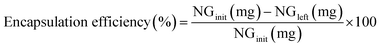Open Access Article
Open Access ArticlePectin-encapsulated amorphous naringenin with enhanced aqueous solubility prepared by simple coacervation and precipitation
Weerapha
Panatdasirisuk
a,
Weerachai
Chantana
ab,
Adisak
Hawan
a,
Phimonwan
Wongjeen
a and
Verawat
Champreda
 *a
*a
aNational Center for Genetic Engineering and Biotechnology (BIOTEC), National Science and Technology Development Agency (NSTDA), Khlong Nueng, Khlong Luang District, Pathum Thani 12120, Thailand. E-mail: verawat@biotec.or.th; Tel: +66-2564-6700 (ext. 3446)
bMolecular Medicine, Multidisciplinary Program, Mahidol University Rama VI Road, Ratchathewi District, Bangkok 10400, Thailand
First published on 5th March 2026
Abstract
This study presents a sustainable approach for enhancing the solubility and processability of naringenin, a poorly water-soluble bioactive flavonoid, through fruit peel-inspired encapsulation. Amorphous naringenin nanoparticles were first prepared by anti-solvent precipitation using polyvinylpyrrolidone (PVP) as a crystallization inhibitor. Subsequently, ethanol-induced coacervation with pectin, a biopolymer abundant in citrus peel, was employed to encapsulate and recover the nanoparticles under ambient conditions, avoiding energy-intensive drying methods. The process yielded microcapsules with significantly reduced crystallinity, as confirmed by X-ray diffraction, and improved dissolution behavior in both deionized water and simulated intestinal fluid compared to pristine naringenin. High-methoxy pectin demonstrated superior encapsulation efficiency (up to 76%) and produced more compact and spherical microcapsules than low-methoxy pectin, highlighting the influence of pectin structure on encapsulation performance. This simple, low-energy, and scalable technique not only improves the functional properties of naringenin but also valorizes pectin from fruit by-products, providing a sustainable strategy for the development of functional food ingredients.
Sustainability spotlightPoor water solubility limits the bioavailability and functional application of many natural flavonoids such as naringenin. This work presents an environmentally friendly strategy to overcome this challenge by employing pectin-based encapsulation derived from fruit peel by-products. The process uses a mild, solvent-driven coacervation and precipitation method under ambient conditions, avoiding energy-intensive drying and synthetic polymers. This green and scalable approach not only improves ingredient performance but also valorizes agricultural residues and reduces chemical waste. The work supports the principles of resource efficiency and circular economy, aligning with the United Nations Sustainable Development Goals (SDG 9: Industry, Innovation and Infrastructure; SDG 12: Responsible Consumption and Production; SDG 13: Climate Action). |
1. Introduction
Fruit peels are widely available agricultural by-products generated in large quantities during fruit processing, accounting for 20–50% of the total fruit weight depending on the type. While often discarded, they are rich in bioactive compounds such as flavonoids, pectin, and phenolic acids, with flavonoid levels frequently higher than in the pulp or juice, making them a valuable but underutilized source for food and health-related applications.1,2Among these phytochemicals, flavonoids like naringin and its aglycone, naringenin, are notable for their antioxidant, anti-inflammatory, and cardioprotective effects.3,4 However, despite their pharmacological potential, their application is often limited by poor oral bioavailability. Naringin is poorly absorbed through the gastrointestinal tract and is typically hydrolyzed to naringenin, which itself has low solubility and limited absorption, with only about 15% being bioavailable.4,5
Various strategies have been developed to improve flavonoid solubility and bioavailability, including nanocrystallization to reduce particle size and solid dispersion to lower crystallinity. Encapsulation further enhances solubility and protects flavonoids from environmental stressors, while enabling controlled release.6 A variety of microencapsulation methods such as spray drying, freeze drying, coacervation, liposomes, ionic gelation, and emulsions have been employed, each with distinct advantages and limitations.6–11
A previous study explored a simple method for enhancing naringenin solubility, with solid dispersion selected for its ability to reduce crystallinity, a major barrier to dissolution, while requiring fewer excipients and simpler preparation compared to other techniques.12 The work demonstrated that dissolving a polymer–flavonoid mixture (PVP/naringenin–hesperetin, 80/20 w/w) in ethanol followed by solvent evaporation yielded a solid dispersion with significantly reduced crystallinity and markedly improved solubility in phosphate buffer (pH 6.8) containing 2% Tween 20.12 In this context, solvent evaporation plays a critical role in obtaining the solid product, with techniques such as spray drying or freeze-drying commonly employed. However, these methods are energy-intensive, requiring either high heat or reduced pressure. Alternative approaches such as precipitation or coagulation may be considered to remove excess solvent prior to drying. In this study, we explore the potential of pectin, a naturally abundant polymer found in fruit peels as a low-cost, functional encapsulation material.
Pectin offers multiple functional benefits, including emulsification, gelling, and binding, and has been used in encapsulation via techniques like spray drying and complex coacervation13–18 With citrus peel containing up to 30% pectin on a dry basis,19–21 extracted pectin aligns well with circular economy strategies. As an encapsulation matrix, it can enhance the stability and expand the applicability of sensitive bioactive compounds.6,22
In this work, naringenin was prepared as an amorphous nanoparticle using PVP as a crystallization inhibitor. The process involved dissolving naringenin and PVP in ethanol, followed by introducing the solution into water as a non-solvent. Pectin was added to the aqueous phase, resulting in the spontaneous formation of encapsulated naringenin. The encapsulation efficiency of pectin and its ability to coagulate naringenin nanoparticles were evaluated to facilitate their separation prior to drying. The final product was characterized in terms of particle size, crystallinity, and morphology, while its dissolution behavior was assessed in both deionized water and simulated intestinal fluid.
2. Materials and methods
2.1 Materials
Naringenin (90%), pectin (high ester, DE 65–68%, from apple), pectin (low ester, DE 30–35%, from apple), and bile salt from pig were purchased from Myskinrecipes, Thailand. Ethanol (95%) was purchased from RCI Labscan, Thailand. KCl, KH2PO4, NaHCO3, MgCl2 and (NH4)2CO3, NaCl, CaCl2 were purchased from Carlo Erba, Italy.2.2 Solubility test
Naringenin solubility was evaluated in 95% ethanol and deionized water. Initially, 10 mg of naringenin was added to 5 mL of each solvent in separate vials, followed by stirring for 2 hours at room temperature. Additional naringenin was incrementally added until visible precipitation occurred, indicating saturation. The suspensions were then filtered through a 0.22 µm nylon syringe filter to remove undissolved particles. The resulting clear solutions were dried in an oven at 70 °C overnight to evaporate the solvent. The dried residue was weighed, and solubility was calculated and reported in mg mL−1.2.3 Preparation of naringenin reprecipitate and amorphous naringenin nanoparticle
To reprecipitate naringenin, ethanolic solution of naringenin was prepared by dissolving 100 mg of naringenin in 1 mL of warm 95% ethanol. The naringenin ethanolic solution was instantly dropped into 19 mL DI water. To prepare amorphous naringenin nanoparticle (NGPVP), 10 mg naringenin and 10 mg PVP was dissolved in 1 mL of 95% ethanol. The mixture was then added dropwise into 19 mL DI water. The morphology of the obtained products was characterized using SEM (Hitachi, SU5000). SEM specimens were prepared by dropping the obtained sample onto aluminium foil and then drying it in an oven overnight. The samples were coated using Au sputter coater (Quorum, Q15R S, USA) at sputter current 15 mA, sputter time 80 s. SEM images were acquired at an accelerating voltage of 5 kV with a spot intensity of 15.2.4 Pectin encapsulated naringenin particle preparation
A stock of 2% w/v pectin solution was prepared by dissolving pectin in DI water. The pectin solution was heated at 100 °C and stirred for 2 hours or until a clear solution was obtained, then the solution was cooled down to room temperature. Pectin solutions at concentrations of 0.005–0.2% w/v were prepared, diluted from stock solution. In a separated container, a mixture of 1% w/v naringenin and 1% w/v PVP in 95% ethanol was prepared. Then, 5 mL of ethanolic mixture was dropped into 95 mL of a stirring pectin solution at each concentration. The naringenin core/pectin shell was formed spontaneously and precipitated after the solution was left stand still for 10 min. The supernatant was taken to the naringenin concentration evaluation. The supernatant was diluted using ethanol for complete dissolution of naringenin. The encapsulation efficiency was calculated using an equation below, where NGinitial is the initially added amount of naringenin and NGleft is the amount of naringenin remaining in the supernatant.The encapsulated products were analyzed the particle size, polydispersity index (PDI), using Zetasizer (Nano ZS ZEN3600, Malvern Panalytical). The powder samples were prepared by decanting the supernatant, followed by freeze-drying. The resulting encapsulated products were designated as NGPVP-HM and NGPVP-LM, where HM and LM indicate the use of high-methoxy pectin and low-methoxy pectin, respectively, during the encapsulation process. The powder products were then subjected to X-ray diffractometry and FTIR spectroscopy. The crystallinity was evaluated using X-ray diffractometer (D8 Advance, Bruker, Germany). Powder-XRD measurements were performed using Cu Kα radiation and operating at 2000 W with 30 mA and 40 kV. Samples were measured in the 2θ range of 5–45° at a step interval of 0.02° at ambient temperature. The chemical functionality was evaluated using Attenuated Total Reflectance Fourier-Transform Infrared (ATR-FTIR) spectroscopy, operated at room temperature in the range between 450 and 4000 cm−1.
2.5 Determination of soluble naringenin content
The sample was filtered using a syringe filter with 0.2 µm pore size, then diluted by ethanol and DI water to the appropriate dilution. The final concentration of ethanol was fixed to 50% v/v to prevent further precipitation of naringenin during the measurement. The sample was analyzed using UV-vis spectrophotometer and a quartz cuvette, measuring at 288 nm. The amount of naringenin was calculated from a standard curve prepared from known concentration of naringenin.2.6 Dissolution profile evaluation
The dissolution profiles of the powder samples were evaluated using a dialysis membrane technique. The samples were loaded into dialysis bags with a molecular weight cutoff of 6–8 kDa. The amount of powder introduced into each dialysis bag was adjusted to be equivalent to 5 mg of naringenin. For the encapsulated samples (NGPVP-HM and NGPVP-LM), the actual naringenin content was calculated based on their respective encapsulation efficiencies. In the case of NGPVP, the naringenin content was determined by redissolving the sample in 50% (v/v) ethanol, followed by spectrometric analysis as described in Section 2.5. The dialysis bag was immersed in a 5 mL stirred medium including DI water or simulated intestinal fluid (SIF). The concentration of naringenin outside the dialysis bag was measured at time intervals and plotted as dissolution profile. The stimulated intestinal fluid (SIF) was prepared as follows, the stock salt solution was prepared by mixing 6.8 mL of 37.3 g per L KCl, 0.8 mL of 68 g per L KH2PO4, 42.5 mL of 84 g per L NaHCO3, 9.6 mL of 117 g per L NaCl, 1.1 mL of 30.5 g per L MgCl2·6H2O, and 40 µL of 0.3 M CaCl2. Then the SIF was prepared by mixing of 50 mL of stock salt solution and 80 mg of bile salt.2.7 Statistical analysis
All analyses were performed using Minitab statistical software (Version 22.4.0.0, Minitab Ltd, UK). Data were analyzed by one-way analysis of variance (ANOVA). All results were expressed as mean ± standard deviation (SD) (n = 3). Post hoc multiple comparisons were performed using Tukey's honestly significant difference (HSD) test. Differences were considered statistically significant at p < 0.05.3. Results and discussion
Pristine naringenin exhibited clear crystallinity, as observed by SEM (Fig. 1a and d). Its aqueous solubility was extremely low, at less than 5 µg mL−1. In contrast, naringenin was highly soluble in 95% ethanol, with a solubility of 58.8 ± 1.7 mg mL−1. Consistent with the literature, the water solubility of naringenin has been reported to be similarly low, with values of 2.523 ± 0.062 µg mL−1 reported by Stasiłowicz-Krzemień et al.23 and 1.94 µg mL−1 reported by Nesalin et al.24 | ||
| Fig. 1 SEM images of (a and d) pristine naringenin, (b and e) naringenin precipitate, (c and f) NGPVP precipitate. | ||
Although naringenin solubility is pH-dependent, the extent of improvement remains limited. For example, in buffered solutions at pH 1.2, 6.8, and 7.4, the solubility values were reported to be 2.69, 3.08, and 5.55 µg mL−1, respectively.24 Such poor aqueous solubility is commonly observed in flavonoids, including both aglycone and glucoside forms. For instance, kaempferol exhibits a low water solubility of 0.113 mg mL−1 at 30 °C.25 In comparison, flavonoid glucosides generally show improved water solubility due to the presence of polar sugar moieties. For example, naringin exhibits a solubility of up to 500 µg mL−1,26 while liquiritin has been reported to reach 980 µg mL−1.27 However, despite their higher solubility, glucosides must be enzymatically hydrolyzed into their aglycone forms in the intestine prior to absorption. Overall, the extremely low solubility of flavonoids is a well-recognized and inherent limitation of this class of compounds. This challenge persists even as new flavonoids with promising biological potential, such as liquiritin, hesperidin, or plant extracts derived from citrus peels, roselle calyces, green tea leaves, buckwheat seeds, etc. continue to be identified and reported.28,29
To enhance the solubility of naringenin, a simple and practical approach involving the reduction of crystallinity and particle size was employed. As previously reported, polyvinylpyrrolidone (PVP) has been widely used in the preparation of solid dispersions to improve drug solubility. Kanaze et al.12 demonstrated the successful formation of an amorphous solid dispersion by combining PVP with a flavonoid mixture (naringenin and hesperetin) in ethanol, followed by solvent evaporation. The solubility of the solid-dispersed naringenin–PVP was found to increase by 51.4-fold when dissolved in a buffer medium at pH 6.8. In another study, Kumar and Abraham30 prepared naringenin nanoparticles using an antisolvent nanoprecipitation method, wherein an ethanolic solution of naringenin was added dropwise into an aqueous PVP solution. This process yielded naringenin nanocrystals. Both studies underscore the effectiveness of PVP in enhancing the solubility of poorly water-soluble compounds.
In the present work, we combined the concepts of solid dispersion and nanoprecipitation. A solution of naringenin and PVP in ethanol was prepared and then introduced into water, serving as a non-solvent. This induced rapid precipitation, while the presence of PVP effectively inhibited crystal growth, leading to the formation of nano-sized amorphous naringenin particles (NGPVP). Fig. 1 shows the morphology of the obtained particles with the Fig. 1c and f clearly exhibits round particle without the edge.
Although the amorphous naringenin/PVP nanoparticles (NGPVP) were successfully obtained, they remained as a fine dispersion in the aqueous medium (Fig. 2b). The particle size is too small to be separated by coarse sieve filtration or sedimentation. In addition, conventional drying techniques, such as spray drying or freeze-drying, are energy intensive and may potentially degrade flavonoid compounds. Therefore, an alternative approach to recover the amorphous particles without the use of excessive heat or vacuum was considered.
 | ||
| Fig. 2 Appearance for the precipitation process (a) naringenin precipitate, (b) NGPVP precipitate, (c) NGPVP-HM precipitate, (d) NGPVP-LM precipitate. | ||
To facilitate the separation, a pectin-assisted encapsulation strategy was employed. Pectin, a naturally derived polysaccharide with amphiphilic and gel-forming properties, was introduced into the aqueous phase prior to nanoprecipitation. When the ethanolic solution of NGPVP was added into the pectin solution, spontaneous precipitation was observed (Fig. 2c and d). This behavior is attributed to coacervation, a phase separation phenomenon where macromolecules such as pectin aggregate and encapsulate solutes in a dense, polymer-rich phase.31 The interaction is likely enhanced by ethanol-induced dehydration of pectin chains, promoting the formation of a pectin shell around the hydrophobic NGPVP core. This process is categorized as simple coacervation where only one polymer is involved. It is simpler, suitable for a hydrophobic core, and cost effective compared to complex coacervation where another polymer like protein is introduced to generate ionic interaction. In either coacervation system, pectin plays a crucial role as a wall-forming material due to its natural origin, excellent biocompatibility, and ability to form a protective polymeric matrix.32–35
To optimize encapsulation conditions, the effect of pectin concentration and methoxyl content on encapsulation behavior was investigated. High methoxy (HM) and low methoxy (LM) pectin were tested at varying concentrations (0.1–1% w/v). SEM images (Fig. 3) revealed that both types of pectin were able to induce coalescence and encapsulation of NGPVP particles, with increasing pectin concentration resulting in more defined and compact microcapsules. Notably, high methoxy pectin tended to produce more discrete, spherical particles, whereas low methoxy pectin led to a smeared or aggregated morphology, especially at lower concentrations.
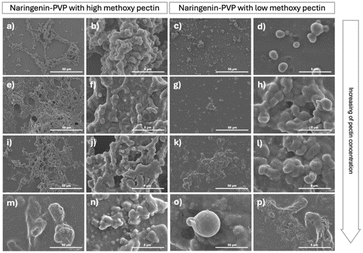 | ||
| Fig. 3 SEM images of pectin encapsulated naringenin using HM pectin and LM pectin at various concentrations (a–d) 0.1% w/v pectin, (e–h) 0.2% w/v pectin, (i–l) 0.4% w/v pectin, (m–p) 1% w/v pectin. | ||
This morphological difference may be attributed to the higher degree of methyl esterification in HM pectin, which renders the polymer more hydrophobic. The increased number of methyl groups could enhance hydrophobic interactions with the naringenin-rich core, promoting more efficient surface adsorption and coalescence into compact particles. In contrast, LM pectin contains more free carboxyl groups, which increase its hydrophilicity and may reduce its affinity toward hydrophobic surfaces, thereby resulting in less defined encapsulation. This behavior aligns with previous studies that reported stronger interactions between hydrophobic bioactive compounds and HM pectin matrices.13 Thus, the degree of methoxylation plays a critical role not only in gelation properties but also in driving interfacial interactions during coacervation-based encapsulation of hydrophobic compounds like naringenin.
From FTIR results (Fig. 4), PVP exhibits a characteristic absorption band at 1651 cm−1, corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration, along with a band at 1289 cm−1 attributed to C–N stretching. Naringenin shows sharp characteristic peaks at 1628 cm−1 and 1600 cm−1, which are assigned to the C
O stretching vibration, along with a band at 1289 cm−1 attributed to C–N stretching. Naringenin shows sharp characteristic peaks at 1628 cm−1 and 1600 cm−1, which are assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of the conjugated carbonyl group and the C
O stretching vibration of the conjugated carbonyl group and the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of the aromatic rings, respectively. Additionally, naringenin exhibits characteristic phenolic C–O stretching vibrations in the fingerprint region at 1156 cm−1, accompanied by several adjacent bands.
C stretching vibrations of the aromatic rings, respectively. Additionally, naringenin exhibits characteristic phenolic C–O stretching vibrations in the fingerprint region at 1156 cm−1, accompanied by several adjacent bands.
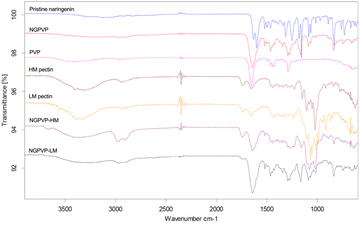 | ||
| Fig. 4 FTIR spectra of powder samples: pristine naringenin (blue), NGPVP (red), PVP (pink), HM pectin (brown), LM pectin (yellow), NGPVP-HM 0.05% (purple), NGPVP-LM 0.05% (black). | ||
For NGPVP, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching band is slightly shifted from 1651 cm−1 to a lower wavenumber at 1644 cm−1, while shoulder peaks around 1628 cm−1 and 1600 cm−1, characteristic of naringenin, remain observable. This shift indicates the presence of intermolecular interactions, most likely hydrogen bonding between naringenin and PVP, whereas the continued presence of naringenin-related bands suggests that its molecular structure remains intact. Similar shifts arising from naringenin–PVP interactions have been reported in PVP–naringenin nanoparticles by Kumar et al.30 and in PVP-based solid dispersions by Kanaze et al.12
O stretching band is slightly shifted from 1651 cm−1 to a lower wavenumber at 1644 cm−1, while shoulder peaks around 1628 cm−1 and 1600 cm−1, characteristic of naringenin, remain observable. This shift indicates the presence of intermolecular interactions, most likely hydrogen bonding between naringenin and PVP, whereas the continued presence of naringenin-related bands suggests that its molecular structure remains intact. Similar shifts arising from naringenin–PVP interactions have been reported in PVP–naringenin nanoparticles by Kumar et al.30 and in PVP-based solid dispersions by Kanaze et al.12
Pectin exhibited characteristic absorption bands at 1739, 1652, and 1020 cm−1, corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of ester groups, the C
O stretching of ester groups, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of carboxylate groups, and the C–O stretching of the pectin backbone, respectively, in good agreement with the literature.36 For pectin-encapsulated NGPVP, the FTIR spectra reflected the superposition of the spectral features of each component. It should be noted that the relative peak intensities were influenced by the amount of each component in the formulation. In the encapsulated samples, 50 mg of naringenin and 50 mg of PVP were used, while the amount of pectin was calculated to be 4.7, 9.5, 19, 47.5, 95, and 190 mg for the process pectin concentrations of 0.005%, 0.01%, 0.02%, 0.05%, 0.1% and 0.2% w/v, respectively.
O stretching of carboxylate groups, and the C–O stretching of the pectin backbone, respectively, in good agreement with the literature.36 For pectin-encapsulated NGPVP, the FTIR spectra reflected the superposition of the spectral features of each component. It should be noted that the relative peak intensities were influenced by the amount of each component in the formulation. In the encapsulated samples, 50 mg of naringenin and 50 mg of PVP were used, while the amount of pectin was calculated to be 4.7, 9.5, 19, 47.5, 95, and 190 mg for the process pectin concentrations of 0.005%, 0.01%, 0.02%, 0.05%, 0.1% and 0.2% w/v, respectively.
For the FTIR results of encapsulated naringenin (0.05% pectin), the characteristic band of NGPVP at 1643 cm−1 was still observed at the same position, indicating that the addition of pectin did not disrupt the interaction between NG and PVP. However, upon the addition of pectin, for example, in the NGPVP-HM sample at a pectin concentration of 0.05%, this band split into two peaks, with a new band appearing at 1634 cm−1. This newly emerged band can be attributed to the carboxylate groups of pectin. Interestingly, the shift of this band to a slightly lower wavenumber (1634 cm−1) suggests that pectin in the encapsulation system experiences a different chemical environment compared to pectin alone, likely due to interactions with NGPVP and possible formation of a pectin coating layer.
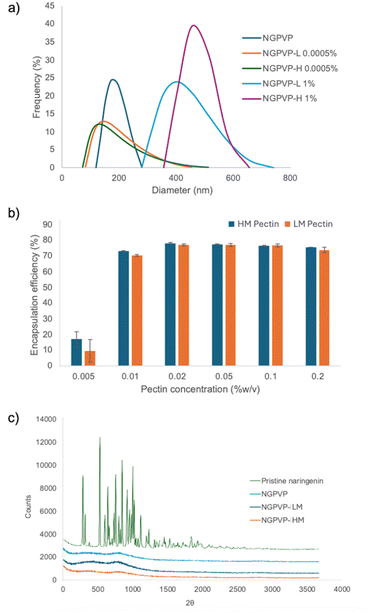
Particle size analysis (Fig. 5a) showed that the bare NGPVP particles exhibited an average hydrodynamic diameter of approximately 200 nm. Upon encapsulation with pectin, the measured particle size increased, supporting the formation of a polymeric shell around the nanoparticle core. However, caution must be taken when interpreting DLS data in this context, particularly under conditions where aggregation or precipitation occurs. At high pectin concentrations (e.g., 1% w/v), rapid precipitation was observed shortly after the addition of the ethanolic NGPVP solution into the pectin-containing aqueous phase. In such cases, the particles tend to sediment quickly, and the DLS measurements predominantly capture the fraction of smaller, suspended particles that remain in solution. Therefore, the reported particle sizes may not represent the full population or average size of the encapsulated material under these conditions. The true size of the larger coacervates or microcapsules formed during precipitation is likely to exceed the detection range of DLS or be lost from measurement due to sedimentation. Interestingly, when a very low concentration of pectin (0.0005% w/v) was used, insufficient to induce visible precipitation, the DLS profile showed a broadened distribution with a distinct tail extending toward larger particle sizes. This suggests that even at sub-gelling concentrations, pectin molecules may adsorb onto the surface of NGPVP particles and increase the hydrodynamic diameter. The gradual size increase observed under these dilute conditions provides indirect evidence of surface interaction between pectin and the naringenin/PVP complex. This may be attributed to weak hydrophobic interactions or hydrogen bonding between the polymer and the flavonoid.
The encapsulation efficiency (Fig. 5b) was found to be strongly dependent on the concentration. At the lowest tested concentration (0.005% w/v), encapsulation was minimal, with HM pectin yielding approximately 20% efficiency and LM pectin only about 10%. This suggests that the amount of pectin available at this level was insufficient to fully coat or aggregate with the dispersed NGPVP particles, resulting in significant loss of naringenin to the supernatant.
A steep increase in encapsulation efficiency was observed as the pectin concentration was increased from 0.005% to 0.01% w/v, suggesting a critical threshold above which sufficient polymer is available to initiate effective coacervation or interfacial complexation. Beyond this threshold, encapsulation efficiency plateaued, reaching values of approximately 76% for both HM and LM pectin in the range of 0.02–0.2% w/v. This plateau indicates that, beyond a certain pectin-to-particle ratio, additional pectin does not contribute significantly to further encapsulation, either due to surface saturation or aggregation-induced steric hindrance that limits further coverage. The plateau values of 78% for NGPVP-HM and 77% for NGPVP-LM are relatively high when compared with the broad range of encapsulation efficiencies reported in the literature (30–90%).37–39 These values are comparable to those obtained from more complex encapsulation techniques, suggesting that the pectin-assisted approach employed in this study provides efficient encapsulation while maintaining a relatively simple and mild processing strategy.
Statistical analysis supported these observations. One-way ANOVA revealed that the encapsulation efficiency increased sharply when the pectin concentration increased from 0.005 to 0.01% (p < 0.05), while no statistically significant improvement was observed at higher concentrations (p > 0.05), indicating saturation of the encapsulation process. One-way ANOVA further confirmed a significant effect of pectin concentration (p < 0.05). However, no statistically significant difference was observed between HM and LM pectin at concentrations ≥0.02% (p > 0.05).
Despite the lack of statistical significance between HM and LM at higher pectin concentrations, a consistent visual trend was observed, in which HM pectin exhibited slightly higher encapsulation efficiency than LM pectin across all concentrations. Mechanistically, this trend may be attributed to the higher degree of methyl esterification in HM pectin, which enhances hydrophobic interactions with the poorly soluble naringenin core. In contrast, the higher content of free carboxyl groups in LM pectin increases solubility and electrostatic repulsion, potentially limiting the formation of condensed interfacial layers around the hydrophobic particle surface. To validate this hypothesis, further studies employing pectin samples with systematically varied degrees of methoxylation would be required.
At the highest concentration tested (0.2% w/v), a slight decline in encapsulation efficiency was observed. This may be attributed to excess pectin molecules forming non-productive aggregates in solution, or to competitive self-association of pectin chains that reduces their availability for particle surface interaction. Similar observations have been reported in polysaccharide-based encapsulation systems, where high polymer concentrations can lead to macroscopic phase separation or entanglement, thus reducing encapsulation efficiency.8,13
The XRD patterns (Fig. 5c) clearly indicate the crystalline nature of pristine naringenin, as evidenced by the presence of numerous sharp and intense diffraction peaks. This crystallinity is significantly reduced upon incorporation with PVP, as shown by the broad and diminished peaks in the NGPVP. The disappearance of sharp diffraction signals suggests that PVP effectively disrupts the crystalline lattice of naringenin, promoting its transformation into an amorphous state. Moreover, samples subjected to further encapsulation, including NGPVP-HM and NGPVP-LM, maintain their amorphous nature, as no sharp peaks reappear in their diffraction profiles. This observation confirms that the encapsulation process, involving a pectin matrix, does not induce recrystallization of naringenin.
Overall, the results can be mechanistically explained as follows. NG and PVP interact to form an amorphous core, while pectin chains subsequently capture these amorphous particles, promoting their agglomeration and precipitation. In the first step, a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NG
1 NG![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PVP ratio is effective in preventing crystallization. For the subsequent encapsulation step, only a small amount of pectin is required, with an NG
PVP ratio is effective in preventing crystallization. For the subsequent encapsulation step, only a small amount of pectin is required, with an NG![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) pectin weight ratio of 4
pectin weight ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 being sufficient.
1 being sufficient.
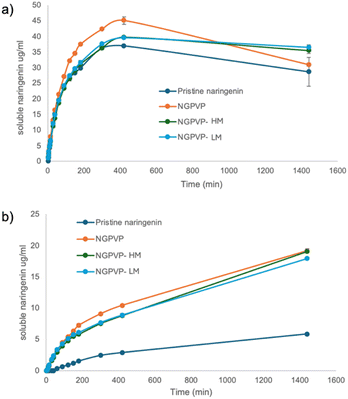
The dissolution profiles of the various naringenin formulations at 37 °C are shown in Fig. 6. Pristine naringenin exhibited poor solubility especially in DI water, consistent with its known low aqueous solubility. The incorporation of PVP significantly enhanced naringenin solubility, as seen in the NGPVP formulation, which achieved the highest maximum soluble concentration in both media. This improvement is attributed to the amorphous state induced by PVP. Interestingly, after further encapsulation with pectin, the NGPVP-HM and NGPVP-LM samples demonstrated slightly reduced initial solubility compared to NGPVP yet still showed substantial improvement over pristine naringenin. This suggests that the pectin layer does not significantly hinder the dissolution capacity of the amorphous naringenin–PVP complex.
In DI water (Fig. 6b), the impact of PVP and pectin on solubility was more pronounced than in SIF (Fig. 5a), possibly because the intestinal fluid itself enhances the solubilization of naringenin to some extent, minimizing the relative difference between formulations.
Notably, after prolonged dissolution (1440 min) in SIF, the NGPVP formulation exhibited a marked decline in solubility, which may be due to reprecipitation or recrystallization of naringenin at high supersaturation levels. In contrast, both NGPVP-HM and NGPVP-LM formulations maintained a more stable solubility over time. One-way ANOVA revealed that at 1440 min (in SIF) the solubility of NGPVP was not different from NG, but the NGPVP-LM, NGPVP-HM were significantly higher than NG group (p < 0.05). This stabilizing effect is likely due to the presence of pectin, which may act as a barrier around naringenin particles, inhibiting recrystallization or aggregation during the dissolution process.
In deionized water (DI), the solubilities of NGPVP, NGPVP-LM, and NGPVP-HM were 19.15, 19.05, and 17.91 µg mL−1, respectively, which are approximately 3–4-fold higher than that of NG (5.83 µg mL−1). At 1440 min, no statistically significant differences were observed among NGPVP, NGPVP-HM, and NGPVP-LM, as determined by one-way ANOVA (p > 0.05). Although this level of enhancement is modest compared to values reported in the literature, substantially higher solubilities have been achieved using other strategies. For example, a phospholipid complex increased the aqueous solubility from 43.83 ± 0.039 to 79.31 ± 0.718 µg mL−1,40 while complexation with hydroxypropyl-β-cyclodextrin (HP-β-CD) enhanced the solubility of naringenin from 4.38 µg mL−1 to as high as 1272.31 µg mL−1.41 In another study, the solubility of naringenin (2.523 ± 0.062 µg mL−1) was improved by nearly 210-fold in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 Naringenin
3 Naringenin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HP-β-CD system.23 A variety of formulation approaches, including liposomes, nanoparticles, self-nanoemulsifying drug delivery systems, nanosuspensions, solid dispersions, and inclusion complexes, have also been extensively reviewed.42
HP-β-CD system.23 A variety of formulation approaches, including liposomes, nanoparticles, self-nanoemulsifying drug delivery systems, nanosuspensions, solid dispersions, and inclusion complexes, have also been extensively reviewed.42
Nevertheless, the performance of a formulation should not be judged solely by the magnitude of solubility enhancement. From an energy saving and process efficiency perspective, the present approach offers a practical advantage, as the product is obtained directly as a precipitate that can be readily recovered and processed into a powder, an aspect that has been comparatively less explored. In practical terms, as demonstrated in this study, out of 100 mL of water in the system, approximately 95 mL can be removed simply by decantation, leaving only about 5 mL to be removed by thermal drying. This implies that roughly 95% of the drying energy can be saved.
In addition, the choice and amount of excipients are critical for real applications; minimizing additive content is generally preferable, especially when food or pharmaceutical grade materials are required. In our system, PVP was used at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio relative to naringenin, while the amount of pectin was four times lower than that of naringenin. Thus, the total additive content in our process is substantially lower than in typical inclusion based approaches, such as cyclodextrin complexation, which usually require at least a 1
1 ratio relative to naringenin, while the amount of pectin was four times lower than that of naringenin. Thus, the total additive content in our process is substantially lower than in typical inclusion based approaches, such as cyclodextrin complexation, which usually require at least a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio, corresponding to approximately five times the mass of naringenin. Nevertheless, further improvement in solubility will still be necessary for our system to better compete with existing techniques. The pectin induced precipitation strategy could also be combined with other solubility enhancement agents, such as surfactants, phospholipids, or other crystallization inhibitors, which will be the subject of future studies.
1 molar ratio, corresponding to approximately five times the mass of naringenin. Nevertheless, further improvement in solubility will still be necessary for our system to better compete with existing techniques. The pectin induced precipitation strategy could also be combined with other solubility enhancement agents, such as surfactants, phospholipids, or other crystallization inhibitors, which will be the subject of future studies.
From an application perspective, the use of this system in functional food formulations will require consideration of additional ingredients and subsequent processing steps to obtain a final product with acceptable stability, sensory properties, and performance. Additionally, the formulation could be evaluated in vivo to determine whether the improvements observed in vitro translate into enhanced bioavailability and biological efficacy, as the behavior in complex biological systems may differ from that observed in the present in vitro experiments. Overall, this study offers a practical and scalable platform for formulating poorly water-soluble flavonoids using food-grade materials, while underscoring the need for further optimization and validation before translation to functional food or pharmaceutical products.
4. Conclusion
This study successfully demonstrated a simple and scalable method for encapsulating naringenin using pectin, inspired by the natural composition of citrus fruit peels. The integration of PVP facilitated the formation of amorphous naringenin nanoparticles with significantly improved solubility compared to pristine crystalline naringenin. The subsequent encapsulation with pectin, particularly high methoxy pectin, effectively promoted the separation of these nanoparticles from the aqueous medium through coacervation, offering an alternative to energy-intensive drying methods. The pectin layer not only aided in the recovery of the encapsulated material but also contributed to maintaining the amorphous state of naringenin and stabilizing its solubility over time. This fruit peel-inspired approach provides a cost-effective and efficient strategy for enhancing the bioavailability and applicability of naringenin and potentially other hydrophobic bioactive compounds, aligning well with circular economy principles by utilizing agricultural by-products.Author contributions
Weerapha Panatdasirisuk: writing – original draft, methodology, validation, data curation, formal analysis, conceptualization, visualization, validation. Weerachai Chantana: methodology, formal analysis, data curation, writing – review & editing. Adisak Hawan: data curation, formal analysis. Phimonwan Wongjeen: data curation, formal analysis. Verawat Champreda: supervision, conceptualization, writing – review & editing, project administration, validation, funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting the findings of this study are fully available within the article.Acknowledgements
This research was supported by the National Science, Research and Innovation Fund, Thailand Science Research and innovation (TSRI) (Grant No. FFB670076/0337) through the National Science and Technology Development Agency (P2351693). WP received funding support from the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation [grant number B13F660064].References
- R. K. Saini, A. Ranjit, K. Sharma, P. Prasad, X. Shang and K. G. M. Gowda, et al., Bioactive Compounds of Citrus Fruits: A Review of Composition and Health Benefits of Carotenoids, Flavonoids, Limonoids, and Terpenes, Antioxidants, 2022, 11(2), 239 CrossRef CAS PubMed.
- B. Singh, J. P. Singh, A. Kaur and M. P. Yadav, Insights into the chemical composition and bioactivities of citrus peel essential oils, Food Res. Int., 2021, 143, 110231 CrossRef CAS PubMed.
- L. H. Yao, Y. M. Jiang, J. Shi, F. A. Tomás-Barberán, N. Datta and R. Singanusong, et al., Flavonoids in food and their health benefits, Plant Foods Hum. Nutr., 2004, 59(3), 113–122 CrossRef CAS PubMed.
- V. S. Shilpa, R. Shams, K. K. Dash, V. K. Pandey, A. H. Dar and M. S. Ayaz, et al., Phytochemical Properties, Extraction, and Pharmacological Benefits of Naringin: A Review, Molecules, 2023, 28(15), 5623 CrossRef CAS PubMed.
- J. Stabrauskiene, D. M. Kopustinskiene, R. Lazauskas and J. Bernatoniene, Naringin and Naringenin: Their Mechanisms of Action and the Potential Anticancer Activities, Biomedicines, 2022, 10(7), 1686 CrossRef CAS PubMed.
- F. Flamminii, M. Paciulli, A. Di Michele, P. Littardi, E. Carini and E. Chiavaro, et al., Alginate-based microparticles structured with different biopolymers and enriched with a phenolic-rich olive leaves extract: A physico-chemical characterization, Curr. Res. Food Sci., 2021, 4, 698–706 CrossRef CAS PubMed.
- G. Ozkan, P. Franco, I. De Marco, J. Xiao and E. Capanoglu, A review of microencapsulation methods for food antioxidants: Principles, advantages, drawbacks and applications, Food Chem., 2019, 272, 494–506 CrossRef CAS PubMed.
- D. J. McClements, Designing biopolymer microgels to encapsulate, protect and deliver bioactive components: Physicochemical aspects, Adv. Colloid Interface Sci., 2017, 240, 31–59 CrossRef CAS PubMed.
- W. Lu, A. L. Kelly and S. Miao, Emulsion-based encapsulation and delivery systems for polyphenols, Trends Food Sci. Technol., 2016, 47, 1–9 CrossRef CAS.
- Z. Fang and B. Bhandari, Encapsulation of polyphenols – A review, Trends Food Sci. Technol., 2010, 21(10), 510–523 CrossRef CAS.
- F. Ardestani, A. Haghighi Asl and A. Rafe, Phase separation and formation of sodium caseinate/pectin complex coacervates: effects of pH on the complexation, Chem. Biol. Technol. Agric., 2022, 9(1), 83 CrossRef CAS.
- F. I. Kanaze, E. Kokkalou, I. Niopas, M. Georgarakis, A. Stergiou and D. Bikiaris, Dissolution enhancement of flavonoids by solid dispersion in PVP and PEG matrixes: A comparative study, J. Appl. Polym. Sci., 2006, 102(1), 460–471 CrossRef CAS.
- A. Rehman, T. Ahmad, R. M. Aadil, M. J. Spotti, A. M. Bakry and I. M. Khan, et al., Pectin polymers as wall materials for the nano-encapsulation of bioactive compounds, Trends Food Sci. Technol., 2019, 90, 35–46 CrossRef CAS.
- M. Yavuz-Düzgün, B. Zeeb, J. Dreher, B. Özçelik and J. Weiss, The Impact of Esterification Degree and Source of Pectins on Complex Coacervation as a Tool to Mask the Bitterness of Potato Protein Isolates, Food Biophys., 2020, 15(3), 376–385 CrossRef.
- D. V. Mendanha, S. E. Molina Ortiz, C. S. Favaro-Trindade, A. Mauri, E. S. Monterrey-Quintero and M. Thomazini, Microencapsulation of casein hydrolysate by complex coacervation with SPI/pectin, Food Res. Int., 2009, 42(8), 1099–1104 CrossRef CAS.
- B. Chen, H. Li, Y. Ding and H. Suo, Formation and microstructural characterization of whey protein isolate/beet pectin coacervations by laccase catalyzed cross-linking, LWT–Food Sci. Technol., 2012, 47(1), 31–38 CrossRef CAS.
- T. Li, B. Wan, R. Jog, A. Costa and D. J. Burgess, Pectin microparticles for peptide delivery: Optimization of spray drying processing, Int. J. Pharm., 2022, 613, 121384 CrossRef CAS PubMed.
- X. Sun, R. G. Cameron, J. A. Manthey, W. B. Hunter and J. Bai, Microencapsulation of tangeretin in a citrus pectin mixture matrix, Foods, 2020, 9(9), 1200 CrossRef CAS PubMed.
- A. H. Rouse and P. G. Crandall, Nitric Acid Extraction of Pectin from citrus peel, Proc. Fla. State Hortic. Soc., 1976, 89, 166–168 CAS.
- M. Khan, N. Bibi and A. Zeb, Optimization of Process Conditions for Pectin Extraction from Citrus Peel, Sci. Technol. Dev., 2015, 34(1), 9–15 CrossRef.
- C. D. May, Industrial pectins: Sources, production and applications, Carbohydr. Polym., 1990, 12(1), 79–99 CrossRef CAS.
- X. Huang, T. Li and S. Li, Encapsulation of vitexin-rhamnoside based on zein/pectin nanoparticles improved its stability and bioavailability, Curr. Res. Food Sci., 2023, 6, 100419 CrossRef CAS PubMed.
- A. Stasiłowicz-Krzemień, M. Gołębiewski, A. Płazińska, W. Płaziński, A. Miklaszewski and M. Żarowski, et al., The Systems of Naringenin with Solubilizers Expand Its Capability to Prevent Neurodegenerative Diseases, Int. J. Mol. Sci., 2022, 23(2), 755 CrossRef PubMed.
- J. A. J. Nesalin, R. S, S. G. N and V. Chandy, Determination of Naringenin Saturation Solubility in Diverse Dissolution Medium Using UV-Visible Spectrophotometric Analysis, Int. J. Res. Publ. Rev., 2024, 5(9), 3405–3410 CrossRef.
- L. A. Jiménez-Ortega, K. Juarez-Moreno, P. Quiñonez-Angulo, J. León-Félix, P. de J. Bastidas-Bastidas and C. Chaidez, et al., Boosting the Solubility and Bioactivity of Flavonoids in Nontoxic Betaine-Based Natural Eutectic Solvents, ACS Sustain. Chem. Eng., 2025, 13(32), 12933–12945 CrossRef.
- M. Chen, R. Li, Y. Gao, Y. Zheng, L. Liao and Y. Cao, et al., Encapsulation of hydrophobic and low-soluble polyphenols into nanoliposomes by ph-driven method: Naringenin and naringin as model compounds, Foods, 2021, 10(5), 963 CrossRef CAS PubMed.
- T. T. H. Nguyen, S. J. Jung, H. K. Kang, Y. M. Kim, Y. H. Moon and M. Kim, et al., Production of rubusoside from stevioside by using a thermostable lactase from Thermus thermophilus and solubility enhancement of liquiritin and teniposide, Enzyme Microb. Technol., 2014, 64–65 Search PubMed.
- L. Zhou, J. Z. Peng and P. Zhou, Liquiritin: A natural flavonoid with potential cardiovascular protection, Ital. J. Food Sci., 2024, 36(2), 216–223 CAS.
- S. A. Sindhu Bharadhi, N. Karthikeyan, A. Thangaiah, R. Balasubramaniam, A. Thiyagarajan and R. Jacob, Active compound analysis of ethanolic extract of roselle calyces (Hibiscus sabdariffa L.), Qual. Assur. Saf. Crop Foods, 2023, 15(2), 117–128 CrossRef.
- R. P. Kumar and A. Abraham, PVP- coated naringenin nanoparticles for biomedical applications – In vivo toxicological evaluations, Chem.-Biol. Interact., 2016, 257, 110–118 CrossRef CAS PubMed.
- W. xin Jiang, J. ru Qi, J. song Liao and Y. X. quan, Acid/ethanol induced pectin gelling and its application in emulsion gel, Food Hydrocolloids, 2021, 118, 106774 CrossRef CAS.
- F. Ardestani, A. Haghighi Asl and A. Rafe, Characterization of caseinate-pectin complex coacervates as a carrier for delivery and controlled-release of saffron extract, Chem. Biol. Technol. Agric., 2024, 11(1), 118 CrossRef CAS.
- H. Jamshidian and A. Rafe, Complex coacervate of wheat germ protein/high methoxy pectin in encapsulation of d-limonene, Chem. Biol. Technol. Agric., 2024, 11(1), 60 CrossRef CAS.
- M. Raei, A. Rafe and F. Shahidi, Rheological and structural characteristics of whey protein-pectin complex coacervates, J. Food Eng., 2018, 228, 25–31 CrossRef CAS.
- M. Raei, F. Shahidi, M. Farhoodi, S. M. Jafari and A. Rafe, Application of whey protein-pectin nano-complex carriers for loading of lactoferrin, Int. J. Biol. Macromol., 2017, 105, 281–291 CrossRef CAS PubMed.
- E. E. Santos, R. C. Amaro, C. C. C. Bustamante, M. H. A. Guerra, L. C. Soares and R. E. S. Froes, Extraction of pectin from agroindustrial residue with an ecofriendly solvent: use of FTIR and chemometrics to differentiate pectins according to degree of methyl esterification, Food Hydrocolloids, 2020, 107, 105921 CrossRef CAS.
- M. R. Smruthi, I. Nallamuthu and T. Anand, A comparative study of optimized naringenin nanoformulations using nano-carriers (PLA/PVA and zein/pectin) for improvement of bioavailability, Food Chem., 2022, 369, 130950 CrossRef CAS PubMed.
- D. K. Takma, S. Bozkurt, M. Koç, F. Korel and H. Ş. Nadeem, Characterization and encapsulation efficiency of zein nanoparticles loaded with chestnut fruit shell, cedar and sweetgum bark extracts, Food Hydrocolloids Health, 2023, 4, 100151 CrossRef CAS.
- Y. Amini, S. Amel Jamehdar, K. Sadri, S. Zare, D. Musavi and M. Tafaghodi, Different methods to determine the encapsulation efficiency of protein in PLGA nanoparticles, Biomed Mater Eng., 2017, 28(6), 613–620 CAS.
- A. Semalty, M. Semalty, D. Singh and M. S. M. Rawat, Preparation and characterization of phospholipid complexes of naringenin for effective drug delivery, J. Inclusion Phenom. Macrocyclic Chem., 2010, 67(3), 253–260 CrossRef CAS.
- J. Wen, B. Liu, E. Yuan, Y. Ma and Y. Zhu, Preparation and physicochemical properties of the complex of naringenin with hydroxypropyl-β-cyclodextrin, Molecules, 2010, 15(6), 4401–4407 CrossRef CAS PubMed.
- R. Joshi, Y. A. Kulkarni and S. Wairkar, Pharmacokinetic, pharmacodynamic and formulations aspects of Naringenin: An update, Life Sci., 2018, 215, 43–56 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |