Comparative assessment of albumins from ten pulses: techno-functional properties and amino acid profiles for sustainable food applications
Received
23rd October 2025
, Accepted 20th December 2025
First published on 22nd December 2025
Abstract
Pulse albumins, often recovered as by-products during pulse processing, have attracted increasing interest due to their favorable solubility and nutritional properties. However, compared with extensively studied animal proteins such as whey protein, systematic information on the techno-functional properties and amino acid profiles of pulse albumins across different species remains limited, restricting their targeted application in food systems. This study addresses the gap by systematically investigating the techno-functional properties and amino acid profiles of albumin fractions extracted from ten major pulses: Vigna radiata (mung bean, MB), Vigna angularis (adzuki bean, RB), Phaseolus vulgaris (red kidney bean, RK; white kidney bean, WK), Cicer arietinum (kabuli chickpea, KCP; desi chickpea, DCP), Pisum sativum (green pea, GP; yellow pea, YP), Vicia faba (broad bean, BB), and Lens culinaris (lentil, LP). All albumins exhibited excellent pH-independent solubility (>70%). Notable variations were observed: P. vulgaris albumins showed superior solubility at high concentrations and water-holding capacity, while P. sativum albumins had outstanding foaming properties. Most pulse albumins surpassed whey protein in foaming but were inferior in emulsification. Amino acid scores met FAO/WHO standards, with P. vulgaris albumins displaying the highest nutritional indices. These findings demonstrate pronounced interspecific differences in the techno-functional and nutritional characteristics of pulse albumins, providing evidence that species selection is critical for targeted food applications.
Sustainability spotlight
This work contributes to sustainable food development by assessment of pulse albumins—protein fractions typically recovered as by-products from pulse processing. Compared with conventional plant-derived protein isolates, pulse albumins offer a renewable and low-carbon alternative with excellent solubility and functionality and balanced amino acid composition. Utilizing these underexploited proteins supports resource efficiency and waste minimization. By systematically comparing albumins from ten pulse species, this study provides a scientific foundation for their targeted use in plant-based formulations, thereby promoting the circular utilization of pulse resources and contributing to the achievement of United Nations Sustainable Development Goals (SDGs).
|
1. Introduction
As the global population continues to increase and the need for sustainable food sources intensifies, plant proteins have garnered significant interest for their environmental benefits. In addition to being nutritionally rich, plant proteins significantly reduce greenhouse gas emissions and represent a promising alternative to animal-based proteins.1 According to the Food and Agriculture Organization (FAO) of the United Nations, pulses are defined as dry-harvested leguminous crops except high oil content seeds, such as soybeans and peanuts.2 The global production of pulses in 2023 reached approximately 103 million tons, with kidney bean (Phaseolus vulgaris) as the dominant species.3 This species is widely cultivated in countries such as India, Myanmar, Brazil, and China. Other major pulse crops included chickpeas (Cicer arietinum), dry peas (Pisum sativum), fava beans (Vicia faba), and lentils (Lens culinaris). China is one of the world's largest producers and consumers of pulses, with mung beans (Vigna radiata), kidney beans (Phaseolus vulgaris), chickpeas (Cicer arietinum), peas (Pisum sativum), broad beans (Vicia faba), and lentils (Lens culinaris) widely cultivated.
Pulses are high-yield crops with a substantial protein content of 20–30%, which is two to three times greater than that of most cereals (8–15%),4 making them an important dietary source of plant-based protein. According to the Osborne classification, pulse proteins are categorized into albumins, globulins, prolamins, and glutelins. Among these, globulins are the main storage proteins, typically accounting for 50–70% of the total protein content.5 However, their poor water solubility and tendency to aggregate at pH 4–5 limit their application in food systems that require high solubility. In contrast, although albumins are present in smaller amounts (10–25%),5 they demonstrate distinct advantages in solubility across a wider pH range compared to globulins.6 Solubility is a key property of pulse albumins, as it underpins their applicability in food systems and also a pre-requisite for other related functional properties such as foaming and emulsification.7
Existing research indicates that albumin derived from certain pulses exhibits significant functional properties. For instance, Djoullah et al.8 demonstrated that pea albumin retains high solubility (around 85%) even at pH 5, whereas pea globulin solubility drops sharply to ∼20% under the same conditions. Yang et al.9 reported that albumin fractions from mung beans and yellow peas exhibit foaming capacities comparable to or superior to those of whey and egg white proteins. Beyond their favourable processing properties, some varieties of pulse albumins were investigated for their amino acid profiles. Chagas et al.10 found that albumins in Phaseolus vulgaris varieties demonstrated a higher cysteine content than globulins, making them valuable for compensating for the general deficiency of sulfur-containing amino acids in pulse proteins. Moreover, pulse albumins are rich in various bioactive components. Previous studies have demonstrated that white kidney bean extract rich in alpha-amylase inhibitors exhibits beneficial health effects in preventing hyperglycemic episodes and fat accumulation.11 Pulse albumins contain trypsin inhibitors which can also inhibit proteases produced by pathogens; therefore, they hold potential for use as antibacterial agents.12
The current research on pulse albumins remains significantly underexplored compared to studies on water-soluble proteins from animal sources (e.g., cow's milk whey protein). While the techno-functional properties—such as solubility, emulsifying, and foaming capacities—of albumins from specific individual pulses have been studied to some extent,6,13 systematic comparative analyses across different pulse species are strikingly scarce. Concurrently, on the nutritional front, a clear contrast exists on the pulse globulins which are known to be deficient in essential sulfur-containing amino acids,14 whereas comprehensive comparative studies assessing and contrasting the amino acid profiles of albumins from diverse pulses are notably absent, leaving their nutritional potential inadequately understood. Therefore, a systematic comparative study of the techno-functional properties and amino acid profiles of different pulse albumins is of considerable scientific and practical significance.
In this study, we conducted a comprehensive investigation of the techno-functional properties and amino acid profiles of albumins isolated from ten major pulse species: Vigna radiata (mung bean, MB), Vigna angularis (adzuki bean, RB), Phaseolus vulgaris (red kidney bean, RK; white kidney bean, WK), Cicer arietinum (kabuli chickpea, KCP; desi chickpea, DCP), Pisum sativum (green pea, GP; yellow pea, YP), Vicia faba (broad bean, BB), and Lens culinaris (lentil, LP). To ensure the representativeness of the samples used in this study, the ten selected pulse varieties cover the dominant species commonly grown and consumed both in China and globally, and all were sourced from their typical major producing regions in China. Using a uniform extraction and analytical protocol, we evaluated the techno-functional properties against whey protein as a benchmark and analysed amino acid composition. This approach provides a more comprehensive understanding of pulsed albumin and offers a broader perspective on the potential of pulse albumins in food applications.
2. Materials and methods
2.1 Materials
Ten varieties of pulses were used in this study, including Vigna radiata (mung bean, MB), Vigna angularis (adzuki bean, RB), Phaseolus vulgaris (red kidney bean, RK; white kidney bean, WK), Cicer arietinum (kabuli chickpea, KCP; desi chickpea, DCP), Pisum sativum (green pea, GP; yellow pea, YP), Vicia faba (broad bean, BB), and Lens culinaris (lentil, LP). To ensure representativeness, all pulse seeds were purchased from major producing areas in China during the year 2024, and each batch was confirmed to be uniform in variety. MB and RB were obtained from Heilongjiang Province, China; RK, WK, and BB were obtained from Yunnan Province, China; KCP and DCP were obtained from Xinjiang, China; GP and YP were obtained from Qinghai Province, China; and LP was sourced from Shanxi Province, China. All seeds were packed in airtight containers and stored in a dry environment at 4 °C until use. All the reagents and chemicals utilized for the research were of analytical reagent grade.
2.2 Methods
2.2.1 Extraction of pulse albumins.
The protein fractions were extracted using the procedure by Yang et al.9 with minor adjustments. Pulses were milled using a high-speed blender (model MX-H2201, Panasonic Appliances (China) Co., Ltd). Then, the flour was sieved through a standard 60-mesh sieve to obtain uniform flour (Fig. 1). The flour was defatted by mixing with n-hexane at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (w/v) ratio with continuous stirring for 60 min. The mixture was then centrifuged, and the upper organic solvent layer was discarded. This defatting procedure was repeated three times. The defatted flour was subsequently collected by filtration and air-dried at room temperature in a fume hood overnight to ensure complete evaporation of residual n-hexane.
3 (w/v) ratio with continuous stirring for 60 min. The mixture was then centrifuged, and the upper organic solvent layer was discarded. This defatting procedure was repeated three times. The defatted flour was subsequently collected by filtration and air-dried at room temperature in a fume hood overnight to ensure complete evaporation of residual n-hexane.
 |
| | Fig. 1 A schematic overview of the pulse protein extraction process. | |
After defatting, the flour was dispersed with deionized water to prepare a 10% (w/v) suspension; the initial pH of the suspension was approximately 6.52–6.67. The pH of the suspension was adjusted to 8.0 using 1 M NaOH, and the mixture was stirred for 2 h and then centrifuged at 8000g for 30 min at 4 °C using a high-speed refrigerated centrifuge (64RL, Beckman Coulter, USA), after which the supernatant was collected and filtered. To isolate the globulin fraction, the pH of the supernatant was adjusted to 4.5 with 1 M HCl and stirred continuously for 1 h, followed by another centrifugation step under the same conditions. The resulting supernatant and precipitate were collected separately. The supernatant was dialyzed using a 5 kDa membrane for 48 hours to obtain albumin. The sediment was redispersed in ultrapure water and stirred for 2 h while gradually adjusting the pH to 7.0. Finally, both albumin and globulin fractions were freeze-dried using a freeze dryer (Christ, Osterode am Harz, Germany). The extraction yields, mass yield and purities of the albumin and globulin fractions were calculated as described in eqn (1), (2) and (3), respectively. Each extraction process was performed in triplicate.
| |  | (1) |
| | 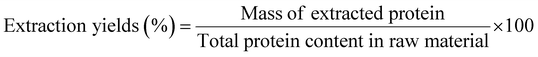 | (2) |
| | 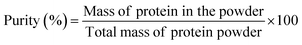 | (3) |
2.2.2 Sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE).
The protein composition of albumin and globulin fractions extracted from the ten pulses was analyzed using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), following the protein quantification method described by Xing et al.15 with slight modifications. Samples were homogenized with loading buffer (Solarbio, P1015) at a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ratio, heated in a boiling water bath for 5 min, and centrifuged (5000g, 4 °C, 10 min) to obtain electrophoretic preparations. Each sample mixture (10 µL) was then applied to precast gels. A 10–250 kDa molecular weight pre-stained protein marker (Sangon Biotech, C520010-0001) was used as the standard. Electrophoresis was performed at a constant voltage (80 V) until the dye front reached the bottom of the gel. After electrophoresis, gels were stained with Coomassie brilliant blue R-250 for 1 h and destained using a 20% methanol and 10% acetic acid solution until a clear background was obtained. The stained gels were scanned, and protein molecular weights were estimated using a ChemiDoc™ MP Imaging System (Bio-Rad Laboratories Inc., CA, USA). Each SDS-PAGE analysis was performed in triplicate.
1 (v/v) ratio, heated in a boiling water bath for 5 min, and centrifuged (5000g, 4 °C, 10 min) to obtain electrophoretic preparations. Each sample mixture (10 µL) was then applied to precast gels. A 10–250 kDa molecular weight pre-stained protein marker (Sangon Biotech, C520010-0001) was used as the standard. Electrophoresis was performed at a constant voltage (80 V) until the dye front reached the bottom of the gel. After electrophoresis, gels were stained with Coomassie brilliant blue R-250 for 1 h and destained using a 20% methanol and 10% acetic acid solution until a clear background was obtained. The stained gels were scanned, and protein molecular weights were estimated using a ChemiDoc™ MP Imaging System (Bio-Rad Laboratories Inc., CA, USA). Each SDS-PAGE analysis was performed in triplicate.
2.3 Determination of the techno-functional properties of pulse albumins
2.3.1 Protein solubility.
Protein solubility of albumin was determined according to the method of Yang et al.16 with slight modifications. Briefly, a protein solution of 3% (w/v) concentration was prepared and stirred for 30 min under different pH conditions (3.0, 4.0, 5.0, 6.0, 7.0, 8.0 and 9.0) and concentration gradients (1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9% and 10%, w/v). Subsequently, the solution was centrifuged at 8000g for 20 min. The protein concentration in the supernatant was measured using the Coomassie brilliant blue method, with three replicates per experimental group. Solubility was defined as the ratio of protein content in the supernatant to the total protein content. Each experiment was performed in triplicate.
2.3.2 Water-holding capacity (WHC) and oil-holding capacity (OHC).
Water-holding capacity (WHC) and oil-holding capacity (OHC) were investigated according to the method described by Habib et al.17 with slight modifications. In brief, 1.0 g of the sample was weighed into a centrifuge tube, followed by the addition of 5 mL distilled water (or rapeseed oil for OHC analysis). The mixture was vortexed and allowed to stand for 30 min. Subsequently, centrifugation was conducted at 6000g for 20 min at room temperature. The centrifuge tube with the protein residue was subsequently weighed, and the WHC/OHC value was calculated using eqn (4). Each experiment was performed in triplicate.| |  | (4) |
where W0: initial sample weight, W1: tube weight, and W2: tube weight with the residue after centrifugation.
2.3.3 Foam formation and stability.
The foaming capacity was determined following the method of Narale et al.18 with minor modifications. Briefly, a 3% (w/v) protein solution was prepared, and its pH was adjusted to various values (3.0, 4.0, 5.0, 6.0, 7.0, 8.0 and 9.0). After that, the solution was homogenized for 1 min at 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 rpm using a Polytron PT 3100 homogenizer. The total volume of foam and the liquid phase was recorded immediately after homogenization. After 30 min of quiescent standing, the remaining foam volume was measured again. The foaming capacity (FC) was calculated using eqn (5) and foam stability (FS) was determined using eqn (6). Each experiment was performed in triplicate.
500 rpm using a Polytron PT 3100 homogenizer. The total volume of foam and the liquid phase was recorded immediately after homogenization. After 30 min of quiescent standing, the remaining foam volume was measured again. The foaming capacity (FC) was calculated using eqn (5) and foam stability (FS) was determined using eqn (6). Each experiment was performed in triplicate.| | 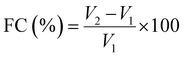 | (5) |
| |  | (6) |
where V: foam volume after 30 min of standing (mL), V1: volume of the protein solution before homogenization (mL), and V2: total volume of foam and the liquid phase after homogenization (mL).
2.3.4 Emulsion formation and stability.
The emulsifying properties were determined according to the method of Zheng et al.19 with slight modifications. Briefly, a 3% (w/v) protein solution was prepared and adjusted to different pH values (3.0, 4.0, 5.0, 6.0, 7.0, 8.0, and 9.0), followed by the addition of 1 mL of soybean oil. The mixture was homogenized at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 2 min, using a Polytron PT 3100 homogenizer. Subsequently, 50 µL aliquots were withdrawn from the bottom of the emulsion and mixed with 5 mL of 0.1% (w/v) SDS solution. The absorbance (A0) was immediately measured at 500 nm using a UV spectrophotometer, followed by a second measurement (A30) after 30 min. All experiments were performed in triplicate. The emulsifying capacity (EC) was calculated using eqn (7), and emulsion stability (ES) was calculated using eqn (8). Each experiment was performed in triplicate.
000 rpm for 2 min, using a Polytron PT 3100 homogenizer. Subsequently, 50 µL aliquots were withdrawn from the bottom of the emulsion and mixed with 5 mL of 0.1% (w/v) SDS solution. The absorbance (A0) was immediately measured at 500 nm using a UV spectrophotometer, followed by a second measurement (A30) after 30 min. All experiments were performed in triplicate. The emulsifying capacity (EC) was calculated using eqn (7), and emulsion stability (ES) was calculated using eqn (8). Each experiment was performed in triplicate.| | 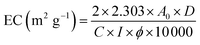 | (7) |
| |  | (8) |
where A0: absorbance at 500 nm measured immediately, A30: absorbance at 500 nm after 30 min, D: dilution factor, C: sample concentration (g mL−1), ϕ: volume fraction of soybean oil in the emulsion (%), and I: optical path length (cm).
2.3.5 Amino acid analysis.
The amino acid composition of pulse albumins was determined using an L-8900 Amino Acid Analyzer (Hitachi High-Tech Science Co., Japan), following the method described by Bidlingmeyer et al.20 with slight modifications. Briefly, approximately 30 mg of each freeze-dried albumin sample was hydrolyzed in 6 M hydrochloric acid (HCl) under vacuum at 110 °C for 24 hours in sealed glass tubes to release protein-bound amino acids. After hydrolysis, the hydrolysates were evaporated to dryness under reduced pressure at 40 °C, reconstituted in citrate buffer (pH 2.2), and filtered through a 0.22 µm membrane prior to injection. The measurement was performed in triplicate.
2.3.6 Statistical analysis.
All experiments were performed in triplicate. The results are presented as mean ± standard deviation, with error bars indicating the standard deviation. Statistical differences were considered significant when p < 0.05. Statistical analyses were conducted using SPSS Statistics 25 followed by one-way ANOVA with the Duncan post hoc test.
3. Results and discussion
3.1 Fractionation of albumin and globulin
Fig. 2A shows the mass yield of albumin and globulin fractions from these pulses. The albumin mass yield ranged from 5.27% (LP) to 7.55% (WK), while the globulin mass yield ranged from 13.52% (LP) to 17.88% (WK). Fig. 2B shows the extraction yields of albumin and globulin fractions from these pulses. Overall, albumin extraction yields ranged from 18.62% (YP) to 25.72% (WK), while globulin extraction yields showed higher values ranging from 55.40% (LP) to 61.15% (WK). The extraction ratio of albumin to globulin was approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, consistent with the previously reported ratio range of these proteins in pulses (1
3, consistent with the previously reported ratio range of these proteins in pulses (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 1
3 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.3).21 Ye et al.22 reported albumin and globulin extraction yields of 36.30% and 52.51%, respectively, in chickpeas through fractional extraction, aligning with the trends observed in this study. However, numerical differences may arise from species-specific characteristics. Fig. 2C further analyses the purity of albumin and globulin from the ten pulses. The results show that the purity of albumin ranged from 73.88% to 77.19% on a wet basis [w.b.], while that of globulin ranged from 77.87% to 81.47% [w.b.], indicating that the purity of the two types of proteins varied less among different bean species and was above 70%. The lower purity of albumin is due to its extraction from the water-soluble fraction, which may retain traces of non-protein components like carbohydrates and phenolics.
6.3).21 Ye et al.22 reported albumin and globulin extraction yields of 36.30% and 52.51%, respectively, in chickpeas through fractional extraction, aligning with the trends observed in this study. However, numerical differences may arise from species-specific characteristics. Fig. 2C further analyses the purity of albumin and globulin from the ten pulses. The results show that the purity of albumin ranged from 73.88% to 77.19% on a wet basis [w.b.], while that of globulin ranged from 77.87% to 81.47% [w.b.], indicating that the purity of the two types of proteins varied less among different bean species and was above 70%. The lower purity of albumin is due to its extraction from the water-soluble fraction, which may retain traces of non-protein components like carbohydrates and phenolics.
 |
| | Fig. 2 The extraction yield (A), mass yield (B) and purity (C) of ten types of pulse albumin and globulin. Data are presented as mean ± SD (n = 3). Different lowercase letters (a–e) indicate statistically significant differences among the albumin samples, while different uppercase letters (A–F) indicate significant differences among the globulin samples (p < 0.05). | |
3.2 SDS-PAGE
Fig. 3 presents the SDS-PAGE profiles of albumin (A) and globulin (G) fractions from the ten pulse varieties. The results indicated that the albumin fractions exhibited fewer electrophoretic bands and predominantly lower molecular weights. Most albumin fractions exhibited electrophoretic bands corresponding to molecular weights between 15 and 98 kDa, primarily consisting of low-molecular-weight components. These proteins mainly corresponded to PA1 albumin (4–18 kDa) and PA2 albumin (23–26 kDa).9 Albumin fractions also contained 30–35 kDa bands, which were considered to belong to phytohemagglutinin.23 A high molecular weight band at 98 kDa was observed, which might correspond to the lipoxygenase based on previous studies.24 Distinct variations were observed in the electrophoresis patterns of the albumin samples. Lanes RK-A and WK-A showed bands at around 40 kDa, which might be associated with α-amylase inhibitors with subunit molecular weights of approximately 40–45 kDa.25 The band observed at 20–25 kDa is likely attributed to low-molecular-weight subunits or partial degradation products. A 60 kDa band observed in lane KCP-A and lane DCP-A was probably from the water-soluble subunit of vicilins.26 Lane LP-A exhibited faint electrophoretic bands, possibly reflecting its inherently low albumin content.
 |
| | Fig. 3 (A–C) SDS-PAGE of the ten types of pulse albumin and globulin. A – albumin; G – globulin. A representative gel image from three independent experiments is shown. | |
Globulins exhibited more electrophoretic bands and higher molecular weights and included a variety of protein subunits, primarily 7S and 11S subunits, with bands distributed across a broad molecular weight range (20–70 kDa).27 Specifically, lanes MB-G, RB-G, RK-G and WK-G displayed concentrated bands at 40–50 kDa, confirming 7S globulins as their primary subunits.9 Lane KCP-G and lane DCP-G showed prominent bands near 35 kDa (lanes KCP-G and DCP-G), corresponding to 11S legumin subunits.28 Lanes GP-G, YP-G and BB-G shared similar banding patterns, with subunits clustered at 20, 35, 40, 50, and 70 kDa. Notably, the 70 kDa subunit was absent in lane RB-G and lane LP-G.
3.3 Protein solubility
3.3.1 Pulse albumin solubility at different pH.
The solubility of albumin fractions from the ten pulse varieties across varying pH levels is illustrated in Fig. 4A. All pulse albumins (3%, w/v) exhibited high solubility (>76%) throughout this pH range. In contrast, the solubility of whey protein (WP) ranged from 59.18% to 82.27%, with a notable decline at pH 5 (59.18 ± 1.79%). At pH 5, the solubility of assayed albumin fractions remained higher than that of WP, with WK showing the highest value (85.15 ± 2.06%) and RB the lowest (76.73 ± 2.51%). Although the isoelectric points of most pulse proteins typically fall between pH 4 and 5,29 the solubility of pulse albumin remained relatively stable across pH 3–9, demonstrating pH-independent behaviour. These differences may be linked to variations in protein subunit composition. Albumin enriched with hydrophilic amino acids (e.g., glutamic acid and aspartic acid) may possess enhanced solubility.30 The findings align with established trends in protein solubility reported in the literature. Mundi et al.13 observed minimal solubility of kidney bean globulin within pH 4–6, whereas albumin maintained high solubility across a broad pH range. Similarly, Nwachukwu et al.31 reported that flaxseed albumin exhibited significantly higher solubility than globulin within pH 3–9. This study further confirms that pulse albumin, with its good solubility across a wide pH spectrum, holds potential for expanding its applications in food and beverage formulations.
 |
| | Fig. 4 Effect of pH (A) and concentration (B) on the solubility of the ten types of pulse albumin. Values shown in the heatmap represent mean values of three replicates (n = 3). Statistical analysis was performed using mean ± SD, as described in the Methods section. | |
3.3.2 Pulse albumin solubility at different concentrations.
The solubility profiles of albumin fractions from the ten pulses under varying concentration conditions are presented in Fig. 4B. As shown, all pulse albumins, along with WP, exhibited a decreasing trend in solubility with increasing protein concentrations. At low concentrations (1–3%, w/v), solubility remained relatively high across all samples, exceeding 80%, with RK (89.21 ± 2.23%) showing the highest solubility at a 3% (w/v) concentration. However, with an increasing concentration to moderate and high levels (5–10%, w/v), solubility progressively declined to a range of 67.81–76.40% at 10% (w/v). This trend aligns with the phenomenon of enhanced intermolecular interactions and aggregate formation in proteins at higher concentrations.32 Notably, pulse albumins exhibited higher solubility than WP at all concentrations, with more pronounced differences observed above 5% (w/v). For example, at a concentration of 10% (w/v), the solubility of WK remained high at 76.40 ± 2.60%, compared with that of WP, which decreased to 65.61 ± 1.27%. Pulse albumins demonstrated a smaller reduction in solubility with increasing concentrations indicating their superior ability to remain soluble at higher protein levels. Among the ten pulses, RK and WK showed the highest solubility performance, maintaining values above 76% even at a 10% (w/v) concentration. In contrast, RB and BB displayed the lowest solubility at this concentration, decreasing to 67.81 ± 2.03% and 68.86 ± 1.99%, respectively.
3.4 Water-holding capacity (WHC) and oil-holding capacity (OHC)
Fig. 5 shows the water-holding capacity (WHC) and oil-holding capacity (OHC) of albumins from the ten pulse sources. The results revealed significant differences in both WHC and OHC among the different pulse albumins. In terms of WHC (Fig. 5A), WK (3.99 ± 0.13 mL g−1), RK (3.67 ± 0.10 mL g−1), and YP (3.51 ± 0.06 mL g−1) exhibited higher values than WP (3.38 ± 0.15 mL g−1), while the other pulse albumins showed lower WHC with MB recording the lowest value (2.04 ± 0.02 mL g−1). Nevertheless, all pulse albumins displayed significantly higher WHC than the commercial soy protein isolate (1.26 mL g−1).33
 |
| | Fig. 5 WHC (A) and OHC (B) of the 10 types of pulse albumin. Data are presented as mean ± SD (n = 3). Different lowercase letters indicate statistically significant differences (P < 0.05). | |
However, pulse albumins demonstrated lower OHC than WP (2.44 ± 0.11 mL g−1) (Fig. 5B). Among them, KCP (2.22 ± 0.12 mL g−1), RB (2.01 ± 0.14 mL g−1), and YP (1.95 ± 0.08 mL g−1) had relatively higher OHC, whereas LP had the lowest value (1.11 ± 0.09 mL g−1). The high WHC of pulse albumins may result from their high levels of polar or charged amino acids, which can form hydrogen bonds with water molecules, enhancing the protein's water-binding ability.34 Conversely, the lower proportion of hydrophobic amino acids may reduce their interaction with lipids, resulting in relatively lower OHC.35 In addition, a comparison with the solubility data (Fig. 4) suggests that pulse albumins with higher solubility generally exhibited higher WHC values. This finding aligns with the underlying mechanism that albumin fractions, rich in polar and charged amino acids, readily interact with water.34 Proteins with high solubility possess a greater number of hydrated molecules and an expanded structure in solution, which subsequently facilitates increased water entrapment and retention within the protein matrix, thereby leading to a higher WHC. Therefore, the superior WHC of certain pulses like WK and RK can be partially attributed to their excellent solubility, which synergistically enhances their potential application in aqueous food systems.
3.5 Foaming capacity (FC) and foam stability (FS)
Foaming capacity (FC) refers to the ability of proteins to incorporate air and generate foam during whipping, while foam stability (FS) reflects the ability of the formed foam to maintain its structure over time. These two techno-functional properties are crucial in the formulation of aerated beverages, protein drinks, and bakery products. FC and FS are primarily influenced by the molecular structure of proteins, their interfacial behaviour, and environmental factors such as pH.36 As shown in Fig. 6, both the FC and FS of the ten pulse albumins exhibited significant pH dependence within the pH range of 3–9. In general, the lowest FC and FS values were observed near pH 5, and both increased markedly as the pH increased. This trend is consistent with findings by Lawal et al.37 who reported enhanced foaming properties of African locust bean albumin under alkaline conditions. In alkaline environments, increased net surface charge and reduced hydrophobic interactions among protein molecules enhance protein flexibility. This facilitates their migration and unfolding at the air–water interface, promoting foam formation and stabilization.38 Our previous experiments also confirmed that the solubility of pulse albumins increases under alkaline conditions, which is a prerequisite for achieving good foaming properties. Compared with WP, pulse albumins exhibited superior FC and FS across the entire pH range. WP showed the highest FC (34.57 ± 1.19%) at pH 9, while all tested pulse albumins exhibited higher FC values at the same pH, ranging from 57.07% (DCP) to 75.67% (GP), which were approximately 1.6 to 2.2 times greater than that of WP. Similarly, the FS of WP peaked at 26.72% at pH 9, whereas the FS of pulse albumins ranged from 57.13% (LP) to 76.22% (GP), which is about 2.14 to 2.85 times higher than that of WP. These results indicate that pulse albumins outperform WP in both foaming capacity and foam stability, demonstrating strong potential as techno-functional property protein alternatives. Foaming performance also varied among the different pulse albumins. Pea albumins, particularly GP, consistently demonstrated superior and stable foaming capacity (FC) across all pH levels, with values ranging from 62.76% to 75.67%. These were followed by YP, which also exhibited excellent FC, ranging from 53.81% to 71.84%. In contrast, RB (48.31–61.89%) and DCP (47.99–57.04%) showed comparatively lower values. In terms of FS, GP achieved the highest stability at pH 9 (76.24 ± 1.17%) but dropped significantly to 49.72 ± 1.22% at pH 5. In contrast, KCP demonstrated more stable FS across pH levels, with values of 73.92 ± 2.23% at pH 9 and 62.24 ± 0.25% at pH 5. On the other hand, DCP (48.89–57.78%) and LP (43.49–57.75%) exhibited relatively weak foam stability across the tested pH range.
 |
| | Fig. 6 Effect of pH on the foaming properties (A) and foaming stability (B) of the ten types of pulse albumin. Values shown in the heatmap represent mean values of three replicates (n = 3). Statistical analysis was performed using mean ± SD, as described in the Methods section. | |
3.6 Emulsifying properties
Emulsifying activity (EAI) indicates a protein's capacity to adsorb at the oil–water interface, while emulsifying stability (ESI) measures its effectiveness in forming and maintaining stable emulsions. As shown in Fig. 7, the EAI and ESI values of all ten pulse albumins across the pH range of 3–9 were generally lower than those of whey protein (WP). This may be attributed to the highly hydrophilic nature and relatively low content of hydrophobic groups in pulse albumins. Such strong hydrophilicity may limit their interaction with the oil phase, thereby weakening their emulsifying capacity. The EAI of pulse albumins exhibited clear pH dependence. The lowest EAI values were observed around pH 5–6, near their isoelectric point, whereas significantly higher values were recorded under alkaline conditions (pH 8–9).
 |
| | Fig. 7 Effect of pH on the emulsifying activity (A) and emulsion stability (B) of the ten types of pulse albumin. Values shown in the heatmap represent mean values of three replicates (n = 3). Statistical analysis was performed using mean ± SD, as described in the Methods section. | |
This trend is likely due to increased negative surface charges and partial unfolding of protein molecules under alkaline pH, which expose more hydrophobic regions, promoting faster migration and adsorption at the oil–water interface.23 Among the tested samples, YP and KCP exhibited superior emulsifying activity, with YP showing the highest EAI of 6.25 ± 0.02 m2 g−1 at pH 9, significantly outperforming the other pulse albumins. In contrast, MB showed lower emulsifying ability, with an EAI of 3.94 ± 0.03 m2 g−1 at pH 9, decreasing to 1.30 ± 0.02 m2 g−1 at pH 5. The trend observed for ESI was similar to that for EAI, with the lowest stability observed near pH 5 and significant improvements under acidic or alkaline conditions. GP and YP exhibited the highest ESI values. These observations align with previous reports. For instance, Shevkani et al.39 found that both the EAI and ESI of kidney bean proteins dropped significantly at pH 4.5, close to their isoelectric point. Similarly, Oluwajuyitan et al.40 reported that pea albumin formed smaller emulsion droplet sizes at pH 9 compared to pH 5, further supporting the enhanced emulsification performance under alkaline conditions.
3.7 Amino acid composition profile
3.7.1 Amino acid composition and content.
The amino acid composition and content of albumins from the ten pulse types are summarized in Table 1. Pulse albumins were found to be rich in both variety and total content of amino acids. Among them, glutamic acid + glutamine was the most abundant (12.69–18.84%), followed by aspartic acid + asparagine (8.26–12.71%), indicating that acidic amino acids are the primary components of pulse albumins. This finding is consistent with that of Stałanowska et al.,41 who also identified acidic amino acids as predominant constituents in plant proteins. Pulse albumins contained a substantially higher proportion of negatively charged amino acids (NCAA: 23.79–30.98%) compared to positively charged amino acids (PCAA: 11.08–16.74%). The abundance of negatively charged residues, particularly carboxyl groups, enhances hydrophilicity and solubility by strengthening protein–water interactions.30 In addition, lysine content was relatively high, ranging from 5.51% (RB) to 9.04% (WK); this aligns with the results of Mundi et al.13 who observed a lysine content of 7.50% in kidney bean albumin. The sulfur-containing amino acids ranged from 0.65% (BB) to 1.59% (MB) for methionine and from 1.16% (MB) to 2.76% (GP) for cysteine. These values are consistent with those reported by Ye et al.22 for chickpea albumin, which contained 0.72% methionine and 2.53% cysteine. The ratio of essential amino acids to total amino acids (E/T) ranged from 36.26% (RB) to 45.06% (WK), all exceeding the FAO/WHO recommended ideal value of 36%. Notably, WK (43.50 ± 0.07%), RK (43.50 ± 0.07%), GP (41.66 ± 0.21%), and YP (41.46 ± 0.12%) exhibited higher essential amino acid contents. Branched-chain amino acid (BCAA) levels varied from 11.02% (LP) to 17.78% (RK). RK (17.78 ± 0.20%) and WK (17.67 ± 0.10%) showed significantly higher BCAA contents compared to the other pulses, such as lentils (11.02 ± 0.12%). The BCAA levels in RK, WK, GP, and YP exceeded the 15.44% reported for flaxseed globulin,31 suggesting superior potential for promoting muscle protein metabolism.
Table 1 Amino acid composition of 10 types of pulse albumin. Values are expressed as mean ± standard deviation (n = 3). Different lowercase letters indicate a statistically significant difference within each row (P < 0.05)a
| Amino acid (%) |
Vigna
|
Phaseolus
|
Cicer
|
Pisum
|
Vicia
|
Lens
|
| MB |
RB |
RK |
WK |
KCP |
DCP |
GP |
YP |
BB |
LP |
|
Asx: aspartic acid + asparagine. Glx: glutamic acid + glutamine. E/T: ratio of essential to total amino acids. BCAA: branched chain amino acids (valine, isoleucine, and leucine). PCAA: positively charged amino acids (histidine and lysine). NCAA: negatively charged amino acids (Asx, asparagine + aspartic acid; Glx, glutamine + glutamic acid).
|
| Asx |
8.26 ± 0.17h |
10.08 ± 0.05d |
12.14 ± 0.19b |
12.71 ± 0.04a |
9.67 ± 0.04f |
9.31 ± 0.02g |
11.69 ± 0.08c |
12.00 ± 0.07b |
9.85 ± 0.06e |
9.89 ± 0.02e |
| Thr |
4.03 ± 0.06ef |
3.95 ± 0.03f |
5.95 ± 0.02b |
6.58 ± 0.11a |
4.44 ± 0.26d |
4.19 ± 0.07e |
5.49 ± 0.08c |
5.80 ± 0.08b |
3.98 ± 0.05f |
4.03 ± 0.11ef |
| Ser |
5.14 ± 0.09a |
5.13 ± 0.04a |
4.74 ± 0.03b |
4.11 ± 0.08e |
3.65 ± 0.02g |
4.66 ± 0.02b |
4.39 ± 0.06d |
4.01 ± 0.02f |
4.48 ± 0.05c |
3.73 ± 0.06g |
| Glx |
17.29 ± 0.05b |
15.30 ± 0.02e |
18.84 ± 0.03a |
17.32 ± 0.04b |
15.96 ± 0.05d |
12.69 ± 0.05h |
16.76 ± 0.03c |
16.74 ± 0.02c |
15.04 ± 0.09f |
13.90 ± 0.11g |
| Gly |
5.78 ± 0.03a |
5.26 ± 0.05b |
4.07 ± 0.04f |
4.04 ± 0.02g |
4.44 ± 0.02c |
3.54 ± 0.03g |
4.31 ± 0.04d |
4.25 ± 0.05e |
5.29 ± 0.06b |
4.44 ± 0.02c |
| Ala |
4.49 ± 0.03bc |
4.00 ± 0.07c |
4.02 ± 0.01c |
3.84 ± 0.06c |
4.34 ± 0.06bc |
4.51 ± 0.03bc |
4.08 ± 0.05b |
5.42 ± 0.05a |
5.58 ± 0.07a |
4.95 ± 0.03b |
| Cys |
1.16 ± 0.07g |
1.42 ± 0.09f |
2.29 ± 0.03bc |
2.18 ± 0.05c |
2.17 ± 0.09c |
1.99 ± 0.02d |
2.76 ± 0.10a |
2.37 ± 0.02b |
2.74 ± 0.09a |
1.60 ± 0.23e |
| Val |
3.50 ± 0.07e |
3.37 ± 0.08f |
5.14 ± 0.06a |
5.18 ± 0.04a |
3.55 ± 0.09e |
3.32 ± 0.08f |
4.88 ± 0.03b |
4.39 ± 0.06c |
4.07 ± 0.06d |
3.50 ± 0.01e |
| Met |
1.59 ± 0.09a |
1.24 ± 0.02c |
1.49 ± 0.02b |
1.47 ± 0.05b |
1.16 ± 0.04d |
0.94 ± 0.02e |
1.01 ± 0.06e |
0.95 ± 0.06e |
0.65 ± 0.05g |
0.85 ± 0.04f |
| Ile |
3.58 ± 0.06f |
3.48 ± 0.06f |
5.67 ± 0.11a |
5.69 ± 0.02a |
4.70 ± 0.07c |
4.01 ± 0.03e |
5.37 ± 0.1b |
5.65 ± 0.15a |
4.35 ± 0.08d |
3.34 ± 0.05g |
| Leu |
5.20 ± 0.05e |
5.05 ± 0.10f |
6.98 ± 0.04a |
6.80 ± 0.05b |
6.17 ± 0.07d |
5.11 ± 0.09ef |
6.79 ± 0.12b |
6.32 ± 0.04c |
6.28 ± 0.05cd |
4.19 ± 0.09g |
| Tyr |
2.04 ± 0.03f |
1.6 ± 0.01g |
2.30 ± 0.04e |
2.58 ± 0.02c |
3.28 ± 0.03a |
2.47 ± 0.05d |
3.00 ± 0.05b |
3.01 ± 0.05b |
2.60 ± 0.04c |
2.51 ± 0.01d |
| Phe |
2.97 ± 0.05f |
3.27 ± 0.05de |
4.53 ± 0.05b |
5.20 ± 0.03a |
4.47 ± 0.24b |
3.37 ± 0.21de |
3.19 ± 0.11e |
3.38 ± 0.14d |
3.67 ± 0.02c |
3.42 ± 0.10d |
| Lys |
5.81 ± 0.03f |
5.51 ± 0.03h |
8.95 ± 0.04a |
9.04 ± 0.08a |
7.80 ± 0.09c |
6.74 ± 0.08d |
7.87 ± 0.10c |
8.10 ± 0.07b |
6.38 ± 0.10e |
5.65 ± 0.15g |
| His |
2.76 ± 0.05e |
2.49 ± 0.02g |
3.44 ± 0.05b |
3.63 ± 0.02a |
2.98 ± 0.09c |
1.75 ± 0.03h |
3.60 ± 0.02a |
3.60 ± 0.04a |
2.86 ± 0.04d |
2.61 ± 0.02f |
| Arg |
2.50 ± 0.13h |
4.89 ± 0.03b |
4.06 ± 0.04c |
4.07 ± 0.05c |
3.88 ± 0.05d |
3.03 ± 0.01g |
3.46 ± 0.02e |
3.12 ± 0.04f |
4.06 ± 0.01c |
5.09 ± 0.01a |
| Pro |
1.93 ± 0.06f |
2.17 ± 0.04e |
2.29 ± 0.05d |
2.31 ± 0.15d |
3.43 ± 0.05a |
2.21 ± 0.07de |
3.06 ± 0.07b |
3.00 ± 0.03b |
2.64 ± 0.02c |
2.21 ± 0.02de |
| E/T |
37.74 ± 0.25g |
36.26 ± 0.32h |
43.50 ± 0.07b |
45.06 ± 0.23a |
40.97 ± 0.25d |
39.85 ± 0.41e |
41.66 ± 0.21c |
41.46 ± 0.12c |
38.16 ± 0.14f |
36.33 ± 0.14h |
| BCAA |
12.27 ± 0.09f |
11.90 ± 0.11g |
17.78 ± 0.2a |
17.67 ± 0.10a |
14.42 ± 0.21e |
12.44 ± 0.19f |
17.04 ± 0.20b |
16.36 ± 0.17c |
14.71 ± 0.16d |
11.02 ± 0.12h |
| PCAA |
11.08 ± 0.11g |
12.89 ± 0.03e |
16.45 ± 0.09b |
16.74 ± 0.02a |
14.66 ± 0.22c |
11.52 ± 0.05f |
14.92 ± 0.12c |
14.82 ± 0.04c |
13.30 ± 0.08d |
13.34 ± 0.16d |
| NCAA |
25.55 ± 0.23e |
25.38 ± 0.03e |
30.98 ± 0.18a |
30.03 ± 0.08b |
25.63 ± 0.09e |
22.00 ± 0.05h |
28.45 ± 0.09d |
28.74 ± 0.09c |
24.89 ± 0.12f |
23.79 ± 0.13g |
3.7.2 Amino acid scores (AASs).
The amino acid score (AAS) is defined as the ratio of the content of essential amino acids in the test protein to that in the FAO/WHO reference protein. A value close to or greater than 100 indicates higher nutritional quality of the protein. As shown in Table 2, the AAS values for lysine in all pulse albumins were above 100 (ranging from 100.22 ± 0.61 to 164.45 ± 1.46), confirming their potential to compensate for the lysine deficiency typically found in cereal proteins.42 In addition, the scores for threonine, isoleucine, and phenylalanine + tyrosine were generally close to or exceeded 100. Notably, the AAS values of methionine + cysteine in RK (108.16 ± 0.44), WK (104.43 ± 2.69), and GP (107.62 ± 4.14) were above the reference value of 100, indicating an improved sulfur-containing amino acid profile that compensates for a common nutritional limitation of pulse proteins. In contrast, the AAS values of leucine (Leu) and valine (Val) were generally lower.
Table 2 Amino acid scores (AASs) of the 10 types of pulse albumin. Values are expressed as mean ± standard deviation (n = 3). Different lowercase letters indicate statistically significant differences within each row (P < 0.05)
| Essential amino acid |
Vigna
|
Phaseolus
|
Cicer
|
Pisum
|
Vicia
|
Lens
|
FAO/WHO |
| MB |
RB |
RK |
WK |
KCP |
DCP |
GP |
YP |
BB |
LP |
| Leu |
74.22 ± 0.68e |
72.14 ± 1.43f |
99.64 ± 0.64a |
97.18 ± 0.78b |
88.2 ± 0.93d |
73.05 ± 1.24ef |
96.98 ± 1.69b |
90.31 ± 0.58c |
102.16 ± 1.30cd |
59.82 ± 1.27g |
70 |
| Ile |
89.43 ± 1.56f |
86.97 ± 1.56f |
141.71 ± 2.79a |
142.18 ± 0.57a |
117.46 ± 1.64c |
100.15 ± 0.72e |
134.28 ± 2.56b |
141.28 ± 3.69a |
126.64 ± 2.34d |
83.38 ± 1.28g |
40 |
| Lys |
105.7 ± 0.47f |
100.22 ± 0.61h |
162.75 ± 0.8a |
164.45 ± 1.46a |
141.83 ± 1.65c |
122.51 ± 1.40d |
143 ± 1.86c |
147.24 ± 1.33b |
134.27 ± 0.21e |
102.65 ± 2.65g |
55 |
| Met + Cys |
78.46 ± 2.51d |
75.90 ± 2.10d |
108.16 ± 0.44a |
104.43 ± 2.69a |
95.26 ± 3.62b |
83.74 ± 0.47c |
107.62 ± 4.14a |
94.92 ± 2.24b |
96.83 ± 4.01b |
69.85 ± 5.87e |
35 |
| Phe + Tyr |
83.53 ± 0.90f |
81.18 ± 0.70f |
113.73 ± 1.35b |
129.82 ± 0.85a |
129.14 ± 3.46a |
97.26 ± 2.90e |
103.08 ± 1.67d |
106.64 ± 2.91c |
104.47 ± 0.76cd |
98.83 ± 1.82e |
60 |
| Thr |
100.83 ± 1.40e |
98.79 ± 0.63e |
148.79 ± 0.41b |
164.38 ± 2.82a |
110.93 ± 6.54d |
104.68 ± 1.84e |
137.27 ± 2.07c |
144.99 ± 2.01b |
113.77 ± 2.66e |
100.78 ± 2.65e |
40 |
| Val |
70.02 ± 1.39e |
67.37 ± 1.60f |
102.83 ± 1.29a |
103.53 ± 0.89a |
70.94 ± 1.89e |
66.46 ± 1.53e |
97.65 ± 0.56b |
87.73 ± 1.17c |
85.41 ± 0.92d |
70.04 ± 0.24f |
50 |
3.7.3 Essential amino acid index (EAAI).
Table 3 presents the Essential Amino Acid Index (EAAI) of the albumin fractions from the ten pulses. The EAAI is an indicator of food protein quality, reflecting the ratio of the essential amino acid content in the protein to human requirements. The closer the EAAI is to 100, the more similar the essential amino acid profile is to that of a reference protein, indicating a higher nutritional value. As presented in Table 3, EAAIs for WK (126.70 ± 0.95), RK (123.30 ± 0.54), GP (115.65 ± 0.70), YP (113.44 ± 0.52), and KCP (105.13 ± 1.08) exceeded the reference value of 100, signifying superior nutritional value. In contrast, BB (98.96 ± 0.32), DCP (87.23 ± 1.29), MB (81.11 ± 0.52), RB (78.11 ± 0.31), and LP (77.66 ± 1.20) exhibited comparatively lower EAAI values. Among all samples, WK and RK showed significantly higher EAAI scores than the other samples, indicating a more balanced essential amino acid composition and superior nutritional quality.
Table 3 Essential amino acid index (EAAI) and biological value (BV) of the 10 types of pulse albumin. Values are expressed as mean ± standard deviation (n = 3). Different lowercase letters indicate statistically significant differences within each row (P < 0.05)
|
|
Vigna
|
Phaseolus
|
Cicer
|
Pisum
|
Vicia
|
Lens
|
| MB |
RB |
RK |
WK |
KCP |
DCP |
GP |
YP |
BB |
LP |
| EAAI |
85.14 ± 0.47h |
82.39 ± 0.28i |
123.3 ± 0.54b |
126.7 ± 0.95a |
105.13 ± 1.08e |
90.77 ± 1.19g |
115.65 ± 0.70c |
113.44 ± 0.52d |
98.96 ± 0.32f |
81.98 ± 1.10i |
| BV |
81.11 ± 0.52h |
78.11 ± 0.31i |
122.7 ± 0.58b |
126.41 ± 1.04a |
102.9 ± 1.18e |
87.24 ± 1.29g |
114.36 ± 0.76c |
111.95 ± 0.57d |
96.16 ± 0.35f |
77.66 ± 1.2i |
3.7.4 Biological value (BV).
Biological value (BV) is defined as the percentage of absorbed nitrogen from dietary protein that is retained and utilized by the body, reflecting the efficiency of protein utilization after digestion and absorption. A higher BV indicates a more efficiently utilized protein. As shown in Table 3, WK (126.41 ± 1.04) and RK (122.70 ± 0.58) had the highest BV values among all samples, followed by GP (114.36 ± 0.76), YP (111.95 ± 0.57), and KCP (102.90 ± 1.18), while LP exhibited the lowest BV (77.66 ± 1.20). The variation in the BV among different pulse albumins highlights significant differences in protein quality and utilization efficiency. WK and RK, possessing the highest BV among all samples, demonstrate high efficiency of protein absorption and utilization, identifying them as high-quality protein sources. Proteins with a high BV offer a distinct advantage in practical diets, as they provide essential amino acids more effectively, thereby enhancing overall nutritional utilization.
3.8 Integrated comparison of techno-functional properties and amino acid profiles of the ten pulse albumins
Fig. 8 provides a comprehensive overview of the functional and compositional profiles of the ten pulse albumins, revealing distinct differences in processing characteristics and amino acid composition across the pulse varieties. In terms of functional properties, Phaseolus vulgaris albumins (WK and RK) consistently ranked among the top samples for water-holding capacity (WHC) and protein solubility (PS). Pisum sativum albumins (GP and YP) demonstrated excellent foaming and emulsifying capacities, whereas Vigna albumins (MB and RB) exhibited relatively low WHC and foaming ability. Within Cicer arietinum, KCP showed higher oil-holding capacity (OHC), while DCP recorded the lowest foaming performance. Amino acid analysis revealed that all albumins had multiple essential amino acid scores exceeding FAO/WHO reference values. Among them, P. vulgaris albumins (WK and RK) achieved the highest values for the essential-to-total amino acid ratio (E/T), essential amino acid index (EAAI), and biological value (BV), highlighting their superior nutritional quality. By contrast, MB and LP albumins presented the lowest E/T, EAAI, and BV, reflecting weaker protein quality. Collectively, these results underline the outstanding overall performance of WK and RK albumins, the specialized foaming and emulsifying advantages of GP and YP, and the limitations of MB, RB, and LP. MB and RB ranked among the lowest for WHC and PS, while RB and LP showed the lowest E/T, EAAI, and BV, indicating relatively lower amino acid quality.
 |
| | Fig. 8 Comparison of techno-functional properties and amino acid analysis of the ten pulse albumin. | |
4. Conclusions
This study provides a systematic comparison of the techno-functional properties and amino acid composition of albumins from ten pulse species. Overall, all pulse albumins exhibited excellent solubility (>70%) across a broad pH range, confirming their potential as highly soluble protein fractions for diverse food applications. Nevertheless, notable interspecific differences were observed, reflecting inherent variations in amino acid composition, subunit structure, and molecular characteristics. Among the tested species, Phaseolus vulgaris albumins (WK and RK) showed high solubility even at high protein concentrations, possessed excellent water-holding capacity, and demonstrated the highest E/T, EAAI, BV, and BCAA content. These features make them promising candidates for high-protein beverages and foods. Pisum sativum albumins (GP and YP) demonstrated outstanding foaming capacity and stability, along with excellent emulsifying properties, suggesting their applications in aerated and emulsion-based formulations. Within Cicer arietinum, KCP exhibited excellent emulsifying activity and oil-holding capacity, whereas DCP showed comparatively lower functionality, particularly foaming capacity. Albumins from Vigna species (MB and RB) and Lens culinaris (LP) displayed lower nutritional scores and less favorable techno-functional properties (WHC, OHC, and EAI), which may limit their use in direct applications.
In summary, these findings highlight the differences in the techno-functional properties and amino acid composition of albumins among different pulse species. These findings deepen the understanding of pulse albumin techno-functional properties and offer practical insights for the targeted use of different pulse species in food applications.
Conflicts of interest
There are no conflicts to declare.
Data availability
The data would be available on request.
Acknowledgements
This work was co-financed by the Fundamental Research Funds for the Central Universities (CXCYL2025004 and KYLH2025006) and National Natural Science Foundation of China (no. 32372440). The authors would also like to acknowledge a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
References
- X. Zhang, T. Zhang, Y. Zhao, L. Jiang and X. Sui, Structural, extraction, and safety aspects of novel alternative proteins from different sources, Food Chem., 2024, 436, 137712 CrossRef PubMed.
- C. P. Marinangeli, J. Curran, S. I. Barr, J. Slavin, S. Puri, S. Swaminathan, L. Tapsell and C. A. Patterson, Enhancing nutrition with pulses: defining a recommended serving size for adults, Nutr. Rev., 2017, 75(12), 990–1006 CrossRef PubMed.
-
FAO, FAO Statistics Data 2024, 2024, available from: https://www.fao.org/faostat/en/≠data, accessed 2 April 2024 Search PubMed.
- W. Zhang, I. D. Boateng, J. Xu and Y. Zhang, Proteins from legumes, cereals, and pseudo-cereals: Composition, modification, bioactivities, and applications, Foods, 2024, 13(13), 1974 CrossRef PubMed.
- R. J. Youle and A. H. Huang, Albumin storage proteins in the protein bodies of castor bean, Plant Physiol., 1978, 61(1), 13–16 CrossRef PubMed.
- T. D. Oluwajuyitan and R. E. Aluko, Structural and functional properties of fava bean albumin, globulin and glutelin protein fractions, Food Chem.: X, 2025, 25, 102104 Search PubMed.
- S. M. Bessada, J. C. Barreira and M. B. P. Oliveira, Pulses and food security: Dietary protein, digestibility, bioactive and functional properties, Trends Food Sci. Technol., 2019, 93, 53–68 CrossRef.
- A. Djoullah, Y. Djemaoune, F. Husson and R. Saurel, Native-state pea albumin and globulin behavior upon transglutaminase treatment, Process Biochem., 2015, 50(8), 1284–1292 CrossRef.
- J. Yang, R. Kornet, C. F. Diedericks, Q. Yang, C. C. Berton-Carabin, C. V. Nikiforidis, P. Venema, E. van der Linden and L. M. Sagis, Rethinking plant protein extraction: Albumin—From side stream to an excellent foaming ingredient, Food Struct., 2022, 31, 100254 Search PubMed.
- E. P. Chagas and L. G. Santoro, Globulin and albumin proteins in dehulled seeds of three Phaseolus vulgaris cultivars, Plant Foods Hum. Nutr., 1997, 51(1), 17–26 CrossRef.
- S. Wang, C. Guo, Z. Xing, M. Li, H. Yang, Y. Zhang, F. Ren, L. Chen and S. Mi, Dietary intervention with α-amylase inhibitor in white kidney beans added yogurt modulated gut microbiota to adjust blood glucose in mice, Front. Nutr., 2021, 8, 664976 Search PubMed.
- J. Y. Kim, S. C. Park, I. Hwang, H. Cheong, J. W. Nah, K. S. Hahm and Y. Park, Protease inhibitors from plants with antimicrobial activity, Int. J. Mol. Sci., 2009, 10(6), 2860–2872 CrossRef PubMed.
- S. Mundi and R. E. Aluko, Physicochemical and functional properties of kidney bean albumin and globulin protein fractions, Food Res. Int., 2012, 48(1), 299–306 Search PubMed.
- M. Carbonaro and A. Nucara, Legume Proteins and Peptides as Compounds in Nutraceuticals: A Structural Basis for Dietary Health Effects, Nutrients, 2022, 14(6), 1188 CrossRef PubMed.
- G. Xing, C. V. L. Giosafatto, A. Carpentieri, R. Pasquino, M. Dong and L. Mariniello, Gelling behavior of bio-tofu coagulated by microbial transglutaminase combined with lactic acid bacteria, Food Res. Int., 2020, 134, 109200 Search PubMed.
- J. S. Yang, F. F. Dias and J. M. de Moura Bell, Optimizing alkaline and enzymatic extraction of black bean proteins: a comparative study of kinetics, functionality, and nutritional properties, Sustainable Food Technol., 2025, 3(1), 188–203 Search PubMed.
- M. Habib, S. Singh, N. A. Sagar, S. Ahmad, I. Qureshi, S. Jan, K. Jan and K. Bashir, Physicochemical and functional characterization of pumpkin seed protein isolate, Sustainable Food Technol., 2025, 3(2), 445–455 Search PubMed.
- B. A. Narale, A. Mounika and A. Shanmugam, Modifications of physicochemical, functional, structural, and nutritional properties of a field bean protein isolate obtained using batch and continuous ultrasound systems, Sustainable Food Technol., 2024, 2(2), 470–484 RSC.
- Z. Zheng, M. Wang, J. Li, J. Li and Y. Liu, Comparative assessment of physicochemical and antioxidative properties of mung bean protein hydrolysates, RSC Adv., 2020, 10(5), 2634–2645 RSC.
- B. A. Bidlingmeyer, S. A. Cohen and T. L. Tarvin, Rapid analysis of amino acids using pre-column derivatization, J. Chromatogr. B: Biomed. Sci. Appl., 1984, 336(1), 93–104 Search PubMed.
- K. Shevkani, N. Singh, Y. Chen, A. Kaur and L. Yu, Pulse proteins: Secondary structure, functionality and applications, J. Food Sci. Technol., 2019, 56(6), 2787–2798 Search PubMed.
- J. Ye, N. Shi, P. Rozi, L. Kong, J. Zhou and H. Yang, A comparative study of the structural and functional properties of chickpea albumin and globulin protein fractions, Food Bioprocess Technol., 2024, 17(10), 3253–3266 Search PubMed.
- C. F. Ajibola, S. A. Malomo, T. N. Fagbemi and R. E. Aluko, Polypeptide composition and functional properties of African yam bean seed (Sphenostylis stenocarpa) albumin, globulin, and protein concentrate, Food Hydrocolloids, 2016, 56, 189–200 CrossRef.
- Y. W. Chang, I. Alli, A. T. Molina, Y. Konishi and J. I. Boye, Isolation and characterization of chickpea (Cicer arietinum L.) seed protein fractions, Food Bioprocess Technol., 2012, 5(2), 618–625 CrossRef.
- J. Mundy, J. Hejgaard and I. Svendsen, Characterization of a bifunctional wheat inhibitor of endogenous α-amylase and subtilisin, FEBS Lett., 1984, 167(2), 210–214 Search PubMed.
- A. Djoullah, Y. Djemaoune, F. Husson and R. Saurel, Native-state pea albumin and globulin behavior upon transglutaminase treatment, Process Biochem., 2015, 50(8), 1284–1292 CrossRef.
- G. V. Portari, O. L. Tavano, M. A. D. Silva and V. A. Neves, Effect of chickpea (Cicer arietinum L.) germination on the major globulin content and in vitro digestibility, Food Sci. Technol., 2005, 25, 807–812 CrossRef CAS.
-
Joint WHO/FAO/UNU Expert Consultation, Protein and Amino Acid Requirements in Human Nutrition: Report of a Joint FAO/WHO/UNU Expert Consultation, 2007 Search PubMed.
- Y. A. Adebowale, U. Schwarzenbolz and T. Henle, Protein isolates from Bambara groundnut (Voandzeia Subterranean L.): chemical characterization and functional properties, Int. J. Food Prop., 2011, 14(4), 758–775 Search PubMed.
- S. R. Trevino, J. M. Scholtz and C. N. Pace, Amino acid contribution to protein solubility: Asp, Glu, and Ser contribute more favorably than the other hydrophilic amino acids in RNase Sa, J. Mol. Biol., 2007, 366(2), 449–460 Search PubMed.
- I. D. Nwachukwu and R. E. Aluko, Physicochemical and emulsification properties of flaxseed (Linum usitatissimum) albumin and globulin fractions, Food Chem., 2018, 255, 216–225 CrossRef CAS.
- Y. A. Adebowale, U. Schwarzenbolz and T. Henle, Protein isolates from Bambara groundnut (Voandzeia Subterranean L.): chemical characterization and functional properties, Int. J. Food Prop., 2011, 14(4), 758–775 Search PubMed.
- M. Du, J. Xie, B. Gong, X. Xu, W. Tang, X. Li, C. Li and M. Xie, Extraction, physicochemical characteristics and functional properties of Mung bean protein, Food Hydrocolloids, 2018, 76, 131–140 Search PubMed.
- Z. Chen, J. Wang, W. Liu and H. Chen, Physicochemical characterization, antioxidant and anticancer activities of proteins from four legume species, J. Food Sci. Technol., 2017, 54(4), 964–972 CrossRef CAS.
- I. A. Wani, D. S. Sogi, U. S. Shivhare and B. S. Gill, Physico-chemical and functional properties of native and hydrolyzed kidney bean (Phaseolus vulgaris L.) protein isolates, Food Res. Int., 2015, 76, 11–18 CrossRef CAS.
- S. Damodaran, Protein stabilization of emulsions and foams, J. Food Sci., 2005, 70(3), R54–R66 CrossRef CAS.
- O. S. Lawal, K. O. Adebowale, B. M. Ogunsanwo, O. A. Sosanwo and S. A. Bankole, On the functional properties of globulin and albumin protein fractions and flours of African locust bean (Parkia biglobossa), Food Chem., 2005, 92(4), 681–691 CrossRef CAS.
- C. F. Chau, P. C. Cheung and Y. S. Wong, Functional properties of protein concentrates from three Chinese indigenous legume seeds, J. Agric. Food Chem., 1997, 45(7), 2500–2503 CrossRef CAS.
- K. Shevkani, N. Singh, A. Kaur and J. C. Rana, Structural and functional characterization of kidney bean and field pea protein isolates: A comparative study, Food Hydrocolloids, 2015, 43, 679–689 CrossRef CAS.
- T. D. Oluwajuyitan and R. E. Aluko, Structural and functional properties of fava bean albumin, globulin and glutelin protein fractions, Food Chem.: X, 2025, 25, 102104 CAS.
- K. Stałanowska, J. Szablińska-Piernik, A. Okorski and L. B. Lahuta, Zinc oxide nanoparticles affect early seedlings' growth and polar metabolite profiles of pea (Pisum sativum L.) and wheat (Triticum aestivum L.), Int. J. Mol. Sci., 2023, 24, 14992 CrossRef PubMed.
- S. H. Gorissen, J. J. Crombag, J. M. Senden, W. H. Waterval, J. Bierau, L. B. Verdijk and L. J. van Loon, Protein content and amino acid composition of commercially available plant-based protein isolates, Amino Acids, 2018, 50, 1685–1695 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article a,
Benjamin K.
Simpson
b and
Xin
Rui
a,
Benjamin K.
Simpson
b and
Xin
Rui
 *a
*a
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (w/v) ratio with continuous stirring for 60 min. The mixture was then centrifuged, and the upper organic solvent layer was discarded. This defatting procedure was repeated three times. The defatted flour was subsequently collected by filtration and air-dried at room temperature in a fume hood overnight to ensure complete evaporation of residual n-hexane.
3 (w/v) ratio with continuous stirring for 60 min. The mixture was then centrifuged, and the upper organic solvent layer was discarded. This defatting procedure was repeated three times. The defatted flour was subsequently collected by filtration and air-dried at room temperature in a fume hood overnight to ensure complete evaporation of residual n-hexane.

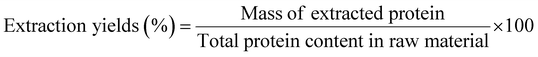
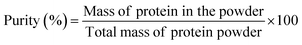
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ratio, heated in a boiling water bath for 5 min, and centrifuged (5000g, 4 °C, 10 min) to obtain electrophoretic preparations. Each sample mixture (10 µL) was then applied to precast gels. A 10–250 kDa molecular weight pre-stained protein marker (Sangon Biotech, C520010-0001) was used as the standard. Electrophoresis was performed at a constant voltage (80 V) until the dye front reached the bottom of the gel. After electrophoresis, gels were stained with Coomassie brilliant blue R-250 for 1 h and destained using a 20% methanol and 10% acetic acid solution until a clear background was obtained. The stained gels were scanned, and protein molecular weights were estimated using a ChemiDoc™ MP Imaging System (Bio-Rad Laboratories Inc., CA, USA). Each SDS-PAGE analysis was performed in triplicate.
1 (v/v) ratio, heated in a boiling water bath for 5 min, and centrifuged (5000g, 4 °C, 10 min) to obtain electrophoretic preparations. Each sample mixture (10 µL) was then applied to precast gels. A 10–250 kDa molecular weight pre-stained protein marker (Sangon Biotech, C520010-0001) was used as the standard. Electrophoresis was performed at a constant voltage (80 V) until the dye front reached the bottom of the gel. After electrophoresis, gels were stained with Coomassie brilliant blue R-250 for 1 h and destained using a 20% methanol and 10% acetic acid solution until a clear background was obtained. The stained gels were scanned, and protein molecular weights were estimated using a ChemiDoc™ MP Imaging System (Bio-Rad Laboratories Inc., CA, USA). Each SDS-PAGE analysis was performed in triplicate.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 rpm using a Polytron PT 3100 homogenizer. The total volume of foam and the liquid phase was recorded immediately after homogenization. After 30 min of quiescent standing, the remaining foam volume was measured again. The foaming capacity (FC) was calculated using eqn (5) and foam stability (FS) was determined using eqn (6). Each experiment was performed in triplicate.
500 rpm using a Polytron PT 3100 homogenizer. The total volume of foam and the liquid phase was recorded immediately after homogenization. After 30 min of quiescent standing, the remaining foam volume was measured again. The foaming capacity (FC) was calculated using eqn (5) and foam stability (FS) was determined using eqn (6). Each experiment was performed in triplicate.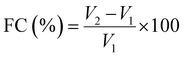

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 2 min, using a Polytron PT 3100 homogenizer. Subsequently, 50 µL aliquots were withdrawn from the bottom of the emulsion and mixed with 5 mL of 0.1% (w/v) SDS solution. The absorbance (A0) was immediately measured at 500 nm using a UV spectrophotometer, followed by a second measurement (A30) after 30 min. All experiments were performed in triplicate. The emulsifying capacity (EC) was calculated using eqn (7), and emulsion stability (ES) was calculated using eqn (8). Each experiment was performed in triplicate.
000 rpm for 2 min, using a Polytron PT 3100 homogenizer. Subsequently, 50 µL aliquots were withdrawn from the bottom of the emulsion and mixed with 5 mL of 0.1% (w/v) SDS solution. The absorbance (A0) was immediately measured at 500 nm using a UV spectrophotometer, followed by a second measurement (A30) after 30 min. All experiments were performed in triplicate. The emulsifying capacity (EC) was calculated using eqn (7), and emulsion stability (ES) was calculated using eqn (8). Each experiment was performed in triplicate.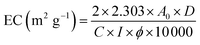

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, consistent with the previously reported ratio range of these proteins in pulses (1
3, consistent with the previously reported ratio range of these proteins in pulses (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 1
3 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.3).21 Ye et al.22 reported albumin and globulin extraction yields of 36.30% and 52.51%, respectively, in chickpeas through fractional extraction, aligning with the trends observed in this study. However, numerical differences may arise from species-specific characteristics. Fig. 2C further analyses the purity of albumin and globulin from the ten pulses. The results show that the purity of albumin ranged from 73.88% to 77.19% on a wet basis [w.b.], while that of globulin ranged from 77.87% to 81.47% [w.b.], indicating that the purity of the two types of proteins varied less among different bean species and was above 70%. The lower purity of albumin is due to its extraction from the water-soluble fraction, which may retain traces of non-protein components like carbohydrates and phenolics.
6.3).21 Ye et al.22 reported albumin and globulin extraction yields of 36.30% and 52.51%, respectively, in chickpeas through fractional extraction, aligning with the trends observed in this study. However, numerical differences may arise from species-specific characteristics. Fig. 2C further analyses the purity of albumin and globulin from the ten pulses. The results show that the purity of albumin ranged from 73.88% to 77.19% on a wet basis [w.b.], while that of globulin ranged from 77.87% to 81.47% [w.b.], indicating that the purity of the two types of proteins varied less among different bean species and was above 70%. The lower purity of albumin is due to its extraction from the water-soluble fraction, which may retain traces of non-protein components like carbohydrates and phenolics.








