Open Access Article
Open Access ArticleValorization of sunflower (Helianthus annuus L.) and canola (Brassica napus L.) meals through protein coprecipitation: physicochemical, functional, and structural characterization
Nitzia Thalía Flores-Jiménezab,
José Armando Ulloa *a and
Judith Esmeralda Urías-Silvas
*a and
Judith Esmeralda Urías-Silvas c
c
aCentro de Tecnología de Alimentos, Universidad Autónoma de Nayarit, Ciudad de la Cultura Amado Nervo, Tepic 63155, Nayarit, Mexico. E-mail: arulloa5@gmail.com
bSecretariat of Science, Humanities, Technology and Innovation, Av. Insurgentes Sur 1582, Crédito Constructor, Demarcación Territorial Benito Juárez 03940, Ciudad de Mexico, Mexico
cTecnología Alimentaria, Centro de Investigación y Asistencia en Tecnología y Diseño del Estado de Jalisco A. C., Avenida Normalistas 800, Colinas de la Normal, Guadalajara 44270, Jalisco, Mexico
First published on 23rd March 2026
Abstract
Canola–sunflower protein coprecipitates (CSPoCos) were prepared using different flour ratios, and their physicochemical, functional, and structural properties were analyzed. The crude protein content of CSPoCos ranged from 88.19% to 91.21%, which was higher than that of canola protein precipitate (CPoPc, 82.25%) and sunflower protein precipitate (SPoPc, 86.19%). In addition, protein coprecipitation (PoC) increased the apparent and compact densities and Carr and Hausner indices while decreasing the particle size and modifying the morphology, which were measured through dynamic light scattering and scanning electron microscopy, respectively. Glutelins (52.71–60.30%) were the major protein fraction in CSPoCos. In addition, PoC increased the water holding capacity, oil holding capacity, least gelation concentration, emulsifying activity index, foam capacity and foam stability. The improvement in these functional properties could be due to the formation or combination of various covalent and non-covalent interactions between the individual protein sources. Moreover, PoC modified the secondary and tertiary protein structures of CSPoCos, which was manifested by a significant increase (p < 0.05) in surface hydrophobicity, in comparison to CPoPc and SPoPc. On the other hand, the values for in vitro antioxidant capacity, in vitro digestibility, sulfhydryl groups and disulfide bridges of the CSPoCos were higher than those of CPoPc. Electrophoresis revealed that CSPoCos had subunits with molecular weights ranging from 15 to 49 kDa. Overall, CSPoCos had better functional properties than single canola and sunflower proteins, making the application of PoC an attractive alternative for obtaining protein powders with improved qualities for use as food ingredients.
Sustainability spotlightOil extraction from oilseeds such as sunflower and canola is one of the processes that generates the most by-products. Among these by-products, de-oiled sunflower and canola meals stand out due to their high protein content. This research proposes the use of de-oiled sunflower and canola meals to produce protein coprecipitates with improved physicochemical, functional and structural properties compared to the individual sources. Numerous studies have reported the valorization of proteins from oilseed industry waste as a strategy for their use as food ingredients, the development of new products, or the improvement of existing products, consistent with this research. The results obtained in this study are aligned with Sustainable Development Goals 2 (Zero Hunger), 9 (Industry, Innovation, and Infrastructure), and 12 (Responsible Consumption and Production). |
1 Introduction
The continuously growing world population demands a sufficient supply of protein through alternative sources.1 In this case, the recovery of proteins from agro-industrial by-products, including oilseed meals, represents an attractive alternative.2 Canola (Brassica napus L.) and sunflower (Helianthus annuus L.) are the second and third most important oilseed crops in the world, respectively, after soybeans.3,4 According to FAOSTAT,5 70 and 56 million metric tons of canola and sunflower were produced worldwide, respectively, with Canada and Ukraine being the largest producers of these crops. Oilseeds are an interesting source of protein since the meals obtained after oil extraction are by-products that can be valorized. Typically, residual oilseed meals from oil extraction may contain 35% to 40% protein on a dry basis. These oilseeds are recognized as an alternative protein source with potential use in human nutrition, although to date they are mainly used for animal consumption.6,7One of the alternatives for the recovery of proteins for human consumption from oilseed meals is protein precipitates (PoPc) such as concentrates or isolates.8 However, each oilseed meal has limited nutritional value due to the presence of anti-nutrients or a low functional quality, which restricts its use as a food ingredient.9,10 Therefore, there has been interest in formulating food products by combining proteins to solve these drawbacks, which can be achieved through PoB (protein blending) and PoC (protein coprecipitation) using two or more sources (plant–plant, animal–animal or plant–animal proteins).11–13 The differences between these protein products are based on the preparation method. PoB can be obtained through the direct mixing of protein isolates or concentrates, while PoC can be prepared by precipitation or by heating.14
The formation of PoB and PoCos involves different interactions that give rise to different structures. These structures present in PoB and PoCos can have a synergistic effect on their techno-functional, nutritional and biological properties,9,11,15,16 depending mainly on the various intrinsic properties (type of proteins, protein concentration, amino acid composition, structure, conformation, molecular weight, and surface properties) of the protein sources and processing conditions such as pH, temperature, ionic force, meal: solvent ratio, etc.12,17–20
For example, Wu et al.13 studied the effect of mixing soybean and zein proteins. The results of this study revealed that the soybean-zein (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; w/w at pH 7) PoB improved the solubility, foaming properties and water holding capacity, in contrast to the zein proteins. These researchers pointed out that the increase in these techno-functional properties was a consequence of the fusion of the two proteins and the modification of their secondary structure. In addition, Zhou et al.21 observed that PoB obtained from pea and grass carp protein (1
1; w/w at pH 7) PoB improved the solubility, foaming properties and water holding capacity, in contrast to the zein proteins. These researchers pointed out that the increase in these techno-functional properties was a consequence of the fusion of the two proteins and the modification of their secondary structure. In addition, Zhou et al.21 observed that PoB obtained from pea and grass carp protein (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; w/w at pH 7) isolates enhanced the solubility and foaming properties compared to grass carp protein isolate. These results are due to the fact that the protein molecules of pea and grass carp have opposite charges, and thus their intermolecular electrostatic interactions are strengthened to generate soluble protein aggregates.
1; w/w at pH 7) isolates enhanced the solubility and foaming properties compared to grass carp protein isolate. These results are due to the fact that the protein molecules of pea and grass carp have opposite charges, and thus their intermolecular electrostatic interactions are strengthened to generate soluble protein aggregates.
Conversely, in the case of PoC, Zhang et al.22 demonstrated that the association of ginseng and Schisandra (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; w/w) proteins in the form of PoCo by alkaline extraction (pH 9) and isoelectric precipitation (pH 3.5) increased the water and oil holding capacities compared to individual proteins. This improvement resulted from the increase in hydrogen bonding interactions and hydrophilic groups exposed under alkaline conditions. In another study, Kristensen et al.16 reported that pea and whey (20
1; w/w) proteins in the form of PoCo by alkaline extraction (pH 9) and isoelectric precipitation (pH 3.5) increased the water and oil holding capacities compared to individual proteins. This improvement resulted from the increase in hydrogen bonding interactions and hydrophilic groups exposed under alkaline conditions. In another study, Kristensen et al.16 reported that pea and whey (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80; v/v) PoCos, obtained by alkaline extraction (pH 11) and isoelectric precipitation (pH 4.6), produced new electrostatic interactions and disulfide bonds between these proteins, increasing their solubility and surface hydrophobicity.
80; v/v) PoCos, obtained by alkaline extraction (pH 11) and isoelectric precipitation (pH 4.6), produced new electrostatic interactions and disulfide bonds between these proteins, increasing their solubility and surface hydrophobicity.
To date, this work constitutes the first report on obtaining PoCos from canola and sunflower as an alternative protein source. Therefore, the objective was to determine the physicochemical, functional, and structural attributes of CSPoCos to evaluate their potential application as a novel food ingredient.
2 Materials and methods
2.1 Material and chemicals
Canola meal (CM) was provided by Forrajes y Alimentos San Cayetano, S. de R.L. de C.V. (Tepic, Nayarit, México), while sunflower meal (SM) was obtained from New Country Organics, Inc. (Lubbock, Texas, USA). The contents of crude protein (N × 6.25), moisture, ash, fat, and total carbohydrate contents for CM, according to the AOAC19 methods, were 39.72% ± 0.32%, 9.76% ± 0.14%, 7.05% ± 0.04%, 2.96% ± 0.25%, and 31.74% ± 0.81%, respectively. The proximal composition of SM is presented in Section 2.2, since it was necessary to remove a significant amount of oil. All chemicals employed in this research were reagent grade and purchased from Sigma-Aldrich, Fermont and J.T. Baker (Ciudad de México, México).2.2 Preparation of defatted sunflower flour (DSF)
Because SM contained a considerable amount of lipids (21.23% ± 0.25%), it was necessary to apply a step to reduce the percentage of this component, according to the following procedure. Firstly, lots of 100 g of SM were placed in 1 L of ethyl ether with stirring at 500 rpm for 1 h, during two successive cycles and replacing the spent solvent with fresh solvent. The material obtained from the second defatting cycle was placed in a plastic container for complete desolventization, and then in an extractor hood for 12 h to obtain DSF, which was stored in airtight bags at room temperature for subsequent characterization. The contents of crude protein (N × 6.25), moisture, ash, fat, and total carbohydrates of DSF were 33.54% ± 0.89%, 8.81% ± 0.04%, 6.76% ± 0.80%, 0.49% ± 0.18%, and 50.40% ± 0.61%, respectively.2.3 Preparation of CPoPc, SPoPc and CSPoCos
CPoPc and SPoPc were obtained using 100% CM and DSF, respectively, as the source of proteins. CSPoCos were obtained employing batches of 200 g pastes, with the following proportions: 70%CM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30%DSF, 50%CM
30%DSF, 50%CM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50%DSF, and 30%CM
50%DSF, and 30%CM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70%DSF (w/w) by alkaline extraction and isoelectric precipitation, following the method reported by Tian et al.12 Briefly, CM, DSF and mixtures thereof were suspended in distilled water (1
70%DSF (w/w) by alkaline extraction and isoelectric precipitation, following the method reported by Tian et al.12 Briefly, CM, DSF and mixtures thereof were suspended in distilled water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, w/v), adjusted to pH 11 with 1 M NaOH by magnetic stirring at 20 °C for 45 min, and the insoluble residue was separated by centrifugation at 8000×g and 14 °C for 10 min. After, the pH of the protein suspensions was adjusted to 4 (for CM) and 4.5 (for DSF and mixtures) with 1 M HCl by magnetic stirring at 20 °C for 30 min. Subsequently, the slurries were centrifuged at 8000×g at 14 °C for 10 min and PoPc and PoCos were re-suspended in distilled water in a ratio of 1
20, w/v), adjusted to pH 11 with 1 M NaOH by magnetic stirring at 20 °C for 45 min, and the insoluble residue was separated by centrifugation at 8000×g and 14 °C for 10 min. After, the pH of the protein suspensions was adjusted to 4 (for CM) and 4.5 (for DSF and mixtures) with 1 M HCl by magnetic stirring at 20 °C for 30 min. Subsequently, the slurries were centrifuged at 8000×g at 14 °C for 10 min and PoPc and PoCos were re-suspended in distilled water in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v) at pH 7 with 1 M NaOH. Finally, the protein dispersions were lyophilized in a freeze dryer model FreeZone 10 L (Labconco, USA) to obtain CPoPc, SPoPc and CSPoCos (CSPoCo70/30, CSPoCo50/50 and CSPoCo30/70, where the first subindex in each material represents the CM proportion and the second subindex is the DSF proportion used in this preparation).
10 (w/v) at pH 7 with 1 M NaOH. Finally, the protein dispersions were lyophilized in a freeze dryer model FreeZone 10 L (Labconco, USA) to obtain CPoPc, SPoPc and CSPoCos (CSPoCo70/30, CSPoCo50/50 and CSPoCo30/70, where the first subindex in each material represents the CM proportion and the second subindex is the DSF proportion used in this preparation).
2.4 Extraction yield and protein purity
The extraction yield of the samples was calculated using two parameters (weight yield and protein yield). The weight yield was determined as the ratio between the total weight of the recovered CPoPc, SPoPc and/or CSPoCos and the initial weight of CM and/or DSF used in the extraction process (eqn (1)). The protein yield was estimated by comparing the total protein content in the recovered CPoPc, SPoPc and/or CSPoCos and the initial protein content in CM and/or DSF (eqn (2)). The protein purity was calculated according to eqn (3), as follows:
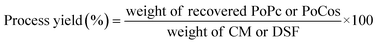 | (1) |
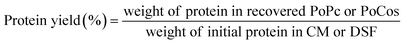 | (2) |
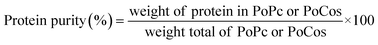 | (3) |
2.5 Composition and physicochemical characteristics
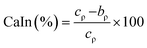 | (4) |
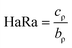 | (5) |
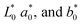 numbers of the white pattern plate employed as the reference were 94.44, −0.20 and 3.87, respectively. The total variation in color (ΔE) was estimated as:
numbers of the white pattern plate employed as the reference were 94.44, −0.20 and 3.87, respectively. The total variation in color (ΔE) was estimated as:
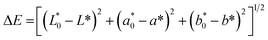 | (6) |
In addition, the hue angle (Hab) was calculated as the arc tangent of the ratio of yellowness to redness, as shown in eqn (7).
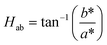 | (7) |
Chroma (C*) is considered as the purity of color or colorfulness of a sample. It was determined as the Euclidean distance of the color vector (L, a, b) for a sample from the lightness, axis as shown in eqn (8)
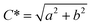 | (8) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/v) ratio with 1 h of stirring at 4 °C was used to extract Al, which was recovered in the supernatant obtained by centrifugation for 20 min at 6000×g and stored under refrigeration at 6 °C for subsequent analysis. To the resulting precipitate, 30 mL of a 50 mM tris-HCl + 0.4 M NaCl solution (pH 8) was added for Gl extraction, and the mixture was centrifuged under the same conditions. Next, 30 mL of 70% ethanol was added to the new precipitate to obtain Pr, and subsequently 30 mL of 0.1 M NaOH was added to the final precipitate to recover Gx. The separation and storage of the supernatant in each extraction stage were performed under the same conditions as for the albumin fraction. The total protein content of each fraction was measured using the Bradford assay26 with albumin from bovine serum as the reference.
10 (w/v) ratio with 1 h of stirring at 4 °C was used to extract Al, which was recovered in the supernatant obtained by centrifugation for 20 min at 6000×g and stored under refrigeration at 6 °C for subsequent analysis. To the resulting precipitate, 30 mL of a 50 mM tris-HCl + 0.4 M NaCl solution (pH 8) was added for Gl extraction, and the mixture was centrifuged under the same conditions. Next, 30 mL of 70% ethanol was added to the new precipitate to obtain Pr, and subsequently 30 mL of 0.1 M NaOH was added to the final precipitate to recover Gx. The separation and storage of the supernatant in each extraction stage were performed under the same conditions as for the albumin fraction. The total protein content of each fraction was measured using the Bradford assay26 with albumin from bovine serum as the reference.2.6 Functional characteristics
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 1 min. Then, 50 µL of emulsion from the bottom of the tube was taken after 0 min (for EAcIn) and 10 min (for EStIn) and added to 5 mL of 0.1% SDoS (sodium dodecyl sulfate) solution. After shaking in a vortex mixer for 5 s, the absorbance of the emulsions was recorded at 500 nm. EAcIn and EStIn were calculated using the following equations:
000 rpm for 1 min. Then, 50 µL of emulsion from the bottom of the tube was taken after 0 min (for EAcIn) and 10 min (for EStIn) and added to 5 mL of 0.1% SDoS (sodium dodecyl sulfate) solution. After shaking in a vortex mixer for 5 s, the absorbance of the emulsions was recorded at 500 nm. EAcIn and EStIn were calculated using the following equations:
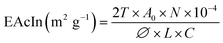 | (9) |
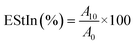 | (10) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm and poured into a graduated tube. The foam volume at 1 min was recorded as V1min, the foam volume at 20 min as V20min, and FmC and FmS were calculated as follows:
000 rpm and poured into a graduated tube. The foam volume at 1 min was recorded as V1min, the foam volume at 20 min as V20min, and FmC and FmS were calculated as follows:
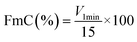 | (11) |
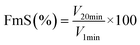 | (12) |
2.7 Biochemical properties
| IVPoDi (%) = 210.464 − 18.10(Z) | (13) |
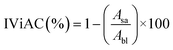 | (14) |
2.8 Structural properties
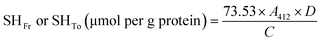 | (15) |
 | (16) |
2.9 Statistical analysis
The physicochemical, functional, biochemical and structural characteristics of PoPc and CSPoCos were reported as the mean ± standard deviation of three batches. Data analysis involved one-way ANOVA and comparison of means was performed using Fisher's Protected LSD (statistical significance considered at p < 0.05) using Statistica software version 7.1 (TIBCO Software, Inc., USA).3 Results and discussion
3.1 Extraction yield and protein purity
The yield and purity of protein materials are important factors associated with their production methods, which determine their economic feasibility.35 The results for the product yield, protein recovery, and purity of PoPc and CSPoCos are presented in Table 1. The product yield ranged from 5.20%–17.10%, following the order of CPoPc < CSPoCo70/30 < CSPoCo50/50 < CSPoCo30/70 < SPoPc. These results are comparable to those of Wintersohle et al.,36 who found a product yield ranging from 8.3% to 18.89% for mung bean PoPc obtained by alkaline extraction and isoelectric precipitation. Meanwhile, the protein recovery reached values of 11.76% to 53.07% in the order of CPoPc < CSPoCo70/30 < CSPoCo50/50 < CSPoCo30/70 < SPoPc. The maximum protein recovery obtained in this study was similar to that reported for the PoPc of sunflower (55.67%),6 Hermetia illucens (55%),37 and Japanese quince (54%).38 In the case of protein purity, PoC significantly increased (p < 0.05) this parameter from 82.18% and 86.22% for CPoPc and SPoPc, respectively, to values ranging from 88.25% to 92.20% for CSPoCos (Table 1). The purity of the protein obtained in the present study for both PoPc (82.18–86.22%) and PoCos (88.25–92.20%) ranged between those reported for Japanese quince PoPc (83.88%).39| Samples | Process yield (%) | Protein recovery (%) | Protein purity (%) |
|---|---|---|---|
| a Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same column indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||
| CPoPc | 5.20 ± 0.26e | 11.76 ± 0.60e | 82.18 ± 0.02e |
| SPoPc | 17.10 ± 0.36a | 53.07 ± 1.16a | 86.22 ± 0.06d |
| CSPoCo70/30 | 11.40 ± 0.53d | 29.92 ± 1.37d | 88.25 ± 0.13c |
| CSPoCo50/50 | 13.27 ± 0.25c | 37.18 ± 0.70c | 89.15 ± 0.03b |
| CSPoCo30/70 | 16.23 ± 0.40b | 50.48 ± 1.27b | 92.20 ± 0.02a |
3.2 Composition and physicochemical characteristics
| Components (%) | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||||
| Protein | 82.25 ± 0.56d | 86.19 ± 0.32c | 88.19 ± 0.45b | 89.17 ± 0.54b | 91.21 ± 0.92a |
| Moisture | 4.42 ± 0.13a | 3.20 ± 0.04c | 3.63 ± 0.03b | 2.80 ± 0.09d | 3.21 ± 0.07c |
| Ash | 3.83 ± 0.11a | 3.67 ± 0.29a | 2.74 ± 0.08b | 2.78 ± 0.02b | 2.40 ± 0.13c |
| Lipids | 1.23 ± 0.30a | 0.95 ± 0.58ab | 0.91 ± 0.57b | 0.86 ± 0.02b | 0.87 ± 0.10b |
| Carbohydrates | 8.27 ± 0.55a | 5.99 ± 0.37b | 4.53 ± 0.73c | 4.39 ± 0.52c | 2.31 ± 0.43d |
The protein content of CSPoCos was higher than pea-grass carp (87.91%)19 and soybean-brewers’ spent grain (83.67%)40 PoCos but lower than those of soybean-wheat (91.5–94%)12 and sheep whey-casein (97.5%)41 PoCos.
The proximate composition of PoPc from canola42 and sunflower43 was similar to that of CPoPc and SPoPc obtained in this study.
| Properties | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a bρ = bulk density, cρ = compact density, CaIn = Carr's index, HaRa = Hausner's ratio, aw = water activity, and Trb = turbidity. Values are the mean of three determinations of independent batches ± standard deviations. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||||
| Density | |||||
| bρ (g cm−3) | 0.326 ± 0.01b | 0.358 ± 0.01a | 0.370 ± 0.01a | 0.372 ± 0.01a | 0.366 ± 0.00a |
| cρ (g cm−3) | 0.430 ± 0.01d | 0.436 ± 0.02d | 0.499 ± 0.01ab | 0.488 ± 0.01bc | 0.482 ± 0.00c |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| Flow characteristics | |||||
| CaIn (%) | 24.24 ± 2.19a | 17.74 ± 1.72b | 25.69 ± 1.21a | 23.73 ± 1.76a | 24.05 ± 1.02a |
| Flow | Fair | Good | Fair | Fair | Fair |
| HaRa | 1.32 ± 0.04a | 1.22 ± 0.03b | 1.35 ± 0.02a | 1.31 ± 0.03a | 1.32 ± 0.02a |
| Cohesion | Intermediate | Intermediate | Intermediate | Intermediate | Intermediate |
| aw | 0.41 ± 0.01a | 0.20 ± 0.03c | 0.25 ± 0.01b | 0.14 ± 0.01d | 0.11 ± 0.01e |
| Trb | 0.11 ± 0.00e | 0.90 ± 0.01a | 0.34 ± 0.01d | 0.41 ± 0.01c | 0.48 ± 0.01b |
Regarding cρ, significantly (p < 0.05) higher values were observed for CSPoCos (0.482–0.499 g cm−3) compared to CPoPc (0.430 g cm−3) and SPoPc (0.436 g cm−3) (Table 3). According to various studies, bρ and cρ are influenced by factors such as particle size, size distribution, shape, surface area, roughness, and particle arrangement of powder products.45,46 The bρ and cρ values obtained in this study for CPoPc and SPoPc of 0.326 g cm−3 and 0.430 g cm−3 and 0.358 g cm−3 and 0.436 g cm−3 were higher than those of 0.156 g cm−3 and 0.200 g cm−3 obtained for soursop seed PoPc, respectively.28
Conversely, HaRa measures the cohesion of particles, and therefore its value determines high or low fluidity in powders.48 Powders with an HaRa of 1.2 are classified as free-flowing, while those with values of 1.2–1.4 or >1.4 are considered to be of intermediate or high cohesion, respectively.30 CPoPc, SPoPc, CSPoCo70/30, CSPoCo50/50, and CSPoCo30/70 had HaRa values of 1.32, 1.22, 1.35, 1.31 and 1.32, respectively, and therefore were classified as intermediate cohesion powders (Table 3). Specifically, the optimal CaIn and HaRa values depend on the potential applications of the powdered products. Therefore, in some cases, higher or lower CaIn and HaRa values may be preferable.48
López-Mártir et al.28 reported for soursop seed PoPc fair flowability (CaIn of 22%) and intermedia cohesiveness (HaRa of 1.28), although Rodríguez-Rivera et al.32 found that groundnut paste PoPc presented good flowability (CaIn of 18.33%) and intermediate cohesiveness (HaRa of 1.22).
The aw values of 0.41, 0.21 and 0.31 have been reported for PoPc of canola,42 guamuchil,24 and gourd,50 respectively.
| Color | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a L* = lightness, a* = +redness, −greenness, b* = +yellowness, −blueness, ΔE = total variation of color, C* = chroma, Hab = hue angle. Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||||
| L* | 51.01 ± 0.37b | 40.45 ± 0.54e | 54.07 ± 0.38a | 47.34 ± 0.55c | 44.41 ± 0.83d |
| a* | 2.11 ± 0.03b | −0.92 ± 0.07e | 2.84 ± 0.21a | 1.16 ± 0.10c | −0.45 ± 0.02d |
| b* | 18.56 ± 0.16a | 4.72 ± 0.16e | 17.21 ± 0.76b | 11.27 ± 0.18c | 8.32 ± 0.22d |
| ΔE | 49.25 ± 0.21d | 57.68 ± 0.62a | 46.10 ± 0.56e | 51.10 ± 0.01c | 53.57 ± 0.27b |
| C* | 18.19 ± 0.32a | 4.81 ± 0.17e | 17.44 ± 0.79b | 11.33 ± 0.19c | 8.33 ± 0.22d |
| Hab | 83.19 ± 0.44d | 100.88 ± 0.30a | 80.65 ± 0.37e | 84.13 ± 0.44c | 93.14 ± 0.16b |
All PoPc and PoCos showed positive values for a* (except SPoPc and CSPoCo30/70) and b* chromaticity (Table 4). High b* values indicate a yellow tone with a slight reddish tone due to small a* values (Fig. 1). SPoPc and CSPoCo30/70 exhibited a slightly greenish hue, as expressed by their negative a* values, because these protein samples contained a considerable amount of sunflower proteins compared to the other samples (Fig. 1).
In a study with pea-grass carp, PoCos19 were found to have L*, a* and b* values of 74.21, −0.44, and 12.92, respectively. In their study, Flores-Jiménez et al.42 reported L*, a*, and b* values of 51.27, 3.38, and 15.17 for canola PoPc, respectively, while Alexandrino et al.6 obtained values of 51.66 (L*), 7.02 (a*), and 2.21 (b*) for sunflower PoPc.
Hab indicates whether an object is red (0°), yellow (90°), green (180°) and (270°).50 SPoPc and CSPoCo30/70 showed Hab values of 100.88° and 93.14°, respectively, confirming their greenish–yellowish tones due to a higher proportion of sunflower protein. In contrast, the lower Hab values for CPoPc (83.19°), CSPoCo50/50 (84.13°), and CSPoCo70/30 (80.65°) indicated their reddish–yellowish tones, which can be associated with a higher content of canola proteins. Furthermore, CPoPc and CSPoCo30/70 presented higher C* values compared to SPoPc, CSPoCo50/50 and CSPoCo70/30, indicating their greater color intensity/vividity.
Naik et al.54 and López-Mártir et al.55 previously reported C* values of 18.25 and 13.87 in melon and noni PoPc, respectively, which were higher than those obtained for SPoPc, CSPoCo50/50 and CSPoCo30/70 in this study.
| Protein fraction (%) | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||||
| Albumin (Al) | 9.33 ± 0.51d | 46.22 ± 1.11a | 31.28 ± 1.01c | 30.57 ± 1.64c | 38.74 ± 1.13b |
| Globulin (Gl) | 9.31 ± 0.63a | 9.64 ± 0.52a | 7.15 ± 0.15c | 8.45 ± 0.76ab | 7.63 ± 0.53bc |
| Glutelin (Gx) | 78.15 ± 0.69a | 43.77 ± 1.09d | 60.30 ± 0.43b | 59.96 ± 2.04b | 52.71 ± 1.13c |
| Prolamin (Pr) | 3.21 ± 0.53a | 0.98 ± 0.36bc | 1.26 ± 0.07b | 1.02 ± 0.09bc | 0.92 ± 0.06c |
As in this study, Gx and Al were the main protein fractions in PoPc obtained from canola (glutelins 57.18% and albumins 23.09%),42 orange seeds (glutelins 70.13% and albumins 10.39%),56 and noni seeds (glutelins 64.62% and albumins 24.12%).55
3.3 Functional characteristics
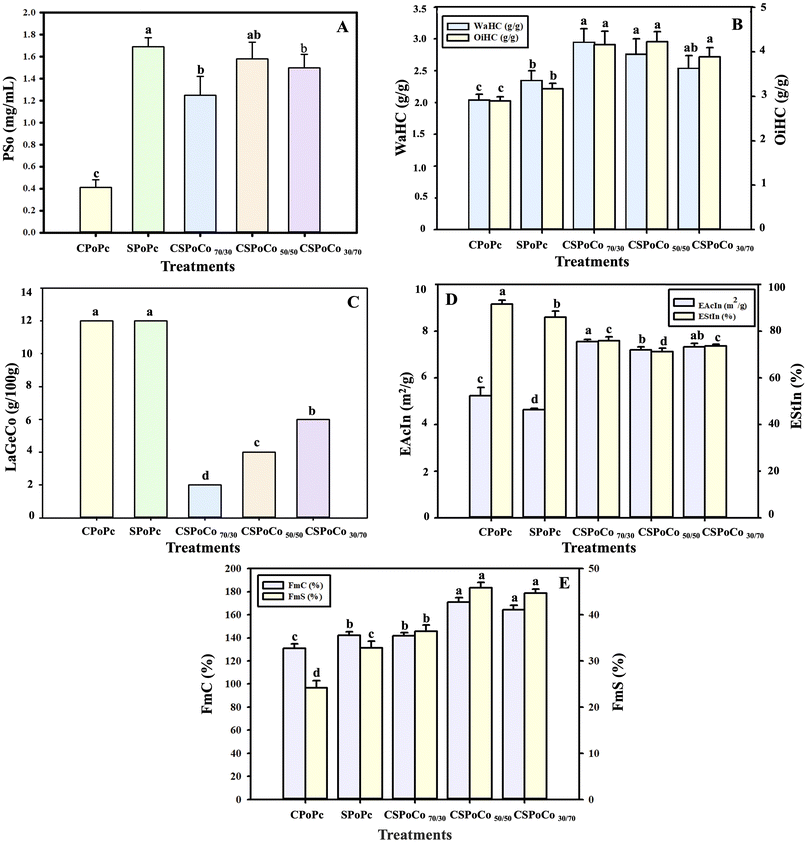
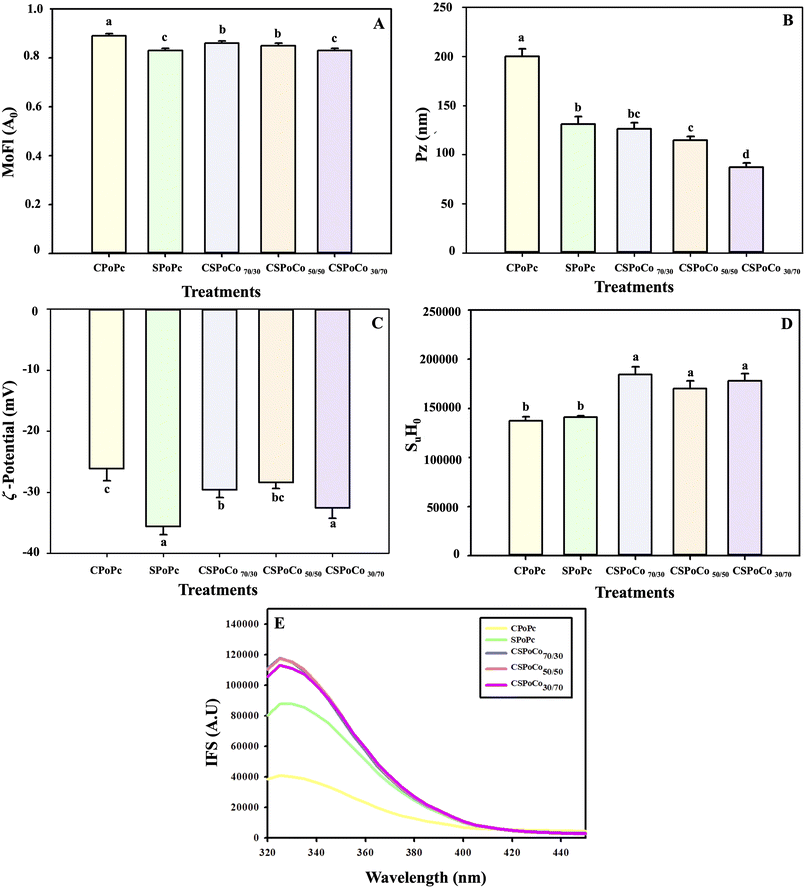
The increase in the PSo of CSPoCos was due to the higher inclusion of sunflower proteins that contain a higher proportion of the Al protein fraction (Table 5). Moreover, the fusion of canola and sunflower polymers may have strengthened the intermolecular electrostatic interactions to generate soluble protein aggregates, as can be observed in Fig. 6B. In a study by Zhou et al.21 with PoCo from pea and grass carp using alkaline extraction-isoelectric precipitation, an increase in PoSo (at pH 7) of 14.3% and 22.9% was observed, in contrast to the pea and grass carp proteins, respectively. In addition, Tian et al.12 reported that PoCo from soybean and wheat proteins had a higher PoSo value (pH 7) than soybean and wheat protein isolates. This modification in PoCo from these protein sources was due to the secondary and tertiary structural changes, in contrast to soybean and wheat protein isolates.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3; w/w), WaHC increased by 6.6% and 9.8%, in contrast to pea protein isolate and lentil protein isolate, respectively.59
3; w/w), WaHC increased by 6.6% and 9.8%, in contrast to pea protein isolate and lentil protein isolate, respectively.59Conversely, the OiHC of CSPoCos (3.88–4.22 g oil per g protein) was significantly higher (p < 0.05) compared to that of CPoPc (2.89 g oil per g protein) and SPoPc (3.17 g oil per g protein) (Fig. 2B). These results may be due to PoC inducing greater exposure of the hydrophobic groups (Fig. 6D), which facilitated better interaction with the oil and led to an increase in OiHC.
| Sulfhydryl groups (µmol g−1 protein) | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower pastes. | |||||
| Free (SHFr) | 12.47 ± 0.48d | 17.87 ± 0.07a | 16.05 ± 0.04b | 15.02 ± 0.26c | 14.98 ± 0.19c |
| Total (SHTo) | 16.20 ± 0.68b | 20.37 ± 0.3a | 16.67 ± 0.42b | 16.18 ± 0.13b | 16.79 ± 0.49b |
| Bridges (SSBr) | 1.87 ± 0.60b | 1.25 ± 0.28a | 0.31 ± 0.40c | 0.58 ± 0.17c | 0.91 ± 0.41bc |
| Secondary structure (%) | CPoPc | SPoPc | CSPoCo70/30 | CSPoCo50/50 | CSPoCo30/70 |
|---|---|---|---|---|---|
| a Values are the mean of three determinations of independent batches ± standard deviation. Different letters within the same row indicate significant differences (p < 0.05) among the groups. CSPoCo70/30 = 70% canola/30% sunflower, CSPoCo50/50 = 50% canola/50% sunflower and CSPoCo30/70 = 30% canola/70% sunflower. The subscripts for each coprecipitate correspond to the proportions of canola and sunflower meals. | |||||
| β-sheet | 49.55 ± 0.68c | 60.96 ± 1.10a | 52.13 ± 0.45b | 51.42 ± 0.28b | 52.58 ± 0.69b |
| α-helix | 8.40 ± 0.87b | 14.64 ± 0.53a | 8.79 ± 0.42b | 8.67 ± 0.77b | 8.64 ± 0.58b |
| β-turn | 29.77 ± 0.54a | 10.72 ± 1.08d | 23.81 ± 0.97c | 28.39 ± 0.35b | 24.11 ± 0.44c |
| Random coil | 11.57 ± 0.39b | 13.67 ± 0.54a | 14.44 ± 0.32a | 12.07 ± 0.15b | 14.63 ± 0.45a |
In addition, PoC significantly (p < 0.05) increased the FmS of CSPoCos with respect to CPoPc and SPoPc. The highest increase in FmS of 47.26% and 28.44% was observed for CSPoCo50/50 compared to CPoPc and SPoPc, respectively (Fig. 2E). In general, it was observed that the higher the proportion of sunflower proteins in PoCos, the better the foaming properties achieved. Furthermore, the increase in the FoP of CSPoCos was possibly due to the interaction between the canola and sunflower protein molecules, resulting in smaller particles and an increase in SuH0 (Fig. 6B and D). In a study by Kristensen et al.9 with a PoCo of pea and whey, an increase in foaming properties was reported, in contrast to the pea PoPc, possibly due to the interaction of the polymers used, which generated smaller particles.
3.4 Biochemical properties
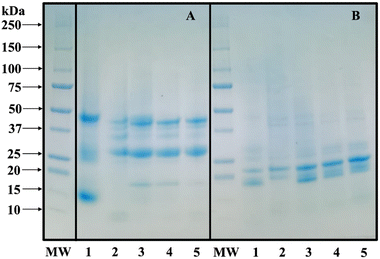
Under non-reducing conditions (Fig. 3A), the molecular weight distribution of CPoPc consisted of four main fractions with the following molecular weights: ∼49 kDa, 30 kDa, 25 kDa, and 15 kDa, which correspond to the molecular weights of cruciferin (∼50–20 kDa) and napin (∼7–11 kDa) of the protein fractions reported by Cháirez-Jiménez et al.7 However, in the presence of the reducing agent β-mercaptoethanol (Fig. 3B), the band at ∼49 kDa for CPoPc was dissociated into its two polypeptides (∼20 kDa and ∼30 kDa), as reported by Flores-Jiménez et al.42 In the case of SPoPc, the main bands detected were ∼49, 38, 35, 29, 25 and 8 kDa under non-reducing conditions (Fig. 3A). The subunits with a molecular weight between 20 and 50 kDa correspond to helianthinin (11S globulins), while those between 10–19 kDa correspond to 2S albumin. Under reducing conditions (Fig. 3B), 11S globulin dissociated into its two acidic subunits with a molecular weight of ∼38–49 kDa and 25–29 kDa, as well as in its basic subunits with a molecular weight of 20–23 kDa.67
Alternatively, under non-reducing conditions, CSPoCos presented seven bands with molecular weights of ∼49, 38, 35, 29, 25, 18, and 15 kDa, which are related to the 11S globulins and 2S albumins fractions of canola and sunflower proteins (Fig. 3A). Under reducing conditions (Fig. 3B), the molecular weight distribution of CSPoCos consisted of nine main fractions, with molecular weights of ∼49, 38, 35, 29, 25, 23, 20, 18, and 12 kDa. Interestingly, all the protein bands from canola and sunflower were present in all CSPoCos under both reducing and non-reducing conditions. However, under non-reducing conditions, an additional 18 kDa band appeared in CSPoCos (Fig. 3A), with this band in CSPoCos70/30 being more intense compared to the other coprecipitates, which is likely due to its higher canola content. According to Guidi et al.,59 protein interactions can be observed through the appearance or disappearance of bands in the electrophoretic profile. This suggests a strong interaction between the polypeptide chains of both protein sources, which is directly related to their functional properties (Fig. 2). Similar results were observed in a PoCo of Bambara groundnut protein isolate and fish collagen, which showed the presence of the characteristic bands of both polymers.65
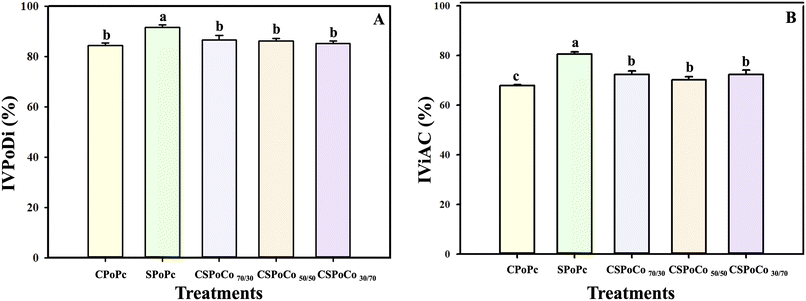
Other studies have revelated IVPoDi values of 83.9% and 90.7% for canola42 and sunflower6 PoPc, respectively, which are similar to the values obtained for CSPoCos and PoPc in this study.
Alu'datt et al.18 found an increase of up to 50.4% in IViAC for soybean and flaxseed PoCo compared to soybean PoPc. The authors of this study concluded that the increase in IViAC is due to the fact that PoC increases the proportion of phenolic compounds and certain amino acids with antioxidant activity.
3.5 Structural characterization
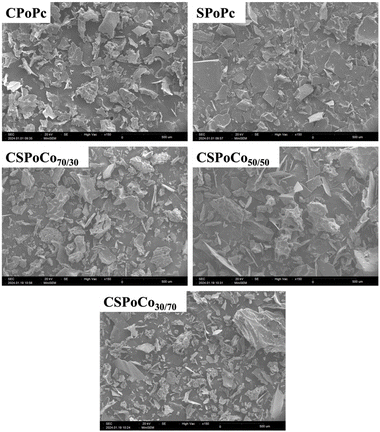
According to a previous study, the presence and proportion of protein fractions based on solubility can influence the microstructure of proteins, as seen by SEM.30 Thus, Al gives a lamellar appearance, Gx a rough surface with several tiny pores, and Gl a structure composed of small globules, which is consistent with the amount of protein fractions of Gx, Al and Gl in CPoPc, SPoPc and CSPoCos. For example, CPoPc had the highest Gx content (Table 5), presenting a more porous and rougher surface, while SPoPc had a higher proportion of Al, showing lamellar structures. In addition, CSPoCos was observed to have a greater balance between Gx and Al fractions, and thus a combination of porous/rough and soft/smooth structures was detected (Table 5 and Fig. 5). Therefore, the surface appearance of CSPoCos observed through SEM appears to be associated with the protein composition of canola and sunflower, which in turn could also influence its physicochemical and functional properties.74
The ζ-potential is related to the net surface charge and the ionic strength of proteins, affecting their adsorption and unfolding of their polypeptide chains, and therefore their functionality.81 The ζ-potential values of CPoPc, SPoPc, and CSPoCos were negative (Fig. 6C). PoC increased the negative value of ζ-potential in the range of 8.09% (CSPoCo70/30)–19.94% (CSPoCo30/70) compared with CPoPc. On the contrary, PoC diminished the negative values for CSPoCo70/30 (20.27%) and CSPoCo50/50 (25.22%) with respect to SPoPc.
Changes in ζ-potential may be associated with modifications in the amount of ionizable amino and carboxyl groups exposed on the surface caused by PoC.80 Previous research has shown that protein dispersions with high absolute ζ-potentials are characterized by strong intermolecular electrostatic repulsion, which is sufficient to resist agglomeration, thereby improving the dispersibility and stability of the protein system.10 Therefore, ζ-potential is related to the improvement in the functional properties of CSPoCos because the interactions between the two polypeptides inhibited structural folding and maintained their charged surface (Fig. 2). PoCos of rice and casein78 and rice and cod10 proteins also increased the negative value of ζ-potential compared to the individual sources.
Overall, PoC increased the SHFr content (20.13–28.71%) of CSPoCos, in contrast to CPoPc, whereas the S–SBr content decreased in CSPoCos in the ranges of 105.5–503.2% and 37.4–303.2% in contrast to CPoPc and SPoPc, respectively, which could imply unfolding of the polypeptide chain and thus a modification in the initial proportion of sulfhydryl groups in the new polymeric structures. Conversely, the SHTo content did not change significantly (p > 0.05) in CSPoCos with respect to CPoPc, but its value was significantly reduced (p < 0.05) compared to SPoPc (Table 6).
Interestingly, in CSPoCos, it was observed that almost all the sulfhydryl groups corresponded to SHFr, which also demonstrated the structural rearrangement of its proteins due to the effect of PoC, similar to the effect caused by other physical treatments such as ultrasound81 and high pressure.34
Table 7 shows the secondary structure content of PoPc and PoCos from canola and sunflower. Compared with CPoPc and SPoPc, the secondary structures of CSPoCos were significantly different (p < 0.05). In CPoPc, SPoPc, and CSPoCos, β-sheets were the predominant secondary structures, followed by β-turns, random coils and α-helices. PoC increased the relative β-sheet content from an initial value of 49.55% for CPoPc to 52.13%, 51.42% and 52.58% for CSPoCo70/30, CSPoCo50/50 and CSPoCo30/70, respectively; however, these values were lower than that of 60.96% obtained for SPoPc. In contrast, the α-helix content in CSPoCos was not significantly (p > 0.05) different from that of CPoPc. In the case of β-turns, PoC significantly reduced (p < 0.05) its content in the range of 4.86% (CSPoCo50/50) to 25.0% (CSPoCo70/30) compared to CPoPc. The reason for these changes could be attributed to the proportion of different protein sources in PoCos. This composition induces a new equilibrium in the secondary structures by modifying molecular interactions, such as hydrogen bonds, disulfide bonds, and hydrophobic and van der Waals forces. Consequently, these structural transformations are linked to the enhancement of PoSo, WaHC, OiHC, LaGeCo, EAcIn, FmC, and FmS (Fig. 2). Tan et al.,11 Tian et al.,12 and Wang et al.78 found that the PoC of proteins from soybean-tilapia, soybean-wheat, and soybean-rice, respectively, modified the secondary structure in contrast to individual protein sources, as found in this study.
4 Conclusions
PoC of canola and sunflower proteins by alkaline extraction-isoelectric precipitation altered their physicochemical, functional and structural characteristics compared to CPoPc and SPoPc. The protein content, ρb, ρc, CaIn, HaRa, WaHC, OiHC, LaGeCo, EAcIn, FmC, FmS, SuHo, and IFS values of CSPoCos increased with respect to original protein source, while their Pz and EStIn values decreased. In general, the moisture, L*, aw, Trb, MoFl, ζ-potential, IVPoDi, IViAc, Al, Gx, Pr, SHFr, SHTo, S–SBr, α-helix, β-sheet, and β-turn values varied between those corresponding to CPoPc and SPoPc. SDS-PAGE results revealed the characteristic bands of the two coprecipitated protein sources, in addition to the appearance of a new band, indicating an interaction between the two polymers. The improvement in the functional characteristics of CSPoCos was due to the unfolding and interactions of the canola and sunflower polypeptide chains, giving rise to new protein structures. Specifically, the values of PoSo, WaHC, FmC and FmS were higher in CSPoCo50/50 compared to the other CSPoCos. Thus, PoC proved to be an effective method for modifying canola and sunflower proteins, which could considerably improve the functional quality of plant proteins, making them a good alternative to animal proteins as food ingredients. Further research into the nutritional and sensory characteristics of CSPoCos could expand their use in the production of functional foods.Author contributions
Nitzia Thalía Flores-Jiménez: investigation; methodology; writing – original draft preparation; editing. José Armando Ulloa: investigation; writing – original draft; review; editing; conceptualization, supervision; data curation; funding acquisition. Judith Esmeralda Urías-Silvas: software; resources; supervision; validation.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this manuscript.Data availability
Data will be made available on request.Acknowledgements
The authors thank the Secretariat of Science, Humanities, Technology and Innovation (SECIHTI) of Mexico for the postdoctoral scholarship awarded to Nitzia Thalía Flores Jiménez (CVU: 601948), and the Patronage to Administer the Special Tax Destined to the Autonomous University of Nayarit (UAN) for the financial support.References
- F. Arrutia, E. Binner, P. Williams and K. W. Waldron, Oilseeds beyond oil: Press cakes and meals supplying global protein requirements, Trends Food Sci. Technol., 2020, 100, 88–102, DOI:10.1016/j.tifs.2020.03.044.
- A. Görgüç, C. Bircan and F. M. Yilmaz, Sesame bran as an unexploited by-product: Effect of enzyme and ultrasound-assisted extraction on the recovery of protein and antioxidant compounds, Food Chem., 2019, 283, 637–645, DOI:10.1016/j.foodchem.2019.01.077.
- M. Aider, M. Ndiaye and A. Karim, Optimization of canola meal bleaching by hydrogen peroxide, protein extraction and characterization of their functional properties, Future Foods, 2023, 8, e100282, DOI:10.1016/j.fufo.2023.100282.
- H.-M. Liu, X.- Y Liu, Y.-Y. Yan, J.-H. Gao, Z. Qin and X.-D. Wang, Structural properties and antioxidant activities of polysaccharides isolated from sunflower meal after oil extraction, Arab. J. Chem., 2021, 14, e103420, DOI:10.1016/j.arabjc.2021.103420.
- FAOSTAT, FAO Statistical Database, 2025, https://openknowledge.fao.org/bitstreams/f8c73abe-d26d-4d47-9272-c9b464cc0fc1/download, accessed 05 May 2025 Search PubMed.
- T. D. Alexandrino, R. A. Ferrari, L. M. de Oliveira, R. de Cássia and M. T. B. Pacheco, Fractioning of the sunflower flour components: Physical, chemical and nutritional evaluation of the fractions, Food Sci. Technol., 2017, 84, 426–432, DOI:10.1016/j.lwt.2017.05.062.
- C. Cháirez-Jiménez, C. Castro-López, S. Serna-Saldívar and C. Chuck-Hernández, Partial characterization of canola (Brassica napus L.) protein isolates as affected by extraction and purification methods, Heliyon, 2023, 9, e21938, DOI:10.1016/j.heliyon.2023.e21938.
- R. Singh, S. Langyan, S. Sangwan, B. Rohtagi, A. Khandelwal and M. Shrivastava, Protein for human consumption from oilseed cakes: a review, Front. Sustain. Food Syst., 2022, 6, e856401, DOI:10.3389/fsufs.2022.856401.
- H. T. Kristensen, Q. Denon, I. Tavernier, S. B. Gregersen, M. Hammershøj, P. Van der Meeren, K. Dewettinck and T. K. Dalsgaard, Improved food functional properties of pea protein isolate in blends and co-precipitates with whey protein isolate, Food Hydrocoll., 2021, 113, e106556, DOI:10.1016/j.foodhyd.2020.106556.
- R. Wang, T. Wang, W. Feng, Q. Wang and T. Wang, Rice proteins and cod proteins forming shared microstructures with enhanced functional and nutritional properties, Food Chem., 2021, 354, e129520, DOI:10.1016/j.foodchem.2021.129520.
- L. Tan, P. Hong, P. Yang, C. Zhou, D. Xiao and T. Zhong, Correlation between the water solubility and secondary structure of tilapia-soybean protein co-precipitates, Molecules, 2019, 24, e4337, DOI:10.3390/molecules24234337.
- T. Tian, X. Tong, K. Ren, J. Cao, Y. Yuan, J. Yang, J. Zhu, L. Miao, S. Yang, A. Yu, H. Wang and L. Jiang, Influence of protein ratios on the structure and gel properties of soybean-wheat co-precipitated proteins, Food Sci. Technol., 2022, 170, e114045, DOI:10.1016/j.lwt.2022.114045.
- D. Wu, W. Wu, N. Zhang, O. P. Soladoye, R. E. Aluko, Y. Zhang and Y. Fu, Tailoring soy protein/corn zein mixture by limited enzymatic hydrolysis to improve digestibility and functionality, Food Chem: X, 2024, 23, 101550, DOI:10.1016/j.fochx.2024.101550.
- N. T. Flores-Jiménez, J. A. Ulloa and J. E. Urías-Silvas, Techno-functional and structural characteristics of coprecipitates and protein mixtures: A Review, J. Food Sci., 2025, 90, e70569, DOI:10.1111/1750-3841.70569.
- J. He, R. Wang, W. Feng, Z. Chen and T. Wang, Design of novel edible hydrocolloids by structural interplays between wheat gluten proteins and soy protein isolates, Food Hydrocoll., 2020, 100, e105395, DOI:10.1016/j.foodhyd.2019.105395.
- H. T. Kristensen, A. H. Møller, M. Christense, M. S. Hansen, M. Hammershøj and T. K. Dalsgaard, Protein-protein interaction of a whey-pea protein co-precipitate, Int. J. Food Sci. Technol., 2021, 56, 5777–5790, DOI:10.1111/ijfs.15165.
- C. G. Soria-Hernández, S. O. Serna-Saldívar and C. Chuck-Hernández, Comparison of physicochemical, functional and nutritional properties between proteins of soybean and a novel mixture of soybean-maize, Appl. Sci., 2020, 10, e6998, DOI:10.3390/app10196998.
- M. H. Alu’datt, D. G. Al-U’datt, C. C. Tranchant, M. N. Alhamad, T. Rababah, S. Gammoh, A. Almajwal and I. Alli, Phenolic and protein contents of differently prepared protein co-precipitates from flaxseed and soybean and antioxidant activity and angiotensin inhibitory activity of their phenolic fractions, NFSJ, 2020, 21, 65–72, DOI:10.1016/j.nfs.2020.11.001.
- X. Zhou, C. Zhang, L. Zhao, W. Cao, C. Zhou, X. Xie and Y. Chen, Functionality of pea-grass carp co-precipitated dual-protein as affected by extraction pH, Foods, 2022, 11, e3136, DOI:10.3390/foods11193136.
- H. T. Kristensen, M. Christensen, M. S. Hansen, M. Hammershøj and T. K. Dalsgaard, Mechanisms behind protein-protein interactions in a β-lg-legumin co-precipitate, Food Chem., 2022, 373, e131509, DOI:10.1016/j.foodchem.2021.131509.
- X. Zhou, C. Zhang, W. Cao, C. Zhou, H. Zheng and L. Zhao, A comparative functional analysis of pea protein and grass carp protein mixture via blending and co-precipitation, Foods, 2021, 10, e3037, DOI:10.3390/foods10123037.
- H. Zhang, H. Wang, H. Zhou, J. Shi, Z. Wan, G. Li and M. Yan, Synergistic effect in the co-extraction of Ginseng and Schisandra protein, Front. Nutr., 2024, 11, 1482125, DOI:10.3389/fnut.2024.1482125.
- AOAC, Official Methods of AOAC International, AOAC International, Gaithersburg, MD, 21th edn, 2019 Search PubMed.
- N. T. Flores-Jiménez, J. A. Ulloa, J. E. Urías-Silvas, J. C. Ramírez-Ramírez, P. U. Bautista-Rosales and R. Gutiérrez-Leyva, Influence of high-intensity ultrasound on physicochemical and functional properties of a guamuchil Pithecellobium dulce (Roxb.) seed protein isolate, Ultrason. Sonochem., 2022, 84, e105976, DOI:10.1016/j.ultsonch.2022.105976.
- S. N. Serrano-Sandoval, D. Guardado-Félix and J. A. Gutiérrez-Uribe, Changes in digestibility of proteins from chickpeas (Cicer arietinum L.) germinated in presence of selenium and antioxidant capacity of hydrolysates, Food Chem., 2019, 285, 290–295, DOI:10.1016/j.foodchem.2019.01.137.
- M. Bradford, A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding, Anal. Biochem., 1976, 72, 248–254, DOI:10.1016/0003-2697(76)90527-3.
- I. Balderas-León, A. Cardador-Martínez, D. K. Baigts-Allende, C. Velázquez-Carriles and J. M. Silva-Jara, Sustainable approach from underutilized Leucaena leucocephala biomass by polyphenols composition and protein functional properties assessment, Chem. Pap., 2024, 78, 5513–5525, DOI:10.1007/s11696-024-03492-5.
- K. U. López-Mártir, J. A. Ulloa, J. E. Urías-Silvas, P. Rosas-Ulloa, J. C. Ramírez-Ramírez and J. A. Resendiz-Vazquez, Modification of the physicochemical, functional, biochemical and structural properties of a soursop seed (Annona muricata L.) protein isolate treated with high-intensity ultrasound, Ultrason. Sonochem., 2024, 105, e106870, DOI:10.1016/j.ultsonch.2024.106870.
- G. Briceño-Islas, M. Estarrón-Espinosa and J. E. Urías-Silvas, Revalorization of coconut from Guerrero, Mexico: isolation and characterization of oils and proteins, ACS Food Sci. Technol., 2023, 3, 648–657, DOI:10.1021/acsfoodscitech.2c00410.
- N. T. Flores-Jiménez, J. A. Ulloa and J. E. Urías-Silvas, Assessment of the physicochemical, functional and structural characteristics of a defatted flour from guamuchil (Pithecellobium dulce (Roxb.) seeds, Fut. Foods, 2024, 9, e100351, DOI:10.1016/j.fufo.2024.100351.
- N. Mesfin, A. Belay and E. Amare, Effect of germination, roasting, and variety on physicochemical, techno-functional, and antioxidant properties of chickpea (Cicer arietinum L.) protein isolate powder, Heliyon, 2021, 7, e08081, DOI:10.1016/j.heliyon.2021.e08081.
- A. E. Rodríguez-Rivera, J. A. Ulloa, J. E. Silvas Urías, J. C. Ramírez Ramírez and J. A. Resendíz Vazquez, Physicochemical, techno-functional, biochemical and structural characterization of a protein isolate from groundnut (Arachis hypogaea L.) paste treated with high-intensity ultrasound, Food Chem., 2025, 464, e141848, DOI:10.1016/j.foodchem.2024.141848.
- G. Briceño-Islas, L. Mojica and J. E. Urías-Silvas, Functional chia (Salvia hispanica L.) co-product protein hydrolysate: An analysis of biochemical, antidiabetic, antioxidant potential and physicochemical properties, Food Chem., 2024, 460, e140406, DOI:10.1016/j.foodchem.2024.140406.
- D. Wu, C. Wu, Z. Wang, F. Fan, H. Chen, W. Ma and M. Du, Effects of high pressure homogenize treatment on the physicochemical and emulsifying properties of proteins from scallop (Chlamys farreri), Food Hydrocoll., 2019, 94, 537–545, 10.1039/c9fo00560a.
- X. E. Ren, C. Li, F. Yang, Y. Huang, C. Huang, K. Zhang and L. Yan, Comparison of hydrodynamic and ultrasonic cavitation effects on soy protein isolate functionality, J. Food Eng., 2020, 265, e109697, DOI:10.1016/j.jfoodeng.2019.109697.
- C. Wintersohle, I. Kracke, L. M. Ignatzy, L. Etzbach and U. Schweiggert-Weisz, Physicochemical and chemical properties of mung bean protein isolate affected by the isolation procedure, Curr. Res. Food Sci., 2023, 7, e100582, DOI:10.1016/j.crfs.2023.100582.
- B. K. Mintah, R. He, A. A. Agyekum, M. Dabbour, M. K. Golly and H. Ma, Edible insect protein for food applications: Extraction, composition, and functional properties, J. Food Process Eng., 2020, 43, e13362, DOI:10.1111/jfpe.13362.
- S. Albe-Slabi, O. Mesieres, C. Mathé, M. Ndiaye, O. Galet and R. Kapel, Combined effect of extraction and purification conditions on yield, composition and functional and structural properties of lupin proteins, Foods, 2022, 11, e1646, DOI:10.3390/foods11111646.
- D. Lazdiņa, D. Segliņa, Z. A. Zvaigzne, A. Butlers and I. Ciproviča, Effect of defatting method on japanese quince (Chaenomeles japonica) fruit seed protein isolate technological properties, Foods, 2025, 14, e234, DOI:10.3390/foods14020234.
- M. H. Alu'datt, S. Gammoh, T. Rababah, M. Almomani, M. N. Alhamad, K. Ereifej and S. Abou Nasser, Preparation, characterization, nanostructures and bio functional analysis of sonicated protein co-precipitates from brewers' spent grain and soybean flour, Food Chem., 2018, 240, 784–798, DOI:10.1016/j.foodchem.2017.08.015.
- J. M. Al-saadi and H. C. Deeth, Preparation and functional properties of protein coprecipitate from sheep milk, Int. J. Dairy Technol., 2011, 64, 461–466, DOI:10.1111/j.1471-0307.2011.00701.x.
- N. T. Flores-Jiménez, J. A. Ulloa, J. E. Urías-Silvas, J. C. Ramírez-Ramírez, P. Rosas-Ulloa, P. U. Bautista-Rosales, Y. Silva-Carrillo and R. Gutiérrez-Leyva, Effect of high-intensity ultrasound on the compositional, physicochemical, biochemical, functional and structural properties of canola (Brassica napus L.) protein isolate, Food Res. Int., 2019, 121, 947–956, DOI:10.1016/j.foodres.2019.01.025.
- B. G. Subaşı, F. Casanova, E. Capanoglu, F. Ajalloueian, J. J. Sloth and M. A. Mohammadifar, Protein extracts from de-oiled sunflower cake: Structural, physico-chemical and functional properties after removal of phenolics, Food Biosci., 2020, 38, e100749, DOI:10.1016/j.fbio.2020.100749.
- R. Suhag, A. Kellil and M. Razem, Factors influencing food powder flowability, Powders, 2024, 3, 65–76, DOI:10.3390/powders3010006.
- H. Ding, B. Li, I. Boiarkina, D. I. Wilson, W. Yu and B. R. Young, Effects of morphology on the bulk density of instant whole milk powder, Foods, 2020, 9, e1024, DOI:10.3390/foods9081024.
- A. Averardi, C. Cola, S. E. Zeltmann and N. Gupta, Effect of particle size distribution on the packing of powder beds: A critical discussion relevant to additive manufacturing, Mater. Today Commun., 2020, 24, e100964, DOI:10.1016/j.mtcomm.2020.100964.
- D. S. Shah, K. K. Moravkar, D. K. Jha, V. Lonkar, P. D. Amin and S. S. Chalikwar, A concise summary of powder processing methodologies for flow enhancement, Heliyon, 2023, 9, e16498, DOI:10.1016/j.heliyon.2023.e16498.
- J. S. Weaver, J. Whiting, V. Tondare, C. Beauchamp, M. Peltz, J. Tarr, T. Q. Phan and M. A. Donmez, The effects of particle size distribution on the rheological properties of the powder and the mechanical properties of additively manufactured 17-4 PH stainless steel, Addit. Manuf., 2021, 39, e101851, DOI:10.1016/j.addma.2021.101851.
- L. Červenka, M. Frühbauerová and H. Velichová, Functional properties of muffin as affected by substituing wheat flour with carob powder, Potr. S. J. Food Sci., 2019, 13, 212–217, DOI:10.5219/1033.
- Y. Silva-Carrillo, J. A. Ulloa, J. E. Silvas, J. C. Ramírez Ramírez and R. Gutierrez Leyva, Physicochemical and functional characteristics of a gourd (Cucurbita argyrosperma Huber) seed protein isolate subjected to high-intensity ultrasound, Heliyon, 2024, 10, e32225, DOI:10.1016/j.heliyon.2024.e32225.
- R. Zhao, M. So, H. Maat, N. J. Ray, F. Arisaka, Y. Goto, J. A. Carver and D. Hall, Measurement of amyloid formation by turbidity assay-seeing through the cloud, Biophys. Rev., 2016, 8, 445–471, DOI:10.1007/s12551-016-0233-7.
- M. A. Malik, H. K. Sharma and C. S. Saini, High intensity ultrasound treatment of protein isolate extracted from dephenolized sunflower meal: Effect on physicochemical and functional properties, Ultrason. Sonochem., 2017, 39, 511–519, DOI:10.1016/j.ultsonch.2017.05.026.
- R. Pandiselvam, S. Mitharwal, P. Rani, M. A. Shanker, A. Kumar, R. Aslam, Y. T. Barut, A. Kothakota, S. Rustagi, D. Bhati, S. A. Siddiqui, M. W. Siddiqui, S. Ramniwas, A. Aliyeva and A. M. Khaneghah, The influence of non-thermal technologies on color pigments of food materials: An updated review, Curr. Res. Food Sci., 2023, 6, e100529, DOI:10.1016/j.crfs.2023.100529.
- M. Naik, V. Natarajan, N. Modupalli, S. Thangaraj and A. Rawson, Pulsed ultrasound assisted extraction of protein from defatted Bitter melon seeds (Momardica charantia L.) meal: Kinetics and quality measurements, Food Sci. Technol., 2022, 155, e112997, DOI:10.1016/j.lwt.2021.112997.
- K. U. López-Mártir, J. A. Ulloa, J. E. Urías-Silvas, P. Rosas-Ulloa and B. E. Ulloa-Rangel, High-intensity ultrasound affects the physicochemical, structural and functional properties of proteins recovered from noni (Morinda citrifolia) seeds, Sustain. Food Technol., 2025, 3, 700–713, 10.1039/D4FB00321G.
- P. Rosas-Ulloa, J. A. Ulloa, B. E. Ulloa Rangel and K. U. López Mártir, Protein isolate from orange (Citrus sinensis L.) seeds: Effect of high-intensity ultrasound on its physicochemical and functional properties, Food Bioprocess Technol., 2023, 16, 589–602, DOI:10.1007/s11947-022-02956-4.
- N. Yousefi and S. Abbasi, Food proteins: Solubility & thermal stability improvement techniques, Food Chem. Adv., 2022, 1, e100090, DOI:10.1016/j.focha.2022.100090.
- L. Miron, G. Montevecchi, G. Bruggeman, L. I. Macavei, L. Maistrello, A. Antonelli and M. Thomas, Functional properties and essential amino acid composition of proteins extracted from black soldier fly larvae reared on canteen leftovers, Innov. Food Sci. Emerg. Technol., 2023, 87, e103407, DOI:10.1016/j.ifset.2023.103407.
- S. Guidi, A. F. Formica and C. Denkel, Mixing plant-based proteins: Gel properties of hemp, pea, lentil proteins and their binary mixtures, Food Res. Int., 2022, 161, e111752, DOI:10.1016/j.foodres.2022.111752.
- R. Kaur and G. Ghoshal, Sunflower protein isolates-composition, extraction and functional properties, Adv. Colloid Interface Sci., 2022, 306, e102725, DOI:10.1016/j.cis.2022.102725.
- N. T. Flores-Jiménez, J. A. Ulloa, R. I. Ortiz-Basurto and J. E. Urías-Silvas, Application of high-intensity ultrasound to modify the rheological properties of a guamuchil Pithecellobium dulce (Roxb.) seed protein isolate, Int. J. Food Prop., 2023, 26, 739–751, DOI:10.1080/10942912.2023.2183171.
- H. Niu, Y. Li, J. Han, Q. Liu and B. Kong, Gelation and rheological properties of myofibrillar proteins influenced by the addition of soybean protein isolates subjected to an acidic pH treatment combined with a mild heating, Food Hydrocoll., 2017, 70, 269–276, DOI:10.1016/j.foodhyd.2017.04.001.
- C. Wu, X. Yan, T. Wang, W. Ma, X. Xu and M. Du, A self-sorted gel network formed by heating a mixture of soy and cod proteins, Food Funct., 2019, 10, 5140–5151, 10.1039/c9fo00560a.
- P. Shen, F. Twilt, B. Deng, J. Peng, K. Schroen, L. M. Sagis and J. Landman, Oil-water interface and emulsion stabilization by pulse proteins, Food Hydrocoll., 2025, 163, e111093, DOI:10.1016/j.foodhyd.2025.111093.
- M. S. Awal, S. Benjakul, T. Prodpran and K. Nilsuwan, Characteristics and properties of co-precipitated protein and film based on Bambara groundnut protein isolate and fish skin acid-soluble collagen, J. Agri. Food Res., 2024, 18, e101430, DOI:10.1016/j.jafr.2024.101430.
- A. Joshi, A. S. Virdi, R. Kaur, A. Kumar and N. Singh, Deciphering food proteins: the applications of SDS-PAGE in food science, Food Biosci., 2025, 66, e106126, DOI:10.1016/j.fbio.2025.106126.
- W. Jia, D. S. Sethi, A. J. van der Goot and J. K. Keppler, Covalent and non-covalent modification of sunflower protein with chlorogenic acid: Identifying the critical ratios that affect techno-functionality, Food Hydrocoll., 2022, 131, e107800, DOI:10.1016/j.foodhyd.2022.107800.
- P. Purohit, H. Rawat, N. Verma, S. Mishra, A. Nautiyal, Anshul, S. Bhatt, N. Bisht, K. Aggarwal, A. Bora, H. Kumar, P. Rawal, A. Kumar, R. Kapoor, J. Sehrawat, M. A. Rather, B. Naik, V. Kumar, S. Rustagi, M. S. Preet and A. K. Gupta, Analytical approach to assess anti-nutritional factors of grains and oilseeds: A comprehensive review, J. Agric. Food Res., 2023, 14, e100877, DOI:10.1016/j.jafr.2023.100877.
- N. Qadir and I. A. Wani, Functional properties, antioxidant activity and in-vitro digestibility characteristics of brown and polished rice flours of Indian temperate region, Grain Oil Sci. Technol., 2023, 6, 43–57, DOI:10.1016/j.gaost.2022.12.001.
- G. L. Zabot, E. K. Silva, L. B. Emerick, M. H. F. Felisberto, M. T. P. Silva Clerici and M. A. A. Meireles, Physicochemical, morphological, thermal and pasting properties of a novel native starch obtained from annatto seeds, Food Hydrocoll., 2019, 89, 321–329, DOI:10.1016/j.foodhyd.2018.10.041.
- L. Jiang, J. Wang, Y. Li, Z. Wang, J. Liang, R. Wang, Y. Chen, W. Ma, B. Qi and M. Zhang, Effects of ultrasound on the structure and physical properties of black bean protein isolates, Food Res. Int., 2014, 62, 595–601, DOI:10.1016/j.foodres.2014.04.022.
- H. B. Jadhav, Foaming properties of protein-based particles and their mixture with other food-grade particles—A review, Food Biomacromol., 2025, 2, 325–339, DOI:10.1002/fob2.70019.
- H. Jiang, N. Zhang, L. Xie, G. Li, L. Chen and Z. Liao, A comprehensive review of the rehydration of instant powders: Mechanisms, influencing factors, and improvement strategies, Foods, 14, e2883, DOI:10.3390/foods14162883.
- D. R. Kim, Y. Jung, S. J. Rho and Y. R. Kim, Sonication of sesame meal protein isolates modified its microstructural and functional properties, Food Sci. Technol., 2013, 186, e115242, DOI:10.1016/j.lwt.2023.115242.
- S. Yan, J. Xu, S. Zhang and Y. Li, Effects of flexibility and surface hydrophobicity on emulsifying properties: Ultrasound-treated soybean protein isolate, Food Sci. Technol., 2021, 142, e110881, DOI:10.1016/j.lwt.2021.110881.
- P. G. Argudo and J. J. Giner-Casares, Folding and self-assembly of short intrinsically disordered peptides and protein regions, Nanoscale Adv., 2021, 3, 1789–1812, 10.1039/d0na00941e.
- H. Yang, Y. Qu, Y. Su, Y. Liu, T. Chen, H. Wang and Q. Shen, Uncovering the chemical bonding basis for ultrasound treatment-induced improvement in the molecular flexibility of myofibrillar proteins from low-salt meat batters with added methylcellulose, Food Sci. Technol., 2024, 203, e116408, DOI:10.1016/j.lwt.2024.116408.
- T. Wang, M. Yue, P. Xu, R. Wang and Z. Chen, Toward water-solvation of rice proteins via backbone hybridization by casein, Food Chem., 2018, 258, 278–283, DOI:10.1016/j.foodchem.2018.03.084.
- V. M. Pizones Ruiz-Henestrosa, M. J. Martinez, C. C. Sánchez, J. M. Rodríguez Patino and A. M. Pilosof, Mixed soy globulins and β-lactoglobulin systems behaviour in aqueous solutions and at the air–water interface, Food Hydrocoll., 2014, 35, 106–114, DOI:10.1016/j.foodhyd.2013.04.021.
- M. L. Chihi, J. L. Mession, N. Sok and R. Saurel, Heat-induced soluble protein aggregates from mixed pea globulins and β-lactoglobulin, J. Agric. Food Chem., 2016, 64, 2780–2791, DOI:10.1021/acs.jafc.6b00087.
- J. Choi, C. Fuentes, J. Fransson, M. Wahlgren and L. Nilsson, Separation and zeta-potential determination of proteins and their oligomers using electrical asymmetrical flow field-flow fractionation (EAF4), J. Chromatogr. A., 2020, 1633, e461625, DOI:10.1016/j.chroma.2020.461625.
| This journal is © The Royal Society of Chemistry 2026 |