 356 mg protein per g DW substrate | TRL 5–7 (pilot/lab scale for protein; TRL 8–9 only for enzyme/koji applications) | Both feed & food (GRAS; long history in food fermentation) | GRAS organism; well-established fermentation technology; converts soy whey into protein-rich biomass; reduces COD by ∼60%; high biomass yield (up to 4.94 g DW per L) under optimized conditions | High nucleic acid content typical of mycoprotein; requires RNA reduction for human food; morphology (pellet size) affects mass transfer; needs pH and nutrient control to avoid growth inhibition | 123 |
| Fungi (Neurospora intermedia) | Pistachio dehulling waste (PBM, hot-water extract); literature also supports oat hulls and other lignocellulosic residues | 20.4–27.1% protein (up to 31.4% at low COD/N with N-supplementation); biomass yield 0.37–0.49 g biomass per g initial COD | TRL 5–7 (lab to pilot scale) | Both feed & food (edible filamentous fungus and traditional fermented foods) | Edible species; grows on low-sugar, high-COD waste; converts phenolic/aromatic compounds; improves cereal-based foods and fermented products; compatible with biorefinery concepts | Lower protein content than bacteria/algae; slower growth; morphology control needed; sensory acceptance depends on product formulation | 124 |
| Fungi (Rhizopus arrhizus/Rhizopus spp.) | Sugar beet pulp (semi-solid fermentation; with ultrasonication or hydrothermal pretreatment) | 53.7 → 93.82–134.11 mg per g DW protein (i.e., ∼5.4% → 9.4–13.4% w/w); essential amino acid ratio improved from 0.81 to 1.96; biomass yield up to ∼0.49 g biomass per g initial COD | TRL 5–7 (lab to pilot scale for protein biomass from waste) | Mainly suited for feed, with potential for food use in edible species after comprehensive safety assessment and nucleic acid reduction | Enhances substrate protein content ∼2.5×; strong pectinolytic and cellulolytic activity; grows well on low-value agro-waste; improves essential amino acid balance; compatible with semi-solid fermentation | High nucleic acid content (≈225–271 mg g−1, requires heat treatment for reduction); morphology control needed; slower than bacteria/yeast; process optimization required | 125 |
| Insects (black soldier fly larvae, Hermetia illucens) | Post-consumer restaurant food leftovers (chicken, meat, rice, vegetables, and bread); poultry feed (control) and 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 mix 50 mix |
33.1–35.7% DM crude protein; 35.8–42.5% lipids; balanced essential amino acids | TRL 7–9 | Primarily animal feed; limited human food uses | High bioconversion efficiency; dual products (protein + frass); processes diverse wastes; low land and water use | Regulatory limits for food use; substrate safety concerns; risk of heavy metal accumulation | 126 |
| Insects (black soldier fly larvae, Hermetia illucens) | Organic agro-industrial by-products; kitchen and food waste (including mixed fruit–vegetable residues) | 39.38–48.20% crude protein (DM basis); rich in essential amino acids (leucine, lysine, valine, and histidine dominant); lipid 25.69–38.36% DM | TRL 7–9 | Primarily animal feed; strong aquaculture potential | High protein with balanced amino acids; converts diverse wastes; produces protein plus nutrient-rich frass; low land and water use | Substrate-dependent quality; chitin affects digestibility; safety depends on the waste source; food-use regulations remain strict | 127 |
4. Innovative technologies for waste to protein conversion
Innovative technologies to convert waste streams into alternative proteins are drawing substantial attention due to their potential for improving global food security, supporting circular economies, and reducing the environmental footprint of food production. There are several key technological approaches, including protein recovery from seafood & agro-food side streams, next-generation algal & fungal processes and integrated waste-to-protein systems in the circular bioeconomy.4.1 Protein recovery from seafood and agro-food side streams
The pH-shift technique, which uses acid or alkaline solubilization followed by isoelectric precipitation has been used to extract proteins from fish-processing waste such as herring, salmon, and cod side streams. At the pilot scale, this method can achieve protein recovery yields exceeding 80% depending on optimization parameters. Protein isolates from Tra catfish head-backbone and abdominal cut-offs reached recoveries of 83.0–88.9% under optimal conditions (pH 12, solvent-to-material ratio 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, extraction duration 150 minutes), also leading to a significant reduction of fat and ash content in the isolates compared to raw materials.128 Life cycle assessment (LCA) studies show that pH-shift technology can reduce the carbon footprint, acidification, and water use by more than 95% compared to conventional fish protein hydrolysate production, with sodium hydroxide use and energy sourcing as the main environmental impact drivers.129
1, extraction duration 150 minutes), also leading to a significant reduction of fat and ash content in the isolates compared to raw materials.128 Life cycle assessment (LCA) studies show that pH-shift technology can reduce the carbon footprint, acidification, and water use by more than 95% compared to conventional fish protein hydrolysate production, with sodium hydroxide use and energy sourcing as the main environmental impact drivers.129
Emerging “green refining” technologies utilize microbial and enzyme treatments, physical methods like ultrasound and pulsed electric field, and solvent extraction for recovering proteins from food and seafood residues. These methods can yield significant quantities of functional protein and other bioactive compounds suitable for nutraceutical and additive applications.130
Enzymatic hydrolysis uses enzymes such as Alcalase to convert fish side-stream proteins into hydrolysates, often producing bioactive peptides with antioxidant properties. For Tra catfish processing by-products, optimal hydrolysis (enzyme/substrate ratio 3%, 50 °C, 3 hours) produced a degree of hydrolysis of about 22.5–24%, protein recovery up to 81.5%, and DPPH-radical scavenging activity as high as 86.1%.128 Similar optimization strategies for small fish like Dagaa led to protein recoveries up to 71% and 83% degree of hydrolysis.131 Emulsion formation during high oil content hydrolysis can inhibit protein recovery, and thus pre-treatments or emulsion management is needed for maximal yields.132 Ultrasound-assisted extraction (UAE) and other advanced, non-thermal pre-treatments (e.g., pulsed electric field) enhance protein yields, solubility, and functional properties. In sea bass side streams, ultrasound treatments optimized at 30 min and 50 °C resulted in high protein recovery and antioxidant activity; importantly, the extracts tested negative for mycotoxins under optimal conditions.133 When used before enzymatic hydrolysis of mackerel side streams, ultrasonication increased the degree of hydrolysis and protein solubility, generating more favourable peptide profiles and reduced bitterness.134 These strategies are increasingly considered “green” technologies due to less energy and chemical usage compared to traditional methods.135 Process waters from seafood processing, notably salted cod, contain dissolved proteins and minerals that can be recovered using filtration and flocculation. Studies showed that flocculation with Levasil RD442 at 1.23% and a maturation period of 1 hour achieved a protein recovery of 51.8% in high-salt process waters; ultrafiltration further increased protein recovery up to 57% in the retentate.136 When integrated, these methods reduce nutrient-rich effluent burdens and boost overall process sustainability.
Agro-industrial residues, such as apple pomace, are effective substrates for submerged fungal propagation (e.g., basidiomycetes such as Pleurotus sapidus). After just 4 days, this method yielded fungal biomass with 21% true protein (dry matter), 4% lipid, and valuable amino and fatty acid profiles, supporting the upcycling of agro-industrial waste into vegan and speciality protein products.137 Similar approaches using food industry side streams and basidiomycetes emphasize the route's efficiency, scalability, and production of bioactive secondary metabolites.
4.2 Next-generation algal & fungal processes for waste-to-protein conversion
Algal biomass, both microalgae and seaweed (macroalgae), is recognized as a highly promising feedstock for upcycling waste streams into protein and other value-added compounds. Microalgae, in particular, are attractive due to their rapid growth, high protein content (30–70% dry biomass in some strains), and their ability to utilize waste-derived nutrients and CO2, thus contributing to both protein supply and environmental sustainability.138,139 Cultivation systems frequently use nutrient-rich effluents from wastewater treatment, digestate from anaerobic digestion, or flue gases, maximizing circular bioeconomy benefits.65 The transition to integrated algal biorefinery concepts has enabled multi-product streams, significantly improving process economics. Recent techno-economic analyses indicate that valorizing all proteins, lipids, and carbohydrates from algal biomass may reduce costs by up to 40% compared to single-product systems.140 Quantitative reports show that microalgal protein extraction can achieve yields of 50–65% conversion efficiency from harvested biomass depending on the species and method.138 Advanced fractionation using ionic liquids, membrane technologies, and green chemical approaches allows the isolation of high-quality protein isolates with preserved amino acid profiles.Recent studies demonstrate that, when using nutrient-rich anaerobic digestate as a feedstock, microalgae productivity can reach 20–40 g dry weight per m2 per day, with protein comprising 40–60% of the dry biomass.33 High-value seaweeds can yield protein extraction efficiencies up to 60% (e.g., enzyme-assisted extraction in Eucheuma denticulatum) without compromising co-product quality.141 Integrated wastewater–microalgae systems can achieve nitrogen and phosphorus removal rates approaching 90%, while simultaneously producing protein-rich algal biomass for feed or food supplementation.138 Life cycle assessments have shown a reduction in greenhouse gas emissions by over 70% compared to conventional soybean production when microalgae are cultivated on waste streams with CO2 recycling.139 Additionally, microalgal bioresource upgrading may contribute to biofuel yields with a higher heating value (HHV) of 35.7 MJ kg−1, further improving process integration and energy circularity.142
Filamentous fungi and fungal consortia (including basidiomycetes and ascomycetes) have emerged as efficient upcyclers of a wide range of agricultural, lignocellulosic, and food industry wastes into protein-rich biomass. These organisms metabolize complex polymers via specialized enzymes, making them particularly effective on recalcitrant substrates such as cellulose, lignin, and food processing residues.143 Mycoprotein systems (e.g., Fusarium venenatum as in Quorn® production) can yield 45–50% protein (dry mass basis) from starchy or sugary industrial byproducts, with fermentation productivities upwards of 0.15–0.3 g L−1 h−1.144 Optimized submerged fermentation of agro-industrial side streams using basidiomycetes can yield fungal biomass with 18–24% true protein (dry matter basis) after 3–5 days, depending on the substrate composition and strain choice.143 Case studies with Pleurotus sapidus grown on apple pomace have reported protein yields of 21% dry matter.65 Enhanced pre-treatments (e.g., enzymatic hydrolysis and ultrasonic disruption) can increase overall protein yields from complex substrates by 10–30%.
In synthetic biology-enabled processes, new fungal strains are being engineered for (a) faster growth, (b) optimized protein secretion, and (c) improved utilization of heterogeneous waste streams. These developments are beginning to close the yield gap with existing bacterial and algal systems.144
Fungal fermentation of agro-wastes is often combined with enzyme production, organic acid recovery, or other bioactive compounds to create multi-product biorefinery models.145 Lignin-modifying fungi can achieve waste conversion rates of 40–55% of initial dry mass, combining protein recovery with pollutant removal.146 Fungal systems also offer high flexibility in process design, tolerance of substrate impurities, and favourable life cycle environmental metrics.
Next-generation algal and fungal platforms for waste-to-protein conversion are advancing rapidly, with consistently high yields in optimized, integrated systems, significant environmental and circular economy benefits as demonstrated via life cycle and techno-economic analyses, emerging synthetic biology and process control tools enabling further gains in conversion efficiency, substrate flexibility, and scalability.33,144
4.3 Integrated waste-to-protein systems in the circular bioeconomy
Integrated waste-to-protein systems are designed to valorize diverse organic side streams (food, agricultural, industrial, and municipal waste) into high-value protein products using microbial, insect, algal, or fungal bioprocesses. These systems maximize resource efficiency, facilitate multi-product recovery (proteins, lipids, and energy), and reduce environmental footprints, forming a key pillar of the circular bioeconomy model.A central tenet of a circular bioeconomy is the reincorporation of waste streams into productive cycles via biotechnological and biorefinery platforms. For instance, recycling chemically defined spent media (CDSM) from mammalian cell culture into microbial fermentation feed enables the production of recombinant proteins by E. coli. The results indicate that CDSM supplemented with 2% glycerol can maintain E. coli growth and recombinant protein production rates equivalent to conventional media, with proteomic analysis confirming upregulation of protein synthesis enzymes and downregulation of carbohydrate metabolism enzymes. Globally, this approach could valorize hundreds of millions of liters of bioprocess industry waste annually.49 Modern integrated systems may involve multi-stage biorefineries, such as combining BSFL bioconversion with anaerobic digestion (AD) and hydrothermal carbonization (HTC). This sequence efficiently upgrades fat-rich food residues, improves biogas yields, and recovers high-value larval protein and biomass, while also optimizing energy recovery and reducing external input by leveraging thermal integration across stages.147 In the cited study with BSFL integration, effective fat removal, improved digestate dewaterability, and a sustainable process chain for circular valorization of food industry waste were demonstrated, leading to high multi-product yields and energy neutrality for the system.147 In food industry contexts within northern Europe, by-product quantification (whey: 2318.3 kilo tonnes per year in Denmark, sugar beet pulp: 1421.3 kt per year) and mapping of valorization methods confirm the regional scale and substrate potential for bioprocesses.148
A comparative process using CDSM waste in E. coli fermentation showed that protein yields in the test (CDSM-fed) were nearly identical on both short-term (4-hour screen) and long-term (16-hour batch) timeframes to those using standard media, with no loss in production efficacy.49
Circular integration entails significant GHG emission reductions, improved resource utilization, and waste minimization. In Brazil, for example, the integration of biomethane from urban and agro-industrial waste into the national energy matrix supports both emission goals and energy security, with technical and economic assessments outlining positive impacts for public policy and investment.149 Advanced biotechnologies, including microbial consortia engineering, automation, and “smart” resource tracking (e.g., using digital twin and big data approaches), are increasingly embedded in these systems to enhance efficiency and traceability.150 Integrated waste-to-protein systems are projected to play a pivotal role in meeting future protein demand with minimal land, water, and energy inputs.
5. Food and feed applications of waste-derived proteins
Waste-derived proteins are gaining traction as sustainable ingredients in various human food applications, offering potential benefits in terms of nutrition, environmental impact, and cost-effectiveness. These proteins, sourced from microbial or insect biomass grown on organic waste streams, can be incorporated into meat analogues, dairy alternatives, and baked products, enhancing their nutritional profiles and lessening the environmental impact.5.1 Integration in human foods
Waste-derived proteins represent a promising and increasingly mainstream solution for fortifying and replacing protein in meat analogues, dairy alternatives, and baked goods. These proteins, sourced from microbial or insect biomass grown on organic waste streams, can be incorporated into meat analogues, dairy alternatives, and baked products, enhancing their nutritional profiles and lessening the environmental impact.42 However, consumer acceptance and regulatory hurdles remain significant challenges for widespread adoption.Alternative and hybrid meat products increasingly use upcycled proteins from sources such as brewers' spent grain (BSG) and insect flour. For instance, hybrid sausages developed with 22% BSG and 10% insect flour can effectively replace up to 35% of the turkey meat without compromising sensory qualities or consumer acceptance. These inclusions significantly enhance the final product's protein, fiber, and micronutrient profiles—BSG delivers additional protein and fiber, while insect flours contribute minerals such as iron and zinc.151 Black soldier fly larvae, cultivated on various food industry wastes, can yield a crude protein content of up to 42% and a fat content of 29%. These attributes enable substantial protein recovery for use in meat analogues, closing nutrient loops and offering a direct avenue for food waste valorization.152 Nevertheless, sensory issues such as strong flavors and public acceptance pose challenges for broader adoption.
Product optimization using waste-derived proteins can achieve textural parity with conventional meat, as demonstrated when combined formulations achieve optimal chewiness and color. The majority of sensory parameters (such as juiciness and odour) remain comparable to traditional products, with some enhancements in color and appearance that are positively rated by consumers.151
Studies confirm significant protein enhancement from waste sources such as apricot seed press cake (APC), which contains nearly 49.8% protein. Utilization of APC in dairy alternative matrices, such as apricot seed milk, delivers a protein content of 8.3%, surpassing many other types of plant-based milk. Further processing into powdered forms maintains a protein concentration of 38.5%, rendering these powders suitable for long-term storage and application across diverse food systems.154 Quinoa, featuring higher protein concentration and advantageous amino acid distribution, is similarly upcycled in dairy alternatives and supports nutritional fortification as well as fulfilling the demands for gluten-free products.155
Over 50 studies confirm the immense potential of plant food byproducts, especially fiber- and protein-rich fractions, in generating dairy alternatives with novel techno-functional features. Those derived from byproducts not only address sustainability but often demonstrate improved bioactive or health-promoting properties, though further work is needed to enhance palatability and consumer satisfaction.156 Fermentation and enzymatic pretreatments are gaining ground as ways to improve the digestibility and sensory profile of waste-enriched dairy analogues, with notable increases in protein accessibility and bioactive peptide generation.145
Waste valorization in baked goods commonly exploits protein-rich byproducts such as brewer's spent grains, wheat bran, and oilseed cakes. Brewer's spent grain protein isolate, for example, demonstrates postprandial amino acid uptake in humans comparable to widely consumed pea protein, indicating its suitability as a functional ingredient for protein-enriched baked goods.157
In extrusion-processed snacks, upcycled biomass such as vegetable pomace and BSG can account for 5–30% of the total formulation, improving protein content, dietary fiber, and antioxidant activity without negatively impacting product acceptability when carefully optimized.158 Addition of such waste-derived protein can increase total protein by 2–6 g per 100 g product, representing a nutritionally meaningful contribution relative to the Recommended Dietary Allowance (RDA), particularly when such snacks are consumed regularly or in populations with suboptimal protein intake.
Challenges include the maintenance of product expansion, texture, and appearance. Higher inclusion rates may reduce the expansion index and brightness, as observed in extruded breakfast cereals, necessitating co-processing or blending strategies. Sensory acceptance remains largely positive if levels are optimized and off-flavors from waste matrices are adequately masked.158 Alongside protein, waste stream components add value through polyphenols, dietary fiber, and potentially bioactive peptides with antioxidant, antihypertensive, and antimicrobial benefits.159,160
5.2 Integration of waste-derived proteins in animal feeds
Waste-derived proteins have significant potential as sustainable and cost-effective ingredients in animal feeds for livestock, aquaculture, and pets. The increasing demand for animal protein necessitates the exploration of alternative protein sources that can reduce reliance on traditional feed ingredients, such as soybean meal and fishmeal, which are associated with environmental concerns and fluctuating prices. Waste-derived proteins offer a viable solution by converting organic waste streams into valuable feed resources, promoting a circular economy in the agriculture and aquaculture sectors.The use of BSFL in livestock feed has been shown to enhance feed conversion efficiency and reduce greenhouse gas emissions, contributing to more sustainable livestock production. Similarly, SCPs can provide a balanced amino acid profile and improve the digestibility of livestock feeds.
In the United States, an estimated 500 million metric tons (MMT) of total feed was consumed by livestock in 2023. Of this, 37% comprised circular (i.e., waste-derived or co-product) ingredients such as animal protein meals, animal-derived fats, soybean meal, and corn distillers' dried grains (DDG). Sector-specific proportions of circular ingredient use included swine (28%), poultry (32–36%), beef (35%), dairy (50%), goats and sheep (52%), equine (77%) and aquaculture (see below). However, domestic feed usage of rendered animal protein and fat decreased from 8.95 MMT (2019) to 4.8 MMT (2023), partly due to evolving feed formulations.160 Waste-derived ingredients like insect meal and copra meal offer balanced protein profiles but may require processing to enhance digestibility and nutrient absorption.161,162 Notably, insect meals, while generally suitable, can have reduced digestibility at high inclusion levels due to chitin content.
Insect meal supplementation (Hermetia illucens) in Nile tilapia diets as up to 50% replacement for soy concentrate resulted in improved or unchanged growth and nitrogen utilization, but higher inclusion could impair growth if not balanced for nutrients.166 Duckweed (Lemna spp.), grown on nutrient-rich wastewater, provides protein-rich biomass for poultry and fish, and is associated with favorable impacts on animal productivity and product quality.167 Fish protein hydrolysates from waste streams can be directly used as feed supplements, supporting the circular bioeconomy.163,164
The use of rendered poultry and fish byproducts remains common, both for protein and palatability. The nutritional adequacy of these feeds is generally comparable to high-quality conventional ingredients when carefully formulated. Growth in pet food sectors, especially for premium or hypoallergenic diets, has furthered interest in insect-based proteins.169 The use of these alternative proteins can address consumer concerns about the sustainability and ethical sourcing of pet food ingredients. Furthermore, waste-derived proteins can offer unique nutritional benefits for pets, such as improved digestibility and reduced allergenicity.
6. Food-grade vs. feed-grade: safety & regulatory differentiation
Waste-derived proteins face fundamentally different requirements depending on whether they are intended for animal feed or human food. Feed-grade proteins are subject to comparatively flexible regulatory frameworks, allowing the use of a wider range of pre-consumer organic residues and certain industrial by-products. Consequently, most commercial waste-to-protein systems currently target aquaculture, poultry, and livestock feeds.In contrast, food-grade proteins must comply with strict safety, traceability, and quality standards. Substrates must be free from hazardous contaminants, pathogens, heavy metals, and chemical residues, which severely restricts the use of post-consumer and mixed municipal waste. Additionally, food applications demand consistent sensory properties, low nucleic acid content (especially in microbial proteins), and allergen risk assessment.
Nucleic acid content is one of the most important technical barriers separating food- and feed-grade waste-derived microbial proteins. High nucleic acid intake is associated with elevated purine metabolism and uric acid formation in humans, increasing the risk of hyperuricemia, which has historically limited direct human consumption of untreated single-cell proteins. Consequently, food-grade microbial proteins are generally required to undergo nucleic acid reduction to levels commonly targeted below ∼2% of dry weight, based on toxicological and nutritional guidance, whereas feed-grade applications tolerate substantially higher nucleic acid contents.170 This requirement disproportionately constrains bacterial SCP, as bacterial biomass typically contains higher nucleic acid fractions than yeasts or filamentous fungi, reflecting their rapid growth and high ribosomal content.170 Achieving food-grade specifications, therefore, requires additional processing steps such as heat treatment, alkaline extraction, or enzymatic degradation, which increase production costs and reduce overall protein yield. As a result, yeast-based systems have dominated food-grade development pathways due to their inherently lower nucleic acid burden, greater consumer acceptance, and long history of regulatory approval, including the GRAS status for several species.170,171
Microbial safety represents a critical regulatory concern for waste-derived proteins. Microbiological safety requirements differ between food- and feed-grade waste-derived proteins. Food-grade applications require the absence of key pathogens such as Salmonella, Listeria monocytogenes, and pathogenic E. coli in defined sample sizes, necessitating stringent hygienic processing, validated kill steps (e.g., heat treatment and fermentation-based inactivation), and extensive batch testing.170,172 The use of BSFL as a protein source in animal feed is increasingly popular; however, the safety of both the larvae and the substrates used for their rearing remains insufficiently studied, despite its importance for economic viability. Comprehensive microbial analysis of black soldier fly larvae reared on common production substrates (kitchen waste, fruit waste, and brewer's waste) revealed that larvae generally had high microbial loads compared to substrates. However, safety indicator microbes, including Staphylococcus aureus, Salmonella typhi, Escherichia coli, and Bacillus cereus, remained within regulatory limits established by KEBS, Codex Alimentarius, and European Union standards.173 The study found low levels of S. typhi in BSFL samples from a brewery (50 ± 0 CFU g−1) and fruit waste (60 ± 5 CFU g−1) compared to other microbes, while E. coli, B. cereus, and S. aureus were present both in substrates and BSF larvae but within regulatory limits for edible insect products.173 Based on these results, the use of safe substrates such as kitchen and fruit waste for BSFL rearing is recommended.
Heavy metal accumulation and chemical contaminants represent significant safety considerations requiring regulatory oversight. Microorganisms bioaccumulate metals from waste substrates, particularly cadmium, lead, and mercury, at levels substantially higher (10–100 times) than those found in the original substrate. In the context of waste-derived proteins, food applications are subject to stringent contaminant limits imposed by food safety regulations to protect human health. For example, EU legislation (e.g., Commission Regulation (EU) 2023/915 and associated contaminant provisions) sets maximum levels for toxic elements such as cadmium and lead that vary by food category, reflecting differences in dietary exposure and vulnerability, and necessitates rigorous waste stream qualification, continuous monitoring, and robust risk assessment frameworks.174 In contrast, feed applications are regulated under a distinct set of standards (e.g., Directive 2002/32/EC on undesirable substances in feed), which generally allow higher tolerances for certain contaminants appropriate to animal physiology.175 Nonetheless, feed safety systems include traceability requirements and monitoring to prevent accumulation of contaminants in animal tissues and subsequent transfer into the human food chain. These dual regulatory tracks require protein producers to implement tailored quality control, analytical testing, and documentation systems that align with the specific safety objectives of food and feed regulatory frameworks.
Food-grade authorization generally requires extensive safety dossiers demonstrating toxicological safety (often including subchronic feeding and genotoxicity studies where relevant), allergenicity assessment through sequence homology and digestibility testing, nutritional evaluation relative to conventional proteins, and validation of production processes to ensure batch-to-batch consistency.170,172 In the European Union, approval under the Novel Food framework commonly requires several years and substantial financial and technical investment, whereas in the United States, GRAS determination can be faster but still depends on robust scientific evidence and expert consensus.176,177
Waste-derived proteins face additional regulatory scrutiny regarding traceability and source control. Regulatory authorities typically require detailed documentation of waste origin and handling to minimize contamination risks, making heterogeneous or poorly characterized waste streams—such as mixed municipal waste particularly challenging for food-grade authorization.
Consumer acceptance represents another major barrier. Products derived from “waste” or insects often evoke psychological resistance linked to disgust, perceived risk, and lack of familiarity. Perceptions of “waste-to-protein” concepts differ markedly across regions and should be carefully considered in technology deployment strategies.178 African and Southeast Asian countries are promising markets for insect-based bioconversion systems due to existing cultural acceptance of insects as food.179 In contrast, microbial fermentation technologies are already well established in Europe and North America, where products such as Quorn™ are widely accepted as mainstream foods, making these regions suitable for further expansion of microbial protein platforms. For global adoption, waste-derived protein products must be perceived as safe, high-quality, and trustworthy. Accordingly, conversion and upgrading processes must comply with food and feed chain requirements and adhere strictly to regulatory hygiene, quality, and safety standards.180
Feed-grade registration typically follows more streamlined regulatory pathways than food-grade approval, such as through The Association of American Feed Control Officials (AAFCO) definitions in the United States or inclusion under EU feed material and additive regulations.181 These pathways generally emphasize compositional analysis, safety evaluation, and, where necessary, target-species feeding trials, making approval timelines substantially more accessible than those for food-grade proteins.182 However, regulatory restrictions on allowable substrates significantly constrain the waste valorization potential of feed-grade systems. In the European Union, the use of certain waste streams—such as animal manures, some animal by-products, and post-consumer food waste—is restricted or prohibited for specific feed and insect-rearing applications, despite their often-high bioconversion efficiency. These constraints can force commercial operations to rely on cleaner but less circular substrates, reducing the environmental benefits of waste-to-protein systems.170
Regulatory distinctions between food- and feed-grade waste-derived proteins create bifurcated market pathways.183 Food-grade applications can command premium prices that may justify higher production and regulatory costs, but they face extended development timelines and continued uncertainty regarding consumer acceptance of ingredients derived from waste streams, even when framed within “upcycled food” narratives.170,184 In contrast, feed-grade applications typically access much larger and more established markets, but operate under commodity pricing structures that demand economies of scale, low-cost feedstocks, and high process efficiency to remain competitive.182,185
Strategic pathway selection in waste-derived protein systems must align regulatory requirements with substrate characteristics and production economics. Clean, well-characterized, and traceable industrial side-streams—such as spent yeast or ethanol stillage—tend to favor food-grade development pathways due to lower contamination risk and greater regulatory acceptability.186,187 In contrast, heterogeneous municipal or agricultural wastes, while technically capable of supporting protein production suitable for human nutrition, are under current regulatory and societal conditions more realistically directed toward feed or non-food applications because of traceability challenges and perceived safety risks.187,188 The long-term commercial viability of circular bioeconomy protein systems will therefore depend strongly on the evolution of regulatory frameworks, particularly regarding controlled waste substrate use, traceability, and acceptance of waste-to-food pathways.170,178,185
From a socio-economic perspective, waste-to-protein systems offer opportunities for job creation, regional waste valorization, and reduced dependence on imported protein crops. However, high capital costs and technical complexity may exclude small producers and concentrate benefits among large industrial actors. Without inclusive policy frameworks, these technologies risk reinforcing existing inequalities rather than supporting equitable food system transformation.
Thus, successful deployment of waste-derived proteins requires integrated consideration of safety, societal values, regulatory clarity, and economic inclusivity alongside technological development.
7. Challenges and knowledge gaps in waste-to-protein conversion
Waste-to-protein technologies are increasingly viewed as a cornerstone of circular and sustainable food systems, yet their large-scale deployment is constrained by a combination of technical, safety, regulatory, economic, and system-level challenges. A primary technical limitation arises from the intrinsic variability of waste feedstocks. Waste streams differ widely in composition depending on their origin, seasonality, and handling practices, leading to fluctuations in nutrient content, presence of inhibitors, and physical characteristics. This variability directly affects microbial or insect growth, protein yield, and product consistency, thereby undermining process stability and scalability. To address this, robust and flexible bioprocesses are needed, including mixed microbial consortia, adaptive process control, and effective pretreatment strategies to homogenize waste streams and buffer against fluctuations.45Safety is a critical barrier to acceptance and regulation of waste-derived proteins. Waste feedstocks may contain toxicants such as heavy metals, pesticides, and industrial chemicals that can bioaccumulate in microbial or insect biomass, posing acute and chronic health risks.189,190 In parallel, novel microbial and insect proteins may introduce allergenicity concerns, particularly for sensitive populations, necessitating systematic allergenicity assessment, appropriate labelling, and processing strategies to reduce allergenic potential.191 Waste substrates can also carry pathogenic microorganisms, increasing the risk of foodborne disease if not adequately controlled. This requires stringent sterilization and sanitation procedures, along with rapid and reliable detection methods for contaminants and pathogens.192,193 Together, these safety issues strongly influence regulatory decisions, public perception, and market acceptance.
A further limitation is the lack of standardized processing and quality assessment protocols. Differences in waste pretreatment, fermentation, harvesting, and protein extraction methods lead to substantial variation in nutritional quality, safety, and functional properties of the final products.194 Current quality assessment often emphasizes crude protein content, while neglecting parameters such as amino acid balance, digestibility, allergenicity, and techno-functional properties relevant for food formulation.195 The development of standardized protocols and rapid, cost-effective analytical tools is therefore essential for ensuring consistent quality, regulatory compliance, and industrial scalability.
Knowledge gaps also exist regarding the long-term stability and shelf-life of waste-derived proteins. Proteins are susceptible to degradation through oxidation, hydrolysis, and aggregation during storage and processing, which can reduce nutritional value, impair functionality, and compromise safety.196 Limited data are available on how different processing methods, packaging materials, and storage conditions affect stability. Systematic studies are needed to define optimal handling and storage strategies that ensure product quality throughout distribution and use.
Regulatory and certification frameworks represent another major barrier. Many regions lack clear and specific regulations for waste-derived proteins, creating uncertainty for producers, investors, and consumers.197 Existing certification systems are often poorly aligned with circular bioeconomy principles and do not adequately account for the unique features of waste-based feedstocks and processes.198 Clear, science-based regulations for safety, quality, and labelling are required, together with streamlined and harmonized certification pathways to facilitate market entry and international trade. Public skepticism toward products derived from “waste” further influences policy support and regulatory stringency, emphasizing the importance of transparent communication, education, and stakeholder engagement.199
Economic and market-related challenges remain substantial. Production costs of waste-derived proteins are often higher than those of conventional protein sources due to energy-intensive operations, complex pretreatment steps, and high capital investment for specialized infrastructure.200 Scaling up requires significant financial resources, automation, and skilled labor, while current production capacities are still limited.197 In addition, reliable supply chains are difficult to establish because waste feedstocks are geographically dispersed, seasonally variable, and costly to collect and transport. Efficient logistics, diversified feedstock sourcing, and supportive policy incentives are therefore essential to improve economic competitiveness.197,201
At the system level, many waste-to-protein platforms fail to fully valorize all components of waste streams, leaving residual fractions that still require disposal and reducing overall sustainability.202 Integrated biorefinery concepts that combine protein production with generation of fuels, fertilizers, or biochemicals, as well as cascading use of by-products, are needed to maximize resource efficiency. Limited coordination among waste generators, processors, and end-users further restricts circularity and leads to fragmented value chains.203 Moreover, comprehensive life cycle assessment (LCA) data are still scarce, making it difficult to quantify true environmental benefits and trade-offs across technologies.204 Standardized LCA methodologies are needed to enable transparent comparison and informed policy and investment decisions.
Finally, although waste-to-protein systems have the potential to reduce land use, waste disposal, and greenhouse gas emissions relative to conventional protein production, their environmental performance varies widely across technologies and contexts.199 Energy demand particularly for aeration, drying, agitation, and lighting remains a major sustainability bottleneck.196 Energy recovery from waste streams and integration of renewable energy sources are therefore crucial to improve environmental performance. Efficient water management through recycling and reuse is also necessary, as many systems remain water-intensive.199,204 Without careful optimization guided by robust LCA, waste-to-protein technologies risk shifting environmental burdens rather than delivering genuine sustainability gains.
8. Waste-derived protein development: near-term deployment vs. long-term transformation
The temporal dimension of waste-derived protein development reveals a critical strategic divergence between immediately deployable solutions and transformative future technologies. Near-term deployment pathways leverage existing industrial infrastructure, established microbial platforms, and regulatory frameworks to achieve rapid market entry with minimal disruption. Yeast-based SCP systems represent the most commercially mature microbial protein pathways, benefiting from decades of industrial fermentation experience and established regulatory acceptance, including the GRAS status for widely used species such as Saccharomyces cerevisiae and long-standing food and feed use of Candida utilis.Industrial side-streams such as spent yeast from brewing and bioethanol operations, ethanol stillage, and whey from dairy processing are produced in high volumes and originate from well-defined, traceable processes, making them attractive as feedstocks for valorization into protein products.186,205 These streams are generally easier to document and characterize than heterogeneous waste materials, which can facilitate regulatory risk assessments for feed and, in some cases, food applications when appropriate safety controls are implemented (e.g., contaminant monitoring and nucleic acid reduction). However, compatibility with food-grade regulatory requirements still depends on demonstrated safety, consistency, and processing controls rather than on feedstock origin alone. Several studies demonstrate that mixed yeast cultures can simultaneously remove organic pollutants and generate protein-rich biomass from waste streams. For example, mixed cultures of Candida utilis, Rhodotorula mucilaginosa, and Kluyveromyces marxianus grown on tequila vinasse achieved superior pollutant removal and high biomass yield compared with monocultures, illustrating the combined potential for waste treatment and SCP production.206 Mixed yeast fermentations using whey also showed enhanced chemical oxygen demand removal and increased SCP productivity relative to single strains, further supporting the use of mixed cultures for integrated valorization of organic wastes.207
Among waste-derived alternative protein technologies, insect-based systems, particularly BSFL, have reached some of the highest TRLs.208 Commercial insect meal production is already established in multiple regions, especially for animal feed, and regulatory frameworks in jurisdictions such as the European Union, the United States, Canada, and Australia now permit specific insect proteins for use in aquaculture and other livestock feeds. Regulatory approval for insect-derived proteins—especially from black soldier fly larvae (Hermetia illucens)—has been achieved in multiple jurisdictions for use in animal feeds, including aquaculture and, increasingly, poultry and pig feed under evolving regulatory frameworks. In the European Union, processed insect proteins have been permitted in aquaculture since 2017 and in poultry and pig feeds since regulatory updates in 2021.209Hermetia illucens larvae typically contain high levels of protein—often reported in the ∼40–60% dry matter range making them a valuable alternative to conventional protein sources in feed applications.210 Insect-based systems are being actively scaled through coordinated research and innovation initiatives. For example, the EU Horizon 2020 project SUStainable INsect CHAIN (SUSINCHAIN, 2019–2023) focused on improving rearing practices, processing, and value-chain coordination to support upscaling of insect protein, primarily for feed applications. Such initiatives illustrate how targeted projects are helping move insect-based waste-to-protein systems from pilot toward early commercial readiness.208,211
Algal systems integrated into wastewater treatment represent a near-term pathway for combined nutrient removal and biomass generation. High-rate algal ponds and rotating/attached algal biofilm concepts are primarily deployed for nutrient removal, but they also generate algal biomass that can be valorized, with reported protein contents commonly in the ∼30–50% dry-weight range and reaching ∼50% or higher under favorable species selection and operating conditions.212–215
Long-term pathways in waste-to-protein research target breakthrough innovations that address fundamental limitations of near-term approaches, particularly substrate flexibility, production cost, and direct food applications. Bacterial SCP systems utilizing gaseous substrates (e.g., CO2, H2, CH4, and syngas) are widely viewed as a transformative opportunity, although they remain at relatively early technology readiness levels due to engineering complexity and regulatory uncertainty.216,217 Emerging studies on co-cultivation using biogas-derived substrates suggest potential improvements in biomass yield and amino acid profiles compared to monocultures, indicating a possible route toward nutritionally competitive microbial protein. Co-cultivation of MOB and HOB on biogas + H2/O2 enabled stable mixed cultures and showed synergistic growth compared with monocultures. Several MOB–HOB combinations produced 1.1–3.8-fold higher protein concentrations and up to 6.1-fold higher essential amino acid content. The best consortium (Methyloparacoccus murrelli with Cupriavidus necator) generated nutritionally competitive microbial protein requiring substantially less biomass than meat, eggs, or tofu to meet amino acid needs. These results demonstrate that MOB–HOB co-cultivation offers a promising route for sustainable microbial protein production from safe, waste-derived gaseous substrates.218 In principle, such systems enable protein production independent of arable land and conventional agriculture, but their large-scale food application will depend on advances in nucleic acid reduction technologies and clearer regulatory pathways.
Precision fermentation platforms based on genetically engineered microorganisms represent a long-term pathway for waste-to-protein valorization, with the potential to convert waste-derived substrates into high-value recombinant or functional proteins.219 Although such systems are technically feasible and already applied at the industrial scale on refined feedstocks, their extension to waste-based substrates and direct food applications faces significant regulatory and societal barriers, particularly regarding the approval and acceptance of genetically modified organisms.
Fungal SSF has been widely explored for valorizing lignocellulosic agricultural residues, with numerous studies demonstrating increased protein content and improved nutritional profiles following fungal bioconversion of substrates such as brewer spent grain, rice straw, and corn stover.220 Studies highlight SSF's potential as a sustainable circular bioeconomy strategy for transforming waste into value-added bioproducts, while also noting technical challenges related to moisture control, heat transfer, and process optimization that currently constrain industrial scale-up.221
Integrated/cascading biorefineries are widely framed as a long-term circular bioeconomy strategy, converting waste/biomass into a portfolio of products (fuels, chemicals, materials and other co-products) to maximize value recovery.222 Techno-economic studies frequently report improved feasibility for integrated multi-product schemes relative to single-product pathways, but emphasize that commercialization depends on advanced process integration and separations, robust process control, and market development for multiple outputs.223
The near-term vs. long-term distinction has strategic implications: near-term systems can be deployed more rapidly but are constrained by substrate restrictions, feed-market focus, and incremental cost gains, whereas long-term systems may deliver larger sustainability benefits (e.g., reduced land dependence and broader food-grade applications) but require sustained investment and regulatory clarity before reaching the commercial scale.208,224
Policy frameworks must accommodate this temporal duality through differentiated support mechanisms: streamlined regulatory pathways and infrastructure investment for near-term systems; fundamental research funding and pilot-scale demonstration facilities for long-term technologies. The successful transition to waste-derived protein systems will ultimately depend on maintaining parallel development pathways that capitalize on near-term opportunities while simultaneously advancing transformative innovations that redefine sustainable food production for the coming decades.
9. Conclusions
The waste to resource conversion stands as a fundamental pillar in advancing circular economy principles within sustainable food systems. The substantial generation of organic wastes and by-products across the agri-food sector presents both an immense challenge and a strategic opportunity to reconfigure these materials as valuable feedstocks for alternative protein production.Waste valorization pathways for alternative proteins should be recognized not merely as supplementary sustainability measures but as central components to circular food economies. Novel protein sources derived from insect bioconversion, microbial fermentation, and the refinement of agricultural and industrial residues exemplify this paradigm shift. These processes internalize waste management within protein production, challenging the linear “take-make-dispose” model and fostering regenerative systems capable of continuous nutrient cycling.
Realizing the full potential of alternative proteins from waste streams mandates a concerted interdisciplinary approach that bridges food science, biotechnology, and circular economy policy. Innovations in biotechnology enable optimized microbial and insect-based protein production, while advances in food science address critical issues of safety, sensory quality, and nutritional adequacy. Concurrently, enabling policy frameworks are essential to support research translation, promote regulatory harmonization, and incentivize circular practices at scale. Coordinated collaboration among stakeholders across academia, industry, and governance will foster the development of resilient, equitable, and transparent food systems aligned with global sustainability agendas.
While waste-to-protein technologies offer powerful tools for building sustainable and circular food systems, their impact will depend on realistic deployment strategies. Near-term emphasis should be placed on scalable, safe, and socially acceptable systems, particularly for animal feed and selected food applications. Longer-term innovations must continue to be supported, but without delaying immediate action on available solutions. Only by aligning technological progress with regulatory readiness, consumer trust, and socio-economic inclusion can waste-derived proteins move from promising concepts to mainstream contributors to global food security.
The development and adoption of alternative proteins from waste streams can contribute to the achievement of several Sustainable Development Goals (SDGs), providing a pathway towards a more sustainable and equitable food system. These encompass SDG 2 (Zero Hunger), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), highlighting the multifaceted benefits of alternative proteins in addressing global challenges. By providing sustainable protein sources, reducing environmental impacts, and promoting resource efficiency, alternative proteins can help achieve a more sustainable and equitable food system that supports both human nutrition and ecological health.
Author contributions
Sunny Dhiman: conceptualisation, writing – original draft, writing – review & editing, visualization, software, methodology, investigation, data curation, formal analysis; Gunjan Mukherjee: writing – review & editing, supervision, project administration, formal analysis; Anu Kumar: formal analysis; Bhanu Krishan: formal analysis.Conflicts of interest
There are no conflicts to declare.Data availability
No datasets were generated or analyzed during the current study.References
- U. Nations, World population projected to reach 9.8 billion in 2050, and 11.2 billion in 2100, https://www.un.org/en/desa/world-population-projected-reach-98-billion-2050-and-112-billion-2100, accessed 25 September 2025.
- FAO, The Future of Food and Agriculture – Trends and Challenges, Food and Agriculture Organization of the United Nations, Rome, 2017 Search PubMed.
- S. Jafarzadeh, Z. Qazanfarzadeh, M. Majzoobi, S. Sheiband, N. Oladzadabbasabad, Y. Esmaeili, C. J. Barrow and W. Timms, Curr. Res. Food Sci., 2024, 9, 100882 CrossRef CAS PubMed.
- M. Nikbakht Nasrabadi, A. Sedaghat Doost and R. Mezzenga, Food Hydrocoll., 2021, 118, 106789 CrossRef CAS.
- J. H. Chiang, W. Tay, D. S. M. Ong, D. Liebl, C. P. Ng and C. J. Henry, Food Struct., 2021, 28, 100183 CrossRef CAS.
- P. Kumar, A. A. Abubakar, A. K. Verma, P. Umaraw, M. Adewale Ahmed, N. Mehta, M. Nizam Hayat, U. Kaka and A. Q. Sazili, Crit. Rev. Food Sci. Nutr., 2023, 63, 11830–11858 CrossRef CAS PubMed.
- A. Espinosa-Marrón, K. Adams, L. Sinno, A. Cantu-Aldana, M. Tamez, A. Marrero, S. N. Bhupathiraju and J. Mattei, Front. Sustain., 2022, 3, 841106 CrossRef.
- K. B. Kc, G. M. Dias, A. Veeramani, C. J. Swanton, D. Fraser, D. Steinke, E. Lee, H. Wittman, J. M. Farber, K. Dunfield, K. McCann, M. Anand, M. Campbell, N. Rooney, N. E. Raine, R. V. Acker, R. Hanner, S. Pascoal, S. Sharif, T. G. Benton and E. D. G. Fraser, PLoS One, 2018, 13, e0205683 CrossRef PubMed.
- L. B. Safdar, M. J. Foulkes, F. H. Kleiner, I. R. Searle, R. A. Bhosale, I. D. Fisk and S. A. Boden, Plant Commun., 2023, 4, 100716 CrossRef CAS PubMed.
- K. Dave, A. Kumar, N. Dave, M. Jain, P. S. Dhanda, A. Yadav and P. Kaushik, Sustainability, 2024, 16, 6026 Search PubMed.
- J. Zwinkels, J. Wolkers-Rooijackers and E. J. Smid, LWT, 2023, 184, 114979 Search PubMed.
- State of Alternative Protein series – The Good Food Institute, https://gfi.org/state-of-alternative-proteins/, accessed 27 September 2025.
- Alternative Protein Market Size & Share, Statistics Report 2034, https://www.gminsights.com/industry-analysis/alternative-protein-market, accessed 27 September 2025.
- F. R. P. Cedeno, O. J. Olubiyo and S. Ferreira, J. Biol. Eng., 2025, 19, 44 CrossRef CAS PubMed.
- O. A. Oladejo, O. Akintaro, M. O. Ayoola, P. E. Kolawole, A. O. Oguntunji and O. M. Alabi, Niger. J. Anim. Prod., 2024, 1362–1365 Search PubMed.
- S. I. Patsios, A. Dedousi, E. N. Sossidou and A. Zdragas, Sustainability, 2020, 12, 1398 CrossRef CAS.
- M. Gil, M. Rudy, P. Duma-Kocan, R. Stanisławczyk, A. Krajewska, D. Dziki and W. H. Hassoon, Sustainability, 2024, 16, 7701 Search PubMed.
- P. Sangiorgio, A. Verardi, S. Dimatteo, A. Spagnoletta, S. Moliterni and S. Errico, J. Insects Food Feed, 2022, 8, 509–524 Search PubMed.
- A. Skendi, K. G. Zinoviadou, M. Papageorgiou and J. M. Rocha, Foods, 2020, 9, 1243 Search PubMed.
- I. Jain, R. Kaur, A. Kumar, M. Paul and N. Singh, Int. J. Food Sci. Technol., 2024, 59, 6797–6820 Search PubMed.
- S. Dhiman, B. Thakur, S. Kaur, M. Ahuja, S. Gantayat, S. Sarkar, R. Singh and M. Tripathi, Dispos. Sustain., 2025, 6, 258 Search PubMed.
- S. Dhiman, A. Kumar, K. Sharma and T. Dhewa, Indian J. Microbiol., 2025, 65(3), 1452–1483 Search PubMed.
- P. Pal, A. K. Singh, R. K. Srivastava, S. S. Rathore, U. K. Sahoo, S. Subudhi, P. K. Sarangi and P. Prus, Foods, 2024, 13, 3007 Search PubMed.
- N. Nirmal, C. F. Anyimadu, A. C. Khanashyam, A. E. A. Bekhit and B. K. Dhar, Sustain. Dev., 2025, 33, 3958–3969 CrossRef.
- I. V. Dos Anjos, N. Coelho, H. Duarte, D. N. Proença, M. F. Duarte, R. Barros, S. Raposo, S. Gonçalves, A. Romano and B. Medronho, Polymers, 2025, 17, 2251 CrossRef CAS PubMed.
- G. Venkatesh, Circ. Econ. Sustain., 2022, 2, 231–279 CrossRef.
- H. Wang, I. Seekamp, A. Malzahn, A. Hagemann, A. K. Carvajal, R. Slizyte, I. B. Standal, A. Handå and K. I. Reitan, Aquaculture, 2019, 502, 232–241 CrossRef.
- K. M. Eggink, I. Lund, P. B. Pedersen, B. W. Hansen and J. Dalsgaard, PLoS One, 2022, 17, e0275213 CrossRef CAS PubMed.
- N. Pliantiangtam, P. Chundang and A. Kovitvadhi, Insects, 2021, 12, 682 CrossRef PubMed.
- M. Von Massow, K. Parizeau, M. Gallant, M. Wickson, J. Haines, D. W. L. Ma, A. Wallace, N. Carroll and A. M. Duncan, Front. Nutr., 2019, 6, 143 CrossRef PubMed.
- V. Narisetty, N. Adlakha, N. Kumar Singh, S. K. Dalei, A. A. Prabhu, S. Nagarajan, A. Naresh Kumar, J. Amruthraj Nagoth, G. Kumar, V. Singh and V. Kumar, Bioresour. Technol., 2022, 363, 127856 CrossRef CAS PubMed.
- X. Cao, P. N. Williams, Y. Zhan, S. A. Coughlin, J. W. McGrath, J. P. Chin and Y. Xu, Soil & Environ. Health, 2023, 1, 100038 Search PubMed.
- M. Logan and C. Visvanathan, Waste Manag. Res., 2019, 37, 27–39 CrossRef CAS PubMed.
- G. Šelo, M. Planinić, M. Tišma, S. Tomas, D. Koceva Komlenić and A. Bucić-Kojić, Foods, 2021, 10, 927 CrossRef PubMed.
- C. H. Edwards, P. Ryden, A. M. Pinto, A. Van Der Schoot, C. Stocchi, N. Perez-Moral, P. J. Butterworth, B. Bajka, S. E. Berry, S. E. Hill and P. R. Ellis, J. Funct.Foods, 2020, 68, 103918 CrossRef CAS.
- R. N. Gavril (Raţu), F. Stoica, F. D. Lipşa, O. E. Constantin, N. Stănciuc, I. Aprodu and G. Râpeanu, Foods, 2024, 13, 2694 CrossRef PubMed.
- A. H. L. Wan, S. J. Davies, A. Soler-Vila, R. Fitzgerald and M. P. Johnson, Rev. Aquacult., 2019, 11, 458–492 CrossRef.
- T. Varzakas, G. Zakynthinos and F. Verpoort, Foods, 2016, 5, 88 CrossRef PubMed.
- J. Gaffey, G. Rajauria, H. McMahon, R. Ravindran, C. Dominguez, M. Ambye-Jensen, M. F. Souza, E. Meers, M. M. Aragonés, D. Skunca and J. P. M. Sanders, Biotechnol. Adv., 2023, 66, 108168 CrossRef CAS PubMed.
- M. Abdurrazaq, M. A. Muhammad and B. B. Maidambe, UMYU J. Microbiol. Res., 2025, 10, 215–220 Search PubMed.
- Y. Liu, W. R. Aimutis and M. Drake, Foods, 2024, 13, 1010 CrossRef CAS PubMed.
- A. Abedfar, F. Abbaszadeh and F. Mardiha, Chem. Biomol. Eng., 2025, 10, 8–15 Search PubMed.
- D. Bertasini, R. L. Binati, D. Bolzonella and F. Battista, Chemosphere, 2022, 296, 134076 CrossRef CAS PubMed.
- A. Tropea, A. Ferracane, A. Albergamo, A. G. Potortì, V. Lo Turco and G. Di Bella, Fermentation, 2022, 8, 91 CrossRef CAS.
- T. Ayodele, A. Tijani, M. Liadi, K. Alarape, C. Clementson and A. Hammed, Front. Biosci., Elite Ed., 2024, 16, 40 CrossRef CAS PubMed.
- P. Thiviya, R. Kapilan and T. Madhujith, Trop. Agric. Res., 2021, 32, 503 Search PubMed.
- A. E. Akinduyite, G. O. Abu and O. K. Agwa, Adv. Microbiol., 2022, 51–63 CrossRef.
- T. Deuchande, J. F. Fundo, D. Rodrigues, M. E. Pintado and A. L. Amaro, Waste Biomass Valoriz., 2024, 15, 3553–3567 CrossRef CAS.
- C. D. Lynch and D. J. O'Connell, PLoS One, 2022, 17, e0266921 CrossRef CAS PubMed.
- D. Díaz-Vázquez, M. V. Garibay, A. Fernández Del Castillo, D. A. Orozco-Nunnelly, C. Senés-Guerrero and M. S. Gradilla-Hernández, Front. Chem. Eng., 2022, 4, 1013873 Search PubMed.
- S. Tsakona, A. Papadaki, N. Kopsahelis, V. Kachrimanidou, S. Papanikolaou and A. Koutinas, Foods, 2019, 8, 300 Search PubMed.
- P. L. Tang and W. H. Wong, J. Sains Malays., 2024, 53, 3383–3394 CrossRef CAS.
- R. Gao, Q. He, Y. Chen, Y. Duan and L. Xie, IOP Conf. Ser. Earth Environ. Sci., 2018, 171, 012035 CrossRef.
- V. Braho, T. Sar and M. J. Taherzadeh, Syst. Microbiol. Biomanuf., 2024, 4, 675–686 Search PubMed.
- M. Al-Farsi, A. Al Bakir, H. A. Marzouqi and R. Thomas, Production of single cell protein from date waste, in By-Products of Palm Trees and Their Applications, Millersville, PA, 2019, DOI:10.21741/9781644900178-26.
- O. Z. Wada, U. Onwusogh, A. S. Vincent, G. Mckay and H. R. Mackey, Biomass Conv. Bioref., 2023, 13, 16569–16583 CrossRef CAS.
- A. Poladyan, S. Blbulyan, M. Sahakyan, O. Lenz and A. Trchounian, Microb. Cell Fact., 2019, 18, 201 CrossRef CAS PubMed.
- R. K. Le, T. Wells Jr, P. Das, X. Meng, R. J. Stoklosa, A. Bhalla, D. B. Hodge, J. S. Yuan and A. J. Ragauskas, RSC Adv., 2017, 7, 4108–4115 RSC.
- F.-M. Kerckhof, M. Sakarika, M. Van Giel, M. Muys, P. Vermeir, J. De Vrieze, S. E. Vlaeminck, K. Rabaey and N. Boon, Front. Bioeng. Biotechnol., 2021, 9, 733753 CrossRef PubMed.
- M. Liu, S. Huang, P. Yan, X. Wu, H. Yin and L. Wang, Appl. Microbiol. Biotechnol., 2025, 109, 157 Search PubMed.
- L. M. Gómez Martínez, A. Rodriguez Montaña, J. J. Martínez, L. Y. Rache Cardenal and L. M. Borrás Sandoval, Trop. Subtrop. Agroecosyst., 2025, 28(2), 115 Search PubMed.
- L. Stasiak-Różańska, J. Gawor, K. Piwowarek, A. Fabiszewska and T. Aleksandrzak-Piekarczyk, Foods, 2025, 14, 1573 CrossRef PubMed.
- P. Klüber, F. F. Gurusinga, S. Hurka, A. Vilcinskas and D. Tegtmeier, Appl. Environ. Microbiol., 2024, 90, e00991–24 CrossRef PubMed.
- M. Shumo, F. M. Khamis, F. L. Ombura, C. M. Tanga, K. K. M. Fiaboe, S. Subramanian, S. Ekesi, O. K. Schlüter, A. Van Huis and C. Borgemeister, Front. Microbiol., 2021, 12, 687103 CrossRef PubMed.
- K. Rasool, S. Hussain, A. Shahzad, W. Miran, K. A. Mahmoud, N. Ali and F. Almomani, Rev. Environ. Sci. Biotechnol., 2023, 22, 527–562 CrossRef CAS.
- E. E. Ziganshina, S. S. Bulynina, K. A. Yureva and A. M. Ziganshin, Plants, 2022, 11, 3583 CrossRef CAS PubMed.
- M. Su, M. Dell'Orto, B. Scaglia, G. D'Imporzano, A. Bani and F. Adani, Molecules, 2022, 27, 422 CrossRef CAS PubMed.
- S. Y. Lee and D. C. Stuckey, J. Clean. Prod., 2022, 357, 131975 Search PubMed.
- G. Proietti Tocca, V. Agostino, B. Menin, T. Tommasi, D. Fino and F. Di Caprio, Rev. Environ. Sci. Biotechnol., 2024, 23, 93–132 CrossRef.
- D. De Francisci, Y. Su, A. Iital and I. Angelidaki, Environ. Technol., 2018, 39, 581–592 CrossRef CAS PubMed.
- I. Khan, D. D. Gupta and A. Gupta, Nat. Environ. Pollut. Technol., 2024, 23, 1801–1816 CrossRef CAS.
- J. Pierson, G. R. Makkena, S. Kumar, V. Kumar, V. Vivekanand, H. Husain, M. Ayser and V. Balan, Fermentation, 2023, 9, 909 CrossRef CAS.
- M. Saleh, A. Al-Zayat, Z. Sharawy and T. Azzam, Egypt. J. Nutr. Feeds, 2023, 26, 469–487 CrossRef.
- M. Kaombo, A. W. Mayo, R. Kimwaga and S. Gabrielsson, Tanz. J. Engrg. Technol., 2019, 38, 130–151 CrossRef.
- O. K. Mosibo, G. Ferrentino and C. C. Udenigwe, Foods, 2024, 13, 733 CrossRef CAS PubMed.
- B. Koç Bilican and A. S. Maruška, Mühendis. Bilim. Tasar. Derg., 2024, 12, 132–139 CrossRef.
- A. Singh, V. Anand, A. Bai, Z. Gabnai and S. K. Prajapati, Period. Polytech.–Chem. Eng., 2023, 67, 355–366 CrossRef CAS.
- I. M. Bradley, Y. Li and J. S. Guest, Environ. Sci. Technol., 2021, 55, 12574–12584 CrossRef CAS PubMed.
- M. Buccaro, A. Toscano, M. Balzarotti, I. Re, D. Bosco and M. Bettiga, Sustainability, 2023, 15, 2195 CrossRef.
- M. G. Ahmed, S. A. Gouda, S. Donia and N. M. Hassanein, Biomass Conv. Bioref., 2025, 15, 5447–5462 CrossRef CAS.
- H. Nadeem, Futuristic Biotechnol., 2021, 09–13 CrossRef.
- D. Sousa, A. Venâncio, I. Belo and J. M. Salgado, J. Sci. Food Agric., 2018, 98, 5248–5256 CrossRef CAS PubMed.
- M. Asadollahzadeh, A. Ghasemian, A. Saraeia, H. Resalati and M. J. Taherzadeh, BioResources, 2018, 13, 5013–5031 CAS.
- S. Ahmed, G. Mustafa, M. Arshad and M. I. Rajoka, BioMed Res. Int., 2017, 2017, 1–9 Search PubMed.
- T. Sar, C. Uwineza, M. J. Taherzadeh and A. Mahboubi, Biotechnology, 2025, 14, 76 CrossRef PubMed.
- S. Sajad Hashemi, K. Karimi and M. J. Taherzadeh, Bioresour. Technol., 2021, 337, 125356 CrossRef CAS PubMed.
- R. B. Nair, M. M. Kabir, P. R. Lennartsson, M. J. Taherzadeh and I. S. Horváth, Appl. Biochem. Biotechnol., 2018, 184, 48–62 CrossRef CAS PubMed.
- J. He, M. Tang, F. Zhong, J. Deng, W. Li, L. Zhang, Q. Lin, X. Xia, J. Li and T. Guo, Crit. Rev. Biotechnol., 2024, 44, 1515–1532 CrossRef CAS PubMed.
- M. H. Aminuddin, N. H. Elias, J. H. Moh and N. Zainalabidin, Int. J. Biomass Util. Sustain. Energy, 2023, 1, 39–50 Search PubMed.
- T. Veldkamp, K. Van Rozen, H. Elissen, P. Van Wikselaar and R. Van Der Weide, Animals, 2021, 11, 3082 CrossRef PubMed.
- M. Bothma, G. M. Teke, E. Pieterse, D. Diedericks, E. Van Rensburg, J. F. Görgens and R. Den Haan, J. Insects Food Feed, 2024, 11, 937–951 Search PubMed.
- J. Y. Kim, J. Park, D.-J. Lee, Y.-B. Choi, Y. Choi, W.-K. Park, B. Koo, K. Park, D. Lee and E. E. Kwon, npj Sci. Food, 2025, 9, 65 CrossRef PubMed.
- T. Uushona, A. Simasiku and N. P. Petrus, Welwitschia Int. J. Agric. Sci., 2019, 1, 35–40 Search PubMed.
- J. Liu, H. Li, Z. Liu, D. Xu, X. Zhu, L. Hu and H. Mo, Entomol. Exp. Appl., 2025, 173, 616–626 Search PubMed.
- S. Lu, N. Taethaisong, W. Meethip, J. Surakhunthod, B. Sinpru, T. Sroichak, P. Archa, S. Thongpea, S. Paengkoum, R. A. P. Purba and P. Paengkoum, Insects, 2022, 13, 831 CrossRef PubMed.
- G. Bosch, S. Zhang, D. G. A. B. Oonincx and W. H. Hendriks, J. Nutr. Sci., 2014, 3, e29 CrossRef PubMed.
- V. Maurer, M. Holinger, Z. Amsler, B. Früh, J. Wohlfahrt, A. Stamer and F. Leiber, J. Insects Food Feed, 2016, 2, 83–90 Search PubMed.
- X. Li, Y. Dong, Q. Sun, X. Tan, C. You, Y. Huang and M. Zhou, Animals, 2022, 12, 486 CrossRef PubMed.
- M. Cullere, G. Tasoniero, V. Giaccone, G. Acuti, A. Marangon and A. Dalle Zotte, Animal, 2018, 12, 640–647 CrossRef CAS PubMed.
- M. Cullere, G. Tasoniero, V. Giaccone, R. Miotti-Scapin, E. Claeys, S. De Smet and A. Dalle Zotte, Animal, 2016, 10, 1923–1930 CrossRef CAS PubMed.
- A. Di Pasquale, M. Zoccola, A. Mohod, G. Dalla Fontana, A. Anceschi and S. Dalle Vacche, Polymers, 2025, 17, 1582 CrossRef CAS PubMed.
- R. Rosa, R. Spinelli, P. Neri, M. Pini, S. Barbi, M. Montorsi, L. Maistrello, A. Marseglia, A. Caligiani and A. M. Ferrari, ACS Sustainable Chem. Eng., 2020, 8, 14752–14764 CrossRef CAS.
- C. Rashama, E. M. Motsetsi, R. Christian and T. S. Matambo, Energy Sci. Eng., 2024, 12, 3986–3994 CrossRef CAS.
- M. Psarianos, R. Schneider, Ö. Altuntas, G. Dimopoulos, P. Taoukis and O. K. Schlüter, Biofuel Res. J., 2024, 11, 2211–2224 CrossRef CAS.
- T. Hahn, E. Tafi, A. Paul, R. Salvia, P. Falabella and S. Zibek, J. Chem. Technol. Biotechnol., 2020, 95, 2775–2795 CrossRef CAS.
- T. Hahn, E. Tafi, N. Von Seggern, P. Falabella, R. Salvia, J. Thomä, E. Febel, M. Fijalkowska, E. Schmitt, L. Stegbauer and S. Zibek, Waste Biomass Valoriz., 2022, 13, 1993–2008 CrossRef CAS.
- A. Vauterin, B. Steiner, J. Sillman and H. Kahiluoto, J. Clean. Prod., 2021, 320, 128799 CrossRef.
- S. Smetana, E. Schmitt and A. Mathys, Resour. Conserv. Recycl., 2019, 144, 285–296 CrossRef.
- P. J. Shafer, Y. H. Chen, T. Reynolds and E. J. B. Von Wettberg, Front. Sustain. Food Syst., 2022, 5, 721985 CrossRef.
- S. Lievens, G. Poma, J. De Smet, L. Van Campenhout, A. Covaci and M. Van Der Borght, J. Insects Food Feed, 2021, 7, 383–396 Search PubMed.
- P. Schrögel and W. Wätjen, Foods, 2019, 8, 288 CrossRef PubMed.
- A. Hardy, D. Benford, H. P. J. M. Noteborn, T. I. Halldorsson, J. Schlatter, R. A. Solecki, M. Jeger, H. K. Knutsen, S. More, A. Mortensen, H. Naegeli, C. Ockleford, A. Ricci, G. Rychen, V. Silano and D. Turck, EFSA J. , 2015, 13(10), 4257 CrossRef.
- A. Van Huis, Agric. Food Secur., 2015, 4, 20 CrossRef.
- F. A. Madau, B. Arru, R. Furesi and P. Pulina, Sustainability, 2020, 12, 5418 CrossRef.
- S. Cappellozza, M. G. Leonardi, S. Savoldelli, D. Carminati, A. Rizzolo, G. Cortellino, G. Terova, E. Moretto, A. Badaile, G. Concheri, A. Saviane, D. Bruno, M. Bonelli, S. Caccia, M. Casartelli and G. Tettamanti, Animals, 2019, 9, 278 CrossRef PubMed.
- A. Shirvanyan, A. Daniyarova, A. Vassilian, A. Poladyan, G. Kumar, D. Orynbekov, K. Bekbayev and K. Trchounian, BMC Biotechnol., 2025, 25, 136 CrossRef CAS PubMed.
- C. Hermansen, R. Siao, G. G. Chua, M. R. X. Lee, A. Thong, M. Weingarten, N. Lindley and E. C. Peterson, Microorganisms, 2025, 13, 1558 CrossRef CAS PubMed.
- A. Lalić, J. Jagelavičiūtė, T. Rezić, Z. Trivunović, D. Žadeikė and L. Bašinskienė, Sustainability, 2025, 17, 782 Search PubMed.
- G. Bayon-Vicente, L. Toubeau, M. Gilson, G. Gégo, N. Landgey, S. Krings and B. Leroy, Front. Bioeng. Biotechnol., 2025, 13, 1529032 Search PubMed.
- R. Lo Coco, M. Pezzuto, A. Jelic, S. Cazzaniga and N. Frison, Bioresour. Technol., 2026, 440, 133413 CrossRef CAS PubMed.
- F. Li, D. K. Amenorfenyo, Y. Zhang, N. Zhang, C. Li and X. Huang, Front. Environ. Sci., 2021, 9, 770633 Search PubMed.
- A. Silkina, J. I. Gayo-Peláez, F. Fernandes, C. Fuentes-Grünewald, R. V. Kapoore and K. W. Tang, J. Appl. Phycol., 2025, 37, 1003–1012 Search PubMed.
- F. Pratama, R. T. Rahardja, A. R. Rachmadi, W. Q. Salam, K. Kho, A. Adelie and P. V. P. Devanthi, J. Fungi, 2025, 11, 349 Search PubMed.
- J. Toghiani, N. Fallah, B. Nasernejad, A. Mahboubi, M. J. Taherzadeh and N. Afsham, Sci. Rep., 2025, 15, 5873 Search PubMed.
- Z. Gaizauskaite and D. Zadeike, Appl. Sci., 2025, 15, 13165 CrossRef CAS.
- S. I. Alqurashi, Front. Sustain. Food Syst., 2025, 9, 1689902 CrossRef.
- N. F. N. M. Zulkifli, A. Y. Seok-Kian, L. L. Seng, S. Mustafa, Y.-S. Kim and R. Shapawi, PLoS One, 2022, 17, e0263924 CrossRef CAS PubMed.
- H. T. Nguyen, H. N. D. Bao, H. T. T. Dang, T. Tómasson, S. Arason and M. Gudjónsdóttir, Foods, 2022, 11, 1531 CrossRef CAS PubMed.
- E. Cadena, O. Kocak, J. Dewulf, I. Undeland and M. Abdollahi, Sustainability, 2025, 17, 323 CrossRef CAS.
- V. Venugopal and A. Sasidharan, Front. Nutr., 2022, 9, 974447 CrossRef PubMed.
- O. L. Awuor, M. Edward Kirwa, M. Betty and M. F. Jackim, Ind. Chem., 2017, 3(1) DOI:10.4172/2469-9764.1000122.
- K. Chiodza and N. J. Goosen, Biomass Conv. Bioref., 2024, 14, 24069–24080 CrossRef CAS.
- F. Al Khawli, N. Pallarés, F. J. Martí-Quijal, E. Ferrer and F. J. Barba, Appl. Sci., 2021, 11, 2160 CrossRef CAS.
- J. Cropotova, K. Kvangarsnes, J. Stangeland and T. Rustad, Front. Sustain. Food Syst., 2024, 8, 1500359 CrossRef.
- S. Kendler, A. Sasidharan and T. Rustad, Front. Sustain. Food Syst., 2024, 7, 1331113 Search PubMed.
- J. Coque, C. Jacobsen, B. Forghani, A. Meyer, G. Jakobsen, J. J. Sloth and A.-D. M. Sørensen, Mar. Drugs, 2023, 21, 558 CrossRef CAS PubMed.
- J. Ahlborn, A. Stephan, T. Meckel, G. Maheshwari, M. Rühl and H. Zorn, Int. J. Recycl. Org. Waste Agric., 2019, 8, 447–455 CrossRef.
- V. S. Uma, Z. Usmani, M. Sharma, D. Diwan, M. Sharma, M. Guo, M. G. Tuohy, C. Makatsoris, X. Zhao, V. K. Thakur and V. K. Gupta, Phytochem. Rev., 2023, 22, 1015–1040 CrossRef CAS PubMed.
- H. Onyeaka, T. Miri, K. Obileke, A. Hart, C. Anumudu and Z. T. Al-Sharify, Carbon Capture Sci. Technol., 2021, 1, 100007 Search PubMed.
- L. M. L. Laurens, J. Markham, D. W. Templeton, E. D. Christensen, S. Van Wychen, E. W. Vadelius, M. Chen-Glasser, T. Dong, R. Davis and P. T. Pienkos, Energy Environ. Sci., 2017, 10, 1716–1738 Search PubMed.
- M. D. Torres, N. Flórez-Fernández and H. Domínguez, Mar. Drugs, 2019, 17, 314 Search PubMed.
- F. Li, Y. Li, K. S. Novoselov, F. Liang, J. Meng, S.-H. Ho, T. Zhao, H. Zhou, A. Ahmad, Y. Zhu, L. Hu, D. Ji, L. Jia, R. Liu, S. Ramakrishna and X. Zhang, Nano-Micro Lett., 2023, 15, 35 CrossRef CAS PubMed.
- D. Jiménez, F. Dini-Andreote and J. Van Elsas, Biotechnol. Biofuels, 2014, 7, 92 Search PubMed.
- C. Jo, J. Zhang, J. M. Tam, G. M. Church, A. S. Khalil, D. Segrè and T.-C. Tang, Mater. Today Bio, 2023, 19, 100560 CrossRef CAS PubMed.
- S. A. Siddiqui, Z. Erol, J. Rugji, F. Taşçı, H. A. Kahraman, V. Toppi, L. Musa, G. Di Giacinto, N. A. Bahmid, M. Mehdizadeh and R. Castro-Muñoz, Bioresour. Bioprocess., 2023, 10, 85 Search PubMed.
- A. K. Singh, M. Bilal, H. M. N. Iqbal, A. S. Meyer and A. Raj, Sci. Total Environ., 2021, 777, 145988 CrossRef CAS PubMed.
- L. Bataglia, A. Conversano, D. Di Bona, D. Sogni, D. Voccia, E. Mazzoni and L. Lamastra, Energies, 2025, 18, 2826 CrossRef CAS.
- R. Soloha, L. K. Lukasa and E. Dace, Biomass Conv. Bioref., 2025, 15, 4349–4370 CrossRef.
- L. S. Hocevar, C. D. A. DAquino, C. T. Alves and A. Á. B. Santos, Rev. Gestão Soc. e Ambiental, 2025, 19, e012707 CrossRef.
- S. Alonso-Muñoz, R. González-Sánchez, M.-S. Medina-Salgado and F.-E. García-Muiña, Econ. Res.–Ekon. Istr., 2023, 36, 2142636 Search PubMed.
- C. Talens, R. Llorente, L. Simó-Boyle, I. Odriozola-Serrano, I. Tueros and M. Ibargüen, Foods, 2022, 11, 3396 CrossRef CAS PubMed.
- Y.-S. Wang and M. Shelomi, Foods, 2017, 6, 91 CrossRef PubMed.
- U. Shah and Dr M. Hajoori, Int. J. Res. Appl. Sci. Eng. Technol., 2022, 10, 1311–1317 CrossRef.
- T. G. Dikme and A. A. Hayaloğlu, Eur. Food Res. Technol., 2025, 251, 2291–2305 CrossRef CAS.
- V. Angeli, P. Miguel Silva, D. Crispim Massuela, M. W. Khan, A. Hamar, F. Khajehei, S. Graeff-Hönninger and C. Piatti, Foods, 2020, 9, 216 CrossRef CAS PubMed.
- M. Iriondo-DeHond, E. Miguel and M. D. Del Castillo, Nutrients, 2018, 10, 1358 CrossRef PubMed.
- M. Ummels, L. JanssenDuijghuijsen, J. J. Mes, C. Van Der Aa, R. Wehrens and D. Esser, Nutrients, 2023, 15, 3196 CrossRef CAS PubMed.
- J. Pennells, I. Bless, P. Juliano and D. Ying, in From Biomass to Biobased Products, IntechOpen, 2023 Search PubMed.
- R. N. Raţu, I. D. Veleşcu, F. Stoica, A. Usturoi, V. N. Arsenoaia, I. C. Crivei, A. N. Postolache, F. D. Lipşa, F. Filipov, A. M. Florea, M. A. Chiţea and I. S. Brumă, Agriculture, 2023, 13, 1559 CrossRef.
- C. Starkey, P. Davis and L. Moody, J. Anim. Sci., 2025, 103, 66–67 CrossRef.
- L. Gasco, I. Biasato, S. Dabbou, A. Schiavone and F. Gai, Animals, 2019, 9, 170 CrossRef PubMed.
- J. K. M. Punzalan and K. A. Rosentrater, Animals, 2024, 14, 1689 CrossRef PubMed.
- K. Hua, J. M. Cobcroft, A. Cole, K. Condon, D. R. Jerry, A. Mangott, C. Praeger, M. J. Vucko, C. Zeng, K. Zenger and J. M. Strugnell, One Earth, 2019, 1, 316–329 CrossRef.
- M. R. Chaklader, J. Howieson, M. J. Foysal, M. A. Hanif, H. M. R. Abdel-Latif and R. Fotedar, Front. Nutr., 2023, 10, 1145068 Search PubMed.
- D. Badillo, S. Z. Herzka and M. T. Viana, PLoS One, 2014, 9, e107523 Search PubMed.
- C. Dietz and F. Liebert, Aquac. Rep., 2018, 12, 43–48 CrossRef.
- M. Sońta, A. Rekiel and M. Batorska, Ann. Anim. Sci., 2019, 19, 257–271 Search PubMed.
- M. Li, C. Mao, X. Li, L. Jiang, W. Zhang, M. Li, H. Liu, Y. Fang, S. Liu, G. Yang and X. Hou, Foods, 2023, 12, 4073 CrossRef CAS PubMed.
- L. Pinotti, C. Giromini, M. Ottoboni, M. Tretola and D. Marchis, Animal, 2019, 13, 1365–1375 CrossRef CAS PubMed.
- J. Hadi and G. Brightwell, Foods, 2021, 10, 1226 Search PubMed.
- K. Piwowarek, E. Lipińska, E. Hać-Szymańczuk, M. Kieliszek and I. Ścibisz, Appl. Microbiol. Biotechnol., 2018, 102, 515–538 CrossRef CAS PubMed.
- K. J. Ong, J. Johnston, I. Datar, V. Sewalt, D. Holmes and J. A. Shatkin, Compr. Rev. Food Sci. Food Saf., 2021, 20, 5421–5448 CrossRef PubMed.
- A. H. Msunje, E. Nyakeri and E. Okuto, East Afr. J. Agric. Biotechnol., 2025, 8, 179–200 CrossRef.
- Regulation – 2023/915 – EN – EUR-Lex, https://eur-lex.europa.eu/eli/reg/2023/915/oj/eng, accessed 16 January 2026.
- European Parliament and Council of the European Union, Directive 2002/32/EC of 7 May 2002 on Undesirable Substances in Animal Feed, Official Journal of the European Communities, 2002, vol. L140, pp. 10–22, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32002L0032 Search PubMed.
- Navigating Novel Foods, https://www.efsa.europa.eu/en/news/navigating-novel-foods-what-efsas-updated-guidance-means-safety-assessments, accessed 17 January 2026.
- GRAS – the faster regulatory pathway to food and feed market, https://medfilesgroup.com/generally-recognised-as-safe-gras/, accessed 17 January 2026.
- E. Piercy, W. Verstraete, P. R. Ellis, M. Banks, J. Rockström, P. Smith, O. C. Witard, J. Hallett, C. Hogstrand, G. Knott, A. Karwati, H. F. Rasoarahona, A. Leslie, Y. He and M. Guo, Green Chem., 2023, 25, 808–832 RSC.
- T.-K. Kim, H. I. Yong, Y.-B. Kim, H.-W. Kim and Y.-S. Choi, Food Sci. Anim. Resour., 2019, 39, 521–540 CrossRef PubMed.
- S. B. Meyer, A. M. Wilson, M. Calnan, J. Henderson, J. Coveney, D. McCullum, A. R. Pearce, P. Ward and T. Webb, BMC Public Health, 2017, 17, 189 CrossRef PubMed.
- AAFCO Votes Yes on New Pathway for Animal Food Ingredients, https://www.aafco.org/news/aafco-votes-yes-on-new-regulatory-pathway-for-animal-food-ingredients/, accessed 17 January 2026.
- S. Albrektsen, R. Kortet, P. V. Skov, E. Ytteborg, S. Gitlesen, D. Kleinegris, L. Mydland, J. Ø. Hansen, E. Lock, T. Mørkøre, P. James, X. Wang, R. D. Whitaker, B. Vang, B. Hatlen, E. Daneshvar, A. Bhatnagar, L. B. Jensen and M. Øverland, Rev. Aquacult., 2022, 14, 1790–1812 CrossRef.
- Converting food waste to protein… first-time research reveals the benefits and burdens, https://www.foodnavigator.com/Article/2023/02/23/Converting-food-waste-to-protein-first-time-research-reveals-the-benefits-and-burdens/, accessed 17 January 2026.
- A. Nikhil Swaraj, J. A. Moses and L. Manickam, Sustainable Food Technol., 2025, 3, 648–664 RSC.
- A. Ritala, S. T. Häkkinen, M. Toivari and M. G. Wiebe, Front. Microbiol., 2017, 8, 2009 CrossRef PubMed.
- A. S. Oliveira, J. O. Pereira, C. Ferreira, M. Faustino, J. Durão, M. E. Pintado and A. P. Carvalho, Peptide-rich extracts from spent yeast waste streams as a source of bioactive compounds for the nutraceutical market, Innov. Food Sci. Emerg. Technol., 2022, 81, 103148 Search PubMed.
- P. T. Sekoai, Y. Roets-Dlamini, F. O'Brien, S. Ramchuran and V. Chunilall, Microorganisms, 2024, 12, 166 CrossRef CAS PubMed.
- T. Linder, Front. Sustain. Food Syst., 2019, 3, 32 Search PubMed.
- B. Bajić, D. Vučurović, Đ. Vasić, R. Jevtić-Mučibabić and S. Dodić, Foods, 2022, 12(1), 107 Search PubMed.
- A. Franco, V. Pucciarelli, S. A. Hosseini, E. Schmitt, F. Bovera, C. Scieuzo and P. Falabella, Insects, 2025, 16, 508 Search PubMed.
- X. Lin, F. Wang, Y. Lu, J. Wang, J. Chen, Y. Yu, X. Tao, Y. Xiao and Y. Peng, Curr. Res. Food Sci., 2023, 7, 100596 Search PubMed.
- S. I. A. R. Sharma, Int. J. Behav. Sci., 2024, 39, 37 CrossRef.
- H. Mbareche, M. Veillette, M.-È. Dubuis, B. Bakhiyi, G. Marchand, J. Zayed, J. Lavoie, G. J. Bilodeau and C. Duchaine, J. Air Waste Manage. Assoc., 2018, 68, 1198–1210 Search PubMed.
- N. Praeg and T. Klammsteiner, J. Environ. Manage., 2024, 356, 120622 CrossRef CAS PubMed.
- F. F. B. Hasnan, Y. Feng, T. Sun, K. Parraga, M. Schwarz and M. Zarei, Foods, 2023, 12, 4243 Search PubMed.
- B. Erickson, J. E. Nelson and P. Winters, Biotechnol. J., 2012, 7, 176–185 CrossRef CAS PubMed.
- Z. Wang, L. Zhou, Z. Zhu and L. Du, Renew. Agric. Food Syst., 2026, 41, e2 CrossRef.
- A. Safavi, R. Thrastardottir, R. I. Thorarinsdottir and R. Unnthorsson, Sustainability, 2024, 16, 9063 Search PubMed.
- M. Li, C. Mao, X. Li, L. Jiang, W. Zhang, M. Li, H. Liu, Y. Fang, S. Liu, G. Yang and X. Hou, Foods, 2023, 12, 4073 CrossRef CAS PubMed.
- N. J. Salazar-López, G. A. Barco-Mendoza, B. S. Zuñiga-Martínez, J. A. Domínguez-Avila, R. M. Robles-Sánchez, M. A. V. Ochoa and G. A. González-Aguilar, Bioengineering, 2022, 9, 623 CrossRef PubMed.
- Z. Abro, I. Macharia, K. Mulungu, S. Subramanian, C. M. Tanga and M. Kassie, Front. Insect Sci., 2022, 2, 968042 Search PubMed.
- A. Artola, X. Font, J. Moral-Vico and A. Sánchez, Front. Chem. Eng., 2024, 6, 1463785 Search PubMed.
- E. Molina-Peñate, M. Del Carmen Vargas-García, A. Artola and A. Sánchez, Waste Manage., 2023, 161, 92–103 CrossRef PubMed.
- N. Paris, A. Fortin, N. Hotte, A. Rasooli Zadeh, S. Jain and L. Hénault-Ethier, J. Clean. Prod., 2024, 460, 142450 CrossRef CAS.
- J. Ferreira, P. Lennartsson and M. Taherzadeh, Energies, 2014, 7, 3872–3885 CrossRef.
- D. Díaz-Vázquez, D. A. Orozco-Nunnelly, C. Yebra-Montes, C. Senés-Guerrero and M. S. Gradilla-Hernández, Biomass Bioenergy, 2022, 161, 106471 CrossRef.
- J. S. S. Yadav, J. Bezawada, C. M. Ajila, S. Yan, R. D. Tyagi and R. Y. Surampalli, Bioresour. Technol., 2014, 164, 119–127 Search PubMed.
- T. Veldkamp, N. Meijer, F. Alleweldt, D. Deruytter, L. Van Campenhout, L. Gasco, N. Roos, S. Smetana, A. Fernandes and H. J. Van Der Fels-Klerx, Insects, 2022, 13, 281 CrossRef PubMed.
- Alternative Protein – A Comparison of the Regulation of Insect Protein as Food and Feed, https://www.mofo.com/resources/insights/211028-alternative-protein-industry-series, accessed 18 January 2026.
- H. Su, B. Zhang, J. Shi, S. He, S. Dai, Z. Zhao, D. Wu and J. Li, Insects, 2025, 16, 830 Search PubMed.
- Susinchain – Sustainable Insect Chain, https://susinchain.eu/, accessed 18 January 2026.
- I. B. Magalhães, A. S. A. d. P. Pereira, T. A. Silva, J. Ferreira, M. Q. Braga, E. A. Couto, P. P. Assemany and M. L. Calijuri, J. Water Proc. Eng., 2024, 66, 105929 Search PubMed.
- V.-T. Nguyen, V.-A. Le, Q.-H. Do, T.-N.-C. Le and T.-D.-H. Vo, Sci. Total Environ., 2024, 912, 168911 Search PubMed.
- D. L. Sutherland and P. J. Ralph, N. Z. J. Bot., 2020, 58(4), 334–357 Search PubMed.
- A. López-Sánchez, A. L. Silva-Gálvez, Ó. Aguilar-Juárez, C. Senés-Guerrero, D. A. Orozco-Nunnelly, D. Carrillo-Nieves and M. S. Gradilla-Hernández, J. Environ. Manage., 2022, 308, 114612 CrossRef PubMed.
- Y. Ma, T. Liu, Z. Yuan and J. Guo, Water Res., 2024, 259, 121820 Search PubMed.
- C. Woern and L. Grossmann, Biotechnol. Adv., 2023, 69, 108240 Search PubMed.
- F.-M. Kerckhof, M. Sakarika, M. Van Giel, M. Muys, P. Vermeir, J. De Vrieze, S. E. Vlaeminck, K. Rabaey and N. Boon, Front. Bioeng. Biotechnol., 2021, 9, 733753 Search PubMed.
- J. Coronel-León, D. Maza, I. García-Álvarez De Toledo, A. Jofré, B. Martín, X. Serra and S. Bover-Cid, Foods, 2025, 15, 117 CrossRef PubMed.
- A. A. Conceição, T. D. Mendes, S. Mendonça, B. F. Quirino, E. G. D. Almeida and F. G. D. Siqueira, Fermentation, 2022, 8, 402 CrossRef.
- M. Perwez and S. Al Asheh, Biotechnol. Rep., 2025, 45, e00873 Search PubMed.
- C. Caldeira, A. Vlysidis, G. Fiore, V. De Laurentiis, G. Vignali and S. Sala, Bioresour. Technol., 2020, 312, 123575 CrossRef CAS PubMed.
- D. Pérez-Almada, Á. Galán-Martín, M. D. M. Contreras and E. Castro, Sustain. Energy Fuels, 2023, 7, 4031–4050 RSC.
- R. Kumar, A. Guleria, Y. S. Padwad, V. Srivatsan and S. K. Yadav, Crit. Rev. Food Sci. Nutr., 2025, 65, 3496–3545 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |